Translate this page into:
Feasibility of biofilm production capacity by Levilactobacillus brevis isolated from motal cheese and evaluation of biofilm resistance produced in vitro and in yogurt
⁎Corresponding authors. khodaparast@um.ac.ir (Mohammad Hossein Haddad Khodaparast), e.shamloo@yahoo.com (Ehsan Shamloo)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Traditional dairy products are a unique source which have been considered for the extraction of indigenous probiotic strains in recent years. In this study, biofilm formation power of Levilactobacillus brevis that isolated from Mutal traditional cheese were investigated. Survival was assessed during 21 days of storage time and at the presence of residues antibiotics as well as gastrointestinal conditions. The results showed after 120 min of treatment in high acidic conditions (pH 2.0), the survival rate decreased only 0.75 log CFU/mL in biofilm formed. The antibiotic susceptibility evaluated of probiotic to enrofloxacin, sulfadimidine, tetracycline, and oxytetracycline showed reducing the bacterial population in the biofilm form only 2.6 log. probiotic strain that isolated from indigenous dairy sources showed excellent resistance in the biofilm state. Therefore, extracting strong probiotic strains from indigenous resources, it can significantly improve functional products and fermentory engineering.
Keywords
Probiotics
Biofilm
Levilactobacillus brevis
Yogurt
Antibiotic residues
Indigenous dairy sources
1 Introduction
The nutritional importance and high profitability of lactic acid bacteria caused serious efforts to identify and isolate these bacteria, especially in developing countries. Studies conducted in recent years on indigenous products of different provinces of Iran show that the biodiversity of these bacteria is very high in different indigenous products, including dairy products (Granato et al., 2010; Nezhad et al., 2020). On the other hand, dairy products are the best carriers of probiotics. Probiotics are beneficial microorganisms that have health effects, and if consumed in sufficient quantities, they can improve the functioning of the digestive system (Bujňáková and Kmeť, 2012). If indigenous potential probiotics can be successfully identified, it will be useful and beneficial to consumers in several ways because the isolation environment is compatible with the food environment of many industries, especially dairy industry (Furukawa, 2015; Sadishkumar and Jeevaratnam, 2017). In addition, the genetic resources available in native ecosystems are excellent sources for the production of starters and probiotics. If new probiotics can be discovered from these sources, unique properties not found in commercial strains can be obtained. It is worth noting that most of the commercially available strains are genetically modified, which makes its use a controversial issue (Bujňáková and Kmeť, 2012). On the other hand, the presence of antibiotics in the food industry, especially among fermented foods such as dairy products, cheese and yogurt, creates adverse effects on the starter culture and probiotic bacteria (Nguyen et al., 2014).The survival of probiotics in different stages of production, storage, passing and surviving in the digestive system is still a challenging issue. In the last decade, bacterial biofilm has been considered as a natural phenomenon to increase the survival of probiotics (Speranza et al., 2020). It seems that bacterial biofilm can solve the problem of survival because this phenomenon is a simple, convenient and natural technique for the survival of bacteria in the face of environmental stress (Zeinab Rezaei et al., 2021a). In the biofilm structure, bacteria produce a strong network of extracellular polysaccharides. The hydrophobic polysaccharides present in this structure limit the entry and absorption of harmful substances into the biofilm network and protect bacteria from adverse effects (Zeinab Rezaei et al., 2021b).Therefore, inspired by the protective structure of biofilm against many environmental stresses, this study was designed with the aim of using this natural phenomenon to enhance probiotic bacteria isolated from native origin and increase their survival during storage, product processing and digestive tract.
2 Experimental
In this study, L. brevis that was previously isolated and identified from motal cheese by Azizi et al. were used (Azizi et al., 2017). Fresh cow's milk for yogurt production was obtained from the Faculty of Agriculture of Ferdowsi University of Mashhad. MRS broth and MRS agar (De Man, Rogosa, Sharpe, Merck KGA, Germany) were purchased. Tetracycline 500 mg (each tablet containing 500 mg tetracycline / Iranian Animal Drug Production Company), oxytetracycline (100 mg, each tablet containing 100 mg of oxytetracycline / Zagros Pharmaceutical Company), Enrofloxacin 10 % (Royal Daru Company / Each ml of the drug contains 100 mg of enrofloxacin) and sulfadimidine 2.5 (Each blus contains 2.5 g of sulfadimidine, Iranian Animal Drug Production Company) were obtained.
2.1 Preparation of biofilm in culture medium
One milliliter of L. brevis suspension (1.5 × 108 CFU/mL) inoculated with fresh MRS broth was dispensed in a 24 well microplate and incubated at 30 ℃ for 48 h. After incubation, the medium was poured, and the plates were washed twice with distelited water to remove planktonic cells (Aoudia et al., 2016).
2.2 Preparation of biofilm in milk
This method was developed by modeling the biofilm production in an MRS broth medium (Aoudia et al., 2016). For the preparation of biofilm in real food environments, milk was used as a substrate precursor. The polystyrene straight-sided jar with a volume of 150 mL with polypropylene screw cap was used. The polystyrene straight-sided jar with a volume of 150 mL with polypropylene screw cap was used. 2 mL of strain suspension (1.5 × 108 CFU/mL) inoculated with 18 mL of pasteurized fresh milk (3 % fat) and poured into container and incubated for 48 h at 30 ℃. Then sample was washed and the biofilm was used for yogurt preparation.
2.3 Microstructure
The three-dimensional structure of the biofilm was observed using a LEO1450VP scanning electron microscope (Germany). Biofilm samples were fixed using 2.5 % glutaraldehyde for 24 h at 4 ℃, then washed thrice for 15 min in 10 mM sodium cacodylate buffer by gentle mixing at room temperature and dehydrated in a graded ethanol series 15 min each at 50, 70, 80, 90 and 95, 2 × 15 min at 100 % and 3 × 15 min in t-butyl alcohol. Finally air-dried at room temperature. After sputter coating with gold, the biofilm sample was observed by SEM with resolution 2.5 nm and Maximum Voltage 35kv. Images were taken in different magnifications at a voltage of 20 kV (Kubota et al., 2009).
2.4 Preparation of probiotic yogurt
Sterile and homogenized bovine milk was heated at 92 °C for 12 min and rapidly cooled to 44 °C. The direct starter cultures (Micromilk S.R.I., Italia; 2 kg of batch starter/100 kg milk, consisting of Streptococcus thermophilus and Lactobacillus delbrueckii spp. bulgaricus) were added to milk and was transferred to container (150 mL) containing biofilm. The planktonic cells of L. brevis (1.5 × 108 CFU/mL) were inoculated in control group of samples and were incubated at 37 °C. After reached the pH about 4.5, the samples were kept at 4 °C for three weeks to assessment the viability (Li et al., 2017; Yangilar and Yildiz, 2018).
2.4.1 Properties of yogurt
The pH value of the yogurt was measured at regular time intervals (1, 7, 14, and 21 days) during storage at 4 °C. Each yogurt sample (1 g) was mixed and measured pH using a pH meter (Martini, Mi 151, China). Syneresis of yogurt was determined using a centrifugal method. 25 g of yogurt was centrifuged (at 1500 × g for 10 min) then measure the amount of whey separated, and the syneresis was expressed as a percentage of the amount of whey to the initial amount of yogurt (Domagała, 2009).
2.4.2 Sensory evaluation
Five points hedonic scale (from 1, very dislike to 5, extremely like) was used to evaluate the quality of yogurts such as appearance, texture, taste, and overall acceptance during the storage period of 1, 3, 7, 14, and 21 days (Singh and Muthukumarappan, 2008).
2.5 Enumeration of probiotic bacteria
Sterial dilutions of each sample was prepared.Then were plated on set MRS agar containing 10 mg/L of vancomycin and incubated in a plastic anaerobic jar with C type gas pack sachet (Merck KGaA, Darmstadt, Germany) at 37 °C for 48 h. The total number of viable bacteria was expressed as Log CFU/g (Li et al., 2017).
2.6 Viability in gastrointestinal condition
Potassium chloride (1.12 g/L), sodium chloride (2.0 g/L), calcium chloride (0.11 g/L), and potassium phosphate monobasic (0.4 g/L) were used for preparation Simulated gastric juice (SGJ). After sterilization at 121 °C for 15 min, pepsin (0.26 g/L) was added, and the pH was adjusted (∼2) by adding 1 N HCl. 1 g of the bacterial biofilm and 3 mL of the suspension containing free cells of L. brevis were separately added into the containers 30 mL of the gastric juice and digested on a shaker at a rate of 90 rpm at 37 °C. Bacterial survival was determined by the conventional plate counting method using an MRS agar plate in 0, 30, 60, 90, and 120 min. To assess survival in the gut, bovine bile salt (0.18 g/L) (Sigma-Aldrich, USA) and porcine pancreatin (1.95 g/L) were added to previous containers and adjusted the pH to 7.0 using 1 N NaHCO3. Viable bacteria were cultured in 60, 120, 180, and 240 min (Gebara et al., 2013; Madureira et al., 2011).
2.7 Determination of MIC
According to the active ingredient of each antibiotic, several concentrations (1, 2, 4, 8, 16, 32, 64, 128, 256, 512, 1024 µg mL-1) were prepared (Zhang et al., 2013). 180 µL of culture medium containing 1.5 × 108 CFU mL−1 from L. brevis was poured into each well. 20 µL of each antibiotic concentration was added and incubated for 48 h at 30 °C. After incubation, the culture medium was drained and washed twice with 0.5 mL of 150 mM NaCl solution. The microplate was then stained for 45 min with 1 mL of 0.05 % (v/v) of crystalline violet solution and washed twice. One mL of 96 % ethanol (v/v) was added, and the optical density was determined at 595 nm (Yangilar and Yildiz, 2018).
2.8 Viability of L. Brevis in biofilm
The viability of L. brevis was tested in the storage period (3 days) at 4 °C. 1 mL of biofilm solution was mixed with 9 mL of sterile peptone water (1 g/L). After sequential dilutions, appropriate dilutions were plated on set MRS. Then, they were incubated in an anaerobic jar with C type gas pack sachet (Merck KGaA, Darmstadt, Germany) at 37 °C for 72 h. The total counts of the viable bacteria were reported (Li et al., 2017).
2.9 Antibiotic susceptibility of L. Brevis in biofilm and planktonic forms
Fresh milk with was heated at 92 °C for 10 min and cooled to 42 °C. Then yogurt starter was added to it (3.6 % w/v). The last concentration of each antibiotic that the L. brevis could not grow in the previous step was prepared. It was then added to the milk and mixed thoroughly. 100 mL of antibiotic-contaminated milk was added to each container containing the biofilm. Also, in planktonic samples, 1 mL of the solution with 1.5 × 109 CFU/mL of L. brevis was added to 9 mL of milk (10 mL to 90 mL of antibiotic-contaminated milk). The control sample was yogurt containing biofilm without antibiotic. All samples were incubated at 42 °C till the pH reached 4.6 then stored at 4 °C to be assessed (Li et al., 2017; Yangilar and Yildiz, 2018).
2.10 Antibacterial activity of probiotic biofilm
L. brevis biofilm was formed in in a microplate with 24-well. After washed, 1 mL per well of fresh Brain Heart Infusion broth (BHI; Merck, Germany) was inoculated with 1.5 × 108 CFU/ml of each pathogenic bacteria (L. monocytogenes ATCC7644 and P. aeruginosa PTCC 1074) was dispensed and incubated for 48 h at 30 °C. After the incubation time, the medium was removed from each well, the microplates were washed. The number of L. monocytogenes and P. aeruginosa were counted by the spread plate method in selective media (Oxford-Listeria-Selective-Agar (Base) and Pseudomonas agar base, respectively). Control groups were included biofilm of L. monocytogenes and biofilm of P. aeruginosa Without the presence of biofilm L. brevis (Aoudia et al., 2016).
2.11 Antibacterial activity of planktonic cells
One ml per well of fresh MRS broth culture was inoculated with 1.5 × 108 CFU/ml of L. brevis and was dispensed in a 24-wells microplate. Subsequently, 1 mL of it was added per well of fresh BHI broth inoculated with 1.5 × 108 CFU/ml of each pathogenic bacteria (L. monocytogenes ATCC7644 and P. aeruginosa PTCC 1074) and incubated for 48 h at 30 °C. After washed, evaluation of microorganisms was performed by the spread plate method (Aoudia et al., 2016).
2.12 Statistical analysis
All the tests were performed in triplicate or more replications, and results were presented as mean values and the standard deviation. The one-way analysis of variance (ANOVA) was performed, and significant difference between treatment groups was determined with Duncan’s multiple range test at p < 0.05.using the statistical analysis system (SPSS Inc., Chicago, IL, USA).
3 Results and discussion
3.1 Microstructure of biofilm
The microstructure of the biofilm showed (Fig. 1) a coherent and three-dimensional network that, similar to a strong skeleton, was able to maintain this unique biological network. Probiotic bacteria in this coherent and natural structure have gained more resistance to environmental stresses such as changes in temperature, pH and the presence of antibiotic residues compared to their counterparts in the plankton state, so their survival rate has increased (Jones and Versalovic, 2009; Kubota et al., 2008; Rueda-Robles et al., 2022).
Scanning electron microscopy images of biofilm of L. brevis in (a, b) MRS agar and (c, d) milk.
3.2 Viability during storage time
The viability of probiotic bacteria in biofilm and planktonic form is compared in Fig. 2. The bacterial population in the biofilm showed an increase about 0.25 log during storage time, while in the planktonic form there was a 3.5 log decrease in the probiotic population.The results show the protective effect of biofilm on probiotics. There is no significant difference between the whole and pulverized biofilm, and this result can make the application of biofilm easier and more convenient on an industrial scale. Biofilm is unique structure that enable bacteria to survive against environmental challenges such as osmotic changes, metal toxicity, dehydration, radiation, host immunity, antimicrobial agents, and disinfectants (Z Rezaei et al., 2021). This significant and protective effect is due to the network structure consisting of hydrophilic and hydrophobic exopolysaccharides that exist in the biofilm (Zeinab Rezaei et al., 2021a). Survival of probiotic bacteria until consumption and in the digestive tract is of significant importance (Kalantarmahdavi et al., 2021). In this study, the viability of bacteria increased with the probiotic biofilm production method (González-Ferrero et al., 2018; Holkem et al., 2017).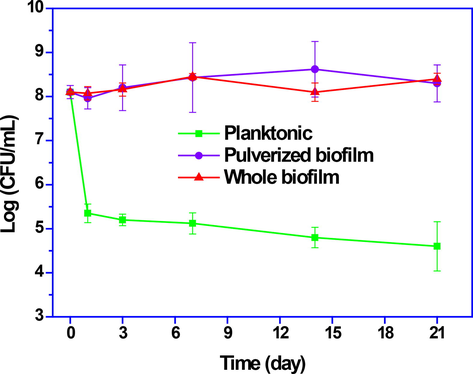
Viability of L. brevis during storage for 21 days at 4 °C.
3.2.1 Syneresis and pH of yogurt
The results of syneresis and pH are shown in Fig. 3. Yogurt samples containing pulverized biofilm and whole biofilm have a lower pH than yogurt samples containing plankton form of probiotics, which is probably due to the protective effect of biofilm on probiotics. Biofilm has been able to maintain the bacterial population because of its protective effect. The pH of yogurt is affected by the activity of yogurt bacteria becuse more lactic acid will be produced (Koohestani et al., 2018).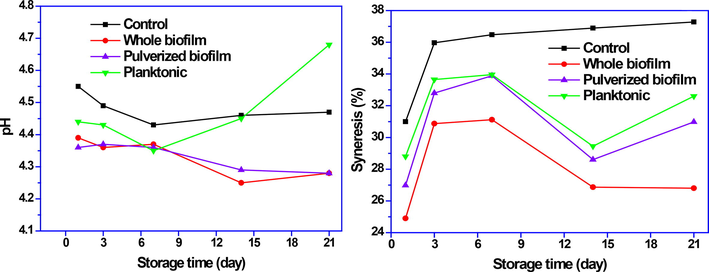
Physicochemical properties of probiotic yogurt with L. brevis.
The amount of syneresis of yogurt can have a significant effect on the appearance quality and overall acceptance of the product. In the dairy industry, compounds such as pectin and gum are used to reduce or prevent syneresis (Lee and Lucey, 2010). The study of the biofilm structure shows the presence of exopolysaccharide compounds in it (Terraf et al., 2016). Therefore, when probiotic bacteria are used in the form of biofilm in dairy products, in addition to the health benefits they can also technically effective in the product and reduce the consumption of stabilizers.
3.3 Sensory evaluation
The color of yogurt containing biofilm was significantly better than the control samples. It also smelled better.(Fig. 4, A). It can be concluded that the acidity of yogurt can affect its pleasant smell. On the other hand, the presence of polysaccharide and protein compounds in the biofilm causes an increase in yogurt solids, and as a result, more light scattering occurs and the color of yogurt becomes whiter (Mousavi et al., 2019). Yogurt texture is also an important parameter. The biofilm showed a more appropriate texture in yogurt and provided more acceptable feeling than control samples. This property was due to the extracellular polysaccharides in biofilm.
Sensory evaluation of probiotic yogurt made by planktonic and biofilm forms of probiotic.
(p < 0.05).Fig. 4, B). The present results showed that the taste of yogurt samples prepared with biofilm was significantly better than the control samples and this property was maintained during the storage time (Fig. 4, C). Biofilm contains about 6–7 % protein and polysaccharides, these compounds may affect other tissue properties (Mousavi et al., 2019; Terraf et al., 2016). In addition, the number of bacteria in yogurt has increased in the form of biofilm, which increases the production of lactic acid and improves the acidity and taste of the product. The new yogurt showed industrial potential and a higher overall acceptance score in sensory evaluation. (Fig. 4, D). Comparing the results of this study with other techniques such as nanoencapsulation and microencapsulation(Iravani et al., 2015; Yao et al., 2020), showed that biofilms do not cause negative sensory changes and can be used to improve the organoleptic properties of yogurt and similar products.
3.4 Gastrointestinal tolerance
In gastric conditions, the free cell viability of L. brevis was decreased to 3.2 Log CFU/mL after 30 min and decreased to undetectable levels after 90 min (Fig. 5a). Survival continued in the biofilm form in the intestine and by the fourth hour of digestion there were 106 viable bacteria in the intestine. (Fig. 5b). In comparison, the viability of L. brevis was greatly enhanced by biofilm in the high acid condition (pH 2.0) to the extent that only 0.25 Log CFU/mL reduction were observed in 120 min. The comparison of the results of this study with studies conducted on commercial strains shows the greater survival power of indigenous strains, which can be attributed to the intra-strain difference of probiotics. In recent years,studies were focused on the isolation and identification of probiotic bacteria from traditional dairy sources. On the other hand, more studies should be done on the annual economy and losses caused by starter imports (Zeinab Rezaei et al., 2021a). Even compared to different methods of increasing survival, including nanoencapsulation and encapsulation, the biofilm technique showed a higher power for the survival of native probiotic (Huq et al., 2017; Liao et al., 2019; Sohail et al., 2011).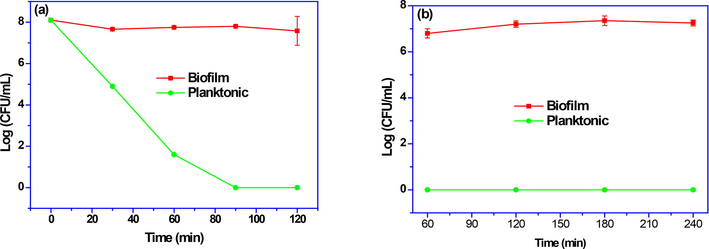
Viability of lactic acid bacteria biofilm during the exposure to (a) the simulated gastric condition and (b) the simulated intestine condition.
3.5 Antibiotic resistance
3.5.1 MIC
Enrofloxacin showed the most growth inhibitory effect on the probiotic strain. In the presence of other antibiotics, there was growth in different concentrations of antibiotics, the results of which are shown in the (Fig. 6a). Comparison of the results of this study with the study conducted by Rezaei et al. on commercial strains showed that the native strain of Lactobacillus brevis has significant resistance to the presence of tetracycline antibiotic compared to commercial strains (Zeinab Rezaei et al., 2021b).One reason for the existence of this greater inherent resistance can be considered intra-species differences and the type of source of bacterial isolation (Aoudia et al., 2016).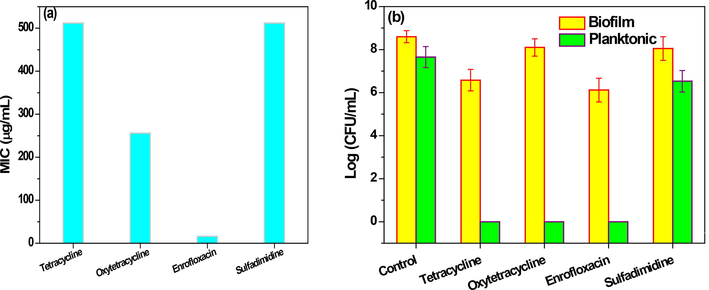
The minimum inhibitory concentration of antibiotic (MIC) in vitro (a), Biofilm and planktonic antibiotic susceptibility (b).
3.5.2 Antibiotic susceptibility
Antibiotic resistance of the indigenous strain of probiotic in the yogurt are shown in the Fig. 6. L. brevis in the planktonic form was the most vulnerable and was under the inhibitory effect of various antibiotics. On the contrary, there was significant resistance in biofilm form. Comparison of the results with the results of the study of commercial strains conducted by Rezaei et al. showed that the L. brevis in the biofilm form is capable of significant resistance in the presence of oxytetracycline with a survival difference of about 1.5 logs. These results can highlight the better survival power of L. brevis and their high resistance. Traditional dairy products are good candidates for the isolation of new probiotic strains. This group of natural functional foods contains a complex ecosystem of microorganisms (Neut et al., 2017). Several studies showed the sensitivity of probiotic bacteria to a variety of antibiotics. so far no study has been done on indigenous probiotic strains in the food environment. In 2014, the World Health Organization mentioned antibiotic resistance as a serious problem. On the other hand, many dairy food industries have been affected by the presence of antibiotic compound residues. Due to its coherent structure and rich in hydrophilic and hydrophobic compounds, probiotic biofilm can play a unique protective role in maintaining the probiotic bacterial population (Grossova et al., 2017). Therefore, probiotic biofilm due to its coherent structure and rich in hydrophilic and hydrophobic compounds, can play a unique protective role in maintaining the probiotic bacterial population in against antibiotics (Zeinab Rezaei et al., 2021b). In the post-antibiotic era, the biofilm approach is proposed as a new natural microbial technique for efficiency in increasing the survival of probiotics (Hobley et al., 2015; Holkem et al., 2017).
3.6 Antagonistic activity
In this study, the antagonistic power of the indigenous probiotic strain against two important food pathogens was investigated. The results are given in the Fig. 7. L. brevis probiotic strain was able to reduce the bacterial population of Listeria monocytogenes and Pseudomonas aeruginosa by 1.4 and 1.2 log, respectively, in planktonic form. In contrast, in the biofilm form of. L. brevis, the bacterial population of the pathogens Listeria monocytogenes and Pseudomonas aeruginosa showed a 4.9 and 3.2 log decrease in growth, respectively. Another useful approach of probiotics is to inhibit the growth of pathogens. Probiotics have been shown to reduce their infections and symptoms (Tung et al., 2009). Numerous studies have shown that indigenous strains separated from indigenous dairy products are capable of inhibiting some gastrointestinal diseases compared to commercial strains and have significant antibacterial properties (Z Rezaei et al., 2021). Organic acids such as lactic acids, short chain fatty acids, proteins, active peptides, bacteriocins and enzymes create many well -known roles in antagonistic activity (Chen et al., 2020). Comparison of the results of the antagonistic activity of commercial probiotic bacteria on food pathogens with the present study showed that the indigenous probiotic strain had more antagonistic activity either in the planktonic form or in the biofilm form. Considering that the biofilm mechanism showed more antagonistic power than the planktonic form of bacteria, it can be concluded that the biofilm structure has been able to maintain the microbial population due to its protective role and more antimicrobial compounds are produced by bacteria in the biofilm form (Zeinab Rezaei et al., 2021a).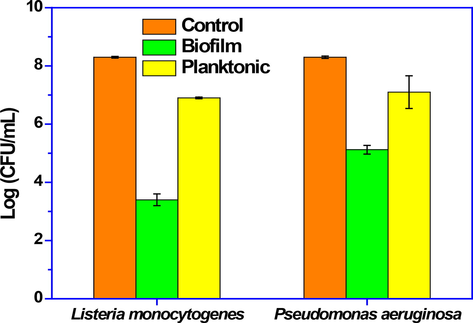
Antagonistic activity of L. brevis in biofilm and planktonic form on the growth of food pathogen.
4 Conclusions
The present study showed that indigenous dairy are potential and excellent probiotic sources. On the other hand, the form of probiotic biofilm showed the best ability to ferment and survival of the probiotic, leading to the improvement of bacteria resistance in simulated digestive conditions of the gastrointestinal tract, storage in the refrigerator and in the presence of residual antibiotics. Therefore, probiotic biofilm can be introduced as a superior technology for the preparation of probiotic products to fermentation and dairy industry. In addition, functional food productions can be improve by identifying and separating indigenous probiotic strains and introducing to the food industry.
Acknowledgments
Zeinab Rezaei was partially supported by a grant from Ferdowsi University of Mashhad (No.FUM-1400-26087). The authors would like to thank the Faculty of Ferdowsi University of Mashhad, for providing raw materials, the facilities, and the fnancial support that make this project possible.
Authors’ contributions
Z.R. A. S , E.SH and M.H.HK. contributed equally to this study. Z.R Created the original idea. Z.R. and F.N. expanded the idea. Z.R. and M.Y. carried out the experiments, and M.H.HK. and E.SH directed the project. All authors analyzed and interpreted the data and contributed to the writing of the manuscript. All authors read and approved the fnal manuscript.
Funding
This research is the result of a POST. DOC thesis with code 140026087, and this work was supported by the Ferdowsi University of Mashhad, Mashhad, Iran.
Availability of data and materials
The corresponding author could provide all experimental data on a valid request.
Declarations
Ethics approval and consent to participat: This article does not contain any studies with human participants or animals performed by any of the authors.
Consent for publication: Not applicable.
Competing interests: The authors declare that they have no competing interests.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Biofilms of Lactobacillus plantarum and Lactobacillus fermentum: effect on stress responses, antagonistic effects on pathogen growth and immunomodulatory properties. Food Microbiol.. 2016;53:51-59.
- [CrossRef] [Google Scholar]
- The biodiversity of Lactobacillus spp. from Iranian raw milk Motal cheese and antibacterial evaluation based on bacteriocin-encoding genes. Amb Express. 2017;7:1-10.
- [CrossRef] [Google Scholar]
- Functional properties of Lactobacillus strains isolated from dairy products. Folia Microbiol. (Praha). 2012;57:263-267.
- [CrossRef] [Google Scholar]
- Functional probiotics of lactic acid bacteria from Hu sheep milk. BMC Microbiol.. 2020;20:1-12.
- [CrossRef] [Google Scholar]
- Instrumental texture, syneresis and microstructure of yoghurts prepared from goat, cow and sheep milk. Int. J. Food Prop.. 2009;12:605-615.
- [CrossRef] [Google Scholar]
- Studies on formation, control and application of biofilm formed by food related microorganisms. Biosci. Biotechnol. Biochem.. 2015;79:1050-1056.
- [CrossRef] [Google Scholar]
- Viability of Lactobacillus acidophilus La5 in pectin–whey protein microparticles during exposure to simulated gastrointestinal conditions. Food Res. Int.. 2013;51:872-878.
- [CrossRef] [Google Scholar]
- Soybean protein-based microparticles for oral delivery of probiotics with improved stability during storage and gut resistance. Food Chem.. 2018;239:879-888.
- [CrossRef] [Google Scholar]
- Probiotic dairy products as functional foods. Compr. Rev. food Sci. food Saf.. 2010;9:455-470.
- [CrossRef] [Google Scholar]
- Probiotic biofilm on carrier surface: A novel promising application for food industry. Acta Aliment.. 2017;46:439-448.
- [CrossRef] [Google Scholar]
- Giving structure to the biofilm matrix: an overview of individual strategies and emerging common themes. FEMS Microbiol. Rev.. 2015;39:649-669.
- [CrossRef] [Google Scholar]
- Production of microcapsules containing Bifidobacterium BB-12 by emulsification/internal gelation. LWT-Food Sci. Technol.. 2017;76:216-221.
- [CrossRef] [Google Scholar]
- Alginate based nanocomposite for microencapsulation of probiotic: Effect of cellulose nanocrystal (CNC) and lecithin. Carbohydr. Polym.. 2017;168:61-69.
- [CrossRef] [Google Scholar]
- Technology and potential applications of probiotic encapsulation in fermented milk products. J. Food Sci. Technol.. 2015;52:4679-4696.
- [CrossRef] [Google Scholar]
- Probiotic Lactobacillus reuteri biofilms produce antimicrobial and anti-inflammatory factors. BMC Microbiol.. 2009;9:1-9.
- [CrossRef] [Google Scholar]
- Edible Films Incorporating with Lactobacillus plantarum Based on Sourdough, Wheat Flour, and Gelatin: Films Characterization and Cell Viability During Storage and Simulated Gastrointestinal Condition. Starch - Stärke. 2021;73:1-10.
- [CrossRef] [Google Scholar]
- Effects of cell-free supernatant of Lactobacillus acidophilus LA5 and Lactobacillus casei 431 against planktonic form and biofilm of Staphylococcus aureus. In: Veterinary Research Forum. Faculty of Veterinary Medicine. Urmia, Iran: Urmia University; 2018. p. :301-306.
- [Google Scholar]
- Biofilm formation by lactic acid bacteria and resistance to environmental stress. J. Biosci. Bioeng.. 2008;106:381-386.
- [CrossRef] [Google Scholar]
- Stress resistance of biofilm and planktonic Lactobacillus plantarum subsp. plantarum JCM 1149. Food Microbiol.. 2009;26:592-597.
- [CrossRef] [Google Scholar]
- Formation and physical properties of yogurt. Asian-Australasian J. Anim. Sci.. 2010;23:1127-1136.
- [CrossRef] [Google Scholar]
- Influence of Lactobacillus plantarum on yogurt fermentation properties and subsequent changes during postfermentation storage. J. Dairy Sci.. 2017;100:2512-2525.
- [CrossRef] [Google Scholar]
- Oligosaccharides as co-encapsulating agents: effect on oral Lactobacillus fermentum survival in a simulated gastrointestinal tract. Biotechnol. Lett.. 2019;41:263-272.
- [CrossRef] [Google Scholar]
- Protective effect of whey cheese matrix on probiotic strains exposed to simulated gastrointestinal conditions. Food Res. Int.. 2011;44:465-470.
- [CrossRef] [Google Scholar]
- Texture and sensory characterization of functional yogurt supplemented with flaxseed during cold storage. Food Sci. Nutr.. 2019;7:907-917.
- [CrossRef] [Google Scholar]
- Antibiotic susceptibility of probiotic strains: Is it reasonable to combine probiotics with antibiotics? Med. Mal. Infect.. 2017;47:477-483.
- [CrossRef] [Google Scholar]
- Technological characteristics of Lactobacillus spp. isolated from Iranian raw milk Motal cheese. LWT. 2020;133:110070
- [CrossRef] [Google Scholar]
- Tetracycline antibiotics and resistance mechanisms. Biol. Chem.. 2014;395:559-575.
- [CrossRef] [Google Scholar]
- Biofilm formation and antagonistic activity of Lacticaseibacillus rhamnosus (PTCC1712) and Lactiplantibacillus plantarum (PTCC1745) AMB Express. 2021;11:1-7.
- [CrossRef] [Google Scholar]
- A survey on biofilm formation of Lactobacillus rhamnosus (PTCC 1637) and Lactobacillus plantarum (PTCC 1745) as a survival strategy of probiotics against antibiotic in vitro and yogurt. J. Food Process. Preserv.. 2021;e15991
- [CrossRef] [Google Scholar]
- Biofilm Formation and Antibacterial Properties of Lactobacillus Isolated from Indigenous Dairy Products. J. food Qual. hazards Control. https:// 2021
- [CrossRef] [Google Scholar]
- Effect of Probiotics on Host-Microbiota in Bacterial Infections. Pathogens. 2022;11:986.
- [CrossRef] [Google Scholar]
- In vitro probiotic evaluation of potential antioxidant lactic acid bacteria isolated from idli batter fermented with Piper betle leaves. Int. J. Food Sci. Technol.. 2017;52:329-340.
- [CrossRef] [Google Scholar]
- Influence of calcium fortification on sensory, physical and rheological characteristics of fruit yogurt. LWT-Food Sci. Technol.. 2008;41:1145-1152.
- [CrossRef] [Google Scholar]
- Survivability of probiotics encapsulated in alginate gel microbeads using a novel impinging aerosols method. Int. J. Food Microbiol.. 2011;145:162-168.
- [CrossRef] [Google Scholar]
- Biofilm formation by potentially probiotic Saccharomyces cerevisiae strains. Food Microbiol.. 2020;87:103393
- [CrossRef] [Google Scholar]
- Biofilms of vaginal Lactobacillus reuteri CRL 1324 and Lactobacillus rhamnosus CRL 1332: kinetics of formation and matrix characterization. Arch. Microbiol.. 2016;198:689-700.
- [CrossRef] [Google Scholar]
- Prevention of Clostridium difficile infection with Saccharomyces boulardii: a systematic review. Can. J. Gastroenterol.. 2009;23:817-821.
- [CrossRef] [Google Scholar]
- Effects of using combined essential oils on quality parameters of bio-yogurt. J. Food Process. Preserv.. 2018;42:1-9.
- [CrossRef] [Google Scholar]
- Progress in microencapsulation of probiotics: A review. Compr. Rev. Food Sci. Food Saf.. 2020;19:857-874.
- [CrossRef] [Google Scholar]
- The association of biofilm formation with antibiotic resistance in lactic acid bacteria from fermented foods. J. Food Saf.. 2013;33:114-120.
- [CrossRef] [Google Scholar]







