Translate this page into:
Evaluate the effect of graphitic carbon nitride nanosheets decorated with copper nanoparticles on biofilm and icaA gene expression in Staphylococcus aureus isolated from clinical samples
⁎Corresponding authors. salavati@kashanu.ac.ir (Masoud Salavati-Niasari)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
The emergence, spread, and persistence of multidrug-resistant (MDR) bacteria have become a public health threat. Therefore, it is urgent to investigate novel antimicrobial materials without antibacterial resistance. In this regard, Copper/graphitic carbon nitride nanocomposites (Cu/g-C3N4 NCs) with different loading amounts of Cu nanoparticles (NPs) have been successfully prepared via the co-precipitation-sonication approach. The purity of the nanostructures was verified by the X-ray diffraction (XRD) pattern. The antibacterial activity of Cu/g-C3N4 NCs versus tested bacteria were determined by the microdilution technique. Inhibition of biofilm formation was also evaluated using the microtiter plate method. In addition, the ability of Cu/g-C3N4 NCs to the expression level of the icaA gene was assessed by the Real-Time PCR technique. Our results showed that the doping of graphitic carbon nitride with Cu-NPs can improve the antibacterial activity of g-C3N4. So, sample 4 (Cu:g-C3N4 1;1), with higher Cu content, had the highest antibacterial activity toward tested bacteria. The result also showed that sample 4 recorded the largest inhibition zone diameters against tested bacteria, including Staphylococcus aureus (29 mm) and Pseudomonas aeruginosa (16 mm). In addition, Cu/g-C3N4 NCs exhibited a 65% reduction in biofilm formation for Staphylococcus aureus. Moreover, icaA gene was down-regulated after treatment with Cu/g-C3N4 NCs. In conclusion, our study showed that extract Cu/g-C3N4 NCs can use as a promising anti-biofilm agent against biofilm–producing bacteria.
Keywords
Antimicrobial activity
Nanostructures
Cu/g-C3N4 nanocomposites
Antibacterial
Biofilm
1 Introduction
The emergence of multidrug-resistant organisms is considered one of the principal public health challenges since it has a major impact on human health worldwide and causes an increase in the average length of hospital stays, morbidity, mortality, and healthcare costs. Bacterial resistance to antibiotics can be affected by related genetic changes, the impermeability of the bacteria to the antibiotic, and changing the drug target (Giraldi et al., 2019). Many mechanisms are involved in bacterial resistance to antibiotics. One of the main factors contributing to drug resistance is biofilm formation (Gajdács et al., 2021). The biofilm formation of bacteria leads to acute infections such as urinary tract infection, otitis media, osteomyelitis, wound infection, endocarditis and cystic fibrosis. These infections are today a major cause of treatment failure. Various genes induced biofilm formation in bacterial isolates. For example, the presence of icaABCD locus is an essential factor for biofilm formation in S. aureus. So that, this locus induced polysaccharide intercellular adhesin (PIA) expression that plays an important role in the initial attachment and biofilm formation of S. aureus (Cucarella et al., 2001). Thus, the decrease in the expression of this locus (icaA gene) has an important effect to reduce of biofilm formation. Moreover, several studies have demonstrated a direct connection between antimicrobial resistance and biofilm formation. Bacterial biofilms can be more resistant to antibiotics, compared to the planktonic state (Rashki et al., 2022a). To decrease treatment defeat in these infections, it is necessary to design new promising materials to inhibit biofilm formation. Over the last few decades, nanotechnology has been widely studied in fields such as the treatment of cancer, bacterial infections, and drug delivery. These sciences can have an important role in the development of new materials because of their unique physicochemical characteristics and biomedical applications (Zhang et al., 2013; Abdulhusain et al., 2022; Mahde et al., 2022). In recent years, nanostructure-based antimicrobial has appeared as an encouraging agent for the treatment of bacterial infections. Recently, carbon-based materials have been widely studied in the fields of drug delivery, cancer therapy, and antibacterial (Sert et al., 2021; Thurston et al., 2020). Among these, graphitic carbon nitride (g-C3N4) is a class of polymeric materials mainly composed of CN bonds that have emerged as an encouraging prospect for a variety of applications (Naseri et al., 2017). Moreover, g-C3N4 has drawn tremendous concentration because of its simple preparation, superior biocompatibility, excellent physicochemical stability, photocatalytic applications, and degradation of pollutants in water (Oves et al., 2020; Wang et al., 2020). However, graphitic carbon nitride (g-C3N4) has no inherent antibacterial activity, and therefore cannot be used on its own and its use as antimicrobial agent is limited. To empower graphitic carbon nitride with anti-bacterial properties, new efficient nanomaterials have been designed based on graphitic carbon nitride for antibacterial applications (Huang et al., 2014). Several case studies report that the doping nanomaterials (e.g. GO, TiO2, Ag, AgBr, etc.) with g-C3N4 is the most facile method to improve the antibacterial and antibiofilm performance of g-C3N4 (Ma et al., 2016; Xu et al., 2019; Zhou et al., 2016). In this regard, researchers have shown that several g-C3N4-based heterostructures, such as g-C3N4/CdS, g-C3N4/TiO2, g-C3N4/NaNbO3, g-C3N4/BiOI, g-C3N4 nanosheets/Bi2MoO6, g-C3N4 nanosheet/carbon dots/Ag6Si2O7, g-C3N4 nanosheet/MgO, and ZnO/g-C3N4 could improve the antibacterial and antibiofilm activities because of their synergistic effect (Deng et al., 2017; Li et al., 2015; Li et al., 2019). Moreover, Murugesan et al. demonstrated that the g-C3N4/silver iodide nanocomposite could interfere with the bacterial DNA and disturb the bacterial metabolic pathway as a novel antibacterial agent. This nanocomposite can be used for the growth inhibition of bacteria (Murugesan et al., 2018). For this reason, the preparation of g-C3N4-based nanomaterials has been the focus of considerable attention in recent years. In this study, various amounts of Cu NPs were added to g-C3N4 dispersion to enhance the antibacterial and antibiofilm performance of g-C3N4, which was termed Cu/g-C3N4 nanocomposites. In this regard, we introduce a new nanocomposite containing g-C3N4 and different contents of copper for its potential as antimicrobial and biofilm agent. Besides, the ability of Cu/g-C3N4 NCs to the expression level of the icaA gene was evaluated by the Real-Time PCR technique. The preparation of Cu/g-C3N4 nanocomposite was confirmed via X-ray diffraction (XRD) and scanning electron microscopy (SEM). Therefore, we speculated that the Cu/g-C3N4 nanocomposite with significant antibacterial performance could enhance the clinical application of the g-C3N4 nanosheets.
2 Materials and methods
2.1 Materials
All chemical reagents supplied within the analytic standard, copper nitrate 99% Cu (NO3)2, and sodium dodecyl sulfate (SDS) ≥ 99, melamine 99.9% (C3H6N6), L-Ascorbic acid > 98% were obtained from Merck and applied without further purification. Also, Mueller Hinton broth and Mueller Hinton agar were acquired from Merck (Germany).
2.2 Synthesis of Cu particle
First, 300 mg of Cu(NO3)2·3H2O was dissolved in 10 ml of deionized water. This solution was agitated for 30 min at 25 °C. After that, 300 mg of SDS was added to the Cu2+ solution. Then, ascorbic acid was added dropwise to the above mixture under a stirrer to reduce Cu2+ to Cu0. The brick-red solution was centrifuged and washed with deionized water. Finally, the prepared particle was dried at 50 °C for 24 h (Begletsova et al., 2018; Cheng et al., 2006).
2.3 Preparation of Cu/g-C3N4 nanocomposites via co-precipitation-sonication method
g-C3N4 was prepared according to our previous paper (Yousefzadeh et al., 2022). In a typical procedure, a specific quantity of melamine (3.0 g) was directly heated at 550 °C for 4 h. The yellow powder of graphitic carbon nitride was obtained and used for further tests. Then, 300 mg of g-C3N4 was dispersed in 15 ml of deionized water (DIW) for 0.5 h, and the mixture was stirred for 1 h. Next, the various weight ratios of Cu(NO3)2 (mass ratio of Cu: g-C3N4 0.3:1 (sample 1), 0.4:1 (sample 2), 0.5:1 (sample 3), and 1:1 (sample 4)) were dissolved in 10 ml of DIW and mixed with the g-C3N4 suspension. Then, ascorbic acid was added to suspensions under a stirrer until the formation of a transparent solution. The above suspension was agitated for 1 h and centrifuged for 15 min at 7000 rpm. Eventually, the precipitate was washed repeatedly with deionized water and dried at 50 °C for 24 h (Orooji et al., 2020).
2.4 Bacterial isolates
In the present study, the antibacterial performance of Cu/g-C3N4 NCs (samples 1–4) was evaluated against isolates obtained from clinical samples the Shahid Beheshti hospital of Kashan, Iran (Escherichia coli, Pseudomonas aeruginosa, Staphylococcus aureus, Enterococcus faecalis, Klebsiella pneumonia, and Enterobacter cloacae).
2.5 Antimicrobial activity
2.5.1 Measurement of the minimum inhibitory concentration (MIC)
The broth microdilution technique was used to evaluate the antibacterial performance of the Cu/g-C3N4 NCs (samples 1–4) based on the Clinical and Laboratory Standards Institute (CLSI) standards. Mueller Hinton broth medium was added to 96-well microplate. Afterward, 100 μl of dispersed Cu/g-C3N4 NCs was added to each well to obtain a final concentration range from 2500 to 2.44 μg/mL by a series of d two-fold dilutions. Then, bacterial suspension (1 × 107 CFU/mL) was diluted to obtain a final concentration 5 × 105 CFU/ml and was added to a 96-well plate. Next, the plate was incubated at 37 °C for 18–24 h. The MIC was determined as the lowest concentration of Cu/g-C3N4 at which no discernible growth was observed (Karami et al., 2021).
2.5.2 Measurement of the minimum bactericidal concentration (MBC)
The MBC was defined by plating 10 μl of the test solutions on the Blood Agar plate. The number of colonies was counted on the plate after 24 h. The MBC is the minimum concentration of the test compound needed to kill the bacteria (Karami et al., 2021).
2.5.3 Agar disk-diffusion approach
Disk diffusion test was applied to appraise the antimicrobial activity of the Cu/g-C3N4 NCs (samples 1–4). A sterile cotton swab was used to streak the bacterial suspension (1 × 107 CFU/mL) on the surface of the Muller Hinton agar. Then, sterile commercial blank disks were impregnated with samples 1–4 and placed on Muller Hinton agar and incubated at 37 °C for 24 h. The photographs of the plates were taken to evaluate the inhibition zone for the bacteria.
2.6 Biofilm formation assay
In this study, biofilm formation was evaluated using the microtiter plate method for all bacterial isolates. First, 100 µl of bacterial suspension (107 CFU/ml) added to 96 well plates and incubated at 37 °C for 24 h. To eliminate non-adherence cells, wells washed with sodium phosphate buffer two times. We then applied methanol (200 µl) for 20 min to fixation of attached cells. To stain the wells 0.1% crystal violet for 15 min was used. The dye was resuspended with 33% glacial acetic acid (200 µl) (Maisetta et al., 2016; Rashki et al., 2022a). The absorbance value was obtained using a Microplate Reader at 570 nm. Based on absorbance values, bacterial isolates were classified into the following four groups: non-biofilm (OD570 ≤ ODc), weak (OD570 > ODc and ≤ 2 × ODc), moderate (OD570 > 2 × ODc and ≤ 4 × ODc), and strong (OD570 > 4 × ODc). The cutoff value (ODc) is defined the mean OD570 of the negative control (Ghanizadeh et al., 2021).
2.7 Biofilm inhibition effect
We assessed the ability of Cu/g-C3N4 nanocomposite (sample 4) with MIC and sub-MIC (1/2 and 1/4) concentrations to inhibit the biofilm formation of isolates produced strong biofilm at 24 h using 96-well plates. For this purpose, 100 μl bacterial suspension produced strong biofilm (1 × 107 CFU/mL) in Tryptic Soy Broth (TSB) medium (Merck, Germany) was seeded into 96-well plates in the presence of 100 μl various concentrations of tests compounds and incubated at 37 °C for 24 h. After 24 h, wells were washed twice with phosphate-buffered saline (PBS) to remove non-adhered cells. The formed biofilms were fixed with methanol (20 min). The plate's wells were then stained with 0.1% (w/v) crystal violet for 15 min, and glacial acetic acid was used to re-solubilize crystal violet. After 15 min, the OD of each well was measured with a spectrophotometer at 570 nm. The following equation was used for the calculation of the inhibition percentage:
2.8 Analysis of gene expression related to biofilm using real-time qPCR
In this research, we explored effect Cu/g-C3N4 NCs on the expression of icaA gene using real-time PCR method. Isolates that produced strong biofilm were chosen to assess gene expressions of icaA. First, RNA of the bacterial cells treated with sub-MIC (1/2 MIC) concentration was extracted with RNX Plus protocol. To assay purity of the extracted RNA, the absorbance of RNA was measured at 260 and 280 nm by UV spectroscopy (Thermo Scientific, USA). Then, the cDNA (complementary DNA) was synthesized based on kit (Pars Tous, Iran) guidelines. The primer sequences and reference gene (glyA gene) were listed in Table 1. The fold change was normalized according to the 2−△△CT formula.
Gene
Primer sequences
Primer length
icaA
F-3ʹ-ACGTTGGCTACTGGGATACTG-5ʹ
R-3ʹ-CTTCCAAAGACCTCCCAATG-5ʹ21
20
glyA
F-3ʹ-GGTGGCTGTGAGTTTGTAGATG-5ʹ
R-3ʹ-CGCTTGTGAACCTGAATGTG-5ʹ22
20
3 Results and discussion
3.1 Characterization
Fig. 1 reveals the XRD patterns of pure Cu NPs, pristine g-C3N4 and Cu/g-C3N4 nanocomposites with different mass ratios. Fig. 1a depicts high purity and crystallinity of Cu (JCPDS No. 085-1326). Fig. 1b shows the pattern of carbon nitride (JCPDS No. 87-1526), exhibiting two distinct peaks. The sharpest one, occurring at 26.5°, and the smallest one, at 13.1°, are indexed as (0 0 2) and (0 0 1) planes of C3N4, respectively. Fig. 1(c-f) indicates the XRD patterns of different contents of Cu on the g-C3N4. Cu (JCPDS No. 085-1326) and g-C3N4 (JCPDS No. 87-1526) are present in all four patterns. As can be seen, increasing the Cu content from 0.3 g to 1.0 g decreased the intensity of carbon nitride while increasing the intensity of Cu peaks.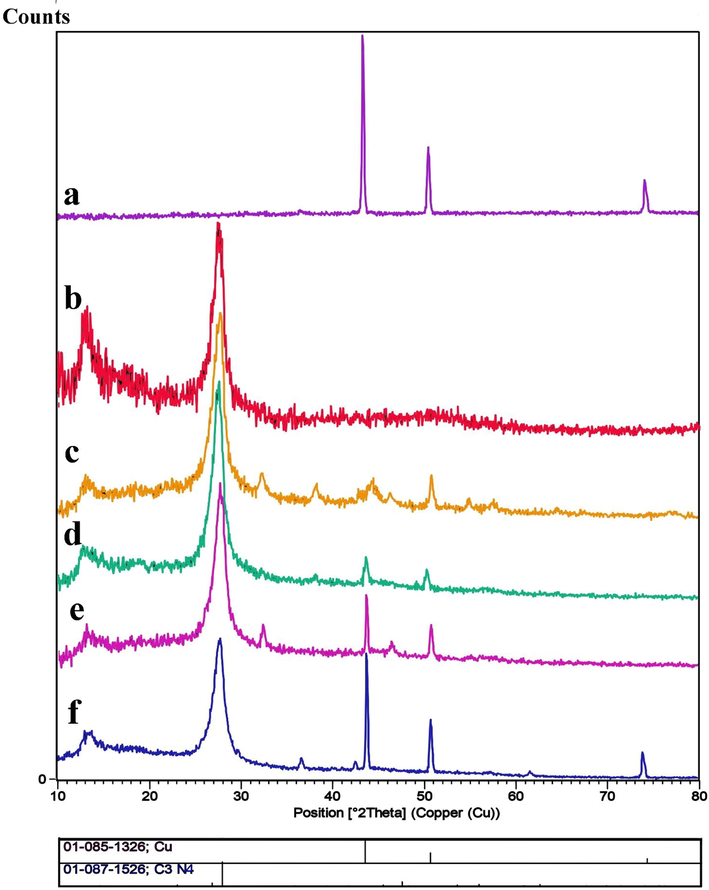
XRD patterns of a) Cu, b) g-C3N4, c) Cu/g-C3N4 (0.3:1), d) Cu/g-C3N4 (0.4:1), e) Cu/g-C3N4 (0.5:1), and f) Cu/g-C3N4 (1:1), nanocomposites.
The FESEM images of Cu particles fabricated with and without SDS as a surfactant are depicted in Fig. 2. Fig. 2a and b display regular pyramidal particles composed in the presence of SDS, while the irregular triangular particles were formed in the absence of SDS (Fig. 2c and d). Cu particles in the presence of SDS have better morphology; therefore, SDS was used in the further tests to prepare Cu/g-C3N4 nanocomposites. SDS can affect the regularity and distribution of copper particles in certain conditions. SDS is a commonly used surfactant in various chemical and biological applications due to its ability to reduce surface tension and stabilize colloidal suspensions. When added to a copper particle solution, SDS molecules can adsorb onto the surface of the particles and form a protective layer, preventing agglomeration and promoting dispersion. This can lead to more uniform distribution of copper particles in the solution. Fig. 3(a-d) reveals the FESEM photographs of Cu/g-C3N4 NPs with various mass ratios. These images demonstrate the nanoflake architecture of g-C3N4 and Cu NPs arranged on the g-C3N4 surface. Typically, expanding the quantity of Cu leads to an accumulation in the diameter of Cu NPs on the carbon nitride surface. Fig. 4(a-d) shows TEM images of sample 4 with a 1:1 mass ratio of Cu:g-C3N4 at various scales (80, 60, 40, and 20 nm). These pictures corroborated the FESEM findings and demonstrated the distribution of Cu NPs on the g-C3N4 substrate. EDS spectroscopy is a scientific method used for chemical description and elemental analysis of materials. Fig. 5a and b depict the EDS spectra of Cu and Cu/g-C3N4, representing all peaks allocated to Cu, C, and N elements. Therefore, both samples are thoroughly purified.
FESEM images of Cu NPs with (a,b) and without (c,d) SDS.

FESEM images of Cu/g-C3N4 nanocomposites in different mass ratios a) 0.3:1, b) 0.4:1, c) 0.5:1, and d) 1:1.

TEM images of Cu/g-C3N4 nanocomposites (sample 4).
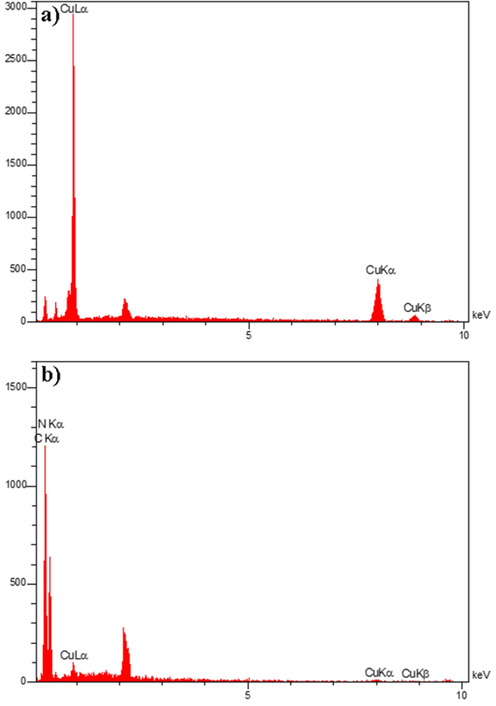
EDS spectra of a) Cu and b) Cu/g-C3N4 nanocomposites.
3.2 Antimicrobial activity
In this study, the antimicrobial activity of Cu/g-C3N4 NCs, and g-C3N4 was evaluated by the broth microdilution method. As revealed in Table 2, samples 3 and 4 (with higher Cu content) were more effective against the tested bacterial species than other samples. Also, the results demonstrated that these samples were more effective against gram-positive than gram-negative bacteria. According to the results, the loaded Cu-NPs on the g-C3N4 could improve its antibacterial activity. Furthermore, according to MIC values, we understand that sample 4 could inhibit the growth of test bacteria in lower concentrations compared to other samples. In this study, the inhibition zone diameters of tested bacteria using disk diffusion method. Table 3 and Fig. 6. The results showed that sample 4 had larger inhibition zone than the other samples. The inhibitory growth zone of Staphylococcus aureus (29 mm) is greater than that of the other microorganisms tested. According to research, the presence of Cu NPs is responsible for the antibacterial activity of Cu/g-C3N4 NCs (Sun et al., 2017). The exact mechanism of antibacterial activity is not fully understood, and several other factors may contribute to the antibacterial action (Sun et al., 2017; Thurston et al., 2020). According to studies, the surface of Cu NPs plays a crucial role in antibacterial activity. Copper antibacterial functionality is associated with various mechanisms, including damaging the microbial DNA, altering bacterial protein synthesis and altering membrane integrity. Some research has evaluated the antibacterial activity of the loaded nanoparticles on the g-C3N4. For instance, Orooji et al. investigated the antibacterial action of silver iodide/graphitic carbon nitride nanocomposites against tested bacteria. The results demonstrated that silver iodide could enhance the g-C3N4 antibacterial effect (Orooji et al., 2020). In a study by Chongchong et al., the g-C3N4/Ag nanocomposites showed better bacterial activity than g-C3N4 (Liu et al., 2016). The findings of this research are similar to the results of the present study. It can be confirmed that the loaded nanoparticles on the g-C3N4 can enhance the antibacterial performance of the g-C3N4. (Cabiscol Català et al., 2000; Thurston et al., 2020). It is concluded that Cu NPs have the potential to enhance the antibacterial effect of g-C3N4 against resistant pathogenic microbes.
Bacteria
Cu/g-C3N4 NCs
g-C3N4
Sample 3
Sample 4
MIC
MBC
MIC
MBC
MIC
MBC
Staphylococcus aureus
1250
1250
312.5
2500
–
–
Entrococcus fecalis
–
–
–
–
–
–
Escherichia coli
1250
2500
625
1250
–
–
Pseudomonuas aeruginosa
1250
1250
1250
2500
–
–
Klebsiella pneumonia
–
–
–
–
–
–
Enterobacter cloacea
–
–
1250
5000
–
–
Bacteria
Sample 3
Sample 4
Staphylococcus aureus
14
29
Entrococcus fecalis
–
–
Escherichia coli
11
15
Pseudomonuas aeruginosa
10
19
Klebsiella pneumonia
–
–
Entrobacter cloacea
12
16
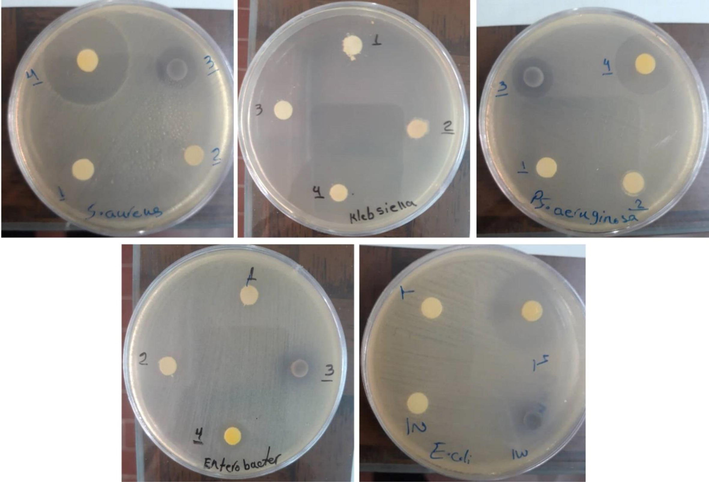
Zones of growth inhibition of Cu/g-C3N4 nanocomposites (samples 1, 2, 3, and 4) on tested microorganisms S. aureus, klebsiella pneumoniae, pseudomonas aeruginosa, Entrobacter cloacea and, Escherichia coli.
3.3 Antibiofilm activity
Biofilm formation is the main cause of chronic bacterial infections and could lead to an increase in antibiotic resistance. Preformed biofilm by blocking the diffusion of antimicrobial agents into the biofilm matrix could enhance the resistance of bacteria (Abebe, 2020). In this study, only S. aureus isolates were strong biofilm producers. Therefore, in this study, we assayed the ability of Cu/g-C3N4 NCs (sample 4) to inhibit the formation of biofilms of S. aureus at 24 h. The results demonstrated that Cu/g-C3N4 NCs significantly inhibited biofilm formation of S. aureus strain in a concentration-dependent manner. As quantified by crystal violet, Cu/g-C3N4 inhibited biofilm formation in the S. aureus strain by 65% at the MIC concentration (Fig. 7). Given that the extracellular matrix is a major structural component of microbial biofilms (Karygianni et al., 2020). One of the main reasons for the effect of Cu-NPs on inhibited biofilm formation in the S. aureus is their particle size, as smaller particles with large surface area have more interaction with extracellular matrix bacteria. So that by destroying the extracellular matrix leads to the weakening of the biofilm and facilitates the entry of the drug into the inner layer of the biofilm (Bi et al., 2021).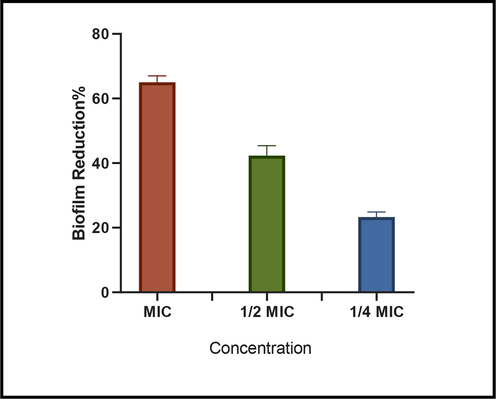
Effect of Cu/g-C3N4-NCs (sample 4) at MIC, ½ MIC and ¼ MIC concentration on the formation of Staphylococcus aureus biofilm analyzed via crystal violet staining.
3.4 Analysis of icaA gene expression in the presence of Cu/g-C3N4 nanocomposites using Real-time qPCR
To further understand the antibiofilm mechanism of prepared Cu/g-C3N4 NCs, change the gene expression related to biofilm S. aureus in the presence of this nanocomposites was evaluated. For this purpose, we compared the expression of icaA gene in S. aureus isolated after treatment with prepared nanocomposite by RT-PCR technique. So far, there is no similar study has been evaluated the effects of Cu/g-C3N4 NCs on the expression of this gene. In other words, we assessed the expression of icaA genes for the first time. As shown in Fig. 8, following treatment with Cu/g-C3N4 NCs, the expression level of the icaA gene was down-regulated as much as 2.26. Although, the expressions of icaA gene in isolates treated with antibiotic decreased by 6 folds. In general, antibiotic has reduced gene expression more than Cu/g-C3N4 NCs. The exact mechanism of this phenomenon is still unclear. Generally, icaA gene facilitate bacterial attachment to the surfaces and as a result, leading the formation of biofilm. Therefore, the factors that cause changes in the expression level of this gene can help to reduce biofilm formation. Thereby Cu/g-C3N4 NCs with reduced icaA expression can have an impact important on the biofilm inhibition of S. aureus.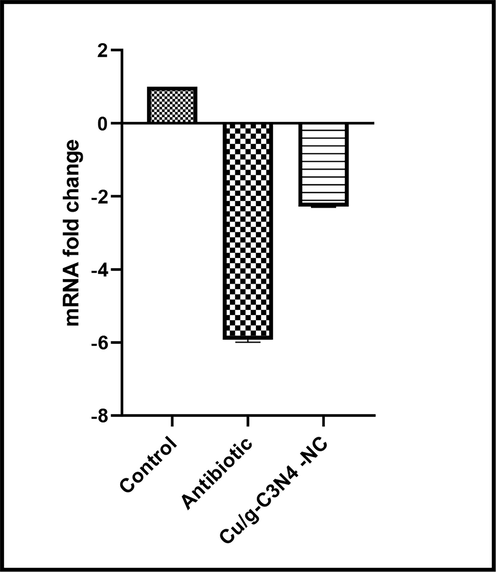
The effect of Cu/g-C3N4- NCs on icaA gene expression. All graphs represent mean SEM.
4 Conclusion
Our results showed that the doping of Cu-NPs with graphitic carbon nitride can lead to an improved antibacterial activity of g-C3N4. So, Cu/g-C3N4 nanocomposite is more effective against tested gram-positive and gram-negative bacteria, especially S. aureus. Also, this nanocomposite could be used to reduce or prevent biofilm formation on medical devices. The biofilm inhibition in S. aureus isolate was iacA gene independent as a result of down-regulation of iacA gene led to biofilm inhibition in S. aureus after treatment by Cu/g-C3N4 nanocomposite. Therefore, Cu/g-C3N4 could be applied for the treatment of medical device-related infections. Given the potential of Cu/g-C3N4 as a promising antimicrobial and antibiofilm material could be utilized to reduce antibacterial resistance among clinical isolates. This compound has the potential to prevent or treat pathogen-caused infections.
CRediT authorship contribution statement
Somaye Rashki: Software, Investigation, Methodology, Formal analysis. Mojgan Ghanbari: Writing – review & editing, Writing – original draft, Software. Zainab Hashim Khudhair: Writing – review & editing, Data curation. Zeynab Marzhoseyni: Software, Data curation, Visualization. Zakaria Bameri: Software, Data curation, Validation. Saleh Afsharikhah: Software, Conceptualization, Data curation, Validation. Waleed K. Abdulsahib: Writing – review & editing, Data curation. Ali Nazari-Alam: Software, Funding acquisition, Data curation, Validation. Masoud Salavati-Niasari: Formal analysis, Methodology, Writing – review & editing, Writing – original draft, Conceptualization, Methodology, Supervision, Project administration, Investigation, Data curation, Validation, Resources, Visualization, Funding acquisition.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Anabasis articulata exerts an anti-arthritic effect on adjuvant-induced arthritis in rats. J. Adv. Pharm. Technol. Res.. 2022;13(4):276-280.
- [CrossRef] [Google Scholar]
- The role of bacterial biofilm in antibiotic resistance and food contamination. Int. J. Microbiol.. 2020;2020:1705814.
- [CrossRef] [Google Scholar]
- Chemical synthesis of copper nanoparticles in aqueous solutions in the presence of anionic surfactant sodium dodecyl sulfate. Colloids Surf A Physicochem Eng Asp. 2018;552:75-80.
- [Google Scholar]
- Therapeutic strategies against bacterial biofilms. Fundam. Res.. 2021;1(2):193-212.
- [Google Scholar]
- Oxidative stress in bacteria and protein damage by reactive oxygen species. Int. Microbiol.. 2000;3(1):3-8.
- [Google Scholar]
- Modifier effects on chemical reduction synthesis of nanostructured copper. Appl. Surf. Sci.. 2006;253(5):2727-2732.
- [Google Scholar]
- Bap, a Staphylococcus aureus surface protein involved in biofilm formation. J. Bacteriol.. 2001;183(9):2888-2896.
- [Google Scholar]
- Enhanced visible-light-driven photocatalytic bacteria disinfection by g-C3N4-AgBr. Colloids Surf. B Biointerfaces. 2017;152:49-57.
- [Google Scholar]
- No correlation between biofilm formation, virulence factors, and antibiotic resistance in Pseudomonas aeruginosa: results from a laboratory-based in vitro study. Antibiotics. 2021;10(9):1134.
- [Google Scholar]
- Genetic diversity, antimicrobial resistance pattern, and biofilm formation in Klebsiella pneumoniae isolated from patients with coronavirus disease 2019 (COVID-19) and ventilator-associated pneumonia. Biomed Res. Int.. 2021;2021
- [CrossRef] [Google Scholar]
- Healthcare-associated infections due to multidrug-resistant organisms: a surveillance study on extra hospital stay and direct costs. Curr. Pharm. Biotechnol.. 2019;20(8):643-652.
- [Google Scholar]
- Metal-free disinfection effects induced by graphitic carbon nitride polymers under visible light illumination. Chem. Commun.. 2014;50(33):4338-4340.
- [Google Scholar]
- Facile fabrication of Tl 4 HgI 6 nanostructures as novel antibacterial and antibiofilm agents and photocatalysts in the degradation of organic pollutants. Inorg. Chem. Front.. 2021;8(10):2442-2460.
- [Google Scholar]
- Biofilm matrixome: extracellular components in structured microbial communities. Trends Microbiol.. 2020;28(8):668-681.
- [Google Scholar]
- Enhanced visible-light-driven photocatalytic inactivation of Escherichia coli using g-C3N4/TiO2 hybrid photocatalyst synthesized using a hydrothermal-calcination approach. Water Res.. 2015;86:17-24.
- [Google Scholar]
- Graphitic carbon nitride (g-C3N4) nanosheets functionalized composite membrane with self-cleaning and antibacterial performance. J. Hazard. Mater.. 2019;365:606-614.
- [Google Scholar]
- “One pot” green synthesis and the antibacterial activity of g-C3N4/Ag nanocomposites. Mater. Lett.. 2016;164:567-570.
- [Google Scholar]
- Enhanced disinfection application of Ag-modified g-C3N4 composite under visible light. Appl Catal B. 2016;186:77-87.
- [Google Scholar]
- Kinetic adsorption and release study of sulfadiazine hydrochloride drug from aqueous solutions on GO/P(AA-AM-MCC) composite. Int. J. Drug Deliv. Technol.. 2022;12(4):1583-1589.
- [CrossRef] [Google Scholar]
- Anti-biofilm properties of the antimicrobial peptide temporin 1Tb and its ability, in combination with EDTA, to eradicate Staphylococcus epidermidis biofilms on silicone catheters. Biofouling. 2016;32(7):787-800.
- [Google Scholar]
- Photocatalytic performance and antibacterial activity of visible light driven silver iodide anchored on Graphitic-C3N4 binary composite. Environ. Nanotechnol. Monit. Manage.. 2018;10:253-263.
- [Google Scholar]
- Graphitic carbon nitride (gC 3 N 4)-based photocatalysts for solar hydrogen generation: recent advances and future development directions. J. Mater. Chem. A. 2017;5(45):23406-23433.
- [Google Scholar]
- Facile fabrication of silver iodide/graphitic carbon nitride nanocomposites by notable photo-catalytic performance through sunlight and antimicrobial activity. J. Hazard. Mater.. 2020;389:122079.
- [Google Scholar]
- Synthesis and antibacterial aspects of graphitic C3N4@ polyaniline composites. Coatings. 2020;10(10):950.
- [Google Scholar]
- Delivery LL37 by chitosan nanoparticles for enhanced antibacterial and antibiofilm efficacy. Carbohydrate Polym.. 2022;291
- [CrossRef] [Google Scholar]
- Improvement in performance of g-C3N4 nanosheets blended PES ultrafiltration membranes including biological properties. Colloids Surf. A Physicochem. Eng. Asp. 2021;623:126571.
- [Google Scholar]
- Antibacterial activity of graphene oxide/g-C3N4 composite through photocatalytic disinfection under visible light. ACS Sustain. Chem. Eng.. 2017;5(10):8693-8701.
- [Google Scholar]
- Development of photoactive g-C3N4/poly (vinyl alcohol) composite hydrogel films with antimicrobial and antibiofilm activity. ACS Appl. Bio Mater.. 2020;3(3):1681-1689.
- [Google Scholar]
- Recent progress in gC 3 N 4 quantum dots: synthesis, properties and applications in photocatalytic degradation of organic pollutants. J. Mater. Chem. A. 2020;8(2):485-502.
- [Google Scholar]
- Combination of CuS and g-C3N4 QDs on upconversion nanoparticles for targeted photothermal and photodynamic cancer therapy. Chem. Eng. J.. 2019;360:866-878.
- [Google Scholar]
- Fabrication of TlSnI3/C3N4 nanocomposites for enhanced photodegradation of toxic contaminants below visible light and investigation of kinetic and mechanism of photocatalytic reaction. J. Mol. Liquids. 2022;349
- [CrossRef] [Google Scholar]
- Advanced materials and processing for drug delivery: the past and the future. Adv. Drug Deliv. Rev.. 2013;65(1):104-120.
- [Google Scholar]
- Well-Dispersed Fe2O3 nanoparticles on g-C3N4 for efficient and stable photo-fenton photocatalysis under visible-light irradiation. Eur. J. Inorg. Chem.. 2016;2016(34):5387-5392.
- [Google Scholar]







