Translate this page into:
Cs2SnI6 perovskites nanostructures as excellent photocatalytic degradation of organic dye pollutants in water under visible light: Synthesis and characterization
⁎Corresponding author. salavati@kashanu.ac.ir (Masoud Salavati-Niasari)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Pollution of surface waters containing chemical dye combinations and/or biological materials can have adverse effects on human health and other organisms, even in small quantities. The photocatalytic oxidation procedure has been widely regarded as a commercially viable method of removing environmental pollutants. In recent research, Caesium-hexaiodostannate nanostructures (Cs2SnI6) were prepared through a co-precipitation approach and their photocatalytic performance was analyzed. A number of surfactants were examined for their influence on the structure, purity, and morphology of the sample. It was found in the SEM data that the presence of surfactants has a negative impact on the morphology, which may be due to the inability to remove surfactants by calcination at high temperatures. Photocatalytic activity of Cs2SnI6 catalysts was demonstrated over a variety of organic dyes, including methylene blue (MB), rhodamine b (RhB), methyl orange (MO), and methyl violet (MV). Upon exposure to visible light for 180 min, 5 ppm methylene blue showed the greatest degradation (84.0%). Cs2SnI6 nanostructures are being applied for the first time in photocatalytic applications. Due to their wide bandgap (1.7 eV) in the visible region, they may be a suitable candidate for water purification. Moreover, the results indicated that the efficiency of photocatalysis was affected by various parameters, including pH, dye concentration, dye types, catalyst dosages, and surfactants. In the recyclability test, Cs2SnI6 nanostructures were found to be stable, and the removal efficiency was reduced by 6.9% after the fifth run.
Keywords
Perovskites
Semiconductor
Co-Precipitation method
Cs2SnI6 nanostructures
Visible-Light-Driven photocatalysis
1 Introduction
It is widely accepted that population growth and industrial development are not only reducing clean water resources, but also causing degradation in water quality, which negatively impacts the health and safety of humans and other living organisms (Ajibade & Oluwalana, 2021; Zhang et al., 2022). Furthermore, wastewater contains a variety of organic compounds, including dyes, medicines, and personal care products that are often resistant to natural degradation (Li et al., 2022a). Although their concentration is very low, the damage they cause cannot be ignored (Li et al., 2022b). Diverse industries can release sewage directly into the environment, which can have a negative impact on aquatic life and human health (Sharwani et al., 2021). In order to purify them, a great deal of energy must be expended (Khan, 2021). As a result of chemical spills, agricultural runoff, and industrial effluent, toxic coloring agents are discharged into water systems (Islam et al., 2021; Lavanya et al., 2014). In many countries, regulatory authorities and societies face problems as a result of stability, acute toxicity, and durability in the ecosystem for long periods of time (Kubra et al., 2021). During the dyeing process, a significant amount of water is used and discharged into the ecosystem on a regular basis. Textile dyeing industry effluents, which contain carcinogenic and hazardous dyes, are poisonous to mammals, aquatic microorganisms, and fish, threaten to destroy the aquatic environment by destroying hydrophyte plants in their photosynthesis (Fu et al., 2011; Nasar, 2021). Increasing amounts of chemical oxygen from dye wastewater produced by photographic and textile industries are causing an ecological crisis due to their unsuitable color, toxicity, and resistance to biological, photochemical, and chemical degradation (Kumar et al., 2021; Waheed et al., 2021; Zhou et al., 2021). Most colorants are mutagenic, carcinogenic, and life-threatening. Dye can cause allergic reactions, such as respiratory infections, contact dermatitis, respiratory tract irritation, and even cancers of the liver, bladder, and kidney. Highly poisonous textile dyes can reduce the penetration of light in water, reduce the transparency and quality of water, and affect the photosynthetic behavior of hydrophytic plants, leading to a lack of oxygen in aquatic environments (Al Kausor & Chakrabortty, 2021). Contaminants such as these pose a great threat to the aquatic ecosystem as well as to human health. In an attempt to remove these pollutants, methods such as ozonation, biochemical treatment, and electrochemical oxidation have been used, but they are hindered by several limitations (Sui et al., 2020; Xu et al., 2022). The purification of these toxic colorants in organic micro-pollutants (OMP) has always attracted the attention of scientists. As a general rule, conventional methods of eliminating organic dyes do not meet the standards for water quality and are not compatible with existing water sources. Due to its ability to remove toxic pollutants entirely to mineral acids, water, and carbon dioxide, photocatalysts based on semiconductors have gained considerable attention for their use in the refinement of environmental contaminants in sewage (Arunachalam et al., 2021; Ghanbari & Salavati-Niasari, 2018; Liu et al., 2011; Zhang et al., 2007).
A semiconductor-based photocatalysis technology is the most lucrative due to its potential applications in a variety of fields, particularly in the decomposition of toxic contaminants(Pasternak & Paz, 2013; Wu et al., 2010). As a result of the advancement of nanotechnology, photocatalysis has become one of the most important approaches in wastewater treatment. Two aspects of issues related to water are of particular interest to researchers: first, simple and low-cost access to clean drinking water, and second, appropriate and low-cost treatment of effluents to limit environmental harm. All around the world, significant amounts of research are conducted daily in these fields. Several technologies and methods are being developed and optimized for the treatment of water and wastewater, including the use of photocatalysts. The process of photocatalysis involves the use of a semiconductor material that is capable of producing electron holes in the presence of visible or ultraviolet light (Karami et al., 2021).
Perovskites are characterized by a wide range of structural and compositional variations that offer many possibilities for overcoming the challenges posed by MPbX3 and MSnX3 perovskites halides. As with perovskites oxides, halide perovskites are also available (Attfield et al., 2015). The BI6 octahedron undergoes a phase transition that reduces symmetry during cooling, resulting in a coordinated tilt that affects its conductivity and bandgap (Amat et al., 2014; Chung et al., 2012; Moller, 1958; Swainson et al., 2015). Besides, halides can create ordered double perovskites with the general equation M2N'N“X6, wherein the rock-salt ordering of the N' and N” metals are present. The order is usually due to an extensive charge difference between the two metals (Anderson et al., 1993). Substitution of one site of N cation with an empty site in the M2N'N“X6 double perovskite results in the vacancy-ordered double perovskites, M2NX6. Commonly mentioned as antifluorite compounds, these defective perovskites embrace the K2PtCl6 structure type, which is defined as isolated [BX6]2−octahedra units bridged with M+ cations (Maughan et al., 2016; Wang et al., 2019). There are some similarities between M2NX6 perovskites and MNX3 perovskites. A close-packed ionic lattice similar to that of ABX3 perovskites is maintained, despite the removal of all other [BX6] octahedrons. Additionally, vacancy-ordered structures undergo coordinated octahedral tilts and rotations during phase transitions, reducing their symmetry during the cooling process (Abrahams et al., 1989; Henkel et al., 1980; Lee et al., 2014; Saparov et al., 2016). Cs2SnI6 exhibits a cubic structure at ambient temperature with a space group of Fm3m, as indicated by its radius ratio ∼ of 0.94 (Shin et al., 2019). The mixture is characterized by n-type electrical conductivity, strong visible light absorption, moisture stability, and air stability, which are all beneficial for photovoltaic devices (Lee et al., 2014; Saparov et al., 2016). One of the most critical factors in the selection of light for photocatalysis is the catalyst bandgap. We have conducted a wide range of photocatalysis research using semiconductor nanoparticles in our lab. In this field, new materials are constantly being created, optimized, and utilized. It is known that Cs2SnI6 has a bandgap that is suitable in the visible region; 1.622 eV (Qiu et al., 2016). Because of its low cost, environmental friendliness, and high efficiency, the cesium tin iodide perovskite semiconductor (Cs2SnI6) has recently attracted considerable attention as a semiconductor for light-emitting diodes, solar cells, catalytic electrodes in fuel cells, superconductivity, sensors, and photodetectors. (Rasukkannu et al., 2018).
As one of the primary objectives of this paper, Cs2SnI6 nanocomposite will be synthesized in a simple and cost-effective manner. Synthesis of nanocomposite Cs2SnI6 was accomplished by co-precipitating. The characterization of Cs2SnI6 has been performed using a variety of methods, including XRD, SEM, EDS, TEM, BET, and DRS. As part of this study, Cs2SnI6 was examined for its photocatalytic activity. It is a first-of-its-kind study that will investigate pH, dye type, catalyst dosage, dye concentration, scavenger type, and surfactant type.
2 Experimental
2.1 Materials and characterization
The reagents used in this study were all of analytical quality. Potassium iodide (KI), Cesium chloride (CsCl), Tin(IV) chloride pentahydrate (SnCl4·5H2O), Polyethylene glycol (PEG-6000), Sodium dodecyl sulfate (SDS), polyvinyl pyrrolidone (PVP-25000), sodium dodecylbenzene sulfonate (SDBS), Ethylenediamine tetra acetic acid (EDTA), Sodium salicylate (NaHSal), Benzoic acid (BA), 1,4-Benzoquinone (BQ), sodium azide (NaN3), were purchased from Merck Company and applied without additional purification. We employed methyl violet (C24H28N3Cl), rhodamine B (C28H31ClN2O3), methyl orange (C14H14N3NaO3S), and methylene blue (C16H18ClN3S) as contaminants for studying the photocatalytic behavior of Cs2SnI6 nanostructures.
XRD (X-ray diffraction) patterns were collected using a Philips diffractometer with X'PertPro monochromatized Cu K radiation (λ = 1.54) to determine the type of structure and purity of the as-synthesized nanoparticles. The shape and distribution of nanoparticles were examined using FESEM (field emission scanning electron microscopy) (Mira3 TESCAN). The nanoparticles were evaluated by transmission electron microscopy using a JEM-2100 TEM, and by energy dispersive spectrometry using a Philips XL30 microscope. Catalyst specific surface areas were calculated using the Brunauer-Emmett-Teller (BET) technique. To determine adsorption/desorption, an automated gas adsorption analyzer (Tristar 3000, Micromeritics) was used to pump liquid N2 at −196 °C.
2.2 Synthesis route
Precursors must be prepared before Cs2SnI6 nanoparticles can be synthesized. SnI4 was synthesized using the co-precipitation method in the first step. A solution of 1.5 mmol SnCl4·5H2O and a solution of 3.0 mmol KI were dissolved in distilled water in two beakers and then combined. Second, stoichiometric amounts of CsCl (1.5 mmol) and LiI (1.5 mmol) were dissolved in distilled water in two beakers for the synthesis of CsI (if surfactant (1.5 mmol) was used, it was added at this point). Following the mixing and stirring of the solutions, the reaction was carried out for 15 min. CsI solution was added to a beaker containing SnI4 and the solution turned yellow. Following 30 min of stirring, the precipitate was washed several times with ethanol to remove residual surfactant and dried in an oven at 65 °C. It is apparent from the black precipitate obtained that nanoparticles have been formed (Scheme 1). A variety of tests and analyses were conducted on the precipitate resulting from the reaction.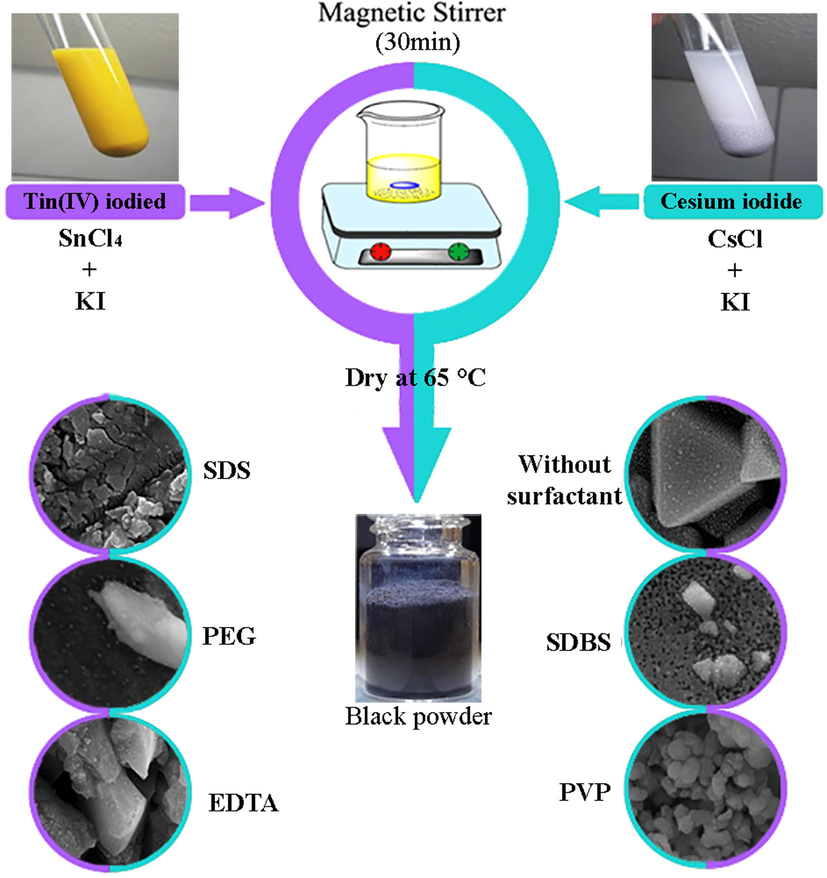
Schematic diagram of the fabrication of Cs2SnI6 nanostructures.
2.3 Photocatalyst process
In order to conduct the photocatalysis procedure, different amounts of the optimum samples were added to 100 ml of dye solution at different concentrations (such as 5, 10, and 15 ppm). During the photocatalysis process, the solution was aerated for 20 min. The photocatalytic process was carried out using an Osram light (400 W) with a wavelength ranging between 400 and 780 nm. As soon as the aeration process had been completed, the first sample was collected. To reach equilibrium, the mixture containing catalyst and dye was blended in the dark for 30 min before turning on the lamp below visible rays. The reaction medium was then illuminated with visible light. Despite the fact that the initial sampling was conducted after 30 min, subsequent samplings were conducted every 30 min. Photocatalysis was performed at room temperature without external light or catalyst, and after 120 min, hardly any dye was degraded without light and catalyst. A UV–visible spectrophotometer was used to measure the absorbance of the collected samples in order to verify the discoloration. The percentage of discoloration was calculated by using the following formula (Eq. 1), where A0 represents the quantity of dye absorption before and after exposure to light, and At represents the amount of dye absorption after exposure to light:
2.4 Cyclic voltammetry (CV) test
Electrochemical energy storage capacity and attributes of products were assessed using the cyclic voltammetry (CV) technique. The probe solution of potassium ferricyanide/potassium ferrocyanide (K3(Fe(CN)6)/K4(Fe(CN)6) in the 0.1 M phosphate buffer solution (pH = 7.2) was applied for CV test at the scan rate of 0.1 Vs−1. Electrochemical cells were thermostatized at 25 °C in 2 M KOH electrolyte. These electrodes consist of the working electrode, Pt electrode, and Ag/AgCl electrode. In order to prepare the working electrode without a binder, a thin layer of as-fabricated materials was coated on a copper substrate using the drop cast method. Through the current between the as fabricated electrode and the counter electrode, the potential of the fabricated working electrode was measured against the reference electrode.
3 Results and discussion
3.1 Characterization of Cs2SnI6
The XRD pattern of Cs2SnI6 as synthesized is shown in Fig. 1a. Each diffraction peak is associated with cesium tin iodide with JCPDS No. 073–0330 having a cubic crystal structure (space group = Fm-3m). The effect of several surfactants on the purity and structure of Cs2SnI6 was studied, including SDS, SDBS, NaHSal, PEG6000, PVP, and EDTA. Fig. 1b and 1c illustrate the XRD pattern of Cs2SnI6 prepared with SDS and SDBS as anionic surface active agents. Tin iodide with JCPDS No. 075–1559 was composed as a by-product along with the main phase. A small amount of unknown phase was also formed in the presence of SDBS. When sodium salicylate (NaHSal) is used as a surface-active agent, it can result in certain impurities such as unknown phase and SnI4 in the sample (Fig. 1d). In Fig. 1e and 1f, polymeric surface-active agents, such as PEG6000 and PVP, were applied in order to obtain pure Cs2SnI6 with high crystallinity. Furthermore, Cs2SnI6 and SnI4 were formed as a result of EDTA acting as a chelating agent. The pure phase of Cs2SnI6 was obtained under three different conditions, including the absence of surfactants and the presence of polymeric agents such as PVP and PEG6000. Based on the Scherrer equation, the crystal size of the samples was determined to be between 36 and 54 nm (Table 1). (Karami et al., 2021; Tahir et al., 2020).
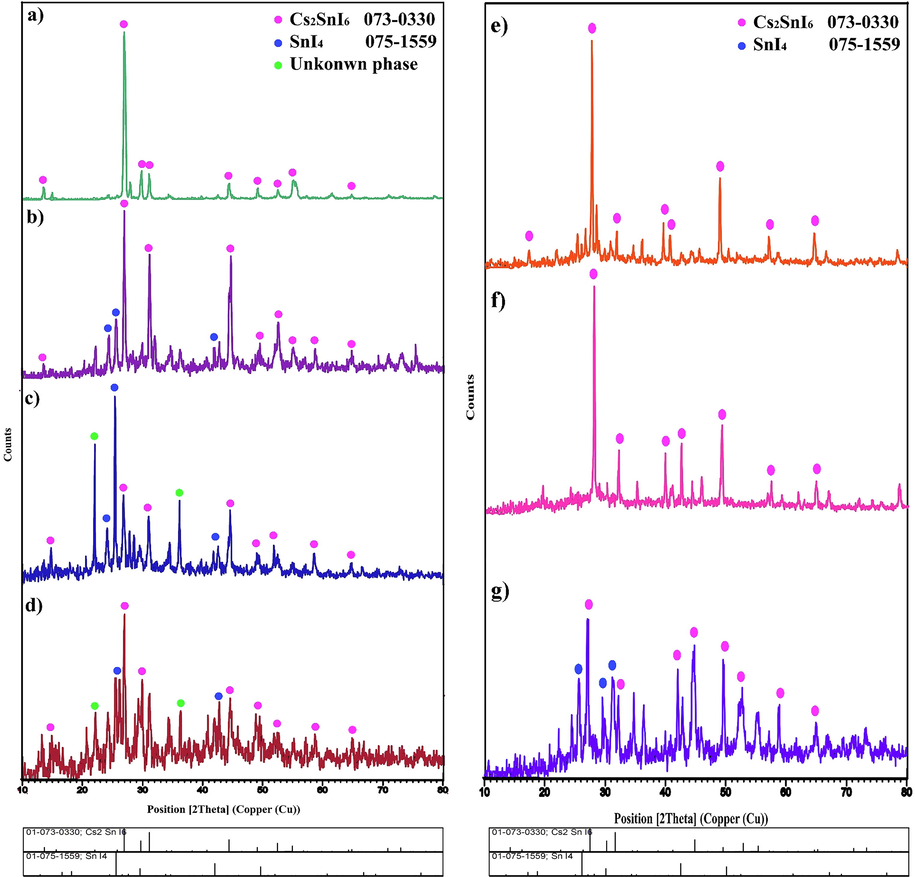
XRD patterns of Cs2SnI6 nanostructures (samples 1–7).
Sample No.
Type of Capping Agent
Products
Crystal Size (nm)
1
–
Cs2SnI6
40.1
2
SDS
Cs2SnI6, SnI4
40.7
3
SDBS
Cs2SnI6, SnI4, Unknown
43.2
4
NaHSal
Cs2SnI6, SnI4, Unknown
51.3
5
PEG6000
Cs2SnI6
36.6
6
PVP
Cs2SnI6
48.7
7
EDTA
Cs2SnI6, SnI4
54.6
The morphology of the synthesized nanoparticles was examined using SEM. A variety of surfactants were used to prepare nanoparticles, as previously mentioned. It was expected that the morphologies of nanoparticles would differ due to the use of different types of surfactants. The images in Fig. 2a and b illustrate synthesized nanoparticles without surfactants. Particles are arranged in a regular pattern and are in different polyhedral states. As can be seen in the 200 nm scale image, the polyhedral structures are formed by tiny nanoparticles. In this sample, a bulk nanostructure is observed. In contrast, different surfactants formed agglomerates of considerable size, contrary to expectations. In the case of SDS, an anionic surfactant (Fig. 2c and d), flakes were formed without being properly distributed. In the presence of SDBS, an anionic surfactant, small nanoparticles with some agglomerations were obtained (Fig. 2e and f). PEG-6000, an anionic (polymeric) surfactant, was used to produce bulk nanostructures in Fig. 2g and h. Further, when PVP was used as a polymeric surfactant, the d-spheres appeared quite clear (Fig. 2i and j). As in sample 1, tiny particles are observed on the surface of the nanoparticles that form the sphere in this sample (sample 6). EDTA was also used as a chelating agent in the preparation of the bulk nanostructures (Fig. 2k and l). As a result of the use of various surfactants, diverse morphologies are produced with larger sizes and irregular shapes. The synthesis of nanostructures with controlled size, shape, and aspect ratio has made extensive use of surfactants of different ionic phases (cationic, anionic, and non-ionic) (Carswell et al., 2003). In general, surfactants are thought of as capping agents, structure guiding agents, or templates. Every surfactant has its own mechanism for preparing nanostructures. During the fabrication procedure, surfactants are adsorbed to the growing crystal and, depending on the starting material concentration and surfactant characteristics, they can limit the growth rate of crystal faces, thereby assisting in the regulation of crystal sizes and shapes (Santra et al., 2001). It is possible that some surfactants may adversely affect the structure and morphology of products. It is possible that the lack of particles with the appropriate morphology and size is due to the inability to remove surfactants through calcination at high temperatures. The HRTEM images of Cs2SnI6 are shown in Fig. 3. In accordance with SEM results, the particles are arranged in a regular pattern. These photographs clearly demonstrate the coexistence of tiny nanoparticles. In order to demonstrate the high crystallinity of Cs2SnI6, parallel lines were specified for the crystal planes. The lattice planes are presented by the interplanar space of 3.2 Å, which conforms to the crystal planes (2 2 2) of Cubic Cs2SnI6 crystals.
FESEM micrographs of Cs2SnI6 nanostructures (samples 1–7).

TEM micrographs of Cs2SnI6 nanostructures (sample 1).
Similarly, Fig. 4(a-e) illustrates the EDS mapping images and EDS spectrum of Cs2SnI6. As shown in Fig. 4(a-d), the related maps of I, Sn, and Cs elements exhibit glossy zones similar to the calcification zones. It is evident from the EDS spectrum of Cs2SnI6 that the product contains I, Sn, and Cs elements, indicating its high purity. In this spectrum (Fig. 4e), the W% and A% of each element are clearly visible. It is also important to note that the Cs2SnI6 sample lacked any other elements. The Fig. 4f illustrates the N2 adsorption–desorption isotherm of Cs2SnI6, along with its associated pore size distribution profile. A microporous or mesoporous material is implied by the isotherm of type III (Karami et al., 2021). The isotherm exhibits a H3-type hysteresis loop, which corresponds to groove pores formed by flack particles in a nonrigid generation (Chen et al., 2018). Total pore volume at p/p0 = 0.989 was obtained at 0.018 cm3g−1, average porosity diameter was estimated at 17.6 nm, and specific aBET was achieved at 4.1 m2g−1. Fig. 4g displays the porous size distribution obtained from the desorption part of the isotherm via the BJH technique. It can be seen that the pore distribution of the generated Cs2SnI6 is multi-peak.
(a-d) EDS mapping, e) EDS spectrum, f) N2 adsorption/desorption isotherm, g) BJH plot, h) DRS spectrum, i) (αhυ)2 vs. hυ plot, and j) cyclic voltammetry of Cs2SnI6 nanostructures (sample 1).
DRS spectrum of Cs2SnI6 (sample 1) is shown in Fig. 4h. The absorption peaks from 200 to 400 nm verify the semiconducting properties of the prepared Cs2SnI6. The calculated bandgap was determined using the following equation (Ghanbari & Salavati-Niasari, 2018):
that α is absorption constant; hυ is the energy of the photon (eV); r is 2 for the direct or 1/2 for the indirect transitions; Eg is bandgap, and β is constant. Fig. 4i indicates the plot of (αhυ)2 vs. hυ for Cs2SnI6 (sample 1). Bandgap was calculated by partially linear extrapolating this curve to a point (αhυ)2 = 0 to be 1.7 eV.
The CV diagram of Cs2SnI6 is shown in Fig. 4j. The CV characteristics curves was used to calculate the conduction band (CB) and the valence band (VB).
3.2 Photocatalytic behavior
To fully understand the photocatalytic process, several factors must be considered, including light source, dye types, pH, catalyst dosages, dye concentrations, etc. (Ghanbari & Salavati-Niasari, 2018). In order to assess the effect of visible irradiation on the degradation of MB at ambient temperature, i.e., photolysis, a control test was conducted first. The dye was rather degraded in visible light after 180 min of irradiation, as illustrated in Fig. 5a (about 6 percent). The experiment was then conducted in the dark in order to evaluate the adsorption of dye on the catalysts. A UV–Visible spectrometer was used following centrifugation to monitor the aliquot at 10 min intervals for 60 min. Adsorption capacity (Qt) is expressed as earlier by Ajibade et. al. (Ajibade & Oluwalana, 2021):
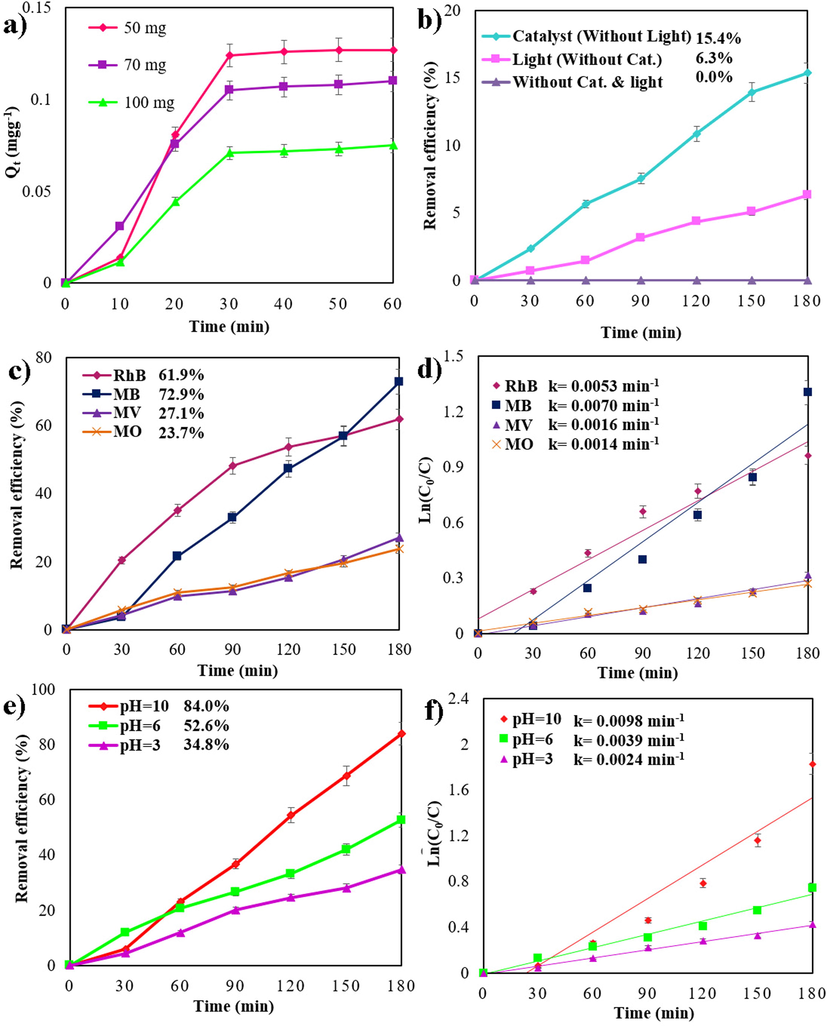
A) time profile adsorption capacity in different catalyst dosages, b) effect of light and catalyst on the photocatalytic degradation, photocatalytic degradation of different dyes over sample 1 and Plots of ln(C0/C) vs time (c and d), photocatalytic degradation in three different pH over sample 1 and Plots of ln(C0/C) vs time (e and f) under visible-light.
3.2.1 Organic pollutant types
An illustration of the influence of dye species on Cs2SnI6 nanoparticles can be found in Fig. 5c. The removal efficiency of MB, RhB, MV, and MO is approximately 72.9, 61.9, 27.1, and 23.7%, respectively. Based on the results of this study, it can be concluded that the efficiency of cationic organic pollutants is greater than that of anionic dyes, since iodide ions (I-) are negatively charged within the nanostructure. Fig.10a illustrates how pH solution affects MB discoloration.
3.2.2 Effect of pH of the solution
We examined the impact of the pH of the solution on the removal efficiency (Fig. 5e). In acidic (pH = 3), neutral (pH = 6), and alkaline (pH = 10) media, the degradation efficiency was approximately 34.8%, 52.6%, and 84.0%, respectively. Based on these results, alkaline media (pH = 10) showed the greatest increase in efficiency. Because methylene blue is a cationic dye, it acts efficiently at pH > 7 by increasing the concentration of OH groups in the solution. It is reasonable to assume that the concentration of OH groups will increase with increasing pH. In other words, the generation of OH in the solvent will be extended, and the photocatalytic efficiency will be enhanced.
3.2.3 Cs2SnI6 doses
Moreover, the removal efficiency yield of Cs2SnI6 (sample 1) was examined in relation to its dose. As noticed in Fig. As shown in Fig. 6a, the catalytic efficiency is largely dependent on the catalyst dosage. Changing the nanocatalyst dosage from 50 to 70 mg enhanced the degradation efficiency of Cs2SnI6 from 72.9 to 78.5%. By increasing the nanocatalyst dosage from 70 to 100 mg, the catalytic yield was increased from 78.5 to 84.0%. Therefore, increasing the Cu2HgI4 doses increases the surface area of the catalyst and enhances dye adsorption on Cs2SnI6.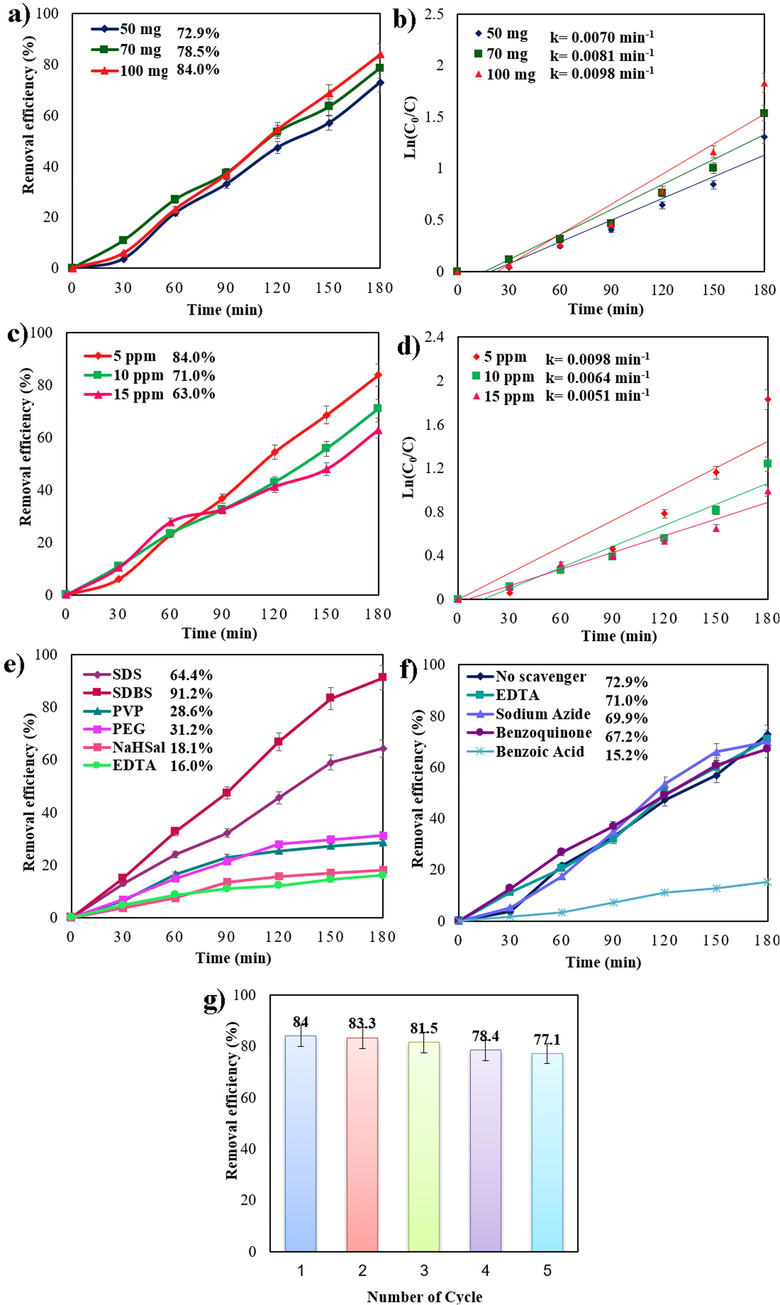
Photodegradation of various organic dyes over sample 1 and Plots of ln(C0/C) vs time in three different catalyst dosages (a and b), concentration of organic pollutants (c and d), different surfactants (e), effect of various scavengers (f), and cycling runs in the photocatalytic degradation of 5 ppm MB (g) under visible light.
3.2.4 Concentration of organic pollutants
Fig. 6c illustrates the effect of different dye concentrations (5, 10, and 15 ppm) of MB after 180 min. A decrease in dye concentration from 10 ppm to 5 ppm increases the degradation percentage from 71.0% to 84.0%, while an increase in dye concentration reduces dye decolorization. By increasing pollutant concentrations from 5 ppm to 15 ppm, the removal efficiency declined from 84.0 to 63.0%. The binding sites on the Cs2SnI6 surface are occupied by a large number of MB molecules at 15 ppm. It is evident that enhancing the dye concentration reduces the degradation percentage (de Luna et al., 2013).
3.2.5 Kinetic study
Langmuir–Hinshelwood mechanism was used to calculate the rate constants (eq.4) of the photocatalytic reaction of organic dyes (Kumari & Meena, 2020). Where k is the rate constant (min−1) for Pseudo-first order reaction; C0 is the primary concentration of contaminating solution; and C is the pollutant concentration at t time. The rate constant (k) for Pseudo-first order reaction has been defined from ln(C0/C) linear correlations versus time. Fig. 6b, 6d, and 6f indicate that the maximum photocatalytic degradation is obtained at a greater rate constant (k = 0.0098 min−1).
3.2.6 Effect of surfactant
Fig. 6e illustrates MB photodegradation in the presence of various surfactants. Due to the better interaction between MB, a cationic dye, and anionic surfactants, the removal efficiency is improved in the presence of anionic surfactants such as SDS (64.4%) and SDBS (91.2%). There are three factors that contribute to the excellent photocatalytic activity of Cs2SnI6 in the presence of SDBS: i) The presence of SnI4 with a band gap of 1.52 eV results in a narrower band gap. According to the XRD pattern of sample 3 (SDBS), SnI4 peak intensity is higher in sample 3 than in sample 2 (SDS), indicating that sample 3 contains a greater amount of SnI4. Due to the narrower bandgap, more radiation can be used to excite more charge carriers, leading to enhanced photocatalytic activity. The particles that are formed in the presence of SDBS are smaller than those formed by SDS, resulting in a larger specific surface area, as shown by the FESEM images of samples 2 and 3. It is generally accepted that a photocatalyst's specific surface area is closely associated with its photocatalytic activity. A higher surface area not only boosts the contact area between photocatalyst and the reactant, but it also supplies more active sites through the photocatalytic operation, which improves photocatalytic performance (Song et al., 2015). iii) smaller particle size. The photoinduced charge carriers have a shorter diffusion distance due to the smaller particle size, which causes them to migrate quickly to the catalyst surface and successfully separate (Peng et al., 2015).
3.2.7 Mechanism study through scavenger test
In aqueous catalyst suspensions, it has been completely demonstrated that valence band holes (h+) and conduction band electrons (e-) are produced when light energy exceeds the band gap of the catalyst (1.7 eV). Using photoinduced electrons, the colorant can be reduced or reacted with electron acceptors, such as O2 dissolved in water or adsorbed on the catalyst surface, thereby reducing it to O2•− (radical of superoxide anion). Photoinduced holes can react with H2O and OH– to form OH• radicals or oxidize organic dyes to produce R+ ions. As well as other high oxidant species (peroxide radicals), they are reported to be responsible for the photodegradation of organic dyes. Multiple initial reactive species, including 1O2, H•, h+, HO•, and O2•− can be created through photocatalytic degradation methods in UV–Vis/semiconductors. The formation of O2•− can prevent the recombination of photogenerated charge carriers. The HO• might be produced within the e− → O2•− → H2O2 → OH• way. Besides, the OH• radicals are created by several stages of reduction O2•− in the process. It was reported that the water separated in subsequent molecular layers and on the TiO2 surface has three roles (1) preserving charges (inhibiting recombination of electron-holes), (2) performing as an electron acceptor (creation of H atoms in a reaction of photoinduced electrons with protons on the surface, –OH2 + ), and (3) performing as an electron donor (reaction of water with photoinduced holes to produce OH• radicals). As claimed by prior investigations (Jiang et al., 2015), the principal active oxygen species produced through photocatalytic, and oxidation reactions are OH• and 1O2 radicals, respectively. Relying on the above thoughts, we can suggest that the possibility of creating OH• should be much higher than the O2•− formation. Nevertheless, OH• is a powerful, unselective oxidant that drives to the complete or incomplete mineralization of numerous organic compounds. According to the above statements, •OH, 1O2, and O2•− are the active species in the mechanism of photocatalytic degradation of organic dyes. Therefore, the literatures have been indicated that the reactive •OH performs the main role and, 1O2 and O2•− perform an insignificant role in the photocatalytic degradation of organic dyes (Tzeng et al., 2016). The •OH may be produced by an h+ with H2O. The 1O2 may be created by an h+ with O2−• species. The O2−• may be formed through an e− with O2 and/or 1O2 with e−species (Rahimzade et al., 2021). Therefore, •OH can help as the most essential active species in this research.
Scavenger experiments were accomplished by employing Benzoic acid (BA) for •OH, EDTA for h+, Benzoquinone (BQ) for •O2−, and sodium azide for 1O2 to demonstrate the function of active species in the degradation of organic dyes (Rahimzade et al., 2021). As noticed in Fig. 6f, the rate of photocatalytic decomposition decreased from 72.9% to 15.2% by adding benzoic acid to the mixture, confirming that hydroxyl radicals (•OH) are the most prominent active species in the photocatalytic reactions. Regardless, the photocatalytic efficiency declined from 72.9% to 67.2%, 71.0%, and 69.9%, respectively, by adding benzoquinone, EDTA, and sodium azide to the mixture separately, illustrating the impact of •O2−, h+, and 1O2 on the degradation of methylene blue was not significant. An electron-hole pair can be produced by illuminating visible light to the Cs2SnI6 photocatalyst (Sakthivel et al., 2003). Active hydroxyl radicals can be generated from the electron (e−) and hole (h+) reaction in the presence of water and oxygen molecules, which can recreate a role in the decolorization of organic pollutants (Wenderich & Mul, 2016). The possible mechanism to degrade methylene blue under visible light is as [33,46]:
3.2.8 Reusability and stability
Cs2SnI6 was tested for reusability in order to determine its stability. Catalysts were centrifuged, rinsed with water, parched at 70 °C for 18 h, and then reprocessed for five periods under the same conditions. Cs2SnI4 exhibits remarkable durability and maintains its total photocatalytic efficiency over five cycles, as shown in Fig. 6g. Over the course of the fifth test, the photocatalytic efficiency decreased by 6.9%. Fig. 7 shows the XRD pattern of the decolorized Cs2SnI6 nanostructures. This figure illustrates that all diffraction peaks are coordinated with Cs2SnI6 (reference code: 073–0330), which has a cubic crystal structure (space group: Fm-3 m). Following the destruction of organic colorants, it may be concluded that the host composition remains intact. Cs2SnI6 is therefore highly stable in an aqueous solution.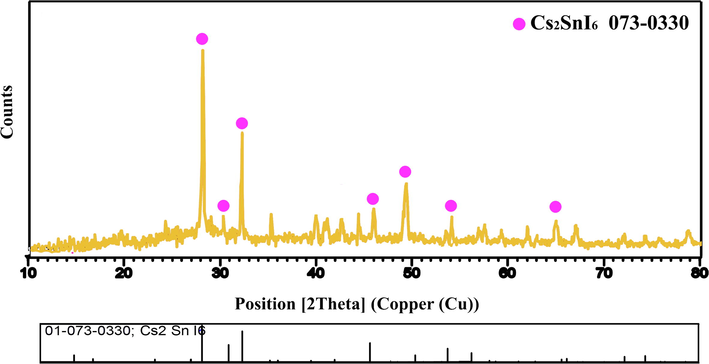
XRD pattern Cs2SnI6 nanostructures (sample 1) after photocatalytic reaction.
3.2.9 Comparison of photocatalytic behavior
Table 2 compares the photocatalytic efficiency of several iodide nanostructures. In conclusion, Cs2SnI6 nanostructures have the potential to degrade MB efficiently, and can compete with other iodide nanostructures as photocatalysts. It is possible to introduce Cs2SnI6 as a new nanophotocatalyst for the treatment of water.
Catalyst
Highest degradation (%)
Lowest degradation (%)
Catalyst dosage (mg)
Source of light
Ref.
Cs2SnI6 nanostructures
84.0 and 91.2% (MB)
23.7 (MO)
100
Vis
This work
Tl4HgI6 nanostructures
76.9 (RhB)
48.9 (ThB*)
70
UV
(Karami et al., 2021)
Cu2HgI4 nanostructures
92.4 (MO)
41.1 (RhB)
70
Vis
(Abkar et al., 2022)
Rb2HgI4 nanostructures
72.1 (AB1**)
48.1 (RhB)
70
Vis
(Abkar et al., 2021)
Tl4Cdl6 nanostructures
85.7 (AB1)
49.1 (MB)
50
UV
(Ghanbari & Salavati-Niasari, 2018)
Tl4PbI6 nanostructures
72.6 (ThB)
47.8 (RhB)
50
Vis
(Rahimzade et al., 2021)
Cu2CdI4/CuI nanocomposites
66.0 (MB)
29.1 (MO)
50
UV
(Ghanbari et al., 2016)
Ag2CdI4 nanostructures
95.3 (RhB)
57.1 (AB1)
50
UV
(Ghanbari et al., 2017b)
TlCdI3 nanostructures
94.6 (MB)
27.0 (MO)
50
UV
(Ghanbari et al., 2017a)
4 Future prospects
There has been extensive debate regarding recent developments in the fabrication of Cs2SnI6 nanostructures, with special emphasis on their photocatalytic properties. The presence of toxic organic dyes in wastewater generated by a variety of industries has produced undesirable contaminants in the environment. With a suitable bandgap in the visible area, Cs2SnI6 is a good candidate for removing toxic organic pollutants from wastewater.
5 Conclusions
A simple and rapid co-precipitation method was successfully used to fabricate Cs2SnI6 nanostructures. Based on the DRS results, Cs2SnI6 has a bandgap of approximately 1.7 eV, which is suitable for activation in the visible range. The photodecomposition of dyes revealed that Cs2SnI6 has a higher potential to decompose cationic dyes due to the presence of negatively charged iodide ions (I-) in the nanostructure. As a result of photocatalytic activity, Cs2SnI6 decomposed 5 ppm MB approximately 84.0% in an alkaline medium after 180 min below the visible spectrum. Due to the interaction between MB as a cationic dye and SDS, removal efficiency was enhanced in the presence of SDBS (91.2%) as an anionic surfactant. According to these results, Cs2SnI6 alone or in combination with other semiconductors performs excellently in the visible region as a photocatalyst for the purification of water in the future.
CRediT authorship contribution statement
Fatemeh Yousefzadeh: Formal analysis, Investigation, Software, Methodology, Formal analysis. Mojgan Ghanbari: Investigation, Supervision, Data curation, Writing – original draft, Writing – review & editing. Elmuez A. Dawi: Writing – review & editing, Software, Visualization, Data curation, Validation. Masoud Salavati-Niasari: Formal analysis, Methodology, Writing – review & editing, Writing – original draft, Conceptualization, Supervision, Project administration, Investigation, Data curation, Validation, Resources, Visualization.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Facile preparation and characterization of a novel visible-light-responsive Rb2HgI4 nanostructure photocatalyst. RSC Adv.. 2021;11(49):30849-30859.
- [CrossRef] [Google Scholar]
- Sonochemical synthesis and characterization of Cu2HgI4 nanostructures photocatalyst with enhanced visible light photocatalytic ability. Arab. J. Chem.. 2022;15(1):103536
- [Google Scholar]
- Structural and thermal dependence of normal-mode condensations in K2TeBr 6. Acta Crystallogr. B. 1989;45(1):26-34.
- [Google Scholar]
- Enhanced photocatalytic degradation of ternary dyes by copper sulfide nanoparticles. Nanomaterials. 2021;11(8):2000.
- [Google Scholar]
- Graphene oxide based semiconductor photocatalysts for degradation of organic dye in waste water: a review on fabrication, performance enhancement and challenges. Inorg. Chem. Commun.. 2021;129:108630
- [Google Scholar]
- Cation-induced band-gap tuning in organohalide perovskites: interplay of spin–orbit coupling and octahedra tilting. Nano Lett.. 2014;14(6):3608-3616.
- [Google Scholar]
- B-cation arrangements in double perovskites. Prog. Solid State Chem.. 1993;22(3):197-233.
- [Google Scholar]
- Recent developments in the use of heterogeneous semiconductor photocatalyst based materials for a visible-light-induced water-splitting system—A brief review. Catalysts. 2021;11(2):160.
- [Google Scholar]
- Adsorbed surfactants as templates for the synthesis of morphologically controlled polyaniline and polypyrrole nanostructures on flat surfaces: from spheres to wires to flat films. J. Am. Chem. Soc.. 2003;125(48):14793-14800.
- [Google Scholar]
- Model construction of micro-pores in shale: a case study of Silurian Longmaxi Formation shale in Dianqianbei area, SW China. Petroleum Exploration Dev.. 2018;45(3):412-421.
- [CrossRef] [Google Scholar]
- CsSnI3: semiconductor or metal? High electrical conductivity and strong near-infrared photoluminescence from a single material. High hole mobility and phase-transitions. J. Am. Chem. Soc.. 2012;134(20):8579-8587.
- [Google Scholar]
- Adsorption of Eriochrome Black T (EBT) dye using activated carbon prepared from waste rice hulls—Optimization, isotherm and kinetic studies. J. Taiwan Inst. Chem. Eng.. 2013;44(4):646-653.
- [CrossRef] [Google Scholar]
- Effective adsorption of anionic dye, alizarin red S, from aqueous solutions on activated clay modified by iron oxide. Ind. Eng. Chem. Res.. 2011;50(16):9712-9717.
- [Google Scholar]
- Simple synthesis-controlled fabrication of thallium cadmium iodide nanostructures via a novel route and photocatalytic investigation in degradation of toxic dyes. Inorg. Chim. Acta. 2017;455:88-97.
- [Google Scholar]
- Synthesis and characterization of Ag2CdI4 nanoparticles and photo-degradation of organic dyes. J. Mater. Sci. Mater. Electron.. 2017;28(8):6272-6277.
- [Google Scholar]
- Tl4CdI6 nanostructures: facile sonochemical synthesis and photocatalytic activity for removal of organic dyes. Inorg. Chem.. 2018;57(18):11443-11455.
- [Google Scholar]
- Synthesis of different morphologies of Cu2CdI4/CuI nanocomposite via simple hydrothermal method. J. Mater. Sci. Mater. Electron.. 2016;27(10):11092-11101.
- [Google Scholar]
- Elastic constants and softening of acoustic modes in A2MX6-crystals observed by Brillouin scattering. Zeitschrift für Physik B Condensed Matter. 1980;37(4):321-332.
- [Google Scholar]
- Step towards the sustainable toxic dyes removal and recycling from aqueous solution-A comprehensive review. Resour. Conserv. Recycl.. 2021;175:105849
- [Google Scholar]
- Controlled hydrothermal synthesis of BiOxCly/BiOmIn composites exhibiting visible-light photocatalytic degradation of crystal violet. J. Hazard. Mater.. 2015;283:787-805.
- [Google Scholar]
- Facile fabrication of Tl 4 HgI 6 nanostructures as novel antibacterial and antibiofilm agents and photocatalysts in the degradation of organic pollutants. Inorg. Chem. Front.. 2021;8(10):2442-2460.
- [Google Scholar]
- State-of-the-art developments in carbon-based metal nanocomposites as a catalyst: photocatalysis. Nanoscale Advances. 2021;3(7):1887-1900.
- [Google Scholar]
- Enhanced toxic dye removal from wastewater using biodegradable polymeric natural adsorbent. J. Mol. Liq.. 2021;328:115468
- [Google Scholar]
- Biosorption: the removal of toxic dyes from industrial effluent using phytobiomass-A review. Plant Arch. 2021;21:1320-1325.
- [Google Scholar]
- Green synthesis of gold nanoparticles from Lawsoniainermis and its catalytic activities following the Langmuir-Hinshelwood mechanism. Colloids Surf A Physicochem Eng Asp. 2020;606:125447
- [Google Scholar]
- Degradation of toxic dyes: a review. Int. J. Curr. Microbiol. App. Sci.. 2014;3(6):189-199.
- [Google Scholar]
- Air-stable molecular semiconducting iodosalts for solar cell applications: Cs2SnI6 as a hole conductor. J. Am. Chem. Soc.. 2014;136(43):15379-15385.
- [Google Scholar]
- Designing oxygen vacancy mediated bismuth molybdate (Bi2MoO6)/N-rich carbon nitride (C3N5) S-scheme heterojunctions for boosted photocatalytic removal of tetracycline antibiotic and Cr (VI): Intermediate toxicity and mechanism insight. J. Colloid Interface Sci. 2022
- [Google Scholar]
- Photocatalytic degradation of tetracycline antibiotic by a novel Bi2Sn2O7/Bi2MoO6 S-scheme heterojunction: performance, mechanism insight and toxicity assessment. Chem. Eng. J.. 2022;429:132519
- [Google Scholar]
- Facile fabrication of hierarchical porous TiO2 hollow microspheres with high photocatalytic activity for water purification. Appl Catal B. 2011;104(3–4):234-238.
- [Google Scholar]
- Defect tolerance to intolerance in the vacancy-ordered double perovskite semiconductors Cs2SnI6 and Cs2TeI6. J. Am. Chem. Soc.. 2016;138(27):8453-8464.
- [Google Scholar]
- Selective contacts drive charge extraction in quantum dot solids via asymmetry in carrier transfer kinetics. Nat. Commun.. 2013;4(1):1-9.
- [Google Scholar]
- Utilization of tea wastes for the removal of toxic dyes from polluted water—a review. Biomass Convers. Biorefin. 2021:1-17.
- [Google Scholar]
- On the similarity and dissimilarity between photocatalytic water splitting and photocatalytic degradation of pollutants. ChemPhysChem. 2013;14(10):2059-2070.
- [Google Scholar]
- Controlled synthesis of thin BiOCl nanosheets with exposed 001 facets and enhanced photocatalytic activities. CrstEngComm. 2015;17(20):3845-3851.
- [Google Scholar]
- Top-down strategy toward versatile graphene quantum dots for organic/inorganic hybrid solar cells. ACS Sustain. Chem. Eng.. 2015;3(4):637-644.
- [Google Scholar]
- Lead-free mesoscopic Cs2SnI6 perovskite solar cells using different nanostructured ZnO nanorods as electron transport layers. physica status solidi (RRL)–Rapid Res. Lett.. 2016;10(8):587-591.
- [Google Scholar]
- Simple preparation of chitosan-coated thallium lead iodide nanostructures as a new visible-light photocatalyst in decolorization of organic contamination. J. Mol. Liq.. 2021;341:117299
- [CrossRef] [Google Scholar]
- A first-principle study of the electronic, mechanical and optical properties of inorganic perovskite Cs2SnI6 for intermediate-band solar cells. Mater. Lett.. 2018;218:233-236.
- [Google Scholar]
- Solar photocatalytic degradation of azo dye: comparison of photocatalytic efficiency of ZnO and TiO2. Sol. Energy Mater. Sol. Cells. 2003;77(1):65-82.
- [CrossRef] [Google Scholar]
- Synthesis and characterization of silica-coated iron oxide nanoparticles in microemulsion: the effect of nonionic surfactants. Langmuir. 2001;17(10):2900-2906.
- [Google Scholar]
- Thin-film deposition and characterization of a Sn-deficient perovskite derivative Cs2SnI6. Chem. Mater.. 2016;28(7):2315-2322.
- [Google Scholar]
- Sustainable fabrication of silver-titania nanocomposites using goji berry (Lycium barbarum L.) fruit extract and their photocatalytic and antibacterial applications. Arab. J. Chem.. 2021;14(12):103456
- [Google Scholar]
- Surface state-mediated charge transfer of Cs2SnI6 and its application in dye-sensitized solar cells. Adv. Energy Mater.. 2019;9(3):1803243.
- [Google Scholar]
- Enhanced photocatalytic activity of cadmium-doped Bi2WO6 nanoparticles under simulated solar light. J. Nanopart. Res.. 2015;17(3):1-10.
- [Google Scholar]
- Preparation and characterization of g-C3N4/Ag–TiO2 ternary hollowsphere nanoheterojunction catalyst with high visible light photocatalytic performance. J. Alloy. Compd.. 2020;823:153851
- [Google Scholar]
- From soft harmonic phonons to fast relaxational dynamics in CH3NH3PbBr3. Phys. Rev. B. 2015;92(10):100303
- [Google Scholar]
- Photocatalytic degradation and hydrogen evolution using bismuth tungstate based nanocomposites under visible light irradiation. Int. J. Hydrogen Energy. 2020;45(43):22833-22847.
- [Google Scholar]
- Photolysis and photocatalytic decomposition of sulfamethazine antibiotics in an aqueous solution with TiO2. RSC Adv.. 2016;6(73):69301-69310.
- [Google Scholar]
- Removal of hazardous dyes, toxic metal ions and organic pollutants from wastewater by using porous hyper-cross-linked polymeric materials: a review of recent advances. J. Environ. Manage.. 2021;287:112360
- [Google Scholar]
- In situ construction of a Cs2SnI6 perovskite nanocrystal/SnS2 nanosheet heterojunction with boosted interfacial charge transfer. J. Am. Chem. Soc.. 2019;141(34):13434-13441.
- [Google Scholar]
- Methods, mechanism, and applications of photodeposition in photocatalysis: a review. Chem. Rev.. 2016;116(23):14587-14619.
- [Google Scholar]
- Constructing supramolecular self-assembled porous g-C3N4 nanosheets containing thiophene-groups for excellent photocatalytic performance under visible light. Appl. Surf. Sci.. 2022;578:152064
- [Google Scholar]
- Anatase TiO2 nanoparticles immobilized on ZnO tetrapods as a highly efficient and easily recyclable photocatalyst. Appl Catal B. 2007;76(1–2):168-173.
- [Google Scholar]
- Achievement of giant cryogenic refrigerant capacity in quinary rare-earths based high-entropy amorphous alloy. J. Mater. Sci. Technol.. 2022;102:66-71.
- [Google Scholar]
- Single-atom Pt-I3 sites on all-inorganic Cs2SnI6 perovskite for efficient photocatalytic hydrogen production. Nat. Commun.. 2021;12(1):4412.
- [CrossRef] [Google Scholar]







