Translate this page into:
Photodynamic therapy: A special emphasis on nanocarrier-mediated delivery of photosensitizers in antimicrobial therapy
⁎Corresponding author at: Department of Pharmaceutical Sciences, College of Pharmacy and Health Sciences, Ajman University, Ajman P.O. Box 346, United Arab Emirates. s.boddu@ajman.ac.ae (Sai HS. Boddu)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Resistance to antimicrobial drugs is an impending healthcare problem of growing significance. In the post-antibiotic era, there is a huge push to develop new tools for effectively treating bacterial infections. Photodynamic therapy involves the use of a photosensitizer that is activated by the use of light of an appropriate wavelength in the presence of oxygen. This results in the generation of singlet oxygen molecules that can kill the target cells, including cancerous cells and microbial cells. Photodynamic therapy is shown to be effective against parasites, viruses, algae, and bacteria. To achieve high antimicrobial activity, a sufficient concentration of photosensitizer should enter the microbial cells. Generally, photosensitizers tend to aggregate in aqueous environments resulting in the weakening of photochemical activity and lowering their uptake into cells. Nanocarrier systems are shown to be efficient in targeting photosensitizers into microbial cells and improve their therapeutic efficiency by enhancing the internalization of photosensitizers into microbial cells. This review aims to highlight the basic principles of photodynamic therapy with a special emphasis on the use of nanosystems in delivering photosensitizers for improving antimicrobial photodynamic therapy.
Keywords
Antibacterial
Photosensitizer
Photodynamic therapy
Nanocarriers
Antimicrobial drug resistance
1 Introduction
The field of antimicrobial therapy has always been a constant challenge, particularly given the resistance developed towards various antimicrobial agents and a wide variety of pathogens encountered. Every year, approximately 700,000 lives are lost worldwide due to infections related to drug-resistant pathogens (Organization 2019). This number of deaths related to drug-resistant pathogens is expected to grow to 10 million people a year by 2050, according to the estimates made by the government-commissioned O’Neill report (Chokshi et al., 2019). A variety of new antimicrobial regimens are being developed and refined to combat the resistance associated with microbial infections. However, the development of resistance towards antimicrobials occurs by various mechanisms, which include thickening of the outer wall, encoding of new proteins that prevent drug penetration, and onset of mutants deficient in those porin channels that allow the influx of foreign chemicals. Antimicrobial resistance has progressively compromised the treatment results (Jori et al., 2006, Morehead and Scarbrough 2018). An alternative approach that eliminates the development of antimicrobial resistance needs to be cultivated. Antimicrobial photodynamic therapy (aPDT) is considered a promising strategy to alleviate resistance associated with microbial infections. The term ‘photodynamic action’ is used to distinguish photosensitized reactions from physicochemical processes occurring in emulsions of photographic films (Dougherty et al., 1998). Photodynamic therapy (PDT) was originally developed for treating cancers and has achieved regulatory approval in several countries (Brown et al., 2004, Huang 2005, dos Santos et al., 2019). PDT is based on the utilization of a photosensitizer (PS) localized in certain cells, which could be activated by low doses of visible light of an appropriate wavelength. Activation of the PS generates singlet oxygen and free radicals that are cytotoxic to the target cells (Ryskova et al., 2010). PDT is also used in antimicrobial therapy as the development of bacterial resistance toward singlet oxygen and free radicals are highly unlikely (Ghorbani et al., 2018, Magadla et al., 2019). The singlet oxygen has a short half-life and the diffusion of singlet oxygen is limited to a few nanometers; hence, the cytotoxicity is confined to immediate cells. However, the delivery of PS in the vicinity of target cells is challenging (George and Kishen 2007). In this review, we provide comprehensive and updated information on the aPDT, with special emphasis on the use of nanosystems in delivering photosensitizers to microbial cells.
The history of PDT has been nicely summarized by Abdel-kader (Abdel-kader 2016) and Rajesh et al. (Rajesh et al., 2011). The first regulatory approval was received for Photofrin® for the treatment of bladder cancer and successive U.S. approvals for its use in oesophageal cancer and endobronchial non-small cell lung cancer. Further, PDT is also approved for the treatment of conditions such as age-related macular degeneration. Though many substances exhibit photosensitizing properties, very few have made it to clinical trials and are commercially available (Allison et al., 2004). An updated list of commercially approved photosensitizers is presented in Table 1. Over the past few years, PDT was found to be promising against various types of microorganisms, such as Gram-positive and Gram-negative bacteria, fungi viruses, and parasites (Almeida et al., 2020). The chemical structures of commonly used photosensitizers are shown in Fig. 1. Several review articles recapitulated the effectiveness of PDT in treating various cancers of the head and neck, pancreas, brain, lung, prostate, intraperitoneal cavity, breast and skin (Agostinis et al., 2011, dos Santos et al., 2019, Zhao et al., 2021); however, very few review articles highlighted its application in antimicrobial therapy (Rajesh et al., 2011, Sobotta et al., 2019, Anas et al., 2021, Youf et al., 2021, Zhao et al., 2021). This review is mainly focused on PDT and its application in antimicrobial therapy. The use of PDT in conjunction with a nanoparticulate approach to increase photosensitizer concentration in the target cells was emphasized in this review.
Trade Name
Photosensitizer
Clinical application
Photofrin®
Hematoporphyrin derivative (HpD)
Oesophageal cancer, cervical cancer, gastric cancer
Ameluz®
Porphyrin precursor
Actinic keratosis and basal cell carcinoma
Hexvix®
Hexaminolevulinate
Bladder cancer
Foscan®
Temoporfin
Head and neck cancer
AlaCare®
5-aminolevulinic acid, a photosensitizing precursor
Actinic keratosis
Levulan®
Amino Levulinic acid (ALA)
Actinic keratosis
Metvix®
m-ALA
Actinic keratosis, basal cell carcinoma
Visudyne®
Vertiporfin
Wet-age related macular degeneration
Laserphyrin®
Talaporfin sodium
Early centrally located lung cancer
Redaporfin®
Bacteriochlorin
Advanced Head and neck cancer
Photoheme®
HpD
Skin, breast and gastrointestinal cancer
Photochlor®
2-(1-Hexyloxyethyl)-2-devinyl pyropheophorbide-a (HPPH)
Oesophageal cancer
Photolon™
Chlorin e6-PVP
Chlorin e6 polyvinylpyrrolidone, Ce6-PVP
Redaporphine® (LUZ11)
Bacteriochlorin
Biliary tract cancer
List of commonly used photosensitizers.
2 Photodynamic therapy for bacterial diseases
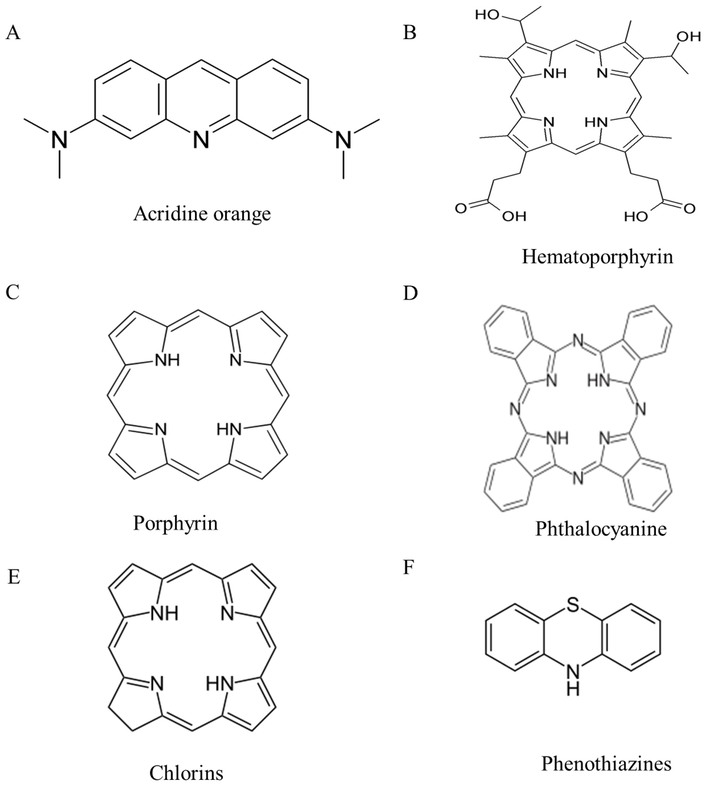
Since the early days of PDT, it was well known that microorganisms can be killed by using a combination of a photosensitizer and harmless visible light. The prerequisite for photosensitization of a microbial cell is the binding/internalization of photosensitizers into the cytoplasmic membrane (Malik et al., 1992). In the 1990 s, researchers reported the differences in susceptibility of Gram-positive bacteria and Gram-negative bacteria towards PDT. The difference in susceptibility is majorly due to the variation in the structural anatomy and physiology of their cell membranes (Fig. 2). Anionic or neutral photosensitizer molecules were found to be internalized efficiently by the Gram-positive bacteria, whereas the Gram-negative bacteria were relatively resistant (Wainwright 1998, Huang et al., 2010). The outer wall of the Gram-positive bacteria consists of a peptidoglycan layer linked to teichuronic acid groups, which exhibit a high level of porosity (Nikaido 1994). This allows large molecules (molecular weight: 30–57 kDa) to readily pass through the cytoplasm (Lambert 2002). Therefore, the Gram-positive bacteria don’t act as a permeability barrier for photosensitizer molecules with a molecular weight less than 1800 Da (Jori et al., 2006). But the outer wall of the Gram-negative bacteria has a heterogeneous composition consisting of peptidoglycan, lipoproteins, and lipopolysaccharides, which are densely packed and give the wall a negative charge (Nikaido 1994). Only very low molecular weight photosensitizer molecules can pass through the cell membrane. Studies have shown that photosensitizer molecules pass through the Gram-negative bacterial membrane when a cationic agent such as polymixin is administered (Malik et al., 1992, Jori and Brown 2004). Some researchers have achieved better penetration by using cationic porphyrins or phthalocyanines (Merchat et al., 1996, Minnock et al., 1996). Recent studies have shown that anionic or neutral photosensitizer molecules can penetrate through the cell membrane when linked to an oligomer (such as poly-lysine), which exists as a cation at the physiological pH (Soukos et al., 1997). Photosensitivity also depends on the physiological state of the bacteria. Cells in the logarithmic phase are found to be more susceptible compared to the cells in the stationary phase (Jori et al., 2006). The differences between the Gram-positive and Gram-negative bacteria are listed in Table 2. Due to differences in the cell structure, photosensitizer molecules should undergo some modifications based on the type of bacteria involved; to achieve sufficient penetration into the bacterial cytoplasm. Unlike antibiotics, photosensitizer upon excitation with light results in a broad spectrum of action on diverse organisms such as bacteria, protozoa, and fungi. Moreover, continuous use of antibiotics might result in an antibiotic resistance pattern; while the bactericidal effect of a photosensitizer is independent of an antibiotic resistance pattern with limited adverse effect profile and damage to the host tissue (Ghorbani et al., 2018). PDT does not exhibit cytotoxicity, phototoxicity and mutagenicity in treated eukaryotic and prokaryotic cells in a wide therapeutic window. A literature search on May 30, 2022, using the United States National Library of Medicine and the National Institutes of Health's PubMed database between the years 2000 and 2021 showed around 4387 articles with the keywords “Antimicrobial Photodynamic Therapy”. There has been a steady increase in the number of publications over the last two decades, which indicates a growing interest in the application of PDT for antimicrobial action (Fig. 3). Moreover, several clinical trials are underway to evaluate the efficacy of PDT to treat diverse infectious diseases, acne vulgaris, dental caries, onychomycosis (tinea cruris, tinea pedis, and interdigital mycoses), HPV infection, and leg ulcers. These are well summarized in a recently published review (Youf et al., 2021).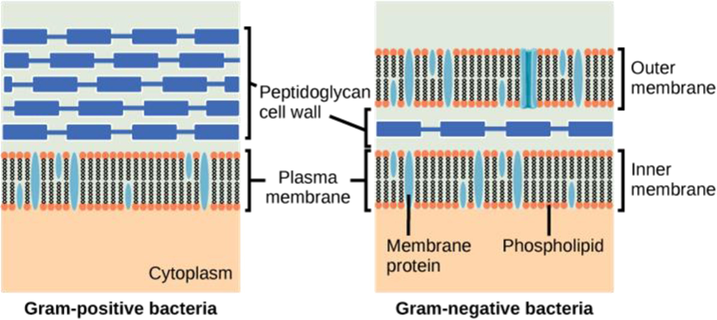
Schematic representation of cell membrane of Gram-positive and Gram-negative bacteria.
Character
Gram positive bacteria
Gram negative bacteria
Nature of cell wall
Rigid
Flexible
Gram stain
Purple
Pink/Red
Peptidoglycan layer
Multi- layered (thick)
Single-layered (thin)
Teichoic acids
Present
Absent
Periplasmic space
Absent
Present
Lipo-polysaccharide content
High
Virtually none
Resistance to physical disruption
Low
High
Inhibition by basic dyes
Low
High
Antibiotic resistance
Low
High
Susceptibility to anionic detergents
Low
High
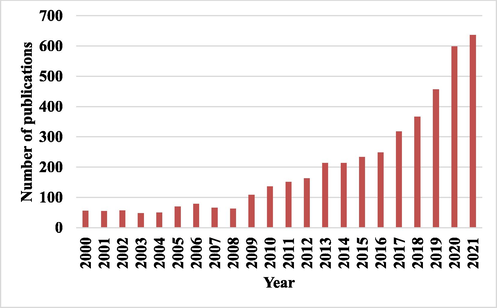
The number of publications from 2000 to 2021 that contain the terms “antimicrobial photodynamic therapy” (PubMed: Accessed in June 2022).
3 Mechanisms of photodynamic therapy
There are three basic principles of PDT: (a) the Grotthus-Draper Law, also called as Principle of Photochemical Activation, states that for a photosensitizer to be activated, the light used must be of an appropriate wavelength so that it can be absorbed by the system to bring about a photochemical change, (b) the Stark Einstein Law (also called as Photoequivalence law) states that each mole of photosensitizer that is involved in the photochemical reaction absorbs one quantum of the light emitted, and (c) the Bunsen-Roscoe Law states that a photochemical response is proportional to the product of light intensity and exposure. When a photosensitizer is illuminated by the light of an appropriate wavelength, the chromophore gets transformed from its stable, quiescent ground state to an excited single state. Here, the molecule might undergo an intersystem crossing into a long-lived high-energy triplet state or it might emit fluorescence to come back to its original stable ground state. The triplet state is also unstable and can undergo a non-radiative transition, giving rise to phosphorescence or it might transfer energy to another molecule (Griesbeck 1991). However, the triplet-singlet state transitions are forbidden as the change in electron spins is required (Castano et al., 2004). In the body, where oxygen levels are significant, the triplet state readily transfers energy to the surrounding oxygen or substrate molecules, which absorb energy and convert into a singlet state oxygen or cytotoxic substrate radicals (Weishaupt et al., 1976). Photosensitization reactions induced by the photosensitizer activation, are known to cause damage to the biological membranes through the oxidation of proteins, nucleic acids and lipids. This damage is triggered by both type I and type II reactions (Fig. 4).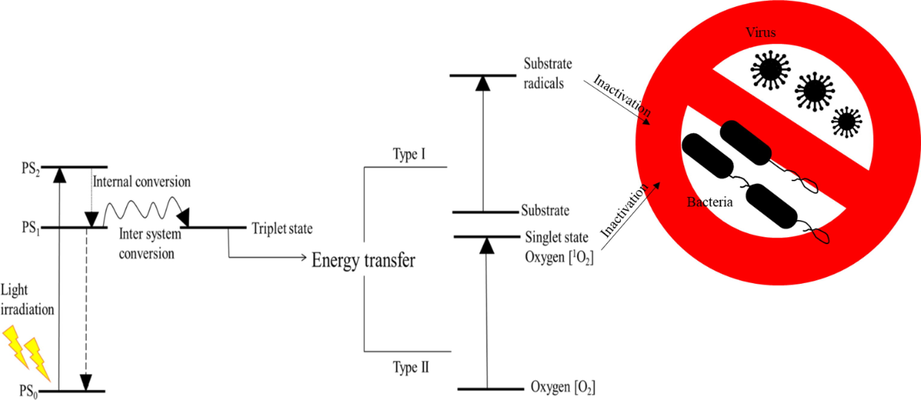
Schematic representation of mechanism underlying photodynamic therapy. PSo- Photosensitizer in ground state; PS1 and PS2- Photosensitizer in excited states (n = 1 and 2, respectively).
3.1 Types of reactions
3.1.1 Type I reactions
Type I reaction mechanisms involve electron transfer between the excited photosensitizer and substrate, yielding free radicals such as superoxide and hydroxyl radicals (Ochsner 1997). In anoxic environments, the energy from the excited photosensitizer is transferred to a substrate (such as a cell membrane or an organic substrate), which eventually forms substrate radicals by the transfer of a proton or an electron. These substrate radicals cause cell death by the destruction of the cell membrane. In hypoxic environments, the substrate radicals may further react with oxygen to produce reactive oxygen species (cytotoxic). These reactions depend on the substrate concentration. hν + PS → PS1 → PS3 PS3 + S → S°+ +PS°3- PS°3- + O2 → PS + O°2-→HO°; H2O2
In type I photoreaction, the excited photosensitizer in the triplet state (PS3) captures an electron from the substrate and forms two radicals: substrate (S°+) and photosensitizer (PS°3-) radicals. In hypoxia environments (where O2 is present), the photosensitizer radical transfers an electron to the molecular oxygen, forming a superoxide radical (O°2-).
3.1.2 Type II reactions
In a type II reaction, the energy is directly transferred to the molecular oxygen to form an excited state of singlet oxygen that is known to be an energized molecular oxygen (0.98 eV or + 1.57 × 10-19 J). The singlet state oxygen is a very strong oxidizing agent and damages the biological membrane through oxidation. It has a very short half-life (nanoseconds) and the diffusion is limited only to a small distance (about 100 nm). Hence, the cytotoxicity is limited to the immediate cells (Ryskova et al., 2010). hν + PS → PS1 → PS3 PS3 + O2 → PS+ 1O2 1O2 + S → S(O)
In type II reactions, the photosensitizer transfers its excitation energy to the molecular oxygen in the ground state (O2) resulting in the singlet state oxygen (1O2), which oxidizes substrates such as proteins and nucleic acids. Both type I and type II reactions may occur simultaneously and the ratio between these processes depends on the type of photosensitizer used, the concentrations of substrate and oxygen, and the binding capacity of the photosensitizer to the substrate (Dougherty et al., 1998, Milanesio et al., 2005). It is generally believed that the single state oxygen (1O2) generated from type II reactions is largely responsible for the PDT effect (Pineiro et al., 2001, Pineiro et al., 2002). However, recent studies have reported an amplified PDT response with the radicals generated from type I reactions, especially under low O2 environments (Vakrat-Haglili et al., 2005, Silva et al., 2010).
3.1.3 Type III reactions
A few researchers have suggested a different mechanism that occurs in parallel with type I and type II reactions (Gál 1992). These reactions occur between the photosensitizer in the triplet state (PS3) and the free radicals present in the system. This reaction is also known as a triplet-doublet process (or) a modified type I reaction (Gal 1992, Yoon et al., 2013). Very less is known about these reactions. Type I and type II reactions are considered to be the most common mechanisms for bacterial cell death following PDT. The mechanism of photoinactivation induced by three different cationic porphyrins (Tri-Py(+)-Me-PF, Tetra-Py(+)-Me and Tri-SPy(+)-Me-PF) on E. coli was studied in the presence of a singlet oxygen quencher (sodium azide) or free radical scavengers (d-mannitol and l-cysteine). The use of scavengers helps in identifying the relative importance of the two pathways. The study concluded that photoinactivation of the bioluminescent E. coli by Tri-Py(+)-Me-PF, Tetra-Py(+)-Me and Tri-SPy(+)-Me-PF occurs mainly through singlet oxygen (type II) than free radicals (type I) mechanism (Tavares et al., 2011). These reactions result in functional damage, morphological changes and cell membrane damage in bacteria. Functional damage includes the oxidation of protein–protein cross-links, inhibition of metabolic processes such as glucose transport and DNA synthesis, and inactivation of essential enzymes (Amos-Tautua et al., 2019). Morphological changes in bacteria mainly occur in the mesosome structure, while cellular membrane damage is a result of the breaking down of cellular contents and subsequent disruption of the membrane transport system and enzymes (Jori and Roncucci 2006). Some microorganisms can produce antioxidant enzymes, such as catalase, superoxide dismutase and peroxidase, which confer protection against some ROS; however, this does not hold for singlet oxygen (1O2) species produced by photosensitizers as singlet oxygen can degrade these antioxidant enzymes.
4 Light sources
The clinical approach of PDT depends on the selection of an appropriate light source and a light delivery system (Brancaleon and Moseley 2002). The light should penetrate the tissues to be absorbed by the photosensitizer. Generally, light with a shorter wavelength has less tissue penetration and is mostly absorbed, resulting in skin photosensitivity. Hence, a photosensitizer with maximum absorption at longer wavelengths is more suitable. The ‘optical window’ of a living tissue ranges between 600 nm and 1300 nm; however, wavelengths greater than 850 nm hardly generate singlet state oxygen (Wilson et al., 1985). Studies have postulated that the wavelength of light ranging from 600 nm − 700 nm has 50–200 % more penetration compared to the light of shorter wavelengths (Keijzer et al., 1989, Wu et al., 1993). The penetration of light into a tissue also depends on the optimal characteristics of the tissue, which can be defined by its optimal penetration depth (OPD) (Gardner et al., 1996, Richards-Kortum and Sevick-Muraca 1996). OPD is defined as the depth at which the intensity of the propagating light is attenuated to 37 % of its initial value. At 635 nm, the OPD of the brain is 800 µm, whereas for the bladder it is 4 mm (Shackley et al., 2000). Hence, the choice of the light source must be determined by considering the location and depth of the lesion, the action spectrum of the photosensitizer (relative effectiveness at different wavelengths for generating a desired response), the optimal wavelength to generate high yields of singlet state oxygen, fluence rate of light (high fluency of light causes oxygen depletion), cost and size of the light source (Juzeniene et al., 2007). Most light sources used for PDT were developed to have an optimized output near wavelengths between 600 and 700 nm. Based on the source, light sources can be divided into laser and non-laser sources (Kim and Darafsheh 2020).
4.1 Laser sources
A laser is an ideal light source for PDT due to its monochromaticity and high-intensity coherent light. Since, coherence is lost after penetration into the tissue, this property would be of no importance in PDT. The monochromaticity allows the delivery of light of a precise wavelength where the photosensitizer has its maximum absorption. Two laser systems are popularly used for the photodynamic inactivation of bacteria: Helium-Neon laser and diode laser (Calin and Parasca 2009). Helium-Neon laser is a gas laser developed by Javan and coworkers in 1961. It operates at wavelengths in the red part of the visible spectrum. The best and most widely used Helium-Neon laser operates at 632.8 nm. The power of the laser beam is between 0.5 and 50 mW (Calin and Parasca 2009). These have been used extensively for PDT of S. aureus, P aeruginosa (DeSimone et al., 1999), Streptococcus mutans (Zanin et al., 2005), and Fusobacterium nucleatum (Chan and Lai 2003). Diode lasers represent a breakthrough in the clinical use of PDT as they can emit at a longer wavelength of 630–950 nm (Brancaleon and Moseley 2002). These are made up of semiconductor substances and were easily portable due to their light weight (Mang 2004). They have an air cooling system utilizing 120 V power and an ability to deliver both continuous and pulsed light (Brancaleon and Moseley 2002). The limitation of using these lasers is that they offer only a single output wavelength of light (Brancaleon and Moseley 2002). They have been used for the photodynamic treatment of Staphylococcus epidermidis (Carvalho Pde et al., 2006), Actinobacillus actinomycetemcomitans, Fusobacterium nucleatum, Porphyromonas gingivalis, and Streptococcus sanguis (Chan and Lai 2003).
4.2 Non-laser sources
Non-laser sources used in aPDT majorly include lamps and light-emitting diodes (LEDs). Two types of lamps are available, lamps that emit a continuous spectrum (incandescent lamps) and lamps with a spectrum in bands (metallic vapor lamps) (Calin and Parasca 2009). The maintenance of lamps is easy and cheap. Filters are employed to narrow the spectrum to an appropriate wavelength (Calin and Parasca 2009). Incandescent lamps consist of a glass bulb with a tungsten filament inside the lamp. The glass bulb can be filled with a vacuum or an inert gas. Light is produced by heating the filament using an electric current. These lamps can deliver power up to 250 mW/cm2 over a wide range of spectrums (Brancaleon and Moseley 2002). Incandescent lamps have been used for the photodynamic inactivation of Gram-positive bacteria such as S. aureus and Gram-negative bacteria such as P aeruginosa (Huang et al., 2010). Gaseous discharge lamps or Xenon lamps are based on the electric discharge between two electrodes in a vapor or gas atmosphere. The emitted radiation depends on the gas. Xenon and mercury vapor lamps are the most commonly used lamps (Calin and Parasca 2009). In xenon lamps, the cathode is a metallic tube containing zirconium dioxide and the anode is a metallic plate. They emit a broad-spectrum light ranging from 300 nm to 1200 nm and are characterized by large output and high fluence rates. Xenon lamps have been employed for photodynamic inactivation of S. aureus, E. coli (Maclean et al., 2008), and Staphylococcus epidermidis (Guo et al., 2010). Light-emitting diodes (LEDs) are used in the aPDT because of their low cost and versatility. They consist of an array of semiconducting LED chips and are used to emit low-powered broad-spectrum light (Schmidt et al., 1996). The choice of the emission wavelength ranges from ultraviolet (350 nm) to near-infrared (1100 nm) (Brancaleon and Moseley 2002). LEDs have been used in inactivating bacteria, such as S. aureus, Porphyromonas gingivalis (Chui et al., 2013), and E. faecalis (Rios et al., 2011). In the following section, we will elaborate on various photosensitizers used in antibacterial therapy.
5 Photosensitizers
aPDT is highly dependent on the photosensitizer used. Photosensitizers are chemical compounds that are capable of absorbing light of an appropriate wavelength and transforms it into useful energy (Sharman et al., 1999). More than 400 natural and synthetic photosensitizers are known and developed in the past few years, which include dyes, drugs, cosmetics and chemicals (Santamaria and Prino 1972). In aPDT, an ideal photosensitizer should possess the following properties: (a) minimal tendency to aggregate, (b) capable of generating high triplet states (PS3) with a long half-life, and also be able to generate singlet oxygen and other reactive oxygen species (ROS), (c) rapid clearance from the body and low skin phototoxicity, (d) retain preferentially in the target tissue, (e) minimum dark toxicity and be cytotoxic only in the presence of light, (f) have a strong absorbance at longer wavelengths (600–800 nm), where the tissue penetration of light is the maximum, (g) water-soluble, (h) stable, chemically pure, and easily synthesized (Wöhrle et al., 1998, Sharman et al., 1999).
5.1 Classification of photosensitizers and their use in antimicrobial therapy
Photosensitizers used in aPDT can be classified in various ways. Depending on when photosensitizers were generated, they are categorized under three generations. This is useful for doctors and scientists working in clinics. Photosensitizers can also be classified based on the chemical structure. This classification is widely accepted by chemists, but has limited utility in clinics.
5.2 Traditional photosensitizers classification
5.2.1 First-generation photosensitizers
The naturally occurring porphyrins and their derivatives are considered the first generation of photosensitizers developed in the 1970 s. These compounds showed good photodynamic activity; however, they exhibited prolonged cutaneous phototoxicity due to low absorption bands at red wavelengths with a little dark cytotoxicity. These sensitizers are retained in cutaneous tissues for 2–3 months during which patients must avoid bright light (Wöhrle et al., 1998). Moraxella catarrhalis is a prevalent bacterial infection in children, which causes pediatric otitis media (OM). Photofrin showed significant bactericidal activity against both planktonic and biofilm-associated M. catarrhalis (Luke-Marshall et al., 2014).
5.2.2 Second-generation photosensitizers
They were developed in the late 1980 s to overcome the disadvantages of first-generation photosensitizers. These are chemically pure substances and have a high absorbance at longer wavelength regions (675–800 nm). Light with these wavelengths can penetrate to a depth of 2–3 cm (Wöhrle et al., 1998). They also have high triplet state yields with photosensitivity lasting for a short time (Juzeniene et al., 2007). Photofrin and other first-generation porphyrin related structures have a few disadvantages which include: difficulty in purifying derivatives, prolonged photosensitivity due to poor clearance, and weak absorption at longer wavelengths (Detty et al., 2004). These disadvantages led to the development of second-generation photosensitizers that are reported to have lesser photosensitivity, better in-depth light absorption and higher purity. The second-generation photosensitizers include expanded porphyrins (benzoporphyrin derivative, boronated porphyrin), chlorophyll derivatives (chlorins, purpurins), dyes (xanthenes, phthalocyanines), Boron-dipyrromethene (BODIPY), and intrinsic photosensitizers such as 5-aminolevulinic acid (Malacarne et al., 2022). 5-amino levulinic acid is converted to a photosensitizer, proto-porphyrin IX in situ (Koteeswaran 2000). The first photosensitizers to be clinically approved are dyes (Allison and Sibata 2010). Though the second-generation photosensitizers are 100 times more active when compared with the previous generation, they cause significant pain during the therapy due to severe skin photosensitivity (Koteeswaran 2000).
5.2.3 Third-generation photosensitizers
First and second-generation photosensitizers have been conjugated to various modifiers such as antibodies, liposomes, and nanoparticles, to increase the selectivity of photosensitizer to a particular tissue (Allison and Sibata 2010, Dube et al., 2018, Soy et al., 2019). Compared to first and second-generation photosensitizers, they bind specifically to the target cells and sensitize their killing without affecting the normal cells.
5.3 Classification based on chemical structure: chemically, photosensitizers can be divided into three categories
5.3.1 Porphyrin-related structures
Porphyrin photosensitizers are developed in the 1970 s and early 1980 s. They consist of a substituted aromatic macrocyclic ring containing four pyrrole-type residues that are connected by four methine groups (Fig. 1C). Porphyrins have demonstrated a significant broad spectrum of action against Gram-positive and Gram-negative bacteria at very low 0.1–5 μM concentrations. Early studies by Orenstein et al. showed that a complex of deuteroporphyrin-hemin complex was able to exhibit a strong bactericidal effect in S. aureus (Orenstein et al., 1997). However, in the case of Gram-negative bacteria such as E. coli and P aeruginosa, a pre-treatment of cells with either EDTA or nitrilotriacetic acid, and sodium hexametaphosphate was needed to make the bacteria susceptible to PDT through the removal of Mg2+ and Ca2+ ions (Malik et al., 1992). The photosensitizers in the first generation are mostly porphyrin-based or hematoporphyrin derivatives (HpD). Though HpD was demonstrated as a powerful photosensitizer, its chromatography showed several components (Juzeniene et al., 2007). HpD was treated with 5 % sulfuric acid and acetic acid for purification and the resulting purified form is known as Photofrin (Wöhrle et al., 1998). Photofrin is a commercially available porphyrin photosensitizer and has the longest clinical history among all photosensitizers (Allison et al., 2004).
Zoltan et al. reported antibacterial activity of meso-tetra(pyren-1-yl)porphyrin and Ni(II), Cu(II) and Zn complexes in E. coli. An efficient inactivation of E. coli was observed in the presence of Ni(II) and Cu(II) complexes and this was attributed to the high generation of singlet oxygen by the compounds (Zoltan et al., 2010). Porphyrins with nitro group substitutions have also been studied for tuning redox and photophysical properties. For example, Rahimi et al. examined the photoinactivation of 5,10,15,20-tetrakis(4-nitrophenyl)porphyrin (TNPP) and its zinc porphyrin complex (ZnTNPP) in P. aeruginosa and B. subtilis. The authors concluded that both compounds exhibited significant photoinactivation activity against both P. aeruginosa than B. subtilis in nutrient agar (Rahimi et al., 2016). A recent review by Amos-Tautua et al., highlighted the antibacterial photodynamic effects of porphyrins in a detailed manner (Amos-Tautua et al., 2019).
5.3.2 Phthalocyanine-related structures
Phthalocyanine, a tetrapyrrole derivative, is used in several areas of photonics such as optical memory, optical power limiting, and photomedicine (Fig. 1D) (Drobizhev et al., 2006). They resemble porphyrins in their structure, but have maximum absorption peaks at longer wavelengths (650–700 nm) (Wiederkehr 1996). Studies have shown that Gram-positive bacteria are more susceptible to phthalocyanine compared with Gram-negative bacteria, which has lipopolysaccharide in the outer membrane. Cationic photosensitizers such as zinc pyridinium phthalocyanine were found to have better photoinactivation of Gram-positive and Gram-negative cells than the anionic or neutral photosensitizers (Minnock et al., 1996). Metal cations such as Zn(II), Ga(III), and Si(IV) in tetra-N-methylpyridyloxy-phthalocyanine showed higher fluorescence quantum yields and singlet oxygen quantum yields compared to In (III) and Ge (IV) (Mantareva et al., 2013). The uptake and efficacy of phthalocyanine also depend on the number of hydrophilic groups (Paquette et al., 1991). Hence, the uptake kinetics and cell retention are different for hydrophobic and hydrophilic phthalocyanines. Various metallic complexes of phthalocyanine such as zinc phthalocyanine show photobiological activity, which is two times more efficient than other commonly used photosensitizers such as methylene blue, with low cytotoxicity and high capacity to penetrate through the cellular membrane (Moreira et al., 2008). Amphiphilic phthalocyanines are obtained by the direct sulfonation of the parent system and the activity is related to the degree of sulfonation. Disulfonic acid of aluminum phthalocyanine is more effective compared to tetrasulfonic acid derivatives (Bonnett 1995). One limitation of using phthalocyanine photosensitizers is that they show aggregation in solution, which leads to low photosensitization efficiency due to low quantum yields of excited states (Moreira et al., 2008).
5.3.3 Chlorins and bacteriochlorins
Chlorin core is obtained by the reduction of pyrrole double bond and further reduction of the second pyrrole double bond gives the bacteriochlorins (Fig. 1E). They show maximum absorption peaks at longer wavelengths (650–670 nm for chlorins and 730–800 nm for bacteriochlorins). As these are reduced forms, they are prone to oxidation. To prevent oxidation, large substituents are placed next to the reduced pyrrole ring (Nyman and Hynninen 2004). Metalated chlorins and bacteriochlorins have efficient photosensitizing activity as metalation strengthens the absorption of red light and moves their red absorption bands to longer wavelengths. These have a high quantum yield of excited states (Nyman and Hynninen 2004). However, these substances (e.g., tin ethyl etiopurpurin, SnET2) impart long-term skin photosensitization, which limits their utility (Sharman et al., 1999, Detty et al., 2004). Synthetic derivatives have also been developed and evaluated for use as photosensitizers.
5.3.4 Phenothiazine derivatives
These are an important class of photosensitizers that are majorly used in antimicrobial therapy (Fig. 1F). Methylene blue is currently being used for decontamination of freshly frozen plasma units (Wainwright 2002). Most photosensitizers in this class bring about the antimicrobial effect by their association with the nucleic acids inside the cell. Improved phototoxicity is observed against the Gram-positive and Gram-negative bacteria by modifying the structure. The addition of methyl or ethyl groups or nitro groups to methylene blue resulted in enhanced phototoxicity as these derivatives are more resistant to reduction compared to methylene blue. The reduction of methylene blue results in the formation of colorless, neutral species which do not show phototoxicity (Maisch 2009). Toluidine blue is another phenothiazine dye that predominantly shows phototoxicity against oral bacteria (Maisch 2009).
5.3.5 BODIPYs
BODIPYs are 4,4-difluoro-4-bora-3a,4a-diaza-s-indacene derivatives that were first reported in 1968. They are resulted from an unexpected reaction between 2,4-dimethylpyrrole and boron trifluoride etherate (BF3·Et2O). BODIPYs exhibit high fluorescence emission (Φ) with high molar extinction coefficient (ε) in the visible region. In addition, they are less affected by environmental conditions such as solvent polarity and pH, and show good resistance to photobleaching (Wittmershaus et al., 2001). The absorption and emission properties of BODIPY can be fine-tuned through the addition substituents on the main structure. For example, substitution by conjugating group at the 2/6-, 3/5-position, aromatic ring fusion with pyrrole moiety, and aza-nitrogen atom substitution at the meso-position results in red-shifts of the absorption and emission wavelengths (Liu et al., 2019). BODIPYs have shown excellent results as PSs in aPDT both in vitro and in vivo. BODIPYs with halogen atoms showed better antimicrobial photodynamic activity, especially halogenated structures with cationic groups (Agazzi et al., 2019). Though several studies reported the anticancer activity of BODIPYs, only a few studies highlighted its activity against microorganisms. Further research into assessment of antimicrobial activity using infection models is required.
6 Factors controlling the effectiveness of photodynamic therapy
The efficiency of aPDT depends on various factors such as the efficiency of the photosensitizer to produce a singlet state of oxygen, delivery system, light fluence rate, the dose of the light, and localization of the photosensitizer in the target tissue. Some of the factors that contribute to treatment success are described below.
6.1 Optimum tissue penetration depth of photoactivating light
The depth of light penetration determines the efficiency of PDT. Currently, PDT is limited to topical, subcutaneous, and endoscopic applications because of the limited tissue depth at which the singlet state oxygen can be generated (Tremblay et al., 2003). The light typically used to activate the photosensitizer cannot approach 5–10 cm in depth. Tissues have a high penetration in a phototherapeutic window ranging between 600 nm − 850 nm (Starkey et al., 2008). At shorter wavelengths, the light is attenuated by the hemoglobin and at longer wavelengths, the light is absorbed by water (Mir et al., 2008, Starkey et al., 2008). In a recent study, one- and two-photon activated pyropheophorbide-a methyl ester, a second-generation photosensitizer derived from chlorophyll a, was evaluated for efficacy in killing human cervical, lung and ovarian cancer cells.
6.2 Localization of photosensitizer in the target tissue
PDT depends on the retention of the photosensitizer in the target cell. Normal cells can also accumulate photosensitizers just like the bacterial cells. This can lead to undesired toxic effects. Hence, it would be beneficial to selectively accumulate photosensitizer in the bacterial cells. Higher localization of the photosensitizer in the bacterial cells, leads to improved therapeutic efficacy at a reduced dose (Lee and Kopelman 2011). Initially, researchers have attempted to develop new photosensitizers by changing the physicochemical properties such as logP value to improve their specificity (Rosenkranz et al., 2000). Hence, second-generation photosensitizers had a better efficacy compared to first-generation photosensitizers. In the third generation, photosensitizers have been conjugated to carriers such as liposomes, micelles, nanoparticles (NPs) to target them to specific cells (Josefsen and Boyle 2008).
6.3 Time interval between administration of photosensitizer and light exposure
The interval between the administration of photosensitizer and light exposure is critical for the efficiency of the treatment. In the case of topical administration, there is no delay in light exposure after the administration of photosensitizer (Lee and Kopelman 2011). With other modes of administration, the photosensitizer needs to accumulate in the bacterial cell before light exposure. When systemically administered, the delay in light exposure should not be more than the half-life of the photosensitizer. The photosensitizer-light interval of currently approved photosensitizers is pretty long. For example, photofrin has an interval in the range of 40–50 h and Foscan has an interval between 90 and 110 h (Lee and Kopelman 2011).
7 Nanosystems in photodynamic antimicrobial therapy
The ability of photosensitizers to generate bactericidal efficacy depends on their physicochemical, photophysical, and photochemical properties. The key physicochemical properties that affect acid dissociation constant (pKa) include solubility and lipophilicity, which indirectly affect the drug dissolution rate and absorption. Likewise, the photochemical stability of a photosensitizer affects the safety and treatment efficacy (Breskey et al., 2013). Moreover, PDT tends to damage normal cells and result in side-effects such as burns, swelling, pain, and scarring in the treatment area, shortness of breath. Skin problems, such as redness, stinging, swelling, or itching are also reported (Borgia et al., 2018). The use of nanosystems in aPDT can overcome the physicochemical and photochemical challenges of PSs while alleviating the side effects. Further nanocarrier systems enhance the targetability of photosensitizer towards microbial infections. The advantages associated with the use of nanocarriers in aPDT are highlighted in Fig. 5. The ability of PDT to eradicate microbial infections could be synergized with the use of nanotechnology. Through nanotechnology, it is possible to increase the antimicrobial effect, reduce the photosensitizer concentration and precisely deliver the photosensitizer to the target region (Polat and Kang 2021). Here, we mainly focus on the latest developments in the use of nanosystems in aPDT. Based on the functionality, nanosystems in aPDT can be broadly categorized into passive nanosystems and active nanosystems (Chatterjee et al., 2008, Perni et al., 2011, Nora et al., 2017).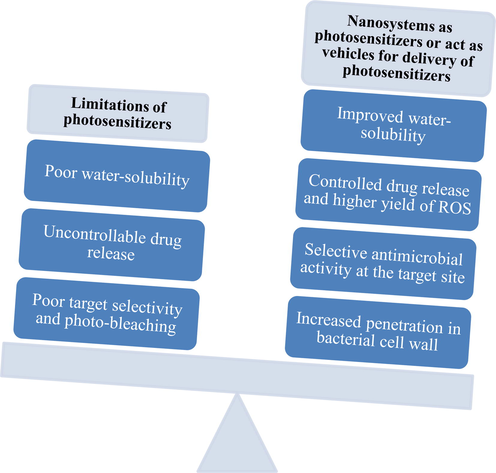
Limitations of photosensitizers and advantages associated with the use of nanosystems as photosensitizers or carriers of photosensitizers.
7.1 Passive nanosystems
Most photosensitizers suffer from low bioavailability and poor bio-distribution. Photosensitizers can be physically entrapped inside NPs which exhibit improved penetration of the photosensitizer into the bacterial cells (Garapati et al., 2015). Nanocarriers reduce the ability of target cells to efflux photosensitizer molecules and thus modulate drug resistance. They can prevent the photosensitizer to self-aggregate to form ineffective dimers or trimers. Passive nanosystems mainly act as a carrier of photosensitizer molecules to deliver them to the target cells, while an active nanosystem acts through participation in the photosensitizer excitation process. In a passive nanosystem, the photosensitizer is either loaded into the nanocarrier system or conjugated to the surface of nanocarrier system with the help of a linker. Nanocarriers are being increasingly used to control the release of photosensitizer at the target site (Chatterjee et al., 2008). There is a vast literature on the application of NPs in PDT for cancer (Kopelman et al., 2005, Chen and Zhang 2006, Wieder et al., 2006). But, the research related to the application of nanocarriers in aPDT is limited when compared to its application in cancer therapy. Based on the material composition, passive nanosystems can be sub-classified into biodegradable and non-biodegradable nanosystems. Biodegradable nanosystems include polymeric nanoparticles, liposomes and polymeric micelles, while non-biodegradable nanosystems include ceramic, gold, iron oxide, and polyacrylamide nanosystems. The delivery system can be selected based on the barrier properties of the target site, physicochemical properties of the photosensitizer, the dose of photosensitizer to be delivered, and patient acceptability.
7.1.1 Biodegradable systems
Biodegradable nanoparticles are made of polymers that can degrade in the biological environment and release the photosensitizer. These polymers degrade by simple hydrolytic or enzymatic hydrolytic processes. The chemical composition of biodegradable polymers can be designed in such a way that, they can accommodate photosensitizer molecules with varied physicochemical properties such as molecular weight, solubility, and charge. The polymer degradation and drug release kinetics can be optimized by changing the polymer composition. Incorporating site-specific moieties on the surface of these NPs allows active targeting of bacterial cells (Li 2013). The advantages of these NPs include high drug-loading and sustained drug release. The use of biodegradable NPs in PDT began in 1990 to control the release of photosensitizers. Previously, biodegradable NPs-loaded with antibiotics have been used to target bacterial cells (Toti et al., 2011).
7.1.1.1 Polymeric nanoparticles
Among the biodegradable polymers, poly-lactic-co-glycolic acid (PLGA) is used as a drug delivery carrier due to its favorable properties such as biocompatibility, biodegradability, bioresorbability and mechanical strength (Li 2013). PLGA has been used to prepare methylene blue-loaded NPs for photosensitization of E. faecalis, which led to 99 % bacterial killing. The synergism of PLGA-NPs and light contributed to a 10-fold killing of E. faecalis in the infected root canals of human teeth (Soukos and Goodson 2011). Tsai et al., showed that biodegradable NPs reduced the dose of photosensitizer to induce photodynamic inactivation and improved the overall antimicrobial efficacy (Chen et al., 2012).
Bacterial cell walls are negatively charged due to their chemical composition. Moreover, during infection, bacteria release organic acids such as lactic and acetic acids by the way of anaerobic glycolysis creating an acid environment in the vicinity. In an attempt to better target the bacterial cell wall, Liu et al., developed charge-switching polymeric NPs of chlorin e6 (Ce6) for urinary tract infection treatment. Nanoparticles were developed using poly(HDDA-co-DBPA)-mPEG copolymer. These NPs exhibited a charge-switching behavior in the vicinity of bacteria resulting in a remarkable bactericidal activity in Gram-positive and -negative bacteria. After irradiation, ROS generated by Ce6 decreased the bacterial infection. The polymeric NPs exhibited low cytotoxicity and high anti-bacterial activity. After treatment with PDT in a cystitis-induced mouse model, a significant decrease in bacterial cells was observed. This study concluded that charge exhibiting polymeric NPs could be beneficial in the treatment of UTIs (Liu et al., 2015).
Many photosensitizer molecules have negative charge potential. So free photosensitizer application may lead to low efficiency in certain parts of the body such as the stomach (pH 1.0–2.0), vagina (pH 4.0–5.0), bladder (pH 4.5–8.0), and skin (pH 4.0–5.5), have an acidic environment that helps in the survival of bacteria. Hence, it is believed that the positive charge of a photosensitizer increases its uptake and phototoxicity on bacterial species. For instance, indocyanine green-loaded PLGA-NPs coated with chitosan showed a good antimicrobial photodynamic effect on P.gingivalis (Nagahara et al., 2013). In a recent study, the synergistic effect of chemical and aPDT for wound healing was tested using multifunctional light-responsive NPs consisting of quaternary ammonium chitosan and photosensitizer chlorin e6 (Ce6) in addition to the magnesium-epigallocatechin-3-gallate complex. Up on irradiation with light (660 nm), magnesium ions were effectively released into the medium, which accelerated wound healing in mammalian cells. Nanoparticles with a positive surface charge adhered to the bacterial surface and generated ROS in the presence of laser irradiation destroying the bacterial membrane structure (Hu et al., 2019). This multifunctional nanoparticulate approach appears to be promising in simultaneously destroying the pathogenic bacteria and accelerating the wound healing process. In the near future, polymeric NP research discussed above must be further studied in preclinical and clinical studies.
7.1.1.2 Liposomes
Liposomal delivery of photosensitizers received tremendous attention due to its ability to improve photosensitizer’s loading capacity, safety, and selective delivery. Liposomes are used in the delivery of both lipophilic and hydrophilic photosensitizers. Lipophilic photosensitizers can be dissolved in the lipid bilayer, while hydrophilic photosensitizers can be suspended in the aqueous inner phase of liposomes (Q Mesquita et al., 2018). Preliminary investigations indicate that photosensitizers loaded in liposomes enhance the cytocidal effects on bacteria. The behavior of liposomes in the biological milieu depends on their physicochemical properties such as the type/amount of lipids used in the formulation and the degree of drug encapsulation in the lipid bilayer (Ferro et al., 2006). In a study by Bombelli et al., the antibacterial activity of m-tetrahydroxy-phenylchlorin loaded into mixed cationic liposomes composed of varying ratios of dimyristoyl-sn-glycero-phosphatidylcholine/cationic surfactants derived from l-prolinol was reported. This study observed a clear correlation between the physicochemical and biological features of formulations with their interaction with the biological environment (Bombelli et al., 2008). The use of cationic agents such as cetyltrimethyl ammonium bromide (CTAB) and dimethyldioctadecylammonium chloride (DOTAC) in the preparation of liposomes improved the overall microbial activity. For example, CTAB-liposomes composed of various ratios of dimyristoyl-sn-glycero-phosphatidylcholine (DMPC) and CTAB significantly enhanced the photodynamic efficacy of chlorine e6 against C. albicans (Yang et al., 2013). Likewise, methylene blue-loaded liposomes made up of a mixture of DOTAC, cholesterol (Chol) and dipalmitoylphosphatidylcholine (DPPC) showed higher efficiency in terms of bacterial cell toxicity and penetration into the bacterial biofilm of E. Coli as compared with methylene blue aqueous solutions (Bani et al., 2017). Photosensitizer loaded in liposomes were used in the photodynamic inactivation of endodontic infections. For instance, aluminum chloride phthalocyanine entrapped in cationic liposomes exhibited preferential absorption/phototoxicity in bacterial cells when compared to eukaryotic dental pulp cells. In addition, a clinical study in volunteers showed a mean reduction of 82 % of total bacteria in the treated cavities following the application of PDT (Longo et al., 2012). Visudyne® is the first clinically-approved liposomal photosensitizer used in the treatment of age-related macular degeneration. Taking into consideration the translational success of this formulation, it is reasonable to consider that liposomes for aPDT might soon enter clinical testing (Ghosh et al., 2019).
7.1.1.3 Polymeric micelles
Polymeric micelles are colloidal structures made up of amphiphilic block copolymers that tend to self-assemble in water. The block copolymers are composed of a hydrophobic polymer such as polylactic acid (PLA) and a hydrophilic polymer such as polyethylene glycol (PEG). They are widely used in the delivery of poorly water-soluble drugs such as hydrophobic photosensitizers. Unlike polymeric NPs, liposomes, polymeric micelles are dynamic and reversible in nature (Awad et al., 2022). Polymeric micelles made up stimuli-responsive copolymers can enhance the delivery of drugs into microbial biofilms/infection sites, which are characterized by lower pH compared to healthy tissues (Albayaty et al., 2020). In a recent study, pH-responsive micelles were prepared by conjugating chlorin e6 to polyethylene glycol-b-poly(2-(hexamethyleneimino) ethyl methacrylate-co-aminoethyl methacrylate) (PHMEMA). The polymeric micelles of PEG-PHMEMA-Ce6 remained negatively charged in the physiological environment, which prolonged blood circulation. However, in the microenvironment of bacterial infection micelles exhibited a positive charge that promotes attachment to the negatively charged bacterial membrane (Fig. 6). The in vivo experiment in a subcutaneous infection model in mice showed a good antibacterial effect of pH-sensitive micelles upon exposure to 660 nm laser (0.865 W/cm2) for 10 min (Wang et al., 2021). A different strategy to enhance the delivery of photosensitizers is by using enzyme-sensitive polymers by targeting lipases, which are secreted by many microbes. Lipase-sensitive micelles constructed from (methoxy poly (ethylene glycol)-block-poly(ε-caprolactone) (PCL- mPEG) were loaded with hypocrellin A in the core. This study reported that minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of mPEG-PCL/ hypocrellin A micelles after light irradiation were 0.69 and 1.38 mg/L, respectively, in methicillin-resistant S. aureus (MRSA) infections. Further, mPEG-PCL/ hypocrellin A micelles increased the survival rate of mice (induced with acute peritonitis) to 86 %, suggesting the potential of lipase-sensitive polymer micelles in treating methicillin-resistant S. aureus antibiotic-resistant bacterial infection. In a different study, polymeric micelles were combined with silver NPs to achieve a synergistic antibacterial effect. In this approach, copolymer micelles of poly(aspartic acid)-block-poly(ε-caprolactone) (PAsp-b-PCL) and photosensitizer protoporphyrin IX (PpIX) were prepared and decorated with silver NPs. This strategy showed a robust eradication of subcutaneous infections (drug-resistant S. aureus) (Hou et al., 2020). Pluronic F-127 polymer micelles were shown to be highly suitable nanosystems for the cationic porphyrins solubilization such as pyridyl-containing meso-arylporphyrins. Nanomicelles of Pluronic F-127 reduced the photosensitizer dose by two times and four times in the absence and presence of light, respectively. E. coli bacteria inhibition by Pluronic F-127 nanomicelles of pyridyl-containing meso-arylporphyrins was slightly lower than compared to the killing effect on S. aureus (Zhdanova et al., 2020). The flexibility offered by polymeric micelles in the molecular design of various photosensitizers can lead to new ways of increasing efficiency for the photodynamic inactivation of microorganisms.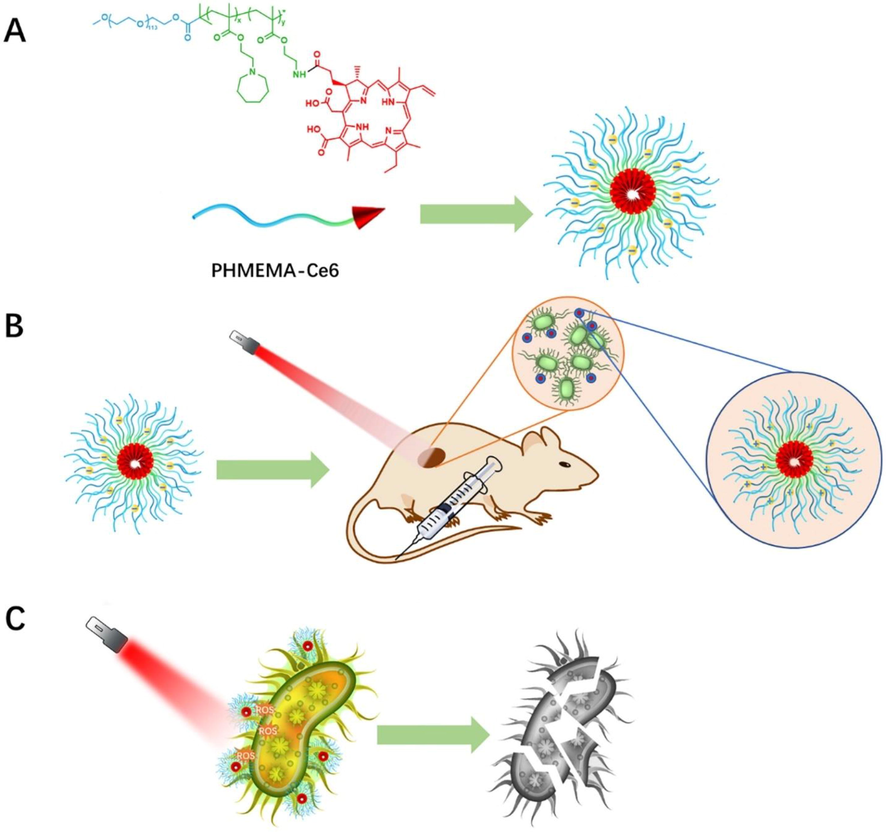
(A) Structure scheme of PHMEMA-Ce6 prodrug polymer and the process of self-assembly. Bule part: PEG, green part: PHMEMA, red part: Ce6. (B) pH-responsive charge conversion of PEG-b-P(HMEMA-co-AEMA)-Ce6 (PHMEMA-Ce6) micelle at acidic infection microenvironment. (C) Binding ability and PDT antibacterial effect of the micelle. ().
Reproduced with permission from Wang et al., 2021
7.1.2 Non-biodegradable nanoparticles
Non-biodegradable NPs including metallic NPs have garnered considerable attention due to their ability to control the spread of infections. These NPs can bind to the bacterial cell surface, causing membrane damage and alteration in membrane potential resulting in cytoplasmic leakage and cellular damage (Akhtar et al., 2021). Photosensitizers are not toxic to target cells by themselves rather they act like catalysts, which upon radiation convert the dissolved oxygen to toxic products. So, photosensitizer molecules should be properly activated to achieve a successful antimicrobial action. In such conditions, non-biodegradable polymers can be used as carriers in place of free photosensitizers (Chatterjee et al., 2008). The following are the advantages of non-biodegradable NPs over biodegradable NPs: (a) size, shape, and porosity of non-biodegradable NPs can be easily controlled, (b) inert materials used in the preparation of non-biodegradable NPs make them stable to environmental fluctuations, and (c) unaffected by the microbial attack (Li 2013). Most of the non-biodegradable NPs are ceramic based-especially silica, but a few of them are made from organic polymeric NPs.
7.1.2.1 Ceramic nanoparticles
Ceramic NPs offer several advantages over organic polymeric NPs such as stability and lower particle size. The particle size, shape, and porosity of ceramic NPs can be better controlled over polymeric NPs (Roy et al., 2003). Zhang et al., studied the effect of photosensitizer-loaded silica NPs as compared to free photosensitizer. This study reported that the association of photosensitizer with NPs made it more resistant to photobleaching. They also showed the superior killing efficiency of photosensitizer-loaded silica NPs in comparison to the free photosensitizer in Methicillin resistant S.aureus and S.epidermidis (Guo et al., 2010). Mirzahosseinipour et al. investigated the antimicrobial photodynamic effect of curcumin silica nanoparticles on planktonic and biofilms of P.aeruginosa, S.aureus and compared the effect with free curcumin. This study concluded that curcumin-silica nanoparticles showed a significant reduction in the number of bacteria in planktonic conditions and biofilm production. Moreover, curcumin-silica nanoparticles showed wound healing properties as identified through the in vitro scratch assay (Mirzahosseinipour et al., 2020). MCM-41 belongs to the family of silicate and alumosilicate solids that have gained importance due to their uniform pore structure, large surface area, high pore volume, biocompatibility, and good chemical and thermal stability. In a recent study, ZnO/Pd-MCM-41 was synthesized using a hydrothermal procedure and its antibacterial activity was tested in the presence of visible light. ZnO/Pd-MCM-41 showed promising antibacterial activity against E. coli, P. aeruginosa and S. aureus with inhibition zones of 17 ± 0.4 mm, 18 ± 0.4 mm and 22 ± 0.2 mm, respectively (Nazir et al., 2021).
7.1.2.2 Gold nanoparticles
The antimicrobial action of metal NPs such as gold nanoparticles (AuNPs) is well known. Further AuNPs have been used extensively in therapeutics and diagnosis because of their small size and large surface-area-to-volume ratio (Dhamecha et al., 2016). AuNPs have suitable optical properties and facilitate the bioconjugation of molecular probes on the surface. These properties make them a suitable choice of drug carriers in nanomedicine for the killing of diseased cells and microbes. When conjugated with cationic thiazine photosensitizers such as methylene blue, AuNPs were effective in the inhibition of biofilms of S. mutans. Further, increasing the concentration of photosensitizer along with AuNPs reduced bacterial biofilm formation more effectively (Lavaee et al., 2022). More complex systems such as the coating of chitosan onto gold–silver core–shell NPs were used in the delivery of toluidine blue. Akhtar et al., through in vitro studies showed that toluidine blue conjugated chitosan-coated gold-silver core-shells (TBO–chitosan–Au–AgNPs) mediated PDT was effective in combating diabetic foot ulcer caused by multi-drug resistant strains (Akhtar et al., 2021). TBO-chitosan-Au-AgNPs-mediated PDT demonstrated a promising anti-bacterial therapeutic approach, which lead to synergistic healing of diabetic foot ulcer caused by multi-drug resistant bacterial strains (Akhtar et al., 2021). More recently biogenic gold NPs (nanoparticles made by a living organism such as fungi) were shown to prevent fast photobleaching and enhance the photoactivity/bactericidal effect of methylene blue (MB) molecules (Maliszewska et al., 2021). An interesting aspect about gold nanoparticles is they are non-toxic and being used in other therapies (Dykman and Khlebtsov 2012). Hence, it is plausible that clinical approval and eventual application of gold nanoparticles will be easier to achieve than other nanocarriers systems.
7.1.2.3 Iron oxide nanoparticles
Iron oxide NPs, especially magnetite (Fe3O4) and maghemite (γ-Fe2O3), are considered promising nanotheranostic agents due to characteristics such as superparamagnetic behavior, high saturation magnetization, and ability to act as heat mediators in magnetic hyperthermia and biocompatibility. Iron oxide NPs exhibit a large surface-to-volume ratio, which permits the functionalization of drug molecules on their surface. Further, iron oxide NPs can be directed to the required location using an external magnetic field (Cabana et al., 2020, Toledo et al., 2020). In a recent study, Fe3O4 NPs were prepared by the co-precipitation of iron (II) and iron (III) salts in aqueous solution and covered with methylene blue-entrapped silica (magnetite-silica-methylene blue). In vitro studies in E. coli showed eradication following photodynamic activation of Fe3O4-silica-methylene blue (Fig. 7). However, these results should be further tested in preclinical assays (Toledo et al., 2020). De Santana et al., reported the synthesis and characterization of superparamagnetic iron oxide NPs-curcumin conjugates through bonding between the keto-enol moiety of curcumin and the Fe atoms present on the surfaces of the SPIONs using a method free of organic solvents as a dispersant. The conjugate not only showed photodynamic action when irradiated with blue LED light, but also displayed heating power under an alternating magnetic field (AMF). A total reduction in the bacterial load against S. aureus in the planktonic phase was observed with the application of blue light at 3.12 J cm−2 (de Santana et al., 2020).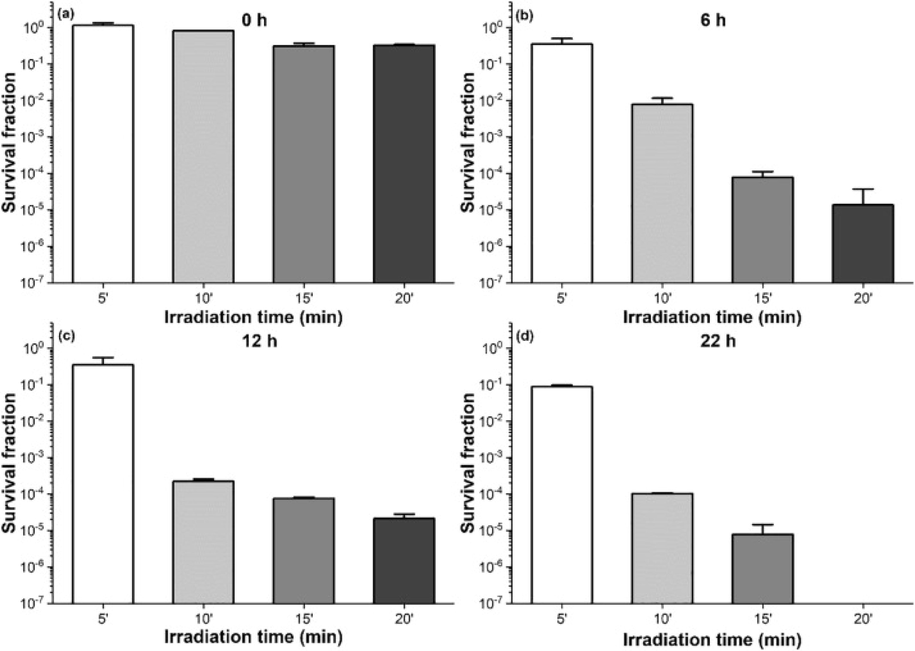
Mean values ± standard deviation of E. coli survival fraction following Fe3O4-2SiO2-MB-mediated PDT. (a) no MB release; (b) after 6 h of MB release; (c) after 12 h of MB release; (d) after 22 h of MB release. ().
Reproduced with permission from Toledo et al., 2020
7.1.2.4 Polyacrylamide nanoparticles
Polyacrylamide polymers are used to synthesize NPs of photosensitizer molecules through encapsulation in the non-porous core (Gualdesi et al., 2021). Polyacrylamide polymer is suitable in the preparation of NPs due to its low cost, ease in chemical modification, biocompatibility and ease in preparation of NPs (Li 2013, Gualdesi et al., 2021). Polyacrylamide polymer was found to be successful in delivering various photosensitizers such as methylene blue, Azure B, neutral red, and their halogenated derivatives for the elimination of bacterial infections (Tang et al., 2005, Tang et al., 2008, Gualdesi et al., 2016). It is possible to produce NPs of small size that allows rapid diffusion of 1O2 and removal from the body through renal clearance. Polyacrylamide nanoparticles inhibited the growth of Gram-positive bacteria with a higher efficacy compared to Gram-negative bacteria (Gualdesi et al., 2019). In a recent study, polyacrylamide NPs of monobrominated derivatives of Azure B and Neutral Red were prepared and characterized to overcome difficulties in solubility, aggregation and optimize photodynamic efficiency. The ability of both monobrominated derivatives of Azure B and Neutral Red to produce singlet oxygen was significantly increased with respect to their free forms (Gualdesi et al., 2021).
7.2 Active nanoparticles
They act as catalysts to generate singlet state oxygen from the dissolved oxygen available in the biological environment. These serve as light sources for activating NPs, which in turn emit light of high luminescence. To serve as a light source, NPs should have an emission spectrum similar to the photosensitizer. Moreover, NPs must be non-toxic, stable, and soluble in the biological environment (Li 2013). Active targeting NPs can be sub-classified by the mechanism of activation.
7.2.1 Photosensitizer bound to the surface of nanoparticles
Photosensitizer is covalently bound to the surface of NPs functionalized with amino groups, carboxyl groups on the surface to link it with the photosensitizer (Perni et al., 2011). The bacteria can easily bind to NPs which are then killed by exposure to the singlet oxygen produced by the photosensitizer (Perni et al., 2011). Rose Bengal-decorated silica NPs showed up to eight-orders-of-magnitude reduction in Gram-positive bacteria, Methicillin-resistant S. aureus, with photodynamic action (Guo et al., 2010). In another study, chitosan NPs functionalized with rose-bengal showed higher toxicity in E. faecalis biofilms and disruption of biofilm structure (Shrestha et al., 2014). In a recent study, Ce6-conjugated hollow ZnFe2O4 nanoparticles were investigated for antibacterial inactivation with low-irradiance light in the near-infrared range (Thakur et al., 2022). This study concluded that ZnFe2O4–Ce6 nanocarriers exhibited excellent antibacterial activities (greater than98 %) under low-irradiance light at 660 nm. Moreover, higher sensitivity was observed in Gram negative bacteria (E. coli) than Gram-positive cells. These studies clearly indicate the efficacy of PS-conjugated nanocarriers in combating antibiotic-resistant bacteria.
7.2.2 Carbon-based nanosystems
Carbon-based nanomaterials have received increased attention in PDT due to advantages such as ease of synthesis, dispersibility in water, nontoxicity, biocompatibility, manageable optical properties, and ability to exhibit antibacterial activity via a type II mechanism (Nie et al., 2020). These systems mainly include carbon-quantum dots and fullerenes. Carbon-quantum dots are considered to be safe and biocompatible and have an ability to generate ROS for PDT applications following light absorption. Carbon-quantum dots upon visible light illumination (λ ≥ 420 nm, 65 ± 5 mW/cm2; 60 min) efficiently eradicated E. coli and S. aureus by 6 log units (Nie et al., 2020). In a recent study, graphene quantum dots-loaded with curcumin as a photosensitizer were fabricated and tested for antibacterial activity in the presence of blue-light (405 nm) irradiance of 30 J cm−2. At a curcumin concentration of 100 µm, graphene quantum dots loaded with curcumin showed a ∼ 3.5 log10 enhanced CFU reduction against P aeruginosa, MRSA, E. coli and C. albicans. Fullerenes (Cn, n = 60, 70, 72, 76, 84, or 100) are allotropes of carbon consisting of closed-cage carbon molecules arranged in the shape of a soccer ball. They contain sp2 hybridized carbon atoms with fused rings of 5–7 atoms. Fullerenes transfer energy from incident radiation to the surrounding oxygen. They produce different reactive oxygen species such as 1O2, superoxide anions (O2.−), and free radicals (.OH) based on the solvents. With polar solvents, they produce free radicals (such as superoxide and hydroxyl radical) and with non-polar solvents, they produce singlet oxygen molecules (Chatterjee et al., 2008). Wiesner et al. showed that hydroxylated and polymer-coated fullerenes are better generators of singlet state oxygen when compared to titanium dioxide NPs (Brunet et al., 2009). Fullerenes possess several advantages over conventional photosensitizers such as: high photostability and resistance to photobleaching, the flexibility of functionalization, ability to undergo type I and type II pathways, higher ROS quantum yield, the possibility of oxygen-independent photo-killing by electron transfer, and broad-spectrum antimicrobial activity against bacteria and fungi (Qi et al., 2019). In a novel approach, BOPHY fluorophore ((bis(difluoroboron)-1,2-bis((1H-pyrrol-2-yl)methylene)hydrazine) was covalently attached to a fullerene C60 moiety through a pyrrolidine ring and tested for antimicrobial activity. BOPHY–fullerene C60 dyad (BP-C60) combination exhibited higher ROS production efficiency and photodynamic inactivation of bacteria such as S. aureus and E. coli (Gonzalez Lopez et al., 2022).
7.2.3 Upconversion nanoparticles
They are modified composites that convert low-energy light to high-energy light, which in turn activates the photosensitizer. They provide advantages such as deep tissue penetration of light and enables the delivery of photosensitizer to specific tissues (Chatterjee et al., 2008). Zinc oxide is a reliable semiconductor used as a photocatalytic antibacterial agent due to its biocompatible nature and low cost. When exposed to solar radiation, zinc oxide can produce electrons (e−) and photogenerated holes (h+) resulting in the generation of various reactive oxygen species (ROS) with antibacterial properties (He et al., 2014). However, zinc oxide has a wide bandgap (3.24 eV) that requires excitation by UV light. In an attempt to broaden the absorption range of zinc oxide, lanthanide-doped upconversion nanocrystals (UCNs) were coupled to permit absorption of NIR light and subsequently emit UV/Vis emissions through the upconversion process. Hybrid core–shell-shell nanocomposites made up of NaYF4:Yb,Tm core, amorphous SiO2 shell and ZnO layer showed NIR-driven antibacterial performance (Tou et al., 2017). More recently an integrated antibacterial nanohybrid approach has reported excellent bactericidal effect/biocompatibility through combining upconversion nanoparticles with silver nanoclusters. In this approach, upconversion nanoparticles and methylene blue molecules were encapsulated with silica microspheres via microemulsion and further silver ions (Ag+) decorated on the surface of silica spheres to produce UCNPs@SiO2(methylene blue)@AgNCs nanohybrids. These nanohybrids when irradiated at 980 nm for 10 min resulted in a 100 % killing rate of both Gram-positive S. aureus and Gram-negative E. coli (Liu et al., 2020). In this technique, the molecular photosensitizer can be excited using infrared light. This is advantageous as infrared light can penetrate deeper in biological tissues than visible and UV light. Moreover, infrared light would cause less damage in the surrounding tissue (Díaz-Moscoso 2012). Thus, nanocarrier systems in combination with PDT could be an interesting approach in clinical treatment of antibiotic-resistant bacteria.
8 Current challenges and future perspectives on the role of nanosystems in aPDT
PDT was initially developed as cancer therapy to destroy proliferating blood vessels. The use of nanosystems in PDT enables the delivery of photosensitizer near the tumor location and thus improves the treatment outcomes. For instance, liposomal benzoporphyrin derivative (Visudyne®) was approved for the treatment of choroidal neovascularization in 2000 (Fenton and Perry 2006). Delivery of photosensitizers using nanosystems helps evade physicochemical barriers and biological barriers to a large extent and enhance the therapeutic outcomes by overcoming the multidrug resistance (dos Santos et al., 2021). Now it has become apparent that PDT can be considered as a modality in the treatment of localized infections. Despite several pieces of literature evidence describing photodynamic inactivation of bacteria/virus, PDT is far from being considered as a standard therapy for infectious diseases (Sharma et al., 2012). This could be attributed to the lack of antimicrobial photosensitizers with optimum physicochemical properties and the inability to deliver them into infected tissues. Nevertheless, this approach still presents some limitations and, in this section, we will discuss the challenges and ways to improve future research in this area.
Several questions related to the use of nanosystems in aPDT remain unanswered. Choosing the right nanomaterial is a critical parameter that determines the efficiency of PDT. The selective accumulation of the photosensitizer in target cells is required to avoid damage to healthy cells. Therefore, photosensitizer molecules should be delivered using an appropriate delivery system designed to overcome microbial barriers such as cell walls and biofilms. Bacterial cell walls maintain a negative surface charge under physiological conditions, therefore, cationic NPs are capable of showing better electrostatic interactions for effective bacterial targeting (Gao et al., 2014). The use of pH/enzyme-responsive polymeric micelles and antibody-decorated NPs could be tested for systemic administration in controlling deeply localized infections such as peritonitis and periodontitis (Wang et al., 2021, Awad et al., 2022). For treating superficial infections, inorganic nanomaterials are better suited as they have a low safety profile unlike biodegradable NPs (Managa and Nyokong 2015). For example, silver NPs have shown promising results against microbes present in the oral cavity (Wang et al., 2019). Another approach for enhancing the treatment efficacy against bacterial infections would be to conjugate targeting ligands that are specifically found on bacterial cells to the surface of nanocarrier systems containing a photosensitizer. These ligands include vancomycin (targets peptidoglycans present on the surface of Gram-positive bacteria), polymyxin (targets lipopolysaccharides present on the surface Gram-negative bacteria, or zinc(II)-bis(dipicolylamine) (targets phosphatidylserine present on the surface of both Gram-positive and Gram-negative bacteria) (Lu et al., 2017). In one study, bacteria-targeting liposomes were prepared by conjugating two bacteria-targeting ligands (antimicrobial peptide WLBU2 and lectin Wheat Germ Agglutinin (WGA)) to the surface of temoporfin incorporated liposomes. WLBU2 appeared to be a better bacteria-targeting ligand than WGA in eradicating methicillin resistant S. aureus and P. aeruginosa (Yang 2012). However, the addition of such targeting ligands to the surface of nanosystems is an expensive affair when compared to the low price antibiotic therapy (Bartlett et al., 2013).
It is still unclear whether photosensitizer should be encapsulated inside nanosystems or covalently attached for better antimicrobial activity. Encapsulation of photosensitizers into nanoparticles improves singlet oxygen production by decreasing photobleaching and aggregation, while covalent linking of photosensitizers to the surface of nanoparticles prevents the premature release of photosensitizers in the media and subsequent loss of efficacy/appearance of side effects (Díaz-Moscoso 2012, Klausen et al., 2020). The side-effects related to premature release of the drug should be studied. The toxicity associated with nanosystems, especially non-biodegradable nanosystems, remains a grave matter of concern. The injudicious use of nanomaterials may lead to accumulation in the environment and this could result in unseen dangers (Khan et al., 2021). For instance, several metallic NPs such as iron, silver, platinum, palladium, gold, and metal oxide are found to cause damage to the cell membrane, DNA and proteins. Further, these particles tend to enter the bloodstream and reach the vital organs of the human body resulting in toxicity (Khan et al., 2021). Despite hurdles, researchers continue to publish data in support of PDT as an alternate therapy for the treatment of bacterial infections. In fact, photosensitizer-nanoformulations have not currently been granted regulatory approval to treat infectious diseases. More efforts are required to get closer to commercialization and clinical application. Nanotechnology-based PDT holds incredible potential for managing resistant bacterial infections, but the safe use of this procedure requires continued evaluation to ensure patient safety and protection to healthcare professionals. Since nanotechnology-based PDT has shown various advantages, it is reasonable to believe that this technique will be popular not only in cancer therapy, but also in treating skin diseases and microbial infections.
9 Conclusion
Despite the use of a wide variety of antibiotics, the treatment of microbial infection still faces several challenges due to the emergence of antibiotic resistance. As a result, PDT has emerged as one of the promising therapeutic options for microbial diseases in order to combat resistance by bacterial strains. The use of nanosystems such as liposomes, polymeric NPs, polymeric micelles, and various inorganic NPs have shown to enhance the antimicrobial action of photosensitizers. Nanocarriers can serve as a platform for improving the cellular uptake, bioavailability, distribution, and pharmacokinetics of photosensitizers. They offer several advantages when compared to free photosensitizer molecules. Nanocarriers increase the concentration of photosensitizer reaching the target cell and thereby increases the yield of singlet state oxygen inside the cell. New ways to enhance the effectiveness of aPDT by combining with nanocarriers are constantly being discovered. From the literature it is clear that a large amount of preclinical evidence exists in support of using aPDT in combination with nanocarrier systems. In the near future, nanoparticles that can better meet the complex biofilm microenvironment should be designed for enhanced therapeutic effect against drug resistant microbes. A majority of in vitro studies discussed in this review indicate the potential of nanotech-based PDT in the elimination of drug-resistant microbes. However, further examination into the long-term toxicity, pharmacokinetics, and pharmacodynamic properties of nanotech-based PDT through appropriate preclinical and clinical studies is required. To conclude, the combination of PDT and nanotechnology may offer new nanophotosensitizer formulations that are capable of treating both superficial and deep-seated infections.
10 Consent for publication
Authors give consent for information to be published in the Arabian Journal of Chemistry.
Author contributions
S.H.S.B., C.P., A.K.T, conceptualized and developed the idea; S.J., S.H.S.B., R.J.B. prepared the manuscript; C.G., S.J., S.H.S.B., A.K.T., C.P., and K.M.R, provided critical inputs and designs for figures. R.J.B., S.H.S.B., K.M.R., H.Y., and A.K.T edited the manuscript. A.K.T. supervised the study and provided funding support for the team.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Abdel-kader, M. H., 2016. The journey of PDT throughout history: PDT from Pharos to present.
- BODIPYs in antitumoral and antimicrobial photodynamic therapy: an integrating review. J. Photochem. Photobiol. C: Photochem. Rev.. 2019;40:21-48.
- [CrossRef] [Google Scholar]
- A nano phototheranostic approach of toluidine blue conjugated gold silver core shells mediated photodynamic therapy to treat diabetic foot ulcer. Sci. Rep.. 2021;11:24464.
- [CrossRef] [Google Scholar]
- pH-Responsive copolymer micelles to enhance itraconazole efficacy against Candida albicans biofilms. J. Mater. Chem. B. 2020;8:1672-1681.
- [Google Scholar]
- Oncologic photodynamic therapy photosensitizers: a clinical review. Photodiagnosis Photodyn Ther.. 2010;7:61-75.
- [CrossRef] [Google Scholar]
- Antimicrobial photodynamic therapy in the control of COVID-19. Antibiotics. 2020;9:320.
- [Google Scholar]
- Application of porphyrins in antibacterial photodynamic therapy. Molecules. 2019;24:2456.
- [CrossRef] [Google Scholar]
- Advances in photodynamic antimicrobial chemotherapy. J. Photochem. Photobiol. C: Photochem. Rev.. 2021;49:100452
- [CrossRef] [Google Scholar]
- Nanomaterials enabling clinical translation of antimicrobial photodynamic therapy. J. Control. Release. 2022;346:300-316.
- [CrossRef] [Google Scholar]
- Methylene blue-containing liposomes as new photodynamic anti-bacterial agents. J. Mater. Chem. B. 2017;5:2788-2797.
- [Google Scholar]
- Seven ways to preserve the miracle of antibiotics. Clin. Infect. Dis.. 2013;56:1445-1450.
- [Google Scholar]
- New cationic liposomes as vehicles of m-tetrahydroxyphenylchlorin in photodynamic therapy of infectious diseases. Mol. Pharm.. 2008;5:672-679.
- [CrossRef] [Google Scholar]
- Photosensitizers of the porphyrin and phthalocyanine series for photodynamic therapy. Chem. Soc. Rev.. 1995;24:19-33.
- [Google Scholar]
- Early and late onset side effects of photodynamic therapy. Biomedicines. 2018;6:12.
- [CrossRef] [Google Scholar]
- Laser and non-laser light sources for photodynamic therapy. Lasers Med. Sci.. 2002;17:173-186.
- [CrossRef] [Google Scholar]
- Photodynamic therapy: occupational hazards and preventative recommendations for clinical administration by healthcare providers. Photomed. Laser Surg.. 2013;31:398-407.
- [CrossRef] [Google Scholar]
- The present and future role of photodynamic therapy in cancer treatment. Lancet Oncol.. 2004;5:497-508.
- [CrossRef] [Google Scholar]
- Comparative photoactivity and antibacterial properties of C60 fullerenes and titanium dioxide nanoparticles. Environ. Sci. Tech.. 2009;43:4355-4360.
- [Google Scholar]
- Iron oxide mediated photothermal therapy in the second biological window: a comparative study between magnetite/maghemite nanospheres and nanoflowers. Nanomaterials (Basel). 2020;10:1548.
- [CrossRef] [Google Scholar]
- Light sources for photodynamic inactivation of bacteria. Lasers Med. Sci.. 2009;24:453-460.
- [CrossRef] [Google Scholar]
- Photodynamic inactivation of in vitro bacterial cultures from pressure ulcers. Acta Cir. Bras.. 2006;21(Suppl 4):32-35.
- [Google Scholar]
- Mechanisms in photodynamic therapy: part one—photosensitizers, photochemistry and cellular localization. Photodiagn. Photodyn. Ther.. 2004;1:279-293.
- [CrossRef] [Google Scholar]
- Bactericidal effects of different laser wavelengths on periodontopathic germs in photodynamic therapy. Lasers Med. Sci.. 2003;18:51-55.
- [Google Scholar]
- Nanoparticles in photodynamic therapy: an emerging paradigm. Adv. Drug Deliv. Rev.. 2008;60:1627-1637.
- [CrossRef] [Google Scholar]
- Chitosan nanoparticles for antimicrobial photodynamic inactivation: characterization and in vitro investigation. Photochem. Photobiol.. 2012;88:570-576.
- [CrossRef] [Google Scholar]
- Using nanoparticles to enable simultaneous radiation and photodynamic therapies for cancer treatment. J. Nanosci. Nanotechnol.. 2006;6:1159-1166.
- [Google Scholar]
- Antimicrobial effect of photodynamic therapy using high-power blue light-emitting diode and red-dye agent on Porphyromonas gingivalis. J. Periodontal. Res.. 2013;48:696-705.
- [CrossRef] [Google Scholar]
- Conjugation of superparamagnetic iron oxide nanoparticles and curcumin photosensitizer to assist in photodynamic therapy. Colloids Surf. B Biointerfaces. 2020;196:111297
- [CrossRef] [Google Scholar]
- Bactericidal effect of 0.95-mW helium-neon and 5-mW indium-gallium-aluminum-phosphate laser irradiation at exposure times of 30, 60, and 120 seconds on photosensitized Staphylococcus aureus and Pseudomonas aeruginosa in vitro. Phys. Ther.. 1999;79:839-846.
- [Google Scholar]
- Current clinical and preclinical photosensitizers for use in photodynamic therapy. J. Med. Chem.. 2004;47:3897-3915.
- [Google Scholar]
- Nepenthes khasiana mediated synthesis of stabilized gold nanoparticles: characterization and biocompatibility studies. J. Photochem. Photobiol. B Biol.. 2016;154:108-117.
- [Google Scholar]
- Soft versus hard nanoparticles in the delivery of aromatic macrocycles for photodynamic therapy of cancer. Int. J. Med. Biomed. Res.. 2012;1:12-23.
- [Google Scholar]
- Photodynamic therapy in cancer treatment - an update review. J. Cancer Metast. Treat.. 2019;5:25.
- [CrossRef] [Google Scholar]
- Nanophotosensitizers for cancer therapy: a promising technology? J. Phys.: Mater. 2021
- [Google Scholar]
- Near-infrared two-photon absorption in phthalocyanines: enhancement of lowest gerade-gerade transition by symmetrical electron-accepting substitution. J. Chem. Phys.. 2006;124:224701
- [Google Scholar]
- Improved photophysical and photochemical properties of thiopheneethoxy substituted metallophthalocyanines on immobilization onto gold-speckled silica nanoparticles. Photochem. Photobiol.. 2018;94:521-531.
- [CrossRef] [Google Scholar]
- Gold nanoparticles in biomedical applications: recent advances and perspectives. Chem. Soc. Rev.. 2012;41:2256-2282.
- [CrossRef] [Google Scholar]
- Inactivation of methicillin-resistant Staphylococcus aureus (MRSA) by liposome-delivered photosensitising agents. J. Photochem. Photobiol. B. 2006;83:98-104.
- [CrossRef] [Google Scholar]
- Effect of photosensitizers in chemical and biological processes: the MTO mechanism in photodynamic therapy. Biochem. Biophys. Res. Commun.. 1992;186:1032-1036.
- [Google Scholar]
- Effect of photosensitizers in chemical and biological processes: the MTO mechanism in photodynamic therapy. Biochem. Biophys. Res. Commun.. 1992;186:1032-1036.
- [CrossRef] [Google Scholar]
- Nanoparticle approaches against bacterial infections. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol.. 2014;6:532-547.
- [CrossRef] [Google Scholar]
- Development and characterization of erythrosine nanoparticles with potential for treating sinusitis using photodynamic therapy. Photodiagn. Photodyn. Ther.. 2015;12:9-18.
- [Google Scholar]
- Fluorescence spectroscopy of tissue: recovery of intrinsic fluorescence from measured fluorescence. Appl. Opt.. 1996;35:1780-1792.
- [CrossRef] [Google Scholar]
- Advanced noninvasive light-activated disinfection: assessment of cytotoxicity on fibroblast versus antimicrobial activity against Enterococcus faecalis. J. Endod.. 2007;33:599-602.
- [Google Scholar]
- Photosensitizers in antibacterial photodynamic therapy: an overview. Laser Ther.. 2018;27:293-302.
- [CrossRef] [Google Scholar]
- Liposomal formulations of photosensitizers. Biomaterials. 2019;218:119341
- [CrossRef] [Google Scholar]
- Gonzalez Lopez, E. J., A. M. Sarotti, S. R. Martínez, et al., 2022. BOPHY-Fullerene C60 Dyad as a Photosensitizer for Antimicrobial Photodynamic Therapy. Chemistry – A European Journal. 28, e202103884. https://doi.org/https://doi.org/10.1002/chem.202103884.
- Griesbeck, A. G., 1991. Essentials of Molecular Photochemistry. Von A. Gilbert und J. Baggott. Blackwell Scientific Publications, Oxford 1991. XII, 538 S. kartoniert £ 17.95. - ISBN 0-632-02429-1. Angewandte Chemie. 103, 1554-1555. https://doi.org/10.1002/ange.19911031143.
- Synthesis and physicochemical properties of polyacrylamide nanoparticles as photosensitizer carriers. Int. J. Pharm.. 2016;512:213-218.
- [Google Scholar]
- Halogenated phenotiazine as photoantimicrobial agent against Staphylococcus aureus. evaluation of the vehiculization in polymeric nanoparticles. Dyes Pigm.. 2019;170:107625
- [Google Scholar]
- New poly(acrylamide) nanoparticles in the development of third generation photosensitizers. Dyes Pigm.. 2021;184:108856
- [CrossRef] [Google Scholar]
- Rose Bengal-decorated silica nanoparticles as photosensitizers for inactivation of gram-positive bacteria. Nanotechnology. 2010;21:065102
- [Google Scholar]
- Rose Bengal-decorated silica nanoparticles as photosensitizers for inactivation of gram-positive bacteria. Nanotechnology. 2010;21:065102.
- [CrossRef] [Google Scholar]
- Photogenerated charge carriers and reactive oxygen species in ZnO/Au hybrid nanostructures with enhanced photocatalytic and antibacterial activity. J. Am. Chem. Soc.. 2014;136:750-757.
- [CrossRef] [Google Scholar]
- Silver-decorated, light-activatable polymeric antimicrobials for combined chemo-photodynamic therapy of drug-resistant bacterial infection. Biomater. Sci.. 2020;8:6350-6361.
- [Google Scholar]
- Synergistic chemical and photodynamic antimicrobial therapy for enhanced wound healing mediated by multifunctional light-responsive nanoparticles. Biomacromolecules. 2019;20:4581-4592.
- [CrossRef] [Google Scholar]
- A review of progress in clinical photodynamic therapy. Technol. Cancer Res. Treat.. 2005;4:283.
- [Google Scholar]
- Huang, L., T. Dai and M. Hamblin, 2010. Antimicrobial Photodynamic Inactivation and Photodynamic Therapy for Infections. Photodynamic Therapy. C. J. Gomer, Humana Press. 635: 155-173
- Photosensitized inactivation of microorganisms. Photochem. Photobiol. Sci.. 2004;3:403-405.
- [CrossRef] [Google Scholar]
- Photodynamic therapy in the treatment of microbial infections: basic principles and perspective applications. Lasers Surg. Med.. 2006;38:468-481.
- [CrossRef] [Google Scholar]
- Photodynamic therapy: novel third-generation photosensitizers one step closer? Br. J. Pharmacol.. 2008;154:1-3.
- [Google Scholar]
- Milestones in the development of photodynamic therapy and fluorescence diagnosis. Photochem. Photobiol. Sci.. 2007;6:1234-1245.
- [CrossRef] [Google Scholar]
- Fluorescence spectroscopy of turbid media: autofluorescence of the human aorta. Appl. Opt.. 1989;28:4286-4292.
- [Google Scholar]
- The potential exposure and hazards of metal-based nanoparticles on plants and environment, with special emphasis on ZnO NPs, TiO2 NPs, and AgNPs: A review. Environ. Adv.. 2021;6:100128
- [CrossRef] [Google Scholar]
- Light sources and dosimetry techniques for photodynamic therapy. Photochem. Photobiol.. 2020;96:280-294.
- [CrossRef] [Google Scholar]
- Design of photosensitizing agents for targeted antimicrobial photodynamic therapy. Molecules. 2020;25:5239.
- [Google Scholar]
- Multifunctional nanoparticle platforms for in vivo MRI enhancement and photodynamic therapy of a rat brain cancer. J. Magn. Magn. Mater.. 2005;293:404-410.
- [Google Scholar]
- Cellular impermeability and uptake of biocides and antibiotics in Gram-positive bacteria and mycobacteria. J. Appl. Microbiol.. 2002;92:46S-54S.
- [CrossRef] [Google Scholar]
- The effect of photodynamic therapy by gold nanoparticles on Streptococcus mutans and biofilm formation: an in vitro study. Lasers Med. Sci.. 2022;37:1717-1725.
- [CrossRef] [Google Scholar]
- Lee, Y.-E. and R. Kopelman, 2011. Polymeric Nanoparticles for Photodynamic Therapy. Biomedical Nanotechnology. S. J. Hurst, Humana Press. 726: 151-178
- Li, W. T., 2013. Nanoparticles for Photodynamic Therapy.
- Hybrids of upconversion nanoparticles and silver nanoclusters ensure superior bactericidal capability via combined sterilization. ACS Appl. Mater. Interfaces. 2020;12:51285-51292.
- [CrossRef] [Google Scholar]
- Structural modification of BODIPY: improve its applicability. Chin. Chem. Lett.. 2019;30:1815-1824.
- [Google Scholar]
- Surface charge-conversion polymeric nanoparticles for photodynamic treatment of urinary tract bacterial infections. Nanotechnology. 2015;26:495602
- [Google Scholar]
- Photodynamic therapy disinfection of carious tissue mediated by aluminum-chloride-phthalocyanine entrapped in cationic liposomes: an in vitro and clinical study. Lasers Med. Sci.. 2012;27:575-584.
- [Google Scholar]
- Nanoparticle targeting of Gram-positive and Gram-negative bacteria for magnetic-based separations of bacterial pathogens. Appl. Nanosci.. 2017;7:83-93.
- [CrossRef] [Google Scholar]
- Moraxella catarrhalis is susceptible to antimicrobial photodynamic therapy with Photofrin. Lasers Surg. Med.. 2014;46:712-717.
- [CrossRef] [Google Scholar]
- Evaluation of one- and two-photon activated photodynamic therapy with pyropheophorbide-a methyl ester in human cervical, lung and ovarian cancer cells. J. Photochem. Photobiol. B Biol.. 2014;132:102-110.
- [CrossRef] [Google Scholar]
- High-intensity narrow-spectrum light inactivation and wavelength sensitivity of Staphylococcus aureus. FEMS Microbiol. Lett.. 2008;285:227-232.
- [CrossRef] [Google Scholar]
- Physicochemical and antimicrobial photodynamic chemotherapy (against E. coli) by indium phthalocyanines in the presence of silver–iron bimetallic nanoparticles. Polyhedron. 2019;162:30-38.
- [CrossRef] [Google Scholar]
- A new strategy to destroy antibiotic resistant microorganisms: antimicrobial photodynamic treatment. Mini Rev. Med. Chem.. 2009;9:974-983.
- [Google Scholar]
- BODIPYs in PDT: a journey through the most interesting molecules produced in the last 10 years. Int. J. Mol. Sci.. 2022;23:10198.
- [Google Scholar]
- Photodynamic inactivation of Gram-negative bacteria: problems and possible solutions. J. Photochem. Photobiol. B Biol.. 1992;14:262-266.
- [Google Scholar]
- Biogenic gold nanoparticles decrease Methylene Blue photobleaching and enhance antimicrobial photodynamic therapy. Molecules. 2021;26:623.
- [CrossRef] [Google Scholar]
- Photodynamic antimicrobial chemotherapy activity of gallium tetra-(4-carboxyphenyl) porphyrin when conjugated to differently shaped platinum nanoparticles. J. Mol. Struct.. 2015;1099:432-440.
- [Google Scholar]
- Lasers and light sources for PDT: past, present and future. Photodiagn. Photodyn. Ther.. 2004;1:43-48.
- [CrossRef] [Google Scholar]
- Metallophthalocyanines for antimicrobial photodynamic therapy: an overview of our experience. J. Porphyrins Phthalocyanines. 2013;17:399-416.
- [Google Scholar]
- Meso-substituted cationic porphyrins as efficient photosensitizers of gram-positive and gram-negative bacteria. J. Photochem. Photobiol. B. 1996;32:153-157.
- [Google Scholar]
- Q Mesquita, M., C. J Dias, M. G. P M S Neves, et al., 2018. Revisiting Current Photoactive Materials for Antimicrobial Photodynamic Therapy. Molecules. 23, 2424. https://doi.org/10.3390/molecules23102424
- Porphyrin-fullerene C60 dyads with high ability to form photoinduced charge-separated state as novel sensitizers for photodynamic therapy. Photochem. Photobiol.. 2005;81:891-897.
- [CrossRef] [Google Scholar]
- Photoinactivation of bacteria. Use of a cationic water-soluble zinc phthalocyanine to photoinactivate both gram-negative and gram-positive bacteria. J. Photochem. Photobiol. B.. 1996;32:159-164.
- [CrossRef] [Google Scholar]
- Photodynamic inhibition of acetylcholinesterase after two-photon excitation of copper tetrasulfophthalocyanine. Lasers Med. Sci.. 2008;23:19-25.
- [CrossRef] [Google Scholar]
- Antimicrobial photodynamic and wound healing activity of curcumin encapsulated in silica nanoparticles. Photodiagn. Photodyn. Ther.. 2020;29:101639
- [CrossRef] [Google Scholar]
- Photodynamic therapy: porphyrins and phthalocyanines as photosensitizers. Aust. J. Chem.. 2008;61:741-754.
- [Google Scholar]
- Antimicrobial photodynamic therapy using a diode laser with a potential new photosensitizer, indocyanine green-loaded nanospheres, may be effective for the clearance of Porphyromonas gingivalis. J. Periodontal. Res.. 2013;48:591-599.
- [CrossRef] [Google Scholar]
- Nazir, S., K. Tahir, R. Irshad, et al., 2021. Photo-assisted inactivation of highly drug resistant bacteria and DPPH scavenging activities of zinc oxide graphted Pd-MCM-41 synthesized by new hydrothermal method. Photodiagnosis and Photodynamic Therapy. 33, 102162. https://doi.org/https://doi.org/10.1016/j.pdpdt.2020.102162.
- Carbon quantum dots: A bright future as photosensitizers for in vitro antibacterial photodynamic inactivation. J. Photochem. Photobiol. B Biol.. 2020;206:111864
- [CrossRef] [Google Scholar]
- Prevention of drug access to bacterial targets: permeability barriers and active efflux. Science. 1994;264:382-388.
- [Google Scholar]
- Nora, B., M. Paolo, I. Marcello, et al., 2017. Can Nanotechnology Shine a New Light on Antimicrobial Photodynamic Therapies? Photomedicine. T. Yohei. Rijeka, IntechOpen: Ch. 4.
- Research advances in the use of tetrapyrrolic photosensitizers for photodynamic therapy. J. Photochem. Photobiol. B Biol.. 2004;73:1-28.
- [CrossRef] [Google Scholar]
- Photophysical and photobiological processes in the photodynamic therapy of tumours. J. Photochem. Photobiol. B. 1997;39:1-18.
- [Google Scholar]
- The use of porphyrins for eradication of Staphylococcus aureus in burn wound infections. FEMS Immunol. Med. Microbiol.. 1997;19:307-314.
- [Google Scholar]
- New report calls for urgent action to avert antimicrobial resistance crisis. Joint News Release. 2019;29
- [Google Scholar]
- Sulfonated phthalimidomethyl aluminum phthalocyanine: the effect of hydrophobic substituents on the in vitro phototoxicity of phthalocyanines. Photochem. Photobiol.. 1991;53:323-327.
- [Google Scholar]
- Nanoparticles: their potential use in antibacterial photodynamic therapy. Photochem. Photobiol. Sci.. 2011;10:712-720.
- [Google Scholar]
- Pineiro, M., M. M. Pereira, A. d. A. Rocha Gonsalves, et al., 2001. Singlet oxygen quantum yields from halogenated chlorins: potential new photodynamic therapy agents. Journal of Photochemistry and Photobiology A: Chemistry. 138, 147-157
- New halogenated phenylbacteriochlorins and their efficiency in singlet-oxygen sensitization‡. Chem. A Eur. J.. 2002;106:3787-3795.
- [CrossRef] [Google Scholar]
- Natural photosensitizers in antimicrobial photodynamic therapy. Biomedicines. 2021;9
- [CrossRef] [Google Scholar]
- Novel nanomaterial-based antibacterial photodynamic therapies to combat oral bacterial biofilms and infectious diseases. Int. J. Nanomed.. 2019;14:6937-6956.
- [CrossRef] [Google Scholar]
- Microwave-assisted synthesis of 5,10,15,20-tetrakis(4-nitrophenyl)porphyrin and zinc derivative and study of their bacterial photoinactivation. Quart. J. Iran. Chem. Commun.. 2016;4:175-185.
- [Google Scholar]
- Antimicrobial photodynamic therapy: an overview. J. Indian Soc. Periodontol.. 2011;15:323-327.
- [CrossRef] [Google Scholar]
- Quantitative optical spectroscopy for tissue diagnosis. Annu. Rev. Phys. Chem.. 1996;47:555-606.
- [CrossRef] [Google Scholar]
- Evaluation of photodynamic therapy using a light-emitting diode lamp against< i> Enterococcus faecalis</i> in extracted human teeth. J. Endod.. 2011;37:856-859.
- [Google Scholar]
- Targeted intracellular delivery of photosensitizers to enhance photodynamic efficiency. Immunol. Cell Biol.. 2000;78:452-464.
- [Google Scholar]
- Ceramic-based nanoparticles entrapping water-insoluble photosensitizing anticancer drugs: a novel drug-carrier system for photodynamic therapy. J. Am. Chem. Soc.. 2003;125:7860-7865.
- [CrossRef] [Google Scholar]
- Photodynamic antimicrobial therapy. Cent. Eur. J. Biol.. 2010;5:400-406.
- [CrossRef] [Google Scholar]
- Santamaria, L. and G. Prino, 1972. List of the photodynamic substances. Res Prog Org Biol Med Chem. 3 Pt 1, XI-XXXV
- Light-emitting diodes as a light source for intraoperative photodynamic therapy. Neurosurgery. 1996;38:552-557.
- [Google Scholar]
- Light penetration in bladder tissue: implications for the intravesical photodynamic therapy of bladder tumours. BJU Int.. 2000;86:638-643.
- [Google Scholar]
- Photodynamic therapy for cancer and for infections: what is the difference? Isr J. Chem.. 2012;52:691-705.
- [CrossRef] [Google Scholar]
- Photodynamic therapeutics: basic principles and clinical applications. Drug Discov. Today. 1999;4:507-517.
- [CrossRef] [Google Scholar]
- Photoactivated rose bengal functionalized chitosan nanoparticles produce antibacterial/biofilm activity and stabilize dentin-collagen. Nanomed.: Nanotechnol. Biol. Med.. 2014;10:491-501.
- [CrossRef] [Google Scholar]
- Mechanisms of singlet-oxygen and superoxide-ion generation by porphyrins and bacteriochlorins and their implications in photodynamic therapy. Chemistry. 2010;16:9273-9286.
- [CrossRef] [Google Scholar]
- Non-porphyrinoid photosensitizers mediated photodynamic inactivation against bacteria. Dyes Pigm.. 2019;163:337-355.
- [CrossRef] [Google Scholar]
- Photodynamic therapy in the control of oral biofilms. Periodontology. 2011;2000(55):143-166.
- [CrossRef] [Google Scholar]
- The effect of charge on cellular uptake and phototoxicity of polylysine chlorin(e6) conjugates. Photochem. Photobiol.. 1997;65:723-729.
- [Google Scholar]
- Photophysicochemical properties and photodynamic therapy activity of chloroindium(III) tetraarylporphyrins and their gold nanoparticle conjugates. Porphyrin Sci. Women 2019:207-218.
- [Google Scholar]
- New two-photon activated photodynamic therapy sensitizers induce xenograft tumor regressions after near-IR laser treatment through the body of the host mouse. Clin Cancer Res.. 2008;14:6564-6573.
- [CrossRef] [Google Scholar]
- Photodynamic characterization and in vitro application of methylene blue-containing nanoparticle platforms. Photochem. Photobiol.. 2005;81:242-249.
- [Google Scholar]
- Encapsulation of methylene blue in polyacrylamide nanoparticle platforms protects its photodynamic effectiveness. Biochem. Biophys. Res. Commun.. 2008;369:579-583.
- [Google Scholar]
- Mechanisms of photodynamic inactivation of a gram-negative recombinant bioluminescent bacterium by cationic porphyrins. Photochem. Photobiol. Sci.. 2011;10:1659-1669.
- [CrossRef] [Google Scholar]
- Photosensitizer-conjugated hollow ZnFe2O4 nanoparticles for antibacterial near-infrared photodynamic therapy. ACS Appl. Nano Mater.. 2022;5:1533-1541.
- [CrossRef] [Google Scholar]
- Methylene blue-covered superparamagnetic iron oxide nanoparticles combined with red light as a novel platform to fight non-local bacterial infections: a proof of concept study against Escherichia coli. J. Photochem. Photobiol. B Biol.. 2020;209:111956
- [CrossRef] [Google Scholar]
- Targeted delivery of antibiotics to intracellular chlamydial infections using PLGA nanoparticles. Biomaterials. 2011;32:6606-6613.
- [CrossRef] [Google Scholar]
- Sequential coating upconversion NaYF4:Yb, Tm nanocrystals with SiO2 and ZnO layers for NIR-driven photocatalytic and antibacterial applications. Mater. Sci. Eng. C. 2017;70:1141-1148.
- [CrossRef] [Google Scholar]
- Endobronchial phototoxicity of WST 09 (Tookad), a new fast-acting photosensitizer for photodynamic therapy: preclinical study in the pig. Photochem. Photobiol.. 2003;78:124-130.
- [Google Scholar]
- The microenvironment effect on the generation of reactive oxygen species by Pd-bacteriopheophorbide. J. Am. Chem. Soc.. 2005;127:6487-6497.
- [Google Scholar]
- Photodynamic antimicrobial chemotherapy (PACT) J. Antimicrob. Chemother.. 1998;42:13-28.
- [Google Scholar]
- The emerging chemistry of blood product disinfection. Chem. Soc. Rev.. 2002;31:128-136.
- [Google Scholar]
- Rapid antibiofilm effect of Ag/ZnO nanocomposites assisted by dental LED curing light against facultative anaerobic oral pathogen Streptococcus mutans. ACS Biomater. Sci. Eng.. 2019;5:2030-2040.
- [Google Scholar]
- Bacterial infection microenvironment sensitive prodrug micelles with enhanced photodynamic activities for infection control. Colloid Interface Sci. Commun.. 2021;40:100354
- [CrossRef] [Google Scholar]
- Identification of singlet oxygen as the cytotoxic agent in photoinactivation of a murine tumor. Cancer Res.. 1976;36:2326-2329.
- [Google Scholar]
- Intracellular photodynamic therapy with photosensitizer-nanoparticle conjugates: cancer therapy using a ‘Trojan horse’. Photochem. Photobiol. Sci.. 2006;5:727-734.
- [Google Scholar]
- Wiederkehr, N., 1996. The aggregation behavior of Zinc-Tetracarboxy-Phthalocyanine and Its Spectral Sensitization on Titanium Dioxide Films.
- In vivo and post mortem measurements of the attenuation spectra of light in mammalian tissues. Photochem. Photobiol.. 1985;42:153-162.
- [Google Scholar]
- Spectral properties of single BODIPY dyes in polystyrene microspheres and in solutions. J. Fluoresc.. 2001;11:119-128.
- [Google Scholar]
- Photodynamic therapy of cancer: second and third generations of photosensitizers. Russ. Chem. Bull.. 1998;47:807-816.
- [CrossRef] [Google Scholar]
- Analytical model for extracting intrinsic fluorescence in turbid media. Appl. Opt.. 1993;32:3585-3595.
- [Google Scholar]
- Photodynamic inactivation of chlorin e6-loaded CTAB-liposomes against Candida albicans. Lasers Surg. Med.. 2013;45:175-185.
- [CrossRef] [Google Scholar]
- Yang, K., 2012. Application of liposomes for antimicrobial photodynamic therapy.
- Advance in photosensitizers and light delivery for photodynamic therapy. Clin. Endoscopy. 2013;46:7-23.
- [Google Scholar]
- Antimicrobial photodynamic therapy: latest developments with a focus on combinatory strategies. Pharmaceutics. 2021;13:1995.
- [CrossRef] [Google Scholar]
- Susceptibility of Streptococcus mutans biofilms to photodynamic therapy: an in vitro study. J. Antimicrob. Chemother.. 2005;56:324-330.
- [Google Scholar]
- Nanomaterial-based strategies in antimicrobial applications: progress and perspectives. Nano Res.. 2021;14:4417-4441.
- [CrossRef] [Google Scholar]
- Synthesis and photodynamic antimicrobial activity of amphiphilic meso-arylporphyrins with pyridyl moieties. Dyes Pigm.. 2020;181:108561
- [CrossRef] [Google Scholar]
- Synthesis, photochemical and photoinduced antibacterial activity studies of meso-Tetra(pyren-1-yl)porphyrin and its Ni, Cu and Zn complexes. Scient. Pharma.. 2010;78:767-789.
- [CrossRef] [Google Scholar]







