Translate this page into:
Microbiologically influenced corrosion of circulating cooling systems in power plants – A review
⁎Corresponding authors at: School of Materials Science and Engineering, University of Science and Technology of China, Shenyang 110016, PR China (Boxin Wei). ljchenxju@163.com (Lijuan Chen), bxwei17s@imr.ac.cn (Boxin Wei)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University. Production and hosting by Elsevier.
Abstract
The problems caused by microbiologically influenced corrosion (MIC) have become increasingly prominent, which has caused very serious economic losses. The research on the mechanism and protective measures of MIC is becoming more and more important. In this paper, combined with the unique environment of the circulating water cooling system, the influence of the unique environmental factors of the system on MIC is analyzed. Several kinds of bacteria known to cause microbial corrosion are summarized, including sulfate-reducing bacteria, nitrate-reducing bacteria, iron oxidizing bacteria, etc. The current MIC mechanisms and related research progress are reviewed. Several common MIC monitoring methods are summarized. The commonly used protection methods and the ideas of MIC technologies in the recent stage are listed. Finally, the future research direction of the MIC is prospected.
Keywords
Microbiologically influenced corrosion
Circulating cooling system
Corrosion mechanism
Sulfate reducing bacteria
Prevention
1 Introduction
Microbiologically Influenced Corrosion (MIC) is a phenomenon wherein the physiological activity of microorganisms within the biofilm, attached to the surface of a material, contributes to the corrosion or degradation of metallic materials through direct or indirect means (Wei et al., 2022; Zhou et al., 2022; Xu et al., 2023). According to relevant survey statistics, the cost of corrosion in the United States in 2013 reached 2.5 trillion U.S. dollars, accounting for more than 3.4 % of gross domestic product (GDP). Meanwhile, China's annual corrosion losses represent 3.4–5 % of its total GDP, equivalent to an expenditure of 3.8–5.7 trillion yuan in 2021. The loss attributed to MIC accounts for approximately 20 % of this total.
Since the discovery of MIC in 1910, the significance of MIC research has become increasingly evident. MIC exhibits both material and environmental universality, affecting materials such as carbon steel, stainless steel, and copper alloys. Microorganisms are commonly found in various environments, including the oil/gas industry, aviation systems, and marine infrastructure (Xu et al., 2023). As a critical component of industrial public systems, circulating cooling water in petrochemical, electric power, metallurgical, and other industries can account for 50 % to 90 % of a company's total water usage. However, MIC remains a significant concern in the field of circulating cooling water treatment. After multiple cycles of cooling water, microorganisms grow on pipe surfaces and form biofilms, leading to the MIC of steel pipes and even causing safety problems such as perforation and leakage. This paper reviews the characteristics, influencing factors, and research progress of MIC in industrial circulating cooling water systems, outlines control strategies of MIC, summarizes the main challenges in the current control process of MIC, and provides insights into the future research and protection of MIC in circulating cooling water systems.
2 Unique environments of circulating cooling water system
Due to the scarcity of fresh water in Northern China, reclaimed water from municipal wastewater discharge has been extensively utilized in circulating cooling systems of power plants. However, the water quality in these circulating cooling water systems significantly contributes to pipe corrosion and scaling due to the presence of Cl-, SO42-, and Ca2+. Environmental conditions such as pH, temperature, and nutrients promote microbial adherence and growth, as well as biofilm formation.
2.1 Cause of MIC
A circulating cooling water system is a type of water supply system designed for cooling and recycling purposes, encompassing both open and closed configurations. In Northern China, where water resources are scarce, the open-type circulating cooling water system is widely employed due to its water-saving capabilities. The water system typically has relatively low water quality requirements. As a result, reclaimed water can be utilized as a supplementary source for circulating cooling water in the thermal power plants, thereby reducing the demand for water resources. However, it should be noted that reclaimed water often exhibits high salinity, ammonia and nitrogen content, hardness, and complex microbial populations. The published standard practices can be used to guide diagnosis of MIC in the field:
-
●
TM0194-2014, Field Monitoring of Bacterial Growth in Oil and Gas Systems;
-
●
TM0212-2018-SG, Detection, Testing, and Evaluation of Microbiologically Influenced Corrosion on Internal Surfaces of Pipelines.
The unique environment of the circulating cooling system, combined with the specific water quality of reclaimed water, creates favorable conditions for the growth and reproduction of microorganisms.
2.2 Affecting factors
2.2.1 Temperature
In the circulating cooling water system of a thermal power plant, the circulating water temperature is typically maintained between 10 °C and 40 °C. This optimal temperature range encourages microorganisms to form biological sludge that adheres to the cooling water pipeline. As summarized in Table 1, the majority of these microorganisms thrive and reproduce at temperatures ranging from 20 °C to 40 °C. Within this temperature range, the microorganisms rapidly convert oxygen and methanol in the circulating water into carbonic acid, which further is transformed into carbon dioxide (CO2) for discharge. The CO2 produced during this process dissolves in water, forming carbonic acid, which subsequently decreas the pH and total alkalinity of the circulating water, resulting in the accelerating corrosion.
Group
T (°C)
pH
Metabolic mechanism
References
Sulfate-reducing bacteria (SRB)
0–70
4.5–9.0
Reduction of SO42- to S2- and H2S
(Yu et al., 2013)
Iron-reducing bacteria (IRB)
10–50
4–8
Reduction of Fe3+ to Fe2+
(Herrera and Videla, 2009)
Iron-oxidizing bacteria (IOB)
10–40
6–8
Oxidation of Fe2+ to Fe3+
(Liu et al., 2021)
Nitrate-reducing bacteria (NRB)
5–40
6–9
Reduction of nitrate to nitrogen or ammonia
(Starosvetsky et al., 2008)
Acid-producing bacteria (APB)
21–40
7–10
Metabolism produces acids that promote corrosion
(Xu et al., 2016)
In a thermodynamic perspective, a temperature increase results in more active statue of material in water, leading to an elevated diffusion coefficient. This enhancement in the diffusion coefficient accelerates the dispersion of dissolved oxygen in water towards the cathodic region of the corroded metal surface, consequently increasing the oxygen concentration difference in the vicinity of the corroded metal surface and expediting corrosion. Furthermore, a reduction in overpotential can also hasten the cathodic reduction process of oxygen or hydrogen ions and the anodic dissolution process of the metal, thereby increasing the metal's corrosion rate.
2.2.2 CO2
In the circulating cooling water system, the continuous circulation of water and the addition of supplementary water, coupled with the prolonged exposure of the cooling tower to the outdoor environment, result in the consistent ingress of CO2. Consequently, the CO2 dissolves in the circulating cooling water, forming carbonic acid. Table 2 shows the key reactions of pipeline steel in CO2-H2O natural gas system. The anodic reactions which occurs is the oxidation of Fe. The cathodic reactions are hydrogen evolution from a combination of H+, H2CO3, and HCO3−. Fe2+ ions released by anodic reaction will accumulate within the process fluid, participating in the products formation. Once the product of the activities of Fe2+ and CO32− ions reach and exceed a particular saturation limit, FeCO3 can form on steel surface, which is considered the main corrosion product at lower temperatures (generally below 80 °C) in CO2 environments.
Name
Reactions
Equilibrium Potential in V or pH
Iron dissolution
Hydrogen evolution
Products formation
Carbonic acid further reacts with water, forming bicarbonate and hydrogen ions, subsequently leading to a decrease in the pH of circulating cooling water. When the pH reaches a critical level, the protective oxide layer on the metal surface is compromised, exposing the underlying metal. Consequently, the metal reacts with ions, molecules, and oxygen present in the circulating cooling water, causing corrosion and damage. As the pH of circulating cooling water decrease, it provides a favorable environment for the growth and reproduction of microorganisms. Referring to Table 1, the optimal pH range for the proliferation of most corrosion-causing microorganisms is between 4 and 9. Prolonged exposure to this pH range accelerates microbial growth and reproduction, promoting biofilm formation and exacerbating metal corrosion.
Additionally, CO2 serves as a vital carbon source for microbial metabolism. An increase in carbon dioxide content enhances the growth rate and metabolic activity of microorganisms, thereby promoting microbial corrosion. Hence, it is crucial to control CO2 levels in the circulating cooling water systems of thermal power plants to prevent the formation of carbonic acid through the reaction of carbon dioxide and water. This control mitigates the reduction in pH and the corrosion rate of metals.
2.2.3 Ammonia and nitrogen contents
The elevated ammonia content in the supplementary water of the circulating cooling water system can adversely affect its normal operation. Ammonia nitrogen can foster the proliferation of microorganisms within the system, leading to the formation of slime and corrosion. These byproducts can coat the heat exchanger surface, diminishing its cooling efficiency and obstructing the cooling water channels. Moreover, this prevents corrosion inhibitors and scale inhibitors from reaching the metal surface, thereby inhibiting their protective functions and creating concentrated corrosion cells, which ultimately result in the corrosion of metal equipment.
Furthermore, the nitrification reaction of ammonia nitrogen in the circulating cooling water system generates a significant amount of nitric acid, causing a decrease in the system's pH. This results in acidic corrosion and electrochemical corrosion of pipes, such as copper and carbon steel pipes. Simultaneously, the cooling tower's cement structure may also be affected by acidic corrosion, leading to a sanding phenomenon.
Additionally, the presence of high levels of organic matter, phosphorus, and nitrogen in the medium water provides essential nutrients for algae. Ammonia nitrogen serves as a critical nutrient source for the nitrification reactions of nitrifying and nitrosating bacteria. The cooling water system offers favorable conditions for the growth of these bacteria.
2.2.4 Scale
The circulating water system in the thermal power plants provides an ideal environment for various microorganisms to thrive due to suitable temperatures, ample ventilation, and sufficient light. Consequently, the formation of biological scale is inevitable in such conditions. biological scale is a challenging-to-remove mixture of microorganisms, their metabolites, algae, suspended matter, and sediments formed during the water treatment process. In terms of microbial corrosion, biological scale can pose the following risks to circulating cooling water systems in thermal power plants:
Generation of acidic substances: Microorganisms and organic matter within biological scale can produce a significant amount of organic acids, hydrogen sulfide, and other acidic substances. This lowers the water's pH value, leading to metal surface corrosion and equipment damage.
-
●
Formation of microbatteries: Biological scale can create a microbial film on metal surfaces, forming microbatteries and causing microbial corrosion. Microorganisms produce various chemical substances through metabolic processes, such as acidic and redox substances, which induce electrochemical reactions on metal surfaces, resulting in corrosion.
-
●
Acceleration of metal corrosion: After biological scale formation on metal surfaces, it can adsorb and degrade organic matter, causing changes in the metal surface potential and accelerating metal corrosion.
-
●
Increased maintenance costs: Biological scale can adhere to metal surfaces, rendering equipment surfaces uneven and reducing heat transfer efficiency. Additionally, biological scale can cause blockages, clogs, and malfunctions, increasing equipment maintenance costs.
2.2.5 Flow rate
The flow rate is also an important factor causing corrosion of circulating water pipelines. In most cases, the flow of the medium will accelerate the corrosion of the material. In the high-speed flow stat, the surface of the metal is damaged by the interaction of mechanical erosion and electrochemical corrosion. On the one hand, the corrosion process leads to an increase in the surface roughness of the material and promotes the erosion process. On the other hand, the corrosion product film on the surface of the material is easily washed by the medium to break or fall off, and even produces erosion pits. The exposed metal and the surrounding corrosion product film form the characteristics of 'small anode-large cathode', which induces localized corrosion. At the same time, the erosion of the medium promotes the transport of the reactants towards the wall and the corrosion products away from the wall, thus promoting the corrosion process. However, the flow of the fluid medium on the metal surface does not always increase the corrosion rate of the metal. In some special cases, the corrosion rate of the metal decreases with the increase of the flow rate. For example, due to the erosion effect of a specific flow rate, the surface of the material is smoother and the product film is more uniform and dense, thereby reducing the corrosion rate.
2.2.6 Oxygen content
Thermal power plants usually use an open circulating water cooling system, which will make the oxygen content in the circulating water system higher. When it enters the cooler, after a long period of oxidative corrosion, there will be corresponding oxygen corrosion products. Corrosion products accumulate on the surface of the material to form a loose and porous corrosion layer. This can easily lead to the formation of oxygen concentration cell inside and outside the corrosion site, thus aggravating the oxygen corrosion. In addition, the excessive oxygen content in the system will lead to the growth and reproduction of aerobic bacteria such as iron-oxidizing bacteria, which will accelerate the corrosion of materials.
3 Microbial community characteristics
As early as 1895, despite the absence of any suggestion that microorganisms could contribute to material corrosion, Beijerinck (Beijerinck, 1895) reported that the presence of an anaerobic microorganism in soil with the ability to produce hydrogen sulfide (H2S) and metal sulfides. This organism was subsequently named “Spirillum desu‘lfuricans.” It was not until 1910 that Gaines first proposed the idea that microorganisms could indeed cause corrosion in metals. To date, an increasing number of microorganism species involved in corrosion processes have been identified. The commonly observed microbial species are presented in Table 3.
Group
Strain name
Phylum/class
Family
References
SRB
Desulfopila corrodens (IS4)
Deltaproteobacteria
Desulfobulbaceae
(Dinh et al., 2004)
Desulfovibrio ferrophilus (IS5)
Deltaproteobacteria
Desulfovibrionaceae
(Dinh et al., 2004)
NRB
Bacillus licheniformis ATCC 14580
Firmicutes
Bacillaceae
(Xu et al., 2013)
Prolixibacter sp. MIC1-1
Bacteroidetes
Prolixibacteraceae
(Iino et al., 2015)
Methanogenic archaea
Methanobacterium sp. IM1
Euryarchaeota
Methanobacteriaceae
(Dinh et al., 2004)
Methanococcus maripaludis KA1
Euryarchaeota
Methanococcaceae
(Uchiyama et al., 2010)
Methanococcus maripaludis Mic1c10
Euryarchaeota
Methanococcaceae
(Mori et al., 2010)
Methanosarcina acetivorans
Euryarchaeota
Methanosarcina
(Holmes et al., 2019)
Acetogenic bacteria
Sporomusa sp. GT1
Firmicutes
Veillonellaceae
(Kato et al., 2015)
3.1 SRB
According to previous research (Zhou et al., 2022), MIC induced by SRB is the most severe, accounting for approximately 70 % of cases. SRB can generate a micro-anaerobic environment on the steel surface through biofilm formation, subsequently obtaining energy for their growth and expediting the corrosion of pipeline steel via physiological metabolic activities. Within this environment, sulfate, sulfite, and monomeric sulfur are reduced to H2S by oxidizing organic compounds or molecular hydrogen. The study by Jia et al. (Jia et al., 2018) showed that the lower biogenic H2S concentration in the liquid phase resulted in enhanced MIC at neutral pH condition. Biogenic H2S is not the primary corrosive agent in SRB MIC. The depletion of hydrogen on the metal cathode surface results in a decrease in local partial pressure, acting as a cathodic depolarization (Fig. 1). Numerous studies have been conducted to investigate the corrosion characteristics of SRB on metallic materials. For example, Zhao et al. (Zhao et al., 2019) studied the effects of different chloride ion concentrations on the corrosion behavior of Q235 steel in sterile solution and SRB-containing solution. The results showed that the corrosion of Q235 steel will be promoted by SRB under the condition of lower chloride ion concentration. E. Ilhan et al. (Ilhan-Sungur et al., 2017) found that SRB could adhere to and form biofilms on the steel surface, even corroding materials with good corrosion resistance. Previous studies (Ilhan-Sungur et al., 2017; Sungur and Cotuk, 2005) used inorganic sulfide as a control and found that the metabolites of SRB also exhibited a substantial corrosive effect on 304 stainless steel.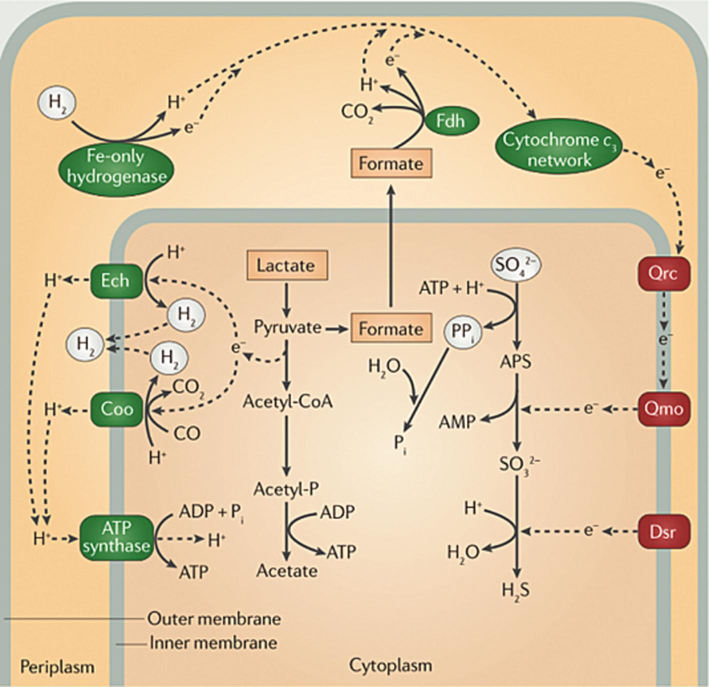
Anaerobic respiration process of SRB (Zhou et al., 2011).
It is important to note that SRB can use sugars, amino acids, alcohols, carboxylic acids and aromatic compounds as electron donors while reducing sulfur-containing or metallic compounds in multiple valence states (Dinh et al., 2004). However, most publications are based on the design of culture medium, which cannot fully demonstrate the operating conditions. The MIC induced by SRB in real world is more complex.
3.2 NRB
The significant production of nitrogen oxides during the combustion process in the thermal power plants results in the formation of nitrates through precipitation. These nitrates can stimulate the growth of NRB. NRB is a class of bacteria capable of reducing nitrate to nitrite and reducing nitrite to gaseous nitrogen compounds or isotropic conversion to NH4+ by denitrification. M. Etique et al. (Etique et al., 2014) reported that Klebsiella mobilis can reduce nitrate using organic carbon sources and can oxidize Fe2+ to Fe3+ through coupled nitrate reduction, with NRB effectively facilitating these reactions. Xu et al. (Xu et al., 2013) determined that NRB can form a biofilm on metal surfaces, a 14.5-depth biofilm could develop on the C1018 carbon steel surface in a one-week corrosion test, leading to the formation of 14.5 deep corrosion pits and a weight loss of 0.89 mg/cm2.
3.3 Iron-oxidizing bacteria (IOB)
Iron-oxidizing bacteria (IOB) are ubiquitously found in diverse environments (Emerson et al., 2010; Dong et al., 2018). These microorganisms exhibit various physiological characteristics, which can oxidize different forms of iron. During corrosion process, Fe2+ remains stable at low pH levels, however, once the pH reaches neutral or higher, Fe2+ becomes highly susceptible to oxidation by O2. Consequently, IOB can effectively utilize Fe2+ for energy metabolism under anaerobic conditions. As a result, IOB are predominantly autotrophic and strictly anaerobic, thriving in neutral, mildly acidic, or even highly acidic environments.
3.4 Acid-producing bacteria (APB)
Acid-producing bacteria (APB) generate various organic and inorganic acid metabolites, which lead to a decrease in pH within the microenvironment beneath the biofilm. This exposes the metal to chemical corrosion, potentially resulting in severe pitting or even perforation leakage. A typical APB, Acidithiobacillus caldus (A. caldus), forms biofilms that secrete sulfuric acid and hydrogen sulfide, which synergistically disrupt the passivation film on steel and accelerate pitting corrosion (Dong et al., 2018).
3.5 Slime-forming bacteria (SFB)
Slime-forming bacteria (SFB) represent a group of microorganisms capable of producing substantial amounts of extracellular polymeric substances (EPS). These substances primarily comprise polysaccharides, proteins, nucleic acids, and lipids, serving as the matrix structures for biofilms that form on metal surfaces. Common SFBs include Clostridium, Flavobacterium, Desulfovibrio, Desulfotomaculum, and Pseudomonas. By generating EPS, SFBs can modulate the oxygen concentration on the metal surface, thereby creating a microenvironment conducive to the growth of other microorganisms (Vigneron et al., 2016).
4 Corrosion mechanisms
The nature of MIC can be described as an abiotic process in which biological factors influence electro-/chemical corrosion. Currently, there is no definitive conclusion or definition regarding the MIC mechanism, as the process typically involves the synergistic effects of various mechanisms, making it impossible to identify a single, unified mechanism.
Contemporary research on microbial corrosion primarily involves macroscopic analysis and interpretation, focusing on factors such as the biofilm and metabolites produced by microorganisms, changes in metal surface composition and structural properties following corrosion, and the acquisition of metal surface images and electrochemical data to infer the underlying corrosion mechanism. Due to the complexity of microbial interactions and the variability of microbial corrosion, it is necessary to determine which corrosion mechanism plays a dominant role based on the specific corrosion situation.
Many scholars have investigated the mechanisms of MIC, with the prevailing mechanisms currently identified as: cathodic depolarization, metabolite-induced corrosion, concentration cell differences, and both direct and indirect electron transfer mechanisms.
4.1 Mechanism of cathodic depolarization
The cathodic depolarization mechanism is currently the most widely accepted explanation for MIC induced by SRB. This mechanism involves SRB adsorbing onto the metal surface, where they facilitate the electrochemical reduction of H+ to H in an anaerobic environment (see Fig. 2). These hydrogen atoms are subsequently converted to hydrogen sulfide (H2S) by the bacteria's hydrogenase enzymes, reducing the local partial pressure at the cathode and leading to the corrosion.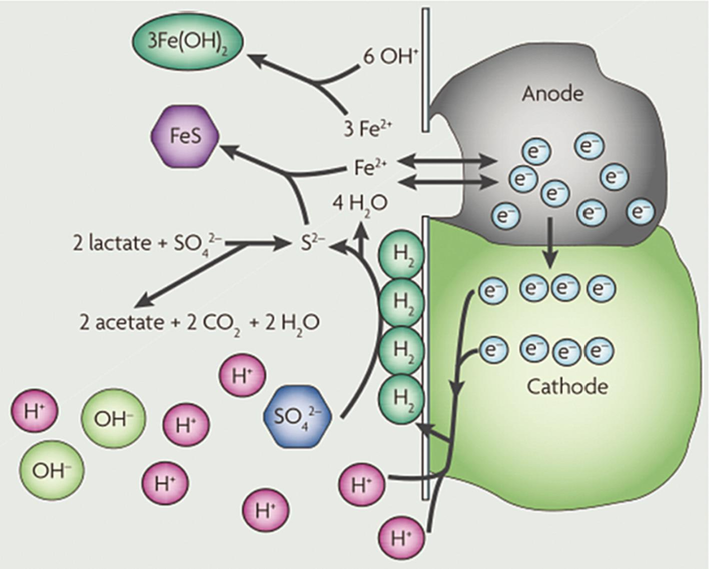
Mechanism diagram of cathodic depolarization of hydrogenase (Wei et al., 2021; Muyzer and Stams, 2008).
The role of SRB in this process is to remove hydrogen atoms from the metal surface, promoting the conversion of iron (Fe) into soluble ferrous ions (Fe2+), which then react with hydroxide ions (OH–) to generate secondary corrosion products, such as iron sulfide (FeS) and iron(II) hydroxide (Fe(OH)2) (Muyzer and Stams, 2008; Jia et al., 2019). The relevant reactions involved in the cathodic depolarization mechanism are as follows:
Anode reaction:
Cathodic reaction
Cathodic depolarization
Corrosion products
Corrosion products
The total reaction is:
The cathodic depolarization theory exhibits some limitations when applied to elucidate the mechanism of SRB corrosion of metals. Kip et al. (Kip and Veen, 2015) demonstrated that in SRB corrosion process, the addition of acetic acid as an electron donor for SRB led to competition between acetic acid and cathodic hydrogen, resulting in increased sulfide production from SRB and causing severe chemical corrosion. Nevertheless, no cathodic depolarization of metals or accelerated corrosion was observed when acetate was not added, and metals served as the sole electron donor for SRB. From the biological standpoint, it fails to capture microorganisms involvement in the MIC process. Surface-associated hydrogenases and presumably formate dehydrogenases are needed to mediate an apparent electron uptake and helped to remove the accumulation of [H], thus accelerating the corrosion. However, it cannot give the reasonable answer for the MIC caused by microorganisms without these enzymes (Tang et al., 2021). Consequently, the explanation based on the cathodic depolarization theory does not comprehensively account for the corrosion mechanism of SRB and exhibits certain limitations.
4.2 Corrosion mechanism of metabolites
4.2.1 H2S corrosion
It is generally believed that H2S corrosion is due to gaseous H2S dissolved in the aqueous phase and then dissociated to produce protons. Proton reduction hydrogen production is a cathodic reaction in the process of steel corrosion:
In addition, acidic H2S can also contribute to the cathodic process by a direct reduction on the steel surface (Zeng et al., 2014):
The SRB utilize hydrogenase to reduce H2S at the cathode of the metal surface, generating a metabolite that rapidly reacts with Fe to produce FeS. This FeS can act as either a cathodic or anodic reactant on the metal surface, thereby accelerating corrosion (Li et al., 2014). The overall corrosion reaction is thus written as:
Research has demonstrated that the proportion of dissolved H2S in water can influence the metal's corrosion rate (Dall’Agnol et al., 2014). Meanwhile, the produced H2 can lead to hydrogen penetration and cracking corrosion of the metal (Biezma, 2001; Raman et al., 2005).
4.2.2 Corrosion mechanism of FeS
The metabolism of SRB produces a dense or loose iron sulfide (FeS) film (Liu et al., 2022; Wei et al., 2021). Previous study has demonstrated (Lv et al., 2022) that the initial formation of the FeS film results in a tightly structured film that provides effective protection for the metal. However, when the sulfide concentration fluctuates from very low to very high, a more loosely structured film is formed. The FeS film exhibits electrical conductivity, enabling electron transfer from the metal surface and consequently accelerating metal corrosion. Zhang et al. (Zhang et al., 2023) found that the biofilms containing FeS accumulated in water-line and under water regions induced a galvanic effect, accelerating the corrosion rate (Fig. 3).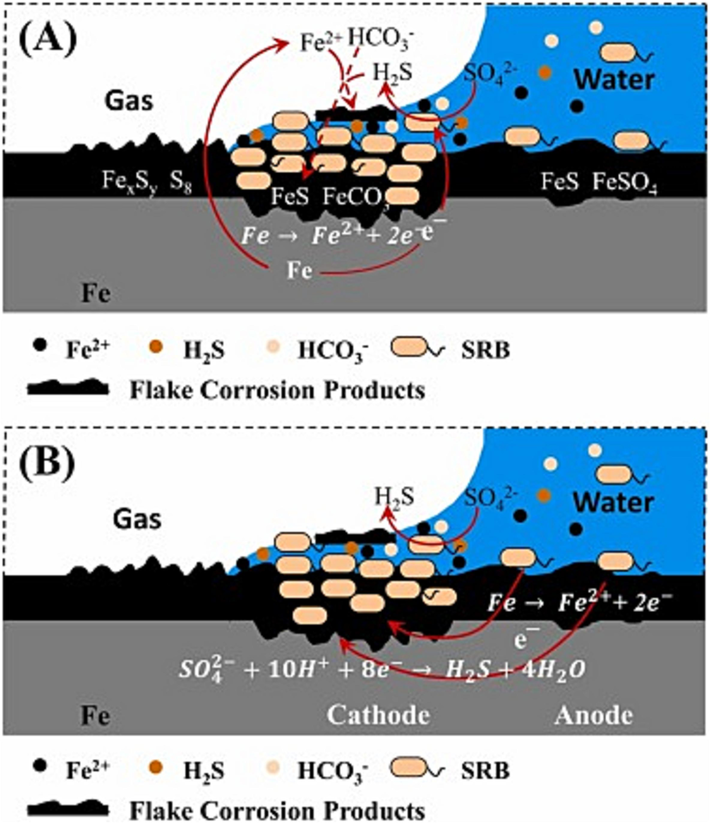
Schematic diagram of the process of water-line SRB corrosion: (A) in the water-line region; (B) in the liquid-phase region Zhang et al., 2023).
4.3 Concentration cell
Concentration cells primarily comprise oxygen concentration cells, metal ion concentration cells, and activated passivation cells, with oxygen concentration cells typically being the primary factor in localized corrosion of metallic materials. The uneven distribution of biofilms generated by microorganisms on metal surfaces leads to non-uniform oxygen diffusion, resulting in the formation of oxygen concentration gradients across different areas of the metal surface, creating the oxygen concentration cells. Research indicates that the electrode potential in oxygen-rich zones is more positive, acting as the metal cathode, while the electrode potential in oxygen-poor zones is more negative, serving as the metal anode. This potential difference prompts electron flow from the cathode to the anode, thereby causing corrosion (Abdolahi et al., 2014).
4.4 Mechanism of direct and indirect electron transfer
It has been demonstrated that SRB can directly acquire electrons from the metal surface under starvation, accelerating metal dissolution. Dinh et al. (Ht, 2004) isolated a specific SRB strain from the marine environment capable of directly utilizing electrons from the metal surface to reduce sulfate. This bypasses the need for chemically generated H2 as an electron source, and the classical theory of cathodic depolarization cannot explain the corrosion behavior of such bacteria. Instead, direct and indirect electron transfer mechanisms provide a comprehensive explanation for this phenomenon. The mechanisms for the direct uptake have been reported for some bacteria, including SRB, NRB, methanogenic archaea and acetogenic bacteria (Table 4).
Group
Strain name
Phylum/class
Family
SRB
Desulfopila corrodens (IS4)
Deltaproteobacteria
Desulfobulbaceae
Desulfovibrio ferrophilus (IS5)
Deltaproteobacteria
Desulfovibrionaceae
NRB
Bacillus licheniformis ATCC 14580
Firmicutes
Bacillaceae
Prolixibacter sp. MIC1-1
Bacteroidetes
Prolixibacteraceae
Methanogenic archaea
Methanobacterium sp. IM1
Euryarchaeota
Methanobacteriaceae
Methanococcus maripaludis KA1
Euryarchaeota
Methanococcaceae
Methanococcus maripaludis Mic1c10
Euryarchaeota
Methanococcaceae
Methanosarcina acetivorans
Euryarchaeota
Methanosarcina
Acetogenic bacteria
Sporomusa sp. GT1
Firmicutes
Veillonellaceae
As shown in Figs. 4 and 5, direct electron transfer encompasses two methods: (A) depending on contact between nanoconductors and the conductor surfacep; and (B) relying on direct contact between outer membrane-bound redox-active proteins, such as pigment C protein, and the substrate conductor surface. In 2005, Reguera et al. (Reguera et al., 2005) found that “geopili” (bacterial hair proteins) on the surface of the Geobacter sulfurreducens play a crucial role in electron transfer between bacteria and metals during contact. They introduced the concept of nanoconductors (pili), which facilitate electron transfer and enhance energy transfer efficiency between the bacterium and its environment. This finding elucidated the direct electron exchange process between microorganisms and electrode surfaces. Enning et al. (Enning et al., 2012) proposed the concept of electromicrobially influenced corrosion (EMIC), which considers the corresponding electron transfer mechanisms between metals and bacteria. Specifically, bacteria can utilize electrons from the metal surface for growth and metabolism, resulting in metal corrosion. This is primarily attributed to the role of cytochrome C proteins on the cell membrane surface and the electron transfer of biological nanoconductors (e.g., pili). Jia (Jia et al., 2017) designed the starvation experiment, in which the carbon source available was removed, and they found that SRB still greatly enhanced the steel corrosion. This also means that SRB can directly use Fe0 as electron donor to maintain the energy required for their growth. Similar enhanced corrosion under energy starvation has been reported for both methanogens and NRB.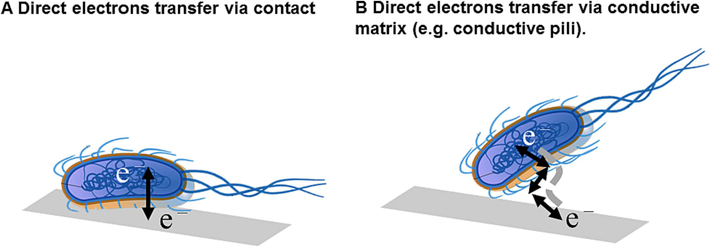
Schematic drawings of two direct electron transfer (Wei et al., 2022).
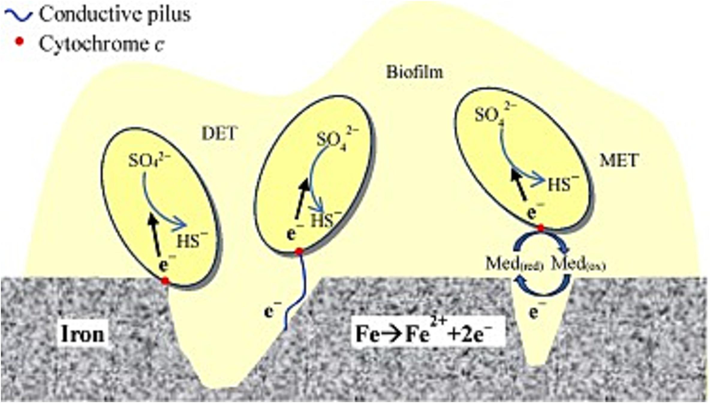
Schematic drawings of direct and indirect electron transfer in MIC by SRB (Li et al., 2018).
Indirect electron transfer involves the utilization of soluble electron carriers secreted by bacteria for growth and metabolism, primarily through cytochrome C proteins on the cell membrane surface to transfer electrons from the metal surface (Enning et al., 2012). Flavin adenine dinucleotide (FAD) and riboflavin are common electron transfer mediators. Recently, the research group lead by Jin (Jin et al., 2023). has been confirmed that microorganisms can directly obtain electrons from metal surfaces through conductive nanowires, leading to a new mechanism of metal material corrosion. This work can provide new targets for monitoring MIC in the future and designing new materials. Jia et al (Jia et al., 2017). found that riboflavin and FAD can increased the MIC rate and pitting of C1018 steel by Pseudomonas aeruginosa grown as a NRB biofilm. Currently, research on electron carrier transfer mechanisms remains in its infancy, and the mediator's role in the electron transfer process warrants further in-depth exploration and investigation.
The basic process of MIC in cooling water is systematically summarized and a “two-step corrosion model” is proposed, as shown in Fig. 6. In fact, the MIC occurred in the cooling water systerm is attributed to the coupling effect of abiotic and multi-biological factors, which also explains that the corrosion rate measured in the laboratory is far less than that observed in the field. The presence of calcium and magnesium ions in circulating water leads to the formation of a scale layer, which in turn forms a difference in oxygen concentration on the surface of the sample. When the oxygen content is high, oxygen absorption corrosion plays a dominant role, while when the oxygen is depleted or the content is very low, anaerobic microorganisms (SRB) will accelerate the corrosion process.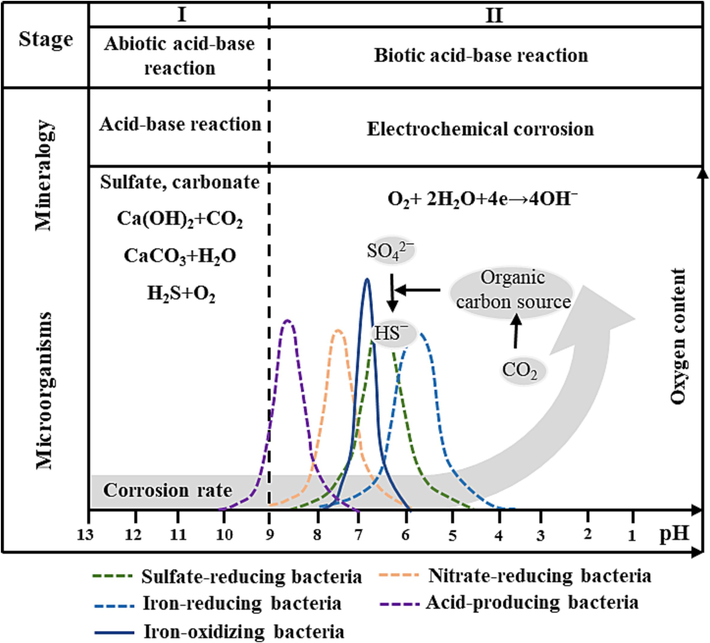
Two-step corrosion model in cooling water system.
5 The role of biofilm
The concept of “biofilm” was introduced in 1978, and MIC is closely associated with biofilm formation. Bacteria settle on metal surfaces and become immobilized or sessile cells. These sessile cells constitute the so-called biofilm, which promotes corrosion through various mechanisms. The formation of biofilms involves the following primary stages (Fig. 7):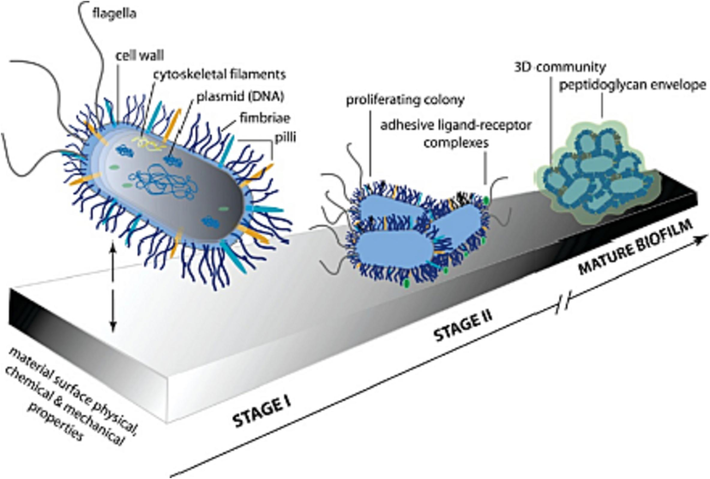
Schematic of bacterial cell structure and adhesion model that precedes organization of a mature biofilm (Lichter et al., 2009).
Initial adsorption: organic and inorganic ions from the environment are adsorbed onto the metal surface, forming an adsorption film. Microorganisms then attach to this film through electrostatic or van der Waals forces.
Biofilm formation: microbial cells proliferate on the material surface and secrete extracellular polymers, which gradually form a cohesive biofilm. These extracellular polymers primarily consist of polysaccharides, proteins, and lipids that firmly adhere microbial cells together, creating a stable microbial community.
Biofilm maturation: as the microbial community continues to develop, the biofilm becomes increasingly stable and mature. Microorganisms continuously secrete extracellular polymers, thickening the biofilm and creating complex microenvironments within it, thus providing optimal conditions for microbial growth and metabolism.
Biofilm disruption: when the microbial community within the biofilm reaches a certain density, the biofilm begins to degrade and disintegrate, releasing microorganisms and metabolites, which contribute to further corrosion and material failure.
It is important to note that even non-corrosive microorganisms can influence the corrosion process through biofilm formation. Concentration gradients, such as oxygen concentration cells or Fe2+ concentration cells, can form anodic and cathodic regions, leading to localized pitting corrosion. In some cases, biofilms can act as diffusion barriers, resulting in reduced corrosion. Pérez et al. (Pérez et al., 2007) demonstrated that desulfurizing Vibrio biofilms initially inhibit steel MIC. Biofilms in natural environments typically consist of multiple bacterial species, and their interactions may cause more severe MIC than single species typically studied under laboratory conditions. Therefore, for MIC studies in circulating water systems, it is essential to investigate the community structure of biofilms and environmental conditions.
Additionally, due to the complexity of actual operating conditions in circulating water pipelines, water flow generates shear stresses in the biofilm, potentially reducing the biofilm's mechanical properties and leading to its detachment. This process can facilitate the formation of electrochemical cells, such as primary cells, and promote localized corrosion. Consequently, understanding the interaction between flow rate and biofilm is crucial for MIC studies in circulating water systems.
6 Research methods of MIC
In the last 30 years, there has been rapid development in materials science, biological science, and instrumental analysis (Jia et al., 2019; Xie et al., 2018; Xie et al., 2019). Researchers have used various tools to study the changes in materials and microorganisms during MIC, as listed in Table 5. Dong and Khan et al. (Khan et al., 2019; Dong et al., 2020) recently reviewed MIC research techniques and recommended them to readers.
Tools
Methods
Function
Biology assays
Most probable number method (MPN)
Cell count
Plate count
Hemocytometer counting
Fluorescent dyes
Bacterial enumeration
Flow cytometry
Physiologic states
16 s RNA
Microbial communities
Chemical tool
Gas chromatography
Concentration measurement
Time of flight secondary ion mass spectrometry (TOF-SIMS)
Bacterial contribution
Image
Scanning electron microscope (SEM)
Biofilm morphology
Transmission electron microscope (TEM)
Biofilm morphology
Focused ion beam (FIB)
FIB milling
Spectral
X-ray diffraction (XRD)
Component
X-ray photoelectron spectroscopy (XPS)
Component
Confocal laser scanning microscope (CLSM)
Live/dead cell characterization
Inductive coupled plasma emission spectrometer (ICP)
Ion content
Corrosion techniques
Weight loss
Corrosion rate
Linear polarization (LP)
Linear polarization resistance
Electrochemical impedance spectroscopy (EIS)
Biofilm capacitance
Electrochemical noise (EN)
Pit susceptibility
Cyclic voltammetry (CV)
Corrosion process
Potentiodynamic polarization
Corrosion reaction kinetics
6.1 Wire beam electrode (WBE)
The wire beam electrode technology, also known as array electrode technology, lies between conventional electrochemical technology and micro-area electrochemical technology. Comprising multiple metal wires with diameters ranging from several to tens of microns, the wire beam electrode can be made of the same material or a combination of different materials. Common devices of this technology include cylindrical, fiber, grid, and other forms of wire beam electrodes. By bundling these wires, a densely arranged electrode array is formed, and the end of each wire bundle is isolated to ensure independent operation between the electrodes. When a potential is applied to the test system, each tiny electrode on the wire beam electrode produces a non-interfering current response, resulting in an electrochemical signal with higher resolution and improved sensitivity and accuracy. When microorganisms come into contact with the metal surface, they release metabolites such as electrons, ions, gases, and organic acids, which alter the potential in the surrounding environment. The wire beam electrode can detect these potential changes to infer the degree and type of microbial corrosion. A zero-resistance galvanometer can measure the two-dimensional current distribution on the surface point by point, and the localized corrosion factor LF of the metal (Dong et al., 2011), can be calculated:
、
represent the coupling current values of individual electrodes in the anode or cathode regions at a specific moment, while Nc and Na denote the number of single electrodes belonging to the cathode and anode regions on the surface at a given time, respectively. By analyzing the trends in LF, valuable insights into the metal surface corrosion process can be obtained. Naing (Naing Aung et al., 2006) initially employed wire beam electrode technology to investigate the variations in cathode and anode on the wire beam electrode surface under distinct corrosive media conditions, thereby determining the impact of various corrosive media on steel corrosion mechanisms. Dong et al. (Dong et al., 2011) studied the heterogeneous corrosion of mild steel under SRB biofilm by WBE technique. They found the galvanic current maps can give a good indication of localized corrosion by SRB (Fig. 8). Building upon this foundation, the research team further examined carbon steel corrosion in diverse environments, such as dry, wet, and chloride salt conditions, and incorporated rust inhibitors in their experiments. Through a thorough analysis and evaluation of the wire beam electrode test results, the team demonstrated the high accuracy of this testing method. This evaluation technique can effectively assess the efficacy of materials and processing methods in preventing metal corrosion, holding significant experimental importance.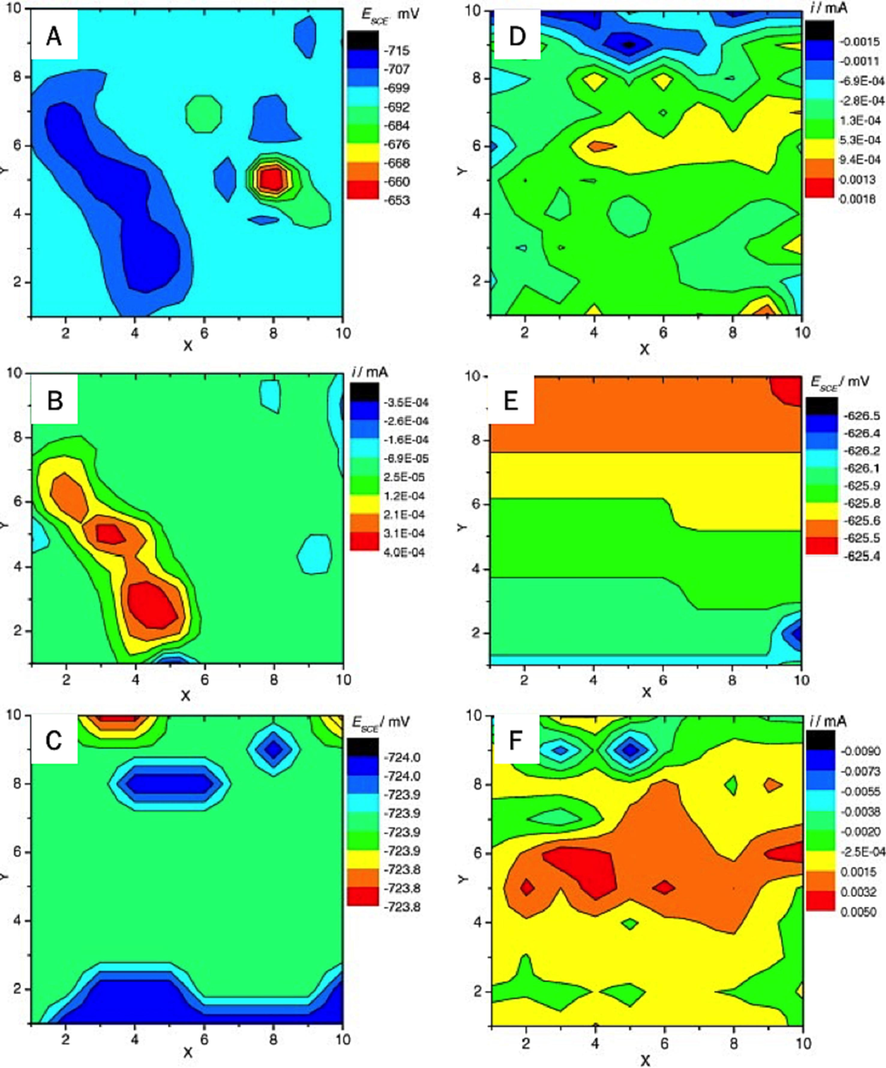
Potential and current maps of the WBE after exposed to SRB-incubated medium for (A and B) 2 h, (C and D) 7 days and (E and F) 15 days (Dong et al., 2011).
The wire beam electrode technology, also known as array electrode technology, has the capability to provide information on the change of electrochemical parameters at adjacent points on the electrode surface. This includes not only the average signal provided by a single electrode but also a series of important information, such as surface distribution and difference size. By scanning the microprobe, differences in electrochemical characteristics in different regions of the electrode surface can be distinguished, enabling researchers to obtain more accurate electrochemical information and making up for defects in traditional electrochemical research. Furthermore, the structural parameters of the wire beam electrode, such as wire beam spacing, number, and diameter, have no impact on the study of paint coating inhomogeneity. The wire beam electrode technology is a bottom-based test technology, meaning that the requirement for electrode surface flatness is not high, and it is not affected by the thickness of the electrode surface coating. Additionally, its fast scanning speed facilitates the study of material corrosion degree evolution with time, and data synchronization is high, enhancing its research value. However, it is important to note that the wire beam electrode is composed of a large number of unit electrodes, and the failure of one electrode may lead to the failure of the entire wire beam electrode measurement. As such, the wire beam electrode technology is currently limited to corrosion monitoring in the laboratory, and its promotion requires further study.
6.2 Electrochemical noise (EN)
Electrochemical noise (EN) refers to the random non-equilibrium fluctuation of state parameters (potential, current) in electrochemical kinetics. During the electrode reaction process, the local activity of the electrode surface, the formation and destruction of the passivation film, the generation of bubbles on the electrode surface, etc., these in-situ information related to the electrode reaction can be reflected in the electrochemical noise. According to the type of measurement signal, electrochemical noise can be divided into potential noise and current noise. The noise resistance can be obtained by calculating the ratio of the standard deviation of the two. Studies have shown that there is consistency between noise resistance and linear polarization resistance. Electrochemical noise technology includes time domain and frequency domain analysis methods. It provides material corrosion information by measuring the potential and current fluctuations generated by spontaneous corrosion reactions. It has great application prospects in effectively distinguishing uniform corrosion from localized corrosion and monitoring and research of localized corrosion behaviors (such as pitting, pitting, etc.) of equipment (Obot et al., 2019). It has little interference on the surface state of the research object, and can achieve continuous detection and rapid detection.
6.3 Electrochemical impedance spectroscopy (EIS)
Electrochemical Impedance Spectroscopy is a high-resolution and non-invasive electrochemical test method. It operates based on the interaction between AC electrical signals in the circuit and the electrochemical reaction of the system. In the MIC monitoring, EIS technology can be used to examine the interaction between the metal surface and biofilm formed by microbial growth, as well as the effect of microorganisms on the metal surface. EIS technology applies an external small AC electrical signal to stimulate the electrochemical reaction of the system. The degree, type, and location of microbial corrosion are evaluated by measuring the impedance spectrum of the system.
Electrode surfaces covered in microbial growth will cause potential changes on the electrode surface due to the metabolites produced by microorganisms. This causes changes in charge transfer and ion transfer, which affects the conductivity of ions in the electrolyte. EIS technology applies a small sine wave signal to the system, measures the impedance response of the system, and evaluates the microbial corrosion according to the change of impedance value and phase angle (Qu et al., 2015; Li et al., 2017). The impedance spectrum's lower frequency indicates greater impedance, which may indicate the formation and thickness of the microbial membrane, leading to blocked ion transport.
EIS technology is an accurate, sensitive, and quantitative high-resolution, non-invasive electrochemical testing method in microbial corrosion monitoring. It can continuously monitor the development process of microbial corrosion, detect problems in time, and take corresponding prevention and control measures to ensure the safe operation of industrial equipment. However, this technology has limitations. The analysis of complex electrochemical impedance spectroscopy can be difficult, affecting the repeatability of experimental results. In the future, computer technology's development will improve the EIS technology data processing capabilities, and it is expected to be promoted in more fields.
Although there are a large number of publications on MIC, most laboratory experiments begin with the following designed test procedures:
-
●
Microbial methods are used to culture, count and quantify bacteria in solution.
-
●
In the presence of bacteria, the corrosion rate and reaction kinetics were monitored by weight loss and electrochemical techniques.
-
●
To characterize the morphology and composition of the biofilm after short-term inoculation.
-
●
Study the changes in the physical and chemical environment of the solution (such as H2S and pH) by chemical analysis.
-
●
Finally, bacteria were associated with MIC.
It should be noted that the results of the report are changes in the steel corrosion process with the participation of bacteria, but the role of bacteria in corrosion is not involved. In fact, only electrochemical monitoring can reveal the real-time corrosion reaction and dynamic change of pipeline steel in the presence of bacteria. Therefore, the following important questions have not been answered in the MIC study of circulating water system:
-
●
How does the bacterial community under the biofilm formed on the surface of the pipeline interact with the environment?
-
●
What electrochemical conditions change at the metal-biofilm / solution interface when bacteria precipitate on a living biofilm?
-
●
How does biofilm structure and mechanical property affect the corrosion of pipeline steel?
Traditional techniques cannot provide sufficient information on the various activities of bacteria at the metal/electrolyte interface. MIC is a local phenomenon, and most publications use electrochemical techniques such as EIS and LPR, which reflect the average signal on the surface area of the exposed sample. The electrochemical technique is applied to the system to evaluate the MIC that requires an external signal, which may cause the surface of the mount to be non-invertible.
7 Protective measures against MIC
MIC control is a challenge in industries. For controlling internal MIC of cooling water systerm, traditional physical methods such as pigging and ultraviolet (UV) irradiation are difficult to apply and could not achieve better results. At present, the common methods for MIC of metal materials are mainly chemical method, physical method, biological control and protective coating (Popoola et al., 2013).
7.1 Chemical method
The Chemical method is commonly used to prevent MIC. These methods control the growth of microorganisms by using chemical reagents such as biocides and corrosion inhibitors. The most commonly used chemical fungicides are biocides and non-oxidized (Rasheed et al., 2019; Fang et al., 2021), as shown in Table 6. However, the environment in practical application poses a greater challenge to the performance of biocides. From a microbial point of view, the sessile cells underneath a biofilm are far more difficult to eradicate than planktonic cells suspended in the liquid. Therefore, Xu et al. (Xu et al., 2012) suggested that a biocide stress is required for recalcitrant industrial biofilms. They found D-amino acids can be used as biocide enhancers for THPS. In addition, biofilms are likely to develop resistance to biocides, making it failed for a long-term use (Vance et al., 2005).
Fungicides
Characteristic
Dosage/ (mg.L-1)
Oxidizing type
Cl2
Effective in killing bacteria and algae, pH-dependent
0.1 ∼ 0.2
ClO2
Effective in killing bacteria, fungi and seaweed
0.1 ∼ 1.0
O3
Effective in killing bacteria and algae, pH-dependent
0.2 ∼ 0.5
Non-oxidizing
Glutaraldehyde
Effective in killing bacteria, fungi and algae, pH broad spectrum
10 ∼ 70
Isothiazolinone
Effective in killing bacteria and seaweed
0.9 ∼ 10
Quaternary ammonium salt
Effective in killing bacteria and algae, surfactant
8 ∼ 35
Tetrakis (hydroxymethyl) phosphonium sulfate
Effectively kills bacteria, fungi and seaweed
7.2 Physical method
The physical method is mainly through some physical methods such as the use of static magnetic field, ultrasonic treatment, ultraviolet irradiation and pigging method, etc. The pigging method is currently a more advanced anti-corrosion technology, with good application and promotion prospects. Pigging method is a method of removing sediment, impurities and corrosion products in the pipeline to achieve the purpose of corrosion protection. In the industrial field, the pipeline system is a key component of transporting liquid and gas. In order to ensure the normal operation of the pipeline and prolong the service life, corrosion protection is very important. The application of pigging method in corrosion protection mainly includes the following aspects:
Remove deposits and impurities in pipelines: Deposits and impurities in pipelines can cause localized corrosion, microbial corrosion and stress corrosion. Through pigging, these substances can be effectively removed and the corrosion risk of pipelines can be reduced.
Removal of corrosion products in the pipeline: Corrosion products in the pipeline will accelerate the occurrence and expansion of corrosion. Pigging method can remove these corrosion products, thereby slowing the process of corrosion.
Restore the coating of the pipeline: The coating of the pipeline may be damaged due to long-term use. The pigging method can remove the dirt and impurities on the surface of the coating and restore the integrity of the coating.
Improve the operation efficiency of the pipeline: the pigging method can reduce the resistance in the pipeline and improve the operation efficiency of the pipeline. This can not only reduce energy consumption, but also reduce the corrosion risk caused by high-speed flow of pipelines.
The principle of pigging method mainly includes the following:
-
●
Mechanical pigging: Direct cleaning of the pipeline through tools such as scrapers and brushes. This method is suitable for removing large amounts of sediment and impurities.
-
●
Chemical pigging: By adding chemical reagents, such as cleaning agents, corrosion inhibitors, etc., the corrosion products in the pipeline are dissolved, dispersed and stabilized. This method is suitable for removing corrosion products that are difficult to remove by mechanical pigging.
-
●
Pneumatic pigging: Through high-pressure gas, such as air, nitrogen, etc., will be the deposit and impurities in the pipe blown out. This method is suitable for removing lighter sediments and impurities.
-
●
Hydraulic pigging: Through high-pressure water or other liquids, the sediment and impurities in the pipeline out. This method is suitable for removal of heavy sediment and impurities.
7.3 Biological method
Biocontrol is mainly through competition, antagonism and symbiosis with other kinds of microorganisms to prevent microorganisms. Some scholars also think that the use of low-concentration EPS inhibition mechanism (Lv et al., 2023), but also a future research ideas, low-concentration EPS on the surface of steel after the formation of a dense passivation layer, this prevents cathodic depolarizing agents such as oxygen from reaching the metal surface, thus preventing electron transfer.
In addition, studies have shown that NRB can be used as a potential biological inhibitor to control SRB-induced MIC in circulating water pipelines, and researchers use NRB to reduce MIC through competitive effects (Javaherdashti and Alasvand, 2019). In the co-culture of SRB and NRB, the energy obtained by nitrate reduction was greater than that obtained by sulfate reduction (Voordouw et al., 2009). Therefore, the electron preferred transfer for SRB sulfate reduction is used for NRB nitrate reduction. In addition, NO3-can be reduced to NO2-by denitrification of NRB, which increases the redox potential of the environment and hinders SRB-driven sulfate reduction.
7.4 Coating protection
The protective coating method is mainly to reduce the roughness of the metal surface by coating a hydrophobic coating on the metal surface, thereby preventing the adhesion of microorganisms. Anti-bacterial coatings can also be applied to the metal surface to kill corrosive microorganisms.
7.5 Antibacterial steel and alloy
To protect against microbial corrosion, numerous researchers have introduced innovative ideas, laying the groundwork for future efficient and cost-effective corrosion control methods. Some experts argue that incorporating copper (Cu) alloy elements into metals can effectively prevent microbial corrosion. Nan et al. (Nan et al., 2012) conducted a preliminary study on the antibacterial behavior of a newly designed Cu-containing 200 stainless steel. The results show that the Cu-rich precipitation caused by saturated Cu in the steel matrix is not only an important factor for its excellent antibacterial properties, but also an important reason for its corrosion resistance comparable to that of commercial 200 stainless steel. This caused bacterial cell wall membranes to rupture, thereby eliminating the bacteria. Similarly, Yang and colleagues (Yang et al., 2020; Shi et al., 2017) introduced 1.0 % Cu alloy elements to pipeline steel and treated it at 500 °C for one hour, resulting in the precipitation of a Cu-rich phase. This caused bacterial cell wall membranes to rupture, thereby eliminating the bacteria.
Overall, it is anticipated that these provide a reliable and convenient technical pathway for the MIC control of pipelines in practice. Understanding the timing of biofilm formation and when they become aggressive in practice may be essential in the MIC control. Therefore, corrosion monitoring necessitates the creation of sensors capable of distinguishing between biological and abiotic corrosion, which involves developing new biofilm-monitoring sensors in the future.
8 Conclusion and outlook
The problem of the MIC has become increasingly prominent in the cooling water systerm, which has caused very serious economic losses, and the research on MIC mechanism and protective measures has become increasingly important. At present, our understanding of microbial corrosion is still relatively limited. The existing microbial corrosion mechanism is not comprehensive and clear. Many corrosion phenomena cannot be fully and accurately described based on existing theories. Therefore, it is necessary to integrate the collaborative research of various corrosion mechanisms and continue to explore the unknown MIC mechanism. Continuing to explore and improve the corrosion mechanism of microorganisms can provide guidance and help for MIC protection in the circulating cooling water system of thermal power plants in the future. Based on the data collected on site, the test results are presented more realistically. Various factors of natural gas pipeline MIC should be considered in mechanism, modeling and control. In-depth understanding of the electron transfer process between Fe0 and bacteria can promote the development of MIC mechanism under the action of microbial mixed bacteria through molecular biological methods, and can further promote derivative applications, such as the development of bioelectrochemical technology. The development of models to describe or predict the MIC of circulating water systems is attractive in the future. The effective model of MIC involves not only the chemical and electrochemical phenomena at the metal-biofilm interface and under the biofilm, but also the conditions of the service environment, such as the chemical and physical conditions collected on site. The dynamics of chemical, electrochemical and biological processes at the metal / biofilm and solution interfaces support a range of phenomena from adsorption and production growth to cell electron transduction. Because many interfaces have nanoscale structures that control these phenomena. The new trend of MIC testing in the future should focus on the measurement of bioelectrochemical flux at the nanoscale. In addition, modern computing technologies such as machine learning, deep learning, and simulation provide promising alternatives for the diagnosis and modeling of MIC processes. The development of computer-based simulation models may be the best hope for understanding and controlling MICs in laboratory research and practice. Therefore, through the interdisciplinary integration of multiple disciplines, combined with today 's cutting-edge research techniques, in-depth exploration of MIC can help us better understand and control the MIC existing in the circulating water cooling system of thermal power plants.
In addition, the study of microbial corrosion mechanism should be combined with multi-field and multi-disciplinary research. The interpretation of various corrosion phenomena does not need to be completely constrained by the existing corrosion mechanism. It is necessary to put forward new research ideas and methods in combination with the current known mechanism, boldly put forward new assumptions, and continue to expand the understanding of the occurrence and inhibition of microbial corrosion on the premise of grasping microbial corrosion as a whole. MIC is a problem that industrial corrosion department and HSE deal with together. The best mitigation strategy for circulating water system MIC is to integrate the MIC management plan in the overall management system of the organization to improve the understanding of system safety and reliability over time. At the same time, there is an urgent need to develop guidelines or standards for laboratory and field MIC testing.
Acknowledgement
This work was supported by the National Natural Science Foundation of China (No. 52261022).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Microbially influenced corrosion of steels by Pseudomonas aeruginosa. Corros. Rev.. 2014;32:129-141.
- [Google Scholar]
- über Spirillum desulfuricans als Ursache von Sulfatreduktion. Zentralbl Bakteriol.. 1895;49–59
- [Google Scholar]
- The role of hydrogen in microbiologically influenced corrosion and stress corrosion cracking. Int. J. Hydrog. Energy.. 2001;26:515-520.
- [Google Scholar]
- Dall’Agnol, L.T., Cordas, C.M., Moura, J.J.G. 2014. Influence of respiratory substrate in carbon steel corrosion by a Sulphate Reducing Prokaryote model organism, Bioelectrochemistry. 97, 43–51.
- Severe microbiologically influenced corrosion of S32654 super austenitic stainless steel by acid producing bacterium Acidithiobacillus caldus SM-1. Bioelectrochemistry.. 2018;123:34-44.
- [Google Scholar]
- Microbiologically influenced corrosion of 304L stainless steel caused by an alga associated bacterium Halomonas titanicae. J. Mater. Sci. Technol.. 2020;37:200-206.
- [Google Scholar]
- Heterogeneous corrosion of mild steel under SRB-biofilm characterised by electrochemical mapping technique. Corros. Sci.. 2011;53:2978-2987.
- [Google Scholar]
- Iron-Oxidizing Bacteria: An Environmental and Genomic Perspective. Annu. Rev. Microbiol.. 2010;64:561-583.
- [Google Scholar]
- Marine sulfate-reducing bacteria cause serious corrosion of iron under electroconductive biogenic mineral crust. Environ. Microbiol.. 2012;14:1772-1787.
- [Google Scholar]
- Abiotic Process for Fe(II) Oxidation and Green Rust Mineralization Driven by a Heterotrophic Nitrate Reducing Bacteria (Klebsiella mobilis) Environ. Sci. Technol.. 2014;48:3742-3751.
- [Google Scholar]
- Preparation and Performance Evaluation of a Novel Bactericide for Sulfate Reducing Bacteria. Mater. Sci. Forum.. 2021;1035:624-629.
- [Google Scholar]
- Role of iron-reducing bacteria in corrosion and protection of carbon steel. Int. Biodeterior. Biodegrad.. 2009;63:891-895.
- [Google Scholar]
- A Membrane-Bound Cytochrome Enables Methanosarcina Acetivorans to Conserve Energy from Extracellular Electron Transfer. mBio.. 2019;10:e00789-e00819.
- [Google Scholar]
- D. Ht, K. J, M. M, H. Aw, S. M, W. F, Iron corrosion by novel anaerobic microorganisms, Nature. 427 (2004).
- Iron Corrosion Induced by Nonhydrogenotrophic Nitrate-Reducing Prolixibacter sp. Strain MIC1-1. Appl. Environ. Microbiol.. 2015;81:1839-1846.
- [Google Scholar]
- Isolation of a sulfide-producing bacterial consortium from cooling-tower water: Evaluation of corrosive effects on galvanized steel. Anaerobe.. 2017;43:27-34.
- [Google Scholar]
- An Introduction to Microbial Corrosion - ScienceDirect. Biol. Treat. Microb. Corros. 2019:25-70.
- [Google Scholar]
- Jia, Ru, Tan, Jie, Long, Jin, Peng, Blackwood, Daniel, John, Effects of biogenic H2S on the microbiologically influenced corrosion of C1018 carbon steel by sulfate reducing Desulfovibrio vulgaris biofilm, Corros. Sci. J. Environ. Degrad. Mater. Its Control. (2018).
- Microbiologically influenced corrosion of C1018 carbon steel by nitrate reducing Pseudomonas aeruginosa biofilm under organic carbon starvation. Corros. Sci.. 2017;127:1-9.
- [Google Scholar]
- Electron transfer mediators accelerated the microbiologically influence corrosion against carbon steel by nitrate reducing Pseudomonas aeruginosa biofilm. Bioelectrochemistry.. 2017;118:38-46.
- [Google Scholar]
- Effects of ferrous ion concentration on microbiologically influenced corrosion of carbon steel by sulfate reducing bacterium Desulfovibrio vulgaris. Corros. Sci.. 2019;153:127-137.
- [Google Scholar]
- Microbiologically influenced corrosion and current mitigation strategies: A state of the art review. Int. Biodeterior. Biodegrad.. 2019;137:42-58.
- [Google Scholar]
- Accelerated microbial corrosion by magnetite and electrically conductive pili through direct Fe0-to-microbe electron transfer. Angew. Chem. Int. Ed.. 2023;62:e202309005.
- [Google Scholar]
- Isolation of Acetogenic Bacteria That Induce Biocorrosion by Utilizing Metallic Iron as the Sole Electron Donor. Appl. Environ. Microbiol.. 2015;81:67-73.
- [Google Scholar]
- Corrosion Inhibition of X80 Steel in Simulated Marine Environment with Marinobacter aquaeolei. Acta Metall. Sin. Lett.. 2019;32:1373-1384.
- [Google Scholar]
- Anaerobic microbiologically influenced corrosion mechanisms interpreted using bioenergetics and bioelectrochemistry: A review. J. Mater. Sci. Technol.. 2018;34:1713-1718.
- [Google Scholar]
- Microbiologically influenced corrosion behavior of S32654 super austenitic stainless steel in the presence of marine Pseudomonas aeruginosa biofilm. J. Mater. Sci. Technol.. 2017;33:1596-1603.
- [Google Scholar]
- Effect of H_2S concentration on the corrosion behavior of pipeline steel under the coexistence of H_2S and CO_2. Int. J. Miner. Metall. Mater.. 2014;21:388-394.
- [Google Scholar]
- Design of Antibacterial Surfaces and Interfaces: Polyelectrolyte Multilayers as a Multifunctional Platform. Macromolecules.. 2009;42:8573-8586.
- [Google Scholar]
- Mchanistic investigations of corrosion and localized corrosion of X80 steel in seawater comprising sulfate-reducing bacteria under continuous carbon starvation. Corrosion. Communications. 2022;8:70-80.
- [Google Scholar]
- Study of biofilm-influenced corrosion on X80 pipeline steel by a nitrate-reducing bacterium, Bacillus cereus, in artificial Beijing soil. Colloids Surf. B Biointerfaces.. 2021;197:111356
- [Google Scholar]
- Accelerated performance recovery of anaerobic granular sludge after temperature shock: Rapid construction of protective barriers (EPS) to optimize microbial community composition base on quorum sensing. J. Clean. Prod.. 2023;392:136243
- [Google Scholar]
- Corrosion of X65 steel by Pseudomonas sp. in planktonic and sessile states and inhibition effect of cathodic polarization. Corrosion. Communications. 2022;8:58-69.
- [Google Scholar]
- Iron corrosion activity of anaerobic hydrogen-consuming microorganisms isolated from oil facilities. J. Biosci. Bioeng.. 2010;110:426-430.
- [Google Scholar]
- The ecology and biotechnology of sulphate-reducing bacteria. Nat. Rev. Microbiol.. 2008;6:441-454.
- [Google Scholar]
- Novel corrosion experiments using the wire beam electrode: (II) Monitoring the effects of ions transportation on electrochemical corrosion processes. Corros. Sci.. 2006;48:39-52.
- [Google Scholar]
- Antibacterial Behavior of a Cu-bearing Type 200 Stainless Steel. J. Mater. Sci. Technol.. 2012;28:1067-1070.
- [Google Scholar]
- Electrochemical noise (EN) technique: review of recent practical applications to corrosion electrochemistry research. J. Adhes. Sci. Technol.. 2019;1–44
- [Google Scholar]
- Influence of Desulfovibrio sp. biofilm on SAE 1018 carbon steel corrosion in synthetic marine medium. Corros. Sci.. 2007;49:3580-3597.
- [Google Scholar]
- Corrosion problems during oil and gas production and its mitigation. Int. J. Ind. Chem.. 2013;4:35.
- [Google Scholar]
- Corrosion behavior of cold rolled steel in artificial seawater in the presence of Bacillus subtilis C2. Corros. Sci.. 2015;91:321-329.
- [Google Scholar]
- Hydrogen embrittlement of a low carbon steel during slow strain testing in chloride solutions containing sulphate reducing bacteria. Mater. Sci. Technol.. 2005;21:1094-1098.
- [Google Scholar]
- Recent advancements of nanomaterials as coatings and biocides for the inhibition of sulfate reducing bacteria induced corrosion. Curr. Opin. Chem. Eng.. 2019;25:35-42.
- [Google Scholar]
- Extracellular electron transfer via microbial nanowires. Nature.. 2005;435:1098-1101.
- [Google Scholar]
- Study on Microbiologically Influenced Corrosion Behavior of Novel Cu-Bearing Pipeline Steels. Acta Metall. Sin.. 2017;53:153-162.
- [Google Scholar]
- Electrochemical behaviour of stainless steels in media containing iron-oxidizing bacteria (IOB) by corrosion process modeling. Corros. Sci.. 2008;50:540-547.
- [Google Scholar]
- Characterization of Sulfate Reducing Bacteria Isolated from Cooling Towers. Environ. Monit. Assess.. 2005;104:211-219.
- [Google Scholar]
- Tang, H.Y., Yang, C., Ueki, T., Pittman, C.C., Xu, D., Woodard, T.L., Holmes, D.E., Gu, T., Wang, F., Lovley, D.R. Stainless steel corrosion via direct iron-to-microbe electron transfer by Geobacter species. ISME J. 15(10):3084-3093.
- Iron-Corroding Methanogen Isolated from a Crude-Oil Storage Tank. Appl. Environ. Microbiol.. 2010;76:1783-1788.
- [Google Scholar]
- Vance, I., Thrasher, D.R. 2005. Reservoir Souring: Mechanisms and Prevention, in: Pet. Microbiol., John Wiley & Sons, Ltd, pp. 123–142.
- Complementary Microorganisms in Highly Corrosive Biofilms from an Offshore Oil Production Facility. Appl. Environ. Microbiol.. 2016;82:2545-2554.
- [Google Scholar]
- Sulfide remediation by pulsed injection of nitrate into a low temperature Canadian heavy oil reservoir. Environ. Sci. Technol.. 2009;43:9512-9518.
- [Google Scholar]
- Effect of sulfate-reducing bacteria on corrosion of X80 pipeline steel under disbondecoating in a red soil solution. J. Mater. Sci. Technol.. 2021;87:1-17.
- [Google Scholar]
- Effect of uniaxial elastic stress on corrosion of X80 pipeline steel in an acidic soil solution containing sulfate-reducing bacteria trapped under disbonded coating. Corros Sci. 2021;193:109893
- [Google Scholar]
- Internal microbiologically influenced corrosion of natural gas pipelines: A critical review. J. Nat. Gas Sci. Eng.. 2022;102:104581
- [Google Scholar]
- Synergistic effect of sulphate-reducing bacteria and external tensile stress on the corrosion behaviour of X80 pipeline steel in neutral soil environment. Eng. Fail. Anal.. 2018;91:382-396.
- [Google Scholar]
- Effect of strain rate and sulfate reducing bacteria on stress corrosion cracking behaviour of X70 pipeline steel in simulated sea mud solution. Eng. Fail. Anal.. 2019;100:245-258.
- [Google Scholar]
- A synergistic d-tyrosine and tetrakis hydroxymethyl phosphonium sulfate biocide combination for the mitigation of an SRB biofilm. World J. Microbiol. Biotechnol.. 2012;28:3067-3074.
- [Google Scholar]
- Laboratory investigation of microbiologically influenced corrosion of C1018 carbon steel by nitrate reducing bacterium Bacillus licheniformis. Corros. Sci.. 2013;77:385-390.
- [Google Scholar]
- Mechanistic modeling of biocorrosion caused by biofilms of sulfate reducing bacteria and acid producing bacteria. Bioelectrochemistry.. 2016;110:52-58.
- [Google Scholar]
- Novel Cu-Bearing Pipeline Steels: A New Strategy to Improve Resistance to Microbiologically Influenced Corrosion for Pipeline Steels. Acta Metall. Sin.. 2020;56:385-399.
- [Google Scholar]
- Accelerated anaerobic corrosion of electroactive sulfate-reducing bacteria by electrochemical impedance spectroscopy and chronoamperometry. Electrochem. Commun.. 2013;26:101-104.
- [Google Scholar]
- Temperature Responses of Ammonia-Oxidizing Prokaryotes in Freshwater Sediment Microcosms. PLoS ONE.. 2014;9:e100653.
- [Google Scholar]
- Mechanism of microbiologically induced anaerobic water-line corrosion of 980 high strength steel in nutrient-rich artificial seawater. Corros. Sci.. 2023;220:111268
- [Google Scholar]
- Effect of Chloride Ion Concentration on Corrosion of Q235 Steel in Sulfate-Reducing Bacteria Containing Solution. Int. J. Electrochem. Sci.. 2019;14:875-885.
- [Google Scholar]
- Direct microbial electron uptake as a mechanism for stainless steel corrosion in aerobic environments. Water Research. 2022;219:118553
- [Google Scholar]
- How sulphate-reducing micro- organisms cope with stress: lessons from systems biology. Nat. Rev. Microbiol.. 2011;9:452.
- [Google Scholar]







