Translate this page into:
Genus Morinda: An insight to its ethnopharmacology, phytochemistry, pharmacology and Industrial Applications
⁎Corresponding authors at: Industrial Chemistry Unit, Department of Physical Sciences, Landmark University, PMB 1001, Omu-Aran, Nigeria. oladeji.oluwole@lmu.edu.ng (Oluwole Solomon Oladeji)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Background of the study
The genus Morinda of the Madder family, (Rubiaceae) has been widely documented in traditional medicine due to its therapeutic properties and also, contributed a great deal in chemical industry. Different parts of Morinda species have traditionally been used to treat malaria, diabetes, memory loss, cancer, inflammation, skin infections, and typhoid fever.
Aim and Objectives
The review provide a critical and innovative information on the traditional uses, phytochemical constituents, and industrial applications of the genus Morinda. This will help researchers understand future research trends by bridging the gap between documented literature and contemporary uses.
Methodology
All the systematic literature data or information on the genus Morinda was collected via selected electronic databases, including Scopus, PubMed, Web of Science, Springer, Medline, ChemSpider, Taylor and Francis, Google Scholar, SciFinder, ScienceDirect and Wiley. Relevant book chapters, Wikipedia and books were also explored.
Results
The study reveals that different parts of Morinda plants have been extensively used for folkloric therapeutic purposes and are a plethora of mineral or nutritional benefits and secondary metabolites. Several classes of bioactive compounds have been elucidated from Morinda plants via spectroscopic and chromatographic phytochemical analyses. Compounds such as terpenoids, glycosides, anthraquinones, polyphenols, steroids, saponins and reducing sugars are among the bioactive substances reported in the genus. Plant extracts, fractions and isolates of Morinda plants have exhibited pronounced antidiabetic, antioxidant, antiplasmodial, antidepressant, wound healing, anticancer, and anti-inflammatory effects (in vitro and in vivo). These pharmacological activities exhibited could be linked to the presence of secondary metabolites reported. The applications of this genus is well documented in textile, metallurgical, agrochemicals and food industries.
Conclusion
A number of reports on Morinda showed significant therapeutic effect against several diseases such as cancer, dermal infection, diabetes, inflammation, malaria, typhoid, cholera and memory loss, however, there were several drawback in previous reports including mechanism of action, drug dose, controls and toxicological data of extracts or isolates. In view of this, further studies should emphasis on targeting active secondary metabolites which responsible for the therapeutic activities, structural elucidation and toxicological assessment. Furthermore, industrial uses require authentication.
Keywords
Morinda
Phytochemistry
Pharmacology
Industrial applications
Traditional uses
1 Introduction
The genus Morinda (Rubiaceae), with about 102 species of trees, shrubs, and vines, is widely dispersed in tropical, sub-tropical and temperate regions (Ban et al., 2013). The folklore applications of Morinda citrifolia L., Morinda lucida, and Morinda officinalis have been reported globally (Koumaglo et al., 1992a,b; Wong et al., 2002). Almost all portions of the Morinda species showed therapeutic effectiveness for the treatment of malaria, diabetes, hepatitis, cancer, typhoid, and hypertension, according to ethnobotanical and ethnopharmacological results (Kamiya et al., 2005; Xiang et al. 2008). This genus is widely dispersed in China with approximately twelve species, namely, Morinda officinalis, Morinda shuhuaensis, Morinda parvifolia, Morinda umbellate, and Morinda citrifolia, etc. (Su et al., 2005). The genus Morinda contain assortment of secondary metabolites with diverse structural moieties such as iridoids, anthraquinones, quinones, terpenes, lignans, alkaloids and glycosides (Kang et al., 2017; Salomone et al., 2017; Chen et al., 2013; Potterat et al., 2007; Takashima et al., 2007; Zhang et al., 2018). The pronounced anti-inflammatory, antimicrobial, antiviral, anti-osteoporotic and antioxidant activities of this genus could be attributed to the presence of anthraquinones and glycosides (Akihisa et al., 2007; Locatelli, 2011; Locatelli et al., 2012; Zengin et al., 2015). Currently, many research groups are working on isolation of secondary metabolites or pharmacophores from the genus Morinda. These have led to the discovery of wide array of compounds with diverse biological properties. Thus, in this review, we present up to date information on the phytochemistry of the genus Morinda and also give a detailed information on its biological and industrial applications.
2 Methodology
In order to accomplish this goal, a relevant data on the genus Morinda was explored using scientific databases (Science Direct, Google Scholar, Web of science, Asian Science Citation Database, PubMed, Scifinder, Chinese Biomedical Literature Database, Scopus, Chinese Scientific Journal Database, Chinese Science Citation Database, books and other literature sources. These databases are explored and exhaustive list of published articles available on Morinda are procured or downloaded. The bibliographic research was accomplished via keywords such as “Morinda, traditional uses, phytochemistry, pharmacology, toxicology”.
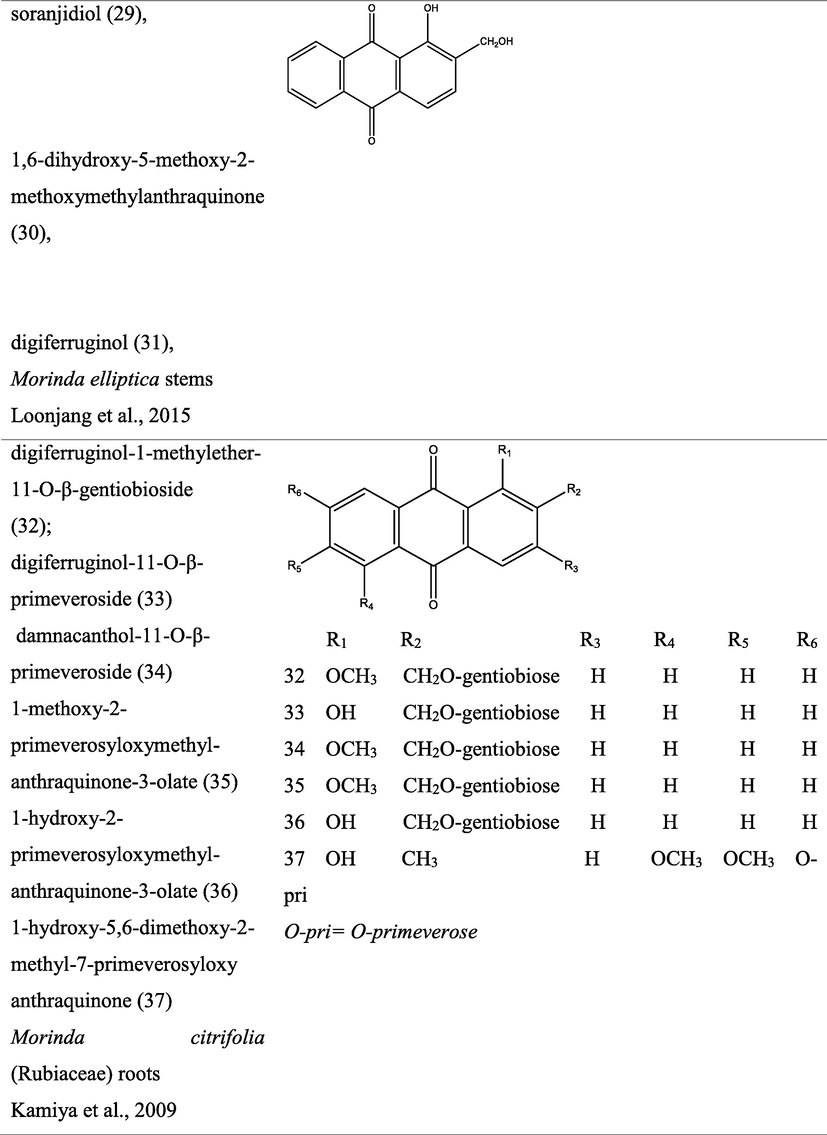
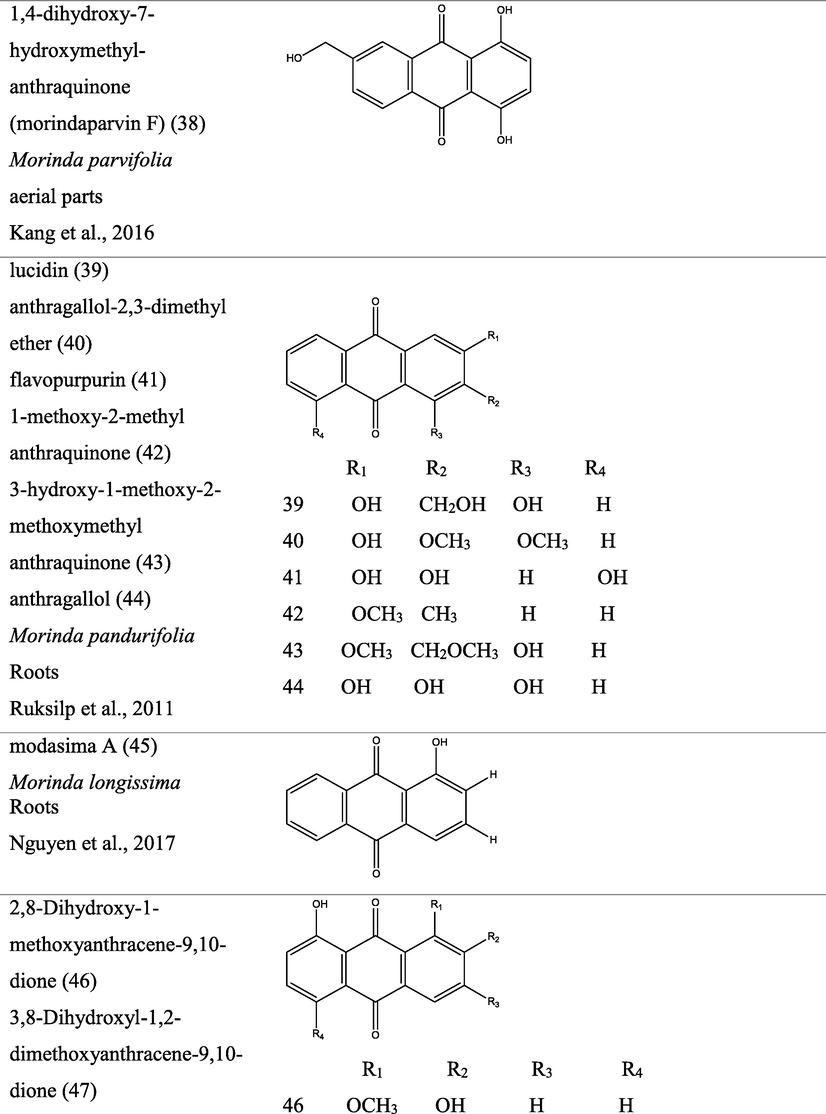
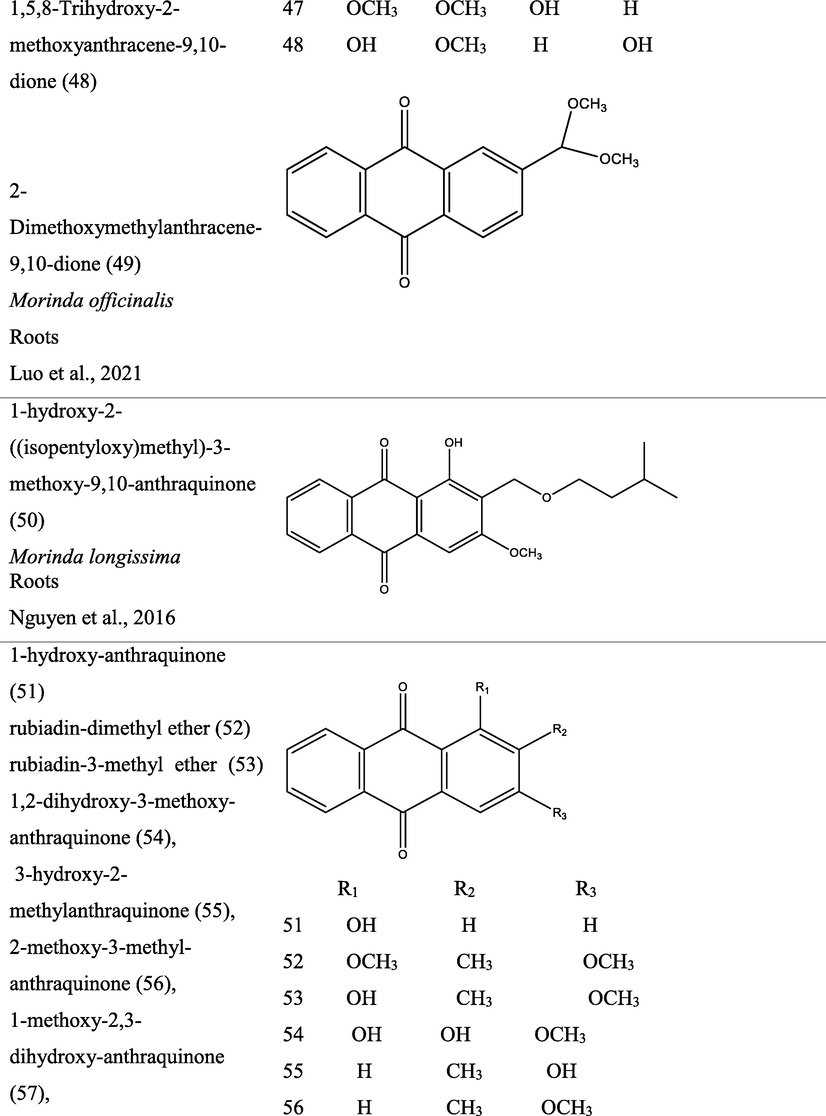
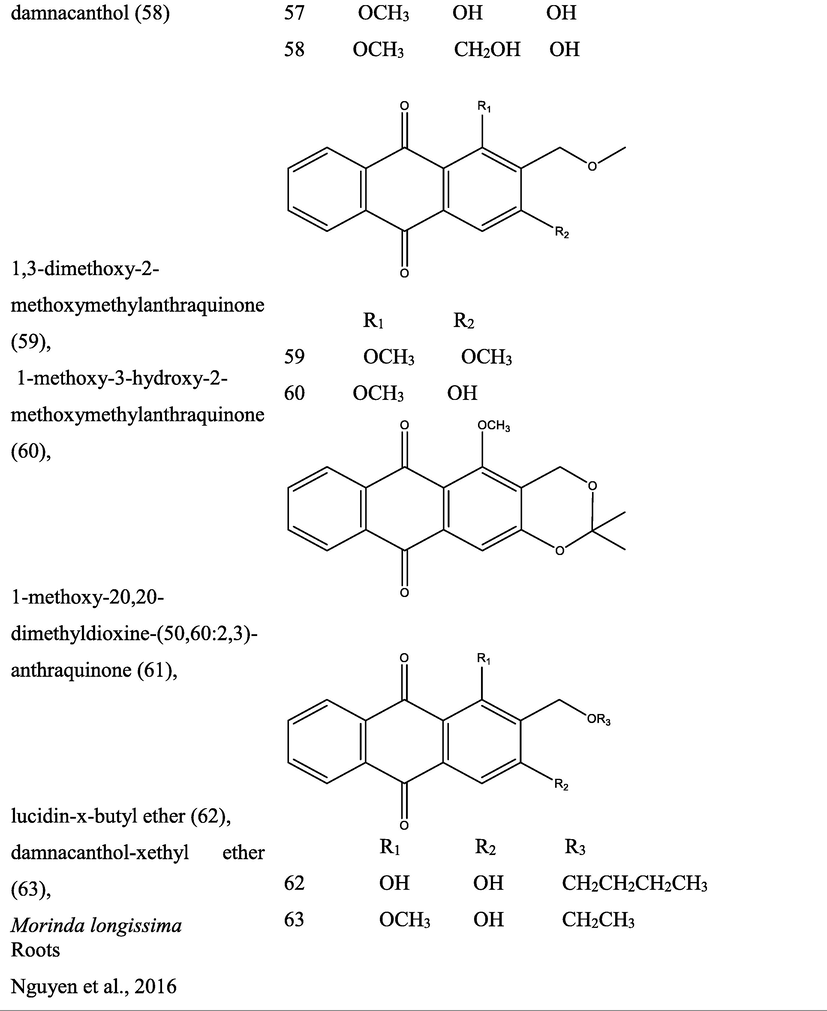
3 Botany
The genus Morinda consist of flowering plants in the madder family, Rubiaceae. The generic name originated from the Latin words morus “mulberry”, (appearance of the fruits), and indica, meaning “of India (Quattrocchi, 2000). It is extensively dispersed in tropical regions of the earth, with about 80 species of vines, shrubs or trees (Fig. 1). This genus produce aggregate or multiple fruits that can be fleshy or dry (Potterat and Hamburger, 2007; Sambamurty, 2005).
Pictures of different species of Morinda, {I- Morinda citrifolia; II- Morinda tinctoria; III- Morinda morindiodes; IV- Morinda elliptica; V- Morinda pubescens; VI- Morinda parvifolia; VII- Morinda officinalis; VIII- Morinda lucida}.
4 Ethnopharmacological uses of the genus Morinda
The scientific study of folkloric usage of medicinal plants by a particular group of people for the treatment of diseases or infections is known as ethnobotany. Traditionally, different parts of Morinda are extensively used in China, Thai, African, Tahitian, Hawaiian, Fiji, India and Ayurveda traditional medicine for the treatment of acute and chronic infections (Oladeji et al., 2022). In Taiwan, fruit juice of M. citrifolia is used as a cure for diabetes and hypertension (Lin et al., 2007). In tropical Pacific islands, fruit juice is used as antidote for headache, high blood pressure, diarrhea and diabetes (Siddiqui et al. 2008). In South-East Asia countries Brunei, Timor-Leste, Burma (Myanmar), Indonesia, Cambodia, Malaysia, the Philippines, Laos, Vietnam, Singapore and Thailand, powdered leaves of M. lucida is macerated in fresh palm wine which is widely used in treatment of high blood pressure, swollen spleen, diabetes, cough and malaria (Ee et al., 2009a,b). In Thai, wood and bark of M. coreia is generally used in alleviating fever and malaria (Chokchaisiri et al., 2019). In Liberia, dried stem-bark and leaves of M. morindoides macerated in water is used in treatment of worms, malaria and fever (Kiazolu et al., 2016). A broad ethnopharmacological uses of the genus Morinda is presented in Table 1.
Plant name
Traditional uses
References
Morinda citrifolia L.
In Asia, fruits, roots, barks, and leaves are used for the treatment of diabetes, arthritis and high blood pressure.
Wang et al., 2002a,b
In Malaysia, roots, barks, fruits, flowers and stems are widely used the treatment of diabetes, high blood pressure and many other illnesses
Bruggnecate,1992
The fruit juice is highly effective in preventing diabetes, hypertension and arteriosclerosis.
Kamiya et al., 2010a,b
In South-East Asia, the fruits are used to treat diabetes, swollen spleen, liver diseases and cough.
Ee et al., 2009a,b
In India, leaves, roots and seeds are curative agents for diarrhea, fevers, urinary tract infections, constipation, ulcers, nausea and asthma.
Swetal and Krishanamurthy, 2013
In Benin Republic, different parts of the plant is used in treatment of tuberculosis, cough and skin infections.
Sina et al., 2021
In Africa and Asia, the plant is used to treat blood pressure, anxiety, diabetes, colds, flu and depression.
Ellethy, 2019
Morinda coreia Buch.–Ham
Different parts of the plant is widely used in treatment of gastropathy, stomach ulcer, hernia, dyspepsia, fever, diarrhea and gout.
Cimanga et al., 2003
In north-eastern Thailand, the bark and wood are used as an antimalarial agent.
Kanchanapooma et al., 2002
Therapeutic agents in ameliorating arthritis, diarrhea, gout, diabetes, wounds, hypertension, gastropathy, dyspepsia and stomach ulcer.
Chokchaisiri et al., 2019; Pawlus et al., 2005
Morinda elliptica Ridl
In Thailand, different parts of the plant is used for the treatment of, cholera, diarrhea, headache, fever and hemorrhoids.
Loonjang et al., 2015a,b
In Malaysia, it is used in the treatment of cholera, headaches, diarrhoea, hemorrhoids and fever.
Ali et al., 1997
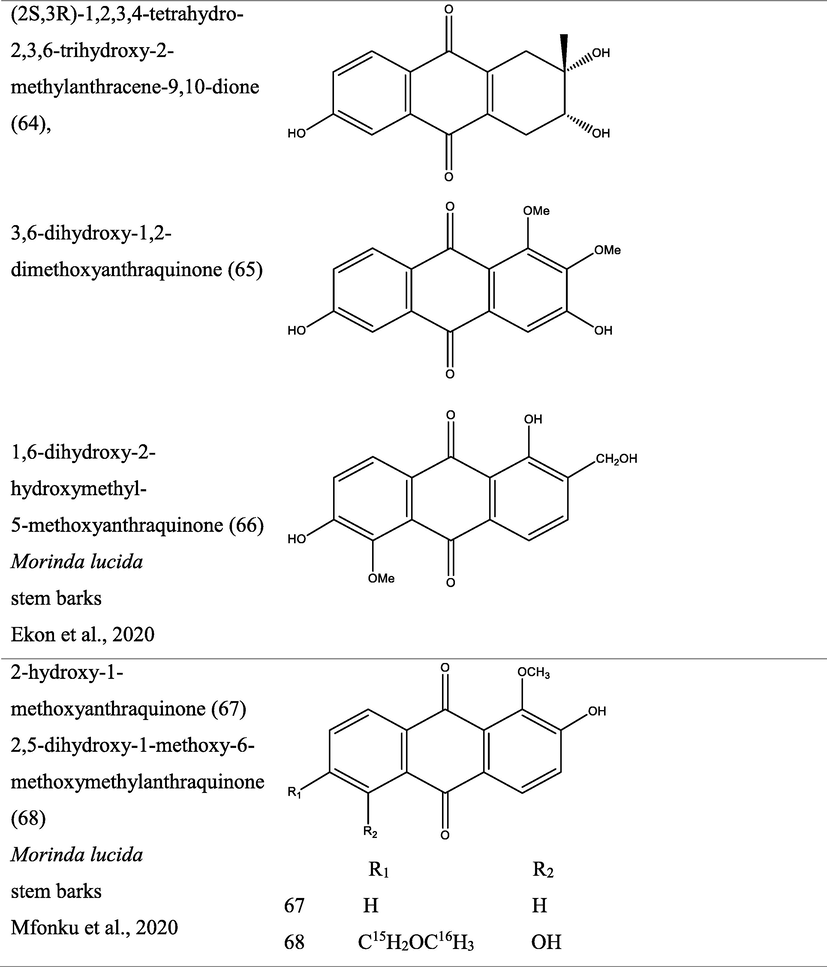
Morinda lucida Bentham
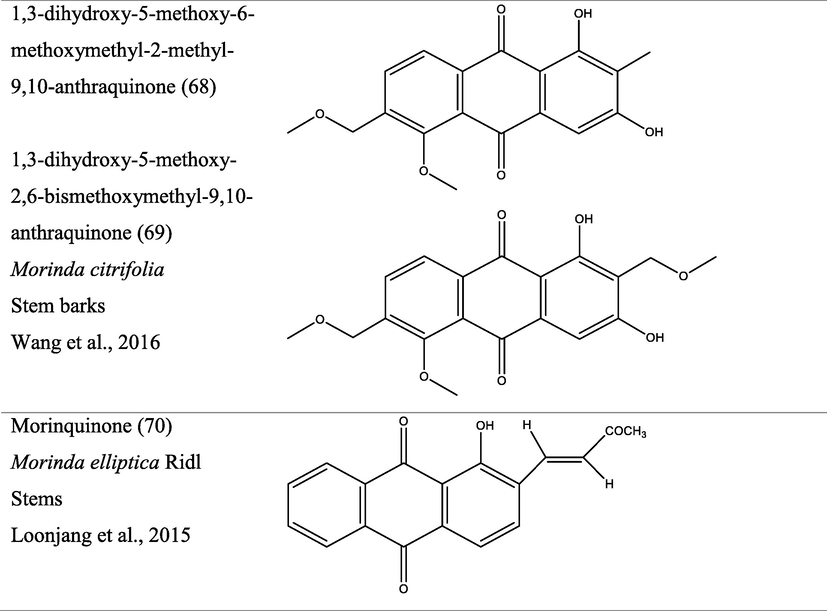
In Africa, stem, leaves and roots are commonly used in the treatment of malaria, yellow fever, trypanosomiasis and feverish conditions during child birth.
Chithambo et al., 2017
In West Africa, it is used in the treatment of fevers, hypertension, jaundice, gastric ulcer dysentery, cerebral congestion and diabetes.
Adeleye et al., 2018
In Nigeria, the leaves are squeezed or macerated in palm wine for the management of high blood pressure and malaria.
Osakwe et al., 1999
In Central and West Africa, leaves, roots and bark is used in treatment of malaria.
Idowu et al., 2010
Used as remedy for fever, dysentery, abdominal colic, and intestinal worm infestation.
Odutuga et al., 2010
In Nigeria and West Africa, the plant is used for the treatment of diabetes and malaria.
Elias et al., 2007Elufioye et al., 2013
Morinda morindoides (Baker) Milne Redhead
As malaria, worms, fever therapies. It is also used as a laxative
Kiazolu et al., 2016
In Africa and Asia, leaves decoction is used to treat diarrhea, amoebiasis or malaria.
Cimanga et al., 2007
In Democratic Republic of Congo, aqueous decoction of fresh leaves, is used for the treatment of diarrhoea and constipation.
Newinger, 2000
Morinda officinalis var. officinalis
Dried rhizomes is used for tonifying kidney, strengthening the bones and muscles, dispelling wind-dampness to alleviate impotence, menstrual disorders and rheumatoid arthritis.
Cai et al., 2021
In China and northeast Asia, it serve as tonics for nourishing kidney, strengthening bone and enhancing immunofunction in the treatment of impotence, menstrual disorders, osteoporosis, diabetes and inflammatory diseases such as rheumatoid arthritis and dermatitis
Wang et al., 2002a,b
Roots decoction help nourish the kidneys, enhance the immune function, and strengthen the bones by warming and tonifying the kidney.
Lin et al., 2010
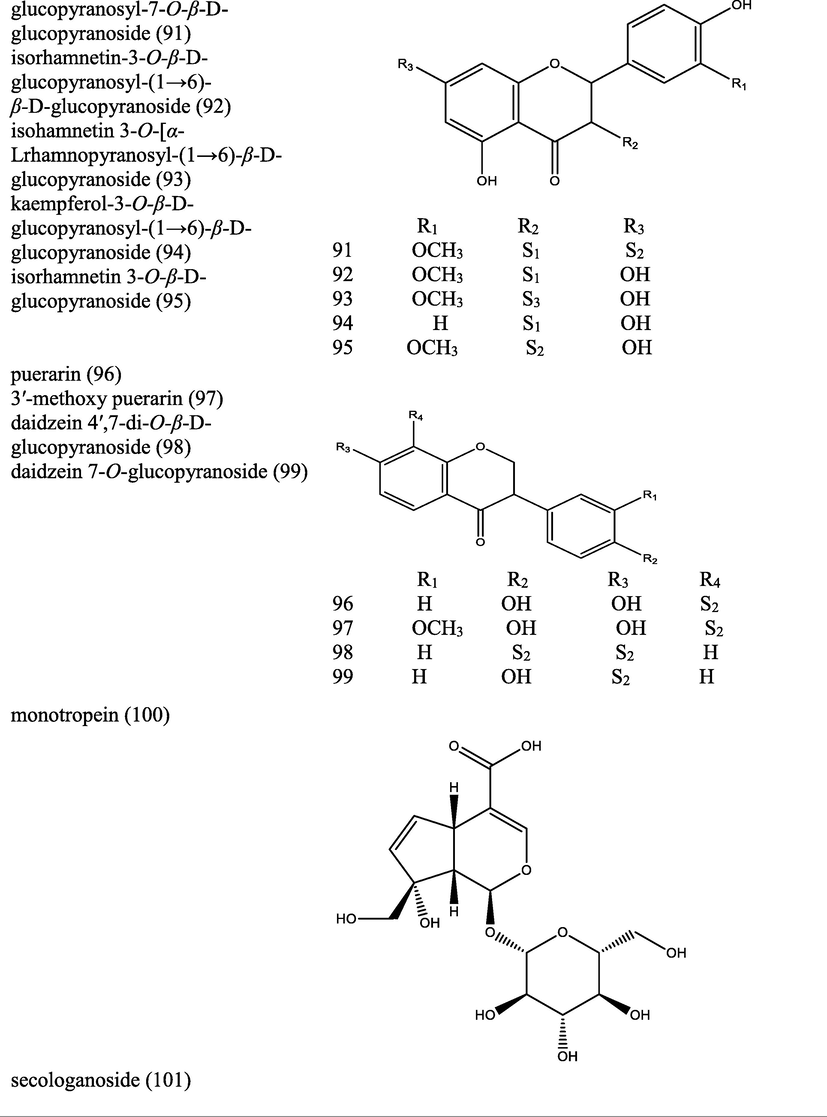
Used for the treatment of bone protection, andrological and gynaecological healthcare
Luo et al., 2021
In China, dried roots used as tonics to nourish kidney, strengthen muscle and enhance immunity in the treatment of impotence, osteoporosis, depression and inflammatory diseases such as rheumatoid arthritis and dermatitis.
Yu et al., 2019
Dried roots is used in China for invigorating kidney, strengthening muscle and bone, consolidating physical strength.
Dried roots is used to reinforce kidney function, strengthen the tendons and bones and relieve rheumatic condition.
(Li et al., 2007;
Morinda pandurifolia
Roots, bark, stems, leaves and fruits are used for the treatment of diabetes, hypertension and cancer.
Ruksilp et al., 2011
Morinda parvifolia Bartl. ex DC
Used in the treatment of tracheitis, coughing, and dyspepsia
Su et al., 2018Su et al., 2019a,b
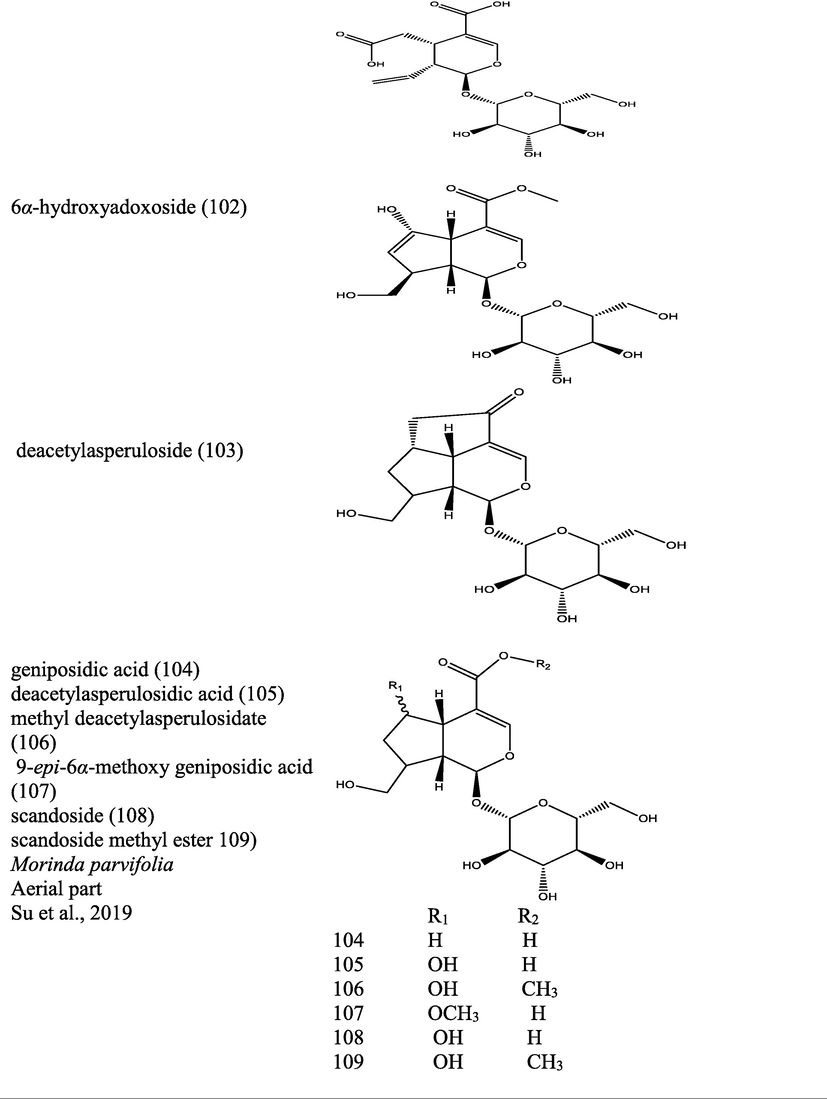
Morinda pubescens
Bark is used in treating eczema, fever, ulcer and glandular swellings. Leaves are useful for digestive disorders and venereal diseases.
Nivas et al., 2011Wee, 1992
Morinda tinctoria (Roxb.)
In Ayurveda and Siddha, dried leaves and roots are used as astringent, deobstrent, emmenagogue and pain reliever in gout.
In South India, different parts of the plant is used for the treatment of arthritis, diarrhoea, gastric ulcer, liver diseases and diabetes
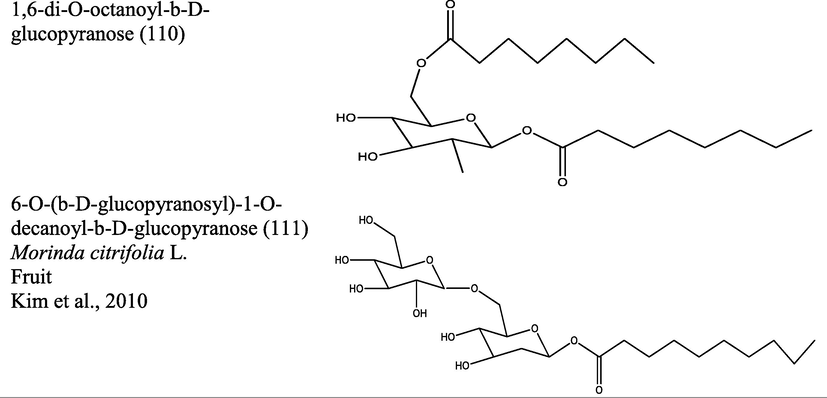
Dinakaran et al., 2008
Morinda umbellata L.
Dried roots, bark, stems, and leaves are used in the treatment of fever, coughing, stomach ache, and acute hepatitis.
Li et al., 2019
5 Secondary metabolites of the genus Morinda
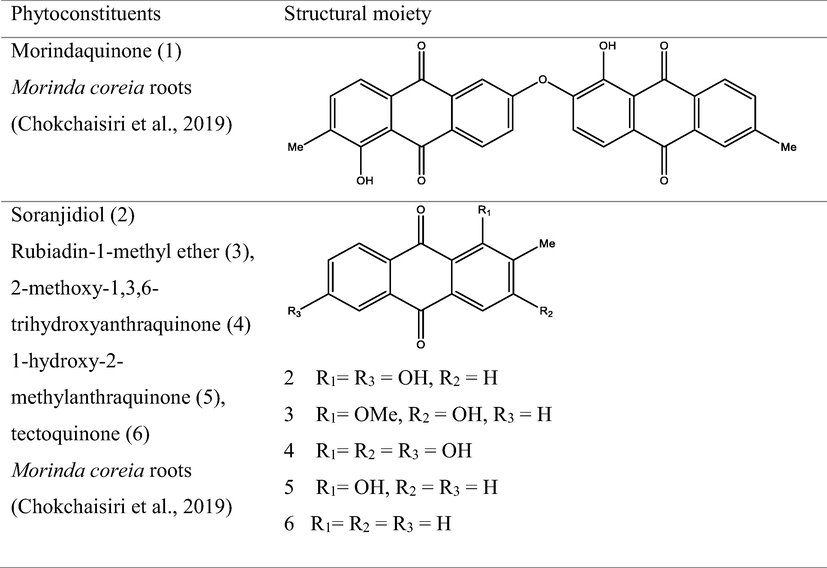
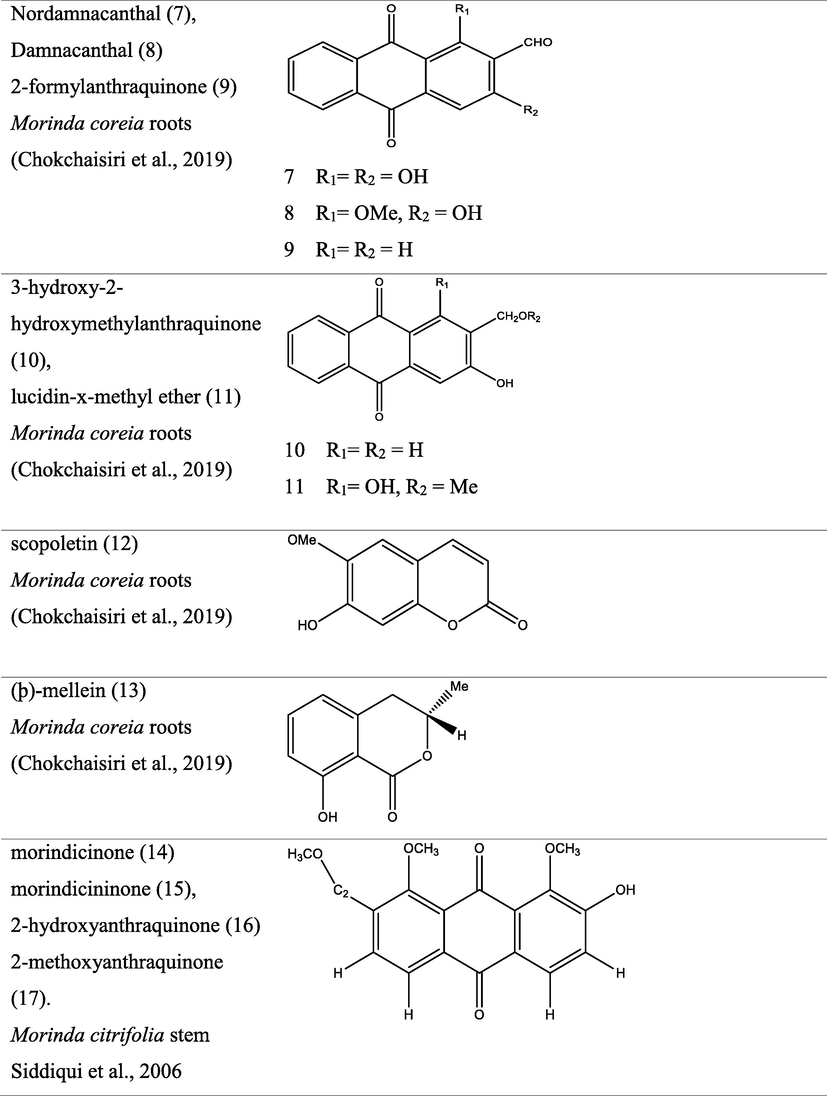
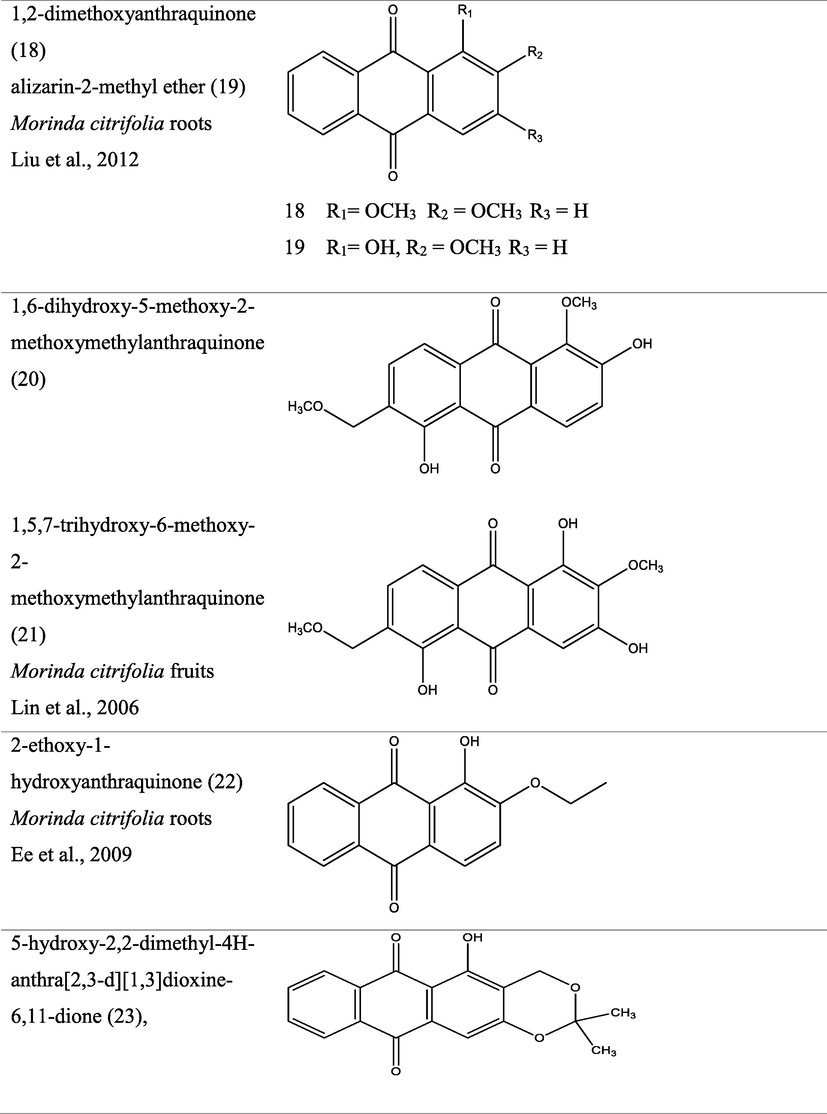
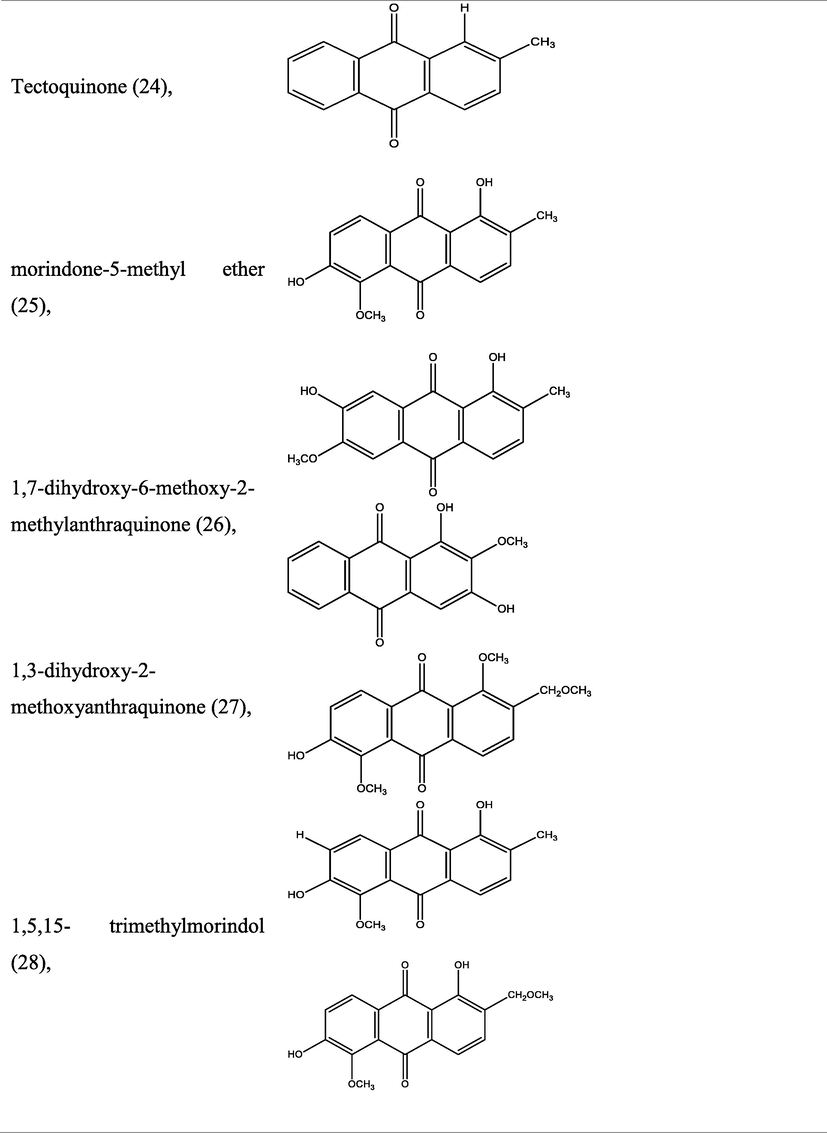
The genus Morinda has afforded an array of secondary metabolites with prominent therapeutic potencies for potential drug discovery. An assortment of secondary metabolites with unique structural moiety belonging to various groups of phytochemicals have been isolated from this genus and their therapeutic potency have been extensively studied via in vivo and in vitro experimental procedures. The abundant nature-synthesized metabolites from the genus Morinda with their unique structural moiety are listed in Tables 2–3 while other metabolites are described (Fig. 2–5).
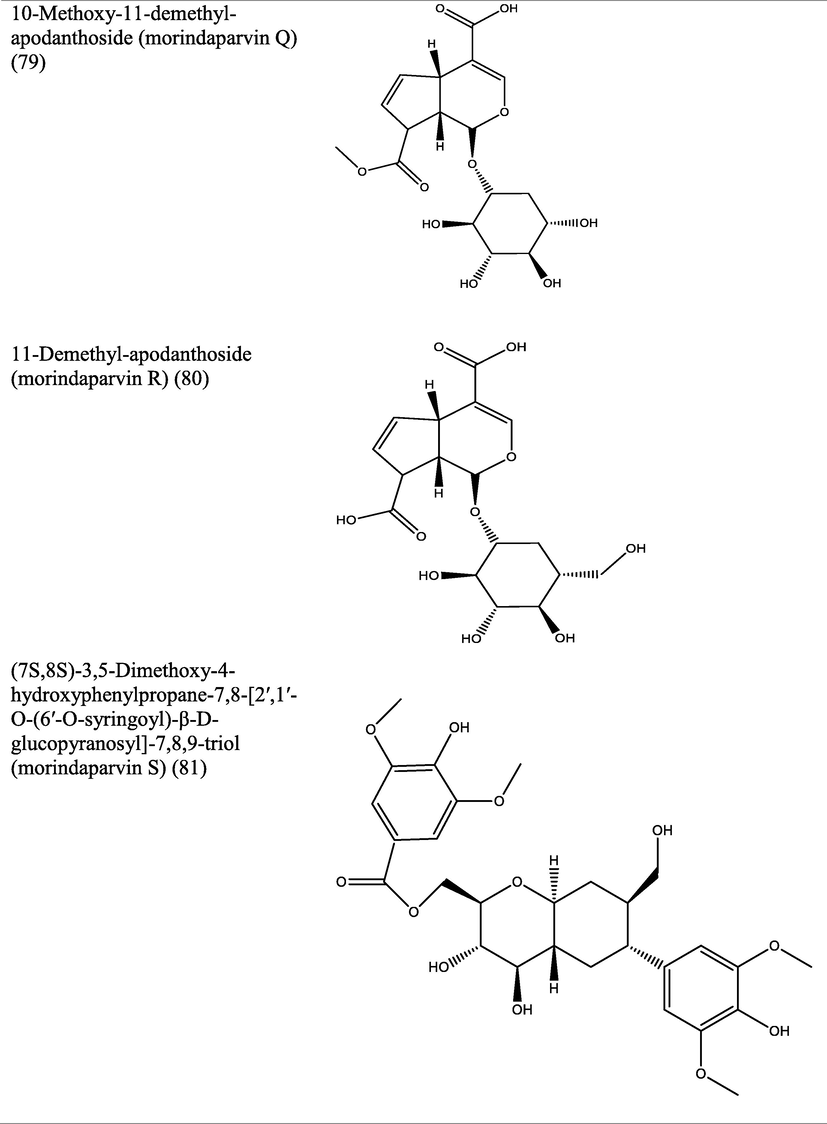
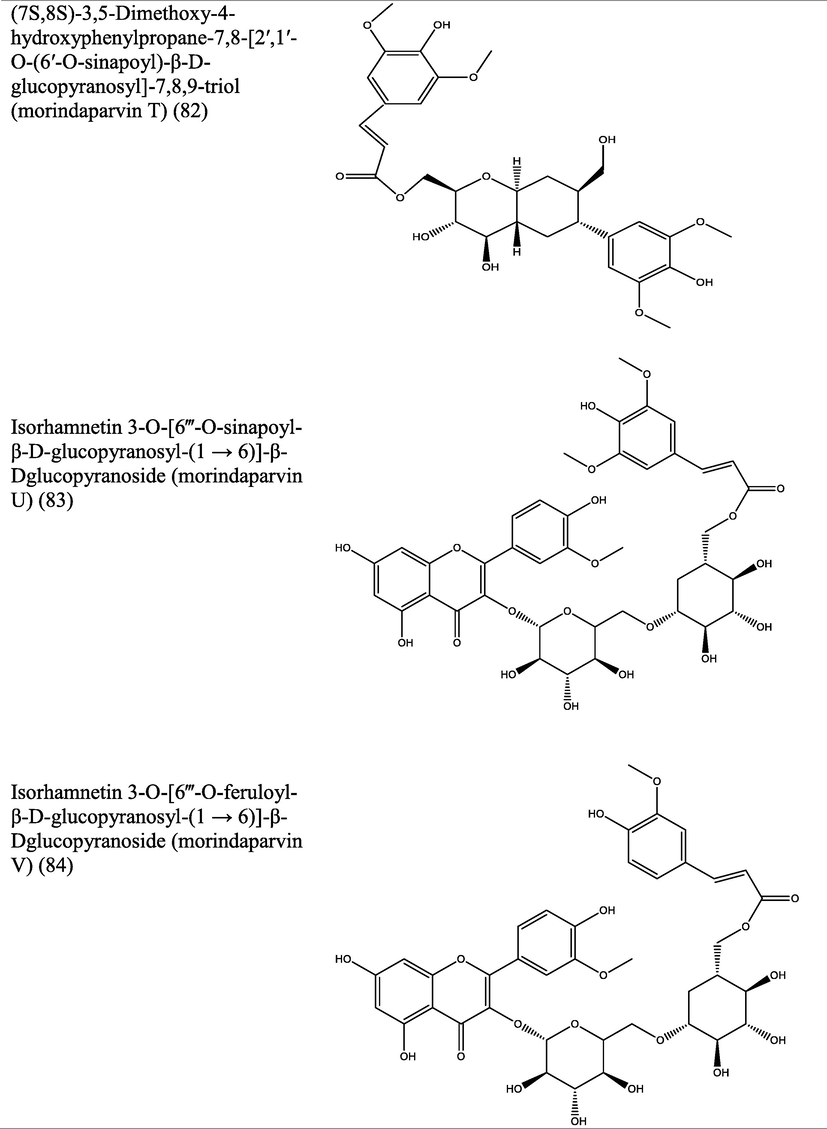
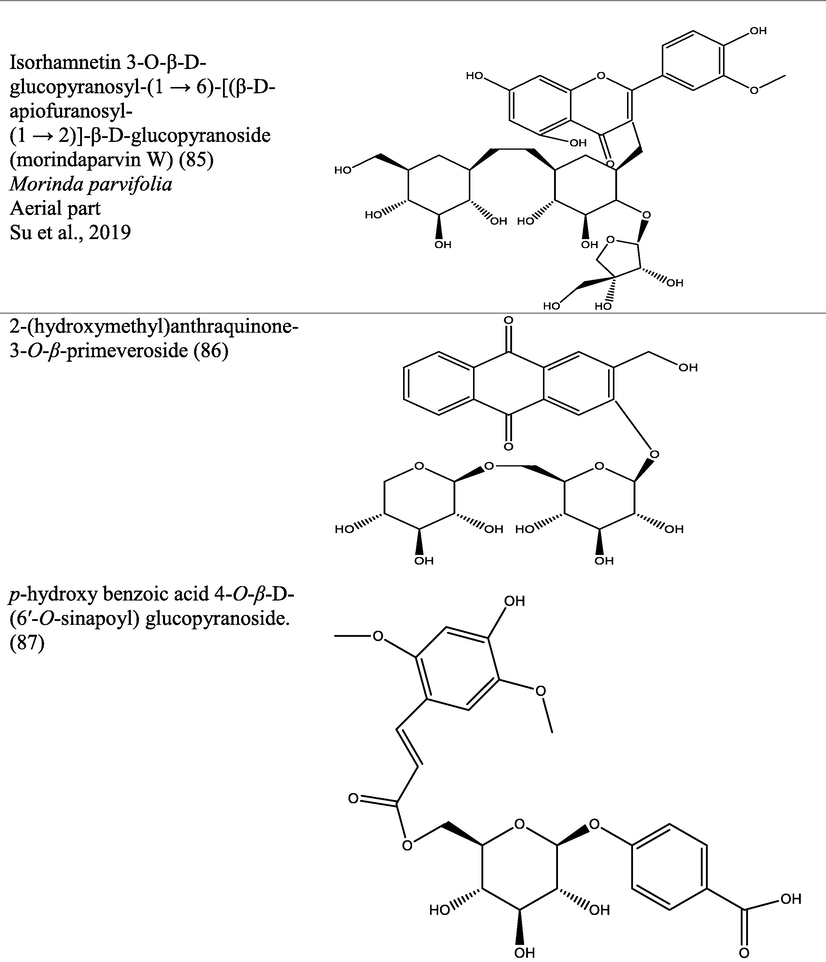
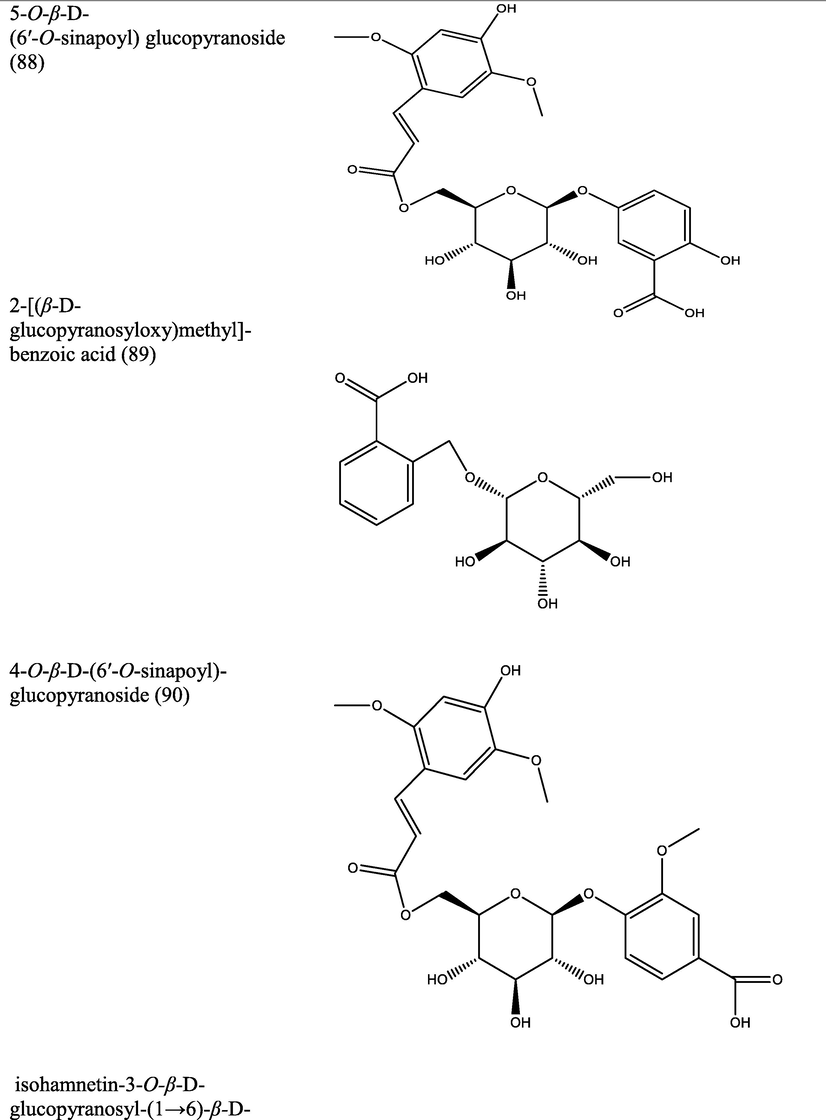
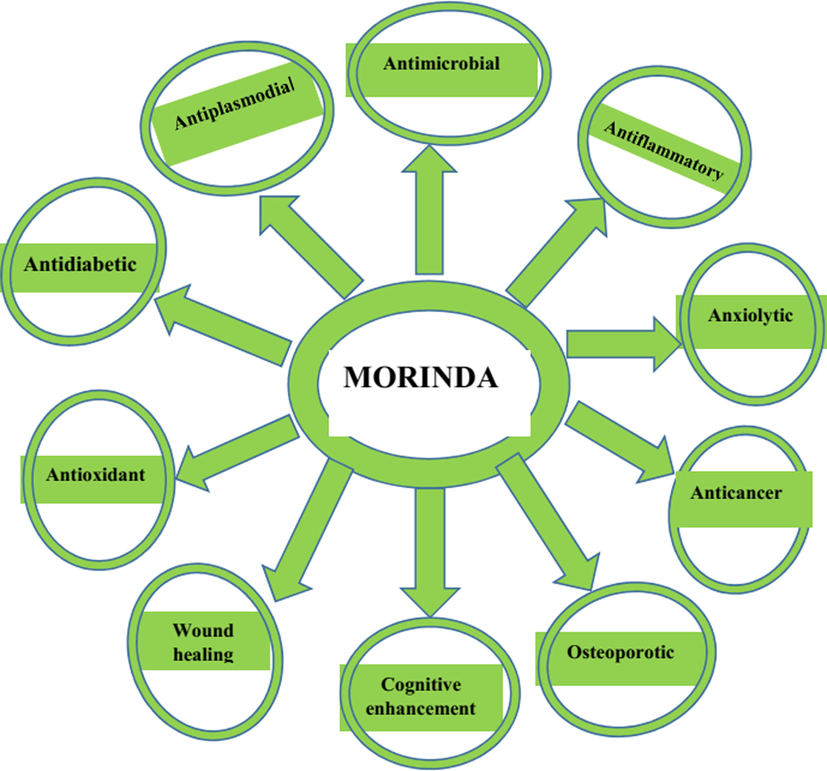
Pharmacological properties shown by different extracts of Morinda species.
5.1 Other isolated phytochemical compounds
5.1.1 Phytosterols and polyphenols
Plant sterols and polyphenolic compounds are two essential secondary metabolites found in medicinal plants (Fig 3). Some important phytosterols such as stigmasterol (112), β-sitosterol (113) and campesterol (114) were isolated from M. lucida leaf and stem bark respectively (Chithambo et al., 2017; Nweze, 2012). Also, pharmacologically active phytosterols such as daucosterol (115), scopoletin (116), 3β, 20(R) - butyl, 5-alkenylcholesterol (117), 3β,5-alkenyl-spirostol (118) have been isolated from the root of M. offinalis (Li et al., 2010a,b).
Few polyphenolic compounds have been isolated from the genus Morinda. Some of the isolated metabolites include epicatechin (119) and catechin hydrate (120). These compounds were isolated from the leaf of M. lucida (Okeniyi et al., 2015).
5.1.2 Terpenes, terpenoids and iridoids
Several terpenoids and terpenes such as monoterpenes, diterpenes, and triterpenes have been isolated from the different parts of the genus Morinda. Structural moieties of selected terpenes, iridoids and terpenoids are shown in Fig. 4. The pronounced antimalarial activity of this genus could be linked to this class of secondary metabolite. From literature, leaf is widely used in folkloric application compared to other parts such as bark, stem, leaves, fruit and root, this could be linked to the assorted terpenes reported in leaf (Bekono et al., 2020).
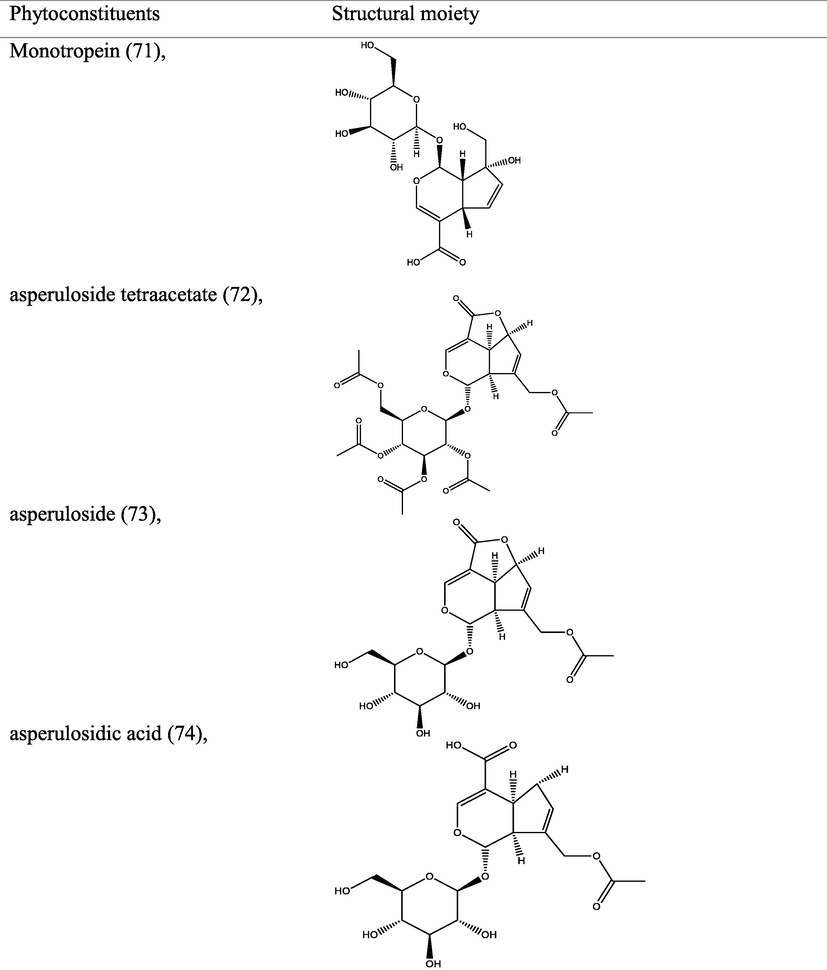
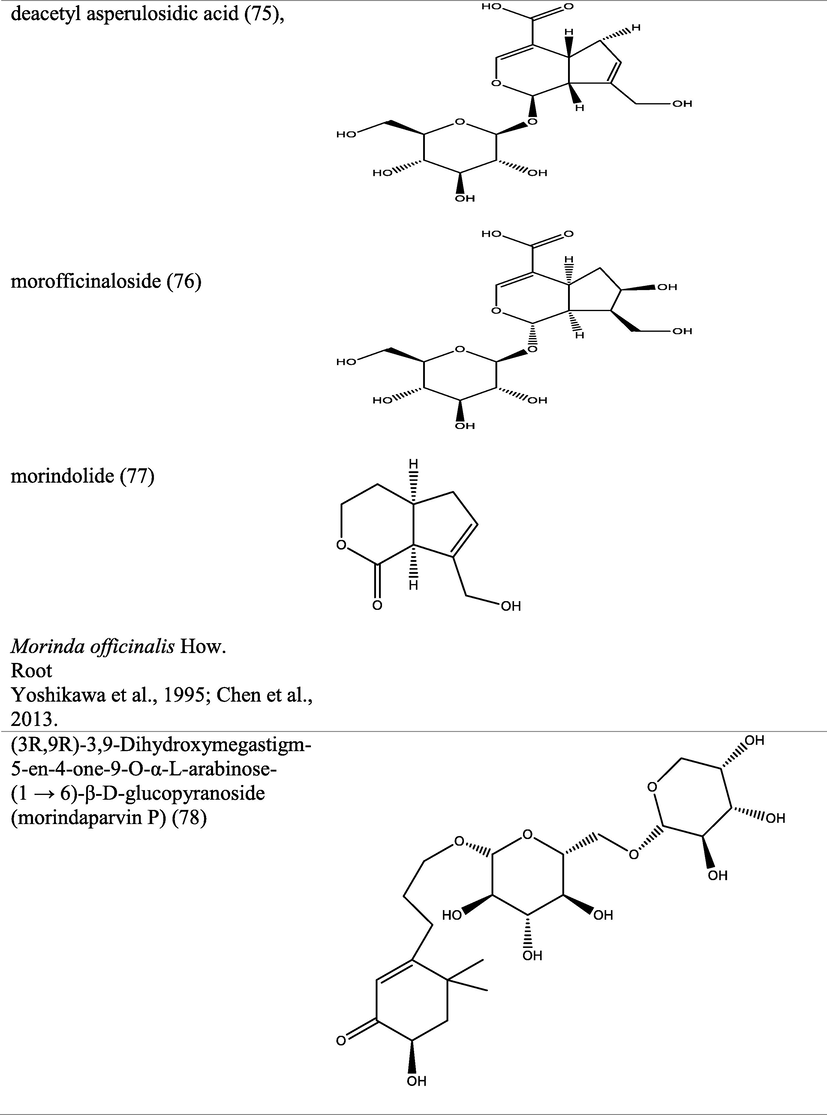
Iridoids are large group of phytochemicals with monoterpenoids-based structural moiety on a cyclopentan-[C]-pyran skeleton and display assorted health benefits. Eleven anti-inflammatory iridoids have been isolated from the aerial parts of M. officinalis How and characterized via spectroscopic techniques. The metabolites isolated were identified as officinaloside A-G (121–127), monotropein (128), asperulosidic acid (129), daphylloside (130), asperuloside (131) (Cai et al., 2021). Cytotoxic Iridoids isolated from the aerial parts of M. umbellata were identified as umbellatolides A (132) and B (133) via characterization by spectroscopic techniques (Ban et al., 2013).
A small number of monoterpenes, diterpenes and triterpenes have been isolated from the genus Morinda, however, despite the numbers, they exhibited exceptional biological potentials. A diterpene identified as phytol (134) and triterpenes identified as ursolic acid (135), cycloartenol (136) and oleanolic acid (137) were isolated from leaf and stem-bark of M. lucida respectively (Chithambo et al., 2017; Elufioye et al., 2015). A monoterpenoids of an iridoid ferulate, identified as Oruwacin (138) and a tetracyclic iridoids, molucidin (139) were isolated from the leaves of M. lucida (Suzuki et al., 2015; Adesogan, 1979). The volatile oils isolated from leaf and root of M. lucida were characterized as identified as cis-sabinene hydrate (140), cis-ρ-menth-2-en-1-ol (141), trans-ρ-menth-2-en-1-ol (142), borneol (143), neryl acetate (144), β-fenchyl alcohol (145) (Okoh et al., 2011; Owolabi et al., 2014).
Some of the monoterpenes which have been isolated from the genus Morinda include α-terpinene (145), β-bisabolene (146), α-thujene (147), camphene (148), 1,8-cineole (149), α-terpinyl acetate (150), sabinene (151), β-pinene (152), myrcene (153), α-phellandrene (154), α-3-carene (155), γ-terpinene (156), ρ-cymene (157), limonene (158), δ-terpineol (159), ρ-terpineol (160), nerol (161), linalool (162), terpinen-4-ol (163) and trans-sabinyl acetate (164) (Okoh et al., 2011; Owolabi et al., 2014). From essential oils isolated from leaf and root of M. lucida, about eight sesquiterpenes were isolated and characterized via spectroscopic s leaf and root oils respectively (Okoh et al., 2011; Owolabi et al., 2014).
5.1.3 Organic and fatty acids
Organic acids from plants are well documented for their antimicrobial, antitumor, antithrombotic as well as other applications (Chen, 2009). Few organic acids have been isolated from the genus Morinda (fig 5). Two organic acids identified as fumaric acid (165) and sucinnic acid (166) were isolated from the root of M. owerensis. These isolates displayed exceptional antidepressant activity (Cui and Yang, 1995; Zhang et al., 2010). In addition, two bioactive organic acids identified as 2-hydroxy-methyl benzoic acid (167) and 1,2-benzedicarboxylic acid (168) were isolated from the root oil (Okoh et al., 2011).
Some of the isolated fatty constituents from stem and root of M. lucida include hexacosanoic acid (169), hexadecanoic acid (170), dodecanoic acid (171), tetradecanoic acid (172) and 9-octadecanoic acid (173) (Adesida and Adesogan, 1972; Okoh et al., 2011),
5.1.4 Polysaccharides, mono- and oligosaccharides
Another prominent group of constituents in M. lucida is saccharides (Fig. 6). A diversity of oligosaccharides have been isolated from Morinda such as bajijiasu (174), mannose (175), nystose (176), 1F-fructofuranosyl nystose (177), inulintype hexasaccharide (178), heptasaccharide (179), sucrose (180), inulin-type trisaecharide (181), inulotriose (182), inulotetraose (183) and inulopentaose (184) (Li et al., 2004; Chen, 2009).
6 Pharmacological activities of the genus Morinda
Crude extracts, fractions, and isolated metabolites from Morinda spp. have been shown to have a wide range of therapeutic properties. The effects of a wide range of pharmacological properties, ranging from anti-infectious to pain and neurological disorders, are thoroughly investigated (Fig. 2).
6.1 Anti-infectious effects
6.1.1 Antimicrobial potentials
The antimicrobial potency is regarded as one of the most widely studied curative application of the genus Morinda. The activities reported in this section will be shown on the significance to human health via minimum inhibitory concentration (MIC) or minimum bactericidal concentration (MBC). The in vitro antifungal potency of hexane, ethyl acetate and methanol extracts of M. citrifolia were investigated in Aspergillus flavus, Aspergillus niger, Candida albicans, Fusarium species, Penicillium species, Rhizopus species and Trichophyton mentagrophytes. The percentage inhibitory of 62.06% and 79.30% were exhibited by ethyl acetate and methanol extract against T. mentagrophytes while activities of ≤ 50% was exhibited by methanolic extract against Fusarium. Penicillium and Rhizopus spp. C. albicans, A. niger and A. flavus were resistant to all solvent extracts tested (Jayaraman et al., 2008). Similarly, n-hexane, ethyl acetate and methanolic extracts of M. citrifolia were tested against Salmonella typhi, Klebsiella pneumonia, Shigella flexneri, Aeromonas hydrophila, Staphylococcus aureus, Enterococcus faecalis, Lactococcus lactis, Pseudomonas aeruginosa, Streptococcus thermophilus, Bacillus subtilis, Chromobacterium violaceum, Pseudomonas aeruginosa, Vibrio harveyi, Escherichia coli, Salmonella paratyphi, Klebsiella pneumonia and Vibrio cholera. Methanolic extract significantly developed resistance against all tested microbial isolates while hexane extracts displayed sparingly activities against some isolates (Jayaraman et al., 2008). The remediation potency of fruit juice obtained from M. citrifolia was tested against Enterococcus faecalis inoculated teeth cultured for 37 days in a CO2 atmosphere (37 °C). The inhibitory activity (smear layer removed) demonstrated by the fruit juice is comparable to NaOCl (when in synergy with EDTA) when viewed under electron microscopy. Though, toxicological profile and biocompatibility via preclinical and clinical trials were not reported (Murray et al., 2008). The antibacterial activity of butanol extract of M. citrifolia fruit partitioned with methanol was assessed in vitro against C. albicans, S. aureus and E. coli. A significant activity was displayed against C. albicans while moderate activity was exhibited against S. aureus. The activity could be linked to asperulosidic acid and deacetylasperulosidic acid analyzed (West et al., 2012). The damnacanthal isolated from M. citrifolia fruit exhibited pronounced in vitro anti-viral activity against human immunodeficiency viruses type 1 (HIV-1) accessory proteins in a Hela cells via an unfamiliar mechanism. Further studies to elucidate the mechanism of M. citrifolia that could be resourceful in assessing the efficacy as antiviral agents is inevitable (Kamata et al., 2006).
The scientific investigations of the genus Morinda could be traced to undocumented facts about its therapeutic potency in management of microbial infections. Several reports on the antimicrobial potency of leaves, bark and fruits of M. lucida have been documented, some of these are detailed below. The chloroform crude extract of M. lucida leaf was reported to exhibit pronounced bactericidal activity at 100 mg/mL and bacteriostatic activity at concentration of 12.5 to 100 mg/mL, authenticating the folkloric usage of this plant for the management of typhoid fever (Musa et al., 2014). Even at a low concentration of 10 mg/mL, leaf extract of Brimstone tree exhibited strong inhibitory activity against P. aeruginosa and S. aureus (Addy et al., 2013). Polar solvent extracts (methanolic and aqueous) of stems, roots and bark of M. lucida were active against bacteria isolates such as E. coli, S. paratyphi, S. typhi and S. typhorium with promising activity higher than ciprofloxacin and chloramphenicol (concentrations of 5, 10 and 20 mg/mL) (Fakoya et al., 2014). M. lucida root (erthanolic) extract exhibited strong inhibitory activities against gram positive bacteria isolates such as Micrococcus luteus and S. aureus (Chukwujekwu et al., 2005). Similarly, ethanolic and aqueous extracts of Brimstone tree (stem-bark) exhibited strong inhibitory activities against S. typhi, P. aeruginosa, Klebsiella pneuoniae, S. aureus, Flavobacterium sp. and Bacillus subtilis,(Adomi and Umukoro, 2010). Methanolic extract of M. lucida leaves also exhibit in vitro and in vivo antimicrobial potency against E. coli (Ogundare and Onifade, 2009). The antifungal activity of M. lucida leaves extract against isolates such as C. albicans is well documented (Anowi et al., 2013).
The antibacterial potency of fresh and fermented fruit juice of M. citrifolia was experimented in ten bacterial isolates using well diffusion method. The assay was carried out taking note of bactericidal concentration (MBC) and Minimum Inhibitory concentration (MIC). After incubation, MBC values ranges from 10 to 29 mg/mL while MIC from 2.5 to 5.0 mg/mL respectively. This indicate that fresh and fermented fruit juice exhibit bacteriostatic effects on bacteria strains. However, Fresh juice has a bacteriostatic effect on S. epidermidis and E. coli, while fermented juice induce bacteriostatic effect on S. epidermidis, Streptococcus oralis and Enterococcus faecalis. (Sina et al., 2021).
In spite of the plethora antimicrobial applications of this genus, there is scarcity of documented information on the antibacterial compounds isolated and the mechanisms of action of such metabolites.
6.1.2 Antiplasmodial/ antimalarial potentials
Malaria is a parasitic disease of global health concern especially in the tropical and subtropical regions. Over 228 million cases of malaria was reported in 2018, with 93% from Africa (Oladeji et al., 2020, 2021; WHO, 2015). This parasitic infection is caused by Plasmodium species transmitted by bite of female Anopheles mosquitoes (Oladeji et al., 2021; Oladeji and Oyebamiji, 2020; Saini et al., 2022). In view of this, several scientific investigations have explored and authenticate the therapeutic potency (antiplasmodial and antimalarial) of medicinal plants (Table 4).
S/n
Plant Name
Plant part (Type of extract)
Plasmodium specie (Assay or technique)
Activity
Reference
1
Morinda lucida
Leaf, bark
(n-hexane, ethyl acetate and ethanol)
Plasmodium berghei NK65 (in vivo)
(Curative)The ethanolic crude extracts (200 and 400 mg/kg) significantly increased the packed cell volume. The lowest inhibitory effect was observed in 200 mg/kg ethanolic bark extract treated group with activity of 72.16%.
Oladeji et al., 2021
2.
Morinda lucida
Leaf
(Partially purified cysteine-stabilised peptide)
P. falciparum W2 (in vitro) and P. berghei NK65 (in vivo).
(Suppressive)PPCSP extract was not active against P. falciparum W2 in vitro (IC50: >50 μg/mL) but was active against P. berghei NK65 in vivo, causing 75.8% reduction in parasitaemia at 125 mg/kg.
Adebayo et al., 2017
3.
Morinda lucida
leaves, stem bark, root bark
Plasmodium berghei (in vivo)
(Suppressive)Each extract produced a degree of chemosuppression of parasitaemia, with the most promising being the chromatographic fractions of the stem bark extract and the highest dose producing activity of 96.4%.
Obih et al. (1985)
4.
Morinda lucida
Leaf
Plasmodium berghei NK65 (in vivo)
(Curative and suppressive)In established infection, there was a sharp fall in parasitaemia from the second day of treatment in the groups administered chloroquine (5 mg/kg body weight) and M. lucida extract (administered subcutaneously) whereas a daily increase in parasitaemia was observed in the control group.
Makinde and Obih (1985)
5.
Morinda lucida
Leaf
(Petroleum ether)
Plasmodium berghei NK65 (in vivo)
(Curative and suppressive)The fractions demonstrated schizontocidal activity against early and established infections but showed less activity when evaluated for repository effect.
Awe and Makinde, 1998
6.
Morinda lucida
Leaf
(Ethanolic and dichloromethane)
P. falciparum (in vitro)
(Curative)Activity of 60.0% inhibition of P. falciparum growth at 6 μg/mL
Tona et al., 1999
7.
Morinda lucida
Leaf
(Aqueous)
Plasmodium berghei NK65 (in vivo)
(Curative, prophylactic and suppressive)The extracts produced significant dose-dependent activity against the parasite. The extract caused 50.2% inhibition of parasite growth at 200 mg/kg body weight on day 4 post-inoculation.
8.
Morinda lucida
Leaf
(Aqueous)
P. falciparum (in vivo)
(Curative)The extract caused 92.7% reduction in parasite density after one week of treatment.
The curative antiplasmodial potency of stem bark and leaf extracts of M. lucida was investigated in Plasmodium berghei NK65 infected Swiss Albino mice using curative assay. The solvent extracts significantly increase the packed cell volume and decrease in body weight of infected mice was observed after treatment. Chloroquine treated group exhibited 100% inhibition while the lowest inhibitory effect of 72.16% was observed in 200 mg/kg ethanolic bark extract treated group. The pronounced activity could be linked to high phenol, flavonoids and tannin contents in the extracts (Oladeji et al., 2021). Partially purified cysteine-stabilized peptide extract of M. lucida leaf was evaluated against P. berghei NK65 (in vivo) and P. falciparum W2 (in vitro). The purified extract exhibited no inhibitory activity against P. falciparum W2 (IC50: >50 μg/mL, in vitro), on the other hand, it exhibited 75.8% parasitaemia reduction against P. berghei NK65 (in vivo) (Adebayo et al., 2017). The schizontocidal potency of leaf extract of M. lucida was assessed in P. berghei infected Swiss Albino mice and inhibitory effect against early and established infections were assessed using chloroquine as control. A single dose of the extract was administered on daily basis while combination of pyrimethamine with extract was evaluated. For early infection, leaf extract exhibited close inhibitory activity to that exhibited by chloroquine treated group, however, daily decrease was observed in chloroquine (5 mg/kg bw) treated and extract treated groups (administered intravenously) with a significant reduction in level of parasitaemia starting from day 2. Furthermore, a pronounced reduction of 70.0% and 80.5% chemosuppresssion of parasitaemia in infected mice of pyrimethamine (1.2 mg/kg bw) and 1/6 dilution of stock of extract were observed after treatment (Makinde and Obih, 1985).
The antiplasmodial activities of solvent fractions of Brimstone tree have been reported. The fractions obtained from petroleum ether extract (leaves) was appraised in vivo against P. berghei berghei. The fractions exhibited significant schizontocidal activity against early and established infections, however, it showed no repository effect (Awe and Makinde, 1998). The curative potency of methanolic root extract and synergic effect of root and leaf extracts of M. lucida were assessed in plasmodium infected mice. The combined extracts produced higher parasitaemia inhibitory activity compared to activity exhibited by single extract (root extract) (Umar et al., 2013). Dichloromethane and ethanolic extracts of M. lucida leaves exhibited about 60.0% inhibitory activity against P. falciparum (in vitro) at a concentration of 6 μg/mL. Aqueous extract of M. lucida leaf was investigated against early, established and residual parasitaemia infections in P. berghei NK65–infected mice. The leaf extracts all exhibited strong inhibitory activity against the infected mice (dose-dependent). The inhibitory activity of 50.2% was observed in 200 mg/kg b.w treated group after four-day inoculation .
6.2 Effects on digestive disorders
6.2.1 Purgative/anti-diarrhoeal activity
Purgatives or laxatives are chemical substances or drugs which aid bowel movement, treat constipation and loosen stools. The folkloric usage of genus Morinda as purgative is well known, however, few scientific validation have been reported. The oral administration of methanolic extract of Brimstone tree leaves significantly induced purging in experimental mice. In a similar study, oral administration of 12.5 to 100 mg/kg body weight of M. lucida methanolic leaf extract induced purging of wet feaces in Swiss albino rats (Olajide et al., 1999). The aqueous fruit and leaf extract of Brimstone tree lessen castor oil-induced diarrhea in rat model (Adejo et al., 2015). These differences in the activities and possible toxicity of administered extracts need more clarification.
6.3 Effects on pain and neurological disorder
6.3.1 Cognition enhancement
Cognitive disorders (CDs), also known as neurocognitive disorders, are a broad category of mental health illnesses that affect problem-solving, perception, memory, and learning abilities.
Plants have been used in folkloric medicine in several parts of Africa and Asia to treat memory loss, improve memory, and cure any neurological illnesses associated with aging or other interesting factors. The ethyl acetate fractions collected from the leaf of M. lucida was assessed for its butyrylcholinesterase and acetylcholinesterase activities. The fractions significantly increase the function of cholinergic neuron and potency in repairing cholinergic memory impairment. The activity exhibited was linked to the presence of phytol which act as major inhibitor to cholinergic impairment (Elufioye et al., 2013). In scopolamine-induced amnesic mice, the leaf extract (M. lucida) has been shown to improve cognitive function (Elufioye and Hameed, 2017). Administration of aqueous extract of M. owariensis (11.25 to 45 g/kg) significantly enhance the memory and learning impairment caused or induced by D-galactose in experimental mice. The dose concentrations administered for 28 days significantly reduced serum glycosylated hemoglobin, aldose reductase and the damage to brain cells however, glycosylation end products was increased (Ye et al., 2015).
The call for memory enhancers and boosters derived from natural or synthetic sources is growing on a daily basis as a result of environmental and societal influences on the human brain and emotions. The memory enhancement potency of dried fruits of M. citrifolia was assessed in scopolamine induced memory impairment in mice with concentrations ranging from 5 to 400 µg/mL. Ethanolic extract and ethyl acetate fraction successfully enhance the memory of experimental mice while butanol fraction had no effects on impaired experimental mice. The activity reported followed the pathway reported by Pachuari et al., that is, acetylcholinesterase inhibitory and cerebral circulation augmentation. Secondary metabolites such as scopoletin, rutin, quercetin analyzed could be responsible for this activity (Pachauri et al., 2012). Also, administrations of various dosage of aqueous extracts of M. owariensis significantly enhanced memory capacity of rats induced by natrium nitrosum and D-galactose after 30 days. The extract decreased malonaldehyde, mRNA expression and activity of monoamine oxidase-B, however, SOD activity was increased under step-down test (Wang et al., 2013). According to the reports of Chen et al., oligosaccharides of M. owariensis lessen memory or learning impairments or dysfunction caused by β-amyloid in experimental mice. This compound improve oxidation resistance, monoamine neurotransmitter, brain energy metabolism and also, reduce brain neuron apoptosis and injury to the cholinergic system. In a similar study, a dimeric fructose isolated from M. owariensis identified as Bajijiasu lessen Aβ-induced memory or learning impairments, boost energy metabolism, anti-oxidation and attenuated cholinergic system damage in neurotoxicity rats caused by injection of Aβ25–35 into the bilateral CA1 region of the experimental rat. The compound also increase potentiation but also, reduce time lapse of the frequency of population spike in the anoxic condition and enhance the resurgence after re-oxygenation. These features indicate the potency of Bajijiasu in enhancing and protecting memory and neurons against anoxic injury.
6.3.2 Anti-rheumatoid arthritis activity
Morinda plants are well documented in traditional Chinese medicine as antidote to rheumatism. Ethanolic extracts of M. oweriensis reduced paw swelling, serum levels of IL-6, interleukin-lβ (IL-lβ) and TNF-α in adjuvant arthritis rats (Shi et al., 2015). It also decrease prostaglandin E2 caused by Freund's complete adjuvant, and the writhing occurrence of mice caused by glacial acetic acid (Chen et al., 2011). In some part of China, combination of two or more medicinal plants, that is, Morinda species and other plants have significantly reduce rheumatism in man. This synergy was assessed in the combination of M. oweriensis and Achyranthes bidentata (ethanolic root) at dosage of 75, 150 and 300 µg/ml. The synergy increased the mRNA, proliferation, cyclin D1 of chondrocytes and protein expressions of cyclin-dependent kinase 4 (Wu et al., 2017). Similarly, synergic reactions between the roots of M. owariensis and Achyranthes bidentata significantly enhanced the proliferation of chondrocytes however, based on synergic effects, the rations was not optimized. According to spectroscopic data, ethanolic extract of M. owariensis exceptionally reduced the effects of glucose and lipid metabolism in rats with carrageenan-induced acute inflammation. The polysaccharides isolated from M. owariensis and Eucommia ulmoides at dosage concentrations of 800, 400, 200, 100 and 50 µg/mL significantly enhanced the rat chondrocytes (in vitro) (Fu et al., 2014).
6.3.3 Anti-fatigue and anti-aging activity
Fatigue is an overall feeling of tiredness which could be linked to lifestyle, mental or physical health conditions. The genus Morinda is well authenticated in folkloric medicine as energy booster or anti-fatigue therapeutic substances. Polysaccharides of M. owariensis extended swimming time of experimental mice and hepatic glycogen, and subsequently reduced blood lactic acid and serum urea nitrogen at dosage of 50, 100 and 200 mg/kg. The polysaccharides MP-1, MP-2 and MP-3 were analyzed to be 27.32%, 7.85% and 20.57%, respectively (Zhang et al., 2009). The anti-aging effects of genus Morinda is known to proceed via antiapopotic or anti-oxidative mechanism. Ethanolic leaf extract of M. owariensis significantly decreased the content of MDA, increased apoptotic index of purkinje fibers, expression of Bax protein in the cerebellum and SOD, the expression of B cell leukemia-2 gene product (Bcl-2) protein at 700 mg/kg and 2100 mg/kg body weight in aged rats. The extract also induce the immune system via spleen and thymus gland, B lymphocyte stimulation index, level of IL-2 and the number of positive CD28 + cells in D-galactose damaged rats (Wang et al., 2013). Similarly, M. owariensis leaf extracts (1 g/ml) enhanced cell vitality, mRNA expression of myosin heavy chain-α and reduced the activity of β-galactosidase and mRNA expression of myosin heavy chain-β in cardiomyocytes damaged by D-galactose (Zhang et al., 2011).
6.3.4 Effect on muscle contractility
Muscle contractility is a major concern in Africa caused by shortened muscle cells under stress or pressure. To the best of our knowledge, there has been no documented report on the use of genus Morinda as muscle relaxants in African folkloric medicine despite been listed among genus with cardio-active potencies. In spite of this, Ettarh and Emeka, reported that aqueous extract obtained from Brimstone tree leaf exhibited a significant vasorelaxant effect by endothelium-dependent and endothelium-independent mechanisms on vascular muscle (smooth) of rat aorta (Ettarh and Emeka, 2004). Similarly, aqueous extract of M. lucida exhibited pronounced relaxant effect on both pregnant and non-pregnant mice (uterine smooth muscle). The extract repressed the frequency of contractile responses and inhibited responses of contractile to acetylcholine and oxytocin in the muscle (Elias et al., 2007).
6.3.5 Anti-inflammatory activity
The human body is designed in a way that self-defense or healing could be accomplished by activating certain some body cells or tissues. Generally, certain drugs or chemicals have potency in reactivation or restoration of weaken cells. One of the common natural substance is the genus Morinda. This genus is widely used in folk medicine in lessen pain and treat inflammation either acute or chronic (Choi et al., 2005; Palu et al., 2012). The methanolic extract of M. owariensis significantly inhibited cyclooxygenase (COX-2), iNOS and TNF-α expression through down-regulation of the NF-κB binding activity (Kim et al., 2005). The activity was enhanced by pretreatment with either 20 mg/kg or 30 mg/kg monotropein. This lessen stretching episodes and extended activity in experimental mice (hot plate), also, increase antinociceptive properties and reduced acute paw edema caused by carrageenan in test organisms. These activities could be linked to monotropein administered (Choi et al., 2005). This compound could also inhibit mRNA expression of IL-1β, COX-2, iNOS and TNF-α, lessen the DNA binding activity, degradation of IκB-α and lessen phosphorylation in LPS induced RAW 264.7 macrophages (Shin et al., 2013; Zhang et al., 2016).
The fruit juice extracted from M. citrifolia obtained from Costa Rica inhibited the activity of cyclooxygenase (COX)-1 and -2 (in vitro) (Dussoss et al., 2011). In another study, M. citrifolia fruit juice from Tahiti reduced carrageenan paw edema in experimental rats, with anti-inflammatory effects similar to celecoxib, indomethacin, and acetylsalicylic acid (Su et al., 2001). Seed oils extracted from M. citrifolia inhibited both 5-LOX and COX-2 enzymes at concentrations of 0.5 and 1 mg/mL. The seed oil was reported to exhibit no side or toxic effects on test rats after examining for 28 days via patch test. The extracted seed oil demonstrated low risk of non-comedogenic or allergic dermatitis triggered by excessive fatty acid contents or linoleic acids in the seed (Palu et al., 2012). The anti-inflammatory potency of dietary supplements of M. citrifolia (400 mg, 38 in placebo group while 42 were treated with M. citrifolia) was assessed in women (100) using double-blind placebo-controlled approach by evaluating parameters such as menstrual blood loss, age, erythrocyte sedimentation rate, body size, packed cell volume, red blood cells and pain. There was no significant difference between the two groups after treatment period. To enhance its potency further study should involve the use of high dosage with a larger test population.
Several solvent extracts of genus Morinda have exhibited strong anti-inflammatory potentials (in vivo or in vitro). Methanolic and aqueous extracts of M. lucida leaf significantly inhibited acute inflammation and antipyretic activity in rabbit and experimental rat (Akah and Nwambie, 1994), and also, showed strong immunorestorative (in vivo) and immunostimulatory activities (in vitro). The dichloromethane extract of M. lucida leaf shown strong anti-inflammatory activity by inhibiting COX-2 activity while petroleum ether extract demonstrated strong inhibitory activity against COX-1 and COX-2 (Chukwujekwu et al., 2005). Similarly, aqueous extract of Brimstone tree doubled total IgG, IgG1, and IgG2a responses to ovalbumin when compared to control groups (Nworu et al., 2012). Ethanolic extract of M. lucida leaf improve immune system of Balb/c mice induced with rhabdomyosarcoma cells (Ala et al., 2017). Damnacanthal isolated from stem, bark and roots of M. lucida significantly suppressed and regulate lipopolysaccharide-induced nuclear factor-κB of paw and ear ede0ma in rats and mice (Koumaglo et al., 1992a,b; Nualsanit et al., 2011). These findings authenticate the folkloric uses of genus Morinda for the treatment of anti-inflammatory, immuno-inflammatory, immunostimulatory and infections.
6.3.6 Antiosteoporotic activity
Osteoporosis is a prevalent health disease marked by low bone mass and structural degradation of the bone, which increases the risk of fractures. The genus Morinda is widely used to treat bone atrophy in Traditional Chinese medicine (TCM). A clinical investigation including 50 patients found that the aqueous extract of M. owaiensis root is effective in the treatment of postmenopausal osteoporosis (Long et al., 2013). The administration of an aqueous extract of M. owaiensis root (50, 100, and 200 mg/kg) to sciatic neurectomized mice 3 days after neurectomy for 6 weeks for a prevention study, or 2 weeks after neurectomy for 12 weeks for a therapeutic study. These significantly and dose dependently suppressed the decrease in hind limb thickness, tibia failure load, and bone mineral density (BMD) with an increase in serum osteoclacin levels, as well as reversed (Seo et al., 2005). The administration of MO ethanol extracts to ovariectomized rats increased total BMD and trabecular BMD of the tibia, improved the levels of phosphorus, calcium, and osteoprotegerin (OPG), decreased the levels of deoxypyridinoline/Cr (DPD/Cr), tartrate resistant acid phosphates (TRAP), adrenocorticotropic hormone (ACTH), and corticosterone (CORT) in (Li et al., 2009). Antiosteoporotic activity was confirmed in eight anthraquinone compounds, particularly for suppressing osteoclastic bone resorption (Wu et al., 2009a,b). Moreover, osteoclasts synthesized from rat bone marrow cells were used to investigate the mechanism of these anthraquinones' inhibitory effect on bone resorption. In an osteoblast-osteoclast coculture system, serum from rats given MO aqueous extracts suppressed mRNA expression of carbonic anhydrase II (CA II) and nuclear factor of activated T cells (NFAT), as well as osteoclast differentiation and bone resorption activity. The current study, on the other hand, did not describe the chemical contents transported from M. owaiensis to rat serum, nor did it specify the active principles for reducing bone resorption (Zheng et al., 2013). Antiosteoporotic properties of MO polysaccharides have also been discovered. Oral administration of MO polysaccharides (100 or 300 mg/kg) to ovariectomized rats for 30 days enhanced BMD and bone mineral content while lowering cytokine levels in the blood, findings indicate that MO polysaccharides may prevent avert bone loss caused by ovariectomy (Zhu et al., 2008). However, there was no positive control in this trial, and serum biochemical indicators of bone metabolism, such as ALP and osteocalcin, as well as TRAP and cathepsin K activity, were not measured.
6.3.7 Antidepressant-like activity
Depression is one of the most frequent and severe disorders in the world, with significant socio - economic consequences. Compounds such as polysaccharides and oligosaccharides isolated from M. owariensis have been shown to have antidepressant-like activity in animal and cell experiments as well as human clinical trials (Qiu et al., 2016).
Post-traumatic stress disorder (PTSD) is a serious psychiatric disorder linked to the production of allopregnanolone. At a dose of 25.0–50.0 mg/kg, MO inulin-type oligosaccharides reversed behavioral deficits in single prolonged stress-treated rats, reduced freezing time in the contextual fear paradigm, increased the time and entries in open arms in the elevated plus maze test without affecting locomotor activity in the open field test, and increased the levels of allopregnanolone in the prefrontal cortex, hippocampus, and amygdala, demonstrating that (Qiu et al., 2016). Polysaccharides and oligosaccharides abundant in MO root. In an experimental depression model of rats, administration of MO polysaccharides reduced the number of T-type labyrinth test errors and increased the number of neurons and level of superoxide dismutase (SOD) in the hippocampus, indicating that MO polysaccharides could mitigate oxidative stress reactions and improve cognitive behavior (Liu, 2011). Administration of MO oligosaccharides (50, 25 and 12.5 mg/kg/d) for 14 days significantly enhanced sucrose preference in stressed rats, decreased the immobility time, and increased the protein levels of brain-derived neurotrophic factor (BDNF), glycogen synthase kinase-3β (GSK-3β), glutamate receptor subunit-1(GluR1), postsynaptic density-95(PSD 95) and synapsin 1 in the hippocampus, indicating that MO oligosaccharides exerted a therapeutic effect in the rat model of chronic stress-induced depression by regulating the BDNF signaling pathway and enhancing synaptic plasticity (Xu et al., 2015). The minimum effective dose of MO extracts for mice was found to be 75 mg/kg in this study, with desipramine serving as a positive control. It does not, however, provide a comparison of differences between MO extracts and desipramine. Furthermore, following 15 days of treatment of MO aqueous extract, 17-month Kunming mice were subjected to an intensive swimming test. MO aqueous extracts increased the swimming duration of the loaded mice and the levels of norepinephrine, epinephrine, and dopamine in the brain tissue, while decreasing the amount of 5-HTP, suggesting that MO may have a favorable effect on the brain via modulating monoamine neurotransmitter levels (Zhang et al., 2014).
6.4 Other activities
6.4.1 Wound healing activity
Few reports of the genus Morinda have been reported. Although, leaf juice (1 mjg/mL), ethanolic and methanolic extracts (10 – 200 mg/mL) and hexane fractions (10 – 200 mg/mL) of M. citrifolia demonstrated distinct wound healing potentials in mice models. Ethanolic and methanolic leaf extracts exhibited strong wound healing potency compared to activity shown by control. This activity could be linked to ligand binding to PDGF/A2A receptor which initiated wound closure (Palu et al., 2010). The results should be substantiated via further in vivo assays and mechanism of activity needs further clarification.
6.4.2 Gastric ulcer healing activity
The gastro-esophageal (in vivo) antiinflammatory effects of Thai dried mature M. citrifolia fruit (unripe) aqueous extract in rats were reported at 0.63–2.50 g/kg. The probable mechanisms of action was observed to contribute to the emergence of acute gastric lesions and also, blocking esophageal reflux. The antisecretory activity of the extract is similar to that exhibited by lanzoprazole and ranitidine (Mahattanadul et al., 2011). Twenty healthy volunteers aged 18 to 45 were given a single dose of Thai M. citrifolia fruit extract (aqueous) containing 25.15 ± 0.11 lg/ml scopoletin in an open-label, two-period crossover research. After 30 min, the volunteers were administered one 300 mg ranitidine tablet and blood samples were collected for 12 h at fixed intervals. The presence of scopoletin in M. citrifolia stimulated the 5-HT4 receptor. To substantiate the findings, comprehensive clinical assessments needs to be carried out with patients with suffering from stomach motility defects or symptoms.
6.4.3 Immunity enhancing activity
The investigation (in vivo) of immunity potency of M. citrifolia (Tahitian and commercial fruit juices) on mice was observed to significantly enhanced the immunity of tested organisms. This indicated that M. citrifolia restrains the immune system by activating CB2 receptors, inhibiting interleukin-4, and augmenting the production of interferon gamma cytokines. Further study should focus on comprehensive in vivo and clinical assays to investigate the dosage and mode of action of M. citrifolia on the immune system (Palu et al., 2008).
6.4.4 Anticancer activity
Cancer is a huge public health concern around the world (Siegel et al., 2014). It refers to a group of disorders in which aberrant cells develop and spread abnormally (ACS, 2015). In ethnobotanical surveys done among traditional medicinal practitioners in South West Nigeria, M. lucida was referenced in recipes for the management of cancers and cancer-related disorders (Sowemimo et al., 2007). Generally, infusions and decoctions of M. lucida leaf and stem bark have been reported to be used in the cancer therapy in South West Nigeria (Ashidi et al., 2010). Aqueous extract of M. lucida leaf displayed antiproliferative action (in vitro) by triggering apoptotic cell death in HL-60 cells (Appiah-opong et al., 2016). Moreover, molucidin isolated from M. lucida leaves has been reported to be cytotoxic, with an MLD50 for some cancer cell lines of less than 10 μM. LoVo (colon cancer cell line) and KATO III (stomach cancer cell line) have MLD50 values as low as 0.11 and 0.21 μM, respectively, with strong selectivity indices (SI) for several cancer cell lines (Suzuki et al., 2015). The most active components of the methanolic extract of M. lucida leaf were cytotoxic to monkey kidney epithelial cells (LLCMK2), with the Median lethal dose (MLD50) ranging from 15.625 g/mL to 31.25 g/mL. Sulphated polysaccharide inhibits metastasis by disrupting the interface between glycosaminoglycan and certain proteins, while damnacanthal impedes tumor formation either by restricting with the growth of ras gene activation (Hiramatsu et al., 1993) or by increasing apoptosis in human colorectal cancer cell lines.
6.4.4.1 Anticancer activity (In vitro)
Based on the extracts' toxicological profiles, Thai M. citrifolia fresh and dried leaf dichloromethane extracts were reported to be more effective and presumably safer in treating cancer than M. citrifolia pure components such as damnacanthal, rutin, and scopoletin. The study included four human cancer cell lines (epidermoid carcinoma, cervical carcinoma, breast carcinoma, and hepatocellular carcinoma), as well as a Vero (African green monkey kidney) cell line. The pure compounds, rutin and scopoletin, reduced anti-proliferative effects on all human cancer cell lines, whereas both M. citrifolia extracts had an inhibitory effect on epidermoid carcinoma and cervical carcinoma cells. Only damnacanthal, demonstrated potent cytotoxic effects on all human cancer cell lines and African green monkey kidney cell lines (Kamiya et al., 2010a,b). Glycosides derived from M. citrifolia fruit juice (6-O-(b-D-glucopyranosyl)-1-Ooctanoyl-b-D-glucopyranose and asperulosidic acid) were effective in suppressing the induced transformation in the mouse epidermal JB6 cell line (Liu et al., 2001). The Indian M. citrifolia fruit juice, cisplatin (an anti-cervical cancer medicine), or a combination of the two were used to treat HeLa and SiHa cell lines, and it was observed that all of the tested formulations were able to induce apoptosis in the cell lines. However, cisplatin had a quite higher cell mortality rate than M. citrifolia fruit juice, although their combination produced comparable efficacy (Gupta et al., 2013). In comparison to hamster (6%) or human laryngeal (13%) cell lines, the crude extract of Hawaiian M. citrifolia fruit was found to substantially inhibit neuroblastoma (36%) and breast cancer (29%) cell lines, but had no effect on green monkey kidney (0%) cell lines (Arpornsuwan and Punjanon, 2006). Moreover, the fermented Hawaiian M. citrifolia juice served as a dendritic cell anti-proliferative, stimulating both splenocytes and B cells to produce IgG and IgM. (Wong, 2004).
6.4.4.2 Anticancer activity (In vivo)
In another study, mice treated with Hawaiian M. citrifolia fruit ethanol precipitate combined with anti-cancer drugs such as cisplatin, adriamycin, mitomycin-C, bleomycin, etoposide, 5-fluorouracil, vincristine, or camptothecin showed synergistic beneficial effects after being exposed to sarcoma tumor cells. When M. citrifolia was coupled with paclitaxel, cytosine arabinoside, or immunosuppressive anticancer medicines such cyclophosphamide, methotrexate, or 6-thioguanine, no beneficial effects were observed. Mice treated with the fruit ethanol precipitate alone, on the other hand, had a cure rate of 25–45%. (Furusawa et al., 2003). Rats with cancer in certain organs were administered M. citrifolia juice in their drinking water at a concentration of 10%. The creation of DNA adducts was reduced after one week, depending on the sex and organ. Female rats had heart 30 percent reduction, liver 42 percent reduction, lungs 41 +-/percent reduction, and kidneys 80 percent reduction, while male rats had heart 60 percent reduction, liver 70 percent reduction, lungs 50 percent reduction, and kidneys 90 percent reduction (Chan-Blanco et al., 2006). Chemically induced carcinogenesis in the rat esophagus was also prevented using freeze-dried Tahitian M. citrifolia fruit (Stoner et al., 2010). Another anticancer study was carried out on three groups of eight mice, each of which was given an Ehrlich ascites tumor. Oral M. citrifolia juice (a commercial product from the Netherlands), doxorubicin (a powerful anticancer medicine), and a combination of both were administered to the first, second, and third groups, respectively. The study revealed that M. citrifolia fruit juice, either singly or in synergistic effects with doxorubicin, could be effective in the treatment of breast cancer (Taskin et al., 2009).
6.4.5 Antidiabetic activity
The hypoglycemic effect of Tahitian M. citrifolia fruit juice was examined by administering it to male Sprague– Dawley rats each day at a dose of 1 ml/150 g body weight for 4 weeks before diabetic induction with alloxan (Horsfal et al., 2008). Blood glucose levels increased after induction, but then decreased significantly due to M. citrifolia juice prophylaxis against the diabetogenic drug alloxan (Horsfal et al., 2008). For 10 days, induced steroid diabetic (diabetes type 2) female Wistar rats were given 1.8 and 3.6 ml/kg of Indian M. citrifolia commercial fruit juice orally. Dexamethasone significantly reduced blood glucose levels in diabetic rats compared to control rats. M. citrifolia fruit juice provided greater benefits than rosiglitazone at a higher oral dose (3.6 ml/kg, twice a day), but caused liver damage in rats (Puranik et al., 2013). On the other hand, streptozotocin-induced diabetic rats were regulated by fermented juice of South American M. citrifolia. Diabetic standard animals given the hypoglycemic drug glibenclamide, as well as diabetic experimental animals given 2 ml/kg M. citrifolia twice a day for 20 days, had blood glucose levels of 125 mg/dl and 150 mg/dl, respectively, when compared with untreated diabetic rats with fasting blood sugar of 360.0 mg/dl. Nayak et al. (2011) found that saponins and flavonoids in M. citrifolia fruit, such as rutin, may act as secretagogues by increasing insulin production. M. citrifolia has shown promise in vivo findings as a natural antidiabetic drug, but scientists should be concerned about its toxicity, especially in the liver. Preclinical trials will be recommended if the doses are modified within a reasonable level with proven efficacy.
7 Industrial applications of the genus Morinda
7.1 Nutrition and Morinda
Food is a valuable natural resource, a basic human requirement, and a national treasure. Several Morinda species are used as food supplements or as drinks. M. citrifolia L., also known as cheese fruit or noni, is mostly consumed by people in the Pacific region, Polynesians, America, and Mengkudu in Malaysia, and its juice has been approved as a commercial food as a dietary supplement by the European Union (Yang et al., 2009). In the United States, the juice derived from the fruit is sold as a dietary supplement under the name “Noni” (Sina et al., 2021). The polysaccharides recovered from noni fruit are acidic heteropolysaccharides with galacturonic acid, galactose, rhamnose, and arabinose as monosaccharide components. Noni fruit juice was discovered to extend the lives of mice injected with Lewis lung cancer. It has recently become popular in Taiwan as a dietary supplement (Lin et al., 2007).
Morinda is widely used in Asia as a healthcare products for nutritious soups and drinks (Luo et al., 2021). Teas made from the leaves are used as a general febrifuge and a tonic for young children when taken orally (Lawal et al., 2012). Also, noni juice is commonly used nowadays as a nutritional supplement or meal for the alleged prevention of ailments like as diabetes, high blood pressure, and arteriosclerosis, and is commonly made as a drip/exudate from senescing fruits stored in fermentation vessels. The fruits of M. citrifolia L. are typically utilized to manufacture beverage products due to its distinctive flavor and health benefits. Various ethnic groups have used the fresh leaves of M. citrifolia L. as a vegetable or traditional medicine.
Noni fruit juice, both fermented and unfermented, has become a popular nutritional supplement in recent years, with health claims based on some of its constituents, notably flavonoids (Deng et al., 2007; Takashima et al., 2007). There have been several reports of hepatotoxicity of noni juice in humans between 2005 and 2011, raising health concerns, including hepatitis (Yu et al., 2013). However, this concern is still debated, as no causal link between liver injury cases and juice consumption has been documented (European Food Safety Authority, 2006). Noni juice is traditionally made by fermenting noni fruits in sealed barrels, glasses, or other vessels for 10 to 2 months, then collecting the juice that drips from the fruits, pasteurizing (or not), and bottling it (Nelson, 2006). Noni powder is prepared by dehydrating noni pulp or whole noni fruits in a dehydrator or outside in the sun. The impact of noni juice on enhancing endurance was proven in two clinical studies on athletes and postmenopausal women (Langford et al., 2004; Palu et al., 2008). The extensive use of Morinda products as food or supplements necessitated the urgent validation or authentication of its health effects for quality control purposes.
7.2 Nanotechnology and Morinda
Nanotechnology is a field of research and technology that focus on building 'matters' on the atomic or molecular level, such as materials and gadgets. To our knowledge, there are only a few published articles on the use of Morinda species in the synthesis of nanoparticles. The fruit extract of M. citrifolia L. was employed in the sol–gel process to synthesize CeO2-NPs. In the synthesis of CeO2-NPs, the extracts served as a weak base source, as well as oxidizing and capping agents. Plant based materials such as Morinda species have been encapsulated into chitosan nanoparticle (CHs NPs) via several methods such as precipitation, micro-emulsion, ionic gelation and organic solvent evaporation. This network improved drug stability by allowing for regulated release at the medication's impact site.
7.3 Textile and Morinda
The root of M. citrifolia is used in local textile industry in the production of local dye (yellow or red) (Siddiqui et al., 2006). Similarly, M. pandurifolia leaves, bark, and wood produce a yellowish-red pigment that can be used to colour garments. It's also used to manufacture the morindone dye that goes by the brand name “Suranji” in India. Morindone is used to dye cotton, silk, and wool in red, chocolate, and purple hues. When the plant is three to four years old, the coloring matter is gathered primarily from the root bark. Morindin is the active ingredient isolated as a glucoside, which when hydrolyzed produces the dye. With an aluminium mordant, it produces a yellowish-red color, chocolate with a chromium mordant, and dull purple to black with an iron mordant. The principal colorant was identified as morindone, a red-colored anthraquinone that has been used as a traditional dye for decades, and was derived from the roots of M. tinctoria.
7.4 Metallurgy and Morinda
In hydrochloric and sulphuric acid, M. tinctoria extract efficiently prevented mild steel corrosion.
7.5 Reagents and Morinda
This magical fruit has been used in the production of several chemicals or reagents such as anthraquinone flavonol glycosides, iridoid glycosides, lipid glycosides, and triterpenoids (Liu et al., 2018).
7.6 Natural preservative and Morinda
A natural preservative identified as butylated hydroxytoluene was isolated from M. citrifolia and was efficient in inhibiting warmed-over flavor in formerly stewed beef pies, via reduction of lipid oxidation, improving color stability and the shelf life of the final aerobically wrapped pies (Nathan et al., 2012). It has been proven to be more efficient in reducing various phytopathogenic fungus, including Olivea neotectonae, the causal agent of rust in teak and Stagonosporopsis cucurbitacearum (Sc).
8 Conclusion and future prospects
Therapeutic plants have been used in treatment of diseases since time immemorial. Plants have played significant roles in drug discovery and serve as lead in unearthing drugs with pronounced health benefits. Nature has given man a structured, complex and functional bioresource with assortment of potentials in ameliorating diseases and ailments. Plant-based (phytochemical) research has made notable innovations precisely in the field of antimalarial (artemisinin compounds) or anticancer (flavonoids) treatments. In spite of this, usage of herbal products decreases with more significance directed towards synthetic drugs or repurposed drugs. One of the well explored genus in Asia, Africa, Europe, Latin America and some part of Europe is Morinda. The genus has played immeasurable role in treating numerous diseases such as malaria, diabetes, inflammation, cancer, cholera, memory loss and gastrointestinal infection. This review highlights the traditional uses, phytochemical profile, pharmacology and industrial applications of Morinda. The secondary metabolites reported include glycosides, anthraquinones, alkaloids, reducing sugars, phenolic compounds, terpenoids, steroids, mono- or poly- saccharides and saponins. These metabolites contribute to marked pharmacological activities exhibited by this genus.
Nevertheless, there are still a number of details that require further research to fill the present-day knowledge gap. There are reports of secondary metabolites diversity in plants is influenced by geographical distribution, adaptation to climatic conditions, extraction procedure, solvent polarity and preparation methods. Thus, it is crucial to elucidate these effects via in vitro or in vivo pharmacological assays. The understanding on chemical diversity will enhance documentation of relevant biomarker compounds with significant therapeutic potency. More studies into the antimicrobial, wound healing, antidiabetic, anxiolytic, antinociceptive, antiplasmodial, anti-inflammatory, antioxidant and antipyretic effects needs to be place in order via in vivo and in vitro experimental models. More research should be focused on developing processes that might aid substantiate the genus therapeutic effects and toxicity. Thus, isolation and clinical trials should be thoroughly investigated, as they may serve as a lead to the discovery of potent pharmacophores, satisfying scientists' biotechnological craves for innocuous therapeutics to address existing health challenges.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests that could have appeared to influence the work reported in this paper.
References
- ACS, (American Cancer Society), 2015. Cancer facts and figures. www.cancer.org (accessed 3 October 2018).
- Phytochemical screening and antimicrobial study on the leaves of Morinda lucida (Rubiaceae) J. Nat. Sci. Res.. 2013;3:131-136.
- [Google Scholar]
- Cysteine-stabilised peptide extract of Morinda lucida (Benth) leaf exhibits antimalarial activity and augments antioxidant defense system in Plasmodium berghei-infected mice. J. Ethnopharmacol.. 2017;207:118-128.
- [Google Scholar]
- Morinda lucida leaf and fruit extracts may attenuate diarrhea arising from ebola. Basic Res. J. Med. Clin. Sci.. 2015;4:95-100.
- [Google Scholar]
- Traditional and medicinal uses of Morinda lucida. J. Med. Plants Stud.. 2018;6:249-254.
- [Google Scholar]
- Oruwal, a novel dihydroanthraquinone pigment from Morinda lucida Benth. J. Chem. Soc., Chem. Commun.. 1972;405–406
- [Google Scholar]
- Antibacterial activity of aqueous and ethanol crude extracts of the root barks of Alstonia boonei and preliminary phytochemical test of Morinda lucida. J. Med. Plants. 2010;4:644-648.
- [Google Scholar]
- Evaluation of Nigerian traditional medicines : 1. Plants used for rheumatic (inflammatory) disorders. J. Ethnopharmacol.. 1994;42:179-182.
- [Google Scholar]
- Anti-inflammatory and potential cancer chemoprevebtive constituents of the fruits of Morinda citrifolia (Noni) J. Nat. Prod.. 2007;70:754-757.
- [Google Scholar]
- Anti-anaemia potential and safety of Morinda lucida leaf extracts in balb/c mice induced with rhabdomyosarcoma cells anti-anaemia potential and safety of Morinda lucida leaf extracts in balb/c mice induced with rhabdomyosarcoma cells. Afr. J. Environ. Heal. Sci.. 2017;4:61-68. F, A.D.
- [Google Scholar]
- Preliminary phytochemical investigations and evaluation of anti -microbial activity of methanol extract of the leaves of Morinda lucida Benth (Rubiaceae) J. Pharm. Res.. 2013;2:1-5.
- [Google Scholar]
- Antiproliferative, antioxidant activities and apoptosis induction by Morinda lucida and Taraxacum officinale in human HL-60 leukemia cells. J. Glob. Biosci.. 2016;5:4281-4291.
- [Google Scholar]
- Tumor cell-selective antiproliferative effect of the extract from Morinda citrifolia fruits. Phytother. Res.. 2006;20(6):515-517.
- [Google Scholar]
- Ethnobotanical survey and cytotoxicity testing of plants of South-western Nigeria used to treat cancer, with isolation of cytotoxic constituents from Cajanus cajan Millsp. leaves. J. Ethnopharmacol.. 2010;128:501-512.
- [Google Scholar]
- Effect of petroleum ether fractions of Morinda lucida on Plasmodium berghei berghei in mice. Pharm. Biol.. 1998;36:301-304.
- [Google Scholar]
- Two new 11-noriridoids from the aerial parts of Morinda umbellate. Phytochem. Lett.. 2013;6:267-269.
- [Google Scholar]
- The potential of anti-malarial compounds derived from African medicinal plants: a review of pharmacological evaluations from 2013 to 2019. Malar. J.. 2020;19:183.
- [Google Scholar]
- Iridoids with anti-inflammatory effect from the aerial parts of Morinda officinalis How. Fitoterapia. 2021;153:104991
- [Google Scholar]
- Chan-Blanco, Yanine, Vaillant, Fabrice, Perez, Ana Mercedes, Reynes, Max, Brillouet, Jean-Marc, Brat, Pierre, 2006. The noni fruit (Morinda citrifolia L.): a review of agricultural research, nutritional and therapeutic properties. J. Food Compos. Anal. 19(6-7), 645–654.
- Observation of the effect of Morinda offcinalis How on rat rheumatoid arthritis. Mil. Med. J. Southeast China. 2011;13:305-309.
- [Google Scholar]
- Chemical constituents of Morinda officinalis. Yunnan. J. Tradit. Chin. Med. Mater. Med.. 2009;15:63-64.
- [Google Scholar]
- Puerarin improves metabolic function leading to hepatoprotective effects in chronic alcohol-induced liver injury in rats. Phytomedicine. 2013;20:849-852.
- [Google Scholar]
- Anti-malarial synergy of secondary metabolites from Morinda lucida Benth. J. Ethnopharm.. 2017;199:91-96.
- [Google Scholar]
- Antinociceptive anti-inflammatory effect of monotropein isolated from the root of Morinda officinalis. Biol. Pharm. Bull.. 2005;10:1915-1918.
- [Google Scholar]
- Chokchaisiri, S., Siriwattanasathien, Y., Thongbamrer, C., Suksamrarn, A., Rukachaisirikul, T., 2019. Morindaquinone, a new bianthraquinone from Morinda coreia roots, Nat. Product Res. DOI: 10.1080/14786419.2019.1705820.
- Antibacterial, anti-inflammatory and antimalarial activities of some Nigerian medicinal plants. South Afr. J. Bot.. 2005;71:316-325.
- [Google Scholar]
- Complement inhibiting iridoids from Morinda morindoide. J. Nat. Prod.. 2003;66(1):97-102.
- [Google Scholar]
- Antiamoebic activity of iridoids from Morinda morindoides leaves. Planta Med.. 2007;73:849-850.
- [Google Scholar]
- Study on antidepressant activity of Morinda officinalis in the ingredients of Chinese medicine. China J. Chin. Mater. Med.. 1995;20:36-39.
- [Google Scholar]
- Noni as an anxiolytic and sedative: a mechanism involving its gamma-aminobutyric acidergic effects. Phytomed.. 2007;14(7–8):517-522.
- [Google Scholar]
- Macroinvertebrate colonization and breakdown of leaves in an astatic pond in south India. J. Environ. Biol.. 2008;29:249-252.
- [Google Scholar]
- Characterization, anti-oxidative and anti-inflammatory effects of Costa Rican noni juice (Morinda citrifolia l.) J. Ethnopharmacol.. 2011;133, 1:108-115.
- [Google Scholar]
- A new anthraquinone from Morinda citrifolia roots. Nat. Prod. Res.. 2009;23:1322-1329.
- [Google Scholar]
- A new anthraquinone from Morinda citrifolia roots. Nat. Prod. Res.. 2009;23(14):1322-1329.
- [Google Scholar]
- New anthraquinone derivatives from the stem barks of Morinda lucida Benth. Phytochem. Lett.. 2020;39:94-98.
- [Google Scholar]
- Morinda lucida reduces contractility of isolated uterine smooth muscle of pregnant and non-pregnant mice. Niger. J. Physiol. Sci.. 2007;22:129-134.
- [Google Scholar]
- Potential antitumor activity of nonsteroidal anti-inflammatory drugs against Ehrlich ascites carcinoma in experimental animals. Int. J. Health Sci. (Qassim). 2019;13:11-17.
- [Google Scholar]
- Cognitive-enhancing properties of Morinda lucida (Rubiaceae) and Peltophorum pterocarpum (Fabaceae) in scopolamine-induced amnestic mice. Afr. J. Tradit. Complementary Altern. Med.. 2017;14:136-141.
- [Google Scholar]
- Acetyl and butyrylcholinesterase inhibiting constituent from Morinda lucida Benth (Rubiaceae) Br. J. Pharmaceut. Res.. 2015;6:358-365.
- [Google Scholar]
- Cholinesterase inhibitory activity of Morinda lucida. J. Med. Plants Res. 2013;7:734-737.
- [Google Scholar]
- Morinda lucida extract induces endothelium- dependent and -independent relaxation of rat aorta. Fitoterapia. 2004;75:332-336.
- [Google Scholar]
- Possible antimicrobial activity of Morinda lucida stem bark, leaf and root extracts. Afr. J. Biotechnol.. 2014;13:471-475.
- [Google Scholar]
- Assaying polysaccharide in couplet drugs of Bajitian (Radix Morindae Officinalis) and Duzhong (Cortex Eucommiae) and its effects on the proliferation of rat chondrocyte. Rheum. Arthritis. 2014;3:23-26.
- [Google Scholar]
- Antitumour potential of a polysaccharide-rich substance from the fruit juice of Morinda citrifolia (noni) on sarcoma 180 ascites tumour in mice. Phytother. Res.. 2003;17(10):1158-1164.
- [Google Scholar]
- Induction of mitochondrial-mediated apoptosis by Morinda citrifolia (noni) in human cervical cancer cells. Asian Pac. J. Cancer Prev.. 2013;14(1):237-242.
- [Google Scholar]
- Induction of normal phenotypes in ras-transformed cells by damnacanthal from Morinda citrifolia. Cancer Lett.. 1993;30(73):161-166.
- [Google Scholar]
- Anti diabetic effect of fruit juice of Morinda citrifolia (Tahitian Noni Juice) on experimentally induced diabetic rats. Nigerian J. Health Biomed. Sci.. 2008;7(2):34-37.
- [Google Scholar]
- Ethnobotanical survey of antimalarial plants used in Ogun State. Southwest Nigeria. Afr. J. Pharmacy Pharmacol.. 2010;4:55-60.
- [Google Scholar]
- Antibacterial, antifungal and tumor cell suppression potential of Morinda citrifolia fruit extracts. Int. J. Integr. Biol.. 2008;3(1):44.
- [Google Scholar]
- Cell-based chemical genetic screenidentifies damnacanthal as an inhibitor of HIV-1 Vpr induced cell death. Biochem. Biophys. Res. Commun.. 2006;348(3):1101-1106.
- [Google Scholar]
- Inhibitory effect of anthraquinones isolated from the Noni (Morinda citrifolia) root on animal A-, Band Y-families of DNA polymerases and human cancer cell proliferation. Food Chem.. 2010;118(3):725-730.
- [Google Scholar]
- New anthraquinone glycosides from the roots of Morinda citrifolia. Fitoterapia. 2009;80:196-199.
- [Google Scholar]
- Inhibitory effect of anthraquinones isolated from the Noni (Morinda citrifolia) root on animal A-, B- and Y-families of DNA polymerases and human cancer cell proliferation. Food Chem.. 2010;118:725-730.
- [Google Scholar]
- Chem. Pharm. Bull.. 2005;53:1597.
- Study on the constituents of aerial part of Morinda parvifolia. Zhongyaocai.. 2017;40:111-113.
- [Google Scholar]
- Naphthohydroquinones, naphthoquinones, anthraquinones, and a naphthohydroquinone dimer isolated from the aerial parts of Morinda parvifolia and their cytotoxic effects through up-regulation of p53. Phytochem.. 2016;130(2016):144-151.
- [Google Scholar]
- Kiazolu, J.B., Intisar, A., Zhang, I., Wang, Y., Zhang, R., Wu, Z., Zhang, W., 2016. Phytochemical screening and chemical variability in volatile oils of aerial parts of Morinda morindoides, Nat. Product Res., DOI: 10.1080/14786419.2016.1154058.
- Identification of novel fatty acid glucosides from the tropical fruit Morinda citrifolia L. Phytochem. Lett.. 2010;3:238-241.
- [Google Scholar]
- In-vitro and in-vivo anti-inflammatory and antinociceptive effects of the methanol extract of the roots of Morinda officinalis. J. Pharm. Pharamcol.. 2005;57:607-615.
- [Google Scholar]
- Effects of three compounds extracted from Morinda lucida on Plasmodium falciparum. Planta Med.. 1992;58:533-534.
- [Google Scholar]
- Effects of the compounds extracted from Morinda lucida on Plasmodium falciparum. Planta Med.. 1992;58:533-534.
- [Google Scholar]
- Effects of Morinda citrifolia on quality of life and auditory function in postmenopausal women. J. Altern. Complement. Med.. 2004;10(5):737-739.
- [Google Scholar]
- Ethnomedicinal and Pharmacological properties of Morinda lucida. J. Nat. Prod.. 2012;5:93-99.
- [Google Scholar]
- Cytotoxic quinones from the aerial parts of Morinda umbellata L. Phytochem.. 2019;167:112096
- [Google Scholar]
- Chemical constitution of the roots from Morinda officinalis. J. South-Cent. Univ. Natl. (Nat. Sci. Ed.). 2010;29:53-55.
- [Google Scholar]
- Inhibitory effects of Morinda officinalis extract on bone loss in ovariectomized rats. Molecules. 2009;14:2049-2061.
- [Google Scholar]
- Effect of Radix Morinda officnalis polysaccharide on expression of Cbfa1mRNA in osteoblast cultured in vitro. China J. Traditional Chinese Med. Pharm.. 2007;22:517-519.
- [Google Scholar]
- Active anti-aging constituents from Morinda officinalis How. Cent. South Pharm.. 2010;9:101-102.
- [Google Scholar]
- The cytoprotective effect of inulin-type hexasaccharide extracted from Morinda officinalis on PC12 cells against the lesion induced by corticosterone. Life Sci.. 2004;75:1531-1538.
- [Google Scholar]
- Lignans and anthraquinones from the fruits of Morinda citrifolia. Nat. Product Res.. 2007;21(13):1199-1204.
- [Google Scholar]
- Study situation and prospects of the research on Morinda officinalis. Subtrop. Plant. Sci.. 2010;39:74-78.
- [Google Scholar]
- Two novel glycosides from the fruits of Morinda citrifolia (noni) inhibit AP-1 transactivation and cell transformation in the mouse epidermal JB6 cell line. Cancer Res.. 2001;61(15):5749-5756.
- [Google Scholar]
- Effect of polysaccharides from Morinda offcinalis on the oxidative stress and congitive-behaviors in an experimental depression model of rats. China Mod. Dr.. 2011;49:2-5.
- [Google Scholar]
- Anthraquinones from Morinda officinalis roots enhance adipocyte differentiation in 3T3-L1 cells. Natural Product Res.. 2012;26(18):1750-1754.
- [Google Scholar]
- A new pair of enantiomeric lignans from the fruits of Morinda citrifolia and their absolute configuration. Nat. Prod. Res.. 2018;32(8):933-938.
- [Google Scholar]
- Anthraquinones: analytical techniques as a novel tool to investigate on the triggering of biological targets. Curr. Drug Targets. 2011;12:366-380.
- [Google Scholar]
- Development and application of high-performance liquid chromatography for the study of two new oxyprenylated anthraquinones produced by Rhamnus species. J. Chromatogr. A. 2012;1225:113-120.
- [Google Scholar]
- Effect of medicinal indianmulberry root in the treatment of postmenopausal osteoporosis. China Med. Pharm.. 2013;20:80-81.
- [Google Scholar]
- Loonjang, K., Duangjinda, D., Phongpaichit, S., Sawangjaroen, N., Rattanaburi, S., Mahabusarakam, W., 2015. A new anthraquinone from Morinda elliptica Ridl., Natural Prod. Res., DOI: 10.1080/14786419.2015.1009062.
- Anti-inflammatory naphthoates and anthraquinones from the roots of Morinda officinalis. Bioorg. Chem.. 2021;110:104800
- [Google Scholar]
- Effects of Morinda citrifolia aqueous fruit extract and its biomarker scopoletin on reflux esophagitis and gastric ulcer in rats. J. Ethnopharmacol.. 2011;134(2):243-250.
- [Google Scholar]
- Screening of Morinda lucida leaf extract for antimalarial action on Plasmodium berghei berghei in mice. Afr. J. Med. Med. Sci.. 1985;14:59-63.
- [Google Scholar]
- Isolation and selective glycosylation of antisalmonellal anthraquinones from the stem bark of Morinda lucida Benth. (Rubiaceae) Phytochem. Lett.. 2020;37:80-84.
- [Google Scholar]
- Evaluation of Morinda citrifolia as an endodontic irrigant. J. Endod.. 2008;34(1):66-70.
- [Google Scholar]
- Anti-typhoid potentials of crude and fractions of defeated chloroform extract of Morinda lucida leaves. Int. J. Appl. Biol. Res.. 2014;6:74-82.
- [Google Scholar]
- Noni puree (Morinda citrifolia) mixed in beef patties enhanced color stability. Meat Sci.. 2012;91(2):131-136.
- [Google Scholar]
- Nelson, Scot C., 2006. Species profiles for Pacific island agroforestry Morinda citrifolia noni. In: Elevitch, Craig R. (Ed.). Permanent Agriculture Resources <http://www.traditionaltree.org/>.
- African Traditional Medicine. A Dictionary of Plant Use and Applications. With Supplement: Search System for Diseases. Stuttgarts: Medipharm Scientific Publishers; 2000. p. :343-344.
- Anthraquinones from Morinda longissima and their insulin mimetic activities via AMP-activated protein kinase (AMPK) activation. Bioorg. Med. Chem. Lett.. 2017;27:40-44.
- [Google Scholar]
- Antioxidant potential of Morinda pubescens Sm. fruits. J. Pharm. Res.. 2011;4:829-831.
- [Google Scholar]
- Damnacanthal-induced anti-inflammation is associated with inhibition of NFB activity. Inflamm. Allergy - Drug Targets. 2011;10:455-463.
- [Google Scholar]
- In vitro anti-trypanosomal activity of Morinda lucida leaves. Afr. J. Biotechnol.. 2012;11:1812-1817.
- [Google Scholar]
- Supplementation with aqueous leaf extract of Morinda lucida enhances immunorestoration and upregulates the expression of cytokines and immunostimulatory markers. Immunol. Invest.. 2012;41:799-819.
- [Google Scholar]
- Investigation of various extracts of Morinda lucida for antimalarial actions on Plasmodium berghei berghei in mice. Afr. J. Med. Med. Sci.. 1985;14:45-49.
- [Google Scholar]
- Anti-diabetic effect of Morinda lucida stem Bark extract on alloxan-induced diabetic rats. Res. J. Pharmacol.. 2010;4(8):78-82.
- [Google Scholar]
- The antimicrobial activity of Morinda lucida leaf extract on Escherichia coli. J. Med. Plants Res.. 2009;3:319-323.
- [Google Scholar]
- Biochemical characterisation of the leaf of Morinda lucida: Prospects for environmentally-friendly steel-rebar corrosion-protection in aggressive medium. Miner. Met. Mater. Soc. Annu. Meet. Suppl. Proc. 2015:637-643.
- [Google Scholar]
- Composition and antioxidant activities of leaf and root volatile oils of Morinda lucida. Nat. Prod. Commun.. 2011;6:1537-1541.
- [Google Scholar]
- The genus Senna (Fabaceae): A review on its traditional uses, botany, phytochemistry, pharmacology and toxicology. South African J. Botany. 2021;138:1-32.
- [Google Scholar]
- Natural Products as Sources of Antimalarial Drugs: Ethnobotanical and Ethnopharmacological Studies. Scientifica. 2020;7076139
- [Google Scholar]
- Antiplasmodial activity of Morinda lucida Benth. Leaf and bark extracts against Plasmodium berghei infected mice. Saudi J. Biological Sci.. 2022;29, 4:2475-2482.
- [Google Scholar]
- Stellaria media (L.) Vill.- A plant with immense therapeutic potentials: phytochemistry and pharmacology. Heliyon. 2020;6, 6:e04150
- [Google Scholar]
- Evaluation of the antidiabetic property of Morinda lucida leaves in streptozotocin-diabetic rats. J. Pharm. Pharmacol.. 1999;51:1321-1324.
- [Google Scholar]
- The feeding value of Dialium guineense as a supplement to West African dwarf sheep fed natural grass hay. Anim. Res. Dev.. 1999;49:24-31.
- [Google Scholar]
- Insecticidal activity and chemical composition of the Morinda lucida essential oil against pulse beetle Callosobruchus maculatus. Sci. World J. 2014:1-8.
- [Google Scholar]
- Protective effect of fruits of Morinda citrifolia L. on scopolamine induced memory impairment in mice: a behavioral, biochemical and cerebral blood flow study. J. Ethnopharmacol.. 2012;139, 1:34-41.
- [Google Scholar]
- Wound healing effects of noni (Morinda citrifolia L.) leaves: a mechanism involving its PDGF/A2A receptor ligand binding and promotion of wound closure. Phytother. Res.. 2010;24, 10:1437-1441.
- [Google Scholar]
- The effects of Morinda citrifolia l. (noni) on the immune system: Its molecular mechanisms of action. J. Ethnopharmacol.. 2008;115, 3:502-506.
- [Google Scholar]
- Noni seed oil topical safety, efficacy, and potential mechanisms of action. J. Cosmet., Dermatol. Sci. Appl.. 2012;2, 2:74-78.
- [Google Scholar]
- An anthraquinone with potent quinone reductase-inducing activity and other constituents of the fruits of Morinda citrifolia (Noni) J. Nat. Prod.. 2005;68:1720-1722.
- [Google Scholar]
- Morinda citrifolia (Noni) fruit–phytochemistry, pharmacology, safety. Planta Med.. 2007;73:191-199.
- [Google Scholar]
- Preclinical evaluation of antidiabetic activity of noni fruit juice. Int. J. Bioassays.. 2013;2(2):475-482.
- [Google Scholar]
- The inulintype oligosaccharides extract from Morinda officinalis, a traditional Chinese herb, ameliorated behavioral deficits in an animal model of post-traumatic stress disorder. Metab. Brain Dis.. 2016;31:1143-1149.
- [Google Scholar]
- CRC World Dictionary of Plant Names. III: M-Q. CRC Press; 2000. p. :1730.
- Anthraquinones and an iridoid glycoside from the roots of Morinda pandurifolia. Biochem. Systematics and Ecol.. 2011;39:888-892.
- [Google Scholar]
- Traditional uses, bioactive composition, pharmacology, and toxicology of Phyllanthus emblica fruits: A comprehensive review. J, Ethnopharm.. 2022;282:114570
- [Google Scholar]
- Molecular bases underlying the hepatoprotective effects of coffee. Nutrients.. 2017;9:85.
- [Google Scholar]
- Taxonomy of Angiosperms. International Pvt Ltd: I. K; 2005. p. :404.
- Effect of Morindae radix extracts on experimental osteoporosis in sciatic neurectomized mice. Phytother. Res.: PTR. 2005;19:231-238.
- [Google Scholar]
- Effect of different processed products and extracts of Morinda officinalis root on aajuvant-induced arthritis in rats. J. Chin. Med. Mater.. 2015;38:1626-1629.
- [Google Scholar]
- Monotropein isolated from the roots of Morinda officinalis ameliorates proinflammatory mediators in RAW 264.7 macrophages and dextran sulfate sodium (DSS)-induced colitis via NF-kappaB inactivation. Food Chem. Toxicol.: Int. J. Publ. Br. Ind. Biol. Res. Assoc.. 2013;53:263-271.
- [Google Scholar]
- Isolation and structure determination of two new constituents from the fruits of Morinda citrifolia Linn. Nat. Prod. Res.. 2008;22:1128-1136.
- [Google Scholar]
- New anthraquinones from the stem of Morinda citrifolia Linn. Nat. Prod. Res.. 2006;20(12):1136-1144.
- [Google Scholar]
- Phytochemical composition and in vitro biological activities of Morinda citrifolia fruit juice. Saudi J. Biol. Sci.. 2021;28:1331-1335.
- [Google Scholar]
- Toxicity and mutagenic activity of some selected Nigerian plants. J. Ethnopharmacol.. 2007;113:427-432.
- [Google Scholar]
- Multipleberrytypes preventn-nitrosomethylbenzylamine-induced esophageal cancer in rats. Pharm. Res.. 2010;27(6):1138-1145.
- [Google Scholar]
- J. Nat. Prod.. 2005;68:592.
- Selective cox-2 inhibition of Morinda citrifolia (Noni) in vitro. Nashville, Tennessee, USA: Loews Vanderbilt Plaza; 2001.
- Eight new glycosides with hepatoprotective activity isolated from the aerial parts of Morinda parvifolia. Bioorganic Chem.. 2019;87:867-875.
- [Google Scholar]
- Glycosides of naphthohydroquinones and anthraquinones isolated from the aerial parts of Morinda parvifolia Bartl. ex DC (Rubiaceae) increase p53 mRNA expression in A2780 cells. Phytochem.. 2018;152:97-104.
- [Google Scholar]
- Anti-inflammatory glycosides isolated from the aerial parts of Morinda parvifolia. Phytochem. Lett.. 2019;33:55-60.
- [Google Scholar]
- New anti-trypanosomal active tetracyclic iridoid isolated from Morinda lucida Benth. Bioorg. Med. Chem. Lett.. 2015;25:3030-3033.
- [Google Scholar]
- A brief review on noni (Morinda citrifolia L.) – a herbal remedy for better health. Glob. J. Res. Med. Plants Indig. Med.. 2013;2:337-347.
- [Google Scholar]
- New constituents from the leaves of Morinda citrifolia. Chem. Pharm. Bull.. 2007;55:343-345.
- [Google Scholar]
- Apoptosis-inducing effects of Morinda citrifolia L. and doxorubicin on the Ehrlich ascites tumor in Balb-c mice. Cell Biochem. Funct.. 2009;27, 8:542-546.
- [Google Scholar]
- Antiplasmodial efficacy of methanolic root and leaf extracts of Morinda lucida. J. Nat. Sci. Res.. 2013;3:112-123.
- [Google Scholar]
- Morinda officialis How extract improves microwave-induced reproductive impairment in male rats. Zhonghua Nanke Xazhi. 2013;19:340-345.
- [Google Scholar]
- Two new anthraquinones with antiviral activities from the barks of Morinda citrifolia (Noni) Phytochem. Letters. 2016;15:13-15.
- [Google Scholar]
- Morinda citrifolia (Noni): A literature review and recent advances in Noni research. Acta Pharmacol. Sin.. 2002;23:1127-1141.
- [Google Scholar]
- Morinda citrifolia (Noni): a literature review and recent advances in noni research. Acta Pharmacol. Sin.. 2002;23:1127-1141.
- [Google Scholar]
- Wee, Y.C., 1992. A Guide to Medicinal Plants. Singapore Science Center.
- Antimicrobial activity of an iridoid rich extract from Morinda citrifolia fruit. Curr. Res. J. Biol. Sci.. 2012;4(1):52-54.
- [Google Scholar]
- WHO, 2015. World Malaria Report.
- Wong, D.K., 2004. Are immune responses pivotal to cancer patient’s long term survival? Two clinical case-study reports on the effects of Morinda citrifolia (noni). Hawaii Med. J. 63(6), 182–184.
- Morinda citrifolia (Noni): a literature review and recent advances in Noni research. Acta. Aeologica Sin.. 2002;23:1127-1141.
- [Google Scholar]
- Influence on free amino acids constituents of Morindae officinalis How processed by common methods. Sci. Technol. Food Ind.. 2009;19:105-107.
- [Google Scholar]
- Antiosteoporotic activity of anthraquinones from Morinda officinalis on osteoblasts and osteoclasts. Molecules. 2009;14:573-583.
- [Google Scholar]
- An experimental study on the effect of Morinda-achyranthes alcohol extract on the expression of Gl/s cyclin and gene in cartilage cells of SD rats. Rheum. Arthritis. 2017;25:5-8.
- [Google Scholar]
- Antimicrobial anthraquinones from Morinda angustifolia. Fitoterapia. 2008;79(7–8):501-504.
- [Google Scholar]
- Study on the mechanism of antidepressant oligosaccharides. Chin. J. Clin. Pharmacol.. 2015;31:1539-1542.
- [Google Scholar]
- Chemical constituents from the seeds of Morinda citrifolia. Chinese J. Nat. Med.. 2009;7(2):119-122.
- [Google Scholar]
- Effect of Morinda officinalis extract on learning and memory impairment in mice. J. Guangzhou Med. Univ.. 2015;43:23-26.
- [Google Scholar]
- Methyl 2-naphthoates from a traditional Chinese herb Morinda officinalis var. officinalis. Tetrahedron. 2019;75:3793-3801.
- [Google Scholar]
- Effect of MOO on Large conductance calcium-activated potassium channels (BKca)in ECV304 cell proliferation. Lishizhen Med. Mater. Med. Res.. 2013;24:123-126.
- [Google Scholar]
- Zengin, G., Locatelli, M., Ceylan, R., Aktumsek, A., 2015. Anthraquinone profile, antioxidant and enzyme inhibitory effect of root extracts of eight Asphodeline taxa from Turkey: can Asphodeline roots be considered as a new source of natural compounds? J. Enzym. Inhib. Med. Chem. doi:http://dx.doi.org/10.3109/14756366.2015.1063623.
- Structural characterization and anti-fatigue activity of polysaccharides from the roots of Morinda officinalis. Int. J. Biol. Macromol.. 2009;44:257-261.
- [Google Scholar]
- Chin. J. Nat. Med.. 2010;8:192-195.
- Zhang, Hong-Cai, Wang, Yu, Yu, Chun-Miao, Yu, Dong-Hua, Chen, Ping-Ping, Liu, Shu-Min, 2014. Two new saccharide fatty acid esters from the fruit of Morinda citrifolia L. and their ABTS radical scavenging activities. Rec. Nat. Prod. 8, 1, 25–31.
- Morinda officinalis How. - A comprehensive review of traditional uses, phytochemistry and pharmacology. J. Ethnopharmacol.. 2018;213:230-255.
- [Google Scholar]
- Effect of Morinda officinalis on expression of myosin and actin gene in aging rat myocardial cells induced by D-galactose. Chin. J. Gerontol.. 2011;31:4836-4839.
- [Google Scholar]
- Monotropein isolated from the roots of Morinda officinalis increases osteoblastic bone formation and prevents bone loss in ovariectomized mice. Fitoterapia. 2016;110:166-172.
- [Google Scholar]
- Effect of Morinda officinalia-containing serum on the expression of CA II and NFAT2 mRNA in osteoblast-osteoclast co-culture system. Chin. J. Osteoporos.. 2013;19:120-124.
- [Google Scholar]
- Protective effect of polysaccharides from Morinda officinalis on bone loss in ovariectomized rats. Int. J. Biol. Macromol.. 2008;43:276-278.
- [Google Scholar]







