Translate this page into:
Concentration of ochratoxin A in coffee products and probabilistic health risk assessment
⁎Corresponding authors. sadighara@farabi.tums.ac.ir (Parisa Sadighara), Mahmood_yousefi70@yahoo.com (Mahmood Yousefi)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Coffee is a beverage that people enjoy a lot in their daily lives and is an integral part of people's social life. In this study, the detection of ochratoxin A (OTA) in coffee was carried out by High-performance liquid chromatography with fluorescence (HPLC-FLD) detectors. Furthermore, the amount of ochratoxin A (OTA), Margin of exposure (MOEs), and Hazard quotient (HQ) in different types of coffees; instant, classic, and roasted coffee were calculated using Monte Carlo simulation (MCS) method. The average OTA concentration was in the rage of 3.6 to 26.6 µg/kg. The content of classic and instant coffee found to have.
OTA, is below the maximum limit defined by the European Union legislation. The maximum limit for these two types of coffee is 10 µg/kg. The daily intakes of the OTA through classic and instant coffee were also found to be lower than the Tolerance daily intake proposed by Joint FAO/WHO Expert Committee on Food Additives (JECFA). MOEs (neoplastic effect) in adults was classic coffee (171026) > roasted coffee (15390) > instant coffee (8549) and also MOEs (non-neoplastic effects) in children was classic coffee (55790) > roasted coffee (5020) > instant coffee (2789). Consumers of instant coffee are at cancer risk based on neoplastic effects and also consumers of instant coffee and roasted coffee are at cancer risk based on non-neoplastic effects (MOEs lower than 10,000 value). HQ (nephrotoxic effect) in adults was instant coffee (0.132) > roasted coffee (0.097) > classic coffee (0.012). HQ due to consumption of coffee products was lower than 1, hence consumers are at safe non-cancer risk. Therefore, it is recommended reducing the concentration of mycotoxins in coffee products.
Keywords
Coffee products
HPLC
Mycotoxins
Ochratoxin A
Risk Assessment
1 Introduction
Human exposure to contaminated food is a global concern that necessitates food safety assessments. Ochratoxin A (OTA) is considered the second major group of mycotoxins after aflatoxin(Solfrizzo et al., 2009, Woo and El-Nezami 2015, Mokhtarian et al., 2020). OTA is a nephrotoxin produced by Aspergillus and Penicillium species with carcinogenicity in the kidneys (Sorrenti et al., 2013). Other toxic effects, including hepatotoxicity, teratogenic and immune toxicity have been reported with OTA(Solfrizzo et al., 2013, Podolska et al., 2017). The human exposure is usually through food. Its half-life is also relatively long in the body (Heussner and Bingle 2015).
Coffee is one of the major beverages consumed worldwide. The coffee is rich in antioxidant component including flavonoids content. It reduces the risk of some diseases including diabetes and cardiovascular disease (Malir et al., 2014, Karami et al., 2022). Coffee is very valuable but can be exposed to various pest and fungi (Paterson et al., 2014). The most important factor limiting the coffee trade in the world is its contamination with OTA. Extensive studies have shown that about 10 % of coffees are contaminated with OTA (Vatinno et al., 2008). OTA in coffee is usually produced by Aspergillus Flavi, Aspergillus Nigri, and Aspergillus Circumdati (Tozlovanu and Pfohl-Leszkowicz 2010). These strain is distributed worldwide (Cabañes and Bragulat 2018). This strain is able to contaminate coffee beans during the drying process and is a source of OTA in green coffee beans. In order to reduce ochratoxin, the drying process should be done on the same day of harvest. Before drying, the coffee beans need to be sorted. The amount of active water in coffee beans should be controlled during their storage(Estrada‐Bahena et al., 2022). Damaged and immature beans are also removed (Poltronieri and Rossi 2016). To control this toxin, it is necessary to evaluate the drying processes of coffee beans. In a study that measured various mycotoxins, including Ochratoxin A, Aflatoxin B1, Fumonisin B2, Sterigmatocystin, Beauvericin and Enniatin A in coffee beans, the amount of Ochratoxin A was higher than other mycotoxins (Bessaire et al., 2019). The previous studies show that 12 % of ochratoxin A exposure is caused by drinking coffee (Sueck et al., 2019).
OTA has been identified in both roasted and green coffees. It is necessary to regularly measure the amount of this toxin in coffee samples to evaluate good agricultural methods and post-harvest handling (Malir ret al., 2014). Risk assessment of contaminants and toxins is a tool to identify different people in the community who are at risk of potential food hazards(Grajewski et al., 2019). So far; no comprehensive study has been done on the amount of OTA in a variety of instant coffee products in Iran. The aim of this study was to evaluate the amount of OTA and estimate carcinogenic and noncarcinogenic risk in various type coffee products.
2 Material and method
2.1 Reagents and materials
HPLC grade water was purchased from Merck (Darmstadt, Germany). Extra pure and HPLC grade acetonitrile, acetic acid and other chemicals were obtained from Samchun (Seoul, Republic of Korea). Ochratoxin A standard were purchased from the Sigma–Aldrich (Seelze, Germany) and Immunoaffinity Column for Ochratoxin A was purchased from aoki immunoclean (Berlin, Germany).
2.2 Coffee samples
20 samples of high-consumption brands of instant coffee, 10 samples of classic coffee and 10 samples of ground roasted coffee powder offered in the markets of Tehran/Iran were randomly selected and purchased from different stores. The purchased samples are coded and transferred to the laboratory in appropriate conditions.
2.3 Preparation of samples
The technique for extraction, cleanup and determination of ochratoxin A was based on method EN 14132:2003 [X]. The classic coffee was ground in a laboratory mill and mixed. Twenty-five grams of the milled sample was mixed with 1 g of sodium chloride. Then, 100 ml of extraction solvent (acetonitrile and distillated water (84:16 v/v) was added and shake for 30 min. The extract was passed through filter paper. Ten milliliters of the resulting extract were diluted with 50 ml of Phosphate-buffered saline (PBS) buffer (pH = 7.4) and cleaned up with aoki immunoclean Ochratoxin A columns. First, bring the immunoaffinity column with ochratoxin A antibody to room temperature and prepare by passing 10 ml of PBS buffer. Pass 55 ml of the diluted extraction solution through the prepared column at a rate of 3 ml/min. Wash the column with 10 to 20 ml of PBS and then dry it with positive air pressure. Then wash the column with 1.5 ml of methanol-acetic acid mixture, with volume ratio 98:2, and place it in a clean vial, Add 1.5 ml of HPLC water to it and mix. 50 μL amounts were injected into the HPLC-FLD system in determination step.
2.4 Apparatus
Chromatographic analysis was carried out using an Agilent HPLC system (Wilmington, USA) equipped with Agilent G1321B fluorescence detector (FLD) with a 1200 series quaternary pump, an Eclipse-XDB-C18 analytical column (250 mm × 4.6 mm, 5 µm) and auto sampler was used. The extract that was passed through immunoaffinity columns containing ochratoxin specific antibodies in preparation step, was injected into the HPLC-FLD for determination. The detector wavelength was set at 333 and 477 nm. The spectral bandwidth was 10 nm for both excitation and emission. The volume of the injection was 50 µL. The mobile phase (acetronitrile/water/acetic acid 49.5: 49.5:1.0 v/v/v) was maintained at a flow rate of 1 ml/min.
2.5 Calibration
The measurement of OTA in food has been validated with a HPLC / FLD(Lobeau et al., 2005). This method is one of the most sensitive methods for measuring OTA (Karami et al., 2019).
Analytical method performance, i. e. limit of detections and quantifications (LODs and LOQs, respectively), linear dynamic ranges (LDRs), extraction recoveries (ER) and intra and inter-day precision (RSDs) were calculated by three spiking level of ochratoxin A in three day. Linearity dynamic ranges (LDR) of 1.5 – 12 μg/kg (R2 = 0.996) obtained for coffee sample (Fig. 1). The LOD was obtained 0.5 μg/kg practically based on signal-to-noise ratio of 3. LOQ was 1.5 μg/kg and RSDs for intra and inter day precision was 1.5 % and 4.2 %, respectively. The obtained relative recovery (RR%) of the OTA from coffee was about 98 %.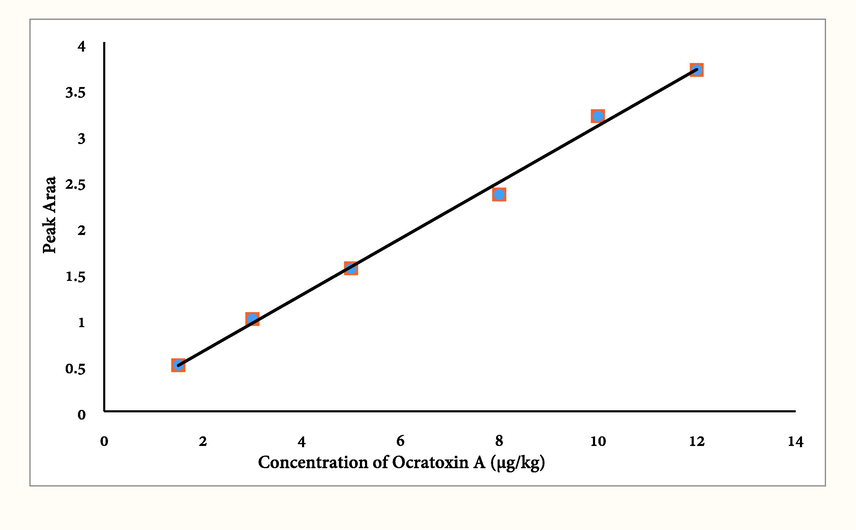
OTA standard curve (1.5, 3, 5, 8, 10, 12 µg/Kg).
2.6 Health risk assessment
The health risk was estimated in adults and children due to ingestion of OTA present in coffee. Health risk of OTA was calculated using equation (1):
Where, CDI is chronic daily intake; C, level of OTA in instant coffee, classic coffee and roasted coffee; IR, ingestion rate of coffee; EF, exposure frequency (350 days/year); ED, exposure duration (70 years); AT, average-life time (for cancer risk is 365 × 70 and for non-cancer risk is ED × 365) and BW, body weight (70 kg). Per capita coffee consumption in adults was calculated through a questionnaire among 450 participants. Ingestion rate for instant coffee, classic coffee and roasted coffee was estimated 15.81 ± 6.34 gr/n-d (range 3.12 to 28.50 gr/n-d); 4.20 ± 1.62 (range 1.07 to 7.50 gr/n-d), and 4.57 ± 2.10 (range 1.21 to 8.32 gr/n-d), respectively. The Margin of Exposures (MOEs) due to OTA in coffee was calculated by Equation (2)(Authority 2020, Sadighara and Ghanati 2022):
BMDL10 is value benchmark dose limit (µg/kg-d); and CDICancer-risk, chronic daily intake (µg/kg-d). The BMDL10 of OTA for neoplastic and non-neoplastic effects is equal to 14.5 µg/kg-d and 4.73 µg/kg-d (EFSA 2020), respectively. When MOEs is less than 10,000, consumers are at health risk (Adetunji et al., 2018, Authority 2020, EFSA 2020).
Hazard quotient (HQ) due to OTA was calculated by Equation (3)(Authority 2020):
HQ is hazard quotient; CDINon-cancer, estimated daily intake (µg/kg-d); and TDI, tolerable daily intake (µg/kg-d). TDI is considered for OTA is equal to 0.017 µg /kg body weight/day based on nephrotoxic in pig (Ostry et al., 2015, EFSA 2017). When HQ is greater than 1 value, consumers are health risk(Chemsafety 2020).
2.7 Uncertainty analysis
In order to decreased uncertainty, Monte Carlo simulation (MCS) method used to calculate risk according to C, IR and BW parameters in the Oracle Crystal Ball software (Ver.11.1.2.4). Estimate of risk was conducted with 5,000 iterations in MCS. Distribution of level of OTA was selected lognormal by fitting analysis and distribution ingestion rate was selected log-normal and body weight was normal distribution (Fakhri et al., 2018, Shahrbabki et al., 2018, Fakhri et al., 2019, Karami et al., 2019, Atamaleki et al., 2020).
2.8 Statistical analysis
Statistical analysis was performed by SPSS version 21. The Mean ± Standard error of the mean (SEM) was calculated. Kolomogorov-smirnov was used for testing normality. All data were nonparametric. Therefore, Kruskal-Wallis test was used to determine the differences between groups.
3 Results and discussion
3.1 Concentration of OTA
A human can expose to mycotoxin with food. Detection of contaminants and their risk assessment in different categories of food and feed improve human health(Mohajer et al., 2021).
In the present study, the level of ocharatoxin A was determined in instant, classic and roasted/ ground coffee. There is a significant difference among amount of OTA between groups (p = 0.007).
Ochratoxin A is the most potent renal carcinogens. A study found that 39 % of mice given very low doses of ochratoxin developed renal cancer (Hajok et al., 2019).The permissible limit of ochratoxin is from 1 to 50 µg/kg for food and from 5 to 300 µg/kg for animal feed (Visconti et al., 1999). According to EU regulations, the permissible level of ochratoxin is 5 µg/kg for roasted coffee and 10 µg/kg for other types of coffee.
Table 1 shows the content of ochratoxin in various types of samples. According to Table 1, OAT is lowest level in classic coffee and highest level in roasted coffee. Mean concentrations of OTA in instant, classic and roasted coffee samples were 10.4 µg/kg, 3.6 µg/kg and 26.6 µg/kg, respectively. In most samples of instant coffee, the amount of OA was less than the allowable limit, but in three samples, the amounts were higher. OTA levels found in roasted coffee in the present study ranged from 2 to 79 μg/ kg.
Type of coffee
Average ± SEM (µg/kg)
MRL EU regulation (µg/kg)
Instant coffee
10.40 ± 3.90
10
Classic coffee
3.60 ± 3.80
10
Roasted coffee
26.60 ± 17.90
5
The amount of OTA in roasted samples was above than allowable range (5 µg/kg). So far, very different amounts of OTA have been reported in coffees, including 79.0 μg/kg in Turkish coffees and 0.35 μg/kg in Taiwanese coffees(Khaneghah et al., 2019). Relatively high levels have been reported in Guatemalan coffees. The level of OTA in coffee from Guatemala were detected 28.4 µg/kg (Sueck et al., 2019). In a comprehensive study conducted in Poland to assess the risk, 43 % of instant coffees were contaminated with OTA (Hajok et al., 2019). In a study, 62 % of instant coffee samples were contaminated with this toxin(Pokrzywa et al., 2022).
Previous studies have shown that the rate of reduction of ochratoxin by roasting is very variable, which can be from zero to 100 % (Vatinno et al., 2008). One study has shown that the roasting process does not reduce the amount of OTA. This type of coffee may be a major source of OTA for humans (Tozlovanu and Pfohl-Leszkowicz 2010). In another study, it was found that it did not even decrease at 250° C (Lobeau et al., 2005). This toxin is stable against processing temperatures (Hajok et al., 2019). Furthermore, these coffees do not have packaging and trademark. The amount of OTA also depends on the season, time and storage conditions (Hajok et al., 2019). Therefore, it is possible that the storage conditions of roasted samples were not suitable. Studies have generally shown that OTA levels are higher in non-trademark coffees (Tozlovanu and Pfohl-Leszkowicz 2010).
3.2 Probabilistic risk assessment in coffee
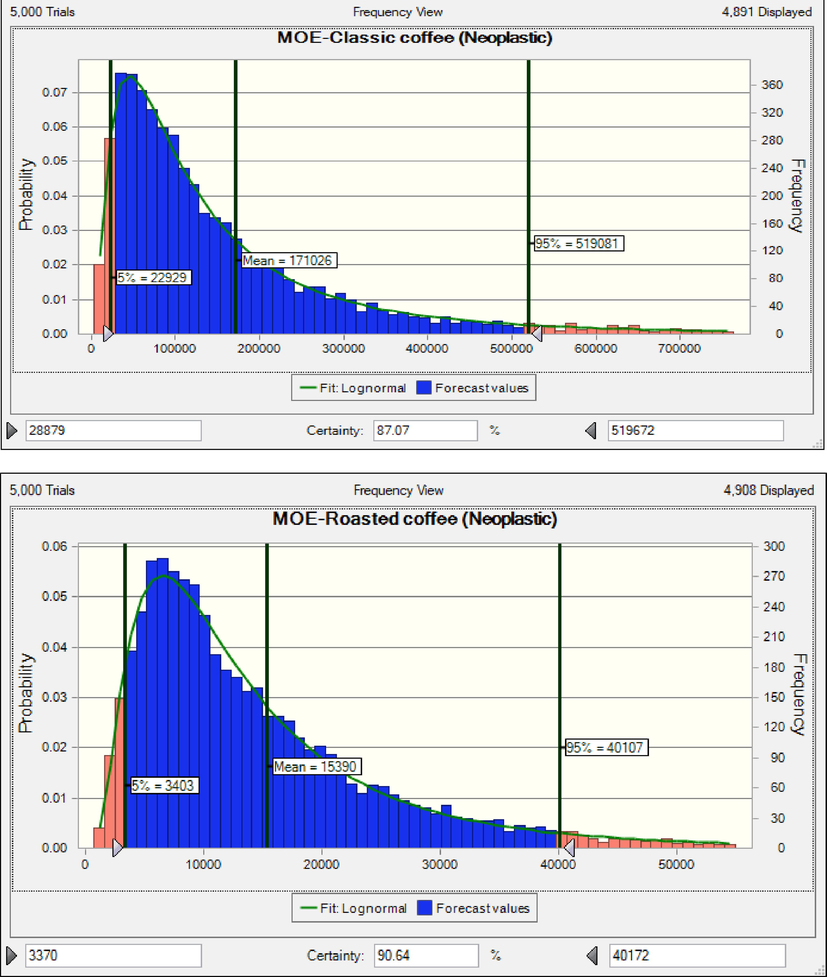
When MOEs are less than 10,000, the exposed population is at carcinogenic health risk(Adetunji et al., 2018, Authority 2020, EFSA 2020). The rank order of coffee products based on MOEs (neoplastic effect) in adults was classic coffee (171026) > roasted coffee (15390) > instant coffee (8549) (Fig. 2).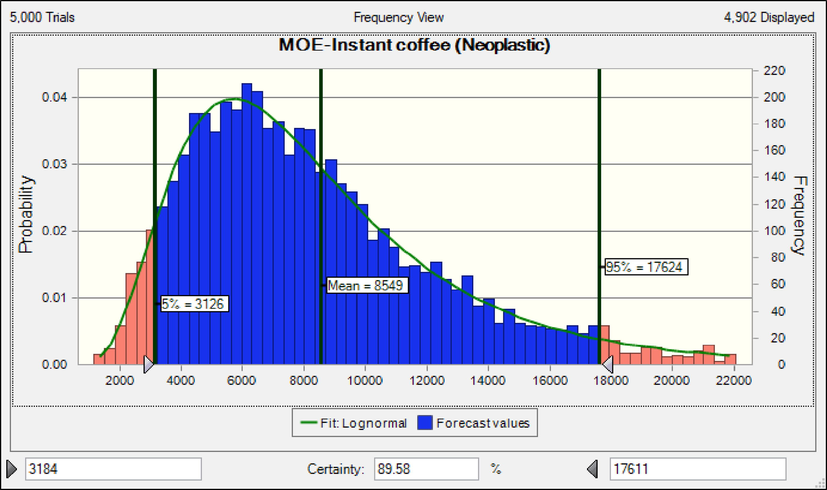
MOEs in adults due to consumption coffee products content of OTA based neoplastic effect.
MOEs in adults due to consumption coffee products content of OTA based neoplastic effect.
The results shows that consumers of instant coffee are at cancer risk based on neoplastic effects. The rank order of coffee products based on MOEs (non-neoplastic effects) in children was classic coffee (55790) > roasted coffee (5020) > instant coffee (2789) (Fig. 3). The results shows that consumers of instant coffee and roasted coffee are at cancer risk based on non-neoplastic effects (Adetunji et al., 2018, Authority 2020, EFSA 2020). When HQ is less than 1 value, consumers are acceptable range risk (EPA 2014). The rank order of coffee products based on HQ (nephrotoxic effect) in adults was instant coffee (0.132) > roasted coffee (0.097) classic coffee (0.012) (Fig. 4). HQ due to consumption of instant coffee, roasted coffee and classic coffee was lower than 1, hence consumers are at safe non-cancer risk. Similar to the results of the present research, the results of the research conducted on Cambodian coffees and Saudi market. The calculated HQ were less than 1(Almasoud et al., 2022, Oeung et al., 2022).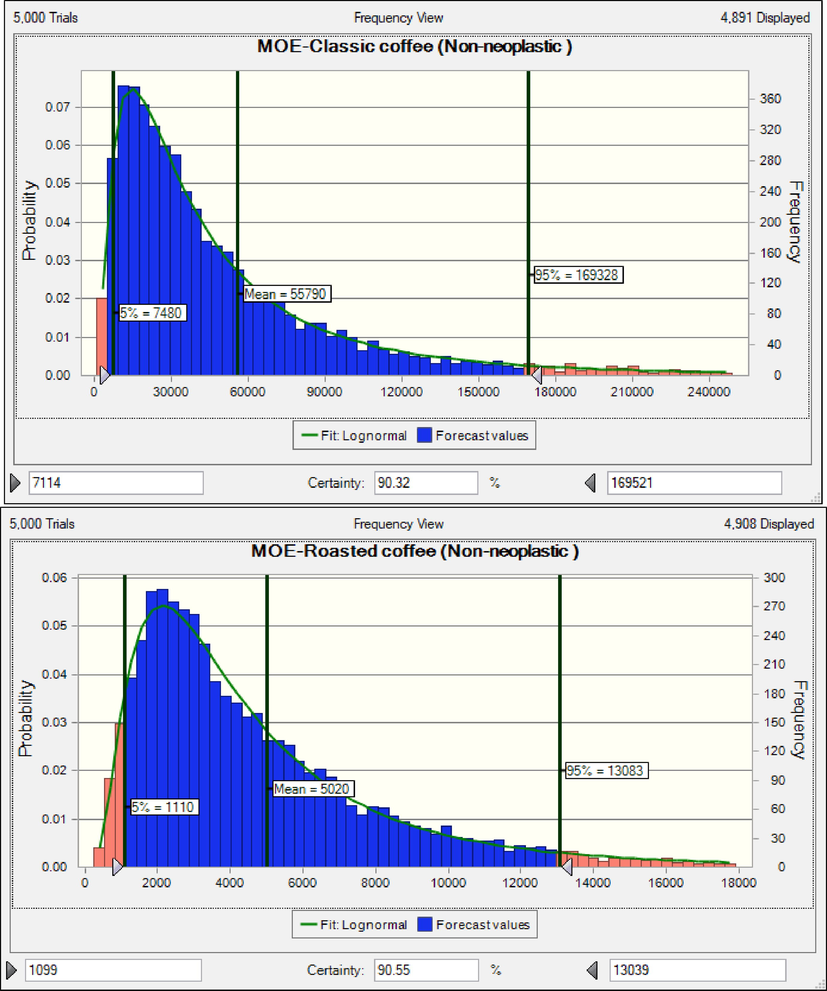
MOEs in children due to consumption coffee products content of OTA based non-neoplastic effect.
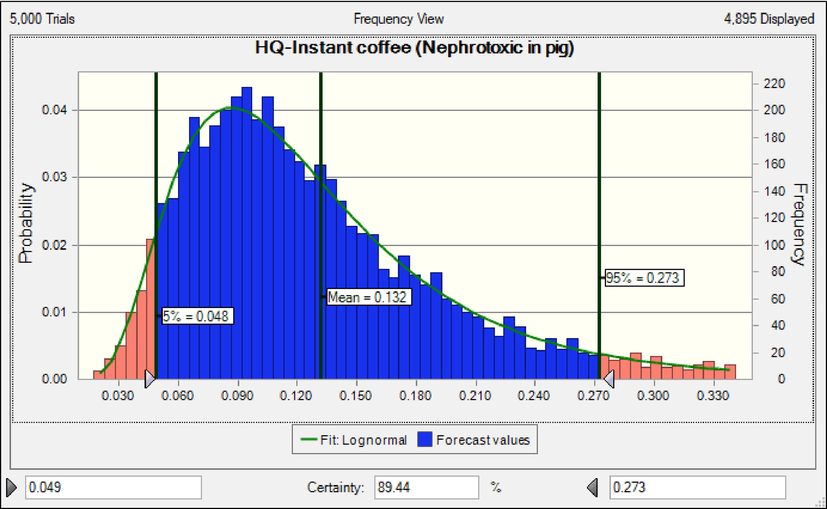
HQ in adults due to consumption coffee products content of OTA based nephrotoxic effect.
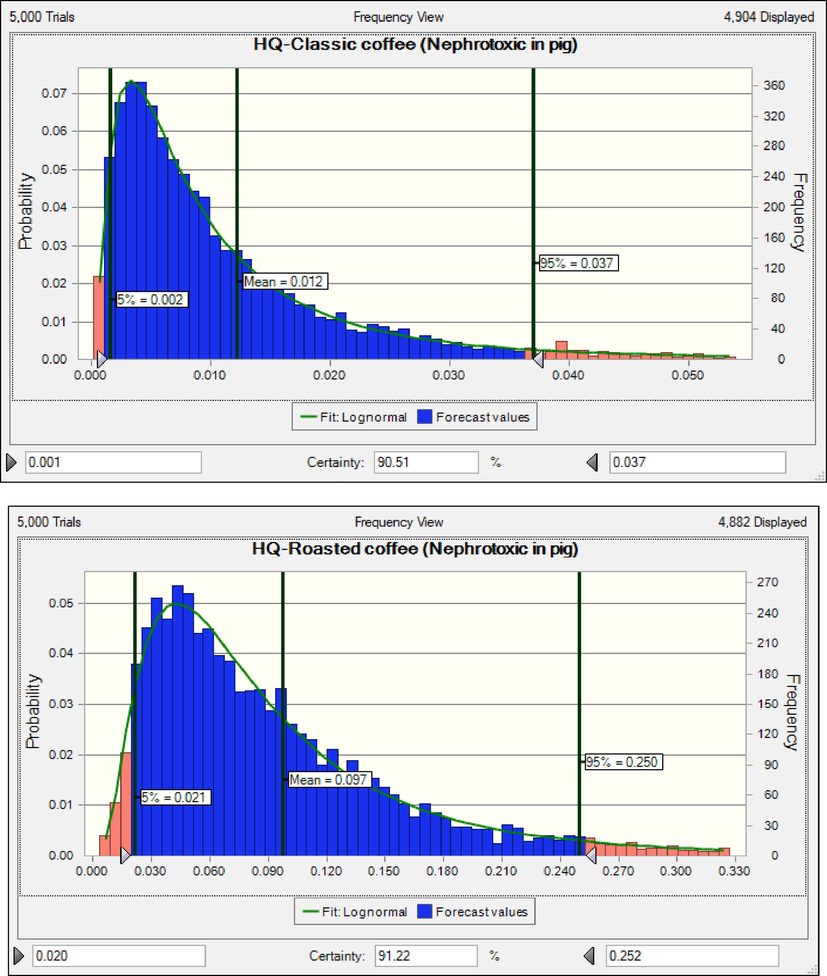
HQ in adults due to consumption coffee products content of OTA based nephrotoxic effect.
4 Conclusion
In this study, the level of OTA in instant, classic and roasted coffee was determined by means of HPLC-fluorescence detection. The level of instant and classic was within the permissible level. The rank order of coffee products based on the concentration of OTA was roasted coffee > instant coffee > classic coffee. The carcinogenic risk assessment based on neoplastic effects revealed that ingestion of instant coffee can have risk for consumers and also based on non-neoplastic effects ingestion of both instant coffee and roasted coffee can have cancer risk for consumers. Although, non-carcinogenic risk assessment based on HQ revealed that consumers are at a safe range of risk. Therefore, it is recommended to reduce its concentration in coffee with proper agricultural and storage methods. Comprehensive research studies in this regard are recommended for the future.
Acknowledgment
We thank the Chemical Analysis Center, Iranian Institute of R&D in Chemical Industries (IRDCI).
Funding
Thanks for financial support of Chemical Analysis Center, Iranian Institute of R&D in Chemical Industries (IRDCI).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Adetunji, M. C., O. P. Alika, N. P. Awa, et al., 2018. Microbiological quality and risk assessment for aflatoxins in groundnuts and roasted cashew nuts meant for human consumption. J. Toxicol. 2018,
- Occurrence of ochratoxin A in green and roasted coffee beans from the Saudi market and a possible exposure assessment. JSFA Rep.. 2022;2:255-260.
- [Google Scholar]
- The concentration of potentially toxic elements (PTEs) in eggs: a global systematic review, meta-analysis and probabilistic health risk assessment. Trends Food Sci. Technol.. 2020;95:1-9.
- [Google Scholar]
- Outcome of a public consultation on the draft risk assessment of aflatoxins in food. Wiley Online Library; 2020.
- Mycotoxins in green coffee: Occurrence and risk assessment. Food Control. 2019;96:59-67.
- [Google Scholar]
- Black aspergilli and ochratoxin A-producing species in foods. Curr. Opin. Food Sci.. 2018;23:1-10.
- [Google Scholar]
- Chemsafety. 2020. How to Calculate Hazard Quotient (HQ) and Risk Quotient (RQ). https://www.chemsafetypro.com/Topics/CRA/How_to_Calculate_Hazard_Quotients_(HQ)_and_Risk_Quotients_(RQ).html#:∼:text=What%20Is%20Hazard%20Quotient%20(HQ,health%20risks%20of%20air%20toxics.
- EFSA, 2020. Risk assessment of ochratoxin A in food. https://efsa.onlinelibrary.wiley.com/doi/epdf/10.2903/j.efsa.2020.6113. EFSA J. 18, 9-17
- EFSA, 2020. SCIENTIFIC OPINIONADOPTED: 30 January 2020doi: 10.2903/j.efsa.2020.6040 https://efsa.onlinelibrary.wiley.com/doi/epdf/10.2903/j.efsa.2020.6040. EFSA J. 18, 28-36
- EPA, 2014. National Air Toxics Assessment. https://www.epa.gov/national-air-toxics-assessment/nata-glossary-terms#hq: 5-12.
- Influence of water activity on physical properties, fungal growth, and ochratoxin A production in dry cherries and green-coffee beans. J. Food Process. Preserv.. 2022;46:e16226.
- [Google Scholar]
- Probabilistic risk assessment (Monte Carlo simulation method) of Pb and Cd in the onion bulb (Allium cepa) and soil of Iran. Environ. Sci. Pollut. Res.. 2018;25:30894-30906.
- [Google Scholar]
- Aflatoxin M1 in human breast milk: a global systematic review, meta-analysis, and risk assessment study (Monte Carlo simulation) Trends Food Sci. Technol.. 2019;88:333-342.
- [CrossRef] [Google Scholar]
- Occurrence and risk assessment of mycotoxins through polish beer consumption. Toxins. 2019;11:254.
- [Google Scholar]
- A risk assessment of dietary exposure to ochratoxin A for the polish population. Food Chem.. 2019;284:264-269.
- [Google Scholar]
- Comparative ochratoxin toxicity: a review of the available data. Toxins. 2015;7:4253-4282.
- [Google Scholar]
- Determination and health risk assessment of acrylamide levels in instant coffee products available in Tehran markets by GC-MS. Int. J. Environ. Anal. Chem.. 2022;1–10
- [Google Scholar]
- Non-carcinogenic health risk assessment due to fluoride exposure from tea consumption in iran using monte carlo simulation. Int. J. Environ. Res. Public Health. 2019;16:4261.
- [Google Scholar]
- The concentration and prevalence of ochratoxin A in coffee and coffee-based products: a global systematic review, meta-analysis and meta-regression. Fungal Biol.. 2019;123:611-617.
- [Google Scholar]
- Development of a new clean-up tandem assay column for the detection of ochratoxin A in roasted coffee. Anal. Chim. Acta. 2005;538:57-61.
- [Google Scholar]
- Effect of Aloe vera extract on reducing aflatoxin B1 in eggs of laying hen and egg yolk oxidative stability. Biointerface Res. Appl. Chem.. 2021;11:12680-12688.
- [Google Scholar]
- Aflatoxin B1 in the Iranian pistachio nut and decontamination methods: a systematic review. Qual. Assurance Saf. Crops Foods. 2020;12:15-25.
- [Google Scholar]
- Assessment of ochratoxin A exposure risk from the consumption of coffee beans in Phnom Penh, Cambodia. Food Addit. Contamin.: Part B. 2022;15:71-77.
- [Google Scholar]
- Ochratoxin A dietary exposure of ten population groups in the Czech Republic: comparison with data over the world. Toxins. 2015;7:3608-3635.
- [Google Scholar]
- Influence of the cultivar and nitrogen fertilisation level on the mycotoxin contamination in winter wheat. Qual. Assurance Saf. Crops Foods. 2017;9:451-461.
- [Google Scholar]
- Coffee and wine with ochratoxin A–exposure risk assessment resulting from its consumption. Medycyna Ogólna i Nauki o Zdrowiu.. 2022;28:190-195.
- [Google Scholar]
- Challenges in specialty coffee processing and quality assurance. Challenges. 2016;7:19.
- [Google Scholar]
- The aflatoxin B1 content of peanut-based foods in Iran: a systematic review. Rev. Environ. Health. 2022;37:29-33.
- [Google Scholar]
- Probabilistic non-carcinogenic and carcinogenic risk assessments (Monte Carlo simulation method) of the measured acrylamide content in Tah-dig using QuEChERS extraction and UHPLC-MS/MS. Food Chem. Toxicol.. 2018;118:361-370.
- [Google Scholar]
- The use of mycotoxin methodology in practice: a need for harmonization. Qual. Assurance Saf. Crops Foods. 2009;1:121-132.
- [Google Scholar]
- Results of a proficiency test for multi-mycotoxin determination in maize by using methods based on LC-MS/(MS) Qual. Assurance Saf. Crops Foods. 2013;5:15-48.
- [Google Scholar]
- Toxicity of ochratoxin a and its modulation by antioxidants: a review. Toxins. 2013;5:1742-1766.
- [Google Scholar]
- Occurrence of the ochratoxin a degradation product 2′ R-ochratoxin a in coffee and other food: an update. Toxins. 2019;11:329.
- [Google Scholar]
- Ochratoxin A in roasted coffee from French supermarkets and transfer in coffee beverages: comparison of analysis methods. Toxins. 2010;2:1928-1942.
- [Google Scholar]
- Determination of Ochratoxin A in green coffee beans by solid-phase microextraction and liquid chromatography with fluorescence detection. J. Chromatogr. A. 2008;1187:145-150.
- [Google Scholar]
- Determination of ochratoxin A in wine by means of immunoaffinity column clean-up and high-performance liquid chromatography. J. Chromatogr. A. 1999;864:89-101.
- [Google Scholar]
- Mycotoxins in Asia: is China in danger? Qual. Assurance Saf. Crops Foods. 2015;7:3-25.
- [Google Scholar]







