Translate this page into:
Ceria doped mixed metal oxide nanoparticles as oxidation catalysts: Synthesis and their characterization
⁎Corresponding author. sfadil@ksu.edu.sa (S.F. Adil)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Mixed metal nanoparticles (NPs) have attracted significant attention as catalysts for various organic transformations. In this study, we have demonstrated the preparation of nickel–manganese mixed metal oxide NPs doped with X% nano cerium oxide (X = 1, 3, 5 mol%) by a facile co-precipitation technique using surfactant and surfactant free methodologies. The as-synthesized materials were calcined at different temperatures (300 °C, 400 °C, and 500 °C), and were characterized using various spectroscopic techniques, including, FTIR and XRD. SEM analysis, TEM analysis and TGA were employed to evaluate the structural properties of the as-prepared catalyst. These were evaluated for their catalytic behaviour towards the conversion of benzyl alcohol to benzaldehyde, which was used as a model reaction with molecular oxygen as oxidant. Furthermore, the effect of the variation of the percentage of nano ceria doping and the calcination temperature on the performance of as-prepared mixed metal catalysts was also evaluated. The kinetic studies of the reactions performed employing gas chromatographic technique have revealed that the mixed metal oxide catalyst doped with 5% nano ceria displayed excellent catalytc activity, among various catalysts synthesized.
Keywords
Catalysis
Ceria nanoparticles
Mixed metal oxide
Nickel manganese oxide
Benzyl alcohol oxidation
1 Introduction
Oxidation catalysis is one of the most important subjects for the scientists around the world due to its evident role played in the industrial process for the production of high value chemicals. Among various oxidation reactions, oxidation of alcohols has been studied extensively, which commonly leads to the formation of industrially important starting materials, including aldehydes and ketones. A variety of transition metals and metal oxides based catalysts have been reported for the catalytic oxidation of alcohols, among them mixed metal oxide nanocatalysts were found to be very effective, which have exhibited excellent catalytic activities for such reactions. Furthermore, it has been widely reported that the catalytic activity of mixed metal oxide nanocatalysts enhanced significantly upon doping with other elements (Fu et al., 2003; Siddiqui et al., 2012; Adil et al., 2015; Alabbad et al., 2013).
Variety of elements have been applied as dopants, among them cerium doped compounds have also been found in the literature. For instance, ceria doped Mn/TiO2 nanocatalyst was used for the selective catalytic reduction of NO with NH3. The as-prepared catalyst was found to be resistant to SO2 poisoning (Wu et al., 2009). In other example, Ce-doped SnO2 thin films have been used for the photoluminescence studies, which were found to be highly sensitive toward the selective sensing of butanone (Chen et al., 2012; Jiang et al., 2010). Similarly, CeO2 based catalyst was found to be fast degradation agents of pesticides such as parathion methyl and other organophosphates (Janos et al., 2014). Apart from this, the ceria doped mixed metal oxide nanocatalysts have also been applied for various organic transformations. For instance, Pt/ceria-doped catalysts were employed for the partial oxidation of methane (Salazar et al., 2006), and Au/doped-ceria catalysts for the preferential CO oxidation reaction (Avgouropoulos et al., 2008), for WGS reaction (Andreeva et al., 2009) and also for the total oxidation of methane and light hydrocarbons (Terribile et al., 1999).
In our previous study, we have reported the use of nickel manganese mixed metal oxide based catalysts for the catalytic oxidation of benzyl alcohol, using various dopants (Siddiqui et al., 2012; Adil et al., 2015; Alabbad et al., 2013). In the continuation of our work in the area of selective alcohol oxidation to aldehydes using mixed metal oxides, herein, we report the synthesis of nano ceria doped nickel manganese mixed oxide nanocatalysts and their application toward the oxidation of benzyl alcohol as a model compound. For this purpose, various catalysts were prepared by altering the w/w% of ceria doping on nickel manganese mixed oxide, which were characterized by SEM, TEM, XRD, FTIR and TGA. The catalytic activities of as-prepared catalysts were evaluated and the conversions were monitored by gas chromatography.
2 Material and method
2.1 Materials used
Ni(NO3)2⋅6H2O (Nickel Nitrate), Mn(NO3)2⋅4H2O (Manganese Nitrate), Ce(NO3)3⋅6H2O (Cerium Nitrate hexahydrate), NaHCO3 (Sodium bicarbonate), C6H5CH2OH (Benzyl alcohol), C6H5CHO (Benzaldehyde) and C6H5CH3 (Toluene) [Merck grade] were purchased from Sigma Aldrich Pure Chemical Industries Co., Ltd. They were used as received without further purification.
2.2 Synthesis of ceria doped on nickel manganese mixed oxide [CeO2 (X%)–NiMnO]
In a typical approach to synthesize the X% (X = 1, 3, 5 mol%) ceria doped nickel–manganese mixed oxide stoichiometric volume of 0.2 M solution of nickel nitrate and manganese nitrate were mixed in a round bottom flask, then it was added with desired volume of 0.2 M solution of cerium nitrate solution. The resulting solution was heated to 90 °C, while stirring using a mechanical stirrer. To this solution, 1 M solution of NaHCO3 was added dropwise until the solution attained a pH of 9. The solution was continued to stir at the same temperature for about 3 h and then left for stirring over night at room temperature. The solution was filtered using a Buchner funnel under vacuum and then dried at 70 °C overnight. The resulting product obtained was then calcined at different temperatures i.e. 300 °C, 400 °C, 500 °C which were characterized by various analytical techniques including, SEM, TEM, FTIR, and XRD in order to obtain structural information of the product.
2.3 Catalyst testing
In a typical reaction run, 300 mg of calcined catalyst was loaded in a glass flask pre-charged with 0.2 ml (2 mmol) benzyl alcohol with 10 ml toluene as solvent; the mixture was then refluxed at 100 °C along with vigorous stirring. Oxygen was bubbled at a flow rate of 20 ml min−1 into the mixture once the reaction temperature was attained. After reaction, the solid catalyst was separated by centrifugation and the liquid samples were analyzed by gas chromatography to evaluate the conversion of the desired product by (GC, 7890A) Agilent Technologies Inc., equipped with a flame ionization detector (FID) and a 19019S-001 HP-PONA capillary column.
3 Result and discussion
3.1 Catalyst characterization
The morphology of nanoparticles and elemental analysis was determined using Scanning Electron Microscopy (SEM) Jeol SEM model JSM 6360A (Japan). Transmission Electron Microscopy (TEM) was carried out using Jeol TEM model JEM-1101 (Japan) which was used to determine the shape and size of nanoparticles. Powder X-ray diffraction studies were carried out using a D2 phaser (Bruker) X-ray diffractometer. Thermogravimetric Analysis (TGA) was carried out using Perkin–Elmer Thermogravimetric Analyzer 7. Fourier transform infrared spectroscopy (FT-IR) was used to see the presence of functional groups.
3.1.1 Microscopic and spectroscopic characterization of the catalysts
3.1.1.1 Scanning Electron Microscopy (SEM)
The SEM images of all the synthesized catalysts were measured and it was revealed that the catalysts were devoid of distinct features, pointing to the absence of well-defined morphology. There is no change in the surface morphology when calcined at different temperatures i.e. 300 °C, 400 °C, 500 °C as observed in Fig. 1.
SEM images of CeO2 (5%)–NiMnO calcined at different temperatures (a) 300 °C, (b) 400 °C and (c) 500 °C.
3.1.1.2 Transmission Electron Microscopy (TEM)
The as prepared catalyst calcined at different temperatures was subjected to transmission electron microscopy in order to ascertain the nanoparticle size formed. It was found that the particles formed were in varying sizes from 6 to 12 nm. However there was no correlation found between the particle size and conversion rate, signifying that there must be other factors governing the catalytic performance of the catalyst. The TEM image for the catalyst CeO2 (5%)–NiMnO calcined at temperature 400 °C is given in Fig. 2.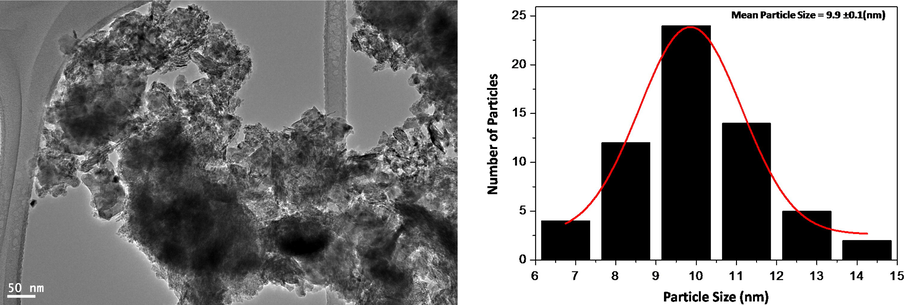
TEM images of CeO2 (5%)–NiMnO calcined at 400 °C and particle size distribution graph.
3.1.1.3 X-ray Diffraction (XRD)
The XRD analysis of the prepared catalyst was carried out and it confirmed that the mixed metal oxides formed were crystalline in nature. Different patterns of the diffractograms have been obtained which indicates the formation of different phases of the mixed metal oxides. The XRD patterns obtained are given in Fig. 3. The phase identification and crystallite size calculation will be carried out and shall be reported later.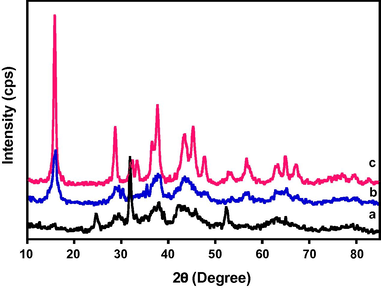
XRD spectra of the catalyst CeO2 (5%)–NiMnO calcined at different temperatures (a) 300 °C, (b) 400 °C and (c) 500 °C.
3.1.1.4 FT-IR
Fig. 4 shows FT-IR spectra of the synthesized catalysts at different calcination temperatures (300, 400 and 500 °C). High wave number region reveals stretching vibrations of the hydroxyl groups. The characteristic bands of ν OH, located at approximately 3450 cm−1 were found to be present in all prepared catalyst. The decrease in the intensity of these bands with an increase of the calcination temperature depicts the decrease of hydroxyl group on the catalyst. Similar trends are visible for the bands located at around 1650 cm−1, identified as fingerprint region for bending vibrations of the hydroxyl groups (Gac, 2007). Significant peaks are observed in the range of 400–700 cm−1 typical of different oxides of manganese (Dubal et al., 2010; Maslen et al., 1995).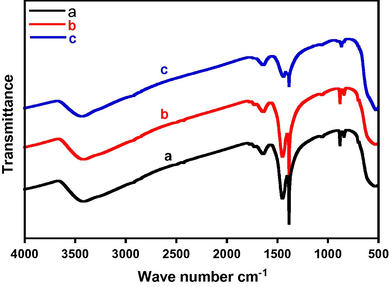
FTIR spectra of the catalyst CeO2 (5%)–NiMnO calcined at different temperatures (a) 300 °C, (b) 400 °C and (c) 500 °C.
3.1.1.5 TGA
The degradation pattern obtained from the thermogravimetric analysis of the catalyst, displayed varied thermal stability. The catalyst calcined at 300 °C displayed a ∼20% weight loss, while the 400 °C showed a weight loss of ∼15%. The catalyst with 500 °C displayed best thermal stability with <10% weight loss. The degradation pattern is given in Fig. 5.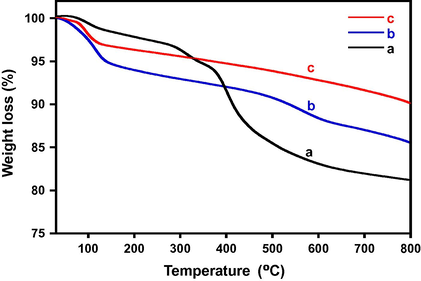
Thermal stability pattern of the CeO2 (5%)–NiMnO catalyst calcined at different temperatures (a) 300 °C, (b) 400 °C and (c) 500 °C.
3.2 Catalytic evaluation as oxidation catalysts
In order to determine the catalytic oxidation properties of synthesized catalysts, oxidation of benzyl alcohol was used as a model reaction and the reaction was carried out in the presence of the as prepared catalyst. The percentage of ceria doping and the calcination temperature was varied, in order to confirm the optimum % of ceria doping required and the calcination temperature for the best catalytic performance of the synthesized catalyst. The reaction was carried out for 5 h and different results were obtained as mentioned below. A graphical illustration of the results obtained is given in Fig. 6.![Graphical representation of time vs conversion (%) [A] for the catalyst CeO2 (5%)–NiMnO catalyst calcined at different temperatures (a) 300 °C, (b) 400 °C and (c) 500 °C [B] CeO2 (X%)–NiMnO catalyst calcined at 400 °C wherein X = (a) 1%, (b) 3% and (c) 5%.](/content/184/2015/8/6/img/10.1016_j.arabjc.2015.05.008-fig6.png)
Graphical representation of time vs conversion (%) [A] for the catalyst CeO2 (5%)–NiMnO catalyst calcined at different temperatures (a) 300 °C, (b) 400 °C and (c) 500 °C [B] CeO2 (X%)–NiMnO catalyst calcined at 400 °C wherein X = (a) 1%, (b) 3% and (c) 5%.
3.2.1 CeO2 (1%)–NiMnO catalyst
When 1% CeO2 doped NiMnO catalyst calcined at different temperatures was used, a 33% and 30% conversion product was obtained by using the catalyst calcined at 300 °C and 500 °C respectively. The 400 °C calcined catalyst yielded a 77% conversion product.
3.2.2 CeO2 (3%)–NiMnO catalyst
When 3% CeO2 doped NiMnO catalyst calcined at different temperatures was used, 35%, 83% and 33% conversion product was obtained by using catalyst calcined at 300 °C, 400 °C and 500 °C respectively.
3.2.3 CeO2 (5%)–NiMnO catalyst
When 5% CeO2 doped NiMnO catalyst calcined at 400 °C was used, a 100% conversion product was obtained while using catalyst calcined at 300 °C and 500 °C yielded 37% and 35% to the conversion product respectively.
From the above mentioned results it was evident that the calcination temperature played an important role in the catalytic performance of prepared catalyst and a direct relation between percentage of ceria in the catalyst and its catalytic performance could be observed, thus confirming that ceria nanoparticles play the role of a promoter. It can be concluded that the 400 °C calcination temperature and 5% doping of ceria nanoparticles is the best combination for the highest catalytic performance.
4 Conclusions
Ceria doped nickel manganese oxide exhibits high activity and stability for the oxidation of benzyl alcohol using molecular oxygen as a source of oxygen. A synergistic effect between calcination temperatures and the chemical kinetics of the reaction was observed and it was confirmed that calcination temperature plays a vital role forming an active and durable catalyst. Among different percentages of ceria doped catalyst, CeO2 (5%)–NiMnO gave the 100% conversion from benzyl alcohol to benzaldehyde at 400 °C. Hence, this catalyst can be considered the best among the synthesized catalysts and its performance toward various aromatic and aliphatic alcohols is being explored and shall be reported later as a full paper.
Acknowledgement
This project was supported by King Saud University, Deanship of Scientific Research, College of Science, Research Center.
References
- Vanadia supported on nickel manganese oxide nanocatalysts for the catalytic oxidation of aromatic alcohols. Nanoscale Res. Lett.. 2015;10(52):1-9.
- [Google Scholar]
- Liquid phase selective oxidation of aromatic alcohols employing nanoparticles of zirconia supported on nickel manganese oxide: synthesis, characterization and catalytic evaluation. Asian J. Chem.. 2013;25(16):8927-8932.
- [Google Scholar]
- Gold catalysts supported on ceria doped by rare earth metals for water gas shift reaction: influence of the preparation method. Appl. Catal., A. 2009;357:159-169.
- [Google Scholar]
- Catalytic performance and characterization of Au/doped-ceria catalysts for the preferential CO oxidation reaction. J. Catal.. 2008;256:237-247.
- [Google Scholar]
- Photoluminescence of undoped and Ce-doped SnO2 thin films deposited by sol–gel-dip-coating method. Appl. Surf. Sci.. 2012;258:3255-3259.
- [Google Scholar]
- A novel chemical synthesis and characterization of Mn3O4 thin films for supercapacitor application. Appl. Surf. Sci.. 2010;256:4411-4416.
- [Google Scholar]
- Active nonmetallic Au and Pt species on ceria-based water–gas shift catalysts. Science. 2003;301:935-938.
- [Google Scholar]
- The influence of silver on the structural, redox and catalytic properties of the cryptomelane-type manganese oxides in the low-temperature CO oxidation reaction. Appl. Catal., B. 2007;75:107-117.
- [Google Scholar]
- Cerium dioxide as a new reactive sorbent for fast degradation of parathion methyl and some other organophosphates. J. Rare Earth. 2014;32:360-370.
- [Google Scholar]
- Highly sensitive and selective butanone sensors based on cerium-doped SnO2 thin films. Sens. Actuators, B. 2010;145:667-673.
- [Google Scholar]
- Electron density and optical anisotropy in rhombohedral carbonates. III. Synchrotron X-ray studies of CaCO3, MgCO3 and MnCO3. Acta Crystallogr. B. 1995;B51:929-939.
- [Google Scholar]
- Catalytic partial oxidation of methane over Pt/ceria-doped catalysts: effect of ionic conductivity. Appl. Catal., A. 2006;310:54-60.
- [Google Scholar]
- Nano-gold supported nickel manganese oxide: synthesis, characterisation and evaluation as oxidation catalyst. Oxid. Commun.. 2012;35:476-481.
- [Google Scholar]
- Catalytic combustion of hydrocarbons with Mn and Cu-doped ceria ± zirconia solid solutions. Catal. Today. 1999;47:133-140.
- [Google Scholar]
- Effect of ceria doping on SO2 resistance of Mn/TiO2 for selective catalytic reduction of NO with NH3 at low temperature. Catal. Commun.. 2009;10:935-939.
- [Google Scholar]







