Translate this page into:
Protective effects of neoagarotetraose against oxidative stress via Nrf2/HO-1 signaling pathway in hydrogen peroxide-induced HepG2 cells
⁎Corresponding author at: College of Chemical Engineering, Huaqiao University, No.686, Jimei Avenue, Xiamen 361021, China. yejenny@hqu.edu.cn (Jing Ye)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Abstract
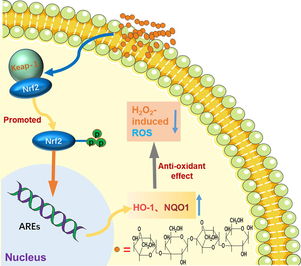
Neoagarooligosaccharides (NAOS), possessing a variety of bioactivities, are mainly produced by β-agarases cleaving the β-1,4-glycosidic bond of agar or agarose. Previous study indicated that NAOS has antioxidant effect, however, the NAOS monomers on the activity of scavenging ROS has not been deeply clarified till now. In this study, the protective effects of NAOS especially neoagarotetraose (NA4) against H2O2-stimulated oxidative damage in HepG2 cells and their underlying molecular mechanism were investigated. The results indicated that NA4 could inhibit the production of reactive oxygen species (ROS) and enhance the activities of antioxidant enzymes including superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px) in H2O2-induced HepG2 cells. Moreover, NA4 activated the mRNA and protein expression levels of the nuclear factor erythroid-2-related factor 2 (Nrf2), which further up-regulated the mRNA and protein expression of the downstream antioxidant enzymes including heme oxygenase-1 (HO-1) and NAD(P)H quinone oxidoreductase 1 (NQO1), indicating that NA4 could attenuate H2O2-induced oxidative damage through Nrf2 pathway in HepG2 cells. The study suggested that NA4 could be an effective antioxidant candidate and it may be developed as a promising component in healthy food exerting antioxidation effect.
Keywords
Neoagarotetraose
Hydrogen peroxide
Oxidative
HepG2 cells
Nrf2 signaling pathway
- NAOS
-
Neoagarooligosaccharides
- NA4
-
neoagarotetraose
- SOD
-
superoxide dismutase
- CAT
-
catalase
- GSH-Px
-
glutathione peroxidase
- Nrf2
-
nuclear factor-erythroid 2
- HO-1
-
heme oxygenase-1
- ROS
-
reactive oxygen species
- Keap1
-
kelch-like ECH associated protein 1
- NQO1
-
NAD(P)H quinone oxidoreductase 1
- ARE
-
antioxidant response element
- DMEM
-
Dulbecco’s modiffed Eagle’s medium
- DMSO
-
dimethyl sulfoxide
- DCFH/DA
-
2,7-dichloroflurescein diacetate
- MTT
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- PMSF
-
phenylmethanesulfonyl fluoride
- BCA
-
bicinchoninic acid
- PVDF
-
polybinylidene difluoride
- HRP
-
horseradish peroxidase
Abbreviations
1 Introduction
Reactive oxygen species (ROS), regarded as by-product of oxygen metabolism, are produced by aerobic organisms in the body (He, Bu, Xie, & Liang, 2019). In normal circumstances, the antioxidant defense system in vivo would remove the produced ROS, maintaining the free radicals in our body staying in a dynamic equilibrium by keeping the generation and elimination of ROS dynamically balanced (He et al., 2020). However, the homeostatic system in the body would be disrupted when it comes to overproduced ROS, which can lead to oxidative stress, causing lipid peroxidation, protein and enzyme degradation, and nucleic acid damage, and eventually resulting in a wide range of diseases including aging, diabetes, obesity, cardiovascular disease, neurological disorders, and cancer, etc (Hamzeh, Wongngam, Kiatsongchai, & Yongsawatdigul, 2019; Meng, Wang, & Liu, 2020; Wang et al., 2021).
Antioxidants can keep the cells and tissues away from the oxidative damage caused by endogenous ROS and thus prevent the development of diseases through a variety of biological mechanisms, among which the most critical antioxidant pathways in the body is the Nrf2 signaling (Yao et al., 2020). Generally, Nrf2, under physiological condition, binds to kelch like ECH associated protein 1 (Keap1) to be fixed in cytosol and kept at low level (Moritani et al., 2020). In response to oxidative stress, Nrf2 dissociates from the Nrf2 and Keap1 complex, then translocate to the nucleus, where Nrf2 combines to antioxidant response element (ARE), thereby activating the expression of cellular enzymes of antioxidant defense system including the first line and phase II detoxification enzymes, such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px), heme oxygenase-1 (HO-1) and NAD(P)H quinone oxidoreductase 1 (NQO1) (Darwish et al., 2020; Wang et al., 2020).
Neoagarooligosaccharides (NAOS) are natural oligosaccharides mainly generated by β-agarases cleaving the β-1,4 glycosidic bond of polysaccharide agar or agarose from red seaweeds (Hong et al., 2017a,b,c). NAOS has been reported to possess a variety of bioactivities such as anti-obesity, prebiotic properties, hypoglycemic effect, cholesterol-lowering properties, hepatoprotective effect, antitumor, whitening and skin-moisturizing effect, anticariogenic activity, antioxidation and anti-inflammation (Hong et al., 2017a,b,c; Kim, Yun, Yu, Kim, & Kang, 2017; Lee et al., 2017; Oh et al., 2022; Yang et al., 2017). Previous studies have found that NAOS has a protective effect against acetaminophen-induced liver injury, and NAOS achieves its protective effect against liver injury mainly by activating Nrf2 and enhancing antioxidant effects (Yang et al., 2021; Yang et al., 2020), however, the studied NAOS was the mixture of oligosaccharides with different degree of polymerization, the NAOS monomers on the activity of scavenging ROS has not been clarified in this study. To deeply understand the antioxidant effect of NAOS monomer and its potential mechanism is cornerstone for the development of NAOS as nutritional supplements.
Therefore, in the present study, the antioxidant effect of neoagarooligosaccharides especially neoagarotetraose (NA4) on H2O2-induced oxidative damage in HepG2 cells was evaluated through cell viability, ROS production and intracellular antioxidant enzyme activity. In additional, the genes and proteins expression levels of Nrf2 and downstream enzymes including HO-1 and NQO1 were examined to confirm whether the potential mechanism of NA4 on H2O2-induced oxidative stress was related to Nrf2 signaling pathway. Our present study would provide the possibility of NA4 to be developed to be potential chemopreventive candidate.
2 Materials and methods
2.1 Material and chemicals
3-(4,5dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), dimethylsulfoxide (DMSO) and 2, 7-dichloroflurescein diacetate (DCFH/DA) were purchased from Sigma (St. Louis, MO). Penicillin (100 U·ml−1) and streptomycin (100 µg·ml−1) were purchased from Lonza (Castleford, UK). Dulbeco’s modified Eagle’s medium (DMEM), Hank’s balanced salt solution (HBSS), PBS (pH 7.4) and foetal bovine serum (FBS) were purchased from Gibco® (Grand Island, NY, USA.). Superoxide dismutase (SOD), reduced glutathione (GSH), glutathione peroxidases (GSH-Px) and catalase (CAT) reagent kits were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). BCA protein assay kit, ROS assay, radio immunoprecipitation assay (RIPA) lysis buffer, Trizol and phenylmethanesulfonyl fluoride (PMSF) were obtained from Beyotime Biotechnology (Shanghai, China). Primary antibodies used for western blotting analysis were rabbit anti-Nrf2 (1:1000), rabbit anti-HO-1 (1:1000) and rabbit anti-NAD(P)H quinone oxidoreductase 1 (NQO1, 1:1000) were purchased from Abcam (Cambridge, UK). β-actin and Lamin B1 were obtained from Bioworld Technology, Inc. (Nanjing, China). All other reagents were obtained from Sigma-Aldrich.
2.2 Cell lines and culture
HepG2 cells were purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). They were cultured in DMEM supplemented with 10 % FBS, 100 U·ml−1 of penicillin and 100 µg·ml−1 of streptomycin under a humidified environment at 37 °C in an incubator with 5 % CO2. The cells were split at 80–90 % confluency using 0.25 % trypsin solution.
2.3 Preparation of neoagarooligosaccharides
The preparation of neoagarooligosaccharide was carried out as described previously (Lin, Ye, Huang, Yang, & Xiao, 2019). In brief, 3 % agar solution was completely dissolved in 0.1 M Tris-HCl solution by heating and then cooled to 45 °C. Subsequently, the solution was treated with β-agarase for 4 h, after inactivation with boiling water bath for 15 min and centrifugation for 30 min (12,000 rpm), the insoluble agar was removed and NAOS was in the supernatant. The lyophilized NAOS powder was resuspended in 0.1 M NH4HCO3,which was loaded onto a Bio-Gel P2 column (1.8 × 150 cm, Bio-Rad Laboratories, Hercules, CA, USA), eluting with NH4HCO3 at a flow rate of 0.4 ml·min−1 to obtain neoagarobiose (NA2), neoagarotetraose (NA4), neoagarohexaose (NA6), neoagarooctaose (NA8), neoagarodecaose (NA10), and neoagarododecaose (NA12).
2.4 Chemical characterization of neoagarooligosaccharides
The obtained NA2, NA4, NA6, NA8, NA10, and NA12 was characterized by ESI-TOF-MS and the purities of them were detected by HPLC. ESI-TOF-MS was carried out on a Q Exactive Hybrid Quadrupole Orbitrap mass spectrometer (Thermo, Bremen, Germany) coupled with an ESI source in positive ion mode. HPLC was performed on an Agilent 1260 equipped with an evaporative light scattering detector and an Asahipak NH2P-50 4E multimode column (250 mm × 4.6 mm, 5 μm). The mobile phase was water: acetonitrile (35:65) with flow rate set at 1.0 ml·min−1. The detector atomization temperature was 75 °C and the column temperature was set at 30 °C.
2.5 MTT assay for cell viability evaluation
MTT assay was used to measure the cell viability. In brief, HepG2 cells were seeded in 96-well plates (Thermo Fisher Scientific, Waltham, MA, USA) and cultured for 24 h. After appropriate treatments, the cells were incubated with 20 μl 0.5 mg·ml−1 MTT reagent. Following incubation at 37 °C for 4 h, media were discarded from each well, 100 μl DMSO was added, cell viability was determined by measuring absorbance of the solubilized formazan product at 570 nm.
2.6 ROS assay
The level of ROS in HepG2 cells was measured using DCFH/DA probe (He et al., 2019). Briefly, HepG2 cells at a density of 4 × 105 cells/well were seeded in 6-well plates. The control and model groups were added DMEM with 10 % FBS and NAOS groups were treated with 50, 100, 200 μg·ml−1 NAOS. After incubation for 24 h, the medium of model and NAOS groups were replaced by 500 μM H2O2 and control group by DMEM. After being exposed to H2O2 for 6 h, the medium was discarded and the cells were rinsed with PBS. 10 μM DCFH/DA was added to each well and the cells were incubated at 37 °C for 30 min shielded from light. The probe was washed off with PBS for three times and the fluorescence intensity of each well was observed with the inverted fluorescence microscope (Nikon, Tokyo, Japan).
2.7 Measurement of SOD, CAT and GSH-Px
The activities of SOD, CAT and GSH-Px were measured using the commercial kits (Nanjing Jiancheng Biology Engineering Institute, Nanjing, China). In brief, HepG2 cells after the treatment were collected and homogenized with RIPA lysis buffer and then centrifuged for 20 min at 12000 rpm, 4 °C. The SOD, CAT and GSH-Px levels of the supernatant were detected according to the manufacturer’s instructions.
2.8 Real-Time qPCR
Total RNA in cells of all the group was extracted using Trizol regent according to the manufacture’s instruction, the RNA was then reverse-transcribed into the first-stand complementary DNA (cDNA) using PrimeScript RT Master Mix, followed by intercalator qPCR assay using TB green Premix Ex Taq II. The primers for real-time qPCR (RT-qPCR) were listed in Table 1. The expression of target genes was normalized to the expression of β-actin and analyzed by the 2ΔΔct method.
Name
Sequence (5′-3′)
NQO-1
F: 5′-GTGGTGGAGTCGGACCTCTATG-3′
R: 5′-AAGCCAGAACAGACTCGGCAG-3′
HO-1
F: 5′-ATGACACCAAGGACCAGAG-3′
R: 5′-TAAGGACCCATCGGAGAAG-3′
Nrf2
F: 5′-AAGCTTTCAACCCGAAGCAC-3′
R: 5′-TTTCCGAGTCACTGAACCCA-3′
β-actin
F: 5′-AGGCCAACCGCGAGAAGATG-3′
R: 5′-AGGCCAACCGCGAGAAGATG-3′
2.9 Western blot analysis
The protein expression of Nrf2, HO-1, and NQO1 was measured by western blot. Briefly, HepG2 cells were incubated in 6-well plates for 24 h. After appropriate treatments, the cells were collected and lysed with ice-cold RIPA lysis buffer. The protein concentration was determined by bicinchoninic acid (BCA) protein assay kit (Pierce, Rockford, IL, USA) according to the manufacturer's protocol. The loading buffer was added and the samples were boiled for 5 min. Equal amounts of lysate protein (20 μg/lane) were separated by 10 % SDS-PAGE and then transferred to a polybinylidene difluoride (PVDF) membrane. After that, the membranes were blocked in 5 % (w/v) skimmed milk and then the blots were incubated with specific primary antibodies overnight at 4 °C followed by incubating with horseradish peroxidase (HRP)-conjugated secondary antibody for 2 h and then the secondary antibody was washed off as before. Finally, the blots were probed using enhanced chemiluminescence (ECL) and auto radiographed. β-actin and Lamin B1 were used as internal controls.
2.10 Statistical analysis
The data are presented as mean ± standard deviation (SD). Statistical significance was assessed by the one-way ANOVA procedure followed by Tukey test using SPSS 16.0 software (IBM, USA). A value of p < 0.05 was considered to be statistically significant in this work. All experiments were performed in triplicate.
3 Result and discussion
3.1 The characterization of neoagarooligosaccharides
After the separation on Bio-Gel P2 column, the obtained fractions were detected by ESI-TOF-MS with the results displayed in Table 2. The ESI-TOF-MS analysis confirmed that the purified neoagarooligosaccharides were NA2, NA4, NA6, NA8, NA10, and NA12, respectively. And the results of FTIR and NMR detection were the same as our previous study (Lin et al., 2019a,b). The purity of the obtained neoagarooligosaccharides was detected by HPLC-ELSD, the results in Fig. 1 turned out to be 99.4 %, 99.0 %, 98.3 %, 98.0 %, 97.5 % and 97.8 % for NA2, NA4, NA6, NA8, NA10, and NA12, respectively, indicating enzymatic hydrolysis was an effective method for the preparation of NAOS with high purities. Hong et al also used crude DagA agarase to hydrolyze agar to prepare NAOS (Hong et al., 2017a,b,c), the composition of the NAOS powder was composed of NA2, NA4, and NA6, which were present in the ratio 3:69:28, respectively, and the purity of NAOS (NA2 + NA4 + NA6) in the powder was 65 ± 5 % (Hong et al., 2017a,b,c).
Purified oligosaccharides
ESI-TOF-MS signals (m/z) [M + Na]+
Calculated molecular weight
neoagarobiose
347.0948
347.0954
neoagarotetraose
653.1887
653.1905
neoagarohexaose
959.2865
959.2856
neoagarooctaose
1265.3753
1265.3807
neoagarodecaose
1571.4751
1571.4757
neoagarododecaose
1877.5752
1877.5708

The purities of neoagarooligosaccharides detected by HPLC.
3.2 The effect of NAOSs on HepG2 cells
Generally, cell viability is regarded as an effective indicator of toxicity on cells and ideal antioxidants should have low toxicity, we firstly detected the effect of NAOSs including NA2, NA4, NA6, NA8, NA10, and NA12 on the viability of HepG2 cells. As shown in Fig. 2a, after 24 h of treatment of neoagarooligosaccharide monomers with different concentrations (25, 50, 100, 200, and 400 μg·ml−1), the cell viability was over 95 %, indicating that all the six neoagarooligosaccharide monomers with concentrations of 25 to 400 μg·ml−1 had no obvious cytotoxicity, and statistics was also analyzed with no significance with all of the concentrations screened. Our findings are consistent with those of Yang et al. who showed that administration of up to 3 mg /ml of NAOS to HepG2 cells also did not produce cytotoxic effects (Yang et al., 2017).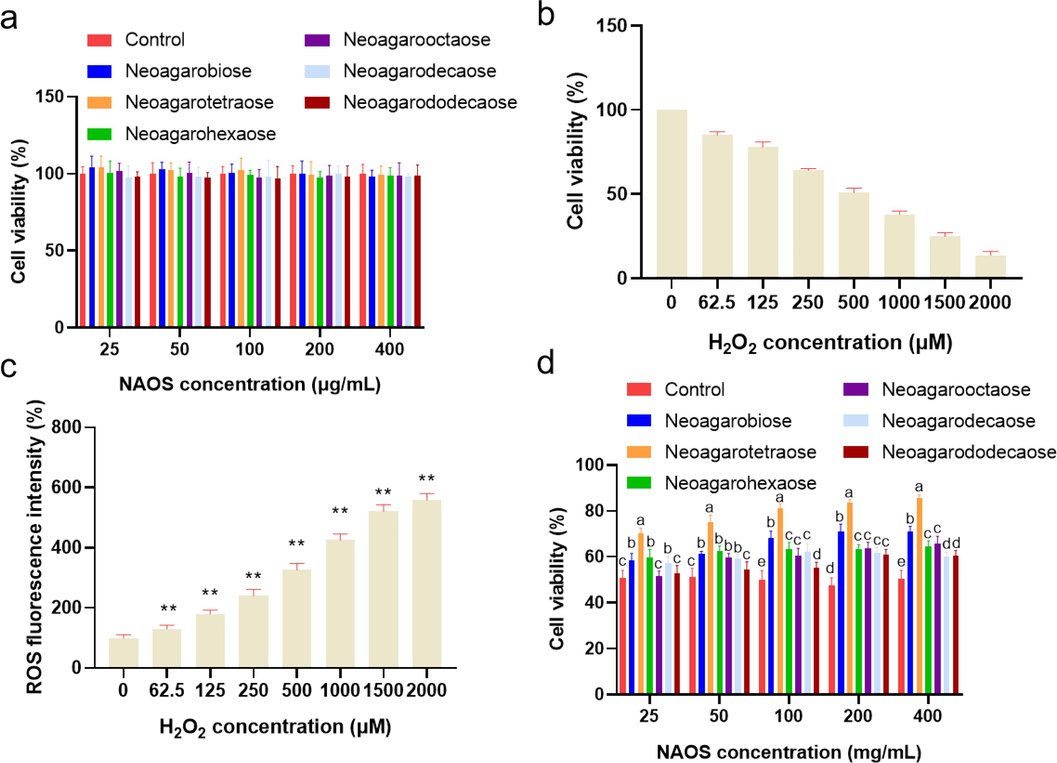
(a) Effect of NAOS on the viabilities of HepG2 cells. (b) and (c) Effect of H2O2 on the viabilities of HepG2 cells. Data were expressed as the mean ± SD, *P < 0.05, ** P < 0.01 (d) Effect of NAOS on the viabilities of H2O2-induced HepG2 cells. Bars with different lowercase letters are statistically different from each other (p < 0.05).
3.3 Evaluation of H2O2 concentration on cell viability
Hydrogen peroxide is commonly used to induce oxidative stress response in cells (Kim & Jang, 2009). The H2O2 concentration on cell viability which is usually used as a direct indicator of cytotoxicity was evaluated to find out the appropriate H2O2 concentration for inducing intracellular oxidative stress. As shown in Fig. 2b, the cell viability was negatively correlated with the concentration of H2O2. When the H2O2 concentration was 500 µM, the cell viability was down to 50.94 %, and the cell viability was dramatically decreased with the H2O2 concentration higher than 500 µM.
It has been reported that H2O2 can directly damage macromolecules such as DNA and lipids, allowing oxidative damage to spread to the cell, resulting in the consequent production of intracellular ROS (Yi, Din, Zhao, & Liu, 2020). Thus, the intracellular concentration of ROS is usually used to indicate the oxidative damage of cells. As could be seen in Fig. 2c, the intracellular oxidative stress in HepG2 cells gradually increased with the increase of H2O2 concentration. Therefore, H2O2 concentration of 500 µM was chosen to induce intracellular oxidative stress for further study.
3.4 The effects of NAOSs on cell viability of H2O2-induced HepG2 cells
Cell viability is often used as an important indicator to evaluate the toxicity of a substance or its protection against toxic damage on cells. Therefore, to evaluate the protective effect of the six neoagarooligosaccharide monomers on H2O2-induced injury to HepG2 cells, the cell viability was measured using MTT assay. The result shown in Fig. 2d indicated that cells treated with H2O2 alone were seriously damaged with the cell mortality rate as high as 50 % after 2 h incubation. Whereas, NAOS had obvious protective effect on H2O2-induced HepG2 cells, the cell death ratio was significantly reduced with comparison to H2O2 alone. Notably, the effect of NA4 was better than the other monomers. The cell viability significantly increased with NA4 concentration increased from 25 µg·ml−1 to 100 µg·ml−1, then the cell viability mildly increased with NA4 concentration higher than 100 µg·ml−1, indicating NA possessed potential antioxidant effect against the H2O2-induced oxidative damage in HepG2 cell. Therefore, NA of 25, 50 and 100 µg·ml−1 was taken to further investigate the protective effect on HepG2 cells damage induced by H2O2.
3.5 Preventive effect of NA4 on ROS generation
Excessive ROS in cells can give rise to oxidative stress, leading to lipid peroxidation, proteins and enzymes damage, and DNA mutation, which eventually cause aging, cardiovascular disease, obesity, diabetes, cancer, etc (Gowd, Xie, Sun, & Chen, 2020). Therefore, the preventive effect of NA4 on lipid peroxidation in HepG2 cells was investigated. As a visual indicator of toxicity, cell morphology was firstly examined. As shown in Fig. 3a, HepG2 cells of control group grew well with the shape of round or polygon, clear boundary and tight arrangement. In contrast, the cells treated with 500 µM H2O2 lost their good shape, cytoplasm shrunk, membrane bubbled and turned translucent with the number greatly reduced. After the treatment of NA4, the number of cells significantly increased in a dose-dependent manner as well as cell boundary turned complete and clear.
(a) The preventive effects of NA4 on morphology and ROS production in H2O2-induced HepG2 cells. (Ⅰ: Control; Ⅱ: H2O2 (500 μM); Ⅲ: H2O2 + 25 μg/ml Neoagarotetraose; Ⅳ: H2O2 + 50 μg/ml Neoagarotetraose; Ⅴ: H2O2 + 100 μg/ml Neoagarotetraose) (b) Quantitative analysis of the DCF fluorescence of NA4 against H2O2-induced HepG2 damage. NA4 on the activities of CAT (c), GSH-Px (d), and SOD (e). Data were expressed as the mean ± SD, *p < 0.05, ** p < 0.01, *** p < 0.001.
The intracellular ROS levels were measured using FACS. The results shown in Fig. 3a and b indicated that the ROS in the cells treated with H2O2 significantly increased with comparison to control group, indicating the increased oxidative stress in HepG2 cells. After the treatment of different concentration of NA4, the H2O2-stimulated intracellular ROS level was dose-dependently decreased. The concentration of 100 µg·ml−1 NA4 showed the better effect than the other two groups. The study of Yang et al also demonstrated that NAOS possesses the antioxidant properties against tert-butyl hydroperoxide (t-BHP)-induced ROS formation (Yang et al., 2020), but the NAOS used in their study was the crude product of enzymatic hydrolysis of agar, and there is no way to know which degree of polymerization of NAOS played a role.
3.6 Effect of NA4 on the activities of antioxidant enzymes
Normally, the produced ROS would be eliminated by the endogenous antioxidants and antioxidant enzymes including CAT, GSH-Px, and SOD (Yi et al., 2020). Therefore, the ROS in the body is maintained at a certain level and in a dynamic balance, showing no damage to cells and tissues. Excessive H2O2 would break this balance, lead to the increase of ROS and decrease of the activities of antioxidant enzymes. In order to investigate the protective effect of NA4 on the antioxidant enzymes activities, the contents of those enzymes in HepG2 cells were determined. As shown in Fig. 3c-e, H2O2 stimulation remarkably suppressed the activities of CAT, GSH-Px, and SOD, showing the oxidative stress in HepG2 cells. Whereas, NA4 significantly increased the content of CAT in a dose-dependent manner. As for GSH-Px and SOD, their activities were dramatically up-regulated with NA4 concentration of 50 µg·ml−1, and as the concentration of NA4 increased, the activities of the two enzymes increased (p < 0.01). Thus, NA4 can increase the activities of antioxidant enzymes in HepG2 cells, and has a good effect of eliminating the enhanced intracellular ROS generated by H2O2. Notably, HepG2 cells exposure to H2O2 can lead to the formation of ROS, thus result in oxidative stress, which in turn will generate ROS or dysfunction of intracellular antioxidant system. Among which, CAT, GSH-Px, and SOD are the critical antioxidants and major scavengers of free radical in the cell. Previous reports indicated that H2O2 triggered the depletion of CAT, GSH-Px, and SOD (Liu et al., 2023b; Meng et al., 2024). In agreement with the previous studies, our results demonstrated that H2O2 caused the decrease of the activities of CAT, GSH-Px, and SOD. Usefully, the H2O2-induced the depletion of these antioxidants was reversed by NA4 treatment, indicating that an enhance in the anti-oxidative enzyme activities from NA4 administration could beneficially restrain oxidative stress.
3.7 The up-regulation of antioxidant enzymes genes and protein expressing by NA4
Nrf2 is a well-known transcription factor responding to oxidative damage. Generally, Nrf2 is bound to Keap1 as an inactive complex, upon oxidative stress, Nrf2 is dissociate from the complex and translocates into nucleus after phosphorylation, leading to the expression of cytoprotective and antioxidant genes including HO-1, NQO1, SOD, etc (Lee et al., 2018; Pang et al., 2016; Yi et al., 2020). In order to investigate the underlying mechanism of the protective effect of NA4 on antioxidant stress in HepG2 cells, the key genes and proteins expression of Nrf2 and its downstream antioxidant enzymes such as HO-1 and NQO1 were assessed in our study. As shown in Fig. 4a-c, the mRNA level of Nrf2 was found to be lower in cells with H2O2 treatment alone, and remarkable increases were observed in HepG2 cells after the treatment of NA4 in a dose-dependent manner, meanwhile, the mRNA expression of HO-1 and NQO1 were dramatically higher in NA4 groups compared to H2O2 alone in HepG2 cells. The same tendency was discovered in the protein expressions of Nrf2, HO-1, and NQO1 (Fig. 4d and e). The enhanced Nrf2 protein level indicated that Nrf2 was activated, subsequently, HO-1 and NQO1 levels were positively to Nrf2 expression in HepG2 cells. The results revealed that NA4 could exert the protection of the H2O2-induced oxidative damage in HepG2 cells through Nrf2-mediated antioxidant signaling. Increasing researches has confirmed that Nrf2 is a critical regulator of the internal antioxidant enzymes, exhibiting the protective effect against oxidative damage (Dai, Zhang, Zhang, Ma, & Zhang, 2023). HO-1, one of the downstream antioxidant enzyme of Nrf2, possesses antioxidant and anti-inflammatory properties. It has been reported that HO-1 is rate-limiting enzyme in the degradation of heme, catalyzing heme metabolism into biliverdin, carbon monoxide, free iron ions, and bilirubin. Oxidative stress could up-regulate the expression of HO-1 gene through the level of Nrf2 (Hattori, Narita, Mitsui, Uehara, & Ichihara, 2022). A growing body of studies have shown Nrf2 gene knockout mice have greatly reduced activities and contents of various antioxidant enzymes in vivo, revealing the indispensable role of Nrf2 in regulating cellular avoidance of oxidative damage (Li et al., 2020; Xiang et al., 2022). Our present study indicated the Nrf2 signaling pathway could be activated by NA4, playing the protective role against oxidative stress by re-regulating the expression levels of the related antioxidant genes including Nrf2, HO-1, and NQO1. Lui et al. showed that Paeonia lactiflora petal flavonoid prevented oxidative stress by activating the Nrf2 pathway, which enhanced the expression levels of Nrf2, HO-1, and NQO1, thereby triggered the antioxidant defense responses (Liu et al., 2023a).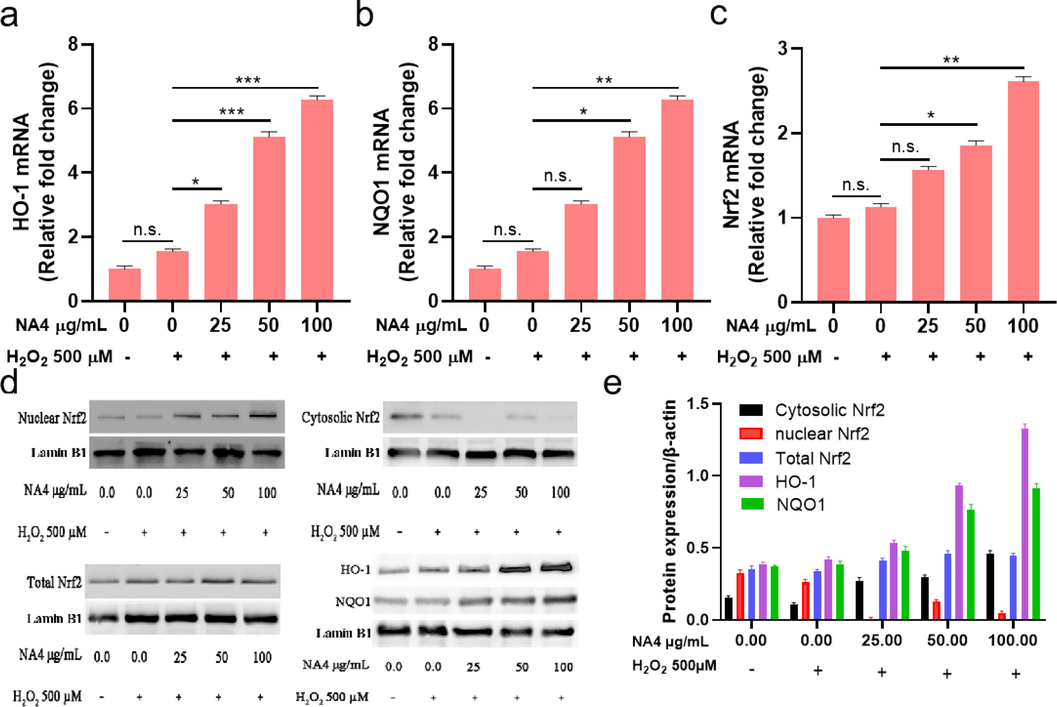
The Nrf2 (a), NQO1 (b), and HO-1 (c) mRNA level of NA4 on H2O2-induced HepG2 cells. (d) and (e) Effects of NAO on Nrf2, HO-1, and NQO1 protein levels in H2O2-induced HepG2 cells. Data were expressed as the mean ± SD, *p < 0.05, ** p < 0.01, *** p < 0.001.
4 Conclusions
In the present study, NA4, a neoagarooligosaccharide monomer, displayed significant protective effect against H2O2-stimulated oxidative stress in HepG2 cells in a dose dependent manner at the concentration of 25 to 100 µg·ml−1. NA4 with concentrations of 25 to 400 μg·ml−1 had no cytotoxicity on HepG2 cells and it could inhibit ROS production and enhance the activities of antioxidant enzymes including CAT, SOD, and GSH-Px in H2O2-stimulated oxidative damage HepG2 cells. Moreover, NA4 activated the mRNA and protein expression levels of Nrf2, which further increased the mRNA and protein expressions of the downstream antioxidant enzymes including HO-1 and NQO-1, suggesting that NA4 could attenuate H2O2-induced oxidative damage through Nrf2 pathway in HepG2 cells. The study indicated that NA4 has the potential to be developed as a promising antioxidant component in healthy food.
CRediT authorship contribution statement
Yayan Huang: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Validation, Writing – original draft. Fudi Lin: Data curation, Investigation, Methodology. Bingde Zheng: Data curation, Software. Yucheng Yang: Conceptualization, Investigation, Software. Na Zhang: Methodology, Data curation, Investigation. Xueqin Zhang: Methodology, Investigation. Qinglin Hong: Conceptualization, Supervision. Meitian Xiao: Funding acquisition, Investigation, Project administration. Jing Ye: Funding acquisition, Supervision, Writing – review & editing.
Acknowledgements
This work was supported by the National Nature Science Foundation of China (Grant No. 22078120), Open Research Fund of Academy of Advanced Carbon Conversion Technology, Huaqiao University (Grant No. AACCT0001), the Foreign Cooperation Project of Fujian Province (Grant No. 2021I0016), Nature Science Foundation of Fujian Province (Grant No.2022J01314), and the Natural Science Foundation of Xiamen (Grant No. 3502Z20227042). The authors appreciate Instrumental Analysis Center of Huaqiao University for analysis support.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Protective effect of agar oligosaccharide on male Drosophila melanogaster suffering from oxidative stress via intestinal microflora activating the Keap1-Nrf2 signaling pathway. Carbohydr. Polym.. 2023;313:120878
- [Google Scholar]
- Deoxynivalenol-induced alterations in the redox status of HepG2 cells: identification of lipid hydroperoxides, the role of Nrf2-Keap1 signaling, and protective effects of zinc. Mycotoxin Res.. 2020;36(3):287-299.
- [Google Scholar]
- Phenolic profile of bayberry followed by simulated gastrointestinal digestion and gut microbiota fermentation and its antioxidant potential in HepG2 cells. J. Funct. Foods. 2020;70:103987
- [Google Scholar]
- Cellular and chemical antioxidant activities of chicken blood hydrolysates as affected by in vitro gastrointestinal digestion. Poult. Sci.. 2019;98(11):6138-6148.
- [Google Scholar]
- The protective effects of blueberry honey from Canada against H2O2-induced cytotoxicity in human buccal mucosal cells. J. Oral Biosci.. 2022;64(3):359-365.
- [Google Scholar]
- Antioxidant activities and protective effects of duck embryo peptides against H2O2-induced oxidative damage in HepG2 cells. Poult. Sci.. 2019;98(12):7118-7128.
- [Google Scholar]
- Cytotoxic effects of polystyrene nanoplastics with different surface functionalization on human HepG2 cells. Sci. Total Environ.. 2020;723:138180
- [Google Scholar]
- In vitro and in vivo investigation for biological activities of neoagarooligosaccharides prepared by hydrolyzing agar with β-agarase. Biotechnol. Bioproc. e.. 2017;22(4):489-496.
- [Google Scholar]
- Toxicological evaluation of neoagarooligosaccharides prepared by enzymatic hydrolysis of agar. Regul. Toxicol. Pharm.. 2017;90:9-21.
- [Google Scholar]
- Anti-obesity and anti-diabetic eEffect of neoagarooligosaccharides on high-fat diet-induced obesity in mice. Mar. Drugs. 2017;15(4):90-101.
- [Google Scholar]
- Protective mechanism of quercetin and rutin using glutathione metabolism on HO-induced oxidative stress in HepG2 cells. Ann. N. Y. Acad. Sci.. 2009;1171:530-537.
- [Google Scholar]
- Different levels of skin whitening activity among 3,6-anhydro-l-galactose, agarooligosaccharides, and neoagarooligosaccharides. Mar. Drugs. 2017;15(10):132-141.
- [Google Scholar]
- Evaluation of in vitro anti-oxidant and anti-inflammatory activities of Korean and Chinese Lonicera caerulea. Nutr. Res. Pract.. 2018;12(6):486-493.
- [Google Scholar]
- Neoagarohexaose-mediated activation of dendritic cells via Toll-like receptor 4 leads to stimulation of natural killer cells and enhancement of antitumor immunity. BMB Rep.. 2017;50(5):263-268.
- [Google Scholar]
- Hepatocyte-specific Nrf2 deficiency mitigates high-fat diet-induced hepatic steatosis: Involvement of reduced PPARgamma expression. Redox Biol.. 2020;30:101412
- [Google Scholar]
- The Potential of neoagaro-oligosaccharides as a treatment of type II diabetes in mice. Mar. Drugs. 2019;17(10):541-557.
- [Google Scholar]
- Acidic hydrolyzed xylo-oligosaccharides bioactivity on the antioxidant and immune activities of macrophage. Food Res. Int.. 2023;163:112152
- [Google Scholar]
- Composition and antioxidant activity of Paeonia lactiflora petal flavonoid extract and underlying mechanisms of the protective effect on H2O2-induced oxidative damage in BRL3A cells. Hortic. Plant J.. 2023;9(2):335-344.
- [Google Scholar]
- Dietary Chito-oligosaccharide attenuates LPS-challenged intestinal inflammation via regulating mitochondrial apoptotic and MAPK signaling pathway. Int. Immunopharmacol.. 2024;126:111153
- [Google Scholar]
- Cytoprotection activity evaluation of (E)-3-(3,4-Dichlorophenyl)-1-(3,4-Dihydroxyphenyl)- Prop-2-En-1-One as a new antioxidant against H2O2-induced oxidative damage in PC12 Cells. Pharm. Chem. J.. 2020;53(10):942-946.
- [Google Scholar]
- Protective effects of rice peptide oryza peptide-P60 against oxidative injury through activation of Nrf2 signaling pathway in vitro and in vivo. ACS Omega. 2020;5(22):13096-13107.
- [Google Scholar]
- Neoagarooligosaccharides modulate gut microbiota and alleviate body weight gain and metabolic syndrome in high-fat diet-induced obese rats. J. Funct. Foods. 2022;88:104869
- [Google Scholar]
- A novel GSK-3beta inhibitor YQ138 prevents neuronal injury induced by glutamate and brain ischemia through activation of the Nrf2 signaling pathway. Acta Pharmacol. Sin.. 2016;37(6):741-752.
- [Google Scholar]
- MicroRNA-based regulatory mechanisms underlying the synergistic antioxidant action of quercetin and catechin in H2O2-stimulated HepG2 cells: Roles of BACH1 in Nrf2-dependent pathways. Free Radic. Biol. Med.. 2020;153:122-131.
- [Google Scholar]
- The double-edged roles of ROS in cancer prevention and therapy. Theranostics. 2021;11(10):4839-4857.
- [Google Scholar]
- Structural characterization of a novel marine polysaccharide from mussel and its antioxidant activity in RAW264.7 cells induced by H2O2. Food Biosci.. 2022;47:101659
- [Google Scholar]
- Neoagarooligosaccharides enhance the level and efficiency of LDL receptor and improve cholesterol homeostasis. J. Funct. Foods. 2017;38:529-539.
- [Google Scholar]
- Hepatoprotective effect of neoagarooligosaccharide via activation of Nrf2 and enhanced antioxidant efficacy. Biol. Pharm. Bull.. 2020;43(4):619-628.
- [Google Scholar]
- Neoagarooligosaccharide Protects against Hepatic Fibrosis via Inhibition of TGF-beta/Smad Signaling Pathway. Int. J. Mol. Sci.. 2021;22(4):2041.
- [Google Scholar]
- Insoluble-bound polyphenols of adlay seed ameliorate H2O2-induced oxidative stress in HepG2 cells via Nrf2 signalling. Food Chem.. 2020;325:126865
- [Google Scholar]
- Effect of soybean peptides against hydrogen peroxide induced oxidative stress in HepG2 cells via Nrf2 signaling. Food Funct.. 2020;11(3):2725-2737.
- [Google Scholar]







