Translate this page into:
Extraction of fucoxanthin, antioxidants and lipid from wet diatom Chaetoceros simplex var. calcitrans by liquefied dimethyl ether
⁎Corresponding author. kanda.hideki@material.nagoya-u.ac.jp (Hideki Kanda)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
The co-production of lipids and high value-added products from microalgae is a key milestone for practical, microalgae-derived food and biofuel production. However, the use of ethanol as a solvent for lipid extraction requires a drying pretreatment to avoid wastewater contamination and large energy consumption during solvent recovery. Previous studies on lipid production have reported liquefied dimethyl ether (DME) as an alternative solvent that can extract lipids from high water content microalgae with low energy consumption by eliminating the traditional drying step. In this study, liquefied DME is used as a solvent to extract fucoxanthin, antioxidants, and lipids from wet diatom Chaetoceros simplex var. calcitrans. DME extracted 9.2 mg/g-dry microalgae of fucoxanthin from wet sample; this is slightly less than the 11.9 mg/g-dry microalgae extracted from dry sample by ethanol. The total phenolic content and antioxidant capacity of the liquefied DME extracts were only slightly lower than those of the ethanol extract. Using DME, all water and 22.7 wt% of lipid, and 5.6 wt% of atty acid were extracted. Lipids extracted with liquefied DME have a higher C/N, an indicator of suitability for conversion to biofuel, than lipids obtained by ethanol extraction. The microalga contains high levels of unsaturated fatty acids. Liquefied DME extraction is sustainable method because liquefied DME can extract most of the lipids and functional components without drying the diatom. DME avoids risks associated with ethanol.
Keywords
Antioxidant
Green solvent
Lipid
Microalgae
Xanthophyll
Subcritical fluid
- DME
-
Dimethyl ether
- MVR
-
Mechanical vapor recompression
- TPC
-
total polyphenolic compound content
- DPPH
-
di(phenyl)-(2,4,6-trinitrophenyl)iminoazanium
Abbreviations
1 Introduction
Diatoms are a major group of microalgae that are considered effective tools for combating global warming owing to their high photosynthetic capacity and high atmospheric CO2 absorption capacity (Kanda et al., 2020a). Diatoms are among the most abundant unicellular photosynthetic algae, accounting for approximately 40 % of total marine primary production (Fu et al., 2022). In food and biofuel production from microalgae, the co-production of valuable by-products, is important from a cost perspective. However, in order for microalgae to be considered a sustainable source of both food and fuel, the problems associated with the microalgae processing must be overcome. The CO2 emissions during diatom cultivation and conversion must be lower than the CO2 absorbed by diatoms. Unfortunately, with current methods, the energy input during drying is too high and the energy balance is negative (Lardon et al., 2009). If this energy input is based on fossil fuels that emit CO2 during combustion, the CO2 emissions will increase, thereby offsetting the benefits from its capture. Even if green energy, such as wind or solar, is used to power drying, the negative energy balance remains problematic. This is because it means that electrical energy is used for microalgae conversion with low efficiency. However, mechanical vapor recompression (MVR) technology, a method of reducing drying energy in liquid concentration, is not applicable after the liquid has been concentrated into a muddy form (Han et al., 2021). This is because the dried powder covers the surface of the heat transfer tubes, inhibiting heat transfer. The application of MVR to microalgae is difficult because the microalgae are collected by centrifugation in a muddy state.
To extract lipids from diatoms while avoiding the drying process, an organic solvent that is miscible with water should be used. However, when ethanol is used, the extracted solution is a mixture of water, lipids, and ethanol. Consequently, this solution requires treatment by benzene or other chemicals to recover the ethanol and prevent it from contaminating wastewater through azeotrope formation. In a previous study that used a mixture of supercritical carbon dioxide (SC-CO2) and ethanol to extract lipids and carotenoids from the microalgae Chlorella sorokiniana, prior drying of the sample was required (Morcelli, et al., 2021). Also, in a previous study that used a mixture of SC-CO2 and ethanol to extract water from high water content carrots, the dried carrots in this previous study retained their bright β-carotene color, suggesting limited β-carotene extraction. (Brown, et al., 2008) Moreover, even with SC-CO2 + ethanol, the solution to the problem of ethanol separation from extracted solution is limited, as the extracted solution is a mixture of water, lipid, and ethanol. To solve the water separation problem, an extraction solvent that has a low boiling point must be used so that the solvent can be evaporated at 40 °C through solar heating (Kanda et al., 2015; Li et al., 2014). Further, the solvent must have a large difference in boiling point from that of water to ensure that the residue in the water after evaporation is little, and that it is nontoxic, and safe. Liquefied dimethyl ether (DME) has been proposed as a substance that meets these requirements (Kanda et al., 2020a).
DME is the simplest ether, and it is considerably different from common ethers, such as ethyl ether. The European Union permits DME as an extraction medium (Commission E Commission Directive (EU), 2016), and the Food and Drug Administration, USA classifies DME as “Generally Recognized as Safe (GRAS)” (Food and Drug Administration, 2017). Owing to its molecular structure, it has a normal boiling point as low as − 24.8 °C and is gaseous in the standard state; therefore, DME must be pressurized to 0.59 MPa before use as a liquid solvent at 25 °C (Wu et al., 2011). This low boiling point ensures that no residues remain in extracts (Kanda et al., 2021a). Additionally, bioassays have confirmed that aqueous solutions of DME gas are nontoxic to microorganisms at ambient pressure (Kanda et al., 2021a). Furthermore, DME is polar and forms weak hydrogen bonds (Tatamitani et al., 2002), which makes it partially miscible with water in its liquefied state (Chai et al., 2022; Holldorff et al, 1988; Tallon and Fenton, 2010). Therefore, when liquefied DME is used as an extraction solvent, the DME can diffuse through the water surrounding the high water content substance and make contact with the target. Furthermore, unlike other alkyl ethers, the autoxidation of DME is comparable to that of liquefied petroleum gas (LPG) and it can be safely worked with using routine LPG handling techniques (Naito et al., 2005). In addition to the aforementioned issues, another problem with extraction from microalgae is their high cost. The cost of extracts is driven up by the cost of energy inputs in the extraction process. However, because DME can evaporate with heat from inexpensive solar hot water, the cost of DME recovery is lower than that of ethanol, and thus, the use of DME can reduce the cost of the extracts obtained from microalgae. Unfortunately, this cost reduction alone is insufficient for achieving economic viability. Therefore, the extraction of valuable products using microalgae is important.
Carotenoids are valuable products found in microalgae (Machmudah et al., 2018). Some species of Chaetoceros, a typical diatom, contain high concentrations of the carotenoid fucoxanthin (Trimborn et al., 2019). Fucoxanthin also exerts effects in humans, such as an antidiabetic effect (Jung et al., 2012), improvement in cholesterol metabolism (Beppu et al., 2012; Hu et al., 2012), anti-obesity effect (Maeda et al., 2005), anti-proliferative effect on cancer cells (Hosokawa et al., 2004), and anti-inflammatory effect (Shiratori et al., 2005). Carotenoids are hydrophobic (Honda et al., 2018); this implies that the presence of water is detrimental to extraction and, therefore, drying is necessary. Given that carotenoids are antioxidants, drying microalgae at high temperatures in air for efficient extraction presents a dilemma as this process can degrade the carotenoids. Ethanol is the typical solvent used for fucoxanthin extraction from dried microalgae (Kim et al., 2012). However, one issue with using ethanol in food processing is that the use of ethanol in food or drink is prohibited in Islamism, which is followed by one-fourth of the world’s population (Kettani, 2010). Additionally, recent large-scale health studies have indicated that even small amounts of ethanol can have adverse health effects (GBD, 2018 Alcohol Collaborators, 2018); thus, ethanol should be avoided if possible.
Therefore, the use of liquefied DME, which avoids the air drying of microalgae and using of ethanol during extraction, as a solvent offers an effective solution for carotenoid extraction. Based on this, previous studies have reported that carotenoids, such as lutein (Kanda et al., 2020b) and fucoxanthin (Billakanti et al., 2013; Kanda et al., 2014), can be extracted with liquefied DME without drying of the high water content macroalgae. Moreover, previous studies have also confirmed that catechins (Kanda et al., 2013), resveratrol (Kanda et al., 2021b), which are polyphenols with antioxidant properties, and carotenoids can be extracted from plants with liquefied DME. During extraction, intracellular solutes are dissolved in the solvent and removed by passing them through the pores of the extracellular matrix. Intermolecular interactions between the solute and pore walls are theorized to significantly affect extraction equilibrium (Kanda et al., 2022). Polysaccharides are the main component of the extracellular matrix in previously studied macroalgae (Garcia-Vaquero et al., 2017), whereas in the case of diatoms, the extracellular matrix is a silica cell wall (Kanda et al., 2020a), and their chemical compositions are very different. Therefore, the molecular interactions between the extract and pore walls of the extracellular matrix are significantly different when using diatoms, and this should be investigated. Based on this background, this study investigated the possibility of extracting fucoxanthin and antioxidants from Chaetoceros simplex var. calcitrans using liquefied DME, which is a suitable solvent for lipid extraction.
In other words, using liquefied DME as the extraction solvent makes it possible to avoid the most CO2-emitting process, the pretreatment of drying, to produce lipids and functional substances through photosynthesis, which is the primary purpose of diatom. The differences in lipid extraction from microalgae with conventional solvents and liquefied DME are shown in Fig. 1. Diatoms are covered with inorganic SiO2 cell walls, and it is unclear whether this inhibits the extraction of fucoxanthin and antioxidants. Therefore, it is important to confirm that liquefied DME can extract fucoxanthin and antioxidants from diatoms in order to reduce the cost of lipid-derived biofuels.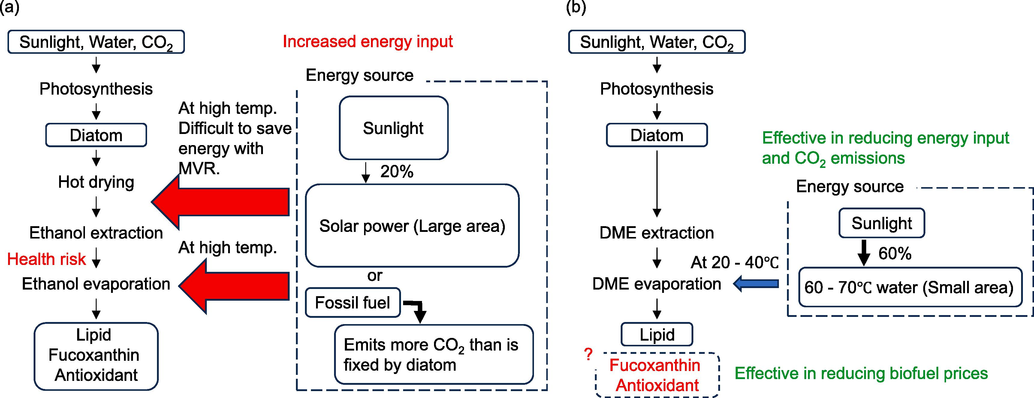
Procedure for lipid extraction from microalgae. (a) conventional solvents, (b) liquefied DME.
2 Materials and methods
2.1 Materials
The C. simplex var. calcitrans used in this study was purchased from Alnur Co., Ltd. (Tokyo, Japan). The original strain was from the Culture Collection of Algae and Protozoa (CCAP) 1085/3 at the UK Scottish Marine Institute. During liquefied DME extraction, the dried samples were mixed with BG-11 broth (C3061, Sigma-Aldrich, MO, U.S.A.) and the moisture content was set to 90.0 %, which was reported to be the typical moisture content at the state after centrifugation by a previous study (Kanda et al., 2020a). Moisture content was determined from the difference between the initial weight and the weight of the dried product after heating to 107 °C to a constant weight; drying at 107 °C is the condition that defines the total moisture content of lignite and peat, which are similar to biomass, in ISO 13909. However, this drying temperature is set low to eliminate significant weight errors due to evaporation of volatile matter, and in reality the moisture content may be underestimated because some of the moisture does not evaporate due to strong hydrogen bonding of the biomass functional groups. In the case of ethanol extraction, the moisture content was similarly adjusted to 90.0 % with BG-11 before drying in air at 50 °C. Liquefied DME was purchased from Tamiya, Inc. (Air Can 420D, Shizuoka, Japan) and used without further purification. Fucoxanthin (95.0+ % (HPLC, mixture of isomers), 90.0+ % (HPLC, trans)), HPLC-grade acetonitrile, HPLC-grade water, gallic acid (98.0–103.0 %), sodium carbonate, and Folin-Ciocalteu reagent were purchased from FujiFilm Wako Pure Chemical Corporation, Osaka, Japan.
2.2 Liquefied DME extraction
Both the DME extraction apparatus used in this study and all operations were performed in accordance with the methods outlined in a previous report (Kanda et al., 2020a). As shown in Fig. 2, the equipment consisted of a metal tank (TVS-1: Taiatsu Techno, Saitama, Japan) containing the liquefied DME, an extraction column (HPG-10–5: Taiatsu Techno, capacity 10 mL, inner diameter 11.6 mm × length 190 mm), and a tank (HPG-96–3: Taiatsu Techno, capacity 96 mL) for collecting the extracted liquid, connected in series, with valves in the middle and at the end of the DME flow.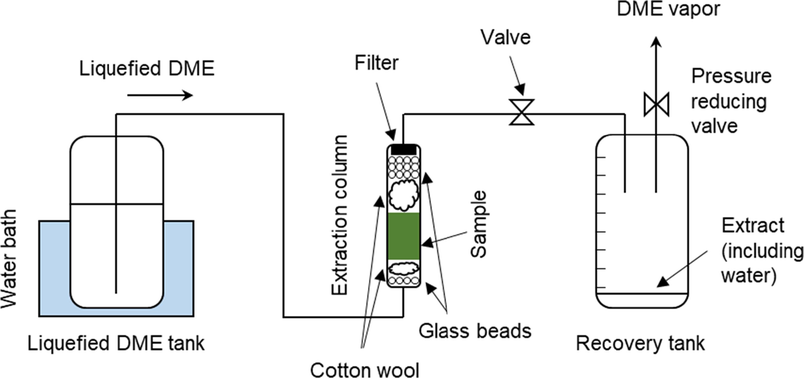
Schematic of liquefied DME extraction.
The extraction column and container in which the extract was collected were made from pressure-resistant glass covered with a clear polycarbonate cylinder for safety and had a volumetric scale printed on them. First, 5.43 ± 0.05 g of wet C. simplex var. calcitrans was packed into the lower half of the extraction column (5 mL volume) and cotton and colorless glass beads were added to the cavity of the extraction column to prevent sample migration. Next, colorless glass beads were used because the completion of fucoxanthin extraction with liquefied DME is determined by a color change in the colorless liquefied DME, which is caused by fucoxanthin extraction. A cellulose filter with a pore size of 0.65 μm (Advantech Toyo Corporation, Tokyo, Japan) was placed at the outlet of the extraction column to prevent the sample from flowing from the column. The tubing connecting each vessel was made of SUS316 and had an inner diameter of 1/16 in..
The inside of the metal tank containing liquefied DME was heated to 35 ± 1 °C in a water bath to increase the saturated vapor pressure of the liquefied DME inside to 0.79 ± 0.02 MPa. As the pressure of the vessel in which the extracted liquid is collected was lower than that of the tank, the liquefied DME was supplied using differential pressure as the driving force. When the supplied liquefied DME passed through a fine tube made of SUS316, a noncontact infrared thermometer confirmed that the temperature decreased rapidly to 25 °C owing to the large specific surface area of the fine tube. The flow rate of liquefied DME was adjusted to 10 ± 1 mL min−1 (=6.61 g min−1) (Wu et al., 2011) by manually adjusting a valve installed in the 1/16 SUS316 tube, based on the volume scale of the container in which the extracted liquid was collected. This was done to determine the rate of increase of liquefied DME. Each time the volume of the extracted solution in the collection vessel increased, it was promptly replaced with a new empty collection vessel. After the filled recovery vessel was disconnected from the apparatus, the end valve was opened and the pressure was reduced to atmospheric pressure to induce DME evaporation. This resulted in an incremental volume of extractant that corresponded with the incremental volume of liquefied DME. After full extraction, the residue was removed from the extraction column and, once the DME had evaporated, the recovery vessel contained only the residual water and the organic extractables, including fucoxanthin. The weight of water extracted was determined by examining the difference in weight before and after air drying at 60 °C overnight. Organic extracts were recovered in an 80:20 (v/v) acetonitrile:water mixture and used for subsequent HPLC analysis. To eliminate the effect of light on fucoxanthin degradation, the surfaces of the extraction and recovery tanks were covered with aluminum foil. To confirm reproducibility, the entire experiment, from extraction to analysis, was repeated thrice, and the three deviations were noted as errors. The reason why the dependence of pressure on the extraction phenomenon is not examined as in SC-CO2 extraction, which is a similar high-pressure extraction technology, is that unlike SC-CO2, DME is a liquid, so the dependence of solubility on pressure is small. The reason why the dependence on temperature is not considered is that, from the viewpoint of reducing operating energy, liquefied DME extraction is assumed to operate at atmospheric temperatures because solar hot water is used as a heat source for boiling liquefied DME and because it is cooled by air, groundwater, or seawater. (Kanda et al., 2015).
2.3 Ethanol extraction
Ethanol, which is commonly used for total fucoxanthin determination, was used as an organic solvent for comparison purposes. Extraction conditions were based on those used in a previous study (Kanda et al., 2014). First, the dry sample was disrupted using a food mill (IFM-800DG, Iwatani Corporation) for 1 min. The amounts of dried C. simplex var. calcitrans. and ethanol used for the extraction were 3.00 g and 300 mL, respectively. Ethanol Soxhlet extraction was performed for 2.5 h; subsequently, the ethanol was evaporated using a rotary evaporator (SB-1200, Eyela Co., Ltd., Tokyo, Japan). To eliminate the effects of light on fucoxanthin degradation, the surfaces of the grinder and extraction equipment were covered with aluminum foil to shield them from light.
2.4 Fucoxanthin-focused characterizations
2.4.1 Quantitative analysis of fucoxanthin
Reversed-phase HPLC was used to quantify the amount of fucoxanthin extracted from the samples. The reversed-phase HPLC system consisted of a degasser (DG–980–50, Jasco Co., Inc., Tokyo, Japan), pump (PU-980, Jasco Co., Inc.), column heater (U–620, Sugai Chemie, Inc., Wakayama, Japan), and ultraviolet–visible (UV–vis) detector (UV-970, Jasco Co., Inc.); it was controlled by Jasco-Borwin (Ver 1.5) software via an LC-Net II/ADC controller (Jasco Co., Inc.). For the isolation of fucoxanthin, an Inertsil ODS-3 column (250 mm × 4.6 mm × 5 μm, GL Science, Tokyo, Japan) was used, with an acetonitrile:water (80/20 (v/v)) eluent flowing at a rate of 1.0 mL/min. The temperature inside the column heater was maintained at 40 °C and the UV–vis detection wavelength was set to 445 nm (Kanda et al., 2014).
2.4.2 Hansen solubility parameter analysis
To examine the differences in the amount of fucoxanthin and antioxidant extracted by the liquefied DME and ethanol extraction methods, solvent compatibility was evaluated using Hansen solubility parameters. Hansen solubility parameter is a measure of the miscibility of two substances. This parameter is based on the idea that intermolecular interactions are split into three components, and the two substances whose values for these components are close to each other are more likely to mix. The three components are the energy due to dispersion forces (δd), energy due to interdipole forces (δp), and energy due to hydrogen bonds, π–π stacking interactions, coordination bonds, and charge transfer interactions (δh) (Hansen, 2004). The three-dimensional distance (Ra) between substances “1″ and ”2″ is calculated by equation (1) below, with a smaller Ra value representing easier mixing.
The idea behind the Hansen solubility sphere is that solvents with component values that fall inside the sphere are better able to dissolve the specified substance, whereas those solvents with component values outside the sphere do not dissolve the said substance (Redelius, 2004; Schoener et al., 2016). Essentially, a substance with a large sphere radius is soluble in numerous solvents, whereas that with a small radius has a more limited range of potential solvents. The exact shape, position, and radius of the Hansen spherical surface differ depending on the combination of solvents and solutes.
However, note that the Hansen solubility parameters do not consider the effects of isomerism, degree of polymerization, or molecular size, which limits their prediction accuracy. The Hansen solubility parameters used in this study were taken from the HSPiP software (ver. 5.3, 2020, https://hansen-solubility.com/), developed by Hansen. The Hansen solubility parameters of liquefied DME are δd = 15.2 MPa0.5, δp = 6.1 MPa0.5, δh = 5.7 MPa0.5. Note that although DME was saturated with water derived from diatoms in the extraction column, the Hansen solubility parameters of water-saturated liquefied DME are not significantly different from those of pure liquefied DME (Kanda et al., 2021b). The Hansen solubility parameters of ethanol are δd = 15.8 MPa0.5, δp = 8.8 MPa0.5, δh = 19.4 MPa0.5 (Kanda et al., 2021b).
2.5 FT-IR characterization
Chemical changes in the original samples, extracted lipids, and residues of C. simplex var. calcitrans produced by liquefied DME extraction were examined using Fourier transform infrared spectroscopy (FT-IR, PerkinElmer Spectrum Two, PerkinElmer Japan Co., Ltd., Yokohama, Japan). The list of IR signals, their corresponding typical functional groups and the possible compounds of diatoms were taken from previous studies (Murdock and Wetzel, 2009; Scarsini et al., 2021). Further, IR data was compared with the IR signal of fucoxanthin (Chen et al., 2022; Noviendri et al., 2016; Yip et al., 2014) to confirm extraction.
2.6 Antioxidant-focused characterizations
2.6.1 Total polyphenolic compounds content assay
As antioxidants can be extracted by liquefied DME, the total polyphenolic compound content (TPC), which is representative of the antioxidant properties, was determined using the Folin–Ciocalteu method (Blainski et al., 2013). First, 1 mL of extracted sample was mixed with 5 mL of deionized water and 6 mL of 7.5 % (w/w) sodium carbonate. After 10 mins, Folin–Ciocalteu reagent (0.5 mL) was added and the mixture was stirred for 5 min. After 2 h of incubation in the dark at room temperature, the TPC was estimated using a UV–vis spectrophotometer to examine the absorbance at 750 nm (V-550, Jasco), which is representative of the equivalent amount of gallic acid (range, 20–100 mg/L) (Chhouk et al., 2017).
2.6.2 DPPH radical scavenging activity assay
The antioxidant capacity of the extract was tested using a di(phenyl)-(2,4,6-trinitrophenyl) iminoazanium (DPPH, other names 2,2-diphenyl-1-(2,4,6-trinitrophenyl)hydrazin-1-yl, 2,2-diphenyl-1-picrylhydrazyl, 1,1-diphenyl-2-picrylhydrazyl radical, and 2,2-diphenyl-1-(2,4,6-trinitrophenyl)hydrazyl diphenylpicrylhydrazyl) radical scavenging activity assay. The UV–vis detection wavelength was 517 nm. Note that the absorption peak of fucoxanthin, which is present in large amounts in the extract, overlaps the absorption wavelength range of 517 nm (Chhouk et al., 2017; Fernández-Agulló et al., 2013), DPPH was first separated from fucoxanthin by HPLC and subsequently quantified according to a method reported in a previous study (Flieger and Flieger, 2020). The HPLC protocol for performing the DPPH radical scavenging activity assay while separating fucoxanthin is as follows. First, the extracts were diluted with methanol to a concentration range of 0.01–2.00 mg/mL. Subsequently, the samples were thoroughly stirred with 2.7 mL of DPPH solution (6 × 10−5 M in methanol) and allowed to stand for 1 h at room temperature in the dark. Next, the solution was injected into an HPLC system with the same instrumentation, column, eluent, temperature, and injection volume as in the fucoxanthin assay mentioned in section 2.4.1.
The DPPH radical scavenging activity was determined using Equation (2):
2.7 Other components characterizations
2.7.1 Organic micro element analysis
As this study aimed not only to extract fucoxanthin and antioxidants but also to co-produce foods and biofuels, the subsequent analyses were conducted from the perspective of foods and biofuels. Elemental analysis was performed using an elemental analyzer (2400 Series II CHNS/O Elemental Analyzer, PerkinElmer Japan Co., Ltd., Yokohama, Japan) to further determine the constituent elements of the organic materials that make up the original sample, lipids, and C. simplex var. calcitrans residue.
2.7.2 FE-SEM characterization
As the contamination of silica and salts in the culture medium with lipids was suspected, the elemental compositions of the original samples, the lipids, and the C. simplex var. calcitrans residues were determined using a field-emission scanning electron microscope (FE-SEM, JSM-7500F, JEOL, Tokyo, Japan). The samples were coated with osmium up to a thickness of 10 nm using a sputtering apparatus (Osmium Plasma Coater OPC-60A, Filgen, Nagoya, Japan); the acceleration voltage was 20.0 kV. The elements measured were Si, which constitutes the cell wall, and Na, which is contained in NaNO3, is the main component of the BG-11 medium (Waterbury and Sranier, 1981). For FE-SEM, as the error of each measurement was large, five measurements were made and the largest and smallest values were rejected, leaving only the data from the middle three measurements.
2.7.3 Fatty acids analysis
The fatty acid components in the extracted lipids were converted to fatty acid methyl esters (FAMEs) by acid-catalyzed esterification using a fatty acid methylation kit (06482–04; Nacalai Tesque, Kyoto, Japan). Impurities were removed using an FAME purification kit (06483–94; Nacalai Tesque) and analysis was conducted using gas chromatography-mass spectrometry (GC–MS; 7890A GC system and 5975C inert XL MSD with a triple-axis detector, Agilent Technologies Japan, Ltd., Hachioji, Japan) according to the NIST mass spectral database. A phenyl arylene capillary column (HP-5MS; 30 m × 0.25 mm i.d., Agilent Technologies Tokyo Ltd., Japan) was employed. The temperature was initially held at 100 °C for 5 min, before being increased to 300 °C at 4 °C min−1. The injector and detector temperatures were set to 250 °C and the split ratio was 1:1 (with a helium gas flow rate of 24 mL min−1 and injection volume of 1.0 μl). Qualitative and quantitative analysis of the detected FAME was performed by comparing the peak area and retention time of the FAME standard (Supelco 37 Component FAME Mix; Sigma-Aldrich, St. Louis, MO, USA) with the mass spectrum. The NIST mass spectral database was used for the qualitative analysis of C16:2 and C16:3, and a calibration curve for C16:1 was substituted for the quantitative analysis. (Kanda et al., 2020a).
3 Results and discussion
3.1 Fucoxanthin-focused characterizations
The extraction operation was terminated when the color of the extract exiting the extraction column changed from light green to colorless and transparent, which was the color of the original liquefied DME. The total extraction time was 50 min, and the total volume of DME flowing was 331 g (500 mL). Fig. 3 shows the appearance of the wet and dry samples of C. simplex var. calcitrans, the residues after extraction with liquefied DME and ethanol, and the lipids gained from ethanol extraction. Before extraction, the sample was a slurry, as shown in Fig. 3(a), whereas the residue after liquefied DME extraction was a very bright yellowish-green dry solid, as shown in Fig. 3(c). The color of the residue was not only much lighter than the pre-extraction sample in Fig. 3(b) but also lighter than the residue from the ethanol extraction shown in Fig. 3(d). This suggests that several pigments, including fucoxanthin, were extracted. As shown in Fig. 3(e) and (f), the lipids extracted with liquefied DME and ethanol were both dark greenish, whereas in the case of ethanol, some solid-like powder was observed in the lipids.
Photographic images of (a) C. simplex var. calcitrans slurry, (b) original dry C. simplex var. calcitrans, (c) residue obtained by DME extraction, (d) residue obtained by ethanol extraction, (e) lipid obtained by DME extraction, and (f) lipid obtained by ethanol extraction.
Changes over time in the amount of water, lipids, and fucoxanthin extracted from C. simplex var. calcitrans by liquefied DME are shown in Fig. 4(a)–(c). Temperature and pressure stabilized early in the experiment. Water, lipids, and fucoxanthin all begin to be extracted from the early stages of extraction, influenced by their solubility in liquefied DME and their diffusion resistance in the sample. In other words, while water is being extracted, hydrophobic lipids and fucoxanthin are also being extracted at the same time. This is thought to be due to the partial miscibility of liquefied DME with water (Holldorff et al., 1988), which allows liquefied DME to diffuse into the aqueous phase surrounding the diatom and contact the lipids and fucoxanthin in the diatom, and DME molecules cover the lipids and fucoxanthin to assist diffusion in the aqueous phase. Note that 0.54 g of dry biomass was extracted with 331 g of solvent, but this does not mean that a large amount of solvent is required for practical application. This apparently large amount of solvent is because the extraction characteristics were studied over a finite column length. In practical use, the amount of solvent is reduced in the pseudo-mobile phase by feeding the excess solvent drained from the column where extraction has been completed into the not-completed diatom packed in the next column. Therefore, the amount of solvent required for extraction can be considered based on the solubility in the early stage of extraction. In the early stages of extraction, the amount of extracted material increased in proportion to the amount of DME; this trend can be explained by the saturated solubility of each substance in liquefied DME. However, at the end of the extraction phase, this slope became shallower. This was attributed to the hindrance of mass transfer due to diffusion resistance during extraction (Dimić et al., 2021) or restraints due to intermolecular interactions between the plant extracellular matrix and the solute, which reduce the saturation concentration of the solute in the solvent (Kanda et al., 2022). This trend was common in water, lipids, and fucoxanthin, which is consistent with previous findings that liquefied DME has favorable extraction properties for both water and fucoxanthin from macroalgae (Kanda et al., 2014). In other words, whether the extracellular matrix is composed of polysaccharides or silica does not seem to have a significant effect on the ease of extraction by liquefied DME.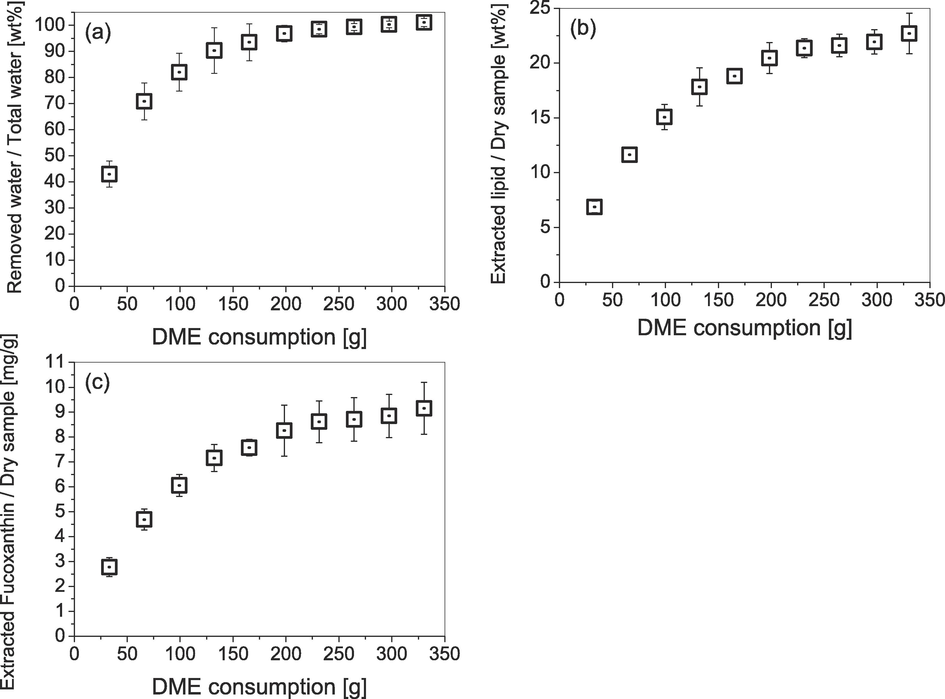
Changes over time in the amounts of materials extracted from C. simplex var. calcitrans by liquefied DME: (a) water; (b) lipid; (c) fucoxanthin.
As shown in Fig. 4(a), by the end of extraction, 101.2 ± 1.6 % of the total water content of C. simplex var. calcitrans was recovered. As the water content extracted by liquefied DME exceeded 100 % of the total water content, chemically bound water that was still present in the dry state could have been removed. As shown in Fig. 4(b), by the end of extraction, the lipids extracted with liquefied DME from the C. simplex var. calcitrans with a high moisture content accounted for 22.7 ± 1.9 % of the dry weight of the C. simplex var. calcitrans. Whereas lipids extracted with ethanol accounted for 23.4 ± 1.4 % of the dry weight. Fig. 4(c) shows the change in the amount of fucoxanthin extracted from C. simplex var. calcitrans using liquefied DME over time. Evidently, at the end of extraction, fucoxanthin extracted with liquefied DME accounted for 9.2 ± 0.9 mg/g of the dry mass of C. simplex var. calcitrans, whereas that extracted with ethanol accounted for 11.9 ± 0.8 mg/g. Essentially, even when using wet samples, liquefied DME had a fucoxanthin extraction capacity that was only slightly less than that of conventional ethanol extraction. Comparing the extracted fucoxanthin content with, for example, the macroalgae Undaria pinnatifida, the amount of fucoxanthin extracted from U. pinnatifida by liquid DME is 0.390 mg/g-dry (Kanda et al., 2014), the amount in C. simplex var. calcitrans is 23.6 times higher than U. pinnatifida. Thus, the fucoxanthin content of C. simplex var. calcitrans was found to be very high.
For fucoxanthin, an analysis using Hansen solubility parameters was performed and the Ra values for DME and ethanol were calculated. The Hansen solubility parameters for fucoxanthin, estimated using HSPiP software, were δd = 17.3 MPa0.5, δp = 4.1 MPa0.5, δh = 4.7 MPa0.5. The calculated Ra values for fucoxanthin versus liquefied DME and ethanol based on these parameters were 4.8 MPa0.5 and 15.7 MPa0.5, respectively. Essentially, the Hansen solubility parameter predicts that liquefied DME should have better fucoxanthin extraction performance than ethanol, a prediction that is inconsistent with the fact that ethanol extracts slightly more fucoxanthin. However, a previous study contradicts the assumed relationship between the extraction amount and the Ra of liquefied DME and ethanol for resveratrol, a polyphenol, and its glycoside, as well as for fucoxanthin. (Kanda et al., 2021b) The fact that the Ra of liquefied DME is significantly smaller than that of ethanol is attributed to the fact that the dh of liquefied DME is significantly smaller than that of ethanol. Hansen's solubility parameters for the DME/water mixture, corrected for DME partially mixing with water, are δd = 15.2 MPa0.5, δp = 6.1 MPa0.5, and δh = 7.1 MPa0.5, the variation in the parameter is not as great. (Kanda et al., 2021b) This may be because the dependence of Hansen solubility parameters of liquefied DME on density has not been fully investigated. For example, in the case of supercritical carbon dioxide, it is known that Hansen solubility parameters change significantly with density due to changes in temperature and pressure. (Hoshino, et al., 2022) In other words, the DME parameters inferred from the molecular structure do not consider the effect of density, which is a candidate source of error.
The slopes of the plots in Fig. 4 show the solubility of water, lipids, and fucoxanthin in, 0.064 ± 0.0056 g/g, 1.13 ± 0.079 mg/g, and 0.046 ± 0.0053 mg/g of liquefied DME, respectively. Evidently, water was preferentially extracted from extracellular free water; hence, these values were close to the saturated solubility in the bulk (0.075 g/g-DME) (Holldorff and Knapp, 1988). By contrast, as lipids and fucoxanthin are intracellular, as noted in the introduction, they are predicted to possess decreased solubility owing to diffusion resistance (Dimić et al., 2021) and binding effects from intermolecular interactions with the cells constituent matrix (Kanda et al., 2022). Therefore, the solubilities of lipids and fucoxanthin may be lower than those in the bulk liquefied DME.
Fig. 5 shows the FT-IR spectra of the native C. simplex var. calcitrans along with the resulting residues and extracts obtained from liquefied DME and ethanol extraction. Table 1 summarizes the peaks that were found, the functional groups suggested by these peaks, the corresponding substances, and the samples in which the peaks were detected.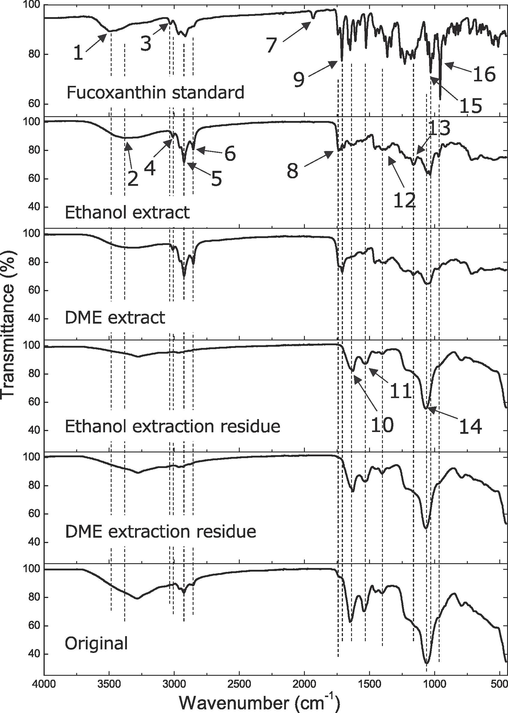
FTIR spectra of native C. simplex var. calcitrans and its residues and extracts after liquefied DME and ethanol extractions.
Peak number
Wave number (cm−1)
Functional groups
Compounds
Samples with peaks detected
1
3481
OH asymmetric expansion/contraction
Fucoxanthin
2
3400
CH = CH stretching
Lipid
Ethanol extract
DME extract
3
3029
C–H stretching
Fucoxanthin
4
3010
=C–H stretching
Lipid
Ethanol extract
DME extract
5
2924
CH = CH asymmetric stretching
Lipid
FucoxanthinEthanol extract
DME extract
Native sample
6
2853
CH3, CH2 symmetry stretching
Lipid
FucoxanthinEthanol extract
DME extract
Native sample
7
1930
Allen C = C = C stretching
Fucoxanthin
8
1735
C = O stretching
Lipid
Ethanol extract
DME extract
Native sample
9
1712
Ketone C = O stretching
Fucoxanthin
DME extract
10
1627
C = O stretching
Protein
FucoxanthinEthanol residue
DME residue
Native sample
11
1530
Amide II C-N stretching
N–H angular vibrationProtein
Ethanol residue
DME residue
Native sample
12
1440
CH2 symmetry in-plane variation, CH3 inverse symmetry variable angle
Lipid
All samples
13
1183
Ester C-O stretching
Fucoxanthin
Ethanol extract
DME extract
14
1065
Si-O-Si asymmetric expansion/contraction
Silica
(Smaller peak)
Ethanol extract
DME extract
(Larger peak)
Ethanol residue
DME residue
Native sample
15
1030
Ester C-O stretching
Fucoxanthin
Ethanol extract
DME extract
16
957
C–H out-of-plane bending vibrations
Fucoxanthin
All samples
First, the characteristic fucoxanthin peaks 13 and 15 were detected in both the liquefied DME and ethanol extracts. Additionally, the fucoxanthin peak 16 was observed as a small peak in both extracts and appeared to have remnants as an inflection point; however, it was buried by other, larger peaks in the residue and native sample. Peaks 5 and 6, which are common to both fucoxanthin (Chen et al., 2022; Noviendri et al., 2016; Yip et al., 2014) and lipids, were detected in both extracts and native samples, and were particularly strongly detected in the extracts. Among the characteristic lipid peaks of 2, 4, 8, and 12, (Murdock and Wetzel, 2009; Scarsini et al., 2021), peaks 2 and 4 were detected only in extracts, peak 8 was detected in both extracts and native samples, and peak 12 was present in all samples, including in the residue. Peak 7 is allenic C = C = C stretching, which was detected only in the standard fucoxanthin and was not detected in all algal samples in this study. In a previous study (Chen et al., 2022), it was reported that the peak 7 disappears completely in fucoxanthin complexed with mixed lipid or cholesterol or gelatin. The algal samples used in this study were also mixed with other substances, supporting the findings of the previous study. Generally, fucoxanthin and lipids were detected prominently in the extracts but less prominently in the residues.
Protein-derived peaks 10 and 11 were also detected in both residues and native samples, thus indicating that the protein was not extracted by liquefied DME or ethanol (Murdock and Wetzel, 2009; Scarsini et al., 2021). Peak 14, which resulted from the silica cell walls characteristic of diatoms (Murdock and Wetzel, 2009; Scarsini et al., 2021), was weakly detected in the extracts, and strongly detected in the residues and native samples. The detection of small peaks in the extract samples is indicative of extract contamination with silica cell walls.
3.2 Antioxidant-focused characterizations
The TPCs with antioxidant activity were 16.3 ± 0.7 mg GAE/g-dry algae sample for the liquefied DME extract and 25.7 ± 1.4 mg GAE/g-dry algae sample for the ethanol extract. As shown in Fig. 6, the IC50 was then measured from the absorption intensity at 517 nm. Peaks derived from DPPH was detected at 8 min in retention time and peaks derived from fucoxanthin at 20 min, confirming that other antioxidants and fucoxanthin were separated. As the added lipids increase, DPPH is consumed by the antioxidants, decreasing the DPPH peak and conversely increasing the amount of fucoxanthin detected. The IC50 was determined from the decreasing of the DPPH peaks at various added lipid concentrations. In the DPPH radical scavenging activity assay, the IC50 was 55.7 ± 3.3 µg / mL-lipid for the DME extract and 48.9 ± 2.3 µg / mL-lipid for the ethanol extract. Essentially, the extraction capacity of liquefied DME was slightly lower than that of ethanol for phenols and antioxidant substances.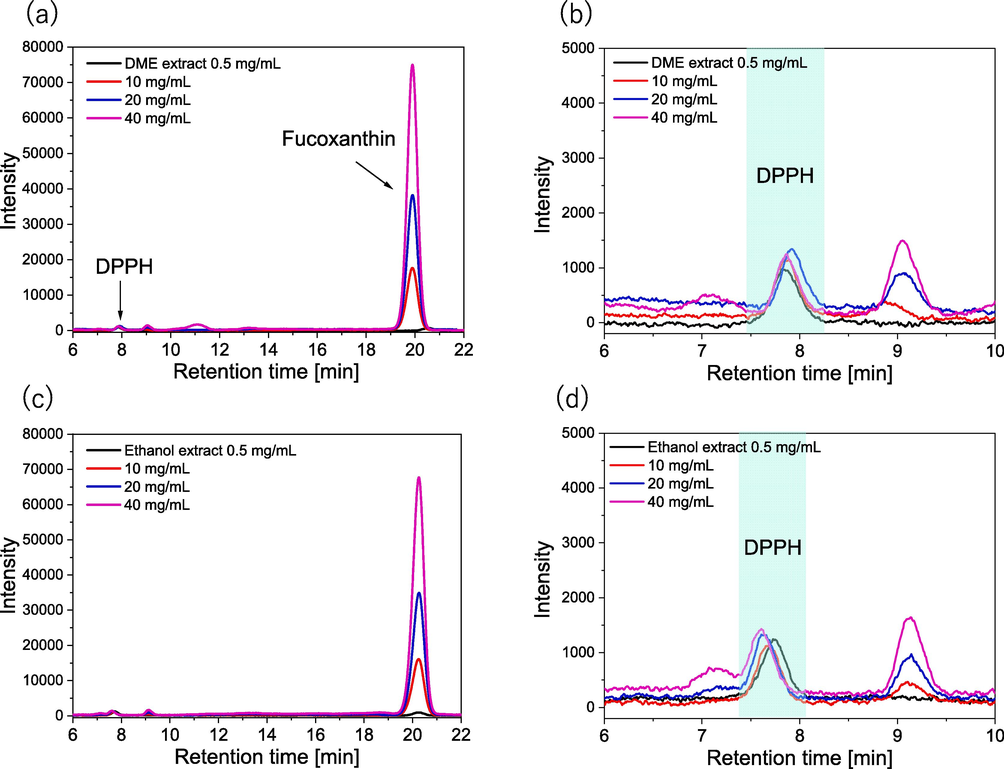
HPLC chromatograms in the DPPH radical scavenging activity assay: (a-b) DME extracts, (c-d) ethanol extracts.
In a previous study (Kanda et al., 2023), curcumin and antioxidants were extracted from Curcuma longa L. (turmeric) with liquefied DME, the TPC content was 68.45 ± 3.65 mg GAE/g-dry and the IC50 values were 67.21 ± 4.79 mg/mL. In other words, the antioxidant activity of this diatom is approximately 1/4 of that of turmeric, but considering that turmeric contained very potent antioxidants, this diatom is considered to contain a non-negligible amount of antioxidants as a lipid by-product that can be used for biofuels and other purposes.
3.3 Other components characterizations
Evidently from Table 2, the elemental ratios determined by CHNS-corder confirmed that the lipids were enriched with C and H, whereas the residues were enriched with N and O. This finding is consistent with that of a previous study that examined different diatoms (Kanda et al., 2020a). Comparing the results from the two extraction solvents, the proportions of C and H were clearly higher for liquefied DME extraction. The C/N ratio is considered an important indicator in the choice of biomass conversion technology when lipids are used as biofuels. If the C/N ratio is greater than 30, thermochemical conversion processes such as methyl esterification, combustion, gasification, and pyrolysis are suitable, whereas if the C/N ratio is less than 30, biochemical processes such as fermentation are more appropriate (Dahlquist, 2013). The C/N ratio of the native sample was 5.2, thus indicating that it is suitable for biochemical processing. Whereas the C/N ratio of the lipid obtained by liquefied DME extraction was 43.8, thus indicating that a thermochemical conversion process should be used. However, the C/N ratio of lipids obtained by ethanol extraction was 27.9; thus, the suitability of the thermochemical conversion process was unclear. * Dry ash-free (daf) basis. The error was the standard deviation. ** Determined by difference.
Wt %*
C
H
N
O**
S
C/N (–)
Native
36.57 ± 0.26
5.57 ± 0.05
7.02 ± 0.03
50.06 ± 0.40
0.78 ± 0.07
5.2
Liquefied DME extraction
Residue
27.07 ± 0.09
4.10 ± 0.16
7.23 ± 0.03
60.88 ± 0.17
0.71 ± 0.03
3.7
Lipid
70.14 ± 0.32
9.47 ± 0.31
1.60 ± 0.00
18.64 ± 0.09
0.15 ± 0.04
43.8
Ethanol extraction
Residue
27.54 ± 0.10
4.35 ± 0.21
7.80 ± 0.04
59.59 ± 0.31
0.72 ± 0.01
3.5
Lipid
54.06 ± 0.22
7.89 ± 0.12
1.94 ± 0.02
35.76 ± 0.29
0.36 ± 0.01
27.9
Fig. 7 shows FE-SEM images of the native sample, along with the lipids, and residues obtained by both liquefied DME extraction and ethanol extraction. The ratios of the constituent elements are listed in Table 3. In lipids extracted with liquefied DME and ethanol, the Si ratio decreased. Conversely, in the residue, the Si ratio increased. This implies that neither of the extraction operations extracted silica or its precursor components that make up the cell wall. In terms of the Na ratio of the original sample, 2.71 % of Na derived from the BG-11 culture was detected. The lipid extracted by liquefied DME contained only 0.26 wt% Na, whereas the lipid extracted from ethanol contained 5.20 wt% Na, a higher Na content than that in the original sample. Essentially, Na remained in the residue during liquefied DME extraction but was transferred to the extracted lipids during ethanol extraction. The transfer of Na to lipids was further examined by focusing on the solubility of NaNO3, the main component of BG-11, in ethanol. First, the concentration of NaNO3 in 99.9 mL BG-11 was 150 mg (Waterbury and Sranier, 1981). Because the sample was 27 mL of BG-11 mixed with diatoms, dried, and mixed with 300 mL (236.7 g) of ethanol, the NaNO3 concentration in ethanol was 0.063 wt%. Here, the saturated solubility of NaNO3 in ethanol was approximately 0.04 wt% (Zhang et al., 2017). However, this saturated solubility corresponds to approximately two-thirds of the NaNO3 present in BG-11 being transported into ethanol, which does not explain the almost complete removal of Na from the residue with ethanol, as shown by the FE-SEM images. Therefore, this suggests that the ethanol solvated lipids promote the dissolution of Na in ethanol. The error reported is the standard deviation.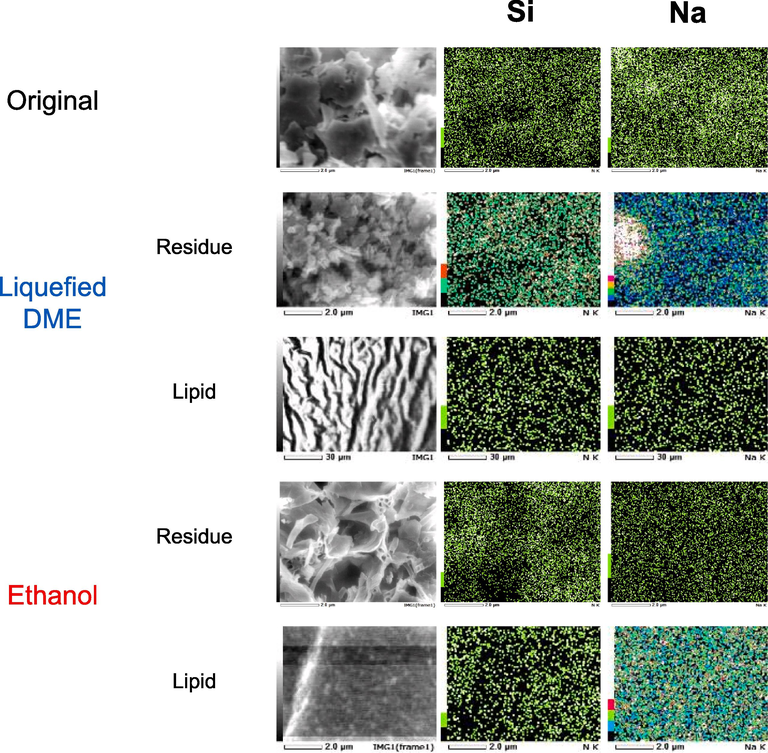
FE-SEM images of original C. simplex var. calcitrans and its residues and extracts after liquefied DME and ethanol extractions.
Wt %
Si
Na
Original
7.36 ± 0.72
2.71 ± 0.69
Liquefied DME extraction
Residue
4.58 ± 0.32
4.51 ± 0.54
Lipid
0.12 ± 0.03
0.26 ± 0.06
Ethanol extraction
Residue
4.09 ± 0.09
0.46 ± 0.04
Lipid
0.05 ± 0.01
5.20 ± 0.60
In contrast to ethanol residue, some areas in the liquefied DME residue exhibited a high concentration of Na. Therefore, these results indicate that the liquefied DME also functioned as an antisolvent for NaNO3 in this study, an idea that can be backed up by previous findings that reported liquefied DME function as an antisolvent for the crystallization of glycine from an aqueous solution (Kanda et al., 2020c). Salt contamination of lipids is undesirable for the use of lipids as food and biofuel. In other words, liquefied DME is superior to ethanol in preventing salt contamination of lipids.
The total fatty acid content was 5.6 ± 0.2 wt% of the dry weight of C. simplex var. calcitrant for the liquefied DME extract, and 4.3 ± 0.5 wt% for the ethanol extract, thus indicating that liquefied DME had a higher extraction rate for fatty acids than ethanol. Table 4 presents the fatty acid composition of the lipids extracted using liquefied DME and ethanol from C. simplex var. calcitrans. Of the fatty acid methyl esters detected, those with less than 1 wt% of total fatty acids were excluded from the list. Error is standard deviation.
wt %
C14:0
C16:3
C16:1
C16:2
C16:0
C20:4
C20:5
Liquefied DME extraction
4.28 ± 0.15
23.35 ± 1.44
23.34 ± 2.66
16.84 ± 1.50
3.31 ± 0.09
1.08 ± 0.08
23.28 ± 3.36
Ethanol extraction
7.47 ± 0.49
19.65 ± 0.36
27.84 ± 3.11
12.45 ± 0.28
4.49 ± 0.39
1.15 ± 0.02
22.56 ± 1.49
The fatty acid composition of the lipids extracted with liquefied DME was almost identical to that of lipids extracted with ethanol, consisting primarily of C16 and C20 unsaturated fatty acids. Evidently from Table 4, the major fatty acid components of the extracted lipids were palmitoleic acid (C16:1), hexadecatrienoic acid (C16:3), eicosapentaenoic acid (C20:5), and hexadecadienoic acid (C16:2), with a content of 23.34–27.84 %, 23.35–19.65 %, 22.56–23.28 %, and 12.45–16.84 %, respectively. This fatty acid type is nearly equivalent to the types of fatty acids that are predominantly found in lipids extracted from another diatom, C. gracilis, namely C16:1, C16:3, and C20:5 (Kanda et al., 2020a). This high unsaturated fatty acid ratio indicates that C. simplex var. calcitrans lipid is more suitable as a functional food than as a biofuel.
In general, lipids with a high composition of unsaturated fatty acids are liquid at room temperature, while those with a high composition of saturated fatty acids have a higher melting point and are solid at room temperature. On the other hand, lipids with high unsaturated fatty acid composition are susceptible to degradation due to oxidation, while lipids with high saturated fatty acid composition have good oxidation stability. Since these properties are inherited even after conversion to biofuel, lipids with high unsaturated fatty acid compositions are more suitable as raw materials for use in cold climates during winter. However, too high a degree of unsaturation impairs oxidation stability, so care must be taken to avoid degradation during fuel production and long-term storage. Therefore, an appropriate balance in fatty acid composition of biofuel is necessary.
3.4 Study strengths and limitations
The advantage of extracting lipids and functional substances from diatoms is to take their extremely high lipid synthesis capacity, which allows them to efficiently fix atmospheric CO2. However, diatoms are very water content when harvested from their culture medium, so drying is conventionally required as a pretreatment for ethanol extraction. The advantage of liquefied DME extraction is that drying of diatoms is not required prior to extraction, thus avoiding the conventional problem of emitting more CO2 than is fixed by the diatoms upon drying. Moreover, the fucoxanthin content of this diatom is an order of magnitude higher than that of macroalgae U. pinnatifida, most of which can be extracted with liquefied DME. In other words, fractionation of high-value-added fucoxanthin can reduce the price of lipid-derived biofuels. In other words, the results support the practical application of diatom-derived biofuels in terms of both energy balance and price. In addition, liquefied DME was found to be able to extract many of the antioxidants as well, which could be expected to further drive down prices.
The disadvantage is that slightly less fucoxanthin and antioxidants are extracted than with ethanol. However, this problem is limited when the priority is to take advantage of the high CO2 reduction capacity of diatoms. Another problem is the susceptibility to oxidation due to the high content of unsaturated fatty acids in the lipids from C. simplex var. calcitrans. However, flowability is improved, so this should also be considered.
4 Conclusion
To effectively utilize the CO2 absorption capacity of diatoms, we propose DME as an alternative solvent for the coproduction of lipids and other high value-added materials without the energy-intensive drying pretreatment. This study shows that fucoxanthin, antioxidants, and lipids can be extracted from wet diatoms using liquefied DME without the drying pretreatment. Ethanol extraction, which has traditionally been used to extract fucoxanthin, can extract slightly more fucoxanthin and antioxidants than DME, but consumes more energy in the drying pretreatment than is obtained by the diatoms in sunlight. Ethanol also has health risks, and the volatile, non-residual DME is an option for those who do not wish to use ethanol.
Acknowledgments
This study was performed by the Environment Research and Technology Development Fund JPMEERF20223C05 of the Environmental Restoration and Conservation Agency of Japan.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this study.
References
- Effects of dietary fucoxanthin on cholesterol metabolism in diabetic/obese KK-A(y) mice. Lipids Health Dis.. 2012;11:112.
- [CrossRef] [Google Scholar]
- Enzyme-assisted extraction of fucoxanthin and lipids containing polyunsaturated fatty acids from Undaria pinnatifida using dimethyl ether and ethanol. Process. Biochem.. 2013;48:1999-2008.
- [CrossRef] [Google Scholar]
- Application and analysis of the folin ciocalteu method for the determination of the total phenolic content from Limonium brasiliense L. Molecules. 2013;18:6852-6865.
- [CrossRef] [Google Scholar]
- Drying of foods using supercritical carbon dioxide — Investigations with carrot. Innov. Food Sci. Emerg. Technol.. 2008;9:280-289.
- [CrossRef] [Google Scholar]
- Vapor-liquid equilibrium (VLE) prediction for dimethyl ether (DME) and water system in DME injection process with Peng-Robinson equation of state and composition dependent binary interaction coefficient. J. Petrol. Sci. Eng.. 2022;211:110172
- [CrossRef] [Google Scholar]
- Fucoxanthin loaded in palm stearin- and cholesterol-based solid lipid nanoparticle-microcapsules, with improved stability and bioavailability in vivo. Mar. Drugs.. 2022;20:237.
- [CrossRef] [Google Scholar]
- Extraction of phenolic compounds and antioxidant activity from garlic husk using carbon dioxide expanded ethanol. Chem. Eng. Process.. 2017;117:113-119.
- [CrossRef] [Google Scholar]
- Commission E Commission Directive (EU), 2016. 2016/1855 of 19 October 2016 amending Directive 2009/32/EC of the European Parliament and of the Council on the approximation of the laws of the Member States on extraction solvents used in the production of foodstuffs and food ingredients. Off. J. Eur. Union. 284, 19–20. http://data.europa.eu/eli/dir/2016/1855/oj.
- Dahlquist, E., 2013. Technologies for Converting Biomass to Useful Energy: Combustion, Gasification, Pyrolysis, Torrefaction and Fermentation. CRC Press Inc, London.
- Supercritical fluid extraction kinetics of cherry seed oil: Kinetics modeling and ANN optimization. Foods. 2021;10:1513.
- [CrossRef] [Google Scholar]
- Influence of solvent on the antioxidant and antimicrobial properties of walnut (Juglans regia L.) green husk extracts. Ind. Crops Prod.. 2013;42:126-132.
- [CrossRef] [Google Scholar]
- The [DPPH●/DPPH-H]-HPLC-DAD method on tracking the antioxidant activity of pure antioxidants and goutweed (Aegopodium podagraria L.) hydroalcoholic extracts. Molecules. 2020;25:6005.
- [CrossRef] [Google Scholar]
- Food and Drug Administration, 2017. GRAS Notice for the Use of Dimethyl Ether as an Extraction Solvent. Notice No, G.R.A.S. GRN 000741.
- Diatom morphology and adaptation: Current progress and potentials for sustainable development. Sustain. Horiz.. 2022;2:100015
- [CrossRef] [Google Scholar]
- Polysaccharides from macroalgae: Recent advances, innovative technologies and challenges in extraction and purification. Food Res. Int.. 2017;99:1011-1020.
- [CrossRef] [Google Scholar]
- GBD, 2018. Alcohol use and burden for 195 countries and territories, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet. 392, 1015–1035. .
- Experimental investigation of a batched mechanical vapor recompression evaporation system. Appl. Therm. Eng.. 2021;192:116940
- [CrossRef] [Google Scholar]
- 50 years with solubility parameters-past and future. Prog. Org. Coat.. 2004;51:77-84.
- [CrossRef] [Google Scholar]
- Binary vapor-liquid-liquid equilibrium of dimethyl ether-water and mutual solubilities of methyl chloride and water: Experimental results and data reduction. Fluid Phase Equilib.. 1988;44:195-209.
- [CrossRef] [Google Scholar]
- Honda, M., Sato, H., Takehara, M., Inoue, Y., Kitamura, C., Takemura, R., Fukaya, T., Wahyudiono, Kanda, H., Goto, M., 2018. Microwave-accelerated Z-isomerization of (all-E)-lycopene in tomato oleoresin and enhancement of the conversion by vegetable oils containing disulfide compounds. Eur. J. Lipid Sci. Technol. 120, 1800060. http://dx.doi.org/10.1002/ejlt.201800060.
- Hoshino, Y., Wahyudiono, Machmudah, S., Hirayama, S., Kanda, H., Hoshino, M., Goto, M., 2022. Extraction of functional components from freeze-dried Angelica furcijuga leaves using supercritical carbon dioxide. ACS Omega, 7, 5104–5111.https://doi.org/10.1021/acsomega.1c06105.
- Fucoxanthin induces apoptosis and enhances the antiproliferative effect of the PPARγ ligand, troglitazone, on colon cancer cells. Biochim. Biophys. Acta.. 2004;1675:113-119.
- [CrossRef] [Google Scholar]
- Combination of fucoxanthin and conjugated linoleic acid attenuates body weight gain and improves lipid metabolism in high-fat diet-induced obese rats. Arch. Biochem. Biophys.. 2012;519:59-65.
- [CrossRef] [Google Scholar]
- Promising antidiabetic potential of fucoxanthin isolated from the edible brown algae Eisenia bicyclis and Undaria pinnatifida. Fish. Sci.. 2012;78:1321-1329.
- [CrossRef] [Google Scholar]
- Kanda, H., Kamo, Y., Machmudah, S., Wahyudiono, Goto, M., 2014. Extraction of fucoxanthin from raw macroalgae excluding drying and cell wall disruption by liquefied dimethyl ether. Mar. Drugs. 12, 2383–2396. https://doi.org/10.3390/md12052383.
- Kanda, H., Hoshino, R., Murakami, K., Wahyudiono, Zheng, Q., Goto, M., 2020a. Lipid extraction from microalgae covered with biomineralized cell walls by using liquefied dimethyl ether. Fuel. 262, 116590. https://doi.org/10.1016/j.fuel.2019.116590.
- Kanda, H., Wahyudiono, Machmudah, S., Goto, M., 2020b. Direct extraction of lutein from wet macroalgae by liquefied dimethyl ether without any pretreatment. ACS Omega. 5, 24005–24010. https://doi.org/10.1021/acsomega.0c03358.
- Kanda, H., Katsube, T., Hoshino, R., Kishino, M., Wahyudiono, Goto, M., 2020c. Ethanol-free antisolvent crystallization of glycine by liquefied dimethyl ether. Heliyon. 6, e05258. https://doi.org/10.1016/j.heliyon.2020.e05258.
- Kanda, H., Ando, D., Hoshino, R., Yamamoto, T., Wahyudiono, Suzuki, S., Shinohara, S., Goto, M., 2021a. Surfactant-free decellularization of porcine aortic tissue by subcritical dimethyl ether. ACS Omega. 6, 13417–13425. https://doi.org/10.1021/acsomega.1c01549.
- Kanda, H., Oishi, K., Machmudah, S., Wahyudiono, Goto, M., 2021b. Ethanol-free extraction of resveratrol and its glycoside from Japanese knotweed rhizome by liquefied dimethyl ether without pretreatments. Asia-Pac. J. Chem. Eng. 16, e2600. https://doi.org/10.1002/apj.2600.535.
- Kanda, H., Wahyudiono, Goto, M., 2022. Thermodynamic model of extraction equilibrium in cylindrical nanopores validated with molecular dynamics simulation. Chem. Eng. Sci. 248, 117115. https://doi.org/10.1016/j.ces.2021.117115.
- Production of decaffeinated green tea leaves using liquefied dimethyl ether. Food Bioprod. Process.. 2013;91:376-380.
- [CrossRef] [Google Scholar]
- Energy-saving lipid extraction from wet Euglena gracilis by the low-boiling-point solvent dimethyl ether. Energies. 2015;8:610-620.
- [CrossRef] [Google Scholar]
- Ethanol-free extraction of curcumin and antioxidant activity of components from wet Curcuma longa L. by liquefied dimethyl ether. Arabian J. Chem.. 2023;16:104585
- [CrossRef] [Google Scholar]
- Kettani, H., 2010. 2010 World Muslim Population. Proceedings of the 8 th Hawaii International Conference on Arts and Humanities, Honolulu, Hawaii, January 2010. https://www.ohio.edu/orgs/muslimst/downloads/World_Muslim_Population_2010.pdf.
- A potential commercial source of fucoxanthin extracted from the microalga Phaeodactylum tricornutum. Appl. Biochem. Biotechnol.. 2012;166:1843-1855.
- [CrossRef] [Google Scholar]
- Life-cycle assessment of biodiesel production from microalgae. Environ. Sci. Technol.. 2009;43:6475-6481.
- [CrossRef] [Google Scholar]
- Simultaneous production of bio-solid fuel and bio-crude from vegetal biomass using liquefied dimethyl ether. Fuel. 2014;116:370-376.
- [CrossRef] [Google Scholar]
- Supercritical fluids extraction of valuable compounds from algae: Future perspectives and challenges. Eng. J.. 2018;22:13-30.
- [CrossRef] [Google Scholar]
- Fucoxanthin from edible seaweed, Undaria pinnatifida, shows antiobesity effect through UCP1 expression in white adipose tissues. Biochem. Biophys. Res. Commun.. 2005;332:392-397.
- [CrossRef] [Google Scholar]
- Supercritical fluid (CO2+ethanol) extraction of chlorophylls and carotenoids from Chlorella sorokiniana: COSMO-SAC assisted prediction of properties and experimental approach. J CO2 Util. 2021;51:101649
- [CrossRef] [Google Scholar]
- FT-IR microspectroscopy enhances biological and ecological analysis of algae. Appl. Spectrosc. Rev.. 2009;44:335-361.
- [CrossRef] [Google Scholar]
- A comparative study on the autoxidation of dimethyl ether (DME) comparison with diethyl ether (DEE) and diisopropyl ether (DIPE) J. Loss Prevent Process. Ind.. 2005;18:469-473.
- [CrossRef] [Google Scholar]
- Fabrication of fucoxanthin-loaded microsphere (F-LM) by two steps double-emulsion solvent evaporation method and characterization of fucoxanthin before and after microencapsulation. J. Oleo Sci.. 2016;65:641-653.
- [CrossRef] [Google Scholar]
- Bitumen solubility model using Hansen solubility parameter. Energy Fuels. 2004;18:1087-1092.
- [CrossRef] [Google Scholar]
- Metabolite quantification by fourier transform infrared spectroscopy in diatoms: Proof of concept on Phaeodactylum tricornutum. Front. Plant Sci.. 2021;12:756421
- [CrossRef] [Google Scholar]
- High throughput research and evaporation rate modeling for solvent screening for ethylcellulose barrier membranes in pharmaceutical applications. Drug Dev. Ind. Pharm.. 2016;42:1700-1707.
- [CrossRef] [Google Scholar]
- Effects of fucoxanthin on lipopolysaccharide-induced inflammation in vitro and in vivo. Exp. Eye Res.. 2005;81:422-428.
- [CrossRef] [Google Scholar]
- The solubility of water in mixtures of dimethyl ether and carbon dioxide. Fluid Phase Equilib.. 2010;298:60-66.
- [CrossRef] [Google Scholar]
- Weak, improper, C−O...H−C hydrogen bonds in the dimethyl ether dimer. J. Am. Chem. Soc.. 2002;124:2739-2743.
- [CrossRef] [Google Scholar]
- Susceptibility of two Southern Ocean phytoplankton key species to iron limitation and high light. Front. Mar. Sci.. 2019;6:167.
- [CrossRef] [Google Scholar]
- Isolation and growth of cyanobacteria from marine and hypersaline environments. The Prokaryotes. 1981;221–223
- [CrossRef] [Google Scholar]
- An equation of state for the thermodynamic properties of dimethyl ether. J. Phys. Chem. Ref. Data.. 2011;40:023104
- [CrossRef] [Google Scholar]
- Characterisation and stability of pigments extracted from Sargassum binderi obtained from semporna, sabah. Sains Malays.. 2014;43:1345-1354.
- [Google Scholar]
- Facile synthesis and temperature-dependent luminescence of single-phase dual-emitting CdTe@NaNO3 composite. Mater. Res. Bull.. 2017;95:419-425.
- [CrossRef] [Google Scholar]







