Translate this page into:
Phytochemical profiles and protein glycation inhibitory activities of three oak species
⁎Corresponding author. jwjeong@hnibr.re.kr (Jin-Woo Jeong)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The Quercus genus (oaks) comprises valuable plant resources that are used in many fields, including cosmetics, foods, and pharmaceuticals. Nonetheless, overall chemical profiling and anti-glycation component identification have not been thoroughly performed. In the present study, 70 % ethanolic and water extracts from three oak species (Quercus dentata, Q. serrata, and Q. aliena) showed antioxidant and anti-glycation potential. Thus, the components of the three oak species were profiled using ultra-performance liquid chromatography coupled with electrospray ionization and quadrupole time-of-flight mass spectrometry. The analysis showed that phenolic acids, ellagitannins, ellagic acid, procyanidin, and flavonoid (quercetin, myricetin, kaempferol, isorhamnetin, and taxifolin) glycosides are the main phenolic compounds. Based on the fluorescence assay, they act as strong inhibitors of non-enzymatic advanced glycation end-products (AGEs) formation in the bovine serum albumin (BSA) and skin proteins (collagen and elastin). Furthermore, mass fragmentation analysis demonstrated that ellagitannin, procyanidin B1 and flavonoid glycosides effectively trapped methylglyoxal (MGO), a reactive carbonyl intermediate and an important precursor of AGEs, to generate mono-, di-, or tri-MGO. Collectively, ellagic acid, ellagitannins, procyanidin B1, and flavonoid glycosides, the main active ingredients of three oak species (Q. dentata, Q. serrata, and Q. aliena), may be employed as lead structures in the development of functional foods or drugs to prevent diseases caused by aging and excessive sugar consumption.
Keywords
Quercus
Oak
Anti-glycation
Methylglyoxal-adduct
Ellagitannin
Advanced glycation end products
UPLC-Q-TOF-MS
1 Introduction
The Maillard reaction (MR) is a complex series of chemical reactions between reducing sugars and proteins that leads to protein browning, fluorescence, and crosslinking (Jia et al., 2023; Mazumder et al., 2019). Advanced glycation end products (AGEs), which are produced in the later stages of the MR, can accumulate in long-lived tissue proteins, such as collagen and lens crystallin, and may play a role in the development of metabolic illnesses such as diabetes, diabetic complications, and neuronal disorders (Liu et al., 2022). Reducing sugars, including glucose and fructose, react non-enzymatically with the H2N-protein through a three-step non-enzymatic protein glycation process. The carbonyl group (–COOH) of glucose combines with the H2N-protein to form Schiff bases, followed by a stable Amadori rearrangement product (ARP) (Grillo and Colombatto, 2008). These ARPs then undergo various irreversible dehydration and rearrangement reactions leading to the formation of reactive carbonyl compounds (RCCs) such as methylglyoxal (MGO). Subsequently, RCCs react with H2N, guanidine, and SH groups in proteins, ultimately generating irreversible AGEs (Chaudhuri et al., 2018; Choei et al., 2004). Recent studies have suggested that AGEs derived from α-hydroxyaldehyde, such as glyceraldehyde (GLAD) and glycoaldehyde, show stronger neurotoxicity and stronger promotion of radical formation than AGEs derived from glucose (Takeuchi et al., 2000). In addition, the browning activity due to α-hydroxyaldehyde was approximately 2000 times greater than glucose in the early stage of the MR. These GLADs are formed from GLAD-3-phosphate, an intermediate of glycolysis, or fructose through the polyol pathway or food intake (Choei et al., 2004). The identification of active substances that can reduce carbonyl stress (accumulation of RCCs) and prevent AGE formation is thus vital.
The Quercus genus comprises tree species commonly named oaks and belongs to the Fagaceae family. The genus contains more than 450 species distributed worldwide. Oaks are used as natural antioxidants in food preservation and play an important role in wine maturation in oak barrels (Morales, 2021). Several studies have demonstrated that oak extracts possess antioxidant, antiseptic, antibacterial, antifungal, anti-acne, anti-inflammatory, antibiofilm, and anti-diabetic properties (Burlacu et al., 2020; Othón-Díaz et al., 2023; Taib et al., 2020). Particularly, Quercus mongolica and its constituents (ellagic acid and kaempferol derivatives) showed inhibitory activities against AGE formation and digestive enzymes including α-glucosidase and α-amylase (Yin et al., 2018). The bioactive components of oaks include polar or moderately polar compounds (e.g., ellagic acid, tannins, flavonoids, flavonoid glycosides, and phenolic acids), and nonpolar compounds (e.g., terpenoids, fatty acids, and sterols) (Morales, 2021). However, the overall phytochemical profile and identification of the anti-glycation compounds of Quercus dentata Thunb, Quercus serrata Murray, and Quercus aliena Blume have not yet been completed. This study aimed to address this gap in our knowledge by profiling the constituents of the leaves, stems, and fruits from these oak species using ultra-performance liquid chromatography coupled with electrospray ionization quadrupole time-of-flight mass spectrometry (UPLC-ESI-Q-TOF-MS); furthermore, we aimed at clarifying their antioxidant and protein (serum albumin, collagen, and elastin) glycation inhibitory potential in vitro.
2 Materials and methods
2.1 Chemicals and reagents
In this study, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS), bovine serum albumin (BSA), monosaccharides, and methylglyoxal solution (40 %, v/v), thioflavin T (ThT) and standards including aminoguanidine HCl (AG) and l-ascorbic acid were all purchased from Sigma-Aldrich (St. Louis, MO, USA). 1,1-Diphenyl-2-picrylhydrazyl radical (DPPH) was purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). All the solvents used for instrumental analysis were purchased from Merck (Darmstadt, Germany).
2.2 Plant materials
Quercus dentata Thunb and Q. serrata Murray were collected from the Aphaedo and Chupodo Islands (Shinan-gun, Jeollanam-do, Republic of Korea), respectively, in July 2021. Quercus aliena Blume was collected from Jindo Island (Jindo-gun, Jeollanam-do, Republic of Korea) in August 2021. The plants were authenticated by Dr. S Han of the Honam National Institute of Biological Resources (voucher no. shan2021-16 for Q. dentata, shan2021-48 for Q. serrata, and shan2021-95 for Q. aliena). The 70 % ethanol (EtOH) and aqueous extracts from the different parts (leaves, fruits, twigs) of three oak species were received from the Bank of Bioresources from Island and Coast (BOBIC), Republic of Korea (registered no. HNIBR NP40, NP42, NP154, and NP156 for Q. dentata; NP145, NP147, NP148, NP150, NP169, and NP171 for Q. serrata; and NP259, NP261, NP262, NP264, NP289, and NP291 for Q. aliena).
2.3 UPLC-ESI-Q-TOF-MS analysis
Separation of 70 % EtOH and the aqueous extract from the different organs of the three oak species studied was performed using a C18 column (1.7 μm, 2.1 × 100 mm) and Waters ACQUITY UPLC I-Class Plus System (Waters Corporation, Milford, MA, USA). The gradient mobile phase consisted of MS-grade water (A) and MS-grade acetonitrile (ACN) (B), with 0.1 % MS-grade HCOOH: 0–10 min, B 2 % to 11 %; 10–12 min, B 11 to 11 %; 12–14 min, B 11 to 20 %; 14–20 min, B 20 to 23 %; 20–35 min, B 23 to 50. The column was heated to 30 °C, and the sample injection volume was 1 μL (0.2 mL/min flow rate). Mass analysis was conducted using Q-TOF-MS (Xevo G2-XS, Waters Corporation, Milford, MA, USA) with a mass range of 50–1500 Da in the negative ion mode, at 40 V cone voltage, and 350 ℃ desolvation temperature; further, 0.5 mM HCOONa and 0.2 ng/mL [Leu5]-enkephalin were used for mass calibration and correction.
2.4 Antioxidant assay
The radical-scavenging activities extracted from the different organs of the three oak species under study were assessed using the ABTS and DPPH radical scavenging method, as previously described, with l-ascorbic acid as the standard (Dudonné et al., 2009).
2.5 Protein glycation inhibitory assays
The BSA glycation inhibitory assay was conducted as previously described (Seong et al., 2021; Vinson and Howard, 1996). To formulate the glucose/fructose-induced AGEs reaction solution (ARS), 0.01 g/mL BSA in 0.05 M sodium phosphate buffer (pH 7.4) was added to 0.4 M monosaccharides, which include d-(−)-fructose and d-(+)-glucose, and 0.02 % NaN3. Various doses of extracts from the three oak species and their constituents were added to the ARS, and the mixtures were incubated at 37 °C for one week. To formulate the BSA-fructose model (gBSAfru), 0.01 g/mL BSA in 0.05 M sodium phosphate buffer (pH 7.4) was added to 0.4 M d-(−)-fructose and 0.02 % NaN3 (Yamaguchi et al., 2000). Various doses of AG and oak-derived constituents were added to the gBSAfru, and the mixtures were incubated at 50 °C for 72 h. A microplate reader (Biotek, Winooski, VT, USA) was used to measure AGEs generation at excitation and emission wavelengths of 350 and 450 nm, respectively. Glycation of collagen and elastin was induced by glyceraldehyde, and the anti-glycation activities of the test compounds were determined following manufacturer instructions (Cosmo Bio Co., Ltd., Carlsbad, USA). AG was used as a reference substance for the protein glycation assays (Thornalley, 2003).
2.6 Fluorescence assay for AGEs and protein oxidation products
The specific AGEs produced in the gBSAfru model were measured by fluorescence at their characteristic excitation and emission wavelengths: argpyrimidine (APN) at 320 and 380 nm, crossline (CL) at 380 and 440 nm, pentosidine (PEN) at 335 and 385 nm, and vesperlysine (VL) at 366 and 442 nm (Awasthi and Saraswathi, 2016). Additionally, the protein oxidation products generated in the gBSAfru model were also measured by fluorescence at their characteristic excitation and emission wavelengths: dityrosine (di-Tyr) at 330 and 415 nm, N′-formyl kynurenine (NFK) at 325 and 434 nm, and kynurenine at 365 and 480 nm (Zhang et al., 2022). Fluorescence from the glycated samples were measured by scanning with excitation wavelengths at 335 and 370 nm and emission wavelengths in the range of 350–600 nm.
2.7 β-Amyloid protein aggregation assay
Glycated protein (gBSAfru) was incubated with 20 μg/mL ThT solution at 37 °C for 1 h (Bouma et al., 2003). Subsequently, the fluorescence spectra of the reaction mixture were measured by scanning with an excitation wavelength at 435 nm and emission wavelengths in the range of 460–600 nm, with intensity recorded at 485 nm for the calculation of the inhibition rate.
2.8 Analysis of MGO adducts of the components from the three oak species under physiological conditions
MGO was mixed with components from different parts of the three oak species at a molar ratio of 3:1 in 1 × phosphate-buffered saline (PBS) (Ni et al., 2021). After 24 h of incubation at 37 °C, the compound–MGO reaction solution was filtered using a 0.2 μm pore syringe and the filtrate was analyzed a 5-μm reversed-phase analytical column (250 × 4.6 mm, C18 silica) and high-performance liquid chromatography (HPLC) (Ultimate™ 3000, ThermoScientific, Pittsburgh, PA, USA). The gradient mobile phase consisted of MS-grade water (A) and MS-grade ACN (B), with 0.1 % MS-grade HCOOH: 0–65 min, B 5 %–25 %. The column was heated to 30 °C, and the sample injection volume was 2.5 μ L (0.5 mL/min flow rate). MS conditions were the same as those mentioned in Section 2.3.
2.9 Statistical analysis
The 50 % effective and inhibitory concentrations (EC50 and IC50, respectively; μg/mL or μM) obtained from dose/inhibition curves were expressed as means ± standard deviation (SD) obtained from three independent experiments.
3 Results
3.1 Antioxidant and anti-glycation potentials of the extracts from the three oak species
To determine the antioxidant and anti-glycation potentials of the three oak species studied herein, the ABTS·+ and DPPH radical-scavenging assay and the monosaccharide-induced BSA glycation assay were conducted with appropriate reference compounds (Fig. 1). Aqueous extracts of the different organs of the Q. dentata, Q. serrata, and Q. aliena, showed strong antioxidant activity, with more than 90 % ABTS·+ and DPPH radical-scavenging activity at 50 μg/mL. In addition, the 70 % ethanolic extracts of Q. dentata (leaves and twigs), Q. serrata (leaves, twigs, and fruits), and Q. aliena (leaves and twigs) showed strong antioxidant activity, similar to that of the aqueous extracts. However, the 70 % ethanolic extract of the Q. aliena fruit showed no activity at the tested concentrations. For the anti-glycation activity, all extracts, except for the 70 % ethanolic extract from the Q. aliena fruits, showed good inhibition against monosaccharide-induced AGE formation. Among them, the 70 % ethanolic and aqueous extracts derived from the Q. aliena twigs showed the strongest anti-glycation, with IC50 values of 7.06 and 7.49 μg/mL, respectively.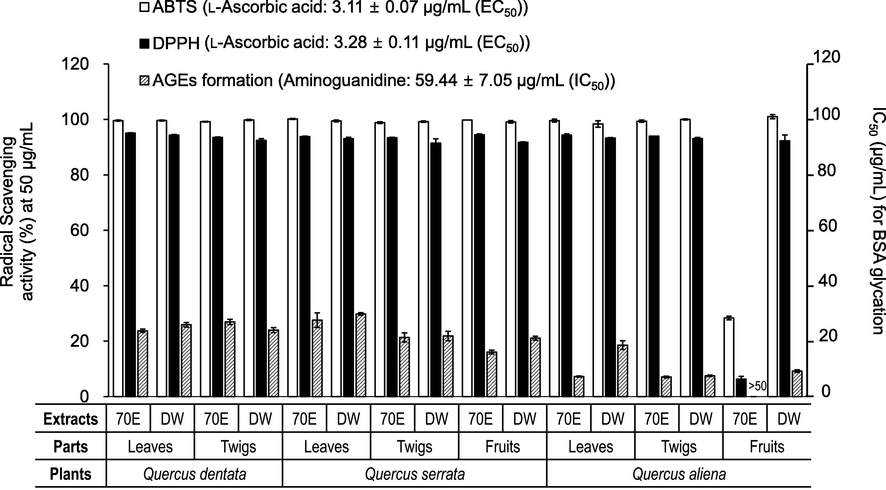
Antioxidant and AGEs formation (BSA/monosaccharide system) inhibitory activities of 70% EtOH (70E) and water (DW) extracts from different parts of three oak species (Quercus dentata, Q. serrata, and Q. aliena).
3.2 UPLC-ESI-Q-TOF-MS profiles of the three oak species
The chemical constituents of the leaves, twigs, and fruits from the three oak species (Q. dentata, Q. serrata, and Q. aliena) were analyzed using UPLC-ESI-Q-TOF-MS (Figs. 2–3 and Table 1). As a result, 47 phytochemicals—eleven phenolic acids (1–11), ten ellagitannins (12–21), ellagic acid (22), two catechins (25, 26), two B-type procyanidins (23, 24), five taxifolin derivatives (27–29, 32, 36), three myricetin derivative (30, 31, 33), six quercetin derivatives (34, 35, 37, 39, 41, 43), four kaempferol derivative (38, 40, 44, 45), one isorhamnetin derivative (42), and two liganas (46 and 47)—were detected in the negative ion mode of the MS and tentatively identified by interpreting the high-resolution mass spectra (fragmentation patterns obtained from high energy function).
The UPLC-MS base peak intensity (BPI) chromatogram of various extracts from Quercus dentata (A), Q. serrata (B), and Q. aliena (C).
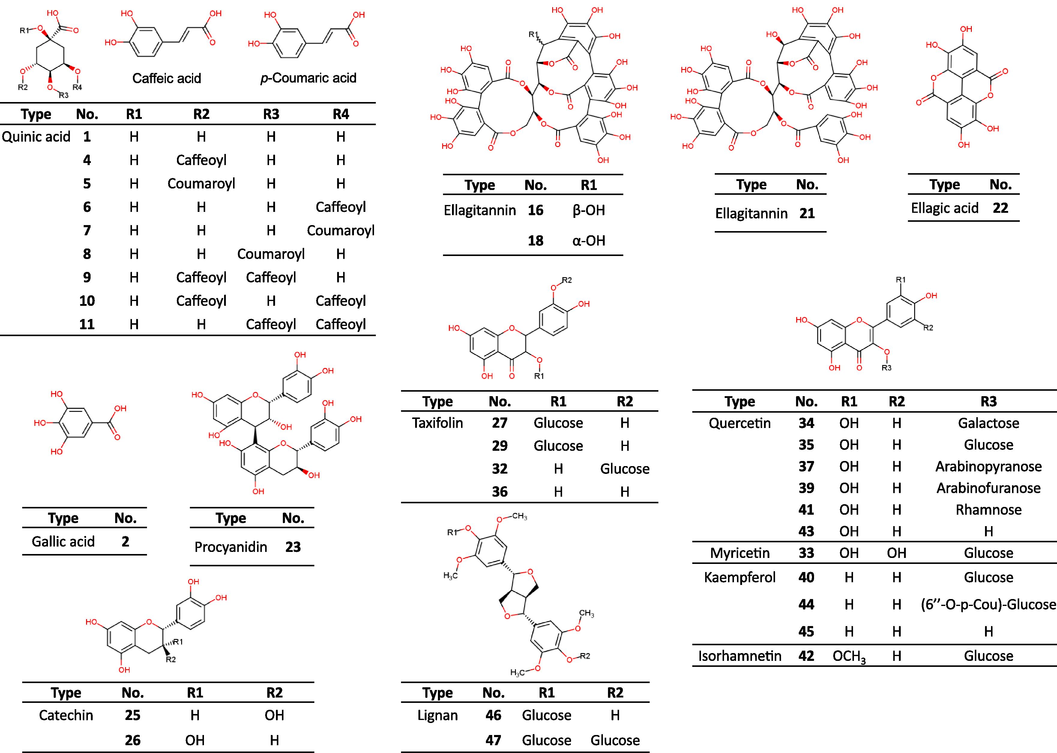
Structures of the phytochemicals derived from Quercus dentata, Q. serrata, and Q. aliena.
Type
No.
RTa (min)
[M–H]− (m/z)
Error (ppm)
Molecular Formula
MS Fragment
Phenolic class
Putative structure
I
1
1.23
191.0559
1.6
C7H12O6
Phenolic acid
Quinic acid*
2
3.08
169.0137
0.0
C7H6O5
Phenolic acid
Gallic acid*
3
3.38
343.0672
2.0
C14H16O10
191.0559
Phenolic acid
Galloylquinic acid
4
6.89
353.0899
7.4
C16H18O9
191.0559
Phenolic acid
Neochlorogenic acid*
5
8.86
337.0919
−1.2
C16H18O8
119.0500, 163.0387, 191.0559
Phenolic acid
3-O-Coumaroylquinic acid*
6
9.50
353.0899
−1.2
C16H18O9
191.0559
Phenolic acid
Chlorogenic acid*
7
12.15
337.0919
−1.2
C16H18O8
191.0559
Phenolic acid
5-O-Coumaroylquinic acid*
8
12.86
337.0919
−1.2
C16H18O8
119.0500, 163.0387, 173.0458, 191.0559
Phenolic acid
4-O-Coumaroylquinic acid*
9
16.78
515.1187
−0.6
C25H24O12
173.0458, 179.0353, 191.0559
Phenolic acid
3,4-Dicaffeoylquinic acid*
10
17.13
515.1187
−0.6
C25H24O12
179.0353, 191.0559
Phenolic acid
3,5-Dicaffeoylquinic acid*
11
18.02
515.1187
−0.6
C25H24O12
173.0458, 179.0353, 191.0559
Phenolic acid
4,5-Dicaffeoylquinic acid*
II
12
3.23
783.0709
3.6
C34H24O22
300.9988, 481.0636
Ellagitannin
Di-HHDP-glucose(pedunculagin) isomer I
13
3.63
1101.0721
2.5
C48H30O31
300.9988,
Ellagitannin
Valoneoyl-NHTP-glucose isomer I
14
4.44
1101.0721
2.5
C48H30O31
300.9988,
Ellagitannin
Valoneoyl-NHTP-glucose isomer I
15
4.49
783.0709
3.6
C34H24O22
300.9988, 481.0636
Ellagitannin
Di-HHDP-glucose(pedunculagin) isomer II
16
4.85
933.0642
0.9
C41H26O26
300.9988,
Ellagitannin
Vescalagin*
17
5.35
783.0709
3.6
C34H24O22
300.9988, 481.0636
Ellagitannin
Di-HHDP-glucose(pedunculagin) isomer III
18
6.24
933.0642
0.9
C41H26O26
300.9988, 631.0605
Ellagitannin
Castalagin*
19
8.01
783.0709
3.6
C34H24O22
300.9988, 481.0636
Ellagitannin
Di-HHDP-glucose(pedunculagin) isomer IV
20
10.96
933.0642
0.9
C41H26O26
300.9988,
Ellagitannin
Vescalagin/Castalagin isomer
21
10.96
935.0810
2.0
C41H28O26
300.9988, 633.0742
Ellagitannin
Casuarinin
22
15.57
300.9988
1.3
C14H6O8
300.9988
Ellagic acid
Ellagic acid*
III
23
8.36
577.1331
−2.6
C30H26O12
289.0710, 407.0787
Flavan-3-ol
Procyanidin B1*
24
8.61
577.1331
−2.6
C30H26O12
289.0710, 407.0787
Flavan-3-ol
Procyanidin B-type isomer
25
9.45
289.0710
−0.7
C15H14O6
Flavan-3-ol
(+)-Catechin*
26
12.46
289.0710
−0.7
C15H14O6
Flavan-3-ol
(−)-Epicatechin*
27
13.77
465.1031
−0.4
C21H22O12
285.0424
Flavanonol
(2R,3R)-(+)-Glucodistylin*
28
14.22
465.1031
−0.4
C21H22O12
285.0424
Flavanonol
Taxifolin-O-hexoside
29
14.43
465.1031
−0.4
C21H22O12
285.0424
Flavanonol
(2S,3S)-(+)-Glucodistylin*
30
14.83
479.0843
3.5
C21H20O13
316.0212
Flavonol
Myricetin-O-hexoside
31
15.01
479.0843
3.5
C21H20O13
316.0212
Flavonol
Myricetin-O-hexoside
32
15.46
465.1031
−0.4
C21H22O12
285.0424
Flavanonol
Taxifolin-3′-O-glucopyranoside
33
15.74
463.0884
1.5
C21H20O12
316.0212
Flavonol
Myricitrin (myricetin-3-rhamnoside)*
34
15.84
463.0884
1.5
C21H20O12
301.0344, 300.0285
Flavonol
Hyperoside*
35
16.00
463.0884
1.5
C21H20O12
301.0344, 300.0285
Flavonol
Isoquercitrin*
36
15.97
303.0512
2.3
C15H12O7
Flavanonol
Taxifolin*
37
16.58
433.0803
7.4
C20H18O11
301.0344, 300.0285
Flavonol
Quercetin-3-O-alpha-L-arabinopyranoside*
38
16.58
447.0947
4.5
C21H20O11
285.0329
Flavonol
Kaempferol-O-hexoside
39
16.85
433.0755
−3.7
C20H18O11
301.0344, 300.0285
Flavonol
Avicularin*
40
17.03
447.0947
4.5
C21H20O11
285.0329
Flavonol
Astragalin*
41
17.18
447.0947
4.5
C21H20O11
301.0344, 300.0285
Flavonol
Quercitrin*
42
17.28
477.1042
1.9
C22H22O12
314.0422
Flavonol
Isorhamnetin-3-O-glucoside*
43
22.54
301.0344
−1.3
C15H10O7
Flavonol
Quercetin*
44
23.15
593.1277
−3.0
C30H26O13
285.0329, 447.0947
Flavonol
Tiliroside*
45
25.95
285.0385
−4.9
C15H10O6
Flavonol
Kaempferol*
IV
46
15.29
741.2628
3.0
C34H46O18
417.1578, 579.2067, 787.2662
Lignans
Syringaresinol-di-O-glucoside*
47
17.16
579.2067
−1.9
C28H36O13
417.1578
Lignans
Syringaresinol-4-O-glucoside*
According to the UPLC-ESI-Q-TOF-MS results, quinic acid (1), gallic acid (2), (+)-catechin (25), ellagitannins (16–18), ellagic acid (22), and flavonoid glycosides appeared to be common compounds in Q. dentata, Q. serrata, and Q. aliena. Interestingly, taxifolin (36) and its glycosides (27–29) were only detected in Q. serrata (Fig. 2B). Compared to that in other Quercus spp., the peak of quercitrin (41) was significantly higher in the UPLC-MS base peak intensity (BPI) chromatogram of the extracts from Q. serrata leaves (Fig. 2B). As shown in Fig. 2C, the galloylquinic acid (3), ellagitannins of the di-hexahydroxydiphenoyl (HHDP)-glucose structure (12, 15, 17, 19) and galloyl-di-HHDP-glucose structure (21) was relatively high in Q. aliena when compared with the values for the other oak species.
3.2.1 Identification of phenolic acids
Quinic acid (1) and its derivatives (3–11) in the three oak species were identified and deduced in accordance with the core-compound diagnostic ion at m/z 191.0559 (quinic acid) and fragmentation pattern. Compound 3 was identified as galloylquinic acid due to the loss of galloyl moiety (170 Da–H2O). Compounds 4 and 6 were characterized as caffeoylquinic acids due to the loss of caffeoyl moiety (180 Da–H2O). Similarly, compounds 5, 7, and 8 were characterized as coumaroylquinic acids due to the loss of coumaroyl moiety (164 Da–H2O). Compounds 9–11 were identified as dicaffeoylquinic acids due to their common fragment ions at m/z 179.0353 [M-caffeoyl-quinic acid-H]–. Compared with reference standards, compounds 2 and 4–11 were identified as gallic acid, neochlorogenic acid, 3-O-coumaroylquinic acid, chlorogenic acid, 5-O-coumaroylquinic acid, 4-O-coumaroylquinic acid, 3,4-dicaffeoylquinic acid, 3,5-dicaffeoylquinic acid, and 4,5-dicaffeoylquinic acid, respectively.
3.2.2 Identification of ellagitannin and ellagic acid
The ellagitannins (12–21) in the three oak species were identified and deduced in accordance with the core-compound diagnostic ion at m/z 300.9988 (ellagic acid). Compounds 12, 15, 17, and 19 were characterized as di-HHDP-glucose structure type ellagitannins due to their common fragment ion at m/z 481.0636 [M-HHDP-H]–. Compound 21 was identified as galloyl-di-HHDP-glucose structure type ellagitannin, casuarinin, due to their common fragment ion at m/z 633.0742 [M-HHDP-H]– and 783.0709 [M-galloyl-H]–. Compounds 13 was characterized as vescavaloninic acid due to its doubly charged fragment ions (m/z 550.0341 [M-2H]2– and 528.0372 [M-H-COOH]2–) and its daughter ions (m/z 631.0605 [M-valoneoyl-H, vescalin]–, 1039.0681 [M-H2O-COOH]– and 1083.0632 [M-H-H2O]–). Similarly, compounds 14 was characterized as castavaloninic acid due to its doubly charged fragment ions (m/z 550.0341 [M-2H]2– and 528.0372 [M-H-COOH]2–) and its daughter ions (m/z 631.0605 [M-valoneoyl-H, castalin]– and 1057.0863 [M-COOH]–). Compounds 16, 18, and 20 were characterized as HHDP-NHTP type ellagitannins due to their common fragment ion at m/z 631.0605 [M-HHDP-H]– and 613.0481 [M-HHDP-H2O-H]–. Compared with reference standard, compound 16 and 18 were identified as vescalagin and castalagin, respectively.
3.2.3 Identification of flavonoids
The flavonoids (23–45) in the three oak species were identified and deduced in accordance with the main aglycone diagnostic ions (catechin, taxifolin, myricetin, quercetin, kaempferol, isorhamnetin) as well as the fragmentation pattern. Compounds 23 and 24 were characterized as B-type procyanidins due to their diagnostic ion at m/z 289.0710 and fragmentation ion at m/z 407.0787, which resulted from retro-Diels-Alder (rDA) reaction and the loss of water. Compared with reference standard, compound 23 was identified as procyanidin B1. Taxifolin derivatives (27–29 and 32) were the major flavanonols in Q. serrata under investigation, according to their common fragment ion at m/z 285.0424. Compared with reference standard, compounds 27, 29, 32, and 36 were identified as (2R,3R)-(+)-glucodistylin, (2S,3S)-(+)-glucodistylin, taxifolin-3′-O-glucopyranoside, and taxifolin, respectively. Myricetin derivatives (33 and 33) were the major flavanonols in Q. dentata under investigation, according to their common fragment ion at m/z 316.0212 (deprotonated myricetin). Compared with reference standard, compound 33 was identified myricitrin (myricetin-3-O-α-L-rhamnopyranoside). Quercetin derivatives (34, 35, 37, 39, and 41) were the common flavonols in Quercus species under investigation, according to their common fragment ions at m/z 301.0344 (quercetin) and 300.0285 (deprotonated quercetin). Compared with reference standard, compounds 34, 35, 37, 39, 41 and 43 were identified hyperoside, isoquercitrin, quercetin-3-O-α-L-arabinopyranoside, avicularin (quercetin-3-O-α-L-arabinofuranoside), quercitrin, and quercetin, respectively. Kaempferol derivatives (38 and 40) were the common flavonols in Quercus species under investigation, according to their common fragment ion at m/z 285.0329 (kaempferol). Compound 44 was identified due to the loss of coumaroyl moiety (164 Da–H2O) and glucose (180 Da–H2O). Compared with reference standards, compounds 38, 40, 44, and 45 were identified as kaempferol-O-hexoside, astragalin (kaempferol-3-O-glucopyranoside), tiliroside (6′′-O-trans-p-coumaroylastragalin), and kaempferol, respectively. Compound 42 was identified as isorhamnetin-3-O-glucopyranoside due to its daughter ion at m/z 314.0422 (deprotonated isorhamnetin) and comparison with reference standard.
3.2.4 Identification of lignans
Syringaresinol derivatives (46 and 47) were detected in Q. aliena under investigation, according to their singly charged pseudo molecular ions (m/z 741.2628 for 46 and 579.2067 for 47) and aglycone diagnostic ion (m/z 417.1578, syringaresinol). Compared with reference standards, compounds 46 and 47 were identified as syringaresinol-di-O-glucoside and syringaresinol-4-O-glucoside, respectively.
3.3 Inhibitory effects of the oak-derived components on protein glycation
Protein (BSA, collagen, and elastin) glycation inhibitory activities of the major components derived from the different organs of the three oak species studied herein were tested, and the results are shown in Fig. 4A. Castalagin (18) and ellagic acid (22) showed outstanding inhibitory activity against both glucose/fructose-induced BSA glycation and fructose-induced BSA glycation, followed by quercetin glycosides (34, 35, and 41) > taxifolin glycosides (27 and 29), taxifolin (36), procyanidin B1 (23) > caffeoylquinic acids (4 and 6), myricitrin (33), and isorhamnetin-3-O-glucoside (42) > (+)-catechin (25) > gallic acid (2). In addition to BSA glycation, skin protein glycation inhibition assays were performed using glyceraldehyde as a glycation inducer. In the collagen glycation assay, castalagin (18), procyanidin B1 (23), quercetin derivatives (34, 35, and 41), myricitrin (33), astragalin (40), and isorhamnetin-3-O-glucoside (42) showed almost 50 % inhibition of the collagen glycation at 0.2 mM. However, taxifolin (36) and its glycosides (27 and 29) showed weak inhibitory potentials, whereas the caffeoylquinic acids (4 and 6) showed no inhibitory activity. With the elastin glycation assay, ellagic acid (22) showed the strongest anti-glycation activity (78.26 % inhibition at 0.2 mM), followed by quercetin derivatives (34, 35, and 41), isorhamnetin-3-O-glucoside (42), and myricitrin (33). However, taxifolin (36) and its glycosides (27 and 29) showed weak inhibitory potential against skin protein glycation, whereas caffeoylquinic acids (4 and 6) showed no inhibitory activity at 0.2 mM. The anti-glycation efficacy of the three oak species is thus believed to be due to mainly the presence of ellagitannins and flavonoid glycosides.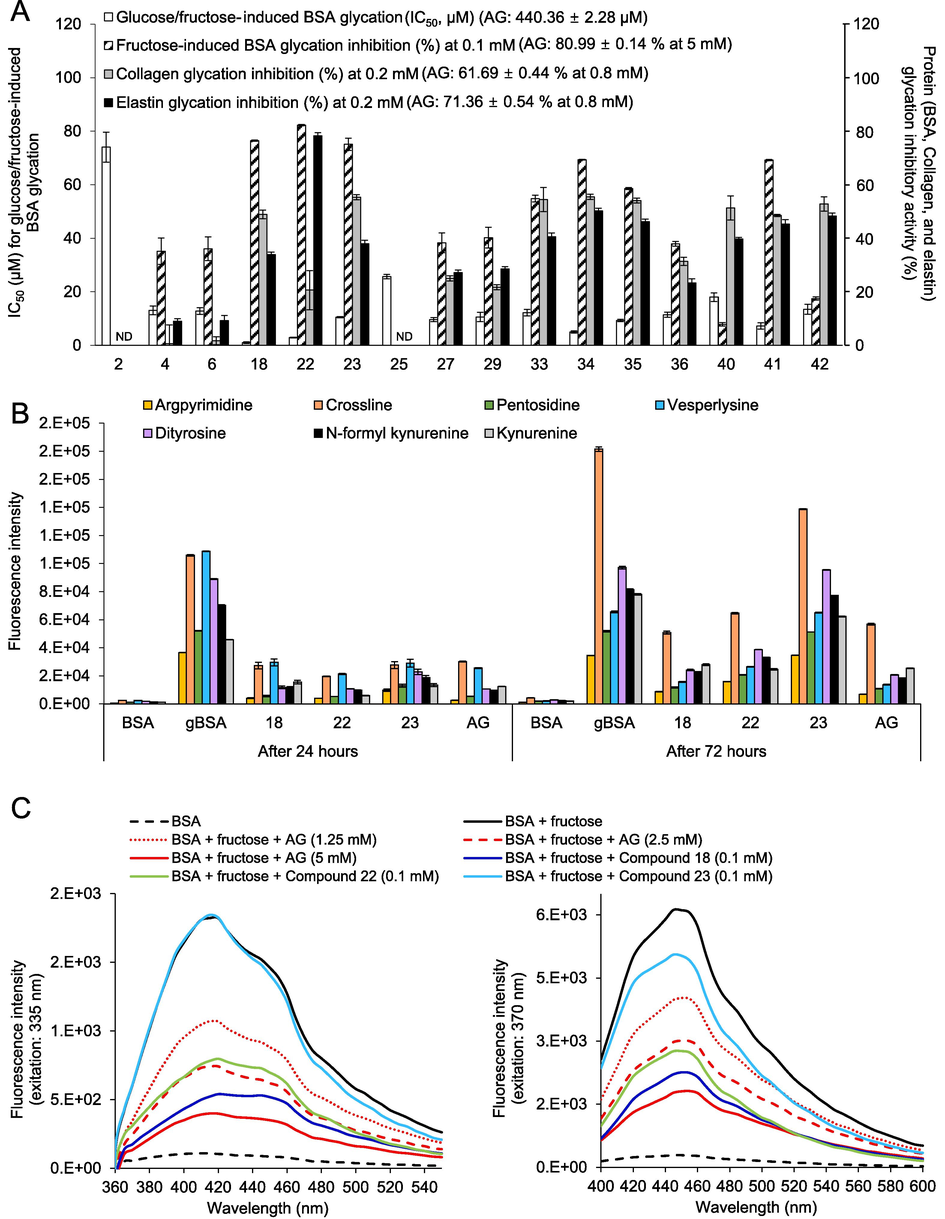
Effects of oak-derived components and aminoguanidine (AG) on the formation of total fluorescent AGEs in the BSA-glucose/fructose model, the BSA + fructose (gBSAfru) model, the collagen + glyceraldehyde model, and the elastin + glyceraldehyde model (A). Effects of the active compounds and AG on the formation of protein glycoxidation products and four specific fluorescent AGEs in the gBSAfru model (B). Fluorescence emission spectra of BSA, gBSAfru, and gBSAfru + active components excited at 335 and 370 nm (C).
3.4 Effects of the oak-derived components on fluorescent AGEs
The inhibitory effects of the oak-derived components on fluorescent AGEs in the gBSAfru model were evaluated by measuring the fluorescence intensity of cross-linked AGEs (CL, PEN, and VL) and non-cross-linked AGEs (APN) (Perrone et al., 2020). As shown in Fig. 4B, both cross-linked AGEs and non-cross-linked AGEs increased significantly after BSA glycation. However, a notable decrease in the fluorescence intensity of CL, PEN, VL, and APN was observed upon treatment with AG and oak-derived active compounds 18, 22, and 23 for 24 h. Especially, AG and compounds 18 and 22 dramatically reduced the levels of these specific fluorescent AGEs even after 72 h of glycation. The results were further validated through emission fluorescence spectra at excitation wavelengths of 335 (for PEN and APN) and 370 nm (for CL and VL), as illustrated in Fig. 4C.
3.5 Effect of the oak-derived components on protein glycoxidation
The inhibitory effects of the oak-derived components on protein glycoxidation in the gBSAfru model were also evaluated by measuring the fluorescence intensity of representative glycoxidation markers, including di-Tyr, NFK, and kynurenine. As shown in Fig. 4B, the levels of these three protein glycoxidation markers increased significantly after BSA glycation. However, a notable decrease in the fluorescence intensity of di-Tyr, NFK, and kynurenine was observed upon treatment with AG and oak-derived active compounds 18, 22, and 23 for 24 h. Especially, AG and compounds 18 and 22 significantly reduced the levels of these glycoxidation markers even after 72 h of glycation. The results were further validated through emission fluorescence spectra at excitation wavelengths of 335 (for di-Tyr and NFK) and 370 nm (for kynurenine), as illustrated in Fig. 4C.
3.6 Inhibition of the oak-derived components on aggregation of β-amyloid fibrillation
Formation of β-amyloid fibrillation in the gBSAfru model was assessed using a ThT-labeled fluorescence assay (Bouma et al., 2003). Fig. 5A and B illustrate the fluorescence spectra and the inhibition rates of amyloid fibrillation in the gBSAfru model treated with oak-derived components, respectively. The fluorescence intensity in the gBSAfru model was signficantly higher than that of native BSA, indicating substantial aggregation of the produced AGEs with BSA and subsequent formation of cross-linked structures. In contrast, treatment with AG and oak-derived active compounds 18 and 22 significantly decreased BSA aggregation. As shown in Fig. 5B, compounds 18 and 22 at a concentration of 0.1 mM prevented 44.85 % and 56.27 % of amyloid-like fibril formation, respectively, surpassing the inhibition observed with AG (35.08 %).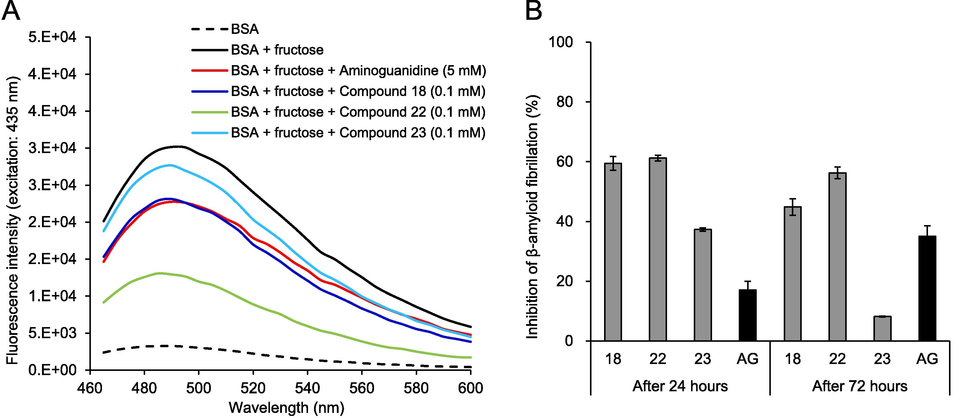
The fluorescence spectra (A) and inhibition rates (B) of oak-derived components on the formation of β-amyloid fibrillation in the BSA-fructose model.
3.7 MGO trapping capacities of the oak-derived compounds
The reaction mixture of MGO with anti-glycation active compounds from the three oak species at a 3:1 M ratio was analyzed using HPLC-ESI-Q-TOF-MS (Fig. 6 and Table 2). For the MGO-compound reaction mixture, several new peaks appeared in the HPLC chromatogram (Fig. 6). Based on the mass fragmentation patterns of the peaks generated after the reaction (Table 2), all the tested flavonoids (23, 27, 29, 33–35, 40–42) were shown to form MGO adducts, including mono-MGO adducts (M1) with an increase of 72 mass units (C3H4O2) and di-MGO adducts (M2) with an increase of 144 mass units (C3H4O2 × 2). Interestingly, tri-MGO adducts (M3) with an increase of 216 mass units (C3H4O2 × 3) were detected in the MGO-procyanidin B1 (23) reaction mixture, together with mono- and di-MGO adducts. On the basis of the present results and previous studies (Li et al., 2014), unsubstituted carbons at flavonoid (C6- and C8-position of A-ring) or procyanidin (C6- and C8-position of A-ring, C6′-position of D-ring, and C4′-position of F-ring) scaffold were highly possible MGO trapping sites (Fig. 7). NA No adducts were detected.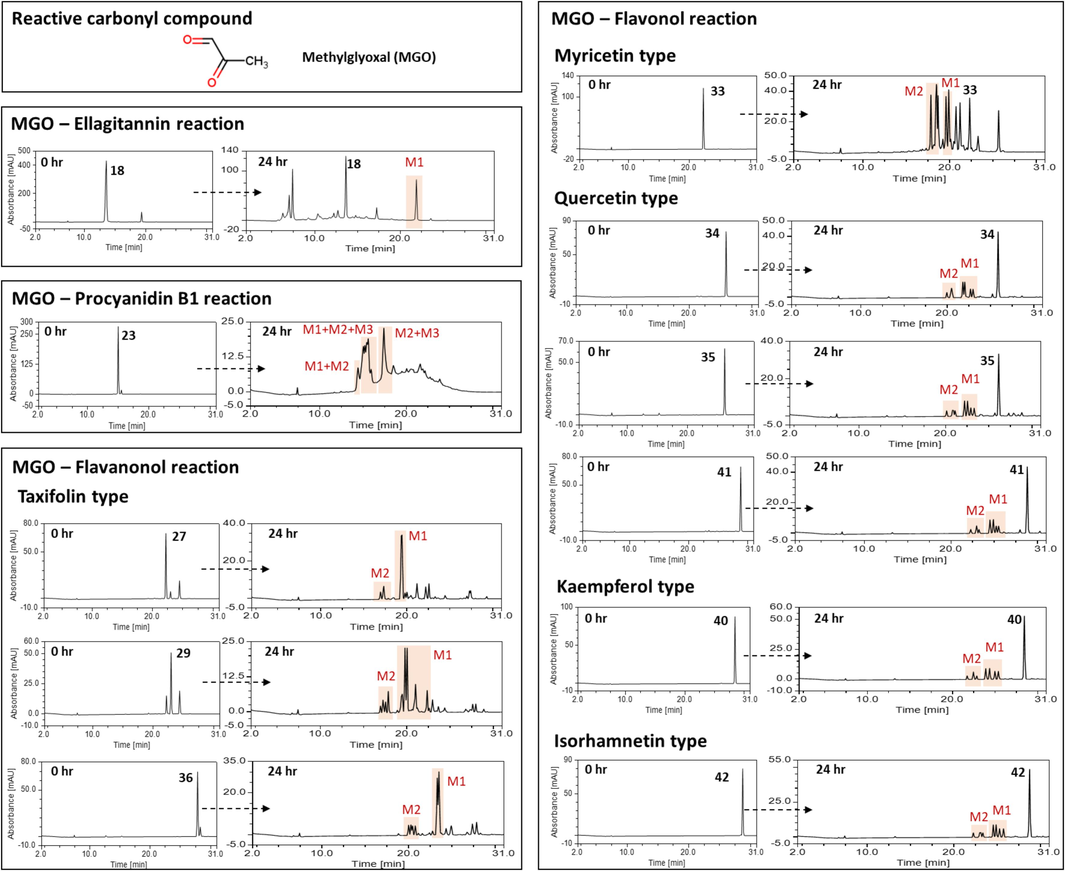
The HPLC chromatogram of compounds after incubation with methylglyoxal (MGO) for 24 h (M1, MGO-compound adduct; M2, di-MGO-compound adduct; M3, tri-MGO-compound adduct).
Compounds
Detected MGO–compound adducts [M − H]-
Neochlorogenic acid (4)
NA
Chlorogenic acid (6)
NA
Castalagin (18)
371.0044 (M1)
Ellagic acid (22)
NA
Procyanidin B1 (23)
793.1993 (M3), 721.1783 (M2), 649.1534 (M1)
(2R,3R)-(+)-Glucodistylin (27)
609.1467 (M2), 537.1241 (M1)
(2S,3S)-(−)-Glucodistylin (29)
609.1467 (M2), 537.1241 (M1)
Myricitrin (33)
607.1320 (M2), 535.1113 (M1)
Hyperoside (34)
607.1320 (M2), 535.1113 (M1)
Isoquercitrin (35)
607.1320 (M2), 535.1113 (M1)
Taxifolin (36)
447.0956 (M2), 375.0742 (M1)
Astragalin (40)
591.1354 (M2), 519.1144 (M1)
Quercitrin (41)
591.1354 (M2), 519.1144 (M1)
Isorhamnetin-3-O-glucoside (42)
621.1455 (M2), 549.1252 (M1)
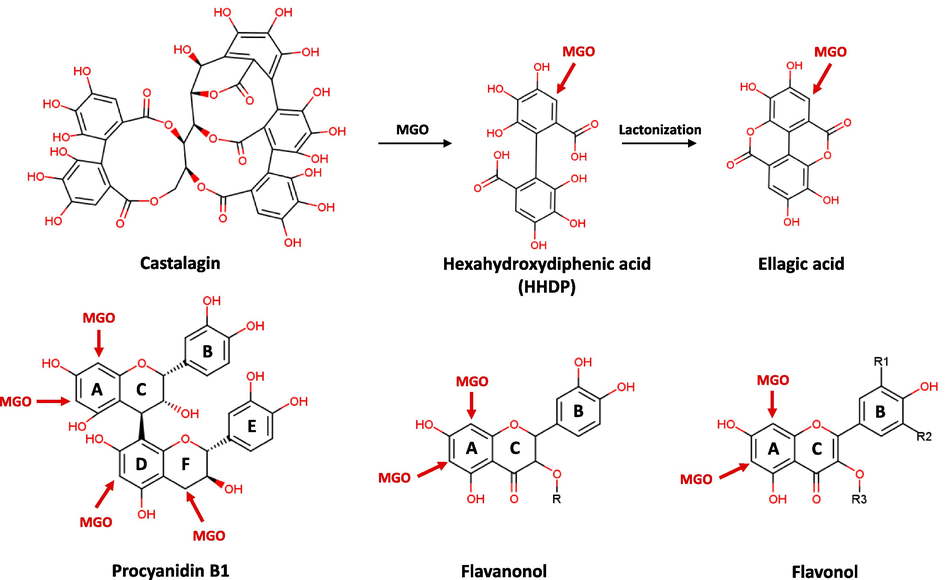
Possible mechanism for production of MGO-compound adducts.
Unlike flavonoids and procyanidin, castalagin (18), an ellagitannin, could trap MGO by binding mono-MGO to the degradation product rather than itself. For the MGO–compound 18 reaction mixture, new peaks appeared in the HPLC chromatogram after one day of incubation with MGO. Among them, a peak at the retention time (RT) of 7.02 min was observed to have the molecular formula of C27H19O18 (m/z of 631.0596 [M–H]−) with a fragment ion of m/z 300.9974, suggesting that this product is castalin. The peak at a RT of 21.87 min was observed to have a molecular formula of C17H7O10 (m/z of 371.0044 [M–H]−) with a fragment ion of m/z 300.9953 [ellagic acid–H]-, suggesting that this product is an ellagic acid-MGO (Fig. 6 and Table 2). Based on the fragmentation patterns of the peaks formed after the reaction, it was predicted that the HHDP moiety of castalagin (18) reacts with MGO and dissociates from the parent body to form M1 (Fig. 7). In contrast, caffeoylquinic acid (4 and 6) and ellagic acid (22) did not form adducts with MGO. Taken together, the HPLC-Q-TOF-MS analysis confirmed that flavonoids and ellagitannins derived from oaks formed mono-, di-, or tri-MGO adducts, thereby eliminating MGO, which is a reactive carbonyl intermediate and an important precursor of AGEs.
4 Discussion
Herein, we investigated the antioxidant and anti-glycation effects of 70 % ethanolic and aqueous extracts of the three organs (leaves, twigs, and fruits) of the Q. dentata, Q. serrata, and Q. aliena. As a result, all tested extract except for 70 % ethanolic extract from the Q. aliena fruits showed strong radical (ABTS·+ and DPPH) scavenging activity as well as good inhibition against monosaccharide-induced AGE formation. In particular, the 70 % EtOH extracts of the leaves and twigs of Q. aliena demonstrated anti-glycation effects that were 1.5 to 2 times stronger than those of onion (Allium cepa L.) extract, which has been validated as an effective anti-glycation agent in several studies. Additionally, these extracts exhibited comparable antioxidant activity (Kim and Kim, 2003; Yang et al., 2018). Therefore, the overall phytochemical profile of the leaves, stems, and fruits from these active oak species was analyzed using UPLC-Q-TOF-MS. For MS analysis, MSe acquisition mode was employed to obtain a full catalog of information for both precursor (low energy function) and fragment ions (high energy function) in a single analysis. The first, collected at low energy, yields accurate mass precursor ion spectra. The second, at high energy, yields the exact mass of the fragment ions. Based on the UPLC-ESI-Q-TOF-MS analysis, eleven phenolic acids (gallic acid and quinic acid derivatives), ellagic acid, ten ellagitannins, twenty-three flavonoid derivatives, and two lignans were identified. Interestingly, taxifolin and its glycosides were only detected in Q. serrata. In addition, the galloylquinic acid (3) and ellagitannins of the di-HHDP-glucose structure and galloyl-di-HHDP-glucose structure (casuarinin, 21) were relatively high in Q. aliena when compared with the values for the other oak species.
Several studies have demonstrated that the phytochemicals found in oak species have antioxidant effects (Othón-Díaz et al., 2023). However, there has been little investigation into how they function in the glycation of diverse proteins and how they interact with a reactive carbonyl intermediate. Therefore, the active ingredients were systematically explored by confirming the ability of the main oak components to inhibit glycation of various proteins (BSA, collagen, and elastin).
Serum albumin glycation was induced by monosaccharides (glucose and fructose). Ellagic acid (22) and castalagin (18) showed notable effects, and flavonoid glycosides (quercetin > taxifolin > myricetin > isorhamnetin > kaempferol) and procyanidin B1 (23) showed strong inhibitory effects. Based on these results, it was confirmed that components with an ellagic acid-type structure can effectively inhibit BSA glycation and that the effect varies depending on the number of hydroxyl groups in the flavonoid.
Ellagic acid and ellagitannin are bioactive polyphenols commonly found in oak trees and are abundant in some fruits (almonds, pomegranates, and raspberries) (Landete, 2011). Ellagitannins are complex ellagic acid derivatives that are hydrolyzed by acids or bases to produce HHDP, which is then spontaneously lactonized to ellagic acid (Evtyugin et al., 2020). Ellagic acids and ellagitannins, similarly to other polyphenols, exhibit a variety of biological actions such as antioxidant, anti-estrogenic, and estrogenic effects, as well as prebiotic and anti-inflammatory activities, suggesting that they may be beneficial to human health (Larrosa et al., 2010; Sharifi-Rad et al., 2022). In our previous study, tellimagrandin II, the first ellagitannin formed from 1,2,3,4,6-penta-O-galloyl-glucose (PGG), exhibited strong inhibition of protein glycation by trapping RCCs and excellent binding ability to AGE receptors (RAGE) (Seong et al., 2023). Similarly, Ma et al. (2015) demonstrated that PGG inhibited early glycation by preventing sugar induced conformational changes of the protein (α-helix to β-sheet) via molecular docking simulations and matrix assisted laser desorption/ionization time of flight-TOF analysis (Ma et al., 2015). In line with these findings, the present study is the first to demonstrate the inhibitory effects of oak-derived ellagitannin, castalagin (18), against protein glycation.
During aging and for patients affected by diabetes, AGE accumulation has been detected in various tissues, including the glomerular basement membrane, skeletal/smooth vascular muscle, elastin, and articular collagen (Gkogkolou and Böhm, 2012). Proteins with long biological half-lives, such as elastin and collagen, which have slow turnover rates of more than 10 years, are more prone to glycation and AGE generation than other proteins (Muir et al., 2021). Collagen and elastin are the major fibers that constitute the extracellular matrix (ECM). Elastin provides skin elasticity and collagen provides tensile strength. In particular, the skin collagen content was greater than 70 %, whereas that of elastin was approximately 2 % (Uitto, 1986). Collagen is not only employed as a mechanical support system for cells and tissues in the skin, but is also an active component capable of interacting with cells and influencing numerous cellular functions, such as proliferation, migration, and differentiation. During glycation, reducing sugar binds to the free lysine or arginine residues of elastin and collagen to form a Schiff base. The accumulation of cross-linked AGEs in skin proteins has various negative effects, including reduced solubility and flexibility of the collagen triple helix; decreased arterial/myocardial compliance; and increased vascular stiffness, diastolic function, and systolic hypertension (Gkogkolou and Böhm, 2012). In addition, skin elasticity declines with age, and this trend was more pronounced in patients with diabetes who experienced increased glycative stress (Yonei et al., 2010). Therefore, the anti-glycation potential of the active oak ingredients against two skin proteins (elastin and collagen) was evaluated. The glycation of these two skin proteins was induced by glycoaldehyde, which is an α-hydroxyaldehyde, and the anti-glycation potential was measured for active compounds at the same dose (0.2 mM). As a result, ellagic acid significantly inhibited elastin glycation, while having little to no impact on collagen glycation. In contrast, ellagitannins had a minimal impact on the glycation of elastin and a nearly 50 % inhibitory effect on the glycation of collagen. Furthermore, flavonoid glycosides, excluding taxifolin and its glycosides, inhibit collagen and elastin glycation by approximately 50 %. Recent studies have revealed that rutin, a representative quercetin rutinoside, is abundant in various foods, and its metabolites can effectively prevent AGE formation in glucose-induced collagen glycation (Cervantes-Laurean et al., 2006). In addition, Chang et al. reported that cranberry juice polyphenols, including procyanidins and quercetin, can effectively prevent MGO-induced collagen glycation and cross-linking (Chang et al., 2022). Urios and colleagues demonstrated that 3′-OH, 4-oxo, and delta2-3 in the flavonoid scaffold are important for inhibitory activity against collagen glycation by measuring the formation of PEN, which is a specific marker of protein cross-link as well as glycoxidation, using HPLC (Urios et al., 2007). Taken together, it was proven that the flavonols are more effective than flavanonols at inhibiting the glycation of skin proteins and that the inhibitory effects increase alongside the number of hydroxyl groups linked to the B-ring of the flavonoids.
To elucidate the mechanism of action of the oak-derived active ingredients in inhibiting protein glycation, their effects on the middle- and end-stages of the protein glycation process were evaluated. MGO is the representative RCC generated during the middle-stage of the protein glycation. MGO influences the formation of AGEs and cross-linked aggregates in the later stages of protein glycation by covalently binding to functional groups of proteins and releasing free radicals (Chaudhuri et al., 2016). The MGO-trapping capacities of oak-derived active ingredients were assessed using HPLC-Q-TOF-MS. Several new peaks were observed in the HPLC chromatogram, which indicated that the active ingredients formed adducts with mono-, di-, and tri-MGO. The binding aspect of oak-derived active ingredients to MGO was confirmed by Q-TOF-MS analysis of the precursor ion spectrum and MS fragmentation pattern of the newly created MGO adduct using the low-energy and high-energy functions of the MSe acquisition mode, respectively. In the MGO-catalagin (18) reaction mixture, mono-MGO was trapped by HHDP, which formed when the parent molecule (18) was broken, thus creating an adduct. For flavonoids (23, 27, 29, 33–36, 40–42), MGO molecules combined with the unsubstituted carbons (C6- and C8-positions) at the A-ring of the flavonoid to form mono- and di-MGO adducts. Interestingly, tri-MGO adducts were detected in the MGO-procyanidin B1 (23) reaction mixture, together with mono- and di-MGO adducts. However, because the ellagic acid (22) and caffeoylquinic acids (4 and 6) did not form adducts with MGO, further research on their other anti-glycation mechanisms should be conducted.
The generated advanced glycation end-products (AGEs) can lead to intramolecular protein cross-linking, further promoting β-amyloid fibrillation associated with β-sheet formation during the later stages of protein glycation. These protein aggregations may impair both mechanical properties and biological functions (Zhang et al., 2022). Furthermore, the non-enzymatic glycation is accompanied by oxidation. During glycoxidation, tyrosine (Tyr) and tryptophan (Trp) residues are readily modified, resulting in the formation of fluorescent products. Particularly, Tyr residues are converted to kynurenine, while Trp residues are transformed into di-Tyr and NFK. Glycoxidation can also induce conformational changes in proteins, leading to the formation of amyloid fibrils (Eze and Tola, 2020). In the current study, castalagin (18) and ellagic acid (22) were found to inhibit the formation of cross-linked AGEs (CL, PEN, and VL), non-cross-linked AGEs (APN), and glycoxidation products (di-Tyr, NFK, and kynurenine) by measuring the fluorescence intensity and spectra at specific excitation and emission wavelengths of fluorescent AGEs and glycoxidation products generated from BSA glycation. Additionally, the glycated BSA solution, when incubated with ThT, exhibited increased fluorescence, indicating the presence of a β-sheeted conformation compared to the native BSA solution. However, castalagin (18) and ellagic acid (22) effectively reduced the ThT fluorescence intensity resulting from BSA glycation, demonstrating their strong potential to inhibit protein aggregation. Therefore, the potent inhibitory effects of the three oak species (Q. dentata, Q. serrata, and Q. aliena) on protein glycation appear to be due to the presence of ellagitannins and flavonoids, which possess inhibitory potential against protein glycation, glycoxidation, and aggregation, as well as RCC-trapping abilities.
5 Conclusions
The components of three oak species (Q. dentata, Q. serrata, and Q. aliena) were profiled, and their anti-glycation effects were systematically examined. This study is the first to demonstrate that castalagin (18), a major ellagitannin in oak, acts as an inhibitor of non-enzymatic protein (BSA, collagen, and elastin) glycation by trapping RCCs and inhibiting AGE production. In addition, the anti-glycation efficiency and RCC-trapping ability of the oak-derived flavonoids were examined, and a structure–activity correlation was established. Taken together, the results of our study might help deepen our understanding of the different oak components and aid in the development of functional foods and pharmaceuticals to manage various disorders caused by aging and excessive sugar consumption. Thus, the active ingredients in oak leaves, twigs, and fruits (mainly ellagitannins and flavonoid derivatives) are promising natural anti-glycation compounds with preventive and therapeutic potential against AGE-related disorders in multiple tissues.
CRediT authorship contribution statement
Su Hui Seong: Writing – review & editing, Writing – original draft, Methodology, Investigation, Formal analysis, Conceptualization. Bo-Ram Kim: Resources, Investigation. Seahee Han: Resources. Jin-Ho Kim: Formal analysis. Sua Im: Resources. Tae-Su Kim: Formal analysis. Chan Seo: Formal analysis. Ha-Nul Lee: Formal analysis. Jung Eun Kim: Resources. Ji Min Jung: Resources. Myoung Lae Cho: Resources, Conceptualization. Kyung-Min Choi: Supervision. Jin-Woo Jeong: Supervision, Project administration, Funding acquisition.
Funding
This work was supported by a grant from the Honam National Institute of Biological Resources (HNIBR), funded by the Ministry of Environment (MOE) of the Republic of Korea (HNIBR202302115), and the Korea Environment Industry & Technology Institute (KEITI) through a project aimed at advancing multi-ministerial national biological research resources, funded by the Korea Ministry of Environment (MOE) (RS-2023-00230402).
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Carbonyl scavenging and chemical chaperon like function of essential amino acids attenuates non-enzymatic glycation of albumin. RSC Adv.. 2016;6(29):24557-24564.
- [CrossRef] [Google Scholar]
- Glycation induces formation of amyloid cross-β structure in albumin. J. Biol. Chem.. 2003;278(43):41810-41819.
- [CrossRef] [Google Scholar]
- A comprehensive review of phytochemistry and biological activities of Quercus species. Forests. 2020;11(9):904.
- [CrossRef] [Google Scholar]
- Inhibition of advanced glycation end product formation on collagen by rutin and its metabolites. J. Nutr. Biochem.. 2006;17(8):531-540.
- [CrossRef] [Google Scholar]
- Cranberry juice polyphenols inhibited the formation of advanced glycation end products in collagens, inhibited advanced glycation end product-induced collagen crosslinking, and cleaved the formed crosslinks. J. Agric. Food Chem.. 2022;70(49):15560-15569.
- [CrossRef] [Google Scholar]
- The role of advanced glycation end products in aging and metabolic diseases: bridging association and causality. Cell Metab.. 2018;28(3):337-352.
- [CrossRef] [Google Scholar]
- Glyceraldehyde-derived advanced glycation end products in Alzheimer’s disease. Acta Neuropathol.. 2004;108(3):189-193.
- [CrossRef] [Google Scholar]
- Comparative study of antioxidant properties and total phenolic content of 30 plant extracts of industrial interest using DPPH, ABTS, FRAP, SOD, and ORAC assays. J. Agric. Food Chem.. 2009;57(5):1768-1774.
- [CrossRef] [Google Scholar]
- Recent advances in the production and applications of ellagic acid and its derivatives. A Review. Molecules. 2020;25(12):2745.
- [CrossRef] [Google Scholar]
- Eze, F. N., Tola, A. J., 2020. Protein glycation and oxidation inhibitory activity of Centella asiatica phenolics (CAP) in glucose-mediated bovine serum albumin glycoxidation. Food Chem., 332, 127302. 10.1016/j.foodchem.2020.127302.
- Advanced glycation end products. Dermatoendocrinology. 2012;4(3):259-270.
- [CrossRef] [Google Scholar]
- Advanced glycation end-products (AGEs): involvement in aging and in neurodegenerative diseases. Amino Acids. 2008;35(1):29-36.
- [CrossRef] [Google Scholar]
- Mechanism of natural antioxidants regulating advanced glycosylation end products of Maillard reaction. Food Chem.. 2023;404:134541
- [CrossRef] [Google Scholar]
- Protein glycation inhibitory and antioxidative activities of some plant extracts in vitro. J. Agric. Food Chem.. 2003;51(6):1586-1591.
- [CrossRef] [Google Scholar]
- Ellagitannins, ellagic acid and their derived metabolites: A review about source, metabolism, functions and health. Food Res. Int.. 2011;44(5):1150-1160.
- [CrossRef] [Google Scholar]
- Ellagitannins, ellagic acid and vascular health. Mol. Aspects Med.. 2010;31(6):513-539.
- [CrossRef] [Google Scholar]
- Quercetin inhibits advanced glycation end product formation by trapping methylglyoxal and glyoxal. J. Agric. Food Chem.. 2014;62(50):12152-12158.
- [CrossRef] [Google Scholar]
- Formation mechanism of AGEs in Maillard reaction model systems containing ascorbic acid. Food Chem.. 2022;378:132108
- [CrossRef] [Google Scholar]
- The hydrolyzable gallotannin, penta-O-galloyl-β-D-glucopyranoside, inhibits the formation of advanced glycation endproducts by protecting protein structure. Mol. Biosyst.. 2015;11(5):1338-1347.
- [CrossRef] [Google Scholar]
- In vitro and in vivo inhibition of maillard reaction products using amino acids, modified proteins, vitamins, and genistein: A review. J. Food Biochem.. 2019;43(12):e13089
- [CrossRef] [Google Scholar]
- Oak trees (Quercus spp.) as a source of extracts with biological activities: A narrative review. Trends Food Sci. Technol.. 2021;109:116-125.
- [CrossRef] [Google Scholar]
- Collagen glycation detected by its intrinsic fluorescence. J. Phys. Chem. B. 2021;125(39):11058-11066.
- [CrossRef] [Google Scholar]
- Vitexin inhibits protein glycation through structural protection, methylglyoxal trapping, and alteration of glycation site. J. Agric. Food Chem.. 2021;69(8):2462-2476.
- [CrossRef] [Google Scholar]
- Antioxidants in Oak (Quercus sp.): Potential application to reduce oxidative rancidity in foods. Antioxidants. 2023;12(4):861
- [CrossRef] [Google Scholar]
- Advanced glycation end products (AGEs): biochemistry, signaling, analytical methods, and epigenetic effects. Oxid. Med. Cell. Longev.. 2020;2020:3818196
- [CrossRef] [Google Scholar]
- Discovery of flazin, an alkaloid isolated from cherry tomato juice, as a novel non-enzymatic protein glycation inhibitor via in vitro and in silico studies. J. Agric. Food Chem.. 2021;69(12):3647-3657.
- [CrossRef] [Google Scholar]
- Phytochemical profiling of Symplocos tanakana Nakai and S. sawafutagi Nagam. leaf and identification of their antioxidant and anti-diabetic potential. J. Pharm. Biomed. Anal.. 2023;233:115441
- [CrossRef] [Google Scholar]
- Ellagic acid: A review on its natural sources, chemical stability, and therapeutic potential. Oxid. Med. Cell. Longev.. 2022;2022:3848084
- [CrossRef] [Google Scholar]
- Medicinal uses, phytochemistry, and pharmacological activities of Quercus species. Evid. Based Complement. Alternat. Med.. 2020;2020:1920683
- [CrossRef] [Google Scholar]
- Neurotoxicity of advanced glycation end-products for cultured cortical neurons. J. Neuropathol. Exp. Neurol.. 2000;59(12):1094-1105.
- [CrossRef] [Google Scholar]
- Use of aminoguanidine (Pimagedine) to prevent the formation of advanced glycation endproducts. Arch. Biochem. Biophys.. 2003;419(1):31-40.
- [CrossRef] [Google Scholar]
- Connective tissue biochemistry of the aging dermis: Age-related alterations in collagen and elastin. Dermatol. Clin.. 1986;4(3):433-446.
- [CrossRef] [Google Scholar]
- Flavonoids inhibit the formation of the cross-linking AGE pentosidine in collagen incubated with glucose, according to their structure. Eur. J. Nutr.. 2007;46(3):139-146.
- [CrossRef] [Google Scholar]
- Inhibition of protein glycation and advanced glycation end products by ascorbic acid and other vitamins and nutrients. J. Nutr. Biochem.. 1996;7(12):659-663.
- [CrossRef] [Google Scholar]
- Antioxidative and anti-glycation activity of garcinol from Garcinia indica fruit rind. J. Agric. Food Chem.. 2000;48(2):180-185.
- [CrossRef] [Google Scholar]
- In vitro protein tyrosine phosphatase 1B inhibition and antioxidant property of different onion peel cultivars: A comparative study. Food Sci. Nutr.. 2018;7(1):205-215.
- [CrossRef] [Google Scholar]
- Identification and inhibitory activities of ellagic acid- and kaempferol-derivatives from Mongolian oak cups against α-glucosidase, α-amylase and protein glycation linked to type II diabetes and its complications and their influence on HepG2 cells’ viability. Arab. J. Chem.. 2018;11(8):1247-1259.
- [CrossRef] [Google Scholar]
- Anti-glycation effect of mixed herbal extract in individuals with pre-diabetes mellitus. Anti Aging Med.. 2010;7(5):26-35.
- [CrossRef] [Google Scholar]
- Inhibition on α-glucosidase activity and non-enzymatic glycation by an anti-oxidative proteoglycan from Ganoderma lucidum. Molecules. 2022;27(5):1457.
- [CrossRef] [Google Scholar]







