Translate this page into:
New mechanistic insights on Justicia vahlii Roth: UPLC-Q-TOF-MS and GC–MS based metabolomics, in-vivo, in-silico toxicological, antioxidant based anti-inflammatory and enzyme inhibition evaluation
⁎Corresponding author. Kashifur.rahman@iub.edu.pk (Kashif ur Rehman Khan)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Abstract
Identification of bioactives in nbutanol extract of the Justicia vahlii was performed using UPLC-QTOF-MS and GC–MS analysis. The acute oral toxicity test in mice and MTT assay confirmed the non-toxic nature of the extract. The extract showed antioxidant based anti-inflammatory potential in dose dependent manner. The extract showed significant in vitro enzyme inhibition potential.
Abstract
Justicia vahlii Roth. (acanthaceae) is an important medicinal food plant used in pain relief and topical inflammation. The present study aimed to evaluate phytochemical composition, toxicity, anti-inflammatory, antioxidant and enzyme inhibition potential of n-butanol extract of J. vahlii (BEJv). The extract prepared through maceration was found rich in total phenolic contents (TPC) 196.08 ± 6.01 mg of Gallic acid equivalent (mg GAE/g DE) and total flavonoid contents (TFC) 59.08 ± 1.32 mg of Rutin equivalent (mg RE/g DE). The UPLC-Q-TOF-MS analysis of BEJv showed tentative identification of 87 compounds and 19 compounds were detected in GC–MS analysis. The HPLC-PDA quantification showed the presence of 14 polyphenols amongst which kaempferol (3.45 ± 0.21 µg/ mL DE) and ferulic acid (2.31 ± 1.30 µg/ mL DE) were found in highest quantity. The acute oral toxicity study revealed the safety and biocompatibility of the extract up to 3000 mg/kg in mice. There was no effect of BEJv on human normal liver cells (HL 7702) and very low cytotoxic effect on liver cancer cells (HepG2) and breast cancer cells (MCF-7). In anti-inflammatory evaluation, the BEJv treated groups showed significant inhibition (p < 0.001) of late phase carrageenan induced paw edema at 400 mg/kg and increased the levels of oxidative stress markers; catalase, superoxide dismutase (SOD) and glutathione (GSH) while decreased the inflammatory markers; interleukin-1beta (IL-β) and tumor necrosis factor alpha (TNF-α) in paw tissue of mice. BEJv displayed highest results in Ferric reducing antioxidant power (FRAP) assay 97. 21 ± 2.34 mg TE (trolox equivalent)/g DE, and highest activity 3.32 ± 0.31 mmol ACAE (acarbose equivalent)/g D.E against α-glucosidase. Docking study showed good docking score by the tested compounds against the various clinically significant enzymes. Conclusively the current study unveiled J. vahlii as novel non-toxic source with good antioxidant-mediated anti-inflammatory potential which strongly back the traditional use of the species in pain and inflammation.
Keywords
Justicia vahlii
Chemical profiling
Anti-inflammatory
Oxidative stress
Enzyme inhibition
- BEJv
-
n-butanol extract of Justicia vahlii
- DPPH
-
2,2-diphenyl-1-picrylhydrazyl)
- ABTS
-
(2,2-azinobis(3-ethylbenothiazoline)-6-sulfonic acid)
- CUPRAC
-
Cupric reducing antioxidant capacity
- FRAP
-
Ferric Reducing Antioxidant Power
- GAE
-
Gallic acid equivalent
- RE
-
Rutin equivalent
- ACAE
-
Acarbose equivalent
- TUE
-
Thiourea equivalent
- KAE
-
Kojic acid equivalent
- DE
-
Dry extract
- p.o
-
Per orally
- i.p
-
Intraperitoneally
- r.t
-
Retention time
- SOD
-
Superoxide dismutase
- GSH
-
Glutathione reductase
- UPLC-QTOF-MS
-
Ultra-performance liquid chromatography- quadrupole time-of-flight-mass spectrometry
- GCMS
-
Gas chromatography-mass spectrometry
- TNF
-
Tumor necrosis factor
- IL
-
Interleukin
- MTT
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide
Abbreviations
1 Introduction
Inflammation is an immune response of the living organism or vascularized tissue(s) against burn, trauma, infection, injury or pathological alterations in the body (Ouda et al., 2021). Over the decades, number of studies revealed the interrelationship of inflammation and oxidative stress (Yesmin et al., 2020). Oxidative stress has been described as the imbalance between the production of reactive oxygen species (ROS) (free radicals) such as hydroxyl, superoxide anion, peroxyl and hydroperoxyl, and the antioxidant defense system (Ahmad et al., 2019). Normally cells produce low concentration of ROS for maintenance of cellular functions. However in pathological conditions, the imbalance of the physiological mechanism causes over production of ROS leading to overexpression of inflammatory mediators which initiate and maintain inflammation (Ansari et al., 2020). The increased ROS production activates the cellular defense mechanism against oxidative stress to eliminate the ROS from the cell. Various enzymes such CAT, SOD and endogenous antioxidant like GSH are the part of the cellular antioxidant defense system (Fukui and Zhu 2010). SOD demolish oxidative stress by converting reactive oxygen (O2–) to hydrogen peroxide (H2O2), and the H2O2 is then converted to water and oxygen molecules by CAT (Ansari et al., 2020). Whereas, the free radical mediated release of inflammatory markers decrease the levels of the endogenous antioxidant markers and devastate the inflammation (Li et al., 2020).
Considerable research studies showed the involvement of oxidative stress mediated inflammation in etiology of many diseases such as diabetes, aging, skin tissue damages and peptic ulcer (Tandon et al., 2004, Babu et al., 2015, Asmat et al., 2016). Therefore, the developing drugs that counter the oxidative stress are considered suitable choice for combating the disease(s) that involve oxidative stress. The therapeutic agents used for the management of diabetes are based on targeting enzymes α-amylase and α-glucosidase. The malfunctioned antioxidant defense mechanism of human body may contribute drastically to the developed insulin resistance. So, there is need of therapeutic agents with maximum desired effects and minimal unwanted effects. Clinically significant enzymes are being considered as one of the suitable drug targets while addressing a pathophysiological condition. For instance tyrosinase can be targeted to avoid the overproduction of melanin, which is the underlying cause of various skin disorders. Similarly through the inhibition of urease, an enzyme responsible for initiation and development of peptic ulcer, the condition can be treated (Bakht et al., 2014, Ullah et al., 2019, Jeon et al., 2021). The intense need of new drug candidates with good safety profile increased the interest of researchers to seek the desired products from natural sources. Because nature has a long history of contribution in provision of functional bioactives (Pieczykolan et al., 2021).
J. vahlii Roth. with local name kodasoori and bhekkar and synonyms Justicia diffusa and Rustellaria vahlii (https://www.theplantlist.org), is an important medicinal food plant of the family acanthaceae used traditionally in treating tooth pain and various skin and respiratory disorders and as food additive (Begum and Khan 2001, Ignacimuthu et al., 2008, Maheshwari et al., 2012, Farooq et al., 2019, Basit et al., 2021). The use of advance hybrid techniques such UPLC-Q-TOF-MS and GC–MS for the identification of functional bioactives responsible for the pharmacological potential of plant species has been increased in last two decades due to the multiple advantages of the techniques (Qi et al., 2014). To the best of our knowledge and literature search there is very limited literature available to date on pharmacological and phytochemical aspects of J. vahlii. Therefore, the current study was designed to evaluate comprehensively phytochemical profile, toxicity, anti-inflammatory, antioxidant and enzyme inhibition potential of n-butanol extract of J. vahlii (BEJv). In this study the traditional use of the species in inflammation was scientifically explored.
2 Material and methods
2.1 Plant material collection
The whole plant material of J. vahlii was collected from District Bannu, Khyber Pakhtunkhwa, Pakistan, which was identified by Prof. Dr. Ghulam Sarwar at the Department of Botany, The Islamia University of Bahawalpur, Punjab, Pakistan, and a specimen was deposited in the herbarium of the university with a voucher number 145/Botany for future record and reference. The plant material was washed thoroughly with distilled water to remove the impurities and dust, followed by shade drying at ambient temperature in a well-ventilated room.
2.2 Preparation of extract
The dried plant material (5 kg) was then subjected to grinding followed by extraction with n-butanol (8 L). The soaked material was shaken mechanically with a glass rod occasionally for 15 days, and then it was filtered two times using Whatman No. 1 filter paper. The process of filtration was repeated for three times to get fine filtrate. The filtrate was concentrated under reduced pressure at 45 ˚C using rotary evaporator which yielded a semisolid mass of 67 g of n-butanol extract of J. vahlii (BEJv). The percent (%) yield was calculated on dry weight basis using the below formula;
2.3 Phytochemical profiling
2.3.1 Total Phenolic (TPC) and flavonoid (TFC) content determination
The total phenolic contents (TPC) and total flavonoid contents (TFC) were determined using Folin-Ciocalteu and aluminium trichloride methods respectively (Uysal et al., 2018). The results of TPC were expressed as milligrams of gallic acid equivalents per gram of dry extract (mg GAE/g DE) and TFC were noted as milligrams of rutin equivalents per gram of dry extract (mg RE/g DE).
2.3.2 UPLC-Q-TOF-MS analysis
The chemical composition of BEJv was evaluated using Triple TOF™ 5,600 (AB Sciex, Foster, USA) LC-MS mass spectrometry with a Duo Spray TM ion source. The chromatographic separation was performed by using a Prominence TM UPLC system (Shimadzu, Japan). The apparatus employed for analysis include CBM-20A lite controller system, Autosampler Shimadzu SIL-30AC. The identification of compounds was carried out in accordance with the Natural Product HR-MS/MS Spectral Library 1.0 software database, which contains approximately 1000 plus Chinese standard reference compounds, previous literature support and Metlin database (Hu et al., 2020, Saleem et al., 2020).
2.3.3 Polyphenolic quantification using HPLC-PDA analysis
HPLC-PDA analysis was applied for quantification of polyphenolic compounds in BEJv using standard described method (Locatelli et al., 2017). The standard compounds; chlorogenic acid, catechin, syringic acid, vanillic acid, p-hydroxy benzoic acid, naringin, ferulic acid, o-coumaric acid, narigenin, cinnamic acid, p-coumaric acid, 2,3-dimethoxy-benzoic acid, rutin, 3-hydroxy-4-dimethoxy-benzoic acid, 3-hydroxy benzoic acid, epicatechin, gallic acid, gentesic acid, kaempferol, quercetin, apigenin, emodin, skullcapeflavone were purchased from sigma Aldrich (Shanghai, China). The results were presented as μg/mg dry extract (DE).
2.3.4 GC–MS analysis
The BEJv was further characterized by employing GC–MS Agilent 7890B equipped with Mass hunter acquisition software. The instrument has an ultra-inert capillary non polar column (HP-5MS) with proportions of 30 mm £ 0.25 mm ID £ 0.25 mm film. The carrier gas used was Helium and flow rate set was 1 ml/min. Temperature set for injector operation was 250 °C and the temperature of oven was set in such a manner; 50 °C for five min, then gradually increased at 100 °C/ min to reach 250 °C, and finally to 3000 °C for 10 min at 70 °C/ min. The sample was dissolved in methanol, added to GC–MS vial placed in auto sampler, and 1uL of sample extract solution was injected and the split ratio was 20:1. The compound identification was performed using NIST-17 MS Library search while the percentage composition was computed from GC peak areas (Achakzai et al., 2019, Hayat and Uzair 2019).
2.4 Effect of BEJv on human normal and tumoral cell lines
2.4.1 Cell culture
The HL 7702 cell line (normal human liver cells), HepG2 (human liver cancer cell line) and MCF-7 (human breast cancer cell lines were obtained from the Cell Bank of Type Culture Collection of Chinese Academy of Sciences (Shanghai, China). The cells were grown in Dulbecco Modified Eagle’s Medium (DMEM) and Roswell Park Memorial Institute Medium (RPMI) culture media at 37 °C with 5 % CO2 and 95 % humidity in a gas incubator (BB 16 gas incubator, Heraeus Instruments GmbH). The cells were supplemented with 1 % non-essential amino acid (NEAA), 10 % Fetal Bovine Serum (FBS) and 1 % PEST.
2.4.2 In vitro MTT assay
The cytotoxicity study against HL 7702 (normal human liver cells), HepG2 (human liver cancer cells) and MCF-7 (human breast cancer cell lines) was carried out by (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) tetrazolium (MTT) assay using methodology prescribed (Chavan et al., 2020). Briefly in this method the cell lines were seeded in 96-well plates at a density of 1 × 104 cells/well and incubated for 24 h. After the incubation the cells were then treated with the extract at 25, 50, 100, 200 and 300 μg/mL and time interval of 24 and 48 h. Then MTT reagent (5 mg/mL in 20 ml phosphate buffered-saline (PBS)) was added per well and the cells were further incubated at 37 °C for 4 h with CO2. The medium was then removed and 150 μL DMSO was added to dissolve the formazan. Then after 10 min the absorbance were recorded at 490 nm by a microplate reader (BioTek, USA) to assess the cell viability.
2.5 In vitro antioxidant potential
Antioxidant potential of the BEJv was evaluated by using DPPH (2,2 diphenyl-1-picrylhydrazyl), ABTS (2,2 azinobis (3-ethylbenothiazoline)-6-sulfonic acid), ferric reducing antioxidant power (FRAP) and cupric reducing antioxidant capacity (CUPRAC) as per the standard protocols described by (Khurshid et al., 2019). Results were expressed in milligrams trolox equivalent per gram of dry extract (mg TE/g DE).
2.6 In vitro enzyme inhibition
The in vitro enzyme inhibition studies of the extract were performed against α-amylase, α-glucosidase and tyrosinase, following previously described protocols (Khurshid et al., 2019). The α-amylase and α-glucosidase activities were expressed as milli mole acarbose equivalent (mmol ACAE/g DE), while tyrosinase inhibition was expressed in milligram kojic acid equivalent (mg KAE/g DE). anti-urease assay was performed in accordance with the procedure described (Weatherburn 1967) with slight modifications. Total volume of solution used in the assay was 200 µL. 15 μL of urease, 15 μL of phosphate buffer (1 M, pH: 7), and 15 μL of the test sample (5 mg/mL) were added to a 96-well plate and incubated at 37 °C for 15 min. Then 40 μL of urea as a substrate was mixed and re-incubated in the same conditions. After incubation, pre-read was taken by measuring the absorbance at 630 nm. Then 45 μL of phenol and 70 μL of alkali reagents were added to the mixture. The reaction mixture was incubated again at 37 °C for 50 min. Absorbance was measured again at 630 nm as post-read. Methanol was taken as a negative control and thiourea was taken as a positive control. Results were expressed as milligrams thiourea equivalent per gram of dry extract (mg TUE/g DE).
2.7 In vivo toxicological evaluation
2.7.1 Animals
BALB/c mice weighing 25–30 g were purchased from the animal house of the National Institute of Health Rawalpindi Pakistan and were acclimatized at 25 ± 2 °C under a 12 h dark/light cycle for 7 days. Animals were housed six mice per cage with ad libitum access to food and water. The experimental procedures for this study were approved by the Institutional Ethical Committee of Faculty of Pharmacy, The Islamia University of Bahawalpur (Permit number 41-2021/PAEC) which were in accordance with the ethics and guidelines of the UK Animals (Scientific Procedures) Act 1986 and conformed to the Animal Research: Reporting of In Vivo experiments (ARRIVE) (Liu et al., 2016).
2.7.2 Acute oral toxicity test
Acute Oral toxicity assessment was conducted according to Test No. 423 of the Organization for Economic Co-operation for Development (OECD) guidelines and standard protocols described (OECD. 1994, Syahmi et al., 2010). The mice were categorized into 3 groups with 6 mice in each group. The control group was treated with normal saline (10 ml/kg p.o.), group 2 and 3 received BEJv via oral gavage tube dissolved in normal saline at 1000 mg/kg and 3000 mg/kg respectively. The animals were continuously observed for the first 2 h and then every 24 h post dose administration for 14 days for spontaneous activity, aggressiveness, cyanosis, ataxia, tail pinch response, righting reflex, writhing, convulsions, catalepsy and bizarre behavior. On day 14 all the animals were euthanized by using ketamine 350 mg/kg body weight and blood was collected for biochemical analysis and vital organs for histopathological examination.
2.8 anti-inflammatory potential of BEJv
2.8.1 Carrageenan induced paw edema model
The anti-inflammatory potential of BEJv was evaluated by using carrageenan induced paw edema model in mice according to previously established method (Yang et al., 2020) with minor modifications. The mice were divided into 5 groups (n = 6). Group 1 was control group which received normal saline (10 ml/kg), group 2 was positive control and received dexamethasone subcutaneously (1 mg/kg) while groups 3–5 received orally BEJv at dose of 100 mg/kg and 200 mg/kg and 400 mg/kg. After 60 min of dosing all the mice were injected carrageenan (100 µL, 1 %) subcutaneously into the right hind paw. The change in the volume of the paw was measured as marker of induction of inflammation. The initial volume (V1) was measured before carrageenan administration and after injection volume (V2) was measured at 0.5, 1, 2, 4 and 5 h using the plethysmometer (Ugo Basile) and percent inhibition of edema was calculated by using the below formula:
% Inhibition = (Vcontrol – Vtreated)/ Vcontrol × 100.
All the animals were sacrificed using ketamine 350 mg/kg immediately at the 5th hour of the experiment and the paw tissue portion was separated quickly. Homogenates of paw sample (10 % w/v) of different treatment groups were prepared using tris-HCl buffer (0.1 M and pH 7.4) and sterilized water and used for estimation of oxidative stress markers and inflammatory markers.
2.8.2 Effect of BEJv on oxidative stress markers
CAT levels in the paw tissue homogenate was estimated according to standard protocols (Doherty et al., 2010). Briefly, H2O2-phosphate buffer (3 ml) was taken in an experimental cuvette and then enzyme extract (40 µL) was added rapidly followed by thorough mixing. The absorbance of reaction mixture was recorded using UV-spectrophotometer at 240 nm. Results were expressed in U/mg protein. Levels of SOD in the homogenate was quantified using standard protocol (Kakkar et al., 1984) with slight modifications. In this method, the superoxide anion mediated reduction of nitro blue tetrazolium (NBT) to formazan was measured at 540 nm. The SOD addition stopped the reaction and the extent of the inhibition was expressed in U/mg protein as enzyme activity. GSH was measured according to established method (Farombi et al., 2007) with minor modifications. Briefly, the homogenate (0.1 ml) was mixed with 2.4 ml of phosphate buffer. 5, 5′-dithiobis(2-nitrobenzoic acid) (DTNB) solution was added to make the final volume 3 ml. The readings were recorded at 412 nm using spectrophotometer. Results were expressed in nmol/mg protein.
2.8.3 Effect of BEJv on IL-1β and TNF-α expression (using ELISA kit)
The tissue samples from all the treated groups for determination of IL-1β and TNF-α were prepared according to the standard method (Khan et al., 2019) with minor modifications. The sample was then homogenized and centrifuged at 3000 g for 10 min and the supernatant was stored at −20 °C. IL-1β and TNF-α quantification was carried out using commercially available TNF-α and IL-1β ELISA kit (eBioscience, Inc., San Diego, CA).
2.9 In silico molecular docking studies
Three bioactives identified in the UPLC-Q-TOF-MS analysis of BEJv namely skullcapflavone-II, sesaminol and conidendrin were selected for docking studies with α-amylase, α-glucosidase, tyrosinase and urease. Structures of the compounds were downloaded from the zinc database (Irwin et al., 2012). AM1 semi-emperical method using guassian 09 software was applied for optimization of 3D structures to the ground state energy. Structure of the target proteins; urease, tyrosinase and α-amylase were downloaded from protein data bank using PDB ID: 4H9M, 4P6R, and 4GQR respectively (Berman et al., 2000). To date, no crystal structure of α-glucosidase is reported in the protein data bank; hence first homology model was generated using previously described method (Yamamoto et al., 2010). Discovery Studio Client software was used for the preparation of all target protein structures, which removes water molecules, heteroatoms, assign charges and add hydrogens and missing residues (if any). After preparation of targets structures, the active site was defined using co-crystal ligands and centroid on all residues within 10 Å co-crystal ligands for each targeted protein. Autodock4 software (Molinspiration Database) was used to add polar hydrogens, neutralize the protein structures using Kollman united atom charges and to perform the docking calculations. Docking the three inhibitors was performed in a 60 × 60 × 60 grid box with 0.375 Å distance between points using Lamarckian genetic algorithm (LGA) for the prediction of binding free energies. Two hundred fifty conformations were produced for each enzyme-inhibitor complex and the control ligand. The docked conformations were ranked into clusters based on the binding free energy (ΔG). Discovery studio 5.0 visualizer was used to visualize to docked inhibitors at the active site and identify the intermolecular interactions with the active site.
2.10 Statistical analysis
Each assay was performed in triplicate. All the results were presented as mean ± standard deviation of at least 3 experiments in triplicate. The data were analyzed using one-way ANOVA, followed by post hoc Tukey’s multiple comparison test. The level of p < 0.05 was used to determine statistical significance. All calculations were carried out through Graph Pad Prism 7.0 software.
3 Results and discussion
3.1 TPC, TFC and UPLC-Q-TOF-MS analysis
The current study is the first report on comprehensive phytochemical composition, toxicological and pharmacological evaluation of BEJv. Plant secondary metabolites particularly phenols and flavonoids are responsible for the biological functions of plants. Previous investigations have demonstrated the identification, extraction and pharmacological effects of different bioactives (Ahmad et al., 2019, Pervaiz et al., 2020). Solvent selection is an important step in extraction of natural products from plant origin. n-butanol is considered as one of the most suitable solvents used in extraction processes (Liste and Alexander 2002).
In the present study BEJv was evaluated for determination of total phenolic and flavonoid contents (Table 1). The quantification revealed the BEJv as rich in TPC 196.08 ± 6.01 mg GAE/g DE and TFC (59.08 ± 1.32 mg RE/ g DE). Higher phenolic contents than flavonoids in the extract were expected as flavonoid is a subclass of phenols. The extract with 100 % n-butanol solvent showed higher quantity of phenols and flavonoids that showed the n-butanol as suitable choice for preparation of plant extracts with high yield of phenols and flavonoids (Jan et al., 2013, Jing et al., 2015). All values are expressed as mean ± SD., (n = 3). RE: rutin equivalent; GAE: gallic acid equivalent; DE: dry extract.
BEJv
Yield of extract (g)
67
Percent yield
1.34 %
TFC mg RE/g DE
59.08 ± 1.32
TPC mg GAE/g DE
196.08 ± 6.01
Based on rich quantity of phenols and flavonoids observed in the preliminary quantification, BEJv was subjected to chemical profiling using highly sophisticated technique UPLC-QTOF-MS. The present analysis showed the identification of total 87 different phytoconstituents, 48 in positive mode of ionization (Table 2) and 39 in negative mode of ionization (Table 3). Total ionic chromatograms of positive and negative mode are displayed in Figs. 1 & 2 respectively. Peak detection was based on comparing accurate mass with ± 5 error of accuracy and isotope ratio with a tolerance of 10 % difference with the established database using Peakview 1.2 software (ABSciex, USA). Among the identified compounds the number of flavonoids and their derivatives were in abundance, which were tentatively identified in positive mode and in negative mode of ionization. Second highest class of secondary metabolites were phenolic compounds followed by alkaloids. Moreover, hydroxycinnamic acid glycoside, lignans, and coumarine, stilbene and phenylpropaonoid, iridoid glycoside, diterpene, quinic acid, anthocyanin glycoside and anthocyanin flavonoid each were tentatively identified in the current analysis of BEJv. The present analysis showed the presence of number of phytoconsituents which are reported with good antioxidant and anti-inflammatory potential in previous investigations (Maleki et al., 2019, Piva et al., 2021). RT: retention time; B. peak: base peak; error pm = ± 5. RT: retention time; B. peak: base peak; error pm = ± 5.
Peak no.
RT (min)
Mol. mass
Tentative identification
Mol. Formula
Adduct
B. peak (m/z)
Error ppm
Class
M/S Product ions
Reference
1
1.01
289.71
Epicatechin
C15H14O6
+H
290.71
1.9
Flavanol
109.05, 151.23, 203.05, 245.08
(Zhuang et al., 2018
2
3.95
189.10
Castanospermine
C8 H15 NO4
+H
190.10
−1.4
Alkaloid
74.02, 88.03, 116.03, 144.10, 149.06
(Rajana et al., 2018)
3
5.34
227.09
Flindersine
C14 H13 NO2
+H
228.10
−1.1
Alkaloid
79.05, 103.05, 107.05, 149.05, 209.87, 228.10
(Cantrell et al., 2005)
4
5.36
195.08
Damascenine
C10 H13 NO3
+H
196.09
−4.4
Alkaloid
Nil
(Sieniawska et al., 2019)
5
6.03
180.04
Caffeic acid
C9 H8 O4
+H
181.04
0.2
Phenol
65.04, 82.04, 93.03, 120.95, 121.95, 123.00, 181.04
(Wang et al., 2015)
6
6.03
198.05
Syringic acid
C9 H10 O5
+H
199.05
−0.9
Phenol
65.04, 77.03, 97.02, 120.03, 136.93, 140.04, 155.06, 180.91
(Li et al., 2022)
7
7.04
159.06
Echinopsine
C10 H9 NO
+H
160.07
−4.5
Alkaloid
Nil
(Liu et al., 2021)
8
8.45
194.05
Ferulic acid
C10 H10 O4
+H
195.06
−1.4
Hydroxycinnamic acid
117.03, 145.02, 149.05, 177.05, 195.04
(Li et al., 2022)
9
8.7
192.04
Scopoletin
C10 H8 O4
+H
193.04
−0.8
Coumarin
64.84, 65.04, 71.93, 81.07, 93.03, 103.01, 112.96, 121.02, 133.89, 149.02, 151.90, 162.90, 193.09
(Fischer et al., 2011)
10
9.63
354.09
Chlorogenic acid
C16 H18 O9
+H
355.10
−0.2
Phenolic
59.05, 103.07, 117.03, 145.02, 177.05, 250.96, 266.99, 308.88, 355.06
(Clifford et al., 2007)
11
10.1
138.03
Sesamol
C7 H6 O3
+H
139.03
2.1
Phenolic
60.98, 77.03, 83.04, 102.94, 119.08, 120.95, 121.04, 139.10
(Mekky et al., 2021)
12
10.42
272.06
Naringenin
C15 H12 O5
+H
273.07
0.6
Flavonoid
119.05, 147.04, 153.01, 273.07
(Mekky et al., 2021)
13
11.23
302.07
Hesperetin
C16 H14 O6
+H
303.08
−0.2
Flavonoid derivative
153.01, 177.05, 285.03, 303.08
(Mekky et al., 2021)
14
11.48
505.13
Peonidin 3-O-(6′'-acetyl-glycoside)
C24H25O12
+H
506.14
3.4
Anthocyanin
nil
(Bennett et al., 2021)
15
11.81
310.10
Cinnamoyl hexose
C15H18O7
+H
311.11
−0.4
Phenolic acid
128.06, 166.06, 167.06, 181.08, 247.09, 293.10, 311.11
(Farag et al., 2022)
16
11.99
666.17
Naringin 6′-malonate
C30H34O17
+H
667.18
−1.3
Flavonoid
85.03, 153.01, 255.08, 273.07, 315.09, 521.12
(Alam et al., 2022)
17
12.14
259.08
Skimmianine
C14H13NO4
+H
260.09
−0.5
Alkaloid
87.06, 102.94, 128.05, 156.07, 182.91, 200.92, 218.92, 242.08
(Chang et al., 2021)
18
12.31
544.12
Paeoniflorin sulfonate
C23H28O13S
+H
545.13
−4.3
Monoterpene
nil
(Basit et al., 2021)
19
12.34
254.05
Daidzein
C15H10O4
+H
255.06
−1.2
Flavonoid
nil
(Bragagnolo et al., 2021)
20
14.02
340.13
6-Prenylnaringenin
C20H20O5
+H
341.13
−0.8
Flavonoid
137.05, 187.07, 291.10, 323.12, 341.09
(Martinez and Davies 2015)
21
14.12
544.15
3,5-Diferuloylquinic acid
C27H28O12
+H
545.16
−1
Phenolic acid
85.03, 291.06, 307.09, 308.10, 324.10, 365.10, 383.11, 526.15, 545.16
(Clifford et al., 2006)
22
15.13
386.10
4-Caffeoylquinic acid
C20H18O8
+H
387.10
−0.3
Phenolic acid
135.04, 137.05, 148.55, 149.02, 208.94, 247.07, 275.06, 351.08
(Chen et al., 2021)
23
15.22
533.12
Pelargonidin 3-O-(6′'-succinyl-hexoside)
C25H25O13
+H
534.13
−3.8
Anthocyanin
nil
(Diretto et al., 2019)
24
15.35
330.07
Jaceosidin
C17H14O7
+H
331.08
0.1
Flavonoid
124.05, 139.07, 139.01, 212.04, 255.06, 313.07
(Cui et al., 2021)
25
16.45
496.15
Negundoside
C23H28O12
+H
497.16
−5
iridiod glycoside
120.95, 211.08, 220.96, 288.16, 309.05, 346.94, 399.18, 479.22, 496.16
(Meena et al., 2022)
26
16.83
344.07
5-O-Galloylquinic acid
C14H16O10
+H
345.08
−2.3
Tannin
191.21
(Maldini et al., 2011)
27
17.81
479.11
Petunidin 3-O-glycoside
C22 H23 O12
+H
480.12
1.9
Flavonoid
258.32, 274.04, 302.56, 317.61
(Downey and Rochfort 2008)
28
18.58
154.02
Gentisic acid
C7 H6 O4
+H
155.03
0
Phenolic acid
Nil
(Li et al., 2022)
29
18.88
493.13
Malvidin 3-O-hexoside
C23H25O12
+H
494.14
1.6
Anthocyanin
180.01, 243.23, 271.43
(Downey and Rochfort 2008)
30
19.78
402.13
Nobiletin
C21H 22O8
+H
403.13
−0.9
Flavonoid
Nil
(Ho and Kuo 2014)
31
21.26
270.05
Apigenin
C15H10O5
+H
271.05
−0.5
Flavonoid
121.02, 149.02, 271.06
(Basit et al., 2021)
32
21.45
331.08
Malvidin
C17H15O7
+H
332.08
1.2
Anthocyanidin
Nil
(Choi et al., 2010)
33
22.01
372.12
Tangeretin
C20H20O7
+H
373.12
−1
Flavonoid
Nil
(Alam et al., 2022)
34
22.02
342.11
Tetramethylkaempferol
C19H18O6
+H
343.11
−1
Flavonoid
Nil
(Ma et al., 2013)
35
22.49
305.12
Rohitukine
C16H19NO5
+H
306.13
−0.6
Alkaloid
176.03, 236.05, 306.13
(Jain et al., 2014)
36
23.57
228.07
Resveratrol
C14H12O3
+H
229.08
−1.1
Polyphenol
Nil
(Tisserant et al., 2016)
37
24.28
452.13
3-Hydroxyphloretin 2′-O-hexoside
C21H24O11
+H
453.14
1.9
Phenolic glucoside
Nil
(Leng et al., 2022)
38
28.93
370.10
Sesaminol
C12 H18 O7
+H
371.09
2.3
Lignan
Nil
(Mekky et al., 2021)
39
29.45
358.10
Gardenin B
C19H18O7
+H
359.11
4.5
Flavonoid
Nil
(Brito et al., 2021)
40
31.35
449.10
Cyanidin 3-O-hexoside
C21H21O11
+H
450.11
−0.9
Anthocyanin
Nil
(Mekky et al., 2021)
41
33.43
148.05
Cinnamic acid
C9H8O2
+H
149.05
0.1
Phenolic acid
Nil
(Mekky et al., 2021)
42
33.91
521.12
Petunidin 3-O-(6′'-acetyl-hexoside)
C24H25O13
+H
522.13
−0.1
Anthocyanin
Nil
(Chen et al., 2021)
43
33.98
561.12
Vitisin A
C26H25O14
+H
562.13
−3.8
Proanthocyanin
339
(Gordillo et al., 2012)
44
34.53
300.06
Hispidulin
C16H12O6
+H
301.07
2.4
Flavonoid
nil
(Cui et al., 2021)
45
35.54
535.14
Malvidin 3-O-(6′'-acetyl-hexoside)
C25H27O13
+H
536.15
2.8
Anthocyanin
331
(Talpur et al., 2017)
46
35.91
370.10
Sesaminol
C20H18O7
+H
371.11
2.7
Lignan
Nil
(Brigante et al., 2022)
47
36.97
164.04
p-coumaric acid
C9H8O3
+H
165.04
1.3
Polyphenol
163,159
(Cui et al., 2010)
48
37.59
276.21
Linolenic acid
C18H30O2
+H
277.21
3.7
Fatty acid
Nil
(Basit et al., 2021)
Peak no.
RT (min)
Mol. mass
Tentative identification
Mol. Formula
Adduct
B. peak (m/z)
Error ppm
Class
M/S product ions
Reference
49
1.01
290.07
Catechin
C15H14O6
-H
289.07
−1
Polyphenol
205.12, 246.43
(Brito et al., 2021)
50
2.64
295.06
Caffeoyl aspartic acid
C13H13NO7
-H
294.06
3.9
Phenolic acid
132.76, 175.34,
(Farag et al., 2016)
51
2.8
316.07
Protocatechuic acid 4-O-glucoside
C13H16O9
-H
315.07
−0.2
Phenolic glycoside
108.04, 109.03, 152.01, 153.03, 198.91, 216.91, 260.86, 278.87, 296.87, 315.07
(Basit et al., 2021)
52
2.86
519.11
Pelargonidin 3-O-(6′'-malonyl-hexoside)
C24H23O13
[M−2H]
517.10
−1.7
Anthocyanin glycoside
151.09, 470.74, 481,34
(Tian et al., 2017, Nowicka et al., 2019, Peniche-Pavía and Tiessen, 2020
53
5.23
342.09
Caffeic acid 4-O-hexoside
C15H18O9
-H
341.08
−0.2
Phenolic acid
89.02, 119.03, 134.04, 135.04, 161.04, 179.03, 248.88, 322.83, 341.10
(Dong et al., 2016, Zhang et al., 2016)
54
5.33
326.10
p-Coumaric acid 4-O-hexoside
C15H18O8
-H
325.09
0
Phenolic acid
119.05, 163.04, 238.89, 288.82, 306.84, 325.01
(Zhang et al., 2016, Qian et al., 2020)
55
6.12
356.11
Feruloyl hexose
C16H20O9
-H
355.10
−0.4
Hydroxycinnamic acid glycoside
149.06, 134.03, 178.02, 193.05, 300.86, 318.86, 355.09
(López-Velázquez et al., 2020)
56
6.93
431.09
Kaempferol 3-O-deoxyhexoside
C21H19O10
-H
430.09
−0.6
Flavonoid glycoside
255.07, 285.78
(Ning et al., 2019)
57
8.55
368.11
4-Feruloylquinic acid
C17H20O9
-H
367.10
−2.4
Quinic acid
155.31, 173.21, 193.08
(Cheiran et al., 2019)
58
8.98
374.10
Skullcapflavone-II
C19H18O8
-H
373.09
−2.2
Flavonoid
229.34, 257.29, 272.31, 285.43, 300.09, 328.41
(Zheng et al., 2012)
59
9.63
636.16
Kaempferol 3-O-(6′'-acetyl-glycoside) 7-O-deoxyhexoside
C29H32O16
-H
635.16
−0.6
Flavonoid
Nil
(Kitahiro et al., 2019)
60
9.88
208.07
Caffeic acid ethyl ester
C11H12O4
-H
207.06
4
Hydroxycinnamic acid
181.43, 199.06
(Sáenz-Navajas et al., 2010)
61
10.27
390.13
Polydatin
C20H22O8
-H
389.12
−1.8
Polyphenol
134.04, 143.05, 165.05, 178.02, 193.05, 257.11, 389.13
(Zhu et al., 2012)
62
10.36
624.16
Isorhamnetin 3-O-hexoside 7-O-deoxyhexoside
C28H32O16
-H
623.15
−4.4
Flavonoid
255.02, 299.07, 314.12, 315.41
(Lee and Shaari 2022)
63
11.01
488.13
6′'-O-Acetylglycitin
C24H24O11
-H
487.12
−3.8
Isoflavonoid
337.07, 372.77, 487.13
(Zhu et al., 2022)
64
11.24
610.18
Hesperidin
C28H34O15
-H
609.18
−2.6
Flavonone
301.07, 489.14, 609.18
(Hu et al., 2015)
65
11.48
597.14
Delphinidin-3-O-sambubioside
C26H29O16
[M−2H]
595.13
0
Anthocyanin flavonoid
66.98, 169.09, 549.76, 523.13,
(Tian et al., 2017, Majdoub et al., 2019)
66
12.04
420.14
Apigenin 7-O-hexoside
C21H24O9
-H
419.13
−1.6
Flavonoid
151.07, 269,78, 311.23
(Lee and Shaari 2022)
67
12.59
338.10
4-p-Coumaroylquinic acid
C16H18O8
-H
337.09
−1.3
Phenolic acid
96.97, 160.01, 161.05, 175.04, 198.90, 220.90, 272.83, 300.85, 337.09
(Kiselova-Kaneva et al., 2022)
68
12.65
772.20
Quercetin 3-O-hexosyl-deoxyhexosyl-hexoside
C33H40O21
-H
771.20
4.4
Flavonoid glycoside
Nil
(Kelebek 2016)
69
13.92
610.15
Rutin
C27H30O16
-H
609.14
−0.7
Flavonoid
Nil
(Ferracane et al., 2010)
70
14.12
564.14
Apigenin 7-O-apiosyl-hexoside
C26H28O14
-H
563.14
0.1
Flavonoid glycoside
193.05, 207.03, 369.08, 457.06, 563.23
(Tang et al., 2019)
71
14.21
930.27
1,2′-Disinapoyl-2-feruloylgentiobiose
C44H50O22
-H
929.26
−4.9
Phenolic acid
481.23, 499.08, 511.46, 529.03
(Lin et al., 2011)
72
15.46
474.07
Chicoric acid
C22H18O12
-H
473.07
−1.4
Phenylpropanoid
121.03, 225.05, 313.06, 401.08, 418.77, 454.82
(Khoza et al., 2016)
73
15.55
388.15
Trachelogenin
C21H24O7
-H
387.14
−3.5
Lignan
Nil
(Basit et al., 2021)
74
15.57
286.04
Kaempferol
C15H10O6
-H
285.04
0.9
Flavonoid
93.41, 117.23, 143.25, 227.68, 255,46, 268.73, 285.05
(Fathoni et al., 2017)
75
15.63
516.12
3,4-Dicaffeoylquinic acid
C25H24O12
-H
515.11
−1.5
Phenolic acid
293.08, 321.07, 424.79, 478.79
(Parejo et al., 2004)
76
16.63
550.16
Isoliquiritin apioside
C26H30O13
-H
549.16
−0.6
Flavonoid
119.044, 450.825, 549.159
(Basit et al., 2021)
77
19.71
356.12
Conidendrin
C20H20O6
-H
355.11
1.2
Lignan
Nil
(Eklund et al., 2004)
78
20.95
358.14
Matairesinol
C20H22O6
-H
357.13
−0.4
Lignan
Nil
(Dias et al., 2017)
79
21.23
406.12
Piceatannol 3-O-hexoside
C20H22O9
-H
405.11
−3.8
Stilbene
121.03, 249.03, 322.85, 337.15, 405.10
(Goufo et al., 2020)
80
23.56
423.26
Senbusine B
C23H37NO6
-H
422.25
−1.1
Diterpene alkaloid
Nil
(Jaiswal et al., 2013)
81
27.04
284.06
Oroxylin A
C16H12O5
-H
283.06
2.5
Flavonoid
240.23, 268.05
(Fong et al., 2014)
82
28.45
212.08
Pinosylvin
C14H12O2
-H
211.07
4.2
Stilbene
Nil
(Fu et al., 2019)
83
29.06
284.16
Stearic acid
C18H36O2
-H
283.09
1
Fatty acid
283.12
(Basit et al., 2021)
84
31.53
408.19
6-Geranylnaringenin
C25H28O5
-H
407.18
0.9
Flavonoid
183.32, 274.56, 339.24
(Basit et al., 2021)
85
33.03
342.34
Caffeic acid-O-hexoside
C15H18O9
-H
341.34
1.6
Phenolic glycoside
135.89, 179.56
(Álvarez-Fernández et al., 2015)
86
36.51
256.43
Palmitic acid
C16H32O2
-H
255,45
2.7
Fatty acid
255.12
(Basit et al., 2021)
87
38.61
299.05
Kaempferide
C16H11O6
-H
298.04
−2
Flavonoid
Nil
(Zehl et al., 2011)
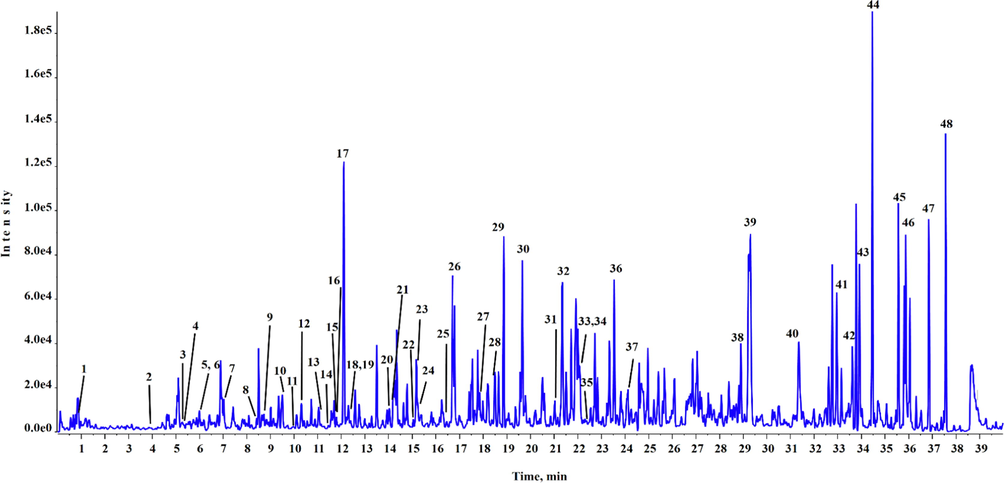
UPLC-Q-TOF-MS chromatogram of BEJv in positive mode of ionization.
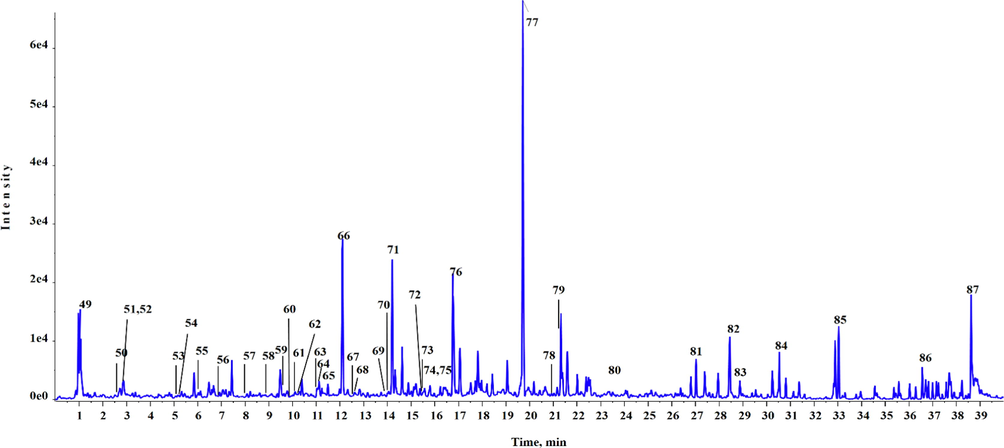
UPLC-Q-TOF-MS chromatogram of BEJv in negative mode of ionization.
Moreover, BEJv was subjected for further evaluation of polyphenolic quantification using HPLC-PDA analysis. The extract was tested for the presence of 23 phenolic compounds (as standards). Quantification of the compounds was done according to their retention time and confirmed by comparing their UV/Vis spectra to the standard compounds. The analysis revealed presence of 14 compounds in BEJv (Table 4). Among the quantified compounds kamepferol was found with highest quantity (3.45 ± 0.21 µg/mg DE) followed by ferrulic acid (2.78 ± 0.31 µg/mg DE), whereas, apigenin, vannilic acid and catechin were found below limit of detection (<0.01 µg/mg DE). Compounds p-hydroxy benzoic acid, o-coumaric acid, p-coumaric acid, 2,3-dimethoxy-benzoic acid, 3-hydroxy-4-dimethoxy-benzoic acid, 3-hydroxy benzoic acid, epicatechin, gallic acid, emodin were not detected in BEJv. The results of the quantification were expressed in µg/mg dry extract (DE). The yield of dry extract was 67 g. All the values are presented as mean ± SD., (n = 3). nd: not detected; BLD: below limit of detection (<0.1 µg/ g DE.); DE: dry extract.
S. No.
Standard name
Polyphenols quantified (µg/mg DE)
1
Kaempferol
3.45 ± 0.21
2
Syringic acid
0.45 ± 1.2
3
Caffeic acid
0.98 ± 0.03
4
Cinnamic acid
1.07 ± 0.26
5
Vannilic acid
BLD
6
Gentesic acid
1.87 ± 0.08
7
Rutin
BLD
8
Ferrulic acid
2.31 ± 1.30
9
Skullcapflavone
1.78 ± 0.27
10
Apigenin
BLD
11
Conidendrin
2.78 ± 0.31
12
Chlorogenic acid
1.65 ± 0.06
13
Naringenin
0.18 ± 0.05
14
Catechin
BLD
GC–MS is considered highly reliable and widely used spectroscopic technique for the analysis of volatile components in plant extracts (Vieira et al., 2020). To extend the phytocochemical evaluation of BEJv to volatile compounds GC–MS technique was applied in the present study. The analysis depicted tentative presence of 19 compounds (Table 5. and Fig. 3). The tentative identification of the compounds was carried out according to NIST library database search. Major class observed in the analysis was fatty acid and their derivatives. In detail methyl linolenate (Rt = 11.26 min) was found in highest quantity (3.69 %) followed by Piperonylcyanoacetic acid hydrazide (Rt = 29.40 min, 2.80 %), methyl linoleate (RT = 11.18 min, 2.14 %), methyl palmitate (Rt = 9.41 min, 1.64 %) and linoleic acid (Rt = 11.73 min, 1.16 %). A potent phenolic antioxidant sesamol (Zhou et al., 2021) was observed at (Rt = 4.83) with 0.17 % peak area. The good antioxidant and pharmacological potential of BEJv might be due to the presence of potent secondary metabolites found in GC–MS characterization of the extract. R.t: retention time; pct area: percent area.
Peak no.
RT (min)
Pct. Area
Compound name
Mol. Formula
Mol. Mass
Quality
1
4.83
0.67
Sesamol
C7H6O3
138.1
95
2
6.15
0.35
2,4-di-tert-butylphenol
C14H22O
206.3
97
3
8.02
0.53
Coniferol
C10H12O3
180.2
97
4
8.75
0.22
Phytone
C18H36O
268.5
90
5
9.41
1.64
Methyl palmitate
C17H34O2
270.5
99
6
9.72
0.87
Palmitic acid
C16H32O2
256.4
99
7
11.18
2.14
Methyl linoleate
C19H34O2
294.5
99
8
11.26
3.69
Methyl linolenate
C19H32O2
292.5
99
9
11.40
0.46
Phytol
C20H40O
296.5
91
10
11.52
0.24
Methyl stearate
C19H38O2
298.5
99
11
11.64
0.51
Linoleic acid
C18H32O2
280.4
99
12
11.73
1.16
Linolenic acid
C18H30O2
278.4
99
13
12.62
0.81
Heliotropine
C8H6O3
150.1
79
14
19.61
0.27
Di-n-2-propylpentylphthalate
C24H38O4
390.6
62
15
25.75
0.65
Cubebinolide
C20H18O6
354.4
83
16
27.69
1.16
2-ethylacridine
C15H13N
207.2
65
17
29.40
2.80
Piperonylcyanoacetic acid hydrazide
C11H11N3O3
233.2
60
18
30.50
0.76
4-Methyl-2-trimethylsilyloxy-acetophenone
C12H18O2Si
222.3
68
19
36.43
0.84
Diethyl bis(trimethylsilyl) silicate
C10H28O4Si3
296.5
70
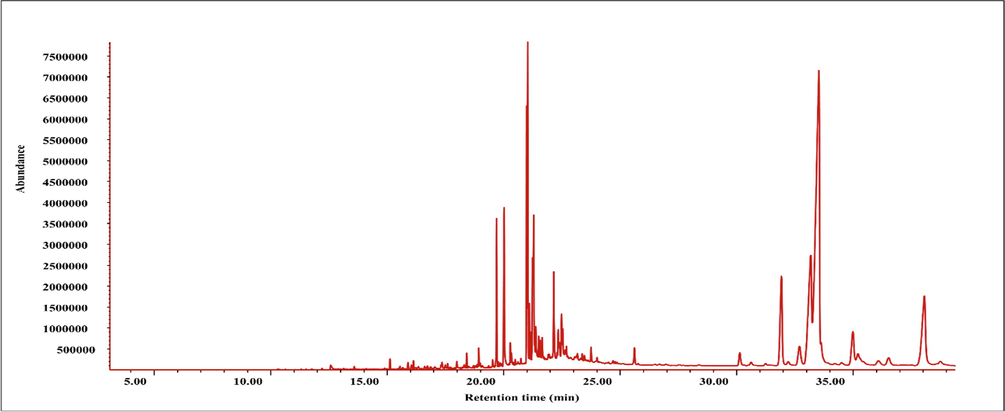
GC–MS chromatogram of BEJv.
3.2 Effect of BEJv on human normal and tumoral cell lines
Moreover, MTT assay was used to assess and evaluate the toxicity of BEJv against normal human liver cells (HL 7702), human liver cancer cells (HepG2) and human breast cancer cell (MCF-7) at various concentrations (25, 50, 100, 200 and 300 μg/mL). The biocompatibility and cell viability evaluation of BEJv were first time reported against the aforementioned cell lines. The extract showed no statistically different percent cell viability compared to the control group after 24 h and 48 h against human normal liver cells (Fig. 4A and B). These findings confirm the safety profile of BEJv with no deteriorating effect on the normal liver cells and also rectify the results of in vivo acute oral toxicity test. In case of liver cancer cells the extract showed moderate toxicity (p < 0.05) after 24 h at 200 and 300 μg/mL while lower cell viability was observed after 48 h at 300 μg/mL compared to control. Similarly BEJv showed moderate toxicity (p < 0.05) against breast cancer cell lines after 24 h and relatively higher toxicity (p < 0.01) at 100 μg/mL and p < 0.001 at 200 and 300 μg/mL after 48 h as compared to the control group. These findings suggest very low cytotoxicity effect of BEJv.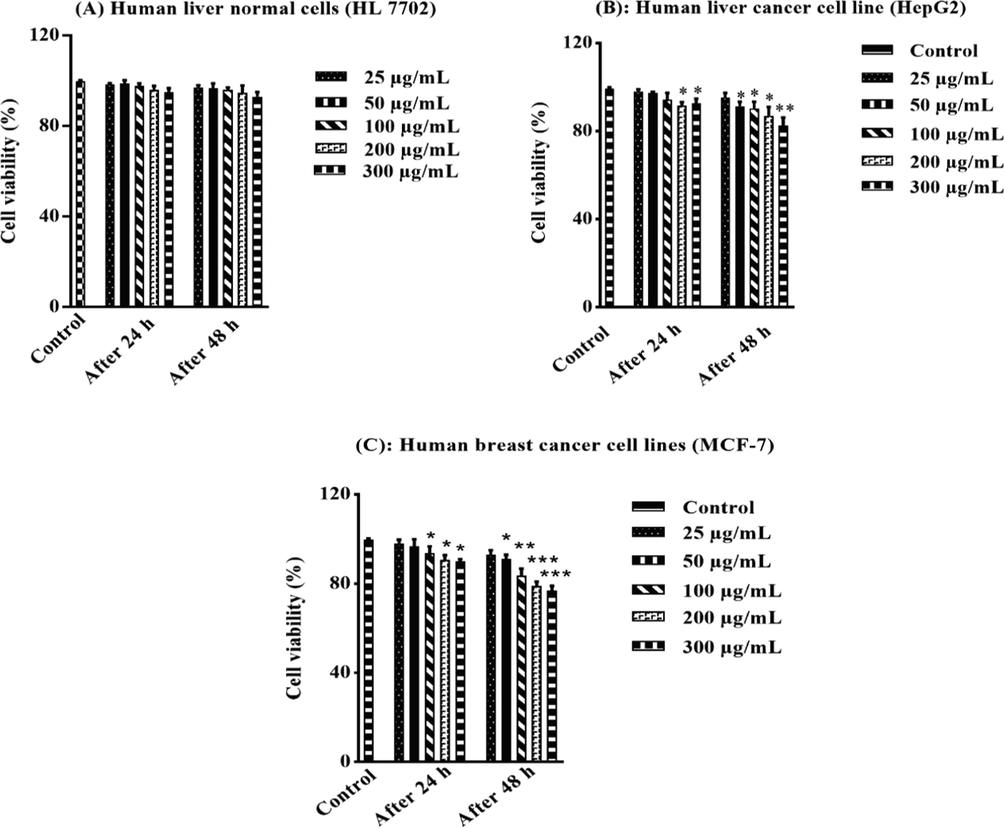
Cytotoxicity using MTT assay. Percent cell viability against human liver normal cells (HL 7702) after 24 h and 48 h (A), percent cell viability against human liver cancer cell line (HepG2) after 24 h and 48 h (B), percent cell viability against human breast cancer cell lines (MCF-7) after 24 h and 48 h (C). All the value are expressed as mean ± SD (n = 3). One-way ANOVA (with post hoc analysis- Tukey‟s multiple comparison test) was applied and the symbols used for significance were shown as *, ** and *** when p-value was<0.05, 0.01 and<0.001 respectively.
3.3 In vitro antioxidant
The involvement of free radicals in the etiology of various diseases is well understood. The free radicals induce toxic reactions such as peroxidation of the membrane lipids which leads to fragmentation and cross linking of macromolecules that causes cell death (Umamaheswari et al., 2008). Antioxidants scavenge the free radicals and minimize their toxic reactions in the pathogenesis of diseases (Fang et al., 2002). There are two categories of antioxidants; endogenous which include CAT, SOD and GSH and exogenous which include the bioactive contents such as flavonoids and phenols (Lee et al., 2004). Role of nature is immense in provision of antioxidant agents. In the present study the antioxidant potential of BEJv was studied using four different methods. BEJv showed highest power reducing activity in the FRAP assay (97.21 ± 2.34 mg TE/g DE) followed by CUPRAC (89.32 ± 3.21 mg TE/g DE), while in free radical scavenging assay BEJv showed higher results in DPPH (45.46 ± 1.8 mg TE/g DE) than ABTS (41.13 ± 1.3 mg TE/g DE) as shown in Table 6. The antioxidant activity may be due to the presence of flavonoids and phenols in the BEJv as previous studies correlated the antioxidant activity of plant extracts to the presence of phenols and flavonoids. The results also ratify the presence of functional phytoconstituents with antioxidant potential tentatively identified in the chemical profiling of BEJv (Santos et al., 2013, Ruankham et al., 2021). (Saleem et al., 2020). Some other species of genus Justicia have also been reported with good antioxidant potential (Mondal et al., 2019). All the values are expressed as mean ± SD (n = 3). TE: trolox equivalent; ACAE: acarbose equivalent; TUE: Thiourea equivalent; DE: Dry extract.
BEJv
DPPH mg. TE/g DE
45.46 ± 1.8
ABTS mg TE/g DE
41.13 ± 1.3
FRAP mg TE/g DE
97. 21 ± 2.34
CUPRAC mg TE/g DE
89.32 ± 3.21
α- glucosidase mmol ACAE/g DE
3.32 ± 0.31
β-amylase
mmol ACAE/g DE1.23 ± 0.6
Tyrosinase
mg KAE/g DE193.21 ± 0.76
Urease
mg TUE/g DE217.05 ± 4.5
3.4 In vitro enzyme inhibition
α-glucosidase and α-amylase inhibitors are used for the treatment of diabetes mellitus. Researchers are employing natural inhibitors of the enzymes for better outcomes in terms of promising results with minimal side effects as the synthetic sources of the enzyme inhibitors lack surety of efficacy and safety (Zengin et al., 2018). In in vitro enzyme inhibition, BEJv was tested to evaluate their inhibition potential against α-glucosidase and α-amylase (Table 6). The BEJv showed moderate to high inhibition potential against the tested enzymes. The BEJv showed comparatively higher inhibition of α-glucosidase (3.32 ± 0.31 mmol ACAE/g DE) compared to α-amylase inhibition (1.23 ± 0.6 mmol ACAE/g DE). The higher α-glucosidase activity of BEJv might be due to the presence of potent α-glucosidase inhibitors of phenol and flavonoids revealed in UPLC-Q-TOF-MS analysis of BEJv (Elmazar et al., 2013, Singh et al., 2021). The detailed in silico molecular docking revealed and confirmed the enzyme inhibition potential by docking three bioactives identified in UPLC-Q-TOF-MS analysis of BEJv.
Tyrosinase is an enzyme with therapeutic significance, involved in the melanogenesis process and its inhibitors prevent skin hyperpigmentation, a common cause of the skin darkening (Ciganović et al., 2019). In the current study the melanin reducing potential of BEJv was evaluated through tyrosinase inhibition. BEJv presented inhibition (193.21 ± 0.76 mg KAE/g DE) (Table 6). The in silico study showed significant docking score of the compounds against tyrosinase. The anti-tyrosinase potential of J. vahlii might be attributed to the presence of bioactives reported with good tyrosinase inhibition activities in literature (Farasat et al., 2020). The antityrosinase results proved the traditional use of J. vahlii in skin disorders.
Urease is the enzyme secreted by bacteria H.pylori in the human stomach, initiating and exaggerating the severity of peptic ulcer (Mobley 1996). In the current investigation BEJv showed significant inhibition (217.05 ± 4.5 mg TUE/g DE) of urease,. The phytochemical profiling has revealed the presence of phenols and flavonoids which were also confirmed by UPLC-Q-TOF-MS, which have a promising role in urease inhibition. The pyrogallol and catechol derivatives have been reported with H. pyloric urease inhibition (Xiao et al., 2010). Therefore, the results of the in vitro urease inhibition assay can be correlated to the diverse phytochemical profile of the species. Moreover, the three compounds skullcapeflavone, sesaminol and conidendrin showed good docking score against urease in in silico molecular docking study.
3.5 In vivo toxicity evaluation
The therapeutic applications of herbal drugs without scientific evidence may be associated with serious concerns regarding their toxicity profiles. Therefore, toxicity assessment of herbal drugs are carried out in different animal models to ensure their safety for human use (Liu et al., 2019). In the present study, oral administration of BEJv at the dose of 1000 mg/kg and 3000 mg/kg, exhibited no morbidity and mortality in mice compare to the control group. Although, there was a slight change observed in the color of stool at 3000 mg/kg. This change might be due to excretion of some ingredients of the extracts via stool. Biochemical and hematological parameters were found normal in both treated and control group. No gross lesions, distortion, deformation and toxicity related signs and symptoms in vital organs were observed after administration of BEJv for 14 days in comparison to the control group (Fig. 5 and Table 7 & 8). These findings suggest that BEJv is non-toxic, biocompatible, and the species is safe for use as food additive and therapeutic applications. All values are expressed as mean ± SD., (n = 3). *p < 0.05, **p < 0.01, ***p < 0.001 compared with control group. ANOVA (one way) followed by Tukey’s test. All values are expressed as mean ± SD., (n = 3).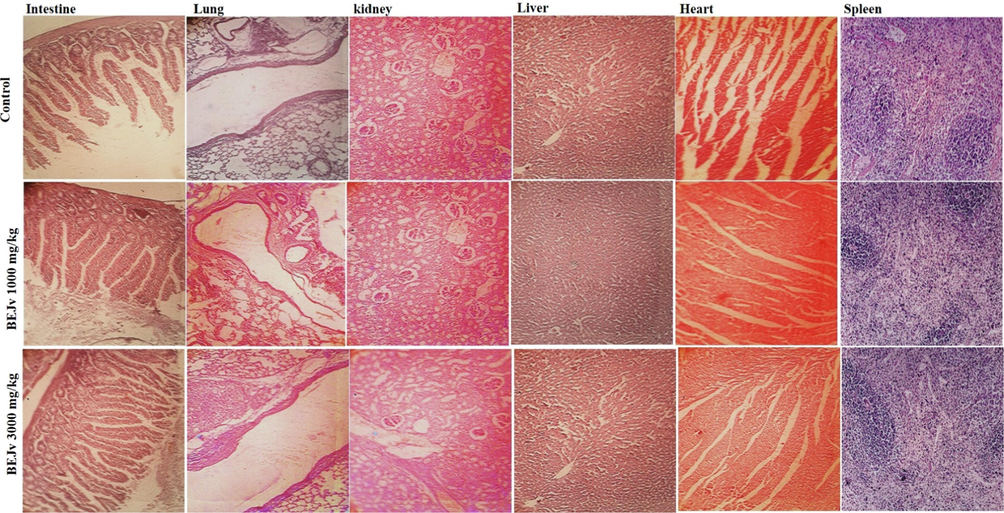
Representative photomicrographs of histopathological examination of vital organs of mice in acute oral toxicity assessment of BEJv at 1000 mg/kg and 3000 mg/kg.
Organ
Control
BEJv 3000 mg/kg
Spleen
0.201 ± 0.19
0.189 ± 0.18*
Kidney
1.450 ± 0.23
1.427 ± 0.17
Lung
0.636 ± 0.34
0.745 ± 0.13*
Heart
0.417 ± 0.10
0.432 ± 0.21
Liver
3.913 ± 0.67
3.941 ± 0.61
Biochemical parameter
Control group
Treated group (BEJv 3000 mg/kg)
Liver Function Tests
ALT (U/L)
16.34 ± 1.1
17.41 ± 3.1
AST (U/L)
74.31 ± 1.3
81.87 ± 1.9
ALP (U/L)
73.51 ± 2.3
75.61 ± 2.5
Hematological parameters
Hemoglobin (g/100 ml)
13.16 ± 2.1
13.1 ± 1.3
Total RBC (mL/cu. mm.)
7.35 ± 1.3
7.27 ± 1.9
PCV (%)
39.73 ± 1.2
40.63 ± 3.2
MCV (cu. micron)
51.03 ± 1.3
48.41 ± 1.3
MCH (Pico gram)
15.43 ± 1.03
15.90 ± 4.1
MCHC (%)
37.31 ± 2.2
36.42 ± 1.2
Total WBC (per cu. mm.)
4875 ± 1132
4989 ± 1076
Neutrophils (%)
31.21 ± 3.5
29.52 ± 4.5
Lymphocytes (%)
70.15 ± 3.7
71.65 ± 3.5
Eosinophils (%)
1.13 ± 0.19
1.23 ± 0.15
3.6 Effect of BEJv on carrageenan induced paw edema, oxidative stress parameters and inflammatory mediators
The carrageenan induced paw edema model is a widely employed preliminary model for evaluation of anti-inflammatory potential of plant extracts (Piva et al., 2021). The carrageenan induced edema occurs in two phases. The initial phase is predominantly non-phagocytic edema which lasts from 1 to 1.5 h followed by later phase from 2 to 6 h with increase edema induction. The first phase is characterized by excessive secretion of inflammatory mediators such as histamine and serotonin while in later phase, there is overproduction of cyclooxygenase takes place which cause an increase in the formation of prostaglandins and free radicals, initiating inflammation by further secretion of inflammatory mediators such as interleukins and cytokines (Ayal et al., 2019). In the current study, the subcutaneous administration of carrageenan (1 %) induced inflammation by increasing volume of mice paw in control group (Table 9). The results were in accordance with the previously published reports (Piva et al., 2021). The BEJv treated groups showed inhibition of the edema in dose dependent manner with relatively lower inhibition at 100 mg/kg and highest percent inhibition (p < 0.001) at 400 mg/kg compared to the control group. At the end of 0.5 h BEJv showed highest inhibition (19.43 %) at 400 mg/kg. Similarly at the end of 1 h the BEJv showed inhibition; 11.37 %, 17.53 % and 35.98 % (p < 0.05) at 100, 200 and 400 mg/kg respectively. After 2 h of carrageenan injection the inhibition showed by BEJv was 13.08 %, 30.37 % (p < 0.05) and 37.91 % (p < 0.01) at 100, 200 and 400 mg/kg. Significant inhibition was shown by BEJv 34.40 % (p < 0.05) at 200 mg/kg and 43.57 % (p < 0.01) at 400 mg/kg after 4 h compared to control group. Highest inhibition 52.01 % (p < 0.001) at 400 mg/kg was observed at 5 h. Animals treated with standard dexamethasone 5 mg/kg showed significant inhibition 28.06 % (p < 0.05), 49.28 % (p < 0.01), 56.54 % (p < 0.001), 72.59 % (p < 0.001) % and 73.99 % (p < 0.001) after 0.5, 1, 2, 4 and 5 h respectively compared to the control group (Fig. 6) which is consistent with the previous reports (Piva et al., 2021). The results showed that BEJv significantly inhibited edema in later phase. The later phase edema inhibition by BEJv might be due to the suppression of prostaglandins by inhibiting cyclooxygenase and so its related products. All values are expressed as mean ± SD., (n = 3). *p < 0.05, **p < 0.01, ***p < 0.001 compared with control group. ANOVA (one way) followed by post hoc analysis- Tukeys multiple comparison test.
The paw volume (mm), mean ± SD
Groups
Initial volume
0.5 h
1 h
2 h
4 h
5 h
Normal saline (1 ml/kg)
0.194 ± 0.01
0.196 ± 0.10
0.211 ± 0.02
0.214 ± 0.12
0.218 ± 0.003
0.223 ± 0.06
Dexa
0.173 ± 0.02
0.141 ± 0.02 *
0.107 ± 0.003 **
0.093 ± 0.01***
0.061 ± 0.15 ***
0.058 ± 0.23 ***
BEJv 100 mg/kg
0.187 ± 0.01
0.181 ± 0.01
0.187 ± 0.05
0.186 ± 0.13
0.173 ± 0.01
0.178 ± 0.03
BEJv 200 mg/kg
0.183 ± 0.01
0.182 ± 0.01
0.174 ± 0.03
0.149 ± 0.02 *
0.143 ± 0.02 *
0.129 ± 0.21 **
BEJv 400 mg/kg
0.193 ± 0.10
0.161 ± 0.03
0.137 ± 0.17 *
0.137 ± 0.21 **
0.123 ± 0.14 **
0.107 ± 0.14 ***
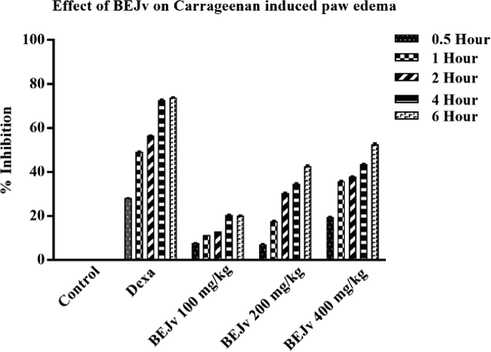
Percent inhibition of carrageenan induced paw edema by BEJv at 100, 200 and 400 mg/kg after 0.5, 1, 2, 4 and 6 h. All values are expressed as mean ± SD., (n = 3) *p < 0.05 compared with control group; ** p < 0.01, *** p < 0.001 compared with control group. ### p < 0.001 compared with normal saline group One-way ANOVA (with post hoc analysis- Tukey‟s multiple comparison test).
The prostaglandins initiate production of ROS and down regulate the oxidative stress markers, that further develop and maintain inflammation (Ansari et al., 2020, Ouda et al., 2021). Therefore, in the present study the effect of BEJv on oxidative stress markers; CAT, SOD and GSH was studied to know the possible anti-inflammatory mechanism of BEJv. The BEJv treated groups showed significant increase in CAT levels 45.31 ± 1.6 U/mg protein (p < 0.01) at 200 mg/kg and 46.67 ± 1.6 U/mg protein (p < 0.01) at 400 mg/kg. Similarly SOD and GSH levels were found higher in BEJv treated animals at 200 mg/kg (p < 0.01) and 400 mg/kg (p < 0.01) as compared to control group. The control group showed a decrease in CAT, SOD and GSH levels (Table 10) that are consistent with previous reports (Ammar et al., 2018). These findings suggest that the increasing effect on oxidative stress markers might be one of the possible mechanisms involved in the anti-inflammatory effect of BEJv. All values are expressed as mean ± SD., (n = 3) *p < 0.05 compared with control group; * p < 0.05, ** p < 0.01, *** p < 0.001 compared with control. One-way ANOVA (with post hoc analysis- Tukey‟s multiple comparison test) using graph pad prism 7.0 was applied.
Treatment and dose
CAT (U/mg protein)
SOD (U/mg protein)
GSH (nmol/mg protein)
BEJv
Carrageenan + Normal saline (10 ml/kg)
21.19 ± 2.1
22.17 ± 1.1
19.41 ± 2.5
Carrageenan + Dexa (75 mg/kg)
51.37 ± 2.4**
44.32 ± 1.2**
43.31 ± 1.9***
Carrageenan + BEJv (100 mg/kg)
37.64 ± 2.2
32.13 ± 2.1
31.23 ± 4.1*
Carrageenan + BEJv (200 mg/kg)
45.31 ± 1.6**
38.29 ± 3.1**
44.34 ± 1.9**
Carrageenan + BEJv (400 mg/kg)
46.67 ± 1.6**
39.13 ± 3.1**
44.84 ± 1.9**
The inflammatory mediators such as IL-1β and TNF-α play a key role in carrageenan induced edema by down regulating the levels of oxidative stress markers that contribute in the maintenance of inflammation (Ansari et al., 2020). In the present study, a significant increase in the levels of IL-1β and TNF-α was observed after carrageenan injection in the paw tissue of mice, these alterations are consistent with previous reports (Mansouri et al., 2015). BEJv treated groups showed a significant decrease in the levels of IL-1β at 200 mg/kg (p < 0.05) and 400 mg/kg (p < 0.001) while non-significant decrease was observed in mice treated with 100 mg/kg of BEJv compared to the control group (Fig. 7A). Similarly TNF-α levels were also decreased by BEJv at 200 mg/kg (p < 0.05) and 400 mg/kg (p < 0.01) compared to the control group. The standard dexamethasone (1 mg/kg) produced same but high yield effect compared to the control group (Fig. 7B). Conclusively it is suggested that the anti-inflammatory effect of BEJv might be due to increase in oxidative stress markers and decrease in inflammatory mediators. These findings support the traditional use of J. vahlii in pain and respiratory disorders.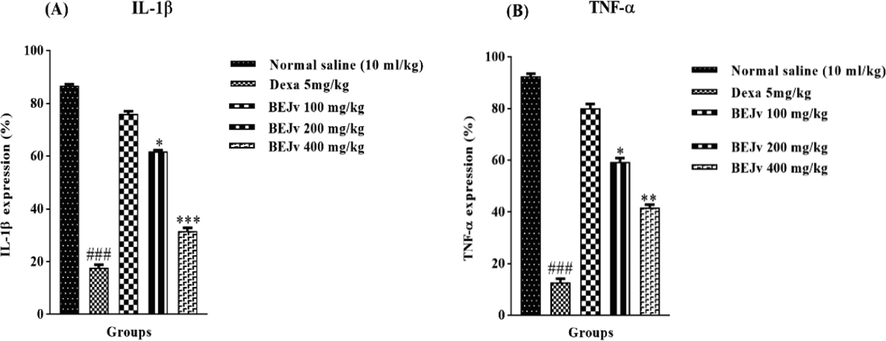
Effect of BEJv on; IL-1β (A) and TNF-α (B) expression. All the value are expressed as mean ± SD (n = 3). The software “GraphPad Prism-5” was used and one-way ANOVA (with post hoc analysis- Tukey’s multiple comparison test) was applied. *p < 0.05, **p < 0.01, ***p < 0.001 compared with standard. ### Significance p < 0.001 compared with normal saline group.
Various investigations revealed the role of phytoconstituents particularly flavonoids, phenols, alkaloids and lignans in attenuation of inflammation by demolishing ROS and free radicals (Ammar et al., 2018). The UPLC-Q-TOF-MS analysis of BEJv showed tentative identification of number of anti-inflammatory flavonoids such as Naringenin, Hesperetin, Daidzein, Jaceosidin, Apigenin, Hispidulin and Rutin and phenols such as Caffeic acid, Syringic acid and Chlorogenic acid as well as alkaloids and lignans which have been proved as significant anti-inflammatory agents in previous studies (Moon et al., 2006, Agrawal 2011, Conti et al., 2013). Various species of justicia were also reported with ant-inflammatory potential (Corrêa and Alcântara, 2012) that ratify our results. The current study validate, the traditional use of J. vahlii in pain, skin and respiratory disorders correlated with inflammation (Tandon et al., 2004, Babu et al., 2015, Asmat et al., 2016).
3.7 In silico studies
Recently molecular docking is considered as reliable and efficient tools used for prediction of binding free energy and correlation of experimental pharmacological activities. In the present study-three selected compounds as the major compounds were tested via in silico approaches against four enzymes namely α-amylase, α-glucosidase, tyrosinase and urease against which the extract and s were evaluated. The active site of the tested enzymes was detected and authenticated through docking the control ligands. Results of the docking are displayed in Table 11 and Fig. 8. The binding affinities of the compounds docked with three enzymes are tabulated in Table 11 with the predicted inhibition constants. More over details of the residual amino acids possibly involved in the interactions with the compounds at the active sites are also listed in the Table 11. Skullcapflavone has shown highest binding free energy (-8.2 kcal/mol) against urease in comparison to other enzymes studied. The inhibition might be due to high number of hydrogen bonds formed with Lys 445, Phe 441, Tyr 32 and Gln 471 at the active site of the enzyme as shown in Fig. 8. Similarly, sesaminol displayed highest binding free energy (-10.2 kcal/mol) against tyrosinase in comparison to other enzymes studied. Conidendrin showed highest results against α-amylase with binding energy of −8.3 kcal/mol. Hydrogen bonding and pi-pi interactions are observed as the dominant interactions between the compounds and the studied enzymes (Fig. 8). HB*: Hydrogen bonding.
Binding energy (kcal/mol)
Interaction site
Skullcapflavone-II
α-amylase
−7.2
Asp 197 (HB), Glu 233, Ala 198, Leu 162, Lys 200, Ile 235, His 201 (HB), Leu 165, Thr 163, Tyr 62, Gln 63, Trp 59, Trp 58, His 305 Arg 195
α-glucosidase
−8.1
Ser 311 (HB), Pro 312, Asp 307, Asp 242, His 280 (HB), Phe 303, Gln 353, Phe 159, Glu 411, Tyr 158 Arg 315, Phe 314, Thr 310
Urease
−8.2
Lys 445 (HB), Phe 441, Pro 446, Asn 447, Gln 459, Tyr 32 (HB), Val 473, Gln 471 (HB), Val 33, Ala 37, Ile 568
Tyrosinase
−7.8
Thr 69 (HB), Asn 439 (HB), Thr 448, Gln 437, Arg 97, Glu 451, Arg 114, Gly 107, Ser 106, Pro 445, Thr 98 (HB), His 100, Val 68
Sesaminol
α-amylase
−9.3
Tyr 62 (HB), His 101, Asp 197, Ala 198, Glu 233, Val 234, Lys 200, Leu 162, His 201, Ile 235, Asp 300, Trp 58, Thr 163, Trp 59, Gln 63 (HB), Leu 165
α-glucosidase
−9.6
Ser 298, Lys 13, Thr 290, Cys 341, Asp 341, Trp 343, Lys 16 (HB), Ala 292, His 295, Arg 270, Val 266, Asn 259
Urease
−8.4
Lys 445 (HB), Val 36, Phe 566, Tyr 32, Ala 150, Tyr 475
Tyrosinase
−10.2
Cys 101, His 100, Asn 102, Glu 66, Arg 64, Gly 63, Cys 99, Gly 107 (HB), Ser 106, Tyr 226, Pro 446 (HB)
Conidendrin
α-amylase
−8.3
Leu 162, Thr 163, Gln 63, Leu 165, Tyr 62, Trp 58, Asp 300 (HB), His 299, Asp 197, Arg 195, Glu 233, Ala 198, His 305 (HB)
α-glucosidase
−7.3
Asp 144, Ile 150, Pro 151, Phe 173, Phe 166 (HB), Thr 165, Ser 162, Arg 176, Gly 160, Trp 238, Lys 148
Urease
−7.7
Glu 159, Leu 215, His 216, Ser 228, Asp 229, Tyr 232, Asp 230, Val 233, Lys 212, Lys 211
Tyrosinase
−7.5
Glu 34, Pro 80, His 81, Ser 38, Arg 55, Ala 35, Met 40, Cys 42 (HB), Pro 43, Gln 29, Pro 51, Asp 82 (HB), His 75
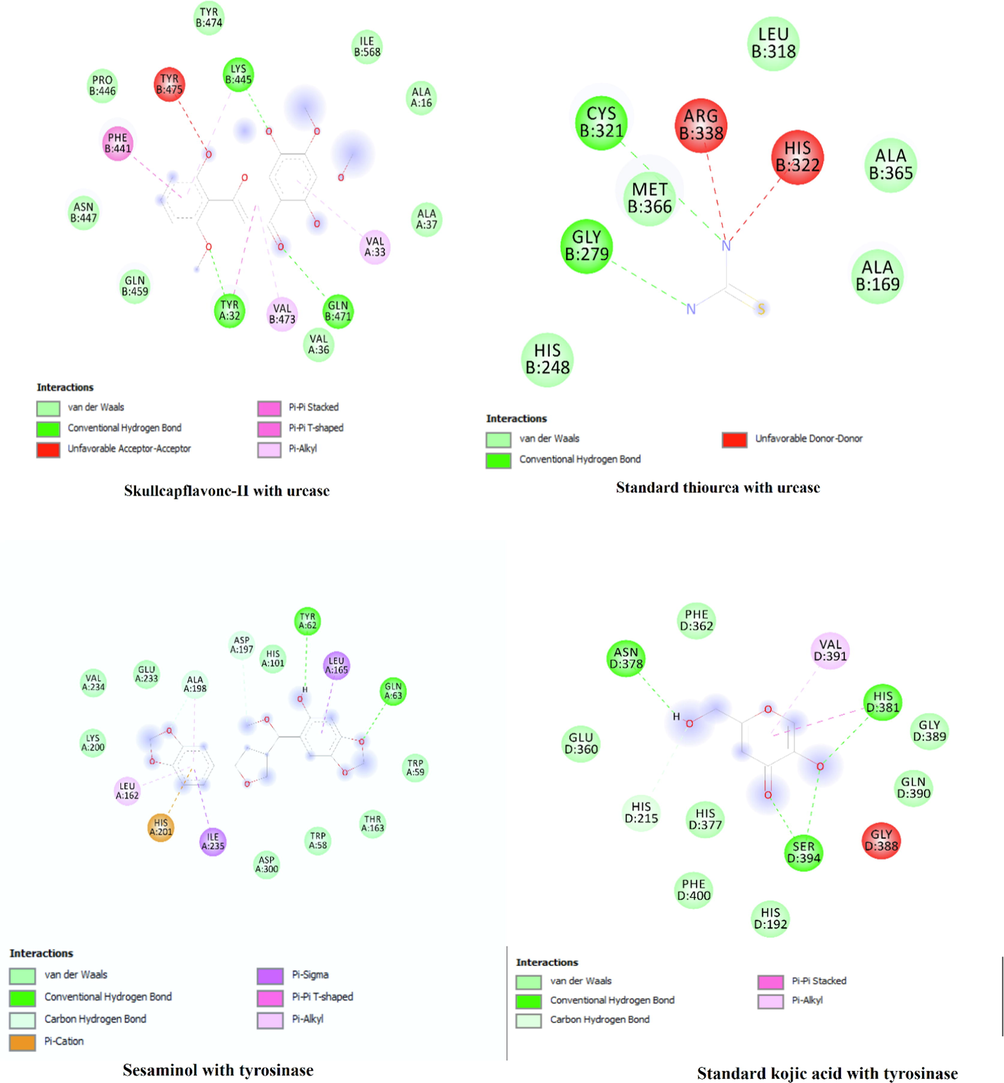
The docked compounds at the active site of the enzyme and their interactions.
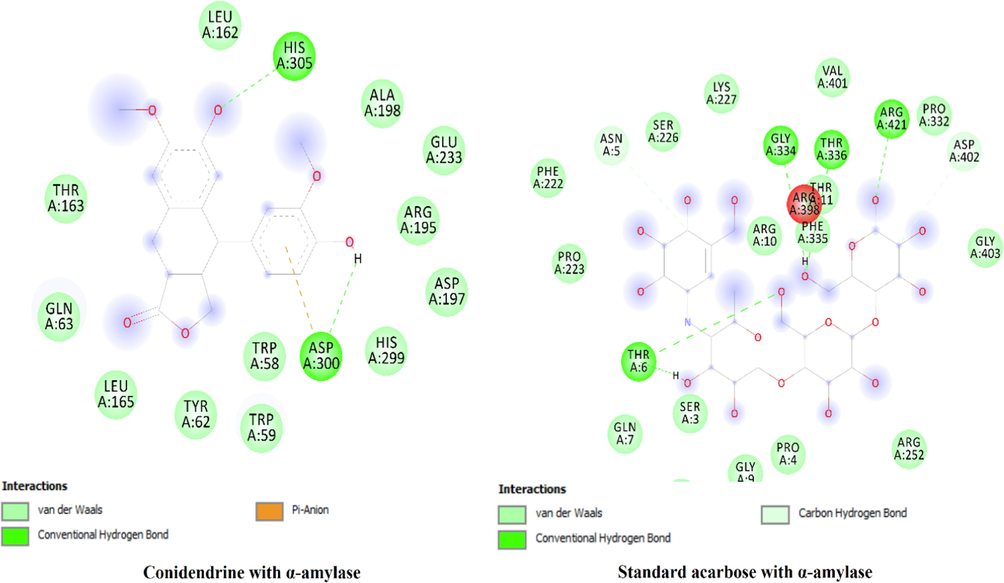
The docked compounds at the active site of the enzyme and their interactions.
4 Conclusion
The studies conducted suggested that BEJv presented an anti-inflammatory potential via modulating release of oxidative stress markers and inflammatory mediators which strongly support the traditional use of the species in inflammation and pain. Acute oral toxicity study and MTT assay suggest that the species is safe, non-toxic and biocompatible, can be consumed as food additive. The UPLC-Q-TOF-MS and GC–MS profiling revealed the tentative identification of functional bioactives with abundance of flavonoids and phenolic compounds in BEJv. The extract presented strong to moderate in vitro antioxidant and enzyme inhibition potential. Theoretically, docking study has confirmed the inhibition ability of the three selected compounds against α- amylase, α- glucosidase, tyrosinase and urease and their interactions with the active site are elucidated. Further studies can be done on the isolation of potentially bioactive compounds and sub-acute toxicity of the species.
Acknowledgements
The authors acknowledge Researchers Supporting Project number (RSP2022R504), King Saud University, Riyadh, Saudi Arabia and Department of Pharmaceutical Chemistry, Faculty of Pharmacy, The Islamia University of Bahawalpur, Punjab, Pakistan.
Funding: The authors are thankful to Researchers Supporting Project number (RSP2022R504), King Saud University, Riyadh, Saudi Arabia.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Achakzai, J.K., Anwar Panezai, M., Kakar, M.A., et al., 2019. In vitro anticancer MCF-7, anti-inflammatory, and brine shrimp lethality assay (BSLA) and GC-MS analysis of whole plant butanol fraction of Rheum ribes (WBFRR). 2019,
- Pharmacological activities of flavonoids: a review. Int. J. Pharm. Sci. Nanotechnol.. 2011;4:1394-1398.
- [Google Scholar]
- In vitro bioactivity of extracts from seeds of Cassia absus L. growing in Pakistan. J. Herb. Med.. 2019;16:1-5.
- [Google Scholar]
- Citrus flavonoids as potential therapeutic agents: A review. Phytother. Res.. 2022;36:1417-1441.
- [Google Scholar]
- Composition of nonanthocyanin polyphenols in alcoholic-fermented strawberry products using LC–MS (QTRAP), high-resolution MS (UHPLC-Orbitrap-MS), LC-DAD, and antioxidant activity. J. Agric. Food Chem.. 2015;63:2041-2051.
- [Google Scholar]
- Anti-inflammatory activity and phenolic composition of prickly pear (Opuntia ficus-indica) flowers. Ind. Crops. Prod.. 2018;112:313-319.
- [Google Scholar]
- Oxidative stress and inflammation in osteoarthritis pathogenesis: Role of polyphenols. Biomed. Pharmacother.. 2020;129:110452
- [Google Scholar]
- Diabetes mellitus and oxidative stress—A concise review. Saudi Pharm. J.. 2016;24:547-553.
- [Google Scholar]
- Evaluation of wound healing and anti-inflammatory activity of the leaves of Calpurnia aurea (Ait.) Benth (fabaceae) in mice. Wound Med.. 2019;25:100151
- [Google Scholar]
- Effect of curcumin in mice model of vincristine-induced neuropathy. Pharm. Biol.. 2015;53:838-848.
- [Google Scholar]
- In vitro antibacterial and antifungal activity of different solvent extracted samples of Alhagi maurorum. 2014;27:1955-1961.
- Basit, A., Ahmad, S., Naeem, A., et al., 2021. Chemical profiling of Justicia vahlii Roth (Acanthaceae), using UPLC-QTOF-MS and GC-MS analysis and evaluation of acute oral toxicity, antineuropathic and antioxidant activities. 114942.
- Justicia vahlii Roth. (Acanthaceae)-A new angiospermic record for Bangladesh. Bangladesh J. Bot.. 2001;30:73-74.
- [Google Scholar]
- Untargeted metabolomics of purple and orange-fleshed sweet potatoes reveals a large structural diversity of anthocyanins and flavonoids. Sci. Rep.. 2021;11:1-13.
- [Google Scholar]
- Metabolomics as a tool to study underused soy parts: In search of bioactive compounds. Foods.. 2021;10:1308.
- [Google Scholar]
- Comparative metabolite fingerprinting of chia, flax and sesame seeds using LC-MS untargeted metabolomics. Food Chem.. 2022;371:131355
- [Google Scholar]
- Antimicrobial, antioxidant, volatile and phenolic profiles of cabbage-stalk and pineapple-crown flour revealed by GC-MS and UPLC-MSE. Food Chem.. 2021;339:1-11.
- [Google Scholar]
- Isolation and identification of antifungal and antialgal alkaloids from Haplophyllum sieversii.. 2005;53:7741-7748.
- Identification and characterization of quinoline alkaloids from the root bark of Dictamnus dasycarpus and their metabolites in rat plasma, urine and feces by UPLC/Qtrap-MS and UPLC/Q-TOF-MS. J. Pharm. Biomed. Anal.. 2021;204:114229
- [Google Scholar]
- Characterization, antioxidant, antimicrobial and cytotoxic activities of green synthesized silver and iron nanoparticles using alcoholic Blumea eriantha DC plant extract. Mater. Today Commun.. 2020;24:101320
- [Google Scholar]
- Simultaneous identification of low-molecular weight phenolic and nitrogen compounds in craft beers by HPLC-ESI-MS/MS. Food Chem.. 2019;286:113-122.
- [Google Scholar]
- Identification of phenolic compounds in Australian grown dragon fruits by LC-ESI-QTOF-MS/MS and determination of their antioxidant potential. Arabian J. Chem.. 2021;14:103151
- [Google Scholar]
- Analysis and tentative structure elucidation of new anthocyanins in fruit peel of Vitis coignetiae Pulliat (meoru) using LC-MS/MS: Contribution to the overall antioxidant activity. J. Sep. Sci.. 2010;33:1192-1197.
- [Google Scholar]
- Glycerolic licorice extracts as active cosmeceutical ingredients: Extraction optimization, chemical characterization, and biological activity. Antioxidants.. 2019;8:445.
- [Google Scholar]
- Characterization by LC-MS n of four new classes of chlorogenic acids in green coffee beans: dimethoxycinnamoylquinic acids, diferuloylquinic acids, caffeoyl-dimethoxycinnamoylquinic acids, and feruloyl-dimethoxycinnamoylquinic acids. J. Agric. Food Chem.. 2006;54:1957-1969.
- [Google Scholar]
- Profiling the chlorogenic acids and other caffeic acid derivatives of herbal Chrysanthemum by LC− MS n. J. Agric. Food Chem.. 2007;55:929-936.
- [Google Scholar]
- Comparison of beneficial actions of non-steroidal anti-inflammatory drugs to flavonoids. J. Biol. Regul. Homeost. Agents.. 2013;27:1-7.
- [Google Scholar]
- Chemical constituents and biological activities of species of Justicia: a review. Rev. Bras. Farmacogn.. 2012;22:220-238.
- [Google Scholar]
- Rapid Identification of Chemical Constituents in Artemisia argyi Lévi et Vant by UPLC-Q-Exactive-MS/MS. J. Food Qual. 2021;2021
- [Google Scholar]
- LC− MS determination and pharmacokinetics of p-coumaric acid in rat plasma after oral administration of p-coumaric acid and freeze-dried red wine. J. Agric. Food Chem.. 2010;58:12083-12088.
- [Google Scholar]
- In vitro schistosomicidal and antiviral activities of Arctium lappa L. (Asteraceae) against Schistosoma mansoni and Herpes simplex virus-1. Biomed. Pharmacother.. 2017;94:489-498.
- [Google Scholar]
- Differential accumulation of pelargonidin glycosides in petals at three different developmental stages of the orange-flowered gentian (Gentiana lutea L. var. aurantiaca) PLoS ONE. 2019;14:e0212062
- [Google Scholar]
- Biomarkers of oxidative stress and heavy metal levels as indicators of environmental pollution in some selected fishes in Lagos, Nigeria. Am. Eurasian J. Agric. Environ. Sci.. 2010;7:359-365.
- [Google Scholar]
- Identification of compounds in an anti-fibrosis Chinese medicine (Fufang Biejia Ruangan Pill) and its absorbed components in rat biofluids and liver by UPLC-MS. J. Chromatogr. B. 2016;1026:145-151.
- [Google Scholar]
- Simultaneous separation by reversed-phase high-performance liquid chromatography and mass spectral identification of anthocyanins and flavonols in Shiraz grape skin. J. Chromatogr. Add.. 2008;1201:43-47.
- [Google Scholar]
- Reactions of the natural lignan hydroxymatairesinol in basic and acidic nucleophilic media: formation and reactivity of a quinone methide intermediate. Org. Biomol. Chem.. 2004;2:2229-2235.
- [Google Scholar]
- Phytol/Phytanic acid and insulin resistance: potential role of phytanic acid proven by docking simulation and modulation of biochemical alterations. PLoS ONE. 2013;8:e45638
- [Google Scholar]
- Metabolomics driven analysis of Erythrina lysistemon cell suspension culture in response to methyl jasmonate elicitation. J. Adv. Res.. 2016;7:681-689.
- [Google Scholar]
- Comparative Metabolite Fingerprinting of Four Different Cinnamon Species Analyzed via UPLC–MS and GC–MS and Chemometric Tools. Molecules. 2022;27:2935.
- [Google Scholar]
- In silico assessment of the inhibitory effect of four flavonoids (Chrysin, Naringin, Quercetin, Kaempferol) on tyrosinase activity using the MD simulation approach. BioTechnologia.. 2020;101:193-204.
- [Google Scholar]
- Biomarkers of oxidative stress and heavy metal levels as indicators of environmental pollution in African cat fish (Clarias gariepinus) from Nigeria Ogun River. Int. J. Environ. Res. Public Health.. 2007;4:158-165.
- [Google Scholar]
- Ethnomedicinal knowledge of the rural communities of Dhirkot, Azad Jammu and Kashmir. Pakistan. J. Ethnobiol. Ethnomed.. 2019;15:1-30.
- [Google Scholar]
- Identification of nonvolatile compounds in clove (Syzygium aromaticum) In: From Manado. AIP Conference Proceedings, AIP Publishing LLC. 2017.
- [Google Scholar]
- Metabolic profile of the bioactive compounds of burdock (Arctium lappa) seeds, roots and leaves. J. Pharm. Biomed. Anal.. 2010;51:399-404.
- [Google Scholar]
- Identification and quantification of phenolic compounds from pomegranate (Punica granatum L.) peel, mesocarp, aril and differently produced juices by HPLC-DAD–ESI/MSn. Food Chem.. 2011;127:807-821.
- [Google Scholar]
- Development of a SPE-LC/MS/MS method for simultaneous quantification of baicalein, wogonin, oroxylin A and their glucuronides baicalin, wogonoside and oroxyloside in rats and its application to brain uptake and plasma pharmacokinetic studies. J. Pharm. Biomed. Anal.. 2014;97:9-23.
- [Google Scholar]
- Screening, and identification of the binding position, of xanthine oxidase inhibitors in the roots of Lindera reflexa Hemsl using ultrafiltration LC–MS combined with enzyme blocking. Biomed. Chromatogr.. 2019;33:e4577
- [Google Scholar]
- Mitochondrial superoxide dismutase SOD2, but not cytosolic SOD1, plays a critical role in protection against glutamate-induced oxidative stress and cell death in HT22 neuronal cells. Free Radic. Biol. Med.. 2010;48:821-830.
- [Google Scholar]
- Application of LC–MS and tristimulus colorimetry to assess the ageing aptitude of Syrah wine in the Condado de Huelva DO (Spain), a typical warm climate region. Anal. Chim. Acta. 2012;732:162-171.
- [Google Scholar]
- A reference list of phenolic compounds (including stilbenes) in grapevine (Vitis vinifera L.) roots, woods, canes, stems, and leaves. Antioxidants. 2020;9:398.
- [Google Scholar]
- Biological potential and GC-MS analysis of phytochemicals of Farsetia hamiltonii (Royle) Biomed. Res.. 2019;30
- [Google Scholar]
- Hesperidin, nobiletin, and tangeretin are collectively responsible for the anti-neuroinflammatory capacity of tangerine peel (Citri reticulatae pericarpium) Food Chem. Toxicol.. 2014;71:176-182.
- [Google Scholar]
- Simultaneous determination of ten compounds in rat plasma by UPLC-MS/MS: application in the pharmacokinetic study of Ma-Zi-Ren-Wan. J. Chromatogr. B. 2015;1000:136-146.
- [Google Scholar]
- Identifying the chemical markers in raw and wine-processed Scutellaria baicalensis by ultra-performance liquid chromatography/quadrupole time-of-flight mass spectrometry coupled with multiple statistical strategies. Biomed. Chromatogr.. 2020;34:1-14.
- [Google Scholar]
- Ethnobotanical study of medicinal plants used by Paliyar tribals in Theni district of Tamil Nadu. India. Fitoterapia.. 2008;79:562-568.
- [Google Scholar]
- ZINC: a free tool to discover chemistry for biology. J Chem Inf Model.. 2012;52:1757-1768.
- [Google Scholar]
- Dysoxylum binectariferum bark as a new source of anticancer drug camptothecin: bioactivity-guided isolation and LCMS-based quantification. Bioorg. Med. Chem. Lett.. 2014;24:3146-3149.
- [Google Scholar]
- A comparative study on the traditional Indian Shodhana and Chinese processing methods for aconite roots by characterization and determination of the major components. Chem. Cent. J.. 2013;7:1-16.
- [Google Scholar]
- Assessment of antioxidant potential, total phenolics and flavonoids of different solvent fractions of Monotheca buxifolia fruit. Osong Public Health Res. Perspect.. 2013;4:246-254.
- [Google Scholar]
- Melanin-decolorizing activity of antioxidant enzymes, glutathione peroxidase, thiol peroxidase, and catalase. Mol. Biotechnol.. 2021;63:150-155.
- [Google Scholar]
- Antioxidant potential, total phenolic and total flavonoid contents of Rhododendron anthopogonoides and its protective effect on hypoxia-induced injury in PC12 cells. BMC Complement. Altern. Med.. 2015;15:1-12.
- [Google Scholar]
- A modified spectrophotometric assay of superoxide dismutase. Indian J. Biochem. Biophys.. 1984;21:130-132.
- [Google Scholar]
- LC-DAD–ESI-MS/MS characterization of phenolic constituents in Turkish black tea: Effect of infusion time and temperature. Food Chem.. 2016;204:227-238.
- [Google Scholar]
- Antinociceptive properties of 25-methoxy hispidol A, a triterpinoid isolated from Poncirus trifoliata (Rutaceae) through inhibition of NF-κB signalling in mice. Phytother. Res.. 2019;33:327-341.
- [Google Scholar]
- Identification of hydroxylcinnamoyl tartaric acid esters in Bidens pilosa by UPLC-tandem mass spectrometry. S. Afr. J. Bot.. 2016;103:95-100.
- [Google Scholar]
- Phytochemical composition and in vitro pharmacological investigations of Neurada procumbens L. (Neuradaceae): A multidirectional approach for industrial products. Ind. Crops Prod.. 2019;142:111861
- [Google Scholar]
- High resolution LC-MS/MS characterization of polyphenolic composition and evaluation of antioxidant activity of Sambucus ebulus fruit tea traditionally used in Bulgaria as a functional food. Food Chem.. 2022;367:130759
- [Google Scholar]
- Phytochemical characterization of Rosa multiflora Thunb. (Rosaceae) in Japan and South Korea, with a focus on the bioactive flavonol glycoside ‘multiflorin A’. J. Nat. Med.. 2019;73:555-565.
- [Google Scholar]
- Reactive oxygen species, aging, and antioxidative nutraceuticals. Compr. Rev. Food Sci. Food Saf.. 2004;3:21-33.
- [Google Scholar]
- LC–MS metabolomics analysis of Stevia rebaudiana Bertoni leaves cultivated in Malaysia in relation to different developmental stages. Phytochem. Anal.. 2022;33:249-261.
- [Google Scholar]
- Identification of Phenolic Compounds in Australian-Grown Bell Peppers by Liquid Chromatography Coupled with Electrospray Ionization-Quadrupole-Time-of-Flight-Mass Spectrometry and Estimation of Their Antioxidant Potential. ACS Omega. 2022;7:4563-4576.
- [CrossRef] [Google Scholar]
- Comparing the LC-MS Phenolic Acids Profiles of Seven Different Varieties of Brown Rice (Oryza sativa L.) Foods.. 2022;11:1552.
- [Google Scholar]
- UHPLC-PDA-ESI/HRMS/MS n analysis of anthocyanins, flavonol glycosides, and hydroxycinnamic acid derivatives in red mustard greens (Brassica juncea Coss variety) J. Agric. Food. Chem.. 2011;59:12059-12072.
- [Google Scholar]
- Butanol extraction to predict bioavailability of PAHs in soil.. 2002;46:1011-1017.
- Identification of β-carboline and canthinone alkaloids as anti-inflammatory agents but with different inhibitory profile on the expression of iNOS and COX-2 in lipopolysaccharide-activated RAW 264.7 macrophages. J. Nat. Med.. 2019;73:124-130.
- [Google Scholar]
- Adherence to ARRIVE guidelines in Chinese journal reports on neoplasms in animals. PLoS ONE. 2016;11:e0154657
- [Google Scholar]
- Identification and quantification analysis of the chemical constituents from Mahonia fortune using Q-Exactive HF Mass Spectrometer and UPLC–ESI-MS/MS. J. Pharm. Biomed. Anal.. 2021;196:113903
- [Google Scholar]
- Vincristine-induced peripheral neuropathy: a mini-review. Neurotoxicology.. 2020;161–171
- [Google Scholar]
- Multicomponent pattern and biological activities of seven Asphodeline taxa: potential sources of natural-functional ingredients for bioactive formulations. J. Enzyme. Inhib. Med. Chem.. 2017;32:60-67.
- [Google Scholar]
- Phenolic profile associated with chilling tolerance induced by the application of a hot water treatment in bell pepper fruit. J. Food Sci.. 2020;85:2080-2089.
- [Google Scholar]
- Identification of regioisomers of methylated kaempferol and quercetin by ultra high performance liquid chromatography quadrupole time-of-flight (UHPLC–QTOF) tandem mass spectrometry combined with diagnostic fragmentation pattern analysis. Anal. Chim. Acta. 2013;795:15-24.
- [Google Scholar]
- Medicinal plant resources of Lankamalleswara wild life sanctuary, eastern Ghats, Andhra Pradesh. Vegetos.. 2012;25:94-101.
- [Google Scholar]
- Evaluation of the availability of delphinidin and cyanidin-3-O-sambubioside from Hibiscus sabdariffa and 6-gingerol from Zingiber officinale in colon using liquid chromatography and mass spectrometry detection. Eur. Food Res. Technol.. 2019;245:2425-2433.
- [Google Scholar]
- Phenolic compounds from Byrsonima crassifolia L. bark: Phytochemical investigation and quantitative analysis by LC-ESI MS/MS. J. Pharm. Biomed. Anal.. 2011;56:1-6.
- [Google Scholar]
- Anti-inflammatory effects of flavonoids.. 2019;299:125124
- A study of the mechanisms underlying the anti-inflammatory effect of ellagic acid in carrageenan-induced paw edema in rats. Indian J. Pharmacol.. 2015;47:292.
- [Google Scholar]
- Stereospecific quantitation of 6-prenylnaringenin in commercially available H. lupulus-containing natural health products and dietary supplements. Res. Pharm. Sci.. 2015;10:182.
- [Google Scholar]
- Studies on physicochemical, phytochemicals, chromatographic profiling & estimation and in-silico study of Negundoside in roots & small branches of Vitex Negundo plant. Phytomedicine Plus.. 2022;2:100205
- [Google Scholar]
- Metabolic profiling of the oil of sesame of the Egyptian cultivar ‘Giza 32’employing LC-MS and tandem MS-based untargeted method. Foods.. 2021;10:298.
- [Google Scholar]
- The role of Helicobacter pylori urease in the pathogenesis of gastritis and peptic ulceration. Aliment. Pharmacol. Ther.. 1996;10:57-64.
- [Google Scholar]
- Hepatoprotective and antioxidant activities of Justicia gendarussa leaf extract in carbofuran-induced hepatic damage in rats. Chem. Res. Toxicol.. 2019;32:2499-2508.
- [Google Scholar]
- Dietary flavonoids: effects on xenobiotic and carcinogen metabolism. Toxicolo. in vitro.. 2006;20:187-210.
- [Google Scholar]
- Simultaneous UPLC–TQ-MS/MS determination of six active components in rat plasma: application in the pharmacokinetic study of Cyclocarya paliurus leaves. Chin. Med.. 2019;14:1-11.
- [Google Scholar]
- Comparison of polyphenol content and antioxidant capacity of strawberry fruit from 90 cultivars of Fragaria× ananassa Duch. Food Chem.. 2019;270:32-46.
- [Google Scholar]
- OECD, 1994. OECD Guidelines for the Testing of Chemicals, Organization for Economic.
- In vivo Anti-inflammatory Activity of Aqueous Extract of Carthamus caeruleus L Rhizome Against Carrageenan-Induced Inflammation in Mice. Jordan J. Biol. Sci.. 2021;14:529-535.
- [Google Scholar]
- Separation and characterization of phenolic compounds in fennel (Foeniculum vulgare) using liquid chromatography− negative electrospray ionization tandem mass spectrometry. J. Agric. Food. Chem.. 2004;52:3679-3687.
- [Google Scholar]
- Anthocyanin profiling of maize grains using DIESI-MSQD reveals that cyanidin-based derivatives predominate in purple corn, whereas pelargonidin-based molecules occur in red-pink varieties from Mexico. J. Agric. Food Chem.. 2020;68:5980-5994.
- [Google Scholar]
- Multidirectional insights into the phytochemical, biological, and multivariate analysis of the famine food plant (Calligonum polygonoides L).: A novel source of bioactive phytocompounds. Food Res. Int.. 2020;137:1-7.
- [Google Scholar]
- Antioxidant, Anti-Inflammatory, and Anti-Diabetic Activity of Phenolic Acids Fractions Obtained from Aerva lanata (L.) Juss. Molecules.. 2021;26:3486.
- [Google Scholar]
- Anti-inflammatory activity and chemical composition of aqueous extract and essential oil from leaves of Ocimum selloi Benth. J. Ethnopharmacol.. 2021;275:114136
- [Google Scholar]
- Comprehensive identification of minor components and bioassay-guided isolation of an unusual antioxidant from Azolla imbricata using ultra-high performance liquid chromatography—quadrupole time-of-flight mass spectrometry combined with multicomponent knockout and bioactivity evaluation. J. Chromatogr. A. 2020;1609:460435
- [Google Scholar]
- Identification, method development, validation, and characterization of Aza sugars by an ion-chromatography, high-resolution mass spectrometer. and LC-MS/MS.. 2018;9:1-16.
- [Google Scholar]
- Sesamin and sesamol attenuate H2O2-induced oxidative stress on human neuronal cells via the SIRT1-SIRT3-FOXO3a signaling pathway. Nutr. Neurosci.. 2021;24:90-101.
- [Google Scholar]
- Characterization of taste-active fractions in red wine combining HPLC fractionation, sensory analysis and ultra performance liquid chromatography coupled with mass spectrometry detection. Anal. Chim. Acta. 2010;673:151-159.
- [Google Scholar]
- UHPLC-MS phytochemical profiling, biological propensities and in-silico studies of Alhagi maurorum roots: a medicinal herb with multifunctional properties. Drug. Dev. Ind. Pharm.. 2020;46:861-868.
- [Google Scholar]
- Antinociceptive and antioxidant activities of phytol in vivo and in vitro models. Neuroscience. 2013;2013:1-9.
- [Google Scholar]
- Nigella damascena L. essential oil and its main constituents, damascenine and β-elemene modulate inflammatory response of human neutrophils ex vivo. Food Chem. Toxicol.. 2019;125:161-169.
- [Google Scholar]
- Phytochemical and Pharmacological Insight on Sesamol: An Updated Review. Curr. Bioact. Compd.. 2021;17:112-119.
- [Google Scholar]
- Acute oral toxicity and brine shrimp lethality of Elaeis guineensis Jacq., (oil palm leaf) methanol extract. Molecules. 2010;15:8111-8121.
- [Google Scholar]
- Analysis and characterization of anthocyanin from phalsa (Grewia asiatica) MOJ Food Process Technol.. 2017;5:00127.
- [Google Scholar]
- Oxidative stress and antioxidants status in peptic ulcer and gastric carcinoma. J. Physiol. Pharmacol.. 2004;48:115-118.
- [Google Scholar]
- Lc-esi-qtof/ms characterization of phenolic compounds from medicinal plants (hops and juniper berries) and their antioxidant activity. Foods.. 2019;9:7.
- [Google Scholar]
- Phenolic compounds extracted by acidic aqueous ethanol from berries and leaves of different berry plants. Food Chem.. 2017;220:266-281.
- [Google Scholar]
- 13C NMR and LC-MS Profiling of stilbenes from elicited grapevine hairy root cultures. J. Nat. Prod.. 2016;79:2846-2855.
- [Google Scholar]
- Tyrosinase inhibition and anti-melanin generation effect of cinnamamide analogues. Bioorg. Chem.. 2019;87:43-55.
- [Google Scholar]
- In vitro antioxidant activities of the fractions of Coccinia grandis L. leaf extract. Afr. J. Tradit. Complement. Altern. Med.. 2008;5:61-73.
- [Google Scholar]
- Novel in vitro and in silico insights of the multi-biological activities and chemical composition of Bidens tripartita L. Food Chem. Toxicol.. 2018;111:525-536.
- [Google Scholar]
- Rapid qualitative profiling of metabolites present in Fusarium solani, a rhizospheric fungus derived from Senna spectabilis, using GC/MS and UPLC-QTOF/MSE techniques assisted by UNIFI information system. Eur. J. Mass Spectrom.. 2020;26:281-291.
- [Google Scholar]
- Simultaneous determination of caffeic acid and its major pharmacologically active metabolites in rat plasma by LC-MS/MS and its application in pharmacokinetic study. Biomed. Chromatogr.. 2015;29:552-559.
- [Google Scholar]
- Phenol-hypochlorite reaction for determination of ammonia. Anal. Chem.. 1967;39:971-974.
- [Google Scholar]
- The synthesis, structure and activity evaluation of pyrogallol and catechol derivatives as Helicobacter pylori urease inhibitors. Eur. J. Med. Chem.. 2010;45:5064-5070.
- [Google Scholar]
- Crystal structures of isomaltase from Saccharomyces cerevisiae and in complex with its competitive inhibitor maltose. FEBS J.. 2010;277:4205-4214.
- [Google Scholar]
- In vitro and in vivo anti-inflammatory effects of different extracts from Epigynum auritum through down-regulation of NF-κB and MAPK signaling pathways. J. Ethnopharmacol.. 2020;261:113105
- [Google Scholar]
- Membrane stabilization as a mechanism of the anti-inflammatory activity of ethanolic root extract of Choi (Piper chaba) Clin. Phytoscience.. 2020;6:1-10.
- [Google Scholar]
- Identification and quantification of flavonoids and ellagic acid derivatives in therapeutically important Drosera species by LC–DAD, LC–NMR, NMR, and LC–MS. Anal. Bioanal. Chem.. 2011;400:2565-2576.
- [Google Scholar]
- Identification of phenolic components via LC–MS analysis and biological activities of two Centaurea species: C. drabifolia subsp. drabifolia and C. lycopifolia. J. Pharm. Biomed. Anal.. 2018;149:436-441.
- [Google Scholar]
- Screening of potential xanthine oxidase inhibitors in Gnaphalium hypoleucum DC. by immobilized metal affinity chromatography and ultrafiltration-ultra performance liquid chromatography-mass spectrometry. Molecules. 2016;21:1242.
- [Google Scholar]
- Fast profiling of chemical constituents in Yiqing Capsule by ultra-performance liquid chromatography coupled to electrospray ionization tandem mass spectrometry. J. Sep. Sci.. 2012;35:174-183.
- [Google Scholar]
- Preparations and antioxidant activities of sesamol and it's derivatives. Bioorganic Med. Chem. Lett.. 2021;31:127716
- [Google Scholar]
- Simultaneous determination of stilbenes, phenolic acids, flavonoids and anthraquinones in Radix polygoni multiflori by LC–MS/MS. J. Pharm. Biomed. Anal.. 2012;62:162-166.
- [Google Scholar]
- LC-ESI-QTOF-MS/MS Characterization and Estimation of the Antioxidant Potential of Phenolic Compounds from Different Parts of the Lotus (Nelumbo nucifera) Seed and Rhizome. ACS Omega. 2022;7:14630-14642.
- [Google Scholar]
- Chemical profiling and quantitation of bioactive compounds in Platycladi Cacumen by UPLC-Q-TOF-MS/MS and UPLC-DAD. J. Pharm. Biomed. Anal.. 2018;154:207-215.
- [Google Scholar]
Appendix A
Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2022.104135.
Appendix A
Supplementary data
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







