Translate this page into:
Preparation of gelatin/Ag NPs under ultrasound condition: A potent and green bio-nanocomposite for the treatment of pleomorphic hepatocellular carcinoma, morris hepatoma, and novikoff hepatoma
⁎Corresponding author. bkarmakar@ghcollege.ac.in (Bikash Karmakar)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Regarding applicative, facile, green chemical research, a bio-inspired approach is being reported for the sonochemical synthesis of Ag nanoparticles by gelatin as a natural reducing/stabilizing and solid support agent without using any toxic and harmful reagent. The biosynthesized Ag NPs@gelatin bionanocomposite were characterized by advanced physicochemical techniques like ultraviolet–visible (UV–Vis), Fourier Transformed Infrared spectroscopy (FT-IR), Scanning Electron Microscopy (SEM), High-Resolution Transmission Electron Microscopy (HR-TEM), Energy Dispersive X-ray spectroscopy (EDX), and X-ray Diffraction (XRD) study. It has been established that gelatin-stabilized silver nanoparticles have a spherical shape with a mean diameter from ∼5–10 nm. To survey the antioxidant potentials of Ag NPs@gelatin, one of the common antioxidant techniques i.e., DPPH was used. The results of DPPH test proved excellent antioxidant properties of Ag NPs@gelatin in comparison to the positive control used i.e., butylated hydroxytoluene. The IC50 of Ag NPs@gelatin was 250 µg/mL in the antioxidant test. Determination of anti-liver cancer effects of Ag NPs@gelatin was carried out by the MTT assay and against pleomorphic hepatocellular carcinoma (SNU-387), morris hepatoma (McA-RH7777), and novikoff hepatoma (N1-S1 Fudr) cell lines. The results of MTT assay confirmed removing SNU-387, McA-RH7777, and N1-S1 Fudr cell lines after treating with low concentrations of Ag NPs@gelatin. The IC50 of the Ag NPs@gelatin was 192, 144, and 228 µg/mL against SNU-387, McA-RH7777, and N1-S1 Fudr cell lines, respectively. The best result of anti-human liver cancer effects of Ag NPs@gelatin against the above cell lines was seen in the case of the N1-S1 Fudr cell line. According to the above results, significant anti-liver cancer effects of Ag NPs@gelatin is confirmed.
Keywords
Gelatin
Silver nanoparticles
Pleomorphic hepatocellular carcinoma
Morris hepatoma
Novikoff hepatoma
1 Introduction
Hepatocellular carcinoma (HCC) is the most common type of primary liver cancer. Hepatocellular carcinoma occurs most often-in people with chronic liver diseases, such as cirrhosis caused by hepatitis B or hepatitis C infection (Shetty et al., 2016; Forner et al., 2012). The risk of hepatocellular carcinoma, the most common type of liver cancer, is higher in people with long-term liver diseases. It's also higher if the liver is scarred by infection with hepatitis B or hepatitis C. Hepatocellular carcinoma is more common in people who drink large amounts of alcohol and who have an accumulation of fat in the liver (Shetty et al., 2016). The treatments are with surgery, liver transplant surgery, destroying cancer cells with heat or cold, delivering chemotherapy or radiation directly to cancer cells, radiation therapy, targeted drug therapy, and immunotherapy (Forner et al., 2012). The biological distribution of therapeutic agents can be a fundamental factor in these fractures. Inadequate concentration at unwanted concentration and target sites elsewhere, leading to dose-limiting poisoning (Shetty et al., 2016; Forner et al., 2012). The biological distribution of drug agents is controlled largely by the drugs ability to penetrate biological barriers. Strategy for Adding Targeting Sections to Therapeutic Nanoparticles to Improve Location Specification to date, despite 30 years of effort in pharmaceutical companies and many laboratories, it has not yet been able to produce clinically approved drugs (Trojer et al., 2013; You et al., 2012). This failure is because the addition of molecular agents increases the targeting of cognitive characteristics. However, it does so in the face of much greater difficulty in managing biological barriers (Forner et al., 2012; Trojer et al., 2013; You et al., 2012).
Nanotechnology is defined in different ways in several countries, which affects the nanodrugs clinical validation. However, what these different definitions have in common is the use of nanoscale structures. There are several distinct benefits to using nanotechnology in the diseases treatment (Mao, 2016; De Jong and Borm, 2008). Nanoparticles, especially metal nanoparticles and metal oxides, have been widely used by medical consumers and manufacturers. The mechanism of nanoparticle-induced toxicity against cancer cells is the production of reactive oxygen species (ROS) (Borm et al., 2006; Stapleton and Nurkiewicz, 2014). Excessive production of reactive oxygen species can lead to oxidative stress, disruption of normal physiological maintenance, and oxidation regulation. These effects in turn lead to DNA damage, unregulated cell signaling pathways, changes in cell evolution, cytotoxicity, apoptotic death, and the onset of cell death (Borm et al., 2006; Stapleton and Nurkiewicz, 2014; Patra et al., 2018; Kiasat et al., 2013).
Critical-deterministic factors can affect the production of reactive oxygen species. These critical-deterministic factors include shape, size, nanoparticle surface area, particle surface baroelectricity, surface-forming groups, Particle solubility, metal ion emission from nanomaterials and nanoparticles, optical activation, model of cell reactions, inflammatory effects and ambient pH (Celardo et al., 2011; Itani and Al Faraj, 2019; Guilbert et al., 1996; Miller and Krochta, 1997). Metal nanoparticles and oxides of metal nanoparticles due to their optical properties due to the large active area and high atomic number, amplify the photoelectric and Compton effects of both X-ray and gamma-ray interactions with the adsorbent in the diagnostic and therapeutic range. Finally, they can lead to the development of methods for the destruction of tumor cells and reduce their survival with minimal side effects in radiation therapy (Patra et al., 2018; Kiasat et al., 2013; Celardo et al., 2011; Itani and Al Faraj, 2019; Guilbert et al., 1996). As a result, increasing industrial knowledge in the field of scalable nanoparticle synthesis, along with the design of multifunctional nanoparticles, will dramatically change the strategies of microenvironmental preparation and therapeutic-diagnostic nanoparticles for cancer treatment (Itani and Al Faraj, 2019; Guilbert et al., 1996; Miller and Krochta, 1997).
Gelatin is made up of a mixture of peptides and proteins produced by partial hydrolysis of naturally occurring collagen. The hydroxyl and carboxyl groups on its chain backbones are one of its amazing features that enables the possibility to form hydrogen bonds with metals (Mi, 2005; Ahmad et al., 2011; Thi and Nguyen, 2016). Due to non-toxic effects of gelatin to human, it is interesting and valuable compounds in pharmaceuticals and biomedical usages. Therefore, we used gelatin in research work as an ecofriendly natural polymer with minimum side effects and importantly as a natural reducing/stabilizing and solid support agent for fabrication silver nanoparticles without using any toxic and harmful reagent. Then, the prepared Ag NPs@gelatin bionanocomposite were characterized by advanced physicochemical techniques. In addition, the properties of Ag NPs@gelatin bionanocomposite against common human liver cancer cell lines i.e. pleomorphic hepatocellular carcinoma (SNU-387), morris hepatoma (McA-RH7777), and novikoff hepatoma (N1-S1 Fudr) were evaluated.
2 Experimental
2.1 Materials and methods
The chemicals required for catalyst synthesis and catalyst applications were purchased from Sigma Aldrich.
2.2 Synthesis of Ag NPs@gelatin bionanocomposite
To initiate the reaction 20 mL of a freshly prepared aqueous solution of AgNO3 (10 mM) was added to 100 mL of an aqueous gelatin suspension under ultrasonic conditions and left for 10 min at 60 °C. Then the pH of the suspension was adjusted to 11.0 (NaOH, 3 wt%) and the reaction mixture was kept with sonication at 60 °C for 60 min. The progress of the reaction could be monitored by the change in color from watery yellow to brown color of the aqueous solution was due to the excitation of the surface plasmon resonance (Stapleton and Nurkiewicz, 2014) and SPR band which both play an important role in the confirmation of silver nanoparticles formation (Patra et al., 2018). The prepared Ag NPs@gelatin were collected by centrifugation and washed several times with DI-water. Ag content in the catalyst was 3.6 mmol/g, as determined by ICP-OES analysis.
2.3 Antioxidant activities of Ag NPs@gelatin bionanocomposite
α, α-diphenyl-β-picrylhydrazyl (DPPH) free radical scavenging method offers the first approach for evaluating the antioxidant potential of a compound, an extract or other biological sources. This is the simplest method, wherein the prospective compound or extract is mixed with DPPH solution and absorbance is recorded after a defined period. However, with the advancement and sophistication in instrumental techniques, the method has undergone various modifications to suit the requirements, even though the basic approach remains same in all of them (Lu et al., 2021).
In this method, 1 mL of different concentrations of the nanoparticles (0–1000 µg/ml) (with 1 mL of DPPH (300 µmol/l) combined and then the final volume of the combination with methanol reached 4000 µl. The falcons were then vertexed and kept in the dark for 60 min. The absorbance was read at 517 nm. The DPPH radical inhibition percentage was calculated using the following equation (Lu et al., 2021):
2.4 Anti-liver carcinoma properties of Ag NPs@gelatin bionanocomposite
Cell culture is the process by which cells are grown under controlled conditions, generally outside their natural environment. After the cells of interest have been isolated from living tissue, they can subsequently be maintained under carefully controlled conditions. These conditions vary for each cell type, but generally consist of a suitable vessel with a substrate or medium that supplies the essential nutrients (amino acids, carbohydrates, vitamins, minerals), growth factors, hormones, and gases (CO2, O2), and regulates the physio-chemical environment (pH buffer, osmotic pressure, temperature). Most cells require a surface or an artificial substrate to form an adherent culture as a monolayer (one single-cell thick), whereas others can be grown free floating in a medium as a suspension culture. The lifespan of most cells is genetically determined, but some cell culturing cells have been “transformed” into immortal cells which will reproduce indefinitely if the optimal conditions are provided. In practice, the term “cell culture” now refers to the culturing of cells derived from multicellular eukaryotes, especially animal cells, in contrast with other types of culture that also grow cells, such as plant tissue culture, fungal culture, and microbiological culture (of microbes). The historical development and methods of cell culture are closely interrelated to those of tissue culture and organ culture. Viral culture is also related, with cells as hosts for the viruses. The laboratory technique of maintaining live cell lines (a population of cells descended from a single cell and containing the same genetic makeup) separated from their original tissue source became more robust in the middle 20th century (Lu et al., 2021).
In this study, common human liver cancer cell lines i.e. pleomorphic hepatocellular carcinoma (SNU-387), morris hepatoma (McA-RH7777), and novikoff hepatoma (N1-S1 Fudr) were used to evaluate the anticancer effect of Ag NPs@gelatin bionanocomposite on cell culture.
These cells in DMEM culture medium (Gibco, USA) with 10% FBS (Gibco, USA) and penicillin/streptomycin (100 μl/100 μg/ml) in an incubator containing 5 % Carbon dioxide with 90% humidity was stored at 37 °C. Then, when about 80% of the flask was filled, cell passage was performed and about 5 × 104 cells (per square centimeter) were placed in 24 house bacterial petri dishes in the usual environment. The cells were treated with different concentrations of nanoparticles 24 h later and kept in this condition for 3 days. The survival rate of cultured cells was prepared with different concentrations. In this experiment, cells were cultured at 3 × 104 cells/well in 24-well plates and kept in an incubator at 37 °C for 24 h. Then the old culture medium was taken out of the wells and the cells were treated with different concentrations of nanoxidro. This test was performed on the first, second and third days after exposing the cells to the compounds; thus, at the appropriate time after culturing the cells in plates of 24 cells, the culture medium was removed and about 300 μl of fresh medium containing 30 μl of MTT solution was added to each cell. After 3–4 h of incubation at 37 °C, MTT solution is removed and 200 μl (Dimethyl Sulfoxide, Merck, USA, 100%) DMSO is added to each house. Then the sample absorption was read at 570 wavelengths using ELISA rider (Expert 96, Asys Hitch, Ec Austria). This experiment was repeated 3 times and each time, four wells were considered for each nano oxide concentration. Cell survival percentage was evaluated by the following formula (Lu et al., 2021):
The results were evaluated as Mean ± SE using SPSS software version 12 and statistical tests of variance of completely randomized block design. Drawing graphs in Excel software was performed and the significance level of the differences was considered p < 0.05.
3 Results and discussion
Regarding the cytotoxicity of metal nanoparticles, several studies have been performed and it has been determined that the toxicity of metal nanoparticles is more than the non-nanoparticle state or mass of metal materials (Yang et al., 2011; Xinli, 2012; Allen, 2002; Byrne et al., 2008). However, the effects of nanoparticles on human health or the environment in general are still debated and need further research and study. Studies have also shown that metal nanoparticles can be effective at the cellular, microcellular, and molecular levels, such as proteins and genes (Gao et al., 2015; Mohammed et al., 2016; Li and Gu, 2014; Torchilin, 2007). Evaluation of metal nanoparticles on cellular and animal models indicates that these particles cause cytotoxicity by inducing the production of passive oxygen species. In this regard, a decrease in the amount of glutathione, an increase in the amount of passive oxygen and an increase in lipid peroxidation have been reported in samples treated with metal nanoparticles (Torchilin, 2007; Pranali, 2013; Zhang et al., 2014; Gao et al., 2002; Davis et al., 2008). According to previous research, metal nanoparticles have a toxic activity on cancerous cells, which may be because of the relatively tumor cells acidic environment in which the release of metal ions from metal nanoparticles occurs more rapidly. In addition, the metal toxic effects on cancer cells could be because of the interaction of metal atoms with functional groups of proteins, as well as phosphate and nitrogen bases of the DNA molecule (Davis et al., 2008; Matsumura et al., 2004; Nie et al., 2007; Namvar et al., 2014; Sankar et al., 2014). Another effect of metal nanoparticles on cancer cells is growth inhibition by inhibiting cell division, which results in their interaction with the structure and organization of the actin cytoskeleton. Evidence and results of several studies indicate the induction of apoptosis because of the use of metal nanoparticles in cancer cells (Zhang et al., 2014; Gao et al., 2002; Davis et al., 2008; Matsumura et al., 2004; Nie et al., 2007; Namvar et al., 2014). This seems to happen through the production of reactive oxygen; thus, the active surfaces of metal nanoparticles yield free radicals by releasing metal ions, which these free radicals destroy DNA, especially the mitochondrial membrane, causing the release of cytochrome c and the induction of apoptosis from the mitochondrial pathway (Zhang et al., 2014; Gao et al., 2002; Davis et al., 2008; Matsumura et al., 2004; Nie et al., 2007; Namvar et al., 2014; Sankar et al., 2014).
3.1 Characterization of the prepared Ag NPs@gelatin bionanocomposite
The formation of Ag NPs in gelatin was established with UV–Visible spectrophotometer. The presence of specific peaks 440 nm confirmed the formation of Ag NPs as shown in Fig. 1. The broad peaks formed by Ag NPs in hydrogel is an indication of the reducing action of gelatin to silver nanoparticles. The successful biogenic synthesis of Ag NPs was primarily confirmed by a visual color change from watery yellow to brown color.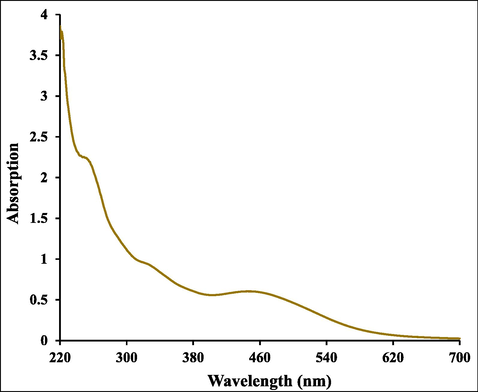
UV–visible absorption spectra of Ag NPs@gelatin bionanocomposite.
The formation mechanism of Ag NPs@gelatin bionanocomposite and the attachment of gelatin can be explained on the basis of their FT-IR spectra (Fig. 2). The spectra of gelatin (Fig. 2a) showed vibration bands at 3420 cm−1 (N—H stretch coupled with hydrogen bonding), 3088 cm−1 (alkenyl C—H stretch), 2962 cm−1 (CH2 asymmetrical stretching), 1629 cm−1 (C⚌O stretch/HB coupled with COO—), 1530 cm−1 (N—H bend coupled with CN stretch), 1442 cm−1 (CH2 bend), 1241 cm−1 (NH bend), and 1078 cm−1 (C—O stretch) (Thi and Nguyen, 2016). Fig. 2b, showing the vibrational peaks of the Ag NPs@gelatin bionanocomposite, looks very similar to that of gelatin. This implies that gelatin is successfully encapsulated over the Ag NPs and there occurs no change in the internal core. Nevertheless, there occurs a slight relocation of the peaks in the spectrum of final material, which can be anticipated due to strong bond formation of Ag NPs with gelatin functions.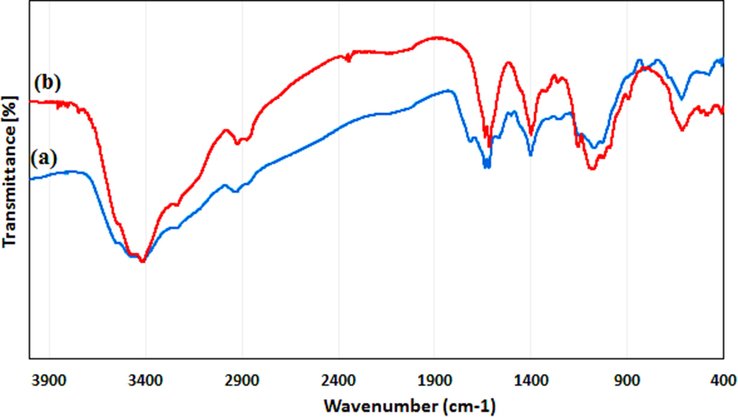
FT-IR spectrum of (a) gelatin and (b) Ag NPs@gelatin bionanocomposite.
The surface morphology of the Ag NPs@gelatin bionanocomposite was investigated by the FE-SEM study, as shown in Fig. 3. It shown the quasi-spherical shaped nanoparticles of mean diameter of nano. For further structural inherence of the nanocomposite, TEM analysis was carried out (Fig. 4). The TEM measurements revealed the presence of spherically shaped nanoparticles in the matrix (Fig. 4). The mean diameter of Ag NPs was in the range of ∼5–10 nm. In the image, the nanoparticles were well dispersed without any aggregation.
FE-SEM image of the Ag NPs@gelatin bionanocomposite.

TEM images of the Ag NPs@gelatin bionanocomposite.
The prepared Ag NPs@gelatin bionanocomposite was validated from EDX analysis, as presented in Fig. 5. The profile shows a major and distinct signal of Ag appeared at 2.95 keV. However, some other peaks of C, N and O as constitutional elements are also observed. They can be corresponded to the gelatin hydrogel, being associated with the Ag NPs surfaces. On X-ray scanning of a section of SEM image, the compositional map can be identified. It shows a uniform distribution of all the elements over the composite surface which is very significant for a bionanocomposite, particularly for the active species (Fig. 6).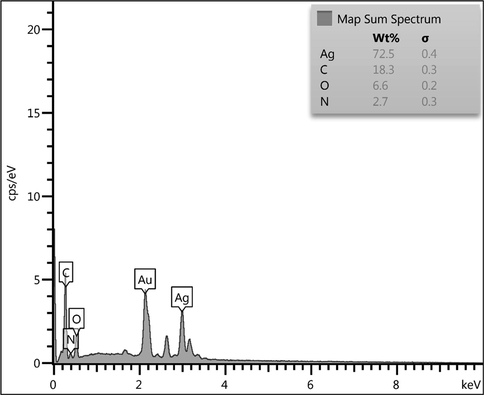
EDX spectrum of the Ag NPs@gelatin bionanocomposite.

Elemental mapping of Ag NPs@gelatin bionanocomposite.
The nature of crystalline phases of Ag NPs@gelatin bionanocomposite was investigated by XRD analysis, being shown in Fig. 7. While the initial phase in the 2θ region up to 15° was attributed to gelatin hydrogel and non-crystalline. Additionally, the diffraction peaks at 2θ = 38.1°, 44.3°, 64.6° and 77.2° confirms the Ag crystalline phases. These corresponds to the diffraction on fcc silver (1 1 1), (2 0 0), (2 2 0) and (3 1 1) planes (JCPDS No. 87-0717).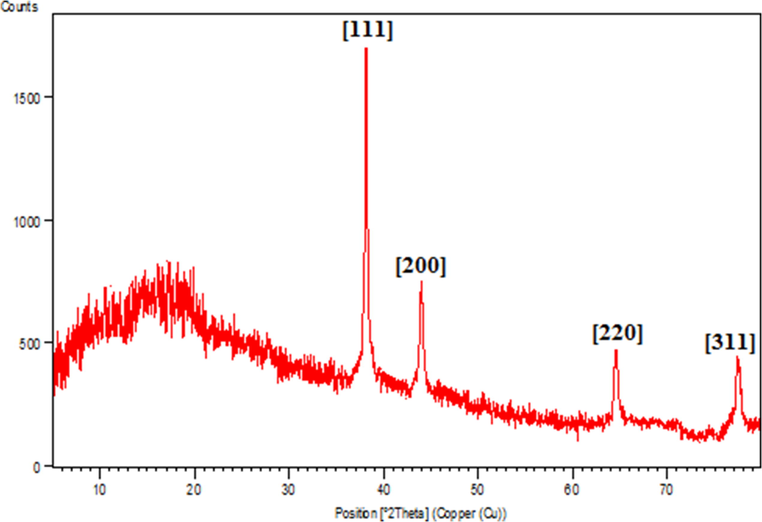
XRD pattern of the Ag NPs@gelatin bionanocomposite.
3.2 Antioxidant properties of Ag NPs@gelatin bionanocomposite
In this study, we assessed the antioxidant properties of Ag NPs@gelatin bionanocomposite by using the DPPH test as a common free radical.
The scavenging capacity of Ag NPs@gelatin bionanocomposite and BHT at different concentrations expressed as percentage inhibition has been indicated in Fig. 8. In the antioxidant test, the IC50 of Ag NPs@gelatin bionanocomposite and BHT against DPPH free radicals were 203 and 250 µg/mL, respectively (Table 1 and Fig. 8).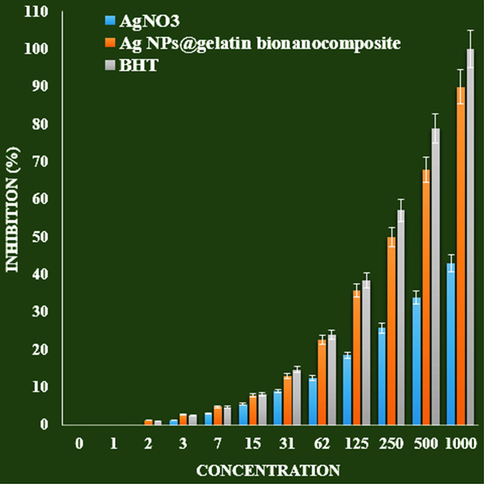
The antioxidant properties of AgNO3, Ag NPs@gelatin bionanocomposite, and BHT against DPPH.
AgNO3 (µg/mL)
Ag NPs@gelatin bionanocomposite (µg/mL)
BHT (µg/mL)
IC50 against DPPH
–
250 ± 0
203 ± 0
3.3 Anti-liver carcinoma effects analysis of Ag NPs@gelatin bionanocomposite
Unique properties of nanoparticles such as particle size, high surface-to-volume ratio, their targeting ability, loading ability of water-insoluble drugs and controlled release, and responsiveness to the stimulant drug can make them a good candidate for cancer treatment (Sankar et al., 2014; Beheshtkhoo et al., 2018; Radini et al., 2018; Katata-Seru et al., 2018; Sangami and Manu, 2017; Beyene et al., 2017; Chen and Schluesener, 2008; Alexander, 2009; Bhattacharya, 2011; Huang et al., 2017; Conde et al., 2012; Rai et al., 2014). When the nanoparticles are injected into the bloodstream, they need to pass through the walls of the arteries to reach the target site and then release the drug. Unlike small molecules, nanoparticles, because of their relatively large size, cannot pass through the tight connections between the endothelial cells of healthy blood vessels. Tumor blood vessels, due to their leaky walls, allow nanoparticles to pass well. Targeted drug delivery to the tumor by nanoparticles is done in both passive and active targeting (Rai et al., 2014; Jo et al., 2015; Riehemann et al., 2009; Bhattacharyya et al., 2011; Day et al., 2009; Pelaz et al., 2015). In passive targeting, tumor tissue properties are used for drug delivery. In tumor tissue, due to rapid cell growth, rapid angiogenesis occurs, but the distance between vascular endothelial cells is too large, causing macromolecules and nanoparticles to leak from blood vessels into tumor tissue (Pelaz et al., 2015; Sperling and Parak, 2010; Sau et al., 2010). On the other hand, the lymphatic system of tumor tissue is not complete and is not able to collect nanoparticles and insert them into the bloodstream; As a result, nanoparticles accumulate in the tumor tissue and release their drug. For passive targeting, the nanoparticle size should usually be below 200 nm. Active targeting of the tumor is achieved by binding a ligand such as antibodies, peptides, aptamers and some small molecules such as folic acid to nanoparticles (Riehemann et al., 2009; Bhattacharyya et al., 2011; Day et al., 2009; Pelaz et al., 2015; Sperling and Parak, 2010; Sau et al., 2010).
In this research, the treated cells with different concentrations of the present Ag NPs@gelatin bionanocomposite were assessed by MTT assay for 48 h about the cytotoxicity properties on normal (HUVEC) and human liver malignancy cell lines i.e. pleomorphic hepatocellular carcinoma (SNU-387), morris hepatoma (McA-RH7777), and novikoff hepatoma (N1-S1 Fudr) (Fig. 9,10).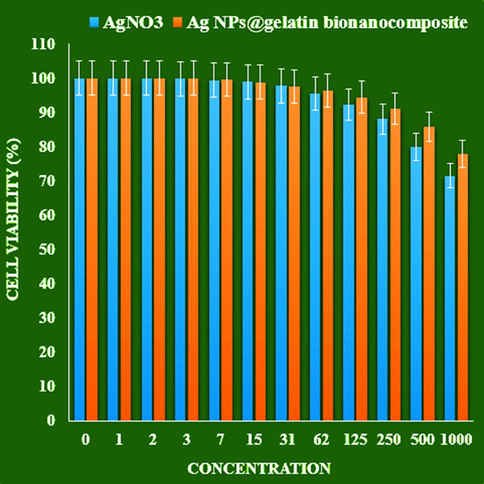
The cytotoxicity effects of AgNO3 and Ag NPs@gelatin bionanocomposite against normal cell line.
The absorbance rate was evaluated at 570 nm, which represented viability on normal cell line (HUVEC) even up to 1000 μg/mL for Ag NPs@gelatin bionanocomposite (Table 2 and Fig. 9).
AgNO3 (µg/mL)
Ag NPs@gelatin bionanocomposite (µg/mL)
IC50 against HUVEC
–
–
IC50 against pleomorphic hepatocellular carcinoma (SNU-387)
–
192 ± 0
IC50 against morris hepatoma (McA-RH7777)
–
144 ± 0
IC50 against novikoff hepatoma (N1-S1 Fudr)
–
228 ± 0
The viability of malignant liver cell line reduced dose-dependently in the presence of Ag NPs@gelatin bionanocomposite.
The IC50 of Ag NPs@gelatin bionanocomposite were 192, 144, and 228 µg/mL against pleomorphic hepatocellular carcinoma (SNU-387), morris hepatoma (McA-RH7777), and novikoff hepatoma (N1-S1 Fudr) cell lines, respectively (Table 2 and Fig. 10).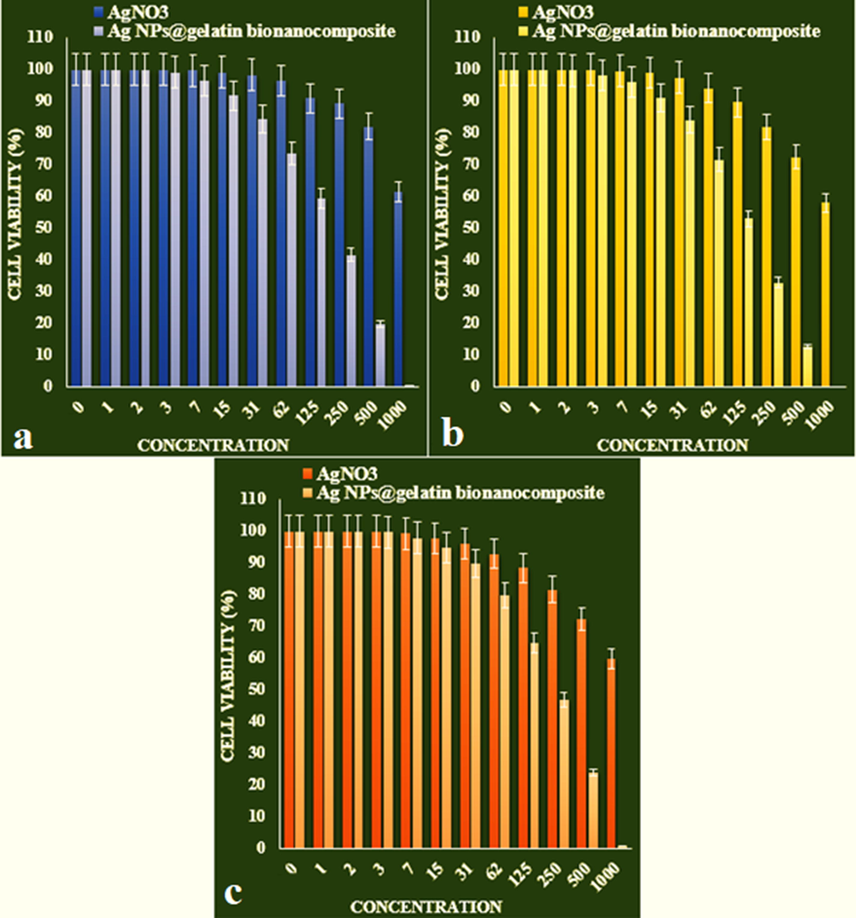
The anti-human liver cancer properties of AgNO3 and Ag NPs@gelatin bionanocomposite against liver cancer cell lines (a: pleomorphic hepatocellular carcinoma (SNU-387), b: morris hepatoma (McA-RH7777), c: novikoff hepatoma (N1-S1 Fudr)).
4 Conclusion
In this study, highly stable silver nanoparticles were successfully introduced into gelatin polymers using a green and simple procedure avoiding any hazardous chemicals. Then structural characterization of Ag NPs@gelatin bionanocomposite was performed by UV–Vis, FT-IR, TEM, SEM, EDS and XRD analysis. It was found that silver nanoparticles uniformly dispersed over gelatin and the average diameter of the particles were in the range of 5–10 nm. The viability of malignant liver cell lines reduced dose-dependently in the presence of Ag NPs@gelatin bionanocomposite. The IC50 of Ag NPs@gelatin bionanocomposite were 192, 144, and 228 and 131 µg/mL against pleomorphic hepatocellular carcinoma (SNU-387), morris hepatoma (McA-RH7777), and novikoff hepatoma (N1-S1 Fudr) cell lines, respectively. The Ag NPs@gelatin bionanocomposite showed the best antioxidant activities against DPPH. It seems that the anti-human liver cancer effect of recent nanoparticles is due to their antioxidant effects.
Acknowledgment
The authors would like to thank the Deanship of Scientific Research at King Khalid University, Abha, KSA for funding this work under Grant number (R.G.P.2/35/43). The authors extend their appreciation to the Taif University Researchers Supporting Project number (TURSP-2020/38), Taif University, Taif, Saudi Arabia.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Molecules. 2011;16:7237-7248.
- Ligand-targeted therapeutics in anticancer therapy. Nat. Rev. Cancer. 2002;2(10):750-763.
- [Google Scholar]
- Green synthesis of iron oxide nanoparticles by aqueous leaf extract of Daphne mezereum as a novel dye removing material. Appl. Phys. A. 2018;124:363-369.
- [Google Scholar]
- Synthesis paradigm and applications of silver nanoparticles (Ag NPs), a review. Sustain. Mater. Technol.. 2017;13:18-23.
- [Google Scholar]
- Toxicity testing in the 21 century: Defining new risk assessment approaches based on perturbation of intracellular toxicity pathways. PLoS One. 2011;6:e20887
- [Google Scholar]
- The potential risks of nanomaterials: a review carried out for ECETOC. Particle Fibre Toxicol.. 2006;3(1):11.
- [Google Scholar]
- Active targeting schemes for nanoparticle systems in cancer therapeutics. Adv. Drug Del. Rev.. 2008;60(15):1615-1626.
- [Google Scholar]
- Pharmacological potential of cerium oxide nanoparticles. Nanoscale. 2011;3(4):1411-1420.
- [Google Scholar]
- Nanoparticle therapeutics: an emerging treatment modality for cancer. Nat. Rev. Drug Discov.. 2008;7(9):771-782.
- [Google Scholar]
- Drug delivery and nanoparticles: applications and hazards. Int. J. Nanomed.. 2008;3(2):133-149.
- [Google Scholar]
- Diacyllipid-polymer micelles as nanocarriers for poorly soluble anticancer drugs. Nano Lett.. 2002;2(9):979-982.
- [Google Scholar]
- Liposome encapsulated of temozolomide for the treatment of glioma tumor: preparation, characterization and evaluation. Drug Discov. Ther.. 2015;9(3):205-212.
- [Google Scholar]
- Prolongation of the shelf-life of perishable food products using biodegradable films and coatings. LWT-Food Sci. Technol.. 1996;29(1–2):10-17.
- [Google Scholar]
- Current applications and future prospects of nanomaterials in tumor therapy. Int. J. Nanomed.. 2017;12:1815-1825.
- [Google Scholar]
- siRNA conjugated nanoparticles-a next generation strategy to treat lung cancer. Int. J. Mol. Sci.. 2019;20(23):6088.
- [Google Scholar]
- Size, surface charge, and shape determine therapeutic effects of nanoparticles on brain and retinal diseases. Nanomed Nanotechnol. Biol. Med.. 2015;11:1603-1611.
- [Google Scholar]
- Green synthesis of iron nanoparticles using Moringa oleifera extracts and their applications: Removal of nitrate from water and antibacterial activity against Escherichia coli. J. Mol. Liq.. 2018;256:296-304.
- [Google Scholar]
- J. Serb. Chem. Soc.. 2013;78:469-476.
- Recent progress in doxorubicin nano-drug delivery systems for reserving multidrug resisitance. Anti-infection Pharm.. 2014;11(3):177-181.
- [Google Scholar]
- Synthesis of a reusable composite of graphene and silver nanoparticles for catalytic reduction of 4- nitrophenol and performance as anti-colorectal carcinoma. J. Mater. Res. Technol.. 2021;12:1832-1843.
- [Google Scholar]
- Mechanisms of silver nanoparticle-induced toxicity and important role of autophagy. Nanotoxicology. 2016;10:1021-1040.
- [Google Scholar]
- Phase I clinical trial and pharmacokinetic evaluation of NK911, a micelleencapsulated doxorubicin. Br. J. Cancer. 2004;91:1775-1781.
- [Google Scholar]
- Biomacromolecules. 2005;6:975-987.
- Oxygen and aroma barrier properties of edible films: a review. Trends Food Sci. Tech.. 1997;8(7):228-237.
- [Google Scholar]
- Transdermal delivery of vancomycin hydrochloride using combination of nano-ethosomes and iontophoresis: in vitro and in vivo study. Drug Deliv.. 2016;23(5):1558-1564.
- [Google Scholar]
- Cytotoxic effect of magnetic iron oxide nanoparticles synthesized via seaweed aqueous extract. Int. J. Nanomed.. 2014;19:2479-2488.
- [Google Scholar]
- Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol.. 2018;16:71.
- [Google Scholar]
- Surface functionalization of nanoparticles with polyethylene glycol: Effects on protein adsorption and cellular uptake. ACS Nano. 2015;9:6996-7008.
- [Google Scholar]
- Deshpande, Current trends in the use of liposomes for tumor targeting. Nanomedicine. 2013;8(9):1509-1528.
- [Google Scholar]
- Biosynthesis of iron nanoparticles using Trigonella foenum-graecum seed extract for photocatalytic methyl orange dye degradation and antibacterial applications. J. Photochem. Photobiol. B. 2018;183:154-163.
- [Google Scholar]
- Broad-spectrum bioactivities of silver nanoparticles: The emerging trends and future prospects. Appl. Microbiol. Biotechnol.. 2014;98:1951-1961.
- [Google Scholar]
- Synthesis of Green Iron Nanoparticles using Laterite and their application as a Fenton-like catalyst for the degradation of herbicide Ametryn in water. Environ. Technol. Innov.. 2017;8:150-163.
- [Google Scholar]
- Anticancer activity of Ficus religiosa engineered copper oxide nanoparticles. Mat. Sci. Eng. C. 2014;44:234-239.
- [Google Scholar]
- Properties and applications of colloidal nonspherical noble metal nanoparticles. Adv. Mater.. 2010;22:1805-1825.
- [Google Scholar]
- Epidemiology of hepatocellular carcinoma (HCC) in hemophilia. Criti. Rev. Oncol. Hematol.. 2016 Mar;1(99):129-133.
- [Google Scholar]
- Surface modification, functionalization and bioconjugation of colloidal inorganic nanoparticles. Philos. Trans. Ser. A Math. Phys. Eng. Sci.. 2010;368:1333-1383.
- [Google Scholar]
- Vascular distribution of nanomaterials. Wiley Interdiscipl. Rev. Nanomed. Nanobiotechnol.. 2014;6(4):338-348.
- [Google Scholar]
- Green Process Synth. 2016;5:467-472.
- Targeted pharmaceutical nanocarriers for cancer therapy and imaging. AAPS J.. 2007;9:E128-E147.
- [Google Scholar]
- Charged microcapsules for controlled release of hydrophobic actives Part II: surface modification by LbL adsorption and lipid bilayer formation on properly anchored dispersant layers. J. Colloid Interface Sci.. 2013;409:8-17.
- [Google Scholar]
- Applications of nanocarriers with tumor molecular targeted in chemotherapy. Chemistry.. 2012;75(7):621-627.
- [Google Scholar]
- Liposome based delivery systems in pancreatic cancer treatment: from bench to bedside. Cancer Treat. Rev.. 2011;37(8):633-642.
- [Google Scholar]
- The progress of silver nanoparticles in the antibacterial mechanism, clinical application and cytotoxicity. Mol. Biol. Rep.. 2012;39:9193-9201.
- [CrossRef] [Google Scholar]
- Polymeric micelles: Nanocarriers for cancer-targeted drug delivery. AAPS PharmSciTech. 2014;15:862-871.
- [Google Scholar]







