Translate this page into:
Extraction and characterization of total phenolic and flavonoid contents from bark of Swietenia macrophylla and their antimicrobial and antioxidant properties
⁎Corresponding author at: Department of Biotechnology, Manipal of Institute of Technology, MAHE, Manipal, 576104, Karnataka, India. subbalaxmi.s@manipal.edu (Subbalaxmi Selvaraj)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
In the current study, the bioactive compounds such as total phenols, flavonoids, hydrolyzable tannins, and gallic acid were extracted from the bark of Swietenia macrophylla using four different solvents such as ethanol, methanol, acetone, and water. Among them, acetone exhibited the highest contents of bioactive compounds. To optimize the extraction process, a statistical approach was adopted using the central composite design (CCD) of response surface methodology (RSM). The five parameters at five different levels were chosen in the design of the experiments. A total of 32 experimental runs given by the design were fitted into the second-order regression model equation. The analysis of the model shows the best fit of the experimental data with an R2 of 0.9971 and a model F-value of 191.73. The optimal conditions of acetone concentration (56 %), the volume of acetone (22 mL), agitation speed (173 rpm), extraction temperature (31 °C), and extraction time (28 h) were noted from desirability function and showed a 2.0-fold increase in the contents of bioactive compounds when compared to unoptimized conditions. Further, the antimicrobial activity of 5 % (w/v) extract was tested against two-gram positive strains Bacillus sp, and Staphylococcus aureus, and two-gram negative strains Escherichia coli, and S. marcescens. The extract exhibited the 21 mm and 18 mm clear zone of diameter with 5 mm standard disc against the gram-positive strains tested whereas no clear zone was found against gram-negative strains. Finally, the antioxidant property was electrochemically analyzed using cyclic voltammetry and Differential pulse voltammetry, which confirmed the presence of multiple antioxidants in the extract.
Keywords
Solvent extraction
Central composite design
Antimicrobial activity
Antioxidant property
Cyclic voltammetry
Differential pulse voltammetry
1 Introduction
Swietenia macrophylla is the binomial name of the big-leaf mahogany or Honduran mahogany. The plant species belongs to the Meliaceae family and is native to South America, Mexico, and Central America. The plant is also found and cultivated in regions such as Singapore, the Philippines, Malaysia, Hawaii, and southern parts of India. The wood is characterized as being dense and strong and has been used extensively to produce furniture, doors, boats, musical instruments, and other purposes. The use of various parts of S. macrophylla has been widespread in folk medicine to treat diseases due to its pharmacological activities. Through the upregulation by p53 in HCT116 colorectal carcinoma cells, samples of S. macrophylla exhibit mitochondrial-mediated apoptosis-inducing capabilities (Goh et al., 2014), which is integral in treating tumors. S. macrophylla also exhibits antimicrobial, antioxidant effects, antidiabetic, anti-inflammatory, and antimutagenic activities (Goh et al., 2014; Moghadamtousi et al., 2013). Other reports also suggest activities such as antidiarrheal, anti-infective anti-nociceptive, acaricidal antiviral, antimalarial, antifeedant, hypolipidemic, and heavy metal phytoremediation activity (Moghadamtousi et al., 2013). Many parts of S. macrophylla contain important medical compounds, a significant part being the bark which has indicated high levels of total phenolic content.
Total phenolic compound (TPC) refers to the concentration of phenolic content present in plant substrates. They are secondary metabolites containing an aromatic ring bearing at least one hydroxyl group (Tungmunnithum et al., 2018). Phenolic compounds are considered integral plant constituents due to their redox properties (Soobrattee et al., 2005). The use of diet sources rich in natural phenolic compounds has been prevalent due to their antioxidant activity. Due to the presence of hydroxyl groups which can act as electron donors, phenolic compounds exhibit antioxidant action (Bendary et al., 2013). It was also observed that these compounds stimulate the synthesis of endogenous antioxidant molecules in cells, further adding to their antioxidant properties (Côté et al., 2010). Reports also suggest that phenolic compounds inhibit free radicals, decompose peroxides, inactivate metal ions, and inhibit oxygen scavenging in biological systems, further preventing an oxidative disease burden (Babbar et al., 2015). Due to the ease of use, wide applicability, and high efficiency, solvent extraction is one of the most used methods for the extraction of compounds. The extraction of TPC was conducted in this study via the use of solvents such as water, ethanol, methanol, and acetone on dried and ground plant samples. Based on the physical and chemical characteristics, the efficiency of extraction varies on the solvent and method used, as indicated by the quantification of TPC by the Folin-Ciocalteu test. The simplicity and low cost of this method make solvent extraction a favorable method for use. It is also seen that under different temperature and pH conditions, the polarities of solvents change, allowing us to optimize our operating conditions for maximum efficiency. This is the first report on the extraction of bioactive compounds from the bark of S. macrophylla, which represents a high yield of total phenolic, flavonoids, and hydrolyzable tannins with antimicrobial and antioxidant properties.
Phenolic compounds (PCs) are widely used in the food processing industry due to their antimicrobial activity and anti-putrefaction properties acting on fruits and vegetables. Food products are fortified with various naturally occurring PCs to increase their shelf life. Also, the antioxidant property of PCs can reduce the concentration and inhibit the activity of reactive oxygen species (ROS). This helps in ensuring safe food intake and lesser spoilage of food at a lower cost and without the use of synthetic additives (Martillanes et al., 2017). Further, PCs have been used in the packaging of meat, fish, and oils. Compounds such as tannins interact with proteins by either hydrogen bonds or non-covalent bonds and can be developed into biofilms that exhibit antimicrobial activity against Listeria innocua and E. coli and also exhibit antioxidant properties, whereas gallic acid and caffeic acid inhibit the growth of Bacillus subtilis and S. aureus (Cano et al., 2020). PCs also possess the capability of absorbing ultraviolet radiation as they contain chromophores in their structure. This property is the basis of UV protection in cosmetic products, with compounds such as quercetin, resveratrol, and hydroxycinnamic acids termed sun protection factor (SPF). Owing to their free radical scavenging property, PCs can effectively reduce the wrinkling of the skin and enhance the whitening of skin with no major side effects or irritation (Albuquerque et al., 2021). Furthermore, PCs from tea and oak bark have been used as natural dyes in the textile industry over synthetic dyes due to their high biodegradability and no additional cost (Bonet-Aracil et al., 2016). Due to their multiple activities that provide protection and maintenance of good health, phenolic compounds are a widely discussed potential pharmaceutical agent. Most of the activity is focused on the antioxidation and radical scavenging properties, PCs exhibit inhibitory activity against cancer, Alzheimer’s disease, diabetes, inhibition of DNA, protein, or lipid oxidation, among others (Albuquerque et al., 2021).
Antioxidants are the reducing agents which could be easily oxidized at the inert electrodes. It was found from the literature that compounds with low oxidation potential exhibit high antioxidant activity (Keffous et al., 2016). Ascorbic acid and phenolic compounds are the common antioxidant compounds found in plants. In this present study, the antioxidant activity of the plant extract was studied using Cyclic Voltammetry and Differential Pulse Voltammetry. The lower the oxidation potential of components of extracts higher would be the antioxidant capacity. The lower the oxidation potential, the easier it is to donate electrons to the system, resulting in free radicals (Arulpriya et al., 2010; SousaT et al., 2010).
Extraction of bioactive compounds from plant cells is one of the most important steps in using the sample as a source for use in pharmaceutical or industrial uses. The use of solvent extraction has been ubiquitous in designing economical and sustainable models for extraction. The solvent used for extraction first penetrates through the solid substrate, solubilizes any soluble solutes into the solvent, and diffuses the solutes out of the solid substrate. The solvent can then be treated to extract the solutes via various methods (Zhang et al., 2018). The selection of solvent can be done based on solubility, cost, safety, and selectivity. Further, inter-miscibility and the polarity of the solvents are essential to be considered while focussing on the extraction of specific compounds. The optimization of the solvent extraction processes via methods such as Central composite design (CCD) and response surface methodology is essential in understanding the optimal conditions for the extraction of specific bioactive compounds and designing an optimal protocol for conducting the extraction. Further, it is essential in obtaining the maximum yield of bioactive compounds and comparing the experimental values with the predicted model, indicating the validity of the model (Gullón et al., 2017). The electrochemical methods are rapid and robust when compared with the chemical methods of analysis of the antioxidant property. The time required for optimization is lesser. Because there are fewer chemicals used in the analysis, waste management is minimal (Arteaga et al., 2012).
2 Materials and methods
2.1 Plant material
The bark of S. macrophylla was collected from the botanical garden located in Manipal, Karnataka-India (13°22′11″ N, 74°47′6″ E). The bark was dried at room temperature in shades, milled and sieved (300-µm particle size); powdered plant materials were stored in an airtight container at room temperature and preserved under darkness to avoid oxidation of compounds (Rhazi et al., 2019; Muñiz-Márquez et al., 2013).
2.2 Reagents and standards
Ethanol, methanol, acetone, and distilled water were used as extraction solvents. These solvents were of analytical grade. Rhodanine, Bovine serum albumin (BSA), sodium dodecyl sulfate (SDS), tri-ethanolamine, potassium hydroxide, sodium nitrite, aluminum chloride, tannic acid, sodium hydroxide, Rutin, sodium carbonate, Folin-Ciocalteau reagent, ferric chloride, and phosphate buffer solution were used as reagents in this study and all procured from Hi-media Laboratories Pvt. ltd., Mumbai-India and Sigma Aldrich Co. LLC, Mumbai-India.
2.3 Solvent extraction
The one gram of powdered bark was taken in four different tubes, to which 20 mL of 50 %v/v different solvents such as methanol, ethanol, and acetone were added individually, whereas in the fourth tube 20 mL of pure distilled water was added. Then the tubes were kept for extraction in the rotary shaker for 48 h at 32 °C and 170 rpm. All experiments were carried out independently to screen the best extraction solvent. Thereafter, the extraction mixture was centrifuged for 10 min at 4 °C and 4000 rpm. After centrifuge, the extract (supernatant) was separated and the solvent was evaporated in the water bath at temperatures equivalent to the solvent’s boiling point till the volume of extract was reduced to half. After evaporation, the extracts were collected in vials and preserved at 4 °C for further analysis of the contents of total phenolic compounds, gallic acid, hydrolyzable tannic acid, and flavonoids (Muñiz-Márquez et al., 2013; Naima et al., 2015; Makkar, 2003). All the experiments were carried out in triplicates and average values with standard deviations were reported.
3 Estimation of bioactive compounds
3.1 Estimation of total phenolic compound
The total phenolic compound (TPC) of the S. macrophylla bark extracts was estimated by using the Folin-Ciocalteu method (Blainski et al., 2013). Briefly, 0.2 mL plant extract diluted to 1 mL with distilled water was mixed with 10 mL Folin-Ciocalteau reagent (10 %v/v). After 5 min, 8 mL of 7.5 %w/v of aqueous sodium carbonate was added to the mixture and incubated at room temperature for 2 h. After incubation, the reaction mixture was read at 710 nm using UV-spectrophotometer against a blank without extract. The amount of total phenolic content in extract was expressed as mg of gallic acid equivalents (GAE)/ gram dry weight of plant material (gdw).
3.2 Estimation of total flavonoid content
The total flavonoid content (TFC) of the S. macrophylla bark extracts was estimated by using the spectrophotometric method (Kim et al., 2003; Zhishen et al., 1999). Briefly, 0.2 mL plant extract diluted five times with distilled water was mixed with 2.4 mL of distilled water. To this, 0.3 mL of 5 % sodium nitrite was added, mixed well, and incubated for 5 min. Then, 0.3 mL of 10 % Aluminum chloride was added, mixed well, and incubated for 6 min. Finally, 2 mL of 1 M sodium hydroxide was added, mixed in the reaction mixture, and read the absorbance at 510 nm. using UV-spectrophotometer against a blank without extract. The amount of TFC in the extract was expressed as mg of rutin equivalents/gram dry weight of plant material (gdw) i.e. mg rutin/gdw.
3.3 Estimation of hydrolysable tannic acid
The hydrolyzable tannic acid content of the S. macrophylla bark extracts was estimated by using the protein precipitation method of Ann-Hagerman and Larry-Butler (Ann-Hagerman and Larry-Butler, 1978). Briefly, 0.2 mL extract was diluted to 1 mL with distilled water and mixed with 2 mL of BSA solution. Incubated for 15 min at 30 °C, then centrifuged at 4 °C and 7000 rpm for 15 min. After centrifuge, the supernatant was discarded and to the pellet 4 mL of SDS-tri ethanolamine solution was added to dissolve the pellet. Further, 1 mL of ferric chloride solution was added, mixed immediately, and incubated for 15 min at 30 °C. After incubation, the absorbance was read using UV-spectrophotometer at 510 nm against a blank without extract. A standard curve was attained by finding the absorbance of standard solutions of tannic acid with concentrations ranging from 0.1 to 1 mg/mL.
3.4 Estimation of gallic acid content
The gallic acid content of the S. macrophylla bark extracts was estimated by using the spectrophotometric method (Sharma et al., 2000). Briefly, 0.25 mL extract was mixed with 0.25 mL citrate buffer (0.05 M and pH 5.0) and 0.3 mL of methanolic rhodanine, mixed well, and incubated for 5 min at 30 °C. After 5 min, 0.2 mL of 0.5 N potassium hydroxide was added and incubated at 30 °C for a further 5 min. Thereafter, 4 mL of distilled water was added and incubated at 30 °C for 10 min. Finally, the absorbance was read using UV-spectrophotometer at 520 nm against a blank without extract (i.e. instead of extract, citrate buffer was added). A standard curve was attained by finding the absorbance of standard solutions of 0.05 mM gallic acid in citrate buffer and expressed in mg/mL.
4 Optimization of solvent extraction of a total phenolic compound by central composite design (CCD)
Based on the preliminary studies, we aim to optimize the extraction of TPC from the plant material S. macrophylla bark using the solvent acetone. For this objective, CCD of Response surface methodology (RSM) was adopted to optimize the five process parameters for extractions of total phenolic compound, hydrolyzable tannin acid, gallic acid, and total flavonoids. The five process parameters were: Acetone concentration (A), %v/v; Volume of acetone (B), mL; Agitation speed (C), rpm; Extraction temperature (D), °C; and Extraction time (E), h. The independent parameters with their levels and codes are shown in Table 1. The experimental design matrix developed by the statistical software MINITAB 17.0 (Trial version) is shown in Table 2, which is comprised of 32 experimental runs with five different levels comprising six center points, 16 cube points, and 10 axial points. The levels of each parameter were chosen based on the preliminary studies (Fig. 1). The value of total phenolic content (response, Y) of the 32 different runs was fitted to a second-order polynomial equation as follows:
A, Acetone concentration (%v/v); B, Volume of acetone (mL); C, Agitation speed (rpm); D, Extraction temperature (°C); and E, Extraction time (h).
Parameters
Coded levels
−2
−1
0
+1
+2
A: Acetone concentration, %v/v
30
40
50
60
70
B: Volume of acetone, mL
10
15
20
25
30
C: Agitation speed, rpm
70
120
170
220
270
D: Extraction temperature, °C
24
28
32
36
40
E: Extraction time, h
0
12
24
36
48
Run
A
B
C
D
E
TPC Yield, mg GAE/gdw
Experimental
Predicted
1
60 (+1)
15 (-1)
120 (-1)
36 (+1)
36 (+1)
5.35 ± 0.021
4.39
2
40 (-1)
25 (+1)
120 (-1)
36 (+1)
36 (+1)
1.32 ± 0.014
0.07
3
40 (-1)
15 (-1)
220 (+1)
36 (+1)
36 (+1)
7.37 ± 0.022
7.21
4
40 (-1)
25 (+1)
220 (+1)
36 (+1)
12 (-1)
3.46 ± 0.026
3.08
5
50 (0)
20 (0)
170 (0)
32 (0)
24 (0)
49.53 ± 0.046
49.25
6
50 (0)
20 (0)
170 (0)
40 (+2)
24 (0)
12.65 ± 0.011
14.48
7
50 (0)
20 (0)
170 (0)
32 (0)
0 (-2)
16.59 ± 0.022
16.72
8
70 (+2)
20 (0)
170 (0)
32 (0)
24 (0)
28.29 ± 0.034
29.43
9
50 (0)
20 (0)
170 (0)
32 (0)
24 (0)
49.53 ± 0.046
49.25
10
60 (+1)
25 (+1)
120 (-1)
36 (+1)
12 (-1)
12.11 ± 0.016
10.93
11
50 (0)
20 (0)
170 (0)
32 (0)
48 (+2)
20.92 ± 0.027
22.48
12
60 (+1)
15 (-1)
120 (-1)
28 (-1)
12 (-1)
18.29 ± 0.024
18.18
13
40 (-1)
15 (-1)
120 (-1)
28 (-1)
36 (+1)
7.99 ± 0.016
7.67
14
60 (+1)
15 (-1)
220 (+1)
28 (-1)
36 (+1)
16.58 ± 0.041
16.77
15
50 (0)
30 (+2)
170 (0)
32 (0)
24 (0)
20.71 ± 0.051
22.71
16
40 (-1)
15 (-1)
220 (+1)
28 (-1)
12 (-1)
11.25 ± 0.048
11.94
17
50 (0)
20 (0)
70 (-2)
32 (0)
24 (0)
23.75 ± 0.062
25.91
18
60 (+1)
25 (+1)
220 (+1)
36 (+1)
36 (+1)
41.97 ± 0.031
41.09
19
50 (0)
20 (0)
170 (0)
32 (0)
24 (0)
49.53 ± 0.046
49.25
20
60 (+1)
25 (+1)
120 (-1)
28 (-1)
36 (+1)
36.33 ± 0.032
35.28
21
60 (+1)
25 (+1)
220 (+1)
28 (-1)
12 (-1)
19.57 ± 0.037
19.54
22
50 (0)
20 (0)
270 (+2)
32 (0)
24 (0)
26.53 ± 0.038
26.06
23
50 (0)
20 (0)
170 (0)
32 (0)
24 (0)
49.53 ± 0.046
49.25
24
50 (0)
20 (0)
170 (0)
32 (0)
24 (0)
49.53 ± 0.046
49.25
25
40 (-1)
15 (-1)
120 (-1)
36 (+1)
12 (-1)
7.56 ± 0.011
7.10
26
50 (0)
10 (-2)
170 (0)
32 (0)
24 (0)
8.54 ± 0.021
8.23
27
40 (-1)
25 (+1)
120 (-1)
28 (-1)
12 (-1)
20.47 ± 0.044
19.93
28
50 (0)
20 (0)
170 (0)
32 (0)
24 (0)
49.53 ± 0.046
49.25
29
30 (-2)
20 (0)
170 (0)
32 (0)
24 (0)
6.89 ± 0.011
7.45
30
50 (0)
20 (0)
170 (0)
24 (-2)
24 (0)
28.87 ± 0.053
28.73
31
60 (+1)
15 (-1)
220 (+1)
36 (+1)
12 (-1)
1.42 ± 0.033
1.47
32
40 (-1)
25 (+1)
220 (+1)
28 (-1)
36 (+1)
3.12 ± 0.026
2.88
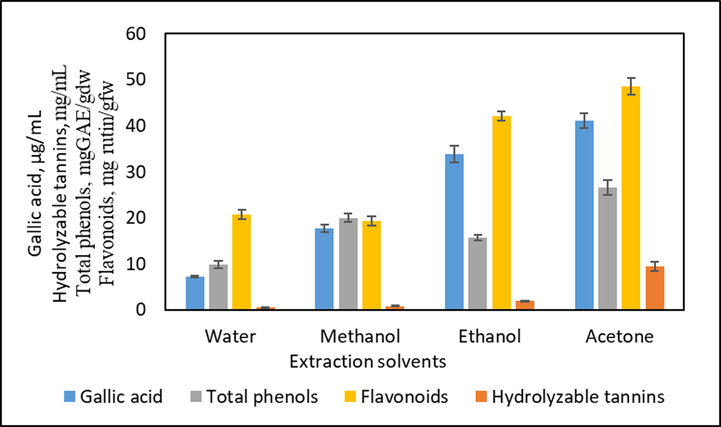
Effect of different solvents on the extraction of total phenolic compounds from the bark of S. macrophylla.
5 Characterization of extracted total phenolic compounds
5.1 Thin-layer chromatography (TLC)
In this study, the resolution of hydrolyzable tannins, gallic acid, total phenols, and flavonoids in the bark extract of S. macrophylla were analyzed using TLC as described elsewhere (Das et al., 2019; Banu and Nagarajan, 2014). The silica gel sheet of dimensions 6 cm × 15 cm and thickness of 175–225 µm (Merck, TLC Silica gel 60 F254) was used to perform the TLC. The straight line parallel to a smaller dimension was drawn 1 cm from the end of the plate with the pencil on either side. The two spots were marked with a 2 cm distance on the straight line on one side only while keeping the margin of 1 cm between a wall of the plate and spots. These spots were useful to load the plant extracts and standard for analysis. The silica gel sheets were activated by soaking the sheet with methanol and then drying at 110 °C for 10 min. The shots of bark extract of S. macrophylla and specific standard were spotted individually on the activated silica gel plate with the help of a wing micropipette and placed in a chromatographic chamber comprising different solvent systems. The spotted plates are placed in such a way that the spots do not touch the solvent. The chamber was closed with the lid and allowed to stand for a sufficient amount of time to separate the phytochemicals. Once the solvent reaches the second line, plates were removed and air-dried at room temperature. The specific spraying reagents were sprayed, the colored spots developed were marked, and dried and their distance was measured from the center of the spot. The retention factor Rf of the sample was calculated using the formula shown below:
The composition of the specific solvent system (i.e. mobile phase) and the type of spraying reagents used to separate the phytochemical constituents qualitatively of bark extracts of S. macrophylla is shown in Table 3 as described elsewhere (Banu and Nagarajan, 2014).
Phytoconstituents
Stationary phase
Mobile phases
Spraying reagent
Standard
Phenols
Precoated Silica gel sheets
Ethyl acetate: Toluene: Formic acid (2.2: 1.1: 1.1)
FeCl3 reagent
Gallic acid
Flavonoids
Ethyl acetate: Butanol: Formic acid (2.5: 1.5: 0.5)
AlCl3 reagent
Rutin
Tannins
Methanol: Water (6: 4)
FeCl3 reagent
Tannic acid
Gallic acid
Ethyl acetate: Butanol: Formic acid (5:4:1)
FeCl3 reagent
Gallic acid
5.2 Fourier transform infrared spectroscopy (FTIR) spectroscopy
Structural characterization of plant extract was carried out by Fourier Transform Infrared spectroscopy (SHIMADZU). The IR peaks were analyzed to confirm the presence of functional groups.
6 Application studies
6.1 Antimicrobial activity
The antimicrobial activity of bark extract of S. macrophylla was tested against two strains of Gram-positive bacteria (Bacillus species and S. aureus) and two strains of Gram-negative bacteria (E. coli and S. marcescens). These strains were procured from the culture collection of the Institute of Microbial Technology, Chandigarh, India. All the above-mentioned strains are pre-cultured overnight in nutrient broth media at 32 °C and 180 rpm.
Next, the agar well diffusion method was adopted to check the antimicrobial activity of plant extract as described elsewhere (Gonelimali et al., 2018; Mostafa et al., 2018). The 0.1 mL of overnight grown strains were spread plated on sterilized and solidified nutrient agar plates individually. The wells of size 0.25 cm in diameter were drilled using the sterile cork borer. Then, 0.1 mL of steam-sterilized plant extract (5 %w/v) was added to the respective wells and placed in the refrigerator for 30 min to diffuse well the plant extract into the agar. Later, the plates were incubated at 37 °C for 24 h. The antibacterial activity was calculated by measuring the clear zone formation around the well using a Vernier caliper. Tannin acid and gallic acid at a concentration of 0.1 %w/v were taken as a positive control.
6.1.1 Determination of minimum inhibitory concentration (MIC)
The MICs of acetone extracts of S. macrophylla bark were estimated by loading the different concentrations of extracts ranging between 5 % (w/v) to 20 % (w/v) into the wells of nutrient agar plates spread with individual pathogenic strains as mentioned above. Further, the loaded nutrient agar plates were incubated for 24 to 72 h at 37 °C in the incubator. The lowest concentration of an extract with a maximum zone of growth inhibition on the agar plate was noted and which gives the values of MIC.
6.2 Antioxidant property studies
Electrochemical analysis of antioxidant property of plant extract was executed with SP 150 Biologic potentiostat with a three-electrode system. Ag/AgCl and platinum electrodes were used as reference and counter electrodes respectively. A glassy carbon electrode of 3 mm diameter was used as the working electrode. Cyclic voltammetry (CV) and differential pulse voltammetry (DPV) studies were carried out to check the antioxidant property. Stainless steel was also used as the working electrode in CV. The extracts were analyzed using 0.1 M phosphate buffer at pH 7. The extracts were prepared by dissolving 5 mL of the plant extract in 25 mL of methanol and ultrasonication was done for 30 min (40 kHz frequency) at room temperature. Post ultrasonication, the extract was subjected to CV and DPV by adding 200 mg/mL of extract concentration to 20 mL of 0.1 M PBS buffer (pH 7.0). The concentration of the extract was increased from 23.5 µM to 235 µM. CV was performed to analyze electrochemical responses between 0.0 and 1.25 V potential window. Scan rates varied between 10 and 100 mV/s. DPV was conducted between − 0.50 V to 1.00 V potential window, with a pulse amplitude of 20 mV and pulse width of 50 ms.
7 Results and discussion
7.1 Impact of different extraction solvents
The total phenolic contents of the S. macrophylla bark were extracted by using different commonly used solvents such as water, methanol, ethanol, and acetone at 50 % v/v and the results are shown in Fig. 1. The results specified that the use of different solvents resulted in different extraction yields of bioactive compounds. 50 % acetone extract showed a higher yield of all the bioactive compounds quantified, which could be due to the high solubility of these compounds in acetone. The highest concentrations of bioactive compounds such as TPC, flavonoids, hydrolyzable tannins, and gallic acid were found to be 26.537 ± 1.56 mg GAE/gdw, 48.584 ± 1.76 mg Rutin/gdw, 9.491 ± 1.03 mg/mL, and 41.11 ± 1.534 µg/mL respectively in 50 % acetone extract. The lowest concentrations of these compounds were observed in water extract (Fig. 1). The choice of significant solvents plays an important role in the extraction of bioactive compounds and their applications in food products and therapeutics (Lasano et al., 2019). Solvents and their polarities will influence the solubility of bioactive compounds in a sample. The solvent penetration into the plant materials and extraction of bioactive compounds from them is more efficient with solvents having similar polarities of compounds (Dhanani et al., 2017; Mohammedelnour et al., 2017). Hence, the effect of different solvents on extraction was carried out to select the most influential solvent to recover phenolic compounds. In this study, the order of high yield of bioactive compounds by using solvents was as follows:
Acetone > Ethanol > Methanol > Water.
Dirar et al (Dirar et al., 2019) reported that the acetone and ethanol solvents are efficient in extracting a higher amount of phenolic and flavonoid compounds from plant sources. Our results are in good agreement with the results reported (N. F. Lasano NF, N. S. Ramli, A. H. Hamid, R. Karim, M. S. Dek, R. Shukri, 2019; Dhanani et al., 2017).
7.2 Optimization of solvent extraction of a total phenolic compound by central composite design (CCD)
The five factors acetone concentration (A), the volume of acetone (B), agitation speed (C), extraction temperature (D), and extraction time (E) were selected based on our preliminary studies to study their interactions and also to find their optimal levels using CCD. The experimental design matrix for the extraction of TPC from S. macrophylla bark is depicted in Table 2. The CCD showed an enhanced TPC yield of 49.53 mg GAE/gdw at 50 % (v/v) acetone concentration, 20 mL volume of acetone, agitation speed of 170 rpm, 32 °C of extraction temperature, and extraction time of 24 h (Run 5, 9, 19, 23, 24, 28 in Table 2). Next, the results were fitted to a second-order regression equation (Eq. (2)).
Where Acetone concentration (A), %v/v; Volume of acetone (B), mL; Agitation speed (C), rpm; Extraction temperature (D), °C; and Extraction time (E), h.
The outcome of an analysis of variance (ANOVA) specifies that the regression model predicts and fits the experimental results very well presenting a high R2 and F- values (Table 4). In this study, the linear (except C), quadratic, and interactive (except AD, BC, and BD) model terms were found to be statistically significant for the extraction of TPC from the bark of S. macrophylla as the P-value was < 0.05 (Table 4). The interaction effects among the factors were visualized with 3D- surface plots as shown in Fig. 2. The interaction between AB, AC, AE, BE, CD, CE, and DE have shown a positive effect on the extraction of TPC representing that the levels selected were converging towards the center point (Level 0). Whereas the interaction between AD, BC, and BD showed a negative effect could be due to the different combinations of factor levels, even though the factors A, B, and D are individually significant. Acetone concentration (A), %v/v; Volume of acetone (B), mL; Agitation speed (C), rpm; Extraction temperature (D), °C; and Extraction time (E), h. *Statistically Significant (95% confidence interval); NS- Statistically Not Significant (95% confidence interval). R2 = 99.71 %; Adj R2 = 99.19; and Predicted R2 = 92.79 %.
Source
DF
Sum of Squares
Contribution %
Mean Square
F-Value
P-Value
Model
20
8478.67
99.71
423.93
191.17
0
*
Linear
5
1393.44
16.39
278.69
125.67
0
*
A
1
724.68
8.52
724.68
326.79
0
*
B
1
314.51
3.7
314.51
141.83
0
*
C
1
0.03
0
0.03
0.01
0.906
NS
D
1
304.45
3.58
304.45
137.29
0
*
E
1
49.77
0.59
49.77
22.44
0.001
*
Square
5
5915.91
69.57
1183.18
533.55
0
*
A2
1
1008.39
11.86
1740.61
784.93
0
*
B2
1
1465.97
17.24
2091.71
943.25
0
*
C2
1
660.29
7.77
992.12
447.39
0
*
D2
1
1169.79
13.76
1400.89
631.73
0
*
E2
1
1611.48
18.95
1611.48
726.73
0
*
2-Way Interaction
10
1169.33
13.75
116.93
52.73
0
*
A × B
1
343.55
4.04
343.55
154.92
0
*
A × C
1
24.01
0.28
24.01
10.83
0.007
*
A × D
1
2.89
0.03
2.89
1.3
0.278
NS
A × E
1
322.02
3.79
322.02
145.22
0
*
B × C
1
0.01
0
0.01
0.01
0.94
NS
B × D
1
8.67
0.1
8.67
3.91
0.074
NS
B × E
1
50.27
0.59
50.27
22.67
0.001
*
C × D
1
228.31
2.69
228.31
102.96
0
*
C × E
1
103.94
1.22
103.94
46.87
0
*
D × E
1
85.66
1.01
85.66
38.63
0
*
Error
11
24.39
0.29
24.39
Lack of Fit
6
24.39
0.29
24.39
Pure Error
5
0
0
0
Total
31
8503.07
100
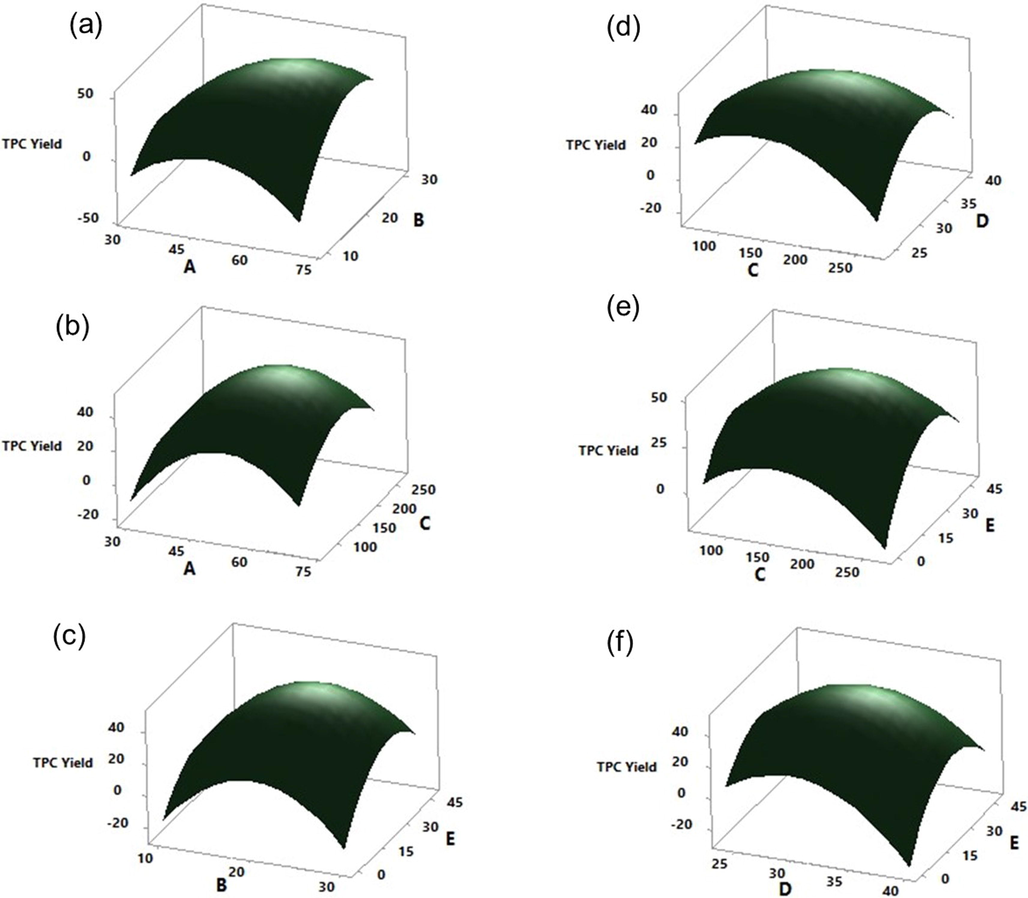
Surface plots showing the interaction effects of the significant factors on the extraction of the TPC process from the bark of S. macrophylla with the remaining factors held constant at the center level of the central composite design: (a) Acetone concentration (A) and Volume of acetone (B); (b) Acetone concentration (A) and Agitation speed (C); (c) Volume of acetone (B) and Extraction time (E); (d) Agitation speed (C) and Extraction temperature (D); (e) Agitation speed (C) and Extraction time (E); and (f) Extraction temperature (D) and Extraction time (E).
The accuracy of the model is tested by using the normal probability plot (Fig. 3). It is the plot between the residuals and the predicted response that also helps to check the normality assumption (Sebayang et al., 2017). If the data points are scattered randomly in the plot shows an inadequate fit and gives meager results. Fig. 3 depicts that the normality assumption is satisfied as all data points are along a straight line indicating that the model provides an adequate estimate of the actual system. Hence, it can be established that the regression model equation (Eq.2) is very much satisfactory to explain the extraction of the TPC process using acetone solvent.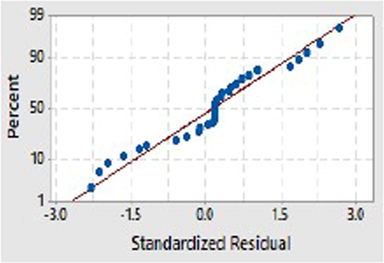
The normal probability plots.
The regression model (Eq. (2)) was solved by using a response optimizer tool of MINITAB 17.0. The acquired optimal levels of individual factors for the improved TPC yield are as follows: Acetone concentration (A) = 50 %(v/v), Volume of acetone (B) = 20 mL, Agitation speed (C) = 170 rpm, Extraction temperature (D) = 32 °C, and Extraction time (E) = 24 h; all of them were found to be present in the experimental levels (Table 1). Under these optimal conditions, the predicted TPC yield was 52.085 mg GAE/gdw (Fig. 4).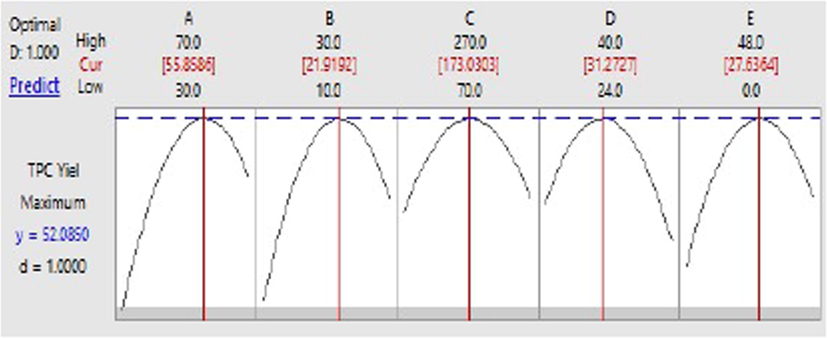
The optimization plot for extraction of TPC from the bark of S. macrophylla.
Lastly, the regression model equation (Eq. (2)) was validated by performing the experiments in triplicates at the above-mentioned optimal levels. It was found that the experimental and predicted TPC yield was 51. 24 ± 0.89 mg GAE/gdw and 52.085 mg GAE/gdw respectively (Table 5), which indicates the adequacy of the model. This optimization method exhibited a 2.0-fold increase in the extraction of TPC from the bark of S. macrophylla using acetone as a solvent when compared to unoptimized conditions (26.537 ± 1.56 mg GAE/gdw). Che Sulaiman et. al (Che Sulaiman et al., 2017) reported an increase of 1.3 times on the extraction of TPC from Clinacanthus nutans Lindau leaves with the use of RSM for optimization, indicating that CCD employed in optimization of TPC extraction from S. macrophylla correlated with better results as there was a 2.0-fold increase in TPC yield. This is the first report on the extraction of bioactive compounds from the bark of S. macrophylla, which represents a high yield of total phenolic, flavonoids, and hydrolyzable tannins with antimicrobial and antioxidant properties.
Parameters
Optimal values
TPC yield, mg GAE/gdw
Predicted
Experimental
A: Acetone concentration, %v/v
56
52.085
51. 24 ± 0.89
B: Volume of acetone, mL
22
C: Agitation speed, rpm
173
D: Extraction temperature, °C
31
E: Extraction time, h
28
7.3 Characterization of extracted total phenolic compounds
7.3.1 Thin-layer chromatography (TLC)
The TLC system was used to separate and identify the contents of flavonoids, phenolic compounds, tannic acid, and gallic acid in acetone extracts of S. macrophylla bark. In this system, Precoated silica gel sheets were used as the stationary phase and the equivalent solvent system was used as the mobile phase as shown in Table 3. Gallic acid, Rutin, and Tannic acid were used as a standard to identify the phenolic compounds and gallic acid, flavonoids, and tannic acid respectively. These compounds were separated in the TLC system and further identified by spraying their respective coloring agents as depicted in Table 3. The colored spots were spotted on the system indicating the presence of phenolic compounds, flavonoids and tannic acids with Rf values of standards Gallic acid, Rutin and Tannic acid are 0.624, 0.958, and 0.978 respectively (Fig. 5a-c). However, the compound gallic acid was not separated and identified (Fig. 5d). This indicates the absence of the component, or presence in the least concentrations in the bark of S. macrophylla, and also showed the gallic acid content of 41.11 ± 1.534 µg/mL using a spectrophotometric method as shown in Fig. 1, whereas other components are in the terms of mg/mL. The values of Rf mentioned for the phenols, flavonoids and tannic acid are in good agreement with reports reported elsewhere (Dinakaran et al., 2019), which represents the presence of bioactive compounds.
The qualitative analysis of bioactive compounds in acetone extracts of S. macrophylla bark: Phenols (a), Flavonoids (b), Hydrolysable tannins (c), and Gallic acid (d) using thin layer chromatography with reference standards gallic acid (G), rutin (R), tannic acid (T), and gallic acid (G) respectively.
7.4 Fourier transform infrared spectroscopy (FTIR) spectroscopy
The FTIR of the studied sample is displayed in Fig. 6. The band at 3309 cm−1 is related to hydroxyl (OH wagging) of a phenolic group (Barud et al., 2013). A band was found at 1638 cm−1 due to the C⚌O stretching vibration of flavonoids. A shoulder at 1700 cm−1 may be due to stretching vibration of C⚌O group (Mot et al., 2011), 1509 cm−1 related to aromatic ring deformations (Franca et al., 2014), 1375 cm-1C-O stretching of acid groups or rocking vibration of aromatic CH, and 1236 cm−1 due to vibration of C—O group of polyols of hydroxy-flavanoids (Silva et al., 2014). The results are in accord with Chukwuneke et al. (Chukwuneke, 2012), indicating that the functional groups identified are similar to the ones presented in this study.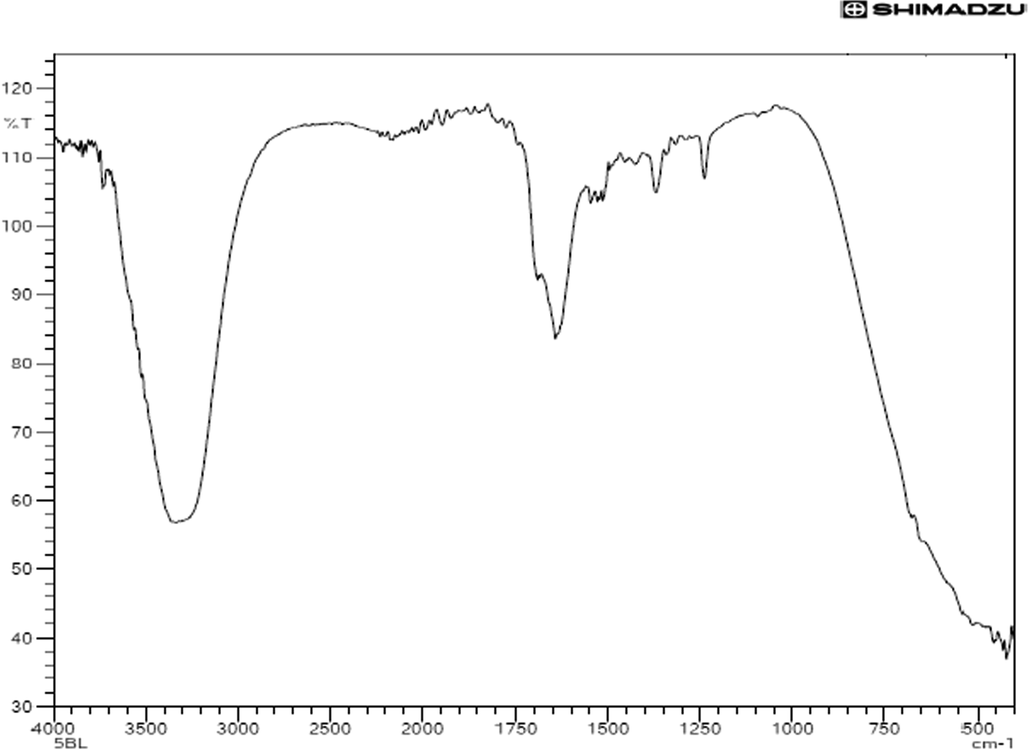
FTIR spectra of bark extract of S. macrophylla.
7.5 Application studies
7.5.1 Antimicrobial activity and minimum inhibitory concentration
The antimicrobial activity of the extract of S. macrophylla bark at 5 % (w/v) was tested against four pathogenic strains i.e. two-gram positive strains Bacillus sp, and S. aureus, and two-gram negative strains E. coli, and S. marcescens. In this study, the extract exhibited the area of inhibition (AOI) against the gram-positive strains tested whereas no AOI was found against gram-negative strains tested. Bacillus sp. and S. aureus showed an AOI of 181.4 mm2 (21 mm clear zone of diameter with 5 mm standard disc) and 113.1 mm2 (18 mm clear zone of diameter with 5 mm standard disc) respectively after 72 h of incubation at 37 °C (Fig. 7). The effectiveness of the extract was estimated by measuring the MICs against the strains with AOI. The extract of S. macrophylla bark showed the MIC value of 20 % (w/v) and 15 % (w/v) against Bacillus sp and S. aureus respectively. Our results showed a good antimicrobial effect against gram-positive bacteria, and also the results are in good agreement with Yuan et al (Yuan et al., 2021). This is the first report on the extraction of bioactive compounds from the bark of S. macrophylla, which represents a high yield of total phenolic, flavonoids, and hydrolyzable tannins with antimicrobial and antioxidant properties.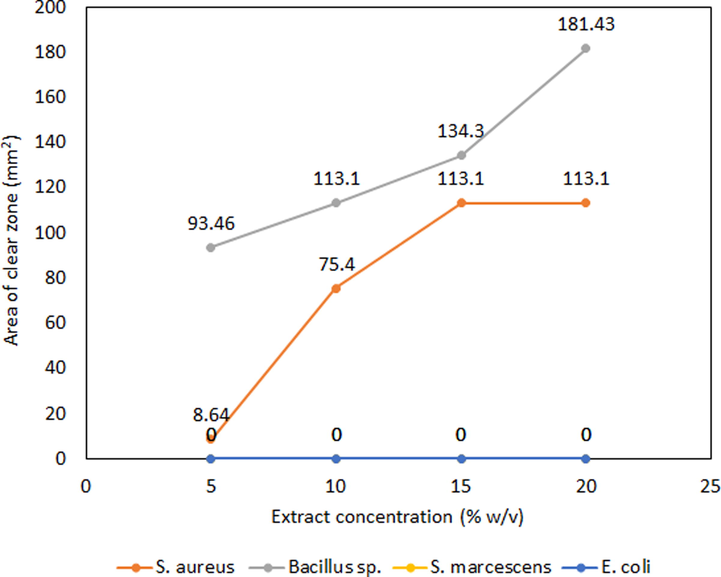
Results of antimicrobial activity and minimum inhibitory concentration.
7.5.2 Electrochemical evaluation of antioxidant properties
The evaluation of the antioxidant properties of plant extract was carried out using CV and DPV. Fig. 8 depicts the redox behavior of compounds present in the plant extract with well-defined oxidation and reduction peaks; in which the CVs were recorded from − 0.2 to 1.4 V at various scan rates. The anodic peak potentials (Epa) as shown in Fig. 8a, 0.17 V and 0.81 V were due to the presence of various antioxidants with varying oxidation potentials (Phenolic compounds and flavonoids). These results confirm that plant extract contains multiple antioxidants. The low and high oxidation potential values obtained from voltammograms indicate that the extract contains both compounds with good and intermediate antioxidant capacity.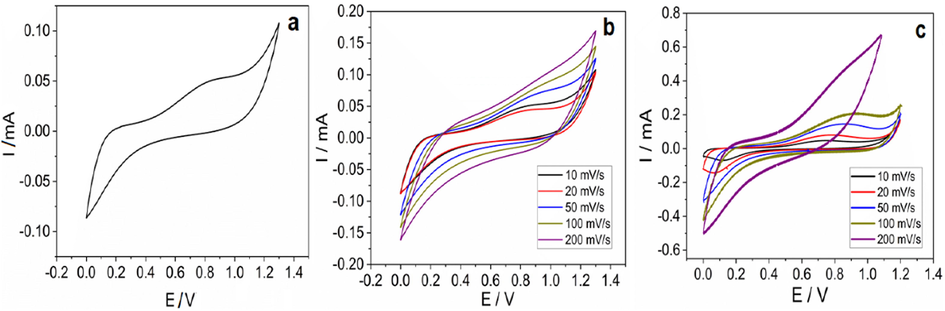
CV of 141 µM plant extract in phosphate buffer. a) at 10 mV/s; b) at various scan rates using glassy carbon as the working electrode; c) at various scan rates using stainless steel as the working electrode.
The CVs were analyzed separately in two different setups by taking glassy carbon and stainless steel(ss) as the working electrodes. The graphs obtained (Fig. 8b and c) confirm the presence of identical peak potentials. As can be seen from Fig. 8b and c that the intensities of the CV increase with an increase in the scan rate. Although a higher scan rate implies greater sensitivity, the charging current also rises. Since it is a diffusion-controlled process at higher scan rates, the graphs look slightly distorted.
Fig. 9 shows the DPV graph for the bark extract of S. macrophylla. This also confirms the presence of a lower oxidation potential peak in agreement with the CV results leading to the antioxidant property. As can be seen from Figure b there is an increase in peak current density with the increase in the concentration of plant extract. This shows the linear relation between the two parameters. The peak current density for this approach is determined by the electron transfer kinetics and the diffusion coefficient of the electroactive species, in addition to the concentration.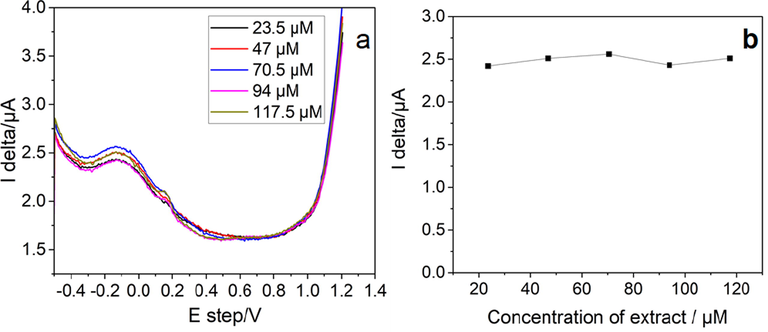
DPV plots for the bark extract of S. macrophylla. a) DPV of extract in phosphate buffer; and b) Variation of the peak current density, in DPV with a concentration of plant extract.
8 Conclusion
The statistical optimization of extraction of total phenolic and flavonoid content from barks of S. macrophylla showed a 2.0-fold increase when compared to unoptimized conditions. The qualitative analysis of extract using the TLC system and FTIR showed the presence of bioactive compounds i.e. phenols, flavonoids, and tannins. The results of antimicrobial activity of S. macrophylla bark extract showed an area of inhibition against only the gram-negative strains tested (E. coli, and S. marcescens) at 5 % (w/v). Further study is crucial to discover the effect of different extraction methods and solvent systems to increase antimicrobial activity. Antioxidant properties were found in all of the species present in the plant extract. It was also proven using electrochemical methods, that plant extracts include electroactive species with oxidation potentials.
The presence of high levels of hydrolyzable tannins in the extract can be used to produce gallic acid via bacterial fermentation-based methods, which can be exploited in industrial applications of the pharmaceutical, cosmetic, and food industries due to its high antioxidant properties. The sample can be further used as an inexpensive source of phenolic compounds and flavonoids due to the high concentration of free phenolic compounds and flavonoids. These compounds can be then extracted, purified, and used in therapeutics due to their antioxidant, antimalarial, antiangiogenic, anticancer, and antimicrobial properties.
9 Authors’ contribution
Abhinav conducted, investigated, and validated this work; Subbalaxmi designed, supervised, and wrote the original draft and was involved in reviewing and editing; Sowmya analyzed the results and also wrote the original draft, and reviewed, and edited this work; Shounak provided resources to carry out this work. All authors have read and approved the final version of the manuscript.
Acknowledgments
The authors of this study are grateful to the Manipal Institute of Technology, MAHE for providing facilities for the conducting of research practices. The contribution and support provided by the faculty and staff of the Department of Biotechnology-MIT, Department of Chemistry-MIT, and the Department of Electronics and Communication Engineering-MIT have been duly acknowledged and appreciated.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Phenolic compounds: current industrial applications, limitations and future challenges. Food Funct.. 2021;12(1):14-29.
- [CrossRef] [Google Scholar]
- E. Ann-Hagerman, G. Larry-Butler G, Protein precipitation method for the quantitative determination of tannins, J, Agri, Food Chem., 26(4) (1978) 809–812. https://doi.org/10.1021/jf60218a027.
- Comparison of the simple cyclic voltammetry (CV) and DPPH assays for the determination of antioxidant capacity of active principles. Molecules. 2012;17(5):5126-5138.
- [CrossRef] [Google Scholar]
- P. Arulpriya, P. Lalitha, S. Hemalatha, (2010) Cyclic voltammetric assessment of the antioxidant activity of petroleum ether extract of Samanea saman (Jacq.)Merr, Adv. Appl. Sci. Res., 1(3) (2010) 24-35.
- Therapeutic and nutraceutical potential of bioactive compounds extracted from fruit residues. Crit. Rev. Food Sci. Nutr.. 2015;55(3):319-337.
- [CrossRef] [Google Scholar]
- TLC and HPTLC fingerprinting of leaf extracts of Wedelia chinensis (Osbeck) Merrill. J. Pharmacogn. Phytochem.. 2014;2(6):29-33.
- [Google Scholar]
- Antimicrobial Brazilian propolis (EPP-AF) containing biocellulose membranes as promising biomaterial for skin wound healing. Evid. Based Complement. Altern. Med. 2013:PP.1-10.
- [CrossRef] [Google Scholar]
- Antioxidant and Structure–activity relationships (SARs) of some phenolic and anilines compounds. Ann. Agric. Sci.. 2013;58(2):173-181.
- [CrossRef] [Google Scholar]
- A. Blainski, G.C. Lopes, J.C.P. De Mello, Application and analysis of the folin ciocalteu method for the determination of the total phenolic content from Limonium brasiliense L., Molecules, 18(6) (2013) 6852-6865. https://doi.org/10.3390%2Fmolecules18066852.
- UV protection from cotton fabrics dyed with different tea extracts. Dyes Pigm.. 2016;134(C):448-452.
- [CrossRef] [Google Scholar]
- Use of tannins to enhance the functional properties of protein based films. Food Hydrocoll.. 2020;100:105443
- [Google Scholar]
- Effects of temperature, time, and solvent ratio on the extraction of phenolic compounds and the anti-radical activity of Clinacanthus nutans Lindau leaves by response surface methodology. Chem. Cent. J.. 2017;11:54.
- [CrossRef] [Google Scholar]
- J. L. Chukwuneke, M. C. Ewulonu, I. C. Chukwujike, P. C. Okolie, Physico-chemical analysis of pyrolyzed bio-oil from swietenia macrophylla (mahogany) wood, Heliyon 5 (6) (20129), pp. e01790. https://doi.org/10.1016/j.heliyon.2019.e01790.
- Bioactive compounds in cranberries and their biological properties. Crit. Rev. Food Sci. Nutr.. 2010;50(7):666-679.
- [CrossRef] [Google Scholar]
- Effect of different extraction techniques on total phenolic and flavonoid contents, and antioxidant activity of betelvine and quantification of its phenolic constituents by validated HPTLC method. 3 Biotech.. 2019;9(1):37.
- [CrossRef] [Google Scholar]
- Effect of extraction methods on yield, phytochemical constituents and antioxidant activity of Withania somnifera. Arab. J. Chem.. 2017;10(1):S1193-S1199.
- [CrossRef] [Google Scholar]
- Profiling and determination of phenolic compounds in poly herbal formulations and their comparative evaluation. J. Tradit. Complement. Med.. 2019;9(4):319-327.
- [CrossRef] [Google Scholar]
- Effects of extraction solvents on total phenolic and flavonoid contents and biological activities of extracts from Sudanese medicinal plants. S. Afr. J. Bot.. 2019;120:261-267.
- [CrossRef] [Google Scholar]
- Propolis - based chitosan varnish: drug delivery, controlled release and antimicrobial activity against oral pathogen bacteria. BMC Complement. Altern. Med.. 2014;14(478):1-11.
- [CrossRef] [Google Scholar]
- Swietenia macrophylla King induces mitochondrial-mediated apoptosis through p53 upregulation in HCT116 colorectal carcinoma cells. J. Ethnopharmacol.. 2014;153(2):375-385.
- [CrossRef] [Google Scholar]
- Antimicrobial properties and mechanism of action of some plant extracts against food pathogens and spoilage microorganisms. Front. Microbiol.. 2018;9:1639.
- [CrossRef] [Google Scholar]
- Optimization of solvent extraction of antioxidants from Eucalyptus globulus leaves by response surface methodology: characterization and assessment of their bioactive properties. Ind. Crop. Prod.. 2017;108:649-659.
- [CrossRef] [Google Scholar]
- Determination of the antioxidant activity of Limoniastrum feei aqueous extract by chemical and electrochemical methods. Cogent Chem.. 2016;2(1):1186141.
- [CrossRef] [Google Scholar]
- Quantification of polyphenolics and their antioxidant capacity in fresh plums. J. Agri. Food Chem.. 2003;51(22):6509-6515.
- [CrossRef] [Google Scholar]
- N. F. Lasano NF, N. S. Ramli, A. H. Hamid, R. Karim, M. S. Dek, R. Shukri, Effects of different extraction solvents on polyphenols and antioxidant capacity of peel, pulp and seed kernel of kuini (Mangifera odorata), Orient Pharm. Exp. Med., 19(3) (2019) 277-86. https://doi.org/10.1007/s13596-019-00383-z.
- Quantification of tannins in tree and shrub foliage: A laboratory manual. Springer Science and Business Media; 2003.
- S. Martillanes, J. Rocha-Pimienta, M. Cabrera-Bañegil, D. Martín-Vertedor, J. Delgado-Adámez, Application of phenolic compounds for food preservation: Food additive and active packaging, in: M. Soto-Hernandez, M. Palma-Tenango, M. d. R. Garcia-Mateos (Eds.), Phenolic Compounds-Biological activity, Intech Open, London UK, 2017, pp. 39-58. .
- Biological activities and phytochemicals of Swietenia macrophylla King. Molecules. 2013;18(9):10465-10483.
- [CrossRef] [Google Scholar]
- Effect of solvent types on phenolics content and antioxidant activities of Acacia polyacantha gum. Int. Food Res. J.. 2017;24:S369-S377.
- [Google Scholar]
- Antimicrobial activity of some plant extracts against bacterial strains causing food poisoning diseases. Saudi J. Biol. Sci.. 2018;25(2):361-366.
- [Google Scholar]
- Rapid and effective evaluation of the antioxidant capacity of propolis extracts using DPPH bleaching kinetic profiles, FT-IR and UV–vis spectroscopic data. J. Food Compost. Anal.. 2011;24(4–5):516-522.
- [CrossRef] [Google Scholar]
- Ultrasound-assisted extraction of phenolic compounds from Laurus nobilis L. and their antioxidant activity. Ultrason. Sonochem.. 2013;20(5):1149-1154.
- [CrossRef] [Google Scholar]
- R. Naima, M. Oumam, H. Hannache, A. Sesbou, B. Charrier, A. Pizzi, F. Charrier–El Bouhtoury, Comparison of the impact of different extraction methods on polyphenols yields and tannins extracted from Moroccan Acacia mollissima barks, Ind. Crops Prod., 70 (2015) 245-252. https://doi.org/10.1016/j.indcrop.2015.03.016.
- Green extraction process of tannins obtained from Moroccan Acacia mollissima barks by microwave: Modeling and optimization of the process using the response surface methodology RSM. Arab. J. Chem.. 2019;12(8):2668-2684.
- [CrossRef] [Google Scholar]
- A.H. Sebayang, M.H. Hassan, H.C. Ong HC, Optimization of reducing sugar production from Manihot glaziovii starch using response surface methodology, Energies, 10(1) (2017) 35. https://doi.org/10.3390/en10010035.
- A Spectrophotometric method for assay of tannase using rhodamine. Anal. Biochem.. 2000;279:85-89.
- [CrossRef] [Google Scholar]
- Membranes from latex with propolis for biomedical applications. Mater. Lett.. 2014;116(1):235-238.
- [CrossRef] [Google Scholar]
- Phenolics as potential antioxidant therapeutic agents: mechanism and actions. Mutat. Res.. 2005;579(1–2):200-213.
- [CrossRef] [Google Scholar]
- Determination of the relative contribution of phenolic antioxidants in orange juice by voltammetric methods. J. Food Comp. Anal.. 2010;17(5):619-633.
- [CrossRef] [Google Scholar]
- Flavonoids and other phenolic compounds from medicinal plants for pharmaceutical and medical aspects: An overview. Medicines. 2018;5(3):93.
- [CrossRef] [Google Scholar]
- Antibacterial activity and mechanism of plant flavonoids to gram-positive bacteria predicted from their lipophilicities. Sci. Rep.. 2021;11:10471.
- [CrossRef] [Google Scholar]
- Techniques for extraction and isolation of natural products: a comprehensive review. Chin. Med.. 2018;13(1):1-26.
- [CrossRef] [Google Scholar]
- The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem.. 1999;64(4):555-559.
- [CrossRef] [Google Scholar]







