Translate this page into:
Mechanistic action of lead removal by the native isolate Bacillus amyloliquefaciens ON261680.1
⁎Corresponding author. Bassma.hassan@pua.edu.eg (Bassma H. Elwakil)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Lead (Pb) is one of the substantial hazard pollutants that cause soil pollution. Bioremediation is an eco-friendly and cost-effective technique that provides a convenient solution for lead removal. In the present study, lead resistant bacterial strains were isolated from industrial effluents contaminated soil with lead minimum inhibitory concentrations reached 15000 ppm. The most resistant isolate was identified by 16S rRNA sequencing as Bacillus amyloliquefaciens ON261680.1. Taguchi statistical design was applied to optimize the environmental and nutritional factors that lead to maximum lead removal. The optimized conditions for the highest lead removal of lead concentration, incubation temperature, incubation time, peptone, yeast, beef extract, NaCl concentrations, pH, inoculum size and inoculum age were 10000 ppm, 30 °C, 96 hrs incubation, 0.5 g, 0.5 g, 1.5 g, 6 g, 9, 15 mL and 48 hrs old culture, respectively. The most significant factors (p < 0.05) were incubation time, pH, lead concentration, and incubation temperature. Mechanistic action was investigated through employing Fourier transform infrared spectroscopy (FTIR), Energy-dispersive X-ray spectroscopy (EDX) and transmission electron microscopy (TEM). It was revealed that Bacillus amyloliquefaciens ON261680.1 removed the lead pollutant through bio-adsorption, deposition and complexation on the cell surface. The present study may pave the way to use Bacillus amyloliquefaciens ON261680.1 as an efficient bioremediation tool.
Keywords
Lead bioremediation
Taguchi statistical design
Lead resistant bacteria
Mechanistic action
1 Introduction
Heavy metal pollution was considered as hazardous dilemma that influencing aquatic and terrestrial life (Kalaimurugan et al., 2020). Lead (Pb) is one of the most widely distributed contaminants present in soil, water, and air (Kushwaha et al., 2018) due to its economic importance in several industries e.g., batteries, pigments, metal smelting, the production of lead arsenate pesticides and lead-containing water pipes (Hanfi et al., 2020). Lead pollution can result in reversible injury to the central nervous, hematopoietic, hepatic, and renal systems failures (Patel et al., 2018).
Bacteria possess several metabolism-dependent and metabolism-independent methods for the uptake and accumulation of toxic metals. Bioremediation is an approach for metal recovery and remediation is the elimination of these hazardous metals from soil, sludge, and wastewater via microbe-based technology (Manoj et al., 2020). Microorganisms possess several cellular mechanisms for efflux, extracellular sequestration, metal modification, and precipitation of Lead ions, among others (Pratush et al., 2018). The microbial cells might potentially modify their shape to promote the generation of the siderophores to support the intracellular bioaccumulation of Lead (Sharma et al., 2018). Bacillus amyloliquefaciens has been used as a biocontrol agent (Ngalimat et al., 2021) to enhance plant growth directly (by nitrogen fixation, phytohormone production and phosphate solubilization) or indirectly by improving the plant health (Chowdhury et al., 2015). The forceful effect of Bacillus amyloliquefaciens in promoting plant growth activity as well as organophosphorus pesticide (OP) removal, paved the way for more recent investigations of the possible use of Bacillus amyloliquefaciens in agricultural and environmental remediations (Meng et al., 2019). Moreover, Maarof et al. (2018) reported that Bacillus amyloliquefaciens strain KIK-12 can effectively reduce molybdate to molybdenum with optimum molybdate concentration ranging between 30 and 50 mM.
The adsorption process is a metabolically independent method of heavy metal accumulation that relies mostly on surface features, such as some main functional groups present on the cell membrane and electrostatic interactions (Zabochnicka-witek & Krzywinos, 2014). The process of adsorption is also impacted by numerous elements like pH, temperature, ionic strength, absorbent quantity, and size, etc., (Timkova et al., 2018). Teng et al. (2019) isolated and identified some potent strains of inorganic phosphate-solubilizing bacteria (iPSB) capable of immobilizing lead by dissolving insoluble phosphate components. In the present study, native lead resistant microorganisms were isolated from industrial effluents contaminated soil and screened for their bioremediation potential. The most potent lead resistant microorganism was identified and statistically optimized using Taguchi design to maximize the lead removing efficiency. Moreover, mechanistic studies of the possible bioremediation mechanisms were investigated (Fig. 1).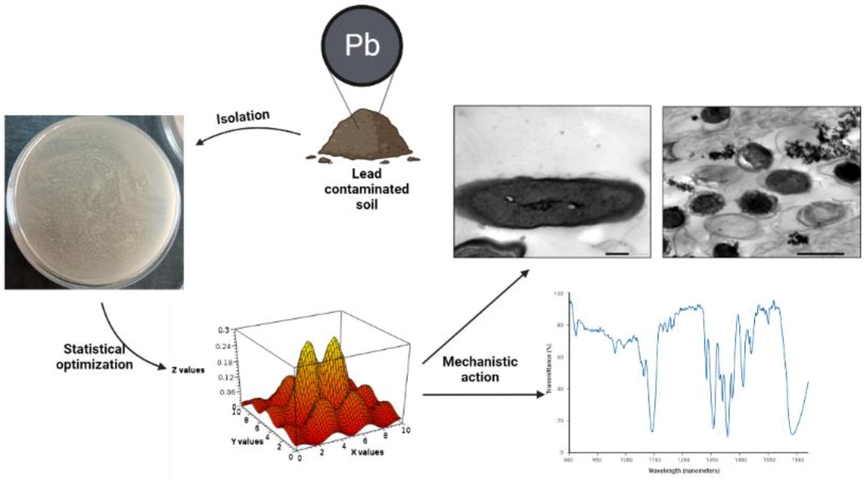
Graphical presentation of the study design.
2 Materials and methods
2.1 Sample collection
Soil samples were obtained from industrial effluents polluted area in Alexandria, Egypt. All soil samples were stored in labelled sterile containers before being delivered to the laboratory for analysis (Kang et al., 2015).
2.2 Isolation of lead resistant bacteria
The lead resistant bacteria were isolated from soil samples using serial dilution technique where 1 g soil was suspended in sterilized distilled water (10 mL), vortexed for 10 min then serially diluted up to 10-6 dilutions. 100 µL of each dilution were distributed over lead-containing (100 ppm) nutrient agar medium using spread plate technique. The plates were incubated for 48 hrs at 37 °C. Single colonies were purified through streaking technique over lead-containing (100 ppm) nutrient agar medium (Dey et al., 2016).
2.3 Detection of the minimum inhibitory concentration of lead
The minimal inhibitory concentration (MIC) of lead was assessed using serial dilutions ranged from 1000 to 25000 ppm. Nutrient agar plates were supplemented with 100 µL of each lead concentration. 0.5 McFarland of lead-resistant bacterial suspensions (1.5 × 108 CFU/mL) was adjusted (OD 800 nm) then, 20 μL of each suspension was streaked over the lead-supplemented nutrient agar plates surface. The inoculated plates were daily observed for a week to assess the bacterial growth. The minimum inhibitory concentration was defined as the lowest concentration that inhibited the visible microbial growth (Nokman et al., 2019). A single colony of the most promising bacterial strain with the highest MIC value was selected and identified by 16S rRNA gene sequencing (Sharma & Shukla, 2021). The bacterial DNA was extracted and amplified against forward (5́-AGTTTGATCCTGGCTCAG-3́) and reverse (5́-GGCTTACCTTGTTACGACTT-3́) 16S rRNA primers. Multiple sequence alignment was performed after sequencing the 16S rDNA in accordance with the National Center for Biotechnology Information (NCBI) database. Finally, the phylogenetic tree of the most resistant lead isolate was created through distance matrix analysis using the NT system (Shahin et al., 2022).
2.4 Bacterial lead removal optimization using experimental Taguchi statistical design
Qualitek-4 software (V. 14.5, Nutek Inc., MI, USA) was used to design Taguchi statistical experiments for optimizing the environmental (lead concentration, pH, incubation temperature, inoculum size, inoculum age and incubation time) and nutritional (peptone, beef extract, yeast extract, NaCl) factors to maximize the bacterial bioremediation of lead (Taran et al., 2015). Table 1 showed the factors and their levels, while Table 2 showed the experimental matrix. Each run was prepared and conducted according to the experimental Taguchi statistical design (Table 2) then at end of each experiment, flasks were centrifuged (12000 rpm for 5 min) and the supernatants were assessed for lead concentration using atomic absorption spectrophotometry. All the experiments were done in triplicates (Taran et al., 2013).
Factor
Level 1
Level 2
Level 3
X1
Incubation time (h)
48
72
96
X2
pH
5
7
9
X3
Lead concentration (ppm)
5000
10,000
15,000
X4
Incubation temperature (℃)
30
35
40
X5
Inoculum size (ml)
5
10
15
X6
Inoculum age (h)
24
48
72
X7
Peptone (g)
0.5
1.5
2.5
X8
Beef extract (g)
0.5
1.5
2.5
X9
Yeast extract (g)
0.5
1.5
2.5
X10
NaCl (g)
4
5
6
X1
X2
X3
X4
X5
X6
X7
X8
X9
X10
1
1
1
1
1
1
1
1
1
1
1
2
1
1
1
1
2
2
2
2
2
2
3
1
1
1
1
3
3
3
3
3
3
4
1
2
2
2
1
1
2
2
2
2
5
1
2
2
2
2
2
3
3
3
3
6
1
2
2
2
3
3
1
1
1
1
7
1
3
3
3
1
1
1
3
3
3
8
1
3
3
3
2
2
2
1
1
1
9
1
3
3
3
3
3
3
2
2
2
10
2
1
2
3
1
2
3
1
2
3
11
2
1
2
3
2
3
1
2
3
1
12
2
1
2
3
3
1
2
3
1
2
13
2
2
3
1
1
2
3
2
3
1
14
2
2
3
1
2
3
1
3
1
2
15
2
2
3
1
3
1
2
1
2
3
16
2
3
1
2
1
2
3
3
1
2
17
2
3
1
2
2
3
1
1
2
3
18
2
3
1
2
3
1
2
2
3
1
19
3
1
3
2
1
3
2
1
3
2
20
3
1
3
2
2
1
3
2
1
3
21
3
1
3
2
3
2
1
3
2
1
22
3
2
1
3
1
3
2
2
1
3
23
3
2
1
3
2
1
3
3
2
1
24
3
2
1
3
3
2
1
1
3
2
25
3
3
2
1
1
3
2
2
2
1
26
3
3
2
1
2
1
3
3
3
2
27
3
3
2
1
3
2
1
1
1
3
2.5 Statistical analysis
Taguchi DOEs results were statistically analyzed using fixed-effects model of analysis of variance (ANOVA) in Qualitek-4 software (Taran et el., 2015).
2.6 Mechanistic action of the Pb removal
In order to determine the possible bio-adsorption mechanism, cells before (control) and after Pb2+ exposure was collected by centrifugation at 940 × g for 3 min then the bacterial pellets were washed twice with PBS buffer (Zhu et al., 2019), and studied using transmission electron microscope (TEM) (JEM-100 CX Joel).
Moreover, the control, the treated cells and the supernatant were lyophilized and Fourier-transform infrared spectroscopy (FTIR) (Perkin Elmer, Inc., Waltham, MA, USA) was applied to study the variations of functional groups on the bacterial cell surfaces. Lyophilized samples were studied through energy-dispersive X-ray spectroscopy (EDX) to determine the Pb content% in the prepared samples (control, treated supernatants and treated cell pellets).
2.6.1 Pb compound characterization
The untreated (control) and Pb2+ ions treated cells were collected, washed with deionized distilled water, lyophilized and subjected to X-Ray diffraction (XRD) analysis (XRD-BRUKER AXS, Cu Kα radiation source (λ = 0.154 nm), scan rate = 10˚/ min).
3 Results and discussion
3.1 Isolation of lead-resistant bacterial colonies
From lead-contaminated soils, certain lead-resistant bacterial strains were identified. The microbial communities in the examined soils evolved a tolerance mechanism to hazardous chemicals (Wang et al., 2018). Five distinct organisms were chosen for further study from various colonies found on nutrient agar plates treated with 100 ppm lead.
3.2 Determination of lead MIC
Maximum lead tolerance was 15000 ppm for isolate 2 followed by 1000, 5000, 5000 and 10000 ppm for isolates 3, 5, 1 and 4 respectively. Therefore isolate 2 was selected and identified (as the highest lead resistant isolate) using 16 s rRNA profile. Similarly, Rittmann (2004) mentioned that Aspergillus niger RH 17 showed the highest lead resistivity reaching 15000 ppm when grown on lead-supplemented agar plates. However, Acinetobacter strain DF4/PUTK2 showed increased reduction percentage with increased concentration of lead up to 15000 ppm, while 30000 ppm reported a lethal effect (Muhammad et al., 2021). Ansari and Malik, (2007) reported the lead MIC value of Bacillus spp. as 1600 ppm. On the other hand, Raja et al. (2006) stated that Bacillus spp. could resist lead concentration up to 8000 ppm.
3.3 Identification of isolate 2 using 16S rRNA profile
Isolate 2 was selected and identified as Bacillus amyloliquefaciens with 99.3% similarity and then deposited at GenBank with accession number ON261680.1. Fig. 2 showed the phylogenetic tree of Bacillus amyloliquefaciens ON261680.1.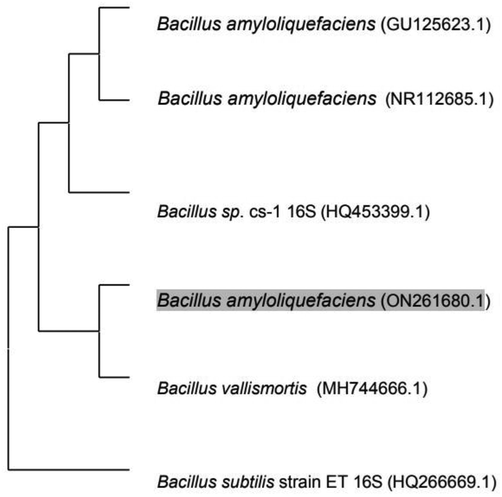
Phylogenetic tree of Bacillus amyloliquefaciens ON261680.1.
3.4 Bioremediation of lead according to DOEs of environmental and nutritional factors
Optimum temperature, pH, concentration, inoculum size, inoculum age, incubation time, peptone, yeast extract, beef extracts and NaCl concentrations were evaluated using Taguchi optimization matrix demonstrated 27 orthogonal arrays (Table 2) with regression equation:
In the regression equation (Eq. (1), the positive values signify the positive effect of the tested variables while the negative values indicate their inverse effect on the response (remaining lead concentration) (Omidi et al., 2017). Data in Table 3 showed that the highest bioremediation of lead by Bacillus amyloliquefaciens ON261680.1 was 99.840 % at 30 °C, 10000 ppm, pH 9, 96 h incubation, 15 mL inoculum size, 48 h inoculum age, 0.5 g peptone, 0.5 g yeast extract, 1.5 g beef extract and 6 g NaCl. Fig. 3 showed the interactions between the significant factors (concentration, temperature, pH time). Raja et al. (2006) revealed that Pseudomonas BC15 and Bacillus sp. J1 showed 65 and 75% Lead accumulation respectively. However, Wang et al. (2018) reported that heavy-metal uptake is an exothermic reaction, and the temperature elevation destroys the metal uptake sites on the surface of the bacterial cell wall or induces ion exudation into solution by bacteria. Gómez-Ramírez et al. (2015) studied Microbacterium oxydans CM3 and CM7 Pb-bioremediation optimization potential using response surface methodology (RSM). The optimum conditions for Pb removal by strain CM3 was observed at pH 6.81, temperature 35.31 °C, the Pb concentration of 337.35 mg/L, with 60.42% removal. However, strain CM7 showed maximum Pb remediation at temperature 35.65, pH 5.28, the Pb concentration of 346.95 mg/L and the Pb removal was 59.05%.
Time (h)
pH
Conc (ppm)
Temp (℃)
Inoculum size (ml)
Inoculum age (h)
Peptone (g)
Beef extract (g)
Yeast extract (g)
NaCl (g)
Remaining lead (ppm)
1
48
5
5000
30
5
24
0.5
0.5
0.5
4
44.56
2
48
5
5000
30
10
48
1.5
1.5
1.5
5
48.256
3
48
5
5000
30
15
72
2.5
2.5
2.5
6
41.97
4
48
7
10,000
35
5
24
0.5
1.5
1.5
5
129.4
5
48
7
10,000
35
10
48
1.5
2.5
2.5
6
121.9
6
48
7
10,000
35
15
72
2.5
0.5
0.5
4
113.837
7
48
9
15,000
40
5
24
0.5
2.5
2.5
6
236.3
8
48
9
15,000
40
10
48
1.5
0.5
0.5
4
221.4
9
48
9
15,000
40
15
72
2.5
1.5
1.5
5
228.1
10
72
5
10,000
40
5
48
2.5
0.5
1.5
6
124.27
11
72
5
10,000
40
10
72
0.5
1.5
2.5
4
112.28
12
72
5
10,000
40
15
24
1.5
2.5
0.5
5
132.8
13
72
7
15,000
30
5
48
2.5
1.5
2.5
4
229.9
14
72
7
15,000
30
10
72
0.5
2.5
0.5
5
210.22
15
72
7
15,000
30
15
24
1.5
0.5
1.5
6
209.9
16
72
9
5000
35
5
48
2.5
2.5
0.5
5
33.99
17
72
9
5000
35
10
72
0.5
0.5
1.5
6
30.93
18
72
9
5000
35
15
24
1.5
1.5
2.5
4
32.36
19
96
5
15,000
35
5
72
1.5
0.5
2.5
5
228.51
20
96
5
15,000
35
10
24
2.5
1.5
0.5
6
227.344
21
96
5
15,000
35
15
48
0.5
2.5
1.5
4
212.22
22
96
7
5000
40
5
72
1.5
1.5
0.5
6
28.33
23
96
7
5000
40
10
24
2.5
2.5
1.5
4
25.35
24
96
7
5000
40
15
48
0.5
0.5
2.5
5
29.37
25
96
9
10,000
30
5
72
1.5
2.5
1.5
4
34.33
26
96
9
10,000
30
10
24
2.5
0.5
2.5
5
30.47
27
96
9
10,000
30
15
48
0.5
1.5
0.5
6
16.0
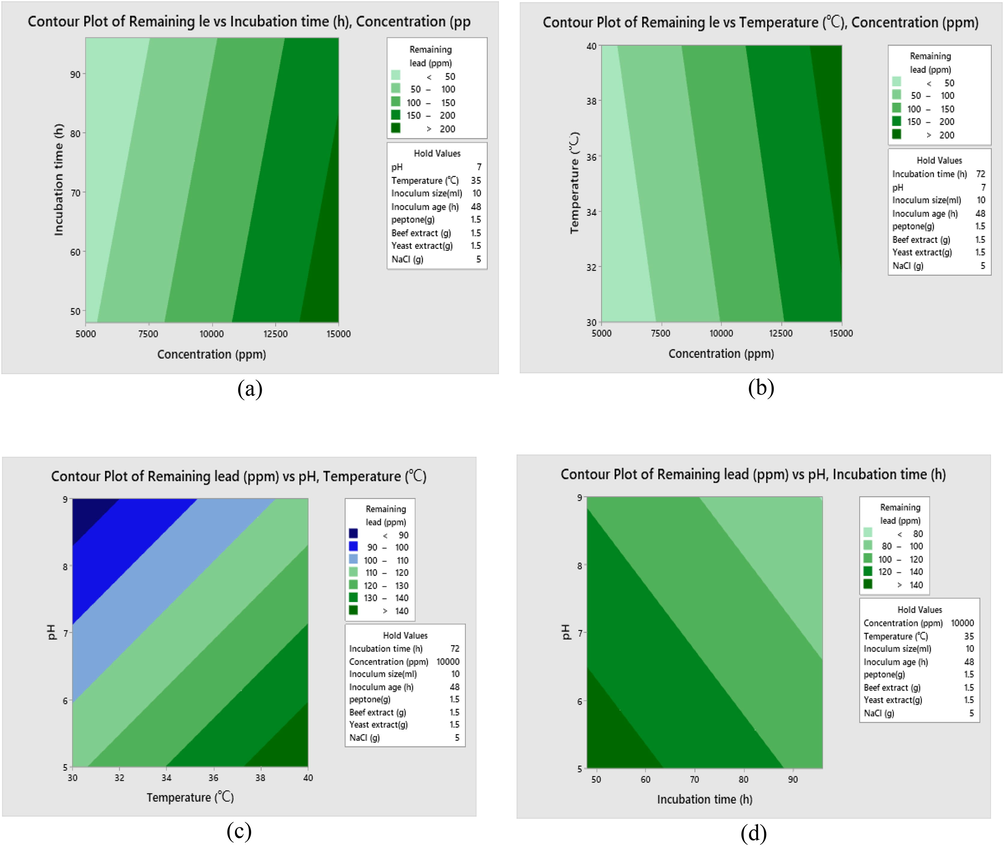
Surface plot of nutritional and environmental factors using Taguchi DOEs: remaining lead versus initial lead concentration and incubation time (a), remaining lead versus initial lead concentration and temperature (b), remaining lead versus pH and temperature (c) and remaining lead versus pH and incubation time (d).
3.5 Statistical analysis of Taguchi DOEs
Statistical analysis was done using fixed-effects model of analysis of variance (ANOVA) in Qualitek-4 software. Analysis of variance showed that time, pH, concentration and temperature were the significant factors (p-value < 0.05) while inoculum size, inoculum age, peptone, beef extract, yeast extract and NaCl were not significant (p-value ˃ 0.05) (Fig. 3a,b,c,d and 4b). Metal biosorption is a complex process includes ion exchange, precipitation and complexation (Meng et al., 2020). Analysis of variance (ANOVA) results (Table 4) are based on calculation correspond to sum of the squares which applied for calculation of factors effect, Fisher’s F-ratios and P-values were also represented. The normal probability of residuals (Fig. 4c) indicates almost no serious destruction of the DOE assumptions. The model summary presents high R2-value of 93.65% for Pb removal and specifies that there is agreement between the experimental and predicted outcomes. Satisfactory normal distribution of the results confirms the normality assumptions made earlier and the independence of the residuals (Dil et al., 2016). The results correspond to plotting residual values vs. the number of experiments (Fig. 4a) shows random distribution of the residual values around zero and proof accuracy of the selected model (Asfaram et al., 2016).
Source
DF
Adj SS
Adj MS
F-Value
P-Value
Regression
10
175,343
17,534
23.58
0.000
Time
1
6954
6954
9.35
0.008
pH
1
5282
5282
7.10
0.017
CONC
1
158,443
158,443
213.11
0.000
TEMP
1
4128
4128
5.55
0.032
Inoculum size
1
296
296
0.40
0.537
Inoculum age
1
89
89
0.12
0.734
Peptone
1
64
64
0.09
0.773
Beef extract
1
14
14
0.02
0.893
Yeast extract
1
66
66
0.09
0.769
NaCl
1
6
6
0.01
0.927
Error
16
11,896
743
Total
26
187,238
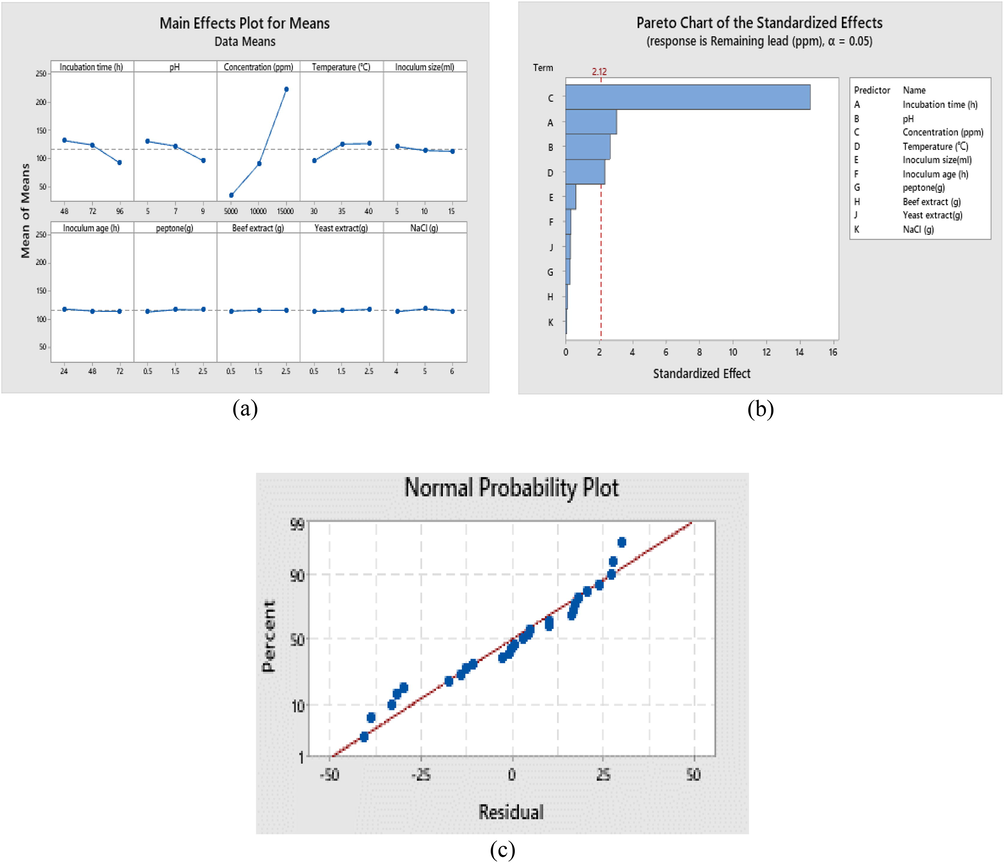
Main effects of the studied variables (a), Pareto chart (b) and normal plot of the optimized lead removal using Taguchi DOEs.
Model Summary.
S
R-sq
R-sq(adj)
R-sq(pred)
27.2669
93.65%
89.68%
82.61%
3.6 Mechanistic action of the Pb removal using Bacillus amyloliquefaciens ON261680.1
EDX analysis of untreated control supernatant revealed the high presence of Pb ions while no traces of Pb ions were noticed in the biologically treated supernatant sample (Fig. 5a and c). It is worth noting that Pb was present on the treated bacterial cell surface (Fig. 5b) which clearly indicates the possible bio-adsorption of Pb ions with possible formation of Pb-containing compounds on Bacillus amyloliquefaciens ON261680.1 cells surface. Chen et al. (2016) suggested the possible mechanism behind the observed lead precipitation as insoluble complexes on the microbial cell surface that can be attributed to the presence of phosphate, amine, hydroxyl and carboxyl groups which were served as metal attachment sites on the cell surface. Consistently, the present study supported that several functional groups of the cell wall (e.g., O–H, N–H, amide C = O and P-O as indicated by FTIR, Fig. 7) provide a net overall negative charge to the cell surface, and concurrently a high binding affinity for Pb2+ metal cations via counterion interactions. At the same time, the presence of single pair electrons on N and O delocalizes into the 6p empty orbit of Pb2+ and can form coordination bond. (Fig. 9).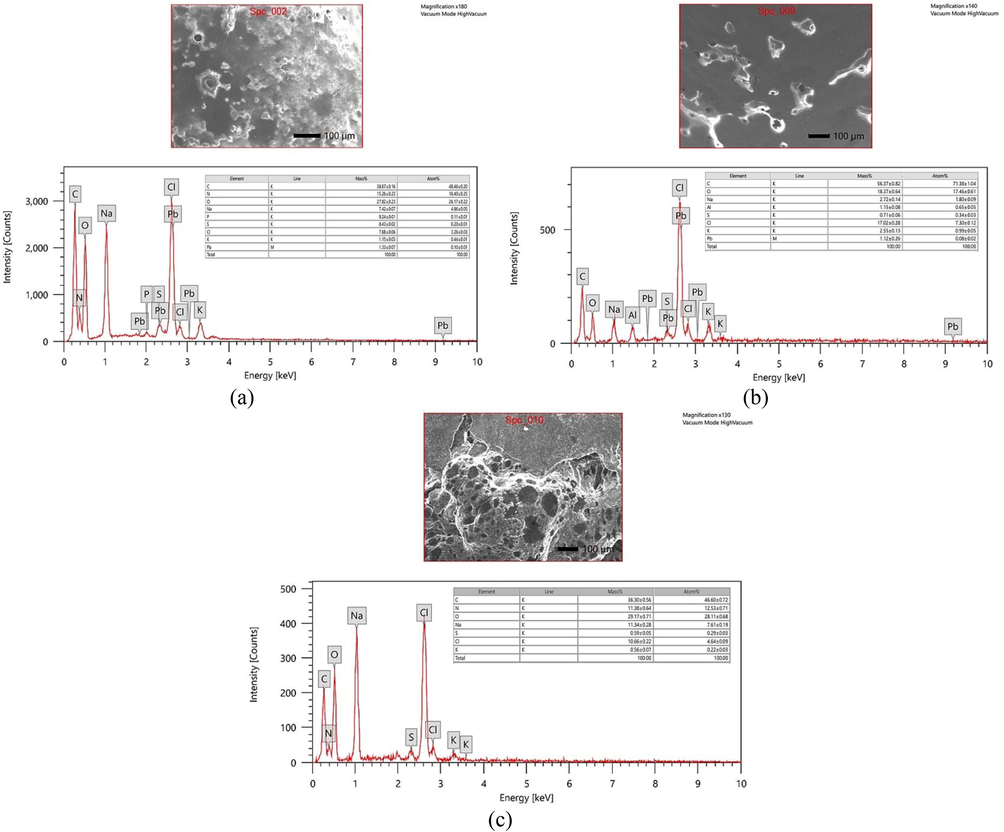
EDX analysis of control untreated culture (a), bacterial cell surface (b) and cultural supernatant (c) after treatment.
Transmission electron microscopy (TEM) was applied to Bacillus amyloliquefaciens ON261680.1 control cells before and after incubation with lead using the optimum conditions. Fig. 6A indicated that the control bacterial cells showed typical intact cell with native morphology. While Fig. 6B showed lead bio-adsorption inside the Bacillus amyloliquefaciens ON261680.1 cells. Zhu et al. (2022) mentioned that the surface of Acinetobacter calcoaceticus/IBWS700 appeared stacked, wrinkled, and shortened with the appearance of a large number of stacks after Pb adsorption, which may be due to these reasons, i, physical adsorption related to the special surface and the microporous structure of the extracellular polysaccharides secreted by iPSB20–3; ii, electrostatic adsorption dependent on the pH; iii, complexation, substitution, and cation-π interactions between Pb and functional groups on the IBWS700 surface (Deng et al., 2019).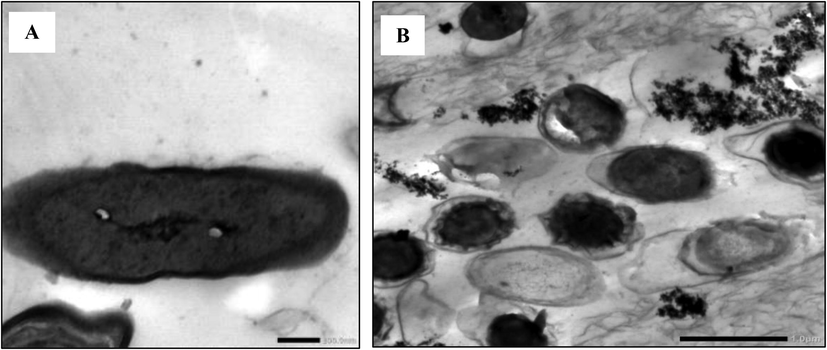
TEM examination of Bacillus amyloliquefaciens ON261680.1 control cells (A) and after incubation with lead (B).
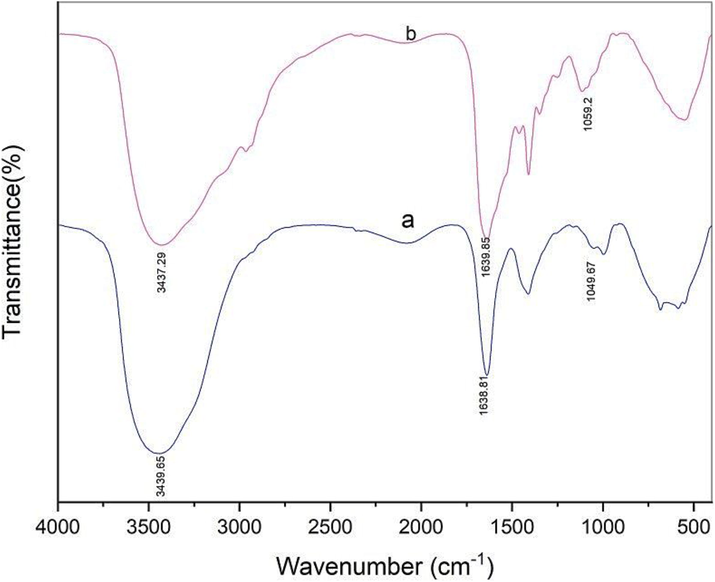
FTIR analyses of bacterial cells before (blue) and after (red) treatment with Pb.
FTIR spectra was executed to assess whether the binding of Pb(II) ions to Bacillus amyloliquefaciens ON261680.1 cells involved the surface functional groups. Due to the interaction of Pb(II) ions with the surface functional groups, the peaks may shift to lower or higher wavenumbers after lead loading. The spectra are depicted in Fig. 7 and the corresponding results are listed in Table 5. The spectra confirmed alterations of molecular vibrations of specific functional groups in the bacterial cell surface before and after adsorption of Pb(II). The Bacillus cell walls are known to contain large amounts of anionic polymers, teichoic acids and teichuronic acids (Tempest et al., 1968). The distinct absorption bands before and after the adsorption process are corresponding to the stretching vibrations of O–H, N–H, amide C = O and P-O of PO4-3. These absorption bands (Table 5) were shifted after adsorption of Pb(II) ions as shown in Fig. 7. It was found that the stretching vibration bands of O–H and N–H bonds were shifted by 2.36 cm−1 to higher wavenumbers. This shifting of peaks to higher wavenumbers can be attributed to increased bond strength. In contrast, the absorption bands of amide C = O and phosphate P-O bonds were shifted to lower wavenumbers by ca. 1.04 and 9.53 cm−1, respectively, indicating the bond weakening. The change in wavenumber of stretching vibrations of the functional groups displayed that these functional groups can be responsible for the effective performance of bacteria during Pb(II) uptake. Accordingly, we could argue that the Pb(II) ions are bound to the functional groups of Bacillus amyloliquefaciens ON261680.1 cell surface through complexation mechanism by providing overall negative charge of the bacterial cell surface along with selective chelating agents for binding to this toxic Pb2+ metal cations.
Functional Groups of IR absorption bands
Wavenumber cm−1
Before adsorption of Pb(II) ion
After adsorption of Pb(II) ion
Shift differences
Absorption bands
O–H
3439.65
3437.29
2.36
N–H
C = O (amide)
1638.81
1639.85
1.04
P-O
1049.67
1059.20
9.53
The information about attribution of Pb bio-precipitation and bio-adsorption by microbial metabolism is relatively rare (Zhu et al., 2019). XRD of the treated bacterial cell surface (Fig. 8) showed strong peak at 30.287◦, corresponding to pyromorphite (Pb5(PO4)3Cl (Log Ksp = − 84.4)) (Zhang et al., 2020) as indicated by comparing the XRD spectra in Fig. 8b with the standard card (JCPDS card: 00–019-0701) provided by the International Centre for Diffraction Data using X Pert High Score Plus. The supernatants contained many components from the culture and metabolites of Bacillus amyloliquefaciens ON261680.1, such as PO4 2-, Cl- and low molecular weight organic acids, which could react with Pb2+ for the formation of the corresponding insoluble complex. Furthermore, the significance of pH factor observed in the optimization of Bacillus amyloliquefaciens ON261680.1 indicates that ion exchange is also involved in lead removal (Fig. 9).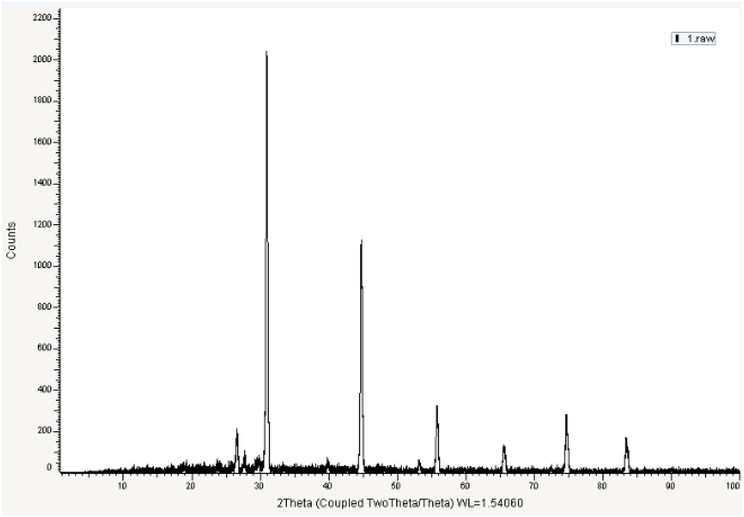
XRD analysis of Pb2+ treated cells.
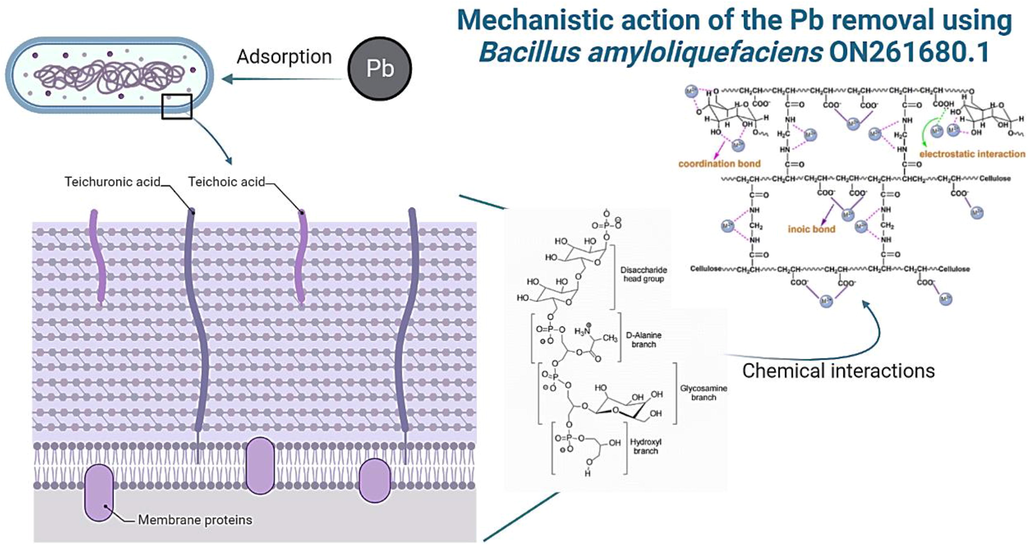
Mechanism of Lead adsorption in Bacillus amyloliquefaciens ON261680.1 cells.
This was in agreement with that reported by Jaafar et al. (2016) who declared the possible mechanism of Pb bioremediation using Deinococcus radiodurans is through Pb (II) immobilization on microbial cell surface.
Biomineralization of heavy metals through biological treatment, especially microbial bioremediation was recently regarded as an efficient and attractive remediation technique and has been widely employed in situ (Chen et al., 2016). Several studies have proposed a significant mechanism of biomineralization associated with the formation of pyromorphite (Bai et al., 2014) which is a stable insoluble complex that hindering Pb bioavailability and toxicity in the environment (Mohapatra et al., 2019). Moreover, pyromorphite formation is suggested as a remediation management for Pb immobilization (Meng et al., 2020).
4 Conclusion
Data of the present study reported that Bacillus amyloliquefaciens ON261680.1 can be considered as a novel lead- resistant bacterium isolated from industrial effluents. The results concluded that Taguchi approach was an effective statistical design in optimizing the environmental and nutritional factors for the maximum bioremediation of lead by Bacillus amyloliquefaciens ON261680.1 reaching 99.84% removal. Furthermore, several mechanistic action studies showed that lead was deposited and precipitated on Bacillus amyloliquefaciens ON261680.1 cell surface through EDX, XRD, FTIR and TEM studies. Data confirmed the evolvement of the cell wall functional groups in Pb bio-adsorption on the bacterial cell surface in a form of pyromorphite (Pb5(PO4)3Cl through complexation mechanism by providing overall negative charge to the cell surface along with selective chelating agents for binding to this toxic Pb2+ metal cations. In general, the present study indicates the effectiveness of Bacillus amyloliquefaciens ON261680.1 in lead bioremediation to adjuvant pyromorphite (insoluble and less toxic complex) which can pave the way for biological treatment of industrial wastewater and polluted areas.
5 Financial & competing interests’ disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties. No writing assistance was utilized in the production of this manuscript.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Biosorption of nickel and cadmium by metal resistant bacterial isolates from agricultural soil irrigated with industrial wastewater. Bioresour. Technol.. 2007;98(16):3149-3153.
- [Google Scholar]
- Statistical experimental design, least squares-support vector machine (LS-SVM) and artificial neural network (ANN) methods for modeling the facilitated adsorption of methylene blue dye. RSC Adv.. 2016;6(46):40502-40516.
- [Google Scholar]
- Biosorption mechanisms involved in immobilization of soil Pb by Bacillus subtilis DBM in a multi-metal-contaminated soil. J. Environ. Sci.. 2014;26(10):2056-2064.
- [Google Scholar]
- Biomineralization of Pb (II) into Pb-hydroxyapatite induced by Bacillus cereus 12–2 isolated from Lead-Zinc mine tailings. J. Hazard. Mater.. 2016;301:531-537.
- [Google Scholar]
- Biocontrol mechanism by root-associated Bacillus amyloliquefaciens FZB42–a review. Front. Microbiol.. 2015;6:780.
- [Google Scholar]
- Adsorption behaviour and mechanisms of cadmium and nickel on rice straw biochars in single-and binary-metal systems. Chemosphere. 2019;218:308-318.
- [Google Scholar]
- Isolation and characterization of arsenic-resistant bacteria and possible application in bioremediation. Biotechnol. Rep,. 2016;10:1-7.
- [Google Scholar]
- Modeling of quaternary dyes adsorption onto ZnO–NR–AC artificial neural network: analysis by derivative spectrophotometry. J. Ind. Eng. Chem.. 2016;34:186-197.
- [Google Scholar]
- Microbacterium oxydans and Microbacterium liquefaciens: A biological alternative for the treatment of Ni-V-containing wastes. J. Environ. Sci. Health A. 2015;50(6):602-610.
- [Google Scholar]
- Heavy metal contamination in urban surface sediments: sources, distribution, contamination control, and remediation. Environ. Monit. Assess.. 2020;192(1):1-21.
- [Google Scholar]
- Biosorption of some heavy metals by Deinococcus radiodurans isolated from soil in Basra Governorate-Iraq. J. Bioremediat. Biodegrad.. 2016;7(332):2.
- [Google Scholar]
- Isolation and characterization of heavy-metal-resistant bacteria and their applications in environmental bioremediation. Int. J. Environ. Sci. Technol.. 2020;17(3):1455-1462.
- [Google Scholar]
- Bioremediation of lead by ureolytic bacteria isolated from soil at abandoned metal mines in South Korea. Ecol. Eng.. 2015;74:402-407.
- [Google Scholar]
- A critical review on speciation, mobilization and toxicity of lead in soil-microbe-plant system and bioremediation strategies. Ecotoxicol. Environ. Saf.. 2018;147:1035-1045.
- [Google Scholar]
- Isolation and Characterization of a Molybdenum-reducing Bacillus amyloliquefaciens strain KIK-12 in Soils from Nigeria with the Ability to grow on SDS. J. Environ. Microbiol. Toxicol.. 2018;6(1):13-20.
- [Google Scholar]
- Understanding the molecular mechanisms for the enhanced phytoremediation of heavy metals through plant growth promoting rhizobacteria: a review. J. Environ. Manage.. 2020;254:109779
- [Google Scholar]
- Lead removal from water by a newly isolated Geotrichum candidum LG-8 from Tibet kefir milk and its mechanism. Chemosphere. 2020;259:127507
- [Google Scholar]
- Complete genome sequence of Bacillus amyloliquefaciens YP6, a plant growth rhizobacterium efficiently degrading a wide range of organophosphorus pesticides. J. Integr. Agric.. 2019;18:2668-2672.
- [Google Scholar]
- Active and passive biosorption of Pb (II) using live and dead biomass of marine bacterium Bacillus xiamenensis PbRPSD202: Kinetics and isotherm studies. J. Environ. Manage.. 2019;247:121-134.
- [Google Scholar]
- Studying the behavior of the light-off bioreporter DF4/PUTK2 as a light-on assay against lead. Biocatal. Agric. Biotechnol.. 2021;35:102069
- [Google Scholar]
- A review on the biotechnological applications of the operational group Bacillus amyloliquefaciens. Microorganisms. 2021;9(3):614.
- [Google Scholar]
- Screening and molecular identification of heavy metal resistant Pseudomonas putida S4 in tannery effluent wastewater. Biocatal. Agric. Biotechnol.. 2019;18:101052
- [Google Scholar]
- Synthesis and characterization of Au-NPs supported on carbon nanotubes: application for the ultrasound assisted removal of radioactive UO22+ ions following complexation with Arsenazo III: spectrophotometric detection, optimization, isotherm and kinetic study. J. Colloid Interface Sci.. 2017;504:68-77.
- [Google Scholar]
- Heavy metal contamination in river water and sediments of the Swarnamukhi River Basin, India: risk assessment and environmental implications. Environ. Geochem. Health. 2018;40(2):609-623.
- [Google Scholar]
- Adverse effect of heavy metals (As, Pb, Hg, and Cr) on health and their bioremediation strategies: a review. Int. Microbiol.. 2018;21(3):97-106.
- [Google Scholar]
- Isolation and characterization of a metal-resistant Pseudomonas aeruginosa strain. World J. Microbiol. Biotechnol.. 2006;22(6):577-585.
- [Google Scholar]
- Rittmann, B. E. (2004). The new frontier of oxidized contaminants. In Proc. 4th Intl. Conf. Remediation of Chlorinated and Recalictrant Compounds. Paper 4C-29, in: A.R. Gavaskar and A.S.C. Chen (Eds.), Remediation of Chlorinated and Recalcitrant Compounds—2004. Proceedings of the Fourth International Conference on Remediation of Chlorinated and Recalcitrant Compounds (Monterey, CA; May 2004). ISBN 1-57477-145-0, published by Battelle Press, Columbus, OH, www.battelle.org/bookstore.
- Micrococcus lylae MW407006 pigment: production, optimization, nano-pigment synthesis, and biological activities. Biology. 2022;11(8):1171.
- [Google Scholar]
- Lead bioaccumulation mediated by Bacillus cereus BPS-9 from an industrial waste contaminated site encoding heavy metal resistant genes and their transporters. J. Hazard. Mater.. 2021;401:123285
- [Google Scholar]
- Contemporary enzyme based technologies for bioremediation: a review. J. Environ. Manage.. 2018;210:10-22.
- [Google Scholar]
- Optimal conditions for the biological removal of arsenic by a novel halophilic archaea in different conditions and its process optimization. Pol. J. Chem. Technol.. 2013;15(2)
- [Google Scholar]
- Biological removal of nickel (II) by Bacillus sp. KL1 in different conditions: optimization by Taguchi statistical approach. Pol. J. Chem. Technol.. 2015;17(3)
- [Google Scholar]
- Influence of growth condition on the concentration of potassium in Bacillus subtilis var. niger and its possible relationship to cellular ribonucleic acid, teichoic acid and teichuronic acid. Biochem. J.. 1968;106(1):237-243.
- [Google Scholar]
- Characterization of phosphate solubilizing bacteria isolated from heavy metal contaminated soils and their potential for lead immobilization. J. Environ. Manage.. 2019;231:189-197.
- [Google Scholar]
- Biosorption and bioaccumulation abilities of actinomycetes/streptomycetes isolated from metal contaminated sites. Separations. 2018;5(4):54.
- [Google Scholar]
- Isolation of lead-resistant Arthrobactor strain GQ-9 and its biosorption mechanism. Environ. Sci. Pollut. Res.. 2018;25(4):3527-3538.
- [Google Scholar]
- Potentials of biosorption and bioaccumulation processes for heavy metal removal. Pol. J. Environ. Stud.. 2014;23(2)
- [Google Scholar]
- Effective passivation of lead by phosphate solubilizing bacteria capsules containing tricalcium phosphate. J. Hazard. Mater.. 2020;397:122754
- [Google Scholar]
- The immobilization effects on Pb, Cd and Cu by the inoculation of organic phosphorus-degrading bacteria (OPDB) with rapeseed dregs in acidic soil. Geoderma. 2019;350:1-10.
- [Google Scholar]
- Bioremediation of lead-contaminated soil by inorganic phosphate-solubilizing bacteria immobilized on biochar. Ecotoxicol. Environ. Saf.. 2022;237:113524
- [Google Scholar]







