Translate this page into:
A greener RP-HPTLC-densitometry method for the quantification of apremilast in nanoformulations and commercial tablets: Greenness assessment by analytical eco-scale, ChlorTox, and AGREE methods
⁎Corresponding authors at: Department of Pharmaceutics, College of Pharmacy, King Saud University, P.O. Box 2457, Riyadh 11451, Saudi Arabia(F. Shakeel), Department of Pharmaceutical Sciences, College of Pharmacy, AlMaarefa University, P.O. Box 71666, Ad Diriyah 13713, Saudi Arabia(S. Alshehri). fsahmad@ksu.edu.sa (Faiyaz Shakeel), sshehri@um.edu.sa (Sultan Alshehri)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
The greener “high-performance thin-layer chromatography (HPTLC)” methods for apremilast (APM) analysis in pharmaceutical products and biological fluids are not currently available in the literature. Accordingly, this study involves the development and validation of a rapid, simple, economical, and greener reversed-phase HPTLC methodology for the estimation of APM in prepared nanoparticles (NPs), nanoemulsion, and marketed tablets. The estimation of APM was conducted using “RP-18 silica gel 60 F254S HPTLC plates” as the stationary phase. The combination of ethanol/water (65:35, v/v) was used as the greener mobile phase for APM analysis. The greenness of the method was predicted using three different approaches, namely Analytical Eco-Score (AES), ChlorTox, and the Analytical GREENness (AGREE) approaches. The λmax = 238 nm was used for the APM detection. By contrasting its single band at Rf = 0.61 ± 0.01 with those of pure APM, the HPTLC peaks for APM in an prepared NP formulation, nanoemulsion, and marketed tablets were identified. In the concentration range of 100–700 ng/band, the proposed analytical methodology was linear. The values of AES, ChlorTox, and AGREE were determined to be 93, 0.66 g, and 0.89, respectively, demonstrated an outstanding greener profile for the existing method. The amount of APM in the tablet, NP formulation, and nanoemulsion was found to be 98.40, 101.60, and 99.37 %, respectively. According to the findings of validation tests and pharmaceutical analysis, the suggested analytical technique could be successfully applied for the routine examination of APM in marketed tablets and laboratory generated nanoformulations.
Keywords
Apremilast
Greenness tools
RP-HPTLC
Nanoformulations
Validation
1 Introduction
The United States Food and Drug Administration (FDA) recently approved the small-molecule medication apremilast (APM) for the treatment of plaque arthritis and psoriatic arthritis (FDA, 2014; Schafer et al., 2014; Souto and Gomez-Reino, 2017). It is also proposed for the treatment of several other diseases, but its approval is pending for these treatments (De Luca et al., 2019; Maloney et al., 2020). APM is marketed as Otezla® immediate-release tablets containing 10, 20, or 30 mg of the drug (EMA, 2014; FDA, 2014). It has been characterized as poorly soluble and poorly permeable drug and shows poor bioavailability after oral administration (FDA, 2014; Shakeel et al., 2017a). The bioavailability of APM after oral administration of Otezla® tablets was found to be 20–30 % (FDA, 2014). It has been reported to be poorly soluble in water and various buffer solutions (Shakeel et al., 2017a; Madan et al., 2019). However, it is freely soluble in carbitol, ethyl acetate, and dimethyl sulfoxide; sparingly soluble in polyethylene glycol-400; and slightly soluble in methanol, ethanol, isopropanol, 1-butanol, 2-butanol, ethylene glycol, and propylene glycol (Shakeel et al., 2017b).
The quantification of APM in its bulk form and pharmaceutical preparations has been reported using both qualitative and quantitative analytical approaches (Kulkarni and Deshpande, 2021). Several ultraviolet (UV) spectrophotometric methods have been reported for the estimation of APM in its bulk form and in marketed or in-house prepared pharmaceutical products (Ashok Chakravarthy et al., 2017; Intwala and Doshi, 2017; Ravisankar et al., 2017). Various “high-performance liquid chromatography (HPLC)”-based methods are also used for APM analysis in its different formulations (Anerao et al., 2017; Mathrusri et al., 2017; Sonawane et al., 2018; Chaudhari and Shirkhedkar, 2019). HPLC methods have also been applied for the identification of impurities in APM (Xiong et al., 2016; Landge et al., 2017; Foroughbakhshfasaei et al., 2018). “Liquid chromatography-mass spectrometry (LC-MS)” technique was also applied for the identification and characterization of process-related substances and the degradation products of APM (Lu et al., 2017). Ultra-performance LC-MS/MS (UPLC-MS/MS)-based methods are also applied for APM estimation in rat plasma and its pharmacokinetic analysis (Chen et al., 2016; Iqbal et al., 2016). Tandem mass spectrometry (MS/MS) was utilized to identify the breakdown products of APM after its determination utilizing a stability-indicating “high-performance thin layer chromatography (HPTLC)” approach (Bhole et al., 2019). The quantification of APM in both its bulk form and pharmaceutical preparations has also been done using the HPTLC method (Chaudhari and Shirkhedkar, 2017).
Although, various analytical methods have been reported for the determination of APM in its pure form, pharmaceutical products, and biological samples, none of them are greener. In addition, literature HPTLC methods employ toxic solvents in their mobile phases. Quality control and drug analysis laboratories use large amounts of toxic solvents for pharmaceutical and bioanalytical procedures, causing issues in terms of environmental toxicity (Tantawy et al., 2020a). Moreover, the environmental impacts of analytical and bioanalytical techniques are largely neglected or paid scant attention (Tantawy et al., 2020a; Wadie et al., 2023a). However, the greener analytical technologies or green analytical chemistry (GAC) are garnering increasing research interest (Ostovan et al., 2018; Ibrahim et al., 2019). Despite the various beneficial characteristics of green solvents, they have not been applied frequently, either alone or in combination, as the eluents for the HPTLC assay of pharmaceutical compounds (Ibrahim et al., 2019; Tantawy et al., 2020a; Wadie et al., 2023a).
Greener HPTLC methods present many advantages for the quantifications of drugs and pharmaceuticals compared with conventional HPTLC methods (Rezk et al., 2019; Tantawy et al., 2020b; Wadie et al., 2023b). In addition, APM in pharmaceutical preparations or biological samples has not been quantitatively analyzed using any greener analytical methods. The potential of ethanol/water as the green eluent system has been studied in the analysis of several drugs/phytochemicals such as rivaroxaban, antipsychotics, diosmin, hesperidin, flufenamic acid, and lenvatinib using HPTLC method (Alam et al., 2020; Foudah et al., 2020; Foudah et al., 2021; Hameed et al., 2021; Alam et al., 2023; Bang and Bhatt, 2023). The National Environmental Method Index, Analytical Eco-Score (AES), Green Analytical Procedure Index, Red, Green, and Blue, ChlorTox, and the Analytical GREENness (AGREE) are just a few of the numerous qualitative and quantitative techniques that have been described in the literature to assess the greenness profiles of analytical methodologies (Keith et al., 2005; Plotka-Wasylka, 2018; Nowak and Koscielniak, 2019; Duan et al., 2020; Pena-Pereira et al., 2020; Nowak et al., 2021; Nowak et al., 2023). In the present study, three different tools, namely, AES, ChlorTox, and AGREE approaches were used to gauge the greener profile of the present methodology (Duan et al., 2020; Pena-Pereira et al., 2020; Nowak et al., 2023). With respect to analytical chemistry, the proposed work will add some knowledge in the field as the combination of ethanol/water has been studied as the green solvent systems for the determination of various drugs/phytochemicals (Alam et al., 2020; Foudah et al., 2020; Foudah et al., 2021; Hameed et al., 2021; Alam et al., 2023; Bang and Bhatt, 2023). However, this work will contribute significantly with respect to pharmaceutical assay of APM as no greener analytical method is reported for the analysis of APM. Accordingly, this study aimed to establish and validate a greener, simple, rapid, and sensitive reversed phase HPTLC (RP-HPTLC) assay for the quantitative analysis of APM in marketed tablet dosage forms and in-house prepared polymeric nanoparticle (NP), and nanoemulsion formulations. The proposed analytical technique was validated in accordance with the “International Council on Harmonization (ICH)” Q2 (R1) recommendations (ICH, 2005).
2 Materials and methods
2.1 Materials
APM was acquired from “Beijing Mesochem Technology Pvt. Ltd. (Beijing, China)”. Poly(lactic-co-glycolic acid) (PLGA; 50:50), poly(vinyl alcohol) (PVA), and Tween-80 were acquired from “Sigma Aldrich (St. Louis, MO, USA)”. Lauroglycol-FCC and Transcutol-HP were procured from “Gattefosse (Lyon, France)”. HPLC-grade ethanol and dichloromethane (DCM) were acquired from “E-Merck (Darmstadt, Germany)”. Deionized water was obtained using a “Milli-Q unit”. Commercial APM tablets (Otezla®, Amgen Inc., CA, USA) were purchased from local market in “Riyadh, Saudi Arabia”. Other materials of high purity and analytical grade were used in this study.
2.2 Chromatography conditions
The HPTLC-based densitometric estimation of APM in its bulk form, commercial tablets, NP, and nanoemulsion formulations was performed utilizing an “HPTLC CAMAG TLC system (CAMAG, Muttenz, Switzerland)”. The quantitation of APM was performed via “10 x 20 cm glass backed plates pre-coated with RP silica gel 60 (F254S plates, E-Merck, Darmstadt, Germany)” at an ambient temperature (22 °C). A “CAMAG Automatic TLC Sampler 4 (ATS4) Sample Applicator (CAMAG, Muttenz, Switzerland)” was used to apply the samples as 6 mm bands. The sample applicator was loaded to a “CAMAG microliter Syringe (Hamilton, Bonaduz, Switzerland)”. The 150 nL/s application rate for the APM quantitation was maintained constant. In a “CAMAG automatic developing chamber 2 (ADC2) (CAMAG, Muttenz, Switzerland)” utilizing ethanol/water (65:35, v/v) solvent system (mobile phase), RP-TLC plates were developed linearly ascending at an 80 mm distance. Earlier, the mobile phase was saturating the developing chamber for 30 min at 22 °C. The absorbance/reflectance mode was used for the RP-HPTLC-densitometric scanning, and the maximum wavelength (λmax) used was 238 nm. Scan rates and slit dimensions were both held constant at 20 mm/s and 4 x 0.45 mm2, respectively. Each quantitative analysis of APM was carried out at least for three times (n = 3) and the baseline was monitored. The software utilized for data analysis and processing was “WinCAT’s (version 1.4.3.6336, CAMAG, Muttenz, Switzerland)”.
2.3 APM calibration plot
A stock solution (SS) of pure APM was prepared by dissolving 10 mg of APM in 10 mL ethanol. Then, 1 mL of the SS was diluted again using the mobile phase to get a final SS with a concentration 100 μg/mL. Different volumes of the SS were taken and diluted to obtain APM concentrations of 100, 200, 300, 400, 500, 600, and 700 ng/band. The same volume (20 µL) of different solutions of APM were applied to RP-TLC plates and the HPTLC response for each concentration of APM was recorded. The calibration plot was constructed for the concentration (100–700 ng/band) of APM vs. the measured HPTLC area.
2.4 Sample processing for the determination of APM in commercial tablet dosage forms
Twenty-five commercial tablets (each containing 5 mg of APM) were weighed and the average weight was recorded. Then, the tablets were crushed and mixed thoroughly to obtain fine powder. A quantity of the powder containing 5 mg of APM was dissolved in ethanol and diluted with mobile phase to make a 50-mL SS. The SS was sonicated for around 10 min after being filtered to remove any insoluble materials. The mobile phase was then used to dilute 1.0 mL of the material above the SS once more. The diluted SS was used for the quantification of APM by the proposed methodology.
2.5 Preparation and characterization of APM-loaded PLGA NPs
A single emulsion and solvent evaporation method were employed to create APM-loaded PLGA NPs (Anwer et al., 2019). Briefly, 150 mg of PLGA were dispensed into the necessary volume of DCM to create the organic phase. The organic phase was then mixed with 10 mg of APM in 5 mL. Dispersing 250 mg of PVA in 10 mL of deionized water produced the aqueous phase. Under probe sonication for around three minutes at 60 % voltage efficiency with a “Probe Sonicator (Ultrasonic Processor, gx-130, Berlin, Germany)” at 25 °C, the organic and aqueous phases were combined into an emulsion. Then, APM-loaded PLGA NPs were separated from the bulk aqueous phase utilizing “High-Speed Centrifugation (Hermle Labort Echnik, Germany)” at 16,000 rpm for 20 min. In order to remove the unadsorbed drug and excessive PVA, the NPs were washed three times using deionized water and then freeze dried. APM-loaded NPs were evaluated physico-chemically for particle size, polydispersity index (PDI), zeta potential (ZP), and entrapment efficiency (EE). The details of the procedures used for the characterization of the PLGA NPs are included in our previous publication (Anwer et al., 2019).
2.6 Sample processing for the quantitation of APM in PLGA NPs
50 mL of SS was created by diluting approximately 25 mg of a PLGA NP formulation (having 5 mg of APM) in ethanol utilizing greener mobile phase. This SS was sonicated for around 10 min after being filtered to remove any insoluble materials. The greener mobile phase was then used to dilute this SS by 1.0 mL once more. The proposed method was used to analyze the APM in PLGA NPs using the diluted SS.
2.7 Preparation and characterization of APM-loaded nanoemulsion
The APM-loaded nanoemulsion formulation was prepared using an aqueous phase titration method, as previously described (Abushal et al., 2022). In a nutshell, 150 µL of the Lauroglycol-FCC (oil phase) were mixed with 5 mg of APM. Then, with continuous votexing, 300 µL of Tween-80 (a surfactant) and 300 µL of Transcutol-HP (a cosurfactant) were introduced to the oil phase containing the APM. The 250 µL of deionized water was then added to the above solution in dropwise manner with continuous shaking till clear and transparent solution obtained. APM-loaded nanoemulsion was evaluated physico-chemically for thermodynamic stability, self-nanoemulsifictaion efficiency, droplet size, PDI, ZP, refractive index (RI), and percentage of transmittence (%T). The details of the procedures used for the characterization of the nanoemulsion are included in our previous publication (Abushal et al., 2022).
2.8 Sample processing for the quantitation of APM in nanoemulsion
A nanoemulsion formulation containing 5 mg of APM was dispersed in the amount of 1.0 mL in ethanol, and 50 mL of SS was created by diluting it with greener mobile phase. This SS was sonicated for around 10 min after being filtered to remove any insoluble materials. The greener mobile phase was then used to dilute this SS by 1.0 mL once more. The proposed method was used to analyze APM in nanoemulsion utilizing the diluted SS.
2.9 Analytical method validation
According to the ICH Q2 (R1) recommendations, the proposed HPTLC-densitometry method for the measurement of APM was validated for several parameters in bulk solution (ICH, 2005). Plotting APM concentrations versus measured densitometric response allowed us to assess the linearity range. In the band range of 100–700 ng, linearity was attained. A standard addition/spiking methodology was used to calculate the accuracy for the greener densitometric assay as a percentage of recovery (ICH, 2005). A pre-determined APM solution of APM (200 ng/spot) was spiked with extra 0–150 % of APM. The final solutions were re-analyzed using the greener densitometric assay. The % recovery of APM was calculated at each concentration level.
The greener densitometric assay's precision was assessed using intra- and interday repeatability. The quantification of APM at concentrations of 200, 300, and 400 ng/band (n = 6) on the same day allowed for the evaluation of intraday fluctuation. The quantification of APM at the same concentrations over the course of three days (n = 6) allowed for the identification of interday fluctuation.
By purposefully making minor changes to the greener mobile phase composition during APM analysis, the robustness for the greener densitometric assay was assessed. For this, the original ethanol/water (65:35, v/v) greener mobile phase was changed to ethanol/water (67:33, v/v) and ethanol/water (63:37, v/v), and the required changes in densitometric response and retardation factor (Rf) values were recorded at each set of conditions.
By using the standard deviation (SD) technique, the sensitivity for the greener densitometric method was evaluated as “limit of detection (LOD) and limit of quantification (LOQ)” (ICH, 2005). The “LOD and LOQ” for APM were derived by equations (1) and (2) (Alam et al., 2020; Foudah et al., 2020):
Where, S is the slope of the calibration curve for APM, and SD is the standard deviation of the intercept.
The specificity for the greener densitometric assay was assessed by contrasting the Rf and UV-absorption spectra of APM in the PLGA NP and nanoemulsion formulations with those of pure APM.
2.10 Greenness assessment
The greenness profile of the existing method for determining APM was evaluated using three separate methods: AES, ChlorTox, and AGREE (Duan et al., 2020; Pena-Pereira et al., 2020; Nowak et al., 2023). AES is a semi-quantitative approach, which considers all steps of analytical procedures, instruments, and waste. Analysis of compounds with no or minimal use of reagents, low energy consumption, and no waste is expected to be an ideal analysis with 100 points. If any of these parameters are deviated, penalty points are assigned, and total penalty points are subtracted from 100 (Duan et al., 2020). The ChlorTox technique states that the ChlorTox score is determined using equation (3) (Nowak et al., 2023):
Where msub is the mass of the substance of interest needed for a single analysis, CHsub is the chemical risks of the substance of interest, CHCHCl3 is the chemical hazard of standard chloroform. With the aid of the safety data sheet from “Sigma Aldrich (St. Louis, MO, USA)”, the values of CHsub and CHCHCl3 were determined using the weighted hazards number (WHN) model (Nowak et al., 2023). AGREE score for the current method for the determination of APM was evaluated using the AGREE-metric technique (Pena-Pereira et al., 2020). The “AGREE: The Analytical Greenness Calculator (version 0.5, Gdansk University of Technology, Gdansk, Poland, 2020)” was used to gauge the AGREE scores in the range from 0.0 to 1.0 for the current procedure.
2.11 Determination of APM in commercial tablet dosage forms, PLGA NP and nanoemulsion formulations
Under the same experimental conditions as those used for the measurement of pure APM, the processed samples of commercial tablet dosage forms, produced PLGA NPs, and nanoemulsion were spotted to the RP-TLC plates. The peak area of APM in produced PLGA NPs, marketed tablet dosage forms, and nanoemulsion was compiled. The content of APM in all dosage forms was determined using APM calibration curve.
2.12 Statistical evaluation
All data are presented as mean ± SD of three or six independent experiments. The value was statistical significance at p = 0.05. The MS Excel (2016) program was used to derive all parameters.
3 Results and discussion
3.1 Preparation and evaluation of APM-loaded PLGA NPs and nanoemulsion
Table 1 lists the components of the APM-loaded PLGA NPs and nanoemulsion and their evaluation criteria. Utilizing a “Malvern Particle Size Analyzer (Malvern Instruments Ltd., Holtsville, NY, USA)”, the particle size, PDI, and ZP of PLGA NPs were assessed. However, the EE was calculated using its standard formula reported previously in literature (Anwer et al., 2019). The particle size and PDI of the PLGA NPs are 307.30 ± 8.50 nm and 0.31, respectively. The ZP is −43.40 ± 2.60 mV. The EE is 61.10 ± 1.90 %. These results demonstrate that the PLGA NP formulation was successfully prepared in the laboratory. The nanoemulsion formulation was found to be thermodynamically stable against centrifugation, heating and cooling cycles, and freeze-pump thaw cycles. In addition, prepared nanoemulsion passed the self-nanoemulsification test with grade A (Abushal et al., 2022). The droplet size and PDI of the nanoemulsion are 17.505 ± 0.247 nm and 0.147 ± 0.014, respectively (Table 1). The ZP is −13.350 ± 0.840 mV. The RI and %T are 1.337 ± 0.001 and 99.15 ± 0.131 %, respectively (Table 1). These results demonstrate that the nanoemulsion formulation of APM was successfully prepared in the laboratory.
Formulation component
Evaluation parameter
PLGA NPs
APM (mg)
10
Particle size ± SD (nm)
307.30 ± 8.50
PLGA (mg)
150
PDI
0.31
PVA (mg)
250
ZP ± SD (mV)
−43.40 ± 2.60
EE ± SD (%)
61.10 ± 1.90
Nanoemulsion
APM (mg)
5
Droplet size ± SD (nm)
17.505 ± 0.247
Lauroglycol-FCC (µL)
150
PDI
0.147 ± 0.014
Tween-80 (µL)
300
ZP ± SD (mV)
−13.350 ± 0.840
Transcutol-HP (µL)
300
RI ± SD
1.337 ± 0.001
Deionized water (µL)
250
T ± SD (%)
99.15 ± 0.131
3.2 Method development
Based on the literature report, there was a necessity to develop a greener densitometric assay for APM analysis. Hence, the aim of this work involves the development and validation of an economical and greener RP-HPTLC technique for the estimation of APM in marketed tablets, PLGA NP, and nanoemulsion formulations. RP-densitometric techniques have a number of benefits over normal-phase HPTLC methods, including the avoidance of non-polar fractions from the sample in the TLC plates, the avoidance of interference from the presence of impurities, the formation of compact spots, and detection clarity (Ahmed et al., 2019; Rezk et al., 2019). The greener mobile phase for RP-densitometric measurement of APM was studied in the proposed experiment using various ethanol and water ratios. The optimized greener mobile phase was developed under chamber saturation circumstances. The ethanol/water (65:35, v/v) mixture, out of all the ethanol and water combinations tested, displayed a well-resolved, symmetrical, and compact chromatogram for APM with Rf = 0.61 ± 0.01 (Fig. 1A). Therefore, this greener combination was used for the determination of APM in our PLGA NP formulation and marketed formulation. The spectral bands were assessed densitometrically, and the maximum densitometric response was found at λmax = 238 nm. Accordingly, all subsequent analyses were carried out at 238 nm.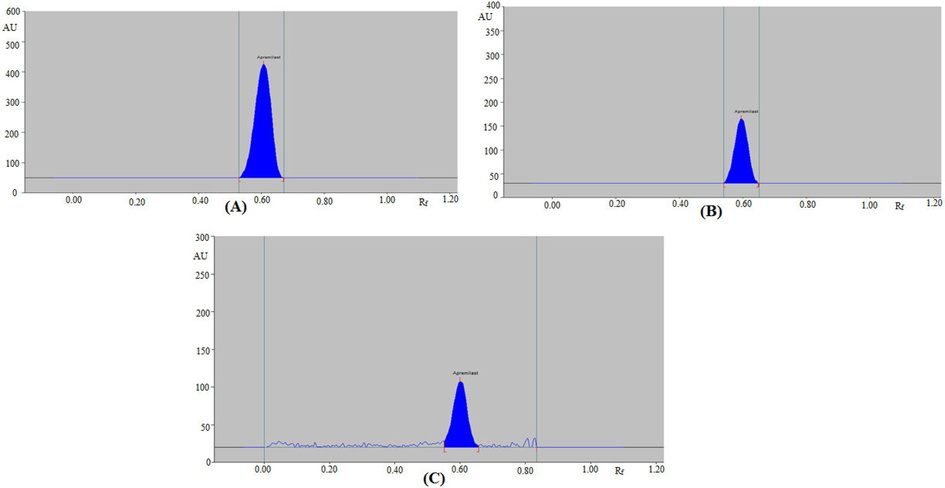
High-performance thin layer chromatography (HPTLC)-densitograms of (A) standard apremilast (APM), (B) prepared NPs, and (C) prepared nanoemulsion.
3.3 Method validation
All the validation parameters of the proposed assay were validated as per ICH recommendations (ICH, 2005). The APM calibration curve is linear in the concentration range of 100–700 ng/band. The concentration and measured HPTLC area have a good linear relationship as a result (Table 2). The APM's determination coefficient (R2) is 0.9995, which has a highly significant level of significance (p < 0.05). The values of slope and intercept of the calibration curve depend on their x-axis and y-axis values, respectively. In this study, the recorded HPTLC area of APM (y-axis) were much higher than its studied concentrations (x-axis) and hence intercept value of APM calibration curve was much higher than its slope value. The APM linearity range for two HPTLC methods has been reported as 100–600 ng/band and 250–1500 ng/band, respectively (Chaudhari and Shirkhedkar, 2017; Bhole et al., 2019). The APM linearity range for the present methodology was better than reported methods (Chaudhari and Shirkhedkar, 2017; Bhole et al., 2019).
Parameters
Values
Linearity range (ng/band)
100–700
Regression equation
y = 30.94x − 2390.90
Determination coefficient
0.9995
Slope ± SD
30.94 ± 1.45
Intercept ± SD
2390.90 ± 38.43
Standard error of slope
0.59
Standard error of intercept
15.68
95 % confidence interval of slope
28.39–33.48
95 % confidence interval of intercept
2323.39–2458.41
LOD ± SD (ng/band)
4.09 ± 0.14
LOQ ± SD (ng/band)
12.29 ± 0.42
The accuracy of the method was determined in terms of % recovery. The APM % recoveries were recorded in the range of 98.74–100.62 %. The APM % RSDs were recorded in the range of 1.03–1.54 %. The % recovery are within 100 ± 2 % and the % RSD values are ± 2 %, indicating that the proposed technique is highly accurate for the determination of APM. The % recovery for two HPTLC methods has been reported as 99.44–99.86 and 100.12–101.83 %, respectively (Chaudhari and Shirkhedkar, 2017; Bhole et al., 2019). The % recovery for the present methodology was similar to the reported methods (Chaudhari and Shirkhedkar, 2017; Bhole et al., 2019).
The method's precision was calculated as a % RSD (Table 3). The recorded values of % RSD are within ± 2 %, demonstrating that the proposed technique is highly precise for the estimation of APM. The % RSD for two HPTLC methods has been reported as 0.28–0.60 and 0.19–0.85 %, respectively (Chaudhari and Shirkhedkar, 2017; Bhole et al., 2019). The % RSD for the present methodology was also similar to the reported methods (Chaudhari and Shirkhedkar, 2017; Bhole et al., 2019).
Conc. (ng/band)
Intra-day precision
Inter-day precision
Area ± SD
Standard error
RSD (%)
Area ± SD
Standard error
RSD (%)
200
3695.51 ± 38.92
15.89
1.05
3545.42 ± 40.45
16.51
1.14
300
6971.81 ± 91.61
37.40
1.31
7089.14 ± 98.65
40.28
1.39
400
9986.28 ± 178.29
72.80
1.78
9789.67 ± 180.67
73.77
1.84
The variation (as % RSD) upon introducing small deliberate changes in the mobile phase composition is 1.19–1.33 %. The Rf value for robustness evaluation was found to be 0.61–0.63. The small variation in Rf values and the lower % RSD value show that the proposed technique is robust.
The “LOD and LOQ” of the proposed methodology are 4.09 ± 0.14 and 12.29 ± 0.42 ng/band, respectively (Table 2). These data show that the suggested method is sensitive enough to detect and quantify APM over a wide range of concentration levels.
The specificity and peak purity of the method for APM were assessed by comparing the overlaid spectra of pure APM and APM in PLGA NPs and nanoemulsion. Fig. 2 displays the superimposed spectra of pure APM and APM in laboratory developed PLGA NPs and nanoemulsion. The peak purity of standard APM and APM in NPs and nanoemulsion was assessed by comparing the spectra at the peak start (S), peak apex (M), and peak end (E) positions of the spot (El-Kimary et al., 2014; El-Kimary and Ragab, 2015). The computed values of r (S,M) and r (M,E) of standard APM, NPs, and nanoemulsion were greater than 0.99, indicating the peaks’ homogeneity (El-Kimary et al., 2014, 2018). APM in its pure form and as a PLGA NPs, and nanoemulsion showed the highest RP-HPTLC-densitometric response at λmax = 238 nm. Similar superimposed spectra, Rf data, and λmax of APM in produced PLGA NPs, nanoemulsion, and pure APM demonstrated the green RP-HPTLC method's peak purity and specificity.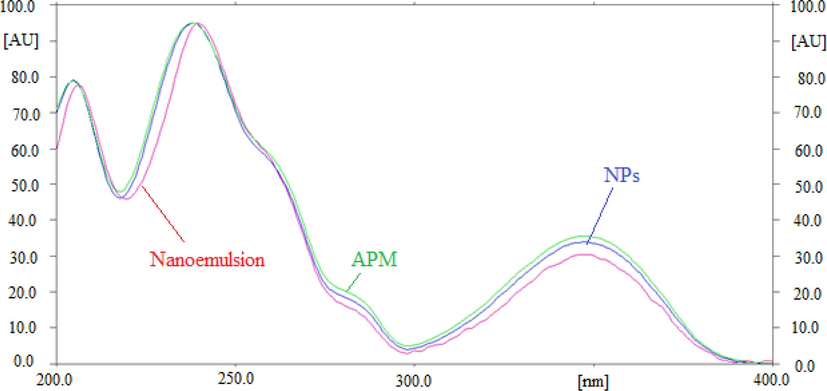
UV-absorption spectrum of pure APM, prepared nanoparticles (NPs), and nanoemulsion, superimposed.
3.4 Greenness assessment
Numerous quantitative and qualitative approaches are established for the greenness assessment of pharmaceutical assays (Keith et al., 2005; Plotka-Wasylka, 2018; Nowak and Koscielniak, 2019; Duan et al., 2020; Pena-Pereira et al., 2020; Nowak et al., 2021; Nowak et al., 2023). In the present work, three different approaches, namely AES, ChlorTox, and AGREE approaches were used to assess the greenness of the current approach (Duan et al., 2020; Pena-Pereira et al., 2020; Nowak et al., 2023).
AES is a good semi-quantitative approach, which considers all the steps of the analytical procedures, instruments, and waste. The results of AES scores with penalty points for the present method in comparison to reported HPTLC methods are included in Table 4. An excellent greenness was indicated by an AES value greater than 75, an adequate greenness by an AES value less than 75 but greater than 50, and an inadequate greenness by an AES value less than 50 (Duan et al., 2020). The AES score of the present method was derived to be 93, indicated an excellent greenness profile. The AES scores for the literature HPTLC methods were recorded as 81 and 73, respectively (Chaudhari and Shirkhedkar, 2017; Bhole et al., 2019). The AES results indicated that the present method was much greener than reported HPTLC methods (Chaudhari and Shirkhedkar, 2017; Bhole et al., 2019).
Reagents/instruments/waste
Penalty points
HPTLC (Chaudhari and Shirkhedkar, 2017)
HPTLC (Bhole et al., 2019)
Present method
Ethanol
4
Water
0
Toluene
12
12
Ethyl acetate
4
Methanol
12
Ethyl acetate
Instruments
0
0
0
Waste
3
3
3
Total penalty points
19
27
7
AES score
81
73
93
Table 5 includes the findings of the individual greener solvent ChlorTox scores and the overall ChlorTox for the suggested technique in comparison to reported HPTLC methods. The total ChlorTox value for the suggested approach was anticipated to be 0.66 g, indicating that it was both safer and environmentally friendly (Nowak et al., 2023). The total ChlorTox values for literature HPTLC methods were anticipated to be 2.20 and 2.71 g, respectively, indicating that it was both unsafe and less green (Chaudhari and Shirkhedkar, 2017; Bhole et al., 2019). The ChlorTox results indicated that the present method was much safer and greener than reported HPTLC methods (Chaudhari and Shirkhedkar, 2017; Bhole et al., 2019).
Stage
Solvent/reagent
Relative hazard (CHsub/CHCHCl3)
msub (mg)
ChlorTox (g)
Total ChlorTox (g)
Reference
Sample preparation
Ethanol
0.26
1300
0.33
HPTLC analysis
Ethanol
0.26
1300
0.33
0.66
Present method
Sample preparation
Methanol
0.56
2000
1.12
HPTLC analysis
Toluene
0.86
800
0.68
Ethyl acetate
0.34
1200
0.40
2.20
(Chaudhari and Shirkhedkar, 2017)
Sample preparation
Methanol
0.56
2000
1.12
HPTLC analysis
Toluene
0.86
1600
1.37
Methanol
0.56
400
0.22
2.71
(Bhole et al., 2019)
The AGREE approach is the most widely used quantitative approach for greenness assessment as it consumes all 12 GAC principles (Pena-Pereira et al., 2020). The AGREE score of greater than 0.75 indicated an excellent greenness, the AGREE score of less than 0.75 but greater than 0.50 indicated the adequate greenness, and AGREE score of less than 0.50 indicated inadequate greenness (Pena-Pereira et al., 2020). The recorded overall AGREE scale utilizing 12 components/principles of GAC is summarized in Fig. 3. The overall AGREE scale for the proposed HPTLC method was recorded as 0.89. The AGREE results again demonstrated the current method's excellent green features. Overall, the results of all greenness approaches indicated the excellent greener profile of the current method for the determination of APM in nanoformulations and commercially available tablets compared to literature HPTLC methods (Chaudhari and Shirkhedkar, 2017; Bhole et al., 2019).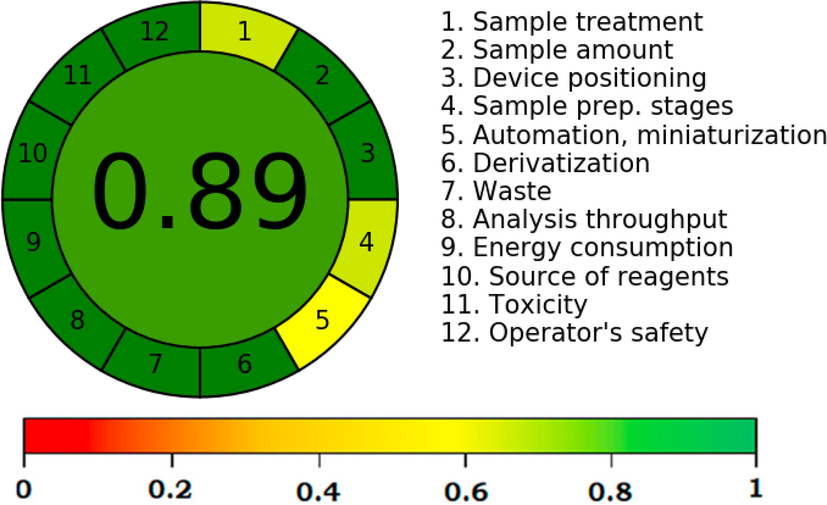
Analytical GREEnness (AGREE) scale for the greener HPTLC assay of APM.
3.5 Quantitative analysis of APM in marketed tablets, PLGA NP, and nanoemulsion formulations
The HPTLC peaks for APM in commercial tablets, PLGA NP, and nanoemulsion formulations was verified by comparing its single TLC spectra at Rf = 0.61 ± 0.01 with that of pure APM. The HPTLC densitograms for APM in our PLGA NPs and nanoemulsion are shown in Fig. 1B and 1C, respectively. The densitograms of APM NPs and nanoemulsion are similar to that of the pure APM. The 3D tracks of the pure APM and different formulations are presented in Fig. 4, showing that λmax is the same for all the samples investigated. The APM content of the marketed tablets, PLGA NPs, and nanoemulsion were estimated from the calibration curve of APM. The APM content of the commercial tablets was calculated to be 4.92 ± 0.57 mg for 5 mg of APM. However, the APM content of the in-house developed PLGA NPs was found to be 5.08 ± 0.76 mg for 5 mg of APM. On the other hand, the APM content of the in-house developed nanoemulsion was found to be 4.96 ± 0.62 mg for 5 mg of APM. Thus, the % contents of APM in the marketed tablets, PLGA NPs, and nanoemulsion are 98.40 %, 101.60 %, and 99.37 %, respectively. The mean % APM recovery in in-house developed tablet formulations has been reported as 99.13 ± 0.75 % (Chaudhari and Shirkhedkar, 2017). The mean % APM recovery in tablet formulations was recorded as 99.70 ± 0.23 % by another report (Bhole et al., 2019). The results of the proposed method for the determination of APM in marketed tablets were compared with reported HPTLC methods using the Student’s t-test and the variance ratio F-test (Chaudhari and Shirkhedkar, 2017; Bhole et al., 2019). The recorded t and F values did not exceed their theoretical values, indicating no significance differences in the performance of compared method in terms of precision and accuracy. The % contents of APM are within ± 2 %, demonstrating that our proposed greener RP-HPTLC technique can be successfully utilized for the quantitative analysis of APM in pharmaceutical dosage forms containing APM as one of the constituents.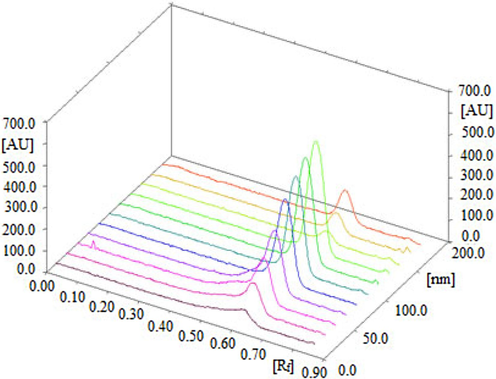
3D view of standard APM (first eight tracks), marketed tablets (track 9), NPs (track 10), and nanoemulsion (track 11).
4 Conclusion
A greener RP-HPTLC technique for the quantitative analysis of APM in its bulk form, marketed tablets, PLGA NP, and nanoemulsion formulations was developed and validated. The PLGA NP formulation was prepared by adopting a “single emulsion and solvent evaporation method” and characterized physico-chemically. The APM-loaded nanoemulsion was prepared using an aqueous phase titration approach and characterized physico-chemically. The proposed analysis technique was found to be simple, accurate, precise, robust, sensitive, selective, and greener for the estimation of APM. The results of AES, ChlorTox, and AGREE assessment showed an excellent greenness characteristic of the current method in comparison to reported HPTLC methods for the determination of APM. Furthermore, it was successfully applied for the quantitative analysis of APM in marketed tablets, PLGA NP, and nanoemulsion formulations. This technique is the first validated method utilizing RP-18 silica gel for the estimation of APM in in-house developed NP and nanoemulsion formulations. Overall, the method has been found more linear, safe, and greener than reported HPTLC methods. The proposed technique may be applied to the quantitative analysis of APM in different pharmaceutical dosage forms having APM as one of the constituents.
Acknowledgments
“Authors are thankful to the Researchers Supporting Project number (RSPD2024R1040), King Saud University, Riyadh, Saudi Arabia for supporting this work”. “The authors also thank Prince Sattam bin Abdulaziz University for supporting this work via project number (PSAU/2023/R/1444)”. “The authors also thank AlMaarefa University for their generous support”.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Self-nanoemulsifying drug delivery system (SNEDDS) of apremilast: In vitro evaluation and pharmacokinetics studies. Molecules. 2022;27:E3085.
- [Google Scholar]
- Antidiabetic, antixidant, molecular docking and HPTLC analysis of miquelianin isolated from Euphorbia schimperi C. Presl. Saudi Pharm. J.. 2019;27:655-663.
- [Google Scholar]
- Ecofriendly densitometric RP-HPTLC method for determination of rivaroxaban in nanoparticle formulations using green solvents. RSC Adv.. 2020;10:2133-2140.
- [Google Scholar]
- Development and validation of a stability-indicating greener HPTLC method for the estimation of flufenamic acid. Separations. 2023;10:E39.
- [Google Scholar]
- Stability-indicating related substances method of apremilast by HPLC and synthesis and chracterization of related impurities using mass and NMR spectroscopy. Int. J. Curr. Med. Pharm. Res.. 2017;3:1378-1385.
- [Google Scholar]
- Preparation of sustained release apremilast-loaded PLGA nanoparticles: in vitro characterization and in vivo pharmacokinetic study in rats. Int. J. Nanomed.. 2019;14:1587-1595.
- [Google Scholar]
- Development and validation of ultraviolet-visible spectroscopic method for the estimation of assay of sugammadex sodium, apremilast, riociguat, and vorapaxar sulfate drugs in active pharmaceutical ingredient form. Asian J. Pharm. Clin. Res.. 2017;10:241-250.
- [Google Scholar]
- Development of green RP- and green NP-HPTLC methods for estimation of lenvatinib and comparative evaluation by AGREE. ACS Sus. Chem. Eng.. 2023;11:2249-2263.
- [Google Scholar]
- A stability indicating HPTLC method for apremilast and identification of degradation products using MS/MS. J. Pharm. Sci. Res.. 2019;11:1861-1869.
- [Google Scholar]
- Box-Behnken design for validation of high-performance thin-layer chromatography/densitometry method for robustness determination of apremilast in bulk and in house tablets. Pharm. Methods. 2017;9:9-15.
- [Google Scholar]
- Design of experiment avenue for development and validation of RP-HPLC-PDA method for determination of apremilast in bulk and in in-house tablet formulation. J. Anal. Sci. Technol.. 2019;10:E10.
- [Google Scholar]
- Determination of apremilast in rat plasma by UPLC-MS-MS and its application to a pharmacokinetic study. J. Chromatogr. Sci.. 2016;54:1336-1340.
- [Google Scholar]
- Efficacy and safety of apremilast for Behcet’s syndrome: a real-life single-centre Italian experience. Rheumatol.. 2019;59:171-175.
- [Google Scholar]
- A green HPLC method for determination of nine sulfonamides in milk and beef, and its greenness assessment with analytical eco-scale and greenness profile. J. AOAC Int.. 2020;103:1181-1189.
- [Google Scholar]
- A validated high-performance thin-layer chromatographic method for the assay of two binary mixtures containing omeprazole or its isomer esomeprazole in pharmaceutical dosage forms. J. Planar Chromatogr.. 2015;28:74-82.
- [Google Scholar]
- High-performance thin-layer chromatographic assay of metformin in urine using ion-pair solid-phase extraction: Application for bioavailability and bioequivalence study of new microbeads controlled release formulation. J. Planar Chromatogr.. 2014;27:377-384.
- [Google Scholar]
- Novel validated HPTLC method for the analysis of two binary mixtures containing tamsulosin hydrochloride with antimuscarinic agents. J. Chromatogr. Sci.. 2018;56:81-91.
- [Google Scholar]
- EMA, 2014. Assessment report on Otezla (apremilast), CHMP, EMA.
- FDA, 2014. US Department of Health and Human Services, Food and Drug Administration Centre for Drug Evaluation and Research (CDER), Guidance for Industry, Clinical Pharmacology and Biopharmaceutics review, March 2014.
- Validated LC method for determination of enantiomeric purity of apremilast using polysaccharide-type stationary phases in polar organic mode. Chromatographia. 2018;81:1613-1621.
- [Google Scholar]
- A green RP-HPTLC-densitometry method for the determination of diosmin in pharmaceutical formulations. Processes. 2020;8:E817.
- [Google Scholar]
- A sustainable reversed-phase HPTLC method for the quantitative estimation of hesperidin in traditional and ultrasound-assisted extracts of different varieties of citrus fruit peels and commercial tablets. Agronomy. 2021;11:E1744.
- [Google Scholar]
- A novel green HPTLC method for simultaneous analysis of four antipsychotics in their pharmaceutical formulations: Assessment by eco-scale. Rec. Pharm. Biomed. Sci.. 2021;5:E16.
- [Google Scholar]
- Green RP-HPLC method for simulatneous determination of moxifloxacin combinations: investigation of the greenness for the proposed method. Microchem. J.. 2019;148:151-161.
- [Google Scholar]
- International conference on harmonization (ICH), Q2 (R1): validation of analytical procedures–text and methodology, Geneva, Switzerland, 2005.
- Development and validation of sophisticated analytical method for the estimation of apremilast. Pharm. Sci. Monit.. 2017;8:267-276.
- [Google Scholar]
- Determination of apremilast in rat plasma by UPLC-MS/MS in ESI-negative mode to avoid adduct ions formation. Bioanalysis. 2016;8:1499-1508.
- [Google Scholar]
- An introduction to the national environmental methods index. Env. Sci. Technol.. 2005;39:173A-176A.
- [Google Scholar]
- Analytical methods for determination of apremilast from bulk, dosage form and biological fluids: a critical review. Crit. Rev. Anal. Chem.. 2021;51:258-267.
- [Google Scholar]
- Development and validation of stability indicating rapid RP-LC method for determination of process and degradation related impurities of apremilast, an anti-inflammatory drug. Am. J. Anal. Chem.. 2017;8:380-394.
- [Google Scholar]
- Identification and characterization of process-related substances and degradation products in apremilast: process optimization and degradation pathway elucidation. J. Pharm. Biomed. Anal.. 2017;141:70-78.
- [Google Scholar]
- Preparation, characterization and in vitro evaluation of tablets containing microwave-assisted solid dispersions of apremilast. Polym. Med.. 2019;48:17-24.
- [Google Scholar]
- Off-label studies on apremilast in dermatology: a review. J. Dermatol. Treat.. 2020;31:131-140.
- [Google Scholar]
- A new stability indicating RP-HPLC method for the determination of apremilast-an antirheumatic drug. Res. J. Pharm. Technol.. 2017;10:1160-1164.
- [Google Scholar]
- What color is your method? Adaptation of the RGB additive color model to analytical method evaluation. Anal. Chem.. 2019;91:10343-10352.
- [Google Scholar]
- White analytical chemistry: An approach to reconcile the principles of green analytical chemistry and functionality. Trends Anal. Chem.. 2021;138:E116223.
- [Google Scholar]
- How to evaluate methods used in chemical laboratories in terms of the total chemical risk? a ChlorTox Scale. Green Anal. Chem.. 2023;5:E100056.
- [Google Scholar]
- Hydrophilic multitemplate molecularly imprinted biopolymers based on a green synthesis strategy for determination of B family vitamins. ACS Appl. Mater. Interf.. 2018;10:4140-4150.
- [Google Scholar]
- AGREE-Analytical GREEnness metric approach and software. Anal. Chem.. 2020;92:10076-10082.
- [Google Scholar]
- A new tool for the evaluation of the analytical procedure: Green analytical procedure index. Talanta. 2018;181:204-209.
- [Google Scholar]
- Development and validation of stability-indicating UV spectrophotometric method for determination of apremilast in bulk and pharmaceutical dosage form. Indian J. Res. Pharm. Biotechnol.. 2017;5:47-53.
- [Google Scholar]
- Novel determination of a new antiviral combination; sofosbuvir and velpatasvir by high performance thin layer chromatographic method; application to real human samples. Microchem. J.. 2019;146:828-834.
- [Google Scholar]
- Apremilast is a selective PDE4 inhibitor with regulatory effects on innate immunity. Cell Signal. 2014;26:2016-2029.
- [Google Scholar]
- Solubility and thermodynamics of apremilast in different mono solvents: determination, correlation and molecular interactions. Int. J. Pharm.. 2017;523:410-417.
- [Google Scholar]
- Solubility and thermodynamic function of apremilast in different (Transcutol + water) cosolvent mixtures: measurement, correlation and molecular interactions. J. Ind. Eng. Chem.. 2017;56:99-107.
- [Google Scholar]
- Chromatographic method development and validation of assay of apremilast in bulk and tablet dosage form. Eur. J. Biomed. Pharm. Sci.. 2018;3:412-417.
- [Google Scholar]
- Souto, A., Gomez-Reino, J.J., 2017. Apremilast for the treatment of psoriatic arthritis. Expert Rev. Clin. Immunol. 11, 1291-1290.
- Novel HPTLC densitometric methods for determination of tamsulosin HCl and tadalafil in their newly formulated dosage form: Comparative study and green profile assessment. Biomed. Chromatogr.. 2020;34:E4850.
- [Google Scholar]
- Stability-indicating HPTLC method for the simultaneous detection and quantification of alfuzosin hydrochloride, solifenacin succinate along with four of their official impurities. Microchem. J.. 2020;157:E104905.
- [Google Scholar]
- A novel smartphone HPTLC assaying platform versus traditional densitometric method for simultaneous quantification of alfuzosin and solifenacin in their dosage forms as well as monitoring content uniformity and drug residues on the manufacturing equipment. RSC Adv.. 2023;13:11642-116651.
- [Google Scholar]
- Sustainable and smart HPTLC determination of silodosin and solifenacin using a constructed two illumination source chamber with a smartphone camera as a detector: Comparative study with conventional densitometric scanner. Sus. Chem. Pharm.. 2023;33:E101095.
- [Google Scholar]
- Identification, characterization and HPLC quantification of impurities in apremilast. Anal. Methods. 2016;8:1889-1897.
- [Google Scholar]







