Translate this page into:
Advances and challenges of MOF derived carbon-based electrocatalysts and photocatalyst for water splitting: A review
⁎Corresponding author. zj517@mail.tsinghua.edu.cn (Naseem Iqbal) naseem@uspcase.nust.edu.pk (Naseem Iqbal)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Environmental pollution and energy shortage are substantial fears to the modern world's long-term sustainability. Water splitting is an essential technique for eco - friendly and sustainable energy storage, as well as a pollution-free method to produce hydrogen. In this regards Metal–organic frameworks have emerged as the most competent multifunctional materials in recent times, due to its large surface areas, adjustable permeability, easy compositional alteration, and capability for usage as precursors with a wide range of morphological forms. Further, MOF-derived carbon-based nanomaterials also offer significant benefits in terms of tunable morphological features and hierarchical permeability, as well as ease of functionalization, making them extremely effective as catalysts or catalysts supports for a wide variety of important reactions. Recent developments in carbon-based MOFs as catalysts for overall water splitting are discussed in this review. We explore how MOFs and carbon-based MOFs might well be beneficial, as well as which methods should be explored for future development. We divided our review into two sections: photocatalytic and electrocatalytic water splitting, and we gathered published literature on carbon-based MOFs materials for their outstanding activity, offers helpful methods for catalysts design and analysis, as well as difficulties This study highlights the developments in MOF derived materials as photo and electro catalysts by explaining respective approaches for their use in overall water splitting.
Keywords
Hydrogen evaluation reaction
Oxygen evaluation reaction
Metal organic framework
Derivatives
Water splitting
1 Introduction
Continuous economic expansion and an ever-growing world population at a pace of approximately 1.05 % annually, demand a consistent rise in energy consumption (Ager and Lapkin, 2018). The present energy sector's reliance on fossil fuels, oil, and natural gas puts our long-term development at risk due to finite reserves. Furthermore, the detrimental impact of fossil fuel consumption has been adequately shown by its disastrous impact on our planet's environment, climate, and overall health (Ahmad et al., 2015). Hence, conventional fossil fuel reserves depletion and the environmental hazards associated with their use, have necessitated the development of clean, renewable and sustainable energy sources as an alternate energy source (Xu et al., 2018; Zhang and Guan, 2021).
Sustainable energy sources often generate energy sporadically, demanding the storage of energy for future use (Mousazade et al., 2020; Wen et al., 2020). Therefore, much research has been directed toward the use of hydrogen (H2) as an environmentally preferable energy carrier in a post-fossil fuel era. At the moment, it is widely accepted that H2 may be the optimal solution for addressing the triple threat of fatigue, pollution, and climate change consequences (Acar and Dincer, 2015). It is possible to produce hydrogen from water by dividing it into its constituent components. A variety of energy sources can be used to split an H-O-H bond, including electricity, heat, and light. Electrolysis, thermolysis, and photolysis are the most common types of energy sources used in water splitting operations, with electrolysis and thermolysis being the most common (Rizvi et al., 2020). Overall water splitting is a chemical process that converts water to oxygen and hydrogen:
Here water is split into gaseous oxygen, together with protons and electrons, at the anode. The protons and electrons recombine to form hydrogen gas at the cathode (Yaqoob et al., 2021). By far, the energy conversion technology has piqued the interest of many people owing to its high conversion efficiency, low environmental impact, and potentially broad variety of applications (Li and Li, 2017). Electrocatalytic and photocatalytic hydrogen generation using water have already been examined as potential approaches for storing renewable energy in the form of chemical bonds to overcome the intermittent character of renewable energy sources such as solar irradiation (Liu et al., 2021; Anantharaj et al., 2016; Chen et al., 2022).
One of the strategies for producing hydrogen is photocatalytic water splitting as illustrated in Fig. 1, which utilizes photonic energy, the most abundant source of energy on the planet. According to previous studies, solar-based H2 production via photocatalysis produces almost no global warming or air pollution and is stored safely (Dubey et al., 2014). As a result, H2 is being evaluated as a potential significant energy source in the future, as it is toxic-free and can generate significant amounts of energy from natural sources such as light energy and water, which are both clean, long-lasting, and renewable sources of energy (Tee et al., 2017).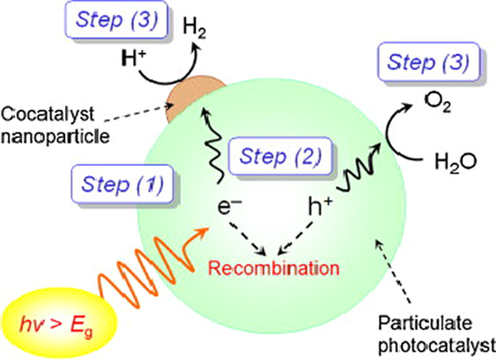
Schematic representation of steps involve in water splitting via photocatalyst (Maeda, 2011). Exclusive rights 2011 Elsevier.
Another strategy of hydrogen production is electrocatalytic water splitting, has been proposed as a viable solution to the renewable energy intermittency problem as an electrical current is used to split water electrolytically, and the electrical energy is converted to chemical energy at the electrode-solution junction via charge transfer processes in a device known an electrolyze. At the anode, water interacts to create oxygen and protons, however at the cathode, hydrogen is evolved (Yaqoob et al., 2020; Mei et al., 2021). Besides, it is estimated that electrolysis can provide just 3.9 % of the world's hydrogen consumption (Matheu et al., 2019). In comparison to conventional SMR, electrolytic water splitting is described as having “zero” CO2 emissions (since oxygen will be the only by-product) (Hunter et al., 2016). However, as electrolysis are usually electrically powered, which would be primarily produced through the ignition of coal and natural gas and carbon dioxide is released as a by-product as an outcome. The electrochemical/catalytic water-splitting reaction is now being driven by sustainable harvesting technologies (such as wind generators or photovoltaic cells), and this is progressively becoming the focus of study in the current era. Using photochemical or photocatalytic water splitting to produce hydrogen is a potential alternative for hydrogen generation that is aimed toward reducing CO2-emissions and making use of sustainable resources like water and sunshine (Albonetti et al., 2019; Wang et al., 2020).
On top of that, water electrolysis has a significant history and is often considered to become the most appealing and convenient way to generate hydrogen and oxygen. At the moment, the majority of commercial uses for water splitting catalysts are limited due to their high cost and scarcity of earth-scarce metals. There have thus been considerable efforts to substitute these precious metals and utilize more plentiful, robust, active, and cost-effective transition electrocatalyst (Walter et al., 2010). Recent advances in developing materials for water splitting. Long-term operating stability is a significant concern and materials are therefore needed, which play a protective function and characteristically stable in acute conditions (acid, base, lighting, heat) (Buriak et al., 2018). For electrocatalytic water splitting the development of efficient electrocatalysts that may efficiently accelerate slow kinetic processes and allow for both water reduction and oxidation at low overpotentials is crucial to ongoing research efforts (Hanif et al., 2019).
In this regards, a novel class of porous materials known as metal organic frameworks stand out as a promising water splitting candidate (Voitic and Hacker, 2016) as they are distinguished by a high degree of crystallinity, porosity, and pore size that significantly exceeds that of other porous materials, and the development of MOF as a catalyst has piqued the interest of researchers over the last decade (Yaqoob et al., 2019; Noor et al., 2019). As MOFs will draw a lot of attention in the future for catalytic applications like water splitting owing to their wide variety of inorganic and organic constituents and following are some of the most common benefits of MOFs and MOFs based composites for using as catalyst: (i) All of the building units, such as metal nodes, organic ligands, and channels, could be modified, resulting in a variety of frameworks, morphologies, sizes, and functional areas; (ii) organic ligands and metal nodes, including both are employed as catalytic centers and light-harvesting centers, to increase active surface area and shorten the photo-generated charges transmission distance; (iii) The porosity of the structure allows encapsulation of additional functional elements in order to create a novel composites and achieve synergetic catalysis; (iv) nanopores may provide the substratum with a unique chemical environment that is able, like the secondary structure of the active site, to interact with the catalytic active site and further enhances its activities and selectivity; (v) The simulation at molecular level of the natural photosynthesis system and single-site catalysts loading can be carried out over certain control of the contact between metal and ligand; (vi) MOFs to create particular functional metal complexes might be utilized as a precursor to get new structures which cannot be achieved using conventional techniques.
Despite considerable success in using MOF-based composites as electro /photocatalysts, additional effort is needed to address the subsequent challenges. such as, MOFs' quantum efficiency of hydrogen generation is poor, demanding further research into the complex chemistry of MOFs for photocatalytic and electrocatalytic applications.
The moisture sensitivity and excellent stability in aqueous environments over a wide pH range of some MOFs has long been regarded as a crucial issue that restricts their applicability (Noor et al., 2019; Noor et al., 2021; Yaqoob et al., 2021). Additionally, the inclusion of noble metals, which are scarce and costly and are not economically desired for large-scale use, improved the efficacy of most reported MOF-based catalysts. Furthermore, certain MOFs' organic linkers are complicated and costly, posing a challenge for their use. Therefore, development of mixed organic linkers, exploration of mixed-organic linkers and transition metal-based catalysts and use of carbon-based co-catalysts including graphene may be an interesting approach in the future to minimize the cost of MOF-based catalysts (Wen et al., 2020; Huang et al., 2022). Further as photocatalyst, several MOF have a limited light harvesting capability by nature, which is a major hindrance to high photocatalytic performance (Gao et al., 2022). MOF materials' soft structures can collapse during the photo water splitting reaction, prompting concerns about their stability. Furthermore, the charge carrier separation and transfer are limited by the intrinsic weak conductivity of MOF material. Therefore, subsequent techniques can be considered in order to address these issues. To begin, more conductive MOF materials must be explored and developed in order to increase electron–hole separation and transmission in a photocatalysts system. For constructing MOF-based solar-driven water splitting, a thorough knowledge of charge transport across the interfaces of MOF based composites is essential. Further, in situ spectroscopic and structural studies of the reaction intermediates, charge transfer, and products, as well as advanced characterization techniques, can give significant information. Additionally, to explain the unclear water splitting process on MOF-based materials, it is required to integrate experimental and theoretical techniques (Li et al., 2021).
Despite these problems, catalytic water splitting over MOFs-based catalysts has a promising future due to its distinct attributes such as large surface area, tunable pore volume, size, 3D structure, and rich coordinating chemistry (Nguyen, 2021) and the synergistic impact of MOFs framework and porous characteristics has provided a new set of possibilities for these MOFs in a wide variety of zones, notably in catalysis (Zhang et al., 2020; Sarwar et al., 2018). The outer coordinating sphere impacts of MOFs-based catalyst, such as hydrogen bonding and proton transfer transmission, must be noted. This tends to imply that MOF catalytic sites may be approached by designing of its framework through integrating different active ligands and metal components, and therefore can exhibit extremely amazing synergic effect, resulting in significantly enhanced catalytic performance (Ipadeola and Ozoemena, 2020; Noor et al., 2020).
Furthermore, carbon materials have been proven to be active materials for OER and HER throughout the last decade. Carbon compounds as an effective material and will also act as catalyst supports have both been actively researched in recent years for overall water splitting, with benefits in both structural characteristics and intrinsic activity (Liu et al., 2021) and can be employed as multifunctional supports to promote charge and mass transport when combined with a metal organic framework. Likewise, carbon products derived from MOFs have superior features such as a large surface area, a variable pore size, high porosity, high conductivity, and stability (Yang et al., 2021). Further, graphene sheets have been demonstrated as a component in the fabrication of new heterojunctions with various MOFs for water splitting. The graphene oxides (GO), reduced graphene oxide (rGO) were chosen for their textural and electrical features.
In the design and synthesis of cost-effective high-activity electro/photocatalysts for the water splitting system, MOFs are commonly used as templates to fabricate variety of carbon based catalysts such as carbon based materials composited with metal, metal oxides and metal carbides. Chen et al. prepared Co@N-CNTs@rGO by a new and efficient method for producing ultrafine Co nanoparticles enclosed in nitrogen-doped carbon nanotubes (N-CNTs) mounted onto rGO. Pyrolysis of coreshell ZIF-67@ZIF-8 evenly attached on graphene oxide yielded hybrid composites with 3D hierarchical structure (GO). Organic ligands derived from core-shell ZIF-67@ZIF-8 crystals were gradually changed into N-doped carbon nanotubes during carbonization due to the catalytic activity of the Co species, resulting in tightly linked Co cores and NCNTs shells. Simultaneously, the graphene oxide was thermally reduced to rGO. It revealed that integrating Co nanoparticles, N-CNTs, and graphene sheets into a 3D hierarchical design resulted in highly exposed active sites and increased diffusion kinetics and mass transfer, owing to the catalyst's synergistic interactions, large interfacial area, and plentiful porosity (Chen et al., 2018).
Above and beyond, the shortage of pure water, hydrogen generation from saltwater electrolysis is becoming more appealing. If saltwater is directly electrolyzed rather than pure water, it may not only store clean energy but also create fresh drinking water, which is very useful in desert regions. According to statistics, seawater comprises for>97 % of earth's water resources over 1.33 billion cubic kilometers. Further, the effective implementation of seawater splitting will be extremely beneficial to the growth of the sustainable society (Mohammed-Ibrahim and Moussab, 2020). Additionally, given the availability of saltwater on the planet, using actual seawater for the creation of hydrogen to generate energy will effectively tackle the problems of fossil energy scarcity and pollution. Because of the numerous advantages of seawater splitting, more and more attention is being paid to seawater electrolysis research (Wang et al., 2021) and much improvement has already been achieved in seawater electrolysis research. The development of suitable electrode catalysts with selective and constant electrocatalytic efficiency in seawater is a critical step in the implementation of seawater electrolysis. Electrocatalysts with inherent corrosion resistance or selective surface chemistry are being suggested as promising solutions for improving the long-term stability of electrocatalysts in seawater (Huang et al., 2019). However, high overpotential, poor stability and low activity remain issues with the developed electrocatalyst. Furthermore, as sodium chloride is present in saltwater, a significant overpotential will result in a major unfavorable reaction of chlorine evolution and electrode degradation. Furthermore, it is well understood that saltwater is a complex solution containing up to 3.5 % different salts, in which multiple competing redox processes and dramatic pH swings result in catalyst deterioration and biofouling (Wang et al., 2022).
A large number of strategies for preparing electro catalysts for sea water splitting have recently been reported. Through structural and morphological engineering of various catalysts the electro catalyst performance in seawater splitting is improved. In addition, carbon-based electro catalysts have recently been discovered to have large surface area and elevated surface activity, allowing them to widen the interfacial interaction between the electrode and electrolyte and thus improve electron, ion, and reactions product transfer (Liu et al., 2020). As a consequence, most carbon-based catalysts may be capable of achieving the efficiency of seawater splitting, and appropriate materials as precursors are required for the creation of carbon-based electro catalysts. A suitable precursor can aid to boost the catalyst's effectiveness and broaden its range of applicability. Amongst several precursors, zeolite imidazolate frameworks which are built by coordinating metal ions with imidazole linkers, have received a lot of attention because of metal ions, that are distributed equally as catalytic active centers and the particular frame structure can be preserved after heating treatment. The overall conductivity and stability of ZIF-derived materials, however, limit their use in electro catalysts. Mixing with a durable and conducting substrate has been shown to be a successful solution to this problem (Khan et al., 2021). Zhang et al. reported the carbonization of a composite material made up of ZIF and carbon paper (ZIF-L-Co@CP) to produce a novel multifunctional catalyst (Co/ Co3O4@C). Surprisingly, the OER property of Co/Co3O4@C seems to be much superior in real seawater than in 1 M KOH. The ability of Co/Co3O4@C (1.929 V) to achieve a current density of 10 mA/cm2 is significantly lower than that of ZIFL-Co@CP, though there is still a small difference when compared to IrO2 (1.858 V). Co/Co3O4@C also shows good catalytic activity for HER in seawater. Overall, Co/ Co3O4@C displays excellent HER and OER catalytic activities in seawater, which can be used to enhance the electrophilic adsorption and OH– oxidation during the oxygen evaluation reaction (Zhang et al., 2020). To our understanding, the majority of carbon-based catalysts reported have excellent water splitting ability in alkaline electrolytes, but only a few have been used in actual seawater.
The purpose of this article is to discuss the current advancement of MOF-based materials for photocatalytic and electrocatalytic water splitting processes. Numerous key variables affecting the activity of water splitting reactions are reviewed, and techniques for catalyst design are highlighted. The authors emphasize significant problems in the domains of photocatalytic and electrocatalytic water splitting and provide some insights on current developments in the production of MOF-based catalysts. Future research directions are indeed discussed, with a particular emphasis on obtaining the required MOF functionality and developing framework correlations in order to discover and explain the variables that affect catalytic activity. This article will offer an overview of current developments in this dynamic sector, and also some suggestions for the future development of highly effective photocatalysts and electrocatalysts for water splitting based on MOFs.
1.1 Stability challenges and its solutions of MOFs during water splitting
An entirely new family of porous materials, known as metal–organic frameworks, has been created by combining metal ions and organic ligands. They have been widely used in a wide range of fields due to the various of functions they can perform owing to its regular and well-defined structures and relatively high surface areas as well as its capacity to be tailored in many different ways (Gong et al., 2021). However, according to the latest findings, majority of MOF types are unstable when exposed to water and losing their structural integrity. Because of metal and oxygen connections inside the frameworks are rather weak, they are rapidly targeted by molecules of water and thus it loses its structure, preventing their operability in water splitting either by electro catalytic way or photo catalytically. Further, the charge and mass transmission may be hampered by the intrinsically poor conductivity and tiny pore size, which is difficult to compensate for even if a much more permeable and conducting support is used. Likewise, majority of metal active sites are firmly encircled by organic ligands, rendering them unable to participate to the reaction. In addition, sustaining the MOF structure during the harsh oxidative conditions of the OER process is also difficult (Fan et al., 2017).
To address these problems, researchers have worked hard to create a variety of ways to improve MOF stability. In general, the stability of MOFs is governed primarily by the metal and ligand bond strength when it come in contact with water. For improving stability Pearson's hard/soft acid/base (HSAB) concept may be used to estimate the strength of metal–ligand coordination bonds. Stable MOFs may be made via the de novo synthesis approach by linking high-valent metal ions (hard acids) with carboxylate-based ligands (hard bases). UiO-66 and MIL-101 are some well-known examples for water splitting photo catalytically (Luo et al., 2020). Similarly, stable MOFs may be made by combining low-valent metal ions (soft acids) with azolate ligands (soft bases), such as Ni3(BTP)2, ZIF-8, and PCN-602, among others (Gao et al., 2021). Another way to improve the stability of MOF based catalysts for water splitting is by the introduction of rigid and hydrophobic linkers utilization during MOF synthesis. High-connected metal clusters are usually linked with rigid organic linkers that are generally very stable. This typically leads to dense and rigid frameworks. Because some isostructural MOFs have organic linkers that get longer, this can make the framework less stable. On the other hand, making low permeability or interfaces by adding hydrophobic groups to the linkers can also make MOFs more stable.
Fortunately, the insertion of hydrophobic functional groups inside the frameworks has the potential to significantly strengthen the M−O bonds, thereby significantly increasing the water resistance of the MOFs. For example, inserting a CH3 functional group inside the framework may improve its water/moisture stability. In this regards, Liu et al. reported Cu-MOFs complexes for oxygen evaluation reaction. He suggested, the incorporation of hydrophobic functional groups (methyl) on the ligands attributes to the stability of Cu-MOF significantly, which plays key role in better electrocatalytic activity for oxygen evaluation reaction during water splitting in an alkaline media by showing high electrocatalytic activity with lower overpotential values such as 1.54 V and low Tafel slope value i.e., 122 mV/dec. Lastly, via introduction of hydrophobic layer on the surface. Aside from the internal elements mentioned above, the outer surface of MOFs, has also an impact on MOF stability. The surface wettability of MOFs can be changed by coating hydrophobic substances on the exterior of water - soluble MOFs or by heating them after they've been made, which makes them more stable. With the above methods, a lot of stable MOF materials were prepared for oxygen evaluation reaction either thru photocatalytic and or electrocatalytic way of water splitting (Zhu et al., 2018).
2 Photocatalytic water splitting
A number of studies is focusing on photocatalytic water splitting to produce oxygen and hydrogen as a possible source of renewable energy generation which is not dependent on fossil fuels and emits no carbon dioxide. Photocatalytic water splitting is termed as an artificial photosynthesis, since it is comparable to photosynthesis process in green plants by utilizing solar energy (Abe, 2010; Shi et al., 2014). In addition, hydrogen production from organic precursors from wastewater possibly will achieved with photocatalytic processes by utilizing sun energy that hit the surface of earth each day (Clarizia et al., 2014). The photocatalyst-assisted water splitting technology has received considerable recognition because to its ecologically friendly conversion of solar energy to clean and storable chemical energy. The ideal and ultimate objective for practical applications is to split the water without requiring a sacrificial reagent. Unfortunately, photocatalytic water splitting confronts number of challenges due to thermodynamic and kinetics limitations as water splitting is an uphill process with a Gibbs free energy of 237 kJ/mol (Xu et al., 2018). Generally, the oxygen evolution half-reaction, which has several stages and a high activation barrier, limits the total water-splitting process. A photocatalyst must not only have a proper band structure with adequate redox power, but also have a high separation efficiency of photogenerated carriers and a sufficient quantity of active sites to provide a desirable total water splitting efficiency. Rarely a photocatalyst meet all of the above cited criteria.
For photo-catalytically splitting of water, there are more than one methods are presented (Yang et al., 2021). One method is to split water into oxygen and hydrogen using a single visible-light-responsive photocatalyst with enough capability to accomplish total water splitting. In such system the photocatalyst must have an appropriate thermodynamic potential for water splitting i.e., a relatively low band gap to collect visible photons, and a high level of durability over photo corrosion. These strict criteria mean that the range of photocatalysts that can reliably split water in a single process is restricted (Srinivas et al., 2021).
Therefore, various techniques, such as doping, metal loading, heterojunction construction, and others, have been researched to improve the photoconversion efficiency of photocatalysts throughout last few decades. Among these proposed solutions, combining two semiconductors to make a type-II heterojunction photocatalyst is one of the simplest ways to improve photocatalytic activities, owing to its efficacy in spatially isolating photogenerated electron–hole pairs via the band gaps of two semiconductors. In a typical type-II heterojunction, semiconductor A conduction band and valence band are both greater than those of semiconductor B. (Fig. 3a). As a consequence of band alignment, photogenerated electrons in semiconductor A conduction band will migrate to semiconductor B conduction band upon light irradiation. Consequently, photogenerated holes in semiconductor B valence band will move to semiconductor A valance band. As photogenerated holes and electrons collect on semiconductor A and semiconductor B, a type-II heterojunction photocatalyst can accomplish spatial separation of the electrons and holes to improve photocatalytic activity. However, there are a number of issues that prevent type-II heterojunction photocatalysts from being widely used (Lu et al., 2022). More in detail, type-II heterojunction photocatalysts' reduction and oxidation reactions occur for semiconductor B with a relatively low reduction potential and semiconductor A with a lower oxidation potential, respectively. As a result, type-II heterojunction photocatalysts' redox capacity will be considerably diminished. Additionally, electrons in semiconductor A and holes in semiconductor B have a lot of difficulty during migration to the electron-rich conduction band and hole-rich valance band regions of semiconductor B and A, respectively, due to electrostatic repulsion between hole–hole and electron–electron. To overcome these issues, new heterostructure photocatalytic systems are desperately needed (Xu et al., 2020).
Alternatively, the second method is the use of two distinct photocatalysts in a two-step excitation method, inspired by the Z-scheme (a natural photosynthesis of green plants), and in 2013, Yu et al. came up with the idea of a direct Z-scheme photocatalyst to explain why the TiO2/g-C3N4 composite had a high photocatalytic formaldehyde (HCHO) degradation rate. In recent times, the Z-scheme configuration in photocatalytic based systems is one of the most up to date strategies to enhance the performance of catalyst in contrast to single semiconductor photocatalyst utilizations. Usually, Z-scheme systems are classified with redox intermediates, without intermediaries of electrons, and with solid state electron intermediaries (Li et al., 2016). Generally, reactants, photocatalyst, photoreactor and light for the emission of hydrogen are required in a photocatalytic system. Water act a reactant, for photocatalyst, it must be functioning with light, either visible or UV light. Absorbing light in the visible range might be an assurance to capture considerable part of the energy evolved by the sunlight (Tahir and Amin, 2013). Photocatalytic reaction starts with light irradiation equal to the band gap of photocatalyst may excite the electron from valance band to conduction band. Generated electron and hole pair is separated and use it for reduction and oxidation (Acar et al., 2014).
Compared to the one-step water splitting system, developing a direct Z-scheme photocatalyst significantly suppresses electron–hole recombination and isolates the photocatalytic reaction oxidation and reduction sites. As a result, numerous direct Z-scheme photocatalysts have now been investigated in order to maximize the effectiveness of photocatalytic hydrogen production (Li et al., 2021; Lu et al., 2019). Further, Z-scheme water splitting system has the benefit of a broader spectrum of visible light that's because the Gibbs free energy needed to drive a photocatalyst may be lowered, as well as the separation of hydrogen and oxygen is also possible (Nasir et al., 2020; Nishioka et al., 2019), following one step and two step photocatalysis of water splitting is shown in Fig. 2.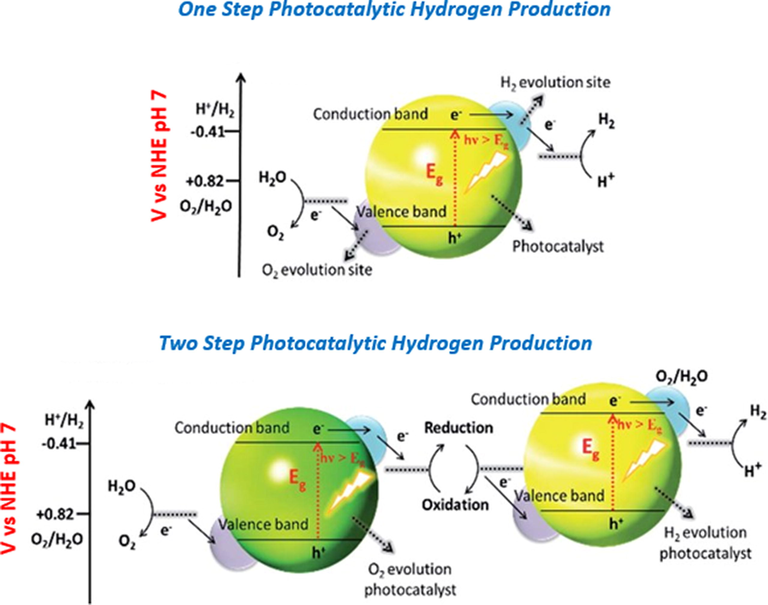
Representation of one step and two step photocatalytic hydrogen production through a photocatalysts for water splitting (Maeda and Domen, 2010). Exclusive rights 2010 American Chemical Society.
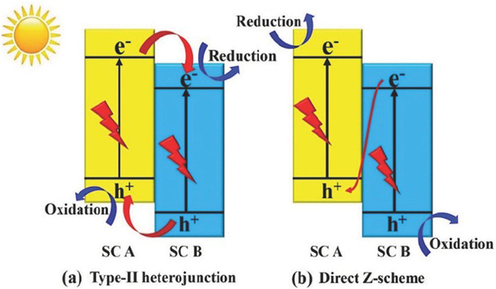
Charge-carrier separation mechanisms on (a) type-II heterojunction and (b) direct Z-scheme for water splitting via two distinct photocatalysts (Low et al., 2017). Exclusive rights 2017 Wiley Online Library.
Further, the structure of a direct Z-scheme photocatalyst is identical to that of a type-II heterojunction photocatalyst (Fig. 3a,b), but the mechanism of charge carrier migration is different. In a typical direct Z-scheme system, the charge-carrier migration pathway resembles the letter “Z”. During the photocatalytic reaction, the photogenerated in semiconductor B with lower reduction ability recombine with the photogenerated holes in semiconductor A with lower oxidation ability. As a result, the direct Z-scheme photocatalyst's redox capability can be improved. It should also be noted that charge-carrier migration in direct Z-scheme photocatalysts is physically more feasible than in type-II heterojunction photocatalysts, because the migration of photogenerated electrons from semiconductor B conduction band to semiconductor A photogenerated hole-rich valance band is preferred owing to the electrostatic attraction between the hole and the electron (Low et al., 2017).
Furthermore, the performance of photocatalytic water splitting may be determined by analyzing the efficiencies of total water splitting in the absence of sacrificial reagents. H2 and O2 are created simultaneously in a 2:1 stoichiometric ratio during total water splitting (eq. 1–2). To explore the kinetics of the proton reduction or water oxidation half reactions, sacrificial reagents are used to rapidly consume the photo - induced holes or electrons, thereby rendering the related half reaction as rate determining step. For instance, lactic acid, methanol, or triethanolamine are often employed as hole scavengers in the H2 generation half reaction (eq. 3–4), whereas Ag+, Fe3+ or IO3 are typically utilized as electron scavengers in the O2 evolution half reaction (eq. 5–6) (Li and Li, 2017). The overall photocatalytic water splitting mechanism for Hydrogen production is demonstrated in below equations (Fajrina and Tahir, 2019).
Photocatalysis is a potential way to convert solar energy into chemical energy, various co-catalysts are frequently utilized to increase charge carrier separation at the semiconductor-electrolyte interface, hence enhancing the charge transport kinetics of the holes for water oxidation. Unfortunately, noble metal co-catalysts such as Pt, Au, and Ag nanoparticles are exceedingly costly. This necessitates the fabrication of photocatalysts that are cost effective (Wang et al., 2022). In this regard metal–organic frameworks have subsequently emerged as novel photocatalysts, due to their intrinsic structural properties of having a high specific surface area with a high porosity (Li et al., 2016). In comparison to conventional photocatalysts, MOFs exhibit a number of significant benefits in photo-induced reactions. MOFs, for example, can facilitate the exchange and dissemination of molecules due to their desired topology and varied pore structure, which inherits the benefits of porous materials like zeolite, carbon nanomaterials and covalent organic frameworks (COFs). Additionally, numerous catalytic active sites are accessible as a result of the porous nature and large surface area of MOFs (Limwichean et al., 2021). The most unique characteristic of MOFs is their chemical and physical features, which can be augmented through careful design and tuning of their structure and composition; consequently, numerous reports have indeed been devoted to identifying the appropriate modification strategy for MOF photocatalysts for particular photo-induced applications. Numerous studies on the use of MOFs in photocatalytic water splitting have already been published in recent years as illustrated in Fig. 4.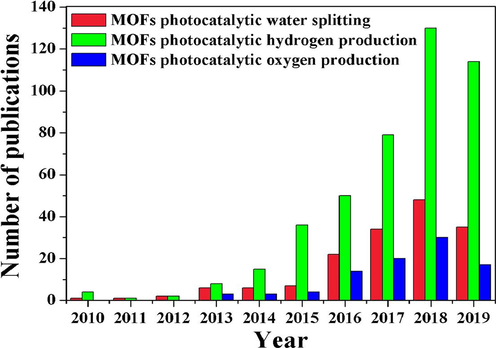
The latest study advances in photocatalytic water splitting using metal–organic frameworks (Luo et al., 2020). Exclusive rights 2020 Elsevier.
2.1 Fabrication strategies for MOF derived photocatalysts
Many studies have shown that MOF-based photocatalysts with a porous structure can give various benefits during the photocatalytic activity, including abundant carrier charge transfer pathways, many reaction active sites, and excessive adsorption and desorption channels. In comparison to other semiconductor-based photocatalysts, MOF-based photocatalysts have the following advantages: i) the organic and inorganic constituents in MOFs contributes to enhance the structural stability; ii) the adjustable structure of MOFs promotes carrier charge separation and transfer rate. iii) MOFs with a high porosity may efficiently supply a large number of light absorption sites, resulting in enhanced light absorption capabilities (Sun et al., 2021). Such as Bag et al. reported CdS@NU-1000 and CdS@NU-10001% rGO composites for water splitting reaction under the irradiation of visible light exhibits 12.1 time more than CdS, due to its high surface area provided by rGO substrate and porosity is provided by MOF structure (Bag et al., 2017). Further, many researchers are inspired by these benefits, and they try to synthesize various types of MOFs and their composite as photocatalysts using variety of strategies, such as hydrothermal reaction method, solvothermal method, solution precipitation route, sonication method, microwave strategy, oil bath approach, template method, and so on (as shown in Fig. 5) (Su et al., 2018; Qian et al., 2021).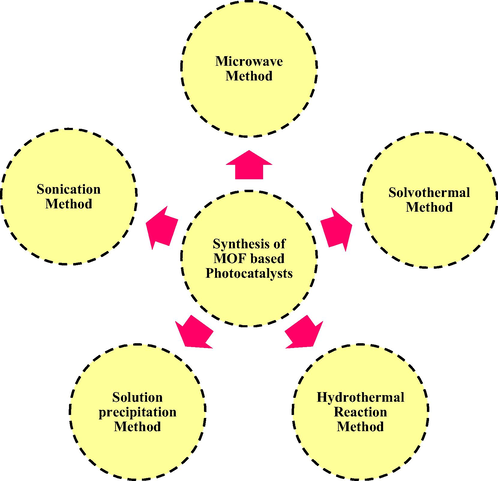
synthetic routes of MOF based photocatalysts.
2.1.1 Solvothermal method
To synthesize MOFs and their composites as photocatalysts, the solvothermal approach is preferred because of its high yield, ease of use, low pressure and low temperature but selecting a suitable organic solvent is crucial points as organic solvents serve as both a structure-directing agent and a synthesis medium throughout the synthesis process. Various organic solvents, such as N,N-dimethylformamide, dimethyl sulfoxide, N,N-dimethylformamide and toluene, have been widely used to fabricate MOFs and their composites up to now. Among above mentioned organic solvents the DMF is most often selecting for the synthesis of MOF and its composites, due to its high boiling point and solubility, for instance, Ye et al. reported rGO/ZnIn2S4 nanocomposites were synthesized by utilizing a one-pot solvothermal technique from InCl3, ZnCl2, and TAA in the presence of varying amounts of GO in a 1:1 mixture of DMF and EG. To make rGO/ZnIn2S4 nanocomposites with a sheet-on-sheet morphology, a simple one-pot solvothermal process was established (Ye et al., 2014). Further, Huang et al. reported a MOF (PCN-624) was effectively prepared in the presence of DMF at 120 °C for 72 h through the solvothermal technique. Specifically, by incorporating the 12-connected nodes such as [Ni8(OH)4(H2O)2Pz12] into PCN-624 during the solvothermal process, the as-prepared sample demonstrated significantly increased photocatalytic stability under a variability of conditions, including organic solvents, strong acid, and base solutions (Huang et al., 2018). Besides, numerous reports illustrated that MOFs and its carbon based composites were also synthesized in a variety of sizes and morphologies via the solvothermal method, for example, the polyhedral PCN-250-Fe3 with a size of 13 m, UiO-68-TZDC over sizes ranging from 10 to 20 m, and the octahedral UiO-68.
Though the solvothermal approach has been widely applied to manufacture MOFs and their composite-based photocatalysts, the organic solvents' toxicity and environmental unfriendliness significantly limit their widespread deployment. Additionally, it is difficult to eliminate organic solvents afterwards the synthesis. Thus, it is critical to create green, safe, and environmentally acceptable solvents for the solvothermal approach of fabricating MOFs and their composite-based photocatalysts in the near future.
2.1.2 Hydrothermal method
Hydrothermal reactions can also be used to prepare MOFs and their composite-based photocatalysts. The reaction parameters, including the reaction temperature, solvents, pH, solvents, precursors, and reaction time, may be adjusted flexibly during the hydrothermal reaction process. MOFs and their composites with adjustable size and structure may be readily created by carefully controlling the synthesis conditions. Several research groups took use of the benefits of the hydrothermal reaction approach to manufacture high-performance MOFs and composite-based photocatalysts. Karthik et al in 2018 reported a reduced graphene oxide based MOF i.e., NH2– MIL-125(Ti)/rGO photocatalyst for water splitting, prepared via hydrothermal method at 150 °C for 48 h. The strong π − π connection was generated by combining NH2-MIL-125(Ti) MOF with rGO and as a result of this interaction an enhanced photocatalytic hydrogen production ∼9.1-fold is measured in contrast to NH2-MIL-125(Ti) MOF.
Even though the hydrothermal reaction approach is an effective method for preparing MOFs and composite-based photocatalysts with adjustable size and shape, it still has significant drawbacks, such as low yield and extended reaction times. Furthermore, the creation mechanism of MOFs and their composites during the hydrothermal reaction phase remains unknown.
2.1.3 Solution precipitation method
Solution precipitation has been a frequently utilized approach for the fabrication of inorganic nanomaterials during the last several years. Since the cations are precipitating simultaneously, this technique is also known as the “co-precipitation method.” In comparison to hydrothermal reaction, the solution precipitation technique is more simple and quicker for the synthesis of MOFs and their composites due to the ease of control and mild experimental conditions. Numerous research groups have taken use of these advantages to synthesize MOFs and their composites for use as photocatalysts. For instance, in 2021, Peng et al. synthesized Zn-ZIF-L composites by a one-step solution precipitation approach. In this study, Zn(NO3)2·3H2O and MIM were combined in ultrapure water and then stirred at a low speed for>4 h to create white precipitates. After that, the prepared Zn-ZIF-L was employed as a precursor to produce the two-dimensional (2D NC) N-doped nano porous carbon composites. Due to their high porosity and large surface area, these 2D NC composites exhibit a quick activity.
Apparently, the one-step solution precipitation approach is an easily manageable procedure for fabricating MOFs and their composites. Additionally, by carefully controlling the synthesis parameters, like stirring speed, reaction duration, and temperature, homogeneous morphology MOFs and their composites may be obtained. We expect that this approach will attract more interest in the near future for the fabrication of MOFs and their composites.
2.1.4 Sonication method
The sonication approach has been established and developed as an efficient method for fabricating MOFs and their composites. In general, energy is transferred during sonication synthesis by acoustic cavitation, which would be the production, growth, and implosion of bubbles within the solvent. A sonication procedure like this, along with the produced high pressure and energy, can produce novel chemical effects and make it easier to construct a new structure. Numerous studies have been published so far employing the sonication approach to synthesis MOFs and composite-based photocatalysts. For instance, Abazari et al. reported g-C3N4/ Ti-MIL125-NH2 composite photocatalysts via sonication method. To make the g-C3N4/Ti-MIL125-NH2 composites, the g-C3N4 powder was first disseminated in a combination of methanol and DMF; the TPOT and 2-ATA were then added to the aforesaid solution and sonicated for 20 min. The as-fabricated g-C3N4 based Ti-MIL125-NH2 composites have a wide surface area and strong light absorption capabilities (Li et al., 2018).
Besides, the tremendous energy and high pressure created during the sonication process help to create a stable structure. Because of these features, the sonication process has a lot of potential for producing high-quality, large-scale, and long-lasting MOFs based photocatalyst composites at quite a low cost.
2.1.5 Microwave method
The microwave approach has recently been recognized as a strong approach for generating functional materials. Throughout the reaction progression, the frequency of electromagnetic irradiation typically varies from 0.2 to 300 GHz, and energy may be transmitted quickly from the electromagnetic irradiation to the reaction precursor. The microwave technique is safer and quicker than the hydrothermal and solvothermal methods. Many research groups used this strategy to synthesize MOFs and their composite photocatalysts, fascinated by the benefits listed above.
For instance, Zhu et al. reported rGO based composites with MOF derived ZnO, synthesized via microwave assisted method. In summary, 16 mg of MOF-derived ZnO was introduced to a 20 mL graphene oxide solution, which was subsequently treated for 10 min at 150 °C with a microwave irradiation strength of 150 W using an automatic focused microwave system (Zhu et al., 2017). Though the microwave technique has been effective in fabricating MOFs and their composite photocatalysts, there are still major drawbacks in controlling the shape and size.
2.2 Basics of photocatalytic water splitting
The capability of photocatalyst to generate e-/h+ for the water splitting depends on number of factors that comprises of; (i) it should have greater capability to absorb wide range spectrum light to confirm the conversion of maximum photons in to charge carriers i.e., e- and h+. (ii) The photocatalyst's band gap should be large enough i.e., 1.23 eV to fulfill the thermodynamic requirement of the overall water splitting process (Ullah et al., 2018): (iii) The photocatalyst's conduction band edge must be relatively negative to the H+/H2 redox potential (0.0 V vs. NHE), while the valence band edge must be relatively positive to the O2/H2O redox potential (1.23 V vs. NHE). Furthermore, for oxygen and hydrogen generation the redox potential is dependent on the electrolyte pH (Ni et al., 2007): (iv) Another most important factor is the lifetime of the charges that must be protracted enough to participate in a particular redox process. This means photogenerated charges must reach the surface reaction sites before their recombination (Luo et al., 2020).
After the photocatalytic tests, TOC analysis was performed on the solutions to determine the degradation of organic sacrificial agents in the solutions. TOC values have been correlated with the overall concentration of organics in the solution (Kumaravel et al., 2019).
2.3 Challenges of photocatalytic water splitting
The rapid recombination process, poor absorption capacity, redox potential variance, and high diffusion barrier for charge transfer in successful catalysis processes are the main challenges that need to be focused. Additional difficulties connected with water splitting catalysts include poor absorption capability, structural degradation owing to photo corrosion, and recombination of rapid charge carriers (e /h+). In addition to many other materials investigated over the past decades, MOFs are developing materials with the ability to solve the related problems owing to its intrinsic catalytic capability and tunable characteristics (Li et al., 2021). Some of the other challenges are:
2.3.1 Light absorption
In order to mollify the thermodynamic requirement of 1.23 V for photocatalytic water splitting, light absorbing materials with a sufficient band gap are required. To overcome kinetic limitations and successfully release molecular products, some overpotential is needed. The greater the overpotential, the quicker the electron transport to the catalytic site for accelerating the redox process. However, significant overpotential limits the choice of photocatalyst to broad band gap materials that exclusively use UV light. Various methods have been used to optimize photocatalytic material absorption ranges while considering thermodynamic and kinetic limitations (Kato et al., 2003; Marcus, 1957).
2.3.2 Charge separation
After generating (e /h+) pairs, their isolation must be sustained until they can be used effectively in specific redox processes. Unfortunately, the majority of charge carriers recombine prior to participating in redox reactions due to its relatively limited survival time (nanoseconds) in comparison to water reduction (milliseconds) and oxidation (seconds) processes. Numerous methods have been developed to extend the lifespan of charge carriers in order to solve this challenge (Tang et al., 2008; Huang et al., 2018).
2.3.3 Effective charge utilization
In the photo-assisted water splitting process, the interfacial charges (e /h+) are transferred to the adsorbate molecules (OH2/H+ /OH), resulting in the formation of redox products (H2/O2), which is the rate determining factor. Usually, just one reaction, presumably oxidation or reduction, occurs to effectively generate the required product, while the remaining half of the reaction is scavenged by the sacrificial agents. During the reduction half reaction (HER), reductive sacrificial agents such as triethanolamine and many other are used to scavenge the holes. In comparison, during the oxidation half reaction (OER), electron acceptor scavengers (e.g., Na2S2O8) are employed to obstruct the HER reaction. But the sacrificial agent's redox potential has to be greater than that of the photocatalyst, which may be fine-tuned through pH modification (Ullah et al., 2021).
2.4 Key strategies for improving MOF-Based photocatalyst water splitting performance
Many strategies have been developed to optimize the desirable characteristics of photocatalysts while also resolving related difficulties in order to increase the total photon to hydrogen conversion efficiency. Therefore, motivated by the aforementioned structural characteristics, photocatalysts of the MOF type have lately been extensively studied and categorized in to two subdivision, MOFs and MOF based compounds. Though, MOFs as photocatalysts face certain bottlenecks such as their low quantum efficiency. There are certain key strategies that has been adopted for improving the MOF-Based photocatalyst water splitting performance it includes:
-
By band gap engineering
-
By active sites engineering
-
By coupling of metal organic framework with other functional materials
2.4.1 By band gap engineering
Due to the fact that photocatalytic activity is largely reliant on the photocatalyst's electronic structure, it is critical to modify the bandgap in order to use the maximum amount of visible photons and reach the goal of 10% solar to fuel conversion efficiency and by integrating the benefits of inorganic and organic chemistry, metal–organic frameworks (MOFs) offer a flexible platform for photocatalysts (Wang et al., 2018).
The majority of MOFs may be thought of as an assembly of semiconductor quantum dots that are dispersed uniformly across the structures and separated through tunable organic linkers. Both organic linkers and inorganic metal nodes may be readily tailored for light harvesting via the use of appropriate organic linkers, metal nodes, and their adjustable coordination modes (Li et al., 2021). Researchers have published large number of publications utilizing MOFs for photocatalysis across the globe in recent decades, but only a few dozens of papers concentrate on the engineering of band gaps from a theoretical and experimental standpoint. The band gap modulation of MOFs was addressed in fewer review papers (Guo et al., 2021).
according to Lu et al., band gaps may be reduced, via increasing the degree of linker conjugation, selecting electron-rich metal nodes and organic molecules, and decorating linkers with nitro and amino groups. The modifying photoelectronic performance of MOFs for premium photocatalysis was summarized by Duan et al. in 2019. Based on the categorization of energy photocatalysis. Many researchers presented various methods for band gap engineering of semiconductor-type MOFs (Zhan et al., 2019).
Furthermore, Volkmer et al. suggested a number of alternative methods for lowering the band gap energy, such as (i) by increasing the degree of conjugation of the linker to achieve VB at a higher energy level, (ii) choosing a metal with diffuse and partially unoccupied d-orbitals as the node to lower the CB energy level, and (iii) by putting rich electronic fragments into the nodes of MOFs to lower the CB energy level (Kondo et al., 2021). In addition, Allendorf et al. suggested three broad strategies for modifying MOF electrical properties, such as (i) by changing the metal ion, (ii) by altering the organic linker, and (iii) by introducing specific organic groups into the frameworks (Yao et al., 2021). A series of MOF photocatalysts were produced by Wang and Huang et al via a Schiff-base reaction involving aldehyde[2/3/3- pyridine carboxaldehyde (2/3/4-PA)] and NH2 in the NH2-MIL-125 MOF groups as shown in Fig. 6a. XPS valence band spectrum and Mott-Schottky plots correspondingly have been used to evaluate CB and VB potential of heterocycle grafted NH2-MIL-125 and the experimental findings show that the decrease of the bandwidth of implanted MIL-125 is positive with the aromatic heterocycle's electron donating ability as illustrated in Fig. 6b. The limited capacity to absorb light and the challenging re-combination of semiconducting MOFs restrict artificial photosynthesis (Karthik et al., 2020). The insertion via simple post-synthetic coordination of metal ions into MOFs may not only increase optical absorption but also enhance the solar-to-fuel transformation. Moreover, the effective incorporation of transition metal ions (Ti2+, Ni2+, Co2+ or Cu2+), via coordinating with –NH2 Group, by means of a post-synthetic change into NH2-MIL-125 and referred as Ni-MOF, Co-MOF, Ti-MOF, and Cu-MOF and illustrated in Fig. 6c. The UV–Visible tests reveals, absorption changes into the visible area as a result of the d-d transitions of coordinated M2+ ions. The band gaps of Ni-MOF, Cu-MOF, Co-MOF and Ti-MOF, compared to Mott-Schottky and Tauc plots, are 1.9 V, 1.7 V, 2.2 V, and 2.7 V correspondingly (Fig. 4b). Based on the coordination of M2+, the VB potential of all M−me MOF are lower than the pure Ti-MOF since it only interacts with organic linkers (Huang et al., 2021).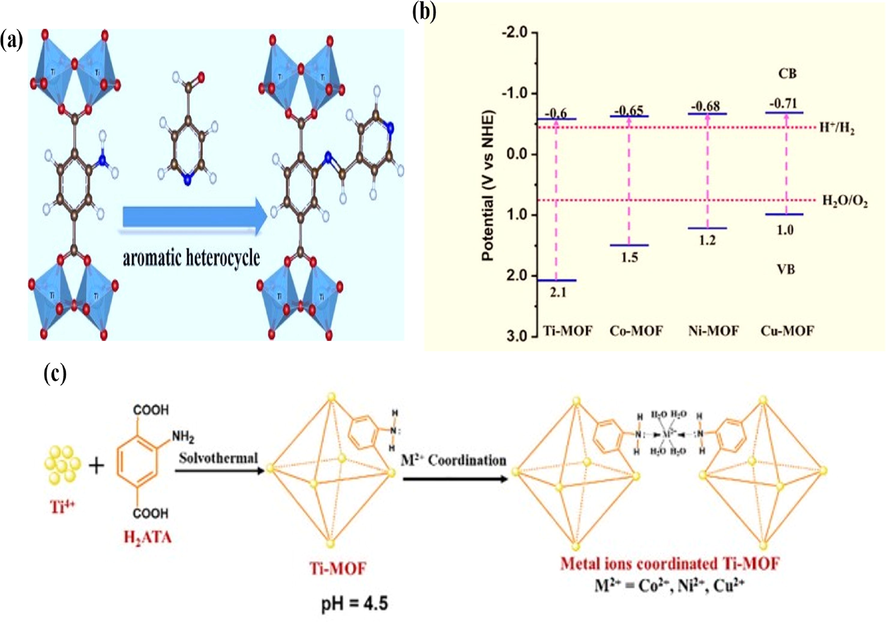
(a) Schematic of NH2-MIL-125 post-synthetic grafting with aromatic heterocycles; (b) Band levels of Ti-MOF, Co-MOF, Ni-MOF and Cu-MOF(c) Schematic depiction of M2+ ions covalently linked to NH2-MIL-125 (Guo et al., 2021). Exclusive rights 2021 Elsevier.
2.4.2 By active sites engineering
Recently, a wide range of MOFs have been developed and manufactured, with the variability owing to the wide range of metal nodes and organic ligands. Metal nodes are, in general, the most important component in catalytic performance, and they may be purposefully chosen to create MOFs with the best activity (Wang et al., 2021). Using MOFs as carriers for photo catalytically active species is a more advanced way to employ them for photocatalysis. This technique has been extensively used for the encapsulation of a range of active sites, ranging from semiconductor nanoparticles to molecular catalysts and overall based on transition metal complexes. In this context, the MOF may either serve as a passive container or take part in the process of charge transfer (Hu et al., 2021). One of the most appealing aspects of employing MOFs to sustain active species is that these groups covalently linked to the framework or enclosed in its cavities. This method has been shown to avoid the discharge of homogeneous catalysts, which are typically made up of precious metals and are soluble under certain reaction conditions (Hu et al., 2021). In 2011 Lin and colleagues published a variety of UiO-67(Zr) materials doped with Re-, Ir- and Ru- complexes, which were used for water oxidation, respectively. The photocatalytic activity of the solids is similar to that of homogenous counterparts. Furthermore, the catalysts' recyclable status was verified, demonstrating their heterogeneous character (Karthik and Neppolian, 2021).
2.5 Carbon based MOFs as photocatalysts in water splitting
It is well understood that MOFs alone exhibit low electrical conductivity. This constraint prevents them from demonstrating their full potential in the electrochemical sector. While conductive MOFs have been discovered lately, they are not yet frequently used owing to numerous restrictions. Apart from their low conductivity, MOFs suffer structural instability due to the weak coordinated connection between organic ligand and metal node, as well as a high moisture vulnerability, that further restricts their commercial use. Some functional carbon additives have been incorporated into MOF catalysts to improve the capacity to transfer charges. Graphene (2D polyaromatic carbon allotrope) has attracted great attention in the field of hydrogen production from water splitting because of its high theoretical surface area, outstanding electro chemical stability, superior electrostatic conductivity with few additional exclusive chemical and physical proprieties, it may be rolled into a 0D, 1D, 3D structure of carbon atoms known as fullerenes, carbon nanotube and a graphite assembly. Another derivative of graphene is reduced-graphene oxide (rGO), is formed by the reduction graphene oxide by number of synthetic approaches. It offers number of substantial characteristics when it is utilized as photocatalyst for the splitting of water as it offer more active sites, increase the separation of charges, and suppress the recombination of charges. Furthermore, availability of oxygenated functional groups just like GO have also been increase the number of active sites for water splitting. In addition, with band gap of 11.69 eV it also creates a heterojunction when it mixes with other light absorbing substances, and further improves the separation of charges and suppressing the recombination of charges thus overall, it plays a significant role in water splitting (Xie et al., 2013). Furthermore, for various photocatalysts graphene is a good support material due to its large surface area. Besides, the presence of defective sites serve as a nucleation site for the metal oxide part of the MOFs. Likewise, reduced graphene oxide (rGO) with a negatively charged surface, is a desirable adsorbent for metal oxides as well. And among them the presence of attractive forces will avoid the agglomeration of following nanoparticles improving the photocatalyst's stability and reusability (Fadlalla and Babu, 2019).
The use of MOF structures to decorate a graphene sheet reduces charge recombination while simultaneously facilitating charge carrier separation and transfer. Karthik et al in 2018 reported a photo catalyst i.e., NH2-MIL-125(Ti) MOF and reduced graphene oxide based NH2-MIL-125(Ti) MOF composite for hydrogen production via water splitting. The strong π − π connection was generated by combining NH2-MIL-125(Ti) MOF with rGO, and as a result of this interaction an enhanced photocatalytic hydrogen production ∼9.1-fold is observed in contrast to NH2-MIL-125(Ti) MOF. Overall mechanism involve in hydrogen production is illustrated in Fig. 7.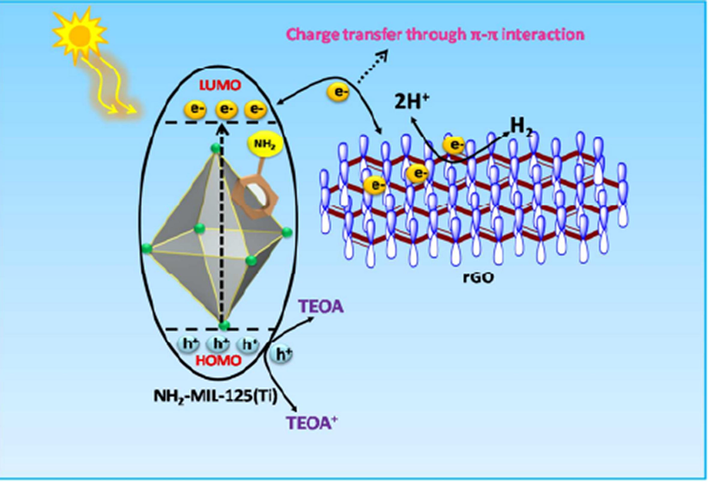
Schematic mechanism for photocatalytic H2 production under visible-light irradiation (Karthik et al., 2018). Exclusive rights 2018 American Chemical Society.
In photocatalytic activity of water splitting following steps are included; (i) visible light is absorbed by a linker 2-amino terephthalic acid, (ii) an electron excitation occurred from the organic linker's HOMO to the LUMO, (iii) following photoexcited electron was then transported to the titanium-oxo cluster of MOF through the ligand to cluster charge transfer mechanism (LCCT), (iv) finally this photogenerated electrons are swiftly transported to rGO via π − π interactions. The transfer of electron through rGO is then engaged in the H+ ions reduction into H2. and the photogenerated holes oxidise the TEOA ( triethanolamine a sacrificial reagent) and form TEOA + ions (Karthik et al., 2018).
Bag et al. in 2012 reported CdS@NU-1000 and CdS@NU-10001% RGO composites for water splitting reaction under the irradiation of visible light. Theses Zr(IV) based MOF i.e., NU-1000 with CdS and rGO based photocatalysts are prepared via using CdS, RGO, and NU-1000 in various proportions, a ternary composite was created: CdS@NU-1000/RGO. The hydrogen production performance of CdS was enhanced by the combination of NU-1000 and rGO, i.e., ternary composite, because of availability of high catalytic sites and reaction centers created by avoiding the agglomeration of CdS nanoparticles caused by NU-1000′s large pore size and minimal charge carrier recombination. Using visible light, CdS@NU-1000 has 9.35 times the photocatalytic activity of commercial CdS whereas CdS@NU-1000/1% rGO has 12.1 times the photocatalytic activity, overall comparison is shown in Fig. 8.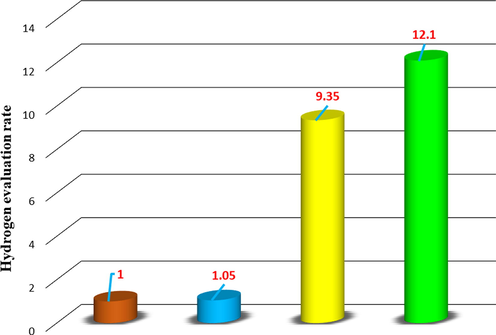
Hydrogen evolution rate under visible light from photocatalyst.
Further, following ternary composites overall water splitting process mechanism, where CdS's lowest conduction band potential and reduced graphene oxide's fermi level are –0.65 V and –0.08 V against NHE, correspondingly, photogenerated electrons from CdS might be transported to NU-1000 and subsequently transferred to RGO sheets. To determine if the holes in CdS could oxidise the NU-1000, the ternary composite was irradiated in aqueous solution without the addition of sacrificial agents to preserve the direct contact between the holes in CdS and the pyrene tetracarboxilic acid fragments of MOF. Thus, this research shows unequivocally the benefit of MOF-supported CdS nanoparticles for improving photocatalytic H2 evolution activity while maintaining high stability (Bag et al., 2017).
Zhang et al. in 2018 reported composites of NH2-UiO-66/g-C3N4 and CD@NH2-UiO-66/g-C3N4 with g-C3N4 and Carbon nanodots for photocatalytic water splitting. CDs were incorporated into the pores of NH2-UiO-66 to create a ternary composite. The inclusion of CDs efficiently enhances visible light absorption areas, improves charge carrier lifespan, and reduces charge carrier recombination. As a result, CD@NH2-UiO-66/g-C3N4 had better photocatalytic performance for visible-light-driven hydrogen production (>420 nm), with a hydrogen evolution rate of 2.930 mmolg-1 h−1, which is 32.4, 38.6, and 17.5 times greater than g-C3N4, NH2-UiO-66, and NH2-UiO-66/g-C3N4, correspondingly. The substantial enhancement of photocatalytic capabilities in the ternary composite may be attributed primarily to the ability of CDs as cocatalysts to efficiently increase electron interfacial charge transfer in the composite system, resulting in a reduction in photo - generated charge recombination. Further, g-C3N4 and NH2-UiO-66 have comparable light absorption, g-C3N4 has a substantially higher CB than NH2-UiO-66, which implies that from g-C3N4 even more photogenerated electrons may move to NH2-UiO-66′s CB and from the CB of NH2-UiO-66 the photogenerated electrons is then readily transferred to CDs and then further reduce H+ into H2, whereas the holes restricted in g-C3N4 and NH2-UiO-66 allow the electron donor sodium ascorbate to be oxidized, Furthermore, including CDs into NH2-UiO-66 improves the material's light absorption capacity, perhaps leading to an increase in H2 generation (Zhang et al., 2018).
Further, for the optimization of the NH2-UiO-66 photocatalyst Zhang et al. in 2020 reported a Pt@NH2-UiO-66@MnOx (PUM) heterostructure photocatalyst using Pt and MnOx as cocatalysts is constructed as illustrated in Fig. 9a. The PUM sample produces the most hydrogen when compared to the pristine NH2-UiO-66, NH2-UiO-66@MnOx (UM) and Pt@NH2-UiO-66 (PU) samples. Pt prefers to capture electrons as a cocatalyst, whereas MnOx prefers to accumulate holes. Electrons and holes move within and outside of the MOF based photocatalyst after being generated from NH2-UiO-66, collecting on the matching cocatalysts, and then participating in the redox processes as illustrated in Fig. 9b. The lifespan of photogenerated electrons and holes is enhanced by the PUM photocatalyst, which favors electron–hole separation. In addition, the PUM sample can help with overall water splitting even when there are no sacrificial agents, which shows that it could be used to change MOF-type semiconductors so that the overall water-splitting reaction can happen.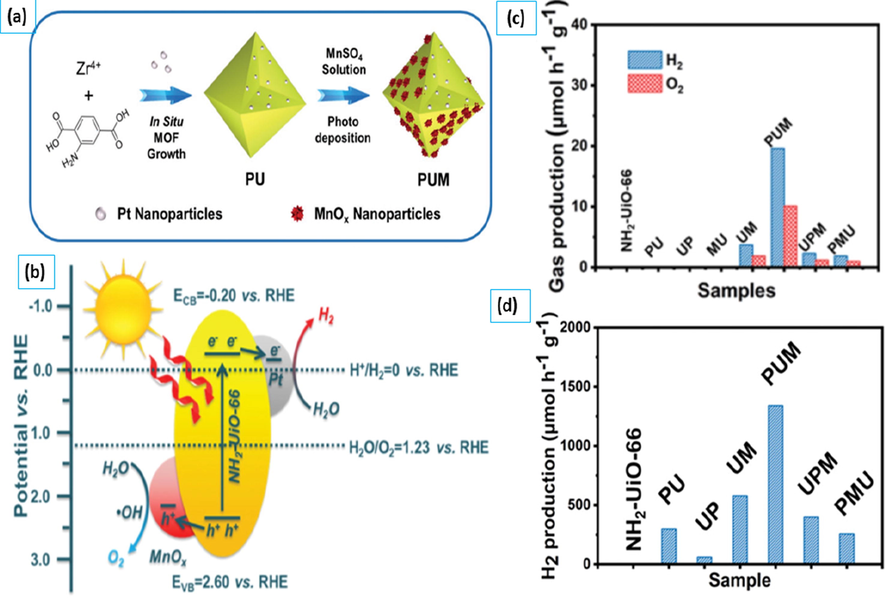
(a) Schematic of Pt@NH2-UiO-66@MnOx preparation; (b) Schematic diagram for overall water splitting by Pt@NH2-UiO-66@MnOx (c) Hydrogen production rates in the absence of sacrificial agent (d) Hydrogen production rates in the presence of sacrificial agent (Zhang et al., 2020). Exclusive rights 2020 Wiley Online Library.
Fig. 9c shows the hydrogen production capability of PUM, UM and PU, in the absence of sacrificial agents only the UM, PUM, and UPM can catalyze the splitting of water into hydrogen and oxygen. Despite the fact that the PUM samples hydrogen and oxygen generation rates is just 19.6 µmol / g/ h and 10.1 mol/ g/ h, are substantially lower than those achieved in the presence of sacrificial agents as illustrated in Fig. 9d (Zhang et al., 2020).
Table 1 and 2 illustrates comparison study from literature by carbon based MOFs as photocatalysts for OER and HER.
MOF-based Photocatalyst
Sacrificial reagents
Illumination (nm)
Activity (µmol h−1 g−1)
Activity (mmol g-1h−1)
Ref.
Bi-based MOF
AgNO3
/
≈180
-
(Wang et al., 2015)
ZnIn2S4@NH2-MIL-125(Ti)
Na2S and Na2SO3
> 420 nm
–
2.204
(Nasalevich et al., 2015)
2.6 wt% CoOx NPs-MIL-101
[Ru(bpy)3]2+–Na2S2O8
/
≈11,000
-
(Han et al., 2015)
3.9 wt% CoOx NPs-MIL-101
[Ru(bpy)3]2+–Na2S2O8
/
≈15,000
-
(Han et al., 2015)
4.9 wt% CoOx NPs-MIL-101
[Ru(bpy)3]2+–Na2S2O8
/
≈13,000
-
(Han et al., 2015)
MnTD/MIL-101
Ceric ammonium nitrate
520 nm
2,250
-
(Hansen et al., 2014)
MIL-101
Ceric ammonium nitrate
520 nm
125
-
(Hansen et al., 2014)
Porous Co3O4 nanocages
[Ru(bpy)3]2+–Na2S2O8
–
≈7,900
-
(Wei et al., 2015)
Porous MnxCo3−xO4 nanocages
[Ru(bpy)3]2+–Na2S2O8
–
≈4,900
-
(Wei et al., 2015)
Porous FexCo3−xO4 nanocages
[Ru(bpy)3]2+–Na2S2O8
–
4,000
-
(Wei et al., 2015)
MOF-based Photocatalyst
Sacrificial reagents (vol%)
Illumination (nm)
Activity (µmol h−1 g−1)
Activity (Mmol g-1h−1)
Ref.
UiO-66
Na2S, Na2SO3
/
0
-
(Zhou et al., 2015)
UiO-66/CdS
Na2S, Na2SO3
/
1,700
-
(Zhou et al., 2015)
UiO-66/CdS/1% rGO
Na2S, Na2SO3
/
2,100
-
(Zhou et al., 2015)
UiO-66/CdS/1% rGO
Na2S and Na2SO3
> 420 nm
–
2.100
(Yuan et al., 2015)
UiO-66/CdS/rGO
Na2S and Na2SO3
> 420 nm
–
2.09
(Yuan et al., 2015)
UiO-66/CdS
Lactic acid
420 nm
1,250
-
(Shen et al., 2015)
UiO-66
Lactic acid
420 nm
0
-
(Shen et al., 2015)
g-C3N4/UiO-66 (1:1, w/w)
Lactic acid
420 nm
1,141
-
(Shen et al., 2015)
Ti-MOF-Ru(tpy)2
TEOA
500 nm
≈200
-
(Zhou et al., 2013)
Pt based MOF-253
CH3CN
440 nm
≈58,000
(Zhou et al., 2013)
Pt@UiO-66
TEOA
> 420 nm
3.9
(He et al., 2014)
2.54 mg g−1 of RhB/Pt@UiO-66
TEOA
> 420 nm
5.6
-
(He et al., 2014)
11.92 mg g−1 of RhB/Pt@UiO-66
TEOA
> 420 nm
≈100
-
(He et al., 2014)
ErB dye-sensitized Pt/UiO-66 octahedrons
Methanol
420 nm
460
-
(Yuan et al., 2015)
Hollow Fe2O3-TiO2-PtOx
Lactic acid
1,100
(Pham et al., 2013)
Co3O4/TiO2 p–n heterojunction
Methanol
7,000
-
(Bala et al., 2015)
UiO-66/g-C3N4
Ascorbic acid
420 nm
14.11
-
(Wang et al., 2015)
CdS@MIL-101(Cr)
Lactic acid
> 420 nm
14.66
-
(Meng et al., 2019)
CD@NH2-UiO-66/g-C3N4
Sodium ascorbate
> 420 nm
–
2.930
(Zhang et al., 2018)
rGO/MOF/Co–Mo–S
TEOA
> 420 nm
0.678
(Liu et al., 2017)
CFB/NH2-MIL-125(Ti)
Triethanolamine
> 420 nm
–
1.123
(Song et al., 2019)
Ni NPs based MOF-5
TEOA
520 nm
3,022
-
(Zhen et al., 2016)
In summary, one of the most important processes in the emerging renewable energy industry is the conversion of solar energy to chemical energy. Photocatalytic water splitting is an excellent way for hydrogen production because it is simple and clean. Just the photocatalyst, water and sunlight are needed to make hydrogen at room temperature. Even though this method of converting solar energy into usable energy has a lot of benefits, making hydrogen via photocatalytic water splitting is really not ready for use so far. This is mostly because the photocatalysts have a very high rate of electron–hole recombination. overall water splitting comprises of both HER and OER reactions. In comparison to HER, OER is slow in terms of kinetics, owing to the elevated overpotential values, with high energy barrier, and also the rate-determining step which involve multiple electrons. Therefore, relative to HER in photocatalytic water splitting, OER growth is limited.
Numerous semiconductor-based photo - catalysts have already been explored and used to produce photocatalytic hydrogen, but MOFs are a new type of photocatalyst because they have high surface area, evident active sites, and can be changed in structure. Innate pores and high specific surface areas make it easier for reactant molecules to adsorb to the active sites. Further, concise active sites are good for studying the photocatalytic reaction mechanism, and number of strategies are reported for the fabrication of various MOF derived photocatalysts where ligand and metal active sites are controlled by varying reaction conditions and using various solvents makes it easier to investigate the effects of different metal water splitting capabilities (Feng et al., 2019).
3 Electrocatalytic water splitting
Electrochemical water splitting has long been regarded as a viable and effective method of transforming water into renewable hydrogen and oxygen. Electrocatalytic water splitting comprises of two half-reactions and might be represented in several ways depending on the reaction considerations (Huang et al., 2018). Further, during the electrocatalytic water splitting process, both the hydrogen evolution reaction and the oxygen evolution reaction are significant half-reactions. Noble metal oxides like platinum for HER and RuO2 for OER have been utilized as potential catalysts, but they have two drawbacks: (i) the high cost and scarcity of materials, which preclude large-scale deployment; and (ii) the loading of catalysts onto glassy carbon, which necessitates the use of binders and additives, which raises contact resistance and degrades electrocatalytic performance. Direct fabrication of non-noble metal compounds, including transition metal complexes, on conductive interfaces, is one typical strategy for overcoming the aforementioned challenges (i.e., copper foams, nickel foams, carbon cloths) (Yang et al., 2021).
Moreover, transition-metal oxides or hydroxides and their derivatives are often the most important active species, resulting in comparatively higher activity and better durability. For electrochemical water splitting a potential of 1.23 V is needed under normal circumstances, equating to an energy input of G = 237.1 kJ/mol (Baig et al., 2021). But to achieve a significant current density, large overpotentials are needed, due to the slow kinetics of the hydrogen evolution reaction and the oxygen evolution reaction and thus results in poor energy conversion efficiencies (Zaman et al., 2021). Consequently, in real electrolyzers, the input potential for water splitting is considerably higher than 1.23 V, therefore high activity suffers from poor explication and the creation of an O = O bond and the loss of four electrons and four protons, resulting in slow kinetics, thermodynamics, and a high overpotential, which is a major bottleneck in the entire water splitting process. Additionally, OER can also be used to generate electrons for carbon dioxide reduction processes for fuel generation. OER is a laborious, two-step process that results in a double bond between two oxygen atoms (O = O), which is hard to activate and sluggish to move (Liu et al., 2018; Anwar et al., 2020).
Different assessment criteria may be used to assess the activity of a water-splitting catalyst. The slope of the Tafel is an essential metric in determining the activity of a catalyst. It's a graph that shows the relationship between the current produced in an electrochemical cell and the electrode potential of a particular metal. In basic terms, the Tafel plot shows an electrode's ability to generate current in accordance with a potential change given to it. It has a relationship between the response rate and the overpotential. Because a lower overpotential is needed to get a large current when the Tafel slope (mV/decade) is low, a lower overpotential is needed to acquire a high current. The Tafel slope for the Pt/C electrode is stated to be 30.3 mV/dec in 0.5 M H2SO4, while the Tafel slope for RuO2 is said to be 89 mV/dec in 1.0 M KOH (Noor et al., 2021).
Overpotential is another important consideration for evaluating a catalyst's potential for water splitting. Overpotential is defined as the potential provided to start the reaction and liberate the entity at the electrodes that is larger than that of the equilibrium voltage. To split water into its constituents, a voltage of at least 1.23 V is necessary, although owing to the complicated reaction kinetics, a significant overpotential is usually required. An ideal catalyst for water splitting must have a low Tafel slope and a large current density. The crystallinity, size, and dimensionality of a catalyst have a big impact on electron transport, which affects the Tafel slope and water-splitting overpotential. As a result, the MOF catalysts have a favorable impact on these assessment criteria. The inclusion of metal centers strengthens the charge transfer characteristics of MOFs (Salahuddin et al., 2021).
Farther, in contrast to HER, OER is strongly dependent on the electrolyte solution pH value and show comparatively different mechanism in alkaline and acidic media (Xiong et al., 2021). As hydrogen and oxygen are generated in acidic medium by oxidizing two water molecules at a potential of 0.404 V. In an alkaline medium, the OH group is converted to oxygen and water at a cost of 1.230 V (vs RHE). As a result of the preceding, it can be concluded that the process operates more efficiently in an alkaline conditions than in an acidic one.
Moreover, one of the primary goals in developing an efficient and cost-effective electrocatalysts for water splitting that substitutes the most active but most expensive noble Pt electrocatalyst (Hu et al., 2019). A variety of non-noble metal- based and carbon-based electrocatalysts have been created based on their outstanding activity, low cost, and abundant availability on the earth, with an emphasis on modifying the aforementioned characteristics, the metallicity of an electrocatalyst is another critical approach that affects the electron density, electrical conductivity, and electron transit between the adsorbate and the catalyst surface (Hu et al., 2020). Therefore, in electrocatalytic water splitting, the goal of using electrocatalysts is to decrease the reaction overpotential and increase the rate of the reaction as often as feasible, thus lowering total amount of energy needed. for alkaline media, transition metal-based catalysts i.e., nickel electrodes for HER and stainless steel composites for OER are usually employed. Besides, for acidic media, noble metal-based catalysts such as platinum for hydrogen evaluation reaction and RuO2 for oxygen evaluation reaction are often required. Further, precious metal-based electrocatalysts and noble metal-free electrocatalysts are prohibitively expensive for large-scale H2 generation from electrolysis of water due to their high cost, limited supply, and low stability (Guan et al., 2017).
The catalyst activity may be efficiently increased by a process known as synergetic interaction between various kinds of materials. It is defined as two or more active components in sequential or supplementary form showing nonlinear cumulative effects of their distinct characteristics. Synergetic effect is defined as the creation of hybrids/composites with enhanced activity and increased accessible active sites as a consequence of combining catalytic material and/or conductive support in the development of water-splitting catalyst. MOF-based catalysts have a greater potential for creating synergetic effects that enhance catalytic activity as it exists among MOF and conducting substrates provide more active sites. One of the key elements influencing the performance of MOF-based composite catalysts and may be engineered for operation at various functioning levels as well. Intra-molecular synergistic effects might be seen in various metal groups of a multi-metallic MOF (Yaqoob et al., 2020).
3.1 Mechanism for HER and OER on MOF-derived electrocatalysts
Currently, electrocatalytic water splitting can be done via MOF derived electrocatalysts in an acidic or alkaline electrolyte as shown in Fig. 10. Overall water splitting comprises of two reactions hydrogen evaluation reaction and oxygen evaluation reaction, HER is considered to be a two-electron transfer process including three probable reaction steps such as Volmer, Heyrovsky, and Tafel reaction and two alternative mechanisms such as Volmer-Heyrovsky or Volmer-Tafel mechanism are involved in acidic or alkaline electrolytes. The potential step reactions are represented by the subsequent equations 1–5, where M denotes electro catalyst active sites. whereas, * denotes reaction intermediates adsorbed on the catalysis sites. Clearly, the way HER works in acidic electrolytes is different from the way it works in alkaline electrolytes. In acidic media, the abundance of protons may facilitate the synthesis of H ∗ during the Volmer reaction, whereas in alkaline media, H ∗ is generated by the adsorption and dissociation of water molecules on catalytic active sites. After then, the adsorbed H ∗ would be either joined with another produced H ∗ to produce H2 molecule by Tafel reaction or an electron transfer reaction with a proton or water molecule to form an H2 via Heyrovsky reaction. Overall, the binding strength between the catalytic site and the electrode is important for the best HER electrocatalysts.
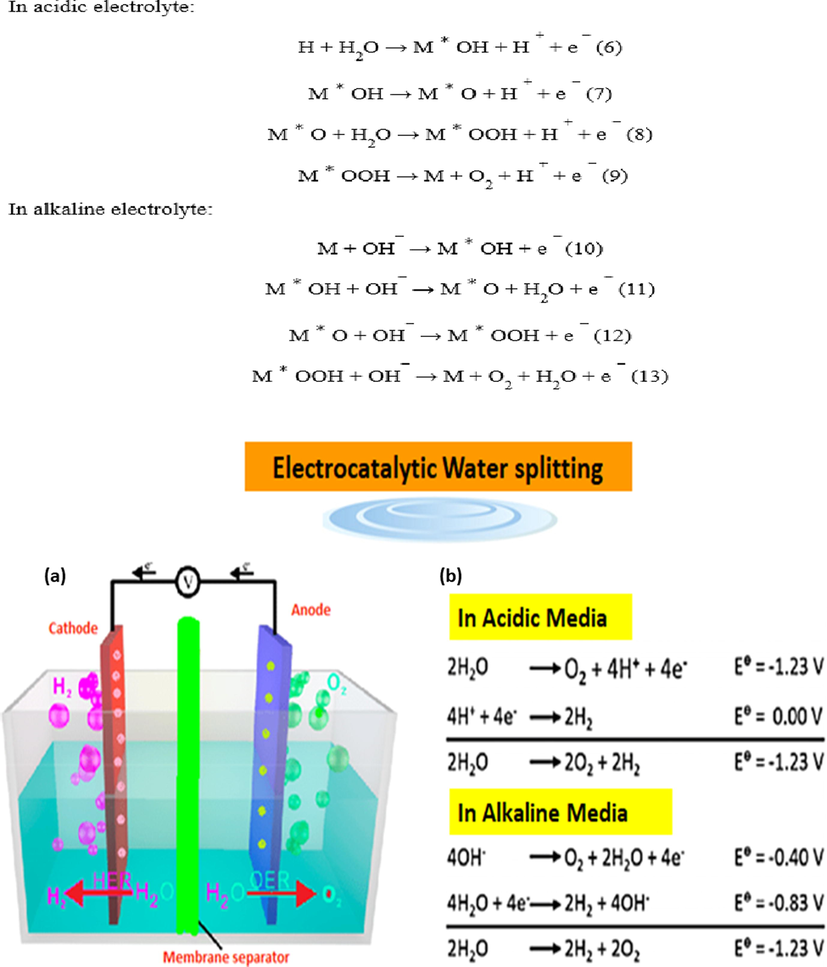
(a) Electrocatalytically splitting of water in cell (b) water splitting reactions in acidic and alkaline media. Exclusive rights 2020 American Chemical Society.
The OER mechanism is of more intricate than that of HER, comprising a four-electron transferring. As illustrated in equations 6–13, in acidic and alkaline or acidic media, hydroxyl ions or water molecules are employed as oxygen-containing reactants. The water molecules or hydroxyl ions are progressively oxidized to oxygen-containing intermediates of ∗ OOH, ∗OH and ∗ O after being adsorbed on the MOF derived electro catalysts active sites, with an electron transfer occurring at each step reaction. Finally, ∗OOH is oxidized further and converted into an oxygen molecule. The above four sequential proton and electron transport stages are thermodynamically advantageous during the oxidation process, which involves a variety of intermediates formation such as ∗ OH, ∗O, and ∗ OOH. The step with the largest energy barrier determines the reaction rate. Due to the large activation energy necessary to break the energy barrier, the OER is kinetically slow. To optimize OER activity, the bonding strength between MOF derived catalysts containing oxygen-based species should be mediated in a systematic manner (Zhang et al., 2020).
In acidic electrolyte:
In alkaline electrolyte:
3.2 Challenges for electrocatalytic water splitting
For electrocatalytic water splitting there are number of challenges that need to be focus such as, (i) HER and OER electrocatalysts are often produced under distinct circumstances, and the current methods for nonprecious catalyst synthesis frequently result in incompatible or complex integration. (ii) hydrogen and oxygen evaluation reactions are tightly linked in traditional water electrolysis, which may result in the production of explosive hydrogen and oxygen gas blends. Not only does the gas crossover create safety concerns, but it also decreases energy proficiency by converting O2 back to water on the cathode side. Meanwhile, the sharing of gas mixtures and electrocatalysts may result in the formation of reactive oxygen species, which can damage the membrane and result in premature device failure. (iii) Due to the fact that OER is a four-electron reaction, it needs a much greater overpotential than HER in order to maintain the same current density, contrary to expectations, the product O2 is not a very useful component of OER. (iv) It remains challenging to procure cost-effective H2 storage and transportation systems utilize on-demand. (v) Hydrogen production from HER in an alkaline medium is the water splitting phase (i.e., the breakage of the strong H–OH bond), which needs extra energy to generate H* intermediates (Xiong et al., 2022).
3.3 Fabrication of MOF derived electrocatalysts
There are several important issues that must be addressed in order to better understand MOF and MOF-derived electrocatalysts water splitting activity. Currently, a number of MOF precursors have been studied individually, generally in distinct dimensions (i.e., 1D, 2D, and 3D). It is broadly accepted that a three-dimensional MOF demonstrates greater stability in terms of shape and structure due to its high porosity but has a lower active site usage. In comparison to the 3D MOFs, other 1D/2D MOF has a higher usage of active sites but is less stable in terms of structure and morphology with low porosity (Li et al., 2021). As a result, combining the benefits of the 3D MOF with those of the 1D/2D MOF is very desirable, although it remains a difficult task for researchers (Aneeshkumar et al., 2021; Tang et al., 2022). Second, MOF and MOF-based composites work better as an electrocatalyst for water splitting, they are readily collapsed, fused, and aggregated at high temperatures, reducing the accessibility of the catalyst active sites, and reducing its mass transfer capacities. As a consequence, their electrocatalytic performance deteriorates.
In order to address these difficulties, a lower temperature treatment is essential, but low temperature carbonization treatment, will result in poor electrical conductivity owing to the low degree of graphitization of the composite. As a result, finding the right temperature to achieve an optimal balance among particle distribution, graphitization degree and surface assembly in MOF-based composite systems remains a challenge (Zaman et al., 2021). Besides, most papers seldom explore the stability of MOF-based catalysts in real-world applications and disregard the degradation process. As a result, a concerted effort must be made to understand the foundations of degrading activity, as well as the development of MOF-based electrocatalysts that display high electrocatalytic activity and long-term stability across a variety of working conditions (Song et al., 2019).
In this regards, various MOF-derived catalysts have already been reported recently, including transition metal oxides, sulphides, selenides, phosphides, carbides, and more. During the fabrication of MOF derived electrocatalysts, MOFs are always used as sacrificial templates; when they're pyrolyzed, many different types of carbon nanostructure, metal oxides, metal composites, and metal carbides and nitrides can be formed and can be functionalized during, before, or after pyrolysis (Bavykina et al., 2020). It has also been noticed that heteroatom doping with nitrogen (N), phosphorous (P), boron (B), etc., increases catalytic responsiveness exponentially in carbon-based metal-free and non-precious M/C hybrid electrocatalyst systems. These MOF derivatives greatly outperform direct-MOF electrocatalysts in terms of application, avoiding some major limitations. By and large, prepared MOFs have limited robustness in strongly acidic or alkaline aqueous electrolytes, which are commonly used in electrochemical systems, whereas derivatives of MOFs exhibit significantly greater robustness under severe working conditions (Huang et al., 2019).
Further, from earlier studies general fabrication procedures can be divided into three categories: (i) in situ nitrogen doping, using MOFs containing nitrogen such as zeolitic imidazolate frameworks (ZIFs); (ii) ex situ nitrogen doping using a gas guest molecule, such as ammonia; and (iii) using a second solid material as a nitrogen source mixed with MOFs. Further, metal organic frameworks can also be used as an immediate precursor to make heteroatom-doped porous carbon electrocatalysts, also used as templates and nitrogen-doped carbon can be synthesized during the structural precursor transition for MOFs carrying nitrogen (Zhou et al., 2015; Wang et al., 2020). So, accessibility of homogeneous dispersed catalytic centers and dense active sites it can be employed as an effective precursors for carbon-based catalysts and number of ways are reported in literature discussed as follows:
3.3.1 Fabrication of MOF derived electrocatalysts via pyrolysis
MOFs used as sacrificial precursors is a common method for obtaining carbon based substances by pyrolyzing MOFs in gaseous environment. During pyrolysis organic ligands of MOFs can be transformed into carbon based materials such as amorphous carbon, or carbon nanotubes in the final composite electrocatalysts and the relevant metal-based nanoparticles such as metals, alloys, or metal-based compounds can be formed from metal nodes. The obtained carbon substrate can inhibit metal-based nanoparticles from further aggregating (Zhang et al., 2020). Further, adjusting MOFs and pyrolysis conditions can influence the electrocatalytic performance of pyrolysis products. The porous structure of the original MOFs can be partially inherited by the MOF-derived catalyst. Although the specific surface area and pore volume of the derivatives may be compromised during the reaction as the porous structure get collapsed, they can still perform well as catalysts. Likewise, researchers may also regulate the morphology and size of the resulting catalysts using this approach, making it possible to analyze the active regions of as-prepared catalysts (Han et al., 2021). In addition, bimetallic MOF materials have also been used as a successful approach to create effective catalysts via direct carbonization, to circumvent the atom type limitation of a MOF precursor. Because of the synergetic impact of bimetal doping, the efficiency of bimetallic MOF electrocatalysts is usually substantially higher than that of single atom MOF-derived ones (Radwan et al., 2021; Guan et al., 2021).
3.3.2 Fabrication of heteroatom dopped MOF derived electrocatalysts via pyrolysis
Another way of fabricating MOF derived electrocatalysts is doping with outside heteroatom such as nonmetal sources (N, P, S, and C) and/or metal sources are used as external heteroatom doping sources (Fe, Co, Cu, etc.), might be created strategically that act as an additional heteroatom resource into MOFs as precursors (Yang et al., 2012). Besides, thiourea, urea, cyanamide, dimethyl sulfoxide, melamine, and others are among the secondary nonmetal dopants. Furthermore, the charge density of carbon materials can also be successfully adapted by combining it with highly different electronegativity nonmetal atoms inside carbon frameworks. Thus metal doping would result in a bare active site that is free of aggregation, accordingly enhance the overall catalytic activity (Choi et al., 2012).
3.3.3 Fabrication of MOF derived electrocatalysts from MOF based carbon composites via pyrolysis
Carbon electrocatalysts derived from MOF precursors at an elevated temperatures are well known for having variety of problems such as skeleton distortion and porosity loss occur at temperatures equal or above than 700 °C. Surprisingly, the composites made from MOF-based precursors, such as MOF/carbon nanotubes and MOF/graphene, can overcome the challenges of producing highly effective catalysts and could effectively prevent the MOF skeleton material from breaking down, implying that porous carbon composite catalysts derived from MOF can preserve high surface area and porosity, resulting in an improved conductivity and graphitization degree of the produced materials. As a result, this approach considerably improves the catalysts' catalytic efficiency (Yang et al., 2018; Gong et al., 2009). Fig. 11 shows the possible ways to fabricate and tailor MOFs and MOF-based materials for overall water splitting.
Schematic diagram for possible ways to fabricate and tailor MOFs and MOF-based materials for overall water splitting (Zhang et al., 2021) Exclusive rights 2021 Wiley Online Library.
3.4 MOF derived carbon based electrocatalysts for water splitting
Abundant research work have now been acknowledged for developing many forms of micro and mesoporous materials; among them, MOFs outperformed other materials in terms of structure, surface morphology, and electrical characteristics. Additionally, it exhibits exceptional cycling and thermal stability, enabling them to precisely regulate the porous structure and nanostructure shape of their nanoparticles. All of these physicochemical features have received considerable interest over the last two decades due to their potential utility in a broad variety of electrochemical applications. MOFs are compelling options because they are well-suited to the requirements of next-generation energy storage systems as they have a substantially larger specific surface area (SSA) than conventional nanomaterials, over 10,000 m2 g−1.
When contrasted to certain other electrocatalysts, MOFs offers number of benefits such as i) MOFs exhibit both heterogeneous and homogeneous catalytic characteristics and considered as an attractive platforms for studying catalytic processes because of their perfect crystalline structure at molecular level and highly porous state. ii) It also exhibits superior catalytic performance, improved chemical stability, and simpler recyclability than other electro catalysts due to overall active sites availability for catalytic action, hence boosting the electrocatalytic efficiency. iii) Metal ion dopants or functionalizing an organic ligands may readily change the optical and electrical structure of MOFs without modifying their crystalline structure. iv) Further, MOFs can have many functional sites, that can be used for electrocatalytic overall water splitting (Sun et al., 2022; Tong et al., 2020). v) MOFs have adjustable porous structure and pore environment, allowing for improved selectivity of intended products, particularly in catalytic organic fabrication processes. vi) MOFs may readily be combined with other nanomaterials in enlarged pores or even on the surface. Further, MOF based composites offer the benefit of large intrinsic activity, improved conductivity and charge transport mechanism, reduced nanoparticle aggregation, and a potential synergistic interfacial impact to boost catalytic activity in comparison to every component (Huang et al., 2021).
As MOFs have shown to be good sacrificial templates for MOF-derived materials due to its unique structural and compositional properties. Various carbon–metal, nanostructured porous carbons and metal-oxide composites can be made by varying the synthesis conditions during MOF transformation which are extensively studied in electrochemical water splitting, subsequently certain particular components in them are excellent electrocatalysts for HER and OER. Furthermore, creating unique morphologies (nano cubes, nanorods, nanotubes, nanosheets) and growing them directly on conductive precursors (Ti foil, Ni Foam, carbon paper, Cu Foam, carbon clothes, and so on) can dramatically improve conductivity and charge/mass transfer efficiency, thereby improving water splitting efficiency. However, the process of converting MOFs to MOF-derived materials is still unknown, and the mechanism of MOFs for water splitting is seldom addressed since researchers work to simplify models into useful catalysts that have already been well investigated.
Noticeably, for researchers to develop and manufacture cost-effective high-activity, MOF-derived catalysts for overall water splitting is a crucial challenge. Unfortunately, an efficient HER catalyst may not be able to effectively catalyze OER and vice versa in an acidic/alkaline electrolyte, in spite of the comparable essential parameters used to assess the water splitting performance of those catalysts are similar to those used to evaluate the performance of HER or OER.
3.4.1 MOF-derived carbon materials
A wide range of practices are possible for carbon based materials with various astonishing properties such as high specific surface area, electrical conductivity, and chemical stability. Chemical stability and superior conductivity are typically found in doped carbon materials derived from MOFs with diverse nanostructures. Some MOFs with nitrogen-containing ligands, such as zeolitic imidazolate frameworks, can be annealed in inert gases to produce N-doped carbons (Chen et al., 2021; Chai et al., 2019).
In recent years, carbon materials with heteroatomic dopants (i.e., boron or nitrogen or phosphorous or sulphur), exhibits high conductivity are considered as good at catalysis and have been getting more attention day by day. Recently, MOFs have been considered as good templates for generating carbon nanomaterials due to its huge surface area, good electrical conductivity, and low cost strategy (Wang et al., 2019). Heteroatom-containing MOFs can be exploited as precursors to create metal-free carbon-based electrocatalysts through carbonization. Lei et al. revealed that MOF derived N and O doped carbon materials might be exploited for the electrochemical splitting of water (Lei et al., 2018). This bifunctional catalyst was produced by calcinating the ZIF-8 precursor, accompanied by the catalytic sites electrochemical activation. Among all prepared series of carbon materials such as (ZIF-8 C2-C8) Six hours CPT (C6) carbon catalysts exhibits an astonishing HER activity in 0.5 M H2SO4 electrolyte and display current density of 0.063 mA/cm2 with overpotential of 155 mV and Tafel slope of 54.7 mV/dec as demonstrated in Fig. 12 (b, e). Further, four hours CPT (C4) carbon catalysts exhibits a surprising OER activity in 0.1 M KOH electrolyte and display overpotential of 476 mV and Tafel slope of 78.5 mV/dec as demonstrated in Fig. 12 (c, d).
(a) schematic illustration of carbon electrocatalyst preparation from ZIF-8 (b) electrochemical testing in 0.5 M H2SO4 for HER and (c, d) electrochemical testing in 0.1 M KOH for OER (Zhang et al., 2021) Exclusive rights 2021 Wiley Online Library.
Calcination of MOFs with particular atmospheres, such as NH3, PH3 and H2S gases, can also be used to make carbonaceous materials with heteroatomic dopant. Liu et al. reported an electrocatalyst (Co2P/CoNPC) with N- and P-doping produced by calcining MOF precursors in a PH3 atmosphere. Co2P/CoNPC exhibits an improved OER activity in 1 M KOH electrolyte display overpotential of 326 mV at current density of 10 mA/cm2 and Tafel slope of 72.6 mV/dec as demonstrated in Fig. 13 (b, c). Further, Co2P/CoNPC also exhibits an improved HER activity in 0.1 M KOH electrolyte display overpotential of 208 mV and Tafel slope of 83.9 mV/dec as demonstrated in Fig. 13 (c, d) (Liu et al., 2020).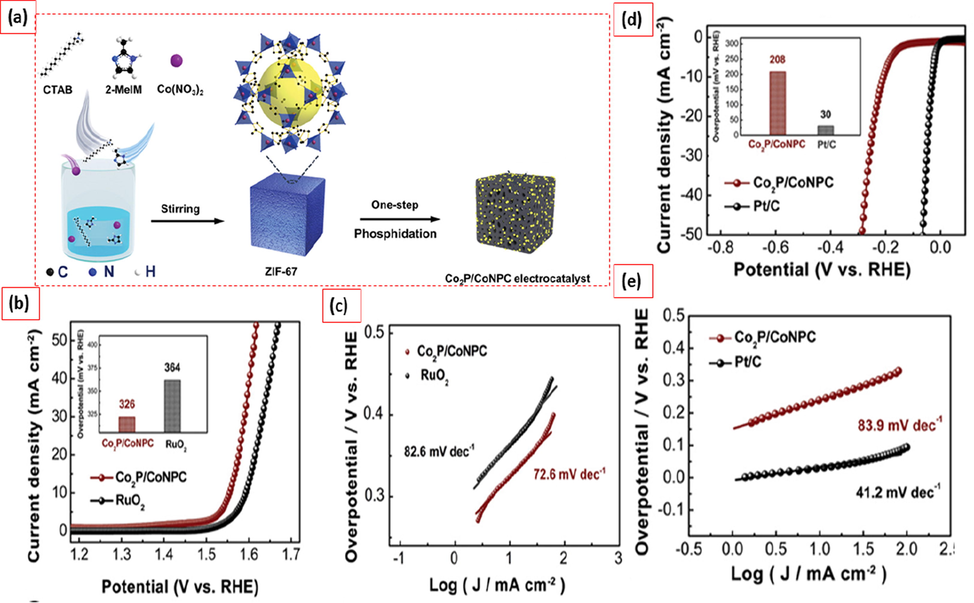
(a) schematic illustration of phosphorous based carbon electrocatalyst preparation from ZIF-67 (b, c) electrochemical testing in 0.1 M KOH for OER and (c, d) electrochemical testing in 0.1 M KOH for HER (Liu et al., 2020) Exclusive rights 2021 Wiley Online Library.
In recent years, novel three-dimensional carbon structures derived from ZIFs such as hollow polyhedrons, carbon nanotube polyhedrons, and carbon nanotube have been reported, resulting in increased surface area, rapid charge and mass movement, and enhanced water splitting electrocatalytic activity. Pan et al. reported a new water splitting electrocatalyst NCNHP (N-doped carbon nanotube hollow polyhedron) and CoP/NCNHP (CoP nanoparticles embedded in an N-doped carbon nanotube hollow polyhedron) produced from core-shell ZIF-8@ZIF-67 and it exhibits overpotentials of 310 mV and −115 mV at 10 mA cm−2 and Tafel slopes of 70 mV dec-1 and −66 mV dec-1 for OER and HER in 1 M KOH, accordingly, the catalyst demonstrated outstanding bifunctional electrocatalytic efficacy. It was ascribed to the NCNHP matrix, and the synergistic impact between CoP and NCNHP, which increased the charge and mass transfer efficiency, and electrolyte stability and achieved a current density of 10 mA cm−2 at 1.64 V, exceeding the majority of reported non-noble-metal electrocatalysts, and displayed remarkable stability for at least 36 h as illustrated in Fig. 14 (Pan et al., 2018).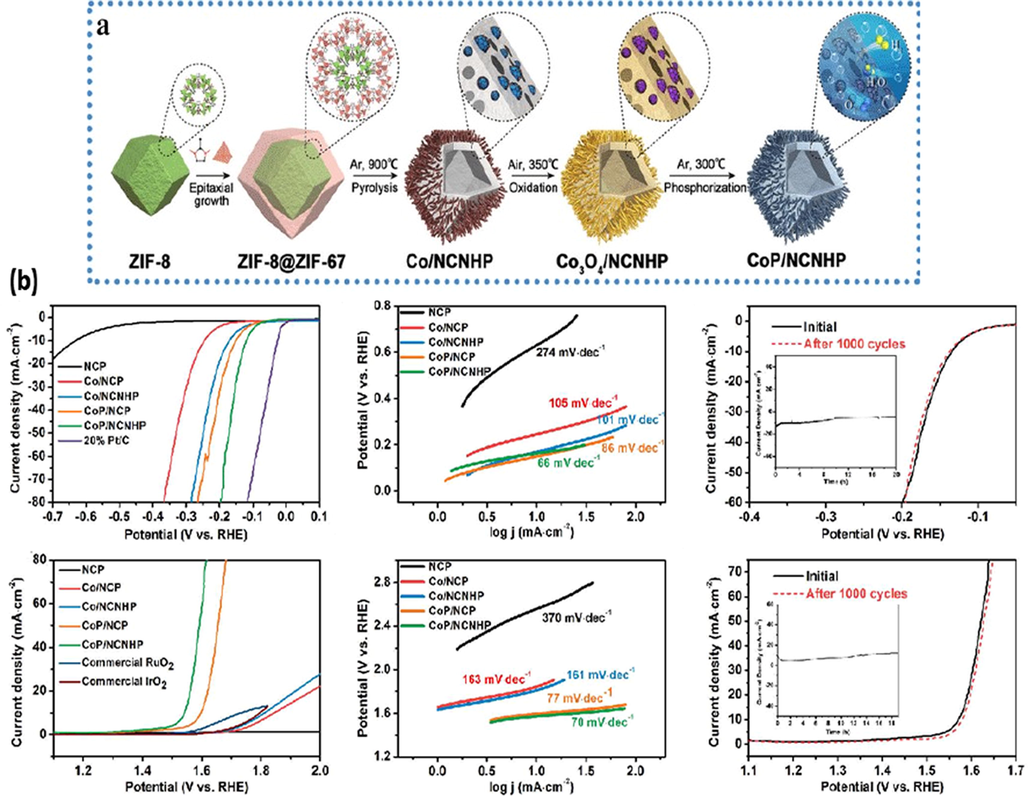
(a) Two step synthesis of CoP/NCNHP and (b) its electrochemical results for overall water splitting (Pan et al., 2018)Exclusive rights 2018 ACS publications.
3.4.2 MOF-derived single-atom materials
Single-atom catalysts have been getting a lot of consideration in the field of catalysis because of their extraordinary catalytic activity, ability to use the most metal atoms and specific selectivity (Mitchell et al., 2018; Zhang and Guan, 2022). However, due to the high surface energy of monatomic catalysts, single atoms may easily move and aggregate into nanoparticles under actual reaction circumstances. To solve this problem, MOFs have become a promising precursor for developing SACs because of their porous structure and availability of different components from which it can be made exactly how they should be. Direct pyrolysis of MOFs is a simple way to make SACs (Yang et al., 2018; Zhang et al., 2019). Further, the N-doped carbon also obtained from the MOF pyrolysis is a good scaffold for metal ions to be absorbed. Li et al. reported a simple way to make atom-distributed Fe–N4 active sites that are embedded in carbon phases that are made by carbonizing ZIF-8 precursors (Li et al., 2019).
Further, Fan et al. recently reported Ni-based SACs (A-Ni-C). Fig. 15a shows schematic illustration for the synthesis of A-Ni-C, where the carbonization of a Ni-MOF was followed by HCl etching and then electrochemical activation led to the formation of A-Ni-C. single atom catalyst such as A-Ni-C exhibits a surprising HER activity in 0.5 M H2SO4 electrolyte display current density of 10 mA/cm2, overpotential of −34 mV and Tafel slope of 41 mV/dec as demonstrated in Fig. 15 (b, c) (Fan et al., 2016). Furthermore, selecting appropriate MOF precursors is a familiar strategy for controlling the chemical properties of MOF derivatives. Controlling the chemical composition of the electrocatalysts can also be done by manipulating the conversion conditions and adding additional components. In most of the cases, these approaches are used in combination to obtain effective electrocatalytic activity and stability.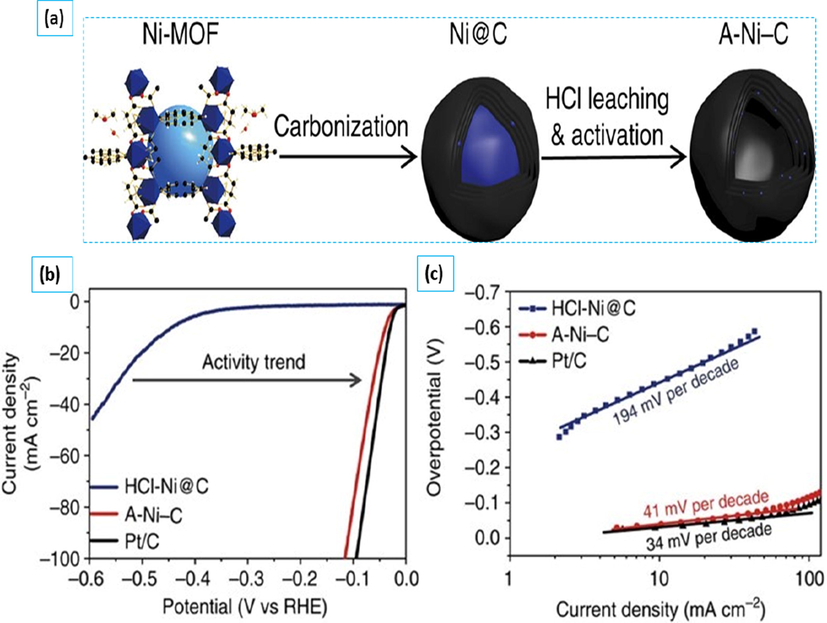
(a) schematic illustration of single atom electrocatalyst (A-Ni-C) preparation from Ni-MOF (b, c) electrochemical testing in 0.5 M H2SO4 for HER (Fan et al., 2016) Exclusive rights 2016 Springer Nature.
Zeolitic imidazolate frameworks are the best scaffolds that can be used to make different types of water splitting electrocatalysts by trying to control morphology structures and chemical constituents. In 2015, you et al. reported porous Co-P/NC nano-polyhedrons made of nanoparticles of CoPx (a mixture of CoP and Co2P) that were integrated in N-doped carbon by direct carbonizing ZIF-67 and then phosphating them. These nanoparticles were used as electrocatalysts for overall water splitting. These nano-polyhedrons had a surface area of 183 m2 g−1 and large mesopores in which CoPx (about 33 nm) particles were dispersed and encased in carbon matrixes, as shown in the Fig. 16. CoP/NC nano polyhedrons with the precise number of pores were very good electrocatalysts for both HER and OER in 1.0 M KOH. They had low overpotentials, which meant they could get 10 mA cm−2 at low overpotentials for OER and HER, respectively. It also worked better than the Pt/IrO2 couple (165 mA cm−2 at 2.0 V) and was very stable over 24 h of electrolysis (You et al., 2015).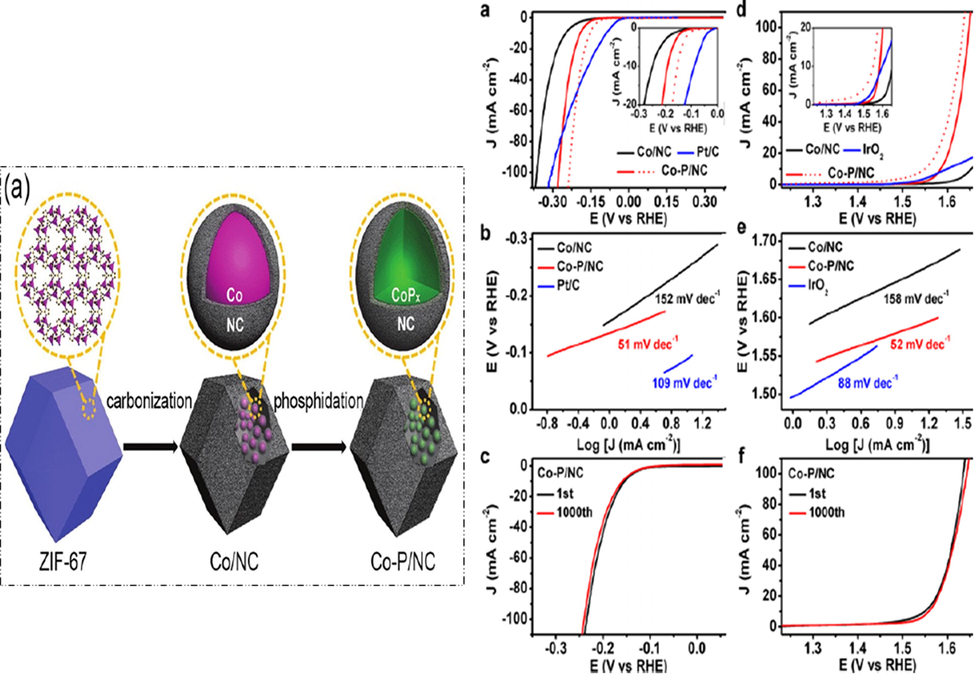
(a) Synthetic scheme of Co-P/NC, (b) Tafel Plots comparison (You et al., 2015). Exclusive rights 2015 American Chemical Society.
3.4.3 MOF-derived composites
Fabrication of MOF derived composites is an alternative efficient approach for improving MOF overall electrocatalytic performance, mechanical characteristics, and stability. Although pure MOFs have a broad variety of structural capabilities, but its composites with other carbon based materials such as graphene oxide reduced graphene oxides and etc. resulting in a distinctive electrochemical activity of the resulting electrocatalysts due to a potential synergic effect. Further, it lowers the aggregation in chemical reactions, increased structural stability with much more catalyst active sites and improved mass transfer are all benefits of MOF-based composite materials (Zhou et al., 2022).
Gopi et al. in 2021 reported an efficient bifunctional electrocatalyst for water splitting, such as graphene oxide based V dopped Nix Fey MOF/GO composites with different ratio of Ni and Fe i.e., V-Ni0.06 Fe0.03 MOF/GO, V-Ni0.06 Fe0.06 MOF/GO, V-Ni0.06 Fe0.09 MOF/GO. Among them, V-Ni0.06Fe0.06 MOF/GO showed superb electrocatalysis for both oxygen and hydrogen evaluation reactions and exhibited superior durability in alkaline, and acidic media. In case of OER, the catalyst V-Ni0.06 Fe0.06 MOF/GO exhibits excellent electrocatalytic activity in 1.0 M KOH i.e., show current density of 579 mA/cm2 with overpotential value of 210 mV, in addition, it exhibits the lowest onset potential value (i.e.,1.44 mV) with smaller value of Tafel slope 97 mV/dec, in comparison to all other prepared V doped V-Nix Fey MOF/GO these values are higher as shown in Fig. 17 a, b. Moreover, for HER the electrocatalytic study is performed in acidic media (0.5 M H2SO4), Like OER the catalyst V-Ni0.06 Fe0.06 MOF/GO shows an admirable performance as well i.e., show current density of 208 mA/ cm2, onset potential of 90 mV to reach stable current density of 10 mA/cm2, and over potential is 130 mV, with smaller Tafel slop value 150 mV/dec. Furthermore, Fig. 17 c shows the comparison of Tafel slope and over potential values, and Fig. 17 d illustrates the overall water splitting by corresponding electrocatalysts (Gopi et al., 2021).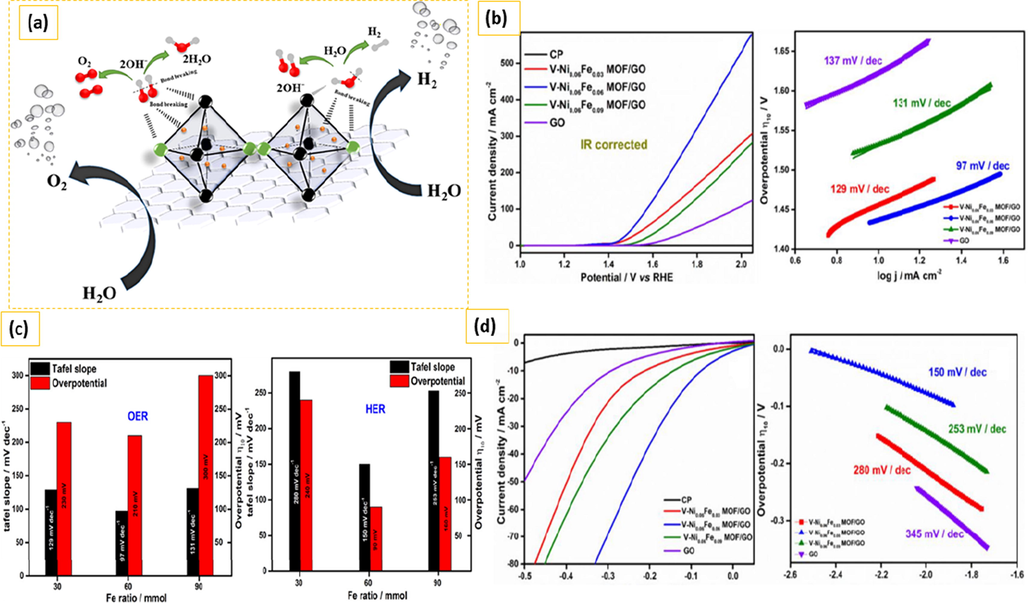
(a) OER LSV and Tafel slopes (b) HER LSV and Tafel slopes (c) comparison of Tafel slope and over potential values, (d) overall water splitting by corresponding electrocatalysts (Gopi et al., 2021). Exclusive rights 2021 ELSEVIER.
In addition, Hu et al. in 2019 reported Co/CoN/Co2P-NPC a ternary metallic carbon based composites for electrocatalytic water splitting and for comparison an electrocatalyst i.e., Co/CoN-NC is also studied. The resulting Co/CoN/ Co2P-NPC exhibits good OER and HER electrocatalytic activity in alkaline solution due to high carbon content of MOFs precursors. A standard three-electrode setup in 1.0 M KOH electrolyte was used to evaluate the electrocatalytic performance of the corresponding prepared catalysts. In comparison to Co/CoN-NC, Co/CoN/ Co2P-NPC exhibits astonishing HER and OER performance. As shown in Fig. 18 a LSV results for OER shows the overpotential 272 mV is needed to achieve the current density of 10 mA/ cm2 for Co/CoN/Co2P-NPC in contrast to Co/CoN-NC needed 306 mV of over potential. Further, Tafel slope value of 62 mV/dec for Co/CoN/Co2P-NPC is lower than Co/CoN-NC (i.e., 76 mV/dec) as illustrated in Fig. 18b. While, for HER LSV results from Fig. 18a Co/CoN/Co2P-NPC show the lower value of overpotential 99 mV is needed to achieve the current density of 10 mA/ cm2 and in case of Co/CoN-NC needs higher overpotential value of 143 mV. Further, for HER the Tafel slope value of 51 mV/dec for Co/CoN/Co2P-NPC is lower than Co/CoN-NC (i.e., 55 mV/dec) as shown in Fig. 18 c (Hu et al., 2019).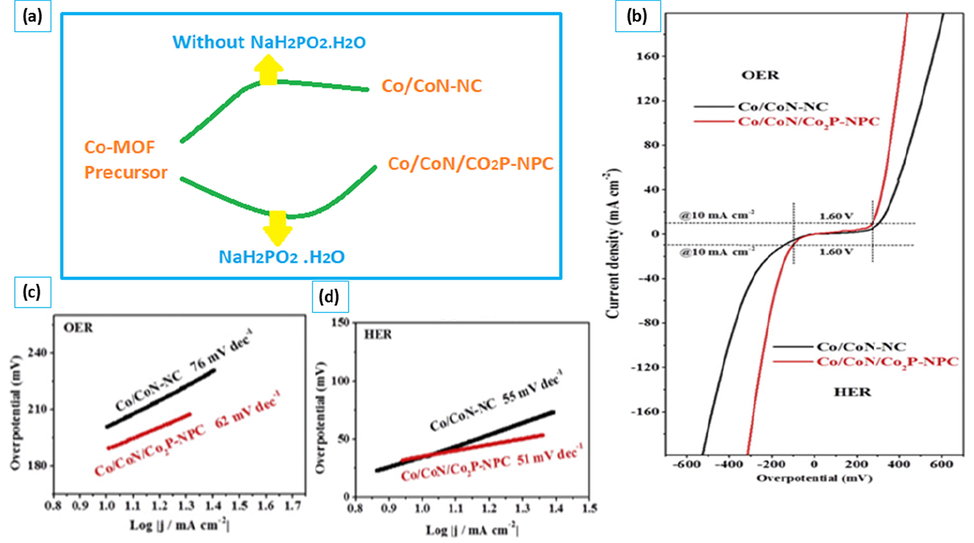
Co/CoN-NC and Co/CoN/Co2P-NPC (a) LSV curves for HER and OER, (b) Tafel plots for OER and (c) Tafel plots for HER (Hu et al., 2019). Exclusive rights 2019 ELSEVIER.
Table 3 and 4 shows comparison study of MOF based electrocatalysts for HER and OER.
Electrocatalysts
Electrolyte
Tafel slope (mV dec−1)
Overpotential (mv)
Ref.
CTGU-5
0.5 M H2 SO4
−125
−388
(Wu et al., 2017)
CoP/rGO-400
1 M KOH
−38
−150
(Jiao et al., 2016)
Co-Ni-Se/C/NF
1 M KOH
−81
−90
(Ming et al., 2016)
CTGU-6
0.5 M H2 SO4
−176
−425
(Wu et al., 2017)
PNC/Co
1 M KOH
−131
−298
(Li et al., 2016)
AB&CTGU-5
0.5 M H2 SO4
−45
−44
(Wu et al., 2017)
UiO-66-NH2-Mo-5
0.5 M H2 SO4
−59
−200
(Dai et al., 2016)
CoSe2@C cages
0.5 M H2SO4
−51
−43
(Kwak et al., 2016)
NENU-500
0.5 M H2 SO4
−96
−237
(Qin et al., 2015)
NENU-501
0.5 M H2 SO4
−137
−392
(Qin et al., 2015)
Co0.85Se@NC
1 M KOH
−125
−230
(Meng et al., 2017)
HUST-201
0.5 M H2 SO4
−79
−192
(Zhang et al., 2018)
Mo-N/C@MoS2
1 M KOH
−64
−117
(Amiinu et al., 2017)
HUST-200
0.5 M H2 SO4
−51
−131
(Zhang et al., 2018)
(GO 8 wt%) Cu‐MOF composite
84
209
(Jahan et al., 2013)
ZIF@LDH@NF-600
1 M KOH
−109
−106
(Jadhav et al., 2021)
Zn0.30Co2.70S4 polyhedra
0.5 M H2SO4
47.5
80
(Huang et al., 2016)
Y − S Co0.85Se/CFP
1 M KOH
−72
−250
(Ao et al., 2018)
CoP CPHs
0.5 M H2SO4
51
133
(Xu et al., 2015)
3D-CNTA
1 M KOH
−135
−185
(Ren et al., 2021)
MoCx nano‐octahedrons
0.5 M H2SO4
53
142
(Wu et al., 2015)
NF@Fe2-Ni2P/C
1 M KOH
30
−39
(Cai et al., 2022)
MoS2/3D‐NPC
0.5 M H2SO4
51
210
(Liu et al., 2015)
Cu0.3Co2.7P/NC
1 M KOH
−120
−220
(Song et al., 2017)
Au@Zn − Fe − C
0.5 M H2SO4
130
123
(Lu et al., 2016)
Layered CoP/rGO composite
0.5 M H2SO4
50
105
(Jiao et al., 2016)
Ni-Fe-P@C NRs
1 M KOH
−93
−79
(Ahn and Manthiram, 2017)
Ni@graphene
1 M KOH
−120
−240
(Kakaei and Ostadi, 2020)
NiFeP
1.0 M KOH
−69
−178
(Du et al., 2019)
Fe3C/Mo2C@NPGC
0.5 M H2 SO4
−45
−98
(Li et al., 2016)
Ni@CoO@CoNC
1 M KOH
−98
−190
(Li et al., 2022)
Cu3P@NPPC-650
0.5 M H2 SO4
−76
−89
(Wang et al., 2018)
MoS2/3D-NPC
0.5 M H2 SO4
−51
−210
(Liu et al., 2015)
Ni2P/C
0.5 M H2 SO4
−113
−198
(He et al., 2018)
Cu-Co9S8
1 M KOH
−132
−62
(Du et al., 2019)
CoP-CNTs
0.5 M H2 SO4
−52
−139
(Wu et al., 2017)
Ni-MoCx -C (HC800)
1.0 M KOH
−83
−123
(Sun et al., 2017)
CoS2 NTA/CC
1 M KOH
−88
−193
(Lu et al., 2020)
Mo2C/C
1.0 M KOH
−64
−165
(Qamar et al., 2016)
Co/Co9S8@ SNGS1000
0.1 M KOH
−96
−350
(Zhang et al., 2016)
A-Ni-C
0.5 M H2 SO4
−41
−34
(Fan et al., 2016)
Zn0.1Co0.9Se2
0.5 M H2SO4
−50
−140
(Wang et al., 2017)
Cr0.6Ru0.4O2
0.5 M H2 SO4
58
178
(Lin et al., 2019)
NGO/Ni7S6
1 M KOH
−146
−370
(Jayaramulu et al., 2017)
CoP@BCN-1
1.0 M KOH
−52
−215
(Tabassum et al., 2017)
Co@NCNT
0.5 M H2SO4
−93
−210
(Tang et al., 2021)
CuCo@NC
0.5 M H2 SO4
−79
−145
(Kuang et al., 2017)
Cu3P@NPPC-650
0.5 M H2 SO4
−76
−89
(Wang et al., 2018)
Co-NC/CNT-800
1 M KOH
−125
−203
(Zhang et al., 2016)
Ni foam, SS mesh
1 M KOH
130, 51
1.74 V, 0.277
(Hu et al., 2019)
NSPC −1000
0.5 M H2SO4
172
(Liu et al., 2019)
CoSe2/CF
1 M KOH
−52
−95
(Ma et al., 2018)
Electrocatalyst
Electrolyte
OER
Ref.
Tafel mV/ dec
Overpotential (η10) mV
Fe:2D-Co-NS@Ni
0.1 M KOH
46
211
(Chen et al., 2021)
NiFe-NFF
1 M KOH
38.9
227
(Qin et al., 2021)
CoP/rGO-400
1 M KOH
66
340
(Jiao et al., 2016)
CoSe2
1 M KOH
79
330
(Liu et al., 2017)
PNC/Co
1 M KOH
76
370
(Li et al., 2016)
Co-Ni-Se/C/NF
1 M KOH
63
275
(Ming et al., 2016)
CeOx/CoS
1 M KOH
50
269
(Xu et al., 2019)
NCNTFs
1 M KOH
93
370.
(Wang et al., 2017)
CoSe2/CF
1 M KOH
41
297
(Ma et al., 2018)
Fe3-Co2
0.1 M KOH
48
225
(Li et al., 2021)
Co-NC/CNT-800
1 M KOH
78
314
(Zhang et al., 2016)
Co@NCNT
1 M KOH OER
116
429
(Tang et al., 2021)
Co/Co9S8@ SNGS1000
0.1 M KOH
80
290
(Zhang et al., 2016)
Ni MOF
1 M KOH
64
346
(Maruthapandian et al., 2018)
Cu-Co9S8
1 M KOH
50
260
(Du et al., 2019)
Ni-Fe-P@C NRs
1 M KOH
40
217
(Ahn and Manthiram, 2017)
Ni-MOF@Fe-MOF
1 M KOH
82
265
(Cheng et al., 2021)
NF@Fe2-Ni2P/C
1 M KOH
52
205
(Cai et al., 2022)
Ni@graphene
1 M KOH
66
370
(Kakaei and Ostadi, 2020)
Co3S4/EC-MOF
1 M KOH
120
226
(Liu et al., 2019)
Mo-N/C@MoS2
0.1 M KOH
72
390
(Amiinu et al., 2017)
Cu-doped RuO2
0.5 M H2SO4
43.96
188
(Li et al., 2021)
Ni@CoO@CoNC
1 M KOH
53
309
(Li et al., 2022)
CoSe2@C cages
1 M KOH
83
200
(Kwak et al., 2016)
FeCoNi
1 M KOH
60
325
(Xu et al., 2018)
CoS2 NTA/CC
1 M KOH
81
276
(Lu et al., 2020)
Cr0.6Ru0.4O2
0.5 M H2SO4
58
178
(Li et al., 2021)
NGO/Ni7S6
0.1 M KOH
45
380
(Jayaramulu et al., 2017)
CoFe-MOF-OH
1 M KOH
44
310
(Zou et al., 2019)
Co0.85Se@NC
1 M KOH
75
320
(Meng et al., 2017)
NGO/Ni7S6
0.1 M KOH
45.4
380
(Stelmachowski et al., 2021)
Cu0.3 Co2.7P/NC
1 M KOH
44
190
(Song et al., 2017)
NixCo3−x O4− y
1 M KOH
53
320
(Antony et al., 2016)
ZIF@LDH@NF-600
1 M KOH
97
318
(Jadhav et al., 2021)
3D-CNTA
1 M KOH
89
360
(Ren et al., 2021)
NiFe MOF/OM-NFH
1 M KOH
123
270
(Li et al., 2019)
Zn0.1Co0.9Se2
1 M KOH
43
340
(Wang et al., 2017)
Ni-Fe-MOF NSs
1 M KOH
56.0
221
(Li et al., 2019)
Y − S Co0.85Se/CFP
1 M KOH
87
300
(Ao et al., 2018)
In summary, electrocatalytic water splitting comprises of hydrogen evolution reaction and the oxygen evolution reaction of water electrolysis produce hydrogen and oxygen from water. Water is split into gaseous oxygen, together with protons and electrons, at the anode. The protons and electrons recombine to form hydrogen gas at the cathode. By far, the energy conversion technology has piqued the interest of many people owing to its high conversion efficiency, low environmental impact, and potentially broad variety of applications (Yagi and Kaneko, 2001). Various MOF based/ derived electrocatalysts are fabricated for electrocatalytic water splitting, include encapsulating metal NPs in carbon nanotubes or carbon shells. As a precursor to the fabrication of core–shell structured hybrid electrocatalysts, MOFs might be used as a latest research path to create effective water splitting MOF-based catalysts. Since we know even less about the transformation process, it is difficult to regulate exactly how these materials will change morphology. Clarity on this problem is critical for the development of large surface area, regular pore structure, and adjustable composition MOF nanostructures for electrochemical water splitting processes (Wang et al., 2017). Table 5 shows the comparison of MOF and MOF derived electrocatalysts for overall water splitting.
Catalyst
Electrolyte
OER
HER
Ref.
Tafel mV/ dec
Overpotential (η10) mV
Tafel mV/ dec
Overpotential (η10) mV
V-Ni0.06Fe0.06 MOF/GO
1 M KOH
97
210
150
90
(Zhang et al., 2021)
CoP/rGO
1 M KOH
66
272
46
107
(Zhang et al., 2021)
NiFeP/CC
1 M KOH
147
425
112
109
(Roger et al., 2017)
Co5.47N NP@N-PC
1 M KOH
54
248
86
149
(Chen et al., 2018)
CoP-NS/C
–
64
292
59
140
(Liu et al., 2017)
CoP NA/CC
–
58
52
58
52
(Liu et al., 2017)
Ni0.9Fe0.1/NC
1 M KOH
45
330
111
231
(Du et al., 2018)
CO2P/CNT
–
68
292
103
132
(Lu et al., 2021)
FeNi@N-CNT (OER)Ni-NPs@NC
(HER)1 M KOH
47.7
300
68
77
(Tao et al., 2016)
Ni2P-CoP
–
64–69
320
64
105
(Liang et al., 2017)
Ni2P/rGO
–
62
142
58
260
(Liang et al., 2017)
NiCOP/CC
–
64
242
68
62
(Du et al., 2017)
Fe-Ni@NC-CNT
1 M KOH
45.47
274
113
202
(Anantharaj et al., 2016)
Co-NCNTFs//NF
1 M KOH
–
141
–
230
(Yuan et al., 2019)
Co3ZnC/Co-NCCP
1 M KOH
188
70
108
295
(Yu et al., 2018)
Co9S8 − NSC@Mo2C
1 M KOH
59.7
89
69.3
293
(Luo et al., 2018)
NiCoFeP/C
65
270
108
149
(Wei et al., 2019)
Co/NBC-900
1 M KOH
70
302
247
117
(Liu et al., 2018)
Co3O4@CuCAT
1 M KOH
75
267
59
100
(Hu et al., 2019)
Co3S4/EC-MOF
1 M KOH
120
226
82
84
(Liu et al., 2019)
NiFe-MOF
0.1 M KOH
34
240
N/A
134
(Duan et al., 2017)
MFN-MOFs
1 M KOH
55.4
294 at 500 mA/cm2
30.1
234 at 500 mA/cm2
(Raja et al., 2019)
NFN-MOF/NF
1 M KOH
58.8
240
35.2
87
(Senthil Raja et al., 2018)
FeNi(BDC)(DMF,F)/NF
1 M KOH
37.4
227 at 60 mA/cm2
96.2
234 at 60 mA/cm2
(Lin et al., 2019)
NiFeZn-MNS/NF
1 M KOH
49
350 at 50 mA/cm2
N/A
180
(Wei et al., 2019)
Cu3(PO4)2/Cu-BDC150-6 foam
1 M KOH
76
241
N/A
145
(Rong et al., 2019)
Ni-ZIF/NieB@NF
1 M KOH
57
234
108
67
(Xu et al., 2020)
4 Conclusion and future perspectives
Hydrogen production using electrocatalysis and photocatalysis is intended to address a variety of environmental and energy-related issues. The formation of an affordable, easily available, and unique catalysts is a critical challenge for achieving effective water splitting. In this regard, MOF derived carbon catalysts may provide various benefits over traditional catalysts due to their distinctive morphologies, superior optical, electrical, and catalytic capabilities, high porosity, crystallinity, structural adjustability, and increased surface area. Beginning with the principles, procedure, and evaluation aspects, and detailed research of MOFs as direct new catalysts, precursors for derived catalysts, and MOF based composites for electrocatalytic and photocatalytic water splitting, various noteworthy outcomes are reported.
We reviewed significant achievements in recent research on fabricating MOF derived carbon based catalysts for effective electro and photo catalytic water splitting. Various MOF derived carbon based materials with high water splitting performance have been developed by exploiting the vast surface area, customizable chemical components, controllable pore structure, programmable topology, and quite well surface functionality. This review highlights a variety of novel synthetic strategies for improving the chemical composition and structural functionalization of catalytic sites in order to enhance their electro/photo catalytic activity, particularly at the atomic scales and for tailoring nanoarchitectures and combinations. The morphological characteristics of MOFs require more comprehensive experimental analysis or theoretical research (Yang et al., 2014). Along with shape, electro/photo catalyst interactions or synergistic effects have a significant impact on the catalytic performance of carbon based MOFs, necessitating more focus on the application of MOF-based electro and photocatalysts for water splitting processes (Jiao et al., 2016). The rapid advancements in catalytic activity, the identification of highly active sites and basic processes contribute to the hydrogen generation industrially (Wang et al., 2017).
This review also summarized the current challenges in the use of carbon based MOFs as photocatalysts for water splitting, with a focus on several important methods for developing high-performance photocatalysts that significantly influenced by its sunlight absorption capacity (i.e., band gap), the charge separation/transfer efficiency, and the number of active sites (Liang et al., 2015). The inclusion of active sites, like Pt nanoparticles, into MOFs was revealed to significantly improve photocatalytic activity. Unfortunately, platinum's limited supplies and expensive cost make it inappropriate for widespread use. Recently, it was shown that extremely effective, small-sized Ni particles implanted in MOF-5 as co-catalysts with negligible overpotential for photocatalytic HER under visible light. This photocatalyst had a low overpotential of 0.37 V, which was similar to Pt@MOF-5.
Although great progress has been made in the field of MOF-based catalysts for electrocatalyst and photocatalytic water-splitting, major work has to be done to make hydrogen production processes practicable and completely economical at an industrial scale. In the future, greater emphasis will be paid for the creation of non-precious metal NPs from MOFs to improve catalytic activity. For this, various research approaches on MOFs for water-splitting may be investigated, along with (i) Researching novel forms of MOFs with design and active sites has a broad scope because only a few are presently used (MIL, UiO, and ZIFs). (ii) As MOFs have a hybrid structure, adjusting the charge transfer capacities of MOFs by fine-tuning the coordination arrangement of metal nodes with organic linkers is an appealing option. (iii) Charge transfer attributes inside different components of MOF derived photo/electrocatalysts are largely unknown; a thorough knowledge of the mechanisms underlying elementary reactions for hydrogen production, light collecting, and electron transfer reactions between the interfaces could result in a rise in hydrogen production efficiency. (iv) So, because exact structure of the majority of MOF based carbon catalysts is still unknown, limitations in comprehending structure-catalysis correlations exist; hence, further extensive study on the morphology of MOF based carbon catalysts should be conducted. (v) Improved stability, direct application of MOF catalysts to a surface of the electrode without the need of binders, and the fabrication of new MOFs are other highly wanted future research subjects (Pham et al., 2014; Toyao et al., 2013).
CRediT authorship contribution statement
Neelam Zaman: Data curation, Writing – original draft. Naseem Iqbal: Conceptualization, Methodology, Supervision. Tayyaba Noor: Writing – review & editing, Supervision.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Recent progress on photocatalytic and photoelectrochemical water splitting under visible light irradiation. J. Photochem. Photobiol., C. 2010;11:179-209.
- [Google Scholar]
- A review on selected heterogeneous photocatalysts for hydrogen production. Int. J. Energy Res.. 2014;38:1903-1920.
- [Google Scholar]
- Impact assessment and efficiency evaluation of hydrogen production methods. Int. J. Energy Res.. 2015;39:1757-1768.
- [Google Scholar]
- Hydrogen from photo-catalytic water splitting process: A review. Renew. Sustain. Energy Rev.. 2015;43:599-610.
- [Google Scholar]
- Direct growth of ternary Ni–Fe–P porous nanorods onto nickel foam as a highly active, robust bi-functional electrocatalyst for overall water splitting. J. Mater. Chem. A. 2017;5:2496-2503.
- [Google Scholar]
- Horizons in Sustainable Industrial Chemistry and Catalysis: Elsevier 2019
- Multifunctional Mo–N/C@ MoS2 electrocatalysts for HER, OER, ORR, and Zn–air batteries. Adv. Funct. Mater.. 2017;27:1702300.
- [Google Scholar]
- Recent trends and perspectives in electrochemical water splitting with an emphasis on sulfide, selenide, and phosphide catalysts of Fe Co, and Ni: a review. ACS Catal.. 2016;6:8069-8097.
- [Google Scholar]
- Electrochemically dealloyed nanoporous Fe 40 Ni 20 Co 20 P 15 C 5 metallic glass for efficient and stable electrocatalytic hydrogen and oxygen generation. RSC Adv.. 2021;11:7369-7380.
- [Google Scholar]
- MOF derived nonstoichiometric NixCo3− xO4− y nanocage for superior electrocatalytic oxygen evolution. Adv. Mater. Interfaces. 2016;3:1600632.
- [Google Scholar]
- MOF-Derived CuPt/NC Electrocatalyst for Oxygen Reduction Reaction. Catalysts. 2020;10:799.
- [Google Scholar]
- Formation of yolk–shelled nickel–cobalt selenide dodecahedral nanocages from metal–organic frameworks for efficient hydrogen and oxygen evolution. ACS Sustainable Chem. Eng.. 2018;6:10952-10959.
- [Google Scholar]
- Efficient photocatalytic hydrogen evolution under visible light by ternary composite CdS@ NU-1000/RGO. Catal. Sci. Technol.. 2017;7:5113-5119.
- [Google Scholar]
- One-step sonochemical synthesis of NiMn-LDH for supercapacitors and overall water splitting. J. Mater. Sci. 2021:1-14.
- [Google Scholar]
- Co-MOF as a sacrificial template: manifesting a new Co 3 O 4/TiO 2 system with ap–n heterojunction for photocatalytic hydrogen evolution. 2015;3:20288-20296.
- Metal–organic frameworks in heterogeneous catalysis: recent progress, new trends, and future perspectives. Chem. Rev.. 2020;120:8468-8535.
- [Google Scholar]
- Chemistry of materials for water splitting reactions (ed:). ACS Publications; 2018.
- Interfacial engineering of nickel/iron/ruthenium phosphides for efficient overall water splitting powered by solar energy. J. Mater. Chem. A. 2022;10:772-778.
- [Google Scholar]
- Bottom-up synthesis of MOF-derived hollow N-doped carbon materials for enhanced ORR performance. 2019;146:248-256.
- “Bimetallic AgNi nanoparticles anchored onto MOF-derived nitrogen-doped carbon nanostrips for efficient hydrogen evolution,” Green. Energy Environ. 2021
- [Google Scholar]
- Design strategies of phosphorus-containing catalysts for photocatalytic, photoelectrochemical and electrocatalytic water splitting. Green Chem. 2022
- [Google Scholar]
- Hierarchical trimetallic Co-Ni-Fe oxides derived from core-shell structured metal-organic frameworks for highly efficient oxygen evolution reaction. Appl. Catal. B. 2021;287:119953
- [Google Scholar]
- In situ formation of cobalt nitrides/graphitic carbon composites as efficient bifunctional electrocatalysts for overall water splitting. ACS Appl. Mater. Interfaces. 2018;10:7134-7144.
- [Google Scholar]
- Ultrafine Co nanoparticles encapsulated in carbon-nanotubes-grafted graphene sheets as advanced electrocatalysts for the hydrogen evolution reaction. Adv. Mater.. 2018;30:1802011.
- [Google Scholar]
- In situ identification of the electrocatalytic water oxidation behavior of a nickel-based metal–organic framework nanoarray. Mater. Horiz.. 2021;8:556-564.
- [Google Scholar]
- Binary and ternary doping of nitrogen, boron, and phosphorus into carbon for enhancing electrochemical oxygen reduction activity. ACS Nano. 2012;6:7084-7091.
- [Google Scholar]
- Copper modified-TiO2 catalysts for hydrogen generation through photoreforming of organics. A short review. Int. J. Hydrogen Energy. 2014;39:16812-16831.
- [Google Scholar]
- Molybdenum polysulfide anchored on porous Zr-metal organic framework to enhance the performance of hydrogen evolution reaction. The Journal of Physical Chemistry C. 2016;120:12539-12548.
- [Google Scholar]
- Self-assemble and in situ formation of Ni1− xFexPS3 nanomosaic-decorated MXene hybrids for overall water splitting. Adv. Energy Mater.. 2018;8:1801127.
- [Google Scholar]
- Metal–organic framework-derived Cu-doped Co9S8 nanorod array with less low-valence Co sites as highly efficient bifunctional electrodes for overall water splitting. ACS Sustainable Chem. Eng.. 2019;7:16917-16926.
- [Google Scholar]
- Controllable synthesized CoP-MP (M= Fe, Mn) as efficient and stable electrocatalyst for hydrogen evolution reaction at all pH values. Int. J. Hydrogen Energy. 2019;44:19978-19985.
- [Google Scholar]
- Nest-like NiCoP for highly efficient overall water splitting. ACS Catal.. 2017;7:4131-4137.
- [Google Scholar]
- Ultrathin metal-organic framework array for efficient electrocatalytic water splitting. Nat. Commun.. 2017;8:1-7.
- [Google Scholar]
- “Synthesis of reduced graphene oxide–TiO2 nanoparticle composite systems and its application in hydrogen production,” international journal of hydrogen energy. 2014;39:16282-16292.
- M. I. Fadlalla and S. G. Babu, “Role of graphene in photocatalytic water splitting for hydrogen production,” in Graphene-Based Nanotechnologies for Energy and Environmental Applications, ed: Elsevier, 2019, pp. 81-108.
- A critical review in strategies to improve photocatalytic water splitting towards hydrogen production. Int. J. Hydrogen Energy. 2019;44:540-577.
- [Google Scholar]
- Atomically isolated nickel species anchored on graphitized carbon for efficient hydrogen evolution electrocatalysis. Nat. Commun.. 2016;7:1-7.
- [Google Scholar]
- A metal–organic-framework/carbon composite with enhanced bifunctional electrocatalytic activities towards oxygen reduction/evolution reactions. Int. J. Hydrogen Energy. 2017;42:17376-17385.
- [Google Scholar]
- Controllable synthesis of metal-organic frameworks and their hierarchical assemblies. Matter. 2019;1:801-824.
- [Google Scholar]
- Metal–organic frameworks for photo/electrocatalysis. Advanced Energy and Sustainability Research. 2021;2:2100033.
- [Google Scholar]
- Defect-rich cobalt pyrophosphate hybrids decorated Cd0. 5Zn0. 5S for efficient photocatalytic hydrogen evolution: Defect and interface engineering. J. Colloid Interface Sci.. 2022;606:544-555.
- [Google Scholar]
- Nitrogen-doped carbon nanotube arrays with high electrocatalytic activity for oxygen reduction. Science. 2009;323:760-764.
- [Google Scholar]
- Bifunctional electrocatalysts for water splitting from a bimetallic (V doped-NixFey) Metal-Organic framework MOF@ Graphene oxide composite. Int. J. Hydrogen Energy 2021
- [Google Scholar]
- Recent progress and prospect of carbon-free single-site catalysts for the hydrogen and oxygen evolution reactions. Nano Res. 2021:1-20.
- [Google Scholar]
- Metal–organic framework derived hollow CoS 2 nanotube arrays: an efficient bifunctional electrocatalyst for overall water splitting. Nanoscale Horiz.. 2017;2:342-348.
- [Google Scholar]
- Band gap engineering of metal-organic frameworks for solar fuel productions. Coord. Chem. Rev.. 2021;435:213785
- [Google Scholar]
- Metal–organic framework immobilized cobalt oxide nanoparticles for efficient photocatalytic water oxidation. J. Mater. Chem. A. 2015;3:20607-20613.
- [Google Scholar]
- High-performance electrocatalyst of vanadium-iron bimetal organic framework arrays on nickel foam for overall water splitting. Chin. Chem. Lett.. 2021;32:2263-2268.
- [Google Scholar]
- ZIF derived PtNiCo/NC cathode catalyst for proton exchange membrane fuel cell. Appl. Catal. B. 2019;258:117947
- [Google Scholar]
- Biomimetic di-manganese catalyst cage-isolated in a MOF: robust catalyst for water oxidation with Ce (IV), a non-O-donating oxidant. 2014;7:317-322.
- Porous Ni2P/C microrods derived from microwave-prepared MOF-74-Ni and its electrocatalysis for hydrogen evolution reaction. Mater. Lett.. 2018;231:94-97.
- [Google Scholar]
- A dye-sensitized Pt@ UiO-66 (Zr) metal–organic framework for visible-light photocatalytic hydrogen production. 2014;50:7063-7066.
- Co-based MOF-derived Co/CoN/Co2P ternary composite embedded in N-and P-doped carbon as bifunctional nanocatalysts for efficient overall water splitting. Int. J. Hydrogen Energy. 2019;44:11402-11410.
- [Google Scholar]
- Nickel foam and stainless steel mesh as electrocatalysts for hydrogen evolution reaction, oxygen evolution reaction and overall water splitting in alkaline media. RSC Adv.. 2019;9:31563-31571.
- [Google Scholar]
- Metal–organic frameworks embedded in a liposome facilitate overall photocatalytic water splitting. Nat. Chem.. 2021;13:358-366.
- [Google Scholar]
- “Co3O4@ Cu-Based Conductive Metal-Organic Framework Core-Shell Nanowire Electrocatalysts Enable Efficient Low-Overall-Potential Water Splitting,” Chemistry–A. European Journal. 2019;25:6575-6583.
- [Google Scholar]
- Efficient hydrogen evolution activity and overall water splitting of metallic Co4N nanowires through tunable d-orbitals with ultrafast incorporation of FeOOH. ACS Appl. Mater. Interfaces. 2019;11:5152-5158.
- [Google Scholar]
- “Enhanced metallicity boosts hydrogen evolution capability of dual-bimetallic Ni–Fe nitride nanoparticles,” Materials Today. Physics. 2020;15:100267
- [Google Scholar]
- “Strategies for the enhanced water splitting activity over metal-organic frameworks-based electrocatalysts and photocatalysts,” Materials Today. NANO 2021100124
- [Google Scholar]
- Rational construction of ultrafine noble metals onto carbon nanoribbons with efficient oxygen reduction in practical alkaline fuel cell. Chem. Eng. J.. 2021;424:130336
- [Google Scholar]
- Highly dispersed Pt clusters encapsulated in MIL-125-NH2 via in situ auto-reduction method for photocatalytic H2 production under visible light. Nano Res. 2021:1-8.
- [Google Scholar]
- Hollow cobalt-based bimetallic sulfide polyhedra for efficient all-pH-value electrochemical and photocatalytic hydrogen evolution. J. Am. Chem. Soc.. 2016;138:1359-1365.
- [Google Scholar]
- Tailor-made pyrazolide-based metal–organic frameworks for selective catalysis. J. Am. Chem. Soc.. 2018;140:6383-6390.
- [Google Scholar]
- Nonpyrolyzed Fe− N Coordination-Based Iron Triazolate Framework: An Efficient and Stable Electrocatalyst for Oxygen Reduction Reaction. ChemSusChem. 2019;12:200-207.
- [Google Scholar]
- Efficient charges separation using advanced BiOI-based hollow spheres decorated with palladium and manganese dioxide nanoparticles. ACS Sustainable Chem. Eng.. 2018;6:2751-2757.
- [Google Scholar]
- Nitrogen treatment generates tunable nanohybridization of Ni5P4 nanosheets with nickel hydr (oxy) oxides for efficient hydrogen production in alkaline, seawater and acidic media. Appl. Catal. B. 2019;251:181-194.
- [Google Scholar]
- Highly graphitized N-doped carbon nanosheets from 2-dimensional coordination polymers for efficient metal-air batteries. Carbon. 2022;188:135-145.
- [Google Scholar]
- Enhanced water splitting electrocatalysis over mnco2o4 via introduction of suitable ce content. ACS Sustainable Chem. Eng.. 2018;7:1169-1177.
- [Google Scholar]
- Earth-abundant heterogeneous water oxidation catalysts. Chem. Rev.. 2016;116:14120-14136.
- [Google Scholar]
- Alkaline water-splitting reactions over Pd/Co-MOF-derived carbon obtained via microwave-assisted synthesis. RSC Adv.. 2020;10:17359-17368.
- [Google Scholar]
- Critical Review, Recent Updates on Zeolitic Imidazolate Framework-67 (ZIF-67) and Its Derivatives for Electrochemical Water Splitting. Adv. Mater. 2021:2107072.
- [Google Scholar]
- A Graphene oxide and copper-centered metal organic framework composite as a tri-functional catalyst for HER, OER, and ORR. Adv. Funct. Mater.. 2013;23:5363-5372.
- [Google Scholar]
- Nanoporous nitrogen-doped graphene oxide/nickel sulfide composite sheets derived from a metal-organic framework as an efficient electrocatalyst for hydrogen and oxygen evolution. Adv. Funct. Mater.. 2017;27:1700451.
- [Google Scholar]
- Metal–organic framework-based CoP/reduced graphene oxide: high-performance bifunctional electrocatalyst for overall water splitting. Chem. Sci.. 2016;7:1690-1695.
- [Google Scholar]
- Nickel nanoparticles coated on the exfoliated graphene layer as an efficient and stable catalyst for oxygen reduction and hydrogen evolution in alkaline media. Mater. Res. Express. 2020;7:055504
- [Google Scholar]
- P. Karthik and B. Neppolian, “Metal–Organic Frameworks (MOFs) with Hierarchical Structures for Visible Light Photocatalysis,” in Nanostructured Materials for Environmental Applications, ed: Springer, 2021, pp. 233-245.
- π–π interaction between metal–organic framework and reduced graphene oxide for visible-light photocatalytic H2 production. ACS Applied Energy Materials. 2018;1:1913-1923.
- [Google Scholar]
- Amine Functionalized Metal-Organic Framework Coordinated with Transition Metal Ions: d–d Transition Enhanced Optical Absorption and Role of Transition Metal Sites on Solar Light Driven H2 Production. Small. 2020;16:1902990.
- [Google Scholar]
- Highly efficient water splitting into H2 and O2 over lanthanum-doped NaTaO3 photocatalysts with high crystallinity and surface nanostructure. J. Am. Chem. Soc.. 2003;125:3082-3089.
- [Google Scholar]
- Seawater electrolysis for hydrogen production: a solution looking for a problem? Energy Environ. Sci.. 2021;14:4831-4839.
- [Google Scholar]
- Flexible Photo-Catalytic Electrode using Graphene, Non-Noble Metal, and Organic Semiconductors for Hydrogen Evolution Reaction. Energy Technology 2021
- [Google Scholar]
- Cu, Co-embedded N-enriched mesoporous carbon for efficient oxygen reduction and hydrogen evolution reactions. Adv. Energy Mater.. 2017;7:1700193.
- [Google Scholar]
- Photocatalytic hydrogen production: role of sacrificial reagents on the activity of oxide, carbon, and sulfide catalysts. Catalysts. 2019;9:276.
- [Google Scholar]
- CoSe2 and NiSe2 nanocrystals as superior bifunctional catalysts for electrochemical and photoelectrochemical water splitting. ACS Appl. Mater. Interfaces. 2016;8:5327-5334.
- [Google Scholar]
- “Metal-free bifunctional carbon electrocatalysts derived from zeolitic imidazolate frameworks for efficient water splitting,” Materials Chemistry. Frontiers. 2018;2:102-111.
- [Google Scholar]
- J. Li, S. Wang, J. Chang, and L. Feng, “A review of Ni based powder catalyst for urea oxidation in assisting water splitting reaction,” Advanced Powder Materials, 2022.
- Transition metal-based bimetallic MOFs and MOF-derived catalysts for electrochemical oxygen evolution reaction. Energy Environ. Sci.. 2021;14:1897-1927.
- [Google Scholar]
- Photocatalytic water splitting on semiconductor-based photocatalysts. Adv. Catal.. 2017;60:1-57.
- [Google Scholar]
- Cobalt nanoparticles embedded in porous N-rich carbon as an efficient bifunctional electrocatalyst for water splitting. J. Mater. Chem. A. 2016;4:3204-3209.
- [Google Scholar]
- Inlaying ultrathin bimetallic MOF nanosheets into 3D ordered macroporous hydroxide for superior electrocatalytic oxygen evolution. Small. 2019;15:1902218.
- [Google Scholar]
- A review of material aspects in developing direct Z-scheme photocatalysts. Mater. Today. 2021;47:75-107.
- [Google Scholar]
- Z-Scheme photocatalytic systems for promoting photocatalytic performance: recent progress and future challenges. Adv. Sci.. 2016;3:1500389.
- [Google Scholar]
- Titanium phosphonate based metal–organic frameworks with hierarchical porosity for enhanced photocatalytic hydrogen evolution. Angew. Chem.. 2018;130:3276-3281.
- [Google Scholar]
- Strategies to improve electrocatalytic and photocatalytic performance of two-dimensional materials for hydrogen evolution reaction. Chin. J. Catal.. 2021;42:511-556.
- [Google Scholar]
- Polyoxometalate-based metal–organic framework-derived hybrid electrocatalysts for highly efficient hydrogen evolution reaction. J. Mater. Chem. A. 2016;4:1202-1207.
- [Google Scholar]
- Large-Scale, Bottom-Up Synthesis of Binary Metal-Organic Framework Nanosheets for Efficient Water Oxidation. Angew. Chem.. 2019;131:7125-7130.
- [Google Scholar]
- Recent progress in advanced electrocatalyst design for acidic oxygen evolution reaction. Adv. Mater.. 2021;33:2004243.
- [Google Scholar]
- “Metal–Organic Framework-Based Materials for Solar Water Splitting,” Small. Science 2021:2000074.
- [Google Scholar]
- Thermally driven structure and performance evolution of atomically dispersed FeN4 sites for oxygen reduction. Angew. Chem.. 2019;131:19147-19156.
- [Google Scholar]
- A simple strategy for fabrication of Pd@ MIL-100 (Fe) nanocomposite as a visible-light-driven photocatalyst for the treatment of pharmaceuticals and personal care products (PPCPs) Appl. Catal. B. 2015;176:240-248.
- [Google Scholar]
- MOF-derived formation of Ni2P–CoP bimetallic phosphides with strong interfacial effect toward electrocatalytic water splitting. ACS Appl. Mater. Interfaces. 2017;9:23222-23229.
- [Google Scholar]
- Morphology-controlled fabrication of nanostructured WO3 thin films by magnetron sputtering with glancing angle deposition for enhanced efficiency photo-electrochemical water splitting. Ceram. Int. 2021
- [Google Scholar]
- Bi-metallic MOFs possessing hierarchical synergistic effects as high performance electrocatalysts for overall water splitting at high current densities. Appl. Catal. B. 2019;258:118023
- [Google Scholar]
- Chromium-ruthenium oxide solid solution electrocatalyst for highly efficient oxygen evolution reaction in acidic media. Nat. Commun.. 2019;10:1-13.
- [Google Scholar]
- G. Liu, Y. Xu, T. Yang, and L. Jiang, “Recent advances in electrocatalysts for seawater splitting,” Nano Materials Science, 2020.
- X. Liu, X. Zhang, D.-S. Li, S. Zhang, and Q. Zhang, “Recent advances in the “on-off” approaches for on-demand liquid-phase Hydrogen evolution,” Journal of Materials Chemistry A, 2021.
- Metal–Organic-Framework-Derived Co2P Nanoparticle/Multi-Doped Porous Carbon as a Trifunctional Electrocatalyst. Adv. Mater.. 2020;32:2003649.
- [Google Scholar]
- Cobalt boron imidazolate framework derived cobalt nanoparticles encapsulated in B/N codoped nanocarbon as efficient bifunctional electrocatalysts for overall water splitting. Adv. Funct. Mater.. 2018;28:1801136.
- [Google Scholar]
- Charge transmission channel construction between a MOF and rGO by means of Co–Mo–S modification. Catal. Sci. Technol.. 2017;7:4478-4488.
- [Google Scholar]
- Water splitting based on homogeneous copper molecular catalysts. J. Photochem. Photobiol., A. 2018;355:141-151.
- [Google Scholar]
- MOF-derived CoSe 2 microspheres with hollow interiors as high-performance electrocatalysts for the enhanced oxygen evolution reaction. J. Mater. Chem. A. 2017;5:15310-15314.
- [Google Scholar]
- Self-standing CoP nanosheets array: a three-dimensional bifunctional catalyst electrode for overall water splitting in both neutral and alkaline media. ChemElectroChem. 2017;4:1840-1845.
- [Google Scholar]
- Self-sacrificial template-directed vapor-phase growth of MOF assemblies and surface vulcanization for efficient water splitting. Adv. Mater.. 2019;31:1806672.
- [Google Scholar]
- Nitrogen and sulfur-codoped porous carbon derived from a BSA/ionic liquid polymer complex: multifunctional electrode materials for water splitting and supercapacitors. RSC Adv.. 2019;9:5189-5196.
- [Google Scholar]
- A binuclear Co-based metal–organic framework towards efficient oxygen evolution reaction. Chem. Commun.. 2021;57:5016-5019.
- [Google Scholar]
- 3D architecture constructed via the confined growth of MoS 2 nanosheets in nanoporous carbon derived from metal–organic frameworks for efficient hydrogen production. Nanoscale. 2015;7:18004-18009.
- [Google Scholar]
- Construction of Co2P/CoP@ Co@ NCNT rich-interface to synergistically promote overall water splitting. Chem. Eng. J. 2021132877
- [Google Scholar]
- Highly efficient CoMoS heterostructure derived from vertically anchored Co5Mo10 polyoxometalate for electrocatalytic overall water splitting. Chem. Eng. J.. 2020;394:124849
- [Google Scholar]
- Metal–organic framework-derived heterojunctions as nanocatalysts for photocatalytic hydrogen production. Inorg. Chem. Front.. 2019;6:3456-3467.
- [Google Scholar]
- Amorphous type FeOOH modified defective BiVO4 photoanodes for photoelectrochemical water oxidation. Chem. Eng. J.. 2022;428:131027
- [Google Scholar]
- Core-Shell Nanocomposites Based on Gold [email protected] Porous Carbons Derived from Metal-Organic Frameworks as Efficient Dual Catalysts for Oxygen Reduction and Hydrogen Evolution Reactions. ACS Catal.. 2016;6:1045-1053.
- [Google Scholar]
- Robust and stable acidic overall water splitting on Ir single atoms. Nano Lett.. 2020;20:2120-2128.
- [Google Scholar]
- Recent progress on metal-organic frameworks based-and derived-photocatalysts for water splitting. Chem. Eng. J.. 2020;383:123196
- [Google Scholar]
- Porous Co9S8/nitrogen, sulfur-doped carbon@ Mo2C dual catalyst for efficient water splitting. ACS Appl. Mater. Interfaces. 2018;10:22291-22302.
- [Google Scholar]
- Highly efficient overall water splitting driven by all-inorganic perovskite solar cells and promoted by bifunctional bimetallic phosphide nanowire arrays. J. Mater. Chem. A. 2018;6:20076-20082.
- [Google Scholar]
- Photocatalytic water splitting using semiconductor particles: history and recent developments. J. Photochem. Photobiol., C. 2011;12:237-268.
- [Google Scholar]
- Photocatalytic water splitting: recent progress and future challenges. The Journal of Physical Chemistry Letters. 2010;1:2655-2661.
- [Google Scholar]
- On the theory of oxidation-reduction reactions involving electron transfer. II. Applications to data on the rates of isotopic exchange reactions. J. Chem. Phys.. 1957;26:867-871.
- [Google Scholar]
- An insight on the electrocatalytic mechanistic study of pristine Ni MOF (BTC) in alkaline medium for enhanced OER and UOR. ChemElectroChem. 2018;5:2795-2807.
- [Google Scholar]
- The development of molecular water oxidation catalysts. Nat. Rev. Chem.. 2019;3:331-341.
- [Google Scholar]
- MOF-on-MOF Strategy to Construct a Nitrogen-Doped Carbon-Incorporated CoP@ Fe–CoP Core-Shelled Heterostructure for High-Performance Overall Water Splitting. Inorg. Chem. 2021
- [Google Scholar]
- In situ coupling of Co 0.85 Se and N-doped carbon via one-step selenization of metal–organic frameworks as a trifunctional catalyst for overall water splitting and Zn–air batteries. J. Mater. Chem. A. 2017;5:7001-7014.
- [Google Scholar]
- Metal-organic framework as nanoreactors to co-incorporate carbon nanodots and CdS quantum dots into the pores for improved H2 evolution without noble-metal cocatalyst. Appl. Catal. B. 2019;244:340-346.
- [Google Scholar]
- MOF-derived Co-doped nickel selenide/C electrocatalysts supported on Ni foam for overall water splitting. J. Mater. Chem. A. 2016;4:15148-15155.
- [Google Scholar]
- The multifaceted reactivity of single-atom heterogeneous catalysts. Angew. Chem. Int. Ed.. 2018;57:15316-15329.
- [Google Scholar]
- Recent advances on hydrogen production through seawater electrolysis. Materials Science for Energy Technologies. 2020;3:780-807.
- [Google Scholar]
- Revisiting Metal-Organic Frameworks for Oxygen Evolution: A Case Study. Inorg. Chem.. 2020;59:15335-15342.
- [Google Scholar]
- Co@ NH 2-MIL-125 (Ti): cobaloxime-derived metal–organic framework-based composite for light-driven H 2 production. Energy Environ. Sci.. 2015;8:364-375.
- [Google Scholar]
- J. A. Nasir, Z. ur Rehman, S. N. A. Shah, A. Khan, I. S. Butler, and C. R. A. Catlow, “Recent developments and perspectives in CdS-based photocatalysts for water splitting,” Journal of Materials Chemistry A, vol. 8, pp. 20752-20780, 2020.
- “Metal–Organic Frameworks for Photocatalytic Water Splitting”. Solar RRL 2021:2100198.
- [Google Scholar]
- A review and recent developments in photocatalytic water-splitting using TiO2 for hydrogen production. Renew. Sustain. Energy Rev.. 2007;11:401-425.
- [Google Scholar]
- “Enhanced water splitting through two-step photoexcitation by sunlight using tantalum/nitrogen-codoped rutile titania as a water oxidation photocatalyst,” Sustainable. Energy Fuels. 2019;3:2337-2346.
- [Google Scholar]
- A highly efficient and stable copper BTC metal organic framework derived electrocatalyst for oxidation of methanol in DMFC application. Catal. Lett.. 2019;149:3312-3327.
- [Google Scholar]
- Electro catalytic study of NiO-MOF/rGO composites for methanol oxidation reaction. Electrochim. Acta. 2019;307:1-12.
- [Google Scholar]
- Nanocomposites of NiO/CuO based MOF with rGO: an efficient and robust electrocatalyst for methanol oxidation reaction in DMFC. Nanomaterials. 2020;10:1601.
- [Google Scholar]
- Recent Advances in Electrocatalysis of Oxygen Evolution Reaction using Noble-Metal, Transition-Metal, and Carbon-Based Materials. ChemElectroChem. 2021;8:447-483.
- [Google Scholar]
- Graphene based FeO/NiO MOF composites for methanol oxidation reaction. J. Electroanal. Chem.. 2021;890:115249
- [Google Scholar]
- Core–shell ZIF-8@ ZIF-67-derived CoP nanoparticle-embedded N-doped carbon nanotube hollow polyhedron for efficient overall water splitting. J. Am. Chem. Soc.. 2018;140:2610-2618.
- [Google Scholar]
- Visible light induced hydrogen generation using a hollow photocatalyst with two cocatalysts separated on two surface sides. PCCP. 2014;16:5937-5941.
- [Google Scholar]
- M.-H. Pham, C.-T. Dinh, G.-T. Vuong, N.-D. Ta, and T.-O. J. N. M.-O. F. Do, “Hollow Fe2O3-TiO2–PtOx Nanostructure with Two Distinct Cocatalysts Embedded Separately on Two Surface Sides for Efficient Visible Light Water Splitting to Hydrogen,” p. 157, 2013.
- Metal–organic framework-guided growth of Mo 2 C embedded in mesoporous carbon as a high-performance and stable electrocatalyst for the hydrogen evolution reaction. J. Mater. Chem. A. 2016;4:16225-16232.
- [Google Scholar]
- A Review of MOFs and Their Composites-Based Photocatalysts: Synthesis and Applications. Adv. Funct. Mater.. 2021;31:2104231.
- [Google Scholar]
- Ultrastable polymolybdate-based metal–organic frameworks as highly active electrocatalysts for hydrogen generation from water. J. Am. Chem. Soc.. 2015;137:7169-7177.
- [Google Scholar]
- Metal-organic frameworks-derived novel nanostructured electrocatalysts for oxygen evolution reaction. Carbon Energy. 2021;3:66-100.
- [Google Scholar]
- Design engineering, synthesis protocols, and energy applications of MOF-derived electrocatalysts. Nano-Micro Letters. 2021;13:1-32.
- [Google Scholar]
- Synergistically well-mixed MOFs grown on nickel foam as highly efficient durable bifunctional electrocatalysts for overall water splitting at high current densities. Nano Energy. 2019;57:1-13.
- [Google Scholar]
- Fabrication strategies of porous precious-metal-free bifunctional electrocatalysts for overall water splitting: Recent advances. Green Energy Environ.. 2021;6:620-643.
- [Google Scholar]
- Synthesis and characterization of Cu-MOF derived Cu@ AC electrocatalyst for oxygen reduction reaction in PEMFC. Catal. Lett.. 2020;150:1397-1407.
- [Google Scholar]
- Earth-abundant catalysts for electrochemical and photoelectrochemical water splitting. Nat. Rev. Chem.. 2017;1:1-13.
- [Google Scholar]
- Self-directed hierarchical Cu3 (PO4) 2/Cu-BDC nanosheets array based on copper foam as an efficient and durable electrocatalyst for overall water splitting. Electrochim. Acta. 2019;313:179-188.
- [Google Scholar]
- ZIF-67 derived MnO2 doped electrocatalyst for oxygen reduction reaction. Catalysts. 2021;11:92.
- [Google Scholar]
- Effect of Co-Ni Ratio in Graphene Based Bimetallic Electro-catalyst for Methanol Oxidation. Fuel cells. 2018;18:189-194.
- [Google Scholar]
- In situ grown bimetallic MOF-based composite as highly efficient bifunctional electrocatalyst for overall water splitting with ultrastability at high current densities. Adv. Energy Mater.. 2018;8:1801065.
- [Google Scholar]
- Noble-metal-free MoS2 co-catalyst decorated UiO-66/CdS hybrids for efficient photocatalytic H2 production. 2015;166:445-453.
- Artificial chloroplast: Au/chloroplast-morph-TiO2 with fast electron transfer and enhanced photocatalytic activity. Int. J. Hydrogen Energy. 2014;39:5617-5624.
- [Google Scholar]
- Interfacial sites between cobalt nitride and cobalt act as bifunctional catalysts for hydrogen electrochemistry. ACS Energy Lett.. 2019;4:1594-1601.
- [Google Scholar]
- Fabrication of NH2-MIL-125 (Ti) incorporated TiO2 nanotube arrays composite anodes for highly efficient PEC water splitting. Sep. Purif. Technol.. 2019;228:115764
- [Google Scholar]
- Bimetallic cobalt-based phosphide zeolitic imidazolate framework: CoPx phase-dependent electrical conductivity and hydrogen atom adsorption energy for efficient overall water splitting. Adv. Energy Mater.. 2017;7:1601555.
- [Google Scholar]
- Constructing Ni/NiS heteronanoparticle-embedded metal–organic framework-derived nanosheets for enhanced water-splitting catalysis. ACS Sustainable Chem. Eng.. 2021;9:1920-1931.
- [Google Scholar]
- P. Stelmachowski, J. Duch, D. Sebastián, M. Lázaro, and A. Kotarba, “Carbon-Based Composites as Electrocatalysts for Oxygen Evolution Reaction in Alkaline Media. Materials 2021, 14, 4984,” ed: s Note: MDPI stays neu-tral with regard to jurisdictional claims in …, 2021.
- Role of interfaces in two-dimensional photocatalyst for water splitting. ACS Catal.. 2018;8:2253-2276.
- [Google Scholar]
- Porous multishelled Ni2P hollow microspheres as an active electrocatalyst for hydrogen and oxygen evolution. Chem. Mater.. 2017;29:8539-8547.
- [Google Scholar]
- Interfacial Design to Enhance Photocatalytic Hydrogen Evolution via Optimizing Energy and Mass Flows. ACS Appl. Mater. Interfaces. 2021;13:21207-21216.
- [Google Scholar]
- MOF-derived three-dimensional ordered porous carbon nanomaterial for efficient alkaline zinc-air batteries. Sci. China Mater. 2022:1-10.
- [Google Scholar]
- Metal-organic frameworks derived cobalt phosphide architecture encapsulated into B/N Co-doped graphene nanotubes for all pH value electrochemical hydrogen evolution. Adv. Energy Mater.. 2017;7:1601671.
- [Google Scholar]
- Advances in visible light responsive titanium oxide-based photocatalysts for CO2 conversion to hydrocarbon fuels. Energy Convers. Manage.. 2013;76:194-214.
- [Google Scholar]
- Mechanism of photocatalytic water splitting in TiO2. Reaction of water with photoholes, importance of charge carrier dynamics, and evidence for four-hole chemistry. J. Am. Chem. Soc.. 2008;130:13885-13891.
- [Google Scholar]
- A needle-like cobalt-based bifunctional catalyst supported on carbon materials for effective overall water splitting. Nanotechnology. 2021;33:065704
- [Google Scholar]
- A review of defect engineering in two-dimensional materials for electrocatalytic hydrogen evolution reaction. Chin. J. Catal.. 2022;43:636-678.
- [Google Scholar]
- MOF-derived noble metal free catalysts for electrochemical water splitting. ACS Appl. Mater. Interfaces. 2016;8:35390-35397.
- [Google Scholar]
- Metal-organic framework derived Co3O4/PPy bifunctional electrocatalysts for efficient overall water splitting. Chin. Chem. Lett.. 2020;31:2295-2299.
- [Google Scholar]
- “Efficient hydrogen production and photocatalytic reduction of nitrobenzene over a visible-light-responsive metal–organic framework photocatalyst,” Catalysis. Science & Technology. 2013;3:2092-2097.
- [Google Scholar]
- Tuning the band gap of TiO2 by tungsten doping for efficient UV and visible photodegradation of Congo red dye. Spectrochim. Acta Part A Mol. Biomol. Spectrosc.. 2018;204:150-157.
- [Google Scholar]
- Supported polyoxometalates as emerging nanohybrid materials for photochemical and photoelectrochemical water splitting. Nanophotonics. 2021;10:1595-1620.
- [Google Scholar]
- Recent advancements in chemical looping water splitting for the production of hydrogen. RSC Adv.. 2016;6:98267-98296.
- [Google Scholar]
- Charge Relays via Dual Carbon-Actions on Nanostructured BiVO4 for High Performance Photoelectrochemical Water Splitting. Adv. Funct. Mater. 2022:2112738.
- [Google Scholar]
- “Quasi-polymeric metal–organic framework UiO-66/g-C3N4 heterojunctions for enhanced photocatalytic hydrogen evolution under visible light irradiation,” Advanced Materials. Interfaces. 2015;2:1500037.
- [Google Scholar]
- MOF-Derived bifunctional Cu3P nanoparticles coated by a N, P-codoped carbon shell for hydrogen evolution and oxygen reduction. Adv. Mater.. 2018;30:1703711.
- [Google Scholar]
- Surfactant-Mediated Morphological Evolution of MnCo Prussian Blue Structures. Small. 2020;16:2004614.
- [Google Scholar]
- New strategies for novel MOF-derived carbon materials based on nanoarchitectures. Chem. 2020;6:19-40.
- [Google Scholar]
- Hollow bimetallic cobalt-based selenide polyhedrons derived from metal–organic framework: an efficient bifunctional electrocatalyst for overall water splitting. J. Mater. Chem. A. 2017;5:17982-17989.
- [Google Scholar]
- Metal-Organic Frameworks Based Photocatalysts: Architecture Strategies for Efficient Solar Energy Conversion. Chem. Eng. J. 2021129459
- [Google Scholar]
- Xie., “Bandgap engineering of organic semiconductors for highly efficient photocatalytic water splitting,”. Adv. Energy Mater.. 2018;8:1801084.
- [Google Scholar]
- “A Bismuth-Based Metal-Organic Framework as an Efficient Visible-Light-Driven Photocatalyst,” Chemistry–A. European Journal 2015:2364-2367.
- [Google Scholar]
- An overview and recent advances in electrocatalysts for direct seawater splitting. Front. Chem. Sci. Eng.. 2021;15:1408-1426.
- [Google Scholar]
- Recent progress in metal-organic frameworks for applications in electrocatalytic and photocatalytic water splitting. Adv. Sci.. 2017;4:1600371.
- [Google Scholar]
- Generally transform 3-dimensional In-based metal-organic frameworks into 2-dimensional Co, N-doped carbon nanosheets for Zn-air battery. J. Power Sources. 2019;440:227158
- [Google Scholar]
- Strategies of designing electrocatalysts for seawater splitting. J. Solid State Chem.. 2022;306:122799
- [Google Scholar]
- J. Wei, Y. Feng, Y. Liu, and Y. J. J. o. M. C. A. Ding, “M x Co 3− x O 4 (M= Co, Mn, Fe) porous nanocages derived from metal–organic frameworks as efficient water oxidation catalysts,” vol. 3, pp. 22300-22310, 2015.
- Ultrathin NiFeZn-MOF nanosheets containing few metal oxide nanoparticles grown on nickel foam for efficient oxygen evolution reaction of electrocatalytic water splitting. Electrochim. Acta. 2019;318:957-965.
- [Google Scholar]
- Carbon-incorporated porous honeycomb NiCoFe phosphide nanospheres derived from a MOF precursor for overall water splitting. Chem. Commun.. 2019;55:10896-10899.
- [Google Scholar]
- Cu Nanoparticles Embedded in N-Doped Carbon Materials for Oxygen Reduction Reaction. Chin. J. Chem .. 2020;38:941-946.
- [Google Scholar]
- Applications of metal–organic framework-derived materials in fuel cells and metal-air batteries. Coord. Chem. Rev.. 2020;409:213214
- [Google Scholar]
- Porous molybdenum carbide nano-octahedrons synthesized via confined carburization in metal-organic frameworks for efficient hydrogen production. Nat. Commun.. 2015;6:1-8.
- [Google Scholar]
- In situ coupling of CoP polyhedrons and carbon nanotubes as highly efficient hydrogen evolution reaction electrocatalyst. Small. 2017;13:1602873.
- [Google Scholar]
- Graphene-based materials for hydrogen generation from light-driven water splitting. Adv. Mater.. 2013;25:3820-3839.
- [Google Scholar]
- T. Xiong, X. Yao, Z. Zhu, R. Xiao, Y. w. Hu, Y. Huang, et al., “In Situ Grown Co‐Based Interstitial Compounds: Non‐3d Metal and Non‐Metal Dual Modulation Boosts Alkaline and Acidic Hydrogen Electrocatalysis,” Small, p. 2105331, 2021.
- “Unveiling the promotion of accelerated water dissociation kinetics on the hydrogen evolution catalysis of NiMoO4 nanorods,” Journal of Energy. Chemistry. 2022;67:805-813.
- [Google Scholar]
- Boronization-Induced Ultrathin 2D Nanosheets with Abundant Crystalline-Amorphous Phase Boundary Supported on Nickel Foam toward Efficient Water Splitting. Adv. Energy Mater.. 2020;10:1902714.
- [Google Scholar]
- Porous CoP concave polyhedron electrocatalysts synthesized from metal–organic frameworks with enhanced electrochemical properties for hydrogen evolution. J. Mater. Chem. A. 2015;3:21471-21477.
- [Google Scholar]
- Trends in activity for the oxygen evolution reaction on transition metal (M= Fe Co, Ni) phosphide pre-catalysts. Chem. Sci.. 2018;9:3470-3476.
- [Google Scholar]
- The role of Cr doping in NiFe oxide/(oxy) hydroxide electrocatalysts for oxygen evolution. Electrochim. Acta. 2018;265:10-18.
- [Google Scholar]
- Stringing MOF-derived nanocages: a strategy for the enhanced oxygen evolution reaction. J. Mater. Chem. A. 2019;7:8284-8291.
- [Google Scholar]
- Direct Z-scheme photocatalysts: principles, synthesis, and applications. Mater. Today. 2018;21:1042-1063.
- [Google Scholar]
- CdS@ Ni3S2 for efficient and stable photo-assisted electrochemical (P-EC) overall water splitting. Chem. Eng. J.. 2021;405:126231
- [Google Scholar]
- N-atoms-coordinated Mn cofactors within a graphene framework as bioinspired oxygen reduction reaction electrocatalysts,“. Adv. Mater.. 2018;30:1801732.
- [Google Scholar]
- Electrochemical synthesis of flower shaped morphology MOFs in an ionic liquid system and their electrocatalytic application to the hydrogen evolution reaction. RSC Adv.. 2014;4:15720-15726.
- [Google Scholar]
- Nanostructured transition metal compounds coated 3D porous core-shell carbon fiber as monolith water splitting electrocatalysts: a general strategy. Chem. Eng. J.. 2021;423:130279
- [Google Scholar]
- Sulfur-doped graphene as an efficient metal-free cathode catalyst for oxygen reduction. ACS Nano. 2012;6:205-211.
- [Google Scholar]
- Recent progress in MOF-derived, heteroatom-doped porous carbons as highly efficient electrocatalysts for oxygen reduction reaction in fuel cells. Adv. Funct. Mater.. 2018;28:1704537.
- [Google Scholar]
- Research Progress of Metal Organic Frameworks/Carbon-Based Composites for Microwave Absorption. Adv. Eng. Mater. 2021:2100964.
- [Google Scholar]
- Recent advances on electrocatalytic and photocatalytic seawater splitting for hydrogen evolution. Int. J. Hydrogen Energy 2021
- [Google Scholar]
- L. Yaqoob, T. Noor, N. Iqbal, H. Nasir, and A. Mumtaz, “Electrocatalytic Performance of NiNH2BDC MOF Based Composites with rGO For Methanol Oxidation Reaction,” 2021.
- Development of nickel-BTC-MOF-derived nanocomposites with rGO towards electrocatalytic oxidation of methanol and its product analysis. Catalysts. 2019;9:856.
- [Google Scholar]
- Nanocomposites of cobalt benzene tricarboxylic acid MOF with rGO: an efficient and robust electrocatalyst for oxygen evolution reaction (OER) Renewable Energy. 2020;156:1040-1054.
- [Google Scholar]
- Development of an efficient non-noble metal based anode electrocatalyst to promote methanol oxidation activity in DMFC. ChemistrySelect. 2020;5:6023-6034.
- [Google Scholar]
- Electrochemical synergies of Fe–Ni bimetallic MOF CNTs catalyst for OER in water splitting. J. Alloy. Compd.. 2021;850:156583
- [Google Scholar]
- Facile one-pot solvothermal method to synthesize sheet-on-sheet reduced graphene oxide (RGO)/ZnIn2S4 nanocomposites with superior photocatalytic performance. ACS Appl. Mater. Interfaces. 2014;6:3483-3490.
- [Google Scholar]
- High-performance overall water splitting electrocatalysts derived from cobalt-based metal–organic frameworks. Chem. Mater.. 2015;27:7636-7642.
- [Google Scholar]
- Metal–Organic Framework-Derived Co3ZnC/Co Embedded in Nitrogen-Doped Carbon Nanotube-Grafted Carbon Polyhedra as a High-Performance Electrocatalyst for Water Splitting. ACS Appl. Mater. Interfaces. 2018;10:6245-6252.
- [Google Scholar]
- Improving photocatalytic hydrogen production of metal–organic framework UiO-66 octahedrons by dye-sensitization. Appl. Catal. B. 2015;168:572-576.
- [Google Scholar]
- Three-dimensional N-doped carbon nanotube frameworks on Ni foam derived from a metal–organic framework as a bifunctional electrocatalyst for overall water splitting. ACS Appl. Mater. Interfaces. 2019;12:3592-3602.
- [Google Scholar]
- Recent advances in the metal–organic framework-based electrocatalysts for the hydrogen evolution reaction in water splitting: a review. RSC Adv.. 2021;11:21904-21925.
- [Google Scholar]
- Recent progress on engineering highly efficient porous semiconductor photocatalysts derived from Metal–organic frameworks. Nano-micro letters. 2019;11:1.
- [Google Scholar]
- Metal–Organic-Framework-Based Photocatalysts Optimized by Spatially Separated Cocatalysts for Overall Water Splitting. Adv. Mater.. 2020;32:2004747.
- [Google Scholar]
- Step-by-step improving photocatalytic hydrogen evolution activity of NH2–UiO-66 by constructing heterojunction and encapsulating carbon nanodots. ACS Sustainable Chem. Eng.. 2018;6:11563-11569.
- [Google Scholar]
- Applications of Atomically Dispersed Oxygen Reduction Catalysts in Fuel Cells and Zinc-Air Batteries. Energy & Environmental Materials. 2021;4:307-335.
- [Google Scholar]
- Two novel polyoxometalate-encapsulated metal–organic nanotube frameworks as stable and highly efficient electrocatalysts for hydrogen evolution reaction. ACS Appl. Mater. Interfaces. 2018;10:31498-31504.
- [Google Scholar]
- Co/Co9S8@ S, N-doped porous graphene sheets derived from S, N dual organic ligands assembled Co-MOFs as superior electrocatalysts for full water splitting in alkaline media. Nano Energy. 2016;30:93-102.
- [Google Scholar]
- Recent advances in metal-organic frameworks and their derived materials for electrocatalytic water splitting. ChemElectroChem. 2020;7:1805-1824.
- [Google Scholar]
- Bismuth single atoms resulting from transformation of metal–organic frameworks and their use as electrocatalysts for CO2 reduction. J. Am. Chem. Soc.. 2019;141:16569-16573.
- [Google Scholar]
- Metal–Organic Frameworks as Heterogeneous Electrocatalysts for Water Splitting and CO2 Fixation. Cryst. Growth Des.. 2021;21:3123-3142.
- [Google Scholar]
- Multifunctional high-activity and robust electrocatalyst derived from metal–organic frameworks. J. Mater. Chem. A. 2016;4:17288-17298.
- [Google Scholar]
- Ultrafine Co 3 O 4 nanolayer-shelled CoWP nanowire array: a bifunctional electrocatalyst for overall water splitting. RSC Adv.. 2020;10:29326-29335.
- [Google Scholar]
- ZIF-L-Co@ carbon fiber paper composite derived Co/Co3O4@ C electrocatalyst for ORR in alkali/acidic media and overall seawater splitting. Int. J. Hydrogen Energy. 2020;45:33028-33036.
- [Google Scholar]
- Designing MOF Nanoarchitectures for Electrochemical Water Splitting. Adv. Mater.. 2021;33:2006042.
- [Google Scholar]
- Small-sized Ni (1 1 1) particles in metal-organic frameworks with low over-potential for visible photocatalytic hydrogen generation. Appl. Catal. B. 2016;190:12-25.
- [Google Scholar]
- Bimetallic metal–organic frameworks and MOF-derived composites: Recent progress on electro-and photoelectrocatalytic applications. Coord. Chem. Rev.. 2022;451:214264
- [Google Scholar]
- Post-synthesis modification of a metal–organic framework to construct a bifunctional photocatalyst for hydrogen production. Energy Environ. Sci.. 2013;6:3229-3234.
- [Google Scholar]
- In situ growth of CdS nanoparticles on UiO-66 metal-organic framework octahedrons for enhanced photocatalytic hydrogen production under visible light irradiation. 2015;346:278-283.
- N-doped carbon-wrapped cobalt nanoparticles on N-doped graphene nanosheets for high-efficiency hydrogen production. Chem. Mater.. 2015;27:2026-2032.
- [Google Scholar]
- Microwave assisted synthesis of reduced graphene oxide incorporated MOF-derived ZnO composites for photocatalytic application. Catal. Commun.. 2017;88:5-8.
- [Google Scholar]
- Metal-organic frameworks and their derivatives as bifunctional electrocatalysts. Coord. Chem. Rev.. 2018;376:430-448.
- [Google Scholar]
- Expediting in-situ electrochemical activation of two-dimensional metal–organic frameworks for enhanced OER intrinsic activity by iron incorporation. ACS Catal.. 2019;9:7356-7364.
- [Google Scholar]







