Translate this page into:
Paclobutrazol ameliorates cigarette smoke-induced chronic obstructive pulmonary disease (COPD) in experimental rats
⁎Corresponding author at: Department of Pulmonary and Critical Care Medicine, The First Affiliated Hospital of Xi’an Medical University, Xi’an City, Shaanxi Province, China. doctor_dx@sina.com (Xin Diao)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Background
The cigarette smoking epidemic is one of the desperate threats experienced all over the world. Cigarettes cause diseases such as chronic obstructive pulmonary disease (COPD), asthma, lung cancer, stroke, heart diseases, etc. Paclobutrazol is a growth regulator hormone that increases antioxidant enzymes, mineral absorption, carbohydrate synthesis, and flowering and fruit production in plants.
Objective
In the current study, we assessed the ameliorative effect of paclobutrazol against chronic cigarette smoke-induced COPD in rats.
Methodology
A side-stream cigarette exposure rat model was used for the study, which is a well-accepted model to evaluate cigarette smoke-induced lung damage. The paclobutrazol treatment was given for 12 weeks, and since the reduction in body weight is the primary symptom of COPD, the food efficiency and the weight gain were measured. Muscular strength was analyzed with a grip strength test, and respiratory functions were assessed with a whole-body plethysmograph. Plasma leptin was measured to detect the fat mass index. C-reactive protein was quantified to assess the level of inflammation. Further bronchoaleveolar fluid was collected and analyzed for the total white blood cell count, neutrophils, lymphocytes, and monocytes to evaluate the inflammation. In order to confirm the anti-inflammatory property of paclobutrazol against cigarette smoke-induced lung inflammation, histopathological analysis of lung tissue was done.
Results
The paclobutrazol treatment increased the body weight, improved respiratory functions, and decreased inflammation in the rats exposed to secondhand cigarette smoke. Our results of histopathological analysis confirm that paclobutrazol has effectively inhibited cigarette smoke-induced lung tissue damage in young Wistrar rats.
Conclusion
Hence, it can be used as a supplementary drug to protect smokers from cigarette smoke-induced lung damage.
Keywords
Smokers
Cigarette
Lung inflammation
Cachexia
Azole drug
Paclobutrazole
1 Introduction
Cigarette smoking is one of the foremost reasons of global preventable mortalities. Every year, more than 7 million mortalities were recorded due to the usage of cigarettes (Hersi et al., 2019; Reid et al., 2016). 80% of the global smoker’s population belongs to the underdeveloped nations (Global burden of diseases, 2021). Worldwide, one in seven adults is addicted to cigarette smoking (Peacock et al., 2018). Smoking not only affects the cigarette smokers but also has a great impact on the health of secondhand smokers. Smoking is a triggering factor for diseases like respiratory illness, cancer, cardiovascular diseases, stroke, osteoporosis, and neonatal deaths (Hersi et al., 2019; WHO, 2017; Mons et al., 2015; Fischer and Kraemer (2015)).
Triazole compounds are boon for plants which protects them from various stress via inducing antioxidant enzyme synthesis (Jaleel et al., 2007). One such azole compound is paclobutrazol. Paclobutrazol [(2RS, 3RS)-1-(4-chlorophenyl)-4,4-dimethyl-2-(1H-1, 2, 4-trizol-1-yl)-pentan-3-ol] (Fig. 1) is a growth regulator which increases the levels of antioxidant enzymes in plants species like Sesamum indicum (Somasundaram et al., 2009), Arachis hypogaea (Sankar et al., 2007), Vigna unguiculata (Manivannan et al., 2007; Srivastava et al., 2010). It reduced lipid peroxidation in Curcuma alismatifolia (Jungklang and Saengnil 2012). Paclobutrazol also increases the levels of non enzymatic antioxidants such as α-tocopherol, ascorbic acid in stress induced plants (Manivannan et al., 2007; Soumya et al., 2017). The antioxidant property of paclobutrazol was well established in plants whereas the beneficiary effect of paclobutrazol in animals was not yet studied. The protective effect of paclobutrazol against the cigarette exposed lung inflammation was analyzed.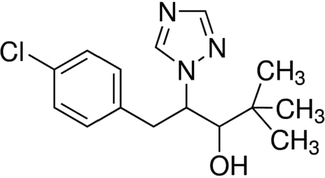
Molecular structure of the paclobutrazol.
Chronic obstructive pulmonary disease, which exemplifies the pathology of airway obstruction due to the infiltration of macrophages and neutrophils, is reported in most cigarette smokers. The toxic chemicals in cigarettes enhance the infiltration of immune cells into the lungs, thereby leading to lung inflammation (Zhang et al. (2020); Celli et al. (2022)). Cigarette smoke induces inflammation of the airways, which leads to obstruction of air flow in COPD patients (Rosenwasser et al., 2022). Apart from pulmonary pathology, COPD patients exhibit other systemic manifestations like reduced body weight, malnutrition, cachexia, and skeletal muscle wasting (De Brandt et al. (2022); Taivassalo and Hepple, 2022). The animals exposed to cigarette smoke exhibited the same pathological symptoms observed in COPD patients (Fricker et al. (2014); Diaz Del Valle et al. (2022); Tomoda et al., 2012). Hence, in the present study, we used a side-stream cigarette-exposed rat model to analyze the ameliorative effect of paclobutrazol against cigarette smoke-induced COPD in rats.
2 Materials & methods
2.1 Chemicals
Paclobutrazol, dexamethasone, and other chemicals were procured from Sigma Aldrich, USA. The assay kits for biochemical assays were obtained from CyrstalChem, USA.
2.2 Experimental animals
Healthy male Wistar rats, aged five weeks and weighing about 115–125 g, were used for the current study. The procedures were explained in detail before the ethical committee, and the same was followed in the present study. The rats were housed in hygienic laboratory conditions maintained at 25 ± 2 °C, 55% relative humidity, and a 12 h light/dark cycle that was strictly maintained. Polycarbonate cages were used to house the rats, and rice husks were used for bedding. The bedding was changed daily and every two days once the cages were changed. The rats were fed ad libidum with a standard laboratory rat pellet diet and reverse osmosis water. The rats were acclimatized for a week in laboratory condition before the initiation of the experiment. The rats were treated with the utmost care and concern; all the experiments were done according to the guidelines of the animal ethical committee (14–2021), Xi’an Medical University, Xi’an City, Shaanxi Province, China.
2.3 Experimental design
The acclimatized rats were randomly divided into five groups, each consisting of 10 rats. Group I are control group rats, which are untreated; Group II are positive control rats exposed to side stream cigarette smoke alone; Group III rats exposed to side stream cigarette smoke were simultaneously treated with 10 mg/kg bodyweight (b.wt) of paclobutrazol; Group IV rats exposed to side stream cigarette smoke were simultaneously treated with 20 mg/kg b.wt of paclobutrazol; and Group V rats exposed to side stream cigarette smoke were simultaneously treated with 2 mg/kg b.wt of standard anti-inflammatory drug dexamethasone. The treatment was conducted for 12 weeks duration.
2.4 Cigarette exposure
Side-stream cigarette smoke was exposed to rats using the whole-body smoke exposure chamber. Twice a day, every morning at 9o'clock and evening at 3o’clock, the rats were exposed to cigarette smoke. The rats were exposed to cigarette smoke for 3 h, with 10 min intervals. About 8–10 cigarettes were exposed to the rats each day, and the same brand of cigarettes, Scissors, was used throughout the experiment. Smoke-free air was exposed to control rats for the same duration twice a day (Anbarasi et al. (2005)).
2.5 Measurement of body weight gain
The body weight and the food intake of rats were measured daily throughout the treatment period. On the day 21 and 77 the 1 h food intake after the treatment was measured. The food efficiency of rats were calculated using the below formula.
2.6 Assessment of grip strength
The grip strength of the rats was measured using the instrument Bioseb’s Grip strength (BIO-GS3). The instrument was positioned horizontally on the table and the rats were positioned on to it by holding the tail. The rats were allowed to grab the metal rod and then pulled backwards in the horizontal plane. The force applied by the rats towards the grids just before the rats lose its grip was recorded. The experiment was conducted on the last day of treatment before injecting drug.
2.7 Evaluation of respiratory functions
On the last day of treatment before the paclobutrazol injection, the rats were subjected to respiratory functional analysis. When the rat was awake, the tidal volume and the peak expiratory flow rate of rats were analyzed using the whole-body plethysmograph with a respiratory function analysis system (Buxco Electronics, USA). The rats were anesthetized to record the functional residual capacity and forced expiratory volume at 100 ms with a forced maneuvers system (Bio-System for Maneuver, Data Sciences International).
2.8 Estimation of plasma leptin & C-reactive protein
After the treatment period, the rats were euthanized, and the blood was collected for the estimation of plasma leptin and C-reactive protein. Blood was collected in the EDTA tubes for the separation of plasma. The blood samples were centrifuged at 1500g for 15 min at 4 °c. The supernatant was collected and stored for further analysis. Plasma leptin and C-reactive protein levels were measured using the commercially available ELISA kit purchased from CyrstalChem,USA. The test was performed according to the instructions provided in the kit.
2.9 Assessment of bronchoalveolar lavage fluid
The anesthetized rats were subjected to the collection of bronchoalveolar lavage fluid. The rats were placed on a clean surgical table, and the chest of each rat was cut open with a surgical knife. The left lung bronchus was ligated, and 4 mL of saline was administered to the trachea and gently withdrawn using a canula. The collected fluid was considered to be the bronchoalveolar lavage fluid. The bronchoalveolar fluid was centrifuged at 500g for 10 min at 4 °C. The pellet was collected and suspended in 1 mL of saline. To the suspension, Turk’s fluid was added, and 1 µl of fluid was subjected to a total WBC count using a hemocytometer. The pellet suspension was mounted on a glass slide and stained with Wright-Giemsa stain to count the monocytes, neutrophils, and lymphocytes.
2.10 Histopathological analysis of lung tissue
The rat’s chest was cut open, and 10% buffered formalin was injected into the lungs at a constant pressure of 25 cm H2O for 4 h.The left lower lobe of the lung was excised and immersed in formalin. The tissue was further subjected to tissue processing and finally embedded in paraffin wax. The paraffin-embedded tissue blocks were sectioned into 5 µm using a microtome. The sections were stained with haematoxylin and eosin stain and microscopically observed for histopathological changes.
2.11 Statistics
The data of the all the experiments were analyzed with one Way ANOVA followed by the post hoc test Student’s-t test using the statistical software GraphPad Prism. The results were expressed as mean ± SEM. p < 0.05 was considered to be statistically significant.
3 Results
3.1 Weight gaining effect of growth regulator paclobutrazol in chronic cigarette smoke induced rats
Fig. 2A&1B depict the results of the paclobutrazol effect on weight gain and the efficiency of food intake in chronically side-stream cigarette-exposed rats. For the first two weeks, not much difference was observed in the weights between the control, cigarette smoke-exposed, and simultaneous paclobutrazol-treated rats, whereas a significant decrease in body weight was observed in only the cigarette smoke-exposed rats from the 7th week onward. After the 12-week treatment period, more cumulative weight gain was observed in the paclobutrazol and dexamethasone-treated rat group than in the cigarette smoke-exposed rats. The food intake, 1 h food intake, and food efficiency were significantly decreased in the cigarette smoke alone group compared to the other groups. Paclobutrazol treatment significantly increased the food intake compared to the cigarette smoke alone that exposed rats.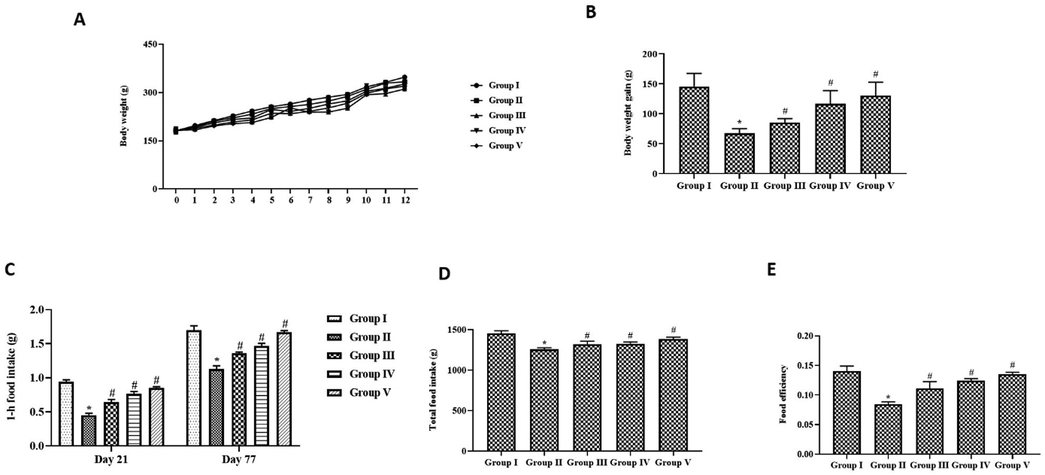
Weight gaining effect of growth regulator paclobutrazol in chronic cigarette smoke induced rats. (A) Body weight (B) Cummulative body weight gain (C) 1-h food intake (D) Total food intake (E) Food efficiency. p < 0.05 was considered to be statistically significant.
3.2 Muscular strengthening effect of growth regulator paclobutrazol in chronic cigarette smoke induced rats
The muscular strengthening effect of paclobutrazol in cigarette smoke-exposed rats was measured using a grip strength meter. Cigarette smoke alone exposed rats, which showed decreased grip strength compared to the sham control rats. Simultaneous paclobutrazol-treated rats exposed to cigarette smoke showed significantly increased grip strength compared to cigarette-alone-exposed rats. No significant change was observed between the paclobutrazol and dexamethasone-treated rats (Fig. 3).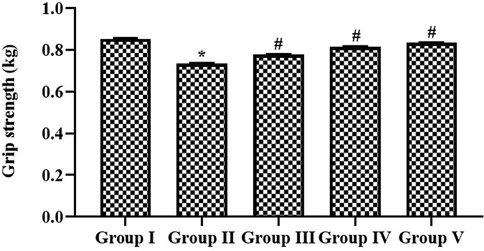
Muscular strengthening effect of growth regulator paclobutrazol in chronic cigarette smoke induced rats. Skeletal muscle strength of control and experimental rats were assessed with Bioseb’s Grip strength instrument. p < 0.05 was considered to be statistically significant.
3.3 Ameliorative effect growth regulator paclobutrazol on respiratory functions in chronic cigarette smoke induced rats
Fig. 4 illustrates the results of respiratory functions analyzed in control and experimental rats using a whole-body plethysmograph. Forced expiratory volume, peak expiratory flow rate, and tidal volume were decreased in the exposed rats to cigarette smoke alone. Simultaneous paclobutrazol treatment increased respiratory functions in rats. Paclobutrazol treatment decreased the lung capacity and functional residual capacity compared to exposure to cigarette smoke alone in rats.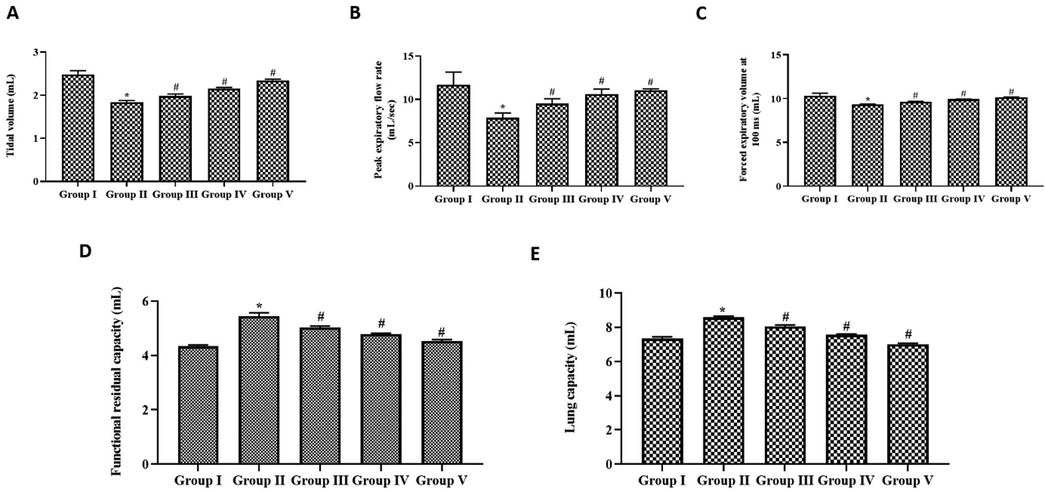
Ameliorative effect growth regulator paclobutrazol on respiratory functions in chronic cigarette smoke induced rats. Respiratory functions of control and experimental rats were analyzed with whole body plethysmograph instrument. (A) Tidal volume (B) Peak expiratory flow rate (C) Forced expiratory volume at 100 ms (D) Functional residual capacity (E) Lung capacity. p < 0.05 was considered to be statistically significant.
3.4 Regulatory effect of growth regulator paclobutrazol on plasma leptin & C-Reactive protein in chronic cigarette smoke induced rats
The fat mass index of control and experimental rats was assessed by measuring the levels of plasma leptin (Fig. 5A). Simultaneous paclobutrazol treatment significantly increased the plasma leptin levels in cigarette smoke-exposed rats. The increase in plasma leptin was observed in a dose-dependent manner in paclobutrazol-treated rats. Fig. 5B depicts the results of C-reactive protein, an inflammatory marker, in control and experimental rats. No significant difference was observed between control, paclobutrazol-treated cigarette-exposed rats, and dexamethasone-treated cigarette-exposed rats. C-reactive protein levels were higher in rats exposed to side stream cigarette smoke alone compared to other treatment groups.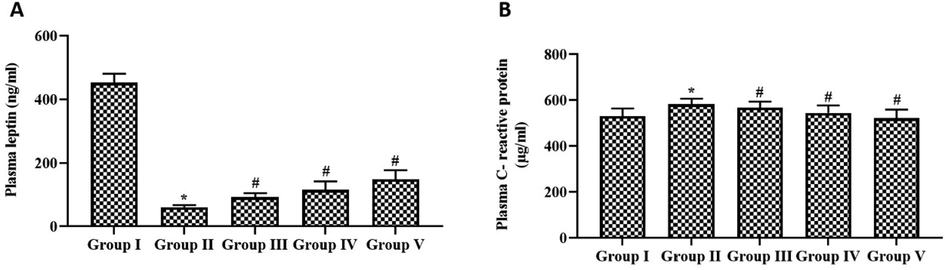
Regulatory effect of growth regulator paclobutrazol on plasma leptin & C-Reactive protein in chronic cigarette smoke induced rats. Fat mass index plasma leptin (A) and inflammatory marker protein C-reactive protein (B) were assessed with commercially available ELISA kit. p < 0.05 was considered to be statistically significant.
3.5 Anti-inflammatory effect of growth regulator paclobutrazol in chronic cigarette smoke induced rats
The anti-inflammatory effect of paclobutrazol was assessed by counting the white blood cells, and the results are depicted in Fig. 6. When cigarette smoke alone was exposed to rats, the total WBC count increased dramatically when compared to control rats. The number of monocytes was decreased, whereas the number of neutrophils and lymphocytes was increased in cigarette-exposed rats. Paclobutrazol treatment significantly decreased the counts of neutrophils and lymphocytes and increased the monocyte count in cigarette-smoke exposed rats. No significant difference was observed between paclobutrazol and dexamethasone-treated rats.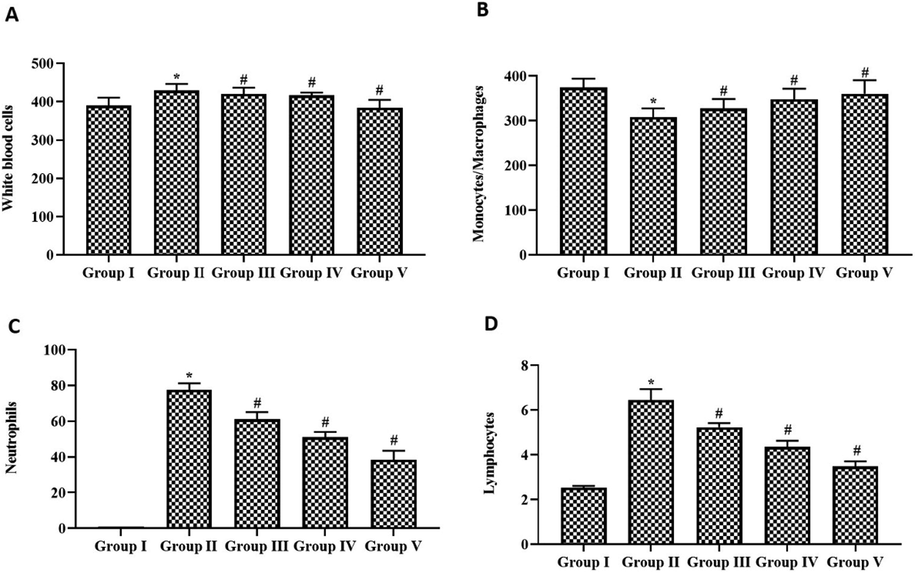
Anti-inflammatory effect of growth regulator paclobutrazol in chronic cigarette smoke induced rats. Bronchoalveolar fluid was collected to analyze (A) Total WBC count (B) Monocytes (C) Neutrophils (D) Lymphocytes. p < 0.05 was considered to be statistically significant.
3.6 Pulmonary protective effect of growth regulator paclobutrazol in chronic cigarette smoke induced rats
Fig. 7 illustrates the representative image of an H&E-stained lung tissue section from control, cigarette smoke-exposed, simultaneous paclobutrazol, and dexamethasone treated rats. Normal lung architecture with undisturbed alveoli and interstitium (6A), whereas cigarette smoke alone exposed rats shown thickened interstitum and alveolar wall due to infiltration of inflammatory cells (6B). The number of disturbed alveolar cells was reduced in paclobutrazol (6C&D) and dexamethasone (6E)-treated rats lung tissue compared to the cigarette smoke alone exposed rats.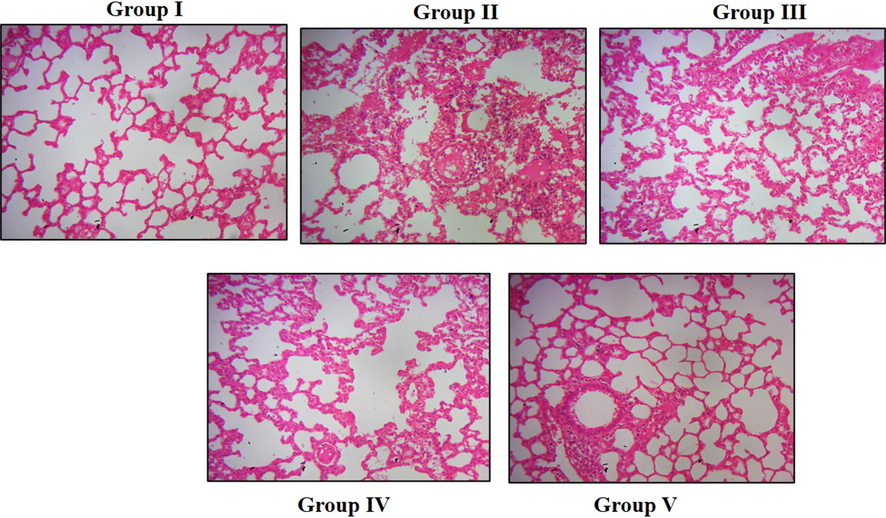
Effect of paclobutrazol on the lung tissues of the chronic cigarette smoke-induced rats The lung tissue was excised from control and experimental rats. The tissue was processed and stained with hameatoxylin and eosin stain. The stained sections were viewed under light microscope and representative images were depicted. (Group I) Control (Group II) Cigarette smoke alone exposed (Group III) Cigarette smoke exposed rats simultaneously treated with 10 mg/kg paclobutrazol (Group IV) Cigarette smoke exposed rats simultaneously treated with 20 mg/kg paclobutrazol (Group V) Cigarette smoke exposed rats simultaneously treated with 2 mg/kg dexamethasone.
4 Discussion
Even though the usage of cigarettes was reduced in developed countries, the middle- and low-income countries still experienced serious health ill effects due to smoking. About 7 million deaths were recorded every year due to cigarette smoking (Hersi et al., 2019; WHO 2015). Cigarettes consist of more than 4000 ingredients, many of which are toxic and cause illness in both smokers and secondhand smokers. Cigarette smoking amplifies the risk of diseases such as respiratory disease, stroke, cancer, inflammatory diseases, diabetes, fatty liver, premature skin ageing, etc. (Ahmed et al., 2020; Lee et al., 2018; Lisboa et al., 2017). COPD is a common disease found in more than 80% of cigarette smokers. Cigarette smoke induces oxidative stress and the infiltration of inflammatory cells, thereby causing severe lung damage (Miller et al., 2014).
Cachexia is a rapid loss of body weight observed in patients suffering from chronic illness (He et al., 2022). Apart from causing respiratory dysfunction, cigarette smoking also decreases the appetite, thereby causing cachexia in healthy individuals (Ho et al., 2022; Thome et al., 2022). 20–40% of COPD patients were reported with cachexia, and the mortality rate in COPD patients with cachexia was high (von Haehling & Anker, 2012; Zhang et al. (2020)). In our study, 12 weeks of side-stream cigarette exposure in healthy male rats reduced their food intake. Nearly 50% of the reduction in cumulative weight gain was observed in rats exposed to secondhand cigarette smoke. The reduction in body weight gain may be due to a loss of appetite induced by the cigarette smoke. Food efficiency and total food intake decreased in cigarette smoke alone, which confirms the loss of appetite. Whereas simultaneous treatment with paclobutrazol increased the intake of food and efficiency in rats, thereby preventing rats from cigarette smoke-induced cachexia.
Cigarette smoking causes skeletal muscle dysfunction, which is reported in smokers without any symptom of respiratory illness (Maltais et al., 2013). COPD patients are prone to skeletal muscle wasting and peripheral muscle damage, which leads to decreased everyday life activity. Therefore, muscle weakness should be considered a serious pathological symptom to be treated on time. Our study results on grip strength assessment in rats also prove that cigarette smoke induces muscle wasting in rats, which causes decreased muscular strength. Both the standard anti-inflammatory drug dexamethasone treatment and paclobutrazol significantly increased the muscular strength in cigarette-exposed rats.
Paclonutrazol treatment significantly increased the respiratory functions in rats treated with cigarette smoke exposure. The forced expiratory volume, tidal volume, and peak expiratory flow rate were increased in rats treated with simultaneous paclobutrazol. The improvement in respiratory function was observed in a dose-dependent manner. The lung capacity and functional residual capacity, which are often increased in emphysema, were also observed in rats treated with cigarette smoke, whereas paclobutrazol treatment decreased the lung capacity and thereby prevented rats from developing cigarette smoke-induced emphysema. Our results correlate with the previous report: gherlin treatment significantly improved lung capacity in whole-body cigarette-exposed rats (Kamiide et al., 2015).
Leptin is an adipokine secreted by the adipocytes that enhances weight gain, and during fasting conditions, the levels of plasma leptin are drastically reduced (Kryfti et al., 2015). The plasma leptin levels were higher in smokers compared to nonsmokers. Cessation of smoking increased food intake and decreased energy expenditure, which proves the correlation between smoking and levels of plasma leptin, which decreases body weight (Driva et al. (2022)). In our study, paclobutrazol treatment decreased the leptin levels in rats exposed to cigarette smoke. This may be the reason for increased food intake and body weight gain in paclobutrazol rats compared to the cigarette smoke alone-exposed rats. C-reactive protein is a plasma protein synthesized by the liver during inflammatory conditions. Increased levels of C-reactive proteins were observed in smokers (Dahdah et al. (2022)). A positive correlation was reported between leptin and C-reactive protein levels (Suleri et al., 2022; Mayyas et al., 2020). In our study, cigarette exposure increased the levels of C-reactive protein, which may be due to the increase in plasma leptin. Paclobutrazol treatment significantly decreased both levels of plasma leptin and C-reactive protein, thereby protecting rats from cigarette smoke-induced inflammation.
It has been reported that the number of white blood cells was increased in smokers compared to non-smokers, which may be due to the increased infiltration of neutrophils to the inflamed site (Suzuki et al., 2022). During inflammation, the cytokine leptin also triggers the activation, infiltration, and survival of inflammatory cells (Kumboyono et al., 2022). Our results also illustrate that cigarette smoke increased the white blood cell count in rats, and the paclobutrazol treatment significantly decreased the WBC count. Further to confirm the anti-inflammatory property of paclobutrazol, we have performed the histopathological analysis of lung tissue. Our results confirmed paclobutrazol effectively inhibited the cigarette smoke-induced inflammation, thereby preventing lung inflammation in the rats.
5 Conclusion
To conclude, in the present study, 12 weeks of treatment with side-stream cigarette smoke in rats decreased food intake, efficiency, and weight gain. It also increased the levels of plasma leptin, C-reactive protein, and white blood cells and decreased the respiratory function in rats, which are characteristic pathologies of COPD. Paclobutrazol treatment significantly increased cumulative body weight, improved respiratory function, and decreased inflammation in cigarette-exposed rats. Overall, our results confirm that paclobutrazol effectively protects the rats from cigarette smoke-induced pulmonary inflammation. Hence, it could be used as a talented medication to treat the patients with COPD. However, additional studies are still needed in the future to understand the precise underlying molecular pathways of the therapeutic effects of paclobutrazol.
Funding
Effect of smoking on CD8 + T cells in serum of patients with chronic obstructive pulmonary disease level: Natural Science Foundation of Shaanxi Province Item Number: 2016SF-066.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- The effects of smoking on IgE, oxidative stress and haemoglobin concentration. Asian Pac. J. Cancer Prev.. 2020 Apr 1;21(4):1069-1072.
- [Google Scholar]
- Creatine kinase isoenzyme patterns upon chronic exposure to cigarette smoke: protective effect of Bacoside A. Vascul. Pharmacol.. 2005 Jan;42(2):57-61.
- [Google Scholar]
- Definition and nomenclature of chronic obstructive pulmonary disease: time for its revision. Am. J. Respir. Crit. Care Med.. 2022 Dec 1;206(11):1317-1325.
- [Google Scholar]
- Immunological insights into cigarette smoking-induced cardiovascular disease risk. Cells. 2022 Oct 11;11(20):3190.
- [Google Scholar]
- Update on the etiology, assessment, and management of COPD Cachexia: considerations for the clinician. Int. J. Chron. Obstruct. Pulmon. Dis.. 2022 Nov;18(17):2957-2976.
- [Google Scholar]
- Impact of airline secondhand tobacco smoke exposure on respiratory health and lung function decades after exposure cessation. Chest. 2022 Sep;162(3):556-568.
- [Google Scholar]
- The effect of smoking cessation on body weight and other metabolic parameters with focus on people with type 2 diabetes mellitus. Int. J. Environ. Res. Public Health. 2022 Oct 14;19(20):13222.
- [Google Scholar]
- Meta-analysis of the association between second-hand smoke exposure and ischaemic heart diseases, COPD and stroke. BMC Public Health. 2015 Dec;1(15):1202.
- [Google Scholar]
- Animal models of chronic obstructive pulmonary disease. Expert. Opin. Drug Discov.. 2014 Jun;9(6):629-645.
- [Google Scholar]
- Global Burden of Disease [database].Washington, DC: Institute of Health Metrics; 2019. IHME,accessed 17 July 2021.
- Impacts of Cigarette Smoke (CS) on Muscle Derangement in Rodents-a systematic review. Toxics. 2022 May 18;10(5):262.
- [Google Scholar]
- Effectiveness of stop smoking interventions among adults: protocol for an overview of systematic reviews and an updated systematic review. Syst. Rev.. 2019 Jan 19;8(1):28.
- [Google Scholar]
- Effects of cigarette smoke exposure on a mouse model of multiple sclerosis. Toxicol. Rep.. 2022 Mar;29(9):597-610.
- [Google Scholar]
- Jaleel CA, Gopi R, Manivannan P, Panneerselvam R. , C. A., Gopi, R., Manivannan, P., & Panneerselvam, R. Responses of antioxidant defense system of Catharanthus roseus (L.) to paclobutrazol treatment under salinity. Acta Physiologiae Plantarum. 2007; 29:205–209
- Jungklang J, Saengnil K. Effect of paclobutrazol on patumma cv. Chiang Mai Pink under water stress. Songklanakarin. Journal of Science and Technology. 2012, 34: 361–366
- Ghrelin ameliorates catabolic conditions and respiratory dysfunction in a chronic obstructive pulmonary disease model of chronic cigarette smoke-exposed rats. Eur. J. Pharmacol.. 2015 May;15(755):88-94.
- [Google Scholar]
- Effects of smoking cessation on serum leptin and adiponectin levels. Tob. Induc. Dis.. 2015 Sep;3(13):30.
- [CrossRef] [Google Scholar]
- Detection of vascular inflammation and oxidative stress by cotinine in smokers: measured through interleukin-6 and superoxide dismutase. Int. J. Gen. Med.. 2022 Sep;16(15):7319-7328.
- [Google Scholar]
- E-cigarette smoke damages DNA and reduces repair activity in mouse lung, heart, and bladder as well as in human lung and bladder cells. Proc. Natl. Acad. Sci. U. S. A.. 2018 Feb 13;115(7):E1560-E1569.
- [Google Scholar]
- Effects of cigarette smoke exposure during suckling on food intake, fat mass, hormones, and biochemical profile of young and adult female rats. Endocrine. 2017 Jul;57(1):60-71.
- [Google Scholar]
- Rationale for earlier treatment in COPD: a systematic review of published literature in mild-to-moderate COPD. COPD. 2013;10(1):79-103.
- [CrossRef] [Google Scholar]
- Changes in antioxidant metabolism of Vigna unguiculata (L.) Walp. by propiconazole under water deficit stress. Colloids Surf. B Biointerfaces. 2007 May 15;57(1):69-74.
- [Google Scholar]
- Comparison of the cardiac effects of electronic cigarette aerosol exposure with waterpipe and combustible cigarette smoke exposure in rats. Life Sci.. 2020 Jun;15(251):117644
- [Google Scholar]
- Inhaled anticoagulation regimens for the treatment of smoke inhalation-associated acute lung injury: a systematic review. Crit. Care Med.. 2014 Feb;42(2):413-419.
- [Google Scholar]
- Mons U, Müezzinler A, Gellert C, Schöttker B, Abnet CC, Bobak M, de Groot L, Freedman ND, Jansen E, Kee F, Kromhout D, Kuulasmaa K, Laatikainen T, O'Doherty MG, Bueno-de-Mesquita B, Orfanos P, Peters A, van der Schouw YT, Wilsgaard T, Wolk A, Trichopoulou A, Boffetta P, Brenner H; CHANCES Consortium. Impact of smoking and smoking cessation on cardiovascular events and mortality among older adults: meta-analysis of individual participant data from prospective cohort studies of the CHANCES consortium. BMJ. 2015 Apr 20;350:h1551.
- Global statistics on alcohol, tobacco and illicit drug use: 2017 status report. Addiction 2018
- [Google Scholar]
- Therapeutic approaches for Chronic Obstructive Pulmonary Disease (COPD) exacerbations. Pathogens. 2022 Dec 10;11(12):1513.
- [Google Scholar]
- Effect of paclobutrazol on water stress amelioration through antioxidants and free radical scavenging enzymes in Arachis hypogaea L. Colloids Surf. B Biointerfaces. 2007 Nov 15;60(2):229-235.
- [Google Scholar]
- Role of Paclobutrazol and ABA in drought stress amelioration in Sesamum indicum L. Global J. Mol. Sci.. 2009;4:56-62.
- [Google Scholar]
- Paclobutrazol: a novel plant growth regulator and multi-stress ameliorant. Ind. J. Plant Physiol.. 2017;22(3):267-278.
- [Google Scholar]
- Effect of paclobutrazol and salinity on ion leakage, proline content and activities of antioxidant enzymes in mango (Mangifera indica L.) Scient. Horticult.. 2010;125:785-788.
- [Google Scholar]
- The long-term impact of elevated C-reactive protein levels during pregnancy on brain morphology in late childhood. Brain Behav. Immun.. 2022 Jul;103:63-72.
- [Google Scholar]
- Response biomarkers of inhalation exposure to cigarette smoke in the mouse lung. J. Toxicol. Pathol.. 2022 Jul;35(3):247-254.
- [Google Scholar]
- Integrating mechanisms of exacerbated atrophy and other adverse skeletal muscle impact in COPD. Front. Physiol.. 2022 Jun;3(13):861617
- [Google Scholar]
- Chronic aryl hydrocarbon receptor activity phenocopies smoking-induced skeletal muscle impairment. J. Cachexia Sarcopenia Muscle. 2022 Feb;13(1):589-604.
- [Google Scholar]
- Changes of ghrelin and leptin levels in plasma by cigarette smoke in rats. J. Toxicol. Sci.. 2012 Feb;37(1):131-138.
- [Google Scholar]
- Cachexia as major underestimated unmet medical need: facts and numbers. Int. J. Cardiol.. 2012;161:121-123.
- [Google Scholar]
- World Health Organization (WHO) (2015) WHO Global Report on Trends in Prevalence of Tobacco Smoking; WHO: Geneva, Switzerland
- World Health Organization (WHO) (2017) WHO report on the global tobacco epidemic, 2017: monitoring tobacco use and prevention policies. http://www.who.int/tobacco/global_report/2017/en/.
- Ameliorative effect of acetylshikonin on cigarette smoke-induced lung inflammation in mice. J. Asian Nat. Prod. Res.. 2020 Nov;22(11):1078-1094.
- [Google Scholar]







