Translate this page into:
Coronarin D attenuates MPTP-induced Parkinson’s disease in mice by inhibition of oxidative stress and apoptosis
⁎Corresponding author at: Neurosurgery Department, The First Affiliated Hospital of Xi' an Jiaotong University, Xi’an, Shaanxi 710061, People’s Republic of China. maodewang@163.com (Maode Wang)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Parkinson’s disease (PD) is a brain-related disease condition, globally it is the second most common neurodegenerative disease that mostly disturbs the brain motor control and action of the aged populations. It involves a tiny, dark portion of the brain denoted as substantia nigra. Dopamine is produced in the substantia nigra for the usage of the brain. This dopamine communicates messages among nerves and controls muscle movements. Coronarin D is a diterpenoid with exclusive pharmacological possessions and its effectiveness over a few neurogenerative diseases exposed to some intense properties. The present study was intended to exhibit the neuroprotective efficacy of Coronarin D over MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) induced animals. Oral management of Coronarin D (10 and 20 mg/kg body wt.) protects the MPTP-induced nigrostriatal dopamine (DA) depletion and its intermediate substances. Behavioral studies like narrow beam walk test, open field test and hang test were conducted to study the movement and actions of Coronarin D treated MPTP induced experimental animals, in which Coronarin D treated group III & IV animals showed better behavioral alterations. Our compound as well attenuated MPTP persuaded oxidative pressure in experimental animals. Apoptotic marker studies displayed that management with Coronarin D upturned MPTP induced programmed cell death, which might be its anti-apoptotic properties. Ultimately to conclude, Coronarin D recovered oxidative pressure, neurochemical alterations, apoptosis, and functional irregularities in investigational mice and propose a potential approach to disease management of neurodegenerative and its related disease.
Keywords
Parkinson’s disease
Coronarin D
Dopamine
Glutathione
Caspases
1 Introduction
Neurological-related disease or disorder that distress both peripheral and central nervous systems. Several factors are grounds for these neuro disorders, such as nerve system injury, reduced blood flow, inflammation, endoplasmic reticulum pressure and oxidative stress, excess protein release in neural tissue, and autoimmune-related neuron damage (Dugger et al. 2017). Depending on the affected area, the loss of neurons, proliferation of glial cells, or brain myelin sheath damage leads to motor damage, behavioral changes, and declined cognitive function (Moher et al. 2009). Moreover, the increased global mortality rate by neurological disorders is mainly caused by stroke and its related disorders, that too placing neuro mortality among the foremost factor of non-traumatic mortality in developing industrialized nations (Sharifi-Rad et al. 2018; Newman et al. 2016). Parkinson's disease (PD) is a complex and common age-related neurodegenerative disease condition or disorder with unclear disease-causing factors. Initially, clinical symptoms of PD include dysfunction of the cardinal motor, inactive tremor, postural instability, a disorder of gait, and neurological damage (Kalia and Lang 2015).
United Kingdom Brain Bank Measures defined diagnostic criteria of Parkinson’s disease, like increased accurateness, but after many years of clinical remark as used in these principles, diagnosing cases remains uncertain (Rizzo et al. 2016). The pathological characteristics of PD are dopamine neuron loss and Lewy body deposits, with a subsequent drop in dopamine levels in the black substance called substantia nigra (Kordower et al. 2013; Bendor et al. 2013). Though the causal molecular mechanisms of PD are not clearly understood, oxidative stress, inflammation and apoptosis are supposed to be involved in the disease condition of PD (Mashima et al. 2018). MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) is categorized as a standard toxin molecule to target comparatively precise neurons that are involved in Parkinson’s disease models since it can effortlessly penetrate the blood–brain blockade network and where glial cells take this MPTP and processed to form MPP+ (1-methyl-4-phenyl pyridinium) over monoamine oxidase-B (Nagatsu and Sawada 2006). Discharged MPP+ is particularly taken in by dopamine transporters into dopaminergic neurons and consequently results in oxidative stress and dysfunctions of mitochondria that ultimately induce apoptosis (Sudhakar et al. 2013; Sandhir et al. 2010).
Despite imperative evidence in learning the disease condition of neurodegeneration, numerous efforts and effective advanced treatment approaches are being executed with negligible translational outcomes (Brotchie and Jenner 2011). In this context, recent researchers focus on the herbal-based compound for neuroprotection of PD with distinct importance on anti-inflammation and anti-oxidative actions, thus, an outstanding validation for the expansion of new neuroprotective agent is much needed. Though, numerous diterpenes, have been recognized for their neurobiological actions. Neurological diseases, such as stroke, Parkinson’s, and Alzheimer’s diseases inclined to become more common (Chang et al. 2007). Diterpenes are excellent natural phytochemical that expose numerous capable biological actions, which includes neuroprotective especially (Lu et al. 2019; Islam et al. 2016). Since Parkinson’s disease is labelled as a steady loss of dopaminergic neurons in the midbrain (Siokas et al. 2021; Salehi et al. 2020).
Previous work shows that pre-treatment with Andrographolide, a diterpenoid eliminated lipopolysaccharide-induced decline in dopamine, in which Andrographolide abridged the immunoreactive neuron damage and reduced immunoreactive dendrites (Wang et al. 2004). Here we focused on Coronarin D, which is a labdane-type diterpene, bestowed with stimulating pharmacologic properties (Chimnoi et al. 2009). Several biological properties of Coronarin D were detected, like inhibition of basic and inducible nuclear factor kappa B (NF-κB) pathway, in bone tissue repair, a key intermediary in inflammation, apoptosis, and invasion, and further cytotoxic action against cancer cells (Kunnumakkara et al. 2008). Previous studies stated the antimicrobial activity of Coronarin D against Candida albicans (Kaomongkolgit et al. 2012).
Among numerous bioactive components, the neuroprotective property of Coronarin D metabolite from Curcuma showed a neuroprotective effect through its antioxidative and anti-inflammatory properties (Pulido-Moran et al. 2016). This Coronarin D is to choose the compound of interest for the present study to investigate the neuroprotective efficacy of Coronarin D on the MPTP-induced animal model of Parkinson's disease by examining the oxidative markers and apoptotic markers.
2 Materials and methods
2.1 Chemicals
Coronarin D, MPTP, TBA (thiobarbituric acid), DTNB (3,5-dithio-bisnitrobenzoic acid) and reduced glutathione were acquired from Sigma-Aldrich, USA. Cyt-c (cytochrome-c), apoptotic markers like bax, bcl-2 and anti-caspase-3, 8, 9 obtained from Santa Cruz Biotechnology, Shanghai Corporation limited. Anti-rabbit, horseradish peroxidase-conjugated secondary antibody purchased from Sigma chemical, Massachusetts, United States. Other laboratory reagents used in the present study were procured from Sigma-Aldrich, USA.
2.2 Experimental animals and treatment protocols
For the investigation, C57bL/6 male mice (28 to 30gm) were obtained from Shanghai Laboratory Animal Centre and adapted to workroom situations before the start of the investigation. Under normal temperature conditions, animals and preserved with pure water and animal pellet feeds ad libitum. All animals were adapted to 12hr darkness and 12hr light cycle at 37 °C and weight checked every week to check the animal health condition. All animal experimentation was directed by following the institutional animal ethics committee.
Animals were randomly divided into four groups (n = 6). Group I animals considered as control given with saline only. Group II animals were injected daily with MPTP (30 mg/kg b.wt) intraperitoneally for seven consecutive days. Group III experimental animals were induced with MPTP (30 mg/kg b.wt) along with Coronarin D (10 mg/kg b.wt) for 14 days, from the 7th day onwards. Group IV animals as well injected with MPTP (30 mg/kg b.wt) along with Coronarin D (20 mg/kg b.wt) for 14 days and from the 7th day itself. Behavior tests like hanging and walking tests were performed on the last day of the treatment (Fig. 1). Formerly, investigational animals fasted overnight sacrificed by cervical dislocation and Substantia nigra tissues were obtained quickly and used for the assessment of dopamine and oxidative tension-associated indications.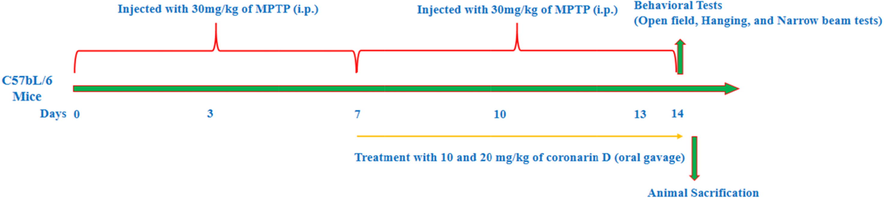
Schematic diagram of experimental design.
2.3 Narrow beam test
Animals with pretraining were permitted to walk on a narrow horizontal immobile wooden beam (length = 100 cm × width = 1 cm) positioned at a height of 100 cm from the ground. The time taken by the animal to cross the flat beam from one end to the other as well as foot slipup errors were calculated (Haydn).
2.4 Hang test
Experimental animals were permissible to grip on a flat horizontal network grid. Further, the flat grid was upturned therefore the animal was allowable to swing upside down, finally hanging phase or time was calculated to measure the motor control (Martino et al. 2020).
2.5 Open field test
Experimental mice were located at one end of the corner of the open area compartment, animal activity and movement were detected for 5 min. When the mice enter the square with both the forelimbs, one time or many times is completed. The number of the center points and marginal squares travelled by the investigation animal was noted down. Actions of mice which includes rearing and grooming were as well noted (Campos et al. 2013).
2.6 Assessment of dopamine and its metabolites
Instantly after obtaining the tissues, fragment striatal tissues were agitated in ice-cold perchloric acid (0.1 M) which contains a pinch of EDTA, and tissues were allowed for centrifugation at 10,000 rpm for 10 min. Then the supernatant solution was collected in a separate tube and further injected (10 µl) into the HPLC (Ultimate 3000 model) and the outcome was denoted in ng/mg wet weight.
2.7 Estimation of thiobarbituric acid reactive substances (TBARS)
Momentarily, the substantia nigra tissue extracts were incubated with phenyl methosulfate (0.2 ml) for 1hr at room temperature. And further tricarboxylic acid (0.4 ml) and thiobarbituric acid (0.4 ml) was added to the mixture. Then the reacting solution was kept for centrifugation at 5000 rpm for 10 min, and finally, the separated solution was kept for boiling for 10 min. Ultimately with subsequent chilling, the final sample solution was read at 534 nm (Shreesh Ojha et al. 2015).
2.8 Assessment of superoxide dismutase (SOD)
The analyzing mixture consists of sodium carbonate buffer (960 µl) which contains xanthine (0.1 mM), EDTA (0.1 Mm), nitro blue tetrazolium chloride (0.025 mM), xanthine oxidase (20 µl) and finally brain supernatant (20 µl) were taken. Ultimately change or alteration in optical density were detected by means of a spectrophotometer at 560 nm (Youdim et al. 2001).
2.9 Determination of catalase activity
To determine the catalase (CAT) activity the evaluating mixture comprised of Tris-HCl buffer (50 µl, with 1 M) at pH 8, which further contains EDTA (5 mM), 10 mM hydrogen peroxide (900 µl), brain tissue supernatant (20 µl) finally to mix well milli Q water (20 µl). The proportion of disintegration of hydrogen peroxide was experimental spectrophotometrically at 250 nm (Singh et al. 2018).
2.10 Examination of glutathione peroxidase activity
The glutathione peroxidase examining mix contains 1 M Tris-HCl (100 µl) at pH 8.0 Further added with 5 mM EDTA, 0.1 M GSH (20 µl), glutathione reductase (100 µl) solution (10Units/ml), 2 mM nicotinamide adenine dinucleotide phosphate solution (100 µl), pure distilled water (600 µl), brain supernatant sample (10 µl) and 7 mM of tert-butyl hydroperoxide (10 µl) and. Oxidation of nicotinamide adenine dinucleotide phosphate solution was analyzed by spectrophotometer at 350 nm (Laurie K. Mischley et al. 2016).
2.11 Assessment of glutathione level
Tissue homogenates from the brain were centrifuged at 15,000 rpm for 15 min at a chilled condition of 4 ◦C. 0.5 ml of supernatant was added to ice-cold 0.1 mM Ellman's reagent solution (4 ml) which comprise 1 M phosphate buffer at pH 8. The changes in optical density were observed spectrophotometrically at 415 nm (Ewa B. Romuk et al. 2017).
2.12 Measurement of apoptotic marker levels
The anti-apoptotic marker like bcl-2 and further pro-apoptotic markers such as bax, cytochrome-c, and caspases (3, 8 and 9) were assessed in the present study. In which relative intensity was compared in MPTP-induced experimental animals and Coronarin D treated animals by means of enzyme-linked immunosorbent assay (ELISA, Fisher Scientific Biotech, Roskilde, Denmark) in accordance with manufacturer instructions.
2.13 Statistical analysis
All data were exposed as mean ± SD of the number of investigations (n = 6). The statistical investigation was executed by means of one-way analysis of variance by SPSS version 16.0 software and individual assessments were attained by using DMRT (Duncan’s multiple range test).
3 Results
3.1 Behavioral alterations in experimental animals
In the behavioral assessment, we examined the balance and motor synchronization skills by narrow walking test and measured the movement and action through an open field test and further explored neuromuscular efficacy and coordination by hang test in experimental and control animals. In above mentioned all test, MPTP alone treated group II animals displayed took extra time to go across the beam with augmented foot slip-up mistakes in the walking test, experimental animals took more hanging phase or time in hang test, abridged center & peripheral movements, ultimately expressed rearing and grooming actions in an open field test in comparison with the group I control animals (p < 0.05), however pre-treatment with Coronarin D in group III & IV (10 mg & 20 mg/kg body wt.) reduced the behavioral irregularities seen in MPTP animals. Especially 20 mg treated group IV animals showed better behavioral alterations when compared to 10 mg treated group III animals. The results were depicted in (Figs. 2-4) consequently.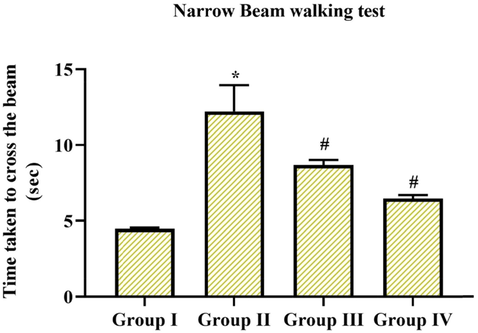
Extent of steadiness and muscular coordination by means of narrow walking test. MPTP-induced animals takes much time period to go across the beam with increased foot slip mistakes, while pre-administration of coronarin D suggestively altered motor defects provoked by MPTP. Data given are mean ± SD, (n = 6), results not allocating common superscript are significant with each other *,#p < 0.05, ANOVA charted by DMRT. Note: Group I: Control; Group II: MPTP-induced PD mice; Group III: MPTP + 10 mg/kg of coronarin D treated mice; Group IV: MPTP + 20 mg/kg of coronarin D treated mice.
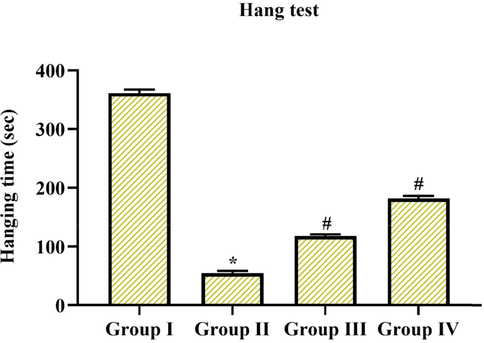
Determination of neuromuscular strength by hang test. Hanging phase of MPTP-induced animals were extremely decreased, in the interim, treatment with coronarin D pointedly improved hanging time of MPTP-induced animals. Results are expressed as mean ± SD, (n = 6), further not sharing common superscript are momentous with each other *,#p < 0.05. Note: Group I: Control; Group II: MPTP-induced PD mice; Group III: MPTP + 10 mg/kg of coronarin D treated mice; Group IV: MPTP + 20 mg/kg of coronarin D treated mice.
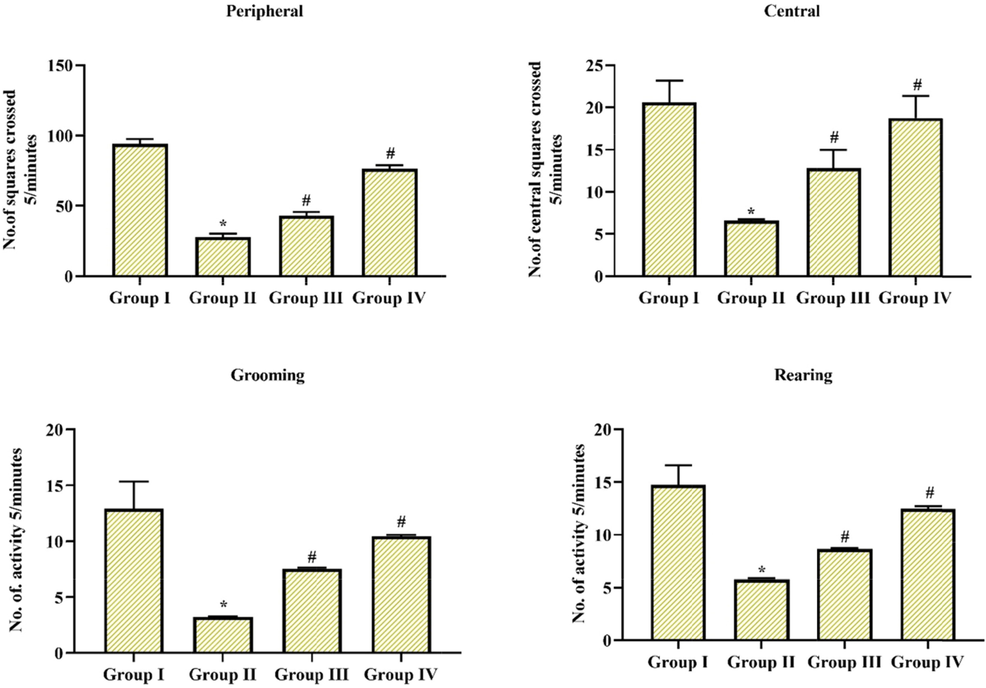
Resolving adaptation capability and motor performance in open field test. Induction of MPTP leads to substantial downregulation in movement and action; while, these reductions were protected by coronarin D treatment. Outcome given are mean ± standard deviation, (n = 6), values not sharing common superscript are significant with each other *,#p < 0.05. Note: Group I: Control; Group II: MPTP-induced PD mice; Group III: MPTP + 10 mg/kg of coronarin D treated mice; Group IV: MPTP + 20 mg/kg of coronarin D treated mice.
3.2 Dopamine and its metabolites preserved by Coronarin D management
Fig. 5 exposed a noteworthy downregulation in dopamine levels and its related substances in MPTP alone treated group II animals in comparison with group I control animals, at the same time pre-administration of Coronarin D enhanced the levels of dopamine and its metabolites in MPTP-induced group III & IV animals. Though, it was detected that both the 10 & 20 mg/kg body wt. exposed better induction, but much significance was observed in 20 mg treated group IV animals.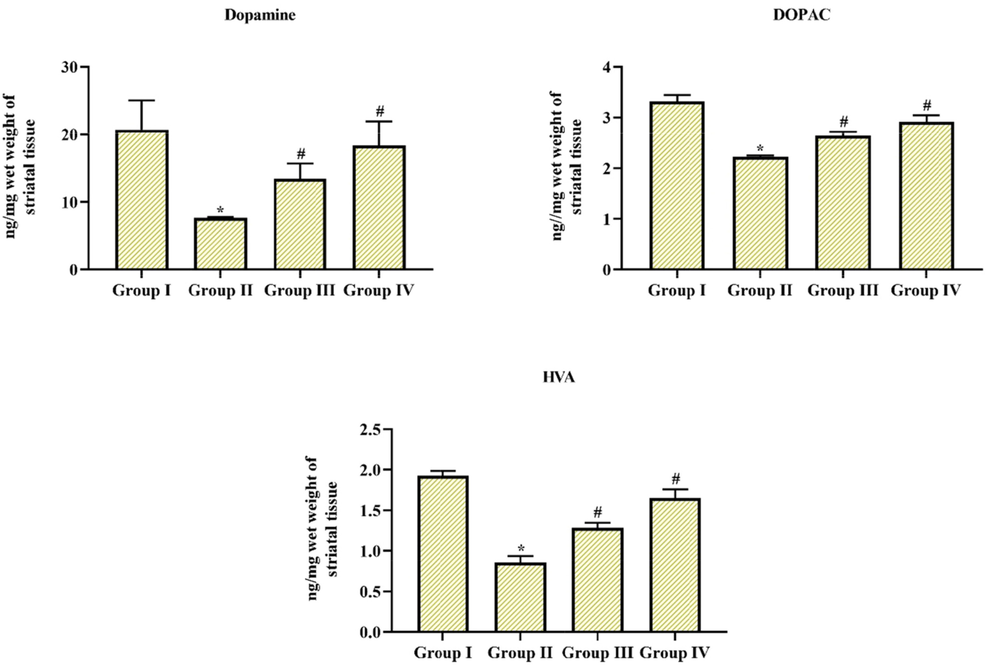
Coronarin D treatment conserved the dopamine depletion and its related substances Treatment of MPTP reduced the dopamine levels as well its connected substances, then management with cornarin D attenuated and restored the dopamine levels in MPTP-treated experimental animals. Results given are mean ± SD,(n = 6), values not sharing common superscript are significant with each other *,#p < 0.05. Note: Group I: Control; Group II: MPTP-induced PD mice; Group III: MPTP + 10 mg/kg of coronarin D treated mice; Group IV: MPTP + 20 mg/kg of coronarin D treated mice.
3.3 Effect of Coronarin D on oxidative and antioxidative markers
Oxidative pressure is the foremost factor to trigger neurodegeneration in diseases like PD. The activities of SOD and CAT and further levels of TBARS were upregulated in MPTP-only induced group II animals. Whereas pre-administration of Coronarin D suggestively downregulated their activities and levels in comparison with MPTP induced as well in group III & IV animals (10 and 20 mg/kg body wt). In addition, the GSH and GPx levels were considerably downregulated in MPTP alone induced group II animals. At the same time, Coronarin D pre-treatment significantly upregulated the GSH and GPx levels (p < 0.05). Precisely the group IV animals treated with (20 mg) of Coronarin D showed some excellent efficacy over oxidative stress. The outcome was depicted in Fig. 6.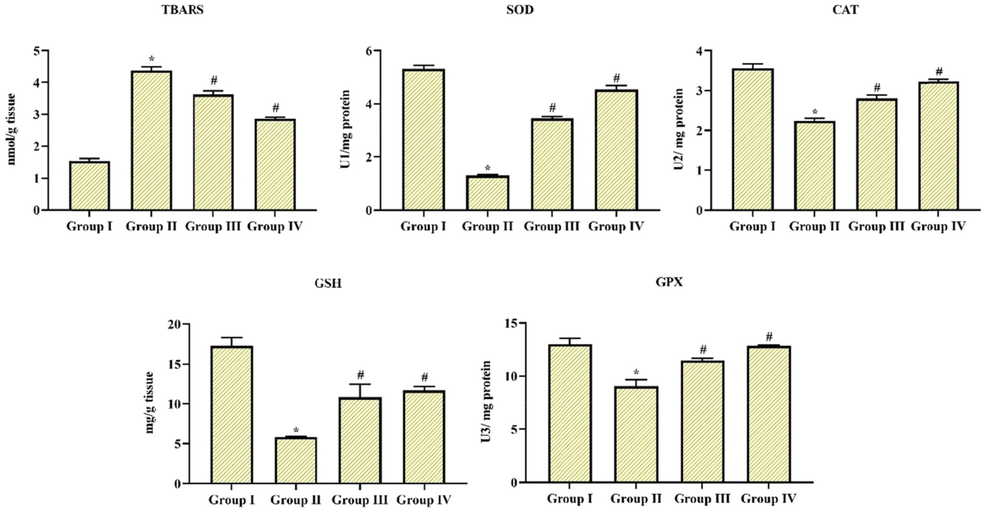
MPTP induced oxidative stress can be attenuated by Coronarin D The altitudes of TBARS and actions of SOD and CAT were upregulated along with a downregulated levels of GSH and GPx in MPTP-treated animals, while, administration with coronarin D significantly enhanced oxidative stress. Results given are mean ± SD, (n = 6), values not sharing common superscript are momentous with each other *,#p < 0.05, one way ANOVA followed by DMRT. Note: Group I: Control; Group II: MPTP-induced PD mice; Group III: MPTP + 10 mg/kg of coronarin D treated mice; Group IV: MPTP + 20 mg/kg of coronarin D treated mice.
3.4 Effect of Coronarin D on apoptotic markers
Apoptosis is described as programmed cell death that plays a crucial role in all types of neurodegenerative diseases, especially PD. The expression figure of bax, caspase 3, caspase 8, and 9 was expressively upregulated in MPTP alone induced group II animals, while Coronarin D treatment significantly downregulated the appearance of these proapoptotic markers in group III & IV animals (10 and 20 mg/kg body wt). In addition, the expressions of cyto c and bcl- 2 were pointedly downregulated in MPTP alone induced animals in comparison with group I control, however Coronarin D pre-treatment suggestively improved the expressions of cyto c and bcl- 2 in group III & IV animals (10 and 20 mg/kg body wt). In apoptotic markers as well 20 mg of Cornarin D showed better results than 10 mg. The results were portrayed in Fig. 7.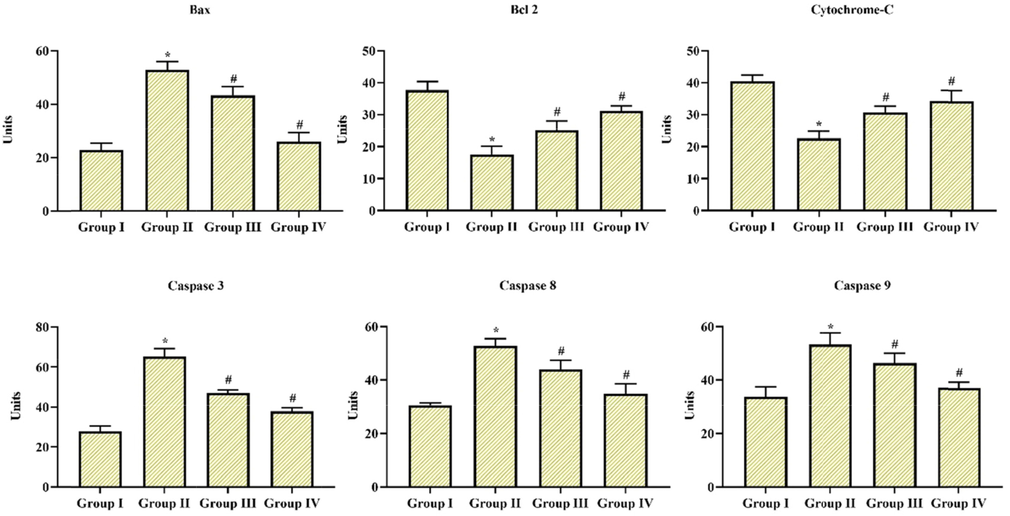
Coronarin D on apoptotic Markers by ELISA Apoptotic marker expressions like proapoptotic and anti-apoptotic molecules caspase 3, caspase 8 and caspase 9, bcl-2 and bax, cytochrome-c, in the substantia nigra was measured by ELIZA kit. The relative intensity was denoted a percentage in all groups of experimental animals. Results given are mean ± standard deviation, values not sharing common superscript are significant with each other *,#p < 0.05. Note: Group I: Control; Group II: MPTP-induced PD mice; Group III: MPTP + 10 mg/kg of coronarin D treated mice; Group IV: MPTP + 20 mg/kg of coronarin D treated mice.
4 Discussion
Dopamine is a significant neurotransmitter accountable for control and balance of motion, especially walking movement. MPTP induction initiated discerning damage of dopaminergic neurons in the two-sided dopaminergic area and leads to a loss in the levels of nigrostriatal DA. Previous studies demonstrate the administration of diterpenoids enhanced MPTP-induced Dopamine depletion, which coincides with the outcome of the present study (Zahra Shahpiri et al. 2016). Coronarin D is an exciting bioactive compound. The pharmacologic properties of this compound might be useful in several domains, such as anti-microbial, antiinflammation, and anticancer (Bailly 2020). Narrow beam walking, open field, and hanging test are used as indications to observe the mechanical damages in MPTP-induced behavioral irregularities in animal models (Nataraj et al., 2016). In our study, MPTP tempted animal behavior alterations are mostly due to extreme loss in midbrain dopaminergic neurons with deterioration of their striatal nerve terminals. At the same time, Coronarin D treatment instigated a recapture of behavioral damage provoked by MPTP, by improving the Dopamine level.
Repression of electron transport chain through oxidation–reduction reaction by 1-methyl-4-phenylpyridinium, a lethal intermediate substance of MPTP, leads to the dripping of electron flow, which might repeatedly produce the fabrication of reactive oxygen species several times when reacts with excess available oxygen (Asokan Prema et al. 2015). Previous studies evidently indicate the induction of MPTP remains to upregulate TBARS level, which is an indication of lipid peroxidation progressions and actions of catalase and superoxide dismutase, further with diminished the levels of GPx and GSH activity which might replicate a modified response to oxidative pressure. Coronarin is stated as the most operative compound which exposes significant strong antioxidant ability and is effective in quenching free radicals (Tian et al. 2020). In the present study, pre-treatment of Coronrin D suggestively retrieved oxidative pressure induced by MPTP due to its biological property to scavenge free radicals to impose its antioxidant properties. Further in our study, MPTP induction downregulated the relative intensity pattern of bcl-2 and augmented cytochrome-c discharge with the upregulated intensity of bax, caspase-3, caspase 8, and caspase 9, thus favoring programmed cell death or apoptosis (Nataraj et al., 2016).
Few reports denote that the management of Coronarin D inhibits constituent and influences neuronal apoptosis, inhibition of invasion, and destruction of differentiation of monocytes into osteoclasts by improving anti-apoptotic and downregulating proapoptotic marker levels. The overall mechanism of Cornarin D over MPTP-induced experimental animals was portrayed in Fig. 8. Numerous studies described that the neuroprotective efficacy of Coronarin D assigned by rectification of inflammation, mitochondrial damage, augmented neural stem cells and neurons which subsidizes indirectly or directly to the disease condition of Parkinson’s disease, Alzheimer’s disease, stroke, lateral sclerosis, and other trauma conditions.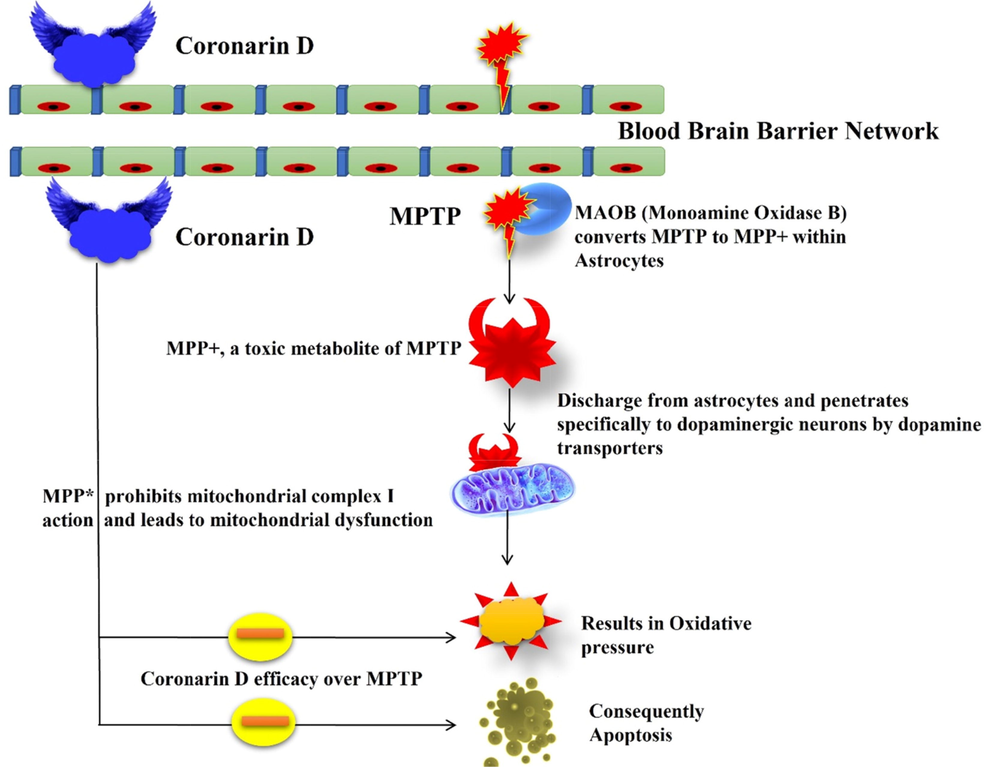
Overall mechanism of Coronarin D.
5 Conclusion
Finally, our results suggest that coronarin D exhibits a recovered behavioral change in MPTP-induced animals, further restoring the depletion of dopamine in MPTP-induced experimental animals. In addition, coronarin D managed oxidative stress and exhibited improved anti-apoptotic and diminished proapoptotic marker levels. These outcomes proved the beneficial properties of coronarin D on the PD. Furthermore, an additional studies are still required in the future to expound the inner mitochondria-mediated preventive and anti-inflammation outcome of Coronarin D over MPTP persuaded neurotoxicity to authorize its efficacy in detail.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Anticancer activities and mechanism of action of the labdane diterpene coronarin D. Pathol. – Res. Pract.. 2020;216(6):152946
- [CrossRef] [Google Scholar]
- Rodent models of Parkinson’s disease: beyond the motor symptomatology. Front. Behav. Neurosci.. 2013;7:175.
- [CrossRef] [Google Scholar]
- Quantitative structure-activity relationship (QSAR) for neuroprotective activity of terpenoids. Life Sci.. 2007;80:835-841.
- [Google Scholar]
- Phytochemical reinvestigation of labdane-type diterpenes and their cytotoxicity from the rhizomes of Hedychium coronarium. Phytochem. Lett.. 2009;2(4):184-187.
- [Google Scholar]
- Pathology of neurodegenerative diseases. Cold Spring Harb. Perspect. Biol.. 2017;9:a028035
- [Google Scholar]
- Diterpenes: advances in neurobiological drug research. Phytother. Res.. 2016;30(6):915-928.
- [Google Scholar]
- Antifungal activity of coronarin D against Candida albicans. Oral Surg. Oral Med. Oral Pathol. Oral Radiol.. 2012;114(1):61-66.
- [Google Scholar]
- Disease duration and the integrity of the nigrostriatal system in Parkinson’s disease. Brain. 2013;136:2419-2431.
- [CrossRef] [Google Scholar]
- Coronarin D, a labdane diterpene, inhibits both constitutive and inducible nuclear factor-κB pathway activation, leading to potentiation of apoptosis, inhibition of invasion, and suppression of osteoclastogenesis. Mol. Cancer Ther.. 2008;7(10):3306-3317.
- [Google Scholar]
- Haydn N. Allbutt Jasmine M. Henderson. Use of the narrow beam test in the rat, 6-hydroxydopamine model of Parkinson's disease. Journal of Neuroscience Methods Volume 159, Issue 2, 30 January 2007, Pages 195-202.
- A review for the neuroprotective effects of andrographolide in the central nervous system. Biomedicine & Pharmacotherapy. 2019;117
- [CrossRef] [Google Scholar]
- Neuromechanical assessment of activated vs. resting leg rigidity using the pendulum test is associated with a fall history in people with Parkinson’s disease. Front. Hum. Neurosci.. 2020;14(602595)
- [CrossRef] [Google Scholar]
- Mashima, K., Takahashi, S., Minami, K., Izawa, Y., Abe, T., Tsukada, N., et al. (2018). Neuroprotective role of astroglia in Parkinson disease by reducing oxidative stress through dopamine-induced activation of pentose-phosphate pathway. ASN Neuro 10:1759091418775562. https://doi.org/10.1177/1759091418775562.
- Laurie K. Mischley, Leanna J. Standish, Noel S. Weiss, Jeannie M. Padowski, Terrance J. Kavanagh, Collin C. White and Michael E. Rosenfeld. Glutathione as a Biomarker in Parkinson’s Disease: Associations with Aging and Disease Severity. Oxidative Medicine and Cellular Longevity Volume 2016, Article ID 9409363, 6 pages http://dx.doi.org/10.1155/2016/9409363.
- Moher D, Liberati A, Tetzla J, Altman DG, Altman D, Antes G, Atkins D, Barbour V, Barrowman N, Berlin JA. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement (Chinese edition). J. Chin. Integr. Med. 2009, 7, 889-896.
- Molecular mechanism of the relation of monoamine oxidase B and its inhibitors to Parkinson's disease: possible implications of glial cells. J. Neural. Transm. Suppl.. 2006;71:53-65.
- [CrossRef] [Google Scholar]
- Lutein protects dopaminergic neurons against MPTP-induced apoptotic death and motor Q8 dysfunction by ameliorating mitochondrial disruption and oxidative stress. Nutr. Neurosci.. 2016;19(6):237-246.
- [CrossRef] [Google Scholar]
- Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod.. 2016;79:629-661.
- [Google Scholar]
- Neuroprotective effect of lycopene against MPTP induced experimental Parkinson’s disease in mice. Neuroscience Letters. 2015 Jul;599:12-19.
- [CrossRef] [Google Scholar]
- Accuracy of clinical diagnosis of Parkinson disease: a systematic review and meta-analysis. Neurology. 2016;86(6):566-576.
- [Google Scholar]
- The evaluation of the changes in enzymatic antioxidant reserves and lipid peroxidation in chosen parts of the brain in an animal model of Parkinson disease. Adv. Clin. Exp. Med.. 2017;26(6):953-959.
- [Google Scholar]
- The Therapeutic Potential of Anthocyanins: Current Approaches Based on Their Molecular Mechanism of Action. Front. Pharmacol.. 2020;11:1300.
- [CrossRef] [Google Scholar]
- Lycopene prevents 3-nitropropionic acid-induced mitochondrial oxidative stress and dysfunctions in nervous system. Neurochem. Int.. 2010;57:579-587.
- [Google Scholar]
- Shahpiri Z, Bahramsoltani R, Hosein Farzaei M, Farzaei F, Rahimi R. Phytochemicals as future drugs for Parkinson's disease: a comprehensive review. Rev Neurosci. 2016 Aug 1;27(6):651-68. https://doi.org/10.1515/revneuro-2016-0004.
- In vitro and in vivo assessment of free radical scavenging and antioxidant activities of Veronica persica Poir. Cell. Mol. Biol.. 2018;64:57-64.
- [Google Scholar]
- Shreesh Ojha, Hayate Javed, Sheikh Azimullah, Salema B Abul Khair, M Emdadul Haque. Neuroprotective potential of ferulic acid in the rotenone model of Parkinson’s disease. Drug Design, Development and Therapy 2015:9.
- Effect of chlorogenic acid supplementation in MPTP-intoxicated mouse. Front. Pharmacol.. 2018;9:757.
- [CrossRef] [Google Scholar]
- ADORA2A rs5760423 and CYP1A2 rs762551 Polymorphisms as Risk Factors for Parkinson’s Disease. J Clin Med. 2021 Jan 20;10(3):381.
- [CrossRef] [Google Scholar]
- Sudhakar Raja Subramaniam and Marie-Francoise Chesselet. Mitochondrial dysfunction and oxidative stress in Parkinson’s disease. Prog Neurobiol. 2013 Jul-Aug; 0: 17-32.
- Phytochemical analysis, antioxidant, antibacterial, cytotoxic, and enzyme inhibitory activities of Hedychium flavum rhizome. Front. Pharmacol.. 2020;11:572659
- [CrossRef] [Google Scholar]
- Andrographolide reduces inflammation-mediated dopaminergic neurodegeneration in mesencephalic neuron-glia cultures by inhibiting microglial activation. J. Pharmacol. Exp. Therap.. 2004;308(3):975-983.
- [Google Scholar]
- Oxidative stress indices in Parkinson's disease : biochemical determination. Methods Mol. Med.. 2001;62:137-153.
- [CrossRef] [Google Scholar]







