Translate this page into:
Identification and quality evaluation of Citrus reticulata cv. Chachiensis varieties based on SNP markers
⁎Corresponding authors. zqy@scau.edu.cn (Qunyu Zhang), Wh@scau.edu.cn (Hong Wu), liuwei@scau.edu.cn (Wei Liu)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The desiccative and mature pericarp of Citrus reticulata cv. Chachiensis (PCRC) contenting rich flavonoids and volatile oil is used as traditional Chinese medicine. However, the biological and mechanical mixing of germplasm seriously affects the quality and clinic efficacy of PCRC. In this study, an identification system based on SNP markers was established which is capable of effectively distinguishing between two main C. reticulata cv. Chachiensis (CRC) varieties of DaZhongyoushen (DZ) and XiZhongyoushen (XZ). Moreover, mechanical mixing was confirmed to exist in another major cultivar DaDong5 (DD). Although the morphological characteristics of DZ and XZ fruits were similar, differences were observed in the contents and compositions of bioactive metabolites between the pericarps of these two CRC varieties. The content of flavonoids, especially hesperidin, in the DZ pericarp was significantly higher than that in the XZ pericarp (P < 0.01). The XZ pericarp contained many specific volatile compounds, and its volatile oil content was slightly higher than that of the DZ pericarp. In conclusion, DZ is more suitable as raw material for producing medicinal PCRC, while XZ is more promising for the development of fragranced products such as Ganpu tea. This study provides the theoretical basis and technical support for the identification and evaluation of CRC germplasm resources, which is instrumental for CRC germplasm resource innovation, further promoting the production of high-quality PCRC.
Keywords
Citrus reticulata cv. Chachiensis
Molecular markers
Variety identification
Bioactive metabolites
Quality evaluation
Germplasm innovation fundamental
1 Introduction
The desiccative and mature pericarp of Citrus reticulata cv. Chachiensis (PCRC) has the effects of regulating Qi, strengthening the spleen, desiccativeing dampness and resolving phlegm (Chinese Pharmacopoeia Commission, 2020). The bioactive metabolites of PCRC mainly include flavonoids, volatile oil, polysaccharides and alkaloids (Luo et al., 2019), which have hypolipidemic, antidepressant and analgesic effects (Radulović et al., 2013; Pinheiro et al., 2014; Correa et al., 2016; Gao et al., 2019; Xiao et al., 2022). In addition, PCRC has a potential preventive effect on the Corona Virus Disease 2019 with the rich flavonoids (Haggag et al., 2020; Liang et al., 2022). It is also widely used as flavored and fragranced products with its high-content volatile oil and pleasant fragrance.
C. reticulata cv. Chachiensis (CRC) belonging to Rutaceae has a long history of cultivation (China National Knowledge Infrastructure, 2011a; Song et al., 2019) and has developed several varieties, including DaZhongyoushen (DZ), XiZhongyoushen (XZ), Dadi, Gaodi, Duanzhimiye and DaDong5 (DD) (Jian, 1988; China National Knowledge Infrastructure et al., 2011b, 2011c, 2011d). Among these varieties, DZ, XZ and DD (bred from DZ) exhibit good quality and high yield (Jian, 2009), and thus are currently the main CRC varieties cultivated in Xinhui District of Guangdong Province, China (China National Knowledge Infrastructure, 2011b, 2011c). The synthesis and accumulation of bioactive metabolites of Chinese medicinal materials are affected by many factors such as plant varieties and cultivation areas (Li et al., 2013; Li et al., 2020). For example, the anti-inflammatory and antitussive effects of fritillariae cirrhosae bulbus fluctuate with its variety and origin (Chen et al., 2017). Similarly, different CRC germplasm also shows different clinic efficacies. Traditionally, DZ is considered the best raw material for PCRC (Jian, 2009). In addition, the long-term hybridization of germplasm has led to the decline in the quality and yield of medicinal plants (Huang et al., 2020; Zhou et al., 2021). In practice, different CRC varieties are usually mixed cultivated since it is often difficult to identify them through morphological characteristics, which further results in the increasingly serious hybridization of CRC varieties and huge obstruction of germplasm innovation, directly affecting the production of high-quality PCRC and the protection of high-quality germplasm resources. In this study, an identification system was established based on SNP markers from genome sequencing, which was then employed to distinguish the two main CRC varieties, DZ and XZ, as well as to evaluate the quality of their pericarps. The results of this study provided scientific basis and technical support for the preservation and innovation of CRC germplasm and the production of high-quality PCRC.
2 Materials and methods
2.1 Plant materials
The sampling CRC trees were preliminary classified as DZ, XZ and DD according to Jian (2009). The leaves were taken from the three CRC varieties, respectively, as reference samples to establish the identification system. The identified fruits were used for morphological observation. All plant materials were collected from Dawantou Orchard, Xinhui District of Guangdong Province, China (22°24′ N, 113°1′ E) in November 2019. In addition, random sampling was performed in three additional orchards in Xinhui District (Fig. 2A). A total of 106 test samples were collected from the 4 above orchards to validate the identification system, and defined as Samples-1, −2, −3 and −4.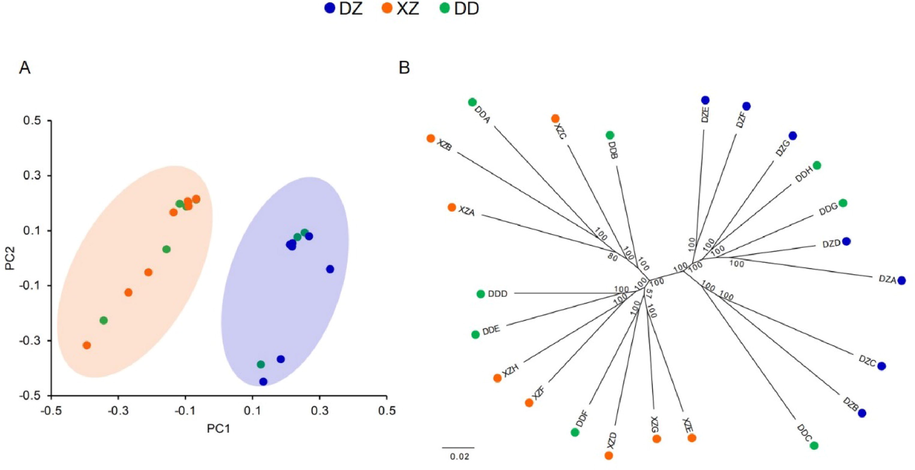
Classification of the reference samples. Blue, orange, and green dots represent reference samples for DZ, XZ, and DD respectively. Samples for DZ and XZ can be independently clustered, while samples for DD is dispersed in both clusters. (A) PCA of the 23 reference samples based on 545,017 SNPs. PC1 indicates the primary significant differences among samples (among varieties), and PC2 shows the secondary significant differences among samples (within a variety). (B) Phylogenetic tree of the reference samples based on 545,017 SNPs.
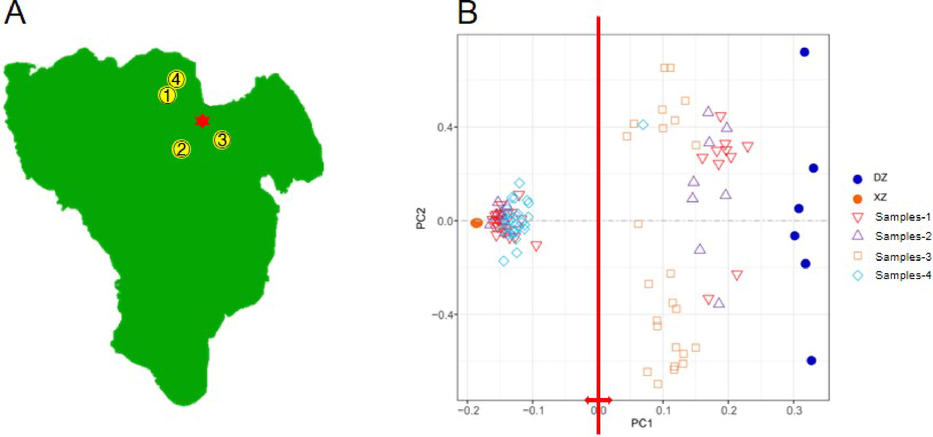
Variety identification of the 106 test samples. (A) Geographic outline of Xinhui District of Guangdong Province, China. The red asterisk indicates Xiongzi Tower, where the produced PCRC is considered to have the best quality. The yellow dots with numbers represent the locations of the sampling orchards. (B) PCA results of the test samples collected from the four orchards based on the identification SNPs. The left side of the red line (negative PC1 values) represents the XZ cluster, while the right side (positive PC1 values) represents the DZ cluster.
2.2 Establishment of the identification system
2.2.1 DNA extraction and sequencing
Genomic DNA of 23 reference samples (i.e., DZA-G, XZA-H and DDA-H) was extracted using a plant genomic DNA kit (#CW0553S; CWBIO, Nanjing, China). The extracted genomic DNA was quality-tested by 1% agarose gel electrophoresis, and quantified with NanoDrop 2000 (Thermo Fisher Scientific, Waltham, MA, US) and Invitrogen Qubit Fluorometer (Life Science, USA). Paired-end sequencing libraries were prepared following the Illumina DNA library protocol with 350-bp insert size. The libraries were quantified with Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA, USA) and sequenced using an Illumina NovaSeq 6000 platform (Illumina, San Diego, CA, USA) with 150 bp read length.
2.2.2 SNP calling and establishment of the identification system
Sequencing data were quality-controlled by FastQC (v0.11.9), and clean reads were aligned to the C. reticulata reference genome (Wang et al., 2018) using BWA (v0.7.17-r1188) (Li et al., 2010) with mem method using default parameters. SNPs were called using DeepVariant (v1.3.0) with the WGS model (Poplin et al., 2018). SNPs that minor allele frequency (MAF) < 0.05 or more than 50% of the samples that not coverage with reads were removed. The principal component analysis (PCA) was performed using PLINK and GCTA (Purcell et al., 2007; Yang et al., 2011). Phylogenetic analysis was conducted based on SNPs and maximum likelihood (ML) method using IQ-TREE2 (v2.0.5) (Minh et al., 2020), with the best fit model TVM + F + R3 base on BIC, and 1000 replicates for ultrafast bootstrap test.
2.2.3 Simplification and verification of the identification system
The fixation indexs (FST) for SNPs were calculated using the R package ‘hierfstat’ in diploids mode following Nei (1987) methods. The SNPs that FST = 1 were complete genetic differentiation positions, and defined as ‘identification SNPs’. The ‘identification SNPs’ were used to divide a total of 106 test samples into different groups by PCA as a verification of the identification system.
2.3 Determination of the characteristics of CRC varieties
2.3.1 Morphological characterization
The fruit weight, fruit shape index (ratio of longitudinal diameter to transverse diameter), seed abortion rate (shriveled seeds were considered aborted), fresh pericarp water content, desiccative pericarp yield per fruit, the weight and thickness (at the equatorial plane) of pericarp, and the number and diameter of sub-dermal secretory cavities were measured.
2.3.2 Bioactive metabolites contents
The volatile oil content of the fresh pericarp was determined according to the Chinese Pharmacopoeia Commission (2020).
The content of total flavonoids in the fresh pericarp was determined by the aluminum nitrate chromogenic with rutin as standards.
2.3.3 Aroma and volatile oil compounds
The volatile oil compounds were analyzed by gas chromatography-mass spectrometry (GCMS) after dehydration by anhydrous sodium sulfate and dilution by dichloromethane. Component analysis used Shimadzu QP-2010 Ultra GCMS system with a chromatographic capillary column (RTX-Wax, 30 m × 0.25 mm × 0.25 μm). The initial temperature of the oven was set at 40 °C (isothermal for 3 min) after which it was increased to 70 °C at the rate of 10 °C/min followed by increasing it to 200 °C at the rate of 5 °C /min for 3 min isothermal. Fragments of 40 to 550 amu were taken at 70 ev. Total GC running time was 30 min. Carrier gas, helium (99.999%) with a flow rate of 1 mL/min; injection volume 0.2 μL without split and injector temperature 240 °C were used.
Headspace gas chromatography (HS-GC) was used to analyze the aroma compounds in the fresh pericarp at room temperature of each CRC variety. Place the pericarp in a beaker sealed with aluminum foil at room temperature for 15 min. Immerse the adsorption column into the beaker for adsorption for 30 min, and then take out the column for detection using the same procedure with GCMS.
Compounds detected by HS-GC and GCMS with SI < 85 were eliminated. The relative content was calculated according to the peak-area ratio.
0.60 g of pericarp was freeze grinded and ultrasound extractioned in 5 mL of methanol for 45 min. After soak for 20 h and dilution by 100 times, 1 mL of the extracting solution was filtered with 0.22 μm membrane into autosampler vials. Liquid chromatography-mass spectrometry (HPLC-MS) was used to determine the contents of nobiletin, hesperidin and tangeretin in the fresh pericarp (Karthikeyan et al., 2021).
2.4 Statistical analysis
Independent T-test of data obtained from each experiment mentioned above was assessed using SPSS Statistics (v22; IBM, NY, USA) to indicate statistical significance between groups. The least significant difference (LSD) method was used for mean variable comparison at P < 0.05 for all statistical tests. All experiments were performed at least in triplicate. Data presented as means values ± standard error (SE).
3 Results
3.1 Establishment of the identification system for CRC varieties
The sequencing information of the 23 reference samples was shown in Supplementary Table S1. A total of 3,966,077 SNPs were detected across the 23 reference samples, and 545,017 SNPs were selected. Results of PCA on all selected SNPs showed that DZ and XZ groups were distributed on different sides of PC1, while reference samples of DD were divided into both two sides (Fig. 1A; Supplementary Table S2). It indicated that there are significant genotypic differences between DZ and XZ. A negative value was indicated XZ, while a positive value represented DZ. Phylogenetic analysis also divided DZ and XZ into two main groups (Fig. 1B). Both PCA and phylogenetic analysis showed that DZ and XZ were two main groups of CRC. However, DD was significantly not an independent group, which could not be distinguished from the other two varieties. Variety DD is considered to be bred from DZ, while some of DD samples were determined as XZ, suggesting mechanical mixing in DD. The remaining DD samples were clustered with DZ without obvious boundaries, indicating that the genetic background of DD and DZ was not significantly different. Therefore, DD samples were excluded from the subsequent analysis.
A total of 376 SNPs with complete genetic differentiation of DZ and XZ (FST = 1) were found (Supplementary Table S3), of which 56.91% were transition mutations and 43.09% were transversion mutations. These SNPs were distributed in the intergenic regions (217; 57.71%), the genic regions (65; 17.29%), upstream of genes (52; 13.83%) and downstream of genes (42; 11.17%), with 7 non-synonymous mutations occurred in the codon regions (Table 1). Note: CR, Citrus reticulata.
Chr.
POS
Gene ID
Note
Function
Amino acid alteration
CR
DZ
XZ
Chr1
12,362,823
LOC102610930
Uncharacterized
–
I108
I108
I108M
Chr1
16,451,243
LOC102629499
Probable disease resistance protein At5g63020
Disease resistance (Lin et al., 2021)
D103
D103
D103G
Chr1
17,608,279
LOC102611408
Probable LRR receptor-like serine/threonine-protein kinase At3g47570
Disease and possible drought resistance (Xu et al., 2017; Zhang et al., 2019)
F470
F470
F470L
Chr3
9,239,579
LOC102613243
Probable disease resistance protein At4g27220
Disease resistance (Li et al., 2018)
Q1135
Q1135E
Q1135
Chr3
17,234,844
LOC102610252
Probable disease resistance protein At4g27220
H1078
H1078N
H1078
Chr9
12,430,161
LOC107178568
Disease resistance protein RGA2-like
Disease resistance (Marimon et al., 2020)
A760
A760T
A760
Chr9
15,182,347
LOC102631206
Putative lipase YOR059C
Maintains lipid droplet morphology (Selvaraju et al., 2014)
R584
R584
R584Q
3.2 Validation of the identification system for CRC varieties
The sequencing information of the 106 test samples was shown in Supplementary Table S1, and their geographic location information was shown in Fig. 2A. PCA was performed on these 106 test samples using the 376 identification SNPs which were complete genetic differentiation of DZ and XZ. In the identification of Samples1-4, PCA analysis showed that the samples were divided into two groups with clear boundaries by PC1, which formed two distinct groups within reference samples of DZ and XZ, respectively. PC1 was the main reference for variety identification. The differences displayed by PC2 showed the richness of genetic diversity within their genetic diversity. PC1 assigned the 106 test samples to either DZ or XZ (Fig. 2B), indicating that the identification SNPs were sufficient and capable for CRC variety identification, and the identification system was reliable.
The samples belonging to XZ were more concentrated on the PC1, while the samples assigned to DZ were more scattered (Fig. 2B), indicating that the genetic diversity among DZ samples is higher. The assignment of Samples-3 and −4 was relatively centralized, with 96.67% of Samples-3 being assigned to XZ and all of Samples-4 being assigned to DZ. On the contrary, the assignment of Samples-1 and −2 were diverse, with 68.75% of Samples-1 and 60.00% of Samples-2 belonging to XZ, and 31.25% of Samples-1 and 40% of Samples-2 being DZ (Supplementary Table S4).
3.3 Comparisons of morphological characteristics and bioactive metabolites contents between the two CRC varieties
Variety DZ had a greater fruit and fresh pericarp weight, while XZ had a higher desiccative pericarp weight and thickness as well as a desiccative pericarp yield per fruit (Fig. 3A). The number of sub-dermal secretory cavities in the DZ pericarp was smaller, while the diameters of those in the XZ pericarp were larger (Fig. 3B). However, the differences in the above indices between the two CRC varieties were not significant which indicating the difficulty of distinguishing DZ and XZ fruits according to morphological characteristics with the naked eye (Supplementary Table S5). Moreover, XZ shows advantages over DZ in volatile oil content of flesh pericarp.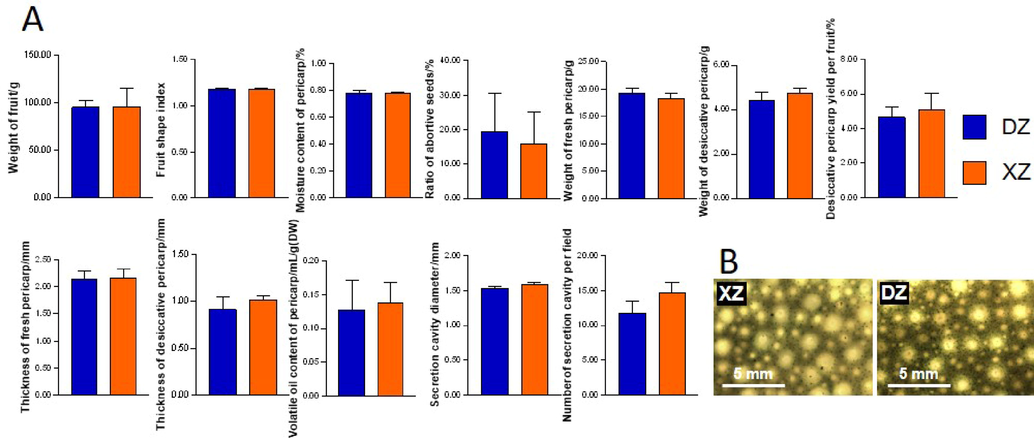
Morphological characteristics and volatile oil contents of the two CRC varieties. (A) Characteristics of the fruit, pericarp, and volatile oil content (n = 3) of the two CRC varieties. The differences in these indices between DZ and XZ were not significant. (B)Sub-dermal secretory cavities in the fresh pericarp of the two CRC varieties (scale bar = 5 mm).
The total flavonoid content was higher in DZ than that in XZ. Among the flavonoids, the contents of nobiletin, hesperidin and tangeretin were all higher in DZ (Fig. 4); specifically, the hesperidin content was significantly higher (P < 0.01).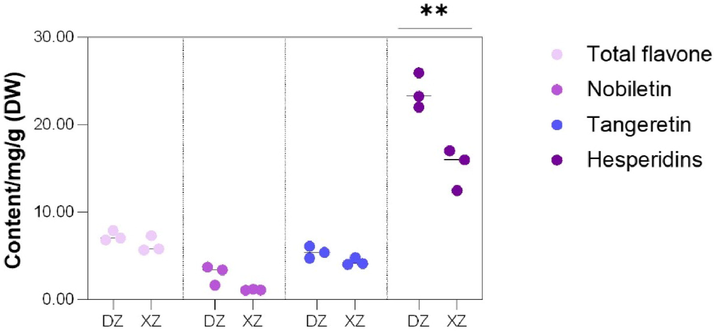
Comparisons of the contents of total flavonoids and individual flavonoid compounds between pericarp of the two CRC varieties. Asterisks indicate significant differences between samples (T-test; ∗∗, P < 0.01).
3.4 Comparisons of aroma and volatile oil compounds between the two CRC varieties
Approximately 30 available aroma compounds (at room temperature) and 40–50 available volatile oil compounds were detected (Supplementary Tables A6 and A7). The aroma and volatile oil compounds of the two CRC varieties were both dominated by terpenes, followed by alcohols and lipids. Compared with DZ, XZ had significantly lower contents of alcohols but significantly higher contents of ketones and phenols. (P < 0.05; Table 2). Note: Group A includes the aroma compounds at room temperature; Group B indicates the volatile oil compounds (n = 3). Asterisks indicate significant differences between samples (T-test; ∗, P < 0.05). ‘-’ indicates that the item was not detected in all three repetitions.
Group
Variety
Terpenes (%)
Alcohols (%)
Lipids(%)
Ketones (%)
Phenols (%)
A
DZ
95.26 ± 0.78
1.51 ± 0.81*
1.36 ± 0.77
–
–
XZ
97.10 ± 0.26
0.62 ± 0.09*
0.81 ± 0.19
–
–
B
DZ
92.75 ± 0.61
1.51 ± 0.24
2.44 ± 0.22
0.03 ± 0.01*
0.07 ± 0.00*
XZ
95.45 ± 0.46
1.11 ± 0.08
2.35 ± 0.30
0.04 ± 0.00*
0.10 ± 0.02*
Methyl N-methylanthranilate is widely considered as the unique volatile component of PCRC(He et al., 2020; Yu et al., 2020) and was identified as one of the volatile compounds in the pericarps of the two CRC varieties. The contents of methyl N-methylanthranilate and linalool in the XZ pericarp were significantly higher than those in the DZ pericarp (P < 0.05). Compounds that were detected in all three replicates of one variety while not detected in all three replicates of the others were defined as the specific compound. Accordingly, the specific aroma compound in DZ was α-phellandrene, while those of XZ included leaf acetate, β-cadinene, α-copaene and (2S)-2-amino-N-ethylpropanamide (Fig. 5A; Supplementary Table S2).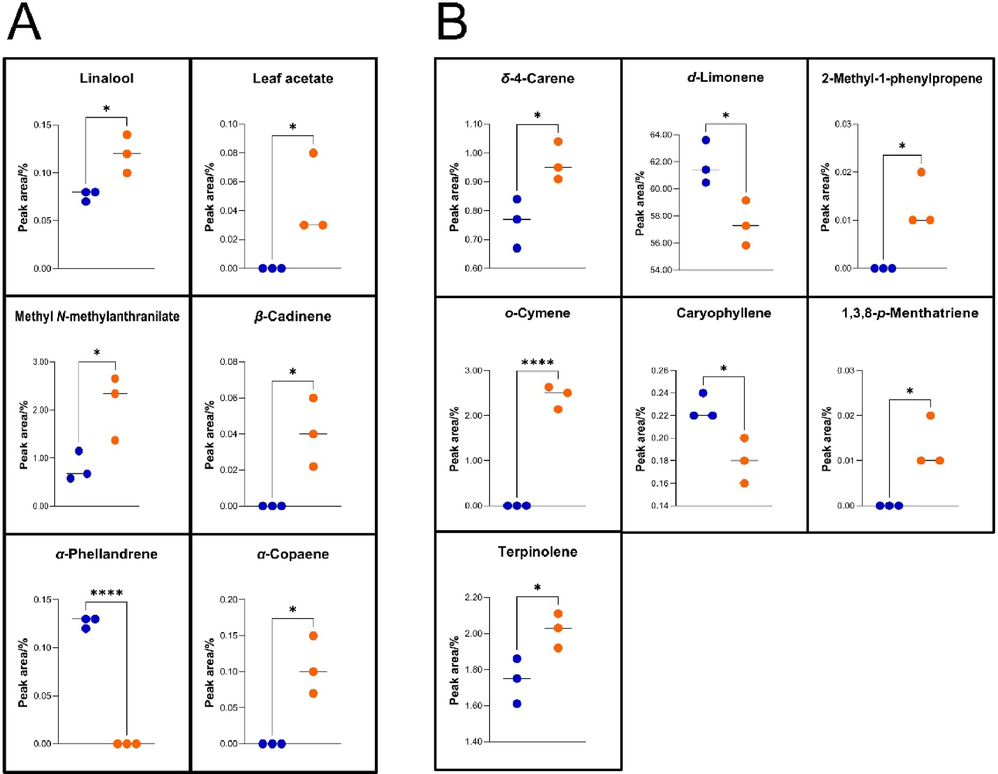
Volatile compounds with significantly different contents between the two CRC varieties. The content was calculated based on the peak-area ratio in the detection results of HS-CG (for aroma compounds) or GCMS (for volatile oil compounds). (A) Aroma compounds (n = 3); (B) volatile oil compounds (n = 3). Asterisks indicate significant differences between samples (T-test; ∗, P < 0.05; ∗∗∗∗, P < 0.0001).
d-Limonene accounted for more than 50% of the total volatile oil content in both XZ and DZ pericarps, suggesting that it is the main compound of PCRC volatile oil. The contents of d-limonene, terpinolene and caryophyllene in DZ were significantly higher, while δ-4-carene was significantly lower compared with those in XZ (P < 0.05). The specific volatile oil compounds in DZ were oxygenates, including (+)-trans-limonene oxide, (+)-citronellal, perillaldehyde, p-cymen-8-ol, (4S,5R)-5-hexyl-4-methyloxolan-2-one, trans-p-mentha-2,8-dienol, cis-p-mentha-2,8-dien-1-ol, bergamotenol and tetradecanal, while those in XZ were not only oxygenates (2-isopropyl-4-methyl anisole, dodecanal, nerol, perillyl alcohol and alpha-elemol), but also hydrocarbons, such as o-cymene, β-elemene, 2-methyl-1-phenylpropene and 1,3,8-p-menthatriene (Fig. 5B; Supplementary Table S2).
4 Discussion
4.1 Variety purification is beneficial for accurate breeding and germplasm innovating of CRC, and stabilizing PCRC quality
The traditional identification methods for CRC varieties mainly rely on the morphological characteristics (Jian, 2009). Our results showed no significant difference in the morphological characteristics between identified DZ and XZ, indicating that identification methods based on morphological characteristics is of low accuracy, which might lead to mechanical and biological mixed cultivating of CRC germplasm. Among the sampling orchards, the XZ and DZ varieties were found to be mechanically mixed in two orchards. Moreover, the DZ samples were clustered more dispersedly in PCA (Fig. 2B), suggesting that biological mixing might also exist.
SNP markers are widely applied in the identification of citrus plants (Endo et al., 2020; Jin et al., 2020), and its combination with PCA is considered more conducive to improving identification accuracy (Horne et al. 2004; Gauch et al. 2019). In this study, SNP markers and PCA were combined to establish an effective system for identification of CRC varieties, which provided technical support for identification and conservation of CRC germplasm resources.
The Citrus Clonal Protection Program (CCPP) is implemented by the University of California, the U.S. Department of Agriculture, the California Department of Food and Agriculture, and the California Citrus Nursery Board (Wang et al., 2013). The basic idea of CCPP is to maintain the characteristics of citrus varieties and to use the most typical and healthy maternal plants as the only source of scions. At present, serious biologically and mechanically mixed cultivation exists in CRC production. The low purity of CRC seedlings will directly affect the stability of their excellent traits, leading to uneven quality of PCRC. The identification system of CRC varieties can be applied to identify the maternal plant, which would provide guarantees for the promotion of high-quality CRC seedlings and fundamental for the innovation of germplasm with clear variety information. Moreover, the stable quality of PCRC (a traditional Chinese medicinal material) and sustainable development of the CRC industry can also be ensured by improving the purity of CRC varieties.
4.2 Differences in bioactive metabolites between DZ and XZ determine their application values
PCRC can be used as both medicine and food (Shi et al., 2020). However, only PCRC with the hesperidin, nobiletin and tangeretin contents passing the thresholds can be used as traditional Chinese medicine (Chinese Pharmacopoeia Commission, 2020). In addition, PCRC has a strong aroma and is also popular as fragranced products such as Ganpu tea (Qi et al., 2020). The contents of hesperidin, nobiletin and tangeretin were higher in the DZ pericarp, and the difference in the hesperidin content reached a significant level (P < 0.01). Therefore, DZ should be protected and utilized as the high-quality raw material for PCRC. In addition, the diverse genetic background of DZ provides abundant breeding materials for the cultivation of new CRC varieties, which, however, also leads to instability of excellent traits (Saha et al., 2022). Therefore, the screening of high-quality DZ individuals, the cultivation of new CRC varieties and the purification of CRC varieties are of important significance and application potentials. By contrast, the genetic background of XZ samples was relatively uniform; XZ samples exhibited a higher volatile oil content than DZ and contained eight specific volatile compounds. Therefore, XZ may be suitably used to develop volatile oil source or fragranced products.
Jian (2009) reported that DZ not only had high yield, but also showed stronger resistance to stress. Among the identification SNPs, the genes at which non-synonymous mutations occurred were mostly related to disease resistance (Li et al., 2018; Zhang et al., 2019; Lin et al., 2021). In addition, 18 genes located upstream of genes were associated with defense against biotic and abiotic stresses (Supplementary Table S9). The above mutations may cause differences in stress resistance among CRC varieties. In summary, the screening of stress-resistant SNP markers should be performed in future work to benefit molecular marker-assisted breeding in CRC.
5 Conclusion
In this study, an identification system of CRC varieties was established based on SNP markers. This system was capable of distinguishing between DZ and XZ varieties and revealed biological and mechanical mixing within the two varieties. The DZ pericarp contained more flavonoids, among which the content of hesperidin was 23.72 ± 1.16 mg/g, significantly higher than that of the XZ pericarp (15.12 ± 1.37 mg/g; P < 0.01). The volatile oil content in the XZ pericarp was 0.05 mL/g higher than that in the DZ pericarp; it included eight specific volatile compounds and was richer in aroma. In conclusion, DZ is more suitable as the raw material for PCRC, while XZ has wider application prospects in the development of fragrance products.
Acknowledgments
This work was supported by the Open Competition Program of Ten Major Directions of Agricultural Science and Technology Innovation for the 14th Five-Year Plan of Guangdong Province [grant numbers 2022SDZG07], the Laboratory of Lingnan Modern Agriculture Project [grant numbers NZ 2021024], the Key realm R&D Program of Guangdong Province [grant numbers 2020B020221001] and Guangdong Provincial Special Fund for Modern Agriculture Industry Technology Innovation Teams [grant numbers 2019KJ125]. We would like to express our special thanks to Mr. Shesheng Su and other citrus growers for their cooperation.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- H.Z.Chen S.Y.Zhang Y.B.Huang S.M.Li J.F.Chen H.AoComparative study on the content determination and the anti-tussive, anti-inflammatory effects of the total alkaloids of Pingbeimu and ChuanbeimuSci. Technol. Food Ind.3820176367doi:10.13386/j.issn1002-0306.2017.15.014
- China National Knowledge Infrastructure, 2011a. Comprehensive utilization and development of Citrus reticulata cv. Chachiensis as raw material of Xinhui Citri Reticulatae Pericarpium. https://t.cnki.net/kcms/detail?v=HFVoyEkArlG7UbTv9YLOIOxILD5vY-5cLzgv12jDkYjvB6rZproKTHE3gcXuSHYIBtwq0o11PIwROc5yczfHRw3nEtme2M6e0CCc4a9GMIm7JVi0TwCAxw==&uniplatform=NZKPT (accessed 10 May 2022).
- China National Knowledge Infrastructure, 2011b. Cultivate industrial resources and promote industrial development. https://t.cnki.net/kcms/detail?v=HFVoyEkArlGnjZvvYACDx9sMbFreuALauV0bZpDhWzxUKrVccsU37RCTBV96U-Gs4T_fsYT3i2EB1LUuKouyNr4cBR_ST3Ju5nMv9QP6EJnvzL65uxOJdA==&uniplatform=NZKPT (accessed 09 October 2021)
- China National Knowledge Infrastructure, 2011c. Cultivation history and variety research of Citri Reticulatae ‘chachi’. https://t.cnki.net/kcms/detail?v=HFVoyEkArlF7ZKR4L_66a6XtIz1EnUISnULVoU4ypZgV9jUlW0fPKXYQcLADYrYxllbKK2bBhoYzKkUA4efHlrh9RjZwOyWmrF36lFH6rFwBv4N3HP8DGg==&uniplatform=NZKPT (accessed 23 July 2022)
- China National Knowledge Infrastructure, 2011d. Geoherbalism code interpretation of Xinhui citri reticulatae pericarpium. https://t.cnki.net/kcms/detail?v=HFVoyEkArlGOOH5S0whrFaSkNa-bNzj6JoGUS4R2izXyrrkoaORBR0ZDNOmxK7gBrUgeIB0zYe4Q0SM3Zq14-QnfwFC6mYH0XTFd-oGdA-Y5PPARp3lkZg==&uniplatform=NZKPT (accessed 14 June 2022)
- Chinese Pharmacopoeia Commission, 2020. Pharmacopoeia of the People's Republic of China: volume I. Beijing. ISBN 9787506700778.
- Methyl-N-methylanthranilate, a pungent compound from Citrus reticulata Blanco leaves. Pharm. Biol.. 2016;54:569-571.
- [CrossRef] [Google Scholar]
- TaqMan-MGB SNP genotyping assay to identify 48 citrus cultivars distributed in the Japanese market. Breed. Sci.. 2020;70:363-372.
- [CrossRef] [Google Scholar]
- Enrichment of polymethoxyflavones from Citrus reticulata ‘Chachi’ peels and their hypolipidemic effect. J. Chromatogr. B. 2019;1124:226-232.
- [CrossRef] [Google Scholar]
- Consequences of PCA graphs, SNP codings, and PCA variants for elucidating population structure. PLoS ONE. 2019;14:6.
- [CrossRef] [Google Scholar]
- Is hesperidin essential for prophylaxis and treatment of COVID-19 Infection? Med. Hypotheses. 2020;144:109957
- [CrossRef] [Google Scholar]
- Specificity analysis of essential oil from Pericarpium Citri Reticulatae ‘Chachiensis. Modern Food Science and Technology. 2020;36:224-231.
- [CrossRef] [Google Scholar]
- Principal component analysis for selection of optimal SNP-sets that capture intragenic genetic variation. Genet. Epidemiol.. 2004;26:11-21.
- [CrossRef] [Google Scholar]
- Investigation of medicinal Chrysanthemum germplasm and analysis of industrial status in China. J. Chinese Med. Mater.. 2020;43:1325-1329.
- [CrossRef] [Google Scholar]
- Jian, C.B., 1988. Standardization cultivation of citrus in Xinhui. South China Fruits. 17, 30-31. https://kns.cnki.net/kcms/detail/detail.aspx?dbcode=CJFD&dbname=CJFD8589&filename=FRUI198803017&uniplatform=NZKPT&v=LQ4zpHp_w9pE8MRBB8RvJETGRIA9U7pgfCkDtMjPxb0QVqbT9x4kCcw6S-SvgtYt
- Jian, C.B., 2009. Citrus and its cultivation in Xinhui. Guangzhou, Guandong. ISBN 978-7-5359-5028-4
- Identification of the ‘Haryejosaeng’ mandarin cultivar by multiplex PCR-based SNP genotyping. Mol. Biol. Rep.. 2020;47:8385-8395.
- [CrossRef] [Google Scholar]
- Assessment of anti-inflammatory and antioxidant effects of Citrus unshiu peel (CUP) flavonoids on LPS-Stimulated RAW 264.7 cells. Plants. 2021;10:2209.
- [CrossRef] [Google Scholar]
- Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics. 2010;26:589-595.
- [CrossRef] [Google Scholar]
- Analysis and evaluation of essential oil components of cinnamon barks using GC-MS and FTIR spectroscopy. Ind. Crops Prod.. 2013;41:269-278.
- [CrossRef] [Google Scholar]
- The effect of developmental and environmental factors on secondary metabolites in medicinal plants. Plant Physiol. Biochem.. 2020;148:80-89.
- [CrossRef] [Google Scholar]
- Heterologous expression of the cotton NBS-LRR gene GbaNA1 enhances Verticillium wilt resistance in Arabidopsis. Front. Plant Sci.. 2018;9:119.
- [CrossRef] [Google Scholar]
- Study on flavonoid and bioactivity features of the pericarp of Citri Reticulatae ‘chachi’ during storage. Arab. J. Chem.. 2022;15:103653
- [CrossRef] [Google Scholar]
- Anthocyanins contribute to fruit defense against postharvest green mold. Postharvest Biol. Tec.. 2021;181:111661
- [CrossRef] [Google Scholar]
- Discrimination of Citrus reticulata Blanco and Citrus reticulata ‘Chachi’ as well as the Citrus reticulata ‘Chachi’ within different storage years using ultra high performance liquid chromatography quadrupole/time-of-flight mass spectrometry based metabolomics approach. J. Pharm. Biomed.. 2019;171:218-231.
- [CrossRef] [Google Scholar]
- Fine mapping and identification of candidate genes for the peach powdery mildew resistance gene Vr3. Hort. Res.. 2020;7:175.
- [CrossRef] [Google Scholar]
- IQ-TREE 2: new models and efficient methods for phylogenetic inference in the genomic era. Mol. Biol. Evol.. 2020;37:1530-1534.
- [CrossRef] [Google Scholar]
- Nei, M., 1987. Molecular Evolutionary Genetics. New York, NY. https://doi.org/10.7312/nei-92038
- Antinociceptive esters of N-methylanthranilic acid: mechanism of action in heat-mediated pain. Eur. J. Pharmacol.. 2014;727:106-114.
- [CrossRef] [Google Scholar]
- A universal SNP and small-indel variant caller using deep neural networks. Nat. Biotechnol.. 2018;36:983-987.
- [CrossRef] [Google Scholar]
- PLINK: A tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet.. 2007;81:559-575.
- [CrossRef] [Google Scholar]
- Characteristic volatile fingerprints and odor activity values in different Citrus-Tea by HS-GC-IMS and HS-SPME-GC-MS. Molecules. 2020;25:6027.
- [CrossRef] [Google Scholar]
- Effects of Methyl and Isopropyl N-methylanthranilates from Choisya ternata Kunth (Rutaceae) on experimental anxiety and depression in Mice. Phytother. Res.. 2013;27:1334-1338.
- [CrossRef] [Google Scholar]
- Rice landraces from haor areas of Bangladesh possess greater genetic diversity as revealed by morpho-molecular approaches along with grain quality traits. Cogent Food Agric.. 2022;8:2075130.
- [CrossRef] [Google Scholar]
- Identification of a phospholipase B encoded by the LPL1 gene in Saccharomyces cerevisiae. BBA-Mol. Cell Biol. L. 2014;1841:1383-1392.
- [CrossRef] [Google Scholar]
- A rapid protocol to distinguish between Citri Exocarpium Rubrum and Citri Reticulatae Pericarpium based on the characteristic fingerprint and UHPLC-Q-TOF MS methods. Food Funct.. 2020;11:3719-3729.
- [CrossRef] [Google Scholar]
- Textual research on Citri Reticulatae Pericarpium, Pericarpium Citri Reticulatae ‘chachiensis’ and Xinhui Citri Reticulatae Pericarpium. J. Chinese Med. Mater.. 2019;42:453-458.
- [CrossRef] [Google Scholar]
- Past and future of a century old Citrus tristeza virus collection: a California citrus germplasm tale. Front. Microbiol.. 2013;4:366.
- [CrossRef] [Google Scholar]
- Genome of wild mandarin and domestication history of mandarin. Mol. Plant. 2018;11:1024-1037.
- [CrossRef] [Google Scholar]
- The antihyperlipidemic equivalent combinatorial components from peel of Citrus reticulata ‘Chachi’. J. Food Drug Anal.. 2022;30:77-87.
- [CrossRef] [Google Scholar]
- Transcriptional factors for stress signaling, oxidative protection, and protein modification in ipt-transgenic creeping bentgrass exposed to drought stress. Environ. Exp. Bot.. 2017;144:49-60.
- [CrossRef] [Google Scholar]
- GCTA: A tool for genome-wide complex trait analysis. Am. J. Hum. Genet.. 2011;88:76-82.
- [CrossRef] [Google Scholar]
- Chemical composition of volatile oil from Citri Reticulatae Pericarpium and antioxidant activity of individual compounds in volatile oil. Food and Fermentation Industry. 2020;47:245-252.
- [CrossRef] [Google Scholar]
- Transcriptome-wide alternative splicing modulation during plant-pathogen interactions in wheat. Plant Sci.. 2019;288:110160
- [CrossRef] [Google Scholar]
- Study on the current situation and development countermeasures of Carthamus tinctorius in Xinjiang. J. Agric. Sci.. 2021;49(199–201):217.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.104948.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







