Translate this page into:
Discovery of 29-O-acyl-toosendanin-based derivatives as potent anti-cancer agents
⁎Corresponding authors. purong328073@163.com (Rong Pu), tanggh5@mail.sysu.edu.cn (Gui-Hua Tang)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Structural modification of natural products is an effective strategy to discover potent lead compounds with improved medicinal performance. Toosendanin (TSN), a natural limonoid with diverse pharmacological properties, was selected as the starting material for structural modification to obtain more active anti-cancer agents in the current study. A library containing 25 structurally diverse derivatives (including 12 new ones) were constructed on the basis of the structure-guided modification of TSN. Subsequent cytotoxic assay of this library discovered that compounds 14, 18, and 25 showed more significant antiproliferative activity than the precursor TSN in MDA-MB-468 cell model and so as compounds 14, 17–19, 21, and 25 in Hela cell model. Among them, the new derivative 29-O-(6-chloronicotinoyl)-toosendanin (25) exhibited the most potent antiproliferative activity (IC50s 0.05–0.06 μM), being more active than TSN (IC50s 0.14–0.24 μM) and even the first-line drug adriamycin (IC50s ∼ 0.07 μM) in both tested cancer cell lines. The SARs study uncovered that the hemiacetal group, the 14,15-epoxy ring, 1-OH, 7-OH, 3-OAc, and 12-OAc were viewed as the essential active groups and the 29-OH was the critically active modification position of TSN for the enhancement of cytotoxicity. The discovering of 25 from TSN-based derivatives might serve as a lead compound for anti-cancer chemotherapy, which may shed light on rationally design TSN-based derivatives for obtaining more potent anti-tumor agents.
Keywords
Toosendanin derivatives
Anti-cancer activity
Structure−activity relationships
1 Introduction
Natural products are evolutionarily endowed with diverse structures and various biological activities and thus served as a revelatory treasure of “privileged scaffolds” in drug development (Cragg et al., 2009). Up to now, approximately 70 % of FDA-approved anti-cancer drugs are natural products or their derivatives (Newman and Cragg, 2020). Toosendanin (TSN) is a highly oxygenated tetracyclic limonoid isolated from the traditional Chinese medicine Melia toosendan in the 1950 s (Zhang et al., 2019). It possesses various biological functions including insecticide (Shi et al., 1986), prejunctional blocker (Shi et al., 1980), anti-botulinum (Shi and Tang, 2004), anti-obesity (Chen et al., 2018), and anti-cancer (Shi and Wang, 2004) activities. Among them, anti-cancer effect of TSN is the most significant research hit in science community (Fan et al., 2022, Luo et al., 2018, Wang et al., 2015, Wang et al., 2018, Zhang et al., 2005, Zhang et al., 2017). For instance, the He group found that TSN inhibited hepatocellular carcinoma cells by inducing mitochondria-dependent apoptosis (He et al., 2010). The Ma group demonstrated that TSN led to the cell arrested at G1/S phase by increasing p53 protein phosphorylation (Cao et al., 2016). The Zhang group showed that TSN suppressed hepatocellular carcinoma metastasis through JAK2/Stat3 and Wnt/β-catenin signaling (Yang et al., 2021). However, the anti-cancer potential of TSN-derived compounds still remains unclear, largely due to insufficient investigation of systematic structural modification of TSN.
Chemical optimization based on structure–activity relationships (SARs) is an established and prevailing strategy to discover more potent leads with excellent medicinal properties in drug development (Eckert and Bajorath, 2007, Kubinyi, 2002, Peltason and Bajorath, 2007). Such strategy is especially prominent and efficent in natural product-based anti-caner research, as exemplified by vinorelbine, a vinblastine-derived chemotherapy medication with therapeutic efficacy for the treatment of breast cancer and non-small cell lung cancer (Xu et al., 2021), and docetaxel, a classical taxol-based clinic drug for the cure of various cancers including prostate cancer, lung cancer and other solid tumors (Bailly, 2022). In the past few decades, there were few reports related to the SARs of TSN in the scientific community, for instance, the Zhang group developed 12 derivatives of TSN by semi-synthesis and evaluated their activity against the pre-third-instar larvae of Mythimna separata in vivo (Xu and Zhang, 2011), and the Xu group obtained 18 alkyl/alkenylacyloxy derivatives from TSN and evaluated their insecticidal activities (Zhang et al., 2013). However, TSN, as an anti-cancer natural product, the potential anti-cancer activity of its derivatives against human cancer cell lines was not well investigated. Therefore, it is of great value to study the potential anticancer activity of TSN-derived compounds.
In the past few years, our group has endeavored to discover anti-cancer agents from natural products and their modified derivatives, and reported some lead compounds (Fan et al., 2021, Zhang et al., 2018, Zhu et al., 2016). In the current study, a total of 25 derivatives classified into four types (types A − D in Fig. 1) were prepared based on the structure-guided modification of TSN by various chemical reactions including acylation, oxygenation, and hydrolysis. All synthesized derivatives were evaluated for their antiproliferative effects on two human cancer cell lines MDA-MB-468 and Hela. The results of bioassay and subsequent SARs studies indicated that 25 was a potent anti-cancer agent against both cancer cell lines, being more potent than TSN and the positive control adriamycin. Herein, the modified methods, cytotoxic activity, and SARs of these compounds were described.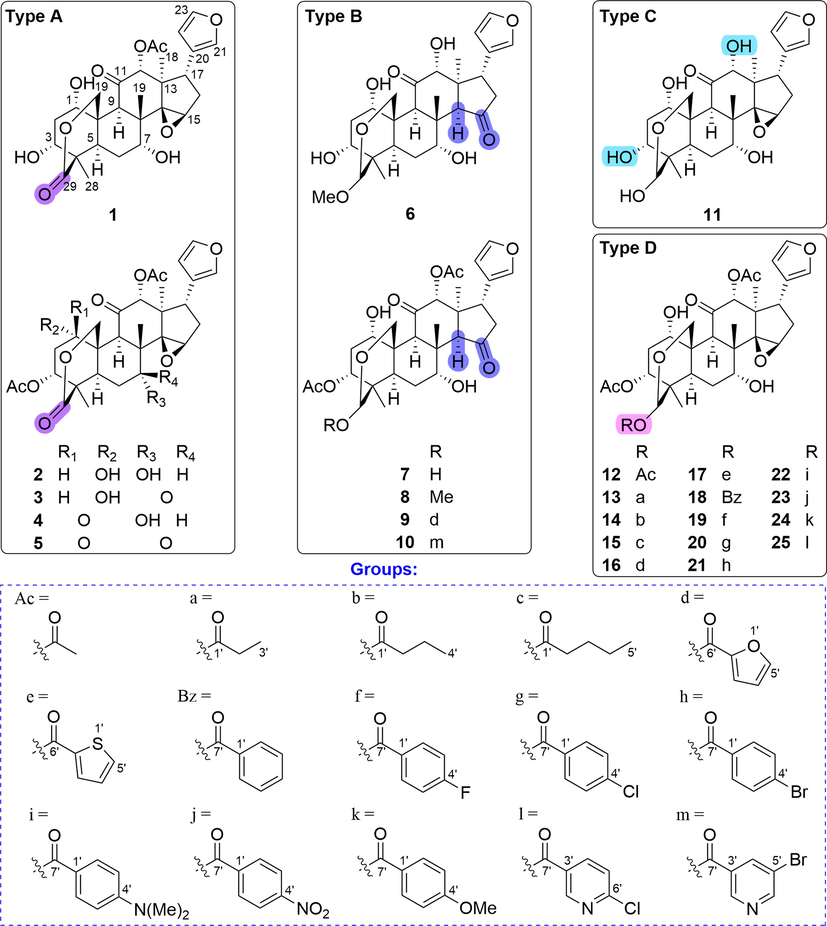
Structures of TSN-based derivatives 1–25.
2 Material and methods
2.1 General information
NMR spectra were measured on a Bruker AM-400 and AM-500 spectrometers at 25 °C. HRESIMS were carried out on a Finnigan LCQ Deca instrument. A Shimadzu LC-20AT equipped with an SPD-M20A PDA detector was used for HPLC, and a YMC-pack ODS-A column (250 × 10 mm, S-5 μm, 12 nm) was used for semipreparative HPLC separation. Silica gel (300–400 mesh, Qingdao Haiyang Chemical Co. ltd.), and Sephadex LH-20 gel (Amersham Biosciences) were used for column chromatography (CC). All solvents were of analytical grade (Guangzhou Chemical Reagents Company, ltd.). The purity of the samples (>95 %) was determined by HPLC, conducted on a Shimadzu LC-20AT series system with Inertsil ODS-SP columns (4.6 mm × 150 mm, 5 μm or 4.6 mm × 100 mm, 5 μm). The samples were eluted with a mixture of MeCN/H2O at a flow rate of 3 mL/min.
2.2 Plant material
The seeds of Melia toosendan Sieb.et Zucc were purchased in June 2020 from Anhui Province, P. R. China, and were authenticated by one of the authors (G. H. Tang). A voucher specimen (accession number: CLZ202006) has been deposited at the School of Pharmaceutical Sciences, Sun Yat-sen University.
2.3 Extraction and isolation
The seeds of M. toosendan (5 kg) were extracted with 95 % EtOH (15 L × 3) at room temperature (rt) to give 600 g of crude extract. The extract was suspended in H2O (3 L) and then partitioned with EtOAc. The EtOAc extract (100 g) was subjected to silica gel CC (CH2Cl2/MeOH, 100:1→0:1) to afford three fractions (Frs. Ι−III). Fr. III (13.7 g) was applied to silica gel CC (PE/EtOAc, 5:1→1:1) to give four fractions (Frs. IIIA−IIID). Fr. IIIC (8.7 g) was separated by Sephadex LH-20 (MeOH) to give two fractions (Frs. IIIC1 and IIIC2). Fr. IIIC1 (8 g) was applied to silica gel CC (PE/EtOAc, 5:1→1:1) to give an amount of toosendanin (TSN, 5 g), which was used as the starting material for the design of various derivatives.
2.4 Structural modification
2.4.1 Preparation of 2–5 by oxygenation of TSN
To a stirred solution of TSN (200 mg, 0.348 mmol) in 2 mL of dichloromethane (DCM) was added excess Dess-Martin Periodinane (1.5 g, 3.54 mmol) at rt and the mixture was stirred overnight. The resulting solution was concentrated by evaporation, washed by H2O (4 mL), and extracted with DCM (5 mL × 3). After removal of the solvents under vacuum, the residue was purified by semi-HPLC (MeCN/H2O, 75:25, 3 mL/min) to give the oxygenated products 2 (14 mg, tR 7 min), 3 (33 mg, tR 9 min), 4 (27 mg, tR 10 min), and 5 (14 mg, tR 11 min), respectively.
2.4.1.1 29-Oxo-toosendanin (2)
White amorphous powder; 1H NMR (CDCl3&CD3OD, 400 MHz) data for the sendanin-type moiety: δH 7.36 (1H, brs, H-23), 7.16 (1H, brs, H-21), 6.15 (1H, brs, H-22), 5.40 (1H, s, H-12), 5.34 (1H, s, H-9), 4.94 (1H, d, J = 14.1 Hz, H-19a), 4.89 (1H, brs, H-3), 4.48 (1H, d, J = 14.1 Hz, H-19b), 4.11 (1H, brs, H-1), 3.80 (1H, brs, H-15), 2.98 (1H, dd, J = 4.1, 13.8 Hz, H-5), 2.92 (1H, dd, J = 6.4, 11.1 Hz, H-17), 2.16 (1H, dd, J = 6.7, 13.5 Hz, H-16a), 2.04 (1H, m, H-2a), 2.00 (1H, m, H-2b), 1.99 (1H, m, H-16b), 1.85 (1H, dt, J = 14.0, 3.7, H-6a), 1.55 (1H, td, J = 1.8, 14.0 Hz, H-6b), 1.52 (3H, s, H-30), 1.30 (3H, s, H-18), 1.28 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.24 (3H, s)], 12-OAc [δH 2.09 (3H, s)]; 13C NMR (CDCl3&CD3OD, 100 MHz) data for the sendanin-type moiety: δC 209.0 (C, C-11), 175.8 (C, C-29), 143.3 (CH, C-21), 141.6 (CH, C-23), 123.5 (C, C-20), 112.6 (CH, C-22), 79.1 (CH, C-12), 74.9 (CH, C-3), 74.4 (CH2, C-19), 72.8 (C, C-14), 70.4 (CH, C-7), 69.7 (CH, C-1), 59.3 (CH, C-15), 50.0 (CH, C-9), 47.4 (C, C-4), 46.7 (C, C-13), 43.9 (C, C-10), 40.4 (C, C-8), 39.5 (CH, C-17), 34.3 (CH2, C-16), 32.6 (CH2, C-2), 28.8 (CH2, C-6), 28.5 (CH, C-5), 22.2 (CH3, C-30), 20.3 (CH3, C-28), 15.8 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 171.8 (C), 21.1 (CH3)], 12-OAc [δC 171.8 (C), 20.9 (CH3)]. The spectroscopic data was identical to meliarachin B (Su et al., 2011).
2.4.1.2 7,29-Dioxo-toosendanin (3)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.35 (1H, t, J = 1.5, H-23), 7.19 (1H, brs, H-21), 6.24 (1H, brs, H-22), 5.20 (1H, d, J = 14.1 Hz, H-19a), 5.00 (1H, d, J = 1.7 Hz, H-3), 4.69 (1H, s, H-12), 4.49 (1H, s, H-9), 4.48 (1H, d, J = 14.1 Hz, H-19b), 4.04 (1H, brs, H-1), 3.92 (1H, s, H-15), 2.98 (1H, dd, J = 5.3, 14.3 Hz, H-5), 2.91 (1H, dd, J = 6.5, 11.1 Hz, H-17), 2.70 (1H, t, J = 14.0, H-6a), 2.49 (1H, dd, J = 5.3, 13.6 Hz, H-6b), 2.33 (1H, dd, J = 6.6, 13.5 Hz, H-16a), 2.19 (1H, m, H-2a), 2.12 (1H, m, H-16b), 2.07 (1H, m, H-2b), 1.38 (3H, s, H-30), 1.18 (3H, s, H-28), 1.07 (3H, s, H-18), for the acyloxy groups: 3-OAc [δH 2.09 (3H, s)], 12-OAc [δH 1.99 (3H, s)]; 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety: δC 206.7 (C, C-7), 203.0 (C, C-11), 172.8 (C, C-29), 142.9 (CH, C-23), 140.9 (CH, C-21), 122.0 (C, C-20), 111.8 (CH, C-22), 81.5 (CH, C-12), 73.6 (CH, C-3), 71.9 (CH2 C-19), 70.2 (CH, C-1), 67.8 (C, C-14), 58.3 (CH, C-15), 54.3 (CH, C-9), 52.1 (C, C-8), 47.6 (C, C-4), 40.9 (CH, C-17), 39.9 (C, C-10), 37.8 (CH2, C-6), 36.6 (CH, C-5), 32.6 (CH2, C-16), 31.2 (CH2, C-2), 21.3 (CH3, C-30), 20.5 (CH3, C-28), 15.3(CH3, C-18), for the acyloxy groups: 3-OAc [δC 169.9 (C), 21.3 (CH3)], 12-OAc [δC 171.1 (C), 19.9 (CH3)]; HRESIMS m/z 593.1994 [M + Na]+ (calcd for C30H34O11Na+, 593.1993).
2.4.1.3 1,29-Dioxo-toosendanin (4)
White amorphous powder; 1H NMR (pyrdine-d5, 400 MHz) data for the sendanin-type moiety: δH 7.58 (1H, brs, H-23), 7.43 (1H, brs, H-21), 6.33 (1H, brs, H-22), 6.04 (1H, s, H-12), 5.74 (1H, dd, J = 1.6, 4.4 Hz, H-3), 5.40 (1H, d, J = 14.6 Hz, H-19a), 5.13 (1H, s, H-9), 5.05 (1H, d, J = 14.6 Hz, H-19b), 4.03 (1H, s, H-15), 3.95 (1H, brs, H-7), 3.83 (1H, dd, J = 4.8, 15.0 Hz, H-2a), 3.26 (1H, dd, J = 6.2, 11.1 Hz, H-17), 3.13 (1H, dd, J = 4.2, 13.2 Hz, H-5), 2.83 (1H, dd, J = 1.7, 15.0 Hz, H-2b), 2.17 (1H, dd, J = 6.4, 13.4 Hz, H-16a), 2.04 (1H, m, H-6a), 1.94 (1H, dd, J = 11.5, 13.2 Hz, H-16b), 1.86 (3H, s, H-18), 1.83 (1H, m, H-6b), 1.31 (3H, s, H-28), 1.31 (3H, s, H-30), for the acyloxy groups: 3-OAc [δH 1.77 (3H, s)], 12-OAc [δH 2.01 (3H, s)]; 13C NMR (pyrdine-d5, 100 MHz) data for the sendanin-type moiety: δC 206.8 (C, C-11), 206.5 (C, C-1), 173.0 (C, C-29), 143.6 (CH, C-23), 141.9 (CH, C-21), 113.0 (CH, C-22), 78.5 (CH, C-12), 77.2 (CH, C-3), 73.4 (C, C-14), 71.3 (CH2, C-19), 69.0 (CH, C-7), 59.7 (CH, C-15), 50.4 (CH, C-9), 49.3 (C, C-10), 47.5 (C, C-4), 46.6 (C, C-13), 43.3 (C, C-8), 42.4 (CH2, C-2), 39.2 (CH, C-17), 38.5 (CH, C-5), 34.5 (CH2, C-16), 30.3 (CH2, C-6), 22.3 (CH3, C-30), 20.0 (CH3, C-28), 16.7 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 170.0 (C), 20.8 (CH3)], 12-OAc [δC 170.9 (C), 21.3 (CH3)]; HRESIMS m/z 593.1997 [M + Na]+ (calcd for C30H34O11Na+, 593.1993).
2.4.1.4 1,7,29-Trioxo-toosendanin (5)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.35 (1H, t, J = 1.5, H-23), 7.16 (1H, brs, H-21), 6.12 (1H, brs, H-22), 5.53 (1H, s, H-12), 5.36 (1H, dd, J = 2.1, 4.5 Hz, H-3), 5.19 (1H, d, J = 15.0 Hz, H-19a), 5.00 (1H, d, J = 15.0 Hz, H-19b), 4.33 (1H, s, H-9), 4.11 (1H, s, H-15), 3.26 (1H, dd, J = 4.6, 15.3 Hz, H-2a), 2.98 (1H, dd, J = 6.5, 11.2 Hz, H-17), 2.86 (1H, t, J = 15.3 Hz, H-6a), 2.64 (1H, dd, J = 2.0, 15.3 Hz, H-2b), 2.45 (1H, m, 6b), 2.32 (1H, m, 16a), 2.04 (1H, m, 16b), 1.45 (3H, s, H-30), 1.29 (3H, s, H-18), 1.23 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.09 (3H, s)], 12-OAc [δH 2.02 (3H, s)]; 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety: δC 205.0 (C, C-7), 203.4 (C, C-1), 202.6 (C, C-11), 171.0 (C, C-29), 143.0 (CH, C-23), 140.8 (CH, C-21), 121.9 (C, C-20), 111.5 (CH, C-22), 77.0 (CH, C-12), 75.1 (CH, C-3), 69.2 (CH2, C-19), 68.2 (C, C-14), 58.4 (CH, C-15), 54.3 (CH, C-9), 51.3 (C, C-8), 48.2 (C, C-10), 47.4 (C, C-4), 46.2 (C, C-13), 44.7 (CH, C-5), 41.4 (CH2, C-2), 39.5 (CH, C-17), 38.0 (CH2, C-6), 33.2 (CH2, C-16), 21.8 (CH3, C-30), 19.1 (CH3, C-28), 15.6 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 169.2 (C), 21.0 (CH3)], 12-OAc [δC 170.0 (C), 20.8 (CH3)]; HRESIMS m/z 591.1813 [M + Na]+ (calcd for C30H32O11Na+, 591.1837).
2.4.2 Preparation of 1 by hydrolysis of 2
To a stirred solution of 2 (50 mg, 0.087 mmol) in 1 mL of MeOH was added excess saturated NaHCO3 soluton at rt, which was kept stirring overnight. After that, the resulting mixture was diluted with H2O (4 mL) and then extracted with EtOAc (5 mL × 3). The obtained organic layers were evaporated in vacuo and then purified by semi-preparative HPLC (MeCN/H2O, 70:30, 3 mL/min) to give compound 1 (20 mg, tR 12 min).
2.4.2.1 3-Deacetyl-29-oxo-toosendanin (1)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.34 (1H, brs, H-23), 7.15 (1H, brs, H-21), 6.21 (1H, brs, H-22), 4.95 (1H, d, J = 14.1 Hz, H-19a), 4.82 (1H, s, H-12), 4.68 (1H, s, H-9), 4.43 (1H, d, J = 14.2 Hz, H-19b), 4.13 (1H, brs, H-1), 3.78 (1H, brs, H-3), 3.71 (1H, d, J = 3.8 Hz, H-15), 2.93 (1H, dd, J = 6.4, 10.9 Hz, H-17), 2.82 (1H, dd, J = 3.6, 13.6 Hz, H-5), 2.24 (1H, dd, J = 6.3, 13.4 Hz, H-16a), 2.11 (1H, m, H-2a), 1.98 (1H, m, H-16b), 1.94 (1H, m, H-2b), 1.83 (1H, m, H-6a), 1.66 (1H, m, H-6b), 1.24 (3H, s, H-28), 1.22 (3H, s, H-18), 1.12 (3H, s, H-30), for the acyloxy group: 12-OAc [δH 1.97 (3H, s)]; 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety: δC 206.9 (C, C-11), 175.3 (C, C-29), 142.7 (CH, C-21), 140.9 (CH, C-23), 122.5 (C, C-20), 112.0 (CH, C-22), 80.7 (CH, C-12), 74.5 (CH, C-3), 72.6 (CH2, C-19), 71.8 (CH, C-1), 71.6 (C, C-14), 70.3 (CH, C-7), 57.7 (CH, C-15), 48.9 (CH, C-9), 48.3 (C, C-4), 47.4 (C, C-13), 43.5 (C, C-8), 40.2 (C, C-10), 39.6 (CH, C-17), 33.1 (CH2, C-16), 33.0 (CH2, C-2), 28.6 (CH2, C-6), 27.0 (CH, C-5), 21.7 (CH3, C-30), 20.2 (CH3, C-28), 16.3 (CH3, C-18), for the acyloxy group: 12-OAc [δC 171.1 (C), 20.6 (CH3)]; HRESIMS m/z 553.2076 [M + Na]+ (calcd for C28H34O10Na+, 553.2044).
2.4.3 Preparation of 6 from TSN
To a solution of TSN (50 mg, 0.087 mmol) in MeOH (1 mL) was added 100 μL of 1 % H2SO4 at rt and was stirred overnight. Subsequently, the reaction mixture was diluted with 4 mL of H2O and extracted with EtOAc (5 mL × 3). The combined organic layers were dried over anhydrous Na2SO4 and then evaporated in vacuo. The resulting residue was separated by semi-preparative HPLC (MeCN/H2O, 80:20, 3 mL/min) to give the product 6 (15 mg, tR 10 min).
2.4.3.1 3,12-Dideacetyl-29-O-methyl-isotoosendanin (6)
White amorphous powder; 1H NMR (CDCl3, 500 MHz) data for the sendanin-type moiety: δH 7.38 (1H, t, J = 1.5 Hz, H-23), 7.30 (1H, brs, H-21), 6.42 (1H, brs, H-22), 4.81 (1H, brs, H-1), 4.45 (1H, s, H-12), 4.16 (1H, brs, H-7), 4.09 (1H, s, H-29), 4.06 (1H, d, J = 11.7 Hz, H-19a), 3.66 (1H, brs, H-3), 3.60 (1H, d, J = 11.7 Hz, H-19b), 3.51 (1H, t, J = 8.9 Hz, H-17), 3.38 (1H, s, H-9), 3.05 (1H, s, H-14), 2.81 (1H, dt, J = 4.4, 15.4 Hz, H-2a), 2.69 (1H, dd, J = 8.4, 18.0 Hz, H-5), 2.61 (1H, m, H-16a), 2.58 (1H, m, H-16b), 2.50 (1H, dt, J = 2.1, 14.6 Hz, H-6a), 1.97 (1H, d, J = 15.5 Hz, H-2b), 1.71 (1H, dt, J = 3.8, 14.7 Hz, H-6b), 1.22 (3H, s, H-30), 0.99 (3H, s, H-28), 0.83 (3H, s, H-18), for the methoxy group: 29-OMe [δH 3.33 (3H, s)]; 13C NMR (CDCl3, 125 MHz) data for the sendanin-type moiety: δC 218.5 (C, C-15), 212.9 (C, C-11), 143.2 (CH, C-23), 140.2 (CH, C-21), 124.1 (C, C-20), 111.2 (CH, C-22), 103.3 (CH, C-29), 79.2 (CH, C-12), 75.4 (CH, C-3), 71.8 (CH, C-1), 69.9 (CH, C-7), 60.5 (CH, C-14), 58.0 (CH2, C-19), 49.0 (CH, C-9), 45.6 (C, C-13), 44.7 (CH2, C-16), 43.9 (C, C-8), 41.6 (C, C-4), 41.4 (C, C-10), 41.3 (CH, C-17), 37.9 (CH2, C-2), 25.2 (CH2, C-6), 24.7 (CH, C-5), 22.6 (CH3, C-30), 21.3 (CH3, C-18), 18.9 (CH3, C-28), for the methoxy group: 29-OMe [δC 55.7 (CH3)]. The spectroscopic data was identical to that reported of meliarachin I (Su et al., 2011).
2.4.4 Preparation of 7 and 17 from TSN
An excess of 2-thiophenecarbonyl chloride (100 μL) was added to a freshly distilled pyridine solution of TSN (100 mg, 0.174 mmol) at rt. The resulting solution was stirred for 1 h and then quenched by adding 2 mL of anhydrous EtOH. The residue obtained by removal of the solvent under vacuum was subjected to semi-preparative HPLC (MeCN/H2O, 70:30, 3 mL/min) to yield compounds 17 (35 mg, tR 12 min) and 7 (25 mg, tR 13 min).
2.4.4.1 Isotoosendanin (7)
White amorphous powder; 1H NMR (pyrdine-d5, 400 MHz) data for the sendanin-type moiety: δH 7.58 (1H, brs, H-21), 7.57 (1H, brs, H-23), 6.45 (1H, brs, H-22), 6.02 (1H, d, J = 4.0 Hz, H-3), 5.52 (1H, s, H-12), 5.41 (1H, s, H-29), 4.79 (1H, d, J = 12.4 Hz, H-19a), 4.78 (1H, brs, H-1), 4.72 (1H, s, H-9), 4.67 (1H, d, J = 12.4 Hz, H-19b), 4.58 (1H, brs, H-7), 3.55 (1H, t, J = 9.1 Hz, H-17), 4.13 (1H, s, H-14), 3.32 (1H, dt, J = 4.7, 15.6 Hz, H-2a), 2.78 (2H, t, J = 9.1 Hz, H-16), 2.38 (1H, m, H-6a), 2.35 (1H, d, J = 15.6 Hz, H-2b), 2.05 (1H, dt, J = 3.8, 14.3 Hz, H-6b), 1.35 (3H, s, H-30), 1.21 (3H, s, H-18), 1.20 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 1.97 (3H, s)], 12-OAc [δH 2.12 (3H, s)]; 13C NMR (pyrdine-d5, 100 MHz) data for the sendanin-type moiety: δC 217.6 (C, C-15), 209.1 (C, C-11), 144.1 (CH, C-23), 141.5 (CH, C-21), 124.3 (C, C-20), 111.7 (CH, C-22), 97.1 (CH, C-29), 80.3 (CH, C-12), 74.7 (CH, C-3), 71.1 (CH, C-1), 69.8 (CH, C-7), 65.2 (CH2, C-19), 59.8 (CH, C-14), 48.4 (CH, C-9), 45.5 (C, C-13), 45.4 (C, C-8), 45.0 (CH2, C-16), 42.9 (C, C-10), 39.0 (C, C-4), 38.9 (CH, C-17), 37.6 (CH2, C-2), 29.2 (CH, C-5), 24.7 (CH2, C-6), 22.3 (CH3, C-18), 21.3 (CH3, C-30), 20.4 (CH3, C-28), for the acyloxy groups: 3-OAc [δC 171.0 (C), 21.7 (CH3)], 12-OAc [δC 171.1 (C), 21.2 (CH3)]. The spectroscopic data was identical to known compound isotoosendanin (Xie and Yuan, 1985).
2.4.4.2 29-O-(2-Thiophenecarbonyl)-toosendanin (17)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.33 (1H, t, J = 1.5 Hz, H-23), 7.13 (1H, brs, H-21), 6.15 (1H, brs, H-22), 5.99 (1H, brs, H-29), 5.42 (1H, d, J = 3.3 Hz, H-3), 5.27 (1H, s, H-12), 4.63 (1H, s, H-9), 4.38 (1H, d, J = 13.3 Hz, H-19a), 4.30 (1H, d, J = 13.3 Hz, H-19b), 3.76 (1H, brs, H-15), 3.69 (1H, brs, H-7), 2.97 (1H, dd, J = 6.3, 11.0 Hz, H-17), 2.90 (1H, dt, J = 4.6, 16.5 Hz, H-2a), 2.76 (1H, dd, J = 4.1, 13.9 Hz, H-5), 2.24 (1H, dd, J = 6.5, 13.2 Hz, H-6a), 2.06 (1H, m, H-16a), 1.98 (1H, m, H-2b), 1.92 (1H, m, H-6b), 1.76 (1H, dt, J = 3.7, 14.3 Hz, H-16b), 1.32 (3H, s, H-18), 1.18 (3H, s, H-30), 0.91 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.12 (3H, s)], 12-OAc [δH 1.98 (3H, s)], 29-O-2-thiophenecarbonyl [δH 7.85 (1H, dd, J = 1.2, 3.8 Hz, H-5′), 7.61 (1H, dd, J = 1.2, 5.0 Hz, H-3′), 7.12 (1H, m, H-4′)]; 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety: δC 206.9 (C, C-11), 142.6 (CH, C-23), 140.8 (CH, C-21), 122.6 (C, C-20), 112.2 (CH, C-22), 95.7 (CH, C-29), 78.8 (CH, C-12), 73.6 (CH, C-3), 72.1 (C, C-14), 70.6 (CH, C-7), 70.1 (CH, C-1), 64.9 (CH2, C-19), 58.5 (CH, C-15), 48.6 (CH, C-9), 46.1 (C, C-13), 42.7 (C, C-8), 41.5 (C, C-10), 39.8 (C, C-4), 38.5 (CH, C-17), 34.9 (CH2, C-2), 33.7 (CH2, C-6), 28.0 (CH, C-5), 26.2 (CH2, C-16), 22.3 (CH3, C-30), 19.9 (CH3, C-28), 15.9 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 170.1 (C), 21.6 (CH3)], 12-OAc [δC 170.6 (C), 20.9 (CH3)], 29-O-2-thiophenecarbonyl [δC 160.9 (C, C-6′), 134.6 (CH, C-3′), 133.7 (CH, C-5′), 132.8 (C, C-2′), 128.2 (CH, C-4′)]. HRESIMS m/z 707.2137 [M + Na]+ (calcd for C35H40O12SNa+, 707.2133).
2.4.5 Preparation of 8 from TSN
To a stirred solution of TSN (50 mg, 0.087 mmol) in 1 mL of MeOH was added excess formic acid (500 μL) at rt. The reaction mixture was then stirred at 50 °C for 4 h. After that, the mixture solution was diluted with 4 mL of H2O and followed by extraction with EtOAc for three times (each 5 mL). The collected organic layers were evaporated in vacuo to yield a crude, which was then purified by semi-preparative HPLC (MeCN/H2O, 70:30, 3 mL/min) to give the product 8 (20 mg, tR 13 min).
2.4.5.1 29-O-Methyl-isotoosendanin (8)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.38 (1H, t, J = 1.6 Hz, H-23), 7.29 (1H, s, H-21), 6.26 (1H, dd, J = 0.7, 1.7 Hz, H-22), 5.15 (1H, s, H-12), 4.91 (1H, d, J = 3.8 Hz, H-3), 4.20 (1H, brs, H-1), 4.17 (1H, s, H-29), 4.12 (1H, d, J = 12.3 Hz, H-19a), 4.02 (1H, brs, H-7), 3.95 (1H, d, J = 12.3 Hz, H-19b), 3.69 (1H, s, H-9), 3.36 (1H, t, J = 9.7 Hz, H-17), 3.20 (1H, s, H-14), 2.84 (1H, dt, J = 4.8, 16.1 Hz, H-2a), 2.59 (2H, dd, J = 3.6, 10.1 Hz, H-16), 2.51 (1H, dd, J = 1.8, 13.7 Hz, H-6a), 1.79 (1H, d, J = 16.1 Hz, H-2b), 1.71 (1H, dt, J = 2.8, 14.2 Hz, H-6b), 1.19 (3H, s, H-30), 1.01 (3H, s, H-18), 0.85 (3H, s, H-28), for the acyloxy and methoxyl groups: 3-OAc [δH 2.09 (3H, s)], 12-OAc [δH 2.09 (3H, s)], 29-OMe [δH 3.32 (3H, s)]; 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety: δC 218.0 (C, C-15), 207.2 (C, C-11), 143.4 (CH, C-23), 140.5 (CH, C-21), 122.5 (C, C-20), 110.5 (CH, C-22), 102.8 (CH, C-29), 79.8 (CH, C-12), 76.6 (CH, C-3), 71.1 (CH, C-1), 69.6 (CH, C-7), 60.4 (CH, C-14), 58.4 (CH2, C-19), 48.3 (CH, C-9), 46.7 (C, C-13), 44.4 (C, C-10), 44.1 (CH2, C-16), 42.5 (C, C-8), 40.4 (C, C-4), 39.6 (CH, C-17), 36.4 (CH2, C-2), 25.9 (CH, C-5), 25.4 (CH2, C-6), 21.7 (CH3, C-18), 20.9 (CH3, C-30), 18.3 (CH3, C-28), for the acyloxy and methoxyl groups: 3-OAc [δC 170.0 (C), 21.1 (CH3)], 12-OAc [δC 170.8 (C), 21.15 (CH3)], 29-OMe [δC 55.6 (CH3)]. The spectroscopic data was identical to that reported of meliarachin G (Su et al., 2011).
2.4.6 Preparation of 9 and 16 from TSN
To a stirred solution of TSN (100 mg, 0.174 mmol) in freshly distilled DCM (2 mL) was added 100 μL of Et3N, followed by the treatment with furoyl chloride (100 μL) at rt. The resulting mixture solution was kept stirring at rt overnight and then quenched with anhydrous EtOH (2 mL). After removal of the solvent under vacuum, the residues were subjected to semi-preparative HPLC (MeCN/H2O, 75:25, 3 mL/min) to afford compounds 9 (20 mg, tR10 min) and 16 (23 mg, tR 13 min).
2.4.6.1 29-O-(2-Furoyl)-isotoosendanin (9)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.39 (1H, brs, H-23), 7.30 (1H, brs, H-21), 6.27 (1H, brs, H-22), 5.97 (1H, s, H-29), 5.48 (1H, d, J = 3.2 Hz, H-3), 5.07 (1H, s, H-12), 4.34 (1H, d, J = 12.7 Hz, H-19a), 4.28 (1H, d, J = 12.7 Hz, H-19b), 4.13 (1H, d, J = 3.1 Hz, H-1), 3.79 (1H, s, H-9), 3.37 (1H, d, J = 9.7 Hz, H-17), 3.23 (1H, s, H-14), 2.87 (1H, dt, J = 4.7, 16.7 Hz, H-2a), 2.73 (1H, dd, J = 3.0, 13.3 Hz, H-5), 2.62 (1H, m, H-16a), 2.60 (1H, m, H-16b), 2.18 (1H, H-6a), 2.10 (1H, H-6b), 1.93 (1H, d, J = 16.6 Hz, H-2b), 1.20 (3H, s, H-30), 1.02 (3H, s, H-18), 0.87 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.12 (3H, s)], 12-OAc [δH 2.09 (3H, s)], 29-O-2-furoyl [δH 7.62 (1H, brs, H-5′), 7.26 (1H, brs, H-3′), 6.52 (1H, brs, H-4′)]; 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety: δC 218.0 (C, C-15), 207.0 (C, C-11), 143.5 (CH, C-23), 140.7 (CH, C-21), 122.2 (C, C-20), 110.5 (CH, C-22), 94.7 (CH, C-29), 79.5 (CH, C-12), 73.8 (CH, C-3), 70.9 (CH, C-1), 69.4 (CH, C-7), 64.8 (CH2, C-19), 60.4 (CH, C-14), 47.9 (CH, C-9), 47.2 (C, C-13), 44.8 (C, C-8), 43.9 (CH2, C-16), 42.1 (C, C-10), 39.5 (C, C-4), 39.3 (CH, C-17), 35.7 (CH2, C-2), 28.5 (CH, C-5), 23.4 (CH2, C-6), 21.6 (CH3, C-18), 20.9 (CH3, C-30), 18.81 (CH3, C-28), for the acyloxy groups: 3-OAc [δC 169.8 (C), 21.6 (CH3)], 12-OAc [δC 170.7 (C), 20.8 (CH3)], 29-O-2-furoyl [δC 157.2 (C, C-6′), 147.5 (CH, C-5′), 143.7 (C, C-2′), 119.6 (CH, C-3′), 112.2 (CH, C-4′)]; HRESIMS m/z 691.2508 [M + Na]+ (calcd for C35H40O13Na+, 691.2361).
2.4.6.2 29-O-(2-Furoyl)-toosendanin (16)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.33 (1H, brs, H-23), 7.13 (1H, brs, H-21), 6.15 (1H, brs, H-22), 6.01 (1H, s, H-29), 5.39 (1H, d, J = 3.4 Hz, H-3), 5.26 (1H, s, H-12), 4.63 (1H, s, H-9), 4.37 (1H, d, J = 13.3 Hz, H-19a), 4.30 (1H, d, J = 13.3 Hz, H-19b), 3.76 (1H, brs, H-15), 3.69 (1H, brs, H-7), 2.98 (1H, dd, J = 6.3, 11.0 Hz, H-17), 2.89 (1H, dt, J = 4.5, 16.4 Hz, H-2a), 2.77 (1H, dd, J = 4.0, 13.9 Hz, H-5), 2.24 (1H, dd, J = 6.5, 13.3 Hz, H-6), 2.06 (1H, m, H-16a), 1.95 (1H, m, H-2b), 1.93 (1H, m, H-6b), 1.76 (1H, dt, J = 3.8, 14.6 Hz, H-16b), 1.32 (3H, s, H-18), 1.18 (3H, s, H-30), 0.90 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.11 (3H, s)], 12-OAc [δH 1.97 (3H, s)], 29-O-2-furoyl [δH 7.62 (1H, brs, H-5′), 7.25 (1H, d, J = 4.3 Hz, H-3′), 6.52(1H, dd, J = 1.7, 3.5 Hz, H-4′)]; 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety: δC 206.9 (C, C-11), 142.6 (CH, C-23), 140.8 (CH, C-21), 122.6 (C, C-20), 112.2 (CH, C-22), 95.4 (CH, C-29), 78.8 (CH, C-12), 73.7 (CH, C-3), 72.1 (C, C-14), 70.6 (CH, C-7), 70.1 (CH, C-1), 64.9 (CH2, C-19), 58.6 (CH, C-15), 48.6 (CH, C-9), 46.1 (C, C-13), 42.7 (C, C-8), 41.5 (C, C-10), 39.8 (C, C-4), 38.5 (CH, C-17), 34.9 (CH2, C-2), 33.7 (CH2, C-6), 28.0 (CH, C-5), 26.2 (CH2, C-16), 22.3 (CH3, C-30), 19.8 (CH3, C-28), 15.9 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 170.1 (C), 21.6 (CH3)], 12-OAc [δC 170.6 (C), 20.9 (CH3)], 29-O-2-furoyl [δC 157.3 (C, C-6′), 147.4 (CH, C-5′), 143.8 (C, C-2′), 119.5 (CH, C-3′), 112.0 (CH, C-4′)]; HRESIMS m/z 691.2383 [M + Na]+ (calcd for C35H40O13Na+, 691.2361).
2.4.7 Preparation of 10 by hydrolysis of TSN
To a stirred solution of TSN (100 mg, 0.174 mmol) in freshly distilled pyridine (2 mL) was added excess 5-bromonicotinoyl chloride (100 μL) at rt. The resulting solution was stirred at rt overnight and then quenched by adding 2 mL of anhydrous EtOH. After removal of the solvent under vacuum, the residues were successively purified by flash CC (PE:EtOAc, 20:1 → 5:1) and semi-preparative HPLC (MeCN/H2O, 70:30, 3 mL/min) to yield the corresponding acylated product 10 (30 mg, tR 12.5 min).
2.4.7.1 29-O-(5-Bromonicotinoyl)-isotoosendanin (10)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.40 (1H, t, J = 1.6 Hz, H-23), 7.31 (1H, brs, H-21), 6.28 (1H, brs, H-22), 6.02 (1H, s, H-29), 5.51 (1H, d, J = 3.1 Hz, H-3), 5.07 (1H, s, H-12), 4.36 (1H, d, J = 13.0 Hz, H-19a), 4.32(1H, d, J = 13.0 Hz, H-19b), 4.15 (1H, brs, H-1), 3.81 (1H, s, H-9), 3.38 (1H, t, J = 9.7 Hz, H-17), 3.23 (1H, s, H-14), 2.87 (1H, dt, J = 4.5, 16.3 Hz, H-2a), 2.77 (1H, dd, J = 3.5, 13.8 Hz, H-5), 2.62 (1H, m, H-16a), 2.60 (1H, m, H-16b), 2.18 (1H, H-6a), 2.10 (1H, H-6b), 1.94 (1H, brs, H-2b), 1.21 (3H, s, H-30), 1.03 (3H, s, H-18), 0.89 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.13 (3H, s)], 12-OAc [δH 2.10 (3H, s)], 29-O-5-bromonicotinyl [δH 9.14 (1H, d, J = 1.6 Hz, H-2′), 8.88 (1H, d, J = 2.2 Hz, H-6′), 8.40 (1H, m, H-4′)]; 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety: δC 218.0 (C, C-15), 207.0 (C, C-11), 143.5 (CH, C-23), 140.7 (CH, C-21), 122.2 (C, C-20), 110.5 (CH, C-22), 95.7 (CH, C-29), 79.5 (CH, C-12), 73.5 (CH, C-3), 70.9 (CH, C-1), 69.4 (CH, C-7), 65.1 (CH2, C-19), 60.4 (CH, C-14), 47.9 (CH, C-9), 47.2 (C, C-13), 44.8 (C, C-8), 43.9 (CH2, C-16), 42.1 (C, C-10), 39.6 (C, C-4), 39.3 (CH, C-17), 35.9 (CH2, C-2), 28.6 (CH, C-5), 23.3 (CH2, C-6), 21.6 (CH3, C-18), 20.8 (CH3, C-30), 18.8 (CH3, C-28), for the acyloxy groups: 3-OAc [δC 169.7 (C), 21.5 (CH3)], 12-OAc [δC 170.7 (C), 20.8 (CH3)], 29-O-5-bromonicotinyl [δC 163.0 (C, C-7′), 155.4 (CH, C-6′), 149.2 (CH, C-2′), 139.9 (CH, C-4′), 126.8 (C, C-3′), 120.9 (C, C-5′)]; HRESIMS m/z 782.1622 [M + Na]+ (calcd for C36H40O12NBrNa+, 780.1626).
2.4.8 Preparation of 11 by alkaline hydrolysis of TSN
To a solution of TSN (100 mg, 0.174 mmol) in 1 mL of MeOH at rt was added excess saturated NaHCO3 solution, which was stirred overnight. The resulting solution was diluted with H2O (4 mL) and then extracted with EtOAc (5 mL × 3). The combined organic phases were evaporated in vacuo and purified by flash CC (PE:EtOAc, 5:1 → 1:1) to give a totally-deacetyl product 11 (45 mg).
2.4.8.1 3,12-Dideacetyl-toosendanin (11)
Colorless oil; 1H NMR (pyrdine-d5, 400 MHz) data: δH 7.57 (1H, brs, H-21), 7.55 (1H, t, J = 1.5 Hz, H-23), 7.02 (1H, brs, H-22), 6.01 (1H, m, H-12), 5.49 (1H, s, H-29), 4.93 (1H, d, J = 13.0 Hz, H-19a), 4.79 (1H, d, J = 12.6 Hz, H-19b), 4.53 (1H, s, H-9), 4.04 (1H, s, H-15), 3.61 (1H, m, H-3), 3.51 (1H, dt, J = 3.7, 14.4 Hz, H-5), 3.33 (1H, dt, J = 5.9, 10.7 Hz, H-17), 2.36 (1H, m, H-2a), 2.31 (1H, m, H-16a), 2.24 (1H, m, H-2b), 2.05 (1H, m, H-6a), 2.05 (1H, m, H-16b), 1.87 (1H, m, H-6b), 1.92 (3H, s, H-18), 1.40 (3H, s, H-30), 1.39 (3H, s, H-28); 13C NMR (pyrdine-d5, 100 MHz) data: δC 216.0 (C, C-11), 142.9 (CH, C-23), 141.7 (CH, C-21), 126.0 (C, C-20), 114.4 (CH, C-22), 97.9 (CH, C-29), 80.1 (CH, C-12), 74.3 (C, C-14), 72.6 (CH, C-1), 72.6 (CH, C-3), 71.1 (CH, C-7), 65.2 (CH2, C-19), 59.2 (CH, C-15), 49.9 (CH, C-9), 47.9 (C, C-13), 43.8 (C, C-8), 43.5 (C, C-10), 42.5 (C, C-4), 41.0 (CH, C-17), 38.9 (CH2, C-2), 34.2 (CH2, C-16), 28.5 (CH, C-5), 27.0 (CH2, C-6), 23.6 (CH3, C-30), 21.9 (CH3, C-28), 15.6 (CH3, C-18). The spectroscopic data was identical to that reported of deacetylated toosendanin (Fischer et al., 2009).
2.4.9 Preparation of 12–15 by acylation of TSN
Each anhydride (including acetic, propionic, butyric, and valeric anhydrides with the amounts of 40, 56, 71, and 86 μL, respectively) was added dropwise to a stirred solution of TSN (50 mg, 0.087 mmol) in freshly distilled pyridine (2 mL) under the protection of N2. Each resulting solution was allowed to keep stirring overnight under the same condition, which was then quenched by adding 2 mL of anhydrous ethanol. After removal of the solvent under vacuum, each residue was successively purified by flash CC (CH2Cl2:MeOH, 100:1 → 50:1) and semi-preparative HPLC (MeCN/H2O, 75:25, 3 mL/min) to afford the corresponding compounds: 12 (from the reaction mixture of TSN and acetic anhydride; 20 mg, tR 8 min), 13 (from the reaction mixture of TSN and propionic anhydride; 23 mg, tR 10.5 min), 14 (from the reaction mixture of TSN and butyric anhydride; 18 mg, tR 11 min), and 15 (from the reaction mixture of TSN and valeric anhydride; 16 mg, tR 12 min).
2.4.9.1 29-O-Acetyl-toosendanin (12)
White amorphous powder; 1H NMR (CD3OD, 500 MHz) data for the sendanin-type moiety: δH 7.40 (1H, t, J = 1.5 Hz, H-23), 7.20 (1H, brs, H-21), 6.16 (1H, d, J = 0.9 Hz, H-22), 5.78 (1H, s, H-29), 5.34 (1H, s, H-12), 5.12 (1H, d, J = 3.9 Hz, H-3), 4.38 (1H, d, J = 12.8 Hz, H-19a), 4.24 (1H, d, J = 12.8 Hz, H-19b), 4.23 (1H, d, J = 4.3 Hz, H-1), 3.81 (1H, brs, H-15), 3.60 (1H, brs, H-7), 2.91 (1H, dd, J = 4.7, 14.2 Hz, H-5), 2.88 (1H, dd, J = 5.9, 11.2 Hz, H-17), 2.66 (1H, dt, J = 4.8, 16.1 Hz, H-2a), 2.12 (1H, dd, J = 6.5, 13.4 Hz, H-16a), 2.02 (1H, m, H-16b), 1.99 (1H, m, H-6a), 1.86 (1H, d, J = 16.1 Hz, H-2b), 1.74 (1H, dt, J = 3.8, 14.2 Hz, H-6b), 1.37 (3H, s, H-18), 1.13 (3H, s, H-30), 0.82 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.09 (3H, s)], 12-OAc [δH 1.94 (3H, s)], 29-OAc [δH 2.07 (3H, s)]; 13C NMR (CD3OD, 125 MHz) data for the sendanin-type moiety: δC 209.1 (C, C-11), 143.7 (CH, C-23), 142.1 (CH, C-21), 124.2 (C, C-20), 113.0 (CH, C-22), 96.2 (CH, C-29), 79.7 (CH, C-12), 74.6 (CH, C-3), 73.5 (C, C-14), 70.7 (CH, C-1), 70.3 (CH, C-7), 66.3 (CH2, C-19), 59.9 (CH, C-15), 50.1 (CH, C-9), 46.9 (C, C-13), 43.9 (C, C-10), 42.6 (C, C-8), 40.3 (C, C-4), 39.9 (CH, C-17), 36.7 (CH2, C-2), 34.8 (CH2, C-16), 29.5 (CH, C-5), 26.4 (CH2, C-6), 20.8 (CH3, C-18), 19.4 (CH3, C-28), 15.8 (CH3, C-30), for the acyloxy groups: 3-OAc [δC 172.7 (C), 22.9 (CH3)], 12-OAc [δC 172.1 (C), 21.3 (CH3)], 29-OAc [δC 171.4 (C), 20.9 (CH3)]. The spectroscopic data was identical to that reported (Xu and Zhang, 2011).
2.4.9.2 29-O-Propionyl-toosendanin (13)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.33 (1H, brs, H-23), 7.13 (1H, brs, H-21), 6.15 (1H, brs, H-22), 5.81 (1H, s, H-29), 5.27 (1H, s, H-12), 4.61 (1H, s, H-9), 4.33 (1H, d, J = 13.2 Hz, H-19a), 4.29 (1H, d, J = 13.5 Hz, H-19b), 3.75 (1H, brs, H-15), 3.67 (1H, brs, H-7), 2.97 (1H, dd, J = 6.3, 11.1 Hz, H-17), 2.81 (1H, dt, J = 4.6, 16.4 Hz, H-2a), 2.72 (1H, dd, J = 4.0, 13.9 Hz, H-5), 2.24 (1H, dd, J = 6.4, 13.5 Hz, H-6a), 2.03 (1H, m, H-16a), 1.99 (1H, m, H-2b), 1.91 (1H, m, H-6b), 1.72 (1H, dt, J = 3.5, 14.3 Hz, H-16b), 1.32 (3H, s, H-18), 1.16 (3H, s, H-30), 0.82 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.10 (3H, s)], 12-OAc [δH 1.97 (3H, s)], 29-O-propionyl [δH 2.40 (1H, q, J = 7.5 Hz, H-2′a) 2.03 (1H, m, H-2′b), 1.15 (3H, t, J = 7.5 Hz, H-3′)], 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety: δC 206.8 (C, C-11), 142.6 (CH, C-23), 140.8 (CH, C-21), 122.6 (C, C-20), 112.0 (CH, C-22), 94.5 (CH, C-29), 78.7 (CH, C-12), 73.8 (CH, C-3), 72.1 (C, C-14), 70.6 (CH, C-7), 70.2 (CH, C-1), 64.8 (CH2, C-19), 58.6 (CH, C-15), 48.6 (CH, C-9), 46.1 (C, C-13), 42.7 (C, C-8), 41.7 (C, C-10), 39.5 (C, C-4), 38.5 (CH, C-17), 35.2 (CH2, C-2), 33.7 (CH2, C-6), 28.5 (CH, C-5), 25.9 (CH2, C-16), 22.5 (CH3, C-30), 19.5 (CH3, C-28), 15.9 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 170.0 (C), 21.6 (CH3)], 12-OAc [δC 170.5 (C), 20.9 (CH3)], 29-O-propionyl [δC 173.4 (C, C-1′), 27.8 (CH2, C-2′), 9.0 (CH3, C-3′)]. The spectroscopic data was identical to that reported (Xu and Zhang, 2011).
2.4.9.3 29-O-Butyryl-toosendanin (14)
White amorphous powder; 1H NMR(CDCl3, 500 MHz) data for the sendanin-type moiety: δH 7.33 (1H, brs, H-23), 7.12 (1H, brs, H-21), 6.14 (1H, brs, H-22), 5.80 (1H, s, H-29), 5.27 (1H, s, H-12), 4.60 (1H, s, H-9), 4.33 (1H, d, J = 13.2 Hz, H-19a), 4.28 (1H, d, J = 13.2 Hz, H-19b), 3.75 (1H, s, H-15), 3.66 (1H, brs, H-7), 2.97 (1H, dd, J = 6.3, 11.0 Hz, H-17), 2.80 (1H, dt, J = 4.5, 16.4 Hz, H-2a), 2.72 (1H, dd, J = 3.9, 14.0 Hz, H-5), 2.23 (1H, dd, J = 6.4, 13.1 Hz, H-6a), 2.05 (1H, m, H-16a), 1.99 (1H, m, H-2b), 1.72 (1H, m, H-16b), 1.31 (3H, s, H-18), 1.16 (3H, s, H-30), 0.82 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.10 (3H, s)], 12-OAc [δH 1.97 (3H, s)], 29-O-butyryl [δH 2.35 (2H, t, J = 7.4 Hz, H-2′) 1.66 (2H, m, H-3′), 0.95 (3H, t, J = 7.4 Hz, H-4′)]; 13C NMR (CDCl3, 125 MHz) data for the sendanin-type moiety: δC 206.8 (C, C-11), 142.6 (CH, C-23), 140.8 (CH, C-21), 122.6 (C, C-20), 112.0 (CH, C-22), 94.4 (CH, C-29), 78.7 (CH, C-12), 73.8 (CH, C-3), 72.1 (C, C-14), 70.5 (CH, C-7), 70.2 (CH, C-1), 64.8 (CH2, C-19), 58.6 (CH, C-15), 48.5 (CH, C-9), 46.1 (C, C-13), 42.7 (C, C-8), 41.6 (C, C-10), 39.4 (C, C-4), 38.5 (CH, C-17), 35.1 (CH2, C-2), 33.7 (CH2, C-6), 28.1 (CH, C-5), 25.9 (CH2, C-16), 22.4 (CH3, C-30), 19.5 (CH3, C-28), 15.9 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 170.1 (C), 21.6 (CH3)], 12-OAc [δC 170.6 (C), 20.9 (CH3)], 29-O-butyryl [δC 172.6 (C, C-1′), 36.4 (CH2, C-2′), 18.4 (CH2, C-3′), 13.7 (CH3, C-4′)]. The spectroscopic data was identical to that reported (Xu and Zhang, 2011).
2.4.9.4 29-O-Valeryl-toosendanin (15)
White amorphous powder; 1H NMR (CDCl3, 500 MHz) data for the sendanin-type moiety: δH 7.33 (1H, brs, H-23), 7.12 (1H, brs, H-21), 6.14 (1H, brs, H-22), 5.80 (1H, s, H-9), 5.27 (1H, s, H-3), 4.60 (1H, s, H-9), 4.33 (1H, d, J = 13.2 Hz, H-19a), 4.28 (1H, d, J = 13.2 Hz, H-19b), 4.26 (1H, brs, H-1), 3.75 (1H, s, H-5), 3.66 (1H, brs, H-7), 2.97 (1H, dd, J = 6.3, 11.0 Hz, H-17), 2.80 (1H, dt, J = 4.5, 16.4 Hz, H-2a), 2.72 (1H, dd, J = 4.0, 14.0 Hz, H-5), 2.23 (1H, dd, J = 6.2, 13.4 Hz, H-6a), 2.06 (1H, dd, J = 1.4, 14.4 Hz, H-16a), 1.99 (1H, m, H-2b), 1.90 (1H, m, H-6b), 1.72 (1H, m, H-16b), 1.31 (3H, s, H-18), 1.16 (3H, s, H-30), 0.82 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.10 (3H, s)], 12-OAc [δH 1.97 (3H, s)], 29-O-valeryl [δH 2.37 (2H, t, J = 7.5 Hz, H-2′) 1.61 (2H, dt, J = 7.5, 15.1 Hz, H-3′), 1.35 (2H, m, H-4′), 0.91 (3H, t, J = 7.4 Hz, H-5′)]; 13C NMR (CDCl3, 125 MHz) data for the sendanin-type moiety: δC 206.8 (C, C-11), 142.6 (CH, C-23), 140.8 (CH, C-21), 122.6 (C, C-20), 112.0 (CH, C-22), 94.5 (CH, C-29), 78.7 (CH, C-12), 73.8 (CH, C-3), 72.1 (C, C-14), 70.5 (CH, C-7), 70.2 (CH, C-1), 64.8 (CH2, C-19), 58.6 (CH, C-15), 48.5 (CH, C-9), 46.1 (C, C-13), 42.7 (C, C-8), 41.6 (C, C-10), 39.4 (C, C-4), 38.5 (CH, C-17), 35.1 (CH2, C-2), 33.7 (CH2, C-6), 28.1 (CH, C-5), 25.9 (CH2, C-16), 22.4 (CH3, C-30), 19.5 (CH3, C-28), 15.9 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 170.1 (C), 21.6 (CH3)], 12-OAc [δC 170.6 (C), 20.9 (CH3)], 29-O-valeryl [δC 172.8 (C,C-1′), 34.2 (CH2, C-2′), 26.9 (CH2, C-3′), 22.3 (CH2, C-4′), 13.8 (CH3, C-5′)]. The spectroscopic data was identical to that reported (Zhang et al., 2013).
2.4.10 Preparation of 18–25 by acylation of TSN
Each acyl chloride (including benzoyl, 4-fluorobenzoyl, 4-chlorobenzoyl, 4-bromobenzoyl, 4-dimethylaminobenzoyl, 4-nitrobenzoyl, 4-anisoyl, and 6-chloronicotinoyl chlorides) (100 μL) was added to a stirred solution of TSN (100 mg, 0.174 mmol) in freshly distilled pyridine (2 mL) at rt. Each resulting solution was then stirred at rt overnight and followed by quenching with 2 mL of anhydrous EtOH. After removal of the solvent under vacuum, each residue was subjected to flash CC (PE:EtOAc, 20:1 → 5:1) and semi-preparative HPLC (MeCN/H2O, 70:30, 3 mL/min) to yield the corresponding acylated compounds: 18 (from the reaction mixture of TSN and benzoyl chloride; 35 mg, tR 12 min), 19 (from the reaction mixture of TSN and 4-fluorobenzoyl chloride; 40 mg, tR 11 min), 20 (from the reaction mixture of TSN and 4-chlorobenzoyl chloride; 50 mg, tR 14 min), 21 (from the reaction mixture of TSN and 4-bromobenzoyl chloride; 40 mg, tR 9 min), 22 (from the reaction mixture of TSN and 4-dimethylaminobenzoyl chloride; 30 mg, tR 12 min), 23 (from the reaction mixture of TSN and 4-nitrobenzoyl chloride; 45 mg, tR 15 min), 24 (from the reaction mixture of TSN and 4-anisoyl; 55 mg, tR 12.5 min), and 25 (from the reaction mixture of TSN and 6-chloronicotinoyl chloride; 50 mg, tR 13.5 min).
2.4.10.1 29-O-Benzoyl-toosendanin (18)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.34 (1H, t, J = 1.6 Hz, H-23), 7.14 (1H, brs, H-21), 6.15 (1H, d, J = 1.0 Hz, H-22), 6.04 (1H, s, H-29), 5.49 (1H, d, J = 3.3 Hz, H-3), 5.28 (1H, s, H-12), 4.65 (1H, s, H-9), 4.42 (1H, d, J = 13.2 Hz, H-19a), 4.36 (1H, d, J = 13.2 Hz, H-19b), 4.31 (1H, brs, H-1), 3.77 (1H, brs, H-15), 3.70 (1H, brs, H-7), 2.98 (1H, dd, J = 6.3, 11.0 Hz, H-17), 2.92 (1H, dd, J = 4.7, 6.5 Hz, H-2a), 2.79 (1H, dd, J = 4.0, 13.9 Hz, H-5), 2.25 (1H, dd, J = 6.6, 13.3 Hz, H-6a), 2.08 (1H, dd, J = 1.9, 14.2 Hz, H-16a), 2.00 (1H, m, H-2b), 1.94 (1H, dd, J = 11.6, 13.7 Hz, H-6b), 1.78(1H, dt, J = 3.9, 14.4 Hz, H-16b), 1.33 (3H, s, H-18), 1.20 (3H, s, H-30), 0.91 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.13 (3H, s)], 12-OAc [δH 1.98 (3H, s)], 29-OBz [δH 8.04 (2H, dd, J = 1.2, 8.3 Hz, H-2′/6′), 7.59 (1H, tt, J = 1.3, 7.0 Hz, H-4′), 7.47 (2H, t, J = 7.7 Hz,H-3′/5′)]; 13C NMR(CDCl3, 100 MHz) data for the sendanin-type moiety: δC 206.8 (C, C-11), 142.6 (CH, C-23), 140.8 (CH, C-21), 122.6 (C, C-20), 112.0 (CH, C-22), 95.3 (CH, C-29), 78.8 (CH, C-12), 73.7 (CH, C-3), 72.1 (C, C-14), 70.6 (CH, C-7), 70.2 (CH, C-1), 65.0 (CH2, C-19), 58.6 (CH, C-15), 48.6 (CH, C-9), 46.1 (C, C-13), 42.7 (C, C-8), 41.7 (C, C-10), 39.8 (C, C-4), 38.5 (CH, C-17), 35.3 (CH2, C-2), 33.7 (CH2, C-6), 28.2 (CH, C-5), 26.0 (CH2, C-16), 22.5 (CH3, C-30), 19.6 (CH3, C-28), 15.9 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 170.1 (C), 21.7 (CH3)], 12-OAc [δC 170.6 (C), 20.9 (CH3)], 29-OBz [δC 165.4 (C, C-7′), 133.8 (CH, C-4′), 130.1(CH × 2, C-2′/6′), 129.4 (C, C-1′), 128.8 (CH × 2, C-3′/5′)]. The spectroscopic data was identical to that reported (Xu and Zhang, 2011).
2.4.10.2 29-O-(4-Fluorobenzoyl)-toosendanin (19)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.33 (1H, t, J = 1.6 Hz, H-23), 7.13 (1H, brs, H-21), 6.14 (1H, d, J = 1.0 Hz, H-22), 6.00 (1H, s, H-29), 5.45 (1H, d, J = 3.5 Hz, H-3), 5.26 (1H, s, H-12), 4.64 (1H, s, H-9), 4.41 (1H, d, J = 13.2 Hz, H-19a), 4.35 (1H, d, J = 13.2 Hz, H-19b), 4.30 (1H, brs, H-1), 3.76 (1H, s, H-15), 3.69 (1H, brs, H-7), 2.96 (1H, dd, J = 6.3, 11.1 Hz, H-17), 2.90 (1H, dt, J = 4.6, 16.3 Hz, H-2a), 2.80 (1H, dd, J = 4.0, 13.9 Hz, H-5), 2.23 (1H, dd, J = 6.5, 13.4 Hz, H-6a), 2.05 (1H, dd, J = 1.5, 14.2 Hz, H-16a), 1.98 (1H, m, H-2b), 1.91 (1H, dd, J = 11.5, 13.4 Hz, H-6b), 1.77 (1H, dt, J = 3.7, 14.2 Hz, H-16b), 1.32 (3H, s, H-18), 1.18 (3H, s, H-30), 0.89 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.12 (3H, s)], 12-OAc [δH 1.97 (3H, s)], 29-O-(4-fluorobenzoyl) [δH 8.05 (2H, dd, J = 5.4, 8.8 Hz, H-2′/6′), 7.13 (2H, t, J = 8.6 Hz, H-3′/5′)]; 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety: δC 206.9 (C, C-11), 142.6 (CH, C-23), 140.8 (CH, C-21), 122.6 (C, C-20), 112.0 (CH, C-22), 95.4 (CH, C-29), 78.8 (CH, C-12), 73.7 (CH, C-3), 72.1 (C, C-14), 70.4 (CH, C-7), 70.1 (CH, C-1), 65.0 (CH2, C-19), 58.6 (CH, C-15), 48.5 (CH, C-9), 46.1 (C, C-13), 42.7 (C, C-8), 41.6 (C, C-10), 39.8 (C, C-4), 38.5 (CH, C-17), 35.3 (CH2, C-2), 33.7 (CH2, C-6), 28.2 (CH, C-5), 25.9 (CH2, C-16), 22.5 (CH3, C-30), 19.5 (CH3, C-28), 15.9 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 170.2 (C), 21.6 (CH3)], 12-OAc [δC 170.6 (C), 20.8 (CH3)], 29-O-(4-fluorobenzoyl) [δC 166.3 (C, C-4′), 164.4 (C, C-7′), 132.6 (CH × 2, C-2′/6′), 125.6 (C, C-1′), 116.0 (CH × 2, C-3′/5′)]; HRESIMS m/z 719.2460 [M + Na]+ (calcd for C37H41O12FNa+, 719.2474).
2.4.10.3 29-O-(4-Chlorobenzoyl)-toosendanin (20)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.34 (1H, t, J = 1.6 Hz, H-23), 7.14 (1H, brs, H-21), 6.15 (1H, d, J = 1.0 Hz, H-22), 6.02 (1H, s, H-29), 5.46 (1H, d, J = 3.5 Hz, H-3), 5.27 (1H, s, H-12), 4.65 (1H, s, H-9), 4.41 (1H, d, J = 13.2 Hz, H-19a), 4.36 (1H, d, J = 13.2 Hz, H-19b), 4.31 (1H, brs, H-1), 3.76 (1H, s, H-15), 3.70 (1H, brs, H-7), 2.98 (1H, dd, J = 6.3, 11.1 Hz, H-17), 2.90 (1H, dt, J = 4.6, 16.3 Hz, H-2a), 2.80 (1H, dd, J = 4.0, 13.9 Hz, H-5), 2.25 (1H, dd, J = 6.5, 13.5 Hz, H-6a), 2.07 (1H, dd, J = 1.7, 14.2 Hz, H-16a), 1.98 (1H, m, H-2b), 1.92 (1H, dd, J = 11.2, 13.3 Hz, H-6b), 1.77 (1H, dt, J = 3.7, 14.3 Hz, H-16b), 1.33 (3H, s, H-18), 1.19 (3H, s, H-30), 0.90 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.13 (3H, s)], 12-OAc [δH 1.98 (3H, s)], 29-O-(4-chlorobenzoyl) [δH 7.97 (2H, d, J = 8.6 Hz, H-2′/6′), 7.44 (2H, d, J = 8.6 Hz, H-3′/5′)]; 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety: δC 206.8 (C, C-11), 142.6 (CH, C-23), 140.8 (CH, C-21), 122.6 (C, C-20), 112.0 (CH, C-22), 95.6 (CH, C-29), 78.8 (CH, C-12), 73.5 (CH, C-3), 72.1 (C, C-14), 70.6 (CH, C-7), 70.1 (CH, C-1), 65.1 (CH2, C-19), 58.6 (CH, C-15), 48.5 (CH, C-9), 46.1 (C, C-13), 42.7 (C, C-8), 41.7 (C, C-10), 39.8 (C, C-4), 38.5 (CH, C-17), 35.3 (CH2, C-2), 33.7 (CH2, C-6), 28.2 (CH, C-5), 26.0 (CH2, C-16), 22.5 (CH3, C-30), 19.5 (CH3, C-28), 15.9 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 170.1 (C), 21.6 (CH3)], 12-OAc [δC 170.6 (C), 20.9 (CH3)], 29-O-(4- chlorobenzoyl) [δC 164.6 (C, C-7′), 140.4 (C, C-4′), 131.4 (CH × 2, C-2′/6′), 129.2 (CH × 2, C-3′/5′), 127.9 (C, C-1′)]; HRESIMS m/z 735.2206 [M + Na]+ (calcd for C37H41O12ClNa+, 735.2179). The spectroscopic data was identical to that reported (Xu and Zhang, 2011).
2.4.10.4 29-O-(4-Bromobenzoyl)-toosendanin (21)
White amorphous powder; 1H NMR (CDCl3, 500 MHz) data for the sendanin-type moiety:δH 7.34 (1H, t, J = 1.5 Hz, H-23), 7.14 (1H, brs, H-21), 6.15 (1H, brs, H-22), 6.02 (1H, s, H-29), 5.45 (1H, d, J = 3.6 Hz, H-3), 5.27 (1H, s, H-12), 4.65 (1H, s, H-9), 4.41 (1H, d, J = 13.2 Hz, H-19a), 4.37 (1H, d, J = 13.2 Hz, H-19b), 4.31 (1H, brs, H-1), 3.76 (1H, s, H-15), 3.70 (1H, brs, H-7), 2.98 (1H, dd, J = 6.3, 11.1 Hz, H-17), 2.90 (1H, dt, J = 4.6, 16.4 Hz, H-2a), 2.80 (1H, dd, J = 4.1, 14.0 Hz, H-5), 2.25 (1H, dd, J = 6.2, 13.3 Hz, H-6a), 2.08 (1H, td, J = 2.0, 14.4 Hz, H-16a), 1.98 (1H, m, H-2b), 1.92 (1H, dd, J = 11.4, 13.4 Hz, H-6b), 1.77 (1H, dt, J = 3.8, 14.3 Hz, H-16b), 1.33 (3H, s, H-18), 1.19 (3H, s, H-30), 0.90 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.13 (3H, s)], 12-OAc [δH 1.98 (3H, s)], 29-O-(4-bromobenzoyl) [δH 7.90 (2H, d, J = 8.5 Hz, H-3′/5′), 7.61 (2H, d, J = 8.5 Hz, H-2′/6′)]; 13C NMR (CDCl3, 125 MHz) data for the sendanin-type moiety: δC 206.8 (C, C-11), 142.6 (CH, C-23), 140.8 (CH, C-21), 122.6 (C, C-20), 112.0 (CH, C-22), 95.6 (CH, C-29), 78.8 (CH, C-12), 73.5 (CH, C-3), 72.1 (C, C-14), 70.6 (CH, C-7), 70.1 (CH, C-1), 65.1 (CH2, C-19), 58.6 (CH, C-15), 48.5 (CH, C-9), 46.1 (C, C-13), 42.7 (C, C-8), 41.7 (C, C-10), 39.8 (C, C-4), 38.5 (CH, C-17), 35.3 (CH2, C-2), 33.7 (CH2, C-6), 28.2 (CH, C-5), 26.0 (CH2, C-16), 22.5 (CH3, C-30), 19.5 (CH3, C-28), 15.9 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 170.1 (C), 21.6 (CH3)], 12-OAc [δC 170.6 (C), 20.9 (CH3)], 29-O-(4-bromobenzoyl) [δC 164.7 (C, C-7′), 132.2 (CH × 2, C-2′/6′), 131.5 (CH × 2, C-3′/5′), 129.1 (C, C-1′), 128.3 (C, C-4′)]; HRESIMS m/z 779.1698 [M + Na]+ (calcd for C37H41O12BrNa+, 779.1674).
2.4.10.5 29-O-(4-Dimethylaminobenzoyl)-toosendanin (22)
White amorphous powder; 1H NMR (CDCl3, 500 MHz) data for the sendanin-type moiety: δH 7.27 (1H, brs, H-23), 7.01 (1H, brs, H-21), 6.67 (1H, brs, H-22), 6.05 (1H, s, H-29), 5.36 (1H, d, J = 4.5 Hz, H-3), 5.06 (1H, s, H-12), 4.79 (1H, s, H-9), 4.55 (1H, d, J = 13.2 Hz, H-19a), 4.41 (1H, d, J = 13.2 Hz, H-19b), 4.28 (1H, d, J = 2.5 Hz, H-1), 3.79 (1H, s, H-15), 3.78 (1H, brs, H-7), 2.98 (1H, m, H-17), 2.90 (1H, m, H-2a), 2.89 (1H, dd, J = 6.3, 11.4 Hz, H-5), 2.76 (1H, m, H-16a), 2.68 (1H, m, H-16b), 2.10 (1H, dd, J = 6.5, 13.8 Hz, H-6a), 1.98 (1H, m, H-2b), 1.80 (1H, m, H-6b), 1.32 (3H, s, H-18), 1.24 (3H, s, H-30), 0.79 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 1.90 (3H, s)], 12-OAc [δH 1.89 (3H, s)], 29-O-(4-dimethylaminobenzoyl) [δH 8.05 (2H, d, J = 9.0 Hz, H-3′/5′), 8.02 (2H, d, J = 9.0 Hz, H-2′/6′), 1.99 (6H, s, N(Me)2)]; 13C NMR (CDCl3, 125 MHz) data for the sendanin-type moiety: δC 206.2 (C, C-11), 142.4 (CH, C-23), 140.8 (CH, C-21), 122.6 (C, C-20), 112.1 (CH, C-22), 96.0 (CH, C-29), 78.9 (CH, C-12), 74.0 (CH, C-3), 72.2 (C, C-14), 71.0 (CH, C-7), 70.8 (CH, C-1), 64.0 (CH2, C-19), 58.6 (CH, C-15), 49.6 (CH, C-9), 46.1 (C, C-13), 42.2 (C, C-8), 41.6 (C, C-10), 40.2 (C, C-4), 38.1 (CH, C-17), 35.5 (CH2, C-2), 33.7 (CH2, C-6), 29.2 (CH, C-5), 26.8 (CH2, C-16), 23.1 (CH3, C-30), 19.9 (CH3, C-28), 16.1 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 169.7 (C), 21.2 (CH3)], 12-OAc [δC 170.5 (C), 20.9 (CH3)], 29-O-(4-dimethylaminobenzoyl) [δC 165.7 (C, C-7′), 153.8 (C, C-4′), 131.7 (CH × 2, C-2′/C-6′), 116.8 (C, C-1′), 110.9 (CH × 2, C-3′/C-5′) 40.2 (CH3 × 2, N(Me)2)]; HRESIMS m/z 744.2987 [M + Na]+ (calcd for C39H47O12NNa+, 744.2990).
2.4.10.6 29-O-(4-Nitrobenzoyl)-toosendanin (23)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.34 (1H, t, J = 1.6 Hz, H-23), 7.14 (1H, brs, H-21), 6.16 (1H, d, J = 1.0 Hz, H-22), 6.05 (1H, s, H-29), 5.46 (1H, d, J = 3.5 Hz, H-3), 5.25 (1H, s, H-12), 4.66 (1H, s, H-9), 4.43 (1H, d, J = 13.2 Hz, H-19a), 4.38 (1H, d, J = 13.2 Hz, H-19b), 4.31 (1H, d, J = 2.9 Hz, H-1), 3.77 (1H, s, H-15), 3.71 (1H, brs, H-7), 2.98 (1H, dd, J = 6.3, 11.1 Hz, H-17), 2.90 (1H, m, H-2a), 2.83 (1H, dd, J = 4.4, 14.2 Hz, H-5), 2.25 (1H, dd, J = 6.6, 13.3 Hz, H-6a), 2.08 (1H, m, H-16a), 1.98 (1H, m, H-2b), 1.92 (1H, m, H-6b), 1.77 (1H, m, H-16b), 1.33 (3H, s, H-18), 1.20 (3H, s, H-30), 0.92 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.14 (3H, s)], 12-OAc [δH 1.98 (3H, s)], 29-O-(4-nitrobenzoyl) [δH 8.32 (2H, d, J = 8.9 Hz, H-3′/5′), 8.21 (2H, d, J = 8.9 Hz, H-2′/6′)]; 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety: δC 206.8 (C, C-11), 142.6 (CH, C-23), 140.8 (CH, C-21), 122.6 (C, C-20), 112.0 (CH, C-22), 96.3 (CH, C-29), 78.9 (CH, C-12), 73.3 (CH, C-3), 72.1 (C, C-14), 71.0 (CH, C-7), 70.0 (CH, C-1), 65.2 (CH2, C-19), 58.6 (CH, C-15), 48.5 (CH, C-9), 46.2 (C, C-13), 42.8 (C, C-8), 41.6 (C, C-10), 40.0 (C, C-4), 38.6 (CH, C-17), 35.3 (CH2, C-2), 33.7 (CH2, C-6), 28.2 (CH, C-5), 26.0 (CH2, C-16), 22.5 (CH3, C-30), 19.5 (CH3, C-28), 16.0 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 170.2 (C), 21.6 (CH3)], 12-OAc [δC 170.6 (C), 20.9 (CH3)], 29-O-(4-nitrobenzoyl) [δC 163.6 (C, C-7′), 151.1 (C, C-4′), 134.8 (C, C-1′), 131.2 (CH × 2, C-2′/6′), 124.0 (CH × 2, C-3′/5′)]. The spectroscopic data was identical to that reported (Xu and Zhang, 2011).
2.4.10.7 29-O-(4-Anisoyl)-toosendanin (24)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.33 (1H, t, J = 1.5 Hz, H-23), 7.13 (1H, brs, H-21), 6.15 (1H, brs, H-22), 6.01 (1H, s, H-29), 5.49 (1H, d, J = 3.4 Hz, H-3), 5.28 (1H, s, H-12), 4.64 (1H, s, H-9), 4.41 (1H, d, J = 13.2 Hz, H-19a), 4.36 (1H, d, J = 13.2 Hz, H-19b), 4.31 (1H, brs, H-1), 3.76 (1H, s, H-15), 3.70 (1H, brs, H-7), 2.98 (1H, dd, J = 7.0, 11.8 Hz, H-17), 2.92 (1H, dd, J = 5.8, 10.5 Hz, H-2a), 2.78 (1H, dd, J = 4.0, 13.8 Hz, H-5), 2.25 (1H, dd, J = 6.2, 13.6 Hz, H-6a), 2.08 (1H, dd, J = 1.7, 14.2 Hz, H-16a), 1.98 (1H, m, H-2b), 1.91 (1H, dd, J = 11.2, 13.3 Hz, H-6b), 1.77 (1H, dt, J = 3.5, 14.2 Hz, H-16b), 1.33 (3H, s, H-18), 1.19 (3H, s, H-30), 0.90 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.13 (3H, s)], 12-OAc [δH 1.98 (3H, s)], 29-O-(4-anisoyl) [δH 8.00 (2H, d, J = 8.9 Hz, H-2′/6′), 6.94 (2H, d, J = 8.9 Hz, H-3′/5′), 3.86 (3H, s, 4′-OMe)]; 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety: δC 206.8 (C, C-11), 142.6 (CH, C-23), 140.8 (CH, C-21), 122.6 (C, C-20), 112.0 (CH, C-22), 95.0 (CH, C-29), 78.8 (CH, C-12), 73.8 (CH, C-3), 72.1 (C, C-14), 70.6 (CH, C-7), 70.2 (CH, C-1), 64.9 (CH2, C-19), 58.6 (CH, C-15), 48.6 (CH, C-9), 46.1 (C, C-13), 42.7 (C, C-8), 41.7 (C, C-10), 39.9 (C, C-4), 38.5 (CH, C-17), 35.3 (CH2, C-2), 33.7 (CH2, C-6), 28.3 (CH, C-5), 26.0 (CH2, C-16), 22.5 (CH3, C-30), 19.6 (CH3, C-28), 15.9 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 170.1 (C), 21.7 (CH3)], 12-OAc [δC 170.6 (C), 20.9 (CH3)], 29-O-(4-anisoyl) [δC 165.1 (C, C-7′), 164.1 (C, C-4′), 132.2 (CH × 2, C-2′/6′),121.7 (C, C-1′), 114.1 (CH × 2, C-3′/5′), 55.6 (CH3, 5′-OMe)]. The spectroscopic data was identical to that reported (Xu and Zhang, 2011).
2.4.10.8 29-O-(6-Chloronicotinoyl)-toosendanin (25)
White amorphous powder; 1H NMR (CDCl3, 400 MHz) data for the sendanin-type moiety: δH 7.33 (1H, t, J = 1.6 Hz, H-23), 7.13 (1H, brs, H-21), 6.15 (1H, d, J = 1.0 Hz, H-22), 6.04 (1H, s, H-29), 5.41 (1H, d, J = 3.4 Hz, H-3), 5.26 (1H, s, H-12), 4.65 (1H, s, H-9), 4.41 (1H, d, J = 13.2 Hz, H-19a), 4.36 (1H, d, J = 13.2 Hz, H-19b), 4.30 (1H, brs, H-1), 3.76 (1H, s, H-15), 3.70 (1H, brs, H-7), 2.98 (1H, dd, J = 6.3, 11.1 Hz, H-17), 2.87(1H, m, H-2a), 2.80 (1H, dd, J = 4.4, 14.6 Hz, H-5), 2.24 (1H, dd, J = 6.6, 13.2 Hz, H-6a), 2.06 (1H, m, H-16a), 1.98 (1H, m, H-2b), 1.93 (1H, dd, J = 11.2, 13.2 Hz, H-6b), 1.78 (1H, m, H-16b), 1.32 (3H, s, H-18), 1.19 (3H, s, H-30), 0.90 (3H, s, H-28), for the acyloxy groups: 3-OAc [δH 2.12 (3H, s)], 12-OAc [δH 1.98 (3H, s)], 29-O-(6-chloronicotinyl) [δH 9.01 (H, dd, J = 0.5, 2.4 Hz, H-2′), 8.22 (H, dd, J = 2.4, 8.3 Hz, H-4′), 7.44 (H, dd, J = 0.5, 8.3 Hz, H-5′)]; 13C NMR (CDCl3, 100 MHz) data for the sendanin-type moiety:δC 206.8 (C, C-11), 142.6 (CH, C-23), 140.8 (CH, C-21), 122.6 (C, C-20), 112.0 (CH, C-22), 96.1 (CH, C-29), 78.9 (CH, C-12), 73.2 (CH, C-3), 72.1 (C, C-14), 70.5 (CH, C-7), 70.0 (CH, C-1), 65.2 (CH2, C-19), 58.6 (CH, C-15), 48.5 (CH, C-9), 46.2 (C, C-13), 42.7 (C, C-8), 41.6 (C, C-10), 39.8 (C, C-4), 38.6 (CH, C-17), 35.2 (CH2, C-2), 33.7 (CH2, C-6), 28.2 (CH, C-5), 26.0 (CH2, C-16), 22.5 (CH3, C-30), 19.6 (CH3, C-28), 15.9 (CH3, C-18), for the acyloxy groups: 3-OAc [δC 170.0 (C), 21.6 (CH3)], 12-OAc [δC 170.6 (C), 20.8 (CH3)], 29-O-(6-chloronicotinyl) [δC 163.4 (C, C-7′), 156.6 (C, C-6′), 151.6 (CH, C-2′), 139.8 (CH, C-4′), 124.6 (CH, C-5′), 124.5 (C, C-3′)]; HRESIMS m/z 736.2172 [M + Na]+ (calcd for C36H40O12NNa+, 736.2131).
2.5 Measurement of cytotoxicity
Cell cytotoxicity was measured by using the MTT method. Briefly, cells were plated in 96-well plates (1 × 103 cells/well) for 24 h. Later, the attached cells were treated with different concentrations of compounds for 96 h. After that, the cells were incubated with 5 mg/mL MTT (Millipore) for another 4 h, and then the suspension was discarded and the dark blue crystals (formazan) were subsequently dissolved in DMSO (Millipore). The absorbance at 570 nm was measured using a FlexStation 3 multifunction microplate reader (Molecular Devices). The cell viability was final calculated as the percentage compared to the control group. Each experiment was performed at least in triplicate.
3 Results and discussion
3.1 TSN and its derivatives from isolation and modification
The starting material, TSN (5 g), was obtained from the EtOH extract of the seeds of M. toosendan. As shown in Fig. 1, 25 derivatives including 12 new ones (1, 3–5, 9, 10, 16, 17, 19, 21, 22, and 25) were obtained by simple structural modification of the natural TSN. The structures of new compounds were determined by analyzing or comparing NMR and HRMS data, and the known compounds are determined by comparing with the NMR data in the literature. Structurally, these modified derivatives could be classified into four groups: type A) 29-carbonyl derivatives of TSN (1–5), type B) non-epoxy derivatives of TSN (6–10), type C) totally-deacetyl TSN (11), and type D) 29-acyloxy derivatives of TSN (12–25).
The synthetic routes to the diverse TSN-based derivatives were presented in Scheme 1. Briefly, the functional groups or different substituents at C-1/3/7/12/29 as well as the 14,15-epoxy ring were the main structural modification sites, which were aimed to investigate their influences on the cytotoxic activity. The oxidation of TSN with Dess-Martin reagent in dichloromethane (DCM) generated four oxygenated products 2–5 (type A). Subsequently, the hydrolysis of 2 gave 3-deacetylate 1 under the condition of an excess of sodium bicarbonate (NaHCO3). Compounds 6 and 8 (type B) without the 14,15-epoxy ring were obtained by treating of TSN with diluted sulfuric acid and formic acid, respectively. Compound 10 (type B) was obtained from TSN by treating with 5-bromonicotinoyl chloride in pyridine solvent at rt. The hydrolation of TSN yielded the totally-deacetyl TSN 11 (type C) under the condition of NaHCO3. Acylation of TSN with different anhydrides (such as acetic, propionic, butyric, and valeric) or acyl chlorides (such as benzoyl, 4-fluorobenzoyl, 4-chlorobenzoyl, 4-bromobenzoyl, 4-dimethylaminobenzoyl, 4-nitrobenzoyl, 4-anisoyl, and 6-chloronicotinoyl) afforded the corresponding esters 12–15 and 18–25 (type D), respectively. While in the pyridine solvent, acylation of TSN with 2-thiophenecarbonyl chloride yielded non-epoxy derivative 7 (type B) and 29-O-2-thiophenecarbonyl ester 17 (type D), and the treatment of TSN with furoyl chloride in solvents triethylamine/DCM also gave two types of products (9 of type B and 16 of type D).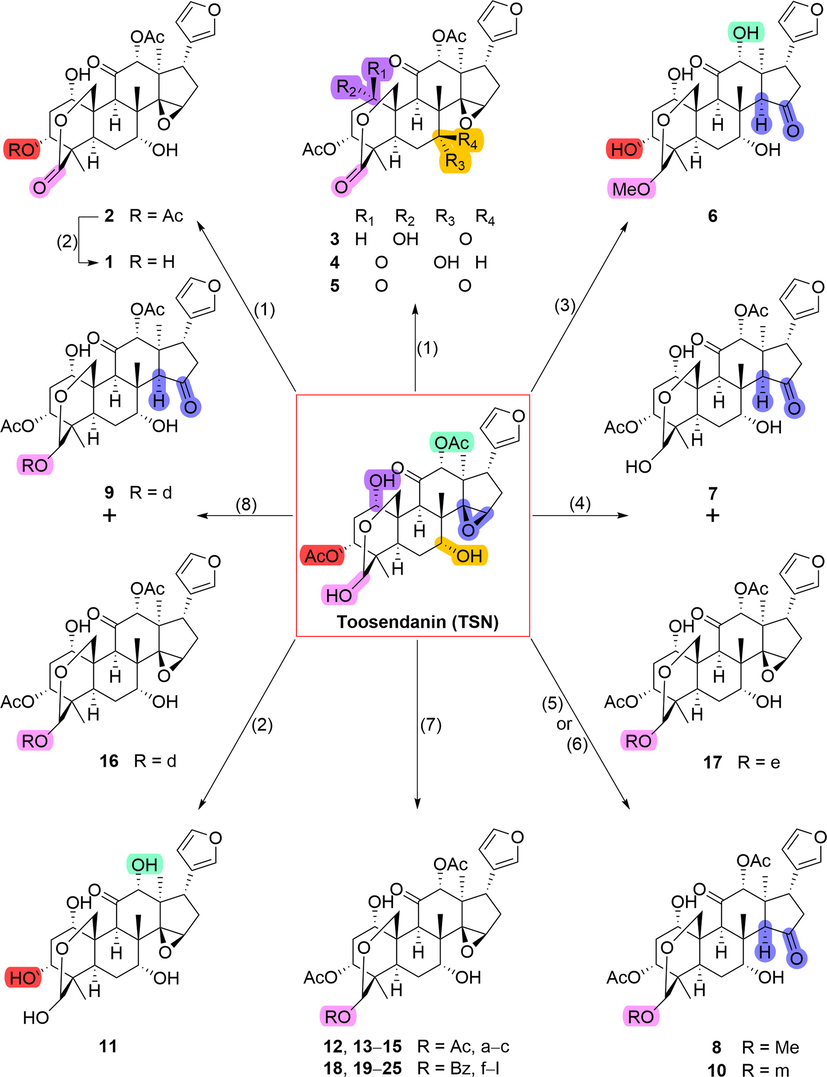
Syntheses of derivatives 1–25 from natural product TSN.a,b. aPlease see Fig. 1 for the structures of groups (Ac, Bz, and a − m). b Reagents and conditions: (1) Dess-Martin Periodinane, DCM, rt, overnight; (2) NaHCO3, MeOH, rt, overnight; (3) 1 % H2SO4, MeOH, rt, overnight; (4) 2-Thiophenecarbonyl chloride, pyridine, 0 °C, 1 h; (5) Formic acid (for 8), MeOH, 50 °C, 4 h; (6) 5-Bromonicotinoyl chloride (for 10), pyridine, rt, overnight; (7) Acetic anhydride (for 12)/propionic anhydride (for 13)/butyric anhydride (for 14)/valeric anhydride (for 15)/benzoyl chloride (for 18)/4-fluorobenzoyl chloride (for 19)/4-bromobenzoyl chloride (for 20)/4-chlorobenzoyl chloride (for 21)/4-(dimethylamino)benzoyl chloride (for 22)/4-nitrobenzoyl chloride (for 23)/4-anisoyl chloride (for 24)/6-chloronicotinoyl chloride (for 25), pyridine or DCM/Et3N (10:1), rt, overnight; (8) Furoyl chloride, DCM/Et3N (10:1), rt, overnight.
3.2 Screening for potential cytotoxicity compounds in MDA-MB-468 and Hela cells
The cytotoxicity of TSN and its derivatives (1–25) against two human cancer cell lines MDA-MB-468 and Hela was measured by the MTT method. Adriamycin, the first-line agent in cancer chemotherapy, was used as the positive drug. The bioassay results showed that the structure modifications in various positions of TSN have different influences on the activity (Fig. 2). Among the four types of TSN derivatives, none of the compounds of types A−C were active in both tested cells, while several compounds of type D had more activity than TSN or comparable effect to TSN. As shown in Fig. 2 and Table 1, the cytotoxic activity of compounds 14, 18, and 25 against MDA-MB-468 cells and compounds 14, 17–19, 21, and 25 against Hela cells was more potent than that of TSN, and five compounds exhibited comparable property to that of TSN in both cells (12, 13, 17, 20, and 21 in Hela cells; 12, 13, 16, 20, and 24 in MDA-MB-468 cells). Compound 25 was the most active TSN-derived molecule with IC50s 0.05–0.06 μM against MDA-MB-468 and Hela cells, being more significant than that of TSN and the positive control adriamycin. In addition, the active compounds 14, 17–19, 21, and 25 were selected to test their cytotoxicity in hepatic cancer cells (HepG2) and human normal breast epithelial cells (MCF10A). The results (Table S1, see Supporting Information) showed that these derivatives except for 21 had more potent than or comparable property to that of TSN. It is worth mentioning that compound 25 with the best activity against MDA-MB-468 and Hela cells also has the strongest activity against hepatic cancer cells, suggesting that it may be a broad-spectrum anticancer molecule. Moreover, all the selected compounds had significant toxicity to MCF10A cells at a concentration of 0.3 μM (Table S2, see Supporting Information), which indicated that these compounds had low even no selectivity to normal cell lines.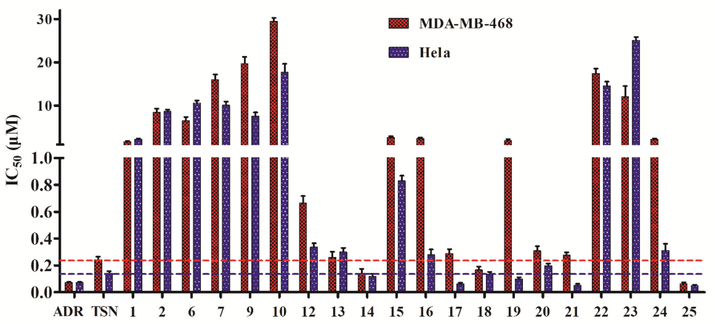
The cytotoxicity of compounds 1–25. Compounds with IC50 values > 50 μM were not shown.
Compd.
MDA-MB-468
Hela
IC50 (μM)
Foldsa
IC50 (μM)
Foldsa
1
1.68 ± 0.165
6.9 ↓
2.29 ± 0.158
16.1 ↓
2
8.48 ± 0.826
40.0 ↓
8.66 ± 0.438
60.7 ↓
3
>50.00
>206.0 ↓
>50.00
>350.0 ↓
4
>50.00
>206.0 ↓
>50.00
>350.0 ↓
5
>50.00
>206.0 ↓
>50.00
>350.0 ↓
6
6.55 ± 0.837
27.0 ↓
10.59 ± 0.579
74.3 ↓
7
16.00 ± 1.23
66.0 ↓
10.12 ± 0.858
71.0 ↓
8
>50.00
>206.0 ↓
>50.00
>350.0 ↓
9
19.74 ± 1.53
81.4 ↓
7.53 ± 0.908
52.8 ↓
10
29.47 ± 0.828
121.5 ↓
17.74 ± 1.93
124.4 ↓
11
21.85 ± 0.323
90.1 ↓
>50.00
>350.0 ↓
12
0.66 ± 0.0534
2.7 ↓
0.33 ± 0.0291
2.4 ↓
13
0.26 ± 0.0433
1.1 ↓
0.30 ± 0.0307
2.1 ↓
14
0.14 ± 0.0322
1.7 ↑
0.11 ± 0.0201
1.2 ↑
15
2.62 ± 0.312
10.8 ↓
0.83 ± 0.0378
5.8 ↓
16
2.43 ± 0.215
10.0 ↓
0.28 ± 0.0383
2.0 ↓
17
0.29 ± 0.0330
1.2 ↓
0.06 ± 0.00939
2.3 ↑
18
0.17 ± 0.0255
1.5 ↑
0.13 ± 0.0169
1.1 ↑
19
2.00 ± 0.300
8.2 ↓
0.10 ± 0.0131
1.5 ↑
20
0.31 ± 0.0354
1.3 ↓
0.20 ± 0.0149
1.4 ↓
21
0.28 ± 0.0195
1.1 ↓
0.05 ± 0.0108
2.8 ↑
22
17.40 ± 1.20
71.8 ↓
14.54 ± 0.986
102.0 ↓
23
12.06 ± 2.44
49.7 ↓
25.08 ± 0.747
176.0 ↓
24
2.22 ± 0.240
9.2 ↓
0.31 ± 0.0516
2.2 ↓
25
0.06 ± 0.00978
3.8 ↑
0.05 ± 0.00548
2.8 ↑
TSN
0.24 ± 0.0223
–
0.14 ± 0.0139
–
ADR
0.07 ± 0.00237
–
0.07 ± 0.00574
–
3.3 Structural optimization strategy and structure–activity relationship (SAR)
Structurally, TSN is a representative of tetranortriterpenoids, which is highly decorated with different oxygenated functional groups. In this study, the structural modification was deployed on the following regions, the hemiacetal, the epoxy group, the free hydroxyls at C-1 and C-7, and the acetyl groups at C-3 and C-12. According to the bioassay results, the products of the oxygenation of the hemiacetal of TSN with or without the free hydroxyls at C-1 or/and C-7 exhibited a dramatical decrease in the activity, as shown by compounds of type A (1–5) vs TSN. This indicated that the present of the hemiacetal group in TSN was essential for the activity, and 1-OH and 7-OH were also indispensable for the cytotoxic effect as the oxidation of these hydroxyls totally lost the activity (3–5 vs 1–2). The cleavage of the epoxy group in TSN also resulted in a significant decrease of the activity, as exampled by compounds of type B (6–10), suggesting that the existence of the epoxy ring was beneficial to the activity. The presence of 3-OAc and 12-OAc in TSN was favorable for the activity, as the hydrolysis of both acetyl groups led to the decrease of the activity (11 vs TSN). The collective information about the activities of compounds of types A−C suggested that 1-OH, 7-OH, 3-OAc, 12-OAc, the hemiacetal group, and the 14,15-epoxy ring were viewed as the essential active functional groups.
A detailed analysis of the bioassay data of compounds of type D showed that different kinds of substituents such as alkyls, phenyls, and aromatic heterocycles at 29-OH have different effects on the activity, which indicated that 29-OH was the key modification position for the enhancement of cytotoxicity. Among the 29-acyloxy derivatives of TSN (type D), compounds with each kind of substituents (alkyls, phenyls, and aromatic heterocycles) have one or more molecules with stronger activity than TSN.
For derivatives with alkyl alteration (12–15), the extension of carbon chain can enhance the activity in both cancer cell lines, as shown by 12 vs 13 vs 14. However, the carbon number of alkyl chain was not allowed to surpass four carbons, as the acylation of 29-OH with valeric anhydride decreased the activity (14 vs 15). Therefore, compound 14, the 29-O-butyryl TSN-based derivative, was the most active molecule in the kind of TSN derivatives with alkyls at 29-OH, whose IC50 values for MDA-MB-468 and Hela cells were 0.14 μM and 0.12 μM, respectively, being more potent than TSN. From the results of this series of compounds, it can be seen that the change of alkyls in 29-OH is a productive strategy to augment the cytotoxicity.
For phenyl series (18–24), the implement of benzoyl and different substituted benzoyls to modify 29-OH of TSN showed different certain influences on the cytotoxic activity in two cancer cell lines. In MDA-MB-468 cells model, except for the 29-O-benzoyl-TSN (18), the cytotoxicities of other compounds (19–24) with substituents on 29-O-benzoyl were all decreased in comparison with that of TSN, showing that the substituent groups in benzene ring were detrimental to the activity. Among these derivatives with different para-substituted benzoyls (19–24), the dimethylamino (see 22) and nitro (see 23) groups displayed significant adverse effect on the activity (their IC50 value with about 72- and 50-folds decreasing than TSN, respectively), while the halogen atoms like chlorine and bromine (see 20 and 21) on the para-position of benzoyl have less reducing effect. In Hela cells model, compounds 18, 19, and 21 exhibited the enhanced activity than TSN, while compounds 22 and 23 have extremely decreased the cytotoxic activity by 102- and 175-folds, respectively. It’s obvious that the substituents such as fluorine and bromine on the para-position of benzoyl were beneficial for the cytotoxic activity and the dimethylamino and nitro groups were detrimental to the activity. The chlorine or methoxy groups on the para-position of benzoyl (see 21 or 24) displayed less effect on the activity. Compounds 19 and 21 displayed the increased activity on Hela cancer cell line while exhibited the decreased effect on MDA-MB-468 cells, which suggested that they have good selectivity.
For derivatives with aromatic heterocycles (16, 17, and 25), the modification of 29-OH with aromatic heterocycles such as furan formyl, thiophene formyl, and chloronicotinoyl indicated that the 6-chloronicotinoyl group (see 25) was the most effective bioactivity fragment. Specifically, the IC50 values of 29-O-(6-chloronicotinoyl)-toosendanin (25) were 0.06 μM for MDA-MB-468 cells and 0.05 μM for Hela cells, respectively, being more than twofold stronger than TSN and comparable to the first-line drug adriamycin.
In general, we obtained a series of compounds of type D through different lead compound optimization strategies including alkyl, phenyl and aromatic heterocyclic group alteration, and explored their effects on the cytotoxicity of human cancer cell lines MDA-MB-468 and Hela. At last, we found that 29-O-butyryl-toosendanin (14) from alkyl series, 29-O-benzoyl-toosendanin (18) from phenyl series, and 29-O-(6-chloronicotinoyl)-toosendanin (25) from aromatic heterocyclic series exhibited the enhanced activity in the two tested cancer cell lines. Strikingly, compound 25 was the strongest bioactive molecule among all the activity-increased 29-OH-modified derivatives. The above-mentioned SARs information was summarized in Fig. 3.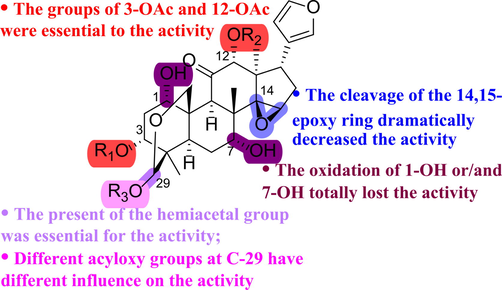
SARs of toosendanin derivatives on cytotoxicity against two cancer cell lines.
4 Conclusions
In summary, a small limonoids library containing 25 structurally diverse compounds (1–25) was constructed on the basis of the structure modification of TSN. Compounds 1, 3–5, 9, 10, 16, 17, 19, 21, 22, and 25 were 12 new derivatives. The cytotoxic assay of this library discovered that three compounds (14, 18, and 25) in MDA-MB-468 cell model and six compounds (14, 17–19, 21, and 25) in Hela cell model were more active than the precursor TSN, respectively. Among these active TSN derivatives, 25 exhibited the most potent antiproliferative activity in both cancer cell models, which was better than that of the first-line drug adriamycin. The SARs analysis indicated that the 29-OH in TSN was the key modification position for the enhancement of cytotoxicity, which may provide insight on rationally design TSN-based derivatives with more potent anti-cancer effect. This is the first report on the anticancer activity of TSN-derived compounds, which suggests that the new derivative 25 may be used as a lead compound for TSN-based anticancer chemotherapy. Whether 25 has higher bioavailability and lower hepatotoxicity than TSN (Wang et al., 2013, Lu et al., 2016) needs further research. In a word, this study may embark the converging research trend toward the intriguing anti-cancer potential of TSN-derived molecules.
Acknowledgements
This research was funded by the Guangdong Basic and Applied Basic Research Foundation, China (Nos. 2021B1515140062 and 2020A1515010841), the Southern Marine Science and Engineering Guangdong Laboratory (Zhuhai) (No. SML2021SP301), the National Natural Science Foundation of China (Nos. 81973203 and 81973195), the Key-Area Research and Development Program of Guangdong Province, China (No. 2020B1111110003), and the Open Program of Shenzhen Bay Laboratory (No. SZBL2021080601007).
References
- The potential value of amlexanox in the treatment of cancer: Molecular targets and therapeutic perspectives. Biochem. Pharmacol.. 2022;197:114895
- [CrossRef] [Google Scholar]
- Toosendanin exerts an anti-cancer effect in glioblastoma by inducing estrogen receptor β- and p53-mediated apoptosis. Int. J. Mol. Sci.. 2016;17:1928.
- [CrossRef] [Google Scholar]
- Toosendanin inhibits adipogenesis by activating Wnt/β-catenin signaling. Sci. Rep.. 2018;8:4626.
- [CrossRef] [Google Scholar]
- Impact of natural products on developing new anti-cancer agents. Chem. Rev.. 2009;109:3012-3043.
- [CrossRef] [Google Scholar]
- Molecular similarity analysis in virtual screening: foundations, limitations and novel approaches. Drug Discovery Today.. 2007;12:225-233.
- [CrossRef] [Google Scholar]
- Discovery of 8,9-seco-ent-kaurane diterpenoids as potential leads for the treatment of triple-negative breast cancer. J. Med. Chem.. 2021;64:9926-9942.
- [CrossRef] [Google Scholar]
- Limonoids from the genus Melia (Meliaceae): Phytochemistry, synthesis, bioactivities, pharmacokinetics, and toxicology. Front. Pharmacol.. 2022;12:795565
- [CrossRef] [Google Scholar]
- Bimodal modulation of the botulinum neurotoxin protein-conducting channel. Proc. Natl. Acad. Sci.. 2009;106(5):1330-1335.
- [CrossRef] [Google Scholar]
- Toosendanin inhibits hepatocellular carcinoma cells by inducing mitochondria-dependent apoptosis. Planta. Medica.. 2010;76:1447-1453.
- [CrossRef] [Google Scholar]
- Similarity and dissimilarity: A medicinal chemist’s view. In: Kubinyi H., Folkers G., Martin Y.C., eds. 3D QSAR in Drug Design. Three-dimensional quantitative structure activity relationships. Vol vol 2. Dordrecht: Springer; 2002.
- [CrossRef] [Google Scholar]
- Integrated analysis of microRNA and mRNA expression profiles highlights the complex and dynamic behavior of toosendanininduced liver injury in mice. Sci. Rep.. 2016;6:34225.
- [CrossRef] [Google Scholar]
- Toosendanin, a natural product, inhibited TGF-β1-induced epithelial-mesenchymal transition through ERK/Snail pathway. Phytother. Res.. 2018;32:2009-2020.
- [CrossRef] [Google Scholar]
- Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J. Nat. Prod.. 2020;83:770-803.
- [CrossRef] [Google Scholar]
- SAR index: Quantifying the nature of structure-activity relationships. J. Med. Chem.. 2007;50:5571-5578.
- [CrossRef] [Google Scholar]
- Cell death and apoptosis-induced effect of toosendanin. Chin. J. Neurosci.. 2004;20:461-465.
- [Google Scholar]
- Cure of experimental botulism and antibotulismic effect of toosendanin. Acta. Pharmacol. Sin.. 2004;25:839-848.
- [Google Scholar]
- Toosendanin—a presynaptic neuromuscular blocking agent. Acta. Physiol. Sin.. 1980;32:293-297.
- [Google Scholar]
- Effect of toosendanin on the sensory inputs of chemoreceptors of the armyworm larvae Mythimna separata. Atca. Entomal. Sin.. 1986;29:233-239.
- [Google Scholar]
- Meliarachins A-K: Eleven limonoids from the twigs and leaves of Melia azedarach. Helv. Chim. Acta.. 2011;94:1515-1526.
- [CrossRef] [Google Scholar]
- Determination of toosendanin in rat plasma by ultra-performance liquid chromatography-electrospray ionization-mass spectrometry and its application in a pharmacokinetic study. Biomed. Chromatogr.. 2013;27:222-227.
- [CrossRef] [Google Scholar]
- Toosendanin inhibits growth and induces apoptosis in colorectal cancer cells through suppression of AKT/GSK-3β/β-catenin pathway. Int. J. Oncol.. 2015;47:1767-1774.
- [CrossRef] [Google Scholar]
- Natural product toosendanin reverses the resistance of human breast cancer cells to adriamycin as a novel PI3K inhibitor. Biochem. Pharmacol.. 2018;152:153-164.
- [CrossRef] [Google Scholar]
- The structure of iso-chuanliansu isolated from Chinese medicine – the bark of Melia. Acta Pharm. Sin.. 1985;20(3):188-192.
- [Google Scholar]
- Natural products-based insecticidal agents 9. Design, semisynthesis and insecticidal activity of 28-acyloxy derivatives of toosendanin against Mythimna separata Walker in vivo. Bioorg. Med. Chem. Lett.. 2011;21:1974-1977.
- [CrossRef] [Google Scholar]
- Metronomic therapy in advanced breast cancer and NSCLC: vinorelbine as a paradigm of recent progress. Expert Rev. Anticancer Ther.. 2021;21(1):71-79.
- [CrossRef] [Google Scholar]
- WWOX activation by toosendanin suppresses hepatocellular carcinoma metastasis through JAK2/Stat3 and Wnt/β-catenin signaling. Cancer Lett.. 2021;513:50-62.
- [CrossRef] [Google Scholar]
- Growth inhibition and apoptosis-induced effect on human cancer cells of toosendanin, a triterpenoid derivative from Chinese traditional medicine. Invest. New Drugs.. 2005;23:547-553.
- [CrossRef] [Google Scholar]
- Combinatorial synthesis of a series of alkyl/alkenylacyloxy derivatives at the C-28 position of toosendanin as insecticidal agents. Comb. Chem. High T. SCR.. 2013;16:394-399.
- [CrossRef] [Google Scholar]
- Toosendanin demonstrates promising antitumor efficacy in osteosarcoma by targeting STAT3. Oncogene.. 2017;36:6627-6639.
- [CrossRef] [Google Scholar]
- Cytotoxic macrocyclic diterpenoids from Jatropha multifida. Bioorg. Chem.. 2018;80:511-518.
- [CrossRef] [Google Scholar]
- Anti-cancer effect of toosendanin and its underlying mechanisms. J. Asian Nat. Prod. Res.. 2019;21:270-283.
- [CrossRef] [Google Scholar]
- Jatrophane diterpenoids as modulators of P-glycoprotein-dependent multidrug resistance (MDR): Advances of structure-activity relationships and discovery of promising MDR reversal agents. J. Med. Chem.. 2016;59:6353-6369.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2022.104283.
Appendix A
Supplementary data
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







