Translate this page into:
Neuroprotective effects of tanshinone IIA in experimental model of Parkinson disease in rats
⁎Corresponding author at: NO. 19, Xiuhua Road, Xiuying District, Haikou, Hainan Province, China. l2381972@yahoo.com (Qiangzi Li)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Neurodegenerative diseases including Parkinson's disease (PD) cause many problems for the patient. Treatment options include the administration of chemical drugs, which are associated with many side effects. Therefore, the current study was aimed to investigate tanshinone IIA effects on the experimental model of PD in rats and to study the neuroprotective mechanisms of this natural product. After preparation of rats and induction of PD by 6-hydroxypopamine (6-OHDA), different concentrations of tanshinone IIA were administered to the rats for 10 days. Apomorphine-induced rotation and muscle rigidity behavioral tests were used to evaluate PD induction and to study the effects of tanshinone IIA. After separation of the hippocampus, biochemical parameters such as lipid peroxidation, protein oxidation, succinic dehydrogenase (SDH), super oxide dismutase (SOD), reactive oxygen species (ROS), malate dehydrogenase (MDH), catalase (CAT), reduced glutathione (GSH), lactate dehydrogenase (LDH), IL-1β, TNF-α, INF-γ, IL-10 cytokines and NF-ĸB protein expression were measured. The results showed that PD rats receiving tanshinone IIA improved their performance in behavioral tests. Decreased oxidative stress indices and increased antioxidant capacity were observed in the hippocampal tissue of PD rats receiving tanshinone IIA. Also, administration of tanshinone IIA to PD rats downregulated inflammatory cytokines TNF-α. IL-1β and INF-γ and decreased NF-ĸB protein expression. Tanshinone IIA has therapeutic effects in PD condition as results of antioxidant and anti-inflammatory properties. Therefore, this natural product is recommended in PD treatment.
Keywords
Tanshinone
Parkinson disease
Rats
Cytokine
Inflammation
1 Introduction
Parkinson's disease (PD) is the second most debilitating and common neurodegenerative disease, with a prevalence of 2% in people over 50 years (Veldman et al., 1998). PD leads to movement disorders such as muscle stiffness, resting tremor, akinesis, bradykinesia, poor balance (Sveinbjornsdottir, 2016), and degeneration of dopaminergic neurons in the dense area of the substantia nigra observed in PD (Song and Kim, 2016). Oxidative stress, increased lipid peroxidation (Nikam et al., 2009), decreased antioxidant enzymes (Mythri et al., 2011), overexpressed inflammatory cytokines including INF-gamma and IL-10 (Rathnayake et al., 2019), DNA fragmentation (Ferrer and Blanco, 2000) and iron accumulation (Xu et al., 2008) are the most important factors in the loss of dopaminergic neurons (Xu et al., 2008).
Angiotensin converting enzyme [ACE] is the major enzyme in the renin-angiotensin system [RAS] and is an exopeptidase that converts angiotensin I to II. Angiotensin II activates NADPH-dependent oxidases, leading to the production of superoxides (Nikam et al., 2009). Angiotensin II degrades dopamine nerves by generating reactive oxygen species [ROS] through AT1 receptors, and manipulation of RAS components is considered for treating Parkinson's patients (Mertens et al., 2009; Perez-Lloret et al., 2017).
Treatment options include dopamine agonists (Antonini et al., 2009), monoamine oxidase B [MAO-B] inhibitors (Teo and Ho, 2013), catechol-o-methyl-transferase [COMT] inhibitors (Kaakkola, 2000), and anticholinergics (Cooper et al., 1992) that are associated with unwanted side effects. Therefore, in recent years, researchers have focused on the use of plant-derived natural products in the treatment of PD (Sengupta et al., 2016). In this regard, the medicinal plants used in Traditional Chinese Medicine (TCM] deserve much attention in the treatment of PD.
Salvia miltiorrhiza, a plant from the Lamiaceae family, is one of the most widely used medicinal plants in China, and its root, Dansen, contains a large amount of tanshinones (Chen et al., 2013). The root extract of this plant has been used in the treatment of gingivitis, pharyngitis, diarrhea, night sweats, coronary heart diseases, and infectious diseases (Sharifi-Rad et al., 2018). The main constituents of the essential oil of this plant include phenolic acids and tanshinones such as salvianolic acid, dihydrotanshinone, tanshinones I, tanshinone IIA and cryptotanshinone (Jia et al., 2019). Tanshinone IIA neuroprotection effects (Dong et al., 2009) and inhibitions of dopaminergic degeneration (Zhang et al., 2019) and MAO-B expression (Prajapati et al., 2021) were reported. It seems that this compound with its anti-inflammatory and anti-oxidant properties (Gao et al., 2012; Lu et al., 2009) can have high therapeutic potential in neurodegenerative diseases including PD. For example, Ren et al. showed that Tanshinone IIA have neuroprotective effects in rat model of PD and the action mechanism was attributed to the NADPH oxidase and iNOS inhibitions (Ren et al., 2015). Tanshinone IIA, also showed the ability to inhibit the α-synuclein aggregation (Ji et al., 2016). However, the mechanism of neuroprotective effects of this compound is not well understood and further studies are needed.
Therefore, the current study was aimed to evaluate the protective impacts of tanshinone IIA in PD and to identify its action mechanisms to gain a broad insight into its therapeutic effects.
2 Materials and methods
2.1 Animals and drug administration
After preparation of 56 Wistar rats [200–250 g], they were divided into 7 groups [n = 8):
Group I: Control 0.9% saline); Group II: Control received 100 mg/kg tanshinone IIA [Sigma-Aldrich, purity ≥ 97%; Cas# 568–72-9, USA), Group III: Sham; Group IV: PD group [8 μg/μl 6-OHDA [Sigma-Aldrich, Cas# 28094–15-7, USA)); Group V: PD group receiving 25 mg/kg tanshinone IIA; Group VI: PD group receiving 50 mg/kg tanshinone IIA; Group VII: PD group receiving 100 mg/kg tanshinone IIA. Tanshinone IIA was administered for 10 days by oral gavage. All animal used procedures were carried out in accordance with the Regulations of Experimental Animal Administration.
2.2 Induction of Parkinson disease
The rats were first weighed and then anesthetized by IP injection of a combination of 100 mg/kg ketamine and 5 mg/kg xylazine. The rats were next fixed in a stereotaxic apparatus [Stoelting, USA). According to the coordinates of the Watson & Paxinos atlas, the SNC core point coordinates [Ap-4.8 mm to bregma, ML 2 mm [left), DV 8.3 mm) from the surface of the skull were determined to create an animal model of PD. In the PD groups were injected 0.8 μg/μl 6-OHDA into the left hemisphere substantia nigra. In the control group, 5 μl of 0.9% saline solution was injected into the left area of the substantia nigra (Deumens et al., 2002).
2.3 Behavioral tests
2.3.1 Apomorphine-induced rotation
6 animals in each group were evaluated for rotating behavior by IP injection of apomorphine hydrochloride [2.5 mg/kg, solved in 0.5 ml of 0.9% normal saline) ten days after PD induction over a period of 60 min. The rats were allowed to acclimatize for 10 min before injection and after one minute they were put in a transparent plastic cylinder [33 cm × 35 cm). The total number of 360-degree rotations was measured for 60 min at 10-minute intervals in a completely solitary room. The number of contralateral rotations [rotation to the opposite side of the lesion, right) was considered as a positive number and ipsilateral rotation [rotation to the lesion site, left) was considered as a negative number. The net number of rotations was calculated by the subtraction of rotations in two directions (Fujita et al., 1996).
2.3.2 Muscle rigidity test
The animals were placed on the table and did not receive a grade if his posture and gait were normal. The animal was given a score of 0.5 when the animal was placed on a table and remained motionless. Then, the animal's right hand was put on a platform [3 cm high). If the rat did not lift its hand for at least 10 s, 0.5 score was considered. The same was then done on the left hand. Finally, the animal's right hand was placed on a 9 cm platform so that it did not come into contact with other parts of the body. If the rats did not move, a score of 1 was given and this operation was repeated on the right hand. The rats in which PD was induced take 3.5score and healthy animals received 0 (Morpurgo, 1962).
2.4 Biochemical parameters
For biochemical tests, the animals were anesthetized 24 h after receiving the last dose of Tanshinone IIA, then after decapitation, the hippocampal tissue of the brain was extracted. The hippocampus was frozen very quickly at −80 °C.
2.4.1 Lipid peroxidation
The lipid peroxidation was measured based on the formation of malondialdehyde [MDA) complex formed with thiobarbituric acid reactive substances [TBARS). In summary, tissue sections were put in Na2PO4/KH2PO4 buffer pH 7.4 isotonized with KCl and containing 200 μM butyl hydroxytoluene and 200 μM deferoxamine. It was homogenized with a homogenizer [Tomy Micro, Smash-MS-100, Germany) at 4000 rpm for 90 s. Then, 200 μl of the homogenized solution mixed with 200 μl of 8% SDS detergent [Sodium dodecyl sulfate) and 750 μl of 20% acetic acid, next it vortexed for 1 min and incubated with 750 μl of 0.8% thiobarbituric acid at 95 °C for 90 min. Then 3 ml of N-butanol was added and centrifuged at 4000 rpm for 5 min. Finally, its absorption was read at 532 nm by spectrophotometer (Lopez-Real et al., 2005).
2.4.2 Protein oxidation
Protein oxidation was evaluated by measuring the carbonyl components of the protein. The carbonyl components of the proteins were measured by spectrophotometry. For this purpose, homogeneity was treated with 1% streptomycin and then centrifuged at 13000 rpm for 5 min. After separation, the supernatant was sonicated for 1 min with 1 M trichloroacetic acid [Ulterasonic System Tecna 3) and then centrifuged at 13000 rpm and then 200 μM 0.5 M NaOH for 3 min. 800 μl of Dinitrophenyl Hedrazine 2–4 in 2 M hydrochloric acid was added to the pellet and kept in the dark at room temperature for 1 h. The solution was then centrifuged in 200 μl of 1 M trichloroacetic acid for 2 min. 500 μl ethyl acetate: ethanol [1:1v/v) was used to wash supernatant. 800 μl of 6 M guanidine was added to each tube containing the pellet and then read by a spectrophotometer [Cecil CE, 2501) at 370 nm (Lopez-Real et al., 2005).
2.4.3 Mitochondrial respiratory enzymes
Measurements of SDH [Succinate Dehydrogenase Assay Kit, Cat# MAK197), MDH [Malate Dehydrogenase Activity Assay Kit, Cat# MAK196) and LDH (Lactate Dehydrogenase (LDH) Assay kit,Cat# MAK066) were done using sigma–aldrich, USA kits.
2.4.4 Reactive oxygen species (ROS), superoxide dismutase (SOD), catalase (CAT) and reduced glutathione (GSH)
Total reactive oxygen species (ROS) (Invitrogen™, Cat# 88-5930-74, USA), superoxide dismutase (SOD) (Invitrogen™, Cat# EIASODC, USA), catalase (CAT) (Invitrogen™, Cat# EIACATC, USA) and reduced glutathione (GSH) (Invitrogen™, Cat# EIAGSHC, USA) kits were used to measure ROS, SOD, CAT and GSH contents in rat hippocampus homogenate.
2.4.5 IL-1β, TNF-α, INF-γ, IL-10, NF-ĸB
IL-1β, TNF-α, INF-γ, IL-10, NF-ĸB, p38 MAPKs and TK were measured using enzyme-linked immunosorbent assay (ELISA). Briefly, rat hippocampal homogenates were prepared and the expression levels of IL-1β (Invitrogen™, Cat # BMS630, USA), TNF-α (Invitrogen™, Cat # BMS622, USA), INF-γ (Invitrogen™, Cat # BMS621, USA), IL-10 (Invitrogen™, Cat # ERA23RB, USA) and NF-ĸB (Invitrogen™, Cat # 85-86083-11, USA) evaluated using kits. Briefly, in 96-well plate wells, 100 ƛ diluted protein monoclonal antibodies were first poured and incubated for 24 h at room temperature. The existing antibodies were then blocked with PBS buffer and 5% bovine serum albumin solution. Then, standard samples were added to each of the wells. After 2 h incubation and re-washing, monoclonal antibodies against each of the proteins were added to the wells, and after 2 h incubation, a conjugated solution diluted in 100 ƛ was added to each well. After incubation for 90 min at room temperature and washing, a solution of chromogenic substrate was added to each well and the reaction blocked after 30 min of incubation in the dark and then the amount of light absorption in specific wavelengths of each protein was read by ELISA-reader and matched with the standard curve.
2.5 Statistical analysis
ANOVA and Tukey post hoc test (P < 0.05) was used to investigate significant differences in means. Data analysis was done by GraphPad prism V.8 software.
3 Results
3.1 Behavioral tests
Induction of PD by neurotoxin 6-OHDA resulted in increased rotations in the apomorphine-induced rotation test (Fig. 1a) and increased muscle rigidity (Fig. 1b). However, administration of tanshinone IIA in a dose-dependent manner reduced the mean rotation and muscle rigidity in Parkinson's rats. The greatest reduction in rotation and muscle rigidity was observed in PD rats receiving 100 mg/kg tanshinone IIA (Fig. 1), indicating Tanshinone IIA prevented lesion and dopaminergic neuron degeneration, thus having neuroprotective effects.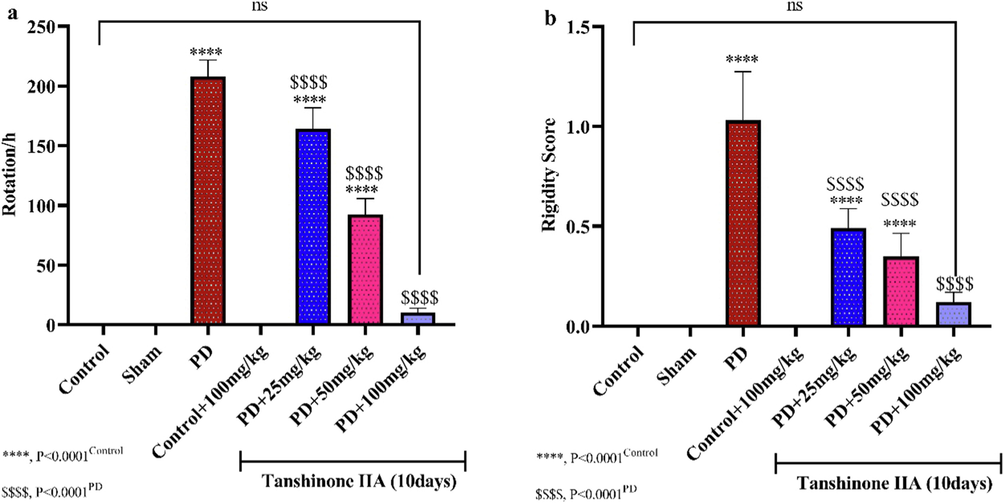
The effect of administering different doses of tanshinone IIA for 10 days on the mean rotation in Apomorphine-induced rotation test (a) and muscle rigidity (b) (n = 6).
3.2 Biochemical parameters
3.2.1 MDA and protein oxidation
The rate of lipid peroxidation and protein oxidation increased in rats administered by 6-OHDA (Fig. 2). However, oral administration of 50 and 100 mg/kg tanshinone IIA to PD rats for 10 days significantly reduced lipid peroxidation and protein oxidation in hippocampal homogenates, indicating the antioxidant effects of tanshinone IIA (see Fig. 3).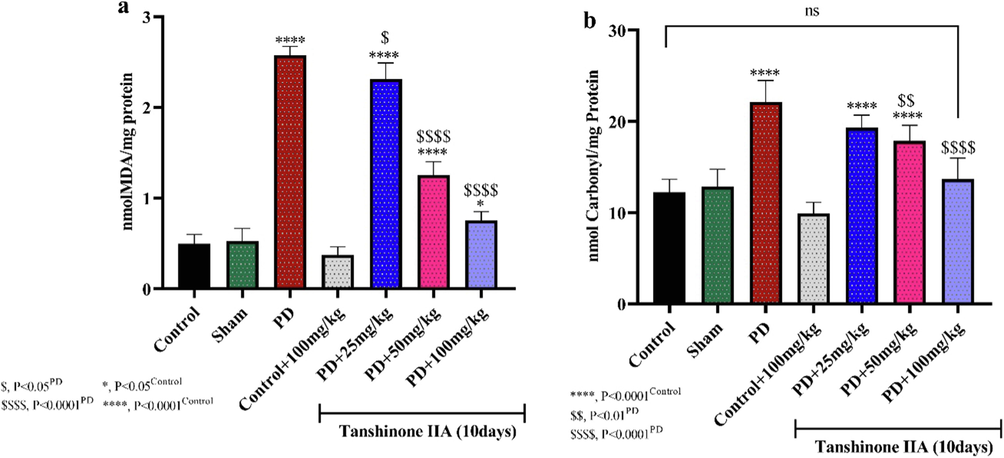
The levels of Malondialdehyde (MDA) (a) and lipid peroxidation (b) in the hippocampus of rats. tanshinone IIA was gavaged to rats for 10 days and hippocampal homogenate prepared 24 h after receiving the last dose (n = 5). Significant reductions in MDA levels and protein peroxidation were observed in the hippocampus of rats receiving 100 mg/kg tanshinone IIA.
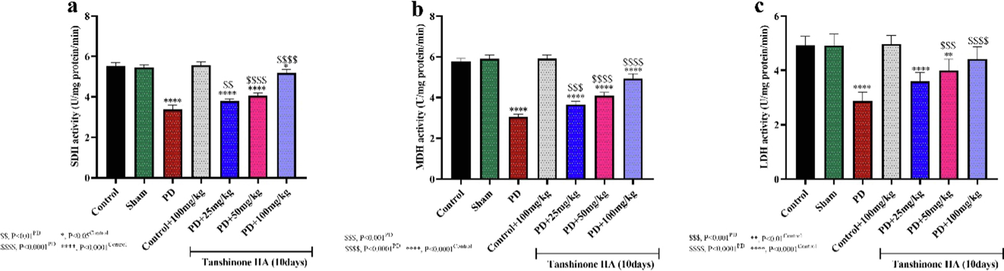
The effect of administering different concentrations of tanshinone IIA for 10 days on the activities of succinate dehydrogenase, lactate dehydrogenase and malate dehydrogenase enzymes in rat hippocampal homogenates (n = 5).
3.2.2 Succinate dehydrogenase, lactate dehydrogenase and malate dehydrogenase
Measurement of the activities of major mitochondrial respiratory enzymes indicated that 6-OHDA-induced Parkinson's disease significantly reduced the activity of the enzymes succinate dehydrogenase, lactate dehydrogenase and malate dehydrogenase (Fig. 2). Oral administration of 100 mg/kg tanshinone IIA for 10 days restored the activity of these respiratory enzymes. In PD groups, administration of tanshinone IIA in a concentration-dependent manner led to a significant increment in the activities of SDH, MDH and LDH compared to PD rats, indicating the protective effects of tanshinone IIA on the activity of respiratory enzymes in Parkinson's disease.
3.2.3 Reactive oxygen species and antioxidant enzymes
The ROS content in the hippocampal tissue of rats in the 6-OHDA group demonstrated a significant augmentation relative to the control-saline group, so that the highest ROS content was observed in PD rats (Fig. 4a). The treatments of tanshinone IIA reduced ROS in the hippocampus of rats and the ROS content in the PD group receiving 100 mg/kg tanshinone IIA did not show a significant difference with the control/saline group. This may indicate the antioxidant properties of tanshinone IIA, which has reduced the oxidative stress induced by PD.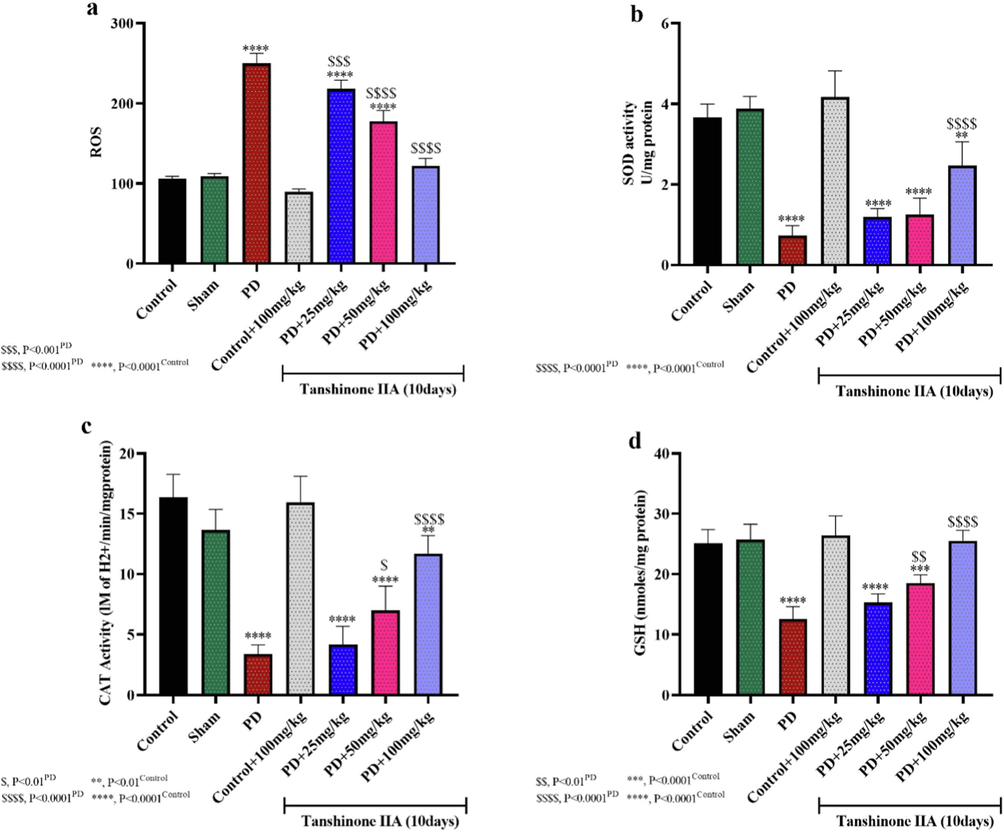
Reactive oxygen species (ROS) content (a), superoxide dismutase (SOD) (B) and catalase (CAT) enzymes activities, as well as reduced glutathione (GSH) in rat hippocampal homogenates (n = 5). Tanshinone IIA was gavage for 10 days and hippocampal homogenate prepared one day after receiving the last dose of tanshinone IIA.
Evaluation the activities of SOD and CAT antioxidant enzymes and reduced glutathione (GSH) levels in rat hippocampal homogenates showed that 6-OHDA-induced PD resulted in a significant reduction in SOD and CAT activities as well as GSH levels compared to the control/saline group (Fig. 4b, c, d). However, administration of 50 and 100 mg/kg tanshinone IIA restored the activity of SOD and CAT enzymes as well as GSH levels. Thus, tanshinone IIA reduces 6-OHDA-induced oxidative stress by increasing its antioxidant capacity.
3.2.4 IL-1β, TNF-α,INF-γ and IL-10
The results of the present study showed an increase in proinflammatory cytokines IL-1β, TNF-α and INF-γ and a decrease in the expression of IL-10 in the hippocampal homogenates of 6-OHDA rats (Fig. 5). However, 100 mg/kg tanshinone IIA decreased IL-1β, TNF-α,INF-γ levels and increased IL-10 expression compared to PD rats, indicating the anti-inflammatory properties of this compound and its therapeutic effects in PD conditions (see Fig. 6).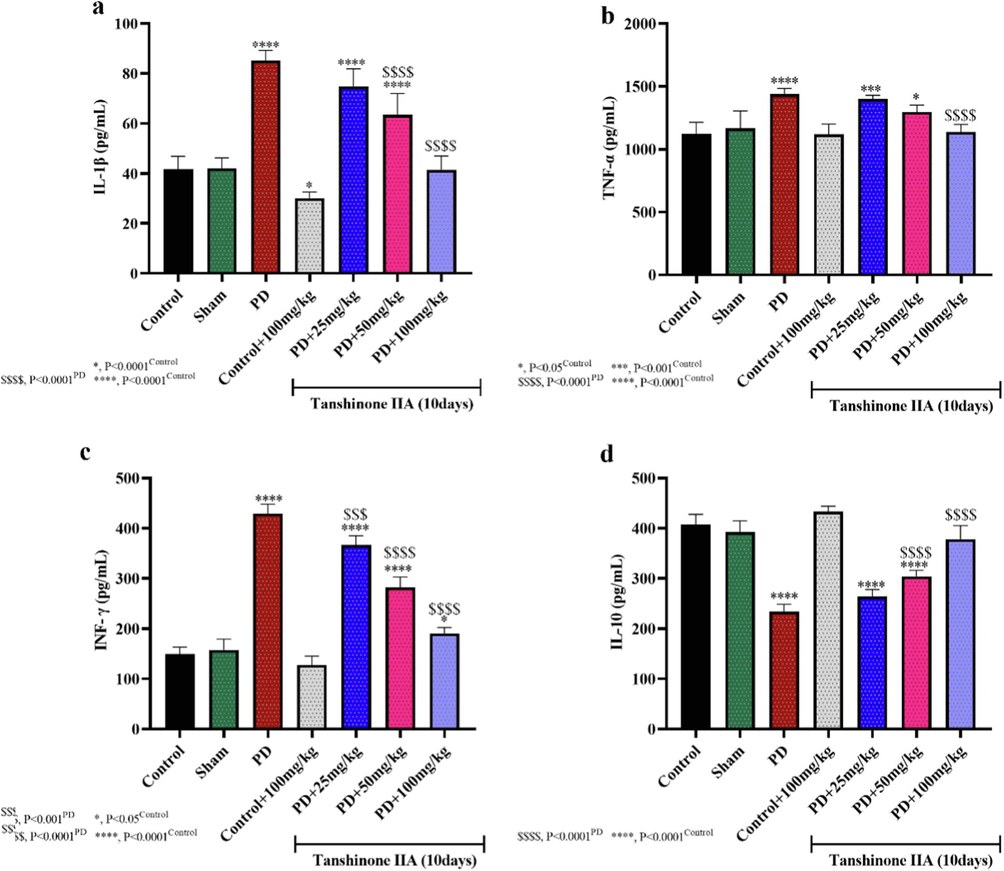
The effect of tanshinone IIA on the expressions of IL-1β (a), TNF-α (b), INF-γ (c) and IL-10 (d) in 6-OHDA -induced PD in rats (n = 5). Tanshinone IIA was administered by oral gavage for 10 days and the expressions of cytokines were studied 24 h after receiving the last dose.
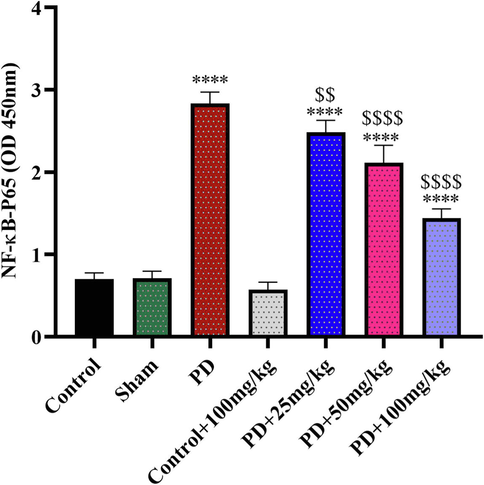
NF-ĸB expressions in rats' hippocampus homogenate (n = 5). ****:P<0.0001,Control/saline; $$: P<0.01,PD; $$$$:P<0.0001,PD. Tanshinone IIA was given to the rats for 10 days and 24 h after last dose administration the hippocampus homogenate prepared.
3.2.5 NF-ĸB
NF-ĸB upregulated in 6-OHDA-induced PD rats, but its expression downregulated in PD rats received tanshinone IIA in dose dependent manner. The treatment of 100 mg/kg tanshinone IIA to PD rats led to the lowest expression of NF-ĸB, indicting anti-inflammation effects of tanshinone IIA.
4 Discussion
In recent years, Traditional Chinese Medicine (TCM) has been more considered in the treatment of diseases due to the side effects of chemical drugs (Lao et al., 2012). In the current study, the impact of tanshinone IIA was studied on the experimental model of PD induced by 6-OHDA injection in rats and the results indicated the healing potential of tanshinone IIA on PD due to its antioxidant and anti-inflammatory effects.
It has been suggested that oxidative stress and free radicals including nitric oxide (NO) and reactive oxygen species (ROS) cause the destruction of brain nerve cells and are involved in the pathophysiology of disorders such as AD and PD (Chen et al., 2012; Emerit et al., 2004). Free radicals oxidize macromolecules such as proteins, nucleic acids, and lipids, disrupting cellular homeostasis (Perry et al., 2008; Hekimi et al., 2011). The body system is protected against oxidative stress by antioxidants such as GSH, SOD, CAT and peroxiredoxin (Nimse and Pal, 2015). In the present study, behavioral tests such as muscle rigidity and apomorphine-induced rotation were used to diagnose PD in animals and investigate the effects of tanshinone IIA. Lipid peroxidation and oxidation of proteins and ROS content were used to evaluate oxidative stress in rats and the results showed an increase in oxidative stress in PD rats. This result is in line with other research that has shown involvement of oxidative stress in PD (Hwang, 2013; Henchcliffe and Beal, 2008).
Studies have shown the therapeutic potential of natural compounds in PD conditions (Nabavi and Sobarzo-Sánchez, 2021; Carrera et al., 2018) and the current study also demonstrated that tanshinone IIA has therapeutic effects on PD. The present study results showed that 100 mg/kg tanshinone IIA improved motor parameters in PD rats. This can be attributed to the neuroprotective effects of tanshinone IIA (Dong et al., 2009), which reduce degeneration of dopaminergic nerves in the SN and thus produce enough dopamine to transmit signals between the substantia nigra and other parts of the brain (Matsumoto and Hikosaka, 2009). In addition, preventing the increase of ROS and improving the antioxidant capacity in rats fed with tanshinone IIA can also be considered as a mechanism for neuroprotective effects of tanshinone IIA.
Studies have shown that serum and cerebrospinal fluid of PD patients have high levels of IL-12, TNF-α and IL- β 1 cytokines (Ferrari and Tarelli, 2011). Also, dopaminergic neurons of the substantia nigra are more sensitive to proinflammatory factors and mediators of oxidative stress due to the decrease in glutathione concentration in these neurons (Collins et al., 2012). In the present study, it was shown that in the experimental model of PD, the concentration of reduced glutathione was decreased and inflammatory cytokines TNF-α, IL-1β and INF-γ increased, which indicates a reduction of antioxidant capacity and an augmentation in inflammation. The onset of PD is associated with microglia cells activation and inflammatory cytokines upregulation (Roodveldt et al., 2008), and these inflammatory factors increase with the progression of PD (Nagatsu and Sawada, 2005). Inflammatory factors damage and reduce the number of dopaminergic neurons in the brain (Herrera et al., 2005). Thus, the use of anti-inflammatory drugs can be considered as a treatment option for PD (Gagne and Power, 2010). In the present study, increase in GSH and IL-10 and decrease in inflammatory cytokines TNF-α, IL-1β and INF-γ were observed in PD rats receiving 100 mg/kg tanshinone IIA, indicating a decrease in inflammation and an increase in antioxidant capacity, which can be responsible for the therapeutic effects of tanshinone IIA in PD conditions. The findings of the current study suggest that tanshinone IIA can prevent the reduction of PD rat function in behavioral tests, which can be attributed to the antioxidant, anti-inflammatory and neuroprotective effects of tanshinone IIA. Decreases in TNF-α cytokine levels were also observed in the hippocampus of PD rats as a result of tanshinone IIA administration. Since TNF-α is one of the most important components of the inflammatory pathway (Zelová and Hošek, 2013), it can be argued that tanshinone IIA may exert its therapeutic effect by altering the expression of inflammatory cytokines. Therefore, tanshinone IIA as a natural product with reduced inflammation can be considered as a treatment option in PD.
In the present study, it was shown that the activities of respiratory enzymes SDH, MDH and LDH decreases after induction of PD in the hippocampus of rats, indicating a defect in the respiratory electron transmission chain and oxidative phosphorylation (Chesworth et al., 1998). However, treatment of PD rats with tanshinone IIA increased the activities of these enzymes. The effects of improving the activity of mitochondrial enzymes in the present study can be attributed to tanshinone IIA antioxidant properties and prevention of damage to the mitochondrial membrane and electron transfer chain.
In the present study, it was shown that NF-ĸB expression levels increased after induction of PD by 6-OHDA. NF-ĸB is a protein whose activation in the nucleus induces the expression of inflammatory cytokine genes such as IL-1β, IL-6 and TNF-α (Moynagh, 2005). Hence, it is a key factor in increasing inflammation in PD (Sivandzade et al., 2019; Kaur et al., 2017). The results of the present study showed that tanshinone IIA reduced the expression of NF-ĸB in the hippocampus of PD rats. Therefore, downregulation the expression of inflammatory cytokines IL-1β, TNF-α and INF-γ can be attributed to decreased expression of NF-ĸB. These results indicate the strong anti-inflammatory and antioxidant effects of tanshinone IIA, which in line with other studies (Ren et al., 2015; Ji et al., 2016). Our study provide new insight about neuroprotective effects of tanshinone IIA in PD condition by providing evidence of its effects on mitochondrial enzyme activities and inflammatory cytokines.
One of the limitations of this study is that it should be further performed in vivo and clinical trials are needed to confirm the effect of tanshinone IIA in PD.
5 Conclusion
It is generally concluded that tanshinone IIA due to its antioxidant and anti-inflammatory properties can have neuroprotective effects on dopaminergic neurons in PD conditions. The use of this natural product can improve the symptoms of this disease and reduce the progression of the disease. However, clinical studies are needed to confirm this.
Acknowledgement
Not applicable.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- A reassessment of risks and benefits of dopamine agonists in Parkinson's disease. Lancet Neurol.. 2009;8(10):929-937.
- [Google Scholar]
- Dopaminergic Neuroprotection with Atremorine in Parkinsoń s Disease. Curr. Med. Chem.. 2018;25(39):5372-5388.
- [Google Scholar]
- Oxidative stress in neurodegenerative diseases. Neural Regenerat. Res.. 2012;7(5):376.
- [Google Scholar]
- Antioxidant activity and components of a traditional chinese medicine formula consisting of Crataegus pinnatifida and Salvia miltiorrhiza. BMC Complement. Alternative Med.. 2013;13(1):99.
- [Google Scholar]
- Electron Transport and Oxidative Phosphorylation. In: Chesworth J.M., Stuchbury T., Scaife J.R., eds. An Introduction to Agricultural Biochemistry. Dordrecht: Springer, Netherlands; 1998. p. :161-171.
- [Google Scholar]
- Contributions of central and systemic inflammation to the pathophysiology of Parkinson's disease. Neuropharmacology. 2012;62(7):2154-2168.
- [Google Scholar]
- Different effects of dopaminergic and anticholinergic therapies on cognitive and motor function in Parkinson's disease: a follow-up study of untreated patients. Brain.. 1992;115(6):1701-1725.
- [Google Scholar]
- Modeling Parkinson's Disease in Rats: An Evaluation of 6-OHDA Lesions of the Nigrostriatal Pathway. Exp. Neurol.. 2002;175(2):303-317.
- [Google Scholar]
- Neuroprotective effects of Tanshinone IIA on permanent focal cerebral ischemia in mice. Phytotherapy Research.. 2009;23(5):608-613.
- [Google Scholar]
- Neurodegenerative diseases and oxidative stress. Biomed. Pharmacother.. 2004;58(1):39-46.
- [Google Scholar]
- Parkinson's Disease and Systemic Inflammation. Parkinson’s Disease.. 2011;2011:436813.
- [Google Scholar]
- N-myc and c-myc expression in Alzheimer disease, Huntington disease and Parkinson disease. Mol. Brain Res.. 2000;77(2):270-276.
- [Google Scholar]
- Expression of dopamine transporter mRNA and its binding site in fetal nigral cells transplanted into the striatum of 6-OHDA lesioned rat. Mol. Brain Res.. 1996;39(1–2):127-136.
- [Google Scholar]
- Anti-inflammatory drugs and risk of Parkinson disease. Neurology.. 2010;74(12):995.
- [Google Scholar]
- Cardiovascular actions and therapeutic potential of tanshinone IIA. Atherosclerosis.. 2012;220(1):3-10.
- [Google Scholar]
- Taking a “good” look at free radicals in the aging process. Trends Cell Biol.. 2011;21(10):569-576.
- [Google Scholar]
- Mitochondrial biology and oxidative stress in Parkinson disease pathogenesis. Nature Clin. Practice Neurol.. 2008;4(11):600-609.
- [Google Scholar]
- Inflammatory process as a determinant factor for the degeneration of substantia nigra dopaminergic neurons. J. Neural Transm.. 2005;112(1):111-119.
- [Google Scholar]
- Inhibition effects of tanshinone on the aggregation of α-synuclein. Food Funct.. 2016;7(1):409-416.
- [Google Scholar]
- Salvia miltiorrhiza in diabetes: A review of its pharmacology, phytochemistry, and safety. Phytomedicine.. 2019;58:152871.
- [Google Scholar]
- Clinical Pharmacology, Therapeutic Use and Potential of COMT Inhibitors in Parkinson’s Disease. Drugs.. 2000;59(6):1233-1250.
- [Google Scholar]
- Neuroinflammation - A major cause for striatal dopaminergic degeneration in Parkinson's disease. J. Neurol. Sci.. 2017;381:308-314.
- [Google Scholar]
- Traditional Chinese Medicine. In: Längler A., Mansky P.J., Seifert G., eds. Integrative Pediatric Oncology. Springer, Berlin Heidelberg: Berlin, Heidelberg; 2012. p. :125-135.
- [Google Scholar]
- Angiotensin-converting enzyme inhibition reduces oxidative stress and protects dopaminergic neurons in a 6-hydroxydopamine rat model of Parkinsonism. J. Neurosci. Res.. 2005;81(6):865-873.
- [Google Scholar]
- Experimental study of the anti-cancer mechanism of tanshinone IIA against human breast cancer. Int. J. Mol. Med.. 2009;24(6):773-780.
- [Google Scholar]
- Two types of dopamine neuron distinctly convey positive and negative motivational signals. Nature. 2009;459(7248):837-841.
- [Google Scholar]
- The role of the central renin-angiotensin system in Parkinson’s disease. Journal of the Renin-Angiotensin-Aldosterone. System.. 2009;11(1):49-56.
- [Google Scholar]
- Effects of antiparkinson drugs on a phenothiazine-induced catatonic reaction. Arch. Int. Pharmacodyn. Ther.. 1962;137:84-90.
- [Google Scholar]
- Evaluation of Markers of Oxidative Stress, Antioxidant Function and Astrocytic Proliferation in the Striatum and Frontal Cortex of Parkinson’s Disease Brains. Neurochem. Res.. 2011;36(8):1452-1463.
- [Google Scholar]
- Parkinsoń s and Alzheimeŕ s Diseases and Natural Products: Pathologies and Medication of the New Times. Curr. Neuropharmacol.. 2021;19(2):112.
- [Google Scholar]
- Inflammatory process in Parkinson's disease: role for cytokines. Curr. Pharm. Des.. 2005;11(8):999-1016.
- [Google Scholar]
- Oxidative stress in Parkinson’s disease. Indian J. Clin. Biochem.. 2009;24(1):98-101.
- [Google Scholar]
- Free radicals, natural antioxidants, and their reaction mechanisms. RSC Adv.. 2015;5(35):27986-28006.
- [Google Scholar]
- Renin-angiotensin system as a potential target for new therapeutic approaches in Parkinson’s disease. Expert Opin. Invest. Drugs. 2017;26(10):1163-1173.
- [Google Scholar]
- Alzheimer disease and the role of free radicals in the pathogenesis of the disease. CNS & Neurol. Disorders-Drug Targets (Formerly Current Drug Targets-CNS & Neurological Disorders). 2008;7(1):3-10.
- [Google Scholar]
- Monoamine Oxidase Inhibition by Major Tanshinones from Salvia miltiorrhiza and Selective Muscarinic Acetylcholine M4 Receptor Antagonism by Tanshinone I. Biomolecules.. 2021;11(7)
- [Google Scholar]
- Selected serum cytokines and nitric oxide as potential multi-marker biosignature panels for Parkinson disease of varying durations: a case-control study. BMC Neurol.. 2019;19(1):1-10.
- [Google Scholar]
- Tanshinone IIA prevents the loss of nigrostriatal dopaminergic neurons by inhibiting NADPH oxidase and iNOS in the MPTP model of Parkinson's disease. J. Neurol. Sci.. 2015;348(1):142-152.
- [Google Scholar]
- Immunological features of α-synuclein in Parkinson's disease. J. Cell Mol. Med.. 2008;12(5b):1820-1829.
- [Google Scholar]
- Plant-Derived Natural Products for Parkinson’s Disease Therapy. In: Essa M.M., Akbar M., Guillemin G., eds. The Benefits of Natural Products for Neurodegenerative Diseases. Cham: Springer International Publishing; 2016. p. :415-496.
- [Google Scholar]
- Salvia spp. plants-from farm to food applications and phytopharmacotherapy. Trends Food Sci. Technol.. 2018;80:242-263.
- [Google Scholar]
- NRF2 and NF-қB interplay in cerebrovascular and neurodegenerative disorders: Molecular mechanisms and possible therapeutic approaches. Redox Biol.. 2019;21:101059.
- [Google Scholar]
- Degeneration of Dopaminergic Neurons Due to Metabolic Alterations and Parkinson’s Disease. Frontiers in Aging. Neuroscience. 2016;8(65)
- [Google Scholar]
- Monoamine oxidase-B (MAO-B) inhibitors: implications for disease-modification in Parkinson’s disease. Translational Neurodegenerat.. 2013;2(1):19.
- [Google Scholar]
- Genetic and environmental risk factors in Parkinson’s disease. Clin. Neurol. Neurosurg.. 1998;100(1):15-26.
- [Google Scholar]
- Neuroprotective effect of the natural iron chelator, phytic acid in a cell culture model of Parkinson's disease. Toxicology. 2008;245(1):101-108.
- [Google Scholar]
- TNF-α signalling and inflammation: interactions between old acquaintances. Inflamm. Res.. 2013;62(7):641-651.
- [Google Scholar]
- Tanshinone IIA protects against dopaminergic neuron degeneration via regulation of DJ-1 and Nrf2/HO-1 pathways in a rodent model of Parkinson’s disease. Trop. J. Pharm. Res.. 2019;18(5):1017-1025.
- [Google Scholar]







