Translate this page into:
Larvicidal activity of plant extracts from Colombian North Coast against Aedes aegypti L. mosquito larvae
⁎Corresponding author at: Pharmacy Department, Faculty of the Pharmaceutical Sciences, Campus Zaragocilla, University of Cartagena, Colombia. fdiazc1@unicartagena.edu.co (Fredyc Díaz Castillo)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
The Aedes aegypti L. mosquito is considered the most important vector of arboviruses in the world. Insecticide-resistant phenomenon is a difficult barrier to overcome for government health entities around the planet. This problem forces to increase the concentrations of insecticides in the environment causing environmental pollution and health threat to human beings. Plants have been used to combat pests for centuries and are an eco-friendly source for the search for molecules with larvicidal activity. In this work, 65 ethanol-soluble extracts from 56 plants of the Colombian Caribbean region were evaluated as potential larvicides against Aedes aegypti mosquito, as well as, for their toxic effects on non-target organism. A high larvicidal activity was found for 16 ethanol extracts, however, the most potent activity against the larvae was obtained for the five plant extracts corresponding to the seeds of Annona squamosa L., Annona cherimolia L., Annona muricata L., Tabernaemontana cymosa Jacq. and Mammea americana L., with LC50 value of 58.44, 65.10, 84.92, 25.02 and 38.58 ppm, respectively. T. cymosa seeds extract was selected for bio-guided fractionation due to the high larvicidal activity showed. Through chromatographic techniques and Nuclear Magnetic Resonance (NMR), five indole alkaloids were isolated and characterized from T. cymosa active fraction; among them, voacangine showed activity with a LC50 of 5.13 ppm indicating a high larvicidal potency besides low toxicity against model organism Caenorhabditis elegans. We also report a new indole alkaloid from T. cymosa. Our study demonstrated the potential of the Colombian Caribbean flora as a host of bioactive plants against important vectors such as the A. aegypti mosquito with potential use in controlled environments.
Keywords
Indole alkaloids
Dengue
Arboviruses
Bio-guided fractionation
Colombian plant extracts
1 Introduction
Mosquito-borne diseases are among the most dangerous illnesses worldwide, given their reemergence and rapid spread. The Aedes aegypti L. mosquito is considered the main vector of many diseases; although it can transmit zika and chikungunya viruses, the vast majority of deaths are consequences of dengue and yellow fever, mainly in children (Saavedra et al., 2018; Jones and Thomas, 2019). In addition, the Aedes aegypti mosquito is also capable of transmitting other important diseases such as Mayaro, West Nile and Rift Valley fevers (Pereira et al., 2008; Mweya et al., 2013; Uraki et al., 2019). Currently, there is an increasing risk of outbreaks worldwide due to changing climatic conditions, poverty, lack of vaccines, mosquitoes anthropophilic diet, simultaneous circulation and reappearance of arboviral diseases (Leta et al., 2018; Shearer et al., 2018; Tjaden et al., 2018; Anoopkumar and Aneesh, 2022; Iwamura et al., 2020;). These situations are more notorious in Africa and South America jungle areas, causing severe public health problems that affect the well-being of vulnerable populations and economic activities, including tourism (Lindholm et al., 2017; Lima et al., 2019). Strategies for the control of arthropod-borne diseases include vector-directed control methods; spraying synthetic insecticides is the most common strategy due to the lack of vaccines or patient-directed treatments (Roiz et al., 2018).
A vaccine for the dengue virus called Dengvaxia has been approved for some countries around the globe. However, clinical tests have shown low effectiveness against some dengue serotypes and the appearance of health problems in infected people after vaccination (Fatima and Syed, 2018). In addition, the absence of specific antiviral treatments against fever diseases viruses and the resistance that mosquitoes are developing against synthetic molecules hinder the control of the disease outbreaks. Furthermore, Aedes aegypti mosquitoes adapt rapidly to changing climatic conditions, thus increasing the geographical distribution of mosquito populations to non-tropical regions (Azeem et al., 2019; Khan and Akram, 2019). Consequently, the transmission rates of arbovirosis have increased dramatically in the last decade, causing 50 to 100 million dengue cases annually. On the other hand, endemic cases of the Chikungunya and Zika viruses have been reported in 44 and 87 countries, respectively, affecting more than two million people in the Americas and with the latent concern about the devastating damage of the Zika virus in unborn children due to its congenital effects and association with Guillem Barré syndrome (Mordecai et al., 2017; Joyce et al., 2018; Lopes Martins et al., 2021). Despite government efforts to control and mitigate dengue transmission, according to the Colombian National Institute of Health, 46.366 cases of dengue have been recorded in Colombia until December of 2021, of which 6.341 have occurred in Cartagena, consolidating itself as the city with the highest incidence of this arbovirus (SIVIGILA, 2021).
The situation described above indicates that using organophosphorus and pyrethroids compounds of synthetic origin as adulticides is not producing the desired effect on Aedes aegypti mosquito populations, probably as a result of the acquired resistance phenomenon (Demok et al., 2019; Smith et al., 2019). Increases in larvae and adult resistance to insecticides impose government control entities to raise concentrations for fumigation processes, causing insecticide residues to reach ecosystems and human communities, affecting people and other organisms (Rahman et al., 2020; Zhang et al., 2020). Therefore, new insecticide molecules with less harmful effects against non-target organisms are needed, mainly due to the constant reports of insecticide resistance; this added to the fact that molecules with new mechanisms of action have not been discovered in the last three decades (Hill et al., 2018).
Plant extracts and their compounds have emerged as viable alternatives for disease vector control due to their insecticidal properties, biodegradability, and economy (Pratheeba et al., 2019). Essential oils derived from plants are also a source of study for their repellent activity against mosquitos. This property has been widely studied to control mosquito bites and prevent the spread of arbovirosis (Sritabutra et al., 2011; Alsarar et al., 2021). These natural products have shown terpenes and iridoids as their main components, which have larvicidal, adulticidal, pupicidal, repellent, and oviposition deterrence activities in mosquitoes (Castillo et al., 2017; Chellappandian et al., 2018).
In the new century, many researchers have focused their efforts on discovering compounds from plants with larvicidal activity against Aedes aegypti due to their several modes of action and biodegradability properties. As a result, more than 130 plants with Lethal Concentrations 50 (LC50) below 100 ppm have been reported, including their phytoconstituents and essential oils (Piplani et al., 2019). Furthermore, the groups of secondary metabolites responsible for the insecticidal properties of some plants have been identified, such as alkaloids, anthraquinones, coumarins, flavonoids, acetogenins, terpenes, and sterols; additionally, Asteraceae, Fabaceae, and Lamiaceae botanical families contain the higher number of larvicidal molecules reported (Pavela et al., 2019).
The aim of the present work was to evaluate the larvicidal activity of 56 plant species from the Colombian Caribbean Region in order to search for new natural alternatives to synthetic insecticides for vector control. Through extract mortality assessment and biodirected isolation using Aedes aegypti larvae as a model, it was possible to identify an indole alkaloid from Tabernaemontana cymosa as a potencial larvicide. Finally, Selectivity Index (SI) for each active extract was obtained as well as for the isolated compound using the fish Poecilia reticulata and Caenorhabditis elegans, respectively. Our findings suggest that Colombian plant extracts and their derivated compound represents a suitable, environmentally friendly and safe alternative for Aedes aegypti larvae field control.
2 Materials and methods
2.1 Mosquitoes rearing
The Aedes aegypti L. eggs were supplied by the Entomological Unit of Health Department Secretary of Atlántico, in Barranquilla, Colombia. The ovitrap with eggs was placed in a tray with dechlorinated water and food; additionally, yeast was added to improve hatching. After two days, the first instar larvae were transferred to a new tray with clean water using plastic pipettes. The larvae trays were maintained at 26 ± 2 °C with a relative humidity of 65 % and 14:10 h light and dark cycles.
Aedes aegypti larvae were fed with commercial dog biscuits until their metamorphosis to pupae stage. Then, they were transferred to an incubation vessel with clean water for their maturation to adult mosquitoes. Next, matured mosquitos were released into the cage. Adult females were fed with a Wistar rat for bloodmeals and males were fed with 10 % sucrose solution. Ovitraps were disposed in the cage one day after the bloodmeal to collect the eggs.
2.2 Preparation of plant ethanol extracts
The plant material (leaves, stems, and seeds) from 56 plant species was collected in the Bolivar department on the Colombian north coast (municipalities of Arjona and Turbaco). Immediately after collection, a sample of the plant material was preserved by spraying with ethanol (95 %), and a voucher was made to ensure the correct identification of the species. The plant identification was carried out by the staff of the Guillermo Piñeres Botanical Garden (JBGP) in Cartagena (Bolívar-Colombia) and the Medellin Botanical Garden in Medellin (Antioquia-Colombia).
The rest of the plant material was taken to the LIFFUC facilities at the University of Cartagena for processing. Each plant’s separated organ (leaves, stems, and seeds) were washed, dried and and ground to powder. Subsequently, 100 g of plant material were weighed and macerated with 95 % ethanol at room temperature (25–27 °C) for three days. Next, the liquid phase was separated by filtration and evaporated under reduced pressure in a rotary vacuum evaporator (Heidolph®- Hei: VAP Advantage) using a temperature range of 40–45 °C. The process was repeated until the depletion of the compounds extracted from the plant material.
2.3 Larvicidal activity of plant extracts against Aedes aegypti
A total of 65 extracts were tested against Aedes aegypti larvae; for this purpose, the WHO protocol was applied with modifications as described in previous studies (Rodríguez-Cavallo et al., 2019; World Health Organization, 2005). First, 20 late-third and early-fourth instar larvae were selected and gently put into a cup containing 99 mL of dechlorinated water, according to the criteria established in the laboratory. Second, each extract's 20.000 ppm stock solution was prepared using Dimethyl sulfoxide (DMSO). Third, one milliliter of the stock solution was added to the cup to obtain a final concentration of 200 ppm in a 100 mL volume; mortality was recorded at 1, 6, 12, 24, 36, and 48 h after extract inoculation. Each test was performed in triplicate; DMSO (1 %) and Temephos (0.05 ppm) were used as negative and positive controls. The concentrations established for bioassays were determined after several preliminary tests, in which the presence of emulsions or high turbidity in the water was observed at extract concentrations above 200 ppm. This situation could cause behavioral changes in the larvae, such as the loss of their characteristic movements, increasing the residence time on the surface of the water, and even death due to mechanical asphyxia, interfering with mortality readings.
Larvae were considered dead when they did not react to physical stimuli like being touched with a blunt pointer in the cervical region or lack of movement when they were exposed to light. The assay was considered invalid when pupation or mortality exceeded 10 % in negative control. The percent of mortality was calculated using the following formula:
In cases where pupae were present, mortality correction was performed according to the Abbott’s formula (Martínez Rodríguez et al., 2022). Thus, the total number of dead larvae in each treatment was calculated as follows:
2.4 Determination of Lethal concentration 50 (LC50) value of the ethanol extracts
Lethal Concentration 50 (LC50) values were calculated for extracts with larvicidal activity above 80 % at 200 ppm. For this, five different concentrations of each extract were evaluated according to an adaptation of protocols previously established (World Health Organization, 2005; Bezerra França et al., 2021). Briefly, serial dilutions were prepared to obtain final concentrations of 5, 10, 50, 100, and 200 ppm from a 20.000 ppm stock solution. Then, 20 late-third and early-fourth instar larvae were placed in a cup with 99 mL of dechlorinated tap water, and 1 mL of stock solution was added to each cup to obtain the treatment concentration (5, 10, 50, 100, and 200 ppm). Mortality was recorded at 24 h after exposure to the extracts. Controls and mortality criteria were used as previously mentioned. Assays were performed by triplecates.
2.5 Bioassay-guided fractionation and isolation
The extracts were selected for active principle seek according to their larvicidal potency and biological activity against the non-target organism. Bio-directed fractionation was carried out by serial Open Column Chromatography (OPC) using Silica Gel (Merck ®, 70–230 Mesh; 60–200 Mesh) as a stationary phase with a 1:20 extract:SiO2 proportion. First, in order to reduce the complexity, 25 g of the total extract were fractionated using a quick elution gradient with hexane, chloroform, ethyl acetate and methanol. The following columns were eluted with mixtures of the organic solvents mentioned above to gradually increase the mobile phase polarity (Hexane 100 % to Methanol 100 %). A fraction was considered as active if its larvicidal activiy is greater than 80 % at 50 ppm.
The eluted fractions were monitored by Thin Layer Chromatography (TLC) with visualization under UV light (254 and 365 nm) and by dipping the plates into a solution of 10 % (v/v) H2SO4 in ethanol for phytochemical analysis. Those fractions with a similar elution pattern were pooled to continue with the biological assays against A. aegypti larvae and their subsequent fractionation. Preparative TLC was performed on Kieselgel 60 F254 plates (Silica gel, 1.0 mm layer thickness, Merck®) when needed in order to isolate compounds from non-complex fractions; afterward, final purification was carried out by High Performance Liquid Chromatography (HPLC) applying normal or reverse phase when necessary (Narayanan et al., 2021).
2.6 Compound elucidation
Standard analytical methods were used for the structural identification of active compounds. These methods included measurements of melting points, UV, and mainly 1H and 13C Nuclear Magnetic Resonance spectroscopy. A Bruker AMX 300 instrument was used to determine the NMR spectra, operating at 300 MHz and 75 MHz for 1H NMR and 13C NMR, respectively. Chemical shifts (δ) are expressed in parts per million (ppm), and the TMS peak is taken as the reference; coupling constants (J) are given in Hertz (Hz) (Botero et al., 2021; Echeverri et al., 2015). Compound chemical structures were confirmed with subsequent 1D and 2D NMR techniques, such as APT, DEPT, COSY, HMQC, and HMBC, then high-resolution mass spectrometry (HR-MS) experiments were developed under needs(Pájaro-González et al., 2022c). The structural elucidation of the compounds was carried out in collaboration with the Organic Chemistry of Natural Products Research Group of the University of Antioquia (Medellin, Colombia).
2.7 Ichthyotoxicity assays
An important characteristic of natural insecticides is their selectivity by calculating the Toxic Concentration 50 (TC50) against a reference organism. For this reason, the Ichthyotoxic activity of the extracts was tested on Poecillia reticulata, commonly known as Guppy fish, to obtain the Selectivity Index (SI) of each extract. The P. reticulata minnows were supplied by the Medical Entomology Unit of the Bolívar Public Health Laboratory. Fish were transferred to the LIFFUC laboratory facilities, and those showing mechanical damage symptoms during transport were discarded. In order to acclimatize and condition the fish, the adults were placed for approximately-two weeks, prior to the toxicological tests, in glass aquariums with standing tap water. Then, the fish were introduced into the aquarium containing aquatic plant species of Elodea and fed weekly with flake-fish commercial food. To avoid stress, the temperature was maintained at 26 °C and the average pH was 7.6; these parameters were regularly measured during the bioassay time. Briefly, ichthyotoxic activity against P. reticulata was carried out using 15 individuals of 3.5 ± 0.2 cm, randomly selected and separated into three different groups of 5 fishes. These fish were kept without food for 24 h prior to testing. Each group was exposed to 200 ppm of each ethanol extract, and mortality was recorded at 0.5, 1, 6, 12, 24, and 30 h. The experiment was performed in duplicate to obtain the average mortality, while the SI was obtained by applying the following formula:
For the TC50 evaluation, fishes were exposed to five different concentrations (1, 5, 10, 25, 50, and 100 ppm) of plant extracts. Considering that the extracts tested were very active against mosquito larvae during the larvicidal assay, the first mortality data collection was recorded at 10 min of exposure, then after 0.5, 1, 6, 12, and 24 h after extract inoculation.
2.8 In vivo experiments on Caenorhabditis elegans
To determine the toxicity of isolated compounds against the model organism Caenorhabditis elegans a dose–response experiment was carried out. Briefly, batches of 10 ± 2 young adult worms (L4 instar) were transferred to 24-well microplates. Each group were treated with different concentrations of the compound (25, 50,100 and 200 ppm) using DMSO as the solvent, on the other hand, a control group was set up with K medium containing 1 % of DMSO (Duran-Izquierdo et al., 2022). After 24 h the number of living and dead organisms in each well was recorded for mortality assessment. Worms that did not present movement within 30 s were considered as dead. Three replicates were established for each treatment, and the tests were performed in triplicate on different days (Olivero-Verbel et al., 2021). The mortality percentage was calculated based on the following formula:
Were DW: Number of Dead worms; AW: Number of Alive worms.
2.9 Statistical analysis
The data collected were subjected to the normality tests of Shapiro-Wilk and Kolmogorov-Smirnov. Probit analysis was carried out to obtain LC50 and TC50 values for larvicidal and ichthyotoxicity activity. For C. elegans assays, the homogeneity of variances was validated through the Barlett test. The means between the different treatments and control were compared using an ANOVA test. When normality was not achieved, the data was processed with Kruskal-Wallis test. After ANOVA, we applied Turkey post-test to compare each treatment with the control. Data are presented as the mean ± standard error. The data were processed with GraphPad Prism Software with confidence limits at 95 % (Hari and Mathew, 2018).
3 Results
3.1 Preparation of plant ethanol extracts
65 ethanol extracts were obtained from 56 plant species collected on the Colombian North Coast. The scientific name, the common name given by the native population, the voucher number and the family to which each of the evaluated plant species belongs are shown in Table S1.
3.2 Larvicidal activity of plant extracts against Aedes aegypti
After 48 h of exposure at 200 ppm, the extracts were classified according to its potency as follows:: a) Null: 0–10 % of larval mortality; b) Low: 11–49 % of larval mortality; c) Moderate: 50–80 % of larval mortality and d) High: 81–100 % of larval mortality. The vast majority of the tested extracts (49) presented null and low larvicidal activity and 16 plant extracts showed high larvicidal activity. Among this last group, the seed extracts of Annona cherimolia, Annona muricata, Annona squamosa, Carica papaya, Tabernaemontana cymosa and Mammea americana showed a 100 % of larvicidal effect at 48 h of exposition (Table S2). The seed extracts of Tabernaemontana cymosa and Mammea americana showed mortality from the first hour of testing, which indicates the high efficacy of these extracts.
3.3 Determination of Lethal concentration 50 (LC50) value of ethanol extracts
The LC50 values of the five most active extracts, corresponding to the following plant species, Tabernaemontana cymosa, Mammea americana, Annona muricata, Annona squamosa, and Annona cherimolia were obtained (Table 1). The ethanol extract of Tabernaemontana cymosa showed the highest toxicity with a LC50 of 25.02 ppm, which would indicate the presence active metabolites with larvicidal activity against A. aegypti. On the other hand, the ethanol extract of Mammea americana exhibited an LC50 of 38.58 ppm.
Extract
LC50 (LCL – UCL)*
LC90 (LCL – UCL)*
Slope ± SD
R2
Annona squamosa
58.44 (24.07 – 141.90)
1449.8 (597.1 – 3520.3)
0.92 ± 1.08
0.931
Annona cherimolia
65.10 (33.18 – 127.71)
685.3 (349.3 – 1344.5)
1.32 ± 0.76
0.801
Annona muricata
84.92 (42.20 – 170.91)
866.5 (430.6 – 1743.9)
1.28 ± 0.78
0.918
Tabernaemontana cymosa
25.02 (22.85 – 27.41)
73.6 (51.6 – 104.8)
2.15 ± 0.19
0.938
Mammea americana
38.58 (34.34 – 43.34)
143.8 (96.2 – 215.1)
1.75 ± 0.16
0.912
3.4 Bioassay-guided fractionation
Tabernaemontana cymosa extract was fractionated as a result of its high larvicidal activity. Four fractions were obtained from the first chromatographic process. Next, fraction 1A (11.4 g) was eluted with a mobile phase of hexane and ethyl acetate mixture (7:3 ratio) followed by fraction F1B (5.3 g) eluted with the same solvents but in a 1:1 proportion. Subsequently, the gradient was increased, eluting the fractions in ethyl acetate (100 %) and methanol (100 %) to obtain fractions F1C (3.5 g) and F1D (1.9 g), respectively. All fraction were subjected to biological evaluation against A. aegypti larvae at 50 ppm. The bio-directed fractionation continued as the biological activity was concentrated in the fractions eluted with hexane and ethyl acetate mixtures, as shown in Fig. 1. The fractions F1A and F1B exerted a potent larvicidal activity since they reached more than 80 % mortality within the first 6 h of treatment. Therefore, these fractions were separated by serial column chromatography as shown in Fig. 2.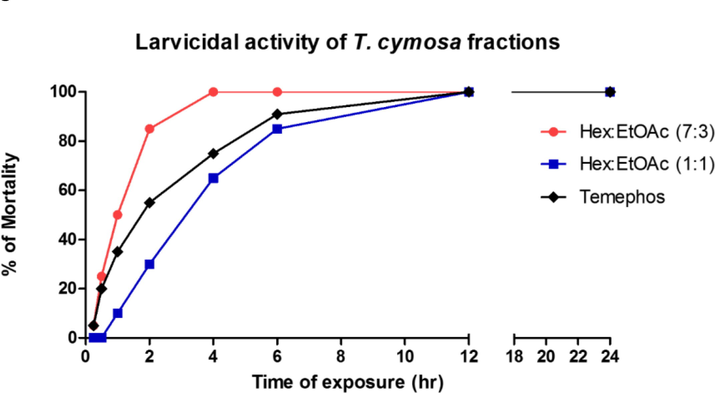
Larvicidal activity of Tabernaemontana cymosa fractions at 50 ppm. F1C (Ethyl acetate) and F1D (methanol) fractions were not active. Hexane (Hex), ethyl acetate (EtOAc).
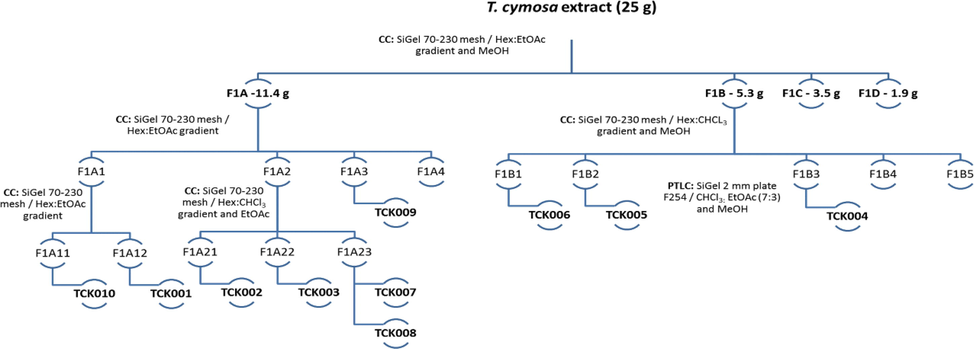
General scheme of compounds isolation from Tabernaemontana cymosa extract. Hexane (Hex); Ethyl acetate (EtOAc); Methanol (MeOH); Column Chromatography (CC); Preparative Thin Layer Chromatography (PTLC). Isolated compounds: voacangine (TcK001), voacangine-7-hydroxyindolenine (TcK002), lupeol (TcK003), rupicoline (TcK004), 3-oxo-voacangine (TcK005), 6-enyl-6-phormyl-voacangine (TcK006), β-sitosterol (TcK007), stigmasterol (TcK008), α-spinasterol (TcK009) and lupeol acetate (TcK010).
The hexane–ethyl acetate (7:3) soluble fraction (F1A – 11.4 g) was the most potent larvicide against A. aegypti, with a 100 % of mortality within the first four hours of treatment. As a consequence, the F1A fraction was chromatographed on a 4.5 × 57 cm SiO2 (200 g; 70–230 mesh) column using a hexane and ethyl acetate elution gradient [12:1, 8:2,6:4 ratios], followed by chloroform and ethyl acetate as can be seen in Fig. 2. Phytochemical screening was simultaneously carried out to monitor all eluted fractions, and those with the similar pattern, were pulled together. Previous studies showed the presence of indole alkaloids in the ethanol extract of T. cymosa (Oliveros-Díaz et al., 2021), therefore, the relative abundance and detection of this kind of metabolites were taken as a criterion to combine the eluted fractions. The presence of alkaloids was determined through chemical treatment using Dragendorff reagent.
After F1A fractionation, four new subfractions were obtained; alkaloids and triterpenes were detected in subfractions F1A1 eluted with Hex:EtOAc (12:1) and F1A2 eluted with Hex:EtOAc (8:2). Both subfractions were active against III and IV instar larvae at 50 ppm after 24 h. However, 100 % mortality was recorded after four hours of treatment for fraction F1A1, indicating a higher concentration of actives compounds. The active subfraction F1A1 was subsequently chromatographed employing the same stationary phase and elution gradient to obtain two fractions, F1A11 and F1A12, which showed precipitates as white powders.
Finally, the precipitates were cleaned using opposite polarity solvent exhibiting different crystallization patterns. The compound TcK010 isolated from F1A11 precipitated as a white powder, while compound TcK001 isolated from F1A12 precipitated as white needles. Thin Layer Chromatography (TLC) analysis also demonstrated differences between both compounds, unlike the F1A11 precipitate, F1A12 precipitate showed spots under UV-Light at 254 and 366 nm. After chemical treatment with Dragendorff and Vainillin reagent, it was determined that the isolated compounds were probably an alkaloid (TcK001) and a triterpenoid (TcK010). The compound TcK001 was highly active for mosquito larvae at 50 ppm; hence it was selected for LC50 experiments.
For F1A2 subfraction, open column chromatography was used with the same experimental conditions arranged for fractionation of F1A1 subfraction. The elution gradient was carried out with a mobile phase composed of hexane and ethyl acetate in proportions 9:1, 8:2 and 6:4, followed by chloroform and ethyl acetate at 100 %. In such manner, the compounds TcK002, TcK003, TcK009 and TcK010 were isolated by crystallization from subfractions F1A11, F1A21, F1A22 and F1A23, respectively, as shown Fig. 2. None of those compounds were active against mosquito larvae.
Bioassay-guided fractionation continued with the F1B fraction using a column of dimensions 3.5 × 57 cm and 120 g SiO2 Merck® (60–230 mesh). A hexane-CHCl3 elution gradient in proportions of 9:1, 7:3 and 1:1 was used as mobile phase, followed by fractions eluted with CHCl3, EtOAc and MeOH. Due to the similarity in TLC patterns, the hexane/CHCl3 9:1 and 7:3 fractions were combined to obtain the F1B1 subfraction, while the hexane/CHCl3 1:1, CHCl3, EtOAc and MeOH constituted the subfractions F1B2, F1B3, F1B4 and F1B5, respectively. Alkaloids were detected in the fractions from F1B1 to F1B3 through TLC using the Dragendorff reagent, and the precipitate called TcK005 was cleaned by washing with a solvent of opposite polarity (hexane). On the other hand, it was necessary to use PTLC to isolate compounds TcK004 and TcK006 with the conditions shown in Fig. 2. After the plates were developed, the bands of interest were cut and extracted with ethyl acetate.
3.5 Compounds elucidation
The isolated and purified compounds were sent to the University of Antioquia in Medellin for NMR experiments (1D and 2D). The NMR spectra were obtained and interpreted in collaboration with Laboratory of Organic Chemistry of Natural Products research group. Five alkaloids were identified and characterized from the active fractions of T. cymosa (TcK001, TcK002, TcK004, TcK005, TcK006); the indole alkaloid voacangine (TcK001) was the main compound and the most active one against A. aegypti larvae. In addition, compound TcK006 was characterized and, as far as we know, correspond to a new indole alkaloid from T. cymosa specie. Likewise, the isolation and elucidation of the known compounds voacangine-7-hydroxyindolenine, rupicoline and 3-oxovoacangine were also achieved.
During the fractionation process, a variety of terpenoids and sterols were isolated, most of them as white powders or solids (Lupeol [TcK003], Sitosterol [TcK007], Stigmasterol [TcK008], Spinasterol [TcK009] and Lupeol acetate [TcK010]). The detection was made by developing TLC plates with vanillin-sulfuric acid (H2SO4) reagent, as mentioned above, and visualizing purple or pink spots when heated. On the other hand, alkaloid detection was achieved when orange or red bands were observed using Dragendorff reagent after immersion and drying of the plates.
Comparative TLC was carried out for the alkaloids detected after fractionation (see Fig. 3), the other resulting compounds were visualized sep arately since they were not visible under any UV wavelength. They were tested by chemical treatment using Vanillin (1 %) and H2SO4 (5 %) reagents before heating until spots were evident (See Figure S1). The chemical structure of each compound is shown in Fig. 4. Spectral and physical characteristics of these compounds are described below: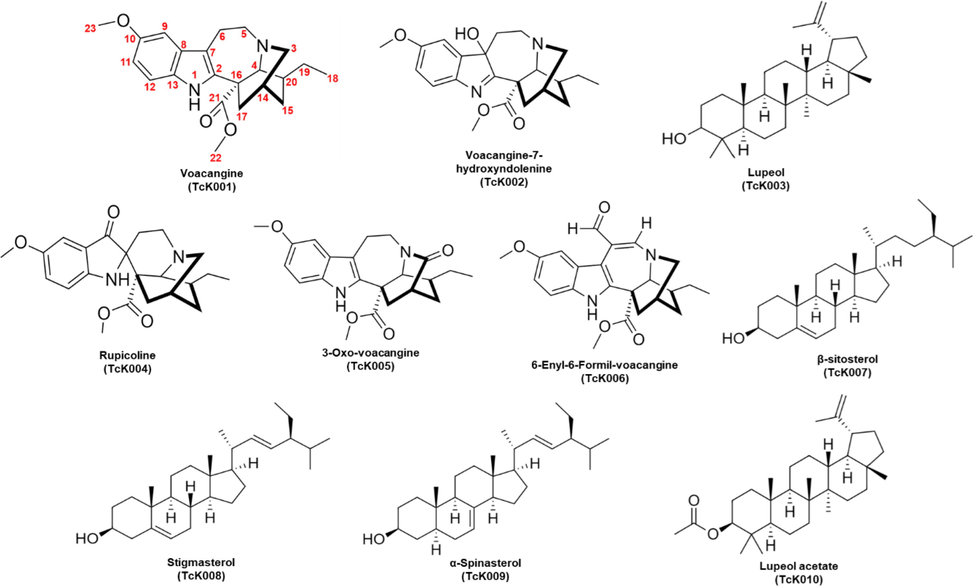
Molecular structure of compounds isolated from Tabernaemontana cymosa.

Comparative Thin Layer Chromatography (TLC) of isolated compounds from T. cymosa. a) UV–vis 254 nm; b) UV–vis 366 nm; c) Chemical identification using Dragendorff reagent. Voacangine (TcK001); voacangine-7-hydroxyindolenine (0 0 2); rupicoline (0 0 4); 3-oxo-voacangine (0 0 5); 6-enyl-6-formil-voacangine (0 0 6).
Voacangine (TcK001) exhibited the following physical and spectral properties: Crystalline needles; Mp: 137–138 °C; Rf: 0.8 (hexane:ethyl acetate 6:4). 1H NMR (300 MHz, CDCl3): δ 7.73 (s, NH), 7.17 (d, J = 8.7 Hz, H-12), 6.96 (d, J = 2.6 Hz, H-9), 6.84 (dd, J = 8.6 and 2.6 Hz, H-11), 3.89 (s, OCH3), 3.73 (s, CO2CH3), 1.44 (q, J = 7.0 Hz, H-19), 0.92 (t, J = 7.0 Hz, H-18). 13C NMR (75 MHz, CDCl3): δ 176.03 (CO2Me), 154.11 (C-10),137.65 (C-2), 130.63 (C-13), 129.31 (C-8), 111.96 (C-11), 111.23 (C-12), 110.24 (C-9), 100.84 (C-7), 57.69 (C-21), 56.15 (OCH3), 55.25 (C-16), 53.24 (C-5), 52.74 (CO2CH3), 51.60 (C-3), 39.27 (C-20), 36.67 (C-17), 32.14 (C-15), 27.44 (C-14), 26.86 (C-19), 22.33 (C-6), 11.82 (C-18).
Voacangine-7-hydroxyindolenine (TcK002) exhibited the following physical and spectral properties: amorphous yellow-green solid; Mp: 135–137 °C; Rf: 0.5 (hexane:ethyl acetate 6:4). 1H NMR (300 MHz, CDCl3): δ 7.36 (d, J = 9 Hz, H-12), 6.91 (s, H-9), 6.80 (dd, J = 6 and 3 Hz, H-11), 3.82 (s, OCH3), 3.70 (s, CO2CH3), 0.86 (t, J = 7 Hz, H-18) ppm. 13C NMR (75 MHz, CDCl3): δ 186.97 (C-2), 174.05 (CO2Me), 159.23 (C-10), 144.90 (C-13), 144.53 (C-8), 121.49 (C-12), 113.85 (C-11), 108.09 (C-9), 88.43 (C-7), 58.68 (C-21), 55.88 (ArOCH3; C16), 53.39 (CO2CH3), 49.22 (C-5), 48.75 (C-3), 37.68 (C-20), 34.63 (C-17), 34.25 (C-6), 32.14 (C-15), 27.08 (C-14), 26.62 (C-19), 11.71 (C-18).
Lupeol (TcK003) exhibited the following physical and spectral properties: crystalline needles; Mp: 212–214 °C; Rf: 0.32 (chloroform). 1H NMR (300 MHz, CDCl3) δ: 4.67 (s, H-29a), 4.56 (s, H-29b), 3.16 (m, H-3), 1.25 (s, H-30), 1.03 (s, H-26), 0.96 (s, H-23), 0.94 (s, H-24), 0.83 (s, H-25), 0.78 (s, H-27), 0.76 (s, H-28). 13C NMR (75 MHz, CDCl3) δ 151.15 (C-20), 109.47 (C-29), 79.17 (C-3), 55.43 (C-5), 50.58 (C-9), 48.44 (C-18), 48.14 (C-19), 43.15 (C-17), 42.97 (C-14), 40.97 (C-8), 40.15 (C-22), 39.01 (C-13), 38.84 (C-4), 38.19 (C-1), 37.31 (C-10), 35.73 (C-16), 34.42 (C-7), 29.99 (C-21), 29.85 (C-23), 28.14 (C-15), 27.55 (C-12), 25.27 (C-2), 21.07 (C-11), 19.45 (C-30), 18.46 (C-6), 18.15 (C-28), 16.27 (C-25), 16.12 (C-26),15.52 (C-24), 14.69 (C-27).
Rupicoline (TcK004) exhibited the following physical and spectral properties: yellow crystals; Mp: 252–253 °C; Rf: 0.0 (hexane: ethyl acetate 6:4). 1H NMR (300 MHz, CDCl3): δ 7.07 (dd, J = 8 and 3 Hz, H-11), 7.02 (s, H-9), 6.76 (d, J = 9 Hz, H-12), 4.29 (s, N—H), 3.95 (s, H-4), 3.76 (s, OCH3), 3.30 (s, CO2CH3), 1.91 (s, H-14), 0.91 (t, J = 6 Hz, H-18). 13C NMR (75 MHz, CDCl3): δ 202.9 (C-7), 174.57 (CO2Me), 154.21 (C-10), 153.82 (C-13), 126.89 (C-11), 121.78 (C-8), 114.12 (C-12), 104.63 (C-9), 55.90 (ArOCH3), 52.07 (C-3), 52.07 (C-16), 52.05 (C-21), 51.31 (CO2CH3), 47.67 (C-5), 35.81 (C-20), 31.1 (C-15), 30.8 (C-17), 28.69 (C-19), 26.08 (C-14), 25.77 (C-6), 12.14 (C-18).
3-oxo-voacangine (TcK005) exhibited the following spectral properties: amorphous white powder; Mp: 253–254 °C; Rf: 0.13 (hexane: ethyl acetate 6:4). 1H NMR (300 MHz, CDCl3): δ 7.92 (s, NH), 7.16 (d, J = 8.7 Hz, H-12), 6.95 (d, J = 2.1 Hz, H-9), 6.84 (dd, J = 8.7 and 2.4 Hz, H-11), 3.88 (s, OCH3), 3.77 (s, CO2CH3), 0.97 (t, J = 7 Hz, H-18). 13C NMR (75 MHz, CDCl3): δ 175.87 (C-3), 173.07 (CO2Me), 154.21 (C-10), 134.65 (C-2), 130.83 (C-13), 128.23 (C-8), 112.59 (C-11), 111.38 (C-12), 109.22 (C-9), 100.48 (C-7), 56.11 (C-16), 56.02 (C-21), 55.60 (OCH3), 53.09 (CO2CH3), 42.72 (C-5), 38.21 (C-20), 35.97 (C-14), 35.49 (C-17), 31.03 (C-15), 27.67 (C-19), 21.18 (C-6), 11.41 (C-18).
6-enyl-6-phormyl-voacangine (TCK006) exhibited the following spectral properties: amorphous white powder; Mp: 256 °C; Rf: 0.27 (hexane: ethyl acetate 6:4). 1H NMR (300 MHz, CDCl3): δ 10.09 (s, H-24), 8.23 (s, H-6), 7.78 (d, J = 3 Hz, H-9), 7.32 (d, J = 9.0 Hz, H-12), 6.90 (dd, J = 9.0 and 3.0 Hz, H-11), 5.03 (bs, H-4), 3.87 (s, OCH3), 3.66 (s, CO2CH3), 0.97 (t, J = 6.0 Hz, H-18). 13C NMR (75 MHz, CDCl3): δ 185.42 (C-24), 172.04 (CO2Me), 165.22 (C-5), 156.94 (C-10), 149.72 (C-2), 129.69 (C-13), 127.31 (C-8), 114.55 (C-11), 114.15 (C-6), 114,13 (C-7), 112.96 (C-12), 103.03 (C-9), 55.95 (OCH3), 53.73 (CO2CH3), 52.61 (C-16), 48.42 (C-3), 47.53 (C-4), 38.29 (C-17), 36.14 (C-20), 30.27 (C-15), 28.24 (C-19), 26.63 (C-14), 11.70 (C-18).
β-sitosterol (TcK007) exhibited the following physical and spectral properties: Amorphous solid, Mp: 137.9–139.6 °C; Rf: 0.2 (chloroform). 1H NMR (300 MHz, CDCl3) 5.40 (m, H-6), 3.25 (m, H-3), 1.36 (bs*, H-22), 1.19 (bs, H-23), 1.05 (s, H-19), 0.85 (d, J = 6.5 Hz, H-27), 0.73 (s, H-18), 0.72 (t, J = 7.8 Hz, H-29). 13C NMR (75 MHz, CDCl3): δ 140.61 (C-8), 121.77 (C-6), 71.84 (C-3), 56.78 (C-14), 56.78 (C-17), 50.14 (C-9), 42.50 (C-4), 40.89 (C-20), 40.27 (C-5), 39.48 (C-12), 38.00 (C-4), 37.16 (C-1), 34.24 (C-10), 31.49 (C-2), 29.74 (C-6), 29.66 (C-16), 25.44 (C-28), 23.05 (C-15), 21.57 (C-11), 21.41 (C-21), 21.14 (C-26), 19.02 (C-19), 13.09 (C-27), 12.30 (C-18), 12.08 (C-29). *Broad signal.
Stigmasterol (TcK008) exhibited the following physical and spectral properties: Amorphous solid; Mp (Mixture with TcK007); Rf: 0.2 (chloroform). 1H NMR (300 MHz, CDCl3) δ: 5.35 (m, H-6), 5.16 (m, H-22), 5.02 (m, H-23), 3.53 (m, H-3), 0.92 (d, J = 6.5 Hz, H-21), 0.85 (bs, H-26), 0.84 (s, H-18), 0.83 (s, H-19), 0.71 (t, J = 7.8 Hz, H-29).13C NMR (75 MHz, CDCl3) δ: 140.77 (C-5), 138.33 (C-22), 129.28 (C-23), 121.74 (C-6), 71.83 (C-3), 56.78 (C-14), 55.96 (C-17), 51.25 (C-9), 50.14 (C-24), 42.32 (C-13), 42.23 (C-4), 40.51 (C-20), 39.79 (C-12), 37.26 (C-1), 36.52 (C-10), 31.91 (C-8), 31.9 (C-7), 31.9 (C-25), 31.68 (C-2), 28.26 (C-16), 25.42 (C-28), 24.32 (C-15), 21.23 (C-26), 21.1 (C-11), 19.83 (C-27), 19.41 (C-19), 18.99 (C-21), 12.26 (C-18), 11.99 (C-29).
α-Spinasterol (TcK009) exhibited the following physical and spectral properties: White needle; Mp 151–153 °C; Rf: 0.17 (chloroform). 1H NMR (300 MHz, CDCl3) δ: 5.21 (m, H-7), 5.19 (m, H-23), 5.10 (m, H-22), 3.65 (m, H-3), 1.06 (d, J = 6.5 Hz, H-21), 0.88 (d, J = 6.2 Hz, H-26), 0.84 (s, H-18), 0.83 (s, H-19), 0.83 (d, J = 6.5 Hz, H-27), 0.59 (t, J = 7.8 Hz, H-29). 13C NMR (75 MHz, CDCl3) δ: 139.72 (C-8), 138.33 (C-22), 129.56 (C-23), 117.60 (C-7), 71.22 (C-3), 56.01 (C-17), 55.26 (C-14), 51.39 (C-24), 49.57 (C-9), 43.42 (C-13), 41.00 (C-20), 40.39 (C-5), 39.59 (C-12), 38.12 (C-4), 37.27 (C-1), 34.36 (C-10), 32.02 (C-25), 31.61 (C-2), 29.77 (C-6), 28.67 (C-16), 25.55 (C-28), 23.16 (C-15), 21.68 (C-11), 21.53 (C-21), 21.26 (C-26), 19.13 (C-27), 13.20 (C-19), 12.41 (C-29), 12.20 (C-18).
Lupeol acetate (TcK010) exhibited the following physical and spectral properties: crystalline needles; Mp: 212–214 °C; Rf: 0.85 (chloroform). 1H NMR (300 MHz, CDCl3) δ: 4.68 (s, H-29), 4.56 (s, H-29), 4.46 (m, H-3), 2.04 (s, H-2′), 1.68 (s, H-30), 1.03 (s, H-25), 0.93 (s, H-28), 0.85 (s, H-26), 0.84 (s, H-23), 0.83 (s, H-26), 0.78 (s, H-27). 13C NMR (75 MHz, CDCl3) δ 171.19 (C-1′), 151.12 (C-20), 109.49 (C-29), 81.12 (C-3), 55.51 (C-5), 50.47 (C-9), 48.42 (C-18), 48.15 (C-19), 43.14 (C-17), 42.96 (C-14), 40.98 (C-8), 40.14 (C-22), 38.52 (C-1), 38.17 (C-4), 37.93 (C-10), 37.22 (C-13), 35.71 (C-16), 34.34 (C-7), 29.97 (C-21), 28.09 (C-2′), 27.57 (C-23), 25.22 (C-15), 23.85 (C-12), 21.48 (C-2), 21.08 (C-11),19.43 (C-30), 18.34 (C-6), 18.14 (C-28), 16.64 (C-24), 16.32 (C-25),16.11 (C-26), 14.64 (C-27).
3.6 Ichthyotoxicity assays
Based on larvicidal results described in sections 3.3 and 3.4, twelve extracts with high larvicidal activity were tested for toxic activity against a non-target organism. All the extracts were highly toxic to the guppy fish at the concentration of 200 ppm after 24 h of exposure (Table S3). Consequently, it was considered unnecessary to carry out the TC50 determination on all extracts. However, TC50 was calculated for Tabernaemontana cymosa and Mammea americana extracts because these two were the most promising extracts against Aedes aegypti larvae. Due to both extracts causing 100 % of fish mortality at different times, the calculation of the TC50 value was developed at 10 min for M. americana and 30 min for T. cymosa. As shown in Table S3, in this study it was found that these larvicide-active extracts are very toxic to the guppy fish used as biological control, with TC50 values of 36.13 and 27.33 ppm for M. americana and T. cymosa, respectively. Additionally, the Selectivity Index (SI) was calculated for both extracts; for T. cymosa and M. americana SI were 1.09 and 0.94 respectively.
3.7 Toxic effect assessment against model organism Caenorhabditis elegans
The indole alkaloid voacangine was selected to toxicity assessement due to the high larvicidal activity showed.The in vivo test results against C. elegans are displayed in Fig. 5., where a concentration-dependent behavior was observed (p < 0.05). None of the treatments showed toxicity against the nematode, since only 10 % mortality was observed at 200 ppm. Thus, the indole alkaloid voacangine did not represent a threat to the nematode survival; hence its impact on human health is probably nule.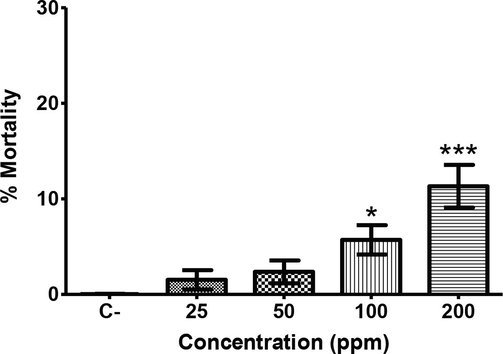
Percent of mortality on C. elegans exposed to voacangine at concentrations ranging from 25 to 200 ppm. *Represent significant differences with respect to the control (p < 0.05).
4 Discussion
Plant extracts have been positioned as the main alternative to search for bioinsecticides for control programs due to their biodegradability characteristics, low cost and environmental safety (Tavares et al., 2021; Abdel Haleem et al., 2022). In this research, 65 plant extracts from the Colombian Caribbean Region were evaluated, of which 16 were classified as highly active at a concentration of 200 ppm (Table S2). Among them, the most potent extracts were Annona cherimolia, Annona muricata, Annona squamosa, Tabernaemontana cymosa and Mammea americana. Larvicidal activity against different mosquito species had been well studied for the Annonaceae species thus the seeds extracts of Annona muricata, Annona squamosa, and Annona cherimolia were considered references since these species and their high toxicity is widely reported in the literature (Mehra and Hiradhar, 2000; Costa et al., 2011; Shad, 2017; de Sousa et al., 2020). As considered from the beginning, the extracts of the Annonaceae family showed a good activity with LC50 of 58.44, 65.10, and 84.92 ppm for Annona squamosa, Annona cherimolia and Annona muricata, respectively. Additionally, the ethanol extracts of two Colombian Coast plants were very actives against mosquitoe larvae. Seeds extract of the wild plant Tabernaemontana cymosa showed the highest toxicity with a LC50 of 25.02 ppm and the seeds extract of Mammea americana, which is a tropical frutal tree, also showed larvicidal activity with a LC50 of 38.58 ppm.
As mentioned above, plants in the Annonaceae family are well known for their biological activity against different mosquito species, with reports of high biological activity at levels below 0.05 % of their seed extracts (Shad, 2017). Furthermore, the obtained data support previous reports, since, for the plants Annona cherimolia, Annona muricata and Annona squamosa we found LC50 values against mosquito larvae very approximated to 70 ppm, as can be seen in Table 1. Although most extracts tested had low larvicidal effects (bellow 20 % of mortality) against the Aedes aegypti mosquito, our results showed two promising native species as larvicides against the world's largest arbovirus vector. The LC50 values found for Tabernaemontana cymosa and Mammea americana seed extracts exceed many of those reported in the literature over the past two decades (Piplani et al., 2019).
For each of the five most promising extracts, their LC90 values were calculated in the same way. As can be seen in Table 1, values above 500 ppm were obtained for the Annonas and values below 200 ppm for the extracts of M. americana and T. cymosa. In our tests we were able to evidence the formation of emulsions when the extracts are tested above 200 ppm, therefore these concentrations are not viable due to their impact on water organoleptic properties, which would imply problems in their field application. Otherwise, the LC90 values for T. cymosa (73 ppm) and M. americana (143 ppm) are acceptable for storage water but the results of ichiotoxicity represent a barrier to overcome in order to avoid damage to ecosystems.
An interesting behavior was evidenced (see Fig. 6), related to the way in which plants concentrate active metabolites in their seeds. A total of nine seed extracts were potent larvicides (greater than80 % of mortality), representing 56 % of the identified active extracts in this study. Fig. 6 also shows this pattern for the two most potent species (red dots for T. cymosa and blue dots for M. americana), where it is seen that for the alkaloids rich specie T. cymosa, the extract of the seeds was very potent while none of the other organs exerted activity. The same behavior happened with Mammea americana; however, the leaves of this plant also showed activity, although moderate. Therefore, we assume that the seeds are the primary organ where active metabolites are concentrated and the study of wild plant seeds could be a valid approach to obtain new active molecules.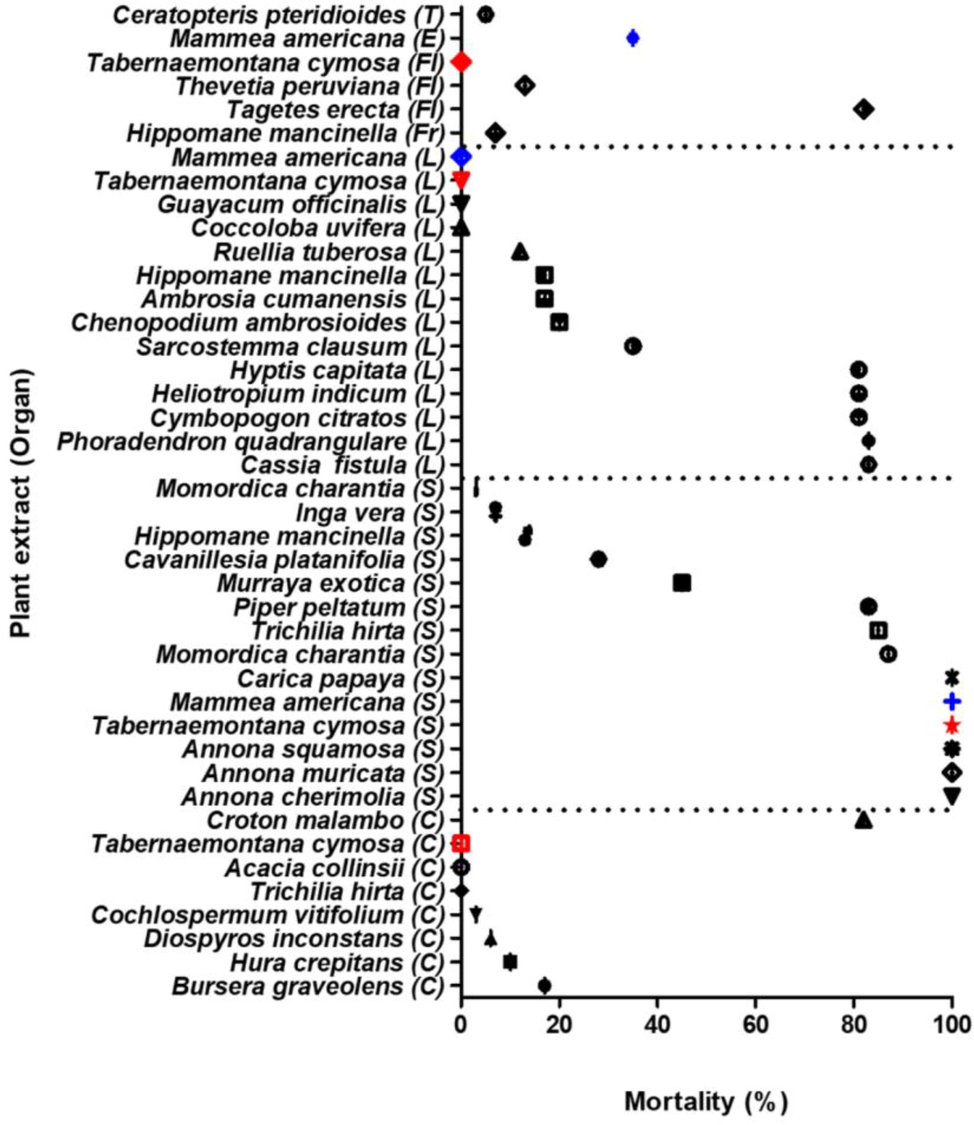
Larvicidal activity of plant extract. Red points indicate T. cymosa and blue points indicate M. Americana. C: stem bark; Fl: flowers; Fr: fruit; L: leaves; E: endosperm; S: seed; T: stem.
The phytochemicals used against mosquitoes differ in their efficacy, as previously mentioned, depending on plant part and species, solvents used in the extraction and secondary metabolites abundance (Arivoli et al., 2016). Previous reports on the chemical composition of Tabernaemontana cymosa showed that alkaloid-type compounds are the most common components present in seed extract of this plant; however, we also found the presence of other metabolites such as triterpenes, sterols, glycosides, saponins, quinones (Beltrán Villanueva et al., 2013). The abundance of alkaloids in the extract of T. cymosa could suppose that these metabolites are responsible for their larvicidal activity, since this family of metabolites has been reported as potential larvicides (Liu et al., 2012; Touré et al., 2017). The antiviral activity of alkaloids isolated from T. cymosa and the extract capacity for carbonylate midgut proteins from A. aegypti mosquito have been proven (Gómez-Calderón et al., 2017; Rodríguez-Cavallo et al., 2019). A bioguided fractionation was carried out to identify the active principles as larvicides of the plant, finding that the most active fractions were those with a high content of alkaloids.
We were able to isolate five alkaloids present in the T. cymosa extract through chromatographic techniques, which showed a characteristic base structure of Iboga class molecules. Fortunately, the major alkaloid called voacangine turned out to be the most potent compound against the larvae of the A. aegypti mosquito with an LC50 of 5.13 ppm as seen in Fig. 7. The interpretation of MNR spectra and the comparison with the literature allowed the identification of a typical spectral imprint of the molecular structure of voacangine, repeating the pattern in two other molecules (voacangine-7-hydroxyindolenine and 3-oxovoacangine) that conserve a similar chemical structure (Kingston, 1978; Pereira et al., 2008; Foudjo et al., 2021). These compounds have been widely discussed in previous studies (Monsalve-Escudero et al., 2021a, 2021b).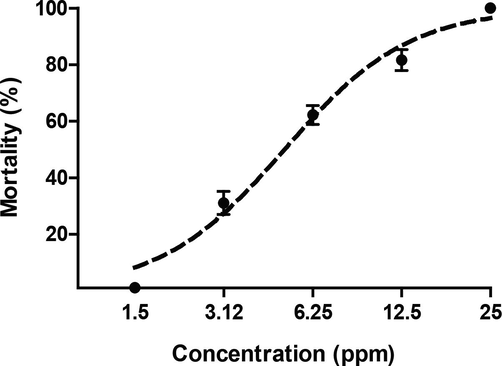
Dose-response curve of voacangine larvicidal effect on A. aegypti larvae.
The 1H NMR spectrum of voacangine (TcK001) showed three well differentiate groups of signals, corresponding to an aromatic and an aliphatic regions, separated by two singlets.The 1H NMR spectrum of this compound showed a triplet at δ 0.89 (t, 3H, J = 7.0 Hz, H-18) indicates a methyl coupled to a methylene at δ 1.44 (q, 2H, J = 7.0 Hz, H-19). 3-proton singlets at δ 3.85 and 3.71 are assigned to the MeO groups at positions 23 and 22. From the HMBC spectrum correlations was possible to establish that these two groups are attached to the aromatic ring (CH3O–, H-23) and to the ester group (CH3O–, H22). In the aromatic region an AMX spin system is observed corresponding to the coupling among three signals at 7.17 ppm (d, J = 8.7 Hz, H-12), 6.84 ppm (dd, J = 8.7 and 2.6 Hz, H-11) and at 6.96 ppm (d, J = 2.6 Hz, H-9). The proton signal belonging to the amino group is observed at a chemical shift of 7.9 ppm. Each proton signal was related to its carbon using 2D NMR HSQC.
Protons signals were then correlated with neighboring carbons using HMBC to corroborate the position of the methoxyl, acetate and ethyl groups in the structure of the molecule. Thus, the methoxyl protons at 3.85 ppm show correlation with the carbon at 110 ppm (C-10), which indicates their location in the aromatic ring of the indole group. The second characteristic methoxyl signal correlates with an ester carbonyl group at 175 ppm thus placing it in the C-22 position. Finally, the spectrum show the strong correlations among protons and carbons of the aromatic system, as expected (Achenbach et al., 1997; Pereira et al., 2008).
In the case of compound TcK005, the same base structure of voacangine was detected by 1H and 13C NMR experiments, except for carbon C-3. According to the 13C NMR spectrum, an additional signal at 175.87 ppm indicates the presence of a ketone carbonyl group binded to a hetrocylic nitrogen in the structure. Besides, the DEPT spectrum of compound TcK005 showed one methylene signal less than voacangine which could be explained because in the Cosy spectrum the methine proton at C-14 showed no correlation with any other proton in the molecule, so the carbonyl should be replacing to the methylene in position C-3 (Thomas and Biemann, 1968; Pereira et al., 2008). The subsequent comparison with the literature corroborated our interpretation, leading us to determine this compound as 3-oxo-voacangine (TcK005) alkaloid.
Compound TcK002 also showed a proton spectral fingerprint corresponding to indole alkaloids, very similar to voacangine, which helped us to identify its base structure. Interestingly, this molecule does not have the proton signal characteristic of the N-heteroatom, which indicates that the nitrogen of the indole group has an unsaturation. The analysis of the carbon spectrum allowed the identification of an oxygenated quaternary carbon with a chemical shift of 88 ppm. This carbon was especially useful for comparisons with the literature, thus identifying the molecule as voacangine-7-hydroxyindolenine (TcK002) (Pereira et al., 2008; Zhang et al., 2018).
Furthermore, the compound TcK004 showed a bright yellow-green fluorescence, which was very striking since the chemical tests with Draggendorff reagent showed that the compound was of the alkaloid type. This behavior on chromatographic plates were the first indication of a totally different indole type alkaloid. Despite this, when conducting the 1H NMR experiments, we realized that it had the same spectral fingerprint as the major compound (voacangine, TcK001). 13C NMR spectra revealed a highly displaced carbonyl ketone at 202 ppm, which is not common in these compounds. Two-dimensional spectra such as HMBC and comparison with literature reports mentioning bright yellow-green fluorescence alkaloids led us to determine that the molecule correspond to the structure of rupicoline (Niemann and Kessel, 1966; Thomas and Biemann, 1968).
Compound TcK006 showed similar spectral characteristics to the indole alkaloid Voacangine (TcK001). Despite detecting the spectral fingerprint of voacangine in the 1H NMR spectrum, the presence of two unprotected signals with chemical shifts of 8.23 and 10.09 ppm was evidenced, which had not been detected in any of the alkaloids previously identified These signals in the HSQC spectrum correlate with carbons at 165.22 and 185.42 ppm, respectively, suggesting an aldehyde group and a highly deshielded vinyl group probably under the influence of another electronegative nucleus. The DEPT spectrum of compound TcK006 showed only four methylenes groups instead of six as shown by the structure of voacangine. The analysis of the HMBC spectrum allowed to place the vinyl proton (8.23 ppm) in position C-5 (165.22 ppm), because it correlates with the methine group at C-4 (5.03/47.57 ppm). The explanation of the unshielding of proton H-5 and carbon C-5 is due to the presence of a neighbor nitrogen in the structure. The HMBC spectrum also shows correlations of the aldehyde proton at 10.09 ppm with carbons signals at 114.15 (C-6), 114.13 (C-7) and 137.32 (C-8) ppm, which indicates that this aldehyde group is located at carbon C-6. (See Fig. 8).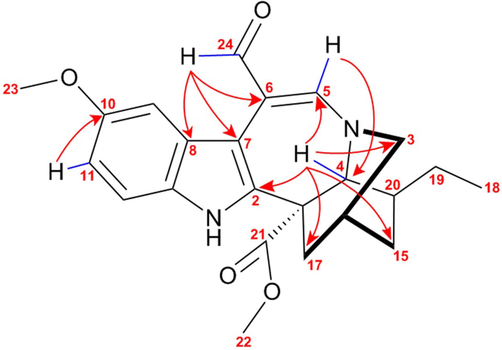
Bidimensional NMR correlations of 6-Enyl-6Formil-voacangine; HSQC correlations are shown with blue bonds and HMBC with red arrows.
Finally, we achieved the structural elucidation of the compound named 6-enyl-6-formyl-voacangine which, after reviewing the databases and literature corresponds, to the best of our knowledge, to a new indole alkaloid isolated from Tabernaemonata cymosa. All NMR spectra is available in Supplementary Materials (Figures S2-S7).
On the other hand, the seeds extract of Mammea americana showed promising activity due to the potency of its larvicidal effect, obtaining a mortality of 43 % in the first hour of exposure. Coumarins are the most representative secondary metabolites in M. americana seed extract, flavonoids and sterols were also detected (Beltrán Villanueva et al., 2013). Some types of coumarins have been reported as promising molecules for mosquito control such as Imperatorin, being this linear coumarins, the most active of 13 relative-compounds tested by Wang and collaborators, obtaining an LC50 of 2.88 ppm for A. aegypti (Wang et al., 2012).The pertinent studies are in course to isolate and characterize the M. americana molecules responsible for the larvicidal activity as possible sources of bioinsecticides. Recent reports have shown that coumarins possess important biological activity against some resistant bacteria. Hence the isolation and evaluation of the larvicidal properties of mammea coumarins is a current approach in ongoing investigations (Pájaro-González et al., 2022c).
In order to check the ecotoxicological repercussions of our extracts, we selected the most active ones and tested them against the guppy fish. Unfortunately, all extracts with high larvicidal activity were also highly toxic against this fish at a concentration of 200 ppm, mainly extracts from the Mammea americana, Momordica charantia, Trichilia hirta, Cassia fistula, Piper peltatum and Croton malambo plants, which exhibited 100 % mortality within the first half hour of testing. The fish exposed to the T. cymosa extract perished in their entirety after 1 h of testing. Due to the high ichthyotoxic activity of these extracts against the fish Poecillia reticulata, we assumed that the determination of TC50 against the fish of all extracts was not feasible. Therefore, the calculation was made for the two most promising extracts as larvicides. As a consequence of its high degree of toxicity, mortality readings were taken at very short times of 10 min for T. cymosa and 30 min for M. americana, obtaining TC50 values of 27.33 ppm and 36.13 ppm, respectively. Additionally, the SI for T. cymosa (SI = 1.09) was similar for M. americana (SI = 0.94), indicating that both extracts have the same toxicity potential.
Since the highest percentage of disease-transmitting mosquito breeding sites in our region correspond to artificial reservoirs inside houses, the application of extracts or pure compounds in tanks, pools or containers with continuous stored water at a controlled concentration is a potential alternative for use in outbreak situations (Anogwih et al., 2015; Anoopkumar et al., 2020; Adeoye-Isijola et al., 2021). Chronic effects have been studied in mosquito larvae for sublethal concentrations, damaging certain processes in the larvae inhibiting their development, reproduction and oviposition (Ramkumar et al., 2019; Chellappandian et al., 2022). In the present study triterpenens and sterol were isolated from the most active plant, this compounds could have potential as endocrine disruptors due to their structural similarity with ecdysteroids and work as Growth Regulators (Savchenko et al., 2022). On the other hand, indole alkaloids target the central nervous system of the larvae causing behavioral changes leading to a loss of reproductive and feeding instincts, consequently, a decrease in the population (Carreño Otero et al., 2018; Balachandran et al., 2021).
Finally, C. elegans bioassays demonstrated that the bioactive alkaloid voacangine did not exert toxicity against the model organism compared with the activity evidenced against Aedes aegypti larvae. These results correlate with the in silico data collected in previous studies (Oliveros-Díaz et al., 2021) due to this compound showed the lowest level of interaction with human proteins, which could be extrapolated to C. elegans considering the high homology between human and nematode proteins (Ayuda-Durán et al., 2020). Since the voacangine LC50 value for C. elegans is greater than 200 ppm, the selectivity index would be above 40, making this compound a viable alternative for destroying mosquito breeding sites in areas with dengue outbreaks without putting the community at risk.
Recently, our research group has published a review where we compile relevant information on the health benefits of plants from the Colombian Caribbean. During the review we found several traditional uses of plants such as Anonna muricata, which has antipyretic and anti-flu properties (Pájaro-González et al., 2022b). Around the world, this plant is known by various names and has more than 40 different uses, from antidiarrheal to antihypertensive and even antimalarial (Coria-Téllez et al., 2018). There are reports for traditional uses of Anonna squamosa to treat lices, ulcers, vomiting and tumours (Kumar et al., 2021). Some Patents have been filed as a consequence of bioactive compounds presents in Mammea americana extract, this compounds have presented antibacterial and pharmaceutical activity as anticancer. Other uses correspond to cosmetic compositions and coumarin-type phytoconstituents with potential insecticidal and trypanocidal activity have also been isolated (Lemus et al., 2021).
The current traditional use for some of the most promising extract tested in this study, shows the health safety of crude extracts.
For the species of the tabernaemontana genus, various studies have also been published on their medicinal properties and traditional uses. These pharmacological activities, including its anti-inflammatory, antibacterial and antioxidant potential, are attributed to its high content of alkaloids (Abubakar and Loh, 2016; Naidoo et al., 2021). Our findings demonstrate that voacangine isolated from T. cymosa may be a candidate for tests in controlled environments, this is support by the ADMET data recently published where it was estimated through FAF-Drugs evaluation that voacangine do not possess toxic chemical groups in its structure, indicating a low rick to humans; additionally, computational calculations of other indole alkaloids identified in the T. cymosa extract against human proteins and toxicity assessment using C. elegans did not showed adverse effect to non-target organism (Oliveros-Díaz et al., 2021; Pájaro-González et al., 2022a).
Overall, our study demonstrated the potential of the Colombian Caribbean region flora as a host of bioactive plants against important vectors such as the A. aegypti mosquito. Through the results reported during this investigation, the extracts of Tabernaemontana cymosa Jacq. and Mammea americana L. species were classified as promising for larvicidal compound isolation. Although the aforementioned extracts have a variety of metabolites in their composition, we were able to correlate the existing literature and the results of our studies, identifying the alkaloid voacangine from T. cymosa as the main responsible for the larvicidal activity of these plant and its low risk to human poppulation. Our results suggest that the extracts are very toxic against accuatic organisms at 200 ppm, however, the potential use of isolated compounds in controlled environments such mosquito breeding sites and the application of sublethal concentrations could favor the field use of natural products without damaging the surrounding biota or humans. Further studies are needed to discover sublethal effects of bioactive extracts and compounds on Aedes aegypti larvae.
The major achieve of this research consisted in the isolation of the active principle of the plant T. cymosa, for being highly active against A. aegypti mosquito larvae showing a great degree of selectivity respect to human models. The indole alkaloid voacangine proved to be a promising compound for the development of bioinsecticides that can be applied in artifitial containers with stored water, which are ideal for mosquito breeding sites. Future studies focused on the fractionation of M. americana are necessary for the isolation of its active principles.
5 Conclusion
The Colombian flora is a potential source to seek larvicidal compounds against Aedes aegypti and other vectors. We report 16 plant extracts with the capacity to eliminate more than the 80 % of a population of larvae, being the ethanol extract of the seeds of T. cymosa the most active one with a LC50 value of 25 ppm with a regular Selectivity index (SI = 1.09) related to the guppy fish. In addition, its active principle, the alkaloid voacangine, is a potential alternative for the control of diseases transmitting vector Aedes aegypti, since it obtained an LC50 of 5.13 ppm, being one of the most promising compounds recently reported with very high selectivity against C. elegans nematode (SI greater than 40) and low toxicity for human models. Therefore, we conclude that the isolated compounds are a better alternative for the field control of A. aegypti larvae since the crude extract has a wide variety of compounds that can cause damage to non-target organisms. On the other hand, Mammea americana seeds extract was also highly toxic to the larvae with a LC50 value of 38.58 ppm, however, it showed toxicity against the model organism for extracts. Finally, subsequent studies with a bio-directed fractionation could help identify the active principles of each of the active plants found in this study and their ecotoxicological characteristics.
Acknowledgements
We acknowledge the University of Cartagena, as they provided the facilities for the experiments reported in this work. This investigation also received financial support from COLCIENCIAS grant No. 1107-519-28634.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Screening and evaluation of different algal extracts and prospects for controlling the disease vector mosquito Culex pipiens L. Saudi J. Biol. Sci.. 2022;29:933-940.
- [CrossRef] [Google Scholar]
- A review on ethnobotany, pharmacology and phytochemistry of Tabernaemontana corymbosa. J. Pharm. Pharmacol.. 2016;68:423-432.
- [CrossRef] [Google Scholar]
- Alkaloids and other compounds from seeds of Tabernaemontana cymosa. Phytochemistry. 1997;45:325-335.
- [CrossRef] [Google Scholar]
- Molecular characterization, gas chromatography mass spectrometry analysis, phytochemical screening and insecticidal activities of ethanol extract of Lentinus squarrosulus against Aedes aegypti (Linnaeus) Mol. Biol. Rep.. 2021;48:41-55.
- [CrossRef] [Google Scholar]
- Mosquitocidal and repellent activities of essential oils against Culex pipiens L. Entomol. Res.. 2021;50:182-188.
- [Google Scholar]
- Potential for integrated control of Culex quinquefasciatus (Diptera: Culicidae) using larvicides and guppies. Biol. Control. 2015;81:31-36.
- [CrossRef] [Google Scholar]
- A critical assessment of mosquito control and the influence of climate change on mosquito-borne disease epidemics. Environ., Develop. Sustain.: Multidiscip. Approach Theor. Pract. Sustai. Develop.. 2022;24:8900-8929.
- [Google Scholar]
- Anoopkumar, A.N., E M, A., Sudhikumar, A., 2020. Exploring the mode of action of isolated bioactive compounds by induced reactive oxygen species generation in Aedes aegypti: a microbes based double-edged weapon to fight against Arboviral diseases. International Journal of Tropical Insect Science 40. https://doi.org/10.1007/s42690-020-00104-z
- Larvicidal activity of fractions of Sphaeranthus indicus Linnaeus (Asteraceae) ethyl acetate whole plant extract against Aedes aegypti Linnaeus 1762, Anopheles stephensi Liston 1901 and Culex quinquefasciatus Say 1823 (Diptera: Culicidae) Int. J. Mosq. Res.. 2016;3:18-30.
- [Google Scholar]
- Caenorhabditis elegans as a model organism to evaluate the antioxidant effects of phytochemicals. Molecules. 2020;25:3194.
- [CrossRef] [Google Scholar]
- Chemical composition and repellent activity of native plants essential oils against dengue mosquito. Aedes aegypti. Industrial Crops Prod.. 2019;140:111609
- [CrossRef] [Google Scholar]
- Balachandran, C., Anbalagan, S., Kandeepan, C., Arun Nagendran, N., Jayakumar, M., Fathi Abd_Allah, E., Alqarawi, A.A., Hashem, A., Baskar, K., 2021. Molecular docking studies of natural alkaloids as acetylcholinesterase (AChE1) inhibitors in Aedes aegypti. Journal of Asia-Pacific Entomology 24, 645–652. https://doi.org/10.1016/j.aspen.2021.05.011
- Tamizaje fitoquímico preliminar de especies de plantas promisorias de la costa atlántica colombiana. Rev. Cubana Plant. Med.. 2013;18:619-631.
- [Google Scholar]
- Larvicidal activity and in silico studies of cinnamic acid derivatives against Aedes aegypti (Diptera: Culicidae) Bioorg. Med. Chem.. 2021;44:116299
- [CrossRef] [Google Scholar]
- Increased accumulation of isoflavonoids in common bean (Phaseolus vulgaris L.) tissues treated with 1-oxo-indane-4-carboxylic acid derivatives. Biotechnol. Rep. (Amst.). 2021;29:e00601.
- [Google Scholar]
- Carreño Otero, A.L., Palacio-Cortés, A.M., Navarro-Silva, M.A., Kouznetsov, V.V., Duque L., J.E., 2018. Behavior of detoxifying enzymes of Aedes aegypti exposed to girgensohnine alkaloid analog and Cymbopogon flexuosus essential oil. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology 204, 14–25. https://doi.org/10.1016/j.cbpc.2017.11.002
- Insecticidal and repellent activity of several plant-derived essential oils against Aedes aegypti. J. Am. Mosq. Control Assoc.. 2017;33:25-35.
- [CrossRef] [Google Scholar]
- Botanical essential oils and uses as mosquitocides and repellents against dengue. Environ. Int.. 2018;113:214-230.
- [CrossRef] [Google Scholar]
- Volatile toxin of Limonia acidissima (L.) produced larvicidal, developmental, repellent, and adulticidal toxicity effects on Aedes aegypti (L.) Toxin. Rev.. 2022;41:119-128.
- [CrossRef] [Google Scholar]
- Annona muricata: a comprehensive review on its traditional medicinal uses, phytochemicals, pharmacological activities, mechanisms of action and toxicity. Arabian J. Chem.. 2018;11:662-691.
- [CrossRef] [Google Scholar]
- Costa, E.V., Dutra, L.M., de Jesus, H.C.R., de Lima Nogueira, P.C., de Souza Moraes, V.R., Salvador, M.J., de Holanda Cavalcanti, S.C., dos Santos, R.L.C., do Nacimento Prata, A.P., 2011. Chemical Composition and Antioxidant, Antimicrobial, and Larvicidal Activities of the Essential Oils of Annona Salzmannii and A. pickelii (Annonaceae). Natural Product Communications 6, 1934578X1100600636. https://doi.org/10.1177/1934578X1100600636.
- Dereplication and isolation of larvicidal compounds from annonaceae species against Aedes aegypti. Rev. Bras. Farmacogn.. 2020;30:123-126.
- [CrossRef] [Google Scholar]
- Insecticide resistance status of Aedes aegypti and Aedes albopictus mosquitoes in Papua New Guinea. Parasites Vectors. 2019;12:333.
- [CrossRef] [Google Scholar]
- Hydroalcoholic extract of Haematoxylum brasiletto protects Caenorhabditis elegans from cadmium-induced toxicity. BMC Complement. Med. Ther.. 2022;22:184.
- [CrossRef] [Google Scholar]
- 6-(1,3-Dihydroxy-3-phenylpropylidene)-5-hydroxy-2,2,4-trimethylcyclohex-4-ene-1,3-dione. Molbank. 2015;2015:M853.
- [CrossRef] [Google Scholar]
- Dengvaxia controversy: impact on vaccine hesitancy. J. Glob. Health. 2018;8:020312
- [CrossRef] [Google Scholar]
- Foudjo Melacheu Laura, G., Mfotie Njoya, E., Jouda, J.-B., Wakeu Kweka, B.N., Djama Mbazoa, C., Wang, F., Seguin, E., Wandji, J., 2021. A new cytotoxic indole alkaloid from Tabernaemontana inconspicua stapf. Nat Prod Res 35, 1590–1595. https://doi.org/10.1080/14786419.2019.1628749.
- Antiviral effect of compounds derived from the seeds of Mammea americana and Tabernaemontana cymosa on Dengue and Chikungunya virus infections. BMC Complement. Altern. Med.. 2017;17:57.
- [CrossRef] [Google Scholar]
- Larvicidal activity of selected plant extracts and their combination against the mosquito vectors Culex quinquefasciatus and Aedes aegypti. Environ. Sci. Pollut. Res. Int.. 2018;25:9176-9185.
- [CrossRef] [Google Scholar]
- Genomics, GPCRs and new targets for the control of insect pests and vectors. Curr. Opin. Insect Sci.. 2018;30:99-106.
- [CrossRef] [Google Scholar]
- Accelerating invasion potential of disease vector Aedes aegypti under climate change. Nat. Commun.. 2020;11:2130.
- [CrossRef] [Google Scholar]
- Yellow Fever and Dengue. In: Domachowske J., ed. Introduction to ClinicAl Infectious DiseAses: A Problem-BAsed ApproAch. Cham: Springer International Publishing; 2019. p. :375-383.
- [CrossRef] [Google Scholar]
- Genetic variability of the Aedes aegypti (Diptera: Culicidae) mosquito in El Salvador, vector of dengue, yellow fever, chikungunya and Zika. Parasit. Vectors. 2018;11:637.
- [CrossRef] [Google Scholar]
- Resistance Status to Deltamethrin, Permethrin, and Temephos Along With Preliminary Resistance Mechanism in Aedes aegypti (Diptera: Culicidae) From Punjab, Pakistan. J. Med. Entomol.. 2019;56:1304-1311.
- [CrossRef] [Google Scholar]
- Plant anticancer agents VI: Isolation of voacangine, voacamine, and epivoacorine from Tabernaemontana arborea sap. J. Pharm. Sci.. 1978;67:271-272.
- [CrossRef] [Google Scholar]
- Kumar, M., Changan, S., Tomar, M., Prajapati, U., Saurabh, V., Hasan, M., Sasi, M., Maheshwari, C., Singh, S., Dhumal, S., Radha, Thakur, M., Punia, S., Satankar, V., Amarowicz, R., Mekhemar, M., 2021. Custard Apple (Annona squamosa L.) Leaves: Nutritional Composition, Phytochemical Profile, and Health-Promoting Biological Activities. Biomolecules 11, 614. https://doi.org/10.3390/biom11050614
- Mammea americana: a review of traditional uses, phytochemistry and biological activities. J. Herb. Med.. 2021;29:100466
- [CrossRef] [Google Scholar]
- Global risk mapping for major diseases transmitted by Aedes aegypti and Aedes albopictus. Int. J. Infect. Dis.. 2018;67:25-35.
- [CrossRef] [Google Scholar]
- Lima, M.E. de S., Bachur, T.P.R., Aragão, G.F., 2019. Guillain-Barre syndrome and its correlation with dengue, Zika and chikungunya viruses infection based on a literature review of reported cases in Brazil. Acta Trop 197, 105064. https://doi.org/10.1016/j.actatropica.2019.105064.
- Mosquito exposure and Chikungunya and Dengue infection among travelers during the Chikungunya outbreak in the Americas. Am. J. Trop. Med. Hyg.. 2017;96:903-912.
- [CrossRef] [Google Scholar]
- Mosquito larvicidal activity of alkaloids and limonoids derived from Evodia rutaecarpa unripe fruits against Aedes albopictus (Diptera: Culicidae) Parasitol. Res.. 2012;111:991-996.
- [CrossRef] [Google Scholar]
- Lopes Martins, R., Bruno Lobato Rodrigues, A., de Menezes Rabelo, É., Lima Santos, L., Barreto Brandão, L., Gomes Faustino, C., Luzia Ferreira Farias, A., Maria da Cunha Sá, D., de Castro Cantuária, P., Kardec Ribeiro Galardo, A., Susan Moreira da Silva de Almeida, S., 2021. Development of larvicide nanoemulsion from the essential oil of Aeollanthus suaveolens Mart. ex Spreng against Aedes aegypti, and its toxicity in non-target organism. Arabian Journal of Chemistry 14, 103148. https://doi.org/10.1016/j.arabjc.2021.103148
- Larvicidal Activity of Carbon Black against the Yellow Fever Mosquito Aedes aegypti. Insects. 2022;13:307.
- [CrossRef] [Google Scholar]
- Effect of crude acetone extract of seeds of Annona squamosa Linn. (Family: Annonaceae) on possible control potential against larvae of Culex quinquefasciatus Say. J. Entomol. Res.. 2000;24:141-146.
- [Google Scholar]
- Indole alkaloids inhibit zika and chikungunya virus infection in different cell lines. BMC Complement. Med. Ther.. 2021;21:216.
- [CrossRef] [Google Scholar]
- The Antiviral and Virucidal Activities of Voacangine and Structural Analogs Extracted from Tabernaemontana cymosa Depend on the Dengue Virus Strain. Plants (Basel). 2021;10:1280.
- [CrossRef] [Google Scholar]
- Detecting the impact of temperature on transmission of Zika, dengue, and chikungunya using mechanistic models. PLoS Negl.Trop. Dis.. 2017;11:e0005568.
- [Google Scholar]
- Mweya, C.N., Kimera, S.I., Kija, J.B., Mboera, L.E.G., 2013. Predicting distribution of Aedes aegypti and Culex pipiens complex, potential vectors of Rift Valley fever virus in relation to disease epidemics in East Africa. Infect Ecol Epidemiol 3, 10.3402/iee.v3i0.21748. https://doi.org/10.3402/iee.v3i0.21748.
- Major Bioactive Alkaloids and Biological Activities of Tabernaemontana Species (Apocynaceae) Plants. 2021;10:313.
- [CrossRef] [Google Scholar]
- Phytochemical profile and larvicidal activity of aqueous extract of Ocimum americanum against mosquito vectors. Appl. Nanosci. 2021
- [CrossRef] [Google Scholar]
- The Isolation of Rupicoline and Montanine, Two Pseudoindoxyl Alkaloids of Tabernaemontana Rupicola Benth. 1a. J. Org. Chem.. 1966;31:2265-2269.
- [CrossRef] [Google Scholar]
- Molecular human targets of bioactive alkaloid-type compounds from Tabernaemontana cymose Jacq. Molecules. 2021;26:3765.
- [CrossRef] [Google Scholar]
- The aqueous extract of Fridericia chica grown in northern Colombia ameliorates toxicity induced by Tergitol on Caenorhabditis elegans. Comp. Biochem. Physiol. C Toxicol. Pharmacol.. 2021;244:109026
- [CrossRef] [Google Scholar]
- In vitro and in silico antistaphylococcal activity of indole alkaloids isolated from Tabernaemontana cymosa Jacq (Apocynaceae) Sci. Pharm.. 2022;90:38.
- [CrossRef] [Google Scholar]
- Chapter 1 - A review of medicinal plants used as antimicrobials in Colombia. In: Chassagne F., ed. Medicinal Plants as Anti-Infectives. Academic Press; 2022. p. :3-57.
- [CrossRef] [Google Scholar]
- Mammea B/BA isolated from the seeds of Mammea americana L. (Calophyllaceae) is a potent inhibitor of methicillin-resistant Staphylococcus aureus. Front. Pharmacol.. 2022;13:826404
- [CrossRef] [Google Scholar]
- Plant extracts for developing mosquito larvicides: from laboratory to the field, with insights on the modes of action. Acta Trop.. 2019;193:236-271.
- [CrossRef] [Google Scholar]
- Pereira, P.S., França, S. de C., Oliveira, P.V.A. de, Breves, C.M. de S., Pereira, S.I.V., Sampaio, S.V., Nomizo, A., Dias, D.A., 2008. Chemical constituents from Tabernaemontana catharinensis root bark: a brief NMR review of indole alkaloids and in vitro cytotoxicity. Quím. Nova 31, 20–24. https://doi.org/10.1590/S0100-40422008000100004
- Plant-based larvicidal agents: an overview from 2000 to 2018. Exp. Parasitol.. 2019;199:92-103.
- [CrossRef] [Google Scholar]
- Antidengue potential of leaf extracts of Pavetta tomentosa and Tarenna asiatica (Rubiaceae) against dengue virus and its vector Aedes aegypti (Diptera: Culicidae) Heliyon. 2019;5:e02732.
- [CrossRef] [Google Scholar]
- Toxicity of the organophosphate insecticide sumithion to embryo and larvae of zebrafish. Toxicol. Rep.. 2020;7:317-323.
- [CrossRef] [Google Scholar]
- Culex quinquefasciatus Egg Membrane Alteration and Ovicidal Activity of Cipadessa baccifera (Roth) plant extracts compared to synthetic insect growth regulators. Res Rep Trop Med. 2019;10:145-151.
- [CrossRef] [Google Scholar]
- Protein carbonylation is a mediator in larvicidal mechanisms of Tabernaemontana cymosa ethanolic extract. J. King Saud Univ. – Sci.. 2019;31:464-471.
- [CrossRef] [Google Scholar]
- Integrated Aedes management for the control of Aedes-borne diseases. PLoS Negl.Trop. Dis.. 2018;12:e0006845.
- [CrossRef] [Google Scholar]
- The quantitative structure-insecticidal activity relationships from plant derived compounds against chikungunya and zika Aedes aegypti (Diptera:Culicidae) vector. Sci. Total Environ.. 2018;610–611:937-943.
- [CrossRef] [Google Scholar]
- Ecdysteroids: isolation, chemical transformations, and biological activity. Phytochem. Rev. 2022
- [CrossRef] [Google Scholar]
- Shad, A., 2017. Larvicidal efficacy of ethanolic extracts of Annona squamosa (Annonaceae) over the filarial vector, Culex quinquefasciatus Say (Culicidae).
- Existing and potential infection risk zones of yellow fever worldwide: a modelling analysis. Lancet Glob. Health. 2018;6:e270-e278.
- [CrossRef] [Google Scholar]
- Sistema de Vigilancia Nacional en Salud Pública (SIVIGILA), 2021. Weekly Epidemiological Bulletin: Week 48, 12-14. Colombian National Institute of Health, Colombia. Accessible at: chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://www.ins.gov.co/buscador-eventos/BoletinEpidemiologico/2021_Boletin_epidemiologico_semana_48.pdf [Consulted: 12-12-21].
- CYP-mediated resistance and cross-resistance to pyrethroids and organophosphates in Aedes aegypti in the presence and absence of kdr. Pestic. Biochem. Physiol.. 2019;160:119-126.
- [CrossRef] [Google Scholar]
- Evaluation of herbal essential oil as repellents against Aedes aegypti (L.) and Anopheles dirus Peyton & Harrion. Asian Pac. J. Trop. Biomed.. 2011;1:S124-S128.
- [CrossRef] [Google Scholar]
- Tavares, W.R., Barreto, M. do C., Seca, A.M.L., 2021. Aqueous and Ethanolic Plant Extracts as Bio-Insecticides—Establishing a Bridge between Raw Scientific Data and Practical Reality. Plants 10, 920. https://doi.org/10.3390/plants10050920
- The hydroxyindolenine derivative of voacangine, a new indole alkaloid from Voacanga africana. Tetrahedron. 1968;24:4223-4231.
- [CrossRef] [Google Scholar]
- Mosquito-borne diseases: advances in modelling climate-change impacts. Trends Parasitol.. 2018;34:227-245.
- [CrossRef] [Google Scholar]
- Aedes aegypti Larvicidal Sesquiterpene Alkaloids from Maytenus oblongata. J. Nat. Prod.. 2017;80:384-390.
- [CrossRef] [Google Scholar]
- AgBR1 antibodies delay lethal Aedes aegypti-borne West Nile virus infection in mice. npj Vaccines. 2019;4:1-4.
- [CrossRef] [Google Scholar]
- Larvicidal activity of Cnidium monnieri fruit coumarins and structurally related compounds against insecticide-susceptible and insecticide-resistant Culex pipiens pallens and Aedes aegypti. Pest. Manag. Sci.. 2012;68:1041-1047.
- [CrossRef] [Google Scholar]
- World Health Organization, 2005. Guidelines for laboratory and field testing of mosquito larvicides (No. WHO/CDS/WHOPES/GCDPP/2005.13). World Health Organization
- Studies on alkaloids from Ervatamia pandacaqui. Zhongguo Zhong Yao Za Zhi. 2018;43:1471-1475.
- [CrossRef] [Google Scholar]
- Contamination of neonicotinoid insecticides in soil-water-sediment systems of the urban and rural areas in a rapidly developing region: Guangzhou, South China. Environ. Int.. 2020;139:105719
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2022.104365.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







