Translate this page into:
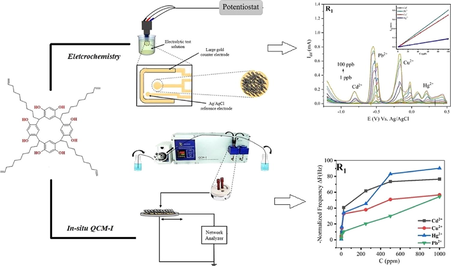
In-situ electrochemical and piezogravimetric studies on the application of macrocyclic resorcinarene tetramer in the development of chemically-modified heavy metals ions detection platform in aqueous media
⁎Corresponding author. shaban.abdul@ttk.hu (Abdul Shaban)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Abstract
The new application of C-dec-9-enylcalix[4]resorcinarene (R1), as an ionophore to detect heavy metals (HMs) cations (Cd2+, Hg2+, Cu2+, and Pb2+) in the aqueous media has been investigated through the preparation of an effective mass-sensitive sensor via the exploitation of a flow-type QCM-I technique. By adjusting the ions’ amounts in model solutions over a wide range of concentrations, acquired changes in the oscillating frequency related to the loading of metal ions on the sensor’s surface were gained, and thus favorable metrological parameters displaying the lowest detection limit (LOD) associated with copper ions (10 ppb). Simultaneously, a novel voltammetric sensor was prepared by modifying gold screen-printed electrodes (SPEs) with R1. Electrochemical characterization employing CV, SWV, and EIS was carried out, showing the success of the electrode modification. Then, the experimental conditions of supporting electrolyte, pH, accumulation time, and accumulation potential were optimized to achieve an enhanced detection. The R1@SPE sensor simultaneously detected the HMs (Cd2+, Hg2+, Cu2+, Pb2+), and the lowest LOD was associated with Pb2+ (0.19 ppb). The selectivity evaluation of the electrochemical sensor was performed by studying the effect of interferences majorly present in water sources (Mg2+, Ni2+, Zn2+, Al3+, and K+) on the SWV detection signals, and it was revealed that the interfering ions did not affect the simultaneous detection of the studied HMs (RSD less than 5%), the voltammetric sensors also presented excellent repeatability and reproducibility (RSD less than 5%).
Keywords
Calix[4]resorcinarene
Mass-sensitive sensor
Heavy metals
Detection
Voltammetric sensors
1 Introduction
Environmental contamination by heavy metals (HMs) is a hazard to the environment and is one of the most vital challenges in modern humanity (Li et al., 2013; Briffa et al., 2020). Trace levels of these toxicants if present in the ecosystem (soil, water surfaces…etc.) can cause dangerous effects on animal health (Woo et al., 2016). Mostly, entering the body via direct consumption of contaminated water and food beverages (Stewart et al., 2011), their toxicity is manifesting in the substitution of essential metals in the human body by interacting with enzymes and nucleic acids through binding to thiol groups of DNA, and so altering the body’s functional properties (Lim and Schoenung, 2010; Stewart et al., 2011; Briffa et al., 2020).
The world health organization (WHO) and US Environmental Protection Agency (EPA) have taken giant steps to enlighten the awareness of the hazard and regulation of the thresholds concentration has been recommended (Mahajan B., 2021; USEPA, 2022). However, the identification and removal of these toxins from scarce water sources are vital necessities for communal protection.
There is a growing need for an ecologically safe, systematic, and novel methodology to remediate toxic heavy metals in water streams. The conventional approaches have limitations in terms of being noneconomic, requiring energy, and inefficiency. Common approaches to removing heavy metals from water sources are not wildly offered over the world. Amongst the best effective progressive remediation processes, adsorption is extensively considered as the maximum effective method for handling a wide variety of heavy metals contaminants in aqueous systems. As a result, much research has focused on the use of low-cost adsorbents for heavy metal removal (Laghrib et al., 2021). For the remediation of environmental pollutants, sonophotocatalytic is a recently applied technique, where the complete improvement in the degradation activity can be credited to the synergic properties of sonolysis and photocatalysis, which lead to the formation of reactive free radicals (Theerthagiri et al., 2021; Yu et al., 2021).
The early environmental detection of HMs is crucial, their recognition in real samples is a difficult task owing to the complexity of various natural matrices (aquatic and river media), besides the very low concentrations of these ions, often below the detection limits of available techniques (Aragay et al., 2011; Elkhatat et al., 2021). On large scale, myriads of analytical procedures and physicochemical tools have been used to gather information on HM ions detection and monitoring in water sources, namely: FES, AAS, ICP-MS, ICP-OES, INAA, and UV/Vis (Kenawy et al., 2000; Hajiaghababaei et al., 2013; Jin and Ainliah, 2021), these approaches provide excellent and complete information in terms of metrological parameters related to sensitivity and selectivity. Nevertheless, the disadvantages and constraints are linked to the difficulty of use (need of skilled trained scientists) and time-consuming analysis (sampling, preparation, calibration, etc.).
At present, the reputation of sensors is well acknowledged, on account of their capability for conducting recognition investigations that were once dominated by analytical chemistry techniques, more advantages are offered by sensors as instrumentation low-cost, portability, data acquisition speed, technical reliability, real-time analyses, label-free onsite employment, and an overtime mapping-out of the target elements’ existence in the studied environment, this later benefit is prohibitively costly when it comes to traditional detection procedures (Fadillah et al., 2020; Wu et al., 2019). The design, construction, integration, and real application of HMs detection chemosensors, are comprehensively studied and reported in the literature, upon employing sensing platforms ranging from inorganic (Prochowicz et al., 2017) and nanomaterials (Deshmukh et al., 2018) to organic and macrocyclic elements (Bettazzi et al., 2012; Luo et al., 2019), by way of illustration calixarenes and resorcinarenes (Arora et al., 2007; Mei C. J. and Ahmad S. A. A., 2021), which had known ever-accelerating progress with regards to synthesis and structural alterations by modifying the cone dimensions. However, the mainstream of calixarene-based chemo-sensing platforms targeting HMs recognition in water matrices is of either electrochemical concept (voltammetry, amperometry, potentiometry, for instance, Ion-Selective-Electrodes (ISEs)) (Ahmadzadeh et al., 2015; Akl and Abd El-Aziz, 2016; Naik et al., 2021; Taghvaei-Ganjali et al., 2009; Pujol et al., 2014; Ebdelli et al., 2011; Göde et al., 2017; Nur Abdul Aziz et al., 2018) or Optical principle (fluorescence, colorimetry, etc.) (Chawla and Gupta, 2015; Echabaane et al., 2013; Erdemir et al., 2016; Lotfi et al., 2017; Mokhtari et al., 2011; Yang et al., 2019). In the field of environmental protection and monitoring, there is a tendency in sensing to use electrochemical impedance spectroscopy (EIS), cyclic voltammetry (CV), and SWV in combination with other measurement methods such as quartz crystal microbalance (QCM) (Briand et al., 2010).
The Quartz Crystal Microbalance with impedance measurements (QCM-I) involves two gold electrodes plated onto a piezoelectric thin quartz disk. Any adsorbed or desorbed mass on the gold surface causes a variation in the oscillation frequency (ΔF); an increase in mass causes a decrease in frequency and vice versa. This ΔF is proportional to the amount of variation in mass (Δm), as shown by the Sauerbrey equation (Eq. (1)) (Sauerbrey, 1959).
Where n is the overtone number and C is the mass sensitivity constant, which only depends on the physical properties of the quartz crystal.
QCM-I employs a network analyzer to permit continuous measurements of resonance parameters e.g. frequency, resonance curve, Full Width at Half Maximum variation (ΔFWHM) calculations, along with monitoring the layer’s viscoelastic properties via dampening energy evaluations, a.k.a. dissipation shifts (ΔD).
Mass-sensitive piezogravimetric techniques, as the QCM, showed their effectiveness in analytical applications for toxic metallic elements ions sensing by engaging numerous surface modifiers (Cao et al., 2011; Chen et al., 2011; Sartore et al., 2011; Emir Diltemiz, S. et al., 2017). However, apart from our published investigations (Eddaif et al., 2019a, 2020; Shaban and Eddaif, 2020), there’s a lack of systematic studies dealing with the utilization of QCM to develop Calix-sensors for detecting HMs ions in aqueous solutions. Some consideration was given to the development of dicarboxyl-Calix[4]arene-based electrochemical sensing platform to detect lead ions in an aqueous solution (NurAbdul Aziz et al, 2018).
For that reason, this contribution aims to study the detection capability of a resorcinarene tetramer (C-dec-9-enylcalix[4]resorcinarene) towards toxic metallic ions, (Cd2+, Hg2+, Cu2+, Pb2+), in aqueous model solutions, by applying the QCM-I, and electrochemical methods (cyclic voltammetry (CV), and square wave voltammetry (SWV)).
2 Experimental
2.1 Chemicals
The used chemicals in the experimental part of this work are listed in Table 1. All chemicals from commercial sources were of analytical grade and used directly without further purification if no special instructions. All solutions were prepared and diluted by ultrapure water from the Milli-Q water purifier system.
Name
Formula
Characteristics
Purchased from
Hydrogen peroxide (30%)
H2O2
Extra pure
Molar Chemicals KFT
Tetrahydrofuran
(THF)C4H8O
Extra pure
Molar Chemicals
KFT
Sulfuric acid (96%)
H2SO4
Analytical grade
Molar Chemicals KFT
Hydrochloric acid (37%)
HCl
Analytical grade
Molar Chemicals KFT
Glacial acetic
acid (+99 %)CH3CO2H
Analytical grade
Sigma-Aldrich
Sodium hydroxide
(+98 %)NaOH
Analytical grade
Sigma-Aldrich
Ammonium acetate
CH3CO2NH4
Analytical grade
Merck
Ammonia (25%)
NH3
Analytical grade
Molar Chemicals KFT
Nitric acid (65%)
HNO3
Analytical grade
Molar Chemicals KFT
Hellmanex III
___
Alkaline cleaning concentrate
Hellma GmbH & Co. KG (Germany)
Pb(II) nitrate
Pb(NO3)2
99.99%
Sigma-Aldrich
Cd(II) nitrate
tetrahydrateCd(NO3)2·4H2O
98%
Sigma-Aldrich
Hg(II) chloride
HgCl2
99.5%
Sigma-Aldrich
Cu (II) nitrate trihydrate
Cu(NO3)2.3H20
Extra pure
Sigma-Aldrich
Mg(II) chloride
hexahydrateMgCl2.6H20
Analytical grade
Molar Chemicals KFT
Al(III) nitrate
nonahydrateAl(NO3)3·9H2O
Analytical grade
Sigma-Aldrich
Ni(II) nitrate
hexahydrateNi(NO3)2·6H2O
Analytical grade
Reanal
K(II) sulfate
K2SO4
99.5 %
Molar Chemicals KFT
K(I) chloride
KCl
+ 99 %
Sigma-Aldrich
Trans-2,cis-6-nonadienal (+96%)
C9H14O
d = 0.866
Sigma-Aldrich
Undecylenic aldehyde
(95%)C11H20O
d = 0.81
Sigma-Aldrich
(S)-(-)-α-Methylbenzylamine
(p ≥ 98%)
C8H11N
d = 0.952
Fluka Chemie
GmbH
(R)-(+)-α-Methylbenzylamine
(p ≥ 98%)
C8H11N
d = 0.952
Fluka Chemie
GmbH
Dodecanal
C12H24O
d = 0.831
Sigma-Aldrich
Paraformaldehyde
HO(CH2O)nH
Reagent grade
Molar Chemicals
KFT
2.2 Preparation of the ionophore
C-dec-9-enylcalix[4]resorcinarene (R1) was prepared by dissolving resorcinol (0.23 mmol, 0.025 g) and 0.23 mmol of aldehyde (undec-10-enal) into absolute ethanol (240 mL) (Aoyama et al., 1989). The solution was cooled down to 0 °C, and 37 mL of concentrated HCl was added, after an hour of stirring, the solution was heated and refluxed for twelve hours. The recrystallization via methanol and a (1:1) mixture of acetone: hexane, gave R1 as a yellowish-orange solid (Yield: 49 %, m.p.:277 °C).
The resorcinarene tetramer was synthesized based on the acid-catalyzed cyclo condensation reactions, viz.: C-dec-9-enylcalix[4]resorcinarene (R1), and was the subject of full structural characterization studies employing FT-IR, 1H/ 13C NMR, powder XRD, and thermal analyses (Eddaif et al., 2019b). The chemical structure of (R1) is presented in Fig. 1.![Molecular structure of C-dec-9-enylcalix[4]resorcinarene (R1).](/content/184/2022/15/5/img/10.1016_j.arabjc.2022.103780-fig2.png)
Molecular structure of C-dec-9-enylcalix[4]resorcinarene (R1).
2.3 Pretreatments of electrodes
For the piezogravimetric analysis, AT-cut gold-plated quartz crystal resonators (QCR) with a 5 MHz fundamental frequency and 14 mm diameter, were used. The QCR’s gold surface was rinsed by acetone (a.r., Reanal), washed with piranha solution (H2O2 (30%) and H2SO4 (96%) in a ratio of (1:2) for 10 min, rigorously rinsed with Milli-Q water, and finally, let dry at room temperature inside a desiccator.
For electrochemical experiments, screen-printed gold electrodes (Pine Research, USA) were used. The electrodes (Pine research, USA) were composed of three electrodes printed system (2 mm OD gold working electrode, a U-shaped large gold counter electrode, and an Ag/AgCl reference electrode) connected to the potentiostat through a connector cable. Potential contaminants on screen-printed electrodes (SPEs) were removed by cyclic voltammetric cleaning by applying 20 CV cycles at 100 mV.s−1 scan rate in 0.05 M H2SO4 solution in the potential range from −1.5 to + 0.5 V. The electrochemical cleaning resulted in stable voltammograms, and the cleaned SPEs were rinsed with Milli-Q water (resistivity/conductivity at 25 °C: 18 MΩ.cm−1 /5.5x10-6 S.m−1) and placed in a desiccator at room temperature until further use.
2.4 Ionophore immobilization on the gold sensing area
For immobilizing the macrocycle onto the gold sensing area, R1 was dissolved in chloroform (a.r., Molar Chemicals Kft) (2 mg.mL−1). With subsequent intensive drop-casting of the ionophore solution on the clean QCR’s gold surface (10 µL) and screen-printed electrodes (2 µL), the resorcinarene based sensing platforms were fabricated. The modified electrodes were later dried at room temperature and kept inside a desiccator (25 °C) awaiting further use.
2.5 QCM-I measurements
The HMs salts (Sigma- Aldrich) (Pb(NO3)2, Cd(NO3)2, HgCl2, and Cu(NO3)2) were mixed at convenient amounts, with Milli-Q water, to prepare the water model solutions with concentration range 5–1000 ppm. Label-free onsite real-time QCM-I detection experiments were performed utilizing the QCM-I instrument (MicroVaccum Ltd, Hungary). The device’s resonance sensitivity in the liquid is 0.2 Hz, the dissipation sensitivity is 1.10−7, and the mass sensitivity is ≤ 1 ng.cm−2.
The apparatus enclosed a 40 μL flow-cell and was automatically thermostated (25 °C), and the applied flowrate was 200 µL.min−1 for injecting the HMs solutions by a peristaltic pump. Experiments were computer-controlled using the BioSense Software V. 3.1 (MicroVaccum Ltd). The data were recorded for selected overtones n = 1, 3, 5, and 7, on behalf of 5, 15, 25, and 35 MHz frequencies, respectively. The normalization of ΔF is accomplished by dividing the change in the measured values by the used overtone number (n) (Eq. (2)):
2.6 Electrochemical experiments
The voltammetric tests, viz. cyclic voltammetry (CV) and square wave voltammetry (SWV) were performed using a Gamry interface 1010E potentiostat/galvanostat apparatus controlled by the Gamry inst. framework software, whereas the electrochemical impedance spectroscopy (EIS) experiments were carried out using a Solartron frequency analyzer/electrochemical interface system (1250/1286) controlled by the ZPlot software.
2.6.1 Electrochemical characterization
EIS is a well-known sensing method to examine the statuses of affinity reactions on electrode surfaces established on the interaction of the analyte with the electrical double-layer or the perturbation of the redox reactions. Electrochemical processes that take place around the vicinity of an electrode surface can be analyzed using the EIS technique. EIS in chemosensor applications focuses on surface actions taking place in a frequency region typically from mHz to KHz.
Prior to the electrochemical characterization tests, the SPE was immersed into the cell containing 0.2 M HCl and the target metal ions with a 3 min stirring time before their accumulation at open circuit potential (OCP). The electrochemical characterization of the bare/modified electrodes in the absence/presence of 1 ppm of HMs dissolved in 0.2 M HCl solution was completed employing the following conditions:
In the case of EIS, the ac voltage was set to Vac = 10 mV amplitude sine wave signal, whereas the dc voltage (bias) remained at the open-circuit potential (OCP) in order to stay in the linear region of the Butler-Volmer equation. The applied frequency range was from 65 kHz to 1 Hz.
Cyclic voltammetry is a measurement procedure in which the potential is driven between the working electrode (WE) and the reference electrode in the form of slow cyclic changing triangle-shaped voltages. At the same time, the current between WE and counter electrode (CE) is recorded which produces an exact analysis of potential oxidizing (Ox) and reducing (Red) species present in the solution. Forward and back CV scans were performed in the potential range from −0.75 V to + 0.5 V at a scan rate = 50 mV.s−1. Square wave voltammetry measurements were executed at frequency = 15 Hz, deposition time = 60 s, deposition potential of −1V within the potential window from −1 to + 0.5 V.
2.6.2 Experimental parameters optimization
Aiming to enhance the detection capacity of the prepared sensor towards various heavy metals, the physicochemical parameters having a direct influence on the SWV signals were optimized. SWV is a fast electroanalytical method that depends on the frequency (Hz) and step height (mV). It generates a peak-shaped symmetrical voltammogram. The current is recorded twice during each cycle of the square wave, once at the end of the forward pulse and once at the end of the reverse pulse. The concentration of the investigated HM ions is proportionate to the current peak. It has a high sensitivity because the current peak is larger than the oxidation/reduction separated signals.
The influence of various supporting electrolytes was examined in order of choosing the most suitable one for offering the highest detection magnitude. The square wave voltammetry conditions were similar to those employed in the electrochemical characterization. The studied electrolytes are HCl 0.2 M (pHi = 0.7), KCl 0.2 M (pHi = 6.8), and acetate buffer ACB 0.2 M (pHi = 5.3), prepared by mixing 15.4 g of ammonium acetate and 2.06 mL of glacial acetic acid in 990 mL of distilled water.
The electrolytic medium’s pH is highly affecting the interactions between the sensing platform and the target ions. Therefore, the detection response in 0.2 M HCl has been investigated by changing the pH values from 0.7 to 8. The pH was adjusted by 0.1 M NaOH (pHi = 13) and was monitored by the ADWA AD8000 digital pH meter. The SWV conditions were similar to those employed in the electrochemical characterization.
The optimization of the accumulation time and potential effects on the R1@SPE sensing platforms were studied within the time-frame of 30–180 s and within potential values from −2V to −1V, with the purpose to produce higher signals and improved detection performance.
2.6.3 Simultaneous electrochemical detection of heavy metals (Calibration studies)
The detection capability of the R1@SPE sensor towards various heavy metals ions, with concentrations ranging from 1 to 100 ppb, was examined under the optimized circumstances, for the determination of the statistical detection characteristics (linear ranges, detection, and quantification limits).
2.6.4 Interference studies
The most common potential interfering metals cations were Al3+, Fe3+, Fe2+, Mg2+, Co2+, Mn2+, Zn2+, Pb2+, Cd2+, Sn2+, and Ni2+, as studied by Pratiwi and coworkers (Pratiwi et al., 2017), where they showed that the presence of common metal ions did not interfere with Cu2+ detection within reasonable tolerance ratios.
The selectivity of the R1@SPE platform for Pb2+, Cd2+, Cu2+, Hg2+ ions was evaluated in the presence of other metals frequently present in environmental samples, Al3+, K+, Mg2+, Zn2+, and Ni2+, usually present in water sources. Interfering ions of 4 ppm (40 folds compared to target ions) were added to 0.2 M HCl containing 100 ppb of heavy metals ions (Pb2+, Cd2+, Cu2+, Hg2+), and SWV detection studies were performed under optimized conditions.
2.6.5 Reproducibility and repeatability
To evaluate the repeatability of the fabricated sensor, its sensing platform was employed for detecting 100 ppb of heavy metals under optimized conditions by running 3 successive measurements. Hence, for assessing the reproducibility, three different SPEs modified by R1 were used to detect 100 ppb of heavy metals ions under optimized conditions. The standard deviation (RSD) was calculated to evaluate these parameters.
3 Results and discussion
3.1 QCM-I studies
3.1.1 Effect of heavy metals ions on unmodified quartz crystal electrodes
As a first step, the effect of heavy metal ion concentration was studied on the bare gold electrodes of quartz crystals, frequency and dissipation shifts through the in-situ QCM-I measurements were recorded. As seen in Fig. 2, the unmodified gold electrodes did not detect any loading of the heavy metals ions on the gold surface (ΔF ≈ 0) (Fig. 2a). A rigid character of the electrodes’ surfaces was dominant, as no changes in dissipation energy were obvious (ΔD ≈ 0) (Fig. 2b). Though, some fluctuations were existing in the majority of plots, which are explained by the electrode’s surface wetting due to straight exposure to heavy metals solutions. The inability of bare gold surfaces to sense heavy metals ions proves that neither physical nor chemical interactions have occurred. It is worth mentioning that the chemical interactions are crucial in increasing the quartz crystal’s sensitivity, where taking advantage of detection networks is compulsory.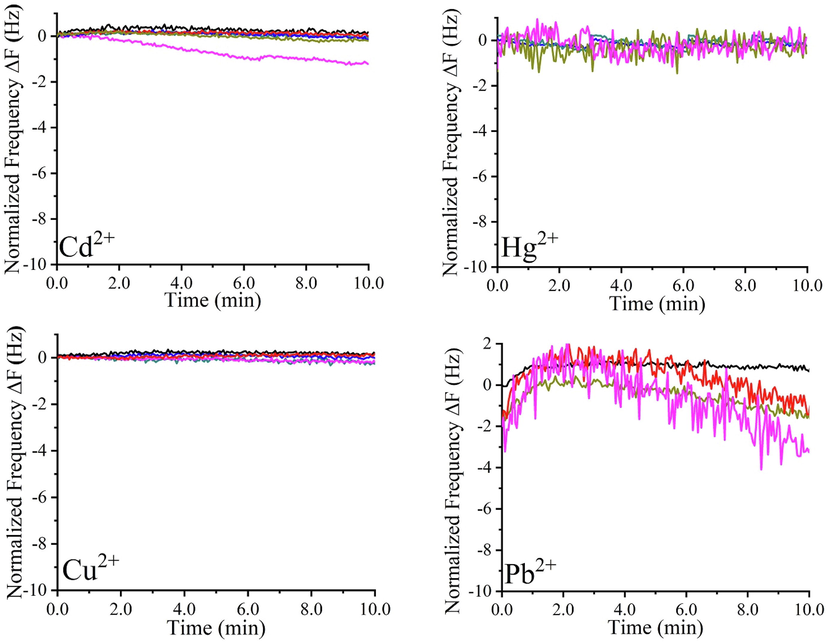
Normalized frequency (a) and dissipation shifts (b) of unmodified QCR against various heavy metals concentrations (0, 5, 25, 250, 500, and 1000 ppm) in time.
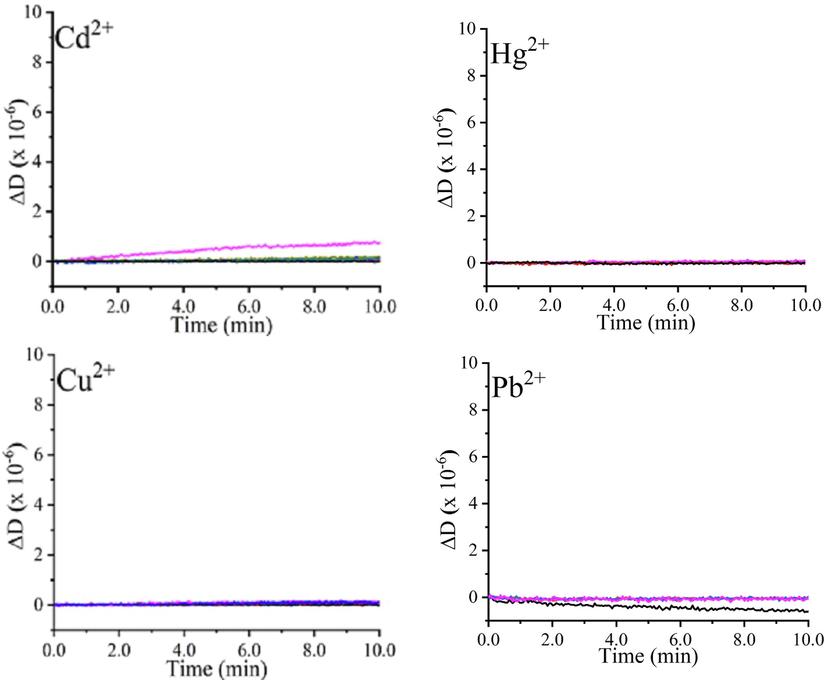
Normalized frequency (a) and dissipation shifts (b) of unmodified QCR against various heavy metals concentrations (0, 5, 25, 250, 500, and 1000 ppm) in time.
3.1.2 Effect of heavy metals ions on the prepared piezogravimetric sensor
Targeting water environmental detection via applied in-situ QCM-I analysis, drop-coated gold electrodes using R1 as sensing platforms were constructed, aiming at evaluating their HMs detection capabilities in model solutions modified via adding toxic metallic elements. Fig. 3 illustrates the normalized frequency (a) and dissipation variations (b) (ΔFN and ΔDN) of Calix-chemosensor R1 in time. Whereas, Table.2 contains a summary of the ΔFN and ΔDN values collected from endpoints of the detection plots.
Normalized frequency (a) and dissipation shifts (b) for ionophore R1 modified QCR against various heavy metals concentrations (0, 5, 25, 250, 500, and 1000 ppm) in time.
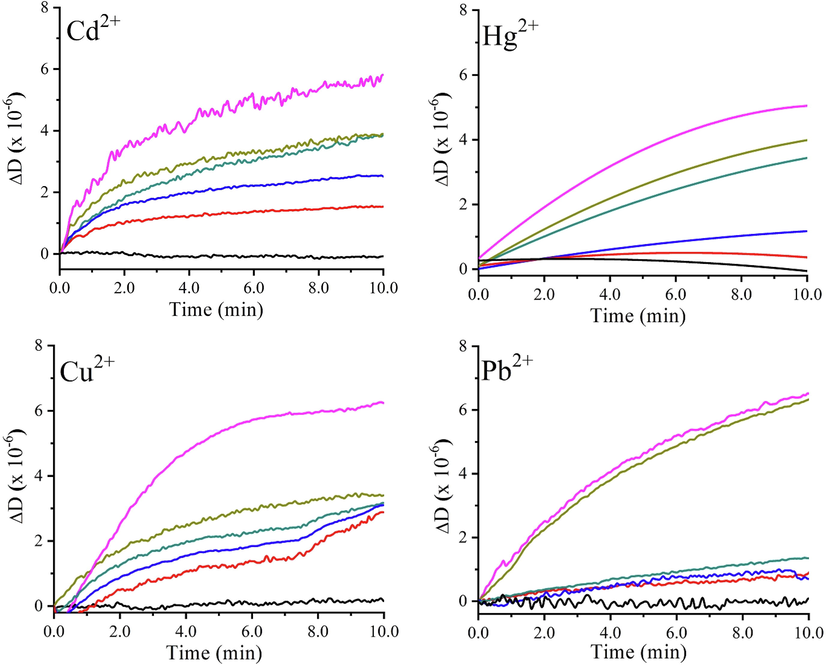
Normalized frequency (a) and dissipation shifts (b) for ionophore R1 modified QCR against various heavy metals concentrations (0, 5, 25, 250, 500, and 1000 ppm) in time.
Metal ions
C
(ppm)
ΔF
(Hz)
ΔD
(x10-6)
Cd2+
0
− 1.14 ± 0.01
0.07 ± 0.02
5
− 9.85 ± 0.04
1.54 ± 0.01
25
− 40.70 ± 0.30
2.51 ± 0.03
250
− 61.70 ± 0.90
3.83 ± 0.04
500
− 73.46 ± 0.78
3.84 ± 0.07
1000
− 76.55 ± 0.50
5.79 ± 0.90
Cu2+
0
− 4.94 ± 0.03
0.12 ± 0.01
5
− 14.42 ± 0.02
2.87 ± 0.04
25
− 32.76 ± 0.07
3.09 ± 0.02
250
− 37.91 ± 0.01
3.19 ± 0.03
500
− 50.91 ± 0.09
3.40 ± 0.09
1000
− 56.68 ± 0.10
6.24 ± 0.10
Hg2+
0
− 0.65 ± 0.08
0.05 ± 0.01
5
− 16.46 ± 0.03
0.36 ± 0.01
25
− 34.27 ± 0.06
1.17 ± 0.02
250
− 45.47 ± 0.07
3.43 ± 0.03
500
− 83.06 ± 0.13
3.98 ± 0.06
1000
− 90.32 ± 0.01
5.05 ± 0.04
Pb2+
0
− 2.13 ± 0.02
0.05 ± 0.03
5
− 6.55 ± 0.01
0.87 ± 0.02
25
− 10.48 ± 0.07
0.71 ± 0.01
250
− 20.36 ± 0.09
1.35 ± 0.04
500
− 29.90 ± 0.04
6.31 ± 0.22
1000
− 54.44 ± 0.03
6.51 ± 0.34
From Fig. 3, it is apparent that the heavy metals ions sensing through the application of Calix-based piezogravimetric sensors disclosed its success since the decrease in frequency with increasing the ions amounts is directly related to the mass loading of heavy metals ions on the quartz crystal sensor’s surface (Fig. 3a).
The resorcinarene thin films’ viscoelastic properties were monitored simultaneously with frequency variations, by recording energy dissipation shifts based on oscillation amplitude variations in time (via ring-down method) (Fig. 3b).
Commonly, the higher is the dissipation shift (ΔD > 2.10-6), the softener is the adlayer, and thus the viscoelastic model description deviates from the Sauerbrey model conditions as previously indicated by (Eq. (1)).
3.1.3 Piezogravimetric sensor’s metrological parameters and ionic selectivity
The sensors’ pertinence rest on detection features manifesting in sensitivity (S), linear range (L.R.), detection, and quantification limits (LOD and LOQ). However, excellent sensing characteristics are considered as small LODs and LOQs, high sensitivities, and wide LRs. To evaluate the metrological parameters, the dynamic ranges based on ΔFN variations for ionophore R1 based piezogravimetric sensor are presented in Fig. 4, whereas Table 3 is recapitulating LRs, LODs, LOQs, and sensitivities of the Calix-based chemosensor.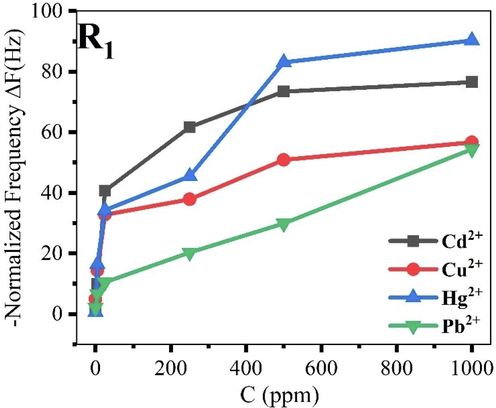
Dynamic ranges for ionophore R1 based on the normalized frequency variation.
HMs
LR
(ppm)S
(Hz/ ppm)LOD
(ppm)LOQ
(ppm)
Cd2+
2–25
1.571
0.61
1.83
Cu2+
0.5–25
1.056
0.01
0.03
Hg2+
3–25
1.215
0.88
2.64
Pb2+
2–1000
0.045
0.47
1.42
The LODs and LOQs were calculated from LOD = 3.3σ/S and LOQ = 10σ/S, where σ is the standard deviation and S is the linear range’s slope or the sensitivity. The developed detection platforms showed respectful sensing characteristics (wide LRs, noble sensitivities, low LODs, and LOQs), based on frequency variations. The obtained LODs in the case of Hg2+, Pb2+, and Cd2+ ions were slightly higher than the recommended thresholds. Nonetheless, in the case of Cu2+, the chemosensor was sensitive since it produced an inferior LOD to limits stated by WHO (2 ppm) and USEPA (1.3 ppm). The selectivity evaluation based on frequency shifts (Fig. 4) indicates that R1 is selective to mercury and cadmium ions.
3.1.4 Hypothetical interpretation of the ionophore adsorption on QC gold surface
Ensuring the best performance of the developed piezogravimetric sensor, resorcinarene detection platforms should be well attached and well-constructed, the ligands affixation on the quartz crystal surface is then ascertained by electrostatic interactions (Mainly VDW -Van der Waals- dispersion forces with a quantum mechanical nature, manifesting in dipoles produced via quantum fluctuations) between heteroatoms, i.e. O, aromatic cycles’ electrons, end carbon chain double bonds, and the gold surface. As appraised by the work of Reimers and coworkers (Reimers et al., 2017), aromatic cycles and carbon chains interact strongly with coinage metals-based surfaces such as gold, resulting in their adsorption via VDW interactions comparable to covalent attachment in terms of strength. For a clarified overview, the free gold atoms can either react with their ‘d’ or ‘s’ orbitals (d10s1 as electronic valence configuration), due to their high reactivity and capability of covalent bonding to carbon atoms forming single, double, and even triple bonds (Tang and Jiang, 2015; Zaba et al., 2014). However, when gold atoms are attached forming a single Au-Au bond via s-s orbitals, their reactivity weakens, which is the case of noble gold surfaces, yet, this fact does not disturb the VDW dispersive forces for those bonds formed via ‘d’ orbitals of gold surface and carbon atoms as an example (Chaudhuri et al., 2009; Reimers et al., 2016). Besides the later interaction, electron flow between O, and gold surface atoms is ensured through a moment dipole polarization creating an electron density stream.
3.2 Electrochemical studies
3.2.1 Electrochemical characterization
Cyclic voltammetry, besides providing exact analysis of possible oxidizing and reducing species existing in the solution, can offer an opportunity to characterize the electrical double-layer, since the current is influenced by charging the capacitive load. The heights of the current peaks show evidence of chemical activity, while the area below the current–voltage curve gives info about the charge transfer capacity of the surface (Arfin, 2021).
The behavior of the R1 based electrochemical sensor was preliminarily examined employing CV. Fig. 5A presents the CV signatures of the bare and modified electrodes in the presence and absence of ions.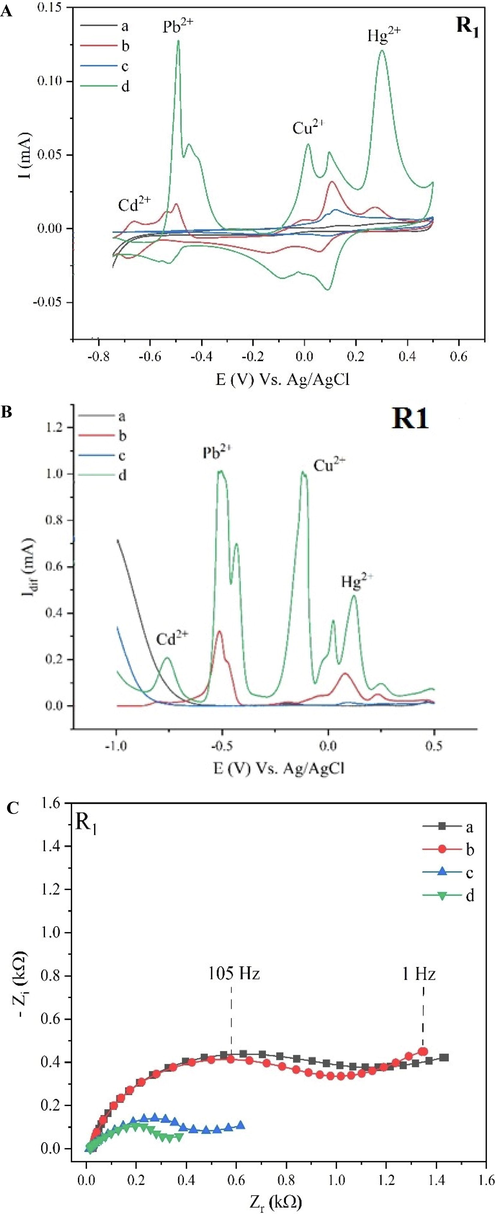
Electrochemical characterization of (a) bare electrode in 0.2 M HCl, (b) bare electrode in the presence of 1 ppm each of heavy metals ions in 0.2 M HCl, (c) modified electrode in 0.2 M HCl, and (d) modified electrode in the presence of 1 ppm each of HMs in 0.2 M HCl through exploring the overlaid CV voltammograms (A), SWV signatures (B), and EIS Nyquist plots (C).
Starting the forward scan at a potential (-0.750 V), the potential increases during the voltammetry curve. The anodic reduction affects the CV curve by increasing current density and showing peaks for Cd, Pb, Cu, and Hg ions at the respected potentials. Due to the diffusion of reduced species at the vicinity of the anode, current density decreases until the inverse potential (+0.500 V) is reached. The back scan starts by decreasing the potential between the electrodes which leads to oxidizing of the reduced species at the cathode having an impact on the current density with cathodic peaks at the respected potential values. Further decrease of the voltage indicates the diffusion limitation until the minus reverse potential is reached. The applied voltage is increased until the starting potential (-0.750 V) is reached again and the potential cycle is finished. Besides the current density value at the peak, the potentials for the anodic and cathodic peaks and their difference are also considered as important parameters of a CV illustration.
As displayed in Fig. 5A, no significant analytical signals (redox peaks) appeared in the absence of ions for the bare (a) and modified electrode (c) in 0.2 M HCl. However, bare gold electrode (b) presented moderate responses when the ions were added to the electrolytic medium, and further modifications of the electrode with ionophore R1 (d) have enhanced the well-defined oxidation current peaks of heavy metals ions on their respective potentials. However, the cathodic reduction peaks of HMs are not well-separated and intense as the anodic ones. The observed increase in the CV current peaks of heavy metals after the electrode modification is owing to the fast electron-transfer rate at the R1@SPE platform and its conductive nature, besides the complexation process between the heavy metals ions in the electrolyte and the impregnated resorcinarenes on the electrodes.
Besides having high sensitivity, SWV can achieve very low detection limits by applying effective discrimination against the charging background current. The SWV signatures of the bare and modified electrodes in the presence and absence of heavy metals were also studied to complement the cyclic voltammetry characterization and are shown in Fig. 5B. The SWV signatures are in agreement with the CV results, where the bare (a) and modified electrode (c) did not represent any anodic signal. After adding the heavy metals to the medium, the appearance of net well-separated peaks of heavy metals ions centered at their pertaining potentials was noticed with enhanced current intensity when coming to the R1@SPE (d) compared to the bare gold electrode (b), meaning that the sensitivity of the electrode has increased notably after its modification, owing to the heavy metals accumulation on its surface, the high adsorption capacity and the large electroactive surface of resorcinarene platform.
EIS is one of the key measurement techniques to study the electrode–electrolyte interfaces.
The most common graphical representation of the EIS data is known as the Nyquist plot. A typical illustration of a Nyquist plot for an idealized faradaic reaction with a probe containing redox couple consists of a semicircle limited by the charge transfer elements (Rct) and double-layer capacitor (CD) shifted on a real axis (Zreal) by the value of the solution resistance (RS).
The value of Rct relates to the radius of the semicircle. The resonance frequency of the parallel combination Rct and CD matches with the highest imaginary impedance part of the curve (Zim).
For higher frequencies, the diffusion effects do not play any significant role, while at low frequencies, the Warburg impedance cannot be neglected. Warburg impedance is related to the diffusion of ions to the oppositely charged electrode. In this case, HM ions in solution diffuse from the bulk fluid to the electrode and as a consequence, increase the Warburg impedance at low frequencies. This hinders the mobility of the slow alternating ions responding to an applied voltage. However, in a high-frequency region, ions have no time to diffuse to a distance before the reversal of an input voltage and thus Warburg impedance does not influence the system response (Dominguez-Benetton et al., 2012).
As mentioned above, Warburg impedance is the dominant parameter for the diffusion-controlled region as the redox couple lowers the charge transfer resistance Rct. The further decrease of the frequency results in a transition from a semicircle to a straight 45° declined line at low frequencies. Adsorption of HMs composite from the analyte of interest to an electrode surface results in the building up of complexes, which are replicated by the changes in material property, i.e., dielectric constant and dependent electrical double-layer capacitor (Dominguez-Benetton et al., 2012).
However, the formation of complexes adsorbing at the electrode/electrolyte interface in the presence of redox couple can be measured by an increase in the Rct value, since the thickness of an insulating layer including the composite and analyte increases or changes its density and thus disturbs a current flow into the electrodes. This is displayed as an extension of the semicircle to a nearly straight line as shown in (Fig. 5C).
As displayed in Fig. 5C, the Nyquist plots displayed a semicircle in the high frequencies associated with an electron transfer limited process, and a further line at low frequencies, indicating a diffusion-controlled process. In the case of bare electrodes (a) and (b), the semicircle diameter equal to the charge transfer resistance (Rct) decreased after the addition of 1 ppm of HMs in 0.2 M HCl. Further decrease in Rct was revealed after modifying the electrodes with R1 (c) and (d). The smallest Rct values were associated with the modified electrodes in the presence of 1 ppm of heavy metals ions in 0.2 M HCl (d). The decrease in Rct values of the modified electrode indicates the electrode surface conductivity improvement from one side and the enhanced electron transfer properties from another. In the present case, we demonstrated in (Fig. 5C) that the charge transfers rate increases even at OCP as a result of the chemical surface modification.
In future contributions, we plan to perform tests where impedance measurements are performed at fixed bias potentials, preferably at potentials corresponding to the maximum current peak of the tested ions. Since a significantly larger Rct difference can be achieved at those potentials due to the influence of the investigated ions. Those measurements will be the basis for the development of alternative sensing methods based on the EIS measurements.
3.2.2 Optimization of the experimental parameters
The optimization of the experimental parameters was achieved to accomplish the best electroanalytical response, and therefore, the best analytical sensitivity. The parameters affecting heavy metals ions sensing were optimized by using SWV.
The supporting electrolyte, pH, accumulation potential, and accumulation time were adjusted to conclude the best conditions for achieving an enhanced detection (Fig. 6).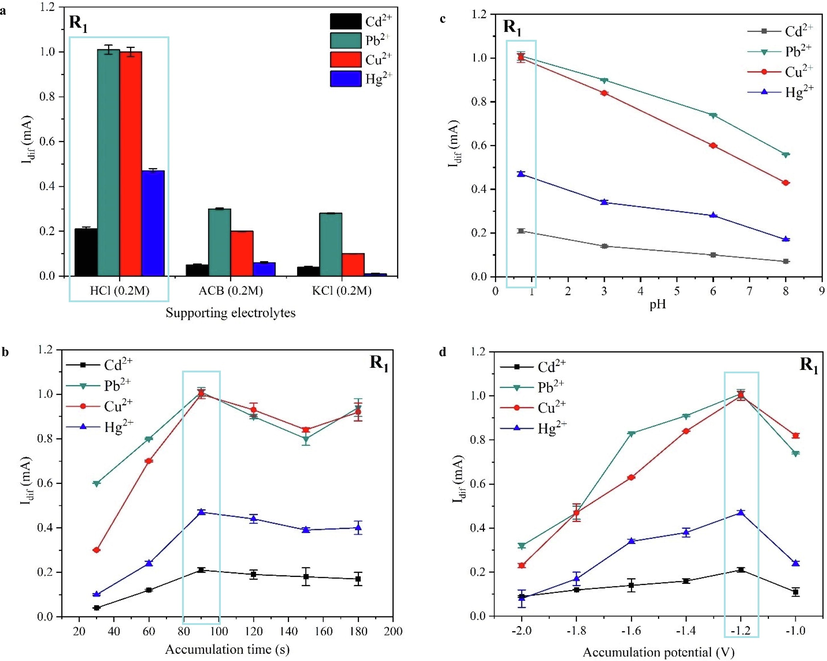
Influence of various supporting electrolytes (a), accumulation time (b), pH (c), and accumulation potential (d) on the electrochemical SWV signals of the modified electrodes in presence of 1 ppm each of HMs.
3.3 Effect of supporting electrolyte
The electrochemical detection of HM by the R1@SPE platform is pH-dependent. The selection of the proper supporting electrolyte and buffer is a significant step in electroanalytical tests because the electrolyte composition and pH influence the properties of the solution as well as the electrode/solution interface, modifying the kinetics and thermodynamics of the charge transfer process, and the adsorption at the electrode surface. Therefore, the pH influence on the electrochemical detection of HM using SWV as a detection technique was investigated.
The electrolytic solution plays a vital role in boosting the medium’s conductivity where the electrochemical detection is taking place, supporting electrolytes viz. 0.2 M HCl (pHi = 0.7), KCl 0.2 M (pHi = 6.8), and ACB 0.2 M (pHi = 5.3) were employed herein for selecting the appropriate one to be utilized in further detection investigations. In Fig. 6a, it was observed that the uppermost SWV current intensities were obtained utilizing 0.2 M HCl as an electrolytic medium, which is explained by its greater conductivity ensuring a fast charge transport process of the HMs and stabilizing them at the modified electrode’s surface. Therefore, 0.2 M HCl was selected as the optimal supporting electrolyte to be used in further detection studies.
3.4 Effect of accumulation time
The effect of accumulation time on the square wave voltammetry signals was studied from 30 to 180 s (Fig. 6b), the current values increased upon the increase of accumulation time up to 90 s, owing to the progressive buildup of HMs on the modified electrode’s surface. After 90 s, the current values dropped gradually due to the surface saturation process, meaning that the equilibrium conditions between the complexed heavy metals and those present in the electrolytic solution were achieved. The 90 s accumulation time was selected as optimal and was employed in further electrochemical examinations.
3.5 Effect of pH
The complexation process of the HMs within the R1@SPE platform is highly affected by the medium’s pH. Hence, the sensing of HMs in HCl 0.2 M (pHi = 0.7) has been examined by adjusting the pH values over a range from 0.7 to 8. The optimal pH of 0.2 M HCl for obtaining the highest SWV currents is 0.7 (Fig. 6c), which is applied in further SWV detection studies. However, while increasing the pH towards alkaline values, a decrease in the peak current values was observed for all HMs, explained by their hydrolysis and the formation of insoluble metal hydroxides on the electrode surface at higher pH values, causing current fluctuations and decrease.
3.6 Effect of accumulation potential
The influence of the accumulation potential on the SWV in the electrochemical determination of HMs was deliberate. The applied potential range was −1V to −2V, (Fig. 6d), the peak currents of various HMs reached their maximum at −1.2 V. Once the potential is shifted towards more negative values, a decrease in the peak currents of HMs is observed. Generally, such a trend is explained by the hydrogen evolution reaction, this later was proved by the presence of bubbles on the electrode’s surface during the measurements. So, an accumulation potential of −1.2 V was employed in further studies as optimal.
3.6.1 Electrochemical determination of HMs
The analytical performance of the proposed electrochemical sensor was examined under the concluded optimal conditions (0.2 M HCl, pH = 0.7), accumulation potential of −1.2 V for an accumulation time of 90 s. Fig. 7a,b present the overlaid square wave voltammograms for the simultaneous electrochemical determination of HMs in the concentration range from 1 to 100 ppb based on the R1@SPE, whereas the corresponding calibration curve is displayed. The peak separation in the SWV signals is large to quantify each metal ion distinctly. On the voltammograms of R1@SPE, some peaks appeared apart from those corresponding to the HMs, owing either to non-complexed analytes trapped on the modified SPE surface (electroactive impurities formerly present in the electrolytic solution) or due to the resorcinarene leaching.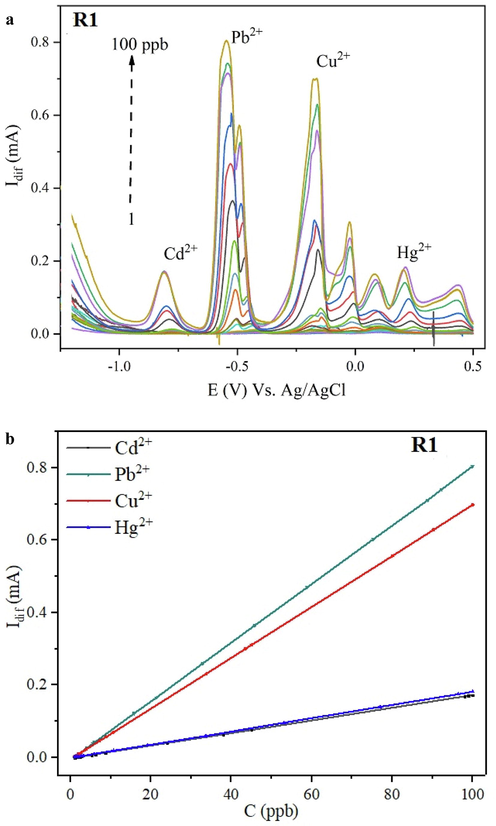
(a) Simultaneous electrochemical determination of the studied HMs in the concentration range from 1 to 100 ppb under optimal conditions based on R1@SPE sensing platform, (b) the insert is presenting the corresponding calibration curve.
Based on the constructed calibration curve (Fig. 7b), a perfect linear relationship between the concentrations of HMs and the responses in SWV peak currents was established (Table 4). The limits of detection (LODs) and limits of quantification (LOQs) were calculated from LOD = 3.3σ/S and LOQ = 10σ/S, respectively. Where σ is the standard deviation of the blank (based on three measurements) and S is the linear range’s slope (the sensor’s sensitivity).
Ions
LR
(ppb)Fitting equation
R2
LOD
(ppb)LOQ
(ppb)
Cd2+
1–100
I = 0.0017*CCd2+ − 0.0016
0.9999
0.37
1.11
Pb2+
1–100
I = 0.0081*CPb2+ − 0.0064
0.9999
0.19
0.57
Cu2+
1–100
I = 0.0070*CCu2+ − 0.0049
0.9999
0.23
0.70
Hg2+
1–100
I = 0.0018*CHg2+ − 0.0006
0.9999
0.41
1.23
The recommended sensor presented wide linear responses and noble sensitivities, the attained LODs and LOQs are much lower than the recommended thresholds stated by the WHO and the USEPA, therefore confirming the ultra-sensitivity of the sensing platform. The sensor has proven to be the most sensitive to lead and copper ions detection, nevertheless cadmium and mercury ions are also well measurable.
3.6.2 Interferences study
The effect of interfering ions on the HMs determination and the sensor’s selectivity was investigated via adding metal ions frequently present in water matrices, i.e. Al3+, K+, Mg2+, Zn2+, and Ni2+. A concentration of 4 ppm (40 folds) of each interfering ion was added to 0.2 M HCl (pH = 0.7) containing 100 ppb of heavy metals; Fig. 8 displays a comparison of the heavy metals peak currents for the developed sensor in the absence (control) and in the presence of interfering ions under the determined optimal conditions.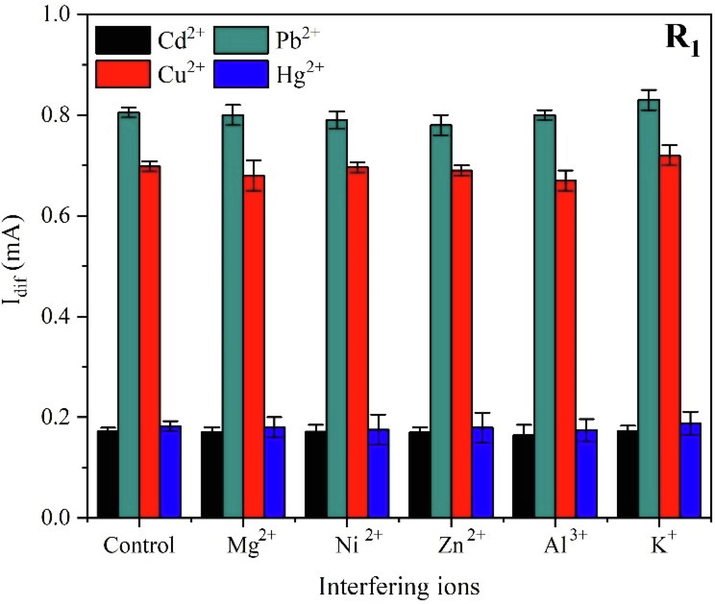
Bar charts representing the heavy metals peak currents for the R1@SPE sensor in the absence (control) and the existence of interfering ions under optimal conditions.
Based on the constructed calibration curves (Fig. 8), a linear relationship between the concentrations of heavy metals ions and the responses in peak currents was established (see correlation coefficients in Table 5). Evaluating Table 5, the calculated signal deviations of 100 ppb of heavy metals ions are less than 5%, so the studied interfering cations did not affect the simultaneous detection of HMs while applying the optimized procedure, which withstands the potential employment of the fabricated sensor for the simultaneous selective determination of heavy metals ions.
Interfering
ionsPeak deviation (%) =
Cd2+
Pb2+
Cu2+
Hg2+
Mg2+
1.17
0.62
2.57
1.10
Ni2+
0.58
1.86
0.30
3.84
Zn2+
1.17
3.10
1.15
1.64
Al3+
4.06
0.62
4.01
− 4.40
K+
− 0.60
− 3.11
− 3.15
− 3.29
3.6.3 Consideration of reproducibility and repeatability
The repeatability, expressed as relative standard deviation (RSD %, n = 3) of the slope of the SWV calibration plot, was determined by measuring the analytical signal of HM ions over the concentration of 100 ppm using the R1@SPE modified platform, three successive scans.
Likewise, the sensor’s reproducibility has been assessed employing 3 tests of R1@SPE modified electrodes to simultaneously detect 100 ppb of heavy metals ions.
The outcomes in terms of residual standard deviation (RSD) are tabulated in Table 6. The RSDs values are less than 5 %, which demonstrates excellent reproducibility and repeatability of the developed sensor platform, permitting its possible real application to determine trace levels of heavy metals ions simultaneously with high analytical selectivity.
% RSD values related to:Heavy metal ions
Cd2+
Pb2+
Cu2+
Hg2+
Reproducibility (3 tests)
4.68
3.93
3.64
3.24
Repeatability (3 tests)
2.19
2.56
1.44
2.18
3.6.4 Assessment of detection and quantification limits of the R1@SPE platform
The sensitivity and quantification limits of Cd2+, Pb2+, Cu2+, and Hg2+ ions disclose significantly good sensitivity of the R1@SPE modified sensor as tabulated in Tables 3 and 6. The calculated sensitivity limits of the selected metal ions displayed a lower value than the sensitivity limits of numerous sensing platforms of modified electrodes described in the literature (Munir et al., 2019; Xiang et al., 2020; Mei C. J. and Ahmad S. A. A., 2021). As shown in Table 7, the sensitivity values of LOD, LOQ, RSD (Repeatability), and LR, of our R1@SPE based sensor, were favorable while the RSD (Reproducibility) values were slightly higher. Furthermore, our R1@SPE based sensor showed good stability, which is due to the insolubility of the modifying material in the water medium for the detection of metal ions. This enhanced stability is further designated by improved reproducibility.
HM
IonsLOD
(ppb)LOQ
(ppb)% RSD (Reproducibility)
% RSD (Repeatability)
LR
(ppb)
R1@SPE sensor platform
Cd2+
0.37
1.11
4.68
2.19
1–100
Pb2+
0.19
0.57
3.93
2.56
1–100
Cu2+
0.23
0.70
3.64
1.44
1–100
Hg2+
0.41
1.23
3.24
2.18
1–100
Cl-DPTU modified GCE modified electrodes (Munir et al., 2019).
Cd2+
6.45
21.5
1.68
3.77
1–5000
Pb2+
11.0
36.8
2.31
3.40
1–5000
Cu2+
7.85
26.2
3.78
3.40
1.1–10000
Hg2+
9.15
30.4
2.05
4.36
1–5000
3.7 Heavy metals ions detection mechanisms
3.7.1 Piezogravimetric detection process
Numerous factors are involved in explaining the piezogravimetric detection mechanism, for instance, the complementarity between the metal ions and the resorcinarene cavity size, besides the molecular structure of ligand (substituents nature and number). Potential complexation interactions between the sensing platform and target ions are mainly of a non-covalent physical nature (VDW, cation-π…etc.). Herein, we enlighten the sensing mechanism in two major phases:
The first phase manifesting in a complexation, or else host–guest interaction between the metals ions (Mn+ = Cd2+, Cu2+, Hg2+, and Pb2+) and the resorcinarene (R1) already attached to the gold surface, either owing to an electron transfer from heteroatoms and nucleophilic elements towards HMs or due to their physical adsorption within the ligands’ cavities, commonly these interactions are of VDW and cation-π origins:
The second stage is the simultaneous mass loading (accumulation) of metals ions on the solution-electrode interface and the piezogravimetric detection of the metal ions buildup on the resonator's surface.
3.8 Electrochemical detection mechanism
The proposed sensing platform achieved low detection limits due to the analytical performance improvement; explained by the high complexation affinity between the heavy metals ions and the hydroxyl groups of the resorcinarenes, affording better conditions for a host–guest reaction, the suggested electrochemical mechanism can be clarified in three steps:
Accumulation: This step consists to accumulate the target metallic cations (2+) on the electrode surface by adsorption, hence, it is a primordial condition that ensures good sensitivity and a higher detection afterward, the physical adsorption of charged heavy metals ions on the modified electrode’s surface (SPE), is mainly performed via electrostatic attractions:
Pre-concentration: At a higher negative potential compared to that of the HM2+/HM couples, the adsorbed heavy metals ions are electrodeposited on the modified SPE through a cathodic reduction from a valence state of (2+) to (0) to enhance the mass transfer rate, permitting the HMs to be deposited at the SPE surface:
Stripping: The ions already electrodeposited on the electrode’s surface are oxidized to ensure their dissolution back to the electrolyte, thus, yielding an analytic signal. Contrary to the preconcentration step, a positive scan has taken place to kinetically promote the separation and the consistent detection of the metallic ions, which led to well-defined voltammograms peaks with higher current intensities, in other words, the electrodeposited HMs are turned back to the electrolytic solution through anodic oxidation, translated by an SWV analytic signal. In this step, a positive scan took place for the consistent determination of the four heavy metals ions:
4 Conclusions
The ionophore (C-dec-9-enylcalix[4]resorcinarene) was synthesized and characterized and utilized as the recognition element for the R1@SPE platform applying electrochemical (CV and SWV), and piezogravimetrical (QCM-I) methods.
The developed R1@SPE sensor displayed excellent sensing behavior towards heavy metals cations (Cd2+, Hg2+, Cu2+, and Pb2+) with good sensitivity, reproducibility, and repeatability, due to the strengths of the applied electrochemical and piezogravimetrical methodologies which allowed the development of miniaturized platform sensors.
Decent metrological characteristics were gained from QCM-I frequency shifts outcomes, especially in the case of copper ions getting a LOD of 10 ppb, moreover, the sensor showed cadmium and mercury ionic selectivity. Additionally, the voltammetric R1@SPE sensor could simultaneously detect the heavy metals ions under the optimized experimental conditions, and the lowest LOD was associated with Pb2+ (0.19 ppb).
The presence of common metal ions (Al3+, K+, Mg2+, Zn2+, and Ni2+) did not interfere with the simultaneous detection of Pb2+, Cd2+, Cu2+, and Hg2+ within reasonable tolerance and offered suitable repeatability and reproducibility.
The obtained results in this work are strengthening the possibility of using QCM-I and voltammetric-based resorcinarene platforms in real-life detection and monitoring of heavy metals ions in water media.
Acknowledgements
The support from the BIONANO_GINOP-2.3.2-15-216-00017 project, as well as the Stipendium Hungaricum Scholarship Program, is appreciated. Many thanks to MicroVacuum Ltd for loaning the QCM-I apparatus.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Highly Selective Detection of Titanium (III) in Industrial Waste Water Samples Using Meso-octamethylcalix[4]pyrrole-Doped PVC Membrane Ion-Selective Electrode. Electrochim. Acta. 2015;178:580-589.
- [CrossRef] [Google Scholar]
- Polyvinyl chloride-based 18-crown-6, dibenzo18-crown-6 and Calix-[6]-arene zinc(II)-potentiometric sensors. Arab. J. Chem.. 2016;9:S878-S888.
- [CrossRef] [Google Scholar]
- Recent Trends in Macro-, Micro-, and Nanomaterial-Based Tools and Strategies for Heavy-Metal Detection. Chem. Rev.. 2011;111:3433-3458.
- [CrossRef] [Google Scholar]
- Arfin T., 2021. 9-Emerging trends in lab-on-a-chip for biosensing applications, Editor(s): Chaudhery Mustansar Hussain, Sudheesh K. Shukla, Girish M. Joshi, In Micro and Nano Technologies, Functionalized Nanomaterials Based Devices for Environmental Applications, Elsevier,199-218,https://doi.org/10.1016/B978-0-12-822245-4.00008-8.
- Calixarenes as sensor materials for recognition and separation of metal ions. Arkivoc. 2007;2:172-200.
- [Google Scholar]
- Molecular recognition. 5. Molecular recognition of sugars via hydrogen-bonding interaction with a synthetic polyhydroxy macrocycle. J. Am. Chem. Soc.. 1989;111:5397-5404.
- [CrossRef] [Google Scholar]
- Dipyridine-Containing Macrocyclic Polyamine – Nafion-Modified Screen-Printed Carbon Electrode for Voltammetric Detection of Lead. Electroanalysis. 2012;24:591-599.
- [CrossRef] [Google Scholar]
- Combined QCM-D and EIS study of supported lipid bilayer formation and interaction with pore-forming peptides. The Analyst. 2010;135:343.
- [CrossRef] [Google Scholar]
- Heavy metal pollution in the environment and their toxicological effects on humans. Heliyon. 2020;6(9):e04691
- [CrossRef] [Google Scholar]
- Detection of heavy metal ions in aqueous solution by P(MBTVBC-co-VIM)-coated QCM sensor. Sens. Actuators B Chem.. 2011;157:34-41.
- [CrossRef] [Google Scholar]
- The structure of the Au(111)/methylthiolate interface: New insights from near-edge x-ray absorption spectroscopy and x-ray standing waves. J. Chem. Phys.. 2009;130:124708
- [CrossRef] [Google Scholar]
- Novel bis-calix[4] arene-based molecular probe for ferric iron through colorimetric, ratiometric, and fluorescence enhancement response. Tetrahedron Lett.. 2015;56:793-796.
- [CrossRef] [Google Scholar]
- Oligonucleotide-functionalized gold nanoparticles-enhanced QCM-D sensor for mercury(II) ions with high sensitivity and tunable dynamic range. Analyst. 2011;136:2572-2577.
- [CrossRef] [Google Scholar]
- Composites Based on Conducting Polymers and Carbon Nanomaterials for Heavy Metal Ion Sensing. Crit. Rev. Anal. Chem.. 2018;48:293-304.
- [Google Scholar]
- The accurate use of impedance analysis for the study of microbial electrochemical systems. Chem. Soc. Rev.. 2012;41:7228-7246.
- [CrossRef] [Google Scholar]
- Electrochemical impedance detection of Hg2+, Ni2+ and Eu3+ ions by a new azo-calix[4]arene membrane. J. Electroanal. Chem.. 2011;661:31-38.
- [CrossRef] [Google Scholar]
- Studies of aluminum (III) ion-selective optical sensor based on a chromogenic calix [4] arene derivative. Spectrochim. Acta. A. Mol. Biomol. Spectrosc.. 2013;115:269-274.
- [Google Scholar]
- Eddaif, Larbi, Shaban, A., Szendro, I., 2019a. Calix[4]Resorcinarene Macrocycles Interactions with Cd 2+, Hg 2+, Pb 2+, and Cu2+ Cations: a QCM‐I and Langmuir Ultra‐thin Monolayers Study. Electroanalysis elan.201900651. https://doi.org/10.1002/elan.201900651.
- Calix[4]resorcinarene macrocycles: Synthesis, thermal behavior, and crystalline characterization. J. Therm. Anal. Calorim. 2019
- [CrossRef] [Google Scholar]
- A Piezogravimetric Sensor Platform for Sensitive Detection of Lead (II) Ions in Water Based on Calix[4]resorcinarene Macrocycles: Synthesis, Characterization, and Detection. Arab. J. Chem.. 2020;13:4448-4461.
- [CrossRef] [Google Scholar]
- Recent trends of copper detection in water samples. Bull Natl Res Cent. 2021;45:218.
- [CrossRef] [Google Scholar]
- Molecular Imprinting Technology in Quartz Crystal Microbalance (QCM) Sensors. Sensors (Basel, Switzerland). 2017;17(3):454.
- [CrossRef] [Google Scholar]
- A highly selective fluorescent sensor based on calix[4]arene appended benzothiazole units for Cu2+, S2− and HSO4− ions in an aqueous solution. Sens. Actuators B Chem.. 2016;228:109-116.
- [CrossRef] [Google Scholar]
- Trends in Polymers Functionalized Nanostructures for Analysis of Environmental Pollutants. Trends Environ. Anal. Chem.. 2020;e00084
- [CrossRef] [Google Scholar]
- A novel electrochemical sensor based on calixarene functionalized reduced graphene oxide: Application to the simultaneous determination of Fe(III), Cd(II), and Pb(II) ions. J. Colloid Interface Sci.. 2017;508:525-531.
- [CrossRef] [Google Scholar]
- Heavy metals determination in water and food samples after preconcentration by a new nanoporous adsorbent. Food Chem.. 2013;141(3):1916-1922.
- [Google Scholar]
- A review on the determination heavy metals ions using calixarene-based electrochemical sensors. Arab. J. Chem.. 2021;14:103303
- [CrossRef] [Google Scholar]
- Determination by AAS of some trace heavy metal ions in some natural and biological samples after their preconcentration using newly chemically modified chloromethylated polystyrene-PAN ion-exchanger. Anal. Sci.. 2000;16:493-500.
- [Google Scholar]
- Laghrib F., Saqrane S. , Lahrich S., El Mhammedi M. A. 2021. Best of advanced remediation process: treatment of heavy metals in water using phosphate materials, International Journal of Environmental Analytical Chemistry, 101:9, 1192 1208, DOI: 10.1080/03067319.2019.1678603.
- Li, M., Gou, H., Al-Ogaidi, I., Wu, N., 2013. Nanostructured sensors for detection of heavy metals: a review. ACS Sustainable Chem. Eng. 2013, 1, 7, 713–723. https://doi.org/10.1021/sc400019a.
- Human health and ecological toxicity potentials due to heavy metal content in waste electronic devices with flat panel displays. J. Hazard. Mater.. 2010;177:251-259.
- [CrossRef] [Google Scholar]
- Multivalent calix[4]arene-based fluorescent sensor for detecting silver ions in aqueous media and physiological environment. Biosens. Bioelectron.. 2017;90:290-297.
- [CrossRef] [Google Scholar]
- Applications of macrocyclic compounds for electrochemical sensors to improve selectivity and sensitivity. J. Incl. Phenom. Macrocycl. Chem.. 2019;95:171-198.
- [CrossRef] [Google Scholar]
- Mahajan B., 2021. Drinking Water Quality Standards (WHO Guidelines), online version, https://civiconcepts.com/blog/drinking-water-quality-standards.
- A review on the determination heavy metals ions using calixarene-based electrochemical sensors. Arabian J. Chem.. 2021;14(9):103303
- [CrossRef] [Google Scholar]
- Analytical applications of calixarenes from 2005 up-to-date. J. Incl. Phenom. Macrocycl. Chem.. 2011;69:1-55.
- [Google Scholar]
- Selective and simultaneous detection of Zn2+, Cd2+, Pb2+, Cu2+, Hg2+ and Sr2+ using surfactant modified electrochemical sensors. Electrochim. Acta. 2019;323:134592
- [CrossRef] [Google Scholar]
- Rapid and highly selective electrochemical sensor based on ZnS/Au-decorated f-multi-walled carbon nanotube nanocomposites produced via pulsed laser technique for detection of toxic nitro compounds. J. Hazard. Mater.. 2021;418:126269
- [CrossRef] [Google Scholar]
- Nur Abdul Aziz, S.F., Zawawi, R., Alang Ahmad, S.A., 2018. An Electrochemical Sensing Platform for the Detection of Lead Ions Based on Dicarboxyl-Calix[4]arene. Electroanalysis 30, 533–542. https://doi.org/10.1002/elan.201700736.
- A selective distance-based paper analytical device for copper(II) determination using a porphyrin derivative. Talanta. 2017;174:493-499.
- [CrossRef] [Google Scholar]
- Interactions of Native Cyclodextrins with Metal Ions and Inorganic Nanoparticles: Fertile Landscape for Chemistry and Materials Science. Chem. Rev.. 2017;117:13461-13501.
- [CrossRef] [Google Scholar]
- Front Chem.. 2014;30;2:19:2014. eCollection.PMID: 24818124
- [CrossRef]
- Gold surfaces and nanoparticles are protected by Au(0)–thiyl species and are destroyed when Au(I)–thiolates form. Proc. Natl. Acad. Sci.. 2016;113:E1424-E1433.
- [CrossRef] [Google Scholar]
- Competition of van der Waals and chemical forces on gold–sulfur surfaces and nanoparticles. Nat. Rev. Chem.. 2017;1:0017.
- [CrossRef] [Google Scholar]
- Polymer-grafted QCM chemical sensor and application to heavy metal ions real-time detection. Sens. Actuators B Chem.. 2011;155:538-544.
- [CrossRef] [Google Scholar]
- Verwendung von Schwingquarzen zur Wugung dunner Schichten und zur Mikrowugung. Z. Phys.. 1959;155:206-222.
- [CrossRef] [Google Scholar]
- Comparative study of a sensing platform via functionalized Calix[4]resorcinarene ionophores on QCM resonator as sensing materials for detection of heavy metal ions in aqueous environments. Electroanalysis elan.202060331. 2020
- [CrossRef] [Google Scholar]
- Organochlorines and heavy metals in wild-caught food as a potential human health risk to the indigenous Māori population of South Canterbury. New Zealand. Sci. Total Environ.. 2011;409:2029-2039.
- [CrossRef] [Google Scholar]
- Taghvaei-Ganjali S., Zadmard R., Zeyaei M., Rahnama K., Faridbod F., Ganjali M-R., 2009. Synthesis of a New Calix[4]Arene and Its Application in Construction of a Highly Selective Silver Ion-Selective Membrane Electrode, Organic Chemistry International, vol. 2009, Article ID 601089. https://doi.org/10.1155/2009/601089.
- Insights into the PhC≡C/Au Interface. J. Phys. Chem. C. 2015;119:10804-10810.
- [CrossRef] [Google Scholar]
- Application of advanced materials in sonophotocatalytic processes for the remediation of environmental pollutants. J. Hazard. Mater.. 2021;412:125245
- [CrossRef] [Google Scholar]
- US Environemtal Protection Agency. 2022. https://www.epa.gov/ground-water-and-drinking-water/table-regulated-drinking-water-contaminants, (Accessed 1 Feb. 2022).
- Potential resource and toxicity impacts from metals in waste electronic devices. Integr Environ Assess Manag. 2016;12:364-370.
- [CrossRef] [Google Scholar]
- Environmental remediation of heavy metal ions by novel-nanomaterials: A review. Environ. Pollut.. 2019;246:608-620.
- [CrossRef] [Google Scholar]
- Xiang H., Cai Q., Li Y., Zhang Z., Cao L., Li K., Yang H., 2020. Sensors Applied for the Detection of Pesticides and Heavy Metals in Freshwaters, J. Sens. 2020, article ID:8503491, 22. https://doi.org/10.1155/2020/8503491.
- Novel Amino-pillar[5]arene as a Fluorescent Probe for Highly Selective Detection of Au3+ Ions. ACS Omega. 2019;4:17903-17909.
- [CrossRef] [Google Scholar]
- Integrated technique of pulsed laser irradiation and sonochemical processes for the production of highly surface-active NiPd spheres. Chem. Eng. J.. 2021;411:128486
- [CrossRef] [Google Scholar]
- Formation of Highly Ordered Self-Assembled Monolayers of Alkynes on Au(111) Substrate. J. Am. Chem. Soc.. 2014;136:11918-11921.
- [CrossRef] [Google Scholar]







