Translate this page into:
Synthesis and evaluation of an environmentally friendly phosphorus-free and nitrogen-free polymer as a scale and corrosion inhibitor
⁎Corresponding author. hualiyiwang@163.com (Xinhua Liu)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Polyepoxysuccinic acid (PESA) was modified by glycidyl (Gly) to obtain a phosphate-free and nitrogen-free polymer (Gly-PESA), and its scale and corrosion inhibition properties were studied.The effects of Gly-PESA concentration, calcium ion concentration and temperature to scale inhibition performance were studied by static scale inhibition method. The corrosion inhibition performance of Gly-PESA was studied by static weight-loss method. The morphology and structure of calcium scale and steel surface were characterized by spectroscopic and microscopic methods. The scale inhibition efficiency of Gly-PESA for CaCO3 and CaSO4 was 98.06% and 92.6%, respectively. Under the same experimental conditions, the scale inhibition effect of Gly-PESA was obviously better than that of PESA. Gly-PESA could effectively inhibit the growth of CaCO3 and CaSO4 crystals and cause their crystal lattice distortion or crystal dispersion. The corrosion inhibition efficiency of Gly-PESA for carbon steel was 78.17%. The adsorption of Gly-PESA on the surface of the steel test piece followed Langmuir adsorption isotherm, and formed a protective film, which increased the corrosion resistance of the steel test piece, thus effectively protecting the steel test piece. Therefore, Gly-PESA was a green scale and corrosion inhibitor, which solved the problem of environmental pollution.
Keywords
Green scale inhibitor
Green corrosion inhibitor
Modified polyepoxysuccinic acid
Interface protection

1 Introduction
With the accelerated development of industrial production and the rapid increase in industrial water consumption, industrial cooling water accounted for more than 60 %–70 % of industrial water. Industrial cooling water could cause corrosion, scaling and microbial slime problems on the surface of equipment and pipes, thereby shortening the service life of equipment and pipes (Quraishi et al., 2010, Htet and Zeng, 2022,Touir et al., 2009). Mineral ions in industrial cooling water systems, such as bicarbonate ions and calcium ions, might settle on the surface of heat transfer equipment (such as heat exchangers, evaporators and condensers) to form scale. In addition, corrosion was also a major problem for cooling water systems, as the consequences of corrosion could lead to loss of reliability, process contamination, reduced system performance, increased maintenance costs, or unplanned downtime (Htet and Zeng, 2022, Yee et al., 2019). The two main operational problems of cooling water systems were corrosion and scaling due to electrochemical redox reactions and the deposition of metal salts on metal surfaces. Therefore, in order to solve these problems in cooling water systems, various inhibitors have been used (Htet and Zeng, 2022, Zeng and Qin, 2012, Sharma and Kumar, 2021).
In the era of world environmental protection laws and regulations, the concept of green chemistry and green water treatment agents has been deeply rooted in people's hearts (Liu et al., 2014). Traditional and natural scale and corrosion inhibitors had problems such as high operating cost and large drug dosage. In addition, corrosion inhibitors and scale inhibitors containing phosphorus had poor scale inhibition effect on calcium phosphate, zinc scale and iron oxide precipitation or caused changes in water quality. At present, PESA and polyaspartic acid (PASP) were identified as scale and corrosion inhibitors for environmentally friendly cooling water treatment in many countries (Leng et al., 2020, Mazumder Jafar, 2020, Liu et al., 2023). Among them, PESA was a new type of green water treatment agent with no phosphorus, no nitrogen, environmental protection and safety. It would not cause the problem of water environment eutrophication in long-term use (Liu et al., 2014, Leng et al., 2020, Yan et al., 2020, Shi et al 2012, Zhang et al., 2010).
PESA was originally developed as a scale inhibitor. It had strong inhibition ability to calcium ion, magnesium ion and iron ion, and was suitable for water treatment with high hardness and high alkalinity. In addition, PESA also had a certain effect on pipeline scaling, corrosion and other problems, and had biodegradability. Traditional corrosion inhibitors and scale inhibitors didn’t have these advantages. However, PESA had some obvious defects, such as single functional group, which was limited in its application scope. Therefore, many researchers have carried out experimental studies on the modification of PESA to expand its application scope (Leng et al., 2020, Kadhim et al., 2021,Liu et al., 2020, Lai et al., 2020, Wei et al., 2022, Al-Itawi et al., 2019, Zhao et al., 2022, Zhang et al., 2021).
Gly contained hydroxyl and ester groups that chelate metal ions (such as calcium ions). This work aimed to introduce Gly into the molecular structure of PESA to obtain Gly-PESA. Because the existence of Gly can improve the corrosion and scale inhibition performance of PESA in a certain extent. The scale inhibition effects of Gly-PESA were studied by static experiments. The corrosion inhibition effect of Gly-PESA to Q235 carbon steel in cooling water was studied by static weight loss method. Nuclear magnetic hydrogen spectroscopy (1H NMR), fourier transform Infrared spectroscopy (FTIR), thermogravimetric analysis (TGA), scanning electron microscopy (SEM), atomic force microscopy (AFM), X-ray photoelectron spectroscopy (XPS) and contact angle (CA) were used to characterize structures of Gly-PESA, the calcium scale or the surface of the steel test piece, which proved the scale inhibition and corrosion inhibition mechanism of Gly-PESA.
2 Experiment
2.1 Equipment and reagent
VERTEX7O Fourier Infrared Spectrometer (Bruker Technology Co., LTD., Germany);SIGMA300 Field Emission Scanning Electron Microscopy (Carl Zeiss AG, Germany);TGA-4000 Thermogravimetric Analyzer (PerkinElmer, USA);Zennium Pro Electrochemical Workstation (ZAHNER-elektrik GmbH & Co.KG, Germany);PulsarTM NMR Spectrometer (Shanghai Hao Lang Scientific Instrument Co., LTD., UK);JC2000 DM Contact Angle Measuring Instrument (Shanghai Zhongchen Digital Technology Equipment Co., LTD., China);AXIS SUPRA X-ray photoelectron Spectrometer (Shimadzu, Japan) and MFP-3D Origin Atomic Force Microscope (Oxford, UK).
Gly (Mass percentage 97 %) (Shanghai Yien Chemical Technology Co., LTD., China); Maleic anhydride, sodium hydroxide, anhydrous calcium chloride, anhydrous sodium sulfate, methanol, anhydrous ethanol, sodium bicarbonate, disodium EDTA (Tianjin Yongda Chemical Reagent Co., LTD. China), all of the above reagents were reagent grade and could be used without further purification; Corrosion pieces of Q235 carbon steel (Gaoyou Qinyou Instrument Chemical Co., LTD., area 20 cm2, China).
2.2 Synthesis of PESA and Gly-PESA
The synthesis of PESA and Gly-PESA was carried out in reference (Leng et al., 2020. Wei et al., 2022. Zhang et al., 2021. Zhang et al., 2017.).
Epoxysuccinic acid (ESA) synthesis: First, 15 mL of distilled water was added to 9.8 g maleic anhydride, and 40 % NaOH solution was slowly added while stirring at 40 °C, and PH was controlled between 5–6. Secondly, added 0.6 g of sodium tungstate and sodium molybdate with a mass ratio of 1:1, droped 10 mL hydrogen peroxide at a slow rate, temperature rised to 65 °C, reaction for 2 h (the above is the cyclization reaction). Finally, the solution was adjusted to acid, then washed with methanol, extracted, filtered, obtained white solid (ESA) and dried for use at 40 °C.
PESA synthesis: Cyclization reaction solution was adjusted to about pH = 11, 0.4 g of initiator (calcium hydroxide) was added in four times, and the temperature was raised to 80 °C for 2 h. After the reaction, the solution was adjusted to acidity, washed with methanol for 5 times to obtain a viscous substance (PESA), and dried at 40 °C for use.
Gly-PESA synthesis: ESA and Gly were put into a three-mouth flask with a mass ratio of 1:0.33, and dissolved with distilled water, adjusting pH = 8. The initiator was added in four times (the amount was 8 % of the amount of reactant). After reaction lasted for 4 h at 80 °C, the solution was adjusted to neutral, and washed for 5 times with methanol. The obtaining viscous substance (Gly-PESA) was dried at 40 °C. The yield of Gly-PESA is about 60 %. The reaction process was shown in Fig. 1: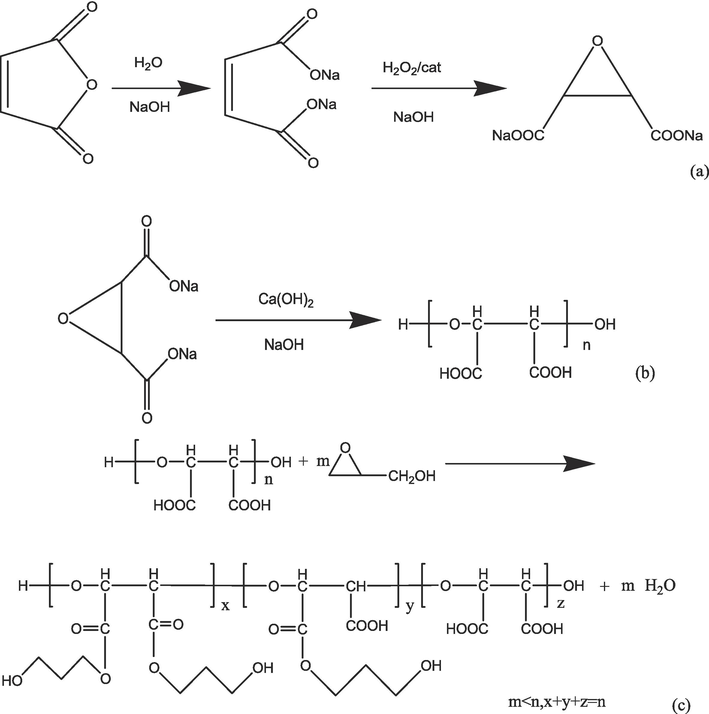
Synthesis of ESA(a), PESA(b) and Gly-PESA(c).
2.3 PESA and Gly-PESA structure analysis and sample characterization
The structures of Gly-PESA were analyzed by FTIR and 1H NMR. The thermal stability of Gly-PESA was proved by TGA. SEM, XPS and FTIR were used to characterize CaCO3 and CaSO4. The surface morphology, film composition and wettability of the steel test pieces were analyzed by SEM, XPS, FTIR and CA.
2.4 Measurement of scale inhibition performance
The scale inhibition effects of PESA and Gly-PESA on CaCO3 and CaSO4 were compared and evaluated referring to the Chinese National standard (GB/T16632-2019) and literature (Liu et al., 2023). The scale inhibition performance test of CaCO3 in the work: ρ(Ca2+) = 200 mg/L and ρ(HCO3–) = 400 mg/L; The scale inhibition performance test of CaSO4: ρ(Ca2+) = 2000 mg/L, ρ(SO42−) = 4000 mg/L.
2.5 Measurement of corrosion inhibition performance
The corrosion test water was tap water [Tangshan,Heiei]: pH=7.8, total hardness was 190 mg/L, conductivity was 624 μs/cm, and the concentrations of Ca2+, Mg2+, Cl− and SO42− were 54.8 mg/L, 27.3 mg/L, 33 mg/L and 60 mg/L respectively. The prepared steel test pieces were immersed in a beaker containing 500 mL simulated cooling water and different doses of Gly-PESA at 30 °C. After soaking for 24 h, removed the steel test pieces, washed (first with 5 % HCl and 3 mg/L hexamethylenetetramine, followed by deionized water), dried, and weighed. Refer to test method of literature (Liu et al., 2023), the corrosion rate(X) and the corrosion inhibition efficiency (ƞ) were calculated. The formulas were (1) and (2).
3 Results and discussion
3.1 Structural characterization of Gly-PESA
The FTIR, 1H NMR and TGA of Gly-PESA were showed in Fig. 2. In the infrared spectrum of PESA in Fig. 2(a), 3477 cm−1/3381 cm−1 and 1627 cm−1/1398 cm−1 were O−H and C⚌O stretching vibration absorption peaks in –COOH, respectively. 1315 cm−1 was C−H deformation vibration, and 1066 cm−1 was C−O−C stretching vibration absorption peak. In the infrared spectrum of Gly-PESA, the hydroxyl peak becomed wider at 3435 cm−1, the absorption peak of − CH2 − was increased at 2932 cm−1, and the peak of C⚌O at 1627 cm−1 and the peak of C−H at 1315 cm−1 became larger. Therefore, FTIR analysis shows that Gly-PESA was successfully obtained.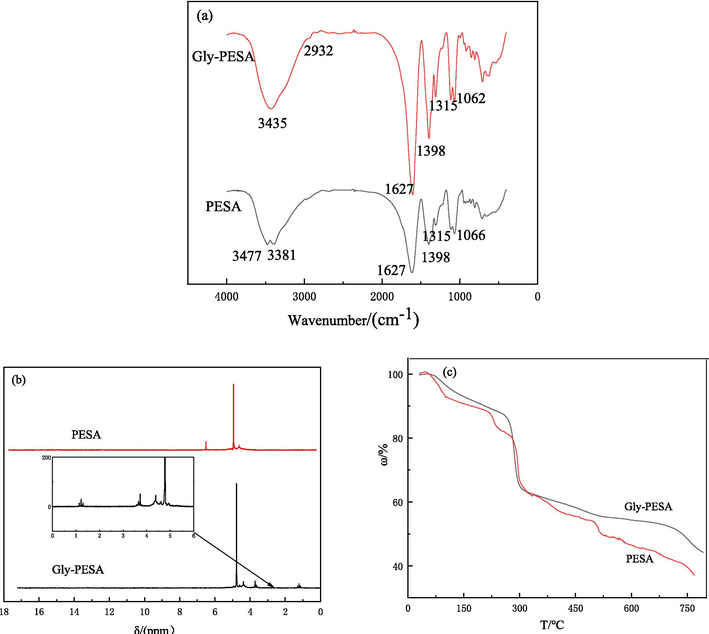
The FTIR(a),1H NMR(b) and TGA(c) of PESA and Gly-PESA.
In 1H NMR of Gly-PESA in Fig. 2(b), besides containing the characteristic peaks in PESA, hydrogen peaks in methylene (−CH2−) and hydroxyl (−CH2OH) in Gly were also found at the chemical shift δ = 3.5 ppm and δ = 1.5 ppm, respectively, which proved the successful synthesis of Gly-PESA.
Fig. 2(c) was the TGA diagram of Gly-PESA and PESA. It could be seen from the TGA curve in Fig. 2(c) that the stability of Gly-PESA and PESA is basically the same. According to the TGA figure of Gly-PESA, the weight loss was smooth at 30 °C–250 °C, which may be caused by the volatilizing of water or other volatile substances, and the weight loss rate was about 10 %–12 %.The weight loss rate was about 28 % at 250 °C–300 °C, which may be due to the fracture of the branch chain of Gly-PESA. Subsequently, the weight loss was gentle between 300 °C and 500 °C, and further gentler between 500 °C and 700 °C, which may be due to the further increase of temperature leading to the break of the main chain of a few Gly-PESA molecules. At 700 °C-800 °C, weight loss was rapid, probably because the temperature was too high to cause many Gly-PESA molecules to decompose faster (Liu et al., 2023). In summary, Gly-PESA had excellent thermal stability in the range of 0 °C-250 °C, and could meet the needs of industrial production.
3.2 Gly-PESA scale inhibition performance analysis
3.2.1 Influence of the dosage of Gly-PESA on the scale inhibition performance to CaCO3 and CaSO4
By changing the dosage of Gly-PESA, the scale inhibition effect to CaCO3 was shown in Fig. 3(a). When the dosage of Gly-PESA was 2 mg/L, the scale inhibition efficiency was 45.7 %. Moreover, with the increase of the dosage of Gly-PESA (2 mg/L–12 mg/L), the scale inhibition efficiency was continuously improved, and the scale inhibition performance of Gly-PESA was obviously better than that of PESA. When the dosage of Gly-PESA was 8 mg/L and 10 mg/L, respectively, the scale inhibition efficiency reached 90.3 % and 98.0 %, respectively. In contrast, the scale inhibition efficiency of Gly-PESA was increased by 10.8 % and 7.7 % over that of PESA, respectively. This was due to the existence of polar groups in Gly-PESA, which enhanced its electrostatic attraction and chelation with calcium ions in water, thus forming a stable calcium ion complex in water, reducing the formation of sediments, and further improving the scale inhibition efficiency (Yan et al., 2020; Zhang et al., 2021; Wang et al., 2017).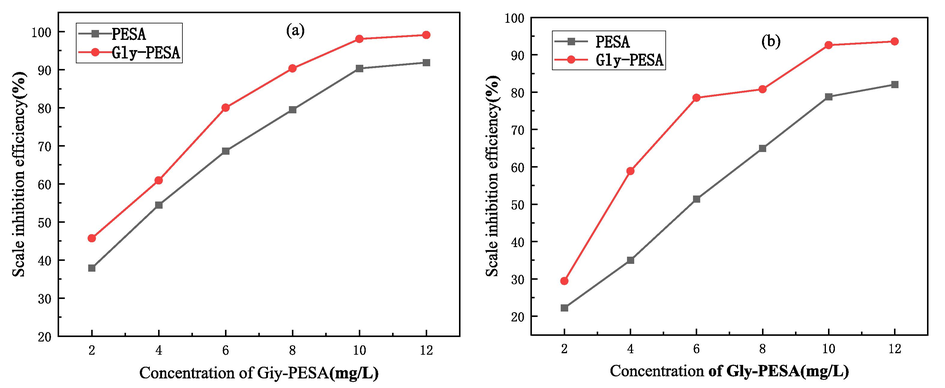
Scale inhibition performance of with different dosage of PESA and Gly-PESA to CaCO3(a) and CaSO4(b).
By changing the dosage of Gly-PESA, the scale inhibition efficiency to CaSO4 was shown in Fig. 3(b). It can be seen from Fig. 3(b) that the scale inhibition efficiency of Gly-PESA increased with increasing dosage. When the dosage of Gly-PESA was 10 mg/L, the scale inhibition efficiency of Gly-PESA was 92.6 %, and the scale inhibition efficiency of Gly-PESA was 13.8 % higher than that of PESA. When the dosage exceeded 10 mg/L, the scale inhibition efficiency of Gly-PESA increased little. In conclusion, the inhibition effect of Gly-PESA to CaSO4 was better than that of PESA.
3.2.2 Effect of temperature on the scale inhibition of Gly-PESA to CaCO3 and CaSO4
The test temperature would affect the formation rate and solubility of CaCO3 and CaSO4 in the system. Therefore, in this work, the concentration of PESA and Gly-PESA was 10 mg/L respectively to study the influence of temperature on the scale inhibition ability of Gly-PESA, and the test results were shown in Fig. 4. Fig. 4 showed that the scale inhibition ability of PESA and Gly-PESA on CaCO3 [Fig. 4(a)] and CaSO4 [Fig. 4(b)] both decreased with the increase of test temperature, but the decrease of Gly-PESA was very small. Moreover, the scale inhibition efficiency of Gly-PESA was always greater than that of PESA for both CaCO3 and CaSO4. This may be because PESA and Gly-PESA adsorb small crystals of calcium scale to release heat, so the increase of system temperature would affect the adsorption amount of PESA and Gly-PESA on small crystals (Liu et al., 2018). In addition, with the increase of system temperature, the velocity of the ion in the system also accelerated, making it easier for forming scale crystals. However, when the temperature was 80 °C, the scale inhibition efficiency of Gly-PESA on CaCO3 and CaSO4 could reach 88.3 % and 85.7 %, respectively. This indicated that Gly-PESA could maintain a good scale inhibition effect at high system temperature.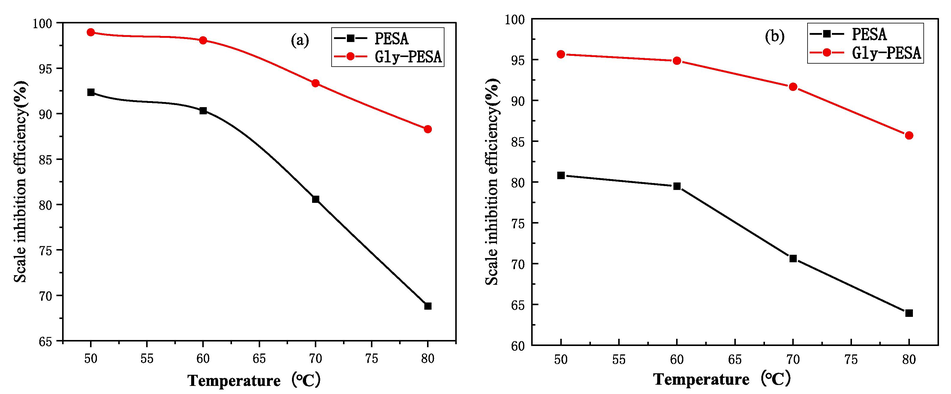
Effects of PESA(Dosage of 10 mg/L) and Gly-PESA(Dosage of 10 mg/L) on the scale inhibition properties of (a) CaCO3 and (b) CaSO4 at different temperatures.
3.2.3 Effect of Ca2+ concentration on the resistance of Gly-PESA to CaCO3 and CaSO4
With the extension of the use of industrial circulating cooling water, the Ca2+ concentration in water will increase. Based on national standard (GB/T16632-2019) in China and the literature (Htet and Zeng, 2022; Zhang et al., 2017; Liu et al., 2023), the concentration range of Ca2+ was selected 200 mg/L-600 mg/L(for CaCO3) and 2000 mg/L-6000 mg/L(for CaSO4) in this work. Therefore, this work studied the effects of Gly-PESA (10 mg/L) on the scale inhibition properties to CaCO3 and CaSO4 at different Ca2+ concentrations, as shown in Fig. 5. As couuld be seen from the curve in Fig. 5, with the increase of Ca2+ concentration, the scale inhibition efficiency of Gly-PESA on CaCO3 [Fig. 5(a)] and CaSO4 [Fig. 5(b)] gradually decreased, but the scale inhibition capacity of Gly-PESA was always stronger than that of PESA. According to the data of CaCO3 in Fig. 5(a), when the Ca2+ concentration reached 500 mg/L, the scale inhibition efficiency of Gly-PESA could still reach 83.2 %, while the PESA could only reach 59.6 %. According to the data of CaSO4 in Fig. 5(b), when the Ca2+ concentration reached 5000 mg/L, the scale inhibition efficiency of Gly-PESA could still reach 82.2 %, while the PESA could only reach 43.8 %. Therefore, it could be concluded that the scale inhibition capacity of PESA and Gly-PESA on CaCO3 and CaSO4 were closely related to the concentration of Ca2+ in the system, but the tolerance of Gly-PESA to calcium ions was always better than that of PESA.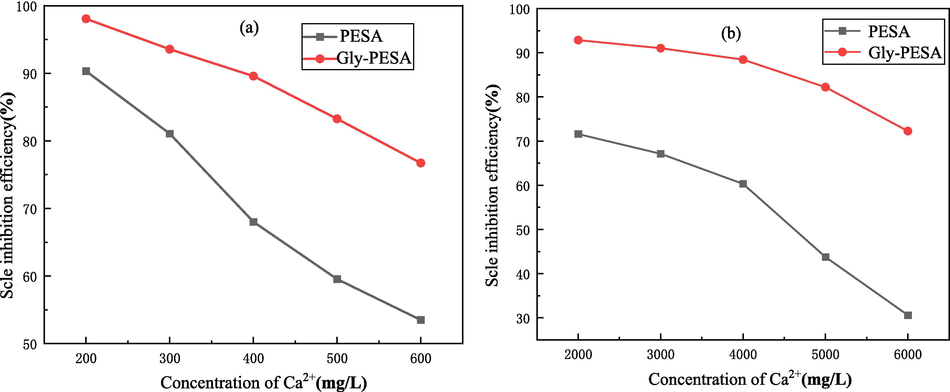
Effects of PESA(Dosage of 10 mg/L) and Gly-PESA(Dosage of 10 mg/L) on the scale inhibition properties of (a) CaCO3 and (b) CaSO4 at different Ca2+ concentrations.
3.3 Structural characterization of calcium scale
In order to further demonstrate the scale inhibition performance of Gly-PESA, SEM, FTIR and XPS were used to characterize the changes of crystal structure of CaCO3 and CaSO4.
3.3.1 SEM characterization of CaCO3 and CaSO4
CaCO3 crystals were prepared in blank system, systems with PESA and Gly-PESA, respectively. The SEM of CaCO3 crystals was shown in Fig. 6. As can be seen from Fig. 6(a) and Fig. 6(b), CaCO3 in the blank group was mainly composed of hexahedral calcite, with a smooth surface and hard texture. Although the crystal pattern of calcium scale added with PESA and Gly-PESA both changed greatly, the crystal distortion of calcium scale with Gly-PESA was more obvious, the crystal growth rule was destroyed, the particles became fine, and showed a good dispersion state, which indicated that Gly-PESA had a stronger interference on the formation of CaCO3 (Liu et al., 2023).![SEM of CaCO3 crystals. [Blank system: (a)/(b); System with PESA(10 mg/L): (c)/(d); System with Gly-PESA(10 mg/L): (e)/(f)].](/content/184/2024/17/12/img/10.1016_j.arabjc.2024.106033-fig6.png)
SEM of CaCO3 crystals. [Blank system: (a)/(b); System with PESA(10 mg/L): (c)/(d); System with Gly-PESA(10 mg/L): (e)/(f)].
The SEM of CaSO4 in the blank system and the scale inhibition system with PESA and Gly-PESA was shown in Fig. 7. As shown in Fig. 7(a) and Fig. 7(b), the crystals in the blank group were regular columnar structures with smooth surfaces. When PESA was added, the crystals in Fig. 7(c) and Fig. 7(d) became small, irregular shapes. When Gly-PESA was added, the crystal in Fig. 7(e) and Fig. 7(f) had no fixed shape, and the crystal structure was more loose. More specifically, CaSO4 crystals were completely broken into a small and unstable powder state.![SEM of CaSO4 crystals. [Blank system: (a)/(b); System with PESA(10 mg/L): (c)/(d); System with Gly-PESA(10 mg/L):(e)/(f)].](/content/184/2024/17/12/img/10.1016_j.arabjc.2024.106033-fig7.png)
SEM of CaSO4 crystals. [Blank system: (a)/(b); System with PESA(10 mg/L): (c)/(d); System with Gly-PESA(10 mg/L):(e)/(f)].
After the addition of scale inhibitor, SEM shows that the crystal structure of CaCO3 and CaSO4 changes. Probably mainly because (i) the complex of scale inhibitor molecules and Ca2+ ions in water formed a stable water-soluble chelate, thereby reducing the concentration of free calcium ions in water. (ii) the scale inhibitor molecules were adsorbed on the active growth point of microcrystals, which led to distortion of calcium scale crystals and increased of internal stress, preventing crystal aggregation and deposition (Liu et al., 2023; Htet T. T. et al., 2022).
3.3.2 FTIR result of CaCO3 and CaSO4
FTIR analysis was performed in this work in order to further confirm the crystal structures of CaCO3 and CaSO4.
The crystal types of CaCO3 were different when different scale inhibitors are added to the scale inhibition system. The FTIR spectrum of CaCO3 crystals were shown in Fig. 8(a), where the adsorption peaks at 711 cm−1 and 873 cm−1 represented the stable calcite and unstable aragonite crystal states of CaCO3, respectively (Meng et al., 2007, Naka, 2003). It was obvious from Fig. 8(a) that CaCO3 in the blank group was mainly calcite (only a small amount of aragonite), while the CaCO3 added with PESA and Gly-PESA was mainly aragonite (only a small amount of calcite). This was because the scale inhibitor molecules interfered with the process which aragonite formed calcite (Meng et al., 2007). In addition, it could be further seen from Fig. 8(a) that the peak of aragonite with Gly-PESA was significantly larger than that of adding PESA, indicating that Gly-PESA had a better scale inhibition effect. This phenomenon could be explained that the adsorption capacity of Gly-PESA on the surface of CaCO3 was obviously stronger than that of PESA, which showed strong lattice distortion. Fig. 8(b) showed that the peaks of CaSO4 scale obtained in different systems were consistent, indicating that the crystal type of CaSO4 did not change, but the strength of the peaks changed, and the peaks with Gly-PESA were smaller than those with PESA, indicating that the crystallinity of CaSO4 in the system with Gly-PESA was worse. This showed that Gly-PESA had better scale inhibition effect on CaSO4 (Zhang et al., 2017, Nan et al., 2022).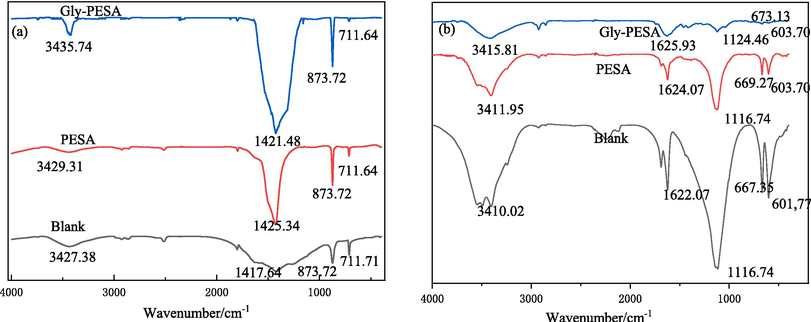
FTIR of CaCO3 (a) and CaSO4 (b) in different systems without or with PESA(10 mg/L) or Gly-PESA(10 mg/L).
3.3.3 XPS characterization of CaCO3 and CaSO4 scale
In order to further explore the adsorption properties of Gly-PESA, we performed XPS analysis of O1s and Ca2p of CaCO3 and CaSO4 formed in different systems.
Fig. 9 showed the O1s XPS spectra of CaCO3 and CaSO4. In addition to the main O peaks of CaCO3 and CaSO4 at 529 eV (Fig. 9(a), (d)) in blank system, peaks of C−O(527 eV) and C⚌O(529.91 eV, 530.56 eV) appeared in Fig. 9(b)/(c)/(e)/(f) after the addition of PESA and Gly-PESA. This again proved that PESA and Gly-PESA adsorbed active sites on the surface of CaCO3 and CaSO4 and interfered with their growth (Zhang et al., 2021). Fig. 10 showed the Ca2p XPS spectra of CaCO3 and CaSO4 scale. In the blank system, the Ca2p spectra of CaCO3 had significant characteristic peaks at 344.38 eV and 347.99 eV respectively. In the system with PESA, the peak displacement of Ca2p spectra was 343.82 eV and 347.47 eV, which might be the change of Ca2+ chemical environment causing the variation of scale crystal pattern. In the system with Gly-PESA, the peak displacement of Ca2p spectra was 345.18 eV and 348.72 eV, which further proved that Gly-PESA changed the crystal structure of CaCO3 (Zhang et al., 2023). The addition of PESA caused the Ca2p peak of CaSO4 to move by 0.18 eV relative to the blank Ca2p peak, while the addition of Gly-PESA caused the Ca2p peak to move by 0.20 eV and 0.17 eV relative to the blank Ca2p peak, respectively, as shown in Fig. 10(e) and Fig. 10(f). These results showed that the addition of Gly-PESA could cause changes of the chemical environment around Ca2+, and the effective groups of Gly-PESA could chelate with Ca2+, increase the electron density around Ca2+, and make the calcium peak migrate (Zhang et al., 2023). The above SEM, FTIR and XPS data results showed that Gly-PESA could be adsorbed on the surface of CaCO3 and CaSO4, affecting the surface morphology and transformation path of crystals, thus effectively inhibiting their formation and deposition in solution.![O1s XPS of CaCO3 (a-c)and CaSO4(d-f). [(a)/(d) Blank system, (b)/(e) System with PESA(10 mg/L), (c)/(f) System with Gly-PESA(10 mg/L)].](/content/184/2024/17/12/img/10.1016_j.arabjc.2024.106033-fig9.png)
O1s XPS of CaCO3 (a-c)and CaSO4(d-f). [(a)/(d) Blank system, (b)/(e) System with PESA(10 mg/L), (c)/(f) System with Gly-PESA(10 mg/L)].
![Ca2p XPS of CaCO3(a-d) and CaSO4(d-f). [(a)/(d) Blank system, (b)/(e) System with PESA(10 mg/L), (c)/(f) System with Gly-PESA(10 mg/L)].](/content/184/2024/17/12/img/10.1016_j.arabjc.2024.106033-fig10.png)
Ca2p XPS of CaCO3(a-d) and CaSO4(d-f). [(a)/(d) Blank system, (b)/(e) System with PESA(10 mg/L), (c)/(f) System with Gly-PESA(10 mg/L)].
3.4 Study on corrosion inhibition performance of Gly-PESA
In this section, weight-loss method, SEM, FTIR, CA and XPS were used to evaluate the corrosion inhibition performance of Gly-PESA.
3.4.1 Weight-loss method
In this section, the corrosion inhibition efficiency of Gly-PESA and PESA with different dosage was measured by weight loss method. The experimental results were shown in Table 1. It can be seen that with the increase of the dosage of Gly-PESA and PESA, the corrosion rate decreased and the corrosion inhibition efficiency increased. When the concentration of Gly-PESA and PESA was 400 mg/L, ƞmax reached 78.17 % and 67.05 %, respectively. This was because the steel surface area covered by Gly-PESA with branched chains was larger, and the chemisorbed effect was stronger.
Gly-PESA
PESA
CInhi
X (g/cm2.h)
ƞ (%)
X (g/cm2.h)
ƞ (%)
0
2.96 × 10−5
/
2.96 × 10−5
/
50
1.74 × 10−5
41.31
1.88 × 10−5
36.56
100
1.39 × 10−5
53.03
1.54 × 10−5
48.01
200
1.06 × 10−5
64.18
1.33 × 10−5
55.12
300
8.40 × 10−6
71.63
1.15 × 10−5
61.14
400
6.46 × 10−6
78.17
9.75 × 10−6
67.05
Firstly, from the point of view of scale and corrosion inhibition: the scale inhibition performance of Gly-PESA synthesized in this work was better than that of arginine-modified PESA[Arg-PESA] in the literature (Zhang et al., 2021), especially in terms of being affected by different temperature and calcium ion concentration. Second, the corrosion inhibition efficiency of Gly-PESA for Q235 steel could reach 78.17 %. the corrosion inhibition performance of Gly-PESA was basically equivalent to that of Arg-PESA (Zhang et al., 2021). The corrosion inhibition efficiency also reached a medium level.Therefore, the Gly-PESA synthesized in this work had the dual function of scale inhibition and corrosion inhibition. In the following research work, in order to further enhance the corrosion inhibition performance of GLY-PESA, this research group will further carry out the compounding work of GLY-PESA.
3.4.2 Surface analysis of the steel test pieces
The surface analysis technique was used to describe the surface state of Q235 steel and to prove the adsorption performance of Gly-PESA. The surface analysis methods we used were SEM, FTIR, AFM, XPS and CA.
SEM and FTIR
Fig. 11(a–c) showed the SEM images of the surface of the steel test pieces before corrosion, in the blank system and in the Gly-PESA system added 400 mg/L. The surface of the steel test piece (before corrosion) had obvious grinding marks and was smooth; The surface of the steel test piece (soaked in the blank solution) was covered with corrosion products. After the addition of Gly-PESA, the damage of the test piece was significantly reduced, the surface became smooth, and the corrosion products were less, which could be inferred that Gly-PESA played a protective role on the surface of the test piece.![SEM (a-c) and FTIR (d) on Q235 steel surface [(a) before corrosion; (b) blank system (c) System with Gly-PESA(400 mg/L); (d) System with Gly-PESA(400 mg/L)].](/content/184/2024/17/12/img/10.1016_j.arabjc.2024.106033-fig11.png)
SEM (a-c) and FTIR (d) on Q235 steel surface [(a) before corrosion; (b) blank system (c) System with Gly-PESA(400 mg/L); (d) System with Gly-PESA(400 mg/L)].
The FTIR spectra of Gly-PESA and the surface of the test piece (soaked in Gly-PESA solution) were shown in Fig. 11(d). As shown in Fig. 11(d), there were O–H stretching vibration absorption peaks in carboxyl group (3435 cm−1), −CH2 − functional group absorption peaks (2932 cm−1, 2859 cm−1), C⚌O stretching and deformation vibration absorption peaks in carboxyl group (1627 cm−1, 1388 cm−1) and C−O−C tensile vibration absorption peak (1051 cm−1) in the FTIR spectrum of the surface of the test piece. Compared with the FTIR diagram of Gly-PESA, it could be inferred that the functional groups of Gly-PESA acted on the metal surface to prevent corrosion reactions due to its adsorption.
-
2)
AFM
AFM was used to study the surface morphology of the test pieces before corrosion, in blank solution and in solution with Gly-PESA, as shown in Fig. 12. It could be clearly seen from Fig. 12 that the steel surface (before corrosion) was the smoothest, and its average surface roughness was 1.88 nm, as shown in Fig. 12(a, b). In the blank system, the height of the steel surface fluctuated greatly, forming the shape of mountains and hills, whose average surface roughness was 12.79 nm, as shown in Fig. 12(c, d). The surface height fluctuation of the steel protected by Gly-PESA was gentler than that of the blank system, and the damage degree was lower, and the average surface roughness was 5.11 nm, as shown in Fig. 12(e, f). In Fig. 12(g), the fluctuation amplitude of the corresponding height profile of the steel surface: steel sheet before corrosion < steel surface in the system with Gly-PESA<steel surface in blank system. The height fluctuation amplitude of the steel surface in the system with Gly-PESA was significantly lower than that of the blank system, indicating that the corrosion rate of the steel was significantly reduced. In summary, the micromorphology of AFM confirmed the results of gravimetric analysis (Gly-PESA has good corrosion inhibition performance), and it could be inferred that Gly-PESA formed a protective film on the steel surface, which had low permeability to corrosive ions and had a good protection ability against steel corrosion.
-
3)
CA
![AFM of Q235 steel surface [(a/b) before corrosion; (c/d) blank system; (e/f) System with Gly-PESA(400 mg/L); (g) corresponding height distribution on Q235 steel surface of new sppecimen and without or with Gly-PESA(400 mg/L)].](/content/184/2024/17/12/img/10.1016_j.arabjc.2024.106033-fig12.png)
- AFM of Q235 steel surface [(a/b) before corrosion; (c/d) blank system; (e/f) System with Gly-PESA(400 mg/L); (g) corresponding height distribution on Q235 steel surface of new sppecimen and without or with Gly-PESA(400 mg/L)].
CA is an angle formed when the liquid was droped onto a metal surface and can be used to characterize the wettability of a metal surface. In this work, the wettability of metal surface before and after corrosion was expressed by measurement of CA, which the corrosion inhibition performance of Gly-PESA was verified. For the test pieces, the values of CA were 118°(before corrosion), 55.5°(blank) and 92.0°(with Gly-PESA) respectively, as shown in Fig. 13. From the value of CA, it could be concluded that the hydrophobicity of the metal surface in the presence of Gly-PESA was somewhat weakened compared with the metal surface (before corrosion), but it was much enhanced compared with the metal surface (in the blank solution). This was due to the fact that the Gly-PESA molecules adsorbed and formed a protective layer on the steel surface. Therefore, the presence of Gly-PESA slowed down the corrosion of steel and improved the corrosion inhibition efficiency.
-
4)
XPS
![CA of Q235 steel surface [(a) before corrosion; (b) blank system; (c) System with Gly-PESA(400 mg/L)].](/content/184/2024/17/12/img/10.1016_j.arabjc.2024.106033-fig13.png)
- CA of Q235 steel surface [(a) before corrosion; (b) blank system; (c) System with Gly-PESA(400 mg/L)].
XPS was a powerful technique for detecting the bonding information of corrosion inhibitors on metal surfaces. Therefore, XPS measurements were performed on the steel test piece immersed in solution with Gly-PESA. Fig. 14 showed the XPS scanning full spectrum and high resolution spectrum of the steel surface. As shown in Fig. 14a, Fe, C and O elements existed on the surface of the steel test piece. In Fig. 14b, the Fe2p3/2 spectrum had three main peaks. The peak at 705.21 eV was attributed to metallic iron (Qiang et al., 2021). The peak value of 708.91 eV was related to FeCl2 and FeOOH (Qiang et al., 2021). In addition, the peak of 724.22 eV was attributed to Fe2O3 and FeCl3 (Wan et al., 2022). The spectrum of C1s (Fig. 14c) had three peaks. 284.70 eV was C−C or C−H, 285.80 eV was C−O or C−O−C, and 288.30 eV was C⚌O (Wan et al., 2022, Aah et al., 2020). The O1s spectrum (Fig. 14d) was decomposed into three peaks. The peak at 526.50 eV corresponded to the oxide of iron, the peak at 528.80 eV corresponded to C−O−C, forming a hydrated iron oxide (i.e. FeOOH), and the peak at 530.5 eV was C−OH, the corresponding compound was a complex of water and iron salts (Qiang et al., 2021, Aah et al., 2020, Gu et al., 2023). XPS further demonstrated that Gly-PESA formed a protective film on the steel surface and prevented the steel from being corroded.![XPS diagram of Q235 steel after soaking in solution with Gly-PESA(400 mg/L) [(a) XPS survey; (b)Fe2p3/2; (c)C1s; (d)O1s].](/content/184/2024/17/12/img/10.1016_j.arabjc.2024.106033-fig14.png)
XPS diagram of Q235 steel after soaking in solution with Gly-PESA(400 mg/L) [(a) XPS survey; (b)Fe2p3/2; (c)C1s; (d)O1s].
3.5 Analysis of corrosion inhibition mechanism
In order to investigate the inhibition mechanism of Gly-PESA, adsorption isothermal equation was used for further analysis. In addition, the whole corrosion inhibition process was explained in detail.
3.5.1 Adsorption isotherm
The corrosion inhibition performance of the organic corrosion inhibitor mainly depended on its adsorption effect on the metal surface. Therefore, in order to explain the corrosion inhibition mechanism, the uses of adsorption isotherms such as Langmuir, Floy-Huggin, Bockris-Swinkels, Frumkin and Temkin isotherms were an effective method (Liu et al., 2023). More specifically, we chose the Langmuir formula for the experiment because it had the best degree of fitting. Therefore, other isotherms weren’t considered.
According to equation (3), Fig. 15 depicted the relationship between Cinh/θ and Cinh, and the discrete points were fitted into a straight line, with the correlation coefficient R2 = 0.99417. This indicated that the adsorption behavior of Gly-PESA molecules conformed to Langmuir formula, indicating that the interaction between molecules can be ignored. The following further used Kads to calculate the adsorption free energy ΔGads0: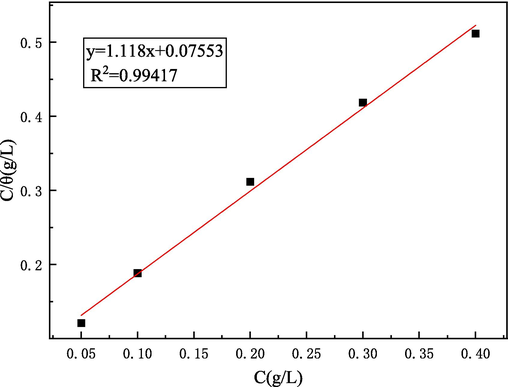
Langmuir adsorption isotherm of Gly-PESA on Q235 steel surface.
|ΔG0ads|≤20 kJ /mol and |ΔG0ads|≥40 kJ/mol were the physical adsorption and chemical adsorption of corrosion inhibitor molecules on metal surface, respectively (Liu et al., 2023; Belkheiri et al., 2024). Based on the above data, it could be concluded that the adsorption behavior of Gly-PESA on the steel surface includes physical adsorption and chemical adsorption. More specifically, in terms of physical adsorption, there was electrostatic attraction when the Gly-PESA molecule was close to the steel surface. In terms of chemisorption, the transferring and sharing of electrons between Gly-PESA molecules and empty d orbitals of iron atoms formed a complex covalent bond.
3.5.2 Corrosion inhibition process
The corrosion inhibition system mainly involved the oxidation of iron atoms in the anode metal and the reduction of oxygen in the cathode. The reactions were as follows (Chen et al., 2019):
According to formulas (5) and (6), it could be found that in the blank corrosion system: (i) the dissolution of the steel test piece was accelerated, corrosion products were formed on the surface (such as compounds of Fe2+ and Fe3+), and (ii) the reduction reaction of oxygen produced OH–, which changed the pH value of the steel surface. When Gly-PESA was added, the corrosion process would be changed. First, due to static electricity, the Gly-PESA molecules were adsorbed on the surface of the steel. The adsorption process could be explained by the displacement reaction of water molecules with Gly-PESA. The substitution reaction equation was as follows (Chen et al., 2019):
[Fe… Gly-PESA]2+(ad) represented the complex on the steel surface. It could also be found from equation (8) that when the concentration of Gly-PESA was low, a dense and uniform protective film cannot be formed. Therefore, in practical applications, the dosage of Gly-PESA was increased so that the Gly-PESA molecules combined with iron ions to form enough complexes to protect carbon steel.
Therefore, in the absence of Gly-PESA, the surface of the steel piece was covered with water molecules and a corrosive medium (such as chloride ions and a small amount of oxygen molecules). After the addition of Gly-PESA, its molecules would adsorb on the surface of the steel, separating the carbon steel from the corrosive medium. In addition, due to the many polar groups of Gly-PESA, a dense protective film could be formed on the surface of carbon steel (such as SEM, AFM, XPS analysis), which increased the resistance to erosion, thereby protecting the steel test piece to slow down corrosion.
4 Conclusion
A new scale and corrosion inhibitor (GLY-PESA) was synthesized with PESA and Gly, and characterized by FTIR, 1H NMR and TGA. The scale inhibition efficiency of CaCO3 and CaSO4 was 98.06 % and 92.6 %, respectively and the scale inhibition effect of Gly-PESA was obviously better than that of PESA under the same experimental conditions. SEM, FTIR and XPS experiments showed that Gly-PESA could effectively inhibit the growth of CaCO3 and CaSO4 crystals and cause their crystal lattice distortion or crystal dispersion. As a green corrosion inhibitor, the inhibition efficiency of Gly-PESA on carbon steel reached 78.17 % and the adsorption of Gly-PESA followed Langmuir adsorption isotherm. In addition, through SEM, FTIR, AFM, XPS and CA, it was proved that Gly-PESA formed a protective film on the surface of carbon steel, which increased the corrosion resistance of carbon steel, and effectively protected the surface of the steel. In summary, Gly-PESA was a kind of environment-friendly non-phosphorus scale and corrosion inhibitor, which had good scale and corrosion inhibition performance in industrial circulating cooling water, and broadened the application range of PESA, providing a new idea for the development of industrial scale and corrosion inhibitor.
5 Author agreement
All authors have approved of the publication of this paper. We declare that the paper is the original research.
CRediT authorship contribution statement
Xinhua Liu: Writing – review & editing, Funding acquisition, Formal analysis, Data curation, Conceptualization. Yuhua Gao: Writing – original draft, Formal analysis. Baojing Luo: Formal analysis. Hongxia Zhang: Formal analysis. Xiaoyu Shi: Writing – review & editing, Funding acquisition. Yuan Zhang: Project administration. Yongguang Gao: Investigation. Ying Wang: Software. Zilin Zheng: Data curation. Nan Ma: Data curation. Jiarui Du: Funding acquisition. Linyan Gu: Data curation.
Acknowledgments
This work was financially supported by Natural Science Foundation of Hebei Province of China (D2022105004), the Foundation of Phased Achievement of the Key Cultivation Project of Tangshan Normal University of China (ZDPY07, ZDPY05), Science and Technology Program of Hebei Academy of Sciences of China (No. 24702), and Science and Technology Research Project of Higher Education of Hebei, China (QN2024182).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Ficus racemosa leaf extract for inhibiting steel corrosion in a hydrochloric acid medium. Alex. Eng. J.. 2020;59(6):4449-4462.
- [CrossRef] [Google Scholar]
- The effect of some green inhibitors on the corrosion rate of Cu, Fe and Al metals. Int. J. Corros. Scale Inhib.. 2019;8(2):199-211.
- [CrossRef] [Google Scholar]
- Advanced evaluation of novel quinoline derivatives for corrosion inhibition of mild steel in acidic environments: A comprehensive electrochemical, computational, and surface study. Int. J. Electrochem. Sci.. 2024;19:100772
- [CrossRef] [Google Scholar]
- Corrosion inhibition performance of threonine- modified polyaspartic acid for carbon steel in simulated cooling water. J. Appl. Polym.. 2019;47242:1-12.
- [CrossRef] [Google Scholar]
- Lycium barbarum leaf extract as biodegradable corrosion inhibitor for copper in sulfuric acid medium. Ind. Crop. Prod.. 2023;203:117181
- [CrossRef] [Google Scholar]
- Study on the Corrosion and Scale Inhibition Mechanism of the Thiourea-Modified Polyepoxysuccinic Acid (CNS-PESA) J. Chem. 2022
- [CrossRef] [Google Scholar]
- Corrosion inhibitors. A review”. Int. J. Corros. Scale Inhib.. 2021;10(1):54-67.
- [CrossRef] [Google Scholar]
- Study on scale and corrosion inhibition performance of ethane-1,2-diaminium-O-O’-dicyclohexyldithiophosphate. Int. J. Electrochem. Sci.. 2020;15(11):11306-11315.
- [CrossRef] [Google Scholar]
- Study on synthesis and modification of polyepoxysuccinic acid. Frontiers Edu. Res.. 2020;3(6):30-33.
- [CrossRef] [Google Scholar]
- Study of corrosion and scale inhibition of polyepoxysuccinic acid derivative. Asian J. Chem.. 2014;26(22):7716-7720.
- [CrossRef] [Google Scholar]
- Scale/corrosion inhibition and biodegradation of lysine modified polyaspartic acid. CIESC J.. 2018;69(5):2127-2136.
- [CrossRef] [Google Scholar]
- Synthesis of polyaspartic acid-glycidyl adduct and evaluation of its scale inhibition performance and corrosion inhibition capacity for Q235 steel Applications. Arab. J. Chem.. 2023;16:104515.
- [CrossRef] [Google Scholar]
- The research progress of water treatment technology on recirculated cooling water. IOP Conf. Ser.: Earth Environ. Sci.. 2020;508
- [CrossRef] [Google Scholar]
- A review of green scale inhibitors: process, types, mechanism and properties. Coatings. 2020;10(10):928.
- [CrossRef] [Google Scholar]
- Hyperbranched polyesters with carboxylic or sulfonic acid functional groups for crystallizationmodification of CaCO3. Macromol. Chem. Phys.. 2007;208:474-484.
- [CrossRef] [Google Scholar]
- Effect of dendrimers on the crystallization of CaCO3 in aqueous solution. Top. Curr. Chem.. 2003;228:141-158.
- [CrossRef] [Google Scholar]
- Study of PBBTCA/PESA complex component as a scale inhiitor against calciuum sulfate. Appl. Chem. Ind.. 2022;51(5):1341-1344.
- [CrossRef] [Google Scholar]
- Fabrication of environmentally friendly Losartan potassium film for corrosion inhibition of mild steel in HCl medium. Chem. Eng. J.. 2021;406:126863
- [CrossRef] [Google Scholar]
- Green approach to corrosion inhibition of mild steel in hydrochloric acid and sulphuric acid solutions by the extract of Murraya koenigii leaves. Mater. Chem. Phys.. 2010;122(1):114-122.
- [CrossRef] [Google Scholar]
- Recent Advances in Metallic Corrosion Inhibition: A Review. J. Mol. Liq.. 2021;322:14862.
- [CrossRef] [Google Scholar]
- Molecular dynamics simulation for interaction of PESA and acrylic copolymers with calcite crystal surfaces. Desalin.. 2012;291:8-14.
- [CrossRef] [Google Scholar]
- Corrosion and scale processes and their inhibition in simulated cooling water systems by monosaccharides derivatives: Part I: EIS study. Desalin. 2009;249(3):922-928.
- [CrossRef] [Google Scholar]
- Soybean extract firstly used as a green corrosion inhibitor with high efficacy and yield for carbon steel in acidic medium. Ind. Crop Prod.. 2022;187:115354
- [CrossRef] [Google Scholar]
- Wang C., Gao B., Zhao P., et al., 2017. Exploration of polyepoxysuccinic acid as a novel draw solution in the forward osmosis process. RSC Adv., 7(49): 30687–30698. https://doi.10.1039/c7ra04036arsc.li/rsc-advances.
- Performance and mechanism study of PESA-IA as a green oilfield scale inhibitor: experimental and molecular dynamics simulation. J. Polym. Res.. 2022;29:525.
- [CrossRef] [Google Scholar]
- Synthesis and Application of a Phosphorous-Free and Non-Nitrogen Polymer as an Environmentally Friendly Scale Inhibition and Dispersion Agent in Simulated Cooling Water Systems. ACS Omega. 2020;5:15487-15494.
- [CrossRef] [Google Scholar]
- Corrosion inhibition of molybdate and nitrite for carbon steel corrosion in process cooling water. J. Eng. Sci. Technol.. 2019;14(4):2431-2444. http://scholars.utp. edu.my/id/eprint/25236
- [Google Scholar]
- Study on a novel composite ecofriendly corrosion and scale inhibitor for steel surface in simulated cooling water. J. Surf. Eng. Mater. Adv. Technol.. 2012;2(3):137-141.
- [CrossRef] [Google Scholar]
- Evaluation of Arginine-Modified Polyepoxysuccinic Acid as Anti-scaling and Anti-corrosion Agent. Chem. Eng. Technol.,. 2021;44(6):1131-1140.
- [CrossRef] [Google Scholar]
- Scale inhibition performance and mechanism of sulfamic/amino acids modified polyaspartic acid against calcium sulfate. Desalin.. 2017;419:152-159.
- [CrossRef] [Google Scholar]
- Testing the formation of Ca-phosphonate precipitates and evaluating the anionic polymers as Ca-phosphonate precipitates and CaCO3 scale inhibitor in simulated cooling water. Corros. Sci.. 2010;52(12):3883-3890.
- [CrossRef] [Google Scholar]
- Synthesis, scale inhibition performance evaluation and mechanism study of 3-amino-1-propane sulfonic acid modified polyaspartic acid. Copolym. J. Mol. Struct.. 2023;1272:134141.
- [CrossRef] [Google Scholar]
- Synthesis and evaluation of amino acid modified polyepoxysuccinic acid as inhibitor of calcium carbonate scale. Water Supply. 2022;22(12):8923-8941.
- [CrossRef] [Google Scholar]






