Translate this page into:
Nickel- and cobalt-based heterogeneous catalytic systems for selective primary amination of alcohol with ammonia
⁎Corresponding author. ywangjung@163.com (Chuanjun Yue)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Abstract
It is one of the critical fields of green chemistry to catalyze the selective conversion of biomass-derived alcohol and ammonia to primary amines with extensive application. Recently, catalytic systems consisting of non-noble metal nickel- and cobalt-based catalysts have been developed for catalytic alcohol amination. This paper reviewed these two types of catalytic systems, which are classified as skeleton Co and Ni catalytic systems, supported and modified Co and Ni catalytic systems, emphasized on catalysts and catalysis, and clearly explained where zero-valent cobalt or nickel is active species for catalytic reaction. In supported catalysts, the catalytic active sites constituted by the catalytic active species and its micro-environment can regulate the efficiency of catalytic the reaction. While in modified catalysts, modifiers such as metal Fe, Re and Bi may modulate the catalytic active sites and change the catalytic selectivity. There are differences in structure and size between catalysts prepared by different methods, resulting in distinct interface and electronic properties for alcohol amination, which determines the structure–activity relationships of the catalytic system.
Keywords
Alcohol
Ammonia
Catalytic amination
Supported catalyst
Modified catalyst
Structure–activity relationships
- TON
-
turnover number
- TOF
-
turnover frequency
- DFT
-
density functional theory
- Cov.
-
conversion
- Sel.
-
selectivity
- Ref.
-
reference
- (CO-, CO2-, NH3-, H2-) TPR
-
(CO-, CO2-, NH3-, H2-) temperature-programmed reduction
- XRD
-
X-ray diffraction
- XPS
-
X-ray photoelectron spectroscopy
- SEM/EDS
-
scanning electron microscopy/energy dispersive X-ray spectrometer
- ESI-MS
-
electrospray ionization-mass spectrometry
- TEM
-
transmission electron microscope
- HR-TEM
-
high-resolution transmission electron microscope
- STEM
-
scanning transmission electron microscope
- STEM-EDX
-
scanning transmission electron microscope-energy dispersive X-ray spectrometer
- FT-IR
-
Fourier transform infrared spectroscopy
- BET
-
Brunauer-Emmett-Teller measurement
- XANES
-
X-ray absorption near edge structure
Abbreviations
1 Introduction
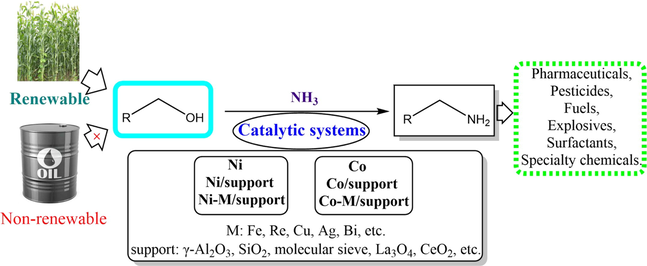
Amine is an indispensable and vital organic compound, widely used in fine chemicals, biology and advanced materials, among which primary amine is a basis for the synthesis of various amines (Geoffrey et al., 2016). Ammonia is an effective and cheap nitrogen source for the preparation of primary amines, because almost all amine compounds in nature can be derived from ammonia under mild conditions. Therefore, producing primary amines from ammonia is meaningful (Yue et al., 2016).
Except for a small amount that can be extracted directly from nature, primary amines are often obtained from following chemical reactions: the aminolysis of halogenated hydrocarbons (Willis. 2007), the amination of alcohol (Shimize et al., 2013), the hydrogenation of nitrile (Laval et al., 2009), the hydroamination of olefins (Klein et al., 2007), the reduction of nitro-compound, and the dehydrogenation coupling of N-H and C-H (Sun et al., 2016; Salomon et al., 2009). Furthermore, the classical amination methods such as the amination of some halogenated hydrocarbons and methanol have been industrialized. To achieve green and sustainable chemical industry development, the selective conversion of biomass-derived alcohol has become a hot spot in the synthesis of primary amine because of its extensive source and environmental friendliness (He et al., 2020; Chen et al., 2021). In recent years, significant progress has been made in the catalysis of alcohol amination by nickel-, cobalt-, ruthenium-, and iridium-based heterogeneous or homogeneous catalytic systems (Bähn et al., 2011; Pelckmans et al., 2017; Liu et al., 2018; Wang et al., 2020). However, it is still a challenge to develop high efficient catalysts for alcohol amination under mild conditions. Fortunately, nickel- and cobalt-based catalysts have shown potential for the catalytic selective primary amination alcohol.
As shown in Scheme 1, the catalytic amination of alcohol with ammonia not only produces amines including primary amine, but also olefin, ether, ketone, nitrile, and polymeric compounds by dehydration, hydrogenolysis, polymerization, cyclization and condensation, which means the design and selection of catalyst is the key to enhance its catalytic selectivity (Wang et al., 2019; Zheng et al., 2020). That is, the catalyst possesses multi-function, and the coordination of the multi-function is the guarantee of catalytic selectivity, otherwise the competitive reactions, resulting in byproducts, will make the catalytic results unsatisfactory. Fortunately, efficient modulation strategies by modification provide the possibility to implement selectively catalytic reactions. At present, the improved heterogeneous nickel-based and cobalt-based catalysts may exhibit almost equivalent catalytic performance to the homogeneous ruthenium-based catalysts. Considering the cost and convenience of post-treatment, the heterogeneous catalyst of alcohol amination is attractive and advantageous. Generally, the catalytic selectivity of catalysts is a part of the catalytic performance (including activity, selectivity, stability, and related mechanical properties, etc.), which overall performance optimization is the guarantee for the catalyst to move towards sustainable industrialization. Therefore, the catalyst for catalytic alcohol amination and its structure–activity relationships are the basis for understanding, designing and preparing the overall optimized catalyst, which depend on the accumulation and summary of numerous studies. In this paper, the catalytic systems based on nickel and cobalt catalysts for alcohol amination are reviewed, in which text materials are organized sequentially and logically according to the time of literature publication.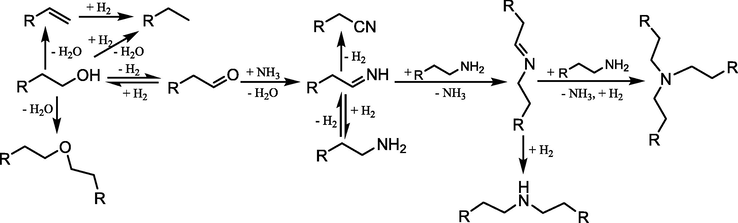
Reactions of alcohol amination with diversified products.
2 Catalytic system of alcohol amination
The catalytic system usually consists of catalyst, catalytic reaction, and catalytic condition. An effective catalytic system is a suitable chemical action process with catalyst of the key. With the development of the catalytic system of alcohol amination, there are three kinds of processes (Scheme 2): dehydration condensation amination, reduction amination and hydrogen transfer amination (Liu et al., 2018). The catalyst for the catalytic amination of alcohol is of dehydration function such as zeolites, silicon oxide, and alumina, which are very effective for the catalytic amination of small molecule alcohols like methanol (Segawa and Tachibana, 1993). The catalytic reductive amination of alcohol requires the dehydrogenation/hydrogenation ability of the catalysts, which undergoes dehydrogenation oxidation, dehydration amination and hydrogenation reduction (Shimizu et al., 2013). The catalytic hydrogen transfer amination of alcohols is a special kind of reductive amination with hydrogen donor. It does not need to be carried out in hydrogen environment, and not only requires the dehydrogenation performance of the catalytic active sites but also enables the removed hydrogen to reduce the imine intermediates. Metal hydride is the active intermediate in catalysis (Alonso et al., 2011). In a word, its advantage is that the atomic economy and safety are better. Therefore, considering catalytic reduction amination and hydrogen transfer amination, the dehydrogenation and hydrogenation nature of catalytic active sites are commonly used. The suitable geometric and electronic structure of the catalytic active site is the basis for the smooth progress of the alcohol amination, and its micro-environment is an essential factor affecting the catalytic performance. For example, the classical preparation process of Raney nickel and cobalt makes it highly active. Because of the higher requirements of the modern chemical industry, the modulation of catalytic active sites has always been the focus of catalysis research (Dong et al., 2020; Ying et al., 2020). First, it is presented the surface area of the catalyst. The larger the surface area of the catalyst is, the more the catalytic active sites would be, the representation is Raney Nickel. As a skeleton catalyst, its catalytic activity sites are slightly influenced by environmental action (including electronic and size effects) due to the difference in dispersion caused by metal loading. Resulting to the 3d orbital in transition metals, irregularity of steps and top angles, and the different aggregation degree on the surface, the intermediate and product on the catalyst surface would change with time and space, which leads to the selectivity of catalysis. The catalyst nature is related to its preparation process, and subsequently affects the catalytic process and reaction results. Thus, it is unreliable to compare the catalytic performance of the same composition catalyst by different preparation methods, because the texture and micro-environment of each catalytic active site on the catalyst surface are not exactly the same, resulting in the different catalytic performance, especially the catalytic selectivity, and the results presented are statistical. Currently, relatively other metals to modify the catalytic active site of the heterogeneous catalysts is a strategy to modulate the catalytic performance, its primary function is to adjust chemical adsorption and control the selectivity of catalytic reaction. Moreover, the heterogeneous catalysts obtained by metal modification would replace part of the scarce and expensive noble metal catalysts, it is manifested in the catalytic amination of alcohol. Based on those above, the cobalt-based and nickel-based catalysts are divided into a skeleton, supported, and modified catalysts for the catalytic selective primary amination alcohol.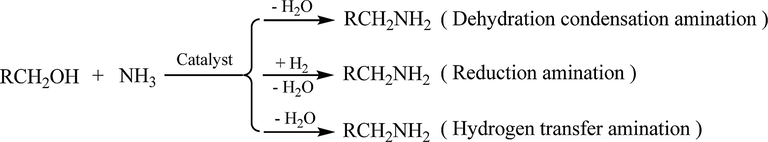
Three kinds of processes for alcohol amination.
2.1 Nickel-based catalytic system
Nickel is a relatively cheap non-precious metal with the outer electron distribution of 3d84s2 and the Pauling’s electronegativity scale of 1.91. The surface of nickel has good adsorption property for hydrogen and excellent hydro-dehydrogenation nature, and the surface activity of zero-valent nickel is high. The nickel-based catalytic system is a purposeful modulation based on the properties of nickel atoms, including skeleton nickel catalyst, supported nickel catalysts containing silicon oxide, and alumina supports, and modified nickel-based catalysts including copper and rhenium modifiers.
2.1.1 Skeleton nickel catalyst
Raney nickel was first discovered by M.Raney, its manufacturing process, that is melt leaching, was published in 1925. At present, it is a commercial catalyst, usually possessing an average surface area of about 100 m2/g, tending to adsorb active hydrogen, and its content is related to parameters with the preparation process, and widely used in the reduction reaction of various organic compounds. It was used to synthesize organic amines in 1939 because of its high activity (Schwoegler and Adkins, 1939). Liu et al. (2017) performed the amination of α-furanol with Raney Ni, and the optimized yield of 78.8% α-amino furan was obtained by using ammonia as the nitrogen source in hydrogen atmosphere at 160 ℃ reaction temperature. Without hydrogen, only 22.2% selectivity of the product could be reached. The XRD and XPS characterization of the catalyst revealed the catalyst was prone to form nickel nitride and deactivated in the absence of hydrogen atmosphere. Wang et al. (2017) used Raney Ni to catalyze the amination of 2-amino-2-methyl-1-propanol in hydrogen and ammonia atmosphere. The 45.6% optimized conversion and 88.3% selectivity, with 1.88 h−1 TON, were obtained under 185 ℃, which were better than that of Raney cobalt. It was found that the pressure of hydrogen regulated the product distribution, temperature and stirring speed also affected the reaction results. Yu et al. (2019) carried out the amination of 1-amino-2-propanol in hydrogen and ammonia atmosphere with Raney nickel as catalyst and potassium carbonate as auxiliary. The yield of 1,2-diaminopropane was 80% with about 1 h−1 TON under 170 ℃, and potassium carbonate promoted the selectivity of the product, while Raney cobalt can only obtain 49.1% yield under the same conditions. Churro et al. (2020) researched the ammonia of cyclohexanol catalyzed by commercial nickel catalyst (81.3% of nickel content and 5.2 nm of nickel diameter), and the yield of cyclohexylamine was 69.3% with 8.1 h−1 TON under the conditions of 2.2:2.2:1 M ratio of ammonia, hydrogen and cyclohexanol, 13.9 g g−1h−1 of space velocity, 200 ℃ and 1 bar pressure in a fixed bed reactor.
In application of the skeleton nickel, the key point should be the optimization of process parameters to obtain better selectivity and yield of product. The surface of skeleton nickel presents catalytic activity based on TON values, and it is sensitive to substrate structure and easy to deactivate in the absence of hydrogen, adding auxiliary is helpful to improve its catalytic performance. After being adsorbed and acted on the surface by ammonia, nickel nitride would form on the catalyst make the catalyst deactivate.
2.1.2 Supported nickel catalyst
The supported catalyst not only makes the active species of heterogeneous catalyst a better dispersion, but also brings some physical properties such as the rigidity with thermal stability and the change of geometric structure of active species at the interface. These features are meaningful for the industrialization of supported catalysts, the modulation of catalytic properties and the stability of catalytic active species due to adsorption (Li et al., 2019). For nickel-based supported catalysts for alcohol amination, a certain amount of acid-base on the surface is beneficial to the catalytic reaction, such as silicon oxide and alumina as nickel-based catalyst supports whose acidity or alkalinity on surface can synergistically play a catalytic role with active species, and be modulated by thermal and aluminum valence states. At the same time, the special surfaces formed by composite silicon oxide and alumina such as various molecular sieves bring great opportunity to tune the catalytic active sites of nickel-based catalysts (Wang et al., 2020a, 2020b) (Fig. 1).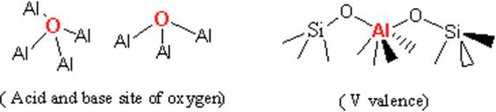
Binding and valence of oxygen and aluminum in alumina.
2.1.2.1 SiO2 support
Silicon oxide is a common catalyst support, and its surface can be adjusted with the variation of preparation method and of silica hydroxyl with acidity. In 1990, Bassili and Baiker (1990) adopted commercial Ni/SiO2 to catalyze the amination of 1-methoxy-2-propanol with NH3. The 70% conversion and 90% selectivity to 2-amino-1-methoxypropane were obtained on the conditions of 190 ℃, 40.5 g g−1h−1 of space velocity and an atmospheric pressure of hydrogen and ammonia in continuous fixed bed reactor, where the presented catalytic efficiency was obviously better than the skeleton nickel (Churro et al., 2020). When the reaction temperature is higher than 190 ℃, the methoxy group would dissociate, which would lead to the decrease of product selectivity, and the catalyst is easily deactivated with hydrogen lacking. Zamlynny et al. (1998) used Ni/SiO2 prepared by impregnation to catalyze the selective amination of cyclohexanol. The optimal 90% conversion and 70% selectivity to cyclohexylamine were obtained using 40 wt% Ni/SiO2 as catalyst under the conditions of 210 ℃, 4 g h−1mol−1 of space velocity, and hydrogen atmosphere. Comparing to the commercial skeleton nickel (Churro et al., 2020) and Ni/SiO2 (Bassili and Baiker, 1990), the nickel-based catalyst here did not show a good catalytic efficiency and might be attributed to the production of the catalyst. Moreover, the results also showed that the catalytic conversion increased as nickel loading, which further revealed the Ni0 was catalytic active species, and Ni2+ was inactive. The reaction process is dehydrogenation amination, while the catalytic process is affected by the acid strength on the surface of the support. Fischer et al. (1999) used commercial Ni/SiO2 (180 cm2/g of BET surface area, 0.17 cm3/g of hole capacity, 4.9 nm of mean pore size, and 5 nm of nickel diameter) as catalyst to catalyze the amination of 1,3-propanediol, 2-methyl-1-propanediol and 2,2-dimethyl-1- propanediol in supercritical ammonia (Tc = 132.4 ℃, Pc = 114.8 bar) and hydrogen atmosphere. The results showed that the substrate structure affected the selectivity of diamine due to dehydration, condensation and degradation in the catalytic reaction, which indicated that the reaction conditions affected the potential competitive reaction performance. For 2-methyl-1,3-propanediol, the 75% conversion and 77% selectivity of monoaminated products can be obtained on the conditions of 135 bar hydrogen and ammonia pressure, 180–235 ℃ and 40000 g s−1mol−1 of space velocity.
Summarily, the nickel catalyst supported by silicon oxide showed good catalytic performance of excellent conversion and selectivity for specific substrates under supercritical conditions. It is also presented that zero-valent nickel is a catalytic active species, and silicon oxide support have a positive effect on the catalytic results to some extent, which depended on the catalyst structure derived from the catalyst preparation process.
2.1.2.2 Al2O3 support
Alumina is also a common catalyst and support in chemical industry with adjustable crystal form, size, and surface properties. γ-Al2O3 is a tetragonal system of disordered transition states with lattice vacancies, which brings chances to tune the catalytic performance of supported catalysts. Shimizu et al. (2013) prepared nickel catalysts with different supports by impregnation method, and for the first time, the heterogeneous catalysts without hydrogen were used in hydrogen transfer reductive amination of alcohols for primary amines. The results showed different crystalline alumina as nickel catalyst support had different effects on alcohol amination, and the support effect would increase as the catalyst surface area expand among the 17 m2/g of ɑ-Al2O3, 112 m2/g of θ-Al2O3, and 124 m2/g of γ-Al2O3. The surface acidity or alkalinity of Ni0 and γ-Al2O3 was the main factor affecting catalytic amination. Additionally, it was found that the electronegativity of these supports was higher than that of simple nickel. Comparing with Raney nickel, the outer electron cloud density of nickel is distinctly reduced. As shown in Fig. 2, supports with different electronegativity have different surface acid and alkalinity, and differed in their efficiency for catalytic alcohol amination. Under the conditions of 0.4 MPa ammonia pressure and 140–160 ℃, the yield of primary amines reached 68-–96% using 1 mol% Ni/γ-Al2O3 as catalyst for alcohols amination, which showed excellent catalytic performance than some homogeneous ruthenium and other noble metal catalysts, and the catalysts could be reused. The reason might be the facilitation on the hydrogen transfer process caused by the acid-base site on the surface of γ-Al2O3. Based on those results, Ni/CaSiO3 catalyst prepared by ion exchange, which was used successfully for the hydrogen transfer amination of a variety of fatty alcohols and aromatic alcohols to produce primary amines (Shimizu et al., 2014).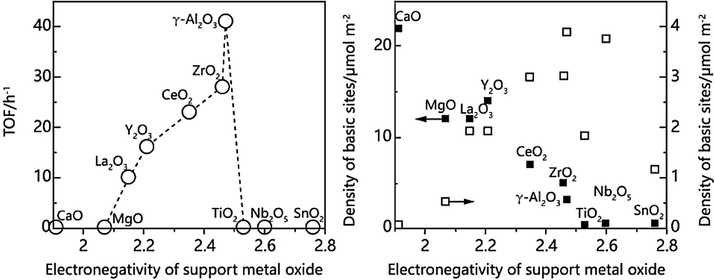
Relationship between electronegativity of different supports of Ni-based catalysts and alkaline sites and catalytic properties (Shimizu et al., 2013, with permission from ACS).
Cho et al. (2013) loaded nickel on commercial γ-Al2O3 surface (194 m2/g of surface area) with impregnation method to get the Ni/γ-Al2O3 catalyst for the catalytic amination of isopropanol. The catalyst with 17 wt% of nickel content and 9.9 nm of particle size showed a good performance with the 90% conversion and 80% selectivity to isopropylamine under 170 ℃. The results presented that the nickel activity of the catalyst is better maintained in the hydrogen atmosphere, because the presence of hydrogen could prevent the formation of nickel nitride and its coverage of the catalyst surface. Moreover, nickel catalysts supported by a variety of crystalline alumina were prepared with impregnation for the reductive amination of isopropanol, and was compared with commercial γ-Al2O3. Studies have shown that the catalytic selectivity was related to the crystal form of γ-, δ-, η-, θ, κ-Al2O3 supports (Cho et al., 2016), and the pyridine adsorption experiment revealed that the catalytic selectivity was depended on the number of Lewis acid on the surface of the catalyst. And γ- and η-Al2O3 exhibited relatively high activity, the yields of isopropanol amination were 48.6% and 59.9% under 160 ℃ and hydrogen atmosphere, respectively.
Dumon et al. (2018) prepared the Ni/γ-Al2O3 catalyst with 138 m2/g of surface area and 18% nickel dispersion by impregnation for the direct amination of 1-octanol. The results showed that the 137 h−1 TOF catalytic efficiency of the catalyst was better than that of the same type of palladium with 23 h−1. DFT calculations revealed that the adsorption of ammonia reduced the catalytic dehydrogenation energy of hydroxyl groups and stabilized imine intermediates to alter the catalytic reaction rate, which demonstrated that ammonia was not only a nitrogen source, but also acted with the catalyst surface to alter the process of alcohol amination, implying that it played a role in the regulatory reaction.
Park et al. (2017) loaded nickle on commercial γ-Al2O3 (194 m2/g of surface area) with impregnation to catalyze the amination of ethanol. The catalyst characterization revealed that nickel loading affected the morphology of the catalyst, the optimal 10 wt% Ni/γ-Al2O3 with the highest 12.0% nickel dispersion and the minimum 8.1 nm nickel particle size among those catalysts presented the excellent performance of both 40% of conversion and selectivity to ethylamine under the conditions of 190 °C, 0.9 g g−1h−1 of space velocity and 1/3/6 M ratio of EtOH/NH3/H2. The presence of ammonia in the catalytic reaction could inhibit the oxidation of acetaldehyde, while the existing hydrogen could prevent further dehydrogenation of imine to nitrile, and also restrained the catalyst deactivation by nitrides.
Among the Ni/γ-Al2O3 catalysts, particle size of nickel affects the catalytic activity. Tomer et al. (2017) loaded nickel on commercial γ-Al2O3 (154 m2/g of surface area) by impregnation with β-cyclodextrin (β-CD) during the catalyst preparation process to improve the dispersion of nickel and obtain relatively uniform particle size nickel, thus increasing the amount of catalytic reduction species on the catalyst surface and enhancing catalytic activity. The leaching of nickel in the catalytic reaction was inhibited to some extent and the stability of the catalyst was improved. The results showed that the 79% conversion and 86% selectivity of 1-octyl amine could be realized by using 15 wt% Ni/γ-Al2O3 as catalyst under the conditions of 160 °C and 7 bar ammonia pressure. Therefore, in the Ni/Al2O3 catalyst, the morphology and surface properties of the support, the particle size of the support and nickel, namely the surface area, and the spatial relationship including the dispersion between them and the relative amount directly affected on the catalytic amination performance.
The composite support of alumina and silicon oxide can also play a support effect in the alcohol amination by nickel-based heterogeneous catalysts. Leung et al. (2018) used commercial Ni/Al2O3-SiO2 catalyst for the primary amination of alcohols in the flow reactor (Fig. 3). In a batch reactor without hydrogen atmosphere, the amination of benzyl alcohol catalyzed with a nickel content of 65 wt% obtain only 44% conversion and 85% selectivity to benzyl amine, while the catalytic amination in continuous process could get the 100% conversion of benzyl alcohol and 100% selectivity to benzyl ammonia, which reduced the possibility of further reaction of the product and greatly improved the catalytic efficiency of the target product. This continuous technology has great significance in reactor engineering of alcohol amination.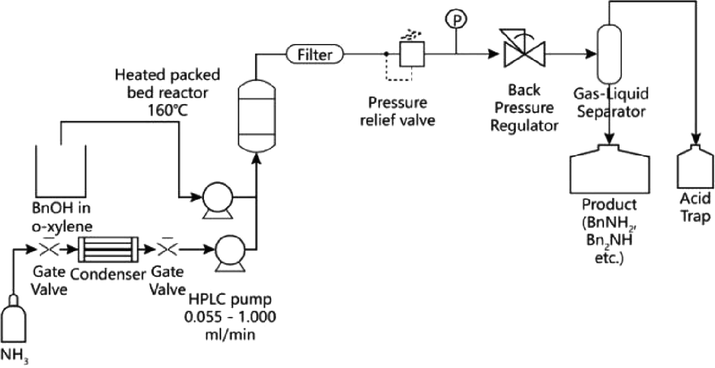
Flow diagram of catalytic alcohol amination by Ni/Al2O3-SiO2 (Leung et al., 2018, with permission from ACS).
Liu et al. (2019) adopted commercial Ni/Al2O3-SiO2 and Raney Ni to catalyze the amination of benzyl alcohols in micro-reactor. The optimal 99% conversion and 58% selectivity of primary amination products for benzyl hydroxy as a structural unit of lignin were obtained using Ni/Al2O3-SiO2 catalyst on the conditions of 140 °C and ammonium carbonate as nitrogen source, which indicated that the catalyst had a good tolerance to the functional groups of substrate, which meant it might be used for catalytic biomass hydroxyl amination. Raney Ni also presented the catalytic activity for the amination of benzyl alcohol with different structures, the 60–100% conversion and the highest 90% primary amine selectivity were reached at 180 °C. Compared Ni/Al2O3-SiO2 to Raney Ni, the former showed better efficacy for benzyl alcohol amination under relatively mild conditions.
Indeed, the catalyst supports of silicon oxide, alumina, and their composite play an important role in improving nickel-based heterogeneous catalysts for the catalytic amination of alcohols. Not only the supports can disperse and stabilize zero-valent nickel active species, but their surface interaction with active nickel and acidity or alkalinity can also regulate the catalytic amination process as well. The overall optimization of catalytic system and the effective combination with new technologies such as flow catalysis may promote the improvement of catalytic performance significantly.
2.1.2.3 Other supports
Compounds with similar surface properties as alumina and silicon oxide supports can also be used as supports for nickel-based catalysts. Tomer et al. (2017) prepared Ni/Al2O3-CeO2 catalyst for the amination of fatty alcohol, using the mixture of commercial γ-Al2O3 (154 m2/g of surface area) and CeO2 (250 m2/g of surface area) as support materials with the methods of impregnation and co-precipitation. Catalytic results showed that the 2 wt% Ni/Al2O3-CeO2 (148 m2/g of surface area with the similar that as 2 wt% Ni/γ-Al2O3) catalyst for 1-octanol amination presented better catalytic selectivity of 80% to 1-octanylamine than that of the catalyst only prepared by impregnation method on the condition of no hydrogen, yet the conversion was not as good as that from the 8 wt% Ni/γ-Al2O3 catalysis. The characterization results from XRD, CO-TPR, SEM/EDS and XPS revealed that the introduction of Ce improved the reducibility of nickel, especially surface nickel, while Ce also promoted the uniform distribution of 2–3 nm nickel (Fig. 4). Different preparation processes originating from the introduction of new component led to various structure of the catalyst, resulting in differential catalytic properties. Therefore, the preparation of catalytic materials needs to be paid enough attention to in catalyst design.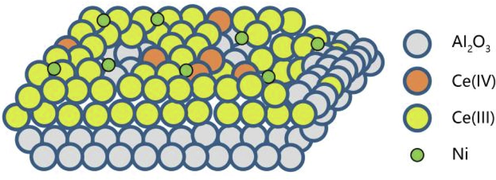
The structure of 2 wt% Ni/Al2O3-CeO2 catalyst (Tomer et al., 2017, with permission from Elsevier).
Ho et al. (2019) prepared nickel catalyst supported by HAP(hydroxyapatite), MgO, γ-Al2O3, and SiO2 with impregnation to catalyze 1-propanol amination in a packed bed reactor. The results showed that in the catalysis of the optimum Ni/HAP at 150 °C, the conversion of 1-propanol was 10.8%, and the selectivity to 1-propanyl amine was 92%. The structure characterization revealed that the Ni particle size of the catalyst is 16.5 nm (minimum belonged to 4.9 nm of Ni/γ-Al2O3) with maximal surface area of 3.9 m2/g among those; kinetic and isotopic studies revealed that the the dehydrogenation of the catalytic reaction as key step depended on the composition of the support and the Ni0 concentration on the surface, showing that Ni0 as the active species and the support could cooperate to complete the decisive step of catalytic amination.
Jeong et al. (2020) sequentially prepared SiO2-Y2O3 composite support by coprecipitation and Ni/SiO2-Y2O3 catalyst by impregnation to catalyze ethanol amination in a fixed bed reactor. The optimal 73% conversion and 35% ethylamine selectivity were obtained using the preferred 10 wt% Ni/SiO2-Y2O3 as catalyst with the 138 m2/g of surface area, 8.1% dispersion of nickel, maximal adsorption for hydrogen and good stability on the conditions of 200 °C, 3.65 h−1 of space velocity and 1/3/6 M ratio of EtOH/NH3/H2. It can be found that the introduction of Y2O3 tuned the surface properties of Ni/SiO2, while the catalytic performance was also improved over Ni/SiO2 (Zamlynnya et al., 1998).
Wang et al. (2020) synthesized USY@Ni catalyst by in situ hydrothermal method of host–guest chemistry (Fig. 5) to catalyze the amination of alcohols.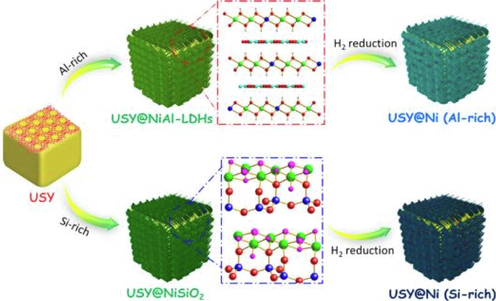
The preparation process of USY@Ni catalyst (Wang et al., 2020, with permission from Elsevier).
The USY@Ni-3 (Si/Al = 3) catalyst featured 20.9% nickel content, 565 m2/g of BET surface area, 6.2% nickel dispersion and 0.142 mmol/g alkali content. XRD characterization showed there was the Ni-Al2O3 crystal phase in the catalyst. Catalytic results showed that the 78% conversion and more than 86% selectivity to 1-octylamine were obtained under the conditions of 180 °C and hydrogen atmosphere. Compared with Raney Ni, the catalyst prepared by this method improved the dispersion of nickel and the selectivity in catalytic reaction.
Li et al. (2020) obtained nickel catalysts supported by SiO2, Al2O3, MgO, TiO2, and ZrO2 by impregnation method in which Ni/ZrO2 presented excellent performance of catalytic amination of 2-hydroxytetrahydropyran from biomass with the 84.7% yield of 5-amino-1-pentanol on the conditions of 80 °C and hydrogen atmosphere in stainless steel reactor. Analysis TPR and NH3-TPR showed that the synergism between reduction of nickel and the acidity of the support made the catalyst exhibit high efficiency, which further suggested that support could efficiently modulate the catalytic properties of active species, manifested as high activity under mild reaction condition.
It can be seen that the support selection of nickel-based supported catalysts and its preparation method not only affected the catalytic active species, but also improved the performance of catalytic alcohol amination by acting with the active species, and it also helped to clarify the structure–activity relationship. However, it still needs better theory to guide.
2.1.3 Modified nickel-based catalysts
The modified nickel-based catalyst can be understood as a further modification of the nickel in nickel-based catalysts mentioned above. This kind of catalyst has the advantages of supported catalyst and multi-level modification of nickel active species, which may improve its catalytic performance, especially for the selectivity.
Hong et al. (2017) prepared Ni-Fe/γ-Al2O3 catalyst with commercial γ-Al2O3 (194 m2/g of surface area) as support by impregnation to catalyze the amination of isopropanol. H2-TPR and XPS characterization revealed that iron would improve the reducibility and stability of nickel, reduced zero-valent nickel was enriched on the catalyst surface, and zero-valent nickel active species on its surface was on the peak when the Fe/Ni molar ratio was 0.3; XRD characterization presented that Ni-Fe alloy was formed in the catalyst. The catalytic performance was that the 83.0% selectivity and 79.3% yield of isopropylamine by the selected 17 wt% Ni-0.3Fe/γ-Al2O3 (134 m2/g of surface area) in a flow reactor with hydrogen atmosphere on the conditions of 150 °C and 500 mL min−1g−1 of space velocity, which showed no sign of deactivation after 100 h of catalyst testing, indicating its stability. The catalytic efficiency of the catalyst was improved by the introduction of iron through the synergistic effect of nickel and iron.
Tomer et al.(2018) prepared bimetallic Ni-Pd/γ-Al2O3 catalysts by the sequential process of adsorbing β-cyclodextrin (β-CD), adding nickel-palladium, removing water and calculating reduction on the γ-Al2O3 support, which was an improved impregnation method. XRD and STEM characterization showed that the both metal contents of nickel and palladium in the catalyst obtained by this method were lower than 5 wt% and 1.0 wt%, respectively, and the distribution of metal particles was more uniform (Fig. 6), which was due to β-CD introduction to isolate and disperse metal particles on the support; ESI-MS and DFT calculations revealed that the preparation process of the catalyst could regulate the interaction between Ni and Pd. The catalyst presented high catalytic performance of 90% conversion and 71% 1-octamine selectivity with 93 TON on the conditions of 160 °C and 7 bar ammonia pressure in a stainless steel reactor. It should be noted that this was a catalysis case with a catalyst that successfully achieved uniform modification and efficient catalytic performance by improving the catalyst preparation process.
Ni-Pd/γ-Al2O3 catalyst by β-cyclodextrin assisted preparation (adapted from Tomer et al., 2018).
Fang et al. (2019) provided low nickel Ni-Pd/CeO2 catalyst and palladium content (<0.5 wt%) by sequential impregnation to catalyze the amination of fatty alcohols. The results showed that the catalytic efficiency of catalysts was affected by the impregnation sequence and Pd loading in the catalyst, presenting that the amination of 1-octanol would be catalyzed to dioctylamine with high selectivity that influenced by cerium oxide support. The introduction of Pd changed the morphology of nickel, and the modification between nickel and palladium could greatly reduce their content in the catalyst, which favored the industrialization of the catalyst.
Re also has a good modification to Ni, and the amination of ethanolamine catalyzed by bimetallic Re-Ni supported by silicon or aluminum and titanium oxides was recorded in patents (King et al., 1998). Ma et al.(2018a, 2018b) prepared Ni-Re/γ-Al2O3 catalyst for the amination of ethanol through impregnation. The results showed that the activity and selectivity of catalytic reaction increased as the adding of Re, and the optimal 50.4% conversion and 82.1% ethylamine selectivity were obtained with the catalyst of optimized Ni15-Re0.5/γ-Al2O3 with 268.1 m2/g of surface area (Note: the number after metal element in the catalyst was a mass percent content.) under the conditions of 170 °C, 60 h−1 of space velocity and hydrogen atmosphere in drip bed reactor. XRD, H2-TPR and acid-base characterization showed that Re improved the dispersion of nickel, and in situ XPS, TEM, and STEM-EDX analysis and DFT calculations revealed that the species dispersed on the ReOx surface played a role in nickel modification. When x ≥ 3, ReOx could reduce the surface energy of the catalyst and stabilize the zero-valent nickel. When x < 3, it helped to adsorb hydrogen in the reaction and improved the reaction activity (Fig. 7). These suggested that the catalytic properties of nickel active species could also be tuned by the variable state of the Re modification. The comparison test with Ni/γ-Al2O3 (224.7 m2/g of surface area) catalyst showed that the catalytic selectivity of Ni-Re/γ-Al2O3 (237.7 m2/g of surface area) catalyst for the amination of ethanol was 65%, with 25 percentage points higher.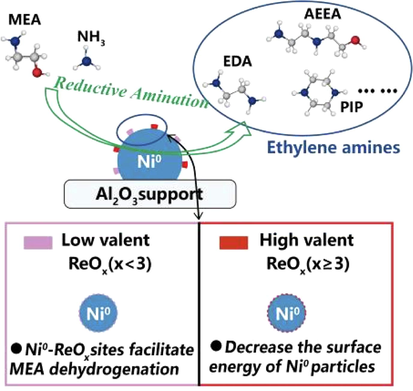
Re role of Ni-Re/γ-Al2O3 catalyst for alcohol amination (Ma et al., 2018a, with permission from ACS).
The group also regulated the size of nickel nanoparticles on the SiO2 support to obtain new Ni-Re/SiO2 catalyst for the amination of ethanolamine. Using the smallest nickel-based catalysts with the size of 4.5–18.0 nm, the catalytic yield was 66.4%, which meant the yield of the product would be affected by the active species distribution. The analysis of XPS and FT-IR showed that the particle size of Ni affected the electronic property of the surface Ni, and then affected the adsorption of ethylamine and the reaction pathway of ethylamine amination (Ma et al., 2019). Ethylenediamine is a widely used product which needs green process of catalysis to produce it. Zhang et al. (2017) adopted NiO/CuO/Al2O3 catalyst prepared by the impregnation to catalyze the synthesis of ethylenediamine from ethylene glycol. The 68.6% conversion and 56.7% selectivity were reached under the optimized conditions of 180 °C and 0.6 MPa in the reactor without hydrogen atmosphere, this catalytic process involved the interaction of copper and nickel. In fact, copper-modified nickel catalyst for catalytic amination of alcohols has been published in patent (Arredondo and Corriga, 2009), which is beneficial to industrial catalytic applications due to the low price. Cui et al. (2013) reported a Raney type polymetallic Ni-Cu/FeOx catalyst prepared by coprecipitation method, with the advantages of cheapness and stability in air. The catalyst could be used in the amination of alcohols effectively. By controlling the amount of ammonia in the catalytic reaction, the selectivity of primary amine products was improved. It was presented that the yield would reach 59–77% for the different alcohols, and the catalyst was easily reused.
In general, nickel-based catalysts show excellent performance in catalytic alcohol amination (Table 1). γ-Al2O3 is an excellent support of nickel-based catalyst with a wide substrate scope, and the new support such as USY plays no less support effect than γ-Al2O3, moreover Fe modification of nickel can better improve the activity of nickel supported γ-Al2O3, manifested as high activity with low temperature, and implying that this modification strategy is effective. At present, for nickel-based catalysts, modifiable metal are limited to iron, copper, rhenium, and palladium, which means metal modifiers need further development. However, the modification of nickel by non-noble metals such as copper and iron still need be further developed in structure refinement with the support of better technology. In combination with modern characterization methods, the relationship between support, nickel and modified nickel metals will be further revealed, which may benefit to provide some valuable evidence for the determination of structure–activity relationship, thus develop the theory of catalysis and guide the design and manufacture of industrial catalysts. * a:sub-substrate, b:SV- space velocity, c:FBR-fixed bed reactor; molar ratio of substrate to reagent.
Entry
Catalytic system
Performance
Ref.
catalyst
substrate
conditions
conv. (%)
sel. (%)
1
Raney Ni
cyclohexanol
NH3/H2/suba = 2.2/2.2/1, 200 °C, 13.9 g g−1h−1 SVb, 1 MPa H2, FBRc.
83.0
83.5
Churro et al., 2020
2
Ni/SiO2
cyclohexanol
NH3/H2/sub = 2/2/1, 210 °C, 4 g h−1 mol−1 SV, 1 MPa (H2 + NH3), FBR.
90
70
Zamlynny et al., 1998
3
Ni/γ-Al2O3
cyclohexanol
0.4 MPa NH3, 0.3 mmol sub, 160 °C, glass tube.
96
99
Shimizu et al., 2013
isopropanol
NH3/H2/sub = 4/6/1, 170 °C, 2.38 g g−1h−1 SV, 1 MPa (H2 + NH3), FBR (microreactor).
90
80
Cho et al., 2013
ethanol
NH3/H2/sub = 3/6/1, 190 °C, 0.9 g g−1h−1 SV, 1 MPa (H2 + NH3), FBR.
40
40
Park et al., 2017
1-octanol
1.3 mmol sub, 7 bar NH3, 210 °C, 4 h, stainless antoclave.
79
86
Tomer et al. 2017
Ni/Al2O3-SiO2
benzyl alcohol
sub/NH3 = 7, 160 °C, flow rate = 0.06 mL/min, flow reactor.
100
100
Leung et al., 2018
5
Ni/USY
1-octanol
sub/NH3 = 15, 5 bar H2, 180 °C, 16 h, high pressure autoclave.
78
86
Wang et al., 2020
6
Ni-Fe/γ-Al2O3
isopropanol
NH3/H2/sub = 8/6/1, 150 °C, 500 mL min−1g−1 SV, 1 MPa (H2 + NH3), flow reactor.
95.5
79.3
Hong et al., 2017
2.2 Cobalt-based catalytic systems
Cobalt is in the third cycle adjacent to nickel in the periodic table, and the outer electrons arrangement of 3d74s2. The electronegativity of Pauling scale is 1.88, and its surface is easy to adsorb hydrogen. Compared with Raney nickel, Raney cobalt has lower efficient in the catalytic primary amine of alcohols, reached 100% conversion of glycerol at 200 °C, but the selectivity to primary amine products of single substitution was extremely low (Ernst et al., 2010). When using Raney cobalt as catalyst in amination of 2-amino-2-methyl-1-propanol at 185 °C, the conversion was 40% and selectivity to 2-methyl-1-propylenediamine was 82% (Wang et al., 2017). Although the total yield was lower than that of Raney nickel, Raney cobalt had shown the potential in the amination of alcohols due to its tunable surface electronic state and its good adsorption of hydrogen and ammonia. Therefore, the modification of cobalt-based catalysts is an important research field in the catalytic amination of alcohols.
2.2.1 Supported cobalt catalyst
Because of the comparability of metal cobalt and nickel in some properties, the support of supported nickel-based catalysts is also suitable as the support of cobalt-based catalysts to some extent. Usually, the catalytic active species with low activity are easy to regulate in catalytic selectivity. So supported cobalt-based catalysts have been developed for catalytic alcohol amination.
2.2.1.1 SiO2 and Al2O3 supports
SiO2 and Al2O3 based on industrial catalytic applications are both of more important alternative supports for cobalt-based catalysts. Sewell et al. (1995) reported Co/SiO2 and Ni/SiO2 catalysts prepared through impregnation for the reduction amination of ethanol. The results showed that the catalytic conversion per pass of ethanol was higher than 60%, and selectivity to ethylamine was more than 50% under the conditions of 180 °C, 0.25 g g−1h−1 of space velocity and 1/2/10 M ratio of ethanol/ammonia/hydrogen in a fixed bed reactor; while the catalytic amination of ethanol was <25% by Ni/SiO2. Obviously, the catalytic efficiency of cobalt-based catalyst was better than that of nickel-based catalyst, and nickel was more suitable to alumina support for ethanol amination (Park et al., 2017). It was considered that the selectivity of catalytic reaction was related to the support and activation process of the catalyst. The efficiency of catalytic ethanol amination by Co/SiO2 was also better than that of Co/γ-Al2O3 and Co/Al2O3-SiO2 catalysis (Sewell et al., 1997). The results of catalyst characterizations showed that the 2.1 m2/g surface area and 8.7% metal dispersion of Co/SiO2 were lower than 6.6 m2/g and 12.7% of Ni/SiO2, respectively. It could be explained as the efficient synergism of the SiO2 support and the Co active species in Co/SiO2, which was determined to be the main factor for the high efficient amination of ethanol by the catalyst. The coking of the catalyst in the process produced cobalt nitride which deactivated the catalyst.
Cho et al. (2012) prepared Co/γ-Al2O3 catalyst for the amination of isopropanol through impregnation. The preferred 23 wt% Co/γ-Al2O3 showed excellent catalytic performance of the 81.5% conversion and 71.7% selectivity to isopropylamine on the conditions of 190 °C, 4.29 g g−1h−1 of space velocity and 1/6/12/14.8 M ratio of 2-propanol/ammonia/hydrogen/nitrogen in a fixed-bed reactor. The catalyst showed no signs of deactivation after 100 h of test. Although the catalyst was slightly less efficient than Ni/γ-Al2O3 (Shimizu et al., 2013), it exhibited higher lifetime (Cho et al., 2013). The results of catalyst characterizations revealed that the catalyst with 23 wt% cobalt content had 124 m2/g surface area, which was the largest in 4–27 wt% Co/γ-Al2O3 catalysts, with the particle size of 12.5–21.5 nm, and the larger particle was preferentially reduced. There were Co3+, Co2+ and Co oxidation states in the catalyst, which were determined by H2-TPR, indicating the adsorption and oxidation capacity of hydrogen. The formation of metal nitride was a common problem, resulting in the deactivation of catalysts.
Niu et al. (2019) reported the new preparation strategy of Co/γ-Al2O3 through impregnation, which introduced the deposition carbon to modify the active sites, in order to improve the selectivity of the catalytic reaction (Fig. 8). The 14.5 wt% Co/γ-Al2O3 with 140 m2/g surface area for amination of 1-butanol provided about 10% conversion with 104.6 h−1 TOF at 180 °C in fixed bed reactor, but for the space barrier on the catalyst surface inhibited the formation of secondary and tertiary amines, the selectivity to primary amine was enhanced, such as 1-octanol and benzyl alcohol with the primary amination increasing from 30 to 50% to 80–90%.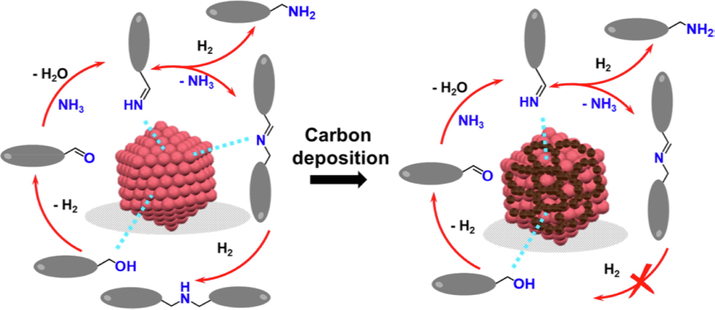
Preparation and modification of Co/γ-Al2O3 by carbon deposition strategy for catalytic selective primary amination of alcohol (Niu et al., 2019, with permission from ACS).
Zhang et al. (2007) prepared Co/γ-Al2O3 and Co/H-ZSM-5 catalysts with impregnation for the amination of ethanolamine. The results from 20 wt% Co/γ-Al2O3 catalysis showed that the 58.2% conversion and 13.6% ethylenediamine selectivity were obtained at 230 °C in a fixed bed reactor with hydrogen atmosphere, whereas the 76.8% conversion and 12.1% selectivity were presented by the catalysis of 20 wt% Co/H-ZSM-5 (the 25 ratio of silicon to aluminum in H-ZSM-5), which meant the H-ZSM-5 support was priority to γ-Al2O3 in this experiment, once again indicating that the efficiency of supported catalysts was determined by whether the active species match support well. Further studies revealed that cobalt in the catalyst was the key to catalytic dehydrogenation and hydrogenation, where zero-valent cobalt was the active species, and the support acted as a dispersion of active cobalt for alcohol amination. It implied that cobalt-based catalysts were similar to nickel-based those for alcohol amination. However, the structure and performance of catalyst was different.
2.2.1.2 Other supports
The cobalt-based catalysts supported by silicon oxide and alumina has been explored in alcohol amination, and this alone is far from enough for the development of this field. Therefore, it is necessary to broaden the selection and design of supports for cobalt-based catalysts. The molecular sieves with silicon, aluminum, and oxygen component has shown the excellent support effect (Irrgang and Kempe, 2020), which presented the match of cobalt active species with the molecular sieve in catalysis for the alcohol amination (Cho et al., 2012; Niu et al., 2019), and also showed the diversity of supports (Fischer et al., 1997). Accordingly, efficient catalysts are expectantly found by efficiently matching more supports and cobalt-active species in the catalysis of alcohol amination.
Yue et al. (2019) used oxide supported Co catalysts, which was prepared by situ liquid phase reduction, to catalyze the reductive amination of 1,2-propanediol. The Co/La3O4 with 10 nm diameter showed an excellent catalytic performance of the optimal 68% conversion and 89% selectivity to 2-amino-1-propanol at 160 °C in a stainless steel reactor. The results of XPS, TEM, and CO2-TPD characterizations revealed that the cobalt-based catalysts prepared by this method allowed cobalt to be dispersed on the surface of the support, while Co-La-O phase was formed at its interface, which stabilized cobalt active species. The alkaline site on the La3O4 surface promoted Co-La-O the dehydrogenation process of hydroxyl groups, leading to high catalytic efficiency. The catalyst shortcoming lay in only presenting 5.1 m2/g BET surface area, which meant it could not maximize the catalytic performance, but it indicated higher selectivity than the Co-La catalyst prepared by coprecipitation (Ichikawa and Fuchu, 1979), suggesting that the catalytic active site can be regulated by preparation methods.
Yuan et al. (2019) used Co@NC catalyst, i.e., cobalt supported by nitrogen-doped carbon, for the amination of carbonyl (it might stem from alcohol). The catalytic results presented that the product yield of 81.8–100% was reached for different carbonyl substrates at 130 °C in hydrogen atmosphere, with the catalyst of 981.2 m2/g surface area and high cobalt dispersion (1.42% of mass percent content and 13.1 nm of average cobalt particle size). Obviously, the amination of carbonyl is more easier than hydroxyl, reflecting that hydroxyl dehydrogenation is a key step in catalyzing alcohol amination. Therefore, the design and preparation of catalyst here need more refinement, such as the precise matching of support and active species, so that the efficiency of catalytic amination can be fundamentally improved, especially selectivity, because there are many competitive reactions during the process of alcohol amination (Scheme 1).
The support of cobalt-based catalyst is diversified, but there has not been an satisfied catalytic system for alcohol amination, which provides chance for the development of cobalt-based catalyst support. At the same time, the preparation of cobalt-based catalyst is also an important aspect of the adjustment of supported cobalt-based catalyst in structure and performance. The methods including in liquid phase reduction and doping methods of special material are also expanded for the preparation of alcohol amination catalysts besides impregnation, which would provide a much more factual basis for the structure–activity relationship of supported cobalt-based catalyst in alcohol amination.
2.2.2 Modified cobalt-based catalysts
The catalysis depends on the valence electrons of active species and the effect of related to atoms, that is, the action of valence electrons of the catalyst alters the way of the reaction, reflecting in the change of potential energy. The modification of cobalt in cobalt-based catalysts regulates their catalytic performance, thus tunes their catalytic efficiency of alcohol amination. The developed modifiers includes iron, nickel, silver, bismuth, barium, platinum, and polymetallic (Baiker and Kijenski, 1985).
Baiker et al. (1999a, 1999b, 1999c) used Co-Fe catalyst, with 8 m2/g surface area and 5 wt% Fe content, prepared by coprecipitation for the amination of diol in a continuous fixed-bed reactor under the supercritical ammonia condition. The result was the 56% conversion and 39% selectivity of monoamine in the amination of 1,3-cyclohexanediol in hydrogen atmosphere under the conditions of 170 °C and 5.55 g h−1mol−1 of space velocity. As for the amination of 1,4-cyclohexandiol, the 76% conversion and the 97% cumulative selectivity to diamine were reached under the conditions of 165 °C and 1.63 g h−1mol−1 of space velocity, which was better than that of Ni/SiO2 (Fischer et al., 1999). The result of the research on the relationship of catalytic selectivity and reaction temperature in the catalytic amination of 1,3-propanediol using the Co-Fe catalyst showed that, above 190° C, the conversion reached 98% while the selectivity was poor, and the by-products were multiple; at relatively low temperature, the conversion was 36%, but the selectivity of monoamine reached 31%. XRD characterization provided the explanation that the Co-Fe catalyst modified by Fe contained more metastable β-Co phase, which helped to improve the activity. The stability of the catalyst was affected by the Fe content and the acidity/alkalinity of the catalytic system, influencing the catalyst lifetime. The Co–Ni/γ-Al2O3 among Co–M/γ-Al2O3 (M: Fe, Ni and Cu) prepared by impregnation for the amination of ethanol showed the excellent catalytic performance of the nearly 100% conversion and 92.5% selectivity to acetonitrile at 420 °C in a continuous fixed bed reactor without hydrogen. While the yield of catalytic ethanol to acetonitrile was 84.2% using the Co20.1Fe2.8/γ-Al2O3 catalyst, clearly indicating the high activity of the modified cobalt-based catalyst (Zhang et al., 2009).
Ibáñez Abad et al. (2018) used silver modified Co–Ag/γ-Al2O3 catalyst for the amination of fatty alcohols with sequential impregnation method. The 90% conversion and 78% selectivity were obtained using the optimized 5 wt% Ag3-Co97/γ-Al2O3 as the catalyst for 1-octanol amination at 200 °C in the improved fixed bed reactor system. H2-TPR, CO-TPD and STEM-EDS characterization results revealed that the introduction of silver had a promoting effect on cobalt, which made cobalt accumulate around silver and strengthen on the reducibility of cobalt, resulting in the activity improving of catalytic amination. Kinetic studies showed that alcohol dehydrogenation was the decisive step of the reaction, and hydrogen pressure had a positive effect on the reaction rate. It was pointed out that the imine produced in the catalytic reaction adsorbed on the catalyst, forming metal nitride might be the original of catalyst deactivation (Ibáñez et al., 2019).
Zhao et al. (2010) obtained Co-Ba-Fe/γ-Al2O3 catalyst by combining three metals and support on the basis of coprecipitation method for 1,2-butandiol amination. The 72.9% conversion and the 73.3% selectivity to 2-amino-1-butanol were got under the optimal conditions of 165 °C and 5 MPa hydrogen pressure in a fixed bed reactor. The results of XRD, XPS and H2-TPR characterizations revealed that Co7Fe3 crystalline phase formed during the catalyst preparation process modified the Co active species and inhibited CoAl2O4 phase, leading to the improvement of the stability and reducibility of the catalyst. Compared with Co-Fe/γ-Al2O3 catalyst (Zhang et al.2009), the introduction of barium changed the structure of Co-Fe crystal phase formed during the preparation of Co-Ba-Fe/γ-Al2O3 catalyst, enhancing the selectivity of catalytic alcohol amination.
Niu et al. (2020) reported the bismuth modified Co-Bi/γ-Al2O3 catalyst prepared by impregnation-coprecipitation method for alcohol amination. The more than 96% conversion and the about 70% selectivity to 1-octylamine were obtained using 10 wt% Co1.0Bi/γ-Al2O3 as the catalyst for 1-octanol under the conditions of 180 °C and 1/4.5/0.85 M ratio of 1-octanol/ammonia/hydrogen in stainless steel reactor. The results of XRD, H2-TPR, STEM, CO-TPR and FT-IR characterizations revealed the Bi introduce in Co-Bi/γ-Al2O3 catalyst not only promoted the reductibility of cobalt and increased the reaction rate, but Bi could migrate near the active site during the catalytic reaction and formed a barrier to prevent the adsorption and hydrogenation of imine, which inhibited the formation of secondary and tertiary amines in the catalytic reaction, then improved the selectivity.
The result of some cobalt-based catalysts modified by metals for the alcohol amination have been reviewed (Baiker and Kijenski, 1985). It is urgent to study the structure–activity relationship because the demand on the heterogeneous catalysis is growing. Modification in reverse is also one of the strategies of the catalyst preparation for alcohol amination. Tong et al. (2019) prepared the cobalt modified Pt–Co/CeO2 catalyst prepared by impregnation method for the reduction amination of alcohols through hydrogen transfer. The 99% conversion and 73.1% selectivity to cyclopentamine were obtained using 1% Pt–1% Co/CeO2 catalyst for cyclopentanol amination under the conditions of 180 °C, 0.2 MPa hydrogen pressure and 0.5 MPa ammonia pressure in stainless steel reactor. The results from XRD, HR-TEM, (H2, CO, NH3)-TPR, XPS and XANES revealed Pt modified by CoOx species in the interface affected the dehydrogenation and prevented the low coordination Pt from hydrogenating the intermediate imine further (Fig. 9).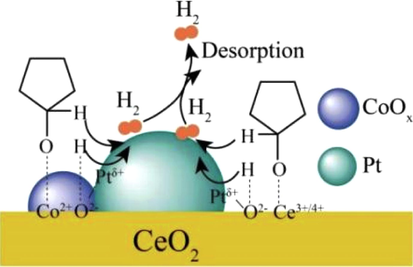
Schematic illustration of catalytic cyclopentanol amination process by Pt–Co/CeO2 (Tong et al., 2019, with permission from Elsevier).
In addition, as palladium was minimally used in the nickel-based catalyst (Fang et al., 2019), the amount of precious metals such as platinum in cobalt-based catalysts may be used as little as possible, but It can supper fully work.
It is clear that cobalt-based catalysts can efficiently catalyze alcohol amination at lower temperature, indicating high catalytic activity and good catalytic selectivity for polyols, especially cobalt modified by metals such as Fe, Ag and Bi in Co/γ-Al2O3 (Table 2). It was understood as the modification of active species with the fundamental change of its catalytic property, usually presenting the formation of new phase. As for heterogeneous catalysis, the regulation of interfacial phase is very important for the selectivity of catalytic reaction. To some extent, the cascade regulation of active species-modifier-support in this type of catalyst can screen the modifier, then to optimize the catalyst, such as the modifier selection. In addition, the size of the catalyst components also affects the activity and selectivity of the catalytic reaction. Those provide an effective way to carry out the substitution of noble metal catalysts in catalysis science. * a: sub-substrate, b: SV- space velocity, c: FBR- fixed bed reactor; molar ratio of substrate to reagent.
Entry
Catalytic system
Performance
Ref.
catalyst
substrate
conditions
conv. (%)
sel. (%)
1
Raney Co
2-amino-2- methyl-1-propanol
0.5 g catlyst, 10.0 g sub, 4.2 g NH3, 1 MPa H2, 185 °C, 7 h, stainless autoclave reactor.
40
82
Wang et al., 2017
2
Co/SiO2
ethanol
NH3/H2/suba = 2/10/1, 180 °C, 0.25 g g−1h−1 SVb, 1 MPa (H2 + NH3), FBRc.
60
50
Sewell et al., 1995
3
Co/γ-Al2O3
isopropanol
NH3/H2/sub = 8/6/1, 190 °C, 4.29 g g−1h−1 SV, 1 MPa (H2 + NH3), FBR.
81.5
71.7
Cho et al., 2012
4
Co/La3O4
1,2-propanediol
2 mL sub, 18 mL aqueous ammonia, 160 °C, 6 h, stailess steel reactor.
68
89
Yue et al., 2019
5
Co-Fe
1,3-cyclohexandiol
NH3/H2/sub = 60/2/1, 165 °C, 1.63 g g−1h−1 SV, 1 MPa (H2 + NH3), FBR.
76
97
Baiker et al., 1999c
6
Co-Ba-Fe/γ-Al2O3
1,2-butandiol
10% in toluene, flow rate: 0.5 mL/min NH3 and 5 mL/h H2, 165 °C, 5 MPa H2, FBR.
72.9
73.3
Zhao et al., 2010
7
Co-Ag/γ-Al2O3
1-octanol
NH3/H2/sub = 9/3.4/1, 200 °C, sub flow rate: 1.2 mL/h, 1 MPa (H2 + NH3), FBR.
90
78
Ibáñez et al., 2018
8
Co-Bi/γ-Al2O3
1-octanol
NH3/H2/sub = 4.5/0.85/1, 180 °C, 5 h, 10 bar (H2 + NH3), stailess steel reactor.
96
70
Niu et al., 2020
3 Catalytic mechanism of alcohol amination
The catalytic reaction mechanism is the catalytic process of unit action, including the catalytic reaction and the catalysis process. The catalyst of catalytic alcohol amination is multi-component in the composition and classified based on understanding, and the catalysis is coordinated effectively among the components. For the catalytic substrate, the process of catalytic alcohol amination may be dehydration amination, dehydrogenation reduction amination and hydrogen transfer amination as well as two by two or all together, where which is dominant in a catalytic process unless it is possible to design and prepare this catalyst with isolated, uniform and well-defined structure of catalytic active sites. For existing catalysts, such as supported Ni/γ-Al2O3 and Co/SiO2, could show catalytic amination activity against alcohols in reactions, and the metal Ni and Co have excellent dehydrogenation and hydrogenation properties. Amination in catalytic conversion process is spontaneous in thermodynamic, while acidic γ-Al2O3 and SiO2 could catalyze dehydration of alcohols, and the exquisite matching of support and metal promotes the reaction to accelerate, that is catalysis. The catalytic dehydrogenation reduction amination of alcohols was usually carried out in hydrogen atmosphere, which maintained the activity of catalyst and promoted the catalytic conversion of reaction intermediate. The catalytic amination of alcohols by hydrogen transfer required the catalyst with the function of hydrogen capture and supply, in order to complete catalytic cycle, in which the activity and stability of the catalyst were the key. For catalytic dehydrogenation reduction amination, Schwegler et al. (1939) proposed a reaction mechanism of “dehydrogenation-imination-hydrogenation amination“, and Shimizu et al. (2013) suggested a similar process above for the hydrogen transfer amination of alcohols. The difference was that the hydrogen of the latter one was derived from the alcohol by dehydrogenation. The catalytic active species undergone a process of metal hydride. But both catalytic decisive steps pointed to dehydrogenation process (Ho et al, 2019; Ibáñez Abad et al., 2018). Dehydrogenation was related to the selective adsorption of the catalyst surface (Wang et al., 2019), and the adsorption strength of ammonia (Dumon et al., 2018). Moreover, carbonyl compound was also detected as intermediate in the catalytic reaction (Baiker et al., 1983). Therefore, the mechanism of catalytic reduction amination and hydrogen transfer amination of alcohols can be described as a cyclic process (Scheme 3), and the degree of hydrogen adsorption is depended on the two difference.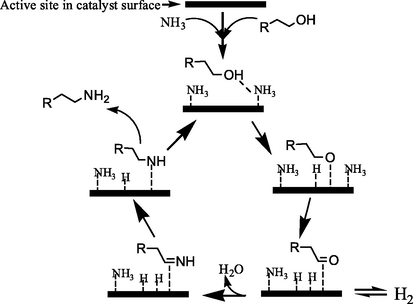
Catalytic dehydrogenation-imination-hydroamination process of alcohol.
The organic conversion process of catalytic alcohol amination has been clarified, but the catalyst needs to be further understood. Considering the adsorption and coordination of nitrogen, some catalysts were easy to form nitride and deactivation without hydrogen atmosphere. Hydrogen could keep the activity and stability of the catalyst. At the same time, the adsorption of ammonia during the catalytic reaction was also one of the factors affecting the catalytic activity and stability. Ibáñez et al. (2019) studied in detail the kinetics of catalytic amination of 1-octanol, presenting the below relationship (Equation (1)) among reaction rate (r), hydrogen pressure (PH2), ammonia pressure (PNH3), the adsorption equilibrium constant (K) and the pressure (Ps) of the substrate.
Therefore, the tacit cooperation of catalytic active species and its micro-environment in supported or modified catalysts accurately regulates the selective adsorption of reactants, and chemisorption and selective conversion are the basis of heterogeneous catalysis, which is determined by the catalytic active site on the catalyst surface. The uniform and consistent catalytic active site is the guarantee of high catalytic selectivity, otherwise the various catalytic competitive reactions as side reactions such as dehydration, ether and polymerization catalyzed by the acidic site on the catalyst will be prominent, due to their advantage over the dehydrogenation in reactivity. The stability of the catalyst can be improved by the composition formulation and preparation process. However, currently reported catalysts for alcohol amination were often performed at high temperature, which limited their catalytic selectivity and the range of catalytic support selection. Therefore, to improve the catalytic activity, the fine modification of cobalt and nickel as non-noble metal representative is inevitable and the selection of modifiers becomes a problem, fundamentally attributed to the dehydrogenation step. More importantly, there is the lack of theoretical guidance for the catalyst design and development. Shimizu et al. (2013) raised an index of electronegativity correlation for measuring the support effect by the activity of active species. The electronegativity index is introduced to evaluate the support modifier selection as overall measurement in cobalt- and nickel-based heterogeneous catalyst design. For example, the supported Pt (electronegativity is 1.89) catalyst is a highly active dehydrogenation and hydrogenation catalyst. When metal modification is introduced into nickel or cobalt, its index relation can be established (Equation (2)).
Correlation factor κ is related to the properties of metals (including metal in support), the degree of interaction and the spatial position are involved in the electron and size of catalytic active site, and interaction between components of catalytic active site during the catalytic process. For the electronegativity not only presents the ability of valence electron, but also relates to the atomic nucleus,which are helpful to make multicomponent heterogeneous catalysts index and facilitate the directional development of catalysts. Actually, Cu/ZrO2 and Cu/γ-Al2O3 also showed the activity for the catalytic amination of alcohols (Chary et al., 2008).
4 Summary and outlook
Alcohol compounds exist in a large number of biomass, and the catalytic dehydration between alcohol and cheap inorganic ammonia is an environmentally friendly way to synthesize all kinds of amines, to which catalyst is the key. Examples of nickel- and cobalt-based catalysts such as Co/SiO2 and Ni/γ-Al2O3 with high catalytic efficiency were representative. While the modified nickel-based and cobalt-based catalysts of Bi and Fe had no less catalytic activity and selectivity than noble metal homogeneous catalysts such as platinum and ruthenium, which demonstrated that the modified catalysts can regulate the performance of the catalysts. It suggested that the electronic and size effects originated from catalyst structure depended on its preparation process to a large extent, and provided a practical basis for the establishment of the accurate structure–activity relationship. Facing industrial applications required for overall properties such as catalytic conversion, selectivity, and stability, the catalytic systems of alcohol amination need further development in following aspects: (1) Preparation methods of catalysts. At present, most of cobalt- and nickel-based catalysts for catalytic amination of alcohols were prepared by the traditional impregnation method, resulting to the low utilization of catalytic active sites. For many catalytic active sites were buried in the bulk phase and could not play a catalytic role. The improved impregnation method could prepare new catalysts with different structure, but it is necessary to develop heterogeneous catalysts with uniform, accurate and isolated catalytic active sites on the surface by an appropriate method, for example of two-dimensional support materials like boron nitride (Zhang et al., 2020). (2) Design of catalysts. The design of new catalysts with high performance based on existing catalysts is fundamental. However, catalysts for alcohol amination often need to operate at high temperatures, which was difficult to improve the selectivity of catalytic reaction and limited the range of support selection. The design and preparation of catalysts based on active species such as metal hydride may drastically improve the activity and reduce the catalytic reaction temperature, which would help enhance the selectivity, solve the catalyst deactivation and develop new processes. In addition, needing more in situ characterization techniques support the design and preparation of catalysts. (3) The lifetime of catalysts. Catalytic hydrogen transfer amination of alcohols features atomic economy. Making metal nitride formation clear and solving the catalyst deactivation in the catalytic process is also the basic problem in this kind of catalytic reaction. The modified heterogeneous catalyst is showing the dawn here.
Acknowledgment
This study was supported by the Nature Science Foundation of the Jiangsu Higher Education Institutions of China (19KJB530004) and the 333 Project in Jiangsu Province of China.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Nickel nanoparticles in hydrogen transfer reactions. Acc. Chem. Res.. 2011;44:379-391.
- [Google Scholar]
- Arredondo, V.M., Corriga, P.J., 2009. Process for the conversion of glycerol to propylene glycol and amino alcohol. U.S. Patent 7619118 B2.
- Catalytic amination of 1-methoxy-2-propanol over silica supported nickel: study of the influence of the reaction parameters. Appl. Catal.. 1990;65:293-308.
- [Google Scholar]
- Catalytic synthesis of higher aliphatic amines from the corresponding alcohols. Catal. Rev.. 1985;27:653-697.
- [Google Scholar]
- Catalytic amination of aliphatic alcohols in the gas and liquid phases: kinetics and reaction pathway. Ind. Eng. Chem. Prod. Res. Dev.. 1983;22:217-225.
- [Google Scholar]
- Expanding the boundary of biorefinery: organonitrogen chemicals from biomass. Acc. Chem. Res.. 2021;54:1711-1722.
- [Google Scholar]
- Amination Reaction of Cyclohexanol Over a Commercial Ni Based Catalyst. Part I: Influence of Operating Conditions. Ind. Eng. Chem. Res.. 2020;59:6565-6579.
- [Google Scholar]
- Reductive amination of 2-propanol to monoisopropylamine over Ni/γ-Al2O3 Catalysts. Catal. Lett.. 2013;143:1319-1327.
- [Google Scholar]
- Effect of an Alumina phase on the reductive amination of 2-propanol to monoisopropylamine over Ni/Al2O3. Catal. Lett.. 2016;146:811-819.
- [Google Scholar]
- Reductive amination of 2-propanol to monoisopropylamine over Co/γ-Al2O3 catalysts. Appl Catal A: Gen. 2012;417–418:313-319.
- [Google Scholar]
- Development of a general non-noble metal catalyst for the benign amination of alcohols with amines and ammonia. Chem. Eur. J.. 2013;19:3665-3675.
- [Google Scholar]
- Characterization and reductive amination of cyclohexanol and cyclohexanone over Cu/ZrO2 catalysts. Catal. Commun.. 2008;9:75-81.
- [Google Scholar]
- Supported metal clusters: fabrication and application in heterogeneous catalysis. ACS Catal.. 2020;10:11011-11045.
- [Google Scholar]
- Direct n-octanol amination by ammonia on supported Ni and Pd catalysts: activity is enhanced by “spectator” ammonia adsorbates. Catal. Sci. Technol.. 2018;8:611-621.
- [Google Scholar]
- Ernst, M., Hoffer, B.W., Melder, J.P., 2010. Method for Producing Amines from Glycerin. U.S. Patent 2010/0240894 A1.
- Nickel-catalyzed amination of 1,3-propanediols differently substituted at C2-position: influence of reactant structure on diamine production. J. Mol. Catal. A: Chem.. 1999;149:197-204.
- [Google Scholar]
- Synthesis of 1,4-diaminocyclohexane in supercritical ammonia. J. Catal.. 1999;182:289-291.
- [Google Scholar]
- Cobalt-catalyzed amination of 1,3-propanediol: effects of catalyst promotion and use of supercritical ammonia as solvent and reactant. J. Catal.. 1999;183:373-383.
- [Google Scholar]
- Sustainable access to renewable N-containing chemicals from reductive amination of biomass-derived platform compounds. Green Chem.. 2020;22:6714-6747.
- [Google Scholar]
- Propanol amination over supported nickel catalysts: reaction mechanism and role of the support. ACS Catal.. 2019;9:2931-2939.
- [Google Scholar]
- Reductive amination of isopropanol to monoisopropylamine over Ni-Fe/γ-Al2O3 catalysts: Synergetic effect of Ni-Fe alloy formation. Appl. Catal. A: Gen.. 2017;542:146-153.
- [Google Scholar]
- Transition-metal-catalyzed reductive amination employing hydrogen. Chem. Rev.. 2020;120:9583-9674.
- [Google Scholar]
- Ichikawa, Y., Fuchu, Yoshisato, E., Nakagawa, K., 1979. Process for preparing amino alcohols. U.S. Patent 4151204A.
- Ru and Ag promoted Co/Al2O3 catalysts for the gas-phase amination of aliphatic alcohols with ammonia. Catal. Sci. Technol.. 2018;8:5858-5874.
- [Google Scholar]
- Direct amination of 1-octanol with NH3 over Ag-Co/Al2O3: promoting effect of the H2 pressure on the reaction rate. Chem. Eng. J.. 2019;358:1620-1630.
- [Google Scholar]
- Characteristics of Si-Y mixed oxide supported nickel catalysts for the reductive amination of ethanol to ethylamines. Catal. Today. 2020;352:287-297.
- [Google Scholar]
- First catalytic hydroaminomethylation in supercritical ammonia. Chem. Eng. Technol.. 2007;30:721-725.
- [Google Scholar]
- King, S.W., Depot, S., Va,W., 1998. Reductive amination catalysts. U.S. Patent 5750790.
- A mild and efficient method for the reduction of nitriles. Tetrahedron Lett.. 2009;50:7005-7007.
- [Google Scholar]
- Advances in primary amine synthesis by reductive amination of alcohols using heterogeneous metal catalysts. J. Chem. Eng. Chinese U. 2018;32:487-498.
- [Google Scholar]
- Switchable synthesis of furfurylamine and tetrahydrofurfurylamine from furfuryl alcohol over Raney Nickel. Catal. Sci. Technol.. 2017;7:4129-4135.
- [Google Scholar]
- Catalysis in flow: Nickel-catalyzed synthesis of primary amines from alcohols and NH3. ACS Sustainable Chem. Eng.. 2018;6:5479-5484.
- [Google Scholar]
- Primary Benzylamines by efficient N-alkylation of benzyl alcohols using commercial Ni catalysts and easy-to-handle ammonia sources. ACS Sustainable Chem. Eng.. 2019;7:11267-11274.
- [Google Scholar]
- Adsorption of Nin (n=1-4) clusters on perfect and O-defective CuAl2O4: A DFT study. Chin. Chem. Lett.. 2019;30:1147-1152.
- [Google Scholar]
- Effective synthesis of 5-amino-1-pentanol by reductive amination of biomass-derived 2-hydroxytetrahydropyran over supported Ni catalysts. Chin. J. Catal.. 2020;41:631-641.
- [Google Scholar]
- Role of ReOx Species in Ni-Re/Al2O3 Catalyst for Amination of Monoethanolamine. J. Phys. Chem. C. 2018;122:23011-23025.
- [Google Scholar]
- Effect of Re promoter on the structure and catalytic performance of Ni-Re/Al2O3 catalysts for the reductive amination of monoethanolamine. RSC Adv.. 2018;8:8152-8163.
- [Google Scholar]
- Effects of Ni particle size on amination of monoethanolamine over Ni-Re/SiO2 catalysts. Chin. J. Catal.. 2019;40:567-579.
- [Google Scholar]
- Catalyst deactivation for enhancement of selectivity in alcohols amination to primary amines. ACS Catal.. 2019;9:5986-5997.
- [Google Scholar]
- Multifaceted role of mobile bismuth promoter in alcohol amination over cobalt catalysts. Green Chem.. 2020;22:4270-4278.
- [Google Scholar]
- Bio-based amines through sustainable heterogeneous catalysis. Green Chem.. 2017;19:5303-5331.
- [Google Scholar]
- Reductive amination of ethanol to ethylamines over Ni/Al2O3 catalysts. Korean J. Chem. Eng.. 2017;34:2610-2618.
- [Google Scholar]
- Heterogeneous Ni catalyst for direct synthesis of primary amines from alcohols and ammonia. ACS Catal.. 2013;3:112-117.
- [Google Scholar]
- Activation of ethylene and ammonia at iridium: C-H versus N–H oxidative addition. Dalton Trans. 2009:7669-7677.
- [Google Scholar]
- Shape-Selective Reactions for Methylamine Synthesis from Methanol and Ammonia. Stud. Surf. Sci. Catal.. 1993;75:1273-1283.
- [Google Scholar]
- N-alkylation of ammonia and amines with alcohols catalyzed by Ni-loaded CaSiO3. Catal. Today. 2014;232:134-138.
- [Google Scholar]
- Reductive amination of ethanol with silica supported cobalt and nickel catalysts. Appl. Catal. A: Gen.. 1995;125:99-112.
- [Google Scholar]
- Effect of activation procedure and support on the reductive amination of ethanol using supported cobalt catalysts. J. Catal.. 1997;167:513-521.
- [Google Scholar]
- Facile preparation of Ni/Al2O3 catalytic formulations with the aid of cyclodextrin complexes: towards highly active and robust catalysts for the direct amination of alcohols. J. Catal.. 2017;356:111-124.
- [Google Scholar]
- Mixed oxides supported low-nickel formulations for the direct amination of aliphatic alcohols with ammonia. J. Catal.. 2017;356:133-146.
- [Google Scholar]
- Cyclodextrin-assisted low-metal Ni-Pd/Al2O3 bimetallic catalysts for the direct amination of aliphatic alcohols. J. Catal.. 2018;368:172-189.
- [Google Scholar]
- Dual functions of CoOx decoration in PtCo/CeO2 catalysts for the hydrogen-borrowing amination of alcohols to primary amines. J. Catal.. 2019;378:392-401.
- [Google Scholar]
- Palladium-catalyzed coupling of ammonia and hydroxide with aryl halides: the direct synthesis of primary anilines and phenols. Angew. Chem. Int. Ed.. 2007;46:3402-3404.
- [Google Scholar]
- Organonitrogen chemicals from oxygencontaining feedstock over heterogeneous catalysts. ACS Catal.. 2020;10:311-335.
- [Google Scholar]
- Rational design of selective metal catalysts for alcohol amination with ammonia. Nat. Catal.. 2019;2:773-779.
- [Google Scholar]
- Reductive amination of 2-amino-2-methyl-1-propanol and ammonia to produce 2-methyl-1,2-propanediamine over Raney Nickel catalyst. ChemistrySelect. 2017;2:8818-8823.
- [Google Scholar]
- Pentacoordinated aluminum species: new frontier for tailoring acidity-enhanced silica−alumina catalysts. Acc. Chem. Res.. 2020;53:2648-2658.
- [Google Scholar]
- Mapping the oxygen structure of γ-Al2O3 by high field solid-state NMR spectroscopy. Nat. Commun.. 2020;11:3620.
- [Google Scholar]
- Host-guest chemistry immobilized nickel nanoparticles on zeolites as efficient catalysts for amination of 1-octanol. J. Catal.. 2020;381:443-453.
- [Google Scholar]
- “More is different:” synergistic effect and structural engineering in double-atom catalysts. Adv. Funct. Mater. 2020 2007423
- [Google Scholar]
- Synthesis of 1,2–propanediamine via reductive amination of isopropanolamine over Raney Ni under the promotion of K2CO3. Chem. Pap.. 2019;73:2019-2026.
- [Google Scholar]
- Preparation of nitrogen-doped carbon supported cobalt catalysts and its application in the reductive amination. J. Catal.. 2019;370:347-356.
- [Google Scholar]
- N-H bond activation of ammonia for catalytic organic reactions. Chem. Word. 2019;60:553-560.
- [Google Scholar]
- Selective amination of 1,2-propanediol over Co/La3O4 catalyst prepared by liquid-phase reduction. Mol. Catal.. 2019;477:110539.
- [Google Scholar]
- Industrial catalysis: Strategies to enhance selectivity. Chin. J. Catal.. 2020;41:1032-1038.
- [Google Scholar]
- Amination of cyclohexanol over metallosilicate-based materials. Appl. Catal. A: Gen. 1998;169:119-125.
- [Google Scholar]
- Synthesis of ethylenediamine by catalytic amination of ethylene glycol. Chin. J. Appl. Chem.. 2017;34:557-562.
- [Google Scholar]
- Amination of ethanolamine over cobalt modified H-ZSM-5 catalysts. Catal. Commun.. 2007;8:1102-1106.
- [Google Scholar]
- Amination of ethanol to acetonitrile over Ni-doped Co/γ-Al2O3 catalyst. Catal. Commun.. 2009;10:1454-1458.
- [Google Scholar]
- The role of iron in the selective amination of 1,2-butanediol to 2-amino-1-butanol over Co-Ba-Fe/γ-Al2O3. React. Kinet. Mech. Cat. 2010;100:449-457.
- [Google Scholar]
- Enhanced and stabilized hydrogen production from methanol by ultrasmall Ni nanoclusters immobilized on defect-rich h-BN nanosheets. PNAS. 2020;117:29442-29452.
- [Google Scholar]







