Translate this page into:
Benzoic-D5 acid as D2 receptor agonist in the treatment of rotenone induced Parkinson’s disease in mice
⁎Corresponding author. umar.sahibzada@gmail.com (Muhammad Umar Khayam Sahibzada)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Abstract
Parkinson's disease is a progressive neurological disorder that affects movement. It is caused by the degeneration of dopamine-producing neurons in the brain. There is no cure for Parkinson's disease, but there are treatments available that can help manage the symptoms. Medications such as levodopa, dopamine agonists and Amantadine (Amd) can help to increase dopamine levels in the brain. Sodium benzoate (Benzoic-D5) is used as preservative in foods due to its antimicrobial potential. Benzoic-D5 showed a pivotal role in ameliorating the symptoms of Parkinson’s disease (PkD) in animal model. The current study is designed to probe the anti-Parkinson potential of Benzoic-D5 in the rotenone (ROT)-induced PkD mice. Behavioral studies showed improvement in sensory-motor functions, and neuromuscular strength in wire hanging test, grip strength, vertical rod test, and swim test. Evaluation of biochemical markers suggested increase in catalase (CAT) and superoxide dismutase (SOD), but decreases in the malondialdehyde (MDA) activity. Dopamine, dihydroxyphenyl acetic acid (DOPAC), and homovanillic acid (HVA) levels were improved in Benzoic-D5-treated animals. Moreover, there were down regulation of pro-inflammatory markers like TNF-α, NF-Kβ, and up regulation of anti-inflammatory IL-4 in the RT-PCR analysis. In molecular docking Benzoic-D5 showed strong binding affinity with an active site of Dopamine D2 receptor. Afterwards, ADMET analysis points to the selected drug’s acceptable properties. Conclusively, in vivo, and in silico studies propose that Benzoic-D5 possesses potentials to ameliorate symptoms of PkD. Binding energy data of Benzoic-D5 (-6.2), shows promising results compared to ROT (-6.0), and Amd (5.8).
Keywords
Sodium Benzoate
Dopamine
Sensory-motor functions
Behavioral modulation
- PKD
-
Parkinson’s disease
- ROT
-
Rotenone
- CAT
-
Catalase
- SOD
-
Superoxide dismutase
- MDA
-
Malondialdehyde
- DOPAC
-
Dihydroxyphenyl acetic acid
- HVA
-
Homovanillic acid
- TNF-α
-
Tumor necrosis factor-α
- NF-Kβ
-
Nuclear factor kappa
- IL-4
-
Interleukin-4
- RT-PCR
-
Real time-Polymerase chain reaction
- EDTA
-
Ethylenediaminetetraacetic acid
- RMSD
-
Root Mean Square Deviation
- LYS
-
Lysine
- VAL
-
Valine
- TYR
-
Tyrosine
- ILE
-
Isoleucine
- ARG
-
Arginine
- GLN
-
Glutamine
- GLY
-
Glycine
- LEU
-
Leucine
- ALA
-
Alanine
- ASP
-
Aspartic acid
- PHE
-
Phenylalanine
- ASN
-
Asparagine
Abbreviations
1 Introduction
Parkinson disease (PkD) is a neurodegenerative disease. PkD is characterized by tremor, bradykinesia, postural instability, stiffness, abnormal body posture, and akinesia. Primary cause of these indications are usually psychological including depression, general non-motor disabilities such as olfactory disorders, constipation, and sleep disorders (Baiano et al., 2020, Innos and Hickey 2021). Moreover, incidence of PkD is 5% at 80, and 1% at 65 years respectively (Tysnes and Storstein 2017).
The ROT is globally known because of its insecticidal, acaricidal, and pesticidal properties (Pan-Montojo et al., 2010). ROT is extracted from stem, root of Derris elliptica (Benth), and Derris malaccensis (Prain), which are native to Southeast Asia, and Indian subcontinent. It is used in powder form to control parasitic mites on chicken as well as lice and ticks on large domestic animals, and plants (Cannon et al., 2009). Countries with agriculture as having economic back-bone are affected directly or indirectly by extensive use of ROT (being an ingredient of pesticides) (Coulom and Birman 2004). Population of the rural areas (70 %) is more inclined to be affected by the exposure of ROT (Sherer et al., 2003).
The ROT is employed as a key neuro-toxicant for induction of animal models of PkD (Bisbal and Sanchez 2019). Exposure to pesticides and insecticides causes neurodegenerative effects. Its high lipophilicity makes it easily cross the blood–brain barrier, and enter cells without relying on specific transporters. Generally understood mechanism by which ROT induces PkD in animals, include damage to dopamine neurons in substantia nigra, primarily as a result of increased oxidative-stress. It has been previously established that ROT inhibits mitochondrial respiratory chain complex-I, leading to oxidative-stress and subsequent deterioration of dopaminergic neurons (Heinz et al., 2017). The ROT-induced suppressed activities of cytosolic antioxidant enzymes superoxide dismutase, and catalase showed further significant increase ROT induced oxidative damages causes the development of neurochemical, neuropathological and behavioral features of PkD in animals and degeneration of dopaminergic neurons (Saravanan et al., 2006).
Amantadine (Amd) is used as anti-parkinson drug for the treatment of parkinsonian symptoms in adults with PkD. These ameliorating effects are suggestive of its potential to up-regulate the dopamine levels (Müller et al., 2019). Due to non-allergic and non-irritating properties, Sodium benzoate (Benzoic-D5) is widely used in a range of cosmetic formulations as preservative agent (Linke et al., 2018). It is also a key ingredient in processed food products (Stanojevic et al., 2009). Its safety and reliability are unquestionable (Williams and Lock 2005). In addition, it has diverse applications in pharmaceutical industry (Hong et al., 2009).
Benzoic-D5 is known to have neuro-protective effects in certain neurological disorders like multiple sclerosis (Mehdi and Hasson 2020). Its excretion in human urine makes basis for its use as a treatment venture for acute hepatic encephalopathy (Bridges et al., 1970, Kubota and Ishizaki 1991). It is also preferred as conjugate drug for controlling hyper-ammonemia that is often found in patients with urea-cycle disorders (Enns et al., 2007), particularly in children (Scaglia et al., 2004, Huh and Farrell 2011).
The objectives of the study were to evaluate the extent of ROT in development of PkD, and also to evaluate the effectiveness of Benzoic-D5 in modulation of behavioral activities as well as biochemical evaluation in PkD mice model. In addition, in-silico studies identified the potential binding affinities of the Amd and Benzoic-D5 with D2 receptors.
2 Material and methods
2.1 Animals
Twenty-eight (28) male albino mice (10–12 months old; 28–32 g), were taken from animal house of The University of Lahore (UOL), Lahore-Pakistan. Mice were housed is separate cages at 12-hours light / dark cycles, and kept under standard temperature (22 ± 2 °C) and humidity (60 ± 2%) with free access to food, and water. Experimental procedures and animal handling were approved by Institutional Research Ethics Committee (IREC # PHR-33153), Faculty of Pharmacy, The University of Lahore, Lahore -Pakistan.
2.2 Drugs and chemicals used
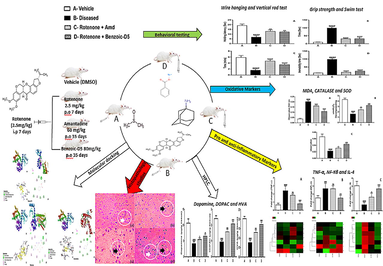
Formalin, acetonitrile (Merck), normal saline (Searle Pakistan LTD), water for injection (Otsuka Pakistan Ltd), Rotenone (Sigma, St. Louis, MO, U.S.A.), Amantadine (PK MERZ) (Brooks Pharmaceutical), Sodium benzoate (CHC-Co), hydrogen peroxide, thiobarbituric acid, phosphoric acid, TRIzol (Ambion), Syber green, cDNA kit, primers, and DNA ladder (Thermo Scientific Fischer), Dopamine, Dihydroxyphenyl acetic acid (DOPAC), Homovanillic acid (HVA), methanol, hydrochloric acid, ethanol, distilled water, paraffin, sodium sulphite (Sigma Aldrich), potassium hydroxide, acetic acid (Daejung), 5,5′- dithiobis-2-nitrobenzoic acid (Ark Pharma), were procured from the relevant company suppliers. All the reagents, and chemicals used in study were 100% pure and of analytical grade (Jing et al., 2007).(See Fig. 1)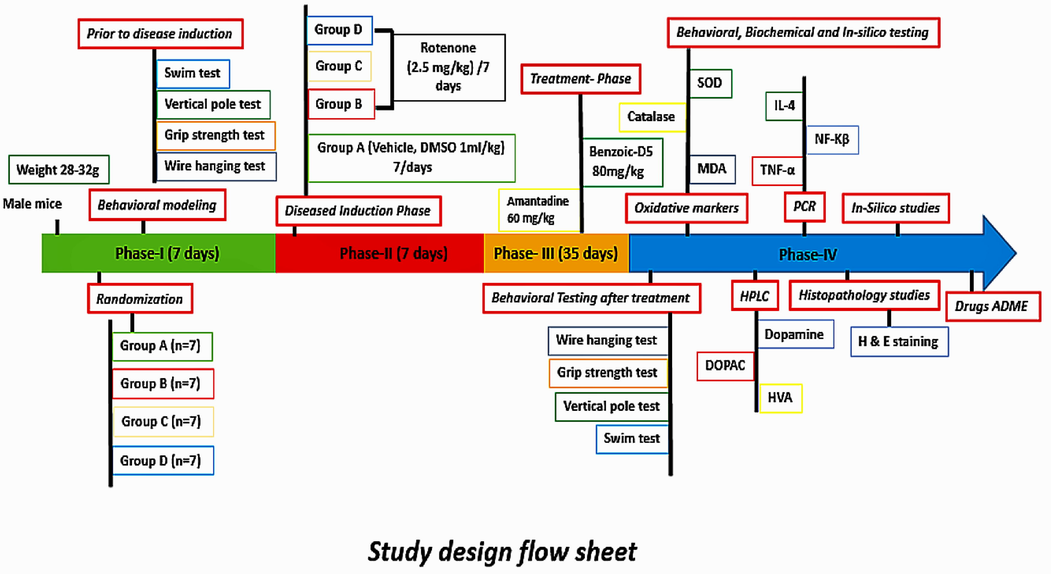
Study design flow sheet.
2.3 Study design
Study was divided into four phases:
Phase-I: Behavioral modeling of the study animals was performed. All 28 mice were trained, and randomly divided into four groups (7 mice in each).
Phase-II: PkD was induced by administering ROT 2.5 mg/kg/day intra-peritoneally (i.p) to groups B, C and D for 7 days. Whereas, in mice of group A, DMSO (vehicle) was introduced (1 ml/kg) i.p for 7 days as negetive control.
Phase-III: Treatment phase, Benzoic-D5, and Amd were given to animals for 5 weeks per oral (p.o), subsequently, behavioral parameters were recorded.
Phase-IV: Animals were sacrificed, blood and organs were collected for biochemical, and histopathological analysis. In-silico studies, pharmacokinetics, and toxicological studies were also carried out using swiss ADME.
2.4 Study groups
The study lasted for 50 days. Animals were randomized into four groups was followed by 7-day; Phase-I (behavioral tests in normal mice). Phase-II lasts for day 8 to day 14, and characterized by disease induction through ROT. Phase-III (treatment phase) starts at day 15, and lasts till 35 days. Phase-IV entails repetition of behavioral, and biochemical tests as well as in-silico analysis.
Group A (Vehicle), (DMSO 1 ml/kg) (Serenilo et al., 2016), i.p for 7 days.
Group B (ROT 2.5 mg/kg, i.p/1 weekdays): Parkinsonism was induced by injecting ROT (2.5 mg/kg, i.p/day) for 7 days (Cannon et al., 2009).
Group C (ROT 2.5 mg/kg, i.p/1 week) + (Amd 60 mg/kg) (Kaefer et al., 2010), per oral (p.o) for 5 weeks after disease induction.
Group D (ROT 2.5 mg/kg, i.p/1 week) + (Benzoic-D5 80 mg/kg) (Kalpana and Rajeswari 2019), p.o for 5 weeks after disease induction.
All animals were observed for 30 min post injection, and then at hourly intervals for next 3 h. PkD was assessed by behavioral testing.
2.5 Dilution
Briefly, ROT was first dissolved in dimethyl sulfoxide (DMSO) to prepare stock solution. Rotenone (50 mg) was weighed on a standard scale before dissolving in 1 ml of 100% dimethyl sulfoxide (DMSO), and stored at − 80 °C for further use. Before injection, the stock solution of ROT was thawed, and further diluted in sunflower oil to obtain a final concentration of 2.5 mg/ml. The sunflower oil used as vehicle of rotenone. To evaluate its neuroprotective efficacy, Benzoic-D5 was initially diluted in water to obtain a final concentration of 80 mg/1 ml, mice that were used as controls received an equivalent volume of vehicle (Javed et al., 2016).
2.6 Biochemical tests
The hematological samples were collected via heart puncture, AST, ALT, Cholesterol, triglyceride, and random glucose were determined by automatic analyzers respectively (automatic biochemistry, and hematological analyzers). WBC count, neutrophils, eosinophils, lymphocytes, were calculated via hematology analyzer (Prasad and Hung 2020).
2.7 Behavioral testing
2.7.1 Wire hanging
A standard linear wire hang apparatus was constructed, comprising a large plastic box of 55 cm long, 40 cm wide by 35 cm deep with a 2.5 mm of diameter wire suspended in the top center of the longest dimension. Wire hanging apparatus was constructed using a 32 cm circle of 2.5 mm wire suspended on a pulley and placed so that the bottom of the loop of wire was positioned in the middle, and at the top of an identical plastic box (Hoffman and Winder 2016).
2.7.2 Grip strength
The grip strength test is a modest non-intrusive method intended to assess mouse muscle power in vivo. In here, we take benefit of the animal's affinity to grip a flat metal bar or framework while suspended to its tail or each of the four appendages. The inverted screen is a 43 cm square of wire mesh consisting of 12 mm squares of 1 mm diameter wire. We placed the mouse in the centre of the wire mesh screen, started a stop clock, and rotated the screen to an inverted position over 2 sec, with the mouse's head declining first. We held the screen steadily 40–50 cm above a padded surface (Takeshita et al., 2017).
2.7.3 Vertical pole
Vertical pole test is used to measure the sensorimotor function of mice. PkD motor tests provide a good read-out of neurological function. Each mouse was placed head upwards at the center of a round pole that was inclined at 90°, and performance was determined by the latency(s) of the mouse to turn downwards, and completely descended the pole. In the habituation period, one day prior to testing, each mouse was allowed to descend through pole. Each testing session lasted for a maximum of 180 s (Kim et al., 2010).
2.7.4 Forced swim
We subjected the animals to a “pre-test session'' to avoid variations, and for maintaining consistency in the immobility time between different groups. Briefly, mice were forced to swim individually for 15 min, in a glass cylinder (21/12 cm) containing fresh water up to a height of 9 cm at a temperature of 25 < 2 °C. This constituted the “pretest session”. Twenty - four hours later, the animals were treated with either a drug (test group) or (vehicle), and each animal was again forced to swim in a similar environment for a period of 6 min in a “real-test session”, and immobility time was recorded (Kara et al., 2018).
2.8 Anti-oxidant potential
2.8.1 MDA Assay
Tissue Malondialdehyde MDA level is one of the final products of polyunsaturated fatty acids peroxidation in the cells was assessed using spectrophotometric method of Ohkawa et al. [16]. MDA determined the measure thio-barbituric acid reactive species (TBARS). One molecule of MDA responds with two molecules of thio-barbituric acid within the acidic medium at 95 °C temperature for 20 mins to make TBARS. The resultant pink item absorbance was estimated at 532 nm. Lipid per oxidation, was quantifiable as indicated by the technique of okahawa (Ohkawa et al., 1979).
2.8.2 Catalase Assay
The determination of serum catalase was done by using UV spectrophotometric method. The measurement was carried out at 240 nm. The serum was homogenized in 150 millimolar phosphate buffer of pH 7 at 4 °C, and was centrifuged at 6000 rpm. The activity of catalase was expressed in unit per gram of serum sample. The absorbance was measured by UV spectrophotometer, and values were compared with standard curve, which was generated from known enzyme catalase (Goth 1991).
2.8.3 Superoxide dismutase assay (SOD)
The quantitative measure of Superoxide dismutases SOD are a group of enzymes that catalyze the dismutation of superoxide radicals (O2 − ) to molecular oxygen (O2) and hydrogen depends on intensity of enzymes to hamper the scavenging effect on superoxide anion radicals, and was assessed using the NBT reduction method with some modifications. Absorbance of the resulting mixture was read at 560 nm against a blank. The activity was monitored as mentioned before (Bhangale and Acharya 2016).
2.9 Evaluation of mRNA expression levels of TNF-α, NFκB, and IL-4
Blood was collected at the end of the investigation, RNA extraction was done through the TRIzol system, getting typical methodology with regards to manufacturer's bearings (Thermo Fisher Scientific, America).
Item was augmented using thermal cycler with 45 cycles of denaturation (95 °C for 10 s), annealing (60 °C for 20 s), and extension (72 °C for 30 s), and evaluated by utilizing (RT-PCR) through Bio-Rad framework. The cDNA was derived from mRNA. The suitable primer was utilized for the pro-inflammatory arbiters for creating the duplicates by RT-PCR. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as a house-keeping gene (Li et al., 2017) with sequence:
5-TCTCTGCTCCT CCCTGTTCT-3′Forward,
5-CTTGCCGTGGGTAGAG TCAT-3′Reverse.
2.10 HPLC
HPLC test was performed to determine the dopamine, DOPAC, and HVA contents. Sample was run in HPLC system (Shimadzo, Japan.), having diode array detector (DAD) and C-18 column were used. The mobile phase was solvent A which consisted of methanol, and deionized water (0.1% acetic acid, v/v) with a ratio of 60:40. The flow rate was 0.8 ml/min, and dopamine was eluted within 15 min. Levels of dopamine, DOPAC and HVA were analyzed at 280, 430, and 312 nm respectively (Baranowska and Zydron 2002).
2.10.1 Analysis of dopamine, DOPAC and HVA
Four mice from each of the groups were decapitated following injecting phenobarbital 40 mg/kg (Tien et al., 2015). The striatum was collected, and then was homogenized in ice-cold 0.1 M HClO4 containing 0.01% EDTA. After being centrifuged (14,000 g, 4 °C, 10 min), the concentration of dopamine, and its metabolites (HVA, and DOPAC) in supernatant were assayed by high performance liquid chromatography (HPLC). Concentrations of dopamine, HVA, and DOPAC were expressed as μg/g of the tissue weight, determination of (DA, HVA). A strong cation-exchange column was used for DA analysis and a C-18 column was used for the analysis of the metabolites (Mounsey et al., 2015).
2.10.2 Sample preparation
Stock solutions of DA (0.5 mmol/L), DOPAC (0.6 mmol/L) and HVA (0.5 mmol/L) were prepared in 0.1 mol/L perchloric acid (HClO4). Calibration standards, with the concentration ranged from 66 nmol/L to 1.3 μmol/L for DA, 74 nmol/L to 1.5 μmol/L for DOPAC and 69 nmol/L to 1.4 μmol/L for HVA, were freshly prepared by successive dilution of the stock solutions with mobile phase and stored at 4℃ as used before this study (Jing et al., 2007).
2.11 Brain histopathology
The mice were decapitated, brain tissue containing the substentia nigra were harvested. These were then mounted, and subsequently dried out through ascending grades of ethanol to absolute ethanol. These were then purged in xylene, impregnated, and installed in paraffin wax (softening point 56 °C). The segments were de-waxed in xylene, hydrated through dropping descending grades of ethanol. Lastly, 5um thick brain tissues were stained with hematoxylin, and eosin dyes (Schneider et al., 2002). The slides were analyzed by pathologist in a blind manner at 40x.
2.12 Pharmacokinetics and toxicity risk assessment
The Swiss-ADME web tool was used to calculate the descriptors such as pharmacokinetics properties, drug-likeness, toxicity alerts, and medicinal chemistry-related parameters of the selected compounds, drug-likeness, ADMET was performed using swiss ADME (Yu and Adedoyin 2003).
2.13 In-Silico studies
To predict the possible targeting by ROT, Amd, and Benzoic-D5 to the D2 receptor, we carried out the in silico studies. For this purpose Auto-Dock vina software was used. The first step was to retrieve required ligand, and target pdb files from major databases. The second step was preparing PDBQT format files for the Target, and Ligand (Target-pdbqt, Ligand-pdbqt), Grid and Docking Parameter file (gpf and dpf). The third step was to perform molecular docking, finally the results were analyzed, by using the Sybyl X2.0. The energy of molecule is minimized, extracted the ligand in Mol-2 file, and dock on auto generated protomol by sybyl 2.0. The binding site was visualized by Discovery studio 2021 in both 2D, and 3D format (Athar et al., 2016).
2.14 Statistical analysis
All data were expressed as the Mean ± SEM. Analysis was performed Using GraphPad Prism software 7.0. The data were analyzed with One-way ANOVA followed by Tukey’s post hoc test. Statistical significance was defined as p < 0.05.
3 Results
3.1 Biochemical tests
3.1.1 Analysis of ALT, AST, Cholesterol, triglyceride and random glucose
The results showed the significant fluctuations of the parameters in the diseased groups, however post-treatment levels displayed normal symmetrical ranges, and similar to the standard group as shown in Table 1. Slight variation in the levels of these parameters can be the result of significant unwanted effects during PkD pathology.
Cytokines
Primer sequences
TnF-α
Forward
Reverse3′- GTCTACTCCTCAGAGCCC-5′
5′-TGAGATCCATGCCATTGGCC-3′
NFκB
Forward
Reverse5′- CAAGGAAGAGGATGTGGGGTT-3′
5′-AGCTGAGCATGAAGGTGGATG-3′
IL-4
Forward
Reverse5′- GGATGTAACGACAGCCCTCT-3′
5′-ACCGAGAACCCCAGACTTGT-3′
3.2 Analysis of WBC’s
The results showed the rise in the WBC levels in the diseased group B. After the treatment with Amd, and Benzoic-D5, there is significant improvement in the WBC levels (group C, D) as shown in Table 2. In diseased group due to the infiltration of neutrophils at the site of inflammation, their numbers were found augmented. Data showing the levels of ALT, AST, Triglyceride, cholesterol, glucose in Group A (Vehicle), Group B (ROT), Group C (Amd 60 mg/kd/day), Group D (Benzoic-D5 80 mg/kg/day). Statistical analysis was done through one-way analysis of variance (ANOVA) trailed by Tuckey’s post hoc test. All the groups were compared with diseased groups. The results are considered significant ∗ if p < 0.005. ∗∗p < 0.05, ∗∗∗p < 0.05. Results were compared in a column with the respective diseased groups.
Group
ALT
u/lAST
u/lTriglyceride
mg/dlCholesterol
mg/dlGlucose
Random mg/dl
A
50 ± 5 ****
70 ± 7****
150 ± 5***
100 ± 15***
113 ± 10**
B
166 ± 7.0
190 ± 15
109 ± 2
65 ± 12
135 ± 7
C
85 ± 7***
107 ± 7***
135 ± 4**
82 ± 3**
120 ± 6*
D
87 ± 5***
95 ± 3***
129 ± 4**
79 ± 3**
128 ± 7*
3.3 Behavioural tests
3.3.1 Wire hanging test
In wire hanging test efficacy of treatment protocols was assessed using neuromuscular, and sensory-motor function parameters. It was observed during experimentation that Group C showed more neuromuscular strength than Group D (p = 0.0119) as shown in Fig. 2A. Although, Group C demonstrate considerable improvement comparing to the Group B Group (0.0006), yet results of Group D being statistically more significant than the Group B (p = 0.0371) implies that Benzoic-D5 improved the neuromuscular strength of mice in wire hanging test as shown in Fig. 2A.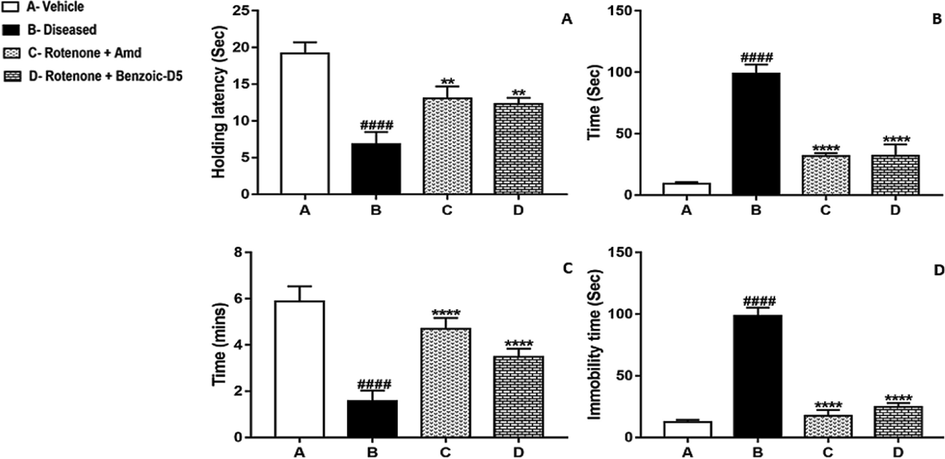
(A) Wire Hanging Test (B) Vertical rod test (C) Grip Strength Test (D) Swim test. The values are mean ± SEM (n = 7). Statistical analysis was done through one way analysis of variance (ANOVA) trailed by Tukey’s post hoc test for all groups. Statistical analysis was carried among all the groups, and showed in the figure. The results are considered significant *if p < 0.05. ** indicates p < 0.005 *** p < 0.001, and **** P < 0.0001 while # showing the non-significance.
3.3.2 Vertical rod test
PkD motor tests provide a good information of neurological functions, and/or dysfunctions of motor systems. During experimentation no significant differences were found between Group C, and Group D (p = 0.999), employing both have equal effects in mice with PkD, as shown in Fig. 2B. However, there is significantly higher difference between Group B, and Group C (p = 0.0001) as well as Group B and Group D (p = 0.0001). This test clearly demonstrate how close, Amd and Benzoic-D5 share the effects against the symptoms of PkD.
3.3.3 Grip strength test
Grip strength test showed no significant difference between Groups C, and D (p = 0.5027) showing effectiveness of Benzoic-D5 in improving grip strength in mice with PkD. Similarly significant difference between Groups B and C (p = 0.0003), Groups B and D (p = 0.0012) remains marginal, again employing a close effectiveness ability of Benzoic-D5 when comparing with Amd. This becomes more pronounced while noting significant difference between Group A, and Group D (p = 0.1762), and relating it with difference between Group A, and Group C (p = 0.8309), as shown in Fig. 2C.
3.3.4 Swim test
Effectiveness of Benzoic-D5 is evident by least significant value (p = 0.0282) as a comparison is made between Group C, and Group D. Also, considerably highest significant values of Group (P < 0.0001), and Group D (P < 0.0001) against Group B confirms the effectiveness of Benzoic-D5 in effectively ameliorating, the deteriorating impact of PkD as shown in Fig. 2D.
3.4 Oxidative stress markers
3.4.1 MDA Assay
Effects of repeated administration of Benzoic-D5 in group D for 5 weeks reduced lipid peroxidation and showed improvement in MDA levels compared to other groups as shown in Fig. 3A. Values are expressed as means ± SD (n = 7). Significant differences by Tukey’s test shows noteworthy (***) change compared to group B (####) indicates significance compared to standard treatment.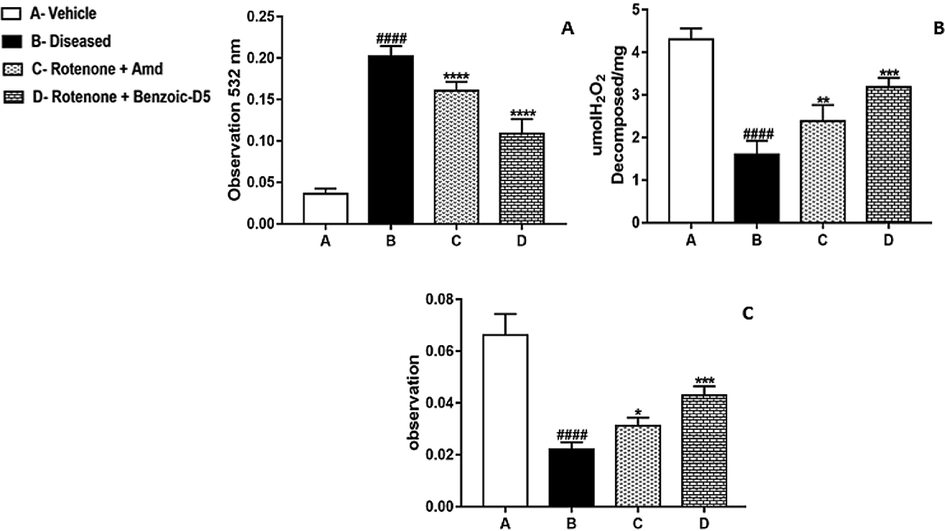
Shows results of (A) MDA, (B) Catalase, and (C) SOD assay, group A (Vehicle), ROT treated group B, (ROT + Amd), and (ROT + Benzoic-D5) group showed effects of repeated administration of Benzoic-D5 80 mg/kg/d for 5 weeks on MDA, catalase, and sodium oxide dismutase levels. Values are means ± SD (n = 7), F (6, 30) = 95.01 (P < 0.0001).
3.4.2 Catalase Assay
In the diseased conditions the catalase levels were significantly reduced in ROT group compared to Benzoic-D5 group and Amd treated groups. In this assay we found that Benzoic-D5 significantly reduced the oxidative stress caused by the rotenone, as shown in Fig. 3B.
3.4.3 SOD Assay
The SOD levels were significantly decreased in ROT group compared to Benzoic–D5 and Amd treated groups. Comparative analysis of different treated groups showed that Benzoic-D5 possesses the more antioxidant potential. It has the capability to avert the ROT induced oxidative stress in the PkD as shown in Fig. 3C.
3.5 Benzoic-D5 suppress mRNA expression levels of TNF-α, NFκB, and improve IL-4expression
See Fig. 4.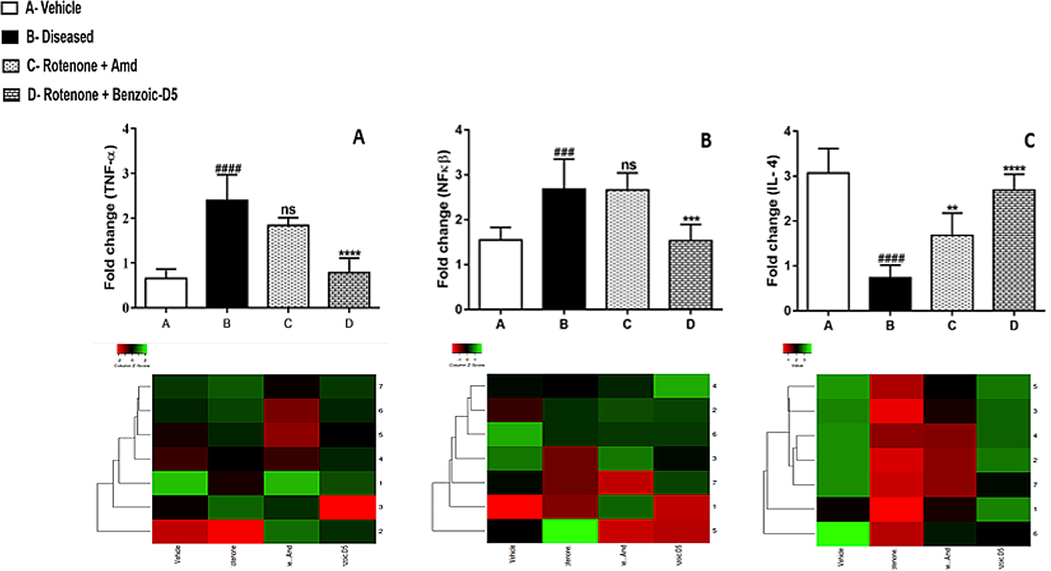
Graphical representation of mean ± SD relative expression levels of TNF-α, NFκB suppressed in group D. However, (Benzoic-D5) treatment causing improvement (****) in IL-4 expression as compared to the group B (####) which indicates significance of treatment in comparison with group B and C. Significantly raised (P < 0.0001) levels of TNF-α found in group B as compared to A, and treated C, and group D, (P = 0.0387) and F (P = 0.9961). Treatment with (Benzoic-D5) (0.5881 ± 0.2401; P < 0.01) considerably suppressed mRNA expression levels of TNF-α. It was also found that, significantly raised (P < 0.01) levels of NF-кB in B group (2.9340 ± 0.4545) as compared to group A (1.4502 ± 0.1285). Both group C and D showed considerably reduced NF-кB expression (2.3209 ± 0.1502) and (1.4998 ± 0.3941) (P < 0.05). Similarly, IL-4 decreased levels were observed in group B (0.7445 ± 0.3061) compared to group A (3.0597 ± 0.6022), while group D (2.776 ± 0.3030) shows significantly up regulation of IL-4 levels.
3.6 Analysis of dopamine, DOPAC and HVA
See Fig. 5.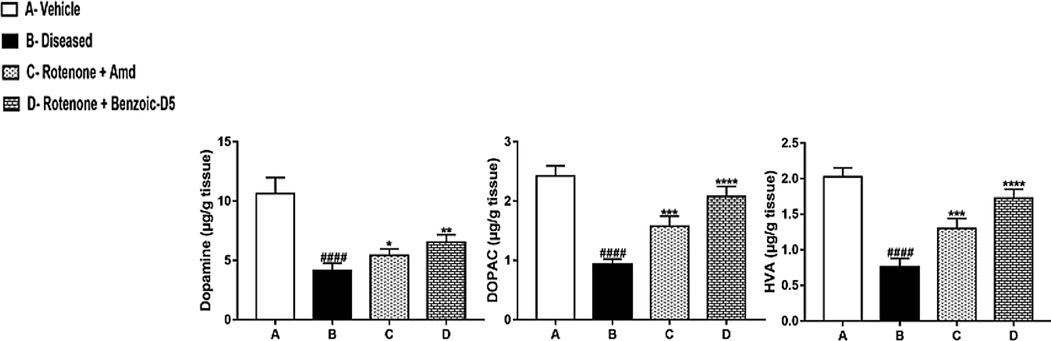
Shows the neurochemical study performed on the striatum tissue. ROT produced a significant (t = 2.54; df = 15; p < 0.05) decrease in the levels of DA of ROT treated group within the striatum, compared to the group A, group C, and D treated groups. However, it is observed the occurrence of a significant increase (t = 3.41; df = 15; p < 0.01) in the DA turnover of the ROT group in comparison to the group A, group C, and D treated groups. Still, considering the striatum, DOPAC turnover demonstrated to be increased (t = 2.72; df = 15; p < 0.01) in group D, compared to the Vehicle group. The analysis of striatum indicated that ROT provoked an increase of the HVA levels (t = 2.149; df = 15, p < 0.05) in Vehicle (t = 2.74; df = 15; p < 0.01), compared to the group D. Besides, HVA showed to be decreased (t = 3.4999; df = 15) and in the same way ROT treatment causes HVA turnover increase (t = 2.86; df = 15; p < 0.05).
3.6.1 Histopathological evaluation
Photomicrograph of (a)(normal, white arrow head) representing normal neurons. (b)(disease, black arrow head) showing Lewy bodies in numbers, (c)(Amd-treated, black arrow head) showed disturbed neurons but intact neurons were also present. (d)(Benzoic-D5 treated) showed normal neuronal cells (white arrow head but also the cells with Lewy body (black arrow head).
3.7 In-Silico studies
The results were analyzed graphically by visualizing protein using Auto-dock and Sybyl-X software. Fig. 7: represented the best docking model of D2 receptor. ROT, Amd and Benzoic-D5 displayed different binding site to the receptor as shown in Fig. 8. The ligand is docked into the binding cavity of the receptor and the putative conformations are explored. The docking results shows that almost all residues contributing towards D2 receptor favorable interactions with ROT, Amd and Benzoic-D5.
The Sybyl docking results were visualized using a discovery studio. 6 cm4 without ligands with encircle interacting area and the amantadine without receptor surface, Receptor pocket with amantadine shown in fig. Protein sequence of amino acid of 6 cm4 has shown in fig containing 4 amino acid chain named 6 cm4 A-1, A-2,A-3 and A-4 chain. The amantadine docked on 6 cm4 receptor with interacting amino acids, in visualization amantadine molecule convert into three hydrocarbon molecule but cycloalkane ring containing —NH2 group remain intact and shows interaction with receptor molecule. There are two types of interactions found, direct non–covalent interactions and supporting moieties. ALA-1093 has shown hydrogen interaction of 2.04A°. There are some van der walls forces also found with amino acid residue of LYS-221, ILE-1003, VAL-1075, TYR-1088, VAL-1094, ALA-1097, and ILE-1100 of A side chain of receptor. Similarly, residue of A side chain i.e. ARG-1093 carbon hydrogen bond interaction with second hydrogen of alkyl amino group and amantadine is cyclic alkyl moiety and has alkyl -alkyl interaction with ARG-220. The ROT docked on 6 cm4 receptor with interacting amino acids GLN-373, ARG-220, ILE-212, TYR-209, GLY-380, ILE-377, VAL-381, TYR-213, LEU-216 ALA-376 and ILE 384 of A side chain. There are two types of interactions found, direct non–covalent interactions and supporting moieties. GLN-373 has shown one hydrogen interaction of 1.97A° there are alkyl-alkyl - and π-π interaction also found with amino acid residue of ILE-384 AR G-220, ILE-212, TYR-209, GLY-380, ILE-377, VAL-381, TYR-213, LEU-216 and ALA-376 (3.88–4.88A) of side chain A of 6 cm4. The benzoic d-5 docked on 6 cm4 receptor with interacting amino acids ARG-217, ARG-220, ILE-1003, ASP-1072, PHE-1067, ASN-1068, LYS-221, VAL-1071 of A side chain. There are two types of interactions found, direct noncovalent interactions and supporting moieties. ARG-217 has shown two hydrogen interaction with both lone pair of oxygen of ligand, 1.88 and 2.07 A° there are π-π interaction also found with amino acid residue of LYS-221, VAL-1071 3.64 A° &4.54 A° and there are some van der walls forces also found with ARG-220, ILE-1003, ASP-1072,PHE-1067,ASN-1068 with side chain A of 6 cm4.
The results of binding energies of ROT Amd and Benzoic-D5 are shown in Fig. 6, supporting information, respectively. The results shows very less significant differences among the groups Root mean square distances (RMSD) showed very less significant differences however in the different ranks and sub ranks the value is varied that is shown in the Fig. 9B. RMSD values should be less than 2 Angstrom.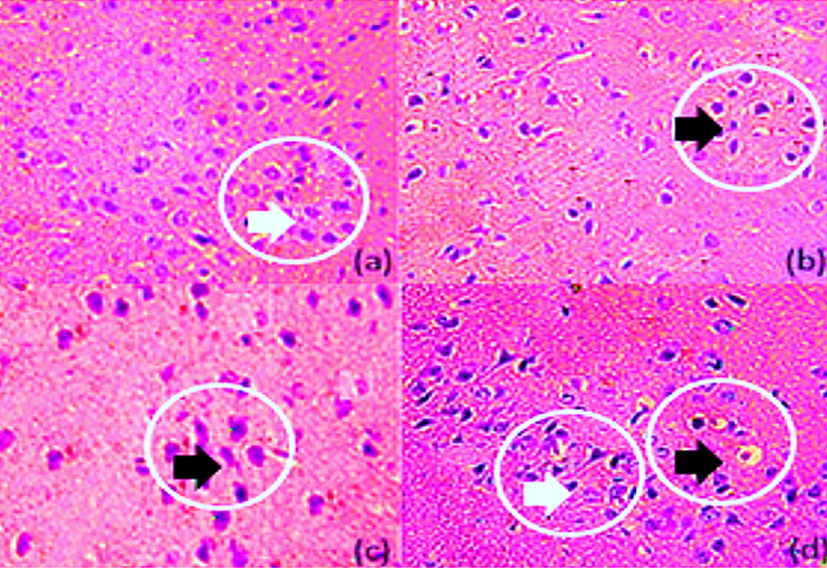
Histopathology of mice brain (Substentia Nigra). (a) Normal (b) Diseased (c) Amd-treated (d) Benzoic-D5 treated.
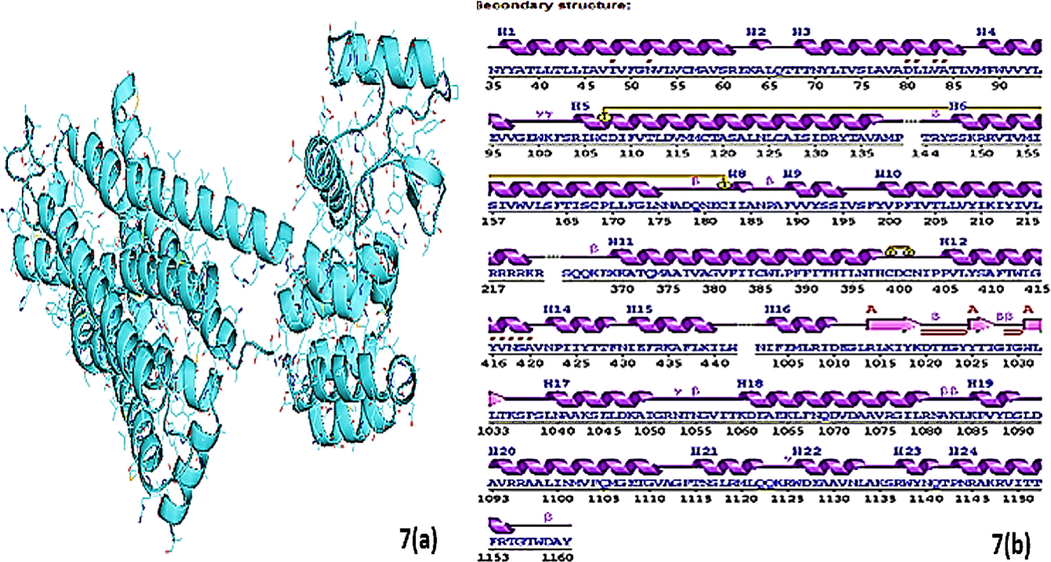
Fig. 7 containing (a and b) sections showing (a) the crystal structure of D2 receptor having PDB id (6 cm4) and (b) the secondary structure of the receptor and position and length of receptor chain.
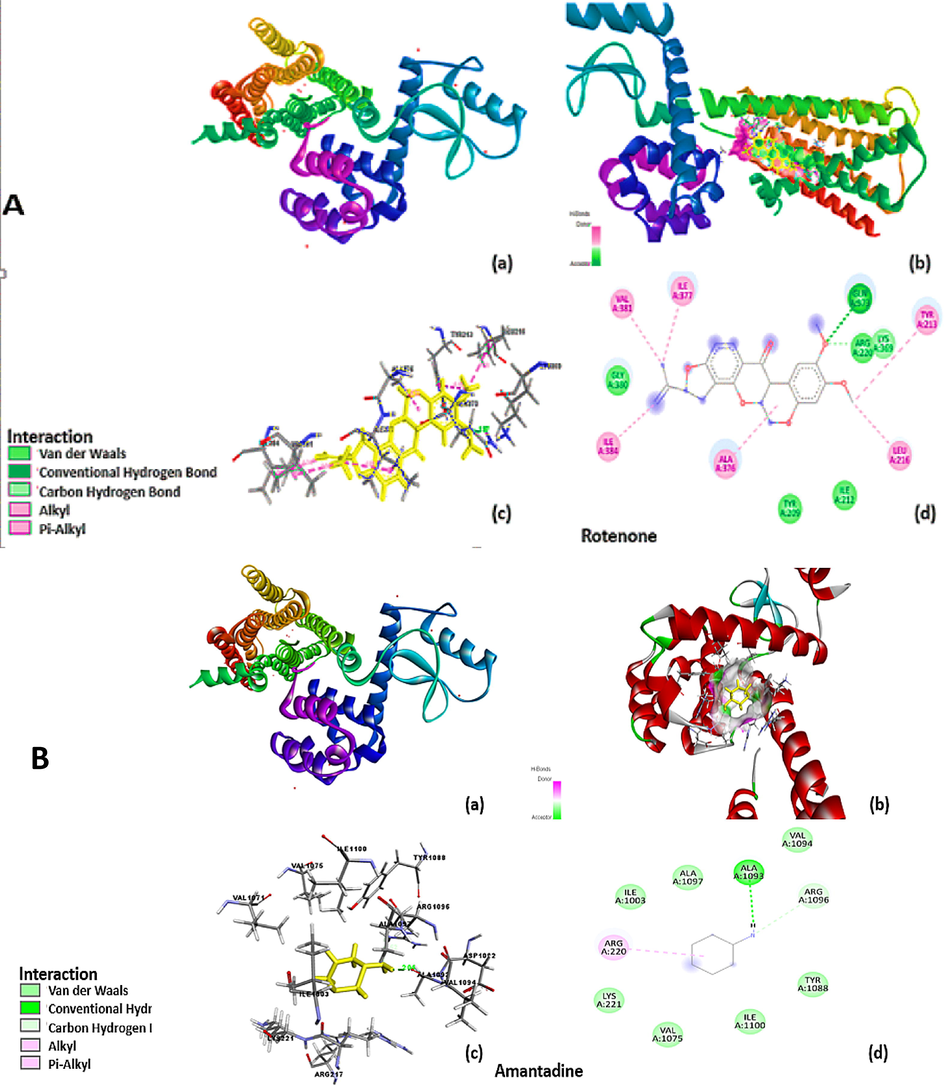
Showing superimposition of the docked ligand (A) ROT (B) Amd and (C) Benzoic-D5 in the main active site of the D2 receptor. The ligand was docked into the binding cavity of the receptor and the putative conformations are explored.
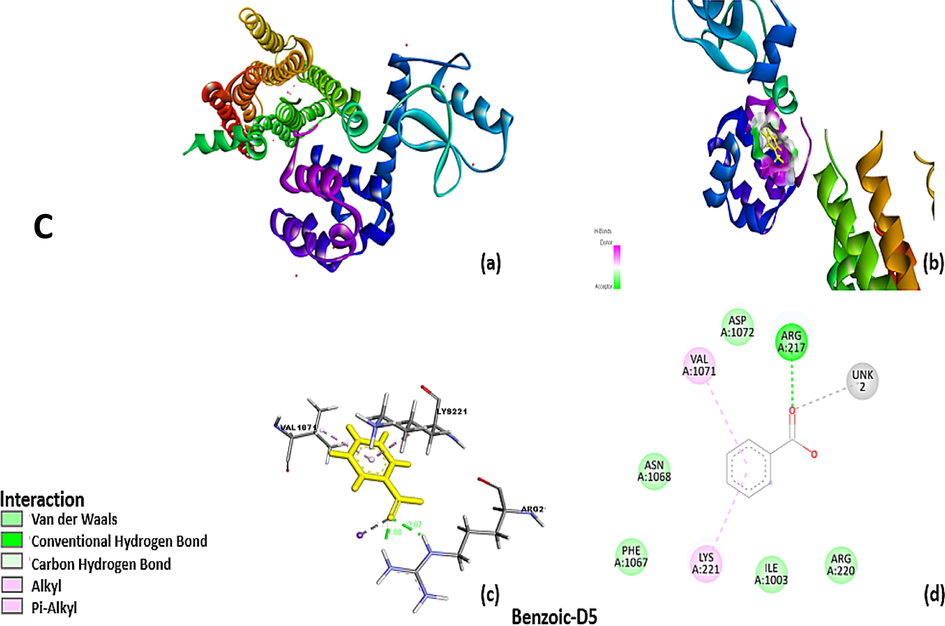
Showing superimposition of the docked ligand (A) ROT (B) Amd and (C) Benzoic-D5 in the main active site of the D2 receptor. The ligand was docked into the binding cavity of the receptor and the putative conformations are explored.
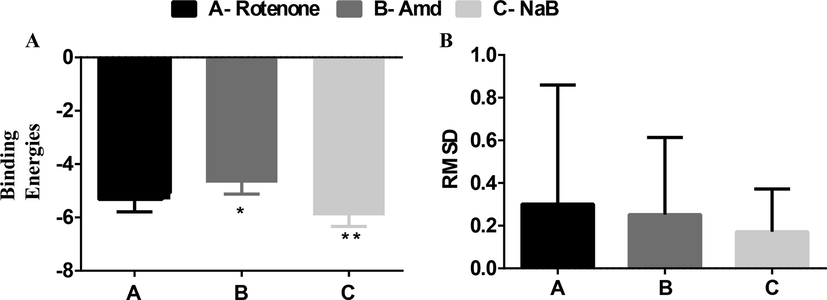
(A) shows the binding energy of ligand (ROT, Amd, and Benzoic-D5) on D2 receptor (B) shows the RMSD values of the rotenone, Amd, and Benzoic-D5.
3.8 Pharmacokinetics and toxicity risk assessment
(See Fig. 10).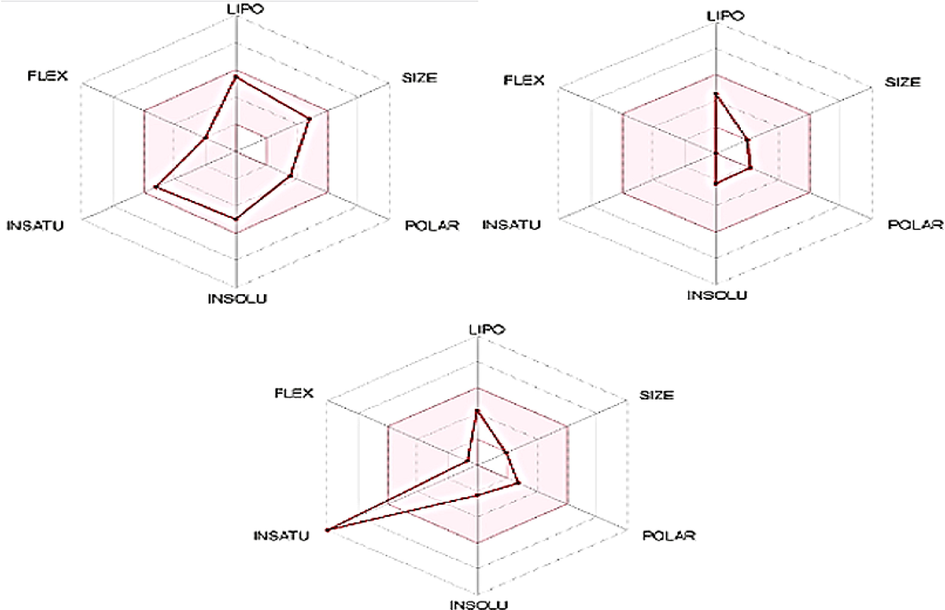
Summarized illustration of ADMET profile of selected drugs; ROT, Amantadine, and Benzoic-D5. The pink colored area is illustrating the acceptable range of drugs such as lipophilicity = XLOGP3 (Range: should be between ne-0.7 and + 5.0, Size = molecular weight (Range: 150 and 500 g/mol) polarity = Total polar surface area (Range: should be between 20 and 130 Å), Solubility = LogS (Range: should not be higher than 6), Saturation = fractions of carbon atoms in sp3 hybridization (Range: should not be<0.25) and flexibility (Range: no more than or equal to 10 rotatable bonds).
3.9 Pharmacokinetic and drug-likeness profile of drugs
(See Table 3). Data showing the levels of WBCs Group A (Vehiclel), Group B (ROT), Group C (ROT + Amd 60 mg/kg/day), Group D (ROT + Benzoic-D5-80 mg/kg/day). Statistical analysis was done through one-way analysis of variance (ANOVA) trailed by Tuckey’s post hoc test for WBC. All the groups were compared with diseased groups. The results are considered significant ∗ if p < 0.005. ∗∗p < 0.05, ∗∗∗p < 0.05. Results were compared in a column with the respective diseased groups.
Group
WBC (109/ L)
Neutrophils (%)
Lymphocytes (%)
Monocytes (%)
Eosinophil (%)
A
13 ± 2.0 **
75 ± 3
20 ± 5
3 ± 1
3 ± 1
B
18 ± 2.0
90 ± 5
5 ± 2
3 ± 1
3 ± 1
C
12 ± 0.8**
75 ± 3
10 ± 3
5 ± 1
5 ± 1
D
12 ± 0.5*
79 ± 3
12 ± 3
6 ± 1
6 ± 1
4 Discussion
Parkinson's disease is a neurodegenerative disorder that affects movement and is characterized by the death of dopamine-producing neurons in the brain. ROT is a pesticide that is known to induce PkD in animal models by inhibiting mitochondrial complex I activity, leading to oxidative stress and neuronal death. This study was executed to investigate the anti-Parkinson potential of the Benzoic-D5. PkD was induced by ROT. Chronic administration of the ROT in mice significantly increased VCM and TDK, the hallmark of the PkD (Khairnar et al., 2021). ROT is a pesticide that is believed to cause severe oxidative stress at the cellular level by inhibiting the mitochondrial respiratory chain complex I. This inhibition leads to a decrease in ATP production and an increase in the production of reactive oxygen species (ROS) (Rekuviene et al., 2017). The increased levels of ROS can cause damage to cellular components, including proteins, lipids, and DNA. In the nigro-striatal region of the brain, this oxidative stress can lead to the degeneration of dopaminergic neurons, which contributes to the development of PKD, Benzoic-D5 causes decrease in level of ROS and thus improves the neuro-muscular performance of the animal as compared to previous study on bee venom (Bava et al., 2023).
There is some evidence to suggest that reduced levels of dopamine can contribute to the initiation of lipid peroxidation and the production of excessive oxidative stress. Dopamine is a neurotransmitter that plays a key role in the brain's reward and pleasure systems, as well as in motor control. Lipid peroxidation is a process that occurs when free radicals react with unsaturated fatty acids in cell membranes, leading to the formation of reactive lipid peroxides. This process can cause damage to cell membranes and other cellular components, and has been implicated in a number of disease processes. Reduced levels of dopamine is one of a triggering factor for initiation of lipid peroxidation that produce excessive oxidative stress (Motawi et al., 2020).
In this study, Benzoic-D5 has shown promising results in improving both behavioral and biochemical changes as well as reversing oxidative stress in a ROT-induced PkD model. Additionally, it appears that the use of Benzoic-D5 has led to a normalization of liver, kidney, and blood markers, which may indicate an improvement in pathological changes as shown in (Tables 1 and 2). Lipid peroxidation is a process in which ROS attack and damage the lipids in cell membranes, resulting in cell damage and death. In PkD, lipid peroxidation is thought to be leading cause of death of dopaminerigic neurons. By preventing lipid peroxidation, Benzoic-D5 may be able to slow the progression of PkD by decreasing the levels of MDA and lipid peroxidation, compared to study performed on europindin (Altharawi et al., 2022).
Fig. 2, suggests behavioral improvement brought about by Benzoic-D5 in animals with PkD and is thought to be due to prevention of this lipid peroxidation by the otherwise generated ROS. Consequently, in absence of peroxidation-cum oxidative stress, neuronal functioning improves significantly. In literature Benzoic-D5 has been cited as have ability to up regulate DJ-1 and Parkin proteins, underscoring its neuroprotective functions by other mechanisms too (Angelopoulou et al., 2021). This is what we have demonstrated in this study. Wire hanging test (Fig. 2A) shows deteriorating effects of ROT in diseased animals and productive outcomes of Benzoic-D5 in treatment groups. Animals treated with Amd showed relatively more neuromuscular strength than the animal group treated with Benzoic-D5. However, significant difference of (p = 0.0119) underlies important anti-Parkinson effects of Benzoic-D5 in mice. Although both Amd and Benzoic-D5 improved neuromuscular strength and were found relatively effective in improvement of muscle functions and coordination. Grip strength test is another modest non-intrusive method intended to measure mouse muscle power in vivo (Saeed et al., 2017).
In PKD, as a result of muscle weakness and dyskinesia, the grip strength of the mice is greatly affected. PKD causes muscle weakness and dyskinesia (involuntary movements), they can occur as a result of the disease, which greatly affect grip strength. Grip strength test is a commonly used measure of muscular strength in mice and rodents, and it can be used to assess the effects of PKD on muscle function. Grip strength is greatly affected in mice with PKD, could be due to a variety of factors, such as inflammation which is caused by oxidative stress (Zaidi et al., 2016). Thus, this behavioral test was utilized to measure animal's affinity to grip a flat metal bar while suspended by its tail. Grip strength remained same for the animals treated with Amd as well as Benzoic-D5 as underlined by the results (p = 0.0527) (Fig. 2B). Also, when statistical values of Amd treated group (p = 0.0003) and Benzoic-D5 treated group (p = 0.0012) were compared against diseased group, it becomes evident that Benzoic-D5 is positioned second to Amd in proving to be effective in reverting dyskinaic effects of PkD This test gives insight into level of catalepsy produced by ROT in mice model of PkD and its positive effects on mice during and after treatment with Amd and Benzoic-D5 in mice models. It was noted during study that Group C presented improved neuro-logical functions than Group B (p < 0.0001) (Fig. 2C). While relating our data of vertical rod test with the outcomes of the study, it becomes evident that the Benzoic-D5-80 mg/kg/day (Group D) provides almost the same results as provided by Amd 60 mg/kg/day.
Swim test was used to examine motor damage and measurement of motor weakness in animals with PKD (Costall and Naylor 1974), as it is considered as an effective tool to assess the extent of effectiveness of anti-depressant medications, upper viability of new drugs, and control trials that are used for blocking or counteracting depressive-like states. In our study Benzoic-D5 80 mg/kg/day demonstrated considerably good effects in comparison with group C treated with Amd 60 mg/kg/day, employing the effective potentials of the latter in diminishing the deteriorating effects of PkD (p ≤ 0.055) (Fig. 2D).
ROT is a potent inhibitor of mitochondrial complex I, which is a crucial enzyme involved in the electron transport chain that generates energy in the form of ATP in cells. By inhibiting complex I, ROT disrupts the normal functioning of the mitochondria, leading to an increase in the production of ROS and oxidative stress. The brain is particularly susceptible to oxidative stress because it has high energy requirements and a high content of polyunsaturated fatty acids, which are vulnerable to lipid peroxidation induced by ROS. Oxidative stress can damage proteins, lipids, and DNA in the brain, leading to neuronal dysfunction and death, and has been implicated in the pathogenesis of neurodegenerative diseases such as PKD. The administration of ROT leads to an increase in oxidative stress in brain. The present investigation suggests that levels of MDA, SOD, and CAT and were decreased in Benzoic-D5 treated group compared to disease group that was in accordance with previous data (Zaidi et al., 2020). Brain defends itself via catalase, and SOD against oxidative stress which leads to neurodegeneration. Glutathione has ability to scavenge H2O2 produced via cellular metabolism. Nitric oxide generation can be measured via nitrite concentration in biological materials. Free radicals, generated throughout stress target membrane lipid which acts on oxygen to produce per-oxy radicals leading to formation of MDA. SOD ameliorates the glutathione levels that develops the relationship between oxidative stress caused by free radical (Abid et al., 2021).
Like Amd the Benzoic-D5 has got the potential to reverse the oxidative stress that was produced due to the disease progression. The substantia nigra is a region of the brain that is involved in movement control, and dysfunction in this area has been implicated in PKD. Oxidative stress, which is an imbalance between the production of ROS and the body's antioxidant defenses, has also been linked to PKD and other neurological disorders. It is possible that the relationship between the improvement of behavioral testing, reduction in oxidative stress, and magnitude of cell death in the substantia nigra of mice brain may be related to the effects of an experimental treatment or intervention aimed at reducing oxidative stress and/or cell death in this region. There was a significant relationship between the between the improvement of behavioral; testing, reduction in oxidative stress and magnitude of cell lose in substantia nigra of mice brain (Nascimento-Ferreira et al., 2013).
ROT is a pesticide that has been shown to induce PKD-like symptoms in animal models by damaging dopaminergic neurons in the brain. Dopamine is a neurotransmitter that is produced by dopaminergic neurons and plays a crucial role in reward-motivated behavior, movement, and mood regulation. DOPAC and HVA are metabolites of dopamine that are produced when dopamine is broken down. Therefore, decreased levels of DOPAC and HVA suggest that there is less dopamine being broken down in the brain, which could be due to a decrease in the number of dopaminergic neurons or a decrease in dopamine release. The decrease in dopamine, DOPAC, and HVA levels in the ROT-treated group suggests that the pesticide has damaged dopaminergic neurons in the brain, resulting in a decrease in dopamine synthesis and release. This is consistent with the neurodegenerative effects of ROT, and it is believed that this mechanism plays a role in the development of PKD. The HPLC results of this study showed improved dopamine, DOPAC and HVA levels in Benzoic-D5 treated group compared to Amd and diseased group (Patil et al., 2021). Treatment with Benzoic-D5 significantly restored the diminished levels of dopamine.
Histopathological findings exhibited that Benzoic-D5 had decreased vacuolation, pigmentation, neutrophils infiltration and shrinkage of neurons as compared to disease group supported by earlier studies (Wu et al., 2021).
Two pro-inflammatory cytokines including tumor necrosis factor-α (TNF-α) and NF-Kβ have been associated with functional consequences of neuro-inflammation leading to neuro-degeneration in PkD models. TNF-α is a pro-inflammatory cytokine that is released by activated microglia, astrocytes, and other immune cells in the brain in response to injury or infection. TNF-α has been shown to be elevated in the brains of patients with PKD and in animal models of the disease, and it is believed to contribute to the neuro-inflammatory response that is characteristic of PKD. NF-κB is a transcription factor that regulates the expression of a wide range of genes involved in inflammation, immune response, and cell survival. NF-κB activation has been observed in both animal models of PKD and in human post-mortem brain tissue from PKD patients, and it is believed to play a role in the neuro-inflammatory response and the death of dopaminergic neurons that is seen in PKD. Together, TNF-α, and NF-κB can activate a cascade of events that contribute to the development and progression of PKD, including oxidative stress, mitochondrial dysfunction, and the activation of other inflammatory pathways. Targeting these pathways has emerged as a potential therapeutic strategy for PKD, with several drugs currently in development that aim to inhibit the neuro-inflammatory response and prevent or slow the progression of the disease (Ndayisaba et al., 2019). Overexpression of TNF-α and NF-Kβ were evident from the present study which was in accordance with previous literature (Guzman-Martinez et al., 2019). However significant decrease in expression of TNF-α and NF-Kβ and improved expression of IL-4, has been observed compared to standard treatment and vehicle treated group. Furthermore, the ADMET profile of selected drugs and Benzoic-D5 was estimated to offer the safe and highly efficacious drug for PkD (Table 4), Benzoic-D5 are natural antioxidants and highly significant for the prevention and treatment of other metabolic diseases also, their biological activities and their bioavailability profile has been discussed in the previous literature (Wang et al., 2020).
Descriptor
Rotenone
Amantadine
Benzoic-D5
Smiles Notion
CC(⚌C)C1CC2⚌C(O1)C⚌CC3⚌C2OC4COC5⚌CC(=C(C⚌C5C4C3⚌O)OC)
C1C2CC3CC1CC(C2)(C3)N
C1⚌CC⚌C(C⚌C1)C(⚌O)O
Formula
C23H22O6
C10H17N
C7H6O2
Molecular weight (MW) (g/mol)
394.4
151.25
122.12
Number of heavy atoms
29
1
9
Number of aromatic Heavy atoms
Number of rotatable bonds
3
0
1
Number of hydrogen bond acceptors (HBA)
6
1
2
Number of hydrogen bond donors (HBD)
0
1
1
Molar Refractivity (MR)
105.71 m3·mol−1
45.54 m3·mol−1
44.15 m3·mol−1
Total polar surface area (TPSA) (Å)
63.22
26.02
40.130
Lipophilicity
LogPo/w (iLOGP)
3.84
2.18
−11.15
LogPo/w (XLOGP3)
4.10
2.44
1.87
LogPo/w (WLOGP)
3.70
1.91
0.05
LogPo/w (MLOGP)
1.93
2.45
1.60
LogPo/w (SILICOS-IT)
4.93
1.91
1.24
Consensus Log Po/w
3.59
2.18
−1.28
Descriptor
Rotenone
Amantadine
Benzoic-D5
Water Solubility
LogS (ESOL)
−4.98
−2.31
−2.29
Solubility (mg/ml)
4.16e-03; 1.06e-1.05 mol/1
7.32e-01; 4.84e-03 mol/1
7.40e-01; 5.13e-03 mol/1
Class
Moderately soluble
Soluble
Soluble
LogS (Ali)
−5.13
−2.63
−2.33
Solubility (mg/ml)
2.90e-03; 7.36e-06 mol/1
3.55e-01; 2.35e-01 mol/1
6.67e-01; 4.63e-03 mol/1
Class
Moderately soluble
Soluble
Soluble
LogS (SILICOS-IT)
−6.00
−1.57
−1.72
Solubility (mg/ml)
3.93e-04; 9.95e-07 mol/1
4.08e + 00; 2.70e-02 mol/1
2.72e + 00; 1.89e-02 mol/1
Class
Poorly soluble
Soluble
Soluble
Distribution
PPB
97.77
79.70
79.702%
BBB Penetration
0.0094
0.608
0.969
Pharmacokinetics
GI absorption
High
High
High
BBB permeant
Yes
Yes
No
P-gp substrate
No
No
Yes
CYP1A2 inhibitor
No
No
No
CYP2C19 Inhibitor
Yes
No
No
CYP2C9 inhibitor
Yes
No
No
CYP2D6 inhibitor
Yes
No
No
CYP3A4 inhibitor
Yes
No
No
Log Kp (skin permeation) (cm/s)
−5.79
−5.49
−5.85
Descriptor
Rotenone
Amantadine
Benzoic-D5
Drug-likeness
Lipinski
Yes: 0 Violation
Yes: 0 Violation
Yes: 0 Violation
Ghose
Yes
No:1 Violation MW<160,
No:3 Violation MW<160,MR<40,# atoms< 20
Veber
Yes
Yes
Yes
Egan
Yes
Yes
Yes
Muegge
Yes
No:2 Violation MW<160,
Heteroatmos <2No:1 Violation MW<200
Bioavailability Score
0.55
0.55
0.55
Medicinal Chemistry
SA score
3.868
3.899
2.215
NP score
1.842
0.553
−0.467
PAINS
No alert
No alert
No alert
Brenk
1 alert
Isolated alkeneNo alert
No alert
Lead likeness
2 Violation MW>350,
XLOGP3> 3.5No:1 Violation MW<250
No:1 Violation MW<250,
Synthetic accessibility
4.52
3.49
1
Toxicophore Rules
Acute Toxicity Rule
0 Alert
0 Alert
0 Alert
Genotoxic Carcinogenicity Rule
0 Alert
0 Alert
0 Alert
NonGenotoxic Carcinogenicity Rule
0 Alert
0 Alert
0 Alert
Skin Sensitization Rule
3 Alert
0 Alert
0 Alert
Aquatic Toxicity Rule
0 Alert
1 Alert
0 Alert
Non-Biodegradable Rule
0 Alert
0 Alert
0 Alert
SureChEMBL Rule
0 Alert
0 Alert
0 Alert
FAF-Drugs4 Rule
0 Alert
0 Alert
0 Alert
Molecular docking of ROT, Amd and Benzoic-D5 to the active site of D2 receptor was performed. Docking pose for proteins was saved for each complex and graphically viewed to identify the ligand–protein interactions. All ligands acquired similar mode of interaction within the active site of the receptor, where they can make similar H-bond interactions with surrounding residues (Jain 2003).
Binding energies and RMSD values also showed the possible interaction of these ligands with the D2 receptor. Although docking analysis can provide an acceptable binding mode, the solvent, temperature and pressure effects could not be taken considered (Fogolari et al., 2003). Therefore, docking results were post processed by more reliable molecular dynamics simulations, to describe the dynamical behavior of the complex at atomic-level by flexibly and firmly treating ligand-receptor complex. Overall, these analyses for binding stabilization further reinforce the accuracy of molecular docking and experimental results. Further research on pharmaco-kinetics and pharmacodynamics of Benzoic-D5 can help to identify the possible mechanism of action.
Molecular infrastructures are important to understand oral bioavailability of drugs, and have been retrieved by the application of In silico methods in this study, following descriptors correlated in vitro and in vivo absorption of available drugs datasets with their molecules structures by physicochemical descriptors to estimate the absorption of similar molecules (Alqahtani 2017). Physiochemical descriptors calculated for the selected drugs in study are in acceptable range such as molecular weight, lipophilicity, number of rotatable bonds, number of hydrogen bond acceptors and donors, and total polar surface area is the highly significant parameters to predict the information of drug adsorption and drug-likeness (Lipinski et al., 1997). In silico models have been offered to estimate the bioavailability, toxicity, and absorption of selected drugs by SwissADME web tool and Osiris property explorer shown in Table 5: Amd and Benzoic-D5 have presented no PAINS and Brenk undesirable medicinal alerts, drug-likeness and drug score is presenting the cumulative properties of selected drugs, Amd and Benzoic-D5 have presented a very good drug score calculated on the basis of drug molecule information The designed systematic analysis has been focused to explain the importance of Benzoic-D5 as therapeutic options for the treatment and prevention of PkD, which is a severe abnormality of the brain progressing towards the multiple other diseases and may lead towards the restriction of muscular mobility of patients. Experimental and computational techniques were applied in this study to produce results, it is noteworthy the selected phytochemicals have presented significant inhibitory activity against acetylcholinesterase which is an active target against PkD.
Behavioral tests
ROT treated
Amd treated
Benzoic-D5 treated
1
Wire hanging test
↓↓↓
↑↑
↑↑↑
2
Vertical pole test
↓↓↓
↑
↑↑↑
3
Forced swim test
↓↓↓
↑↑
↑↑↑
Anti-oxidant test
1
MDA Assay
↓↓↓
↑
↑↑↑
2
Catalase Assay
↓↓↓
↑
↑↑↑
3
SOD Assay
↓↓↓
↑
↑↑↑
Pro-Inflammatory cytokines expression
1
TnF-α
↑↑↑
↓↓
↓↓↓
2
NFκB
↑↑↑
↓↓
↓↓↓
Anti-Inflammatory cytokines expression
1
IL-4
↓↓↓
↑↑
↑↑↑
Neurotransmitter levels
1
Dopamine
↓↓↓
↑↑
↑↑↑
2
DOPAC
↓↓↓
↑↑
↑↑↑
3
HVA
↓↓↓
↑↑
↑↑↑
Biochemical tests
1
ALT
↑↑↑
↓↓
↓↓
2
AST,
↑↑↑
↓↓
↓↓↓
3
Cholesterol,
↓↓↓
↑
↑↑
4
triglyceride
↓
↓↓
↓↓
5
random glucose
↓
↓
↑
Analysis of WBC’s
WBC (109/ L)
↑↑↑
↑↑
↑
2
Neutrophils (%)
↑↑↑
↑
↑
3
Lymphocytes (%)
↑
↑
↑
4
Monocytes (%)
↓
↓↓
↓↓
5
Eosinophil (%)
↓
↓↓
↓↓
In-Silico Studies
1
Binding energy
↓
↑
↑↑
2
RMSD values
↑↑↑
↑↑
↑
5 Conclusion
From the current systematic investigation, it is concluded that Benzoic-D5 has an antioxidant against ROS, and neuro-protective effect against ROT induced PkD. Benzoic-D5 could significantly improve the neuro-muscular parameters, biochemical and histopathological alterations. Furthermore, molecular docking studies and ADMET profile estimation, qRT-PCR analysis supported the use of Benzoic-D5 in PkD and offer new insight for the development of novel drug candidates against PkD, which could be harmonious to the human body.
Acknowledgement
The authors would like to acknowledge Deanship of Scientific Research, Taif University, Taif , Saudi Arabia for supporting this work. Also we would like to thank for the work that had been done by the lab persons and M.Phil-scholars, Arsalan Ahmad, Muhammad Yousaf and Atif Saeed.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Acacia modesta attenuates MnCl2 induced hepatotoxicity, oxidative stress and hepatic inflammation in wistar rats. Pak. J. Pharm. Sci.. 2021;34:265-274.
- [Google Scholar]
- In silico ADME-Tox modeling: progress and prospects. Expert Opin. Drug Metab. Toxicol.. 2017;13:1147-1158.
- [Google Scholar]
- Europinidin Inhibits Rotenone-Activated Parkinson’s Disease in Rodents by Decreasing Lipid Peroxidation and Inflammatory Cytokines Pathways. Molecules. 2022;27:7159.
- [Google Scholar]
- Neuroprotective potential of cinnamon and its metabolites in Parkinson's disease: Mechanistic insights, limitations, and novel therapeutic opportunities. J. Biochem. Mol. Toxicol.. 2021;35:e22720.
- [Google Scholar]
- Pharmacophore model prediction, 3D-QSAR and molecular docking studies on vinyl sulfones targeting Nrf2-mediated gene transcription intended for anti-Parkinson drug design. J. Biomol. Struct. Dyn.. 2016;34:1282-1297.
- [Google Scholar]
- Prevalence and clinical aspects of mild cognitive impairment in Parkinson's disease: a meta-analysis. Mov. Disord.. 2020;35:45-54.
- [Google Scholar]
- Liquid chromatography in the analysis of neurotransmitters and alkaloids. J. Chromatogr. Sci.. 2002;40:224-230.
- [Google Scholar]
- Therapeutic Use of Bee venom and potential applications in veterinary medicine. Vet. Sci.. 2023;10:119.
- [Google Scholar]
- Anti-Parkinson activity of petroleum ether extract of Ficus religiosa (L.) leaves. Adv. Pharmacol. Sci. 2016
- [Google Scholar]
- Neurotoxicity of the pesticide rotenone on neuronal polarization: a mechanistic approach. Neural Regen. Res.. 2019;14:762.
- [Google Scholar]
- A highly reproducible rotenone model of Parkinson's disease. Neurobiol. Dis.. 2009;34:279-290.
- [Google Scholar]
- Mesolimbic involvement with behavioural effects indicating antipsychotic activity. Eur. J. Pharmacol.. 1974;27:46-58.
- [Google Scholar]
- Chronic exposure to rotenone models sporadic Parkinson's disease in Drosophila melanogaster. J. Neurosci.. 2004;24:10993-10998.
- [Google Scholar]
- Survival after treatment with phenylacetate and benzoate for urea-cycle disorders. N. Engl. J. Med.. 2007;356:2282-2292.
- [Google Scholar]
- Protocol for MM/PBSA molecular dynamics simulations of proteins. Biophys. J .. 2003;85:159-166.
- [Google Scholar]
- A simple method for determination of serum catalase activity and revision of reference range. Clin. Chim. Acta. 1991;196:143-151.
- [Google Scholar]
- Neuroinflammation as a common feature of neurodegenerative disorders. Front. Pharmacol.. 2019;10:1008.
- [Google Scholar]
- Mechanistic investigations of the mitochondrial complex I inhibitor rotenone in the context of pharmacological and safety evaluation. Sci. Rep.. 2017;7:1-13.
- [Google Scholar]
- A Modified wire hanging apparatus for small animal muscle function testing. PLoS Curr.. 2016;8
- [CrossRef] [Google Scholar]
- Huh, L. and K. Farrell, 2011. Urea cycle disorders. The Causes of Epilepsy: Common and Uncommon Causes in Adults and Children. 246.
- Using rotenone to model Parkinson’s disease in mice: a review of the role of pharmacokinetics. Chem. Res. Toxicol. 2021
- [Google Scholar]
- Surflex: fully automatic flexible molecular docking using a molecular similarity-based search engine. J. Med. Chem.. 2003;46:499-511.
- [Google Scholar]
- Neuroprotective effect of nerolidol against neuroinflammation and oxidative stress induced by rotenone. BMC Neurosci.. 2016;17:1-12.
- [Google Scholar]
- Rapid determination of dopamine and its metabolites during in vivo cerebral microdialysis by routine high performance liquid chromatography with electrochemical detection. Biomed. Environ. Sci.. 2007;20:317.
- [Google Scholar]
- DNA damage in brain cells and behavioral deficits in mice after treatment with high doses of amantadine. J. Appl. Toxicol.. 2010;30:745-753.
- [Google Scholar]
- Kalpana, V., Rajeswari, V.D., 2019. Preservatives in beverages: Perception and needs. Preservatives and preservation approaches in beverages, Elsevier: 1-30.
- Revisiting the validity of the mouse forced swim test: Systematic review and meta-analysis of the effects of prototypic antidepressants. Neurosci. Biobehav. Rev.. 2018;84:1-11.
- [Google Scholar]
- Diffusion kurtosis imaging detects the time-dependent progress of pathological changes in the oral rotenone mouse model of Parkinson's disease. J. Neurochem. 2021
- [Google Scholar]
- Vertical grid test and modified horizontal grid test are sensitive methods for evaluating motor dysfunctions in the MPTP mouse model of Parkinson's disease. Brain Res.. 2010;1306:176-183.
- [Google Scholar]
- Dose-dependent pharmacokinetics of benzoic acid following oral administration of sodium benzoate to humans. Eur. J. Clin. Pharmacol.. 1991;41:363-368.
- [Google Scholar]
- Validation of suitable reference genes for RT-qPCR data in Achyranthes bidentata Blume under different experimental conditions. Front. Plant Sci.. 2017;8:776.
- [Google Scholar]
- Food additives and their health effects: A review on preservative sodium benzoate. Afr. J. Biotechnol.. 2018;17:306-310.
- [Google Scholar]
- Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev.. 1997;23:3-25.
- [Google Scholar]
- Estimate levels of the liver enzymes and serotonin immune localization in the brain of rats administrated orally with sodium benzoateInternational Journal of. Pharm. Res.. 2020;12
- [Google Scholar]
- Potential therapeutic effects of antagonizing adenosine A 2A receptor, curcumin and niacin in rotenone-induced Parkinson’s disease mice model. Mol. Cell. Biochem.. 2020;465:89-102.
- [Google Scholar]
- Increasing levels of the endocannabinoid 2-AG is neuroprotective in the 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine mouse model of Parkinson's disease. Exp. Neurol.. 2015;273:36-44.
- [Google Scholar]
- Evaluating ADS5102 (amantadine) for the treatment of Parkinson’s disease patients with dyskinesia. Expert Opin. Pharmacother.. 2019;20:1181-1187.
- [Google Scholar]
- Beclin 1 mitigates motor and neuropathological deficits in genetic mouse models of Machado-Joseph disease. Brain. 2013;136:2173-2188.
- [Google Scholar]
- TNFα inhibitors as targets for protective therapies in MSA: a viewpoint. J. Neuroinflammation. 2019;16:1-7.
- [Google Scholar]
- Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem.. 1979;95:351-358.
- [Google Scholar]
- Progression of Parkinson’s disease pathology is reproduced by intragastric administration of rotenone in mice. Nat. Precedings. 2010:1.
- [Google Scholar]
- Evaluation of Antidepressant activity of Ficus carica leaves extract in experimental animals. Res. J. Pharm. Technol.. 2021;14:1267-1273.
- [Google Scholar]
- Behavioral Tests in Neurotoxin-Induced Animal Models of Parkinson’s Disease. Antioxidants.. 2020;9:1007.
- [Google Scholar]
- Rotenone decreases ischemia-induced injury by inhibiting mitochondrial permeability transition in mature brains. Neurosci. Lett.. 2017;653:45-50.
- [Google Scholar]
- Haloperidol induced Parkinson’s disease mice model and motor-function modulation with Pyridine-3-carboxylic acid. Biomedical Research and Therapy.. 2017;4:1305-1317.
- [Google Scholar]
- L-deprenyl protects against rotenone-induced, oxidative stress-mediated dopaminergic neurodegeneration in rats. Neurochem. Int.. 2006;49:28-40.
- [Google Scholar]
- Effect of alternative pathway therapy on branched chain amino acid metabolism in urea cycle disorder patients. Mol. Genet. Metab.. 2004;81:79-85.
- [Google Scholar]
- Improved detection of substantia nigra pathology in Alzheimer's disease. J. Histochem. Cytochem.. 2002;50:99-106.
- [Google Scholar]
- Schinus terebinthifolius administration prevented behavioral and biochemical alterations in a rotenone model of Parkinson’s disease. Rev. Bras. 2016;26:240-245.
- [Google Scholar]
- Subcutaneous rotenone exposure causes highly selective dopaminergic degeneration and α-synuclein aggregation. Exp. Neurol.. 2003;179:9-16.
- [Google Scholar]
- Antimicrobial effects of sodium benzoate, sodium nitrite and potassium sorbate and their synergistic action in vitro. Bulgarian J. Agr. Sci.. 2009;15:307-311.
- [Google Scholar]
- Modified forelimb grip strength test detects aging-associated physiological decline in skeletal muscle function in male mice. Sci. Rep.. 2017;7:42323.
- [Google Scholar]
- Dose of phenobarbital and age of treatment at early life are two key factors for the persistent induction of cytochrome P450 enzymes in adult mouse liver. Drug Metab. Dispos.. 2015;43:1938-1945.
- [Google Scholar]
- α-synuclein promotes progression of Parkinson's disease by upregulating autophagy signaling pathway to activate NLRP3 inflammasome. Exp. Ther. Med.. 2020;19:931-938.
- [Google Scholar]
- Sodium benzoate attenuates D-serine induced nephrotoxicity in the rat. Toxicology. 2005;207:35-48.
- [Google Scholar]
- Oxidative stress, GTPCH1, and endothelial nitric oxide synthase uncoupling in hypertension. Antioxid. Redox Signal.. 2021;34:750-764.
- [Google Scholar]
- ADME–Tox in drug discovery: integration of experimental and computational technologies. Drug Discov. Today. 2003;8:852-861.
- [Google Scholar]
- Evaluation of C. cassia Effectiveness in Behavioral Modulation of Haloperidol Induced Parkinson’s Disease (Mice Model) British J. Pharm. Res.. 2016;6:1-7.
- [Google Scholar]
- Lauric acid: Its role in behavioral modulation, neuro-inflammatory and oxidative stress markers in haloperidol induced Parkinson's disease. Pak. J. Pharm. Sci.. 2020;33:755-763.
- [Google Scholar]







