Translate this page into:
Discovery of α-hexyl cinnamaldehyde and its derivatives as novel larvicides against Aedes albopictus
⁎Corresponding author at: School of Pharmacy and Food Engineering, Wuyi University, Jiangmen 529020, China. zjsheng@wyu.edu.cn (Zhaojun Sheng) wyuchemszj@126.com (Zhaojun Sheng)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Mosquitoes are the vectors of numerous diseases such as malaria, Dengue and Zika. Controlling mosquito vectors is essential for preventing and managing diseases transmitted by mosquitoes. Larvicides target larvae within their breeding habitats before they mature into adult mosquitoes and disperse. In this study, using α-hexyl cinnamaldehyde (HCA) as a lead compound, three series of derivatives comprising a total of 78 compounds were designed and synthesized. The larvicidal activities of these compounds against Aedes albopictus were evaluated, resulting in the identification of ten promising compounds. Among them, furan-3-ylmethyl cinnamate (compound 2v) and (E)-allyl 3-(4-chlorophenyl)acrylate (compound 4c) exhibited the lowest LC50 values (8.0 ppm and 7.2 ppm respectively). In a 96-h acute toxicity assay conducted on zebrafish, 2v showed no or very low toxicity with an LC50 higher than 100 ppm. To elucidate their mode of action, morphological and histopathologic analyses were performed, and the effects of the compounds on the acetylcholinesterase (AChE) and phosphatase activities were evaluated. Results suggested that the active HCA derivatives induce injury to intestinal epithelial cells by inhibiting phosphatase activity, ultimately resulting in the mortality of mosquito larvae.
Keywords
Larvicide
Cinnamaldehyde
Mosquito
Insecticide
Morphology
1 Introduction
Mosquitoes are capable of transmitting numerous deadly and debilitating diseases, thus being regarded as the world's deadliest animals. Aedes albopictus (Skuse, 1894) (Diptera: Culicidae), commonly known as the Asian Tiger mosquito, are extensively found in tropical and subtropical regions, being a major vector for diseases such as Dengue, Chikungunya and yellow fever. (Tolle, 2009; Kraemer et al., 2019) Due to the lack of effective vaccines, the prevention and control of these diseases primarily depend on the control of mosquito vectors.
The life cycle of Ae. albopictus includes eggs, larvae, pupae, and adults. Insecticide treatments primarily target the larval and adult stages. At present, larvicides registered by the USA Environmental Protection Agency (EPA) can be classified into three categories: 1) microbial insecticides such as Bacillus thuringiensis and Bacillus sphaericus; 2) insect growth inhibitors such as S-hydroprene and methoprene; 3) organophosphate insecticides such as temephos. (EPA, n.d.) However, these larvicides have their limitations or drawbacks. For instance, the environmental stability of microbial insecticides needs improvement. (Chattopadhyay et al., 2017; Moustafa, 2018) The limitations of insect growth inhibitors include slow mode of action, stage specificity, and difficulty in monitoring their efficacy. (Singh, 2013) Although organophosphate insecticides like temephos are highly effective, insecticide resistance is progressively increasing due to the extensive and prolonged use. (Francis, 2020) Moreover, temephos exhibits genotoxic effects and adverse effects on mammalian male reproduction and fertility, as well as causing liver damage, even at low doses. (Martínez-Mercado, 2022) Therefore, the discovery and development of new chemical larvicides remain an important area of focus.
The potential for plant extracts such as essential oils (EOs) to act as a natural alternative for controlling mosquito populations has garnered considerable interest in recent years. (Rodrigues et al., 2020) In our previous work, the larvicidal activity of 53 EOs and 26 constituents derived from EOs were screened. (Li, 2023; Sheng et al., 2020) α-Hexyl cinnamaldehyde (HCA) from green tea EO showed the highest activity, with an LC50 of 24.0 ppm. In this study, to develop more potent and novel larvicides, HCA as a lead compound was employed and three series of HCA derivatives were designed by chemical modification at four sites of HCA (Fig. 1). These target compounds were synthesized through several steps. Their larvicidal activity against Ae. albopictus and toxicity against zebrafish were assessed, and the mode of action was investigated.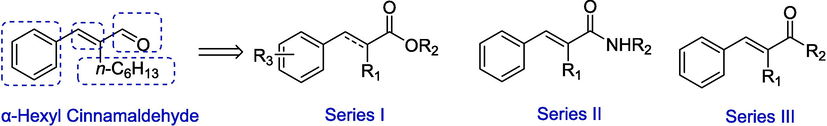
Structures of three series of HCA derivatives.
2 Material and methods
2.1 Instruments and reagents
All reagents were of analytical reagent grade (purity > 98 %) and were used without additional purification steps. Zebrafish (AB strain) was procured from Nanjing Pear Blossom Biotech Co., Ltd., and ACP and ALP assay kits were obtained from Solarbio (Beijing, China). Cinnamaldehyde (7a), α-methyl cinnamaldehyde (7b), α-pentyl cinnamaldehyde (7d) and α-hexyl cinnamaldehyde (7e) were purchased from Bidepharm (Shanghai, China).
The derivatives of HCA were structurally characterized using nuclear magnetic resonance (NMR) (Bruker AVANCE NEO 500 MHz spectrometer) and high-performance liquid chromatography-mass spectrometry (HPLC-MS) (Thermo Fisher Q Exactive).
2.2 Synthesis procedures for compounds in scheme 1
2.2.1 Procedure for the synthesis of 7c
To synthesize of α-propyl cinnamaldehyde (7c), benzaldehyde (2.12 g, 20 mmol) was added to 45 mL of EtOH in an ice bath, followed by the addition of n-pentanal (3.45 g, 40 mmol). Afterward, 2 mL of NaOH (0.80 g, 20 mmol) were added slowly drop-by-drop, and the mixture was stirred overnight at room temperature. The reaction was monitored by thin-layer chromatography (TLC) (petroleum ether/EtOAc = 10/1). EtOH was removed under reduced pressure, and the residue was dissolved in 50 mL of EtOAc. The organic phase was washed with H2O (100 mL) and a saturated solution of NaCl in water (brine) (50 mL), then dried over Na2SO4 and evaporated. The crude product was chromatographed (petroleum ether/EtOAc = 80/1) to obtain pure α-propyl cinnamaldehyde (7c) (2.5 g, yield: 35 %; Rf value = 0.26).
2.2.2 General procedure for the synthesis of 1a-1t and 2a-2ab
Intermediate 7a-7e (9 mmol) was dissolved in 20 mL tetrahydrofuran (THF). Afterward, 2-methyl-2-butene (10.52 g, 135 mmol) and 5 mL of an aqueous solution of NaH2PO4 (5.11 g, 36 mmol) were added. The mixture was stirred for 5 min in an ice bath. Next, 5 mL of an aqueous solution of NaClO2 (3.05 g, 27 mmol) was added dropwise. The reaction mixture changed from colorless to yellow. Stirring was continued at room temperature for 5 h. The reaction was monitored with TLC (petroleum ether/EtOAc = 5/1), until the reaction mixture became colorless·THF was removed under reduced pressure, and the residue was dissolved in 50 mL of EtOAc. The pH of the solution was adjusted to 9–10 with 1 M NaOH (2–3 mL). The mixture was extracted with H2O (100 mL), and the aqueous phase was collected. The pH of the aqueous phase was adjusted to 3–4 with 1 M HCl (1–2 mL), and then extracted with EtOAc (100 mL). The organic phase was dried over Na2SO4, filtered, and concentrated to yield the crude product, which was purified by recrystallization from dichloromethane (DCM) to obtain intermediate 8a-8e (yield: 76 %-88 %).
Intermediate 8a-8e (3.3 mmol) was dissolved in 15 mL DCM. 1-Ethyl-3-(3-dimethyl aminopropyl)carbodiimide hydrochloride (EDCI) (0.69 g, 3.6 mmol) and 4-dimethylaminopyridine (DMAP) (0.44 g, 3.6 mmol) were added. Next, the corresponding alcohol (3 mmol) was added. The mixture was stirred at room temperature for 6–8 h. The reaction solution was washed with H2O (15 mL), brine (15 mL), dried over Na2SO4, filtered, and concentrated to yield the crude product, which was purified by column chromatography (petroleum ether/EtOAc = 200/1 to 50/1) to obtain the final compounds 1a-1 t (yield: 66 %-94 %) and 2a-2ab (yield: 31 %-96 %).
2.2.3 General procedure for the synthesis of 3a-3g and 4a-4k
3-Phenylpropanoic acid or cinnamic acid derivatives (3 mmol) was dissolved in 15 mL of DCM. Then, EDCI (0.69 g, 3.6 mmol) and DMAP (0.44 g, 3.6 mmol) were added. Next, the corresponding alcohol (3 mmol) was added and the mixture was stirred at room temperature for 6–8 h. The reaction solution was washed with H2O (15 mL) and brine (15 mL), dried over Na2SO4, filtered, and concentrated to yield the crude product, which was purified by column chromatography (petroleum ether/EtOAc = 200/1 to 50/1) to obtain the final compounds 3a-3 g (yield: 72 %-92 %) and 4a-4 k (yield: 72 %-95 %).
2.2.4 General procedure for the synthesis of 5a-5e
α-Hexyl cinnamic acid 8e (0.26 g, 1.1 mmol) was dissolved in 15 mL DCM. 1H-benzotriazol-1-yloxytripyrrolidinophosphonium hexafluorophosphate (PyBOP) (0.63 g, 1.2 mmol) and triethylamine (TEA) (0.30 g, 3 mmol) were added, and stirred for 10 min in an ice bath. Then, the corresponding amine (1 mmol) was added. The mixture was stirred at room temperature for 6–8 h. The reaction solution was washed with H2O and brine. The organic phase was dried over Na2SO4, filtered and concentrated to yield the crude product, which was purified by column chromatography (petroleum ether/EtOAc = 50/1) to obtain the final compounds 5a-5e (yield: 65 %-82 %).
2.2.5 General procedure for the synthesis of 6a-6g
Magnesium (0.15 g, 6 mmol) and a small crystal of iodine were added to 5 mL of anhydrous THF under argon. The bromide (4 mmol) was dissolved in 10 mL of anhydrous THF and then added dropwise to the flask. The reaction was initiated by heating the mixture with a heat gun to near boiling point. Next, the reaction mixture was refluxed for 1 h at 60 °C. After cooling to room temperature, cinnamaldehyde (7a) or α-methyl cinnamaldehyde (7b) or α-hexyl cinnamaldehyde (7e) (3.2 mmol) was added in an ice bath. The mixture was stirred for 2–3 h. The reaction was quenched by the addition of 2 mL saturated NH4Cl solution. Then the mixture was filtered, concentrated under reduced pressure, and dissolved in 20 mL of EtOAc. The organic phase was washed with H2O (20 mL) and brine (20 mL), and dried over Na2SO4, filtered and concentrated to yield the crude product, which was purified by column chromatography (petroleum ether/EtOAc = 20:1) to obtain the intermediate 9a-9 g (yield: 55 %-79 %).
9a-9 g (1.5 mmol) was dissolved in 5 mL THF, followed by the addition of Dess-Martin periodinane (DMP) (0.70 g, 1.65 mmol). The reaction mixture was stirred at room temperature for 2 h. After that, THF was evaporated under reduced pressure. The residue was dissolved in EtOAc (20 mL), and washed with saturated NaHCO3 (20 mL), H2O (20 mL), and brine (20 mL). The organic phase was dried over Na2SO4, filtered and concentrated to yield the crude product, which was purified by column chromatography (petroleum ether/EtOAc = 100/1) to obtain the final compounds 6a-6 g (yield: 41 %-80 %).
2.3 Mosquitoes
All mosquitoes were reared in the insectary of Liverpool-Jiangmen Public Health Research Centre of the International Healthcare Innovation Institute (Jiangmen), China. Mosquitoes were maintained at 26 ± 2 °C, with a relative humidity of 70 ± 5 % and a photoperiod of 14 h light:10 h dark. Larvae were fed daily with fish food, and adults were fed with 5 % glucose solution. Fourth instar larvae were selected for use in the bioassays. The resistance level of Ae. albopictus resistant strain to pyrethroids was assessed using the Bottle bioassay method recommended by the CDC (CDC, n.d.) and was determined to be of moderate intensity according to WHO guidelines (WHO, n.d.). Resistance ratios (RRs) were calculated by dividing the LC50 value of the pyrethroid-resistant strain Ae. albopictus by that of the sensitive strain. (Silalahi, 2022) The RR value of the pyrethroid-resistant strain used in this study for deltamethrin was 8.1.
2.4 Larvicidal assay
The larvicidal activities of HCA derivatives were assessed using the immersion method recommended by the WHO, (WHO, n.d.) with few modifications. First, 10 mg of test compound were dissolved in 2 mL of acetone to prepare a stock solution. Subsequently, 1 mL of the stock solution was added to 99 mL of distilled water to obtain a 50 mg/L (or ppm) solution, which was then further diluted to the required concentrations (1–20 ppm) for the test solutions. Next, 20 Ae. albopictus larvae were exposed to each test solution, with no feeding allowed during the test period. After 24 h of exposure, mortality was determined by gently touching the larvae. If there was no physiological activity, the larva was considered dead. Additionally, a blank control consisting of 1 % (v/v) acetone in distilled water was used. Three replicates were conducted for each sample at each concentration. In cases where the blank control showed 3–10 % mortality, adjusted mortality was calculated using Eq. (1) (Abbott, 1925).
2.5 Zebrafish toxicity assay
In this work, the acute toxicity of compounds 2v and 4c against zebrafish was assessed according to OECD Guidelines (OECD, 2019) and the national standard (GB/T 31270.12–2014) of the People's Republic of China. First, a stock solution of 2 g of test compound was prepared in 1 mL MeOH. Then, 50 μL of the stock solution were added to 1 l deionized water to obtain a 100 mg/L (or ppm) solution, which was further diluted to the required concentrations (0.001–10 ppm) for the test solutions. Ten zebrafish of consistent size, aged 2 months, were exposed to the test solution. Their behavior and mortality were observed at 6, 24, 48, 72, and 96 h. Fish that showed no movement when gently touched on the tail were considered dead and promptly removed. The number of dead fish was recorded after 96 h of exposure.
2.6 Enzyme assays
2.6.1 Enzyme extraction
First, mosquito larvae were exposed to HCA derivatives at the corresponding LC50 for 24 h. Next, twenty live fourth instar larvae were collected for ultrasonic homogenization in 0.6 mL of ice-cold 0.1 M sodium phosphate buffer (pH 8.0). The homogenate was then centrifuged at 17000 × G and 4 °C for 15 min, and the resulting supernatant was used as the enzyme source for all enzyme assays. The protein concentration in the supernatant was determined using the Bradford protein assay. In the control group, fourth instar larvae were exposed to 1 % acetone for 24 h, then collected, homogenized, centrifuged, and the supernatant was used for protein quantitative determination.
2.6.2 AChE assay
The AChE assay was performed according to the standard procedures recommended by the WHO, (WHO, n.d.) with slight modifications. The enzyme solution was first diluted to a concentration of 0.05 μg/μL in 0.1 M sodium phosphate buffer (pH 8.0). Next, 80 μL diluted enzyme solution containing 4 μg protein was added to each well of a 96-well microplate and incubated at 28 °C for 10 min. Following this, 10 μL of 4 mM 5,5′-dithiobis(2-nitrobenzoic acid) and 10 μL of 10 mM acetylthiocholine iodide were added to each well, and the final mixture was incubated at room temperature for 30 min. The change in absorbance (ΔOD412) of the mixture was then measured at 412 nm without heating using a microplate reader (Thermo Fisher Scientific Multiskan GO, Finland). All experiments were conducted in triplicate. The enzyme activity assay was calculated using Eq. (2) (WHO, n.d.), where ε is the molar absorption coefficient (13.6 l/(mmol·cm)), P (μg/μL) is the protein concentration of the enzyme solution, V (μL) is the volume of the mixture, L (cm) is the absorption path length, T is the reaction time of the mixture, and Ve (μL) is the volume of the enzyme solution.
2.6.3 Acid and alkaline phosphatase assays
The activities of acid and alkaline phosphatase (ACP and ALP) were assessed using assay kits (BC2135 and BC2145, Beijing Suolaibao Technology Co. LTD). First, a phenol standard solution with a concentration of 10 μmol/mL was diluted to 2.5 μmol/mL using distilled water, and kept as a stocking solution.
Four groups were established, including test group, blank group, standard group and control group. In the test group, 20 μL of enzyme solution, 40 μL of reagent 1 (the main component is disodium benzene phosphate), and 40 μL of reagent 2 (the main component is 4-aminoantipyrine) were sequentially added into a 96-well plate and incubated at 37 °C for 15 min. Then, 120 μL of reagent 3 (the main component is potassium ferricyanide) was added, and the absorbance was measured at 510 nm. In the control group, 40 μL of reagent 1 and 40 μL of reagent 2 were added to the 96-well plate, and then incubated at 37 °C for 15 min. Following this, 120 μL of reagent 3 and 20 μL of enzyme solution were added before measuring the absorbance at 510 nm. In the blank group, 20 μL of distilled water were used instead of the enzyme solution. In the standard group, 20 μL of 2.5 μmol/mL of the phenol standard solution were used as a substitute for the enzyme solution. Finally, the ACP and ALP activities were calculated by Eq. (3), where Atest is the absorbance value of the test group, Ablank is the absorbance value of the blank group, Astandard is the absorbance value of the standard group, Acontrol is the absorbance value of the control group, and Cprotein is the protein concentration of the enzyme solution.
2.7 Morphology and histopathology study
The morphology was studied according to the method reported by Fia et al. (Fiaz, 2019); with few modifications. First, the fourth instar larvae of Ae. albopictus were exposed to solutions of compound 2v, 4c, and temephos at their corresponding LC50 concentrations. After 24 h of exposure, the larvae were collected and fixed in 4 % paraformaldehyde for immersion and morphological studies under a stereomicroscope (Leica/M205FA, 0.5:1 zoom ratio, 7.8 × -160 × magnification range (1 × objective lens, 10 × eyepiece)). Next, both the control and treated larvae were dehydrated using a series of graded alcohols (70 %, 80 %, 95 % and 100 %) and embedded in paraffin. The larvae were then sectioned at a thickness of 5 μm using a microtome (Leica/RM2235) to obtain gut sections. These sections were stained with hematoxylin and eosin (HE) and evaluated under the stereomicroscope.
2.8 Statistical analysis
The mortality rate of Ae. albopictus larvae was calculated using Probit analysis in SPSS 25.0 software (SPSS Inc., Chicago, IL, USA) to determine the LC50 and LC90 values, along with their respective 95 % confidence intervals. The enzyme activities of AChE, ACP and ALP were analyzed using t-test in GraphPad Prism 8.0 (San Diego, CA, USA), with statistical significance set at P < 0.05.
3 Results and discussion
3.1 Synthesis
The synthetic routes to the target compounds are outlined in Scheme 1. There are three series of derivatives: ester derivatives (Series I), amide derivatives (Series II), and ketone derivatives (Series III).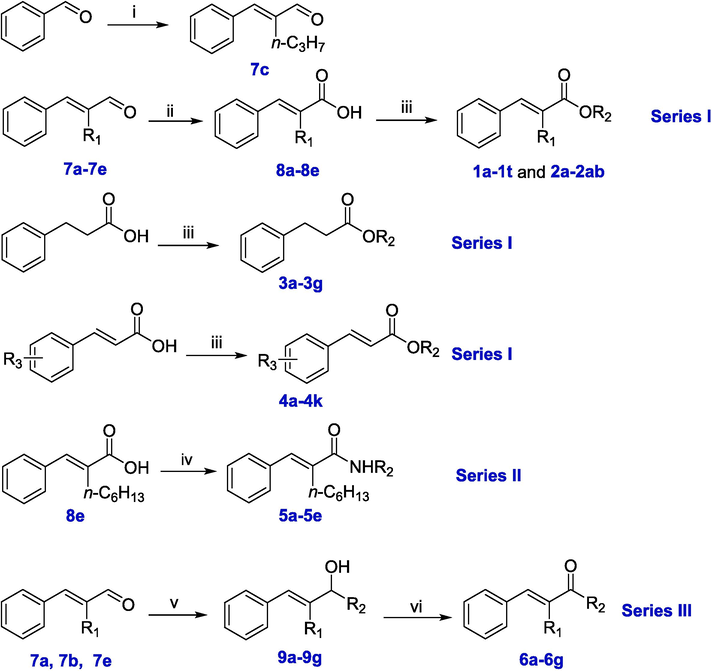
Synthesis of α-hexyl cinnamaldehyde derivatives.
Reagents and conditions: i. n-C4H9CHO, EtOH, NaOH, rt., overnight. ii. 2-methyl-2-butene, NaClO2, NaH2PO4, THF, rt., 5 h. iii. R2OH, EDCI, DMAP, DCM, rt., 6–8 h. iv. R2NH2, PyBOP, DCM, rt., 6–8 h. v. R2MgBr, I2, THF, 0 °C, 2–3 h. vi. DMP, THF, rt., 2 h.
The compounds 7a-7b and 7d-7e are commercially available whereas 7c (α-propyl cinnamaldehyde) was synthesized by condensing benzaldehyde with n-pentanal followed by elimination. Aldehydes 7a-7e were oxidized to carboxylic acids 8a-8e by Pinnick oxidation. Then, acids 8a-8e or 3-phenylpropanoic acid or other cinnamic acid derivatives were condensed with various alcohols in the presence of EDCI and DMAP, resulting in the first series: 1a-1 t, 2a-2ab, 3a-3 g and 4a-4 k (Table 1). Acid 8e was used as a starting compound and was coupled with various amines in the presence of PyBOP, yielding the second series: 5a-5e (Table 1). The nucleophilic addition of aldehyde 7a or 7b or 7e with various Grignard reagents yielded intermediates 9a-9 g. The hydroxyl group of 9a-9 g was selectively oxidized by DMP to carbonyl, thereby yielding the third series: 6a-6 g (Table 1).
No.
R1
R2
No.
R1
R2
No.
R2
R3
1a
CH3
n-C3H7
2i
H
4a
ortho-Cl

1b
CH3

2j
H
4b
meta-Cl

1c
CH3
n-C5H11
2k
H
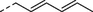
4c
para-Cl

1d
CH3
Bn
2l
H
n-C8H17
4d
2,6-dichloro

1e
CH3

2m
H

4e
3,4-dichloro

1f
n-C3H7
n-C3H7
2n
H
Ph
4f
para-Cl
C2H5
1g
n-C3H7

2o
H
Bn
4g
para-Cl
n-C3H7
1h
n-C3H7
n-C5H11
2p
H

4h
para-Cl
i-C3H7
1i
n-C3H7
Bn
2q
H
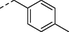
4i
para-Cl
n-C4H9
1j
n-C3H7
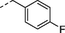
2r
H
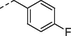
4j
para-Cl

1k
n-C5H11
n-C3H7
2s
H
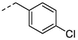
4k
para-Cl
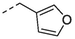
1l
n-C5H11

2t
H
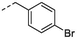
1m
n-C5H11
n-C5H11
2u
H
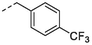
No.
R2
5a
n-C3H7
1n
n-C5H11
Bn
2v
H
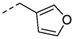
5b

1o
n-C5H11
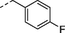
2w
H
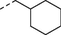
5c
n-C5H11
1p
n-C6H13
n-C3H7
2x
H

5d
Bn
1q
n-C6H13

2y
H
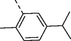
5e
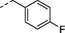
1r
n-C6H13
n-C5H11
2z
H
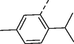
1s
n-C6H13
Bn
2aa
H
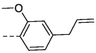
No.
R1
R2
6a
H
n-C3H7
1t
n-C6H13
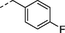
2ab
H
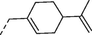
6b
CH3
n-C3H7
2a
H
CH3
No.
R2
6c
n-C6H13
n-C3H7
2b
H
C2H5
3a
n-C2H5
6d
n-C6H13

2c
H
n-C3H7
3b
n-C3H7
6e
n-C6H13
n-C5H11
2d
H

3c

6f
n-C6H13
Bn
2e
H
i-C3H7
3d
i-C3H7
6g
n-C6H13
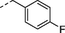
2f
H
n-C4H9
3e
n-C4H9
2g
H

3f

2h
H
n-C5H11
3g
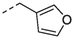
3.2 Larvicidal activity
First, the larvicidal activities of all compounds, at concentrations of 20 ppm and 50 ppm, against sensitive Ae. albopictus were assessed using the larval immersion method. As shown in Table 2, out of 78 compounds, 19 resulted in 100 % mortality at 50 ppm, while 10 led to 100 % mortality at 20 ppm. It’s worth noting that all the active compounds are ester derivatives of HCA.
No.
Mortality (%)
No.
Mortality (%)
No.
Mortality (%)
20 ppm
50 ppm
20 ppm
50 ppm
20 ppm
50 ppm
1a
17 ± 2
100 ± 0
2 h
25 ± 4
95 ± 4
3g
0 ± 0
15 ± 4
1b
43 ± 2
100 ± 0
2i
8 ± 2
95 ± 4
4a
100 ± 0
100 ± 0
1c
17 ± 2
95 ± 0
2j
0 ± 0
48 ± 2
4b
100 ± 0
100 ± 0
1d
30 ± 0
93 ± 2
2k
33 ± 2
95 ± 4
4c
100 ± 0
100 ± 0
1e
5 ± 0
77 ± 2
2l
0 ± 0
28 ± 2
4d
100 ± 0
100 ± 0
1f
2 ± 2
82 ± 2
2m
0 ± 0
22 ± 2
4e
15 ± 4
95 ± 4
1g
40 ± 4
98 ± 2
2n
10 ± 4
47 ± 5
4f
100 ± 0
100 ± 0
1h
0 ± 0
22 ± 2
2o
35 ± 4
95 ± 4
4g
100 ± 0
100 ± 0
1i
2 ± 2
22 ± 2
2p
10 ± 0
38 ± 2
4h
100 ± 0
100 ± 0
1j
0 ± 0
12 ± 2
2q
15 ± 4
20 ± 0
4i
100 ± 0
100 ± 0
1k
2 ± 2
72 ± 2
2r
32 ± 2
88 ± 5
4j
100 ± 0
100 ± 0
1l
2 ± 2
80 ± 0
2s
20 ± 0
75 ± 4
4k
23 ± 2
92 ± 2
1m
0 ± 0
13 ± 2
2t
0 ± 0
0 ± 0
5a
0 ± 0
7 ± 2
1n
0 ± 0
12 ± 2
2u
0 ± 0
60 ± 4
5b
5 ± 0
27 ± 2
1o
0 ± 0
12 ± 2
2v
100 ± 0
100 ± 0
5c
0 ± 0
3 ± 2
1p
2 ± 2
35 ± 4
2w
2 ± 2
95 ± 4
5d
0 ± 0
0 ± 0
1q
8 ± 2
78 ± 2
2x
0 ± 0
12 ± 2
5e
0 ± 0
0 ± 0
1r
0 ± 0
0 ± 0
2y
0 ± 0
3 ± 2
6a
10 ± 0
83 ± 5
1s
0 ± 0
0 ± 0
2z
0 ± 0
7 ± 2
6b
13 ± 2
100 ± 0
1t
0 ± 0
0 ± 0
2aa
0 ± 0
0 ± 0
6c
0 ± 0
63 ± 2
2a
3 ± 2
85 ± 4
2ab
0 ± 0
3 ± 2
6d
18 ± 2
65 ± 0
2b
0 ± 0
93 ± 2
3a
0 ± 0
3 ± 2
6e
0 ± 0
5 ± 0
2c
22 ± 2
100 ± 0
3b
0 ± 0
18 ± 5
6f
0 ± 0
2 ± 2
2d
45 ± 4
100 ± 0
3c
20 ± 4
100 ± 0
6g
0 ± 0
0 ± 0
2e
18 ± 5
100 ± 0
3d
0 ± 0
3 ± 2
HCA
27 ± 3
100 ± 0
2f
45 ± 4
100 ± 0
3e
0 ± 0
42 ± 2
2g
78 ± 2
100 ± 0
3f
0 ± 0
72 ± 2
Next, the 10 most active compounds were selected for further bioassays. Larval mortalities were assessed at different concentrations, and their LC50 and LC90 values are shown in Table 3. Comparative to the lead compounds, these 10 derivatives showed lower LC50 and LC90 values, indicating heightened larvicidal activities. Compound 4c exhibited the lowest LC50 value (7.2 ppm), followed by 2v (8.0 ppm) and 4i (8.8 ppm).
Comp.
Sensitive Ae. albopictus
Resistant Ae. albopictus
LC50 (ppm)
95 % CI (ppm)
LC90 (ppm)
95 % CI (ppm)
LC50 (ppm)
95 % CI (ppm)
LC90 (ppm)
95 % CI (ppm)
2v
8.0
7.7–8.2
11.2
10.7–11.8
18.3
16.3–20.1
27.3
24.2–33.7
4a
9.7
9.4–10.0
13.3
12.8–14.0
NT*
NT
NT
NT
4b
9.3
8.4–9.9
13.6
12.6–15.3
NT
NT
NT
NT
4c
7.2
6.5–7.9
9.2
8.2–12.0
10.8
10.3–11.3
13.8
12.9–15.4
4d
8.8
8.0–9.6
12.5
11.2–14.8
NT
NT
NT
NT
4f
11.1
10.2–12.0
15.7
14.2–18.2
NT
NT
NT
NT
4g
10.2
9.7–10.7
14.3
13.3–16.1
NT
NT
NT
NT
4h
11.2
10.9–11.5
15.6
14.8–16.6
NT
NT
NT
NT
4i
8.8
7.8–9.6
12.0
10.8–14.9
18.4
17.5–19.3
32.9
30.1–36.7
4j
9.0
8.4–9.6
12.6
11.5–14.4
NT
NT
NT
NT
HCA10
24.0
21.2–26.5
34.0
30.4–41.5
NT
NT
NT
NT
Except for the sensitive strain of Ae. albopictus, the larvicidal activity of compounds 2v, 4c, and 4i against the pyrethroid-resistant strain of Ae. albopictus was also evaluated. The LC50 values were 18.3 ppm, 10.8 ppm, and 18.4 ppm, respectively. Their RRs ranged from 1.5 to 2.3, indicating that the pyrethroid-resistant strain is comparatively sensitive to these HCA derivatives.
Cinnamic acid and cinnamaldehyde are common components in natural products, and their application in the control of mosquito vectors has been widely reported. (Fujiwara, 2017; Pavela, 2015) Moreover, several cinnamic acid or cinnamaldehyde derivatives have been reported in recent years. Araujo et al. (Araújo, 2021) synthesized seventeen cinnamic acid derivatives and tested their larvicidal activity against Ae. aegypti. The most potent compound was pentyl cinnamate with a LC50 value of 36 ppm. Franca et al. (Bezerra França, 2021) prepared several n-substituted ethyl cinnamates and tested their larvicidal activity against Ae. aegypti after 48 h of exposure. Ethyl p-chlorocinnamate showed the lowest LC50 value (8.3 ppm). Naresh et al. (Naresh, 2022) synthesized fourteen cinnamic acid thiadiazole derivatives and tested their larvicidal activity against Ae. aegypti. After 24 h of exposure, the most active compound was CA2 with an LC50 value of 82.2 ppm. N’goka et al. (N’goka, 2023) reported the larvicidal and ovicidal activities of some cinnamaldehyde derivatives against Anopheles Gambiae after 24 h of exposure. o-Nitrocinnamaldehyde exhibited the highest activity with an LC50 value of 55 ppm. Our study thus supplements the database of cinnamic acid or cinnamaldehyde derivatives. Moreover, the derivatives in our study show more potent larvicidal activity against mosquitoes than other reports.
3.3 Zebrafish toxicity
Given that mosquito larvicides are often applied directly to water, it's essential to consider their impact on fish safety. As a small vertebrate model organism, zebrafish offers advantages such as short experimental cycle, low testing cost, and adherence to the international “3R” principle of animal welfare protection. It has been applied in environmental toxicology since as early as around 1930. (Hill, 2005) In this study, the 96-hour acute toxicity of compounds 2v and 4c to zebrafish was evaluated. The LC50 value of compound 2v was higher than 100 ppm, indicating no or very low toxicity to zebrafish. In contrast, the LC50 value of compound 4c was 3.5 ppm, indicating its high toxicity to zebrafish.
3.4 Structure-activity relationship (SAR) analysis
Based on the results of the larvicidal activity and toxicity to zebrafish, the SAR of HCA derivatives can be summarized as shown in Fig. 2. Firstly, introducing chlorine into the benzene ring significantly enhances the larvicidal activity but also increases toxicity to fish. Secondly, the presence of the double bond of HCA is essential; replacing it with a single bond markedly reduces larvicidal activity. Thirdly, among the ester derivatives of HCA (Series I) higher activity was observed compared to the amide (Series II) and ketone derivatives (Series III). Within several R2 substituents, the allyl group exhibited high preference. Fourthly, in the α-position, a hydrogen atom is notably preferable over other substituents such as hexyl and amyl.
Summary of SAR for HCA derivatives.
3.5 Morphology study
To elucidate the mode of larvicidal action of these active derivatives, their effects on larval morphology were examined. As shown in Fig. 3, compared to control larvae (Fig. 3A), those exposed to 2v and 4c exhibited grossly damaged digestive tracts, appearing dim. Moreover, the cervix region, linking the caput with the thorax, was elongated (Fig. 3B and 3C). In contrast, larvae exposed to temephos displayed a markedly different phenotype (Fig. 3D), characterized by shrinkage and short stature. Temephos is known to induce larval death primarily by inhibiting AChE, an important and widely studied target of insecticides like organophosphates and carbamates. AChE hydrolyzes acetylcholine neurotransmitters, thereby terminating nerve impulses in the insects nervous system. (Liu, 2015) The morphology analysis suggests that the mode of action of HCA derivatives differs from that of temephos.
Photomicrographs of the whole body of Ae. albopictus larvae. A: control larva; B: larva exposed to 2v; C: larva exposed to 4c; D: larva exposed to temephos.
For further analysis, the larvae exposed to 2v and control larvae were fixed and embedded in paraffin. After, the blocks were sectioned into 5 μm slices. HE staining was performed for microscopic analysis. As shown in Figs. 3 and 4, the abdomen constitutes the major structure of the larvae, with the digestive tract being the largest organ. The basic structure and functions of the intestine are conserved between different species, with the intestine playing a key role in digestion nutrient absorption. (Suzuki, 2020) The intestinal epithelial cells are crucial for controlling intestinal barrier function and nutrient absorption. Ciliated epithelial cells and goblet cells were clearly observed in the intestine of mosquito larvae. (Hixson et al., 2021) After 24 h exposed to 2v, the epithelial cells near the thorax exhibited vacuolation, and a large number of apoptotic bodies of epithelial cells were observed around the bottom of the abdomen. The dysfunction of intestinal epithelial cells resulted in the entry of bacteria and toxins, leading to systemic inflammation. (Ghosh, 2020) Based on these results, it is conceivable that intestinal epithelial cells are one of the targets of 2v, leading to the death of mosquito larvae.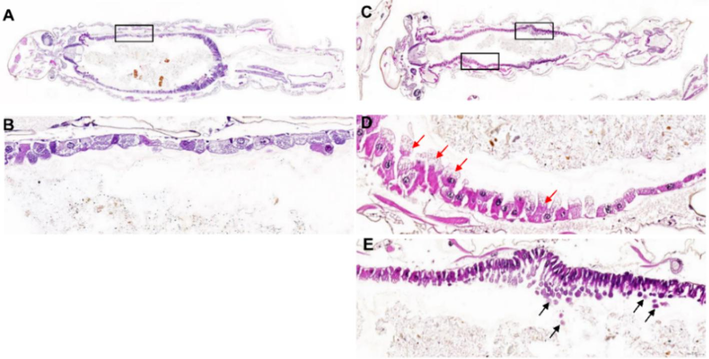
Microscopic images of Ae. albopictus larvae. A-B: control larva; C-E: larva exposed to 2v; red arrow indicates epithelial cells with vacuolation, black arrow indicates these cells with apoptotic bodies.
3.6 Enzyme assays
To further investigate the mechanism of action of these HCA derivatives, the activities of AChE, ACP and ALP in the larvae, exposed for 24 h to a solution of HCA derivatives at their LC50, were evaluated.
As mentioned above, AChE is an important target of nerve agents like temephos. First, the AChE inhibitory activity of 10 active derivatives (2v, 4a-4d, and 4f-4j) was studied. As shown in Fig. 5, although these compounds exhibited some inhibition of AChE at their LC50 concentrations, the inhibitory rates were generally low. This suggests that AChE might not be the main target of these compounds, which is consistent with the morphological results.
AChE inhibitory activity of 10 active compounds.
Phosphatases are enzymes that hydrolyze phosphoric acid monoesters into a phosphate ion and an alcohol. They are classified into two types based on their optimum pH: acid phosphatases (ACP) and alkaline phosphatases (ALP). (Koodalingam, 2012) ALP is widely distributed in several tissues of insects, including the digestive tract, storage tissue, nervous tissue, parts of the reproductive system, certain muscles, and some glands. (Day, 1949) Among these tissues, the midgut exhibits higher ALP activity, (Miao, 2002) playing a crucial role in digestion and nutrient supplying throughout the insect’s body. (Parthiban et al., 2020) The activities of ACP and ALP are closely linked to key physiological processes such as metabolism and cell signaling. (Koodalingam, 2012) Ismail et al. (Ismail, 2020) reported that increased activities of ACP and ALP are associated with the development of resistance in Culex quinquefasciatus. Wang et al. (Wang, 2011) also reported the effects of ALPs on insect resistance to organophosphate insecticides. Thus, phosphatases have been extensively used as accurate and sensitive biomarkers for assessing the toxicity of insecticides. (Koodalingam, 2012; Koodalingam et al., 2011).
In this study, the ACP and ALP activities of mosquito larvae exposed to the 10 most active HCA derivatives at LC50 were also assessed. The results are shown in Fig. 6 and Fig. 7. These HCA derivatives reduced ACP activity by 12–41 % compared to the control, and reduced ALP activity by 14–45 %. Compounds 2v and 4i exhibited the most significant reductions in ACP activity, with 40.5 % and 40.7 % inhibition, respectively. Compound 4j induced the greatest decrease in the ALP activity, with 45.2 % inhibition. Based on the results of enzyme inhibition assays and morphological observation, it can be reasonably inferred that these HCA derivatives disrupt the physiological balance in the digestive tracts by inhibiting ALP and ACP activities, ultimately resulting in larval death.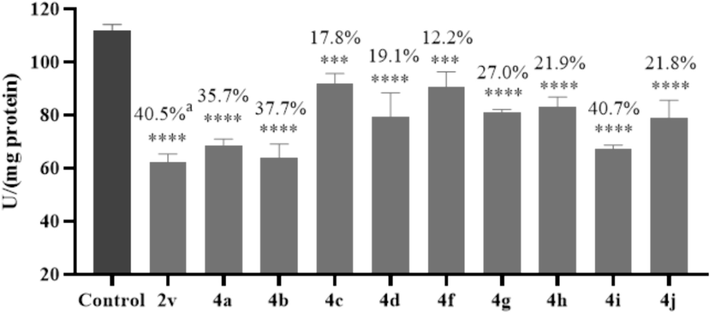
ACP inhibitory activity of 10 active compounds.

ALP inhibitory activity of 10 active compounds.
4 Conclusions
In this study, 78 HCA derivatives were synthesized and characterized by NMR and HRMS, among which ten compounds exhibited significant larvicidal activity against Ae. Albopictus, achieving 100 % mortality at 50 ppm and 20 ppm. Notably, compounds 2v and 4i demonstrated remarkable potency, with LC50 values of 8.0 ppm and 7.2 ppm, respectively, representing an approximately three-fold increase compared to the lead compound HCA. Importantly, compound 2v exhibited no or very low toxicity to zebrafish, indicating its safety for aquatic organisms. Although there have been lots of reports on the mosquito larvicidal activity of plant extracts or synthesized insecticides, mechanism study are often lacked. Our mechanistic studies suggest that these HCA derivatives exert their larvicidal effects through the inhibition of phosphatase activities and damage to intestinal epithelial cells, in contrast to the mode of action of the conventional larvicide temephos. Our study has supplemented the database of cinnamic acid or cinnamaldehyde derivatives, meanwhile provides SAR information and several promising candidates for the development of novel mosquito larvicides. More efforts are devoted to the further improvement of their efficacy and the results will be reported in due course.
CRediT authorship contribution statement
Yuling Li: Conceptualization, Investigation, Methodology, Writing – original draft. Jialun Zou: Data curation, Investigation, Methodology. Xiaofei Ren: Investigation, Methodology. Wenjiao Li: Investigation. Weifeng Wu: Data curation, Investigation. Qiwei Xuan: Investigation. Hanwen Miao: Investigation. Yajie Chen: Methodology, Supervision. Chi-Sing Lee: Supervision. Zhaojun Sheng: Conceptualization, Funding acquisition, Methodology, Project administration, Resources, Supervision, Writing – original draft, Writing – review & editing.
Acknowledgments
The authors gratefully acknowledge Wuyi University-Hong Kong and Macao Joint Research Fund (No. 2021WGALH10), Science and Technology Planning Project of Guangdong Province (2021A1515110916), Innovation team program supported by Guangdong Province (2020KCXTD038), and Project of Innovative and Entrepreneurship for College Students in Wuyi University (No. 202211349209, 202211349340, and 2022CX17) for the financial support. The authors thank Dr. John S. Lomas for his help in correcting the English of this article.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- A Method of Computing the Effectiveness of an Insecticide. J. Econ. Entomol.. 1925;18(2):265-267.
- [Google Scholar]
- Larvicidal activity of cinnamic acid derivatives: Investigating alternative products for Aedes aegypti L. control. Molecules. 2021;26(1):61.
- [Google Scholar]
- Larvicidal activity and in silico studies of cinnamic acid derivatives against Aedes aegypti (Diptera: Culicidae) Bioorg. Med. Chem.. 2021;44:116299
- [Google Scholar]
- CDC, Guideline for Evaluating Insecticide Resistance in Vectors Using the CDC Bottle Bioassay, https://stacks.cdc.gov/view/cdc/21777..
- Recent trends of modern bacterial insecticides for pest control practice in integrated crop management system. 3 Biotech. 2017;7(1):60.
- [Google Scholar]
- The distribution of alkaline phosphatase in insects. Aust. J. Biol. Sci.. 1949;2(1):31-41.
- [Google Scholar]
- Pyriproxyfen, a juvenile hormone analog, damages midgut cells and interferes with behaviors of Aedes aegypti larvae. PeerJ. 2019;7:e7489.
- [Google Scholar]
- Comparative toxicity of larvicides and growth inhibitors on Aedes aegypti from select areas in Jamaica. R. Soc. Open Sci.. 2020;7(3):192041
- [Google Scholar]
- Evaluation of larvicidal activity and ecotoxicity of linalool, methyl cinnamate and methyl cinnamate/linalool in combination against Aedes aegypti. Ecotoxicol. Environ. Saf.. 2017;139:238-244.
- [Google Scholar]
- Intestinal barrier dysfunction, LPS translocation, and disease development. J. Endocr. Soc.. 2020;4(2):bvz039.
- [Google Scholar]
- Zebrafish as a model vertebrate for investigating chemical toxicity. Toxicol. Sci.. 2005;86(1):6-19.
- [Google Scholar]
- Midgut epithelial dynamics are central to mosquitoes’ physiology and fitness, and to the transmission of vector-borne disease. Front. Cell. Infect. Microbiol.. 2021;11
- [Google Scholar]
- The effect of entomopathogenic fungi on enzymatic activity in chlorpyrifos-resistant mosquitoes, Culex quinquefasciatus (Diptera: Culicidae) J. Med. Entomol.. 2020;57(1):204-213.
- [Google Scholar]
- Effect of a Bt-based product (Vectobar) on esterases and phosphatases from larvae of the mosquito Aedes aegypti. Pestic. Biochem. Physiol.. 2012;104(3):267-272.
- [Google Scholar]
- Effects of extract of soapnut Sapindus emarginatus on esterases and phosphatases of the vector mosquito, Aedes aegypti (Diptera: Culicidae) Acta Trop.. 2011;118(1):27-36.
- [Google Scholar]
- Kraemer, M.U.G., Reiner, R.C., Brady, O.J.,nMessina, J.P., Gilbert, M., Pigott, D.M., Yi, D., Johnson, K., Earl, L., Marczak, L.B., Shirude, S., Davis Weaver, N., Bisanzio, D., Perkins, T.A., Lai, S., Lu, X., Jones, P., Coelho, G.E., Carvalho, R.G., Van Bortel, W., Marsboom, C., Hendrickx, G., Schaffner, F., Moore, C.G., Nax, H.H., Bengtsson, L., Wetter, E., Tatem, A.J., Brownstein, J.S., Smith, D.L., Lambrechts, L., Cauchemez, S. Linard, C., Faria, N.R., Pybus, O.G., Scott, T.W., Liu, Q., Yu, H., Wint, G.R.W., Hay, S.I., Golding, N., Past and future spread of the arbovirus vectors Aedes aegypti and Aedes albopictus. Nature Microbiology, 2019. 4(5): p. 854-863.
- Larvicidal, acetylcholinesterase inhibitory activities of four essential oils and their constituents against Aedes albopictus, and nanoemulsion preparation. J. Pest. Sci.. 2023;96(3):961-971.
- [Google Scholar]
- Insecticide resistance in mosquitoes: Impact, mechanisms, and research directions. Annu. Rev. Entomol.. 2015;60(1):537-559.
- [Google Scholar]
- Temephos, an organophosphate larvicide for residential use: A review of its toxicity. Crit. Rev. Toxicol.. 2022;52(2):113-124.
- [Google Scholar]
- Studies on the activity of the alkaline phosphatase in the midgut of infected silkworm, Bombyx mori L. J. Appl. Entomol.. 2002;126(2–3):138-142.
- [Google Scholar]
- Influence of some environmental conditions on stability and activity of Bacillus thuringiensis formulations against the cotton leaf worm, Spodoptera littoralis (Boisd.) (Lepidoptera: Noctuidae) Egypt. J. Biol. Pest Control. 2018;28(1):61.
- [Google Scholar]
- N’goka, V., et al., Larvicidal and Ovicidal Activities of Some Cinnamaldehyde Derivatives against Anopheles Gambiae, Malaria Vector Agent. American Journal of Chemistry, 2023. 13(1): p. 1-10.
- Larvicidal and histopathological efficacy of cinnamic acid analogues: A novel strategy to reduce the dengue vector competence. RSC Adv.. 2022;12(16):9793-9814.
- [Google Scholar]
- OECD, Test No. 203: Fish, Acute Toxicity Test. 2019.
- Annona muricata: An alternate mosquito control agent with special reference to inhibition of detoxifying enzymes in Aedes aegypti. Ecotoxicol. Environ. Saf.. 2020;189:110050
- [Google Scholar]
- Essential oils for the development of eco-friendly mosquito larvicides: A review. Ind. Crop. Prod.. 2015;76:174-187.
- [Google Scholar]
- Larvicidal efficacy of plant extracts and isolated compounds from Annonaceae and Piperaceae against Aedes aegypti and Aedes albopictus. Asian Pac. J. Trop. Med.. 2020;13(9):384-396.
- [Google Scholar]
- Screening of larvicidal activity of 53 essential oils and their synergistic effect for the improvement of deltamethrin efficacy against Aedes albopictus. Ind. Crop. Prod.. 2020;145:112131
- [Google Scholar]
- Insecticide resistance profiles and synergism of field Aedes aegypti from Indonesia. PLoS Negl. Trop. Dis.. 2022;16(6):e0010501.
- [Google Scholar]
- Insect growth regulators: practicle use, limitations and future. J. Eco-Friendly Agric.. 2013;8(1):1-14.
- [Google Scholar]
- Regulation of the intestinal barrier by nutrients: The role of tight junctions. Anim. Sci. J.. 2020;91(1):e13357.
- [Google Scholar]
- Mosquito-borne diseases. Curr. Probl. Pediatr. Adolesc. Health Care. 2009;39(4):97-140.
- [Google Scholar]
- Characterization of soluble and membrane-bound alkaline phosphatase in Nilaparvata lugens and their potential relation to development and insecticide resistance. Arch. Insect Biochem. Physiol.. 2011;78(1):30-45.
- [Google Scholar]
- WHO, Test procedures for insecticide resistance monitoring in malaria vector mosquitoes..
- WHO, Guidelines for laboratory and field testing of mosquito larvicides, WHO/CDS/WHOPES/GCDPP/2005.13..
- WHO, Techniques to detect insecticide resistance mechanisms (Field and laboratory manual), WHO/CDS/CPC/MAL/98.6..
Appendix A
Supplementary material
1H NMR and 13C NMR spectra of final compounds are available in the supporting information. Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2024.105873.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary Data 1
Supplementary Data 1







