Translate this page into:
Biosynthesis of Bixa orellana seed extract mediated silver nanoparticles with moderate antioxidant, antibacterial and antiproliferative activity
⁎Corresponding author. ahasanurrabbi@bcsir.gov.bd (M. Ahasanur Rabbi)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Biosynthesis of metallic silver nanoparticles (AgNPs) has gained much interest and offers an attractive alternate to physical and chemical approaches. In recent year several safe, easy, cost-effective, reproducible, and environmentally friendly synthesis approaches for silver nanoparticles have been developed. In this research work, a simple, cheap, and unexplored method was applied on green synthesis of AgNPs using secondary metabolites extracted from Bixa orellana seeds. The seeds are rich of flavonoids and phenolic compounds which presumably responsible for the fast reduction and stabilization of silver ion into silver nanoparticles. The biosynthesis process is very likely to be able to reduce silver ions under simple physiological conditions. The surface plasmon resonance (SPR) that was appeared at 420 nm in UV–vis spectrum, had confirmed the formation of AgNPs. Moreover, the functional groups in secondary metabolite that act as reducing, capping and stabilizing agents for silver nanoparticles, are identified by Fourier transform infrared (FTIR) spectra. An X-ray diffraction analysis generated four peaks for Bixa orellana seed extract mediated AgNPs positioned at 2θ angles of 38.1°, 44.2°, 64.6°, and 77.5° corresponding to crystal planes (1 1 1), (2 0 0), (2 2 0), and (3 1 1). Field emission scanning electron microscope (FESEM) and transmission electron microscopy (TEM) images confirmed the formation of nanosized silver particles. The z-average of the synthesized particles measured by dynamic light scattering (DLS) was found to be 92.9 nm. AgNPs synthesized exhibited remarkable antioxidant activity, antibacterial and antiproliferative activity against human breast (MCF-7) cell line. On the basis of our results, we conclude that biologically synthesized AgNPs exhibited favorable characteristics and have the potential to be used in biomedical fields.
Keywords
Biosynthesis
Bixa orellana
Antibacterial activity
Antioxidant activity
Antiproliferative activity
1 Introduction
Since last century nanotechnology is a rapidly growing field which is considered as the modern multidisciplinary science involving the fields of physics, chemistry, materials science, biochemistry, and medical science (Porter and Youtie, 2009, Tarafder et al., 2013). The term 'nanotechnology' has been used for a range of technologies, techniques and processes that involve the production, manipulation, and engineering of matter at the nanoscale level (atomic level) to describe extra-high precision and ultra-fine dimensions (Salata, 2004). In recent years, nanomaterials with unique physical, chemical, and biological properties are being used in medicine (Murthy, 2007, Zhang et al., 2008, Rudramurthy et al., 2018, Musielak et al., 2022) chemical sensing (Liu et al., 2018, Saha et al., 2012), imaging (Jin et al., 2008, Nune et al., 2009), drug delivery (Patra et al., 2018, Soppimath et al., 2001, Wilczewska et al., 2012, Yao et al., 2020), cosmetics (Aziz et al., 2019, Fytianos et al., 2020), catalysis (Astruc, 2020, Carabineiro, 2019, Testa et al, 2019), separation science (Che et al., 2019, Phuong et al., 2016) and electronics (Gross et al., 2007, Payel and Pandey, 2022). Nanoparticles (NPs) play an important role in nanotechnology which can be defined as the clusters of atoms that have a size of 1 to 100 nm in at least one dimension. NPs have possessed novel physical and chemical properties which make them different from that of the bulk form including vast surface area to volume proportion, high surface energy, uniformity, specific optical properties, mechanical strengths, and specific magnetizations (Khan et al., 2019).
Synthesis of metal nano-particles is achieved by several techniques which include physical, chemical or biological methods. But physical and chemical methods are not so suitable for the synthesis of silver nano-particles when we consider low toxicity, ecofriendliness, cost effectiveness, and low time consumption (Ahmad et al., 2011, Wu et al.,2010). Instead of conventional chemical synthesis methods, bio-synthesis of nanoparticles using microorganism (Klaus et al., 1999, Nair and Pradeep, 2002), enzymes (Thapa et al., 2017, Willner et al., 2006), and plant extract (Chung et al., 2016, Salayova et al., 2021, Vanlalveni et al., 2021) has been considered as a significant and valuable alternative. Plant source and plant extract are non-toxic in nature and thus provide a better platform for the green synthesis of nanoparticles (He et al., 2017a). Different parts of plant are being used to synthesize metal nanoparticles. In the recent past nanoparticle synthesis using flower (Devanesan and AlSalhi, 2021, Cheng et al., 2020, Mathew et al., 2020, He et al. 2018, Padalia et al., 2015), leaf (Khan et al., 2020, Jain and Mehata, 2017, Kouvaris et al., 2012), fruits (Kumar et al., 2017), fruit peel (Alkhulaifi et al.,2020, Ibrahim, 2015), root (Alishah et al., 2016, Arokiyaraj et al., 2017, Dangi et al., 2020), bark (Ahmed et al., 2016, Bharathi et al., 2018, Rajeshkumar, 2016), stem (Karthiga, 2018, Tahir et al., 2015, Wan Mat Khalir et al, 2020) and seed (Nawabjohn et al., 2022, Kanniah et al., 2021, Masudulla et al., 2021, Salmen, 2020, Shobana et al., 2020, He et al., 2017b) have been reported. Despite a good number of research articles have been found still extensive research work is carried out using plants and their parts for nanoparticle synthesis due to scale up cost effective commercial production. Scientists have been trying to find greener ways to ensure nanoparticles stay dispersed in their target environments with novel and improved properties (Heiligtag and Niederberger, 2013).
In recent time, natural products specially plant mediated synthesis of novel metal nanoparticles is a vastly emerging area of research and has been receiving increased attention due to their eco-friendly, biodegradable and nontoxic nature (Raveendran et al., 2003). In biosynthesis, plant extracts act as provider of both reducing agents for reduction of silver ions into silver metal and a capping agent for stabilizing the produced nanoparticles. Generally, alkaloids, tannins, steroids, phenols, saponins and flavonoids compounds of plant extracts are the major phytochemicals that are induced green synthesis of nanoparticles, due to their reducing properties and high stability of the synthesized nanoparticles (Ahmed et al., 2016). During the past few years various metals have been used to prepare nanoparticles but nanocrystalline silver is one of the best-known nanomaterials which have been extensively used in medicine throughout recorded history (Klasen, 2000, Patra et al., 2008, Strohal et al., 2005). Currently AgNPs have attracted the attention of researchers to employ it in different applications because of their chemical stability, localized surface plasma resonance, catalytic activity, high conductivity and antibacterial activities. Consequently, AgNPs are predominantly engineered for biomedical applications such as targeted drug delivery, bio-labeling, imaging probe, wound healing, endodontics etc. (Nakamura et al., 2019, Shameli et al., 2012).
It has been demonstrated that plant-derived phytochemicals such as, such as polysaccharides, flavonoids, phenolic acids and alkaloids, are capable of effectively reducing metal ions. These compounds also act as stabilizing agent and provide colloidal stability along with preventing agglomeration and stopping uncontrolled growth of metal nanoparticles (Xulu et al., 2022). So, the main goal of the present study is to synthesize and characterize silver nanoparticles for the first time using aqueous seed extract of Bixa orellana which is commonly known as Anatto, Lipstick Tree, Achiote, etc. It is a medium sized evergreen shrub and its height can reach 5–10 m. The fruits of Bixa orellana are red, globular, ovoid capsules. Capsules are arranged in clusters that are looking like red-brown seed pods. 30–45 cone-shaped seeds are contained by each capsule or pod and these seeds are covered in a thin waxy blood-red aril. After full maturation, dried capsules are splited to expose the red seeds. These seeds and the extract have been used for more than 200 years in Europe and North America as food colorant (Chuyen et al. 2012). Hard seeds of Bixa orellana are rich in flavonoids (Abayomi et al., 2014) and carotenoids (Rivera-Madrid et al., 2016, Raddatz-Mota et al., 2017), which can be a promising green resource to synthesize and stabilize metal nanoparticles (Hussain, et al. 2019). There are no reports available for silver nanoparticles (AgNPs) synthesis using bioactive extract of Bixa Orellana seeds and to the best of our knowledge, this is the first report of a rapid, room temperature protocol for AgNPs synthesis using the seed extract.
2 Materials & methods
2.1 Materials
Bixa orellana seeds were collected from remote area of Rajshahi. After washing the seeds were kept at room temperature for drying. The dried seeds were stored in a dry and airtight container for further use. All chemicals used in the investigation were of reagent grade purity. Silver nitrate (AgNO3) was purchased from Mark, India and sodium hydroxide (NaOH) and 2,2-Diphenyl-1-picrylhydrazyl (DPPH) were purchased from Merck, Germany. Four different bacterial strains namely Staphylococcus aureus (ATCC 25956), Escherichia coli (ATCC 27853), Shigella dysenteriae (ATCC 26131) and Shigella boydii (ATCC 27890) were collected from Department of Biochemistry and Molecular Biology, Rajshahi University. These strains were preserved at 4 °C on agar slant and sub cultured at 37 °C for 24 h on nutrient broth media. Antibiotic, Erythromycin standard disc was used as positive control for antibacterial activity and water was distilled using a laboratory (Pyrex glass) distillation apparatus before the experiment.
2.1.1 Preparation of Bixa orellana seed extract
25 g of seed was weighted carefully and transferred it in a beaker of 1000 mL containing 250 mL of distilled water and 1 mL of 1 M Sodium hydroxide (NaOH) solution was added to it. The mixture was warmed at 70 °C with continuous stirring for 30 min on hot plate magnetic stirrer. The 1st extract was collected by filtering with clean muslin cloth. Again 250 mL of distilled water and 1 M NaOH solution was added to that beaker containing seed residue and stirred for 2 h at 70 °C. The 2nd extract was collected by filtering with another muslin cloth and both of the filtrates were mixed together. Finally, the filtrate was filtered again through Whatmann filter paper (Fig. S1) and stored at 4 °C prior synthesis of nanoparticles.
2.1.2 Green synthesis of silver nanoparticle
1 mM silver nitrate solution was used as the source of silver. Seed extract and silver nitrate solution were mixed together with ratio of 1:1. 5 mL of 1 M NaOH solution was added to the mixture to facilitates the reduction of silver ions by generating intermediates that are more easily reduced to zero valence silver (Park et al., 2020). The mixture was continuously stirred at 400–500 rpm on a magnetic stirrer. The flow of nitrogen gas was supplied during synthesis to make inert atmosphere to prevent the formation of silver oxide. The mixture turned into dark brown color within few minutes but the reaction was continued for 2 h to maximize the AgNPs synthesis. The bio synthesized AgNPs were kept at room temperature for 24 h. The prepared AgNPs were washed by using dialysis membrane technique (dialysis membrane 60, Himedia, India). The washed nanoparticles were stored in a cool, dry, dark place and later their characterization was performed.
2.2 Characterization
UV–vis spectral analysis was performed by using Spectrum UV–visible spectrophotometer (Spectrum 500 D, Germany). The absorbance maximum was recorded for the dilute solutions of seed extract and dilute emulsion of synthesized AgNPs with the wavelength range from 200 to 800 nm. Thermogravimetric analyzer, TGA (STA 8000, Perkin Elmer, Netherland) was used to measure the thermal properties for 10.0 mg of the dried powder samples. The sample was dried under a flowing nitrogen atmosphere at a heating rate of 20 °C min−1 and the mass loss in percent was recorded. Fourier Transform IR, FTIR (Perkin Elmer, FTIR-1400, UK) was used as a tool to investigate the structural functionality of Bixa orellana seed extract and synthesized AgNPs. The seed extract and synthesized AgNPs samples were dried at low temperature (∼50 °C) under vacuum and mixed with KBr. The samples were dried again prior analysis and then pressed onto pellets. Samples were scanned over the range between 4000 and 400 cm−1 in deflection mode at room temperature. Optical microscope images of the synthesized AgNPs were taken by N-800 M (China) to study the cluster size and shape. Dilute dispersion of the washed particles was used for optical microscope images and images were captured at 40 × magnification. Field emission scanning electron microscope (FESEM) image provides information of surface structure and morphology of nanomaterials through the scattered secondary electrons by the surface atoms. Synthesized AgNPs was imaged in the FESEM using a JSM-7610F operated in the range of 5 to 15 kV and magnification × 200,000. Before imaging in the SEM dried sample was platinum coated by JEOL Auto fine coater (JEC-3000FC). EDX spectrum was recorded with energy dispersive X-ray spectroscopy adaptor coupled to the SEM. Transmission electron microscope (TEM) images were taken by Taalos F 200X, Thermofisher Scientific for characterizing size and shape of synthesized AgNPs. TEM measurement was prepared by depositing a drop of colloid solution on a 300-mesh copper (Cu) grid coated by carbon film and evaporating the solvent in air at room temperature. Size distribution of the synthesized AgNPs and its polydispersity index (PI) was checked using a particle size analyzer. The colloidal dispersion of NPs was analyzed by Horiba analyzer (SZ-100). Particle size analysis is performed by dynamic light scattering (DLS) at 25 ⁰C with scattering angle 90. Scanning X-ray diffractometer (Bruker D8 Advance, Germany) was used to record the X-ray diffraction (XRD) patterns of the synthesized AgNPs where the intensity measurement was done at 2θ values from 10 to 80°. Here the scanning rate was 1° min−1 with a position-sensitive detector aperture at 25 °C. Semi quantitative phase analysis software was applied to process diffraction spectrum and this software was useful to minimize noises, smooth the data and identify the peaks.
2.2.1 Free radical scavenging activity
The DPPH assay method as described by Shen et al. (2010) was followed here to evaluate free radical scavenging capability of synthesized AgNPs. In this assay, first 0.1 mM solution of DPPH was prepared in methanol. Then 1 mL of this solution was added to 3 mL of methanolic dispersion of synthesized AgNPs. Ascorbic acid was used as reference materials and the DPPH scavenging activity with variable particle concentrations (50, 100 and 200 µg mL−1) were studied. After vigorous shaking, the mixtures were allowed to stand in a dark place at room temperature for 30 min. Then absorbance was taken at 517 nm and the percentage of DPPH radical scavenging was calculated by using the following formula.
where Acontrol was the absorbance of reaction mixture without samples and Asample was the absorbance of the reaction mixture with samples. After three-time replication, the mean values were used to calculate percentage (%) scavenging.
2.2.2 Antibacterial activity assay
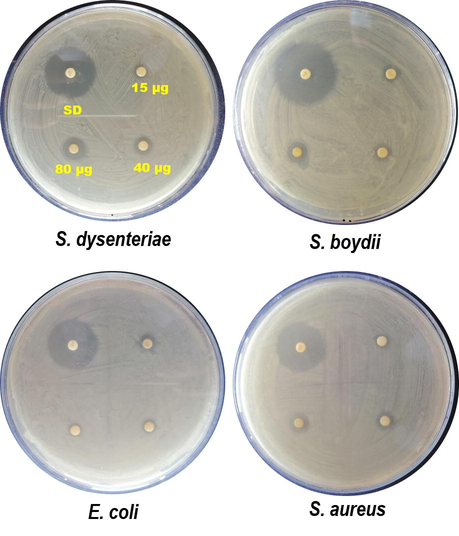
The antimicrobial efficacy of synthesized AgNPs was evaluated by disc diffusion assay method (Habib et al., 2009). Separate sterilized petri dish was used for each type of pathogens and particles. 0.2 mL of cultured test organism was poured on the surface of solid nutrient agar media previously taken into each sterilized petri dish. Then 7.5–40 µL of dispersion containing three different amounts (15, 40 and 80 µg) of respective particles were dropped carefully over filter paper disks that were previously placed on the surface of cultured test organism. Erythromycin standard disc (15 µg/disc) was used as positive control for each petri dish. All the assay plates were placed in the refrigerator at 4 °C for 4 h for allowing sample diffusion. After diffusion, all the plates were incubated at 37 ± 1 °C for 24 h to ensure bacterial growth. After incubation, diameter of the zone of inhibition surrounding each filter paper disc was measured. Each experiment was carried out in triplicates and mean value of inhibition zone for a particular dose was taken to make final results.
2.2.3 Evaluation of the anticancer Activity:
Cell Culture: DMEM medium supplemented with 10 % FBS, neomycin, and streptomycin was taken in a 25 cm2 cell culture flask and it was used to culture the MCF-7 cells at 37 °C in 5 % CO2. Subcultures were carried out at 80–90 % confluence (Kabir et al., 2020).
Assay for antiproliferative activity of AgNPs against MCF-7: MTS assay was done to detect proliferation of MCF-7 cells using the guideline of manufacturer (Promega, USA). For MTS assay, 1 × 104 MCF-7 cells were seeded in 150 μL of DMEM medium contained by the each well of the 96-well flatbottom culture plate and the plate was incubated at 37 °C in a CO2 incubator for 24 h. Then, serially diluted AgNPs in 50 μL of DMEM medium were added to each well of the culture plate and the final concentration of the test sample was 3.90–37.25 μg/mL. Then the plate was incubated at 37 °C in a CO2 incubator for 48 h. Wells containing only MCF-7 cells without AgNPs were used as controls. Then, aliquot of each well was removed, and 100 μL of DMEM medium was added. After that, 10 μL of MTS with phenazine ethosulfate was added to each well and incubated for 2 h. Finally, a micro plate reader with 490 nm filter was used for measuring the absorbance. Three wells were employed for each concentration, and by the following equation, cell proliferation inhibition was estimated.
where A and B designates OD490 nm of the cellular homogenate (control) without AgNPs and with AgNPs, respectively (see Fig. 1).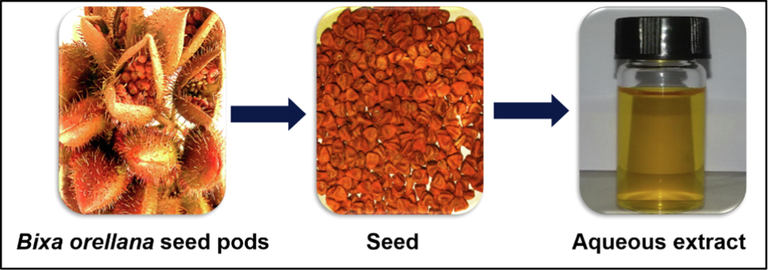
Extraction of reducing agents from Bixa Orellana seeds.
3 Results and discussion
The preparation of silver nanoparticles was done at room temperature by simple bottom-up approach. Biosynthesis of AgNPs using aqueous extract of Bixa orellana seeds can be easily monitored from the instant change in color of the reaction mixture. A distinct color change was observed after addition of colorless silver nitrate aqueous solution to alkaline seed extract (Fig. 2). The color of the seed extract changed from light yellow to dark brown indicating formation of silver nanoparticles (Ahmed et al., 2016).
Color change due to the formation of AgNPs.
UV–visible spectroscopy is one of the most widely used techniques for the analysis of NPs and the synthesized AgNPs were primarily characterized by UV–vis absorption spectrum. The absorption spectrum due to the free electron excitation of the brown colored silver colloids was confirms the production of silver nanoparticles and the surface plasmon absorption band with a maximum of 420 nm (Fig. 3b) indicating the presence of spherical or nearly spherical silver nanoparticles (Kumar et al., 2018, Roy et al., 2017). The absorption spectrum of the seed extract and AgNPs are shown in Fig. 3. The UV–vis spectrum was recorded for extended time period to investigate the stability of AgNPs. The synthesized AgNPs were found to be stable as no precipitation was observed in the sol (Guzman et al., 2009).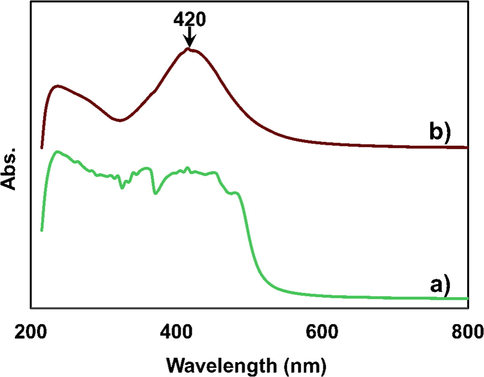
UV–vis spectra of a) Bixa orellana seed extract & b) Bixa orellana seed extract mediated AgNPs.
FTIR analysis of pure Bixa orellana seed extract was carried out to identify the potential functional groups of the phytochemicals in its seed extract and that might have contributed to the process of bio-reduction of silver ions (Ag+) to silver nanoparticles (Ag0). The FTIR spectrum of Bixa orellana seed extract showed peaks at around 2930, 1615 and 1383 cm−1 which correspond to the C—H stretching, C⚌C stretching and C—H bending vibrations respectively which is in good correlation with that of the other reports (Alshehri and Malik, 2020, Devi et al., 2020). FTIR spectrum of AgNPs showed high similarity with the spectra of the seed extract except small change in peak position and intensity (Fig. 4). The dual role of the seed extract as a reducing and capping agent and presence of same functional groups in AgNPs was confirmed by FTIR analysis of synthesized silver nanoparticle. The peaks for C—H stretching, C⚌C stretching and C—H bending vibrations were shifted to 2926, 1610 and 1384 respectively in the FTIR spectrum of synthesized AgNPs. The involvement of biomolecules extracted from Bixa orellana seed in biosynthesizing of nanoparticles was confirmed with shifting of peaks (Monisha et al., 2017). Two broad bands were found in the both spectra at 3600–3200 cm−1 and 1150–1010 cm−1 due to the stretching vibrations of O—H (alcohol/phenol) and C—O (alcohol) respectively (Pirtarighat et al., 2019). The shoulder peak at 1715 cm−1 was found in the FTIR spectrum of B. orellana seed extract due to C⚌O stretching which was shifted to 1710 cm−1 in the AgNPs. There was no excess peak in the range of 500–540, corresponding to free metal ions of silver (Ssekatawa et al., 2021). Thus, the FTIR study reveals the flavonoids the other compounds including OH and CO groups have a vital role as a reducing agent, which reduces Ag+ to Ag0 and as a stabilizing agent in the green synthesis of AgNPs.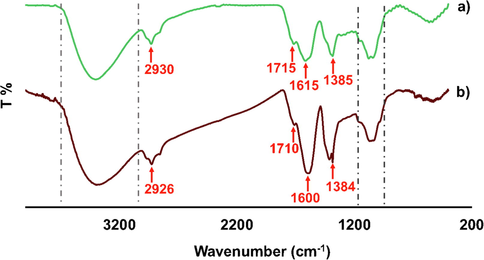
FTIR spectra of Bixa orellana a) seed extract & b) seed extract mediated AgNPs.
Thermogravimetric (TG) curves of silver nanoparticles synthesized by Bixa orellana seed extract is presented in Fig. 5. In initial step of the decomposition process occurring between 40 and 100 °C, weight loss (∼3%) of sample might be associated with the evaporation of moisture adsorbed on the surface of the AgNPs. At a temperature of 100 to 380 °C AgNPs sample again loses about 21 % of its weight due to the consequence of the degradation of the organic biomolecules present at the surface of the nanoparticles as capping agent (Esfanddarani et al., 2018). The reason of this weight loss is the decomposition of phenolic acids, flavonoids and carotenoids which present in the seed extract and are responsible for the stabilization of AgNPs (David and Moldovan, 2020). A gradual loss accounting about 26 % was observed between 350 and 450 °C which might be happened because of the thermal degradation of resistant aromatic compounds present on the surface of the silver nanoparticles. Above 450 °C no weight loss was observed until 800 °C and the residual mass at 800 °C was found to be 49.80 %. Thus, residual mass (∼50 %) of the particles attributed to the presence of metallic silver (Moldovan et al., 2018).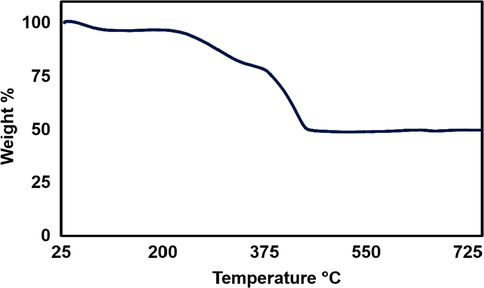
TGA graph of silver nanoparticles synthesized by Bixa orellana seed extract.
The X-ray diffraction (XRD) analysis has confirmed the crystalline nature of the synthesized AgNPs and the powder XRD pattern is shown in Fig. 6. Bragg Reflection peaks as shown by silver nanoparticles were found at 38.1°, 44.2°, 64.6° and 77.5° that correspond to (1 1 1), (2 0 0), (2 2 0) and (3 1 1) planes of face centered cubic (FCC) crystal structure of metallic silver, respectively (Rabbi et al., 2020, Venkatadri et al., 2020, Jemal et al., 2017). The reflection planes have a good match with the Joint Committee of Powder Diffraction Standard (JCPDS File No. 04–0783) corresponding to the reflections of crystal planes. Synthesized silver nanoparticles are composed of pure crystalline silver. The peak corresponding to (1 1 1) plane is more intense than the other planes, suggesting that the (1 1 1) plane is in the predominant orientation.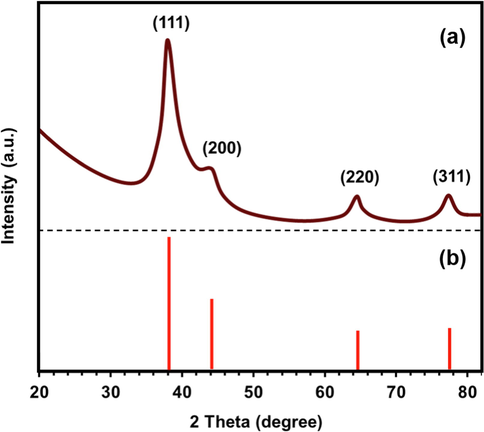
(a) XRD pattern of AgNPs synthesized by Bixa orellana seed extract, (b) JCPDS file No. 04–0783.
Fig. 7 represents (a) Field emission scanning electron microscope (FESEM) and (b) Transmission electron microscope images of the synthesized AgNPs. As shown in Fig. 7(a), the nanoparticles were aggregated in layer with average size distribution varying from approximately 20 to 40 nm. Clustering of nanoparticles may be attributed to the interaction of concentrated nanoparticles with the organic components of Bixa orellana seed extracts used in biosynthesis (Ssekatawa et al. 2021). Aggregation of nanoparticles was induced by solvent evaporation during preparation of the film of nanoparticles for FESEM imaging. SEM image also displayed crystalline and quasi-spherical shapes except few large aggregates. The synthesized AgNPs might be spherical in shape throughout in the colloidal solution (Ahmad and Sharma, 2012).
(a) FESEM and (b) TEM images of AgNPs synthesized by Bixa orellana seed extract.
Inclusion of the organic phytochemicals as capping agent from Bixa orellana seed extract can be observed by difference in contrast of the particle image (Fig. 7b) where the lighter part around the particle represents segment (Palani-velan et al., 2015). This thin layer around AgNPs, suggests that, the silver nanoparticles were successfully capped by water soluble phytochemicals which significantly hindered aggregation of AgNPs (He et al. 2017a). However partial agglomeration observed in the TEM image which might be occurred either during sample preparation or during exposure to electron beam. Partial agglomeration is also confirmed by the optical microscope images (Fig. S2) of dilute dispersion of synthesized AgNPs. The colloidal suspension of synthesized AgNPs was stable and from DLS histogram the average hydrodynamic diameter was around 92.9 nm (Fig. 8). DLS measures only the diameter of nanoparticles dispersed in liquid and not the actual size of the nanoparticles. Subsequently the average size of biosynthesized AgNPs obtained from DLS in this work is larger than that measured from SEM and TEM. It was reported that larger size from DLS than SEM and TEM is due to result of Brownian motion influence (Adebayo-Tayo et al. 2019, Zhang et al. 2016, Lim et al. 2013). According to Mulvaney (1996) the colloidal solutions having a polydispersity index (PI) less than 0.50 are considered to be of “good” quality. The PI for the colloidal solution of AgNPs is 0.310 and despite partial aggregation, individual cluster of AgNPs which are as small as 100 nm are still visible (indicated by yellow box in Fig. 7a). Bixa orellana seed extract mediated bioynthesis process results in the formation of a nearly monodisperse colloidal dispersion of AgNPs. The dispersion was found to be stable which is also in accordance with the result previously reported for colloidal nanoparticles stability (Kartini et al. 2020).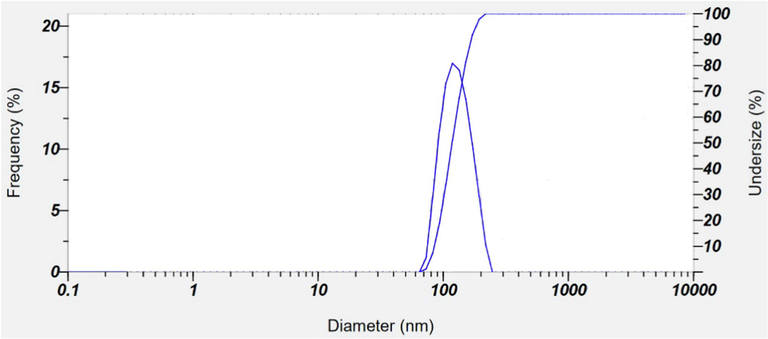
Size distribution of AgNPs as studied by DLS.
Energy-dispersive X-ray spectroscopy (EDX) is a surface analytical technique which is used to estimate the composition or number of metals at the surface of the nanoparticles can be estimated. EDX spectrum is the graphical representation of the ionization energy versus counts. Fig. 9 shows the EDX spectrum of AgNPs synthesized by Bixa orellana seed extract. EDX spectroscopy results confirmed the significant presence of all the peaks of pure metallic Ag with no other elemental contaminants. The AgNPs showed a strong absorption peak at approximately 3 keV, which is the characteristic absorption of metallic silver nano crystal (Aromal and Philip, 2012, Abdel-Hafez et al., 2016). Weaker signals from O & C atoms were appeared which correspond to the organic compounds present as capping agents and originated from Bixa orellana seed extract. Absorption peak for Pt was appeared due to platinum-coating which was used to prepare the sample before SEM/EDX analysis. The weight percentage (W%) of silver, oxygen and carbon were found to be 53.65, 21.34 and 25.01 % respectively. Thus, from EDX analysis the weight percentage of nano silver is about 53 %, this result also supports the finding of the TGA thermograph (see Figs. 5 and 9 ).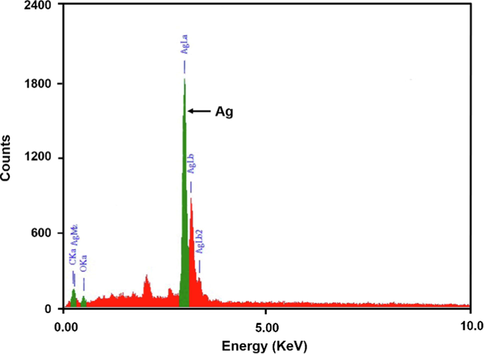
EDX spectrum of AgNPs synthesized by Bixa orellana seed extract.
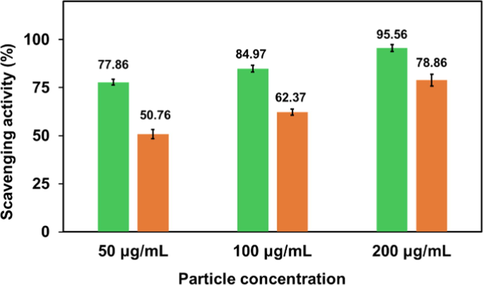
Free radical scavenging activity of ascorbic acid (green column) and synthesized AgNPs (orange column).
Antioxidants that can be natural or synthetic substances, are vital agents to protect or delay cell damage caused by oxidants (Azeez et al., 2017, Bedlovicova et al., 2020). Metal and metal oxide nanoparticles, carbon nanotubes, and various types of nanomaterials have been reported to exhibit antioxidant properties (Valgimigli et al., 2018). To investigate the antioxidant activity of metal nanoparticles DPPH assay is most commonly used. Generally, all green synthesized silver nanoparticles possess higher values of antioxidant activity than plant extract alone (Salari et al., 2019). At room temperature the delocalization of an unpaired electron over the molecule is responsible for stability of DPPH radical which gives rise to the deep violet color. This radical can be reduced in the presence of an antioxidant molecule by accepting an electron or hydrogen (Kedare and Singh, 2011, Kharat and Mendhulkar, 2016) to become stable. The DPPH radical scavenging activity of synthesized AgNPs were found to be 50.76, 62.78 and 78.86 % in presence of 50, 100 and 200 μg/ mL AgNPs respectively (Fig. 10). Thus, some active constituents present in the synthesized AgNPs, are capable of donating electron or hydrogen to a free radical in order to remove odd electron and this electron or hydrogen donation is responsible for radical’s reactivity (Gangwar et al., 2014). Particularly, the presence of phytochemicals like phenolic compounds, terpenoids, and flavonoids in Bixa orellana seed extract act as key players for offering the antioxidant activity of AgNPs. They allow nanoparticles to play role as singlet oxygen quenchers, hydrogen donors, and reducing agents simultaneously (Elemike et al., 2017). The findings can be correlated with the DPPH free radical scavenging activity of pure Bioxa orellana seed extract (Abayomi et al., 2014).
The antibacterial properties of silver have been well known and the green synthesis process can improve the biocompatibility and reduce their potential harmful effects on human health and the environment (Qing et al., 2018). In this study antibacterial activities of the Bixa orellana seed extract mediated silver nanoparticles have been investigated against four pathogens. No inhibition zone was found for the negative control and the antibacterial activities of AgNPs increased with the increase of particle concentration which supports previous findings (Moldovan et al., 2018, Duran et al., 2010). Size, shape and formulation of silver nanoparticles also influence their antibacterial efficacy (Franci et al., 2015). Synthesized AgNPs possessed remarkable antibacterial properties against the growth of all Gram positive and Gram-negative bacterial strains used in this study (Fig. S3). With variable particle concentrations the inhibition zones for AgNPs are in the range of 07–17 mm (Fig. 11). With 80 µg/disc AgNPs, the maximum inhibition (17 mm) zone is observed against Shigella dysenteriae. The above study also suggests that comparatively less toxic AgNPs with moderate antibacterial activity will be a promising bioactive nanomaterial.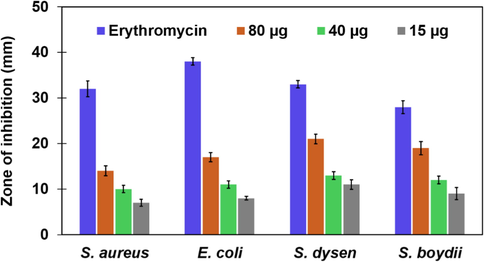
Antibacterial activity of AgNPs synthesized by Bixa orellana seed extract.
The in vitro cytotoxicity of synthesized AgNPs against MCF-7 cell line was determined using the MTS test. It was obvious from Fig. 12, that AgNPs had the ability to diminish MCF-7 cell viability in a dose-dependent manner. AgNPs showed cytotoxicity to EAC cells at four different concentrations after completion of treatment. The growth inhibitory effect of AgNPs was found to get decreased gradually with reduced concentrations. The maximum inhibition (68.3 %) was seen at a concentration of 31.25 µg/ml, while the least inhibition (21.2 %) was obtained at a concentration of 3.90 µg/ml. When the concentration of AgNPs was reduced to ten times lower from higher concentration, the percentage of inhibition became one third of maximum inhibition. So drastic reduction was not observed in growth inhibition by lower dose of AgNPs. Previous study done by Kaler et al. also showed similar type of results where the green synthesized AgNPs have cytotoxicity against MCF-7 cells (Kaler et al., 2013). Moreover, the anticancer activity of Potentilla fulgens mediated AgNPs was tested against MCF-7 cancer cell line (Mittal et al., 2015) where a 42 % of growth inhibition was reported for MCF-7 at 6 μg/mL concentration. In this study, 34.8 % inhibition was observed for AgNPs at 7.81 μg/mL concentration. As compared with this previous observation, AgNPs synthesized from Bixa orellana seed extract showed moderate efficacy against MCF-7 cancer cell line.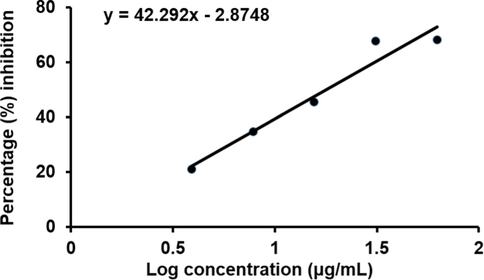
In vitro growth inhibitory activity of AgNPs against MCF-7 cell line. Each value is presented as the mean ± standard deviation from triplicate measurements.
4 Conclusions
In this present study, we have demonstrated the use of Bixa orellana seed extract as the reducing and capping agent for the rapid synthesis of AgNPs at room temperature through a simple one pot synthesis. The dispersion of AgNPs was stable without any external stabilizer. The active phytoconstituents present in the Bixa Orellana seed extract as detected by FTIR may have aided the successful bio-reduction of Ag+ to Ag0 and long-term stabilization of synthesized AgNPs. Average hydrodynamic diameter of the monodispersed AgNPs was found to be 92.9 nm and the XRD peaks attribute to the crystalline FCC structure of silver. Synthesized AgNPs exhibited significant antioxidant activity with moderate antibacterial activity against both Gram-positive and Gram-negative bacteria. Furthermore, synthesized AgNPs showed moderate efficacy against MCF-7 cancer cell line. The synthesis method of AgNPs using Bixa orellana seed extract is energy efficient, cost effective, protecting human health and environment followed by green chemistry principle. This extract highly rich in phenolic compounds and results suggest that the synthesis process of AgNPs can be considered for further scale-up process studies to provide AgNPs in a sufficient amount. This eco-friendly method could be a competitive alternative to the conventional physical or chemical methods used for synthesis of AgNPs and thus has a potential to use in biomedical applications such as fabrication of hospital clothes, gloves and masks to avoid the spread of infection among healthcare workers.
Conflict of interest
The authors confirm that they have no conflicts of interest and the data included in this manuscript have not been published previously.
Acknowledgments
This work was supported by Bangladesh Council of Scientific & Industrial Research (BCSIR), Ministry of Science and Technology, People's Republic of Bangladesh.
References
- In vitro antioxidant activity of Bixa orellana (Annatto) seed extract. J. Appl. Pharm. Sci.. 2014;4(2):101-106.
- [CrossRef] [Google Scholar]
- Assessment of protein silver nanoparticles toxicity against pathogenic Alternaria solani. 3 Biotech. 2016;6:199.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticle using Oscillatoria sp. extract, its antibacterial, antibiofilm potential and cytotoxicity activity. Heliyon. 2019;5:e02502.
- [Google Scholar]
- Synthesis of silver nanoparticles in chitosan: gelatin and chitosan/gelatinbionanocomposites by a chemical reducing agent and their characterization. Molecules.. 2011;16(9):7237-7248.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles using extracts of Ananas comosus. Green Sustain. Chem.. 2012;2:141-147.
- [CrossRef] [Google Scholar]
- Antibacterial efficacy of silver nanoparticles synthesized employing Terminalia arjuna bark extract. Artif. Cells Nanomed. Biotechnol.. 2016;45(6):1-9.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles using Azadirachta indica aqueous leaf extract. J. Radiat. Res. Appl. Sci.. 2016;9(1):1-7.
- [CrossRef] [Google Scholar]
- A green approach for silver nanoparticles using root extract of Chelidonium majus: characterization and antibacterial evaluation. J. Clust. Sci.. 2016;27(2):421-429. 10.1007%2Fs10876-016-0968-0
- [Google Scholar]
- Green synthesis of silver nanoparticles using citrus limon peels and evaluation of their antibacterial and cytotoxic properties. Saudi J. Biol. Sci.. 2020;27(12):3434-3441.
- [CrossRef] [Google Scholar]
- Phytomediated photo-induced green synthesis of silver nanoparticles using Matricaria chamomilla L. and its catalytic activity against Rhodamine B. Biomolecules.. 2020;10(12):1604.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles using Rheum palmatum root extract and their antibacterial activity against Staphylococcus aureus and Pseudomonas aeruginosa. Artif. Cells Nanomed. Biotechnol.. 2017;45(2):372-379.
- [CrossRef] [Google Scholar]
- Green synthesis of gold nanoparticles using Trigonella foenum-graecum and its size-dependent catalytic activity. Spectrochimica Acta. Part A. Molecular and Biomolecular Spectroscopy.. 2012;97:1-5.
- [CrossRef] [Google Scholar]
- Introduction: Nanoparticles in catalysis. Chem. Rev.. 2020;120(2):461-463.
- [CrossRef] [Google Scholar]
- Silver nanoparticles (AgNPs) biosynthesized using pod extract of Cola nitida enhances antioxidant activity and phytochemical composition of Amaranthus caudatus Linn. Appl. Nanosci.. 2017;7:59-66.
- [CrossRef] [Google Scholar]
- Role of nanotechnology for design and development of cosmeceutical: Application in makeup and skin care. Front. Chem.. 2019;7:739.
- [CrossRef] [Google Scholar]
- Brief overview on antioxidant activity determination of silver nanoparticles. Molecules. 2020;25:3191.
- [Google Scholar]
- Biosynthesis of silver nanoparticles using stem bark extracts of Diospyros montana and their antioxidant and antibacterial activities. J. Nanostructure Chem.. 2018;8:83-92.
- [CrossRef] [Google Scholar]
- Supported gold nanoparticles as catalysts for the oxidation of alcohols and alkanes. Front. Chem.. 2019;7:702.
- [CrossRef] [Google Scholar]
- Wood-based mesoporous filter decorated with silver nanoparticles for water purification. ACS Sustain. Chem. Eng.. 2019;7(5):5134-5141.
- [CrossRef] [Google Scholar]
- Phytofabrication of silver nanoparticles using three flower extracts and their antibacterial activities against pathogen Ralstonia solanacearum strain YY06 of bacterial wilt. Front. Microbiol.. 2020;11:2110.
- [CrossRef] [Google Scholar]
- Plant-mediated synthesis of silver nanoparticles: Their characteristic properties and therapeutic applications. Nanoscale Res. Lett.. 2016;11(1):40.
- [CrossRef] [Google Scholar]
- Improvement of bixin extraction yield and extraction quality from annatto seed by modification and combination of different extraction methods. Int. J. Food Sci.. 2012;47:1333-1338.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles using aqueous root extract of Berberis asiatica and evaluation of their antibacterial activity. Chem. Data Collect.. 2020;28:100411
- [CrossRef] [Google Scholar]
- Green synthesis of biogenic silver nanoparticles for efficient catalytic removal of harmful organic dyes. J. Nanomater.. 2020;10(2):202.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles using the flower extract of Abelmoschus esculentus for cytotoxicity and antimicrobial studies. Int. J. Nanomedicine.. 2021;16:3343-3356.
- [CrossRef] [Google Scholar]
- Spectroscopic investigation of green synthesized ZnS nanoparticles encapsulated by sodium carboxy methyl cellulose. Appl. Phys. A.. 2020;126:924.
- [CrossRef] [Google Scholar]
- Potential use of silver nanoparticles on pathogenic bacteria, their toxicity and possible mechanisms of action. J. Braz. Chem. Soc.. 2010;21(6):949-959.
- [CrossRef] [Google Scholar]
- Silver nanoparticles mediated by Costus afer leaf electrochemical properties. Molecules.. 2017;2017:22.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles using flower extract of Malva sylvestris and investigation of their antibacterial activity. IET Nanobiotechnol.. 2018;12:412-416.
- [CrossRef] [Google Scholar]
- Silver nanoparticles as potential antibacterial agents. Molecules.. 2015;20(5):8856-8874.
- [CrossRef] [Google Scholar]
- Nanomaterials in cosmetics: Recent updates. Nanomaterials. 2020;10(5):979.
- [CrossRef]
- Antioxidant capacity and radical scavenging effect of polyphenol rich Mallotus philippenensis fruit extract on human erythrocytes: An in vitro study. Sci. World J.. 2014;2014:279451
- [CrossRef] [Google Scholar]
- Electrical, optical and morphological properties of nanoparticle indium-tin oxide layers. Thin Solid Films.. 2007;515(24):8567-8572.
- [CrossRef] [Google Scholar]
- Synthesis of silver nanoparticles by chemical reduction method and their antibacterial activity. Int. J. Chem. Biomol. Eng.. 2009;2(3):2009.
- [Google Scholar]
- Antimicrobial and cytotoxic activity of di-(2-ethylhexyl) phthalate and anhydrosophoradiol-3-acetate isolated from Calotropic gigentia (Linn.) flower. Micobiology.. 2009;37(1):31-36.
- [Google Scholar]
- Synthesis, characterization and evaluation cytotoxic activity of silver nanoparticles synthesized by Chinese herbal Cornus officinalis via environment friendly approach. Environ. Toxicol. Pharmacol.. 2017;56:56-60.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles using seed extract of Alpinia katsumadai, and their antioxidant, cytotoxicity, and antibacterial activities. RSC Adv.. 2017;7(63):39842-39851.
- [CrossRef] [Google Scholar]
- A green approach of synthesizing of silver nanoparticles and their antibacterial and cytotoxicity activities. New J. Chem.. 2018;42:2882-2888.
- [CrossRef] [Google Scholar]
- Applications of Plant Flavonoids in the Green Synthesis of Colloidal Silver Nanoparticles and Impacts on Human Health. Iran. J. Sci. Technol. Trans. Sci.. 2019;43:1381-1392.
- [CrossRef] [Google Scholar]
- Green synthesis and characterization of silver nanoparticles using banana peel extract and their antimicrobial activity against representative microorganisms. J. Radiat. Res. Appl. Sci.. 2015;8(3):265-275.
- [Google Scholar]
- Medicinal plant leaf extract and pure flavonoid mediated green synthesis of silver nanoparticles and their enhanced antibacterial property. Sci. Rep.. 2017;7:15867.
- [CrossRef] [Google Scholar]
- Synthesis, characterization, and evaluation of the antibacterial activity of Allophylus serratus leaf and leaf derived Callus extracts mediated silver nanoparticles. J. Nanomater.. 2017;4213275
- [CrossRef] [Google Scholar]
- Preparation and characterization of highly fluorescent, glutathione coated near infrared quantum dots for in vivo fluorescence imaging. Int. J. Mol. Sci.. 2008;9(10):2044-2061.
- [Google Scholar]
- Zizyphus mauritiana fruit extract-mediated synthesized silver/silver chloride nanoparticles retain antimicrobial activity and induce apoptosis in MCF-7 Cells through the Fas Pathway. ACS Omega.. 2020;5(32):20599-20608.
- [CrossRef] [Google Scholar]
- Green and rapid synthesis of anticancerous silver nanoparticles by Saccharomyces boulardii and insight into mechanism of nanoparticle synthesis. Biomed. Res. Int. 2013:872940.
- [Google Scholar]
- Green synthesis of antibacterial and cytotoxic silver nanoparticles by Piper nigrum seed extract and development of antibacterial silver based chitosan nanocomposite. Int. J. Biol. Macromol.. 2021;189:18-33.
- [CrossRef] [Google Scholar]
- Preparation of silver nanoparticles by Garcina mangostana stem extract and investigation of the antimicrobial properties. Biotechnol. Res. Innov.. 2018;2(1):30-36.
- [CrossRef] [Google Scholar]
- Process Optimization for Green Synthesis of Silver Nanoparticles Using Indonesian Medicinal Plant Extracts. Processes. 2020;8:998.
- [CrossRef] [Google Scholar]
- Genesis and development of DPPH method of antioxidant assay. J. Food Sci. Technol.. 2011;48(4):412-422.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles using Ipomoea aquatica leaf extract and its cytotoxicity and antibacterial activity assay. Green Chem. Lett. Rev.. 2020;13(4):303-315.
- [CrossRef] [Google Scholar]
- Nanoparticles: Properties, applications and toxicities. Arab. J. Chem.. 2019;12(7):908-931.
- [CrossRef] [Google Scholar]
- Rapid Green Synthesis and Characterization of Silver Nanoparticles Arbitrated by Curcumin in an Alkaline Medium. Molecules. 2019;24(4):719.
- [CrossRef] [Google Scholar]
- Synthesis, characterization and studies on antioxidant activity of silver nanoparticles using Elephantopus scaber leaf extract. Mater. Sci. Eng. C.. 2016;62:719-724.
- [CrossRef] [Google Scholar]
- Historical review of the use of silver in the treatment of burns. I. Early uses. Burns.. 2000;26(2):117-130.
- [CrossRef] [Google Scholar]
- Silver-based crystalline nanoparticles, microbially fabricated. Proc. Natl. Acad. Sci. USA. 1999;96(24):13611-13614.
- [CrossRef] [Google Scholar]
- Green synthesis and characterization of silver nanoparticles produced using Arbutus Unedo leaf extract. Mater. Lett.. 2012;76:18-20.
- [CrossRef] [Google Scholar]
- High conversion synthesis of <10 nm starch-stabilized silver nanoparticles using microwave technology. Sci. Rep.. 2018;8:5106.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nano particles using Andean blackberry fruit extract. Saudi. J. Bio.l Sci.. 2017;24(1):45-50.
- [CrossRef] [Google Scholar]
- Characterization of magnetic nanoparticle by dynamic light scattering. Nanoscale Res. Lett.. 2013;8:381.
- [CrossRef] [Google Scholar]
- Application of gold-nanoparticle colorimetric sensing to rapid food safety screening. Sensors.. 2018;18(12):4166.
- [Google Scholar]
- Synthesis of biogenic silver nanoparticles from the seed coat waste of pistachio (Pistacia vera) and their effect on the growth of eggplant. Nanotechnol. Rev.. 2021;10(1):1789-1800.
- [CrossRef] [Google Scholar]
- Biosynthesis of silver nanoparticle using flowers of Calotropis gigantea (L.) W.T. Aiton and activity against pathogenic bacteria. Arab. J. Chem.. 2020;13(12):9139-9144.
- [CrossRef] [Google Scholar]
- Bio-synthesis of silver nanoparticles using Potentilla fulgens Wall. ex Hook. and its therapeutic evaluation as anticancer and antimicrobial agent. Mater. Sci. Eng. C.. 2015;53:120-127.
- [Google Scholar]
- Biosynthesis of silver nanoparticles using Ligustrum ovalifolium fruits and their cytotoxic effects. J. Nanomater.. 2018;8(8):627.
- [CrossRef] [Google Scholar]
- Investigation of biopolymer electrolyte based on celluloseacetate-ammoniumnitrate for potential use in electrochemical devices. Carbohydr. Polym.. 2017;157:38-47.
- [CrossRef] [Google Scholar]
- Surface plasmon spectroscopy of nanosized metal particles. Langmuir. 1996;12:788-800.
- [CrossRef] [Google Scholar]
- Nanoparticles in modern medicine: State of the art and future challenges. Int. J. Nanomedicine.. 2007;2(2):129-141.
- [Google Scholar]
- Synthesis and potential applications of lipid nanoparticles in medicine. Materials.. 2022;15:682.
- [Google Scholar]
- Coalescence of nanoclusters and formation of submicron crystallites assisted by Lactobacillus strains. Cryst. Growth Des.. 2002;2(4):293-298.
- [CrossRef] [Google Scholar]
- Synthesis and application of silver nanoparticles (Ag NPs) for the prevention of infection in healthcare workers. Int. J. Mol. Sci.. 2019;20(15):3620.
- [CrossRef] [Google Scholar]
- Green synthesis and characterisation of silver nanoparticles using Cassia tora seed extract and investigation of antibacterial potential. Appl. Biochem. Biotechnol.. 2022;194(1):464-478.
- [CrossRef] [Google Scholar]
- Nanoparticles for biomedical imaging. Expert Opin. Drug. Deliv.. 2009;6(11):1175-1194.
- [Google Scholar]
- Green synthesis of silver nanoparticles from marigold flower and its synergistic antimicrobial potential. Arab. J. Chem.. 2015;8(5):732-741.
- [CrossRef] [Google Scholar]
- Rapid decolorization of synthetic melanoidin by bacterial extract and their mediated silver nanoparticles as support. J. Appl. Biol. Biotechnol.. 2015;3(2):006-011.
- [CrossRef] [Google Scholar]
- Effect of NaOH and precursor concentration on size and magnetic properties of FeCo nanoparticles synthesized using polyol method. AIP Adv.. 2020;10:115220
- [CrossRef] [Google Scholar]
- Targeted delivery of gemcitabine to pancreatic adenocarcinoma using Cetuximab as a targeting agent. Cancer Res.. 2008;68(6):1970-1978.
- [CrossRef] [Google Scholar]
- Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnology.. 2018;16(1):71.
- [CrossRef] [Google Scholar]
- Role of nanotechnology in electronics: A review of recent developments and patents. Recent Pat. Nanotechnol.. 2022;16(1):45-66.
- [CrossRef] [Google Scholar]
- Nano sand filter with functionalized nanoparticles embedded in anodic aluminum oxide templates. Sci. Rep.. 2016;6:37673.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles using the plant extract of Salvia spinosa grown in vitro and their antibacterial activity assessment. J. Nanostructure Chem.. 2019;9:1-9.
- [CrossRef] [Google Scholar]
- How interdisciplinary is nanotechnology? J. Nanopart. Res.. 2009;11:1023-1041.
- [CrossRef] [Google Scholar]
- Potential antibacterial mechanism of silver nanoparticles and the optimization of orthopedic implants by advanced modification technologies. Int. J. Nanomedicine.. 2018;13:3311-3327.
- [CrossRef] [Google Scholar]
- Magnetically responsive antibacterial nanocrystalline jute cellulose nano composites with moderate catalytic activity. Carbohydr. Polym.. 2020;251:117024
- [CrossRef] [Google Scholar]
- Achiote (Bixa orellana L.): A natural source of pigment and vitamin E. J. Food Sci. Technol.. 2017;54(6):1729-1741.
- [CrossRef] [Google Scholar]
- Synthesis of silver nanoparticles using fresh bark of Pongamia pinnata and characterization of its antibacterial activity against gram positive and gram negative pathogens. Resource-Efficient Technologies.. 2016;2(1):30-35.
- [CrossRef] [Google Scholar]
- Completely green synthesis and stabilization of metal nanoparticles. J. Am. Chem. Soc.. 2003;125:13940-13941.
- [CrossRef] [Google Scholar]
- Carotenoid derivates in Achiote (Bixa orellana) seeds: Synthesis and health promoting properties. Front. Plant Sci.. 2016;7:1406.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles using Azadirachta indica leaf extract and its antimicrobial study. Appl. Nanosci.. 2017;7:843-850.
- [CrossRef] [Google Scholar]
- Potential applications of engineered nanoparticles in medicine and biology: An update. J. Biol. Inorg. Chem.. 2018;23:1185-1204.
- [Google Scholar]
- Gold nanoparticles in chemical and biological sensing. Chem. Rev.. 2012;112(5):2739-2779.
- [Google Scholar]
- In-vitro evaluation of antioxidant and antibacterial potential of green synthesized silver nanoparticles using Prosopis farcta fruit extract. Iran J. Pharm. Sci.. 2019;18(1):430-445.
- [Google Scholar]
- Applications of nanoparticles in biology and medicine. J. Nanobiotechnol.. 2004;2:3.
- [Google Scholar]
- Green synthesis of silver nanoparticles with antibacterial activity using various medicinal plant extracts: Morphology and antibacterial efficacy. Nanomaterials.. 2021;11(4):1005.
- [CrossRef] [Google Scholar]
- Biosynthesis of silver nanoparticles from date seeds extract and evaluation of antibacterial activity against pathogenic bacteria. Orient. J. Chem.. 2020;36(6):1189-1193.
- [Google Scholar]
- Green biosynthesis of silver nanoparticles using Curcuma longa tuber powder. Int. J. Nanomedicine.. 2012;7:5603-5610.
- [CrossRef] [Google Scholar]
- Antioxidant activity in vitro of selenium-contained protein from the Se-enriched Bifodobacterium animalis 01. Anaerobe.. 2010;16(4):380-386.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles using Artocarpus hirsutus seed extract and its antibacterial activity. Curr. Pharm. Biotechnol.. 2020;21(10):980-989.
- [CrossRef] [Google Scholar]
- Biodegradable polymeric nanoparticles as drug delivery devices. J. Control Release.. 2001;70(1–2):1-20.
- [CrossRef] [Google Scholar]
- Green Strategy-Based Synthesis of Silver Nanoparticles for Antibacterial Applications. Front. Nanotechnol.. 2021;3:697303
- [CrossRef] [Google Scholar]
- Nanocrystalline silver dressings as an efficient anti-MRSA barrier: A new solution to an increasing problem. J. Hosp. Infect.. 2005;60(3):226-230.
- [CrossRef] [Google Scholar]
- An efficient photo catalytic activity of green synthesized silver nanoparticles using Salvadora persica stem extract. Sep. Purif. Technol.. 2015;150:316-324. 10.1016%2Fj.seppur.2015.07.012
- [Google Scholar]
- Nanotechnology: Interdisciplinary science of applications. Afr. J. Biotechnol.. 2013;12:219-226.
- [CrossRef] [Google Scholar]
- Catalysis with carbon nanoparticles. RSC Adv.. 2019;9(47):27659-27664.
- [CrossRef]
- Enzyme-mediated formulation of stable elliptical silver nanoparticles tested against clinical pathogens and MDR bacteria and development of antimicrobial surgical thread. Ann. Clin. Microbiol. Antimicrob.. 2017;16:39.
- [CrossRef] [Google Scholar]
- Antioxidant activity of nanomaterials. J. Mater. Chem. B.. 2018;6:2036-2051.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles using plant extracts and their antimicrobial activities: A review of recent literature. RSC Adv.. 2021;11(5):2804-2837.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles using aqueous rhizome extract of Zingiber officinale and Curcuma longa: In-vitro anti-cancer potential on human colon carcinoma HT-29 cells. Saudi J. Biol. Sci.. 2020;27(11):2980-2986.
- [Google Scholar]
- Biosynthesized silver nanoparticles by aqueous stem extract of Entada spiralis and screening of their biomedical activity. Front. Chem.. 2020;8:620.
- [CrossRef] [Google Scholar]
- Nanoparticles as drug delivery systems. Pharmacol. Rep.. 2012;64(5):1020-1037.
- [CrossRef] [Google Scholar]
- Growing metal nanoparticles by enzymes. Adv. Mater.. 2006;18(9):1109-1120.
- [CrossRef] [Google Scholar]
- The synthesis of nickel nanoparticles by hydrazine reduction. Adv. Powder Technol.. 2010;21(2):165-168.
- [CrossRef] [Google Scholar]
- The Use of Medicinal Plant-Derived Metallic Nanoparticles in Theranostics. Pharmaceutics. 2022;14:2437.
- [CrossRef] [Google Scholar]
- Nanoparticle-based drug delivery in cancer therapy and its role in overcoming drug resistance. Front. Mol. Biosci.. 2020;7:193.
- [CrossRef] [Google Scholar]
- Nanoparticles in medicine: Therapeutic applications and developments. Clin. Pharmacol. Ther.. 2008;83(5):761-769.
- [Google Scholar]
- Silver Nanoparticles: Synthesis, Characterization, Properties, Applications, and Therapeutic Approaches. Int. J. Mol. Sci.. 2016;17(9):1534.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.104675.
Appendix A
Supplementary data
The following are the Supplementary data to this article: