Preparation of enteric capsules with pulsatile drug delivery potential using pullulan and polyacrylic acid resin III
⁎Corresponding authors. tangjb@wfmc.edu.cn (Jinbao Tang), chenlu@wfmc.edu.cn (Lu Chen)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
In this study, the enteric swelling layer of capsules was prepared by optimizing the ratio of pullulan to gellan gum. The enteric-coating solution was prepared by dissolving polyacrylic acid resin III in 85 % ethanol, and the two were combined to form pullulan enteric-coated capsules (PECs). To elaborate further, Pullulan enteric-coated thin films (PEFs) were fabricated using the casting method, and their oxygen permeability (OP) and water vapor permeability (WVP) were assessed. Enteric capsules were prepared employing a secondary impregnation method. The cap gap and surface roughness of the capsules were characterized using Scanning Electron Microscopy (SEM) and Atomic Force Microscopy (AFM). The water content and hygroscopicity of the capsules were examined by employing the drying weight loss method. The pullulan-based enteric capsules (PECs) exhibited attributes such as low water content, hygroscopicity, brittleness, leakage resistance, and high impermeability. Finally, the in vitro release of aspirin from the enteric capsules was investigated. Results demonstrated the excellent combination of the enteric-coating layer and the swelling layer in the PECs. Compared with Gelatin enteric-coated capsules (GECs) on the market, it had a smaller cap gap, lower brittleness, weaker moisture absorption, and a good dissolution effect. Thus, it met the requirements of Chinese pharmacopeia, i.e., the ability to maintain integrity for 2 h in simulated gastric fluid and a drug-release rate reaching 80 % within 30 min in simulated intestinal fluid. This capsule also exhibited certain characteristics of a pulsatile drug-delivery system. This work proposed a potential method of preparing enteric hard capsules by using pullulan combined with polyacrylic acid resin III.
Keywords
Pullulan hard capsule
Pulsatile drug-delivery system
Enteric coating
Aspirin
- GECs
-
Gelatin enteric-coated capsules
- PECs
-
pullulan enteric-coated capsules
- HCl
-
hydrogen chloride
- PBS
-
Phosphate buffered saline
- WVP
-
water-vapor permeability
- OP
-
oxygen permeability
- PEFs
-
Pullulan enteric-coated thin films
- PV
-
peroxide value
- SEM
-
Scanning electron microscopy
- AFM
-
Atomic force microscopy
- RH
-
Relative Humidity
- MSR
-
Moisture sorption rate
Abbreviations
1 Introduction
Hard capsules are some of the largest solid dosage forms prepared in the pharmaceutical industry (Gullapalli and Mazzitelli, 2017). The most common material used to prepare the capsule shells is gelatin (Fundueanu et al., 2008). Coating the surface of the capsule shell with a specific material composite film can prepare sustained-release formulations that deliver drugs to specific parts of the intestine, meeting the drug-loading requirements of different medications (Reix et al., 2012). However, gelatin capsules have a smooth surface that is not conducive to film adhesion, and gelatin is a water-soluble protein containing a large number of carboxyl groups, amino groups, and hydroxyl groups. These functional groups easily form hydrogen bonds with water molecules (Duconseille et al., 2015). When using the aqueous film-coating method, softening, swelling, and even sticking together may occur, and the capsules may become brittle due to water loss during the drying process (Yang et al., 2020, Zulkiflee and Fauzi, 2021, Ding et al., 2024). These issues can be resolved by using non-gelatin materials, such as chitosan and its derivatives, modified starch, and sodium alginate, as alternatives to prepare capsules instead of gelatin (Zhang et al., 2013).
Pullulan is a microbial extracellular polysaccharide produced by the yeast fungus Rhizopus during aerobic fermentation in the form of amorphous mucilage (Prajapati et al., 2013). It is a water-soluble, biocompatible, nontoxic, and film-forming material (Prajapati et al., 2013, Wei et al., 2021, Agrawal et al., 2022). Pullulan is generally recognized as safe by the US Food and Drug Administration (Rai et al., 2021). Plantcaps™ is a commercial hard capsule made primarily from pullulan, and it has the advantages of gelatin capsules (Garbacz et al., 2014). However, due to the physical properties of pullulan, it has strong water solubility when it exists alone. Under the acidic conditions of the stomach, the molecular structure of pullulan is gradually hydrolyzed by gastric proteases (Singh et al., 2021). Therefore, pullulan must be combined with other materials to achieve processability and expected functionality.
Adding polymer materials and plasticizers to form a composite film is commonly used to produce capsule shells (Chen et al., 2021). Most capsule shells are typically prepared by dip-coating technique. When preparing enteric-coated formulations that are resistant to gastric acid or need to be delivered through the stomach for backward dosing, drug delivery is achieved through enteric coating. Anionic polymers such as methacrylic acid ester polymers (copolymer of methacrylic acid and methyl methacrylate or ethyl acrylate (Eudragit®)), cellulose-based polymers such as cellulose acetate phthalate (Aquateric®), or polyethylene derivatives such as polyethylene-co-vinyl acetate (Coateric®) are commonly used to achieve enteric properties (Cole et al., 2002). The materials comprising methyl methacrylate acid and its ester, polymerized in different proportions to form cationic or anionic copolymers, can be categorized into different types based on their ratios and compositions. These products are collectively referred to as Eudragit, with basic models including Eudragit E, L, S, RL, RS, and NE, as well as Eastacryl and Kollicoat (Thakral et al., 2013).
Polyacrylic acid resin can be used as an enteric-coating material, but no specific data are available regarding the types of polyacrylic acid resin suitable for different drug formulations. Based on specific needs, different materials are selected or different types of materials are used in combination. Accordingly, the present work developed an enteric capsule based on pullulan to obtain a high-quality and stable product by developing and optimizing the formula of the capsule's enteric-coating and swelling layers. We aimed to determine whether the combination of pullulan and polyacrylic acid resin materials can form enteric capsules with pulsatile drug-delivery characteristics while also prolonging drug release. The effects of formulation and variables on capsule disintegration were also examined and compared with those of gelatin enteric capsules (GECs). The barrier performance, sealing performance, roughness, and moisture absorption of the capsules were evaluated. Aspirin was selected as the model drug to study the drug release of the capsules in the gastrointestinal tract. The aim was to establish a foundation for the application of pullulan enteric-coated capsules (PECs) as a new dosage form.
2 Materials and methods
2.1 Materials
Pullulan was purchased from Shandong FURIDA Biotechnology Co., Ltd., China. Glycerinum was purchased from Xinpote Chemical Co., Ltd. in Tianjin, China. Gellan gum was purchased from Hefei Qiansheng Biotechnology Co., Ltd., China. Tween-80 was purchased from China National Pharmaceutical Group Chemical Reagent Co., Ltd., China. Polyacrylic acid resin Ⅲ and Ⅱ were purchased from Nanjing DoLai Biotechnology Co., Ltd., China. Acetylsalicylic acid (Aspirin) was purchased from Aladdin Biochemical Technology Co., Ltd. in China. Simulated gastric juice and simulated intestinal juice were purchased from MesGen Biotech Co., Ltd., Shanghai, China. Gelatin enteric capsules were purchased from Shaoxing Kangke Capsule Co., Ltd., China.
2.2 Characterization of pullulan
Referring to the measurement method proposed by Liu (Liu et al., 2018) et al., the molecular weight of pullulan was analyzed by gel-permeation chromatography (GPC). Pullulan samples ranging from 100 mg to 300 mg were dissolved in 100 mL of distilled water to prepare different concentrations of pullulan solutions. After filtration through a 0.22 μm microporous membrane, the molecular weight of pullulan was determined. Analysis was performed using a gel-permeation chromatography (1260 InfinityⅡ MDSs, USA; column, PL aqua gel-OH Mixed-H; mobile phase, 0.1 mol/L sodium nitrate (0.01 % sodium azide); temperature, 45 °C; eluent flow rate, 1.0 mL/min; differential detector; dual-angle laser light scattering detector; sample volume, 50 μL).
2.3 Preparation of PECs
A metal capsule mold and the dipping-coating method (Chen et al., 2021) were used to form PECs. The entire process of preparing PECs included sol preparation, dipping, drying, secondary coating, drying and shaping, cutting and de-shelling, and assembly. Fig. 1 shows the specific preparation methods and requirements. All capsules were cut to the appropriate length for the “0#” capsule mold, and their thickness was measured using a digital caliper. To determine if the capsules were properly formed, the external defects of empty hard capsules were assessed accordingly (Table 1 lists the defect descriptions). Under the corresponding preparation conditions, 10 PECs were randomly selected. If any of the capsules met any of the criteria in Table 1, it was considered an unformed sample. The capsule-forming rate was calculated using formula (1):
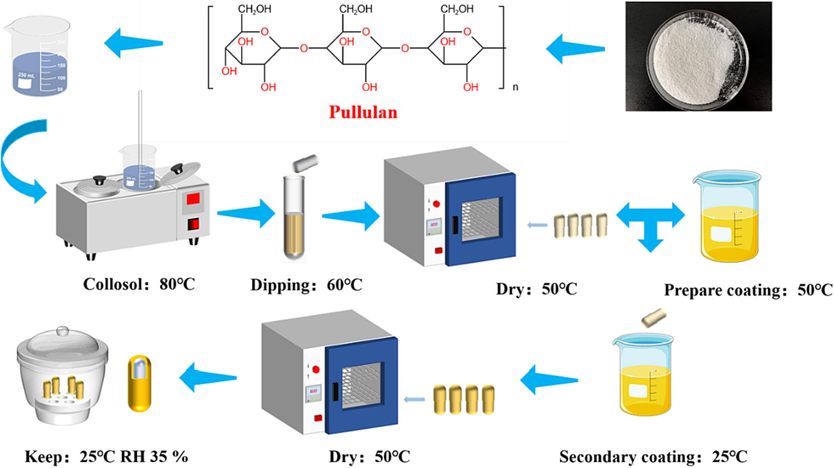
- Preparation of PECs.
| Defect name | Description |
|---|---|
| Broken hole | Any visible holes in the surface |
| No cut/Presence of debris | The cut edge of the cap remains attached to the capsule |
| Fissure/Gap | Cut edge cracks greater than 2 mm, gap greater than 2 mm |
| Short body | The length of the capsule is more than 1 mm shorter than the specified length. |
| Deflated shell | The body cap is flattened or twisted |
| intercut | When the capsule is closed, the weight of the cap is inserted into each other |
| Side scrap | Any excised part left after cutting the cap or body |
The capsule's swelling layer was prepared using different concentrations of pullulan (100–180 g/L) and gellan gum (5–20 g/L). The polymer was plasticized using different concentrations of potassium citrate (0.5–1.2 g/L) and glycerol (10–18 %), the concentration of glycerol is relative to the mass of the polysaccharide in the solution. Table 2 summarizes the test formulations. The optimal concentration of pullulan was determined in group (1), which was crucial to the gel-solution viscosity and the swelling-layer thickness. Group (2) determined the optimal concentration of the gelling agent gellan gum, which determined whether the gel solution properly adhered to the capsule mold. Group (3) determined the optimal glycerol content, thereby affecting the capsule's water content and brittleness. Lastly, Group (4) determined the optimal concentration of potassium citrate, which also greatly affected the capsule's formation.
| Number | Pullulan (g/L) | Gellan gum(g/L) | Glycerin(%) | potassium citrate(g/L) |
|---|---|---|---|---|
| Group (1) | 100 | 10 | 20 | 1 |
| 120 | ||||
| 150 | ||||
| 180 | ||||
| Group (2) | 120 | 5 | 20 | 1 |
| 10 | ||||
| 15 | ||||
| 20 | ||||
| Group (3) | 120 | 10 | 10 | 1 |
| 15 | ||||
| 20 | ||||
| 25 | ||||
| Group (4) | 120 | 10 | 20 | 0.5 |
| 0.8 | ||||
| 1 | ||||
| 1.2 | ||||
An enteric-coating solution was prepared using different concentrations of polyacrylic acid resin II (5 %–12.5 % w/v) or polyacrylic acid resin III (5 %–12.5 % w/v) dissolved in ethanol (65 %–95 % v/v). After adding the emulsifier Tween-80 (4.5 %–7.5 % v/v) to the solution, it was heated in a 50 °C water bath. Table 3 provides the formulation for the enteric-coating solution. In groups (1) and (2) of the testing, different types and optimal concentrations of enteric materials were selected. The entire testing process aimed to achieve dissolution through disintegration experiments. Disintegration experiments require selecting enteric capsules coated with different concentrations of polyacrylic acid resin materials, filling them with equal amounts of talc, and testing the leakage of talc from the capsules by using a disintegration-time instrument (LB-2D, China) in 0.1 M HCl (pH 1.5) and pH 6.8 PBS (Sutthapitaksakul et al., 2022). Group (3) tested the optimal concentration of ethanol, and the appropriate concentration of ethanol as a solvent was found to accelerate the dissolution of enteric materials. The appearance of enteric capsules prepared with different concentrations of ethanol was observed to determine the suitable concentration. Group (4) primarily used a water-content analyzer (Precisa XM60pro, Switzerland) to measure the water content of enteric capsules and screen for the optimal concentration of Tween-80. Adding the appropriate concentration of Tween-80 can reduce the fragility of the capsule.
| Number | Polyacrylic acid resin Ⅱ(%) | Polyacrylic acid resin Ⅲ(%) | Ethyl alcohol(%) | Tween-80 (%) |
|---|---|---|---|---|
| Group (1) | 5 | – | 85 | 6.5 |
| 7.5 | ||||
| 10 | ||||
| 12.5 | ||||
| Group (2) | – | 5 | 85 | 6.5 |
| 7.5 | ||||
| 10 | ||||
| 12.5 | ||||
| Group (3) | – | 10 | 65 | 6.5 |
| 75 | ||||
| 85 | ||||
| 95 | ||||
| Group (4) | – | 10 | 85 | 4.5 |
| 5.5 | ||||
| 6.5 | ||||
| 7.5 | ||||
When the final step of enteric coating was performed on PECs, the influence of various coating times (30–75 s) on the thickness of the PECs needed to be considered. The coating duration determined the quantity of coating liquid that stuck to the swelling layer of the PECs, thereby affecting the final thickness of the capsules.
2.4 Determination of water–vapor permeability (WVP) and oxygen permeability (OP)
Pullulan enteric-coated thin films (PEFs) were prepared using the casting method. The WVP of PEFs was determined and improved according to the method developed by Ngo (Ngo et al., 2020) et al. At room temperature, a suitable amount of anhydrous calcium chloride was filled into a 10 mL vial (generally up to 5 mm below the bottle mouth). PEF was then cut into square pieces with dimensions of 2 × 2 cm2 using a digital thickness gauge to measure the film thickness. The film was then placed over the vial mouth and weighed. After weighing, the vial was placed in a drying chamber with deionized water at the bottom (maintaining a relative humidity (RH) of 100 %). After 24 h, the vial was taken out and weighed again. The weight change was calculated using equation (2):
Li (Li et al., 2022) et al. explained the method of determining the oxygen-barrier properties of films. In a typical procedure, 6.00–8.00 g of fresh soybean oil was placed in a 10 mL vial. PEF, with uniform thickness and no wrinkles, was selected and cut into 2 × 2 cm2 pieces. These pieces were then placed over the vial opening and stored in a CO2 incubator at 60 °C for 15 days. Afterwards, the vials were taken out and the peroxide value (PV) of the soybean oil was determined. The oxygen-barrier properties (OP) of the PEFs were evaluated based on the PV of soybean oil by using the formula shown in equation (3):
The determination of peroxide value (PV): Weigh 2.00 g–3.00 g of the sample and place it in a 250 mL iodine flask. Add 30 mL of a mixture of chloroform and glacial acetic acid (in a 2:3 ratio) to the 250 mL iodine flask, mix it thoroughly with the sample, and let it stand for a while until the mixed solution becomes clear and transparent. At the same time, prepare a saturated potassium iodide solution and store it in a brown reagent bottle. After the mixed solution becomes clear and transparent, add 1.00 mL of saturated potassium iodide solution to the solution, and the solution turns yellow. Shake with an ultrasonic oscillating machine for 30 s, then avoid light and let it stand for 3 min. Then transfer the solution to a 250 mL conical flask, add 100 mL of distilled water, shake well, and immediately titrate with 0.01 mol/L sodium thiosulfate solution. When the solution turns light yellow, add 1 mL of starch indicator, and the solution turns blue. Continue titrating with sodium thiosulfate solution until the blue color of the solution disappears, record the amount of sodium thiosulfate used, and use fresh soybean oil as a blank control, calculate the peroxide value of soybean oil according to the formula.
where PV is the peroxide value of soybean oil, meq/kg; V1 is the volume of sodium thiosulfate solution required for titration, mL; V0 is the volume of sodium thiosulfate standard solution required for blank titration, mL; C is the concentration of sodium thiosulfate solution, mol/L; and M is the mass of the sample to be titrated, g. Data are presented as the mean ± SD, n = 3.
2.5 Scanning electron microscopy (SEM)
Using the method described by Jongebloed (Jongebloed et al., 1990)et al., this study utilized SEM (TESCAN VEGA3, Czech Republic) to observe the gap between the main body and the cap, and randomly select 6 PECs and 6 GECs as samples. The sealed capsules were cut at the closure to expose the cross-section between the main body and the cap and then sputter-coated with platinum. The coated samples were analyzed with an ESEM Model Quanta 200 by FEI under a high vacuum at a high voltage of 12.5 kV with a spot size of 3.5. The maximum gap between the main body and the cap was located and measured from the cross-sectioned samples.
2.6 Atomic force microscopy (AFM)
The research was conducted by AFM (Bruker dimension icon, Germany) in tapping mode at room temperature. An OTESPA-R3 probe was selected, and the scanning speed was set at 0.5 Hz. PECs and GECs were used as samples. The capsule-shell area scanned was 50 μm2 with 256 points per line. The 2D and 3D surface morphology of the capsule shell was obtained. The images were quantitatively analyzed using Ara Research Co Nano expert software to obtain the roughness parameters of the capsule surface. Based on the current data, the average roughness (Sa) of the capsule shell was calculated.
2.7 Measurement of water content and analysis of hygroscopicity
PECs and GECs were dried in a water-content analyzer (Precisa XM60pro, Switzerland) at 105 °C until their weight remained constant. The mass of the capsules before and after reaching a constant weight was recorded, and then water content was calculated using formula (4):
The capsules, which were weighed carefully, were placed separately into two weigh bottles without lids. The bottles were then placed in a sealed, dry chamber at 25 °C and 75 % RH. The moisture absorption of the capsules was measured at 2, 4, 8, 24, 36, 48, 72, 96, and 120 h and calculated according to equation (5):
2.8 In vitro dissolution testing of enteric-coated aspirin capsules
This work selected 6 PECs and 6 GECs, and then each capsule was filled with 100 mg of aspirin. The dissolution test was conducted using a dissolution apparatus (RCZ-8A, China) with 900 mL of simulated gastric fluid (pH 1.5) as the dissolution medium. The test was performed at 37 ℃ and a rotation speed of 100 rpm for 2 h. The drug-release rate of aspirin in the simulated gastric fluid was calculated. Before quantification, the filtered samples were passed through a 0.22 μm polyethersulfone membrane, and the absorbance at 276 nm was measured using a UV-2700i UV–visible spectrophotometer (Shimadzu, Japan). The aspirin concentration in the filtered samples was then calculated based on a standard curve. After completion of the simulation of gastric fluid dissolution, the enteric-coated capsules were added to 900 mL of simulated intestinal fluid (pH 6.8). The rotation speed remained the same, and the duration of measurement was 4 h. Before quantification, the filtered samples were passed through a 0.22 μm polyethersulfone membrane and measured for absorbance at 265 nm by using a UV-2700i spectrophotometer (Shimadzu, Japan). This absorbance value was used to calculate the drug release of aspirin in the simulated intestinal fluid.
2.9 Data analysis
This study utilized Origin 2020 for statistical analysis and presented the results as the mean value ± SD of the measured values (n = 3). ANOVA and mean comparison were performed using the least significant difference test. A probability value (p) less than 0.05 was considered statistically significant.
3 Results and discussion
3.1 Polysaccharide characterization
According to the measurements (Fig. 2), the number-average molecular weight (Mn) of pullulan is 138910 g/mol, the weight-average molecular weight (Mw) is 196690 g/mol, the z-average molecular weight (Mz) is 250458 g/mol, the peak molecular weight (Mp) is 237851 g/mol, and the polydispersity index (Mw/Mn) is 1.4(Chen et al., 2023). Ding (Ding et al., 2020)et al. demonstrated that a higher Mw of pullulan leads to a lower dosage requirement in capsule preparation, and high-molecular-weight pullulan can save costs. Commercially available pullulan has an average molecular-weight distribution ranging from 8.8 × 104 g/mol to 1.0 × 105 g/mol (Liu et al., 2018, Ding et al., 2020), proving that the pullulan used in this study had a relatively high molecular weight and was suitable for capsule preparation (Cheng et al., 2011).
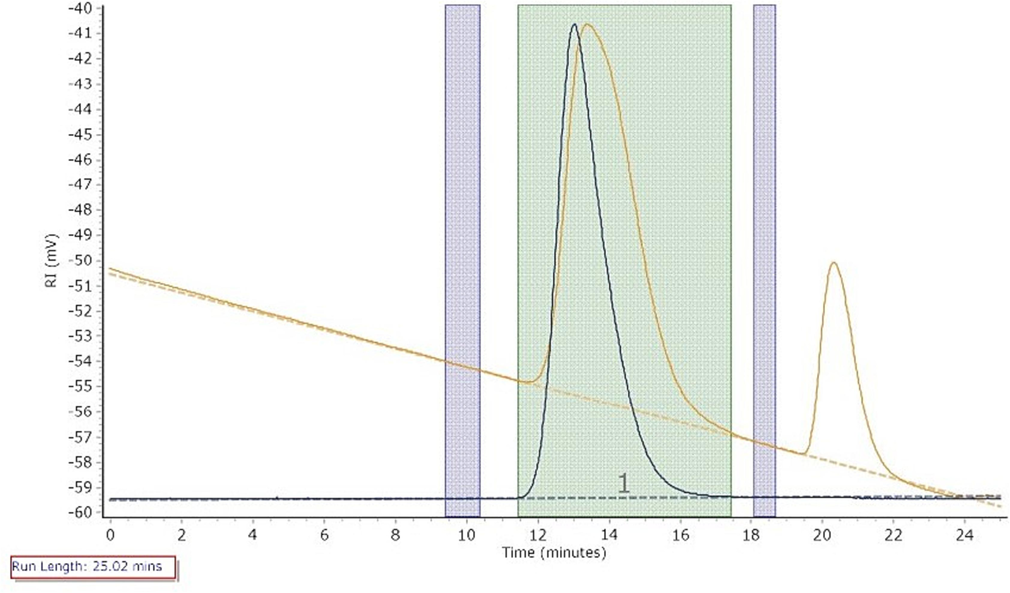
- Chromatogram of Pullulan Molecular Weight.
3.2 Preparation of PECs
This work conducted tests on the formulation of polymer preparations to obtain the optimal ratio for the swelling layer of PECs. The amount of pullulan and gellan gum affected the capsule-wall thickness. Lower concentrations of polymers resulted in thinner capsules that were prone to breakage during processing. Conversely, excessive concentrations of polymers can lead to an overly thick capsule wall, preventing proper fitting of the capsules (Sutthapitaksakul et al., 2022). When the concentration of pullulan ranged within 120–150 g/L, the capsule-formation rate could reach 100 % (Fig. 3A). To ensure a high capsule-formation rate and reduce costs, the optimal concentration of pullulan for preparing the enteric-capsule swelling layer was around 120 g/L. When the concentration of gellan gum is 5 g/L and 20 g/L, the capsule-forming rate is 0; when the concentration of gellan gum is 10 g/L, the capsule-forming rate is 100 %; when the concentration of gellan gum is 15 g/L, the capsule-forming rate is 50 %. Therefore, the optimal concentration of gellan gum is around 10 g/L. Under this condition, the capsule-forming rate is 100 %, and the thickness of the prepared capsule samples also meets the standard (Fig. 3B), indicating that the optimal concentration of gellan gum for preparing the enteric capsule swelling layer was 10 g/L. Glycerol affected the capsule's water content, which in turn affected its brittleness and capsule-formation rate. Therefore, the proportion of glycerol needed to be reduced. Based on Yang (Yang et al., 2020)et al.'s study on the water content of gelatin and non-gelatin capsules, maintaining the water content of plant capsules at around 8 %–10 % is recommended. Our experimental results showed that when the glycerol proportion in the enteric capsule swelling layer was 15 %, the water content of the capsule was around 9 % and the capsule-formation rate reached 100 % (Fig. 3C). As a medication binder, potassium citrate itself can help the gel form more effectively. Adding an appropriate amount of potassium citrate can help the pullulan-gellan gum mixed gel cover the mold better and solidify more quickly. When the concentration of potassium citrate was 1 g/L, the capsule-formation rate could reach 100 % (Fig. 3D). Based on these data, the optimal formulation for the enteric capsule swelling layer was determined to be 120 g/L pullulan, 10 g/L gellan gum, 15 % glycerol, and 1 g/L potassium citrate. The swelling layer of the PECs (pullulan hollow capsules, PHCs) prepared under this condition is shown in Fig. 4A, and the PHCs’ lengths are 21.0 ± 0.06 mm.
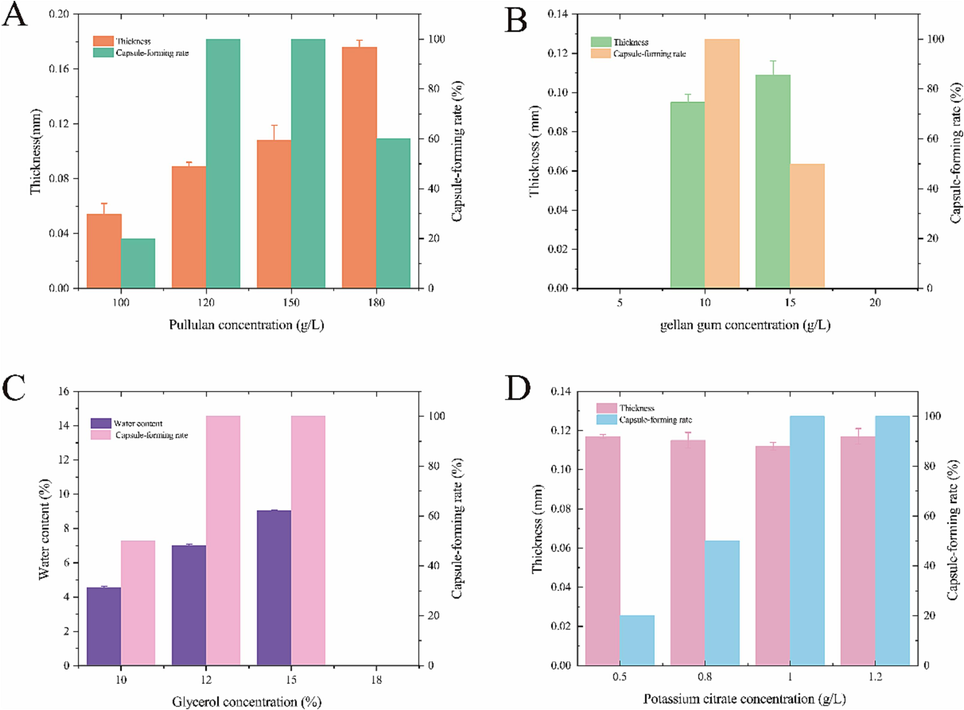
- Effect of pullulan concentration on the thickness of the PEC swelling layer and the capsule-forming rate (A); effect of gellan gum concentration on the thickness of the PEC swelling layer and the capsule-forming rate (B); effect of glycerol concentration on the water content and capsule-forming rate of PECs (C); and effect of potassium citrate concentration on the thickness of the PEC swelling layer and the capsule-forming rate (D).
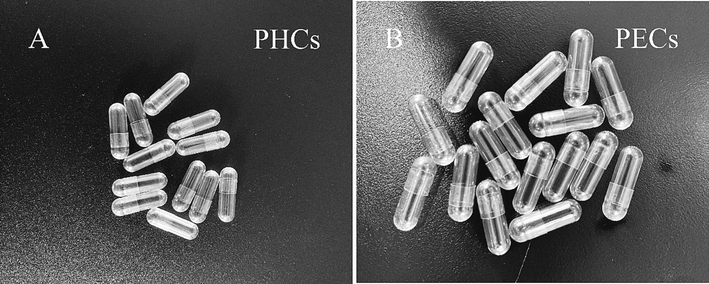
- A: Pullulan hollow capsules (PHCs). B: Pullulan enteric-coated capsules (PECs).
Polypropylene acid resin has a continuous carbon–hydrogen chain structure. It is not absorbed in the body and does not participate in the physiological metabolism of humans. After oral administration, it is excreted from the body and can be used as a material for enteric-coated capsules (O'Halloran et al., 2023). Enteric-type polypropylene acid resin can be divided into polypropylene acid resin II and polyacrylic acid resin III according to the different structures, proportions, and polymerization degrees of the excipients. While the monomer composition of both polyacrylic resins was kept identical, the ratio of the monomer composition differed. Polyacrylic acid resin II is an anionic copolymer synthesized by copolymerizing methyl acrylate and methyl methacrylate in a 1:1 ratio, whereas polyacrylic acid resin III is obtained by copolymerizing methyl acrylate and methyl methacrylate in a 1:2 ratio. Experimental data showed that capsules prepared with polyacrylic acid resin II underwent disintegration in 0.1 M HCl (pH 1.5) within 2 h at the concentration range of 5 %–12.5 % (w/v), which does not meet the requirements for enteric-capsule disintegration (Table 4). When the concentration of polyacrylic acid resin III exceeded 10 % (w/v), capsules prepared with it did not disintegrate in 0.1 M HCl (pH 1.5) within 2 h but completely disintegrated within 1 h in pH 6.8 PBS (Table 4). The variation in monomer composition ratio between polyacrylic acid resin II and polyacrylic acid resin III leads to a disparity in the number of free carboxyl groups. Polyacrylic acid resin II contains fewer free carboxyl groups compared to polyacrylic acid resin III. The carboxyl groups within the molecular structure remain non-dissociated in an acidic environment, causing the macromolecule to adopt a curled conformation. As the pH of the solution increases, the carboxyl groups gradually dissociate, resulting in the extension and solvation of the curled molecules. Higher solution pH leads to a faster dissolution rate. Due to the reduced number of carboxyl groups in polyacrylic acid resin II, it exhibits a higher propensity for disintegration in a high pH environment. To achieve the desired performance similar to that of polyacrylic acid resin III within a phosphate buffer solution while ensuring a high capsule-forming rate, it is necessary to increase the concentration of polyacrylic acid resin II in the enteric coating solution. However, such an increase in concentration inevitably raises the cost associated with the preparation of enteric capsules. Generally, the capsule-shell thickness more easily fit and was less likely to come off within the range of 0.12–0.15 mm. When the concentration of polyacrylic acid resin III was too high (e.g., >10 %), the prepared capsule wall became too thick, so fitting was difficult for the capsule shell (Fig. 6A). Therefore, the suitable concentration of polyacrylic acid resin III as an enteric-coating material was around 10 % (w/v).
| Materials | Polypropylene Acid Resin II | Polypropylene Acid Resin III | ||
|---|---|---|---|---|
| Proportion | Medium | |||
| 0.1 M HCl (pH 1.5) |
PBS (pH 6.8) | 0.1 M HCl (pH 1.5) |
PBS (pH 6.8) | |
| 5 % | 15.0 ± 5.0 min | – | 63.0 ± 2.0 min | – |
| 7.5 % | 30.0 ± 5.0 min | – | 89.0 ± 5.0 min | – |
| 10 % | 66.0 ± 3.0 min | – | >120.0 min | 30.0 ± 5.0 min |
| 12.5 % | 106.0 ± 5.0 min | – | >120.0 min | 45.0 ± 5.0 min |
Data are presented as the mean ± SD, n = 3, -indicates that the experiment was not conducted.
Polyacrylic acid resin Ⅲ is soluble only in polar organic solvents, so ethanol was chosen as the solvent. When the concentration of ethanol is too low, polyacrylic acid resin III cannot completely dissolve, resulting in incomplete enteric capsules (Fig. 5A). When the ethanol concentration was 75 % (v/v), the surface of the enteric capsule was rough and appeared to be in a white swollen state (Fig. 5B). When the ethanol concentration was 85 % (v/v), the prepared enteric capsule had a smooth and transparent surface with no defects in the capsule shell (Fig. 5C). When the ethanol concentration was 95 % (v/v), the thickness of the capsule cap and body did not exceed 0.10 mm, and the capsule wall was too thin (Fig. 6B), causing the capsule cap and body gap to be too large for fitting (Fig. 5D).

- Preparation of PECs using (A–D) 65%, 75%, 85%, and 95% ethanol-based coating solution, respectively.
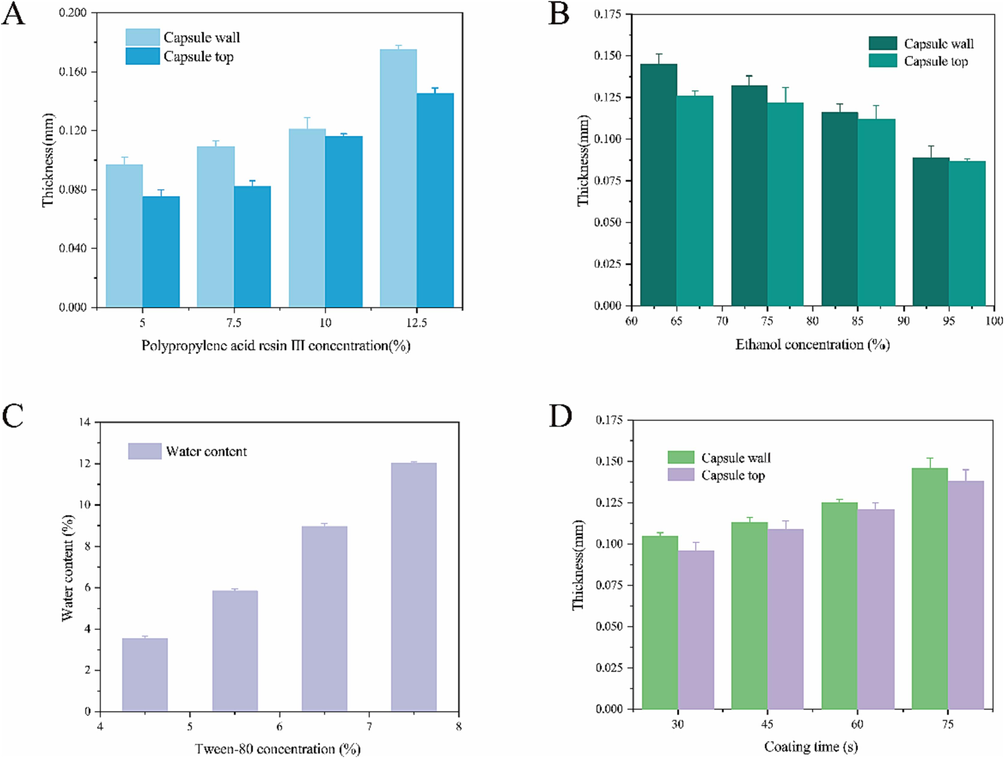
- Effect of the concentration of polypropylene acid resin III on the thickness of the capsule caps and bodies (A); effect of ethanol concentration on the thickness of the capsule caps and bodies (B); effect of Tween-80 concentration on the water content of PECs (C); and effect of coating time on the thickness of enteric-coated capsule caps and bodies (D).
The wetting agent Tween-80 affects the water content of enteric capsules, which in turn affects their brittleness (Zha et al., 2018). When the proportion of Tween-80 was around 4.5 % (w/v), the water content of the prepared enteric capsule shell did not reach 6 %, resulting in low water content and brittleness during capsule extraction. When the proportion of Tween-80 was around 7.5 % (w/v), the water content of the prepared capsule approached 12 %. For plant capsules, excessive water content caused the capsules to become too soft, easily absorb water, and swell, leading to cap separation (Fig. 6C). Therefore, the optimal proportion of Tween-80 to be added to the coating solution was around 6.5 % (w/v).
The coating duration can affect the capsule-shell thickness (Fig. 6D). When the coating time was around 30 s, the swelling layer of the capsule did not absorb enough coating liquid, resulting in a thinner capsule wall (0.090 ± 0.005 mm) that was prone to rupture (Fig. 7A). If the coating time exceeded 60 s, the capsule surface carried too much coating liquid, which affected the transparency of the capsule and led to an excessively thick enteric coating after formation. Consequently, the capsule had difficulty fitting properly (Fig. 7D). However, when the coating time was 60 s, the thickness of the capsule cap and body ranged within 0.12–0.15 mm, allowing for easy fitting and preventing detachment (Fig. 7C). Therefore, controlling the coating time to around 60 s when performing enteric coating on the capsule shell was recommended.

- PECs with various coating times: (A–D) 30, 45, 60, and 75 s, respectively.
The enteric-coated capsules described in this study were obtained by secondary coating on the hollow capsules. An effective enteric-coating solution can provide the enteric-coated capsules with the ability to remain intact and not rupture in a low-pH environment. Simultaneously, it can censure rapid disintegration of the enteric-coated capsules in an appropriately high pH environment, allowing for rapid drug release and therapeutic effect (Patrick et al., 2016). The optimal formulation for the enteric-coating solution of PECs was to dissolve 10 % polyacrylic acid resin III in 85 % ethanol and add 6.5 % Tween-80 during dissolution. After stirring evenly, the coating time was controlled to around 60 s. The PECs prepared by the secondary coating of the enteric-coating solution are shown in Fig. 4B, and the PECs’ lengths are 21.0 ± 0.11 mm. They had a beautiful and delicate appearance and were transparent and free of impurities.
3.3 Characterization of PEFs
The PV value of the small bottle of soybean oil covered by PEFs is 42.1 ± 0.03 meq/kg, and the WVP value of PEFs is 0.036 ± 0.009 g·m·(m2·d)–1. Firstly, from the data, it can be concluded that in this experiment, the peroxide value of the soybean oil is low, and the OP value of PEFs is also low, proving that only a small amount of oxygen has permeated through the PEFs into the bottle, indicating good oxygen barrier properties of PEFs. The low WVP value of PEFs proves that only a small amount of moisture has permeated through the PEFs and reacted with calcium chloride, indicating a low water vapor transmission rate and good moisture barrier properties of PEFs. Ding (Ding et al., 2020) et al. reported that the PV value of soybean oil covered by Prussian-cold gel composite film is 48.9 meq/kg, and the WVP value of the composite film is 0.06 g·m·(m2·d)−1. In comparison, the oxygen and water vapor barrier properties of the PEFs prepared in this research have been improved, particularly showing a significant improvement in moisture barrier properties, which may be attributed to the external coating of the enteric-coating solution prepared with polyacrylic acid resin III. The polyacrylic acid resin III itself was insoluble in water but soluble in organic solvents. Its unique molecular structure was tightly and orderly arranged, forming a film to prevent water molecules and oxygen in the air from entering the capsule. The performance of the PECs prepared in this experiment was better in terms of barrier properties. It prevented the oxidation of the drugs filled in the capsule, enhanced drug stability, and increased drug storage time.
3.4 SEM analysis
Before the start of this study, the PECs were subjected to a tightness test and showed no obvious powder leakage. The above results were based on visual inspection, whereas the size of the gaps between the capsule caps and bodies was determined by SEM analysis (Fig. 8). The enteric coating of the PECs showed good adhesion with the swelling layer, and no layering phenomenon was noticeable. The average gap between the PEC caps and bodies was measured to be 15.36 ± 0.12 μm (Fig. 8A), whereas the average gap between the GEC caps and bodies was measured to be 70.12 ± 0.09 μm (Fig. 8B). The gap between the PECs was significantly smaller than that of GECs. The extremely small gap between the capsules has the PECs high sealing and low leakage properties, confirming that the PECs in this study had good physical characteristics.

- SEM images of homemade PECs (A) and GECs (B).
3.5 AFM analysis
This study utilized AFM to scan the surface of PECs for more precise observation of their roughness. The cap and body surface of the PEC exhibited fewer small particles (Fig. 9A and 9B). Conversely, the GEC cap surface had larger particles (Fig. 9C). The GEC capsule body also had a larger number of small particles (Fig. 8D). The capsule cap and body surfaces of the GEC had a greater height difference than the PEC surface (Fig. 10). Based on calculations, the average roughness (Sa) of the PEC was 6.00 ± 0.12 nm. At the same time, the Sa of the GEC was 26 ± 0.05 nm. A smaller Sa indicated a lower brittleness. PECs had a more uniform surface, lower roughness, and reduced brittleness than GECs (Ramezani Kalmer et al., 2022). The presence of Pullulan imparted good toughness to the PECs. The double-helix structure of gellan gum itself was greatly affected by temperature, resulting in poor flexibility of the individually formed thin film. Meanwhile, pullulan comprised alternating α-(1–4) glycosidic bonds and α-(1–6) glycosidic bonds, and this alternating structure gave pullulan good flexibility and solubility. The combination of these two materials formed a swelling layer, enhancing the toughness of the enteric capsules. The shell formed by pouring the mixed solution of pullulan-gellan gum into the mold simultaneously not only serves as the swelling layer of PECs but also provides a good structural basis for PECs. The mixed solution adheres to the mold, naturally flows downward from top to bottom, and forms a smooth film on the surface of the mold through rotation and extension. The formed film is flexible and smooth, with no wrinkles or ruptures on the mold surface. Based on this, the mold is immersed in an enteric coating solution prepared by completely dissolving polyacrylic acid resin III in ethanol, to completely cover the mold and then dried and cut into PECs. On the one hand, due to the smooth and unwrinkled surface of the capsule swelling layer, the coating solution can smoothly cover it, so the surface of the formed PECs has no ruptured or wrinkled areas. On the other hand, due to the complete dissolution of the prepared enteric coating solution, the polyacrylic acid resin III and ethanol exhibit good compatibility, with no undissolved particles. As a result, the prepared PEC surface has no wrinkles or large particles, and observation by AFM shows that the roughness is small.
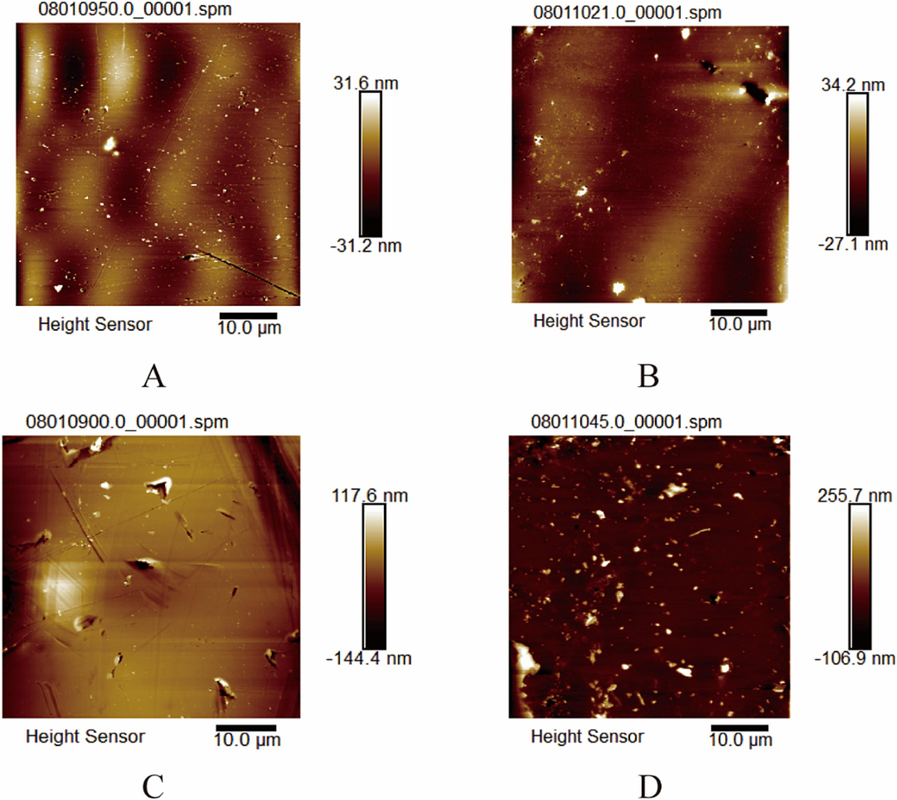
- AFM 2D image of homemade PEC cap (A), homemade PEC body (B), GEC cap (C), and GEC body (D).
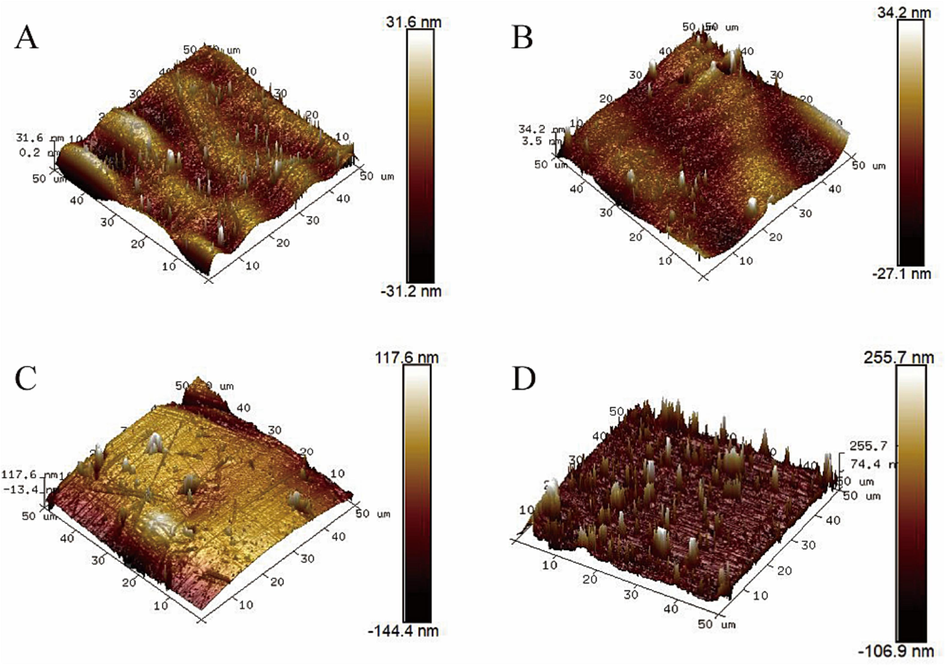
- AFM 3D image of homemade PEC cap (A), homemade PEC body (B), GEC cap (C), and GEC body (D).
3.6 Analysis of water content and hygroscopicity
Before analyzing the moisture-absorption rate of the two types of capsules in this study, their water content was tested, and then the average water content of three identical capsules was calculated. Finally, the average water content of the PECs was found to be 8.12 %, whereas the average water content of the GECs was 12.39 %. The water content of the pullulan polysaccharide-κ-carrageenan-based hard capsules prepared by Zhang (Zhang et al., 2020)et al. ranges from 11.87 % to 12.85 %. Barham (Barham et al., 2015) et al. also confirmed that the water content of gelatin hollow hard capsules commonly used for pharmaceutical purposes ranges from 12.03 % to 16.45 %, thereby demonstrating that gelatin hard capsules generally have a higher water content than pullulan-based hard capsules. The moisture sorption curves of GECs and PECs at 75 %RH are shown in Fig. 11. The moisture-sorption rates of both types of enteric capsules increased with time and gradually reached equilibrium after 100 h, where the moisture-sorption rate remained essentially unchanged (Fig. 11). This equilibrium point was referred to as the equilibrium moisture-sorption rate at constant temperature (Ciper and Bodmeier, 2006). The equilibrium moisture uptake of GECs was 20 %. Conversely, the equilibrium moisture uptake of the PECs prepared in this study was 15 % (Fig. 11). Yang (Yang et al., 2020) et al. investigated the hygroscopicity of pullulan hard capsules and gelatin hard capsules. Within 120 h, the equilibrium moisture uptake of gelatin hard capsules was 19.01 %, whereas the equilibrium moisture uptake of pullulan hard capsules was 16.5 %. Gelatin is a water-soluble protein containing a large amount of carboxyl groups, amino groups, and hydroxyl groups. These functional groups can easily form hydrogen bonds with water molecules, giving gelatin a strong hygroscopic ability. Compared with GECs, PECs had weaker hygroscopic ability at the same temperature and humidity. Additionally, due to the intestinal coating of GECs, they had enhanced water–vapor barrier properties. Therefore, PECs had better quality and can effectively prevent capsule deliquescence or mold growth.
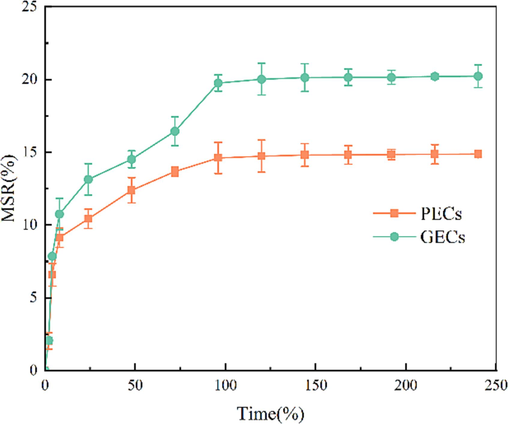
- Comparison of MSR (Moisture sorption rate) between homemade PECs and GECs within 240 h.
3.7 Extracorporeal drug delivery
The PECs and GECs had drug-release rates of less than 10 % in simulated gastric fluid for 120 min. In simulated intestinal fluid, the drug-release rate reached 80 % within the first 30 min and then continued to release. When the drug-release rate reached around 90 % of the indicated amount of aspirin, the release rate remained unchanged, reaching equilibrium (Fig. 12A and 12C). This finding met the requirements of the pharmacopeia for aspirin enteric capsules in terms of in vitro drug release, i.e., the release amount within 2 h was less than 10 % of the indicated amount of aspirin in simulated gastric fluid, and the release amount within 45 min was greater than 80 % of the indicated amount of aspirin in simulated intestinal fluid. GECs showed a proportional relationship between drug release and time within 30 min in simulated intestinal fluid. After 30 min, drug release reached equilibrium (Fig. 12D). The entire drug-release process of GECs in simulated intestinal fluid followed the principles of controlled continuous drug delivery. Conversely, in the first 20 min of simulating intestinal fluid, a minimal amount of dissolution of the PECs occurred. However, starting from the 20th minute, the drug-release rate rapidly increased, and the amount of drug released increased. Within only 5 min, it reached 80 % of the labeled amount of aspirin. Afterward, the drug-release rate decreased and gradually reached equilibrium after 30 min of entering the simulated intestinal fluid (Fig. 12B). The entire process had the typical characteristics of pulsatile drug delivery, where the coating prevented premature release of the drug in gastric fluid. After a certain period, the capsule dissolved in intestinal fluid, leading to a rapid and significant release of the drug within a short timeframe (Patil and Shahiwala, 2014). Throughout the entire enteric-capsule system, a large number of carboxyl groups in the polyacrylic acid resin III gradually dissociated in a high-pH environment, causing the curled molecules to unfold and dissolve. The swelling layer of the enteric capsule formed a three-dimensional network structure by combining with pullulan and gelatin, which absorbed water and weakened the membrane layer. After the enteric coating dissolved, a large amount of buffer solution water molecules came into contact with the swelling layer. When the swelling pressure exceeded the strength of the polymer membrane, the swelling layer ruptured the capsule completely disintegrated, and aspirin started to dissolve (Bussemer et al., 2003). The advantage of this method of administration is that it can protect the gastric mucosa from the damage inflicted by gastric irritants while minimizing the first-pass effect of drug release and ensuring drug efficacy (Pandit et al., 2017).
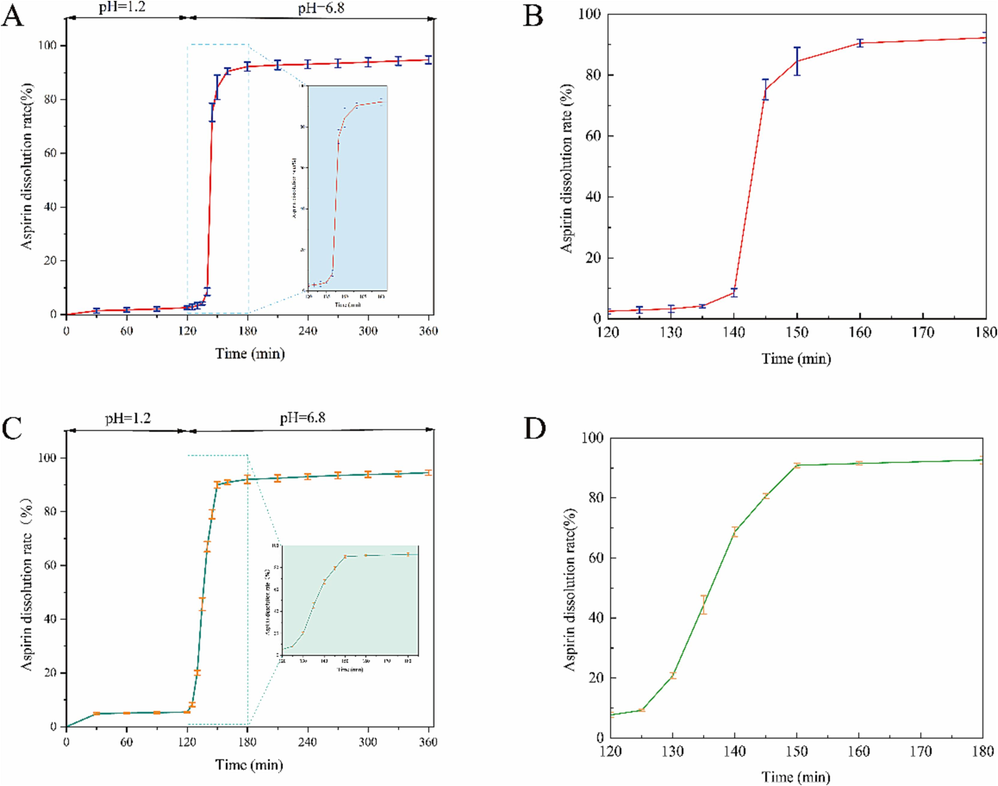
- In vitro drug-release curve of PECs in simulated gastric fluid and simulated intestinal fluid (A). Drug-release profile of PECs within the first hour in simulated intestinal fluid (B). In vitro drug-release curve of GECs in simulated gastric fluid and simulated intestinal fluid (C). Drug-release profile of GECs within the first hour in simulated intestinal fluid (D).
4 Conclusion
The main objective of this study is to design and develop a novel type of PECs different from GECs, which combines microbial polysaccharides with other materials to prepare enteric-coated hard capsules. The combination of pullulan and gellan gum forms the swelling layer of the capsule, and after formulation, polyacrylic acid resin III acts as the enteric coating. Compared to GECs, PECs have better barrier properties, higher sealing performance, and lower roughness in terms of physical aspects. Through design, PECs significantly reduce water content and hygroscopicity. These advantages allow PECs, when filled with drugs, to better prevent drug deterioration and ensure drug stability during storage and transportation. The subsequent drug release characteristics of PECs indicate their potential for pulsatile drug release. This method of administration is of great significance for diseases with a peak in morning onset, such as hypertension, asthma, myocardial infarction, and angina pectoris, by providing the appropriate medication at the right time to achieve the best therapeutic effect. Together with other benefits of microbial extracellular polysaccharides, this study provides a simple, reliable, and cost-effective method for controlling drug release. Therefore, the PECs developed in this study may have broad prospects in clinical trials or customized formulations for specific drugs.
Funding sources
This work was financially supported by the Natural Science Foundation of Shandong Province (grant ZR2022QH173), the Science and technology innovation Program of Weifang Medical University (grant 041125), the National Natural Scientific Foundation of China (81971998).
References
- Pullulan based derivatives: synthesis, enhanced physicochemical properties, and applications. Drug Deliv.. 2022;29:3328-3339.
- [CrossRef] [Google Scholar]
- Moisture diffusion and permeability characteristics of hydroxypropylmethylcellulose and hard gelatin capsules. Int. J. Pharm.. 2015;478:796-803.
- [CrossRef] [Google Scholar]
- A pulsatile drug delivery system based on rupturable coated hard gelatin capsules. J. Control Release. 2003;93:331-339.
- [CrossRef] [Google Scholar]
- High-level production of pullulan and its biosynthesis regulation in Aureobasidium pullulans BL06. Front. Bioeng. Biotechnol.. 2023;11:1131875.
- [CrossRef] [Google Scholar]
- Preparation and characterization of nanostarch-based green hard capsules reinforced by cellulose nanocrystals. Int. J. Biol. Macromol.. 2021;167:1241-1247.
- [CrossRef] [Google Scholar]
- Pullulan: biosynthesis, production, and applications. Appl. Microbiol. Biotechnol.. 2011;92:29-44.
- [CrossRef] [Google Scholar]
- Modified conventional hard gelatin capsules as fast disintegrating dosage form in the oral cavity. Eur. J. Pharm. Biopharm.. 2006;62:178-184.
- [CrossRef] [Google Scholar]
- Enteric coated HPMC capsules designed to achieve intestinal targeting. Int. J. Pharm.. 2002;231:83-95.
- [CrossRef] [Google Scholar]
- An alternative hard capsule prepared with the high molecular weight pullulan and gellan: processing, characterization, and in vitro drug release. Carbohydr. Polym.. 2020;237:116172
- [CrossRef] [Google Scholar]
- Carboxyl-modified nanocellulose (cNC) enhances the stability of cNC/pullulan bio-nanocomposite hard capsule against moisture variation. Carbohydr. Polym.. 2024;328
- [CrossRef] [Google Scholar]
- Gelatin structure and composition linked to hard capsule dissolution: a review. Food Hydrocoll.. 2015;43:360-376.
- [CrossRef] [Google Scholar]
- Preparation and characterization of pH- and temperature-sensitive pullulan microspheres for controlled release of drugs. Biomaterials. 2008;29:2767-2775.
- [CrossRef] [Google Scholar]
- Bio-relevant dissolution testing of hard capsules prepared from different shell materials using the dynamic open flow through test apparatus. Eur. J. Pharm. Sci.. 2014;57:264-272.
- [CrossRef] [Google Scholar]
- Gelatin and non-gelatin capsule dosage forms. J. Pharm. Sci.. 2017;106:1453-1465.
- [CrossRef] [Google Scholar]
- Morphology of donor lens-capsule material studied by SEM. Doc Ophthalmol.. 1990;75:343-350.
- [CrossRef] [Google Scholar]
- Characterization and application in packaging grease of gelatin-sodium alginate edible films cross-linked by pullulan. Polymers (Basel). 2022;14
- [CrossRef] [Google Scholar]
- α-Amylase, glucoamylase and isopullulanase determine molecular weight of pullulan produced by Aureobasidium melanogenum P16. Int. J. Biol. Macromol.. 2018;117:727-734.
- [CrossRef] [Google Scholar]
- Characteristics and antimicrobial properties of active edible films based on pectin and nanochitosan. Int. J. Mol. Sci.. 2020;21
- [CrossRef] [Google Scholar]
- Two-photon polymerization: fundamentals, materials, and chemical modification strategies. Adv. Sci. (weinh). 2023;10:e2204072.
- [Google Scholar]
- Recent advancement and technological aspects of pulsatile drug delivery system - a laconic review. Curr. Drug Targets. 2017;18:1191-1203.
- [CrossRef] [Google Scholar]
- Patented pulsatile drug delivery technologies for chronotherapy. Expert Opin. Ther. Pat.. 2014;24:845-856.
- [CrossRef] [Google Scholar]
- Safety and tolerability of extended-release acetylsalicylic acid capsules: a summary of double-blind comparative studies. Future Cardiol.. 2016;12:627-638.
- [CrossRef] [Google Scholar]
- Pullulan: an exopolysaccharide and its various applications. Carbohydr. Polym.. 2013;95:540-549.
- [CrossRef] [Google Scholar]
- Emerging trends in pullulan-based antimicrobial systems for various applications. Int. J. Mol. Sci.. 2021;22
- [CrossRef] [Google Scholar]
- Effect of different molecular weights of polyethylene glycol as a plasticizer on the formulation of dry powder inhaler capsules: investigation of puncturing size, morphologies, and surface properties. Arab. J. Chem.. 2022;15:104239
- [CrossRef] [Google Scholar]
- Duodenum-specific drug delivery: in vivo assessment of a pharmaceutically developed enteric-coated capsule for a broad applicability in rat studies. Int. J. Pharm.. 2012;422:338-340.
- [CrossRef] [Google Scholar]
- Pullulan in biomedical research and development - a review. Int. J. Biol. Macromol.. 2021;166:694-706.
- [CrossRef] [Google Scholar]
- Effect of a superdisintegrant on disintegration of orally disintegrating tablets determined by simulated wetting test and in vitrodisintegration test. Pharmazie. 2022;77:287-290.
- [CrossRef] [Google Scholar]
- Eudragit: a technology evaluation. Expert Opin. Drug Deliv.. 2013;10:131-149.
- [CrossRef] [Google Scholar]
- Pullulan biosynthesis and its regulation in Aureobasidium spp. Carbohydr. Polym.. 2021;251:117076
- [CrossRef] [Google Scholar]
- Moisture sorption and desorption properties of gelatin, HPMC and pullulan hard capsules. Int. J. Biol. Macromol.. 2020;159:659-666.
- [CrossRef] [Google Scholar]
- Improving dissolution properties by polymers and surfactants: a case study of celastrol. J. Pharm. Sci.. 2018;107:2860-2868.
- [CrossRef] [Google Scholar]
- Developing gelatin-starch blends for use as capsule materials. Carbohydr. Polym.. 2013;92:455-461.
- [CrossRef] [Google Scholar]
- Effects of κ-carrageenan on pullulan's rheological and texture properties as well as pullulan hard capsule performances. Carbohydr. Polym.. 2020;238:116190
- [CrossRef] [Google Scholar]
- Gelatin-polyvinyl alcohol film for tissue engineering: a concise review. Biomedicines. 2021;9
- [CrossRef] [Google Scholar]







