Functionalization of pre-gelatinized Urad bean fermented by Saccharomyces cerevisiae MK-157 as a fat replacer and its impact on physico-chemical, micromorphology, nutritional and sensory characteristics of biscuits
⁎Corresponding author. 786syedarsalanali@gmail.com (Syed Arsalan Ali)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The reduction of saturated fats in baked products has become an important unit operation in the food industry due to the consumers' demand for healthy foods low in saturated fat. In this research, a comprehensive biotechnological technique was explored to enhance the functional, technological, and nutritional characteristics of Urad beans as a fat replacer in biscuits via the combined treatment of gelatinization and fermentation by Saccharomyces cerevisiae MK-157. Fat in biscuits was replaced with fermented and gelatinized Urad bean flour (FGUBF) by S. cerevisiae MK-157 in the following proportions: 10%, 20%, and 30% wt/wt. The peak, final, breakdown, and setback viscosities of flour blends decreased. Protein, ash, crude fiber, antioxidants (DPPH, FRAP, ABTS, hydroxyl scavenging), bioactive compounds (total phenol content (TPC) & total flavonoid content (TFC)), and α-glucosidase inhibition increased, with the level of fat replacement in flour blends and their biscuits. However, FGUBF reduced the ash (4.32%) and fiber (10.96%) contents compared to the control sample (5.11% and 20.32%, respectively). The TPC and TFC of flour blends increased from 35.49 to 90.01 mg GAE/100 g and from 20.83 to 60.45 mg CE/100 g, respectively. However, baking decreased the values of TPC compared to flour blends, whereas TFC increased, and a similar increase in antioxidant activities was observed for fat-replaced biscuits. Phytic acid and total saponins were drastically reduced after fermentation by 98.83% and 98.85%, respectively. The microstructure of biscuits showed disoriented arrangements of protein, starch, fiber, and their complexes with disrupted gluten networks. Fermentation enhanced the In vitro protein digestibility (74.91 to 89.21%) of samples. Biscuits prepared from 20% FGUBF demonstrated desirable dimensional, textural, and sensory properties compared to control biscuits. Based on the results of this investigation, FGUBF might be recommended as an important natural fat substitute with enhanced nutrient bioavailability, antioxidants, and bioactive compounds for functional foods and nutraceuticals.
Keywords
Antioxidants
Urad bean
Fat replacement
Gelatinization
Solid-state fermentation
Biscuits
1 Introduction
Daily intake of high quantities of saturated fat has been reported as one of the significant causes of obesity. It triggers numerous non-communicable diseases and health problems, including cardiovascular diseases, cancer, hypertension, high level of low-density lipoprotein (LDL) cholesterol, and type-II diabetes (Peng and Yao, 2017). Generally, being overweight and obese are associated with high-income countries, but in recent times, these problems have been increasing in low income-countries along with undernutrition (Bhurosy and Jeewon, 2014). According to the World Health Organization, a balanced diet should not contain more than 30% and 10% of caloric values from total fat and saturated fat contents, respectively (WHO, 2018). It has been discovered that consuming saturated fats increases both total and LDL cholesterol levels (Marangoni et al., 2020).
Biscuits are considered one of the most popular bakery items that are inexpensive, easy to prepare, and have a longer shelf life than other bakery products (Saeed et al., 2020a; Yalcin et al., 2023). The main ingredients of biscuits are wheat flour, sugar, and fat which contributes to a poor nutritional profile. There is a dire need to produce low or non-fat, reduced-saturated, and trans-free-fat food products due to the growing impact of nutrition claims on consumers food purchase intentions. It is a major concern, especially for bakery items where solid fats like shortenings and butter are frequently used (Saeed et al., 2020a; Rakshit and Srivastav, 2022). Instead of lowering the total fat in food products, recent studies have focused more on replacing saturated fats, such as partially hydrogenated oil, because of their high trans-fat content (Saeed et al., 2020a; Giuffrè et al., 2022). However, saturated fats cannot be completely replaced with liquid oils or fat replacers without deteriorating the organoleptic properties of the final products (Yazar and Rosell, 2022; Giuffrè et al., 2022; Laganà et al., 2022). Saturated fats have been known to exhibit distinctive functional characteristics, i.e., loaf volume, crumb softness, taste (buttery, rich flavor), and the plasticity of shortenings. Therefore, saturated fats or shortenings could only be partially replaced with protein or carbohydrate-based fat replacers (Peng and Yao, 2017).
Legumes are a valuable and inexpensive source of proteins, dietary fiber, vitamins, minerals, and phytonutrients. Their daily intake is associated with numerous health advantages, including decreased risk of cancer, type-II diabetes, and heart diseases (Hughes et al., 2022; Langyan et al., 2022). Despite the increased growth rate of population, the per capita consumption of legumes has not increased over the past three decades (Hughes et al., 2022). Therefore, legume research should be promoted to produce innovative alternatives for food ingredients that can increase legume consumption and simultaneously satisfy consumer demand for healthy foods (Saeed et al., 2020a; Yazar and Rosell, 2022). Urad bean (Vigna mungo), also known as black gram or black bean, is a member of the Leguminoseae family, high in proteins, minerals, fiber, and phytochemicals. These compounds possess therapeutic properties, including antioxidant, anti-cancer, anti-diabetic, and cholesterol-lowering effects (Hughes et al., 2022; Langyan et al., 2022). The presence of polar compounds in Urad bean generates lubricity and creaminess, while the non-polar compounds provide lipid-soluble flavor-carrying capacity. These functional properties of Urad bean as a fat replacer are comparable to those of fat-containing bakery products (Saeed et al., 2020a; Yemenicioğlu et al., 2020).
In our previous research, raw Urad beans flour had been demonstrated as a fat replacer in biscuits which had successfully replaced 15% of saturated fat due to its functional properties, i.e., aqueous solubility, swelling power, water binding, and emulsifying capacity (Saeed et al., 2020a). Legume flours have high nutritional contents and perform desirably in heat-treated food products, making them appropriate components for use in baked products (Sparvoli et al., 2021). However, legume-based snacks face challenges such as the elimination or reduction of anti-nutritional factors (e.g., enzyme inhibitors, phytic acid, and tannins), low protein digestibility, long cooking time, and meeting consumer acceptance in terms of texture, flavor, appearance, color, and overall product acceptability (Adebo et al., 2022).
Milling legumes into flours can comparatively reduce the cooking time than the whole legume seeds. However, it is not enough to attain the demand of consumers for the preparation of high-quality foods within a minimal cooking time (Romano et al., 2021). Many technological treatments, including thermal treatments and fermentation, may overcome the abovementioned disadvantages (Romano et al., 2021; Adebo et al., 2022; Choe et al., 2022). Before milling, the thermal treatment of legume seeds is essential for utilizing legumes as functional food ingredients. The moist heat treatment causes the gelatinization of legumes starch by absorption of water molecules across the seed coat (Choe et al., 2022). Also, some of the middle lamellae of the cell wall are detached from the seeds of the legume as pectin degrades (Choe et al., 2022). As a result, it can increase the digestibility of macronutrients, including proteins and starch, in legumes flours (Romano et al., 2021; Choe et al., 2022). Fermentation of legumes improves texture, shelf life, taste, aroma, and digestibility and significantly reduces anti-nutrients (Adebo et al., 2022).
For the fermentation of foods, such as dairy items, drinks, and cereal-based products, a wide variety of yeast strains, most notably Saccharomyces cerevisiae, have been used (Tamang and Lama, 2022). Commercial probiotics made from the S. cerevisiae strain boulardii are utilized as food supplements for medicinal purposes (Tamang and Lama, 2022). The fermentation of dried cocoa beans had also been tested using various S. cerevisiae strains, including LM and KY794742 (Tamang and Lama, 2022; Chagas Junior et al., 2021). Bakery items developed by using fermentation techniques can be modified by choosing strains of microbes with particular pro-technological attributes (Bavaro et al., 2021). Exopolysaccharides (EPS) are produced by a number of lactic acid bacteria and yeast strains during fermentation, and they are similar to vegetal polysaccharides used as fat substitutes (Lynch et al., 2018; Lynch et al., 2021). According to their structure and mechanism of biosynthesis, the EPS are typically divided into homo-polysaccharides and hetero-polysaccharides (Bavaro et al., 2021). They are made up of repetitive units of at least two different types of monosaccharides and many types of linkages (Bavaro et al., 2021). EPS are renowned for their beneficial effects on the rheological characteristics of dough, texture, sensorial properties, and shelf-life of the final product (Bavaro et al., 2021).
Generally, only the endosperm of Urad bean is used for food preparations, and approximately 25% of the legume seeds are eliminated as waste by-products during milling (Girish et al., 2012). Whole Urad beans with their shells were treated in the present research to provide these by-products with commercial significance. Additionally, the outer covering/shells of legumes are regarded as organic wastes and are not consumed by humans or used in food items despite being a rich source of several bioactive compounds and antioxidants (Lynch et al., 2018). The literature revealed that treated (pre-gelatinized) Urad bean flour fermented by S. cerevisiae had not been previously used as a fat replacer in baked products. Therefore, the aim of this study was to investigate the combined effect of pre-gelatinization and fermentation of Urad bean as a fat replacer on nutrients, anti-nutrient contents, antioxidants, and bioactive compounds. Moreover, the functional and technological properties of flour and biscuit samples were analyzed for the prediction of fat replacement ability in bakery products. Thus, the utilization of these functional food sources in the biscuit industry will also improve environmental protection by reducing food waste.
2 Materials and methods
2.1 Collection of raw material
Fresh whole seeds of Urad beans (2 kg), refined wheat flour and eggs were purchased in July 2022 from the local market of Karachi, Pakistan. Sugar, salt, soya lecithin, and baking powder were procured from Allied Chemicals (Karachi, Pakistan). Hydrogenated vegetable oil (shortening) was procured from Dalda oil mills, Karachi and stored at 4 °C in amber glass sealed jar. All the chemicals were of analytical reagent grade purchased from Sigma-Aldrich, Germany and Dae-Jung chemicals, South Korea. The culture media was purchased from Oxoid, USA.
2.2 Preparation of raw and gelatinized Urad bean flour
Raw Urad bean flour (RUBF) was prepared by pulverizing in a laboratory mill (3100 Perten Instruments, USA). The equal particle size of ground flour was achieved by subsequently passing through a 60 µm sieve. Gelatinization of Urad bean was performed on pilot plant available at United King Foods (Karachi, Pakistan). Seeds were gelatinized by the hot water (100 °C) treatment for 15 min. The gelatinized seeds were then dehydrated in a forced air oven (05-B1106, Binder, Germany) at 60 °C for 8 h. The dried gelatinized seeds was further treated as mentioned for the RUBF. The flour samples were autoclaved for 20 min at 121 °C and 15 psi. Additionally, gelatenized and raw flour samples were kept in airtight glass containers at 4 °C untile used.
2.3 Microorganism and inoculum preparation
The S. cerevisiae MK-157 strain was selected due to the maximum production of enzymes and antioxidant activities of fermented Urad bean flour from our previous research (Ali et al., 2022). Strain was procured from the culture bank of University of Karachi, Pakistan. S. cerevisiae MK-157 was revived i.e., inoculated on Sabouraud Dextrose Broth (SDB) (Oxoid. USA) followed by incubation at 30 °C for 72 h. Then inoculum was centrifuged for 15 min at 6000g. Supernatant was discarded and pallet was wased thrice with saline water and resuspend in a mineral salt medium (MSM) (Ali et al., 2022). Sample was vortexed for 5 min then optical density (OD 600 nm) was determined and maintained at 0.5 OD.
2.4 Fermentation of gelatinized Urad bean flour
Solid state fermentation of 50 g gelatined Urad bean flour (GUBF) was carried out in perti dish by inoculating 38.8 mL of prepared inoculum. Substrate moisture was maintained at 80% by using MSM and incubated at 37 °C for 44 h. After incubation sample was autoclaved and dried for 24 h at 60 °C. Prior to use, samples were pulverised and sieved through a mesh screen of 60 µm and kept at 4 °C. Similar conditions were applied to the control sample (RUBF) without the addition of inoculum.
2.5 Preparation of fermented gelatinized Urad bean flour blends
To replace the same amount of fat in biscuit samples, fermented gelatinized Urad bean flour (FGUBF) was mixed with wheat flour (WF) in varied proportions of 10, 20, and 30% (w/w). The FGUBF-WF blends were evaluated for proximate composition, functional & technological properties, anti-nutrients, antioxidants, and bioactive compounds. Each batch of fat replacer were prepared in triplicates (i.e., 9 batches were prepared).
2.6 Biscuit preparation
Fat-replaced biscuits were developed according to the method of Saeed et al. (2021). Vanaspati ghee/shortening and powdered sugar were blended with the help of a dough mixer (Kenwood KVL4100W, UK) for 3 min, followed by the incorporation of fresh eggs and soya lecithin and mixed further for 5 min. FGUBF-WF blends, baking powder, and glucose were mixed into the mixture (of ghee, sugar and eggs) with the appropriate amount of water (Table 1). The developed dough was uniformly sheeted (thickness; 0.9 cm), and the biscuits were shaped into a circle (diameter; 3.9 cm) followed by baking in a preheated oven (Westpoint, WF-2610RK, China) at 180 °C for 25 min. After baking, biscuits were kept at room temperature (25 °C) for 10 min and then packed in airtight glass containers. Biscuits were manufactured in triplicate, and each batch contained 27 biscuits.
| Ingredients (g/100 g) | Control | 10% FGUBF | 20% FGUBF | 30% FGUBF |
|---|---|---|---|---|
| All-purpose wheat flour (g) | 100 | 100 | 100 | 100 |
| Fat/Shortening (g) | 40 | 36 | 32 | 28 |
| Icing Sugar(g) | 40 | 40 | 40 | 40 |
| Baking powder (g) | 2 | 2 | 2 | 2 |
| Egg (g) | 13.4 | 13.4 | 13.4 | 13.4 |
| Soya lecithin (g) | 0.25 | 0.25 | 0.25 | 0.25 |
| Salt (g) | 1 | 1 | 1 | 1 |
| Glucose (g) | 0.5 | 0.5 | 0.5 | 0.5 |
| Water (mL) | 20 ± 5 | 20 ± 5 | 20 ± 5 | 20 ± 5 |
| Fermented and Gelatinized Urad Bean Flour (FGUBF) (g) | – | 4 | 8 | 12 |
2.7 Functional properties and gluten content of flour blends
Water absorption and oil absorption capacities of RUBF, GUBF and FGUBF, and FGUBF-WF blends were determined according to the techniques of Ahn et al. (2005). Flour sample (1 g) was added to a tube, and 20 mL of distilled water was added, followed by mixing. Then tubes were centrifuged at 3000g for 10 min. The sediments were weighed and analyzed for water absoption capacity (WAC) after the removal of supernatants. For the analysis of oil absorption capacity (OAC) of flour samples, soybean oil was used in place of distilled water and was calculated similarly. Wet gluten, dry gluten, and gluten index of flour samples were determined by Glutomatic (2200, Pertan Instruments, USA). All the analyses were performed on a dry weight basis in triplicates.
2.8 Technological properties of dough and flour blends
The technology properties of sample are as follows:
2.8.1 Dough rheological behavior
According to the AACC Method 54-21 (2000), Brabender® Frainograph (OHG, Duisburg, Germany) was utilized to measure the impact of incorporating different levels of FGUBF to WF on dough rheology in the mixing process. In accordance with the experimental method, Farinograph water absorption (WA), dough development time (DDT), dough stability (DS), degree of softening (DoS), and farinograph quality number (FQN) were determined.
2.8.2 Pasting behavior of flour blends
The Brabender® Micro-Visco-Amylograph (OHG Duisburg, Germany) was used to measure the pasting characteristics of flour blends under various temperature and time conditions in accordance with AACC Method 22-10 (2000). The gelatinization time (GT) and pasting temperature (PT) were analyzed. In addition, peak (PV), final (FV), breakdown (BDV), and setback (SBV) viscosities were measured in Brabender Units (BUs).
2.9 Proximate composition of flour blends and biscuits
Proximate composition parameters i.e., moisture content (Method 44-40), protein (Method 08-01), ash (Method 46-10), crude fiber (Method 32-10), and fat content (Method 30-25) of RUBF, GUBF, FGUBF, and FGUBF-WF premix were assayed by using standard AACC Method (2000). Carbohydrate content was determined by subtracting 100 from the obtained values of moisture, protein, ash, crude fiber, and fat. The same methodology for moisture, protein, lipid, crude fibre, ash and carbohydrate were used in proximate analysis for biscuit samples (AACC, 2000). Moreover, caloric values were estimated by the Atwater general factor system: 4 kcal/g for carbohydrates and protein and 9 kcal/g for Lipids. All the analyses were performed on a dry weight basis in triplicates.
2.10 Antioxidant activities of flour blends and biscuits
Antioxidants activities of flour and biscuits samples were estimated by extracting the samples in 80% acetone. Variable concentration of extracts were prepared i.e. 10–500 mg/mL. Furthermore, samples were sonicated for 10 min and then vortexed for 35 min. The final extracts were centrifuged at 3500g at 10 min and supernenatant was utilized for analysis (Saeed et al., 2021).
2.10.1 Radical scavenging activity
The methodology of Fan et al. (2012) was utilized for 2,2-Diphenyl-1-Picrylhydrazyl (DPPH) assay. DPPH solution was prepared by mixing 33.9 mg of DPPH in 100 mL methanol. Sample extract and DPPH solution were mixed in equal amount (1 mL) and kept in dark for 30 min. Absorbance was measured through Spectrophotometer (Cary 60 UV–Vis, Agilent Technologies, Malaysia) at 517 nm. The % scavenging activity was calculated from the following formula:
Linear regression analysis was used to assess the IC50 values (mg/mL) of flour blends.
2.10.2 Ferric/Ferricyanide (Fe3+) Reducing Antioxidant Power (FRAP)
The standard method of Gawlik et al. (2016) for FRAP assay was used for the flour samples. Perl’s pursian blue color was prepared in reaction mixtures of solutions. Resulting intensity of color was measured at 700 nm. Increased absorbance readings suggest more FRAP as the intensity of produced color increases. FRAP % was determined as follows:
2.10.3 2,2-Azinobis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) radical scavenging activity
ABTS scavenging activity of flour blends was determined according to the method of Kong et al. (2015). After mixing 100 µL of extract of a sample with 5000 µL of ABTS working solution, the mixture was stored in a dark place for 10 min. The absorbance of the resulting reaction was measured at 734 nm. The % ABTS radical scavenging activity was determined as follows:
2.10.4 Hydroxyl radical scavenging activity (HRSA)
According to the method of De Avellar et al. (2004), hydroxyl free radical scavenging activity was determined. Extract of samples (0.2 mL), phosphate buffer (1 mL, pH; 7.4), 5 mM/L phenanthroline (0.2 mL), 7.5 mM/L FeSO4 (0.2 mL), 0.05% H2O2 (0.2 mL), and distilled water (3.2 mL) were added in a tube. Then tubes were incubated for 30 min at 37 °C, and absorbance was measured at 536 nm.
2.11 Bioactive compounds of flour blends and biscuits
The bioactive compounds of flour blends were determined as follows:
2.11.1 Total phenolic content
Total phenolic content was analysed by the method of Salar and Purewal (2017). The absorbance of the reaction mixture was measured at 765 nm in comparison to a blank sample. The results were expressed as milligrammes of gallic acid equivalent per 100 Gram (mg GAE/100 g) of the extract on a dry weight basis using the standard calibration curve.
2.11.2 Total flavonoid content
Total flavonoid content (TFC) of flour samples was estimated by the method of Dewanto et al. (2002). The absorbance of reaction mixtures was measured at 510 nm. On a dry weight basis, the extract was quantified in mg of Catechin equivalent per 100 g (mg CE/100 g).
2.12 Antinutrient content and In-vitro α-Glucosidase inhibition of flour blends and biscuits
Total saponins in samples were estimated as described by Lai et al. (2013) with minor modifications adapted by De Pasquale et al. (2021). Briefly, 10 mL of petroleum ether and 0.5 g of sample were mixed by shaking for 4 h. The residues were then extracted with 80% (v/v) aqueous methanol with continuous shaking for 4 h. The resulting extract was stored at 4 °C in the dark until further use. The spectrophotometric method was used to determine the total saponin content (TSC) (Lai et al., 2013). Results are presented as dry weight basis. Phytic acid was quantified via Haug and Lantzsch (1983) colorimetric technique. At room temperature, samples of flour blends sample were extracted with 0.2 M HCl. Stoppered test tubes containing 1 mL of iron (III) solution and 0.5 mL of the extracts were heated in a boiling water bath for 30 min. The tubes were then cooled for 15 min. After that, 2 mL of the 2,2-bipyridine solution was added, and the mixture was thoroughly mixed. After incubation for 30 min, absorbance was measured at 519 nm. Sodium salt of phytic acid was used as the reference standard to obtain the standard curve, and the results were expressed as milligrams of phytic acid/100 g of dry sample (mg PA/100 g).
The inhibition of enzyme was estimated by the slightly modified method of Kim et al. (2005). In a test tube, 50 μL of extract of the sample (60–185 mg/mL) was prepared in the phosphate buffer (50 mM, pH 7.0). Then substrate 4-nitrophenyl α-D-glucopyranoside 50 μL of (5 mM), and 100 μL of the enzymatic solution (0.1 U/mL) were added and incubated at 37 °C for 10 min. Reaction mixture was stopped by adding 1 mL of Na2CO3 (1 M). A control assay was performed using distilled water instead of the sample. The absorbance of the sample was recorded at 405 nm. The inhibition of the α-glucosidase was calculated from the following formula:
2.13 Physical properties of fat-replaced biscuits
The physical characteristics i.e. dimensional, textural and color of biscuits samples were determined as follows:
2.13.1 Dimensional analysis
The vernier calliper was used to measure the biscuits dimensions i.e. diameter (mm) and thickness (mm). The spread ratio of biscuits was determined by dividing the values of diameter and thickness (Saeed et al., 2021).
2.13.2 Textural analysis
The breaking force (Hardness) of biscuits was measured by texture analyzer (UTM, Zwick/Roell, Germany) functioning at a three points bend rig style i.e., trigger force, distance, pre-test speed, test speed, post-test speed and load cell: 50 N, 10 mm, 1.0 mm/s, 5.0 mm/s10.0 mm/s and 5 kg respectively (Saeed et al., 2021).
2.13.3 Color analysis
The color values was measured by a portable colorimeter (NH300, China) (Saeed et al., 2021). The colorimeter was set on different locations (thrice) of each biscuit surface and average values were determined. The a* and b* values, which represent the chromaticity values, showed (+) redness/greenness and (+) yellowness/blueness, respectively. While the L* value indicated lightness and was interpreted as 100 for pure white and 0 for black (Choe et al., 2022).
2.14 In vitro protein digestibility of biscuits
In-vitro protein digestibility (IVPD) of biscuit samples was measured according to Almeida et al.’s method (2015). Nitrogen (N) contents of the samples and undigested residues were determined using the Kjeldahl method (AACC, 2000). IVPD was calculated according to the following formula:
2.15 Microstructure of fat-replaced biscuits
Micrographs of the biscuit samples were captured at a magnification of 500× by using Scanning Electron Microscope (SM-6380 A, JEOL USA) (Saeed et al., 2021).
2.16 Sensory evaluation of fat-replace biscuits
The biscuit samples were judged by 95 semi-trained panellists (non-smokers male and female ranging in age from 22 to 55) from the University of Karachi (Pakistan). The sensory training was delivered to train the participants using the sensory profiling techniques for the conventional biscuits and prototypes (Heymann and Lawless, 2013). The aesthetic charaterstics of biscuits i.e., appearance, color, taste, texture, and overall acceptability were assessed by the semi-trained panellists using a 9-point hedonic scale (ranging from 9-like extremely to 1-dislike extremely). The biscuit samples were examined within 24 h after baking under daylight in portable cabins placed within the sensory laboratory. Biscuit samples were kept at room temperature (25 °C) for the analysis. During the analysis, water was provided to the judges so they could rinse their mouths. However, to test the accuracy of the findings, the control biscuit was served twice, simultaneously with the other biscuit samples.
2.17 Statistical analysis
All the anaylysis were performed in triplicates. The data were examined for Analysis of Variance (ANOVA) by using the Statistical Package for the Social Sciences (SPSS) software (Version 17.0. Inc., Chicago, USA). The Duncan's multiple range tests were performed to identify any significant differences (P ≤ 0.05) between the treatments. Correlation analysis was carried out by using OriginPro 2022 software (Version 9.90, USA) to determine the relationships between antioxidant activities (DPPH, FRAP, ABTS, and HRSA), bioactive substances (TPC and TFC), and enzymatic inhibition (Version 9.90, USA). Correlation graphs, also known as Correlograms, were designed to illustrate the results. The values of proximate composition, pasting, rheological, functional,anti-nutritional, α-glucosidase inhibition, and bioactive compounds of flour samples were compared using principal component analysis (PCA). The data of biscuit samples (anti-nutritional factors, antidiabetic potential, bioactive compounds, color values, dimensional, textural, nutritional, In Vitro protein digestibility, and sensory properties) were also subjected to PCA analysis using XLSTAT (Addinsoft, 2020; Version 2019.2.1, Paris, France).
3 Results and discussion
3.1 Functional properties and gluten content of flour blends
Table 2 shows the impact of gelatinization and gelatinization in combination with fermentation on the functional characteristics of RUBF. WAC and OAC of flour are essential functional properties that directly influence the application of flour on food products, especially bakery items Saeed et al. (2020). WAC of FGUBF-WF blends significantly (P < 0.05) increased with the increased concentration of FGUBF in the flour blends. The highest increase in WAC was observed for FGUBF 30% (206.71%), while the lowest increase was observed for 10% FGUBF (160.45%). The amount of water absorbed by WF was directly affected by the addition of FGUBF. The high fiber, protein, and carbohydrate contents of FGUBF-WF blends may be the reason for their higher WAC (Table 2) (Saeed et al., 2020a). Complex carbohydrates, fibers, and proteins from plants have demonstrated their success as effective fat replacers due to their ability to bind water and produce pastes that can mimic the texture and mobility of fats in food systems by providing lubrication, flow properties, and viscosity similar to fat (Patel et al., 2020). The two most desirable properties of bakery fat required for the development of different types of dough recipes are lubrication and moisture retention (Patel et al., 2020). The increased WAC of FGUBF-WF blends depicted their manifestation of these attributes of bakery fat (Rozan et al., 2022). In addition, the pre-treatment of gelatinization significantly enhanced (P < 0.05) the WAC (267.13%) compared to RUBF (256.34%). Similar observation of increased water absorption was reported by Choe et al. (2022) for gelatinized common bean flour.
| Sample | Water absorption capacity % | Oil absorption capacity % |
|---|---|---|
| Wheat flour (Control) | 137.32 ± 2.01a | 103.11 ± 2.02c |
| 10% FGUBF | 146.57 ± 2.11b | 106.69 ± 2.06d |
| 20% FGUBF | 155.82 ± 2.14c | 110.50 ± 2.09f |
| 30% FGUBF | 165.07 ± 2.17d | 114.20 ± 2.10 g |
| RUBF | 256.34 ± 2.19f | 107.73 ± 2.04e |
| GUBF | 267.13 ± 2.22 g | 101.20 ± 2.00b |
| FGUBF | 231.32 ± 2.21e | 92.42 ± 1.01a |
Each value is presented as the mean ± standard deviation (n = 3); the Significant difference (P ≤ 0.05) between the means is indicated by different letters with in the same column. The significance level is calculated by Duncan method. The values were computed using dry weight.
There are a number of potential reasons for the increased WAC after the boiling of legume seeds during gelatinization. The first reason could be the structural alteration of legume seeds (Choe et al., 2022). In addition, the gap between cotyledon cells expands after boiling, which may indicate the breakdown of the middle lamella. In turn, this can lead to more fluid retention and water permeability (Adebo et al., 2022; Choe et al., 2022). The second potential explanation is the gelatinization of starch and fiber swelling during heating which results in the loss of crystalline order with increased exposure of hydroxyl group surfaces that may interact with water molecules (Choe et al., 2022). The increase in hydrophilic amino acid residues caused by protein denaturation during thermal processing could be another possible reason for the increased WAC of GUBF (Choe et al., 2022). However, fermentation of pre-gelatinized legume flour decreased the WAC (231.32%). The development of soluble substances such as sugars and dextrin as a result of macromolecular breakdown during fermentation decreased the total water holding potential (Igbabul et al., 2012). The aforementioned tendency (i.e., reduced WAC) resulted in the intermolecular crosslinking inside the amorphous region of the starch granules due to the higher proportion of low molecular weight sugar fragments (Igbabul et al., 2012). Similar observation of decrease in WAC was depicted by Sharma and Sharma (2022) for fermented foxtail millet flour.
RUBF displayed increased OAC compared to GUBF (Table 2). Greater OAC of RUBF was responsible due to the dry heat treatment (i.e., sterilization) of legume flour that denatures protein molecules without moisture and exposes more hydrophobic regions on the surface (Stone et al., 2021). The electrostatic force and hydrogen bandings play a major role in the interaction between lipids and proteins. The ability of proteins to bind water and oil depends on internal elements, including amino acid content, protein structure, and surface polarity or hydrophobicity (Choe et al., 2022). The combined treatments of fermentation and gelatinization promoted a slight reduction in OAC (92.42%) and suggested that the reduction could be due to the higher amount of polar groups at the protein surface in contact with the neighbouring molecules of oil (Igbabul, et al., 2014). Igbabul et al. (2014) reported comparable findings of decreased OAC for fermented mahogany bean flour. However, the addition of FGUBF to WF enhanced the OAC of flour blends. The highest OAC was observed for 30% FGUBF (130.86%) and the lowest for 10% FGUBF (112.35%). Fat absorption is preferred in baked foods because it helps develop and retain flavor profile and prolongs the shelf life (Saeed et al., 2020a). Consistent observations of increased WAC and OAC of different levels of black gram flour incorporated in wheat flour as fat mimetic were depicted by Saeed et al. (2020a).
The values of gluten content of flour belnds are reported in Table 3. As the concentration of FGUBF in WF increased, the gluten content of flour blends decreased. The observed trend was expected due to the fact that Urad bean lacks gliadins and glutenins and may dilute the gluten network (Saeed et al., 2020a). Highest decrease in gluten index was observed for FGUBF 30% (77.59%), and the lowest decrease for FGUBF 10% (91.32%).
| Sample | Dry gluten % | Wet gluten % | Gluten index% |
|---|---|---|---|
| Wheat flour (Control) | 8.89 ± 0.14d | 26.03 ± 0.12d | 94.10 ± 1.30d |
| 10% FGUBF | 8.76 ± 0.16c | 24.30 ± 0.15c | 91.32 ± 1.24c |
| 20% FGUBF | 7.88 ± 0.18b | 23.11 ± 0.17b | 86.43 ± 1.10b |
| 30% FGUBF | 7.41 ± 0.19a | 21.80 ± 0.19a | 77.59 ± 1.07a |
| RUBF | – | – | – |
| GUBF | – | – | – |
| FGUBF | – | – | – |
Each value is presented as the mean ± standard deviation (n = 3); the Significant difference (P ≤ 0.05) between the means is indicated by different letters with in the same column. The significance level is calculated by Duncan method. The values were computed using dry weight.
3.2 Dough rheological characteristics
The effect of FGUBF-WF blends on dough mixing properties is presented in Table 4. There was a consistent increase in DDT (1.57–4.72 min) and farinograph WA (57.90–65.91%) with the addition of FGUBF in WF dough (Table 3). Increased farinograph WA was due to the presence of hydrophilic compounds (proteins, carbohydrates, dietary fibers, and polyphenolics) in FGUBF that promoted the water-binding capacity of dough (Saeed et al., 2020a). These hydrophilic components have more hydroxyl groups, which accelerates the reaction of OH groups with water molecules (Mironeasa et al., 2019). The DDT and DS are the indicators of flour strength; a decline in these values correlates with the weakening of dough (Mironeasa et al., 2019). The significant increase (P < 0.05) in water absorption may be the reason for the rise in DDT of dough samples prepared from FGUBF-WF blends, which promotes the association among hydrophobic biopolymers and allows minimal possibilities for gluten protein development. Thus weakens the gluten network and increases the time required to optimize dough formation by incorporating FGUBF in WF (Mironeasa et al., 2019). The stability of the control (WF) dough was found to be higher than the dough samples of flour blends. The RUBF does not contain gluten protein, and other proteins do not perform the same function as demonstrated by gluten. Therefore, it has been suggested that the incorporation of FGUBF components in WF dough produced weak and unstable gluten network.
| Samples | Farinograph properties | Micro-Visco-Amylograph properties | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Water absorption (%) | Dough development time (min) | Dough stability time (%) | Degree of softening (BU) | Farinograph quality number | Gelatinization time (min) | Peak viscosity (BU) | Final viscosity (BU) | Breakdown viscosity (BU) | Setback viscosity (BU) | Pasting temperature (°C) | |
| Wheat flour (control) | 57.90 ± 0.20a | 1.57 ± 0.01a | 8.59 ± 0.18d | 34.91 ± 0.90a | 87.71 ± 1.12a | 24.08 ± 0.11a | 1036.42 ± 10.02a | 1539.63 ± 18.12d | 377.81 ± 4.51d | 478.61 ± 7.40d | 58.11 ± 0.23a |
| 10% FGUBF | 61.40 ± 0.27b | 3.30 ± 0.03b | 5.70 ± 0.12a | 74.01 ± 0.81b | 95.72 ± 1.52b | 32.12 ± 0.30b | 1234.15 ± 17.11d | 1478.21 ± 16.04c | 352.17 ± 4.31c | 453.21 ± 7.31a | 61.71 ± 0.30b |
| 20% FGUBF | 63.50 ± 0.33c | 4.31 ± 0.05c | 6.40 ± 0.15b | 122.02 ± 1.30c | 108.51 ± 1.10c | 35.14 ± 0.41c | 1213.19 ± 14.11c | 1393.11 ± 15.06b | 336.14 ± 4.20b | 439.79 ± 6.40b | 62.14 ± 0.34c |
| 30% FGUBF | 65.91 ± 0.51d | 4.72 ± 0.08d | 7.32 ± 0.11c | 167.03 ± 1.70d | 113.72 ± 1.11d | 38.41 ± 0.60d | 1202.25 ± 12.13b | 1340.31 ± 13.34a | 327.49 ± 4.12a | 424.17 ± 5.15c | 63.38 ± 0.35d |
Each value is presented as the mean ± standard deviation (n = 3); the significant difference (P ≤ 0.05) between the means is indicated by different letters with in the same column. The significance level is calculated by Duncan method. FGUBF represents Fermented & gelatinized Urad bean flour.
The farinograph DoS was significantly (P ≤ 0.05) affected by the concentration of FGUBF incorporated in WF. The DoS is associated with a weak gluten network and poor dough strength (Saeed et al., 2020a). The increased values of DoS of dough samples prepared from fermented flour were due to the decreased gluten content and disrupted gluten network (Bakare et al., 2014). Disruption of gluten network was also confirmed by microstructural images of biscuit samples (Fig. 1). Additionally, Urad bean is abundant in phenolic compounds with significant antioxidant activity. The weakening impact on FGUBF-WF dough may be attributed to the increased quantities of phenolic compounds prevalent in FGUBF (Table 4) (Saeed et al., 2020a). Thus, it is possible to accept the changes in dough behavior based on the variations in farinograph WA, DDT, DST, and DoS because comparable findings were reported by Bakare et al. (2014) when different levels of fermented breadfruit flour were incorporated in WF dough.

- Appearance and scanning electron micrographs (at 500× magnification) of fat-replaced biscuits: (a, b); control (full fat biscuits), (c, d); FGUBF 10%, (e, f); FGUBF 20%, and (g, h); FGUBF 30%, respectively. Where, FGUBF; fermented and gelatinized Urad bean flour.
3.3 Pasting profile of flour blends
The textural and sensorial characteristics or mouth feel of baked products are directly influenced by the viscometric properties of flour. The incorporation of FGUBF as a fat replacer in flour blends significantly (P ≤ 0.05) altered the pasting behaviour of WF (Table 4). The pasting temperature of FGUBF-WF blends increased significantly (P < 0.05). The higher pasting temperature of flour blends than WF is ideal for recipes of food that needs to be thickened or require high gel strength (Xu et al., 2019). The rise in pasting temperatures was due to the increased amount of non-starchy compounds in flour blends, i.e., fiber, protein, etc. (Xu et al., 2019). These substances competed with starch for water and reduced water availability, ultimately increasing the values of pasting temperature (Xu et al., 2019). The peak viscosity of flour blends decreased significantly (P < 0.05) from 1234.15 to 1202.25 BU. The reduction in peak viscosity with the increased level of FGUBF in WF was due to the fact that FGUBF had higher WAC than WF, which ultimately resulted in the formation of a weak gluten network (Table 2) (Saeed et al., 2021). In addition, increased levels of protein and fiber contents in flour blends attracted the water molecules and hindered the swelling of starch granules of wheat and legume flours (Saeed et al., 2021). Another reason for decreasing trend of peak viscosity could be the less availability of intact starch granules because of the combined effect of fermentation and gelatinization results in the starch hydrolysis by microbial enzymes and heating in the presence of water, respectively (De Pasquale et al., 2020).
Similarly, with the increased concentration of FGUBF in flour blends, breakdown and setback viscosities decreased from 352.17 to 327.49 BU and 453.21 to 424.17 BU, respectively. The breakdown viscosity is correlated to the sustainability of starch granules at high temperature and shear stress (Xu et al., 2019). The decreasing trend in breakdown viscosity depicted the tendency of flour blends for greater shearing resistance and good paste stability (Stone et al., 2021). The lower values of setback viscosity indicated a lesser degree of retrogradation of starch components, more specifically, amylose content (Saeed et al., 2021). The reduced retrogradation could be attributed to the presence of phenolic components in flour blends (Table 6a), which may interact with the —OH groups of the amylose chain and prevent amylose molecules from arranging themselves together (Saeed et al., 2021). Moreover, results predicted that the incorporation of FGUBF in food products could significantly (P < 0.05) reduce the retrogradation, thereby inhibiting the phenomena of staling and increasing the shelf life (Saeed et al., 2021). Similar observations regarding the pasting profile were reported by Azeez et al. (2022) and Kim et al. (2021) for fermented brown finger millet flour and fermented lettuce incorporated in rice flour, respectively.
3.4 Proximate composition of flour blends and fat-replaced biscuits
The chemical composition of RUBF, GUBF, FGUBF, and FGUBF-WF blends is presented in Table 5. The moisture content of GUBF significantly increased (P < 0.05) after pre-gelatinization. A significant increase was also observed in the combined effect of fermentation and gelatinization. In contrast, crude fiber, ash, and fat contents significantly decreased (P < 0.05) after fermentation. The gelatinization process slightly reduced the protein content (19.89%) by 22.09% compared to RUBF (25.53%). This decrease was because of protein's polar nature, facilitating protein solubilization in an aqueous medium during the gelatinization (Heymann and Lawless, 2013). However, the fermentation of pre-gelatinized legume flour increased the protein content (23.60%) by 18.65%. This increase in protein content occurred due to the fermentation by S. cerevisiae which causes changes in nutrition levels by producing several enzymes and other metabolites (Munishamanna et al., 2017).
| Flour | Biscuits | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | Moisture (%) | Protein (%) | Fat (%) | Ash (%) | Crude fiber (%) | Carbohydrate (%) | Moisture (%) | Protein (%) | Fat (%) | Ash (%) | Crude fiber (%) | Carbohydrate (%) | Energy (Kcal/100 g) |
| Control | 7.02 ± 0.11a | 11.75 ± 0.12a | 0.91 ± 0.01a | 1.15 ± 0.03a | 0.45 ± 0.02a | 78.72 ± 0.61 g | 5.03 ± 0.16c | 1.62 ± 0.01a | 14.01 ± 0.10a | 25.12 ± 0.20d | 0.48 ± 0.26a | 53.74 ± 0.80d | 5.03 ± 0.16c |
| 10% FGUBF | 7.40 ± 0.14b | 12.70 ± 0.13b | 0.96 ± 0.07b | 1.32 ± 0.05b | 1.68 ± 0.07b | 75.94 ± 0.50f | 4.78 ± 0.11a | 2.12 ± 0.03b | 17.91 ± 0.12b | 22.16 ± 0.14c | 2.45 ± 0.07b | 50.58 ± 0.42c | 4.78 ± 0.11a |
| 20% FGUBF | 7.76 ± 0.16c | 13.68 ± 0.15c | 1.01 ± 0.08c | 1.50 ± 0.07c | 1.96 ± 0.08c | 74.09 ± 0.43e | 4.79 ± 0.13b | 2.47 ± 0.01c | 20.01 ± 0.11c | 19.28 ± 0.10b | 3.41 ± 0.10c | 50.04 ± 0.57b | 4.79 ± 0.13b |
| 30% FGUBF | 8.14 ± 0.19d | 14.58 ± 0.16d | 1.07 ± 0.10d | 1.66 ± 0.08d | 2.48 ± 0.10d | 72.07 ± 0.40d | 4.79 ± 0.14b | 2.98 ± 0.02d | 22.73 ± 0.15d | 16.41 ± 0.21a | 4.75 ± 1.03d | 48.34 ± 0.64a | 4.79 ± 0.14b |
| RUBF | 8.21 ± 0.15e | 25.53 ± 0.19 g | 4.64 ± 0.12f | 5.11 ± 0.10 g | 20.32 ± 0.17 g | 36.15 ± 0.27a | – | – | – | – | – | – | – |
| GUBF | 8.75 ± 0.16f | 19.89 ± 0.17e | 4.62 ± 0.11f | 4.71 ± 0.09f | 18.11 ± 0.15f | 43.92 ± 0.30b | – | – | – | – | – | – | – |
| FGUBF | 9.32 ± 0.17 g | 23.60 ± 0.18f | 1.28 ± 0.04e | 4.32 ± 0.08e | 10.96 ± 0.13e | 50.52 ± 0.32c | – | – | – | – | – | – | – |
Each value is presented as the mean ± standard deviation (n = 3); the Significant difference (P ≤ 0.05) between the means is indicated by different letters with in the same column. The significance level is calculated by Duncan method. The values were computed using dry weight.
Moreover, yeast itself is the source of numerous nutrients (Munishamanna et al., 2017). The leading causes of the rise in crude protein content during fermentation are the increase in microbial nitrogen, the ability of microorganisms to enzymatically destroy cell walls, and the creation of single-celled proteins by microbes (Heymann and Lawless, 2013). Similar observations of increases in moisture and protein contents and decreases in crude fiber and ash contents were reported by Rozan et al. (2022) for fermented avocado kernel flour. According to Igbabul et al. (2012), the reduction in crude fiber after fermentation may be caused by the conversion of some of the crude fiber and lignocellulosic components into protein by microbial fermentation. Another reason could be the enzymatic degradation of fibers by the microorganism after fermentation (Igbabul et al., 2012). Furthermore, minerals are utilized to some extent by microorganisms during their process of metabolism (Igbabul et al., 2012), which correlated with the lower value of ash content for FGUBF (4.32%) compared to unfermented samples, i.e., RUBF (5.11%) and GUBF (4.71%). In addition, the treatment of gelatinization also reduces the ash content due to the leaching of mineral content in water (De Pasquale et al., 2020). However, moisture, protein, fat, crude fiber, and ash contents increased in the flour blends as the level of incorporation of FGUBF increased in WF. As the concentration of FGUBF in WF increased, the gluten content of flour blends decreased. The observed trend was expected due to the fact that Urad bean lacks gliadins and glutenins and may dilute the gluten network (Saeed et al., 2020a). Highest decrease in gluten index was observed for FGUBF 30% (77.59%), and the lowest decrease for FGUBF 10% (91.32%). In general, fermentation, together with gelatinization, appeared to have a significant impact on the proximate composition of RUBF. In general, fermentation, together with gelatinization, appeared to have a significant impact on the proximate composition of RUBF.
The nutrient composition of control and fat-replaced biscuits produced from gelatinized legume flour fermented by S. cerevisiae is presented in Table 5. The fat-replaced biscuits depicted higher protein, ash, and fibre contents and lower fat content than the full-fat containing biscuits. As presented in Table 5, FGUBF had greater quantities of protein, ash, and fibre than WF. The same fact could be the possible reason for the increased concentrations of protein, ash, and fiber contents in fat-replaced biscuits (Table 5). The moisture content of FGUBF-WF biscuit samples showed no significant difference (P > 0.05) with the level of fat replacement. However, the contents of carbohydrates and fat significantly declined (P < 0.05), corresponding to the incorporation level of FGUBF in biscuits.
Moreover, the changes in the nutritional profile of biscuits were due to the nutritional contribution of fermentation by S. cerevisiae in GUBF or, more specifically rise in proteins, ash, and fibers that occurred in the biomass (Sharma and Sharma, 2022). Yeast itself is a source of several nutrients that may be associated with these variations in the nutritional levels of biscuit samples (Sandez Penidez et al., 2022). S. cerevisiae is the strain that is most commonly employed for the bio-production of different bio-based materials because it has several benefits, including the capacity to release extracellular enzymes, non-pathogenicity, and low cost (Tamang and Lama, 2022). This is related to the fact that the improvement in nutrients observed in this study is supported by the S. cerevisiae capacity to hydrolyze complex carbohydrates into sugars that are employed as a carbon source in the synthesis of high protein microbial biomass (Tamang and Lama, 2022). The high protein content of FGUBF-WF biscuits is important from a nutritional perspective since Urad bean proteins have a well-balanced amino acid composition with high levels of essential amino acids such as arginine and lysine, which are generally not present in cereal grains (Boye et al., 2010). Moreover, these two amino acids were reported to exhibit anti-hyperlipidemic activity in animal models (Kaushik et al., 2016). Furthermore, the reduction in fat content was due to the obvious reason of fat replacement, which resulted in a reduction in calories of FGUBF-WF biscuits compared to the control. Similar observations of a nutritional profile were reported by Christ- Christ-Ribeiro et al. (2021) for gluten-free cookies formulated from different ratios of bran fermented by S. cerevisiae.
3.5 Dynamic antioxidant profile of flour blends and fat-replaced biscuits
The antioxidant activities of compounds rich in antioxidants have been linked to numerous pathways, including chain reaction inhibition, binding of transition metal ion catalysts, peroxide breakdown, reducing capability, and potential to scavenge free radicals (Kong et al., 2015). A single test is typically insufficient to accurately evaluate the antioxidant activity of a putative antioxidant due to the complication of the oxidation-antioxidation reactions (Kong et al., 2015). Therefore, the antioxidant potential of FGUBF-WF blends and fat-replaced biscuit samples were assessed by DDPH, FRAP, ABTS, and HRSA. The stability of the free radical DPPH has been widely studied to predict the effectiveness of antioxidants (Azeez et al., 2022). Furthermore, hydroxyl free radical is known as the most reactive and toxic radical produced in living organisms. All types of macromolecules, such as lipids, proteins, nucleic acids, and carbohydrates, are susceptible to damage from this free radical (Azeez et al., 2022). At particular locations, the hydroxyl radicals in the cells can easily pass through the cell membranes, resulting in tissue damage and cell death (De Avellar et al., 2004). Therefore, eliminating hydroxyl radicals is crucial for the defense of living (De Avellar et al., 2004).
Antioxidant activities of unfermented flour, gelatinized flour, FGUBF, FGUBF-WF blends, and fat-replaced biscuits made from these flour blends are presented in Table 6a. The thermal treatment of gelatinization decreased the antioxidant potential compared to raw/untreated flour samples (RUBF). According to Choe et al. (2022), thermal treatment of gelatinization caused a reduction in total phenols due to the following reasons: polymerization chain reaction or disintegration of structures of the aromatic ring and partial transfer of soluble phenolic compounds into the processing water. Similar observations of reduced antioxidant activities were depicted by De Pasquale et al. (2020) for gelatinized legume flour. However, the process of fermentation by S. cerevisiae MK-157 significantly enhanced the antioxidant potential of gelatinized legume flour. Highest DPPH, ABTS, FRAP, and Hydroxyl scavenging activities were observed for FGUBF 30% and its biscuit samples which were depicted by their lowest IC50 values of 106.64 mg/mL, 83.42 mg/mL, 94.87 mg/mL, and 101.11 mg/mL for flour blends respectively. The biscuit samples showed IC50 values of 83.14 mg/mL, 51.09 mg/mL, 78.87 mg/mL, and 86.05 mg/mL, respectively for the same antioxidant activities. While the lowest DPPH, FRAP, ABTS, and HRSA, i.e. highest IC50 values were observed for control (WF) (478.21 mg/mL, 327.13 mg/mL, 384.98 mg/mL, and 413.56 mg/mL) and its biscuits (383.12 mg/mL, 267.61 mg/mL, 258.32 mg/mL and 373.76 mg/mL) respectively.
| Flour blends | Biscuits | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Samples | DPPH- Scavenging activity- IC50 (mg/ml) | FRAP-IC50 (mg/ml) | ABTS-Scavenging activity- IC50 (mg/ml) | Hydroxyl Scavenging activity- IC50 (mg/ml) | TPC (mg GAE/100 g DW) | TFC (mg CE/100 g DW) | DPPH- Scavenging activity- IC50 (mg/ml) | FRAP- IC50 (mg/ml) | ABTS-Scavenging activity- IC50 (mg/ml) | Hydroxyl Scavenging activity- IC50 (mg/ml) | TPC (mg GAE/ 100 g DW) | TFC (mg CE/100 g DW) |
| Control | 478.21 ± 4.35g | 327.13 ± 3.52g | 384.98 ± 3.87g | 413.56 ± 4.07g | 35.49 ± 0.11a | 20.83 ± 0.11a | 383.12 ± 3.83d | 267.61 ± 2.52d | 258.32 ± 2.85d | 373.76 ± 3.68d | 28.73 ± 0.12a | 22.41 ± 0.13a |
| 10% FGUBF | 312.40 ± 1.99f | 215.95 ± 1.51f | 281.53 ± 1.06f | 296.32 ± 1.15f | 54.29 ± 0.18b | 36.13 ± 0.78b | 287.40 ± 1.29c | 95.49 ± 0.78c | 119.11 ± 0.74c | 132.86 ± 1.36c | 49.60 ± 0.18b | 45.73 ± 0.42b |
| 20% FGUBF | 185.82 ± 1.21e | 117.14 ± 0.51e | 157.32 ± 0.82e | 173.42 ± 1.09e | 73.10 ± 1.31c | 51.42 ± 1.02c | 110.62 ± 0.81b | 72.64 ± 0.53b | 94.55 ± 0.55b | 101.31 ± 0.74b | 68.53 ± 1.17c | 67.91 ± 1.81c |
| 30% FGUBF | 106.64 ± 1.01d | 83.42 ± 0.35d | 94.87 ± 0.68d | 101.11 ± 0.28d | 92.01 ± 1.42d | 65.45 ± 1.12d | 83.14 ± 0.70a | 51.09 ± 0.19a | 78.87 ± 0.13a | 86.05 ± 0.63a | 89.13 ± 1.72d | 88.74 ± 1.01d |
| RUBF | 59.84 ± 0.92b | 23.81 ± 0.13b | 26.35 ± 0.12b | 32.34 ± 0.19b | 463.32 ± 2.55e | 375.41 ± 2.63f | – | – | – | – | – | – |
| GUBF | 63.82 ± 0.98C | 27.21 ± 0.15c | 29.73 ± 0.14c | 38.10 ± 0.22c | 451.50 ± 2.46 | 363.50 ± 2.50e | – | – | – | – | – | – |
| FGUBF | 56.14 ± 0.12a | 19.92 ± 0.11a | 24.30 ± 0.10a | 28.76 ± 0.14a | 470.18 ± 2.82f | 382.42 ± 2.74 g | – | – | – | – | – | – |
Each value is presented as the mean ± standard deviation (n = 3); the significant difference (P ≤ 0.05) between the means is indicated by different letters with in the same column. The values were computed using dry weight (DW). Control represents wheat flour and its biscuits without fat replacement, DPPH; 2, 2-diphenyl-1-picrylhydrazyl, ABTS; 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid), FRAP; Ferric/Ferricyanide (Fe3+) reducing antioxidant power, GAE; Gallic acid equivalent, CE; Catechin equivalent.
The results of antioxidants methods reported in Table 6a suggested that the antioxidants in flour and biscuit samples prevent the synthesis of different radicals or interact with the radicals to transform them into less harmful or harmless molecules, thereby terminating the radical chain reaction (Azeez et al., 2022). In addition, antioxidants have the ability to act as reducing agents by donating their electors to free radicals, which eventually hinders the production of peroxides (Lee et al., 2020). Literature revealed that fermented legume flour have high antioxidant activities. This may be due to the functional group addition i.e., hydroxyl or amino acid in antioxidant compounds and free phenolic acid production by the activity of microbial enzyme (Girish et al., 2012). Similar findings of increased antioxidant activities of fermented legume flour by lactic acid bacteria were reported by De Pasquale et al. (2020). In addition, the antioxidant activities of FGUBF-WF and biscuit samples were attributed to their phenolic contents, such as gallic, protocatechuic, gentisic, vanillic, syringic, caffeic, and ferulic acids, which have more effective reducing power and radical scavenging activities (Girish et al., 2012). An increase in the TPC of samples after fermentation was also witnessed in this research and reported in Table 6a.
The fermentation process is known to produce bioactive peptides through the proteolytic enzymatic activity of microorganisms (Li and Wang, 2021). The production of bioactive peptides could be the possible reason of the enhanced antioxidant potential of samples (Li and Wang, 2021). Furthermore, Li and Wang (2021) in their study reported that the Bacillus subtilis-fermented chickpea peptide showed strong radical scavenging activities. The data reported in Table 6a revealed that the synthesis of melanoidins during baking improved the antioxidant activities of biscuit samples compared to flour blends (Saeed et al., 2021). Similar observations of an increase in antioxidant activities of flour blends and biscuit samples were reported by Saeed et al. (2020a) when different ratios of black gram flour were incorporated in WF as a fat replacer in biscuits.
3.6 Total phenol content
Phenolic compounds from plant materials are characterized as natural antioxidants and thus attract great attention from researchers (Salar and Purewal, 2017). Numerous studies have demonstrated that these phenolic compounds have health-promoting activities, such as the ability to reduce cardiovascular diseases and prevent other degenerative disorders (Lai, et al., 2013; Salar and Purewal, 2017). Similar to the observations of antioxidant activities, decrease in TPC was observed for GUBF (451.50 mg GAE/100 g DW) compared to RUBF (463.32 mg GAE/100 g DW) due to the same reasons as aforementioned for the thermal treatment of gelatinization (Adebo et al., 2022). De Pasquale et al. (2020) also reported a decrease in TPC for gelatinized legume flour. However, fermentation resulted in an 8.46% increase in the TPC of FGUBF (470.18 mg GAE/100 g DW). The rise in the concentration of TPC in legume flour after fermentation was consistent with the findings of Christ-Ribeiro et al. (2021) for the solid state fermentation of rice bran by S. cerevisiae.
Previous studies have discovered that the majority of phenolic compounds are covalently bound to the components of the cell-wall structure, such as hemicellulose, cellulose, pectin, structural proteins, arabinoxylans, and lignin, whereas only a litter amount of phenolic compounds exist in plant materials as simple and free-soluble esters (Acosta-Estrada et al., 2014). Solid state fermentation, a sophisticated and superior biochemical procedure, enables microorganisms to secrete large amounts of hydrolytic enzymes (cellulases, pectinases, proteases, etc.). These hydrolytic enzymes hydrolyse the covalent bonds such as ether, ester, or glycosidic bonds between cell-wall structure components and insoluble bound phenolics, which ultimately resulted in enhanced extractability of phenolic compounds (Adebo et al., 2022).
Furthermore, the enzymatic potential of the strain S. cerevisiae MK-157 had been previously stated to produce cellulase, xylanase (Shariq and Sohail, 2019) and pectinase (Qadir et al., 2020). The values of TPC increased with the increased proportion of FGUBF in the flour blends and biscuit samples. However, a significant decrease (P < 0.05) in TPC was observed during baking. The highest destruction of TPC was observed in biscuits without fat replacement control, which showed an 18.95% loss after baking. The lowest TPC loss was observed in biscuit samples formulated with 30% FGUBF, which showed a 3.13% loss after baking. Regardless of the reduction in TPC during baking, FGUBF biscuits (10%, 20%, and 30%) displayed the maximum values of TPC in contrast to the control biscuits. Moreover, the formation of melanoidins during baking was insufficient to compensate the decrease in TPC (Saeed et al., 2020a; Sandez Penidez et al., 2022). In addition, the decrease in TPC in biscuit samples was due to the thermal decomposition of polyphenols and the removal of the carboxyl group from phenolic acids during thermal processing. A similar decrease in TPC upon baking was depicted by Sandez Penidez et al. (2022) for biscuit samples made from quinoa fermented by lactic acid bacteria.
3.7 Total flavonoid content
Flavonoids have been suggested as therapeutic agents (because of their potent scavenging and chelating properties) to prevent a wide range of diseases, including cardiovascular and neurological disorders (Barber et al., 2021). The results shown in Table 6a depicted that the FGUBF-WF blends and their fat-replaced biscuits have higher TFC due to the higher concentration of flavonoids in FGUBF. Similarly, the fat-replaced biscuits made from FGUBF-WF blends depicted the highest values (45.73–88.74 mg CE/100 g DW) of TFC, whereas control biscuits showed the lowest value (22.41 mg CE/100 g DW). Furthermore, previous works discovered that the Urad bean is a good source of flavonoid-containing substances such as epicatechin, catechin, rutin, quercetin, kaempferol, vitexin, and isovitexin (Lynch et al., 2018). The existence of these flavonoids in Urad bean may have contributed to the increase in TFC of FGUBF-WF blends and the resultant biscuits. However, fermentation of GUBF had significantly (P < 0.05) increased the TFC from 363.50 to 382.42 mg CE/100 g DW.
The release of bound flavonoid components during fermentation may be due to the increase in acid value, which made them more accessible and contributed to the increase in flavonoid content (Adebo et al., 2022). In addition, compared to FGUBF-WF blends, TFC significantly (P ≤ 0.05) increased during baking. The highest rise in TFC was observed in biscuit samples prepared from 30% FGUBF, which exhibited a 35.58% increase in TFC during baking, while the minimum rise was observed for FGUBF 10%, which depicted a 26.57% increase. The brown-colored pigments, or melanoidins, which are by-products of the Maillard reaction, typically emerge during the baking of biscuits. These pigments may be the cause of the observed variations in the values of TFC (Saeed et al., 2021).
3.8 Anti-nutritional content and ant-diabetic activity of flour blends and biscuits
Legumes contain harmful anti-nutritional chemicals which prevent the effective absorption of various nutrients, such as proteins, carbohydrates, vitamins, and minerals in the human body (Lai et al., 2013). The highest phytic acid (1302.21 mg/100 g) and saponin (1115.10 mg/100 g) contents were identified for RUBF (Table 6b). The concentrations of phytic acid and saponins were significantly (P < 0.05) reduced after gelatinization and fermentation. However, the highest decrease was achieved by the latter process, i.e., fermentation. Gelatinization had an impact on the values of phytic acid and total saponin content because the concentrations of these anti-nutrients in GUBF were up to 26.48% and 38.66% lower than the comparable RUBF. Fermentation of GUBF drastically decreased the contents of phytic acid and saponins by 98.83% and 98.85%, respectively. The degradation of anti-nutrients during gelatinization might be due to the leaching of phytates and aqueous soluble saponins in a heating medium (i.e., water) (Azeez et al., 2022).
| Flour blends | Biscuits | |||||
|---|---|---|---|---|---|---|
| Samples | Phytic acid (mg/100 g DW) | Total saponin (mg/100 g DW) | α-Glucosidase Inhibition - IC50 (mg/mL) | Phytic acid (mg/100 g DW) | Saponin (mg/100 g DW) | α-Glucosidase Inhibition - IC50 (mg/ml) |
| Control | 223.58 ± 1.09b | 1.02 ± 0.02b | ND | 195.19 ± 0.70a | 0.07 ± 0.00a | ND |
| 10% FGUBF | 224.69 ± 1.12c | 1.80 ± 0.03c | 154.30 ± 1.03f | 195.21 ± 0.74a | 0.09 ± 0.00a | 124.87 ± 0.18c |
| 20% FGUBF | 225.80 ± 1.14d | 2.59 ± 0.05d | 128.11 ± 1.01e | 195.23 ± 0.75a | 0.10 ± 0.00ab | 108.62 ± 0.14b |
| 30% FGUBF | 226.91 ± 1.17e | 3.37 ± 0.09e | 115.78 ± 0.98d | 195.27 ± 0.77a | 0.12 ± 0.00b | 97.78 ± 0.10a |
| RUBF | 1302.21 ± 13.02 g | 1115.10 ± 11.78 g | 77.76 ± 0.35c | – | – | – |
| GUBF | 957.34 ± 6.74f | 683.91 ± 4.87f | 75.87 ± 0.27b | – | – | – |
| FGUBF | 11.12 ± 0.06a | 7.83 ± 0.01a | 63.56 ± 0.12a | – | – | – |
Each result is presented as the mean ± standard deviation (n = 3); the significant difference (P ≤ 0.05) between the means in each separate column is denoted by different letters. The significance level is calculated by Duncan method. Where, ND; not detected, DW; dry weight basis. Control represents Wheat flour and its biscuits without fat replacement.
The possible mechanisms for decrease in saponins during fermentation are enzymatic degradation and the production of metabolites which can eliminate or reduce anti-nutrients (De Pasquale et al., 2020). The deterioration of phytic acid during fermentation might be due to the activation of phytases and phosphatases, liberating lower molecular weight biomolecules, including phosphates, inositol, and other nutrients (Menezes et al., 2020). In addition, fermentation by S. cerevisiae was previously reported to produce phytases and phosphatases (Menezes et al., 2020). The levels of phytic acid and total saponin contents of flour blends and their fat-replaced biscuit samples did not differ significantly (P > 0.05) from the control samples. Furthermore, the baking process significantly (P < 0.05) reduced the anti-nutrients compared to flour blends because heat treatment further facilitates the reduction of anti-nutrient compounds (Choe et al., 2022). Therefore, the consumption of biscuits produced from FGUBF-WF blends may provide better bioavailability of carbohydrates, proteins, minerals, vitamins, and other bioactive compounds. Similar trends of decrease in anti-nutrients by gelatinization and combined efcts of fermentation and pre-gelatinization were explored by De Pasquale et al. (2020) for legumes flour and Lee et al. (2020) for biscuit samples produced from fermented okara.
3.9 Dimensional and textural properties of fat-replaced biscuits
The dimensional and textural properties of control and fat-replaced biscuits are shown in Table 7. The incorporation of fermented and pre-gelatinized legume flour as a fat replacer in biscuits had a substantial influence on spread ratio, thickness, and diameter. The diameter of biscuit is directly linked with the spreading ability or the contraction of biscuits during baking (Saeed et al., 2020b). The diameter of fat-replaced biscuit samples increased with the addition of FGUBF due to the enhanced ability to spread during baking. The maximum increment in diameter was recorded for 20% FGUBF (41.77 mm). However, a reduction in diameter was observed for the level of fat replacement above 20%. However, the diameter of all the FGUBF-WF biscuits was still higher than the control biscuits (41.65 mm). The thickness of biscuit samples was increased significantly (P < 0.05) by the addition of FGUBF. In contrast, the highest thickness value was reported for control biscuits (Table 5).
| Sample | Diameter (mm) | Thickness (mm) | Spread Ratio (mm) | Breaking force (N) | L* | a* | b* |
|---|---|---|---|---|---|---|---|
| Control | 41.65 ± 0.56d | 7.84 ± 0.52d | 5.31 ± 0.10a | 21.85 ± 0.39c | 72.85 ± 0.32d | 4.21 ± 0.01a | 27.27 ± 0.10a |
| 10% FGUBF | 41.69 ± 0.40c | 7.08 ± 0.2a | 5.88 ± 0.14d | 16.72 ± 0.21a | 70.14 ± 0.27c | 4.74 ± 0.03b | 27.48 ± 0.12b |
| 20% FGUBF | 41.77 ± 0.88b | 7.25 ± 0.20b | 5.76 ± 0.12c | 20.89 ± 0.23b | 67.11 ± 0.13b | 5.52 ± 0.05c | 28.09 ± 0.17c |
| 30% FGUBF | 40.89 ± 0.51a | 7.62 ± 0.21c | 5.36 ± 0.13b | 23.87 ± 0.30d | 63.30 ± 0.10a | 6.31 ± 0.08d | 28.85 ± 0.19d |
Each result is shown as the mean ± standard deviation (n = 3); the significant difference (P ≤ 0.05) between the means in each separate column is denoted by different letters. The significance level is calculated by Duncan method. Where, control represents biscuits without fat replacement, L* values indicates the lightness (100; perfect white /0; perfect black), a* value indicates; (+) redness/(−) greenness and b* values indicates; (+) yellowness/(−) blueness.
One of the critical physical factors used to assess the quality of biscuits is the spread ratio, which is correlated with the texture and overall mouthfeel of the biscuits (Menezes et al., 2020). The spread ratio is primarily influenced by the expansion of the dough caused by leavening and the gravity flow during baking (Saeed et al., 2020b). A similar trend of increase in spread ratio was observed, as mentioned for the diameter of biscuit samples. In addition, 10% FGUBF (5.88 mm) and 20% FGUBF (5.76 mm) depicted higher values of spread ratio compared to 30% FGUBF (5.36 mm) and control (5.31 mm).
The reduction in the spread ratio of biscuits by 30% of fat replacement could be due to the increased concentration of water-binding components such as protein and fibers (Saeed et al., 2021). Moreover, these components create aggregates with available hydrophilic sites, lowering the amount of free water in the biscuit dough (Lee et al., 2020). During dough mixing, free water moves quickly via these hydrophilic regions, increasing dough viscosity and limiting biscuit spread (Saeed et al., 2021).
Hardness is a textural characteristic of bakery items and a preferred indicator by consumers for evaluating the quality of baked products (Bavaro et al., 2021). Therefore, the lower hardness values are more desirable (Saeed et al., 2021). The hardness of biscuits increased by increasing the fat replacement level. However, the breaking force of 10% FGUBF (16.72 N) and 20% FGUBF (23.87 N) was lower than the full fat-containing biscuits (21.85 N). Furthermore, the textural value of 30% FGUBF was not significantly different (P > 0.05) from the control. Consistent observations regarding dimensional and textural properties were reported by Saeed et al. (2020a) and Saeed et al. (2021) when black gram flour and date pit flour were used as fat-replacers in biscuits, respectively. The increment in hardness values was due to the increased hydrophilic components that trigger the water absorption of dough (Bavaro et al., 2021). This resulted in the development of a fragile gluten network and subsequently hardened the texture of the biscuits (Saeed et al., 2020b). Despite fat replacement, lower breaking strength of fermented legume biscuits was observed. It could be due to the fact that during fermentation, large fiber particles are broken down into smaller fiber particles which provides a crumblier texture. However, large particle size of fibers is associated with firmer texture (Lee et al., 2020). A similar observation of lower values of hardness of fat-replaced bread prepared from quinoa and amaranth flours fermented by Weissella cibaria was reported by Bavaro et al. (2021).
3.10 Color analysis of biscuits
Color, along with texture and flavor, is an essential factor of baked goods because it can considerably affect consumer acceptability (Rozan et al., 2022). The color of the biscuit samples was also affected by the level of fat replacement and represented in terms of L*, a*, and b* values (Table 7). The findings revealed that as the amount of FGUBF increased in biscuit samples, a* and b* values were gradually increased, whereas the L* value gradually decreased. The significant (P < 0.05) decrease in L* value of fat-replaced biscuits might be possible due to the increased contents of dietary fibers, proteins, and phytochemicals (i.e., phenols and flavonoids) (Sandez Penidez et al., 2022). Moreover, changes in color that occurred during the fermentation of GUBF may be responsible for the decrease in L* value and increase in a* and b* values (Sandez Penidez et al., 2022).
FGUBF-WF biscuits showed higher protein content (Table 5) which promoted browning reaction, i.e., generation of Maillard reaction end products (melanoidin pigment) during baking. Saeed et al. (2020a) and Rozan et al. (2022) depicted a similar trend of color profile when black gram flour was used as a fat replacer in biscuits, and fermented avocado kernel flour was used in biscuits preparation, respectively.
3.11 In-vitro protein digestibility of biscuits
The data of In vitro protein digestibility (IVPD) of biscuit samples are illustrated in Table 8. IVPD is one of the significant criteria in the assessment of protein quality and is a marker of protein bioavailability (Espinosa-Páez et al., 2021). It was observed that increased concentration of FGUBF in biscuits promotes protein bio-availability of samples. Control biscuits showed the lowest value of protein digestibility 74.21%, while 30% FGUBF presented the highest protein digestibility (89.28%). The rise in IVPD of fat-replaced biscuits could be because of fermentation that increases net protein utilization (NPR) and protein efficiency ratio (PER) (Azeez et al., 2022). Due to the presence of anti-nutrients in plants, the bioavailability of protein sources from plant origin is more likely to be affected compared to protein from animal origin (Lai et al., 2013) During fermentation, the release of proteolytic enzymes increases, which can break down complex proteins and degrade anti-nutrients, thereby releasing more peptides and amino acids (Azeez et al., 2022). Such interactions make the protein cross-linking more vulnerable to proteolytic attack, thus enhancing their digestibility (Adebo et al., 2022). Similar results of a decrease in anti-nutrients were also observed in this study (Table 6b). Control biscuits showed the lowest digestibility because the amino acid composition of wheat flour is deficient in essential amino acids such as lysine, threonine, and methionine, which may contribute to its poor protein digestibility (Wang et al., 2022). Moreover, baking biscuits increases the protein digestibility of legumes by enhancing the composition of amino acids and lowering the levels of anti-nutrient (Nosworthy et al., 2018), thereby confirming the beneficial effects of utilizing FGUBF as a fat replacer in biscuit production. Consistent observation of enhanced IVPD was reported by Espinosa-Páez et al. (2021) for biscuit samples prepared from fermented Phaseolus vulgaris and Avena sativa.
| Samples | In Vitro protein digestibility % |
|---|---|
| Control | 74.21 ± 1.01a |
| 10% FGUBF | 82.71 ± 1.10b |
| 20% FGUBF | 86.10 ± 1.13c |
| 30% FGUBF | 89.28 ± 1.46d |
Each result is shown as the mean ± standard deviation (n = 3); the significant difference (P ≤ 0.05) between the means in each separate column is denoted by different letters. The significance level is calculated by Duncan method. Where control represents biscuits without fat replacement, FGUBF; fermented and gelatinized Urad bean flour.
3.12 Scanning electron microscopy of fat-replaced biscuits
The scanning electron microscopic images of the internal cross-sectional areas of biscuit samples are presented in Fig. 1. The biscuit formulations show the clusters of proteins and starch that partially encase fat content in the biscuits. In these conditions, biscuits with higher amounts of fat (control and 10% FGUBF) seem to exhibit certain areas where the smooth surfaces can be interpreted as fat enveloping protein-starch complexes (Fig. 1b–d). The control biscuit dough (Fig. 1b) displayed flat surfaces corresponding to the matrix development between wheat starch and gluten. A network of some fragments of developed gluten appeared in 20% FGUBF biscuits (Fig. 1f). However, only a few small fragments of gluten were formed in 30% FGUBF biscuits which may have been caused by insufficient hydration of gluten and wheat endosperm (Fig. 1h) (Saeed et al., 2021).
Furthermore, a few smooth surfaces can be visualized in Fig. 1f for 20% FGUBF biscuits. While no smooth surfaces were noticeable in the biscuits with lower fat content (30% FGUBF), small and large disrupted protein bodies and deformed starch granules can be observed (Fig. 1h). From Fig. 1f–h it can be visualized that there was only a small amount of fat in the biscuits (20% FGUBF and 30% FGUBF) and was enough to lubricate the matrix. However, it was not good enough to generate the uniform smooth layer across the surfaces of these samples (Fig. 1f–h). The increased levels of fat replacement by 30% FGUBF caused the starch granules to be leached out from the discontinuous gluten matrix, which can be easily identified from Fig. 1d. The higher level of fat replacement (30% FGUBF) presented the development of a more compact structure than the other biscuit samples (Fig. 1f–h). The instrumental texture analysis of the fat-replaced biscuit samples further confirmed the observation of compactness in structure, which may have resulted in the increased hardness of biscuits (Table 7) (Saeed et al., 2021). Moreover, the protein matrix suffered degradation due to the dual treatment of gelatinization and fermentation (De Pasquale et al., 2020). Additionally, increased IVPD of biscuit samples correlated with the breakdown of the protein matrix (Table 8). These observations are consistent with the findings of Saeed et al. (2020a) for fat-replaced biscuits produced from legume flour. The literature indicated that despite the fact that microstructural features of fat replacers are substantially different from fat when they are used in food items. However, the end products exhibit similar attributes to the conventional formulation, which was also verified by our results (Saeed et al., 2020a).
3.13 Sensory characteristics of fat-replaced biscuits
Table 9 shows the results of the sensory assessment in terms of appearance, taste, color, texture, and overall acceptability of biscuit samples prepared by replacing varying amounts of fat content with FGUBF. Higher sensory scores for color, texture, and overall acceptability were produced by replacing fat at the ostensibly optimal level of 20% FGUBF, which were significantly (P < 0.05) greater than that of the control biscuits. However, the sensory panellists were not able to differentiate any significant (P > 0.05) difference in appearance and taste between 20% FGUBF and full fat-containing biscuits. Furthermore, color and overall acceptability scores of 10% FGUBF biscuits were significantly (P > 0.05) different from control biscuits.
| Samples | Appearance (9*) | Color (9*) | Taste (9*) | Texture (9*) | Overall Acceptability (9*) |
|---|---|---|---|---|---|
| Control | 8.35 ± 0.15b | 7.22 ± 0.17a | 8.35 ± 0.13d | 8.23 ± 0.15b | 8.07 ± 0.17b |
| 10% FGUBF | 8.33 ± 0.12b | 8.43 ± 0.15c | 8.32 ± 0.15b | 8.22 ± 0. 13b | 8.33 ± 0.16c |
| 20% FGUBF | 8.36 ± 0.17bc | 8.62 ± 0.15d | 8.34 ± 0.12d | 8.38 ± 0. 17c | 8.42 ± 0.19d |
| 30% FGUBF | 7.52 ± 0.13a | 7.89 ± 0.14b | 7.25 ± 0.10a | 7.73 ± 0. 12a | 7.13 ± 0.14a |
Each result is shown as the mean ± standard deviation (n = 3); the significant difference (P ≤ 0.05) between the means in each separate column is denoted by different letters. The significance level is calculated by Duncan method. Where control represents biscuits without fat replacement, FGUBF; fermented and gelatinized Urad bean flour, *9 Points hedonic scale.
The taste of biscuit samples decreased when the level of fat replacement increased beyond 20% FGUBF due to the undesirable mouthfeel and increased hardness. This increased hardness at a high concentration of fat replacement was also confirmed by the findings of textural analyzer (Table 5). These negative consequences were apparently caused by a substantial reduction in the amount of saturated fat in the biscuit recipe (Patel et al., 2020). Generally, fat is considered to aid in soothing and softening the texture of many different types of baked goods, thereby giving them the textural attributes that consumers demand (Yazar and Rosell, 2022). Saeed et al. (2020a) depicted that up to 15% of fat replacement by black gram flour had no negative impact on the sensory profile of biscuits. In contrast, in the present study, up to 20% of fat replacement by gelatinized and fermented legume flour produced satisfactory results for the sensory parameters of biscuit samples.
3.14 Correlation analysis
The correlation plot matrix shows the coefficient values of Pearson correlation (Fig. 2a) and their degree of significance (Fig. 2b) assessed between the DPPH-IC50, FRAP-IC50, ABTS-IC50, HRSA-IC50, α-glucosidase inhibition-IC50, TPC and TFC of flour blends and biscuit samples. The positive and negative correlations are shown in red and purple colors, respectively. The correlation coefficients were inversely proportional to ellipse size. For example, the smaller the size of ellipse, the more will be the positive correlation between the samples. The non-significant difference (P > 0.05) was excluded from the correlogram (Fig. 2b). Strong and positive correlations existed between the different antioxidant activities of flour blends and their biscuit samples. In fact, significant and positive correlations were anticipated because these antioxidant activities work on the same principle, that is, the donation of electron (Dewanto et al., 2002). The negative linear correlation of the IC50 values of DPPH, FRAP, ABTS, HRSA, and α-glucosidase inhibition with the bioactive compounds (TPC and TFC) were observed, which demonstrated the positive contribution of phenolic compounds to the antioxidant potential of fermented legumes (Girish et al., 2012). The lower IC50 values of DPPH, FRAP, ABTS, and HRSA are associated with increased antioxidant activities. These findings suggested that the FGUBF has strong antioxidant potential since the reduction in IC50 values of antioxidant activities may have resulted in the increment of TPC and TFC. Furthermore, IC50 values of α-glucosidase inhibition of flour blends and biscuit samples were also negatively correlated to TPC and TFC, i.e., inhibition of α-glucosidase increased with the increased concentration of these bioactive compounds and antioxidants.
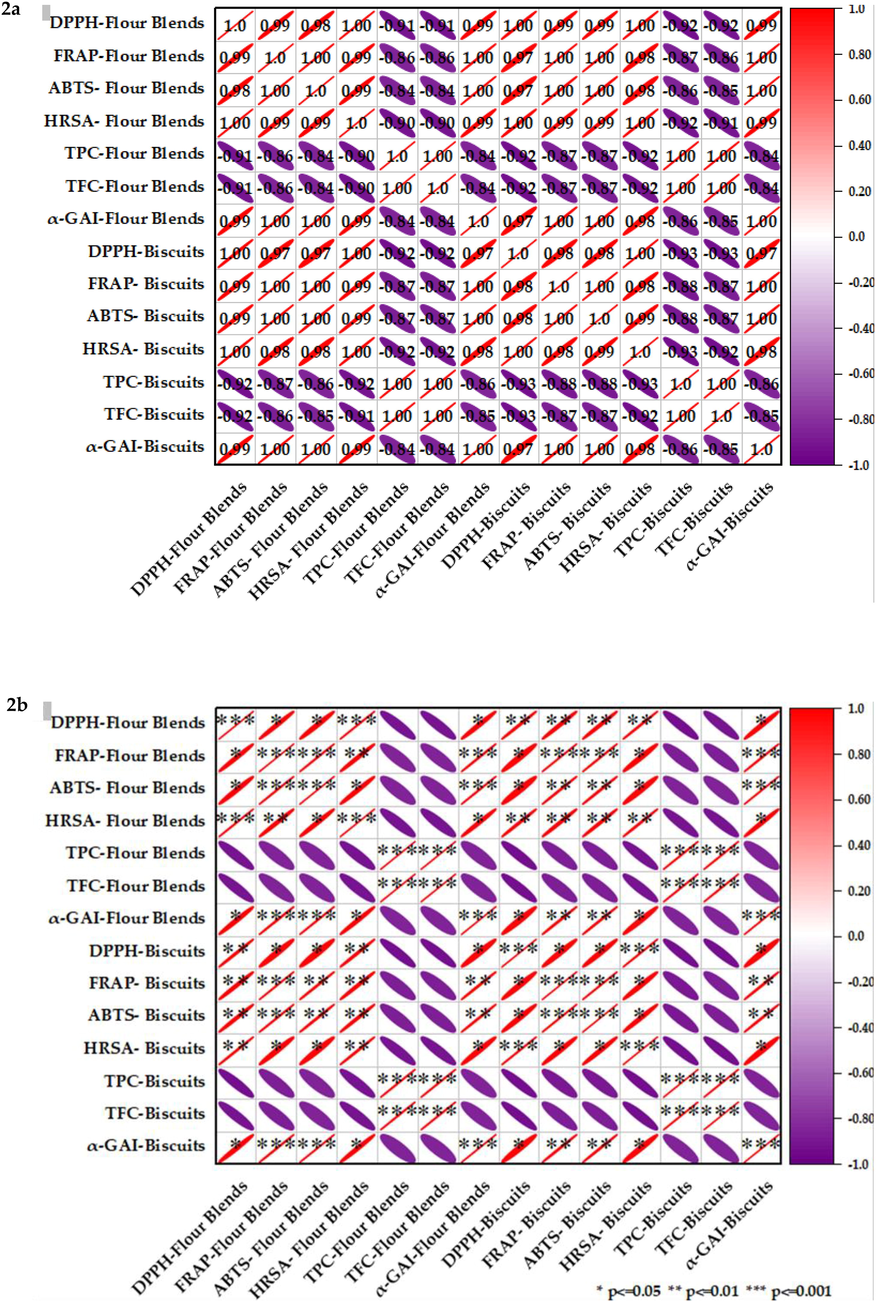
- (a) Correlogram analysis with the determined values of Pearson Coefficient of Correlation between the bioactive compounds (TPC and TFC), IC50 values of antioxidant activities (DPPH, ABTS, HRSA, and FRAP), and IC50 values of α-Glucosidase Inhibition (α-GAI) of flour blends and biscuits. (b) Correlogram with the defined significance levels. Where: TPC; Total phenol content, TFC; Total flavonoid content, DPPH; 2, 2-diphenyl-1-picrylhydrazyl, ABTS; 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid), HRSA; Hydroxyl radical scavenging activity, FRAP; Ferric reducing antioxidant power.
3.15 Principal component analysis
The relationship between the physicochemical properties, functional properties, bioactive compounds, anti-nutrients, pasting properties, and rheological characteristics of WF (control) and FGUBF-WF blends were investigated by using principle component analysis (PCA) (Fig. 3). The PC1 and PC2 accounted 9821% of the cumulative variation among the 24 attributes of the 4 flour samples (Fig. 3). PC1 and PC2 explained 89.23% and 8.98% of the data variation, respectively. 10% FGUBF and 20% FGUBF looked closer to one another in the observation plot, which suggested a positive correlation between them (Fig. 3a). In addition, 30% FGUBF also seemed to be closer to FGUBF 20%. However, control and 30% FGUBF emerged opposite to one another and displayed a negative association. The bi-plot of PC1 versus PC2 demonstrated that maximum attributes were negatively correlated with PC1 and positively correlated with PC2 (Fig. 3b). Furthermore, carbohydrate %, gluten index %, DST, FV, SBV, and BDV were positively correlated to PC1. While physicochemical attributes (protein%, fat% crude fibre%, ash%, and moisture %), bioactive compounds (TPC and TFC), functional properties (WAC% and OAC %), pasting characteristics (BDV and SBV), rheological properties (WA, DST, DDT, DoS, and FQN) and anti-nutrients (PA and TS) were positively correlated with PC2. Besides, negative mutual correlations of PC1 and PC2 were observed for DDT, GT, PV, PT, and glucosidase inhibition which predicted the negative variations of these attributes among the samples.
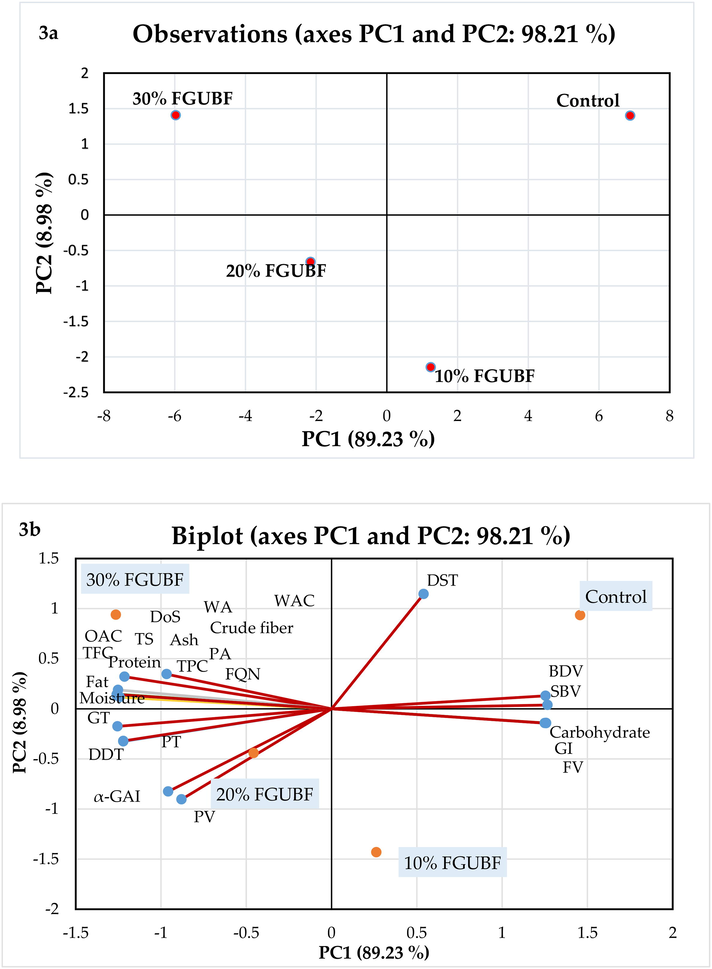
- (a) Principal component analysis with the location of Wheat flour (control), fermented and gelatinized Urad bean (FGUBG), FGUBF 10%, FGUBF 20%, and FGUBF 30%). (b) Biplot of flour blends with the dispersion of characteristics (b6y) in the area delineated by principal component 1 (PC1) and principal component 2 (PC2). Where, WAC; water absorption capacity, OAC; oil absorption capacity, PV; peak viscosity, BD; break down viscosity, SB; setback viscosity, FV; final viscosity, PT; pasting temperature, GT; gelatinization time, DDT; dough development time, WA; water absorption, DS; dough stability, DoS; degree of softening, FQN; farinograph quality number, PA; phytic acid, TS; total saponin, GAI; glucosidase inhibition, TPC; total phenolic content, TFC; total flavonoid content.
Using PCA, the average values of the physical properties, anti-nutrients, bioactive compounds, α-glucosidase inhibition, In vitro protein digestibility, and nutritional profile of fat- replaced biscuits were determined, and the results are illustrated in Fig. 4(a and b). The differences and similarities among the 20 attributes of the 4 biscuit samples were described by PC1 and PC2, which together accounted for 98.44% of the total variations. It was evaluated that the eigenvalue of PC1 and PC2 was 15.54 and 4.14, respectively, which depicted a variability of 77.70% and 20.74%, respectively. 10% FGUBF, 20% FGUBF, and 30% FGUBF were found closer to each other in the observation plot (Fig. 4a), which showed similar physical (diameter, spread ratio breaking force, L*, a*, and b* values), bioactive compounds (TPC and TFC), anti-nutrients (phytic acid and saponin), α-glucosidase inhibition%, and nutritional characteristics (ash, protein, fiber, fat, and energy) of these fat-replaced biscuit samples (Fig. 4b).
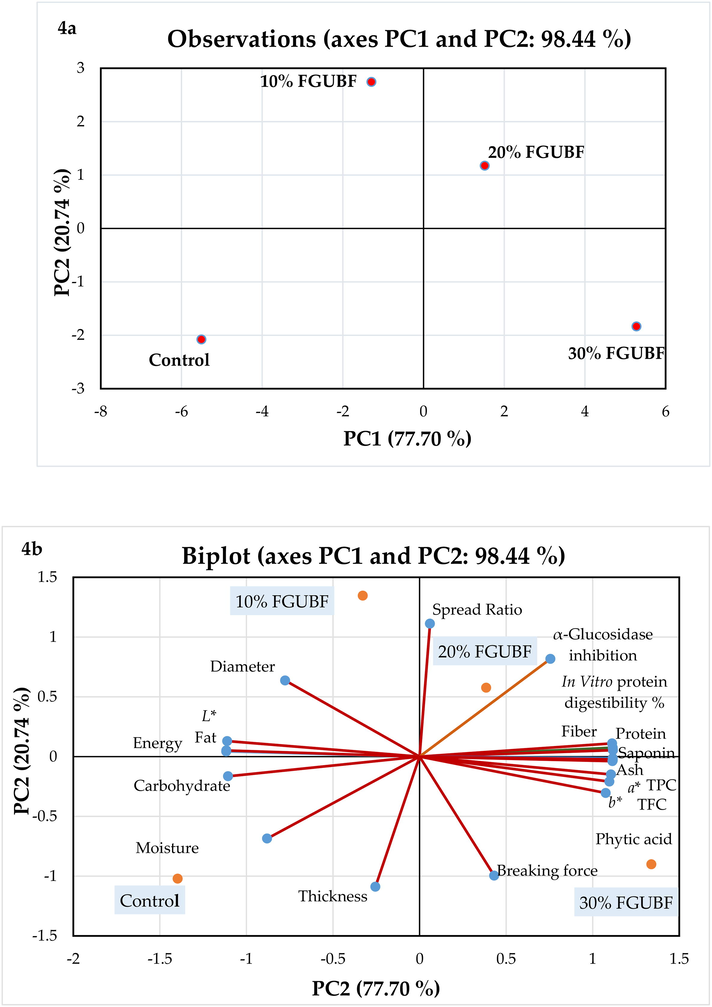
- (a) Principal component analysis with the location of control and fat-replaced biscuit samples(fermented and gelatinized Urad bean (FGUBG), FGUBF10%, FGUBF 20%, and FGUBF 30%). (b) Biplot of fat-replaced biscuit samples illustrating the association between variables (anti-nutrients, bioactive substances, α-Glucosidase inhibition, dimensions, textural, color values, and nutritional properties) in the area delineated by principal component 1 (PC1) and principal component 2 (PC2).
Fig. 5(a and b) shows the average values of sensory properties of biscuit samples determined by PCA. The PC1 and PC2 collectively demonstrated 99.46% of the total variation among the 22 biscuit samples. The eigenvalue of PC1 was >1 and explained 80.15% of the variation. The observation plot in Fig. 5a shows that the control, 10% FGUBF, and 20% FGUBF were close to each other, indicating that these biscuit samples have similar edible characteristics.
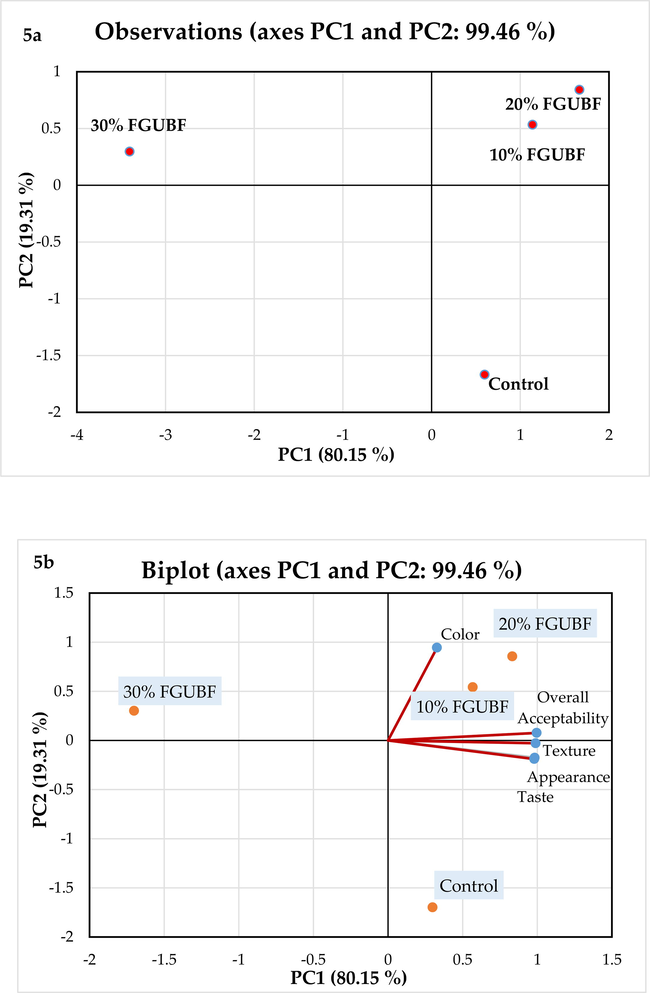
- (a) Principal component analysis and its score plot, illustrating the variability between the sensory parameters of biscuit samples: control and fat-replaced biscuit samples fermented and gelatinized Urad bean (FGUBG), FGUBF10%, FGUBF 20%, and FGUBF 30%). (b) Biplot of fat-replaced biscuit samples describing the relationship within the sensory parameters.
Furthermore, the bi-plot of sensory variables (i.e., taste, texture, color, appearance, and overall acceptability) indicated a favourable association (i.e., positive correlation) between them (Fig. 5b). From the bi-plot, it can be clearly visualized that the 30% FGUBF displayed considerably different traits than the other biscuit samples. This explains the fact that 30% FGUBF had a negative correlation with other biscuit samples and demonstrated a strong influence on the sensory properties of biscuits. PCA and ANOVA depicted similar observations for sensory analysis of fat-replaced biscuit samples (Table 5).
4 Conclusion
In this research, the nutritional profile and technological properties of Urad bean as fat replacer and its resulting biscuits were improved by the combined effects of gelatinization and fermentation with S. cerevisiae MK-157. The thermal treatment of gelatinization activated endogenous enzymes in Urad bean which was further enhanced by the microbial activities during fermentation. These microbial activities significantly (P < 0.05) improved the nutritional characteristics of legumes by degrading anti-nutritional compounds, i.e., phytic acid and saponin content. Although, GUBF reduced the antioxidant potential assessed by DPPH, ABTS, HRSA, and FRAP. However, fermentation of GUBF significantly (P < 0.05) increased the concentrations of antioxidants and bioactive compounds (TPC and TFC) in FGUBF-WF blends and their biscuit samples. FGUBF showed a positive impact on the functional (WAC and OAC) and technological (pasting, rheological, textural) characteristics of flour blends, biscuit dough, and biscuit samples.
Gelatinization and fermentation of legume disintegrated the structure of proteins and polymeric chains of starch molecules, which was reflected in the micrographs of biscuit samples. Furthermore, the incorporation of flour blends in biscuit samples as a fat replacer increased the contents of protein, ash, and fiber, while calories were reduced with respect to the levels of fat replacement. In vitro protein digestibility of biscuit samples was also increased. 20% FGUBF depicted the desirable physical and sensory properties of biscuit samples. Increasing the concentration of FGUBF beyound 20% resulted in the development of hard texture and bitter mouth feel. In combination with gelatinization, fermentation produced encouraging results, making this technique an ideal option for implementation as a new technology in the food sector to develop legume flours with improved nutritional profiles. Simultaneously, this study opens the doors for the legume processing industries by providing value to the waste of legumes. However, the fermention of gelatenized Urad bean flour has been done on a laboratory scale. Therefore, implimentation of this technique on pilot plant or industrial scale for biscuits prepartion may provide certain variations in the final product which needs to be analyzed practically. Conclusively, future investigations are required to validate the reported findings in the In-vivo models.
Author contributions
Conceptualization Syed Arsalan Ali Syed Muhammad Saeed and Angelo Giuffrè; Data curation Syed Arsalan Ali and Muhammad Sohail; Formal analysis Syed Arsalan Ali; Funding acquisition Manal Elkhadragy and Hany Yehia; Investigation Syed Arsalan Ali; Methodology Syed Arsalan Ali Syed Muhammad Saeed Muhammad Sohail and Angelo Giuffrè; Project administration Syed Arsalan Ali; Resources Syed Muhammad Saeed Manal Elkhadragy and Hany Yehia; Software Syed Arsalan Ali and Angelo Giuffrè; Supervision Syed Muhammad Saeed and Angelo Giuffrè; Validation Syed Arsalan Ali and Syed Muhammad Saeed; Visualization Syed Muhammad Saeed; Writing – 52 original draft Syed Arsalan Ali; Writing – review & editing Syed Arsalan Ali Syed Muhammad Saeed Muhammad Sohail Manal Elkhadragy Hany Yehia and Angelo Giuffrè.
Funding
This research was funded by the Princess Nourah bint Abdulrahman University P.O. Box 84428, Riyadh 11671, Saudi Arabia, Researchers Supporting Project number (PNURSP2022R23).
Acknowledgement
The authors would like to thank all colleagues at the Princess Nourah bint Abdulrahman University , P.O. Box 84428, Riyadh 11671, Saudi Arabia Researchers Supporting Project number (PNURSP2022R23). Authors are also thankfull to English Biscuits Manufacturer (PVT .Ltd) Karachi, Pakistan for providing the testing facilities.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- AACC Committee, 2000. Approved methods of the American association of cereal chemists, vol. 1. Amer. Assn. of Cereal Chemists. https://doi.org/10.1016/s0144-8617(01)00358-7.
- Fermentation of cereals and legumes: Impact on nutritional constituents and nutrient bioavailability. Fermentation. 2022;8(2):63.
- [CrossRef] [Google Scholar]
- Functional and thermal properties of wheat, barley, and soy flours and their blends treated with a microbial transglutaminase. J. Food Sci.. 2005;70(6):c380-c386.
- [CrossRef] [Google Scholar]
- A novel approach to improve the nutritional value of black gram (Vigna mungo L.) by the combined effect of pre-gelatinization and fermentation by Lactobacillus sp. E14 and Saccharomyces cerevisiae MK-157: Impact on morphological, thermal, and chemical structural properties. LWT-Food Sci. Technol.. 2022;172:114216
- [CrossRef] [Google Scholar]
- In vitro digestibility of commercial whey protein supplements. LWT-Food Sci. Technol.. 2015;61(1):7-11.
- [CrossRef] [Google Scholar]
- Impact of germination alone or in combination with solid-state fermentation on the physicochemical, antioxidant, in vitro digestibility, functional and thermal properties of brown finger millet flours. LWT-Food Sci. Technol. 2022;154:112734
- [CrossRef] [Google Scholar]
- Proximate, baking, and sensory qualities of biscuits from wheat and fermented breadfruit (Artocarpus communis Frost) flour. Culin. Sci. Technol.. 2014;12(4):316-325.
- [CrossRef] [Google Scholar]
- Flavonoids as human intestinal α-glucosidase inhibitors. Foods. 2021;10(8):1939.
- [CrossRef] [Google Scholar]
- Weissella cibaria short-fermented liquid sourdoughs based on quinoa or amaranth flours as fat replacer in focaccia bread formulation. Int. J. Food Sci.. 2021;56(7):3197-3208.
- [CrossRef] [Google Scholar]
- Overweight and obesity epidemic in developing countries: a problem with diet, physical activity, or socioeconomic status? Sci. World J.. 2014;2014
- [CrossRef] [Google Scholar]
- Pulse proteins: Processing, characterization, functional properties and applications in food and feed. Int. Food Res. J.. 2010;43(10):414-431.
- [CrossRef] [Google Scholar]
- Profile of volatile compounds of on-farm fermented and dried cocoa beans inoculated with Saccharomyces cerevisiae KY794742 and Pichia kudriavzevii KY794725. Molecules. 2021;26(2):344.
- [CrossRef] [Google Scholar]
- Modification of physicochemical, functional properties, and digestibility of macronutrients in common bean (Phaseolus vulgaris L.) flours by different thermally treated whole seeds. Food Chem.. 2022;382:132570
- [CrossRef] [Google Scholar]
- Fermented rice-bran by Saccharomyces cerevisiae: Nutritious ingredient in the formulation of gluten-free cookies. Food Biosci.. 2021;40:100859
- [CrossRef] [Google Scholar]
- Reevaluating the role of 1, 10-phenanthroline in oxidative reactions involving ferrous ions and DNA damage. Biochim. Biophys. Acta. Gen. Subj.. 2004;1675(1–3):46-53.
- [CrossRef] [Google Scholar]
- Nutritional and functional effects of the lactic acid bacteria fermentation on gelatinized legume flours. Int. J. Food Microbiol.. 2020;316:108426
- [CrossRef] [Google Scholar]
- Nutritional and functional advantages of the use of fermented black chickpea flour for semolina-pasta fortification. Foods. 2021;10(1):182.
- [CrossRef] [Google Scholar]
- Processed sweet corn has higher antioxidant activity. J. Agric. Food Chem.. 2002;50(17):4959-4964.
- [CrossRef] [Google Scholar]
- Pleurotus ostreatus: A potential concurrent biotransformation agent/ingredient on development of functional foods (cookies) LWT-Food Sci. Technol.. 2021;148:111727
- [CrossRef] [Google Scholar]
- Chemical constituents with free-radical-scavenging activities from the stem of Fissistigma polyanthum. Pharmacogn. Mag.. 2012;8(30):98.
- [CrossRef] [Google Scholar]
- Markers of antioxidant defense in patients with type 2 diabetes. Oxid. Med. Cell. Longev.. 2016;2016
- [CrossRef] [Google Scholar]
- Nutrient distribution, phenolic acid composition, antioxidant and alpha-glucosidase inhibitory potentials of black gram (Vigna mungo L.) and its milled by-products. Int. Food Res. J.. 2012;46(1):370-377.
- [CrossRef] [Google Scholar]
- Effects of shortening replacement with extra virgin olive oil on the physical–chemical–sensory properties of Italian Cantuccini biscuits. Foods. 2022;11(3):299.
- [CrossRef] [Google Scholar]
- Sensitive method for the rapid determination of phytate in cereals and cereal products. J. Sci. Food Agric.. 1983;34(12):1423-1426.
- [CrossRef] [Google Scholar]
- Heymann, H., Lawless, H.T., 2013. Sensory evaluation of food: principles and practices. Springer Sci. Business Media. https://doi.org/10.1007/978-1-4419-6488-5.
- Legumes—A Comprehensive Exploration of Global Food-Based Dietary Guidelines and Consumption. Nutrients. 2022;14(15):3080.
- [CrossRef] [Google Scholar]
- Effect of fermentation on some functional properties of Mucuna sloanei and Detarium microcarpum. J. Food Technol.. 2012;10(13):83-86.
- [CrossRef] [Google Scholar]
- Effect of fermentation on the proximate composition and functional properties of mahogany bean (Afzelia africana) flour. Curr. Res. Nutr. Food Sci.. 2014;2(1):01-07.
- [CrossRef] [Google Scholar]
- Preparation, characterization and functional properties of flax seed protein isolate. Food Chem.. 2016;197:212-220.
- [CrossRef] [Google Scholar]
- Inhibitory effect of pine extract on α-glucosidase activity and postprandial hyperglycemia. Nutrition. 2005;21(6):756-761.
- [CrossRef] [Google Scholar]
- Effect of Fermented Lettuce Extract on Starch Retrogradation in Korean Rice Cake. Garaetteok. KSBB J.. 2021;36(3):198-203.
- [CrossRef] [Google Scholar]
- Preparation of antioxidant and evaluation of the antioxidant activities of antioxidants extracted from sugarcane products. J. Food Nutr. Res.. 2015;3(7):458-463.
- [CrossRef] [Google Scholar]
- Formulation of biscuits fortified with a flour obtained from bergamot by-products (Citrus bergamia, Risso) Foods. 2022;11(8):1137.
- [CrossRef] [Google Scholar]
- Effect of lactic fermentation on the total phenolic, saponin and phytic acid contents as well as anti-colon cancer cell proliferation activity of soymilk. J. Biosci. Bioeng.. 2013;115(5):552-556.
- [CrossRef] [Google Scholar]
- Nutritional and Food Composition Survey of Major Pulses Toward Healthy, Sustainable, and Biofortified Diets. Front. Sustain. Food Syst.. 2022;6
- [CrossRef] [Google Scholar]
- Incorporation of biovalorised okara in biscuits: Improvements of nutritional, antioxidant, physical, and sensory properties. LWT-Food Sci. Technol.. 2020;134:109902
- [Google Scholar]
- Effect of solid-state fermentation with Bacillus subtilis lwo on the proteolysis and the antioxidative properties of chickpeas. Int. J. Food Microbiol.. 2021;338:108988
- [CrossRef] [Google Scholar]
- Exopolysaccharide producing lactic acid bacteria: Their techno-functional role and potential application in gluten-free bread products. Int. Food Res. J.. 2018;110:52-61.
- [CrossRef] [Google Scholar]
- An update on water kefir: Microbiology, composition and production. Int. J. Food Microbiol.. 2021;345:109128
- [CrossRef] [Google Scholar]
- Advances in our understanding of the structure and functionality of edible fats and fat mimetics. Soft Matter.. 2020;16(2):289-306.
- [CrossRef] [Google Scholar]
- Probiotic potential, antioxidant activity, and phytase production of indigenous yeasts isolated from indigenous fermented foods. Probiotics Antimicrob. Proteins. 2020;12(1):280-288.
- [CrossRef] [Google Scholar]
- Rheological analysis of wheat flour dough as influenced by grape peels of different particle sizes and addition levels. Food Bioproc. Tech.. 2019;12(2):228-245.
- [CrossRef] [Google Scholar]
- Solid state fermentation of mango peel and mango seed waste by different yeasts and bacteria for nutritional improvement. Int. J. Food Ferment. Technol.. 2017;7(1):111-118.
- [CrossRef] [Google Scholar]
- Effect of processing on the in vitro and in vivo protein quality of red and green lentils (Lens culinaris) Food Chem.. 2018;240:588-593.
- [CrossRef] [Google Scholar]
- Applications of fat mimetics for the replacement of saturated and hydrogenated fat in food products. Curr. Opin. Food Sci.. 2020;33:61-68.
- [CrossRef] [Google Scholar]
- Carbohydrates as fat replacers. Annu. Rev. Food Sci. Technol.. 2017;8(1):331-351.
- [CrossRef] [Google Scholar]
- Co-culturing corncob-immobilized yeasts on orange peels for the production of pectinase. Biotechnol. Lett.. 2020;42(9):1743-1753.
- [CrossRef] [Google Scholar]
- Sensory evaluation and storage stability of fat reduced shortdough biscuit using hydrolysable tannin encapsulated double emulsion as fat replacer. LWT. 2022;154:112816
- [CrossRef] [Google Scholar]
- Lentil flour: Nutritional and technological properties, in vitro digestibility and perspectives for use in the food industry. Curr. Opin. Food. 2021;40:157-167.
- [CrossRef] [Google Scholar]
- Fermented Hass avocado kernel: Nutritional properties and use in the manufacture of biscuits. Saudi J. Biol. Sci.. 2022;29(6):103295
- [CrossRef] [Google Scholar]
- Exploring the potential of black gram (Vigna mungo) flour as a fat replacer in biscuits with improved physicochemical, microstructure, phytochemicals, nutritional and sensory attributes. SN Appl. Sci.. 2020;2(12):1-17.
- [CrossRef] [Google Scholar]
- b. Evaluation of the potential of Lotus root (Nelumbo nucifera) flour as a fat mimetic in biscuits with improved functional and nutritional properties. CyTA-J. Food.. 2020;18(1):624-634.
- [CrossRef] [Google Scholar]
- Impact of the incorporation of date pit flour an underutilized biowaste in dough and its functional role as a fat replacer in biscuits. J. Food Process. Preserv.. 2021;45(3):e15218.
- [Google Scholar]
- Phenolic content, antioxidant potential and DNA damage protection of pearl millet (Pennisetum glaucum) cultivars of North Indian region. J. Food Meas. Charact.. 2017;11(1):126-133.
- [CrossRef] [Google Scholar]
- Quinoa sourdough-based biscuits with high antioxidant activity fermented with autochthonous lactic acid bacteria. J. Appl. Microbiol.. 2022;132(3):2093-2105.
- [CrossRef] [Google Scholar]
- Citrus limetta peels: a promising substrate for the production of multienzyme preparation from a yeast consortium. Bioresour. Bioprocess.. 2019;6(1):1-15.
- [CrossRef] [Google Scholar]
- Anti-nutrient & bioactive profile, in vitro nutrient digestibility, techno-functionality, molecular and structural interactions of foxtail millet (Setaria italica L.) as influenced by biological processing techniques. Food Chem.. 2022;368:130815
- [CrossRef] [Google Scholar]
- Sensory characteristics and nutritional quality of food products made with a biofortified and lectin free common bean (Phaseolus vulgaris L.) flour. Nutrients. 2021;13(12):4517.
- [CrossRef] [Google Scholar]
- Effect of roasting pulse seeds at different tempering moisture on the flour functional properties and nutritional quality. Int. Food Res. J.. 2021;147:110489
- [CrossRef] [Google Scholar]
- Probiotic properties of yeasts in traditional fermented foods and beverages. J. Appl. Microbiol.. 2022;132(5):3533-3542.
- [CrossRef] [Google Scholar]
- Enhancing the utilization of rapeseed protein ingredients in bread making by tailored lactic acid fermentation. Food Biosci. 2022:102028.
- [CrossRef] [Google Scholar]
- WHO Organization, 2018. A healthy diet sustainably produced: information sheet. World Health Organization. https://www.who.int/publications/i/item/WHO-NMH-NHD-18.12.
- Effect of germination on the chemical composition, thermal, pasting, and moisture sorption properties of flours from chickpea, lentil, and yellow pea. Food Chem.. 2019;295:579-587.
- [CrossRef] [Google Scholar]
- Effects of chia flour as a fat substitute on the physicochemical, nutritional and sensory properties of biscuits. Int. J. Food Sci. Tech. 2023
- [CrossRef] [Google Scholar]
- Fat replacers in baked products: their impact on rheological properties and final product quality. Crit. Rev. Food Sci. Nutr. 2022:1-24.
- [CrossRef] [Google Scholar]
- review of current and future food applications of natural hydrocolloids. Int. J. Food Sci.. 2020;55(4):1389-1406.
- [CrossRef] [Google Scholar]







