Translate this page into:
Production of Monoclonal antibodies to membrane components of human colorectal cancer HCT-116 cell line for diagnostic purposes
⁎Corresponding author. Maryam.o@yu.edu.jo (Mariam M. Al-Omari)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Colorectal cancer (CRC) is the second leading cause of cancer-related death worldwide. The survival rate of people diagnosed in the early stages of disease is 92 % whereas; less than 11 % of patients survive if reached stage IV. Moreover, only 24 % of cases are diagnosed during stage I. Thus, the early diagnosis of the CRC will save many lives, reduce suffering of patients. Biomarkers that are in use for the diagnosis of CRC such as Carcinoembryonic Antigen (CEA), Carbohydrate Antigen (CA 19–9) suffer from low sensitivity, expressed in other neoplasms, and not expressed in early stages of cancer. This study aims to find a highly sensitive and specific biomarker for early detection of CRC. Hybridoma clones were established by fusing spleen cells from mice hyperimmunized with human cell membrane homogenate of HCT116 with murine myeloma cells X63Ag8. One clone was selected after fusion and designated as D2C5. Isotyping of secretory antibody revealed an IgG1 kappa light chain and was reactive to a protein with molecular weight about 55 kDa using immunoblot. In situ reactivity of the D2C5 showed homogenous and cytoplasmic pattern staining in stage IV and III, discrete and heterogeneous pattern was seen in stage II samples. Moreover, nuclear staining pattern was seen in one slide of stage II samples. In addition, mucinous type was detected. Low peripheral staining was observed in normal colon tissue. These observations suggest the localization of the target protein in the cytoplasm of epithelial cells with high expression in more advanced cases, suggesting a cytokeratin pattern of staining.
Keywords
Monoclonal antibody mAb
Colorectal cancer CRC
Whole cell immunization
Cytokeratin CK
Immunohistochemistry IHC
Membrane protein homogenate MPH
1 Introduction
For over a decade, colorectal cancer (CRC) is the second most common cancer in men and women and the second cause of death (Keum and Giovannucci, 2019). According to Globocan 2020 Jordan, CRC ranked second among cancers reported in both males and females accounting (11.1 %, 10.8 %), respectively. In the same year, CRC ranked third for men and women in distribution of deaths by cancer site. The present diagnostic methods that include colonoscopy, flexible sigmoidoscopy, and stool-based tests have successfully reduced the number of undiagnosed cases; however, a significant number of individuals are diagnosed in late stages (Kolligs, 2016).The early diagnosis of CRC is a big challenge for the health institutions to reduce the morbidity and mortality rates of the disease. The overall survival rate when cancer is limited to colon is 95 % and 82 % for stages one and two, respectively. However, the survival rate is reduced to 61 % with regional spread to lymph nodes (stage three) and only 8 % for patients with distal metastasis (stage four) (Parkin et al., 2005).
The new way of diagnosis and treatment of cancer is the detection biomarkers. Biomarkers defined as substances that appear in association with carcinogenesis and may be specific for each stage of the disease. Cancer biomarkers could refer to DNA, RNA, miRNA, epigenetic changes or antibodies (Lech et al., 2016). Great success has been achieved in the development of diagnostic and therapeutic methods; however, a need for non-invasive, sensitive and specific biomarkers is still a priority. Biomarkers are considered as key factors in the detection and treatment of patients with CRC. Thus, new biomarkers for testing either alone or in combination with preexisting once may aid in the recognition of early stages of the disease (Lech et al., 2016). Carcinoembryonic antigen (CEA) was the first and still remains the most efficacious useful tumor marker in CRC diagnosis (Duffy et al., 2014). However, there are limitations for the use of CEA in CRC diagnosis that include its elevation in other neoplasms and inflammation, rarely identified in stage I CRC and its poor differentiation between benign versus malignant polyps (Duffy et al., 2014). Cancer antigen 19–9 (CA 19–9) is the second most popular biomarker in CRC diagnosis (Lumachi et al., 2012). It is much inferior to CEA in diagnosis but elevated levels of CA19- 9are markers of poor prognosis of the disease (Levy et al., 2008). Other carbohydrate biomarkers such as CA 72–4 showed improved specificity reaching to 89 % − 95 % in screening of patients with CRC but its lower sensitivity reaching to 9–31 % resulted in its limited use (Lumachi et al., 2012). Moreover, CK7-/CK20 + and CK7 + CK20 + phenotype was found to be expressed in 64 % and 20 % of colorectal adenocarcinoma, respectively (Bayrak et al., 2012). Taken together, none of the commercially available antigen biomarkers has the capacity to identify early stages of CRC with high sensitivity and specificity which urges the need for a comprehensive search for a reliable biomarker that helps in the early diagnosis of CRC.
The most efficient way of cancer diagnosis and treatment is the production of monoclonal antibodies (mAbs) that can be produced by hybridoma technique. Thus, in the current study, we aimed to characterize and identify new CRC antigens using monoclonal antibodies produced by hybridoma technology. The production of such mAb may improve the diagnostic capabilities of CRC mainly in early stages of the disease. Moreover, one efficient mAb may substitute the use of panels of mAbs for CRC diagnosis and thus reduce the cost of diagnosis.
2 Material and methods
2.1 Material and chemicals
Human colonic cancer cell line (HCT-116) was purchased from ATCC (USA), HRP labeled goat anti-mouse total IgG was purchased from Abcam (UK), Hypoxanthine, Aminopterin and Thymidine supplement (HAT) and HT were purchased from Euro clone, (E.U). Polyethylene glycol 1450 solution (PEG 1450) was purchased from Sigma-Aldrich (Shanghai, China). DMEM media, McCoy’s media and fetal bovine serum (FBS) were purchased from Euro clone (E.U). Balb/c mice were from the Animal House at Yarmouk University (Irbid-Jordan). All animal experiments followed the guidelines established by Institutional Animal Care and Use Committee (IACUC) (No.16/3/3/310 Jordan University of Science and Technology).
2.2 Cell culture and antigens preparation
Human colorectal carcinoma cell line (HCT-116) was cultured as monolayer in complete McCoy’s media supplemented with 10 % FBS, 2.5 μg/ml Amphotericin B and 0.5 mg/ml penicillin–streptomycin at 37 °C and humidified 5 % CO2. The cells medium was replaced regularly every 2–3 days.
Membrane Protein Homogenate of HCT-116 (HCT-MPH) was extracted and purified using Mem-PER™ Plus Membrane Protein Extraction buffer (Thermo Fisher, USA). HCT-116 cells were trypsinized as mentioned previously. Mem-PER™ Plus protein extraction buffer and Pierce protease inhibitor (Thermo Fisher, USA) were added to cells and incubated for 10 min with gentle shaking and centrifuged for 15 min at 15000g at 4℃. Bradford micro-protein assay was done according to (Bradford, 1976) with modification to adapt microassay protocol.
2.3 Mouse immunization
Three Female BALB/c mice weighing between 23 and 28 g were used for immunization and 2 mice were injected with Freunds adjuvant alone and used as negative controls. HCT-MPH was emulsified with Freund’s complete adjuvant (1:1) at the first injection and in incomplete adjuvant at subsequent injections. Each mouse was injected intraperitoneally (ip) with 0.5 ml containing 200 µg of antigen for 3 boosts at 14 days intervals.
2.4 Measurement of mouse anti HCT-MPH polyclonal antibodies
The detection of anti HCT-MPH in mouse sera and hybridoma supernatants were done by ELISA. Microtiter plates were coated with 10 μg/mL of HCT-MPH diluted in carbonate buffer (pH 9.6) and incubated at 4 °C overnight. Next day, the plates were washed 3 times with 1X PBS containing 0.05 %Tween 20, and then blocked with 300 μl/well 3 % chicken egg albumin. Hundred µl of immunized and control mice sera (diluted 1:100) were added to wells and incubated at 37 °C for 1 h. For hybridoma 100 µl undiluted culture supernatants were used. After washing, HRP-labeled goat anti-mouse IgG (1:2000) was added and incubated at 37 °C for 1 h. Plates were washed and 100 µl freshly prepared substrate solution (high sensitive TMB solution, Abcam, UK) was added into each well and incubated at 37 °C for 10 min. Finally, the reaction was stopped by 2 M H2SO4 (100 μl per well), and measured by micro-plate reader (Multiskan Go, Thermo, USA) at 450 nm wavelength. Mouse with higher titer was used in hybridoma.
2.5 Cell fusion and hybridoma screening
Mouse that showed high anti HCT-MPH antibody titer was selected for hybridoma generation. Splenocytes were isolated from the immunized mouse and mixed with murine X63Ag8 myeloma cells at a ratio of 2:1 with a gradual addition of PEG-1450. Cells were distributed into 12-well micro-titer plates. These cells were cultured in DMEM with 10 % FBS/HAT medium at 37 °C and 5 % CO2 and the media was changed upon need. Clone generation was observed and after confluent cell growth the mixed clones were injected into the peritoneal cavity of primed mouse to remove any contaminants in the culture. Cells were recollected from mouse peritoneal cavity under aseptic conditions and cultured for single clone generation. Following that, supernatant was tested for HCT-MPH specific antibodies by ELISA. Cells in positive clones were subcloned in 96 well plate by limiting dilution for single cell generation.
2.6 Characterization of positive hybridoma against HCT-116 membrane protein
2.6.1 Determination of mAb isotypes
The monoclonal antibody isotypes (IgG1, IgG2a, IgG2b, IgG3 and IgM) was determined by lateral flow isotyping kit according to manufacturer instructions (Abcam, Uk).
2.6.2 Determination of specificity of mAb by IHC
The specificity of mAb to CRC was determined based on its reactivity to different stages of CRC as well as normal colon tissue. Human CRC tissue slides in formalin were obtained from the King Abdullah Hospital Irbid/Jordan. Slides were deparaffinized in xylene, rehydrated in graded alcohol and treated with boiling in 10 mmol/L citrate buffer (pH 6.0) for 1 min using microwave. After that; slides were cooled to room temperature, washed 3 times with PBS, the subsequent steps were done according to manufacturing protocol (Abcam, UK). Then, the section was rehydrated and stained with hematoxylin for 3 min. The slides were ethyl alcohol dehydrated and treated with xylene for 3 min. Finally, the sections were mounted using Canada Balsam and observed under microscope (Nikon 400, Tokyo, Japan).
2.6.3 Identification of antibody-protein reactivity by westeren blot
The reactivity of mAb CRC antigen was determined by western blot. HCT-MPH was electrophoresed in 12 % SDS-PAGE gel, transferred into the nitrocellulose membrane, and blocked with 3 % bovine serum albumin. The membrane was incubated for 1 hr with the culture supernatant, positive mouse serum as positive control and DMEM as negative control. HRP-conjugated anti mouse IgG was added to strips and incubated for 1 hr at RT. Ready to use TMB substrate was poured on washed strips until color development.
3 Result
3.1 Generation of hybridoma from immunized mice
Several fusion trials were done and antibody secreting clones were detected in hybridoma generated from mouse immunized with HCT-MPH. The clones started to appear after the fifth day of fusion (Fig. 1A) and reached high rate of growth at day 10 (Fig. 1B).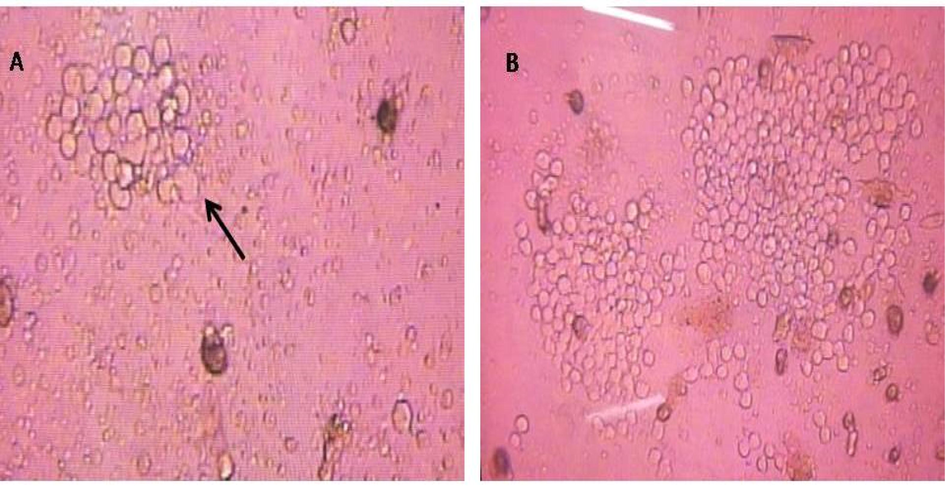
Hybridoma culture at day 5 following fusion (A) showing small cell clone (arrow) and at day 10 showing 2 growing distinct clones (B) (Magnification 40X and 10X, respectively).
3.2 Screening of positive hybridoma clones
Hybridoma clones showed high polyclonal anti-HCT-MPH antibody titers with multiple subclasses IgG1, IgG2a, IgG2b and IgG3. After limiting dilution protocol, one stable clone (D2C5) showed positive results when tested against HCT-MPH. ELISA and lateral flow testing of cultured supernatant from clone (D2C5) gave positive IgG1 kappa light chain isotype result on antibody isotyping strip (Fig. 2).
D2C5clone culture supernatant isotyping using commercial kit (AbCam, UK). The presence of red line below the script indicates positive reaction.
3.3 Reactivity of D2C5 monoclonal anti HCT-MPH to human colon cancer tissue
The reactivity of D2C5 antibody secretions were tested with IHC assay. Different stages of human colonic tissue samples were detected (Table 1). D2C5 showed specific homogeneous reactivity with cytoplasmic cancer tissue in all stages. At stage IV; D2C5 showed strong cytoplasmic reactivity which was stronger than stage III and stage II (Figs. 3, 4 and 5). At stage III; D2C5 showed specific moderate heterogeneous cytoplasmic reactivity (Fig. 4). In some tissue sections nuclear staining was observed (Fig. 4D). Moreover, weak heterogeneous cytoplasmic reactivity was characteristic for stage II sections (Fig. 3). It is noteworthy that D2C5 showed minimal reactivity to normal human colon sections and was localized to the outer most epithelial layer of colon (Fig. 6).
Stage
Grade
Treatment
Intensity
Staining pattern
Pattern
2
G2
D2C5
++
Heterogeneous
Cytoplasmic
2
G2
Media
–
–
–
2
G2
D2C5
+++
Heterogeneous
Cytoplasmic and nuclear
2
G2
Media
–
–
–
2
Mucinous CA
D2C5
+
Homogeneous
Cytoplasmic
2
Mucinous CA
Media
++
–
–
3
G2
D2C5
++
Homogeneous
Cytoplasmic
3
G2
Media
–
–
–
3
G2
D2C5
++
Heterogeneous
Cytoplasmic
3
G2
D2C5
+++
Heterogeneous
Cytoplasmic
4
G1
D2C5
++
Homogeneous
Cytoplasmic
4
G1
Media
–
–
–
4
G2
D2C5
++++
Homogeneous
Cytoplasmic
4
G3
D2C5
++++
Homogeneous
Cytoplasmic
Normal
–
D2C5
+/−
Homogeneous
Surface of outer membrane
Normal
–
Media
–
–
–
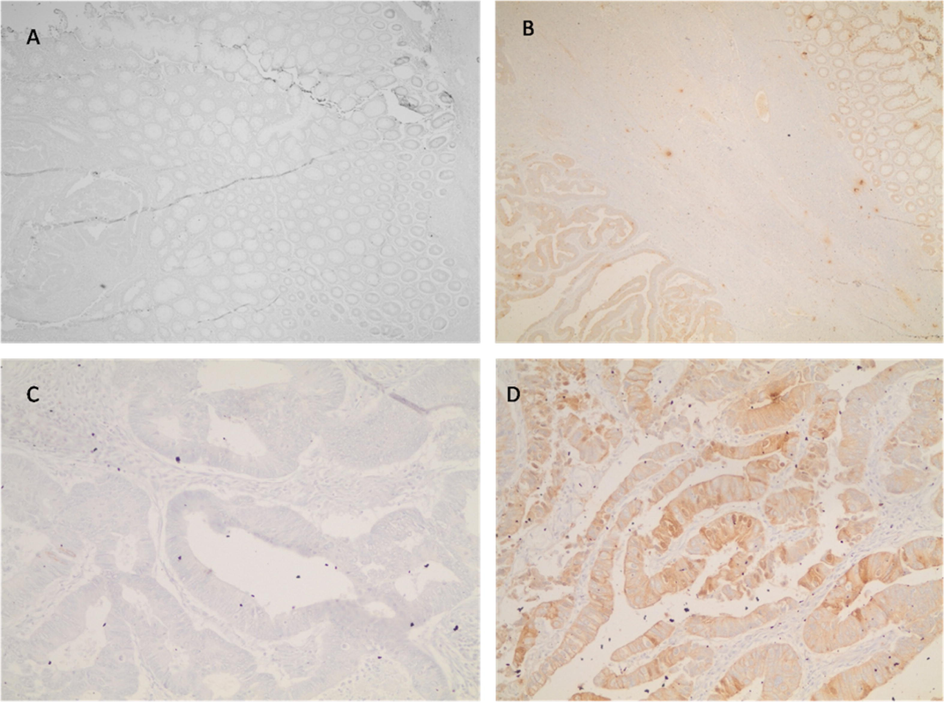
Immunohistochemical staining of colon cancer tissue section (stage II). In A and C DMEM complete medium was added as negative control (Mag. 10X and 40X, respectively). In B and D, D2C5 culture supernatant (Mag. 10X and 40X respectively). Heterogeneous and discrete staining are evident.
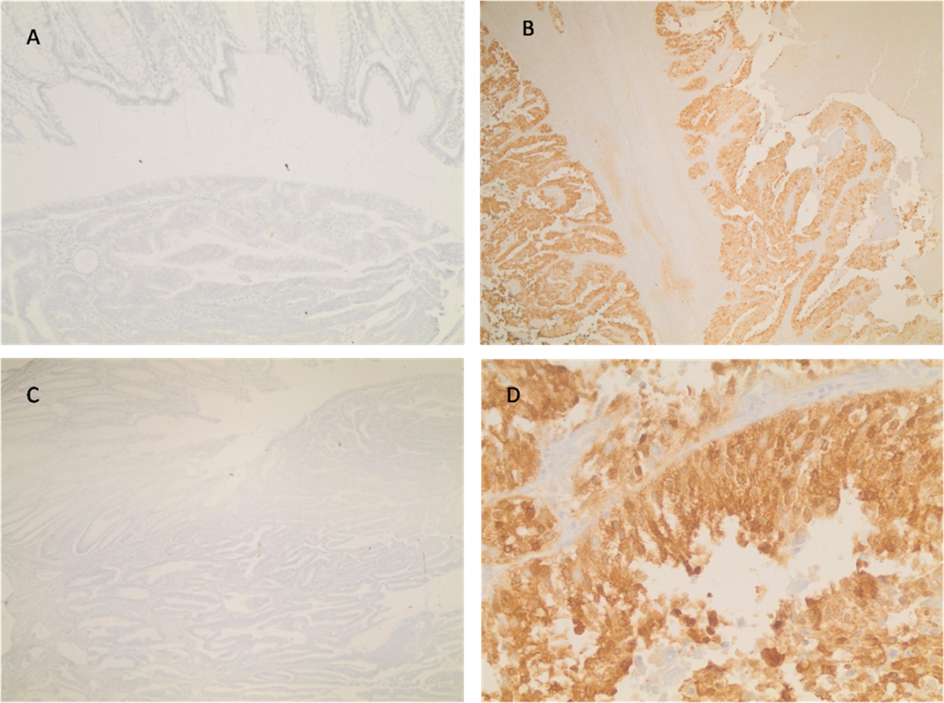
Immunohistochemical staining of colon cancer tissue section (stage III). In A and C, DMEM complete medium as negative control (Mag. 10X). In B and D, D2C5 culture supernatant (Mag. 10X and 40X respectively). Homogenous cytoplasmic and nuclear staining are evident.
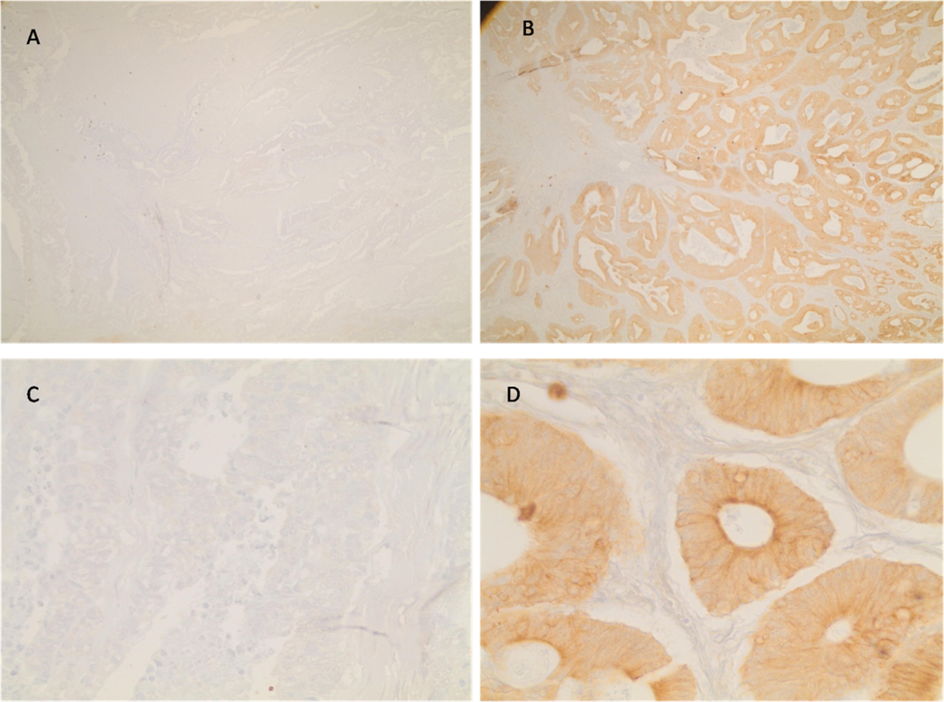
Immunohistochemical staining of colon cancer tissue section (stage IV). In A and C tissue sections were incubated with DMEM complete medium as negative control (Mag. 10X). In B and D sections were incubated with D2C5 culture supernatant (Mag. 10X and 40X, respectively). Homogenous cytoplasmic staining is evident.
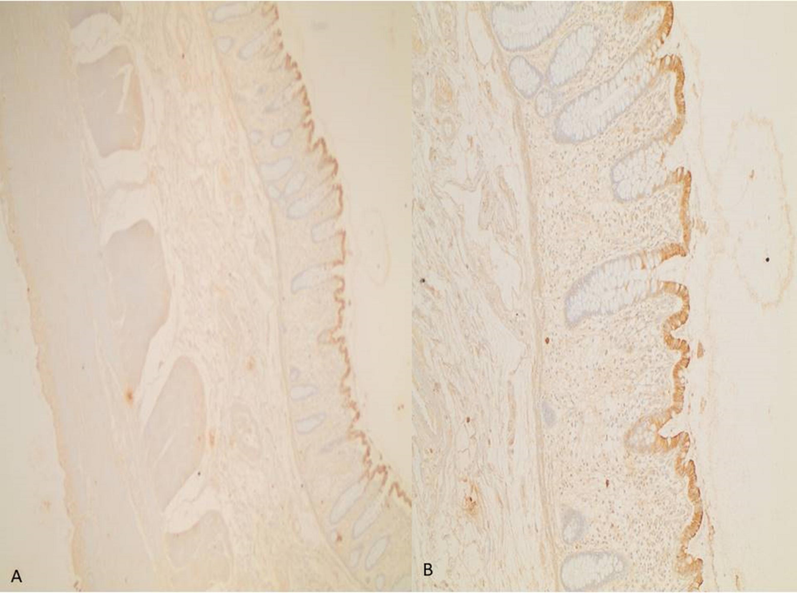
Immunoreactivity of D2C5 clone culture supernatant to normal colon tissue sections (Mag. 10X and 40X for A and B, respectively).
3.4 Identification of protein reactive D2C5 clone
When D2C5 culture supernatants were blotted against HCT-MPH, single band was identified with a molecular weight of about 55 kDa (Fig. 7A). This suggests a member of cytokeratin protein. As evident from the SDS-PAGE protein profile the reactive protein present both in mouse colon cancer tissue and HCT116 cell line homogenate (Fig. 7B).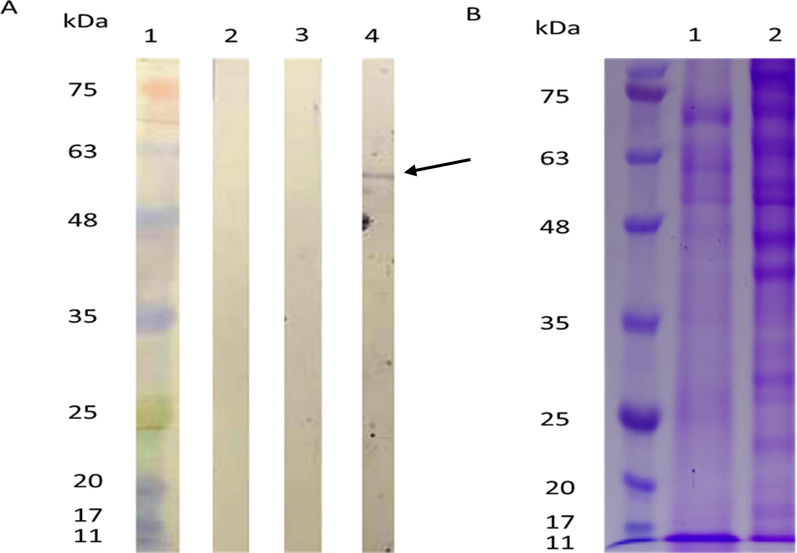
A. Immunoblot results for D2C5 clone culture supernatant to HCT-MPH: protein marker, 2: PBS control, 3: complete medium, 4: D2C5 clone, arrow points at the D2C5 reactive protein. B. SDS-PAGE of protein extracts from mouse colon cancer (1) and HCT-MPH.
4 Discussion
In this study, we generated and characterized a novel monoclonal antibody (mAB); D2C5, using human cell line membrane protein extract which is specific against colorectal cancer antigen. We used hybridoma technology to generate D2C5 mAB. These hybridoma cells were formed via fusion between HCT-MPH and myeloma cell line (X63Ag8). D2C5 antibody produced positive results against HCT-MPH that have been validated by ELISA assay. D2C5 specific reactivity has been also analyzed and confirmed using immunoblot and immunohistochemistry assays.
Several antigen preparation methods were tested, including whole cell fixation using formalin, outer membrane proteins (OMP) extraction from HCT-116, and cell homogenate preparation from mouse colon cancer which has been developed by colitis induction (data are not presented). Unfortunately, the mice did not immunologically respond well to cell homogenate from colon cancer. Moreover, weak responses were induced by fixed cells and OMP. Therefore, HCT-MPH was selected for immunization as it triggered the highest polyclonal antibody response. Despite the poor mice response to immunization with whole tumor cell reported in this study, several studies showed a moderate to high response to whole cell vaccines (Obata et al., 2004; Tian et al., 2014; Xia et al., 2018).
The lack of expression of tumor specific antigens may explain the inability of immune system to attack the growing tumor and eradicate it (Schreiber et al., 2011). Based on that, our study reported a weak immune response after injecting autologous mouse colorectal cancer extract or cell homogenate. The failure of generating of an immune response has been explained by others to be due to the minimal expression of tumor antigens on cancer cells (Arlen et al., 2014).
Our study reported ineffectual immune response after injecting OMP which is inconsistent with the results of a previous study that reported strong and sufficient immune response to OMP (Pal et al., 2006; Chiang et al., 2010). Based on the results reported by (Morrison et al., 1989) trachoma clinical trials and preclinical animal models, it has been concluded that immunization procedures using whole organism vaccines produce poor immune response and cannot fully protect individuals. Furthermore, whole organism vaccines can elicit notorious adverse effects particularly a hypersensitivity reaction upon re-exposure to the organism (Morrison et al., 1989; Hozbor, 2017).
The specificity of D2C5 for human colon cancer of different stages has been examined in our study. Our results indicated that D2C5 instigated differential and specific reactivity to all colon cancer stages. A powerful cytoplasmic reactivity was observed with stage IV compared to stage II and stage III (Fig. 4). The D2C5 immunostaining patterns in normal and cancer colonic tissues are similar to those produced by Cytokeratin 20 (CK20). Cytokeratins (CKs) are a family of more than 20 polypeptides which are used as epithelial differentiation markers. Various cytokeratin subtypes are expressed in a tissue-specific manner on cells of epithelial origin (Chu and Weiss, 2002; Wang et al., 1995). CK20 is highly expressed in colorectal carcinoma as well as in normal colonic epithelium which is mainly localized in the upper compartments of the colonic crypts (Park et al., 2002). The difference in CK20 expression between normal and tumor tissues is due to the fact that CK20 is highly expressed on the surface of non-proliferative epithelium, and it is undetectable in proliferative crypt epithelium (Hernandez et al., 2005). Immunoblot results indicated that D2C5 mAB can recognize a single membrane protein of about 55 KDa (Fig. 7). The protein reactivity and localization in the epithelial layer of tissue sections suggested that D2C5 may target one of the cytokeratin family proteins (Granados-Romero et al., 2017). The molecular weight of human CK 20 protein falls in the range of 46.6 to 50 kDa (Moll et al., 1992). To confirm whether D2C5 can target CK20 or other novel colon cancer antigens, a comprehensive analysis such as proteomics mass spectrometry should be conducted in future studies.
5 Conclusion
In summary, we have successfully generated one clone after fusion (D2C5) and it was of IgG1 kappa light chain and reactive to a protein with macular weight about 55 kDa using immunoblot. The observations from IHC result suggest the localization of the target protein in the cytoplasm of epithelial cells with high expression in more advanced cases, suggesting a cytokeratin pattern of staining. Further identification of target protein using advanced technique is recommended.
Acknowledgment
This work was supported by the Deanship of Scientific Research and Higher Education-Yarmouk University, (Grant No. 29/2017).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Monoclonal antibodies that target the immunogenic proteins expressed in colorectal cancer. World J. Gastrointest. Oncol.. 2014;6(6):170-176.
- [Google Scholar]
- The value of CDX2 and cytokeratins 7 and 20 expression in differentiating colorectal adenocarcinomas from extraintestinal gastrointestinal adenocarcinomas: cytokeratin 7-/20+ phenotype is more specific than CDX2 antibody. Diagn. Pathol.. 2012;23(7):9.
- [Google Scholar]
- A rapid and sensitive method for the quantitation microgram quantities of a protein isolated from red cell membranes. Anal. Biochem.. 1976;72(248):e254.
- [Google Scholar]
- Chiang, C.L.L., Benencia, F., Coukos, G., 2010. Whole tumor antigen vaccines. In: Seminars in Immunology, vol. 22, no. 3, pp. 132–143.
- Keratin expression in human tissues and neoplasms. Histopathology. 2002;40:403-439.
- [Google Scholar]
- Tumor markers in colorectal cancer, gastric cancer and gastrointestinal stromal cancers: European group on tumor markers. Int. J. Cancer. 2014;134(11):2513-2522.
- [Google Scholar]
- CK20 and CK7 protein expression in colorectal cancer: demonstration of the utility of a population-based tissue microarray. Hum. Pathol.. 2005;36:275-281.
- [Google Scholar]
- Outer membrane vesicles: an attractive candidate for pertussis vaccines. Expert Rev. Vaccines. 2017;16:193-196.
- [Google Scholar]
- Global burden of colorectal cancer: emerging trends, risk factors and prevention strategies. Nat. Rev. Gastroenterol. Hepatol.. 2019;16:713-732.
- [Google Scholar]
- Colorectal cancer tumour markers and biomarkers: Recent therapeutic advances. World J. Gastroenterol.. 2016;22:1745.
- [Google Scholar]
- Tumor markers in staging and prognosis of colorectal carcinoma. Neoplasma. 2008;55(2):138.
- [Google Scholar]
- Touch imprint cytology and frozen-section analysis for intraoperative evaluation of sentinel nodes in early breast cancer. Anticancer Res. 2012;32(8):3523-3526.
- [Google Scholar]
- Cytokeratin 20 in human carcinomas. A new histodiagnostic marker detected by monoclonal antibodies. [Research Support, Non-U.S. Gov't] Am. J. Pathol.. 1992;140:427-447.
- [Google Scholar]
- Chlamydial disease pathogenesis. The 57-kD chlamydial hypersensitivity antigen is a stress response protein. J. Exp. Med.. 1989;170:1271-1283.
- [Google Scholar]
- Formalin-fixed tumor cells effectively induce antitumor immunity both in prophylactic and therapeutic conditions. J. Dermatol. Sci.. 2004;34:209-219.
- [Google Scholar]
- Immunization with the Chlamydia trachomatis major outer membrane protein, using adjuvants developed for human vaccines, can induce partial protection in a mouse model against a genital challenge. Vaccine. 2006;24:766-775.
- [Google Scholar]
- Expression of cytokeratins 7 and 20 in primary carcinomas of the stomach and colorectum and their value in the differential diagnosis of metastatic carcinomas to the ovary. Hum. Pathol.. 2002;33:1078-1085.
- [Google Scholar]
- Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science. 2011;331:1565-1570.
- [Google Scholar]
- Cellular immunotherapy using irradiated lung cancer cell vaccine co-expressing GM-CSF and IL-18 can induce significant antitumor effects. BMC Cancer. 2014;14:1-13.
- [Google Scholar]
- Coordinate expression of cytokeratins 7 and 20 defines unique subsets of carcinomas. Appl. Immunohistochem.. 1995;3:99-107.
- [Google Scholar]
- Inhibition of prostate cancer growth by immunization with a GM-CSF-modified mouse prostate cancer RM–1 cell vaccine in a novel murine model. Oncol. Lett.. 2018;15:538-544.
- [Google Scholar]







