Translate this page into:
Determination of the pharmacodynamic substances and mechanism of Shiwuwei Saierdou Pills against cholestatic hepatitis through chemical profile identification and network pharmacology analysis
⁎Corresponding authors at: State Key Laboratory of Southwestern Chinese Medicine Resources, Chengdu University of Traditional Chinese Medicine, 1166 Liutai Avenue, Wenjiang District, Chengdu 611137, China. winter9091@163.com (Shaohui Wang), zhangyi@cdutcm.edu.cn (Yi Zhang)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Abstract
Cholestatic hepatitis (CH) is a liver lesion caused by abnormal bile production, secretion and excretion and has a complex pathogenesis. The Tibetan medicine Shiwuwei Saierdou Pills (SSP) is an empirical Tibetan medicine formula for the treatment of CH, but its chemical composition is complex and the material basis of its efficacy is not yet clear. So, in this study, the main chemical constituents and its blood-incorporated constituents in SSP were analyzed by ultra-high-performance liquid chromatography-quadruple-electrostatic field orbitrap high resolution mass spectrometry (UHPLC-Q-Exactive Orbitrap/MS), Then, the blood-incorporated constituents were subjected to network pharmacology analysis to preliminarily clarify its potential pharmacological substances and mechanism. and further, it was verified through molecular docking and in vivo animal experiments. As a result, a total of 80 chemical components were identified in the SSP, of which 11 were confirmed by reference standards and 20 blood-incorporated constituents (including 10 prototypes and 10 metabolites) were characterized in the serum containing the medicine. The core targets of SSP for the treatment of CH were identified as AKT1, VEGFA, CASP3, SRC and MAPK3 through the screening of the relationship between the blood-incorporated constituents and the targets. Combined with the results of molecular docking, swertiamarin, ellagic acid, taurocholic acid and bellidifolin in the ten prototypes may be the key pharmacodynamic substances for SSP to treat CH. The results of animal experiments showed that SSP could significantly inhibit the pathological changes of the CH rat model, and inhibit the protein expression of AKT1, VEGFA, CASP3, SRC and MAPK3. In summary, we used network pharmacology, molecular docking and animal experiments to preliminarily determine the main medicinal components, targets and pathways of SSP in the treatment of CH, which provides a scientific basis for further revealing the material basis and mechanism of SSP in treating CH.
Keywords
Shiwuwei Saierdou Pills
Cholestatic hepatitis
UHPLC-Q-Exactive Orbitrap/MS
Network Pharmacology
Molecular docking
- CH
-
Cholestatic hepatitis
- SSP
-
Shiwuwei Saierdou Pills
- 2D
-
Two dimensional
- 3D
-
Three dimensional
- AKT1
-
AKT Serine/Threonine Kinase 1
- ALP
-
Alkaline phosphatase
- ALT
-
Alanine aminotransferase, ANIT, α-naphthyl isothiocyanate
- AST
-
Glutathione transaminase
- BP
-
Biological Processes
- CASP3
-
Caspase-3
- CC
-
Cellular Components
- CLD
-
Cholestatic Liver Diseases
- FDA
-
Food and Drug Administration
- GGT
-
Gamma glutamyl transpeptidase
- GO
-
Gene Ontology
- HE
-
Hematoxylin-eosin
- HSCs
-
Hepatic stellate cells
- IL-6
-
Interleukin-6
- KEGG
-
Kyoto Encyclopedia of Genes and Genomes
- MAPK3
-
Mitogen-Activated Protein Kinase 3
- MF
-
Molecular Functions
- PDB
-
Protein Data Bank
- PPI
-
Protein-Protein Interaction
- Rt
-
Retention time
- SD
-
Sprague Dawley
- SDF
-
Software Development File
- SPF
-
Specific pathogen free
- SRC
-
Proto-oncogene tyrosine-protein kinase SRC
- TBIL
-
Total bilirubin
- UDCA
-
Ursodeoxycholic acid
- UHPLC-Q-Exactive Orbitrap/MS
-
ultra-high-performance liquid chromatography-quadruple-electrostatic field orbitrap high resolution mass spectrometry
- VEGFA
-
Vascular Endothelial Growth Factor A
- VEGFR
-
Vascular endothelial growth factor receptor
Abbreviations
1 Introduction
Hepatitis, a prevalent hepatic disorder, is categorized into jaundiced and non-jaundiced types based on the manifestation of jaundice, with cholestatic hepatitis (CH) constituting 2 % to 8 % of the jaundiced cases (Yang and Wang, 2021). CH, also termed biliary or bile duct hepatitis, encompasses a spectrum of liver pathologies instigated by diverse etiological factors. Disruptions at any stage of bile processing-from formation to excretion-might precipitate bile stasis, potentially escalating to hepatic impairment (Lindholm et al., 2022). Correspondingly, epidemiological investigations in China report that over 10 % of chronic liver disease hospital admissions are attributed to CH (Fang et al., 2020). Early detection of cholestasis is challenging; often, clinical findings include nonspecific symptoms alongside raised serum markers of ALP and GGT (Boyer, 2013, Lindholm et al., 2022). Cholestasis has no fixed group and can occur in any population and is generally benign(Li et al., 2020b). While CH may appear in any demographic and often has a benign course, protracted and intense bile obstruction can severely disrupt enterohepatic circulation, advancing toward fibrosis, cirrhosis, carcinomas, and eventual mortality (Han and Tian, 2016). Global mortality from viral hepatitis surpasses a million annually (Li et al., 2018), yet the only drug currently approved by the FDA for the treatment of cholestasis is ursodeoxycholic acid (2015). Hence, novel therapeutic strategies for CH require exploration.
The Shiwuwei Saierdou Pills (SSP) of Tibetan medicine is a compound formulation derived from the empirical amalgamation of two traditional Tibetan remedies: Bawei Zhangyacai Pills and Wuwei Jinse Pills. Chronicled within the seminal Tibetan medical text, 'Lan Liu Li', the Bawei Zhangyacai concoction is heralded for its efficacy in treating biliary tumors within the small intestine, espousing a therapeutic approach that induces clear evacuation of the bowels. Concurrently, the Wuwei Jinse Pills are traditionally prescribed by Tibetan practitioners to alleviate jaundiced hepatitis. The synergistic combination of these two formulas is applied to address an array of hepato-biliary ailments, including hepatic fever, cholecystitis, obstruction of the common bile duct, and cholelithiasis (Pan et al., 2021). Comprising a blend of fifteen fifteen herbs: Swertiae chirayitae Herba, Chrysosplenii Herba, Nitroum, Hypecoe Herba, Lagotis Herba, Aconiti tangutici Herba, Punicae granati Semen, Herpetospermi Semen, Berberidis Cortex, Trogopteri Faeces, Saussureae Herba, Scrofa Faeces, Vladimiriae Radix, Chebulae Fructus and Vermiculitum, SSP represents a complex herbal ensemble. The intricate nature of its phytochemical constitution poses challenges in deciphering the precise foundational components responsible for its purported therapeutic impacts, thus hindering broader.
Serological pharmacological profiling is a potent approach to rapidly discern the biologically active components of Chinese medicinal formulations following oral administration. This process entails tracing and characterizing the metabolites present in systemic circulation, thereby reflecting the distinctive pharmacokinetic properties of orally-administered Chinese medicinal therapies. The Q-Exactive system, founded upon Orbitrap technology, integrates a highly selective quadrupole for ion filtration with high-resolution, precise mass measurement, yielding an analytical method that is both highly discriminating and sensitive (Zhu et al., 2020, Zhang et al., 2021). Network pharmacology extends the boundaries of pharmacological research by mapping drug actions and disease mechanisms within expansive biological networks, underpinned by principles of systems biology (Li et al., 2022a). Concurrently, molecular docking provides an in silico platform for drug screening, efficiently delineating possible molecular targets for pharmaceutical compounds. The intricate nature of Tibetan medicinal formulations poses challenges due to their complex chemical constitution and the pleiotropy of their pharmacological targets (Zhang et al., 2019). This complexity is compounded by the paucity of investigations into the foundational material contributors to their therapeutic efficacy. To bridge this knowledge gap, we amalgamated network pharmacology with molecular docking strategies to unravel the specific pathways through which SSP exert therapeutic effects in the context of CH. This multidisciplinary inquiry aspires to furnish a more rigorous scientific rationale for employing SSP in treating CH.
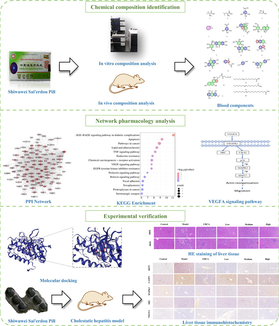
In this investigation, we conducted a comprehensive analysis of the chemical constituents of SSP both in vitro and in the plasma of rats post-gavage administration. Utilizing ultra-high-performance liquid chromatography-quadruple-electrostatic field orbitrap high resolution mass spectrometry (UHPLC-Q-Exactive Orbitrap/MS), we were able to identify and categorize the intricate array of bioactive compounds. Building on these findings, a network pharmacology approach was employed to elucidate the potential biological targets and pathways influenced by SSP's components. Key molecular targets were then subjected to further validation through the use of molecular docking techniques and in vivo experimental models (Fig. 1). The outcomes of this research lay the groundwork for a deeper understanding of the active substances within SSP and their corresponding mechanisms of action, contributing valuable scientific insights. Moreover, these findings underpin the pharmacological validation required for the broader clinical application of SSP as a treatment modality for CH.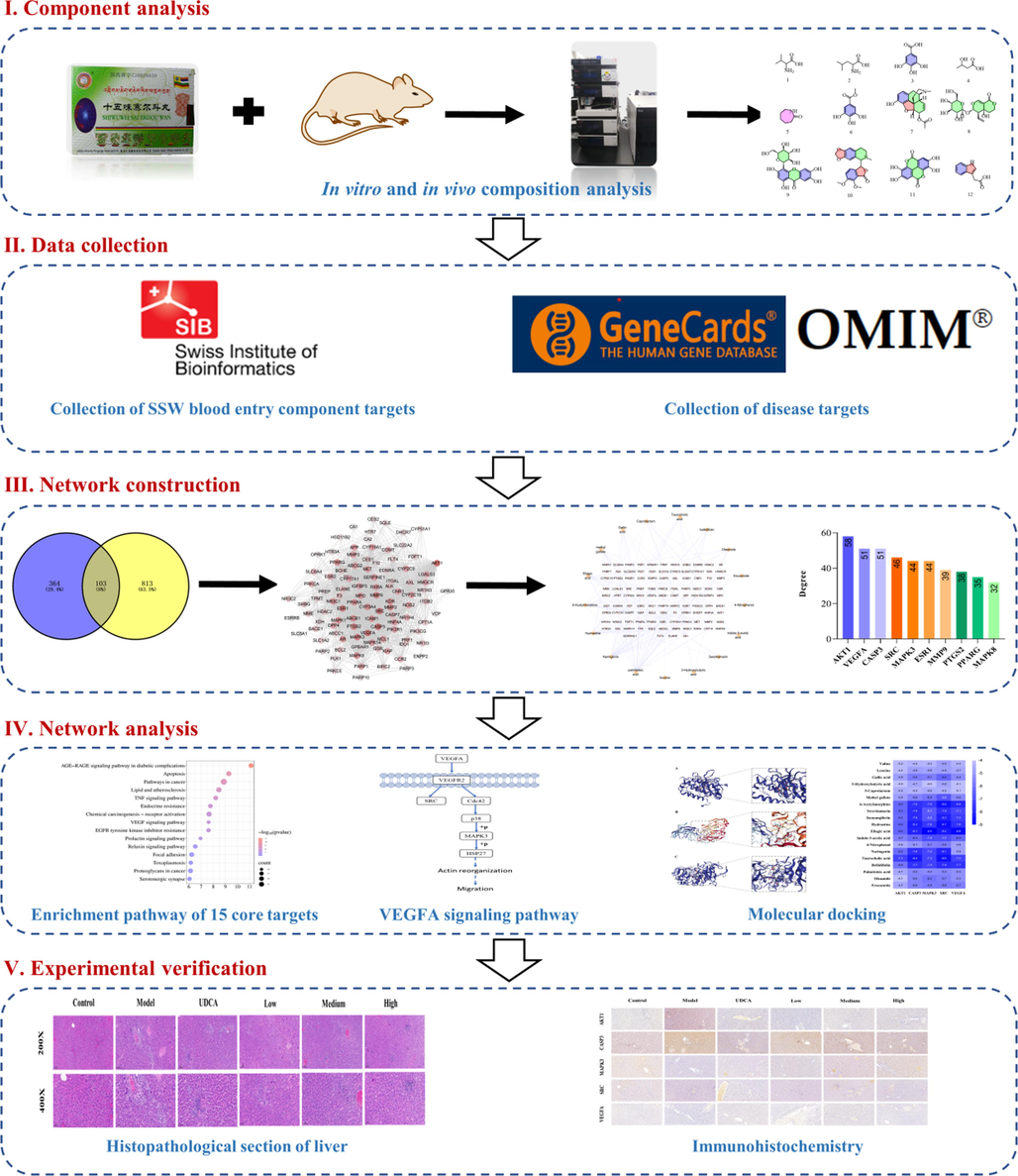
The workflow of research on the pharmacodynamic substances and mechanism of SSP in treating CH.
2 Materials and methods
2.1 Main instruments
Vanquish Ultra Performance Liquid Chromatography coupled with Q Exactive Quadrupole-Electrostatic Field Orbital Trap High Resolution Mass Spectrometer (Thermo Fisher Scientific, USA); Waters Acquity UPLC HSS T3 C18 (2.1 mm × 100 mm,1.8 μm); Electronic Analytical Balance Type BSA124S (Beijing Sartorius Scientific Instruments Co., Ltd., Beijing, China); SB-8200DTS Type Dual Frequency Ultrasound Instrument (Ningbo Xingyi Ultrasound Equipment Co., Ltd., Ningbo, China); TGL-16 M benchtop high-speed frozen centrifuge (Changsha Xiangyi Centrifuge Instrument Co., Ltd., Changsha, China); XW-80A Vortex Mixer (Shanghai Chitang Electronics Co., Ltd., Shanghai, China); UPH-I-10 T UPP Series Ultra Pure Water Machine (Sichuan UPP Ultra Pure Technology Co., Ltd., Chengdu, China); JCS-110020 electronic balance (Harbin Zhong Hui Weighing Instrument Co., Ltd., Haerbing, China); FC-9 Enzyme Labeler (Shanghai Meigu Molecular Instruments, Shanghai, China).
2.2 Main drugs and reagents
SSP (Guomadian Z20026038, Batch No. 20201002) was provided by Qinghai Jiumei Tibetan Medicine Pharmaceutical Co. Ltd. (Xining, China). Protopine (batch no. DSTDY011301, purity ≥ 98 %), Coptisine (batch no. DST201105-003, purity ≥ 98 %), Gallic acid (batch no. DSTDM000801, purity ≥ 98 %), Ellagic acid (batch no. DSTDR000401, purity ≥ 98 %), Dehydrocostus lactone (batch no. DSTDQ004201, purity ≥ 98 %), Costunolide (batch no. DSTDM003002, purity ≥ 98 %), Mangiferin (batch no. DST200719-031, purity ≥ 98 %), Swertiamarin (batch no. DST201020-003, purity ≥ 98 %), Quercetin (batch no. DSTDH002802, purity ≥ 98 %), Hordenine (batch no. DST210615-041, purity ≥ 98 %), Scopoletin (batch no. DST210930-064, purity ≥ 98 %) were obtained from Chengdu Desite Biotechnology Co. Ltd. (Chengdu, China); α-naphthyl isothiocyanate (ANIT), Ursodeoxycholic acid (UDCA), Peanut oil, were obtained from Shanghai Yi'en Chemical Technology Co. Ltd. (Shanghai, China).
2.3 Experimental animals
Specific pathogen free (SPF) grade male SD rats, weight (200 ± 20) g, provided by Chengdu Dashuo Experimental Animal Co. (Animal Licence No. SCXK (Chuan) 2020–030) and from Hunan Enswell Laboratory Animals Co Ltd (Production Certificate No. SCXK(Xiang)2019–0004). All rats were housed for 7 days in 12 h of light and 12 h of darkness, at 23 ± 2 °C and 40 %-70 % humidity, with free access to food and water. The animals were fasted for 12 h before administration and were not water fasted throughout. All animals were housed in the animal house of Chengdu University of Traditional Chinese Medicine, NO: TCM-09–315, Laboratory Animal License No. SYXK(Chuan)2020–124.
2.4 Chromatographic conditions
Chromatographic columns: Waters Acquity UPLC HSS T3 C18 (2.1 mm × 100 mm, 1.8 μm); Mobile phase: 0.1 % formic acid aqueous solution (A) − 0.1 % formic acid acetonitrile solution (B); gradient elution: (0 ∼ 48 min, 5%B;48 ∼ 55 min, 95 %B); Flow rate: 0.3 ml/min; Column temperature: 35℃; Injection volume: 2 μl. The retention time tolerances for this experiment were all in the range of 0.5 min.
2.5 Mass spectrometry conditions
Electrospray ionisation sources (ESI), Positive ion/negative ion scanning mode, Spray voltage: 3500 V(+) / 3500 V(-), Ion transfer tube temperature: 320 °C, Auxiliary gas heating temperature: 350 °C, Auxiliary gas flow rate: 10arb, Sheath gas flow rate: 35arb, Scan mode: Full MS dd-MS2, Full MS resolution: 35000, dd-MS2 resolution: 17500, Scan range: 100 ∼ 1500, Collision energy gradients: 20, 40, 60 eV.
2.6 Preparation of control solutions
Precise weigh the amount of protopine, coptisine, gallic acid, ellagic acid, costunolide, mangiferin, swertiamarin, quercetin, hordenine, scopoletin, dehydrocostus lactone, add chromatographic methanol to 10 ml, weigh, sonicate for 30 min, weigh again and make up the weight to obtain the mixed standard solution.
2.7 Preparation of the test solution
Take about 0.3 g of SSP powder and weigh it precisely. Place it in a 50 ml conical flask, add 10 ml of chromatographic methanol, weigh the mass, sonicate for 30 min, weigh again, make up the lost mass with chromatographic methanol and shake well. The liquid is packed in 1.5 ml EP tubes and centrifuged for 5 min and the supernatant was passed through a 0.22 μm microporous membrane and the filtrate was renewed to obtain the test solution.
2.8 Preparation of the gavage solution
SSP powder, dissolved in pure water to a concentration 10 and 20 times the clinical dosing concentration (1.35 g/10 ml/kg, 2.7 g/10 ml/kg, converted from 1.5 g of SSP for a single dose in 70 kg of human body), for serum medicinal chemistry tests.
SSP powder was dissolved in saline into high (0.95 g/kg), medium (0.47 g/kg) and low (0.24 g/kg) doses (equivalent to 2, 1 and 0.5 times the daily clinical dosage in humans, based on a single dose of 1.5 g of SSP for 60 kg of human body) and used in network pharmacological validation experiments.
2.9 Group dosing and sample collection
For the experiments on the analysis of blood-incorporated constituents in SSP, six male SPF grade SD rats were randomly divided into three groups of two rats each. The control cohort received purified water, while the two treatment cohorts were administered either 10-fold or 20-fold concentrations of SSP via gavage at a dosage of 10 ml/kg. The dosing regimen spanned three days, consisting of morning, afternoon, and evening administrations, with a mandated 12-hour fasting period prior to the final dose, although water access remained unrestricted. Pursuant to the final gavage, and in compliance with approved ethical guidelines, blood collection was performed via the orbital plexus under deep anaesthesia to ensure a humane endpoint for each animal. These samples were collected at specified time points (15, 30, 45, 60, 90, 120, 180, 240, and 360 min post-administration) into heparinized 1.5 ml microcentrifuge tubes, centrifuged at 4000 rpm for 15 min at 4 °C. The resultant plasma was then stored at −80 °C for downstream in vivo chemical composition analysis.
For in vivo animal validation experiments, thirty-six SPF grade SD male rats were randomly divided into 6 groups: normal group, model group, SSW high, medium and low dose group and positive drug group (ursodeoxycholic acid). Rats were dosed at 10 ml/kg once a day for 7 days. After gavage on day 5 of the dosing period, at an interval of 4 h, all groups except the normal group were moulded by gavage with 100 mg/kg ANIT, and the normal group was gavaged with the same volume of peanut oil, and then the dosing was continued at the original dose. Liver tissue was taken from each animal and placed in saline, rinsed to remove excess blood, blotted dry with filter paper and fixed in 4 % paraformaldehyde fixative, dehydrated, paraffin embedded and stained with HE for histopathological sections as well as immunohistochemical studies. The rats were anaesthetized by intraperitoneal injection of 1 % sodium pentobarbital saline solution at a dose of 35 mg/kg in all the above experiments.
2.10 Plasma sample processing
Add 200 μl of plasma, add 3 times the volume of acetonitrile to precipitate the protein, vortex for 3 min, centrifuge at 12,000 r/min for 20 min at 4 °C. Aspirate the supernatant, blow dry under nitrogen at 37 °C, add 200 μl of methanol to the residue, vortex for 3 min, centrifuge at 12,000 r/min at 4 °C, aspirate the supernatant and keep it for the sample. Blank plasma and administered plasma were operated according to this method.
2.11 Data processing
Raw liquid mass data was imported into Compound Discoverer 3.1 and a workflow was established for the identification of unknown compounds with primary and secondary mass deviations of < 5 ppm. The exact mass numbers and molecular formulae of the compounds were obtained from the mass-to-charge ratios of the primary excimer ion peaks, and then combined with the information of the secondary fragment ions, MassFrontier software, mzCloud web database, mzVault local Chinese medicine composition database search and reported literature as well as some of the controls, to analyse and identify the chemical composition of SSP and rat plasma samples. The chemical composition of the pills and rat plasma samples were analyzed and identified.
2.12 Network pharmacological analysis of 20 blood-incorporated constituents
Utilizing the ChemSpider database, molecular files in the 'MOL' format for components that entered the bloodstream were retrieved for analysis of serum samples from rats administered SSP via gavage. Subsequent target prediction for these bioactive molecules was carried out through the SwissTargetPrediction (https://swisstargetprediction.ch/) platform. After removing duplicate targets, the analysis yielded pertinent targets (relevance score > 0) for 20 blood-incorporated constituents. To identify targets associated with CH, a query was performed using the Gene Cards (https: //https://www.genecards.org/) and OMIM (https://omim.org/) databases, from which a curated list of relevant targets was compiled.
The data pertaining to targets associated with both the bioactive components of SSP and CH were integrated using Venny 2.1, which facilitated the identification of overlapping targets. These shared targets between the pharmacological agents and disease states were further analyzed on the STRING database by selecting “multiple proteins” and specifying the species as “Homo sapiens” This process enabled the acquisition of STRING (https://string-db.org/) derived information to construct the protein–protein interaction (PPI) network. The resulting data was imported into Cytoscape 3.9.1 and GraphPad Prism 9.2.0 software suites for graphical representation.
Pathway enrichment analysis was subsequently conducted with the intersecting targets using the DAVID 2021 (Dec. 2021) database. The parameters for this analysis were set to OFFICIAL GENE SYMBOL for the identifier, Gene list for the list type, and “Homo sapiens” for the protein species. Downloaded results encompassed “GOTERM_BP_DIRECT” for biological processes, “GOTERM_CC_DIRECT” for cellular components, “GOTERM_MF_DIRECT” for molecular functions, and “KEGG_PATHWAY” for pathway enrichment. From these findings, the top 10 GO terms and top 15 KEGG pathways, as determined by P-values, were selected for highlighting in the presentation and elaboration of this study.
2.13 Molecular docking
The PDB Format of the top 5 target proteins by degree and the SDF format of the blood-incorporated constituents were collected in the Uniprot and PubChem databases for molecular docking via the CB-DOCK2 website (https://cadd.labshare.cn/cb-dock2/php/index.php) and save the molecular docking results.
2.14 Histological examination
Liver tissues that had been fixed for more than 24 h were removed from the fixative for trimming, dehydration, embedding, sectioning, HE staining and dehydration sealing, followed by microscopic examination and photographic analysis to assess morphological changes in each group.
2.15 Immunohistochemistry
Rat liver tissues previously fixed in 4 % paraformaldehyde fixative were removed and subjected to immunohistochemical experimental steps such as trimming, embedding, sectioning, staining and dehydration, followed by photographic analysis using Image-Pro Plus 6.0 to validate the five core targets closely associated with SSP anti-CH as screened by network pharmacology. Three 200 × fields of view were randomly selected for each section of rat liver tissue in each group to be photographed. The photographs were taken so that as much tissue as possible filled the entire field of view, and to ensure that the background light was as uniform as possible for each photograph. Image-Pro Plus 6.0 was used to select the same brown color as the standard for all positive photographs, and each photograph was analyzed to obtain the cumulative optical density (IOD) and the pixel area of tissue (AREA) for each positive photograph.
3 Results
3.1 In vitro chemical composition identification
Under the above conditions, analysis of the methanolic extracts from SSP yielded the identification of 80 distinct chemical constituents. These included a diverse array of compounds: 17 alkaloids, 11 organic acids, 9 phenylpropanoids, 18 flavonoids, 8 terpenoids, 6 phenols, along with 11 other identified substances. To ensure the reliability of our findings, comparison with reference standards was performed, which confirmed the identity of 11 constituents within this profile. Visualization of the chemical composition was facilitated through both positive and negative ionization modes, which are comprehensively depicted in the mass flow diagrams presented in Fig. 2. Detailed characterizations of the principal chemical entities recognized in our analysis are collated and delineated in Table 1. This array of constituents underscores the complex phytochemical framework that SSP embodies, setting the stage for subsequent bioactivity correlations. Note: A, Alkaloids; B, Organic acids; C, Phenylpropanoids; D, Flavonoids; E, Terpenoids; F, Phenols; G, Others respectively. “*” is the ingredient confirmed by comparing with the reference substance.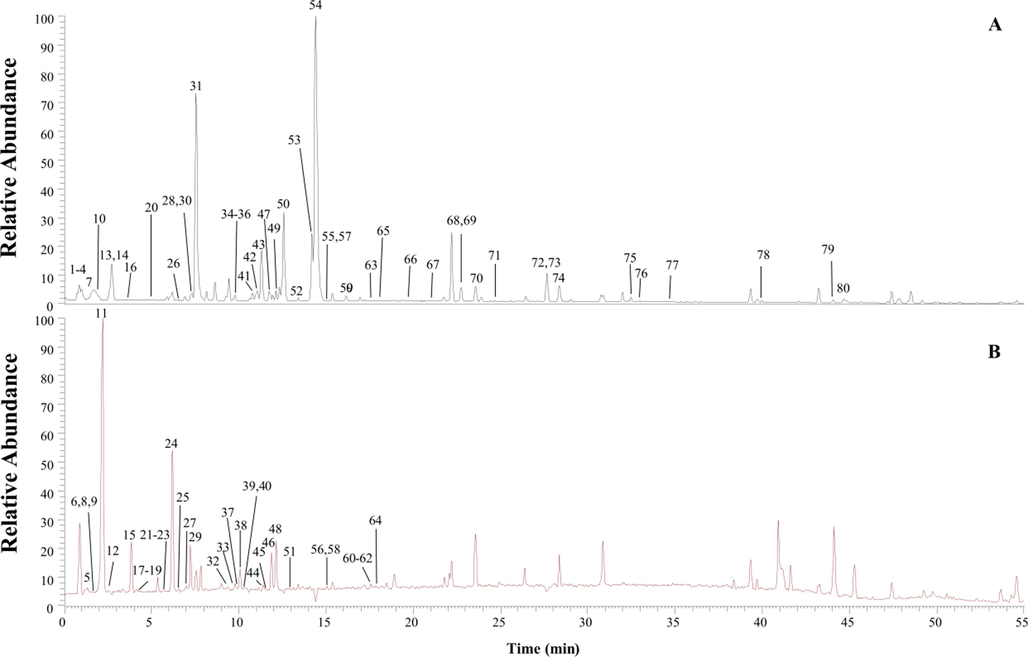
Total ion flow diagram in the positive (A) and negative (B) ion modes of SSP.
NO.
tR/min
Molecular formula
Precursor ion
Measured ion
Ion type
MS/MS(m/z)
Component
Classification
1
0.87
C5H13NO
103.10010
103.10015
[M + H]+
104.10733,58.06580,60.08146
Choline(Bruce et al., 2010, Gill et al., 2020)
B
2
0.91
C7H7NO2
137.04770
137.04772
[M + H]+
138.05504,94.06558,92.05013
Trigonelline(Lang et al., 2008)
A
3
1.02
C5H7NO3
129.04300
129.04297
[M + H]+
130.05000,102.05524,84.04494,77.63916
Pyroglutamic acid(Qu et al., 2002)
G
4*
1.10
C10H15NO
165.11550
165.11551
[M + H]+
166.12263,121.06499,103.05468
Hordenine(Steiner et al., 2016)
A
5
1.29
C7H10O5
174.05226
174.05226
[M−H]-
173.04494,129.01848,111.00776
Shikimic acid (Li et al., 2021)
B
6
1.30
C4H6O5
134.02070
134.02069
[M−H]-
133.01337,115.00274,71.01277
Malic acid (Birkler et al., 2010)
B
7
1.36
C4H4N2O2
112.02770
112.02771
[M + H]+
113.03482,96.00844,70.02937
Uracil (Tafzi et al., 2020)
G
8
1.48
C6H6O6
174.01590
174.01591
[M−H]-
173.08185,137.02399,111.00781,93.03360,85.02845,73.02847
Cis-Aconitic acid (Xiong et al., 2021)
B
9
1.58
C4H6O4
118.02570
118.02575
[M−H]-
117.05405,116.92759,99.02480,73.02842
Succinic acid (Yang et al., 2016)
B
10*
2.01
C10H15NO
165.11550
165.11551
[M + H]+
166.12270,121.06500,103.05466
Hordenine(Steiner et al., 2016)
A
11*
2.19
C7H6O5
170.02090
170.02091
[M−H]-
169.01353,125.02351,124.01560,97.02856,81.03366,79.01788,69.03351
Gallic acid (Huang et al., 2017, Ren et al., 2021)
B
12
2.19
C6H6O3
126.03080
126.03079
[M−H]-
125.02354,97.02837,81.03336,61.39196
Pyrogallol (Dutschke et al., 2021)
F
13
2.75
C12H16N2O
204.12630
204.12631
[M + H]+
205.13388,160.07574,142.06523,132.08086,115.05460,79.79268,67.02159,58.06585
Bufotenin (Costa et al., 2005)
A
14
2.88
C6H6O3
126.03200
126.03202
[M + H-H2O]+
109.02871,81.03405,53.03931
5-Hydroxymethylfurfural (Zhou and Qi, 2017)
G
15
3.85
C7H6O4
154.02590
154.02589
[M−H]-
153.01851,109.02852
2,3-Dihydroxybenzoic (Cheiran et al., 2019)
B
16
3.86
C6H6O3
126.03200
126.03199
[M + H]+
127.03914,97.02858,71.04969,55.01853
Maltol (Li et al., 2011a; Zhang et al., 2002)
G
17
4.13
C16H24O10
376.13701
376.13701
[M−H]-
375.12570,213.07629,195.06602,169.08629,161.04520,151.07524
8-epi-loganic acid (Wang, 2021)
E
18
4.29
C16H18O9
354.09540
354.09535
[M−H]-
353.08743,191.05566,179.03435,173.04437,135.04431
Neochlorogenic acid (Fang et al., 2002)
C
19
4.32
C16H24O10
376.13700
376.13698
[M−H]-
376.13339,375.12900,213.07639,179.03410,169.08633,125.05962,113.02348
Loganic acid (Zhou et al., 2021, Abirami et al., 2022)
E
20
4.89
C12H12N2O
200.09510
200.09513
[M + H]+
201.10213,186.07864,160.07579,114.94859,81.07304
Harmalol (Zhang et al., 2013)
F
21
5.74
C8H8O5
184.03660
184.03663
[M−H]-
183.02925,140.01064,124.01566
Methyl gallate (Gong et al., 2020)
F
22
5.75
C7H6O4
154.02590
154.02588
[M−H]-
153.01851,123.00783,109.02851,108.02068
Gentisic acid (Yao et al., 2020)
F
23
5.90
C16H18O9
354.09520
354.09518
[M−H]-
353.08875,191.05548,161.02380
Chlorogenic acid (Zhang et al., 2010, Choi et al., 2018)
C
24*
6.15
C16H22O10
420.12660
420.12663
[M + HCOOH-H]-
419.11935,179.05539,161.04469,141.01843
Swertiamarin (Li et al., 2011b)
E
25
6.58
C9H6O4
178.02630
178.02628
[M−H]-
179.02368,178.05304,175.96822,177.01865,176.83632,133.02861,121.02853,116.92731,105.03364
Esculetin (Yang et al., 2017)
C
26
6.58
C16H17NO3
271.12090
271.12093
[M + H]+
272.12808,255.10139,237.09093,209.09583,161.05962,143.04916,123.04419,107.04949
Higenamine (Wang et al., 2020)
A
27
6.92
C7H6O4
154.02590
154.02588
[M−H]-
153.01846,152.89421,109.02850,108.02081
Protocatechuic acid (Li et al., 2017)
F
28*
7.22
C19H18O11
422.08491
422.08491
[M + H]+
423.09219,405.08167,359.13364,303.04977,167.01286
Mangiferin (Khurana et al., 2017)
D
29
7.23
C19H18O11
422.08480
422.08482
[M−H]-
421.07764,331.04593,301.03531
Isomangiferin (Aabideen et al., 2020)
D
30
7.26
C16H22O9
358.12650
358.12645
[M + H]+
359.13373,197.08073,179.07018,127.03906
Sweroside (Sheng et al., 2014)
E
31
7.54
C20H23NO4
341.16300
341.16292
[M + H]+
342.16986,297.11203,282.08856,265.08585,237.09108
Magnoflorine (Sharma et al., 2020; Tian et al., 2014a)
A
32
9.32
C34H28O22
788.10790
788.10791
[M−H]-
787.09906,617.07733,465.06805,447.05438,313.05780,169.01355
1,2,3,6-Tetra-O-galloyl-β-D-glucose(Owen et al., 2003)
G
33
9.60
C29H36O16
640.20040
640.20044
[M−H]-
639.19299,477.16125,179.03438,161.02367,133.02861
Plantamajoside (Bai et al., 2017)
C
34
9.61
C15H16O8
324.08460
324.08461
[M + H]+
325.09180,163.03894,135.04411
Skimmin (Lou et al., 2020)
C
35*
9.62
C10H8O4
192.04240
192.04237
[M + H]+
193.04951,178.02605,165.05479,150.03110,137.05975,133.02844,122.03632,105.07030,94.04187
Scopoletin (LI et al., 2022b; Wang et al., 2021; Zeng et al., 2015)
C
36
9.78
C10H10O4
194.05800
194.05802
[M + H]+
195.08740,193.15872,149.05971,145.02841,117.03373
Ferulic acid (Huang et al., 2014; Zhang et al., 2018b)
B
37
9.87
C27H30O16
610.15360
610.15363
[M−H]-
609.14600,343.04520,300.02744,271.02481,151.00293
Rutin (He et al., 2014)
D
38*
10.07
C14H6O8
302.00630
302.00625
[M−H]-
300.99884,283.99612,257.00876,229.01373,185.02370
Ellagic acid (Yan et al., 2014)
F
39
10.32
C21H20O12
464.09580
464.09580
[M−H]-
463.08807,300.02753,271.02475,151.00276
Isoquercitrin (Zhang et al., 2017)
D
40
10.36
C29H36O15
624.20570
624.20570
[M−H]-
623.19843,461.16479,315.10901,179.03447,161.02365,135.04425,133.02858,113.02342
Verbascoside (Plaza et al., 2005, Wu et al., 2006, Xie et al., 2017)
F
41
10.78
C21H25NO4
355.17840
355.17836
[M + H]+
356.18555,192.10184,177.07835
Tetrahydropalmatine (Wang et al., 2019)
A
42
10.96
C27H30O15
594.15860
594.15863
[M + H]+
595.16595,593.15143,285.04044,284.03268,255.02959
Kaempferol-3-O-rutinoside (Dou et al., 2017; Li et al., 2020a)
D
43*
11.30
C20H19NO5
353.12640
353.12639
[M + H]+
354.13342,189.07861,188.07057,149.05974
Protopine (Huang et al., 2014)
A
44
11.32
C25H24O12
516.12680
516.12680
[M−H]-
517.12518,515.11249,353.08768,173.04465,161.02365
4,5-Dicaffeoylquinic (de Souza et al., 2015, Pantoja Pulido et al., 2017)
C
45
11.57
C21H20O11
448.10070
448.10070
[M−H]-
447.09299,301.03516,300.02747,284.03229
Kaempferol-3-glucoside (Abu Bakar et al., 2020)
D
46
11.66
C9H8O3
164.0469
164.0469
[M−H]-
163.03943,119.04929
p-Coumaric acid (Yao et al., 2017)
B
47
11.73
C21H20O10
432.10580
432.10577
[M + H]+
431.09805,268.03778,269.04593
Apigetrin (Yilmaz et al., 2018)
D
48
12.08
C25H24O12
516.12670
516.12666
[M−H]-
515.04901,353.08768,191.05547,179.03429,173.04483,135.04422
Isochlorogenic acid C(Huang et al., 2015)
C
49
12.11
C21H23NO5
369.15760
369.15861
[M + H]+
370.16479,352.15436,336.12405,290.09338,206.08112,189.07838,188.07063,165.09113
Allocryptopine (Huang et al., 2018)
A
50
12.57
C20H19NO4
305.10530
305.10527
[M + H + MeOH]+
338.13846,322.10745,279.08893,265.07349
Jatrorrhizine (Li et al., 2019)
A
51
12.93
C21H20O10
432.10580
432.10581
[M−H]-
431.09824,284.03259,151.00270,107.01329
Afzelin (Abu Bakar et al., 2020, Brito et al., 2021)
D
52
13.40
C29H30O13
586.16870
586.16873
[M + H]+
587.17621,391.10229,373.09274,311.09113,281.08109,247.05984,197.08087
Amarogentin (Kumar and Chandra, 2015)
E
53
13.92
C20H13NO4
331.08450
331.08446
[M + H]+
332.09143,317.06812,274.08600,246.09132,218.09642
Sanguinarine (Xie et al., 2015)
A
54*
14.39
C19H14NO4
320.0909
320.09146
[M + H]+
321.09821,292.09665,290.08130,262.08636,249.07773,234.09081
Coptisine (Cheng et al., 2016)
A
55
15.06
C16H12O5
284.06850
284.06854
[M + H]+
285.07553,270.05209,253.04935,225.05449,214.06219,213.05453,137.02330
Calycosin (Sun et al., 2014)
D
56
15.06
C15H10O6
286.04780
286.04777
[M−H]-
285.04034,270.04449,257.04575,241.05025,217.05042,201.01904,199.03970,198.03168,175.03946,171.04475,151.00288,133.02859,132.02080,121.02863,107.01289
Luteolin (Xie et al., 2017, Jia et al., 2020, Meng et al., 2020)
D
57
15.12
C22H23NO4
365.16310
365.16313
[M + H]+
366.16989,351.14609,350.13846,322.14340,308.12805,306.11206,278.52924
Dehydrocorydaline (Guan et al., 2017)
A
58*
15.14
C15H10O7
302.04270
302.04269
[M−H]-
301.03522,178.99797,151.00284
Quercetin (Zhang et al., 2018a)
D
59
16.15
C20H17NO4
335.11600
335.11604
[M + H]+
336.12286,320.09164,292.09647,278.08099
Berberine (Xu et al., 2015)
A
60
17.49
C16H12O6
300.06340
300.06340
[M−H]-
299.05588,284.03250,136.98711,65.00219
Diosmetin (Shi et al., 2018)
D
61
17.51
C26H45NO7S
515.29160
515.29163
[M−H]-
51428412,124.00652,106.97980,80.96410
Taurocholic acid (Gu et al., 2016)
G
62
17.57
C13H8O6
260.03200
260.03198
[M−H]-
259.02463,231.02921,215.03450,203.03465,187.03944,151.00305
Tetrahydroxyxanthone (Guo et al., 2018)
D
63
17.57
C15H10O3
238.06310
238.06307
[M + H]+
239.06999,221.05798,165.06998,133.08617,121.02856,93.03368,65.03925
3-Hydroxyflavone (Xu et al., 2013)
D
64
17.91
C18H18O4
298.12060
298.12062
[M−H]-
297.11307,189.05511,93.03343
Enterolactone (Parker et al., 2012)
G
65
17.99
C17H14O7
330.07400
330.07403
[M + H]+
331.08096,316.05759,301.03369,168.00523
Jaceosidin (Song et al., 2009)
D
66
19.64
C21H24O6
372.15740
372.15736
[M + H]+
373.16293,355.15359,337.14331,323.12579,305.11682,295.13278,237.11182,177.05486,165.05507,151.07533,137.05975,121.06496
Arctigenin (Zou et al., 2013)
C
67
21.09
C17H14O7
330.07410
330.07410
[M + H]+
331.08130,329.06671,314.04315,299.01971,271.02496
Iristectorigenin B (Mykhailenko et al., 2020)
D
68
22.51
C16H12O5
284.06850
284.06855
[M + H]+
285.07568,270.05209,242.05725,170.11955
5,7-Dihydroxy-4′-methoxyisoflavone (Beszterda et al., 2020)
D
69
22.96
C14H10O6
274.04785
274.04785
[M + H]+
275.04572;273.04037;258.01682;230.02197;186.03223
Bellidifolin (Wang et al., 2015)
D
70
23.43
C17H21NO3
287.15220
287.15218
[M + H]+
289.16284,288.15927,175.07491,161.05911,135.04410,86.09696,84.08147
Piperanine (Friedman et al., 2008)
A
71
24.63
C17H19NO3
285.13650
285.13649
[M + H]+
286.14377,285.13556,215.10611,201.05458,171.04402,143.04916,135.04413,112.07600,98.06050,84.08131,69.07053
Piperine (Chithra et al., 2014)
A
72*
27.66
C15H20O2
232.14658
232.14658
[M + H]+
233.15387,187.14812,159.11681,145.10123,131.08563
Costunolide (Pei et al., 2012)
E
73
28.00
C15H20O2
232.14660
232.14659
[M + H]+
233.15367,215.14308,187.14815,105.07028
Isoalantolactone (Kumar et al., 2014)
E
74*
28.37
C15H18O2
230.13080
230.13083
[M + H]+
231.13792,213.12744,195.11662,185.13248,175.07539,157.10126,143.08556
Dehydrocostus lactone (Kumar et al., 2014)
E
75
32.71
C20H15NO4
333.09980
333.09981
[M + H]+
334.10687,319.08377,276.10138
Dihydrosanguinarine (Xie et al., 2015)
A
76
32.97
C18H30O3
294.21950
294.21963
[M + H-H2O]+
277.21631,249.22151,185.113248,125.09607
9-Oxo-10(E),12(E)-octadecadienoic (Kim et al., 2011)
B
77
34.65
C20H37NO2
323.28260
323.28261
[M + H]+
324.28934,306.52356,109.10160,95.08610,62.06074
Linoleoyl ethanolamide (Palandra et al., 2009)
G
78
39.99
C18H35NO
281.27200
281.27198
[M + H]+
282.27927,247.24199,57.07064
Oleamide (Farha and Hatha, 2019)
G
79
44.07
C22H43NO
337.33450
337.33449
[M + H]+
338.34155,321.31519,303.30380,163.14795,149.13245,135.11687,111.11709
Erucamide (Dabur and Mittal, 2016)
G
80
44.68
C18H37NO
283.28760
283.28755
[M + H]+
284.29474,116.10712,102.09167,71.04993,57.07061
Stearamide (Castillo-Peinado et al., 2019)
G
3.1.1 Alkaloids
Seventeen alkaloid compounds were identified from SSP. Several of these compounds, namely hordenine, protopine and coptisine were compared with the reference standards. For example, the compound dehydrocorydaline (Rt = 15.12 min, C22H23NO4), gives a quasi-ion peak which is m/z 366.17029[M + H]+. It loses one molecule of CH3 and one molecule of CO to form fragment ions m/z 351.14609 [M−CH3]+ and m/z 308.12805 [M−CH3−CH3−CO]+ respectively (Guan et al., 2017) (Fig. 3A).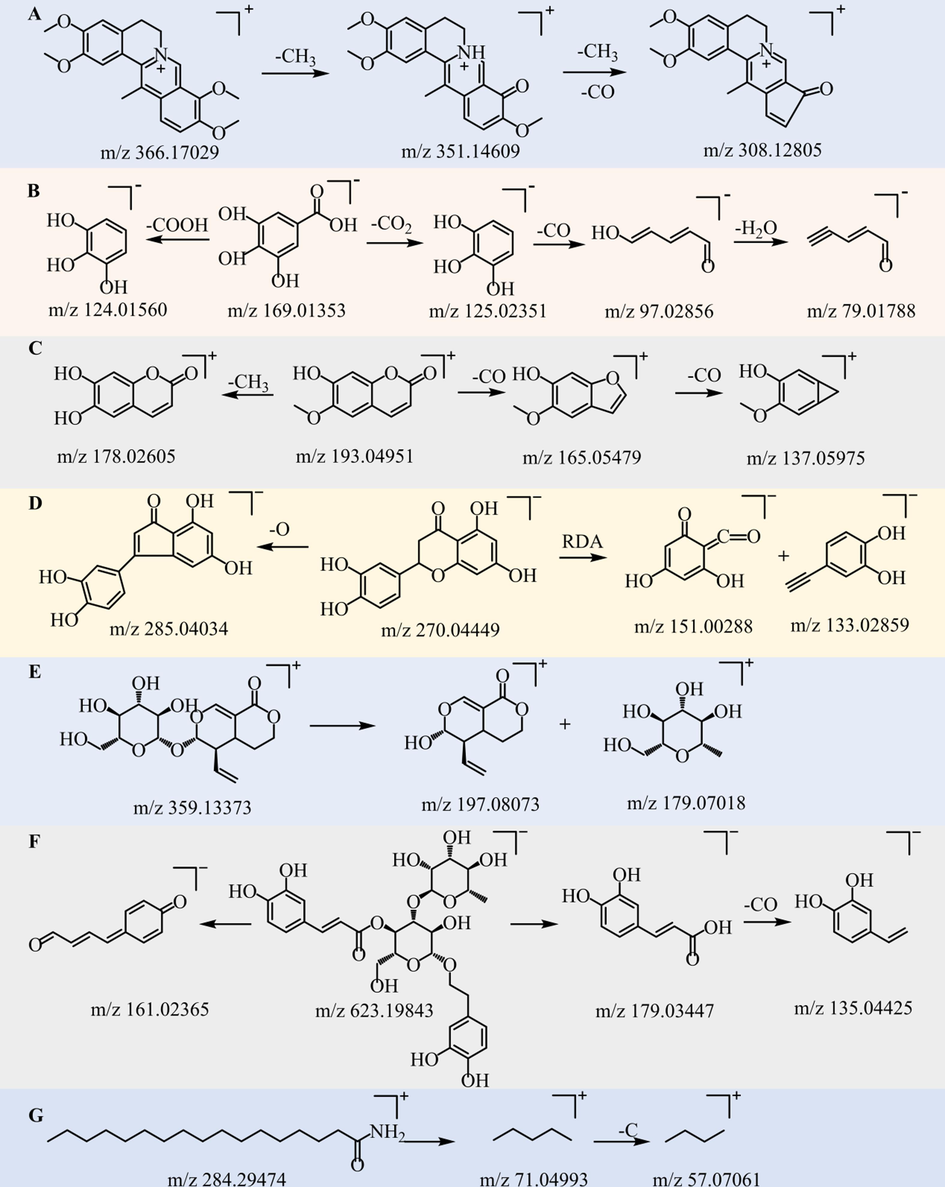
The possible fragmentation pathways of the major components identified from SSP. (A) Dehydrocorydaline. (B) Gallic acid. (C) Scopoletin. (D) Luteolin. (E) Sweroside. (F) Verbascoside. (G) Stearamide.
3.1.2 Organic acids
Eleven organic acids were identified from SSP, including gallic acid, which was accurately identified by comparison with the reference standards. For example, the compound gallic acid (Rt = 2.19 min, C7H6O5), gives a quasi-ion peak which is m/z 169.0136[M-H]-. It loses a COOH molecule to form the fragment ion m/z 124.01560 [M−H−COOH]-. It breaks its C–C bond f to form m/z 125.02351 [M−H−CO2]- and m/z 97.02856 [M−H−CO2−CO]-, respectively, and loses another H2O molecule to form the fragment ion m/z 79.01788 [M−H−CO2−CO−H2O]- (Huang et al., 2017, Ren et al., 2021) (Fig. 3B).
3.1.3 Phenylpropanoids
Nine phenylpropanoids were identified from SSP, among which scopoletin was accurately identified by comparing with the reference standards. For example, scopoletin (Rt = 9.62 min, C10H8O4), gives a quasi-ion peak which m/z is 193.04971[M + H]+. The C-O bond on the side chain of the benzene ring breaks to form fragment ions with m/z 178.02605 [M + H-CH3]+. The benzene ring break formed m/z 165.05479 [M + H-CO]+ and the carbon chain continued to break to form m/z 137.05975 [M + H-2CO]+ (LI et al., 2022b; Wang et al., 2021; Zeng et al., 2015) (Fig. 3C).
3.1.4 Flavonoids
Eighteen flavonoids were identified from SSP, among which mangiferin and quercetin was accurately identified by comparing with the reference standards. For example, luteolin (Rt = 15.06 min, C15H10O6), obtained a quasi-ion peak which m/z is 285.04047[M-H]-. The C-O bond is broken to form m/z 270.04449 [M−H−O]-. After RDA cleavage, m/z 151.00288 and m/z 133.02859 were formed (Xie et al., 2017, Jia et al., 2020, Meng et al., 2020) (Fig. 3D).
3.1.5 Terpenoids
Eight terpenoids were identified from SSP, among which swertiamarin, costunolide and dehydrocostus lactone were accurately identified by comparison with the reference standards. Sweroside (Rt = 7.26 min, C16H22O9), the quasi-ion peak m/z 359.13373 [M + H]+ was obtained. The formation of fragment ions after the glycosidic bond is visible in the secondary mass spectrum m/z 197.08073 and m/z 179.05539 (Yu, 2017) (Fig. 3E).
3.1.6 Phenolics
Six phenolic compounds were identified from SSP, of which ellagic acid was accurately identified by comparison with the reference standards. For example, verbascoside (Rt = 10.36 min, C29H36O15), obtained a quasi-ion peak m/z 623.19843[M-H]-. The glycosidic bond was broken to form m/z 179.03447 and m/z 161.02365, and the loss of a further CO molecule resulted in the formation of m/z 135.04425 (Plaza et al., 2005, Wu et al., 2006, Xie et al., 2017) (Fig. 3F).
3.1.7 Others
Eleven other compounds were identified from SSP. For example, stearamide (Rt = 44.68 min, C18H37NO), obtained a quasi-ion peak of m/z 284.29483 [M + H]+. C–C bond breakage forms m/z 71.04993 and m/z 57.07061(Castillo-Peinado et al., 2019) (Fig. 3G).
3.2 In vivo chemical composition identification
The plasma samples of rats in the dosing and blank groups were analyzed and identified according to the above conditions. The results obtained from the 10-fold dosing group were discarded as they were fewer. A total of 20 chemical components, including 10 prototypes and 10 metabolites, were detected and analyzed in plasma samples from the 20-fold dosing group, net of the chemical components of the blank group. The total positive and negative ion flow diagrams of the samples are shown in Fig. 4, the results of the identification of the main components are shown in Table 2 and the structural formulae of the main components are shown in Fig. 5.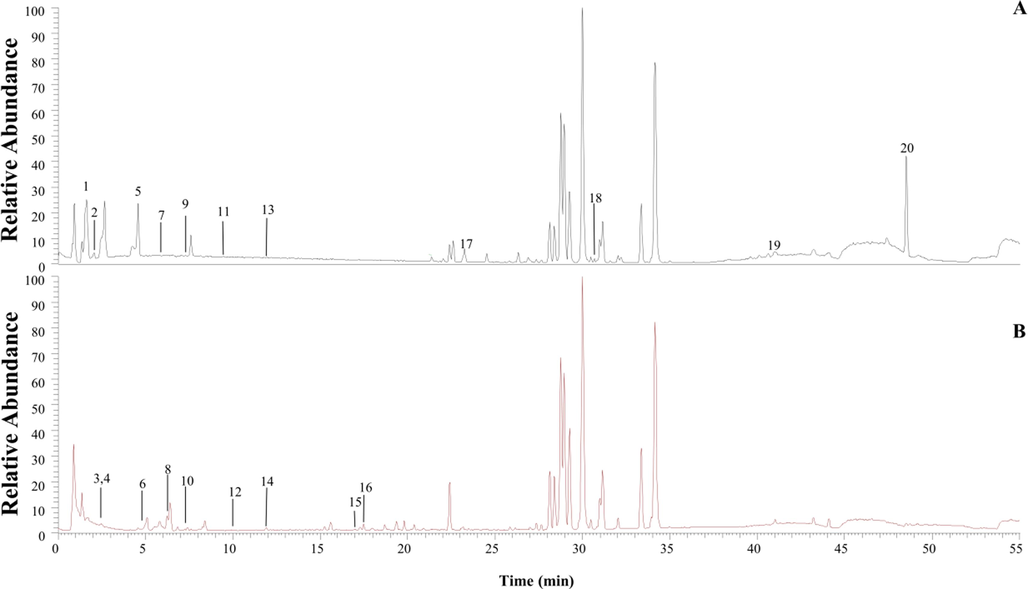
The positive (A) and negative (B) ion chromatogram of the blood-incorporated constituents in SSP.
NO.
tR/min
Molecular formula
Precursor ion
Measured ion
Ion type
MS/MS (m/z)
Component
Classification
1
1.51
C5H11NO2
117.07920
117.07923
[M + H]+
118.08652,72.08141,55.05495
Valine(Virág et al., 2020)
Others
2
2.03
C6H13NO2
131.09
131.09
[M + H]+
132.10202;86.09697;69.07052;55.93526
Leucine (Xiong et al., 2021)
Others
3
2.27
C7H6O5
170.02100
170.02096
[M−H]-
169.01350,125.02351,124.88483,124.01545,97.02839,81.03327,69.03345
Gallic acid (Huang et al., 2017, Ren et al., 2021)
Organic acids
4
2.45
C4H8O3
104.04650
104.04646
[M−H]-
103.03909,59.01276
3-Hydroxybutyric (Zhang et al., 2016a)
Others
5
4.55
C6H11NO
113.08440
113.08438
[M + H]+
114.09161,79.05477,55.01859,55.05495
Caprolactam (Wu et al., 2012)
Others
6
4.89
C8H8O5
184.04
184.04
[M−H]-
183.02948;140.01076;124.01572
Methyl gallate (Gong et al., 2020)
Organic acids
7
6.05
C19H21NO4
327.14720
327.1472
[M + H]+
328.15411,165.07037
6-Acetylmorphine (Ruiz-Colon et al., 2012)
Alkaloids
8
6.21
C16H22O10
420.12700
420.12703
[M + HCOOH-H]-
419.11975,179.05531,161.04503,141.01842
Swertiamarin (Li et al., 2011b)
Terpenoids
9
7.23
C19H18O11
422.08491
422.08491
[M + H]+
423.09116[M + H]+,405.08170[M + H-H2O]+,387.07120,327.04993,303.04974[M + H-C4H8O4]+,273.03925[M + H-C4H8O4-CH2O]+
Mangiferin (Khurana et al., 2017)
Flavonoids
10
7.25
C19H18O11
422.08
422.09
[M−H]-
421.0779;421.07712;331.04596;301.03531
Isomangiferin (Aabideen et al., 2020)
Flavonoids
11
9.63
C21H21NO6
383.13700
383.13701
[M + H]+
384.14429(32),190.08638
Hydrastine (Gupta et al., 2015)
Alkaloids
12
10.05
C14H6O8
302.00640
302.00642
[M−H]-
300.99899,283.99622,257.00824,229.01414,185.02397
Ellagic acid (Yan et al., 2014)
Phenols
13
11.97
C10H9NO2
175.06350
175.06348
[M + H]+
176.07048,130.06517,103.05463
Indole-3-acetic acid (Lin et al., 2015)
Others
14
12.10
C6H5NO3
139.02620
139.02621
[M−H]-
138.01881,108.02070,94.08684
4-Nitrophenol (Hernández et al., 2004)
Phenols
15
16.98
C15H12O5
272.06860
272.06858
[M−H]-
271.06116,227.07135,177.01878,165.01872,151.00288,119.04930,107.01286,93.03355,64.99896
Naringenin (Xu et al., 2020)
Flavonoids
16
17.48
C26H45NO7S
515.29190
515.29187
[M−H]-
514.28442,124.00647,106.97991,80.96426
Taurocholic acid (Gu et al., 2016)
Others
17
23.50
C14H10O6
274.05
274.05
[M + H]+
275.05505;273.04041;258.01697;230.02174;186.03177
Bellidifolin (Wang et al., 2015)
Flavonoids
18
30.69
C16H30O2
254.22
254.22
[M + H]+
255.23184;237.22174;95.08604
Palmitoleic acid (Luo et al., 2022)
Others
19
40.91
C18H35NO
281.27170
281.27165
[M + H]+
282.27896,114.09157,57.07048
Oleamide (Farha and Hatha, 2019)
Others
20
48.52
C22H43NO
337.33450
337.33455
[M + H]+
338.34152,321.31485,303.30420,163.14018,149.13278,135.11661
Erucamide (Dabur and Mittal, 2016)
Others
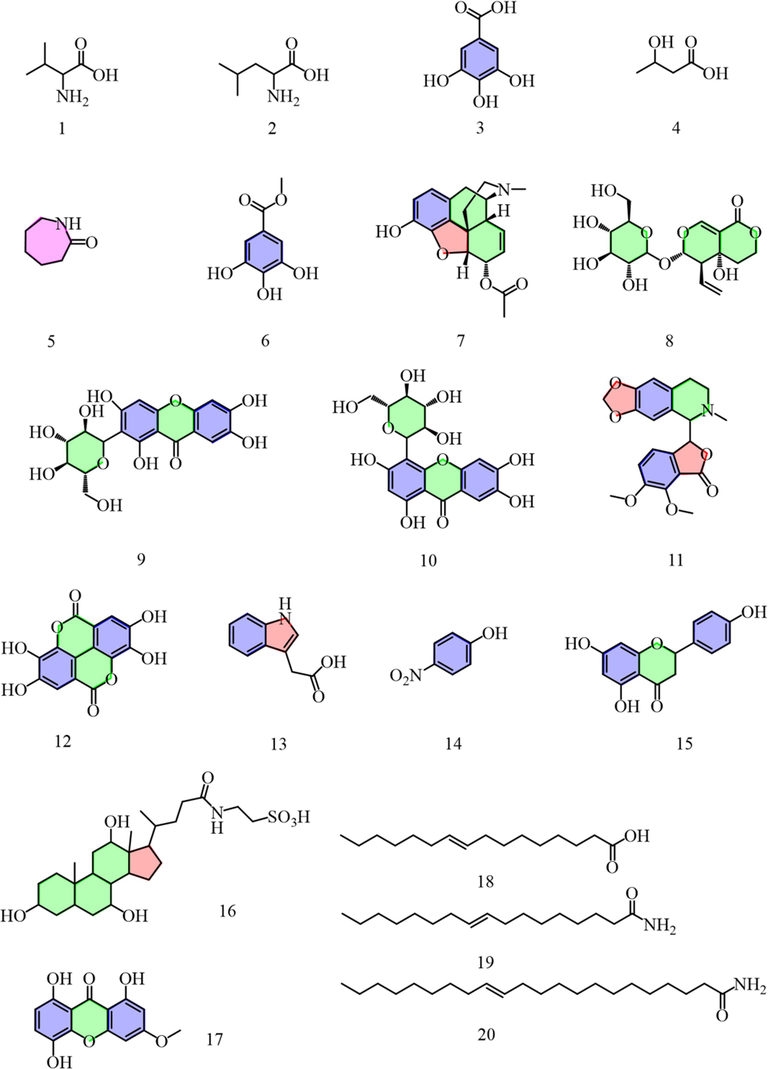
The structural formula of the blood-incorporated constituents in SSP. (1) Valine. (2) Leucine. (3) Gallic acid. (4) 3-Hydroxybutyric. (5) Caprolactam. (6) Methyl gallate. (7) 6-Acetylmorphine. (8) Swertiamarin. (9) Mangiferin. (10) Isomangiferin. (11) Hydrastine. (12) Ellagic acid. (13) Indole-3-acetic acid. (14) 4-Nitrophenol. (15) Naringenin. (16) Taurocholic acid. (17) Bellidifolin. (18) Palmitoleic acid. (19) Oleamide. (20) Erucamide.
3.2.1 Prototype composition identification
A total of ten prototype components were identified from the plasma of rats given SSP by gavage, including gallic acid, methyl gallate, swertiamarin, mangiferin, isomangiferin, ellagic acid, taurocholic acid, bellidifolin, oleamide and erucamide.
Gallic acid (Rt = 2.27 min, C7H6O5), a quasi-ion peak of m/z 169.0136 [M−H]- was obtained. Loss of one carbon dioxide and one carbon monoxide resulted in the formation of m/z 125.02351 [M−H−CO2]- and m/z 97.02839 [M−H−CO2−CO]-, respectively. Loss of two carbon dioxide forms m/z 69.03351 [M−H−CO2−2CO]- (Huang et al., 2017, Ren et al., 2021) (Fig. 6A).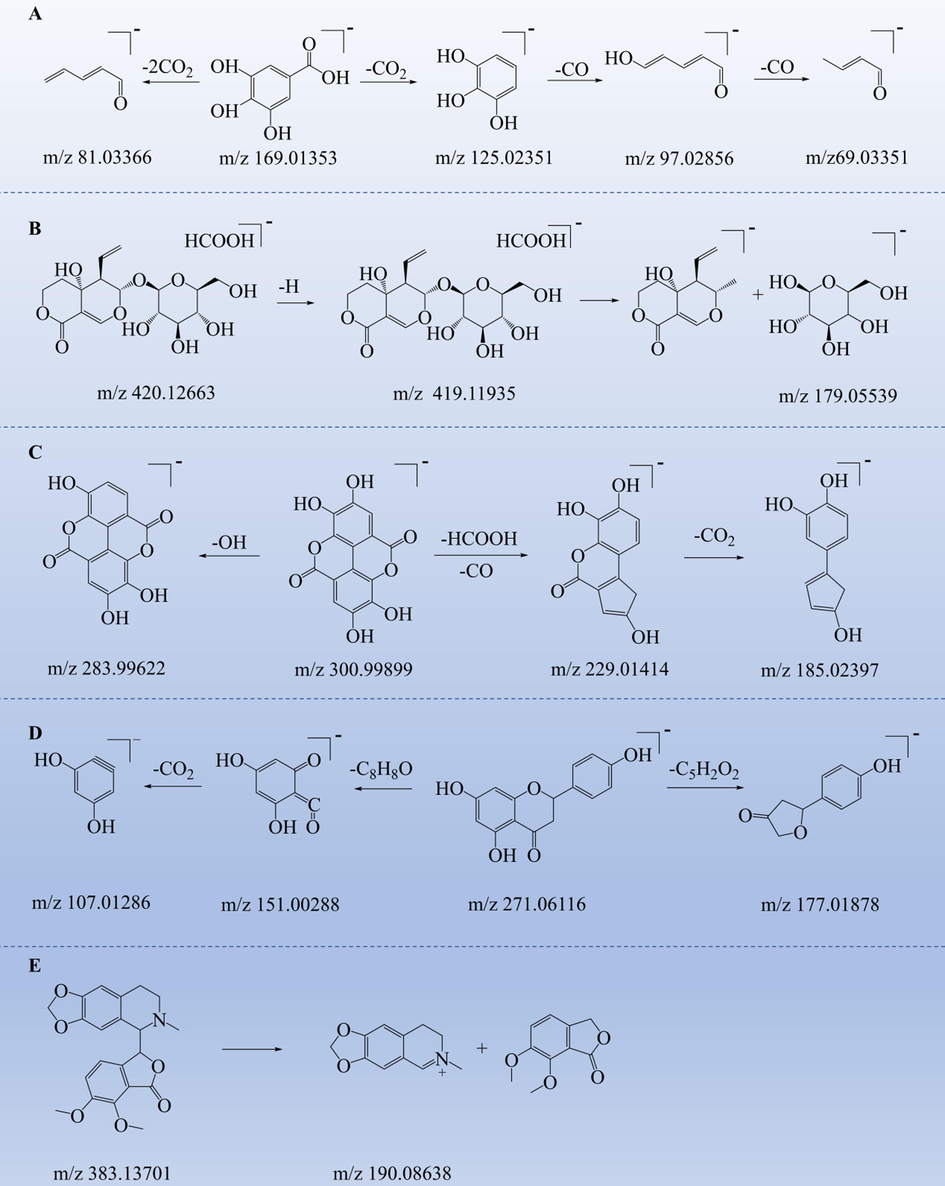
The possible fragmentation pathways of the main blood-incorporated constituents in SSP. (A) Gallic acid. (B) Swertiamarin. (C) Ellagic acid. (D) Naringenin. (E) Hydrastine.
Swertiamarin (Rt = 6.21 min, C16H22O10), a quasi-ion peak of m/z 419.11975 [M + HCOOH-H]- was obtained. Its glycosidic bond is broken to form m/z 179.05331 (Li et al., 2011b) (Fig. 6B).
Ellagic acid (Rt = 10.05 min, C14H6O8), obtained a quasi-ion peak of m/z 300.99899 [M−H]-. The parent ion loses one OH molecule to form m/z 283.99622 [M−H−OH]-; one HCOOH molecule and one CO molecule to form m/z 229.01414 [M−H−CO2−CO]-, and another CO2 molecule to form m/z 185.02397 [M−H−2CO2−CO]- (Yan et al., 2014, Qin et al., 2016) (Fig. 6C).
3.2.2 Metabolite identification
A total of 10 metabolic components, including valine, leucine, 3-hydroxybutyric acid, caprolactam, 6-acetylmorphine, hydrastine, indole-3-acetic acid, p-nitrophenol, palmitoleic acid and naringenin, were identified in the plasma of rats given SSP by gavage.
Naringenin (Rt = 16.98 min, C15H12O5), obtained a quasi-ion peak of m/z 271.06140 [M−H]-. The chemical bond on the C ring is broken to form m/z 177.01878 [M−H−C5H2O2]- and m/z 151.00288 [M−H−C8H8O]-. A CO2 is removed from m/z 151.00288 [M−H−C8H8O]- to form m/z 107.01286 [M−H−C9H8O3]- (Sun et al., 2020) (Fig. 6D).
Hydrastine (Rt = 9.63 min, C21H21NO6), obtained a quasi-ion peak of m/z 384.14429 [M + H]+. The fragment ion m/z 190.08638 [M + H-C10H9O4]+ is seen in the secondary mass spectrum after a chemical bond breakage (Gupta et al., 2015) (Fig. 6E).
3.3 Network pharmacological analysis of 20 blood-incorporated constituents
3.3.1 The targets of SSP blood-incorporated constituents and CH
Employing the SwissTargetPrediction database, our study identified 467 potential targets associated with the blood-incorporated constituents of SSP. These were cross-referenced with a set of 916 targets implicated in CH pathophysiology. This comparative analysis culminated in a subset of 103 common targets, presenting a focused pool of candidates that may mediate SSP's anti-CH effects (Fig. 7A).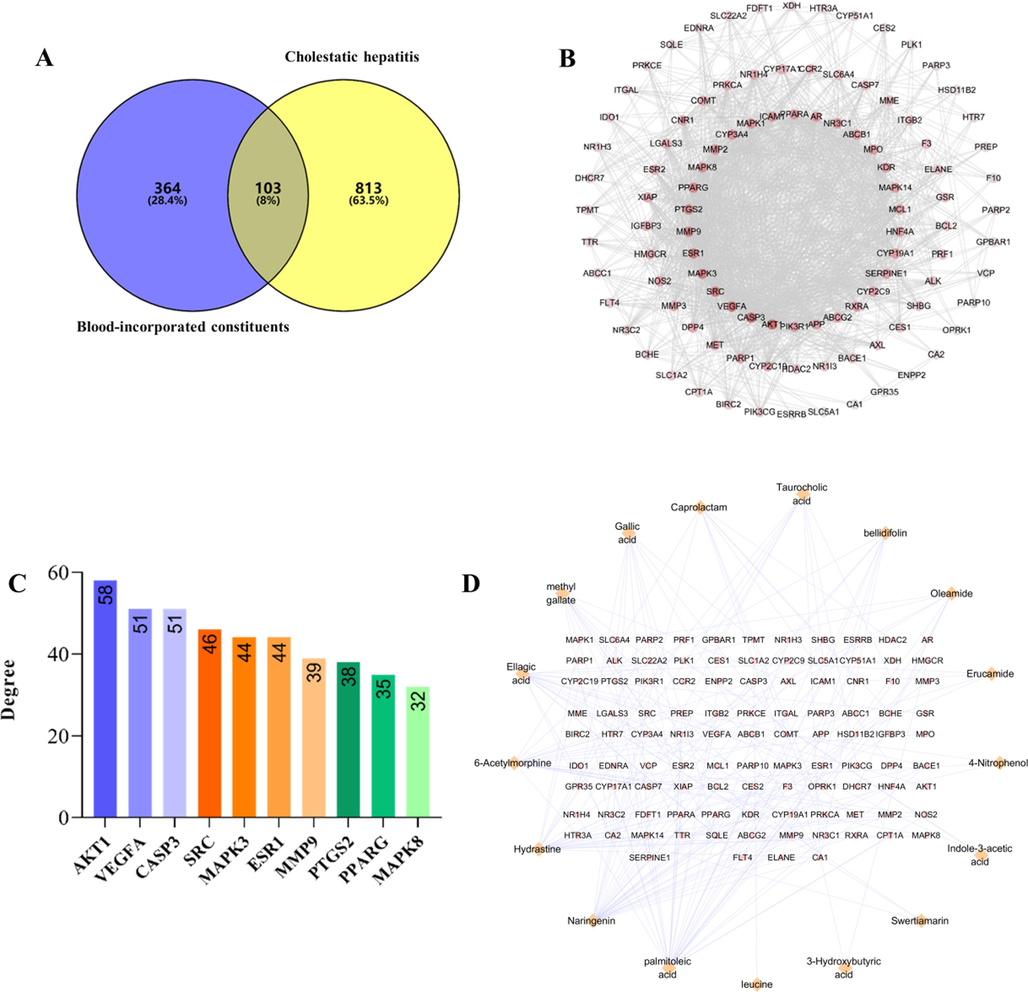
The target PPI network of CH and SSP blood-entering components analysis. (A) Venn plots of the cross-targets between CH and the blood-incorporated constituents in SSP. (B) The PPI network of 103 intersection targets. (C) The degree value of the top 10 intersecting targets. (D) Visualization of the interaction network between SSP blood-incorporated constituents and 103 cross-targets drawn by Cytoscape 3.9.1. The orange square nodes represent blood-incorporated constituents of SSP and the round nodes represent targets for drug component-disease interactions. (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article.)
3.3.2 Protein-protein interaction (PPI) network construction
To elucidate the potential interactions among the 103 overlapping targets identified, we established a PPI network comprising these targets. The network consisted of 103 nodes representing targets and 779 edges denoting interactions, with a median connectivity degree of 15.1, illustrating a complex web of potential inter-target communications (Fig. 7B). Analysis of this PPI network highlighted the top 10 core targets with the highest degree values, namely AKT, VEGFA, CASP3, SRC, MAPK3, ESR1, MMP9, PTGS2, PPARG, and MAPK8, indicating their centrality within the network and their possible significance in SSP's anti-CH action (Fig. 7C). To further explore the relationships between the blood-incorporated constituents of SSP and the intersecting targets, we constructed an interaction network graph. This graphical representation allowed us to discern that, of the 20 components analyzed, three exhibited no direct targets. Conversely, palmitoleic acid, naringenin, and hydrastine emerged as the three constituents with the widest scope of target interactions, hinting at their prominent roles within the therapeutic context of SSP against CH (Fig. 7D).
3.3.3 GO and KEGG pathway enrichment analysis
To delineate the underlying mechanisms of SSP's therapeutic effect on CH, we input the 103 overlapping targets into the DAVID database for GO and KEGG pathway analyses. The GO functional enrichment analysis identified significant associations with 394 biological processes (BP), with top-ranking processes by P-value encompassing responses to drugs, negative regulation of apoptosis, and intracellular receptor signaling pathways. Within cellular components (CC), 56 entities were implicated, with notable structures including receptor complexes, membrane rafts, and the plasma membrane. For molecular functions (MF), 107 functions were highlighted, with steroid binding, enzyme binding, and zinc ion binding surfacing as impactful (Fig. 8A).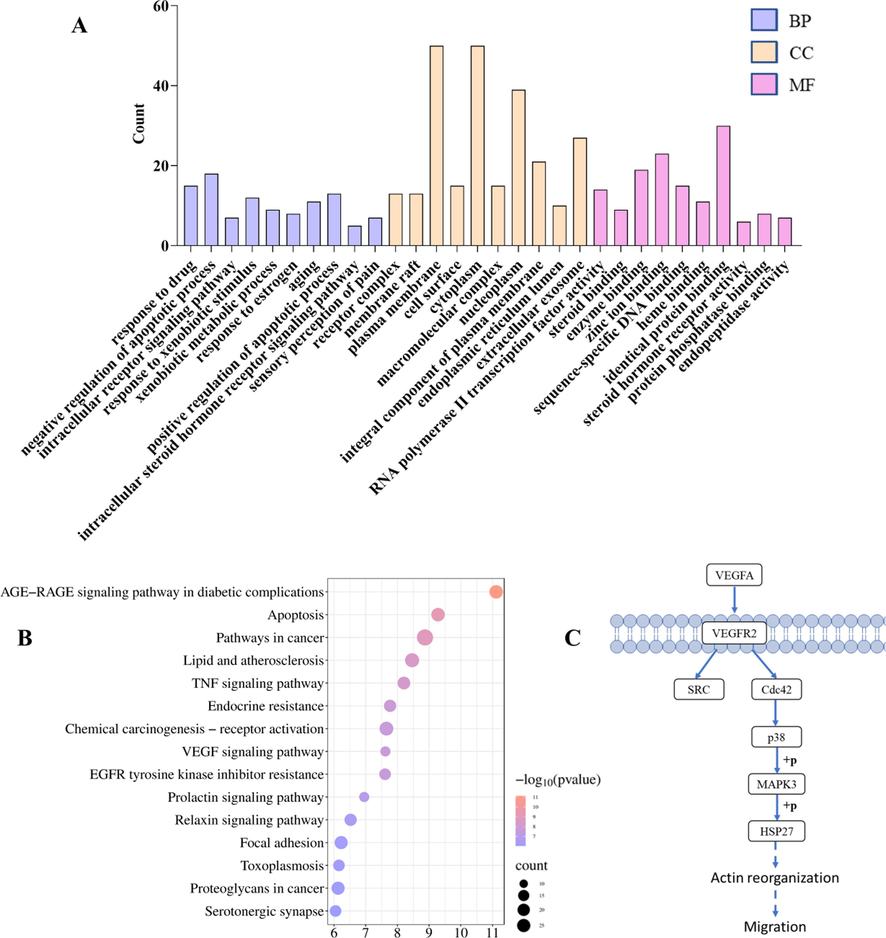
GO and KEGG analysis of the core targets. (A) The top 10 GO terms of hub genes. (B) The top 15 KEGG pathway of hub genes. (C) VEGF signaling pathway.
KEGG pathway enrichment analysis disclosed 142 pathways potentially pertinent to SSP's pharmacodynamics, from which the top 15 pathways were selected based on enrichment significance (Fig. 8B). Some pathways corresponding to the blood-incorporated constituents of SSP stood out, like AGE-RAGE signaling associated with diabetic complications, apoptosis, TNF signaling pathway, and VEGF signaling pathways, etc. Noteworthy pathways not displayed in the top selection included the Rap1 signaling pathway (P = 9.78E-07), fluid shear stress and atherosclerosis (P = 5.05E-06), and the sphingolipid signaling pathway (P = 1.03E-05). Excitingly, we found that top 5 core targets were the most enriched in the VEGF signaling pathway. Therefore, VEGF is considered to be the key pathway for SSP to play its role in the treatment of CH (Fig. 8C).
3.3.4 Molecular docking
Based on the PPI network and pathway enrichment results, we performed molecular docking of the top 5 target proteins in terms of degree (most of which were also significantly enriched in the VEGF signalling pathway) and the 17 blood-incorporated constituents associated with CH, and the detailed results of molecular docking are shown in Fig. 9A. Of all the chemical-target combinations examined, the three pairs exhibiting the lowest binding energies, indicative of the strongest predicted interactions, were: ellagic acid with MAPK3 (Fig. 9B), ellagic acid with VEGFA (Fig. 9C), and hydrastine with SRC (Fig. 9D). In addition to these findings, the docking scores involving six constituents (including swertiamarin, 6-acetylmorphine, hydrastine, ellagic acid, taurocholic acid, and bellidifolin) in relation to the five core targets were uniformly below −6 kcal/mol, underscoring the potential strength and significance of their interactions. It is noteworthy that swertiamarin, ellagic acid, taurocholic acid, and bellidifolin were discerned as prototype components, being those most reliably detected in the bloodstream post SSP administration. These analyses collectively suggest that these four constituents may play an integral role in mediating SSP's therapeutic efficacy against CH, signifying the potential for their advancement as focal points for further study and drug development efforts against this condition.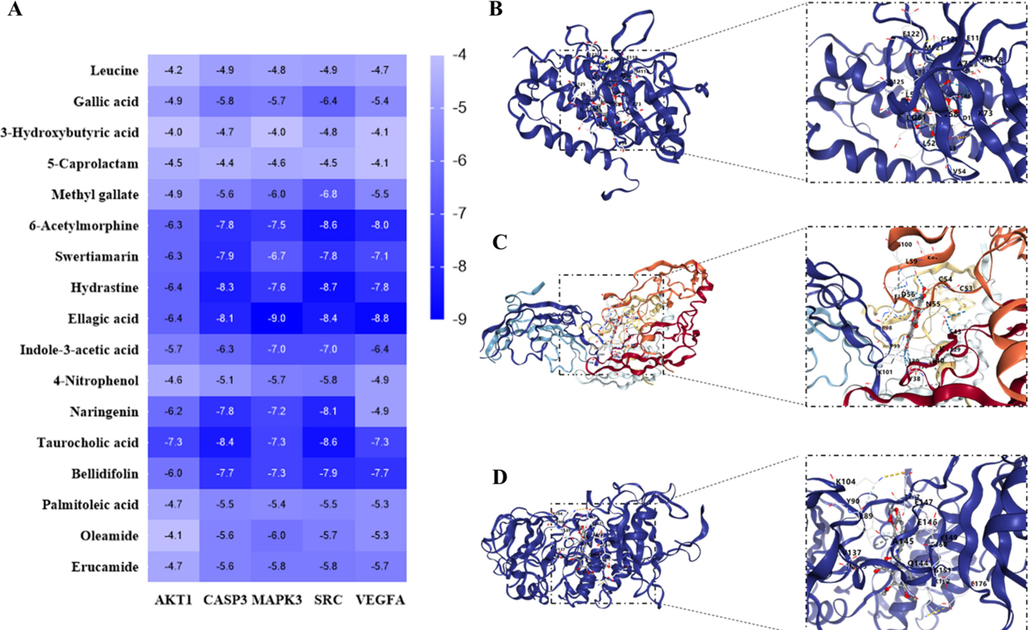
Molecular docking results for the core targets. (A) The heat map of docking results between the core targets (including VEGFA, SRC, MAPK3, CASP3, and AKT1) and 17 blood-incorporated constituents. (B) Ellagic acid-MAPK3 (7NRB). (C) Ellagic acid-VEGFA (3QTK). (D) Hydrastine-SRC (1FMK).
3.3.5 Biological validation
To evaluate the therapeutic potential of SSP for combating CH, our study commenced with histological investigation through H&E staining. In normal control rats, hepatocytes demonstrated a radiating arrangement around central veins, exhibiting typical morphology and intact architecture without prominent histopathological deviations. In contrast, rats from the model control group, depicting a similar radial hepatocyte organization, exhibited discernible pathologic alterations, including hepatocyte vacuolation, infiltration of inflammatory cells within confluent regions, necrotic sites within hepatic tissues accompanied by local congestive or hemorrhagic features. Upon treatment with SSP, we observed a restoration towards normalcy in the arrangement of hepatocytes around central veins across various dosage groups. There was noticeable amelioration in steatotic vacuolization and reduced inflammatory exudation (Fig. 10A). Based on the results of network pharmacological analysis, we obtained that the top five core targets all have numerous studies showing that their aberrant expression or abnormally elevated activity may lead to different degrees of liver pathology, so we selected the top five proteins (AKT1, VEGFA, CASP3, SRC, MAPK3) that are closely related to the SSP anti-CH and verified the targets using immunohistochemistry methods. Notably, most of these Top5 targets are enriched in the VEGF signalling pathway, thus, we examined the expression of these five target proteins using immunohistochemistry, and the immunohistological quantifications revealed elevated expressions of AKT1, VEGFA, CASP3, SRC, and MAPK3 in the model group when compared to normal controls, with significant differences (p < 0.001). Compared with the model group, the UDCA, as well as low, medium, and high doses of SSP, resulted in a dose-dependent attenuation in the expression levels of these proteins when juxtaposed against the model control. These alterations were statistically significant (p < 0.05) (Fig. 10B). These findings corroborate SSP's putative hepatoprotective effect and pave the way for a deeper understanding of SSP's mechanistic role.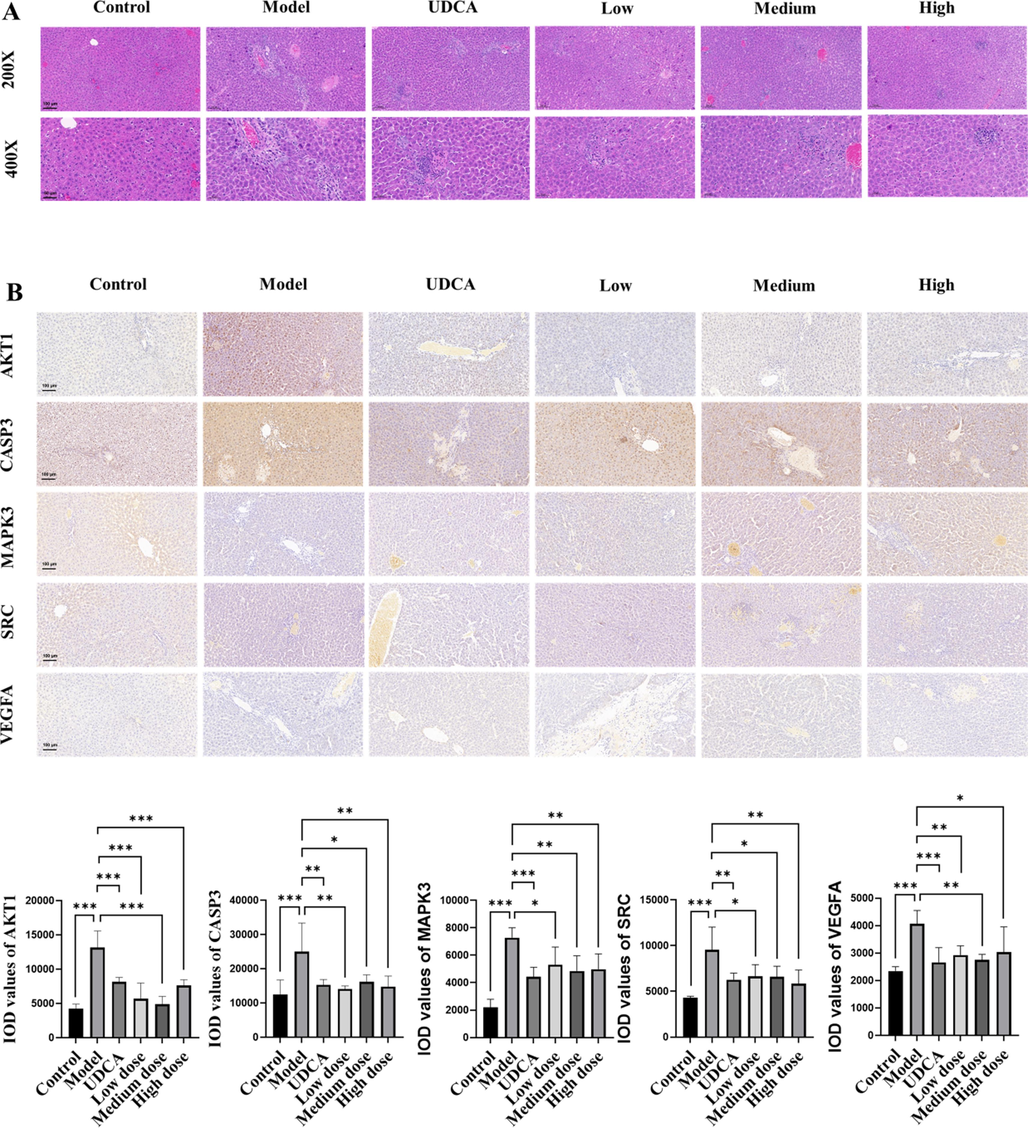
Pathological effect of SSP on CH model rats and verification of core targets. (A) The histopathology of liver tissue was observed by HE staining. Magnification (200×; 400 × ). (B) The changes in the core targets (including VEGFA, SRC, MAPK3, CASP3, and AKT1) of the SSP were detected by immunohistochemical method. Magnification (200 × ). Data were expressed as the mean ± SD (n = 3), *p < 0.05, **p < 0.01, ***p < 0.001.
4 Discussion
This research aimed to delineate the active chemical components and their mechanistic roles underpinning the therapeutic effects of the Tibetan medicine SSP in the management of CH. Through UHPLC-Q-Exactive Orbitrap/MS, we examined both the composition of SSP and the plasma constituents in SSP-dosed rats. We identified 80 chemical entities in vitro and 20 that were systemically assimilated, inclusive of 10 primary compounds and 10 derivative metabolites. Notables among these are gallic acid, swertiamarin, ellagic acid, and bellidifolin, each capable of targeting multiple biological sites. Gallic acid displays a spectrum of bioactivities, including anti-inflammatory, antiviral, and liver-protective effects (Huang et al., 2023). Specific to the liver, it targets and induces apoptosis in hepatic stellate cells and inhibits growth in the SMMC-7721 hepatocellular carcinoma cell line (Sun, 2016, Zheng et al., 2016). Swertiamarin has been reported to substantially abate both serum aspartate aminotransferase (AST) and interleukin-6 (IL-6) levels in models of surgically induced liver injury, and decrease serum alanine aminotransferase (ALT) and total bilirubin (TBIL) in chemical liver injury scenarios (Chen et al., 2016; Tian et al., 2014b; Zhang et al., 2015). Ellagic acid has been observed to reverse acute liver injury markers such as ALT and AST in mice, elevate the expression of VEGF and its receptor VEGFR, and boost CASP3 activity, underscoring its hepatoprotective capacity across both rodent species (Chen et al., 2023; Zhang et al., 2016b; Zhao et al., 2021; Long et al., 2017). Collectively, our findings affirm the medicinal significance of these compounds, ensuring their pivotal status in the exploration of SSP’s mode of action in CH.
Network pharmacology analyses divulged 103 intersecting targets between components absorbed into the bloodstream following SSP administration and those implicated in CH, highlighting a spectrum of potential molecular sites for SSP's action against the disease. Subsequent establishment of a PPI network facilitated the distillation of five central targets of interest-AKT1, VEGFA, CASP3, SRC, and MAPK3-for therapeutic intervention. Kupffer cells (KCs) can instigate the synthesis and release of transforming growth factor-beta1 (TGF-β1), thereby contributing to hepatic inflammation, a response that is augmented in mice subjected to CCl4-induced liver fibrosis mice (Ghavami et al., 2015, Nie et al., 2019, Vaidya et al., 2019). Research suggests that the inhibition of AKT1 may curtail TGF-β1 secretion by KCs, offering a therapeutic lever against liver inflammation (Wu et al., 2020). Meanwhile, VEGFA serves as a trigger for human hepatic stellate cell activation through VEGF-VEGFR pathways, whose attenuation has been documented to lessen the severity of NAFLD progression to hepatocellular carcinoma in mice with hepatocyte-specific VEGFA deletion (Vaidya et al., 2019). Overexpression of CASP3, with ensuing PARP substrate cleavage, precipitates DNA disintegration and cellular apoptosis-a common occurrence across various liver pathologies including inflammation, fibrosis, and cancer (Hengartner, 2000, Decker and Muller, 2002, Osna et al., 2017, Ma et al., 2021). Disparities in SRC expression are influential in liver functionality and bear prognostic significance in hepatocellular carcinoma trajectories (Chatzizacharias et al., 2012, Reinehr et al., 2013, Mantonakis et al., 2017). Additionally, aberrations in MAPK3′s expression or activity may instigate cellular apoptosis or proliferation, potentially influencing the onset, progression, or metastatic spread of diverse cancers, liver cancer included (Taherkhani et al., 2023). In the context of SSP's therapeutic targeting for CH, the elucidated PPI network underscores the five core targets (AKT1, VEGFA, CASP3, SRC, MAPK3) as vital nodes, potentially critical in the effective management of cholestatic hepatitis.
GO and KEGG pathway enrichment analyses were deployed to unravel the pharmacological mechanisms through which SSP counteract CH. According to GO insights, the implicated targets were predominantly correlated with biological processes like drug response, negative regulation of apoptosis, intracellular receptor signaling pathways, and positive regulation of apoptosis. KEGG enrichment suggested apoptosis and the VEGF signaling pathway as central to SSP's CH remediation. VEGF's pivotal role in vascular formation and endothelial gene expression modulation substantiates its influence on endothelial dysfunction and hepatic fibrosis development (Apte et al., 2019, Ntellas et al., 2020). Additionally, hepatocyte-derived VEGFA has been implicated in expediting fibrosis and hepatocarcinogenesis by HSC activation during NAFLD progression (Shen, 2021).
To further decipher SSP's anti-CH modus operandi, molecular docking assessed the interaction potential between 20 systemic components-such as gallic acid, swertiamarin, ellagic acid, and bellidifolin-and five core targets (AKT1, VEGFA, CASP3, SRC, MAPK3). The analysis disclosed affirmative binding affinities, with ellagic acid demonstrating especially stable docking to MAPK3 as evidenced by the lowest binding energy. Complementarily, the therapeutic benefits of SSP in CH treatment, and its impact on the expression of the quintet of core proteins, were substantiated through HE staining and immunohistochemical approaches. HE staining results signified SSP’s efficacy in mitigating inflammatory exudation and hemorrhage within hepatic tissue cells. Immunohistochemistry revealed upregulated expression of the core proteins post-ANIT gavage, while SSP treatment inversely modulated their expression. This indicates SSP’s regulatory effect on the VEGF signaling pathway through these proteins, thus mediating its therapeutic action in CH management.
5 Conclusions
Collectively, in this study, we firstly characterized the in vivo and in vitro chemical composition of Tibetan medicine SSP using UHPLC-Q-Exactive Orbitrap/MS technique, then investigated the blood-incorporated constituents through network pharmacology and molecular docking, and finally performed preliminary validation of key targets through in vivo animal experiments. We found that the main active components of SSP exerting its anti-CH effects include swertiamarin, ellagic acid, taurocholic acid and bellidifolin. These components may exert their anti-CH effects through modulation of key targets such as AKT1, VEGFA, CASP3, SRC and MAPK3. It is worth noting that our current study still has some limitations and we have only assessed the anti-CH efficacy of SSP as a whole. However, whether the main active ingredients of SSP alone are also effective in the treatment of CH, as well as their direct targets and mechanisms are still key research directions that need to be strengthened in the future. Furthermore, in our investigation, the anticipated dose-dependent inhibition across varying concentrations of SSP was not observed. This counterintuitive biological response may be explained by several factors intrinsic to the multifaceted nature of herbal formulations. Pharmacokinetic complexities such as non-linear absorption, distribution, metabolism, or excretion of SSP constituents could contribute to this phenomenon. Moreover, we hypothesize the presence of a ceiling effect, wherein maximum inhibitory efficacy is reached at lower dosages, beyond which no additional suppression of target proteins is discernible. Additionally, adaptive cellular feedback mechanisms might attenuate the inhibitory effects at higher concentrations. Understanding the interplay between these components in SSP and their cumulative biological impact necessitates further elucidation. To address this complexity, future studies will aim to dissect the pharmacological intricacies of SSP, potentially shedding light on these unexpected results and refining our comprehension of its therapeutic mechanisms in CH treatment. As a conclusion, it is hoped that this experiment will provide a reference for subsequent studies on the treatment of CH with the Tibetan medicine SSP.
CRediT authorship contribution statement
Jing Qin: Methodology, Investigation, Writing – original draft. Gelin Xiang: Data curation, Investigation, Visualization. Huimin Gao: Data curation, Investigation, Visualization. Xianli Meng: Supervision, Writing – review & editing, Resources. Shaohui Wang: Conceptualization, Supervision, Writing – review & editing, Resources. Yi Zhang: Conceptualization, Supervision, Writing – review & editing, Resources.
Acknowledgements
This study was funded by the National Natural Science Foundation of China (81973573); the Science and Technology Department of Sichuan Province (2020YFQ0032); the Key R&D and Transformation Program of the Science and Technology Department of Qinghai Province (2020-SF-C33).
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Anti-obesity attributes; UHPLC-QTOF-MS/MS-based metabolite profiling and molecular docking insights of taraxacum officinale. Molecules. 2020;25
- [CrossRef] [Google Scholar]
- Antioxidant and cytoprotective properties of loganic acid isolated from seeds of Strychnos potatorum L. against heavy metal induced toxicity in PBMC model. Drug Chem. Toxicol.. 2022;45:239-249.
- [CrossRef] [Google Scholar]
- Abu Bakar, F. I., M. F. Abu Bakar, N. Abdullah, et al., 2020. Optimization of Extraction Conditions of Phytochemical Compounds and Anti-Gout Activity of Euphorbia hirta L. (Ara Tanah) Using Response Surface Methodology and Liquid Chromatography-Mass Spectrometry (LC-MS) Analysis. Evid Based Complement Alternat Med. 2020, 4501261. 10.1155/2020/4501261.
- VEGF in signaling and disease: Beyond discovery and development. Cell. 2019;176:1248-1264.
- [CrossRef] [Google Scholar]
- UHPLC-MS/MS determination and pharmacokinetic study of plantamajoside in rat plasma after oral administration of single plantamajoside and Plantago asiatica extract. Biomed. Chromatogr.. 2017;31
- [CrossRef] [Google Scholar]
- Comment on the published data concerning the identification of biochanin A and prunetin by LC/ESI-MS. Talanta. 2020;211:120733
- [CrossRef] [Google Scholar]
- A UPLC-MS/MS application for profiling of intermediary energy metabolites in microdialysis samples–a method for high-throughput. J. Pharm. Biomed. Anal.. 2010;53:983-990.
- [CrossRef] [Google Scholar]
- Brito, J. A. G. d., L. d. S. Pinto, C. F. Chaves, et al., 2021. Chemophenetic Significance of Anomalocalyx uleanus Metabolites are Revealed by Dereplication Using Molecular Networking Tools. Molecules. 26, 10.3390/molecules26040925.
- Quantitative measurement of betaine and free choline in plasma, cereals and cereal products by isotope dilution LC-MS/MS. J. Agric. Food Chem.. 2010;58:2055-2061.
- [CrossRef] [Google Scholar]
- Determination of primary fatty acid amides in different biological fluids by LC-MS/MS in MRM mode with synthetic deuterated standards: Influence of biofluid matrix on sample preparation. Talanta. 2019;193:29-36.
- [CrossRef] [Google Scholar]
- Clinical significance of Src expression and activity in human neoplasia. Histol. Histopathol.. 2012;27:677-692.
- [CrossRef] [Google Scholar]
- Simultaneous identification of low-molecular weight phenolic and nitrogen compounds in craft beers by HPLC-ESI-MS/MS. Food Chem.. 2019;286:113-122.
- [CrossRef] [Google Scholar]
- The pharmacology research progress of ellagic acid online first. Modernization Trad. Chinese Med. Mater. Med. World Sci. Technol.. 2023;1–7
- [CrossRef] [Google Scholar]
- Research progress on pharmacological activities of swertiamarin. Drugs & Clinic. 2016;31:1684-1688.
- [CrossRef] [Google Scholar]
- LC-MS/MS determination and urinary excretion study of seven alkaloids in healthy Chinese volunteers after oral administration of Shuanghua Baihe tablets. J. Pharm. Biomed. Anal.. 2016;118:89-95.
- [CrossRef] [Google Scholar]
- LC-MS/MS based identification of piperine production by endophytic Mycosphaerella sp. PF13 from Piper nigrum. Appl. Biochem. Biotechnol.. 2014;173:30-35.
- [CrossRef] [Google Scholar]
- Simultaneous determination of chlorogenic acid isomers and metabolites in rat plasma using LC-MS/MS and its application to A pharmacokinetic study following oral administration of stauntonia hexaphylla leaf extract (YRA-1909) to rats. Pharmaceutics. 2018;10
- [CrossRef] [Google Scholar]
- Occurrence of bufotenin in the Osteocephalus genus (Anura: Hylidae) Toxicon. 2005;46:371-375.
- [CrossRef] [Google Scholar]
- Detection and qualitative analysis of fatty acid amides in the urine of alcoholics using HPLC-QTOF-MS. Alcohol. 2016;52:71-78.
- [CrossRef] [Google Scholar]
- Modulatory effect of Senecio brasiliensis (Spreng) Less. in a murine model of inflammation induced by carrageenan into the pleural cavity. J. Ethnopharmacol.. 2015;168:373-379.
- [CrossRef] [Google Scholar]
- Modulating poly (ADP-ribose) polymerase activity: potential for the prevention and therapy of pathogenic situations involving DNA damage and oxidative stress. Curr. Pharm. Biotechnol.. 2002;3:275-283.
- [CrossRef] [Google Scholar]
- An UHPLC-MS/MS method for simultaneous determination of quercetin 3- O -rutinoside, kaempferol 3- O -rutinoside, isorhamnetin 3- O -rutinoside, bilobalide and ligustrazine in rat plasma, and its application to pharmacokinetic study of Xingxiong injection. Chin. J. Nat. Med.. 2017;15:710-720.
- [CrossRef] [Google Scholar]
- Simultaneous determination of selected catechins and pyrogallol in deer intoxications by HPLC-MS/MS. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci.. 2021;1180:122886
- [CrossRef] [Google Scholar]
- Expert consensus on the diagnosis and treatment of cholestatic liver disease: an update in 5. Journal of Clinical Hepatology. 31, 1563-1574. 10.3969/j.issn.1001-5256.5.10.002.
- LC/MS/MS characterization of phenolic constituents in dried plums. J. Agric. Food Chem.. 2002;50:3579-3585.
- [CrossRef] [Google Scholar]
- Experimental research progress of traditional Chinese medicine in the prevention and treatment of cholestatic liver disease. Chinese J. Integrated Trad. Western Med. Liver Diseases. 2020;30:375-377.
- [CrossRef] [Google Scholar]
- Bioprospecting potential and secondary metabolite profile of a novel sediment-derived fungus Penicillium sp. ArCSPf from continental slope of Eastern Arabian Sea. Mycology. 2019;10:109-117.
- [CrossRef] [Google Scholar]
- Analysis by HPLC and LC/MS of pungent piperamides in commercial black, white, green, and red whole and ground peppercorns. J. Agric. Food Chem.. 2008;56:3028-3036.
- [CrossRef] [Google Scholar]
- Autophagy is a regulator of TGF-β1-induced fibrogenesis in primary human atrial myofibroblasts. Cell Death Dis.. 2015;6:e1696.
- [Google Scholar]
- Comparison of LC-MS/MS and enzymatic methods for the determination of total choline and total carnitine in infant formula and milk products. J. AOAC Int.. 2020;103:1293-1300.
- [CrossRef] [Google Scholar]
- Simultaneous determination of gallic acid, methyl gallate, and 1,3,6-tri-O-galloyl-beta-d-glucose from Turkish galls in rat plasma using liquid chromatography-tandem mass spectrometry and its application to pharmacokinetics study. Biomed. Chromatogr.. 2020;34:e4916.
- [CrossRef] [Google Scholar]
- Simultaneous quantification of chlorogenic acid and taurocholic acid in human plasma by LC-MS/MS and its application to a pharmacokinetic study after oral administration of Shuanghua Baihe tablets. Chin. J. Nat. Med.. 2016;14:313-320.
- [CrossRef] [Google Scholar]
- Identification of metabolites of the cardioprotective alkaloid dehydrocorydaline in rat plasma and bile by liquid chromatography coupled with triple quadrupole linear ion trap mass spectrometry. Molecules. 2017;22
- [CrossRef] [Google Scholar]
- Absorption, metabolism, and pharmacokinetics profiles of norathyriol, an aglycone of mangiferin, in rats by HPLC-MS/MS. J. Agric. Food Chem.. 2018;66:12227-12235.
- [CrossRef] [Google Scholar]
- Hydrastine pharmacokinetics and metabolism after a single oral dose of goldenseal (Hydrastis canadensis) to humans. Drug Metab. Dispos.. 2015;43:534-552.
- [CrossRef] [Google Scholar]
- New progress of pathogenesis and therapeutic agents of hepatic cholestasi. Chinese J. Gastroenterol. Hepatol.. 2016;25:584-588.
- [CrossRef] [Google Scholar]
- Determination of swertianolin in rat plasma by LC-MS/MS and its application to a pharmacokinetic study. Biomed. Chromatogr.. 2014;28:1418-1422.
- [CrossRef] [Google Scholar]
- An estimation of the exposure to organophosphorus pesticides through the simultaneous determination of their main metabolites in urine by liquid chromatography-tandem mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci.. 2004;808:229-239.
- [CrossRef] [Google Scholar]
- Simultaneous determination of paeoniflorin, albiflorin, ferulic acid, tetrahydropalmatine, protopine, typhaneoside, senkyunolide I in Beagle dogs plasma by UPLC-MS/MS and its application to a pharmacokinetic study after Oral Administration of Shaofu Zhuyu Decoction. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci.. 2014;962:75-81.
- [CrossRef] [Google Scholar]
- Research progress on penthorum chinense pursh in common liver diseases. Modernization Trad. Chinese Med. Mater. Med. World Sci. Technol.. 2023;1–6
- [CrossRef] [Google Scholar]
- A UPLC-MS/MS method for simultaneous determination of free and total forms of a phenolic acid and two flavonoids in rat plasma and its application to comparative pharmacokinetic studies of polygonum capitatum extract in rats. Molecules. 2017;22
- [CrossRef] [Google Scholar]
- Biotransformation and tissue distribution of protopine and allocryptopine and effects of Plume Poppy Total Alkaloid on liver drug-metabolizing enzymes. Sci. Rep.. 2018;8:537.
- [CrossRef] [Google Scholar]
- Identification and quantification of phenolic compounds in Vitex negundo L. var. cannabifolia (Siebold et Zucc.) Hand.-Mazz. using liquid chromatography combined with quadrupole time-of-flight and triple quadrupole mass spectrometers. J. Pharm. Biomed. Anal.. 2015;108:11-20.
- [CrossRef] [Google Scholar]
- Pharmacokinetic study of thirteen ingredients after the oral administration of flos chrysanthemi extract in rats by UPLC-MS/MS. Biomed. Res. Int.. 2020;2020:8420409.
- [CrossRef] [Google Scholar]
- Elucidation of stress-induced degradation products of mangiferin: Method development and validation. Biomed. Chromatogr.. 2017;31
- [CrossRef] [Google Scholar]
- 9-oxo-10(E),12(E)-Octadecadienoic acid derived from tomato is a potent PPAR alpha agonist to decrease triglyceride accumulation in mouse primary hepatocytes. Mol. Nutr. Food Res.. 2011;55:585-593.
- [CrossRef] [Google Scholar]
- LC-ESI/MS determination of xanthone and secoiridoid glycosides from in vitro regenerated and in vivo Swertia chirayita. Physiol. Mol. Biol. Plants. 2015;21:51-60.
- [CrossRef] [Google Scholar]
- UPLC/MS/MS method for quantification and cytotoxic activity of sesquiterpene lactones isolated from Saussurea lappa. J. Ethnopharmacol.. 2014;155:1393-1397.
- [CrossRef] [Google Scholar]
- Quantitative investigation of trigonelline, nicotinic acid, and nicotinamide in foods, urine, and plasma by means of LC-MS/MS and stable isotope dilution analysis. J. Agric. Food Chem.. 2008;56:11114-11121.
- [CrossRef] [Google Scholar]
- Li, P., S. Liu, Q. Liu, et al., 2019. Screening of acetylcholinesterase inhibitors and characterizing of phytochemical constituents from Dichocarpum auriculatum (Franch.) W.T. Wang & P. K. Hsiao through UPLC-MS combined with an acetylcholinesterase inhibition assay in vitro. J Ethnopharmacol. 245, 112185. 10.1016/j.jep.2019.112185.
- Dietary flavone from the Tetrastigma hemsleyanum vine triggers human lung adenocarcinoma apoptosis via autophagy. Food Funct.. 2020;11:9776-9788.
- [CrossRef] [Google Scholar]
- Study on protective mechanism of Tibetan medicine Ershiwuwei Songshi Pills on cholestatic liver injury in rats based on FXR signaling pathway. China J. Chinese Mater. Med.. 2020;45:5273-5279.
- [CrossRef] [Google Scholar]
- Analysis on medication regularities of traditional Tibetan medicine treating hepatitis based on data mining. Chinese J. Ethnomed. Ethnopharm.. 2018;27:4-7.
- [CrossRef] [Google Scholar]
- Chemical constituents of vinegar-processed Kansui radix. Food and Drug. 2011;13:183-185.
- [CrossRef] [Google Scholar]
- Development and validation of a LC-ESI-MS/MS method for the determination of swertiamarin in rat plasma and its application in pharmacokinetics. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci.. 2011;879:1653-1658.
- [CrossRef] [Google Scholar]
- Comparative metabolism study on chlorogenic acid, cryptochlorogenic acid and neochlorogenic acid using UHPLC-Q-TOF MS coupled with network pharmacology. Chin. J. Nat. Med.. 2021;19:212-224.
- [CrossRef] [Google Scholar]
- Network pharmacology prediction and molecular docking-based strategy to explore the potential mechanism of Huanglian Jiedu Decoction against sepsis. Comput. Biol. Med.. 2022;144:105389
- [CrossRef] [Google Scholar]
- Identification and attribution of chemical constituents from Erlong Capsules based on UPLC-Q-TOF-MS/MS. Chinese Traditional Patent Medicine. 2022;44:132-141.
- [CrossRef] [Google Scholar]
- Simultaneous determination and pharmacokinetics of danshensu, protocatechuic aldehyde, 4-hydroxy-3-methyloxyphenyl lactic acid and protocatechuic acid in human plasma by LC-MS/MS after oral administration of Compound Danshen Dripping Pills. J. Pharm. Biomed. Anal.. 2017;145:860-864.
- [CrossRef] [Google Scholar]
- Systematic profiling of indole-3-acetic acid biosynthesis in bacteria using LC–MS/MS. J. Chromatogr. B. 2015;988:53-58.
- [CrossRef] [Google Scholar]
- Severe cholestatic hepatitis secondary to SARS-CoV-2. ACG Case Rep. J.. 2022;9:e00753.
- [CrossRef] [Google Scholar]
- Long, Y., J. Luo, M. Hu, et al., 2017. Protective effect of ellagic acid on acute liver injury induced by CCl4 in mice and its mechanism. Journal of Jilin University(Medicine Edition). 10.13481/j.1671-587x.20170321.
- Determination and pharmacokinetic study of skimmin by UHPLC-MS/MS in rat plasma. J. Pharm. Biomed. Anal.. 2020;179:112969
- [CrossRef] [Google Scholar]
- Analysis of chemical composition in different parts of fritillaria thunbergii using UHPLC-QTOF-MS/MS. Chinese J. Mod. Appl. Pharm.. 2022;39:1984-1991.
- [CrossRef] [Google Scholar]
- Ursolic acid reduces hepatocellular apoptosis and alleviates alcohol-induced liver injury via irreversible inhibition of CASP3 in vivo. Acta Pharmacol. Sin.. 2021;42:1101-1110.
- [CrossRef] [Google Scholar]
- FAK, Src and p-Paxillin expression is decreased in liver metastasis of colorectal carcinoma patients. J. Buon.. 2017;22:1097-1106.
- [Google Scholar]
- Rapid identification and analysis of chemical components in heartwood of Dalbergia cochinchinensis by UPLC-Q-TOF-MS/MS. Chinese J. Exp. Trad. Med. Formulae. 2020;26:143-156.
- [CrossRef] [Google Scholar]
- Qualitative and quantitative analysis of Ukrainian Iris species: A fresh look on their antioxidant content and biological activities. Molecules. 2020;25
- [CrossRef] [Google Scholar]
- Akt1 regulates pulmonary fibrosis via modulating IL-13 expression in macrophages. Innate Immun.. 2019;25:451-461.
- [CrossRef] [Google Scholar]
- Old player-new tricks: Non angiogenic effects of the VEGF/VEGFR pathway in cancer. Cancers (Basel). 2020;12
- [CrossRef] [Google Scholar]
- Alcoholic liver disease: Pathogenesis and current management. Alcohol Res.. 2017;38:147-161.
- [Google Scholar]
- Isolation and structure elucidation of the major individual polyphenols in carob fibre. Food Chem. Toxicol.. 2003;41:1727-1738.
- [CrossRef] [Google Scholar]
- Endogenous ethanolamide analysis in human plasma using HPLC tandem MS with electrospray ionization. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci.. 2009;877:2052-2060.
- [CrossRef] [Google Scholar]
- Study on quality standard of Shiwuwei Sai'erdou Pill. Chinese J. Mod. Appl. Pharm. 2021
- [CrossRef] [Google Scholar]
- New caffeic acid derivative from Tithonia diversifolia (Hemsl.) A. Gray butanolic extract and its antioxidant activity. Food Chem. Toxicol.. 2017;109:1079-1085.
- [CrossRef] [Google Scholar]
- Phytoestrogen biomonitoring: an extractionless LC-MS/MS method for measuring urinary isoflavones and lignans by use of atmospheric pressure photoionization (APPI) Anal. Bioanal. Chem.. 2012;402:1123-1136.
- [CrossRef] [Google Scholar]
- A sensitive method for determination of furanodiene in rat plasma using liquid chromatography/tandem mass spectrometry and its application to a pharmacokinetic study. Biomed. Chromatogr.. 2012;26:826-832.
- [CrossRef] [Google Scholar]
- Phenylpropanoid glycosides from Tynanthus panurensis: characterization and LC-MS quantitative analysis. J. Agric. Food Chem.. 2005;53:2853-2858.
- [CrossRef] [Google Scholar]
- Rapid analysis of compounds in leaves of Chinese seabuckthorn and Tibetan seabuckthorn by UPLC / Q-TOF-MS. China J. Chinese Mater. Med.. 2016;41:1461-1468.
- [CrossRef] [Google Scholar]
- Rapid determination of underivatized pyroglutamic acid, glutamic acid, glutamine and other relevant amino acids in fermentation media by LC-MS-MS. Analyst. 2002;127:66-69.
- [CrossRef] [Google Scholar]
- The Src family kinases: distinct functions of c-Src, Yes, and Fyn in the liver. Biomol. Concepts. 2013;4:129-142.
- [CrossRef] [Google Scholar]
- Analysis on chemical constituents in rhizomes of Bergenia scopulosa by UHPLC-Q exactive focus MS/MS enhanced publishing. Chinese J. Exp. Trad. Med. Formulae. 2021;27:118-128.
- [CrossRef] [Google Scholar]
- Simultaneous determination of xylazine, free morphine, codeine, 6-acetylmorphine, cocaine and benzoylecgonine in postmortem blood by UPLC-MS-MS. J. Anal. Toxicol.. 2012;36:319-326.
- [CrossRef] [Google Scholar]
- Berberis lycium Royle fruit extract mitigates oxi-inflammatory stress by suppressing NF-kappaB/MAPK signalling cascade in activated macrophages and Treg proliferation in splenic lymphocytes. Inflammopharmacology. 2020;28:1053-1072.
- [CrossRef] [Google Scholar]
- Shen, H., 2021. The Role of Hepatocyte-derived VEGFA in Liver Regeneration and Non-alcoholic Fatty Liver Disease. Doctor, Naval Medical University.
- Application of a liquid chromatography-tandem mass spectrometry method to the pharmacokinetics, tissue distribution and excretion studies of sweroside in rats. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci.. 2014;969:1-11.
- [CrossRef] [Google Scholar]
- An HPLC-MS/MS method for the simultaneous determination of luteolin and its major metabolites in rat plasma and its application to a pharmacokinetic study. J. Sep. Sci.. 2018;41:3830-3839.
- [CrossRef] [Google Scholar]
- Liquid chromatography-tandem mass spectrometry for the determination of jaceosidin in rat plasma. J. Pharm. Biomed. Anal.. 2009;49:381-386.
- [CrossRef] [Google Scholar]
- A sensitive method for the determination of hordenine in human serum by ESI(+) UPLC-MS/MS for forensic toxicological applications. Anal. Bioanal. Chem.. 2016;408:2285-2292.
- [CrossRef] [Google Scholar]
- Qualitative analysis of flavonoids in tetrastigma hemsleyanum based on UPLC-quadrupole/exactive orbitrap mass spectrometry and mass defect filter method. J. Chinese Mass Spectrometry Soc.. 2020;41:359-367.
- [CrossRef] [Google Scholar]
- Simultaneous determination of calycosin-7-O-beta-d-glucoside, calycosin, formononetin, astragaloside IV and schisandrin in rat plasma by LC-MS/MS: application to a pharmacokinetic study after oral administration of Shenqi Wuwei chewable tablets. Biomed. Chromatogr.. 2014;28:1118-1125.
- [CrossRef] [Google Scholar]
- Sun, Z., 2016. Study on Active Ingredients from Penthorum Chinense Pursh. Master Master, Chongqing University.
- Phenotyping of uracil and 5-fluorouracil metabolism using LC-MS/MS for prevention of toxicity and dose adjustment of fluoropyrimidines. Ther. Drug Monit.. 2020;42:540-547.
- [CrossRef] [Google Scholar]
- Flavonoids as strong inhibitors of MAPK3: A computational drug discovery approach. Int. J. Anal. Chem.. 2023;2023:8899240.
- [CrossRef] [Google Scholar]
- Study on the PK profiles of magnoflorine and its potential interaction in Cortex phellodendri decoction by LC-MS/MS. Anal. Bioanal. Chem.. 2014;406:841-849.
- [CrossRef] [Google Scholar]
- The hepatoprotective effect and chemical constituents of total iridoids and xanthones extracted from Swertia mussotii Franch. J. Ethnopharmacol.. 2014;154:259-266.
- [CrossRef] [Google Scholar]
- Chimeric feeders of mesenchymal stromal cells and stromal cells modified with constitutively active AKT expand hematopoietic stem cells. Regen. Med.. 2019;14:535-553.
- [CrossRef] [Google Scholar]
- Development, validation and application of LC-MS/MS method for quantification of amino acids, kynurenine and serotonin in human plasma. J. Pharm. Biomed. Anal.. 2020;180:113018
- [CrossRef] [Google Scholar]
- Determination and pharmacokinetic study of four xanthones in rat plasma after oral administration of Gentianella acuta extract by UHPLC-ESI-MS/MS. J. Ethnopharmacol.. 2015;174:261-269.
- [CrossRef] [Google Scholar]
- A LC-MS/MS method for simultaneous determination of seven alkaloids in rat plasma after oral administration of Phellodendri chinensis cortex extract and its application to a pharmacokinetic study. J. Sep. Sci.. 2019;42:1351-1363.
- [CrossRef] [Google Scholar]
- A pharmacokinetics study of orally administered higenamine in rats using LC-MS/MS for doping control analysis. Drug Test. Anal.. 2020;12:485-495.
- [CrossRef] [Google Scholar]
- Study on fragmentation patterns of coumarins in Notopterygium inchum with ultrahigh performance liquid chromatography combined with quadrupole time-of-flight mass spectrometry. China J. Chinese Mater. Med.. 2021;46:1179-1190.
- [CrossRef] [Google Scholar]
- Wang, L., 2021. Qualitative Analysis and in vivo metabolic profile study of Lagotis brachystachys Maxim based on UPLC-Q-TOF-MS technolog Master, Jiangxi University of Chinese Medicine.
- TIM-4 interference in Kupffer cells against CCL4-induced liver fibrosis by mediating Akt1/Mitophagy signalling pathway. Cell Prolif.. 2020;53:e12731.
- [Google Scholar]
- Determination of acteoside in Cistanche deserticola and Boschniakia rossica and its pharmacokinetics in freely-moving rats using LC-MS/MS. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci.. 2006;844:89-95.
- [CrossRef] [Google Scholar]
- Determination of caprolactam and 6-aminocaproic acid in human urine using hydrophilic interaction liquid chromatography-tandem mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci.. 2012;885–886:61-65.
- [CrossRef] [Google Scholar]
- Simultaneous quantitative determination of sanguinarine, chelerythrine, dihydrosanguinarine and dihydrochelerythrine in chicken by HPLC-MS/MS method and its applications to drug residue and pharmacokinetic study. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci.. 2015;985:124-130.
- [CrossRef] [Google Scholar]
- Fast identification of constituents of Lagotis brevituba by using UPLC-Q-TOF-MS/MS method. Zhongguo Zhong Yao Za Zhi. 2017;42:2123-2130.
- [CrossRef] [Google Scholar]
- Identification of chemical constituents in the Kadsura Coccinea Fructus based on UHPLC-Q-exactive orbitrap MS. China Pharmaceuticals. 2021;30:55-60.
- [CrossRef] [Google Scholar]
- Validated LC-MS/MS method for the determination of 3-hydroxflavone and its glucuronide in blood and bioequivalent buffers: application to pharmacokinetic, absorption, and metabolism studies. J. Pharm. Biomed. Anal.. 2013;85:245-252.
- [CrossRef] [Google Scholar]
- Comparative pharmacokinetics of puerarin, daidzin, baicalin, glycyrrhizic acid, liquiritin, berberine, palmatine and jateorhizine by liquid chromatography-mass spectrometry after oral administration of Gegenqinlian decoction and active components alignment (ACA) to rats. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci.. 2015;988:33-44.
- [CrossRef] [Google Scholar]
- Pharmacokinetic comparisons of naringenin and naringenin-nicotinamide cocrystal in rats by LC-MS/MS. J. Anal. Methods Chem.. 2020;2020:8364218.
- [CrossRef] [Google Scholar]
- Method development and validation for pharmacokinetic and tissue distributions of ellagic acid using ultrahigh performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS) Molecules. 2014;19:18923-18935.
- [CrossRef] [Google Scholar]
- Simultaneous determination of anemoside B4, phellodendrine, berberine, palmatine, obakunone, esculin, esculetin in rat plasma by UPLC-ESI-MS/MS and its application to a comparative pharmacokinetic study in normal and ulcerative colitis rats. J. Pharm. Biomed. Anal.. 2017;134:43-52.
- [CrossRef] [Google Scholar]
- Efficacy of the combination of ademetionine 1,4 - butanedisulfonate for injection in the treatment of patients with cholestatic hepatitis. Heilongjiang Med. Pharm.. 2021;44:109-110.
- [Google Scholar]
- HPLC-Q-TOF-MS/MS-based analysis of chemical constituents in Choerospondiatis fructus. J. Second Military Med. Univ.. 2016;37:159-166.
- [CrossRef] [Google Scholar]
- Study on the fingerprint chromatography of bran atractylodis macrocephalae rhizoma dispensing granules. Asia-Pac. Trad. Med.. 2020;16:78-83.
- [CrossRef] [Google Scholar]
- Identification of major constituents from propolis by LC-IT-TOF-MS. J. China Pharma. Univ.. 2017;48:178-183.
- [CrossRef] [Google Scholar]
- A comprehensive LC-MS/MS method validation for the quantitative investigation of 37 fingerprint phytochemicals in Achillea species: A detailed examination of A. coarctata and A. monocephala. J. Pharm. Biomed. Anal.. 2018;154:413-424.
- [CrossRef] [Google Scholar]
- Yu, J., 2017. Analysis of Polyphenols in Camellia Nitidissima Chi Master, Guangxi University of Traditional Chinese Medicine.
- Validated LC-MS/MS method for the determination of scopoletin in rat plasma and its application to pharmacokinetic studies. Molecules. 2015;20:18988-19001.
- [CrossRef] [Google Scholar]
- Liquid chromatograph/tandem mass spectrometry assay for the simultaneous determination of chlorogenic acid and cinnamic acid in plasma and its application to a pharmacokinetic study. J. Pharm. Biomed. Anal.. 2010;51:685-690.
- [CrossRef] [Google Scholar]
- Zhang, C., Z. Zheng, D. Wang, et al., 2021. Analysis of Chemical Constituents from Calyxes of Physalis alkekengi var. franchetii by UPLC-Q-Orbitrap HRMS. Journal of Chinese Medicinal Materials. 2600-2608. 10.13863/j.issn1001-4454.2021.11.017.
- LC-MS/MS method for quantitation of seven biomarkers in human plasma for the assessment of insulin resistance and impaired glucose tolerance. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci 2016
- [CrossRef] [Google Scholar]
- Quantitation of beta-carboline and quercetin in alligator weed (Alternanthera philoxeroides (Mart.) Griseb.) by LC-MS/MS and evaluation of cardioprotective effects of the methanol extracts. Drug Discov. Ther.. 2018;12:341-346.
- [CrossRef] [Google Scholar]
- Pharmacokinetics, bioavailability, and tissue distribution study of angoroside C and its metabolite ferulic acid in rat using UPLC-MS/MS. Front. Pharmacol.. 2018;9:1186.
- [CrossRef] [Google Scholar]
- Studies on the structure and activity of phenylic compounds from Erigeron brev iscapus. Chin. Pharm. J.. 2002;21–24
- [CrossRef] [Google Scholar]
- Simultaneous determination of harmine, harmaline and their metabolites harmol and harmalol in beagle dog plasma by UPLC-ESI-MS/MS and its application to a pharmacokinetic study. J. Pharm. Biomed. Anal.. 2013;85:162-168.
- [CrossRef] [Google Scholar]
- Swertianlarin, an herbal agent derived from swertia mussotii franch, attenuates liver injury, inflammation, and cholestasis in common bile duct-ligated rats. Evid. Based Complement. Alternat. Med.. 2015;2015:948376
- [CrossRef] [Google Scholar]
- Development of an LC-MS/MS method for quantification of two pairs of isomeric flavonoid glycosides and other ones in rat plasma: Application to pharmacokinetic studies. Biomed. Chromatogr.. 2017;31
- [CrossRef] [Google Scholar]
- Protective effect of ellagic acid on liver injury in rats with alcoholic fatty liver and its effect on TNF-α and Leptin. Chinese Trad. Patent Med.. 2016;38:1821-1824.
- [CrossRef] [Google Scholar]
- Network pharmacology databases for Traditional Chinese Medicine: Review and assessment. Front. Pharmacol.. 2019;10:123.
- [CrossRef] [Google Scholar]
- Protective effects of ellagic acid against alcoholic liver disease in mice. Front. Nutr.. 2021;8:744520
- [CrossRef] [Google Scholar]
- Zheng, X., J. Yang and Y. Yang, 2016. Research progress on pharmacological effects of gallic acid. Chinese Journal of Hospital Pharmacy. 37, 94-98+102. 10.13286/j.cnki.chinhosppharmacyj.2017.01.22.
- Quantitative analysis of the profiles of twelve major compounds in Gentiana straminea Maxim. Roots by LC-MS/MS in an extensive germplasm survey in the Qinghai-Tibetan plateau. J. Ethnopharmacol.. 2021;280:114068
- [CrossRef] [Google Scholar]
- Simultaneous determination of eight components in radix scrophulariae by HPLC-MS/MS. Mod. Chinese Med.. 2017;19:670-674.
- [CrossRef] [Google Scholar]
- Zhu, L., Y. LI and X. LI, 2020. Preliminary Study on Serum Medicinal Chemistry of Tibetan Medicine Bawei Chenxiang Powder based on UPLC-Q-Exactive Technology. Journal of Chinese Medicinal Materials. 43, 88-94. 10.13863/j.issn1001-4454.2020.01.018.
- Development of an LC/MS/MS method in order to determine arctigenin in rat plasma: its application to a pharmacokinetic study. Biomed. Chromatogr.. 2013;27:1123-1128.
- [CrossRef] [Google Scholar]







