Translate this page into:
Experimental and computational phase behavior investigation for the CO2 + 1H, 1H-perfluorooctyl acrylate and CO2 + 1H, 1H-perfluorooctyl methacrylate systems at high pressure
⁎Corresponding author. hsbyun@jnu.ac.kr (Hun-Soo Byun)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Abstract
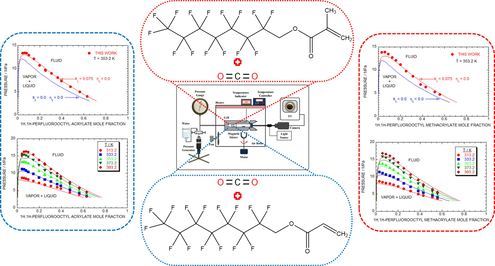
The thermodynamics equilibrium properties for the two-component mixture of 1H, 1H-perfluorooctyl acrylate (PFOA) and 1H, 1H-perfluorooctyl methacrylate (PFOMA) in supercritical CO2 were investigated. The phase behaviour of PFOA + SC-CO2 and PFOMA + SC-CO2 exhibits a type-I model. The vapor and liquid curves of as-proposed two-component models (PFOA + SC-CO2 and PFOMA + SC-CO2) were investiagted by experimentaly as well as numerically (Peng-Robinson equation of state). The as-proposed binary models (PFOA + SC-CO2 and PFOMA + SC-CO2) experimental results were shows a good fit with the numerical simulated results (Peng-Robinson equation of state).
Abstract
The solubility information of fluoro-monomer (meth) acrylate in organic solvents is an important factor affecting their use in numerous engineering practices. This study investigated the phase equilibria of 1H, 1H-perfluorooctyl acrylate (PFOA) and 1H, 1H-perfluorooctyl methacrylate (PFOMA) in supercritical CO2 using optical fiber and contact lenses. The solubility curves investigations were conducted at different temperatures (313.2 to 393.2) K and pressures (3.31 to 16.84) MPa, and the mole fraction of (0.032 to 0.630). Results revealed that the PFOA + SC-CO2 and PFOMA + SC-CO2 systems exhibited a type-I behavior. The RMSD (%) for the PFOA + SC-CO2 [kij = 0.075, ηij = 0.0], and PFOMA + SC-CO2 [kij = 0.075, ηij = 0.0] models using two factors determined at 353.2 K evaluated with the alterable parameters at each T were 5.08 %, and 5.36 %, respectively. The correlation of the experimental response for the PFOA + SC-CO2 and PFOMA + SC-CO2 two component models were examined using Peng-Robinson (PR) equation of state (EOS) involving two parameters (kij, ηij) base on a fluid mixture rule. Additionally, the critical properties (pc, Tc and ω) and vapor pressure of PFOA and PFOMA were assessed using the Joback-Lyderson group impact.
Keywords
High-pressure phase behavior
Carbon dioxide
PFOA
PFOMA
- EOS
-
Equation of state
- OF
-
Objective Function
- PR
-
Peng-Robinson
- vdW1
-
van der Waals one fluid mixing
Abbreviations
Nomenclature
- a Pa∙m6/mol2
-
Attraction factor
- b m3/mol
-
Co-volume factor
- BP MPa
-
Bubble point
- CP
-
Critical point
- DP
-
Dew point
- N
-
Number of the data
- p
-
MPa Pressure
- R
-
8.314462 J/(mol∙K) Universal gas constant
- T
-
K Temperature
- v
-
m3 mol−1 Molar volume
- x
-
Liquid phase mole fractions
Symbols
- α
-
Alpha function
- ω
-
Acentric factor
Greek letters
1 Introduction
Currently, the use and development of polymers, particularly in engineering application, such as coating sources, biomedical resources, aircraft materials, optics, and fibers, have increased. Fluoropolymers have demonstrated attractive and exceptional characteristic behaviors such as superior thermal stability, water resistance and high chemical stability. Particularly, polymers with lengthy fluoro-functional chains have attracted significant interest. The polymers with acrylate and (meth) acrylate repetition group (i.e., monomer) are primarily utilized in a variety of functions which are in medicine, glazing, oil additives, toy industry and building materials (Kirk-Othmer, 1981). Thus, it is essential to further understand the behavior of more knowledge of monomers with acrylate and (meth) acrylate unit in supercritical gases (Choi et al., 2016; Jang et al., 2015). Although few works have investigated the phase behavior of methacrylate-based polymers, the high-cost of fluorine-based monomers and their poor solubility have limited their application in various industries (Ritz and P. L_atalova, J. Kríz, J. Genzer, P. Vlcek, 2008; Mohammed et al., 2016; John et al., 2016; Kim et al., 2017).
There are many different approaches were reported for the green synthesis of chemicals (Wang et al., 2022; Meng et al., 2022; Wei et al., 2023), however the utilization of supercritical fluids technique is considered as economical and eco-friendly. The supercritical fluids (SCFs) techniques along with carbon dioxide (CO2) could be a better substitute in terms of several globally dangerous organic solvents presently applied in the engineering industry due to supercritical carbon dioxide (SC-CO2) is comparatively cheap, nonflammable, and nontoxic (Goetheer et al., 1999). However, polymers exhibit the inadequate miscibility in solvents, most polymers are miscible in SCFs. At various pressure (p), temperature (T) and concentration, the miscibility of polymer in SC-CO2 exposes the extraordinary disparity. Being ecologically benign solvent, Sc-CO2 is non-flammable, nontoxic, cheap solvent and also a by-product mass-produced in commercial processes such as hydrogen, ammonia, and ethanol plants (Cooper and Holmes, 1999; Carlota et al., 2007). Also, it has characteristics properties like zero dipole moment, low dielectric constant, quadrupole moment, easily attainable critical point with and etc. The two-component system containing Sc-CO2 has various practical functions and hence, to have information of phase equilibria of these mixtures is indispensable. So, the SCFs methodology been utilized to numerous industrial practices trade with various polymer such as anti-solvent precipitation, fine particles creation and polymerization (McHugh and Krukonis, 1994; Cooper, 2000; Tomasko et al., 2003). The lab-made examinations have been constantly published by Byun and their team people. They were discussed the thermodynamic properties of polymers phase behavior study under different supercritical fluids, such as carbon dioxide, ethylene, etc. (Ghoderao et al., 168 (2022); Byun, 2021; Dhamodharan et al., 110 (2022); Dhamodharan, 2022). McHugh and their group also reported various polymers thermodynamic properties under supercritical fluids, they were discussed experimental as well as computational analysis (Mallepally et al., 2016; Wu et al., 2014). Radosz’s team performed numerous research works to analyze the phase behavior properties of polymers under supercritical fluids (Albrecht et al., 1996; Folie et al., 1996), and Kiran’s team (Kiran, 2016).
In this work, we performed laboratory investigations on the phase equilibria for the PFOA + SC-CO2 and PFOMA + SC-CO2 mixtures at maximum pressures and elevated temperatures of approximately 16.84 MPa and 393.2 K, respectively. Our key fact of this research work is to analyze the thermodynamic properties (critical point, dew point, and bubble point) of the discussed two component models (PFOA + SC-CO2 and PFOMA + SC-CO2). The machinery performed in this experiment is a static type by synthetic view technique. The three-phases in presentation of these mixtures was not considered as it is beyond the scope of this work. The data of lab-made trails are correlated by utilizing the Peng Robinson equation of state (PR EOS) under van der Waals single fluid mixing rule (vdW1). The calculation results revealed that laboratory data and the estimated data of the two models were consistent. In addition, there is no other previous studies has been reported for the said system and based on the literature report, this is the first time we have performed the phase behavior examination of a binary model comprising of PFOA + SC-CO2 and PFOMA + SC-CO2 mixture.
2 Laboratory investigational section
2.1 Materials
1H, 1H-Perfluorooctyl acrylate (purity > 0.970; CAS 307–98-2) and 1H, 1H-Perfluorooctyl methacrylate (purity > 0.970; CAS 3934–23-4) were obtained from Alfa Aesar Company. Carbon dioxide (purity > 0.999) was purchased form Deok Yang Company. All the materials were utilized as obtained in this research work. The complete details of the utilized materials related to this study is presented in the Table 1. The schematic illustration of the investigated chemicals is presented in Fig. 1.
Chemical name
Mass fraction purity
Molecular formula
Source
CAS RN
Carbon Dioxide
> 0.999
CO2
Deok Yang Co.
124–38-9
1H, 1H-Perfluorooctyl Acrylate
> 0.970
C11H5F15O2
Alfa Aesar Co. (GC)
307–98-2
1H, 1H-Perfluorooctyl Methacrylate
> 0.970
C12H7F15O2
Alfa Aesar Co.
3934–23-4
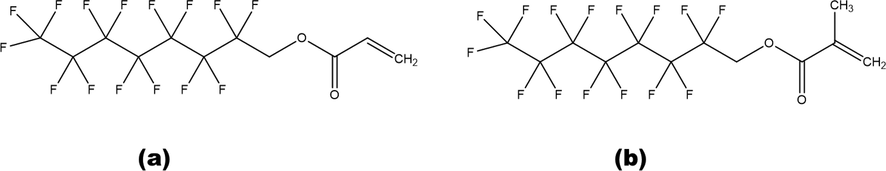
Chemical structure of (a) 1H, 1H-perfluorooctyl acrylate and (b) 1H, 1H-perfluorooctyl methacrylate.
2.2 Device setup and working process
The equipment operated to analyze the phase behavior of two different monomers under SC-CO2 were performed same as our previous works (Dhamodharan et al., 2022; Kwon et al., 2022; Ghoderao et al., 2022; Ghoderao et al., 2022; Choo et al., 2019). The essential component of the equipment consists of a variable-volume view (3 V) cell, and high-pressure (HP) generator. A thick and strong glass window is associated with the front of the 3 V cell to observe the phase changes. Normally, the thawed (liquid) monomers was passed through the cell roughly ± 0.002 g by the assist of a syringe; then the bare cell is cleansed by numerous trails with N2 gas to eliminate pinches of organic matters and unwanted air particles. Afterward, CO2 supplied to the operating 3 V cell roughly ± 0.004 g by the use of a HP bomb. Then the blend inside the 3 V cell compressed to the necessitous pressure by the assist of a wheel type piston.
The 3 V cell pressure was valued with a Heise gauge (having a highest pressure of 34.0 MPa, model CM-53920, made by Dresser Ind.) precise to ± 0.02 MPa. A digital meter (accurate to ± 0.005 %, model 7563, made by Yokogawa), was utilized to monitor the 3 V cell temperature and the maintained temperature was less than ± 0.2 K. A borescope (made by Olympus Corp) built-in with a camera is enclosed side to the glass window to observe phase changes via computer. The light diffusion in the 3 V cell was sustained by a fiberoptic wire associated to an elevated-density illuminator and the borescope. The monomers inside the 3 V cell were robustly blended by the support of a magnetic stirrer, which is operated by an external unit. Firstly, the monomer content compressed to a homogenous phase at steady temperature. The homogeneous phase inside the cell continued at the desired temperature for more than half an hour to accomplish phase equilibrium. Then, the 3 V cell pressure was slightly abridged till a two phases attains. The bubble and dew points were measured, while the first vapor bubble forms and while the earliest teeny mist observed inside the 3 V cell. By changing the pressure and temperature till the critical opalescence, the critical points are marked. The sketch demonstration of the equipment was presented in the Fig. 2.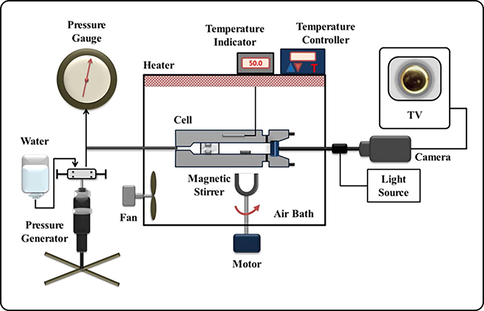
The detailed pictorial representation of experimental apparatus utilized in this research work (McHugh and Krukonis, 1994).
3 Results analysis
3.1 Experimentation
The laboratory trial data of the PFOA + SC-CO2 and PFOMA + SC-CO2 systems were investigated, and the experiment was replicated using the HP equipment at a temperature (T), pressure (p), and mole fraction (x) of 0.2 K, 0.02 MPa, and 0.002, respectively. The solubility isotherms of the PFOA + SC-CO2 and PFOMA + SC-CO2 systems with a change in temperatures from 313.2 K to 393.2 K were constructed depending on the outcomes with two sovereign factors with an estimated error of less than ± 0.1%.
The lab-based trail data of p, composition (x) isotherms at various temperatures, such as 313.2 K to 393.2 K and several pressures raise from 3.31 to 16.84 MPa for the PFOA + SC-CO2 system. Fig. 3a and Table 2 shows the lab-based trail data of PFOA + SC-CO2 system. The Fig. 3a, exposes that, described BP pressure rises with the rise in temperature which is attributes of type-I phase behavior (Byun, 2017; Lee et al., 2018). As shown in Fig. 3a, the critical p of the PFOA + SC-CO2 system is rising in regard to the increasing temperature, that is the early critical pressure was noted at 13.43 MPa in regard to 353.2 K, then the elevated pressure value of 16.36 MPa was accomplished at 393.2 K, which evidence that the critical point (CP) of the PFOA + SC-CO2 system rises with respect to raising T.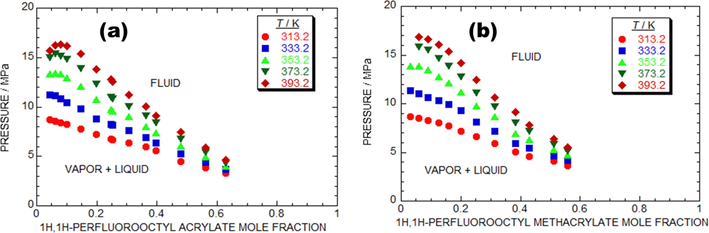
The laboratory experimental results of pressure-composition isotherm for as-proposed binary models. (a) × 1H, 1H-perfluorooctyl acrylate + (1-x) SC-CO2 model at various temperatures such as ●, 313.2.K; ■, 333.2.K; ▲, 353.2.K; ▾, 373.2.K; ◆, 393.2.K. (b) × 1H, 1H-perfluorooctyl methacrylate + (1-x) SC-CO2 model at various temperatures such as ●, 313.2 K; ■, 333.2 K; ▲, 353.2 K; ▾, 373.2 K; ◆, 393.2 K.
1H, 1H-Perfluorooctyl Acrylate Mole Fraction
pa / MPa
Transitionb
Ta / K = 313.2
0.044
8.72
BP
0.063
8.59
BP
0.081
8.42
BP
0.101
8.24
BP
0.147
7.80
BP
0.198
7.20
BP
0.249
6.74
BP
0.253
6.68
BP
0.307
6.35
BP
0.363
5.97
BP
0.396
5.55
BP
0.478
4.48
BP
0.563
3.83
BP
0.630
3.31
BP
T / K = 333.2
0.044
11.22
BP
0.063
11.15
BP
0.081
10.83
BP
0.101
10.45
BP
0.147
9.80
BP
0.198
8.79
BP
0.249
8.22
BP
0.253
8.20
BP
0.307
7.59
BP
0.363
6.93
BP
0.396
6.38
BP
0.478
5.24
BP
0.563
4.41
BP
0.630
3.72
BP
T / K = 353.2
0.044
13.35
DP
0.063
13.43
CP
0.081
13.32
BP
0.101
12.93
BP
0.147
12.10
BP
0.198
10.72
BP
0.249
9.74
BP
0.253
9.61
BP
0.307
8.99
BP
0.363
8.04
BP
0.396
7.35
BP
0.478
6.06
BP
0.563
4.96
BP
0.630
4.00
BP
T / K = 373.2
0.044
15.00
DP
0.063
15.37
DP
0.081
15.11
BP
0.101
14.79
BP
0.147
13.86
BP
0.198
12.31
BP
0.249
10.95
BP
0.253
10.79
BP
0.307
10.04
BP
0.363
9.07
BP
0.396
8.38
BP
0.478
6.79
BP
0.563
5.41
BP
0.630
4.35
BP
T / K = 393.2
0.044
15.70
DP
0.063
16.25
DP
0.081
16.36
CP
0.101
16.17
BP
0.147
15.41
BP
0.198
13.83
BP
0.249
12.76
BP
0.253
12.55
BP
0.307
11.21
BP
0.363
10.03
BP
0.396
9.14
BP
0.478
7.45
BP
0.563
5.85
BP
0.630
4.62
BP
Similarly, the PFOMA + SC-CO2 model was also carried out with the various temperatures and difference pressures same as PFOA + SC-CO2 model. The studied lab-based investigational data of the PFOMA + SC-CO2 system is presented in the Fig. 3b and Table 3. The Fig. 3a, exposes that, described BP pressure rises with the rise in temperature which is attributes of type-I BP (Byun, 2017; Lee et al., 2018). In addition, there was a notable decrease in the solubility of the monomer in CO2 with increase T at a fixed pressure range. Nevertheless, as shown in Fig. 3 (a-b), both two-component models demonstrate type-I model and possibly there were no three phases achieved at various operated temperatures in both the models.
1H, 1H-Perfluorooctyl Methacrylate Mole Fraction
pa / MPa
Transitionb
Ta / K = 313.2
0.032
8.67
BP
0.059
8.49
BP
0.091
8.24
BP
0.125
8.04
BP
0.159
7.70
BP
0.200
7.18
BP
0.252
6.59
BP
0.315
5.90
BP
0.382
5.04
BP
0.430
4.59
BP
0.513
4.07
BP
0.557
3.66
BP
T / K = 333.2
0.032
11.38
BP
0.059
11.05
BP
0.091
10.66
BP
0.125
10.31
BP
0.159
9.90
BP
0.200
9.29
BP
0.252
8.14
BP
0.315
7.14
BP
0.382
5.93
BP
0.430
5.41
BP
0.513
4.69
BP
0.557
4.21
BP
T / K = 353.2
0.032
13.84
DP
0.059
13.87
CP
0.091
13.48
BP
0.125
12.72
BP
0.159
12.10
BP
0.200
11.17
BP
0.252
9.79
BP
0.315
8.66
BP
0.382
6.90
BP
0.430
6.31
BP
0.513
5.31
BP
0.557
4.76
BP
T / K = 373.2
0.059
15.83
BP
0.091
15.49
BP
0.125
14.66
BP
0.159
13.90
BP
0.200
12.76
BP
0.252
11.14
BP
0.315
9.69
BP
0.382
8.00
BP
0.430
7.21
BP
0.513
5.86
BP
0.557
5.10
BP
T / K = 393.2
0.059
16.84
BP
0.091
16.60
BP
0.125
16.04
BP
0.159
15.35
BP
0.200
14.21
BP
0.252
12.48
BP
0.315
10.66
BP
0.382
9.14
BP
0.430
7.83
BP
0.513
6.41
BP
0.557
5.55
BP
3.2 Computational analysis
The PR EOS model was utilized for comparison with the obtained laboratory trail values such as the BP, dew point (DP), and CP of the isothermal phase behavior, and the formulas used are expressed below:
The vdW1 rules are expressed below,
Compounds
Mw
Tb / K
Tc / K
pc / MPa
ω
Carbon Dioxide
44.01
304.2
7.38
0.225
1H, 1H-Perfluorooctyl Acrylate
454.13
481.6a
614.7
1.28
0.708
1H, 1H-Perfluorooctyl Methacrylate
468.16
493.0b
625.6
1.20
0.710
Fig. 4a shows the comparison of the laboratory trail and PR EOS assessed values of the PFOA + SC-CO2 system at T = 353.2 K. The red dots indicate the laboratory trail data counts, and the red mark displays a good fit of computational analysis data obtained by the aid of PR EOS. The blue marks imply outcomes obtained using
, whereas the red mark indicate outcomes obtained using the matched
and
. The independent interaction parameter of the PR EOS exposes good match with the laboratory trail data at 353.2 K. The PR EOS, adjusted value for PFOA + SC-CO2 system are
= 0.0,
= 0.075 at 353.2 K.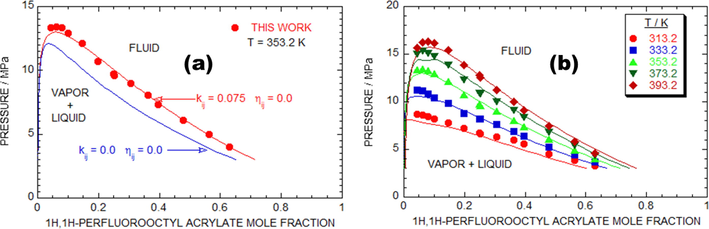
The comparative study analysis of experimental and PR EOS simulated results of as-proposed binary mixtures models. (a) Plot of pressure against mole fraction to illustrate the comparison of experimental data (symbols) for the (1H, 1H-perfluorooctyl acrylate + SC-CO2) {(1-x) SC-CO2 + x (C11H5F15O2)} model with calculations obtained from the PR EOS by
and
set equal to zero (dashed lines), and
equal to 0.075 and
equal to 0.0 (solid lines) at T-353.2 K. (b) 1H, 1H-perfluorooctyl acrylate + SC-CO2 {(1-x) SC-CO2 + x (C11H5F15O2)}.
Fig. 4b, demonstrating the competitive discussion of lab-made trail and computational analysis data of the p-x isotherms at various temperatures (313.2 to 393.2) K for the PFOA + SC-CO2 system engaged using the adjusted data of the sovereign factors of and calculated at T = 353.2 K. In agreement with the Fig. 4b, the lab-based investigational results, and the PR EOS adjusted data of the PFOA + SC-CO2 system shows a decent agreement. The RMSD (%) of all the laboratory trial T was 5.08 % for 70 data counts.
Fig. 5a shows a comparative study analysis of laboratory trail and simulated p-x isotherms for the PFOMA + SC-CO2 system at T = 353.2 K. The red dots denote to lab-based investigational results, and the red line indicates that the laboratory results were well matched with the simulated values obtained using PR EOS. The blue mark indicates the outcomes obtained using
whereas the red marks represent the outcomes obtained using the matched
and
. The two component interaction elements of the PR EOS model were in good agreement with the laboratory trail data at 353.2 K. The PR EOS adjusted data for PFOMA + SC-CO2 system at T = 353.2 K were
= 0.0,
= 0.075.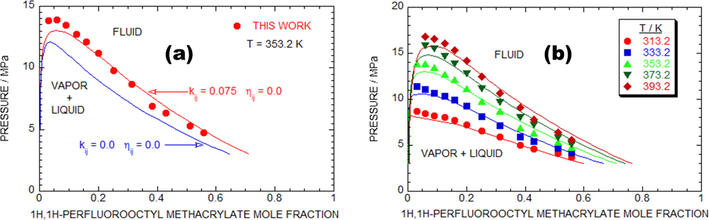
The comparative study analysis of experimental and PR EOS simulated results of as-proposed binary mixtures models. (a) Plot of pressure against mole fraction to illustrate the comparison of experimental data (symbols) for the (1H, 1H-perfluorooctyl methacrylate + SC-CO2) {(1-x) SC-CO2 + x (C12H7F15O2)} model with calculations obtained from the PR EOS by
and
set equal to zero (dashed lines), and
equal to 0.075 and
equal to 0.0 (solid lines) at T-353.2 K. (b) 1H, 1H-perfluorooctyl methacrylate + SC-CO2 {(1-x) SC-CO2 + x (C12H7F15O2)}.
Fig. 5b displays that the comparative discussion of laboratory trail response and computational p-x isotherms at various temperatures from 313.2 K to 393.2 K for the PFOMA + SC-CO2 system engaged the adjusted data of the sovereign factors of and lofted at T = 353.2 K. The image revealed that the obtained laboratory trail results and the PR EOS optimized data of the PFOMA + SC-CO2 system were well matched. The RMSD (%) of all the investigated T was 5.36 % for 58 data counts.
Fig. 6a shows the PR EOS predicted critical curve (CC) for the PFOA + SC-CO2 system. The red marks in Fig. 6a indicate the vapor p of the pure PFOA and CO2, which was assessed using the Lee-Kesler approach (Peng and Robinson, 1976; Poling et al., 2001). The red dots emphasize the CP of the pure CO2 and PFOA. The area above the blue mark indicated that the system is in one phase (fluid), and the area below indicates two phases (vapor and liquid). The blue dashed lines represent the information obtained from the PR EOS for the PFOA + SC-CO2 model at T = 353.2 K.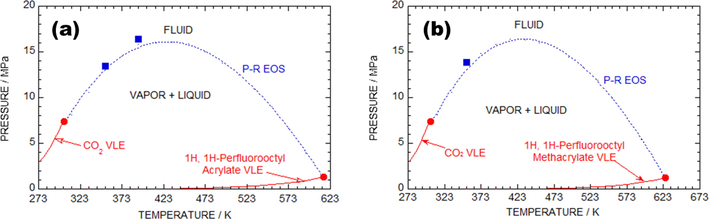
Plot of pressure against temperature for the as-proposed binary models. (a) 1H, 1H-perfluorooctyl acrylate + SC-CO2 model. The solid lines and the solid circles represent the (vapor + liquid) lines and the critical points for pure CO2 and 1H, 1H-perfluorooctyl acrylate. The solid squares are critical points determined from isotherms measured in this work. (b) 1H, 1H-perfluorooctyl methacrylate + SC-CO2 model. The solid lines and the solid circles represent the (vapor + liquid) lines and the critical points for pure CO2 and 1H, 1H-perfluorooctyl methacrylate. The solid squares are critical points determined from isotherms measured in this work.
Fig. 6b shows the PR EOS estimated CC for the PFOMA + SC-CO2 system. The red lines in Fig. 6b correspond to the vapor p of the pure PFOMA and CO2, which was assessed by the help of Lee-Kesler approach (Peng and Robinson, 1976; Poling et al., 2001). The red dots indicate the CP of the pure CO2 and PFOMA. In Fig. 6b, the area above the blue marking represents one phase (fluid), and the below the mark represents two phases (vapor–liquid). The consequential CCs expose type-I system. The blue marking relates to the estimated data accomplished through PR EOS of the PFOMA + SC-CO2 system at T = 353.2 K. Moreover, the examined CCs of the as-proposed two-component models were consistent with the laboratory trail data and computational response, which was obtained using PR EOS with two alterable parameters.
4 Conclusions
In this study, high-pressure laboratory-obtained trial data of the (p, x) isotherm for the (PFOA + SC-CO2 and PFOMA + SC-CO2) two-component models assessed using a synthetic approach equipment (3 V cell) at various temperatures from 313.2 to 393.2 K and a maximum p of ∼ 24 MPa were obtained. Both (PFOA + SC-CO2 and PFOMA + SC-CO2) systems did not exhibit three phases (liquid + liquid + vapor) at any operated temperatures. The PR EOS predicted the phase behavior for the (PFOA + SC-CO2, and PFOMA + SC-CO2) systems using two temperature sovereign mixture synergy factors. The critical curves between the simulated and laboratory-obtained trail data were consistent base on the two-adjustable parameters of the PR EOS. The RMSD (%) for the PFOA + SC-CO2 [ = 0.075, = 0.0], and PFOMA + SC-CO2 [ = 0.075, = 0.0] models using two factors determined at 353.2 K evaluated using the alterable parameters at each T were 5.08 %, and 5.36 %, respectively.
CRediT authorship contribution statement
Duraisami Dhamodharan: Investigation, Writing – original draft. Min-Soo Park: Experimental Assistant, Simulation. Suhail Mubarak: Experimental Assistant, Simulation. Hun-Soo Byun: Conceptualization, Visualization, Supervision, Project administration, Writing – review & editing.
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. 2021R1A2C2006888).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Phase equilibria of saturated and unsaturated polyisoprene in sub-and supercritical ethane, ethylene, propane, propylene, and dimethyl ether. Fluid Phase Equilib.. 1996;117:84-91.
- [CrossRef] [Google Scholar]
- High pressure phase equilibria for the ry mixture of CO2 + 3-phenyl propionitrile and CO2 + 2-phenyl butyronitrile systems. J. Supercrit. Fluids. 2017;120:218-225.
- [CrossRef] [Google Scholar]
- Phase separation of two-and three-component solution for the poly (pentyl acrylate-co-methyl methacrylate) + compressed solvents and copolymer preparation by supercritical dispersion polymerization. J. Ind. Eng. Chem.. 2021;99:158-171.
- [CrossRef] [Google Scholar]
- Boron trifluoride catalyzed polymerisation of 2-substituted-2-oxazolines in supercritical carbon dioxide. Green Chem.. 2007;9:948-953.
- [CrossRef] [Google Scholar]
- Phase behavior for the poly(alkyl methacrylate) + supercritical CO2 + DME mixture at high pressures. Kor. J. Chem. Eng.. 2016;33:277-284.
- [CrossRef] [Google Scholar]
- Phase equilibria and cloud-point behavior for the poly (2-phenylethyl methacrylate) in supercritical CO2 with monomers as co-solvent. J. CO2 Utilization. 2019;31:215-225.
- [CrossRef] [Google Scholar]
- Polymer synthesis and processing using supercritical carbon dioxide. J. Mater. Chem.. 2000;10:207-234.
- [CrossRef] [Google Scholar]
- Synthesis of molded monolithic porous polymers using supercritical carbon dioxide as the porogenic solvent. Adv. Mater.. 1999;11:1270-1274.
- [CrossRef] [Google Scholar]
- Pradnya Prabhakar Ghoderao, Hun-Soo Byun, Binary Equilibrium Behavior for the N, N-Dimethylaniline and N, N-diethylaniline in Supercritical Carbon Dioxide. J. Mol. Liq.. 2022;357:119112
- [CrossRef] [Google Scholar]
- D Dhamodharan, Pradnya NP Ghoderao, Cheol-Woong Park, Hun-Soo Byun. Experimental and computational investigation of two-component mixtures for the alkyl (ethyl, propyl and butyl) oleate in supercritical carbon dioxide. J. Ind. Eng. Chem., 110 (2022) 367-374. 10.1016/j.jiec.2022.03.013.
- Bubble and Dew- Point Measurement of Mixture for the 1H,1H,2H-Perfluoro-1-octene and 1H,1H,2H,2H- Perfluoro-1-octanol in Supercritical CO2. New J. Chem.. 2022;46:7271-7278.
- [CrossRef] [Google Scholar]
- Phase equilibria of poly (ethylene-co-vinyl acetate) copolymers in subcritical and supercritical ethylene and ethylene vinyl acetate mixtures. Fluid Phase Equilib.. 1996;120:11-37.
- [CrossRef] [Google Scholar]
- Co-solvent concentration impact on the cloud point behavior of 2-and 3-ingredient systems of the poly (tridecyl methacrylate) in supercritical CO2. New J. Chem.. 2022;46:2300-2308.
- [CrossRef] [Google Scholar]
- Phase behavioral study of binary systems for the vinyl benzoate, vinyl pivalate and vinyl octanoate with carbon dioxide at high-pressure. J. Mol. Liq.. 2022;358:119131
- [Google Scholar]
- PNP. Ghoderao, D. Dhamodharan, and H-S. Byun. Binary Mixture Phase Equilibria for the Vinyl Laurate, Vinyl Methacrylate and Vinyl Propionate under High Pressure Carbon Dioxide. The J. Chem. Thermodys., 168 (2022) 106746. .
- Opportunities for process intensification using reverse micelles in liquid and supercritical carbon dioxide. Chem. Eng. Sci.. 1999;54:1589-1596.
- [CrossRef] [Google Scholar]
- Phase behavior for the poly(2-methoxyethyl acrylate) + supercritical solvent + cosolvent mixture and CO2 + 2-methoxyethyl acrylate system at high pressure. Kor. J. Chem. Eng.. 2015;32:958-966.
- [CrossRef] [Google Scholar]
- Synthesis and microstructural properties of the scaffold based on a 3-(trimethoxysilyl) propyl methacrylate-POSS hybrid towards potential tissue engineering applications. RSC Adv.. 2016;6:66037-66047.
- [CrossRef] [Google Scholar]
- Electrochemical capacitor performance of 2-(trimethylsilyloxy) ethyl methacrylate-derived highly mesoporous carbon nanofiber composite containing MnO2. J. Electroanal. Chem.. 2017;801:403-409.
- [CrossRef] [Google Scholar]
- Supercritical fluids and polymers-The year in review-2014. J. Supercrit. Fluids. 2016;110:126-153.
- [CrossRef] [Google Scholar]
- Kirk-Othmer, Encyclopedia of Chemical Technology, John Wiley & Sons, New York, 1981
- Optimization Techniques with Fortran. New York: McGraw-Hill; 1973.
- Phase equilibrium study of binary systems for the PGME + CO2 and PGMEA + CO2 mixture at high pressure by experimentally and computationally. Kor. J. Chem. Eng.. 2022;39:2783-2791.
- [CrossRef] [Google Scholar]
- Phase behavior for the 2-(trimethylsilyloxy) ethyl methacrylate and 3-(trimethoxysilyl) propyl methacrylate in supercritical carbon dioxide. Fluid Phase Equilib.. 2018;462:1-5.
- [CrossRef] [Google Scholar]
- Phase behavior and densities of propylene + hexane binary mixtures to 585 K and 70 MPa. J. Chem. Eng. Data. 2016;61:2818-2827.
- [CrossRef] [Google Scholar]
- Supercritical Fluid Extraction: Principles and Practice 2nd. Boston: Butterworth-Heinemann; 1994.
- MoC nanocrystals confined in N-doped carbon nanosheets toward highly selective electrocatalytic nitric oxide reduction to ammonia. Nano Res.. 2022;15:8890-8896.
- [CrossRef] [Google Scholar]
- A.H. Mohammed, M.B. Ahmad, N.A. Ibrahim, N. Zainuddin, Synthesis and monomer reactivity ratios of acrylamide with 3-(trimethoxysilyl) propyl methacrylate and tris (methoxyethoxy) vinylsilane copolymers. Polimery, 61 (11-12) (2016) 758-765. .
- A new two-constant equation of state. Ind. Eng. Chem. Fundamen.. 1976;15:59-64.
- [CrossRef] [Google Scholar]
- B.E. Poling, J.M. Prausnitz, and J.P. O’Connell. Properties of gases and liquids. McGraw-Hill Education. 2001
- P. Ritz, P. L_atalova, J. Kríz, J. Genzer, P. Vlcek, Statistical copolymers of 2-(trimethylsilyloxy) ethyl methacrylate and methyl methacrylate synthesized by ATRP. J. Polym. Sci., Part A: Polym. Chem., 46 (2008) 1919-1923.
- A review of CO2 applications in the processing of polymers. Ind. Eng. Chem. Res.. 2003;42:6431-6456.
- [CrossRef] [Google Scholar]
- Polycrystalline SnSx nanofilm enables CO2 electroreduction to formate with high current density. Chem. Commun.. 2022;58:7654-7657.
- [CrossRef] [Google Scholar]
- A dual-functional Bi-doped Co3O4 nanosheet array towards high efficiency 5-hydroxymethylfurfural oxidation and hydrogen production. Chem. Commun.. 2023;59:442-445.
- [CrossRef] [Google Scholar]
- Architectural effects on the solution behavior of linear and star polymers in propane at high pressures. Ind. Eng. Chem. Res.. 2014;53:10133-10143.
- [CrossRef] [Google Scholar]







