Translate this page into:
Evaluation of crude methanolic mangrove leaves extract for antibiofilm efficacy against biofilm-forming bacteria on a cooling tower wastewater system
⁎Corresponding author rajasekargood@gmail.com (Aruliah Rajasekar)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
The present study demonstrated microbial corrosion protection of MS 1010 on cooling tower water using plant-based inhibitors derived from methanolic extraction of dry mangrove leaves (R.mangle and A.marina), and its assessment of antibacterial activity against corrosive bacteria (B.megaterium) was investigated. FT-IR and GC–MS analyzed the inhibitors component and corrosion behavior of MS 1010 on cooling water, with and without inhibitors were analyzed by EIS and Tafel studies. GC–MS spectra confirmed the presence of Myo-Inositol, 4-C-methyl and chromene as major constituents presented on the R.mangle whereas Lupeol, trifluoroacetate and beta-amyrin compounds were found on the A.marina. In the cooling water, these two inhibitors demonstrated outstanding antibacterial activity and controlled biofilm growth. As plant-based inhibitors were used in cooling water systems, EIS data showed a significant increase in Rct value when compared to the control system. Tafel plot indicates inhibitors have mixed inhibitory effects and for the systems with and without inhibitors, the icorr value was 1.5649A and 2.0875A, respectively. At the optimal dose of 25 ppm, the inhibitory efficiency of MERM and MEAM was 81% and 80%, respectively. The overall discussion reveals that inhibitor substances can be absorbed on the metal surface and then act as a dual role in inhibiting corrosive bacterial growth and barrier to the corrosion process in the cooling water system.
Keywords
Biocorrosion
Bacillus megaterium
Rhizophora mangle
Avicennia marina
Eco-friendly inhibitors
1 Introduction
Mild steel (MS) is the most favorable metal used for various industrial sectors such as oil, gas pipelines, building construction, cooling tower plants, etc. For industrial purposes, MS pipelines are usually used to cool tower plants for circulation and transport the water to specific operating conditions (Kumar Yadav & Soni, 2013; Rajasekar et al., 2007). The cooling tower is damaged due to the metal composition and regular water flow. This water contains rich in presence of microbes that have secreted extracellular polymeric substances and formed biofilm layers on the metal surface that involve metal deterioration is called Microbially Influenced Corrosion (MIC). (Fayyad et al., 2021; Granero et al., 2009). Bacillus megaterium (B.megaterium) is a gram-positive, aerobic spore-forming huge size bacteria, the name megaterium means ‘big animal’ (Greek), and even this microorganism is the biggest of all bacilli (Vejar et al., 2016). In the process of bacterial communication known as quorum sensing (cell signalling hormones), it can release the extracellular polymeric substance (EPS). Acyl homoserine lactones (AHLs)) are responsible for the secretion, synthesis and sense of signal molecules known as autoinducers, as well as communicating the signal to other microbes, which subsequently interact to exchange electrons and assemble to create biofilm layers (Marchand & Collins, 2016; Pusparizkita et al., 2020). In the cooling tower water plant, bio-treatment activities of wastewater treatment units have gathered a large number of microbes allowing for simple and healthy bacterial growth. By increasing heterogeneity on the metal's surface, it can influence metal distraction through the development of biofilm layers. B.megaterium will begin to utilise iron (Fe) as an electron donor, which is oxidized to Fe2+ and Fe3+ to provide the energy needed for the metabolic pathways. Direct transfer electrons can carry electrons through the cell wall to the cyto-plasma via protein cell membranes (Feng Lin et al., 2013). Beneath the biofilm, oxidized metal on the anode might interact with acid compounds generated by B.megaterium to induce corrosion (pitting) products (Rajasekar & Ting, 2011). Corrosion is a significant trouble in the cooling tower system because of the wastewater composition, which includes salt, pH, temperature, impure particles, microbes, and chemical compounds (hydrogen sulfide, sodium chloride), which all impact the metals susceptibility to corrosive substances. The corrosion rate is increased by sodium chloride which prohibits the formation of a protective FeS layer on the iron surface. Green inhibitors are a concentrated mixture of organic compounds including oxygen, sulphur, nitrogen and hydrogen atoms that inhibits microorganism from interacting with metal compounds (Kokilaramani et al., 2021a; Shah et al., 2011). Many researchers are working to develop effective green corrosion inhibitors for a wide range of metals in multiple configurations utilizing various components of plant (organic) materials including fronds, seeds, or bark extracts (Kamaruzzaman et al., 2021; Rahim et al., 2007).
Mangrove leaves include bioactive and secondary compounds, which are non-poisonous, ecologically friendly, organic molecules derived from natural sources and the mangrove leaves extracted were reported to be an effective inhibitor for corrosion (Bothi Raja & Sethuraman, 2008). The mangrove tree is a global, fast-growing species found along tropical and subtropical coastlines and it has been noted for its bio-potentials and distinctive chemical compositions, which are attributed to its therapeutic uses (Cui et al., 2019). Rhizophora mangle (R.mangle) often known as red mangroves, is a kind of mangrove tree that grows in tropical evergreen trees that look like a stilt root and form dense thickets along coastal coastlines. It has thick bark, grey-brown in color, with a smooth edge of oval shape, yellow flowers were blooms in spring and top of the trees has a deeper shade of green than its bottom (Biber, 2006; Okeniyi et al., 2019b; Tan & Kassim, 2011). Another one is the Avicennia marina (A.marina) often known as white mangrove, which has light grey or whitish bark in stiff, brittle, thin flakes and their upper leaves are robust, shiny, and deep green, while the lower leaves are grey or silver-white with tiny hairs and they are the global, fastest-growing species located along with tropical and subtropical areas. (Amaral et al., 2011; Fouda et al., 2017; Shebany, 2012). Table 1 shows the classification of the mangrove plants R.mangle and A.marina. In corrosive environments, plant extracts are preferable to use as an inhibitor owing to their simple synthesis, biocompatibility, widespread availability, and economical efficiency. High inhibitor concentrations allow more surface molecules to be adsorbed, which enhances inhibitor efficiency and slows down the corrosion rate. In that adsorption provides a smooth interface between inhibitors and a metal surface and the metal surface, which is mostly ascribed to the heteroatom in an organic molecule with unsaturated p-bonds, effectively blocks the metal dissolution. The antimicrobial activity of mangrove leaves highly inhibit bacterial growth, it is easily adsorbed on the metal surface and creates protective layers for preventing metal corrosion and microbe communication (Chauhan et al., 2021; Musthafa et al., 2013).
Names
Rhizophora mangle
Avicennia marina
Empire
Eukaryota
Eukaryota
Kingdom
Plantae
Plantae
Subkingdom
Viridiplantae
Viridiplantae
Infrakingdom
Streptophyta
Streptophyta
Superdivision
Embryophyta
Embryophyta
Division
Tracheophyta
Tracheophyta
Subdivision
Spermatophytina
Spermatophytina
Phylum
Spermatophyta
Tracheophyta
Subphylum
Angiospermae
Spermatophytina
Class
Dicotyledonae
Magnoliopsida
Subclass
Dictyotophycidae
Fucophycidae
Superorder
Rosanae
Asteranae
Order
Rhizophorales
Lamiales
Family
Rhizophoraceae
Acanthaceae
Genus
Rhizophora
Avicennia
Species
Rhizophora mangle
Avicennia marina
This research work was aimed to evaluate the efficiency of mangrove leaves as a green corrosion inhibitor, as well as their adsorption behavior in B.megaterium strain isolated from cooling tower water using MS 1010 and the studies inhibition efficiency of methanolic extraction of mangrove leaves was evaluated by Minimal Inhibition Concentration, Biofilm Inhibition Assay, and Weight Loss measurement (WL). Future analysis Electrochemical measurements (Tafel and Polarization), surface studies such as GCMS (Gas chromatography-mass spectrometry), and FTIR (Fourier transform infrared spectroscopy) was done. This is the first report, on using mangrove leaves (green inhibitors) to be considered a potential inhibitor for microbial corrosion (biocorrosion) in the cooling tower water. Recently, many research papers have reported the successfully used of natural products like plant leaf, flower, stem extracts as effective corrosion inhibitors against metal corrosion in acidic and alkaline media. Peach pomace (Vorobyova & Skiba, 2021), Fucus spiralis (Afrokh et al., 2022), purple rice bran (Pal & Das, 2022), Moroccan, Mauritania, and Senegalese gum (el Azzouzi et al., 2022) was investigated for their corrosion inhibition potential. The corrosion prevention mechanism this natural plant extract on mild steel metal demonstrates favorable adsorption and physisorption. This study shows that the leaf extracts from these natural plants R. mangle and A.marina are suitable for corrosion prevention in cooling tower water environment as a green/environmentally friendly inhibitor.
2 Materials and methods
2.1 Bacterial strain and media composition
The bacterial strain, B.megaterium SKR7 was identified from wastewater of the cooling water system. GenBank accession number MT211511.1 was assigned to the 16S rDNA gene sequence submission described by Kokilaramani 2020 (Kokilaramani et al., 2020). The SKR7 strain was recovered at 37 °C for 24 h in Luria-Bertani (LB) medium (5 g yeast extract, 10 g sodium chloride, 10 g tryptone, 15 g agar and final pH (at 25 °C) 7.5 ± 0.2 g/L (Himedia, Mumbai, India)). The well-grown bacterial inoculums were prepared to inoculate a single pure isolated colony in LB broth and shaking it for 24 h in a rotary shaker at (150 rpm) 37 °C.
2.2 Extraction of inhibitors (Mangrove Leaves)
The greenish raw mangrove plants of R.mangle and A.marina leaf samples were gathered from Parangipettai, Tamil Nadu, India (southeast coast of India, 11o29′25.6″ N and 79o45′57.1″ E) during January 2020. The leaves were cleaned with sterile double distilled water to detach dirt before being sent to the laboratory and the samples are left shadow environment at room temperature (RT) for a few days to dry. After the drying process, the leaves are shredded by mixer and followed for Soxhlet extraction, which entails mixing 10 g of powdered mangrove leaves added into 300 mL of methanol for 3 h at RT. From the obtained methanolic extraction of R.mangle (MERM) and A.marina (MEAM) the methanol was evaporated at 64.7 °C then the vaporized samples were stored in a refrigerator for future study (Punniyakotti et al., 2020; Zaher et al., 2022).
2.3 Minimal inhibitory concentration assay
The agar diffusion technique was used to estimate the minimal inhibitory concentration, that suppressed bacterial growth following overnight incubation at 37 °C. In the Mueller Hinton Agar (MHA) plate (Himedia) bacterial culture of SKR7 was swapped and profuse concentrations (10, 25, 50, and 100 ppm) of MERM and MEAM were added to the individual well of the test and control samples were then incubated at 37 °C for 16 h in an incubator. The zones of the sample plates are evaluated after the incubation time (Li et al., 2018).
2.4 Enumeration of total viable count
The SKR7 bacterial cells had been cultured overnight and it was spread on the nutrient agar plates at 37 °C for 24 h with and without inhibitors (MERM and MEAM) at various doses (10, 25, 50, and 100 ppm) and the number of colonies was calculated using the following equation to estimate the TVC values as a colony-forming unit after the inhibition period (Li et al., 2018).
2.5 Biofilm inhibition assay
Overnight cultures of SKR7 (2.2 × 106 CFU/mL) were grown in Mueller Hinton Broth (MHB) medium, 1 mL of MHB as control System 1(S1), System 2 (S2) 1 mL of MHB with 100 μL of (OD 0.500) SKR7 culture, System 3 (S3 (A)) 1 mL of MHB, 100 μL of (OD 0.500) SKR7 culture with added 25, 50, 100 ppm of MERM, System 4 (S4 (A)) 1 mL of MHB and 25, 50, 100 ppm of MERM. The well of S3 (B), S4 (B) was similar to S3 (A) and S4 (A) respectively, the A.marina was added instead of R.mangle. A 96-well microtiter plate was overnight incubated at 37 °C. The inoculum was removed and the 96-well microplate was rinsed in phosphate-buffered saline (PBS) followed by 15 min of incubation with 99% methanol then rinsed with PBS, added 200 μL of 0.1% crystal violet maintained for 5 mins at RT. Lastly, 200 μL of 95% ethanol was added and incubate on an orbital shaker for 30 mins then a UV–Visible spectrophotometer was used to absorbance the measured at 595 nm (Kokilaramani et al., 2021b).
2.6 Biocorrosion studies
2.6.1 Preparation of specimen
The mild steel 1010 (MS 1010) has a chemical composition of (wt %) was about C-0.03, Cu-0.093, Fe-99.64, Mn-0.16, Mo-0.002, Ni-0.04, P-0.01, S-0.026, Si- 0.002. The coupons were made out of MS 1010 and cut into small squares the size of 2.5 cm2 × 2.5 cm2 with a 2 mm hole in the top for weight loss measurement for hanging purposes and 1 cm2 × 1 cm2 for electrochemical analysis (working electrode (WE)). Coupons were polished using various grades of polishing papers to get a smooth and uniform exterior and then 0.3 μM alumina powder was used to polish the surface mirror finish. Each specimen was washed in ethanol and acetone before being air-dried and placed in a desiccator for further studies then the coupons were sterilized for 30 min using a UV light exposure before the experiment (Kokilaramani et al., 2021b).
2.6.1.1 Weight loss method (WL)
The autoclaved cooling tower water (CTW) was utilized as an electrolyte (medium) for the biocorrosion test and the water’s chloride level was kept constant then the specimens are weighted for initial weight before the experiment begins. Including both weight loss and electrochemical experiments which were carried out in triplicate, the coupons were placed on a 500 mL flask with 400 mL of CTW and nutrient broth (NB) 1% as noted a control System 1 (S1), System 2 (S2) 1% of NB was added CTW in 400 mL and 1 mL of (OD 0.500) 16 h SKR7 culture about 2.2 × 106 CFU/mL, System 3 (S3 (A)) 400 mL of CTW and 1% of NB with 1 mL of SKR7 and 25 ppm of MERM, System 4 (S4 (A)) 400 mL of CTW and 1% of NB with 25 ppm of MERM. The experimental systems S3 (B), S4 (B) were similar to S3 (A) and S4 (A) respectively, the A.marina was added instead of R.mangle and incubated at (37 °C) RT for 20 days in an immobile stage. The WL coupons were collected at the end of the biocorrosion study and then soaked in pickling solutions described as (Kokilaramani et al., 2021b) according to ASTM Standard G1-81. It was washed with distilled water before being utilized for the WL study. According to the National Association of Corrosion Engineers (NACE), the experiment of inhibition efficiency (IE) and surface coverage (θ) was evaluated by the following equation (Zulfareen et al., 2016).
where, W0 and W1 - weight loss of MS with and without inhibitor.
where, icorr and icorr(inh) – corrosion current density of MS with and without inhibitor.
2.7 FT-IR
After the weight loss study, biofilm formed coupons were thoroughly dried up and the dried scraped powdered samples were mixed with potassium bromide (KBr) (purified salt) to form a pellet. These pellets were analyzed using FT-IR spectra (PerkinElmer Spectrum IR Version 10.6.0) with wave numbers 400–4000 cm−1 and 8 cm−1 resolution and a 64 scans/spectrum scan rate. FT-IR was used for the analysis to find out the organic functional groups of biofilm layers on the coupons of a metal surface (Rajasekar et al., 2008).
2.8 GC–MS
The examination of inhibitors was attained on an Agilent 7890A GC and 5975C Mass Spectrometer instrument, with 1 μL (volume) of MERM and MEAM taken in the column HP5-MS (Long 30, interior diameter 0.25 μM, thickness 250 μM), the carrier gas is helium.
With 1.0 mL/min flow rate. The oven was programmed between initial temperature: 55 °C for 2 min, increased at 70 °C /min, detector temperature 225 °C, injector volume: 1 μL a carrier gas and total running time of 35.5 min. The compounds were identified by comparing the corresponding mass spectra with library data (NIST05.LIB) (ben Harb et al., 2020; Halim et al., 2014).
2.9 Electrochemical method
For electrochemical measurements, the WE is MS 1010 (surface area: 1.0 cm2), whereas the reference and counter electrodes are Ag/AgCl with KCl saturated calomel electrode (SCE) and a Titanium mesh, respectively. The electrolyte (medium) was obtained from the electrochemical experiment system and electrochemical polarization and impedance measurements were performed using the Metrohm Autolab Version of Nova 2.1.5 software. The frequency range of the applied measurement was 100,000 to 0.1 Hz. The polarization curves were attained by scanning anodically and cathodically from the open circuit potential (OCP) to + 200 and 200 mV SCE at a scan rate of 0.001 V/s. In the AC input, the frequency number is 50, and the amplitude is 0.01 mV, after which the final Rct readings are received. The Tafel polarization was set as a start −0.2 and 0.2 stop potentials and the scan rate was adjusted with a high (1 mA) to low (100 nA) current range by adding constant phase components to the relevant circuit (Kokilaramani et al., 2020).
3 Results
3.1 Minimal inhibition concentration (MIC) assay
The agar well diffusion technique was used to evaluate the minimal inhibitory concentration of MERM and MEAM leaf components. Various inhibitor doses (10, 25, 50, and 100 ppm) were tested, with MERM 2.92 ± 0.1 mm and MEAM 2.81 ± 0.1 mm of 25 ppm observed to be the most effective zone of inhibition. Both mangrove leaves were highly effective against the SKR7 bacterial growth. The total viable count (TVC) was revealed to be 25 ppm of MERM and MEAM suppressed bacterial growth in a random manner when compared to the other concentrations. In 25 ppm of MERM and MEAM, the lowest colonies were counted, whereas the control plate showed no colony growth.
3.2 Biofilm inhibition assay
For the biofilm inhibition assay, 25 ppm of inhibitors MERM and MEAM were employed with SKR7 strains in the biofilm studies using the crystal violet method in a 96-well microtiter plate. Biofilm development on well surfaces was greatly decreased by the presence of inhibitors MERM and MEAM at 25 ppm, this concentration was used for biocorrosion testing. The biofilm formation of SKR7 (S2) is 1.692, and the addition of 25 ppm of biofilm inhibitors MERM S3 (A) and MEAM S3 (B) is 0.534 and 0.619, respectively, according to the data shown in Fig. 1. The violet color fluctuation of wells is visible in the biofilm inhibition assay findings owing to the biofilm retaining crystal violet dye due to the high density of layers. It means that inhibitors can prevent the production of extracellular polymeric substances like proteins, carbohydrates, lipids, and so on (Ibrahim et al., 2021).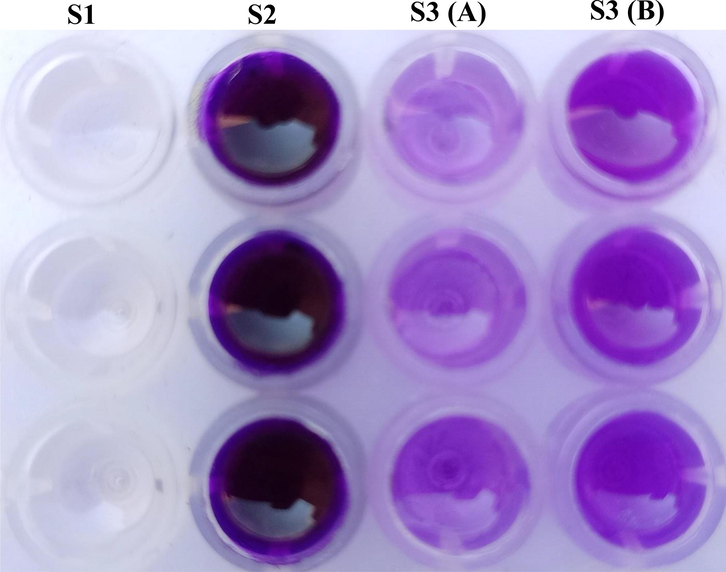
Biofilm Inhibition assay.
3.3 Biocorrosion studies
3.3.1 Weight loss measurement
The weight loss data for MS 1010 coupons placed in cooling tower wastewater with or without inhibitors along SKR7 were summarized in Table 2. Both the mangrove leaf (MERM and MEAM) inhibitors can inhibit the corrosion causing bacteria that have developed biofilm layers in cooling tower wastewater S3 (A) 80.33 ± 1and S3 (B) 86.80 ± 1 and the S2 430.89 ± 1 sample revealed that the presence of SKR7 can induce severe corrosion on the MS surface. The efficiency of corrosion inhibition at 25 ppm of MERM is 78%, and 25 ppm of MEAM is 75%, respectively.
System
Weight loss (mg)
Corrosion Rate (mm/y)
Inhibition Efficiency (%)
S1
27.66 ± 1
0.102
–
S2
430.89 ± 1
1.598
–
S3(A)
80.33 ± 1
0.298
81
S4(A)
12.57 ± 1
0.044
57
S3(B)
86.80 ± 1
0.322
80
S4(B)
12.13 ± 1
0.046
55
3.4 FTIR
The FTIR spectrum parameters of corrosion products including mangrove leaves (R.mangle and A.marina) are identical to alginate standards. Table 3 and Fig. 2 show the FTIR peak results for this experiment. The occurrence of biologically organic substances was validated then peaks were seen at 1054.36 cm−1 showing C—O stretch with the moderate intensity of Alcohols R—CH2—OH (1°) or C⚌C—CH(R)—OH and 612.57 cm−1 indicates the ≡C—H bend with a wide, solid density of Alkynes groups are presented, and this resonant peak shows the bacterial suppression (Okeniyi et al., 2019a; Shahin Lefteh et al., 2021). As detected by a biofilm inhibition study, the presence of mangrove leaves compounds absorption with the metal surface while reducing bacterial development.
Sample
Peak Values (cm−1)
Bond
Intensity
Functional groups
S1
3454.36
N—H stretch
weak-medium
Amides R—C(O)—NH—R
2926.26
C—H stretch
strong
Alkanes & Alkyls
S2
1652.43
C⚌O stretch
strong, broad
Amides
1393.30
CH3 C—H bend
medium
Alkanes & Alkyls
S3(A)
1532.40
N—O symmetric and asymmetric stretch
strong
Nitro compounds
C⚌C—NO2 or Ar—NO2
S4(A)
1054.36
C—O stretch
medium-strong
Alcohols R—CH2—OH (1°) or C⚌C—CH(R)—OH
612.57
≡C—H bend
strong broad
Alkynes
S3(B)
1560.81
N—H bend
medium or strong
Amides
1467.75
C—H bend
strong
Alkanes and Alkyls
S4(B)
1240.55
O⚌C—O—C stretch
strong-very strong
Esters Acetates
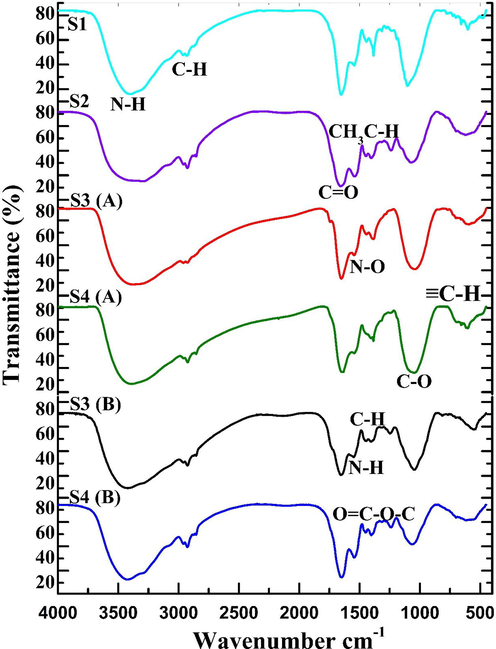
FTIR analysis for MS 1010 coupons system.
3.5 GC–MS analysis
The GC–MS analysis of methanolic extracts of mangrove leaves (R.mangle and A.marina) were identified substances provided in Fig. 3. In the GC–MS study of R.mangle the Myo-Inositol, 4-C-methyl was found at a significant constituent with a peak area of 94.36%, followed by 5,5,8a-Trimethyl-3,5,6,7,8,8a-hexahydro-2H-chromene, Nonanoic acid, Cyclohexane, ethyl, Pyrene, Cyclohexane, 1,2-dimethyl-, trans, Hexane, 3-methoxy was a prominent ingredient. A.marina of GC–MS analysis the Lupeol, trifluoroacetate was significant constituent with a peak area of 65.35% followed by beta.-Amyrin, Hexane, 3-methoxy, Cyclohexane, 1,4-dimethyl were found to be a minor amount. To suppress microbial activity, the Myo-inositol, 4-C-methyl and Lupeol-based compounds are used. The found inhibitory compounds are used to minimize biofilm development and to regulate the corrosion process in the cooling water system (Khattab et al., 2012).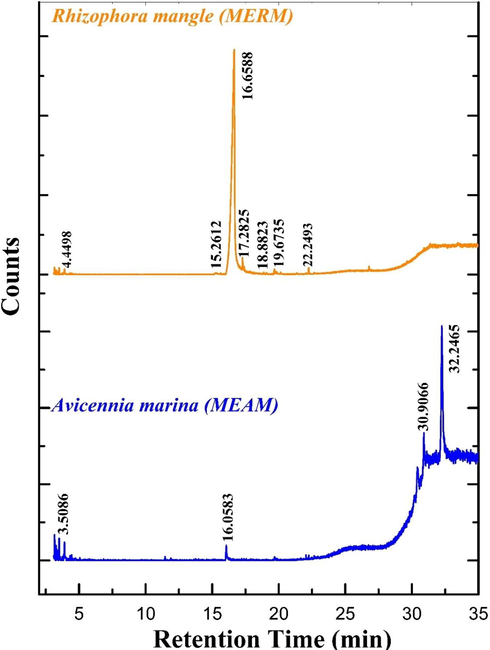
GCMS analysis for green inhibitors of mangrove plant Rhizophora mangle and Avicennia marina.
3.6 Electrochemical studies
3.6.1 Impedance spectroscopy
The results of electrochemical impedance measurements are shown in Table 4 and Fig. 4. Rs – Solution Resistance; Rct - Charge transfer resistance; CPE - Constant Phase Element; Y0 = C = The Capacitance; N - a constant value that is less than unity, 0 < n < 1.
System
Rs (Ωcm2)
Rct (Ωcm2)
CPE Y0
CPE N
S1
32.9
116
0.006682
0.8561
S2
32.3
76.3
0.004856
0.5114
S3(A)
42.5
547
0.007907
0.5355
S4(A)
46.0
226
0.010707
0.2965
S3(B)
55.7
438
0.003155
0.4022
S4(B)
62.6
218
0.003393
0.3605
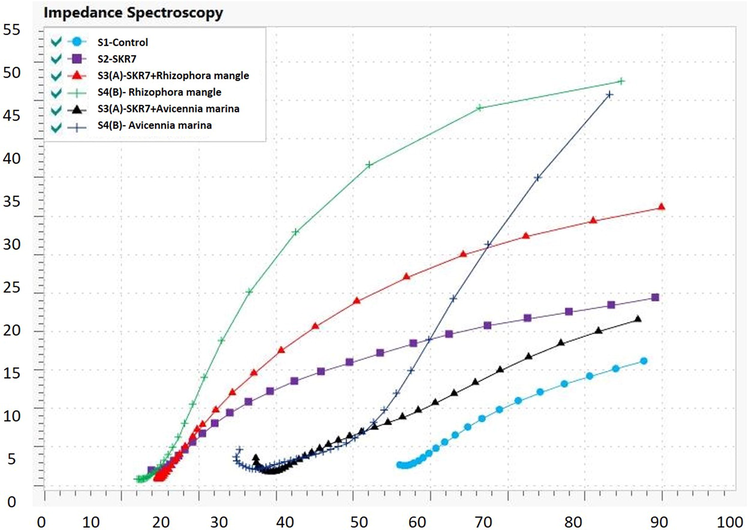
Impedance Spectroscopy for MS 1010 coupons collected from biocorrosion system.
The charge transfer resistance (Rct) of the experiential systems is S1 has 116 Ωcm2, while S2 has lower values 76.3 Ω cm2 absorbs a huge amount of current owing to the addition of SKR7 indicates that the bacteria potentially cause deterioration of the metal, inhibitory system S3 (A), S4 (A) of MERM are 547 Ω cm2, 226 Ω cm2 and S3 (B), S4 (B) of MEAM is 438 Ω cm2, 218 Ω cm2 correspondingly. The inclusion of mangrove leaves, MERM and MEAM, can reduce the current consumption level due to reduced corrosion rates because of the formation of protective layers that limit microbial adhesion on the metal surface. The green inhibitors prevent the microbe binding mechanism through a lone pair electron, hence preventing interaction of bacterial community and secretion of polymeric substances. The production of insoluble complexes as a result of interaction between iron cations caused by adsorption at the electrode/solution interface is attributed to the mechanism of action.
Both MERM and MEAM are mixed (anodic and cathodic) type inhibitors, according to the adsorption mechanism electron atoms of inhibitors (free lone pairs of electrons) such as nitrogen, oxygen, phosphorus and sulfur are vital in forming protective layers on the metal products (vacant-d-orbital). Table 5 and Fig. 5 are the result of Equivalent Circuit for MS 1010 coupons. The polarization absorption of electrochemical coupon S2 rapidly lowers the circuit rate, whereas S3 (A) and S3 (B) increased. Constant phase elements (CPE) have become widely used for generating acceptable impedance spectra. (Pino et al., 2001).
S1 – MS + CW (Control)
Element
Parameter
Value
Estimated Error (%)
Rs
R
32.883
2.370
Rp
R
115.9
7.646
CPE
Y0
0.0066829
11.467
N
0.85614
6.479
χ2
0.80532
S2 – MS+CW+SKR7
Element
Parameter
Value
Estimated Error (%)
Rs
R
32.294
2.559
Rp
R
76.322
8.266
CPE
Y0
0.0048562
19.362
N
0.51146
9.474
χ2
0.48948
S3 (A) - MS+CW+SKR7+MERM
Element
Parameter
Value
Estimated Error (%)
Rs
R
42.49
2.857
Rp
R
547.43
27.013
CPE
Y0
0.0079077
8.417
N
0.53554
7.581
χ2
0.8695
S4 (A) - MS+CW+MERM
Element
Parameter
Value
Estimated Error (%)
Rs
R
46.014
5.198
Rp
R
226.09
53.855
CPE
Y0
0.010707
22.356
N
0.29654
20.285
χ2
0.80767
S3 (B) - MS+CW+SKR7+ MEAM
Element
Parameter
Value
Estimated Error (%)
Rs
R
55.717
4.006
Rp
R
438.16
18.437
CPE
Y0
0.0031557
14.488
N
0.40225
9.432
χ2
0.84624
S4 (B)- MS+CW+ MEAM
Element
Parameter
Value
Estimated Error (%)
Rs
R
62.571
5.586
Rp
R
217.93
20.852
CPE
Y0
0.0033931
26.730
N
0.36052
16.871
χ2
1.1008
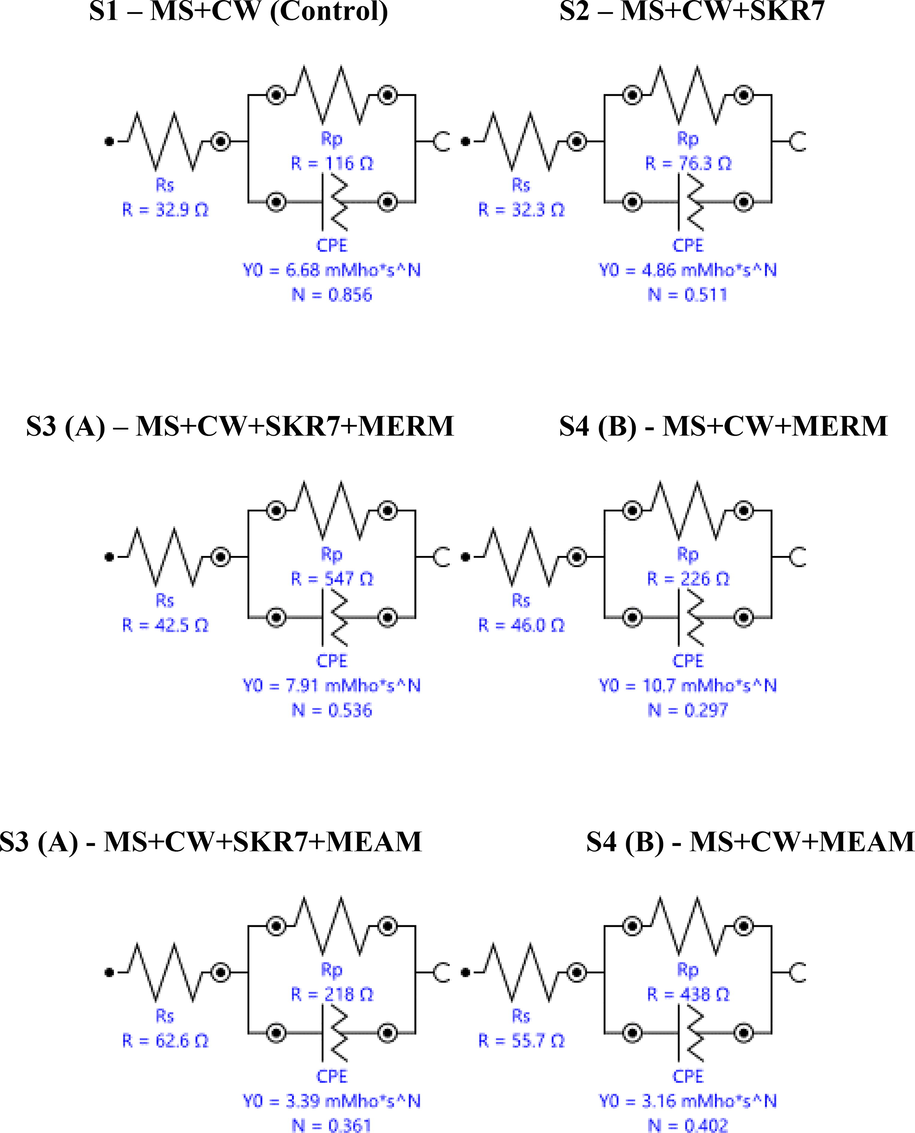
Equivalent Circuit for MS 1010 coupons collected from biocorrosion.
3.6.2 Linear polarization
The linear polarisation of MS 1010 coupons on cooling water is shown in Table 6 and Fig. 6. The bacterium SKR7 was tested on the MS coupons with and without inhibitors for anodic and cathodic polarisation curves. Ecorr- Corrosion Potential; icorr- Corrosion Current Density; βa - Anodic Beta coefficient; βc - Cathodic Beta coefficient; RP- Polarization Resistance; mV/dec – Millivolt/Decade.
System
Ecorr
(V)
icorr (μA/cm2)
βa
(mV/dec)
βc
(mV/dec)
Rp (KΩcm2)
Corrosion Rate
(mm/year)
S1
−0.57946
2.2114
0.043291
0.046952
304.59
0.1647
S2
−0.59815
6.0514
0.028579
0.031072
211.88
1.8065
S3(A)
−0.58472
1.5649
0.054324
0.060331
793.28
0.1843
S4(A)
−0.57651
1.2044
0.026398
0.028673
495.63
0.2425
S3(B)
−0.51204
2.0875
0.062683
0.096332
790.02
0.3545
S4(B)
−0.5285
1.4187
0.02232
0.026475
370.9
0.3731
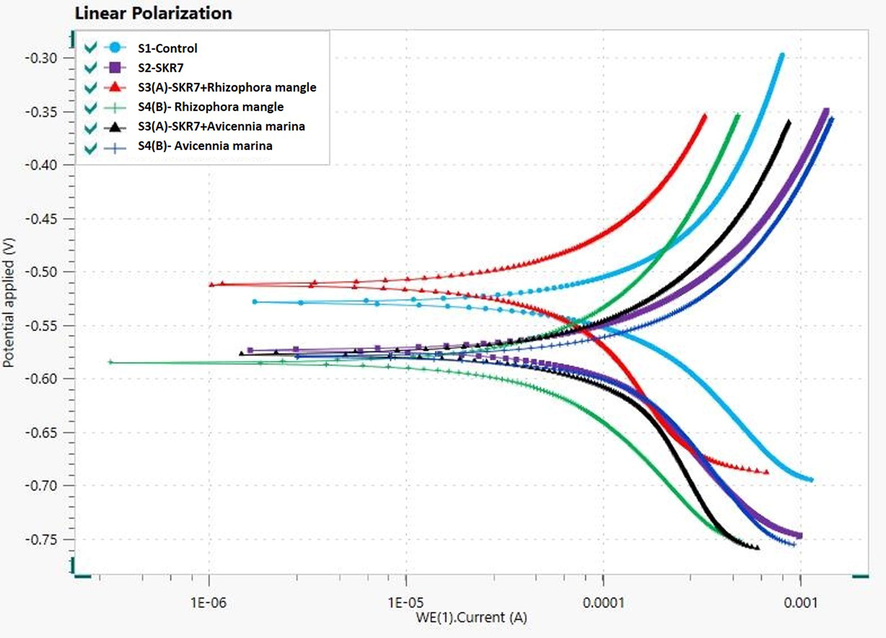
Linear polarization of electrochemical studies for MS 1010 coupons.
The Tafel polarization method is used to evaluate the surface coverage (θ) values of inhibitors to know the corrosion inhibition mechanism and adsorption between mild steel and inhibitor (Preethi Kumari et al., 2017). The corrosion current density (icorr) of S1 is 2.2114 μA/cm2, while S2 SKR7 has a 6.0514 μA/cm2 reached a high corrosion rate of 1.8065. The icorr value was lower in the presence of green inhibitors S3 (A), (B) showing 1.5649 μA/cm2, 2.0875 μA/cm2 and the corrosion rate was partially controlled at 0.1843 and 0.3545. As a result, the inhibitors can prevent both biofilm growth as well as corrosion. The S4 (A), (B) readings of icorr are 1.2044 μA/cm2, 1.4187 μA/cm2 and corrosion rate is 0.2425 and 0.3731 respectively. The S2 Ecorr and icorr values have the potential to rise to result in pitting corrosion. Based on the values of activation energy and free energy of adsorption, the Langmuir adsorption isotherms appear to represent the mechanism of adsorption. MERM and MEAM have been identified as efficient biofilm inhibitors and preventers. MERM is the best inhibitor of inhibiting efficiency than MEAM (Kokilaramani et al., 2020).
4 Discussion
The researchers are successfully finding green inhibitors to inhibit metal corrosion in various environments. Gelatin, gram flour, onion, organic honey, potato, plant roots, leaves, seeds, and floral gums have all been described as potential metal corrosion inhibitors (Argyropoulos et al., 2021). Mangrove plants have natural bioactive compounds that are effective against a variety of microbial pathogens and exhibit an alternate source of antimicrobial agents to combat biofilm formed by microbes. It has biochemically distinct, producing a varied spectrum of novel natural products as well as bioactive compounds that defend against microbial pathogens. The MERM and MEAM have been demonstrated to inhibit the activity of virulence factors such LasA protease, LasB elastase, protease, and pyocyanin pigment. Furthermore, bioactive proteins deactivate bacteria's Quorum Sensing (QS) signals and prevent bacterial communication. (Marzorati et al., 2019). (Ejikeme et al., 2013; Haldhar et al., 2021; Michael, 2014; Okeniyi et al., 2019a; Perencatan et al., 2011; Shah et al., 2011; Tan & Kassim, 2011; Umoren et al., 2018) are all reported, that the mangrove plants worked against corrosion and electrochemical studies provide a good inhibition efficiency result and the SEM, FTIR, GCMS analysis are detailed studies about the inhibitors compound. It was the first report to use mangrove leaves extract to reduce bacterial corrosion on MS 1010 in cooling tower wastewater.
5 Conclusion
In conclusion, this was reported to examine the microbial corrosion inhibition of mangrove trees of R.mangle and A.marina leaves against the B.megaterium strain. MERM and MEAM inhibitors effectively reduce bacterial accumulation by inhibiting biofilm formation caused by forbidden cell–cell communication. According to the results of the FT-IR studies, extract inhibitors are primarily responsible for the adsorption on the metal surface and suppress bacterial growth, which leads to avoiding the corrosion process. A weight loss study has confirmed that the SKR7 bacterium generates high corrosion on metals and plant-based substances prevent corrosion through an adsorption mechanism in which metal d-orbitals interact with inhibitory lone pair electrons. The mechanism process is based on the formation of a layer between the external corrosive medium and the substrate, which is generally provided by the coating structure by its chemical contents. Controlling the transit of corrosive ions becomes important to postpone and, hence, avoid a corrosive reaction once the corrosive medium has penetrated the coating over time. In the results of GCMS, plant compounds exhibit excellent antimicrobial efficacy against microorganisms. EIS and Tafel plots indicated that inhibitors substance to adsorption with metals surface and suppress both anodic and cathodic sites. A comprehensive study concluded that plant-based inhibitors act as excellent antibacterial activity against corrosion-causing bacteria and prevent the corrosion process in the cooling water system.
CRediT authorship contribution statement
Seenivasan Kokilaramani: Writing – original draft, Methodology. Jayaraman Narenkumar: Validation, Writing – review & editing. Mohamad S. AlSalhi: Resources, Funding acquisition, Writing – review & editing. Sandhanasamy Devanesan: Validation, Formal analysis, Writing – review & editing. Parthiba Karthikeyan Obulisamy: Validation, Writing – review & editing. Ramasamy Balagurunathan: Writing – review & editing, Formal analysis. Aruliah Rajasekar: Project administration, Supervision, Validation, Writing – review & editing.
Acknowledgment
The authors are grateful to the Researchers Supporting Project Number (RSP-2021/68), King Saud University, Riyadh, Saudi Arabia.
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to participate
Not applicable.
Consent for publication
All authors agreed to publish this version of the article.
Funding
Not applicable.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
References
- Green approach to corrosion inhibition of carbon steel by fucus spiralis extract in 1 m hcl medium. Biointerface Res. Appl. Chem.. 2022;12(5):7075-7091.
- [CrossRef] [Google Scholar]
- Effect of runoff from acid-sulfate soils on pneumatophores of the grey mangrove Avicennia marina. Marine Freshwater Res.. 2011;62(8):974-979.
- [CrossRef] [Google Scholar]
- Olive leaf extract as a green corrosion inhibitor of reinforced concrete contaminated with seawater. Arabian J. Chem. 2020
- [CrossRef] [Google Scholar]
- Biber, P., 2006. Measuring the effects of salinity stress in the red mangrove, Rhizophora mangle L. https://www.researchgate.net/publication/228340196
- Inhibitive effect of black pepper extract on the sulphuric acid corrosion of mild steel. Mater. Lett.. 2008;62(17–18):2977-2979.
- [CrossRef] [Google Scholar]
- Recent trends in environmentally sustainable Sweet corrosion inhibitors. J. Mol. Liq.. 2021;326
- [CrossRef] [Google Scholar]
- Corrosion inhibition of aluminium by Treculia Africana leaves extract in acid medium. Portugaliae Electrochimica Acta. 2013;30(5):317-328.
- [CrossRef] [Google Scholar]
- Moroccan, Mauritania, and senegalese gum Arabic variants as green corrosion inhibitors for mild steel in HCl: Weight loss, electrochemical, AFM and XPS studies. J. Mol. Liq.. 2022;347
- [CrossRef] [Google Scholar]
- Microbiologically-influenced corrosion of the electroless-deposited NiP-TiNi – Coating. Arabian J. Chem.. 2021;14(12)
- [CrossRef] [Google Scholar]
- Quorum sensing in water and wastewater treatment biofilms. J. Environ. Biol.. 2013;34:437-444.
- [Google Scholar]
- Adsorption and inhibition behavior of Avicennia marina for Zn metal in hydrochloric acid solution. Int. J. Electrochem. Sci.. 2017;12(12):11789-11804.
- [CrossRef] [Google Scholar]
- Dodigen 213-N as corrosion inhibitor for ASTM 1010 mild steel in 10% HCL. J. Appl. Electrochem.. 2009;39(8):1199-1205.
- [CrossRef] [Google Scholar]
- Anticorrosive properties of a green and sustainable inhibitor from leaves extract of Cannabis sativa plant: Experimental and theoretical approach. Colloids Surf., A: Physicochem. Eng. Aspects. 2021;614
- [CrossRef] [Google Scholar]
- A Study of Chemical Compounds in Rhizophora Apiculata. Open Conf. Proc. J.. 2014;4(1):108-110.
- [CrossRef] [Google Scholar]
- Phytochemical composition of Avicennia marina leaf extract, its antioxidant, antimicrobial potentials and inhibitory properties on Pseudomonas fluorescens biofilm. Egyptian J. Aquatic Res. 2021
- [CrossRef] [Google Scholar]
- Khattab, R. A., Gaballa, A., Zakaria, S. M., Allah, A., Ali, E.-S., Sultan Sallam, I., Temraz, T., 2012. Phytochemical Analysis of Avicennia marina and Rhizophora mucronata by GC-MS. In CATRINA (Vol. 7, Issue 1).
- Kokilaramani, S., Al-Ansari, M. M., Rajasekar, A., Al-Khattaf, F. S., Hussain, A. M. R., Govarthanan, M., 2021. Microbial influenced corrosion of processing industry by re-circulating waste water and its control measures - A review. In Chemosphere (Vol. 265). Elsevier Ltd. https://doi.org/10.1016/j.chemosphere.2020.129075.
- Bacillus megaterium-induced biocorrosion on mild steel and the effect of Artemisia pallens methanolic extract as a natural corrosion inhibitor. Arch. Microbiol.. 2020;202(8):2311-2321.
- [CrossRef] [Google Scholar]
- Characterization of methanolic extract of seaweeds as environmentally benign corrosion inhibitors for mild steel corrosion in sodium chloride environment. J. Mol. Liq.. 2021;340
- [CrossRef] [Google Scholar]
- Biocorrosion of mild steel and copper used in cooling tower water and its control. 3. Biotech. 2018;8(3)
- [CrossRef] [Google Scholar]
- Marchand, N., Collins, C.H., 2016. Synthetic Quorum Sensing and Cell-Cell Communication in Gram-Positive Bacillus megaterium 1 2. DOI: 10.1021/acssynbio.5b00099
- Green corrosion inhibitors from natural sources and biomass wastes. Molecules. 2019;24(1)
- [CrossRef] [Google Scholar]
- The corrosion inhibition of mild steel in sulphuric acid solution by flavonoid (Catechin) separated from Nypa Fruticans Wurmb leaves extract. Sci. J. Chem.. 2014;2(4):27.
- [CrossRef] [Google Scholar]
- Mohamad Ikhmal Wan Mohamad Kamaruzzaman, W., Amirah Mohd Nasir, N., Aiman Syafiq Mohd Hamidi, N., Yusof, N., Syaizwadi Shaifudin, M., Muiz Aniq Aiman Mohd Suhaimi, A., Athirah Badruddin, M., Adnan, A., Mohd Norsani Wan Nik, W., & Mohd Ghazali, M.S., 2021. 25 years of progress on plants as corrosion inhibitors through a bibliometric analysis using the Scopus database (1995-2020). Arabian J. Chem. 103655. https://doi.org/10.1016/j.arabjc.2021.103655.
- Anti-quorum sensing potential of the mangrove Rhizophora annamalayana. World J. Microbiol. Biotechnol.. 2013;29(10):1851-1858.
- [CrossRef] [Google Scholar]
- Rhizophora Mangle L. Leaf biochemical characterization: Natural-green total-corrosion inhibition prospect on concrete steel-reinforcement in 3.5% NaCI. Jurnal Teknologi. 2019;81(1):11-21.
- [CrossRef] [Google Scholar]
- Anticorrosion behaviour of Rhizophora mangle L. Bark-Extract on concrete steel-rebar in saline/marine simulating-environment. Sci. World J. 2019
- [CrossRef] [Google Scholar]
- New eco-friendly anti-corrosion inhibitor of purple rice bran extract for boiler quality steel: Experimental and theoretical investigations. J. Mol. Struct.. 2022;1251
- [CrossRef] [Google Scholar]
- Inhibitive Behaviour of Corrosion of Aluminium Alloy in NaCl by Mangrove Tannin. In. Sains Malaysiana. 2011;40(Issue 9)
- [Google Scholar]
- Volatile components of red mangrove bark (rhizophora mangle l.) from Cuba. J. Essent. Oil Res.. 2001;13(2):88-89.
- [CrossRef] [Google Scholar]
- Electrochemical measurements for the corrosion inhibition of mild steel in 1 M hydrochloric acid by using an aromatic hydrazide derivative. Arabian J. Chem.. 2017;10(5):653-663.
- [CrossRef] [Google Scholar]
- Anti-bacterial and anti-biofilm properties of green synthesized copper nanoparticles from Cardiospermum halicacabum leaf extract. Bioprocess Biosyst. Eng.. 2020;43(9):1649-1657.
- [CrossRef] [Google Scholar]
- Evaluation of bio-corrosion on carbon steel by bacillus megaterium in biodiesel and diesel oil mixture. J. Eng. Technol. Sci.. 2020;52(3):370-384.
- [CrossRef] [Google Scholar]
- Mangrove tannins and their flavanoid monomers as alternative steel corrosion inhibitors in acidic medium. Corros. Sci.. 2007;49(2):402-417.
- [CrossRef] [Google Scholar]
- Biodegradation of corrosion inhibitors and their influence on petroleum product pipeline. Microbiol. Res.. 2007;162(4):355-368.
- [CrossRef] [Google Scholar]
- Electrochemical behavior of Serratia marcescens ACE2 on carbon steel API 5L–X60 in organic/aqueous phase. Ind. Eng. Chem. Res.. 2008;47(18):6925-6932.
- [CrossRef] [Google Scholar]
- Role of inorganic and organic medium in the corrosion behavior of bacillus megaterium and pseudomonas sp. in stainless steel SS 304. Ind. Eng. Chem. Res.. 2011;50(22):12534-12541.
- [CrossRef] [Google Scholar]
- Acid corrosion inhibition of copper by mangrove tannin. Pigm. Resin Technol.. 2011;40(2):118-122.
- [CrossRef] [Google Scholar]
- Avicennia marina mediated synthesis of TiO2 nanoparticles: its antibacterial potential against some aquatic pathogens. Inorganic Nano-Metal Chem.. 2021;51(12):1775-1785.
- [CrossRef] [Google Scholar]
- Antimicrobial Activity of Endophytic Fungi Isolated from Avicennia marina Plant, Red Sea, Egypt. In. J. Microbiol. 2012;47
- [Google Scholar]
- A correlation study on the phenolic profiles and corrosion inhibition properties of mangrove tannins (Rhizophora apiculata) as affected by extraction solvents. Corros. Sci.. 2011;53(2):569-574.
- [CrossRef] [Google Scholar]
- Comparative studies on the corrosion inhibition efficacy of ethanolic extracts of date palm leaves and seeds on carbon steel corrosion in 15% HCl solution. J. Adhes. Sci. Technol.. 2018;32(17):1934-1951.
- [CrossRef] [Google Scholar]
- Organic Green Corrosion Inhibitors Derived from Natural and/or Biological Sources for Conservation of Metals Cultural Heritage. In: In Microorganisms in the Deterioration and Preservation of Cultural Heritage. Springer International Publishing. 2021.
- [CrossRef] [Google Scholar]
- Enhanced corrosion of 7075 alloy by the presence of Bacillus megaterium. Int. J. Electrochem. Sci.. 2016;11(11):9723-9733.
- [CrossRef] [Google Scholar]
- Peach Pomace Extract as Efficient Sustainable Inhibitor for Carbon Steel Against Chloride-Induced Corrosion. J. Bio- Tribo-Corrosion. 2021;7(1)
- [CrossRef] [Google Scholar]
- A combined computational & electrochemical exploration of the Ammi visnaga L. extract as a green corrosion inhibitor for carbon steel in HCl solution. Arabian J. Chem.. 2022;15(2)
- [CrossRef] [Google Scholar]
- Synthesis, characterization and corrosion inhibition efficiency of N-(4-(Morpholinomethyl Carbamoyl Phenyl) Furan-2-Carboxamide for brass in HCl medium. Arabian J. Chem.. 2016;9(1):121-135.
- [CrossRef] [Google Scholar]







