Translate this page into:
Effect of gamma irradiation on chemical composition, antioxidant activity, antibacterial activity, shelf life, and cytotoxicity in the peels of two mango varieties grown in Bangladesh
⁎Corresponding authors. saleh@ru.ac.bd (Md. Abu Saleh), malakhan_07@yahoo.com (Mala Khan), szaman@ru.ac.bd (Shahriar Zaman)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Mango (Mangifera indica) is perhaps the most notable sound natural product, however short-lived. A few synthetic compounds like calcium carbide, pesticides, formalin and calcium-based arrangements have been utilizing for its protection, notwithstanding, those synthetic compounds are perilous for our human wellbeing. Besides, mango strip is a food-handling by-product that is discarded since it isn't economically reasonable. For this study, the nutritional, antibacterial, and antioxidant properties of peels from two mango cultivars (Amrapali, and BARI-11) were analyzed with four doses (0.5, 1.0, 1.5, and 2 kGy) of gamma irradiation. The impacts of radiation on storage quality and life span of usability were likewise investigated. Mango peels were found to have many phytochemicals. Be that as it may, radiation altogether impacted tested parameters like fat, protein, carb, fiber, sugar, energy, soluble solids, etc. in both of the mango peels. On the other hand, the peel samples showed 50% DPPH scavenging activity at varied dosages. Moreover, samples exposed to 1.5 kGy of radiation showed higher antioxidant activity. At a radiation dose of 1.5 kGy, amrapali peel proved to be less hazardous to brine shrimp lethality assay than other samples. The widest inhibitory zones against Pseudomonas sp. were obtained at a dosage of 150 µg/disc. After two weeks of storage, the 1.5 kGy radiation dosage left the mangoes' texture firm, indicating that it was extending the fruit's shelf life by greatly reducing the microbial attack. Thus mangoes could be preserved using radiation without significantly affecting their nutritional value.
Keywords
Mango peels
Irradiation
Antioxidant properties
Antibacterial activities
Cytotoxicity
Nutritional qualities
1 Introduction
The nutritious value, distinct flavor, delectable and healthy taste, and wholesomeness of mangoes give it its rank in the world (Saleem Dar et al., 2015). Mangoes are now grown commercially in over 103 countries throughout the world, and output is expanding year after year due to rising consumer demand. Asian nations produce over 77 percent of the world's mangoes, while the America and Africa contribute 13 percent and 9 percent, respectively (Jahurul et al., 2015). The bulk of mango output is consumed fresh, with just approximately 1–2 percent being processed into products such as juices, nectars, jams, jelly powders, fruit bars, flakes, and dried fruits. (Beyene & Araya, 2015). Mango varieties include Fozli, Rani Pasand, Gopal Vogue, Khirshapat (himshagor), Langra, Amropali, Gouramoti, Haribhanga, BARI-4, BARI-11, Katimon, and others are grown in Bangladesh. Recently, it has been revealed that mango agro-industrial byproducts are a source of potent compounds that could be consumed by humans. 35–60% of the peel and seed from mango processing are produced. Phenolic compounds, carotenoids, tocopherols, and sterols are responsible for these mango by-products' antioxidant effects (Gómez-Caravaca et al., 2016) (Abdel-aty et al., 2018).
Peel, which is considered a by-product of industrial processing or consumption of the fruit, contributes approximately 15–20% of the fruit that is typically discarded as waste (Thambi et al., 2016). Mango peel is also high in vitamins C and E. Protease, peroxidase, polyphenol oxidase, xylanase, and amylase activity were found in both raw (green mangoes) and mature mango peels. (Mitra et al., 2013). Mango peel flour is now found in a wide range of foods, including noodles, bread, sponge cakes, biscuits, and other bakery items. It can deliver nutrients as well as color. The exploitation of biological wastes is of major importance since the sector is rapidly being obliged to find an alternate use for its residual matter, such as seed and peels, due to regulation and environmental concerns (Thambi et al., 2016). Mango peels have lately been studied for anti-diabetic, antiproliferative, and antioxidant effects, as well as for enhancing dietary fiber content in cookies (Mannepula et al., 2015). Natural products are well-known for their importance in medication development and chemical biology (Nair & Chanda, 2007a).
Fresh-cut fruit's microbial development has been minimized by the use of chemical preservatives, vacuum and modified environment packing, irradiation techniques, Pulsed electric field (PEF), and efficient temperature and moisture management. (Gasu et al., 2012). Some of these methods like PEF and harmful chemical preservatives can reduce the amount of flavor, color, and nutritional value (Zhang & Weng, 2014)(Noci et al., 2008). If used correctly, irradiation can be an efficient technique to minimize the occurrence of food-borne illnesses by destroying contaminating organisms such as bacteria, molds, and yeasts (Naresh et al., 2015). Recently, dosages of up to 3 kGy of gamma-irradiation in combination with cold storage conditions have been suggested for enhancing shelf life and postponing the establishment of gray mold (Botrytis cinerea) and Rhizopus soft rot in fresh strawberries (Panou et al., 2020).
An important global public health concern is the rise of harmful bacteria that are resistant to antibiotics (Peterson & Kaur, 2018). Numerous studies have demonstrated that plant extracts have an antibacterial impact against germs that cause food poisoning. Bacterial contamination, including Gram-negative bacteria like Salmonella typhi, Escherichia coli, and Pseudomonas aeruginosa, is responsible for the majority of cases of food poisoning (Mostafa et al., 2018). Nowadays, it is common practice to employ the brine shrimp lethality assay (also known as the fairy shrimp or Artemia salina) to determine how deadly a substance is (Sarah et al., 2017). To ascertain a plant extract's capacity for cytotoxicity, a quick, affordable, and simple Artemia salina (Brine shrimp) bioassay is used (Baravalia et al., 2012).
Mango peel was shown to be an effective flavoring matrix, with appreciated odor-active chemicals detected in greater concentrations than in the edible mango fraction. Because of the acknowledged sensory qualities of mango, this fact has stimulated the revalorization of mango peels as a source of natural flavorings, which may be regarded as an option for utilizing those by-products. During the peak season in Bangladesh, faulty mango handling, processing, and preservation techniques can result in huge losses (Oliver-Simancas et al., 2020). Processing and conserving mangoes properly at both the farmer and industrial levels could prevent the loss of the seasonal surplus. Several strategies have been developed over the years to help farmers utilize extra fruits during the busiest time of the year to prevent microbial contamination of their products (Kabir et al., 2017). The purpose of the current investigation was to assess the impact of gamma-ray on the nutritional value and other characteristics of mango peels as well as the self-life of mangoes.
2 Materials and methods
2.1 Sample collection
Fresh, green-yellowish, and free of bruises mango varieties named BARI-11 and Amrapali were collected from Ambagan of Chapainawabganj, Bangladesh during June and July respectively. The day after they were picked, the mangoes were transported to the Bangladesh Atomic Energy Commission in Savar, Dhaka. After washing and air-drying, the mangoes of each kind were then individually wrapped in the thin, aerated polythene bags.
2.2 Gamma irradiation
Mangoes were exposed to radiation at the Gamma Source Division of the Institute of Food and Radiation Biology (IFRB), Bangladesh Atomic Energy Commission (BAEC), Savar, Dhaka. Using Co-60 gamma cells, the whole mangos were exposed to doses of gamma radiation ranging from 0.5 kGy to 2 kGy. The dosage rate throughout the investigation was 5.811 kGy/hr. Some mangoes were left untreated as a control. The mangoes were then maintained in a humidity cabinet at 19 °C for a few days, both the irradiated and non-irradiated varieties.
2.3 Total microbial count and ripening evaluation
Total bacterial load was measured on nutrient agar following some modifications of the method described by Iqtedar et al., 2016. At first mangoes were washed in 100 ml of sterilized distilled water then mango was shaken continually for 5 min before being serially diluted according to ISO standards. On medium plates, aliquots of 0.1 ml from each diluted tube were distributed. The colony-forming unit/mL was used to compute the microbiological count. For each diluted solution, the statistics provided were the mean counts from three Petri dishes. Each control and irradiated sample received three duplicates. The ripening evaluation was conducted based on the color and texture of the mangoes stored at room temperature.
2.4 Sample preparation
The peels were removed with a sharp, sterilized knife on days 1–2 of storage. On average, 1 kg of mango yielded about 200 g of peel. Mango peels were homogenized with a clean grinder for some experiments. Then the peels were dried at room temperature for almost one month before being ground into powder for other measurements.
2.5 Measurement of fat content
Fat content was measured according to Onuh et al., 2017 using soxtec system. Thimbles were dried at 105 °C for two hours. 5 g of crushed samples were placed in each thimble. 60–70 ml of petroleum ether was added to the extraction beakers. A 3-fold replicate was performed for each sample. The thimbles were attached to the extraction column and the beakers were placed under the extraction columns. Thimbles were dipped in solvent and the machine started. Upon completion of the process, the extraction cups were removed and dried in an oven at 105 °C for 30 min to remove moisture. The thimbles were then cooled to room temperature in a desiccator and weighed.
Calculation: % Fat= (F-T/S) x100.
F = weight of cup + fat residue, T = weight of the empty cup, S = Sample weight.
2.6 Measurement of protein
The percentage of protein in mango peel samples was measured according to the AOAC 2001.11 procedure with slight modifications (International, 1970)(Thiex & Scientific, 2002). The digestion mixture (Na2SO4 + CuSO4) was prepared. Then 10 g digestion mixture was taken in each tube. A 2 g sample was taken in each tube except for 1 blank tube. 12 ml H2SO4 was added to each tube. The digestion block (DKL 12 Automatic Digestion Unit) was set up and turned on. The track of tubes was removed after turning the digestor. The tubes were set in an automatic distiller (UDK 159 Automatic Kjeldahl Nitrogen Protein Analyzer) one by one, and the data were collected. It shows the protein percentage in every sample.
2.7 Measurement of ash
At first, the crucibles were dried after washing and cooled in a desiccator and weighed. Then, 2 g of samples were taken in each crucible. After that, the crucibles were carefully placed in the muffle furnace for 4 h at 450 °C. The crucibles were removed from the furnace and cooled in a desiccator. After that, the crucibles were weighed again. The total amount of ash was calculated according to the following formula.
2.8 Measurement of moisture
This experiment was conducted by AOAC method (Horwitz, 1975). The test sample was mixed and ground into a suitable quantity to give sufficient ground material for replicate determination. 5 g of the test sample was weighed in a pre-weighted flat dish and the dish was dried at 100 °C for 3 h. Then the dish was removed, covered, cooled in a desiccator, and weighed. Then it was re-dried for 1 h and repeated until a change in weight between successive dryings at 1 h intervals were ≤ 2 mg. Again, the dish was removed and cooled in a desiccator and weighed.
Percentage of moisture = W2 - W1 /W2- W3 × 100.
Where; W1 is the initial weight of the empty dish, W2 is the weight of the dish + un-dried sample, and W3 is the weight of the dish + dried sample.
2.9 Determination of total sugar
Using a modified version of the method described by Sewwandi et al., 2020, mango peel samples were tested for total sugar content (Sewwandi et al., 2020). In this method, the sample reacts with Fehling's solution, an alkaline tartrate system, to decrease copper sulfate. A combination of Fehling's A and Fehling's B makes up Fehling's solution. Fehling's B is a mixture of a strong base, commonly sodium hydroxide, and a reducing sugar, typically potassium sodium tartrate. Fehling's A is a solution of copper (II) sulfate.
2.10 Dietary fiber determination
The total dietary fiber in the mango peel sample was estimated with the help of the method AOAC 930.10 with some modifications (International, 1970)(Ara et al., 2014). At first, 0.1 g homogenized mango peel samples were taken in each conical flask. For each sample, 6 replications were conducted. In the conical flask, we added 8 ml of concentrated HCl. Then 25 ml DI water was added to each flask. After that the conical flasks were heated in the water bath for 90 min at 80 °C. After removing the flasks from the water bath, they were cooled. Then 50 ml ethanol was added to the flasks. After that, they were kept for 1 h at room temperature. The flasks were heated for 30 min at 60 °C. Then the mixer was filtered with the glass crucible. The glass crucibles were dried at 105 °C for 2 h after completing the filtration. For each sample, two crucibles were used for the protein estimation and two were further used for the ash estimation among the previous crucibles. The dried sample in the crucible was taken out for the measurement of the protein with the help of sulfuric acid. Then the further process was conducted according to the protein estimation process of the AOAC method. For the ash estimation, the two crucibles were kept at 650 °C for 4 h. They were removed and weighed.
2.11 Determination of vitamin C (Ascorbic Acid)
By macerating the sample with stabilizing agents such as 20% metaphosphoric acid, the ascorbic acid level in fruits and vegetables can be determined with the method of AOAC 967.21(Horwitz, 1975) (Nielsen & Nielsen, 2017). 50 mg of sample was taken into a 100 ml volumetric flask, and 25 ml of 20% metaphosphoric acid was pipetted as a stabilizing agent and diluted to volume. 10 ml was pipetted in a small flask and 2.5 ml of acetone was added. Then it was titrated with an indophenol solution until a faint pink color persists for 15 s.
Ascorbic acid/g= (X-B) x(F/E) x(V/Y).
Where X = average ml for test solution titration,
B = average ml for test blank titration,
F = mg ascorbic acid equivalent to one ml indophenol standard solution,
E = no. of g.
V = volume initial test solution and.
Y = volume test solution titrated.
2.12 Total insoluble solid
5 Whatman filter papers were placed in an oven of 100 °C for 2 h. They were weighed and placed in a desiccator. Then 20 g samples were taken in a beaker, and about 20 ml of hot water was added and mixed thoroughly. The mixer was filtered by pre-weighed filter papers. Again, hot water was added to the remained mixer and filtered through the filter papers.
Then the filter papers were dried at 100 °C in a covered dish for two hours and then cooled in a desiccator and weighed. The total percentage of insoluble solids was calculated according to the following formula
2.13 Measurement of titratable acidity
The total titratable acidity (TTA) of a food sample is the quantity of acid or acids present in it. In this study, the procedure described by Islam et al. 2013 was used to determine the titratable acidity. Briefly, In an Erlenmeyer flask, 6 g samples were collected. Then 100 ml of water was added and thoroughly shaken with a shaker. A filter paper was used to filter the mixer. 5 ml of the filtrate was collected and placed in each of the conical flasks. The phenolphthalein indicator was then used to titrate against 0.1 N sodium hydroxide (NaOH). The results were expressed as a percentage of citric acid. It was calculated using the following formula.
2.14 Measurement of carbohydrate
The content of total carbohydrates in the mango peel samples was determined by the following equation (Ara et al., 2014):
Total Carbohydrate (%) = 100 - {Moisture (%) + Protein (%) + Fat (%) + Ash (%)}.
2.15 Measurement of energy
The gross food energy was estimated (Ara et al., 2014) using the value of total carbohydrate, protein, and fat in the samples with the equation written below: -.
Here, TC- Total carbohydrate.
TF-Total fat.
TP-Total protein.
2.16 Extraction of mango peels
For extraction, about 100 g powder of each peel was taken in 1 L conical flasks. Each sample was soaked in 500–600 ml of 95% methanol. The conical flasks with their contents were then sealed and kept on an orbital shaker for continuous shaking at 150 rpm for 2 days at 37 °C. The mixtures were then filtered through Whatman No.1 filter paper. Using a rotary evaporator, the methanolic extract of all two plants was evaporated at 55–60 °C temperature and a rotation speed of 160–180 rpm. After 30 min of the drying process, a slurry concentration was obtained, which was kept in a small vial for further drying. After 20–30 days the solvents were completely evaporated, and the extracts became ready for the experiment.
2.17 Antimicrobial activity assay
The bacterial strains used for this investigation were Pseudomonas sp., Escherichia coli, and Salmonella sp. following the protocol of disc diffusion method (Bauer, 1966), (Nair & Chanda, 2007b). 25 µg/mL, 50 µg/mL, 100 µg/mL and 150 µg/mL of extract were used in each discs. In this investigation, ready-made kanamycin 30 µg/disc was employed as a standard disc for comparison. Then, the dried crude extract discs and reference disc were carefully put on solidified LB agar plates seeded with the test bacterium using a pair of sterile forceps. The antibacterial activity of the test samples was measured after incubation by measuring the width of inhibitory zones in millimeters using a transparent scale.
2.18 Determination of MIC and MBC
Following the Broth dilution method, the minimum inhibitory concentration (MIC) was carried out (Reddy & Mangalekar, n.d.). In short, 1 to 6 tubes were labeled then BHI broth (0.5 ml) was added to tubes labeled 2 to 6. After that, 1 ml of the test material was taken in the first tube then 0.5 ml of the extract from the first tube was transferred to the second tube containing the BHI broth and 0.5 ml was serially transferred to the next tube, till the 6th tube. Thus, extract solutions were serially diluted and concentrations at the concentration of 200,100,50,25,12.5 mg/mL for Amrapali and BARI-11. The tubes were then inoculated with 0.1 ml of the respective bacterial cultures. The lowest concentration of the extract that completely inhibited the growth of the organisms was considered MIC.
MBC (Minimum Bactericidal Concentrations) was calculated with some modifications of G. Parvez, et.al.(Parvez et al., 2016). After removing 0.10 ml of bacterial suspension from the MIC tubes that did not show any growth, the MBC values were obtained. Then, subcultured into Mueller Hinton agar plates and incubated at 37 °C for 24 h. After incubation, the concentration at which no visible growth was seen was denoted as the MBC.
2.19 Antioxidant activity test
At first, the concentration at which BHT (a typical antioxidant) exhibited 50% DPPH scavenging capacity, which was the IC50 value for BHT to scavenge DPPH was calculated (Choi et al., 2000).
Four test tubes were therefore autoclaved and marked with concentrations 1, 2, 3, and control. From the stock solution of Mangifera indica peel extract, 10 µl, 20 µl, and 30 µl solutions were poured into the first three test tubes, respectively and the control test tube was empty. Then, 990 µl, 980 µl, 970 µl, and 1000 µl of the solvent (methanol) were added to the first, second, third, and control test tubes, respectively. As a result, the first three test tubes had extract concentrations of 50 µg, 100 µg, and 150 µg/ml, respectively and 1.5 ml of DPPH solution was added to each test tube. The absorbance of the solutions was measured at 519 nm using a spectrophotometer against a blank after 30 min of incubation and repeated three times.
The absorbance measurement, determination of the percentage of DPPH scavenging activity, and IC50 value of the extract were virtually identical to the BHT experimental protocol. However, while measuring absorbance with a spectrophotometer, a blank solution that included all reagents was employed except the extract.
2.20 Cytotoxicity test on brine shrimp
Cytotoxicity test of the M. indica peel was conducted by following the method of Meyer et al. (Meyer et al., 1982) with some modifications. Here, the extract concentrations in the test tubes were 50 µg, 100. µg, and 150 µg/ml, respectively. The percentage of mortality of brine shrimp nauplii at each concentration for each sample was estimated.
2.21 Statistical analysis
With three repetitions, the experimental design was randomized. One-way analysis of variance (ANOVA) in SPSS was used for analyzing the experimental data and Duncan's Multiple Range Test (DMRT) was used for comparing the significant differences between treatment means. GraphPad Prism 8 was used for preparing graphical figures.
3 Results
3.1 Total microbial count
Table 1 shows that the microbial load in all radiated samples was lower than the control after two weeks of observation. The microbial load in the Amrapali control was 6.8x105 cfu/ml in one week. In contrast, the 1.5 kGy and 2 kGy radiated Amrapali samples had 2.1x102 and 1.4x102 cfu/ml, respectively. The microbial counts in the BARI-11 mango control sample were 6.4x104 cfu/ml. This sample had a significantly greater microbial burden. The microbial load of 1.5 kGy radiated BARI-11 samples was 2.8x102 cfu/ml on week one, and 3.80x102 cfu/ml on week two (Table 1). Results represented as Mean of cfu/mL ± SD. Superscript a,b,c,d shows significant (p ≤ 0.05) difference between cfu/mL values of given doses.
Gamma Irradiation (kGy)
Microbial Load in Amrapali on Nutrient Agar (CFU/mL)
Microbial Load in BARI-11 on Nutrient Agar (CFU/mL)
Week 1
Week 2
Week 1
Week 2
Control
6.8x105 ± 0.52a
7.9x108 ± 0.6a
6.4x104 ± 1.15a
6.9x108 ± 0.64a
0.5
4.1x103 ± 0.85a
3.4x103 ± 0.7ab
3.8x103 ± 0.42a
4.1x104 ± 0.6d
1.0
3.8x103 ± 0.7a
4.2x103 ± 0.68ab
3.5x102 ± 0.45a
5.6x102 ± 1.5d
1.5
2.1x102 ± 0.4a
3.7x102 ± 0.95b
2.80x102 ± 0.55ab
3.80x102 ± 0.25b
2.0
1.4x102 ± 0.4a
2.1x103 ± 0.21a
1.5x102 ± 0.24a
4.5x103 ± 0.67b
3.2 Ripening evaluation
Tables 2 and 3 demonstrate the results of a two-week ripening study of irradiated and control mango samples. The texture of the control and treated samples (0.5 kGy, 1 kGy, 1.5 kGy, and 2.0 kGy) did not alter significantly in the first week. The variations between control and irradiated samples with low dose rates were not immediately apparent; however, after the mangoes were stored, the inconsistencies became apparent. The peels of all of the samples were green for the first week. The control mangoes began to ripen and soften after the second week, as seen by the yellowing of the peel color. After two weeks, the control mangoes were rotten. Mangoes dosed at 2.0 kGy were overly soft, but those dosed at 1.5 kGy had a stronger texture. When compared to BARI-11, Amrapali's changes happened at a faster rate during storage (Table 2 and Table 3).
Gamma Irradiation (kGy)
Ripening Evaluation (Amrapali)
Week 1
Week 2
Texture
Color
Texture
Color
Control
Firm
Yellowish
Over ripe
Yellow
0.5
Firm
Green
Less firm
Yellowish Green
1.0
Firm
Yellowish
Firm
Yellow
1.5
Firm
Green
Firm
Yellowish
2.0
Firm
Yellowish
Firm
Yellowish
Week 1
Week 2
Texture
Color
Texture
Color
Control
Firm
Green
Overripe
Yellowish
0.5
Firm
Green
Less firm
Yellowish Green
1.0
Firm
Green
Firm
Green
1.5
Firm
Green
Firm
Slightly yellow
2.0
Firm
Green
Soft
Slightly yellow
3.3 Total fat and protein content
On day 1, the BARI-11 peel control had the lowest fat level (0.35%) while the Amrapali peel sample with 2 kGy radiation had the highest fat content (0.50%). After 15 days, the fat content in the control samples of both mango cultivars was higher than the other radiated samples as the mangoes were over-ripped. The other samples with different radiation doses had little changes as radiation can delay their ripening (Fig. 1a and 1b).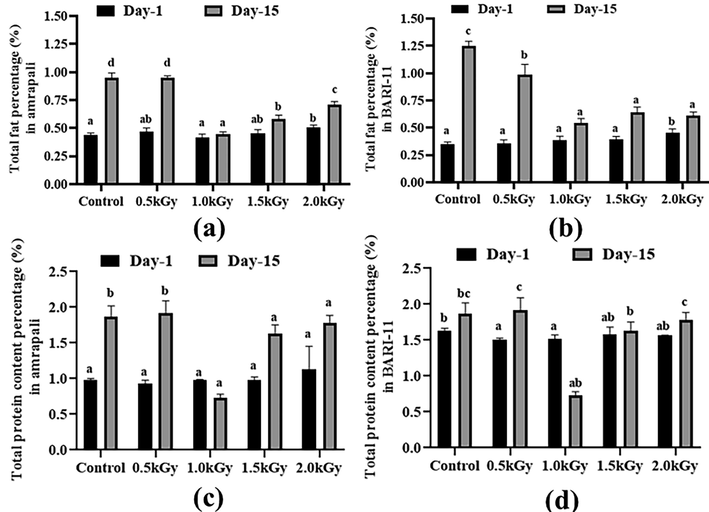
Total fat and protein content (%) of the two cultivars of Mangifera indica peel samples with different gamma irradiation doses on day 1 and day 15. (a) total fat percentage in Amrapali peel, (b) total fat percentage in BARI-11 peel, (c) total protein content in Amrapali peel, (d) total protein content in BARI-11 peel. Different letters indicate significant differences between the mean ± SD of treatments (n = 3) at a P < 0.05 significance level.
The BARI-11 mango with a 1.5 kGy dose had the highest protein concentration in the peel samples, while the Amrapali variety with a 1.0 kGy treatment had the lowest, at 0.93 percent. The protein levels in the control and 0.5 kGy radiated samples were dramatically changed after 15 days. However, the total amount rose overall, and BARI-11 mango peels showed some substantial alterations (Fig. 1c and 1d).
3.4 Total ash and moisture content
Without any doses, the BARI-11 sample had the highest percentage of ash (0.69%). After 15 days, the ash content of all samples rose, with the control samples showing the greatest increase. With no radiation, the highest concentration was found in the BARI-11 peel sample. The ash content of Amrapali mango peels did not alter significantly, whereas BARI-11 mango peels showed some substantial changes (Fig. 2a and 2b).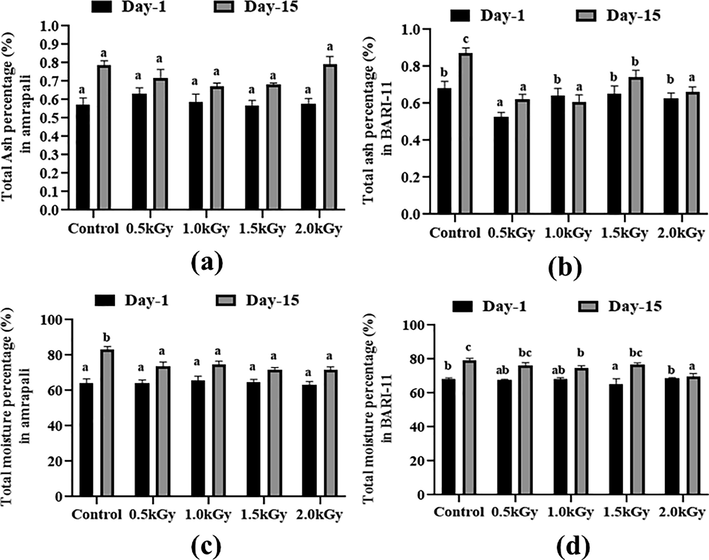
Total ash and a moisture percentage of the two cultivars of Mangifera indica peel samples with different radiation doses on day 1 and day 15. (a) total ash percentage in Amrapali mango peel, (b) total ash percentage in BARI-11 mango peel, (c) total moisture percentage in amrapali mango peel, (d) total moisture percentage in BARI-11 mango peel. Different letters indicate significant differences between the mean ± SD of treatments (n = 3) at a P < 0.05 significance level.
However, the average moisture level of all peel samples of Amrapali mango peel was the same. On day one, the BARI-11 peel had the maximum moisture content (68.43%). After 15 days, the moisture content of all samples increased. The Amrapali control type had the most, while BARI-11 with a 1.5 kGy dose had the least (Fig. 2c and 2d).
3.5 Total sugar and dietary fiber content (%)
The peel samples had a very low sugar content. This was in the range of 0.1 to 0.2 %. The lowest level of 0.1 % was found in the Amrapali peel sample, which received a 1.5 kGy dosage of radiation. After 15 days, all samples revealed a considerable rise in sugar content. The sugar content of the non-radiated samples was higher than that of the radiated samples (Fig. 3a and 3b).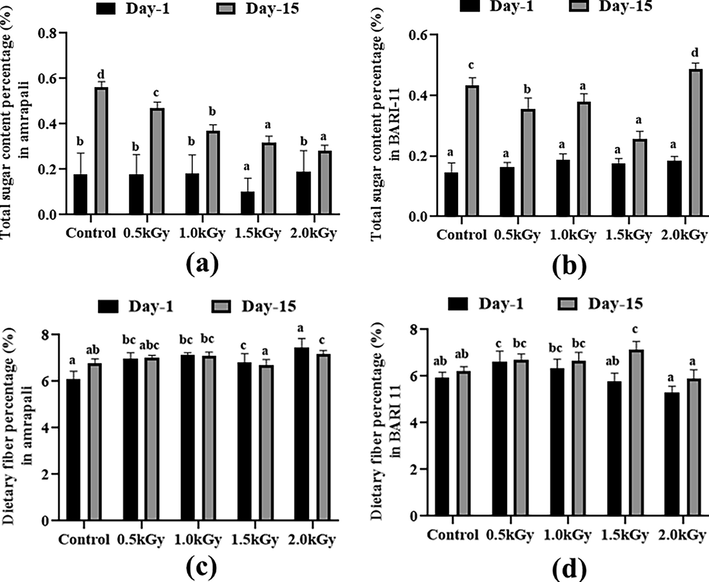
Total sugar and dietary fiber content (%) of the two cultivars of Mangifera indica peel samples with different radiation doses on day 1 and day 15. (a) total sugar percentage in Amrapali mango peel, (b) total sugar percentage in BARI-11 mango peel, (c) Dietary fiber percentage in Amrapali mango peel, (d) Dietary fiber percentage in BARI-11 mango peel. Different letters indicate significant differences between the mean ± SD of treatments (n = 3) at a P < 0.05 significance level.
On day 1, the Amrapali peel sample reached the highest value of 7.44 % dietary fiber with a dosing rate of 2 kGy. In contrast, with a 2 kGy radiation dosage, the BARI-11 mango peels showed a low value of 5.31 %. The amount of dietary fiber in peel samples differed significantly after 15 days. The highest percentage (7.16%) was found in Amrapali mango peels with a 2 kGy dosage rate (Fig. 3c and 3d).
3.6 Vitamin C and total soluble solids (TSS) content
On day 1, the Amrapali and BARI-11 control peels exhibited the greatest levels of ascorbic acid, with 48.12 mg/100 g and 46.07 mg/100 g, respectively. Ascorbic acid was detected in trace levels in the 2 kGy samples. After 15 days, the level of vitamin C in all types of samples had substantially decreased (Fig. 4a and 4b).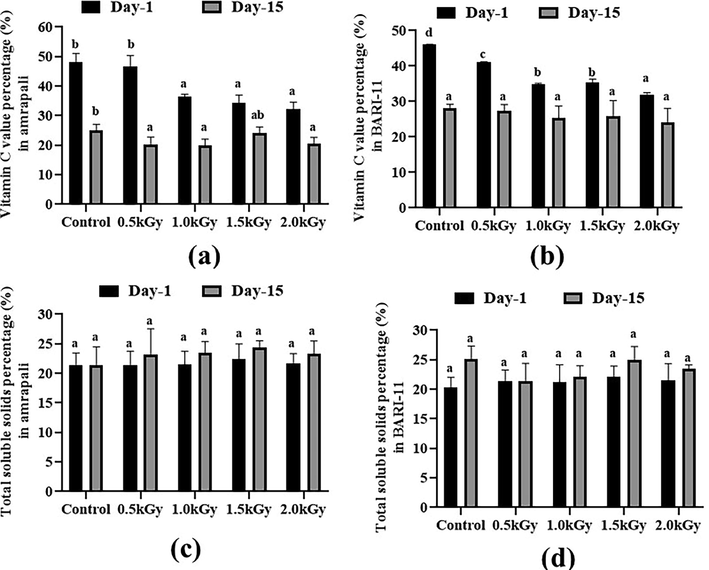
Vitamin C and Total soluble solids content (%) of the two cultivars of Mangifera indica peel samples with different radiation doses on day 1 and day 15. (a) Vitamin C percentage in Amrapali mango peel, (b) Vitamin C percentage in BARI-11 mango peel, (c) Total soluble solids percentage in Amrapali mango peel, (d) Total soluble solids percentage in BARI-11 mango peel. Different letters indicate significant differences between the mean ± SD of treatments (n = 3) at a P < 0.05 significance level.
The maximum value (22.39%) of TSS in the Amrapali peel was estimated at 1.5 kGy radiation dose on day 1. The BARI-11 mango peels with a 1.5 kGy dose rate had a slightly higher rate of total soluble solids than others. The amount of total soluble solids in all the peel samples of two mango cultivars increased non-significantly after 15 days (Fig. 4c and 4d).
3.7 Titratable acidity and total insoluble solids
The Amrapali control sample had a titratable acidity of 0.295 %. On day 1, the other results in all peel samples were more or less comparable, and no significant changes were observed. The titratable acidity content in almost all of the samples increased after 15 days. The maximum TA was found in an Amrapali peel sample with a 1.5 kGy dose, while 0.48 % titratable acidity was found in a BARI-11 peel sample with a 1 kGy dose rate (Fig. 5a and 5b).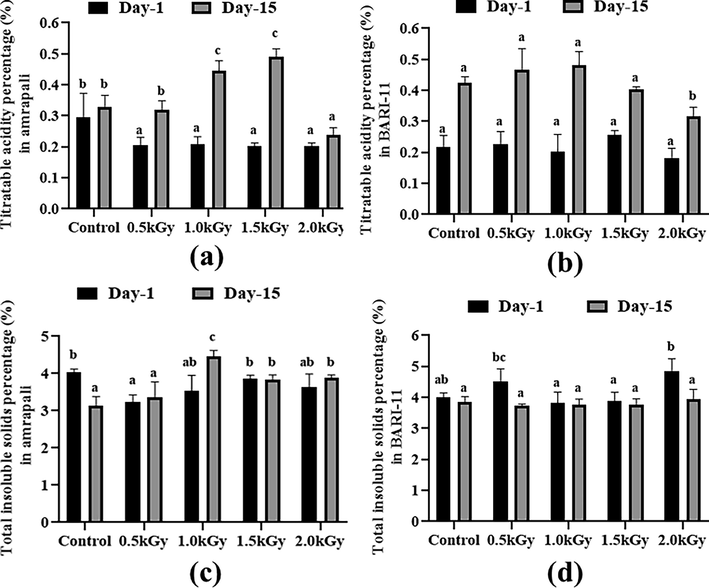
Titratable acidity and total insoluble solids (%) of the two cultivars of Mangifera indica peel samples with different radiation doses on day 1 and day 15. (a) titratable acidity percentage in Amrapali mango peel, (b) titratable acidity percentage in BARI-11 mango peel, (c) total insoluble solid percentage in Amrapali mango peel, (d) total insoluble solid percentage in BARI-11 mango peel. Different letters indicate significant differences between the mean ± SD of treatments (n = 3) at a P < 0.05 significance level.
In the case of total insoluble solids, the BARI-11 had the highest value of 4.86 percent with a dose rate of 2 kGy. On day 1, substantial alterations were observed in the Amrapali mango peel samples with radiation doses of 0.5, 1.0, 1.5, and 2.0 kGy. After 15 days, some samples showed a drop in this metric, while others showed an increase. After two weeks, there were no notable changes in BARI-11 mango peels (Fig. 5c and 5d).
3.8 Carbohydrate and energy content
On day 1, the Amrapali control type had the highest carbohydrate content (34.71%), while BARI-11 with a 1.5 kGy radiation dosage had the highest carbohydrate content among the BARI-11 samples. With a 1 kGy radiation exposure, the greatest quantity was seen in the Amrapali peel sample after 15 days. Amrapali peel samples showed no significant changes on day 1 or day 15, however, BARI-11 peel samples showed some carbohydrate content alterations after two weeks (Fig. 6a and 6b).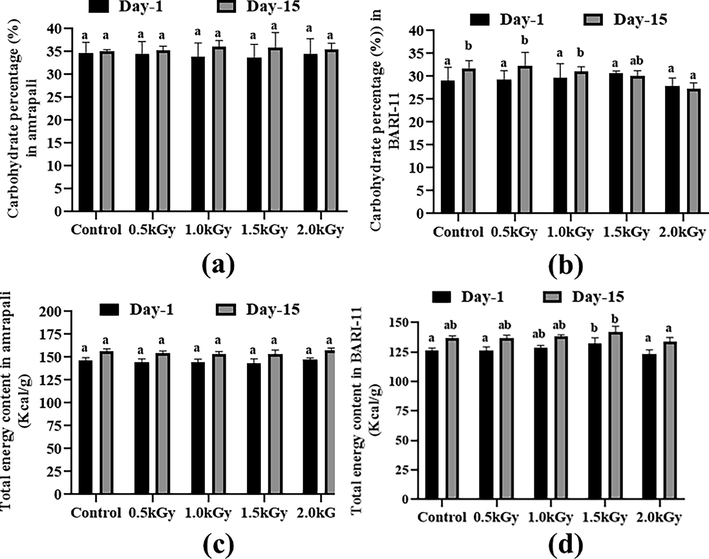
Carbohydrate (%) and energy content (Kcal/g) of the two cultivars of Mangifera indica peel samples with different radiation doses on day 1 and day 15. (a) carbohydrate content (%) in Amrapali mango peel, (b) carbohydrate content (%) in BARI-11 mango peel, (c) total energy content (Kcal/g) in Amrapali mango peel, (d) total energy content (Kcal/g) in BARI-11 mango peel. Different letters indicate significant differences between the mean ± SD of treatments (n = 3) at a P < 0.05 significance level.
The Amrapali peel with a 2 kGy dosage rate had a higher energy content of 147.31 kcal/g on day 1, but there were no significant changes after two weeks. In the case of BARI-11 mango peels figure depicts the fluctuating energy levels of several samples with varied dosages on day 1 and after 15 days of storage (Fig. 6c and 6d).
3.9 Antimicrobial activity
Amrapali control mango peel extracts demonstrated antibacterial activity against Pseudomonas sp. at 100 and 150 µg/disc dosages. However, they were ineffective against Escherichia coli and Salmonella sp. Antibacterial activity against Pseudomonas sp. was also observed in the Amrapali 0.5 kGy dosed sample. Amrapali peel extract had antibacterial activity against Pseudomonas sp. and Salmonella sp. after being exposed to a 1.0 kGy dose of radiation. The sample dosage rate was 150 µg/disc in this case. At a sample dose of 150 µg/disc, Pseudomonas sp. and Salmonella sp. showed susceptibility to Amrapali 2.0 kGy dosed peel extracts (Table 4). Note: Zone size < 10 mm = Resistance (R), Zone size 10–15 mm = Intermediate resistance (I), Zone size > 15 mm = Sensitive (S). Different letters indicate significant differences between mean ± SD of treatments (n = 3) at a P < 0.05 significance level.
Radiation Doses
Sample Dose (µg/disc)
Mean Value of Inhibition Zone (mm)
Pseudomonas sp.
Escherichia coli
Salmonella sp.
Amrapali Control
25
12.23 ± 0.13b
12.23 ± 0.147d
8.45 ± 0.023b
50
13.67 ± 0.153a
9.89 ±.025a
8 ± 0.0a
100
15.45 ± 0.208c
10.13 ± 0.01b
9.5 ± 0.017c
150
15.1 ± 0.02d
11.89 ± 0.015c
11.01 ± 0.01d
Kanamycin 30 µg
26.32 ± 0.01e
25.13 ±.017e
25.53 ± 0.017e
Amrapali 0.5 kGy
25
12.23 ± 0.017a
8 ± 0.0a
8 ± 0.0a
50
12.33 ± 0.015b
8 ± 0.01a
8 ± 0.0a
100
15.54 ± 0.01d
8 ± 0.0a
8.23 ± 0.021b
150
15.1 ± 0.015c
8.78 ± 0.012b
8.27 ± 0.01c
Kanamycin 30 µg
24.12 ± 0.017e
22.24 ± 0.006c
21.23 ± 0.015d
Amrapali 1.0 kGy
25
8 ± 0.0a
9 ± 0.006a
8 ± 0.0a
50
9 ± 0.02b
9.89 ± 0.035b
8 ± 0.0b
100
9.24 ± 0.006c
12 ± 0.015c
9 ± 0.0c
150
15.57 ± 0.015d
13.1 ± 0.071d
15.78 ± 0.006d
Kanamycin 30 µg
24.13 ± 0.017e
22.56 ± 0.006e
23.56 ± 0.015e
Amrapali 1.5 kGy
25
9.67 ± 0.0152a
8 ± 0.0a
8 ± 0.0a
50
10 ± 0.01b
8 ± 0.0a
8 ± 0.0a
100
15.2 ± 0.012c
8 ± 0.0a
8 ± 0.0a
150
15.4 ± 0.012d
8 ± 0.0a
8 ± 0.0a
Kanamycin 30 µg
24.58 ± 0.006e
22.23 ± 0.012b
21.23 ± 0.015b
Amrapali 2.0 kGy
25
8 ± 0.0a
9 ± 0.012a
9 ± 0.0a
50
9 ± 0.012b
9 ± 0.015b
9 ± 0.0a
100
12.1 ± 0.025c
11.44 ± 0.015c
11.12 ± 0.01b
150
15 ± 0.01d
11.88 ± 0.017d
15.1 ± 0.015c
Kanamycin 30 µg
24.2 ± 0.01e
23.56 ± 0.012e
22.23 ± 0.015d
Three bacterial strains were found to be resistant to the peel sample that had not been exposed to radiation. BARI-11 with a 0.5 kGy radiation dose exhibited antibacterial action against Pseudomonas sp. at a dose of 150 µg/disc, but not against Escherichia coli or Salmonella sp. Pseudomonas sp. was susceptible to BARI-11 (1.0 kGy) peel extract at a treatment rate of 150 µg/disc, but Salmonella sp. showed intermediate resistance. The BARI-11 (2.0 kGy) sample had antibacterial action against Pseudomonas sp. at a dosage rate of 150 µg/disc (Table 5). Note: Zone size < 10 mm = Resistance (R), Zone size 10–15 mm = Intermediate resistance (I), Zone size > 15 mm = Sensitive (S). Different letters indicate significant differences between mean ± SD of treatments (n = 3) at a P < 0.05 significance level.
Radiation Doses
Sample Dose (µg/disc)
Mean Value of Inhibition Zone (mm)
Pseudomonas sp.
Escherichia coli
Salmonella sp.
BARI-11 Control
25
8 ± 0.0a
8 ± 0.0a
8.78 ± 0.017a
50
9 ± 0.0b
8 ± 0.0a
9.21 ± 0.006b
100
10.13 ± 0.006c
8 ± 0.0a
9.89 ± 0.006c
150
11.67 ± 0.015d
8 ± 0.01a
10.78 ± 0.023d
Kanamycin 30 µg
23.13 ± 0.006e
22.11 ± 0.021b
21.43 ± 0.017e
BARI-11 0.5 kGy
25
8 ± 0.0a
8 ± 0.0a
9.11 ± 0.006a
50
9.12 ± 0.006b
8.77 ± 0.006b
9 ± 0.0b
100
11.66 ± 0.006c
9 ± 0.0c
9.2 ± 0.01c
150
15.1 ± 0.006d
9.12 ± 0.01d
11 ± 0.021d
Kanamycin 30 µg
23.32 ± 0.017e
21.89 ± 0.006e
22.10 ± 0.01e
BARI-11 1.0 kGy
25
8.99 ± 0.015a
8 ± 0.0a
9.0 ± 0.021a
50
9 ± 0.01b
8 ± 0.0a
9.34 ± 0.012b
100
11 ± 0.025c
9 ± 0.015b
10.78 ± 0.023c
150
15.1 ± 0.01d
9 ± 0.0b
10.69 ± 0.015d
Kanamycin 30 µg
23.32 ± 0.012e
21.56 ± 0.017c
21.88 ± 0.006e
BARI-11 1.5 kGy
25
8 ± 0.0a
8 ± 0.0a
8 ± 0.0a
50
8.88 ± 0.017b
8 ± 0.0a
9 ± 0.0b
100
9.1 ± 0.006c
8 ± 0.0a
10.12 ± 0.006c
150
11.56 ± 0.006d
9 ± 0.021b
10.78 ± 0.005d
Kanamycin 30 µg
23.34 ± 0.005e
21.78 ± 0.005c
21.90 ± 0.015e
BARI-11 2.0 kGy
25
8 ± 0.0a
8 ± 0.0a
9 ± 0.0a
50
8.4 ± 0.015b
8 ± 0.0a
9.21 ± 0.015b
100
11.44 ± 0.021c
8 ± 0.0a
10 ± 0.015c
150
15.85 ± 0.012d
8 ± 0.0a
10.98 ± 0.006d
Kanamycin 30 µg
22.98 ± 0.012e
21.88 ± 0.006b
21.90 ± 0.015e
3.10 Determination of MIC and MBC
In MIC and MBC tests, it was observed that both of the extracts inhibited bacterial growth. The ranges of MIC and MBC in most of the cases were between 50 and 100 µg/ml for Amrapali and BARI-11 and in all cases, MBC values were higher or equal to the MIC values. All of the MIC and MBC values of the extracts are presented in (Table 6).
Radiation Doses
Sample Dose (µg/disc)
Amrapali
BARI-11
MIC
MBC
MIC
MBC
Control
Pseudomonas sp.
50
100
100
100
Escherichia coli
50
100
50
100
Salmonella sp.
100
100
50
100
0.5 kGy
Pseudomonas sp.
50
50
50
100
Escherichia coli
50
100
50
50
Salmonella sp.
100
100
100
100
1.0 kGy
Pseudomonas sp.
100
200
100
100
Escherichia coli
100
100
50
100
Salmonella sp.
100
100
100
100
1.5 kGy
Pseudomonas sp.
100
100
50
100
Escherichia coli
50
100
50
100
Salmonella sp.
100
100
100
200
2.0 kGy
Pseudomonas sp.
100
100
50
100
Escherichia coli
50
100
50
100
Salmonella sp.
50
100
100
100
Kanamycin
Pseudomonas sp.
25
25
12.5
25
Escherichia coli
12.5
25
25
25
Salmonella sp.
25
25
12.5
25
3.11 Antioxidant activity and cytotoxicity test
This study looked at the antioxidant impact (DPPH free radical scavenging activity) of BHT standard at three concentrations: 50 µg, 100 µg, and 150 µg/ml. BHT had significant antioxidant activity, as evidenced by its IC50 value of 36 µg/ml. Antioxidant activity was found in all three mango peel extracts (radiated and non-radiated samples). They had the maximum antioxidant potentiality at a dosing rate of 1.5 kGy. The peel extract of the BARI-11 mango variety had the maximum activity, with a radiation dose of 1.5 kGy, and some significant changes were recorded among the replicates (Fig. 7a).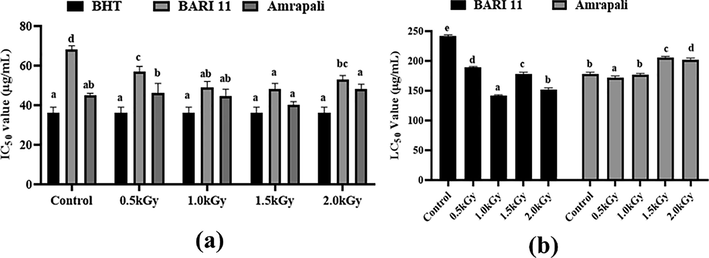
(a) Effect of gamma radiation on antioxidant activity of two varieties of Mangifera indica peels (Amrapali and BARI-11) (b) Cytotoxic activity of Mangifera indica peel extracts (Amrapali and BARI-11) at different concentrations against Artemia. Different letters indicate significant differences between the mean ± SD of treatments (n = 3) at a P < 0.05 significance level.
The cytotoxic impact of Mangifera indica peel extract was assessed using the Brine Shrimp (Artemia salina) lethality assay at various doses. After 24 h, the concentration of 150 µg resulted in the highest death rate, whereas the concentration of 50 µg resulted in the lowest death rate. No mortality was found with the solvent. We calculated the LC50 values with the probit test and presented them in Fig. 7b. The radiated sample of Amrapali with a 1.5 kGy dose rate showed lower toxicity when compared to the other samples, with LC50 values of 206.03 µg/ml, respectively. In the instance of BARI-11, the control sample showed less toxicity (Fig. 7b).
4 Discussion
Food irradiation is an economically viable method for minimizing post-harvest losses, extending the shelf life of perishable goods, improving the hygiene quality of food, and inactivating food-borne viruses and parasites (Ashraf Chaudry et al., 2004). The microbial burden in the control sample was highest at week two. The microbial load was lowered in both types of mangoes at a treatment rate of 1.5 kGy. At 2 weeks, the microbial attack was slightly higher in the 2 kGy irradiated Amrapali and BARI-11 samples, though the 2 kGy radiation had a significant effect in reducing microbial attack on day one. In their study, Iqtedar et al. (2016) found that a 1 kGy radiation exposure rate resulted in the lowest microbial burden for Pakistani mango types named Sindhi (Iqtedar, Kiran, et al., 2016). Radiated mangoes took longer to ripen without hurting the fruit, whereas control mangoes decayed in just two weeks. The texture of the mangoes remained unchanged at a dosage rate of 1.5 kGy than the other dose rates. Similar outcomes were obtained by Mahto and Das, 2013 (Mahto & Das, 2013). Through the reduction or elimination of dangerous bacteria, viruses, parasites, and other microorganisms that might result in foodborne illness, food irradiation helps to decrease microbial attacks and enhance food safety. This is a great technique for decreasing food waste and raising the quality of the food that is available for consumption because it helps to avoid food spoilage and extend the shelf life of perishable foods (Indiarto et al., 2020). The ripening evaluation can be compared with the study of Iqtedar et al., in 2016 where they found dose rate 1 kGy was suitable for preservation (Iqtedar, Amjad, et al., 2016).
The Amrapali peel has the highest fat content of the two varieties. We observed lower fat content in our samples when compared to Ashoush and Gadallah's 2011 study (Ashoush & Gadallah, 2011). The fat level of all samples was high after 15 days due to the overripe mango, but it was higher in the control than in the other samples. In the peel samples, the BARI-11 mango with a 1.5 kGy dosage exhibited the highest protein content. In Indian mango peels, Ajila et al. observed a similar range of protein levels (Ajila et al., 2010). After 15 days, the protein levels in the control and 0.5 kGy radiated samples had altered, but not in the others. The total amount of ash in these findings matched those reported by Ajila et al. in 2007. But they didn’t use radiation to examine ash samples from mango peels. The ash level of all samples increased after 15 days, with the control samples exhibiting the most rise. The moisture content of the peel is similar to that of Indian Raspuri mango peels (Ajila et al., 2007). The moisture content of all samples increased after 15 days. Most percentages were found in the Amrapali control type, whereas the least was found in BARI-11 with a 2 kGy dose. During storage, both irradiated and non-irradiated bananas had the same moisture levels, according to research (Hassan, 2007). This indicates that radiation had little impact on moisture content. There was hardly any sugar in the peel samples. It ranged from 0.1% to 0.3%. Every sample's sugar content had risen after 15 days. The samples that had been exposed to radiation had less sugar than the those that hadn't. When climatic fruits are selected as opposed to when they are eaten when fully ripe, their sugar content differs (Khan et al., 2018).
Mango peel's fiber can promote digestive health and control blood sugar levels. In addition, mango peel's antioxidants have been linked to a number of health advantages, including as lowered oxidative stress and improved heart health (Ajila et al., 2008) (Masibo and He, 2009). The Amrapali peel sample contained the highest dietary fiber content at a dose rate of 2 kGy. Amrapali was found to have 8.4% fiber in an experiment (Tokas et al., 2020), which is somewhat higher than our data. After 15 days, the amount of dietary fiber in the peel samples had not changed appreciably. There were no noticeable changes between irradiated and control dates when researchers examined the effects of various gamma irradiation doses on the fiber content of dates stored in various colored polyethylene bags (Mohammadzai et al., 2010). Amrapali control peels had the highest concentration of ascorbic acid and the radiated samples showed a lower level of vitamin C on day one. After 15 days, the amount of vitamin C in the samples had significantly dropped in some samples compared to day one. This type of result has also been reported by another researcher (Youssef et al., 2002). The greatest titratable acidity in the peels was 0.25 % in the BARI-11 sample with a 1.5 kGy dosage and 0.29% in the Amrapali control. In Amrapali mango pulp, Ara et al., 2014 found 0.32 % titratable acidity. After 15 days, the titratable acidity content of almost all varieties increased. Lebaka et al., 2021 found decreased carbohydrate content in mango peel and pulp samples (Lebaka et al., 2021). The Amrapali and Bari 11 peel samples have similar amounts of carbohydrates. The number of carbohydrates in many samples increased after 15 days, while moisture, ash, and protein levels increased as well. The Amrapali peel sample has the highest level of energy. These findings matched those of Ara et al., 2014, who used Bangladeshi mangoes for their experiments. After 15 days of storage, the energy levels of different mango peels with varying dosages changed.
Infectious diseases, which account for nearly half of all human mortality, are most frequently brought on by microbes (El-desoukey et al., 2020). Antibacterial activities of Mangifera indica peel extracts were tested against E. coli, Pseudomonas sp., and Salmonella sp. The majority of the substances tested in this investigation were effective against Pseudomonas sp. but failed to kill Salmonella sp. Thambi and colleagues have found that mango peel extract has antibacterial action against Salmonella and E. coli (Thambi et al., 2016). At a dosage rate of 1.5 kGy, the mango samples had the highest antioxidant potentiality of the two types, although the control samples also demonstrated antioxidant activity. BARI-11 has the best antioxidant capabilities of the two mango types. This cultivar was developed by BARI (Bangladesh Agricultural Research Institute). The Keitt and Tommy Atkins mango varieties had increased antioxidant activity in the peel and pulp, according to Navarro et al. (Navarro et al., 2019). The findings, on the other hand, were identical to Ashoush and Gadallah's in 2011. They also found that baking biscuits with mango peel powder increased antioxidant levels in the biscuits. The cytotoxic impact of Mangifera indica peel extract at various doses was investigated using the Brine Shrimp (Artemia salina) lethality assay. The radiated samples of Amrapali with a 1.5 kGy dose rate demonstrated decreased toxicity when compared to the other samples. The cytotoxic impact of mango peel extract was also investigated by Chowdhury et al. 2017 (Chowdhury et al., 2017). They found that mango peels are more harmful than the peel extracts we utilized in our investigation. Thus, without affecting the nutritional value, gamma radiation can be used to increase the antioxidant and cytotoxic effects.
5 Conclusion
Based on phytochemical research, we can conclude that mango peels have a wide range of nutritional properties. It has a lot of antioxidant properties and a low toxicity level. This by-product of agro-industrial mango processing could be used to make high-fiber natural meals and a wide range of pharmaceuticals. Because gamma radiation can penetrate the entire fruit and inactivate microorganisms, it's a good way to improve the safety of fruits and vegetables. Our data indicated that 1.5 kGy was the ideal radiation dose for the preservation of fresh mangoes because it produced comparably better results. Thus, radiation may protect fruits from microbial attack and increase their shelf life, allowing people to enjoy them for longer.
Funding
The research was supported by the grants (696/5/52/Bio-11//R.U/2020–2021) provided by the University of Rajshahi, Bangladesh.
CRediT authorship contribution statement
Tabassum Jabin: . Sabiha Kamal: . Shirmin Islam: . Mamudul Hasan Razu: Data curation. Gobindo Kumar Paul: . Pranab Karmaker: Data curation. Mainul Huda: . Mashiur Rahman: Data curation. Md. Moniruzzaman: Data curation. Md. Salah Uddin: . Md. Abu Saleh: Writing – review & editing. Mala Khan: Supervision. Shahriar Zaman: Conceptualization, Supervision.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Phenolic-antioxidant capacity of mango seed kernels: therapeutic effect against viper venoms. Rev. Brasil. Farmacogn.. 2018;2017:4-11.
- [CrossRef] [Google Scholar]
- Valuable components of raw and ripe peels from two Indian mango varieties. Food Chem.. 2007;102(4):1006-1011.
- [CrossRef] [Google Scholar]
- Improvement of dietary fiber content and antioxidant properties in soft dough biscuits with the incorporation of mango peel powder. J. Cereal Sci.. 2008;48(2):319-326.
- [CrossRef] [Google Scholar]
- Characterization of bioactive compounds from raw and ripe Mangifera indica L. peel extracts. Food Chem. Toxicol.. 2010;48(12):3406-3411.
- [CrossRef] [Google Scholar]
- Nutritional evaluation of different mango varieties available in Bangladesh. Int. Food Res. J.. 2014;21(6):2169-2174.
- [Google Scholar]
- Utilization of mango peels and seed kernels powders as sources of phytochemicals in biscuit. World J. Dairy Food Sci.. 2011;6(1):35-42.
- [Google Scholar]
- Irradiation treatment of minimally processed carrots for ensuring microbiological safety. Radiat. Phys. Chem.. 2004;71(1–2):171-175.
- [CrossRef] [Google Scholar]
- Brine shrimp cytotoxicity, anti-inflammatory and analgesic properties of Woodfordia fruticosa Kurz flowers. Iran. J. Pharma. Res.. 2012;11(3):851-861.
- [Google Scholar]
- Bauer, A. W. (1966). Antibiotic susceptibility testing by a standardized single diffusion method. Am. J. Clin. Pathol., 45, 493-496.
- Review of Mango (Mangifera indica) seed-kernel waste as a diet for poultry. J. Biol. Agric. Healthcare. 2015;5(11):156-160.
- [Google Scholar]
- Comparative evaluation of antioxidant potential of alaternin (2-hydroxyemodin) and emodin. J. Agric. Food Chem.. 2000;48(12):6347-6351.
- [CrossRef] [Google Scholar]
- Phytochemical screening and evaluation of cytotoxic and hypoglycemic properties of Mangifera indica peels. Asian Pac. J. Trop. Biomed.. 2017;7(1):49-52.
- [CrossRef] [Google Scholar]
- El-desoukey, R. M. A., Aljor, N. M., & Alaotibi, A. D. (2020). The Phytochemical and Antimicrobial Effect of Mango (Mangifera Indica L .) Peel Extracts on Some Animal Pathogens as Eco-Friendly. 3(4), 34–39
- Gasu, E. K., Appiah, V., Gyamfi, A. A., & Nketsia-Tabiri, J. (2012). Effects of irradiation and chemical preservatives on the microbiological quality of refrigerated fresh-cut mangoes. European Journal of Food Research & Review, 2(4), 101–110. http://www.sciencedomain.org/abstract.php?iid=155&id=1&aid=704%0Ahttps://www.cabdirect.org/cabdirect/abstract/20133180850.
- HPLC-DAD-q-TOF-MS as a powerful platform for the determination of phenolic and other polar compounds in the edible part of mango and its by-products (peel, seed, and seed husk) Electrophoresis. 2016;37(7–8):1072-1084.
- [CrossRef] [Google Scholar]
- Hassan, P. (2007). Shelf Life Extension of Banana (Musa Sapientum) By Gamma Radiation. 47–53.
- Horwitz, W. (1975). Official methods of analysis (Vol. 222). Association of Official Analytical Chemists Washington, DC
- Indiarto, R., Pratama, A. W., Sari, T. I., & Theodora, H. C. (2020). Food Irradiation Technology : A Review of The Uses and Their Capabilities. December. https://doi.org/10.14445/22315381/IJETT-V68I12P216
- International, A. (1970). Official methods of analysis of the Association of Official Analytical Chemists. Association of official analytical chemists.
- Iqtedar, M., Amjad, H., Kaleem, A., & Abdullah, R. (2016). Effect of 60 Co gamma radiation on microbial flora and shelf life of Malus domestica Borkh. September. https://doi.org/10.12692/ijb/9.3.47-52
- Iqtedar, M., Kiran, S., Sarwar, U., Abdullah, R., Kaleem, A., Aftab, M., & Naz, S. (2016). EFFECT OF Co 60 GAMMA RADIATION ON SHELF LIFE OF PAKISTANI MANGOES EVALUATED THROUGH SENSORY AND MICROBIAL ANALYSIS. 6, 121–125.
- Islam, K., Khan, M. Z. H., Sarkar, M. A. R., Absar, N., & Sarkar, S. K. (2013). Changes in Acidity , TSS , and Sugar Content at Different Storage Periods of the Postharvest Mango (Mangifera indica L .) Influenced by Bavistin DF. 2013.
- Mango (Mangifera indica L.) by-products and their valuable components: a review. Food Chem.. 2015;183:173-180.
- [CrossRef] [Google Scholar]
- Antioxidant properties of selected mango varieties and correlation with the shelf-life of their products. Bangladesh J. Scient. Industrial Res.. 2017;52(4):303-308.
- [CrossRef] [Google Scholar]
- Khan, Q. U., Mohammadzai, I., Shah, Z., & Khattak, T. N. (2018). Effect of Gamma Irradiation on Nutrients and Shelf Life of Peach (Prunus persical) Stored at Ambient Temperature. 8–15. https://doi.org/10.2174/2210289201809010008.
- Nutritional composition and bioactive compounds in three different parts of mango fruit. Int. J. Environ. Res. Public Health. 2021;18(2):1-20.
- [CrossRef] [Google Scholar]
- Effect of gamma irradiation on the physico-chemical and visual properties of mango (Mangifera indica L.), cv. “Dushehri” and “Fazli” stored at 20°C. Postharvest Biol. Technol.. 2013;86:447-455.
- [CrossRef] [Google Scholar]
- Mannepula, S., Kumar Bathal, V., Sarathi Reddy Obulam, V., & Vijaya Sarathi Reddy Obulam, C. (2015). A comparative study on utilisation of citrus and mango peels for lactic acid production and optimisation by Rhizopus oryzae in submerged fermentation. European Journal of Biotechnology and Bioscience, 18(9), 18–26. www.biosciencejournals.com.
- Masibo and He, M. (2009). Mango Bioactive Compounds and Related Nutraceutical Properties — A Review June 2013, 37–41. https://doi.org/10.1080/87559120903153524.
- Brine shrimp: a convenient general bioassay for active plant constituents. Planta Med.. 1982;45(5):31-34.
- [Google Scholar]
- Mitra, S. K., Pathak, P. K., Devi, H. L., Chakraborty, I., Chandra, B., Viswavidyalaya, K., Bengal, W., & Bengal, W. (2013). Utilization of Seed and Peel of Mango. 593–596.
- Mohammadzai, I. ullah, Shah, Z., Ihsanullah, I., Khan, H., Khan, H., & Rashid, H. (2010). Effect of gamma irradiation, packaging and storage on the nutrients and shelf life of palm dates. Journal of Food Processing and Preservation, 34(SUPPL. 2), 622–638. https://doi.org/10.1111/j.1745-4549.2009.00421.x.
- Antimicrobial activity of some plant extracts against bacterial strains causing food poisoning diseases. Saudi J. Biol. Sci.. 2018;25(2):361-366.
- [CrossRef] [Google Scholar]
- Nair, R., & Chanda, S. V. (2007b). Antibacterial Activities of Some Medicinal Plants of the Western Region of India. 31, 231–236
- Antibacterial activities of some medicinal plants of the western region of India. Turk. J. Biol.. 2007;31(4):231-236.
- [Google Scholar]
- Enhancing antioxidant activity, microbial and sensory quality of mango (Mangifera indica L.) juice by γ-irradiation and its in vitro radioprotective potential. J. Food Sci. Technol.. 2015;52(7):4054-4065.
- [CrossRef] [Google Scholar]
- Polyphenolic characterization, antioxidant, and cytotoxic activities of mangifera indica cultivars from Costa Rica. Foods. 2019;8(9)
- [CrossRef] [Google Scholar]
- Noci, F., Riener, J., Cronin, D. A., Morgan, D. J., & Lyng, J. G. (2008). Ultraviolet irradiation and pulsed electric fields (PEF) in a hurdle strategy for the preservation of fresh apple Juice. 85, 141–146. https://doi.org/10.1016/j.jfoodeng.2007.07.011
- Mango by-products as a natural source of valuable odor-active compounds. J. Sci. Food Agric.. 2020;100(13):4688-4695.
- [CrossRef] [Google Scholar]
- Onuh, J. O., Momoh, G., Egwujeh, S., & Onuh, F. (2017). Evaluation of the Nutritional , Phytochemical and Antioxidant Properties of the Peels of Some Selected Mango Varieties. 5(5), 176–181. https://doi.org/10.12691/ajfst-5-5-2
- Effect of gamma-irradiation on sensory characteristics, physicochemical parameters, and shelf life of strawberries stored under refrigeration. Int. J. Fruit Sci.. 2020;20(2):191-206.
- [CrossRef] [Google Scholar]
- Alternation of antimicrobial potential of mango peel and pulp after formalin treatment against six bacteria. J. Pharmacogn. Phytochem.. 2016;5(5):158-161.
- [Google Scholar]
- Antibiotic resistance mechanisms in bacteria: relationships between resistance determinants of antibiotic producers, environmental bacteria, and clinical pathogens. Front. Microbiol.. 2018;9(NOV):1-21.
- [CrossRef] [Google Scholar]
- Reddy, H., & Mangalekar, S. B. (n.d.). Evaluation of the Efficacy of Guava Extract as an Antimicrobial Agent on Periodontal Pathogens. 690–697.
- Saleem Dar, M., Oak, P., Chidley, H., Deshpande, A., Giri, A., & Gupta, V. (2015). Nutrient and Flavor Content of Mango (Mangifera indica L.) Cultivars: An Appurtenance to the List of Staple Foods. In Nutritional Composition of Fruit Cultivars. Elsevier Inc. https://doi.org/10.1016/B978-0-12-408117-8.00019-2
- Brine shrimp lethality assay. Bangladesh J. Pharmacol.. 2017;12(2):186-189.
- [CrossRef] [Google Scholar]
- Determination and comparative study of sugars and synthetic colorants in commercial branded fruit juice products. J. Food Qual.. 2020;2020:1-11.
- [Google Scholar]
- Antimicrobial efficacy of mango peel powder and formulation of recipes using mango peel powder (Mangifera indica L.) Int. J. Home Sci. IJHS. 2016;2(2):155-161.
- [Google Scholar]
- Thiex, N. J., & Scientific, U. (2002). Determination of Crude Protein in Animal Feed, Forage, Grain, and Oilseeds by Using Block Digestion with a Copper Catalyst and Steam Distillation into Boric Acid: Collaborative Stu... March 2014.
- Mango peel : a potential source of bioactive compounds and phytochemicals. Austin Food Sci.. 2020;5(1):1-7.
- [Google Scholar]
- Youssef, B. M., Asker, A. A., El-samahy, S. K., & Swailam, H. M. (2002). Combined effect of steaming and gamma irradiation on the quality of mango pulp stored at refrigerated temperature. 35, 1–13
- Effects of Co-60 gamma-irradiation and refrigerated storage on quality of Shatang mandarin. Food Sci. Human Wellness. 2014;3(1):9-15.
- [CrossRef] [Google Scholar]







