Translate this page into:
Degradation-fragmentation of marine plastic waste and their environmental implications: A critical review
⁎Corresponding authors at: FEWWS Program, Center for Sustainable Development, College of Arts and Sciences, Qatar University, P.O. Box: 2713, Doha, Qatar (J.N. Hahladakis); Environmental Science Program, Department of Biological and Environmental Sciences, College of Arts and Sciences, Qatar University, P.O. Box: 2713, Doha, Qatar (M.A. Al-Ghouti). john_chach@yahoo.gr (John N. Hahladakis), ichachladakis@qu.edu.qa (John N. Hahladakis), mohammad.alghouti@qu.edu.qa (Mohammad A. Al-Ghouti)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
This review critically evaluates the plastic accumulation challenges and their environmental (primarily) and human (secondarily) impacts. It also emphasizes on their degradation and fragmentation phenomena under marine conditions. In addition, it takes into account the leachability of the various chemical substances (additives) embedded in plastic products to improve their polymeric properties and extend their life. Regardless of their effectiveness in enhancing the polymeric function of plastic products, these additives can potentially contaminate air, soil, food, and water. Several findings have shown that, regardless of their types and sizes, plastics can be degraded and/or fragmented under marine conditions. Therefore, the estimation of fragmentation and degradation rates via a reliable developed model is required to better understand the marine environmental status. The main parameter, which is responsible for initiating the fragmentation of plastics, is sunlight/UV radiation. Yet, UV- radiation alone is not enough to fragment some plastic polymer types under marine conditions, additional factors are needed such as mechanical abrasion. It should be also mentioned that most current studies on plastic degradation and fragmentation centered on the primary stages of degradation. Thus, further studies are needed to better understand these phenomena and to identify their fate and environmental effects.
Keywords
Plastic waste
Degradation
Marine litter
Fragmentation
Leachate
Plastic additives
- BFRs
-
Brominated flame retardants
- BPA
-
Bisphenol A
- DDT
-
Dichlorodiphenyltrichloroethane
- DSC
-
Differential scanning calorimetry
- EDCs
-
Endocrine-disrupting chemicals
- EDS
-
Energy-dispersive X-ray spectroscopy
- GPC
-
Gel permeation chromatography
- HDPE
-
High-density polyethylene
- LDPE
-
Low-density polyethylene
- LEDCs
-
Less economically developed countries
- LLDPE
-
Linear low-density polyethylene
- MA
-
Mechanical abrasion
- MEDCs
-
More economically developed countries
- MPs
-
Microplastics
- MTBE
-
Methyl tert-butyl ether
- MW
-
Molecular weight
- NPs
-
Nanoplastics
- PAHs
-
Polycyclic aromatic hydrocarbons
- PBDEs
-
Polybrominated diphenyl ethers
- PCBs
-
Polychlorinated biphenyls
- PE
-
Polyethylene
- PET
-
Polyethylene terephthalate
- PM
-
Particulate matter
- PoLA
-
Potentially leaching additives
- POPs
-
Persistent organic pollutants
- PP
-
Polypropylene
- PPos
-
Plastic polymers
- PS
-
Polystyrene
- PU
-
Polyurethane
- PVC
-
Polyvinyl chloride
- PW
-
Plastic waste
- py-GC/MS
-
Pyrolysis-gas chromatography-mass spectrometry
- TOC
-
Total organic carbon
- UDC
-
United development company
- VOCs
-
Volatile organic compounds
- XRD
-
X-ray diffraction
- XRF
-
X-ray fluorescence
Abbreviations
1 Introduction
Plastics are one of the most fundamentally used materials worldwide due to their attractive physicochemical properties and characteristics, which include; weathering resistance, levity, and inexpensive transparent material. In addition, it does not require high maintenance. All these valuable properties help in facilitating the implementation of plastics into various activities including agricultural, mercantile, and industrial activities (Ali et al., 2021; Amobonye et al., 2021; Jenkins and Harrison, 2008). Unlike other types of solid materials, plastics have gained ultimate attention due to their extreme durability and stability, which are generated mainly from their polymeric structure (Rivard et al., 1995). Nevertheless, like any material, plastics have their own disadvantages. The main challenge in using this desired material is its degradability since the majority of plastics are non-degradable and their complete decomposition in landfills may take centuries to occur. These previously mentioned facts clearly elucidate the global current situation of plastic waste (PW) accumulation in the environment (Ali et al., 2021; Barnes et al., 2009; Matjašič et al., 2021). In addition, conventionally used monomers for synthesizing polymers of plastics such as propylene and ethylene are usually invented from fossil hydrocarbons. Moreover, their polymeric properties boost their resistance to being degraded microbially, and even their short-term existence in nature obstructs the production of innovative enzymes, which may have the capability to degrade the common polymers (Amobonye et al., 2021; Mueller, 2006).
In the 1940 s and 1950 s, plastics production started blooming due to the dramatic increase of industrialization, leading to a tremendous universal annual production which was projected to reach 367 million metric tons by 2020 (Tiseo, 2021). However, due to Coronavirus (COVID-19), plastic production declined by approximately 0.3% when compared to the previous year (Tiseo, 2021). Presently, the most dominantly used types of plastics in the market are mainly thermoplastics, such as polyvinyl chloride (PVC), polypropylene (PP), high-density polyethylene (HDPE), and low -and linear low-density polyethylene (LDPE and LLDPE). Besides, other commercially demanded plastics such as polystyrene (PS), polyurethane (PU), and polyethylene terephthalate (PET) (Ali et al., 2021; Gewert et al., 2015; Hahladakis et al., 2018; PlasticsEurope and Group, 2008). Plastics including PVC, polyethylene (PE), PP, and PS are generated from C-C backbone polymers, which exemplify 77% of the global market share. On the other hand, heteroatomic polymers (e.g., C-O backbone polymers), which are mainly PU and PET account for around 18% of the total sales in the plastics market (Ali et al., 2021; Danso et al., 2019; Gewert et al., 2015). The major universal challenge regarding the C-C backbone polymeric structure is the resistance to biodegradation, hydrolysis, and vulnerability to thermal oxidation (Ali et al., 2021; Krueger et al., 2015). However, polymers, which are heteroatomically structured can be possibly degraded via hydrolysis, photo-oxidation, and biodegradation (Gewert et al., 2015). Fig. 1 illustrates these main types of plastics and their structure-based classification.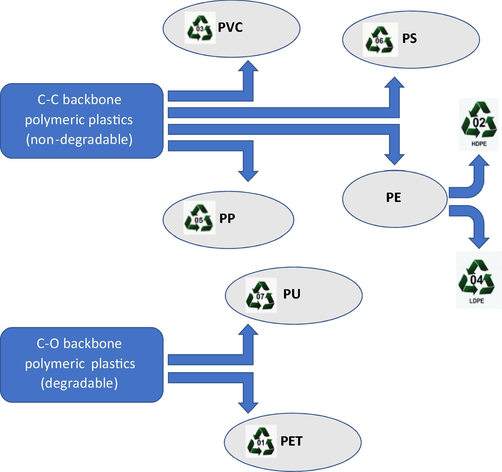
Most utilized plastic types and their structure-based categorization.
Plastic polymers (PPos) usage is not restricted to commercial consumer products but is also used in plentiful applications such as foams, synthesized fibers, adhesives, and coatings (Engelhart, 2010); Hahladakis et al. (2018). The usage of plastics in Europe is generated from various sectors, predominantly from the packaging process, then the construction sector, followed by automotive, electronics, and several other sectors (PlasticsEurope and EPRO, 2008). The excessive use of plastics generates a huge amount of PWs. The universal plastic manufacture was projected to surpass 8.3 billion metric tons from 1950 to 2018, with an annual increase of 5%, which is around 185 million tons of plastics (Ali et al., 2021; Amobonye et al., 2021; Geyer et al., 2017; Jambeck et al., 2015). Nevertheless, about 76% of these manufactured plastics end up as PW which will be further landfilled or emitted into the environment (72%), recycled (14%), and combusted (14%) (Ali et al., 2021; Schulze, 2016). Thus, PWs accumulation will pollute the environment and continuously pose an environmental threat, whilst impacting public health and natural ecosystems (Shah et al., 2008). Moreover, the inadequate disposal of PWs coupled with an impoverished waste management infrastructure and insufficient implementation of recycling technologies which is mainly due to a lack of public awareness have provided PWs as a leading component of marine debris (Hahladakis, 2020). Furthermore, there are multidimensional environmental impacts of terrestrial PWs. However, this plastic litter will eventually end up in the oceans since it is the ultimate sink in our globe (Jambeck et al., 2018; Jambeck et al., 2015).
Plastic materials may be categorized into three groups depending on the size of the particles. Plastics with a size of more than 5 mm are classified as “macroplastics” (Axelsson and van Sebille, 2017). In turn, plastics can potentially undergo degradation and fragmentation processes under various environmental abiotic conditions, e.g., UV light, pH, salinity, and temperature. These processes will lead to tinier pieces of plastics known as “microplastics” which are ranged between 50 μm and 5 mm (Andrady, 2011; Kalogerakis et al., 2017; Wang et al., 2016). The last category of plastics is the “nanoplastics” which are usually characterized by being lesser than 100 nm in particle size (Hahladakis, 2020; Koelmans et al., 2015). Microplastics (MPs) and nanoplastics (NPs) are deemed the most deleterious to humans and living organisms, particularly when present under marine conditions. This is owing to numerous complications, such as the ease of their ingestion by several species (Browne et al., 2008; Hahladakis, 2020; Steer et al., 2017; Teuten et al., 2009), and b) the release of chemical compounds embedded in the polymers (known as additives) that occurs during the degradation-fragmentation of bigger particles. Moreover, any potential accumulation of these fine pieces is a major concern since they are characterized as persistent organic pollutants (POPs) that will further burden humans, living organisms (especially marine), and the environment, considering the potential carcinogenicity of the chemicals they bear (Chen et al., 2019b; Hahladakis, 2020; Hahladakis et al., 2018; Koelmans et al., 2013).
Currently, there is no effective environmentally friendly technique, which is used to treat PWs. However, various reports highlighted the prospective microbial degradation of PWs (Ali et al., 2021; Sarkhel et al., 2019; Yoshida et al., 2016; Zhang et al., 2020). The biodegradation process of plastics in the marine environment involves microorganisms since they are efficient in the degradation of several synthetic polymers such as LDPE, PP, LLDPE, and HDPE. In addition, recent studies included novel isolated microbes, which can effectively degrade PWs in the marine environment, as well as some innovative concepts in which specific enzymes can be cloned to support the degradation and enhance the efficiency (Anjana et al., 2020; Premraj et al., 2004). There are many factors, which affect the degradation and fragmentation process of PW under marine conditions including, but not limited to the polymer characteristics (e.g., high molecular weight, hydrophobicity, and structure). All of these characteristics inhibit its accessibility to microorganisms (Anjana et al., 2020; Hadad et al., 2005). In addition to the effect of the marine environmental conditions, which are biotic (e.g., enzymes, which are secreted by microbes, and hydrophobic characteristics of the polymer) and abiotic factors (e.g., UV, temperature, salinity, and pH) (Ahmed et al., 2018; Anjana et al., 2020). Moreover, there are some additional dynamic factors related to the plastic additives which can influence the degradation of PW such as crystallinity, high molar mass, chain configuration, functional group types, and molecular orientation (Anjana et al., 2020; Min' et al., 2015).
This review aims to a) critically review the degradation and fragmentation of marine PW under biotic and abiotic factors and its environmental implications, b) investigate the complication and/or leaching caused by the presence of several additives embedded in plastics, and c) scrutinize the effect of the various properties of the plastic types (e.g., durability, flexibility, lightweight) on the overall degradation/fragmentation processes. Finally, future recommendations which can be implemented to address the PW issue in the marine environment will be suggested.
2 Methodology
A systematic literature review was performed to critically analyze the degradation and fragmentation of plastic material under marine conditions. In addition, the plastic material implications were reviewed, as well as the different additives and potentially toxic substances/chemicals, which are embedded in them. The data collected was performed mainly by searching various scientific literature databases (ScienceDirect, and Scopus). A list of references was obtained by using different syntax combinations, under which the research papers were searched and carefully chosen. The syntax combinations were “plastic, AND (degradation OR fragmentation), AND marine)”, “plastics, AND degradation, AND marine, AND litter, AND additives, AND release)”. Afterward, the search was mainly focused on very recent publications ranging between 2015 and 2022, which showed results ranging between 301 and 1,086 articles depending on the combination of keywords used and the database used for the search. Subsequently, around 256 scientific research papers were considered based on content, and objectives. On the other hand, several papers were excluded based on an exclusion criterion (e.g., studies addressing only terrestrial plastic degradation-fragmentation, all studies that did not investigate the degradation-fragmentation in aquatic environments and/or the complication caused by the presence of plastic additives).
3 Marine plastic degradation - fragmentation and factors affecting the process
3.1 Degradation of PW
PW can be degraded via physicochemical degradation also known as abiotic degradation, and through biodegradation in which the physical forces of the polymers are primarily broken down (Kyrikou and Briassoulis, 2007). There are several plastic degradation mechanisms in the environment which have been reported, mainly including thermo-oxidative degradation, photodegradation, biodegradation, and hydrolytic degradation (Andrady, 2011). Naturally, the degradation of PWs begins with photodegradation, then by hydrolysis process, and it goes through a thermo-oxidation process. All these reactions result in breaking the PWs into compounds with lower molecular weight (MW), which may be consequently degraded through the activity of microbes (Andrady, 2011; Webb et al., 2013). Yet, this mechanism is extremely slow and it may require centuries to take place completely (Chamas et al., 2020).
3.1.1 Degradation based on the characteristics of the plastic polymer
The plastic degradation process is defined by several environmental circumstances, as well as the physicochemical polymeric characteristics of the PWs as illustrated in Fig. 2. The physicochemical features of PPos play a crucial role in the process of degradation. The vulnerability of PWs to degradation biotically and abiotically depends on the polymer chain length and the backbone composition of the PPos, as such longer carbon chain (e.g., PP), can give the polymer more resistance to degradation (Fotopoulou and Karapanagioti, 2019; Huerta Lwanga et al., 2016). Nevertheless, incorporating heteroatoms as in PU and PET in which oxygen is integrated into their polymers gives the plastic the susceptibility to degrade thermally and biologically (Singh and Sharma, 2008). In addition, the hydrophobic property of the polymer highly influences the efficiency of the degradation process, as the rate of degradation rise with the increase of the hydrophobicity (Padsalgikar, 2017). Moreover, the degradation rate varies depending on the crystallinity of the polymer (Jenkins and Harrison, 2008), which means the more crystalline the structure of the polymer, the more O2 and H2O are required to degrade the plastic. Thus, the higher the MW and/or the crystallinity degree, the lower the rate of degradation (Jenkins and Harrison, 2008). In contrast, attacking an amorphous structure of polymers is via oxygen and water. In addition, the amorphous regions of the polymers are considerably affected by thermal oxidation (Li et al., 2019). In this regard, the MW of the polymer may easily influence the rate of degradation, the higher the MW of the polymer, the slower the rate of degradation owing to its relatively low surface area (Singh and Sharma, 2008). It is also interesting to mention that the production techniques of plastics and the types of additives utilized extremely influence the rate of degradation of plastic products. For example, PP which is manufactured via Ziegler-Natta catalyzation or bulk polymerization is distinguished by its high photodegradation susceptibility compared to PP which is co-polymerized (Tang et al., 2005). Moreover, various additives including stabilizers are mainly utilized to reduce the chromophores which are the hydroperoxide and the carbonyl groups and to lower the rate of degradation (Aldas et al., 2018). As such, the presence of these chromophores results in the photochemical creation of radicals, which helps in the initiation of photodegradation due to the availability of several photooxidative sites. In addition, the existence of metal-to-metal bonds can enhance the process of photodegradation due to the cleavage of the homolytic bond when irradiated (Daglen and Tyler, 2010). Furthermore, the morphological characteristics of the PW have the ability to increase the rate of degradation based on the favorable surface type for biofilm creation. Rough surfaces are found to be the most suitable types of surfaces for creating a biofilm. Thus, the rate of PW degradation increases whenever a rough surface appears (Booth et al., 2018).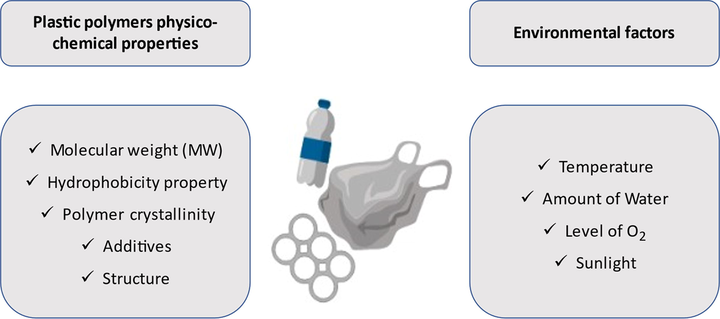
Factors influencing the PW degradation rate.
3.1.2 Degradation based on environmental factors
The climatic conditions, the geographical positioning, and the various pollutants can potentially influence the techniques and degradation rate of plastics (Andrady et al., 2003). Sunlight is one of the most crucial factors which affect plastic degradation. When the intensity of light increases the rate of the photooxidation process also increases, and thus the rate of plastic degradation increases (Kitamoto et al., 2011). Moreover, the abiotic degradation rate also rises with the elevation of temperature (Pischedda et al., 2019), with a duplication of the process rate per 10 °C of increase. The temperature has the ability to influence the mobility of the polymer chain, which will consequently influence its enzymatic activity through microbial degradation, in addition to its effect on the rate of hydrolysis reaction due to influencing the creation of the free radicals, humidity, and the rate of oxygen diffusion (Booth et al., 2018). A previous study shows that a PET chain separation was much greater in 100% humidity and 60 °C conditions by around 500% compared with 45% of relative humidity (Edge et al., 1991). In addition, the photodegradation of PP was improved by the increase in humidity (Fernando et al., 2009), as well as in PE (Jin et al., 2006), and PVC plastic-type (James et al., 2013), coupled with the increase of the concentration of hydroxyl radicals. Nevertheless, a decrease in the intensity of the UV light in the sea will cause a reduction in the rate of photodegradation. However, the increase in humidity level at the surface of the sea will boost the photodegradation reaction due to the solubility of particular photo-stabilizers in the seawater, which leads to efficient degradation (Booth et al., 2018). Moreover, the accessibility of O2 has an influence on the rate of plastic degradation through biodegradation and photodegradation (Queste et al., 2013). It was stated that the degradation process of polymers is speeded up when high levels of oxygen are present due to the swift process between the O2 and carbon radicals, which are released from the products which are initially degraded (Price and Horrocks, 2013). Furthermore, the presence of water is crucial during the degradation process due to the hydrolysis which results in the cleavage of functional groups and thus leads to the polymeric chain cleavage (Pitt, 1992).
3.2 Marine plastic waste degradation
It is appealing to realize that extensively utilized plastics do not naturally degrade to a huge extent whenever it is discarded into the environment (Bonhomme et al., 2003; Marqués-Calvo et al., 2006; Yamada-Onodera et al., 2001; Zheng et al., 2005). This is maybe predictable since one of the major reasons for the extensive application of several polymers is mainly the exceptional properties including extreme durability and stability (Yamada-Onodera et al., 2001; Zheng et al., 2005).
“Degradation” is the most utilized term for the description of the processes which result in the deterioration of the polymer properties (Yousif and Haddad, 2013). The polymeric structure of the plastics can be degraded via either biotic or abiotic pathways (Strlic and Kolar, 2005). For instance, polymers can be completely or partially broken down due to oxygen attack, UV radiation, and/or biological effect, leading to changes in properties (e.g., fragmentation, surface cracks, and discoloration) (Maes et al., 2021). The smaller the fragmented polymer through abiotic degradation, the easier it can pass via the cellular membranes and thus it can pass through the biodegradation by the cellular enzymes within the cells of the microbes. Nevertheless, some of the microbes have an extracellular excretion of enzymes which can potentially degrade a variety of PPos (Shah et al., 2008). The degradation of plastics is mostly initiated at the polymer surface level since it is mainly exposed and thus, it can be easily enzymatically or chemically attacked. Consequently, the MPs degradation is much swifter than the macroplastics due to its larger surface-to-volume ratio. Initially, the degradation of the polymers can be visually observed due to some visual effects such as the surface crazing and the color changes) (Vasile, 2000). When the surface of the plastics is cracked, the further degradation of the interior of the plastic material will be easier, leading to fragmentation and embrittlement (Vasile, 2000). Generally, PWs in the environment are subjected to various conditions, which can stimulate the weathering process.
Yet, a question that usually comes up is: are all types of polymers being degraded effectively? The answer is simply no! Not all types of degradation pathways can degrade all types of polymers in an efficient way. The plastic degradation challenge has been investigated by several authors. The degradation of the most common polymer types was identified to investigate the degradation pathway of the plastics, which are mainly floating in the marine environment. Several findings were found while reviewing the degradation of plastics studies, thus speculative extrapolations of these findings were done in this review. Yet, due to the fact that the smaller the plastic particle, the higher its probability to sink and be unobserved. This leads to a challenge in the investigation, testing, and quantification of these plastic fragments, which are facing much more pressure leading to their further fragmentation. Therefore, most of the literature findings mainly report evidence related to the PWs, which are floating in the marine environment. These plastic particles, which are floating on the surface of the oceans, are mainly subjected to several conditions including solar radiation (with a λ of 300 nm and above), oxidizing circumstances, and moderate temperature. The most crucial factors which initiate the abiotic degradation are mainly the sunlight and the oxygen availability since the temperature level is moderate (Pritchard, 1998). Moreover, few PPos can undergo a hydrolysis process (Gewert et al., 2015).
The degradation process is a chemical alteration, which significantly decreases the average MW of the PPos. Any extent of considerable degradation can potentially diminish the plastic product due to the high dependence of the plastic mechanical integrity consistency on the elevated average MW. Thus, the extensive degradation of the plastics will lead to brittle plastic material, which will eventually be converted to powdery fragments. The more the MW of the PPos is decreased, the more the plastic material gets brittle! (Summers and Rabinovitch, 1999). In addition, it provides a larger surface area for additional reactions, which makes its fragmentation susceptibility much higher. Those smaller fragmented particles may not be even visible to the naked eye due to their tiny size, which creates difficulty in their collection and investigation. The degradation process is usually categorized into several classifications based on the main cause of the degradation. For an example, biodegradation is mainly caused by the living organisms (e.g., microbes), and thermo-oxidation degradation is caused by the slow breaking down of the PPos through oxidation reactions at moderate temperatures. In addition, hydrolysis is caused due to water reacting; the degradation thermally is caused mainly due to high temperatures, and the photodegradation is usually due to the outdoor exposure of light. Generally, the UV-B sunlight radiation is the main initiator for the photo-oxidative degradation of the commonly used PPos including PP, HDPE, nylons, and LDPE, which are under marine environmental conditions. Then, the degradation process keeps proceeding with the thermo-oxidative process for a while without being exposed to more UV radiation. In addition, the catalytic reaction of polymer degradation will keep progressing in the presence of oxygen. The degradation process leads to a big decrease in the MW of the polymer in addition to the generation of oxygen-rich functional groups. While comparing light-induced oxidation and other types of degradation processes, light-induced oxidation was found to be swifter. Moreover, the hydrolysis mechanism is not a largely considered process in seawater. All types of biomaterials (e.g., plastics) can be invariably degraded under marine environmental conditions; however, the rate of degradation is slower. This is applicable to benthic sediments as well. In contrast to the oxidative degradation which is induced by light. PW degradation, which is initiated by UV sunlight, was found to be an extremely effective process when compared to plastics, which are exposed to air or discarded onshore/nearshore. However, while comparing an identical PW which is exposed to UV-radiation (sunlight) at the same location, when placing it in seawater, the degradation rates were found to be extremely slower (Andrady, 2011). It should be noted that PW degradation in the marine environment is slower due to the unoptimized conditions for the degradation of polymers. Yet, biodegradation and abiotic degradation are still most likely to happen but on a large timing scale (Gewert et al., 2015).
Usually, the degradation of plastics is initiated via photodegradation, leading to thermo-oxidative degradation. The UV light produced by the sun has a crucial effect since it is providing the needed energy. This helps in initiating the integration of O2 atoms into the polymer (Andrady, 2011; Raquez et al., 2011), and leads to the brittleness of the PWs. This eventually causes its fragmentation into smaller pieces and tinier pieces until reaching an extremely low MW of these polymer chains, which then can be metabolized via microorganisms (Andrady, 2011; Webb et al., 2013; Zheng et al., 2005). The microbes have the ability to convert the carbon, which is located in the polymer chain into CO2, or it can be also incorporated into the biomolecules (Andrady, 2011; Webb et al., 2013; Yamada-Onodera et al., 2001). Nevertheless, the whole process is extremely slow, and it may take over 50 years for plastic materials to be completely degraded (Müller et al., 2001). Moreover, the photodegradation is drastically declining in seawater conditions owing to the oxygen availability, the lesser temperature, and the hydrolysis rate of the PPos in the ocean which is extremely inconsequential (Andrady, 2011; Webb et al., 2013). Usually, UV-B sunlight radiation is the main initiator of photo-oxidative degradation in the marine environment, especially with the commonly utilized polymers which include PP, HDPE, and LDPE (Andrady, 2011).
PW degradation is mainly depending on the rate of chemical, biological, and physical conditions to which plastics are exposed, where they start to lose their original characteristics. The plastic degradation phenomenon involves 6 main mechanism categories: hydrolysis, thermal degradation, physical degradation, biodegradation, photodegradation, and thermo-oxidative degradation (Maes et al., 2021; Mattsson et al., 2015). In addition, most of these degradation processes occur in seawater at varied rates.
Plastic degradation via sunlight (UV-light) exposure, which is also called photodegradation, results in the “chain scission” phenomenon, which is mainly the breaking down of covalent bonds within the structure of PPos (Gewert et al., 2015). Chain scission can happen at any place within the polymeric structure, leading to monomers. It should be mentioned that some PPos may be hazardous, bioaccumulative, and persistent pollutants, which may potentially harm our environment (Lithner et al., 2011). In other words, the photodegradation of plastics is mainly the decomposition of PPos through the absorption of energy from light to produce free radicals. It is also known as “photooxidative degradation” whenever oxygen is present. Generally, the major light source which is initiating the photodegradation is the near UV light, which is ranging from 290 nm to 400 nm (Singh and Sharma, 2008). By exposing plastics to this light range, and due to the high energy of the C-C bond in the polymeric chain, it starts cleaving (Ravelli et al., 2016). Compared to other techniques of degradation, photodegradation technology is advantageous due to the ability to localize and control the light spatiotemporally in a simplistic, independent, and green approach (Chatani et al., 2014). The photodegradation process is not only beneficial in the usage of natural sunlight to degrade PPos, however, it also decreases the time needed for consequent biodegradation while providing the prospect of complete plastic degradation (Zhang et al., 2021). Photodegradation of plastics includes various mechanisms, for instance, secondary oxidative reactions and crosslinking, and chain scission, which occurs by generating and transferring free radicals (Bracco et al., 2018). There are two major techniques in which free radicals can be generated. The first method is directly irradiating the plastic with UV light without interfering with any catalysts, additives, or chromophores. The second method is based on adding photoactive substances into PPos to accelerate the production of free radicals (Zhang et al., 2021). These photoactive substances comprise photosensitive groups and photoactive fillers, which are combined into polymeric chains. Studies showed that these photoactive additives could be categorized into two main categories, which are, additives, which sacrifice themselves during the photodegradation process in which they produce free radicals when exposed to UV irradiation through decomposition reactions, named as the additive photosensitizer. The other type of additives is mainly photoactive additives which do not sacrifice themselves while constantly producing free radicals, and are known as the additive photocatalyst (Grassie and Scott, 1988). To better understand the plastic degradation mechanism through UV- light irradiation, it should be noted that the UV-light wavelength range which helps initiate the bond cleavage of polymeric structures of C-C or C-H bonds is 280–420 nm (Pospisil and Nespurek, 2000). Since the UV wavelengths, which correspond to the dissociation process of bond energies (C-H bond 420 kJ/mol and C-C bond, 375 kJ/mol) are 290 nm and 320 nm. This leads to the breakage of C-C and C-H bonds and the formation of free radicals (Pospisil and Nespurek, 2000), as illustrated in the initiation part of the process in Fig. 3 (A), where C-C bond was broken when exposed to UV-light radiation at a wavelength shorter than 320 nm, leading to the formation of alkyl radical R.. Afterwards, the propagation step, where the autooxidation begin, as the radical R. reacts with the oxygen and forms ROOֹ. Then, a further reaction occurs between the radical ROO. and polymer to produce hydroperoxide ROOH and R.. Subsequently, ROO. and R. reacts by disproportionation reaction or also known as radical coupling, resulting in the termination step of the photodegradation process. In the photooxidative process, ROOH groups (hydroperoxide) are considered the most crucial intermediates in this extremely photolabile process. Usually, the termination step of the photodegradation reaction combine peroxyl radicals or alkyl radical to produce stable products as illustrated in Fig. 3 (A). Generally, the combined macroalkyl radicals can form branched, disproportionated, or crosslinked products, on the other hand, peroxyl radicals are ultimately undergoing the termination reaction with other free radicals, forming dialkyl peroxides, alcohol, or carbonyl species (Mark and Kroschwitz, 1985). In addition, the oxygen pressure is correlated to the type of termination step (Yousif and Haddad, 2013). In other words, whenever polymer is exposed to light with a wavelength shorter than 320 nm, PPos are supposed to be degraded. It should be noted that the stratosphere act as a filter for UV- light with higher energy, which is ranged between 280 nm and 315 nm, and only less than 2% of this UV- light is reaching our earth, which is ranged between 315 nm and 400 nm (Yousif and Haddad, 2013). Under sunlight irradiation condition, polymers, which mainly consist of C-H and C-C bonds such as PP and PE, have an extremely slow rate of degradation. However, the addition of other chemical bonds can cause a different initiation reaction which in turn can speed up the degradation of the polymer (Wang et al., 2019).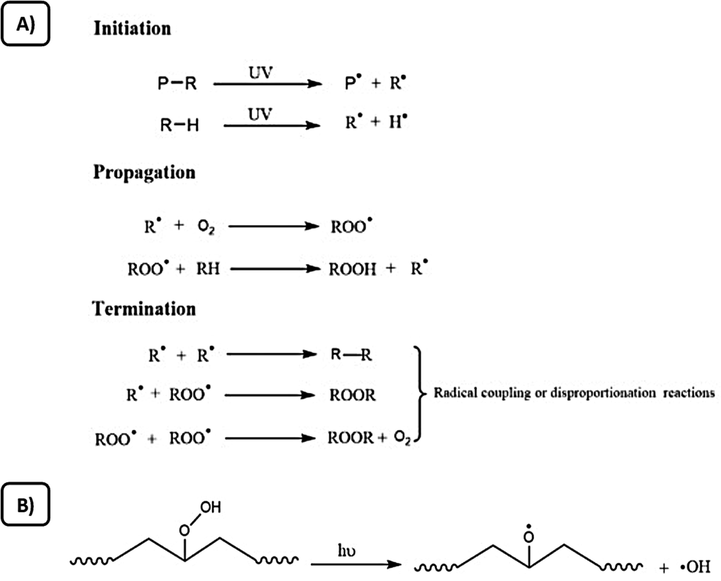
(A) Photodegradation process of polymers via UV- light irradiation (initiation, propagation, and termination). (B) Bond cleavage (O-O) of hydroperoxides mechanism. Modified from (Zhang et al., 2021).
It should be noted that thermodegradation of plastics is a different process, which aims to facilitate successive microbial degradation via degrading/fragmenting macromolecular polymers into smaller particles. When comparing between pyrolysis process and the thermodegradation process, the temperature is much lower in the thermodegradation process than in pyrolysis. In addition, thermodegradation occurs in the presence of oxygen. Thus, the thermodegradation process is correspondent to thermo-oxidation degradation, which means the mechanism of thermo-oxidation degradation and photooxidation are almost similar to each other, as illustrated in Fig. 3 (B). Where polyolefins can produce hydroperoxide group in the presence of oxygen and heating conditions, leading to the breakage of O—O bond of the group and forming hydroxyl radicals and alkoxy groups, and then degradation continues by forming lactone and esters through the autooxidation reactions (Chiellini et al., 2006). Nevertheless, the main difference between thermooxidation and photooxidation is the instability of ketones to light, since the ketonic products are usually thermally stable, yet they are not stable to light (Wiles and Scott, 2006). A study investigated the thermooxidation degradation of PE and showed that the temperature factor is the main factor influencing the degradation rate, rather than the oxygen concentration (Jakubowicz, 2003). In addition, another study concluded that the thermooxidation degradation of plastics (e.g., PE) can improve the degradation efficiency (Khabbaz et al., 1999). It should be also mentioned that the thermooxidation technique helps in significantly reduce the temperature required for the process compared to the pyrolysis technique, thus reducing the equipment needed as well. Nevertheless, the degradation of plastics thermooxidatively requires more time, leading to further consumption of energy compared to the photodegradation process. Therefore, further research should be done focusing on developing techniques to accelerate thermooxidation degradation, as well as to reduce energy consumption.
3.3 Common commercial plastics and their degradation
While understanding the degradation process of plastics, it should be noted that there are two major groups of plastics based on the main chain bonds composition, which are C-C backbone plastics such as PP, PE, PVC, and PS, and heteroatomic plastics which are mainly PU and PET (Gewert et al., 2015). Since the reduction of MW of the polymers leads to a brittle material (Summers and Rabinovitch, 1999), which will increase its susceptibility to being fragmented, leading to larger surface areas.
Polyethylene (PE)
PE (structure shown in Fig. 4) can be abiotically oxidized through UV-radiation exposure, which is the rate-determining step for its environmental degradation (Hakkarainen and Albertsson, 2004; Peacock, 2000; Singh and Sharma, 2008; Vasile and Pascu, 2005). There are several popular products, which can be degraded such as ethane, ethene, propane, propene, hexane, and butane (Vasile, 2000), which makes plastics materials susceptible to fragmentation and thus becomes more brittle (Hakkarainen and Albertsson, 2004). Microorganisms may initiate PE degradation by attacking any terminal methyl groups in the plastic material chemical structure (Booma et al., 1994). The smaller the MW, and particularly whenever it is less than 500 Da, the swifter the biodegradation process (Vasile and Pascu, 2005). In addition, the lifetime of PE is highly dependent on the additives, thus their degradation projections differ extensively (Gewert et al., 2015). A study calculated that a stabilized PE has a lifetime of around 15 years to 20 years in moderate temperatures (Vasile and Pascu, 2005). Moreover, it was computed in a research study that around 0.1% of the polymer carbon is transformed into carbon dioxide annually through the biodegradation process, this study was performed in the laboratory under the best-controlled conditions (Pritchard, 1997). Furthermore, the degradation process is much slower in the marine environment when compared to the terrestrial environment. Yet, the process is still possible via biodegradation and abiotic degradation but a longer time is required for the completer degradation of polymers (Gewert et al., 2015).
Structure of PE.
Polypropylene (PP)
Due to the tertiary carbon atoms in the backbone of its polymeric structure as shown in Fig. 5, PP is less stable than PE, leading to an increase in the susceptibility of being attacked abiotically while compared to the secondary carbon atoms which are present in PE (Beyler and Hirschler, 2002; Grassie and Scott, 1988). In addition, the process mechanisms are identical to PE. Moreover, the chemical structure of the polymer influences its biodegradability (Sayler et al., 2012). It should be noted that the chain branching also affects the polymer degradation, as it strengthens the resistance to aerobic biodegradation due to the existence of the PP tertiary carbon, thus the susceptibility to microbial degradation will be reduced (Sayler et al., 2012; Singh and Dwivedi, 2004).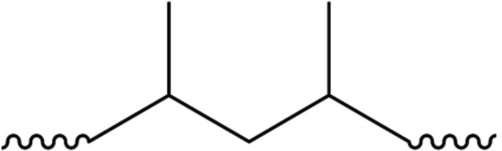
Structure of PP.
Polystyrene (PS)
PS has a higher susceptibility to outdoor weathering (Faber, 1979), especially when it is exposed to UV radiation, where the phenyl rings, as shown in Fig. 6, gets excited leading to transferring the released energy to the closest carbon-hydrogen bond, and thus a hydrogen cleavage will occur resulting in a polymer radical formation (Yousif and Haddad, 2013). The main volatile degradation products which have been identified are known as styrene monomers (Beyler and Hirschler, 2002). There are other products, which are known as oligomers of styrene. These products are produced from the thermooxidation process, and they may include phenol, benzene, ethylbenzene, benzaldehyde, benzoic acid, and ketones (Hoff et al., 1982). Photo-oxidation and thermooxidation processes have a similar degradation pathway. They only differ in their initiation. Therefore, these products can be potentially found as degradation products in the marine environment via the photooxidation process. Polystyrene is a thermoplastic polymer, which has extreme durability concerning biodegradation (Mor and Sivan, 2008). This clearly explains why the rapid biodegradation which is usually seen with PP and PE residues does not happen with this polymer type (Jones et al., 1974). Several additives can be incorporated into the polymer to enhance its physical characteristics depending on its usage (Gewert et al., 2015; Gurman et al., 1987). Moreover, other additions can promote the degradation of PS such as the starch addition (Kiatkamjornwong et al., 1999).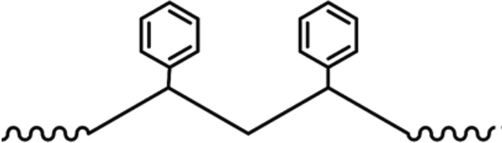
Structure of PS.
Polyvinyl chloride (PVC)
PVC (structure shown in Fig. 7) is one of the polymers which have the least stability compared to other types of polymers, due to its extreme UV radiation sensitivity (Nicholson, 2017; Owen, 1984). Thus, the photo-degradation process is highly significant for this type of polymer (Owen, 1984). When PVC is exposed to the sunlight, its polymer dechlorination will be initiated as a first step, leading to the creation of the conjugated double bond and the formation of polyene, as well as the hydrochloric acid, in addition to a tiny amount of various products (Gewert et al., 2015; Pielichowski and Njuguna, 2005). The dichlorination of PVC and the creation of polyene are schematically represented in Fig. 8 (the chemical reaction was drawn using ChemDraw Professional software). It was also reported that the dechlorination rate of PVC could be boosted under aerobic conditions, especially for polymers with lower MW, in the existence of hydrochloric acid (Beyler and Hirschler, 2002). In addition, the high-temperature levels may improve the degradation rate of PVC, as well as humidity and mechanical stress (Gewert et al., 2015; Jakubowicz et al., 1999). The unsaturated C = C double bonds which are formed have lower stability to photo-degradation which leads to the fragmentation of the polymer backbone into tinier fragments. The degradation of PVC can be visually observed through the discoloration of the polymer (Gewert et al., 2015; Nicholson, 2017). Moreover, it was reported that PVC is highly resistant to the biodegradation process (Summers and Rabinovitch, 1999). It should be also noted that halogens and mainly chlorine have the ability to enhance the resistance to aerobic biodegradation (Gewert et al., 2015). That clearly explains the dechlorination of the polymer (PVC) while biodegrading. Moreover, PVC can potentially incorporate an extensive range of plasticizers and additives, which will modify and enhance the properties of the plastic material (Bruma and Olabisi, 1997; Yoshioka et al., 2008). Nowadays, PVC is rarely utilized in its pure material, owing to the improvements in its characteristics when additives are added (Stringer and Johnston, 2001). To extend the stability and lifetime of the polymer, UV and thermal stabilizers are inserted into the PVC products which are usually subjected to sunlight (Nicholson, 2017). As well as, flexibilities or stabilizers which are incorporated into PVC to enhance its flexibility (Beyler and Hirschler, 2002). Over time, these stabilizers which are added to the various PVC products are expected to leach into the environment (Weschler, 2009). These stabilizers can migrate from the PVC polymer into the environment because they are not bonded chemically. In turn, they can be degraded via microbes (Stringer and Johnston, 2001).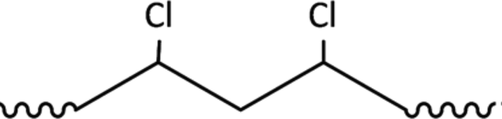
Structure of PVC.
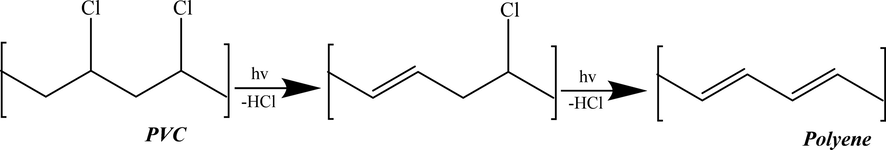
PVC dichlorination and the creation of polyene.
Polyethylene terephthalate (PET)
PET (structure shown in Fig. 9) which one of the widely used polymers in several commercial products. It can be degraded through various pathways under marine conditions including hydrolytic degradation and photo-oxidation. In addition, the photo-degradation process results in the cleavage of the ester bonds which leads to the formation of the vinyl and carboxylic acid groups, or radicals, which will ultimately end up as carboxylic acid groups (Fagerburg and Clauberg, 2004). These carboxylic acids which will be formed will act as a promoter for the degradation of the polymer thermo-oxidatively (Bikiaris and Karayannidis, 1999), as well as photo-oxidatively. PET is highly susceptible to hydrolytic degradation whenever it is under water conditions (Summers and Rabinovitch, 1999). Although it is known that the degradation of PET hydrolytically under room temperature is tremendously slow, it is considered as a crucial process in the degradation of this polymer, especially at minimal temperature (Allen et al., 1991; Venkatachalam et al., 2012). Usually, PET weathering under marine conditions takes place mainly through hydrolytic and photooxidation processes. The degradation of PET can be visually observed through the yellowing of this polymer (Venkatachalam et al., 2012). In the case of the thermo-oxidative degradation of the polymer, there are some main changes, which will occur such as the decrease in the MW of the polymer, as well as the increase in the carboxylic acid groups (Culbert and Christel, 2004). In addition, photo-oxidative degradation shares the same degradation pathway as well as similar results and/or findings. Moreover, the hydrolysis process of the polymer can also result in the increase of the carboxylic acid end groups and the decrease of the polymer MW (Gewert et al., 2015). Polyethylene terephthalate is found to be an extremely resistant polymer to environmental biodegradation (Webb et al., 2013), due to its consolidated structure (Zhang et al., 2004), however, a study captured some low/soft degradation of this polymer through lipase and microbes (Zhang et al., 2004).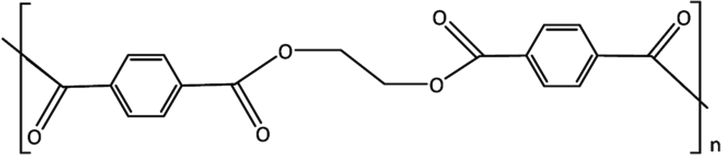
Structure of PET.
Overall, the degradation process of the polymers results in lower MW. Yet, it can lead to deeper effects, where the surface of the plastic material can be randomly cracked. All various types of polymers usually include additives. Almost none of the polymers is used in their pure form (Gewert et al., 2015). Therefore, the plastic degraded products will be liberated as weathered plastic chemicals to the ocean water, as well as the additives which are different chemical substances, which will be released into the environment (Deanin, 1975). All of these chemical substances will end up as environmental pollutants. Moreover, to improve the resistance of the polymers to degradation, stabilizers can be added to the plastic materials in different amounts depending on the final plastic products needed. Mostly, the degradation rates of plastics in the environment depend mainly on additives. Therefore, it is difficult to generalize a quantitative statement regarding the degradation rates because each product will be different in its composition. In addition, most of the experiments were mostly performed under various environmental and non-environmental conditions. Plastics, which are floating, can be protected from UV-light radiation through water, leading to a decrease in the photo-degradation. Furthermore, the condition of the plastic materials after weathering plays a significant role, because the plastic products, which have a smaller sizes will have higher surface areas, which makes them more susceptible to cracking. Thus, they are more susceptible to degradation.
The various polymer types utilized in plastics have different properties which can influence their behavior in several environments, including but not limited to buoyancy hydrophobic and hydrophilic characteristics, density, and tendency toward biodegradation (Maes et al., 2021). One of the most crucial factors in the marine environment is the density of plastics compared to seawater. Generally, the densities of conventional plastics have a range between 0.90 kg.m−3 to 1.39 kg.m−3 when compared to freshwater with a density of 1.0 (pure water), and a density range of 1.020 kg.m−3 to 1.029 kg.m−3 for seawater (Maes et al., 2021). For instance, PE, mainly LDPE, tends to have a density, which is lower than 1 kg.m−3, which makes this type float in both, marine and freshwater. This is the main reason behind being one of the most frequently found types of plastics whenever samples are collected from surface waters. In addition, PP has the tendency to float in freshwater, as well as expanded PS which would be expected to float in seawater (Maes et al., 2021). Furthermore, drinking bottles, which are made of PET are usually found both accumulated on the seabed and floating in the seawater due to the buoyancy effect, which is influenced by water currents, turbulence, and trapped air. Moreover, the buoyancy of PPos can be influenced by the existence of the biofilm, which is present on the surface of the plastics (Napper and Thompson, 2019).
3.4 Fragmentation of PW in the ocean
The vulnerability of the structure coupled with the loss of mechanical integrity may result in breaking the parent plastics into tinier pieces of plastics (Andrady, 2011; Ekvall et al., 2019; ter Halle et al., 2016). Intrinsically, the fragmentation process of plastics results in the alteration in the size distribution of plastic debris in aquatic ecosystems. The fragmentation process does not have the capability to eliminate marine plastic litter from the environment. Yet, it is efficient in accelerating the physicochemical and biochemical processes, which occur at the surface of the marine plastic litter owing to the higher surface-to-volume ratio of tinier pieces (ter Halle et al., 2016). Moreover, the fragmentation of plastic materials may be simulated by the mechanical stress in marine environmental conditions because of the wave action (Cooper and Corcoran, 2010; Weinstein et al., 2016). Moreover, this fragmentation process may be speeded via the potential biodegradation and weathering, which makes the plastic materials more brittle over time (Andrady, 2017; Barnes et al., 2009; Kalogerakis et al., 2017; ter Halle et al., 2016). The fragmentation rate increases as the size of the particle decrease. Additionally, marine plastic litter particles which are bigger than 2 mm have a morphology that looks like a sheet, which makes one face more likely to be exposed to sunlight (ter Halle et al., 2016). Therefore, the fragmentation process will be accelerated leading to the creation of cracks at the surface of the particle, which generates more cubic pieces that seems like the parent fragment (around 1 mm of thickness). Furthermore, the degradation of the cubic pieces will be much swifter due to their tendency to roll at the surface. Accelerated fragmentation may also be a valid justification for the relatively low masses of floating marine litter particles. The fragmentation of marine PW can lead to the generation of tinier particles (less than 1 µm), which are known as NPs.
The fragmentation process tends to influence the PW transport through marine systems since tinier particles are carried in a different way horizontally, as well as vertically than larger plastic pieces (Cole et al., 2011; Gerritse et al., 2020; Zhang, 2017). It should also be mentioned that smaller fragments have a relatively greater surface area compared to their volume, leading to increased rates of degradation, adsorption sites per unit, and decreased buoyancy. This is due to biofouling, which results in the transfer of MPs from the surface to the water–sediment or column (Cózar et al., 2014; Eriksen et al., 2014; Gewert et al., 2015; ter Halle et al., 2016; Zhang, 2017). Hence, the fragmentation process creates a larger specific surface area, leading to more water contact, and thus swifter sorption and/or leaching rates for chemicals (Velzeboer et al., 2014).
Macro- and microplastics are the most found PW types in the ocean, and their environmental impacts are clearly stated in many research papers. However, these plastics can also be degraded in the marine environment via several mechanisms mainly photo-oxidation coupled with microbial degradation. Consequently, the degradation of plastics in the ocean is expected to be the highest in the tropical and sub-tropical regions due to the high pollution levels where plastic marine litter is highly accumulated (Wayman and Niemann, 2021). Degradation and fragmentation mechanisms result in the transformation of larger plastics into smaller fragments, where macro and microplastics can be potentially transformed into NPs. In addition, the smaller the fragments, the higher their degradation due to the greater surface-to-volume ratio, leading to higher rates of degradation.
According to (ter Halle et al., 2016), there are two different behaviors of MPs in the marine environment due to their physicochemical properties. To understand the fragmentation of MPs, different samples (big parallelepipeds and small cubic) were analyzed through their physicochemical properties. It was noticed that the bigger sample (parallelepipeds) was floating at the sea surface while exposing only one surface to the sun as represented in Fig. 10. During sunlight exposure, the photodegradation process will induce fragmentation due to a decrease in mechanical properties. The fragmentation process takes place because of the breaking down near cracks on the surface of the plastic samples, resulting in smaller fragments. However, the cubic samples have the tendency to roll on the water surface. It was also confirmed that the continuous rotation of the cubic appeared to prevent the biofilm growth on the surface of the cube pieces, while edge erosion seemed more likely to happen. These variations in the motion behavior of PW are undoubtedly associated with different fragmentation rates. It was concluded that the cubic sample was fragmented faster than the larger parallelepipeds. Similarly, other studies showed that smaller plastic items can degrade faster (Andrady, 2015; Kershaw, 2015b). Overall, it should be highlighted that these investigations can significantly contribute to a better understanding of the fate of the MPs in oceans.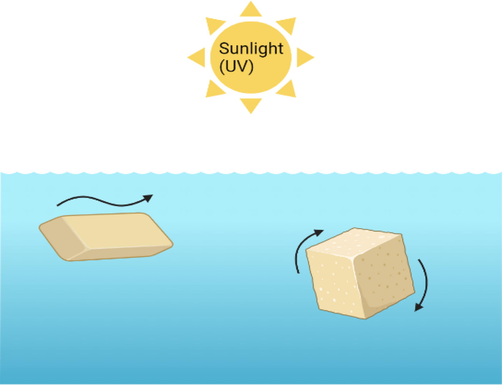
Seawater PW fragmentation based on the shape.
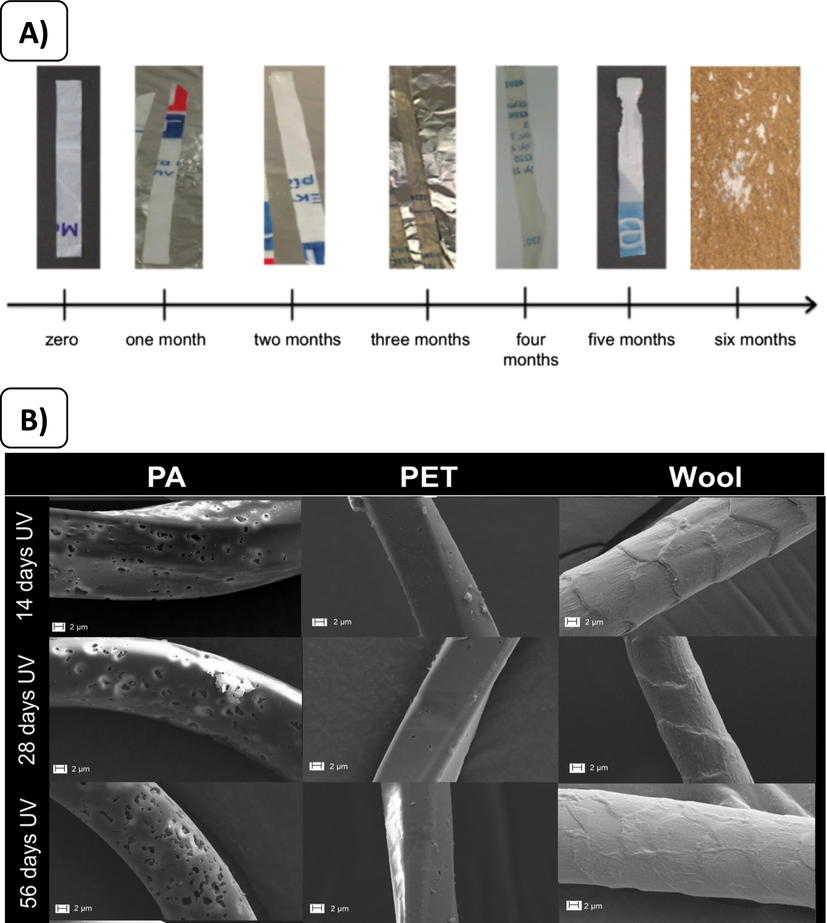
(A) Fragmentation of plastics (HDPE strips) under outdoor onshore (sand) conditions over an experimental period. Source: (Kalogerakis et al., 2017). (B) SEM microscopy of plastics (PET, wool, and PA) under UV exposure for 14, 28, and 56 days. Modified from: (Sørensen et al., 2021).
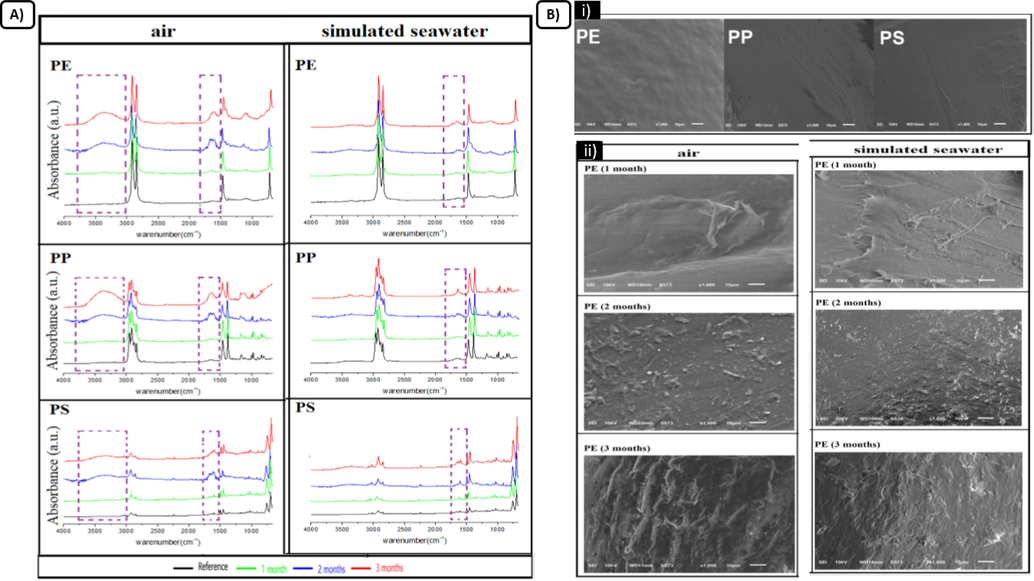
(A) FTIR spectra of PE, PP, and PS exposed to UV irradiation for 1 month, 2 months, and 3 months in two different environments. (B) SEM microscopy of PE, PP, and PS exposed to UV irradiation for 1 month, 2 months, and 3 months in two different environments. (i) SEM microscopy of the plastic types before exposure, (ii) SEM microscopy of PE after exposure in two different environments. Modified from: (Cai et al., 2018).
Recently, the creation of MPs and NPs through fragmentation has been investigated under marine conditions in several studies through accelerated laboratory conditions (photodegradation) (Gigault et al., 2016; Lambert and Wagner, 2016a; Lambert and Wagner, 2016b). However, many plastic fragments tend to be lost in the open exposure systems, leading to an underestimation of the real numbers of plastic fragments formed (Song et al., 2017). Thus, the collection of data through the fragmentation process of plastics in marine environments is needed to estimate the number, production rate, and size of the plastic particles developed by fragmentation.
The fragmentation process can occur through different factors including UV exposure and MA. (Song et al., 2017) shows that some polymer types can be affected by MA such as PS; however, others were unlikely to be fragmented via MA only. To be fragmented, an additional factor needs to be present as well; which is UV exposure/sunlight. These findings can be confirmed by the difference in the mechanical strength of PPos, where PS has lower mechanical strength compared to PP and PE. It was concluded that with the increase of UV exposure time and MA, the fragmentation of PPos increases, despite the fact that the fragmentation rate will differ based on the type of polymer. UV light acts as a producer for initial free radicals by dissociating C-H and C-C polymer backbone bonds (Feldman, 2002), leading to chemical changes, visible fractures and cracks, and surface embrittlement. It was also confirmed that due to chain reactions and cross-linking, UV oxidation can create surface embrittlement to a depth exceeding 100 μm (Feldman, 2002). However, it is interesting to mention that the photooxidation process, alone, cannot directly cause polymer fragmentation. PPos need to be exposed to successive MA as well to be fragmented. This means that an additional physical process is needed to improve the fragmentation of PPos. Some factors and conditions were found to be critical in the rate of photooxidation and propagation of PPos such as oxygen availability, diffusion of radicals, and the depth of UV penetration (Pilař et al., 2015). Furthermore, the rate and degree of fragmentation-degradation coupled with the environmental conditions are determined mainly by the physicochemical characteristics of PPos. In addition, the degree of fragmentation is also affected by the additives (e.g., antioxidants and UV stabilizers) embedded in plastics which hinder the process.
The fragmentation and degradation of several plastic materials were studied and investigated using various methods under different conditions as illustrated in Table 1. To critically evaluate, analyze, and interpret the differences between these studies or to compare their findings, several aspects need to be considered including polymer type, conditions/parameters, the exposure duration, plastic particle size, as well as the additives used in the plastic samples. Since all the aforementioned aspects can potentially affect the degradation and fragmentation process of plastic materials. When comparing two different studies, where scientists inspect the degradation of plastics under marine conditions, all parameters and exposure conditions need to be well explored, as well as the polymer type used, because each polymer type behaves differently from another even though, both, are polymers, but each polymer has its own unique polymeric properties and characteristics. (Ioakeimidis et al., 2016) studied the degradation of PET bottles under marine environmental conditions, where the size of the particles/bottles were unspecified, as well as the exposure duration of the plastics. These PET bottles were collected from the bottom of the sea for the first time. These samples were settled for many years at 150 m-350 m on the seafloor (Ioakeimidis et al., 2016). Collected samples were tested and characterized using ATR-FTIR and SEM analysis to visualize any degradation/fragmentation that occurred on the surface of the plastics. The main findings can be summarized in a significant decrease in the functional groups as shown in Fig. 13, and uneven surfaces and cracks were observed. It was also concluded that PET plastics could maintain their robustness for around 15 years. On the other hand, (Iñiguez et al., 2018) investigated the degradation and recyclability of four different common types of plastics (Nylon, PET, PP, and PE) from unknown items. They were cut into large pieces (area of 60 mm × 10 mm) and subjected to UV-radiation under marine environment conditions for 6.5 months (Iñiguez et al., 2018), then characterized through SEM, TGA, AFM, as well as the Instron universal instrument to check the mechanical properties. The main results can be briefly summarized as a decrease in the strength of the plastics and their elasticity due to the changes in their mechanical and thermal properties; granular oxidation, surface cracks, and flakes were observed, as well as a decrease in surface homogeneity. While focusing on the findings of the mutual type of plastics used in both aforementioned studies, which is PET, it can be deduced that both researchers could be correct, since they almost revealed approximately similar findings as they both showed degradation and fragmentation signs on the surface of the samples, even though the experimental set up was not the same. Yet, there are several major differences, which make each of these studies correct in terms of inspecting the degradation of PET despite any differences. Starting with the different origins of plastics used in the experiment, as some of the plastics were collected from the marine environment directly, which means they were actually under marine conditions, and some other unspecified items were purchased from supermarkets and put under marine conditions. In addition, there is an extreme difference between the exposure durations of both studies, yet even plastics, which were exposed to marine conditions for 6.5 months only showed a good visual degradation sign. Moreover, the additives, which are embedded into each of the plastic samples, were unspecified since plastic manufactural companies use various additives while producing plastic bottles. The authors claim that the rapid degradation illustrated in the findings of (Iñiguez et al., 2018) could be initiated due to the direct impact of some additives on the degradation of plastics. It was reported that certain additives might strengthen the recyclability and degradation of plastics (Pivnenko et al., 2016).
No.
Plastic type
Size of particle
Parameters and conditions investigated
Characterization
Findings
Ref
1
Nylon, PP, and PE
10 mm in diameter
PP, PE, and nylon exposed to benthic conditions at 10 m depth over 12 months
Tensile strength (mechanical properties), SEM, GLM analysis (mass loss)
Fastest fragmentation: Nylon rope, Slowest fragmentation: PP rope.
MP particles and fibers can be formed in benthic environments (in low photodegradation).(Welden and Cowie, 2017)
2
PU
MPs of fraction size (3 mm × 3 mm × 3 mm).
Leaching of additives in simulated acidic, saline, basic water, and in natural waters (river, lake, wetland, and seawater), Leaching time (0 h to 48 h), aged MPs (prior leaching exp) exposed to outdoor/sunlight condition for at least 12 months.
FT-IR and Raman spectroscopy analysis, Fluorescence excitation-emission matrix spectroscopy, and 3-DEEM (to characterize specific fluorescent substances).
The additives leaching process from MPs depends on water environments.
As leaching time and pH increase, the amount of additives released increases.
Leached substances may affect aquatic organisms.
(Luo et al., 2019)
3
PE
1 cm2 – Sample surface
In seawater for 15 days and 33 days at 3 m depth and seafloor at 6 m depth
SEM analysis, Dynamometer (tensile properties)
Degradation signs on the surface of the plastics.
Biofilm was significantly attached.(Eich et al., 2015)
4
PVC and PE
PE sample (2 cm × 2 cm × 10 μm, PVC sample (1 cm length,
4.8 cm2 surface area)Incubation in seawater under laboratory conditions (light/dark, temperature) exposure durations: 12 weeks.
FTIR, GC/MS, DOC, and SEM analysis.
PVC and PE samples significantly leached phthalates into the seawater, within the first month.
PE sample released more phthalates than the PVC.(Paluselli et al., 2019)
5
PP
PP plastic cup- unspecified size
Exposed to different depths of seawater (surface and at 50 cm and 170 cm of depth) for 60 days.
SEM-EDX, FTIR analysis, and microscope analysis
Organic carbon content decreased, erosion observed, discoloration and irregularity on the surface, and alteration in the organic functional groups of PP.
(Khoironi et al., 2020)
6
Nylon, PET, PP, and PE
Unspecified, area of the specimen (60 mm × 10 mm)
Exposed to UV-light in the seawater for 6.5 months
SEM, TGA, AFM, Mechanical properties test (Instron Universal Test instrument)
Decrease in the strength of plastics and their elasticity; granular oxidation, surface cracks, and flakes were observed; decrease in the surface homogeneity.
(Iñiguez et al., 2018)
7
PP
Bags shredded into 7 cm by 1 cm strips
In seawater for 40 weeks at 0.6 m depth.
Instron universal testing instrument, UV spectroradiometer
Less UV transmittance, decrease in tensile strength
(O’Brine and Thompson, 2010)
8
PET
Unspecified
On the seafloor at 150 m-350 m depth for many years (samples collected from the bottom of the sea)
SEM and ATR-FTIR analysis
Decreases in the functional groups, uneven surfaces, and cracks were observed.
(Ioakeimidis et al., 2016)
9
PE
Samples of 1 cm × 2 cm
In marine sediment for 98 days.
Raman, SEM, Goniometer
Decrease in contact angle, and biofilm was attached.
(Nauendorf et al., 2016)
10
PVC
Oval-shaped pellet samples with a diameter of ≈ 100 μm.
UV and Thermal exposure in seawater and air for 210 days.
FTIR, SEM-EDX, BET specific surface area analyzer.
Alteration in the surface area, pore size, functional groups, and morphologies.
(Tang et al., 2018)
11
PP, Polyamide (PA), PET, PE
Rectangular samples of 3.5 cm × 1.5 cm
Exposed to different conditions (seafloor aphotic, euphotic conditions) for 1 year
Differential Scanning Calorimetry (DSC), balance, FTIR
Decrease in weight, alteration in the crystallinity and melting enthalpy, alteration in the functional groups.
(Beltrán-Sanahuja et al., 2020)
12
HDPE
Samples of 1 cm × 20 cm
Exposure to environmental conditions (temperature and sunlight) in seawater and sand for 6 months.
MW measurements, tensile strength, weight loss, FTIR
60% reduction in the MW for 1 sample and the measurement was not possible for the other sample because of the extensive fragmentation, and significantly higher onshore (sand) fragmentation (Fig. 11 (A)) when compared to plastics in seawater.
(Kalogerakis et al., 2017)
13
PS, PP, and LDPE
Macroplastics- samples of 2 cm × 2 cm
The set-up was adjusted to simulate sea swash zone conditions by continuously mixing natural sediments and tap water, at room temperature, atmospheric pressure, and very moderate wave simulation for 24 hr.
Fluorescence microscope, Weight loss (balance), visual inspection.
Greater plastic mass fraction fragmented into MPs when the sediment is coarser.
(Chubarenko et al., 2020)
14
LDPE, PP, and expanded PS
Pellets- Unspecified size
Plastics were exposed to UV for up to 12 months, then subjected to mechanical abrasion (MA) with sand for 2 months.
FTIR, carbonyl index (CI), SEM, fluorescence microscopy
An increase in plastic fragmentation when exposed to UV and MA, a decrease in the size of particles, and a large portion (76.5%) of the PS were fragmented into undetectable fragments.
(Song et al., 2017)
15
LDPE
125 μm – 200 μm
fraction for the particle.Degradation of LDPE under controlled abiotic laboratory conditions (simulated solar radiation and water) for 2000 hrs.
FTIR, Gel Permeation Chromatography (GPC), SEM, Energy-dispersive X-ray Spectroscopy (EDS), Particle size analysis, Mechanical testing, NMR spectroscopy, DSC, and X-ray Diffraction (XRD).
Decrease in particle size, sharp edges, and broadening of the particle size distribution, a significant increase in the rate of secondary MPs, and production of oxygen-containing functional groups due to weathering of MPs.
(Menzel et al., 2022)
16
PP, PS, and PE
Pellets – Unspecified
UV exposure in various environmental conditions (seawater, ultrapure water, and air) for 3 months.
SEM, FTIR, and Raman
Alterations in functional groups, surface cracks, and flakes were observed as illustrated in Fig. 12.
(Cai et al., 2018)
17
PP
Pellets - diameter of 4 mm
Solar radiation, UV-B, and heat exposure in different simulated treatments (seawater-air, dry-darkness, seawater-darkness, dry-air) for 0.5 to 1.5 years.
SEM, and FTIR
Exposure to solar radiation showed 35% of cracks, and UV-B showed 12% of cracks. Floating PP illustrated less degradation compared to dry conditions.
(Tang et al., 2019)
18
PE, PP, and PS
MPs – 1– 5 mm
Irradiation and mechanical stirring exposure in simulated seawater for 72 hrs (stirring without irradiation) – 360 hrs (irradiated samples).
ATR-FTIR, micro-FTIR, DSC, TGA.
A high number of smaller MPs up to 105-106 items/mg of plastics.
(Sorasan et al., 2022)
19
PP (2 different food packaging materials)
MPs − 400–500 μm
Exposed to UV irradiation and mechanical stirring in artificial seawater for 12 days.
GC–MS, SEM-EDS, ATR-FTIR, Thermo gravimetry-differential scanning calorimetry analyzer (TG-DSC), XRD, and Total organic carbon (TOC) analyzer.
PP materials used show a longer aging time compared to pure PP. More micro- and nano-sized particles are produced when exposed to MA and UV in the marine environment. Antioxidant (Irgafos 168) in PP showed a considerable inhibitory impact on the degradation process of MPs in seawater.
(Wu et al., 2021)
20
PP
Pink/Blue Cartridges are about 70 mm × 37 mm × 11 mm in size, Black cartridges are about 70 mm × 37 mm × 15 mm
Exposed to marine environmental conditions for 41 months.
Pyrolysis-gas chromatography-mass spectrometry (py-GC–MS), Infrared (IR) analysis, SEM, CI, sand XRF.
Significant cracks, fractures, surface roughness, and discoloration were observed in highly heterogenous weathering, and extremely significant weathering due to the presence of titanium dioxide pigment.
(Scott et al., 2022)
21
PET, PA, and wool
∼2 mm in size
Exposure to marine environmental conditions UV irradiance (simulated sunlight) in seawater for 56 days.
SEM, and GC–MS
Changes in morphology were observed in PA, changes in the surface morphology and fragmentation were observed in PET and wool particles (Fig. 11 (B)), and leaching chemical substances were detected in seawater after UV exposure.
(Sørensen et al., 2021)
22
LDPE
115 mm × 19 mm × 3.4 mm
UV radiation exposure in artificial seawater and air at the same temperature for maximum exposure of 600 h for seawater (SW) samples and 1100 h for sand/air samples.
FTIR-ATR, optical microscope, and tensile properties.
No spectral signature of weathering is illustrated in FTIR for floating SW samples for 375hrs. Decrease in plastic degradation due to the lower O2 availability in SW while compared to air.
(Andrady et al., 2022)
23
Fibers, filaments, fragments, pellets, rubbers, films, and Paint sheets
Samples were collected by using a manta trawl net with a mesh size of 300 μm.
MPs found in the sediments from
Ría de Vigo, northwest Spain, during the upwelling and downwelling conditions at depth of up to 40 m,
stereomicroscope (magnifications of × 0.63 − × 8) coupled with an image analysis system,
205 plastic samples were collected near the surface (fibers (56%), paint sheets (15%), filaments (7%), and fragments (6%)). The remaining plastics were present in smaller proportions: films (4%), rubbers (2%), and pellets (1%).
(Díez-Minguito et al., 2020)
24
Paint coatings
2 different sizes: 7 × 7 cm2 and 20 × 20 cm2
Paint coatings were exposed to outdoor natural conditions in Morocco for 3 years.
FTIR-ATR and Colorimetric measurements
Changes in the colors were observed, (yellow and more matte color). FTIR-ATR findings showed several changes in the intensity of IR bands.
(Guerguer et al., 2021)
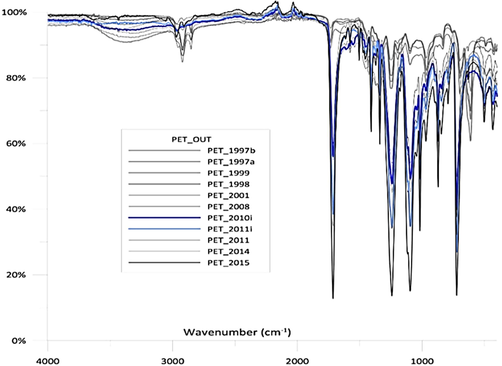
ATR-FTIR spectra of the degraded PET from the Saronikos Gulf (1997 (a and b), 1998, 1999, 2001, 2008, 2011, 2014) and the Ionian Sea (2010i, and 2011i) compared to the non-exposed sample (2015). Modified from: (Ioakeimidis et al., 2016).
It should be noted that paints contribute to marine plastic pollution. Due to the fact that paint pieces contain polymers combined with various additives (Turner, 2021). Nevertheless, paint fragments are usually undetected and consciously disregarded in the pool of MPs, especially in the marine environment. Because of the extensive literature on micro-litter. A study showed that paint coatings of different commercial solar glass mirrors were degraded after being exposed to marine outdoor conditions for 3 years (Guerguer et al., 2021). Changes in some colors were observed. In addition, FTIR-ATR findings showed several changes in the intensity of several IR bands. The reason behind this degradation is not limited to UV-radiation. It is also related to other conditions including salinity, temperature, rainfalls, and humidity. Nevertheless, the amounts of MPs released during the production, use, and disposal of paints are still not clear (Faber et al., 2021). Thus, further studies are needed to take measures to reduce emissions.
4 Potential leaching of plastic waste additives in the marine environment and their complications
Several studies show that PW particulates in marine ecosystems contain significant amounts of various organic pollutants and additives, which can potentially leach into seawater, leading to adverse harmful effects. Many toxic substances and compounds may encompass polychlorinated biphenyls (PCBs), nonylphenol, and organic pesticides, which may comprise dichlorodiphenyltrichloroethane (DDT), polycyclic aromatic hydrocarbons (PAHs), bisphenol A (BPA), and polybrominated diphenyl ethers (PBDEs). All of the aforementioned toxic chemicals have been constantly found in marine plastic litter (Hirai et al., 2011; Mato et al., 2001; Rios et al., 2007). While these toxic chemicals are present, the risks linked to the ingestion of the plastic litter by marine organisms, and the plentiful of these chemicals can be significantly biomagnified, leading to a potential direct risk to human well-being (Hirai et al., 2011). Many studies illustrated that these toxic compounds are associated with several health issues, which may include cancer, diabetes, developmental impairments (hormonal imbalance, neurological impairment, abnormalities in growth), endocrine disruption, DNA hypomethylation, breast cancer, neurobehavioral variations, arthritis (Chung et al., 2011; Schecter et al., 2010; Trudel et al., 2011; Zhou et al., 2011).
The state of the plastic materials after being exposed to several environmental conditions could be affecting their interactions with the environment as well as influencing the potential release of the additives and chemical substances which are embedded (UNEP, 2016). Most of the polymers are initially fabricated as thermostable materials, which means that they cannot break down easily to deliver the intended use of the product (UNEP, 2016). Due to the non-biodegradable property of plastics, their products are considered as persistent pollutants. Biodegradable plastics are accounting for a smaller share nowadays. Yet, these products are constantly expanding in the markets. Nevertheless, not all commercial plastic products are completely, environmentally biodegradable (Kershaw, 2015a; O’Brine and Thompson, 2010). In addition, some biodegradable products can potentially change the geochemistry of the sediments in the ocean and impact the marine species as well as their co-existence (Balestri et al., 2017). To improve the performance of plastic products, several additives are added to most plastic materials, mainly during the formation of the polymers and their shaping, which enhance their functions and aging characteristics. There are numerous additives, which can be used for various types of polymeric products, which include, but are not limited to, antioxidants, plasticizers, pigments, slip agents, thermal stabilizers, and lubricants (Hahladakis et al., 2018). Each of these additives has a distinctive role in improving the polymeric characteristics and the functionality of the final product. For example, pigments are commonly used to deliver a diversity of colors. In addition, slip agents are utilized usually to avoid the stick of the latter together or to the metal surface. Additives can be divided into several categories (Hansen et al., 2013), including functional additives, fillers, colorants, and reinforcements. It should be emphasized that most of these additives are not chemically bound to the polymeric plastic products, and only some additives, which are mainly organic reactive additives, will be part of the polymeric chain due to their polymerization with the plastic particles.
Toxic substances can be potentially leached from plastic products into water, air, and soil. Potentially leaching additives (PoLA) from plastic products to water, food, and air have been studied and detected through chemical analysis, which is mainly conducted in laboratories. The identification of the type and magnitude of the chemical leachate is a complicated task due to its dependence on several factors. Generally, in the short term as well as the long term, any chemical substance and/or degraded polymeric products will bioaccumulate in different environments due to their persistence, leading to potential harm to humans and the environment. The composition of the additives in the plastic products will define what can be leached from the product. Yet, there are several other factors, which are controlling the actual potential emission of these substances in any environment. The presence and/or release of PoLA and other substances in plastic products do not necessarily represent a hazard; there are many additional facets, which needs to be taken into consideration while assessing the risk posed to the receptors including human, animals, and the environment. It needs to be noted that the long-term release of potentially toxic substances into the environment is mitigated and controlled efficiently, where migration mechanisms are used to test the potential release of most of the additives. These are mostly embedded in plastic food products, as well as the leaching/release of PoLA throughout the recycling process of all plastic types (Velis and Brunner, 2013). Numerous research studies investigated the potential release of several additives from different plastic products such as BPA (Brede et al., 2003; Geens et al., 2010; Olea et al., 1996; Sajiki et al., 2007), phthalates (Rijk and Ehlert, 2001; Tønning et al., 2010), volatile organic compounds (e.g., benzene) (Skjevrak et al., 2003), bisphenol-A dimethacrylate (Olea et al., 1996), lead, cadmium, and tin (Al-Malack, 2001), brominated flame retardants (Kim et al., 2006), acetaldehyde and formaldehyde (Mutsuga et al., 2006), 4-nonylphenol (Fernandes et al., 2008; Loyo-Rosales et al., 2004), and several other volatile organic compounds including methyl tert-butyl ether (MTBE) (Henneuse-Boxus and Pacary, 2003). The concentrations of these released plastic products were found to be low in many of these research investigations when compared to legal standard/guideline limits, yet in some cases; these concentrations may be significantly higher. Conspicuously, the low levels at which endocrine-disrupting chemicals (EDCs) can potentially generate an impact was not included in the standard/guideline values (Vandenberg et al., 2012), as well as the mixtures toxicity/harmfulness (Kortenkamp, 2007).
The degradation of plastic products produced depends highly on the type of polymer (Ravve, 2000). In addition, the amount and type of degraded plastic products can be affected by the degradation pathways, as well as the existence of polymerized impurities and/or the environmental conditions including oxygen availability and temperature (La Mantia, 2002; Ravve, 2000). It should be also mentioned that plastics, which contain nitrogen such as nylons release hydrogen cyanide during thermal degradation, as well as plastic products, which contain chlorine such as PVC, which releases dioxins and hydrogen chloride (Hahladakis et al., 2018; Lokensgard, 2004; Ravve, 2000). Moreover, several polymers can depolymerize through chain scission such as polyoxymethylene, which is capable of the complete depolymerization into their initial monomers. Furthermore, there are some other polymers, which can partially depolymerize into their monomers, which include nylons, and polyesters such as polycarbonate and PET (La Mantia, 2002; Ravve, 2000).
The leachability of various chemical substances in seawater was investigated through several research studies. Recently, the leachability of EDCs associated with mesoplastics (large plastic particles which are commonly defined as 5 mm – 10 mm in range (Isobe et al., 2014)) and MPs were studied under various normal life conditions which are usually occurring during the life cycle of plastics (e.g., solar irradiation, autoclaving, microwaving) (Chen et al., 2019a). Findings showed that Estrogens were the main EDCs found on the plastic fragments, which either originated from plastic fabrication or leached out to the surrounding water. In addition, while using an average concentration, Bisphenol A showed the greatest detection frequency compared to other EDCs such as octylphenol, nonylphenol, and bisphenol S. Furthermore, it was found that smaller plastic particles (MPs) leached out larger quantities of EDCs compared to other sizes due to their higher and more efficient sorption capability of seawater. Moreover, results showed that solar irradiation has the ability to increase the concentrations of EDCs leaching out to the seawater (Chen et al., 2019a). Recently, a study investigated the leaching of six different phthalic acid esters from various commonly used commercial plastic types including LDPE, HDPE, and recycled PE. In addition, the effect of some factors on leaching was investigated such as temperature, salinity, and UV irradiation. In addition, GC–MS was used for the identification and characterization of leaching chemical substances. Generally, the leaching of various chemical substances mainly depends on the migration of the additives via the voids of the PPos. Therefore, the leaching rate of chemical substances is mainly depending on the shape and size of the additives in addition to the internal polymeric structure (Basfar, 2002; Tüzüm Demir and Ulutan, 2013). LDPE showed higher leaching rates among other used polymers (HPDE and recycled PE) (Dhavamani et al., 2022), which confirms the fact related to the diffusivity of plastic additives within the polymeric amorphous regions (Jordan et al., 2016; Satkowski, 1990). In other words, the diffusion of plastic additives is related to the ratio of amorphous regions and crystalline arrangements of the internal molecular polymeric structure of plastics. Therefore, the higher the content of amorphous regions, the more flexible and loose the polymer is. This makes the diffusivity of the plastic additives a significant factor in the leaching process. Moreover, the increase in salinity can negatively impact the leaching process (Dhavamani et al., 2022). On the other hand, the increase in UV irradiation and temperature can positively impact the leaching rate. (Dhavamani et al., 2022) found that the leaching of phthalic acid esters from recycled PE and LDPE increased as the temperature increased. It also means that additive leaching rates fluctuate based on the time of the year, where leaching rates increase in hot weather conditions rather than winter conditions. Nevertheless, the leachate dramatically increases with UV radiation exposure. Fig. 14 illustrates the relationship between the degradation of PWs, variables affecting the process, and factors influencing the leachability of additives embedded in plastics.
Understanding the relationship between a) the degradation of PWs, variables affecting the process, and b) factors influencing the leaching of the plastic additives.
5 Plastic accumulation and its environmental implications
5.1 Marine plastic litter accumulation and their overall impacts
According to several projections, starting from 2020, over 400 Mt of PW will be generated annually, and the production of plastics which are estimated to reach double by 2035 with an amount of 800 Mt and reach 1600 Mt by 2050 (Barra and Leonard, 2018). In addition, the production of plastics generates massive quantities of destructive gaseous into the air such as dioxins, carbon monoxides, hydrogen cyanides, and nitrogen oxides, which creates a critical threat to human well-being and the environment. For instance, it was stated that the trace gases generated from the LDPE’s increased in their intensity while incubating for 212 days and it reaches around 5.8 nmol of CH4 per g per day, 3.9 nmol of ethane per grams per day, 9.7 nmol propylene per grams per day, and 14.5 nmol ethylene per grams per days. In addition, findings show that the emission rates of LDPE’s are approx. 2 times more for CH4 and 76 times more for ethylene while incubating samples in the air rather than water (Royer et al., 2018). Therefore, it is important to emphasize that plastics could be a hidden source, which contributes to climate change due to these trace gases, which are estimated to rise with the increase of plastic fabrication and its environmental accumulation.
Regarding the disposal of plastics (Fig. 15), from 1950 to 2018 around 6.3 billion metric tons of the overall plastic manufacturing were discarded as trash, and over 4 billion tons have been only single utilized before its elimination (Geyer et al., 2017). Furthermore, around 4.5 billion tons of the overall produced PWs end up landfilled and/or released into the environment. Thus, it will adversely affect the soil microbial diversity since it was reported that landfills are potentially affecting soil infertility due to the extremely long duration (e.g., over 500 years) required for the complete decomposition of plastics (Chamas et al., 2020). In addition, the degradation of PW can potentially lead to the release of various contaminants (Webb et al., 2013). In the biodegradable plastics case, there are several microorganisms (e.g., Pseudomonas sp. and flavobacteria), which can effectively speed up the biodegradation of these PW when they are landfilled by breaking down their structure using various enzymes (Negoro, 2000).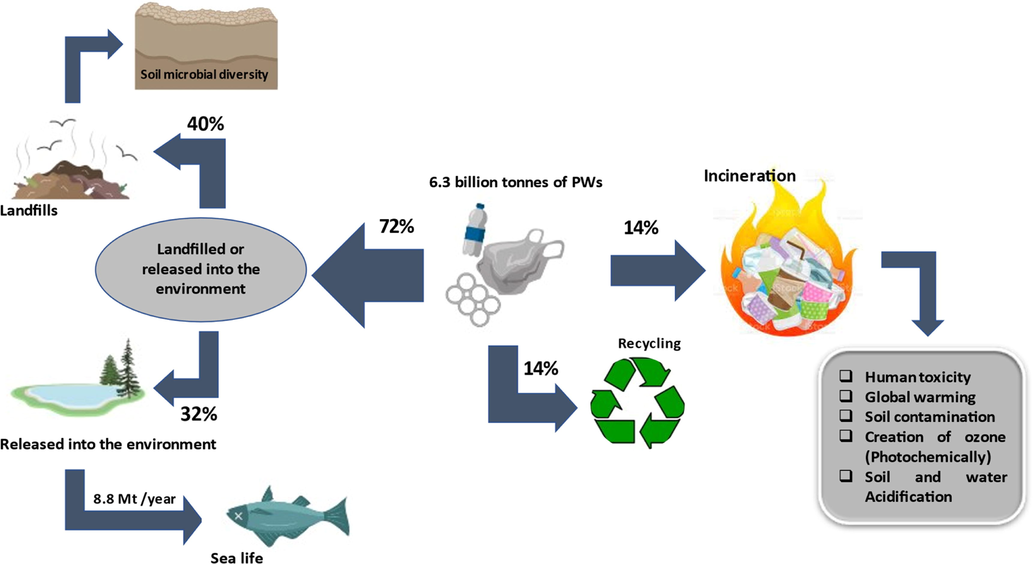
PW and their environmental impact.
Due to industrialization, plastics production started blooming in the mid-50 s, leading to a huge universal annual production which was projected to reach 367 million metric tons by 2020 (Tiseo, 2021). Plastics are suitable for several applications including being an excellent packaging material because of their various properties such as lightweight and potentially transparent material, their low cost, and their outstanding property as oxygen and/or moisture barrier. Other common materials including glass, paper, and metal are being substituted by cost-efficient packaging made of plastic. Almost a third of the generated plastics are thus transformed into packaging materials including single-use items which are generally found in the beach litter (Andrady, 2003). Many types of plastics are utilized in packaging such as PET, PS, PE, PVC, and PP. Their high utilization is well reflected in their manufacturing percentages as shown in Table 2, and therefore, these plastic types particularly will potentially end up in the aquatic ecosystems. Moreover, overfishing, and recreational uses of the sea, as well as the demographic variations by immigrating to the coastal zones, will potentially rise the flow of PWs into the marine environment (Ribic et al., 2010). PW, which are mainly land sources, contributes around 80% of the overall plastic debris. All universal fishing fleets are using plastic gear nowadays (Watson et al., 2006), and most of these gears are consistently lost or even carelessly disposed of in oceans during their usage. The most used plastics in fishing gear applications are polyolefins mainly PE and PP, in addition to nylons (Klust, 1982; Timmers et al., 2005). Therefore, around 18% of the PW which is found in aquatic ecosystems is mainly generated from fishing activities, as well as aquaculture which can considerably contribute to the PW in marine ecosystems (Hinojosa and Thiel, 2009). The rest of the plastic litter is generated from land-based sources including beach waste. The quantification of the floating plastic litter does not reflect the real amounts of PWs in the ocean since the plastic debris, which is located in the sediment, and the middle of the seawater is not included by using this method. The debris’ visibility while floating needs the plastic to be buoyant in the seawater. Nevertheless, only some of the plastics, which are utilized in the marine, have a specific gravity, which is less than the seawater (the average specific gravity of the seawater is approx. 1.025). In addition, the specific gravidities of these various plastics products can be altered due to the fillers and other additives. Therefore, denser plastics including nylons have the tendency to be submerged in the water columns and sometimes even reach the coastal sediments (Andrady, 2011).
Plastic-type
Production (%)*
Products and conventional origin
LDPE, LLDPE
21%
Bottles, plastic bags, nets, straws
HDPE
17%
Jugs (for milk, juice, and water)
PP
24%
Nets, ropes, bottle caps
PS
6%
Containers for food, plastic equipment, and tools
PET
7%
Bottles for beverages
PVC
19%
Cups, bottles, films
It should be noted that the transport of the PW is considerably affected by the shape, density, size of the fragments, and the type of polymers used (Castillo et al., 2016; Isobe et al., 2014; Wright et al., 2013). PW which are discarded are ubiquitous in the oceans and they are found as sunken debris on seafloor sediments (Galgani et al., 2000; Schulz et al., 2015), floating on the surface of seawater (Barnes et al., 2009; Barnes and Milner, 2005), and abandoned on the coastal shores (Barnes and Milner, 2005; Thiel et al., 2013). It has been reported that almost 60% of the plastic material generated is lesser in density than seawater (Andrady, 2011). In addition, buoyant plastics can be easily moved by winds and currents once introduced into the marine ecosystems (Kako et al., 2010), and consequently captured again by coastlines (Kako et al., 2014; Lavers and Bond, 2017). In turn, these plastics will be degraded into tinier pieces under several environmental conditions including sunlight, waves, and changes in temperatures (Andrady, 2011). However, some of these buoyant PW will be carried offshore and go into the oceanic gyres (Eriksen et al., 2019). A significant amount of PW (buoyant) was discovered in the North Pacific Subtropical Gyre mainly in the eastern part, renowned as the Great Pacific Garbage Patch (Chu et al., 2015; Wong et al., 1974; Young et al., 2009), where a comparatively high concentration of PW in the oceans occurs in the region (Eriksen et al., 2014; Law et al., 2014).
Owing to the intrinsic nature of the PW (e.g., durability, slow rates of degradation, and water insolubility), plastic litter has become a challenging, persistent, and significantly abundant material in marine ecosystems (Barnes et al., 2009; Browne et al., 2007; Castillo et al., 2016). It was illustrated that PW abundance (e.g., MPs) in the marine ecosystems has correlated positively with the density of the human population in the contiguous coastal zones. Usually, the increase in human population in the coastal regions results in a higher PW generation, and subsequently their frequent occurrence in the coastal seawater (Castillo et al., 2016; Depledge et al., 2013).
Although the Middle East region is responsible for around 7.3% of the universal plastic generation, only a few studies investigated microplastics (MP) pollution in the region (Castillo et al., 2016). Qatar is a country, which is located in the middle of the western coast of the semi-enclosed Arabian Gulf. The aquatic ecosystem is highly vulnerable to PW because of the continued economic expansion of this nation, where the economic zone increased to around 32,000 km2, which accounts for approximately 15% of the gulf and comprises a 563 km lengthy shoreline (Castillo et al., 2016; Jones et al., 2002). Nevertheless, the marine environmental ecosystem of the country is a precious resource, which provides tourism, fisheries, and recreation activities, as well as, the expanded petrochemical industries, which are located in the coastal area (e.g., oil and gas rigs and other facilities), are considered as anthropogenic activities. This leads to several environmental impacts which will eventually increase the plastic litter levels in the seawaters (Jones et al., 2002). Furthermore, Qatar’s marine environment conditions are extremely affecting the size and quantity distribution of the PW in the ocean where the sea temperatures during the summer may reach up to 35 °C, as well as, the elevated evaporation rates. This leads to higher salinity levels, which may reach 39 ppt to 41 ppt (Castillo et al., 2016; Jones et al., 2002).
5.2 Environmental impacts
In 2020, it was expected that around 400 Mt of PW will be generated in 195 countries, and around 8.8 Mt will end up in the aquatic ecosystems (Koller and Braunegg, 2018; Serrano-Ruiz et al., 2021). The southeast Asia countries mainly the Philippines, Sri Lanka, Indonesia, Malaysia, Thailand, Bangladesh, China, and Vietnam, are classified with the highest rank in the mismanagement of PWs, as such 88% of these wastes end up in the ocean (Ali et al., 2021). PW has the potential to carry several organic pollutants, pathogens, chemicals, and toxic metals (Chamas et al., 2020; Chen et al., 2019a; Cregut et al., 2013; Galloway and Lewis, 2016; Tang et al., 2021). In addition, PW degradation via abiotic factors releases extremely toxic compounds, which leads to the deterioration of the water and soil quality (Chen et al., 2019a). Since oceans keep loading with extreme amounts of plastic debris of various sizes, for instance, it was reported that around 5.25 trillion macro-, micro-, and nanoplastics were found in the ocean with an overall weight of 269 t (Eriksen et al., 2014). Numerous types of PW are hydrophobic by nature, which boosts the aggregation procedure with other pollutants including organic pollutants, polychlorinated biphenyls, and polyaromatic hydrocarbons (PAHs) (Liu et al., 2016). Since the reactivity is highly contingent on the hydrophobic nature of the PW type and the proportion between the surface areas to volume, this directly leads to the efficient sorption property of the PW. Furthermore, the long-term buildup of MPs can influence the food chain due to the ingestion of these particles by animals (Frias et al., 2010). A study identified the histopathological destruction in the fish tissues due to the plastic litter and illustrated the abundance of MPs in fish’s gut with a range of more than 1 mm to 3 mm and the amount of these particles was projected by 2.3 items/g to 15.8 items/g of digestive tissue (Li et al., 2020). Unquestionably, water pollution due to PW is an extreme challenge for biota, including various water bodies in which the waste is discarded such as rivers, ponds, lakes, etc. Several studies have indicated that PW is found universally in many seas and different locations including the Atlantic Ocean, Baltic Sea, Pacific Ocean, Mediterranean Sea, The United states, and Southwest England, depending on different mechanisms (e.g., currents, winds, Man-made factors, as well as the geography of the coastline) (Li et al., 2016). The PW presence acts as a disturber of the natural flow, restricts the reproducibility of the fish, and thus destroys the vital organisms. MPs were found to be present in raw, as well as treated drinking water. A study showed that MPs were found in all tested water samples, with an average occurrence range of 1473 ± 34 particles/L to 3605 ± 497 particles/L in raw water samples and from 338 ± 76 particles/L to 628 ± 28 particles/L in treated water samples (Pivokonsky et al., 2018). These findings explain the urgent need for novel PW adoption techniques, as well as new strict and efficient policies to reduce the amounts of MPs and NPs. Furthermore, another study showed that MPs were present in the drinking water of various freshwater sources in Belgium (Semmouri et al., 2022). Moreover, MPs were found to be polluting commercial salt (Karami et al., 2017; Peixoto et al., 2019), which is a crucial universal ingredient. Studies illustrated that microplastics were found in 128 different brands of commercial salts originated from 38 various countries (Peixoto et al., 2019). The presence ofsuch tiny plastics (MPs and NPs) in natural resources and food for human consumption including seafood sources, drinking water, and salt will eventually have harmful effects on human. In addition, PWs contribute to global warming due to their shade creation which hinders the growth of plankton (Proshad et al., 2017). Consequently, they disturbe the balance of the ecosystems and the natural environment, which in turn affect human health. There are several types of PWs, which can potentially contaminate the soil via surface settlement or by the penetration of the soil layers through various methods including irrigation by utilizing wastewater, biosolids, landfills, or other sources (Darwesh et al., 2021; Horton et al., 2017). The PW fragmentation into MPs on the surface of the soil occurs mainly due to photo-oxidation and temperature (Horton et al., 2017). These fragments/particles can end up in the deep soil layer, contaminate the groundwater and thus deteriorate the soil properties (Scheurer and Bigalke, 2018; Zhu et al., 2018).
Numerous scientists have studied the effects of macroplastics and MP wastes on soil bodies and these investigations demonstrate that the accumulation of these particles can cause histologic damage (Bråte et al., 2016; Bravo Rebolledo et al., 2013; Diepens and Koelmans, 2018). Moreover, a study illustrated that those added microfibers to the soil can extremely impact the soil community and biodiversity which will eventually influence the balance of the ecosystems (Lozano and Rillig, 2020).
Lastly, the incineration of PWs emits various pollutants into the atmosphere, and thus it is deemed as a major source of air pollution. The major impacts, which are initiated due to incineration, are illustrated in Fig. 15. These pollutants are metals, nitrogen oxides (NOx), aldehyde (–CHO), volatile organic compounds (VOCs), carbon dioxides (CO2), carbon monoxide (CO), PAHs, methane (CH4), furan (C4H4O), particulate matter (PM), and several other compounds, which rise the heavy metals level in the environment (Sabiha et al., 2008; Sharma et al., 2013). Some of them are the main contributors to the photochemical ozone creation process.
5.3 Impacts on human well-being
PW can have considerable direct and indirect impacts on human health via inhalation or digestion or dermal exposure as illustrated in Fig. 16. Particularly, the persistency of the MPs can potentially lead to many biological responses (e.g., genotoxicity, inflammation, necrosis, oxidative stress, and apoptosis) (Prata et al., 2020; Proshad et al., 2017). Yet, various critical outcomes can be developed when continuously exposed such as fibrosis, cancer, and tissue damage (Wright and Kelly, 2017). The composition of the polymers can have a sequence of chemical impacts due to the leaching of the chemicals which are not bounded and/or the residues of monomers and/or the combined hydrophobic organic contaminants, all leading to the deterioration of human well-being (Wright and Kelly, 2017). The ingestion of MPs and NPs by animals and humans can possibly permit the cellular entry of endogenous pollutants (Khan et al., 2015; Prata et al., 2020; Wright and Kelly, 2017). In addition, PMs may potentially result in oxidative stress because of the inhalation and consequently end up in intestinal fibrosis and acute inflammation (Nel et al., 2006). Concerning the harm that PMs can cause, a study investigated the PMs which are generated via vinyl or the incineration of plastics and reported that high concentrations of toxic pollutants were released, including the fine particle with a combination of additional harmful compounds which were detected such as benzene, acetone (Barabad et al., 2018). Furthermore, all types of plastics comprise oxygen species, which are reactive where their concentration can drastically rise because of the light interaction and/or the transition metals presence (Wright and Kelly, 2017), which leads to the formation of free radicals through dissociating C-H bonds (Gewert et al., 2015; White and Turnbull, 1994). Moreover, a study illustrated that specific plastic types including PVC, PU, and PS may produce harmful monomers which can potentially be mutagenic and/or carcinogenic monomers (Lithner et al., 2011).
Marine plastic waste degradation-fragmentation and its human health impacts.
How MPs and macro-PW can be transferred into the food chain? Regarding the transfer of macro and micro- PWs in the food chain, a study investigated the transfer of low-density polyethylene microplastics (LDPE-MPs) through earthworms and chicken, and it demonstrated that these MPs show high concentrations of 129.8 MPs/g in chicken feces and 10.2 MPs/gizzard (Huerta Lwanga et al., 2017). Unlike seabirds, the transport of plastic additives directly to humans has not been proven yet (Tanaka et al., 2013).
In general, the exhaustion of PW per year could be projected as 840 plastic fragments per individual (Huerta Lwanga et al., 2017). Therefore, and by considering the PWs mismanagement the universal plastic environmental effects, and human health impacts, there is a crucial necessity for novel technologies for PW disposal and treatments.
6 Future recommendations and potential solutions for a plastic waste challenge in the marine environment
Nowadays, plastic pollution is one of the most critical environmental challenges which are extremely affecting marine environments (Hahladakis, 2020). The lifetime duration of plastic materials is not verified yet. However, so far, it is well validated that plastics take a tremendously long time to break down, thus inhibiting their entrance into the marine environment is urgently required. To address the universal plastic marine waste challenge, there are several effective potential solutions, which can be implemented, which mainly focus on preventing plastic material from flowing into aquatic ecosystems. Most of the plastics in the marine environment have the tendency to be found under the water surface; therefore, any clean-up attempts for the floating PW are not effective. Moreover, the collection of floating plastics on the surface could be expensive due to the usage of costly inefficient cleaning technologies, as well as its potential to impact biodiversity and marine species (Thaler, 2015). On the other hand, cleaning the shorelines could be much simpler and more effective (Hahladakis, 2020). While minimizing the terrestrial PW, the plastic marine waste will be decreased as well since most of the terrestrial waste will end up eventually in the aquatic ecosystems (Jambeck et al., 2018; Jambeck et al., 2015).
Public awareness and encouraging the community to individual actions could be the ultimate solution to tackle the universal plastic marine waste problem because the ideal solutions always lie within us! Even some small changes in our mentalities regarding waste disposal can positively this issue. These individual actions may seem to be insignificant and ineffective on a short-term basis. However, it will help significantly decrease the overall amount of PW, which is improperly disposed of, and thereby decrease the plastic marine debris in the marine environment. There are several global actions such as the “Last Straw”, the “Bye-Bye Plastic Bay”, and the “Clean up Kenya”. Moreover, many countries and cities have banned plastic bottles and bags (e.g., San Francisco) (Levin, 2019). In addition, it is worth mentioning that some other countries are banning single-use plastics including Maharashtra (Indian state) and the EU which tackled the most commonly found plastics on European beaches (e.g., plastic bags, beverages containers, etc), which represents around 70% of all EU marine waste (Union, 2021). Each person can contribute significantly to these implemented actions/programs to address the plastic marine waste issue effectively by becoming an educated responsible consumer via disposing of PW properly, which will effectively help in decreasing the PW amount, which ends up in the aquatic ecosystems. In addition, the proper recycling of plastics results in less raw plastic materials generated, and thus less PW in the marine environment.
Marine plastics can be deteriorated to different degrees, owing to their degradation and fragmentation, which makes ocean cleaning a challenging task. It is projected that around 5.25 trillion plastic pieces float in different large subtropical areas, called “gyres” which are mainly five zones the Indian Ocean, the South and North Atlantic, and the South and North Pacific (Cózar et al., 2014; van Giezen and Wiegmans, 2020). Recently, several ideas were suggested on how to clean the ocean. One of the most developed and well-known ideas is “the Ocean Cleanup”, which is an environmental company devoted to addressing the marine PW issue (van Giezen and Wiegmans, 2020). The Ocean Cleanup acts as a Panacea, which will accumulate the plastics by using sensors systems and ocean vessels that will actively operate, and passively grabs the floating litter. It is a Dutch organization, which aims to implement and develop a technology, which can collect plastics that end up in the oceans through rivers (Roland Holst, 2019). This unique operation is combining both practice and law aspects. The marine cleanup mission is distinctly in line with the overall international policy goals (Roland Holst, 2019; Weller, 2018). Therefore, this technology acts as international support. According to their official website, it was predicted that this technology aims to get rid of around 90% of all floating ocean debris. However, the ocean clean-up techniques aim exclusively at the floating ocean PW, which means it does not decrease the PW generated from land to the marine environment, which leads to continuous cleaning efforts that will never end. In addition, several questions should be raised about the economic feasibility and sustainability of these technological efforts in the long term. In addition, in this case, the PW will still eventually reach the ocean, so, what are its impacts? Furthermore, these types of technologies cannot tackle all the issues since plastics will continue degrading into MPs and even NPs and thus creating an ultimate environmental threat. Presently, clean-up technologies cannot remove these small particles from the marine environment (Cordier and Uehara, 2019). From an effective mitigation perspective, strategies should emphasize on reducing the source, not only developing cleaning techniques (Rochman, 2016; Sherman and van Sebille, 2016). This might be attained via several actions such as enhancing the waste management infrastructure (Jambeck et al., 2015), as well as eliminating the microbeads which are made of plastics from the personal care products (Rochman et al., 2015). Scientists claim that marine PW will persist unless, we, individuals take an action and completely prevent the plastic input (Sherman and van Sebille, 2016). We are the ideal solution! Stopping the plastic input can be only achieved if we, including producers, governments, and companies (e.g., packaging) avoid using plastics, and even individual actions which may seem to be small movements, yet it can make a massive difference in the overall plastic inputs. Nevertheless, further studies needed for the assessment of the economic feasibility of the suggested solutions for the ocean clean up. In addition, the ecological impact of every strategy needs to be assessed in the long term to guarantee the feasibility of the short-term solutions on the mid-term as well as the long-term.
Several countries around the world are introducing specific policies and legislation on plastic pollution. A plastic treaty can contribute vastly to cleaning up our oceans, as the UN convention on the law of the sea includes a commitment in which land-based pollution sources needs to be prevented, controlled, and reduced. In addition, in 2011 the Honolulu strategy was established to address the land-based sourced activities of the marine litter. If these obligations were to be completely satisfied, then the plastic issue would be enormously decreased by now (Zhongming et al., 2018). Furthermore, the United Development Company (UDC), located in Qatar, initiated the “Seabin” project to prevent ocean pollution and ensure the protection of marine species in the region and maintain a healthy ecosystem (UDC, 2021). UDC projected that within 1 year, around 15 tons of MPs, plastic fibers, and marine debris will be filtered and eliminated (UDC, 2021).
The recycling process itself generates emissions arising from plastic additives. These PoLAs, which are not limited to but include toxic metals, PAHs, Persistent Organic Pollutants (POPs), and Brominated Flame Retardants (BFRs) could be emitted during the recycling process. While the potential release of these emissions could occur in any region, underdeveloped nations are more prone to emitting such toxins, mainly due to their uncontrolled recycling ‘stages-steps’ conditions which start with sorting, then reprocessing, and end with recycling. Additionally, not all waste generated in a particular country or region is produced there. The electronic waste generated in Europe for example arises from parts that are produced elsewhere in the world. Depending on the degree of enforcement of EU regulation regarding the import of products, PoLAs could make their way in through these imported products and as a result, end up as waste. Moreover, the difference in standards between European and non-European regulation could mean that products produced outside the EU may have a less stringent requirement in terms of toxic plastic additives and therefore, leads to the presence of these toxic substances in their products. Ultimately, these additives could directly impact the recycling of plastics while others may aid in plastic degradation (Pivnenko et al., 2016).
With governments moving towards sustainability and the circular economy concept, there is likely to be an increase in plastic recycling rates. Nevertheless, technological, and environmental obstacles still exist and need to be tackled to achieve environmentally friendly processes of designing, using, disposing, recycling, and finally recovering plastics, and as a result, replacing a significant portion of raw materials. Similarly, PoLAs, which are present in these plastic products, should be replaced by more environmentally friendly substances. Yet, a significant cut in non-renewable resources and efficient use of energy are challenges that need to be addressed to indeed be sustainable.
7 Conclusions
The degradation potential of plastic debris continues to be a critical concern for marine waste research. The current critical review presented the degradation and fragmentation of marine plastic litter under marine conditions and its environmental implications. The review also discussed the effect of the various properties of the plastic types (e.g., durability, flexibility, lightweight) on the overall degradation/fragmentation procedure, the leaching complication caused by the presence of several additives embedded in plastics, and future recommendations, which can be implemented to address PW issue in marine as well as their recycling and recovering.
Producing, consuming, recovering, and recycling plastics can be influenced by various aspects. Degradation of plastics can result in altering the MW of products as well as changing the mechanical and physicochemical properties of the PPos. In addition, any significant decrease in tensile strength may lead to plastic fragmentation into smaller pieces, which can potentially threaten the environment and human well-being. Moreover, PoLAs, which are embedded in the plastics could leach out from the products to water, and result in environmental potential threats in addition to their direct impact on the degradation of the plastics.
Different studies have reported that plastics can be degraded and fragmented under marine conditions. Various findings showed that different plastic types and sizes can potentially exhibit variations in the degree and rate of degradation and/or fragmentation. Thus, the development of a reliable model to estimate the fragmentation rates is needed to better understand and forecast the marine environmental status. In addition, different investigations confirmed that while comparing onshore and seawater conditions, sunlight/UV radiation factors can fragment the PPos, which are found onshore much faster than in seawater. Moreover, reports confirmed that the main potential destination for PW is the marine environment (e.g., seafloors, and beaches), where PW will be subjected to different factors including UV radiation which is the main initiator of their degradation. Several innovative technologies have been developed to characterize PW degradation and fragmentation. Various research findings showed that marine conditions could deteriorate the tensile strength of PW leading to the fragmentation of PPos into small fragments with the assistance of external forces (e.g., MA). Since the degradation of commercial plastics is affected by the exposed environmental conditions, it is critically important to understand and determine their environmental effects and fate. The majority of the current studies on plastic degradation and fragmentation focused on the primary stages of degradation; therefore, the creation of MPs and NPs under various environmental conditions, especially marine litter investigation, is still in its adolescence. Thus, the persistence of these small particles in the environment is still mysterious. Consequently, to forecast and better understand the key environmental parameters and characteristics, which affect the degradation of PW, further research studies are needed to facilitate the progress of PW reduction technologies.
In conclusion, quite a few challenges remain in the process of using plastics, properly sorting them, and efficiently recycling and recovering them, either due to overlooking essential problems or due to the unavailability of solutions. Therefore, to enhance supply-chain redistribution and ensure that the environment is safeguarded to be able to achieve optimal performance, joint efforts are needed to move society towards recycled plastics as supposed to the extraction of non-renewable resources. Then the process of designing plastics, sorting them, recycling the wastes, and recovering them can gain the necessary attention that it has been lacking. Efforts regarding plastic litter entering the marine environment should not be limited, and further studies need to be implemented to tackle the plastic marine debris in the first place, therefore addressing their degradation of the marine environment.
CRediT authorship contribution statement
Sarra N. Dimassi: Data curation, Investigation, Writing – original draft. John N. Hahladakis: Data curation, Funding acquisition, Project administration, Conceptualization, Investigation, Supervision, Writing – review & editing. Mohamed Najib Daly Yahia: Investigation, Data curation, Supervision. Mohammad I. Ahmad: Investigation, Data curation, Supervision. Sami Sayadi: Funding acquisition, Project administration. Mohammad A. Al-Ghouti: Data curation, Funding acquisition, Project administration, Conceptualization, Investigation, Supervision, Writing – review & editing.
Acknowledgment
This paper was supported by Qatar University Internal Grant (No. QUCG-CAS-21/22-3). The findings achieved herein are solely the responsibility of the authors.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Biodegradation of plastics: current scenario and future prospects for environmental safety. Environ. Sci. Pollut. Res.. 2018;25(8):7287-7298.
- [Google Scholar]
- Migration of lead from unplasticized polyvinyl chloride pipes. J. Hazard. Mater.. 2001;82(3):263-274.
- [Google Scholar]
- Effect of the Prodegradant-Additive Plastics Incorporated on the Polyethylene Recycling. Int. J. Polym. Sci.. 2018;2018:2474176.
- [Google Scholar]
- Degradation of conventional plastic wastes in the environment: A review on current status of knowledge and future perspectives of disposal. Sci. Total Environ.. 2021;771:144719
- [Google Scholar]
- Hydrolytic degradation of poly(ethylene terephthalate): Importance of chain scission versus crystallinity. Eur. Polym. J.. 1991;27(12):1373-1378.
- [Google Scholar]
- Plastic biodegradation: Frontline microbes and their enzymes. Sci. Total Environ.. 2021;759:143536
- [Google Scholar]
- Plastics and the Environment. John Wiley & Sons; 2003.
- Andrady, A.L., 2015. Marine Anthropogenic Litter. In: Bergmann, M., Gutow, L., Klages, M. (Eds.), Springer International Publishing, Cham., pp. 57–72.
- Effects of climate change and UV-B on materials. Photochem. Photobiol. Sci.. 2003;2(1):68-72.
- [Google Scholar]
- Accelerated degradation of low-density polyethylene in air and in sea water. Sci. Total Environ.. 2022;811:151368
- [Google Scholar]
- Review on plastic wastes in marine environment–Biodegradation and biotechnological solutions. Mar. Pollut. Bull.. 2020;150:110733
- [Google Scholar]
- Prevention through policy: Urban macroplastic leakages to the marine environment during extreme rainfall events. Mar. Pollut. Bull.. 2017;124(1):211-227.
- [Google Scholar]
- Biodegradable plastic bags on the seafloor: A future threat for seagrass meadows? Sci. Total Environ.. 2017;605–606:755-763.
- [Google Scholar]
- Emission Characteristics of Particulate Matter, Volatile Organic Compounds, and Trace Elements from the Combustion of Coals in Mongolia. Int. J. Environ. Res. Public Health. 2018;15(8):1706.
- [Google Scholar]
- Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. Royal Soc. B: Biol. Sci.. 2009;364(1526):1985-1998.
- [Google Scholar]
- Drifting plastic and its consequences for sessile organism dispersal in the Atlantic Ocean. Mar. Biol.. 2005;146(4):815-825.
- [Google Scholar]
- Barra, R. and Leonard, S., 2018. Plastics and the circular economy. A STAP document.
- Flame retardancy of radiation cross-linked poly(vinyl chloride) (PVC) used as an insulating material for wire and cable. Polym. Degrad. Stab.. 2002;77(2):221-226.
- [Google Scholar]
- Monitoring polymer degradation under different conditions in the marine environment. Environ. Pollut.. 2020;259:113836
- [Google Scholar]
- Beyler, C.L., Hirschler, M.M., 2002. Thermal decomposition of polymers. In: SFPE Handbook of fire Protection Engineering, pp. 2.
- Effect of carboxylic end groups on thermooxidative stability of PET and PBT. Polym. Degrad. Stab.. 1999;63(2):213-218.
- [Google Scholar]
- Environmental biodegradation of polyethylene. Polym. Degrad. Stab.. 2003;81(3):441-452.
- [Google Scholar]
- Booth, A., Kubowicz, S., Beegle-Krause, C., Skancke, J., Nordam, T., Landsem, E., Throne-Holst, M., Jahren, S., 2018. Microplastic in Global and Norwegian Marine Environments: Distributions, Degradation Mechanisms and Transport. M-918| 2017. Norwegian Environment Agency.
- A review of experimental studies of the role of free-radicals in polyethylene oxidation. Polym. Degrad. Stab.. 2018;155:67-83.
- [Google Scholar]
- Plastic ingestion by Atlantic cod (Gadus morhua) from the Norwegian coast. Mar. Pollut. Bull.. 2016;112(1):105-110.
- [Google Scholar]
- Plastic ingestion by harbour seals (Phoca vitulina) in The Netherlands. Mar. Pollut. Bull.. 2013;67(1):200-202.
- [Google Scholar]
- Increased migration levels of bisphenol A from polycarbonate baby bottles after dishwashing, boiling and brushing. Food Addit. Contam.. 2003;20(7):684-689.
- [Google Scholar]
- Brien, S., 2007. Vinyls industry update. Presentation at the world vinyl forum 2007, Sept. 2007. September). http://vinylinfo.org/wpcontent/uploads/2011/07/VinylIndustryUpdate.pdf (March 20, 2015).
- Ingested microscopic plastic translocates to the circulatory system of the mussel, Mytilus edulis (L.) Environ. Sci. Technol.. 2008;42(13):5026-5031.
- [Google Scholar]
- Microplastic—an emerging contaminant of potential concern? Integr. Environ. Assess. Manage.. 2007;3(4):559-561.
- [Google Scholar]
- Handbook of Thermoplastics. New York: O. Olabisi Dekker; 1997. p. :771-798.
- Observation of the degradation of three types of plastic pellets exposed to UV irradiation in three different environments. Sci. Total Environ.. 2018;628–629:740-747.
- [Google Scholar]
- Prevalence of microplastics in the marine waters of Qatar. Mar. Pollut. Bull.. 2016;111(1):260-267.
- [Google Scholar]
- Degradation Rates of Plastics in the Environment. ACS Sustainable Chem. Eng.. 2020;8(9):3494-3511.
- [Google Scholar]
- The power of light in polymer science: photochemical processes to manipulate polymer formation, structure, and properties. Polym. Chem.. 2014;5(7):2187-2201.
- [Google Scholar]
- Leaching of endocrine disrupting chemicals from marine microplastics and mesoplastics under common life stress conditions. Environ. Int.. 2019;130:104938
- [Google Scholar]
- Marine microplastics bound dioxin-like chemicals: Model explanation and risk assessment. J. Hazard. Mater.. 2019;364:82-90.
- [Google Scholar]
- Oxo-biodegradable carbon backbone polymers–Oxidative degradation of polyethylene under accelerated test conditions. Polym. Degrad. Stab.. 2006;91(11):2739-2747.
- [Google Scholar]
- Perfluoroalkyl sulfonates and carboxylic acids in liver, muscle and adipose tissues of black-footed albatross (Phoebastria nigripes) from Midway Island, North Pacific Ocean. Chemosphere. 2015;138:60-66.
- [Google Scholar]
- On mechanical fragmentation of single-use plastics in the sea swash zone with different types of bottom sediments: Insights from laboratory experiments. Mar. Pollut. Bull.. 2020;150:110726
- [Google Scholar]
- Effects of grilling and roasting on the levels of polycyclic aromatic hydrocarbons in beef and pork. Food Chem.. 2011;129(4):1420-1426.
- [Google Scholar]
- Microplastics as contaminants in the marine environment: A review. Mar. Pollut. Bull.. 2011;62(12):2588-2597.
- [Google Scholar]
- Effects of mechanical and chemical processes on the degradation of plastic beach debris on the island of Kauai. Hawaii. Mar Pollut Bull. 2010;60(5):650-654.
- [Google Scholar]
- How much innovation is needed to protect the ocean from plastic contamination? Sci. Total Environ.. 2019;670:789-799.
- [Google Scholar]
- New insights into polyurethane biodegradation and realistic prospects for the development of a sustainable waste recycling process. Biotechnol. Adv.. 2013;31(8):1634-1647.
- [Google Scholar]
- Culbert, B., Christel, A., 2004. Modern Polyesters: Chemistry and Technology of Polyesters and Copolyesters, pp. 143–194.
- Photodegradable plastics: end-of-life design principles. Green Chem. Lett. Rev.. 2010;3(2):69-82.
- [Google Scholar]
- Plastics: Environmental and Biotechnological Perspectives on Microbial Degradation. Appl. Environ. Microbiol.. 2019;85(19):e01095-01019.
- [Google Scholar]
- Nanosensors and Nanodevices for Smart Multifunctional Textiles. Elsevier; 2021. p. :267-286.
- The effects of salinity, temperature, and UV irradiation on leaching and adsorption of phthalate esters from polyethylene in seawater. Sci. Total Environ.. 2022;838:155461
- [Google Scholar]
- Accumulation of Plastic Debris and Associated Contaminants in Aquatic Food Webs. Environ. Sci. Technol.. 2018;52(15):8510-8520.
- [Google Scholar]
- Observations and idealized modelling of microplastic transport in estuaries: The exemplary case of an upwelling system (Ría de Vigo, NW Spain) Mar. Chem.. 2020;222:103780
- [Google Scholar]
- Aspects of poly(ethylene terephthalate) degradation for archival life and environmental degradation. Polym. Degrad. Stab.. 1991;32(2):131-153.
- [Google Scholar]
- Biofilm and Diatom Succession on Polyethylene (PE) and Biodegradable Plastic Bags in Two Marine Habitats: Early Signs of Degradation in the Pelagic and Benthic Zone? PLoS ONE. 2015;10(9):e0137201
- [Google Scholar]
- Nanoplastics formed during the mechanical breakdown of daily-use polystyrene products. Nanoscale Adv.. 2019;1(3):1055-1061.
- [Google Scholar]
- Engelhart, A., 2010. The fiber year 2009/10. A world survey on textile and nonwovens industry. Oerlikon Textile GmbH & Co. KG, Remscheid.
- Plastic pollution in the world's oceans: more than 5 trillion plastic pieces weighing over 250,000 tons afloat at sea. PLoS ONE. 2014;9(12):e111913
- [Google Scholar]
- Hazardous Chemicals Associated with Plastics in the Marine Environment. Cham: Springer International Publishing; 2019. p. :135-162.
- Faber, M., Marinković, M., de Valk, E., Waaijers-van der Loop, S., 2021. Paints and microplastics. Exploring the possibilities to reduce the use and release of microplastics from paints. Feedback from the paint sector.
- Microbial degradation of recalcitrant compounds and synthetic aromatic polymers. Enzyme Microb. Technol.. 1979;1(4):226-232.
- [Google Scholar]
- Fagerburg, D.R., Clauberg, H., 2004. Modern Polyesters: Chemistry and Technology of Polyesters and Copolyesters, pp. 609–641.
- 4-Nonylphenol (NP) in food-contact materials: Analytical methodology and occurrence. Food Addit. Contam. Part A. 2008;25(3):364-372.
- [Google Scholar]
- Humidity dependence of carbon dioxide generation during photodegradation of biaxially oriented polypropylene in oxygen. Polym. Degrad. Stab.. 2009;94(1):83-89.
- [Google Scholar]
- Hazardous Chemicals Associated with Plastics in the Marine Environment. Cham: Springer International Publishing; 2019. p. :71-92.
- Organic pollutants in microplastics from two beaches of the Portuguese coast. Mar. Pollut. Bull.. 2010;60(11):1988-1992.
- [Google Scholar]
- Litter on the Sea Floor Along European Coasts. Mar. Pollut. Bull.. 2000;40(6):516-527.
- [Google Scholar]
- Marine microplastics spell big problems for future generations. Proc. Natl. Acad. Sci.. 2016;113(9):2331.
- [Google Scholar]
- Intake of bisphenol A from canned beverages and foods on the Belgian market. Food Addit. Contam.: Part A. 2010;27(11):1627-1637.
- [Google Scholar]
- Fragmentation of plastic objects in a laboratory seawater microcosm. Sci. Rep.. 2020;10(1):10945.
- [Google Scholar]
- Pathways for degradation of plastic polymers floating in the marine environment. Environ. Sci. Processes Impacts. 2015;17(9):1513-1521.
- [Google Scholar]
- Marine plastic litter: the unanalyzed nano-fraction. Environ. Sci. Nano. 2016;3(2):346-350.
- [Google Scholar]
- Polymer Degradation and Stabilisation. CUP Archive; 1988.
- Chemical Degradation and Color Changes of Paint Protective Coatings Used in Solar Glass Mirrors. Coatings. 2021;11(4):476.
- [Google Scholar]
- Polystyrenes: A review of the literature on the products of thermal decomposition and toxicity. Fire Mater.. 1987;11(3):109-130.
- [Google Scholar]
- Biodegradation of polyethylene by the thermophilic bacterium Brevibacillus borstelensis. J. Appl. Microbiol.. 2005;98(5):1093-1100.
- [Google Scholar]
- Delineating the global plastic marine litter challenge: clarifying the misconceptions. Environ. Monit. Assess.. 2020;192(5):267.
- [Google Scholar]
- An overview of chemical additives present in plastics: Migration, release, fate and environmental impact during their use, disposal and recycling. J. Hazard. Mater.. 2018;344:179-199.
- [Google Scholar]
- Long Term Properties of Polyolefins. Berlin Heidelberg, Berlin, Heidelberg: Springer; 2004. p. :177-200.
- Hazardous Substances in Plastic Materials. COWI in cooperation with Danish Technological Institute; 2013. p. :7-8.
- Henneuse-Boxus, C., Pacary, T., 2003. Emissions from Plastics. iSmithers Rapra Publishing.
- Floating marine debris in fjords, gulfs and channels of southern Chile. Mar. Pollut. Bull.. 2009;58(3):341-350.
- [Google Scholar]
- Organic micropollutants in marine plastics debris from the open ocean and remote and urban beaches. Mar. Pollut. Bull.. 2011;62(8):1683-1692.
- [Google Scholar]
- Degradation products of plastics: Polyethylene and styrene-containing thermoplastics — Analytical, occupational and toxicologic aspects. Scand. J. Work Environ. Health. 1982;8:1-60.
- [Google Scholar]
- Microplastics in freshwater and terrestrial environments: Evaluating the current understanding to identify the knowledge gaps and future research priorities. Sci. Total Environ.. 2017;586:127-141.
- [Google Scholar]
- Microplastics in the Terrestrial Ecosystem: Implications for Lumbricus terrestris (Oligochaeta, Lumbricidae) Environ. Sci. Technol.. 2016;50(5):2685-2691.
- [Google Scholar]
- Field evidence for transfer of plastic debris along a terrestrial food chain. Sci. Rep.. 2017;7(1):14071.
- [Google Scholar]
- Recyclability of four types of plastics exposed to UV irradiation in a marine environment. Waste Manage.. 2018;79:339-345.
- [Google Scholar]
- The degradation potential of PET bottles in the marine environment: An ATR-FTIR based approach. Sci. Rep.. 2016;6(1):23501.
- [Google Scholar]
- Selective transport of microplastics and mesoplastics by drifting in coastal waters. Mar. Pollut. Bull.. 2014;89(1):324-330.
- [Google Scholar]
- Evaluation of degradability of biodegradable polyethylene (PE) Polym. Degrad. Stab.. 2003;80(1):39-43.
- [Google Scholar]
- Effects of accelerated and natural ageing on plasticized polyvinyl chloride (PVC) Polym. Degrad. Stab.. 1999;66(3):415-421.
- [Google Scholar]
- Challenges and emerging solutions to the land-based plastic waste issue in Africa. Marine Policy. 2018;96:256-263.
- [Google Scholar]
- The effects of humidity on photodegradation of poly(vinyl chloride) and polyethylene as measured by the CO2 evolution rate. Polym. Degrad. Stab.. 2013;98(2):508-513.
- [Google Scholar]
- The effect of crystalline morphology on the degradation of polycaprolactone in a solution of phosphate buffer and lipase. Polym. Adv. Technol.. 2008;19(12):1901-1906.
- [Google Scholar]
- Rapid measurement of polymer photo-degradation by FTIR spectrometry of evolved carbon dioxide. Polym. Degrad. Stab.. 2006;91(5):1086-1096.
- [Google Scholar]
- Biodegradability of photodegraded polymers. I. Development of experimental procedures. Environ. Sci. Technol.. 1974;8(10):919-923.
- [Google Scholar]
- Mechanical properties of low density polyethylene. J Dyn. Behav. Mater.. 2016;2(4):411-420.
- [Google Scholar]
- A decadal prediction of the quantity of plastic marine debris littered on beaches of the East Asian marginal seas. Mar. Pollut. Bull.. 2014;81(1):174-184.
- [Google Scholar]
- Inverse estimation of drifting-object outflows using actual observation data. J. Oceanogr.. 2010;66(2):291-297.
- [Google Scholar]
- Microplastics Generation: Onset of Fragmentation of Polyethylene Films in Marine Environment Mesocosms. Front. Mar. Sci.. 2017;4(84)
- [Google Scholar]
- The presence of microplastics in commercial salts from different countries. Sci. Rep.. 2017;7(1):46173.
- [Google Scholar]
- Kershaw, P., 2015a. Biodegradable Plastics and Marine Litter: Misconceptions, Concerns and Impacts on Our Marine Environments.
- Kershaw, P., 2015b. Sources, Fate and Effects of Microplastics in the Marine Environment: A Global Assessment. International Maritime Organization.
- Chemical and morphological changes of environmentally degradable polyethylene films exposed to thermo-oxidation. Polym. Degrad. Stab.. 1999;63(1):127-138.
- [Google Scholar]
- Influence of polyethylene microplastic beads on the uptake and localization of silver in zebrafish (Danio rerio) Environ. Pollut.. 2015;206:73-79.
- [Google Scholar]
- Evaluation of polypropylene plastic degradation and microplastic identification in sediments at Tambak Lorok coastal area, Semarang, Indonesia. Marine Pollut. Bullet.. 2020;151:110868
- [Google Scholar]
- Degradation of styrene-g-cassava starch filled polystyrene plastics. Polym. Degrad. Stab.. 1999;66(3):323-335.
- [Google Scholar]
- Leaching characteristics of polybrominated diphenyl ethers (PBDEs) from flame-retardant plastics. Chemosphere. 2006;65(3):506-513.
- [Google Scholar]
- Phyllosphere yeasts rapidly break down biodegradable plastics. AMB Express. 2011;1:44.
- [Google Scholar]
- Klust, G., 1982. Netting materials for fishing gear, pp. 2.
- Koelmans, A.A., Besseling, E., Shim, W.J., 2015. Marine Anthropogenic Litter. In: Bergmann, M., Gutow, L., Klages, M., (Eds.), Springer International Publishing, Cham., pp. 325–340.
- Plastic as a Carrier of POPs to Aquatic Organisms: A Model Analysis. Environ. Sci. Technol.. 2013;47(14):7812-7820.
- [Google Scholar]
- Advanced approaches to produce polyhydroxyalkanoate (PHA) biopolyesters in a sustainable and economic fashion. EuroBiotech J.. 2018;2(2):89-103.
- [Google Scholar]
- Ten Years of Mixing Cocktails: A Review of Combination Effects of Endocrine-Disrupting Chemicals. Environ. Health Perspect.. 2007;115(Suppl 1):98-105.
- [Google Scholar]
- Prospects for microbiological solutions to environmental pollution with plastics. Appl. Microbiol. Biotechnol.. 2015;99(21):8857-8874.
- [Google Scholar]
- Biodegradation of Agricultural Plastic Films: A Critical Review. J. Polym. Environ.. 2007;15(2):125-150.
- [Google Scholar]
- La Mantia, F., 2002. Handbook of Plastics Recycling, iSmithers Rapra Publishing.
- Characterisation of nanoplastics during the degradation of polystyrene. Chemosphere. 2016;145:265-268.
- [Google Scholar]
- Formation of microscopic particles during the degradation of different polymers. Chemosphere. 2016;161:510-517.
- [Google Scholar]
- Exceptional and rapid accumulation of anthropogenic debris on one of the world’s most remote and pristine islands. Proc. Natl. Acad. Sci.. 2017;114(23):6052.
- [Google Scholar]
- Distribution of Surface Plastic Debris in the Eastern Pacific Ocean from an 11-Year Data Set. Environ. Sci. Technol.. 2014;48(9):4732-4738.
- [Google Scholar]
- Levin, S., 2019. How San Francisco is leading the way out of bottled water culture.
- Microplastics in fishes and their living environments surrounding a plastic production area. Sci. Total Environ.. 2020;727:138662
- [Google Scholar]
- Crystalline Characteristics, Mechanical Properties, Thermal Degradation Kinetics and Hydration Behavior of Biodegradable Fibers Melt-Spun from Polyoxymethylene/Poly(l-lactic acid) Blends. Polymers (Basel). 2019;11(11)
- [Google Scholar]
- Plastic waste in the marine environment: A review of sources, occurrence and effects. Sci. Total Environ.. 2016;566–567:333-349.
- [Google Scholar]
- Environmental and health hazard ranking and assessment of plastic polymers based on chemical composition. Sci. Total Environ.. 2011;409(18):3309-3324.
- [Google Scholar]
- Sorption of polycyclic aromatic hydrocarbons to polystyrene nanoplastic. Environ. Toxicol. Chem.. 2016;35(7):1650-1655.
- [Google Scholar]
- Industrial Plastics: Theory and Applications. Albany, NY: Thomson Delmar Learning; 2004.
- Migration of Nonylphenol from Plastic Containers to Water and a Milk Surrogate. J. Agric. Food. Chem.. 2004;52(7):2016-2020.
- [Google Scholar]
- Effects of Microplastic Fibers and Drought on Plant Communities. Environ. Sci. Technol.. 2020;54(10):6166-6173.
- [Google Scholar]
- Leaching behavior of fluorescent additives from microplastics and the toxicity of leachate to Chlorella vulgaris. Sci. Total Environ.. 2019;678:1-9.
- [Google Scholar]
- Maes, T., McGlade, J., Fahim, I.S., Green, D.S., Landrigan, P., Andrady, A.L., Costa, M.F., Geyer, R., Gomes, R., Hwai, A.T.S., 2021. From Pollution to Solution: A Global Assessment of Marine Litter and Plastic Pollution.
- Mark, H.F., Kroschwitz, J.I., 1985. Encyclopedia of polymer science and engineering.
- Enzymatic and microbial biodegradability of poly(ethylene terephthalate) copolymers containing nitrated units. Polym. Degrad. Stab.. 2006;91(4):663-671.
- [Google Scholar]
- Critical evaluation of biodegradation studies on synthetic plastics through a systematic literature review. Sci. Total Environ.. 2021;752:141959
- [Google Scholar]
- Plastic Resin Pellets as a Transport Medium for Toxic Chemicals in the Marine Environment. Environ. Sci. Technol.. 2001;35(2):318-324.
- [Google Scholar]
- Nano-plastics in the aquatic environment. Environ. Sci. Processes Impacts. 2015;17(10):1712-1721.
- [Google Scholar]
- Degradation of low-density polyethylene to nanoplastic particles by accelerated weathering. Sci. Total Environ.. 2022;826:154035
- [Google Scholar]
- Improving the compatibility and assessing the biodegradation of polymer composites of low-density polyethylene and polyamide-6. Int. Polym. Sci. Technol.. 2015;42(1):51-56.
- [Google Scholar]
- Biofilm formation and partial biodegradation of polystyrene by the actinomycete Rhodococcus ruber. Biodegradation. 2008;19(6):851-858.
- [Google Scholar]
- Biological degradation of synthetic polyesters—Enzymes as potential catalysts for polyester recycling. Process Biochem.. 2006;41(10):2124-2128.
- [Google Scholar]
- Biodegradation of polyesters containing aromatic constituents. J. Biotechnol.. 2001;86(2):87-95.
- [Google Scholar]
- Migration of formaldehyde and acetaldehyde into mineral water in polyethylene terephthalate (PET) bottles. Food Addit. Contam.. 2006;23(2):212-218.
- [Google Scholar]
- Environmental Deterioration of Biodegradable, Oxo-biodegradable, Compostable, and Conventional Plastic Carrier Bags in the Sea, Soil, and Open-Air Over a 3-Year Period. Environ. Sci. Technol.. 2019;53(9):4775-4783.
- [Google Scholar]
- Microbial colonization and degradation of polyethylene and biodegradable plastic bags in temperate fine-grained organic-rich marine sediments. Mar. Pollut. Bull.. 2016;103(1):168-178.
- [Google Scholar]
- Biodegradation of nylon oligomers. Appl. Microbiol. Biotechnol.. 2000;54(4):461-466.
- [Google Scholar]
- The Chemistry of Polymers. Royal Society of Chemistry; 2017.
- Degradation of plastic carrier bags in the marine environment. Mar. Pollut. Bull.. 2010;60(12):2279-2283.
- [Google Scholar]
- Estrogenicity of resin-based composites and sealants used in dentistry. Environ. Health Perspect.. 1996;104(3):298-305.
- [Google Scholar]
- Degradation and Stabilisation of PVC. Netherlands, Dordrecht: Springer; 1984. p. :197-252.
- Padsalgikar, A., 2017. Plastics in Medical Devices for Cardiovascular Applications. William Andrew.
- Phthalate Release from Plastic Fragments and Degradation in Seawater. Environ. Sci. Technol.. 2019;53(1):166-175.
- [Google Scholar]
- Peacock, A., 2000. Handbook of Polyethylene: Structures: Properties, and Applications, CRC Press.
- Microplastic pollution in commercial salt for human consumption: A review. Estuar. Coast. Shelf Sci.. 2019;219:161-168.
- [Google Scholar]
- Pielichowski, K., Njuguna, J., 2005. Thermal Degradation of Polymeric Materials. iSmithers Rapra Publishing.
- Long-term accelerated weathering of HAS stabilized PE and PP plaques: Compliance of ESRI, IR, and microhardness data characterizing heterogeneity of photooxidation. Polym. Degrad. Stab.. 2015;120:114-121.
- [Google Scholar]
- Biodegradation of plastics in soil: The effect of temperature. Polym. Degrad. Stab.. 2019;170:109017
- [Google Scholar]
- Non-microbial degradation of polyesters: mechanisms and modifications. Spec. Publ.-Roy. Soc. Chem.. 1992;109 7-7
- [Google Scholar]
- Recycling of plastic waste: Presence of phthalates in plastics from households and industry. Waste Manage.. 2016;54:44-52.
- [Google Scholar]
- Occurrence of microplastics in raw and treated drinking water. Sci. Total Environ.. 2018;643:1644-1651.
- [Google Scholar]
- PlasticsEurope, E. and EPRO, E., 2008. The Compelling Facts about Plastics. An Analysis of Plastics Production, Demand and Recovery for 2006 in Europe, Plastics Europe Brussels, Belgium.
- PlasticsEurope, M., Group, M.R., 2008. Business Data and Carts 2007. Status September.
- Environmental exposure to microplastics: An overview on possible human health effects. Sci. Total Environ.. 2020;702:134455
- [Google Scholar]
- Solution structure of 2′,5′ d(G4C4): Relevance to topological restrictions and nature's choice of phosphodiester links. Eur. J. Biochem.. 2004;271(14):2956-2966.
- [Google Scholar]
- Handbook of Fire Resistant Textiles. Woodhead Publishing; 2013. p. :3-25.
- Pritchard, G., 1997. Plastics Additives: An AZ Reference. Springer Science & Business Media.
- Pritchard, G., 1998. Plastics Additives: An A-Z Reference. Dordrecht: Springer Netherlands.
- Proshad, R., Kormoker, T., Islam, M.S., Haque, M.A., Rahman, M.M., Mithu, M.M.R., 2017. Toxic effects of plastic on human health and environment: A consequences of health risk assessment in Bangladesh.
- Spatial extent and historical context of North Sea oxygen depletion in August 2010. Biogeochemistry. 2013;113(1):53-68.
- [Google Scholar]
- Oxidative degradations of oxodegradable LDPE enhanced with thermoplastic pea starch: Thermo-mechanical properties, morphology, and UV-ageing studies. J. Appl. Polym. Sci.. 2011;122(1):489-496.
- [Google Scholar]
- Carbon-Carbon Bond Forming Reactions via Photogenerated Intermediates. Chem. Rev.. 2016;116(17):9850-9913.
- [Google Scholar]
- Principles of Polymer Chemistry. Springer; 2000. p. :449-475.
- Trends and drivers of marine debris on the Atlantic coast of the United States 1997–2007. Mar. Pollut. Bull.. 2010;60(8):1231-1242.
- [Google Scholar]
- Final report. Migration of phthalate plasticizers from soft PVC toys and child care articles. TNO report V3932. EU contract no, EDT/00/503303. Zeist: TNO Nutrition and Food Research; 2001.
- Persistent organic pollutants carried by synthetic polymers in the ocean environment. Mar. Pollut. Bull.. 2007;54(8):1230-1237.
- [Google Scholar]
- Starch esters as biodegradable plastics: Effects of ester group chain length and degree of substitution on anaerobic biodegradation. Enzyme Microb. Technol.. 1995;17(9):848-852.
- [Google Scholar]
- Strategies for reducing ocean plastic debris should be diverse and guided by science. Environ. Res. Lett.. 2016;11(4):041001
- [Google Scholar]
- Scientific Evidence Supports a Ban on Microbeads. Environ. Sci. Technol.. 2015;49(18):10759-10761.
- [Google Scholar]
- The Netherlands: The 2018 Agreement between The Ocean Cleanup and the Netherlands. Int. J. Marine Coastal Law. 2019;34(2):351-371.
- [Google Scholar]
- Production of methane and ethylene from plastic in the environment. PloS One. 2018;13(8) e0200574-e0200574
- [Google Scholar]
- Heavy metal pollution from medical waste incineration at Islamabad and Rawalpindi, Pakistan. Microchem. J.. 2008;90(1):77-81.
- [Google Scholar]
- Bisphenol A (BPA) and its source in foods in Japanese markets. Food Addit. Contam.. 2007;24(1):103-112.
- [Google Scholar]
- Comparative biodegradation study of polymer from plastic bottle waste using novel isolated bacteria and fungi from marine source. J. Polym. Res.. 2019;27(1):16.
- [Google Scholar]
- Satkowski, M.M., 1990. The crystallization and morphology of polyethylene and its blends.
- Biotechnology in the Sustainable Environment. Springer Science & Business Media; 2012.
- Perfluorinated Compounds, Polychlorinated Biphenyls, and Organochlorine Pesticide Contamination in Composite Food Samples from Dallas, Texas, USA. Environ. Health Perspect.. 2010;118(6):796-802.
- [Google Scholar]
- Microplastics in Swiss Floodplain Soils. Environ. Sci. Technol.. 2018;52(6):3591-3598.
- [Google Scholar]
- Comparative analysis of time series of marine litter surveyed on beaches and the seafloor in the southeastern North Sea. Marine Environ. Res.. 2015;106:61-67.
- [Google Scholar]
- Growth Within: A Circular Economy Vision for a Competitive Europe. Ellen MacArthur Foundation and the McKinsey Center for Business and Environment; 2016. p. :1-22.
- Heterogeneous weathering of polypropylene in the marine environment. Sci. Total Environ.. 2022;812:152308
- [Google Scholar]
- Presence of microplastics in drinking water from different freshwater sources in Flanders (Belgium), an urbanized region in Europe. Int. J. Food Contam.. 2022;9(1):6.
- [Google Scholar]
- Biodegradable plastic mulches: Impact on the agricultural biotic environment. Sci. Total Environ.. 2021;750:141228
- [Google Scholar]
- Biological degradation of plastics: a comprehensive review. Biotechnol. Adv.. 2008;26(3):246-265.
- [Google Scholar]
- The impact of incinerators on human health and environment. Rev. Environ. Health. 2013;28(1):67-72.
- [Google Scholar]
- Modeling marine surface microplastic transport to assess optimal removal locations. Environ. Res. Lett.. 2016;11(1):014006
- [Google Scholar]
- Mechanistic implications of plastic degradation. Polym. Degrad. Stab.. 2008;93(3):561-584.
- [Google Scholar]
- Environmental Microbiology and Biotechnology. Springer; 2004.
- Volatile organic components migrating from plastic pipes (HDPE, PEX and PVC) into drinking water. Water Res.. 2003;37(8):1912-1920.
- [Google Scholar]
- Combined Effects of UV Exposure Duration and Mechanical Abrasion on Microplastic Fragmentation by Polymer Type. Environ. Sci. Technol.. 2017;51(8):4368-4376.
- [Google Scholar]
- Ageing and fragmentation of marine microplastics. Sci. Total Environ.. 2022;827:154438
- [Google Scholar]
- UV degradation of natural and synthetic microfibers causes fragmentation and release of polymer degradation products and chemical additives. Sci. Total Environ.. 2021;755:143170
- [Google Scholar]
- Microplastic ingestion in fish larvae in the western English Channel. Environ. Pollut.. 2017;226:250-259.
- [Google Scholar]
- Chlorine and the environment: An overview of the chlorine industry. Environ. Sci. Pollut. Res. Int.. 2001;8(2):146.
- [Google Scholar]
- Strlic, M., Kolar, J., 2005. Ageing and Stabilisation of Paper. National and University Library.
- Weathering of Plastics. Norwich, NY: William Andrew Publishing; 1999. p. :61-68.
- Accumulation of plastic-derived chemicals in tissues of seabirds ingesting marine plastics. Mar. Pollut. Bull.. 2013;69(1–2):219-222.
- [Google Scholar]
- Textural, surface and chemical properties of polyvinyl chloride particles degraded in a simulated environment. Mar. Pollut. Bull.. 2018;133:392-401.
- [Google Scholar]
- Morphology and chemical properties of polypropylene pellets degraded in simulated terrestrial and marine environments. Mar. Pollut. Bull.. 2019;149:110626
- [Google Scholar]
- The effects of chemical structure and synthesis method on photodegradation of polypropylene. J. Appl. Polym. Sci.. 2005;95(2):270-279.
- [Google Scholar]
- A review: Research progress on microplastic pollutants in aquatic environments. Sci. Total Environ.. 2021;766:142572
- [Google Scholar]
- Understanding the Fragmentation Pattern of Marine Plastic Debris. Environ. Sci. Technol.. 2016;50(11):5668-5675.
- [Google Scholar]
- Transport and release of chemicals from plastics to the environment and to wildlife. Philos. Trans. Roy. Soc. B: Biol. Sci.. 2009;364(1526):2027-2045.
- [Google Scholar]
- Thaler, A., 2015. Three facts (and a lot of questions) about the ocean cleanup. Southern Fried Science. http://www.southernfriedscience.com/three-facts-about-the-ocean-cleanup.
- Anthropogenic marine debris in the coastal environment: A multi-year comparison between coastal waters and local shores. Mar. Pollut. Bull.. 2013;71(1):307-316.
- [Google Scholar]
- Timmers, M.A., Kistner, C.A., Donohue, M.J., 2005. Marine debris of the Northwestern Hawaiian Islands: ghost net identification, US Department of Commerce, National Oceanic and Atmospheric Administration ….
- Tiseo, I., 2021. Annual production of plastics worldwide from 1950 to 2020. Statista.
- Phthalates in Products that Children are in Direct Contact with. Environmental Protection Agency; 2010.
- Total Consumer Exposure to Polybrominated Diphenyl Ethers in North America and Europe. Environ. Sci. Technol.. 2011;45(6):2391-2397.
- [Google Scholar]
- Paint particles in the marine environment: An overlooked component of microplastics. Water Res. X. 2021;12:100110
- [Google Scholar]
- Migration of phthalate and non-phthalate plasticizers out of plasticized PVC films into air. J. Appl. Polym. Sci.. 2013;128(3):1948-1961.
- [Google Scholar]
- UDC, 2021. For the First Time in the State of Qatar, on World Environment Day… UDC Inaugurates the ‘SEABIN’ Project to Maintain Clean Waters at the Pearl-Qatar. Available at: https://www.udcqatar.com/en/Media-Room/Press-Releases/UDC-inaugurates-the-Seabin-project-to-maintain-clean-waters--at-The-Pearl-Qatar.
- UNEP, M.P.D., 2016. Microplastics. Global lessons and research to inspire action and guide policy change.
- Union, E., 2021. European Union: Ban on Single-Use Plastics Takes Effect. Available at: https://www.loc.gov/item/global-legal-monitor/2021-07-18/european-union-ban-on-single-use-plastics-takes-effect/.
- Spoilt - Ocean Cleanup: Alternative logistics chains to accommodate plastic waste recycling: An economic evaluation. Transport. Res. Interdisc. Perspect.. 2020;5:100115
- [Google Scholar]
- Hormones and Endocrine-Disrupting Chemicals: Low-Dose Effects and Nonmonotonic Dose Responses. Endocr. Rev.. 2012;33(3):378-455.
- [Google Scholar]
- Handbook of Polyolefins. New York: Marcel Dekker; 2000.
- Vasile, C., Pascu, M., 2005. Practical Guide to Polyethylene. iSmithers Rapra Publishing.
- Recycling and resource efficiency: it is time for a change from quantity to quality. Sage UK: London, England: Sage Publications; 2013. p. :539-540.
- Strong Sorption of PCBs to Nanoplastics, Microplastics, Carbon Nanotubes, and Fullerenes. Environ. Sci. Technol.. 2014;48(9):4869-4876.
- [Google Scholar]
- Venkatachalam, S., Nayak, S.G., Labde, J.V., Gharal, P.R., Rao, K., Kelkar, A.K., 2012. Degradation and Recyclability of Poly (Ethylene Terephthalate). Edited by Hosam El-Din M. Saleh, pp. 75.
- Fate and metabolism of the brominated flame retardant tetrabromobisphenol A (TBBPA) in rice cell suspension culture. Environ. Pollut.. 2016;214:299-306.
- [Google Scholar]
- Exploring hydroperoxides in combustion: History, recent advances and perspectives. Prog. Energy Combust. Sci.. 2019;73:132-181.
- [Google Scholar]
- Fishing gear associated with global marine catches: I. Database development. Fisheries Res.. 2006;79(1):97-102.
- [Google Scholar]
- The fate of plastic in the ocean environment–a minireview. Environ. Sci. Processes Impacts. 2021;23(2):198-212.
- [Google Scholar]
- Plastic Degradation and Its Environmental Implications with Special Reference to Poly(ethylene terephthalate) Polymers. 2013;5(1):1-18.
- [Google Scholar]
- From macroplastic to microplastic: Degradation of high-density polyethylene, polypropylene, and polystyrene in a salt marsh habitat. Environ. Toxicol. Chem.. 2016;35(7):1632-1640.
- [Google Scholar]
- Degradation of common polymer ropes in a sublittoral marine environment. Mar. Pollut. Bull.. 2017;118(1):248-253.
- [Google Scholar]
- Weller, K., 2018. Nations commit to fight plastic pollution together during the UN General Assembly. Available at: https://www.unep.org/news-and-stories/press-release/nations-commit-fight-plastic-pollution-together-during-un-general.
- Weathering of polymers: mechanisms of degradation and stabilization, testing strategies and modelling. J. Mater. Sci.. 1994;29(3):584-613.
- [Google Scholar]
- Polyolefins with controlled environmental degradability. Polym. Degrad. Stab.. 2006;91(7):1581-1592.
- [Google Scholar]
- Quantitative Tar and Plastic Waste Distributions in the Pacific Ocean. Nature. 1974;247(5435):30-32.
- [Google Scholar]
- Plastic and Human Health: A Micro Issue? Environ. Sci. Technol.. 2017;51(12):6634-6647.
- [Google Scholar]
- The physical impacts of microplastics on marine organisms: A review. Environ. Pollut.. 2013;178:483-492.
- [Google Scholar]
- Photo aging and fragmentation of polypropylene food packaging materials in artificial seawater. Water Res.. 2021;188:116456
- [Google Scholar]
- Degradation of polyethylene by a fungus, Penicillium simplicissimum YK. Polym. Degrad. Stab.. 2001;72(2):323-327.
- [Google Scholar]
- A bacterium that degrades and assimilates poly(ethylene terephthalate) Science. 2016;351(6278):1196-1199.
- [Google Scholar]
- Dechlorination behaviour of flexible poly(vinyl chloride) in NaOH/EG solution. Polym. Degrad. Stab.. 2008;93(10):1822-1825.
- [Google Scholar]
- Bringing Home the Trash: Do Colony-Based Differences in Foraging Distribution Lead to Increased Plastic Ingestion in Laysan Albatrosses? PLoS ONE. 2009;4(10):e7623
- [Google Scholar]
- Photodegradation and photostabilization of polymers, especially polystyrene: review. SpringerPlus. 2013;2(1):398.
- [Google Scholar]
- Current technologies for plastic waste treatment: A review. J. Cleaner Prod.. 2021;282:124523
- [Google Scholar]
- Transport of microplastics in coastal seas. Estuar. Coast. Shelf Sci.. 2017;199:74-86.
- [Google Scholar]
- Biodegradation of polyethylene microplastic particles by the fungus Aspergillus flavus from the guts of wax moth Galleria mellonella. Sci. Total Environ.. 2020;704:135931
- [Google Scholar]
- A study on the biodegradability of polyethylene terephthalate fiber and diethylene glycol terephthalate. J. Appl. Polym. Sci.. 2004;93(3):1089-1096.
- [Google Scholar]
- A Review of Plastic Waste Biodegradation. Crit. Rev. Biotechnol.. 2005;25(4):243-250.
- [Google Scholar]
- Zhongming, Z., Linong, L., Xiaona, Y., Wangqiang, Z., Wei, L., 2018. COP24: How a plastics treaty could clean up our oceans.
- Determination of bisphenol A, 4-n-nonylphenol, and 4-tert-octylphenol by temperature-controlled ionic liquid dispersive liquid-phase microextraction combined with high performance liquid chromatography-fluorescence detector. Talanta. 2011;85(3):1598-1602.
- [Google Scholar]
- Exposure of soil collembolans to microplastics perturbs their gut microbiota and alters their isotopic composition. Soil Biol. Biochem.. 2018;116:302-310.
- [Google Scholar]







