Translate this page into:
Micro-titer plate assay for measurement of total phenolic and total flavonoid contents in medicinal plant extracts
⁎Correspondence author at: Department of Pharmaceutical Biology, Faculty of Pharmacy, Universitas Gadjah Mada, Yogyakarta 55281, Indonesia. hertiani@ugm.ac.id (Triana Hertiani)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Total phenolic content (TPC) measured by using the Folin-Ciocalteu method and Total flavonoid content (TFC) based on a complex formation with AlCl3 assay are widely applied as part of herbal extract quality control. Conducting the assay measurements in microtiter plates may save resources and time. The objective of this study was to evaluate the validity of the micro-titer plates assay to determine the TFC and TPC in several medicinal plants’ extracts. To compare the developed TPC and TFC methods to the conventional assays, the confidence intervals of linear regression were used with a significance set as p < 0.05. Both of the standards showed good linearity in the range of 10–60 mg L−1 of gallic acid and 40–200 mg L−1 of quercetin, (R2 > 0.999). Based on the micro-titer plate assay, the limits of detection (LODs), and quantification (LOQs) for TPC ranged between 1.19 mg L−1 and 3.98 mg L−1 whereas for TFC, they were 1.47 mg L−1 and 4.90 mg L−1. The percent coefficient of variation (%CV) of the intra-day and inter-day assays were lower than 1.46% and 1.91%, respectively, showing adequate precision. The recovery ranged between 101.25% and 105.93% for the TPC assay and between 96.78% and 101.28% for the TFC assay. Twelve samples of medicinal extracts were analyzed by validated microplate assay for TPC and TFC and then compared with the conventional method. Notably, there were no significant differences in the TPC and TFC content detected between the microplate and conventional methods for all samples (p > 0.05). In conclusion, the microplate assay can be potentially used for TPC and TFC determinations.
Keywords
Microtiter plate assay
Total Phenolic Content
Total Flavonoid Content
Medicinal plant extracts
1 Introduction
Medicinal plant extracts are widely used as active ingredients in herbal-based products. They contain metabolites, which differ in role and function. The unique and specific composition and chemical properties of each medicinal plant extract make them have high nutritional and medicinal values. A wide array of pharmacological activities were reported such as antibiotic (Oh & Jeon, 2015; Sanhueza et al., 2017), antifungal (Ansari et al., 2013; Zabka & Pavela, 2013), antioxidant (Nayak et al., 2015; Olas, 2018), anti-inflammatory (Ambriz-Pérez et al., 2016), anticancer (Roleira et al., 2015), and antiviral properties and hence have applications in pharmaceutical, food, cosmetics, and fine chemical industries (Balmus et al., 2016; Manach et al., 2004; Rajput et al., 2017; Tungmunnithum et al., 2018). Recent studies have investigated that the antioxidant effects of plant products are mainly attributed to phenolic compounds (Tungmunnithum et al., 2018; Xu et al., 2017).
There are more than 10,000 phenolic compounds that have been isolated from plants so far. In general, phenolic metabolites are divided into seven groups: coumarins, flavonoids, phenolic acids, lignans, lignin, stilbenes, and tannins (Huang et al., 2009). Many reports refer to the synergistic effects of those groups of compounds in exerting pharmacological effects. Therefore, the determination of total phenolic content (TPC) and total flavonoid content (TFC) in a medical plant extract continues to be a valuable tool for the assessment of quality.
Currently, the most common method used to measure the TPC of all types of herbal samples is the Folin–Ciocalteu method. This particular method is based on the reduction of the phosphomolybdate heteropoly acids Mo(VI) center in the heteropoly complex to Mo(V), producing a blue coloration which is measured at around 750 nm (Bobo-García et al., 2015). On the other hand, the TFC in plant extracts is widely measured using an aluminum chloride colorimetric assay. The method is based on the chelate formation of Al(III)-flavonoids. The numerous oxo and hydroxyl groups contained in this group's compounds, contribute a great affinity of flavonoids to bind metal ions such as Al(III), predominantly in a 1:1 ratio, depending on experimental conditions including pH value (Kasprzak et al., 2015; Pyrzynska & Pękal, 2011; Shraim et al., 2021).
The conventional methods for measuring the TPC and TFC are time-consuming, labor-intensive, and use large quantities of reagents (Johnson et al., 2022). The development of a valid high-throughput assay will be a valuable tool to overcome these downsides. In the case of exploration for potential plant extracts as medicinal resources, where a determination of TPC and TFC are needed as preliminary screening steps, the filtrate extracted could be limited. Therefore, the development of this method is needed.
The assays in microplates have been reported with positive results on a wide variety of samples such as apple, green tea, and grape seed extract for TPC and 2,2-diphenyl-1-picryl-hydrazl-hydrate (DPPH) activity assay (Bobo-García et al., 2015); sorghum for TPC and TFC (Herald et al., 2012), seaweeds for TPC (Zhang et al., 2006), ginger for TPC (Johnson et al., 2022), berries for TPC, TFC, and antioxidant capacity (Horszwald & Andlauer, 2011) and Cyphostemma digitatum for TPC and antioxidant capacity (Al-Duais et al., 2009). However, no statistical comparison between the results obtained with the microplate and the conventional methods has been reported. In light of this background, this study aimed to develop and validate the application of the Folin–Ciocalteu and AlCl3 micro-titer plate assays in comparison to the conventional methods, and to implement the method in the determination of TPC and TFC of several plant extracts.
2 Materials and methods
2.1 Materials
Chromolaena odorata L (Kirinyu or Slam Weed) (leaves, roots, stems), Centella asiatica (Pegagan or Pennywort) leaves, Plantago major L (Daun Sendok or Broadleaves Plantain) leaves, Chrysanthemum morifolium var. sheena and Chrysanthemum morifolium var. lamet (Krisan or Florist Daisy) flowers, Coleus amboinicus Lour (Torbagun or Indian Borage) leaves, Brassica oleraceae L.var. sabauda L. (Kubis Ungu or Purple Cabbage) flowers, Brassica oleraceae L. var. capitata forma alba DC (Kubis Putih or White Cabbage) flowers, Folin-Ciocalteu (Merck chemicals), Sodium carbonate anhydrous (Merck chemicals), Methanol (Merck chemicals), aluminum chloride (AlCl3), quercetin (Sigma MCLS), gallic acid (Sigma MCLS), deionized water (Waterone®), Sodium acetate (Merck chemicals), and ethanol 70% (Genera Labora/local).
2.2 Instruments
An ultrasonic chamber (Cole Palmer) was used for the extraction process of some samples, and the rotary evaporator (IKA-RV80) was used to concentrate the macerates. An ultraviolet (UV)-Vis spectrophotometer (Thermo, Genesys 10S) was used for the TPC and TFC measurement by the conventional method, and a microplate reader (Microlab 300) was used for the determination by the microplate assay.
2.3 Sample preparation and extraction protocol
Chromolaena odorata L, Centella asiatica, Chrysanthemum morifolium var. sheena and Chrysanthemum morifolium var. lamet flowers were collected from Sleman, Yogyakarta, Indonesia. Brassica oleraceae L.var. sabauda L. flowers dan Brassica oleraceae L. var. capitata forma alba DC flowers were collected from Magelang, Jawa Tengah, Indonesia. Plantago major L and Coleus amboinicus Lour leaves were collected from Bogor, Jawa Barat, Indonesia. All samples were rendered and dried at the temperature of 50 °C.
Each dried sample was powdered to a particle size of 40 mesh. Each sample was extracted with the extraction method as described in Table 1. Filtrates of each sample were rotary evaporated to produced concentrated extracts and stored at 4 °C until analysis was performed. For stock solution, each extract was diluted in methanol and was sonicated to enhance the solubility of the phytochemical compound. UAE, ultrasound-assisted extraction.
Sample Code
Sample name
Part of plants
Extraction Solvent
Extraction method
CoL
Chromolaena odorata L
Leaves
Ethanol 70%
Maceration
1:8, 3 days, room temperature
CoR
Chromolaena odorata L
Roots
Ethanol 70%
Maceration
1:8, 3 days, room temperature
CoS
Chromolaena odorata L
Stems
Ethanol 70%
Maceration
1:8, 3 days, room temperature
CaLE70
Centella asiatica
Leaves
Ethanol 70%
UAE
1:10, 45 °C, 20 min
CaLE96
Centella asiatica
Leaves
Ethanol 96%
UAE
1:10, 50 °C, 20 min
CaLEA
Centella asiatica
Leaves
Ethyl acetate
UAE
1:10, 50 °C, 20 min
PMLE50
Plantago major L
Leaves
Ethanol 10%
UAE
1: 15, 50 °C, 40 min
CmS
Chrysanthemum morifolium var. sheena
Flowers
Ethanol 70%
Maceration
1:10, 3 days, room temperature
CmL
Chrysanthemum morifolium var. lamet
Flowers
Ethanol 70%
Maceration
1:10, 3 days, room temperature
CaL
Coleus amboinicus Lour.
Leaves
Ethanol 70%
Maceration
1:10, 3 days, room temperature
BoS
Brassica oleraceae L. var. sabauda L.
Flowers
Ethanol 70%
Maceration
1:10, 3 days, room temperature
BoA
Brassica oleraceae L. var. capitata forma alba DC
Flowers
Ethanol 70%
Maceration
1:10, 3 days, room temperature
2.4 Conventional assay for total phenolic content (TPC)
The TPC is determined based on the reaction with the Folin–Ciocalteu reagent (Ribarova et al., 2005). In a 5 mL volumetric flask, 0.1 mL of the diluted extract and 0.1 mL of the Folin–Ciocalteu reagent was added and left to react for 5 min. To complete the reaction, 1 mL of a 7% sodium carbonate solution was added, and the volumetric flask was filled to its volume with deionized water. After 120 min in the darkness at room temperature, the absorbance of the samples at 750 nm was measured in the UV–Vis spectrophotometer. The measured absorbance of the same reaction with water instead of the extract or standard was subtracted from the absorbance of the reaction with the sample. The phenolic content was expressed in gallic acid equivalents (GAE) per gram after the preparation of a standard curve of gallic acid from 10 to 60 mg L−1.
2.5 Microplate assay for TPC
The microplate TPC method was conducted by applying the aforementioned Folin–Ciocalteu methods with some modifications. A total of 20 μL of the diluted extract were mixed with 20 μL Folin–Ciocalteu reagent and shaken for 1 min. The mixture was left for 5 min and then 200 μL of 7% sodium carbonate solution and 10 μL deionized water were added and the mixture was shaken at medium-continuous speed for 1 min. After 120 min in the darkness at room temperature, the absorbance was measured at 750 nm using the microplate reader. The absorbance of the same reaction with water instead of the extract or standard was subtracted from the absorbance of the reaction with the sample. Gallic acid dilutions (10–60 mg L−1) were used as standards for calibration. Phenolic contents are expressed in milligram equivalent of gallic acid per gram of dry extract (mg GAE/g).
2.6 Conventional assay for total flavonoid content (TFC)
The TFC is determined based on the reaction with the aluminum trichloride reagent (Shraim et al., 2021). 500 μL of the diluted extract was mixed with 2 mL methanol and 200 μL of a 10% AlCl3 solution. After incubating for 3 min at room temperature, 200 μL of a 1 M CH3COONa was added to the mixture. The final volume was adjusted to 5 mL with methanol. After 40 min of incubation in the darkness at room temperature, the absorbance of this preparation was measured at 430 nm. A calibration curve was created in parallel under the same operating conditions using quercetin. Flavonoid contents are expressed in milligram equivalent to quercetin per gram of dry extract (mg QE/g).
2.7 Microplate assay for TFC
To microplate, a TFC assay was conducted by applying the aforementioned TFC method with some modifications. A total of 50 μL of the sample extract or standard solution and 100 µL methanol were added to each well. One well on the plate was used as a blank for the microplate absorbance reading and filled up with 150 μL methanol. Following the addition of 20 μL AlCl3 10% to each well, the plate was gently shaken five times each up and down and left to right in the horizontal plane. The plate was kept in incubation for three minutes. After the incubation period, 20 µL CH3COONa 1 M the added and followed by 60 μL Methanol. The plate was then incubated in the darkness at room temperature for 40 min and then the absorbance was measured at 430 nm using a microplate reader.
2.8 Method validation
A validation study was conducted to demonstrate the applicability of this analytical approach to assess the medicinal plant extracts' quality. Validation comprised the assessment of specificity/selectivity, linearity, recovery, precision, the limits of detection (LODs), and quantification (LOQs). To evaluate the linearity of the method, the calibration curves were plotted by absorbance versus concentration of each standard. To prepare the standard solutions, gallic acid (10, 20, 30, 40, 50, and 60 mg L−1) and quercetin (40, 80, 120, 160, and 200 mg L−1) were dissolved in methanol. The linear regression equations were calculated as y = ax ± b, where × was concentration and y was the absorbance of each standard. The acceptance criteria for linearity are that the correlation coefficient (R2) should not be <0.995 over the working range of 80–120% (Food and Drug Administration, 2020). The LODs and LOQs were determined based on the standard deviation (SD) of the response and the slope, using the calibration curve data, and calculated based on the SD of the intercept with y and the slope of the calibration curve (S) according to the Eqs. (1) and (2).
Accuracy was evaluated by determining the method of recovery. The mean percentage of recovery should be within the following ranges. The precision of the intra- and inter-day was evaluated by the repeated assay. The intra-day experiment was obtained by three plates for a single day, and the inter-day was determined for three consecutive days. Analysis was performed in triplicate. The precision was expressed as the percent coefficient of variation (%CV) that calculated from SD compare to the Mean values of four repetitions of the analysis in each concentration.
2.9 Statistical analyses
Statistical comparisons of TPC and TFC between conventional and microtiter methods were performed based on Levene’s test value for equality of variance and t-test value for equality of means of each sample extract. Both methods are considered statistically equal with a 95% confidence interval (CI), while the p-values of Levene’s test and t-test have to be >0.05. This statistical analysis was performed using Minitab 17 Statistical Software.
3 Results and discussion
3.1 Calibration curves and limits of detection and quantification
The standard calibration curves for the microplate methods are presented in Fig. 1. The curves are linear when the concentration of gallic acid is in the range of 10–60 mg L−1 (R2 = 0.999) and the concentration of quercetin is in the range of 40–200 mg L−1 (R2 = 0.999). When comparing these curves with those obtained by conventional methods, it can be seen that the slight changes in the methodology had more influence on the slope of the TPC assay than on the slope of the TFC assay. The slope of the calibration curve for the TFC using the microplate method was 0.005, while the slope in the conventional study was 0.005; for the calibration curves of the TPC using the microplate and conventional method, the slopes were 0.013 and 0.022, respectively. However, comparing the slopes of this work, it can be seen that greater differences in the TPC and TFC microplate methods had a significant impact on the sensitivity of the methods. In this work, the LOD and LOQ for the TPC microplate method were 1.19 and 3.98 mg L−1 GAE, respectively. In the case of the TFC microplate method, the LOD and LOQ were 1.47 and 4.90 mg L−1 of quercetin equivalents, respectively (Table 2). The LOQ and LOQ both for TPC and TFC of the conventional method are lower than the LOD and LOQ of the microplate method. This can demonstrate that the conventional method is more sensitive and accurate than the microplate assay. LOD, limits of detection; LOQ, limits of quantification; TPC, total phenolic content; TFC, total flavonoid content.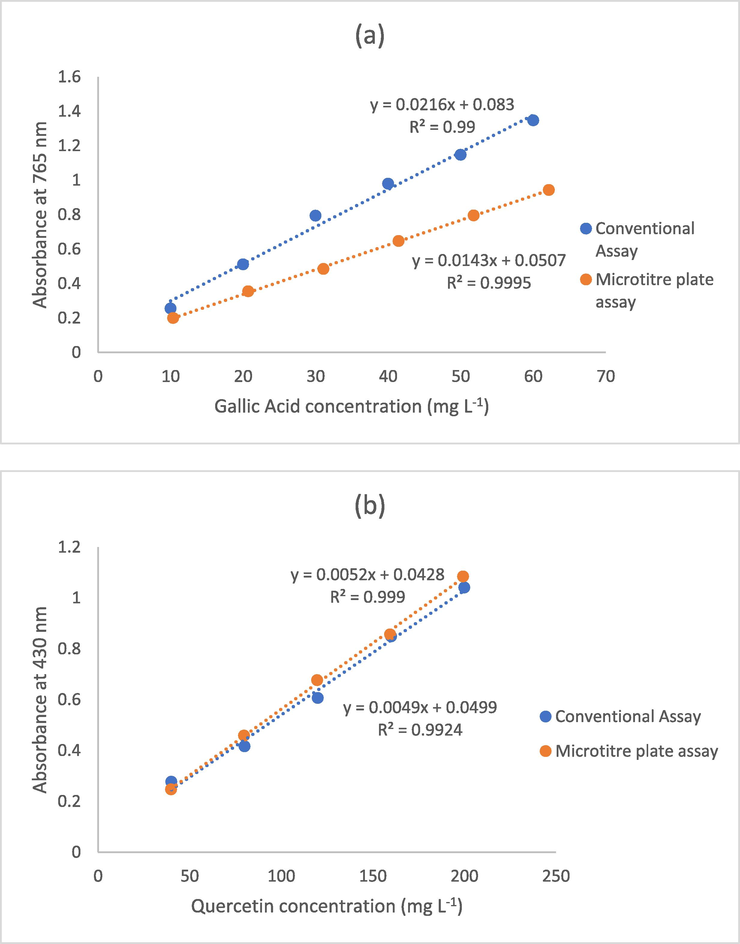
Representative standard curve for 96-well assay: (a) total phenolic content, and (b) total flavonoid content.
Slope
Intercept
Pearson R2
LOD (mg/L)
LOQ (mg/L)
TPC
Conventional assay
0.022
0.083
0.990
0.58
1.93
Microplate assay
0.014
0.05
0.999
1.19
3.98
TFC
Conventional assay
0.005
0.049
0.992
0.80
2.66
Microplate assay
0.005
0.043
0.999
1.47
4.90
3.2 Accuracy and precision
Accuracy was calculated and expressed as % recovery (%REC), which is the difference between the calculated value of the standard obtained after running the microplate assay and the actual value of the prepared standard, expressed as a percentage. The %REC values ranged from 101.30 to 105.93% for the three concentrations of gallic acid and 96.78 to 101.28% for the concentration of quercetin tested, suggesting that the microplate assays can be reproduced with excellent accuracy (Table 3). Similarly, the %CV values were <5% for all three concentrations tested, which is considered good precision. The %CV values for inter-day variability and intra-day reproducibility were 1.46% and 1.91%, respectively for gallic acid. The %CV values for inter-day variability and intra-day reproducibility for quercetin were 1.48% and 0.89%, respectively (Table 4). Values are expressed as mean ± standard deviation of five replicates. Gallic acid was used for total phenolic content and quercetin was used for total flavonoid content; %REC, percentage recovery; %CV, percentage. %CV, percent coefficient of variation; GAE, gallic acid equivalent; QE, quercetin equivalent.
Assay
Std 1
Std 2
Std 3
Total phenolics
20 µg mL−1
30 µg mL−1
40 µg mL−1
Calculated (µg mL−1)
21.19 ± 0.27
31.28 ± 0.23
40.52 ± 0.20
%REC
105.93
104.26
101.30
%CV
1.12
0.66
0.44
Total flavonoid
80 µg mL−1
120 µg mL−1
160 µg mL−1
Calculated (µg mL−1)
77.42 ± 1.38
121.46 ± 2.82
156.04 ± 2.45
%REC
96.78
101.28
97.52
%CV
1.60
2.07
1.40
Assay
Inter-day variability
Intra-day variability
Mean
%CV
Mean
%CV
Total phenolic (GAE)
54.07 ± 0.79
1.46
51.37 ± 1.10
1.91
Total flavonoids (QE)
111.469 ± 1.69
1.48
106.19 ± 1.06
0.89
3.3 Application of the validated microplate assay of TPC on various medicinal plant extracts
A total of 12 extracts were analyzed in four replicates using the optimized microplate TPC method and then compared to the conventional method. The average coefficient of variation on each extract was a range of 0.34–2.29% which is considered good in precision. In comparison, the %CV obtained using the conventional TPC method is approximately 0.75–2.43%.
Table 5(a) shows that the TPC levels of each extract sample varied. The total phenolic of sample extract determined using microplate assay ranged from 8.48 to 28.72 mg/g GAE. The highest TPC was obtained in Centella asiatica leaves which were extracted using 70% ethanol and the lowest TPC was in Brassica oleraceae L. var. capitata f. alba DC extract. The TPC of each plant part shows varying values, for example in Chromolaena odorata L, the part of the plant that contains the highest TPC is the leaf part while the root part contains the smallest TPC. In addition, it can be seen that the solvent used during plant extraction can also make a difference in the TPC levels of the samples. It can be seen in Centella asiatica, the 70% ethanol solvent gave the highest TPC value followed by 96% ethanol and then ethyl acetate. This can be possible due to the better solubility of phenolic compounds in polar solvents.
Sample
96-well assay
Conventional method
Levene’s test
t-test
Value ± SD
%CV
Value ± SD
%CV
p-value
p-value
Total phenolic content (mg/g GAE)
CoL
25.66 ± 0.28
0.93
25.49 ± 0.22
0.75
0.99
0.38
CoS
13.31 ± 0.05
0.34
13.11 ± 0.18
1.22
0.13
0.07
CoR
10.96 ± 0.23
1.82
10.58 ± 0.25
2.02
0.90
0.08
CaLE70
28.72 ± 0.76
2.29
29.08 ± 0.39
1.16
0.14
0.43
CaLE96
26.31 ± 0.46
2.23
26.72 ± 0.75
2.43
0.14
0.38
CaLEA
18.56 ± 0.15
0.70
18.41 ± 0.27
1.27
0.17
0.36
PmLE50
25.79 ± 0.51
1.70
26.46 ± 0.51
1.66
0.97
0.11
CmS
11.07 ± 0.18
1.43
10.83 ± 0.16
1.31
0.91
0.10
CmL
12.32 ± 0.13
0.92
12.07 ± 0.20
1.43
0.23
0.08
CaL
15.64 ± 0.25
1.38
15.88 ± 0.33
1.79
0.58
0.29
BoS
10.626 ± 0.12
0.96
10.83 ± 0.15
1.16
0.71
0.06
BoA
8.48 ± 0.18
1.85
8.75 ± 0.14
1.41
0.65
0.06
Total flavonoid content (mg/g QE)
CoL
21.59 ± 0.08
0.33
21.68 ± 0.30
1.22
0.22
0.58
CoS
3.59 ± 0.08
1.87
3.67 ± 0.03
0.74
0.06
0.09
CoR
2.37 ± 0.04
1.28
2.44 ± 0.05
1.81
0.55
0.07
CaLE70
16.13 ± 0.19
1.03
16.37 ± 0.15
0.81
0.48
0.09
CaLE96
15.89 ± 0.26
1.39
15.76 ± 0.22
1.23
0.80
0.46
CaLEA
10.27 ± 0.19
1.64
10.54 ± 0.15
1.26
0.62
0.07
PMLE50
12.55 ± 0.24
1.67
12.68 ± 0.21
1.45
0.52
0.45
CmS
4.12 ± 0.04
0.87
4.09 ± 0.06
1.28
0.22
0.40
CmL
5.66 ± 0.01
1.52
5.53 ± 0.12
1.89
0.72
0.15
CaL
11.24 ± 0.33
2.56
11.49 ± 0.14
1.03
0.10
0.21
BoS
2.97 ± 0.03
0.77
2.95 ± 0.01
0.29
0.20
0.22
BoA
4.00 ± 0.11
2.38
4.10 ± 0.05
1.13
0.37
0.17
To compare the TPC conventional and microplate methods, the result from both methods were then statistically analyzed using SPSS 23 (IBM Corp., Armonk, NY). Table 5(a) shows the p-values of Levene’s test for equality of variances and the p-value of the t-test for equality of means. In all cases, the p-values for Levene’s test for equality of variance were >0.05, which indicates that the variances among the measurements for each sample (n = 4) were not significantly different at 95% CI. The lack of significant differences in the variance indicates that both methods are equally precise and allow the performance of the p-value of the t-test for equality of means. All the p-values for the p-value of the t-test for equality of means were >0.05, which indicates that there were no significant differences between the mean values for both methods at a 95% CI. Based on the p-values resulting from both statistical tests, it can be said that the conventional and the microplate methods were equivalent in the concentration ranges studied.
3.4 Application of the validated microplate assay of TFC assay on various medicinal plants extracts
The TFC determination by using the optimized microplates assay was also performed on 12 different extracts. The results of the TFC analysis of both microplate and conventional methods are summarized in Table 5(b). The %CV range for the conventional assay was 0.29–1.89% and 0.33–2.56% for the microplate assay. It indicated that the determination of TFC using the microplate method for all sample extracts is considered to have good precision. The TFC of the extract ranges from 2.37 mg/g quercetin equivalents (QE), which is found in Chromolaena odorata L roots extract, to 21.59 mg/g QE which is found in Chromolaena odorata L leaves extract. The diversity of TFC levels from each sample can be seen in Table 5 (b). In Chromolaena odorata L, every part of the plant that contains various TFC content with the highest content is the leaf part while the smallest TFC is in the root part. In TFC, the solvent used during plant extraction can also make a difference in the samples. As can be seen in Centella asiatica, the highest TFC was showed in 70% ethanol solvent, followed by 96% ethanol and then ethyl acetate.
The statistical analysis conducted for both conventional and microplate methods for TFC determination shows that the p-value from all samples is above 0.05. This result indicated that the variances of all samples are equal for both methods. Furthermore, the p-value from the t-test analysis shows that in all samples for both methods are >0.05. So, it can be concluded that there were no significant differences between the microplate and conventional method to determine TFC from the sample extracts.
3.5 General observations
The microplate assay uses fewer resources and provides a rapid measurement in comparison to conventional assays. The microplate assay reduced the time and manpower needed to transfer the reactant solutions to the cuvettes to be manually read in the spectrophotometer and allowed for more replicates to be run on the same extracts than was possible with the conventional methods. The need for numerous cuvettes, transfer pipettes, and test tubes was eliminated. Instead, the only consumables used were pipette tips and 96-well plates. The amounts of the reagent required also decreased. In the conventional TPC assay, the total volume used of FC reagents is 100 µL per reaction, whereas only 20 µL of FC reagent was required per reaction for the microplate assay. In the TFC assay, the AlCl3 used for the microplate assay was only 20 µL per reaction compared to conventional assay that used 200 μL AlCl3 per reaction.
The gallic acid and quercetin absorbances of the two methods showed a correlation with R2 = 0.9987 and R2 = 0.9964, respectively. This linearity was slightly higher for the microplate methods than for the conventional methods, even though all %CV < 5% and indicated good reproducibility of the method when applied to real sample extracts. However, the savings on time, the amounts of samples that can be run per day, and the reduced amount of solvent due to the use of the microplate assays more than compensated for the slightly higher variability especially for detection of lots sample variant.
Previous studies using microplates for the determination of TPC in grape seed apple and green tea extracts by Gloria in 2015 showed that the results of the microtiter and conventional assay methods were linearly correlated. (Bobo-García et al., 2015). Herald's research in 2012 highlighted that the results of TPC determination using a microplate showed good precision with a %CV value <10% on various test samples in the form of flour and brans (Herald et al., 2012). The research conducted by Johnson in 2022 on TPC determination also obtained validated method results with good reproducibility, with CV < 5% (Johnson et al., 2022).
However, several aspects need to be considered in this microtiter test to minimize some errors in the results such as the accuracy or pipetting technique of the researcher and the solubility of the sample in the solvent. If the extract or sample is partly insoluble in the solvent used, it will cause unreliable results because the particles in the sample can interfere with the intensity reading on the microplate reader causing it to become unstable and even higher, and this also applies in the determination with conventional methods. Therefore, the selection of the sample solvent is a critical point in this test.
Determination of total flavonoids with AlCl3 reagent will react with the C4 keto group and between the C3 or C5 OH groups of flavonoids. In addition, aluminum chloride can also form complexes with ortho-dihydroxyl groups in the A or B rings of flavonoid compounds. The method for determining total flavonoids using aluminum chloride reagent is more specific for flavones and flavonols, so it is necessary to develop a microtiter assay method for determining total flavonoids using other methods, such as using the 2,4-dinitrophenylhydrazine reagent, which is more stable for the determination of flavanones (Chang et al., 2002).
The extract samples used in this study are diverse to represent different types of plants both in terms of species but also plant parts (roots, leaves, stems, to flowers). This is to get an idea of the breadth of application of this microtiter method when applied to herbal extracts which contain phytochemical compounds with various physical and chemical properties. The content of these phytochemical compounds is of course also influenced by the type of raw material since when using colored flower parts there will be various dyes, and in leaves containing chlorophyll, or roots and stems containing lignin because these compounds may interfere with the analysis process.
This micro-titter plate method can be applied at the initial screening process stage for the exploration of efficacious natural plants, for example, in the process of extraction optimization which could obtain an extract with limited yield. In this case, this method will be useful because it can minimize the resources needed.
4 Conclusions
The microplate method developed in this study showed several benefits not only can handle large number of samples in one experiment but also reduces the amount of sample and reagent requirements for total phenolic and total flavonoid content. Furthermore, it showed acceptable repeatability and reproducibility. The repeatability, reproducibility, and percentage of recovery for the TPC and TFC microplate methods showed a precision below 5% and accuracy between 96.78 and 105.93%. For the application for several extract, the microplate and the conventional methods are equal based on statistical analysis at a 95% confidence level These microplate assays can be used for routine screening of a large number of samples because it was demonstrated to be as reproducible, efficient, accurate, and precise as the conventional method for determining total phenolic content and total flavonoid content in several extracts.
Acknowledgments
The author is grateful to the Center for Education Financial Services and Indonesia Endowment Funds for Education for financial support as Doctoral Scholarship and the staff at Klinik Bahasa, Office of Research and Publication, Faculty of Medicine, Public Health, and Nursing, Universitas Gadjah Mada, who kindly provided proofreading assistance.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Antioxidant capacity and total phenolics of Cyphostemma digitatum before and after processing: use of different assays. Eur. Food Res. Technol.. 2009;228(5):813-821.
- [CrossRef] [Google Scholar]
- Phenolic compounds: Natural alternative in inflammation treatment. A Review. Cogent Food Agric.. 2016;2(1):1131412.
- [CrossRef] [Google Scholar]
- Natural phenolic compounds: a potential antifungal agent. Microbial Pathogens Strateg. Combating Them: Sci. Technol. Educ.. 2013;1:1189-1195.
- [CrossRef] [Google Scholar]
- The implications of oxidative stress and antioxidant therapies in Inflammatory Bowel Disease: Clinical aspects and animal models. Saudi J. Gastroenterol.: Off. J. Saudi Gastroenterol. Assoc.. 2016;22(1):3.
- [CrossRef] [Google Scholar]
- Intra-laboratory validation of microplate methods for total phenolic content and antioxidant activity on polyphenolic extracts, and comparison with conventional spectrophotometric methods. J. Sci. Food Agric.. 2015;95(1):204-209.
- [CrossRef] [Google Scholar]
- Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J. Food Drug Anal.. 2002;10(3)
- [CrossRef] [Google Scholar]
- Methods, Method Verification and Validation. 2020;II:1-32.
- High-throughput micro plate assays for screening flavonoid content and DPPH-scavenging activity in sorghum bran and flour. J. Sci. Food Agric.. 2012;92(11):2326-2331.
- [CrossRef] [Google Scholar]
- Characterisation of bioactive compounds in berry juices by traditional photometric and modern microplate methods. J. Berry Res.. 2011;1(4):189-199.
- [CrossRef] [Google Scholar]
- Natural phenolic compounds from medicinal herbs and dietary plants: potential use for cancer prevention. Nutr. Cancer. 2009;62(1):1-20.
- [CrossRef] [Google Scholar]
- Johnson, J.B., Mani, J.S., Naiker, M., 2022. Development and Validation of a 96-Well Microplate Assay for the Measurement of Total Phenolic Content in Ginger Extracts. In: Food Analytical Methods (Vol. 15, Issue 2, pp. 413–420). Doi: 10.1007/s12161-021-02127-9.
- Properties and applications of flavonoid metal complexes. RSC Adv.. 2015;5(57):45853-45877.
- [CrossRef] [Google Scholar]
- Polyphenols: food sources and bioavailability. Am. J. Clin. Nutr.. 2004;79(5):727-747.
- [CrossRef] [Google Scholar]
- Effect of processing on phenolic antioxidants of fruits, vegetables, and grains—a review. Crit. Rev. Food Sci. Nutr.. 2015;55(7):887-918.
- [CrossRef] [Google Scholar]
- Synergistic anti-Campylobacter jejuni activity of fluoroquinolone and macrolide antibiotics with phenolic compounds. Front. Microbiol.. 2015;6:1129.
- [Google Scholar]
- Berry phenolic antioxidants–implications for human health? Front. Pharmacol.. 2018;9:78.
- [CrossRef] [Google Scholar]
- Flavonoids as analytical reagents. Crit. Rev. Anal. Chem.. 2011;41(4):335-345.
- [CrossRef] [Google Scholar]
- Rajput, D.S., Dash, D.K., Sahu, A.K., Mishra, K., Kashyap P, & S, P.M., 2017. Brief update on Indian herbs and spices used for diabetes in rural area of Chhattisgarh. In: International Journal of Pharmaceutical Chemistry and Analysis. academia.edu. https://www.academia.edu/download/53241726/IJPCA_41_1-4.pdf.
- Total phenolics and flavonoids in Bulgarian fruits and vegetables. JU Chem. Metal. 2005;40:255-260.
- [Google Scholar]
- Plant derived and dietary phenolic antioxidants: Anticancer properties. Food Chem.. 2015;183:235-258.
- [CrossRef] [Google Scholar]
- Synergistic interactions between phenolic compounds identified in grape pomace extract with antibiotics of different classes against Staphylococcus aureus and Escherichia coli. PLoS One. 2017;12(2):e0172273.
- [Google Scholar]
- Determination of total flavonoid content by aluminum chloride assay: A critical evaluation. LWT. 2021;150:111932
- [CrossRef] [Google Scholar]
- Flavonoids and other phenolic compounds from medicinal plants for pharmaceutical and medical aspects: An overview. Medicines. 2018;5(3):93.
- [CrossRef] [Google Scholar]
- Natural antioxidants in foods and medicinal plants: Extraction, assessment and resources. Int. J. Mol. Sci.. 2017;18(1):96.
- [CrossRef] [Google Scholar]
- Antifungal efficacy of some natural phenolic compounds against significant pathogenic and toxinogenic filamentous fungi. Chemosphere. 2013;93(6):1051-1056.
- [CrossRef] [Google Scholar]
- A simple 96-well microplate method for estimation of total polyphenol content in seaweeds. J. Appl. Phycol.. 2006;18(3):445-450.
- [CrossRef] [Google Scholar]







