Translate this page into:
Flavokawain mitigates ovalbumin (OVA)-induced asthma by regulating NLRP3 inflammasome and inflammatory markers: In-vitro and in-vivo models
⁎Corresponding author at: Department of Respiratory and Critical Care Medicine, Xi’an GaoXin Hospital, Xi'an, 710000, China bushzhang0423c@sina.com (Chuang Zhang)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Background
Asthma is the most prevalent chronic inflammatory ailment, which has major communal health implications for both adults and children worldwide. Flavokawain is a chalcones present in kawa plant extracts. They have anti-inflammatory, antioxidant, anti-cancer, antimicrobial, and other activities.
Methods
In the present investigation, the potential of flavokawain on the IL-4-induced 16HBE cell line and the ovalbumin (OVA)-induced mouse model was analyzed.
Results
The findings revealed that the IL-4-induced 16HBE cell line treated with flavokawain reduced cell apoptosis and increased cell proliferation. The NLRP3 inflammasome activity was suppressed by flavokawain. The level of immune cells was increased in the asthmatic mice, which was effectively reduced by the treatment with flavokawain. IgE, eotaxin, TNF-α and TXB levels were decreased by the flavokawain treatment in the asthmatic mice. Imbalances in Th1 and Th2 cytokines were effectively modulated by the flavokawain treatment in the asthmatic mice. The balance between the Th1 cytokines (IL-12 and IFN-γ) and the Th2 cytokines (IL-4, IL-5, IL-6, and IL-13) was maintained by flavokawain. The histopathology study of lungs revealed that the thickness of the airway membrane and inflammatory cell infiltration was reduced by the flavokawain, which indicates the decrease in inflammation.
Conclusion
These findings recommend that flavokawain can be used as a potent candidate to treat asthma in the future.
Keywords
16HBE cell lines
Ovalbumin
Flavokawain
Allergic asthma
Inflammatory markers
NLRP3 inflammasome
1 Introduction
Asthma is the most prevalent and highly complex inflammatory airway disease, which affects both young people and adolescents. The major characteristics of asthma are inflammation, edema, airflow obstruction, airway narrowing, and mucus hypersecretion. (Dharmage et al., 2019) The prevalence of asthma is notably increased in children, whereas the morbidity and mortality rates are high in adolescents. (Stern et al., 2020) The risk factors for the development of asthma are microbial infections, abnormalities in toll-like receptors (TLR) response, interferon response, and respiratory tract epithelial functions. (Christou et al., 2019) Inflammatory cells such as T cells, eosinophils, and neutrophils are closely associated with the immunological and inflammatory reactions in response to allergens in asthma. (Hamid and Tulic, 2009).
In terms of treatment, the currently existing therapies mainly target the symptoms and fail to provide long-term benefits. Apart from several improvements in the medical field, the gold standard treatments for asthma still lack. The currently prescribed medications are also often reported to have numerous side effects such as damaging the renal, hepatic, cardiovascular, digestive, and immune systems. (García-Menaya et al., 2019) Hence, the need for the development of new therapeutic strategies in order to overcome the treatment complications and achieve successful management of asthma without side effects is highly demanded.
In recent times, researchers have shifted their research interests to the naturally sourced bioactive compounds for the successful management of several inflammatory diseases. The most prevalent flavonoid/isoflavonoid compound found in vegetables and fruits is chalcones. (Li et al., 2019).
The chalcone compound has already been reported to show several biological and pharmacological activities. (Abdullah et al., 2014 Jun; Nazar et al., 2015; Hameed et al., 2016 Apr) Flavokawain A, B, and C, which make up 0.46 percent, 0.015 percent, and 0.012 percent of kava extracts, are chalcones. It was already reported that the chalcones had immense pharmacological activities such as anti-inflammation, antioxidant, antiulcer, antiprotozoal, and antibacterial properties. (Zi et al., 2005).
Previous studies have revealed that flavokawain protects against lung injury and sepsis in mice, (Luo et al., 2021) antifibrotic and antioxidant activities, (Hseu et al., 2019) antitumor effects against gastric cancer, (Hseu et al., 2020) prostate cancer, (Wang et al., 2020) lung cancer, (Li et al., 2020; Phang et al., 2021) and colon cancer. (Jiao et al., 2018) However, the anti-asthmatic activity of flavokawain was not scientifically studied yet. Hence, here were intended to investigate the beneficial properties of flavokawain against the allergic asthma in an interleukin (IL)-4-activated airway bronchial cell lines (in vitro) and murine model of asthma (in vivo) through the regulation of NLRP3 inflammasome pathway (Fig. 1).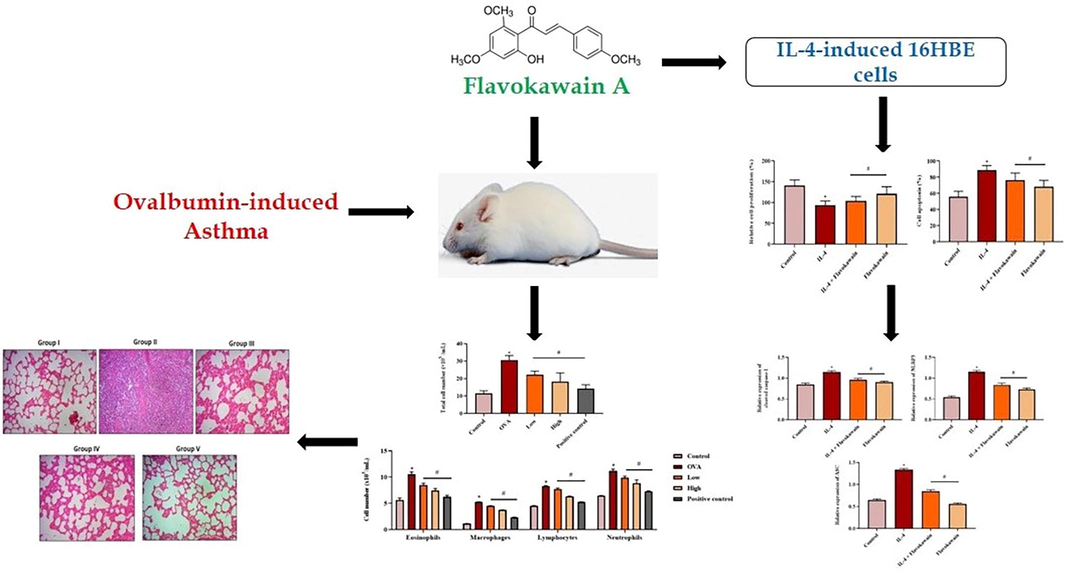
Graphical representation of mode of action of flavokawain A.
2 Materials and methodology
2.1 Chemicals
Aluminium hydroxide, ovalbumin, flavokawain A (Fig. 2), and other chemicals were obtained from Sigma Aldrich, USA. The assay kits for biochemical assays were procured from Thermo Fisher Scientific, USA.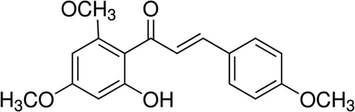
Chemical structure of flavokawain A.
2.2 Cell culture and treatment procedure
The human bronchial epithelial cell line (16HBE) was collected from the American Type Culture Collection (ATCC), USA, and grown on RPMI-1640 medium enriched with 10% FBS, 100 µg/mL of streptomycin, and 100 µg/mL of penicillin. After reaching the 80% of confluency, the cells were trypsinized and used for the additional assays. The 16 HBE cells were grouped into four groups: control, 100 ng/µL of IL-4-activated cells, IL-4 (100 ng/µL) + flavokawain (100 µM) treated cells, and 100 µM of flavokawain alone treated cells.
2.3 Cell growth measurement
The growth levels of control and treated 16 HBE cells were measured by the 5-bromo-2-deoxyuridine (BrdU) technique. Cells were loaded onto the 96-well plates with growth media comprising 100 ng/µL of IL-4 with or without flavokawain (100 µM). After 48 h, wells were loaded with 10 µL of BrdU (1 mg/mL) and incubated for 4 hr. Then added BrdU antibody for 1 hr. Then the cells were cleansed thrice with chilled buffered saline. After that, 200 µL of substrate solution were loaded into wells and incubated for 25 mins before adding H2SO4. Finally, the absorbance was measured by utilizing a spectrophotometer at 490 nm. The relative cell proliferation rates were calculated using the obtained absorbance value. Three duplicate wells were used to test each group.
2.4 TUNEL assay
The level of apoptotic cell death in the control and treated 16 HBE cells was estimated using the TUNEL assay. The 16 HBE cells were cultured for 24 hr and treated with 100 ng/µL of IL-4 with or without flavokawain. The cells were counterstained using DAPI fluorescent dye to perform the cell count. The images of the apoptotic cells were obtained by utilizing a fluorescence microscope. The total number of DAPI-stained cells and TUNEL-positive cells was evaluated. TUNEL-positive/DAPI-stained cells were used to reflect the total % of apoptotic cells. The experiments were carried out thrice and the average was calculated.
2.5 Estimation of NLRP3 inflammasome activity
The expression levels of cleaved caspases-1, NLRP3, and ASC in the control and treated 16 HBE cell lysates were determined by utilizing commercial ELISA assay kits based on the given protocols by the manufacturer (Thermofisher Scientific, USA).
2.6 Experimental animals and treatment groups
The 5–6-week-old BALB/c mice were procured and utilized in the current study. They were maintained at controlled laboratory conditions with a 24 °C temperature and alternative 12 hrs of light and dark cycles. All the animal studies were performed as per the institutional animal ethical committee-approved guidelines. All mice were separated into five groups with 6 in each (n = 6). Group I was considered to be the control group. Group II was intraperitoneally administered with 10 mg/kg of ovalbumin (OVA) along with 1 mg/kg of aluminium hydroxide on the 0th and 5th days to induce asthma. In group III animals, asthma was induced by OVA administration and treated with 25 mg/kg b.wt of flavokawain for 21 days. In group IV animals, asthma was induced by OVA administration and treated with 50 mg/kg b.wt of flavokawain for 21 days. Group V animals were treated with the standard drug dexamethasone.
2.7 Determination of inflammatory cell counts
Blood samples were obtained from the experimental mice in a heparinized tube and then processed for the determination of inflammatory cell counts. The blood samples were then centrifuged at 4 °C for 10 mins at 500g. The obtained pellets were rinsed with 0.5 mL of buffered saline and used for the differential count. An automated cell counter was utilized to count the inflammatory cell counts. Using conventional morphologic determinants, aliquots of the cells were mounted on slides and labeled with Field's dye to detect eosinophils, macrophages, lymphocytes, and neutrophils.
2.8 Determination of eotaxin and IgE level
The bronchoalveolar lavage fluid (BALF) obtained from the experimental animals was centrifuged at 4 °C for 10 min at 1000g. After centrifugation, the supernatant was collected and used for IgE and eotaxin estimations. The status of eotaxin and IgE in the BALF of control and experimental mice was analyzed using commercial ELISA assay kits. The assays were done using the protocols recommended by the manufacturer (Thermofisher Scientific, USA). The tests were duplicated and the average value was used.
2.9 Determination of inflammatory markers
The levels of inflammatory markers including IL-4, IL-5, IL-6, IL-12, IL-13, and IFN-γ in the BALF of control and experimental mice were examined by using commercial ELISA assay kits. The experimental assays were conducted based on the protocols suggested by the manufacturer (Thermofisher Scientific, USA).
2.10 Determination of TNF-alpha and TXB2 levels
The status of TNF-alpha and TXB2 in the BALF of control and experimental mice were analysed using commercial ELISA assay kits based on the guidelines recommended by the manufacturer (Thermofisher Scientific, USA).
2.11 Histopathology studies
For histopathological analysis, the lungs were excised from both control and experimental mice and fixed in 10% formalin. Then dried in a series of graded ethanol, and then paraffin embedded. The paraffinized lung tissues were sectioned into thin slices at a thickness of 5 μm and stained with hematoxylin and eosin. The stained slides were analyzed under an optical microscope.
2.12 Statistical analysis
All data are reported as the mean ± SD of at least 3 replicates. The statistical tool SPSS version 15.0 was used for all statistical analyses. A one-way ANOVA analysis was used to assess the data, followed by a Bonferroni's test. P <.05 was seen as significant.
3 Results
3.1 Effect of flavokawain on cell proliferation and apoptosis
The effect of flavokawain on cell proliferation and apoptosis in the 16HBE cells is shoed in Fig. 3. The IL-4-activated 16HBE cells revealed decreased cell proliferation and elevated apoptosis when compared with the control cells. The IL-4-induced 16HBE cells treated with 100 µM of flavokawain showed increased cell growth and reduced cell apoptosis compared to the IL-4-alone-treated cells. There were no major changes between the control group and the flavokawain alone-treated group. The result revealed that flavokawain suppressed cell apoptosis and boosted cell proliferation.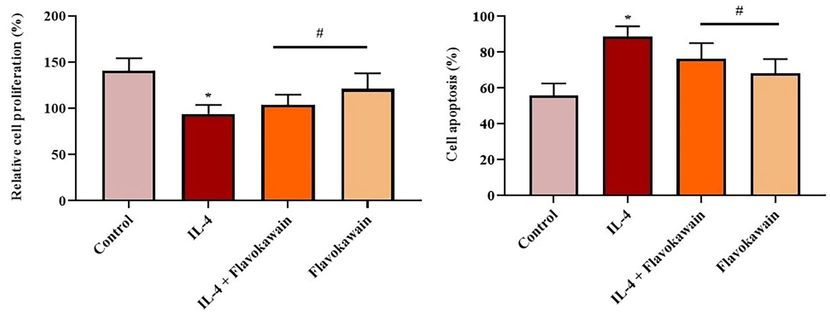
Effect of flavokawain on the cell growth and apoptosis in the IL-4-induced 16HBE cells. Flavokawain effectively increased the proliferation and inhibited the apoptosis in IL-4-stimulated 16HBE cells. Control: control 16HBE cells; IL-4: 16HBE cells treated with IL-4 (100 ng/µL); IL-4 + Flavokawain: 16HBE cells treated with flavokawain (100 µM) IL-4 (100 ng/µL); Flavokawain: 16HBE cells treated with flavokawain (100 µM) alone. The cell growth was determined by BrdU technique and apoptosis was investigated by TUNEL assay. *P <.01 vs control; #P <.05 vs IL-4 group.
3.2 Effect of flavokawain on caspase-1, ASC, and NLRP3 activity
The impacts of flavokawain on the expression of caspase-1, ASC, and NLRP3 in the 16HBE cells was showed in Fig. 4. The expression of caspase-1, ASC, and NLRP3 was upregulated in the IL-4-induced 16-HBE cells when compared with control cells. The caspase-1, ASC, and NLRP3 inflammasome expressions were effectively downregulated in the 100 µM of flavokawain-treated 16HBE cells. Flavokawain alone treated 16HBE cells and did not reveal major changes when compared with control. The results revealed that the flavokawain treatment is effective in regulating NLRP3, cleaved caspase-1, and ASC protein expression.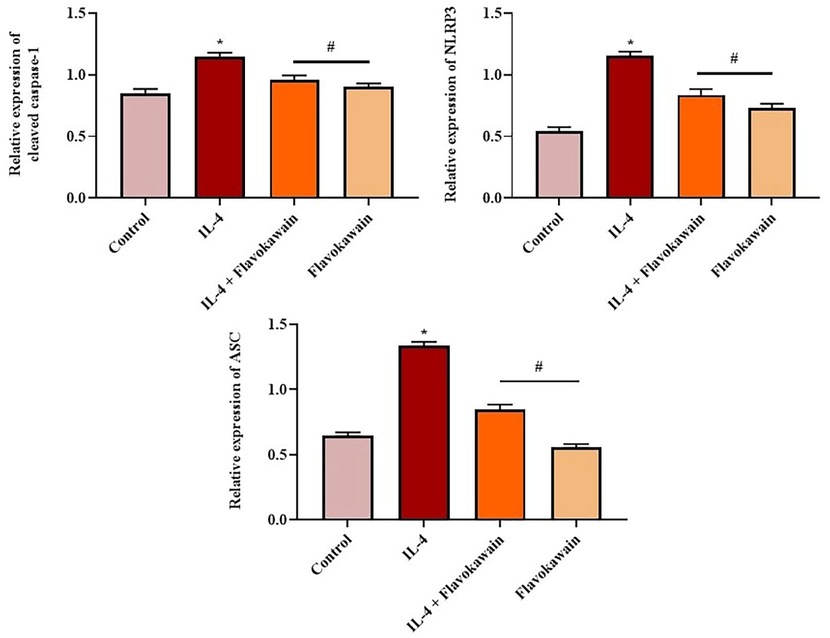
Effect of flavokawain on the expression of NLRP3 inflammasome markers in the IL-4-induced 16HBE cells. Flavokawain treatment effectively suppressed the NLRP3 inflammasome expression in the IL-4-stimulated 16HBE cells. The expression of caspase-1, ASC, and NLRP3 was standardized to GAPDH. Control: control 16HBE cells; IL-4: 16HBE cells treated with IL-4 (100 ng/µL); IL-4 + Flavokawain: 16HBE cells treated with flavokawain (100 µM) IL-4 (100 ng/µL); Flavokawain: 16HBE cells administered with flavokawain (100 µM) alone. *P <.01 vs control; #P <.05 vs IL-4 group.
3.3 Effect of flavokawain on inflammatory cell counts
The effect of flavokawain on the total leukocytes, eosinophils, macrophages, lymphocytes, and neutrophils in the control and experimental animals are revealed in Fig. 5. The OVA-induced mice revealed a boosted level of total leukocytes, eosinophils, macrophages, lymphocytes, and neutrophils when compared with the control. Interestingly, treatment with 25 mg/kg and 50 mg/kg of flavokawain effectively reduced the total leukocyte counts, eosinophils, macrophages, lymphocytes, and neutrophils in the OVA-induced mice. These findings showed the potential of flavokawain to reduce inflammatory cell counts.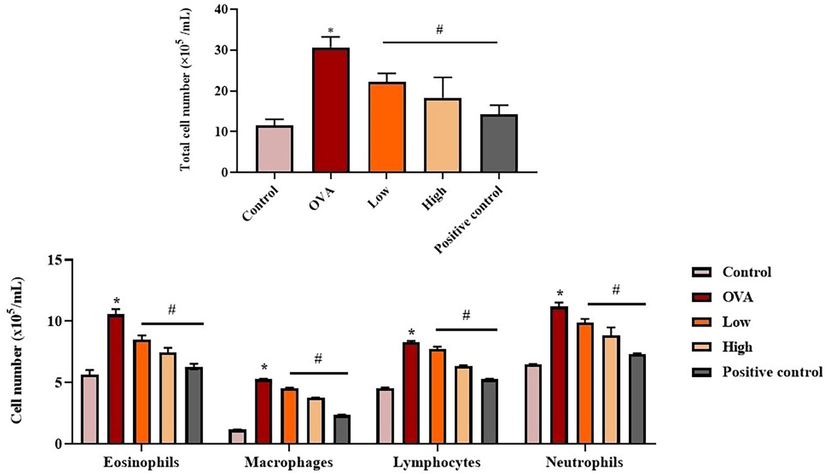
Effect of flavokawain on the inflammatory cell counts in the experimental mice. Flavokawain remarkably reduced the inflammatory cells in the asthma mice. The status of total leukocytes, eosinophils, macrophages, lymphocytes and neutrophils in BALF of experimental mice were determined. *P <.01 vs control; #P <.05 vs OVA group. Control: normal mice; OVA: asthma-induced mice; Low: 25 mg/kg of flavokawain treated asthmatic mice; High: 50 mg/kg of flavokawain treated asthmatic mice; Positive control: standard drug dexamethasone treated asthmatic mice.
3.4 Effect of flavokawain on the IgE and eotaxin levels
The influence of flavokawain on the status of OVA-specific IgE and eotaxin in the BALF of experimental mice are revealed in Fig. 6. There was a significant increment in the eotaxin and IgE status in the BALF of OVA-induced mice when compared with control groups. Meanwhile, treatment with 25 mg/kg and 50 mg/kg of flavokawain effectively depleted the levels of eotaxin and IgE status in the asthmatic mice. This result revealed the impact of flavokawain on the suppression of allergic reactions in the asthma mice.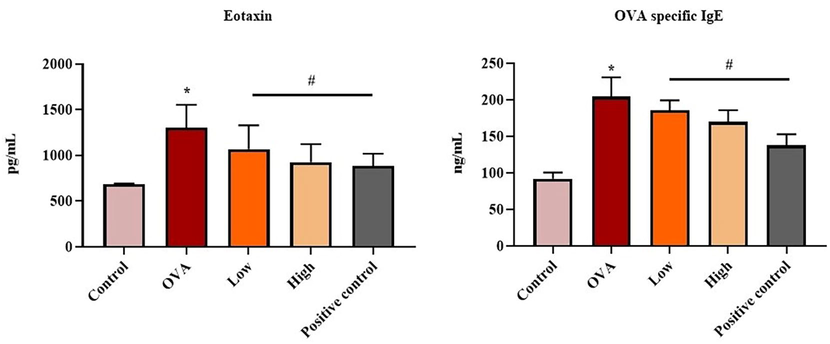
Effect of flavokawain on the IgE and eotaxin in the experimental mice. Flavokawain treatment reduced the IgE and eotaxin levels in asthmatic mice. Level of eotaxin and OVA specific IgE from experimental mice were estimated. *P <.01 vs control; #P <.05 vs OVA group. Control: normal mice; OVA: asthma-induced mice; Low: 25 mg/kg of flavokawain treated asthmatic mice; High: 50 mg/kg of flavokawain treated asthmatic mice; Positive control: standard drug dexamethasone treated asthmatic mice.
3.5 Effect of flavokawain on the inflammatory marker levels
The effect of flavokawain on the levels of inflammatory markers (IL-4, IL-5, IL-12, IL-13, IFN-γ, and IL-6) in the BALF of experimental mice was demonstrated in Fig. 7. The drastic increment in the status of inflammatory markers in the OVA-induced mice when compared with control. Interestingly, 25 mg/kg and 50 mg/kg of flavokawain appreciably reduced these inflammatory marker levels in the OVA induced group. These findings revealed the potential of flavokawain in decreasing the inflammatory marker levels in the OVA-stimulated asthmatic mice.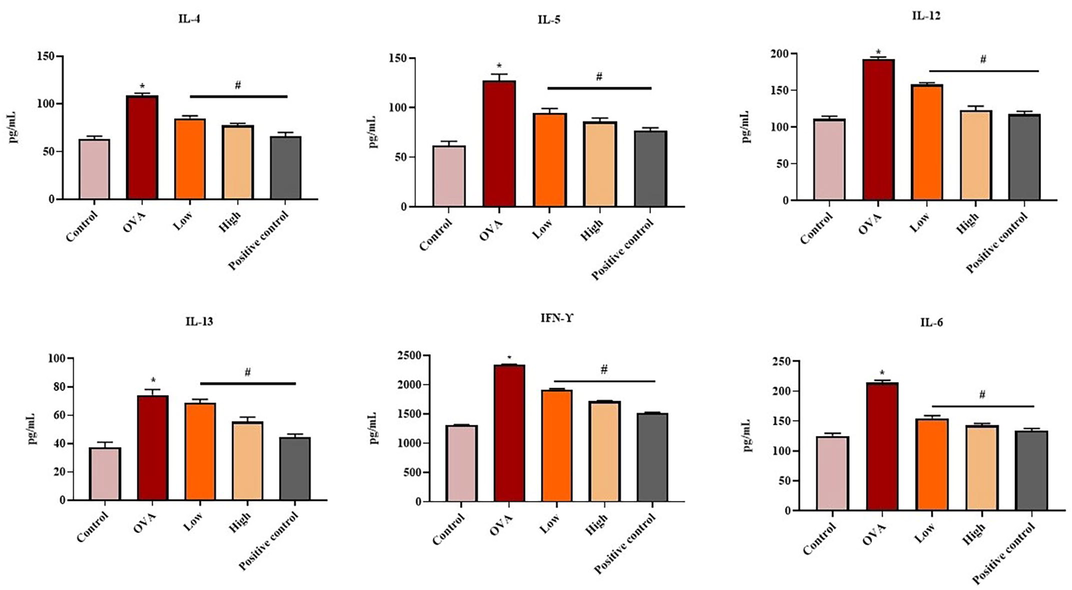
Effect of flavokawain on the inflammatory cytokines level in the experimental mice. Flavokawain treatment suppressed the levels of pro-inflammatory markers (IL-4, IL-5, IL-6, IL-12, IL-13, IFN-γ). Levels of IL-4, IL-5, IL-12, IL-13, IFN-γ, and IL-6 in different group of mice were measured by ELISA kits. *P <.01 vs control; #P <.05 vs OVA group. Control: normal mice; OVA: asthma-induced mice; Low: 25 mg/kg of flavokawain treated asthmatic mice; High: 50 mg/kg of flavokawain treated asthmatic mice; Positive control: standard drug dexamethasone treated asthmatic mice.
3.6 Effect of flavokawain on the TNF- α and TXB2 levels
The impact of flavokawain on the status of TNF- α and TXB2 in the BALF of experimental mice was revealed in Fig. 8. A remarkable increase in the levels of TNF-α and TXB2 was noted in the BALF of OVA-induced mice when compared with the control. Interestingly, the treatment with 25 mg/kg and 50 mg/kg of flavokawain effectively depleted the levels of TNF-α and TXB2 in the OVA-induced mice.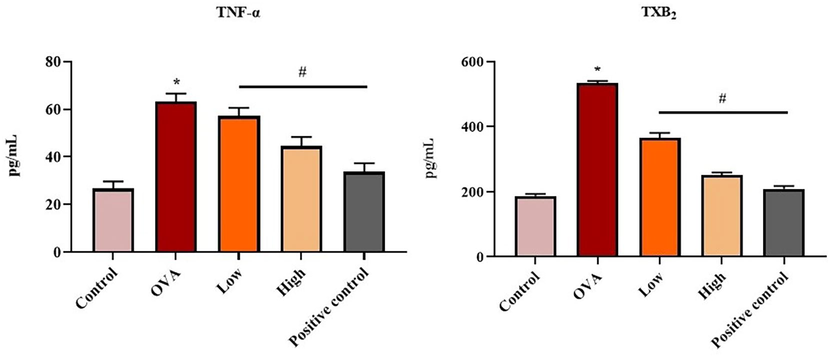
Effect of flavokawain on the TNF-α and TXB2 levels in the experimental mice. Flavokawain treatment reduced the TNF-α and TXB2 levels. The status of TNF-α and TXB2 status of different mice groups were measured by ELISA kits. *P <.01 vs control; #P <.05 vs OVA group. Control: normal mice; OVA: asthma-induced mice; Low: 25 mg/kg of flavokawain treated asthmatic mice; High: 50 mg/kg of flavokawain treated asthmatic mice; Positive control: standard drug dexamethasone treated asthmatic mice.
3.7 Effect of flavokawain on the histoarchitecture of lung tissue
The effect of flavokawain on the lung tissue histology of control and experimental mice was showed in Fig. 9. In the lung histology of control mice (group I), normal lung histology was observed. The lungs of asthma mice (group II) revealed increased inflammatory cell infiltrations in the peribronchial and perivascular regions and thickening of the airway epithelium and mucous membranes. The treatment with 25 mg/kg and 50 mg/kg of flavokawain (groups III and IV) exhibited reduced thickness in the epithelial membranes and inflammatory cell infiltrations. The standard drug dexamethasone treated asthma mice and also revealed decreased inflammatory cell infiltrations and epithelial thickness. These results revealed the potential of flavokawain to decrease the severity of allergic asthma.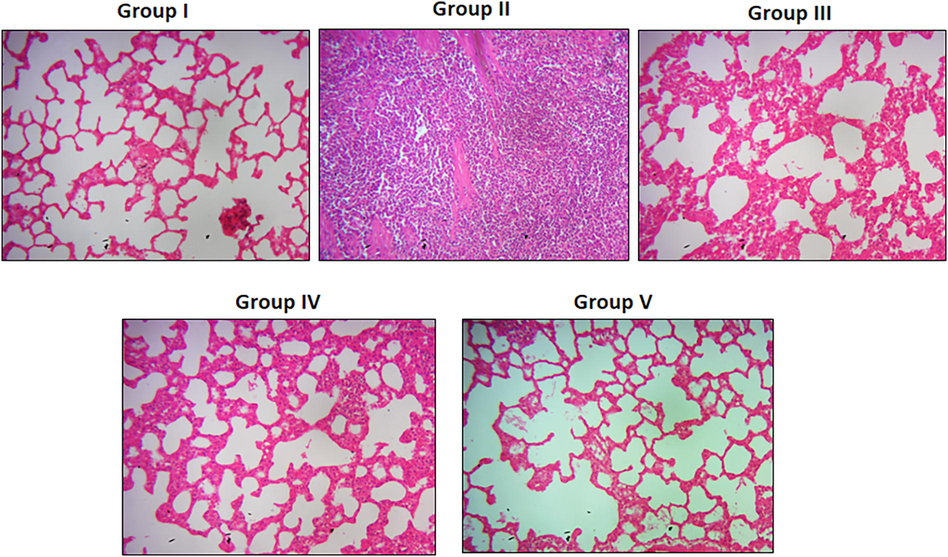
Effect of flavokawain on the lung histopathology of experimental mice. Flavokawain treatment reduced the thickness of airway membrane and inflammatory cell infiltrations in the lung tissues of asthmatic mice. Photomicrograph of lung sections were taken using optical microscope at 45 × magnification. Control: normal mice; OVA: asthma-induced mice; Low: 25 mg/kg of flavokawain treated asthmatic mice; High: 50 mg/kg of flavokawain treated asthmatic mice; Positive control: standard drug dexamethasone treated asthmatic mice.
4 Discussion
Asthma is a recurring chronic airway inflammatory ailment involving several immune and inflammatory regulators. (Zhang et al., 2020) Asthma is characterized by mucus hypersecretion, airway hyperresponsiveness, airway inflammation, and allergen-specific IgE secretion. Recently, the occurrence of asthma has elevated because of the upsurges in internal dust, environmental pollutants, and other allergens. (Zhao and Wang, 2018) Presently, the major therapeutic strategy is the utilization of glucocorticoids to reduce the symptoms of asthma. Meanwhile, conventional anti-inflammatory drugs could trigger several adverse effects and cause health complications. Hence, new treatment strategies to overcome these issues were required with improved efficacy and safety. (Grayson et al., 2018) In this work, in-vivo and in-vitro studies were done to investigate the effectiveness of flavokawain against the asthma model. The exact underlying molecular mechanisms of asthma development have not been uncovered yet, but airway inflammation is closely connected with the production of neutrophils, IgE, inflammatory cytokines, and NLRP3, which aggravate the asthmatic condition. (Dwyer et al., 2021) The increased levels of inflammatory cytokines play a major role in the pathophysiology of asthma. Following the release of cytokines, the mast cells invade the smooth muscle cells and attach to them. The stimulation of mast cells results in the accumulation of inflammatory mediators that result in bronchial hyperresponsiveness. (Wisnu Wardana and Rosyid, 2021).
Inflammatory cells and cytokines (IL-5, IL-14, and IL-4) are closely connected with the progression of asthma. An upsurge in the inflammatory cells is related to the production of various mediators, including leukotrienes, prostaglandins, and other inflammatory mediators, including TNF-α and IL-3, which are released by the eosinophils and mast cells. (Hu et al., 2021) Here, our findings proved that the OVA-induced mice revealed increased levels of leucocytes, eosinophils, macrophages, lymphocytes, and neutrophils when compared with control. Interestingly, the treatment with flavokawain effectively decreased the inflammatory cell counts in the OVA mice. These findings proved that flavokawain is effective in reducing the immune response in allergic asthma mice.
Apoptosis is an imperative cell death mechanism that is vital for the regulation of tissue homeostasis. (de Boer et al., 2008) The apoptosis in the epithelial cells during the development of acute asthma can lead to increased epithelial membrane permeability. (Zhu et al., 2020) Here, it was observed that IL-4-activated 16HBE cells exhibited elevated cell apoptosis and reduced cell proliferation. This indicates that IL-4 causes inflammation that leads to cell death. (Theofani et al., 2019) Interestingly, when the IL-4-activated 16HBE cells were treated with flavokawain, the cell proliferation level was augmented and apoptotic cell death declined. Flavokawain did not affect the viability of 16HBE cells, which indicates the non-toxic property of flavokawain.
The hyperactivation of the NLRP3 inflammasome occurs during asthma development in response to excessive inflammation. (Hosseinian et al., 2015) Inflammation is considered one of the key symptoms of airway diseases, including asthma. The NLRP3 inflammasome has a major contribution to the inflammation process. (Tsuchiya et al., 2019) Apoptosis and pyroptosis are two types of programmed cell death that are regulated by caspases, a family of cysteine proteases. IL-1β converting enzyme, cleaved caspase-1, can induce both pyroptosis and apoptosis in cells, and it is activated by the inflammasome. (Swanson et al., 2019) ASC is a protein that is crucially involved in assembling the inflammasome. (Kelley et al., 2019) NLRP3 is a tripartite protein that consists of three domains. NLRP3 protein (sensor), ASC (adaptor), and caspase-1 (effector) assemble to form NLRP3 inflammasome. (Yang et al., 2019) The NLRP3 inflammasome is a protein complex that promotes the secretion of the cytokines IL-1 and IL-18 and causes an cell death. NLRP3 inflammasome is vital for host immune defense against microorganisms but it is related to pathogenesis mechanisms when hyperactivated. (Yii et al., 2019) In the present investigation, there was an increased expression of ASC, cleaved caspases-1, and NLRP3 protein in the IL-4 activated 16 HBE cells indicating the elevated level of NLRP3 inflammasome expression which contributes to inflammation. Flavokawain significantly suppressed NLRP3 expression, caspases-1, and ASC in IL-4-stimulated 16HBE cells, suggesting that flavokawain can deactivate NLRP3 and restrict inflammation.
In T2 high asthma, eosinophils play a crucial role as a inflammatory cells. Blood eosinophil level is a basic biomarker that relates to sputum eosinophilia and increased blood eosinophils have been proven to predict the further exacerbation of disease. (de Groot et al., 2019) Macrophages are considered to be the primary defense system against pathogens. Lung macrophages have been associated with the commencement and development of asthmatic lung inflammation. (Peebles and Aronica, 2019) Antigen-specific IgE synthesis by B lymphocytes characterises an allergic reaction to an inhaled antigen are participated in the accumulation of cytokines. B and T lymphocytes are involved in activation cells responsible for inflammation reaction. (Radermecker et al., 2018) Neutrophils are the most prevalent immune cell and are recruited to the lungs during the asthmatic condition. (Reichard and Asosingh, 2019) In the OVA-induced mice, the total number of leukocytes is elevated indicating the asthmatic condition. The OVA-induced mice administered with the flavokawain exhibited decrement in the leukocytes. The status of neutrophils, macrophages, lymphocytes, and eosinophils are upregulated in OVA-stimulated mice and downregulated in the flavokawain administered OVA mice, which suggest the anti-inflammatory properties of flavokawain.
The epithelial cells and endothelial cells generate a chemokine known as eotaxin. It is generally found at a very high concentration in the lung endothelium of asthmatic patients. (Adeli et al., 2019) Eotaxin is a chemoattractant that is involved in eosinophil attraction. (Matucci et al., 2018) IgE has the ability to cause exceedingly fast pathological reactions and operate as an immune amplifier that is extraordinarily sensitive. (Jackson et al., 2020) An increased level of IgE performs critical functions in the pathology of asthma. (Zahedi et al., 2021) In the study, it was observed that the OVA-induced mice exhibited an elevated level of IgE and eotaxin, indicating the pathology of asthma. The levels of eotaxin and IgE declined in the OVA-induced mice treated with flavokawain in a dose-based manner. This outcome demonstrates that flavokawain can be a promising agent for treating asthma.
IL-4 and IL-13 are the cytokines generated by the mast cells and Th2 cells in the blood. IL-4 and IL-13 participate in a variety of inflammatory processes, including asthma and atopic inflammation. (Suzukawa et al., 2018) IL-12 is a protein secreted by neutrophils and macrophages that plays a crucial role in the Th1 inflammation process and is implicated in the regulation of inflammation as well as anti-infection immune responses. (Nagase et al., 2020) During allergic reactions, IL-5 is secreted by Th2 cells and participates in eosinophil differentiation. For allergic ailments, IL-5 has been proposed as a therapeutic target. (Piao et al., 2021) IL-6 and IFN-γ are Th2 cytokines and Th1 cytokines, respectively. Inflammatory cells are recruited and activated at the inflammatory site as a result of IL-6 secretion. (Boonpiyathad et al., 2019) An elevated IFN-γ status causes inflammation in the airway and is found in severe asthmatic conditions. (Lee et al., 2021) Disproportion in Th1 (IFN- γ and IL-12) and Th2 (IL-4, IL-13, IL-6, and IL-5) cytokines could lead to asthma. (Boonpiyathad et al., 2019) In this investigation, the status of Th-1 and Th-2 cytokines is elevated in the OVA-induced mice, which indicates the inflammatory condition of the airway. Treatment of the OVA-induced mice with flavokawain in a dose-based manner exhibited suppressed Th1 and Th2 cytokines, which suggests that flavokawain altered the disproportion of Th1 and Th2 cytokines and regulated the inflammatory responses in the asthmatic mice.
TNF-α is a significant cytokine in the immune response and also induces airway inflammation in asthma patients. (Chen et al., 2021) COX-derived TXA2, a strong bronchial smooth muscle spasmogen, is degraded non-enzymatically to produce thromboxane B2 (TXB2). It was already reported that TXB2 was significantly elevated in asthma patients. (Elliot et al., 2018) The OVA-induced mice exhibited a significantly elevated level of TNF-α and TXB2, which revealed the asthmatic condition. It was observed that the treatment with flavokawain suppressed TNF-α and TXB2 in the BALF of OVA-induced mice, which indicates a decrease in airway inflammation.
Asthma is characterized by an increased thickness of the airway wall owing to inflammation and increased mucus accumulation as a result of inflammatory cells infiltration into the airway walls.[49] The histopathology of the OVA-induced lung showed an increased thickness of the airway membrane, which indicates the progression of asthma. The OVA-induced mice treated with 25 mg/kg and 50 mg/kg of flavokawain exhibited thinning of the airway wall and a reduction in inflammatory cell infiltration, which suggests that flavokawain is involved in the protection against the airway remodeling in the OVA-induced mice.
5 Conclusion
Our findings suggest that treatment with flavokawain decreased cell apoptosis and NLRP3 inflammasome activity in the IL-4-induced 16 HBE cell line. In an animal model, flavokawain treatment effectively reduced the level of immune cells, IgE levels, eotaxin, and inflammatory biomarkers in the OVA-induced mice. These findings revealed that flavokawain can be used as a drug candidate in the future to treat allergic asthma.
Acknowledgments
This project was supported by Researchers Supporting Project number (RSP2023R5) King Saud University, Riyadh, Saudi Arabia.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Synthesis, characterization, theoretical, anti-bacterial and molecular docking studies of quinoline based chalcones as a DNA gyrase inhibitor. Bioorg. Chem.. 2014 Jun;54:31-37.
- [Google Scholar]
- Asthma exacerbation related to viral infections: an up to date summary. J. Family Med. Primary Care. 2019 Sep;8(9):2753.
- [Google Scholar]
- Boonpiyathad T, Sözener ZC, Satitsuksanoa P, Akdis CA. Immunologic mechanisms in asthma. InSeminars in immunology 2019 Dec 1 (Vol. 46, p. 101333). Academic Press.
- Immunologic mechanisms in asthma. InSemin. Immunol. Academic Press. 2019 Dec;46:101333
- [Google Scholar]
- Exhaled 15-HETE and thromboxin-B2 are associated with the therapeutic outcome in childhood asthma. Authorea Preprints 2021 Apr
- [Google Scholar]
- Asthma an undermined state of immunodeficiency. Int. Rev. Immunol.. 2019 Mar;38(2):70-78.
- [Google Scholar]
- Altered expression of epithelial junctional proteins in atopic asthma: possible role in inflammation. Can. J. Physiol. Pharmacol.. 2008;86:105-112.
- [Google Scholar]
- Oxidative stress and macrophages: driving forces behind exacerbations of asthma and chronic obstructive pulmonary disease? Am. J. Physiol.-Lung Cell Mol. Physiol.. 2019 Feb;316(2):L369-L384.
- [Google Scholar]
- Human airway mast cells proliferate and acquire distinct inflammation-driven phenotypes during type 2 inflammation. Sci. Immunol.. 2021 Feb;6(56):7221.
- [Google Scholar]
- Inflammation-dependent and independent airway remodelling in asthma. Respirology. 2018 Dec;23(12):1138-1145.
- [Google Scholar]
- Pharmacogenetic factors affecting asthma treatment response. potential implications for drug therapy. Front. Pharmacol. 2019 May;10:520.
- [Google Scholar]
- Advances in asthma in 2017.mechanisms, biologics, and genetics. J. Allergy Clin. Immunol.. 2018 Nov;142(5):1423-1436.
- [Google Scholar]
- Anti-HIV cytotoxicity enzyme inhibition and molecular docking studies of quinoline based chalcones as potential non-nucleoside reverse transcriptase inhibitors (NNRT) Bioorg. Chem.. 2016 Apr;65:175-182.
- [Google Scholar]
- The role of the NLRP3 inflammasome in pulmonary diseases. Ther. Adv. Respirat. Disease. 2015 Aug;9(4):188-197.
- [Google Scholar]
- Anticancer activities of chalcone flavokawain B from Alpinia pricei Hayata in human lung adenocarcinoma (A549) cells via induction of reactive oxygen species-mediated apoptotic and autophagic cell death. J. Cell Physiol.. 2019 Oct;234(10):17514-17526.
- [Google Scholar]
- The in vitro and in vivo anticancer properties of chalcone flavokawain b through induction of ros-mediated apoptotic and autophagic cell death in human melanoma cells. Cancer. 2020 Oct;12(10):2936.
- [Google Scholar]
- Fructose 1, 6-bisphosphatase aggravates oxidative stress-induced apoptosis in asthma by suppressing the Nrf2 pathway. J. Cell Mol. Med. 2021 Jun
- [Google Scholar]
- Ability of serum IgE concentration to predict exacerbation risk and benralizumab efficacy for patients with severe eosinophilic asthma. Adv. Ther.. 2020 Feb;37(2):718-729.
- [Google Scholar]
- Jiao J, Wu J, Wang J, Guo Y, Gao L, Liang H, Huang J, Wang J. Ma Huang Tang ameliorates bronchial asthma symptoms through the TLR9 pathway. Pharm Biol. 2018; 56:580-593
- The NLRP3 inflammasome: an overview of mechanisms of activation and regulation. Int. J. Mol. Sci.. 2019 Jan;20(13):3328.
- [Google Scholar]
- Korean Red Ginseng affects ovalbumin-induced asthma by modulating IL-12, IL-4, and IL-6 levels and the NF-κB/COX-2 and PGE2 pathways. J. Ginseng Res.. 2021 Jul;45(4):482-489.
- [Google Scholar]
- Effect of Syringic acid on antioxidant biomarkers and associated inflammatory markers in mice model of asthma. Drug Develop. Res.. 2019 Mar;80(2):253-261.
- [Google Scholar]
- Activity and mechanism of flavokawain A in inhibiting permeability P–glycoprotein expression in paclitaxel resistance of lung cancer. Oncol. Lett.. 2020 Jan;19(1):379-387.
- [Google Scholar]
- Luo W, Yang LB, Qian CC, Ma B, Manjengwa GM, Miao XM, Wang J, Hu CH, Jin B, Zhang LX, Zheng C. Flavokawain B alleviates LPS-induced acute lung injury via targeting myeloid differentiation factor 2. Acta Pharmacologica Sinica. 2021 Nov 4:1-1.
- Is IgE or eosinophils the key player in allergic asthma pathogenesis? are we asking the right question? Res. Res.. 2018 Dec;19(1):1.
- [Google Scholar]
- The roles of IL-5 and anti-IL-5 treatment in eosinophilic diseases: asthma, eosinophilic granulomatosis with polyangiitis, and eosinophilic chronic rhinosinusitis. Allergol. Int.. 2020;69(2):178-186.
- [Google Scholar]
- Synthesis, structure–activity relationship and molecular docking of cyclohexenone based analogous as potent non-nucleoside reverse-transcriptase inhibitors. J. Mol. Struct.. 2015;1086:8-16.
- [Google Scholar]
- Proinflammatory pathways in the pathogenesis of asthma. Clin. Chest Med.. 2019 Mar 1;40(1):29-50.
- [Google Scholar]
- Flavokawain C exhibits anti-tumor effects on in vivo HCT 116 xenograft and identification of its apoptosis-linked serum biomarkers via proteomic analysis. Biomed. Pharmacother.. 2021 May;1(137):110846
- [Google Scholar]
- Anti-allergic effect of Fructus amomi on ovalbumin-induced asthma mice model. Anat. Bio. Anthropol.. 2021 Jun;34(2):45-52.
- [Google Scholar]
- Reichard A, Asosingh K. Endothelial Cells in Asthma. InAsthma-Biological Evidences. IntechOpen. 2019 May
- Stern J, Pier J, Litonjua AA. Asthma epidemiology and risk factors. InSeminars in Immunopathology 2020 Feb (Vol. 42, No. 1, pp. 5-15). Springer Berlin Heidelberg. 2020 Feb;42(1):5-15.
- Baseline serum CXCL10 and IL-12 levels may predict severe asthmatics' responsiveness to omalizumab. Res. Med.. 2018 Jan;134:95-102.
- [Google Scholar]
- The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat. Rev. Immunol.. 2019 Aug;19(8):477-489.
- [Google Scholar]
- Targeting NLRP3 inflammasome activation in severe asthma. J. Clin. Med.. 2019 Oct;8(10):1615.
- [Google Scholar]
- Caspase-1 initiates apoptosis in the absence of gasdermin D. Nat. Commun.. 2019 May;10(1):1-9.
- [Google Scholar]
- Flavokawain A inhibits prostate cancer cells by inducing cell cycle arrest and cell apoptosis and regulating the glutamine metabolism pathway. J. Pharmaceut. Biomed. Anal.. 2020 Jul;15(186):113288
- [Google Scholar]
- Wisnu Wardana VA, Rosyid AN. Inflammatory Mechanism and clinical implication of asthma in COVID-19. Clin Med Insights: Cir Res Pulmonary Med. 2021 Sep;15:11795484211042711.
- Yang Y, Wang H, Kouadir M, Song H, Shi F. Recent advances in the mechanisms of NLRP3 inflammasome activation and its inhibitors. Cell Death Dis. 2019 Feb;10(2):1-1.
- Blood eosinophil count correlates with severity of respiratory failure in life-threatening asthma and predicts risk of subsequent exacerbations. Clin. Exp. Allergy. 2019 Dec;49(12):1578-1586.
- [Google Scholar]
- Effect of ambient air PM2. 5-bound heavy metals on blood metal (loid) s and children’s asthma and allergy pro-inflammatory (IgE, IL-4 and IL-13) biomarkers. J. Trace Element Med. Bio.. 2021 Dec;68:126826
- [Google Scholar]
- LncRNA-AK149641 associated with airway inflammation in an OVA-induced asthma mouse model. J. Bioenerg. Biomembr.. 2020;52:355-365.
- [Google Scholar]
- Flavokawainreduces NLRP3 inflammasome activation and Th1/Th2 imbalances in both in vitro and in vivo models of asthma. Clin. Exp. Pharmacol. Physiol.. 2020 Aug;47(8):1360-1367.
- [Google Scholar]
- A novel chalcone from kava extract, induces apoptosis in bladder cancer cells by involvement of Bax protein-dependent and mitochondria-dependent apoptotic pathway and suppresses tumor growth in mice. Cancer Res.. 2005 Apr;65(8):3479-3486.
- [Google Scholar]







