Translate this page into:
Research progress on the removal of pesticides in water by Fe3O4-based adsorbents in the past decade: A review
⁎Corresponding author. jgfeng@yzu.edu.cn (Jianguo Feng)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Abstract
There are large amounts of residual pesticide in water, which has caused serious environmental problems worldwide and poses a great threat to environmental organisms and human health. Thus, effectively removing pesticides from the water environment is important, among which adsorption has become a promising method due to its low cost, simple operation, rapidity, and convenience. Fe3O4-based adsorbents have attracted much attention due to their magnetic properties, rapid recovery, and various modification methods. This review summarizes various materials (inorganics, polymers, silica, metal–organic frameworks, multi-layer composites) used to construct Fe3O4-based adsorbents for pesticides removal from water environment. On this basis, the effects of adsorbent dosage, adsorption time, temperature, pH, and other factors on the adsorption capacity of Fe3O4-based adsorbents were discussed. The adsorption mechanism is generalized as follows: van der Waals forces, hydrogen bonds, electrostatic interactions, π-π interactions and hydrophobic interactions. Finally, the future challenges and perspectives of Fe3O4-based adsorbents for practical water purification are proposed.
Keywords
Water environment
Fe3O4-based adsorbents
Pesticide removal
Effect factors
Adsorption mechanism
1 Introduction
The water environment is one of the fundamental elements that constitute the environment, is essential for the existence and development of human society, which can affect the life and development of human beings directly or indirectly (Nemeth et al., 2017, Westall & Brack, 2018). However, a large number of negligent human activities have caused pollution of the water environment, thereby reducing the coherence and functionality of ecosystems and posing a threat to the survival of environmental organisms and the health of humans (Evans et al., 2019, Lee et al., 2017). The introduction of anthropogenic pollutants into the water environment causes damage to the native characteristics of the water, thus making the water bodies inadequate to meet the most basic requirements for human and ecosystem continuity (Deletic & Wang, 2019, Mitchelmore et al., 2019). At present, water pollution has become an urgent global problem affecting the survival of humans and natural life.
Pesticides are synthetic, functional chemicals that are widely used in agriculture, private gardens, indoor public hygiene, and other areas to kill harmful organisms in the environment, control the occurrence of pests and the spread of human diseases, and greatly ensure improved crop yields and human health (Deka et al., 2021, Singh et al., 2020). To ensure the massive food needs resulting from the increase in the global population, pesticides have become an extremely vital factor in food production (de et al., 2020). However, traditional pesticides formulations such as emulsifiable concentrate, wettable powder can cause serious water pollution through subsurface drainage, runoff, leaching, and spray drift (Cryder et al., 2021).
It has been reported that the concentrations of pesticides in the aquatic environment range from 7 ng/L to 121222 ng/L in rivers and lakes, 23 ng/L to 3172 ng/L in wastewater streams, 20 ng/L to 1060 ng/L in groundwater, and 141 ng/L to 14629 ng/L in drinking water sources (Mojiri et al., 2020). Long-term low-dose pesticide exposure can lead to chronic diseases and toxic effects on organisms' immune, neurological, cardiovascular, and genetic systems, inducing carcinogenicity, teratogenicity, and mutagenicity (Kermani et al., 2021, Sidhu et al., 2019, Sun et al., 2019). Hence, the eradication of pesticide residues in the environment has received considerable attention.
At present, the methods of removing pesticide residues in water generally include physical and chemical treatments, such as adsorption (Del Rio et al., 2020), membrane filtration (Abdelhameed et al., 2016), biodegradation (Liu et al., 2022), and chemical catalysis (Vellingiri et al., 2017). However, most of these methods have certain limitations. Although membrane filtration can also purify water, it sometimes leads to pore clogging and high drainage, which reduces pesticide removal efficiency (Ullah et al., 2020). Biodegradation is an environmentally friendly solution. However, due to the use of microorganisms, the requirements for the environment in which they are used are quite strict, and the time required is long, which greatly limits their application (Liu et al., 2021a). Chemical catalytic methods often require the introduction of new catalysts into the system and often face problems such as unknown products after catalytic degradation or even secondary pollution (Zhou et al., 2021). Compared with the above methods, adsorption is a method that uses a solid adsorbent to directly extract pollutants from solid water, thereby purifying wastewater, which has been widely accepted as a promising technique with the advantages of high efficiency, rapidity, and convenience (Keshvardoostchokami et al., 2021, Mondol & Jhung, 2021).
Adsorption has been widely used in industries as an effective means of wastewater treatment (Ighalo et al., 2022). The adsorption effect is influenced by many factors (including self and external factors), among which the adsorbent plays a decisive role in the whole adsorption process (Jain et al., 2021). The adsorption effect of different adsorbates on different adsorbents varies widely, so the selection, combination, or modification of different adsorbents has become a current research trend (Singh et al., 2018). Although traditional adsorbents without magnetism can also remove pesticides from water, they always have limitations such as poor reusability and inconvenient recycling, which can easily cause secondary pollution to water bodies. Recently, Fe3O4-based adsorbents have emerged from numerous types of adsorbents by virtue of their unique magnetism, versatile functions, easy modification, diverse types, and so on (Wang et al., 2018b). Fe3O4 particles are easy to synthesize, low cost, and environmentally friendly, and most importantly, their strong magnetic properties enable them to be easily recovered after adsorption (Fu et al., 2023), whereas the adsorption capacity of purely naked Fe3O4 particles is relatively poor, and they tend to agglomerate in water, which is not conducive to adsorption (Gil et al., 2019). Hence, Fe3O4 particles are often modified by various moieties or materials with particular functions to obtain superior adsorbents with improved adsorption performance and to avoid agglomeration (Wei et al., 2022).
This review discusses the adsorption and purification of pesticides from aqueous solutions based on Fe3O4-modified materials. Although several reviews have been published on adsorbents for pesticide adsorption in water, to our knowledge, there is no systematic review on Fe3O4-based adsorbents for pesticide removal. In addition, adsorption is an interaction between surfaces, which is associated with the physicochemical properties of the adsorbent and the pesticide, so this review also discusses the factors affecting the adsorption effect, the main mechanism of pesticide adsorption and the current problems and development prospects of Fe3O4-based adsorbent applications.
2 Materials used for surface modification of Fe3O4
Many current studies have grafted, coated, or modified Fe3O4 surfaces with modification materials, including inorganics (clays, graphene), polymers, silica, and metal–organic frameworks, to obtain adsorbents with unique properties and excellent adsorption performance, which ensure that the adsorbents have strong magnetic properties for easy recovery. The modification of Fe3O4 to construct adsorbents is shown in Fig. 1.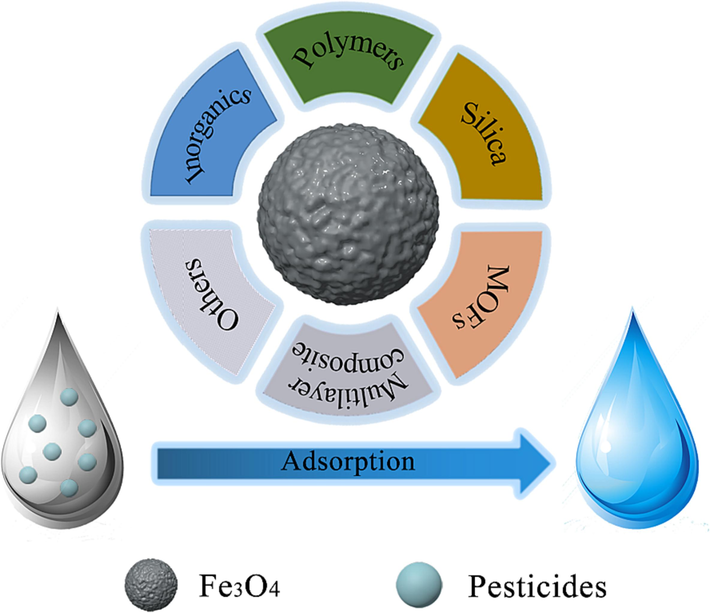
Construction of Fe3O4-based adsorbents by inorganics, polymers, silica, MOFs and multilayer composites.
2.1 Inorganics
Some inorganic substances, such as mud, graphene, and activated carbon, are frequently used to remove pesticides from the water environment. As traditional adsorbents, their low cost is the most prominent advantage, but these adsorbents are usually difficult to recover, which easily causes secondary pollution (Premarathna et al., 2019). The adsorption characteristics of Fe3O4-based adsorbents modified by inorganics for various pesticides removal is shown in Table 1. Boruah et al. prepared Fe3O4 by chemical co-precipitation method and synthesized Fe3O4 / reduced graphene oxide composites by in-situ solution chemistry method, which efficiently adsorbed five triazine pesticides (atrazine, prometryn, simazine, simeton, and ametryn) in aqueous solution with an effective adsorption rate of 93.61 % in 70 min due to the graphene’s aromatic ring (Boruah et al., 2017), although it takes a relative long time. Aydin synthesized magnetic Fe3O4/red mud nanoparticles for the adsorption of organophosphorus pesticides in water environments with high adsorption capacity (>180 µg/g) (Aydin, 2016). Since the adsorbent is constructed from red mud, which has not many groups to form a reversible interaction force, so it shows poor reusability. Herrera-García et al. extracted activated carbon from yam bark and combined it with Fe3O4 to obtain magnetic activated carbon (MAC), which has a high surface area for the removal of 2,4-dichlorophenoxyacetic (2,4-D) acid from water, but its adsorption capacity decreased by 40 % after five cycles, indicating relatively low reusability (Herrera-García et al., 2019). As mentioned in some of the above references, the Fe3O4 adsorbent obtained by inorganic modification has the advantages of low cost, simple preparation, and good stability. However, compared with other adsorbents, its dosage is large, and the most prominent problem is that the reusability is low, which may cause waste of resources to a certain extent. Therefore, using inorganic (mud, activated carbon, etc.) modified Fe3O4 nanoparticles to construct adsorbents has faded out to be the central hot spot in the research field. Kinetic model : pseudo-first-order (PFO), pseudo-second-order (PSO), and Elovich.Isotherm model : Langmuir (L), Freundlich (F), Temkin, and Sips.
Modifiers
Adsorbent
Pesticide
Adsorption temperature (°C)
Adsorption pH
Equilibrium time (min)
Adsorbent dosage (g/L)
Adsorption capacity (mg/g)
Kinetic model
Isotherm model
Mechanism
References
Inorganics
Fe3O4/graphene nanocomposite
Triazine pesticides
25
5
70
0.5
54.8
PSO
L
electrostatic interactions, π-π stacking interactions, hydrophobic interactions
(Boruah et al., 2017)
Magnetic diatomite (m-DE-OTMS)
DDT
25
/
30
4.0
120.0
PSO
L
hydrophobic interactions
(Erol et al., 2019)
Fe3O4/red mud nanoparticles
Organophosphorus pesticides
25
7 (diazinon, malathion, parathion) 3 (chlorpyrifos)
60
5.0
/
PSO
F
/
(Aydin 2016)
Graphene/Fe3O4 nanocomposite
Triazole fungicides
/
2–13
30
0.4
/
PSO
L
electrostatic interaction, hydrogen bonding, π-π stacking, hydrophobic interaction, cation- π interaction
(Wang et al., 2019b)
Amine-modified magnetic diatomite (m-DE-APTES)
endosulfan
25
/
60
4.0
97.2
/
L
/
(Alacabey 2022)
Algerian palygorskite modified with magnetic iron
linuron
20
/
480
2.5
1.70
PSO
F
/
(Belaroui et al., 2018)
Aminoguanidine-modified magnetic graphene oxide (AGu@mGO(R))
chlorpyrifos
/
6.5
30
1.0
85.47
PSO
F
hydrogen bonding, π-π interactions
(Mahdavi et al., 2021)
Magnetic graphene nanocomposite (graphene@SiO2@Fe3O4)
Organophosphorus pesticides
/
7
5
2.0
/
/
/
/
(Wang et al., 2018a)
Sugarcane bagasse (MBo) and peanut shell (MPSo) magnetic-composites
carbofuran, iprodione
/
7
720, 480 (MBo) 480,720 (MPSo)
2.0
175, 89.3 (MBo) 119, 2.76 (MPSo)
Elovich
Sips
/
(Toledo-Jaldin et al., 2020)
Activated carbon modified with Fe3O4
2,4-Dichlorophenoxyacetic Acid
25
2.8
50
2.0
79.05
PSO
L
/
(Herrera-García et al., 2019)
Bioadsorbent from Magnetic Activated Carbon Hybrid (AA@Fe3O4 NPs@AC)
acetamiprid
/
3
30
0.4
357.14
PSO
L
hydrogen bonding, electrostatic interactions, π-π interactions
(Nejadshafiee & Islami, 2020)
Magnetic polyethyleneimine functionalized reduced graphene oxide (Fe3O4@PEI-RGO)
Polar acidic herbicides
/
4
6.5
0.36
/
/
/
electrostatic interactions, π-π stacking interactions
(Li et al., 2017)
2.2 Polymers
Polymers such as chitosan (Yang et al., 2022), poly(indole-co-thiophene) (Ebrahimpour et al., 2017), polydopamine (Wang et al., 2019a) and polyaniline (Goswami & Mahanta, 2020) have abundant sources in nature and are inexpensive and nontoxic, with simple synthesis methods and a certain functionality and purpose compared to other materials. The affluent groups in the polymers allow for grafting modification on the surface of Fe3O4 particles and the possibility of adsorption by forming interactions with pollutants in water (Samaddar et al., 2019). The adsorption characteristics of Fe3O4-based adsorbents modified by polymers for various pesticides removal is shown in Table 2. Wang et al. coated Fe3O4 by polydopamine to prepare Fe3O4@PDA-DES, which could capture and adsorb sulfonylurea herbicides in aqueous solution by hydrogen bonding and π-π interactions within a short time (3 min) at low dosage (0.5 g/L) (Wang et al., 2019a). Polymers can indeed achieve an increase in adsorption capacity, but whether all polymers have the same effect. Goswami et al. prepared Fe3O4 by solvothermal method, and then coated the prepared nanoparticles with polyaniline (PANI) and polypyrrole (PPy) by chemical oxidative polymerization to prepare magnetic Fe3O4-PANI and Fe3O4-PPy for efficient adsorption of 2,4-D in aqueous solution. Fe3O4-PANI achieved an adsorption capacity of 60.97 mg/g for 2,4-D at 90 min, while Fe3O4-PPy achieved 96.15 mg/g for 2,4-D at 60 min (Goswami & Mahanta, 2020), indicating that polypyrrole-modified Fe3O4 can achieve better performance in the adsorption of 2,4-D. Due to the different properties and diverse structures of various polymers, the characteristic groups can interact with the target pesticide molecules, which means that the selection of polymers will directly affect the adsorption capacity. Therefore, it is necessary to select the most suitable polymer material according to the characteristics of the target pesticide molecules before constructing the adsorbent. Compared with the inorganic-based Fe3O4 adsorbent modified with mud and graphene, polymer-modified Fe3O4 adsorbents have the ability to orient easily, and there is more chemisorption to achieve efficient adsorption of pesticides during the adsorption process. However, the inherent characteristics of the polymer itself lead to poor dispersion of the adsorbent, which may be challenging and thus affect its use in practical scenarios and fail to achieve the expected performance. Kinetic model : pseudo-first-order (PFO), pseudo-second-order (PSO), and Elovich.Isotherm model : Langmuir (L), Freundlich (F), Temkin, and Sips.
Modifiers
Adsorbent
Pesticide
Adsorption temperature (°C)
Adsorption pH
Equilibrium time (min)
Adsorbent dosage (g/ L)
Adsorption capacity (mg/g)
Kinetic model
Isotherm model
Mechanism
References
Polymers
Magnetic sporopollenin supported cyanocalixarene (MSP-CyCalix)
chlorpyrifos, hexaconazole
/
6
30
1.0
13.88 12.34
PSO
L
electrostatic interaction, π-π interaction, hydrogen bonding,hydrophobic interaction
(Kamboh et al., 2021)
Magnetic molecularly imprinted polymers (MMIPs)
phosalone, diazinon, chlorpyrifos
25
/
60
1.0
196.07, 192.30, 172.41
PSO
L
/
(Masoumi et al., 2016)
Magnetic polydopamine modified with deep eutectic solvent (Fe3O4 @PDA-DES)
Sulfonylurea herbicides
/
3
15
0.5
/
/
/
hydrogen bonding, π-π interactions
(Wang et al., 2019a)
Matrix-dispersed magnetic molecularly-imprinted polyaniline (MMIP)
chlorpyrifos
25
7
200
20
1.77
PSO
Temkin
physico-chemical interaction
(Saad et al., 2021)
Polyaniline- Fe3O4 and polypyrrole- Fe3O4 magnetic nanocomposites
2, 4-dichlorophenoxyacetic acid
30
/
90 (Fe3O4-PANI) 60 (Fe3O4-PPy)
1.0
60.97 (Fe3O4-PANI) 96.15 (Fe3O4-PPy)
PSO
L
electrostatic interaction, hydrogen bonding, π-π interactions
(Goswami & Mahanta, 2020)
2.3 Silica (SiO2)
Silica (SiO2) is a mesoporous material with high stability, environmental friendliness, great specific surface area and good biocompatibility that is widely used in many fields, such as adsorption and controlled release of drugs, and is often combined with other materials, which can provide more adsorption channels for Fe3O4 and improve the adsorption capacity (Gao et al., 2020, Soltani et al., 2019). It is worth noting that by modifying SiO2 appropriately, such as adjusting its morphology and size, changing its surface charge, and functionalizing the surface, the dispersibility of SiO2 in water can be improved, which can overcome some problems caused by the strong magnetism of Fe3O4 nanoparticles. The adsorption characteristics of Fe3O4-based adsorbents modified by silica for various pesticides removal is shown in Table 3. Farmany et al. prepared Fe3O4 nanoparticles by chemical co-precipitation method, and then synthesized Fe3O4/SiO2 core–shell structure adsorbent by ultrasonic assisted method (Farmany et al., 2016). The adsorbent effectively adsorbs diazinon from water, which not only ensured the magnetic properties of the adsorbent and facilitated recovery but also improved the dispersibility of the adsorbent and avoided the agglomeration of Fe3O4 in water. Tian et al. prepared a multifunctional core–shell microsphere adsorbent by embedding bilayer SiO2 onto Fe3O4 nanoparticles using hydrothermal and impregnation methods. The inner nonporous silica layer served as a protective layer to prevent the magnet from being etched in the adsorption system. The outer layer of mesoporous silica with a high specific surface area and large pore volume ensured its powerful adsorption capacity and efficient adsorption of DDT in water (Tian et al., 2014). The mesoporous structure of SiO2 ensures that the adsorbent has large pores to adequately accommodate external contaminants, and in numerous adsorption applications, SiO2 is an ideal material for constructing adsorbents due to its strong compatibility and adjustability, which is often combined with other functional materials. However, the time-consuming nature of synthesizing SiO2 often limits its application in constructing adsorbents, so the development of a rapid synthesis method is necessary. Kinetic model : pseudo-first-order (PFO), pseudo-second-order (PSO), and Elovich.Isotherm model : Langmuir (L), Freundlich (F), Temkin, and Sips.
Modifiers
Adsorbent
Pesticide
Adsorption temperature (°C)
Adsorption pH
Equilibrium time (min)
Adsorbent dosage (g/ L)
Adsorption capacity (mg/g)
Kinetic model
Isotherm model
Mechanism
References
Silica
Benzenesulfonic acid functionalized core–shell Fe3O4@SiO2
Benzimidazole fungicides
/
3
3
3.33
/
/
/
electrostatic interaction
(Deng et al., 2013)
C18-functionalized ultrafine magnetic silica nanoparticles
Pyrethroid pesticides
/
/
10
3.0
/
/
/
/
(Jiang et al., 2013)
Magnetic mesoporous silica nanocomposite (MSNPs/Fe3O4)
lindane
25
7
360
1.8
/
/
/
/
(El-Said et al., 2017)
Fe3O4/SiO2-NH2 core/shell nanocrystals
diazinon
/
6
25
6.0
206.18
PSO
L
electrostatic interaction
(Farmany et al., 2016)
Multifunctional Fe3O4@nSiO2@mSiO2–Fe core–shell microspheres
DDT
25
/
60
1.25
/
/
/
/
(Tian et al., 2014)
2.4 Metal-organic frameworks (MOFs)
Metal-organic frameworks (MOFs) are a new class of materials that are currently being researched with great enthusiasm and are widely used in important fields such as hydrogen storage, adsorption and separation, drug mitigation, and catalysis (Do et al., 2022, Nguyen et al., 2020). MOFs are considered extremely efficient adsorbents in water environmental pollutant adsorption due to their abundant active sites, extraordinary stability, and excellent specific surface area (Do et al., 2021, Rojas et al., 2022). As a rich variety of functional materials, many researchers will choose the appropriate MOF to construct adsorbents according to their own needs, so as to obtain or enhance some functions. The adsorption characteristics of Fe3O4-based adsorbents modified by MOFs for various pesticides removal is shown in Table 4. Lu et al. firstly synthesized Fe3O4@MXene by co-precipitation method, and then prepared Fe3O4@MXene@MOF-74 by combining the oxygen-containing functional groups and zinc ions in Zn-MOF-74. Fe3O4@MXene@MOF-74 has excellent adsorption performance for triazole fungicides in actual paddy water samples (maximum adsorption capacity: epoxiconazole 120.96 mg/g, tebuconazole 108.26 mg/g), and at the same time, the adsorbent itself has an inhibitory effect on the rice pathogen (Magnaporthe grisea Barr.). This provides a novel concept for the practical application of bifunctional adsorbents (Lu et al., 2022). Li et al. constructed a bilayer MOF adsorbent (Fe3O4@ZIF-8@ZIF-67) through one-pot method and layer-by-layer self-assembly method, which showed high removal rates (>95 %) for fipronil and its metabolites in water and cucumber samples. The data rarely fit the bilayer Freundlich model rather than the monolayer Langmuir model, which may be due to the similar bilayer pore structure of the adsorbent (Li et al., 2020). Despite the widespread interest in MOFs for pesticide removal in water environments, their preparation is usually tedious, and functionalized MOFs and their derivatives are not currently combined with Fe3O4 to obtain more compact and efficient adsorbents. In addition, the adverse effects of MOFs, especially the metal ions they contain, on aquatic organisms have not been extensively studied thus far (Li et al., 2021), which is extremely relevant at the ecological and toxicological levels. Kinetic model : pseudo-first-order (PFO), pseudo-second-order (PSO), and Elovich.Isotherm model : Langmuir (L), Freundlich (F), Temkin, and Sips.
Modifiers
Adsorbent
Pesticide
Adsorption temperature (°C)
Adsorption pH
Equilibrium time (min)
Adsorbent dosage (g/ L)
Adsorption capacity (mg/g)
Kinetic model
Isotherm model
Mechanism
References
Metal-organic frameworks
Magnetic MXene based metal organic frameworks composites (Fe3O4@MXene@MOF-74)
epoxiconazole, tebuconazole
/
7
100
1.0
120.96, 108.26
PSO
L
hydrogen bonding, covalent bonding, π-π stacking interaction, electrostatic interaction, physical interaction
(Lu et al., 2022)
Double layer MOFs (M−ZIF−8@ZIF-67)
fipronil and its metabolites
/
6
45
3.75
/
PSO
F
/
(Li et al., 2020)
Magnetic metal–organic framework composite (Fe3O4/MIL-101 (Fe))
fenitrothion
/
7
60
1.0
58.14
PSO
L
electrostatic interaction, hydrogen bonding, π-π interactions
(Samadi-Maybodi & Nikou, 2021)
Fe3O4@ZnAl-LDH@MIL-53(Al)
Azole fungicides
/
6
5
30.0
43.54–71.79
PSO
L
hydrogen bonding, chemisorption, π-π interaction
(Lu et al., 2021a)
Covalent-organic frameworks
Fluffy ball-like magnetic covalent organic frameworks
Organophosphorus pesticides
25
/
/
0.5
163.9–178.6
PSO
F
π-π, C-H⋯π, C-H⋯S interactions
(Qi et al., 2022)
Magnetic covalent organic framework (Fe3O4@DOPA-Tp)
chlorpyrifos, atrazine
19
/
120
1.0
270, 54
/
F
van der Waals interactions, hydrogen bonding, hydrophobic interactions
(Romero et al., 2020)
2.5 Multilayer composite modification
Although the adsorbents constructed by modification of Fe3O4 with a single type of material do have better adsorption effects on pesticides, they are often limited in application due to the single type of modifier, such as a single type of target, unstable properties of the adsorbent, and single-factor limitations in application (Lin & Lee, 2020). Hence, a growing number of studies have focused on the multilayer composite modification of Fe3O4 to obtain multiple advantages of the adsorbent, optimize adsorption performance, expand its application scope and overcome the shortcomings in practical applications (Foroutan et al., 2022). The common combinations in current research include polymer/graphene (Liu et al., 2017), polymer/SiO2 (Mahpishanian et al., 2015), polymer/MOFs (Senosy et al., 2020), and SiO2/MOFs(Yang et al., 2018). The problems of monolayer-modified adsorbents can be solved by different combinations. The adsorption characteristics of Fe3O4-based adsorbents modified by multilayer composite for various pesticides removal is shown in Table 5. Lu et al. firstly coated a layered double hydroxide (LDHs) on Fe3O4 by co-precipitation method, and then covered a layer of MIL-53 (Al) on its basis by hydrothermal method (Lu et al., 2021a). The two modified materials selected in this study have a high number of active sites for azole fungicides, and once combined, the strong combination can make the adsorption achieve equilibrium in a short time (5 min), which is efficient and fast. In addition, many studies have taken into account the complementary advantages of different materials in the construction of multilayer composite adsorbents. Yang et al. constructed a novel smart adsorbent Fe3O4@SiO2@UiO-67 for the adsorption of the organophosphorus pesticide glyphosate, integrating the advantages of each component with a very high adsorption capacity (256.54 mg/g) (Yang et al., 2018). It is noteworthy that SiO2 plays a role in the adsorbent to protect the Fe3O4 cores, prevent external electron transfer and transition with Fe3O4, and facilitate dendritic modification, as demonstrated in numerous studies. The multilayer composite-modified Fe3O4 adsorbents enhance the performance of the adsorbent in many aspects, but it is a more cumbersome and costly synthesis process due to the variety of modified materials used. In addition, the compatibility between various materials sometimes limits the design and synthesis of the adsorbent, which are urgent problems for long-term practical applications in the future. Kinetic model : pseudo-first-order (PFO), pseudo-second-order (PSO), and Elovich.Isotherm model : Langmuir (L), Freundlich (F), Temkin, and Sips.
Modifiers
Adsorbent
Pesticide
Adsorption temperature (°C)
Adsorption pH
Equilibrium time (min)
Adsorbent dosage (g/L)
Adsorption capacity (mg/g)
Kinetic model
Isotherm model
Mechanism
References
Multilayer composite modification
New magnetic molecularly imprinted polymer nanocomposite (GO/Clay/Fe3O4@PDA MIP)
diazinon
/
7
16
1.24
1.17
PSO, Elovich
F
intraparticle and extraparticle diffusion
(Sohrabi et al., 2022)
Phenyl-modified magnetic graphene/mesoporous silica (MG-MS-Ph)
eight pesticides
25
/
/
2.0
/
PFO
F
/
(Wang et al., 2017)
Mixed hemimicelle SDS-coated magnetic chitosan nanoparticles (MHMS-MCNPs)
diazinon, phosalone, chlorpyrifos.
/
2.5
3
/
/
/
L
hydrophobic and electrostatic interactions
(Ranjbar Bandforuzi & Hadjmohammadi, 2019)
Post-modification of magnetic metal–organic frameworks with β-cyclodextrin (Fe3O4@MIL-100(Fe)/β-CD)
tebuconazole, flusilazole
25
7
50
1.0
64.52–102.10
PSO
L
acid-base interaction, π-complex formation, hydrogen bonding, electrostatic interaction
(Senosy et al., 2020)
Ionic-liquid-functionalized magnetic particles
Sulfonylurea herbicides
/
4
5
0.05
/
/
/
Hydrophobic interaction
(He et al., 2013)
Magnetic graphene oxide and carboxymethyl cellulose (MGOC)
chlorpyrifos
20
6
40
0.4
108.3
PSO
L
hydrogen bonding, surface complexation, π-π interactions
(Dolatabadi et al., 2022)
Superparamagnetic core–shells anchored onto graphene oxide grafted with phenylethyl amine (Fe3O4@SiO2@GO-PEA)
Organophosphorus pesticides
/
6
5
15
/
/
/
π-stacking interaction
(Mahpishanian et al., 2015)
Amino-functionalized Iron oxide inside SBA-15 (RED-Fe-NH2-SBA-15)
glyphosate
/
2.1
1440
5.7
/
/
/
/
(Fiorilli et al., 2017)
Mesoporous KIT-6-magnetite composite (Fe3O4@SiO2@KIT-6)
Pyrethroid pesticides
25
7
10
0.4
/
PSO
L
/
(Zhang et al., 2019a)
Magnetic zeolitic imidazolate framework-8@deep eutectic solvent (M−ZIF−8@DES)
Pyrethroid pesticides
/
6
15
1.6
/
/
hydrogen bonding
(Liu et al., 2019)
Fe3O4-graphene oxide (GO)-β-cyclodextrin (β-CD) nanocomposite
Neonicotinoid pesticide
/
/
30
0.05
1.77–2.96
PSO
L (thiacloprid) F (others)
/
(Liu et al., 2017)
Graphene oxide-based silica coated magnetic nanoparticles functionalized with 2-phenylethylamine (Fe3O4@ SiO2@GO)
chlorpyrifos, parathion, malathion
25
3–11
15
1.5
11.1 (chlorpyrifos) 10.6 (parathion) 10.9 (malathion)
PSO
Sips
hydrogen bonding, π-π interaction
(Wanjeri et al., 2018)
Covalently functionalized Fe3O4@SiO2-MWCNTs core–shell magnetic microspheres
pentachlorophenol
/
2.5
/
0.5
96.4
PSO
F
π-π electron-donor–acceptor interaction, hydrophobic interaction, electrostatic interaction
(Zhou et al., 2014)
Magnetic multi-walled carbon nanotubes @organic framework ZIF-8 (M−M−ZIF−8)
Organophosphorus pesticides
/
4
15
3.75
2.18–3.89
/
F
electronic sharing or exchange
(Liu et al., 2018b)
Magnetic chitosan/activated carbon@UiO-66 bio-nanocomposite
imidacloprid
/
6.5
20
0.6
25.2
PSO
L
electrostatic forces, hydrogen bonding, π-π and n-π interactions
(Motaghi et al., 2022)
Fe3O4@SiO2@UiO-67
glyphosate
25
/
60
0.03
256.54
PSO
L
chemisorption
(Yang et al., 2018)
Fe3O4@ZIF-8@SiO2 core–shell nanoparticles
fenvalerate, β-cyfluthrin, tetramethrin
25
7
40
0.25
316.23, 364.43, 258.69
PSO
Sips
π-π stacking interactions, hydrogen bonds, hydrophobic interactions, pore filling effects
(Meng et al., 2023)
Magnetic graphene oxide functionalized MOF-199 (M−MOF−199)
Triazole pesticides
/
/
15
5.0
3.24–3.74
/
/
hydrophobic interactions, hydrogen bonding, electrostatic interactions, π-π stacking interactions
(Liu et al., 2018a)
Ternary magnetic Fe3O4@C3N4@covalent organic framework (COFs)
Organophosphorus pesticides
/
8
/
7.5
/
PSO
L
/
(Gao et al., 2022)
Metal organic framework functionalized magnetic lignosulfonate (Fe3O4@LS@ZIF-8)
Azole fungicides
/
8
50 (tebuconazole) 160 (imazalil)
20
25.10 (tebuconazole) 100.33 (imazalil)
PSO
L
covalent bonding, π-π interaction, hydrogen bonding
(Lu et al., 2021b)
Ternary metal–organic framework/ multi-walled carbon nanotube/iron oxide nanocomposite (Fe3O4–MWCNT–ZIF67)
butachlor
/
4.5
95
1.4
3.53
PSO
Temkin
/
(Azizzadeh et al., 2022)
3 Factors affecting Fe3O4-based adsorbents for the adsorption of pesticides
The adsorption of pesticides in the water environment is a complex multistep process involving the diffusion and interaction of pesticide molecules on the surface and inside the adsorbent (Soltani et al., 2021). This process is influenced by the following parameters: adsorbent dosage (Liu et al., 2018a), adsorption time (Dinari et al., 2020), temperature (Saad et al., 2021), pH (Wang et al., 2020), external ionic strength (Fu et al., 2023), and natural organic matter (Boruah et al., 2017), which determine the practical application of adsorbents in the remediation of water environments with various contaminants, including pesticides. Fig. 2 shows the factors affecting the adsorption performance of Fe3O4-based adsorbents for pesticides.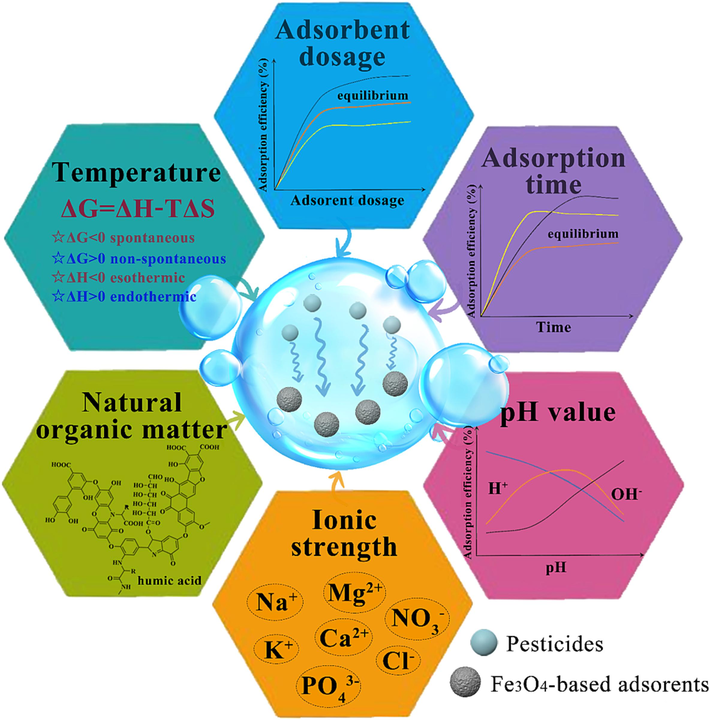
Factors affecting the removal of pesticides from water by Fe3O4-based adsorbents.
3.1 Adsorbent dosage
The solid-to-liquid ratio is an extremely critical factor affecting the adsorption process, which is essentially the adsorbent dosage put into the reaction system. In general, when the adsorbent concentration, pH, temperature and other conditions are the same, the smaller the amount of adsorbent, the higher the adsorption capacity (Morin-Crini et al., 2018). Therefore, it is always desirable to construct an adsorbent that effectively removes the target pollutant with a minimum dosage, thus reducing the purification cost and making magnetic adsorbent recovery more convenient. In general, during the initial stage of adsorption, the adsorption of the target pollutant increases with increasing adsorbent dose and finally reaches equilibrium, which is attributed to the abundance of active sites on the surface of the adsorbent. For example, Wanjeri et al. investigated the effect of an adsorbent (Fe3O4@SiO2@GO) on the adsorption of organophosphorus pesticides at different doses from 2 to 40 mg in a reaction system with a volume of 10 mL and observed that the maximum adsorption was reached at an adsorbent dosage of 15 mg (1.5 g/L) (Wanjeri et al., 2018). When the dosage exceeded 1.5 g/L, the adsorption reached equilibrium as the interactions between the adsorbent surface and organophosphorus pesticide molecules stabilized. After this point, the increase in adsorbent dosage could not improve the adsorption effect and even inhibited adsorption, probably due to the decrease in specific surface area and exposure of active sites caused by the aggregation of excess adsorbent in the water environment. Therefore, in all adsorption operations, the most appropriate adsorbent dosage is always selected because too much or too little cannot obtain the best adsorption effect; of course, as small of an adsorbent dosage as possible to save costs is always desired.
3.2 Adsorption time
Adsorption is a time-dependent process that will reach equilibrium after a certain time because the number of reactants in the system is finite. For the adsorption process, the shorter the time it takes to remove a certain amount of pollutant is, the better the performance of the adsorbent. A long adsorption time may lead to a waste of resources and excessive consumption of energy. Thus, the time taken for adsorption is a significant indicator of the performance of an adsorbent (Sajid et al., 2018). Typically, it can be observed that in the initial stage of adsorption, the large pollutant concentration difference between the adsorbent surface and the internal and external water environment, the large adsorption driving force, and the fact that the adsorbent still possesses an abundance of active sites lead to rapid binding of the pesticide to the adsorbent and a rapid increase in the adsorption rate (Cao et al., 2017). For example, Mahdavi et al. studied the adsorption of chlorpyrifos by the magnetic adsorbent AGu@mGO(R) in 5–50 min. The adsorption rate of chlorpyrifos gradually increased in the first 30 min, and the adsorbent had not reached saturation at this stage. After 30 min, the adsorption rate tended to equilibrate or even slightly decrease due to the lack of remaining adsorption sites, decrease in adsorption driving force, and partial desorption of chlorpyrifos (Mahdavi et al., 2021). In addition, Lu et al. synthesized Fe3O4@LS@ZIF-8 for the adsorption of azole fungicides (tebuconazole and imazalil), and the adsorption of tebuconazole reached equilibrium after 50 min, while the adsorption of imazalil required 160 min (Lu et al., 2021b). Different pesticides have different structures and characteristic groups, thus differing in their rate of interaction with the active site of the adsorbent, which leads to a difference in the equilibrium time. In practical applications, the time point of adsorption equilibrium needs to be explored in advance to ensure that the maximum adsorption effect is obtained and no resources are wasted.
3.3 Temperature
At the thermodynamic aspect, the adsorption process also involves heat transfer, and the temperature determines the speed and even the nature of the reaction between the surface of the adsorbent and adsorbate, whether exothermic or endothermic (Mehta et al., 2022, Soltani et al., 2020). If a certain adsorption process is endothermic, the adsorption capacity of the adsorbent may increase with increasing temperature, probably because of the increased interaction between the surface of the adsorbent and the adsorbate, while the exothermic adsorption process is the exact opposite. Zhou et al. investigated the thermodynamic behavior of covalently functionalized Fe3O4@SiO2-MWCNTs core–shell magnetic microspheres for pentachlorophenol (PCP) adsorption in the range of 288–318 K. The results show that the adsorption capacity of PCP increased with increasing temperature, indicating an endothermic adsorption process. At the same time, the standard enthalpy (ΔHθ) and standard entropy (ΔSθ) of the adsorption process of PCP in water are negative so that the standard free energy (ΔGθ) was also negative, indicating that the adsorption reaction was spontaneous and decreased spontaneously with increasing temperature (Zhou et al., 2014). The same pattern was found by Saad et al., who found that the adsorption of matrix-dispersed superparamagnetic iron oxide nanoparticles (SPIONs) on chlorpyrifos is also an endothermic and spontaneous process. It is worth mentioning that ΔGθ moves in the positive direction when the temperature is higher than 318 K. Therefore, room temperature (25 °C) is most favorable for adsorption (Saad et al., 2021). In practical industrial applications, for energy consumption reasons, adsorption at room temperature is the most economical and desired condition, which requires a precise understanding of the designed adsorbent and the target pesticide properties.
3.4 pH
The initial pH of the system is an essential factor affecting adsorption, which not only determines the form of adsorbate present but also influences the surface charge of the adsorbent (Wang et al., 2020), thus significantly enhancing or inhibiting the adsorption effect, which determines the strength of the interaction (Bahrami et al., 2018). In research studies on the removal of triazole pesticides from water by a magnetic adsorbent compounded with β-cyclodextrin and metal organic frameworks (Fe3O4@MIL-100(Fe)/β-CD), the pH of the reaction system was adjusted to 3–10 by adding appropriate amounts of 0.1 M HCl or 0.1 M NaOH, and the best adsorption effect was finally determined when the solution was neutral. It was observed that as the pH gradually increased from 3, the charge between the adsorbent and the pesticide gradually became the same on opposite sides, resulting in electrostatic repulsion, so the adsorption efficiency decreased. However, the adsorption capacity gradually increased after a pH of 6, peaked at 7, and then equilibrated, indicating that electrostatic interaction is not the main adsorption mechanism (Senosy et al., 2020). However, Wang et al. found that the change in pH had no significant effect on the adsorption of tebuconazole by graphene/Fe3O4. This was attributed to the fact that tebuconazole has two pKa values, electrostatic interactions played no major role, and pH adjustment did not affect the adsorption process via hydrogen bonding and π-π interactions (Wang et al., 2019b). Therefore, the different surface groups of adsorbents and pesticides may be affected differently at different pH values, including changes in the surface charge of adsorbents and ionization of pesticide molecules; thus, adsorbents with a wide range of pH adaptability are worth developing.
3.5 Ionic strength and natural organic matter
The number of adsorption sites available on the adsorbent surface is limited; unfortunately, metal ions and natural organic matter (NOM) in the external water environment may inhibit or promote the binding of target adsorbates and adsorption sites, thus directly or indirectly affecting the removal efficiency of the target contaminant (Zhao et al., 2019). In the adsorption system, as the amount of salt addition increases, the viscosity of the solution increases, preventing the diffusion of pesticide molecules, while the solubility of the pesticide in aqueous solution decreases as the ionic strength increases due to salt precipitation (Zhang et al., 2019b). The adsorption of Fe3O4@PEI-RGO on polar acidic herbicides inhibits the adsorption efficiency due to the addition of NaCl, which may also be due to the ion exchange between NaCl and the adsorbent, which inhibits the electrostatic interaction and occupies the adsorption site of the herbicide (Li et al., 2017). The ionic strength affects the electric potential at the interface, which is external to the adsorbent (external ions often occupy active sites on the adsorbent surface), thus affecting the strength of the adsorbent-pesticide binding. The mechanism of the effect of NOM such as humic acid on adsorption is more complex, as it can change the nature of the adsorbent and improve its dispersion and stability in the water column, thus improving the adsorption performance. In the study of ametryn adsorption by Fe3O4/rGO, the adsorption performance was improved by adding humic acid to the reaction system, which may be attributed to the formation of hydrogen bonds promoted by the presence of NOM (Boruah et al., 2017). NOM can transfer a negative surface charge to the adsorbent and change its adsorption behavior (Karimi Pasandideh et al., 2016), so it is necessary to study the specific effect of NOM on specific adsorbents with a wide range of prospects. In general, the presence of salt ions (including K+, Na+, Ca2+, Mg2+, Cl-, NO3–, SO42-, PO43-, etc.) in the solution increases the viscosity of the solution and causes salt precipitation, which inhibits the adsorption process. In contrast, the presence of NOM, such as humic substances in solution, promotes the adsorption process and increases the adsorption capacity. Therefore, the choice of the external environment will directly affect the adsorption.
In summary, any adsorption process is influenced by a mixture of factors, and the best adsorption performance can be obtained by adjusting multiple factors to achieve an optimal combination. The construction of a low dosage and high efficiency adsorbent that does not require adjustment of system pH, is not affected by external ions, and can exhibit excellent performance at room temperature has been the goal of multiple research studies.
4 Mechanism of pesticide adsorption
As a mass transfer process, adsorption is essentially an interaction between the interface of the adsorbent and the adsorbate (Liu et al., 2021b). Due to some special groups on the surface of the adsorbent and the target adsorbate, as well as the suitability of the pore channels on the surface of the adsorbent to the size of the target molecules, there are some adsorption mechanisms in the adsorption process, including van der Waals interactions, hydrogen bonding, electrostatic interactions, π-π interactions, and hydrophobic interactions, as shown in Fig. 3. These mechanisms have been currently used to explain the Fe3O4-based adsorbent adsorption of organic pollutants, including pesticides, in water environments. It is worth noting that Fe3O4 particles do not have abundant groups on their surface (providing magnetic properties only to the adsorbent), so the above adsorption mechanisms often occur between the modifier and the pesticide.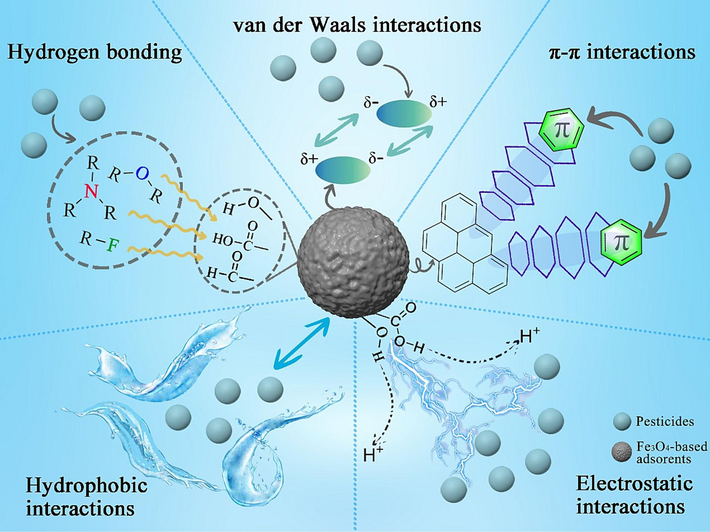
Based on van der Waals interactions, hydrogen bonding, electrostatic interactions, π-π interactions and hydrophobic interactions, the adsorption mechanism of Fe3O4-based adsorbents for pesticides.
4.1 van der Waals interactions
van der Waals interactions, also known as intermolecular forces, include orientation, induction and dispersion forces, which are generated by the polarity of the adsorbent and the pesticide (Huang et al., 2020). Romero et al. demonstrated through model calculations that van der Waals interactions are the main driving force for the adsorption of atrazine and chlorpyrifos by the magnetic covalent organic framework and that van der Waals interactions are always present between the adsorbent and the pesticide molecules at different positions of the simulation, accompanied by changes in intensity (Romero et al., 2020). However, Wanjeri et al. found that van der Waals interactions may lead to lower theoretical values of adsorbent specific surface area, which is unavoidable (Wanjeri et al., 2018). The same problem was found by Dolatabadi et al., as well as in the presence of graphene (GO) (Dolatabadi et al., 2022). For adsorbents with high specific surface area and porosity, van der Waals interactions are extremely prevalent and unavoidable and, in most cases, can be considered a complementary mechanism to explain the observed results.
4.2 Hydrogen bonding
Hydrogen bonding is traditionally defined as occurring between the donor hydrogen atom and the electronegative acceptor atom with lone electron pairs (Brinkley & Gupta, 2004). Since the formation of hydrogen bonds requires the presence of H-acceptor and H-donor sites on the adsorbent and adsorbate, this action is not affected by external factors such as pH (Samadi et al., 2021). Romero et al. developed two models, A and B, for atrazine and chlorpyrifos by theoretical calculations to investigate the adsorption mechanism when using magnetic covalent organic skeletons for the removal of endocrine-disrupting pesticides. The number of hydrogen bonds was statistically analyzed in 40 snapshots, and in the model for atrazine, a higher number of hydrogen bonds (average 3) was observed. In contrast, for chlorpyrifos, only one hydrogen bond was found on average. The reason for this result is that pesticide molecules have different structures and contain different elements, namely, different numbers of H donors or acceptors, which can result in different adsorption amounts of different pesticides (Romero et al., 2020). The degree of strength of hydrogen bonding is related to the number of H donors and acceptors; thus, when designing and constructing adsorbents, the introduction of sufficient H-acceptors or donors to form a large number of hydrogen bonds during the adsorption process is a powerful way to enhance the performance of adsorbents, and this is an idea to be considered for the design of special adsorbents in the future.
4.3 Electrostatic interactions
During adsorption, electrostatic interactions (including electrostatic repulsion and electrostatic gravitational forces) are often observed when the adsorbent and adsorbate have surface charges, which are frequently related to the pH of the adsorption system (Shi et al., 2022). For example, the zeta potential value of the adsorbent Fe3O4/MIL-101(Fe) is 8. When the pH of the system is greater than 8, both the adsorbent and the adsorbate mass (fenitrothion) are negatively charged, whereas when the pH decreases below 6, both the adsorbent and the adsorbate are positively charged. Both cases have a large electrostatic repulsion, which is not favorable for adsorption. When the pH is neutral, the adsorbent surface is negatively charged, fenitrothion is in the form of amphoteric ions, and the electrostatic gravitational force dominates, showing a strong electrostatic interaction (Samadi-Maybodi & Nikou, 2021). As shown in Table 1, although many adsorbents can be used directly without pH adjustment because the reaction system is neutral when applied specifically to pesticide adsorption, some adsorbents still require special pH adjustment when used, which obviously cannot exclude the influence of the nature of the pesticide itself. To fully form electrostatic interactions in the adsorption process to improve the adsorption performance, there are special requirements for the modifiers used in the design of adsorbents; the surface charge should be as opposite as possible to that of the pesticide molecules, and their zeta potential should match the adsorption conditions.
4.4 π-π interactions
When π-electrons are present in both the adsorbent and the pesticide molecule, more specifically, when the presence of aromatic rings is needed, π-π interactions can be observed during the adsorption process (Igwegbe et al., 2021). Due to the specificity required for the adsorbent structure, π-π interactions are often found in materials such as graphene and metal–organic frameworks. Lu et al. analyzed the adsorption behavior of Fe3O4@MXene@MOF-74 for triazole fungicides by FT-IR and XPS. The FT-IR analysis showed that the aromatic C–C and C–H characteristic peaks almost disappeared after adsorption, and XPS analysis showed that the C = O and C = C bonds of Fe3O4@MXene@MOF-74 and the intensity of the triazole fungicides’ aromatic rings was decreased, and these characterization results indicated proved the existence and action of π-π interactions (Lu et al., 2022). In addition, Lu et al. reached the same conclusion by XPS analysis in a study using Fe3O4@ZnAl-LDH@MIL-53(Al) for the adsorption of azole fungicides (Lu et al., 2021a). It is noteworthy that the AGu@mGO(R) nanoadsorbent has a wide adaptability to pH changes (3–11) when applied for the adsorption of chlorpyrifos due to the strong π-π interactions formed between chlorpyrifos molecules in the rich aromatic ring structure domain on the adsorbent surface (Mahdavi et al., 2021). Although π-π interactions are not the dominant adsorption mechanism in some adsorption processes, they are more common for some adsorbents and pesticides that possess electron-donating and electron-withdrawing functional groups.
4.5 Hydrophobic interactions
The hydrophobicity of a substance is closely related to its solubility in water and octanol–water partition coefficient (Kow), while most pesticides have lower solubility in water and larger Kow values, hence their high hydrophobicity (Xu et al., 2020). Hydrophobic interactions are also an important mechanism for the adsorption process between adsorbents and liposoluble pesticides if the adsorbents are also strongly hydrophobic. Erol et al. modified the strongly hydrophobic ligand octadecyltrimethoxysilane (OTMS) onto magnetic diatomaceous earth (m-DE) to obtain the strongly hydrophobic adsorbent m-DE-OTMS, which formed a strong hydrophobic interaction with the equally strongly hydrophobic pesticide DDT in an water environment and achieved efficient adsorption (Erol et al., 2019). This work provides a reference for the targeted design of functional adsorbents. In addition, some MOFs also show hydrophobic properties, so some adsorbents with MOF structures will also have hydrophobic interactions with pesticides. For example, Liu et al. prepared a magnetic copper-based metal organic framework (M−MOF−199), which had a better adsorption effect due to the hydrophobicity of both MOF-199 and triazole pesticides (Liu et al., 2018a). Due to the specificity of the formation conditions of hydrophobic interactions, they are currently not very common in adsorption mechanisms. Nevertheless, the current trend suggests that a large number of hydrophobic materials, such as MOFs, are increasingly used to construct adsorbents (Zhang & Snurr, 2017), and hydrophobic interactions will increasingly appear in adsorption systems to explain the adsorption mechanism.
Based on the above discussion, it can be concluded that the adsorption of pesticides is a complex and interesting process that involves numerous different adsorption mechanisms. In actual adsorption processes, it is not possible to explain it by just one single mechanism; alternatively, a combination of a series of mechanisms is a more appropriate explanation of the observed results.
5 Future directions
Currently, the Fe3O4-based adsorbents constructed by a single modifier are limited by single performance and can no longer meet the high requirements of practical applications. Considering the two development directions of pesticide adsorption specificity and universality, composite multilayer modifications have become a research trend, and various modifiers with different properties can be combined in various ways to purposefully construct multifunctional high-performance magnetic adsorbents. Otherwise a more environmentally friendly approach can be used for the construction of sustainable Fe3O4-based adsorbents, and toxicity assessments, including the potential impact of the adsorbent on ecosystems (especially aquatic environments) and human health, need to be completed to ensure their safety for practical application in water environments.
-
Most of the current studies on adsorption have been carried out on a small scale in the laboratory, and although adsorbent performance on pesticides has been demonstrated, further industrial studies investigating their performance in real complex effluents are lacking.
-
The dispersion stability of the vast majority of adsorbents in the water environment was not considered, especially the addition of Fe3O4. The magnetic properties brought about can also lead to agglomeration of the adsorbent in water, which affects the adsorption performance, and modifiers that can improve the dispersibility are preferred.
-
Some of the modifiers for constructing Fe3O4-based adsorbents have excellent physicochemical properties, but they have some irresistible limitations, such as the time-consuming process of SiO2 preparation, complicated synthesis of MOFs and high cost. More facile preparation approaches and inexpensive materials must be explored in the future.
-
To date, a large number of studies have shown that the adsorption performance can be affected by many external factors, and almost all adsorption studies involve the optimization of adsorption parameters. In practical applications, it is also necessary to consider the influence of various factors to maximize the adsorption effect as much as possible. The development of adsorbents with a wide range of applicability and less susceptible to external factors has always been desired since large-scale adjustment of the reaction system is unrealistic.
-
In addition, to avoid secondary contamination, the next stage after the adsorption of pesticides by Fe3O4-based adsorbents, that is, the possibility of recovery and reuse of the adsorbed substances, is still a completely unexplored research area, which has far-reaching implications for the full utilization of resources.
6 Conclusion
This review attempts to cover the Fe3O4-based adsorbents used for pesticide adsorption in water environments in the last decade. Summarizing the work of previous scholars, it was found that most Fe3O4-based adsorbents could achieve more than 90 % adsorption efficiency for pesticides in water. In general, it is still focused on improving the adsorbent performance (namely, increasing the adsorption capacity). The adsorption of pesticides in water environment is a complex and interesting process, which is affected by many factors and accompanied by a complex mechanism. We believe that using Fe3O4-based adsorbents to remove pesticides in water is an important and effective approach to reducing environmental pollution.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Cu–BTC@cotton composite: design and removal of ethion insecticide from water. RSC Adv.. 2016;6:42324-42333.
- [CrossRef] [Google Scholar]
- Endosulfan elimination using amine-modified magnetic diatomite as an adsorbent. Front. Chem.. 2022;10:907302
- [CrossRef] [Google Scholar]
- Removal of organophosphorus pesticides from aqueous solution by magnetic Fe3O4/red mud-nanoparticles. Water Environ. Res. 2016;88:2275-2284.
- [CrossRef] [Google Scholar]
- Ternary metal-organic framework/multi-walled carbon nanotube/iron oxide nanocomposite for removal of butachlor pesticide. Environ. Sci. Eur.. 2022;34:49.
- [CrossRef] [Google Scholar]
- Adsorption of 2,4-dichlorophenoxyacetic acid using rice husk biochar, granular activated carbon, and multi-walled carbon nanotubes in a fixed bed column system. Water Sci. Technol.. 2018;78:1812-1821.
- [CrossRef] [Google Scholar]
- Adsorption of linuron by an Algerian palygorskite modified with magnetic iron. Appl. Clay Sci.. 2018;164:26-33.
- [CrossRef] [Google Scholar]
- Magnetically recoverable Fe3O4/graphene nanocomposite towards efficient removal of triazine pesticides from aqueous solution: Investigation of the adsorption phenomenon and specific ion effect. Chemosphere. 2017;168:1058-1067.
- [CrossRef] [Google Scholar]
- Tuning adsorption duration to control the diffusion of a nanoparticle in adsorbing polymers. J. Phys. Chem. Lett.. 2017;8:2629-2633.
- [CrossRef] [Google Scholar]
- Removal of urban-use insecticides in a large-scale constructed wetland. Environ. Pollut.. 2021;268:115586
- [CrossRef] [Google Scholar]
- de OGH, Menezes JMC, da Costa JGM, Coutinho HDM, Teixeira RNP, do Nascimento RF. 2020. A socio-environmental perspective on pesticide use and food production. Ecotoxicol. Environ. Saf. 197, 110627. 10.1016/j.ecoenv.2020.110627.
- Nanopesticides: A systematic review of their prospects with special reference to tea pest management. Front. Nutr.. 2021;8:686131
- [CrossRef] [Google Scholar]
- Metal-organic framework@carbon hybrid magnetic material as an efficient adsorbent for pollutant extraction. ACS Appl. Mater. Interfaces. 2020;12:6419-6425.
- [CrossRef] [Google Scholar]
- Water pollution control for sustainable development. Engineering. 2019;5:839-840.
- [CrossRef] [Google Scholar]
- Rapid and selective determination of trace benzimidazole fungicides in fruit juices by magnetic solid-phase extraction coupled with high-performance liquid chromatography-fluorescence detection. Food Anal. Methods. 2013;6:1576-1582.
- [CrossRef] [Google Scholar]
- Dinari M, Atabaki F, Pahnavar Z, Soltani R. 2020. Adsorptive removal properties of bivalent cadmium from aqueous solution using porous poly(N-2-methyl-4-nitrophenyl maleimide-maleic anhydride-methyl methacrylate) terpolymers. Journal of Environmental Chemical Engineering 810.1016/j.jece.2020.104560.
- Do HH, Le QV, Tekalgne MA, Tran AV, Lee TH, Hong SH, Han SM, Ahn SH, Kim YJ, Jang HW, Kim SY. 2021. Metal–organic framework-derived MoSx composites as efficient electrocatalysts for hydrogen evolution reaction. J. Alloys Compd. 85210.1016/j.jallcom.2020.156952.
- Metal-organic-framework based catalyst for hydrogen production: Progress and perspectives. Int. J. Hydrogen Energy. 2022;47:37552-37568.
- [CrossRef] [Google Scholar]
- Adsorption characteristics in the removal of chlorpyrifos from groundwater using magnetic graphene oxide and carboxy methyl cellulose composite. Sep. Purif. Technol.. 2022;300:121919
- [CrossRef] [Google Scholar]
- Poly (indole-co-thiophene)@Fe3O4 as novel adsorbents for the extraction of aniline derivatives from water samples. Microchem. J.. 2017;131:174-181.
- [CrossRef] [Google Scholar]
- Green synthesis of magnetic mesoporous silica nanocomposite and its adsorptive performance against organochlorine pesticides. Int. J. Environ. Sci. Technol. (Tehran). 2017;15:1731-1744.
- [CrossRef] [Google Scholar]
- Magnetic diatomite for pesticide removal from aqueous solution via hydrophobic interactions. Environ. Sci. Pollut. Res. Int.. 2019;26:33631-33641.
- [CrossRef] [Google Scholar]
- Agricultural water pollution: key knowledge gaps and research needs. Curr. Opin. Environ. Sustain.. 2019;36:20-27.
- [CrossRef] [Google Scholar]
- Ultrasond-assisted synthesis of Fe3O4/SiO2 core/shell with enhanced adsorption capacity for diazinon removal. J. Magn. Magn. Mater.. 2016;416:75-80.
- [CrossRef] [Google Scholar]
- Iron oxide inside SBA-15 modified with amino groups as reusable adsorbent for highly efficient removal of glyphosate from water. Appl. Surf. Sci.. 2017;411:457-465.
- [CrossRef] [Google Scholar]
- Cadmium ion removal from aqueous media using banana peel biochar/Fe3O4/ZIF-67. Environ. Res.. 2022;211:113020
- [CrossRef] [Google Scholar]
- Novel phenylalanine-modified magnetic ferroferric oxide nanoparticles for ciprofloxacin removal from aqueous solution. J. Colloid Interface Sci.. 2023;632:345-356.
- [CrossRef] [Google Scholar]
- A bioresponsive system based on mesoporous organosilica nanoparticles for smart delivery of fungicide in response to pathogen presence. ACS Sustain. Chem. Eng.. 2020;8:5716-5723.
- [CrossRef] [Google Scholar]
- Ternary magnetic Fe3O4@C3N4@covalent organic framework for facile extraction and determination of organophosphorus pesticides in fruit. Microchem. J.. 2022;174:107103
- [CrossRef] [Google Scholar]
- Facile fabrication of functionalized core-shell Fe3O4@SiO2@Pd microspheres by urea-assisted hydrothermal route and their application in the reduction of nitro compounds. Environ. Nanotechnol. Monit. Manage.. 2019;11:100220
- [CrossRef] [Google Scholar]
- Polyaniline-Fe3O4 and polypyrrole-Fe3O4 magnetic nanocomposites for removal of 2, 4-dichlorophenoxyacetic acid from aqueous medium. J. Environ. Chem. Eng.. 2020;8:103919
- [CrossRef] [Google Scholar]
- Ionic-liquid-functionalized magnetic particles as an adsorbent for the magnetic SPE of sulfonylurea herbicides in environmental water samples. J. Sep. Sci.. 2013;36:3226-3233.
- [CrossRef] [Google Scholar]
- Activated carbon from yam peels modified with Fe3O4 for removal of 2,4-dichlorophenoxyacetic acid in aqueous solution. Water. 2019;11:2342.
- [CrossRef] [Google Scholar]
- Stronger adsorption of phosphorothioate DNA oligonucleotides on graphene oxide by van der Waals forces. Langmuir. 2020;36:13708-13715.
- [CrossRef] [Google Scholar]
- Adsorption of persistent organic pollutants (POPs) from the aqueous environment by nano-adsorbents: A review. Environ. Res.. 2022;212:113123
- [CrossRef] [Google Scholar]
- Adsorption of ciprofloxacin from water: A comprehensive review. J. Ind. Eng. Chem.. 2021;93:57-77.
- [CrossRef] [Google Scholar]
- Water purification via novel nano-adsorbents and their regeneration strategies. Process Saf. Environ. Prot.. 2021;152:441-454.
- [CrossRef] [Google Scholar]
- Liquid-solid extraction coupled with magnetic solid-phase extraction for determination of pyrethroid residues in vegetable samples by ultra fast liquid chromatography. Talanta. 2013;114:167-175.
- [CrossRef] [Google Scholar]
- Green sporopollenin supported cyanocalixarene based magnetic adsorbent for pesticides removal from water: Kinetic and equilibrium studies. Environ. Res.. 2021;201:111588
- [CrossRef] [Google Scholar]
- Silica-coated magnetite nanoparticles core-shell spheres (Fe(3)O(4)@SiO(2)) for natural organic matter removal. J. Environ. Health Sci. Eng.. 2016;14:21.
- [CrossRef] [Google Scholar]
- A global systematic review, meta-analysis and health risk assessment on the quantity of Malathion, Diazinon and Chlorpyrifos in Vegetables. Chemosphere. 2021;270:129382
- [CrossRef] [Google Scholar]
- Adsorption of phenol on environmentally friendly Fe3O4/ chitosan/ zeolitic imidazolate framework-8 nanocomposite: Optimization by experimental design methodology. J. Mol. Liq.. 2021;323:115064
- [CrossRef] [Google Scholar]
- Application of organic-inorganic hybrid composite particle for removal of heavy metal ions from aqueous solution and its toxicity evaluation. Eur. Polym. J.. 2017;95:335-347.
- [CrossRef] [Google Scholar]
- Magnetic polyethyleneimine functionalized reduced graphene oxide as a novel magnetic solid-phase extraction adsorbent for the determination of polar acidic herbicides in rice. Anal. Chim. Acta. 2017;949:23-34.
- [CrossRef] [Google Scholar]
- Double layer MOFs M-ZIF-8@ZIF-67: The adsorption capacity and removal mechanism of fipronil and its metabolites from environmental water and cucumber samples. J. Adv. Res.. 2020;24:159-166.
- [CrossRef] [Google Scholar]
- Photocatalytic degradation of hazardous organic pollutants in water by Fe-MOFs and their composites: A review. J. Environ. Chem. Eng.. 2021;9:105967
- [CrossRef] [Google Scholar]
- Adsorption of ciprofloxacin in water using Fe3O4 nanoparticles formed at low temperature and high reactant concentrations in a rotating packed bed with co-precipitation. Mater. Chem. Phys.. 2020;240:122049
- [CrossRef] [Google Scholar]
- Recent advances in MOF-derived carbon-based nanomaterials for environmental applications in adsorption and catalytic degradation. Chem. Eng. J.. 2022;427:131503
- [CrossRef] [Google Scholar]
- Magnetic solid-phase extraction of pyrethroid pesticides from environmental water samples using deep eutectic solvent-type surfactant modified magnetic zeolitic imidazolate framework-8. Molecules. 2019;24:4038.
- [CrossRef] [Google Scholar]
- Metal-organic framework preparation using magnetic graphene oxide-beta-cyclodextrin for neonicotinoid pesticide adsorption and removal. Carbohydr. Polym.. 2017;175:584-591.
- [CrossRef] [Google Scholar]
- Determination of triazole pesticides in aqueous solution based on magnetic graphene oxide functionalized MOF-199 as solid phase extraction sorbents. Micropor. Mesopor. Mater.. 2018;270:258-264.
- [CrossRef] [Google Scholar]
- Adsorption and removal of organophosphorus pesticides from environmental water and soil samples by using magnetic multi-walled carbon nanotubes @ organic framework ZIF-8. J. Mater. Sci.. 2018;53:10772-10783.
- [CrossRef] [Google Scholar]
- Orderly porous covalent organic frameworks-based materials: Superior adsorbents for pollutants removal from aqueous solutions. Innovation (Camb). 2021;2:100076
- [CrossRef] [Google Scholar]
- Analysis of the mechanism for enhanced pyrene biodegradation based on the interactions between iron-ions and Rhodococcus ruber strain L9. Ecotoxicol. Environ. Saf.. 2021;225:112789
- [CrossRef] [Google Scholar]
- Synthesis and adsorption properties investigation of Fe3O4@ZnAl-LDH@MIL-53(Al) for azole fungicides removal from environmental water. Sep. Purif. Technol.. 2021;276:119282
- [CrossRef] [Google Scholar]
- Enhanced removal efficiency towards azole fungicides from environmental water using a metal organic framework functionalized magnetic lignosulfonate. Sep. Purif. Technol.. 2021;279:119785
- [CrossRef] [Google Scholar]
- Magnetic MXene based metal organic frameworks composites: Synthesis, characterization and application. J. Environ. Chem. Eng.. 2022;10:108037
- [CrossRef] [Google Scholar]
- Aminoguanidine modified magnetic graphene oxide as a robust nanoadsorbent for efficient removal and extraction of chlorpyrifos residue from water. J. Environ. Chem. Eng.. 2021;9:106117
- [CrossRef] [Google Scholar]
- Superparamagnetic core-shells anchored onto graphene oxide grafted with phenylethyl amine as a nano-adsorbent for extraction and enrichment of organophosphorus pesticides from fruit, vegetable and water samples. J. Chromatogr. A. 2015;1406:48-58.
- [CrossRef] [Google Scholar]
- Recognition and selective adsorption of pesticides by superparamagnetic molecularly imprinted polymer nanospheres. RSC Adv.. 2016;6:49401-49410.
- [CrossRef] [Google Scholar]
- Recent advancements in adsorptive removal of organophosphate pesticides from aqueous phase using nanomaterials. J. Nanostruct. Chem. 2022
- [CrossRef] [Google Scholar]
- Fe3O4@ZIF-8@SiO2 core-shell nanoparticles for the removal of pyrethroid insecticides from water. ACS Appl. Nano Mater.. 2023;6:6581-6593.
- [CrossRef] [Google Scholar]
- Occurrence and distribution of UV-filters and other anthropogenic contaminants in coastal surface water, sediment, and coral tissue from Hawaii. Sci. Total Environ.. 2019;670:398-410.
- [CrossRef] [Google Scholar]
- Pesticides in aquatic environments and their removal by adsorption methods. Chemosphere. 2020;253:126646
- [CrossRef] [Google Scholar]
- Adsorptive removal of pesticides from water with metal–organic framework-based materials. Chem. Eng. J.. 2021;421:129688
- [CrossRef] [Google Scholar]
- Synthesis of silica materials containing cyclodextrin and their applications in wastewater treatment. Environ. Chem. Lett.. 2018;17:683-696.
- [CrossRef] [Google Scholar]
- Simultaneous adsorption of cobalt ions, azo dye, and imidacloprid pesticide on the magnetic chitosan/activated carbon@UiO-66 bio-nanocomposite: Optimization, mechanisms, regeneration, and application. Sep. Purif. Technol.. 2022;284:120258
- [CrossRef] [Google Scholar]
- Bioadsorbent from magnetic activated carbon hybrid for removal of dye and pesticide. ChemistrySelect. 2020;5:8814-8822.
- [CrossRef] [Google Scholar]
- Methodology development on aquatic environmental assessment. Environ. Sci. Pollut. Res. Int.. 2017;24:11126-11140.
- [CrossRef] [Google Scholar]
- Perovskite oxide-based photocatalysts for solar-driven hydrogen production: Progress and perspectives. Sol. Energy. 2020;211:584-599.
- [CrossRef] [Google Scholar]
- Biochar-based engineered composites for sorptive decontamination of water: A review. Chem. Eng. J.. 2019;372:536-550.
- [CrossRef] [Google Scholar]
- Fluffy ball-like magnetic covalent organic frameworks for adsorption and removal of organothiophosphate pesticides. Sci. Total Environ.. 2022;840:156529
- [CrossRef] [Google Scholar]
- Modified magnetic chitosan nanoparticles based on mixed hemimicelle of sodium dodecyl sulfate for enhanced removal and trace determination of three organophosphorus pesticides from natural waters. Anal. Chim. Acta. 2019;1078:90-100.
- [CrossRef] [Google Scholar]
- Metal-organic frameworks in agriculture. ACS Appl. Mater. Interfaces. 2022;14:16983-17007.
- [CrossRef] [Google Scholar]
- Efficient adsorption of endocrine-disrupting pesticides from water with a reusable magnetic covalent organic framework. Micropor. Mesopor. Mater.. 2020;307:110523
- [CrossRef] [Google Scholar]
- Matrix-dispersed magnetic molecularly-imprinted polyaniline for the effective removal of chlorpyrifos pesticide from contaminated water. RSC Adv.. 2021;11:39768-39780.
- [CrossRef] [Google Scholar]
- Removal of heavy metals and organic pollutants from water using dendritic polymers based adsorbents: A critical review. Sep. Purif. Technol.. 2018;191:400-423.
- [CrossRef] [Google Scholar]
- Polymer hydrogels and their applications toward sorptive removal of potential aqueous pollutants. Polym. Rev.. 2019;59:418-464.
- [CrossRef] [Google Scholar]
- Polyaniline-based adsorbents for aqueous pollutants removal: A review. Chem. Eng. J.. 2021;418:129425
- [CrossRef] [Google Scholar]
- Modeling of removal of an organophosphorus pesticide from aqueous solution by amagnetic metal–organic framework composite. Chin. J. Chem. Eng.. 2021;40:323-335.
- [CrossRef] [Google Scholar]
- The post-modification of magnetic metal–organic frameworks with β-cyclodextrin for the efficient removal of fungicides from environmental water. Environ. Sci. Nano. 2020;7:2087-2101.
- [CrossRef] [Google Scholar]
- Adsorption behaviors of triclosan by non-biodegradable and biodegradable microplastics: Kinetics and mechanism. Sci. Total Environ.. 2022;842:156832
- [CrossRef] [Google Scholar]
- Toxicity, monitoring and biodegradation of organophosphate pesticides: A review. Crit. Rev. Environ. Sci. Technol.. 2019;49:1135-1187.
- [CrossRef] [Google Scholar]
- Advances in controlled release pesticide formulations: Prospects to safer integrated pest management and sustainable agriculture. J. Hazard. Mater.. 2020;385:121525
- [CrossRef] [Google Scholar]
- Singh NB, Nagpal G, Agrawal S, Rachna. 2018. Water purification by using Adsorbents: A Review. Environmental Technology & Innovation 11, 187-240. 10.1016/j.eti.2018.05.006.
- Design and synthesis of a new magnetic molecularly imprinted polymer nanocomposite for specific adsorption and separation of diazinon insecticides from aqueous media. Microchem. J.. 2022;175:107087
- [CrossRef] [Google Scholar]
- Soltani R, Pelalak R, Pishnamazi M, Marjani A, Shirazian S. 2021. A water-stable functionalized NiCo-LDH/MOF nanocomposite: green synthesis, characterization, and its environmental application for heavy metals adsorption. Arabian Journal of Chemistry 1410.1016/j.arabjc.2021.103052.
- Facile one-pot synthesis of thiol-functionalized mesoporous silica submicrospheres for Tl(I) adsorption: Isotherm, kinetic and thermodynamic studies. J. Hazard. Mater.. 2019;371:146-155.
- [CrossRef] [Google Scholar]
- A hierarchical LDH/MOF nanocomposite: single, simultaneous and consecutive adsorption of a reactive dye and Cr(vi) Dalton Trans.. 2020;49:5323-5335.
- [CrossRef] [Google Scholar]
- Nano-pesticides: A great challenge for biodiversity? Nano Today. 2019;28:100757
- [CrossRef] [Google Scholar]
- Multifunctional Fe(3)O(4)@nSiO(2)@mSiO(2)-Fe core-shell microspheres for highly efficient removal of 1,1,1-trichloro-2,2-bis(4-chlorophenyl)ethane (DDT) from aqueous media. J. Colloid Interface Sci.. 2014;431:90-96.
- [CrossRef] [Google Scholar]
- Low-cost sugarcane bagasse and peanut shell magnetic-composites applied in the removal of carbofuran and iprodione pesticides. Environ. Sci. Pollut. Res. Int.. 2020;27:7872-7885.
- [CrossRef] [Google Scholar]
- Purification of produced water using oscillatory membrane filtration. Desalination. 2020;491:114428
- [CrossRef] [Google Scholar]
- Metal–organic frameworks as media for the catalytic degradation of chemical warfare agents. Coord. Chem. Rev.. 2017;353:159-179.
- [CrossRef] [Google Scholar]
- Adsorption of agricultural wastewater contaminated with antibiotics, pesticides and toxic metals by functionalized magnetic nanoparticles. J. Environ. Chem. Eng.. 2018;6:6468-6478.
- [CrossRef] [Google Scholar]
- Application of a magnetic graphene nanocomposite for organophosphorus pesticide extraction in environmental water samples. J. Chromatogr. A. 2018;1535:9-16.
- [CrossRef] [Google Scholar]
- Facile synthesis of phenyl-modified magnetic graphene/mesoporous silica with hierarchical bridge-pore structure for efficient adsorption of pesticides. Mater. Chem. Phys.. 2017;198:393-400.
- [CrossRef] [Google Scholar]
- Graphene/Fe3O4 nanocomposite for effective removal of ten triazole fungicides from water solution: Tebuconazole as an example for investigation of the adsorption mechanism by experimental and molecular docking study. J. Taiwan Inst. Chem. Eng.. 2019;95:635-642.
- [CrossRef] [Google Scholar]
- Magnetic polydopamine modified with deep eutectic solvent for the magnetic solid-phase extraction of sulfonylurea herbicides in water samples. J. Chromatogr. A. 2019;1601:53-59.
- [CrossRef] [Google Scholar]
- Designing ZIF-8/hydroxylated MWCNT nanocomposites for phosphate adsorption from water: Capability and mechanism. Chem. Eng. J.. 2020;394:124992
- [CrossRef] [Google Scholar]
- Isotherm and kinetic investigations on the adsorption of organophosphorus pesticides on graphene oxide based silica coated magnetic nanoparticles functionalized with 2-phenylethylamine. J. Environ. Chem. Eng.. 2018;6:1333-1346.
- [CrossRef] [Google Scholar]
- High-efficiency adsorption of phenanthrene by Fe3O4-SiO2-dimethoxydiphenylsilane nanocomposite: Experimental and theoretical study. J. Hazard. Mater.. 2022;422:126948
- [CrossRef] [Google Scholar]
- Encapsulation of hydrophobic guests within metal-organic framework capsules for regulating host-guest interaction. Chem. Mater.. 2020;32:3553-3560.
- [CrossRef] [Google Scholar]
- Yang W, Luo W, Sun T, Xu Y, Sun Y. 2022. Adsorption Performance of Cd(II) by Chitosan-Fe(3)O(4)-Modified Fish Bone Char. Int. J. Environ. Res. Public Health 1910.3390/ijerph19031260.
- The simultaneous detection and removal of organophosphorus pesticides by a novel Zr-MOF based smart adsorbent. J. Mater. Chem. A. 2018;6:2184-2192.
- [CrossRef] [Google Scholar]
- Computational study of water adsorption in the hydrophobic metal-organic framework ZIF-8: Adsorption mechanism and acceleration of the simulations. J. Phys. Chem. C. 2017;121:24000-24010.
- [CrossRef] [Google Scholar]
- Magnetic adsorbent based on mesoporous silica nanoparticles for magnetic solid phase extraction of pyrethroid pesticides in water samples. J. Chromatogr. A. 2019;1598:20-29.
- [CrossRef] [Google Scholar]
- Effects of ionic strength on removal of toxic pollutants from aqueous media with multifarious adsorbents: A review. Sci. Total Environ.. 2019;646:265-279.
- [CrossRef] [Google Scholar]
- Sorption of copper and norfloxacin onto humic acid: effects of pH, ionic strength, and foreign ions. Environ. Sci. Pollut. Res. Int.. 2019;26:10685-10694.
- [CrossRef] [Google Scholar]
- Biodegradation and catalytic-chemical degradation strategies to mitigate microplastic pollution. Sustain. Mater. Technol.. 2021;28:e00251.
- [Google Scholar]
- Kinetics and thermodynamics studies of pentachlorophenol adsorption on covalently functionalized Fe3O4@SiO2–MWCNTs core–shell magnetic microspheres. Chem. Eng. J.. 2014;257:10-19.
- [CrossRef] [Google Scholar]







