Translate this page into:
A comprehensive review of phytochemistry, pharmacology and clinical applications of Uncariae Ramulus Cum Uncis
⁎Corresponding authors. zhangnatprod@163.com (Dong-dong Zhang), ellewang@163.com (Rui Wang)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Objectives
Uncariae Ramulus Cum Uncis (URCU) belonging to the genus Uncaria is widely distributed in China and used in folk medicine, which has the effect of clearing heat and calming the liver, extinguishing wind and settling convulsion. So, it is used to treat hypertension and neurological diseases. Herein, we reported a review on botany, phytochemistry, pharmacology and clinical applications reported from 1973 up to 2022. All the information and studies concerning URCU were summarized from the library and digital databases (e.g. Sciencedirect, SciFinder, Medline PubMed, Google Scholar, and CNKI).
Key findings
A total of 190 articles about URCU have been collected. The phytochemical investigations of URCU revealed the presence of more than 371 chemical components, including alkaloids, terpenoids, flavonoids, phenylpropanoids, phytosterols and phenolics. Moreover, the compounds isolated from URCU possessed a wide spectrum of pharmacology such as anti-hypertension, antiinflammation, anticancer, antioxidant, antiviral, anti-epilepsy, anti-depressant, ischemic brain injury, neuroprotection, anti-Alzheimer's disease, anti-Parkinson's disease and antiasthma.
Summary
In this paper, the botany, phytochemistry, pharmacology and clinical applications of URCU were reviewed. As a source of traditional folk medicine, URCU has high medicinal value and are widely used in medicine. Therefore, we hope our review can help URCU get better development and utilization.
Keywords
Uncariae Ramulus Cum Uncis
Phytochemistry
Pharmacology
Clinical Applications
1 Introduction
As a country that has been using herbal medicine to treat diseases since ancient times, China has abundant natural drug resources and experience in clinical application. Uncariae Ramulus Cum Uncis (URCU) was a common Traditional Chinese Medcicine (TCM) used to extinguish wind and settle convulsion (Zhao, 2021; Tang, 2020). In Chinese Pharmacopoeia (2020 edition), URCU is stem and hook of five species from the genus Uncaria (Chinese Pharmacopoeia Commission, 2020). The plants of URCU, with rich chemical compositions and pharmacological activities, have been used in Traditional Chinese medicines or folk medicines to treat various diseases, which have become a hot spot for phytochemical studies. Currently, more than 371 compounds have been extracted and identified from URCU including alkaloids (Chi, 2017), terpenoids (Wu et al., 2007), flavonoids (Sun et al., 2012c), phenylpropanoids (Shin and Lee, 2013), phytosterols (Zhang, 2013) and phenolics (Yang, 2018). And alkaloids were major compounds. Meanwhile, several studies showed that the compounds and extracts isolated from URCU possessed a wide spectrum of pharmacology in vivo or in vitro such as anti-hypertension (Li et al., 2020), antiinflammation (Kim et al., 2010), anticancer (Kim et al., 2014), antioxidant (Yin et al., 2010), antiviral (Reis et al., 2008), anti-epilepsy (Tang et al., 2017), anti-depressant (Qiao et al., 2021), ischemic brain injury (Xie et al., 2009), neuroprotection (Lee et al., 2003), anti-Alzheimer's disease (Fu et al., 2014), anti-Parkinson's disease (Li et al., 2017b) and antiasthma (Wang et al., 2019). So, it is necessary to review URCU for better research. In this study, we comprehensively summarized research on botany, phytochemistry, pharmacology and clinical application of URCU (Fig. 1). The extant information on these species allows us to provide a scientific basis for future research studies and to explore the potential therapeutic use.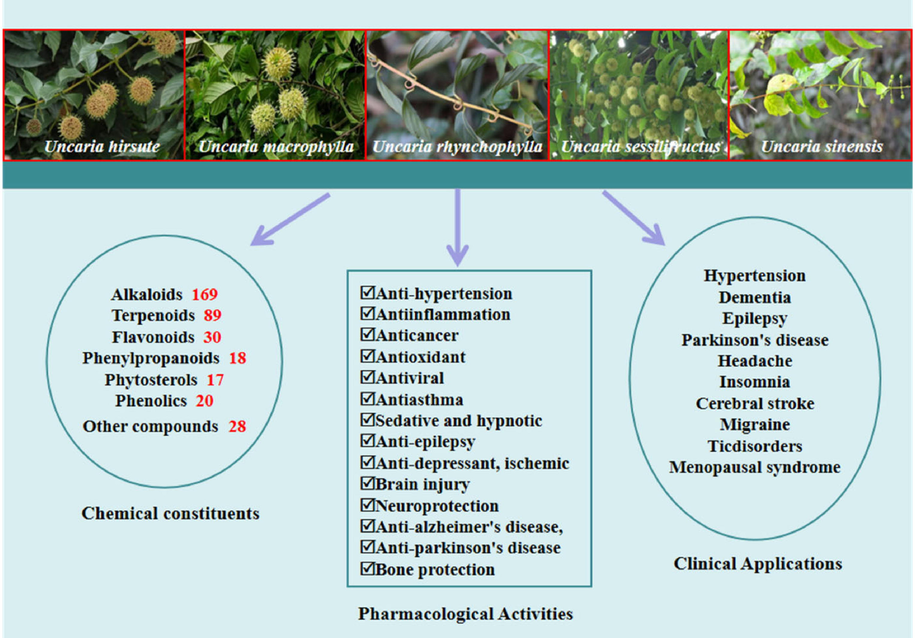
URCU, chemical constituents, pharmacological activities and clinical applications.
2 Search strategy
Comprehensive research and analysis of previously published literature were conducted for studies on the botany, phytochemistry, pharmacology and clinical application properties of URCU. The search was conducted using databases such as Sciencedirect, SciFinder, Medline PubMed, Google Scholar, Baidu Scholar, and CNKI by using the keywords such as Uncaria hirsuta; Uncaria macrophylla; Uncaria rhynchophylla; Uncaria sessilifructus; Uncaria sinensis. Furthermore, part of the analyzed studies was got by a manual search of articles in the reference lists of the included studies. The PRISMA template for determining the list of articles is displayed in Fig. 2. The chemical structures were drawn using ChemDraw Professional 20.0.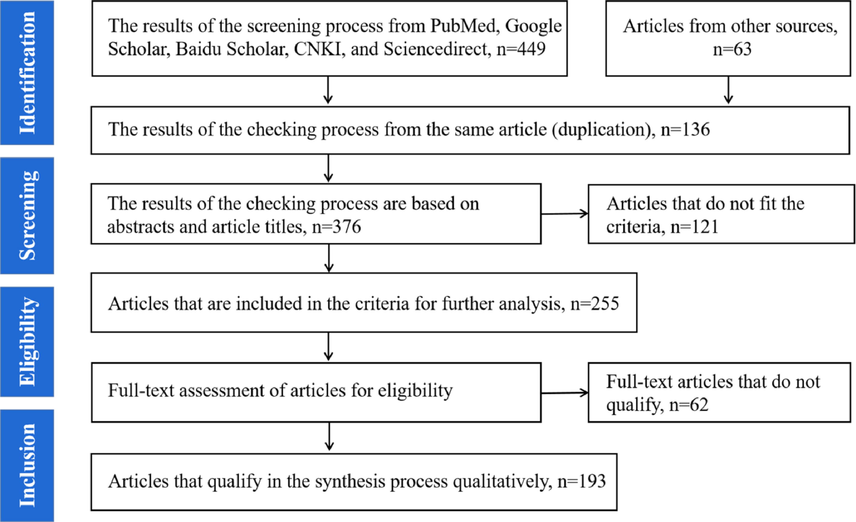
Research Data Search & Selection Flow.
3 Botany, Description and Distribution
URCU was a common TCM used to extinguish wind and settle convulsion. According to the herbal textual research of scholars, URCU mainly referred to Uncaria sinensis (Oliv.) Havil in the Tang Dynasty. In the Song Dynasty, URCU mainly referred to Uncaria rhynchophylla (Miq.) Miq. ex Havil. and U. sinensis. While, URCU mainly included U. sinensis, U. rhynchophylla and Uncaria sessilifructus Roxb.. in the Ming Dynasty. Now, Uncaria hirsuta Havil. and Uncaria macrophylla Wall. are also considered as the source of URCU (Huang et al., 2016). In Chinese Pharmacopoeia (2020 edition), URCU was the stem with hook of five species from the genus Uncaria, including U. hirsuta, U. macrophylla, U. rhynchophylla, U. sessilifructus, U. sinensis now (Chinese Pharmacopoeia Commission, 2020).
According to Chinese Flora, the common botanical morphology of URCU is woody vines, tender branches square or cylindrical, glabrous or pubescent, and nutrient laterals often metamorphose into hook prickles. leaves opposite; axils of lateral veins usually have pits; stipules entire or absent, two shallow lobed or two deeply lobed, ventral base or entire surface with mucor hairs. Headlike inflorescences terminal on lateral branches, and sparsely branched as compound umbrella cone inflorescence. Five flowers; the total pedicel has sparse or dense hairs; bracts linear or linear spoon - shaped; calyx tube short, sepal lobes glabrous or densely hairy; corolla disc-shaped or nearly funnel-shaped, glabrous or densely hairy outside, corolla lobes ovately oblong or elliptic; stamens inserted near throat of corolla tube, filaments short; styles extended, stigma spherical or long rod-shaped, verrucous at the top, ovary-two-chambered, placenta at least one third of the upper diaphragm; most ovules. Capsule two-chambered, outer pericarp thick, longitudinally dehiscent, inner pericarp thick bone, dorsally dehiscent; seeds small, mostly, centrally reticulate, with long wings at both ends, two deeply lobed wings below (Uncaria plant in Flora of China @ efloras.org, 2020). The local name, distribution and morphological features of URCU were shown in Table 1.
Species name
Local name
Distribution
Morphological features
Uncaria hirsuta Havil.
Maogouteng
TaiwanfengtengChina (Guangdong, Guangxi, Guizhou, Fujian and Taiwan)
Leaves leathery, densely hard haired below; stipules deeply 2-lobed, lobes ovate; calyx lobes linear oblong, widest near base (Uncaria plant in Flora of China @ efloras.org, 2020).
Uncaria macrophylla Wall.
Dayegouteng
China (Yunnan, Guangxi, Guangdong, Hainan), India, Bhutan, Bangladesh, Myanmar, northern Thailand, Laos and Vietnam
stipules deeply 2-lobed; leaves nearly leathery, ovate or broadly elliptic, 10–16 cm long, 6–12 cm wide (Uncaria plant in Flora of China @ efloras.org, 2020).
Uncaria rhynchophylla (Miq.) Miq. ex Havil.
Gouteng
China (Guangdong, Guangxi, Yunnan, Guizhou, Fujian, Hunan, Hubei and Jiangxi), Japan
corolla ca. 7 mm; reddish brown or dark red under leaves when dry; headed inflorescence regardless of corolla diameter 5–8 mm (Yang et al., 2018).
Uncaria sessilifructus Roxb.
Baigouteng
Wubingguogouteng
HuaimianwangChina (Guangxi and Yunnan), India, Bangladesh, Bhutan, Myanmar, Nepal, northern Vietnam and Laos
Leaf slightly pink below; calyx lobes oblong, 1 mm long; corolla lobes densely sericeous outside (Chinese Pharmacopoeia Commission, 2020).
Uncaria sinensis (Oliv.) Havil.
Huagouteng
China (Sichuan, Guangxi, Yunnan, Hubei, Guizhou, Hunan, Shaanxi, Gansu)
Stipules entire or absent, broadly triangular or semicircular (Chinese Pharmacopoeia Commission, 2020).
4 Phytochemistry
To date, about 371 chemical constituents have been isolated from URCU, among which, alkaloids are considered the main constituents. Moreover, other reported secondary metabolites from URCU are terpenoids, flavonoids, phenylpropanoids, phytosterols, phenolics and other compounds.
Table 2 shows all phytochemicals isolated from URCU. The reported phytoconstituents included 169 alkaloids (1 ∼ 169), 89 terpenoids (170 ∼ 258), 30 flavonoids (259 ∼ 288), 18 phenylpropanoids (287 ∼ 306), 17 phytosterols (307 ∼ 323), 20 phenolics (324 ∼ 343), 28 other compounds (344 ∼ 371). Each phytochemical has been numbered from (1 ∼ 371) and cited in the text. The structures of chemical constituents have been illustrated in Figs. 3-17 according to the chemical classes. U1:Uncaria hirsuta Havil; U2:Uncaria macrophylla Wall; U3:Uncaria rhynchophylla (Miq.) Jacks; U4:Uncaria sessilifructus Roxb; U5:Uncaria sinensis (Oliv.) Havil. P1: stem and hook; P2: leaves; P3: stem; P4: the aerial part; P5: stem bark; P6: root.
No.
Compounds
From
Part
Ref.
1. Alkaloids
1.1 Indole alkaloids
1.1.1 Tetracyclic monoterpene indole alkaloids
1.
hirsutine
U3
U4
P1
P2
P4(Chi, 2017)(Liu, 2021)(Yu et al., 2022)
2.
hirsuteine
U3
U5
U2
P1
P2
P6(Guo et al., 2018)(Zhang et al., 2015)(Zhang et al., 2015)
3.
epi-allo-corynantheine
U3
P1
(Zhu et al., 1997)
4.
corynantheidine
U2
U3
P4
P1(Wang et al., 2011a)(Gong, 2021)
5.
18,19-dihydrocorynantheine
dihydrocorynantheine
U2
U3
P2
P4
P1
P2
P4(Liu, 2017)(Wang et al., 2011a)(Yu et al., 2021)(Ma et al., 2009b)(Kong et al., 2017)
6.
geissoschizine
U3
P1
(Gong, 2021)
7.
geissoschizine methyl ether
U3
P2
P1(Liu, 2021)(Chi, 2017)
P4
(Kong et al., 2017)
8.
villocarine A
U3
P2
P1(Liu, 2021)(Guo et al., 2018)
9.
corynantheine
U2
U3
P2
P1
P4(Liu, 2017)(Gong, 2021)(Kong et al., 2017)
10.
epi-allo-corynantheine
U2
P2
(Liu, 2017)
(3-ethenyl-1,2,3,4,6,7,12,12b-octahydro-α- (methoxymethylene)-methyl ester)
U3
P1
(Gong, 2021)
11.
uncanidine K
U4
P1
(Yu et al., 2022)
12.
17-O-ethylhirsutine
U4
P1
(Yu et al., 2022)
13.
Z‐geissoschizine
U3
P1
(Yu et al., 2021)
14.
uncarialin A
U3
P1
(Liang et al., 2019)
15.
indole [23-a] quinolizine-a-acetic acid
U3
P4
1P1(Kong et al., 2017)(Gong, 2021)
16.
O-(17)-demethyldihydrocorynantheine
19,20-dihydroisositsirkineU3
P2
P1(Kong et al., 2017)(Gong, 2021)
17.
dihydrositsirikine
U3
P2
(Liu, 2021)
18.
sitsirikine
U2
U3
P2
P1(Liu, 2017)(Guo et al., 2019)
19.
uncarialin E
U3
P1
(Liang et al., 2019)
20.
uncarialin F
U3
P1
(Liang et al., 2019)
21.
uncarialin G
U3
P1
(Liang et al., 2019)
22.
uncarialin H
U3
P1
(Liang et al., 2019)
23.
uncarialin K
U3
P1
(Yu et al., 2021)
24.
17-O-methyl-3,4,5,6-tetradehydrogeissoschizine
U3
P4
(Kong et al., 2017)
25.
rhynchophyllionium A
U3
P1
(Guo et al., 2018)
26.
rhynchophyllionium B
U3
P1
(Guo et al., 2018)
27.
rhynchophyllionium C
U3
P1
(Guo et al., 2018)
28.
rhynchophyllionium D
U3
P1
(Guo et al., 2018)
29.
vallesiachotamine
U3
P2
P4(Aimi et al., 1982)(Kong et al., 2017)
30.
uncarialin C
U3
P1
(Liang et al., 2019)
31.
uncarrhynchophylline A
U3
P1
(Li et al., 2021c)
32.
16R-E-Isositsirikine
U3
P1
(Gong, 2021)
33.
E-geissoschizine methyl ether
U3
P1
(Gong, 2021)
1.1.2 Tetracyclic monoterpenes oxidize indoles alkaloids
34.
rhynchophylline
U2
U3
U4
U5
U1
P4
P2
P6
P2
P1
P1
P1
P2(Wang et al., 2011a)(Phillipson and Hemingway, 1973)(Li et al., 2010)(Liu, 2021)(Liang et al., 2019)(Chi, 2017)(Liu et al., 1993b)(Xin et al., 2008b)
35.
corynoxinic B
U3
P1
(Gong, 2021)
36.
corynoxine B
U2
U3
P4
P2
P1(Wang et al., 2011a)(Phillipson and Hemingway, 1973)(Gong, 2021)
37.
corynoxeine
U2
U3
P1
P2
P2
P1(Liu, 2017)(Liu, 2021)(Chi, 2017)(Yu et al., 2021)
38.
9-hydroxy corynoxeine
U3
P1
(Xie et al., 2013)
39.
18,19-dehydrocorynoxinic acid
U3
P1
(Xie et al., 2013)
40.
isocorynoxeine
U2
U3
P2
P1
P2(Liu, 2017)(Wu et al., 2015)(Aimi et al., 1982)
41.
macrophylline A
U2
P4
(Wang et al., 2011a)
42.
rhynchophylloside B
U3
P1
(Guo et al., 2019)
43.
rhynchophyllic acid
U5
U3
P1
P1(Liu and Feng, 1993)(Gong, 2021)
44.
isorhynchophylline
U2
U3
U4
U1
P2
P4
P3
P6
P2
P1
P2
P2(Phillipson and Hemingway, 1973)
(Wang et al., 2011a)(Liang et al., 2021)(Li et al., 2010)(Liu, 2021)(Liang et al., 2019)(Liu et al., 1993b)(Xin et al., 2008b)
45.
corynoxine
U2
U4
U3
U1
P4
P3
P2
P1
P2(Wang et al., 2011a)(Liang et al., 2021)
(Phillipson and Hemingway, 1973)(Wu et al., 2007)(Xin et al., 2008b)
46.
isocorynoxine
U2
U3
P1
P2(Zhang et al., 2015)(Zhang et al., 2015)
47.
9-hydroxy isocorynoxeine
U3
P1
(Xie et al., 2013)
48.
18,19-dehydrocorynoxinic acid B
U3
P1
(Xie et al., 2013)
49.
22-O-demethyl-22-O-β-d-glucopynosylisocorynoxeine
U3
P2
(Ma et al., 2009b)
50.
isorhynchophyllic acid
U3
U5
P1
P1(Chi, 2017)(Liu and Feng, 1993)
51.
macrophylline B
U2
P4
(Wang et al., 2011a)
52.
macrophylline C
U2
P3
(Liang et al., 2021)
53.
macrophylline D
U2
P3
(Liang et al., 2021)
54.
uncarialin J
U3
P1
(Yu et al., 2021)
55.
5-oxo-isorhynchophylline
U3
P1
(Yu et al., 2021)
56.
macrophyllianium
U2
P4
(Wang et al., 2011a)
1.1.3 N-oxide tetracyclic monoterpene indole alkaloids
57.
hirsuteine N-oxide
U3
P1
(Guo et al., 2018)
58.
geissoschizine N-oxide methylether
U3
P2
P1(Liu, 2021)(Liang et al., 2019)
59.
uncarialin M
U3
P1
(Yu et al., 2021)
60.
uncarialin B
U3
P1
(Liang et al., 2019)
61.
hirsutine N-oxide
U3
P4
P1(Kong et al., 2017)(Liang et al., 2019)
62.
uncarrhynchophylline D
U3
P1
(Li et al., 2021c)
63.
uncarrhynchophylline E
U3
P1
(Li et al., 2021c)
64.
uncarialin I
U3
P1
(Liang et al., 2019)
65.
16-epi-isositsirikine (3S,4S)-N-oxide
U3
P1
(Liang et al., 2019)
66.
rhynchophylline N-oxide
U3
U5
P2
P1(Ma et al., 2009b)(Liu et al., 1993b)
67.
corynoxeine N-oxide
U3
P2
(Liu, 2021)
68.
isorhynchophylline N-oxide
U3
U5
P2
P1(Ma et al., 2009b)(Liu et al., 1993b)
69.
isocorynoxeine N-oxide
U3
P2
(Ma et al., 2009b)
1.1.4 Pentacyclic monoterpene indole alkaloids
70.
3-iso-19-epi-ajmalicine
U1
P2
(Xin et al., 2008b)
71.
akuammigine
U3
P1
(Guo et al., 2018)
72.
3-isoajmallicine
U1
P2
(Xin et al., 2008b)
73.
3β-isodihydrocadambine
U5
P1
(Zhang et al., 2015)
74.
3-isoajmalicine
U2
P2
(Liu, 2017)
75.
rhynchophylloside I
U3
P1
(Guo et al., 2019)
76.
tetrahydroalstonine
U4
U3
U5
P1
P1
P1(Yu et al., 2022)
(Liang et al., 2019)(Liu et al., 1993b)
77.
akuammigine
U4
U3
P1
P1(Yu et al., 2022)(Gong, 2021)
78.
vincoside lactam
(vincosamide)U3
P2
P1(Liu, 2021)(Xin et al., 2009a)
79.
rhynchophylloside F
U3
P1
(Guo et al., 2019)
80.
rhynchophylloside G
U3
P1
(Guo et al., 2019)
81.
2′-O-β-d-glucopyranosyl-11-hydroxyvincoside lactum
U3
P2
(Ma et al., 2009b)
82.
strictosamide
U3
P2
P1(Liu, 2021)(Wu et al., 2015)
83.
rhynchophine
U3
P2
(Aimi et al., 1982)
84.
rubescine
U3
P2
(Aimi et al., 1982)
85.
angustine
U3
P2
(Zhang et al., 2015)
86.
angustidine
U3
P2
(Zhang et al., 2015)
87.
(+)-(19S)-angustoline
U3
P2
(Zhang et al., 2015)
88.
β-yohimbine
U1
P2
(Kam et al., 1992)
89.
uncanidine J
U4
P1
(Yu et al., 2022)
90.
rhynchophylloside H
U3
P1
(Guo et al., 2019)
91.
vincosamide A
U3
P2
(Li, 2017)
92.
uncarialin L
U3
P1
(Yu et al., 2021)
93.
rhynchophylloside J
U3
P1
(Guo et al., 2019)
94.
α-yohimbine
U1
P2
(Kam et al., 1992)
95.
uncarrhynchophylline B
U3
P1
(Li et al., 2021c)
96.
uncarrhynchophylline C
U3
P1
(Li et al., 2021c)
1.1.5 Pentacyclic monoterpenes oxidize indole alkaloids
97.
uncarine F
U4
P2
(Zhang et al., 2015)
98.
uncarine B
U1
U2
P2
P2(Xin et al., 2008b)(Liu, 2017)
99.
uncarine C
(pteropodine)U5
U3
P1
P1(Liu et al., 1993b)(Gong, 2021)
100.
pteropodic acid
U5
P3
(Liu and Feng, 1993)
101.
mitraphylline
U1
U4
U5
U3
P2,
P1
P1
P1(Xin et al., 2008b)(Zhang, 2013)(Liu et al., 1993b)(Gong, 2021)
102.
mitraphyllic acid
U5
P2
P1(Liu et al., 1993a)(Liu and Feng, 1993)
103.
mitraphyllic acid (16–1)-β-d-glucopyranosylester
U5
P2
(Liu et al., 1993a)
104.
uncarine D
U5
P2
(Zhang et al., 2015)
105.
uncarine A
(isoformosanine)U1
U5
P2
P1
P1(Xin et al., 2008b)
(Lin et al., 2020)(Liu et al., 1993b)
106.
isopteropodic acid
U5
P1
(Liu and Feng, 1993)
107.
isomitraphylline
U1
U4
U3
P2
P1
P1(Xin et al., 2008b)(Zhang, 2013)(Gong, 2021)
108.
isomitraphyllic acid
U5
U1
P2
P2(Liu et al., 1993a)(Xin et al., 2008b)
109.
uncaric acid A
U1
P2
(Xin et al., 2008c)
110.
isomitraphyllic acid (16–1)-β-d-glucopyranosyl ester
U5
P2
(Liu et al., 1993a)
111.
uncarine E
(isopteropodine)U3
U5
P1
P1(Gong, 2021)(Liu et al., 1993b)
112.
rhynchophylloside E
U3
P1
(Guo et al., 2019)
113.
rhynchophylloside C
U3
P1
(Guo et al., 2019)
114.
rhynchophylloside D
U3
P1
(Guo et al., 2019)
115.
rhynchophylloside A
U3
P1
(Guo et al., 2019)
1.1.6 N-oxide pentacyclic monoterpene indole alkaloids
116.
uncarine D N-oxide
U5
P2
(Zhang et al., 2015)
117.
uncarine E N-oxide
U5
P2
(Zhang et al., 2015)
118.
isomitraphylline N-oxide
U1
U4
P2
P1(Zhang et al., 2015)(Zhang, 2013)
119.
uncarine B N-oxide
U1
P1
(Pan et al., 2017)
120.
uncarine F N-oxide
U5
P2
(Zhang et al., 2015)
121.
mitraphylline N-oxide
U1
U4
U5
P2
P2
P1(Zhang et al., 2015)
(Zhang et al., 2015)(Liu et al., 1993b)
122.
uncarine C N-oxide
U5
P2
P1(Zhang et al., 2015)(Liu et al., 1993b)
1.1.7 β-carboline alkaloids
123.
bahienoside A
U1
P2
(Xin et al., 2011)
124.
bahienoside B
U1
P2
(Xin et al., 2011)
125.
hirsutaside D
U1
P2
(Xin et al., 2011)
126.
neonaucleoside B
U1
P2
(Xin et al., 2011)
127.
vincoside
U3
P2
(Aimi et al., 1982)
128.
strictosidine
U3
P2
(Ma et al., 2009b)
129.
hirsutaside A
U1
P2
(Xin et al., 2008c)
130.
5β-carboxystrictosidine
U1
P1
(Lin et al., 2020)
131.
indol[2,3-a]quinolizidine
U3
P2
(Liu, 2021)
132.
1,2,3,4-tetrahydro-1-oxo-β-carboline (β-carboline alkaloid)
U3
P1
(Cai et al., 2019)
133.
harmane
U1
U3
P2
P1(Xin et al., 2008b)(Chi, 2017)
134.
croceaine B
U3
P2
(Liu, 2021)
135.
4β-hydroxyisodolichantoside
U3
P2
(Liu, 2021)
1.1.8 Cadambine alkaloids
136.
cadambine
U3
U5
P1
P1(Chi, 2017)(Endo et al., 1983)
137.
cadambinic acid
U3
P2
(Liu, 2021)
138.
3α-dihydrocadambine
U3
U5
P1
P1(Wu et al., 2015)(Endo et al., 1983)
139.
3β-dihydrocadambine
U5
P1
(Endo et al., 1983)
140.
3β-isodihydrocadambine
U5
P1
(Endo et al., 1983)
1.1.9 Dimeric isoechinulin-type alkaloids
141.
(+)-uncarilin A
U3
P1
(Geng et al., 2017)
142.
(+)-uncarilin B
U3
P1
(Geng et al., 2017)
143.
(-)-uncarilin B
U3
P1
(Geng et al., 2017)
144.
(-)-uncarilin A
U3
P1
(Geng et al., 2017)
1.1.10 Other indole alkaloids
145.
(+)-(7R)-3-oxo-7-hydroxy-3,7-seco-dihydrorhynchohylline
U3
P1
(Cai et al., 2019)
146.
(+)-(7S)-3-oxo-7-hydroxy-3,7-seco-dihydrorhyncho-hylline
U3
P1
(Cai et al., 2019)
147.
(+)-(7R)-3-oxo-7-hydroxy-3,7-seco-rhynchohylline
U3
P1
(Cai et al., 2019)
148.
(+)-(7S)-3-oxo-7-hydroxy-3,7-seco-rhynchohylline
U3
P1
(Cai et al., 2019)
149.
hirsutanine D
U1
P1
(Pan et al., 2017)
150.
hirsutanine E
U1
P1
(Pan et al., 2017)
151.
rhynchine A
U3
P1
(Zhou et al., 2021)
152.
rhynchine B
U3
P1
(Zhou et al., 2021)
153.
rhynchine C
U3
P1
(Zhou et al., 2021)
154.
rhynchine D
U3
P1
(Zhou et al., 2021)
155.
rhynchine E
U3
P1
(Zhou et al., 2021)
156.
salacin
U5
P2
(Zhang et al., 2015)
157.
uncarialin D
U3
P1
(Liang et al., 2019)
158.
uncanidine A
U3
P1
(Zhang et al., 2020)
1.2 Other alkaloids
159.
hirsutanine A
U1
P4
(Jia et al., 2014)
160.
hirsutanine B
U1
P4
(Jia et al., 2014)
161.
hirsutanine C
U1
P4
(Jia et al., 2014)
162.
hirsutanine F
U1
P1
(Pan et al., 2017)
163.
protopine
U3
P1
(Wu et al., 2015)
164.
venoterpine
U3
P1
(Yuan, 2022)
165.
(-)-N-methylcytisine
U3
P1
(Yuan, 2022)
166.
rhynchophylloside K
U3
P1
(Guo et al., 2019)
167.
rhynchophylloside L
U3
P1
(Guo et al., 2019)
168.
uncarrhynchoside A
U3
P1
(Li et al., 2021c)
169.
uncarrhynchoside B
U3
P1
(Li et al., 2021c)
2. Terpenoids
2.1 Triterpenoids
2.1.1 Ursane type triterpenoids
170.
ursolic acid
U2
U3
U4
U5
P1
P3
P6
P2
P1
P1
P1(Wu et al., 2007)(Yang, 2018)(Li et al., 2010)(Liu, 2021)(Chi, 2017)(Zhang, 2013)(Chen et al., 2014b)
171.
ursolic aldehyde
U5
P1
(Liu et al., 2011)
172.
α-amyrin acetate
U2
P1
(Wu et al., 2007)
173.
6β-hydroxyursolic acid
U3
P2
(Ma et al., 2009a)
174.
3β,6β,23-trihydroxyurs-12-en-28-oic acid
U2
U3
P1
P2(Wei et al., 2015)(Ma et al., 2009a)
175.
3β-hydroxyurs-12-en-27,28-diolic acid
U3
U2
P1
P1(Deng et al., 2009)(Wu et al., 2007)
176.
uncarinic acid C
U3
P1
(Yoshioka et al., 2016)
177.
uncarinic acid D
U3
P1
(Lee et al., 2000)
178.
quinovic acid
U1
U2
U4
U5
P2
P3
P1
P1(Xin et al., 2009b)(Wei et al., 2015)(Zhang, 2013)(Chen et al., 2014b)
179.
3-O-[β-D-glucopyranosy1]-quinovic acid
U4
U2
P1
P3(Zhang, 2013)(Wei et al., 2015)
180.
quinovic acid-3-O-β-d-fucopyranoside
U5
U2
U4
P1
P3
P3
P1(Chen et al., 2014b)(Wei et al., 2015)
(Fan et al., 2022)(Zhang, 2014)
181.
quinovic acid-3-O-β-d-glucopyranoide (28 → 1)-
β-d-glucopyranosideU5
P1
(Chen et al., 2014b)
182.
3-O-[β-d-quinovpyanosyl]-quinovic acid
U4
P1
(Zhang, 2013)
183.
3β,6β-dihydroxy-urs-12-en-28-oic acid
U3
P1
(Wei et al., 2015)
184.
3β-hydroxy-27-p-(Z)-coumaroyloxyursan-12-en-28-oic acid
U3
P1
(Lee et al., 2000)
185.
3β-hydroxy-27-p-(E)-coumaroyloxyursan-12-en-28-oic acid
U3
P1
(Zhang et al., 2014)
186.
3β,27-dihydroxy-urs-12-en-28-oic acid
U3
P1
(Zhang et al., 2014)
187.
2α-hydroxyursolic acid
U2
P3
(Yang, 2018)
188.
quinovic acid-3-β-O-β-6-deoxy-d-rhamnoside
U4
P1
(Zhang, 2014)
189.
3β,6β,19α,23,27-pentahydroxyures-12-en-28-oci acid
U4
P1
(Zhang, 2014)
190.
22α-hydroxy-3-oxo-urs-12-en-27,28-diolic acid
U1
P2
(Xin et al., 2009b)
191.
6β,19α-dihydroxy-3-oxo-urs-12-en-28-oic acid
U3
U2
P1
P3(Deng et al., 2009)(Wei et al., 2015)
192.
2-oxopomolic acid
U4
P1
(Zhang, 2014)
193.
3β,6β,19α-trihydroxyurs-12-en-28-oic acid
U2
U3
P3
P1(Sun et al., 2012a)(Deng et al., 2009)
U4
P1
(Zhang, 2013)
194.
3β,6β,19α-trihydroxy-23-oxo-urs-12-en-28-oic acid
U2
U3
U4P5
P1
P1(Sun et al., 2012b)(Zhang et al., 2014)(Zhang, 2013)
195.
3β,6β,19α-trihydroxy-urs-12-en-28-oicacid-24-carboxylicacidmethyl ester
U2
P5
(Sun et al., 2012a)
U4
P3
P1(Fan et al., 2022)(Zhang, 2014)
196.
3β,6β,19α-trihydroxy-23-methoxycarbonyl
-urs-12-en-28-oic acidU3
P1
(Zhang et al., 2014)
197.
uncarinic acid H
U3
P1
(Zhang et al., 2014)
198.
pomolic acid
U2
P3
(Wei et al., 2015)
199.
3β,6β,19α,23-tetrahydroxy-urs-12-en-28-oic acid
U4
U2
P1
P3(Zhang, 2013)(Wei et al., 2015)
200.
24-dimethoxymethyl-3β,6β,19α-trihydroxy −12-en-28-oic acid
U4
P3
(Fan et al., 2022)
201.
3β,6β,19α,24-tetrahydroxyurs-12-en-28-oic acid
U3
P1
(Deng et al., 2009)
U4
P1
(Zhang, 2014)
202.
3β,19α,24-trihydroxyurs-12-en-28-oic acid
U3
P1
(Deng et al., 2009)
203.
3β,6β,19α-trihydroxyurs-23-o-12-en-28-oic acid
U3
P1
(Deng et al., 2009)
204.
rotundic acid
U4
P1
(Zhang, 2014)
205.
uncarinic acid I
U3
P1
(Zhang et al., 2014)
206.
uncarisaside A
((3β)-3-(β-d-glucopyranosyloxy)-12-oxopyroquinovic acid β-d-glucopyranosyl ester)U1
P2
(Xin et al., 2009b)
207.
ursolic acid lactone
U3
P2
(Li, 2017)
208.
3β,6β-dihydroxy-urs-12,18-dien-28-oic acid
U2
P3
(Wei et al., 2015)
209.
uncargenin D
U2
P3
(Wei et al., 2015)
210.
uncarinic acid N
U3
P1
(Li et al., 2021b)
211.
uncarinic acid O
U3
P1
(Li et al., 2021b)
212.
uncarinic acid P
U3
P1
(Li et al., 2021b)
2.1.2 Oleanane type triterpenoids
213.
oleanoic acid
U2
U3
U5
P3
P1
P1(Yang, 2018)(Chi, 2017)(Chen et al., 2014b)
214.
uncarinic acid A
U3
P1
(Lee et al., 1999)
215.
uncarinic acid B
U3
P1
(Lee et al., 1999)
216.
uncarinic acid E
U3
P1
(Lee et al., 2000)
217.
3β-hydroxy-27-p-(E)-coumaroyloxyolean-12-en-28-oic acid
U3
P1
(Zhang et al., 2014)
218.
3β,6β-dihydroxy-olean-12-en-28-oic acid
U2
P3
(Wei et al., 2015)
219.
hederagenin
U2
P3
(Wei et al., 2015)
U1
P2
(Liu et al., 2021)
220.
3β,6β,23-trihydroxy-olean-12-en-28-oic acid
U2
U4
P3
P1(Wei et al., 2015)(Zhang, 2014)
221.
cincholic acid 3β-O-β-d-fucopyranoside
U4
P3
(Fan et al., 2022)
P1
(Zhang, 2014)
222.
β-amyrin-3-palmitate
U3
P2
(Liu, 2021)
223.
β-amnyrenol
U3
P1
(Chi, 2017)
224.
uncarinic acid F
U3
P1
(Zhang et al., 2014)
225.
uncarinic acid G
U3
P1
(Zhang et al., 2014)
226.
uncarinic acid J
U3
P1
(Zhang et al., 2014)
227.
3β,6β,19α-trihydroxy-olean-12-en-28-oic acid
U2
P3
(Wei et al., 2015)
228.
3β,6β,19α,23-tetrahydroxy-olean-12-en-28-oic acid
U2
P3
(Wei et al., 2015)
229.
phytolaccoside A
U5
P1
(Liu et al., 2011)
230.
sumresinolic acid
U3
P1
(Deng et al., 2009)
231.
uncargenin C
U3
P1
(Deng et al., 2009)
232.
3-oxo-olean-12-en-28-oic acid
U3
P1
(Shin and Lee, 2013)
233.
uncarinic acid M
U3
P1
(Li et al., 2021b)
234.
3β,19α,23-trihydroxy-6-oxo-olean-12-en-28-oic acid
U4
U3
P3
P1(Fan et al., 2022)(Deng et al., 2009)
235.
uncarilic acid
U4
P1
(Zhang, 2013)
236.
uncarinic acid L
U3
P1
(Li et al., 2021b)
237.
pyrocincholic acid
U1
P2
(Xin et al., 2009b)
238.
pyrocincholic acid ethyl ether
U1
P2
(Xin et al., 2009b)
239.
(3β)-3-(β-d-quinovopyranosyloxy)-
pyrocincholic acid-β-d-glucopyranosyl esterU1
P2
(Xin et al., 2009b)
240.
(3β)-hydroxy-27-norolean-13 (28)-lactone
U1
P2
(Xin et al., 2009b)
241.
3β,23-dihydroxy-12-oxo-olean-28,13β-olide
U2
P3
(Wei et al., 2015)
242.
secouncarilic acid
U4
P1
(Zhang, 2013)
243.
taraxerol
U2
P3
P6(Wei et al., 2015)(Li et al., 2010)
244.
uncarinic acid K
U3
P1
(Li et al., 2021b)
245.
myricadoil
U2
P6
(Li et al., 2010)
246.
friedelin
U4
P1
(Zhang, 2013)
2.1.3 Lupeol type triterpenoids
247.
obtusalin
U4
P1
(Zhang, 2013)
248.
betulin
U4
P1
(Zhang, 2013)
249.
lupenone
U2
P3
(Yang, 2018)
2.1.4 Cycloartenone
250.
24-en-cycloartenone
U4
P1
(Zhang, 2013)
2.1.5 Squalene
251.
squalene
U4
P1
(Zhang, 2013)
2.2 Sesquiterpenes
2.2.1 Megastigmanes
252.
uncarphyllonone A
U3
P1
(Song et al., 2022)
253.
uncarphyllonol A
U3
P1
(Song et al., 2022)
254.
uncarphyllonol B
U3
P1
(Song et al., 2022)
255.
uncarphabscisic acid A
U3
P1
(Song et al., 2022)
256.
uncarphabscisic acid B
U3
P1
(Song et al., 2022)
257.
(6R,9R) −9-hydroxymegastigman-4-en-3-one
U1
P2
(Liu et al., 2021)
2.2.2 Azulenoid
258.
(-)-alloaromadendrene
U4
P1
(Zhang, 2013)
2.3 Flavonoids
2.3.1 Flavonols
259.
kaempferol
U5
U3
U1
P1
P3
P2(Sun et al., 2012c)(Zhang et al., 2022)(Xin et al., 2008a)
260.
quercetin
U5
U3
U2
U1
P1
P1
P2
P2
P2(Sun et al., 2012c)(Chi, 2017)(Liu, 2021)(Liu, 2017)(Xin et al., 2008a)
261.
trifolin
U3
P2
(Aimi et al., 1982)
262.
hyperin
(quercetin-3-O-β-d-galactopyranoside)U1
U3
U5
P1
P2
P1
P1(Xin et al., 2008a)(Huang et al., 2019)(Chi, 2017)(Sun et al., 2012c)
263.
quercitrin
U1
U3
P2
P1(Xin et al., 2008a)(Li et al., 2017a)
264.
isoquercitrin
U1
P2
(Xin et al., 2008a)
265.
rutin
U1
U3
U2
P2
P1
P2
P2(Xin et al., 2008a)(Li et al., 2017a)(Ma et al., 2009a)(Liu, 2017)
266.
quercetin-3-O-robinobioside
U3
U5
P1
P2
P1(Li et al., 2017a)(Li, 2017)(Sun et al., 2012c)
267.
manghaslin
U1
P2
(Xin et al., 2008a)
268.
(+)-uncariols C
U3
P1
(Li et al., 2017a)
269.
(-)-uncariols C
U3
P1
(Li et al., 2017a)
270.
(+)-uncariols D
U3
P1
(Li et al., 2017a)
271.
(-)-uncariols D
U3
P1
(Li et al., 2017a)
272.
afzelin
U1
P2
(Xin et al., 2008a)
273.
kaemferol-3-O-β-d-galactopyranoside
U3
P2
(Ma et al., 2009a)
274.
kaemferol-3-O-β-d-galactopyranosyl-(6–1)-α-L
-rhamnopyranosideU3
P2
(Ma et al., 2009a)
2.3.2 Flavones
275.
buddleoside (linarin)
U5
P1
(Sun et al., 2012c)
2.3.3 Flavan-3-ols
276.
(+)-catechin
U3
U4
P1
P1(Hou et al., 2005)(Zhang, 2014)
277.
(-)-epicatechin
U1
U3
U2
P2
P1
P2
P3(Xin et al., 2008a)(Li et al., 2017a)
(Li, 2017)(Yang, 2018)
278.
uncariol A
U3
P1
(Li et al., 2017a)
279.
uncariol B
U3
P1
(Li et al., 2017a)
280.
cinchonain Ia
U3
P1
(Li et al., 2017a)
281.
cinchonain Ib
U3
P1
(Li et al., 2017a)
282.
cinchonain Ic
U3
P1
(Li et al., 2017a)
283.
cinchonain Id
U3
P1
(Li et al., 2017a)
2.3.4 Homoisoflavone
284.
3-(3-hydroxy-4-methoxybenzyl)-5,7-
dihydroxychroman-4-oneU2
P3
(Yang, 2018)
2.3.5 Chromone
285.
eugenin
U4
U5
P1
P1(Zhang, 2013)(Liu et al., 2011)
286.
noreugenin
U3
P1
(Deng et al., 2009)
287.
2-methyl-5,7-dihydroxy-chromone-
7-O-β-d-glucopyranosideU1
P2
(Liu et al., 2021)
2.3.6 Flavanone
288.
neohesperidin
U1
P2
(Wu and Chan, 1994)
2.4 Phenylpropanoids
2.4.1 Simple phenylpropanoids
289.
trans-anethole
U3
P1
(Shin and Lee, 2013)
290.
p-anisaldehyde
U3
P1
(Shin and Lee, 2013)
2.4.2 Coumarins
291.
umbelliferone
(7-hydroxycoumarin)U1
U4
U2
P2
P1
P2(Wu and Chan, 1994)(Zhang, 2014)(Liu, 2017)
292.
scopoletin
U2
U4
U5
U3
P3
P1
P1
P1(Yang, 2018)(Zhang, 2013)(Liu et al., 2011)(Chi, 2017)
293.
5-hydroxy-7-methoxycoumarin
U4
P1
(Zhang, 2014)
294.
cleomiscosin B
U3
P1
(Deng et al., 2009)
295.
cleomiscosin D
U3
P1
(Deng et al., 2009)
2.4.3 Lignans
296.
(2R,3R,4S)-lyoniresinol-3α-O-β-d-glucopyra-noside
U5
P1
(Sun et al., 2011)
297.
(2R,3S,4R)-lyoniresinol-3α-O-β-d-glucopyra-noside
U5
P1
(Sun et al., 2011)
298.
(2S,3S,4R)-lyoniresinol-3α-O-β-d-glucopyra-noside
U5
P1
(Sun et al., 2011)
299.
(2S,3R,4S)-lyoniresinol-3α-O-β-d-glucopyra-noside
U5
P1
(Sun et al., 2011)
300.
(+)-lyoniresinol
U3
P3
(Zhang et al., 2022)
301.
(–)-lyoniresinol
U3
P3
(Zhang et al., 2022)
302.
isolariciresinol
U3
P3
(Zhang et al., 2022)
Neolignans
303.
(-)-(7S,8R)-dihydrodehydrodiconiferyalcohol
U3
P1
(Zhang et al., 2010)
304.
leptolepisol D
U3
P3
(Zhang et al., 2022)
305.
leptolepisol C
U3
P3
(Zhang et al., 2022)
306.
Threo-3,3′-dimethoxy-4,8′-oxyneoligna-9,4′,7′,9′-tetraol-7(8)
-eneU3
P3
(Zhang et al., 2022)
2.5 Phytosterols
2.5.1 Sitosterols
307.
β-sitosterol
U4
U3
U2
U5
P1
P1
P2
P3
P2
P1
P6
P1(Zhang, 2013)(Chi, 2017)(Ma et al., 2009a)(Yang, 2018)(Liu, 2017)(Wu, 2007)(Li et al., 2010)(Chen et al., 2014b)
308.
β-daucosterol
U4
U3
U2
U5
U1
P1
P1
P2
P3
P1
P1
P1(Zhang, 2013)(Chi, 2017)(Ma et al., 2009a)(Yang, 2018)(Wu, 2007)(Chen et al., 2014b)(Xin et al., 2008a)
309.
sitost-5-ene-3β,7β-diol (7β-hydroxysitosterol)
U3
P1
(Liu et al., 2022)
310.
sitost-5-ene-3β,7α-diol (7α-hydroxysitosterol)
U3
P1
(Liu et al., 2022)
2.5.2 Stigmasterols
311.
(24S)-stigmast-4-en-3-one
U4
P1
(Zhang, 2013)
312.
(24S)-stigmasta-4-en-6β,7α-diol-3-one
U3
P1
(Liu et al., 2022)
313.
stigmasterol
U3
P1
(Duan, 2010)
314.
(24S)- stigmasta-4-en-3β,6β-diol
U3
P1
(Liu et al., 2022)
315.
(24S)- stigmasta-4-en-3β,6α-diol
U3
P1
(Liu et al., 2022)
316.
(24S)-stigmasta-3β,6α-diol
U3
P1
(Liu et al., 2022)
317.
(24S)-stigmasta-3β,5α,6β-triol
U3
P1
(Liu et al., 2022)
U5
P1
(Chen et al., 2014b)
2.5.3 Ergosterols
318.
(22E,24R)-ergosta-7,22-diene-3β,5α,6β-triol
U3
P1
(Liu et al., 2022)
319.
(22E,24R)-6β-methoxyergosta-7,22-diene-3β,5α-diol
U3
P1
(Liu et al., 2022)
320.
(22E,24R)-ergosta-7,9(11),22-triene-3β,5β,6α-triol
U3
P1
(Liu et al., 2022)
321.
(22E,24R)-ergosta-7,22-dien-3β,5α,6α-triol
U3
P1
(Liu et al., 2022)
322.
(22E,24R)-ergosta-7,22-dien-3β,5α- diol-6-one
U3
P1
(Liu et al., 2022)
323.
(22E,24R)-ergosta-7,22-dien-3β,5α-diol-6,5-olide
U3
P1
(Liu et al., 2022)
2.6 Phenolics
324.
ethyl 3,4-dihydroxybenzoate
U2
U3
P3
P1(Yang, 2018)(Chi, 2017)
325.
vanillic acid
U2
U3
P3
P1(Yang, 2018)(Duan, 2010)
326.
3-hydroxy-5-methoxybenzoic acid
U4
P1
(Zhang, 2014)
327.
1,3,5-trimethoxybenzene
U2
U3
P3
P1(Yang, 2018)(Yuan, 2022)
328.
p-hydroxybenzoic acid
U3
P2
(Liu, 2021)
329.
syringic acid
U3
P2
P1(Liu, 2021)(Deng et al., 2009)
330.
p-dihydroxyhenzene
U3
P1
(Duan, 2010)
331.
protocatechuic acid
U3
P2
(Li, 2017)
U4
P1
(Zhang, 2014)
332.
4-methyl-phenol
U4
P3
(Fan et al., 2022)
333.
estragole
U3
P1
(Shin and Lee, 2013)
334.
1,2,3-trihydroxyphenol
U4
P1
(Zhang, 2014)
335.
3,4,5-trimethoxybenzene
U1
P1
(Liu et al., 2021)
Phenolic acids
336.
caffeic acid
U2
P2
(Liu, 2017)
337.
ethyl cafficate
U3
P1
(Chi, 2017)
338.
p-coumaric acid ethyl ester
U3
P3
(Zhang et al., 2022)
339.
methyl caffeate
U3
P2
(Li, 2017)
340.
methylrosmarinate
U3
P1
(Chi, 2017)
341.
chlorogenic acid
U3
U1
P2
P1
P2(Huang, 2019)
(Lin et al., 2020)(Xin et al., 2008a)
342.
chlorogenic acid ethyl ester
U3
P2
(Li, 2017)
343.
1,2-bis(4-hydroxy-3-methoxyphenyl)-1,3-propanediol
U3
P3
(Zhang et al., 2022)
2.7 Other compounds
344.
1-methoxyoctadecan-1-ol
U5
P1
(Ahn et al., 2014)
345.
vitamin E
U4
P1
(Zhang, 2013)
346.
α-tocopherolquinone
U4
P1
(Zhang, 2013)
347.
α-tocopherol
U3
P2
(Li, 2017)
348.
dihydroactinidiolide
U3
P2
(Li, 2017)
349.
palmitic acid
U3
P1
(Chi, 2017)
350.
tetracosane
U3
P1
(Chi, 2017)
351.
glycerol monopalmtate
U3
P1
(Deng et al., 2009)
352.
O-β-d-fructofuranosyl-(2 → 6)-α-d-glucopyranosyl-(1 → 6)-β-d-fructofuranosyl-(2 → 6)-β-d-fructofuranosyl-(2 → 1)-α-D- glucopyranosyl-(6 → 2)-β-D- fructofuranoside
U3
P2
(Liu, 2021)
353.
2-phenethyl-O-α-Lrhamnopyranosyl-(1 → 6)-
β-d-glucopyranosideU3
P3
(Zhang et al., 2022)
354.
1,2:4,5-di-O-isoproylidene-β-d-fructopyranose
U2
P3
(Yang, 2018)
355.
3,4-dehydrotheaspirone
U3
P2
(Li, 2017)
356.
chakyunglupulin A
U3
P2
(Li, 2017)
357.
mannitol
U2
P3
(Yang, 2018)
358.
sucrose
U3
P2
(Liu, 2021)
359.
maackiain
U3
P1
(Yuan, 2022)
360.
(-)-(7S,8R)-dihydrodehydrodiconiferyalcohol
U4
P3
(Fan et al., 2022)
361.
vomifoliol
U3
P3
(Zhang et al., 2022)
362.
dibutyl phthalate
U3
P2
(Liu, 2021)
363.
bis(2-ethylhexyl)phthalate
U4
P1
(Zhang, 2013)
364.
erythroglaucin
U3
P1
(Chi, 2017)
365.
rheochrysidin (physcione)
U3
P1
(Chi, 2017)
366.
uncarophyllofolic acid A
U3
P1
(Wang et al., 2019b)
367.
uncarophyllofolic acid B
U3
P1
(Wang et al., 2019b)
368.
3-diethylamino-5-methoxy-1,2-benzoquinone
U3
P1
(Zhang et al., 2016)
369.
3-ethylamino-5-methoxy-1,2-benzoquinone
U3
P1
(Zhang et al., 2016)
370.
semiphorone
U3
P1
(Gong, 2021)
371.
4-hydorxy-4-methyl-2-pentanone
U3
P1
(Gong, 2021)
Structures of tetracyclic monoterpene indole alkaloids in URCU.
Structures of tetracyclic monoterpenes oxidize indoles alkaloids in URCU.
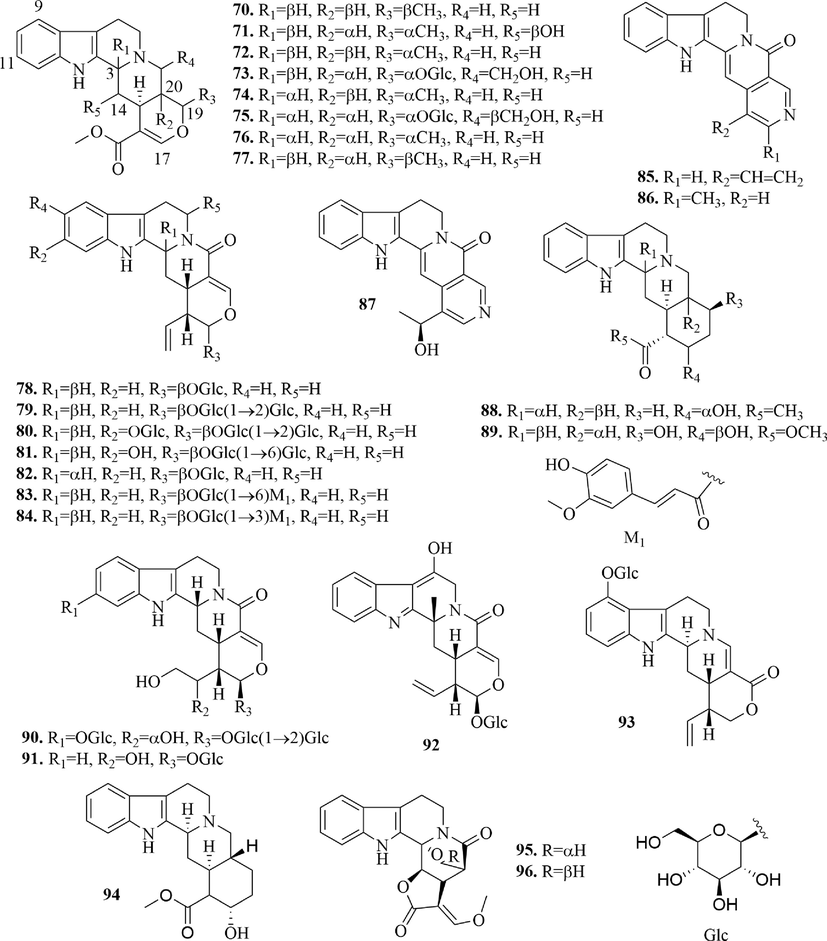
Structures of pentacyclic monoterpene indole alkaloids in URCU.
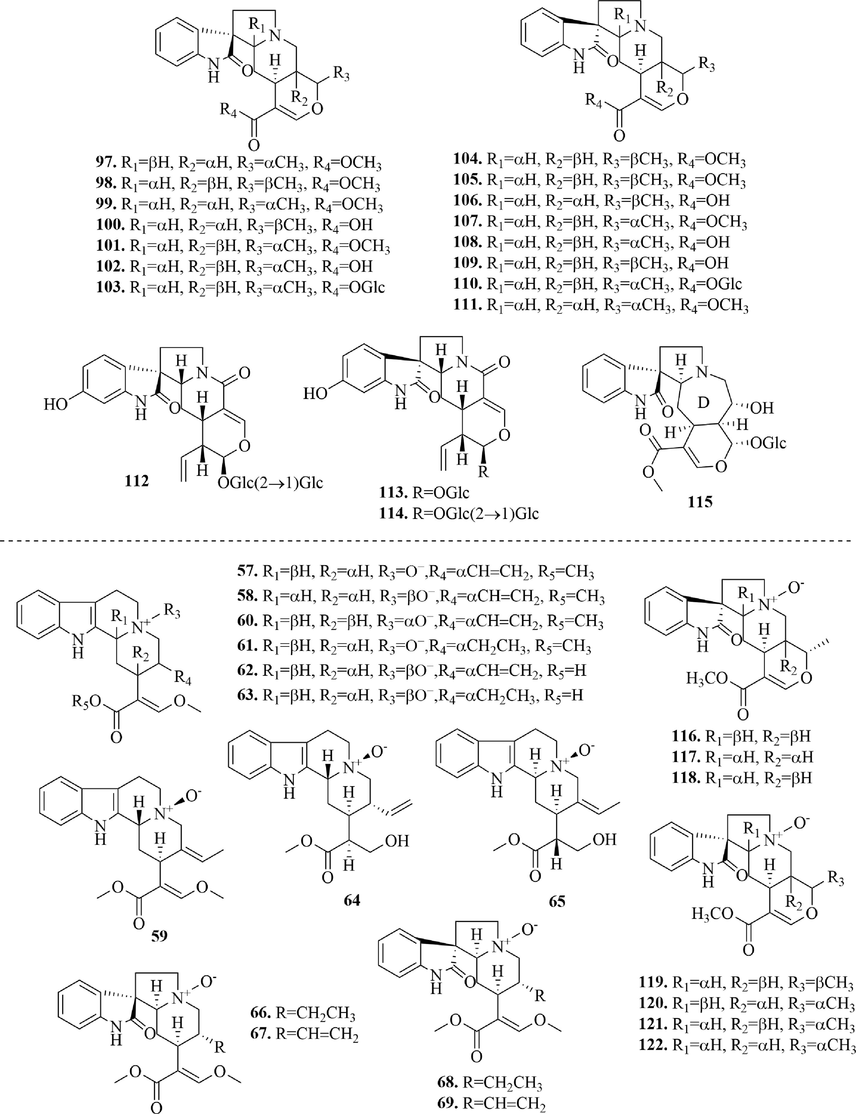
Structures of pentacyclic monoterpenes oxidize indole alkaloids and N-oxide monoterpene indole alkaloids in URCU.
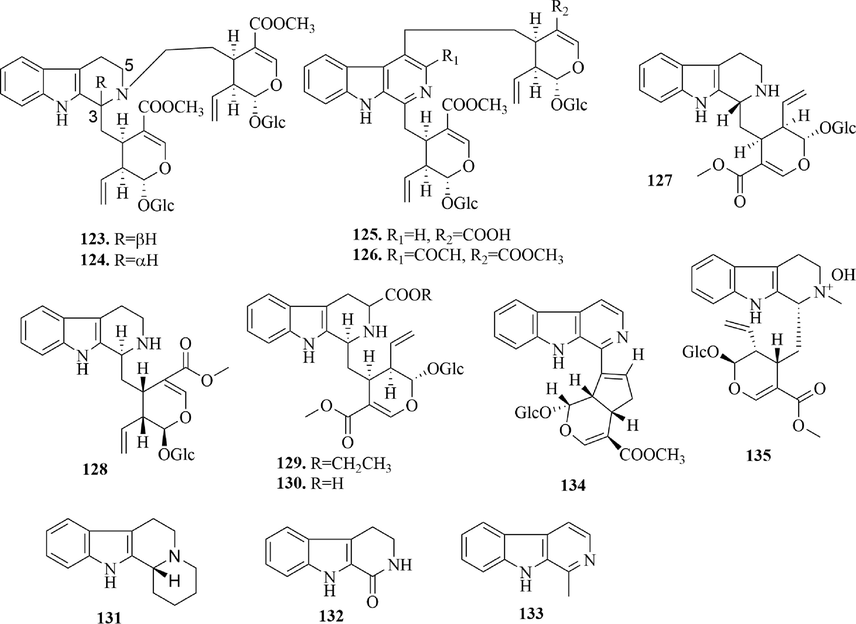
Structures of β-carboline alkaloids in URCU.
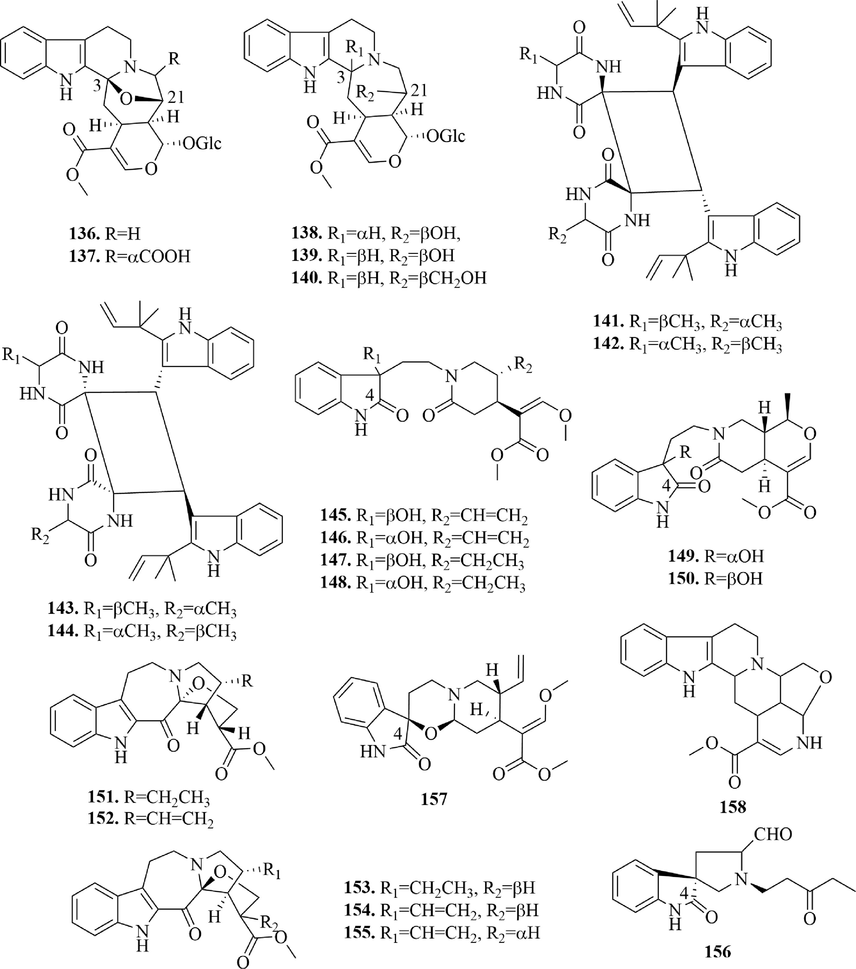
Structures of cadambine alkaloids, dimeric isoechinulin-type alkaloids and other indole alkaloids in URCU.
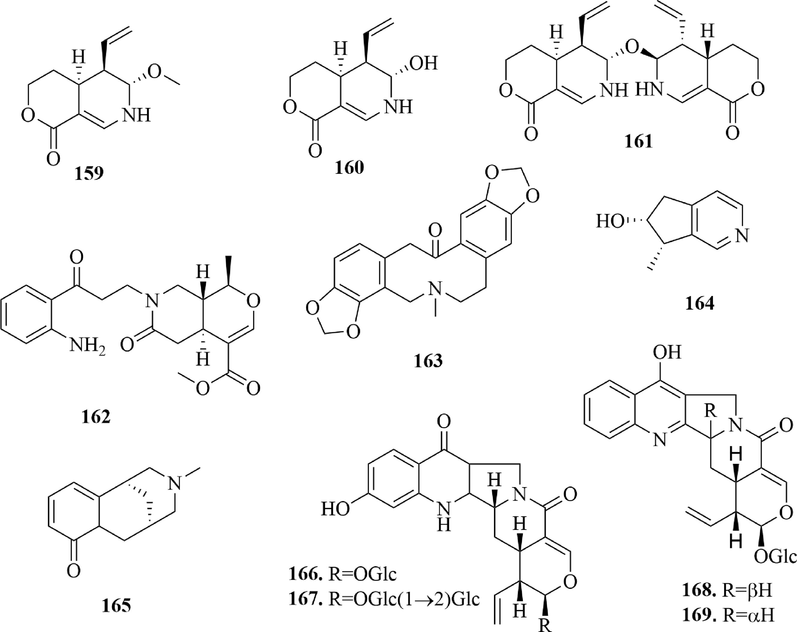
Structures of other alkaloids in URCU.
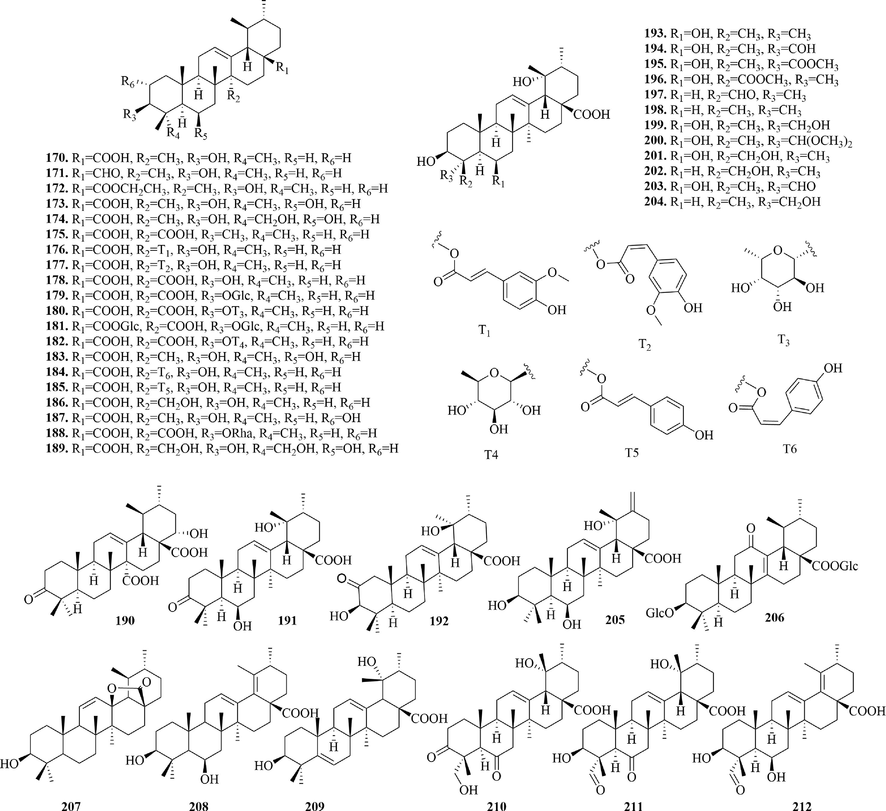
Structures of ursane type triterpenoids in URCU.
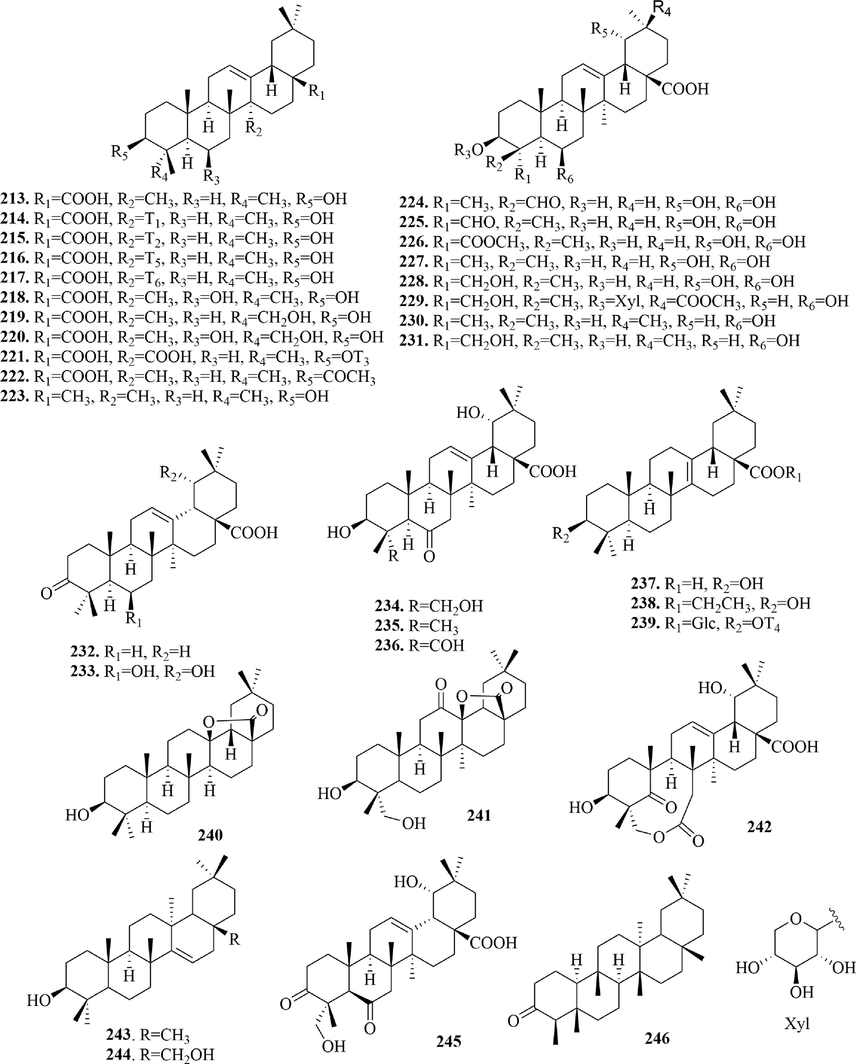
Structures of oleanane type triterpenoids in URCU.
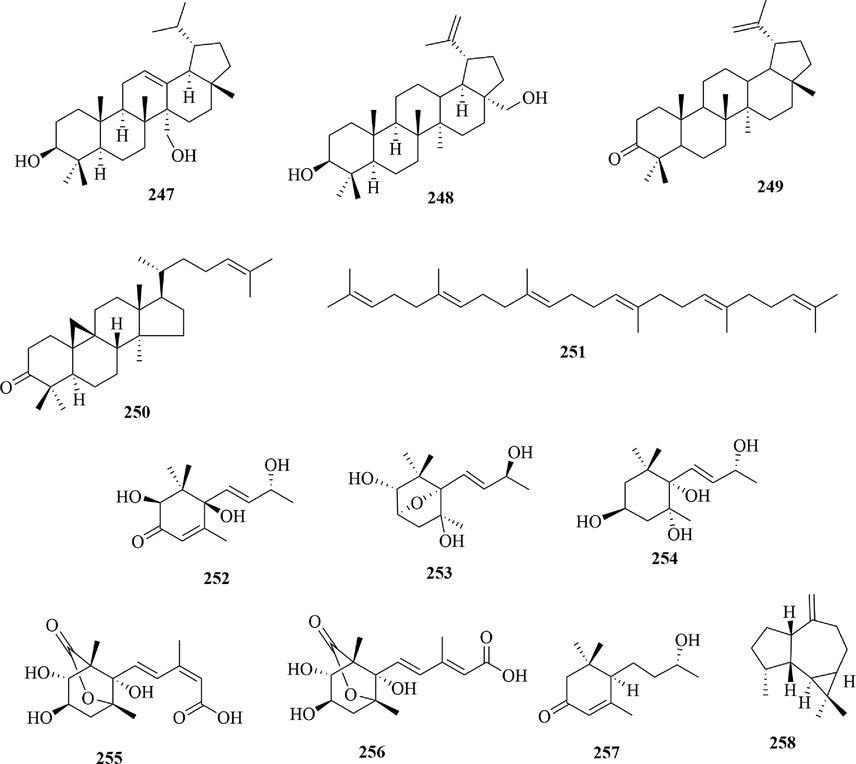
Structures of other triterpenoids and sesquiterpenes in URCU.
Structures of flavonoids in URCU.
Structures of phenylpropanoids in URCU.
Structures of phytosterols in URCU.
Structures of phenolics in URCU.
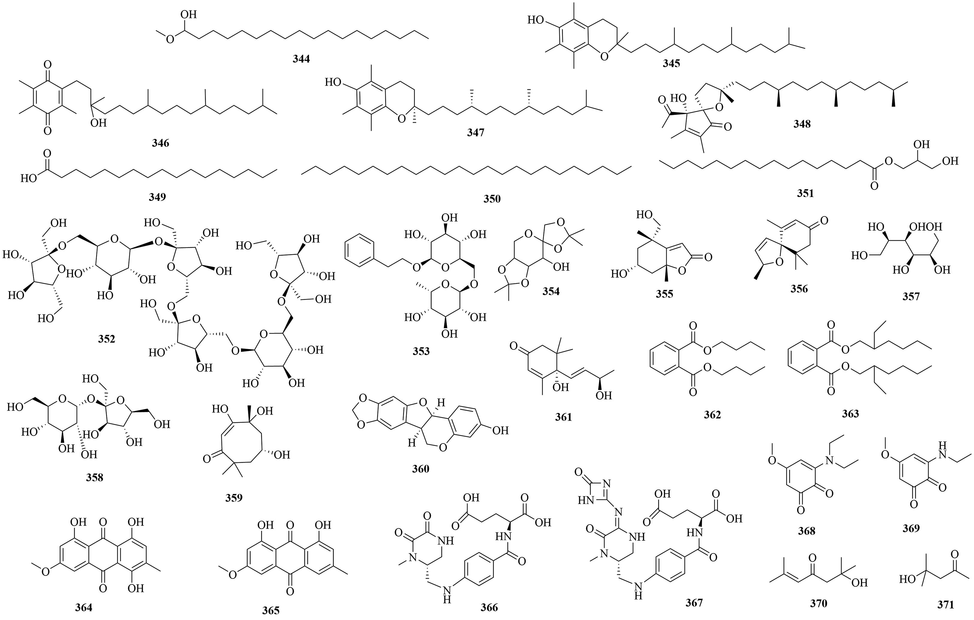
Structures of other compounds in URCU.
4.1 Alkaloids
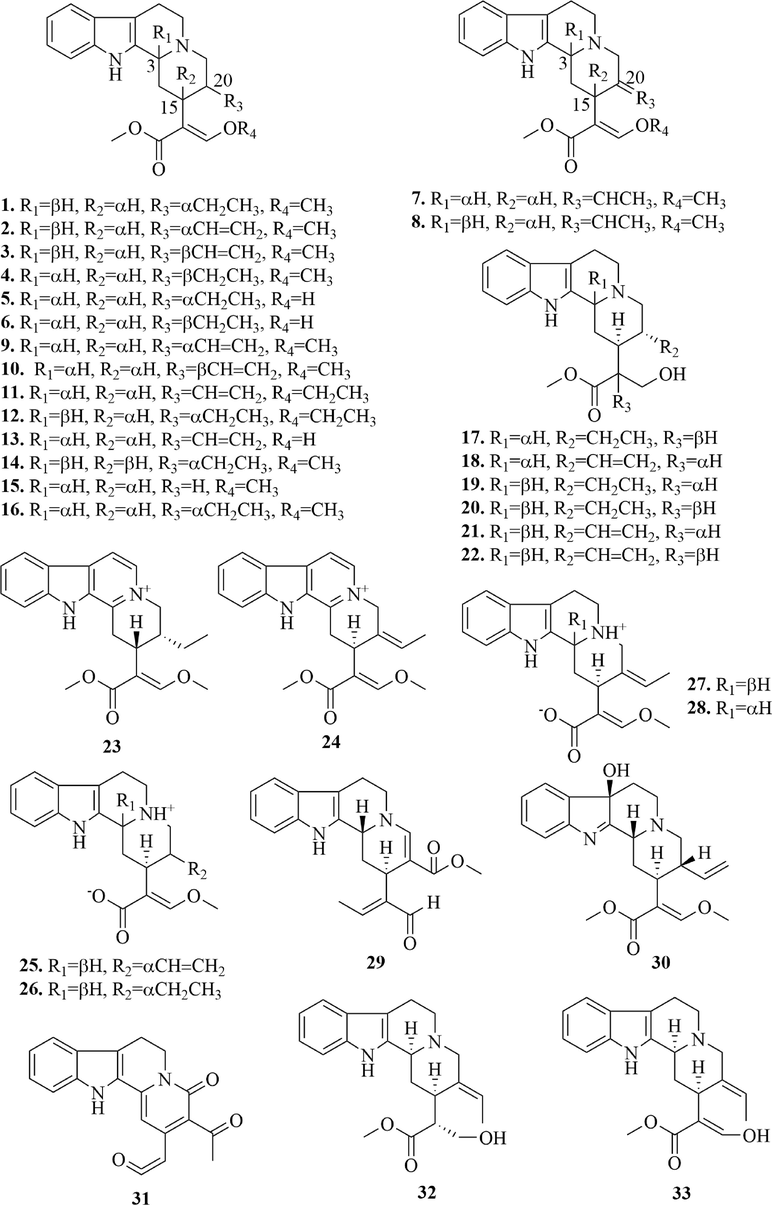
Currently, more than 169 alkaloids have been isolated and identified from URCU, among which, indole alkaloids were the main alkaloids. There were 158 indole alkaloids, which included 122 monoterpene indole alkaloids, 13 β-carboline alkaloids, 5 cadambine alkaloids, 4 dimeric isoechinulin-type alkaloids and 14 other indole alkaloids. The specific structures of compounds were shown in Figs. 3-9.
4.1.1 Monoterpene indole alkaloids
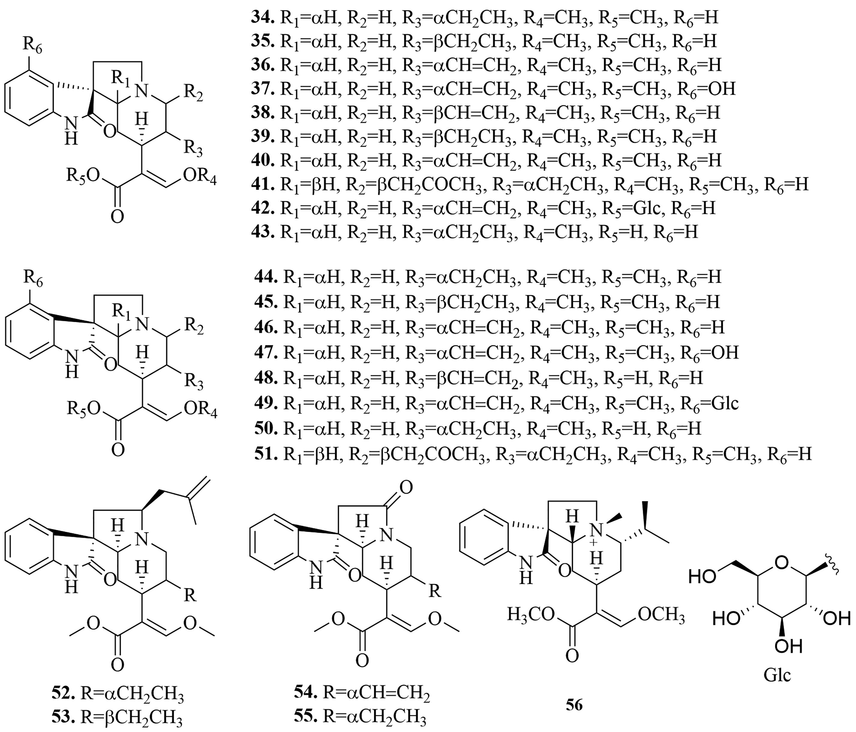
Monoterpene indole alkaloids are also known as secoiridoid alkaloids, whose basic skeleton is formed by the manish reaction of secologanin and tryptamine. According to the skeleton type and oxidation state, they can be divided into tetracyclic monoterpene indole alkaloids (1 ∼ 33), tetracyclic monoterpenes oxidize indoles alkaloids (34 ∼ 56), N-oxide tetracyclic monoterpene indole alkaloids (57 ∼ 69), pentacyclic monoterpene indole alkaloids (70 ∼ 96), pentacyclic monoterpenes oxidize indole alkaloids (97 ∼ 115), N-oxide pentacyclic monoterpene indole alkaloids (116 ∼ 122). The specific structures were shown in Figs. 3-6.
(1) Monoterpene indole alkaloids
33 tetracyclic monoterpene indole alkaloids (1 ∼ 33) (Fig. 3) and 27 pentacyclic monoterpene indole alkaloids (70 ∼ 96) (Fig. 5) were reported from URCU. Tetracyclic monoterpene indole alkaloid’s 15-position was mostly α-H and the 20-position mostly had ethylene or ether, which may be related to the secologanin in the synthesis pathway. Pentacyclic monoterpene indole alkaloids mostly formed glycosides at hydroxyl group of 17 or 19-position. Whereas rhynchophylloside J (93) formed glycosides at 9-position hydroxyl and rhynchophylloside H (90) formed glycosides at 11 and 17-position hydroxyl groups. In addition, it’s worth noting that tetracyclic monoterpene indole alkaloid didn’t form glycoside. Uncarrhynchophylline A (31) was a monoterpene 22-norindoloquinolizidine alkaloid with a unique ketene unit and uncarrhynchophylline B (95) and uncarrhynchophylline C (96) were a pair of monoterpene indoloquinolizidinealkaloid epimers possessing an oxygen-bridge between C-3 and C-19 to form an oxazinane ring. Meanwhile, the E-ring is a five-membered lactone ring.
(2) Oxidized monoterpene indole alkaloids
Oxidized monoterpene indole alkaloids are the 2-position oxidation of monoterpene indole alkaloids, which is a typical feature of Uncaria alkaloids. At present, 23 tetracyclic monoterpenes oxidize indole alkaloids (34 ∼ 56) (Fig. 4) and 19 pentacyclic monoterpenes oxidize indole alkaloids (97 ∼ 115) (Fig. 6) were isolated from URCU. Rhynchophylloside A (1 1 5) represented a new subtype of oxindole alkaloid with a seven-membered d-ring, rhynchophylloside D (1 1 4) and E (1 1 2) were the two oxindole alkaloid diglycosides, which were firstly isolated from the genus Uncaria.
(3) N-oxide monoterpene indole alkaloids
N-oxide monoterpene indole alkaloids are nitrogen oxides oxidized from N-4 in monoterpene indole alkaloids. 13 N-oxide tetracyclic monoterpene indole alkaloids (57 ∼ 69) and 7 N-oxide pentacyclic monoterpene indole alkaloids (116 ∼ 122) (Fig. 6) were reported in this article.
4.1.2 β-carboline alkaloids
Carboline alkaloid is a kind of alkaloid with a pyridylindole structure, which can be divided into α, β, γ, and δ-carboline according to different cyclization methods. In URCU, all carboline alkaloids were β-carboline alkaloids (123 ∼ 135) (Fig. 7) and all substitutions occur in C-ring. By observing the structure, we found that most of the substitutions were at 3-position. Meanwhile, some β-carboline alkaloids had secologanin at the 3-position, whose formation might be related to the carbon bond cleavage between the 3 and 21-position of pentacyclic triterpenoids.
4.1.3 Cadambine alkaloids
Cadambine alkaloid is a kind of pentacyclic indole alkaloid, whose d-ring is a heptatomic ring. 5 cadambine alkaloids (136 ∼ 140) were reported in this article (Fig. 8). Among cadambine alkaloids, the hydroxyl groups at 3 and 21-position of cadambine (1 3 6) and cadambinic acid (1 3 7) formed an oxygen bridge.
4.1.4 Dimeric isoechinulin-type alkaloids
(±)-Uncarilin A and (±)-uncarilin B (141 ∼ 144), two pairs of unusual dimeric isoechinulin-type enantiomers, were isolated from U. rhynchophylla (Fig. 8), which contained two characteristic units: indole and diketopiperazine. Geng et al. thought the diketopiperazine core was condensed by “head to tail” cyclization of tryptophan and alanine. Subsequent incorporation of mevalonic acid afforded neoechinulin A, which was transformed to yield compounds 141 ∼ 144 via intermolecular [2 + 2] cycloaddition. And, they thought the formation of this kind of compound might be related to the endophytic fungus in U. rhynchophylla.
4.1.5 Other indole alkaloids
14 other indole alkaloids (145 ∼ 158) (Fig. 8) were reported from URCU. Among other indole alkaloids, compounds 145 ∼ 150 and 156 ∼ 157 were oxindoles, which had carbonyl in 4-position. Notably, hirsutanine D and E (149 ∼ 150) were two 3-oxo-3,7-seco-oxindole alkaloids. Moreover, rhynchine A-E (151 ∼ 155) were five new indole alkaloids with an unprecedented skeleton. The new skeleton was characterized by an indole moiety and a 2-oxa-8-azatricyclo[6,5,01,5,01,8]tridecane core, forming a unique 6/5/7/5/5 ring system. Rhynchophyllosides K-L (166 ∼ 167) were two alkaloids with a quinolone nucleus. Meanwhile, uncanidine A (1 5 8) was a novel Uncaria alkaloid which possessed a 6/5/6/6/6/5 hexacyclic ring system.
4.1.6 Other alkaloids
In addition to indole alkaloids, 11 other alkaloids (159 ∼ 169) (Fig. 9) were reported from URCU. Hirsutanine A-C (159 ∼ 161) were three monoterpenoid alkaloids, which were isolated from U. hirsuta. Notably, hirsutanine C (1 6 1) was the first dimeric monoterpenoid alkaloid obtained from the genus Uncaria. Uncarrhynchoside A and B (168 ∼ 169) were rare camptothecin-related monoterpene alkaloids from the Uncaria plants. Meanwhile, hirsutanine F (1 6 2) is the first 3-oxo-3,7-seco-oxindole alkaloid with ring B opened and degraded isolated from the Uncaria genus.
4.2 Terpenoids
Terpenoids are compounds derived from mevalonic acid with (C5H8)n general formula. Terpenoids, which were reported from URCU, can be divided into triterpenoids (170 ∼ 251) and sesquiterpene (252–258). 82 triterpenoids included 43 ursane type triterpenoids, 34 oleanane type triterpenoids, 3 lupeol type triterpenoids, 1 cycloartane and 1 squalene. The specific structures were shown in Figs. 10-12.
4.2.1 Ursane type triterpenoids
Ursane type triterpenoids (170 ∼ 212) (Fig. 10) are also known as α-aromatic resin, whose basic skeleton is a pentacyclic nucleus of polyhydropinene with a gem-dimethyl at 4-position, and a methyl substitution at 19 and 20-position, respectively. According to the number and location of double keys, they can be divided into Δ12 ursane type (mostly), Δ13 ursane type, Δ5, 12 ursane type and Δ12, 18 ursane type. Most ursane type triterpenoids from URCU had carboxyl at 17-position and β-hydroxyl substitution at 3-position. Compounds 179 ∼ 180, 182 and 188 formed saponins at the C-3 hydroxyl group with different sugars, respectively. Whereas, compound 181 and uncarisaside A (2 0 6) formed saponins with glucose at both 3-position hydroxyl group and 17-position carboxyl group. Compound 206 was a special 12-oxo ursane type triterpenoid. Notably, ursolic acid lactone (2 0 7) formed a pentalactone ring at the 13-position of hydroxyl group and at the 17-position of carboxyl group.
4.2.2 Oleanane type triterpenoids
Oleanane type triterpenoids (213 ∼ 246) (Fig. 11) are also known as β-aromatic resin, whose basic skeleton is a pentacyclic nucleus of polyhydropinene with a gem-dimethyl at 4 and 20-position, respectively. In this article, most oleanane type triterpenoids had carboxyl at 17-position and β-hydroxyl substitution at 3-position. According to the number and location of double keys, they also could be divided into Δ12 oleanane type (mostly), Δ13 oleanane type, Δ14 oleanane type and double bond-free oleanane type. In general, oleanane type triterpenoids exist in the form of saponins. But in URCU, only cincholic acid 3-O-β-d-fucopyranoside (2 2 1), phytolaccoside A (2 2 9) and (3β)-3-(β-d-quinovopyranosyloxy)-pyrocincholic acid-β- d-glucopyranosyl ester (2 3 9) were saponins. It’s worth noting that (3β)-hydroxy-27- norolean-13(28)-lactone (2 4 0) and 3β,23-dihydroxy-12-oxo-olean-28,13β-olide (2 4 1) both formed a pentalactone ring by the dehydration condensation reaction of the hydroxyl group at C-13 position and the carboxyl group at 17-position. In addition, secouncarilic acid (2 4 2) was the first oleanane-type 5,6-secotriterpenoid, which had a nine-membered lactone ring.
4.2.3 Lupeol type triterpenoids
Lupeol type triterpenoid is a kind of triterpenoid, whose five rings are all trans-condensed. Especially, the lupeol type triterpenoid’s E-ring is a five-membered ring. Meanwhile, it has an α-isopropyl substitution at the 19-position of the E-ring. At present, three lupeol type triterpenoids were isolated from URCU including obtusalin (2 4 7), betulin (2 4 8) and lupenone (2 4 9) (Fig. 12).
4.2.4 Sesquiterpenes
Sesquiterpenes (252 ∼ 258) are a class of terpenoids composed of three isoprene units (15 carbons) (Fig. 12). In URCU, 7 sesquiterpenes were reported, which included 6 megastigmanes (252 ∼ 257) and 1 azulenoid (2 5 8). Megastigmane, also known as lonone, is a kind of monocyclic sesquiterpene. In general, most megastigmanes have a 1,1-dimethylcyclohexane (alkene) structure and exist in the form of glycoside, whereas all megastigmanes in this article are aglycones. Notably, compounds 252 ∼ 254 and 257 lost two carbons in the decarboxylation reaction of source synthesis, so they were special norsesquiterpenoids. Uncarphabscisic acid A (2 5 5) and uncarphabscisic acid B (2 5 6) formed a pentalactone ring. In addition, azulenoid is an aromatic derivative synthesized by the parallel synthesis of a five-membered ring and seven-membered ring, and its molecular structure has a high conjugated system. At present, only one azulenoid, (-)-alloaromadendrene (2 5 8), was isolated from U. sessilifructus.
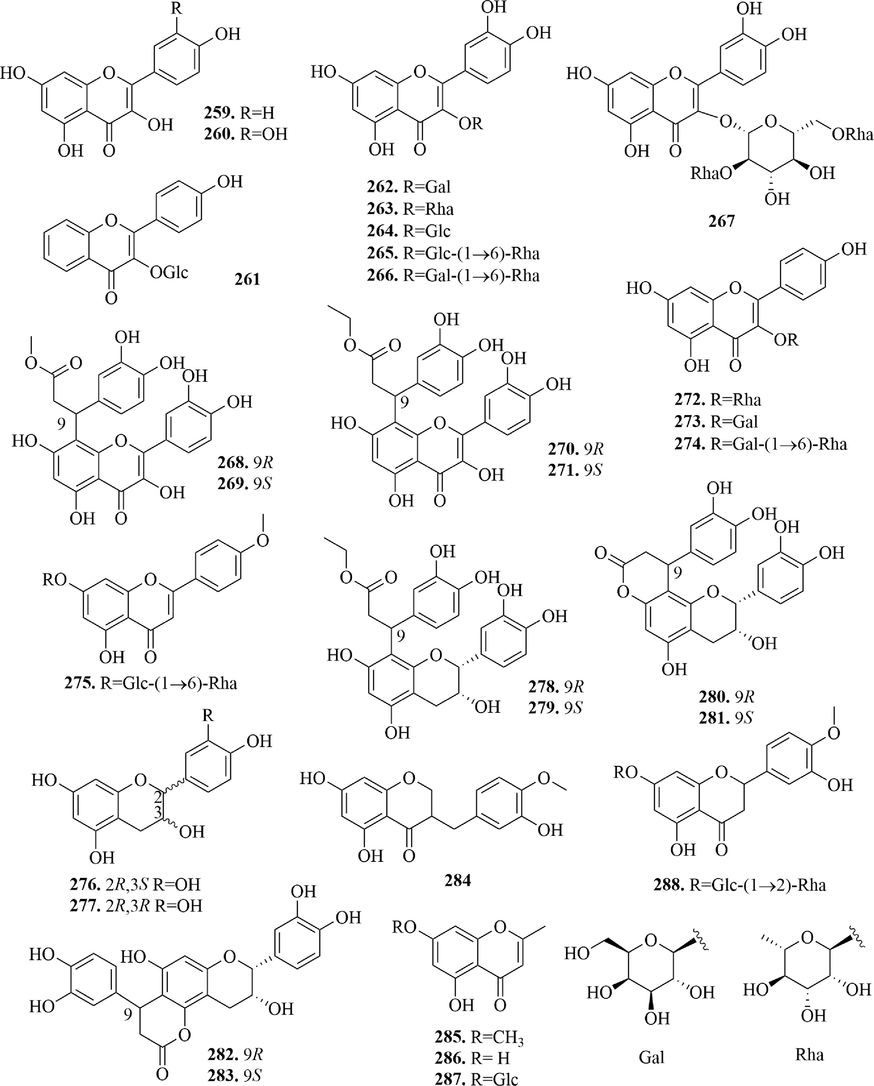
4.3 Flavonoids
According to structures, 30 flavonoids are divided into flavonols (259 ∼ 274), flavone (2 7 5), flavan-3-ols (276 ∼ 283), homoisoflavone (2 8 4), chromone (285 ∼ 287) and flavanones (2 8 8) (Fig. 13). Although the parent nucleus of flavonols and flavone both are 2-phenyl chromone, flavonols connect hydroxyl or other oxygen-containing groups at 3-position. In URCU, most flavonols mostly formed glycosides with various sugars at the 3-position. Notably, two pairs of phenylpropanoid-substituted flavonol enantiomers, (±)-uncariols C and D (268 ∼ 271), were isolated from the leaves of U. rhynchophylla, which had phenylpropanoid substitution at 8-position and two configurations at 9-position.
Flavan-3-ols are also known as catechins, whose parent nucleus is the 3,4-2H-2-phenyl-1-benzopyran ring. And the C-2 and 3 of flavan-3-ol are chiral carbons, which are generally (2R, 3S) and (2R, 3R) in plants. Six phenylpropanoid-substituted flavan-3-ols were isolated from the leaves of U. rhynchophylla and whose configurations were (2R, 3R). Uncariol A (2 7 8), uncariol B (2 7 9), cinchonain Ia (2 8 0) and cinchonain Ib (2 8 1) had phenylpropanoid substitutions at C-8. Whereas, cinchonain Ic (2 8 2) and cinchonain Id (2 8 3) had phenylpropanoid substitutions at C-6, which have two configurations at 9-position. It’s worth that the phenylpropanoid substituents of compounds 280 ∼ 283 formed a six-membered lactone ring with the benzene ring, respectively.
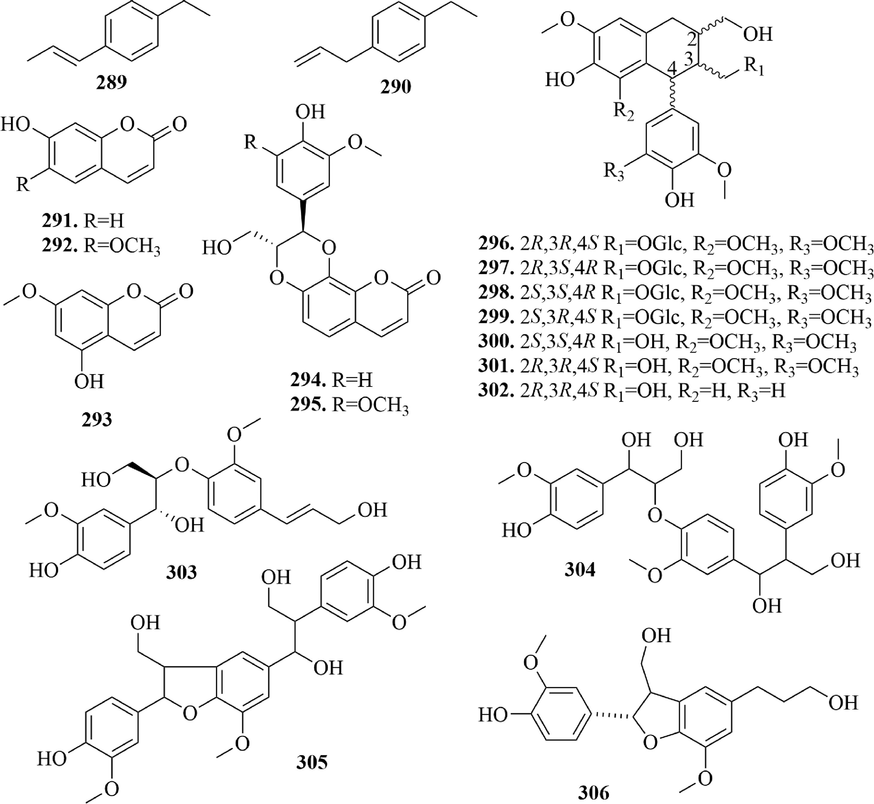
4.4 Phenylpropanoids
Phenylpropanoids, a class of compounds consisting of a benzene ring linked to three carbons (C6-C3), can be divided into simple phenylpropanoids (289 ∼ 290), coumarins (291 ∼ 295) and lignans (296 ∼ 306) (Fig. 14). The parent nucleus of coumarins is pyranone nucleus, which is formed by dehydration cyclization of cis-hydroxycinnamic acid. And most coumarins have hydroxyl substitution at the 7- position. It is noteworthy that phenylpropanoid substitutions of cleomiscosin B (2 9 4) and cleomiscosin D (2 9 5) both form dioxane with the hydroxyl groups of 7 and 8-position. Lignans are natural compounds synthesized from two C6-C3 units. And most of the lignans in URCU were arylnaphthalenes, and there were two configurations at 2, 3 and 4-position, respectively. Neolignans are compounds formed by linking the aliphatic hydrocarbon carbon of a phenylpropanoid to the benzene ring of another phenylpropanoid. At present, four neolignans, compounds 303 ∼ 306, were reported from URCU.
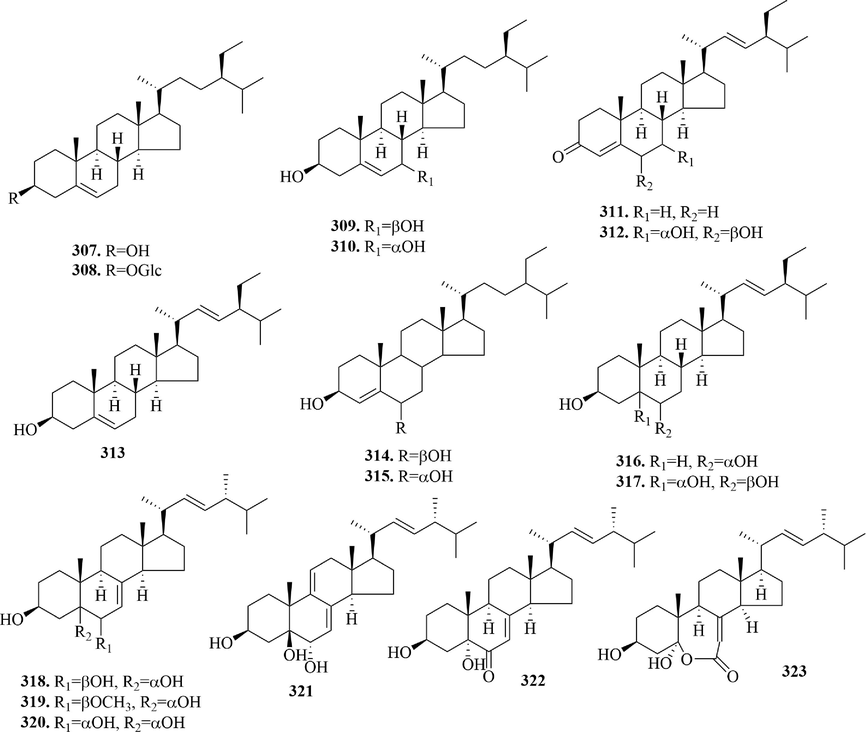
4.5 Phytosterols
Phytosterols are steroid derivatives with C-17 side chains of 8–10 carbon atoms, which is a natural active substance widely distributed in plants. Meanwhile, it’s one of the components of the cell membrane in plants. Phytosterols in URCU include sitosterols (307 ∼ 310), stigmasterols (311 ∼ 317) and ergosterols (318 ∼ 323) (Fig. 15). Ergosterol is an essential precursor for vitamin D synthesis. It’s worth that (22E,24R)-ergosta-7,22-dien-3β,5α-diol-6,5-olide (3 2 3) was a special ergosterol, whose B ring was a 7-membered lactone ring. In addition, most phytosterols existed in a free state, only β-daucosterol formed glycoside with glucose at the 3-position hydroxyl.
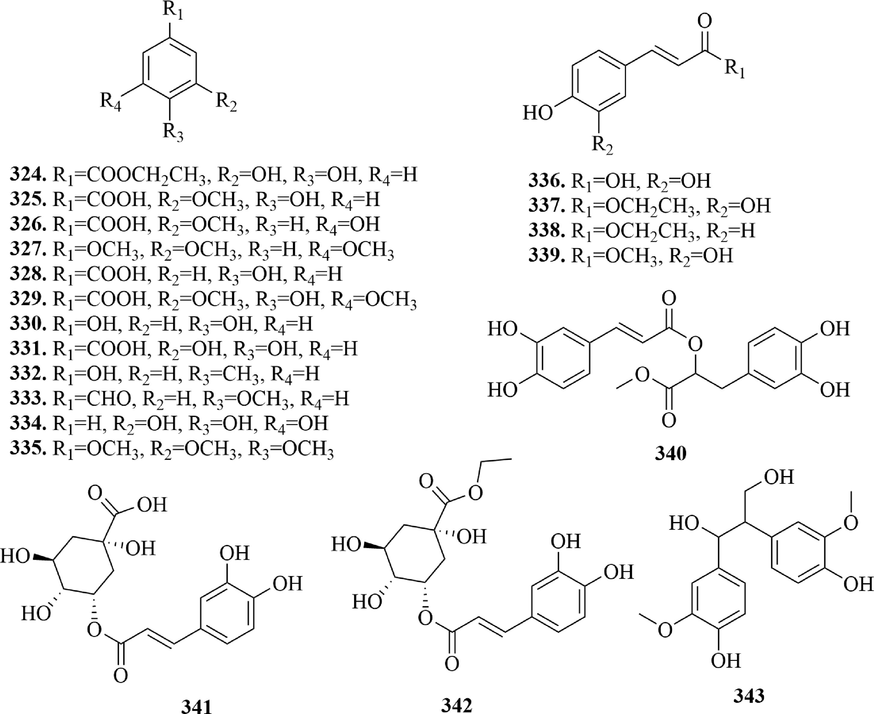
4.6 Phenolics
Phenolic compounds are important secondary metabolites in plants, which have good antioxidant activity for the existence of phenolic hydroxyl, phenolic compounds. While phenolic acid is a kind of organic acid containing a phenol ring. 12 phenolics (324 ∼ 335) and 8 phenolic acids (336 ∼ 343) have been isolated from URCU. The specific structures of compounds were shown in Fig. 16.
4.7 Other compounds
In addition, 28 other compounds (344 ∼ 371) were isolated from URCU, including saccharides, phthalates, anthraquinones, vitamins, ortho benzoquinones, folate-derived analogues and miscellaneous compounds. The specific structures of other compounds were shown in Fig. 17.
5 Pharmacological activity
5.1 Anti-hypertension
Hypertension is the most common chronic disease and the main risk factor causing cardiovascular and cerebrovascular diseases (Oparil et al., 2018). In vitro, dihydrocorynantheine (5) (IC50 = 6.73 μg/mL) exhibited a significant vasodilation effect against phenylephrine (Phe)-induced contraction in rat thoracic aorta rings (Wang et al., 2011a). 95 % ethanol extract of U. rhynchophylla could exert vasodilatory effects for Phe-induced contraction in SD rat’s aortic rings (EC50 = 0.028 mg/mL) via activating NO/sGC/cGMP signaling pathways, PGI2, G protein-coupled M3- and β2 receptors, and all the potassium channels except the Kca channel (Loh et al., 2017). Rhynchophylline (34) and isorhynchophylline (44) inhibited the contraction of arterial vessels of isolated rats induced by 60 mM KCl (20–30 μM) and induced by Phe and U46619 (100 and 200 μM, respectively) via l-type Ca2+ channels and other Ca2+ channels (Zhang et al., 2004). Uncarialin A (14) exhibited a relaxation effect against Phe-induced contraction (IC50 = 0.18 μM) in the manner by significantly inhibiting l-type calcium channel subunit alpha-1C (Cav1.2) via the hydrogen bond interaction with amino acid residue Met1186 (Yun et al., 2020). Likewise, rhynchine A (1 5 1) and B (1 5 2) showed strong inhibitory activities against the Cav3.1 calcium channel with IC50 values of 6.86 and 10.41 μM (Zhou et al., 2021). Geissoschizine methyl ether (7) (EC50 = 0.744 μM) was found to alleviate NE (norepinephrine)-induced aorta strip contraction through increasing NO and blocking voltage-dependent Ca2+ channels (Yuzurihara et al., 2002). In vivo, isorhynchophylline (44) (0.245 mg/kg) exhibited a strong anti-hypertensive role in SHRs by attenuating hypertension-induced the activation of the renin-angiotensin system and sympathetic hyperactivity (Li et al., 2020). The signal pathways related to the anti-hypertensive effect of URCU were shown in Fig. 18. By summarizing, it is found that compounds in URCU mainly dilate blood vessels to reduce blood pressure.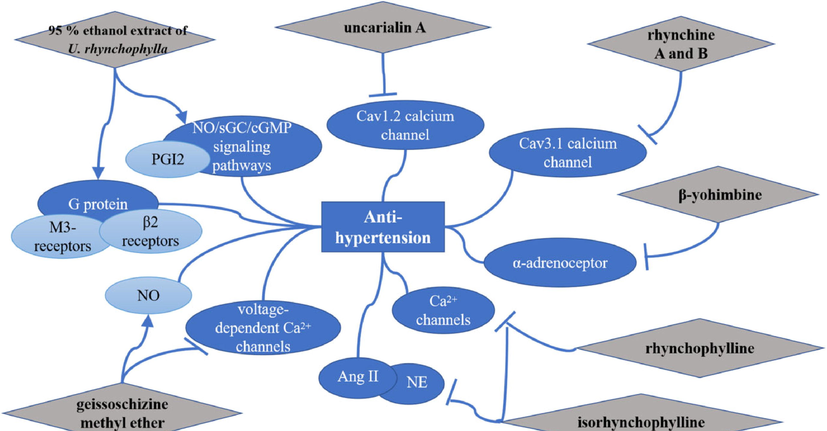
The signal pathways related to the anti-hypertension effect of URCU.
5.2 Anti-inflammation
The development of inflammation is often accompanied by the increase of inflammatory factors such as IL and TNF-α. In vitro, Kim et al. verified that water extract of U. rhynchophylla (1 mg/mL) had inhibitory effects on LPS-induced NO and IL-1β production in RAW264.7 Cells through blocking the phosphorylation of Akt and mitogen-activated protein kinase (MAPK) (Kim et al., 2010). In LPS induced N9 microglial cells, rhynchophylline (34) and isorhynchophylline (44) (0.3–30 μM) dose-dependently abated the production of inflammatory cytokines such as TNF-α, IL-1β and NO by inhibiting iNOS protein expression and blocking the activation of NF-κB and ERK and p38 MAPKs (Yuan et al., 2009). And compound 44 (30 or 40 μM) had a better anti-inflammatory effect in LPS induced murine alveolar macrophages cells by activating the TLR4/NF-κB/nod-like receptor protein 3 (NLRP3) inflammasome pathway (Zhou et al., 2019). In vivo, U. rhynchophylla alkaloids extracts (35, 70, and 140 mg/kg) effectively prevented inflammation by inhibiting serum and placental levels of pro-inflammatory cytokines, including IL-6, IL-1β, tumor necrosis factor-α (TNF-α), and interferon-γ (IFN-γ) (Wu and Xiao, 2019) in LPS-induced preeclampsia model rats. In summary, URCU can effectively reduce the release of inflammatory factors to achieve anti-inflammatory effects in LPS-induced in vitro and in vivo models.
NO is an important physiological transmitter and intracellular chemical messenger in the body, which plays a complex role in the inflammatory response. In vitro, corynoxeine (37), isocorynoxeine (40), rhynchophylline (34), isorhynchophylline (44) and vincoside lactam (74) exhibited inhibitory activities on LPS-induced NO release in primary cultured rat cortical microglia with IC50 value of 15.7, 13.7, 18.5, 19.0 and 16.4 μM, respectively (Yuan et al., 2008). Strictosidine (1 2 8) manifested a potent inhibitory activity on LPS-induced NO release in N9 microglia cells with IC50 value of 8.3 μM (Ma et al., 2009b). Also, uncarinic acid I (2 0 5), 3β-hydroxy-27-p-(E)- coumaroyloxyursan-12-en-28-oic acid (1 8 5) and 3β-hydroxy-27-(E)-coumaroyl -oleanen-12-en-28-oic acid (2 1 7) exhibited inhibitory effects on LPS-induced NO production in RAW264.7 cells with IC50 value of 1.48, 7.01, and 1.89 μM, respectively (Zhang et al., 2014). The signal pathways related to the anti-inflammation effect of URCU were shown in Fig. 19.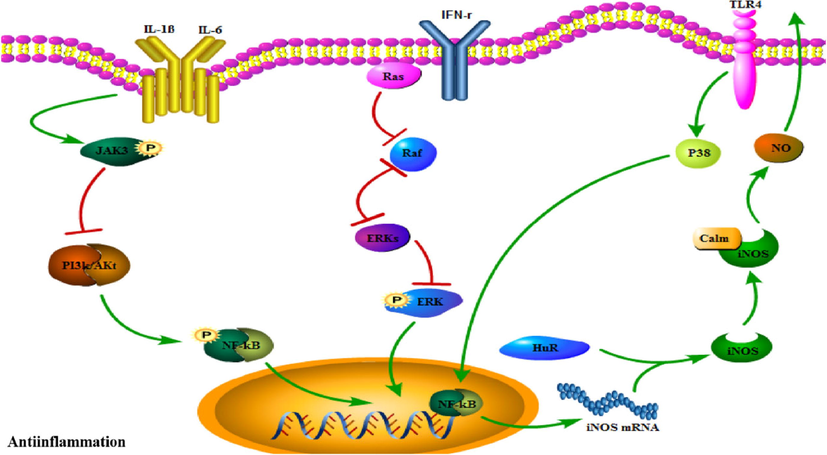
The signal pathways related to the anti-inflammation effect of URCU.
5.3 Anticancer
As a global public health problem, cancer seriously endangers human life and health. Studies have found that natural drugs can achieve anti-tumor effects by inhibiting and killing tumor cells, inducing apoptosis, affecting related proteins and enzymes, regulating body immunity, and enhancing antioxidant effects (Liu et al., 2015). Killing tumor cells through the cytotoxic activity of compounds or extracts is a more direct anticancer method. In vitro, Kim et al. found the n-BuOH fraction of U. rhynchophylla (0.05, 0.1, and 0.2 mg/mL) has strong cytotoxicity towards HepG2 cells via up-regulating expression levels of caspases 7 and 8 and poly ADP ribose polymerase (PARP) (Kim et al., 2014). Rhynchophylline (34) (130 µM) was found to induce HepG2 cell apoptosis by eliminating the phosphorylations of p38, ERK, JNK, CREB, Akt and STAT3 signals and strengthening the phosphorylation of p53 signals. Moreover, C-X-C chemokine receptor type 4 (CXCR4), matrix metallopeptidase-9 (MMP-9), and MMP-2 expression were inhibited upon rhynchophylline treatment (Lee et al., 2017). Meanwhile, uncarinic acid E (2 1 6) (6, 12, 24, 48 µM) also caused apoptosis in HepG2 cells via accumulating p53, altering the Bax/Bcl-2 ratio and activating caspases (Zhao et al., 2006). Ursolic acid (1 7 0) and rhynchophylline (34) (50, 25, 12.5, 6.25 μM) could inhibit the proliferation of HepG2 cells and induce apoptosis. Compound 170 more significantly acted as a disincentive to the growth of HepG2 cells than rhynchophylline (Wu et al., 2017). Sun et al. reported that 3β,6β,19α-trihydroxy-olean-12-en-28-oic acid (2 2 0) exhibited cytotoxicity in MCF-7 and HepG2 cells with IC50 = 78.2 and 73.9 µg/mL, respectively (Sun et al., 2012b).
Suppressing tumor cell proliferation by blocking cell cycle is also an effective way. In vitro, Uncarinic acid A (2 1 4), uncarinic acid B (2 1 5), uncarinic acid C (1 7 6), uncarinic acid D (1 7 7), uncarinic acid E (2 1 7), 3β-hydroxy-27-(E)-coumaroyl-oleanen −12-en-28-oic acid (2 1 0), 3β-hydroxy-27-p-(E)-coumaroyloxyursan-12-en-28-oic acid (1 8 5) and 3β-hydroxy-27-p-(Z)-coumaroyloxyursan-12-en-28-oic acid (1 8 4) restrained the growth of HCT-15, MCF-7, A549, and HT-1197 cells with IC50 values of 0.5–6.5 μM (Lee et al., 2000). 3-diethylamino-5-methoxy-1,2-benzoquinone (3 6 8) and 3-ethylamino-5-methoxy-1,2-benzoquinone (3 6 9) showed weak antiproliferative activities on A549, HepG2 and A2780 cells (IC50 = 50.2–98.8 μM). Isorhynchophyllic acid (50) significantly inhibited the proliferation of A549, HepG2 and A2780 cells with IC50 value of 5.8, 12.8 and 11.8 μM, respectively (Zhang et al., 2016). In vivo, hirsutine (1) (40–80 µM) also limited tumor growth in the A549 xenograft mouse model through GSK-3β dephosphorylation and accelerated apoptosis via ROCK1/phosphatase and tensin homolog (PTEN)/PI3K/Akt signaling (Zhang et al., 2018). In vitro, hirsutine (1) (10, 25 and 50 μM) had an inhibitory effect on Jurkat Clone E6-1 cells which could inhibit cell growth in the S and G2/M phases. Meanwhile, it also could promote cell death upon elevating Bax, cleaved-caspase 3/9, Cyto-c protein, caspase-3 and 9, and decreasing Bcl-2 protein (Meng et al., 2021). Corynantheidine (4) exhibited moderate cytotoxicity against HL-60 and SW480 cells with IC50 values of 13.96 and 23.28 µM, respectively (Wang et al., 2011a). In conclusion, both extracts and compounds from URCU can achieve anti-tumor effects in a variety of ways. Although anticancer is not the traditional use of URCU, the development of new uses of URCU through modern research is also an effective use of URCU resources.
Breast cancer, the most common cancer in the world, is a major global health challenge, which seriously affects the quality of life of patients. It is worth noting that URCU has a certain therapeutic effect on breast cancer. In vitro, Chen et al. found that treatment of MDA-MB-231 cells with U. rhynchophylla proanthocyanidins (UPAs) (5, 10, 20, 30 and 40 µg/mL) increased G2/M cell cycle arrest. Further research showed that UPAs inhibited cell viability and migration ability by increasing cellular ROS production, loss of mitochondrial membrane potential, Bax/Bcl-2 ratio and cleaved caspase 3. Meanwhile, it was interesting that the cytotoxic effects of 5-FU against MDA-MB-231 cells could be enhanced by UPAs (Chen et al., 2017). In addition, hirsutanine D-F (149, 150 and 162) (100 µM) exhibited a slight inhibition effect on the proliferation of the breast cancer cell MDA-MB-23 cells by 18.1 %, 20.5 % and 15.9 %, respectively (Pan et al., 2017). Moreover, hirsutine (1) remarkably reduced the viability of human breast cancer MCF-7 and MDA-MB-231 cells with IC50 values of 447.79 and 179.06 µM. Compound 1 induced apoptosis of MDA-MB-231 cells by decreasing the Bax/Bcl-2 ratio and activating caspase 9 and 3 (Huang et al., 2018). Furthermore, compound 1 (IC50 = 62.82 µM) also showed an inhibition effect for MCF-7 cells via down-regulating HIF-1α, Snail and MMP-9, and up-regulating E-cadherin (Zhai et al., 2017).
Multidrug resistance is one of the main reasons for the failure of tumor treatment, which greatly limits the selection and use of cancer drugs. In vitro, 5 μg/mL total alkaloids of Uncaria reversed multidrug resistance (MDR) for vincristine on KBv200 cell line by 16.8-fold (Zhang et al., 2001). Isorhynchophylline (44) (0.5, 1.0 and 1.5 mg/L) reversed the MDR of A549/DDP cells by restraining the efflux of chemotherapeutic drugs and enhancing the induction of apoptosis by chemotherapeutic drugs (Zhou et al., 2009). The signal pathways related to the anti-cancer effect of URCU were shown in Fig. 20.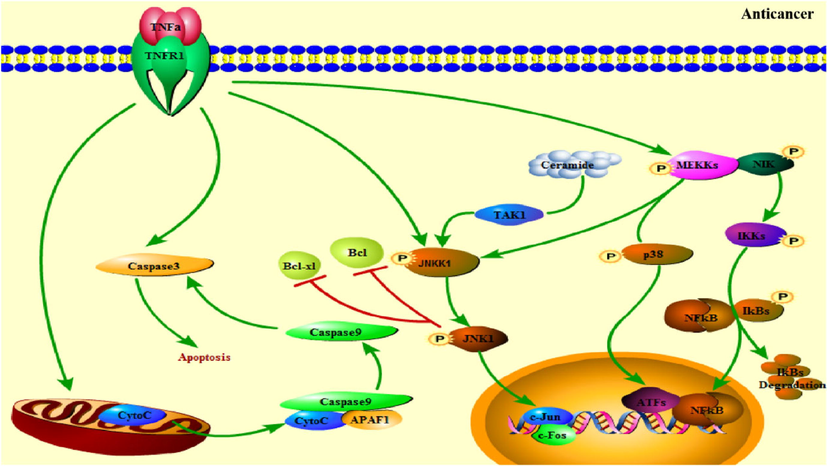
The signal pathways related to the anti-cancer effect of URCU.
5.4 Antioxidant
Oxidation inhibitor can effectively inhibit the oxidation reaction of free radicals at low concentrations, which is the main research and development direction of health products and cosmetics enterprises. In vitro, Yin et al. found that different extracts of U. rhynchophylla had strong antioxidant capacity, and the order of antioxidant capacity was ethanol extract > ethyl acetate extract > chloroform extract > petroleum ether extract (IC50 = 20.432, 1.547, 0.0283 and 0.00326 g/L) (Yin et al., 2010). Uncariol A (2 7 7), uncariol B (2 7 8), (±)-uncarilin A (141, 142), (±)-uncarilin B (143, 144), cinchonain Ia-Id (279–282), quercetin (2 5 9), (-)-epicatechin (2 7 6), methyl caffeate (3 3 6), quercetin-3-O-robinobioside (2 6 5) and rutin (2 6 4) showed comparable DPPH radical scavenging potentials with IC50 values were 22.26, 16.12, 10.28, 11.32, 12.67, 14.34, 15.72, 8.27, 3.22, 5.84, 7.52, 8.21, 5.35, 8.14, and 2.13 μM, respectively (Li et al., 2017a).
5.5 Antiviral
Dengue virus (DENV) is transmitted to humans by Aedes mosquitoes and is a public health issue worldwide. No antiviral drugs specific for treating dengue infection are currently available (Reis et al., 2008). In vitro, mitraphylline (1 0 1), isomitraphylline (1 0 7) and uncarine C-F (99, 104 and 97) (1 μg/mL) were found to have significant inhibitory effects by lowering Dengue virus (DENV)-antigen cell rates. Moreover, these compounds exerted strong immunomodulation via declining TNF-α, IFN-α and IL-10 levels (Reis et al., 2008). In addition, hirsutine (1) (10 μM) showed antiviral activities against all DENV serotypes by inhibiting the viral particle assembly, budding, and release step (Hishiki et al., 2017). Thus, compounds 1, 97, 99, 101, 104 and 107 may be the potential candidate to treat DENV.
5.6 Antiasthma
Asthma is a chronic inflammatory disease characterized by airway remodeling and inflammation. And proliferation of airway smooth muscle cells (ASMCs) is key to the progression of asthma (Li et al., 2021a). In vitro, rhynchophylline (34) (10 µM) inhibited the proliferation of ASMCs by inhibiting TGF-β1-mediated Smad and MAPK signaling pathways (Wang et al., 2019). In addition, it (40 or 80 mg/kg) also suppressed ASMC autophagy by suppressing the JAK2/STAT3 signal to achieve anti-asthma effect (Li et al., 2021a). Zhu reported that isorhynchophylline (44) could induce the apoptosis of ASMCs by up-regulating miR-200a and deactivating the FOXC1/NF-kB pathway to achieve anti-asthma effect (Zhu et al., 2020).
5.7 Sedative and hypnotic
Insomnia is a sleep disorder, which seriously affects the quality of human life. Sedation and hypnosis are traditional applications of URCU (Chen et al., 2019). In vivo, Chen et al. found that the stem hook, branch and leaf extracts of U. rhynchophylla, U. macrophylla and U. hirsuta at 15 g/kg could significantly inhibit the number of spontaneous activities in mice and prolong the sleep time of mice induced by pentobarbital (Chen et al., 2019). It was found that corynoxine (37) and corynoxine B (36) (30 mg/kg), isorhynchophylline (44) and geissoschizine methyl ether (7) (100 mg/kg) significantly reduced autonomic activity in mice. Meanwhile, compound 36 (30 mg/kg), 44 and 7 (60 mg/kg) also could inhibit the activity of mice (Sakakibara et al., 1999). Notably, rhynchophylline (34) (5, 10, 15 mg/kg) can exhibit a sedative effect by raising 5-hydroxyindole acetic acid (5-HIAA) in rat brain striata and hippocampus, and decreasing concentrations of norepinephrine (NE) on hippocampus and frontoparietal lobe of cortex (Lu et al., 2003). Meanwhile, oral administration of compound 37, 36, 34 and 44 (100 mg/kg) prolonged the hypnosis duration induced by thiopental in ICR mice (Sakakibara et al., 1998).
5.8 Anti-epilepsy
Epilepsy is a chronic disease in which sudden abnormal discharges of brain neurons cause transient brain dysfunction (Xu et al., 2001). In vivo, Wang et al. found that rhynchophylline (34) (10, 20, 40 mg/kg/d) showed a good antiepilepsy effect by inhibiting the expression of Toll-like receptor 4 and enhancing the activity of SOD (Wang and Cai, 2018). The ethanol extract of U. rhynchophylla (1 g/mL) could reduce the peak potential of pyramidal cells in the CA1 region of rat hippocampal slices induced by pilocarpine, which may be related to the inhibitory effect of rhynchophylline (31) on calcium influx and glutamate release (Xu et al., 2001). Oral U. rhynchophylla extract (1 g/kg, 5d/wk) can inhibit the excessive expression of S100 B protein and receptor of advanced glycation end products (RAGE) through RAGE pathway to reduce epilepsy in kainic acid (KA) induced epileptic SD rats (Tang et al., 2017). Research proved that 70 % alcohol extract of U. rhynchophylla (1 g/kg/day) and rhynchophylline (0.25 mg/kg) exhibited anti-convulsive effects in KA-induced rats by inhibiting IL-1β and BDNF gene expressions via suppressing TLR and neurotrophin signaling pathways (Ho et al., 2014). Meanwhile, rhynchophylline (34) (100 μM) effectively reduced the severity of seizures and neuronal hyperexcitation by inhibiting the current of persistent sodium (INaP) and N-methyl-d-aspartate receptor (NMDAR) (Shao et al., 2016). Geissoschizine methyl ether (7) (1–30 μM) showed antiepileptic activities by inhibiting voltage-gated sodium (Nav), calcium (Cav), and delaying the currents of rectifier potassium (IK) and the ligand-gated nicotinic acetylcholine (nACh) (IC50 = 1.3–13.3 μM). Meanwhile, in the electroshock-induced mouse seizure model, geissoschizine methyl ether (50–100 mg/kg) suppressed generalized tonic-clonic seizures. In the 6-Hz-induced mouse seizure model, oral administration of compound 7 (100 mg/kg) reduced treatment-resistant seizures (Xie et al., 2020). The signal pathways related to the anti-epilepsy effect of URCU were shown in Fig. 21.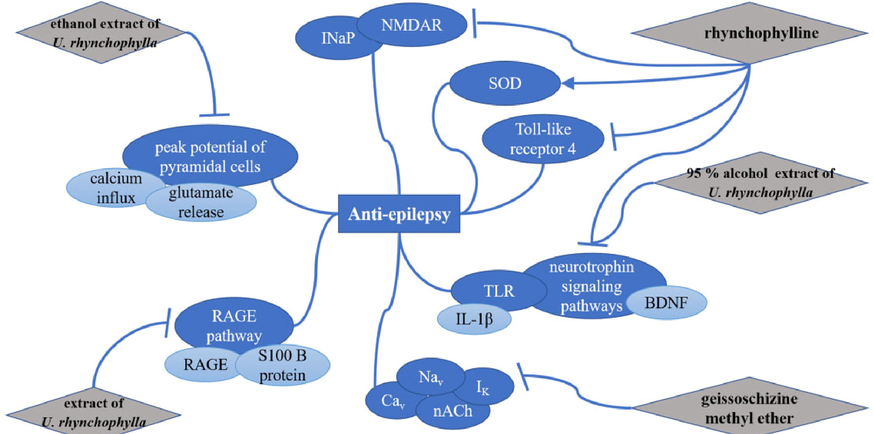
The signal pathways related to the anti-epilepsy effect of URCU.
5.9 Anti-depression
Depression is a mental disease with abnormal low spirit as the main clinical manifestation, which is reflected in the lack of monoamine neurotransmitters in the brain, especially norepinephrine (NE), 5-hydroxytryptamine (HT) and dopamine (DA) (Zhang et al., 2017). In vitro, uncarialin E (19), G (21), J (54) and K (23), 3α-dihydrocadambine (1 3 8), isorhynchophylline (34), hirsuteine (2), akuammigine (77), Z-geissoschizine (13) and corynoxeine (37) displayed significant magonistic effects towards 5-HT1A receptor, whose EC50 values were 2.2, 0.1, 7.86, 7.32, 1.6, 2.0, 2.24, 1.18, 1.52, and 3.75 μM, respectively (Liang et al., 2019). In vivo, U. rhynchophylla EtOH extract displayed an agonistic effect against the 5-HT1A receptor with an EC50 value of 17.42 μg/mL, which could ameliorate CUMS-induced depression-like behaviors in mice (Qiao et al., 2021). In reserpine-induced depression model mouses, isorhynchophylline (44) (10, 20 and 40 mg/kg) showed antidepressant activity by significantly up-regulating NE and 5-HT levels and inhibiting MAO-A activity in hippocampus and frontal lobe of mice (Xian et al., 2017). Isorhynchophylline (44) (20 or 40 mg/kg/d) reversed CUMS-induced depression via enhancing neurotrophins and up-regulating the phosphatidylinositol 3-kinase/protein kinase B/glycogen synthase kinase-3β (PI3K/ Akt/ GSK-3β) pathway (Xian et al., 2019). And, rhynchophylline (34) (25 mg/kg) exhibited rapid antidepressant-like effects by inhibiting EphA4 ephexin1 signaling and activating BDNF-tropomyosin receptor kinase (TrkB) signaling in a mice model of social defeat (Zhang et al., 2017).
Melatonin (MT), secreted by the pineal gland, can coordinate circadian rhythm and neuroendocrine processes by activating MT1 and MT2 receptors. MT receptors can be used as a target for the treatment of depression, which could treat major depressive disorder by normalizing disturbances of circadian rhythms (Ekmekcioglu, 2006). In vitro, (±)-Uncarilins A and B (141–144) showed activities on the MT1 and MT2 receptors, at the tested concentration of 0.25 mM. And (-)-Uncarilins B (1 4 4) possessed the most potent activities on MT1 and MT2 receptors, with agonistic rates of 11.26 % and 52.44 % (Geng et al., 2017). In addition to alkaloids, catechin (2 7 5) manifested agonistic effects on melatonin receptors (MT1 and MT2), which inhibited MT1 and MT2 activities with EC50 values of 25.8 μM and 47.3 μM respectively (Ekmekcioglu, 2006).
5.10 Ischemic brain injury
Ischemic brain injury is also known as stroke, which is a group of brain damage caused by sudden rupture of blood vessels in the brain or blood can not flow into the brain due to vascular obstruction (Ramos et al., 2017). In vivo, U. macrophylla alkaloids can significantly reduce the volume of cerebral infarction in rat cerebral ischemia model and reduce the damage of neurological function caused by ischemia (Xie, 2009). Methanol extracts of U. rhynchophylla (100–1000 mg/kg) were found to significantly protect hippocampal CA1 neurons against transient forebrain ischemia via inhibiting induction of cyclooxygenase-2 (COX-2) expression in hippocampus (Suk et al., 2002). In the permanent middle cerebral artery occlusion model, rhynchophylline (34) (30 mg/kg) not only improved neurological deficits and brain edema through activating the PI3K/Akt/mammalian target of rapamycin (mTOR) signaling pathway and inhibiting the TLRs/NF-κB pathway, and reduced infarct volume by increasing claudin-5 and BDNF (Huang et al., 2014). Notably, it (0.02 or 0.2 mg/mL) might protect cerebral ischemia by inhibiting necrosis and apoptosis of primary astrocytes in rats induced by ischemia-repefusion (IR) (Gao et al., 2009). Subsequent studies have shown that rhynchophylline (34) (3 μg/mL) protected astrocytes by inducing nuclear factor E2-related factor 2 (Nrf2) nuclear translocation via PI3K signaling pathway to alleviate oxidative damage induced by IR (Ying et al., 2012). In a rat model with MCAO and reperfusion-induced (I/R) injury, isorhynchophylline (44) (20 mg/kg) attenuated the infarct volume and improved the neurological function in I/R injury rats through reducing the neuronal death rate, brain water content, and aquaporin-4 expression in the ischemic penumbra of I/R injury rats’ brains. Besides, it (20 mg/kg) also treated microglial activation and inflammatory response via inhibiting lκB-α degradation, NF-κBp65 activation and CX3CR1 expression (Deng et al., 2021).
5.11 Neuroprotection
URCU has the effect of extinguishing wind and settling convulsion. So, it is used to neurological diseases since ancient times (Li et al., 2021d). Modern pharmacological studies have shown that URCU has excellent neuroprotective activity. In vitro, the crude alkaloids of U. rhynchophylla (1, 10 and 100 μg/mL) showed a protective effect against NMDA-induced cytotoxicity in the hippocampal slices by suppressing the NMDA-induced expressions of apoptosis-related genes such as c-jun, p53, and Bax (Lee et al., 2003). Rhynchophylline (34) (5 or 50 μM) presented a protective effect on DA-induced apoptosis of NT2 cells through suppressing DNA degradation (Shi and Kenneth, 2002). The results showed that isorhynchophylline (44) (10 or 50 μM) significantly elevated cell viability, decreased the levels of intracellular ROS and MAD, increased the level of glutathione, and stabilized mitochondrial membrane potential in Aβ25-35-treated PC12 cells via significantly suppressing the formation of DNA fragmentation and the activity of caspase-3 and moderating the ratio of Bcl-2/Bax (Xian et al., 2012). Notably, 5β-carboxystrictosidine (1 3 0) and chlorogenic acid (3 4 0) (12.5, 25, 50 or 100 μM) could protected mouse nerve growth factor (mNGF)-differentiated PC12 cells against toxicity induced by 6-hydroxydopamine (OHDA). They could scavenge ROS with IC50 values of 24.5 and 19.7 μM and reduce intracellular calcium levels with respective IC50 values of 46.9 and 27 μM, respectively. Meanwhile, they also inhibited caspase 3 and 9 activities with respective IC50 values of 25.6 and 24.5 μM for 1 and 19.4 and 16.3 μM for 2 (Lin et al., 2020). Meanwhile, it (10–50 µM) inhibits MPP+-triggered neurotoxicity of primary cerebellar granule neurons via activating PI3-K/Akt/GSK3β/MEF2D signaling pathway (Hu et al., 2018). Zheng et al. found that rhynchophylline (34) exhibited a protective effect against the MPTP-induced decrease in MPP+-induced neurotoxicity in PC12 cells (20 µM) by activating the PI3K/Akt signaling pathway. In vivo, it also exhibited a protective effect against the MPTP-induced decrease in tyrosine hydroxylase (TH)-positive fibers in C57BL/6 mice (30 mg/kg) by activating the PI3K/Akt signaling pathway (Zheng et al., 2021). The signal pathways related to the neuroprotection effect of URCU were shown in Fig. 22.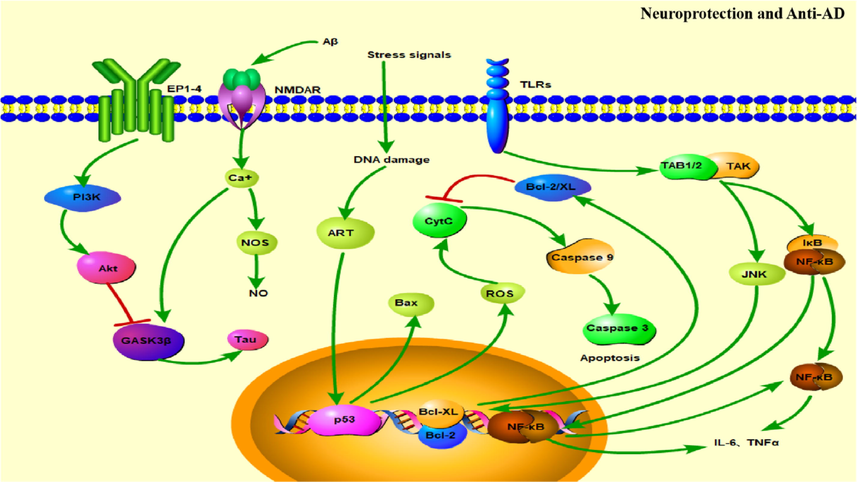
The signal pathways related to the neuroprotection and anti-AD effect of URCU.
5.12 Anti-Alzheimer's disease
Alzheimer’s disease (AD) is the most common neurodegenerative disorder, characterized by progressive neuronal loss with amyloid β-peptide (Aβ) plaques. In vivo, 70 % ethanol extract of U. rhynchophylla (400 mg/kg/d) could attenuate Aβ deposition and Aβ-mediated neuropathology in 5 × FAD mice by alleviating gliosis and neurodegeneration, and impairing adult hippocampal neuron damage (Shin et al., 2018). Rhynchophylline (34) (50 mg/kg/d) effectively reduced the EphA4 activity in the hippocampus of amyloid precursor protein (APP)/presenilin 1 (PS1) transgenic mice by blocking the EphA4- dependent signaling (Fu et al., 2014).
Oligomers of the amyloid-β 42 protein (Aβ42) could cause synaptic dysfunction in the pathology of Alzheimer’s disease (AD). In vivo, Fujiwara et al. found that three extracts of U. rhynchophylla (100 mg/mL) all could inhibit the aggregation of Aβ1-40 and Aβ1-42 (Fujiwara et al., 2006). Whereas rhynchophylline (34) (IC50 = 9.0 μM) could remold the spontaneous discharges disturbed by Aβ and counteract the deleterious effect of Aβ1-42 (Shao et al., 2015). In Aβ1-42-induced SD rats, compound 34 (100 µM) efficiently rescued the Aβ1-42-induced spatial learning and memory deficits by reducing extrasynaptic NMDARs-mediated excitatory postsynaptic currents and downregulating GluN2B-NMDAR expression in the DG region (Yang et al., 2018). In vitro, Uncarinic acid C (1 7 6) (50 μM) was identified as a specific inhibitor of the nucleation phase of Aβ42 aggregation. And structure–activity studies suggested that both a C-27 ferulate and a C-28 carboxylic acid group are required for its inhibitory activity (Yoshioka et al., 2016).
During the AD process, abnormally hyperphosphorylated tau protein may impede mitochondrial movement and affect mitochondrial distribution along the axons of cortical neurons, which would induce apoptosis and regional-specific neurodegeneration. Based on the pathological process of AD, corynoxine, isocorynoxeine, dihydrocorynatheine, isorhynchophylline and hirsutine were identified as key alkaloids that regulate tau phosphorylation (Zeng et al., 2021). In vitro, treatment with isorhynchophylline (44) (1, 10 or 50 μM) inhibited tau hyperphosphorylation by enhancing the expression of phosphorylated cAMP response element binding protein (p-CREB) through PI3K/Akt/GSK-3β signaling pathway (Xian et al., 2013). In vivo, it (20 or 40 mg/kg) could significantly ameliorate the cognitive deficits induced by Aβ25-35 in the rats, which also could attenuate Aβ25-35-induced neuronal apoptosis in the hippocampus by down-regulating the ratio of Bcl-2/Bax, cleaved caspase-3 and caspase-9, as well as suppressing the tau protein hyperphosphorylation. The mechanistic study showed the neuroprotection of IRN is via inhibiting the GSK-3β activity and activating PI3K/Akt signaling pathway. (Xian et al., 2014a).
The massive accumulation of Aβ could directly damage the cell membrane and result in oxidative stress and ROS release. Ultimately it would lead to mitochondrial apoptosis and neuroinflammation. This suggested that we could treat AD by strengthening the anti-oxidation and anti-inflammatory functions of brain tissue. In vivo, Li et al. revealed that isorhynchophylline (44) (40 mg/kg) improved cognitive impairment in TgCRND8 transgenic mice. It could reduce Aβ generation and deposition (Aβ40, Aβ42) through modulating the amyloid precursor protein (APP) processing and phosphorylation. This process mainly including up-regulation of β-site APP cleaving enzyme-1 (BACE-1), phosphorylated APP (Thr668), presenilin-1 (PS-1) and anterior pharynx-defective-1 (APH-1), as well as insulin-degrading enzyme (IDE). Meanwhile, isorhynchophylline (44) also could inhabit tau hyperphosphorylation and neuroinflammation (TNF-α, IL-6 and IL-1β) and attenuate the ratios of p-c-Jun/c-Jun and p-JNK/JNK through inhibiting the activation of JNK signaling pathway. Microglia (Iba-1) and astrocytes (GFAP) were suppressed by isorhynchophylline, as well. (Li et al., 2019). Xian et al. demonstrated that it (20 or 40 mg/kg) was able to ameliorate cognitive deficits induced by d-gal in mice through increasing GSH, SOD, CAT and NF-κB and decreasing MDA, PGE2, NO, COX-2 and iNOS (Xian et al., 2014b).
Based on the cholinergic hypothesis of AD pathogenesis, inhibition of acetylcholinesterase (AChE) activity could significantly increase the content of central acetylcholine, enabling the accumulation of acetylcholine at synapses, thereby improving the cognitive function of patients. In vitro, isorhynchophylline (44) (20 or 40 mg/kg) could inhibit AChE activity, and reduce oxidative damage to brain tissue via NF-κB signaling pathway (mainly NF-κBp65 and IκBα) to improve AlCl3-induced learning and memory impairment in mice (Li et al., 2018). Notably, geissoschizine methyl ether (7), geissoschizine methyl ether N-oxide (58) and rhynchophylloside J (93) exhibited inhibitory activity against AChE with IC50 values of 3.7 μg/mL, 23.4 μM, and 10.5 μM, respectively (Yang et al., 2012; Jiang et al., 2015; Guo et al., 2019).
By summarizing the literature on URCU treatment AD, It can be proved that URCU could treat AD by reducing the accumulation of Aβ (especially oligomers of the Aβ42), reducing abnormally hyperphosphorylated tau protein and inhibiting AChE. This could provide direction for subsequent research. The signal pathways related to the anti-AD effect of URCU were shown in Fig. 22.
5.13 Anti-Parkinson's disease
Parkinson’s disease (PD) is a progressive, age-related, neurodegenerative disorder characterized by tremors, rigidity, and cognitive impairment. In vitro, 95 % EtOH extract of U. rhynchophylla (20 μg/mL) inhibited the expression of HSP90, which also could suppress MPP+-induced SH-SY5Y cell apoptosis and autophagy through increasing the expressions of Bcl-2, Cyclin D1, p-ERK, p-PI3K p85, PI3K p110α, p-AKT, and LC3-I and decreasing cleaved caspase 3, Bax, p-JNK, p-p38, and LC3-II. Meanwhile, it also markedly decreased the apoptotic ratio and elevated mitochondrial transmembrane potential (DΨm) (Lan et al., 2018). In 6-OHDA induced PC12 cells, water extract of U. rhynchophylla (0.01–5 μg/mL) significantly reduced cell death and the generation of ROS, increased GSH levels, and inhibited caspase-3 activity. In addition, studies showed that isorhynchophylline (44) (0.3–100 μM) significantly reduced MPP+-induced cell death and oxidative stress in PC12 cells by blocking the generation of ROS in upstream of the apoptosis signal-regulating kinase 1 (ASK1)/JNK pathway and the inositol-requiring enzyme 1 (IRE1)/caspase-12 pathway (Li et al., 2017b). Meanwhile, in vivo, posttreatment with water extract of U. rhynchophylla (5 mg/kg/d) significantly reduced dopaminergic neuronal loss in substantia nigra pars compacta in 6-OHDA-lesioned rats (Shim et al., 2009). In conclusion, the chemical constituents or extracts of URCU have been proven to have anti-PD effect in vitro and in vivo.
Accumulation of α-synuclein (α-syn) in the brain is a pathogenic feature and also a causative factor of parkinson’s disease. In vitro, corynoxine (45) (25 μM) promoted the clearance of wild-type and A53T α-syn uclein, and suppressed p-Akt, p-mTOR, and p-p70 S6 kinase levels via up-regulating the Akt/mTOR pathway (Chen et al., 2014a). Furthermore, corynoxine B (36), an enantiomer of corynoxine (45), might restore the deficient cytosolic translocation of HMGB1 and autophagy in cells overexpressing SNCA by blocking SNCA-high mobility group box 1 (HMGB1) interaction (Song et al., 2014). Finaly, isorhynchophylline (44) (6.25, 12.5, 25 μM) promoted clearance of wild-type, A53T and A30P α-syn monomers, α-syn oligomers and α-syn/synphilin-1 aggresomes in neuronal cells via the autophagy-lysosome pathway. Notably, the autophagy was dependent on the function of Beclin 1 (Lu et al., 2012). Thus, compounds 45, 36 and 44 may be the potential candidate to treat PD by reducing the accumulation of α-syn in the brain. The signal pathways related to the anti-PD effect of URCU were shown in Fig. 23.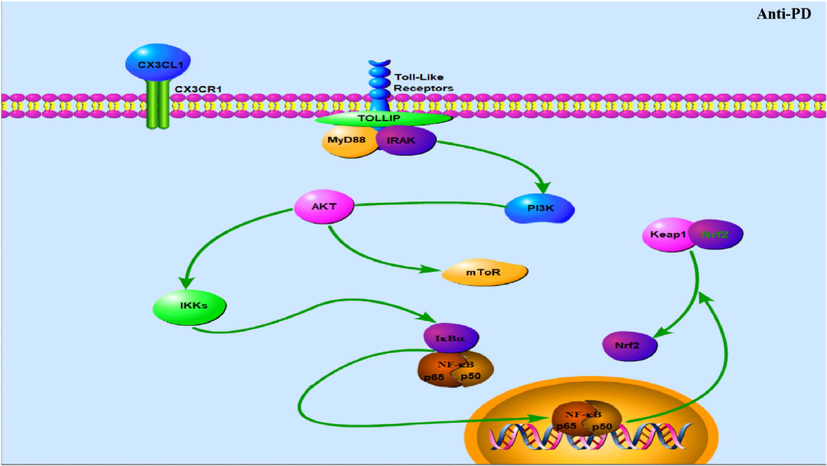
The signal pathways related to the anti-PD effect of URCU.
5.14 Bone protection
Regulation of osteoclast differentiation and activity is a major target for preventing and treating pathological bone diseases. In vitro, water extract of the hooks and stems of U. sinensis could inhibit RANKL-induced differentiation of murine bone marrow macrophages and RAW264.7 cells into osteoclasts by inhibiting the activation of NF-kB and the expression of nuclear factor of activated T-cells, cytoplasmic 1, and suppress RANKL-induced bone loss with a significant amelioration of trabecular bone micro-structures. Furthermore, it also reduced serum TRAP5b activity and C-terminal cross-linked telopeptide of type I collagen levels (Ha et al., 2017). These results suggest that U. sinensis could be a promising herbal candidate for preventing and treating bone diseases such as osteoporosis.
All the pharmacological effects of this genus are summarized in Table 3. “–” means no reports were found.
Pharmacological effects
Effective fraction/ Compounds
Vitro or vivo
Models
Dosage
Pathway or possible target site
Ref.
Anti-hypertension
isorhynchophylline
In vivo
SHRs
0.245 mg/kg
renin-angiotensin and sympathetic system↓
(Ang II, NE↓)(Li et al., 2020)
dihydrocorynantheine
In vitro
Phe-induced contraction in rat thoracic aorta rings
IC50 = 6.73 µg/mL
–
(Wang et al.,2011a)
95 % ethanol extract of
U. rhynchophylla
In vitro
Phe-induced contraction in SD rats aortic rings
EC50 = 0.028 mg/mL
NO/sGC/cGMP signaling pathways, PGI2,
G protein-coupled M3- and β2 receptors, and all the potassium channels except the Kca channel ↑(Loh et al., 2017)
rhynchophylline and isorhynchophylline
In vitro
contraction of arterial vessels of isolated rats induced by 60 mM KCl
IC50 = 20–30 μM
l-type Ca2 + channels and a variety of other Ca2 + channels↓
(Zhang et al., 2004)
(by 1 μM Phe)
IC50 = 100 μM
(by 10 nM U46619)
IC50 =200 μM
uncarialin A
In vitro
Phe-induced contraction of rat mesenteric arteries
IC50 = 0.18 μM
l-type calcium channel subunit alpha-1C (Cav1.2)↓
(Yun et al., 2020)
rhynchine A and B
In vitro
HEK293T cells and molecular docking
IC50 = 6.86 and 10.41 μM
Cav3.1 calcium channel ↓
(Zhou et al., 2021)
geissoschizine methyl ether
In vitro
NE- rats aorta strip induced contraction
EC50 = 0.744 μM
NO↑, voltage-dependent Ca2 + channels↓
(Yuzurihara et al., 2002)
Antiinflammation
water extract of
U. rhynchophylla
In vitro
RAW 264.7 cells
1 mg/mL
Akt and MAPK ↓
(NO and IL-1β↓)(Kim et al., 2010)
U. rhynchophylla
alkaloids extractsIn vivo
LPS-induced preeclampsia rat model
35, 70, and 140 mg/kg
(gavage)IL-6, IL-1β, TNF-α, and IFN-γ ↓
(Wu and Xiao, 2019)
rhynchophylline and isorhynchophylline
In vitro
LPS-induced N9 microglial cells
0.3–30 μM
NF-κB and ERK and p38 MAPKs ↓
iNOS protein ↓
(TNF-α, IL-1β and NO↓)(Yuan et al., 2009)
isorhynchophylline
In vitro
LPS-stimulated murine alveolar macrophages
(30 or 40 μM)
TLR4/NF-κB/NLRP3pathway↑
(TNF-α, IL-1β, IL-6, and PAI-1↓)(Zhou et al., 2019)
corynoxeine, isocorynoxeine, rhynchophylline, isorhynchophylline and vincoside lactam
In vitro
LPS-induced primary cultured rat cortical microglia
IC50 = 15.7, 13.7, 18.5, 19.0 and 16.4 μM
–
(Yuan et al., 2008)
strictosidine
In vitro
LPS-induced N9
microglia cellsIC50 = 8.3 μM
–
(Ma et al., 2009b)
uncarinic acid I, 3β-hydroxy-27-p-(E)-coumaroyloxyursan-12-en-28-oic acid and 3β-hydroxy-27-(E)
-
coumaroyl-oleanen-12-en-
28-oic acidIn vitro
LPS-induced
RAW264.7 cellsIC50 = 1.48, 7.01, and 1.89 μM
–
(Zhang et al., 2014)
Anticancer
n-BuOH fraction of
U. rhynchophylla
In vitro
HepG2 cells
0.05, 0.1, and 0.2 mg/mL
caspases 7, 8 and PARP↑
(Kim et al., 2014)
rhynchophylline
In vitro
HepG2 cells
130 µM
p38, ERK, JNK, CREB, Akt and STAT3 signals↓ and p53 signals↑
CXCR4, MMP-9, and MMP-2↓(Lee et al., 2017)
uncarinic acid E
In vitro
HepG2 cells
6, 12, 24, 48 µM
p53↑, Bax/Bcl-2 ↓ and caspases↑
(Zhao et al., 2006)
ursolic acid and rhynchophylline
In vitro
HepG2 cells
50, 25, 12.5 and 6.25 μM
–
(Wu et al., 2017)
3β,6β,19α-trihydroxy-
olean-12-en-28-oic acidIn vitro
MCF-7 and HepG2 cells
IC50 = 78.2 and 73.9 µg/mL
–
(Sun et al., 2012b)
U. rhynchophylla proanthocyanidins
In vitro
MDA-MB-231 cells
IC50 = 5, 10, 20, 30 and 40 µg/mL
G2/M cell cycle arrest
(ROS, Bax/Bcl-2 and cleaved caspase 3 ↑
, mitochondrial membrane potential ↓)(Chen et al., 2017)
hirsutanine D-F
In vitro
MDA-MB-231 cells
100 µM
–
(Pan et al., 2017)
hirsutine
In vitro
MCF-7 and
MDA-MB-231 cellsIC50 = 447.79 and 179.06 µM
Bax/Bcl-2 ↓
caspase 9 and 3 ↑(Huang et al., 2018)
hirsutine
In vitro
MCF-7 cells
IC50 = 62.82 µM
HIF-1α, Snail and MMP-9 ↓
E-cadherin↑(Zhai et al., 2017)
hirsutine
In vivo
A549 xenograft mouse model
40–80 µM
ROCK1/PTEN/PI3K/Akt signaling ↑
(Zhang et al., 2018)
uncarinic acid A,
uncarinic acid B,
uncarinic acid C,
uncarinic acid D,
uncarinic acid E,3β-hydroxy-27-(E)
-coumaroyl-oleanen-12-en-28 -oic acid, 3β-hydroxy-27-p-(E)
-coumaroyloxyursan-12-en-28-oic acid and 3β-hydroxy-27-p-(Z)
-
coumaroyloxyursan-12-en-28-oic acidIn vitro
HCT-15, MCF-7, A549, and HT-1197 cells
IC50 = 0.5–6.5 μM
–
(Lee et al., 2000)
3-diethylamino-5-methoxy-1,2-benzoquinone and 3-ethylamino-5-methoxy-1, 2-benzoquinone
In vitro
A549, HepG2
and A2780 cellsIC50 = 50.2–98.8 μM
–
(Zhang et al., 2016)
isorhynchophyllic acid
IC50 = 5.8, 12.8 and 11.8 μM
hirsutine
In vitro
Jurkat Clone E6-1 cells
10, 25 and 50 μM
Bax, cleaved-caspase 3/9,
Cyto-c protein, caspase-3 and 9 ↑
Bcl-2 protein ↓(Meng et al., 2021)
corynantheidine
In vitro
HL-60 and SW480 cells
IC50 = 13.96,
23.28 µM–
(Wang et al., 2011a)
total alkaloids of Uncaria
In vitro
MDR for vincristine on KBv200 cell line
5 μg/mL
–
(Zhang et al., 2001)
isorhynchophylline
In vitro
MDR of A549/DDP cells
0.5, 1.0 and 1.5 mg/L
efflux of chemotherapeutic drugs ↓
(Zhou et al., 2009)
Antioxidant
petroleum ether extract,
chloroform extract,
ethyl acetate extract,
and ethanol extract of U. rhynchophylla
In vitro
OH radical
scavenging assay20.432, 1.547, 0.0283 and
0.00326 g/L–
(Yin et al., 2010)
Uncariol A, uncariolB, (±)-uncarilin A, (±)-uncarilin B,cinchonain Ia-Id, quercetin, (-)
-epicatechin,
methyl caffeate,
quercetin-3-O-robinobioside and rutinIn vitro
DPPH radical
scavenging assayIC50 = 22.26, 16.12, 10.28, 11.32, 12.67, 14.34, 15.72, 8.27, 3.22, 5.84, 7.52, 8.21, 5.35, 8.14, and 2.13 μM, respectively
–
(Li et al., 2017a)
Antiviral
including mitraphylline, isomitraphylline and uncarine C-F
In vitro
Dengue Virus
1 μg /mL
DENV-antigen ↓
TNF-α, IFN-α and IL-10 ↓(Reis et al., 2008)
hirsutine
In vitro
A549 cells infected
with DENV-110 μM
viral particle assembly, budding,
and release step ↓(Hishiki et al., 2017)
Antiasthma
rhynchophylline
In vitro
TGF-β1 induced hyperplasia of ASMCs
10 µM
TGF‑β1‑induced Smad and MAPK signaling pathways ↓
(Smad4 and phosphorylation of Smad2 and Smad3 ↑, p-ERK1/2 and p-p38 ↑, Smad7↓)(Wang et al., 2019)
rhynchophylline
In vivo
OVA induced
BALB/c mice asthma40 or 80 mg/kg
(gavage)JAK2/STAT3 signaling pathway↓
(IL-6 ↓, SOD, CAT ↑)(Li et al., 2021a)
ASMCs isolated from BALB/c mice
10 μM or 20 μM
LC3 II, beclin-1, and ATG5 ↑,P62 ↓
isorhynchophylline
In vivo
OVA induced
BALB/c mice asthma40 mg/kg
(gavage)FOXC1/NF-kB pathway↓
miR-200a↑
(IL-E, IL-13, IL-4, and IL-5↓)(Zhu et al., 2020).
In vitro
ASMCs isolated from BALB/c mice
10 μM
Sedative and hypnotic
ethanol extracts of
U. rhynchophylla,
U. macrophylla
and U. hirsute
In vivo
Spontaneous activity test in Kunming mice
15 g/kg
(gavage)–
(Chen et al., 2019)
pentobarbital sodium induced
Kunming mice sleep time test
corynoxine and
corynoxine BIn vivo
ICR mice
30 mg/kg
(gavage)mediating of the central dopaminergic system
(Sakakibara et al., 1999)
isorhynchophylline and geissoschizine methyl ether
100 mg/kg
(gavage)
rhynchophylline
In vivo
Wistar rats
5, 10, 15 mg/kg
(intravenous injection)5-HIAA↑, NE↓
(Lu et al., 2003)
corynoxine, corynoxine B, rhynchophelline, and isorhynchophelline
In vivo
ICR mice
100 mg/kg
(gavage)
(Sakakibara et al., 1998)
Anti-epilepsy
rhynchophylline
In vivo
lithium chloride-pilocarpine induced model of SD rats after status convulsion
10、20、40 mg/kg/d
(intraperitoneal injection)Toll-like receptor 4↓,
SOD↑(Wang et al., 2018)
ethanol extract of
U. rhynchophylla
In vitro
SD rat hippocampal slices induced by pilocarpine
1 g/mL
peak potential of pyramidal cells in CA1 region of rat hippocampal slices ↓
(calcium influx and glutamate release ↓)(Xu et al., 2001)
U. rhynchophylla extract
In vivo
KA induced
epileptic SD rats1 g/kg, 5 d/wk
(gavage)RAGE pathway ↓
(S100 B protein and RAGE ↓)(Tang et al., 2017)
rhynchophylline
In vivo
KA induced
epileptic SD rats0.25 mg/kg
TLR and neurotrophin signaling pathways↓
(IL-1β and BDNF ↓)(Ho et al., 2014)
70 % alcoholextract of
U. rhynchophylla
1 g/kg/d
rhynchophylline
In vivo
pilocarpine induced SD rats
100 μM
INaP and NMDAR ↓
(Shao et al., 2016).
geissoschizine methyl ether
SD rats
(IC50 = 1.3–13.3 μM)1–30 μM
(gavage)Nav, Cav, IK and nACh ↓
(Xie et al., 2020)
In vivo
maximal electroshock -induced mouse
50–100 mg/kg
(gavage)
In vivo
6-Hz-induced mouse
seizure model100 mg/kg
(gavage)
Anti-depression
U. rhynchophylla
EtOH extractIn vivo
CUMS-induced depression-like
behaviours in miceEC50 = 17.42 μg/mL
5-HT1A↑
CREB,BDNF and PKA↑(Qiao et al., 2021)
isorhynchophylline
In vivo
reserpine-induced BALB/c mouse depression model
10, 20 and 40 mg/kg (gavage)
NE, 5-HT ↑
MAO-A ↓(Xian et al., 2017)
uncarialin E, G, J and K, 3α-dihydrocadambine, isorhynchophylline, hirsuteine, akuammigine, Z-geissoschizine and corynoxeine
In vitro
Chinese hamster ovary
CHO-K1 cell lineEC50 = 2.2, 0.1, 7.86, 7.32, 1.6, 2.0, 2.24, 1.18, 1.52, and 3.75 μM
5-HT1A receptor ↓
(Liang et al., 2019)
(±)-uncarilins A and B
In vitro
HEK293 cells
0.25 mM
MT1 and MT2 receptors↑
(Geng et al., 2017)
catechin
In vitro
HEK293 cells
EC50 = 25.8 μM and 47.3 μM
MT1 and MT2 receptors↑
(Ekmekcioglu, 2006)
isorhynchophylline
In vivo
CUMS-induced depression-like behaviors
20 or 40 mg/kg/d (gavage)
neurotrophins and
PI3K/ Akt/ GSK-3β pathway ↑(Xian et al., 2019)
rhynchophylline
In vivo
Social defeat C57BL/6 mice
25 mg/kg
(intraperitoneal injection)EphA4 ephexin1 signaling ↓
and BDNF-TrkB signaling ↑(Zhang et al., 2017)
Ischemic brain injury
U. macrophylla alkaloids
In vivo
Focal ischemic model SD rats
–
–
(Xie et al., 2009)
methanol extracts of
U. rhynchophylla
In vivo
transient global ischemia using 4-vessel occlusion model in Wistar rats
100–1000 mg/kg
(intraperitoneal injection)COX-2 ↓
(Suk et al., 2002)
rhynchophylline
In vivo
pMCAO model SD rats
30 mg/kg
(intraperitoneal injection)PI3K/Akt/mTOR signaling pathway↑
and TLRs/NF-κB pathway↓
(claudin-5 and BDNF↑)(Huang et al., 2014)
rhynchophylline
In vitro
primary astrocytes in rats induced by ischemia-repeffusion
0.02 or 0.2 mg/m
necrosis and apoptosis of astrocytes↓
(Gao et al., 2009)
rhynchophylline
In vitro
astrocytes induced by ischemia reperfusion
3 μg/mL
PI3K signaling pathway↓
(Nrf2 nuclear translocation↑)(Yin et al., 2012)
isorhynchophylline
In vivo
a rat model with
MCAO and I/R injury (microglial)20 mg/kg
(gavage)aquaporin-4 expression ↓, lκB-α ↑
NF-κBp65, CX3CR1 expression↓(Deng et al., 2021)
Neuroprotection
crude alkaloids of
U. rhynchophylla
In vitro
NMDA-induced cytotoxicity in the hippocampal slices
1, 10, 100 μg/mL
c-jun, p53, and bax↓
(Lee et al., 2003).
rhynchophylline
In vitro
DA-induced apoptosis of
NT2 cells5 or 50 μM
DNA degradation↓
(Shi and Kenneth, 2002)
isorhynchophylline
In vitro
Aβ25-35-treated PC12 cells
10 or 50 μM
ROS and MAD↓, glutathione↑
DNA fragmentation ↓
caspase-3 ↓ and Bcl-2/Bax ↑(Xian et al., 2012).
5β-carboxystrictosidine
and chlorogenic acidIn vitro
6-OHDA induced mNGF
-differentiated PC12 cells12.5, 25, 50 or 100 μM
ROS↓ (IC50 = 24.5 and 19.7 μM) intracellular calcium levels↓
(IC50 = 46.9 and 27 μM)caspase 3↓
(IC50 = 25.6 and 19.4 μM)caspase 9 ↓
(IC50 = 24.5 and 16.3 μM)(Lin et al., 2020).
rhynchophylline
In vivo
MPTP-induced neurotoxicity
in C57BL/6 mice30 mg/kg
(intraperitoneal injection)PI3K/Akt signaling pathway↑
(LDH and ROS↓)(Zheng et al., 2021)
In vitro
MPP + -induced neurotoxicity in PC12 cells
20 µM
PI3K/Akt signaling pathway↑
(Bax and caspase-3↓, Bcl-2 ↑)
rhynchophylline
In vitro
MPP + -triggered neurotoxicity of primary cerebellar
granule neurons10–50 µM
PI3-K/Akt/GSK3β/MEF2D signaling pathway ↑
(Hu et al., 2018)
Anti-Alzheimer's disease
70 % ethanol extract of
U. rhynchophylla
In vivo
Aβ-mediated neuropathology in 5 × FAD mice
400 mg/kg/d
(gavage)gliosis, neurodegeneration and hippocampal neuron damage ↓
(Shin et al., 2018)
rhynchophylline
In vivo
APP/PS1 transgenic mice
50 mg/kg/d
(gavage)EphA4-dependent signaling ↓
(EphA4 ↓)(Fu et al., 2014)
water, methanol and ethanol extracts of U. rhynchophylla
In vitro
Aβ1-40 and Aβ1-42
100 mg/mL
–
(Fujiwara et al., 2006)
rhynchophylline
In vivo
Aβ1-42-induced spontaneous discharges in the hippocampal CA1 region of SD rats
IC50 = 9.0 μM
(intrahippocampal injection)spontaneous discharges ↓
(Shao et al., 2015)
rhynchophylline
In vivo
Aβ1-42-induced spatial cognition function impairment of SD rats
100 µM
(intrahippocampal injection)extrasynaptic NMDARs-mediated excitatory postsynaptic currents↓
(GluN2B-NMDAR expression ↓)(Yang et al., 2018)
uncarinic acid C
In vitro
Aβ42
50 μM
–
(Yoshioka et al., 2016)
isorhynchophylline
In vitro
-induced neurotoxicity in rat PC12 cells
1, 10 or 50 μM
PI3K/Akt/GSK-3β signaling pathway
(tau protein↓ and p-CREB↑)(Xian et al., 2013)
isorhynchophylline
In vivo
Aβ25-35-induced neuronal apoptosis in hippocampus
20 or 40 mg/kg
(gavage)GSK-3β↓ and
PI3K/Akt signaling pathway↑
(Bcl-2/Bax, cleaved caspase-3,9 and tau protein hyperphosphorylation ↓)(Xian et al., 2014a)
isorhynchophylline
In vivo
TgCRND8 transgenic mice
40 mg/kg
(gavage)Aβ40, Aβ42↓ and
APP processing and phosphorylation↓
(BACE-1, Thr668, PS-1, APH-1 and IDE↑)
TNF-α, IL-6 and IL-1β↓
tau hyperphosphorylation↓
JNK signaling pathway ↑
(p-c-Jun/c-Jun and p-JNK/JNK↓)(Li et al., 2019)
isorhynchophylline
In vivo
d-gal induced
cognitive deficits of mice20 or 40 mg/kg
(gavage)GSH, SOD, CAT and NF-κB ↑
MDA, PGE2, NO, COX-2 and iNOS ↓(Xian et al., 2014b)
isorhynchophylline
In vivo
AlCl3-induced learning and memory impairment in mice
20 or 40 mg/kg
NF-κB signaling pathway↓
(NF-κBp65 and IκBα↓)
AChE ↓(Li et al., 2018)
geissoschizine methyl ether
In vitro
AChE
IC50 = 3.7 μg/mL
–
(Yang et al., 2012)
geissoschizine
methyl ether N-oxideIn vitro
AChE
IC50 = 23.4 μM
–
(Jiang et al., 2015)
rhynchophylloside J
In vitro
AChE
IC50 = 10.5 μM
–
(Guo et al., 2019)
Anti-Parkinson's disease
95 % EtOH extract of
U. rhynchophylla
In vitro
MPP + -induced SH-SY5Y cells
20 μg/mL
Bcl-2, Cyclin D1, p-ERK, p-PI3Kp85, PI3Kp110α, p-AKT, and LC3-I ↑
cleaved caspase 3, Bax, p-JNK,
p-p38, and LC3-II.↓,
DΨm↓(Lan et al., 2018)
water extract of
U. rhynchophylla
In vitro
6-OHDA induced PC12 cells
0.01–5 μg/mL
ROS↓, GSH↑ and caspase-3↓
(Shim et al., 2009)
In vivo
6-OHDA-lesioned rats
5 mg/kg/d
(gavage)dopaminergic neuronal loss ↓
isorhynchophylline
In vitro
MPP + -induced PC12 cells
0.3–100 μM
ASK1/JNK pathway and
IRE1/caspase-12 pathway↓
(ROS↓)(Li et al., 2017b)
corynoxine
In vitro
PC12 cells
25 μM
wild-type and A53T α-syn uclein↓ Akt/mTOR pathway ↑
(p-Akt, p-mTOR, and p-p70 S6↓)(Chen et al., 2014a)
corynoxine B
In vitro
PC12 cells
–
SNCA-HMGB1 interaction↓
(Song et al., 2014)
isorhynchophylline
In vitro
neuronal cells
6.25, 12.5, 25 μM
wild-type, A53T and A30P α-syn monomers↓, α-syn oligomers and α-syn/synphilin-1 aggresomes ↓
(Lu et al., 2012)
Bone protection
water extract of the hooks and stems of U. sinensis
In vitro
RANKL-induced murine bone marrow macrophages and RAW264.7 cells
10–80 μg/mL
NF-kB and T-cells, cytoplasmic 1↓,
serum TRAP5b and C-terminal cross-linked telopeptide of type I collagen↓(Ha et al., 2017)
6 Clinical application
As a traditional Chinese medicine, URCU had the effect of clearing heat and calming the liver, extinguishing wind and settling convulsion. So, URCU preparations are more widely used to treat hypertension and neurological diseases. This article collected Chinese patent medicines or preparations, which contained URCU, such as empirical prescriptions used in folklore, in-hospital preparations, and marketed drugs in Table 4.
Prescription Name
Prescription composition
Functions and Treatments
Gastrodia and Uncaria Decoction
(Tian Ma Gou Teng Yin)Shi jue ming, Tian ma, Yi mu cao、Du zhong、Gou teng, Huang qin,
Niu xi, Sang ji sheng, Shou wu teng, Zhi ziPrimary hypertension
Pregnancy hypertension
Vascular dementia
Parkinson 's disease
Insomnia
Cerebral stroke
Gouteng Jiangya Decoction
San qi, Bai shao, Tian ma, Zhi zi, Sang ji sheng, Du zhong, Mu li, Di long, Gou teng,
Luo bu ma, Suan zao ren, Gan cao, Xiang fu, Dan shenSenile hypertension complicated with depression
Tianteng Jiangya Decoction
Tian ma, Ju hua, Gou qi zi, Sheng di huang, Niu xi, Bai shao yao, Jue ming zi,
Gou teng, Ge teng, Du zhongPrimary hypertension
Lingjiao Gouteng Decoction
Sheng di, Gou teng, Ju hua, Fu shen, Bai shao, Zhu ru, Sang ye,
Chuan bei, Gan cao, Ling yang jiaoSenile hypertension
Acute cerebral hemorrhage
Tianma Jiangya Granules
Tian ma, Gou teng, Shi jue ming, Zhi zi, Niu xi, Yi mu cao, Suan zao ren
Hypertension
Insomnia
Qinggan Antihypertension Granule
Huang lian, Gou teng, Ze xie, Xuan shen
Primary hypertension
Tengfu Jiangya Tablets
Gou teng, Lai fu zi
Hypertension
Gouteng powder
Gou teng,Chen pi,Bna xia,Mai dong,Fu lin,Fu shen,Ren shen,Ju hua,Fang feng,Gan cao,Shi gao
Alzheimer's disease
Insomnia
Tianma Gouteng Granules
Shi jue ming, Tian ma, Yi mu cao、Du zhong、Gou teng, Huang qin,
Niu xi, Sang ji sheng, Shou wu teng, Zhi ziAlzheimer's disease
Chaibei Zhixian Decoction
Chai hu, Tian ma, Zhe bei mu, Di long, Ban xia, Shi chang pu, Mu li
Epilepsy
Jiawei Tianma Gouteng Yin
Shi jue ming, Tian ma, Yi mu cao、Du zhong、Gou teng, Huang qin, Niu xi, Sang ji sheng, Shou wu teng, Zhi zi, Hong jing tian, Jiang huang
Parkinson 's disease
Zhichan particles
Huang qi, Dan shen, Gou teng, Zhi mu, Bai shao, Da hunag, Sheng ma
Parkinson 's disease
Pinggan Maitong Tablets
Tian ma, Gou teng, Shi jue ming, Tian ma, Dan nan xing, Tian zhu huang,
Bei mu, Zhi zi, Shui zhi, Di longParkinson 's disease
Pinggan Huayu Decoction
Tian ma, Gou teng, Chuan qiong, Bai shao, Dang gui, Sheng di, Huang qin,
Dan pi,Dan shen, Di long, Wu gong, Gan caoHeadache
Xinlekang tablets
Gou teng, Suan zao ren, Luo fu mu
Insomnia
Zhenan decoction Decoction
Di huang, Dang gui, Bai shao, Tian ma, Gou teng, Suan zao ren, Ye jiao teng, Xia ku cao, Huang qin, Da zao
Insomnia
Huatan Tongluo Decoction
Ban xia, Ju hong, Zhi qiao, Chan xiong, Hong hua, Yuan zhi, Shi chang pu, Fu lin, Dang shen, Dan shen, Tian ma, Gou teng, Xiang fu, San qi, Dang gui, Chi shao, Ge gen, Gan cao
Cerebral stroke
Sanchong Banxia Baizhu
Tian ma DecoctionQuan xie, Wu gong, Jiang can, Gou teng, Tian ma, Fu lin, Pu huang, Ban xia,
Bai zhu, Bai shao, Chuan xiong, Gan caoCerebral stroke
Tianwu Xiaoyao Decoction
Tian ma, Gou teng, Chai hu, Shi jue ming, Bai shao, Dang gui, Ge gen, Chuan niu xi,
Wu gong, Quan xie, Chuan xiong, Yan hu suo, Bai zhi, Fu lin, Gan caoMigraine
Touteng Granules
Tian ma, Gou teng, Chai hu, Huang qin, Chuan lian zi, Chuan xiong, Bai shao, Dang gui,
Yan hu suo, Zhi zi, Yu jin, Jiang huang, Jiang can, Chan tui, Gan caoMigraine
Yangxue Qingnao Granules
Dang gui, Chuan xiong, Zhen zhu mu, Bai shao, Xia ku cao, Jue ming zi, Yan hu suo,
Xi xin, Di huang, Ji xue teng, Gou tengMigraine
Gouju Qingxiang Decoction
Gou teng, Ju hua, Xia ku cao, Zhi zi, Qing xiang zi, Mu li, Dan shen, Gan cao
Migraine
Shentian Zhidong Decoction
Tai zi shen, Tian ma, Bai zhu, Fu lin, Shi chang pu, Yu jin, Gou teng,
Zhen zhu mu, Yi zhi ren, Bai shaoTicdisorder
Qingre Pinggan Granules
Xia ku cao, Tian ma, Gou teng, Ju hua, Chan tui, Man jin zi, Ji li, Shi hu, Bai shao,
Long gu, Mu li, Dang shen, Chen pi, Gan caoTicdisorder
Qinggan Dingjing Decoction
Shi chang pu, Yu jin, Gou teng, Tian ma, Shi jue ming, Quan xie, Jiang can,
Chuan xiong, Ju hua, Gan caoTicdisorder
Gouteng Yinzi
Quan xie, Chan tui, Fang feng, Jin yin hua, Chuan xiong, Lian qiao, Jiang can, Jie geng,
Niu bang zi, Dan dou chi, Gou teng, Dang shen, Tian ma, Gan caoTicdisorder
Sangye Gouteng Decoction
Sang ye, Gou teng, Ju hua, Chan tui, Jiang can, Can sha, Hu lu cha, Lian qiao,
Huang qin, Gan cao, Pu gong ying, Chai hu, Chen piTicdisorder
Gouteng Dingfeng Decoction
Gou teng, Chuan lian zi, Shi jue ming, Gou qi, Bai ji li, Quan xie, Chan tui,
Di long, Sheng di, Shu di, Gan caoTicdisorder
Tianteng Zhichou Granules
Tian ma, Gou teng, Long gu, Mu li, Jiang can, Chen pi, Bai zhu, Gan cao
Ticdisorder
Wenshen Gouteng Decoction
Dang shen, Gou teng, Lian zi xin, He huan pi, Fu shen, Fu xiao mai,
Huai shan yao, Bu gu zhiMenopausal syndromes
Juhua Gouteng Decoction
Ju hua, Gpu teng, Bai Shao, Fu lin, Ji li, Niu xi, Di huang, Dan pi, Long gu, Mu li
Menopausal syndromes
Tiangou Erxian Decoction
Tian ma, Gou teng, Shi ju ming, Huai niu xi, Ye jiao teng, Sang ji sheng, Du zhong, Huang qin, Zhi zi, Chuan xiong, Dang gui, Yi mu cao, Xian mao, Xian ling pi, Ba ji tian, Huang bai
Menopausal syndromes
Qingxin Zishen Fang
Lian zi xin, Huang lian, Gou teng, Chao zao ren, Di huang,
Shan zhu yu, Dan shen, Fu xiao maiMenopausal syndromes
Gouqin Disui Decoction
Gou teng, Huang qin, Bai shao, Dang gui, Chuan xiong, Mu gua, Ren dong teng, Gan cao
Hemifacial spasm
Shaoyao Gouteng Muer Tang
Bai shao, Gou teng, Gan cao, Yu li ren, Quan xie, Tian ma, Jiang can, Bai ju zi, Hei mu er
Postherpetic neuralgia
6.1 Applications in the treatment of hypertension
Hypertension is a clinical syndrome characterized by increased systemic arterial blood pressure, which can be divided into primary hypertension and secondary hypertension (Oparil et al., 2018). For the treatment of hypertension, TCM has the advantages of fewer side effects, flexible dialectical treatment and remarkable curative effect.
Total of 42 cases of hypertension were treated with Gastrodia and Uncaria Decoction. The Blood pressure and incidence of adverse reactions was significantly shortened and the effective rates were 95.24 % (Zhao, 2021). In addition, 53 cases of senile hypertension complicated with depression were treated with Gouteng Jiangya Decoction combined with paroxetine and captopril. The effective rate of the treatment group was 96.23 % (Ye et al., 2019). Liu et al. found that Gastrodia and Uncaria Decoction combined with labetalol and magnesium sulfate injection was beneficial to control blood pressure level and significantly reduce adverse reactions (Liu et al., 2020). Xu et al. used Tianteng Jiangya Decoction combined with enalapril in the treatment of essential hypertension, the effective rate was 93.75 %. Meanwhile, headache and palpitation symptoms were significantly reduced (Xu and Zou, 2017). Dai used ionic antagonist and Lingjiao Gouteng Decoction to treat 45 elderly hypertensive patients. After 3 months of treatment, the effective rate was 93.3 % (Dai, 2017). Tianma Jiangya Granule and amlodipine were used to treat 30 patients with hypertension. After treatment, the blood pressure of patients was well controlled (Gao and Ding, 2017). Chen used Qinggan Antihypertension Granule in the treatment of 30 patients with hypertension, the effective rate was as high as 96.6 % (Chen, 2005). Li used Tengfu Jiangya Tablets to treat 30 patients with hypertension, and the effective rate was 93 % (Li, 2016).
6.2 Applications in the treatment of neurological diseases
6.2.1 Treatment of dementia
Dementia is a progressive disorder of intelligence, including Alzheimer’s disease and vascular dementia. Li et al. used Tianma Gouteng Yin combined with nimodipine to treat 50 patients with vascular dementia and found that it could effectively improve the cognitive function, daily living ability and dementia degree of patients (Li et al., 2021d). Xu et al. used Gouteng powder in the treatment of 35 cases of alzheimer's disease patients and found that the treatment of AD is reliable, can improve the treatment efficiency and improve the cognitive function of patients (Xu et al., 2016). Chen et al. used Tianma Gouteng Granule to treat 40 patients with alzheimer's disease and found that it could significantly improve the cognitive function and self-living ability of patients (Chen and Guan, 2018).
6.2.2 Treatment of epilepsy
Epilepsy is a chronic disease of sudden abnormal discharge of brain neurons, resulting in transient brain dysfunction. Liu reported 40 cases of drug-resistant epilepsy treated with Chaibei Zhixian Decoction combined with carbamazepine. Three months later, the attack frequency and attack degree of patients were reduced. At the same time, the patient’s energy and attention are improved (Liu, 2020).
6.2.3 Treatment of Parkinson's disease
Parkinson’s disease, also known as tremor paralysis, is characterized by tremor, rigidity and pseudofacial appearance. Hu et al. used Jiawei Tianma Gouteng Yin combined with madopar and senfuluo to treat 20 patients with early Parkinson’s disease. After 1 month, the patient’s condition was improved. After discontinuing the use of Jiawei Tianma Gouteng Yin to continue to use madopar and senfuluo for one month, the therapeutic effect decreased (Hu et al., 2017). Yang et al. used Tianma Gouteng decoction combined with madopar tablets in the treatment of 48 patients with parkinson's disease had a good curative effect, which could improve the patient’s tremor, insomnia and other main symptoms (Yang et al., 2017). Gu reported that the effective rate of Zhichan particles combined with Madopar in the treatment of 38 patients with Parkinson’s disease was 97.30 % (Gu, 2019). Lin reported that Pinggan Maitong Tablets combined with dobutazide tablets in the treatment of parkinson's 29 cases, the patient’s movement disorders were reduced (Lin, 2019).
6.2.4 Treatment of headache
Wang et al. used Pinggan Huayu Decoction to treat 38 cases of headache and found that the headache of patients was effectively improved. And the curative effect is better than flunarizine hydrochloride capsules group (Wang, 2012). Wu et al reported that Tianma Gouteng Yin was superior to flunarizine capsules in treating headache (Wu and Chen, 2020).
6.2.5 Treatment of insomnia
Insomnia usually refers to sleep difficulty or difficulty in maintaining sleep, which is a common set of sleep disorders. Cai et al found that Gouteng Powder was effective in treating insomnia. Patients with parkinson's disease often have sleep disorders (Cai and Wang, 2020). Wang et al. used Tiangouteng decoction combined with Madopar to treat 38 patients with parkinson's insomnia. After treatment, the total sleep time was prolonged and the number of awakenings was reduced (Wang et al., 2013). Wang et al found that after taking Xinlekang tablets, the total sleep time of 43 patients with insomnia was prolonged, the sleep latency and wake time after sleep were shortened, and the sleep efficiency was improved (Wang et al., 2011b). Kong used Tianma Jiangya Granule combined with amlodipine to treat 30 patients with essential hypertension complicated with sleep disorders, and the effective rate was 74.6 % (Kong, 2020). Li used Zhenan decoction combined with zopiclone tablets in the treatment of 32 elderly patients with insomnia. The sleep quality, sleep latency and sleep persistence of the patients were better than those of the control group (Li, 2019).
6.2.6 Treatment of cerebral stroke
On the basis of conventional treatment, zhao et al. used Tian Ma Gou Teng Yin to treat 17 cases of lacunar cerebral infarction, 15 cases were cured, 2 cases were improved, and the effective rate was 100 % (Zhao and Zhang, 2014). Wu used Huatan Tongluo Decoction to treat 30 patients with transient cerebral ischemia. The neurological deficits of patients were restored and daily living abilities were improved (Wu, 2016). Likewise, Xu et al. used Sanchong Banxia Baizhu Tianma Decoction to treat 45 patients, of which 36 cases were significantly improved, accounting for 80 %; 5 cases were improved, accounting for 11.11 % (Xu, 2019).
6.2.7 Treatment of migraine
Cai used Tiangong Xiaoyao decoction combined with flunarizine hydrochloride capsules to treat migraine. The clinical effective rate of 42 patients was 92.86 % (Cai, 2021). Wang reported that headache granules reduced the number and duration of migraine attacks in 35 patients (Wang, 2021). Tang used Yangxue Qingnao Granule to treat 45 patients with migraine, found that it could effectively improve the symptoms of migraine patients and reduce the possibility of recurrence (Tang, 2020). Li ussed Gouju Qingxiang decoction combined with sibelium to treat 31 migraine patients, the symptoms of patients were significantly improved (Li, 2020).
6.2.8 Treatment of ticdisorders
Ticdisorder is a complex and chronic neuropsychiatric disorder characterized by rapid, involuntary, sudden, repetitive, single or multiple muscle movements and vocal tic. Su used Shentian Zhidong decoction in the treatment of tic disorder, the frequency, complexity and tic frequency of 30 patients were reduced (Su, 2017). Fan used Qingre Pinggan granules to treat 30 patients, including 1 case of clinical control, 11 cases markedly effective and 16 cases effective, the total effective rate was 93.30 %, which could effectively reduce the frequency, intensity and complexity of motor and vocal tic (Fan, 2020). Xu used Gouteng Yinzi to treat 30 cases of tic disorder, the effective rate was 93.33 %, and the therapeutic effect was remarkable. Sangye Gouteng Decoction can significantly improve the motor tic and tic symptoms of children in the middle and late stages of tic disorder treatment (Xu, 2020). Gouteng Dingfeng Decoction in the treatment of 31 patients with ticdisorder, 8 cases were cured, 14 cases markedly effective, 6 cases effective, 3 cases ineffective, the total effective rate was 90.3 % (Zhang, 2019). In addition, the effect of Tianteng Zhichou Granules had the same effect as sulpiride hydrochloride in improving motor tic, and the effect is better than sulpiride hydrochloride in improving the type and complexity of vocal tic (Mou, 2020).
6.3 Applications in the treatment of menopausal syndrome
The menopausal syndrome refers to a series of physical and mental symptoms caused by the fluctuation or reduction of sexual hormones in women before and after menopause. Zong et al. used Wenshen Gouteng Decoction combined with tamoxifen in the treatment of 30 patients with perimenopausal syndrome after breast cancer surgery. It was found that it could not only alleviate the symptoms of patients with the menopausal syndrome but also improve the endocrine level of patients (Zong et al., 2021). Lin reported that Juhua Gouteng Decoction combined with nilestriol promoted the rapid disappearance of symptoms in 88 patients, improved the secretion of sex hormones, increased bone mineral density, reduced endometrial thickness (Lin, 2022). Likewise, Tiangou Erxian Decoction in the treatment of 60 patients with menopausal syndromes had a good curative effect, which can improve symptoms (Zhou et al., 2017). Moreover, Yao used Qingxinzishen Decoction to treat 30 patients with menopausal syndrome, and the total clinical effective rate was 86.67 %. It could effectively alleviate the clinical symptoms of patients and improve the level of sex hormone indicators (Yao, 2021).
6.4 Applications in other diseases
Jiang reported that the therapeutic effect of Gouqin Disui Decoction in the treatment of 30 HFS patients was remarkable. The total effective rate of the experimental group was 83.33 % (Jiang, 2018). Deng et al reported that Shaoyao Gouteng Muer Decoction had good clinical efficacy in the treatment of postherpetic neuralgia, and the effective rate of 28 patients was 92.86 % (Deng et al., 2016). Li et al. used Lingjiao Gouteng Decoction combined with glycerol fructose to treat acute cerebral hemorrhage with remarkable clinical effect, which could effectively promote the reduction of cerebral hematoma and peripheral edema, improve cerebral blood flow, alleviate local inflammatory response, protect cerebral nerve cells (Li et al., 2021e).
7 Discussion
This review summarized current research development regarding botany, phytochemistry, pharmacology and clinical application of URCU. More than 371 compounds have been isolated and identified from this genus. Meanwhile, modern pharmacological research revealed that URCU had significant pharmacological properties including anti-hypertension, antiinflammation, anticancer, antioxidant, antiviral, anti-epilepsy, anti-depressant, ischemic brain injury, neuroprotection, anti-Alzheimer's disease, anti-Parkinson's disease and antiasthma. Regardless, there are still several aspects that need to be concerned in the further development of URCU.
Firstly, we collected 75 papers about the chemical constituents of URCU, including 45 papers about U. rhynchophylla, 9 papers about U. hirsuta, 10 papers about U. macrophylla, 7 papers about U. sinensis and 4 papers about U. sessilifructus. In addition, 371 compounds have been reported from URCU, including 53 from U. hirsuta, 60 from U. macrophylla, 253 from U. rhynchophylla, 56 from sessilifructus and 51 from U. sinensis (Fig. 24). Based on these statistics, we knew that the current phytochemical studies on URCU mainly focused on U. rhynchophylla and other URCU had not yet been comprehensively investigated.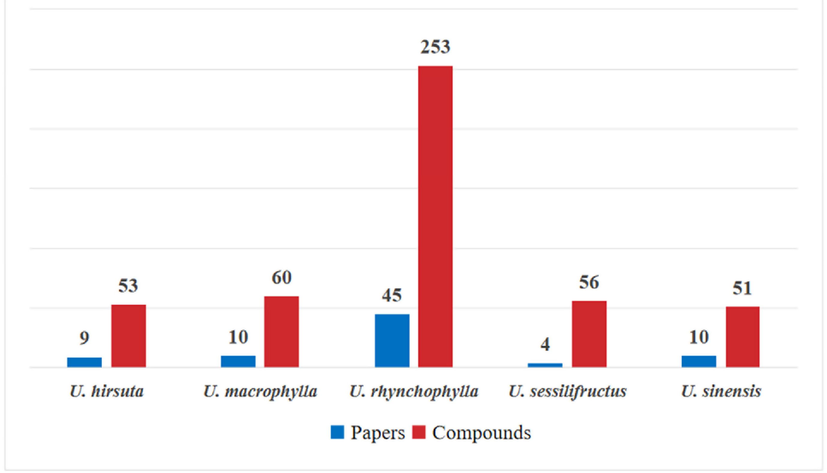
The amount of all published chemical reports and secondary metabolites regarding URCU.
Secondly, 169 alkaloids have been reported from URCU, including 122 monoterpene indole alkaloids, 13 β-carboline alkaloids, 5 cadambine alkaloids, 4 dimeric isoechinulin-type alkaloids, 14 other indole alkaloids and 11 other alkaloids (Figs. 25-26). Meanwhile, monoterpene indole alkaloids are the main alkaloids. It is worth noting that the chemical structure of monoterpene indole alkaloids is often unstable due to the existence of multiple chiral centers. The structure is easily affected by many factors such as temperature, PH and solvent polarity, which also leads to the difficulty in the separation of alkaloids from URCU. How to solve this problem is the breakthrough and key to finding more novel alkaloids.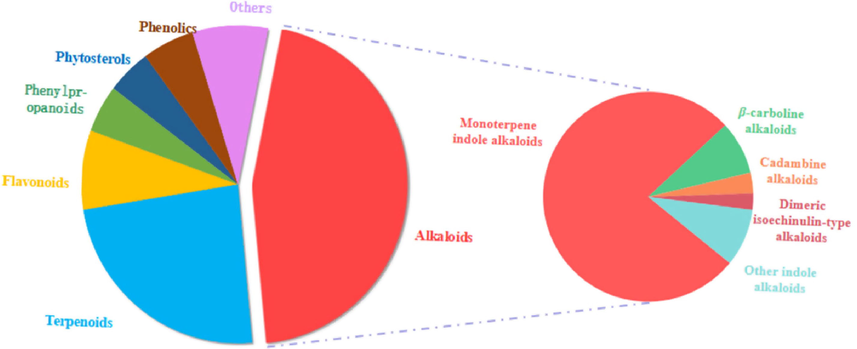
Distribution of the secondary metabolites among URCU.
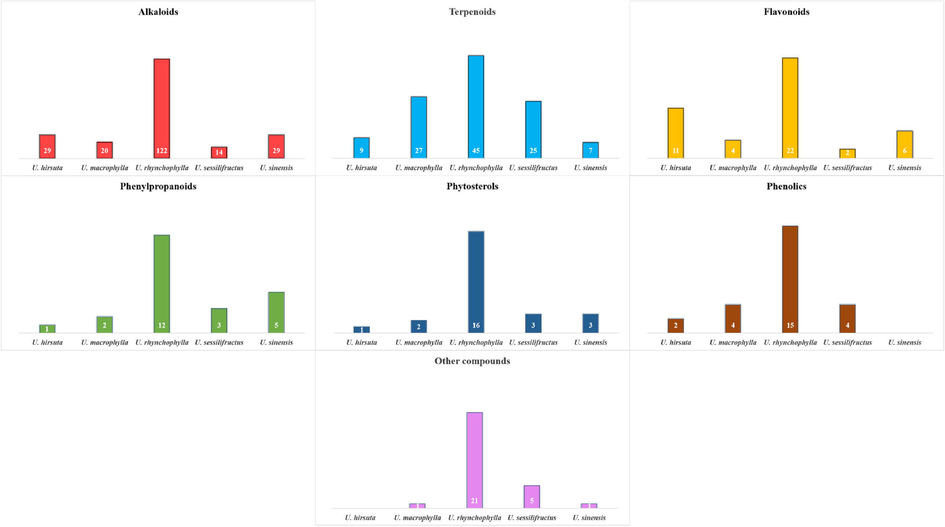
The amount of each chemical classes among URCU.
Thirdly, monoterpene indole alkaloids are the main active compounds, which have a variety of pharmacology activities. Rhynchophylline (34) and isorhynchophylline (44) showed excellent activity in anti-Alzheimer's disease (Yang et al., 2018; Xian et al., 2014a), antiinflammation (Yuan et al., 2009) and anti-hypertension (Zhang et al., 2004). Hirsutine (1) has significant anticancer (Huang et al., 2018) and antiviral (Moradi et al., 2018) effects. But more monoterpene indole alkaloids with various activities also need to be found in this genus. Of course, it can not be ignored that monomeric compounds with outstanding pharmacological activities can be considered the source of new drugs with excellent therapeutic effects.
Fourth, the traditional medicinal part of URCU is the stems with hooks. Leaves, stems without hooks and other parts are not effectively utilized. After reviewing the chemical constituents of URCU, we found that there were also a large number of active components in leaves and stems. Therefore, attention should also be paid to the development of the non-medical parts of URCU, to realize the effective utilization of URCU resources.
Finally, URCU are valuable plant resources that deserve further research and development. This review could be a useful tool in assisting researchers in the selection of interesting species or isolated compounds for further studies, as well as expand the research of URCU.
8 Conclusion
URCU (Uncariae Ramulus Cum Uncis) is a common TCM used to extinguish wind and settle convulsion, which has been used in Traditional Chinese medicines or folk medicines to treat various diseases. This review summarized all the compounds of URCU, including alkaloids, terpenoids, flavonoids, phenylpropanoids, phytosterols and phenolics. Alkaloids were generally considered major bioactive ingredients in URCU, exhibiting various important qualities. In addition, pharmacological studies showed that compounds and extracts isolated from URCU possessed a wide range of pharmacological activities, such as anti-hypertension, antiinflammation, anticancer, antioxidant, antiviral, anti-epilepsy, anti-depressant, ischemic brain injury, neuroprotection, anti-alzheimer's disease, anti-parkinson's disease and antiasthma. In short, as a source of traditional folk medicine, the URCU has been widely used in medicine. Therefore, we believe it’s necessary to review it, which will help to gain a greater understanding and appreciation of URCU.
Author contributions
All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
Funding
We thank the foundations of the National Natural Science Foundation of China (82104368) and Shaanxi Provincial Science and Technology Department Project (2021JQ-744) for financial support of this study.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Neuroprotective effect of 1-methoxyoctadecan-1-ol from Uncaria sinensis on glutamate-induced hippocampal neuronal cell death. J Ethnopharmacol.. 2014;155:293-299.
- [CrossRef] [Google Scholar]
- Studies on plants containing indole alkaloids. VIII. Indole alkaloid glycosides and other constituents of the leaves of Uncaria rhynchophylla Miq. CHEM & PHARM BULL.. 1982;30:4046-4051.
- [CrossRef] [Google Scholar]
- Clinical efficacy of Tiangong Xiaoyao Decoction in the treatment of migraine patients with hyperactivity of liver yang. Fujian: Fujian University of Traditional Chinese Medicine; 2021. [Dissertation]
- Alkaloids in water extract of Uncaria rhynchophylla. Acta Pharm Sin.. 2019;54:1075-1081.
- [CrossRef] [Google Scholar]
- Professor Wang Zhonglin uses Gouteng Powder to treat insomnia. J Cardiovasc Cerebrovasc Dis Integr Trad Chin Western Med.. 2020;18:2352-2353.
- [Google Scholar]
- Qinggan Jiangya Granule in the treatment of essential hypertension clinical research [Dissertation]. Shandong: Shandong University of Traditional Chinese Medicine; 2005.
- Chen Z.J., Yan Y.M., Zhou Y.T., Zhu C., Huo C.Y., Liu W., Luo C.H. and Mo Z.X., 2019. Studies on the inhibitory effects of extracts from stems and leaves of four Uncaria plants on the central nervous system. Chin J Clin Pharmacol. 35, 157-160. https://doi.org/10.13699/j.cnki. 1001-6821.2019.02.018.
- Clinical Observation on 40 Cases of Alzheimer 's Disease Treated with Tianma Gouteng Granule. Henan Trad Chin Med.. 2018;38:1182-1184.
- [CrossRef] [Google Scholar]
- Proanthocyanidins from Uncaria rhynchophylla induced apoptosis in MDA-MB-231 breast cancer cells while enhancing cytotoxic effects of 5-fluorouracil. Food Chem Toxicol.. 2017;107:248-260.
- [CrossRef] [Google Scholar]
- Corynoxine, a Natural Autophagy Enhancer, Promotes the Clearance of Alpha-Synuclein via Akt/mTOR Pathway. J Neuroimmune Pharm.. 2014;9:380-387.
- [CrossRef] [Google Scholar]
- Study on non-alkaloids in Uncaria rhynchophylla. Mountain Agr Biol.. 2014;33:91-94.
- [Google Scholar]
- Systematic Separation of Chemical Constituents from Uncaria rhynchophylla. Shandong: Shandong University of Traditional Chinese Medicine; 2017. [Dissertation]
- Pharmacopoeia of the People's Republic of China. 2020;Vol. 1:268.
- Dai, S.X., 2017. Clinical observation of Lingjiao Gouteng decoction combined with calcium antagonist in the treatment of senile hypertension. Combined trad Chin western med cardiovasc dis electronic magazine. 5, 127 + 130.
- Deng, M.C., Jiao, W., Dong, W.W., Yang, C.B., Lu, R.H., 2009. Study on chemical constituents of Uncaria rhynchophylla. Nat Prod Res Dev. 21, 242-245. https://doi.org/10.16333/ j.1001-6880.2009.02.026.
- Deng, Y., Tan, R., Li, F., Liu, Y., Shi, J., Gong, Q., 2021. Isorhynchophylline Ameliorates Cerebral Ischemia/Reperfusion Injury by Inhibiting CX3CR1-Mediated Microglial Activation and Neuroinflammation. Front Pharmacol. https://doi.org/12:10.3389/fphar.2021.574793.
- Clinical Observation on 28 Cases of Postherpetic Neuralgia Treated with Gouteng Muer Decoction. J Shanxi Univ of Trad Chin Med.. 2016;17:46-47.
- [Google Scholar]
- Study on chemical constituents and antioxidant activity of Uncaria rhynchophylla [Dissertation]. Guangxi: Guangxi Normal University; 2010.
- Melatonin receptors in humans: biological role and clinical relevance. Biomedicine Pharmacother.. 2006;60:97-108.
- [CrossRef] [Google Scholar]
- Clinical observation of Qingre Pinggan Granule in the treatment of children with tic disorder hyperactivity of liver wind type. Heilongjiang: Heilongjiang University of Traditional Chinese Medicine; 2020. [Dissertation]
- A novel pentacyclic triterpene from the canes of Uncaria sessilifructus (Rubiaceae) Nat Prod Res.. 2022;36:668-673.
- [CrossRef] [Google Scholar]
- Blockade of EphA4 signaling ameliorates hippocampal synaptic dysfunctions in mouse models of Alzheimer’s disease. P Natl A Sci.. 2014;111:9959-9964.
- [CrossRef] [Google Scholar]
- Uncaria rhynchophylla, a Chinese medicinal herb, has potent antiaggregation effects on Alzheimer's β-amyloid proteins. J Neurosci Res.. 2006;84:427-433.
- [CrossRef] [Google Scholar]
- Gao, Y., Ding, B.Y., 2017. Clinical Observation of Compound Tianma Jiangya Granule in the Treatment of Hypertensive Liver Yang and Blood Pressure Variability. J Clin Chin Med. 29, 510-514. https://doi.org/10.16448/ j.cjtcm.2017.0172.
- Preventive effect of the rhynchophylline on the lesion of cultured primary astrocytes in rats induced by ischemia-reperfusion. Pharmaceut Clin Res.. 2009;17:1-4.
- [CrossRef] [Google Scholar]
- (±)-Uncarilins A and B, Dimeric Isoechinulin-Type Alkaloids from Uncaria rhynchophylla. J Nat Prod.. 2017;80:959-964.
- [CrossRef] [Google Scholar]
- Study on anti-inflammatory chemical constituents of Gongshuang Huazizhu and Gouteng. Yunnan: Yunnan University; 2021. [Dissertation]
- Zhichan Clinical study of Granule in treating Parkinson 's disease with dyskinesia. Shanghai: Shanghai University of Traditional Chinese Medicine; 2019. [Dissertation]
- Guo, Q., Yang, H., Liu, X., Si, X., Liang, H., Tu, P., Zhang, Q., 2018. New zwitterionic monoterpene indole alkaloids from Uncaria rhynchophylla. Fitoterapia. 127:47-55. https://doi.org/ 10.1016/j.fitote.2018.01.013.
- Glucoconjugated Monoterpene Indole Alkaloids from Uncaria rhynchophylla. J Nat Prod.. 2019;82:3288-3301.
- [CrossRef] [Google Scholar]
- Water extract of Uncaria sinensis suppresses RANKL-induced bone loss by attenuating osteoclast differentiation and bone resorption. Integr Med Res.. 2017;6:434-442.
- [CrossRef] [Google Scholar]
- Hirsutine, an Indole Alkaloid of Uncaria rhynchophylla, Inhibits Late Step in Dengue Virus Lifecycle. Front Pharmacol.. 2017;8:10. https://doi.org/3389/fmicb.2017.01674
- [Google Scholar]
- Uncaria rhynchophylla and rhynchophylline improved kainic acid-induced epileptic seizures via IL-1β and brain-derived neurotrophic factor. Phytomedicine.. 2014;21:893-900.
- [CrossRef] [Google Scholar]
- Monoamine oxidase B (MAO-B) inhibition by active principles from Uncaria rhynchophylla. J Ethnopharmacol.. 2005;100:216-220.
- [CrossRef] [Google Scholar]
- Hu, Q.L., Zhong, S.S., Zeng, Z.L., Liao, H.Y., Hu, Y.X., Yang, H., Li, Z.G., Bao, Z.Y., Luo, J.P., Liao, S.X., 2017. Clinical study on the neuroprotective effect of Jiawei Tianma Gouteng Decoction in the treatment of early Parkinson’s disease. Chin mod drug appl. 11, 136-137. https://doi.org/10.14164/ j.cnki.cn11-5581/r.2017.16.080.
- Hu, S., Mak, S., Zuo, X., Li, H., Wang, Y., Han, Y., 2018. Neuroprotection Against MPP+-Induced Cytotoxicity Through the Activation of PI3-K/Akt/GSK3β/MEF2D Signaling Pathway by Rhynchophylline, the Major Tetracyclic Oxindole Alkaloid Isolated From Uncaria rhynchophylla. Front Pharmacol. 9, 10. https://doi.org/3389/fphar.2018.00768.
- Determination of five active components in Uncaria rhynchophylla leaves. Nat Prod Res Dev.. 2019;31:1731-1737.
- [Google Scholar]
- Study on Herbology of Uncaria. Chin Tradit Herbal Drugs.. 2016;39:2902-2906.
- [CrossRef] [Google Scholar]
- Hirsutine induces apoptosis of human breast cancer MDA-MB-231 cells through mitochondrial pathway. Acta physiologica Sin.. 2018;70:40.
- [Google Scholar]
- Neuroprotective Effects of Rhynchophylline Against Ischemic Brain Injury via Regulation of the Akt/mTOR and TLRs Signaling Pathways. Molecules.. 2014;19:11196-11210.
- [CrossRef] [Google Scholar]
- New monoterpenoid alkaloids from the aerial parts of Uncaria hirsuta. Nat Prod Res.. 2014;28:971-975.
- [CrossRef] [Google Scholar]
- Clinical study on the improvement of quality of life in patients with idiopathic hemifacial spasm. Shandong: Shandong University of Traditional Chinese Medicine; 2018. [Dissertation]
- Geissoschizine methyl ether N-oxide, a new alkaloid with antiacetylcholinesterase activity from Uncaria rhynchophylla. Nat Prod Res.. 2015;29:842-847.
- [CrossRef] [Google Scholar]
- Alkaloid distribution in Malaysian Uncaria. Phytochemistry.. 1992;31:2031-2034.
- [CrossRef] [Google Scholar]
- Uncaria rhynchophylla Inhibits the Production of Nitric Oxide and Interleukin-1β Through Blocking Nuclear Factor κB, Akt, and Mitogen-Activated Protein Kinase Activation in Macrophages. J Med Food.. 2010;13:1133-1140.
- [CrossRef] [Google Scholar]
- n-butanol fraction of Uncaria rhynchophylla induces apoptosis in human hepatoma cancer cells through activation of PARP. Biotechnol Bioproc E.. 2014;19:411-418.
- [CrossRef] [Google Scholar]
- Clinical Study of Compound Tianma Jiangya Granule in the Treatment of Hypertensive Patients with Sleep Disorder of Yin Deficiency and Yang Hyperactivity [Dissertation]. Anhui: Anhui University of Traditional Chinese Medicine; 2020.
- Tetracyclic indole alkaloids with antinematode activity from Uncaria rhynchophylla. Nat Prod Res.. 2017;31:1403.
- [CrossRef] [Google Scholar]
- Uncaria rhynchophylla Ameliorates Parkinson’s Disease by Inhibiting HSP90 Expression: Insights from Quantitative Proteomics. Cell Physiol Biochem.. 2018;47:1453-1464.
- [CrossRef] [Google Scholar]
- Isorhynchophylline, a Potent Plant Alkaloid, Induces Apoptotic and Anti-Metastatic Effects in Human Hepatocellular Carcinoma Cells through the Modulation of Diverse Cell Signaling Cascades. Int J Mol Sci.. 2017;18:1095.
- [CrossRef] [Google Scholar]
- Inhibition of Phospholipase Cγ1 and Cancer Cell Proliferation by Triterpene Esters from Uncaria rhynchophylla. J Nat Prod.. 2000;63:753-756.
- [CrossRef] [Google Scholar]
- Alkaloid fraction of Uncaria rhynchophylla protects against N-methyl-d-aspartate-induced apoptosis in rat hippocampal slices. Neurosci Lett.. 2003;348:51-55.
- [CrossRef] [Google Scholar]
- Uncarinic acids: Phospholipase Cγ1 inhibitors from hooks of Uncaria rhynchophylla. Bioorg Med Chem Lett.. 1999;9:1429-1432.
- [CrossRef] [Google Scholar]
- The clinical efficacy of traditional Chinese medicine compound Tengfu Jiangya Tablets in the treatment of hypertension with hyperactivity of liver yang and the effect of its components on autophagy of vascular endothelial cells [Dissertation]. Shandong: Shandong University of Traditional Chinese Medicine; 2016.
- Clinical study of Zhenan Decoction in the treatment of senile insomnia with blood deficiency and liver hyperactivity syndrome. Shandong: Shandong University of Traditional Chinese Medicine; 2019. [Dissertation]
- Gou Ju Qing Xiang Decoction in the treatment of migraine with hyperactivity of liver Yang. Xinjiang: Xinjiang Medical University; 2020. [Dissertation]
- Suppression of autophagy through JAK2/STAT3 contributes to the therapeutic action of rhynchophylline on asthma. BMC Complement Med.. 2021;21:21.
- [CrossRef] [Google Scholar]
- New phenylpropanoid-substituted flavan-3-ols and flavonols from the leaves of Uncaria rhynchophylla. Fitoterapia.. 2017;116:17-23.
- [CrossRef] [Google Scholar]
- Chemical constituents from leaves of Uncaria rhynchophylla. Chin Tradit Herbal Drugs.. 2017;48:1499-1505.
- [Google Scholar]
- Minor triterpenes from an aqueous extract of the hook-bearing stem of Uncaria rhynchophylla. J Asian Nat Prod Res.. 2021;23:307-317.
- [CrossRef] [Google Scholar]
- Clinical study of Tianma Gouteng decoction combined with nimodipine in the treatment of mild to moderate vascular dementia. J Cardiovasc Cerebrovasc Dis Integr Trad Chin Western Med.. 2021;19:1899-1901.
- [Google Scholar]
- Isorhynchophylline alleviates learning and memory impairments induced by aluminum chloride in mice. Chin Med.. 2018;13:29.
- [CrossRef] [Google Scholar]
- Isorhynchophylline ameliorates cognitive impairment via modulating amyloid pathology, tau hyperphosphorylation and neuroinflammation: Studies in a transgenic mouse model of Alzheimer’s disease. Brain. Behav, Immun.. 2019;82:264-278.
- [CrossRef] [Google Scholar]
- Clinical Study on Lingjiao Gouteng Decoction Combined with Glycerol Fructose in the Treatment of Acute Cerebral Hemorrhage. New Trad Chin Med.. 2021;53:24-29.
- [Google Scholar]
- Study on chemical constituents of Uncaria macrophylla. Chin mod appl pharm.. 2010;27:1193-1195.
- [CrossRef] [Google Scholar]
- Deciphering the Mechanism of the Anti-Hypertensive Effect of Isorhynchophylline by Targeting Neurotransmitters Metabolism of Hypothalamus in Spontaneously Hypertensive Rats. ACS Chem Neurosci.. 2020;11:1563-1572.
- [CrossRef] [Google Scholar]
- Isorhynchophylline Attenuates MPP+-Induced Apoptosis Through Endoplasmic Reticulum Stress- and Mitochondria-Dependent Pathways in PC12 Cells: Involvement of Antioxidant Activity. NeuroMol Med.. 2017;19:480-492.
- [CrossRef] [Google Scholar]
- Minor alkaloids from an aqueous extract of the hook-bearing stem of Uncaria rhynchophylla. J Asian Nat Prod Res.. 2021;23:513-526.
- [CrossRef] [Google Scholar]
- Uncarialins A-I, Monoterpenoid Indole Alkaloids from Uncaria rhynchophylla as Natural Agonists of the 5-HT1A Receptor. J Nat Prod.. 2019;82:3302-3310.
- [CrossRef] [Google Scholar]
- A pair of new oxindole alkaloids isolated from Uncaria macrophylla. Nat Prod Res.. 2021;1–7
- [CrossRef] [Google Scholar]
- Clinical observation of Ping Gan Mai Tong tablets in the treatment of phlegm-heat wind type Parkinson ' s disease [Dissertation]. Xinjiang: Xinjiang Medical University; 2019.
- Clinical analysis of Juhua Gouteng Decoction in the treatment of perimenopausal syndrome of yin deficiency and yang hyperactivity. Chin foreign med treat.. 2022;41:9-13.
- [CrossRef] [Google Scholar]
- In vitro and in vivo evaluation of the neuroprotective activity of Uncaria hirsuta Haviland. J Food Drug Anal.. 2020;28:147-158.
- [CrossRef] [Google Scholar]
- Chemical constituents of Arthropoda and Uncaria macrophylla [Dissertation]. Yunnan: Yunnan University of Traditional Chinese Medicine; 2017.
- Clinical study of Chaibei Zhixian Decoction in the treatment of drug-resistant epilepsy and its anti-drug resistance mechanism of main blood components in rats with epilepsy induced by KA. Beijing: Beijing University of Traditional Chinese Medicine; 2020. [Dissertation].
- Studies on the Chemical Constituents of the Non-Medicinal Parts of Uncaria rhynchophylla from Guizhou Provinceand their Biological Activity [Dissertation]. Guizhou: Guizhou Medical University; 2021.
- Liu, H.M., Yang, J.Z., Liu, G., Feng, X.Z., 1993b. Study on chemical constituents of Uncaria sinensis. Chin Tradit Herbal Drugs. 24: 61-63 + 110.
- Oxindole alkaloids from Uncaria sinensis. Phytochemistry.. 1993;33:707-710.
- [CrossRef] [Google Scholar]
- Effect of Tianma Gouteng Decoction on multi-index monitoring of pregnancy-induced hypertension syndrome and clinical efficacy analysis. J Hebei Trad Chin Med.. 2020;35:32-35.
- [Google Scholar]
- Studies on the Chemical Constituents of Sterols in Uncaria rhynchophylla. Chin J Chin Mater Med.. 2022;47:684-691.
- [CrossRef] [Google Scholar]
- Study on chemical constituents of Uncaria sinensis. Nat Prod Res Dev.. 2011;23 1058-1060+1132. https://doi.org/10.16333/ j.1001-6880.2011.06.013
- [Google Scholar]
- New indole alkaloids glycosides from Uncaria sinensis. Acta Pharm Sin.. 1993;11:849-853.
- [Google Scholar]
- Research Progress on Antitumor Mechanism of Natural Drugs. Med Rev.. 2015;21:1778-1780.
- [Google Scholar]
- Studies on chemical constituents and antitumor activities of stems and leaves of Uncaria rhynchophylla from Guizhou. Guangxi plants.. 2021;41:1061-1069.
- [Google Scholar]
- Mechanisms of Action of Uncaria rhynchophylla Ethanolic Extract for Its Vasodilatory Effects. J Med Food.. 2017;20:895-911.
- [CrossRef] [Google Scholar]
- Isorhynchophylline, a natural alkaloid, promotes the degradation of alpha-synuclein in neuronal cells via inducing autophagy. Autophagy.. 2012;8:98-108.
- [CrossRef] [Google Scholar]
- Effects of rhynchophylline on norepinephrine and 5-hydroxyindoleacetic acid in rat brain. Guizhou med.. 2003;09:771-773.
- [Google Scholar]
- Flavonol glycosides and triterpenes from the leaves of Uncaria rhynchophylla (Miq.) Jacks. J Asian Nat Prod Res.. 2009;4:85-91.
- [Google Scholar]
- Three New Alkaloids from the Leaves of Uncaria rhynchophylla. Helv Chim Acta.. 2009;92:1575-1585.
- [CrossRef] [Google Scholar]
- Inhibitory effect and mechanism of action (MOA) of hirsutine on the proliferation of T-cell leukemia Jurkat clone E6–1 cells. Peerj.. 2021;9:e10692.
- [Google Scholar]
- Clinical study of Tianteng Zhitui Granule in treating tic disorder of spleen deficiency and hyperactivity of liver type [Dissertation]. Hebei: Hebei University; 2020.
- Hypertension. Nat Rev Dis Primers.. 2018;4:18014.
- [CrossRef]
- New monoterpenoid oxindole alkaloid derivatives from the stems of Uncaria hirsuta Havil. and their cytotoxicity and tandem mass spectrometric fragmentation. Fitoterapia.. 2017;116:85-92.
- [CrossRef] [Google Scholar]
- Oxindole alkaloids from Uncaria macrophylla. Phytochemistry.. 1973;12:2795-2798.
- [CrossRef] [Google Scholar]
- Uncaria rhynchophylla ameliorates unpredictable chronic mild stress-induced depression in mice via activating 5-HT1A receptor: Insights from transcriptomics. Phytomedicine.. 2021;81:153436
- [CrossRef] [Google Scholar]
- Ischemic brain injury: New insights on the protective role of melatonin. Free Radic Biol Med.. 2017;104:32-53.
- [CrossRef] [Google Scholar]
- Immunomodulating and antiviral activities of Uncaria tomentosa on human monocytes infected with Dengue Virus-2. Int Immunopharmacol.. 2008;8:468-476.
- [CrossRef] [Google Scholar]
- Effect of oxindole alkaloids from the hooks of Uncaria macrophylla on thiopental-induced hypnosis. Phytomedicine.. 1998;5:83-86.
- [CrossRef] [Google Scholar]
- Effect on locomotion of indole alkaloids from the hooks of Uncaria plants. Phytomedicine.. 1999;6:163-168.
- [CrossRef] [Google Scholar]
- Rhynchophylline Protects Against the Amyloid β-Induced Increase of Spontaneous Discharges in the Hippocampal CA1 Region of Rats. Neurochem Res.. 2015;40:2365-2373.
- [CrossRef] [Google Scholar]
- Anticonvulsant effect of Rhynchophylline involved in the inhibition of persistent sodium current and NMDA receptor current in the pilocarpine rat model of temporal lobe epilepsy. Neurosci.. 2016;337:355-369.
- [CrossRef] [Google Scholar]
- Effect of rhynchophylline on apoptosis induced by dopamine in NT2 cells. Acta Pharmacol Sin.. 2002;23:445-449.
- [CrossRef] [Google Scholar]
- Effects of the hook of Uncaria rhynchophylla on neurotoxicity in the 6-hydroxydopamine model of Parkinson's disease. J Ethnopharmacol.. 2009;126:361-365.
- [CrossRef] [Google Scholar]
- Uncaria rhynchophylla ameliorates amyloid beta deposition and amyloid beta-mediated pathology in 5XFAD mice. Neurochem Int.. 2018;121:114-124.
- [CrossRef] [Google Scholar]
- Ameliorating effect of new constituents from the hooks of Uncaria rhynchophylla on scopolamine-induced memory impairment. Chin J Nat Med.. 2013;11:391-395.
- [CrossRef] [Google Scholar]
- HMGB1 is involved in autophagy inhibition caused by SNCA/α-synuclein overexpression. Autophagy.. 2014;10:144-154.
- [CrossRef] [Google Scholar]
- Megastigmane components in water extract of Uncaria rhynchophylla. Acta Pharm Sin.. 2022;57:1832-1839.
- [CrossRef] [Google Scholar]
- Clinical Observation of Shentian Zhidong Decoction in the Treatment of Multiple Tourette Syndrome with Spleen Deficiency and Liver Hyperactivity [Dissertation]. Heilongjiang: Heilongjiang University of Traditional Chinese Medicine; 2017.
- Neuroprotection by methanol extract of Uncaria rhynchophylla against global cerebral ischemia in rats. Life Sci.. 2002;70:2467-2480.
- [CrossRef] [Google Scholar]
- Sun, G.L., Yang, J.S., Si, Y.K., Xu, X.D., Zhong, M.L., Yuan, J.Q., 2011. Separation and structural identification of lignans from Uncaria sinensis. 2011 China Pharmaceutical Congress and the 11 th China Pharmacists Weekly. 334-342.
- Isolation and structural identification of flavonoids from Uncaria sinensis. Chin Pharm J.. 2012;47:177-179.
- [Google Scholar]
- A New Triterpene from the Plant of Uncaria Macrophylla. Molecules.. 2012;17:504-510.
- [CrossRef] [Google Scholar]
- A New Triterpene From Uncaria macrophylla and Its Antitumor Activity. Molecules.. 2012;17:1883-1889.
- [CrossRef] [Google Scholar]
- Observation on the clinical efficacy of Yangxue Qingnao Granule in the prevention and treatment of vestibular migraine with blood deficiency and liver hyperactivity [Dissertation]. Chengdu: Chengdu University of Traditional Chinese Medicine; 2020.
- Long-Term Intake of Uncaria rhynchophylla Reduces S100B and RAGE Protein Levels in Kainic Acid-Induced Epileptic Seizures Rats. Evid-Based Compl Alt.. 2017;2017:9732854.
- [CrossRef] [Google Scholar]
- Uncaria plant in Flora of China @ efloras.org. (2020). Uncaria plant in Flora of China @ efloras.org (Vol. 2020).
- Clinical study of Pinggan Huayu Decoction in the treatment of 38 cases of vascular headache. Mod Chin med appl.. 2012;6:95.
- [Google Scholar]
- Clinical study of Toutong Granule in the treatment of vestibular migraine [Dissertation]. Shandong: Shandong University of Traditional Chinese Medicine; 2021.
- Effect of rhynchophylline on serum SOD level and TLR4 expression in hippocampus of juvenile rats with persistent convulsion. New Chin med.. 2018;50:6-9.
- [CrossRef] [Google Scholar]
- Two folate-derived analogues from an aqueous decoction of Uncaria rhynchophylla. Chin J Nat Med.. 2019;17:928-934.
- [CrossRef] [Google Scholar]
- Clinical Observation on 30 Cases of Sleep Disorders in Patients with Parkinson 's Disease Treated with Tianma Gouteng Decoction. Zhejiang J Trad Chin Med.. 2013;48:809-810.
- [Google Scholar]
- Macrophyllionium and Macrophyllines A and B, Oxindole Alkaloids from Uncaria macrophylla. J Nat Prod.. 2011;74:12-15.
- [CrossRef] [Google Scholar]
- Triterpenoids from the stems of Uncaria macrophylla. J Chin Pharm Sci.. 2015;24:169-176.
- [Google Scholar]
- Clinical intervention study of Huatan Tongluo Decoction on transient ischemic attack [Dissertation]. Liaoning: Liaoning University of Traditional Chinese Medicine; 2016.
- Wu, Y.H, Chen, J.H., 2020. Tianma Gouteng Decoction in the treatment of 30 cases of liver yang hyperactivity type headache. Fujian trad Chin med. 51 , 22-23 + 29 + 91.
- Constituents of leaves of Uncaria hirsuta Haviland. Journal of the Chinese Chemical Society.. 1994;41:209-212.
- [Google Scholar]
- Wu, W.M., Li, Z.F., Ou, Y.H., Xu, Y., He, M.Z., Chen, J., Feng, Y.L., Yang, S.L., 2015. Chemical constituents analysis of Uncaria rhynchophylla. Chin J Exp Tradit Med Form. 21, 56-58. https://doi.org/10.13422/j. cnki. syfjx. 2015180056.
- Chemical constituents of the non-alkaloid fraction of Uncaria macrophylla. J Southern Medl Univ.. 2007;27:226-227.
- [Google Scholar]
- Evaluation of the effects of Uncaria rhynchophylla alkaloid extract on LPS-induced preeclampsia symptoms and inflammation in a pregnant rat model. Braz J Med Biol Res.. 2019;52:e8273.
- [Google Scholar]
- Effects of Ursolic Acid and Rhynchophylline in Uncariae Ramulus Cum Uncis on Human Hepatoma HepG2 Cells. Chin J Information on Trad Chin Med.. 2017;24:63-66.
- [Google Scholar]
- Protective Effect of Isorhynchophylline Against β-Amyloid-Induced Neurotoxicity in PC12 Cells. Cell Mol Neurobiol.. 2012;32:353-360.
- [CrossRef] [Google Scholar]
- Isorhynchophylline Protects PC12 Cells Against Beta-Amyloid-Induced Apoptosis via PI3K/Akt Signaling Pathway. Evid-Based Compl Alt.. 2013;2013:163057
- [CrossRef] [Google Scholar]
- Isorhynchophylline Treatment Improves the Amyloid-β-Induced Cognitive Impairment in Rats via Inhibition of Neuronal Apoptosis and Tau Protein Hyperphosphorylation. J Alzheimer's Dis.. 2014;39:331-346.
- [CrossRef] [Google Scholar]
- Isorhynchophylline improves learning and memory impairments induced by D-galactose in mice. Neurochem Int.. 2014;76:42-49.
- [CrossRef] [Google Scholar]
- Antidepressant-Like Effect of Isorhynchophylline in Mice. Neurochem Res.. 2017;42:678-685.
- [CrossRef] [Google Scholar]
- Isorhynchophylline exerts antidepressant-like effects in mice via modulating neuroinflammation and neurotrophins: involvement of the PI3K/Akt/GSK-3β signaling pathway. The FASEB J.. 2019;33:10393-10408.
- [CrossRef] [Google Scholar]
- Systematic identification and quantification of tetracyclic monoterpenoid oxindole alkaloids in Uncaria rhynchophylla and their fragmentations in Q-TOF-MS spectra. J Pharmaceut Biomed.. 2013;81–82:56-64.
- [CrossRef] [Google Scholar]
- Antiepileptic geissoschizine methyl ether is an inhibitor of multiple neuronal channels. Acta Pharmacologica Sin.. 2020;41:629-637.
- [CrossRef] [Google Scholar]
- Xie, T.G., 2009. Therapeutic effect and chemical composition analysis of Xietangguang. Uncaria rhynchophylla on cerebral ischemia model in rats. [Dissertation]. Beijing, Capital Normal University.
- Chemical constituents in the leaves of Uncaria hirsuta Haviland. Chin J Nat Med.. 2008;04:262-264.
- [Google Scholar]
- Studies on alkaloid constituents inleaves of Uncaria hirsuta. Chin J Chin Mater Med.. 2008;17:2124-2127.
- [Google Scholar]
- Two new alkaloids from the leaves of Uncaria hirsuta Haviland. Chin Chem Lett.. 2008;19:931-933.
- [Google Scholar]
- Study on alkaloids from Uncaria rhynchophylla. Chin Tradit Herbal Drugs.. 2009;40:204-207.
- [Google Scholar]
- Triterpenoids and tannins from the leaves of Uncaria hirsuta. Helvetical Chemical Acta.. 2009;92:638-644.
- [Google Scholar]
- Bis(monoterpenoid) indole alkaloid glucosides from Uncaria hirsuta. Phytochem Lett.. 2011;4:380-382.
- [CrossRef] [Google Scholar]
- Sanchong Banxia Baizhu Tianma Decoction in the treatment of acute ischemic stroke patients with wind phlegm and blood stasis. Fujian: Fujian University of Traditional Chinese Medicine; 2019. [Dissertation]
- Clinical observation of Gouteng Decoction in the treatment of children with tic disorder caused by wind [Dissertation]. Heilongjiang: Heilongjiang University of Traditional Chinese Medicine; 2020.
- Clinical observation of Gouteng Powder extract in the treatment of senile dementia. Shanxi Trad Chin Med.. 2016;32:18-20.
- [Google Scholar]
- Effect of Uncaria rhynchophylla on evoked field potential of hippocampal slices in epileptic rats. Chin J Appl Physiol.. 2001;03:52-54.
- [Google Scholar]
- Clinical Efficacy of Tianteng Jiangya Decoction in the Treatment of Essential Hypertension. J Clin Ration Drug Use.. 2017;10:112-114.
- [Google Scholar]
- Geissoschizine methyl ether, a corynanthean-type indole alkaloid from Uncaria rhynchophylla as a potential acetylcholinesterase inhibitor. Nat Prod Res.. 2012;26:22-28.
- [CrossRef] [Google Scholar]
- Rhynchophylline suppresses soluble Aβ1-42-induced impairment of spatial cognition function via inhibiting excessive activation of extrasynaptic NR2B-containing NMDA receptors. Neuropharmacol.. 2018;135:100-112.
- [CrossRef] [Google Scholar]
- Clinical Observation of Tianma Gouteng Decoction in the Treatment of Parkinson 's Disease. Clin Med Res Practice.. 2017;2:97-98.
- [Google Scholar]
- Yang, M.C., 2018. Studies on the chemical constituents of two medicinal plants, Uncaria rhynchophylla and E. grandis [Dissertation]. Yunnan: Yunnan Normal University.
- Clinical study and network pharmacology analysis of Qing Xin Zi Shen Decoction in the treatment of perimenopausal syndrome with disharmony between heart and kidney. Nanjing: Nanjing University of Traditional Chinese Medicine; 2021. [Dissertation]
- Clinical observation of Compound Gouteng Jiangya Decoction in the treatment of senile hypertension with depression. Trad Chin med rep.. 2019;25:100-103.
- [Google Scholar]
- Antioxidant activity of different solvent extracts and total alkaloids from Uncaria rhynchophylla. J Guangxi Norm Univ (Natural Science Edition).. 2010;28:31-34.
- [CrossRef] [Google Scholar]
- Protective effect and regulatory mechanism of rhynchophylline-induced Nrf2 nuclear translocation on ischemia-reperfusion injury in rat astrocytes. Strait Pharmaceut.. 2012;24:28-31.
- [Google Scholar]
- Semisynthesis and Structure-Activity Studies of Uncarinic Acid C Isolated from Uncaria rhynchophylla as a Specific Inhibitor of the Nucleation Phase in Amyloid β42 Aggregation. J Nat Prod.. 2016;79:2521-2529.
- [CrossRef] [Google Scholar]
- Uncarialins J-M from Uncaria rhynchophylla and Their Anti-depression Mechanism in Unpredictable Chronic Mild Stress-Induced Mice via Activating 5-HT1A Receptor. Chin J Chem.. 2021;39:1331-1343.
- [CrossRef] [Google Scholar]
- Two new monoterpene indole alkaloids from Uncaria sessilifructus. Chin J Chin Mater Med. 2022:1-9.
- [Google Scholar]
- Chemical constituents of Uncaria rhynchophylla. J Baoji Univ of Arts and Sciences.. 2022;42:44-49.
- [Google Scholar]
- Alkaloids from the Leaves of Uncaria rhynchophylla and Their Inhibitory Activity on NO Production in Lipopolysaccharide-Activated Microglia. J Nat Prod.. 2008;71:1271-1274.
- [CrossRef] [Google Scholar]
- Anti-inflammatory effects of rhynchophylline and isorhynchophylline in mouse N9 microglial cells and the molecular mechanism. Int Immunopharmacol.. 2009;9:1549-1554.
- [CrossRef] [Google Scholar]
- The inhibition effect of uncarialin A on voltage-dependent L-type calcium channel subunit alpha-1C: Inhibition potential and molecular stimulation. Int J of Biol Macromol.. 2020;159:1022-1030.
- [CrossRef] [Google Scholar]
- Geissoschizine methyl ether, an indole alkaloid extracted from Uncariae Ramulus et Uncus, is a potent vasorelaxant of isolated rat aorta. Eur J Pharmacol.. 2002;444:183-189.
- [CrossRef] [Google Scholar]
- Therapeutic Mechanism and Key Alkaloids of Uncaria rhynchophylla in Alzheimer’s Disease From the Perspective of Pathophysiological Processes. Front Pharmacol 2021
- [CrossRef] [Google Scholar]
- Effect of hirsutine on hypoxia-induced migration and invasion abilities in human breast cancer MCF-7 cells. Chin J Pathophysiol.. 2017;33:2009-2014.
- [Google Scholar]
- The chemical constituents and bioactivities of Dai medicine 'Huaimianwang' -Baigouteng [Dissertation]. Yunnan: Yunnan University of Traditional Chinese Medicine; 2013.
- Chemical constituents of two plants [Dissertation]. Yunnan: Yunnan University of Traditional Chinese Medicine; 2014.
- Clinical observation of Gouteng Dingfeng Decoction in the treatment of hyperactivity of liver wind type Tourette syndrome [Dissertation]. Zhejiang: Zhejiang University of Traditional Chinese Medicine; 2019.
- In vitro vasodilator mechanisms of the indole alkaloids rhynchophylline and isorhynchophylline, isolated from the hook of Uncaria rhynchophylla (Miquel) Naunyn-Schmiedeberg's Archives Pharmacol.. 2004;369:232-238.
- [CrossRef] [Google Scholar]
- Two new ortho benzoquinones from Uncaria rhynchophylla. Chin J Nat Med.. 2016;14:232-235.
- [CrossRef] [Google Scholar]
- Hirsutine induces mPTP-dependent apoptosis through ROCK1/PTEN/PI3K/GSK3β pathway in human lung cancer cells. Cell Death Dis.. 2018;9:598.
- [CrossRef] [Google Scholar]
- A new monoterpenoid indole alkaloid from Uncaria rhynchophylla. Japan Institute Heterocyclic Chem.. 2020;104:945-951.
- [CrossRef] [Google Scholar]
- Chemical Constituents of Uncaria rhynchophylla. Chem Nat Comp.. 2022;58:129-131.
- [CrossRef] [Google Scholar]
- Study on active constituents of traditional Chinese medicine reversing multidrug resistance of tumor cells in vitro. Chin Med Mat.. 2001;24:655-657.
- [Google Scholar]
- New triterpenic acids from Uncaria rhynchophylla: Chemistry, NO-inhibitory activity, and tandem mass spectrometric analysis. Fitoterapia.. 2014;96:39-47.
- [CrossRef] [Google Scholar]
- Increased EphA4-ephexin1 signaling in the medial prefrontal cortex plays a role in depression-like phenotype. Sci Rep.. 2017;7:7133.
- [CrossRef] [Google Scholar]
- Medicinal uses, phytochemistry and pharmacology of the genus Uncaria. J Ethnopharmacol.. 2015;173:48-80.
- [CrossRef] [Google Scholar]
- Clinical Study of Tianma Gouteng Decoction in Treating Hypertension. Inner Mongolia Trad Chin Medicine.. 2021;40:69-70.
- [Google Scholar]
- The Course of Uncarinic Acid E-Induced Apoptosis of HepG2 Cells from Damage to DNA and p53 Activation to Mitochondrial Release of Cytochrome. Biol Pharm Bull.. 2006;29:1639-1644.
- [CrossRef] [Google Scholar]
- Clinical Observation on 17 Cases of Lacunar Cerebral Infarction Treated with Tianma Gouteng Decoction. J Practical Chin Med.. 2014;28:36-38.
- [CrossRef] [Google Scholar]
- Protection by rhynchophylline against MPTP/MPP+-induced neurotoxicity via regulating PI3K/Akt pathway. J Ethnopharmacol.. 2021;268:113568
- [CrossRef] [Google Scholar]
- Clinical Observation of Tiangou Erxian Decoction in Treating Premenopausal and Postmenopausal Syndromes. J Practical Chin Med.. 2017;33:765.
- [Google Scholar]
- Rhynchines A-E: Cav3.1 Calcium Channel Blockers from Uncaria rhynchophylla. Org Lett.. 2021;23:9463-9467.
- [CrossRef] [Google Scholar]
- Isorhynchophylline exerts anti-inflammatory and anti-oxidative activities in LPS-stimulated murine alveolar macrophages. Life Sci.. 2019;223:137-145.
- [CrossRef] [Google Scholar]
- Reversal effect of isorhynchophylline on lung adenocarcinoma drug-resistant cell line A549/DDP. Chin J New Drugs.. 2009;18:1339-1342.
- [Google Scholar]
- Application of radioligand-receptor binding assays in the search for the active principles of the traditional Chinese medicine ‘Gouteng’. Phytother Res.. 1997;11:231-236.
- [CrossRef] [Google Scholar]
- Isorhynchophylline exerts anti-asthma effects in mice by inhibiting the proliferation of airway smooth muscle cells: The involvement of miR-200a-mediated FOXC1/NF-κB pathway. Biochem Bioph Res Co.. 2020;521:1055-1060.
- [CrossRef] [Google Scholar]
- ‘Wenshen Gouteng Decoction’ in the treatment of 30 cases of perimenopausal syndrome after breast cancer surgery. Jiangsu trad Chin med.. 2021;53:32-34.
- [Google Scholar]







