Translate this page into:
Optimization of extraction process of Dioscorea nipponica Makino saponins and their UPLC-QTOF-MS profiling, antioxidant, antibacterial and anti- inflammatory activities
⁎Corresponding author. chunliantian@syau.edu.cn (Chunlian Tian)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Dioscorea nipponica Makino exhibits many biological activities, including relieving cough, eliminating phlegm and preventing asthma. The present study extensively evaluated the extraction process, major components, antioxidant, antibacterial and anti-inflammatory activities of total saponins extraction from Dioscorea nipponica Makino. In this study, the optimal extraction process of total saponins extract was optimized by single-factor test and response surface methodology as follows: extraction time 25 min, ethanol concentration 50 % and liquid to material ratio 55:1 ml/g, and the extraction rate was 1.72 %. Eighteen components were initially analyzed by UPLC-QTOF-MS method. Although total saponins extract exhibited mild antibacterial activities against Escherichia coli, Salmonella, Staphylococcus aureus and Streptococcus, and antioxidant activities against ferric-ion, ABTS and DPPH radicals, the perfect anti-inflammatory activity of TSE was demonstrated by significantly reducing the content of NO and the phagocytic activity in LPS induced RAW 264.7 cells, which provided a theoretical basis for the research and development of new anti-inflammatory Chinese medicine.
Keywords
Dioscorea nipponica Makino
Saponins
Extraction process
Effective constituents
Anti-inflammatory activity
- BBD
-
Box-Behnken design;
- LPS
-
lipopolysaccharide;
- TSC
-
total saponins content;
- TSE
-
total saponins extract
Abbreviations
1 Introduction
With the extensive use of antibiotics, antibiotic resistance is becoming more and more serious. At the same time, the residues of antibiotics in animal food also seriously affect people's quality of life (Ababneh and Alkhazali, 2019; Kumar et al., 2021). Antioxidant plays an important role in medicine and life, which is not only protecting easily oxidized drugs, but also inhibiting aging and immune stress (Agnieszka, 2021; Farid et al., 2020). Inflammation is a beneficial defense response against many stimulate, but an excessive inflammatory response may lead to some inflammatory diseases, such as pneumonia, chronic bronchitis and rheumatoid arthritis (Wang et al., 2014; Wang et al., 2020; Choi et al., 2018). At present, the main treatment is drug therapy from the steroidal and non-steroidal anti-inflammatory drugs in clinic, but there are also many adverse reactions, including gastrointestinal reactions and sticky damage (Baló-Banga et al., 2018; Dona et al., 2016). Therefore, it is an urgent to develop and research antioxidant, antibacterial, anti-inflammatory drugs from natural plants.
Dioscorea nipponica Makino (D. nipponica), as a species of the Dioscorea genus from the family Dioscoreaceae. (Ou-Yang et al., 2018), is a traditional Chinese medicine, and mainly produced in Northeast of China, Hebei and Shanxi provinces. It’s main active ingredients are saponins such as dioscin, pseudoprotodioscin and protodioscin, etc. (Ou-Yang et al., 2018; Tang et al., 2013; Tang et al., 2015; Li et al., 2010). Saponins have a variety of pharmacological effects, including anti-inflammatory (Yin et al., 2020; Zhang et al., 2016; Liu et al., 2017), antioxidant (Yang et al., 2019), anti-parasitic (Zhou et al., 2021), cardiac diseases (Li et al., 2021), anti-allergic diarrhea (Huang et al., 2021), treatment of gouty arthritis (Zhou et al., 2020), atherosclerotic cardiovascular (Sun et al., 2020), hepatorenal damages (Li Y et al., 2021), mastitis (Ran et al., 2020), skin cancer (Wang et al., 2020), diabetic nephropathy (Cai et al., 2020).
However, there was no systematic and comprehensive study on preparation process, main components, anti-inflammatory, antimicrobial and antioxidant activities of the total saponins extract (TSE) from D. nipponica. Thus, this experiment was conducted to research these gaps.
2 Materials and methods
2.1 Plant collection
D. nipponica was purchased from Shenyang Tasly Pharmacy and identified by licensed Chinese pharmacist Yao Cong. The medicinal materials were crushed and sifted through the screen for the further extraction.
2.2 Chemicals and reagents
2, 4, 6-tri (2-pyridyl)-s-triazine (TPTZ), 2, 2-diphenyl-1-picrylhydrazyl (DPPH) and 2, 2′ - azino - bis(3-ethylbenzothiazoline-6-sulfonic acid diammonium salt) (ABTS) were provided from Sigma-Aldrich Chemie (Steinheim, Germany) for the determination of antioxidant activities. Lipopolysaccharide (LPS) from Escherichia coli and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Nutritional broth and modified Edwards medium were bought form Beijing Solaibao Technology Co., ltd (Beijing, China), peptone soy broth and Salmonella Shigella agar medium from Qingdao Haibo Biotechnology Co., ltd (Qingdao, China). TLR4 inhibitor TAK-242 was offered by Wuhan Bode Biological Engineering Co., ltd (Wuhan, China). Other analytical chemicals and reagents were obtained from Beijing Dingguo Changsheng Biotechnology Co., ltd (Beijing, China).
2.3 Cells and microbial strains
RAW 264.7 cells were bought from the Shanghai Cell Bank of the Chinese Academy of Sciences (Shanghai, China).
Four standard strains, including Escherichia coli (ATCC 25922), Salmonella (ATCC 14028), Staphylococcus aureus (ATCC 29213) and Streptococcus (ATCC 49619) were purchased from American Type Culture Collection for antibacterial test, and stored at College of Animal Science and Veterinary Medicine, Shenyang Agricultural University.
2.4 Instruments
Liquid chromatograph analysis was carried out on Agilent 1290 Infinity II (Palo Alto, CA, USA), and the mass spectrometer analysis with Agilent 6546 QTOF. The data acquisition and processing were performed by MassHunter Quantitative Analysis 10.0 software.
2.5 Preparation method of TSE
A suitable amount (about 0.5 g) of fine powder from D. nipponica was accurately weighed and placed in a conical flask. The extraction time, ethanol concentration and ratio of solvent and material were confirmed according to the test scheme. The ultrasonic extraction was performed twice, and the filtrate was combined and concentrated to dryness for subsequent experiments.
2.6 Optimization the extraction process of TSE from D. nipponica
2.6.1 Determination of total saponins content (TSC)
Total saponins content (TSC) was measured by perchloric acid colorimetric method according to the report of Tian et al. (2020). A linear regression equation was drawn with absorbance (Y) of reference substance of diosgenin as ordinate and concentration (X) as abscissa, Y = 0.0236X-0.0634 (r = 0.9991). After the preparation of the extract solution, 0.5 ml of the extraction solution was taken for color reaction, and its absorbance value was determined at 410 nm. The concentration can be calculated when the measured absorbance value was inserted into the standard curve, and TSC was defined as follows: extraction yield of total saponins (w/w, %) = mass of total saponins expressed as g of diosgenin equivalents per g dry weight of D. nipponica samples × 100 %.
2.6.2 Optimization the extraction process of TSE by single factor experiments
Three single factors, including extraction time (X1, min), ethanol concentration (X2,%) and ratio of solvent and material (X3, ml/g), were investigated with the yield of TSE as response value. The five different levels of the three single factors were as follows: 20, 30, 40, 50 and 60 min for X1 factor, 20, 40, 60, 80 and 100 % for X2 factor, 20:1, 30:1, 40:1, 50:1 and 60:1 ml/g for X3 factor. When the influence of any single factor was investigated on the yield of TSE, the intermediate level was selected for the other two factors.
2.6.3 Optimization the extraction process of TSE by response surface methodology
According to the optimization results of single factor experiments, a three-factor and three-level response surface analysis experiment was designed by Box-Behnken method using Design Expert 8.0.6 with extraction time (X1, min), ethanol concentration (X2,%) and ratio of solvent and material (X3, ml/g) as independent variables and the yield of TSE as investigation index.
2.7 The compositions of TSE analyzed by UPLC-QTOF-MS
2.7.1 Sample preparation for HPLC-MS analysis
The extraction solution was prepared with 0.5 g of medicinal materials according to the optimal extraction process optimized by response surface experiments. The extraction solution was concentrated up to dryness, and the residue was dissolved in 10 ml of chromatographic methanol and filtered by 0.22 μm microfiltration membrane for UPLC-QTOF-MS analysis.
2.7.2 UPLC-QTOF-MS and LC-DAD conditions and parameters
The samples were separated and analyzed on C18 chromatographic column (Poroshell 120 series, 150 mm × 2.1 mm, i.d. 2.7 μm) with the mobile phases (A) water and (B) acetonitrile. The gradient elution procedure of the mobile phases was as follows: 90 %-70 % of A for 1–15 min, keeping 70 % of A for 5 min, 70 %-5% of A for 12 min, keeping 5 % of A for 3 min, and then 5 %-90 % of A for 1 min with a flow rate of 0.3 ml/min at column temperature 30 °C. The detection conditions for UPLC-QTOF-MS were set up with positive ion mode, drying gas flow 7 L/min and 250 °C, nebulizer pressure 35 psi, capillary voltage 3.5 KV, sheath gas flow 11 L/min and 325 °C.
2.8 Determination the antioxidant activities of TSE
The antioxidant activities of TSE from D. nipponica was evaluated by three methods, including ferric-ion reducing antioxidant power (FRAP) assay, ABTS radical scavenging activity assay and DPPH radical scavenging activity assay. The reducing ability and free radical scavenging ability of the extract were evaluated according to the specific operations reported in literature (Tian et al., 2018). 0.1 ml of different concentration of TSE was mixed with 0.4 ml of FRAP reagent and reacted for 30 min at 37 °C, and then the absorbance was measured at 593 nm. FRAP of TSE was expressed as mmol Fe2+ per 100 μg/ml. 0.1 ml of sample solution was added with 0.9 ml of ABTS or DPPH working solution, and the absorbance was measured at 734 nm or 517 nm after incubation for 30 min at room temperature or 37 °C, respectively. The scavenging ability was calculated by the following equation: [(A0-A1)/A0] × 100 %, where A0 was the absorbance of ABTS or DPPH, and A1 for ABTS or DPPH with sample. The free radical scavenging ability can be expressed by IC50 (mg/ml), which is TSE concentration of inhibition 50 % free radicals.
2.9 Determination the antimicrobial activities of TSE
2.9.1 Bacterium solution preparation
50 µl of Escherichia coli, Salmonella, Staphylococcus aureus and Streptococcus preserved in glycerin were inoculated onto Luria-Bertani nutrient broth, Salmonella Shigella agar, peptone soy broth and improved Edward medium, respectively. The plates were incubated at 37 °C for 30 min, and then inverted for further culture for 12–18 h. The typical single colony was selected and enriched in 3 ml of Mueller Hinton broth medium, and then cultured at 37 °C and 170 RPM until the logarithmic growth phase. The bacterial liquid was diluted with Nutrient Broth medium to 1 × 105CFU/ml and stored in a refrigerator at 4 °C for later use.
2.9.2 Samples preparation
The extract was prepared according to the optimized extraction process of response surface test and dissolved in sterile water to the concentration from 1.953 to 1000 µg/ml.
2.9.3 Antibacterial activity assay
The antibacterial activity of TSE was determined with microbroth dilution method according to the method reported by Tian et al.(2019), and the minimal inhibitory concentration (MIC) value was the major evaluation index. After the mixture of the TSE solution and the bacterial liquid with the volume ratio 1:1 incubation at 37 °C for 24 h, the inhibition of bacteria was observed with the antibiotic gentamicin as positive control and sterile water as negative control.
2.10 Determination the anti-inflammatory activities of TSE
2.10.1 Cell culture
RAW 264.7 cells were cultured in DMEM high glucose medium containing 10 % fetal bovine serum under the condition of 5 % CO2 and 37 °C.
2.10.2 Samples solution preparation
According to the optimized extraction process, TSE was prepared and dissolved with DMEM high glucose medium with the final concentration of 31.25, 62.5, 125 g/ml. After filtered with 0.22 μm of microfiltration membrane, the solution of TSE was preserved at −20 °C for later use.
2.10.3 Cell viability
MTT assay was adopted to evaluate the effect of TSE on cell viability based on the report by Tian et al. (2018). Cells were incubated with 6.25, 12.5, 25 μg/ml of TSE, DEX, TAK-242, LPS and DMEM high glucose medium for 18 h, respectively. After cells cultured with 20 μl of MTT reagent (5 mg/ml) for 4 h, the absorbance value was measured by microplate reader detection at 570 nm.
2.10.4 Measurement of NO content
According to the reported Griess reagent method (Tian et al., 2018), the cell grouping was the same as the assay of cell viability and the absorbance value of the reaction solution was measured at 540 nm after culturing for 18 h. The content of NO in cell supernatant was calculated by the established sodium nitrite standard curve.
2.10.5 Measurement of phagocytic activity
Based on the method reported in the literature (Tian et al., 2020), the effect of TSE on phagocytic activity was observed with neutral red dye. After cultured for 18 h and discarded the cell supernatant, the cells were mixed with 100 μl of neutral red solution (100 mg/ml, dissolved in normal saline) and cultured further in a cell incubator for 1 h. The supernatant was poured out and added with 100 μl of cell lysate for overnight at room temperature in darkness, and then the absorbance value was detected at 540 nm with a microplate analyzer.
2.11 Statistical analysis
The IC50 values were calculated by Prime 5 software, and other data was statistically analyzed with SPSS 17.0 software, and p > 0.05 expressed no significant difference.
3 Results and discussions
3.1 Optimization of extraction process of TSE from D. Nipponica
3.1.1 The optimization of single factor experiments
According to literatures (Hu et al., 2012; Luo et al., 2018; Ren et al., 2015) and preliminary experimental studies (Tian et al., 2020), three main factors, including extraction time, ethanol concentration and ratio of solvent and material, can affect the content of saponins extracted by ultrasonic extraction method. Therefore, this study mainly investigated the effects of the above three factors on the yield of TSE from D. nipponica.
The effects of five different levels of three factors on the extraction yield of TSE were shown in Fig. 1. When the extraction time reached 30 min, the yield of TSE was the highest, and decreased with the extension of the extraction time. When the ethanol concentration reached 40 %, the content of total saponins was higher than that of other concentration groups, which can be explained by similarity of polarity between saponins and 40 % ethanol. With the increase of ratio of solvent and material, the extraction ratio of TSE also will increase, and the highest content of total saponins was gained at ratio of solvent and material of 50:1, but TSC showed a trend of decline gradually when ratio of solvent and material than 50:1. This may be due to the increased dissolution of other impurity components.
Effect of extraction time (X1, min), ethanol concentration (X2,%), solvent to material (X3, ml/g) on the extraction yield of total saponins from D.nipponica.
According to the results of single factor test, three levels were selected for the next step of response surface analysis for the three factors, and the three levels are 25, 30, 35 min for extraction time (X1), 30, 40, 50 % for ethanol concentration (X2) and 45:1, 50:1, 55:1 for ratio of solvent and material (X3, ml/g), respectively.
3.1.2 Optimization of extraction conditions by a BBD
Based on the single-factor design, the Box-Behnken method was used to design a three-factor, three-level response surface analysis test using Design Expert 8.0.6 with extraction time (X1), ethanol concentration (X2) and liquid to material ratio (X3) as independent variables (see Table 1). The final equation of TSE was fitted out by multiple regression analysis as follows:
Y(%) = 1.28 + 0.060X1 + 0.054X2 + 0.039X3-0.13X1X2 + 0.030X1X3 + 0.097X2X3 + 0.27X12-0.030X22-0.080X32
Run
Variable levels
Response 1
(R1)
A
Extraction time
(min)B
Ethanol concentration
(%)C
Ratio of solvent
to material
(ml/g)
1
30
40
50
1.23
2
30
30
55
1.07
3
30
50
55
1.35
4
25
50
50
1.64
5
35
40
45
1.51
6
25
40
55
1.48
7
35
30
50
1.65
8
35
40
55
1.52
9
30
50
45
1.07
10
30
30
45
1.18
11
30
40
50
1.27
12
25
30
50
1.25
13
30
40
50
1.29
14
35
50
50
1.52
15
30
40
50
1.33
16
30
40
50
1.27
17
25
40
45
1.35
The results of further analysis of the fitted quadratic regression equation by precision, reliability and accuracy were shown in Table 2, the model had a P-value < 0.0001 (P < 0.01) and the Lack of Fit is 0.7101 (P > 0.05), which indicated that the model had good fitting accuracy and could be used for subsequent correlation analysis and prediction. The test error was low, which shown that the model can be used for subsequent correlation analysis and prediction; the correlation coefficient R2 = 0.9856 indicated a good correlation between the predicted and actual values; the value of Adj R-Squared and Pred R-Squared were 0.9671 and 0.9221; the coefficient of variation (C.V., 2.37 %) < 10 % showed a better model with perfect accuracy and precision (see Table 3).
Source
Sum of
SquaresDF
Mean Square
F Value
p-value,
Prob>FSignificant
model
0.49
9
0.055
53.33
< 0.0001
significant
A
0.029
1
0.029
27.98
0.0011
**
B
0.023
1
0.023
22.45
0.0021
**
C
0.012
1
0.012
11.67
0.0112
*
AB
0.068
1
0.068
65.68
< 0.0001
**
AC
0.0036
1
0.0036
3.50
0.1037
BC
0.038
1
0.038
36.94
0.0005
**
A2
0.30
1
0.30
292.17
< 0.0001
**
B2
0.004
1
0.004
3.74
0.0943
C2
0.027
1
0.027
26.34
0.0013
**
Residual
0.007
7
0.001
Lack of Fit
0.002
3
0.0006
0.49
0.7101
not significant
Pure Error
0.005
4
0.001
Cor Total
0.50
16
R2
0. 9856
Adj R2
0.9671
Pred R2
0.9221
Adeq Precision
24.232
C.V.%
2.37
No.
Retention time (min)
Formula
Experimental m/z of [M−H]-
Experimental m/z of [M + H]+
Molecular mass
Error (mDa)
Compounds’ name
1a
1.135
C11H12O7
255.0514
256.0587
−0.39
Tartaric acid
2
1.759
C51H84O22
1047.5385
1048.5466
−1.15
Protodioscin
3
1.779
C51H84O23
1063.5343
1064.5415
−1.21
Protogracillin
4
4.47
C12H14N2O4
251.1027
250.0956
−0.19
Cyclo-(Ser-Tyr)
5
7.048
C7H6O2
121.0295
122.0368
−0.02
Benzoic acid
6
28.074
C19H18O3
293.1185
295.1331
294.1258
−0.07b/-0.21c
1,7-bis(4-hydroxyphenyl)hepta-4E,6E-dien-3
7
28.112
C20H20O4
323.1292
325.1437
324.1364
−0.24/-0.27
7-(4-hydroxy-3-methoxyphenyl)-1-(4-hydroxyphenyl)-4E,6E-heptadien-3-one
8
30.25
C52H86O22
1061.5548
1062.5619
−0.87
Methyl protodioscin
9
30.417
C52H86O23
1077.549
1078.5577
−1.14
Methyl protogracillin
10
30.769
C51H82O22
1045.5232
1046.5305
−0.77
Pseudo protogracillin
11
31.986
C33H52O8
577.3739
576.3665
−0.33
Diosgenin-3-O-β-d-glucopyranoside
12
32.122
C18H32O3
293.2125
294.2198
−0.29
(5S,6E,8Z)-5-Hydroxy-6,8-octadecadienoic Acid
13
32.526
C27H38O3
411.2895
410.2821
−0.03
25-d-Spirosta −3,5-diene
14
32.985
C51H82O21
1029.5281
1031.5421
1030.5421
−0.54/0.25
Protogracellin
15
33.033
C45H72O16
867.4752
869.4894
868.484
−0.18/0.04
Dioscin
16
33.123
C45H72O17
883.4704
885.4840
884.4844
−0.44/-0.25
Gracillin
17
33.168
C39H62O12
723.4318
722.4245
−0.33
Prosapogenin A
18
33.553
C18H30O2
277.2175
278.2247
−0.14
α-Linolenic acid
19
33.975
C18H32O2
279.2331
280.2404
−0.2
Linoleic acid
20
34.309
C27H40O3
413.3054
412.2982
−0.44
Δ3,5-deoxytigogenin
21
34.511
C18H34O2
281.2488
282.2558
0.08
Oleic acid
22
34.835
C27H42O3
415.3208
414.3136
−0.18
Diosgenin
23
35.366
C18H36O2
283.2645
284.2714
0.09
Stearic acid
Fig. 2 described the effects of three factors on the yield of TSE with 3D response surface curves. As can be seen from Fig. 2a, when the ethanol concentration was a constant value, TSC declined at the begining and rose up in late, and was the highest at extraction time 25 min; while extraction time was constant, TSC increased with the increasing of ethanol concentration, and reached the maximum at 50 % ethanol concentration, and the interaction of extraction time and ethanol concentration had a greater effect on the yield of TSE (P < 0.0001).When the extraction time was constant, there was a positive correlation between TSC and the liquid to material ratio, TSC was the maximum value at the the liquid to material ratio 55:1 ml/g (Fig. 2b). TSC was increasing with the improving of ethanol concentration, and reached the maximum at 50 % ethanol concentration; and the interaction exhibited a greater effect on TSC between the ethanol concentration and the liquid to material ratio (P = 0.0005, Fig. 2c).
A Response surface plot of ethanol concentration (X1,%) and ratio of solvent to material (X2, ml/g) on the extraction yield of total saponin of D.nipponica; b Response surface plot of ratio of solvent to material (X2, ml/g) and extraction time (X3, min) on the extraction yield of total saponin of D.nipponica; c Response surface plot of ethanol concentration (X1,%) and extraction time (X3, min) on the extraction yield of total saponin of D.nipponica.
3.2 Chemical components analysis of TSE from D. Nipponica
In order to further reveal the pharmacodynamic substance basis of TSE from D. nipponica, the main chemical components of the extract were preliminarily inferred and analyzed by UPLC-QTOF-MS. According to the reported literature (Li and Leng, 2019), there were twenty-three compounds were inferred based on the information of formula, molecular mass, m/z of [M−H]- and [M + H]+, including thirteen steroidal saponins (protodioscin, protogracillin, methyl protodioscin, methyl protogracillin, pseudoprotogracillin, diosgenin-3-O-β-d-glucopyranoside, protogracellin, dioscin, gracillin, prosapogenin A, Δ3,5-deoxytigogenin, diosgenin, 25-d-Spirosta −3,5-diene), seven organic acid compounds (tartaric acid, benzoic acid, 5S,6E,8Z-5-Hydroxy-6,8-octadecadienoic acid, α-linolenic acid, linoleic acid, oleic acid, stearic acid), two flavonoids (1,7-bis (4-hydroxyphenyl) hepta-4E, 6E-dien-3,7- (4-hydroxy-3-methoxyphenyl)-1-(4-hydroxyphenyl)-4E,6E-heptadien-3-one), and one amino acid compound (CycloSer-Tyr). The preliminary inference of the main chemical components in the TSE from D. nipponica will provide an important reference for further study of pharmacological mechanism.
3.3 The antioxidant activities of TSE from D. Nipponica
In this study, three antioxidant assay methods, including FRAP, ABTS and DPPH radicals scavenging activity assays, were used to evaluate the antioxidant activity of TSE. TSE exhibited a milder antioxidant activity than BHT (IC50 DPPH 85.18 μg/ml, IC50 ABTS 31.90 μg/ml, 0.56 mmol Fe2+ for FRAP) with IC50 DPPH 2906 μg/ml, IC50 ABTS 74 μg/ml, 0.025 mmol Fe2+ for FRAP.
Relevant studies had confirmed the certain antioxidant activity of monomer saponins. Selim S (2016) indicated that the diosgenin showed an effective antioxidant activity against DPPH free radical than VC, and could significantly reduce the content of SOD, LPO and MDA (Kanchan DM, 2016; Kiasalari Z, 2017). Combined with the results of this study, it can be concluded that the antioxidant activity of saponins extract was weaker than that of than their monomeric compounds. However, the price of saponins crude extract is relatively cheap, and it will exhibit a good prospect of application as antioxidant in food and medicine industry.
3.4 The antibacterial activities of TSE from D. Nipponica
Microbroth dilution method was applied for evaluation the antibacterial activities of TSE from D. nipponica against Escherichia coli, Salmonella, Staphylococcus aureus and Streptococcus. MIC values were 62.5 mg crude drug/ml for Escherichia coli, Staphylococcus aureus and Streptococcus, and 125 mg crude drug/ml for Salmonella.
The antibacterial activities of monomer saponins and their new formulations were confirmed by related studies. The antimicrobial activity of diosgenin was demonstrated with a significant zone of inhibition against Gram-positive and Gram-negative bacteria (Khan H, 2015). SevincIlkar Erdagi (2020) found that Genipin crosslinked gelatin-diosgenin-nanocellulose hydrogels exhibited a excellent antibacterial effect towards Gram-positive and Gram-negative bacteria. However, there were few reports on the antibacterial activity of crude saponins. This study revealed the antibacterial activity of crude saponins, laying a foundation for the screening of green and healthy antibacterial drugs from Traditional Chinese medicine.
3.5 Evaluation of the anti-inflammatory effects of TSE on LPS induced RAW 264.7 cells
3.5.1 Effect of TSE on the cell viability of LPS induced RAW 264.7 cells
The cytotoxicity of TSE on RAW 264.7 cells was described in Fig. 4. The cell viability ratio was>98 %, which was indicated that three test concentrations of TSE, LPS, Dex, and TAK-242 all exhibited no cytotoxicity.
Chromatographic profile of the chemical components from TSE of D.nipponica at 204 nm.

Effect of TSE from D.nipponica on cell viability in LPS-induced RAW 264.7 cells.
3.5.2 Effect of TSE on the NO content in LPS induced RAW 264.7 cells
The effect of TSE from D. nipponica on the content of inflammatory mediator NO was described in Fig. 5. It can be seen that 6.25, 12.5 and 25 µg/ml of TSE can significantly reduce the level of NO with dose-dependent, and there was no significant difference between the 25 µg/ml of TSE treatment group with the control group.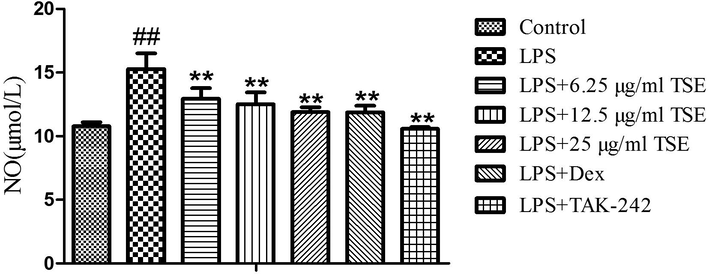
Effects of TSE from D.nipponica on the NO content in LPS induced RAW 264.7 cells (#compared with the control, *compared with LPS, **/ ## P < 0.01).
Nitric oxide, as an important inflammatory mediator, is a major reference index to evaluate the anti-inflammatory effect of drugs. A large number of studies had revealed that saponins can reduce the amount of nitric oxide produced in inflammatory response. The total saponins from Dioscorea nipponica Makino can decrease the content of NO in carbon tetrachloride-induced liver injury in mice (Yu et al., 2014). Diosgenin glucoside, as a relatively common monomer compound of saponins, can also suppress the synthesis of promote inflammation molecule NO in regulating microglial M1 polarization (Wang et al., 2017). Sapogenin diosgenin can exhibit anti-inflammatory properties by inhibiting the production of NO induced by IL-1β (Wang et al., 2015). This study revealed that the extract can reduce the content of NO, which was consistent with literature reports. However, which signaling pathways play a regulatory role needs to be further revealed in future studies.
3.5.3 Effect of TSE on the phagocytic activity of LPS induced RAW 264.7 cells
The phagocytic activity of RAW 264.7 cells after treatment with 6.25, 12.5 and 25 µg/ml of TSE from D. nipponica was shown in Fig. 6. The phagocytic capacity of the LPS model group was very significantly (P < 0.01) up-regulated compared to the control group, and significantly (0.05 < P < 0.01) down-regulated in three drugs groups compared to the LPS model group with a dose-dependent trend. The difference between the 25 µg/ml of TSE and the control group was not significant (P > 0.05).
Effects of TSE from D.nipponica on phagocytic index in LPS induced RAW 264.7 cells (#compared with the control, *compared with LPS, **/ ## P < 0.01).
Cytophagocytosis is the function of certain cells in organism to recognize foreign body and swallow it and destroy it, which is the basic defense mechanism of organism. Phagocytosis plays a major role in the genesis and development of inflammation. Therefore, it is often used as an important index to measure the anti-inflammatory effect of drugs. In a certain concentration range, the phagocytosis activity increased with the increasing concentration of diosgenin 3-O-α-l-rhamnopyranosyl(1 → 4)-α-l-rhamnopyranosyl (1 → 4)-[α-l-rhamnopyranosyl (1 → 2)-β-d-glucopyranoside, diosgenin 3-O-α-l-rhamnopyranosyl (1 → 2)-α-l-arabino furanosyl-β-d-glucopyranoside and diosgenin, but with the further increasing of dosage, the phagocytosis activity began to decrease (Zhang et al., 2007). Studies had also been reported that dioscin and diosgenin all can induce macrophage M2-to-M1 phenotype or lymphocyte transformation, and enhance phagocytic capability of macrophages in vitro (Cui et al., 2020; He et al., 2012). TSE can exert anti-inflammatory effects by directly affecting phagocytosis activity, but the mechanism of anti-inflammatory effects needs to be further explored in the future.
4 Conclusion
This research comprehensively explored the extraction process, main active ingredients, and the biological activities of TSE from D. nipponica in vitro. The optimal extraction process of TSE was extraction time 25 min, 50 % ethanol solution, and the ratio of solvent to material 55:1 (ml/g); eighteen compounds were preliminarily inferred with UPLC-QTOF-MS method, including thirteen steroidal saponins, two organic acids, two flavonoids, and one amino acid. Although TSE showed mild antibacterial activities against Escherichia coli, Salmonella, Staphylococcus aureus and Streptococcus, and antioxidant activities against ferric-ion, ABTS and DPPH radicals, the stronger anti-inflammatory activity of TSE was demonstrated by significantly reducing the content of NO and the phagocytic activity.The above research content laid a preliminary foundation for further research on the anti-inflammatory effect and mechanism of TSE from D. nipponica, and provided an important reference for the screening of new anti-inflammatory Chinese medicines.
Acknowledgments
We are grateful for the financial support from Scientific Research Fund of Liaoning Province Education Department (Grant No.LSNQN202004).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- The impact of antibiotic abuse: health and economic burden. Biomed. J. Sci. Tech. Res.. 2019;16(1):11794-11797.
- [Google Scholar]
- Application of natural antioxidants in the oxidative stabilization of fish oils: A mini-review. J. Food Process. Pres.. 2021;45(4)
- [Google Scholar]
- Baló-Banga, J.M., Schweitzer, K. 2018. Nemszteroid gyulladáscsökkentők okozta adverz reakciók differenciáldiagnosztikája. In vitro és in vivo módszerek [The differential diagnosis of adverse reactions to non-steroidal anti-inflammatory drugs. In vitro and in vivo methods]. Orvosi hetilap, 159(38), 1556-1566.
- Dioscin protects against diabetic nephropathy by inhibiting renal inflammation through TLR4/NF-κB pathway in mice. Immunobiology.. 2020;225(3):151941
- [Google Scholar]
- Bis (3-bromo-4,5-dihydroxybenzyl) ether, a novel bromophenol from the marine red alga Polysiphonia morrowii that suppresses LPS-induced inflammatory response by inhibiting ROS-mediated ERK signaling pathway in RAW 264.7 macrophages. Biomed Pharmacother.. 2018;103:1170-1177.
- [Google Scholar]
- Cui, L.Y., Yang, G.D., Ye, J.N., Yao, Y.N., Lu, G.H., Chen, J.J., Fang, L.J., Lu, S., Zhou, J.Y. 2020. Dioscin elicits anti-tumour immunity by inhibiting macrophage M2 polarization via JNK and STAT3 pathways in lung cancer. J Cell Mol Med. Aug;24(16):9217-9230.
- Hypersensitivity reactions to non-steroidal anti-inflammatory drugs. Curr Pharm Des.. 2016;22(45):6784-6802.
- [Google Scholar]
- Genipin crosslinked gelatin-diosgenin-nanocellulose hydrogels for potential wound dressing and healing applications. Int J Biol Macromol.. 2020;15(149):651-663.
- [Google Scholar]
- Assessment of antioxidant, immune enhancement, and antimutagenic efficacy of fennel seed extracts in irradiated human blood cultures. J. Radiat. Res. Appl. Sci.. 2020;13(1):260-266.
- [Google Scholar]
- Anti-tumour and immunomodulating activities of diosgenin, a naturally occurring steroidal saponin. Nat. Prod. Res.. 2012;26(23):2243-2246.
- [Google Scholar]
- Optimization of ultrasonic-assisted extraction of total saponins from Eclipta prostrasta L. using response surface methodology. J. Food Sci.. 2012;77(9):975-982.
- [Google Scholar]
- Anti-allergic diarrhea effect of diosgenin occurs via improving gut dysbiosis in a murine model of food allergy. Molecules.. 2021;26(9):2471.
- [Google Scholar]
- Kanchan, D.M., Soman,i G.S., Peshattiwar, V.V., Kaikini, A.A., Sathaye, S. Renoprotective effect of diosgenin in streptozotocin induced diabetic rats. Pharmacol Rep. 2016 Apr;68(2):370-7.
- Antimicrobial and inhibition on heat-induced protein denaturation of constituents isolated from Polygonatum verticillatum rhizomes. Nat Prod Res.. 2015;29(22):2160-2163.
- [Google Scholar]
- Diosgenin ameliorates development of neuropathic pain in diabetic rats: Involvement of oxidative stress and inflammation. Biomed Pharmacother.. 2017 Feb;86:654-661.
- [Google Scholar]
- Determination of antibiotic residues in bovine milk by HPLC-DAD and assessment of human health risks in Northwestern Himalayan region. India. J. Food Sci. Technol. 2021:1-10.
- [Google Scholar]
- Li J., Leng, J.H. 2019. Network pharmacological mechanism of Dioscorea nipponica Makino in the treatment of painful diabetic peripheral neuropathy. Chinese patent med.41(10):9.
- Dioscin ameliorates methotrexate-induced liver and kidney damages via adjusting miRNA-145-5p-mediated oxidative stress. Free Radic Biol Med.. 2021;169:99-109.
- [Google Scholar]
- Anti-thrombotic activity and chemical characterization of steroidal saponins from Dioscorea zingiberensis C.H. Wright. Fitoterapia.. 2010;81(8):1147-1156.
- [Google Scholar]
- Dioscin and diosgenin: Insights into their potential protective effects in cardiac diseases. J Ethnopharmacol.. 2021;28(274):114018
- [Google Scholar]
- Protodioscin ameliorates oxidative stress, inflammation and histology outcome in Complete Freund's adjuvant induced arthritis rats. Apoptosis.. 2017;22(11):1454-1460.
- [Google Scholar]
- Evaluation of the nitric oxide and nitrite scavenging capability, n-nitrosamine formation inhibitory activity, and pptimization of ultrasound-assisted aqueous two-phase system extraction of total saponins from Coreopsis tinctoria flowering tops by response surface methodology. App. Biochem. Biotech.. 2018;184(3):763-776.
- [Google Scholar]
- Dioscorea nipponica Makino: a systematic review on its ethnobotany, phytochemical and pharmacological profiles. Chem Cent J.. 2018;12(1):57.
- [Google Scholar]
- Dioscin Improves Pyroptosis in LPS-Induced Mice Mastitis by Activating AMPK/Nrf2 and Inhibiting the NF-κB Signaling Pathway. Oxid Med Cell Longev.. 2020;2020:8845521.
- [Google Scholar]
- Microwave-assisted extraction and a new determination method for total steroid saponins from Dioscorea zingiberensis C.H. Wright. Steroids. 2015;104:145-152.
- [Google Scholar]
- Anti-inflammatory, antioxidant and antiangiogenic activities of diosgenin isolated from tradit ional medicinal plant, Costus speciosus (Koen ex. Retz.) Sm. Nat Prod Res.. 2016 Aug;30(16):1830-1833.
- [Google Scholar]
- Estrogenic and anti-inflammatory effects of pseudoprotodioscin in atherosclerosis-prone mice: Insights into endothelial cells and perivascular adipose tissues. Eur J Pharmacol.. 2020;15(869):172887
- [Google Scholar]
- Cardioprotective effect of total saponins from three medicinal species of Dioscorea against isoprenaline-induced myocardial ischemia. J Ethnopharmacol.. 2015;4(175):451-455.
- [Google Scholar]
- Quantitative comparison of multiple components in Dioscorea nipponica and D. panthaica by ultra-high performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry. Phytochem Anal.. 2013;24(4):413-422.
- [Google Scholar]
- Extraction process, component analysis, and in vitro antioxidant, antibacterial and anti-inflammatory activities of total flavonoids extracts from Abutilon theophrasti Medic. leaves. Mediat. Inflamm.. 2018;2:1-17.
- [Google Scholar]
- Extraction technology, component analysis, and in vitro antioxidant and antibacterial activities of total flavonoids and fatty acids from Tribulus terrestris L. fruits. Biomed. Chromatogr.. 2019;33:4474.
- [Google Scholar]
- Anti-inflammatory activity in vitro, extractive process and HPLC-MS characterization of total saponins extract from Tribulus terrestris L. fruits. Ind. Crops Prod.. 2020;150:112343
- [Google Scholar]
- Anti-inflammatory effects of (-)-epicatechin in lipopolysaccharide stimulated raw 264.7 macrophages. Trop. J. Pharm. Res.. 2014;13:1415-1419.
- [Google Scholar]
- Luteolin alters macrophage polarization to inhibit inflammation. Inflammation.. 2020;43(1):95-108.
- [Google Scholar]
- Diosgenin inhibits IL-1β-induced expression of inflammatory mediators in human osteoarthritis chondrocytes. Int J Clin Exp Pathol.. 2015;8(5):4830-4836.
- [Google Scholar]
- Antitumor effects of dioscin in A431 cells via adjusting ATM/p53-mediated cell apoptosis. DNA damage and migration. Oncol Lett.. 2020;21(1):59.
- [Google Scholar]
- Diosgenin glucoside provides neuroprotection by regulating microglial M1 polarization. Int Immunopharmacol.. 2017;50:22-29.
- [Google Scholar]
- Recent Advances in the Pharmacological Activities of Dioscin. Biomed Res Int.. 2019;14:5763602.
- [Google Scholar]
- A New NLRP3 Inflammasome Inhibitor, Dioscin. Promotes Osteogenesis. Small.. 2020;16(1):e1905977.
- [Google Scholar]
- Protective effects of the total saponins from Dioscorea nipponica Makino against carbon tetrachloride-induced liver injury in mice through suppression of apoptosis and inflammation. Int Immunopharmacol.. 2014;19(2):233-244.
- [Google Scholar]
- Immuno-stimulating properties of diosgenyl saponins isolated from Paris polyphylla. Bioorg. Med. Chem. Lett.. 2007;17(9):2408-2413.
- [Google Scholar]
- Potential neuroprotection of protodioscin against cerebral ischemia-reperfusion injury in rats through intervening inflammation and apoptosis. Steroids.. 2016;113:52-63.
- [Google Scholar]
- Antiparasitic efficacy of herbal extracts and active compound against Gyrodactylus kobayashii in Carassius auratus. Front Vet Sci.. 2021;6(8):665072
- [Google Scholar]
- Anti-inflammation effects of the total saponin fraction from Dioscorea nipponica Makino on rats with gouty arthritis by influencing MAPK signalling pathway. BMC complementary medicine and therapies.. 2020;20(1):261.
- [Google Scholar]







