Translate this page into:
Balanophora dioica ethanol extract ameliorates isoproterenol-induced myocardial injury by suppressing fibrosis, inflammation and apoptosis by regulating TLR4/MyD88/NF-κB signaling pathway
⁎Corresponding authors. tweizhuo@126.com (Weizhuo Tang), xiaoshu2397@163.com (Xiaoshu Zhang)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Balanophora dioica R.Br.ex Royle, belongs to the the Balanophoraceae family, has been documented as Chinese folk medicine in Lahu nationality in China for millennia. Myocardial injury is the main manifestation of cardiovascular disease. However, the role of Balanophora dioica ethanol extract (BDEE) in improving myocardial injury is unclear. The main purpose of this study is to clarify the pharmacological effect and molecular mechanisms of BDEE on myocardial injury. The major compounds of BDEE were analyzed by GC-MS and HPLC-FT-ICR-MS. The male C57BL/6 mice were randomly divided into 6 groups (n = 10), the control group, the isoproterenol (ISO) model group, ISO + 100, 200, and 400 mg/kg/d of BDEE groups, and the BDEE group (400 mg/kg/d). The heart weight/body weight was calculated, serum biochemistry markers CK, LDH, and myocardial enzyme indexes MDA, SOD, CAT, GSH-Px were measured by corresponding kits. Heart tissue histopathology was detected by hematoxylin-eosin and Sirius Red staining. Immunohistochemistry and western blotting were used to determine the protein expression associated with myocardial fibrosis, inflammation and apoptosis. TUNEL staining was used to evaluate the apoptosis level. The results demonstrated that BDEE can restore cardiac morphology and serum biochemical level, and restrain ISO-induced myocardial fibrosis by regulating TGF-β1/Smad3 pathway. Additionally, BDEE can reduce inflammation-related TLR4, MyD88, IRAK-1, TRAF-6, and NF-κB expression, and also inhibit cardiomyocyte apoptosis by regulating the expression of Bcl-2, Bax, Caspase-9 and Caspase-3. The above results indicated that BDEE improved ISO-induced myocardial injury by regulating myocardial fibrosis, inflammation and apoptosis, and it may be developed as a potential drug to prevent cardiovascular diseases.
Keywords
Balanophora dioica
Myocardial injury
Myocardial fibrosis
Inflammation
Apoptosis
TLR4/MyD88/NF-κB signaling pathway
- α-SMA
-
α-smooth muscle actin
- BDEE
-
Balanophora dioica ethanol extract
- CAT
-
catalase
- CK
-
creatine kinase
- COL1A1
-
Collagen Type I Alpha 1 Chain
- COL3A1
-
Collagen Type III Alpha 1 Chain
- DAPI
-
4′,6-Diamidino-2-phenylindole dihydrochloride
- GAPDH
-
Glyceraldehyde 3-phosphate dehydrogenase
- GC-MS
-
gas chromatography-mass spectrometry
- GSH-Px
-
Glutathione peroxidase
- H&E
-
Hematoxylin-Eosin
- HRP
-
Horseradish peroxidase
- HW/BW
-
heart weight /body weight
- IRAK-1
-
Interleukin 1 Receptor Associated Kinase 1
- ISO
-
isoproterenol
- LDH
-
lactate dehydrogenase
- MDA
-
malondialdehyde
- MyD88
-
Myeloid Differentiation Factor 88
- NC membrane
-
nitrocellulose filter membrane
- NF-κB
-
Nuclear Factor kappa-B
- ROS
-
reactive oxygen species
- Smad3
-
SMAD Family Member 3
- SOD
-
superoxide dismutase
- TGF-β1
-
transforming growth factor-β1
- TLR4
-
Toll Like Receptor 4
- TRAF-6
-
TNF Receptor Associated Factor 6
Abbreviations
1 Introduction
Cardiovascular diseases (CVDs) represent one of the intractable disorders that currently threatening human life and health and also are the leading cause of mortality and morbidity. In parallel, the CVDs mortality has gradually increased by 21.1 % from 2007 to 2017 (Collaborators, 2018). At present, the prevalence rate of cardiovascular disease in China is continuing to rise, and it is estimated that the number of CVD patients is 330 million, accounting for 23.57 % of the total number of people in China (Wang and Hu, 2023). Myocardial injury is a common pathology feature of CVDs, caused by a multitude of factors including common cardiac ischemia, hypoxia, myocardial degeneration, fibrosis, necrosis and other pathological changes. Serious lesions can lead to heart failure, such as arrhythmia, cardiogenic shock, and even sudden death (Long et al., 2020). ISO is a non-selective β-adrenergic receptor (β-AR) agonist, which enhances myocardial systolic force, accelerates heart rate, increases myocardial oxygen consumption, leads to myocardial ischemia and hypoxia, myocardial inflammation and fibrosis, and myocardial cell necrosis, thus causing myocardial injury by activating the β1 receptor on the myocardium (Nichtova et al., 2012). The myocardial injury model that was induced by ISO has the characteristics of short modeling time, stability and reliability, low cost, easy of imitation. Therefore, it is often used to simulate ischemic myocardial injury, drug-induced myocardial injury, myocardial infarction and other diseases (Saqib et al., 2022). A large number of studies have shown that the pathogenesis of myocardial injury is related to myocardial fibrosis, apoptosis, inflammation and oxidative stress (Farag et al., 2021). ISO can make the expression levels of fibrosis markers Collagen Type I Alpha 1 Chain (COL1A1), Collagen Type III Alpha 1 Chain (COL3A1) and α-smooth muscle actin (α-SMA) significantly up-regulated in myocardial injury mice (Sun et al., 2021a). ISO can also impact TGF-β1/Smad3 signaling pathway (Liu et al., 2019a) and TLR4/MyD88/NF-κB pathway (Xu et al., 2020), and influence proapoptotic protein (Bax and Caspase-3) and anti-apoptotic protein Bcl-2 (Fan et al., 2021). Drugs are an extremely important part of the comprehensive measures for the prevention and treatment of cardiovascular diseases. The vast majority of patients control the development of disease and prevent the occurrence of cardiovascular accidents by taking appropriate medications on a long- term. But at present, many drugs for the treatment of cardiovascular diseases have serious side effects (Hippisley-Cox and Coupland, 2010). Therefore, it is particularly important to find effective and low toxic drugs to improve or treat myocardial injury from natural plants.
Balanophora dioica R.Br.ex Royle, belongs to the Balanophoraceae family, which is widely distributed in Fujian, Yunnan, Sichuan and other places, and is one of the folk remedy of the Lahu nationality in Yunnan, China. It has a long history of drug use in Chinese folk. In the Compendium of Materia Medica written by Li Shizhen in Ming Dynasty, it is called “Ge-hua-cai” with liver protection. In recent years, it has often been used as a health food, and many Yunnan local people use Balanophora dioica to make soup and wine, because it has effects on analgesic and anti-inflammatory, and it was traditionally used to treat chronic hepatitis, impotence and other diseases. According to the National Compilation of Chinese Herbal Medicine and Chinese Materia Medica records, it is cold-natured and slightly bitter in taste. Then, it was used to cool blood and stop bleeding, clear heat and detoxification, treat cough, vomiting of blood, schizophrenia, hemorrhoids and other diseases (Editorial Board of Chinese Materia Medica of the State Administration of Traditional Chinese Medicine, 1999). It is found that Balanophora dioica contains several phytochemical compounds, including different phenolic acids (gallic acid, palmitic acid) (Wei et al., 2017), flavonoids (Trilobatin, Phlorizin), sterols (β-sitosterol, taraxasterol) (Tao et al., 2012), phenylpropanoids (Coniferin), lignans (isolariciresinol, burselignan) and other compounds (Ho et al., 2010, Pan et al., 2008). The diversity of its compounds indicates that Balanophora dioica contains a variety of biological activities. Modern pharmacological studies have confirmed that Balanophora dioica has the effect on anti-inflammation, analgesic (Ruan et al., 2003). Inflammation is closely related to myocardial injury and is an important factor leading to myocardial injury. Therefore, it can be speculated that BDEE may also play a role in inflammation during the pathological process of myocardial injury, so as to improve or prevent cardiovascular disease. However, the molecular mechanism of BDEE improving myocardial injury has not been reported. In this study, the protective effect and mechanism of BDEE against ISO-induced myocardial injury were discussed, which provides a theoretical basis for the development of cardioprotective drugs and also offers the possibility of discovering Balanophora dioica.
2 Materials and methods
2.1 Chemicals and reagents
Balanophora dioica was collected from Yunnan Province of China in 2022 (Zhang et al., 2008), kept in desiccators and authenticated by Professor Jincai Lu of the Shenyang Pharmaceutical University. The voucher specimen (No. 20220126) was deposited in the Herbarium of Shenyang Pharmaceutical University (Shenyang, China). ISO was purchased from Sigma (America), CK, LDH, SOD, CAT, GSH-Px and MDA kits were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China), Anti-caspase9 Rabbit pAb, TGF-β1 was purchased from Wanleibio (Shenyang, China). Anti-GAPDH Rabbit pAb was purchased from Servicebio (Wuhan, China). Other antibodies were all obtained from Beyotime Biotechnology (Shanghai, China).
2.2 Preparation and GC-MS, HPLC-FT-ICR-MS analysis of BDEE
2.2.1 The preparation of BDEE
Balanophora dioica (1000 g), the dried whole plant (dry in the shade), was extracted using 75 % ethanol (1:10, w/v) by reflux extraction for 1 h each time, and extracted totally twice (Wei et al., 2019, Xia, 2023). Then the extraction solvents were combined and filtrated, followed by freeze-drying (-50℃, 24 h) to obtain BDEE.
2.2.2 The GC-MS analysis of BDEE
The BDEE was analyzed by using gas chromatography-mass spectrometry (GC-MS) in an Agilent 8890 GC System with a mass spectrometer Agilent 5977B GC/MSD. The capillary column used was Agilent 122-1732 DB-17 (30 m × 0.25 mm × 0.25 μm). The BDEE (1.0 μL) was injected into the sample injector (280℃). Helium gas was used as a carrier gas at a constant flow rate of 12 mL/min and the injection mode split was 10:1. The oven temperature was programmed at 40℃-10 min hold and it was raised to reach 180℃-10 min hold at a rate of 10 ℃/min, then it was raised to reach 260℃-10 min hold at a rate of 10 ℃/min, and finally it was raised to reach 280℃-10 min hold at a rate of 10 ℃/min. The scan range of MS was 50–300 m/z, and the total time of MS was 64 min. The compounds shown in the chromatogram were computed by the total areas with the retention time. The other characteristics of the identified compounds and structure of the major compounds were established. The peak area to total area ratio and electronic libraries (standard mass spectral library, NIST 11) were used to determine the identification of detected substances in the BDEE.
2.2.3 The HPLC-FT-ICR-MS analysis of BDEE
2 mg BDEE was taken and placed in a centrifuge tube, 2 mL of chromatographic methanol was added. Then, it was treated with ultrasound for 10 min to make it fully dissolved. 1 mL of filtrate was taken and filtered with 0.45 μm microporous filter membrane. The filtrate obtained was used for HPLC analysis. The compounds of BDEE were determined by an Agilent 1260 HPLC system coupled with a Bruker Solarix 7.0 T FT-ICR-MS system. The separation of HPLC was by C18 column (1.8 μm × 150 mm × 2.1 mm, Welch, China) at 25℃. Elution was performed with a mobile phase of A (ddH2O) and B (methanol). The procedure used a flow rate of 400 μL/min and an injection volume of 5 μL to perform gradient elution as follows: 0–8 min (30–50 % B), 8–35 min (50–62 % B), 35–40 min (62–100 % B). The analytes were monitored by mass spectrometry with an electrospray ion source operating in the positive ion mode and negative ion mode. The optimized parameters were as follows: the nebulizer gas pressure was 4.0 bar. The dry gas flow rate was 8.0 L/min and the dry gas temperature was 200℃. The ion accumulation time was 0.15 s and the TOF was 0.6 ms. The capillary voltage was 4.5 kV and the end plate offset was 500 V. The full-scan MS data was 100–1000 m/z range. The collision energy was 14.0 V initially or it can be adjusted according to the fragments. FT-MS control, Bruker Compass-Hystar and Data Analysis Software (Bruker, Germany) were used to control the equipment and for data acquisition and analysis.
2.3 Animal studies
Sixty C57/BL6 mice (male, 6 weeks old, 18–22 g) were purchased from Liaoning Changsheng Biotechnology Co., Ltd, license number: SCXK (Liao) 2020–0001. The study adhered strictly to the National Institutes of Health Guide for the care and use of laboratory animals and was approved by the Laboratory Animal Committee of Shenyang Pharmaceutical University. All mice were kept at a temperature of 23 ± 1℃ and a relative humidity of 50–60 %, with a 12 h light/dark cycle, and with free access to water and food. After 1 week of acclimatization, the animals were randomly divided into 6 groups (10 in each group). The normal control group was given 0.9 % saline (10 mg/kg/d) by intragastric injection and subcutaneous injection. The ISO model group was given 0.9 % saline by gavage and subcutaneous injection of ISO (10 mg/kg/d) to induce myocardial injury for 21 days (Han et al., 2020, Xue et al., 2021, Zhang et al., 2023). 100, 200 and 400 mg/kg/d of BDEE were gavaged in the BDEE low-dose group, BDEE medium-dose group and BDEE high-dose group, and ISO (10 mg/kg/d) was injected subcutaneously. The BDEE drug group was given 400 mg/kg/d of BDEE by gavage. The experiments in each group lasted for 21 days. On the 22nd day, the mice were executed at anesthesia. Eyeballs were taken for blood. The mouse was fixed with one hand, and the eye skin on the blood side was gently pressed to make the eyeball congested and protruding. The eyeball was picked up with elbow forceps and quickly removed. At the same time, the mouse's heart was lightly pressed to increase the rate at which the hearts pumped blood. When the blood ran out, the mice were killed by dislocating. The hearts and livers were taken for follow-up experiments. The myocardial tissue of the left ventricle of mice was taken, fixed with 4 % paraformaldehyde, paraffin-embedded, and other myocardial tissue were stored at −80℃ for subsequent experiments. The blood was removed from the heart and liver in normal saline, dried with filter paper, weighed, and the heart index HW/BW were calculated. Then the whole blood was centrifuged at 3500 r/min for 15 min, and the supernatant was carefully sucked out and stored at −80℃ for subsequent experiments.
2.4 Histopathological examination
Take the embedded paraffin blocks and cut into 4 μm sections using standard histological methods. Left ventricular sections were stained with H&E and Sirius red. Liver sections were stained with H&E and Masson.
2.5 Biochemical index analysis
Serum LDH and CK activities were measured with LDH and CK kits. 0.1 g of the heart tissue was mixed in normal saline at a ratio of 1:9, and the supernatant fluid was taken after centrifugation at 3500 r/min for 15 min for the detection of oxidative stress markers such as SOD, MDA, CAT and GSH-Px. These indicators were achieved using the corresponding kit according to the instructions.
2.6 Immunohistochemistry
The protein expression of COL1A1, α-SMA and TLR4 in the heart was determined by immunohistochemistry. The paraffin-embedded of the left ventricle of mice was taken, and sliced. After dewaxing paraffin sections to water, the slices were immersed in pH 6.0 citric acid resistant solution and placed in microwave oven for antigen repair. The following experimental steps were as follows: natural cooling, block endogenous peroxidase, serum seal, adding primary antibody, adding secondary antibody, DAB color rendering, overdyed nucleus, and the results are interpreted under a white light microscope (Nikon, E100).
2.7 TUNEL staining
The paraffin-embedded of the left ventricle of mice was taken, according to the TUNEL kit requirements for reaction. Rinsing, dehydration, sealing, fluorescence microscope observation. Normal cardiomyocytes showed blue, and apoptotic cells showed green. The apoptosis rate of myocardial cells in each group was obtained by Image analysis software.
2.8 Western blotting
Myocardial tissues of mice were taken and weighed, and they were lysed with lysis solution (RIPA: Phosphoproteinase inhibitor A: Phosphoproteinase inhibitor B: PMSF = 100:1:1:1), then were grinded using a tissue grinder. The homogenates were centrifuged at 12,000 g, −4℃ for 15 min to collect the supernatant. Protein content in each group were determined by BCA kit. We obtained protein samples through adding buffered solution, boiling for 10 min after mixing. Then the protein in each group was separated by SDS-polyacrylamide gel electrophoresis. Transferring of separated proteins onto nitrocellulose filter membrane (NC membrane). blocking with 5 % skimmed milk powder at room temperature for 1.5 h. NC membrane was incubated with primary antibody overnight at 4℃. Then wash the membrane with TBST at 100 r for 5 min a total of 5 times to remove unbound primary antibodies. Incubation with horseradish peroxidase-labeled goat anti-mouse (1: 2000) and anti-rabbit antibody (1:1000) for 1 h. Washing membrane with TBST 5 times, 5 min/time. Finally, the ECL luminescence kits were used, the gray value was quantified by image analysis software image-J.
2.9 Statistical analysis
All the data were presented as the mean ± SEM and were analyzed with one-way ANOVA using GraphPad Prism version 9 (GraphPad Software Inc., San Diego, CA, USA). The data were statistically significant when P < 0.05.
3 Results
3.1 Dentification of main compounds in BDEE
GC–MS analysis results indicated that BDEE mainly contains 15 compounds (Fig.S1 and Table S1), while eight components identified from BDEE by HPLC-MS at positive ion mode (Fig.S3 and Table S2), and 17 components identified by HPLC-MS at negative ion mode (Fig.S4 and Table S3). The major components are luteolin-7-O-β-D-glucoside, 1-O-(E)-caffeoyl-3-O-galloyl-β-D-glucopyranose, palmitic acid, phloridzin, trilobatin, quercimeritrin, and ethyl brevifolin carboxylate. The information on all preliminarily identified compounds is in supporting information. Therefore, we speculate that phenols and flavonoids of BDEE maybe play a role in improving ISO-induced myocardial injury.
3.2 BDEE improves myocardial injury induced by ISO
In this study, the heart of the control group was smooth, bright red in color and normal in shape and size, while the heart of the ISO group was dark red in color and significantly increased in size. Compared with the ISO group, the heart of the BDEE administration group was close to normal in color and significantly reduced in size (Fig. 1A). The HW/BW in the ISO group was significantly higher than that in the control group, while the HW/BW in the BDEE group was significantly reduced in a dose-dependent manner (Compared with the Con group, the ISO model group increased HW/BW by 56.12 %, compared with the ISO model group, the ISO + 100, 200, 400 mg/kg BDEE group decreased by 18.06 %, 26.19 %, 34.55 %, respectively), suggesting that BDEE could alleviate ISO-induced myocardial hypertrophy (Fig. 1B). Histopathology indicated that the control group showed normal myocardial tissue structure by H&E staining, while the ISO group showed inflammatory cell infiltration vacuoles and interstitial edema, which were improved after BDEE administration. And the myocardial fibrosis was observed by Sirius red staining. The results showed that the degree of myocardial fibrosis in the ISO group was significantly higher than that in the control group, while the degree of myocardial fibrosis in the BDEE group was significantly improved (Fig. 1C). These results suggest that BDEE has a myocardial protective effect. Liver tissue H&E and Masson staining showed that BDEE had no drug toxicity compared with the control group (Fig. 1D).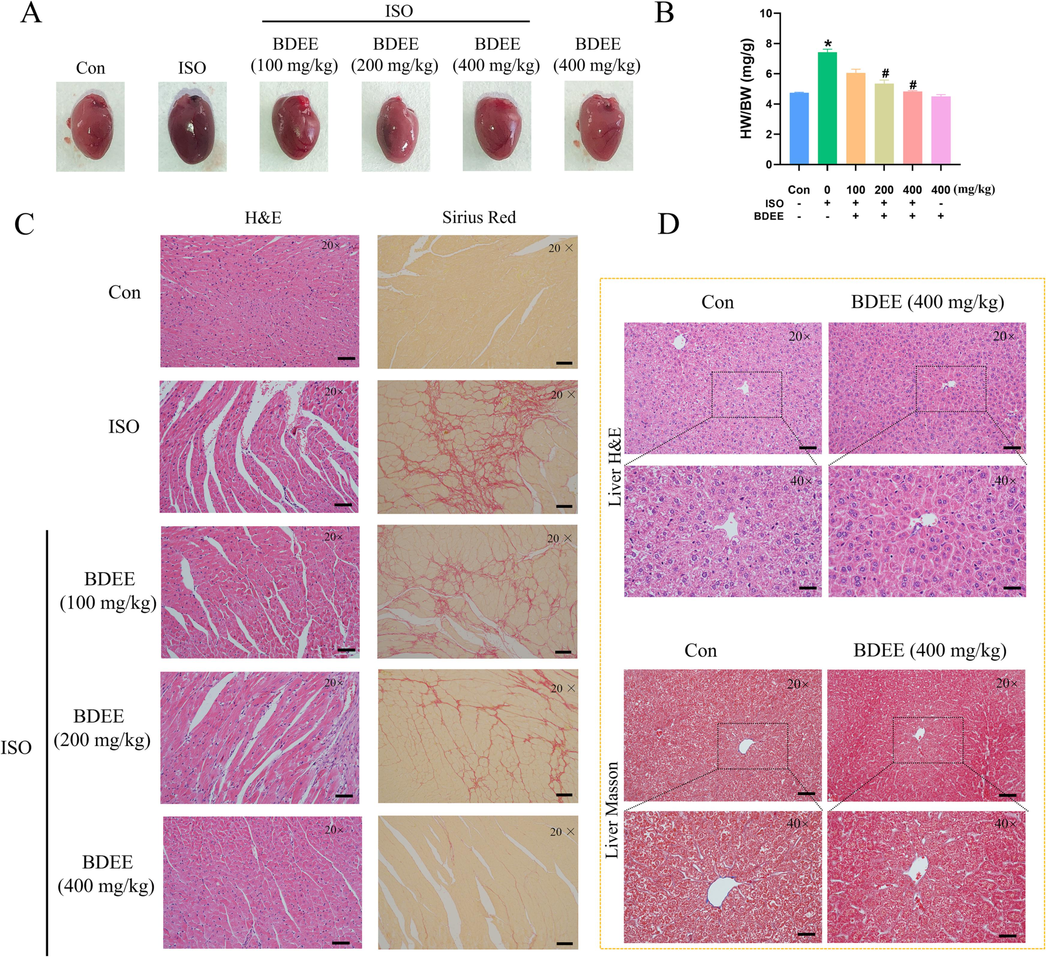
BDEE improves myocardial injury induced by ISO. (A) Morphology of the hearts of mice in each group. (B) The HW/BW were calculated to determine the effects of BDEE. (C) H&E staining and Sirius Red staining of heart sections, magnification: 20 ×, scale bar = 50 μm. (D) Masson staining and H&E staining of liver section, magnification: 20 × and 40 ×, scale bar = 50 μm. Values are given as mean ± SEM, n = 6. *P < 0.05 compared with Con group; #P < 0.05 compared with ISO group.
3.3 BDEE reduces ISO-induced oxidative stress
When the body produces too much reactive oxygen species (ROS) or clears them slowly, it can cause damage to cells and tissues. The heart is one of the organs more strongly affected by ROS. ISO can produce excessive free radicals through oxidative stress and lead to myocardial injury. Its morphological changes and energy metabolism are similar to that of human myocardial ischemia. In this process, SOD activity decreased, lipid peroxidation increased, and MDA content increased. In the process of oxidative stress, myocardial cells rupture or membrane permeability increases, so that CK, LDH and other enzymes exudate, resulting in increased enzyme activity in the serum of myocardial injury mice.
The results of this experiment showed that BDEE could significantly reduce the leakage of CK and LDH (Fig. 2A and B) (Compared with the ISO model group, the ISO + 100, 200, 400 mg/kg BDEE group decreased CK by 18.35 %, 30.39 %, 58.61 %; the ISO + 100, 200, 400 mg/kg BDEE group decreased LDH by 11.71 %, 27.05 %, 55.25 %, respectively), inhibit the accumulation of lipid peroxidation metabolite MDA in myocardium (Fig. 2C) (Compared with the ISO model group, the ISO + 100, 200, 400 mg/kg BDEE group decreased MDA by 17.51 %, 38.69 %, 66.16 %, respectively), and increase the levels of SOD, CAT and GSH-Px (Fig. 2D-F) (Compared with the ISO model group, the ISO + 100, 200, 400 mg/kg BDEE group increased SOD by 12.17 %, 37.01 %, 41.15 % and increased CAT by 12.71 %, 21.51 %, 55.25 % and increased GSH-Px by 7.22 %, 12.15 %, 40.69 %, respectively). It is suggested that it can reduce ISO-induced oxidative stress, stabilize the cell membrane and protect the myocardium of ISO-induced myocardial injury in mice.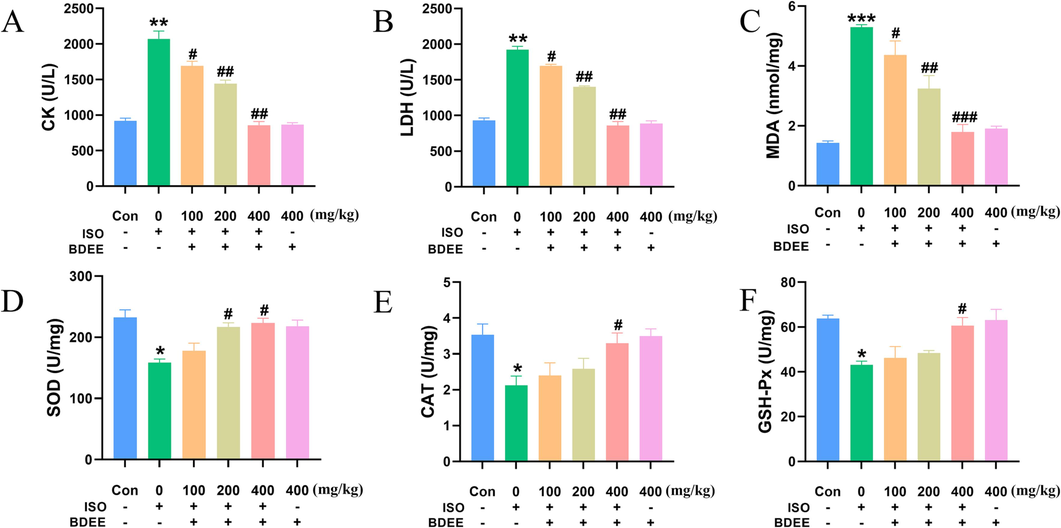
BDEE reduces ISO-induced oxidative stress. Effects of BDEE on the levels of CK (A), LDH (B). Effects of BDEE on concentration of MDA and activities of SOD, CAT, GSH-Px. (C-F). Note: The “+” means to give the drug, and “-” means not to give the drug. The results are presented as means ± SEM, n = 6. ***P < 0.001, **P < 0.01, *P < 0.05, compared with Con group; ### P < 0.001, ##P < 0.01, #P < 0.05, compared with ISO group.
3.4 BDEE inhibits myocardial fibrosis induced by ISO
The immunohistochemical results of COL1A1 and α-SMA were shown in Fig. 3A-B. Compared with the control group, the expressions of COL1A1 and α-SMA were increased in the model group, but decreased in a dose-dependent manner after BDEE administration. Western blotting results were shown in Fig. 3C-G. Compared with the control group, the protein expressions of COL1A1, COL3A1, α-SMA, TGF-β1, P-Smad3/Smad3 in myocardial cells in model group were significantly increased. (Compared with the Con group, the ISO model group increased the protein expression levels of COL1A1, COL3A1, α-SMA, TGF-β1, P-Smad3/Smad3 by 2.05, 2.10, 1.01, 1.55, and 1.51 times respectively). Compared with the model group, the protein expression levels of COL1A1, COL3A1, α-SMA, TGF-β1, P-Smad3/Smad3 in the BDEE administration group were decreased in a dose-dependent manner. It was consistent with the literature and suggested that BDEE may improve ISO-induced myocardial fibrosis in mice through TGF-β1/Smad3 pathway (Sun et al., 2021b).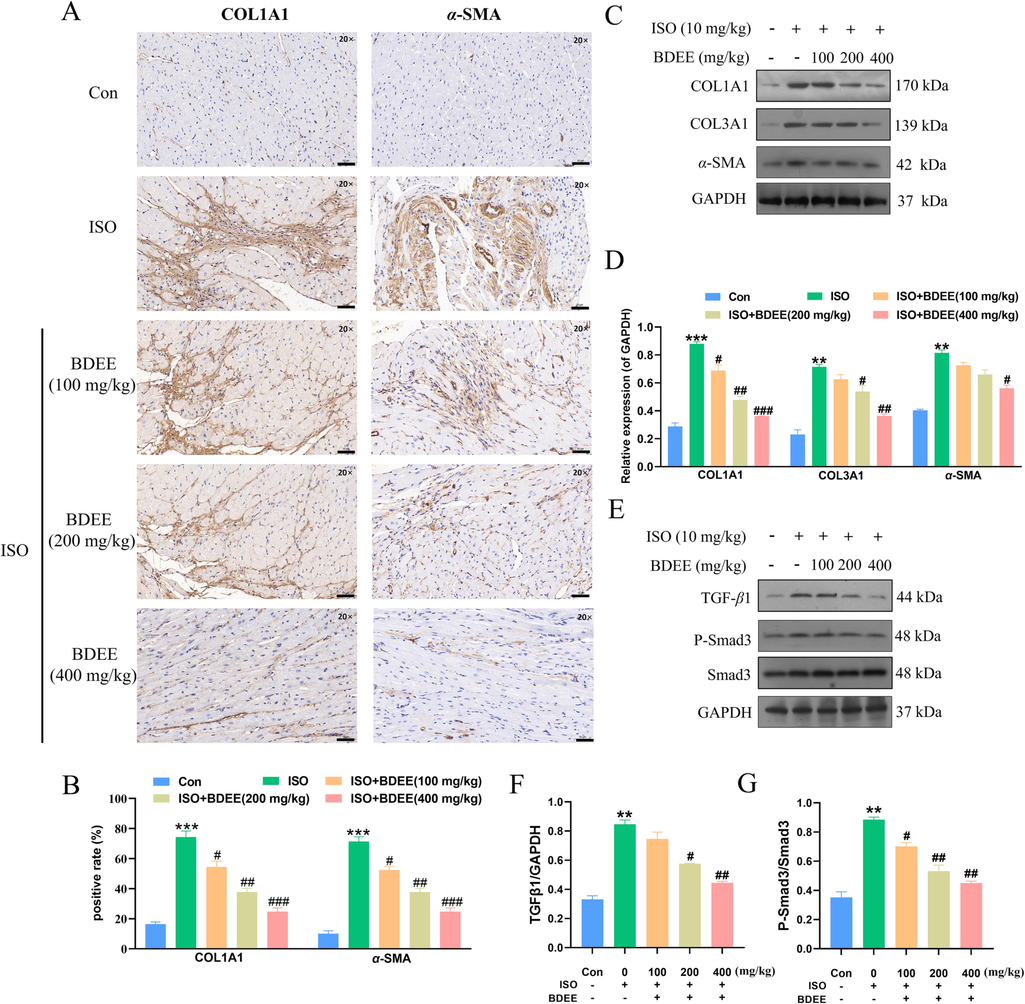
BDEE inhibits myocardial fibrosis induced by ISO. (A) Photomicrographs of histological sections of heart depicting expression of COL1A1 and α-SMA, magnification: 20 ×, scale bar = 50 μm. (B) Quantitative image analysis for immunohistochemical staining of COL1A1 and α-SMA, expressed as optical densities (OD). (C) Effects of BDEE on the expression of COL1A1, COL3A1, α-SMA in heart tissue were determined by western blotting. (D) Quantification of the COL1A1, COL3A1, α-SMA protein expression normalized to GAPDH. (E) Effects of BDEE on the expression of TGF-β1, P-Smad3 and Smad3 in each group. Quantification of the TGF-β1 (F) normalized to GAPDH, P-Smad3/Smad3 (G) protein expression. Results are represented as means ± SEM, n = 3. ***P < 0.001, **P < 0.01, compared with Con group; ### P < 0.001, ##P < 0.01, #P < 0.05, compared with ISO group.
3.5 BDEE can reduce the inflammation of ISO-induced myocardial injury mice
TLR4 is an important target protein in inflammatory pathways. Immunohistochemical results of TLR4 (Fig. 4A and B) showed that compared with the control group, TLR4 expression in ISO group was significantly increased, while after BDEE administration, TLR4 expression decreased in a dose-dependent manner. These results suggest that TLR4 plays a role in that BDEE improves the inflammatory response of ISO-induced myocardial injury mice. The role of TLR4 in reducing inflammation and other target proteins related to TLR4 were further verified by Western blotting. Western blotting results (Fig. 4C and D) showed that, compared with control group, TLR4, MyD88, IRAK-1, TRAF-6, NF-κB protein expression in ISO group increased. (Compared with the Con group, the ISO model group increased the protein expression levels of TLR4, MyD88, IRAK-1, TRAF-6, NF-κB by 1.64, 3.12, 0.43, 3.19 and 1.16 times, respectively) However, after BDEE administration, there were different degrees of decline, these results indicated that BDEE could reduce inflammatory response by regulating the expression of TLR4, MyD88, IRAK-1, TRAF-6 and NF-κB. It was further revealed that BDEE may reduce ISO-induced myocardial inflammation in mice and improve myocardial injury by regulating TLR4/MyD88/NF-κB pathway.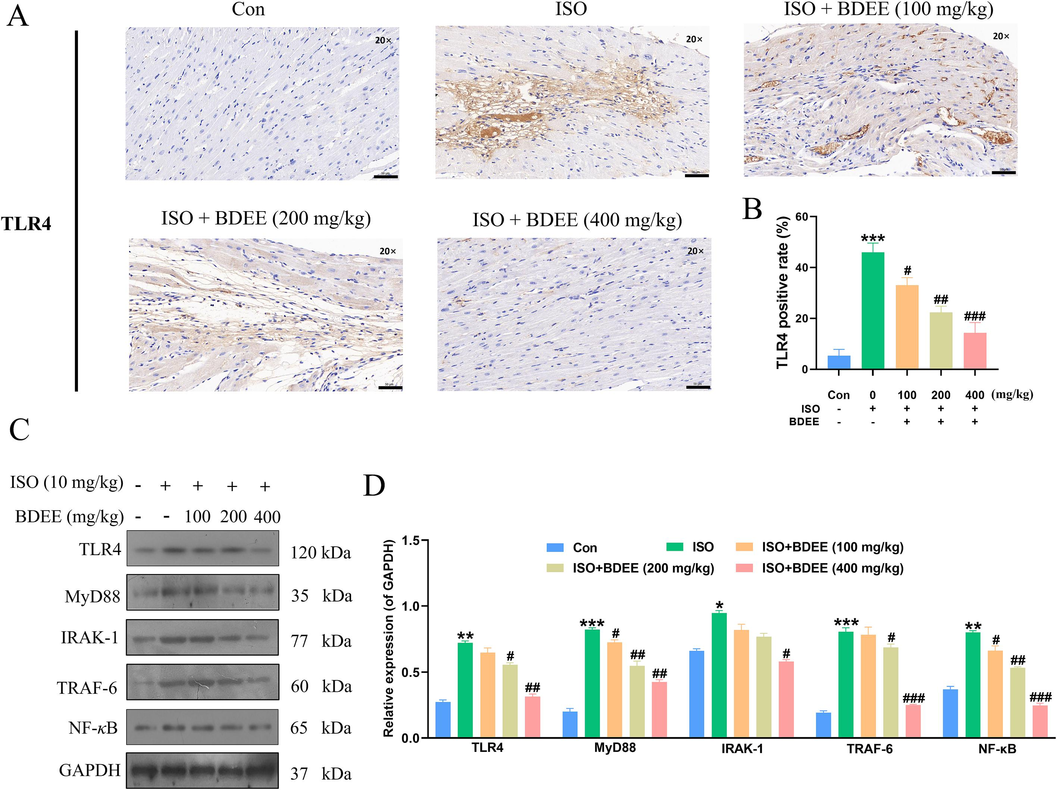
BDEE can reduce the inflammation of ISO-induced myocardial injury mice. (A) Photomicrographs of histological sections of heart depicting expression of TLR4, magnification: 20 ×, scale bar = 50 μm. (B) Quantitative image analysis for immunohistochemical staining of TLR4, expressed as optical densities (OD). (C) The effects of BDEE on the protein expression of TLR4, MyD88, IRAK-1, TRAF-6 and NF-κB in heart tissue were determined by western blotting. (D) Relative intensities of TLR4, MyD88, IRAK-1, TRAF-6 and NF-κB were calculated by normalization to the GAPDH in each group. Values are given as mean ± SEM, n = 3. ***P < 0.001, **P < 0.01, *P < 0.05, compared with Con group; ###P < 0.001, ##P < 0.01, #P < 0.05, compared with ISO group.
3.6 BDEE may achieve myocardial protection by inhibiting ISO-induced apoptosis
TUNEL staining results were shown in Fig. 5A and B. Compared with the control group, the apoptosis rate of myocardial cells in the model group was significantly increased. Compared with the model group, the apoptosis rate was decreased after BDEE administration. (Compared with the Con group, the ISO model group increased TUNEL-positive cells by 6.99 times, compared with the ISO model group, the ISO + 100, 200, 400 mg/kg BDEE group decreased by 2.28, 0.52, and 7.73 times, respectively). Western blotting results showed that (Fig. 5C–H), compared with the control group, Bcl-2 protein expression was decreased and Bax, Caspase-9 and Caspase-3 protein expression were significantly increased in the model group. Compared with the model group, the expression level of Bcl-2 increased in a dose-dependent manner, while the protein expression levels of Bax, Caspase-9 and Caspase-3 decreased in a dose-dependent manner. It was consistent with the literature and indicated that BDEE inhibited ISO-induced apoptosis by regulating Bcl-2, Bax, Caspase-9 and Caspase-3, thus improving myocardial injury (Bi et al., 2018, Nagoor Meeran et al., 2019).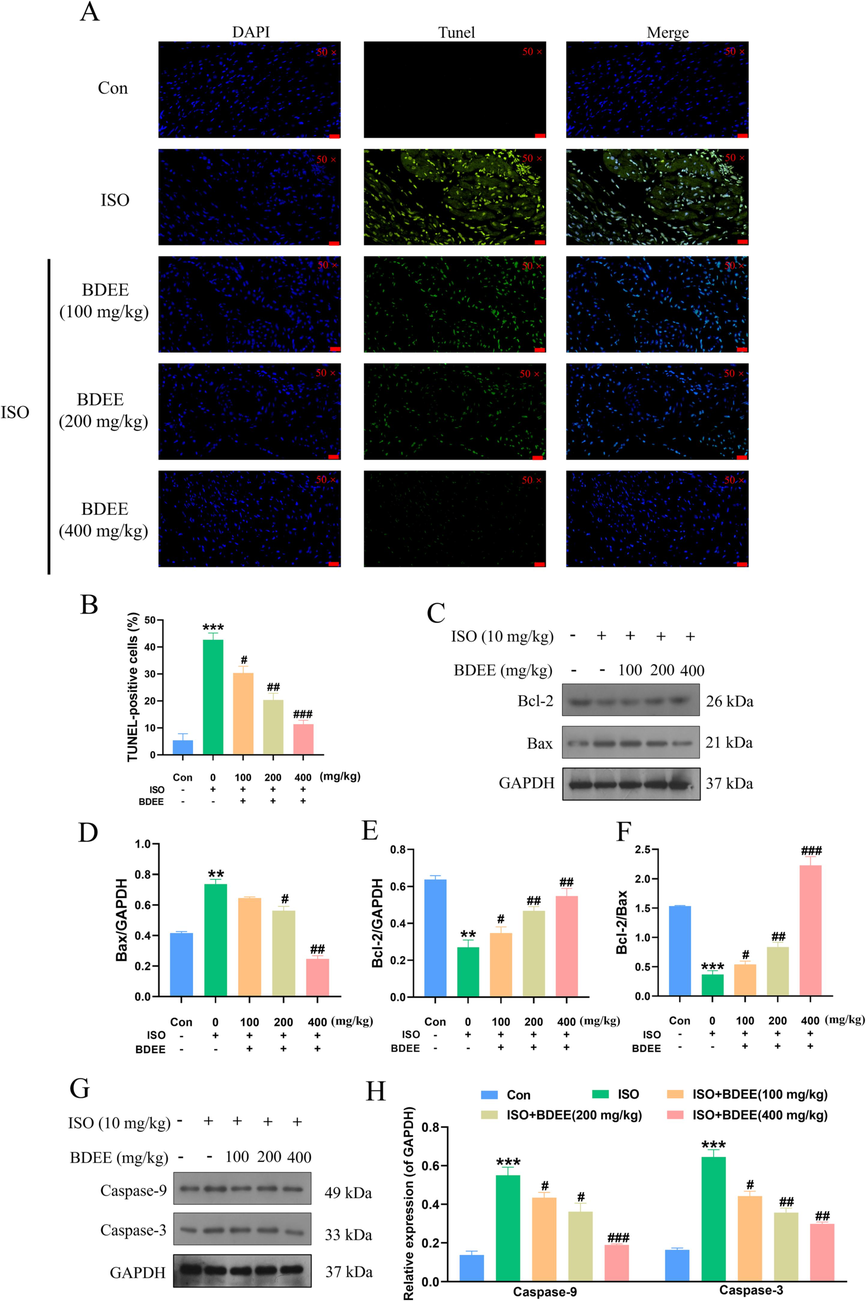
BDEE may achieve myocardial protection by inhibiting ISO-induced apoptosis. (A) Representative microscopic photographs of TUNEL-stained heart sections, magnification: 50 ×, scale bar = 20 μm. (B) The histogram showed the relative proportion of TUNEL-positive cells in the heart of mice. (C) Effects of BDEE on the expression of Bcl-2, Bax in each group. Quantification of the Bax (D), Bcl-2 (E) protein expression normalized to GAPDH. (F) Quantification of Bcl-2/Bax ratio in protein expression level. (G) Effects of BDEE on the expression of Caspase-9 and Caspase-3 in each group. (H) Quantification of the Caspase-9, Caspase-3 protein expression normalized to GAPDH. Results are represented as means ± SEM, n = 3. ***P < 0.001, **P < 0.01, compared with Con group; ###P < 0.001, ##P < 0.01, #P < 0.05, compared with ISO group.
4 Discussion
ISO can promote the proliferation of cardiac fibroblasts and collagen formation, which is mediated by Ang II (Gregori et al., 2013). After binding to its corresponding receptor, Ang II can not only promote the proliferation of cardiac fibroblasts, but also stimulate the secretion of TGF-β1 by cardiac fibroblasts, promoting collagen production and causing cardiac interstitial fibrosis. The TGF-β1/Smads signaling pathway plays an important regulatory role in the process of tissue fibrosis. At present, many experiments have confirmed that inhibition of TGF-β1/Smad3 signaling pathway can inhibit myocardial fibrosis, making TGF-β1/Smad3 become one of the main targets of anti-fibrosis treatment (Liu et al., 2019b). In this study, compared with the ISO model group, the expression of TGF-β1/Smad3 in the myocardium of the mice in each dose group was decreased, suggesting that the BDEE can regulate the TGF-β1/Smad3 pathway.
ISO can cause myocardial injury by inducing inflammatory response. In this study, TLR4, MyD88, and NF-κB protein levels were increased in myocardial injury model mice. TLRs are an important family of recognition receptors that can induce natural immune responses (Vallejo, 2011). TLR4 is a member of TLRs. As an important receptor of inflammatory response, it is an important member of the immune defense system and a bridge between immune response and chronic inflammation. TLR4 activates the MyD88 pathway and ultimately activates NF-κB, a key transcription factor that activates the inflammatory response (Lim and Staudt, 2013). And it was found that TLR4 activated NF-κB by MyD88, IRAKs and TRAF6 (Pham, 2006). NF-κB p65 dissociation occurs nuclear translocation, activation enters the nucleus. Anti-inflammation is one of the main effects of BDEE. After administration of BDEE in mice with myocardial injury, H&E staining showed that myocardial necrosis and inflammatory infiltration were significantly reduced, and Sirius red staining showed that myocardial collagen deposition was reduced. It is suggested that BDEE may protect myocardial cells, reduce myocardial necrosis and inflammatory response, reduce myocardial collagen deposition and play a protective role in myocardial injury by inhibiting myocardial inflammation. Combined with the results of this study and a large number of earlier studies, it can be concluded that BDEE may inhibit myocardial inflammation by inhibiting TLR4/MyD88/NF-κB signaling pathway.
In recent years, it has been reported that cardiomyocyte apoptosis plays an important role in the occurrence and development of cardiovascular diseases such as atherosclerosis and myocardial ischemia–reperfusion injury. (Li et al., 2020, Wang et al., 2016). Excessive cardiomyocyte apoptosis is an important factor causing cardiac dysfunction and structural changes, leading to myocardial injury. Therefore, inhibition of cardiomyocyte apoptosis can reduce myocardial injury. TUNEL staining is a classical method to assess apoptosis (Deng et al., 2021). In this study, TUNEL staining showed that BDEE could significantly improve ISO-induced cardiomyocyte apoptosis. Bcl-2 family proteins play an important role in the regulation of apoptosis. Bcl-2 can inhibit the occurrence of apoptosis, and Bax can promote the occurrence of apoptosis. The two cooperate to control the apoptosis of cells by controlling their content. Studies have found that Bcl-2 can regulate mitochondrial membrane permeability, triggering Caspase cascade, leading to cardiomyocyte apoptosis (Edlich, 2018). The precise regulation of cardiomyocyte apoptosis pathway is inextricably linked to the Caspase family. Caspase-9 belongs to the initial type of Caspases, and its main function is to initiate apoptosis and regulate the activity of effector Caspases. Caspase3 belongs to the effector Caspases, and its main function is the executor and terminator of apoptosis. Its activation can promote the formation of apoptotic bodies and cause cardiomyocyte apoptosis. Combined with the results of this study, it was concluded that BDEE could improve myocardial injury by inhibiting cardiomyocyte apoptosis and regulating Bcl-2, Bax, Caspase-9 and Caspase-3.
5 Conclusions
In conclusion, the study shows that BDEE protects the ISO-induced myocardial injury mice mainly by regulating TLR4/MyD88/NF-κB pathway to inhibit the formation of myocardial fibrosis, reduce inflammatory reaction, and lessen cell apoptosis. This research provides a theoretical basis for improving myocardial injury, and also affords a scientific basis for the development and utilization of Balanophora dioica.
CRediT authorship contribution statement
Ting Gao: Conceptualization, Data curation, Writing – original draft, Methodology, Project administration. Minjie Li: Formal analysis, Visualization, Project administration. Meng Zhang: Formal analysis, Visualization, Project administration. Yuxi Xiang: Methodology, Software. Zilong Huang: Methodology, Software. Weizhuo Tang: Funding acquisition, Writing – review & editing, Supervision, Resources, Investigation. Xiaoshu Zhang: Funding acquisition, Writing – review & editing, Supervision, Resources, Investigation.
Acknowledgments
This work was supported by National Natural Science Foundation of China (grant No. 81703389), Training Program for Excellent Young Innovators of Changsha (grant No. kq2305014), Education Fund Item of Liaoning Province (grant No. LJKZ0906), Natural Science Foundation of Science and Technology Department of Liaoning Province (grant No. 2021-MS-215), Youth Development Support Plan of Shenyang Pharmaceutical University (grant No. ZQN2021010) and Natural Science Fund of Hunan Province (grant No. 2022JJ30638).
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Bi, F., Xu, Y., Sun, Q., 2018. Catalpol pretreatment attenuates cardiac dysfunction following myocardial infarction in rats. Anatol. J. Cardiol. 19:296–302. 10.14744/AnatolJCardiol.2018.33230.
- Collaborators, G.B.D.C.o.D., 2018. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 392(10159), 1736-1788. 10.1016/S0140-6736(18)32203-7.
- XPA serves as an autophagy and apoptosis inducer by suppressing hepatocellular carcinoma in a PI3K/Akt/mTOR dependent manner. J. Gastrointest. Oncol.. 2021;12(4):1797-1810.
- [CrossRef] [Google Scholar]
- Editorial Board of Chinese Materia Medica of the State Administration of Traditional Chinese Medicine. Chinese Materia Medica [M]. Shanghai: Shanghai Science and Technology Press, 1999.
- BCL-2 proteins and apoptosis: recent insights and unknowns. Biochem. Biophys. Res. Commun.. 2018;500(1):26-34.
- [CrossRef] [Google Scholar]
- Plantago asiatica L. seeds extract protects against cardiomyocyte injury in isoproterenol- induced cardiac hypertrophy by inhibiting excessive autophagy and apoptosis in mice. Phytomedicine. 2021;91:153681
- [CrossRef] [Google Scholar]
- Thymoquinone dose-dependently attenuates myocardial injury induced by isoproterenol in rats via integrated modulations of oxidative stress, inflammation, apoptosis, autophagy, and fibrosis. Naunyn Schmiedebergs Arch. Pharmacol.. 2021;394(8):1787-1801.
- [CrossRef] [Google Scholar]
- Inadequate RAAS suppression is associated with excessive left ventricular mass and systo-diastolic dysfunction. Clin. Res. Cardiol.. 2013;102(10):725-733.
- [CrossRef] [Google Scholar]
- [6]-gingerol ameliorates ISO-induced myocardial fibrosis by reducing oxidative stress, inflammation, and apoptosis through inhibition of TLR4/MAPKs/NF-κB pathway. Mol. Nutr Food Res. 2020;64:e2000003.
- [Google Scholar]
- Unintended effects of statins in men and women in England and Wales: population based cohort study using the QResearch database. BMJ. 2010;340:c2197
- [CrossRef] [Google Scholar]
- Screening, determination and quantification of major antioxidants from Balanophora laxiflora flowers. Food Chem.. 2010;122(3):584-588.
- [CrossRef] [Google Scholar]
- Ginsenoside Rb1 attenuates cardiomyocyte apoptosis induced by myocardial ischemia reperfusion injury through mTOR signal pathway. Biomed. Pharmacother.. 2020;125:109913
- [CrossRef] [Google Scholar]
- Toll-like receptor signaling. Cold Spring Harb. Perspect. Biol.. 2013;5(1):a011247
- [CrossRef] [Google Scholar]
- Liu, M., Ai, J., Feng, J., Zheng, J., Tang, K., Shuai, Z., Yang, J., 2019. Effect of paeoniflorin on cardiac remodeling in chronic heart failure rats through the transforming growth factor β1/Smad signaling pathway. Cardiovasc. Diagn Ther. 9(3), 272-280. 10.21037/cdt.2019.06.01.
- Isorhamnetin inhibits liver fibrosis by reducing autophagy and inhibiting extracellular matrix formation via the TGF-β1/Smad3 and TGF-β1/p38 MAPK pathways. Mediators Inflamm.. 2019;2019:6175091.
- [CrossRef] [Google Scholar]
- Long, B., Brady, W.J., Koyfman, A., Gottlieb, M., Cardiovascular complications in COVID-19. (1532-8171 (Electronic)). 10.1016/j.ajem.2020.04.048.
- α-Bisabolol abrogates isoproterenol-induced myocardial infarction by inhibiting mitochondrial dysfunction and intrinsic pathway of apoptosis in rats. Mol. Cell. Biochem.. 2019;453(1–2):89-102.
- [CrossRef] [Google Scholar]
- Morphological and functional characteristics of models of experimental myocardial injury induced by isoproterenol. Gen. Physiol. Biophys.. 2012;31(2):141-151.
- [CrossRef] [Google Scholar]
- Separation of flavanone enantiomers and flavanone glucoside diastereomers from Balanophora involucrata Hook. f. by capillary electrophoresis and reversed-phase high-performance liquid chromatography on a C18 column. J. Chromatogr. A. 2008;1185(1):117-129.
- [CrossRef] [Google Scholar]
- Neutrophil serine proteases: specific regulators of inflammation. Nat. Rev. Immunol.. 2006;6(7):541-550.
- [CrossRef] [Google Scholar]
- Ruan, H.L., Li, J., Zhao, X.Y., Zhang, Y.H., Xiang, M., Wu, J.Z., 2003. Studies on Anti-inflammatory and Analgesic effects of Balanophora involucrate. J Archtcm. 1009 - 5276(2003) 06 - 0910 – 02.
- Metabolomics based mechanistic insights to vasorelaxant and cardioprotective effect of ethanolic extract of Citrullus lanatus (Thunb.) Matsum. & Nakai. seeds in isoproterenol induced myocardial infraction. Phytomedicine. 2022;100:154069
- [CrossRef] [Google Scholar]
- Forsythiaside B inhibits myocardial fibrosis via down regulating TGF-β1/Smad signaling pathway. Eur. J. Pharmacol.. 2021;908:174354
- [CrossRef] [Google Scholar]
- Xanthohumol attenuates isoprenaline-induced cardiac hypertrophy and fibrosis through regulating PTEN/AKT/mTOR pathway. Eur. J. Pharmacol.. 2021;891:173690
- [CrossRef] [Google Scholar]
- BACE inhibitory flavanones from Balanophora involucrata Hook. f. Fitoterapia. 2012;83(8):1386-1390.
- [CrossRef] [Google Scholar]
- Role of toll-like receptors in cardiovascular diseases. Clin. Sci. (Lond.). 2011;121(1):1-10.
- [CrossRef] [Google Scholar]
- Interpretation of report on Cardiovascular health and diseases in China 2022. Chin J. Cardiovasc. Med.. 2023;28(4):297-312.
- [CrossRef] [Google Scholar]
- Scutellarin protects cardiomyocyte ischemia-reperfusion injury by reducing apoptosis and oxidative stress. Life Sci.. 2016;157:200-207.
- [CrossRef] [Google Scholar]
- Phenolic acids from Balanophora involucrata and their bioactivities. Fitoterapia. 2017;121:129-135.
- [CrossRef] [Google Scholar]
- Lignan constituents from Balanophora involucrate and their anti-inflammatory activities. Chin. Tradit. Herb. Drug. 2019;50(08):1915-1920.
- [CrossRef] [Google Scholar]
- Xia, D.Y., 2023, Study on the mechanism of the therapeutic effect of Balanophora involucrata on acetic acid gastric ulcer in rats. [D]. Hubei Minzu University. DOI: 10.27764/d.cnki.ghbmz.2022.000021.
- Modified citrus pectin ameliorates myocardial fibrosis and inflammation via suppressing galectin-3 and TLR4/MyD88/NF-κB signaling pathway. Biomed. Pharmacother.. 2020;126:110071
- [CrossRef] [Google Scholar]
- 8-Gingerol ameliorates myocardial fibrosis by attenuating reactive oxygen species, apoptosis, and autophagy via the PI3K/Akt/mTOR signaling pathway. Front. Pharmacol.. 2021;12:711701
- [CrossRef] [Google Scholar]
- Phlorizin ameliorates myocardial fibrosis by inhibiting pyroptosis through restraining HK1-mediated NLRP3 inflammasome activation. Heliyon. 2023;9:e21217.
- [Google Scholar]
- Zhang, S.Y., Fu, K.C., Ni, Y., Zhao, Q., 2008. Medicinal plant resources for treating liver disease of Dai nationality in Menglian County. Chin. J. Ethn. Med. (02),36-37. 10.16041/j.cnki.cn15-1175.2008.02.040.
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2024.105662.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







