Translate this page into:
Analytical methods for quantification of non-steroidal anti-inflammatory drugs in pharmaceutical and biological samples: An overview of developments in the last decade
⁎Corresponding author at: Department of Pharmaceutical Sciences, College of Pharmacy and Health Sciences, Ajman University, Ajman P.O. Box 346, United Arab Emirates. s.boddu@ajman.ac.ae (Sai HS. Boddu)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Nonsteroidal anti-inflammatory drugs (NSAIDs) are the most commonly used pharmaceuticals in both human and animal medicines for the treatment of certain conditions such as inflammation, fever, and pain. The role of validated analytical methods has become highly important in the quantification of drug substances from their pharmaceuticals as precise product quality control is required. In the present review, we have summarized various sample preparation methods and analytical methods developed for the quantification of NSAIDs during the past decade (2012-till date). Furthermore, an in-depth description of numerous techniques including chromatography (89), UV spectrophotometry (5), spectroflurometry (4), IR spectroscopy (3), electrophoresis (4), and electrochemical (9) approaches used to determine and quantify NSAIDs are provided. Based on the matrix utilized, the following details were discussed: analytical conditions, detection limits, and solvent used in sample preparation. The present compilation provides valuable insights and crucial information on quantification methods for NSAIDs and would assist the scientific community to select the best and economical method for drug analysis in pharmaceuticals and biological samples.
Keywords
NSAIDs
Sample preparation
Analytical methods
Spectroscopy
Chromatography
Electrochemical techniques
- µLPME
-
Microfluidic-based liquid-phase microextraction
- µSPE
-
Micro solid phase extraction
- AA
-
Arachidonic acid
- BR
-
Britton Robinson
- CE
-
Capillary electrophoresis
- CMS
-
Capillary microsample
- CMD
-
Cerebral micro dialysis
- COX
-
Cyclooxygenase
- CPE-MWCNT
-
Carbonate paste-multiwalled carbon nanotubes electrode
- CPE
-
Cloud point extraction
- CV
-
Cyclic voltammetry
- CZE
-
Capillary zone electrophoresis
- DAD/PDA
-
Photodiode array
- DBS
-
Dried blood spot
- DPS
-
Dried plasma spot
- DSS
-
Dried saliva spot
- DUS
-
Dried urine spot
- DES
-
Deep eutectic solvent
- DLLME
-
Dispersive liquid–liquid microextraction
- DMSPE
-
Dispersive magnetic solid phase extraction
- DPV
-
Differential pulse voltammetry
- EME
-
Electro membrane extraction
- FTIR
-
Fourier transform infrared
- FD
-
Fluorescence detection
- Fe3O4/SiO2/TiO2
-
Magnetite/silica/titania composite
- FID
-
Flame ionization detector
- FL
-
Spectro fluorimetry
- GC
-
Gas chromatography
- GC-QqQ-MS/MS
-
Gas chromatography-tandem mass spectrometry with the triple quadrupole
- GC-FID
-
Gas chromatography-flame ionization detection
- GC-EI-MS
-
Gas chromatograph–electron impact-mass spectrometer
- GPC
-
Gel permeation chromatography
- HCl
-
Hydrochloric acid
- HDTMA-ZSM5/Fe2O3
-
zeolite-based composite decorated with iron oxide magnetic nanoparticles and modified with hexadecyltrimethylammonium bromide surfactant
- HF-LPME
-
Hollow fiber liquid-phase microextraction
- HILIC
-
Hydrophilic interaction liquid chromatography
- HPLC
-
High-performance liquid chromatography
- HRMS
-
High-resolution mass spectroscopy
- HPLC-FD
-
high-performance liquid chromatography-florescence detector
- HPLC-MS
-
High-performance liquid chromatography-mass spectrometry
- HPLC-PDA
-
High-performance liquid chromatography-photo diode array
- HPLC-MS/MS
-
High-performance liquid chromatography-tandem mass spectrometry
- HRMS
-
High-resolution mass spectrometry
- IR
-
Infrared
- LC
-
Liquid chromatography
- LLE
-
Liquid-liquid extraction
- LLME
-
Liquid-liquid microextraction
- LPME
-
liquid-phase microextraction
- LOD
-
Limit of detection
- LOQ
-
Limit of quantitation
- LC-HRMS
-
Liquid chromatography-high resolution mass spectrometry
- MCM-41
-
Mesoporous silica material
- MDSPE
-
Magnetic dispersive solid-phase extraction
- MEKC
-
Micellar electrokinetic capillary chromatography
- MEPS
-
Microextraction by packed sorbent
- MIP
-
Molecularly imprinted polymer
- MP A
-
Moblie phase A
- MP B
-
Moblie phase B
- MS/MS
-
tandem mass spectrometry
- MS
-
Mass spectrometry
- MSPD
-
Matrix solid-phase dispersion method
- MSPE
-
Magnetic solid phase extraction
- NaOH
-
Sodium hydroxide
- RDSE
-
Rotating disk sorptive extraction
- RP
-
Reversed phase
- SEME-SFOD
-
Surfactant-enhanced emulsification microextraction method based on solidification of floating organic drop
- SFC
-
Supercritical fluid chromatography
- SPE
-
Solid-phase extraction
- SPME
-
Solid phase microextraction
- SPMTE
-
Solid phase membrane tip extraction
- TAALLM
-
Tandem air agitated liquid–liquid microextraction
- TFME
-
Thin film microextraction
- TX
-
Thromboxane
- UA-Dµ-SPE-S-UA-LLME-SFO
-
Ultrasound assisted dispersive micro solid-phase extraction coupled with salting-out ultrasound-assisted liquid–liquid microextraction based on solidification of a floating organic droplet
- UHPLC
-
Ultra-high performance liquid chromatography
- UPLC
-
Ultra-performance liquid chromatography
- UV
-
Ultraviolet
Abbreviations
1 Introduction
Non-steroidal anti-inflammatory drugs (NSAIDs) constitute some of the highly recognized categories of pharmaceutical agents having a variety of advantages. These agents are generally employed as therapeutics in humans and animals due to their ability to function as antipyretic, analgesic, and anti-inflammatory agents (I. Olives et al., 2012; Izadi et al., 2020). Drugs included within this category differ in terms of their chemical properties and almost all agents possess varying amounts of therapeutic efficacy (Kress et al., 2016; Modi et al., 2012; Smith, 2014). NSAIDs are widely employed to alleviate several debilitating conditions including fever, migraines, menstrual irregularities, rheumatoid arthritis, osteoarthritis, gout, and postoperative complications (Katturajan and Sabina, 2021; McCarberg and Gibofsky, 2012). Several reports in the literature have suggested that NSAIDs have certain functions in reducing the risk of acquiring malignancies such as colorectal, breast, ovarian, hepatocellular, prostate, pancreatic, and head and neck cancers (Amici et al., 2006; Bindu et al., 2020; Ruder et al., 2011). These agents act on cyclooxygenase enzymes (COX-1 and COX-2) that are involved in the conversion of arachidonic acid (AA) into prostaglandins (PGs) and also generate thromboxane’s (TX), which is ultimately involved in the modulation of the inflammatory response (Omran, 2013; Shishov et al., 2019; Smith et al., 1998). NSAIDs can be further subdivided into COX-1 selective, COX-1 non-selective, and COX-2 selective based on their mechanism of action (Haag et al., 2008). The physicochemical properties of selected NSAIDs are outlined in Table 1.
Class
Drugs
Mol. weight (g/mol)
Log P
pKa
Water solubility (mg/mL)
Nonselective COX inhibitors
Salicylic acid derivatives
Aspirin
180.16
1.18
3.50
10.0
Pyrazole derivatives
Phenylbutazone
308.37
3.16
4.50
47.5
Anthranilic acid derivatives
Mefenamic acid
241.28
5.12
4.20
20.0
Flufenamic acid
281.23
5.25
3.88
9.1
Propionic acid derivatives
Ibuprofen
206.29
3.97
5.30
21.0
Naproxen
230.26
3.18
4.15
15.9
Ketoprofen
254.28
3.12
4.45
51.0
Acetic acid derivatives
Diclofenac
296.15
4.51
4.15
2.37
Ketorolac
255.27
2.10
3.84
0.86
Indole derivatives
Indomethacin
357.79
4.27
4.50
0.93
Enolic acid derivatives
Piroxicam
331.35
3.06
6.30
23.0
Meloxicam
351.40
1.16
4.47
22.0
Tenoxicam
337.38
1.19
2.21
14.1
Selective COX-2 inhibitors
Coxibs
Celecoxib
381.37
3.53
11.10
0.000503
Etoricoxib
358.84
2.79
16.19
0.00328
Sample preparation is considered as one of the crucial steps in an analytical process, as it plays a significant role in mitigating the impact of interfering factors on the analytical performance of various techniques. Moreover, appropriate sample preparation can be useful in several analytical methods (Li et al., 2020). Techniques such as homogenization, solid-phase extraction (SPE), liquid–liquid extraction (LLE), and centrifugation are commonly employed in sample preparations to improve the sensitivity, specificity, and accuracy of the analytical results (Yilmaz et al., 2020). This review provides an overview of different analytical techniques used in the detection and quantification of NSAIDs in pharmaceutical preparations and biological samples. Moreover, it gives preliminary data on developed analytical methods (chromatography, spectrophotometry, spectroscopy, electrophoresis, and electrochemical techniques) that have been used in the determination of NSAIDs containing pharmaceuticals and biological samples.
2 Pharmacokinetics and metabolism
Drugs included in the NSAID category possess both hydrophilic groups, such as carboxylic or enolic groups, and lipophilic groups, such as aromatic rings or halogen atoms (Starek and Krzek, 2009). The gastrointestinal tract serves as the primary route of absorption for most of these drugs. These medications typically have high bioavailability making them highly effective agents. However, certain NSAIDs, such as diclofenac, are subjected to hepatic first-pass metabolism, which reduces their bioavailability. Conversely, prodrugs, such as sulindac and parecoxib, require hepatic metabolism to convert to their active metabolites, sulindac sulphide, and valdecoxib, through which they can exert their action. A majority of NSAIDs that exist in the plasma remain in highly ionized forms. The strong binding capacity (>97 %) of NSAIDs to plasma proteins is a result of their favorable amphiphilic properties, which displace other drugs from protein binding leading to increased persistence in the body and a high duration of action (Starek and Krzek, 2009). The volume of distribution is generally low for NSAIDs ranging from 0.1 to 0.3 L/kg of body weight since they are lipophilic in nature, and are not metabolized or excreted by the liver or kidneys. The half-life of NSAIDs varies to a great extent depending on their physicochemical properties. For example, the half-life of aspirin is 0.25–0.3 h, while for piroxicam it is 45–50 h (Awtry and Loscalzo, 2000). These pharmacokinetic parameters are influenced by the patient’s age, protein binding, and the distribution of the drug (Wongrakpanich et al., 2018).
3 Sample preparation
A brief summary of pre-treatment methods that are utilized for the analysis of NSAIDs in biological specimens is provided in this section. Recent developments in analytical instrumentation have resulted in improvements in sensitivity, efficiency, and selectivity. Nevertheless, there are a few challenges in directly analyzing low or trace amounts of analytes in complicated samples. These difficulties include the existence of matrix effects, interferences, and analytical instrument incompatibilities. In order to overcome these difficulties, scientists frequently isolate and purify the target analyte before analysis using sample preparation procedures like extraction, separation, or purification (Ocaña-González et al., 2016; Yan and Wang, 2013).
Sample preparation is an essential step in the analytical process, where different issues that may come up when analyzing complex samples are carefully considered. Once the barriers have been identified, particular actions can be considered, like eliminating substances that interfere, boosting the analyte concentration, and converting the analytes into a form that is appropriate for separation and detection. These actions aid in guaranteeing the accuracy and dependability of the analytical results (Ansari and Karimi, 2017). Pre-treatment procedures are essential for the analysis of NSAIDs in biological samples differ significantly from those needed for the analysis of NSAIDs in pharmaceutical products because the matrix of biological samples containing NSAIDs can vary widely in terms of concentration levels, interferences present, and whether the sample is in a liquid or solid state. The objective of using analytical techniques for pharmaceutical products and bulk drugs is to evaluate the quality of these substances and identify any impurities or degradation products that might be present. The efficacy, safety, and regulatory compliance of drugs are guaranteed by this procedure. Utilizing analytical techniques for medications such as NSAIDs also has the benefit of allowing for the long-term monitoring of the drug's stability and the detection of any alterations in the drug's quality that might happen during transportation or storage. (I. Olives et al., 2012).
In biological matrices such as blood, serum, plasma, saliva, urine, sweat or tissues, the estimation of NSAID is essential for pharmacokinetic and pharmacodynamic studies (Al-Khateeb and Dahas, 2021). During analysis of therapeutic drugs in different biological sample matrices, selecting a suitable sample preparation method is considered to be the most important step in the analytical process. This is a result of the intricacy of such matrices, which include a range of endogenous substances that can impede the analysis, including proteins, salts, and particulate matter. Furthermore, the presence of drugs in biological matrices at very low concentrations often complicates the analysis even more. As a result, selecting a reliable and safe sampling technique is crucial (Saito and Nakagami, 2020; Vasconcelos and Fernandes, 2017). One example of a bioanalytical analysis technique is the protein precipitation method, which typically involves centrifuging the sample first and adding an organic solvent, such as methanol or acetonitrile (Lakshmana and K. Suriyaprakash, 2012). For sample preparation in complicated matrices, the traditional procedures of LLE, protein precipitation with subsequent centrifugation, and SPE have been utilized most frequently (Carasek and Merib, 2015; Płotka-Wasylka et al., 2016). Recently, there has been a shift towards miniaturization and automation in sample preparation techniques in order to address the issues of time-consuming, labour-intensive, and costly procedures, as well as the negative environmental impact of the use and disposal of large amounts of organic solvents (Magiera et al., 2013). Furthermore, different microextraction techniques have been developed for sample preparation in bioanalytical applications. These techniques include solid-phase microextraction (SPME), micro extraction by packed sorbent (MEPS), stir bar sorptive extraction (SBSE), thin-film microextraction (TFME), liquid-phase microextraction (LPME), dispersive liquid–liquid microextraction (DLLME), single-drop microextraction (SDME), electromembrane extraction (EME), solvent bar microextraction (SBME), etc. These techniques have become more acceptable due to their advantages in terms of their simple and rapid operation, accuracy, and sensitivity, during clinical investigations. Therefore, continuous improvement of novel sample preparation and microfluidics-based techniques is necessary to accelerate bioanalytical research (Alexovič et al., 2018; Boyaci et al., 2015; Buszewski and Szultka, 2012; Daryanavard et al., 2021; Kabir et al., 2017; Vas and Vékey, 2004).
Microsampling is an another approach of sample preparation which require small sample volumes (<50 μL) of biological fluids. It has gained significant attention at ICH level and reflected in ICH M10, SA3, and S11 guidelines. This technique assists remote sampling, easy shipment, storage and analysis during clinical trials. Example of microsampling techniques linclude cloud point extraction (CPE), microdialysis, cerebral microdialysis (CMD), dried blood spot (DBS), dried plasma spot (DPS), dried saliva spot (DSS), dried urine spot (DUS), volumetric absorptive microsample (VAMS), capillary microsample (CMS), spin column extraction (SCE) (Ingle et al., 2022; Londhe and Rajadhyaksha, 2020).
4 Analytical techniques
Although reviews for the quantification of NSAIDs have been published in the past, none of these reviews include all the reported analytical techniques for the analysis of NSAIDs in pharmaceuticals and biological samples. Additionally, a lot of fresh analytical methods have been reported in the recent past. With the development of novel sample preparation procedures as well as sophisticated chromatographic and spectrometric techniques during the past ten years, drug analysis has experienced enormous expansion. Therefore, an updated and comprehensive review that can briefly explain various analytical methodologies for NSAIDs is required. Furthermore, a concise tabular explanation of each analytical technique along with its procedure and specifics, such as analytical conditions, matrices, and limit of detection (LOD) is mentioned. A graphical representation of different analytical techniques used for quantification NSAIDs in pharmaceuticals and biological samples is shown in Fig. 1.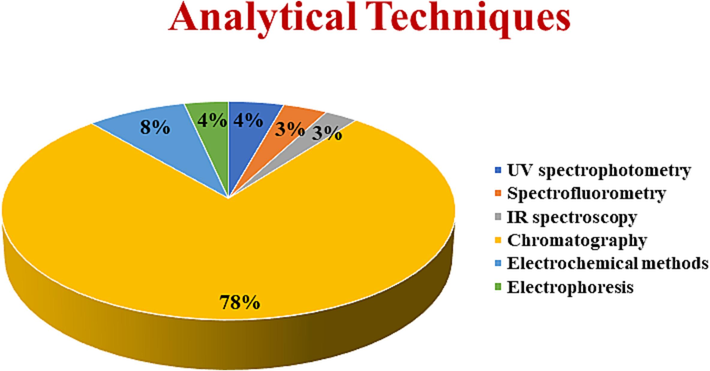
Graphical representation of different analytical techniques used for NSAIDs determination in pharmaceuticals and biological samples.
4.1 Spectrometric and spectroscopic techniques
Spectrometric and spectroscopic methods have several advantages over other analytical methods such as being an easy, uncomplicated procedure as well as low reagent consumption. Thus, they are considered cost-effective and time-saving methods as compared to most of the other methods. Furthermore, these methods can be used for the determination of drugs in laboratories where modern and expensive equipment such as gas chromatography (GC) or high-performance liquid chromatography (HPLC) are not available.
4.1.1 UV spectrophotometric technique
Several studies have reported UV spectrophotometric techniques for the determination of NSAIDs in pharmaceuticals and biological samples. A summary of these studies is presented in Table 2 and Table 3. Liposome-encapsulated diclofenac sodium was prepared by Goh et al. and analyzed using a simple, economical, and reliable UV spectrophotometric technique employing dimethyl sulfoxide as a solvent. The detection wavelength was 295 nm with a linearity range of 5–35 µg/mL and a 0.9978 regression coefficient. The LOD and limit of quantitation (LOQ) were 1.19 and 3.62 µg/mL, respectively (Goh et al., 2014). Zaazaa et al. carried out an analysis of ibuprofen with famotidine in a combined form wherein they analyzed ibuprofen in the form of tablet matrices through ratio difference UV spectrophotometric analysis with methanol and 0.1 N NaOH as solvents. The wavelength applied for detection was 262.5–271.7 nm. The LOD and LOQ values were found to be 13.68 and 41.47 µg/mL, respectively. This ratio difference method was simpler than the Q-absorption and absorption correction methods as it involves fewer data processing steps (Lotfy et al., 2015; Zaazaa et al., 2015). For the simultaneous estimation of naproxen, imatinib, and nalbuphine in a quaternary mixture and human urine, Belal et al. used the spectrophotometric technique. This work used a univariate method (extended derivative ratio) as well as the multivariate method (partial least squares in original and derivative mode). They asserted that these methods work in laboratories without liquid chromatographic equipment and can be used for quality control analysis without the need for any preliminary separation steps (Belal et al., 2018). Mabrouk et al. analyzed ketorolac in biological samples of human plasma with 0.1 N HCl and phosphate buffer as solvents to simulate human conditions. The wavelength used was 223 nm. The LOD value was found to be 0.08 µg/mL with a linearity range of 2–20 µg/mL. This method differentiated and measured ketorolac after the removal of potential interfering substances in the blank biological matrix without using large volumes of organic solvents, such as methanol, acetonitrile, and n-hexane, sophisticated instruments (Mabrouk et al., 2020). The described spectrophotometric methods showed several advantages such as simplicity, low cost, and speed of analysis. However, disadvantages such as the inability to analyze related substances and less sensitivity often make them unattractive in analysis.
Analyte
Formulation
Solvent(s)
Detection (nm)
Linearity (µg/mL)
LOD (μg/mL)
Ref.
Diclofenac sodium
Liposome encapsulated formulation
Dimethyl sulfoxide
295.0
5–35
1.19
(Goh et al., 2014)
Ibuprofen
Tablet
Methanol and 0.01 N Sodium hydroxide
262.5–271.7
50–600
13.68
(Zaazaa et al., 2015)
Naproxen
Tablet
Methanol
239.0
1–6
0.09
(Belal et al., 2018)
Mefenamic acid
Tablet
0.1 N Sodium hydroxide
285
2–10
–
(Pabla et al., 2018)
Analyte
Matrix
Sample Preparation
Sorbents/Solvents used in Sample Preparation
Detection (nm)
Linearity (μg/mL)
LOD (μg/mL)
Ref.
Naproxen
Human urine
Weighing, dilution, filtration
Methanol
239.0
1.0–6.0
0.09
(Belal et al., 2018)
Ketorolac
Human plasma
SPE
Ion exchange MIP (Chitosan-based)
223.0
2–20
0.08
(Mabrouk et al., 2020)
4.1.2 Spectrofluorimetric technique
Table S1 and Table S2 summarize the studies utilizing spectrofluorimetric techniques for the estimation of NSAIDs in pharmaceuticals and biological samples. Lian et al. used a complex imprinted membrane as the recognition material to develop a simple and effective solid-surface fluorescence method for determining naproxen in a capsule dosage form. The extraction, concentration, and detection steps were combined into one step by a complex imprinted membrane, which might have improved the effectiveness of the analytical process. A satisfactory linearity was attained between 0.50 and 20 mg/L concentrations, and LOD was 0.11 mg/L at emission and excitation wavelengths of 352 and 284 nm, respectively (Lian et al., 2013).
Amjadi et al. performed the biological analysis of celecoxib in human serum using ratiometric fluorescent nanosensor at emissive and excitatory wavelengths of 550–440 and 360 nm, respectively. They have developed a new sensor for selective identification and quantitative determination of target analyte, by integrating the exceptional selectivity of the molecularly imprinted polymer (MIP), the effective characteristics of the ratiometric approach, and the sensitivity of mesoporous silica. The LOD and linearity range was found to be 57 µM and 0.08–0.90 µM, respectively (Amjadi and Jalili, 2018). Attala et al. used an improved first derivative synchronous spectrofluorimetric technique to identify amlodipine and celecoxib in a combined dosage form and human plasma. Compared to traditional native fluorescence techniques, synchronous spectrofluorimetric simplified emission spectra, narrows spectral bands, and constricts spectral range. To increase the fluorescence intensity, a complex was formed between the drugs and sodium dodecyl sulphate. Linearity, LOD, LOQ, precision, and accuracy of the validation parameters were within acceptable limits (Attala et al., 2020). Only being able to analyze fluorescent compounds or requiring a derivatization step that increases complexity and analysis time, and typically prevents the analysis of related substances, are its limitations, which restrict the use of this technique in quality control applications.
4.1.3 IR spectroscopic techniques
Haskell et al. analyzed ibuprofen in tablet formulation and urine sample using transmission Fourier transform infrared (FTIR) method in which LOD values were found to be 0.77 µg/mL in both samples and the linearity range was found to be 10–100 µg/mL. The main outcomes of this method were analytical simplicity, improved rapidity, improved accuracy, and improved sensitivity for ibuprofen quantification. The range from 1807 to 1461 cm−1 was used to create a partial least square calibration model as shown in Table S3 and Table S4 (Khaskheli et al., 2013). For the determination of four NSAIDs, etodolac, tolfenamic acid, bumadizone, and diacerein, either alone or in the presence of their degradation products, Hassib et al. used ATR-FTIR methods like direct measurement, first derivative, and second derivative with minimal sample preparation. To get over the spectrum overlap and enable simultaneous assessment of drugs and their degradation product, derivative spectroscopy was utilized for data processing. From the pharmaceutical preparation, active constituents were extracted in chloroform, and then directly measured in liquid form. The LOD of etodolac, bumadizone, and tolfenamic acid were observed to be 1.523, 2.773, and 1.193 μg/mL, respectively. The reported techniques could serve as a substitute for methods that utilize the separation processes. IR spectroscopic technique has the potential to decrease both chemical waste and costly laboratory testing (Hassib et al., 2017). Nevertheless, this technique does not allow analyte separation.
4.2 Chromatographic techniques
Several chromatographic techniques were used in the analysis of NSAID drugs such as high-performance liquid chromatography-UV (HPLC-UV), high-performance liquid chromatography-floroscence detector (HPLC-FD), high-performance liquid chromatography-mass spectrometry (HPLC-MS), high-performance liquid chromatography-photo diode array (HPLC-PDA), high-performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS), ultra-high performance liquid chromatography-tandem mass spectrophotometry (UHPLC-MS/MS), supercritical fluid chromatography (SFC), gas chromatography- flame ionization detector (GC-FID), gas chromatography-mass spectrometry (GC–MS), gas chromatography-tandem mass spectrometry (GC–MS/MS), and liquid chromatography-high resolution mass spectrometry (LC-HRMS), and hydrophilic liquid chromatography (HILIC). These methods are listed in Table S5 and Table S6. The quantitation of NSAIDs in pharmaceutical formulations (tablet, capsule, caplet, eye drops, and suspension) and biological samples (human plasma, urine, blood, breast milk, rat plasma, rabbit tissue, swine muscle, etc.) using chromatographic techniques with various types of detectors, including UV, fluorescence, PDA, MS, and MS/MS are represented in Fig. 2.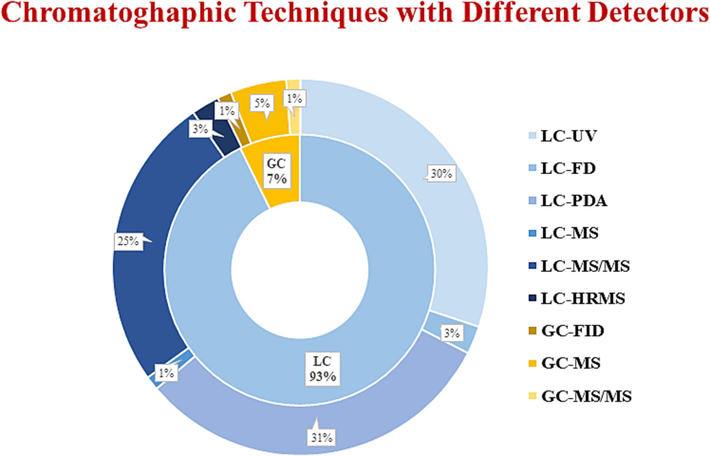
Summary of various chromatographic techniques along with different detectors used for NSAIDs determination in pharmaceuticals and biological samples.
The determination of mefenamic acid in urine and pharmaceutical samples was carried out by Rezaei Kahkha et al. using HPLC-UV following pipette-tip solid phase microextraction using carbon nanotubes modified with zinc sulphide. The method was observed to have good linearity in the range of 0.7–100 µg/L. The LOD was found to be 0.075 µg/L. Additionally, a quick analysis time of 9 min was attained (Rezaei Kahkha et al., 2016). For the biological sample analysis of naproxen, diclofenac, and mefenamic acid, the HPLC-UV analytical technique was employed by Aqda et al. which was analyzed in the plasma sample. The mobile phase of methanol and water in an 80:20 proportion was pumped at a flow rate of 1.0 mL/min with the C18 column as a stationary phase. LOD values were found to be 1.80 µg/mL, 2.40 µg/mL, and 2.00 µg/mL for naproxen, diclofenac, and mefenamic acid, respectively (Golzari Aqda et al., 2018). In 2023 Han et al. coupled the Fe3O4@magnetic ionic liquid hypercrosslinked polymer composite based magnetic solid-phase extraction (MSPE) with HPLC-DAD to detect specific NSAIDs in urine and water samples with sensitivity. Using Friedel-Crafts alkylation, a magnetic ionic liquid hyper crosslinked polymer composite was produced as an MSPE adsorbent. The developed composite was able to extract NSAIDs from real samples with a wide linear range, low LODs, and satisfactory recoveries (Han et al., 2023). Using a simple cysteine-triggered in situ growth strategy, Ji et al. created amino-bearing metal–organic frameworks modified on cotton fibers and used as an in-situ SPME adsorbent combined with HPLC-UV for the extraction and quantitation of three NSAIDs i.e. diclofenac sodium, ketoprofen, and flurbiprofen in human plasma samples. The experiment yielded good linear range, good reproducibility, good recoveries (66.5 % − 98.9 % with less than 6.62 % relative standard deviations), and satisfactory sensitivity. For the quantitative analysis and pretreatment of NSAIDs in complex samples, the suggested method showed potential (Ji et al., 2023).
For the detection and quantification of NSAIDs such as acetylsalicylic acid, ketoprofen, diclofenac, naproxen, and ibuprofen in human urine, Magiera et al. reported a novel method based on MEPS and an RP-UHPLC-UV. The analytes were separated using a binary mobile phase (aqueous 0.1 % trifluoroacetic acid:Acetonitrile) in the gradient elution mode on a core–shell C18 column (100 × 3.0 mm × 2.7 µm). For the target drugs, good linearity (1.07–16.2 ng/mL) and LOD were reported (Magiera et al., 2013). For the simultaneous separation and determination of NSAIDs in human urine and plasma, Ferrone et al. developed graphene/Fe3O4-based dispersive magnetic solid phase extraction coupled with UHPLC-PDA. It was found that using an isocratic elution mode with acetonitrile and 10 mM potassium dihydrogen phosphate (pH 2) in water (50:50, v/v) pumped at a flow rate of 0.55 mL/min resulted in higher separation efficiency, better peak shape, and short run time (Ferrone et al., 2018). The chiral HPLC is used to distinguish between the enantiomers of naproxen, etodolac, and ibuprofen. Hewala et al. performed the analysis of the etodolac drug which existed in their R- and S- enantiomers in the form of tablet pharmaceutical preparation and human plasma through HPLC-DAD. The mobile phase was hexane, isopropanol, and trifluoracetic acid in a 90:10:0.1 proportion. The stationary phase was the Kromasil Cellucoat Chiral column (Hewala et al., 2014). In order to quickly determine S-ibuprofen and R-ibuprofen at low concentration levels typically present in human breast milk, León-González et al. combined the optimized vortex-assisted matrix solid-phase dispersion method (MSPD) procedure with direct chiral LC-UV detection. Upon analysis, LOD was found to be 0.042 µg/g for R-ibuprofen and 0.045 µg/g for S-ibuprofen (León-González and Rosales-Conrado, 2017). LC-MS and LC-MS/MS are widely used to determine NSAID residues in complex biological matrices such as human plasma, urine, beef liver, whole blood, and animal tissues. LC coupled to MS or MS/MS is preferred over other detectors due to its high analytical selectivity and sensitivity. The accurate identification of poisons or drugs is the primary objective of toxicology investigations. Since substances with chemically similar structures may potentially cause interference, it is important to accurately discriminate drugs from all potential sources of interference. This can be done by using LC-MS/MS techniques. In order to simultaneously quantify sixty drugs that are frequently found in postmortem blood, Al-Asmari has developed an LC-MS/MS technique. The method could be used for NSAIDs like acetaminophen, ibuprofen, mefenamic acid, and diclofenac as well as a wide range of other compounds and their metabolites in forensic toxicology cases. Gradient elution was carried out using biphenyl columns (50 × 3.0 mm, 2.7 µm) at a flow rate of 0.30 mL/min. This method was found to quickly and accurately identify multiple drugs. The author has claimed that this technique is for the multianalyte screening of sixty drugs and their metabolites that are commonly encountered in postmortem toxicology and can be applied to the routine analysis of autopsy blood samples (Al-Asmari, 2020). Sun et al. developed a UPLC-MS/MS method for the detection of NSAIDs in swine kidneys, fat, muscle, and liver. Phosphorylated acetonitrile was used to extract the swine tissue samples, which were then purified using a hydrophile-lipophile balance (HLB)-SPE column and separated using an AcquityTM UPLC BEH shield RP18 column in gradient elution mode, and 0.1 % formic acid in acetonitrile and water as a mobile phase. The method was able to separate and detect ten different types of NSAIDs in 10 min with good recovery and repeatability. The suggested technique would be helpful for regulatory monitoring of NSAID residues in swine fat, liver, kidneys, and muscle (Sun et al., 2023). There are some drawbacks to LC techniques, including sample pretreatment or extractions, derivatization, lengthy analysis times, significant waste disposal, and high instrument and maintenance costs.
The fact that GC requires a derivatization step before analysis, which adds to the experimental effort, makes it a less popular method than LC, but it was still used to determine NSAIDs in a number of different matrices. The extraction of salicylic acid (hydrolysis product of aspirin), diclofenac, and ibuprofen, in human urine is done before their detection by GC-FID, Barfi et al. compared two dispersive-based LLME methods which include USE-AALLME and LDS-DLLME. The findings demonstrated that USE-AALLME combined with GC-FID was significantly more efficient method, offering high analyte recoveries without the need for a derivatization step and detection limits of 0.1–1.0 µg/L (Barfi et al., 2015). Ibuprofen and its four likely metabolites were simultaneously determined in equine urine samples using a novel assay developed by Waraksa et al. using GC-EI-MS. In the sample preparation step, methyl-derivatization was carried out by adding 100 mL of ethyl acetate, 50 mL of methyl iodide, and 50 mg of anhydrous potassium carbonate. An Agilent HP-1 MS column (17 m × 200 µm, 0.11 µm) was used for the separation. As a carrier gas, helium was used with a constant flow rate of 1.8 mL/min. The LOD for ibuprofen, 1-hydroxy ibuprofen, 2-hydroxy ibuprofen, 3-hydroxy ibuprofen, and carboxy ibuprofen was found to be 0.58, 0.20, 1.37, 0.19, and 1.33 µg/mL, respectively, and also produced a satisfactory linear concentration range (Waraksa et al., 2018). Szpot et al. developed an ultra-sensitive GC-QqQ-MS/MS with an electron impact ionization source for direct detection of diclofenac in whole blood samples. The multiple-reaction monitoring mode was used to determine the analytes. Helium served as the carrier gas and the SH-RXI-5MS column (30.0 m × 0.25 mm, 0.25 µm) served as the stationary phase. The column temperature was initially maintained at 60 °C for 2 min before being raised to 320 °C and maintained for another 2 min. The method had a regression coefficient of 0.999 and was linear from 0.1 to 200 ng/mL. Moreover, the LOD and LOQ attained were 0.05 ng/mL and 0.1 ng/mL, respectively, both of which were relatively low (Szpot et al., 2021). GC has been widely used for NSAIDs analysis, however, further research is needed to address a few drawbacks such as the need for a derivatization step for compounds that exhibit low volatility and have poor thermal stability.
4.3 Electrochemical techniques
Recently, a number of electrochemical techniques have drawn a lot of interest because they have been found successful in quantifying various pharmaceutical compounds. In most scenarios, sample pre-treatment is unnecessary, non-destructive, and takes less time. Its main advantage over conventional methods is that they enable the low-cost simultaneous analysis of multiple analytes at once. For the analysis of NSAIDs, both potentiometric and voltammetric techniques were reported; however, voltammetry is the technique that is most frequently employed due to its high sensitivity.
4.3.1 Potentiometry techniques
Potentiometric methods are an intriguing alternative to traditional drug testing because they offer straightforward procedures, inexpensive electrodes, and good sensitivity. Potentiometric techniques used in the analysis of NSAIDs are listed in Table S7. A new potentiometric sensor based on doped polypyrrole films for the detection of diclofenac was prepared and characterized in work reports by Oliveira et al. Diclofenac anion is incorporated into polypyrrole film during polymer electrochemical synthesis to create a membrane with a selective potentiometric response for the dopant ion. The pharmaceutical analysis of diclofenac existed in the form of tablet preparation and the solvent used was 10 mL of hydroalcoholic solution (20 %). With an electric charge of 35 mC and an electric current density of 0.045 mA/cm2, it was observed that the sensor displayed a linear dynamic response in the concentration range from 3.1 × 10-4 to 1.1 × 10-2 mol/L (Oliveira et al., 2014).
4.3.2 Voltammetric techniques
Electroanalytical approaches, particularly voltammetric techniques are also used in the analysis of pharmaceuticals in recent years due to their precise ability to quantify analytes, less expensive equipment, and sensitivity. Various voltammetric methods have been highlighted in Table S8 and Table S9. The differential pulse voltammetry (DPV) method is primarily employed to determine NSAIDs in pharmaceuticals and biological samples. Aguilar-Lira et al. carried out a DPV analysis of diclofenac analyte which was present in the form of tablet pharmaceutical preparation. The solvent used was Britton-Robinson buffer (BR buffer) having pH 8. The electrodes used for the analysis were carbonate paste-multiwalled carbon nanotubes electrodes (CPE-MWCNT). The analyte concentration was found to be 2.49–10 µmol/L and the LOD value was 0.74 µmol/L (Aguilar-Lira et al., 2017). Hendawy et al. performed an analysis of naproxen in human plasma biological samples in a DPV technique. The electrode employed was a nanomaterial-based carbon paste electrode in which the concentration range of the analyte was found to be 4.35–65.5 µM and the LOD value was 6.255 µM and a quick run time of 66 s (Nigović et al., 2018). Although electrochemical techniques possess many benefits such as selectivity and accuracy, drawbacks such as extended analysis time and the necessity of complicated sample preparation steps reduce the enthusiasm for their use.
4.4 Electrophoresis techniques
Electrophoresis methods (Table S10 and Table S11) like capillary zone electrophoresis (CZE) and micellar electrokinetic capillary chromatography (MEKC) have many benefits, including high separation efficiency, short run times, easy-to-use instruments, low operating costs, and compatibility with relatively small volumes. It has been demonstrated to be among the most effective methods for analyzing biological samples. Zhang et al. performed an analytical estimation of naproxen in biological samples of human urine in which a borate buffer of pH 10.0 was used as a solvent. Capillary electrophoresis with chemiluminescence was used for analysis. The voltage was adjusted to 16 kV due to its high separation efficiency and selectivity of chemiluminescence system towards analytes. This method could determine naproxen in relatively complicated matrices. The linear concentration range was found to be 10.0–2000 µg/L and LOD and LOQ values were 2.7 µg/L and 2.8 µg/L, respectively (Zhang et al., 2018). Dal et al. carried out an analysis of piroxicam in tablet formulation in which borate buffer having pH 9.0 along with 10 % v/v methanol was used as a solvent. The analysis was conducted using capillary zone electrophoresis. The LOD and LOQ values are 0.07 µg/mL and 0.19 µg/mL, respectively (Dal et al., 2014). To analyze five pharmaceutical formulations containing three NSAIDs, El-Kommos et al. created a quick and precise MEKC method. All mixtures were separated using a 20 mM borate buffer of pH 9, 100 mM sodium dodecyl sulphate, and methanol (15 % v/v), with a voltage of 15 kV. The detection was carried out at 214 nm. The technique could be used in quality control labs not only for the binary mixtures under investigation but also for potential future binary mixtures with the same components (El-Kommos et al., 2013).
5 Conclusion
Due to the fact that NSAIDs are frequently prescribed for a wide range of indications, their analysis becomes an important aspect to ensure proper quality control. The present review summarizes various analytical techniques and sample preparation methods of NSAIDs both in pharmaceutical preparations and biological samples reported in the previous decade (2012-present). Appropriate analysis of NSAIDs not only ensures proper quality control but can also be employed in their pharmacokinetic and pharmacodynamic studies in order to examine drug toxicity, stability, and interaction studies, which provide valuable insights into the drug profile of NSAIDs. The commonly used pre-treatment techniques include LLE, SPE, and the environmentally friendly novel techniques including LLME (DLLME, HF-LPME). Upon evaluation, we observed that the most widely utilized separation and analytical techniques are LC using UV, FD, MS, and tandem MS/MS techniques for their estimation. LC methods have several advantages over other methods in terms of simple to perform and cost-effective, which is why they are widely used in the clinical monitoring. However, other chromatographic methods such as GC are not commonly employed in the estimation of NSAIDs. Another example of a method that has come to light is spectrophotometric analysis in UV along with fluorimetry. A drawback of this technique is that in spite of widely available pieces of equipment, their use is limited, especially with a complex matrix. In recent times, electrochemical (voltammetric) methods have gained attention for the estimation of pharmaceutical products as they possess the advantages of being cheap, widely available, rapid to perform, simple to perform, and provide results that are more sensitive than HPLC and spectrometric methods of analysis. To conclude, it would be helpful for researchers to gain insights into the various methods employed for the estimation of NSAIDs and select the method most convenient for their research.
Author contributions
Bhavarth Dave, Jyoti Solanki, Kunal Maheshwari: Gathered information from the mentioned references and finalized the layout of the article along with the addition of content and prepared Figures, diagrams, and tables. All four authors also contributed to the subsequent manuscript draft review, editing, and subsequent changes during the manuscript preparation.
Palak Parikh, Ketan Ranch, Anuradha Gajjar, Bharaneeswar Renukuntla: Design of content and skeleton, Manuscript draft review and editing, Figures and diagram conception, overall monitoring and guidance throughout the study duration.
Nisha Parikh, Sai HS Boddu: Topic conception, preparation of the article, and formatting it according to the specified requirements.
Funding
The authors declare that no funds have been received for the preparation of the manuscript. The authors would like to acknowledge Ajman University for their support towards Article Processing Charges.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Artificial neural network for the voltamperometric quantification of diclofenac in presence of other nonsteroidal anti-inflammatory drugs and some commercial excipients. J. Electroanal. Chem.. 2017;801:527-535.
- [CrossRef] [Google Scholar]
- Method for the identification and quantification of sixty drugs and their metabolites in postmortem whole blood using liquid chromatography tandem mass spectrometry. Forensic Sci. Int.. 2020;309:110193
- [CrossRef] [Google Scholar]
- Achievements in robotic automation of solvent extraction and related approaches for bioanalysis of pharmaceuticals. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2018;1092:402-421.
- [CrossRef] [Google Scholar]
- Extra-thermodynamic study of the retention of anti-inflammatory 2-arylpropionic acid derivatives on a heat-resistive stationary phase: Application of HTLC approach for pharmaceutical and biological analysis. Microchem. J.. 2021;169:106597
- [CrossRef] [Google Scholar]
- Green method development approach of superheated water liquid chromatography for separation and trace determination of non-steroidal anti-inflammatory compounds in pharmaceutical and water samples and their extraction. Arab. J. Chem.. 2021;14:103226
- [CrossRef] [Google Scholar]
- Indomethacin has a potent antiviral activity against SARS coronavirus. Antivir. Ther.. 2006;11:1021-1030.
- [CrossRef] [Google Scholar]
- A molecularly imprinted dual-emission carbon dot-quantum dot mesoporous hybrid for ratiometric determination of anti-inflammatory drug celecoxib. Spectrochim. Acta A Mol. Biomol. Spectrosc.. 2018;191:345-351.
- [CrossRef] [Google Scholar]
- Novel developments and trends of analytical methods for drug analysis in biological and environmental samples by molecularly imprinted polymers. TrAC - Trends Anal. Chem.. 2017;89:146-162.
- [CrossRef] [Google Scholar]
- Effect of Aliquat®336 on supported liquid membrane on electromembrane extraction of non-steroidal anti-inflammatory drugs. Microchem. J.. 2021;168
- [CrossRef] [Google Scholar]
- Coupling of two centrifugeless ultrasound-assisted dispersive solid/liquid phase microextractions as a highly selective, clean, and efficient method for determination of ultra-trace amounts of non-steroidal anti-inflammatory drugs in complicated matrices. Anal. Chim. Acta Elsevier B.V. 2018
- [CrossRef] [Google Scholar]
- Vortex-assisted surfactant-enhanced emulsification microextraction based on solidification of floating organic drop combined with high performance liquid chromatography for determination of naproxen and nabumetone. J. Chromatogr. A. 2015;1425:17-24.
- [CrossRef] [Google Scholar]
- An enhanced first derivative synchronous spectrofluorimetric method for determination of the newly co-formulated drugs, amlodipine and celecoxib in pharmaceutical preparation and human plasma. Spectrochim. Acta A Mol. Biomol. Spectrosc.. 2020;240:118533
- [CrossRef] [Google Scholar]
- Validation of rapid RP-HPLC method for concurrent quantification of amlodipine and celecoxib in pure and formulation using an experimental design. Microchem. J.. 2020;152:104365
- [CrossRef] [Google Scholar]
- A modified zeolite/iron oxide composite as a sorbent for magnetic dispersive solid-phase extraction for the preconcentration of nonsteroidal anti-inflammatory drugs in water and urine samples. J. Chromatogr. A. 2019;1603:33-43.
- [CrossRef] [Google Scholar]
- Comparison of ultrasound-enhanced air-assisted liquid-liquid microextraction and low-density solvent-based dispersive liquid-liquid microextraction methods for determination of nonsteroidal anti-inflammatory drugs in human urine samples. J. Pharm. Biomed. Anal.. 2015;111:297-305.
- [CrossRef] [Google Scholar]
- Bazregar, M., Rajabi, M., Yamini, Y., Asghari, A., Hemmati, M., 2016. Tandem air-agitated liquid-liquid microextraction as an efficient method for determination of acidic drugs in complicated matrices, Analytica Chimica Acta. Elsevier Ltd. 10.1016/j.aca.2016.03.005.
- Magnetic solid phase extraction of mefenamic acid from biological samples based on the formation of mixed hemimicelle aggregates on Fe3O4 nanoparticles prior to its HPLC-UV detection. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2014;945–946:46-52.
- [CrossRef] [Google Scholar]
- New spectrophotometric/chemometric assisted methods for the simultaneous determination of imatinib, gemifloxacin, nalbuphine and naproxen in pharmaceutical formulations and human urine. Spectrochim. Acta A Mol. Biomol. Spectrosc.. 2018;198:51-60.
- [CrossRef] [Google Scholar]
- Non-steroidal anti-inflammatory drugs (NSAIDs) and organ damage: A current perspective. Biochem. Pharmacol.. 2020;180:114147
- [CrossRef] [Google Scholar]
- Non-aqueous capillary electrophoresis for the analysis of acidic compounds using negative electrospray ionization mass spectrometry. J. Chromatogr. A. 2014;1323:163-173.
- [CrossRef] [Google Scholar]
- Sample preparation with solid phase microextraction and exhaustive extraction approaches: Comparison for challenging cases. Anal. Chim. Acta. 2015;873:14-30.
- [CrossRef] [Google Scholar]
- Past, present, and future of solid phase extraction: A review. Crit. Rev. Anal. Chem.. 2012;42:198-213.
- [CrossRef] [Google Scholar]
- Membrane-based microextraction techniques in analytical chemistry: A review. Anal. Chim. Acta. 2015;880:8-25.
- [CrossRef] [Google Scholar]
- Application of a hybrid ordered mesoporous silica as sorbent for solid-phase multi-residue extraction of veterinary drugs in meat by ultra-high-performance liquid chromatography coupled to ion-trap tandem mass spectrometry. J. Chromatogr. A. 2016;1459:24-37.
- [CrossRef] [Google Scholar]
- Online eluent-switching technique coupled anion-exchange liquid chromatography-ion trap tandem mass spectrometry for analysis of non-steroidal anti-inflammatory drugs in pig serum. J. Chromatogr. A. 2015;1422:222-229.
- [CrossRef] [Google Scholar]
- Rapid analysis of pharmaceuticals and personal care products in fish plasma micro-aliquots using liquid chromatography tandem mass spectrometry. J. Chromatogr. A. 2015;1383:104-111.
- [CrossRef] [Google Scholar]
- Optimisation by response surface methodology of microextraction by packed sorbent of non steroidal anti-inflammatory drugs and ultra-high performance liquid chromatography analysis of dialyzed samples. J. Pharm. Biomed. Anal.. 2016;125:114-121.
- [CrossRef] [Google Scholar]
- Validated method for the determination of piroxicam by capillary zone electrophoresis and its application to tablets. J. Anal. Methods Chem.. 2014;2014
- [CrossRef] [Google Scholar]
- In-situ synthesis of nanocubic cobalt oxide @ graphene oxide nanocomposite reinforced hollow fiber-solid phase microextraction for enrichment of non-steroidal anti-inflammatory drugs from human urine prior to their quantification via high-performance liqu. J. Chromatogr. A. 2021;1641:461984
- [CrossRef] [Google Scholar]
- Recent applications of microextraction sample preparation techniques in biological samples analysis. Biomed. Chromatogr.. 2021;35:1-31.
- [CrossRef] [Google Scholar]
- Polyamidoamine with a hyper-branched structure grafted on modified magnetic graphene oxide for the trace separation of diclofenac and acetaminophen followed by high-performance liquid chromatography determination. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2023;1227
- [CrossRef] [Google Scholar]
- Development and validation of RP-HPLC-PDA method for simultaneous determination of dexketoprofen and thiocolchicoside in pharmaceutical dosage form. J. Pharm. Res.. 2013;6:604-608.
- [CrossRef] [Google Scholar]
- Simultaneous electrochemical analysis of ibuprofen and paracetamol by clay modified carbon paste electrode: Analytical application in human blood. Anal. Bioanal. Electrochem.. 2022;14:18-31.
- [Google Scholar]
- Selective micellar electrokinetic chromatographic method for simultaneous determination of some pharmaceutical binary mixtures containing non-steroidal anti-inflammatory drugs. J. Pharm. Anal.. 2013;3:53-60.
- [CrossRef] [Google Scholar]
- Es’haghi, Z., Esmaeili-Shahri, E., 2014. Sol-gel-derived magnetic SiO2/TiO2 nanocomposite reinforced hollow fiber-solid phase microextraction for enrichment of non-steroidal anti-inflammatory drugs from human hair prior to high performance liquid chromatography, Journal of Chromatography B: Analytical Technologies in the Biomedical and Life Sciences. Elsevier B.V. 10.1016/j.jchromb.2014.09.030.
- Dispersive magnetic solid phase extraction exploiting magnetic graphene nanocomposite coupled with UHPLC-PDA for simultaneous determination of NSAIDs in human plasma and urine. J. Pharm. Biomed. Anal.. 2018;161:280-288.
- [CrossRef] [Google Scholar]
- An efficient sample preparation method based on dispersive liquid–liquid microextraction associated with back extraction for trace determination of acidic pharmaceuticals. Arab. J. Chem.. 2020;13:1924-1932.
- [CrossRef] [Google Scholar]
- Nanocrystalline cellulose as a biotemplate for preparation of porous titania thin film as a sorbent for thin film microextraction of ketorolac, meloxicam, diclofenac and mefenamic acid. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2020;1142:122039
- [CrossRef] [Google Scholar]
- Woven cotton yarn-graphene oxide-layered double hydroxide composite as a sorbent for thin film microextraction of nonsteroidal anti-inflammatory drugs followed by quantitation through high performance liquid chromatography. Anal. Chim. Acta. 2020;1097:94-102.
- [CrossRef] [Google Scholar]
- Detection and quantification of cimicoxib, a novel COX-2 inhibitor, in canine plasma by HPLC with spectrofluorimetric detection: Development and validation of a new methodology. J. Pharm. Biomed. Anal.. 2013;83:28-33.
- [CrossRef] [Google Scholar]
- Rapid spectrophotometric determination, characterization and anti-inflammatory efficacy evaluation of nanoencapsulated diclofenac sodium. J. Drug Deliv Sci. Technol.. 2014;24:361-366.
- [CrossRef] [Google Scholar]
- Porous eco–friendly fibers for on–line micro solid–phase extraction of nonsteroidal anti–inflammatory drugs from urine and plasma samples. J. Chromatogr. A. 2018;1574:18-26.
- [CrossRef] [Google Scholar]
- Cyclooxygenase selectivity of nonsteroidal anti-inflammatory drugs and risk of stroke. Arch. Intern. Med.. 2008;168:1219-1224.
- [CrossRef] [Google Scholar]
- Combustion fabrication of magnetic porous carbon as a novel magnetic solid-phase extraction adsorbent for the determination of non-steroidal anti-inflammatory drugs. Anal. Chim. Acta. 2019;1078:78-89.
- [CrossRef] [Google Scholar]
- Dispersive solid-phase extraction of non-steroidal anti-inflammatory drugs in water and urine samples using a magnetic ionic liquid hypercrosslinked polymer composite. J. Chromatogr. A. 2023;1689
- [CrossRef] [Google Scholar]
- Switchable-hydrophilicity solvent liquid–liquid microextraction of non-steroidal anti-inflammatory drugs from biological fluids prior to HPLC-DAD determination. J. Pharm. Biomed. Anal.. 2019;174:509-517.
- [CrossRef] [Google Scholar]
- Quantitative analysis of anti-inflammatory drugs using FTIR-ATR spectrometry. Spectrochim. Acta A Mol. Biomol. Spectrosc.. 2017;186:59-65.
- [CrossRef] [Google Scholar]
- Nanomaterial-based carbon paste electrodes for voltammetric determination of naproxen in presence of its degradation products. J. Anal. Methods Chem.. 2019;2019
- [CrossRef] [Google Scholar]
- Enantioselective HPLC-DAD method for the determination of etodolac enantiomers in tablets, human plasma and application to comparative pharmacokinetic study of both enantiomers after a single oral dose to twelve healthy volunteers. Talanta. 2014;130:506-517.
- [CrossRef] [Google Scholar]
- Huang, C., Li, Y., Yang, J., Peng, J., Tan, J., Fan, Y., Wang, L., Chen, J., 2018. Hyperbranched mixed-mode anion-exchange polymeric sorbent for highly selective extraction of nine acidic non-steroidal anti-inflammatory drugs from human urine, Talanta. Elsevier B.V. 10.1016/j.talanta.2018.07.033.
- Development of an extraction and purification method for the determination of multi-class pharmaceuticals and endocrine disruptors in freshwater invertebrates. Talanta. 2015;132:373-381.
- [CrossRef] [Google Scholar]
- Current developments of bioanalytical sample preparation techniques in pharmaceuticals. J. Pharm. Anal.. 2022;12:517-529.
- [CrossRef] [Google Scholar]
- Non-steroidal anti-inflammatory drugs in the environment: Where were we and how far we have come? Environ. Pollut.. 2020;267:115370
- [CrossRef] [Google Scholar]
- Facile and efficient preparation of amino bearing metal-organic frameworks-coated cotton fibers for solid-phase extraction of non-steroidal anti-inflammatory drugs in human plasma. J. Chromatogr. A. 2023;1705
- [CrossRef] [Google Scholar]
- Recent trends in microextraction techniques employed in analytical and bioanalytical sample preparation. Separations. 2017;4:1-15.
- [CrossRef] [Google Scholar]
- MCM-41 solid phase membrane tip extraction combined with liquid chromatography for the determination of non-steroidal anti-inflammatory drugs in human urine. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2013;940:59-65.
- [CrossRef] [Google Scholar]
- Joint inflammation: Insights of osteoarthritis, gouty and rheumatoid arthritis and its prevalence, mechanism, medications and remedies. Indian J. Pharm. Sci.. 2021;83:886-898.
- [CrossRef] [Google Scholar]
- Khaskheli, A.R., Sirajuddin, Sherazi, S.T.H., Mahesar, S.A., Kandhro, A.A., Kalwar, N.H., Mallah, M.A., 2013. Estimation of ibuprofen in urine and tablet formulations by transmission Fourier Transform Infrared spectroscopy by partial least square. Spectrochim Acta A Mol Biomol Spectrosc 102, 403–407. 10.1016/j.saa.2012.10.021.
- Acute pain: A multifaceted challenge - The role of nimesulide. Curr. Med. Res. Opin.. 2016;32:23-36.
- [CrossRef] [Google Scholar]
- Screening of over 100 drugs in horse urine using automated on-line solid-phase extraction coupled to liquid chromatography-high resolution mass spectrometry for doping control. J. Chromatogr. A. 2017;1490:89-101.
- [CrossRef] [Google Scholar]
- Lakshmana, S., K. Suriyaprakash, T.N., 2012. Extraction of Drug from the Biological Matrix: A Review. Applied Biological Engineering - Principles and Practice. 10.5772/32455.
- Simultaneous determination of flurbiprofen and its hydroxy metabolite in human plasma by liquid chromatography-tandem mass spectrometry for clinical application. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2014;971:58-63.
- [CrossRef] [Google Scholar]
- Determination of ibuprofen enantiomers in breast milk using vortex-assisted matrix solid-phase dispersion and direct chiral liquid chromatography. J. Chromatogr. A. 2017;1514:88-94.
- [CrossRef] [Google Scholar]
- Recent advances in applications of metal–organic frameworks for sample preparation in pharmaceutical analysis. Coord. Chem. Rev.. 2020;411:213235
- [CrossRef] [Google Scholar]
- A rapid analysis of piroxicam in beagle plasma applying evaporation-free liquid-liquid extraction by supercritical fluid chromatography-tandem mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2018;1100–1101:93-99.
- [CrossRef] [Google Scholar]
- Zr-based metal-organic framework-modified cotton for solid phase micro-extraction of non-steroidal anti-inflammatory drugs. J. Chromatogr. A. 2018;1576:19-25.
- [CrossRef] [Google Scholar]
- Novel metal ion-mediated complex imprinted membrane for selective recognition and direct determination of naproxen in pharmaceuticals by solid surface fluorescence. Talanta. 2013;116:460-467.
- [CrossRef] [Google Scholar]
- Simultaneous determination of parecoxib sodium and its active metabolite valdecoxib in rat plasma by UPLC-MS/MS and its application to a pharmacokinetic study after intravenous and intramuscular administration. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2016;1022:220-229.
- [CrossRef] [Google Scholar]
- Microextraction by packed sorbent and high performance liquid chromatography determination of seven non-steroidal anti-inflammatory drugs in human plasma and urine. J. Chromatogr. A. 2014;1367:1-8.
- [CrossRef] [Google Scholar]
- Opportunities and obstacles for microsampling techniques in bioanalysis: Special focus on DBS and VAMS. J. Pharm. Biomed. Anal. 2020
- [CrossRef] [Google Scholar]
- A comparative study of the novel spectrophotometric methods versus conventional ones for the simultaneous determination of Esomeprazole magnesium trihydrate and Naproxen in their binary mixture. Spectrochim. Acta A Mol. Biomol. Spectrosc.. 2015;151:538-546.
- [CrossRef] [Google Scholar]
- Development and validation of ultra-performance liquid chromatography method for the determination of meloxicam and its impurities in active pharmaceutical ingredients. Ann. Pharm. Fr.. 2018;76:187-200.
- [CrossRef] [Google Scholar]
- Ibuprofen analysis in blood samples by palladium particles-impregnated sodium montmorillonite electrodes: Validation using high performance liquid chromatography. Mater. Sci. Eng. C. 2016;69:616-624.
- [CrossRef] [Google Scholar]
- Chitosan-based molecular imprinted polymer for extraction and spectrophotometric determination of ketorolac in human plasma. Spectrochim. Acta A Mol. Biomol. Spectrosc.. 2020;241:118668
- [CrossRef] [Google Scholar]
- Application of statistical experimental design to the optimisation of microextraction by packed sorbent for the analysis of nonsteroidal anti-inflammatory drugs in human urine by ultra-high pressure liquid chromatography. J. Chromatogr. A. 2013;1304:1-9.
- [CrossRef] [Google Scholar]
- Porphyrin-functionalized graphene oxide sheets: An efficient nanomaterial for micro solid phase extraction of non-steroidal anti-inflammatory drugs from urine samples. J. Chromatogr. A. 2019;1607:460387
- [CrossRef] [Google Scholar]
- Programmable flow-based dynamic sorptive microextraction exploiting an octadecyl chemically modified rotating disk extraction system for the determination of acidic drugs in urine. J. Chromatogr. A. 2014;1368:64-69.
- [CrossRef] [Google Scholar]
- Determination of non-steroidal anti-inflammatory drugs in water and urine using selective molecular imprinted polymer extraction and liquid chromatography. J. Pharm. Biomed. Anal.. 2016;131:48-53.
- [CrossRef] [Google Scholar]
- Need to Develop New Nonsteroidal Anti-Inflammatory Drug Formulations. Clin. Ther.. 2012;34:1954-1963.
- [CrossRef] [Google Scholar]
- Determination of phenylbutazone and flunixin meglumine in equine plasma by electrochemical-based sensing coupled to selective extraction with molecularly imprinted polymers. Sens. Actuators B Chem.. 2013;179:226-231.
- [CrossRef] [Google Scholar]
- Correlation analysis based on the hydropathy properties of non-steroidal anti-inflammatory drugs in solid-phase extraction (SPE) and reversed-phase high performance liquid chromatography (HPLC) with photodiode array detection and their applications to bio. J. Chromatogr. A. 2019;1605:360351
- [CrossRef] [Google Scholar]
- Toxicopathological overview of analgesic and anti-inflammatory drugs in animals. J. Appl. Pharm. Sci.. 2012;2:149-157.
- [Google Scholar]
- Polyester fabric-based nano copper-polyhedral oligomeric silsesquioxanes sorbent for thin film extraction of non-steroidal anti-inflammatory drugs. Anal. Chim. Acta. 2023;1270
- [CrossRef] [Google Scholar]
- Simple and sensitive method for the analysis of ketorolac in human plasma using high-performance liquid chromatography. J. Young Pharm.. 2013;5:98-101.
- [CrossRef] [Google Scholar]
- Establishment of analytical method for quantification of anti-inflammatory agents co-nanoencapsulated and its application to physicochemical development and characterization of lipid-core nanocapsules. Arab. J. Chem.. 2020;13:2456-2469.
- [CrossRef] [Google Scholar]
- Critical development by design of a rugged HPLC-MS/MS method for direct determination of ibuprofen enantiomers in human plasma. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2015;992:67-75.
- [CrossRef] [Google Scholar]
- A novel sorbent based on electrospun for electrically-assisted solid phase microextraction of six non-steroid anti-inflammatory drugs, followed by quantitation with HPLC-UV in human plasma samples. Anal. Chim. Acta 2023341839
- [CrossRef] [Google Scholar]
- High-throughput determination of nonsteroidal anti-inflammatory drugs in human plasma by HILIC-MS/MS. J. Pharm. Biomed. Anal.. 2014;88:71-80.
- [CrossRef] [Google Scholar]
- Electrochemical determination of nepafenac topically applied nonsteroidal anti-inflammatory drug using graphene nanoplatelets-carbon nanofibers modified glassy carbon electrode. J. Electroanal. Chem.. 2018;817:30-35.
- [CrossRef] [Google Scholar]
- New developments in microextraction techniques in bioanalysis. A review. Anal. Chim. Acta. 2016;905:8-23.
- [CrossRef] [Google Scholar]
- Simultaneous quantitative determination of celecoxib and its two metabolites using liquid chromatography-tandem mass spectrometry in alternating polarity switching mode. J. Pharm. Biomed. Anal.. 2015;107:32-39.
- [CrossRef] [Google Scholar]
- Potentiometric determination of Diclofenac using an ion-selective electrode prepared from polypyrrole films. J. Electroanal. Chem.. 2014;732:11-16.
- [CrossRef] [Google Scholar]
- I. Olives, A., Gonzalez-Ruiz, V., Antonia Martin, M., 2012. Isolation and quantitative methods for analysis of non-steroidal anti-inflammatory drugs. Antiinflamm Antiallergy Agents Med Chem 11, 65–95. 10.2174/187152312803476273.
- An in vitro spectrometric method for determining the partition coefficients of non-steroidal anti-inflammatory drugs into human erythrocyte ghost membranes. Spectrochim. Acta A Mol. Biomol. Spectrosc.. 2013;104:461-467.
- [CrossRef] [Google Scholar]
- Method development and validation of mefenamic acid, dicyclomine hydrochloride, and pamabrom of marketed formulation by ultraviolet. Pharmaspire. 2018;10(2):64-67.
- [Google Scholar]
- Boron-doped diamond electrode as efficient sensing platform for simultaneous quantification of mefenamic acid and indomethacin. Diam. Relat. Mater.. 2020;105
- [CrossRef] [Google Scholar]
- Modern trends in solid phase extraction: New sorbent media. TrAC - Trends Anal. Chem.. 2016;77:23-43.
- [CrossRef] [Google Scholar]
- High performance liquid chromatography with photo diode array for separation and analysis of naproxen and esomeprazole in presence of their chiral impurities: Enantiomeric purity determination in tablets. J. Chromatogr. A. 2017;1497:110-117.
- [CrossRef] [Google Scholar]
- An effective microfluidic based liquid-phase microextraction device (μLPME) for extraction of non-steroidal anti-inflammatory drugs from biological and environmental samples. Anal. Chim. Acta. 2016;946:56-63.
- [CrossRef] [Google Scholar]
- Determination of mefenamic acid in urine and pharmaceutical samples by HPLC after pipette-tip solid phase microextraction using zinc sulfide modified carbon nanotubes. Anal. Methods. 2016;8:5978-5983.
- [CrossRef] [Google Scholar]
- Solid phase extraction as a cleanup step before microextraction of diclofenac and mefenamic acid using nanostructured solvent. Talanta. 2013;105:173-178.
- [CrossRef] [Google Scholar]
- Micro QuEChERS-based method for the simultaneous biomonitoring in whole blood of 360 toxicologically relevant pollutants for wildlife. Sci. Total Environ.. 2020;736:139444
- [CrossRef] [Google Scholar]
- Non-steroidal anti-inflammatory drugs and colorectal cancer risk in a large, prospective cohort. Am. J. Gastroenterol.. 2011;106:1340-1350.
- [CrossRef] [Google Scholar]
- Forced degradation of nepafenac: Development and validation of stability indicating UHPLC method. J. Pharm. Biomed. Anal.. 2016;123:42-52.
- [CrossRef] [Google Scholar]
- Ryu, J.H., Park, J.S., Jo, M. ho, Kim, J. Il, Shim, W.S., Kim, B.H., Yim, S.V., Hong, J., Lee, K.T., 2015. Development and validation of an LC-MS/MS method for the determination of pelubiprofen and its active metabolite, trans-alcohol, in human plasma and its application to pharmacokinetic study. J Chromatogr B Analyt Technol Biomed Life Sci 983–984, 62–67. 10.1016/j.jchromb.2014.12.037.
- Sample preparation for the analysis of drugs in biological fluids. Handbk. Anal. Separ.. 2020;7:1-13.
- [CrossRef] [Google Scholar]
- Electrospun NiFe layered double hydroxide/Nylon 6 composite nanofibers as a sorbent for micro solid phase extraction by packed sorbent of non-steroidal anti-inflammatory drugs in human blood. J. Chromatogr. A. 2020;1614:460718
- [CrossRef] [Google Scholar]
- Simultaneous determination for oxicam non-steroidal anti-inflammatory drugs in human serum by liquid chromatography-tandem mass spectrometry. Forensic Sci. Int.. 2013;227:100-102.
- [CrossRef] [Google Scholar]
- A new approach for microextraction of non-steroidal anti-inflammatory drugs from human urine samples based on in-situ deep eutectic mixture formation. J. Mol. Liq.. 2018;272:738-745.
- [CrossRef] [Google Scholar]
- HPLC-MS/MS determination of non-steroidal anti-inflammatory drugs in bovine milk based on simultaneous deep eutectic solvents formation and its solidification. Microchem. J.. 2019;150:104080
- [CrossRef] [Google Scholar]
- An effervescence-assisted dispersive liquid–liquid microextraction based on deep eutectic solvent decomposition: Determination of ketoprofen and diclofenac in liver. Microchem. J.. 2020;156:104837
- [CrossRef] [Google Scholar]
- Pharmacological analysis of cyclooxygenase-1 in inflammation. PNAS. 1998;95:13313-13318.
- [CrossRef] [Google Scholar]
- Smith, H.S., 2014. Nonsteroidal Anti-Inflammatory Drugs; Acetaminophen, Encyclopedia of the Neurological Sciences. Elsevier Ltd. 10.1016/B978-0-12-385157-4.00214-1.
- A review of analytical techniques for determination of oxicams, nimesulide and nabumetone. Talanta. 2009;77:925-942.
- [CrossRef] [Google Scholar]
- Development of a detection method for 10 non-steroidal anti-inflammatory drugs residues in four swine tissues by ultra-performance liquid chromatography with tandem mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2023;1223
- [CrossRef] [Google Scholar]
- Development and validation of Ketorolac Tromethamine in eye drop formulation by RP-HPLC method. Arab. J. Chem.. 2017;10:S928-S935.
- [CrossRef] [Google Scholar]
- Application of ultra-sensitive GC-QqQ-MS/MS (MRM) method for the determination of diclofenac in whole blood samples without derivatization. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2021;1179
- [CrossRef] [Google Scholar]
- Simultaneous determination of polar pharmaceuticals and personal care products in biological organs and tissues. J. Chromatogr. A. 2014;1355:193-205.
- [CrossRef] [Google Scholar]
- Fast off-line FPSE-HPLC-PDA determination of six NSAIDs in saliva samples. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2020;1144:122082
- [CrossRef] [Google Scholar]
- Solid-phase microextraction: A powerful sample preparation tool prior to mass spectrometric analysis. J. Mass Spectrom.. 2004;39:233-254.
- [CrossRef] [Google Scholar]
- Magnetic solid phase extraction for determination of drugs in biological matrices. TrAC - Trends Anal. Chem.. 2017;89:41-52.
- [CrossRef] [Google Scholar]
- Solid phase microextraction with poly(deep eutectic solvent) monolithic column online coupled to HPLC for determination of non-steroidal anti-inflammatory drugs. Anal. Chim. Acta. 2018;1018:111-118.
- [CrossRef] [Google Scholar]
- Determination of vitacoxib, a novel COX-2 inhibitor, in equine plasma using UPLC–MS/MS detection: Development and validation of new methodology. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2017;1061–1062:270-274.
- [CrossRef] [Google Scholar]
- Simultaneous determination of ibuprofen and its metabolites in complex equine urine matrices by GC-EI-MS in excretion study in view of doping control. Journalof Pharmaceutical and Biomedical Analysis. 2018;152:279-288.
- [CrossRef] [Google Scholar]
- Wongrakpanich, S., Wongrakpanich, A., Melhado, K., Rangaswami, J., 2018. A Comprehensive Review of Non-Steroidal Anti-Inflammatory Drug Use in The Elderly. Aging Dis 9, 143–150. 10.14336/AD.2017.0306.
- In-line carbon nanofiber reinforced hollow fiber-mediated liquid phase microextraction using a 3D printed extraction platform as a front end to liquid chromatography for automatic sample preparation and analysis: A proof of concept study. Talanta. 2018;185:611-619.
- [CrossRef] [Google Scholar]
- Recent development and applications of dispersive liquid-liquid microextraction. J. Chromatogr. A. 2013;1295:1-15.
- [CrossRef] [Google Scholar]
- Simultaneous determination of naproxen and esomeprazole in beagle dog plasma by supercritical fluid chromatography-tandem mass spectrometry coupled with evaporation-free liquid-liquid extraction. J. Pharm. Biomed. Anal.. 2021;193:113668
- [CrossRef] [Google Scholar]
- Determination of diclofenac in pharmaceutical preparations by voltammetry and gas chromatography methods. J. Pharm. Anal.. 2015;5:153-160.
- [CrossRef] [Google Scholar]
- TiO2 nanoparticles and C-Nanofibers modified magnetic Fe3O4 nanospheres (TiO2@Fe3O4@C–NF): A multifunctional hybrid material for magnetic solid-phase extraction of ibuprofen and photocatalytic degradation of drug molecules and azo dye. Talanta. 2020;213:120813
- [CrossRef] [Google Scholar]
- Multi-residue determination of 210 drugs in pork by ultra-high-performance liquid chromatography–tandem mass spectrometry. J. Chromatogr. A. 2016;1463:49-59.
- [CrossRef] [Google Scholar]
- Green synthesis of magnetic carbon nanodot/graphene oxide hybrid material (Fe3O4@C-nanodot@GO) for magnetic solid phase extraction of ibuprofen in human blood samples prior to HPLC-DAD determination. J. Pharm. Biomed. Anal.. 2020;179:1-10.
- [CrossRef] [Google Scholar]
- Application of the ratio difference spectrophotometry to the determination of ibuprofen and famotidine in their combined dosage form; Comparison with previously published spectrophotometric methods. Spectrochim. Acta A Mol. Biomol. Spectrosc.. 2015;143:251-255.
- [CrossRef] [Google Scholar]
- Rapid determination of 54 pharmaceutical and personal care products in fish samples using microwave-assisted extraction—Hollow fiber—Liquid/solid phase microextraction. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2017;1051:41-53.
- [CrossRef] [Google Scholar]
- Determination of naproxen in human urine by capillary electrophoresis with chemiluminescence detection. Glob Drugs Ther. 2018;3:1-5.
- [CrossRef] [Google Scholar]
- Ultra-performance liquid chromatography-tandem mass spectrometry determination and depletion profile of flunixin residues in tissues after single oral administration in rabbits. J. Chromatogr. B Anal. Technol. Biomed. Life Sci.. 2013;934:8-15.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.105446.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







