Translate this page into:
Analysis and health risk assessment of heavy metals in four common seaweeds of Marchica lagoon (a restored lagoon, Moroccan Mediterranean)
⁎Corresponding author. Abderahmane.rahhou@ump.ac.ma (Abderrahmane RAHHOU)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
The Marchica lagoon (Morocco, Mediterranean) is a wetland classified as a RAMSAR site, which has been the subject of restoration activities after several years of pollution. The heavy metal (loïdes) content was studied in four common species of seaweed (Gracilaria sp., Alsidium corallinum, Caulerpa prolifera and Chaetomorpha sp.) for the first time, and was added to the basic studies carried out on sediments and water of this ecosystem. The heavy metals with mean values (mg/kg) can be sequenced in descending order, Al > Sr > Rb > Ba > As > Ti > V > Ni > Pb > Cr > Cd > Li > Co for Chaetomorpha sp., and the following order Al > Sr > As > Pb > Ti > Ba > Rb > V > Li > Cr > Co > Ni > Cd for Caulerpa prolifera. The mean concentrations (mg/kg) in Gracilaria sp. followed the sequence Al > Sr > Ti > Rb > Pb > As > Co > Ba > V > Ni > Cr > Li > Cd, and the following order Al > Sr > Ti > Rb > As > Ba > Pb > V > Co > Ni > Cr > Li > Cd in Alsidium corralinum. The Pb and As levels in studied seaweeds were above the CEVA standards. The health risk analysis showed that consuming the seaweed under study posed a serious risk. All examined seaweed had HI values greater than 1.0, and arsenic is the element that strongly contributes to these values, which can suggest a potential negative impact caused by this metalloid. Red seaweeds showed the highest MPI, followed by green seaweeds.
Keywords
Seaweeds
Health risk
Heavy metals
Marchica Lagoon
1 Introduction
Around the world, the production of macroalgae or seaweeds is increasing due to its many potential uses in the food and other sectors, such as animal feed, fertilizers, plant biostimulants, and as a source of bioactives for cosmetics and biomedical purposes(Araújo et al., 2021; FAO, 2018). Red algae (Rhodophyta), brown algae (Phaeophyceae), and green algae (Chlorophyta) are the basic groups into which marine macroalgae are classified (Apaydin et al., 2010). Depending on biological and environmental factors, species, area of origin and time of collection, the chemical composition and bioaccumulation of metals by macroalgae can vary considerably (Chen et al., 2018; Roleda et al., 2019; Véliz et al., 2023). Due to their high bioaccumulation capacity of heavy metals, including most toxic elements (As, Pb, Hg, and Cd), seaweed intake may also be strongly associated to a transfer of hazardous heavy metals to humans (Bouga and Combet, 2015; Ma et al., 2018; Olsson et al., 2020). Heavy metals and hazardous metalloids are considered as dangerous, endocrine-disrupting, and bioaccumulative metals (Kibria et al., 2021), other metals such as chromium, nickel, and aluminum, are essential in small doses, and toxics at excessive intake and exposure (Tepe and Dede, 2023; Yalçın Tepe, 2014). Through its Commission Recommendation (EU) 2018/ 464, the European Commission emphasized the necessity to monitor the levels of toxic metals in seaweeds used as food, food additives, and feed products. Regarding this, the European Commission has established maximum levels of (3.0 mg/kg, 0.1 mg/kg, 3 mg/kg) for Cd, Hg, and Pb respectively; inseaweeds used as food supplements made up entirely or primarily of dried seaweed. Regarding Arsenic in seaweed destined for animal feed, values of (40 mg/kg of total As) and (2 mg/kg for inorganic As) have been specified as maximum levels for animal (Lähteenmäki-Uutela et al., 2021). Seaweeds are crucial since they control the nutritional fluxes in marine habitats and effectively impact the water quality (Wilson, 2002). Heavy metals sources are primarily natural and crustal and, to a minor extent, anthropogenic (Kodat and Tepe, 2023). Due to human activities such as agriculture, industrial and domestic discharges, heavy metals are transported to aquatic environments (Baghour, 2017). Macroalgae have been utilized around the world to the monitoring of heavy metal pollution in aquatic ecosystems (Anbazhagan et al., 2021; Baghour, 2017; Conti and Cecchetti, 2003; Farias et al., 2017; Skalli et al., 2019). Current findings revealed the presence of heavy metals in several taxa of seaweed from various regions of the world (Ali et al., 2021; Augustsson et al., 2021; Chen et al., 2021; El-Said, 2013; Filippini et al., 2021; Hossain et al., 2022; Jayasekera and Rossbach, 1996; Khandaker et al., 2021; Öbek et al., 2021; Paz et al., 2019; Peng et al., 2022). The majority of seaweeds are found growing on sediments in benthic environments near coastlines where heavy metal concentrations are frequently high (Aknaf et al., 2022; Gao et al., 2012; Oujidi et al., 2021). In addition, sediments in the marine environment are well recognized for being the main repository for heavy metals and for being essential in the release and transportation of metals (Kodat and Tepe, 2023; Pan and Wang, 2012). As a result, seaweeds can absorb heavy metals from adjacent waters and sediments (Hurd et al., 2014), and in other instances, they were reported to bioaccumulate metal levels greater than those in seawater (Çelik and Oehlenschläger, 2007).
For the present study, it is considered that the aquatic environment of the lagoon of Marchica (NE-Morocco, Mediterranean) has been subjected to a strong natural and anthropic pressure (wastewater discharges, deposits of old iron mine, and agriculture) and storm water runoff for several years and that waters of the ecosystem in which macroalgae grow have been impacted in several parts (Oujidi et al., 2020; Aknaf et al., 2022). While it is clear that the effect of metal pollution on the environment must be analyzed, some studies have been carried out on the contamination of water and sediments by heavy metals (Oujidi et al., 2020; Aknaf et al., 2022), other studies have been pointed on macroalgae (Ramdani et al., 2015), and microalgae (Riouchi et al., 2022). (Kassi̇la et al., 2019) studied the seaweed (Gracilaria gracilis, Rhodophyta) farming in Nador lagoon as an additional activity for fishermen community. No research has been conducted on the accumulation of hazardous pollutants such as metals, in most common seaweeds growing in the Marchica Lagoon. Consequently, the current study will focus on the amount of heavy metal pollution in naturally occurring macroalgae in the Marchica lagoon.
This study aimed to determine the content of non-essential metal-(loids) (Li, Co, Ti, V, Cr, Al, Ni, Sr, Rb, Cd, Pb, Ba, and As) in four species of seaweeds from Marchica Lagoon, and the health risk assessments using targeted hazard quotient (THQ) and hazard index (HI) (Ali et al., 2021; Arisekar et al., 2021; Töre et al., 2021) due to consumption of these seaweeds as food and feed. These taxa include to the green seaweeds, Caulerpa prolifera and Chaetomorpha sp.; and the red seaweeds Gracilaria sp., and Alsidium corallinum. Therefore, some non-consumable seaweed might be able to be farmed along the coasts of Marchica lagoon to decrease pollution levels.
2 Materials and methods
2.1 Study area
With a length of 25 km, a width of 7.5 km, and an approximate area of 115 km2, Marchica lagoon (NE-Morocco, Mediterranean) is one of the biggest coastal lagoons in the Mediterranean Sea. It is located on the north-eastern coast of Morocco between the “Cap des trois fourches” and the “Cap de l'eau”, more precisely between latitudes 35°05′N −35°14′N and longitudes 2°44′W − 2°56′W (Fig. 1). It is separated from the sea by a sand bar (25 km). In 2010, a new inlet was built. It's about 300 m wide and 6.5 m deep, in order to improve exchange of the lagoon's waters with those of the Mediterranean Sea.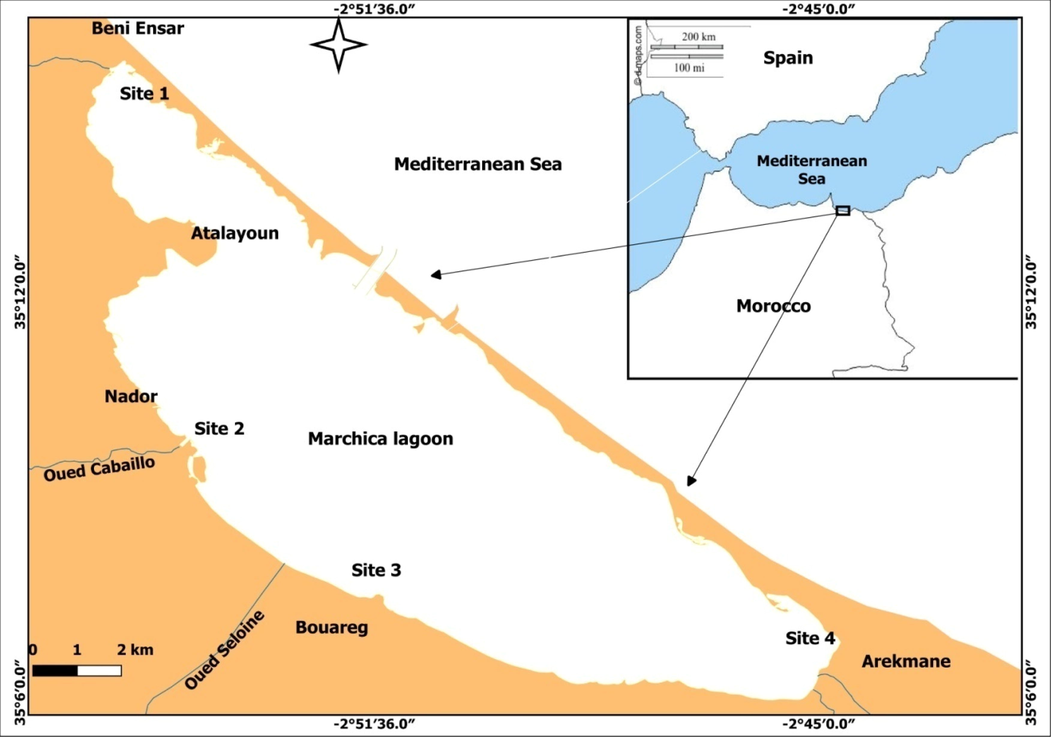
Map of the study area and sampling’s locations.
2.2 Samples collection and preparation
Fresh seaweed samples were collected from the bottom by diving from the main locations containing naturally occurring seaweeds in the Marchica Lagoon (Fig. 1). A sampling campaign was carried out in October 2021. On-site, the samples were washed with seawater, then placed into different plastic bags and transported to the laboratory. The seaweeds were cleaned with distilled water to remove any epiphytes and attached objects. The samples were then cut into smaller pieces, and exposed to air to ensure proper drying and to facilitate subsequent grinding. To eliminate water, the samples were dried in a programmed closed system oven set to 60 °C,until the material is at a constant dry weight (Żbikowski et al., 2007).The samples comprised of two groups viz. red algae Gracilaria sp.(from site 3) and Alsidium corallinum (from site 2), and green algae Caulerpa prolifera (from site 4) and Chaetomorpha sp. (from site1) were collected. Three independent sampling stations were used for each taxon and each site described in the Fig. 1. The site 1 is characterized by the seagrass meadow in the bottom and the Chaeotomorpha sp. in the surface. The site 3 is characterized by the cultivation of the Gracilaria sp, while Alsidium corralinium is the only seaweed that occurred in the site 2. The site 4 is characterized by the Caulerpa prolifera meadow. The choice of sampling sites is based on the naturally occurrence of these algae, with significant abundance and coverage.
2.3 Sample analysis
All used reagents were “For analysis” grade. HNO3 (65%), H2O2 (30%), and HCl (37%) were used for the digestion of seaweeds. A Sigma Aldrich multi-element stock standard solution (Periodic Table Mix 1 for ICP) 10 mg/L was used to prepare working standards.
For the digestion of samples, a high-pressure laboratory microwave (MW) oven (Milestone Ethos UP, Italy) was used (Majer et al., 2014). Weighed samples were transferred to 50 mL Teflon vessels of the MW system and a mixture of 8 mL concentrated nitric acid and 2 mL hydrogen peroxide was added. The MW digestion has been run with the following program: 30 min ramp to 210 °C and 20 min hold at 210 °C (both 1800 W). The digested samples were transferred in volumetric flasks, 2 mL concentrated HCl was added and samples were diluted to a final volume of 50 mL with MilliQ water. Blank samples were passed through the same procedure. The digestion procedure was performed in duplicate for each algae sample.
The chemical elements determination was carried out using an Agilent 5100 simultaneous vertical dual view (ICP-OES). Instrumental parameters for ICP-OES measurements of studied elements were as follow: RF incident power (1.2 kW); plasma argon flow rate (12 Lmin−1); auxiliary argon flow rate (1 Lmin−1); nebulizer argon flow rate (0.7 Lmin−1); nebulizer (concentric), spray chamber (cyclonic double pass); axial viewing, three replicates.
To establish the accuracy of the method, a recovery test was conducted. Prior to digestion, all samples were spiked with a precise quantity of standard solution of the analyzed elements. The spiked samples underwent the same digestion and analysis conditions as the non-spiked samples. The recoveries observed in the spiked samples fell within the range of 91% to 107%. Detailed results from the recovery test can be found in Table 1.
Element
Added, mg kg−1
Recovery, %
RSD, %
Li
0.5
91
5
Cd
0.5
103
6
Co
1
93
4
Cr
1
102
2
Ti
5
105
5
V
5
96
2
Ni
5
95
4
Ba
5
97
6
Pb
5
102
3
As
5
93
4
Rb
5
94
5
Sr
20
107
5
Al
20
102
3
2.4 Health risk assessment
Based on exposure doses, the targeted hazard quotient (THQ), and hazard index (HI) established by the US EPA's IRIS database, a possible health risk assessment was estimated (Ali et al., 2021; Arisekar et al., 2021; US EPA, 2013).
The Metal Pollution Index (MPI) was used to measure the quantities of various metals in all studied seaweed species (Giusti, 2001; Rajaram et al., 2020).
Where Cfi = Concentration (mg/kg) for the metal “i” in the sample. And “n’’ the number of investigated metals.
The HI was determined by comparing the mean metal concentrations in the current study's seaweed samples to worldwide standards set by the US EPA. The total of all THQs for all metals is defined as HI. The formulae are as follows (Ali et al., 2021; Arisekar et al., 2021; US EPA, 2013):
Targeted Hazard Quotient (THQ) = Exposure Dose/RfD.
Hazard Index (HI) = Sum of THQ.
EDi: Exposure dose of the metal “i”.
Ci = mean concentration of the heavy metal “i” in the seaweed samples (mg kg-1).
Di: daily intake of seaweeds (e.g., 5.2 g/capita/day).
Ed: average exposure duration (e.g., 70 years).
Bw: average weight (e.g., 70 kg).
At: average lifetime (e.g., 70 years).
RfD: recommended reference dose (RfD).
Based on the US EPA (2013) guidelines HI < 1 seems to no health risk are expected to occur.
If HI ≥ 1, there is moderate or high risk for adverse human health effects (Ali et al., 2021; Arisekar et al., 2021).
2.5 Statistics
The significant difference between the heavy metals concentrations among the studied seaweeds with respect to species was evaluated by one-way ANOVA. Subsequently, Pearson’s correlation analysis was conducted to explore the relationship between investigated elements in all studied seaweeds. ANOVA and Pearson’s correlation analysis were performed in SPSS 26.0 software. The map of the lagoon was produced with QGIS 2.18 software.
3 Results and discussion
3.1 Heavy metals and MPI contents of studied seaweeds
In the present study, the statistical analysis indicated that levels of Sr, Rb, Li, and Cd varied significantly among all the studied species (P < 0.05); however, there were no differences in the concentrations of the other examined elements. In this study, Cd was detected only in the species Chaetomorpha sp., and Co detected only in red seaweeds. The heavy metals and metalloids with mean values (mg/kg) (Table 2) can be sequenced in descending order, Al > Sr > Rb > Ba > As > Ti > V > Ni > Pb > Cr > Cd > Li > Co for Chaetomorpha sp., and the following order Al > Sr > As > Pb > Ti > Ba > Rb > V > Li > Cr > Co > Ni > Cd, for Caulerpa prolifera. The mean concentrations (mg/kg) for heavy metals and metalloids present in Gracilaria sp. followed the sequence Al > Sr > Ti > Rb > Pb > As > Co > Ba > V > Ni > Cr > Li > Cd, and the following order Al > Sr > Ti > Rb > As > Ba > Pb > V > Co > Ni > Cr > Li > Cd in Alsidium corralinum. nd: not detected; *: significant variation between species; n = number of samples.
Li
Co
Ti
V
Cr
Al
Ni
Sr*
Ba
Pb
Cd*
As
Rb*
Caulerpa prolifera (n = 6)
0.3 ± 0.12
nd
7.2 ± 5.19
2.2
nd
261.33 ± 207.69
nd
126.03 ± 34.81
4.06 ± 0.23
7.65 ± 7.14
nd
11.80 ± 2.1
4.05 ± 2.47
Alsidium corallinum(n = 6)
1.04 ± 0.28
5.05 ± 0.91
39.63 ± 14.83
5.66 ± 2.55
1.15 ± 0.49
1617.66 ± 427.84
4.55 ± 0.07
77.06 ± 31.17
9.53 ± 3.58
8.1
nd
10.6
26.33 ± 5.13
Gracilaria sp. (n = 6)
1.06 ± 0.71
10.2 ±
31.96 ± 26.12
5.36 ± 5.25
2.75 ± 0.49
1563 ± 1042.58
4.4 ± 3.67
100.63 ± 95.09
9.00 ± 7.97
16.2
nd
12.04 ± 3.89
25.06 ± 5.96
Chaetomorpha sp. (n = 6)
0.28 ± 0.07
nd
10.63 ± 2.71
3.63 ± 0.37
1.70 ± 0.6
1652.33 ± 2406.78
3.33 ± 0.56
368.66 ± 80.13
16.03 ± 2.00
2.86 ± 0.76
0.3 ± 0
12.07 ± 2.52
20.23 ± 3.45
Heavy metal bioaccumulation in seaweeds has been observed to be highly variable between species (Malea and Kevrekidis, 2014; Rajaram et al., 2020; Sun et al., 2019). Some species potentially take metals directly from sediment via their thallus and rhizoids, which have significantly greater metal concentrations than the water column (Żbikowski et al., 2006).
In this regard, results of (Aknaf et al., 2022; Oujidi et al., 2021) show that the surface sediments and surface waters of Marchica lagoon are rich in trace metals and that the most impacted areas are Beniensar (Site 1), Arekmane (Site 4), the mouth of Wadis and wastewater treatment plant discharges area. In addition, the mineralogical composition of deposits from iron mine of Nador contains significant quantities of accessory metals such as Ni, Cr, Pb, and Cd (Oujidi et al., 2021).
When the metals contents in selected seaweeds was assessed using the MPI, red seaweeds showed the highest MPI following this order; Alsidium corralinum (26.21) > Gracilaria sp.(24.07) > Chaeotomorpha sp.(17.38) > Caulerpa prolifera (16.36). Heavy metals and macroalgae have a relationship based on their ability to absorb heavy metals, which is dependent on cell wall polysaccharides (Karthick et al., 2012).
3.2 Health risk assessment
A targeted hazard quotient (TQH) for each element was determined using 5.2 g dry weight of seaweed meal per day (as a dietary item) and the Recommended Reference Dose (RfD) for heavy metals as a reference value (Table 2).The TQH was<1.0 for all elements except As, suggesting that the predicted exposure doses was less than the RfD set by the US EPA. Additionally, when the hazard index (HI) was computed with mean metal levels included, all seaweeds had HI values more than 1.0. Arsenic contributes more than any other element to high values of HI, which may suggest a possible hazardous effect related to this element (Table 3). Moreover, the HI values of examined seaweed species were ordered in declining order as follows: Gracilaria sp. > Chaetomorpha sp. > Caulerpa prolifera > Alsidium corallinum. nd: not determined. Different seaweed species exhibited varying preferred uptake of particular metals. The Pb level in red seaweeds and Caulerpa prolifera revealed in our study exceeds the limits imposed by organizations such as (CEVA, 2019).
RfD (µg/g/day)
Elements
Li
Al
V
Cr
Sr
Co
Ni
As
Ba
Pb
Cd
0.002
1.0
0.009
0.003
0.6
0.02
0.02
0.0003
0.2
0.004
0.001
THQ for each element and TH in studied seaweeds
THQ
Li
Al
V
Cr
Sr
Co
Ni
As
Ba
Pb
Cd
HI= ∑THQ
Caulerpa prolifera
0.016
0.019
0.018
nd
0.016
nd
nd
2.922
0.002
0.142
nd
3.13
Alsidium corallinum
0.039
0.12
0.047
0.028
0.01
0.019
0.017
2.625
0.004
0.15
nd
3.06
Gracilaria sp.
0.039
0.116
0.044
0.068
0.012
0.038
0.016
2.983
0.003
0.301
nd
3.62
Chaetomorpha sp.
0.011
0.123
0.03
0.042
0.046
nd
0.012
2.989
0.006
0.053
0.022
3.33
The Cd contents in the examined seaweeds (0.5 mg kg −1) were within the limits specified by (CEVA, 2019). Therefore, the arsenic’s levels revealed in our study for all tested seaweeds were greater than the CEVA recommended limits. The high levels of arsenic, which is considered to be a limiting factor causing a health risk, is mainly from natural sources, with even small anthropogenic contributions. The Marchica lagoon watershed, and principally the Gourougou region, is characterized by volcanic rocks (El Bakkali et al., 1998) and it’s well known that mining deposits of old mine site of Nador has been dumped into the lagoon for many years (Oujidi et al 2021). Hence, arsenic in various forms is a main constituent of more than hundreds of ores, namely iron oxides and most metamorphic rocks (Garelick et al., 2009). (Xu et al., 2023) reported high concentrations of arsenic in the natural system near the vanadium-tellurium-iron mine. Likewise,(Swartjes and Janssen, 2016) found naturally occurring arsenic in iron ore lumps, which was used to build an artificial peninsula. Pearson’s correlations (parametric) for heavy metals concentrations in all studied algae are shown in the Table 4. Significant positive correlations (p < 0.05) were found between studied elements such as the following combinations: NiCr, CrV, CrAl, NiAl, RbCo, RbAl, BaAl and BaV. Highly significant positive correlations (p < 0.01) were observed between the following elements: TiLi, VLi, VTi, CoLi, CoTi, CoV, NiLi, NiTi, CdSr, and NiCo. No correlations observed between As and Pb with the other elements studied. These findings suggested that the combined two metals exhibited comparable bioaccumulation behavior in seaweeds (Peng et al., 2022). Results of (Arisekar et al., 2021) showed positive correlations between these combinations CoCr; NiCr; AsCr, AsNi, CdCr, PbAs, PbCr, and PbNi in the red seaweeds, and showed also, positives correlations between As and (Co, Ni, Pb, and Cr) in the green seaweeds. *. Significance at 0.05 level; **. Significance at 0.01 level.
Li
Al
Ti
V
Cr
Sr
Co
Ni
As
Rb
Ba
Pb
Cd
Li
1
Al
0.506
1
Ti
0.974**
0.505
1
V
0.913**
0.576
0.929**
1
Cr
0.54
0.631*
0.45
0.613*
1
Sr
−0.226
0.384
−0.2
0.14
0.438
1
Co
0.875**
0.421
0.921**
0.903**
0.448
−0.086
1
Ni
0.780**
0.642*
0.814**
0.942**
0.707*
0.358
0.802**
1
As
−0.236
0.084
−0.292
−0.1
0.385
0.529
−0.139
0.008
1
Rb
0.788**
0.599*
0.764**
0.823**
0.573
0.095
0.623*
0.830**
−0.364
1
Ba
0.384
0.624*
0.426
0.692*
0.714**
0.754**
0.489
0.869**
0.23
0.604*
1
Pb
−0.462
−0.304
−0.491
−0.56
−0.443
−0.282
−0.408
−0.544
−0.095
−0.248
−0.487
1
Cd
−0.263
0.396
−0.252
0.02
0.386
0.838**
−0.238
0.231
0.383
0.159
0.537
−0.07
1
4 Comparative study with other works
In our study, red seaweeds accumulated great contents of Pb, Ni, Co, V, Rb, Cr, Ti and Li. While, green seaweeds accumulated Cd, As, Sr, and Al. In contrast, results of (Chakraborty et al., 2014) indicated that Rhodophyta species in general bound and accumulated less Cr than other algae groups (i.e., Chlorophyta and Phaeophyceae), and results of (Peng et al., 2022) stated that green algae had higher contents of Pb than other phylum, which may have been attributed to environmental factors. This disagreement is probably due to spatial variation of heavy metal in waters and sites collection of seaweeds, as previously demonstrated by (Khairy and El-Sheikh, 2015) which indicated that the comparatively high levels of several heavy metals in macroalgae imply their high amounts in the waters of the investigated locations as well as the algae's potential to absorb them. Tables (5, 6, and 7) compare the values revealed in our study to a selection of studies around the world.
In the matter of green seaweed, when the current data was compared to those previously published by several authors. For Caulerpa prolifera (Table 5), it was shown that (Arisekar et al., 2021; Intawongse et al., 2018) reported lower values of As and Pb along the marine coastal of India and Thailand than that reported in this study, but higher values of Cd, Cr, Co and Ni. Higher values of Cd, and Pb reported by (Sanchiz et al., 2000, 1999) along the East coast and Mar Menor lagoon (Spain). On the other hand, our findings, showed greater levels of Pb and As but lower levels of Co, Cd, and Ni than those reported by (Al-Shwafi and Rushdi, 2008) along the gulf of Aden. nd: not detected.
Study area
As
Ba
Cr
Ni
Cd
Pb
Co
V
Sr
Al
Reference
And man coast of Thailand
–
–
–
–
0.89
0.97
–
–
–
–
(Intawongse et al., 2018)
Gulf of Mannar, India
0.25
–
0.14
0.2
0.07
0.07
0.05
–
–
–
(Arisekar et al., 2021)
the Gulf of Aden
–
–
13.2
5.3
0.93
2.5
0.8
–
–
–
(Al-Shwafi and Rushdi, 2008)
Palk Bay coast, India
–
–
–
–
4.13
10.54
–
–
–
–
(Rajaram et al., 2020)
East Coast of Spain
–
–
–
–
–
23
–
–
–
–
(Sanchiz et al., 1999)
Mar Menor, spain
–
–
–
–
0,20
37
–
–
–
–
(Sanchiz et al., 2000)
Marchica lagoon
11.8
4.07
nd
nd
nd
7.65
nd
2.2
126
261
This study
Chaetomorpha sp. in the present study recorded lower values for Pb than that reported by (Wong et al., 1979) for algae collected from the Holo Harbor in Hong Kong. (Arulkumar et al., 2019; Rajaram et al., 2020) detected higher levels for Cd, and Pb in Palk Bay coast (India) than that reported in the present study (Table 6). (Al-Homaidan, 2007) detected higher values for Ni, Cd and Pb in algae from Saudi coast Arabian Gulf than that reported in the present study. Chaeotomorpha sp. in this study recorded higher values of Sr, Pb, Cd, and As but lower concentrations of Co, and Ni than that collected from fish pond aquaculture in Portugal by (Afonso et al., 2018). nd: not detected.
Study area
As
Ba
Cr
Ni
Cd
Pb
Co
V
Sr
Al
Reference
fish pond aquaculture (Portugal)
6.37
–
5.49
1.83
0.02
1.66
0.62
–
59
–
(Afonso et al., 2018)
Saudi coast Arabian Gulf
–
–
–
44.13
1.12
30.5
–
–
–
–
(Al-Homaidan, 2007)
Tolo Harbour, Hong Kong
–
–
–
–
–
24
–
–
–
–
(Wong et al., 1979)
coast of Palk Bay, India
–
–
–
–
8.51
5.24
–
–
–
–
(Arulkumar et al., 2019)
Palk Bay coast, India
–
–
–
–
0.57
9
–
–
–
–
(Rajaram et al., 2020)
Marchica Lagoon
12.1
16.03
1.7
3.33
0.3
2.87
nd
3.63
368.6
1652
This study
Regarding red seaweeds, comparison of the contents of As, Cr, Pb, and Ni in the present study indicated that Gracilaria sp. (Table 7) showed higher levels than those published in the literature by (Arisekar et al., 2021; Caliceti et al., 2002; Malea and Kevrekidis, 2014). On the other hand, Gracilaria sp. reported a lower concentrations of Ni, Cd, and Pb than that recorded by (Khaled et al., 2014) (Egyptian coast) and lower value of Cr, Cd, and Ni than that detected by (Ali et al., 2021) (Sudanese Red Sea). While it recorded a higher levels of As in comparison with that collected from the Egyptian coast (El-Said, 2013), Gracilaria sp. reported lower values of Ni, Cd, and Co. nd: not detected.
Study area
As
Ba
Cr
Ni
Cd
Pb
Co
V
Sr
Al
Reference
Aegean Sea greece
–
–
–
–
0.49
8.6
–
–
–
–
(Boubonari et al., 2008)
Egyptian Sea
–
–
–
7.82
0.26
41.16
–
–
–
–
(Khaled et al., 2014)
Venice Lagoon Italy
15
–
0.7
1.1
0.6
6.9
–
–
–
–
(Caliceti et al., 2002)
Gulf of Mannar, South India
1.19
–
1.17
1.06
0.26
0.5
0.37
–
–
–
(Arisekar et al., 2021)
Northern Aegean Sea
8.42
94.46
2.77
2.19
0.08
5.13
0.93
10.06
53.09
(Malea and Kevrekidis, 2014)
Sudanese Red Sea
–
–
12
8.0
0.082
1.02
–
–
–
–
(Ali et al., 2021)
Egyptian coast
3.1
–
–
9.1
12.4
–
16.8
–
–
–
(El-Said, 2013)
Portuguese coast
17.57
–
0.534
1.48
0.065
1.12
1.534
3.8
–
196.17
(Cabrita et al., 2016)
Marchica Lagoon
12.05
9
2.75
4.4
nd
16.2
10.2
5.37
100.63
1563
This study
5 Conclusion
This study is the first baseline to assess the hazards and risks associated with ingesting naturally occurring green and red seaweeds in Marchica lagoon (NE-Morocco, Mediteranean). This baseline study, which focused on the bioaccumulation of a total of 13 elements, including toxic heavy metals, by different algal species, confirmed the practical utility and importance of algae in the decontamination of Marchica lagoon through their potential for metals’ uptake. It showed that investigated seaweeds contain various concentrations of metals, with specific-species degree of accumulation. Considering the research area's coastal urbanization and anthropogenic pressure, as well as the associated discharges; consumption of these seaweeds was shown to constitute a significant risk to human health, while the bioaccumulation of toxic metals was found below the hazard quotient (THQ < 1), Arsenic was considered to be the main element contributing to high values of HI (THQ greater than 1 for As). Red seaweeds recorded high values of the metal pollution index (MPI) than green seaweeds. Therefore, we suggested that the cultivation of macroalgae in the Marchica lagoon has the potential of heavy metals uptake in this coastal area, and can be subsequently valued in fields that do not cause risks to human health such as energy recovery (El Asri et al., 2017). Moreover, a further investigation of other elements like mercury, and arsenic’s speciation (organic and inorganic) would be helpful to highlight the real hazardous effect of these elements.
Author contributions
[M.A], [M.B], and [A.R] contributed to the study conception and design; [B.O], [N.E], and [M.B] for supervision. Material preparation, data collection and analysis were performed by [A.S],[A.R],[ML],[A.A],[N.E],[M.K ], [J.I], and [P.P]. The first draft of the manuscript was written by [A.R],[M.L], and [MB] and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Composition and bioaccessibility of elements in green seaweeds from fish pond aquaculture. Food Res. Int.. 2018;105:271-277.
- [CrossRef] [Google Scholar]
- The chemical characterization and its relationship with heavy metals contamination in surface sediment of Marchica Mediterranean Lagoon (North of Morocco) Environ. Sci. Pollut. Res.. 2022;29:4159-4169.
- [CrossRef] [Google Scholar]
- Al-Homaidan, 2007. Heavy metal concentrations in three species of green algae from the Saudi coast of the Arabian Gulf. Int. j. food, agric. environ. (Print) 5, 354–358.
- Bioaccumulation and health risk assessment of toxic metals in red algae in Sudanese Red Sea coast. Toxin Rev.. 2021;40:1327-1337.
- [CrossRef] [Google Scholar]
- Heavy metal concentrations in marine green, brown, and red seaweeds from coastal waters of Yemen, the Gulf of Aden. Environ. Geol.. 2008;55:653-660.
- [CrossRef] [Google Scholar]
- Health risk assessment and bioaccumulation of metals in brown and red seaweeds collected from a tropical marine biosphere reserve. Mar. Pollut. Bull.. 2021;164:112029
- [CrossRef] [Google Scholar]
- Apaydin, G., Aylikci, V., Cengi̇z, E., Saydam, M., Küp, N., Tiraşoğlu, E., 2010. Analysis of Metal Contents of Seaweed (Ulva lactuca) from Istanbul, Turkey by EDXRF. Turkish Journal of Fisheries and Aquatic Sciences 10, 215–220. https://doi.org/10.4194/trjfas.2010.0209.
- Current status of the algae production industry in Europe: An emerging sector of the blue bioeconomy. Front. Mar. Sci.. 2021;7
- [CrossRef] [Google Scholar]
- Heavy metal concentrations in the macroalgae, seagrasses, mangroves, and crabs collected from the Tuticorin coast (Hare Island), Gulf of Mannar, South India. Mar. Pollut. Bull.. 2021;163:111971
- [CrossRef] [Google Scholar]
- Metals accumulation in edible marine algae collected from Thondi coast of Palk Bay, Southeastern India. Chemosphere. 2019;221:856-862.
- [CrossRef] [Google Scholar]
- Trace and major elements in food supplements of different origin: Implications for daily intake levels and health risks. Toxicol. Rep.. 2021;8:1067-1080.
- [CrossRef] [Google Scholar]
- The red macroalga Gracilaria bursa-pastoris as a bioindicator of metals (Fe, Zn, Cu, Pb, Cd) in oligohaline coastal environments. Fresenius Environ. Bull.. 2008;17:2207-2216.
- [Google Scholar]
- Emergence of seaweed and seaweed-containing foods in the UK: Focus on labeling, iodine content, toxicity and nutrition. Foods. 2015;4:240-253.
- [CrossRef] [Google Scholar]
- Tracing seaweeds as mineral sources for farm-animals. J. Appl. Phycol.. 2016;28:3135-3150.
- [CrossRef] [Google Scholar]
- Heavy metal contamination in the seaweeds of the Venice lagoon. Chemosphere. 2002;47:443-454.
- [CrossRef] [Google Scholar]
- High contents of cadmium, lead, zinc and copper in popular fishery products sold in Turkish supermarkets. Food Control. 2007;18:258-261.
- [CrossRef] [Google Scholar]
- CEVA , 2019. Centre d’Etude et de Valorisation des Algues: Synthèse règlementaire - algues alimentaires:Revue actualisée du statut réglementaire des algues alimentaires et microalgues en France et en Europe (ingrédients, compléments alimentaires, Novel Food) [WWW Document]. URL https://www.ceva-algues.com/document/synthese-reglementaire-algues-alimentaires/ (accessed 9.20.22).
- Benthic macroalgae as biological indicators of heavy metal pollution in the marine environments: A biomonitoring approach for pollution assessment. Ecotoxicol. Environ. Saf.. 2014;100:61-68.
- [CrossRef] [Google Scholar]
- An investigation of toxic metal levels (Pb, Cd, Cr, As, Hg) in dried porphyra and laminaria collected from coastal cities, China. Biol. Trace Elem. Res.. 2021;199:3987-3997.
- [CrossRef] [Google Scholar]
- Distribution of metals and metalloids in dried seaweeds and health risk to population in southeastern China. Sci. Rep.. 2018;8:1-7.
- [CrossRef] [Google Scholar]
- A biomonitoring study: trace metals in algae and molluscs from Tyrrhenian coastal areas. Environ. Res.. 2003;93:99-112.
- [CrossRef] [Google Scholar]
- Comparison of energy recovery after anaerobic digestion of three Marchica lagoon algae (Caulerpa prolifera, Colpomenia sinuosa, Gracilaria bursa-pastoris) Sustain. Mater. Technol.. 2017;11:47-52.
- [CrossRef] [Google Scholar]
- El Bakkali, S., Bourdier, J.-L., Gourgaud, A., 1998. Caractérisation et stratigraphie de dépôts volcanoclastiques marqueurs dans le Miocène supérieur du bassin de Melilla-bas Kert (Rif oriental, Maroc). Comptes Rendus de l’Académie des Sciences - Series IIA - Earth and Planetary Science 327, 93–100. https://doi.org/10.1016/S1251-8050(98)80038-4.
- Bioaccumulation of key metals and other contaminants by seaweeds from the Egyptian Mediterranean sea coast in relation to human health risk. Hum. Ecol. Risk Assess. Int. J.. 2013;19:1285-1305.
- [CrossRef] [Google Scholar]
- FAO, 2018. The State of World Fisheries and Aquaculture: Sustainability in Action. Security Research Hub Reports.
- In situ assessment of Ulva australis as a monitoring and management tool for metal pollution. J. Appl. Phycol.. 2017;29:2489-2502.
- [CrossRef] [Google Scholar]
- Heavy metals and potential risks in edible seaweed on the market in Italy. Chemosphere. 2021;263:127983
- [CrossRef] [Google Scholar]
- Geochemistry of organic carbon and nitrogen in surface sediments of coastal Bohai Bay inferred from their ratios and stable isotopic signatures. Mar. Pollut. Bull.. 2012;64:1148-1155.
- [CrossRef] [Google Scholar]
- Garelick, H., Jones, H., Dybowska, A., Valsami-Jones, E., 2009. Arsenic Pollution Sources, in: Reviews of Environmental Contamination Volume 197, Reviews of Environmental Contamination and Toxicology. Springer New York, New York, NY, pp. 17–60. https://doi.org/10.1007/978-0-387-79284-2_2.
- Heavy metal contamination of brown seaweed and sediments from the UK coastline between the Wear river and the Tees river. Environ. Int.. 2001;26:275-286.
- [CrossRef] [Google Scholar]
- Trace element bioaccumulation in edible red seaweeds (Rhodophyta): A risk assessment for consumers. Environ. Pollut.. 2022;307:119560
- [CrossRef] [Google Scholar]
- Hurd, C.L., Lobban, C.S., Bischof, K., Harrison, P.J. (Eds.), 2014. Nutrients, in: Seaweed Ecology and Physiology. Cambridge University Press, Cambridge, pp. 238–293. https://doi.org/10.1017/CBO9781139192637.007.
- Bioaccessibility of heavy metals in the seaweed Caulerpa racemosa var. corynephora: Human health risk from consumption. Instrum. Sci. Technol.. 2018;46:628-644.
- [CrossRef] [Google Scholar]
- Use of seaweeds for monitoring trace elements in coastal waters. Environ. Geochem. Health. 1996;18:63-68.
- [CrossRef] [Google Scholar]
- Ecological implications of trace metals in seaweeds: Bio-indication potential for metal contamination in Wandoor, South Andaman Island. Egypt. J. Aquat. Res.. 2012;38:227-231.
- [CrossRef] [Google Scholar]
- Kassi̇la, J., Nhhala, H., Gi̇vernaud, T., Monsouri̇, M., Abrehouch, A., Mosfi̇oui̇, A., 2019. Opportunities for the development of seaweed farming as a supplementary income for small-scale fishermen in Nador lagoon: Experimental cultivations of Gracilaria gracilis (Stackhouse). Mediterranean Fisheries and Aquaculture Research 2, 12–26.
- Antioxidant activity and mineral composition of three Mediterranean common seaweeds from Abu-Qir Bay, Egypt. Saudi J. Biol. Sci.. 2015;22:623-630.
- [CrossRef] [Google Scholar]
- Distribution of heavy metals in seaweeds collected along Marsa-Matrouh beaches, Egyptian Mediterranean Sea. Egypt. J. Aquat. Res.. 2014;40:363-371.
- [CrossRef] [Google Scholar]
- Elevated concentrations of metal(loids) in seaweed and the concomitant exposure to humans. Foods. 2021;10:381.
- [CrossRef] [Google Scholar]
- Climate change impacts on pollutants mobilization and interactive effects of climate change and pollutants on toxicity and bioaccumulation of pollutants in estuarine and marine biota and linkage to seafood security. Mar. Pollut. Bull.. 2021;167:112364
- [CrossRef] [Google Scholar]
- A holistic approach to the assessment of heavy metal levels and associated risks in the coastal sediment of Giresun, southeast Black Sea. Heliyon. 2023;9:e16424.
- [Google Scholar]
- European Union legislation on macroalgae products. Aquac. Int.. 2021;29:487-509.
- [CrossRef] [Google Scholar]
- Total and inorganic arsenic contents in seaweeds: Absorption, accumulation, transformation and toxicity. Aquaculture. 2018;497:49-55.
- [CrossRef] [Google Scholar]
- Majer, A.P., Petti, M.A.V., Corbisier, T.N., Ribeiro, A.P., Theophilo, C.Y.S., Ferreira, P.A. de L., Figueira, R.C.L., 2014. Bioaccumulation of potentially toxic trace elements in benthic organisms of Admiralty Bay (King George Island, Antarctica). Marine Pollution Bulletin 79, 321–325. https://doi.org/10.1016/j.marpolbul.2013.12.015.
- Trace element patterns in marine macroalgae. Sci. Total Environ.. 2014;494–495:144-157.
- [CrossRef] [Google Scholar]
- Assessment of potential health risk associated with the use of Cladophora fracta as mulch. Environ. Geochem. Health. 2021;43:2175-2191.
- [CrossRef] [Google Scholar]
- Biochemical composition of red, green and brown seaweeds on the Swedish west coast. J. Appl. Phycol.. 2020;32:3305-3317.
- [CrossRef] [Google Scholar]
- Effects of the watershed on the seasonal variation of the surface water quality of a post-restoration coastal wetland: The case of the Nador lagoon (Mediterranean sea, Morocco) Reg. Stud. Mar. Sci.. 2020;35:101127
- [CrossRef] [Google Scholar]
- Seasonal and spatial patterns of ecotoxicological indices of trace elements in superficial sediments of the marchica lagoon following restoration actions during the last decade. Diversity. 2021;13:51.
- [CrossRef] [Google Scholar]
- Trace metal contamination in estuarine and coastal environments in China. Sci. Total Environ. Special Sect.: Rev. Trace Metal Pollut. China. 2012;421–422:3-16.
- [CrossRef] [Google Scholar]
- Toxic metals (Al, Cd, Pb and Hg) in the most consumed edible seaweeds in Europe. Chemosphere. 2019;218:879-884.
- [CrossRef] [Google Scholar]
- Species-specific bioaccumulation and health risk assessment of heavy metal in seaweeds in tropic coasts of South China Sea. Sci. Total Environ.. 2022;832:155031
- [CrossRef] [Google Scholar]
- Health risk assessment and potentiality of green seaweeds on bioaccumulation of trace elements along the Palk Bay coast, Southeastern India. Mar. Pollut. Bull.. 2020;154:111069
- [CrossRef] [Google Scholar]
- Ramdani, Mohamed, Sahnouni, F., Haloui, B., Roger, N.E., Mesfioui, F.A., Ramdani, Mohammed, 2015. Macroepiphytes and macroalgae (Phaeophyceae, rhodophyceae and chlorophyceae) in the nador lagoon (Morocco).
- Ecology and seasonal distribution of two genera of diatoms, Cocconeis and Licmophora, recorded in the Nador Lagoon (North-East Morocco) AACL Bioflux. 2022;15:314-326.
- [Google Scholar]
- Variations in polyphenol and heavy metal contents of wild-harvested and cultivated seaweed bulk biomass: Health risk assessment and implication for food applications. Food Control. 2019;95:121-134.
- [CrossRef] [Google Scholar]
- Sanchiz, C., García-Carrascosa, A.M., Pastor, A., 1999. Bioaccumulation of Hg, Cd, Pb and Zn in Four Marine Phanerogams and the Alga Caulerpa prolifera (Försskal) Lamouroux from the East Coast of Spain 42, 157–164. https://doi.org/10.1515/BOT.1999.018.
- Sanchiz, C., M. García-Carrascosa, A., Pastor, A., 2000. Heavy Metal Contents in Soft-Bottom Marine Macrophytes and Sediments Along the Mediterranean Coast of Spain. Marine Ecology 21, 1–16. https://doi.org/10.1046/j.1439-0485.2000.00642.x.
- Seaweed by-products for food and bioremediation. Valoris. Seaweed By-Prod. 2019:197-219.
- [Google Scholar]
- Concentrations of various elements in seaweed and seawater from Shen’ao Bay, Nan’ao Island, Guangdong coast, China: Environmental monitoring and the bioremediation potential of the seaweed. Sci. Total Environ.. 2019;659:632-639.
- [CrossRef] [Google Scholar]
- Assessment of health risks due to arsenic from iron ore lumps in a beach setting. Sci. Total Environ.. 2016;563–564:405-412.
- [CrossRef] [Google Scholar]
- Tepe, A.Y., Dede, Ö.T., 2023. Chromium, in: Reference Module in Food Science. Elsevier. https://doi.org/10.1016/B978-0-12-822521-9.00111-8.
- Levels of toxic metals in edible fish species of the Tigris River (Turkey); Threat to public health. Ecol. Indic.. 2021;123:107361
- [CrossRef] [Google Scholar]
- US EPA, 2013. Integrated Risk Information System [WWW Document]. URL https://www.epa.gov/iris (accessed 9.20.22).
- Chemical composition and heavy metal content of Chilean seaweeds: Potential applications of seaweed meal as food and feed ingredients. Food Chem.. 2023;398:133866
- [CrossRef] [Google Scholar]
- Nutritional value of detritus and algae in blenny territories on the Great Barrier Reef. J. Exp. Mar. Biol. Ecol.. 2002;271:155-169.
- [CrossRef] [Google Scholar]
- Metal contents of the two marine algae found on iron ore tailings. Mar. Pollut. Bull.. 1979;10:56-59.
- [CrossRef] [Google Scholar]
- Mine waste water self-purification (arsenic) in neutral hydrogeochemical ecosystem: A case study from V-Ti-Fe mine tailings. Geochemistry. 2023;83:125947
- [CrossRef] [Google Scholar]
- Yalçın Tepe, A., 2014. Toxic Metals: Trace Metals – Chromium, Nickel, Copper, and Aluminum, in: Motarjemi, Y. (Ed.), Encyclopedia of Food Safety. Academic Press, Waltham, pp. 356–362. https://doi.org/10.1016/B978-0-12-378612-8.00205-5.
- Distribution and relationships between selected chemical elements in green alga Enteromorpha sp. from the southern Baltic. Environ. Pollut.. 2006;143:435-448.
- [CrossRef] [Google Scholar]
- Comparison of green algae Cladophora sp. and Enteromorpha sp. as potential biomonitors of chemical elements in the southern Baltic. Sci. Total Environ.. 2007;387:320-332.
- [CrossRef] [Google Scholar]







