Translate this page into:
Response surface methodology (RSM) for optimization of gelatin extraction from pangasius fish skin and its utilization for hard capsules
⁎Corresponding author at: Aquatic Product Technology Department, Faculty of Fisheries and Marine Sciences, IPB University, 16680, Bogor, Indonesia. mnurilmala@apps.ipb.ac.id (Mala Nurilmala)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Abstract
This study was aimed to investigate the use of the response surface methodology (RSM) to improve the extraction process of pangasius Pangasianodon hypopthalmus fish skin gelatin. RSM was conducted based on central composite design (CCD) consisting of the two factors, namely, citric acid concentration and extraction time. The yield, moisture content, ash content, pH, viscosity, and setting point were all positively correlated with citric acid concentration, whereas the extraction duration influenced the yield, gel strength, moisture content, and setting point. The combination of these two factors improved the physicochemical properties of the final gelatin preparation. The optimized extraction process was found at the citric acid concentration of 0.3 % and the extraction time of 8 h, producing the gelatin of the quality within the standard GMIA. The gelatin resulted from the optimized processing condition was used as a raw material for hard capsules. The characteristics of the capsules produced were in accordance with those of the commercially available hard capsules.
Keywords
Fish skin
Gelatin
Hard capsule
Pangasius
Process optimization
RSM
1 Introduction
Gelatin is the result of thermal denaturation of collagen from the animal skins, connective tissues, and bones (Nurilmala et al., 2021). The amount of gelatin production worldwide tends to increase every year. Commercial gelatin is mostly derived from the bones and skins of terrestrial animals such as porcine and bovine sources. Bovine gelatin could be contaminated with the pathogens causing diseases such as mad cow disease, foot and mouth disease and Bovine Spongiform Encephalopathy (BSE) (Mad-Ali et al., 2017). In addition, there are limitations of the use of porcine and bovine materials for gelatin from the religious reasons. Muslims are prohibited from eating pork, while Hindus are prohibited from eating beef (Rezaei and Motamedzadegan, 2012).
Gelatin from fish can be an alternative to meet the increasing demand for the halal gelatin. Fishery by-products such as skin can be a source for gelatin (Nurilmala et al., 2020a; Nurilmala et al., 2021). Pangasius (Pangasianodon hypopthalmus) skin is a potential source for sustainable production of gelatin. Pangasius is a freshwater fish having a high commercial value and the prospect of development in the community. Production of the gelatin from pangasius skin has been reported recently (Mahmoodani et al., 2014, Barmon et al., 2020, Nurilmala et al., 2021). However, optimization of the extraction process is still required (Nurilmala et al., 2021).
The quality of gelatin depends on the physicochemical properties which are strongly influenced by the origin of the raw material and the processing method. Fish gelatin is generally less stable and has poorer gelling properties than those from terrestrial animals limiting the application (Mahmoodani et al., 2014). The quality of gelatin is determined by the length of the polypeptide chains. Longer chains with higher molecular weights will produce gelatin with better functional properties and higher molecular weight. Extraction conditions that affect the quality of gelatin include the method of preparation (pretreatment), extraction temperature and time, and the intrinsic properties of collagen. The alkaline pretreatment favors the removal of non-collagenous proteins and lipids (Uranga et al., 2021).
Improving the quality of extracted gelatin can be maximized using response surface methodology (RSM), which is a collection of combined mathematical and statistical techniques that help evaluating processing factors to build a variable model with optimized conditions, and then describe the response with the highest yield (Fan et al., 2017). Differences in the concentration of citric acid and extraction time need to be examined to obtain the optimal conditions for extracting gelatin from pangasius fish skin. It has been reported the optimized extraction conditions of acid concentration and extraction time showed gelatin with the best quality (Mahmoodani et al., 2014).
On the other hand, capsules are used as a shield for drugs, protecting them from environmental influences and help preventing oxidation of the contents of the capsules (Yang et al., 2020). There are two types of capsules, namely hard and soft ones. The hard capsules have a cylindrical shape and consisting of the two parts. The part of short cylindrical shape is called the cap, while the long cylindrical shape is called the body. Whereas, soft capsules consist of only one part. They are usually used for medicines that need to be dissolved in oil or other liquids to help the drug being absorbed in the stomach (Deepak et al., 2014). Capsules that are widely used are hard capsules because they can be used for both solid and liquid drugs (Fauzi et al., 2020).
The objective of this study was to produce gelatin from the pangasius skin. RSM was conducted to determine the optimized processing conditions. Then, the resulted gelatin was applied to the production of hard capsules, and further the physiochemical properties of the capsules obtained were compared with the commercially available ones.
2 Materials and methods
2.1 Skin preparation
The skin from the specimens of fresh pangasius (Pangasianodon hypopthalmus) was cleaned from waste materials such as meat scraps and fat, using a sharp knife. The skin was then washed intensively with tap water. The washed skin was then cut with to a size of about 1 × 1 cm and washed again with tap water as above. The cleaned skin thus prepared was stored in polyethylene bags at −18 °C for until use.
2.2 Gelatin preparation
As a pretreatment, the skin was treated with NaOH, then with citric acid as follows. In the first treatment, the skin was immersed in 0.05 M NaOH solution with a ratio of the skin and the solvent 1:4 (w/v) for 1 h (Nurilmala et al., 2021) followed by washing with tap water. The second treatment was immersion in citric acid with different concentrations (w/v) according to the formula obtained from the running software with an upper limit of 0.3%, the lower limit of 0.1%, with the midpoint of 0.2%. The pretreatment process was carried out for 12 h, and then washed with distilled water until the pH became around 7. The gelatin was extracted with distilled water from the treated skin at 65 °C for different extraction time according to the running software formula with the lower limit of 6 h, the upper limit of 8 h, with the midpoint of 7 h. The ratio of the skin and distilled water for the extraction was 1:1. The treated gelatin was filtered through calico cloth and cotton to produce liquid gelatin, which was then dried using an oven at 50 °C for 24 h. The resulting gelatin was analyzed for the yield, air content, ash content, pH, gel strength, and viscosity.
2.3 Experimental design
Experimental design of gelatin extraction from the pangasius fish skin was carried out by using RSM based on Central Composite Design (CCD). RSM was used to produce product formulations towards the external production factors such as citric acid concentration (X1) and extraction time (X2) as the independent variables. Applications for making a gelatin trial design was done by using the Design Expert software version 10. The independent variables and treatment codes can be seen in Table 1.
Variables
Code
Range and level
-α (-1.414)
−1
0
1
α (1.414)
Acid concentration (%)
X1
0.0585
0.1
0.2
0.3
0.3414
Extraction time (h)
X2
5.58
6
7
8
8.41
The running software resulted in 13 treatments with the independent variables of citric acid concentration and extraction time. The response variables in this study were yield, gel strength, moisture, ash content, pH, viscosity and setting point. The experimental design can be seen in Table 2.
Treatment
Factor codes
Actual factors
X1
X2
Citric acid concentration (%)
Extraction time (h)
1
α (1.414)
0
0.3414
7
2
0
0
0.2
7
3
0
0
0.2
7
4
0
-α (-1.414)
0.2
5.58
5
-α (-1.414)
0
0.0585
7
6
1
1
0.3
8
7
0
α (1.414)
0.2
8.41
8
1
−1
0.3
6
9
−1
1
0.1
8
10
0
0
0.2
7
11
0
0
0.2
7
12
0
0
0.2
7
13
−1
−1
0.1
6
2.4 Preparation of hard capsules
The hard capsules were made from the obtained gelatin. Solid gelatin was mixed with distilled water at the ratio of 1:2 (w/w), then dissolved in a melter at around 85–90 °C. The dissolved gelatin was printed on a capsule mold plate at around 50–60 °C. The pin bar that would be used to be printed on the capsule was smeared in advance with soybean oil. The pin bar was dipped in a plate containing the gelatin solution, and then the wet capsules were removed and dried at around 25–27 °C for approximately 30 min. The dried capsules were then removed from the pin bars. The obtained capsules were analyzed for the acidity degree, moisture content, ash content, capsules cap length and body, dimensions, and disintegration time and and compared with commercial capsules.
2.5 Chemical composition
Chemical composition was determined on pangasius fish skin moisture, ash, fat, and protein contents (AOAC, 2005). The crude protein was measured using a Kjeldahl method, while fat content was determined by a Soxhlet method. However, for the gelatin and capsules, only moisture and ash contents were determined.
2.5.1 Yield
The yield was obtained by calculating the ratio of dry gelatin weight to the weight of skin (raw material) before the gelatin extraction (AOAC, 1995).
2.5.2 Acidity degree
The pH value of gelatin was determined according to Hue et al. (2017) for 1% gelatin (w/v) dissolved in distilled water at around 55–60 ⁰C under constant stirring for 30 min, followed by cooling down to 25 ⁰ C. The solution pH was directly measured using a pH meter (Metrohm, Switzerland).
2.5.3 Gel strength
(GMIA, 2019) Dry gelatin was put into a bloom jar, and dissolved in 100 mL of distilled water to prepare 6.67% (w/v) solution and periodically stirred at 65 °C. The solution was cooled for 15 min at a room temperature and stored at 10 °C for 17 h. Gel strength or bloom was determined using a gelometer (GCA, Grace Instrument, USA).
2.5.4 Viscosity
Gelatin solution of 6.67% (w/v) was put into an Erlenmeyer flask and brought to 100 mL with distilled water. The dissolved gelatin was heated to 61–62 °C. The viscosity (GMIA, 2019) of the solution was measured by using a Brookfield TV-10 digital viscometer (Toki Sangyo, Japan) with spindle No. 1 (Model LV) at 100 rpm and was expressed in centipoises (cP).
2.5.5 Setting point
The setting point was measured as reported (Nurilmala et al., 2020a). A glass cylinder set at 35 °C and a water bath set at 15 °C were used. Briefly, the gelatin (7.5 g) was dissolved in 50 mL distilled water, and heated in a water bath until the solution temperature reached 35 °C. The gelatin solution was put into the tube. The filter paper was inserted in gelatin solution. Then, and this solution was placed into a glass cylinder, which had been submerged in water at 15 °C. The solution was stirred with a thermometer until the gelatin began to harden as indicated by the filter paper which has stopped moving.
2.6 Sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS-PAGE)
According to the method of He et al. (2020), 2 mg gelatin was dissolved in 1 mL of SDS 5% and heated at 85 °C for 1 h, followed by centrifugation for 5 min at room temperature at 12400 rpm, and 20 µL of the supernatants were taken. These were pretreated with SDS at 85 °C for 10 min. As much as 5 µL of the sample solutions were applied to 15% polyacrylamide slab gel and run at 100 V for 3 h. The gel was then soaked in 25 mL of Coomassie Brilliant Blue staining solution for approximately 2 h.
2.7 Fourier transform infrared (FTIR) spectrometry
FTIR analysis was performed according to Dai et al. (2020) to determine the functional groups and presence of the resulting gelatin. Dried gelatin (2 mg) was mashed with KBr powder in a mortar to homogeneity, then put into a pellet mold, compacted and vacuumed in a pellet molding machine. The pellet was put into the cell which was placed in the cell placement chamber and then fired with IR light from an IR-408 infrared spectrophotometer (PerkinElmer, Boston, MA, USA). The histogram was then analyzed to obtain qualitative and quantitative data.
2.8 Capsule weight and dimension measurement
The weight of the capsules was measured using an analytical balance as described in Nurilmala et al. (2020b), while dimensional measurements were carried out for the length and diameter of the capsules using a caliper, after separating the capsule shell into the capsule body and cap.
2.9 Disintegration time
Disintegration time was measured as described in Nurilmala et al. (2020b). Briefly, the capsule shell put in a glass beaker containing 100 mL of distilled water was set at 37 °C for imitating human body temperature. The time for complete solubilization of the capsule shell was defined as the resistance value of the capsule shell in water.
2.10 Statistical analysis
Independent T-test (Ross and Wilson, 2017) was used to analyze the characteristics of capsules from the skin gelatin and commercially available capsules.
3 Results and discussion
3.1 Chemical composition of pangasius skin
The chemical composition is presented in Table 3. The results showed the pangasius skin contained high protein and very low ash, indicating that it is a good source of gelatin. High protein content is needed to obtain higher yield, while low ash content is required to get a good quality of gelatin. The protein content of pangasius skin in this study was slightly lower than those of the two closely related secies, P. sutchi (Mahmoodani et al., 2014) and P. bocourti (Prommajak and Raviyan, 2013). In contrast, the lipid content was higher than those of the two species. High lipid content is undesirable during gelatin production because of resulting in lower quality (Kim et al., 2020). Low contents of ash, lipid and other impurities are important for the quality of gelatins.
Parameter
This experiment
P. sutchi1
P. bocourti2
Moisture (%)
58.67 ± 0.53
50.03 ± 0.27
60.86 ± 0.65
Ash (%)
0.11 ± 0.00
4.14 ± 0.18
0.18 ± 008
Fat (%)
13.44 ± 0.31
6.95 ± 0.17
2.19 ± 0.64
Protein (%)
27.54 ± 0.12
30.91 ± 0.28
35.83 ± 2.61
3.2 Optimization of the gelatin extraction procedures
The results of the characteristics of gelatin based on the experimental design are shown in Table 4. There were 13 gelatin formulas from the Design Expert program version 10 with a central composite design (CCD) that were used in this study. The results of the 13 designs showed different results for each response.
Run
X1
X2
Yield (%)
Gel strength (bloom)
Moisture content (%)
Ash content (%)
pH
Viscosity (cP)
Setting point (°C)
1
0.3414
7
20.5
250.31
6.29
0.97
5.67
63
23.5
2
0.2
7
14
279.37
6.50
0.30
6.51
57
21
3
0.2
7
18
264.73
6.40
0.96
6.84
67
19.5
4
0.2
5.58
12
220.96
5.82
0.39
6.19
68
20
5
0.0585
7
14.5
169.84
6.31
0.96
6.95
53
21
6
0.3
8
18
220.93
7.09
0.96
6.67
63
21
7
0.2
8.41
21
168.36
6.90
0.96
6.76
71
21
8
0.3
6
16.5
203.30
5.96
0.93
5.54
69
24.5
9
0.1
8
16.5
154.79
6.18
0.95
6.75
68
20
10
0.2
7
18
213.04
5.60
0.96
6.60
66
20
11
0.2
7
16
283.81
5.81
0.40
5.65
62
21.5
12
0.2
7
18
259.02
5.47
0.30
6.33
63
20.5
13
0.1
6
12.5
228.57
5.54
0.96
6.76
69
20
The mathematical models for each response are provided in Table 5. The mathematical models for the yield and moisture response followed linear regression, while the gel strength, ash, pH, viscosity and setting point followed quadratic regression. These models were developed after ignoring insignificant variables and were found to be statistically significant with P values < 0.05 and lack of fit value (>0.05).
Response
Equation
P value
Yield
Y = 2.227858X2 + 17.4826X1-2.8697
0.0018
Gel strength
Y = –32.7910X22-2507.8999X12 + 397.0523X2-403.1971X1-1051.5923
0.0151
Moisture
Y = 0.4123X2 + 1.6340X1 + 2.92977
0.0274
Ash
Y = 0.1401X22 + 25.8633X12-1.8772X2-0.3625X + 17.8136
0.0444
pH
Y = 2.8500X1X2-0.4370X2-23.8379X1 + 10.3392
0.0080
Viscosity
Y = 3.5010X22-225.0676X12-37.50000X1X2-40.6094X2 + 376.4556X1 + 161.9257
0.0352
Setting point
Y = 86.6632X128.7500X1X2 + 1.4718X2 + 37.8820X1 + 11.4170
0.0120
3.3 Yield
Yield is a very important factor for gelatin production industry for economic reason. The model equation indicated that the yield was positively correlated with the citric acid concentration and extraction time. The 3D surface plot showed the effect of extraction time and citric acid concentration on gelatin yield (Fig. 1A). The extraction yield increased sharply when the extraction time was prolonged from 6 h to 8 h and the citric acid concentration increased from 0.1% to 0.3%. It is well understood that acid could breakdown collagen structure into subunit chains like α and β as well as high molecular weight components. When the extraction time was prolonged, there will be more breakdown of collagen and increase in the gelatin yield. The yield of gelatin produced in this study varied between 12% and 21%. Using modeling calculation, the maximum yield was 20.54% at the citric acid concentration of 0.3% and extraction time of 8 h, though the actual experimental yield was 22.50%. The yield was higher than those of blackspotted croaker (Protonibea diacanthus) (19.19%), tuna (Thunnus thynnus) (18.1%) (Haddar et al., 2011), Nile tilapia (18.1%) (Songchotikunpan et al., 2008), Atlantic salmon (Salmo salar) (11.3%) and Atlantic cod (Gadus morhua) (10.1%) (Arnesen and Gildberg, 2007). In addition to the extrinsic factors such as extraction time, pH, and temperature, the variation of gelatin yield is also caused by the intrinsic factors including types of fish, collagen content, fish age, and organ sources.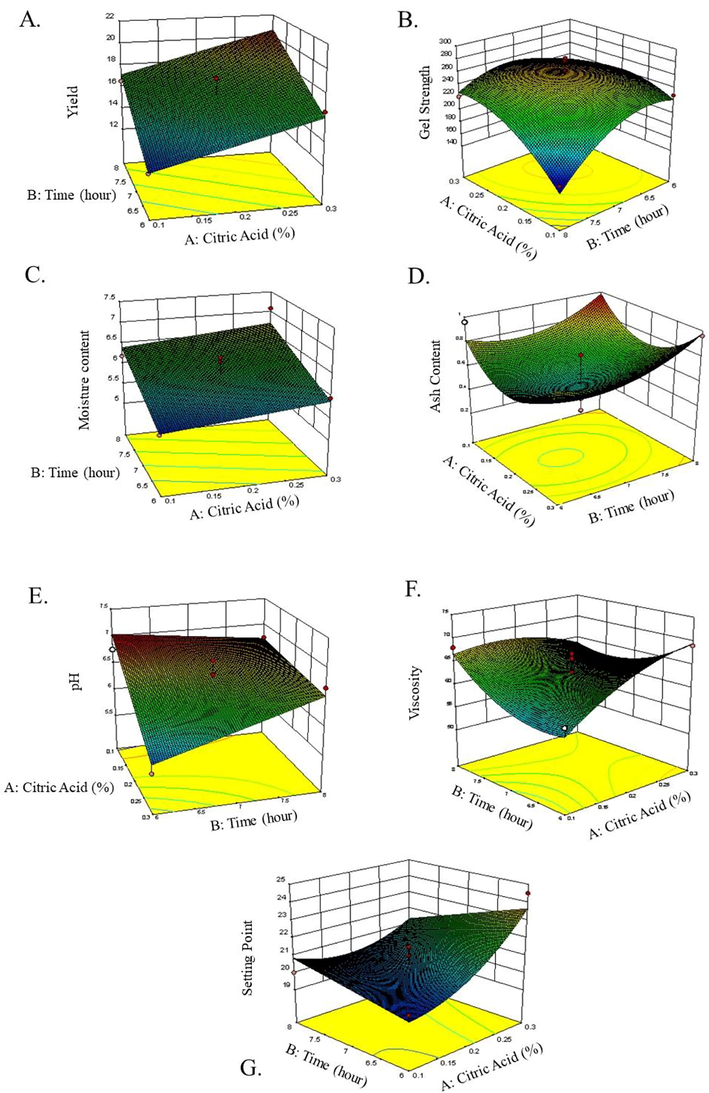
Response surface plots of the effects of citric acid concentration and extraction time on yield (A), gel strength (B), moisture (C), ash (D), pH (E), viscosity (F), and setting point (G).
3.4 Gel strength
Gel strength is a measure of the hardness, stiffness, strength, and compressibility of the gel at a certain temperature and is also influenced by concentration and molecular weight (Irwandi et al., 2009). The strength of the gelatin gel in this study was 154.79–283.81 bloom. The model equation showed that gel strength was positively correlated with the extraction time but negatively correlated with the citric acid concentration, the quadratic effect of extraction time, and the quadratic effect of citric acid concentration. Surface contour images of the gel strength response can be seen in Fig. 1B. The highest gel strength was found in the citric acid concentration of 0.25% with an extraction time for 7 h. The gel strength of pangasius skin gelatin was higher than those of perch (Monsur et al., 2014) and unicorn leatherjacket (Aluterus monoceros) counterparts (Hanjabam et al., 2015). Gel strength is the most important attribute of gelatin and determines the quality of produced gelatin. The application of gelatin was determined by the range of gel strength values. Usually, the gel strength of commercial gelatins ranges from 100 to 300 bloom, but gelatins with gel strength of 250–260 bloom are the most desirable and suitable for a wide range of applications in the food industry especially in the processing of jellies, canned meat, marshmallows and yoghurts. Some species of warm-water fish gelatins have been reported to exhibit high gel strengths, close to that of porcine gelatin, while the gelatin from cold water fish species have poorer physical properties. This is due to a lower content of the amino acids, proline and hydroxyproline in the gelatin from cold water fish species. Factors that affect gel strength are differences in the contents of proline and hydroxyproline as well as the distribution of molecular weight and the type of pretreatments such as the concentration of acid used (Hue et al., 2017).
3.5 Moisture content
Moisture content is an important parameter for determining acceptability, appearance, texture, quality of materials, and durability of materials. The moisture content of gelatin obtained in this study was in range of 5.47–7.08%. The model equation showed that the moisture content is positively correlated with the concentration of citric acid and the extraction time. Surface contour of the gelatin moisture content can be seen in Fig. 1C. The highest moisture content was found in the citric acid concentration of 0.3% with an extraction time of 8 h. The lowest moisture content was in the blue area or the area with 0.1% citric acid with an extraction time of 6 h. The difference in moisture content may be influenced by the drying process using an oven and drying time.
3.6 Ash content
The model equation showed that the response of ash content was positively correlated with the quadratic effect of the citric acid concentration, the quadratic effect of the extraction time but was negatively correlated with the linear effect of citric acid concentration, reducing the linear effect of extraction time. The surface contours and 3D surfaces in Fig. 1D showed that the highest ash content was in the red color or in the citric acid area with a concentration of 0.1% with an extraction time of 6 and 8 h and a citric acid concentration of 0.3% with the extraction time of 6 and 8 h. The lowest ash content was shown in the blue area or the area with 0.2% citric acid with extraction time of 6.5–7 h.
3.7 pH
Measurement of the pH value of the gelatin solution is important, because the pH of the gelatin solution affects other gelatin properties such as viscosity, gel strength, and also affects the application of gelatin in the product. The pH of the gelatin produced in this study was 5.54–6.95%. Different values of acid strength effect on the number of H+ ions that is bound to proteins, then contributing the acidity level of the gelatin produced. The model equation showed that the pH response is positively correlated with the interaction of citric acid concentration and extraction time, but is negatively correlated with the linear effect of citric acid concentration as well as extraction time. Surface contours of pH can be seen in Fig. 1E, which shows that the highest pH is found in the red color or in the citric acid region with a concentration of 0.1% with an extraction time of 8 h. The lowest pH was in the blue area or the area with 0.3% citric acid with an extraction time of 6 h.
3.8 Viscosity
Viscosity is the second most important physical parameter for gelatin. The gelatin viscosity produced in this study was 53–71 mPa-s (cP). The model equation showed that the viscosity response value was positively correlated with the linear effect of citric acid concentration and the quadratic effect of the extraction time concentration, but was negatively correlated with the linear effect of extraction time, the interaction effect of citric acid concentration and extraction time, and the quadratic effect of citric acid concentration. Surface contours of viscosity can be seen in Fig. 1F, which showed that the highest viscosity was found in the red color or in the citric acid region with a concentration of 0.3% with an extraction time of 6 h. The lowest viscosity was shown in the blue area or the area with 0.1% citric acid with an extraction time of 6 h.
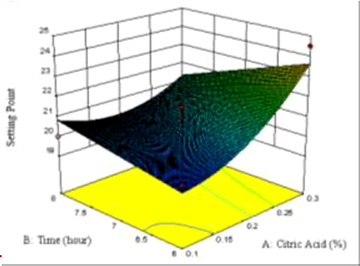
3.9 Setting point
The setting point is the temperature required when the gelatin solution in a certain concentration forms a gel. The setting point of gelatin in this study was 19.5–24.5 °C. The equation of the model showed that the setting point is positively correlated with the quadratic effect of the extraction citric acid concentration and the linear effect of extraction time, but was negatively correlated with the linear effect of the citric acid concentration and the interaction of citric acid concentration and extraction time. The surface contours of viscosity is presented in Fig. 1G. The highest setting point was found in the red color or in the citric acid area with a concentration of 0.3% with an extraction time of 6 h. The lowest setting point was shown in the blue area or the area with 0.1% citric acid with an extraction time of 6 h. The difference in setting point can be caused by differences in the contents of glycine and hydroxyproline in the gelatin, which resulting in the loss of hydrogen bonds in the gelatin solution. In this study, glycine conten was 30.17%, while hydroxyproline was 1.33% (data not shown). The setting point is also influenced by the concentration of gelatin in solution, pH, and molecular weight (Mad-Ali et al., 2017).
3.10 Optimal state validation
Optimal conditions were found at 0.3 % citric acid concentration and 8 h of extraction time based on selection from all response variables of gelatin. Validation was carried out by comparing the optimal prediction results given by the software with the actual value. The optimal prediction value given by the software will be followed by a 95% confidence interval (CI). The comparison of the actual response value with the predicted response value is shown in Table 6. Note: CI = Confidence Interval.
Response
Actual values
Predicted values
95% CI
Low
High
Yield (%)
22.50 ± 0.00
20.54
18.52
22.57
Gel strength (bloom)
191.85 ± 8.19
228.21
185.28
271.15
Moisture content (%)
5.19 ± 0.46
6.71
6.22
7.20
Ash content (%)
0.79 ± 0.05
0.96
0.69
1.23
pH
6.07 ± 0.16
6.53
6.07
6.99
Viscosity (mps)
62 ± 3.21
63.97
58.02
69.93
Setting point (°C)
20.67 ± 0.33
21.29
19.80
22.78
The actual value of the yield response was greater than the predicted value, but was still in the 95% CI scale. The actual value of the response gel strength, ash content, pH, viscosity, and setting point were smaller than the respective predicted values. The results obtained were still within the 95% CI scale. The actual value of moisture content was smaller than the predicted value and was not included in the 95% CI scale. The value of moisture obtained still met the standard of GMIA 2019.
3.11 SDS-PAGE and FTIR analysis
SDS-PAGE and FTIR analysis were carried out to determine the optimum condition. The gelatin preparation in this study showed the bands of the subunits, namely, α1, α2, β and γ chains based on the SDS-PAGE patterns (Fig. 2). The molecular weights of these subunits were 82 kDa for α1, 97 kDa for α2, 211 kDa for β, and 276 kDa for γ, respectively. The α1, α2 and β chains were the typical triple helix chains consisting of collagen molecule, while the γ chain forms the double structure of the triple helix. The range of molecular weights corresponds to the molecular weight of gelatin in general. Boran et al. (2010) reported that the range of molecular weights was 80–125 kDa for the α chain, and 160–250 kDa for the β chain.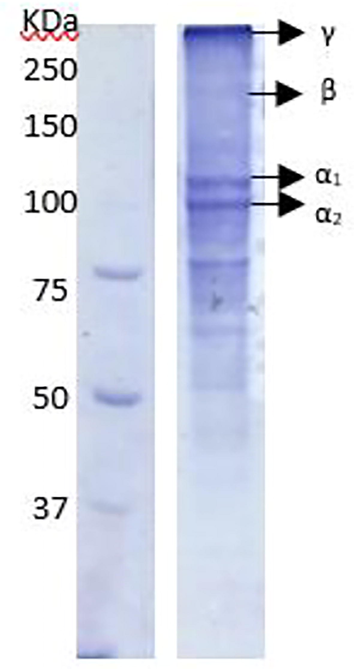
SDS-PAGE pattern of the gelatin preparation.
FTIR analysis was carried out to compare the gelatin preparation of this study with that reported by Maryam et al. (2019). Carbonyl (C = O), amine (NH), and hydroxyl (OH) groups are generally present in the gelatin structure and give rise to five main peaks, namely, amide A (the wavenumber region from 3440 to 3201 cm−1), amide B from 3000 to 2923 cm−1), amide I (from 1698 to 1633 cm−1), amide II (from 1543 to 1447 cm−1) and amide III (the wave number region at 1365 to 1200 cm−1) (Shahvalizadeh et al. 2021; Ashrafi et al. 2023). As shown in Fig. 3, the results of this study showed that there were five peaks, namely, the wavenumbers at 3440; 2933; 1639; 1437 to 1332; 1250 cm−1 were the regions of amide A, amide B, amide I, amide II and amide III, respectively.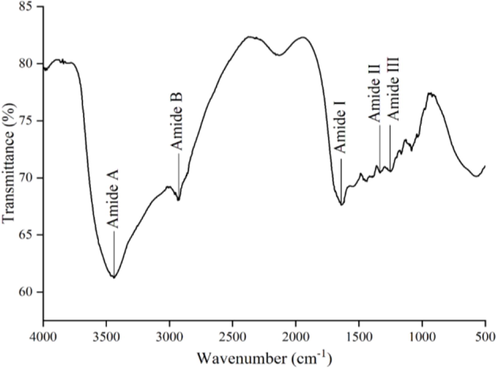
Fourier transform infrared (FTIR) of fish skin gelatin.
3.12 Characteristics of hard capsules
The capsules made in this study were those with size 0. The characteristics of the hard capsules are shown in Table 7. Note: The same superscript letters do not show any significant difference (p > 0.05).
Specification
This study
Commercially available capsules
Body length (mm)
17.88 ± 0.08a
17.90 ± 0.00a
Body diameter (mm)
6.54 ± 0.01a
6.80 ± 0.14a
Cap length (mm)
10.63 ± 0.22a
10.62 ± 0.12a
Diameter cap (mm)
7.05 ± 0.06a
7.27 ± 0.03a
Capsule weight (g)
86.80 ± 2.00a
99.00 ± 1.41b
Moisture (%)
14.58 ± 0.55a
15.35 ± 0.07a
Ash content (%)
0.58 ± 0.28a
1.15 ± 0.20a
Acidity degree (pH)
6.18 ± 0.02a
5.75 ± 0.10b
Disintegration time (min)
1.46 ± 0.00a
5.90 ± 0.28a
The gelatin capsules made from pangasius skin gave the characteristics that were in accordance with the commercial capsules based on the dimensions, weight, moisture content, ash content, pH, and disintegration time. The capsules made from fish gelatin were found to have similar properties as the commercial capsules. Fig. 4 shows the process of production pangasius fish skin gelatin using RSM and the application for hard capsules.
Preparation procedures of skin gelatin and hard capsules.
4 Conclusion
Pangasius fish skin can be made into high quality gelatin with characteristics in accordance with GMIA standard (2019) based on the gel strength, viscosity, acidity (pH), moisture content and ash content. Optimization of the process for producing gelatin from pangasius skin using the RSM method resulted in the optimum response at 0.3% citric acid and extraction time for 8 h for the response of yield, gel strength, moisture, ash content, pH, viscosity and setting point. In addition, the obtained gelatin was found to be the good source for producing hard capsules for Moslem community.
Acknowledgment
The author would like to thank the Indonesia Endowment Fund for Education (LPDP) for the innovative productive research funding grant (RISPRO) for the invitation No. PRJ-76/LPDP/2019 given to Mala Nurilmala. We would also like to thank PT Kapsulindo Nusantara for providing the laboratorium.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- AOAC. 1995. Official Method of Analysis. The Association of Official Analytical Chemist, Inc.
- AOAC. 2005. Official Method of Analysis. The Association of Official Analytical of Chemist, Inc.
- Extraction and characterization of gelatine from Atlantic salmon (Salmo salar) skin. Bioresour. Technol.. 2007;98(1):53-57.
- [CrossRef] [Google Scholar]
- Application of poultry gelatin to enhance the physicochemical, mechanical, and rheological properties of fish gelatin as alternative mammalian gelatin films for food packaging. J. Food.. 2023;12(3):670.
- [CrossRef] [Google Scholar]
- Gelatin extraction from the Bangladeshi pangas catfish (Pangasius pangasius) waste and comparative study of their physicochemical properties with a commercial gelatin. IJETS.. 2020;7(2):13-23.
- [CrossRef] [Google Scholar]
- Effect of extraction condition in the sensory and instrumental characteristics of fish gelatin gels. J. Food Sci.. 2010;75(9):S469-S476.
- [CrossRef] [Google Scholar]
- Effect of interaction between sorbitol and gelatin on gelatin properties and its mechanism under different citric acid concentrations. Food Hydrocoll.. 2020;101(2020):1-9.
- [CrossRef] [Google Scholar]
- Soft gelatin capsules: development, applications, and recent patents. Int. Res. J. Invent. Pharm. Sci.. 2014;2(3):163-173.
- [Google Scholar]
- Extraction of gelatin from salmon (Salmo salar) fish skin using trypsin-aided process: optimization by Plackett-Burman and response surface methodological approaches. J. Food Sci. Technol.. 2017;54(12):4000-4008.
- [CrossRef] [Google Scholar]
- Characterization, disintegration, and dissolution analyses of carrageenan-based hard-shell capsules cross-linked with maltodextrin as a potential alternative drugs delivery system. Int. J. Polym. Sci.. 2020;1(3):1-7.
- [CrossRef] [Google Scholar]
- GMIA.,2019. Gelatin Handbook. Gelatin Manufactures Institute of America
- Physicochemical and functional properties of gelatin from tuna (Thunnus thynnus) head bones. J. Food Nutr. Res.. 2011;50(3):150-159.
- [Google Scholar]
- Optimisation of gelatin extraction from Unicorn leatherjacket (Aluterus monoceros) skin waste: response surface approach. J. Food Sci. Technol.. 2015;52(2):976-983.
- [CrossRef] [Google Scholar]
- A new method of gelatin modified collagen and viscoelastic study of gelatin-collagen composite hydrogel. Macromol. Res.. 2020;28:861-868.
- [CrossRef] [Google Scholar]
- Physicochemical characterization of gelatin extracted from European perch (Perca fluviatilis) and Volga pikeperch (Sander volgensis) skins. Turkish J. Fish. Aquat. Sci.. 2017;17(6):1117-1125.
- [CrossRef] [Google Scholar]
- Extraction and characterization of gelatin from different marine fish species in Malaysia. Int. Food Res. J.. 2009;16:381-389.
- [Google Scholar]
- Extraction of crude gelatin from duck skin: effects of heating methods on gelatin yield. Poult. Sci.. 2020;99(1):590-596.
- [CrossRef] [Google Scholar]
- Characteristics and gelling properties of gelatin from goat skin as affected by drying methods. J. Food Sci. Technol.. 2017;54(6):1646-1654.
- [CrossRef] [Google Scholar]
- Optimization of extraction and physicochemical properties of gelatin from pangasius catfish (Pangasius sutchi) skin. Sains Malays.. 2014;43(7):995-1002.
- [Google Scholar]
- Effects of pretreatment on properties of gelatin from perch (Lates Niloticus) skin. Int. J. Food Prop.. 2014;17(6):1224-1236.
- [CrossRef] [Google Scholar]
- Characterization and antioxidant activity of collagen, gelatin, and the derived peptides from yellowfin tuna (Thunnus albacares) skin. Mar. Drugs.. 2020;18(2):98.
- [CrossRef] [Google Scholar]
- Evaluation of the properties of tuna skin gelatin as a hard capsule material. Fish Sci.. 2020;86:917-924.
- [CrossRef] [Google Scholar]
- Pangasius fish skin and swim bladder as gelatin sources for hard capsule material. Int. J. Biomater.. 2021;2021:1-6.
- [CrossRef] [Google Scholar]
- Physical properties of gelatin extracted from skin of Thai panga fish (Pangasius bocourti Sauvage) Food Appl. Biosci. J.. 2013;1(3):131-145.
- [Google Scholar]
- The effect of processing conditions on the extraction yield and melting point of whitecheek shark (Carcharhinus dussumieri) skin gelatin. IFSTRJ. 2012;8(2):150-157.
- [CrossRef] [Google Scholar]
- Ross, A., Willson, V.L., 2017. Independent Samples T-Test. In: Basic and advanced statistical tests. SensePublishers, Rotterdam. Doi: 10.1007/978-94-6351-086-8_3.
- Shahvalizadeh, R., Ahmadi, R., Davandeh, I., Pezeshki, A., Moslemi, S.A.S., Karimi, S., Rahimi, M., Hamishehkar, H., Mohammadi., M., 2021. Antimicrobial bionanocomposite films based on gelatin, tragacanth, and zinc oxide nanoparticles-Microstructural, mechanical, thermo-physical, and barrier properties. Food Chem. 354, 1-11. Doi: 10.1016/j.foodchem.2021.129492
- Extraction and electrospinning of gelatin from fish skin. Int. J. Biol. Macromol.. 2008;42(3):247-255.
- [CrossRef] [Google Scholar]
- Uranga, J., Zarandona, I., Andonegi, M., Guerrero, P., de la Caba, K., 2021. Biopolymers derived from marine sources for food packaging applications, in: Athanassiou, A. (Eds.), Sustainable Food Packaging Technology., Spain, pp. 35–56. Doi: 10.1002/9783527820078.CH2.
- Moisture sorption and desorption properties of gelatin, HPMC, and pullulan hard capsules. Int. J. Biol. Macromol.. 2020;5(1):1-23.
- [CrossRef] [Google Scholar]







