Translate this page into:
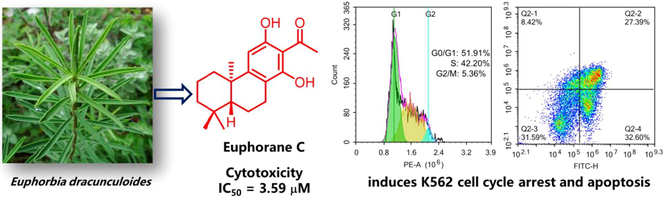
Euphorane C, an unusual C17-norabietane diterpenoid from Euphorbia dracunculoides induces cell cycle arrest and apoptosis in human leukemia K562 cells
⁎Corresponding authors. emmalao@163.com (Wu-Ling Liu), lshangg@163.com (Shang-Gao Liao)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Abstract
Four new polycyclic diterpenoids euphoranes A − D (1–4), together with 12 known terpenoids (5–16) were isolated from the whole plants of Euphorbia dracunculoides. Compound 2 represents the first example of aromatic 15,16,17-trinorabietane diterpenoid in Euphorbia species, and euphorane C (3) is an unusual 17-norabietane diterpenoid. The absolute configurations of 1, 2, and 4 were determined by single crystal X-ray diffraction, while that of 3 was established with the aid of ECD and 1D NMR quantum chemical calculation. Moreover, the absolute stereochemistry of 11 was demonstrated for the first time by crystallographic data. Compounds 1–16 were screened for antiproliferative activities against five human cancer cell lines, and 3 showed significant cytotoxicity against the human leukemia K562 cells with IC50 value of 3.59 μM. Preliminary mechanistic investigation revealed that 3 could induce cell cycle arrest at G0/G1 phase and apoptosis in K562 cells.
Keywords
Euphorbia dracunculoides
Cytotoxicity
Apoptosis
Abietane diterpenoid
Norabietane
1 Introduction
Natural products are important sources of “privileged structures” in drug development (Cragg et al., 2009). For anti-cancer agents, about 70 % of FDA-approved antitumor drugs are either natural products or their derivatives (Newman et al., 2020). Among them, the nonspecific “higher diterpenes” (also known as polycyclic diterpenoids) with a common 6/6/6 ring system from Euphorbia species are of particular significance due to their extensive physiological activities. Polycyclic diterpenoids are well-known for their structural diversity with many different skeletal types, such as rosanes, abietanes, atisanes, and kauranes (Shi et al., 2008; Vasas et al., 2014; Xu et al., 2021). Abietane diterpenoids from Euphorbia are usually substituted by an α,β-unsaturated γ-lactone ring at C-12 and C-13. Kauranes comprise a perhydrophenanthrene unit (A, B, and C rings) fused with a cyclopentane (D ring), while in atisanes, the perhydrophenanthrene unit is fused with a cyclohexane (Vasas et al., 2014). Recent studies have shown that polycyclic diterpenoids may be potential chemical entities in future antitumor drug development. For example, jolkinolide B, an abietane diterpene, could induce apoptosis and sensitize bladder cancer to mTOR inhibitors (Sang et al., 2021; Sang et al., 2022). 8,9-Seco-ent-kaurene, a kaurane diterpene, could effectively suppress triple-negative breast cancer cells via Akt signaling pathway (Fan et al., 2021).
Euphorbia dracunculoides, a perennial herb of the genus Euphorbia, is mainly distributed in North Africa, South Europe, and Southwest Asia (Wang et al., 2015a). Previous phytochemical studies showed that the plant is rich in diterpenes with different skeletal types, such as lathyranes, myrsinanes, and tiglianes, etc. (Chi et al., 2016; Dai et al., 2016; Wang et al., 2015a, 2015b, 2016, 2017). In our ongoing efforts to discover antitumor diterpenoids from Euphorbia plants (Chen et al., 2022; Yan et al., 2019), four new polycyclic diterpenoids (1–4) and 12 known compounds were isolated from the EtOAc fraction of E. dracunculoides. Compounds 2 and 3 represent unusual examples of aromatic abietane norditerpenoids in Euphorbia species. All the compounds were tested for antiproliferative activities against five human cancer cell lines, and 3 showed significant activity against the human leukemia K562 cells with micromolar IC50. Herein, we reported the isolation, structural identification, and cytotoxicity of these compounds.
2 Material and methods
2.1 General information
Optical rotations were tested on a Rudolph Autopol I automatic polarimeter. UV spectra were tested on a Shimadzu UV-2450 spectrophotometer, and IR spectra were recorded on Bruker Tensor 37 infrared spectrophotometers. ECD spectra were measured on an Applied Photophysics Chirascan spectrometer. NMR data were measured on Bruker AM-400/500 spectrometers at 25 °C. HRESIMS was performed on a Waters Micromass Q-TOF spectrometer. Column chromatography (CC) was performed on silica gel (Particle size 37–48 μm, Qingdao Haiyang Chemical Co., ltd.), reversed-phase C18 (RP-C18) (S-50 μm, 12 nm, YMC Co., ltd.), D101 macroreticular resin (Polystyrene, non-polar, particle size 0.315–0.7 mm, pore diameter 9–15 nm, surface area 500–550 m2/g, Donghong Chemical Co., ltd.), and Sephadex LH-20 gel (Amersham Biosciences). A Shimadzu LC-20 AT equipped with a SPD-M20A PDA detector was used for HPLC. An YMC-pack ODS-A column (250 × 10 mm, S-5 μm, 12 nm) was used for semi-preparative HPLC separation. All solvents were analytical grade (Guangzhou Chemical Reagents Company, ltd.).
2.2 Plant material
The whole plant of Euphorbia dracunculoides Lam. (Euphorbiaceae) were collected in July 2020 from Dali city, Yunnan Province, China and identified by one of the authors, Qing-De Long. A voucher specimen (number: HZDJ202007) was deposited at the School of Pharmacy, Guizhou Medical University.
2.3 Extraction and isolation
The air-dried powder of E. dracunculoides (20 kg) was extracted with 95 % EtOH (3 × 50 L) at room temperature to give a crude extract (3 kg), which was suspended in H2O (3 L) and then partitioned with EtOAc. The resulting EtOAc extract (800 g) was subjected to D101 macroreticular resin eluted with a MeOH/H2O gradient (30:70 → 100:0) to afford four fractions (Fr. I − Fr. IV). Fr. II was separated by silica gel chromatographic column (CC) (CH2Cl2/MeOH, 200:1 → 10:1) to give three fractions (Fr. IIa − Fr. IIc). Fr. IIc was recrystallized in MeOH to yield 8 (1.03 g). Fr. III was subjected to silica gel CC eluted with a petroleum ether (PE)/EtOAc gradient (10:1 → 1:3) to give three fractions (Fr. IIIa − Fr. IIIc). Fr. IIIa was separated by RP-C18 silica gel CC eluted with MeOH/H2O (50:50 → 100:0) to give Fr. IIIa1 and Fr. IIIa2. Fr. IIIa2 was separated by Sephadex LH-20 (MeOH), followed by semipreparative HPLC (MeCN/H2O, 70:30, 3 mL/min) to afford 15 (11 mg, tR 8.4 min) and 11 (31 mg, tR 7.3 min). Fr. IIIc was separated by Sephadex LH-20 (MeOH) followed by semipreparative HPLC (MeCN/H2O, 75:25, 3 mL/min) to give 12 (8 mg, tR 12.3 min), 14 (9 mg, tR 14.1 min), and 16 (10 mg, tR 14.5 min). Fr. IV (140 g) was separated by silica gel CC (PE/EtOAc) to give five fractions (Fr. IVa − Fr. IVe). Fr. IVa was separated by RP-C18 silica gel CC followed by HPLC (MeCN/H2O, 75:25, 3 mL/min) to yield 1 (38 mg, tR 14.5 min) and 13 (5 mg, tR 10.1 min). Fr. IVb was separated by RP-C18 silica gel CC (MeOH/H2O, 60:40 → 90:10) to give Fr. IVb1 and Fr. IVb2. Fr. IVb1 was purified by HPLC (MeCN/H2O, 90:10, 3 mL/min) to give 3 (30 mg, tR 12.0 min). Fr. IVb2 was purified by HPLC (MeCN/H2O, 70:30, 3 mL/min) to afford 2 (24 mg, tR 13.3 min) and 7 (20 mg, tR 16.0 min). Fr. IVd was separated by silica gel CC to give Fr. IVd1 and Fr. IVd2. Fr. IVd1 was separated by HPLC (MeCN/H2O, 80:20, 3 mL/min) to give 4 (11 mg, tR 11.8 min) and 5 (5 mg, tR 8.1 min). Fr. IVd2 was purified by HPLC (MeCN/H2O, 70:30, 3 mL/min) to afford 6 (15 mg, tR 12.1 min). Fr. IVe was separated by silica gel CC (CH2Cl2/MeOH, 200:1 → 10:1), Sephadex LH-20 (MeOH), followed by HPLC (MeCN/H2O, 80:20, 3 mL/min) to afford 10 (8 mg, tR 15.5 min) and 9 (10 mg, tR 12.5 min).
2.3.1 14β-Hydroxy-ent-abieta-13(15)-ene-12α,16-olide (euphorane A) (1)
Colorless needles; mp 190.7–193.6 °C; [α]20D − 80.8 (c 0.28, MeOH); UV (MeCN) λmax (log ε) 221 (4.13) nm; IR (KBr) νmax 3421, 2924, 1742, 1684, 1476, 1366, 1261, 1226, 1088, 1034, 832, and 735 cm−1; 1H and 13C NMR data see Tables 1 and 2, respectively; HRESIMS m/z 339.1920 [M + Na]+ (calcd for C20H28O3Na, 339.1931).
Position
1a
2b
3c
4c
1
0.97, dd (13.1, 4.0)
β 1.32, m
β 1.35, m
0.96, md
1.83, m
α 2.18, dt, (13.3, 3.9)
α 2.08, d (12.2)
1.51, m
2
1.46, md
1.58, m
1.45, m
1.82, m
1.59, m
1.75, dt (13.3, 3.3)
1.62, md
3
1.17, m
1.22, m
1.13, ddd (17.0, 13.4, 3.7)
3.44, m
1.45, md
1.46, dt (13.1, 3.5)
1.37, m
5
1.26, dd (12.1, 4.7)
1.25, m,
1.23, dd (12.9, 2.0)
0.82, m,
6
α 1.98, m
α 1.60, m
α 1.59, md
1.53, m
β 2.15, m
β 1.88, dd (13.1, 7.8)
β 1.86, dd (12.9, 8.0)
7
5.99, s
α 2.75, dd (17.3, 6.0)
α 3.05, dd (17.5, 6.1)
α 2.47, dt (13.3, 3.2)
β 2.43, ddd (17.3, 11.8, 7.8)
β 2.70, ddd (17.5, 11.7, 8.0)
β 0.88, m
8
9
2.42, m
1.46, m
11
β 1.55, md
6.25, d (2.3)
6.70, s
1.75, m
α 2.29, ddd (13.5, 6.2, 2.8)
12
4.58, dd (11.8, 6.2)
2.87, dd (6.2, 2.9)
13
6.10, d (2.3)
5.41, d (2.9)
14
5.05, s
15
2.33, m
16
2.95, s
17
1.97, s
4.88, dd (4.0, 2.3)
5.05, dd (4.0, 2.3)
18
0.91, s
0.95, s
0.91, s
1.20, s
19
0.88, s
0.92, s
0.86, s
0.96, s
20
0.86, s
1.16, s
1.18, s
0.73, s
Position
1a
2b
3c
4c
1
38.7
40.3
39.0
37.0
2
18.5
20.4
19.9
28.1
3
42.1
42.9
42.1
78.4
4
32.8
34.4
34.0
39.6
5
49.5
51.7
50.3
55.0
6
23.7
19.8
19.2
19.6
7
129.6
25.1
24.6
31.7
8
135.8
114.7
115.1
48.4
9
44.1
153.3
159.8
51.9
10
36.2
38.9
39.0
38.4
11
27.0
103.3
102.9
26.0
12
78.3
156.4
160.1
44.2
13
123.7
100.3
109.3
75.6
14
70.5
156.4
163.1
212.6
15
162.3
205.9
43.9
16
174.9
33.9
143.9
17
9.1
110.6
18
33.0
33.8
33.7
29.3
19
21.8
22.1
22.2
16.8
20
13.1
25.2
24.7
14.3
2.3.2 12,14-Dihydroxy-15,16,17-trinorabieta-8,11,13-triene (euphorane B) (2)
Colorless crystals; mp 176.8–179.0 °C; [α]20D − 45 (c 1.5, MeOH); UV (MeCN) λmax (log ε) 202 (3.63) nm; IR (KBr) νmax 3306, 2925, 1593, 1456, 1310, 1132, 1041, 1012, and 838 cm−1; 1H and 13C NMR data see Tables 1 and 2, respectively; HRESIMS m/z 259.1707 [M − H]− (calcd for C17H23O2, 259.1704).
2.3.3 12,14-Dihydroxy-17-norabieta-8,11,13-triene-15-one (euphorane C) (3)
Yellowish oil; [α]20D − 40.5 (c 0.2, MeOH); UV (MeCN) λmax (log ε) 276 (3.19) and 350 (2.42) nm; ECD (9.9 × 10−4 mol/L, MeCN) λmax (Δε) 230 (+3.08), 275 (−1.33), 350 (+0.37) nm; IR (KBr) νmax 3255, 2926, 1622, 1585, 1416, 1269, 1073, 1001, and 831 cm−1; 1H and 13C NMR data see Tables 1 and 2, respectively; HRESIMS m/z 325.1784 [M + Na]+ (calcd for C19H26O3Na, 325.1774).
2.3.4 13β-Acetoxy-3α-hydroxy-ent-atis-16-ene-14-one (euphorane D) (4)
Colorless crystals; mp 145.0–148.6 °C; [α]20D + 17.5 (c 0.4, MeOH); UV (MeCN) λmax (log ε) 203 (3.46) nm; IR (KBr) νmax 3442, 2930, 1746, 1729, 1370, 1230, 1055, 1033, 975, 946, 898, and 735 cm−1; 1H and 13C NMR data see Tables 1 and 2, respectively; HRESIMS m/z 383.2205 [M + Na]+ (calcd for C22H32O4Na, 383.2193).
2.4 ECD and 1D NMR calculations
The structure of 3 was studied by quantum chemical calculation. Details of calculation procedure was provided in the Supplementary Material.
2.5 Crystallographic data
Euphorane A (1) was recrystallized in EtOH to give colorless crystals. Euphorane B (2) was recrystallized in PE/CH2Cl2 (5:1) to give colorless crystals. Euphorane D (4) was recrystallized in PE/EtOAc (4:1) to give colorless crystals. (23E)-25-methoxycycloart-23-en-3β-ol (11) was recrystallized in MeOH to afford colorless needles. Their X-ray crystallographic data have been deposited at the Cambridge Crystallographic Data Centre, CCDC Numbers 2184857 (1), 2184871 (2), 2184873 (4), and 2184858 (11).
2.5.1 Euphorane A (1)
C20H28O3 (M = 316.42 g/mol): monoclinic, space group P21 (no. 4), a = 10.0198(8) Å, b = 7.9319(6) Å, c = 10.6903(5) Å, β = 97.363(6)°, V = 842.62(10) Å3, Z = 2, T = 100.00(10) K, μ(Cu Kα) = 0.648 mm−1, Dcalc = 1.247 g/cm3, 16479 reflections measured (8.34° ≤ 2Θ ≤ 157.626°), 3418 unique (Rint = 0.1437, Rsigma = 0.0824) which were used in all calculations. The final R1 was 0.0715 (I > 2σ(I)) and wR2 was 0.1897 (all data). Flack parameter = 0.1(3).
2.5.2 Euphorane B (2)
C17H26O3 (M + H2O = 278.38 g/mol): monoclinic, space group P21 (no. 4), a = 12.5386(3) Å, b = 7.8476(2) Å, c = 15.9368(4) Å, β = 96.696(2)°, V = 1557.45(7) Å3, Z = 4, T = 99.99(10) K, μ(Cu Kα) = 0.631 mm−1, Dcalc = 1.187 g/cm3, 30,951 reflections measured (5.584° ≤ 2Θ ≤ 154.208°), 6420 unique (Rint = 0.1043, Rsigma = 0.0721) which were used in all calculations. The final R1 was 0.0523 (I > 2σ(I)) and wR2 was 0.1492 (all data). Flack parameter = − 0.11(14).
2.5.3 Euphorane D (4)
C22H32O4 (M = 360.47 g/mol): monoclinic, space group P21 (no. 4), a = 7.42590(10) Å, b = 11.16680(10) Å, c = 23.2313(3) Å, β = 90.0980(10)°, V = 1926.42(4) Å3, Z = 4, T = 99.99(10) K, μ(Cu Kα) = 0.668 mm−1, Dcalc = 1.243 g/cm3, 38,225 reflections measured (3.804° ≤ 2Θ ≤ 144.254°), 6856 unique (Rint = 0.0797, Rsigma = 0.0561) which were used in all calculations. The final R1 was 0.0448 (I > 2σ(I)) and wR2 was 0.1261 (all data). Flack parameter = 0.07(14).
2.5.4 (23E)-25-methoxycycloart-23-en-3β-ol (11)
C31H52O2 (M = 456.72 g/mol): monoclinic, space group P21 (no. 4), a = 12.8731(3) Å, b = 7.2205(2) Å, c = 14.8704(3) Å, β = 90.868(2)°, V = 1382.05(6) Å3, Z = 2, T = 100.00(10) K, μ(Cu Kα) = 0.496 mm−1, Dcalc = 1.098 g/cm3, 27759 reflections measured (5.944° ≤ 2Θ ≤ 158.296°), 5618 unique (Rint = 0.1236, Rsigma = 0.0701) which were used in all calculations. The final R1 was 0.0571 (I > 2σ(I)) and wR2 was 0.1532 (all data). Flack parameter = − 0.1(3).
2.6 Cell culture
The human cancer cell lines HEL (erythroleukemia), K562 (chronic myelogenous leukemia), PC3 (prostatic cancer), MCF-7 (breast cancer), and HepG2 (liver cancer) were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA) and maintained in RPMI 1640 (HyClone) containing 5 % fetal bovine serum (GIBCO) at 37 °C under a humidified 5 % CO2 atmosphere.
2.7 Cytotoxicity assay
Cytotoxicity of compounds 1–16 against cancer cell line was assessed using MTT method. 5 × 103 Cells per well were seeded into 96-well plates. After 24 h, cells were treated with tested compounds at different concentrations for 72 h. Then, cells were cultured with MTT for 4 h. After removing the supernatant, DMSO was added to dissolve the formazan crystals. The absorbance values were measured at 490 nm. The IC50 values of indicated compounds were determined based on the relative survival curves.
2.8 Apoptosis analysis
For apoptosis measurement, K562 cells (5 × 105/well) were seeded into 6-well plates and treated with different concentrations of 3 (4 μM and 8 μM) or DMSO (control) for 24 h and 48 h. After being washed by pre-cooled PBS, cells were double stained using FITC-Annexin V/PI Apoptosis Detection Kit (BD Biosciences, NJ, USA) and analyzed on flow cytometry according to manufacturer guideline.
2.9 Cell cycle analysis
To assess cell cycle, K562 cells (5 × 105/well) were seeded into 6-well plates and treated with different concentrations of 3 (4 μM and 8 μM) or DMSO (control) for 12 h. After being collected and washed with pre-cooled PBS, cells were permeabilized and fixed in paraformaldehyde containing 0.1 % triton X-100. Then, cells were washed twice with pre-cooled PBS, followed by incubation with the mixture of Ribonuclease A (RNaseA, Takara Bio, Beijing, China) and propidium iodide (PI, BD Biosciences, NJ, USA) in the dark for 30 min at 37 °C. Finally, cell cycle was analyzed by FACS Calibur flow cytometer (BD Biosciences, NJ, USA).
3 Results and discussion
The whole plants of E. dracunculoides were extracted with 95 % EtOH and then partitioned with EtOAc. The obtained EtOAc extract was separated by various column chromatographic methods to afford compounds 1–16 (Fig. 1).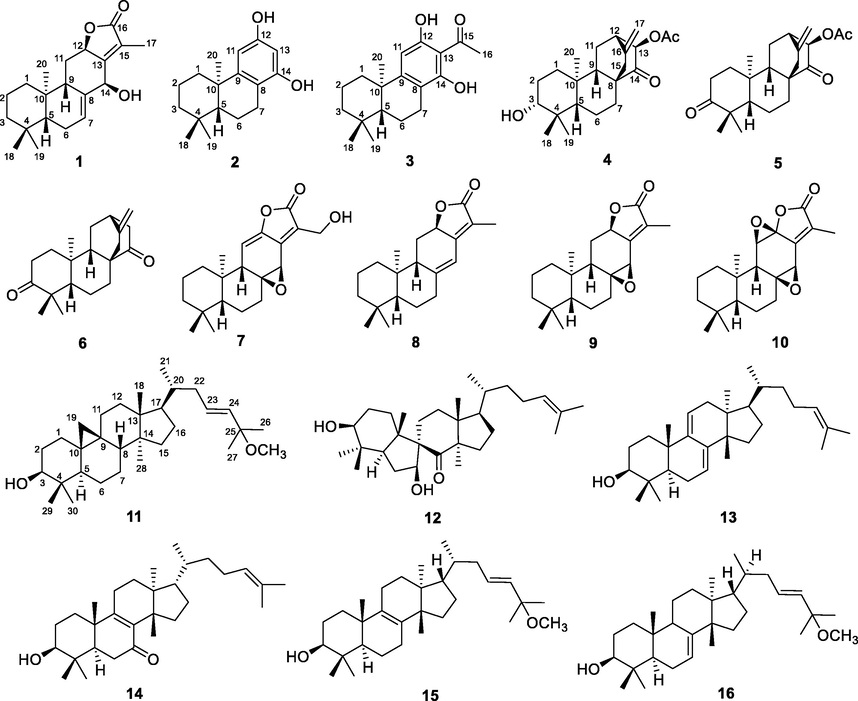
Structures of compounds 1–16.
Compound 1 was isolated as colorless needles and showed a molecular formula of C20H28O3 according to the HRESMS at m/z 339.1920 [M + Na]+ (calcd for C20H28O3Na, 339.1931). Its IR spectrum displayed the absorptions for hydroxyl (3421 cm−1) and carbonyl (1742 cm−1) groups. The 1H NMR data (Table 1) of 1 showed signals for four methyls (δH 0.86, 0.88, 0.91, and 1.97), two oxygenated methines [δH 4.58 (1H, dd, J = 11.8, 6.2 Hz) and 5.05 (1H, s)], an olefinic proton [δH 5.99 (1H, s)] and a series of multiplets for aliphatic protons. The 13C NMR data of 1 (Table 2) showed 20 carbon resonances, which was classified as an ester carbonyl (δC 174.9), two double bonds (δC 123.7, 129.6, 135.8, and 162.3), four sp3 methines (two oxyganated at δC 70.5 and 78.3), five sp3 methylenes, four methyls, and two sp3 quaternary carbons (δC 32.8 and 36.2). The above NMR data were very similar to those of a reported abietane diterpenoid, fischeriolide A (Lee et al., 2016), except for the replacement of the oxygenated methine (δC 65.7, C-11) in fischeriolide A by a methylene (δC 27.0) in 1. This was supported by 1H–1H COSY correlations (Fig. 2) from the methylene protons (H2-11) to H-9 and H-12. Further HMBC and 1H–1H COSY analysis confirmed the gross structure of 1.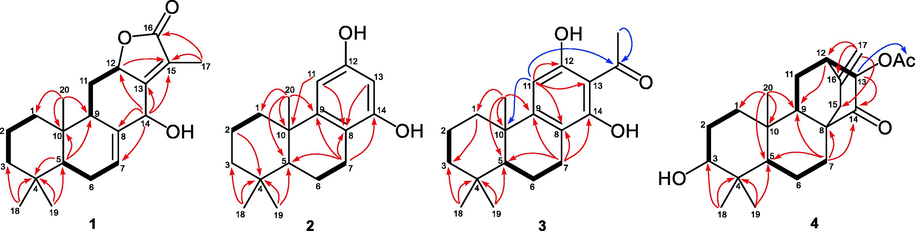
Key 1H−1H COSY (
 ) and HMBC (arrows) correlations of 1–4.
) and HMBC (arrows) correlations of 1–4.
The relative configuration of 1 was partially determined by a NOESY experiment. As shown in Fig. 3, the correlations of H-5/H-9 and H3-20/H-6α indicated that these protons were pseudoaxially oriented on the half-chair conformational B ring. H-5 and H-9 were arbitrarily designated as β-orientation, while CH3-20 was determined as α-orientation. Thus, the NOESY correlation from H3-20 to H-12 assigned H-12 to be α-orientation. As the absence of NOESY correlation from H-14 to H-9, the remaining configuration of H-14 was difficult to confirm. Fortunately, a qualified crystal of compound 1 was obtained via recrystallization in EtOH, and subsequent X-ray diffraction analysis accurately assigned the absolute configuration of 1 as 5R,9S,10R,12R,14S (Fig. 4). Thus, compound 1 was assigned as 14β-hydroxy-ent-abieta-13(15)-ene-12α,16-olide and was given the trivial name euphorane A.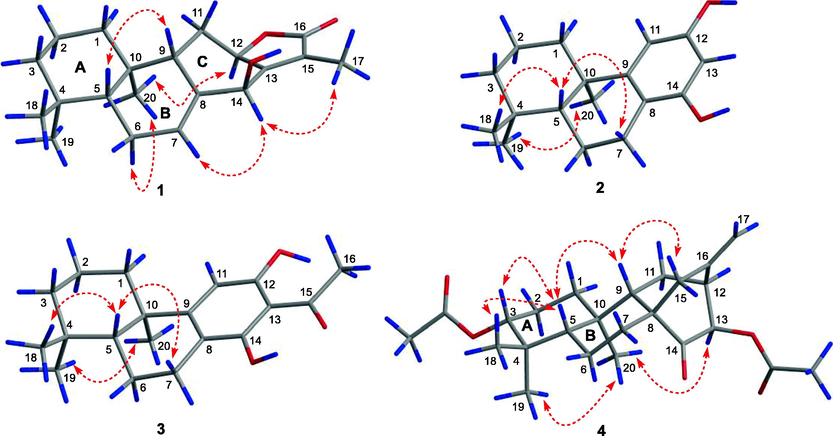
Key NOESY correlations (
 ) of 1–4.
) of 1–4.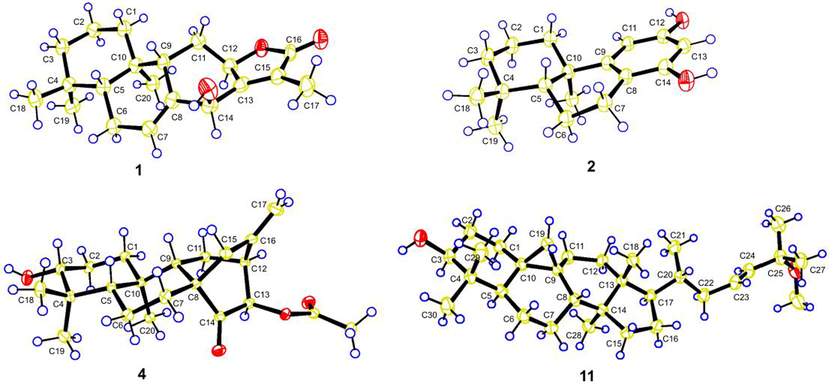
X-ray structures of compounds 1, 2, 4, and 11.
Compound 2 had a molecular formula of C17H24O2 as determined by the HRESIMS at m/z 259.1707 [M − H]− (calcd C17H23O2, 259.1704) and 13C NMR data. The 1D NMR data of 2 were similar to those of a known podocarpane-like trinorditerpene, 7-deoxynimbidiol (Alvarez-manzaneda et al., 2007; Xiong et al., 2006), with the major differences focusing on the 13C chemical shifts in the aromatic ring C (e.g. δC 141.2, 141.5, and 143.6 in 7-deoxynimbidiol and δC 153.3, 156.4, and 156.4 in 2), suggesting that the two phenolic hydroxyls in 2 might be at C-12 and C-14 respectively. This was supported by HMBC correlations from an aromatic proton (δC 6.25, H-11) to C-10 and C-8, and from another aromatic proton (δC 6.10, H-13) to C-8 (Fig. 2). The relative configuration was determined to be the same as that of 7-deoxynimbidiol based on their similar 13C NMR data around rings A and B. This assignment was also supported by the NOESY correlations of H3-20/H3-19 and H3-18/H-5 (Fig. 3). To determine the absolute configuration of 2, a single crystal X-ray diffraction analysis was conducted, and the results showed that 2 possessed a 5R,10R configuration (Fig. 4), contrary to that of 7-deoxynimbidiol (5S,10S). Thus, compound 2 was assigned as an unusual aromatic 15,16,17-trinorabietane diterpenoid and was named euphorane B.
Compound 3 (euphorane C) had a molecular formula of C19H26O3 as determined by its HRESIMS and 13C NMR data. The 1D NMR data of 3 were very similar to those of 2, except for the absence of signals for an aromatic methine (δC 100.3, δH 6.10) and the presence of additional signals for an aromatic quaternary carbon (δC 109.3) and an ethanone fragment (δC 205.9 and 33.9, δH 2.95) in 3, suggesting that 3 was a C-13 acetylated derivative of 2. Observation of a four-bond W-type HMBC correlation from the aromatic proton (δH 6.70, H-11) to the ketone carbonyl (δC 205.9) confirmed this assignment. Similar 13C NMR data around the aromatic ring C of 3 and a synthesized analogue S1 (Fig. 5A) (Shockley et al., 2014) also supported this conclusion. Further computational calculation of the 1D NMR data of two positional isomers 3a and 3b (Fig. 5A) by gauge-independent atomic orbital (GIAO) method and analyzed by the improved probability DP4 + method confirmed this structure. As indicated in Fig. 5B, Tables S4 and S5, a final DP4 + probability score of 100.00 % and 0.00 % was obtained for 3b and 3a respectively, and the experimental 13C NMR data around ring C in 3 matched well with those calculated for isomer 3b (Fig. 5A, Table S2, S4). These results indicated that compound 3 had a structure as depicted and was a rare C17-norabietane diterpenoid.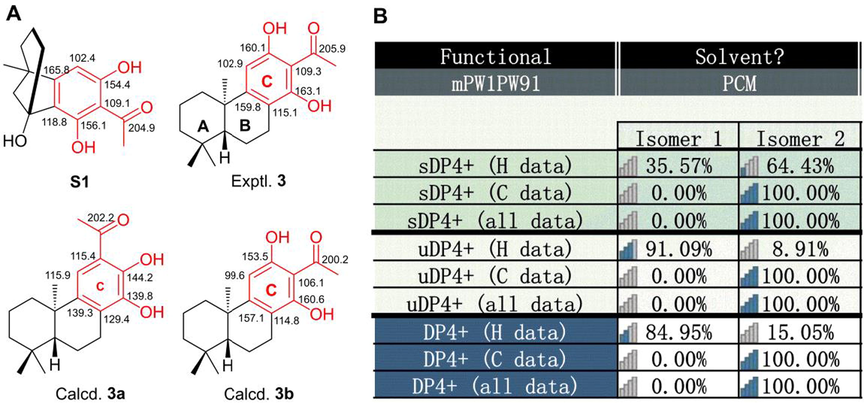
1D NMR calculation of 3. (A) Structures of S1, 3, 3a, and 3b, and their experimental or calculated 13C NMR data around ring C. (B) DP4 + probability of 1D NMR data of 3a (isomer 1) and 3b (isomer 2).
The relative configuration of 3 was determined to be the same as that of 2 based on their similar 1D NMR data around rings A and B. This assignment was also supported by the NOESY correlations of 3 (Fig. 3). Then, the absolute stereochemistry of 3 could be studied by comparison of its experimental and calculated ECD spectra. As shown in Fig. 6, the experimental ECD curve of 3 (3b) showed four Cotton effects around 200 (−), 230 (+), 275 (−), and 350 (+) nm respectively, which matched well with those calculated for the isomer 5R,10R-3b, indicating that 3 possessed the same absolute configuration (5R,10R).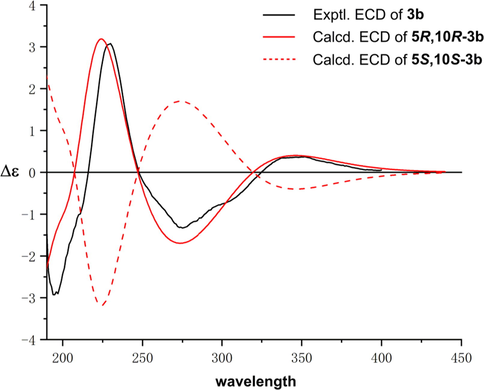
The experimental and calculated ECD curves of 3.
Euphorane C (3) is an unusual C17-norabietane diterpenoid, and its biosynthetic pathway was proposed in Scheme 1. Briefly, proton-initiated polyene cyclization of trans-geranylgeranyl diphosphate could successively produce (+)-copalyl diphosphate and pimarane-like diterpene. Then, 1,2-migration of methyl from C-13 to C-15 constructed the characteristic isopropyl group of the abietane-like precursor, which further underwent the aromatization to give an important intermediate i (Ravn et al., 2002; Yang et al., 2002). The hydroxylations of C-12 and C-14 followed by oxidation of CH3-17 in i afforded intermediate ii. Finally, the C17-norabietane diterpenoid (3) might be generated from ii via decarboxylization and oxidation reactions (Reis et al., 2014; Wang et al., 2015; Yun et al., 2018).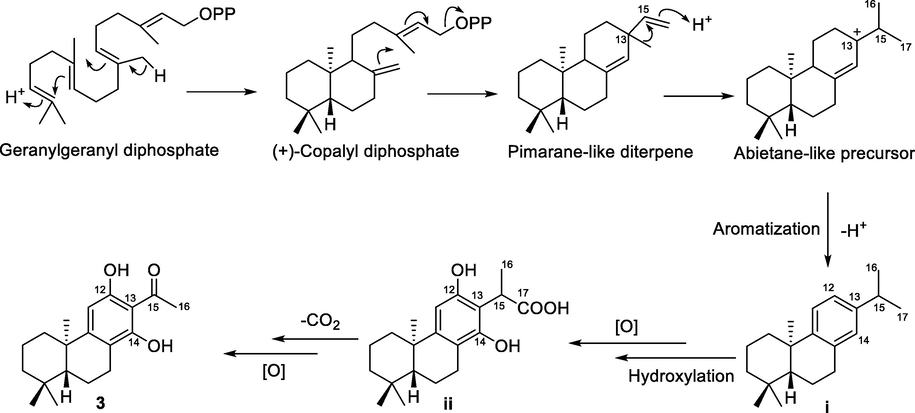
Proposed Biosynthetic Pathway of 3.
Compound 4 was isolated as colorless crystals. Its molecular formula was determined as C22H32O4 by the HRESIMS at m/z 383.2205 [M + Na]+ (calcd for C22H32O4Na, 383.2193). The 1D NMR data of 4 were very similar to those of a reported ent-atisane diterpenoid, ent-(3β,13S)-3,13-dihydroxyatis-16-en-14-one (Zhang et al., 2012), except for the presence of an additional acetyl in 4, suggesting that 4 was an O-acetylated derivative of the reported compound. This was supported by its 2D NMR data (Fig. 2), and the acetoxyl group was located at C-13 by the HMBC correlation from H-13 to the carbonyl (δC 170.5). The relative configuration of 4 was determined by the NOESY experiment. As shown in Fig. 3, the NOESY correlations of H-5/H-3, H-5/H3-18, and H3-20/H3-19 suggested that H-5, H-3, CH3-19, and CH3-20 occupied the axial positions of the chair conformational A ring. H-3 and H-5 were arbitrarily designated as β-orientations, CH3-19 and CH3-20 were accordingly determined as α-orientations. Thus, the NOESY correlations of H-5/H-9 and H-9/H2-15 established the β-orientation of H-9 and C-8 − C-15, while the NOESY correlation of H3-20/H-13 assigned H-13 as α-orientation. Furthermore, the absolute stereochemistry of 4 was determined by single crystal X-ray diffraction experiment to be 3R,5S,8S,9S,10R,12S,13R (Fig. 4). Thus, compound 4 was assigned as 13β-acetoxy-3α-hydroxy-ent-atis-16-ene-14-one and was named euphorane D.
Compound 11 was identified as (23E)-25-methoxycycloart-23-en-3β-ol, a known cycloartane triterpenoid, by comparing its 1D NMR data with those reported (Cabrera et al., 1996; Zhang et al., 2005). However, the absolute stereochemistry of 11 remains to be determined. Thus, a recrystallization experiment was carried out, and a qualified crystal of 11 was obtained in MeOH. The crystallographic data exactly assigned the absolute stereochemistry of 11 as 3S,5R,8S,9S,10R,13R,14S,17R,20R (Fig. 4).
Other known compounds, 13-acetyloxy-atis-16-ene-3,14-dione (5) (Jia et al., 1990), ent-3,14-dioxo-16-atisene (6) (Lal et al., 1989), 17-hydroxyjolkinolide A (7) (Che et al., 1999), jolkinolide E (8) (Lal et al., 1990), jolkinolide A (9) (Lal et al., 1990), jolkinolide B (10) (Che et al., 1999), spiroinonotsuoxodiol (12) (Handa et al., 2010), agnosterol (13) (Emmons et al., 1989), kansenone (14) (Wang et al., 2003), 3β-hydroxy-25-methyloxylanosta-8,23-diene (15) (Fang et al., 2015), and cornusalterin A (16) (Kim et al., 2011) were identified by comparison of their NMR data with those reported in the literature.
The antiproliferative activities of compounds 1–16 were evaluated on five human cancer cell lines. As shown in Table 3, most of the tested compounds only showed mild to none inhibitory activity against the cancer cells. Compounds 10 and 13 imparted cytotoxicity against the chronic myelogenous leukemia cell line (K562) with IC50 values of 7.23 and 5.63 μM respectively. Notably, compound 3 demonstrated significant antiproliferative activity against four of the cancer cell lines with micromolar IC50, and had the highest activity against K562 cells (IC50 = 3.59 μM). Thus, the preliminary mechanism of 3 on inhibiting K562 cells was further investigated by using flow cytometry. As shown in Fig. 7, the treatment of 3 at 4 and 8 μM dose- and time-dependently induced apoptosis of K562 cells as compared to the DMSO treated group. Furthermore, we tested whether 3 could arrest cell cycle. The results showed that 3 treatments (4 and 8 μM) changed cell cycle progression as shown by increased G0/G1 and reduced G2/M cell population (Fig. 8), suggesting that 3 arrested K562 cell cycle at G0/G1 phase. Taken together, compound 3 could arrest cell cycle at G0/G1 phase, finally leading to the apoptosis of K562 cells. Doxorubicin is the positive control.
Compounds
Cell lines
MCF7
HEL
PC3
HepG2
K562
1
7.10
> 20
> 20
> 20
21.4
2
20.98
> 20
> 20
> 20
18.23
3
> 20
10.23
10.22
6.95
3.59
4
> 20
> 20
> 20
> 20
> 20
5
> 20
> 20
> 20
> 20
> 20
6
> 20
> 20
> 20
> 20
> 20
7
> 20
9.60
> 20
> 20
17.23
8
> 20
> 20
> 20
> 20
> 20
9
> 20
> 20
> 20
> 20
> 20
10
> 20
10.65
8.76
> 20
7.23
11
> 20
> 20
> 20
> 20
> 20
12
> 20
> 20
> 20
> 20
> 20
13
> 20
16.3
> 20
> 20
5.63
14
> 20
> 20
> 20
> 20
> 20
15
> 20
> 20
> 20
> 20
> 20
16
> 20
> 20
> 20
> 20
> 20
Doxorubicin
1.02
0.42
0.63
1.87
0.96
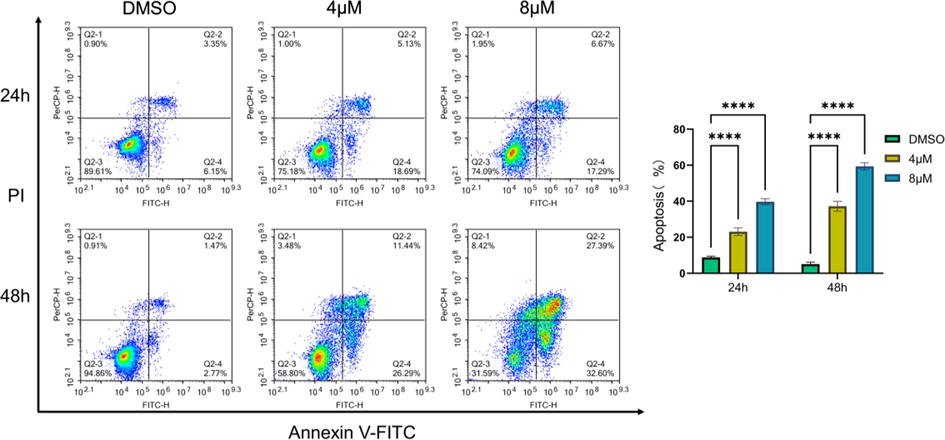
Compound 3 induced apoptosis of K562 cells. The corresponding quantitative analysis was included. Data represented the mean ± SD of at least three independent experiments. ****P < 0.0001 vs DMSO treated group.
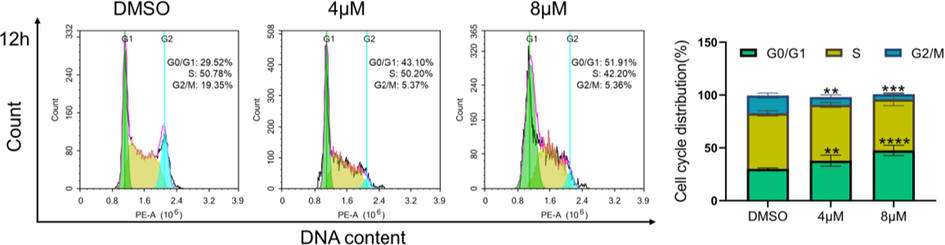
Compound 3 arrested K562 cell cycle at G0/G1 phase. Quantitative analysis was included. Data represented the mean ± SD of at least three independent experiments. **P < 0.01, ***P < 0.001 and ****P < 0.0001 vs DMSO treated group.
4 Conclusions
Polycyclic diterpenoids, such as abietanes (6/6/6 ring system) with 20α-CH3 and 5β-H relative configuration, were common natural products in Euphorbia species. However, abietane trinor or norditerpenoids were rarely discovered in Euphorbia plants (Shi et al., 2008; Vasas et al., 2014; Xu et al., 2021). In the study, chemical investigation of E. dracunculoides led to the isolation of four new polycyclic diterpenoids (1–4) and 12 known terpenoids. Among them, euphorane B (2) represents the first example of aromatic 15,16,17-trinorabietane diterpenoid in Euphorbia species, while 3 is an unusual 17-norabietane diterpenoid. Biological assay indicated that 3 significantly inhibited the proliferation of K562 cells with micromolar IC50. Preliminary mechanism investigation revealed that 3 could induce cell cycle arrest at G0/G1 phase, causing apoptosis of K562 cells. These findings not only expanded the structural diversity of abietane diterpenoids, but also provided potential chemical scaffold for the development of anti-leukemia drugs. Studies toward the in vivo efficacy and in-depth mechanism of 3 are still in progress.
Acknowledgements
This work was supported by National Natural Science Foundation of China (22167009), China Postdoctoral Science Fund (2021M693809), Postdoctoral Science Fund of Guizhou Medical University (YJ20073), and the funds from Guizhou Science and Technology Department (Nos. 2020-5006 and 2020-6011).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Novel synthetic strategy toward abietane and podocarpane-type diterpenes from (−)-sclareol: synthesis of the antitumor (+)-7-deoxynimbidiol. Tetrahedron Lett.. 2007;48:8930-8934.
- [CrossRef] [Google Scholar]
- Cycloartane derivatives from Tillandsia usneoides. J. Nat. Prod.. 1996;59:343-347.
- [CrossRef] [Google Scholar]
- Diterpenes and aromatic compounds from Euphorbia fischeriana. Phytochemistry. 1999;52:117-121.
- [CrossRef] [Google Scholar]
- An unusual indole-diterpenoid with C-17 norcassane skeleton from Euphorbia fischeriana induces HEL cell cycle arrest and apoptosis. Fitoterapia. 2022;159:105195
- [CrossRef] [Google Scholar]
- A new myrinsol diterpenoid ester from Euphorbia dracunculoides. Chem. Nat. Compd.. 2016;52:1041-1043.
- [CrossRef] [Google Scholar]
- Impact of natural products on developing new anti-cancer agents. Chem. Rev.. 2009;109:3012-3043.
- [CrossRef] [Google Scholar]
- A new tigliane-type diterpene from Euphorbia dracunculoides Lam. Nat. Prod. Res.. 2016;30:1639-1645.
- [CrossRef] [Google Scholar]
- lH and 13C NMR assignments for lanostan-3β-ol derivatives: revised assignments for lanosterol. Magn. Reson. Chem.. 1989;27:1012-1024.
- [CrossRef] [Google Scholar]
- Discovery of 8,9-seco-ent-kaurane diterpenoids as potential leads for the treatment of triple-negative breast cancer. J. Med. Chem.. 2021;64:9926-9942.
- [CrossRef] [Google Scholar]
- Terpenoids from Euphorbia pekinensis. J. Asian Nat. Prod. Res.. 2015;17:1213-1219.
- [CrossRef] [Google Scholar]
- An unusual lanostane-type triterpenoid, spiroinonotsuoxodiol, and other triterpenoids from Inonotus obliquus. Phytochemistry. 2010;71:1774-1779.
- [CrossRef] [Google Scholar]
- Two diterpenes from Euphorbia sieboldiana. Phytochemistry. 1990;29:2343-2345.
- [CrossRef] [Google Scholar]
- Tirucallane triterpenoids from Cornus walteri. J. Nat. Prod.. 2011;74:54-59.
- [CrossRef] [Google Scholar]
- New oxidised ent-atisene diterpenes from Eupnorbza fidjiana. Tetrahedron Lett.. 1989;30:3205-3208.
- [CrossRef] [Google Scholar]
- Ent-pimarane and ent-abietane diterpenes from Euphorbia fidjiana. Phytochemistry. 1990;29:2239-2246.
- [CrossRef] [Google Scholar]
- Diterpenoids from the roots of Euphorbia fischeriana with inhibitoryeffects on nitric oxide production. J. Nat. Prod.. 2016;79:126-131.
- [CrossRef] [Google Scholar]
- Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J. Nat. Prod.. 2020;83:770-803.
- [CrossRef] [Google Scholar]
- Mechanism of abietadiene synthase catalysis: stereochemistry and stabilization of the cryptic pimarenyl carbocation intermediates. J. Am. Chem. Soc.. 2002;124:6998-7006.
- [CrossRef] [Google Scholar]
- Diterpenes from Euphorbia piscatoria: synergistic interaction of lathyranes with doxorubicin on resistant cancer cells. Planta Med.. 2014;80:1739-1745.
- [CrossRef] [Google Scholar]
- Jolkinolide B targets thioredoxin and glutathione systems to induce ROS-mediated paraptosis and apoptosis in bladder cancer cells. Cancer Lett.. 2021;509:13-25.
- [CrossRef] [Google Scholar]
- Jolkinolide B sensitizes bladder cancer to mTOR inhibitors via dual inhibition of Akt signaling and autophagy. Cancer Lett.. 2022;526:352-362.
- [CrossRef] [Google Scholar]
- Chemical and pharmacological research of the plants in genus Euphorbia. Chem. Rev.. 2008;108:4295-4327.
- [CrossRef] [Google Scholar]
- A Catalytic, Enantioselective formal synthesis of (+)-dichroanone and (+)-Taiwaniaquinone H. Org. Lett.. 2014;16:6362-6365.
- [CrossRef] [Google Scholar]
- Euphorbia diterpenes: isolation, structure, biological activity, and synthesis (2008–2012) Chem. Rev.. 2014;114:8579-8612.
- [CrossRef] [Google Scholar]
- Structurally diversifified diterpenoids from Euphorbia dracunculoides. Tetrahedron. 2015;71:5484-5493.
- [CrossRef] [Google Scholar]
- A new lathyrane diterpenoid ester from Euphorbia dracunculoides. Chem. Nat. Compd.. 2016;52:1037-1040.
- [CrossRef] [Google Scholar]
- Euphane and tirucallane triterpenes from the roots of Euphorbia kansui and their in vitro effects on the cell division of Xenopus. J. Nat. Prod.. 2003;66:630-633.
- [CrossRef] [Google Scholar]
- ent-Abietane-type and related seco-/nor-diterpenoids from the rare chloranthaceae plant Chloranthus sessilifolius and their antineuroinflammatory activities. J. Nat. Prod.. 2015;78:1635-1646.
- [CrossRef] [Google Scholar]
- Two new myrinsol diterpenoids from Euphorbia dracunculoides Lam. Chin. Chem. Lett.. 2015;26:121-123.
- [CrossRef] [Google Scholar]
- Natural and Semisynthetic Tigliane Diterpenoids with New Carbon Skeletons from Euphorbia dracunculoides as a Wnt Signaling Pathway Inhibitor. Org. Lett.. 2017;19:3911-3914.
- [CrossRef] [Google Scholar]
- Isolation, synthesis, and anti-tumor activities of a novel class of podocarpic diterpenes. Bioorg. Med. Chem. Lett.. 2006;16:786-789.
- [CrossRef] [Google Scholar]
- Diterpenoids from the genus Euphorbia: structure and biological activity (2013–2019) Phytochemistry. 2021;190:112846
- [CrossRef] [Google Scholar]
- Euphonoids A−G, cytotoxic diterpenoids from Euphorbia fischeriana. Phytochemistry. 2019;166:112064
- [CrossRef] [Google Scholar]
- Biosynthesis of abietane diterpenoids in cultured cells of Torreya nucifera var. radicans: biosynthetic inequality of the FPP part and the terminal IPP. Tetrahedron. 2002;58:1265-1270.
- [CrossRef] [Google Scholar]
- A new ent-norabietant diterpenoid from roots of Euphorbia lathyris L. Tetrahedron Lett.. 2018;59:2813-2815.
- [CrossRef] [Google Scholar]
- Bisyinshanic acids A and B, two novel diterpene dimers from the roots of Euphorbia yinshanica. Helv. Chim. Acta. 2012;95:1672-1679.
- [CrossRef] [Google Scholar]
- Cycloartanes from the red alga Galaxaura sp. J. Asian Nat. Prod. Res.. 2005;7:59-65.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2022.104203.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1
Supplementary data 2
Supplementary data 2







