Translate this page into:
Maximizing fermentable feedstocks from Camellia oleifera seed oil extraction residues: Green pretreatment and enzymatic hydrolysis for effective valorization
⁎Corresponding authors at: Hunan Engineering Technology Research Center for Comprehensive Development and Utilization of Biomass Resources, College of Chemistry and Bioengineering, Hunan University of Science and Engineering, 425199 Yongzhou, Hunan, China (W. Dessie). dwubliker@yahoo.com (Wubliker Dessie), lichangzhu2013@aliyun.com (Changzhu Li), dong6758068@163.com (Zuodong Qin)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
This study aimed to convert wastes generated during the extraction of oil from Camellia oleifera seeds, including the fruit shell (COFS) and seed cake (COSC), into fermentable feedstocks using pretreatment and enzymatic hydrolysis processes. The application of a green acid catalyst in the thermochemical pretreatment of the COFS and COSC mixture yielded the highest sugar of 28 g/L. Solid-state fermentation (SSF) was conducted on-site using various waste biomass substrates to produce enzymes for the saccharification process. This study found that COFS was the ideal substrate for generating SSF enzymes capable of breaking down lignocellulosic biomass. A novel enzymatic hydrolysis strategy was developed to minimize water consumption and enhance substrate utilization efficiency. By fine-tuning the enzymatic hydrolysis process, an additional sugar of 20.58 g/L was achieved using thermochemically pretreated solid residues and SSF enzymes. Standard techniques were used to analyze the raw materials, residual solids, and hydrolysates to assess the efficiency of each processing step. In general, the use of SSF enzymes and green pretreatment methods, reduction of water consumption in the process, and implementation of an efficient biomass utilization approach, enhance the overall efficiency and eco-friendliness of this study.
Keywords
Camellia oleifera
Lignocellulose pretreatment
Solid-state fermentation
Enzymatic hydrolysis
Fermentable sugar
- COSOER
-
Camellia oleifera seed extraction residue
- COSF
-
C. oleifera fruit shell
- COSC
-
C. oleifera seed cake
- PP
-
Pomelo peel
- PDA
-
Potato dextrose agar
- AH
-
Authohydrolysis
- TH
-
Thermal hydrolysis
- TCH
-
Thermochemical hydrolysis
- SSF
-
Solid-state fermentation
- FTIR
-
Fourier transform infrared spectroscopy
- SEM
-
Scanning electron microscopy
- DNS
-
Dinitrosalicylic acid
- w/v
-
Weight per volume
Abbreviations
1 Introduction
Camellia oleifera is an evergreen plant that belongs to the group of widely cultivated woody oil species worldwide, including in subtropical areas of China and Southeast Asian countries (Deng et al., 2020, Liu et al., 2024). The seeds of C. oleifera are used to produce a high-quality edible oil. Oil extraction process from this seed generates huge amount of waste products. The major C. oleifera seed oil extraction residues (COSOERs) includes C. oleifera fruit shell (COFS) and C. oleifera seed cake (COSC). In order to reduce China's nearly 70 % dependency on imported edible vegetable oil, the country has ambitiously focused on cultivating woody oil crops, including the endogenous C. oleifera (Yang et al., 2022). As a result, the annual fruit yield of camellia in Southern China has reached 7 to 8 million tons (Tang et al., 2022). Related to this, in 2020, the generation of COFS and COSC reportedly reached approximately 3.68 million tons and over 5 million tons, respectively, in this area (Chen et al., 2022, Liu et al., 2024). Generally, these wastes are either disposed of in landfills or burned in oil processing industries. This leads to a significant loss of bioresources and can also result in severe environmental and waste management issues. Given the abundance of COSOERs and the potential hazards associated with their improper disposal, it is essential to develop effective strategies to address this issue.
In recent years, the development of sustainable processes for the production of value-added products from waste biomass has become an important focus of research (Liao et al., 2020). COSOERs contain a significant amount of lignocellulosic components and other essential bioactive ingredients. COFS can contain up to 13.9─21.0 % cellulose, 23.5─31.6 % hemicellulose, and 30.1─36.2 % lignin (Liu et al., 2024). Similarly, COSC comprises nearly 40 % carbohydrates, 15─25 % crude fiber, and 15 % crude protein (Wu et al., 2022, Liu et al., 2024). According to the latest reviews (Quan et al., 2022, Liu et al., 2024), the potential applications of COSOERs have been primarily explored in two areas, namely the extraction of functional bioactive components and the development of bio-based materials and/or chemicals. Nonetheless, although functional bioactive components, such as saponins, tannins, flavonoids, phenolics, and others, are present in COSOER, they only form a small proportion of its composition. Consequently, the extraction of these compounds from COSOERs could potentially result in the generation of a large amount of additional waste materials. Furthermore, the valorization of COSOERs to produce biochar and related products at high operational temperatures (Liu et al., 2022) may result in the unnecessary loss of some biomass components.
An alternative option would be to utilize COSOERs to generate fermentable sugars that can be employed for the production of green fuels and chemicals. The solid residues released afterwards can then be repurposed for other applications (including biochar) (Dessie et al., 2023), facilitating efficient utilization of biomass and easing waste management issues. However, the presence of exceptionally high lignin (Wang et al., 2021) and a hard cell wall structure (Chen et al., 2023) in COFS, as well as the availability of antimicrobial factors such as saponin in COSC (Qian et al., 2018), limit the implementation of the aforementioned strategy. Previous attempts to fractionate lignocellulosic components in C. oleifera fruit hulls (Sun et al., 2016, Tang et al., 2017) involved multi-step pretreatment processes that required the use of large volumes of various chemicals (solvents), as well as a saccharification step utilizing commercial enzymes. This approach would require extensive separation steps, consume a significant amount of time, chemical, energy, water and equipment, which could ultimately increase bioprocessing costs and raise sustainability concerns. The efficiency of converting lignocellulosic biomass into fermentable sugars is a crucial factor in determining the feasibility of value-added production from second-generation feedstocks (Besada-Lombana et al., 2023). At this point, developing simple, efficient and sustainable strategies for converting COSOERs into valuable products is essential.
Advantages such as relatively low cost, high efficiency, minimal generation of inhibitors, and reduced corrosion (Dessie et al., 2022, Nair et al., 2023, Rahim et al., 2023) have sparked growing interest in utilizing oxalic acid for biomass pretreatment. Not only can it be produced from renewable resources (Panyamao et al., 2023), but it can also be recovered, recycled, and degraded during the pretreatment process (Cheng et al., 2018, Hu et al., 2018). Overall, the utilization of oxalic acid represents a promising, greener, and more efficient alternative to traditional pretreatment methods for lignocellulosic biomass. Reports show that the cost of enzymes remains a significant challenge in the production of biofuels and chemicals from lignocellulosic biomass (Carpio et al., 2022), accounting for up to 40 % of the total production cost (Arora et al., 2015). In the current study, on-site enzymes were produced through solid-state fermentation (SSF) using cheap waste biomass resources. SSF technology offers numerous advantages in this area, such as production of high enzyme concentration, minimal energy consumption, easy operation, and reduced environmental impact (Dessie et al., 2018a, Dessie et al., 2018b; Liu et al., 2021).
The objective of this study is to develop efficient pretreatment and enzymatic hydrolysis processes for converting COSOERs into fermentable sugar. The pretreatment, SSF, and enzymatic hydrolysis steps were thoroughly optimized to attain the desired objectives. Simply put, the on-site enzymes produced by SSF were utilized to convert the lignocellulosic components in pretreated COSOERs into fermentable hydrolysates, including sugars. The processing steps implemented in this study were evaluated by characterizing the raw materials, residual solids, and hydrolysates.
The current study addressed the following aspects to achieve the designated objectives:
-
Optimization of COSOERs pretreatment by employing various pretreatment methods with both individual and mixed substrates.
-
Evaluation of fungal SSF performance utilizing (1) different individual and mixed substrates and (2) various concentrations of saponin.
-
Assessment of enzymatic hydrolysis efficiency through three distinct saccharification strategies (Fig. 1).
-
Investigation into the efficiency of each processing step (SSF, pretreatment, and enzymatic hydrolysis) by analyzing fungal growth, characterizing raw materials, solid residues, and hydrolysates of pretreatment and enzymatic hydrolysis.
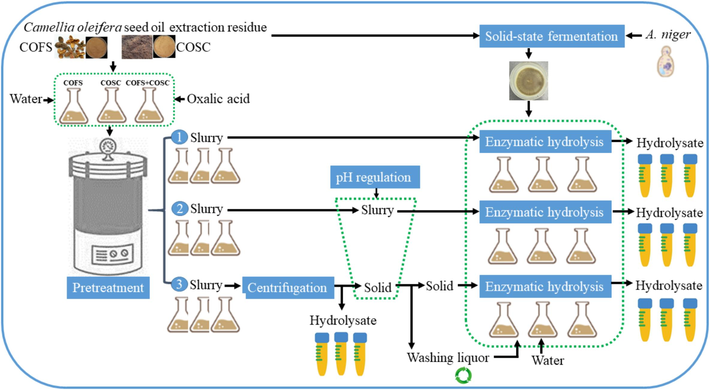
-
C. oleifera seed oil extraction residues pretreatment and enzymatic hydrolysis process.
2 Materials and methods
2.1 Chemicals and raw materials
All the chemicals utilized in this research were of reagent grade. Raw biomass materials such as COFS, COSC, and pomelo peel (PP) were used in this study. COFS and COSC were collected from C. oleifera seed oil extraction cite in Yongzhou City, Hunan Province, China. Fresh pomelo fruits were obtained from nearby fruit shops in Yongzhou City, Hunan Province, China, specifically in proximity to Hunan University of Science and Engineering, and subsequently transported to the laboratory. Following the manual separation of each component of the fruit, the PP was cut into small cubes using a knife and then subjected to drying in an oven at 55 °C for 24 h. These raw materials were smashed with the aid of a stainless-steel mini laboratory grinder and subsequently screened using a laboratory wire mesh sieve with a pore size of 0.5 mm as explained previously (Dessie et al., 2022).
2.2 Pretreatment
In this study, an initial substrate mass ratio of 10 % (w/v) was utilized, where 10 g of substrate was mixed with 100 ml of water for each experiment. The effectiveness of three distinct pretreatment methods (namely autohydrolysis (AH), thermal hydrolysis (TH), and thermochemical hydrolysis (TCH)) was evaluated using COFS, COSC, and a 1:1 mixture of these substrates. AH involved briefly mixing the respective substrates with water at room temperature, while TH and TCH were carried out at 121 °C for 30 min, with the latter involving the addition of 2 % (w/v) oxalic acid as described in previous work (Dessie et al., 2022). To separate the hydrolysates and solid residues, the slurries generated after pretreatment were subjected to centrifugation at 10,000g for 20 min. Following this step, the hydrolysates underwent additional filtration using Whatman no.1 filter paper while the solid residues were dried in an oven at 55 °C for 24 h. The resulting hydrolysates and solid residues were then utilized for subsequent analyses and characterization studies.
2.3 Fungal cultivation
Aspergillus niger was utilized in this study for SSF to produce enzymes capable of degrading COFS and COSC. The fungus was cultured on potato dextrose agar (PDA) and incubated at 30 °C. The spore suspensions were obtained by carefully agitating the agar plate with the addition of sterile water.
2.4 Fungal growth potential
In order to assess the growth potential of A. niger on a range of substrates (including COFS, COSC, and PP), a 10 % (w/v) mass ratio of individual or equal mixed amounts of the substrates were utilized with the addition of 1.5 % (w/v) agar. In addition, the growth potential of A. niger was evaluated using 0, 5, 10, and 15 % saponin mixed relative to the base PDA medium. Following preparation, the media were sterilized at 121 °C for 15 min. A drop of fungal spore suspension was subsequently added to the center of each petri dish, which contained the respective medium (Dessie et al., 2022). These plates were then incubated at 30 °C, and the diameter of the mycelia was measured periodically to determine the radial growth of the fungi.
2.5 Solid-state fermentation
SSF was conducted using 2 g of biomass substrate in petri dishes. The SSF medium was adjusted to nearly 70 % moisture content using distilled water and sterilized at 121 °C for 30 min. A spore suspension of A. niger (2 × 107 spores/mL) was evenly spread on the surface of the sterilized SSF medium and incubated at 30 °C.
2.6 Enzymatic hydrolysis
COFS, COSC, and their mixtures were pretreated using Erlenmeyer flasks, and SSF mashes containing enzyme mixtures with the potential to degrade lignocellulosic biomass were produced, as described in the previous subsections. These pretreatment and SSF products were utilized in enzymatic hydrolysis step. The enzymatic hydrolysis was optimized following three strategies as shown in Fig. 1. Initially, hydrolysate suspension (slurry) was collected after pretreatment of biomass substrate. In the first two strategies, the SSF mash was added to the pretreatment slurry. However, in the second strategy, the pH was adjusted to meet the optimal requirements of the enzyme before starting the enzymatic hydrolysis process. In the third method, the solid residues generated after pretreatment and centrifugation were considered for further enzymatic hydrolysis. The solid was washed by alkaline solution to adjust the pH suitable for hydrolysis. Then, the SSF mash was added to this solid (10 % mass (w/v)), in which the washing liquor was recycled and used for enzymatic hydrolysis (Fig. 1 #3). The pH for the second and the third methods were adjusted to be nearly 5.0 using 1 M NaOH. Enzymatic hydrolysis was conducted at 50 °C and 200 rpm in a shaker for 24 h (Dessie et al., 2022). Samples were collected at regular intervals and subjected to the same procedures outlined in the pretreatment section to obtain hydrolysates and solid residues for subsequent analysis.
2.7 Fourier-transform infrared spectroscopy analysis
Fourier transform infrared spectroscopy (FTIR) was used to analyze the raw substrates, as well as the solid residues generated after pretreatment and enzymatic hydrolysis. FTIR analysis in this study was performed using a Thermo Nicolet IS10 (USA) spectrometer with a scanning range of 400–4000 cm−1, a resolution of 4.0 cm−1, and 32 scans.
2.8 Scanning electron microscopy analysis
The samples utilized for FTIR analysis were also examined for their morphological characterization using a scanning electron microscopy (SEM). For this purpose, a Hitachi SU8010 cold field emission SEM operating at 3.0 kV was employed in the study.
2.9 Reducing sugar determination
The amount of reducing sugar in the hydrolysates was determined using Miller's dinitrosalicylic acid (DNS) method (Miller, 1959) with some modifications. A known amount of the sample and pre-prepared DNS solution were combined in a test tube and heated in a water bath for 10 min. The mixture was then allowed to cool and diluted to a predetermined ratio with water. Absorbance was measured at 540 nm using a GENESYS™ 180 UV–Visible Spectrophotometer (Shanghai, China).
2.10 Statistical analysis
Microsoft Excel and OriginPro 2018 software were employed for statistical analysis. Each experiment was conducted in triplicate, and the resulting values were expressed as the mean of the replicates ± standard deviation. Statistical significance was determined with P values < 0.05.
3 Result and discussion
3.1 Solid-state fermentation
A. niger is a widely recognized industrial strain renowned for its capacity to efficiently utilize various waste biomass resources in SSF, with the capability to produce up to 19 different types of enzymes (Orzua et al., 2009). In this study, the growth potential of A. niger using COSOERs substrates was assessed. This evaluation was particularly focused on substrates with high lignin content and rigid cell structures, such as COFS, while also considering the presence of antimicrobial factors like saponin in COSC. The results revealed that fungal growth in COFS substrates was remarkably rapid and efficient, with a mean radial growth of 81.5 mm within 8 days (Fig. 2). The least SSF performance was shown when COSC was utilized as a substrate. In our previous study, PP was utilized in SSF for this application (Dessie et al., 2022). Mixing PP with the highly recalcitrant industrial hemp residue improved SSF performance compared to using a single substrate. It was hypothesized that the presence of sugars in the PP would stimulate fungal transcriptional regulators to produce enzyme cocktails for degrading lignocellulosic components in the industrial hemp residue. However, in the current study, although A. niger exhibited better growth performance on PP compared to COSC, it could not surpass the performance achieved with COFS. The SSF performance of A. niger on various biomass substrates individually, in mixture, and overall is presented as follows.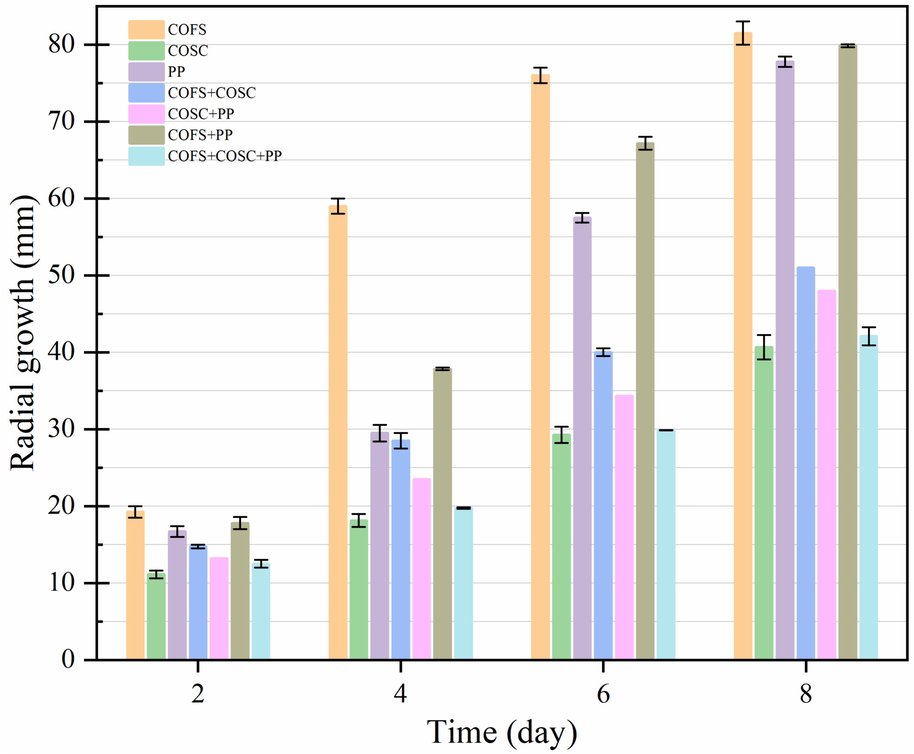
Radial growth of A. niger on agar plates using different biomass substrates. The values represent the mean of triplicates ± SD. COFS: C. oleifera Fruit Shell; COSC: C. oleifera Seed Cake; PP: Pomelo Peel.
COFS > PP > COSC.
COFS + PP > COFS + COSC > COSC + PP > COFS + COSC + PP.
COFS > COFS + PP > PP > COFS + COSC > COSC + PP > COFS + COSC + PP > COSC.
The high availability of lignocellulosic components in COFS may offer a suitable solid support for A. niger to grow and produce lignocellulose-degrading enzymes. It is worth mentioning that the substrates used in SSF did not undergo any pretreatment or nutrient supplementation. Additionally, the SSF performance of COFS (Fig. 2) was comparable to that of the commonly used fungal commercial medium, PDA (Fig. 3). This confirms that COFS can be utilized as a cheap and sustainable source for the production of fungal-based SSF enzymes to degrade lignocellulosic biomass during the enzymatic hydrolysis process.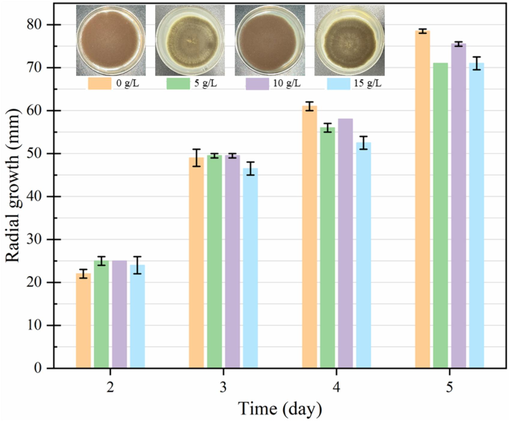
Radial growth of A. niger on potato dextrose agar plates containing 0─15 g/L saponin. The plate photographs were captured on the fifth day of growth. The values represent the mean of triplicates ± SD.
It is known that COSOERs contain a significant amount of saponin. A recent study revealed that COFS and COSC may contain up to 2.8─5.0 % and 10─15 % (dry weight) saponin, respectively (Liu et al., 2024). Saponins have been reported to possess antimicrobial effects, including antifungal activities (Kuo et al., 2010, Yu et al., 2022). In the current study, the SSF performance of A. niger was evaluated using PDA as a base medium supplemented with different concentrations (0─15 g/L) of saponin. The initial concentration of PDA was established at 46 g/L in accordance with the manufacturer's specifications. Nonetheless, the proportion of PDA in the medium diminishes in relation to the quantity of saponin to be added. As expected, the best overall SSF performance was achieved when A. niger was cultivated in saponin-free PDA, with a mean radial growth of 78.5 mm within 5 days. (Fig. 3). Nevertheless, during the initial stage, enhanced fungal growth was observed when different concentrations of saponin were added to PDA. Starting from the third day of cultivation, A. niger on PDA with 15 g/L saponin began to display lower growth compared to the other groups. The highest final growth was observed in PDA medium without saponin supplementation, followed by the addition of 10 g/L saponin. Based on these findings, it can be inferred that the addition of up to 15 g/L saponin (equivalent to 15 % saponin in a 10 % w/v mass ratio of COSOERs substrate) may not significantly inhibit the growth of A. niger. Indeed, A. niger is a well-established strain with the capability to grow on a wide range of substrates, demonstrating potential for degrading saponin (Qian et al., 2018).
3.2 Pretreatment
In most lignocellulose valorization strategies pretreatment is required before proceeding the enzymatic hydrolysis step. The major aim of pretreatment is to overcome the recalcitrance of the biomass substrate by breaking down the cell wall of lignocellulose, thereby increasing the accessibility of cellulose for enzymatic hydrolysis (Dessie et al., 2022, Singh Satish et al., 2023). To this end, the current study evaluated the effectiveness of various COSOERs pretreatment strategies, including AH, TH, and TCH.
3.2.1 Fourier-transform infrared spectroscopy analysis of pretreated solids
FTIR analysis was conducted using solid powders of the raw material and pretreatment residues. The results of the analysis can be seen in Fig. 4. The FTIR results were discussed based on previous studies as shown in Table S-1. A broad absorption band in the range of 3400─3300 cm─1, which is linked with the O─H stretching of cellulose and hemicellulose (Dessie et al., 2022), indicates the presence of a large number of hydroxyl groups in the lignocellulosic components, particularly for raw COSOERs. The peaks at 2930 and 2850 cm─1 appeared due to the C─H stretching in cellulose (Gulati et al., 2014, Yan et al., 2022). These peaks are sharp and enhanced for COSC-TCH, with the second peak being absent in COFS except for COFS-TCH. This implies, on one hand, the effectiveness of TCH in making cellulose readily available for subsequent enzymatic hydrolysis and, on the other hand, the higher degradability potential of COSC, which may be attributed to previous operations during the oil extraction process. The band 1740─1730 cm─1 is associated with C = O stretching of acetyl groups in hemicellulose and aldehydes in lignin (Dessie et al., 2022). The increase in intensity of this peak for TCH samples indicates the possible degradation of hemicellulose and removal of lignin by the combined action of temperature and oxalic acid. This was also partially reflected by the formation of dark-colored solid residues, specifically in COFS-TCH (Fig. 4 a). Peaks at 1650 (Dessie et al., 2021) and 1573 cm─1 (Mohamed et al., 2017), exhibit availability of primary amides and stretching of N─O, respectively. Interestingly, these peaks were not present in COFS samples, but 1650 and 1573 cm─1 were highly enhanced in raw and TCH of COSC, respectively. This may indicate the presence of proteins in COSC-Raw and the possible degradation of proteins after TCH. In fact, COSC is known to contain 12─15 % (w/w) crude protein (Liu et al., 2024).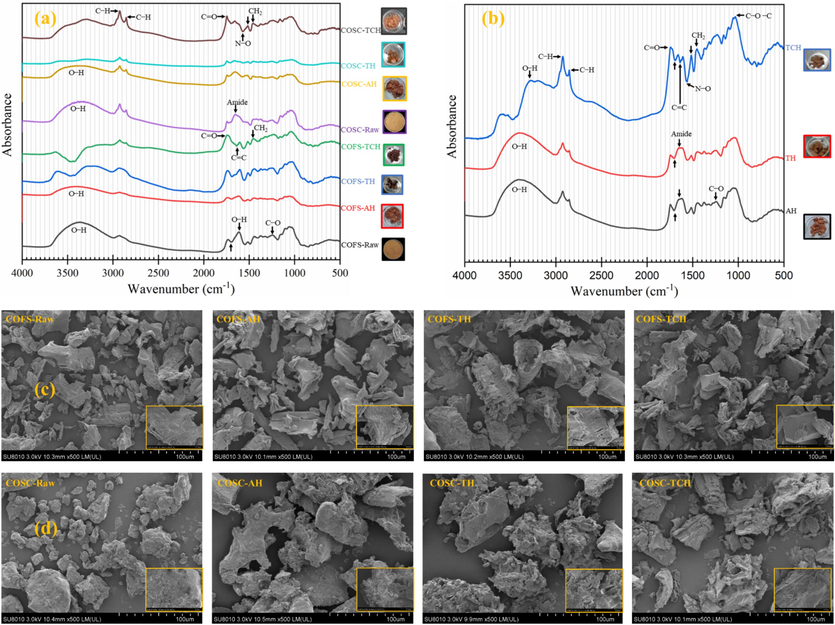
Analysis of raw solids and residual solids after pretreatment using Fourier-transform infrared spectroscopy; (a) Individual substrates, (b) mixture of COFS and COSC, and scanning electron microscope; (c) COFS, (d) COSC. COFS: C. oleifera Fruit Shell; COSC: C. oleifera Seed Cake; AH: Authohydrolysis; TH: Thermal Hydrolysis; TCH: Thermochemical Hydrolysis.
Distinct spectral peaks were observed when a mixture of COSOERs was used in the pretreatment process (Fig. 4 b). These peaks exhibited important attributes of both COFS and COSC (Table S-1). For example, the FTIR peak representing the aromatic skeletal vibration in lignin at 1512 cm─1 (Dessie et al., 2021) was highly enhanced for TCH of COSC and COFS + COSC, which could indicate lignin removal. An exceptionally high peak related to the C─O─C vibration at 1040 cm─1 (Li et al., 2016) was observed for COFS + COSC-TCH. It is known that in the cellulose structure, glucose units are bonded covalently through 1,4′-glycosidic, i.e., C─O─C, linkage (Altaner et al., 2014). Therefore, the aforementioned peak may indicate the availability high amount of exposed cellulose for further enzymatic hydrolysis processes. Intriguingly, a peak at 3285 cm─1 appeared only for COFS + COSC-TCH. This peak characterizes the O─H stretching vibration of the cellulose ring, lignin, and water (Meganathan et al., 2022). Overall, TCH exhibited more absorption peaks, and the highest number of peaks was observed particularly for COFS + COSC-TCH (Fig. 4 b). This indicates significant chemical changes resulting from TCH pretreatment using a combination of COFS and COSC biomass substrates, thus validating its effectiveness.
3.2.2 Scanning electron microscopy analysis of pretreated solids
In general the SEM analysis results are in agreement with FTIR studies with COFS and COSC showed different morphologies related to their origin. The SEM analysis results of COFS and COSC are shown in Fig. 4 c and d, respectively. Accordingly, COFS-Raw displayed a tight and densely packed woody structure, while COFS-AH revealed relatively a thinner surface due to the removal of certain particles by water from the woody material. The SEM analysis of COFS-TH solid residues revealed a rough surface and large holes, attributed to the steam explosion process that caused cracking and degradation of the lignocellulose biomass. Furthermore, the cracks that were created by TH process allowed for better penetration of oxalic acid into the substrate, which could facilitate the degradation of hemicellulose and removal of lignin during the TCH. This led to the formation of a relatively smooth and thin surface layer in the COFS-TCH solid residues, which ultimately increased the surface area and exposed the cellulose for subsequent enzymatic hydrolysis.
On the other hand, COSC-Raw exhibited a granular structure that may be attributed to the compression of the seeds during the previous oil extraction process. The solid residues of COSC-AH showed a more altered structure compared to COSC-Raw, which was more pronounced than the changes observed in COFS upon AH pretreatment. This suggests that COSC is more susceptible to degradation than COFS, owing to the physicochemical alterations that occur during the oil extraction process. Likewise, SEM analysis of COSC-TH solid samples displayed a highly rough and cracked surface, while COSC-TCH solid residues exhibited large holes and exposed fibers. These observations indicate favorable conditions for subsequent enzymatic saccharification processes.
3.2.3 Reducing sugar in pretreated hydrolysates
Pretreatment was optimized using single (COFS or COSC) or mixed (1:1 of COFS and COSC) COSOERs biomass and employing different pretreatment strategies (AH, TH, and TCH). The results revealed that the reducing sugar production performances based on the substrate type and pretreatment method were as follows; COFS + COSC > COSC > COFS and TCH > TH > AH, respectively (Fig. 5). Even though there was not a significant difference in sugar production from COFS and COSC, pretreatment of their mixture provided certain advantages. TCH of COSOERs by far produced the highest amount of reducing sugars. In general, TCH of mixture of COFS and COSC produce the highest sugar of 28 g/L. The TCH of COFS showed the highest enhancement in sugar production, which is 9.22 and 5.24 folds than its AH and TH counterparts, respectively. The high availability of lignocellulosic components, particularly lignin, may make COFS relatively resistant to AH and TH pretreatments, but it shows improved performance during TCH using oxalic acid.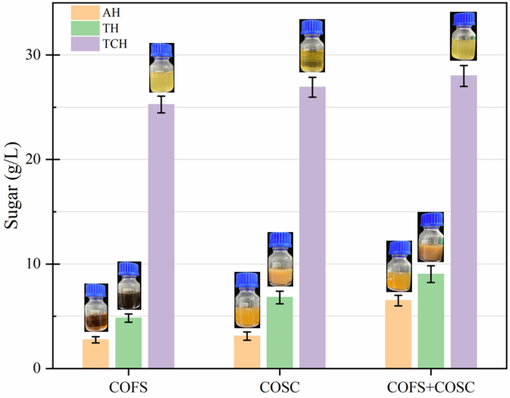
Sugar production after pretreatment. The bottle photographs are hydrolysates of the corresponding pretreatment. The values represent the mean of triplicates ± SD. COFS: C. oleifera Fruit Shell; COSC: C. oleifera Seed Cake; AH: Authohydrolysis; TH: Thermal Hydrolysis; TCH: Thermochemical Hydrolysis.
The color of hydrolysates generated by pretreatment of COSOERs showed variations based on the substrate type and pretreatment method (Fig. 5). Accordingly, TH pretreatment and COFS showed the darkest coloration whereas TCH and COSC revealed lightest (yellowish) coloration in their hydrolysates. According to a previous report, the dark coloration of hydrolysate may appear due to the Maillard reaction (interaction between reducing sugars and amino acids) during TH pretreatment of lignocellulosic materials (Korniłłowicz-Kowalska and Rybczyńska-Tkaczyk, 2020). The colorants present in hydrolysates generated by TH are not easily degradable, which can have a negative impact on enzymatic hydrolysis and fermentation processes. The dark coloration of the hydrolysate needs to be removed through additional processes, which increases expenses. In this study, TCH of COSOERs using oxalic acid served a dual purpose by degrading lignocellulosic polymers and removing colorants from the hydrolysates.
The detailed pH values of the pretreatment are presented in Table 1. Among the pretreatment slurries and hydrolysates, the highest pH was observed in the AH samples, followed by TH and TCH, respectively. Notably, there were no significant pH differences between COFS and COSC. The considerably low pH observed in TCH can be attributed to the presence of oxalic acid. While the pH variation between slurries and hydrolysates was not substantial, a slight reduction was noted in the pH of the slurries. The increase in pH of the hydrolysates could be attributed to the removal of low pH-causing components such as oxalic acid, along with the solid residue, during the solid–liquid separation stage. The values represent the mean of triplicates ± SD. COFS: C. oleifera Fruit Shell; COSC: C. oleifera Seed Cake; COFS + COSC: equal mixture of COFS and COSC; AH: Autohydrolysis; TH: Thermal Hydrolysis; TCH: Thermochemical Hydrolysis.
Substrate
AH
TH
TCH
Slurry
Hydrolysate
Slurry
Hydrolysate
Slurry
Hydrolysate
COFS
5.4 ± 0.25
5.4 ± 0.28
4.2 ± 0.24
4.4 ± 0.21
0.94 ± 0.03
1.25 ± 0.04
COSC
5.5 ± 0.20
5.6 ± 0.23
4.5 ± 0.21
4.7 ± 0.26
0.96 ± 0.03
1.35 ± 0.05
COFS + COSC
5.5 ± 0.22
5.5 ± 0.25
4.4 ± 0.20
4.6 ± 0.25
0.96 ± 0.04
1.34 ± 0.05
3.3 Enzymatic hydrolysis
Following comprehensive optimization of the pretreatment and SSF stages, enzymatic hydrolysis studies were conducted to further produce fermentative sugar from COSOERs. In this study, the solid mashes generated by SSF of A. niger using COFS and the COSOER slurries produced via TH and TCH were chosen for the enzymatic hydrolysis. The enzymatic hydrolysis process was serially optimized to enhance the production of sugars. In the first case, enzymatic hydrolysis was undergone by adding SSF mashes to the pretreated slurries of COSOER without adjusting the pH of the slurry. It is clear that oxalic acid pretreatment of COSOER provided a slurry with low pH values (Table 1). As a result, it is necessary to adjust the pH of this slurry within the optimum range for the enzymes activity. However, pH adjustment of slurry using NaOH solution led to dilution of the pretreated hydrolysate in the second case of enzymatic hydrolysis. In the third strategy, the hydrolysates and solid residues of the slurry was separated via centrifugation and the solid residues were further used for enzymatic hydrolysis. These solid residues were washed using alkaline solution to adjust the pH and the washing liquor was recycled for enzymatic hydrolysis (Fig. 1). In all cases, the pH of the pretreated slurries generated by TH of COSOERs were nearly within the range required by the enzymes (Table 1), and therefore, did not require any adjustment.
3.3.1 Fourier-transform infrared spectroscopy analysis of enzymatically hydrolyzed solids
In general, the FTIR peak changes in the enzymatic hydrolysis samples can be seen as related to the degradation of hemicellulose and the removal of lignin by the previous pretreatment step, and the subsequent cellulose saccharification by the SSF enzymes. The details for the FTIR analysis of enzymatic hydrolysis samples can be found Fig. 6 and Table S-1. To begin with, unlike the pretreatment samples, the shape of extended peak from 3700 to 3000 cm─1 was nearly uniform for all samples generated after enzymatic hydrolysis. The peak at 1740─1730 cm─1 almost disappeared for TCH samples. Similarly, the 1600 and 1248 cm─1 peaks for O─H bending associated with water in hemicelluloses (Dessie et al., 2021) and C─O stretching (Li et al., 2016), respectively, were absent in these enzymatic hydrolysis samples. On the contrary, some FTIR peaks, such as those at 1620─1610, 1085, 824, and 780 cm─1, were present in the enzymatic hydrolysis samples but not in the pretreatment ones. The first peak represents C = C stretching (Mohamed et al., 2017) related to α,β-unsaturated ketone and is exhibited in TCH of COFS and COFS + COSC. The peak at 1085 cm─1 could be due to C─O stretching (Mohamed et al., 2017), while the peaks at 824 and 780 cm─1 can be associated with C─H bending (Dessie et al., 2021). These peaks appeared in COFS-TH, which may indicate the presence of residual undegraded cellulose in this sample, while cellulose degradation occurred in other enzymatic hydrolysis samples.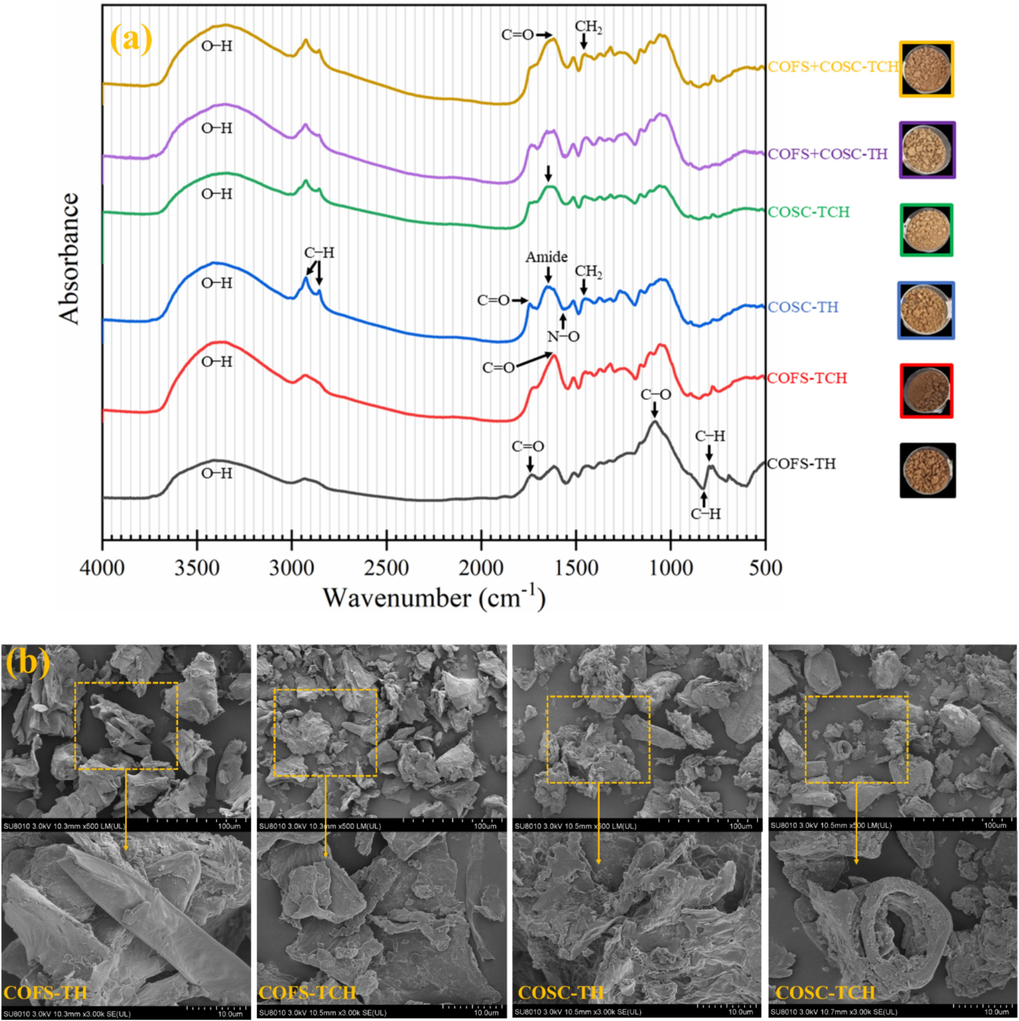
Analysis of residual solids generated after enzymatic hydrolysis using (a) Fourier-transform infrared spectroscopy analysis and (b) scanning electron microscope. COFS: C. oleifera Fruit Shell; COSC: C. oleifera Seed Cake; TH: Thermal Hydrolysis; TCH: Thermochemical Hydrolysis.
3.3.2 Scanning electron microscopy analysis of enzymatically hydrolyzed solids
The SEM analysis results are consistent with the FTIR analyses. The solid residues generated after enzymatic hydrolysis were observed to display a higher degree of degradation compared to their respective raw material and pretreated counterparts. The SEM analysis of enzymatically hydrolyzed solids are shown in Fig. 6 b. The solid residues of COFS-TH revealed the presence of residual fibrous materials, whereas COFS-TCH solids exhibited a thinner surface and a highly negligible fiber content. Similarly, SEM analysis of COSC-TH solids showed a rough surface with remaining fibers, while COSC-TCH solids displayed a large cavity. The results of TH solids suggest incomplete degradation of lignocellulose biomass. In contrast, SEM analysis of solids generated by TCH and subsequent enzymatic hydrolysis demonstrated efficient breakdown of fiber cell walls and other macromolecules.
3.3.3 Reducing sugars in enzymatically hydrolyzed liquids
The enzymatic hydrolysis was undergone for 24 h based on the sugar production pattern. First, enzymatic hydrolysis was conducted using slurries generated by TH. Sugar production increases correspondingly with the increase in hydrolysis time. The highest sugar (16.16 g/L) for TH was achieved using a mixture of COFS and COSC followed by COSC and COFS, respectively (Fig. 7 a). The sugar production in the enzymatic hydrolysis showed improvement compared to the corresponding sugar generated by TH pretreatment. However, in the first strategy (Fig. 1, #1) which employs slurries of TCH, the amount of reducing sugar decreased as the enzymatic hydrolysis progressed, with the poorest performance using COFS (Fig. 7 b). For example, the sugar concentration, which was 25.26 g/L at the beginning of saccharification in the TCH slurry of COFS, decreased to 14.40 g/L at the end of enzymatic hydrolysis. The primary factor for this could be the very low pH of the slurry (Table 1) generated by acidic pretreatment of the substrate. Previous studies have reported that the maximum activity of lignocellulose degrading enzymes produced by A. niger and related fungi occurs at a pH of approximately 5.0, while the optimal operating pH range falls between 4.0 and 6.0 (Singhania et al., 2010, Infanzón-Rodríguez et al., 2022). The enzymes could be deactivated when the pH is below or above this range. Other possible reason for the reduction of the concentration of the sugar might be further degradation of sugars by the acid. The degradation of sugars not only reduces the amount of sugars available in the hydrolysate but can also generate lignocellulose pretreatment degradation products that may inhibit enzymatic hydrolysis. Furthermore, sugars available in the slurries that were produced during TCH of COSOERs could impose negative effect on enzymatic hydrolysis. The inhibitory effects of sugars on lignocellulose enzymatic hydrolysis can be found in previous studies (Miao et al., 2012, Sun et al., 2023).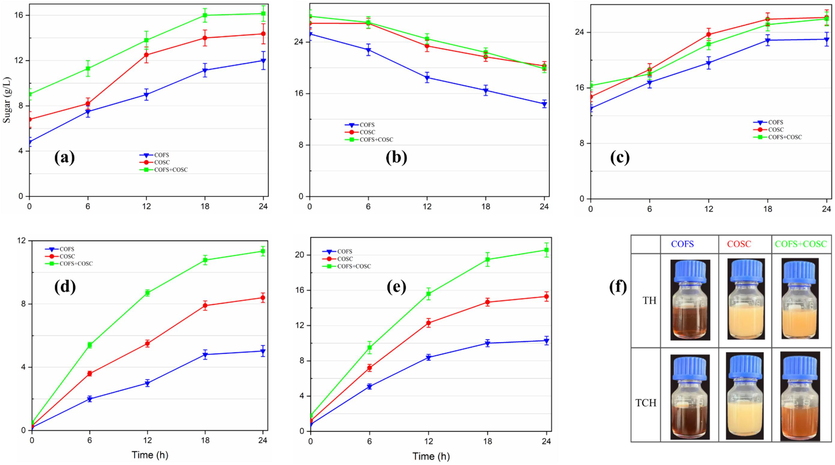
Sugar production after enzymatic hydrolysis using pretreatment slurries of (a) TH, (b) TCH without pH adjustment and (c) TCH with pH adjustment, and solid residues of (d) TH and (e) TCH pretreatment after pH adjustment. (f) Hydrolysates of (d) and (e). The values represent the mean of triplicates ± SD. COFS: C. oleifera Fruit Shell; COSC: C. oleifera Seed Cake; TH: Thermal Hydrolysis; TCH: Thermochemical.
To overcome the aforementioned challenge, the second strategy (Fig. 1 #2) was developed to adjust the pH of COSOER slurries produced by TCH to be closer to the enzyme's optimal activity. The reducing sugar production showed successive improvements as the enzymatic hydrolysis proceeded (Fig. 7 c). However, adjusting the pH with alkaline solutions caused the initial slurry to become diluted, resulting in a lower amount of sugar accumulation at the end of enzymatic hydrolysis than what was achieved even at the end of pretreatment (prior to pH adjustment). For instance, the sugar which was 25.26 g/L in the TCH slurry of COFS reduced to 13.06 g/L after pH adjustment (at the beginning of enzymatic hydrolysis), and finally reached 23.01 g/L after enzymatic hydrolysis. Besides, as explained above, the initially available sugars in the slurry may have inhibitory effect on the enzymatic hydrolysis.
In the third strategy, the sugar-containing hydrolysates generated after pretreatment were preserved (Fig. 1 #3), whereas the solid residues were considered for subsequent enzymatic hydrolysis. Solid residues collected after TCH were washed using alkaline solution to adjust the pH suitable to enzymatic hydrolysis and the washing liquor was recycled for saccharification (Fig. 1, #3). Instead of discarding the washing liquor, it was recycled and used in enzymatic hydrolysis alongside water (Fig. 1). This approach facilitates a decrease in water consumption while addressing environmental concerns associated with improper alkaline waste disposal. The low initial sugar concentration of the slurry used in this enzymatic hydrolysis process serves a dual purpose: it not only reflects the significant amount of sugar recovered during pretreatment but also helps to avoid potential inhibitory effects on enzyme activity that may arise from high sugar concentrations. In the enzymatic hydrolysis step, further degradation of lignocellulosic components occurs and sugar production increases as the saccharification process advances. For both TH and TCH residual solid slurries, the highest sugar were achieved through enzymatic hydrolysis of mixtures of COSOERs, followed by COSC and COFS, respectively. Evidently, TH and TCH pretreatments of COSOER mixtures produced 9.03 and 28 g/L sugar (Fig. 5), and the corresponding solid residues employed for enzymatic hydrolysis to achieve additional 11.34 and 20.58 g/L sugar (Fig. 7 d & e), respectively. During enzymatic hydrolysis, the previously light-colored hydrolysate of COFS (which had undergone TCH) (Fig. 5) tends to darken due to the addition of NaOH, which was utilized to adjust the pH (Fig. 7 f).
The utilization of oxalic acid-based TCH and the incorporation of mixed substrates of COFS and COSC in this study confer significant advantages over other pretreatment methods and individual substrates. These benefits encompass the efficient degradation of lignocellulosic biomass and the elimination of dark colorants from the hydrolysate, thereby positively influencing pretreatment and enzymatic hydrolysis processes. Oxalic acid pretreatment primarily targets the hemicellulose component of the biomass, breaking it down into pentose sugars such as xylose. Additionally, oxalic acid demonstrates potential for lignin removal, as evidenced by SEM and FTIR studies. Consequently, the degradation of hemicellulose and removal of lignin could expose cellulose, creating an optimal environment for SSF enzymes to efficiently hydrolyze it. Moreover, the combination of the robust COFS and the highly degradable COSC offers several advantages. These include the dilution of colorants and recalcitrant components like lignin, as well as enhancement of degradation efficiency.
Alternatively, the inhibitory effect of sugars and pretreatment degradation products of the hydrolysate during enzymatic hydrolysis could be managed by removal of sugars (Miao et al., 2012) and detoxification (Jönsson and Martín, 2016), respectively. However, these strategies require additional processing steps that could increase the production cost and energy consumption (Yuan et al., 2021). The decrease in sugar concentration resulting from dilution in the second strategy could have been alleviated by concentrating the sugar. However, it's crucial to note that concentrating the sugar from the hydrolyte might introduce additional complexities and expenses to the process, such as increased time, energy consumption, and the requirement for additional equipment. Moreover, the pH of acidic pretreated solid residues to be used in enzymatic hydrolysis could be adjusted through water washing. This technique, unfortunately, leads to high water consumption and resource loss. Since water is usually a limiting factor in most bioprocessing, developing strategies to reduce water consumption is crucial to ensure acceptable sustainability metrics for building a bio-based production scheme (Reis et al., 2023).
To further assess the efficiency of the current study, results were compared with recent literature (Table 2). The sugar production performance of this study was found to be encouraging, and even superior to that of most of the research considered in this comparison. The current study developed a simple, effective and sustainable method that can help reduce water consumption and efficiently utilize biomass resources. Embracing green pretreatment methods and employing on-site enzymes generated through SSF, rather than traditional pretreatment and commercial enzymes, further strengthens the applicability of this study in bioprocessing. To end with, the sugar-rich hydrolysates obtained from pretreatment (Fig. 3) and enzymatic hydrolysis (Fig. 7 e) have the potential to be utilized for the production of chemicals and fuels through fermentation (Fig. 1 #3). COFS: C. oleifera Fruit Shell; COSC: C. oleifera Seed Cake; COFS + COSC: COFS and COSC mix; TOFH: Tea Oil Fruit Hull; OPTB: Oil Palm Trunk Biomass; FVW: Fruit and Vegetable Waste; WS: Wheat Straw; CP: Coffee Husk; OA: Oxalic Acid; SSF: Solid-state Fermentation; rpm: revolution per minute; NA: Not Applicable.
Substrate
Pretreatment
Enzymatic hydrolysis
References
Condition
Sugar (g/L)
Condition
Sugar (g/L)
COFS
10 % substrate, 2 % OA, 121 °C, 30 min
25.26
A. niger SSF enzymes, 50 °C, 200 rpm, 24 h
10.30
This study
COSC
10 % substrate, 2 % OA, 121 °C, 30 min
26.92
A. niger SSF enzymes, 50 °C, 200 rpm, 24 h
15.30
This study
COFS + COSC
10 % substrate, 2 % OA, 121 °C, 30 min
28.00
A. niger SSF enzymes, 50 °C, 200 rpm, 24 h
20.58
This study
TOFH
10 % substrate, 52 % acetic acid, 0.64 % H2SO4, 125 °C, 1.7 h
25.50
3 % H2O2, Cellulase, 50 °C, 150 rpm, 48 h
15.00
(Tang et al., 2017)
OPTB
10 % substrate, 1 % OA, 120 °C, 30 min
16.23
Cellulase, 50 °C, 150 rpm, 72 h
22.13
(Bukhari et al., 2021)
Corncob
5 % substrate, 150 mmol/L OA, 140 °C, 150 min
26.67
NA
NA
(Cheng et al., 2018)
OPTB
10 % substrate, 5 % OA, 100 °C, 120 min
0.00
Cellulase, 50 °C, 155 rpm, 48 h
33.36
(Bukhari et al., 2020)
FVW
NA
NA
10 % substrates, A. niger and Rhizopus oryzae SSF enzymes, 50 °C, 500 rpm, 24 h
25.83
(Dessie et al., 2018)
WS
10 % substrate, 190 °C, 20 min
0.00
A. niger SmF enzymes, 50 °C, 150 rpm, 48 h
32.90
(Infanzón-Rodríguez et al., 2022)
CH
10 % substrate, 0.18 mol/L H2SO4, 121 °C, 30 min
24.40
NA
NA
(Dessie et al., 2018)
3.4 Material balance
Strategy 3 (Fig. 1) emerged as the most efficient technique in our study. Consequently, material balance was conducted for this scenario. The solid and liquid generated after pretreatment and enzymatic hydrolysis were analyzed to determine the residual solid and sugar contents, respectively. The residual solid content (%) was calculated using eq. (1), while the sugar yield was calculated based on the milligrams of sugar generated per grams of initial substrate used, as demonstrated in eq. (2). Generally, there was an inverse relationship between residual solid and sugar yield (Fig. 8). Specifically, pretreatment yielded less residual solid (Fig. 8 a) compared to the corresponding enzymatic hydrolysis (Fig. 8 b), thereby leading to higher sugar yields, particularly for TCH (Fig. 8 c). For instance, COFS + COSC-TCH and enzymatic hydrolysis of COS-TP produced the lowest and highest solid residues at 51.2 % and 89.6 %, respectively. The lowest sugar yields of 48.4 mg/g substrate and 50.3 mg/g substrate were achieved from pretreatment and enzymatic hydrolysis, respectively, of COFS-TH. Conversely, the highest sugar yields of 280 mg/g substrate and 205.8 mg/g substrate were generated from pretreatment and enzymatic hydrolysis, respectively, of COFS + COSC-TCH. Although there was no significant difference in sugar yield between TCH pretreatments, enzymatic hydrolysis of COFS + COSC-TCH resulted in a two-fold increase in sugar yield compared to the COFS-TCH counterpart (Fig. 8 d). These results highlighted the effectiveness of the combined effect of oxalic acid-based TCH and the utilization of mixed substrates.
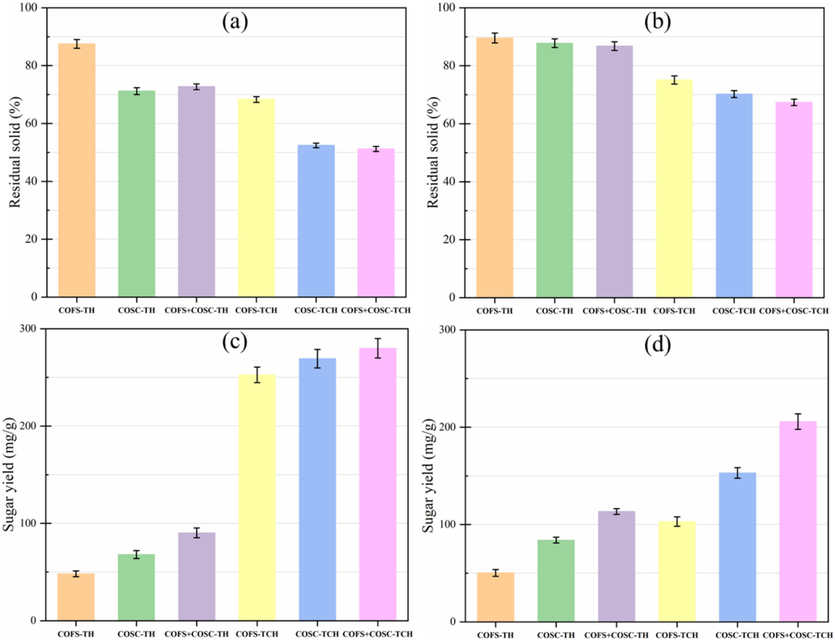
Residual solid content of pretreatment (a) and enzymatic hydrolysis (b), and sugar yield of pretreatment (c) and enzymatic hydrolysis (d). The values represent the mean of triplicates ± SD. COFS: C. oleifera Fruit Shell; COSC: C. oleifera Seed Cake; COFS + COSC: equal mixture of COFS and COSC; TH: Thermal Hydrolysis; TCH: Thermochemical hydrolysis.
4 Conclusions
This study aimed to create simple and efficient methods for pretreating and enzymatically hydrolyzing COSOERs, with the goal of converting them into fermentable feedstocks. Different methods were employed to pretreat COSOERs both individually and in mixed form. As a result, the mixture of COFS and COSC produced the highest sugar of 28 g/L when subjected to TCH pretreatment. Enzymes used for saccharification were on-site produced via SSF using A. niger and cheap biomass resources. The findings indicated that COFS was the ideal substrate for producing SSF enzymes to degrade lignocellulosic biomass. Following this, SSF enzymes were utilized to convert the lignocellulosic components in pretreated COSOERs into fermentable sugar. Through optimizing the enzymatic hydrolysis process, the best result of 20.58 g/L of sugar was achieved using solid residues from a COFS and COSC mixture that had undergone TCH pretreatment. The use of SSF enzymes, adoption of green and mild pretreatment methods, minimized water usage, and implementation of an effective biomass utilization approach collectively enhance the overall effectiveness of this study. However, additional optimization of various parameters, scaling-up studies, and conducting techno-economic and environmental assessments are necessary steps before advancing to the large-scale production stage.
CRediT authorship contribution statement
Wubliker Dessie: Conceptualization, Data curation, Funding acquisition, Investigation, Writing – original draft, Writing – review & editing, Methodology. Jingjing Xiao: Methodology, Writing – review & editing. Jiachen Tang: Methodology, Writing – review & editing. Baiyan An: Methodology, Writing – review & editing. Xiaofang Luo: Writing – review & editing. Meifeng Wang: Writing – review & editing. Yunhui Liao: Writing – review & editing. Rizwan Wahab: Writing – review & editing. Changzhu Li: Funding acquisition, Project administration, Writing – review & editing. Zuodong Qin: Funding acquisition, Project administration, Writing – review & editing.
Acknowledgements
This work was supported by the Ministry of Science and Technology Young Foreign Talent Program, China (QN2022029001), Foundation of State Key Laboratory of Utilization of Woody Oil Resource, China (GZKF202128, GZKF202220), the Young Science and Technology Talents Project of Hunan Province, China (2022RC1168) and Shennongguo Oil Team, Hunan Talent Project, China (2022) No 9. The authors would also like to thank the Researchers Supporting Project Number (RSPD2024R1042), King Saud University, Riyadh, Saudi Arabia.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- How Cellulose Stretches: Synergism between Covalent and Hydrogen Bonding. Biomacromolecules.. 2014;15:791-798.
- [CrossRef] [Google Scholar]
- Bioprospecting thermophilic/thermotolerant microbes for production of lignocellulosic ethanol: A future perspective. Renew. Sustain. Energy Rev.. 2015;51:699-717.
- [CrossRef] [Google Scholar]
- An extracellular glucose sensor for substrate-dependent secretion and display of cellulose-degrading enzymes. Biotechnol. Bioeng. N/a 2023
- [CrossRef] [Google Scholar]
- Organic Acid Pretreatment of Oil Palm Trunk Biomass for Succinic Acid Production. Waste Biomass Valoriz.. 2020;11:5549-5559.
- [CrossRef] [Google Scholar]
- Whole slurry saccharification of mild oxalic acid-pretreated oil palm trunk biomass improves succinic acid production. Ind. Crop. Prod.. 2021;171:113854
- [CrossRef] [Google Scholar]
- Techno-economic evaluation of second-generation ethanol from sugarcane bagasse: Commercial versus on-site produced enzymes and use of the xylose liquor. J. Clean. Prod.. 2022;369:133340
- [CrossRef] [Google Scholar]
- Pyrolysis characteristics of tea oil camellia (Camellia oleifera Abel.) shells and their chemically pre-treated residues: Kinetics, mechanisms, product evaluation and joint optimization. J. Anal. Appl. Pyrolysis.. 2022;164:105526
- [CrossRef] [Google Scholar]
- Camellia oleifera Shell Biochar as a Robust Adsorbent for Aqueous Mercury Removal. Fermentation.. 2023;9:295.
- [Google Scholar]
- A new approach to recycle oxalic acid during lignocellulose pretreatment for xylose production. Biotechnol. Biofuels.. 2018;11:324.
- [CrossRef] [Google Scholar]
- Catalytic co-pyrolysis behaviors and kinetics of camellia shell and take-out solid waste using pyrolyzer – gas chromatography/mass spectrometry and thermogravimetric analyzer. Bioresour. Technol.. 2020;297:122419
- [CrossRef] [Google Scholar]
- Succinic acid production from fruit and vegetable wastes hydrolyzed by on-site enzyme mixtures through solid state fermentation. Bioresour. Technol.. 2018;247:1177-1180.
- [CrossRef] [Google Scholar]
- Bio-succinic acid production from coffee husk treated with thermochemical and fungal hydrolysis. Bioprocess Biosyst. Eng.. 2018;41:1461-1470.
- [CrossRef] [Google Scholar]
- Towards Full Utilization of Biomass Resources: A Case Study on Industrial Hemp Residue and Spent Mushroom Substrate. Processes.. 2021;9:1200.
- [Google Scholar]
- One-pot conversion of industrial hemp residue into fermentable feedstocks using green catalyst and enzyme cocktails generated by solid-state fermentation. Ind. Crop. Prod.. 2022;182:114885
- [CrossRef] [Google Scholar]
- Lignin valorization: A crucial step towards full utilization of biomass, zero waste and circular bioeconomy. Biocatal. Agric. Biotechnol.. 2023;51:102777
- [CrossRef] [Google Scholar]
- Production of carboxymethylcellulose fibers from waste lignocellulosic sawdust using NaOH/NaClO2 pretreatment. Fibers Polym.. 2014;15:680-686.
- [CrossRef] [Google Scholar]
- Eco-Friendly Leaching and Separation of Vanadium over Iron Impurity from Vanadium-Bearing Shale Using Oxalic Acid as a Leachant. ACS Sustain. Chem. Eng.. 2018;6:1900-1908.
- [CrossRef] [Google Scholar]
- Enzymatic hydrolysis of lignocellulosic biomass using native cellulase produced by Aspergillus niger ITV02 under liquid state fermentation. Biotechnol. Appl. Biochem.. 2022;69:198-208.
- [CrossRef] [Google Scholar]
- Pretreatment of lignocellulose: Formation of inhibitory by-products and strategies for minimizing their effects. Bioresour. Technol.. 2016;199:103-112.
- [CrossRef] [Google Scholar]
- Decolorization and biodegradation of melanoidin contained in beet molasses by an anamorphic strain of Bjerkandera adusta CCBAS930 and its mutants. World J. Microbiol. Biotechnol.. 2020;37:1.
- [CrossRef] [Google Scholar]
- Bioactive Saponin from Tea Seed Pomace with Inhibitory Effects against Rhizoctonia solani. J. Agric. Food Chem.. 2010;58:8618-8622.
- [CrossRef] [Google Scholar]
- Fabrication of carbon microspheres with controllable porous structure by using waste Camellia oleifera shells. Mat. Chem. Phy.. 2016;181:518-528.
- [CrossRef] [Google Scholar]
- A sustainable wood biorefinery for low–carbon footprint chemicals production. Science.. 2020;367:1385-1390.
- [CrossRef] [Google Scholar]
- Utilization of camellia oleifera shell for production of valuable products by pyrolysis. Arab. J. Chem.. 2022;15:104348
- [CrossRef] [Google Scholar]
- Diagnostic product ions-based chemical characterization and antioxidative activity evaluation of solid fermentation for Astragali radix produced by Paecilomyces cicadae. Arab. J. Chem.. 2021;14:102908
- [CrossRef] [Google Scholar]
- Valorization of Camellia oleifera oil processing byproducts to value-added chemicals and biobased materials: A critical review. Green Energy Environ.. 2024;9:28-53.
- [CrossRef] [Google Scholar]
- Photocatalytic self-cleaning and antibacterial activity of cotton fabric coated with polyaniline/carbon nitride composite for smart textile application. Phosphorus Sulfur Silicon Relat. Elem.. 2022;197:244-253.
- [CrossRef] [Google Scholar]
- Kinetic studies on the product inhibition of enzymatic lignocellulose hydrolysis. Appl. Biochem. Biotechnol.. 2012;167:358-366.
- [CrossRef] [Google Scholar]
- Use of Dinitrosalicylic Acid Reagent for Determination of Reducing Sugar. Anal. Chem.. 1959;31:426-428.
- [CrossRef] [Google Scholar]
- Chapter 1 - Fourier Transform Infrared (FTIR) Spectroscopy. In: Hilal N., Ismail A.F., Matsuura T., eds. Membrane Characterization. Elsevier; 2017. p. :3-29.
- [Google Scholar]
- Organosolv pretreatment: an in-depth purview of mechanics of the system. Bioresour. Bioprocessing.. 2023;10:50.
- [CrossRef] [Google Scholar]
- Exploitation of agro industrial wastes as immobilization carrier for solid-state fermentation. Ind. Crop. Prod.. 2009;30:24-27.
- [CrossRef] [Google Scholar]
- Efficient Isolation of Cellulosic Fibers from Coffee Parchment via Natural Acidic Deep Eutectic Solvent Pretreatment for Nanocellulose Production. ACS Sustain. Chem. Eng.. 2023;11:13962-13973.
- [CrossRef] [Google Scholar]
- Effects of fermentation on the hemolytic activity and degradation of Camellia oleifera saponins by Lactobacillus crustorum and Bacillus subtilis. FEMS Microbiol. Lett.. 2018;365
- [CrossRef] [Google Scholar]
- Applications of Chinese Camellia oleifera and its By-Products: A Review. Front. Chem.. 2022;10
- [CrossRef] [Google Scholar]
- Biotechnological enhancement of lactic acid conversion from pretreated palm kernel cake hydrolysate by Actinobacillus succinogenes 130Z. Sci. Rep.. 2023;13:5787.
- [CrossRef] [Google Scholar]
- Process strategies to reduce cellulase enzyme loading for renewable sugar production in biorefineries. Chem. Eng. J.. 2023;451:138690
- [CrossRef] [Google Scholar]
- Alkaline Peroxide Pretreatment of Waste Lignocellulosic Sawdust for Total Reducing Sugars. Korean Chem. Eng. Res.. 2023;61:412-418.
- [CrossRef] [Google Scholar]
- Advancement and comparative profiles in the production technologies using solid-state and submerged fermentation for microbial cellulases. Enzym. Microb. Technol.. 2010;46:541-549.
- [CrossRef] [Google Scholar]
- Advances and perspectives on mass transfer and enzymatic hydrolysis in the enzyme-mediated lignocellulosic biorefinery: A review. Biotechnol. Adv.. 2023;62:108059
- [CrossRef] [Google Scholar]
- Biorefining fractionation of the Camellia oleifera Abel. hull into diverse bioproducts with a two-stage organosolv extraction. Ind. Crop. Prod.. 2016;94:790-799.
- [CrossRef] [Google Scholar]
- Bioprocessing of tea oil fruit hull with acetic acid organosolv pretreatment in combination with alkaline H2O2. Biotechnol. Biofuels.. 2017;10:86.
- [CrossRef] [Google Scholar]
- Characterizations and application potentials of the hemicelluloses in waste oil-tea camellia fruit shells from Southern China. Ind. Crop. Prod.. 2022;178:114551
- [CrossRef] [Google Scholar]
- Anatomy and lignin deposition of stone cell in Camellia oleifera shell during the young stage. Protoplasma.. 2021;258:361-370.
- [CrossRef] [Google Scholar]
- Effect of phenolic extracts from Camellia oleifera seed cake on the formation of polar compounds, core aldehydes, and monoepoxy oleic acids during deep-fat frying. Food Chem.. 2022;372:131143
- [CrossRef] [Google Scholar]
- Fabrication of supported acid catalytic composite fibers by a simple and low-cost method and their application on the synthesis of liquid biofuel 5-ethoxymethylfurfural. Green Energy Environ.. 2022;7:165-171.
- [CrossRef] [Google Scholar]
- Fruit economic characteristics and yields of 40 superior Camellia oleifera Abel plants in the low-hot valley area of Guizhou Province. China. Sci. Rep.. 2022;12:7068.
- [CrossRef] [Google Scholar]
- Study on the antifungal activity and mechanism of tea saponin from Camellia oleifera cake. Eur. Food Res. Technol.. 2022;248:783-795.
- [CrossRef] [Google Scholar]
- Potential for reduced water consumption in biorefining of lignocellulosic biomass to bioethanol and biogas. J. Biosci. Bioeng.. 2021;131:461-468.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2024.105815.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary Data 1
Supplementary Data 1







