Translate this page into:
High oleic safflower biolubricant through double transesterification with methanol and pentaerythritol: Production, characterization, and antioxidant addition
⁎Corresponding author. senogalesd@unex.es (S. Nogales-Delgado)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Abstract
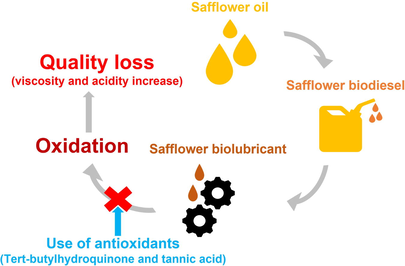
Biodiesel and biolubricant from high-oleic safflower were obtained. Quality parameters of these products were suitable. Oxidative stability of biolubricant was short, needing antioxidant addition. TBHQ had a positive effect and kept biolubricant properties during oxidation.
Abstract
Double transesterification from vegetable oils could play an important role in biodiesel and biolubricant production, with the possible implementation of biorefineries to replace refineries based on petroleum. The oxidative stability of the original sample will influence the quality of the intermediate and final products, recommending highly stable raw materials or the use of antioxidants to keep quality parameters during storage. The aim of this work was to obtain a stable biolubricant, assessing its production through a double transesterification with methanol and pentaerythritol from high-oleic safflower oil and adding antioxidants, paying attention to quality parameters. Consequently, a biorefinery that produced high-quality products was proposed. In conclusion, high biodiesel and biolubricant yields were obtained (>97 and >94%, respectively) with the following chemical conditions for the latter: FAME/alcohol ratio, 1:0.33; pressure, 260 mmHg; catalyst concentration, 1.0%; temperature, 160 °C. The oxidative stability of biodiesel complied with the standard (10.78 h) due to its high methyl oleate content (exceeding 80%), whereas this parameter was shorter for the biolubricant (2.86 h), possibly due to its molecular structure. Consequently, antioxidant addition was needed, and tert-Butylhydroquinone at low concentration (500 ppm) kept viscosity and acid number of high-oleic safflower biolubricant during oxidation conditions (up to 8 h). However, tannic acid did not keep these properties in biolubricant. In conclusion, by using the right antioxidant, all the products of the proposed biorefinery were stable during oxidizing conditions, making this biorefinery more competitive.
Keywords
Transesterification
Oxidative stability
Viscosity
Viscosity index
Acid number
Tert-butylhydroquinone
Tannic acid
- CFPP
-
Cold filter plugging point
- DTG
-
Derivative thermogravimetry
- FT-IR
-
Fourier transform infrared
- FAME
-
Fatty acid methyl ester
- HSO
-
High-oleic safflower oil
- HSBD
-
High-oleic safflower biodiesel
- HSBL
-
High-oleic safflower biolubricant
- TA
-
Tannic acid
- TBHQ
-
Tert-butylhydroquinone
- TG
-
Thermogravimetric analysis
Abbreviations
1 Introduction
Due to the role of energy (or material) market in the current geopolitical situation, as well as the consequences of unpredictable events that can affect or change national strategies, the energy transition is going to play a vital role in the future, which can alter the current economic status of many world powers (Overland et al., 2019) or make local economies more resistant to global crisis such as COVID-19 outbreak (Galanakis et al., 2022). This scenario might be a starting point (or opportunity) for the global integration of bioeconomy (Woźniak and Tyczewska, 2021). Thus, many countries have resorted to green strategies to carry out an energy transition in this post-pandemic era (Tian et al., 2022), promoting domestic feedstock production and advanced biofuel feedstock technology (Duarah et al., 2022). Apart from that, global concern about the environment is an important issue, increasing the scientific interest in the last decade in fields such as bioenergy, among others (Welfle et al., 2020), and implying the implementation of local and global policies, with Sustainable Development Goals (established by the United Nations) being the most important exponent of this trend (United Nations, 2015). Many of these goals have to do with sustainability, poverty reduction, pollution decrease, etc. The use of biofuels or biomaterials could play an important role, complying with many of these requirements in the long run. Specifically, biodiesel and biolubricant production could be a feasible and sustainable replacement for their equivalents from oil or petroleum, as many studies have previously pointed out. Thus, biodiesel production has been widely studied, applying approaches such as exergy analysis, from the efficiency, productivity and sustainability perspectives, in order to optimize the process (Aghbashlo et al., 2018), with specific examples such as the optimization of biodiesel production from waste cooking oil in a ultrasonic reactor (Aghbashlo et al., 2021a). This technique, as well as other tools such as life cycle analysis, can be promising to assess biofuel production from thermodynamic, economic and environmental perspectives (Rosen, 2018).
On the other hand, design, optimization, monitoring and control of biodiesel production are important, and Data-driven machine learning techniques have demonstrated to be effective to modeling such complex processes (Aghbashlo et al., 2021b).
Biodiesel and biolubricants usually share many advantages, like the following (McNutt and He, 2016; Rodionova et al., 2017): a) Use of green feedstock, such as biomass; b) zero CO2 net emissions, especially concerning biodiesel; c) production of biodegradable compounds; d) sustainable economic growth of developing areas; e) promotion of green processes related to circular and local economy.
Biodiesel and biolubricant production can be variable, especially for the latter, where dehydrogenation, epoxidation or transesterification, among others, can be used (do Valle et al., 2018; Owuna et al., 2020). For instance, many studies have covered different kinds of biolubricants, changing both the raw material, alcohol and technique used, as in the case of biolubricants based on 2-ethyl-1-hexyl oleate (Kleinaite et al., 2014), octylated branched biolubricant (Zhang et al., 2020), castor oil biolubricant through hydrolysis and esterification (Greco-Duarte et al., 2017), or canola oil and graphene as nano-additive (Siddaiah et al., 2019), among others. Regarding biodiesel, the process seems to be clearly defined, through a transesterification of vegetable oils with methanol (or ethanol, which is less common), to obtain fatty acid methyl (or ethyl) esters (Huang et al., 2011). Nevertheless, alternative sources can be found, such as feedstock with high free fatty acid levels (Litinas et al., 2020).
Considering the process selected for biolubricant production in this study (through double transesterification with methanol and pentaerythritol), it could meet all the requirements to be considered as a biorefinery. Fig. 1 shows the main steps carried out in this kind of biorefinery based on vegetable oils.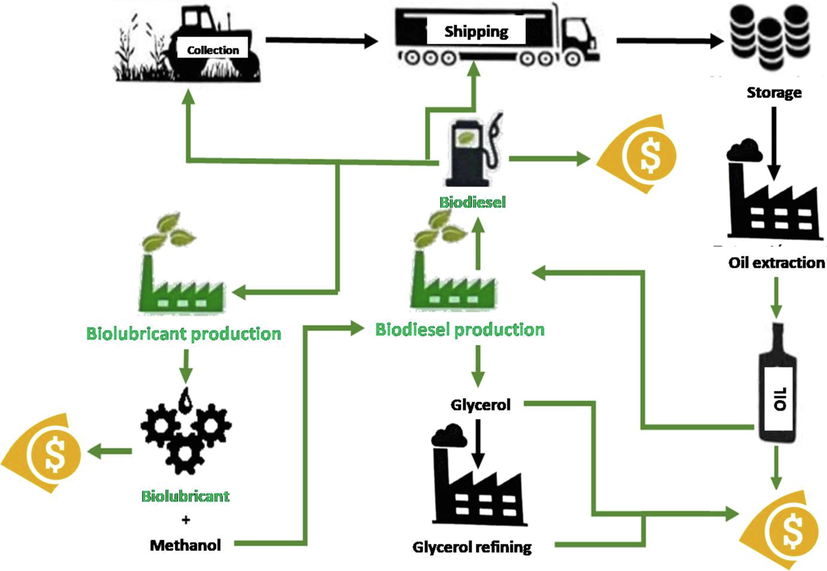
Proposal of a biorefinery based on vegetable oils.
Thus, biodiesel (first transesterification) and biolubricant production (second transesterification) are connected, as biodiesel is the intermediate product to produce biolubricants (following the same kind of reaction and, therefore, similar characteristics for their production). Also, many other steps are connected, such as methanol re-use. Consequently, this process could comply with many of the main requirements of a biorefinery (Liberato et al., 2019; Moncada et al., 2016), as follows: a) It is based on natural and sustainable products; b) a wide range of products are generated, including by-products which could be re-used in this process or valorized, as in the case of glycerol, which can be upgraded into hydrogen (Tabatabaei et al., 2019); c) these products are used for different purposes, providing diversification and making this industry adaptable for changes; d) it contributes to the sustainable and economic growth of developing areas, as some oil crops (such as safflower) can be easily grown in extreme climate conditions or poor soils; e) most of the products used are biodegradable, which is important in the case of leakage or spillage into the environment; f) the atom efficiency is high, as no by-products with difficult management are generated (not even with low molecular weight). Moreover, some by-products evolved might be re-used, as explained previously, and the use of catalysts, both homogeneous or heterogeneous, can be useful in order to make this process more effective compared to petroleum-based processes (Aderibigbe et al., 2020).
Biolubricant production from vegetable oils has been an interesting research field in recent years. In that sense, the use of pentaerythritol has played an important role, as in the case of palm-based pentarerytrhitol ester synthesis, which was optimized to obtain a high-quality product such as high flash point (exceeding 300 °C) and viscosity (12.7 cSt at 100 °C) (Aziz et al., 2014). A thorough study including different biolubricants based on oleate esters of ethylene glycol, 1,2-propanediol, 2,3-butanediol and pentaerythritol stated that the latter, with high viscosity values, was a suitable candidate to replace conventional lubricants (Yao et al., 2014). Equally, biolubricant based on rubber seed oil and different alcohols (including pentaerythritol) pointed out the high quality of polyol esters, with higher viscosity index compared to branched esters (Kamalakar et al., 2013). The esterification of free fatty acids obtained from soybean oil hydrolysis and different alcohols (neopentyl glycol, trimethylolpropane, and pentaerythritol) with lipases showed interesting results, assuring the reusability of lipases (up to 6 consecutive reactions) and high yields in general (Cavalcanti et al., 2018). Finally, different moringa polyesters (one of them produced with pentaerythritol) offered high viscosity index and were non-toxic, which is one of the most important aspects in green chemistry (Moreira et al., 2020).
One of the challenges in this field is the low oxidative stability of bioproducts derived from vegetable oils, which could change their properties during storage (Rodionova et al., 2017). Auto-oxidation can cause a chain reaction, generating free radicals that could contribute to the generation of polymers, free fatty acids, aldehydes, etc., which could increase viscosity (due to polymerization of esters) and acid number (due to free fatty acid generation) in biofuels and biomaterials. Therefore, these undesirable changes would affect the quality of the final products, not complying with the standard for their suitable and specific industrial use.
Considering the interrelation of a biorefinery (see Fig. 1), it is interesting to consider four aspects to keep or enhance the oxidative stability of all the products involved in this process: a) the oxidative stability of the raw material. Fatty acid profile of vegetable oils plays a vital role in the characteristics of the subsequent products. For instance, high amounts of oleic acid (a mono-unsaturated fatty acid) could assure better oxidative stabilities for biodiesel and biolubricant production, whereas high percentages of linoleic or linolenic acid (with double or triple unsaturations and more prone to auto-oxidation) would worsen the properties of these products (Encinar et al., 2020; Martínez et al., 2014). Consequently, it is important to characterize the feedstock, and the selection of genetically modified crops in order to improve oxidative stability (among other properties) can gain importance in the future, as proved for high oleic safflower cultivars in India (Anjani and Yadav, 2017) and Spain (Nogales-Delgado et al., 2021); b) the use of mild conditions to produce biodiesel and biolubricants. This fact is also important to decrease reaction temperature and the use of homogeneous catalysts (the final product could contain traces of metals such as sodium or potassium that can act as catalysts for oxidation processes), which could contribute to auto-oxidation; c) the use of natural or synthetic antioxidants could neutralize free radicals, which could delay most of the undesirable effects of auto-oxidations, as explained previously; d) although the use of stable raw materials is important, it should be considered the final products obtained to understand the final stability obtained. In general, the more complex the final product is, the less stable it is, as it would present more weak points (such as unsaturations) to start auto-oxidation processes. As a consequence, biodiesel (which is a fatty acid methyl ester) is expected to show better oxidative stability values compared to biolubricants (which are fatty acid esters obtained from more complex alcohols, such as pentaerythritol) as it was pointed out in previous studies (Encinar et al., 2020).
In this study, the use of high-oleic safflower and the possible addition of natural and synthetic antioxidants (tannic acid, TA, and tert-butylhydroquinone, TBHQ, respectively) in the corresponding products were studied.
As previously explained, the use of vegetable oils is interesting to produce biodiesel and biolubricants, and the search for genetically modified crops could be an interesting point to improve the characteristics of the products obtained in a biorefinery based on oil crops. Regarding high-oleic safflower, it is an interesting crop mainly due to three reasons: a) First, it is a resistant crop, suitable for extreme climates, offering a high yield in biomass, seed, and oil. Furthermore, its taproot could help to recover soils if it is used as a rotating crop, providing diversification to growers, especially in Spain and Extremadura region (Nogales-Delgado et al., 2019a), where its adaptation would be feasible apart from other different areas such as USA, India, Ethiopia or Romania, among others (Mihaela et al., 2013; Yesilyurt et al., 2020); b) Second, its use to produce some bioproducts included in the proposed biorefinery is well known, especially for biodiesel production, where high yields have been obtained, proving its efficiency in this process (Ilkiliç et al., 2011). Thus, its use in a second transesterification would be feasible, as previous studies have pointed out (Nogales-Delgado et al., 2021); c) Third, as it is a genetically modified crop, its high content in oleic acid (one of the most stable fatty acids due to the fact that it is mono-unsaturated, whereas other fatty acids such as linoleic or linolenic acid are di or tri-unsaturated) could contribute to the stability of the raw material and, subsequently, the intermediate and final products obtained in a biorefinery (as they share common parts of the molecular structure after each transesterification, regardless the alcohol used).
The use of antioxidants to increase oxidative stability of fatty acid esters has been studied, specifically in the case of fatty acid methyl esters (Varatharajan and Pushparani, 2018). Many studies have proved the effectiveness of naturals and, especially, synthetic antioxidants on the improvement of biodiesel stability, keeping their properties during storage, such as oxidative stability after 6 months (Serrano et al., 2013) or longer periods, up to 18 months (Tang et al., 2008). In that sense, the maintenance of viscosity or acid number is vital to keep the characteristics and specific use of this product. It should be noted that, for this purpose, TBHQ is one of the most popular antioxidants used, and some authors have pointed out its effectiveness (clearly exceeding oxidative stability values of 8 h) at low concentrations (usually below 1000 ppm). Thus, in the case of seed biodiesel, 400 ppm were required (Nogales-Delgado et al., 2019b), whereas for camelina biodiesel, 1000 ppm were necessary to comply with the standard (Yang et al., 2017).
Although the effect of antioxidant addition on similar samples (such as FAMEs, biodiesel) has been widely studied, as far as we know, the use of antioxidants in biolubricants is a subject hardly covered in the literature. Nevertheless, keeping the main properties of biodiesel or biolubricants is vital, especially in the case of the latter, where their applications are highly linked to viscosity, which changes during storage. Furthermore, and considering that the biolubricants covered in this study have similar molecular structures compared to fatty acid methyl esters, the effect of antioxidants on them is expected to be similar, which would make these products more competitive in the market compared to mineral lubricants.
According to the above, the aim of this work was to obtain a biolubricant based on high-oleic safflower biodiesel through double transesterification with methanol and pentaerythritol, selecting the right parameters to obtain a high-yield product. Equally, a characterization of the final product and the subsequent antioxidant addition (with TBHQ and tannic acid) was carried out, monitoring some quality parameters during extreme oxidation conditions to check the effectiveness of these antioxidants at keeping these properties. In that sense, the use of antioxidants might be useful to keep the main quality parameters of bioproducts obtained in this biorefinery, enhancing the competitiveness of this kind of facilities compared to traditional refineries.
2 Materials and methods
2.1 Raw material
The raw material used in this study was high-oleic safflower oil, which was collected and mechanically extracted in 2021 by the Agricultural Research Institute “Finca La Orden – Valdesequera”, belonging to CICYTEX (Centro de Investigaciones Científicas y Tecnológicas de Extremadura). Once the oil was filtered and its acidity was checked (below 1%, which allowed to continue the process without any further treatments), it was collected in 25-L containers at room temperature for further treatments.
2.2 Fatty acid methyl ester production (first transesterification)
In order to produce fatty acid methyl esters (biodiesel), transesterification of high-oleic safflower oil (HSO) with methanol was selected. The main chemical conditions to carry out this reaction were selected according to previous studies where a high-yield biodiesel was obtained (Encinar et al., 2020): methanol/oil ratio, 6:1; catalyst concentration (sodium methylate), 0.5% w/w; reaction temperature, 65 °C; stirring rate, 350 rpm; reaction time, 120 min. Afterwards, biodiesel was separated from glycerol through decantation in a separating funnel, and successive washing treatments with distilled water were carried out to remove the catalyst and possible impurities. Finally, the sample was dried at 110 °C to avoid moisture and the final biodiesel was stored in 2.5-L opaque containers for further characterization or treatments.
2.3 Biolubricant production (second transesterification)
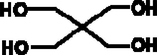
Regarding fatty acid ester production (biolubricant), a second transesterification of fatty acid methyl esters (high-oleic safflower biodiesel, HSBD) with pentaerytrhitol was carried out. The main properties of this superior alcohol are included in Table 1.
Chemical formula
C(CH2OH)4
Molecular structure
Molecular weight, g·mol−1
136.15
Density, g·cm−1 (at 20 °C)
1.39
Melting point, °C
258.4
Boiling point, °C (40.00 hPa)
276
Ignition temperature, °C
490
Thus, it is a reagent in a solid state, and its complex molecular structure (compared to methanol) makes transesterification more difficult, requiring higher temperatures or catalyst concentrations. The experimental facility used for the second transesterification is shown in Fig. 2, where a trap for methanol collection is required as methanol is evolved during the process. Thus, the generic procedure was the following: high-oleic safflower biodiesel (HSBD) was placed into the reactor system, adding the right amount of pentaerythritol and heating the sample to the desired temperature. Once achieved, the corresponding amount of catalyst (sodium methylate) was placed, activating the vacuum pump to obtain the suitable pressure in the reaction system. This point was considered the beginning of the reaction, which took place for two hours with a stirring rate of 350 rpm. Once the process ended, the sample was filtered to remove the surplus pentaerythritol through vacuum filtration, repeating this process until the sample was not cloudy. Finally, high-oleic safflower biolubricant (HSBL) was stored in 2.5-L opaque bottles for further characterization and treatments.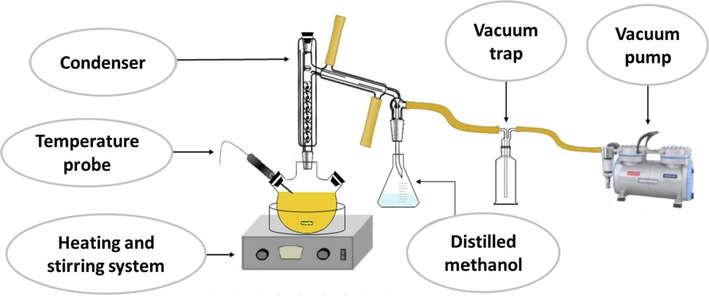
Experimental facility for biolubricant production.
Considering the abovementioned procedure, different experiments were carried out to determine the effect of FAME:pentaerythritol ratio (from 1:1/5 to 1:1/2), pressure (from 760 to 260 mmHg), catalyst concentration (from 0.5 to 1.5% w/w) and temperature (from 100 to 160 °C) on the conversion of HSBD to produce HSBL. This way, Table 2 shows the specifications of each experiment. Afterwards, the best chemical conditions were selected, in order to obtain a high-yield biolubricant for its subsequent characterization.
Experiment1
FAME:pentaerythritol ratio
Pressure, mmHg
[Catalyst]2, %
Temperature, °C
1
1:1/5
760
1
140
2
1:1/4
760
1
140
3
1:1/3
760
1
140
4
1:1/2
760
1
140
5
1:1/4
610
1
140
6
1:1/4
510
1
140
7
1:1/4
260
1
140
8
1:1/4
760
0.5
140
9
1:1/4
760
0.75
140
10
1:1/4
760
1.5
140
11
1:1/4
760
1
100
12
1:1/4
760
1
120
13
1:1/4
760
1
130
14
1:1/4
760
1
150
15
1:1/4
760
1
160
In order to assess the evolution of the chemical reaction, FAME decrease was studied, calculating conversion as follows (see Eq. (1)):
2.4 High-oleic biodiesel and biolubricant characterizaition
2.4.1 Fatty acid methyl ester content
In order to determine FAME content in HSBD (and FAME decrease during HSBL production), gas chromatography coupled to FID detection (Varian 3900, Agilent, Santa Clara, CA, USA) was used. The chromatographic column was a polyethylene glycol column (Zebron ZB-WAX PLUS, Phenomenex, length: 30 m, film thickness: 0.5 µm and i.d.: 0.32 mm) and the main FAMEs determined were methyl oleate, linoleate, linolenate or palmitate, among others. Methyl heptadecanoate was used as an internal standard. All these standards were provided by Sigma-Aldrich (San Luis, USA), and the chromatographic conditions were based on previous studies, according to the standard (Nogales-Delgado et al., 2019a; UNE-EN ISO 12966-2:2011, 2011). Thus, a typical chromatogram obtained under these circumstances can be observed in previous works (Nogales-Delgado et al., 2020).
2.4.2 Moisture and density
Moisture and density determinations were carried out according to the UNE-EN ISO 12937:2000 and UNE-EN-ISO 3675 standards, respectively. Concerning moisture, a Metrohm 870 trinitro plus equipment was used, and density was determined by using a pycnometer (Pobel, Madrid, Spain) (UNE-EN-ISO-12937:2000, 2001; UNE-EN-ISO 3675, 1999).
2.4.3 Viscosity and viscosity index
In the case of HSBD, viscosity was obtained at 40 °C, whereas for HSBL, viscosity was measured at 40 and 100 °C by using an Ostwald viscometer, following the corresponding standard (UNE-EN ISO 3104/AC:1999, 1999). These values were used to calculate viscosity index, according to the standard (ASTM-D2270-10, 2016).
2.4.4 Oxidative stability
Oxidative stability (of HSBD or HSBL) was calculated according to the Rancimat method, as explained elsewhere and in previous studies (Focke et al., 2016; Nogales-Delgado et al., 2019b). Shortly, 3 g of the sample was heated at 110 °C, bubbling synthetic air (10 L·h−1) and connecting the resulting stream of air to 50 mL of distilled water, increasing its conductivity (due to the dilution of oxidation by-products), and monitoring the process with a conductivity meter (Mettler Toledo, Columbus, Ohio, USA). The oxidative stability, expressed in time units (min or h), was the time at which conductivity abruptly increased.
2.4.5 Cold filter plugging point and flash point
Regarding cold filter plugging point (CFPP) determination, the EN 116 standard was followed (UNE-EN 116:2015, 2015). For flash point determination, the Cleveland open-cup method was followed, using the corresponding equipment (Herzog Cleveland semi automatic, Herzog, Landa-Königshofen, Germany), according to the UNE 51-023-90 standard (UNE-EN 51023:1990, 1990).
2.4.6 Acid and iodine numbers
Acid and iodine numbers were determined according to the UNE-EN 12634:1999 and UNE-EN 14111:2003 standards, respectively (UNE-EN-12634:1999, 1999; UNE-EN 14111:2003, 2003). Both parameters were analyzed for HSBD, whereas acid number was determined for HSBL, on account of the fact that this is one of the most interesting parameters to be determined during oxidation processes.
2.4.7 Infrared analysis
FT-IR analysis of HSBL was carried out to identify the main functional groups included in its molecular structure, using the corresponding equipment (Perkin-Elmer Spectrum 3F-IR, Waltham, MA, United States) and measuring in the range of 4000–650 cm−1. The results were processed with computer software.
2.4.8 Thermogravimetry
Thermogravimetric analysis, TG, and derivative thermogravimetry, DTG of the feedstock and biolubricant were carried out by using a thermobalance (STA449F3, QMS403D, VERTEX70, Netzsch–Bruker, Billerica, MA, USA). Around 6 mg of biolubricant was heated from room temperature to 600 °C, with a heating rate of 10 °C·min−1. The carrier gas for pyrolysis was nitrogen (100 mL·min−1). This way, TGA and DTG curves were obtained by monitoring the weight loss of the sample over temperature.
2.5 Antioxidant addition
Once HSBL was obtained, two antioxidants were tested to assess their effectiveness to improve the oxidative stability of the sample. Different concentrations, typically studied in the literature for similar products such as fatty acid methyl esters (from 0 to 1500 ppm) of each antioxidant were added to HSBL (Karavalakis and Stournas, 2010; Nogales-Delgado et al., 2022), measuring the effect on oxidative stability through the Rancimat method. The best concentration was selected to undergo extreme oxidation conditions, as explained in the following section.
2.6 Extreme oxidation conditions
Extreme oxidation conditions were selected to assess the evolution of three main characteristics of HSBL, that is, viscosity, viscosity index and acid number. These conditions were carried out according to previous studies (similar to Rancimat method) (Nogales-Delgado et al., 2022, 2019b), and the procedure was the following: 30 g of biolubricant was heated at 110 °C, bubbling synthetic air (100 L·h−1) for 8 h. During this process, samples at 0, 2, 4, 6 and 8 h were collected to measure their viscosity, viscosity index and acid number (according to previous sections). To sum up, Fig. 3 shows the experimental design for this work, including the main steps carried out.
Experimental design for this work.
3 Results and discussion
3.1 Fatty acid methyl ester (biodiesel) characterization
As it can be seen in Table 3, HSBD complied with all the requirements established by the UNE-EN 14214 standard, including oxidative stability (exceeding 10 h), whose high value is not common in biodiesel obtained from vegetable oils, although some works showed similar value, slightly below 8 h, as in the case of canola biodiesel (Hong et al., 2015). In that sense, there is a strong influence of FAME composition as well as the presence of natural antioxidants in the feedstock (Tang et al., 2008). Specifically, the presence of double bonds in FAMEs, especially with adjacent allylic sites, can promote the decrease in oxidative stability (Lanjekar and Deshmukh, 2016). Thus, specifically regarding common safflower biodiesel (not high oleic), according to the literature, short oxidative stability values were found, as in the case of safflower biodiesel (ranging from 0.57 and 1.46 h), where low amounts of methyl oleate (from 12.76 and 10%) and high amounts of methyl linolenate (from 69.65 and 75%) were found (Mihaela et al., 2013; Nogales-Delgado et al., 2019a). Other biodiesel samples obtained from different vegetable oils had low oxidative stability values, compared to the results found in this study. Thus, in the case of soybean biodiesel, it had low oxidative stability values (ranging from 4.33 to 4.97 h), (de Freitas et al., 2019; De Sousa et al., 2014), whereas sunflower biodiesel had induction points below 3 h (van der Westhuizen and Focke, 2018), increasing this value when mixed with corn biodiesel (around 5 h) (Nogales-Delgado et al., 2019b). For cardoon biodiesel, previous studies showed low values (1.35 h) (Nogales-Delgado et al., 2020). In this case, methyl oleate content in HSB was high, exceeding 80%, whereas methyl linoleate content was much lower compared to these data obtained in the literature. As explained in previous studies, the former is more stable, including one single unsaturation, whereas the latter is more unstable, with two unsaturations. Thus, oxidative stability or induction period decreases with the increase in the percentages of unsaturated fatty acid in raw materials (Sundus et al., 2017). This fact could explain the high oxidative stability obtained in this study, pointing out the importance of a suitable selection of feedstocks (and its influence on the characteristics of the products, as in the case of biodiesel) in a biorefinery with these characteristics (Jose and Anand, 2016), as oxidative stability is related to storage stability of products (Kumar, 2017) and the lower requirement of antioxidants (Yang et al., 2014).
Characteristic
Units
HSBD
EN 14214 limits
Lower
Upper
FAME content
%
97.5
96.5
–
Moisture
mg·kg−1
200
–
500
Density at 15 °C
kg·m−3
869
860
900
Viscosity at 40 °C
mm2·s−1
4.51
3.50
5.00
Oxidative stability
H
10.78
8
–
Flash point
°C
190
101
–
Acid number
mg KOH·g−1
0.13
–
0.50
Iodine number
g I2·100 g−1
88.7
–
120
Methyl linoleate
% (m·m−1)
0.01
–
12
Cold filter plugging point
°C
−4
−10
0
Also, according to the same table, flash point was considerably high, which implies a safe product during storage. The high FAME content obtained, exceeding 97%, proved that the chemical conditions selected in this work were suitable. Viscosity and density values were within the range required by the standard, and the compliance with all the requirements indicated that a high-quality biodiesel was obtained. This fact is vital in a biorefinery, especially considering that biodiesel can act as an intermediate product in this case.
3.2 Biolubricant production
Once HSBD was obtained, the second transesterification with pentaerythritol was carried out, assessing the effect of FAME/alcohol ratio, pressure, catalyst concentration and temperature on conversion, as explained in the following subsections.
3.2.1 Effect of fatty acid methyl ester:pentaerythritol ratio
Regarding FAME:alcohol ratio, Fig. 4 shows the effect of this parameter on conversion. As it can be inferred, the conversion was higher as the amount of alcohol increased, exceeding 65% in the case of 1:1/2 FAME/pentaerythritol ratio. It was equally observed in other studies, as in the case of polyol ester biolubricant produced from Rhodotorula glutinis lipid, whose yield increased with the amount of alcohol (trimethylolpropane) (Ma et al., 2021). Thus, the excess of alcohol contributed to the generation of reaction products. However, the surplus alcohol could imply more difficulties during vacuum filtration process. The same trend (and problem) was observed for other alcohols which are in solid state, such as 2,2-dimethyl-1,3-propanediol or 2-ethyl-2-hydroxymethyl-1,3-propanediol, which were used for biolubricant production through transesterification from cardoon and rapeseed oil, respectively (Encinar et al., 2021; Nogales-Delgado et al., 2020). That is the reason why a ratio of 1:1/3 was selected, where a high conversion was achieved with lower amounts of alcohol.
Effect of Fatty acid methyl ester/pentaerythritol mole ratio on biolubricant production. Chemical conditions: Pressure, 760 mmHg; Catalyst concentration, 1.0% w/w; Temperature, 140 °C; Reaction time, 120 min; Stirring rate, 350 rpm.
3.2.2 Effect of pressure
Fig. 5 shows the effect of pressure (or vacuum) on FAME conversion. Thus, and even though a gradual increase in conversion was observed as pressure decreased, similar results were obtained for 760, 610 and 510 mmHg (ranging from 50.64 to 55.97%), whereas 260 mmHg showed the highest conversion with 67.54%. As in the previous case, the balance was shifted towards product generation, in this case on account of methanol removal. As a consequence, the lowest pressure (that is, 260 mmHg) was selected for further experiments.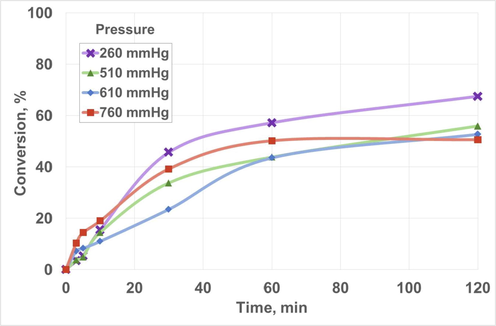
Effect of pressure on biolubricant production. Chemical conditions: Fatty acid methyl ester/pentaerythritol ratio, 1:1/4; Catalyst concentration, 1.0% w/w; Temperature, 140 °C; Reaction time, 120 min; Stirring rate, 350 rpm.
3.2.3 Effect of catalyst concentration
Regarding sodium methylate concentration, Fig. 6 shows the main results obtained. As catalyst concentration was higher, conversion tended to increase, especially concerning 1.00 and 1.50% w/w, where the balance was achieved within the reaction time of 2 h (as conversion was similar at 60 and 120 min, showing a plateau in the figure). In addition, as catalyst concentration increased, the slope of the curves at the initial reaction times (that is, between 0 and 10 min) was higher for the highest catalyst concentration, which was observed in similar studies about biolubricant production, esters derived from ethylene glycol ester or 2-Ethyl-2-hydroxymethyl-1,3-propanediol (Hussein et al., 2021; Nogales-Delgado et al., 2021). Equally, other studies did not observe considerable increases in yield with catalysts loads exceeding 1% w/w, as in the case of biolubricant production from waste cooking oil and ethylene glycol with calcium oxide as a catalyst (Cavalcanti et al., 2018) or palm fatty acid distillate esterification with neopentyl glycol, where a solid acid catalyst was used (Ng et al., 2022). As similar conversion values were obtained for 1.00 and 1.50% w/w (50.64 and 55.97%, respectively), the former was selected for further studies, according to economic criteria.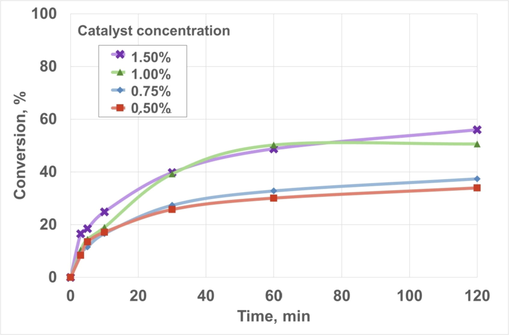
Effect of catalyst concentration on biolubricant production. Chemical conditions: Pressure, 760 mmHg; Fatty acid methyl ester/pentaerythritol ratio, 1:1/4; Temperature, 140 °C; Reaction time, 120 min; Stirring rate, 350 rpm.
3.2.4 Effect of temperature
Concerning temperature, Fig. 7 included the main results obtained for these experiments. As expected, an increase in temperature implied higher conversion values at the end of each experiment, increasing the reaction rate at the beginning of the reaction. This could be due to the endothermic nature of the reaction, increasing chemical product generation with temperature, as observed for the production of biolubricants from different sources and alcohols (Hussein et al., 2021; Ma et al., 2021; Ng et al., 2022). Studies with waste cooking oil and ethylene glycol obtained increasing yields with temperature, reaching maximum values at 130 and 140 °C (Cavalcanti et al., 2018). Considerable conversion values were obtained at 150 and 160 °C, with a sharp increase in conversion in the first 30 min, exceeding 50%. Afterwards, the reaction slowed down gradually, reaching a stationary state from 60 min at 160 °C and obtaining 77.24% conversion at the end of this experiment. Thus, 160 °C was the temperature selected for further experiments to produce the final biolubricant.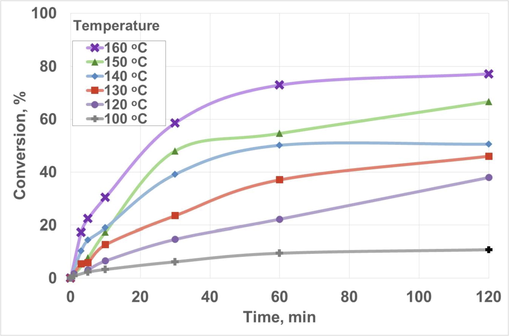
Effect of temperature on biolubricant production. Chemical conditions: Pressure, 760 mmHg; Fatty acid methyl ester/pentaerythritol ratio, 1:1/4; Catalyst concentration, 1.0% w/w. Reaction time, 120 min; Stirring rate, 350 rpm.
3.3 Final biolubricant production and characterization
As explained in previous sections, the most suitable reaction parameters of these experiments were selected to obtain high yields of biolubricant. To sum up, Table 4 shows the chemical conditions for HSBL production, following economic and efficiency criteria.
FAMEs:Alcohol mole ratio
Pressure, mmHg
[Catalyst], %
Temperature, °C
Conversion, %
1:1/3
260
1.0
160
>94
Following these conditions, the yield of the biolubricant obtained was high, as FAME conversion exceeded 90%, according to Table 5, where the most important characteristics of HSBL were included. The high conversion obtained assured a biolubricant with recognizable characteristics, as its purity was considerable. Other biolubricants found in the literature (from the same raw material through double transesterification with methanol and 2-ethyl-2-hydroxymethyl-1,3-propanediol) had lower conversion rates, approximately 93% (Nogales-Delgado et al., 2021). Regarding the rest of characteristics, HSBL had low moisture and acid number, and the viscosity values obtained (77.70 and 12.30 mm2·s−1 at 40 and 100 °C, respectively) allowed this product to be used as a SAE 10 W30 lubricant, that is, for automotive purposes. These values were similar to other pentaerythritol esters observed in the literature, with values ranging from 65.9 and 68.4 mm2·s−1 (or cSt) at 40 °C and between 11.9 and 12.7 mm2·s−1 at 100 °C (Aziz et al., 2014). One interesting property of HSBL was its high viscosity index, which implied lower viscosity changes with temperature (a desirable effect considering the multiple changes in working conditions in industry). Its value was higher compared to other commercial lubricants and biolubricants, like those included in this table or other studies, where a wide range of lubricants and biolubricants were covered, whose viscosity index ranged from 97 to 107 (Barbosa et al., 2021). Furthermore, flash and combustion points were considerably high compared to the commercial biolubricant, which assured safety during storage. These values are in accordance with other biolubricants covered in the literature, where other polyol esters showed similar flash point ranges from 215 to 331 °C (Ho et al., 2019).
Characteristics
Units
HSBL
C. biolubricant
C. lubricant
Conversion
%
94.13
92.90
–
Density at 15 °C
kg·m−3
927
945
870
Moisture
%
0.05
–
–
Acid number
mg KOH·g−1
0.38
0.17
10
Viscosity at 40 °C
mm2·s−1
77.7
73.4
82.6
Viscosity at 100 °C
mm2·s−1
12.3
9.3
12.2
Viscosity index
–
155
103
143
Flash point
°C
260
216
221
Combustion point
°C
271
228
–
Oxidative stability
h
2.86
6.72
–
However, the oxidative stability of HSBL was poor (not achieving 3 h), possibly due to the complexity of the molecule obtained, as it will be explained in following sections. Thus, the use of antioxidants, as expected in this study, is clearly justified. Otherwise, some changes in the properties of HSBL would takes place, especially concerning viscosity and acid number.
Regarding TG and DTG, Fig. 8 shows the TG and DTG curves for high oleic safflower oil and biolubricant. One single degradation step (at 418 °C) was observed for the oil, whereas two main degradation steps (at 231 and 447 °C, which were the peak temperatures found in DTG) were observed in HSBL. The first stage (an almost negligible DTG peak), as observed by other authors and in previous studies, can be due to methyl ester degradation (with temperature peaks at around 220–230 °C (Ferreira et al., 2019; Nogales-Delgado et al., 2019b), which could imply the presence of impurities. The second degradation stage can be attributed to the main thermal degradation stage, as it was a prominent DTG peak. In the case of safflower oil, this process started at 300 °C, finishing at 465 °C, whereas in the case of safflower biolubricant it started at around the same temperature but ended at higher temperature values (480 °C). In addition, the maximum degradation rate (DTG peak) was observed at 419 and 447 °C for oil and biolubricant, respectively, which proved the higher thermal stability of the latter, possibly due to the absence of β-hydrogen in the final ester obtained (HSBL), as pointed out in the literature for a similar comparison between the an original vegetable oil and the final trimethylpropane triester-based biolubricant (Bahadi et al., 2021). The main degradation stage in biolubricant took place at higher temperatures compared to other similar products (Bezerra et al., 2020; Ferreira et al., 2019), although similar temperatures for this stage (at around 450 °C for polyol ester biolubricant produced from Rhodotorula glutinis lipid) were also found (Ma et al., 2021). Thus, the final biolubricant obtained from high-oleic safflower oil and pentaerythritol had a high thermal stability, exceeding many equivalent products included in the literature.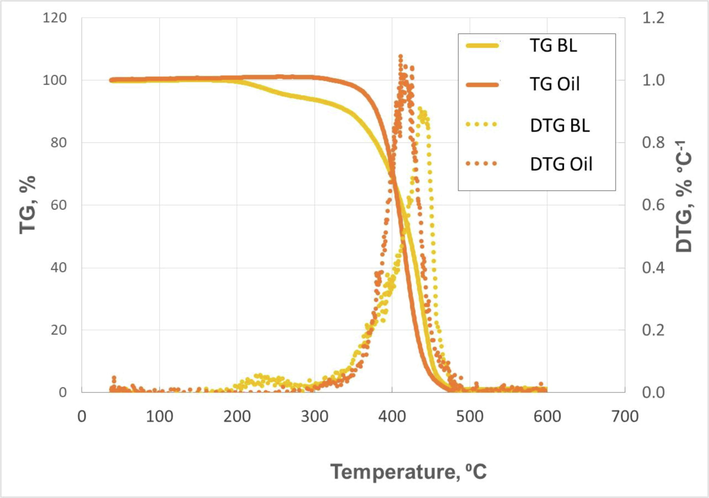
Thermogravimetry (TG) and derivative thermogravimetry (DTG) curves for high-oleic safflower oil and biolubricant.
In the case of IR spectrum, Fig. 9 shows the main results. As it can be observed, a typical spectrum for esters was obtained as found in the literature (for instance, -C = O band at around 1700 cm−1) (Ma et al., 2021), where the absence of a wide and prominent peak at around 3500 cm−1 should be pointed out. This fact implies the absence of hydroxyl groups in the chemical structure of the sample, indicating a high conversion of all the –OH groups in pentaerythritol (which were transesterified) and the absence of surplus pentaerythritol.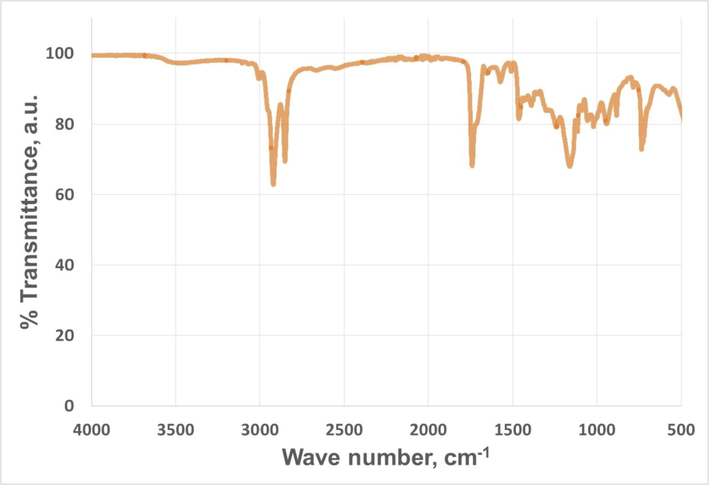
Infrared spectrum of high-oleic safflower biolubricant.
According to the above, the chemical reaction that could have taken place is shown in Fig. 10, where different FAMEs (where 3 methyl oleate molecules and 1 methyl linoleate molecule were the most representative compounds in HOBD) reacted with the four –OH groups in pentaerythritol, assuring the total reaction of the alcohol and the production of the final biolubricant and methanol.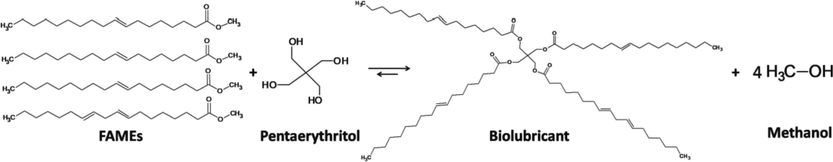
Reaction to produce biolubricant from high-oleic safflower biodiesel through transesterification with pentaerythritol.
3.4 Antioxidant addition in biolubricant
As it was explained in previous sections, the addition of antioxidants was necessary to increase the oxidative stability of HSBL. In this work, two antioxidants were tested (TBHQ and TA), and their effect on oxidative stability (that is, induction point) was included in Fig. 11.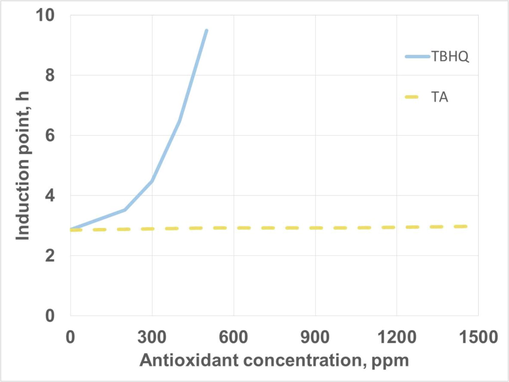
Effect of antioxidant addition on oxidative stability of high-oleic safflower biolubricant.
As it can be observed, TBHQ considerably increased the oxidative stability of HSBL, exceeding 9 h with low amounts (500 ppm). On the contrary, the increase in oxidative stability due to TA was negligible, possibly due to the low solubility in biolubricant. Thus, 500 ppm of TBHQ was selected for further studies, where extreme oxidation conditions on HSBL were tested. These concentrations were in accordance with the effectiveness found for this antioxidant in the literature, where low concentration ranges were observed in antioxidant addition in biodiesel, at around 400 ppm for corn and sunflower biodiesel (Nogales-Delgado et al., 2019b) and 1000 ppm for waste cooking biodiesel (Uğuz et al., 2019).
Three main parameters were studied during extreme oxidation conditions of HSBL, that is, viscosity, viscosity index and acid number evolution. Concerning the former, Fig. 12 shows the evolution of this parameter, where control and doped samples (with TBHQ addition) were compared. Thus, during oxidation, viscosity increased for control samples (increasing 55% at the end of oxidation process), possibly due to polymerization during auto-oxidation of esters. This trend was similar in other biomaterials such as biodiesel, where during storage or extreme oxidation conditions there was a considerable increase in viscosity. On the other hand, the addition of TBHQ showed a positive effect, keeping viscosity during the process, which implies the maintenance of this property during storage, not varying its performance for specific uses. The same effect was shown in previous studies with fatty acid methyl esters during oxidation or storage (Nogales-Delgado et al., 2019b; Nogales-Delgado and Encinar Martín, 2021).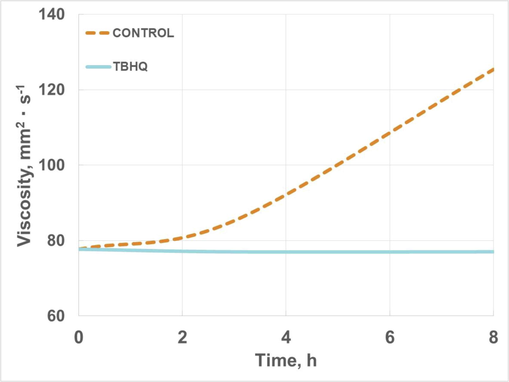
Viscosity evolution of high-oleic safflower biolubricant (with and without TBHQ addition) during oxidation.
Regarding viscosity index evolution, Fig. 13 shows its evolution for control and doped samples. Control samples showed a considerable decrease during oxidation, which implies higher variations in viscosity with temperature, limiting its use for industrial purposes. A similar behavior was observed for biolubricants obtained from cardoon biolubricant obtained through double transesterification with methanol and 2,2-dimethyl-1,3-propanediol, where a decrease in viscosity index was found during storage (Nogales-Delgado and Encinar Martín, 2021). When TBHQ was added, viscosity index decrease was reduced, contributing to keep HSBL properties during oxidation.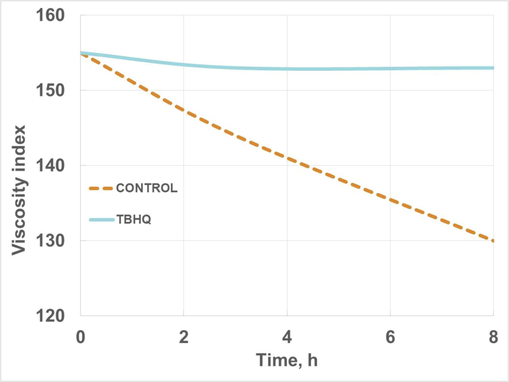
Viscosity index evolution of high-oleic safflower biolubricant (with and without TBHQ addition) during oxidation.
Finally, acid number evolution is included in Fig. 14. As previously shown, control sample underwent an increase in acid number, possibly due to the auto-oxidation process, where many by-products are generated (carboxylic acids, aldehydes, ketones) which can contribute to the raise in this parameter. This trend was also observed in oxidized biodiesel samples, as explained in previous studies (Nogales-Delgado et al., 2019b). Again, TBHQ kept this property during oxidation showing a negligible increase in acid number and proving its effectiveness when it comes to delaying auto-oxidation in HSBL. Compared to the use of TBHQ in different biodiesel samples, similar antioxidant concentrations practically kept acid number during extreme oxidation conditions, assuring low degradation during storage (Nogales-Delgado et al., 2022).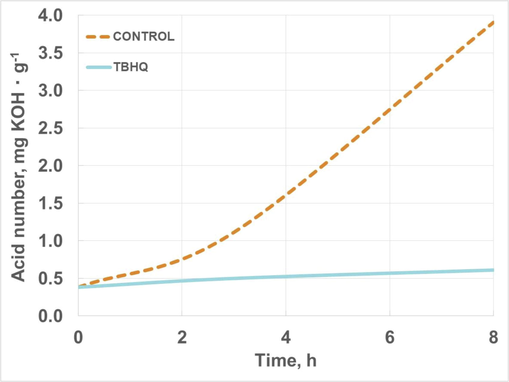
Acid number evolution of high-oleic safflower biolubricant (with and without TBHQ addition) during oxidation.
To sum up, the addition of TBHQ was effective on keeping the main quality parameters of the biolubricant produced during extreme oxidation conditions, which can imply a good performance during the storage of this product, making the use of antioxidants vital to produce high-quality biolubricants.
4 Conclusion and future research directions
The main findings inferred from this work were the following:
-
High-oleic safflower was a suitable raw material for a biorefinery based on vegetable oils to produce biodiesel and biolubricants, which produced a biodiesel that complied with all the requirements established by the UNE-EN 14214 standard.
-
Regarding biolubricant production with pentaerythritol, high yields (exceeding 94%) were obtained with the following chemical conditions: FAME/pentaerythritol ratio, 1:1/3; Temperature, 160 °C; Pressure, 260 mmHg; Catalyst concentration, 1.0% w/w. Reaction time, 120 min; Stirring rate, 350 rpm.
-
The biolubricant obtained showed good properties such as high viscosity index (>150), flash and combustion points (260 and 271 °C, respectively), whereas its oxidative stability was relatively low (<3h), which made the use of antioxidants necessary.
-
TBHQ addition was effective, increasing oxidative stability up to 10 h with low concentrations (500 ppm). Moreover, during oxidation conditions, the main properties of the biolubricant (viscosity, viscosity index and acid number) were kept.
-
On the other hand, the effectiveness of tannic acid was almost negligible under the circumstances followed in this study.
-
This was an introductory research work with a practical purpose, that is, to keep the quality parameters of a biolubricant based on high-oleic safflower oil and pentaerythritol. This study was limited to a specific biolubricant, two antioxidants and the monitorization of the most representative characteristics of a biolubricant during oxidation. Further studies, including the possible application of different antioxidants (both natural and synthetic) with different vegetable oils (showing differences in oxidative stability) could be useful to broaden the knowledge in this field. Thus, the effect of a specific antioxidant on the properties of different components of a biorefinery (for instance, both biodiesel and biolubricant) could be an interesting research field, as well as the study of the retention of other properties such as flash and combustion point or ultraviolet absorption (which are presumably affected during storage or oxidation processes). This way, the use of this additives could contribute to a higher efficiency of biorefineries, making them a suitable replacement for the corresponding petroleum-based refineries.
Acknowledgements
Prof. Dr. José María Encinar: before my career comes to an end, I wish to thank the group of people who has contributed to its development. Firstly, I would like to acknowledge Prof. Dr. José Luis Sotelo Sancho, our “Jefe” (boss), who taught us what Chemical Engineering was and, above all, ended up being our friend. Equally, I want to thank my Department colleagues Vicente Montero (†), Javier Viguera (†), Joaquín Torregrosa (†), Clara Tasis, Gabriel Ovejero, Javier Benítez, Fernando Beltrán, Jesús Beltrán, Manolo González, Juan F. García, Teresa González, Javier Rivas, Juan Luis Acero, Pedro M. Álvarez, Benito Acedo, Francisco J. Real, Joaquín R. Domínguez, Olga Gimeno, Eva Rodríguez, Jesús Frades and Miguel A. Alonso, with whom I have shared many working hours (and party time too), hopes, worries, sadness and, above all, joys. Finally, I would like to express thanks to the colleagues and partners belonging to our research group, especially Juan Félix González (friend and travel companion), Antonio Ramiro, Eduardo Sabio, Jerónimo González, Carmen González, Silvia Román, Gloria Martínez, Nuria Sánchez, Sergio Nogales and Belen Roldán. Thank you all, with your company, we can say: “… que cincuenta años no es nada”.
The authors would like to thank the Junta de Extremadura and FEDER for the financial support (“Junta de Extremadura, Ayudas para la realización de actividades de investigación y desarrollo tecnológico, de divulgación y de transferencia de conocimiento por los Grupos de Investigación de Extremadura” and “Fondos Europeos de Desarrollo Regional (Una manera de hacer Europa)” GR21139 and IB18028, respectively).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Qualitative role of heterogeneous catalysts in biodiesel production from Jatropha curcas oil. Biofuel Res. J.. 2020;7:1159-1169.
- [Google Scholar]
- On the exergoeconomic and exergoenvironmental evaluation and optimization of biodiesel synthesis from waste cooking oil (WCO) using a low power, high frequency ultrasonic reactor. Energy Convers. Manag.. 2018;164:385-398.
- [CrossRef] [Google Scholar]
- Exergoenvironmental analysis of bioenergy systems: A comprehensive review. Renew. Sustain. Energy Rev.. 2021;149
- [CrossRef] [Google Scholar]
- Machine learning technology in biodiesel research: A review. Prog. Energy Combust. Sci.. 2021;85
- [CrossRef] [Google Scholar]
- High yielding-high oleic non-genetically modified Indian safflower cultivars. Ind. Crops Prod.. 2017;104:7-12.
- [CrossRef] [Google Scholar]
- ASTM-D2270-10, 2016. Standard practice for calculating viscosity index from kinematic viscosity at 40 °C and 100 °C.
- Application of response surface methodology (RSM) for optimizing the palm-based pentaerythritol ester synthesis. Ind. Crops Prod.. 2014;62:305-312.
- [CrossRef] [Google Scholar]
- Synthesis, optimization, tribological and rheological properties of trimethylpropane triester-based biolubricant from Malaysian crude palm kernel oil. Arab. J. Chem. 2021:103286.
- [CrossRef] [Google Scholar]
- Biolubricant production under zero-waste Moringa oleifera Lam biorefinery approach for boosting circular economy. Ind. Crops Prod.. 2021;167
- [CrossRef] [Google Scholar]
- Bezerra, R. de C. de F., Rodrigues, F.E.A., Arruda, T.B.M.G., Moreira, F.B. de F., Chaves, P.O.B., Assunção, J.C. da C., Ricardo, N.M.P.S., 2020. Babassu-oil-based biolubricant: Chemical characterization and physicochemical behavior as additive to naphthenic lubricant NH-10. Ind. Crops Prod. 154, 112624. https://doi.org/10.1016/j.indcrop.2020.112624.
- Cavalcanti, E.D.C., Aguieiras, É.C.G., da Silva, P.R., Duarte, J.G., Cipolatti, E.P., Fernandez-Lafuente, R., da Silva, J.A.C., Freire, D.M.G., 2018. Improved production of biolubricants from soybean oil and different polyols via esterification reaction catalyzed by immobilized lipase from Candida rugosa. Fuel 215, 705–713. https://doi.org/10.1016/j.fuel.2017.11.119.
- Evaluation of the oxidative stability and cold filter plugging point of soybean methyl biodiesel/bovine tallow methyl biodiesel blends. Ind. Crops Prod.. 2019;140
- [CrossRef] [Google Scholar]
- Use of natural antioxidants in soybean biodiesel. Fuel. 2014;134:420-428.
- [CrossRef] [Google Scholar]
- do Valle, C.P., Rodrigues, J.S., Fechine, L.M.U.D., Cunha, A.P., Queiroz Malveira, J., Luna, F.M.T., Ricardo, N.M.P.S., 2018. Chemical modification of Tilapia oil for biolubricant applications. J. Clean. Prod. 191, 158–166. https://doi.org/10.1016/j.jclepro.2018.04.062.
- A review on global perspectives of sustainable development in bioenergy generation. Bioresour. Technol.. 2022;348
- [CrossRef] [Google Scholar]
- Biodiesel and biolubricant production from different vegetable oils through transesterification. Eng. Reports. 2020;1–10
- [CrossRef] [Google Scholar]
- Biolubricant Production through Double Transesterification: Reactor Design for the Implementation of a Biorefinery Based on Rapeseed. Processes. 2021;9:1224.
- [CrossRef] [Google Scholar]
- Investigation of the thermal degradation of the biolubricant through TG-FTIR and characterization of the biodiesel – Pequi (Caryocar brasiliensis) as energetic raw material. Fuel. 2019;245:398-405.
- [CrossRef] [Google Scholar]
- Focke, W.W., Westhuizen, I. Van Der, Oosthuysen, X., 2016. Biodiesel oxidative stability from Rancimat data. Thermochim. Acta. https://doi.org/10.1016/j.tca.2016.03.023.
- Bioeconomy and green recovery in a post-COVID-19 era. Sci. Total Environ.. 2022;808
- [CrossRef] [Google Scholar]
- Two-step enzymatic production of environmentally friendly biolubricants using castor oil: Enzyme selection and product characterization. Fuel. 2017;202:196-205.
- [CrossRef] [Google Scholar]
- Biolubricants through renewable hydrocarbons: A perspective for new opportunities. Renew. Sustain. Energy Rev.. 2019;113
- [CrossRef] [Google Scholar]
- Fuel properties of canola oil and lard biodiesel blends: Higher heating value, oxidative stability, and kinematic viscosity. J. Ind. Eng. Chem. 2015
- [CrossRef] [Google Scholar]
- Huang, D., Zhou, H., Lin, L., 2011. Biodiesel: An alternative to conventional fuel, in: Energy Procedia. https://doi.org/10.1016/j.egypro.2012.01.287.
- Experimental investigation and process simulation of biolubricant production from waste cooking oil. Biomass Bioenergy. 2021;144
- [CrossRef] [Google Scholar]
- Biodiesel from safflower oil and its application in a diesel engine. Fuel Process. Technol.. 2011;92:356-362.
- [CrossRef] [Google Scholar]
- Effects of biodiesel composition on its long term storage stability. Fuel 2016
- [CrossRef] [Google Scholar]
- Rubber seed oil-based biolubricant base stocks: A potential source for hydraulic oils. Ind. Crops Prod.. 2013;51:249-257.
- [CrossRef] [Google Scholar]
- Impact of antioxidant additives on the oxidation stability of diesel/biodiesel blends. Energy Fuels. 2010;24:3682-3686.
- [CrossRef] [Google Scholar]
- A cleaner approach for biolubricant production using biodiesel as a starting material. J. Clean. Prod. 2014
- [CrossRef] [Google Scholar]
- Oxidative stability of biodiesel: Causes, effects and prevention. Fuel 2017
- [CrossRef] [Google Scholar]
- A review of the effect of the composition of biodiesel on NOx emission, oxidative stability and cold flow properties. Renew. Sustain. Energy Rev.. 2016;54:1401-1411.
- [CrossRef] [Google Scholar]
- Biodiesel production from high FFA feedstocks with a novel chemical multifunctional process intensifier. Biofuel Res. J.. 2020;7:1170-1177.
- [Google Scholar]
- Enzyme-catalyzed synthesis and properties of polyol ester biolubricant produced from Rhodotorula glutinis lipid. Biochem. Eng. J.. 2021;173
- [CrossRef] [Google Scholar]
- Martínez, G., Sánchez, N., Encinar, J.M., González, J.F., 2014. Fuel properties of biodiesel from vegetable oils and oil mixtures. Influence of methyl esters distribution. Biomass and Bioenergy. https://doi.org/10.1016/j.biombioe.2014.01.034.
- Development of biolubricants from vegetable oils via chemical modification. J. Ind. Eng. Chem. 2016
- [CrossRef] [Google Scholar]
- Perspectives of safflower oil as biodiesel source for South Eastern Europe (comparative study: Safflower, soybean and rapeseed) Fuel 2013
- [CrossRef] [Google Scholar]
- Moncada B, J., Aristizábal M, V., Cardona A, C.A., 2016. Design strategies for sustainable biorefineries. Biochem. Eng. J. 116, 122–134. https://doi.org/10.1016/j.bej.2016.06.009.
- Moringa polyesters as eco-friendly lubricants and its blends with naphthalenic lubricant. Ind. Crops Prod.. 2020;158
- [CrossRef] [Google Scholar]
- Production of sustainable two-stroke engine biolubricant ester base oil from palm fatty acid distillate. Ind. Crops Prod.. 2022;175
- [CrossRef] [Google Scholar]
- The Effect of Antioxidants on Corn and Sunflower Biodiesel Properties under Extreme Oxidation Conditions. JAOCS. J. Am. Oil Chem. Soc. 2019
- [CrossRef] [Google Scholar]
- Safflower Biodiesel: Improvement of its Oxidative Stability by using BHA and TBHQ. Energies 2019:19-22.
- [Google Scholar]
- Cardoon biolubricant through double transesterification: Assessment of its oxidative, thermal and storage stability. Mater. Lett.. 2021;302
- [CrossRef] [Google Scholar]
- Valorization of Cynara Cardunculus L. Oil as the Basis of a Biorefinery for Biodiesel and Biolubricant Production. Energies. 2020;13:5085.
- [CrossRef] [Google Scholar]
- High oleic safflower oil as a feedstock for stable biodiesel and biolubricant production. Ind. Crops Prod.. 2021;170
- [CrossRef] [Google Scholar]
- Effect of tert-butylhydroquinone on biodiesel properties during extreme oxidation conditions. Fuel. 2022;310
- [CrossRef] [Google Scholar]
- The GeGaLo index: Geopolitical gains and losses after energy transition. Energy Strateg. Rev.. 2019;26
- [CrossRef] [Google Scholar]
- Chemical modification of vegetable oils for the production of biolubricants using trimethylolpropane: A review. Egypt. J. Pet.. 2020;29:75-82.
- [CrossRef] [Google Scholar]
- Biofuel production: Challenges and opportunities. Int. J. Hydrogen Energy. 2017
- [CrossRef] [Google Scholar]
- Environmental sustainability tools in the biofuel industry. Biofuel Res. J.. 2018;5:751-752.
- [Google Scholar]
- Long term storage stability of biodiesel: Influence of feedstock, commercial additives and purification step. Fuel Process. Technol.. 2013;116:135-141.
- [CrossRef] [Google Scholar]
- Influence of environmental friendly multiphase lubricants on the friction and transfer layer formation during sliding against textured surfaces. J. Clean. Prod.. 2019;209:1245-1251.
- [CrossRef] [Google Scholar]
- Sundus, F., Fazal, M.A., Masjuki, H.H., 2017. Tribology with biodiesel: A study on enhancing biodiesel stability and its fuel properties. Renew. Sustain. Energy Rev. https://doi.org/10.1016/j.rser.2016.11.217.
- Reactor technologies for biodiesel production and processing: A review. Prog. Energy Combust. Sci.. 2019;74:239-303.
- [CrossRef] [Google Scholar]
- The oxidative stability of biodiesel: Effects of FAME composition and antioxidant. Lipid Technol.. 2008;20:249-252.
- [CrossRef] [Google Scholar]
- Global low-carbon energy transition in the post-COVID-19 era. Appl. Energy. 2022;307
- [CrossRef] [Google Scholar]
- Fuel stability of biodiesel from waste cooking oil: A comparative evaluation with various antioxidants using FT-IR and DSC techniques. Biocatal. Agric. Biotechnol.. 2019;21
- [CrossRef] [Google Scholar]
- UNE-EN 51023:1990, 1990. Petroleum products. Determination of flash and fire points. Cleveland open cup method.
- UNE-EN ISO 3104/AC:1999, 1999. Petroleum products. Transparent and opaque liquids. Determination of kinematic viscosity and calculation of dynamic viscosity (ISO 3104:1994).
- UNE-EN ISO 12966-2:2011, 2011. Animal and vegetable fats and oils – Gas chromatography of fatty acid methyl esters – part 2: preparation of methyl esters of fatty acids.
- UNE-EN 14111:2003, 2003. Fat and oil derivatives. Fatty Acid Methyl Esters (FAME). Determination of iodine value.
- UNE-EN 116:2015, 2015. Diesel and domestic heating fuels – Determination of cold filter plugging point- Stepwise cooling bath method.
- UNE-EN-12634:1999, 1999. Productos petrolíferos y lubricantes. Determinación del índice de ácido. Método de valoración potenciométrica en un medio no acuoso.
- UNE-EN-ISO 3675, 1999. Crude petroleum and liquid petroleum products. Laboratory determination of density. Hydrometer method.
- UNE-EN-ISO-12937:2000, 2001. Productos petrolíferos. Determinación de agua. Método de Karl Fischer por valoración culombimétrica.
- United Nations, 2015. Transforming our world : the 2030 Agenda for Sustainable Development [WWW Document]. URL https://www.un.org/sustainabledevelopment/sustainable-development-goals/ (accessed 8.2.21).
- Stabilizing sunflower biodiesel with synthetic antioxidant blends. Fuel. 2018;219:126-131.
- [CrossRef] [Google Scholar]
- Varatharajan, K., Pushparani, D.S., 2018. Screening of antioxidant additives for biodiesel fuels. Renew. Sustain. Energy Rev. https://doi.org/10.1016/j.rser.2017.07.020.
- A review of the role of bioenergy modelling in renewable energy research & policy development. Biomass Bioenergy. 2020;136
- [CrossRef] [Google Scholar]
- Bioeconomy during the COVID-19 and perspectives for the post-pandemic world: Example from EU. EFB Bioeconomy J.. 2021;1
- [CrossRef] [Google Scholar]
- Improvement on oxidation and storage stability of biodiesel derived from an emerging feedstock camelina. Fuel Process. Technol. 2017
- [CrossRef] [Google Scholar]
- Storage stability of commercially available biodiesels and their blends under different storage conditions. Fuel 2014
- [CrossRef] [Google Scholar]
- Physical and monolayer film properties of potential fatty ester biolubricants. Eur. J. Lipid Sci. Technol.. 2014;116:910-917.
- [CrossRef] [Google Scholar]
- The production of biodiesel from safflower (Carthamus tinctorius L.) oil as a potential feedstock and its usage in compression ignition engine: A comprehensive review. Renew. Sustain. Energy Rev.. 2020;119
- [CrossRef] [Google Scholar]
- Green preparation of branched biolubricant by chemically modifying waste cooking oil with lipase and ionic liquid. J. Clean. Prod.. 2020;274
- [CrossRef] [Google Scholar]







