Translate this page into:
SnCl4/γ-Al2O3 nanoparticles: A reusable and green heterogeneous catalyst for the one-pot synthesis of tetrahydropyridines
⁎Corresponding author. nahidyaghma@yahoo.com (Nahid Yaghmaeiyan)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Nano-SnCl4/γ-Al2O3 as a reusable and green heterogeneous catalyst was prepared by the reaction of tin tetrachloride with γ-alumina. This solid acid catalyst was characterized by fourier transform-infrared spectroscopy (FT-IR), X-ray diffraction (XRD), field emission scanning electron microscopy (FESEM), transmission electron microscopy (TEM), energy-dispersive X-ray spectroscopy (EDS), thermal gravimetric analysis (TGA) and Brunauer-Emmett-Teller (BET) analyses. Tetrahydropyridines were synthesized via the one-pot reaction of 4-substitutd anilines, ethylacetoacetate and 4-substituted aldehydes in the presence of optimized amount (0.06 g) of nano-SnCl4/γ-Al2O3 under thermal conditions (70 °C) in ethanol as a green solvent. The process had short reaction times (5–8 h), good to excellent yields (80–93%), simple work-up and easy purification technique (recrystallization through the hot ethanol). It was found that the catalyst could be reused for three times without any considerable loss of its activity.
Keywords
SnCl4/γ-Al2O3
Nanoparticles
Reusable
Heterogeneous
Tetrahydropyridine
One-pot
1 Introduction
Recently, new organic and medicinal compounds are produced through the one-pot processes called multicomponent reactions (MCRs) (Safaei-ghomi and Masoomi, 2015; Bamoniri et al., 2022; Bamoniri et al., 2023; Bamoniri et al., 2014). By using simple materials in a one pot set up, MCR techniques enable the generation novel and complex molecular structures with minimal synthetic steps. MCRs are environmentally friendly and effectual for the synthesis of heterocyclic compounds (Geetha et al., 2020; Bharti and Parvin, 1806). The advantages of this class of reactions include low response time, high efficiency, clean work-up and easy purification (Bamoniri and Yaghmaeiyan, 2022; Bamoniri et al., 2023; Domling, 2006; Bamoniri et al., 2018; Bamoniri et al., 2019; Yaghmaeiyan et al., 2023).
The atom economy of a reaction is a theoretical measure of the amount of starting material that ends up as the desired final product which can be maximized by use of a suitable green catalyst. Atom economy is the most widely used metrics to measure the greenness of a reaction (Ostovan et al., 2022). In the 21st century, green and sustainable practices have become a critical issue. The deleterious environmental side effects results a senseof responsibility toward environment providing the motivation to follow green approachments such as the prohibition of the consumption of toxic organic solvents and using the heterogeneous catalysts in organic syntheses or compliance with the principles of green chemistry by drug companies (Arabi et al., 2021). Thus, green chemistry has improved and its principles have been used in the synthesis, analytical chemistry, engineering, and pharmaceutical science (Arabi et al., 2022).
Tetrahydropyridines (THPs) are a category of six-membered nitrogen-bearing heterocyclic compounds classified as piperidine or pyridine derivatives. THPs are formed the central core of many natural compounds and medicinal products (Viegas et al., 2004; Hayes et al., 2001; Urushima et al., 2010; Marson, 2012; Cuthbertson and Taylor, 2013; Viegas et al., 2004; Watson et al., 2000). Optimization of the structure of THPs has been widely extended to produce new molecules such as GTS-21 for Alzheimer's disease (Kitagawa et al., 2003) and RO10-5824 for central nervous system disorders (Tancredi et al., 2008). THPs have received a lot of attention due to their medicinal properties such as antihypertensive (Petit et al., 1991), antibacterial (Zhou et al., 2007), analgesic (Madhavi and Redda, 2006), antihyperglycemic (Yeung et al., 1982), antimalarial (Misra et al., 2009; Aeluri et al., 2012), antiemetic (Domino et al., 1999; Mashkovskii and Glushkov, 2001), sedative (Gessner et al., 1985) and anti-cancer (Takasu et al., 2006) activities. Several methods based on cyclization reactions (Sales and Charette, 2005; Zhu et al., 2003; Carballo et al., 2006; Dobbs and Guesne, 2005; Fustero et al., 2007; Davis et al., 2001; Sajadikhah et al., 2012) have been reported for the preparation of THPs and various catalysts including dihydrate oxalic acid (Shi et al., 2013), chiral phosphoric acids (CPAs) (Hazeri et al., 2013), Fe(NO3)3·9H2O (Safaei-Ghomi and Ziarati, 2013), FeCl3/SiO2 NPs (Harichandran et al., 2013), IRA400-Cl Resin/I2/KI Amberlite (Zhang et al., 2014) and SSA (Mirjalili et al., 2013) have been used for the synthesis of them. However, some of these methods have disadvantages such as harsh reaction conditions, difficult processing, possibility of side reactions, high cost of the catalyst, and most importantly, production of acidic and toxic effluents, which is against the principles of green chemistry.
Tin tetrachloride is highly volatile, corrosive, and difficult to handle. In the presence of moisture, it is hydrolyzed and produce HCl (Mukhopadhyay et al., 2011). In this study nano-SnCl4/γ-Al2O3 prepared as a mild solid lewis acid and used for the synthesis of THPs. In a typical procedure, a mixture of 4-substitutd anilines (2 mmol), ethylacetoacetate (1 mmol) and 4-substituted aldehydes (2 mmol) are condensed in the presence of optimized amount of nano-SnCl4/γ-Al2O3 at 70 °C in ethanol as a green solvent (Scheme 1).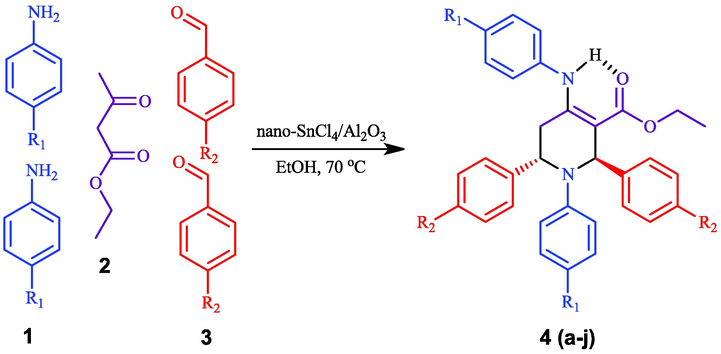
Synthesis of THP derivatives in the presence of nano-SnCl4/γ-Al2O3.
2 Experimental
2.1 Preparation of nano-SnCl4/γ-Al2O3
25 ml of SnCl4 was added drop-wise to a suspension of 5 g of nano-γ-Al2O3 in 50 ml dichloromethane. The mixture stirred at room temperature for 1 h. Then, it filtered and the white solid was separated, washed with dichloromethane and dried at room temperature (Scheme 2). Nano-SnCl4/γ-Al2O3 was obtained in 98% yield and characterized by FT-IR, XRD, FESEM, TEM, EDS. BET and TGA analyses.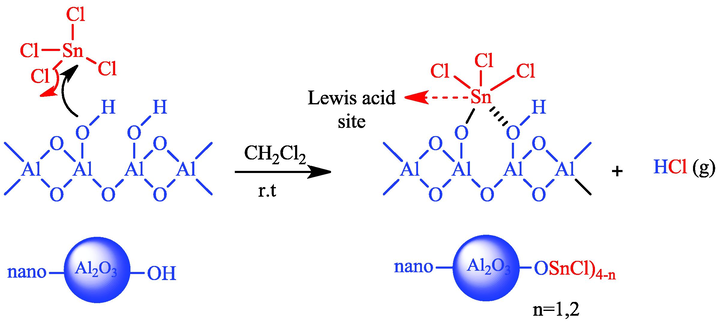
A proposed structure for nano SnCl4/γ-Al2O3.
2.2 Preparation of THPs
A mixture of 4-substitutd anilines (2 mmol), ethylacetoacetate (1 mmol) and 4-substituted aldehydes (2 mmol) are condensed in the presence of optimized amount of nano-SnCl4/γ-Al2O3 (0.06 g) as a solid acid catalyst, at 70 °C, in ethanol as the green solvent of the reaction. After completion of the reaction, as specified by TLC (EtOAc: petroleum ether 3: 7), the catalyst was separated by filtration. The pure product was obtained after evaporation of the solvent from the filtrate and then, recrystallization through the hot ethanol. The results of the synthesis of THPs by nano-SnCl4/γ-Al2O3 are summarized in Table 1.
Entry
R1
R2
Products
Time (h)
Yield (%)
MP (°C)
Ref.
1
H
Me
4a
6
80
230–232
(Sajadikhah et al., 2012)
2
H
Cl
4b
6
80
201–203
(Atrak et al., 2018)
3
H
Br
4c
5
85
218–220
(Atrak et al., 2018)
4
Me
H
4d
5
88
192–194
(Saniger, 1995)
5
Me
Me
4e
6
90
169–171
(Saniger, 1995)
6
Br
H
4f
5
85
200–202
(Saniger, 1995)
7
Br
Cl
4 g
8
81
194–196
(Atrak et al., 2018)
a8
Cl
H
4 h
5
93,89,82,75
197–199
(Malik et al., 2015)
9
Cl
Cl
4i
8
90
212–214
(Lashkari et al., 2013)
10
Br
OMe
4j
5
91
199–201
(Saniger, 1995)
The stability, regeneration, and reusability of the nano-SnCl4/γ-Al2O3 were investigated. The catalyst was recovered after the reaction 8 for three runs. After completion of the reaction for the synthesis of 4 h, the catalyst was separated by simple filtration, washed with dichloromethane (3 × 10 ml) and dried at room temperature without further purification to use for the next run in current reaction under equal conditions. It was found that the catalyst could be reused for 3 times without any considerable loss of its activity (Fig. 1). The structure of the catalyst is not changed after each reaction but the active surface and acidic sites of the catalyst is decreased little by little, due to the contact with the reactants.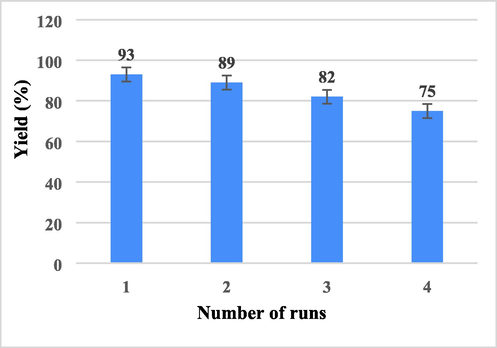
Reusability diagram of nano-SnCl4/γ-Al2O3.
2.3 Identification of nano-SnCl4/γ-Al2O3
ATR spectra of nano-γ-Al2O3 and nano-SnCl4/γ-Al2O3 were compared from 613 to 4000 cm−1 (Fig. 2a-b). In Fig. 2a, the absorption peak at 1630 cm−1 for γ-Al2O3 is ascribed to the bending vibrations of adsorbed water molecules (Wang et al., 2011). The strong broad band centered at 613 cm−1 is coincident with the expected asymmetric vibration frequency of the condensed Al-O groups (Xiao et al., 2014). In FT-IR spectrum of nano-SnCl4/γ-Al2O3 (Fig. 2b), the broad peaks at 3606 and 2971 cm−1 is related to the stretching vibration of hydroxyl groups confirmed by weak band at about 1612 cm−1. This band is caused by the bending vibration of coordinated H2O as well as Sn–OH. The peak at 1098 cm−1 corresponds to the (Sn–OH) vibrations (Bamoniri et al., 2015). Thus, the FT-IR spectrum confirmed the presence of Sn–O bonds and OH groups in nano-SnCl4/γ-Al2O3.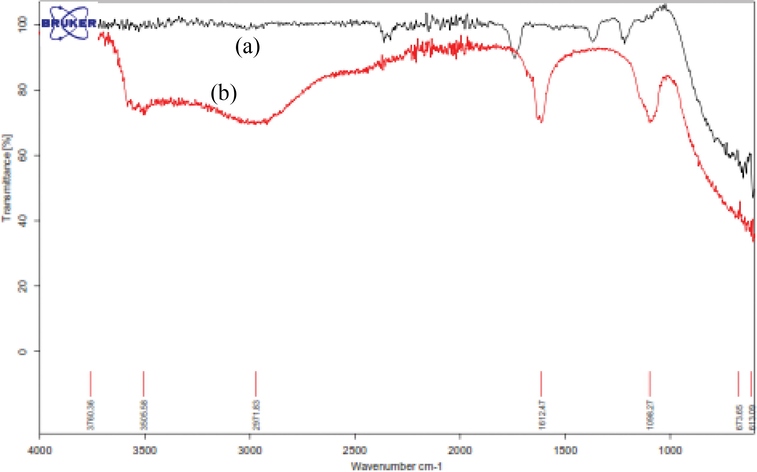
ATR spectra of (a) nano-γ-Al2O3 and (b) nano-SnCl4/γ-Al2O3.
TG-DTG pattern of nano-SnCl4/γ-Al2O3 are presented from 0 to 800 °C (Fig. 3). As can be seen in this pattern, the catalyst is stable until 280 °C. Three ranges are detectable in this pattern. The processes between 0 and 120 °C caused mass changed equal to 6% which attributed to the moisture removal. The catalyst was lost its weight equal to 14% in the range of 120–280 °C. It is an evidence of the SnCln breakdown and the formation of γ-alumina which is stable until 800 °C. In the range of 280–800 °C the mass change in only 3.5% attributed to the moisture removal of γ-alumina surface.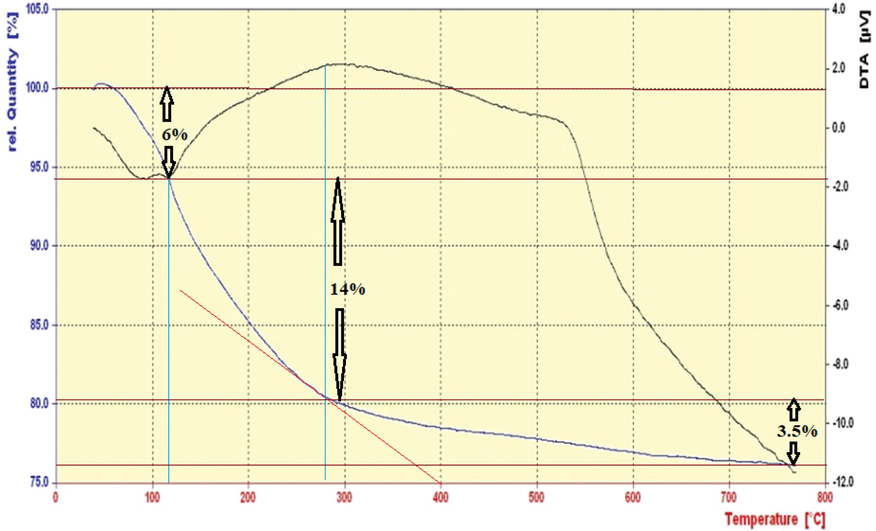
TGA-DTG pattern of nano-SnCl4/γ-Al2O3.
The XRD results of the catalyst in Fig. 4 show the diffraction pattern of nano-SnCl4/γ-Al2O3 peaks at (1 0 1), (3 1 1), (4 0 0) and (4 4 0), at 2θ diffraction angle 32.00, 37.6, 45.66 and 67.20, consecutively. As shown in this figure, three peaks in 2θ = 37.6, 45.66 and 67.20, correspond to the structure of γ-Al2O3. The sharpest peak in 2θ = 32.00 correspond to Sn-O band of nano-SnCl4/γ-Al2O3. The appearance of the peaks which is disturbed and broad shows the amorphous nature of the structure. Using the Scherrer equation show that the average size of nanoparticles is about 35.67 nm.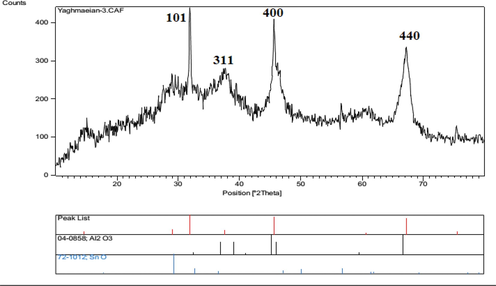
XRD pattern of nano-SnCl4/γ-Al2O3.
In order to obtain the percentage of elements of the catalyst, the EDS analysis of nano-SnCl4/γ-Al2O3 nanoparticles was measured by EDS instrument (Fig. 5). According to this data, the weight percentage of O, Al, Sn and Cl are 59.2, 28.5, 6.6 and 5.7, respectively. The presence of Sn and Cl demonstrate that SnCl4 has reacted readily with /γ-Al2O3. This data are verified the structure of nano-SnCl4/γ-Al2O3.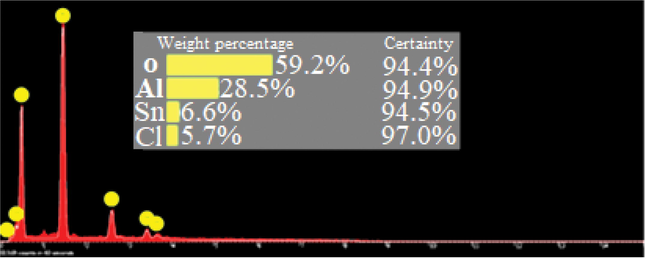
The EDS analysis of nano-SnCl4/γ-Al2O3.
FESEM and TEM images of nano-SnCl4/γ-Al2O3 are displayed in Fig. 6 (a-b). These images are clearly shown the amorphous surface morphology of the nano-SnCl4/γ-Al2O3 with an average size distribution of 35 nm. The micrographs of nano-SnCl4/γ-Al2O3 clearly show highly porous morphology of it. As can be seen from Fig. 6a, the holes in nano-SnCl4/γ-Al2O3 structure makes it a convenient bed for the conversion of bulky molecules. In Fig. 6b, we see a collection of small particles with nearly spherical shape creates big amorphous and non-uniform particles in nano-SnCl4/γ-Al2O3.
(a) FESEM and (b) TEM photographs of nano-SnCl4/γ-Al2O3.
Specific surface area of catalyst was measured by BET theory. The single point surface area at P/P0 = 0.990 is 141.78 m2/g, while the average pore diameter is 11.187 nm and the total pore volume is 0.3965 cm3 g−1. The BET, BJH and adsorption/desorption isotherm plots of nano-SnCl4/γ-Al2O3 are shown in Fig. 7 (a-c).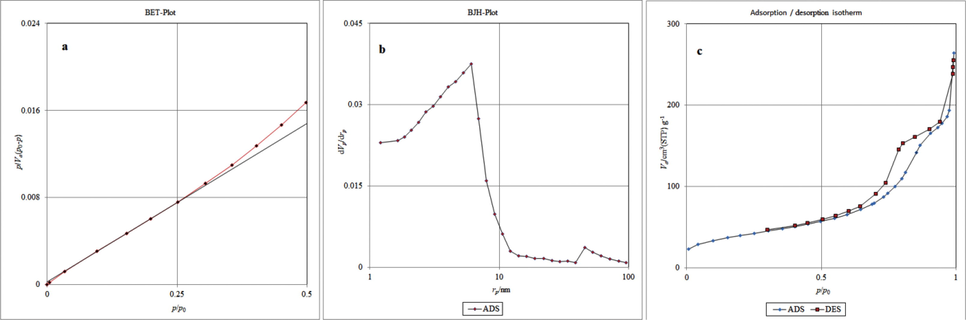
(a) BET (Brunauer–Emmett–Teller), (b) BJH (Barrett-Joyner-Halenda) and (c) adsorption/desorption isotherm plots of nano-SnCl4/γ-Al2O3.
2.4 Optimization of the reaction conditions
Parameters affected on the catalytic activity were investigated and optimized during the synthesis step. Synthesis of 4j was repeated for 13 times in different conditions (by changing the solvent, temperature and amount of the catalyst) and the yield of the reaction was evaluated. The results are summarized in Table 2. As can be seen in this table, the reaction efficiency in the presence of nano-SnCl4/γ-Al2O3, mainly depends on the amount of the catalyst, temperature and type of the solvent. After optimization of these parameters, the best yield was observed with 0.06 g of nano-SnCl4/γ-Al2O3 at 70 °C in ethanol as the green solvent (Table 2, Entry 12). The studies on the effect of catalyst in the reaction was showed that in the absence of catalyst the efficiency was reduced to 10%, after 48 h under solvent-free conditions (Table 2, Entry 1).
Entry
Catalyst (g)
Solvent
Temperature (°C)
Time (h)
Yield (%)
1
Nano SnCl4/γ-Al2O3 (0.00)
Solvent-free
25
48
10
2
Nano SnCl4/γ-Al2O3 (0.01)
Solvent-free
25
48
15
3
Nano SnCl4/γ-Al2O3 (0.01)
CH3CN
25
40
25
4
Nano SnCl4/γ-Al2O3 (0.01)
MeOH
25
35
32
5
Nano SnCl4/γ-Al2O3 (0.01)
EtOH
25
30
40
6
Nano SnCl4/γ-Al2O3 (0.01)
EtOH
50
25
48
7
Nano SnCl4/γ-Al2O3 (0.01)
EtOH
60
20
53
8
Nano SnCl4/γ-Al2O3 (0.01)
EtOH
70
15
58
9
Nano SnCl4/γ-Al2O3 (0.01)
EtOH
80
15
58
10
Nano SnCl4/γ-Al2O3 (0.02)
EtOH
70
13
63
11
Nano SnCl4/γ-Al2O3 (0.04)
EtOH
70
10
78
12
Nano SnCl4/γ-Al2O3 (0.06)
EtOH
70
5
91
13
Nano SnCl4/γ-Al2O3 (0.08)
EtOH
70
5
91
In Table 3, we compared nano- SnCl4/γ-Al2O3 with several catalysts for the synthesis of THPs. As can be seen in this table, nano- SnCl4/γ-Al2O3 is also a highly efficient catalyst under thermal conditions. Except entry 1 which have more mild conditions and shorter reaction time, this work is superior compared to other reported methods.
Entry
Catalyst
Solvent
Conditions
Time (h)
Yield (%)
Ref.
1
AcOH (5 ml)
AcOH
rt
1
92
Lashkari et al., 2013
2
CAN (0.15 mol)
MeCN
rt
35
68
Wang et al., 2011
3
p-TsOH·H2O (0.11 g)
EtOH
rt
12
88
(Malik et al., 2015)
4
SSA (0 0.10 g)
EtOH
60 °C
9
78
Xiao et al., 2014
5
L-proline/TFA (20% mol)
AcCN
20–30 °C
22
75
(Misra et al., 2009)
6
Nano-TiCl4/SiO2 (1/0 g) (0.05)
EtOH
70 °C
7
81
Bamoniri et al., 2015
7
Nano-SnCl4/γ-Al2O3 (0.06)
EtOH
70 °C
5
93
This work
2.5 Mechanism of the reaction
Suggested mechanism for the synthesis of THPs have presented in scheme 3. In this mechanism, at first, the ethylacetoacetate is activated by the solid acid catalyst. Then, nucleophilic attack of amine is occurred to produce intermediate A (Step I). In the next step, the intermediate A condensed with a molecule of aldehyde which activated by the catalyst to produce intermediate B (Step II). In the other hand, the intermediate C produced through the condensation of a molecule of amine with a molecule of aldehyde (Step III). The intermediate D is formed by addition of intermediate B to intermediate C (Step IV). Finally, the product formed in the presence of acid catalyst (Step V). If the reaction is not complete, the intermediates B and C may be formed as the side products along with the main product, but with proper optimization of the reaction conditions in this method, only the main product was obtained without disturbing the side products.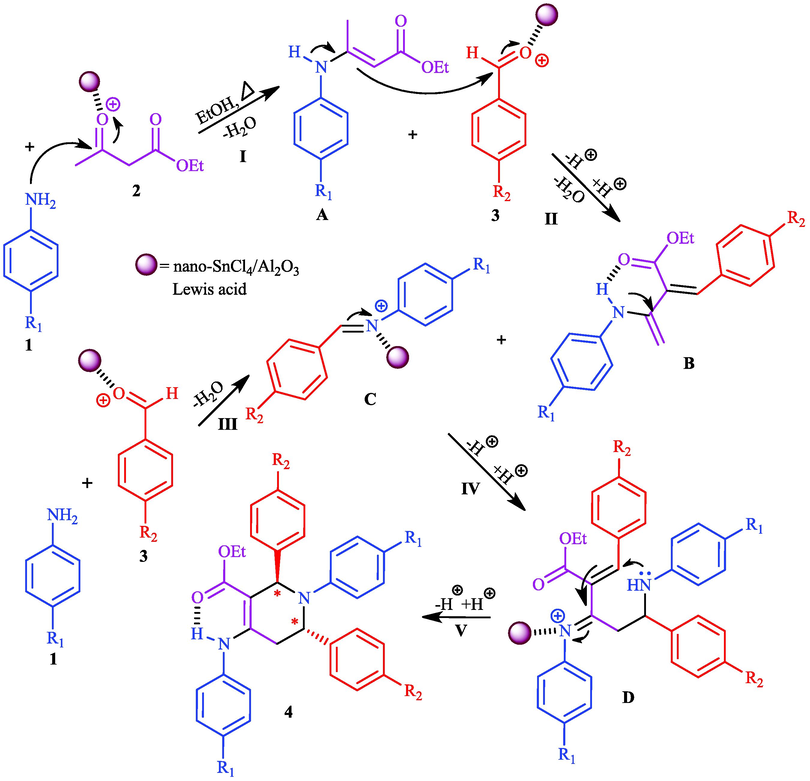
Suggested mechanism for the synthesis of THPs in the presence of nano-SnCl4/γ-Al2O3.
3 Conclusion
In this paper, nano-SnCl4/γ-Al2O3 as a reusable and green heterogeneous catalyst was prepared by the reaction of tin tetrachloride with γ-alumina and characterized by FT-IR, XRD, FESEM, TEM, EDS, BET and TGA teqnigues. We have introduced an efficient and environmentally friendly methodology for the synthesis of tetahydropyridines via condensation of 4-substitutd anilines, ethylacetoacetate and 4-substituted aldehydes using SnCl4/γ-Al2O3 nanoparticles as a solid acid catalyst. This simple procedure have advantages such as easy and clean work up, high yields, reusability of catalyst and heterogeneous conditions.
Acknowledgement
The authors are grateful to University of Kashan, Iran for supporting this work by Grant No. 159189/40.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Asian J. Org. Chem.. 2012;1:71.
- Adv. Mater.. 2021;33:2100543.
- Nat. Commun.. 2022;13:5757.
- J. Mater. Sci.: Mater. Electron.. 2018;29:8347.
- Bulg. Chem. Commun.. 2014;46:79.
- J. Chem. Sci.. 2015;127:885-895.
- Results Chem.. 2022;4:100505
- Iran. J. Catal.. 2018;8:81.
- J. Nanostruct.. 2019;9:219.
- Indian J. Chem. B. 2022;61:599.
- Results Chem.. 2023;5:100870
- Results Chem.. 2023;5:100853
- J. Heterocycle. Chem.. 1806;2015:52.
- Org. Lett.. 2006;8:3837.
- Angew. Chem. Int. Ed.. 2013;52:1490.
- Org. Lett.. 2001;3:3169.
- Synlett 2005:2101.
- Anesth. Analg.. 1999;88:1370.
- Chem. Rev.. 2006;106:17.
- Org. Lett.. 2007;9:5283.
- Chem. Data Collect.. 2020;30:100565
- J. Med. Chem.. 1985;28:311.
- J. Heterocycl. Chem.. 2013;50:539.
- Chem. Commun. 2001:1784.
- J. Chin. Chem. Soc.. 2013;60:355.
- Neuropsychopharmacology. 2003;28:542.
- Synth. Commun.. 2013;43:635.
- J. Heterocycl. Chem.. 2006;43:709.
- Energy Environ. Focus. 2015;4:340.
- Chem. Soc. Rev.. 2012;41:7712.
- Pharm. Chem. J.. 2001;35:179.
- Sci. Iran. 2013;20(3):587.
- Bioorg. Med. Chem.. 2009;17:625.
- Tetrahedron Lett.. 2011;52:5835.
- Adv. Mater.. 2022;34:2203154.
- Eur. J. Med. Chem.. 1991;26:19.
- RSC Adv.. 2015;5:15591.
- J. Iran. Chem. Soc.. 2013;10:135.
- Monatsh. Chem.. 2012;143:939.
- Chin. Chem. Lett.. 2012;23:569.
- Org. Lett.. 2005;7:5773.
- Mater. Lett.. 1995;22:109.
- Adv. Synth. Catal.. 2013;355:1605.
- Tetrahedron. 2006;62:11900.
- Neuropsychopharmacologicum (CINP). 2008;11:293.
- Org. Lett.. 2010;12:4588.
- J. Nat. Prod.. 2004;67:908.
- J. Nat. Prod.. 2004;67:908.
- Sci.. 2011;13:181.
- Org. Lett.. 2000;2:3679.
- Results Chem.. 2023;5:100696
- J. Med. Chem.. 1982;25:720.
- J. Heterocycl. Chem.. 2014;51:442.
- J. Heterocycl. Chem.. 2014;51:442.
- Bioorg. Med. Chem. Lett.. 2007;17:1206.
- J. Am. Chem. Soc.. 2003;125:4716.







